JP2008030979A5 - - Google Patents
Download PDFInfo
- Publication number
- JP2008030979A5 JP2008030979A5 JP2006203888A JP2006203888A JP2008030979A5 JP 2008030979 A5 JP2008030979 A5 JP 2008030979A5 JP 2006203888 A JP2006203888 A JP 2006203888A JP 2006203888 A JP2006203888 A JP 2006203888A JP 2008030979 A5 JP2008030979 A5 JP 2008030979A5
- Authority
- JP
- Japan
- Prior art keywords
- hypochlorous acid
- water
- air
- acid water
- solution
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 18
- QWPPOHNGKGFGJK-UHFFFAOYSA-N Hypochlorous acid Chemical compound ClO QWPPOHNGKGFGJK-UHFFFAOYSA-N 0.000 description 11
- 239000000460 chlorine Substances 0.000 description 7
- 238000004519 manufacturing process Methods 0.000 description 6
- 229910052801 chlorine Inorganic materials 0.000 description 5
- -1 chlorine ions Chemical class 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- ZAMOUSCENKQFHK-UHFFFAOYSA-N chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 3
- 239000000835 fiber Substances 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-N HCl Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 239000004020 conductor Substances 0.000 description 2
- 238000005260 corrosion Methods 0.000 description 2
- 238000007865 diluting Methods 0.000 description 2
- 239000001103 potassium chloride Substances 0.000 description 2
- 235000011164 potassium chloride Nutrition 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000012209 synthetic fiber Substances 0.000 description 2
- 229920002994 synthetic fiber Polymers 0.000 description 2
- 239000004698 Polyethylene (PE) Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 239000003463 adsorbent Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 238000010411 cooking Methods 0.000 description 1
- 239000008151 electrolyte solution Substances 0.000 description 1
- 239000002778 food additive Substances 0.000 description 1
- 235000013373 food additive Nutrition 0.000 description 1
- 239000003365 glass fiber Substances 0.000 description 1
- 239000003595 mist Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 230000001954 sterilising Effects 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Description
次亜塩素酸水は、食品添加物として認められているとともに、給食センター、食品製造プラント、レストランなどの食品調理室などにおける殺菌性洗浄水として利用されている。
次亜塩素酸水の製造は、例えば、食塩、塩化カリウム、塩酸などの水溶液のように塩素イオンを含んだ溶液を直流により電気分解することにより行われる。
そして、次亜塩素酸の生成は、塩素イオン(Cl−)が陽極表面で電解酸化を受けて塩素(Cl2)となり、さらに水と反応し、次亜塩素酸を生成することを利用している。
次亜塩素酸水製造装置としては、特開平9−103786号公報(特許文献1)に開示されているような食塩や塩化カリウムの低濃度溶液を隔膜式電解槽で電解する装置、特開2000−140850号公報(特許文献2)に開示されているような高濃度の塩溶液を連続的に電解し、水で希釈調製する装置、また、特開2000−212786号公報(特許文献3)に開示されているような高濃度の原液を貯留した電解槽を水タンク内に投入して電解し、電解槽内部に生成した電解液をタンク内に混合希釈する装置などが知られている。
Hypochlorous acid water is recognized as a food additive, and is also used as sterilizing washing water in food cooking rooms such as lunch centers, food manufacturing plants, and restaurants.
The production of hypochlorous acid water is performed, for example, by electrolyzing a solution containing chlorine ions with a direct current, such as an aqueous solution of sodium chloride, potassium chloride, hydrochloric acid or the like.
Hypochlorous acid is produced using the fact that chlorine ions (Cl − ) undergo electrolytic oxidation on the anode surface to become chlorine (Cl 2 ), and further react with water to produce hypochlorous acid. Yes.
As an apparatus for producing hypochlorous acid water, an apparatus for electrolyzing a low concentration solution of sodium chloride or potassium chloride in a diaphragm type electrolytic cell as disclosed in JP-A-9-103786 (Patent Document 1), JP-A 2000 -140850 (patent document 2) disclosed in Japanese Patent Application Laid-Open No. 2000-212786 (patent document 3), a device for continuously electrolyzing a salt solution of high concentration and diluting with water. There is known an apparatus for electrolyzing an electrolytic cell storing a stock solution having a high concentration as disclosed in a water tank, and mixing and diluting the electrolytic solution generated inside the electrolytic cell in the tank.
