EP3609986B1 - Alkoxylated amines as fuel additive - Google Patents
Alkoxylated amines as fuel additive Download PDFInfo
- Publication number
- EP3609986B1 EP3609986B1 EP18720112.4A EP18720112A EP3609986B1 EP 3609986 B1 EP3609986 B1 EP 3609986B1 EP 18720112 A EP18720112 A EP 18720112A EP 3609986 B1 EP3609986 B1 EP 3609986B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- propylene
- chain
- independently
- group
- ethylene
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L10/00—Use of additives to fuels or fires for particular purposes
- C10L10/06—Use of additives to fuels or fires for particular purposes for facilitating soot removal
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/222—Organic compounds containing nitrogen containing at least one carbon-to-nitrogen single bond
- C10L1/2222—(cyclo)aliphatic amines; polyamines (no macromolecular substituent 30C); quaternair ammonium compounds; carbamates
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/222—Organic compounds containing nitrogen containing at least one carbon-to-nitrogen single bond
- C10L1/2222—(cyclo)aliphatic amines; polyamines (no macromolecular substituent 30C); quaternair ammonium compounds; carbamates
- C10L1/2225—(cyclo)aliphatic amines; polyamines (no macromolecular substituent 30C); quaternair ammonium compounds; carbamates hydroxy containing
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/234—Macromolecular compounds
- C10L1/238—Macromolecular compounds obtained otherwise than by reactions involving only carbon-to-carbon unsaturated bonds
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/234—Macromolecular compounds
- C10L1/238—Macromolecular compounds obtained otherwise than by reactions involving only carbon-to-carbon unsaturated bonds
- C10L1/2383—Polyamines or polyimines, or derivatives thereof (poly)amines and imines; derivatives thereof (substituted by a macromolecular group containing 30C)
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/234—Macromolecular compounds
- C10L1/238—Macromolecular compounds obtained otherwise than by reactions involving only carbon-to-carbon unsaturated bonds
- C10L1/2383—Polyamines or polyimines, or derivatives thereof (poly)amines and imines; derivatives thereof (substituted by a macromolecular group containing 30C)
- C10L1/2387—Polyoxyalkyleneamines (poly)oxyalkylene amines and derivatives thereof (substituted by a macromolecular group containing 30C)
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L10/00—Use of additives to fuels or fires for particular purposes
- C10L10/04—Use of additives to fuels or fires for particular purposes for minimising corrosion or incrustation
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/18—Organic compounds containing oxygen
- C10L1/182—Organic compounds containing oxygen containing hydroxy groups; Salts thereof
- C10L1/1822—Organic compounds containing oxygen containing hydroxy groups; Salts thereof hydroxy group directly attached to (cyclo)aliphatic carbon atoms
- C10L1/1824—Organic compounds containing oxygen containing hydroxy groups; Salts thereof hydroxy group directly attached to (cyclo)aliphatic carbon atoms mono-hydroxy
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/18—Organic compounds containing oxygen
- C10L1/192—Macromolecular compounds
- C10L1/198—Macromolecular compounds obtained otherwise than by reactions involving only carbon-to-carbon unsaturated bonds homo- or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon to carbon double bond, and at least one being terminated by an acyloxy radical of a saturated carboxylic acid, of carbonic acid
- C10L1/1985—Macromolecular compounds obtained otherwise than by reactions involving only carbon-to-carbon unsaturated bonds homo- or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon to carbon double bond, and at least one being terminated by an acyloxy radical of a saturated carboxylic acid, of carbonic acid polyethers, e.g. di- polygylcols and derivatives; ethers - esters
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/222—Organic compounds containing nitrogen containing at least one carbon-to-nitrogen single bond
- C10L1/224—Amides; Imides carboxylic acid amides, imides
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L2200/00—Components of fuel compositions
- C10L2200/04—Organic compounds
- C10L2200/0407—Specifically defined hydrocarbon fractions as obtained from, e.g. a distillation column
- C10L2200/0415—Light distillates, e.g. LPG, naphtha
- C10L2200/0423—Gasoline
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L2270/00—Specifically adapted fuels
- C10L2270/02—Specifically adapted fuels for internal combustion engines
- C10L2270/023—Specifically adapted fuels for internal combustion engines for gasoline engines
Definitions
- the present invention describes alkoxylated amines as fuel additives to reduce injector deposits in direct injection gasoline engines.
- EP 700985 A1 is known to use butoxylated polyetheramines of mono- or polyamines as additives in fuels for gasoline engines.
- the polyetheramines show a reduction in deposits on the intake valves.
- the polyetheramines are prepared by butoxylation of alkanols, followed by reductive amination of the products obtained with mono- or polyamines.
- the polyetheramines therefore do not carry any free hydroxyl groups, but instead carry alkyl-capped butylene oxide chains.
- WO 2011/076949 discloses the use of mixed ethoxylated/propoxylated polyetheramines of monoamines in alcohol-containing gasoline to improve fuel economy and/or reduce deposits on intake valves and/or injection nozzles.
- EP 905217 A describes the use of ether group-containing compounds in gasoline for direct-injection gasoline engines to reduce emissions. To further reduce injector deposits or combustion chamber deposits, polyalkylamines or polyetheramines should be added to the fuel.
- the object of the present invention was to provide compounds with which the formation of deposits on injection nozzles can be avoided or reduced and/or existing deposits can be removed in direct-injection Otto engines.
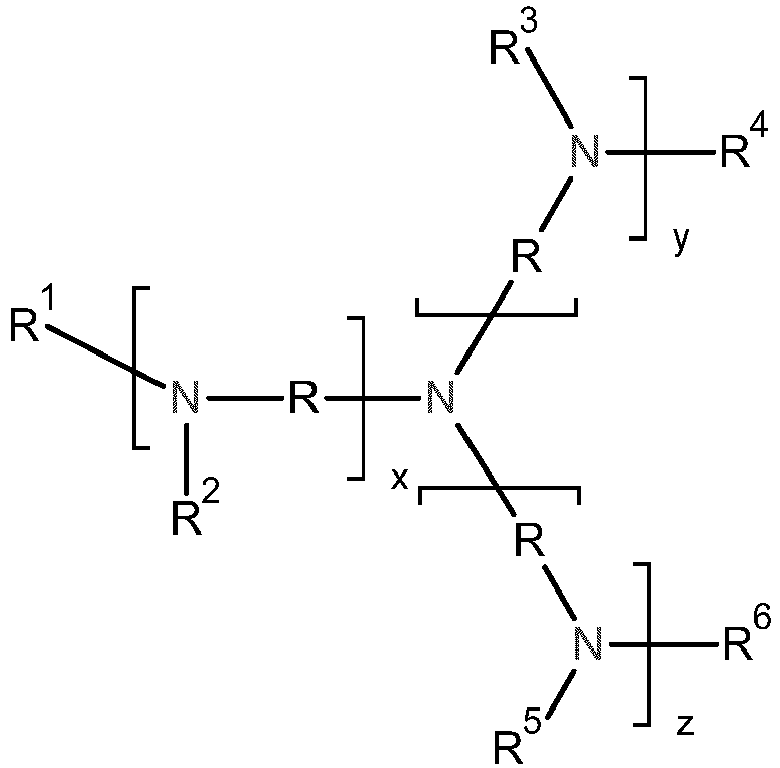
- R 1 , R 2 , R 3 , R 4 , R 5 and R 6 are independently hydrogen or a monovalent organic radical or R 1 and R 2 together and together with the nitrogen atom can form a five- to seven-membered ring.
- Preferred monovalent organic radicals are C 1 - to C 20 -alkyl, C 5 - to C 12 -cycloalkyl, C 6 - to C 12 -aryl or a radical -[-X i -] n -H, particularly preferably C 1 - to C 10 -alkyl, C 5 - to C 6 -cycloalkyl, C 6 - to C 12 -aryl or a radical -[-X i -] n -H, very particularly preferably particularly preferably C 1 - to C 4 -alkyl , especially methyl.
- R 1 and R 2 are preferably both the same and are each C 1 - to C 4 -alkyl and particularly preferably methyl.
- R 1 and R 2 together and together with the nitrogen atom to form a five- to seven-membered ring, particularly a five- or six-membered ring and particularly preferably a six-membered ring.
- the proviso applies that at least one of the radicals R 1 , R 2 , R 3 , R 4 , R 5 and R 6 is not hydrogen and at least one of the radicals R 1 , R 2 , R 3 , R 4 , R 5 and R 6 is a radical -[-X i -] n -H, preferably at least two of the radicals R 1 , R 2 , R 3 , R 4 , R 5 and R 6 are a radical -[-X i -] n -H .
- R is a divalent organic radical, preferably an alkylene radical having 2 to 10 carbon atoms, preferably an alkylene radical having 2 to 6 carbon atoms, particularly preferably an alkylene radical having 2 to 4 carbon atoms, very particularly preferably an alkylene radical having 2 or 3 carbon atoms and in particular an alkylene radical having 3 carbon atoms having alkylene radical.
- R is 1,2-ethylene, 1,2-propylene, 1,3-propylene, 1,2-butylene, 1,3-butylene, 1,4-butylene, 1,5-pentylene, 1,6-hexylene , 1,8-octylene or 1,10-decylene, particularly preferably 1,2-ethylene, 1,2-propylene, 1,3-propylene, 1,2-butylene, 1,4-butylene or 1,6-hexylene , very particularly preferably 1,2-ethylene, 1,2-propylene or 1,3-propylene and for compounds of the formula (I) or (III) in particular 1,3-propylene and for compounds of the formula (II) in particular 1, 2-ethylene or 1,3-propylene, especially 1,2-ethylene.
- w is a positive integer, preferably 1, 2, 3 or 4, particularly preferably 1, 2 or 3, very particularly preferably 1 or 2 and in particular 1.
- x, y and z independently of one another, are positive integers, preferably 1, 2, 3 or 4, particularly preferably 1, 2 or 3, very particularly preferably 1 or 2 and in particular 1.
- the proviso applies that the sum of x, y and z is not equal to zero, preferably the sum of x, y and z is at least 2, particularly preferably at least 3.
- the sum of x, y and z is preferably not greater than 10, particularly preferably not greater than 8, very particularly preferably not greater than 6 and in particular not greater than 5.
- each chain -[-X i -] n - occurring in the compounds the units X i can be statistically distributed, so that both the length of the chains and the distribution of the units X i within the chains are different can be.
- n, p and q independently of one another are positive integers from 1 to 50, preferably from 1 to 25, particularly preferably from 2 to 20 and very particularly preferably from 5 to 15.
- the sum of p and q is preferably from 2 to 50, particularly preferably from 5 to 40, very particularly preferably from 10 to 30 and in particular from 12 to 27.
- C 1 - to C 20 -alkyl are methyl, ethyl, isopropyl , n-propyl, n-butyl, isobutyl , sec -butyl, tert-butyl, n-hexyl, n-heptyl, n-octyl , 2-ethylhexyl, n-decyl, 2-propylheptyl, n-dodecyl, n-tetradecyl, n-hexadecyl, n-octadecyl and n-eicosyl.
- C 1 - to C 10 -alkyl are methyl, ethyl, isopropyl , n-propyl, n-butyl, isobutyl , sec- butyl, tert -butyl , n-hexyl, n-heptyl, n-octyl , 2-ethylhexyl, n-decyl and 2-propylheptyl.
- C 1 - to C 4 -alkyl are methyl, ethyl, isopropyl , n-propyl, n-butyl, isobutyl , sec- butyl and tert -butyl , preferably methyl, ethyl and n-butyl, particularly preferred methyl and ethyl and most preferably methyl.
- C 5 - to C 12 -cycloalkyl examples are cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclodecyl and cyclododecyl, preference being given to cyclopentyl and cyclohexyl.
- C 6 to C 12 aryl examples include phenyl, tolyl, ethylphenyl, benzyl, phenethyl, xylyl and naphthyl.
- reaction products of 1,2-ethylenediamine, diethylenetriamine, triethylenetetramine or 3-(dimethylamino)propylamine preferably diethylenetriamine or 3-(dimethylamino)propylamine and particularly preferably 3-(dimethylamino)propylamine with 10 to 30, preferably 12 to 27, are particularly preferred and more preferably 15 to 25 equivalents of propylene oxide and/or 1,2-butylene oxide.
- the compounds described are used according to the invention in order to avoid or reduce the formation of deposits on direct-injection injection nozzles and/or to remove or reduce existing deposits when operating direct-injection Otto engines with Otto fuels.
- Another advantage of the compounds described is that they can act as demulsifiers as an alternative or in addition to the uses described.
- Another object of the present invention are fuel additive concentrates according to claim 14.
- alkoxylated amines amines with primary and/or secondary amino groups
- HL Sanders et al. Journal of the American Oil Chemists Society, 1969, 46, 167-170 and in WO 2013/076024 described.
- the alkoxylation of amines and polyamines usually takes place in two stages: first, enough alkylene oxide is used to achieve an average degree of alkoxylation of from 0.5 to 1.5, preferably from 0.75 to 1.25, mol of alkylene oxide per NH function. This step is usually carried out in the presence of water (preferably 0.5-10.0% by weight based on the amine used), but can also be carried out in the absence of water.
- the reaction usually takes place at a temperature of from 50.degree. C. to 180.degree. C., preferably from 90.degree. C. to 160.degree.
- the alkylene oxide is preferably metered in over a period of 1 to 10 hours.
- the synthesis of the alkoxylated amines can also take place in one stage, with potassium hydroxide, sodium hydroxide, potassium methoxide, sodium methoxide, potassium acetate or sodium acetate being able to be used as possible catalysts.
- the reaction can also take place without a catalyst, since the amine used can itself catalyze the alkoxylation (see Mihail Ionescu, Chemistry and Technology of Polyols for Polyurethanes, Rapra Technology Limited, 2005).
- the alkylene oxides usually contain 2 or more carbon atoms, preferably 2 to 20 carbon atoms, in particular from 2 to 12 carbon atoms.
- Possible alkylene oxides are ethylene oxide, propylene oxide, isobutylene oxide, 1,2-butylene oxide, 2,3-butylene oxide, 1,2-pentene oxide, 1,2-dodecene oxide, styrene oxide.
- Possible alkylene oxides also include glycidyl ethers, for example 2-ethylhexyl glycidyl ether. Propylene oxide and 1,2-butylene oxide are particularly preferred.
- the metal is advantageously removed from the product for use in fuels or lubricants.
- these methods are known in principle and are described in Mihail Ionescu, Chemistry and Technology of Polyols for Polyurethanes, Rapra Technology Limited, 2005.
- alkali metals or alkaline earth metals can be removed by adsorption on magnesium silicates (commercial products Ambosol® or Magnesol® ).
- Potassium ions can be removed as potassium hydrogen phosphate by precipitation with phosphoric acid and subsequent filtration.
- Alkali metals or alkaline earth metals can also be removed with the aid of regenerable ion exchangers, in which case the alkoxylated amine can also be dissolved in a solvent.
- the compounds (A) according to the invention can be added to the fuels to which additives are to be added individually or as a mixture with other active additive components (co-additives).
- the hydrophobic hydrocarbon group in the above detergent additives which provides sufficient solubility in fuel has a number average molecular weight (Mn) of from 85 to 20,000, especially from 113 to 10,000, especially from 300 to 5000.
- Mn number average molecular weight
- Amines such as ammonia, monoamines or polyamines such as dimethylaminopropylamine, ethylenediamine, diethylenetriamine, triethylenetetramine or tetraethylenepentamine can be used for the amination.
- Corresponding additives based on polypropylene are in particular in the WO-A-94/24231 described.
- additives containing monoamino groups (a) are the compounds obtainable from polyisobutene epoxides by reaction with amines and subsequent dehydration and reduction of the amino alcohols, as they are used in particular in DE-A-196 20 262 are described.
- Additives containing carboxyl groups or their alkali metal or alkaline earth metal salts (d) are preferably copolymers of C 2 -C 40 -olefins with maleic anhydride having a total molar mass of 500 to 20,000, all or part of the carboxyl groups to the alkali metal or alkaline earth metal salts and one remaining Rest of the carboxyl groups are reacted with alcohols or amines.
- Such additives are in particular from EP-A-307 815 known.
- Such additives are mainly used to prevent valve seat wear and, as in the WO-A-87/01126 described, are used advantageously in combination with conventional fuel detergents such as poly(iso)buteneamines or polyetheramines.
- Additives containing sulfonic acid groups or their alkali metal or alkaline earth metal salts are preferably alkali metal or alkaline earth metal salts of an alkyl sulfosuccinate, as is found in particular in EP-A-639 632 is described.
- Such additives serve mainly to prevent valve seat wear and can be used to advantage in combination with conventional fuel detergents such as poly(iso)buteneamines or polyetheramines.
- Additives containing polyoxy-C 2 -C 4 -alkylene groups (f) are preferably polyethers or polyetheramines, which are obtained by reacting C 2 -C 60 -alkanols, C 6 -C 30 -alkanediols, mono- or di-C 2 -C 30 -alkylamines, C 1 -C 30 -alkylcyclohexanols or C 1 -C 30 -alkylphenols with 1 to 30 mol of ethylene oxide and/or propylene oxide and/or butylene oxide per hydroxyl group or amino group and, in the case of the polyetheramines, by subsequent reductive amination with ammonia, monoamines or polyamines.
- Such products are used in particular in EP-A-310 875 , EP-A-356 725 , EP-A-700 985 and US-A-4,877,416 described.
- polyethers such products also have carrier oil properties. Typical examples of these are tridecanol or isotridecanol butoxylates, isononylphenol butoxylates and polyisobutenol butoxylates and propoxylates and the corresponding reaction products with ammonia.
- Additives containing carboxylic acid ester groups (g) are preferably esters of mono-, di- or tricarboxylic acids with long-chain alkanols or polyols, in particular those with a minimum viscosity of 2 mm 2 /s at 100° C., as is the case in particular in DE-A-38 38 918 are described.
