EP1565239B1 - Flüssige zusammensetzungen mit 3,7-diazabicyclo[3,3,1] nonanen zur behandlung von anti-arrhythmischen ereignissen - Google Patents
Flüssige zusammensetzungen mit 3,7-diazabicyclo[3,3,1] nonanen zur behandlung von anti-arrhythmischen ereignissen Download PDFInfo
- Publication number
- EP1565239B1 EP1565239B1 EP03811379A EP03811379A EP1565239B1 EP 1565239 B1 EP1565239 B1 EP 1565239B1 EP 03811379 A EP03811379 A EP 03811379A EP 03811379 A EP03811379 A EP 03811379A EP 1565239 B1 EP1565239 B1 EP 1565239B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- diazabicyclo
- carbon atoms
- tedisamil
- minutes
- infusion
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 239000007788 liquid Substances 0.000 title claims abstract description 45
- 239000008194 pharmaceutical composition Substances 0.000 title claims description 36
- 239000003416 antiarrhythmic agent Substances 0.000 title abstract description 21
- 230000003288 anthiarrhythmic effect Effects 0.000 title abstract description 18
- PTPQJKANBKHDPM-UHFFFAOYSA-N 3,7-diazabicyclo[3.3.1]nonane Chemical class C1NCC2CNCC1C2 PTPQJKANBKHDPM-UHFFFAOYSA-N 0.000 title abstract description 17
- 206010003658 Atrial Fibrillation Diseases 0.000 claims abstract description 80
- 238000011282 treatment Methods 0.000 claims abstract description 67
- 238000006243 chemical reaction Methods 0.000 claims abstract description 66
- 230000033764 rhythmic process Effects 0.000 claims abstract description 65
- 150000003839 salts Chemical class 0.000 claims abstract description 45
- 239000002253 acid Substances 0.000 claims abstract description 41
- 239000012453 solvate Substances 0.000 claims abstract description 32
- BKIMMITUMNQMOS-UHFFFAOYSA-N nonane Chemical class CCCCCCCCC BKIMMITUMNQMOS-UHFFFAOYSA-N 0.000 claims abstract description 24
- 238000011321 prophylaxis Methods 0.000 claims abstract description 22
- 238000001802 infusion Methods 0.000 claims description 149
- 125000004432 carbon atom Chemical group C* 0.000 claims description 44
- -1 3,7-diazabicyclo[3,3,1]nonane compound Chemical class 0.000 claims description 35
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical class OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 claims description 17
- 125000000217 alkyl group Chemical group 0.000 claims description 17
- 230000002763 arrhythmic effect Effects 0.000 claims description 17
- 125000001316 cycloalkyl alkyl group Chemical group 0.000 claims description 14
- 229910052757 nitrogen Inorganic materials 0.000 claims description 14
- 125000002947 alkylene group Chemical group 0.000 claims description 11
- 229910052736 halogen Inorganic materials 0.000 claims description 10
- 150000002367 halogens Chemical group 0.000 claims description 10
- 229910052739 hydrogen Inorganic materials 0.000 claims description 9
- 239000001257 hydrogen Substances 0.000 claims description 9
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 9
- 238000004519 manufacturing process Methods 0.000 claims description 7
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims description 7
- 239000001530 fumaric acid Substances 0.000 claims description 6
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 6
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 4
- 125000003342 alkenyl group Chemical group 0.000 claims description 3
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 3
- 125000006410 propenylene group Chemical group 0.000 claims description 3
- 125000004186 cyclopropylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C1([H])[H] 0.000 claims description 2