この実施例において用いられる空気供給器3は、次亜塩素酸水製造用ガス発生器2の下方より、空気を供給するものとなっている。空気供給器3は、加湿機能を備えることが好ましい。空気供給器3としては、例えば、図2に示すように、吸引する空気を浄化する空気清浄器31、空気清浄器により清浄化された空気を加湿器である水貯留槽33に送気する送気ポンプ32とからなるものが好ましい。また、送気手段としては、ドライフォークノズルを用いてもよい、また、空気供給器としては、水ミストスプレーを用いてもよい。
空気清浄器31としては、物理的に異物を捕捉するフィルターと、化学的に異物を吸着する吸着材(例えば、活性炭)とを備えるものが好ましい。送気ポンプ32より送られる空気は、水貯留槽33内に貯留されている水中に導入される。そして、水貯留槽の上部と次亜塩素酸水製造用ガス発生器2の下部とは、管路34により接続されている。
The air supply device 3 used in this embodiment supplies air from below the
The air purifier 31 preferably includes a filter that physically captures foreign matter and an adsorbent (for example, activated carbon) that chemically adsorbs foreign matter. The air sent from the air feed pump 32 is introduced into the water stored in the water storage tank 33. And the upper part of a water storage tank and the lower part of the
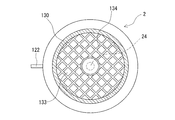
そして、ガス流出側ヘッダ145内に収納された板状部材144の下面には、環状凹部が設けられるとともに、この凹部に通気性メッシュ部材113が配置されており、保水性導電性多孔体25の流出を防止している。ガス流出側ヘッダ145は、ハウジング本体120の一端に固定されている。
通気性メッシュ部材112、113は、塩素耐腐食性非導電性材料(好ましくは、湿塩素耐腐食性非導電性材料)により形成されていることが好ましく、例えば、ガラス繊維、合成繊維、セラミックス繊維などによるメッシュが使用できる。合成繊維としては、ポリエチレン、ポリプロピレンなどのポリオレフィン系繊維、フッ素樹脂系繊維などが使用できる。
An annular recess is provided on the lower surface of the plate-
The
さらに、次亜塩素酸水製造装置1としては、図1に示すように、次亜塩素酸水作製部に設けられた製造次亜塩素酸水のpHもしくは電導度を検知するための検知部45の検知結果に基づき次亜塩素酸水製造装置を制御する制御部8を備えていることが好ましい。
この実施例の制御部8は、検知部45の検知結果に基づき、電源部6、空気供給器3および塩化物水溶液供給器5のすべての作動を制御するものとなっている。しかし、このようなものが好ましいが、いずれか1つのみ、また、任意の2つのみを制御するものであってもよい。
Furthermore, as shown in FIG. 1, the hypochlorous acid water production apparatus 1 has a detection unit 45 for detecting the pH or conductivity of the produced hypochlorous acid water provided in the hypochlorous acid water production unit. It is preferable to include a control unit 8 that controls the hypochlorous acid water production apparatus based on the detection result.