- Aliphatic or aromatic acids can be used as mono-, di- or tricarboxylic acids, and long-chain representatives having, for example, 6 to 24 carbon atoms are particularly suitable as ester alcohols or ester polyols.
- esters are adipates, phthalates, isophthalates, terephthalates and trimellitates of isooctanol, isononanol, isodecanol and isotridecanol. Such products also fulfill carrier oil properties.
- derivatives with aliphatic polyamines such as ethylenediamine, diethylenetriamine, triethylenetetramine or tetraethylenepentamine.
- Such petrol additives are particularly popular US-A-4,849,572 described.
- Additives containing groups (i) produced by Mannich reaction of substituted phenols with aldehydes and mono- or polyamines are preferably reaction products of polyisobutene-substituted phenols with formaldehyde and mono- or polyamines such as ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine or dimethylaminopropylamine.
- Such "polyisobutene Mannich bases" are particularly in the EP-A-831 141 described.
- additive formulations according to the invention can also be combined with other customary components and additives.
- the main ones to be mentioned here are carrier oils without a pronounced detergent effect.
- Suitable mineral carrier oils are fractions obtained during petroleum processing, such as bright stock or base oils with viscosities such as those from the class SN 500-2000; but also aromatic hydrocarbons, paraffinic hydrocarbons and alkoxyalkanols.
- a fraction known as "hydrocrack oil” and obtained in the refining of mineral oil vacuum distillate cut with a boiling range of about 360 to 500°C, obtainable from natural mineral oil which has been catalytically hydrogenated and isomerized and dewaxed under high pressure
- mineral carrier oils mentioned above are also suitable.
- Examples of synthetic carrier oils that can be used according to the invention are selected from: polyolefins (polyalphaolefins or polyinternal olefins), (poly)esters, (poly)alkoxylates, polyethers, aliphatic polyetheramines, alkylphenol-started polyethers, alkylphenol-started polyetheramines and carboxylic acid esters of long-chain alkanols.
- Examples of particularly suitable synthetic carrier oils are alcohol-started polyethers with about 5 to 35 C 3 - to C 6 -alkylene oxide units, mostly selected from propylene oxide, n-butylene oxide and isobutylene oxide units or mixtures thereof.
- Non-limiting examples of starter alcohols suitable for this are long-chain alkanols or phenols substituted with long-chain alkyl, where the long-chain alkyl radical is in particular a straight-chain or branched C 6 - to C 18 -alkyl radical. Preferred examples of this are tridecanol, heptadecanol and nonylphenol.
- the use of the compounds (A) according to the invention can often reduce the use of carrier oils.
- suitable polyethers or polyetheramines are preferably compounds containing polyoxy-C 2 -C 4 -alkylene groups, which are obtained by reacting C 2 -C 60 -alkanols, C 6 -C 30 -alkanediols, mono- or di-C 2 -C 30 -alkylamines, C 1 -C 30 -alkylcyclohexanols or C 1 -C 30 -alkylphenols with 1 to 30 mol of ethylene oxide and/or propylene oxide and/or butylene oxide per hydroxyl group or amino group and, in the case of polyetheramines, by subsequent reductive amination with ammonia, Monoamines or polyamines are available.
- poly-C 2 -C 6 -alkylene oxide amines or functional derivatives thereof can be used as polyetheramines.
- Typical examples of these are tridecanol or isotridecanol butoxylates, heptadecanol or isoheptadecanol butoxylates, isononylphenol butoxylates and polyisobutenol butoxylates and propoxylates and the corresponding reaction products with ammonia.
- carboxylic acid esters of long-chain alkanols are, in particular, esters of mono-, di- or tricarboxylic acids with long-chain alkanols or polyols, such as those in particular DE-A-38 38 918 are described.
- Aliphatic or aromatic acids can be used as mono-, di- or tricarboxylic acids, and long-chain representatives having 6 to 24 carbon atoms are particularly suitable as ester alcohols or ester polyols.
- esters are adipates, phthalates, isophthalates, terephthalates and trimellitates of isooctanol, isononanol, isodecanol and isotridecanol, such as di-(n- or isotridecyl) phthalate or di-(iso-heptadecyl) phthalate .
- Examples of particularly suitable synthetic carrier oils are alcohol-started polyethers having about 5 to 35, preferably about 5 to 30, particularly preferably 7 to 25, C 3 -C 6 -alkylene oxide units, such as selected from propylene oxide, n-butylene oxide and i-butylene oxide units, or mixtures thereof, preferably selected from propylene oxide and i-butylene oxide units.
- suitable starter alcohols are long-chain alkanols or phenols substituted with long-chain alkyl, where the long-chain alkyl radical is in particular a straight-chain or branched C 6 -C 18 -alkyl radical.
- alkanols are decanol, tridecanol, heptadecanol and nonylphenol, particularly preferably branched decanol, tridecanol and heptadecanol.
- customary additives are corrosion inhibitors, for example based on film-forming ammonium salts of organic carboxylic acids or on heterocyclic aromatics in the case of non-ferrous metal corrosion protection; preferred corrosion inhibitors are mono-, di- and polycarboxylic acids which have at least 12 carbon atoms, preferably at least 14, more preferably at least 16 and most preferably at least 18 carbon atoms and preferably have no other functionalities apart from hydrocarbon radicals and carboxyl groups.
- fatty acids examples include fatty acids, dimeric fatty acids, alkyl and alkenylsuccinic acids and hydrolyzed olefin-maleic anhydride copolymers, preference being given to dodecanoic acid (lauric acid), tridecanoic acid, tetradecanoic acid (myristic acid), pentadecanoic acid, palmitic acid (hexadecanoic acid), margaric acid (heptadecanoic acid), stearic acid (octadecanoic acid), Nonadecanoic acid, arachidic acid (eicosanoic acid), behenic acid (docosanic acid), tetracosanoic acid (lignoceric acid), cerotic acid (hexacosanoic acid), triacontanoic acid (melissinic acid), palmitoleic acid [(9Z)-hexadec-9-enoic acid], oleic acid [(9Z)
- antioxidants or stabilizers for example based on amines such as p-phenylenediamine, dicyclohexylamine or derivatives thereof, or on phenols such as 2,4-di-tert-butylphenol or 3,5-di-tert-butyl-4- hydroxyphenylpropionic acid; demulsifiers; antistatic agents; metallocenes such as ferrocene; methylcyclopentadienyl manganese tricarbonyl; lubricity improvers (other than the triazoles of this invention) such as certain fatty acids, alkenyl succinic acid esters, bis(hydroxyalkyl) fatty amines, hydroxyacetamides, or castor oil; and dyes (markers). If necessary, amines are also added to lower the pH of the fuel.
- amines such as p-phenylenediamine, dicyclohexylamine or derivatives thereof, or on phenols such as 2,4-di-tert
- solvents are, for example, aromatics, such as solvent naphtha, preferably naphthalene-depleted solvent naphtha, benzene, toluene, xylene, kerosene with an aromatics content of less than 50% by volume, a sulfur content of less than 10 mg/kg and a flash point according to DIN EN ISO 13736: 2000 -04 above 60 °C and higher alcohols which have a flash point according to DIN EN ISO 13736: 2000-04 above 60 °C, preferably 2-ethylhexanol or 2-propylheptanol.
- aromatics such as solvent naphtha, preferably naphthalene-depleted solvent naphtha, benzene, toluene, xylene, kerosene with an aromatics content of less than 50% by volume, a sulfur content of less than 10 mg/kg and a flash point according to DIN EN ISO 13736: 2000 -04 above 60 °C and higher alcohols which have
- the components or additives can be added to the fuel individually or as a previously prepared concentrate (additive package) together with the compound (A) according to the invention.
- additive compositions according to the invention can be used in all conventional gasoline fuels, as described, for example, in Ullmann's Encyclopedia of Industrial Chemistry, 5th edition 1990, volume A16, p. 719 et seq.
- the petrol is preferably at least one petrol according to DIN EN 228.
- a gasoline with an aromatics content of no more than 60 such as no more than 42 or no more than 35% by volume and/or a sulfur content of no more than 2000, such as no more than 150 or no more than 10 ppm by weight, is possible.
- the aromatics content of petrol is, for example, 10 to 50% by volume, such as 30 to 42% by volume, in particular 32 to 40% by volume, or a maximum of 35% by volume.
- the sulfur content of petrol is, for example, 2 to 500, such as 5 to 100 ppm by weight, or a maximum of 10 ppm by weight.
- the gasoline can, for example, have an olefin content of up to 50% by volume, such as from 6 to 21% by volume, in particular 7 to 18% by volume; a benzene content of up to 5% by volume, such as 0.5 to 1.0% by volume, in particular 0.6 to 0.9% by volume, and/or an oxygen content of up to 25% by volume , such as up to 10% by weight or 1.0 to 2.7% by weight, in particular from 1.2 to 2.0% by weight.
- an olefin content of up to 50% by volume, such as from 6 to 21% by volume, in particular 7 to 18% by volume
- a benzene content of up to 5% by volume, such as 0.5 to 1.0% by volume, in particular 0.6 to 0.9% by volume
- an oxygen content of up to 25% by volume such as up to 10% by weight or 1.0 to 2.7% by weight, in particular from 1.2 to 2.0% by weight.
- petrol fuels that can be mentioned by way of example have an aromatics content of no more than 38 or 35% by volume, an olefin content of no more than 21% by volume, a sulfur content of no more than 50 or 10 ppm by weight, a benzene content of no more than 1, 0% by volume and an oxygen content of 1.0 to 2.7% by weight.
- the content of alcohols and ethers in petrol can vary over a wide range. Examples of typical maximum contents are 15% by volume for methanol, 85% by volume for ethanol, 20% by volume for isopropanol, 15% by volume for tert-butanol, 20% by volume for isobutanol and ether with 5 or more carbon atoms in the molecule, 30% by volume.
- the summer vapor pressure of petrol is usually a maximum of 70 kPa, in particular 60 kPa (each at 37° C.).
- the RON of petrol is usually 75 to 105.
- a typical range for the corresponding MON is 65 to 95.
- the dosage is such that the compound (A) according to the invention is present in the fuel in amounts of 20-2000 ppm by weight, particularly preferably in amounts of 30-1000 ppm by weight, very particularly preferably in amounts of 40-500 ppm by weight, in particular is present in amounts of 50-300 wppm and especially in amounts of 10-100 wppm.
- DMAPA methyl methacrylate
- water 4.1 g, completely desalinated
- the reactor was rendered inert with nitrogen, then it was heated to 130° C. and a pressure of 2.0 bar absolute was established with nitrogen.
- Propylene oxide (465 g; 8.0 mol) was metered in over a period of 6 hours. The reaction was allowed to continue for 6 h at 130° C., the mixture was cooled to 50° C., the reactor was flushed with nitrogen and the product was discharged. The product was then freed from low boilers on a rotary evaporator (90° C./10 mbar/2 h).
- ethylenediamine ⁇ 25 PO was obtained by reacting Quadrol L (ethylenediamine ⁇ 4PO) with 21 equivalents of PO.
- DMAPA DMAPA (408.8 g; 4.0 mol) was placed in a 3.5 l pressure autoclave with an inclined blade stirrer and the stirrer was switched on.
- the reactor was rendered inert with nitrogen, then it was heated to 120° C. and a pressure of 2.0 bar absolute was established with nitrogen.
- 1,2-Butylene oxide (577 g; 8.0 mol) was metered in over a period of 15 hours.
- the reaction was allowed to continue for 6 h at 120° C., the mixture was cooled to 50° C., the reactor was flushed with nitrogen and the product was discharged. The product was then freed from low boilers on a rotary evaporator (90° C./10 mbar/2 h).
- the reactor was flushed as well nitrogen and drained the product.
- the product was freed from low boilers on a rotary evaporator (90° C./10 mbar/2 h).
- the product was then mixed with 30 g of Ambosol® , stirred at 80° C./100 mbar for 2 h and filtered using a pressure suction filter (Filter medium Seitz K 900 depth filter).
- the product was obtained in the form of a yellow oil.
- the fuel was admixed with 80 ppm by weight of the specified products from the synthesis examples or formulations and the FR value was determined over the running time.
- the FR value is a parameter created by the engine control and corresponds to the fuel injection time into the combustion chamber. If the FR value increases during the test run, this indicates deposits on the injector, the larger the increase, the more deposits have formed. On the other hand, if the FR value remains constant or even falls over the course of the remaining run, the injection nozzle remains free of deposits.
Landscapes
- Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Combustion & Propulsion (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Solid Fuels And Fuel-Associated Substances (AREA)
Description
Die vorliegende Erfindung beschreibt alkoxylierte Amine als Kraftstoffadditive zur Verringerung von Injektorablagerungen in direkteinspritzenden Benzinmotoren.The present invention describes alkoxylated amines as fuel additives to reduce injector deposits in direct injection gasoline engines.
Aus
Aus
Aus
Aufgabe der vorliegenden Erfindung war es, Verbindungen zur Verfügung zu stellen, mit denen in direkteinspritzenden Ottomotoren die Bildung von Ablagerungen an Einspritzdüsen vermieden oder verringert und/oder bestehende Ablagerungen entfernt werden können.The object of the present invention was to provide compounds with which the formation of deposits on injection nozzles can be avoided or reduced and/or existing deposits can be removed in direct-injection Otto engines.
Die Aufgabe wurde gelöst durch die Verwendung von Verbindungen (A) ausgewählt aus der Gruppe bestehend aus
- R einen zweibindigen organischen Rest, bevorzugt einen 2 bis 10 Kohlenstoffatome aufweisenden Alkylenrest,
- R1, R2, R3, R4, R5 und R6 unabhängig voneinander Wasserstoff oder einen einbindigen organischen Rest oder einen Rest -[-Xi-]n-H oder R1 und R2 gemeinsam und zusammen mit dem Stickstoffatom einen fünf- bis siebengliedrigen Ring bilden können, bevorzugt Wasserstoff, einen 1 bis 20 Kohlenstoffatome aufweisenden Alkylrest oder einen Rest -[-Xi-]n-H,
- w eine positive ganze Zahl und
- x, y und z unabhängig voneinander Null oder eine positive ganze Zahl
- worin n eine positive ganze Zahl bedeutet, und
- worin jedes Xi für i = 1 bis n unabhängig voneinander ausgewählt ist aus der Gruppe bestehend aus -CH2-CH2-O-, -CH2-CH(CH3)-O-, -CH(CH3)-CH2-O-, -CH2-C(CH3)2-O-, -C(CH3)2-CH2-O-, -C H2-CH(C2H5)-O-, -CH(C2H5)-CH2-O- und -CH(CH3)-CH(CH3)-O-, bevorzugt ausgewählt aus der Gruppe bestehend aus -CH2-CH(CH3)-O-, -CH(CH3)-CH2-O-, -CH2-C(CH3)2-O-, -C(CH3)2-CH2-O-, -CH2-CH(C2H5)-O -, -CH(C2H5)-CH2-O- und -CH(CH3)-CH(CH3)-O-, besonders bevorzugt ausgewählt aus der Gruppe bestehend aus -CH2-CH(CH3)-O-, -CH(CH3)-CH2-O-, -CH2-C(CH3)2-O-, -C(CH3)2-CH2-O-, -CH2-CH(C2H5)-O
- und -CH(C2H5)-CH2-O-, ganz besonders bevorzugt ausgewählt aus der Gruppe bestehend aus -CH2-CH(C2H5)-O-, -CH(C2H5)-CH2-O-, -CH2-CH(CH3)-O- und -CH(CH3)-CH2-O-, und insbesondere ausgewählt aus der Gruppe bestehend aus -CH2-CH(CH3)-O- und -CH(CH3)-CH2-O-, mit der Maßgabe, daß
- die Summe aus x, y und z ungleich Null ist
- mindestens einer der Reste R1, R2, R3, R4, R5 und R6 nicht Wasserstoff ist und
- mindestens einer der Reste R1, R2, R3, R4, R5 und R6 einen Rest -[-Xi-]n-H darstellt,
- R is a divalent organic radical, preferably an alkylene radical having 2 to 10 carbon atoms,
- R 1 , R 2 , R 3 , R 4 , R 5 and R 6 are independently hydrogen or a monovalent organic radical or a radical -[-X i -] n -H or R 1 and R 2 together and together with the nitrogen atom can form a five- to seven-membered ring, preferably hydrogen, an alkyl radical having 1 to 20 carbon atoms or a radical -[-X i -] n -H,
- w a positive integer and
- x, y, and z are independently zero or a positive integer
- where n is a positive integer, and
- wherein each X i for i = 1 to n is independently selected from the group consisting of -CH 2 -CH 2 -O-, -CH 2 -CH(CH 3 )-O-, -CH(CH 3 )-CH 2 -O-, -CH 2 -C(CH 3 ) 2 -O-, -C(CH 3 ) 2 -CH 2 -O-, -CH 2 -CH(C 2 H 5 )-O-, -CH (C 2 H 5 )-CH 2 -O- and -CH(CH 3 )-CH(CH 3 )-O-, preferably selected from the group consisting of -CH 2 -CH(CH 3 )-O-, - CH(CH 3 )-CH 2 -O-, -CH 2 -C(CH 3 ) 2 -O-, -C(CH 3 ) 2 -CH 2 -O-, -CH 2 -CH(C 2 H 5 )-O-, -CH(C 2 H 5 )-CH 2 -O- and -CH(CH 3 )-CH(CH 3 )-O-, particularly preferably selected from the group consisting of -CH 2 -CH( CH3 )-O-, -CH(CH3 ) -CH2 -O-, -CH2 -C(CH3 )2 -O- , -C ( CH3 )2 -CH2 -O- , -CH 2 -CH(C 2 H 5 )-O
- and -CH(C 2 H 5 )-CH 2 -O-, most preferably selected from the group consisting of -CH 2 -CH(C 2 H 5 )-O-, -CH(C 2 H 5 )-CH 2 -O-, -CH 2 -CH(CH 3 )-O- and -CH(CH 3 )-CH 2 -O-, and in particular selected from the group consisting of -CH 2 -CH(CH 3 )-O- - and -CH(CH 3 )-CH 2 -O-, with the proviso that
- the sum of x, y, and z is non-zero
- at least one of the radicals R 1 , R 2 , R 3 , R 4 , R 5 and R 6 is not hydrogen and
- at least one of the radicals R 1 , R 2 , R 3 , R 4 , R 5 and R 6 represents a radical -[-X i -] n -H,
Unter diesen sind die Verbindungen der Formel (I) bevorzugt.Among these, the compounds of formula (I) are preferred.