- QCQCHGYLTSGIGX-GHXANHINSA-N 4-[[(3ar,5ar,5br,7ar,9s,11ar,11br,13as)-5a,5b,8,8,11a-pentamethyl-3a-[(5-methylpyridine-3-carbonyl)amino]-2-oxo-1-propan-2-yl-4,5,6,7,7a,9,10,11,11b,12,13,13a-dodecahydro-3h-cyclopenta[a]chrysen-9-yl]oxy]-2,2-dimethyl-4-oxobutanoic acid Chemical compound N([C@@]12CC[C@@]3(C)[C@]4(C)CC[C@H]5C(C)(C)[C@@H](OC(=O)CC(C)(C)C(O)=O)CC[C@]5(C)[C@H]4CC[C@@H]3C1=C(C(C2)=O)C(C)C)C(=O)C1=CN=CC(C)=C1 QCQCHGYLTSGIGX-GHXANHINSA-N 0.000 claims 1
- 229960002926 tedisamil Drugs 0.000 abstract description 136
- CTIRHWCPXYGDGF-HDICACEKSA-N tedisamil Chemical compound [H][C@]12CN(CC3CC3)C[C@]([H])(CN(CC3CC3)C1)C21CCCC1 CTIRHWCPXYGDGF-HDICACEKSA-N 0.000 abstract description 122
- 238000000034 method Methods 0.000 abstract description 15
- 239000000825 pharmaceutical preparation Substances 0.000 abstract description 12
- 229940127557 pharmaceutical product Drugs 0.000 abstract description 7
- 239000000902 placebo Substances 0.000 description 89
- 229940068196 placebo Drugs 0.000 description 89
- 239000003814 drug Substances 0.000 description 34
- 206010003662 Atrial flutter Diseases 0.000 description 31
- 238000001990 intravenous administration Methods 0.000 description 31
- 229940079593 drug Drugs 0.000 description 30
- 150000001875 compounds Chemical class 0.000 description 24
- 230000000694 effects Effects 0.000 description 23
- 230000000977 initiatory effect Effects 0.000 description 21
- 238000004458 analytical method Methods 0.000 description 20
- 230000037396 body weight Effects 0.000 description 18
- AZPYPAVFNGFTGB-ORRJWUSCSA-N (1S,5R)-3,7-bis(cyclopropylmethyl)spiro[3-aza-7-azoniabicyclo[3.3.1]nonane-9,1'-cyclopentane] (E)-but-2-enedioate (E)-but-2-enedioic acid Chemical compound OC(=O)\C=C\C(=O)O.OC(=O)\C=C\C(=O)O.OC(=O)\C=C\C(=O)O.C(C1CC1)N2C[C@@H]3CN(CC4CC4)C[C@H](C2)C35CCCC5.C(C6CC6)N7C[C@@H]8CN(CC9CC9)C[C@H](C7)C8%10CCCC%10 AZPYPAVFNGFTGB-ORRJWUSCSA-N 0.000 description 14
- 238000002565 electrocardiography Methods 0.000 description 14
- 238000013194 cardioversion Methods 0.000 description 12
- 230000003285 pharmacodynamic effect Effects 0.000 description 12
- 230000036470 plasma concentration Effects 0.000 description 12
- NXXZICGFHYJPGE-UHFFFAOYSA-N 3,7-bis(cyclopropylmethyl)spiro[3,7-diazabicyclo[3.3.1]nonane-9,1'-cyclopentane];dihydrochloride Chemical compound Cl.Cl.C1C(C23CCCC3)CN(CC3CC3)CC2CN1CC1CC1 NXXZICGFHYJPGE-UHFFFAOYSA-N 0.000 description 10
- 238000011156 evaluation Methods 0.000 description 10
- 230000002411 adverse Effects 0.000 description 9
- 238000002347 injection Methods 0.000 description 9
- 239000007924 injection Substances 0.000 description 9
- 238000012216 screening Methods 0.000 description 9
- 230000004044 response Effects 0.000 description 8
- 239000000243 solution Substances 0.000 description 8
- 239000012458 free base Substances 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 238000004088 simulation Methods 0.000 description 7
- 238000012360 testing method Methods 0.000 description 7
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 239000013543 active substance Substances 0.000 description 6
- 239000000725 suspension Substances 0.000 description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 5
- 239000002585 base Substances 0.000 description 5
- 239000002876 beta blocker Substances 0.000 description 5
- 229940097320 beta blocking agent Drugs 0.000 description 5
- 230000036772 blood pressure Effects 0.000 description 5
- 239000012669 liquid formulation Substances 0.000 description 5
- 239000013610 patient sample Substances 0.000 description 5
- 230000000306 recurrent effect Effects 0.000 description 5
- 238000007619 statistical method Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 4
- 208000018452 Torsade de pointes Diseases 0.000 description 4
- 208000002363 Torsades de Pointes Diseases 0.000 description 4