The control unit 8 of this embodiment controls all operations of the
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006203888A JP5070503B2 (en) | 2006-07-26 | 2006-07-26 | Hypochlorous acid water production equipment |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006203888A JP5070503B2 (en) | 2006-07-26 | 2006-07-26 | Hypochlorous acid water production equipment |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2008030979A JP2008030979A (en) | 2008-02-14 |
| JP2008030979A5 true JP2008030979A5 (en) | 2009-08-27 |
| JP5070503B2 JP5070503B2 (en) | 2012-11-14 |
Family
ID=39120805
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2006203888A Expired - Fee Related JP5070503B2 (en) | 2006-07-26 | 2006-07-26 | Hypochlorous acid water production equipment |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP5070503B2 (en) |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101142833B1 (en) | 2010-03-29 | 2012-05-08 | 동인메디텍 주식회사 | Apparatus for manufacturing of hypochlorous acid water |
| KR101398053B1 (en) | 2011-08-23 | 2014-05-27 | (주) 테크윈 | A scrubber system having an apparatus for creating automatic an oxidizing bent and absorbent |
| CN103791536A (en) * | 2014-01-23 | 2014-05-14 | 广西华信恒基科技发展有限公司 | Cooking fume purifying and exhausting device |
| KR101476097B1 (en) * | 2014-04-18 | 2014-12-24 | 안산시 | A sodium hypochlorite generator capable of the real time measurement of the concentration |
| JP6528173B2 (en) * | 2015-04-02 | 2019-06-12 | 株式会社微酸研 | Electrolytic cell and hypochlorous acid water production device |
| CN110314566B (en) * | 2019-05-29 | 2021-09-07 | 山东消博士消毒科技股份有限公司 | Disinfectant preparation system |
| CN110217862A (en) * | 2019-07-01 | 2019-09-10 | 冉金环保科技发展(苏州)有限公司 | It is a kind of to prepare subacidity hypochloric acid water electrolytic cell cooling structure |
| CN112499799A (en) * | 2020-08-10 | 2021-03-16 | 中山市原域企业投资有限公司 | Method and equipment for preparing hypochlorous acid by combining gas-liquid synthesis with ion exchange method |
| WO2022148535A1 (en) * | 2021-01-06 | 2022-07-14 | Condair Group Ag | Method of controlling the conductivity of a liquid which is atomized in order to form an aerosol |
| CN113398867B (en) * | 2021-07-14 | 2022-08-12 | 宁波蓝野医疗器械有限公司 | Thing networking intelligence supplies water hypochlorous acid disinfecting equipment |
-
2006
- 2006-07-26 JP JP2006203888A patent/JP5070503B2/en not_active Expired - Fee Related
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2008030979A5 (en) | ||
| JP5070503B2 (en) | Hypochlorous acid water production equipment | |
| TWI449811B (en) | Electrolytic device for generation of ph-controlled hypohalous acid aqueous solutions for disinfectant applications | |
| JP5191350B2 (en) | Vehicle air purification device | |
| JP4222786B2 (en) | Deodorizing / purifying method and apparatus for exhaust or smoke | |
| JP2006043707A (en) | Apparatus for producing electrolytic water | |
| JP5197306B2 (en) | Air purification device | |
| JP2009112996A5 (en) | ||
| JP2009112996A (en) | Method for sterilizing marine organism in water for seawater-utilizing equipment, and device therefor | |
| JP5370543B2 (en) | Hydrogen peroxide production apparatus and air conditioner, air purifier and humidifier using the same | |
| JP2009208021A (en) | Water absorption device and atomizer | |
| JP2008280549A (en) | Apparatus for producing hydrogen peroxide, and air conditioner, air cleaner and humidifier using the same | |
| KR101076630B1 (en) | Hydrogen water humidifier | |
| WO2022267895A1 (en) | Wet surface cleaning device having electrolysis apparatus and cleaning system | |
| JP5289215B2 (en) | Air sanitizer | |
| CN216639662U (en) | Disinfectant manufacturing installation | |
| JP6617288B2 (en) | Air purification device | |
| CN215799937U (en) | Intelligent control disinfection atomizing and oxygenation multipurpose machine | |
| CN114150332A (en) | Disinfectant liquid manufacturing apparatus and control method thereof | |
| KR101313698B1 (en) | Generation-system for antiseptic solution including chlorine | |
| JP2021120140A (en) | Ozone water generation method, generation sprayer and generation spraying device | |
| KR20210024835A (en) | HOCL Production System with Constant Current Control with Filmless Electrode Structure of Qing Structure | |
| JP2020049031A (en) | Method for removing bacteria, viruses and odorous substances in the air, and air cleaning device using the same | |
| CN204891001U (en) | Multifunction air purifier | |
| JP2007136381A (en) | Ozone water purifier |