In einer bevorzugten Ausführungsform erfüllen die Verbindungen die Formel (III)
- R1, R2 und Xi die oben genannten Bedeutungen haben und
- p und q unabhängig voneinander eine positive ganze Zahl bedeuten.
- R 1 , R 2 and X i have the meanings given above and
- p and q independently represent a positive integer.
Darin bedeuten R1, R2, R3, R4, R5 und R6 unabhängig voneinander Wasserstoff oder einen einbindigen organischen Rest oder R1 und R2 können gemeinsam und zusammen mit dem Stickstoffatom einen fünf- bis siebengliedrigen Ring bilden.R 1 , R 2 , R 3 , R 4 , R 5 and R 6 are independently hydrogen or a monovalent organic radical or R 1 and R 2 together and together with the nitrogen atom can form a five- to seven-membered ring.
Bevorzugt einbindige organische Reste sind C1- bis C20-Alkyl, C5- bis C12-Cycloalkyl, C6- bis C12-Aryl oder einen Rest -[-Xi-]n-H, besonders bevorzugt C1- bis C10-Alkyl, C5- bis C6-Cycloalkyl, C6- bis C12-Aryl oder einen Rest -[-Xi-]n-H, ganz besonders bevorzugt besonders bevorzugt C1- bis C4-Alkyl, insbesondere Methyl.Preferred monovalent organic radicals are C 1 - to C 20 -alkyl, C 5 - to C 12 -cycloalkyl, C 6 - to C 12 -aryl or a radical -[-X i -] n -H, particularly preferably C 1 - to C 10 -alkyl, C 5 - to C 6 -cycloalkyl, C 6 - to C 12 -aryl or a radical -[-X i -] n -H, very particularly preferably particularly preferably C 1 - to C 4 -alkyl , especially methyl.
Bevorzugt sind R1 und R2 beide gleich und jeweils C1- bis C4-Alkyl und besonders bevorzugt Methyl.R 1 and R 2 are preferably both the same and are each C 1 - to C 4 -alkyl and particularly preferably methyl.
Weiterhin ist es möglich, daß R1 und R2 gemeinsam und zusammen mit dem Stickstoffatom einen fünf- bis siebengliedrigen Ring bilden, besonders einen fünf- oder sechsgliedrigen Ring und besonders bevorzugt einen sechsgliedrigen Ring.Furthermore, it is possible for R 1 and R 2 together and together with the nitrogen atom to form a five- to seven-membered ring, particularly a five- or six-membered ring and particularly preferably a six-membered ring.
Dabei gilt die Maßgabe, mindestens einer der Reste R1, R2, R3, R4, R5 und R6 nicht Wasserstoff ist und mindestens einer der Reste R1, R2, R3, R4, R5 und R6 einen Rest -[-Xi-]n-H darstellt, bevorzugt sind mindestens zwei der Reste R1, R2, R3, R4, R5 und R6 ein Rest -[-Xi-]n-H.The proviso applies that at least one of the radicals R 1 , R 2 , R 3 , R 4 , R 5 and R 6 is not hydrogen and at least one of the radicals R 1 , R 2 , R 3 , R 4 , R 5 and R 6 is a radical -[-X i -] n -H, preferably at least two of the radicals R 1 , R 2 , R 3 , R 4 , R 5 and R 6 are a radical -[-X i -] n -H .
R bedeutet einen zweibindigen organischen Rest, bevorzugt einen 2 bis 10 Kohlenstoffatome aufweisenden Alkylenrest, bevorzugt einen 2 bis 6 Kohlenstoffatome aufweisenden Alkylenrest, besonders bevorzugt einen 2 bis 4 Kohlenstoffatome aufweisenden Alkylenrest, ganz besonders bevorzugt einen 2 oder 3 Kohlenstoffatome aufweisenden Alkylenrest und insbesondere einen 3 Kohlenstoffatome aufweisenden Alkylenrest.R is a divalent organic radical, preferably an alkylene radical having 2 to 10 carbon atoms, preferably an alkylene radical having 2 to 6 carbon atoms, particularly preferably an alkylene radical having 2 to 4 carbon atoms, very particularly preferably an alkylene radical having 2 or 3 carbon atoms and in particular an alkylene radical having 3 carbon atoms having alkylene radical.
Bevorzugt ist R 1,2-Ethylen, 1,2-Propylen, 1,3-Propylen, 1,2-Butylen, 1,3-Butylen, 1,4-Butylen, 1,5-Pentylen, 1,6-Hexylen, 1,8-Octylen oder 1,10-Decylen, besonders bevorzugt 1,2-Ethylen, 1,2-Propylen, 1,3-Propylen, 1,2-Butylen, 1,4-Butylen oder 1,6-Hexylen, ganz besonders bevorzugt 1,2-Ethylen, 1,2-Propylen oder 1,3-Propylen und für Verbindungen der Formel (I) oder (III) insbesondere 1,3-Propylen und für Verbindungen der Formel (II) insbesondere 1,2-Ethylen oder 1,3-Propylen, speziell 1,2-Ethylen.Preferably R is 1,2-ethylene, 1,2-propylene, 1,3-propylene, 1,2-butylene, 1,3-butylene, 1,4-butylene, 1,5-pentylene, 1,6-hexylene , 1,8-octylene or 1,10-decylene, particularly preferably 1,2-ethylene, 1,2-propylene, 1,3-propylene, 1,2-butylene, 1,4-butylene or 1,6-hexylene , very particularly preferably 1,2-ethylene, 1,2-propylene or 1,3-propylene and for compounds of the formula (I) or (III) in particular 1,3-propylene and for compounds of the formula (II) in particular 1, 2-ethylene or 1,3-propylene, especially 1,2-ethylene.
Ferner bedeutet w eine positive ganze Zahl, bevorzugt 1, 2, 3 oder 4, besonders bevorzugt 1, 2 oder 3, ganz besonders bevorzugt 1 oder 2 und insbesondere 1.Furthermore, w is a positive integer, preferably 1, 2, 3 or 4, particularly preferably 1, 2 or 3, very particularly preferably 1 or 2 and in particular 1.
Ferner bedeuten x, y und z unabhängig voneinander positive ganze Zahlen, bevorzugt 1, 2, 3 oder 4, besonders bevorzugt 1, 2 oder 3, ganz besonders bevorzugt 1 oder 2 und insbesondere 1.Furthermore, x, y and z, independently of one another, are positive integers, preferably 1, 2, 3 or 4, particularly preferably 1, 2 or 3, very particularly preferably 1 or 2 and in particular 1.
Dabei gilt die Maßgabe, daß die Summe aus x, y und z ungleich Null ist, bevorzugt ist die Summe aus x, y und z mindestens 2, besonders bevorzugt mindestens 3.The proviso applies that the sum of x, y and z is not equal to zero, preferably the sum of x, y and z is at least 2, particularly preferably at least 3.
Bevorzugt ist die Summe aus x, y und z nicht größer als 10, besonders bevorzugt nicht größer als 8, ganz besonders bevorzugt nicht größer als 6 und insbesondere nicht größer als 5.The sum of x, y and z is preferably not greater than 10, particularly preferably not greater than 8, very particularly preferably not greater than 6 and in particular not greater than 5.
Ferner ist jedes Xi für i = 1 bis n unabhängig voneinander ausgewählt ist aus der Gruppe bestehend aus -CH2-CH2-O-, -CH2-CH(CH3)-O-, -CH(CH3)-CH2-O-, -CH2-C(CH3)2-O-, -C(CH3)2-CH2-O-, -C H2-CH(C2H5)-O-, -CH(C2H5)-CH2-O- und -CH(CH3)-CH(CH3)-O-, bevorzugt ausgewählt aus der Gruppe bestehend aus -CH2-CH(CH3)-O-, -CH(CH3)-CH2-O-, -CH2-C(CH3)2-O-, -C(CH3)2-CH2-O-, -CH2-CH(C2H5)-O -, -CH(C2H5)-CH2-O- und -CH(CH3)-CH(CH3)-O-, besonders bevorzugt ausgewählt aus der Gruppe bestehend aus -CH2-CH(CH3)-O-, -CH(CH3)-CH2-O-, -CH2-C(CH3)2-O-, -C(CH3)2-CH2-O-, -CH2-CH(C2H5)-O - und -CH(C2H5)-CH2-O- und ganz besonders bevorzugt ausgewählt aus der Gruppe bestehend aus -CH2-CH(C2H5)-O-, -CH(C2H5)-CH2-O-, -CH2-CH(CH3)-O- und -CH(CH3)-CH2-O-, und insbesondere ausgewählt aus der Gruppe bestehend aus -CH2-CH(CH3)-O- und -CH(CH3)-CH2-O-.Further, each X i for i = 1 through n is independently selected from the group consisting of -CH 2 -CH 2 -O-, -CH 2 -CH(CH 3 )-O-, -CH(CH 3 )- CH 2 -O-, -CH 2 -C(CH 3 ) 2 -O-, -C(CH 3 ) 2 -CH 2 -O-, -CH 2 -CH(C 2 H 5 )-O-, - CH(C 2 H 5 )-CH 2 -O- and -CH(CH 3 )-CH(CH 3 )-O-, preferably selected from the group consisting of -CH 2 -CH(CH 3 )-O-, -CH(CH 3 )-CH 2 -O-, -CH 2 -C(CH 3 ) 2 -O-, -C(CH 3 ) 2 -CH 2 -O-, -CH 2 -CH(C 2 H 5 )-O-, -CH(C 2 H 5 )-CH 2 -O- and -CH(CH 3 )-CH(CH 3 )-O-, particularly preferably selected from the group consisting of -CH 2 -CH (CH 3 )-O-, -CH(CH 3 )-CH 2 -O-, -CH 2 -C(CH 3 ) 2 -O-, -C(CH 3 ) 2 -CH 2 -O-, - CH 2 -CH(C 2 H 5 )-O- and -CH(C 2 H 5 )-CH 2 -O- and most preferably selected from the group consisting of -CH 2 -CH(C 2 H 5 )- O-, -CH(C 2 H 5 )-CH 2 -O-, -CH 2 -CH(CH 3 )-O- and -CH(CH 3 )-CH 2 -O-, and in particular selected from the group consisting of -CH 2 -CH(CH 3 )-O- and -CH(CH 3 )-CH 2 -O-.
Dabei ist zu beachten, daß in jeder in den Verbindungen vorkommende Kette -[-Xi-]n- die Einheiten Xi statistisch verteilt sein können, so daß sowohl die Länge der Ketten als auch die Verteilung der Einheiten Xi innerhalb der Ketten verschieden sein kann.It should be noted that in each chain -[-X i -] n - occurring in the compounds, the units X i can be statistically distributed, so that both the length of the chains and the distribution of the units X i within the chains are different can be.
Ferner bedeuten n, p und q unabhängig voneinander eine positive ganze Zahl von 1 bis 50, bevorzugt von 1 bis 25, besonders bevorzugt von 2 bis 20 und ganz besonders bevorzugt von 5 bis 15.Furthermore, n, p and q independently of one another are positive integers from 1 to 50, preferably from 1 to 25, particularly preferably from 2 to 20 and very particularly preferably from 5 to 15.
Die Summe aus p und q beträgt bevorzugt von 2 bis 50, besonders bevorzugt von 5 bis 40, ganz besonders bevorzugt von 10 bis 30 und insbesondere von 12 bis 27.The sum of p and q is preferably from 2 to 50, particularly preferably from 5 to 40, very particularly preferably from 10 to 30 and in particular from 12 to 27.
Beispiele für C1- bis C20-Alkyl sind Methyl, Ethyl, iso-Propyl, n-Propyl, n-Butyl, iso-Butyl, sek-Butyl, tert-Butyl, n-Hexyl, n-Heptyl, n-Octyl, 2-Ethylhexyl, n-Decyl, 2-Propylheptyl, n-Dodecyl, n-Tetradecyl, n-Hexadecyl, n-Octadecyl und n-Eicosyl.Examples of C 1 - to C 20 -alkyl are methyl, ethyl, isopropyl , n-propyl, n-butyl, isobutyl , sec -butyl, tert-butyl, n-hexyl, n-heptyl, n-octyl , 2-ethylhexyl, n-decyl, 2-propylheptyl, n-dodecyl, n-tetradecyl, n-hexadecyl, n-octadecyl and n-eicosyl.
Beispiele für C1- bis C10-Alkyl sind Methyl, Ethyl, iso-Propyl, n-Propyl, n-Butyl, iso-Butyl, sek-Butyl, tert-Butyl, n-Hexyl, n-Heptyl, n-Octyl, 2-Ethylhexyl, n-Decyl und 2-Propylheptyl.Examples of C 1 - to C 10 -alkyl are methyl, ethyl, isopropyl , n-propyl, n-butyl, isobutyl , sec- butyl, tert -butyl , n-hexyl, n-heptyl, n-octyl , 2-ethylhexyl, n-decyl and 2-propylheptyl.
Beispiele für C1- bis C4-Alkyl sind Methyl, Ethyl, iso-Propyl, n-Propyl, n-Butyl, iso-Butyl, sek-Butyl und tert-Butyl, bevorzugt Methyl, Ethyl und n-Butyl, besonders bevorzugt Methl und Ethyl und ganz besonders bevorzugt Methyl.Examples of C 1 - to C 4 -alkyl are methyl, ethyl, isopropyl , n-propyl, n-butyl, isobutyl , sec- butyl and tert -butyl , preferably methyl, ethyl and n-butyl, particularly preferred methyl and ethyl and most preferably methyl.
Beispiele C5- bis C12-Cycloalkyl sind Cyclopentyl, Cyclohexyl, Cycloheptyl, Cyclooctyl, Cyclodecyl und Cyclododecyl, bevorzugt sind Cyclopentyl und Cyclohexyl.Examples of C 5 - to C 12 -cycloalkyl are cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclodecyl and cyclododecyl, preference being given to cyclopentyl and cyclohexyl.
Beispiele für C6- bis C12-Aryl Phenyl, Tolyl, Ethylphenyl, Benzyl, Phenethyl, Xylyl und Naphthyl.Examples of C 6 to C 12 aryl are phenyl, tolyl, ethylphenyl, benzyl, phenethyl, xylyl and naphthyl.
Folgende Verbindungen der Formel (I) stellen bevorzugte Ausführungsformen dar:
- (la) R sei 1,2-Ethylen, w = 1, R1 bis R3 Methyl und R4 eine Kette -[-Xi-]n-H.
- (Ib) R sei 1,2-Ethylen, w = 1, R1 und R2 Methyl und R3 und R4 jeweils unabhängig voneinander eine Kette -[-Xi-]n-H.
- (Ic) R sei 1,2-Propylen, w = 1, R1 und R2 Methyl und R3 und R4 jeweils unabhängig voneinander eine Kette -[-Xi-]n-H.
- (Id) R sei 1,3-Propylen, w = 1, R1 und R2 Methyl und R3 und R4 jeweils unabhängig voneinander eine Kette -[-Xi-]n-H.
- (Ie) R sei 1,3-Propylen, w = 1, R1 und R2 Ethyl und R3 und R4 jeweils unabhängig voneinander eine Kette -[-Xi-]n-H.
- (If) R sei 1,3-Propylen, w = 1, R1 und R2 n-Butyl und R3 und R4 jeweils unabhängig voneinander eine Kette -[-Xi-]n-H.
- (Ig) R sei 1,2-Ethylen, w = 1, R1 bis R4 eine Kette -[-Xi-]n-H.
- (la) R is 1,2-ethylene, w=1, R 1 to R 3 are methyl and R 4 is a chain -[-X i -] n -H.
- (Ib) R is 1,2 -ethylene, w= 1 , R1 and R2 are methyl and R3 and R4 are each independently a chain -[-X i -] n -H.
- (Ic) R is 1,2-propylene, w=1, R 1 and R 2 are methyl and R 3 and R 4 are each independently a chain -[-X i -] n -H.
- (Id) R is 1,3-propylene, w=1, R 1 and R 2 are methyl and R 3 and R 4 are each independently a chain -[-X i -] n -H.
- (Ie) R is 1,3-propylene, w=1, R 1 and R 2 are ethyl and R 3 and R 4 are each independently a chain -[-X i -] n -H.
- (If) R is 1,3-propylene, w=1, R 1 and R 2 are n-butyl and R 3 and R 4 are each independently a chain -[-X i -] n -H.
- (Ig) R is 1,2-ethylene, w=1, R 1 to R 4 are a chain -[-X i -] n -H.
Unter diesen sind Verbindungen (Id) besonders bevorzugt.Among these, compounds (Id) are particularly preferred.
Folgende Verbindungen der Formel (II) sind bevorzugt:
- (IIa) R sei 1,2-Ethylen, x und y 1, z = 0 und R1 bis R4 und R6 jeweils unabhängig voneinander eine Kette -[-Xi-]n-H.
- (IIb) R sei 1,2-Propylen, x und y 1, z = 0 und R1 bis R4 und R6 jeweils unabhängig voneinander eine Kette -[-Xi-]n-H.
- (IIc) R sei 1,3-Propylen, x und y 1, z = 0 und R1 bis R4 jeweils unabhängig voneinander eine Kette -[-Xi-]n-H und R6 C1- bis C20-Alkyl.
- (IIa) R is 1,2-ethylene, x and y are 1, z=0 and R 1 to R 4 and R 6 are each independently a chain -[-X i -] n -H.
- (IIb) R is 1,2-propylene, x and y are 1, z=0 and R 1 to R 4 and R 6 are each independently a chain -[-X i -] n -H.