- 239000002671 adjuvant Substances 0.000 description 4
- 230000001174 ascending effect Effects 0.000 description 4
- 239000000839 emulsion Substances 0.000 description 4
- 239000004615 ingredient Substances 0.000 description 4
- 238000010253 intravenous injection Methods 0.000 description 4
- 239000011591 potassium Substances 0.000 description 4
- 229910052700 potassium Inorganic materials 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 206010003119 arrhythmia Diseases 0.000 description 3
- 230000001746 atrial effect Effects 0.000 description 3
- 230000000903 blocking effect Effects 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 229940124301 concurrent medication Drugs 0.000 description 3
- 238000013461 design Methods 0.000 description 3
- 238000003745 diagnosis Methods 0.000 description 3
- 229960004166 diltiazem Drugs 0.000 description 3
- HSUGRBWQSSZJOP-RTWAWAEBSA-N diltiazem Chemical compound C1=CC(OC)=CC=C1[C@H]1[C@@H](OC(C)=O)C(=O)N(CCN(C)C)C2=CC=CC=C2S1 HSUGRBWQSSZJOP-RTWAWAEBSA-N 0.000 description 3
- 231100000673 dose–response relationship Toxicity 0.000 description 3
- 230000002349 favourable effect Effects 0.000 description 3
- 238000012544 monitoring process Methods 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 230000000144 pharmacologic effect Effects 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 230000002861 ventricular Effects 0.000 description 3
- 239000008215 water for injection Substances 0.000 description 3
- ITPDYQOUSLNIHG-UHFFFAOYSA-N Amiodarone hydrochloride Chemical compound [Cl-].CCCCC=1OC2=CC=CC=C2C=1C(=O)C1=CC(I)=C(OCC[NH+](CC)CC)C(I)=C1 ITPDYQOUSLNIHG-UHFFFAOYSA-N 0.000 description 2
- 0 CC(*)C(CCN(*)C1)C1C1(*)CC1 Chemical compound CC(*)C(CCN(*)C1)C1C1(*)CC1 0.000 description 2
- 206010007559 Cardiac failure congestive Diseases 0.000 description 2
- 241000208011 Digitalis Species 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- 206010019280 Heart failures Diseases 0.000 description 2
- 206010022095 Injection Site reaction Diseases 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- 102000004257 Potassium Channel Human genes 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 230000002159 abnormal effect Effects 0.000 description 2
- 230000036982 action potential Effects 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 229960005260 amiodarone Drugs 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 238000009530 blood pressure measurement Methods 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N coumarin Chemical compound C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 2
- DDRJAANPRJIHGJ-UHFFFAOYSA-N creatinine Chemical compound CN1CC(=O)NC1=N DDRJAANPRJIHGJ-UHFFFAOYSA-N 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000035487 diastolic blood pressure Effects 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 238000001647 drug administration Methods 0.000 description 2
- 230000009977 dual effect Effects 0.000 description 2
- 230000007717 exclusion Effects 0.000 description 2
- 238000009532 heart rate measurement Methods 0.000 description 2
- 229960000443 hydrochloric acid Drugs 0.000 description 2
- 235000011167 hydrochloric acid Nutrition 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- 238000002483 medication Methods 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 229940124531 pharmaceutical excipient Drugs 0.000 description 2
- 108020001213 potassium channel Proteins 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 238000012552 review Methods 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 230000002459 sustained effect Effects 0.000 description 2
- 230000035488 systolic blood pressure Effects 0.000 description 2