- (IIc) R is 1,3-propylene, x and y are 1, z = 0 and R 1 to R 4 are each independently a chain -[-X i -] n -H and R 6 is C 1 - to C 20 - alkyl.
Unter diesen sind Verbindungen (IIa) besonders bevorzugt.Among these, compounds (IIa) are particularly preferred.
Folgende Verbindungen der Formel (III) sind bevorzugt:
- (IIIa)
- R sei 1,2-Ethylen und R1 und R2 Methyl
- (IIIb)
- R sei 1,2-Ethylen und R1 und R2 Ethyl
- (IIIc)
- R sei 1,2-Ethylen und R1 und R2 n-Butyl
- (IIId)
- R sei 1,2-Ethylen und R1 und R2 gemeinsam eine 1,4-Butylenkette
- (IIIe)
- R sei 1,2-Ethylen und R1 und R2 gemeinsam eine 1,5-Pentylenkette
- (IIIf)
- R sei 1,2-Ethylen und R1 und R2 gemeinsam eine 3-Oxa-1,5-pentylenkette
- (IIIg)
- R sei 1,2-Propylen und R1 und R2 Methyl
- (IIIh)
- R sei 1,2-Propylen und R1 und R2 Ethyl
- (IIIi)
- R sei 1,2-Propylen und R1 und R2 n-Butyl
- (IIIj)
- R sei 1,2-Propylen und R1 und R2 gemeinsam eine 1,4-Butylenkette
- (IIIk)
- R sei 1,2-Propylen und R1 und R2 gemeinsam eine 1,5-Pentylenkette
- (IIII)
- R sei 1,2-Propylen und R1 und R2 gemeinsam eine 3-Oxa-1,5-pentylenkette
- (IIIm)
- R sei 1,3-Propylen und R1 und R2 Methyl
- (IIIn)
- R sei 1,3-Propylen und R1 und R2 Ethyl
- (IIIo)
- R sei 1,3-Propylen und R1 und R2 n-Butyl
- (IIIp)
- R sei 1,3-Propylen und R1 und R2 gemeinsam eine 1,4-Butylenkette
- (IIIq)
- R sei 1,3-Propylen und R1 und R2 gemeinsam eine 1,5-Pentylenkette
- (IIIr)
- R sei 1,3-Propylen und R1 und R2 gemeinsam eine 3-Oxa-1,5-pentylenkette
- (IIIa)
- R is 1,2-ethylene and R 1 and R 2 are methyl
- (IIIb)
- R is 1,2-ethylene and R 1 and R 2 are ethyl
- (IIIc)
- R is 1,2-ethylene and R 1 and R 2 are n-butyl
- (IIId)
- R is 1,2-ethylene and R 1 and R 2 together are a 1,4-butylene chain
- (IIIe)
- R is 1,2-ethylene and R 1 and R 2 together are a 1,5-pentylene chain
- (IIIf)
- R is 1,2-ethylene and R 1 and R 2 together are a 3-oxa-1,5-pentylene chain
- (IIIg)
- R is 1,2-propylene and R 1 and R 2 are methyl
- (IIIh)
- R is 1,2-propylene and R 1 and R 2 are ethyl
- (IIIi)
- R is 1,2-propylene and R 1 and R 2 are n-butyl
- (IIIj)
- R is 1,2-propylene and R 1 and R 2 together are a 1,4-butylene chain
- (IIIk)
- R is 1,2-propylene and R 1 and R 2 together are a 1,5-pentylene chain
- (III)
- R is 1,2-propylene and R 1 and R 2 together are a 3-oxa-1,5-pentylene chain
- (IIIm)
- R is 1,3-propylene and R 1 and R 2 are methyl
- (IIIn)
- R is 1,3-propylene and R 1 and R 2 are ethyl
- (IIIo)
- R is 1,3-propylene and R 1 and R 2 are n-butyl
- (IIIp)
- R is 1,3-propylene and R 1 and R 2 together are a 1,4-butylene chain
- (IIIq)
- R is 1,3-propylene and R 1 and R 2 together are a 1,5-pentylene chain
- (IIIr)
- R is 1,3-propylene and R 1 and R 2 together are a 3-oxa-1,5-pentylene chain
Unter diesen sind Verbindungen (IIIg) besonders bevorzugt.Among these, compounds (IIIg) are particularly preferred.
Besonders bevorzugt sind die Umsetzungsprodukte von 1,2-Ethylendiamin, Diethylentriamin, Triethylentetramin oder 3-(Dimethylamino)propylamin, bevorzugt Diethylentriamin oder 3-(Dimethylamino)propylamin und besonders bevorzugt 3-(Dimethylamino)propylamin mit 10 bis 30, bevorzugt 12 bis 27 und besonders bevorzugt 15 bis 25 Äquivalenten Propylenoxid und/oder 1,2-Butylenoxid.The reaction products of 1,2-ethylenediamine, diethylenetriamine, triethylenetetramine or 3-(dimethylamino)propylamine, preferably diethylenetriamine or 3-(dimethylamino)propylamine and particularly preferably 3-(dimethylamino)propylamine with 10 to 30, preferably 12 to 27, are particularly preferred and more preferably 15 to 25 equivalents of propylene oxide and/or 1,2-butylene oxide.
Die beschriebenen Verbindungen werden erfindungsgemäß verwendet, um beim Betrieb von direkteinspritzenden Ottomotoren mit Ottokraftstoffen die Bildung von Ablagerungen an direkteinspritzenden Einspritzdüsen zu vermeiden oder zu verringern und/oder bestehende Ablagerungen zu entfernen oder zu verringern.The compounds described are used according to the invention in order to avoid or reduce the formation of deposits on direct-injection injection nozzles and/or to remove or reduce existing deposits when operating direct-injection Otto engines with Otto fuels.
Es stellt einen Vorteil der beschriebenen Verbindungen dar, daß diese alternativ oder zusätzlich zu dieser beschriebenen Verwendung als Trägeröl wirken können, so daß man die Menge an Trägeröl in den Additivpaketen (siehe unten) verringern kann.It is an advantage of the described compounds that they can act as an alternative or in addition to this described use as a carrier oil, so that one can reduce the amount of carrier oil in the additive packages (see below).
Es stellt einen weiteren Vorteil der beschriebenen Verbindungen dar, daß diese alternativ oder zusätzlich zu diesen beschriebenen Verwendungen als Demulsifier wirken können.Another advantage of the compounds described is that they can act as demulsifiers as an alternative or in addition to the uses described.
Ein weiterer Gegenstand der vorliegenden Erfindung sind Kraftstoffadditivkonzentrate gemäss Anspruch 14.Another object of the present invention are fuel additive concentrates according to claim 14.
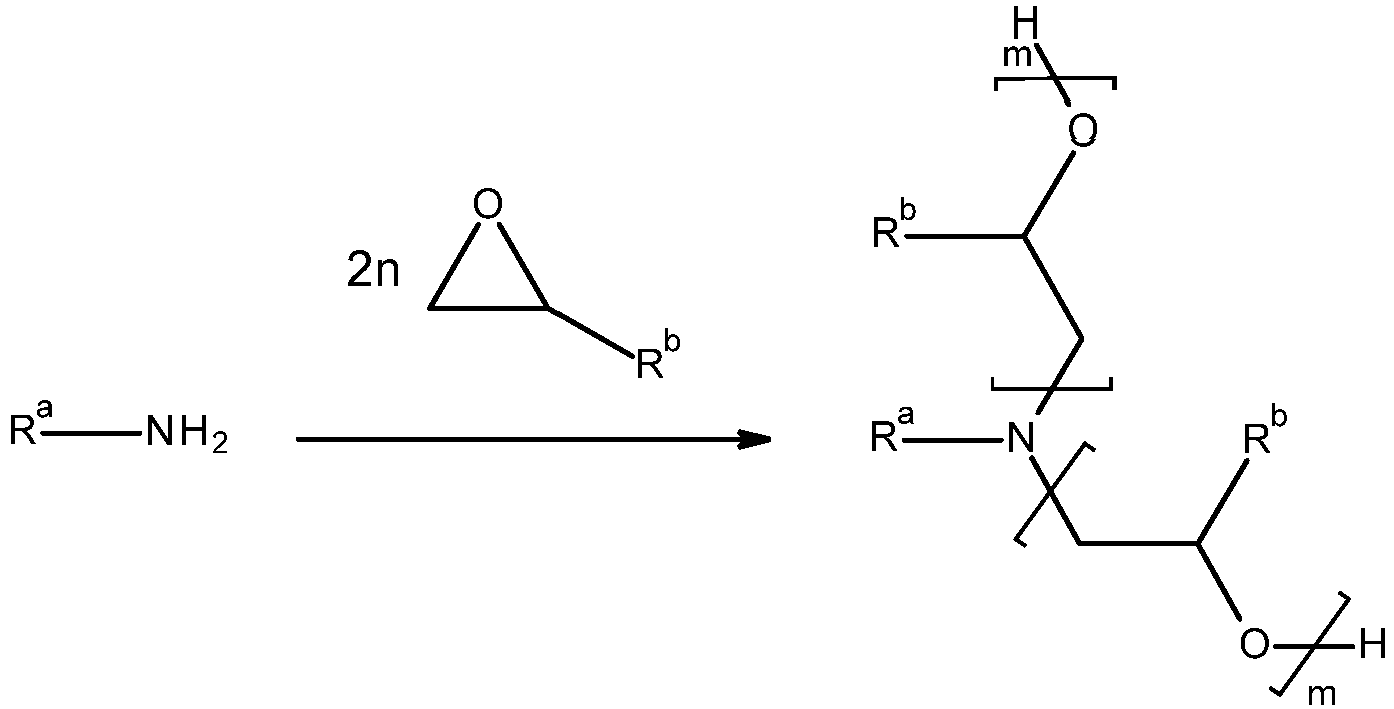
Die Herstellung von alkoxylierten Aminen (Amine mit primären und/oder sekundären Aminogruppen) ist prinzipiell bekannt, beispielsweise wird sie in
Üblicherweise erfolgt die Alkoxylierung von Aminen und Polyaminen zweistufig: Zunächst wird soviel Alkylenoxid eingesetzt, dass ein durchschnittlicher Alkoxylierungsgrad von 0,5 bis 1,5, bevorzugt von 0,75 bis 1,25 mol Alkylenoxid pro NH-Funktion erreicht wird. Dieser Schritt wird üblicherweise in Gegenwart von Wasser (bevorzugt 0,5-10,0 Gew-% bezogen auf das eingesetzte Amin) durchgeführt, kann aber auch in Abwesenheit von Wasser erfolgen. Die Umsetzung erfolgt üblicherweise bei einer Temperatur von 50°C bis 180°C, bevorzugt 90°C bis 160°C. Das Alkylenoxid wird bevorzugt in einem Zeitraum von 1 bis 10 h dosiert.The alkoxylation of amines and polyamines usually takes place in two stages: first, enough alkylene oxide is used to achieve an average degree of alkoxylation of from 0.5 to 1.5, preferably from 0.75 to 1.25, mol of alkylene oxide per NH function. This step is usually carried out in the presence of water (preferably 0.5-10.0% by weight based on the amine used), but can also be carried out in the absence of water. The reaction usually takes place at a temperature of from 50.degree. C. to 180.degree. C., preferably from 90.degree. C. to 160.degree. The alkylene oxide is preferably metered in over a period of 1 to 10 hours.
Der weitere Aufbau von Polyalkylenoxidketten erfolgt unter Basenkatalyse nach Entfernung des Lösungsmittels, insbesondere Wasser. Als basische Katalysatoren werden beispielsweise eingesetzt: Kaliumhydroxid, Natriumhydroxid, Kaliummethanolat oder Natriummethanolat, bevorzugt Kaliumhydroxid oder Natriumhydroxid.The further build-up of polyalkylene oxide chains takes place under base catalysis after removal of the solvent, in particular water. Examples of basic catalysts used are: Potassium hydroxide, sodium hydroxide, potassium methoxide or sodium methoxide, preferably potassium hydroxide or sodium hydroxide.
Der Aufbau der alkoxylierten Amine kann aber auch einstufig erfolgen, wobei als mögliche Katalysatoren Kaliumhydroxid, Natriumhydroxid, Kaliummethanolat, Natriummethanolat, Kaliumacetat oder Natriumacetat eingesetzt werden können.However, the synthesis of the alkoxylated amines can also take place in one stage, with potassium hydroxide, sodium hydroxide, potassium methoxide, sodium methoxide, potassium acetate or sodium acetate being able to be used as possible catalysts.
Die Umsetzung kann prinzipiell aber auch ohne Katalysator erfolgen, da das eingesetzte Amin selbst die Alkoxylierung katalysieren kann (siehe Mihail Ionescu, Chemistry and Technology of Polyols for Polyurethanes, Rapra Technology Limited, 2005).In principle, however, the reaction can also take place without a catalyst, since the amine used can itself catalyze the alkoxylation (see Mihail Ionescu, Chemistry and Technology of Polyols for Polyurethanes, Rapra Technology Limited, 2005).
Die Alkyenoxide enthalten in der Regel 2 oder mehr Kohlenstoffatome, bevorzugt 2 bis 20 Kohlenstoffatome, insbesondere von 2 bis 12 Kohlenstoffatomen. Mögliche Alkylenoxide sind Ethylenoxid, Propylenoxid, iso-Butylenoxid, 1,2-Butylenoxid, 2,3-Butylenoxid, 1,2-Pentenoxid, 1,2-Dodecenoxid, Styroloxid. Mögliche Alkylenoxide umfassen auch Glycidylether, beispielsweise 2-Ethylhexylglycidylether. Besonders bevorzugt sind Propylenoxid und 1,2-Butylenoxid.The alkylene oxides usually contain 2 or more carbon atoms, preferably 2 to 20 carbon atoms, in particular from 2 to 12 carbon atoms. Possible alkylene oxides are ethylene oxide, propylene oxide, isobutylene oxide, 1,2-butylene oxide, 2,3-butylene oxide, 1,2-pentene oxide, 1,2-dodecene oxide, styrene oxide. Possible alkylene oxides also include glycidyl ethers, for example 2-ethylhexyl glycidyl ether. Propylene oxide and 1,2-butylene oxide are particularly preferred.
Wurde ein metallhaltiger Katalysator verwendet wird das Metall für eine Anwendung in Kraft- oder Schmierstoffen vorteilhafterweise aus dem Produkt entfernt. Diese Methoden sind prinzipiell bekannt und sind beschrieben in Mihail Ionescu, Chemistry and Technology of Polyols for Polyurethanes, Rapra Technology Limited, 2005. So können Alkali- oder Erdalkalimetalle durch Adsorption an Magnesiumsilikaten (Handelsprodukte Ambosol® oder Magnesol®) entfernt werden. Kaliumionen können durch Fällung mit Phosphorsäure als Kaliumhydrogenphosphat und anschließender Filtration entfernt werden. Alkali- oder Erdalkalimetalle können auch mit Hilfe von regenerierbaren lonentauschern entfernt werden, wobei das alkoxylierte Amin auch in einem Lösungsmittel gelöst werden kann.If a metal-containing catalyst has been used, the metal is advantageously removed from the product for use in fuels or lubricants. These methods are known in principle and are described in Mihail Ionescu, Chemistry and Technology of Polyols for Polyurethanes, Rapra Technology Limited, 2005. Thus, alkali metals or alkaline earth metals can be removed by adsorption on magnesium silicates (commercial products Ambosol® or Magnesol® ). Potassium ions can be removed as potassium hydrogen phosphate by precipitation with phosphoric acid and subsequent filtration. Alkali metals or alkaline earth metals can also be removed with the aid of regenerable ion exchangers, in which case the alkoxylated amine can also be dissolved in a solvent.
Die erfindungsgemäßen Verbindungen (A) können den zu additivierenden Kraftstoffen einzeln oder im Gemisch mit weiteren wirksamen Additivkomponenten (Co-Additiven) zugesetzt werden.The compounds (A) according to the invention can be added to the fuels to which additives are to be added individually or as a mixture with other active additive components (co-additives).
Als Beispiele können Additive mit Detergenswirkung und/oder mit Ventilsitzverschleißhemmender Wirkung (im folgenden bezeichnet als Detergensadditive) genannt werden. Dieses Detergensadditiv besitzt mindestens einen hydrophoben Kohlenwasserstoffrest mit einem zahlengemittelten Molekulargewicht (Mn) von 85 bis 20 000 und mindestens eine polare Gruppierung ausgewählt aus:
- (a) Mono- oder Polyaminogruppen mit bis zu 6 Stickstoffatomen, wobei mindestens ein Stickstoffatom basische Eigenschaften hat;
- (b) Nitrogruppen, ggf. in Kombination mit Hydroxylgruppen;
- (c) Hydroxylgruppen in Kombination mit Mono- oder Polyaminogruppen, wobei mindestens ein Stickstoffatom basische Eigenschaften hat;
- (d) Carboxylgruppen oder deren Alkalimetall- oder Erdalkalimetallsalzen;
- (e) Sulfonsäuregruppen oder deren Alkalimetall- oder Erdalkalimetallsalzen;
- (f) Polyoxy-C2- bis C4-alkylengruppierungen, die durch Hydroxylgruppen, Mono- oder Polyaminogruppen, wobei mindestens ein Stickstoffatom basische Eigenschaften hat, oder durch Carbamatgruppen terminiert sind;
- (g) Carbonsäureestergruppen;
- (h) aus Bernsteinsäureanhydrid abgeleiteten Gruppierungen mit Hydroxy- und/oder Amino- und/oder Amido- und/oder Imidogruppen; und/oder
- (i) durch Mannich-Umsetzung von substituierten Phenolen mit Aldehyden und Mono- oder Polyaminen erzeugten Gruppierungen;
- (a) mono- or polyamino groups containing up to 6 nitrogen atoms, where at least one nitrogen atom has basic properties;
- (b) nitro groups, optionally in combination with hydroxyl groups;
- (c) hydroxyl groups in combination with mono- or polyamino groups, where at least one nitrogen atom has basic properties;
- (d) carboxyl groups or their alkali metal or alkaline earth metal salts;
- (e) sulfonic acid groups or their alkali metal or alkaline earth metal salts;
- (f) polyoxy-C 2 - to C 4 -alkylene groups terminated by hydroxyl groups, mono- or polyamino groups, where at least one nitrogen atom has basic properties, or by carbamate groups;
- (g) carboxylic acid ester groups;
- (h) moieties derived from succinic anhydride having hydroxy and/or amino and/or amido and/or imido groups; and or
- (i) moieties generated by Mannich reactions of substituted phenols with aldehydes and mono- or polyamines;
Der hydrophobe Kohlenwasserstoffrest in den obigen Detergensadditiven, welcher für die ausreichende Löslichkeit im Kraftstoff sorgt, hat ein zahlengemitteltes Molekulargewicht (Mn) von 85 bis 20,000, insbesondere von 113 bis 10000, vor allem von 300 bis 5000. Als typischer hydrophober Kohlenwasserstoffrest, insbesondere in Verbindung mit den polaren Gruppierungen (a), (c), (h) und (i), kommen der Polypropenyl-, Polybutenyl- und Polyisobutenylrest mit jeweils Mn = 300 bis 5000, insbesondere 500 bis 2500, vor allem 700 bis 2300, in Betracht.The hydrophobic hydrocarbon group in the above detergent additives which provides sufficient solubility in fuel has a number average molecular weight (Mn) of from 85 to 20,000, especially from 113 to 10,000, especially from 300 to 5000. As a typical hydrophobic hydrocarbon group, especially in combination with the polar groups (a), (c), (h) and (i), the polypropenyl, polybutenyl and polyisobutenyl radicals, each with Mn=300 to 5000, in particular 500 to 2500, especially 700 to 2300, are suitable .
Als Beispiele für obige Gruppen von Detergensadditiven seien die folgenden genannt:
Mono- oder Polyaminogruppen (a) enthaltende Additive sind vorzugsweise Polyalkenmono- oder Polyalkenpolyamine auf Basis von Polypropen oder konventionellem (d.h. mit überwiegend mittenständigen Doppelbindungen) Polybuten oder Polyisobuten mit Mn = 300 bis 5000. Geht man bei der Herstellung der Additive von Polybuten oder Polyisobuten mit überwiegend mittenständigen Doppelbindungen (meist in der beta-und gamma-Position) aus, bietet sich der Herstellweg durch Chlorierung und anschließende Aminierung oder durch Oxidation der Doppelbindung mit Luft oder Ozon zur Carbonyl- oder Carboxylverbindung und anschließende Aminierung unter reduktiven (hydrierenden) Bedingungen an. Zur Aminierung können hier Amine, wie z.B. Ammoniak, Monoamine oder Polyamine, wie Dimethylaminopropylamin, Ethylendiamin, Diethylentriamin, Triethylentetramin oder Tetraethylenpentamin, eingesetzt werden. Entsprechende Additive auf Basis von Polypropen sind insbesondere in der
Additives containing mono- or polyamino groups (a) are preferably polyalkenemono- or polyalkenepolyamines based on polypropene or conventional (ie with predominantly central double bonds) polybutene or polyisobutene with Mn=300 to 5000. If polybutene or polyisobutene is included in the preparation of the additives predominantly central double bonds (usually in the beta and gamma position), the production route by chlorination and subsequent amination or by oxidation of the double bond with air or ozone to form the carbonyl or carboxyl compound and subsequent amination under reductive (hydrogenating) conditions is an option. Amines such as ammonia, monoamines or polyamines such as dimethylaminopropylamine, ethylenediamine, diethylenetriamine, triethylenetetramine or tetraethylenepentamine can be used for the amination. Corresponding additives based on polypropylene are in particular in the
Weitere bevorzugte Monoaminogruppen (a) enthaltende Additive sind die Hydrierungsprodukte der Umsetzungsprodukte aus Polyisobutenen mit einem mittleren Polymerisationsgrad P = 5 bis 100 mit Stickoxiden oder Gemischen aus Stickoxiden und Sauerstoff, wie sie insbesondere in
Weitere bevorzugte Monoaminogruppen (a) enthaltende Additive sind die aus Polyisobutenepoxiden durch Umsetzung mit Aminen und nachfolgender Dehydratisierung und Reduktion der Aminoalkohole erhältlichen Verbindungen, wie sie insbesondere in
Nitrogruppen (b), ggf. in Kombination mit Hydroxylgruppen, enthaltende Additive sind vorzugsweise Umsetzungsprodukte aus Polyisobutenen des mittleren Polymerisationsgrades P = 5 bis 100 oder 10 bis 100 mit Stickoxiden oder Gemischen aus Stickoxiden und Sauerstoff, wie sie insbesondere in
Hydroxylgruppen in Kombination mit Mono- oder Polyaminogruppen (c) enthaltende Additive sind insbesondere Umsetzungsprodukte von Polyisobutenepoxiden, erhältlich aus vorzugsweise überwiegend endständige Doppelbindungen aufweisendem Polyisobuten mit Mn = 300 bis 5000, mit Ammoniak, Mono- oder Polyaminen, wie sie insbesondere in
Carboxylgruppen oder deren Alkalimetall- oder Erdalkalimetallsalze (d) enthaltende Additive sind vorzugsweise Copolymere von C2-C40-Olefinen mit Maleinsäureanhydrid mit einer Gesamt-Molmasse von 500 bis 20 000, deren Carboxylgruppen ganz oder teilweise zu den Alkalimetall- oder Erdalkalimetallsalzen und ein verbleibender Rest der Carboxylgruppen mit Alkoholen oder Aminen umgesetzt sind. Solche Additive sind insbesondere aus der
Sulfonsäuregruppen oder deren Alkalimetall- oder Erdalkalimetallsalze (e) enthaltende Additive sind vorzugsweise Alkalimetall- oder Erdalkalimetallsalze eines Sulfobernsteinsäurealkylesters, wie er insbesondere in der
Polyoxy-C2-C4-alkylengruppierungen (f) enthaltende Additive sind vorzugsweise Polyether oder Polyetheramine, welche durch Umsetzung von C2-C60-Alkanolen, C6-C30-Alkandiolen, Mono- oder Di-C2-C30-alkylaminen, C1-C30-Alkylcyclohexanolen oder C1-C30-Alkylphenolen mit 1 bis 30 mol Ethylenoxid und/oder Propylenoxid und/oder Butylenoxid pro Hydroxylgruppe oder Aminogruppe und, im Falle der Polyetheramine, durch anschließende reduktive Aminierung mit Ammoniak, Monoaminen oder Polyaminen erhältlich sind. Derartige Produkte werden insbesondere in
Carbonsäureestergruppen (g) enthaltende Additive sind vorzugsweise Ester aus Mono-, Di- oder Tricarbonsäuren mit langkettigen Alkanolen oder Polyolen, insbesondere solche mit einer Mindestviskosität von 2 mm2/s bei 100°C, wie sie insbesondere in
Aus Bernsteinsäureanhydrid abgeleitete Gruppierungen mit Hydroxy- und/oder Amino- und/oder Amido- und/oder Imidogruppen (h) enthaltende Additive sind vorzugsweise entsprechende Derivate von Polyisobutenylbernsteinsäureanhydrid, welche durch Umsetzung von konventionellem oder hochreaktivem Polyisobuten mit Mn = 300 bis 5000 mit Maleinsäureanhydrid auf thermischen Wege oder über das chlorierte Polyisobuten erhältlich sind. Von besonderem Interesse sind hierbei Derivate mit aliphatischen Polyaminen wie Ethylendiamin, Diethylentriamin, Triethylentetramin oder Tetraethylenpentamin. Derartige Ottokraftstoffadditive sind insbesondere in
Durch Mannich-Umsetzung von substituierten Phenolen mit Aldehyden und Mono- oder Polyaminen erzeugte Gruppierungen (i) enthaltende Additive sind vorzugsweise Umsetzungsprodukte von polyisobutensubstituierten Phenolen mit Formaldehyd und Mono- oder Polyaminen wie Ethylendiamin, Diethylentriamin, Triethylentetramin, Tetraethylenpentamin oder Dimethylaminopropylamin. Die polyisobutenylsubstituierten Phenole können aus konventionellem oder hochreaktivem Polyisobuten mit Mn = 300 bis 5000 stammen. Derartige "Polyisobuten-Mannichbasen" sind insbesondere in der
Zur genaueren Definition der einzelnen aufgeführten Ottokraftstoffadditive wird hier auf die Offenbarungen der obengenannten Schriften des Standes der Technik ausdrücklich Bezug genommen.For a more precise definition of the individual gasoline additives listed, reference is expressly made here to the disclosures of the above-mentioned documents of the prior art.
Die erfindungsgemäßen Additiv-Formulierungen können darüber hinaus mit noch weiteren üblichen Komponenten und Additiven kombiniert werden. Hier sind in erster Linie Trägeröle ohne ausgeprägte Detergenswirkung zu nennen.The additive formulations according to the invention can also be combined with other customary components and additives. The main ones to be mentioned here are carrier oils without a pronounced detergent effect.
Geeignete mineralische Trägeröle sind bei der Erdölverarbeitung anfallende Fraktionen, wie Brightstock oder Grundöle mit Viskositäten wie beispielsweise aus der Klasse SN 500 - 2000; aber auch aromatische Kohlenwasserstoffe, paraffinische Kohlenwasserstoffe und Alkoxyalkanole. Brauchbar ist ebenfalls eine als "hydrocrack oil" bekannte und bei der Raffination von Mineralöl anfallende Fraktion (Vakuumdestillatschnitt mit einem Siedebereich von etwa 360 bis 500°C, erhältlich aus unter Hochdruck katalytisch hydriertem und isomerisiertem sowie entparaffiniertem natürlichen Mineralöl). Ebenfalls geeignet sind Mischungen oben genannter mineralischer Trägeröle.Suitable mineral carrier oils are fractions obtained during petroleum processing, such as bright stock or base oils with viscosities such as those from the class SN 500-2000; but also aromatic hydrocarbons, paraffinic hydrocarbons and alkoxyalkanols. A fraction known as "hydrocrack oil" and obtained in the refining of mineral oil (vacuum distillate cut with a boiling range of about 360 to 500°C, obtainable from natural mineral oil which has been catalytically hydrogenated and isomerized and dewaxed under high pressure) can also be used. Mixtures of the mineral carrier oils mentioned above are also suitable.
Beispiele für erfindungsgemäß verwendbare synthetische Trägeröle sind ausgewählt unter: Polyolefinen (Polyalphaolefine oder Polyinternalolefine), (Poly)estern, (Poly)alkoxylaten, Polyethern, aliphatischen Polyetheraminen, alkylphenolgestarteten Polyethern, alkylphenolgestarteten Polyetheraminen und Carbonsäureester langkettiger Alkanole.Examples of synthetic carrier oils that can be used according to the invention are selected from: polyolefins (polyalphaolefins or polyinternal olefins), (poly)esters, (poly)alkoxylates, polyethers, aliphatic polyetheramines, alkylphenol-started polyethers, alkylphenol-started polyetheramines and carboxylic acid esters of long-chain alkanols.
Beispiele für besonders geeignete synthetische Trägeröle sind alkoholgestartete Polyether mit etwa 5 bis 35 C3- bis C6-Alkylenoxideinheiten, meist ausgewählt unter Propylenoxid-, n-Butylenoxid- und iso-Butylenoxid-Einheiten oder Gemischen davon. Nichtlimitierende Beispiele für hierfür geeignete Starteralkohole sind langkettige Alkanole oder mit langkettigem Alkyl substituierte Phenole, wobei der langkettige Alkylrest insbesondere für einen geradkettigen oder verzweigten C6- bis C18-Alkylrest steht. Als bevorzugte Beispiele sind hierfür Tridecanol, Heptadecanol und Nonylphenol zu nennen.Examples of particularly suitable synthetic carrier oils are alcohol-started polyethers with about 5 to 35 C 3 - to C 6 -alkylene oxide units, mostly selected from propylene oxide, n-butylene oxide and isobutylene oxide units or mixtures thereof. Non-limiting examples of starter alcohols suitable for this are long-chain alkanols or phenols substituted with long-chain alkyl, where the long-chain alkyl radical is in particular a straight-chain or branched C 6 - to C 18 -alkyl radical. Preferred examples of this are tridecanol, heptadecanol and nonylphenol.
Durch den Einsatz der erfindungsgemäßen Verbindungen (A) kann oftmals der Einsatz von Trägerölen verringert werden.The use of the compounds (A) according to the invention can often reduce the use of carrier oils.
Beispiele für geeignete Polyolefine sind Olefinpolymerisate mit Mn = 400 bis 1800, vor allem auf Polybuten- oder Polyisobuten-Basis (hydriert oder nicht hydriert).Examples of suitable polyolefins are olefin polymers with Mn=400 to 1800, primarily based on polybutene or polyisobutene (hydrogenated or non-hydrogenated).
Beispiele für geeignete Polyether oder Polyetheramine sind vorzugsweise Polyoxy-C2-C4-alkylengruppierungen enthaltende Verbindungen, welche durch Umsetzung von C2-C60-Alkanolen, C6-C30-Alkandiolen, Mono- oder Di-C2-C30-alkylaminen, C1-C30-Alkylcyclohexanolen oder C1-C30-Alkylphenolen mit 1 bis 30 mol Ethylenoxid und/ oder Propylenoxid und/oder Butylenoxid pro Hydroxylgruppe oder Aminogruppe und, im Falle der Polyetheramine, durch anschließende reduktive Aminierung mit Ammoniak, Monoaminen oder Polyaminen erhältlich sind. Derartige Produkte werden insbesondere in
Beispiele für Carbonsäureester langkettiger Alkanole sind insbesondere Ester aus Mono-, Di- oder Tricarbonsäuren mit langkettigen Alkanolen oder Polyolen, wie sie insbesondere in der
Weitere geeignete Trägerölsysteme sind beispielsweise beschrieben in
Beispiele für besonders geeignete synthetische Trägeröle sind alkoholgestartete Polyether mit etwa 5 bis 35, bevorzugt etwa 5 bis 30, besonders bevorzugt 7 bis 25, C3-C6-Alkylenoxideinheiten, wie z.B. ausgewählt unter Propylenoxid-, n-Butylenoxid- und i-Butylenoxid-Einheiten, oder Gemischen davon, bevorzugt ausgewählt unter Propylenoxid- und i-Butylenoxid-Einheiten. Nichtlimitierende Beispiele für geeignete Starteralkohole sind langkettige Alkanole oder mit langkettigem Alkyl substituierte Phenole, wobei der langkettige Alkylrest insbesondere für einen geradkettigen oder verzweigten C6-C18-Alkylrest steht. Als bevorzugte Beispiele für Alkanole sind zu nennen Decanol, Tridecanol, Heptadecanol und Nonylphenol, besonders bevorzugt verzweigte Decanol, Tridecanol und Heptadecanol.Examples of particularly suitable synthetic carrier oils are alcohol-started polyethers having about 5 to 35, preferably about 5 to 30, particularly preferably 7 to 25, C 3 -C 6 -alkylene oxide units, such as selected from propylene oxide, n-butylene oxide and i-butylene oxide units, or mixtures thereof, preferably selected from propylene oxide and i-butylene oxide units. Non-limiting examples of suitable starter alcohols are long-chain alkanols or phenols substituted with long-chain alkyl, where the long-chain alkyl radical is in particular a straight-chain or branched C 6 -C 18 -alkyl radical. Preferred examples of alkanols are decanol, tridecanol, heptadecanol and nonylphenol, particularly preferably branched decanol, tridecanol and heptadecanol.
Weitere geeignete synthetische Trägeröle sind alkoxylierte Alkylphenole, wie sie in der
Weitere übliche Additive sind Korrosionsinhibitoren, beispielsweise auf Basis von zur Filmbildung neigenden Ammoniumsalzen organischer Carbonsäuren oder von heterocyclischen Aromaten im Falle von Buntmetallkorrosionsschutz; bevorzugte Korrosionsinhibitoren sind Mono-, Di- und Polycarbonsäuren, die mindestens 12 Kohlenstoffatome, bevorzugt mindestens 14, besonders bevorzugt mindestens 16 und ganz besonders bevorzugt mindestens 18 Kohlenstoffatome aufweisen und bevorzugt außer Kohlenwasserstoffresten und Carboxylgruppen keine weiteren Funktionalitäten aufweisen. Beispiele dafür sind Fettsäuren, Dimerfettsäuren, Alkyl- und Alkenylbernsteinsäuren sowie hydrolysierte Olefin-Maleinsäureanhydrid-Copolymere, bevorzugt sind Dodecansäure (Laurinsäure), Tridecansäure, Tetradecansäure (Myristinsäure), Pentadecansäure, Palmitinsäure (Hexadecansäure), Margarinsäure (Heptadecansäure), Stearinsäure (Octadecansäure), Nonadecansäure, Arachinsäure (Eicosansäure), Behensäure (Docosansäure), Tetracosansäure (Lignocerinsäure), Cerotinsäure (Hexacosansäure), Triacontansäure (Melissinsäure), Palmitoleinsäure [(9Z)-Hexadec-9-ensäure], Ölsäure [(9Z)-Octadec-9-ensäure], Elaidinsäure [(9E)-Octadec-9-ensäure], Erucasäure [(13Z)-Docos-13-ensäure], Linolsäure [(9Z,12Z)-Octadeca-9,12-diensäure], Linolensäure [(9Z,12Z,15Z)-Octadeca-9,12,15-triensäure] und Elaeostearinsäure [(9Z,11E,13E)-Octadeca-9,11,13-triensäure], dimere Ölsäure (
Weiterhin zu nennen sind Antioxidantien oder Stabilisatoren, beispielsweise auf Basis von Aminen wie p-Phenylendiamin, Dicyclohexylamin oder Derivaten hiervon oder von Phenolen wie 2,4-Di-tert.-butylphenol oder 3,5-Di-tert.-butyl-4-hydroxyphenylpropionsäure; Demulgatoren; Antistatikmittel; Metallocene wie Ferrocen; Methylcyclopentadienylmangantricarbonyl; Schmierfähigkeitsverbesserer (andere als die erfindungsgemäßen Triazole) wie bestimmte Fettsäuren, Alkenylbernsteinsäureester, Bis(hydroxyalkyl)fettamine, Hydroxyacetamide oder Ricinusöl; sowie Farbstoffe (Marker). Gegebenenfalls werden auch Amine zur Absenkung des pH-Wertes des Kraftstoffes zugesetzt.Mention should also be made of antioxidants or stabilizers, for example based on amines such as p-phenylenediamine, dicyclohexylamine or derivatives thereof, or on phenols such as 2,4-di-tert-butylphenol or 3,5-di-tert-butyl-4- hydroxyphenylpropionic acid; demulsifiers; antistatic agents; metallocenes such as ferrocene; methylcyclopentadienyl manganese tricarbonyl; lubricity improvers (other than the triazoles of this invention) such as certain fatty acids, alkenyl succinic acid esters, bis(hydroxyalkyl) fatty amines, hydroxyacetamides, or castor oil; and dyes (markers). If necessary, amines are also added to lower the pH of the fuel.