- 238000002560 therapeutic procedure Methods 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- 239000003981 vehicle Substances 0.000 description 2
- SGTNSNPWRIOYBX-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-{[2-(3,4-dimethoxyphenyl)ethyl](methyl)amino}-2-(propan-2-yl)pentanenitrile Chemical compound C1=C(OC)C(OC)=CC=C1CCN(C)CCCC(C#N)(C(C)C)C1=CC=C(OC)C(OC)=C1 SGTNSNPWRIOYBX-UHFFFAOYSA-N 0.000 description 1
- KKJUPNGICOCCDW-UHFFFAOYSA-N 7-N,N-Dimethylamino-1,2,3,4,5-pentathiocyclooctane Chemical compound CN(C)C1CSSSSSC1 KKJUPNGICOCCDW-UHFFFAOYSA-N 0.000 description 1
- 102100021177 ATP-sensitive inward rectifier potassium channel 11 Human genes 0.000 description 1
- 101710122998 ATP-sensitive inward rectifier potassium channel 11 Proteins 0.000 description 1
- 208000004476 Acute Coronary Syndrome Diseases 0.000 description 1
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 1
- 206010003673 Atrioventricular block complete Diseases 0.000 description 1
- 206010008190 Cerebrovascular accident Diseases 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- WDJUZGPOPHTGOT-OAXVISGBSA-N Digitoxin Natural products O([C@H]1[C@@H](C)O[C@@H](O[C@@H]2C[C@@H]3[C@@](C)([C@@H]4[C@H]([C@]5(O)[C@@](C)([C@H](C6=CC(=O)OC6)CC5)CC4)CC3)CC2)C[C@H]1O)[C@H]1O[C@@H](C)[C@H](O[C@H]2O[C@@H](C)[C@@H](O)[C@@H](O)C2)[C@@H](O)C1 WDJUZGPOPHTGOT-OAXVISGBSA-N 0.000 description 1
- LTMHDMANZUZIPE-AMTYYWEZSA-N Digoxin Natural products O([C@H]1[C@H](C)O[C@H](O[C@@H]2C[C@@H]3[C@@](C)([C@@H]4[C@H]([C@]5(O)[C@](C)([C@H](O)C4)[C@H](C4=CC(=O)OC4)CC5)CC3)CC2)C[C@@H]1O)[C@H]1O[C@H](C)[C@@H](O[C@H]2O[C@@H](C)[C@H](O)[C@@H](O)C2)[C@@H](O)C1 LTMHDMANZUZIPE-AMTYYWEZSA-N 0.000 description 1
- 206010013654 Drug abuse Diseases 0.000 description 1
- 206010013700 Drug hypersensitivity Diseases 0.000 description 1
- 206010013710 Drug interaction Diseases 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 206010020850 Hyperthyroidism Diseases 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 206010022086 Injection site pain Diseases 0.000 description 1
- 238000012773 Laboratory assay Methods 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 208000004880 Polyuria Diseases 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 208000007536 Thrombosis Diseases 0.000 description 1
- 206010070863 Toxicity to various agents Diseases 0.000 description 1
- 206010047281 Ventricular arrhythmia Diseases 0.000 description 1
- 206010049761 Ventricular pre-excitation Diseases 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- 229960001138 acetylsalicylic acid Drugs 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 230000007815 allergy Effects 0.000 description 1
- 239000003708 ampul Substances 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000003257 anti-anginal effect Effects 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 230000002253 anti-ischaemic effect Effects 0.000 description 1
- 239000003146 anticoagulant agent Substances 0.000 description 1
- 229940127219 anticoagulant drug Drugs 0.000 description 1
- 230000010100 anticoagulation Effects 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 239000003429 antifungal agent Substances 0.000 description 1
- 229940030600 antihypertensive agent Drugs 0.000 description 1
- 239000002220 antihypertensive agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 230000006793 arrhythmia Effects 0.000 description 1
- 230000003190 augmentative effect Effects 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 150000008107 benzenesulfonic acids Chemical class 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 230000036765 blood level Effects 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 238000007675 cardiac surgery Methods 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 208000026106 cerebrovascular disease Diseases 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 229960004926 chlorobutanol Drugs 0.000 description 1