Beispiele für Lösungsmittel sind beispielsweise Aromaten, wie Solvent Naphtha, bevorzugt Naphthalin-abgereichertes Solvent Naphtha, Benzol, Toluol, Xylol, Kerosin mit einem Aromatengehalt unter 50 Vol%, einem Schwefelgehalt unter 10 mg/kg und einen Flammpunkt nach DIN EN ISO 13736: 2000-04 über 60 °C sowie höhere Alkohole, die einen Flammpunkt nach DIN EN ISO 13736: 2000-04 über 60 °C aufweisen, bevorzugt 2-Ethylhexanol oder 2-Propylheptanol.Examples of solvents are, for example, aromatics, such as solvent naphtha, preferably naphthalene-depleted solvent naphtha, benzene, toluene, xylene, kerosene with an aromatics content of less than 50% by volume, a sulfur content of less than 10 mg/kg and a flash point according to DIN EN ISO 13736: 2000 -04 above 60 °C and higher alcohols which have a flash point according to DIN EN ISO 13736: 2000-04 above 60 °C, preferably 2-ethylhexanol or 2-propylheptanol.
Die Komponenten bzw. Additive können dem Kraftstoff einzeln oder als vorher zubereitetes Konzentrat (Additivpaket) zusammen mit der erfindungsgemäßen Verbindung (A) zugegeben werden.The components or additives can be added to the fuel individually or as a previously prepared concentrate (additive package) together with the compound (A) according to the invention.
Die genannten Detergensadditive (B1) mit den polaren Gruppierungen (a) bis (i) werden dem Kraftstoff üblicherweise in einer Menge von 50 bis 1000 Gew.-ppm, zugegeben. Die sonstigen erwähnten Komponenten und Additive werden, wenn gewünscht, in hierfür üblichen Mengen zugesetzt. raftstoffadditivkonzentrate enthalten
- (A) mindestens eine Verbindung der Formel
- (B1) mindestens eine Verbindung (B1a), bei der es sich um ein Polyisobutenamin handelt, erhältlich durch Hydroformylierung und anschließender reduktiver Aminierung, ganz besonders bevorzugt mit Ammoniak, von Polyisobuten mit Mn = 300 bis 5000,
- (B2) mindestens ein Trägeröl, sowie
- (B3) mindestens einen Korrosionsinhibitor. Kraftstoffadditivkonzentrate sind zusammengesetzt wie folgt,
- (A) 1 - 50 Gew%, bevorzugt 2 - 40 Gew%, besonders bevorzugt 5 - 30 Gew%,
- (B1) 20 - 96,75 Gew%, bevorzugt 40 - 94,5 Gew%, besonders bevorzugt 60 - 89 Gew%,
- (B2) 2 - 30 Gew%, bevorzugt 3 - 25 Gew%, besonders bevorzugt 5 - 20 Gew%, sowie
- (B3) 0,25 - 10 Gew%, bevorzugt 0,5 - 7,5 Gew%, besonders bevorzugt 1 - 5 Gew%, sowie optional weitere Coadditive,
- (A) at least one compound of formula
- (B1) at least one compound (B1a), which is a polyisobuteneamine, obtainable by hydroformylation and subsequent reductive amination, very particularly preferably with ammonia, of polyisobutene with Mn=300 to 5000,
- (B2) at least one carrier oil, and
- (B3) at least one corrosion inhibitor. Fuel additive concentrates are composed as follows,
- (A) 1-50% by weight, preferably 2-40% by weight, particularly preferably 5-30% by weight,
- (B1) 20-96.75% by weight, preferably 40-94.5% by weight, particularly preferably 60-89% by weight,
- (B2) 2-30% by weight, preferably 3-25% by weight, particularly preferably 5-20% by weight, and
- (B3) 0.25-10% by weight, preferably 0.5-7.5% by weight, particularly preferably 1-5% by weight, and optionally further coadditives,
Die erfindungsgemäßen Additivzusammensetzungen sind in allen herkömmlichen Ottokraftstoffen, wie sie beispielsweise in Ullmann's Encyclopedia of Industrial Chemistry, 5. Aufl. 1990, Band A16, S. 719 ff. beschrieben sind, verwendbar.The additive compositions according to the invention can be used in all conventional gasoline fuels, as described, for example, in Ullmann's Encyclopedia of Industrial Chemistry, 5th edition 1990, volume A16, p. 719 et seq.
Daher stellen einen weiteren Gegenstand der vorliegenden Erfindung Kraftstoffzusammensetzungen gemäss Anspruch 15 dar.Therefore, a further subject of the present invention are fuel compositions according to claim 15.
Der Ottokraftstoff ist bevorzugt mindestens ein Ottokraftstoff gemäß DIN EN 228.The petrol is preferably at least one petrol according to DIN EN 228.
Zum Beispiel ist die Verwendung in einem Ottokraftstoff mit einem Aromatengehalt von maximal 60, wie z.B. maximal 42 oder maximal 35 Vol.-% und/oder einem Schwefelgehalt von maximal 2000, wie z.B. maximal 150 oder maximal 10 Gew.-ppm möglich.For example, use in a gasoline with an aromatics content of no more than 60, such as no more than 42 or no more than 35% by volume and/or a sulfur content of no more than 2000, such as no more than 150 or no more than 10 ppm by weight, is possible.
Der Aromatengehalt des Ottokraftstoffes beträgt beispielsweise10 bis 50, wie z.B. 30 bis 42 Vol.-%, insbesondere 32 bis 40 Vol.-%, oder maximal 35 Vol.-%. Der Schwefelgehalt des Ottokraftstoffes beträgt beispielsweise 2 bis 500, wie z.B. 5 bis 100Gew.-ppm, oder maximal10 Gew.-ppm.The aromatics content of petrol is, for example, 10 to 50% by volume, such as 30 to 42% by volume, in particular 32 to 40% by volume, or a maximum of 35% by volume. The sulfur content of petrol is, for example, 2 to 500, such as 5 to 100 ppm by weight, or a maximum of 10 ppm by weight.
Weiterhin kann der Ottokraftstoff beispielsweise einen Olefingehalt bis zu 50 Vol.-%, wie z.B. von 6 bis 21 Vol.-%, insbesondere 7 bis 18 Vol.-%; einen Benzolgehalt von bis zu 5 Vol.-%, wie z.B. 0,5 bis 1,0 Vol.-%, insbesondere 0,6 bis 0,9 Vol.-% und/oder einen Sauerstoffgehalt von bis zu 25 Vol.-%, wie z.B. bis zu 10 Gew.-% oder 1,0 bis 2,7 Gew.-%, insbesondere von 1,2 bis 2,0 Gew.-%, aufweisen.Furthermore, the gasoline can, for example, have an olefin content of up to 50% by volume, such as from 6 to 21% by volume, in particular 7 to 18% by volume; a benzene content of up to 5% by volume, such as 0.5 to 1.0% by volume, in particular 0.6 to 0.9% by volume, and/or an oxygen content of up to 25% by volume , such as up to 10% by weight or 1.0 to 2.7% by weight, in particular from 1.2 to 2.0% by weight.
Insbesondere können solche Ottokraftstoffe beispielhaft genannt werden, welche gleichzeitig einen Aromatengehalt von maximal 38 oder 35 Vol.-%, einen Olefingehalt von maximal 21 Vol.- %, einen Schwefelgehalt von maximal 50 oder 10 Gew.-ppm, eine Benzolgehalt von maximal 1,0 Vol.-% und eine Sauerstoffgehalt von 1,0 bis 2,7 Gew.-% aufweisen.In particular, petrol fuels that can be mentioned by way of example have an aromatics content of no more than 38 or 35% by volume, an olefin content of no more than 21% by volume, a sulfur content of no more than 50 or 10 ppm by weight, a benzene content of no more than 1, 0% by volume and an oxygen content of 1.0 to 2.7% by weight.
Der Gehalt an Alkoholen und Ethern im Ottokraftstoff kann über einem weiten Bereich variieren. Beispiele typischer maximaler Gehalte sind für Methanol 15 Vol.-%, für Ethanol 85 Vol.-%, für Isopropanol 20 Vol.-%, für tert.-Butanol 15 Vol.-%, für Isobutanol 20 Vol.-% und für Ether mit 5 oder mehr C-Atomen im Molekül 30 Vol.-%.The content of alcohols and ethers in petrol can vary over a wide range. Examples of typical maximum contents are 15% by volume for methanol, 85% by volume for ethanol, 20% by volume for isopropanol, 15% by volume for tert-butanol, 20% by volume for isobutanol and ether with 5 or more carbon atoms in the molecule, 30% by volume.
Der Sommer-Dampfdruck des Ottokraftstoffes beträgt üblicherweise maximal 70 kPa, insbesondere 60 kPa (jeweils bei 37°C).The summer vapor pressure of petrol is usually a maximum of 70 kPa, in particular 60 kPa (each at 37° C.).
Die ROZ des Ottokraftstoffes beträgt in der Regel 75 bis 105. Ein üblicher Bereich für die entsprechende MOZ liegt bei 65 bis 95.The RON of petrol is usually 75 to 105. A typical range for the corresponding MON is 65 to 95.
Die genannten Spezifikationen werden nach üblichen Methoden bestimmt (DIN EN 228).The specifications mentioned are determined using standard methods (DIN EN 228).
Eine Ausführungsform der vorliegenden Erfindung stellen Kraftstoffzusammensetzungen dar, enthaltend,
- (A) mindestens eine Verbindung der Formel (III),
- (B1) mindestens eine Verbindung (B1a), bei der es sich um ein Polyisobutenamin handelt, erhältlich durch Hydroformylierung und anschließender reduktiver Aminierung, ganz besonders bevorzugt mit Ammoniak, von Polyisobuten mit Mn = 300 bis 5000,
- (B2) mindestens ein Trägeröl,
- (B3) mindestens einen Korrosionsinhibitor,
- (C) mindestens einen Ottokraftstoff, sowie
optional mindestens einen Alkohol, bevorzugt mindestens ein C1-C4-Alkanol, besonders bevorzugt Methanol oder Ethanol, ganz besonders bevorzugt Ethanol.
- (A) at least one compound of formula (III),
- (B1) at least one compound (B1a), which is a polyisobuteneamine, obtainable by hydroformylation and subsequent reductive amination, very particularly preferably with ammonia, of polyisobutene with Mn=300 to 5000,
- (B2) at least one carrier oil,
- (B3) at least one corrosion inhibitor,
- (C) at least one petrol, and
optionally at least one alcohol, preferably at least one C 1 -C 4 alkanol, particularly preferably methanol or ethanol, very particularly preferably ethanol.
Die Dosierung erfolgt so, daß die erfindungsgemäße Verbindung (A) im Kraftstoff in Mengen von 20 - 2000 Gew.ppm, besonders bevorzugt in Mengen von 30 - 1000 Gew.ppm, ganz besonders bevorzugt in Mengen von 40 - 500 Gew.ppm, insbesondere in Mengen von 50 - 300 Gew.ppm und speziell in Mengen von 10 - 100 Gew.ppm anwesend ist.The dosage is such that the compound (A) according to the invention is present in the fuel in amounts of 20-2000 ppm by weight, particularly preferably in amounts of 30-1000 ppm by weight, very particularly preferably in amounts of 40-500 ppm by weight, in particular is present in amounts of 50-300 wppm and especially in amounts of 10-100 wppm.
Die Gehalte der anderen Co-Additive im Kraftstoff ergeben sich aus den oben für die Kraftstoffadditivkonzentrate angegebenen Mengen im Verhältnis zur Komponente (A).The contents of the other co-additives in the fuel result from the amounts specified above for the fuel additive concentrates in relation to component (A).
Die in diesem Dokument angegebenen Prozent- und ppm-Mengen beziehen sich, soweit nicht anders angegeben, auf Gew.% und Gew.ppm.The percentages and ppm amounts given in this document refer to wt.% and wt.ppm unless otherwise stated.
Die Erfindung wird durch die nachfolgend beschriebenen Beispiele näher erläutert, ohne darauf beschränkt zu sein.The invention is explained in more detail by the examples described below, without being restricted thereto.
Abkürzungen, Analytik und eingesetzte Chemikalien
- OHZ: Hydroxylzahl, bestimmt nach DIN 53240-1
- Gesamtaminzahl: Bestimmt nach DIN EN 13716:2001
- Mn: Zahlenmittleres Molekulargewicht nach DIN 55672-1
- Mw: Massenmitleres Molekulargewicht nach DIN 55672-1
- D: Polydispersität nach DIN 55672-1
- Kaliumgehalte: Bestimmt durch ICP-OES
- DMAPA: 3-(Dimethylamino)propylamin (
CAS 109-55-7 - PO: Propylenoxid (
CAS 75-56-9 - BuO: 1,2-Butylenoxid (
CAS 106-88-7 - Quadrol L® der Firma BASF SE: Ethylendiamin×4PO
- N,N,N',N'-Tetrakis(2-Hydroxypropyl)ethylendiamin,
CAS 102-60-3 - DETA: Diethylentriamin der Fa. BASF SE,
CAS 111-40-0 - Isotridecanol N der BASF SE,
CAS 27458-92-0 - Ambosol®: Hydratisiertes Magnesiumsilikat der Fa. PQ Corporation.
- Viskositäten und Dichten wurden mit einem Stabinger-Viskosimeter nach ASTM D7042 bestimmt.
- OHZ: Hydroxyl number, determined according to DIN 53240-1
- Total number of amines: Determined according to DIN EN 13716:2001
- M n : number-average molecular weight according to DIN 55672-1
- M w : mass-average molecular weight according to DIN 55672-1
- D: Polydispersity according to DIN 55672-1
- Potassium levels: Determined by ICP-OES
- DMAPA: 3-(dimethylamino)propylamine (
CAS 109-55-7 - PO: propylene oxide (
CAS 75-56-9 - BuO: 1,2-butylene oxide (
CAS 106-88-7 - Quadrol L® from BASF SE: ethylenediamine×4PO
- N,N,N',N'-tetrakis(2-hydroxypropyl)ethylenediamine,
CAS 102-60-3 - DETA: Diethylenetriamine from BASF SE,
CAS 111-40-0 - Isotridecanol N from BASF SE,
CAS 27458-92-0 - Ambosol ® : Hydrated magnesium silicate from PQ Corporation.
- Viscosities and densities were determined using a Stabinger viscometer according to ASTM D7042.
In einem 3,5 l Druckautoklaven mit Schrägblattrührer wurden DMAPA (408,8 g; 4,0 mol) und Wasser (4,1 g, vollentsalzt) vorgelegt und der Rührer wurde eingeschaltet. Der Reaktor wurde mit Stickstoff inertisiert, anschließend wurde auf 130°C erhitzt und mit Stickstoff ein Druck von 2,0 bar absolut eingestellt. Propylenoxid (465 g; 8,0 mol) wurde über einen Zeitraum von 6 h zudosiert. Man ließ 6 h bei 130°C nachreagieren, kühlte auf 50°C ab, spülte den Reaktor mit Stickstoff und ließ das Produkt ab. Anschließend wurde das Produkt am Rotationsverdampfer (90°C/10 mbar/2 h) von Niedrigsiedern befreit.DMAPA (408.8 g; 4.0 mol) and water (4.1 g, completely desalinated) were placed in a 3.5 l pressure autoclave with a slanting blade stirrer and the stirrer was switched on. The reactor was rendered inert with nitrogen, then it was heated to 130° C. and a pressure of 2.0 bar absolute was established with nitrogen. Propylene oxide (465 g; 8.0 mol) was metered in over a period of 6 hours. The reaction was allowed to continue for 6 h at 130° C., the mixture was cooled to 50° C., the reactor was flushed with nitrogen and the product was discharged. The product was then freed from low boilers on a rotary evaporator (90° C./10 mbar/2 h).
1H-NMR-Analytik in CDCl3 bestätigte die Struktur. 1 H NMR analysis in CDCl 3 confirmed the structure.
In einem 3,5 l Druckautoklaven mit Schrägblattrührer wurden das Produkt aus Synthesebeispiel 1 (339 g; 1,55 mol) und 50%ige wässrige KOH-Lösung (14,5 g) vorgelegt und der Rührer wurde eingeschaltet. Anschließend wurde auf einen Druck von 10 mbar evakuiert und für 2 h auf 105°C erhitzt, um das in der Startermischung enthaltene Wasser abzudestillieren. Der Reaktor wurde mit Stickstoff inertisiert, anschließend wurde auf 130°C erhitzt und mit Stickstoff ein Druck von 2,0 bar absolut eingestellt. Propylenoxid (2075 g, 35,7 mol) wurde über einen Zeitraum von 7 h zudosiert. Man ließ 10 h bei 130°C nachreagieren, kühlte auf 80°C ab, spülte den Reaktor mit Stickstoff und ließ das Produkt ab. Das Produkt wurde am Rotationsverdampfer (90°C/10 mbar/2 h) von Niedrigsiedern befreit. Anschließend wurde das Produkt mit 73 g Ambosol® versetzt, 2 h bei 80°C gerührt und mit Hilfe einer Druckfilternutsche filtriert (Filtermedium Seitz K 150 Tiefenfilter). Man erhielt 2412 g des Produktes (99,9% der Theorie) in Form eines gelben Öls.The product from synthesis example 1 (339 g; 1.55 mol) and 50% strength aqueous KOH solution (14.5 g) were initially taken in a 3.5 l pressure autoclave with an inclined blade stirrer, and the stirrer was switched on. The system was then evacuated to a pressure of 10 mbar and heated at 105° C. for 2 h in order to distill off the water contained in the starter mixture. The reactor was rendered inert with nitrogen, then it was heated to 130° C. and a pressure of 2.0 bar absolute was established with nitrogen. Propylene oxide (2075 g, 35.7 mol) was metered in over a period of 7 hours. The reaction was allowed to continue for 10 h at 130° C., the mixture was cooled to 80° C., the reactor was flushed with nitrogen and the product was discharged. The product was freed from low boilers on a rotary evaporator (90° C./10 mbar/2 h). Then the product with 73 g Ambosol ® mixed, stirred for 2 h at 80° C. and filtered using a pressure suction filter (Filter medium Seitz K 150 depth filter). 2412 g of the product (99.9% of theory) were obtained in the form of a yellow oil.
OHZ 76,3 mg KOH/g, Gesamtaminzahl 67,9 mg KOH/g, DMAPA-Gehalt nach Flüssigchromatographie < 0,005%, Kaliumgehalt < 10 ppm, Mn 1658 g/mol, Mw 1891 g/mol, D 1,14, kin. Viskosität bei 40°C 113,4 mm2/s.OOH 76.3 mg KOH/g, total amine number 67.9 mg KOH/g, DMAPA content according to liquid chromatography <0.005%, potassium content <10 ppm, Mn 1658 g/mol, Mw 1891 g/mol, D 1.14 , children Viscosity at 40°C 113.4 mm 2 /s.
In analoger Weise zu Synthesebeispiel 2 wurde DMAPA×15 PO erhalten.In a manner analogous to Synthesis Example 2, DMAPA×15 PO was obtained.
OHZ 99,9 mg KOH/g, Mn 1026, Mw 1185 g/mol, D 1,15, Kaliumgehalt < 10 ppm.OHN 99.9 mg KOH/g, M n 1026, M w 1185 g/mol, D 1.15, potassium content < 10 ppm.
In analoger Weise zu Synthesebeispiel 2 wurde Ethylendiamin×25 PO durch Umsetzung von Quadrol L (Ethylendiamin×4PO) mit 21 Äquivalenten PO erhalten.In a manner analogous to Synthesis Example 2, ethylenediamine×25 PO was obtained by reacting Quadrol L (ethylenediamine×4PO) with 21 equivalents of PO.
OHZ 121,3 mg KOH/g, Gesamtaminzahl 72,2 mg KOH/g, Mn 1804 g/mol, Mw 1902 g/mol, D 1,05, Kaliumgehalt < 10 ppm, dyn. Viskosität bei 40°C 198,4 mPas.OHN 121.3 mg KOH/g, total amine number 72.2 mg KOH/g, M n 1804 g/mol, M w 1902 g/mol, D 1.05, potassium content < 10 ppm, dyn. Viscosity at 40°C 198.4 mPas.
In analoger Weise zu Synthesebeispiel 1 wurde DETA×5 PO durch Propoxylierung von DETA erhalten.In a manner analogous to Synthesis Example 1, DETA×5 PO was obtained by propoxylation of DETA.
In analoger Weise zu Synthesebeispiel 2 wurde DETAx25 PO durch Propoxylierung von DE-TA×5 PO (Synthesebeispiel 5) erhalten.In a manner analogous to Synthesis Example 2, DETAx25 PO was obtained by propoxylation of DE-TA×5 PO (Synthesis Example 5).
In einem 3,5 l Druckautoklaven mit Schrägblattrührer wurde DMAPA (408,8 g; 4,0 mol) vorgelegt und der Rührer wurde eingeschaltet. Der Reaktor wurde mit Stickstoff inertisiert, anschließend wurde auf 120°C erhitzt und mit Stickstoff ein Druck von 2,0 bar absolut eingestellt. 1,2-Butylenoxid (577 g; 8,0 mol) wurde über einen Zeitraum von 15 h zudosiert. Man ließ 6 h bei 120°C nachreagieren, kühlte auf 50°C ab, spülte den Reaktor mit Stickstoff und ließ das Produkt ab. Anschließend wurde das Produkt am Rotationsverdampfer (90°C/10 mbar/2 h) von Niedrigsiedern befreit.DMAPA (408.8 g; 4.0 mol) was placed in a 3.5 l pressure autoclave with an inclined blade stirrer and the stirrer was switched on. The reactor was rendered inert with nitrogen, then it was heated to 120° C. and a pressure of 2.0 bar absolute was established with nitrogen. 1,2-Butylene oxide (577 g; 8.0 mol) was metered in over a period of 15 hours. The reaction was allowed to continue for 6 h at 120° C., the mixture was cooled to 50° C., the reactor was flushed with nitrogen and the product was discharged. The product was then freed from low boilers on a rotary evaporator (90° C./10 mbar/2 h).
1H-NMR-Analytik in CDCl3 bestätigte die Struktur. 1 H NMR analysis in CDCl 3 confirmed the structure.
In einem 2 l Druckautoklaven mit Schrägblattrührer wurden das Produkt aus Synthesebeispiel 7 (130 g; 0,528 mol) und 50%ige wässrige KOH-Lösung (6,0 g) vorgelegt und der Rührer wurde eingeschaltet. Anschließend wurde auf einen Druck von 10 mbar evakuiert und für 2 h auf 95°C erhitzt, um das in der Startermischung enthaltene Wasser abzudestillieren. Der Reaktor wurde mit Stickstoff inertisiert, anschließend wurde auf 140°C erhitzt und mit Stickstoff ein Druck von 2,5 bar absolut eingestellt. 1,2-Butylenoxid (875 g, 12,1 mol) wurde über einen Zeitraum von 14 h zudosiert. Man ließ 4 h bei 140°C nachreagieren, kühlte auf 80°C ab, spülte den Reaktor mit Stickstoff und ließ das Produkt ab. Das Produkt wurde am Rotationsverdampfer (90°C/10 mbar/2 h) von Niedrigsiedern befreit. Anschließend wurde das Produkt mit 30 g Ambosol® versetzt, 2 h bei 80°C/100 mbar gerührt und mit Hilfe einer Druckfilternutsche filtriert (Filtermedium Seitz K 900 Tiefenfilter). Man erhielt das Produkt in Form eines gelben Öls.The product from Synthesis Example 7 (130 g; 0.528 mol) and 50% strength aqueous KOH solution (6.0 g) were placed in a 2 l pressure autoclave with an inclined blade stirrer, and the stirrer was switched on. The system was then evacuated to a pressure of 10 mbar and heated at 95° C. for 2 h in order to distill off the water present in the starter mixture. The reactor was rendered inert with nitrogen, then it was heated to 140° C. and a pressure of 2.5 bar absolute was established with nitrogen. 1,2-Butylene oxide (875 g, 12.1 mol) was metered in over a period of 14 hours. The reaction was allowed to continue for 4 h at 140° C., the mixture was cooled to 80° C. and the reactor was flushed as well nitrogen and drained the product. The product was freed from low boilers on a rotary evaporator (90° C./10 mbar/2 h). The product was then mixed with 30 g of Ambosol® , stirred at 80° C./100 mbar for 2 h and filtered using a pressure suction filter (Filter medium Seitz K 900 depth filter). The product was obtained in the form of a yellow oil.
OHZ 63,7 mg KOH/g, Gesamtaminzahl 49,7 mg KOH/g, Mn 2374 g/mol, Mw 2550 g/mol, D 1,07. OHN 63.7 mg KOH/g, total amine number 49.7 mg KOH/g, Mn 2374 g/mol, Mw 2550 g/mol, D 1.07.
AnwendungsbeispieleApplication examples
Es wurde folgende Additivpaketformulierungen hergestellt:
Die Formulierungen waren einphasig und zeigten bei Lagerung über 6 Wochen bei -10 °C, sowie bei Lagerung über -5 °C über 3 Monate keine Phasentrennungen oder Ausfällungen.
** PO/BuO basiertes Trägeröl
*** Friction Modifier, Lösungsmittel und Korrosionsinhibitor
****PO basiertes Trägeröl
** PO/BuO based carrier oil
*** Friction modifier, solvent and corrosion inhibitor
****PO based carrier oil
Einsatz als Trägeröl:
Ventilklebetest im Motor VW Wasserboxer (CEC F-016-96) bei -18°C:
Die Überprüfung des Verhaltens beim Ventilkleben wurde durch Untersuchungen im VW-Wasserboxer-Test nach CEC F-16-T-96 vorgenommen. Als Grundkraftstoff diente ein Eurosuper-Kraftstoff gemäß EN 228. Geprüft wurde nach den Kriterien der Testvorschrift, ob ein "Pass" (kein Ventilkleben in drei aufeinander folgenden Testläufen) oder ein "Fail" (Ventilkleben im ersten, zweiten oder dritten der aufeinander folgenden Testläufe) erfolgte. Das Ventilkleben macht sich hierbei dadurch bemerkbar, dass sich der Motor nur mit Verzögerung oder gar nicht mehr starten lässt. Um eine Differenzierung zu ermöglichen, wurde absichtlich im Grenzbereich des zu erwartenden Ventilklebens getestet. Die angegebenen Dosiermengen der jeweiligen Additive in Gew.-ppm (angegeben als Reinsubstanz-Gehalt, ohne Lösungsmittel) beziehen sich jeweils auf die Gesamtmenge der eingesetzten Ottokraftstoff-Formulierung.
- Kraftstoff: MIRO 95 OKTAN E10
- Formulierung 4 mit 1000 mg/kg: 3 × pass
- Formulierungen 5-7 mit 2325 mg/kg: 3 × pass
Valve sticking test in the VW Wasserboxer engine (CEC F-016-96) at -18°C:
The behavior during valve sticking was checked by investigations in the VW Wasserboxer test according to CEC F-16-T-96. A Eurosuper fuel in accordance with EN 228 was used as the base fuel. The criteria of the test specification were used to determine whether it was a "pass" (no valve sticking in three consecutive test runs) or a "fail" (valve sticking in the first, second or third of the consecutive test runs). ) took place. The valve sticking is noticeable here by the fact that the engine can only be started with a delay or not at all. In order to enable differentiation, tests were deliberately carried out in the limit area of the valve sticking to be expected. The specified dosages of the respective additives in ppm by weight (given as pure substance content, without solvent) relate to the total quantity of the petrol formulation used.
- Fuel: MIRO 95 OCTANE E10
- Formulation 4 at 1000 mg/kg: 3 x pass
- Formulations 5-7 at 2325 mg/kg: 3 x pass
Der Motorentest wurde durchgeführt, wie beschrieben in
Dazu wurde nach einer internen Methode ein kommerziell verfügbarer, aufgeladener Vierzylinder Ottomotor (1,6 L Hubraum) mit Direkteinspritzung mit einem E0 Benzin, enthaltend 7 Vol% sauerstoffhaltige Verbindungen.For this purpose, a commercially available, supercharged four-cylinder Otto engine (1.6 L cubic capacity) with direct injection and E0 gasoline containing 7% by volume of oxygen-containing compounds was tested using an internal method.
Der Kraftstoff wurde mit 80 Gew.ppm der angegebenen Produkte aus den Synthesebeispielen oder Formulierungen versetzt und über die Laufzeit der FR-Wert bestimmt. Der FR-Wert ist ein Parameter, der von der Motorsteuerung erstellt wird und entspricht der Einspritzzeit des Kraftstoffs in den Verbrennungsraum. Wenn sich der FR-Wert im Verlauf des Testlaufs erhöht, deutet dies auf Ablagerungen an der Einspritzdüse hin, je größer die Erhöhung ist, desto mehr Ablagerungen haben sich gebildet. Bleibt der FR-Wert hingegen konstant oder sinkt sogar im Verlauf eines Restlaufs, bleibt die Einspritzdüse frei von Ablagerungen.The fuel was admixed with 80 ppm by weight of the specified products from the synthesis examples or formulations and the FR value was determined over the running time. The FR value is a parameter created by the engine control and corresponds to the fuel injection time into the combustion chamber. If the FR value increases during the test run, this indicates deposits on the injector, the larger the increase, the more deposits have formed. On the other hand, if the FR value remains constant or even falls over the course of the remaining run, the injection nozzle remains free of deposits.
Gleichzeitig werden die Brennraumablagerungen bestimmt (Ablagerungen an der Kolbenoberseite: PTD, Ablagerungen im Zylinderkopf: CHD).
- Testlauf 1: Grundwert ohne Additiv
Startend mit 0 sinkt der FR-Wert zunächst ab um nach ca. 30 Stunden wieder 0 zu erreichen und erreicht nach 50 Stunden Laufzeit einen Endwert von +2,60. - Testlauf 2: Zusatz von 80 Gew.ppm der Verbindung aus Synthesebeispiel 2 in den Kraftstoff
Startend mit 0 sinkt der FR-Wert dauerhaft ab und erreicht nach 50 Stunden Laufzeit einen Endwert von -2,00. - Testlauf 3: Zusatz von 80 Gew.ppm der Verbindung aus Synthesebeispiel 3 in den Kraftstoff
Startend mit 0 sinkt der FR-Wert dauerhaft ab und erreicht - zuletzt wieder leicht ansteigend - nach 50 Stunden Laufzeit einen Endwert von -1,58. - Testlauf 4: Vergleich zu
EP 700985
Startend mit 0 mit einem unadditivierten Kraftstoff erreicht der FR-Wert nach 80 h einen Wert von 7,0 (dirty-up). Danach werden dem Kraftstoff 65 ppm PIBA und 55 ppm Polyetheramin gemäß Herstellungsbeispiel B derEP700985 - Testlauf 5: Zusatz von Synthesebeispiel 2
Startend mit 0 mit einem unadditivierten Kraftstoff steigt der FR-Wert und erreicht nach 60 h einen Wert von 6,8 (dirty-up). Danach werden dem Kraftstoff 65 ppm PIBA, 55 ppm Trägeröl und 55 ppm Synthesebeispiel 2 zugesetzt. Nach weiteren 20 h Laufzeit wird ein FR-Wert von 2,0 erreicht (clean-up). Der relative clean-up beträgt damit 71%.
- Test run 1: basic value without additive
Starting with 0, the FR value initially drops, only to reach 0 again after about 30 hours and reaches a final value of +2.60 after 50 hours of running time. - Test Run 2: Addition of 80 ppm by weight of the compound from Synthesis Example 2 to the fuel
Starting with 0, the FR value drops permanently and reaches a final value of -2.00 after 50 hours of running time. - Test Run 3: Addition of 80 ppm by weight of the compound from Synthesis Example 3 to the fuel
Starting with 0, the FR value drops permanently and finally reaches a final value of -1.58 after 50 hours of running time - finally rising again slightly. - Test run 4: comparison to
EP 700985
Starting with 0 with an additive-free fuel, the FR value reaches a value of 7.0 (dirty-up) after 80 hours. Thereafter, 65 ppm of PIBA and 55 ppm of polyetheramine according to Preparation Example B are added to the fuelEP700985 - Test Run 5: Addition of Synthesis Example 2
Starting with 0 with an unadditized fuel, the FR value increases and reaches a value of 6.8 (dirty-up) after 60 hours. Thereafter, 65 ppm PIBA, 55 ppm carrier oil and 55 ppm Synthesis Example 2 are added to the fuel. After a further 20 h running time, an FR value of 2.0 is reached (clean-up). The relative clean-up is thus 71%.
Reduktion von Brennraumablagerungen in einem M111 (PFI) Motor:
Kraftstoff MIRO E10, 95 Oktan; Test nach CEC F-020-98
Fuel MIRO E10, 95 octane; Test according to CEC F-020-98
Reduktion von Brennraumablagerungen in einem M111 (PFI) Motor:
Kraftstoff MIRO E10, 95 Oktan; Test nach CEC F-020-98
Fuel MIRO E10, 95 octane; Test according to CEC F-020-98
Claims (15)
- The use of compounds (A) selected from the group consisting of
- The use according to claim 1, wherein the R radical is selected from the group consisting of 1,2-ethylene, 1,2-propylene, 1,3-propylene, 1,2-butylene, 1,3-butylene, 1,4-butylene, 1,5-pentylene, 1,6-hexylene, 1,8-octylene and 1,10-decylene.
- The use according to any of the preceding claims, wherein R1, R2, R3, R4, R5 and R6, if they do not represent a -[-Xi-]n-H radical, are independently selected from the group consisting of hydrogen and an alkyl radical having 1 to 12 carbon atoms.
- The use according to claim 3, wherein R1, R2, R3, R4, R5 and R6, if they do not represent a -[-Xi-]n-H radical, are independently selected from the group consisting of methyl, ethyl, isopropyl, n-propyl, n-butyl, sec-butyl and tert-butyl.
- The use according to any of the preceding claims, wherein the compound is a compound of the formula (I) .
- The use according to claim 5, wherein x = 1 or 2 and R is 1,2-ethylene, 1,2-propylene or 1,3-propylene.
- The use according to claim 5 or 6, wherein R1 and R2 are each independently C1- to C4-alkyl and R3 and R4 are each independently a -[-Xi-]n-H radical.
- The use according to claim 7, wherein each Xi for i = 1 to n is independently selected from the group consisting of -CH2-CH(CH3)-O-, -CH(CH3)-CH2-O-, -CH2-C(CH3)2-O-, -C(CH3)2-CH2-O-, -CH2-CH(C2H5)-O- and - CH(C2H5)-CH2-O-.
- The use according to claim 9, wherein the sum total of p and q is from 2 to 50.
- The use according to claim 1, wherein the radicals in formula (I) are defined as follows:(Ia) R is 1,2-ethylene, w = 1, R1 to R3 are methyl and R4 is a -[-Xi-]n-H chain(Ib) R is 1,2-ethylene, w = 1, R1 and R2 are methyl and R3 and R4 are each independently a -[-Xi-]n-H chain(Ic) R is 1,2-propylene, w = 1, R1 and R2 are methyl and R3 and R4 are each independently a -[-Xi-]n-H chain(Id) R is 1,3-propylene, w = 1, R1 and R2 are methyl and R3 and R4 are each independently a -[-Xi-]n-H chain(Ie) R is 1,3-propylene, w = 1, R1 and R2 are ethyl and R3 and R4 are each independently a -[-Xi-]n-H chain,(If) R is 1, 3-propylene, w = 1, R1 and R2 are n-butyl and R3 and R4 are each independently a -[-Xi-]n-H chain, and(Ig) R is 1,2-ethylene, w = 1, R1 to R4 are a -[-Xi-]n-H chain.
- The use according to claim 1, wherein the radicals in formula (II) are defined as follows:(IIa) R is 1,2-ethylene, x and y are 1, z = 0 and R1 to R4 and R6 are each independently a -[-Xi-]n-H chain(IIb) R is 1,2-propylene, x and y are 1, z = 0 and R1 to R4 and R6 are each independently a -[-Xi-]n-H chain, and(IIc) R is 1,3-propylene, x and y are 1, z = 0 and R1 to R4 are each independently a -[-Xi-]n-H chain, and R6 is C1- to C20-alkyl.
- The use according to claim 9, wherein the radicals in formula (III) are defined as follows:(IIIa) R is 1,2-ethylene and R1 and R2 are methyl(IIIb) R is 1,2-ethylene and R1 and R2 are ethyl(IIIc) R is 1,2-ethylene and R1 and R2 are n-butyl(IIId) R is 1,2-ethylene and R1 and R2 are collectively a 1,4-butylene chain(IIIe) R is 1,2-ethylene and R1 and R2 are collectively a 1,5-pentylene chain(IIIf) R is 1,2-ethylene and R1 and R2 are collectively a 3-oxa-1,5-pentylene chain(IIIg) R is 1,2-propylene and R1 and R2 are methyl(IIIh) R is 1,2-propylene and R1 and R2 are ethyl(IIIi) R is 1,2-propylene and R1 and R2 are n-butyl(IIIj) R is 1,2-propylene and R1 and R2 are collectively a 1,4-butylene chain(IIIk) R is 1,2-propylene and R1 and R2 are collectively a 1,5-pentylene chain(IIIl) R is 1,2-propylene and R1 and R2 are collectively a 3-oxa-1,5-pentylene chain(IIIm) R is 1,3-propylene and R1 and R2 are methyl(IIIn) R is 1,3-propylene and R1 and R2 are ethyl(IIIo) R is 1,3-propylene and R1 and R2 are n-butyl(IIIp) R is 1,3-propylene and R1 and R2 are collectively a 1,4-butylene chain(IIIq) R is 1,3-propylene and R1 and R2 are collectively a 1,5-pentylene chain, and(IIIr) R is 1,3-propylene and R1 and R2 are collectively a 3-oxa-1,5-pentylene chain.
- A fuel additive concentrate comprising(A) 1-50% by weight of at least one compound of the formula (I), (II) or (III) as described in any of claims 1 to 13,in which each Xi for i = 1 to n is independently selected from the group consisting of -CH2-CH(CH3)-O-, -CH(CH3)-CH2-O-, -CH2-C(CH3)2-O-, -C(CH3)2-CH2-O-, -CH2-CH(C2H5)-O- and -CH(C2H5)-CH2-O-, andR1, R2 and Xi have the definitions given above andp and q are independently a positive integer,(B1) 20-96.75% by weight of at least one compound (B1a) which is a polyisobuteneamine obtainable by hydroformylation and subsequent reductive amination, most preferably with ammonia, of polyisobutene with Mn = 300 to 5000,(B2) 2-30% by weight of at least one carrier oil selected from the group consisting of polyolefins, (poly)esters, (poly)alkoxylates, polyethers, aliphatic polyetheramines, alkylphenol-started polyethers, alkylphenol-started polyetheramines and carboxylic esters of alkanols having 6 to 24 carbon atoms, and(B3) 0.25-10% by weight of at least one corrosion inhibitor selected from the group consisting of mono-, di- and polycarboxylic acids having at least 12 carbon atoms, ammonium salts of organic carboxylic acids and heterocyclic aromatics, with the proviso that the sum total of all components is always 100% by weight.
- A fuel composition comprising(A) 20 to 2000 ppm by weight of at least one compound of the formula (I), (II) or (III) as described in any of claims 1 to 13,in which each Xi for i = 1 to n is independently selected from the group consisting of -CH2-CH(CH3)-O-, -CH(CH3)-CH2-O-, -CH2-C(CH3)2-O-, -C(CH3)2-CH2-O-, -CH2-CH(C2H5)-O- and -CH(C2H5)-CH2-O-, andR1, R2 and Xi have the definitions given above andp and q are independently a positive integer,(B1) 50 to 1000 ppm by weight of at least one compound (B1a) which is a polyisobuteneamine obtainable by hydroformylation and subsequent reductive amination, most preferably with ammonia, of polyisobutene with Mn = 300 to 5000,(B2) at least one carrier oil selected from the group consisting of polyolefins, (poly)esters, (poly)alkoxylates, polyethers, aliphatic polyetheramines, alkylphenol-started polyethers, alkylphenol-started polyetheramines and carboxylic esters of alkanols having 6 to 24 carbon atoms, (B3) at least one corrosion inhibitor selected from the group consisting of mono-, di- and polycarboxylic acids having at least 12 carbon atoms, ammonium salts of organic carboxylic acids and heterocyclic aromatics,(C) at least one gasoline fuel, and
optionally at least one alcohol, preferably at least one C1-C4-alkanol, more preferably methanol or ethanol, most preferably ethanol.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP17165955 | 2017-04-11 | ||
| PCT/EP2018/058398 WO2018188982A1 (en) | 2017-04-11 | 2018-04-03 | Alkoxylated amines as fuel additives |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP3609986A1 EP3609986A1 (en) | 2020-02-19 |
| EP3609986B1 true EP3609986B1 (en) | 2022-10-12 |
Family
ID=58638671
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP18720112.4A Active EP3609986B1 (en) | 2017-04-11 | 2018-04-03 | Alkoxylated amines as fuel additive |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US11130923B2 (en) |
| EP (1) | EP3609986B1 (en) |
| CN (1) | CN110494534A (en) |
| AU (1) | AU2018253240A1 (en) |
| RU (1) | RU2019135830A (en) |
| SG (1) | SG11201909437RA (en) |
| WO (1) | WO2018188982A1 (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA3265314A1 (en) | 2022-09-09 | 2024-03-14 | Ecolab Usa Inc. | Synthetic lubricity additives for hydrocarbon fuels |
| WO2024105387A1 (en) * | 2022-11-15 | 2024-05-23 | Innospec Fuel Specialties Llc | Compositions, methods and uses |
Family Cites Families (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4129508A (en) * | 1977-10-13 | 1978-12-12 | The Lubrizol Corporation | Demulsifier additive compositions for lubricants and fuels and concentrates containing the same |
| US4409000A (en) * | 1981-12-14 | 1983-10-11 | The Lubrizol Corporation | Combinations of hydroxy amines and carboxylic dispersants as fuel additives |
| US4690687A (en) | 1985-08-16 | 1987-09-01 | The Lubrizol Corporation | Fuel products comprising a lead scavenger |
| ES2032318T3 (en) | 1987-09-15 | 1993-02-01 | Basf Aktiengesellschaft | FUELS FOR OTTO ENGINES. |
| DE3732908A1 (en) | 1987-09-30 | 1989-04-13 | Basf Ag | FUELS CONTAINING POLYETHERAMINE FOR OTTO ENGINES |
| US4877416A (en) | 1987-11-18 | 1989-10-31 | Chevron Research Company | Synergistic fuel compositions |
| US4849572A (en) | 1987-12-22 | 1989-07-18 | Exxon Chemical Patents Inc. | Process for preparing polybutenes having enhanced reactivity using boron trifluoride catalysts (PT-647) |
| DE3826608A1 (en) | 1988-08-05 | 1990-02-08 | Basf Ag | FUELS CONTAINING POLYETHERAMINE OR POLYETHERAMINE DERIVATIVES FOR OTTO ENGINES |
| DE3838918A1 (en) | 1988-11-17 | 1990-05-23 | Basf Ag | FUELS FOR COMBUSTION ENGINES |
| DE4030164A1 (en) | 1990-09-24 | 1992-03-26 | Basf Ag | FUELS FOR COMBUSTION ENGINES AND LUBRICANTS CONTAINING HIGHLY MOLECULAR AMINO ALCOHOLS |
| TW239158B (en) * | 1991-02-15 | 1995-01-21 | Lubrizol Corp | |
| DE4142241A1 (en) | 1991-12-20 | 1993-06-24 | Basf Ag | FUELS FOR OTTO ENGINES |
| DE4309074A1 (en) | 1993-03-20 | 1994-09-22 | Basf Ag | Mixtures suitable as fuel additives |
| DE4313088A1 (en) | 1993-04-22 | 1994-10-27 | Basf Ag | Poly-1-n-alkeneamines and fuel and lubricant compositions containing them |
| AT400149B (en) | 1993-08-17 | 1995-10-25 | Oemv Ag | ADDITIVE FOR UNLEADED FUEL AND THIS CONTAINING FUEL |
| DE4425835A1 (en) | 1994-07-21 | 1996-01-25 | Basf Ag | Use of reaction products made of polyolefins and nitrogen oxides or mixtures of nitrogen oxides and oxygen as additives for fuels |
| DE4425834A1 (en) | 1994-07-21 | 1996-01-25 | Basf Ag | Reaction products made of polyisobutenes and nitrogen oxides or mixtures of nitrogen oxides and oxygen and their use as fuel and lubricant additives |
| DE4432038A1 (en) | 1994-09-09 | 1996-03-14 | Basf Ag | Fuels containing polyetheramines for gasoline engines |
| DE19525938A1 (en) | 1995-07-17 | 1997-01-23 | Basf Ag | Process for the production of organic nitrogen compounds, special organic nitrogen compounds and mixtures of such compounds and their use as fuel and lubricant additives |
| DE19620262A1 (en) | 1996-05-20 | 1997-11-27 | Basf Ag | Process for the preparation of polyalkenamines |
| GB9618546D0 (en) | 1996-09-05 | 1996-10-16 | Bp Chemicals Additives | Dispersants/detergents for hydrocarbons fuels |
| JP3948796B2 (en) * | 1997-09-30 | 2007-07-25 | 新日本石油株式会社 | Unleaded gasoline for in-cylinder direct injection gasoline engines |
| DE10102913A1 (en) | 2001-01-23 | 2002-07-25 | Basf Ag | Alkoxylated alkylphenols used as additives for fuel or lubricant compositions, have a long-chain alkyl group with tertiary or quaternary carbon atoms |
| JP4330828B2 (en) * | 2001-09-19 | 2009-09-16 | 新日本石油株式会社 | Fuel oil additive and fuel oil composition |
| US7435272B2 (en) * | 2002-04-24 | 2008-10-14 | Afton Chemical Intangibles | Friction modifier alkoxyamine salts of carboxylic acids as additives for fuel compositions and methods of use thereof |
| US7402185B2 (en) * | 2002-04-24 | 2008-07-22 | Afton Chemical Intangibles, Llc | Additives for fuel compositions to reduce formation of combustion chamber deposits |
| DE10316871A1 (en) * | 2003-04-11 | 2004-10-21 | Basf Ag | Fuel composition |
| CA2681498A1 (en) | 2007-03-21 | 2008-09-25 | The Lubrizol Corporation | Fuel additives for use in alcohol-fuels |
| WO2011076949A1 (en) | 2009-12-24 | 2011-06-30 | Shell Internationale Research Maatschappij B.V. | Liquid fuel compositions |
| CA2801643A1 (en) * | 2010-06-28 | 2012-01-05 | Basf Se | Alkoxylates and the use thereof |
| KR20140103978A (en) * | 2011-11-23 | 2014-08-27 | 바스프 에스이 | Amine mixture |
| IN2014CN03842A (en) | 2011-11-25 | 2015-09-04 | Basf Se | |
| WO2014023853A2 (en) | 2012-11-06 | 2014-02-13 | Basf Se | Tertiary amines for reducing injector nozzle fouling and modifying friction in direct injection spark ignition engines |
| US20160152912A1 (en) * | 2013-07-26 | 2016-06-02 | Innospec Limited | Diesel fuel compositions and methods of use thereof |
| CN106459811B (en) | 2014-01-29 | 2020-02-18 | 巴斯夫欧洲公司 | Corrosion Inhibitors for Fuels and Lubricants |
-
2018
- 2018-04-03 EP EP18720112.4A patent/EP3609986B1/en active Active
- 2018-04-03 CN CN201880024371.XA patent/CN110494534A/en active Pending
- 2018-04-03 SG SG11201909437R patent/SG11201909437RA/en unknown
- 2018-04-03 WO PCT/EP2018/058398 patent/WO2018188982A1/en not_active Ceased
- 2018-04-03 US US16/500,632 patent/US11130923B2/en not_active Expired - Fee Related
- 2018-04-03 AU AU2018253240A patent/AU2018253240A1/en not_active Abandoned
- 2018-04-03 RU RU2019135830A patent/RU2019135830A/en not_active Application Discontinuation
Also Published As
| Publication number | Publication date |
|---|---|
| RU2019135830A (en) | 2021-05-11 |
| US11130923B2 (en) | 2021-09-28 |
| CN110494534A (en) | 2019-11-22 |
| EP3609986A1 (en) | 2020-02-19 |
| AU2018253240A1 (en) | 2019-10-17 |
| US20200102515A1 (en) | 2020-04-02 |
| WO2018188982A1 (en) | 2018-10-18 |
| SG11201909437RA (en) | 2019-11-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE69703385T2 (en) | ALCOXYACETIC ACID DERIVATIVES | |
| EP2114844B1 (en) | Branched decyl nitrates and the use thereof as combustion improvers and/or cetane number improvers in fuels | |
| EP2270119B1 (en) | Fuel composition | |
| EP1613694B1 (en) | Polyalkene amines with improved applicational properties | |
| EP1612257A2 (en) | Fuel composition | |
| EP0310875A1 (en) | Fuels containing a polyether amine for spark ignition engines | |
| US9951288B2 (en) | Use of an alkoxylated polytetrahydrofuran to reduce fuel consumption | |
| EP2240519B1 (en) | Special polyisobutene amines, and use thereof as detergents in fuels | |
| DE102022131890A1 (en) | Guanidine derivatives as fuel additives | |
| DE102006006252A1 (en) | Fuel additive composition | |
| EP1230330B1 (en) | Use of fatty acid salts of alkoxylated oligoamines as lubricity improvers for petroleum products | |
| DE102004038113A1 (en) | Nitrogen-containing heterocyclic compounds as Reibverschleißvermindernder addition to fuels | |
| EP2782985A1 (en) | Amine mixture | |
| EP3609986B1 (en) | Alkoxylated amines as fuel additive | |
| EP1866396A1 (en) | Use of detergent additives for reducing a particle amount in the exhaust gas of direct injection diesel engines | |
| DE102022132342A1 (en) | Guanidinium salts as fuel additives | |
| EP1537192A1 (en) | Additive mixture for fuel and lubricants | |
| EP1495096A1 (en) | Fuel additive mixtures for gasolines with synergistic ivd performance | |
| EP2646530B1 (en) | Use of the product of reaction of a hydrocarbyl substituted dicarboxylic acid and a nitrogen compound to reduce fuel consumption. | |
| DE102022131356A1 (en) | Formamidines as fuel additives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: UNKNOWN |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE INTERNATIONAL PUBLICATION HAS BEEN MADE |
|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20191111 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| DAV | Request for validation of the european patent (deleted) | ||
| DAX | Request for extension of the european patent (deleted) | ||
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| 17Q | First examination report despatched |
Effective date: 20200824 |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: C10L 10/04 20060101ALI20220321BHEP Ipc: C10L 1/2383 20060101ALI20220321BHEP Ipc: C10L 1/238 20060101ALI20220321BHEP Ipc: C10L 1/182 20060101ALI20220321BHEP Ipc: C10L 1/224 20060101ALI20220321BHEP Ipc: C10L 1/198 20060101ALI20220321BHEP Ipc: C10L 10/06 20060101ALI20220321BHEP Ipc: C10L 1/2387 20060101ALI20220321BHEP Ipc: C10L 1/222 20060101AFI20220321BHEP |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| INTG | Intention to grant announced |
Effective date: 20220504 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 502018010818 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 1524180 Country of ref document: AT Kind code of ref document: T Effective date: 20221115 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG9D |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20221012 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20230213 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20230112 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20230212 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20230113 |
|
| P01 | Opt-out of the competence of the unified patent court (upc) registered |
Effective date: 20230331 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 502018010818 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20230421 Year of fee payment: 6 Ref country code: DE Payment date: 20230427 Year of fee payment: 6 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |
|
| 26N | No opposition filed |
Effective date: 20230713 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20230424 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20230418 Year of fee payment: 6 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230403 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230430 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230430 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230403 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230403 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 1524180 Country of ref document: AT Kind code of ref document: T Effective date: 20230403 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230403 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230403 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 502018010818 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20240403 |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20240430 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20241105 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20240430 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20240403 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20240430 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20240403 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20240430 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20241105 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20240430 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20180403 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20180403 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20221012 |