- 210000002808 connective tissue Anatomy 0.000 description 1
- 239000003433 contraceptive agent Substances 0.000 description 1
- 230000002254 contraceptive effect Effects 0.000 description 1
- 235000001671 coumarin Nutrition 0.000 description 1
- 229960000956 coumarin Drugs 0.000 description 1
- 229940109239 creatinine Drugs 0.000 description 1
- 230000006735 deficit Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 150000001991 dicarboxylic acids Chemical class 0.000 description 1
- 229960000648 digitoxin Drugs 0.000 description 1
- WDJUZGPOPHTGOT-XUDUSOBPSA-N digitoxin Chemical compound C1[C@H](O)[C@H](O)[C@@H](C)O[C@H]1O[C@@H]1[C@@H](C)O[C@@H](O[C@@H]2[C@H](O[C@@H](O[C@@H]3C[C@@H]4[C@]([C@@H]5[C@H]([C@]6(CC[C@@H]([C@@]6(C)CC5)C=5COC(=O)C=5)O)CC4)(C)CC3)C[C@@H]2O)C)C[C@@H]1O WDJUZGPOPHTGOT-XUDUSOBPSA-N 0.000 description 1
- 229960005156 digoxin Drugs 0.000 description 1
- LTMHDMANZUZIPE-PUGKRICDSA-N digoxin Chemical compound C1[C@H](O)[C@H](O)[C@@H](C)O[C@H]1O[C@@H]1[C@@H](C)O[C@@H](O[C@@H]2[C@H](O[C@@H](O[C@@H]3C[C@@H]4[C@]([C@@H]5[C@H]([C@]6(CC[C@@H]([C@@]6(C)[C@H](O)C5)C=5COC(=O)C=5)O)CC4)(C)CC3)C[C@@H]2O)C)C[C@@H]1O LTMHDMANZUZIPE-PUGKRICDSA-N 0.000 description 1
- LTMHDMANZUZIPE-UHFFFAOYSA-N digoxine Natural products C1C(O)C(O)C(C)OC1OC1C(C)OC(OC2C(OC(OC3CC4C(C5C(C6(CCC(C6(C)C(O)C5)C=5COC(=O)C=5)O)CC4)(C)CC3)CC2O)C)CC1O LTMHDMANZUZIPE-UHFFFAOYSA-N 0.000 description 1
- 239000002866 dihydropyridine calcium channel blocker Substances 0.000 description 1
- 229940127292 dihydropyridine calcium channel blocker Drugs 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 230000001882 diuretic effect Effects 0.000 description 1
- 201000005311 drug allergy Diseases 0.000 description 1
- 229940000406 drug candidate Drugs 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 230000003073 embolic effect Effects 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 230000002169 extracardiac Effects 0.000 description 1
- 208000015700 familial long QT syndrome Diseases 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 238000002695 general anesthesia Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 210000002837 heart atrium Anatomy 0.000 description 1
- 208000019622 heart disease Diseases 0.000 description 1
- 230000002489 hematologic effect Effects 0.000 description 1
- 230000000004 hemodynamic effect Effects 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 208000006454 hepatitis Diseases 0.000 description 1
- 231100000283 hepatitis Toxicity 0.000 description 1
- 150000003840 hydrochlorides Chemical class 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 239000003978 infusion fluid Substances 0.000 description 1
- 230000000297 inotrophic effect Effects 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 238000001325 log-rank test Methods 0.000 description 1
- 238000007477 logistic regression Methods 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 238000007726 management method Methods 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 239000004530 micro-emulsion Substances 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 239000007908 nanoemulsion Substances 0.000 description 1
- 239000006070 nanosuspension Substances 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 229940097271 other diuretics in atc Drugs 0.000 description 1
- 238000010979 pH adjustment Methods 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 208000035824 paresthesia Diseases 0.000 description 1
- 229960003742 phenol Drugs 0.000 description 1
- 239000003286 potassium sparing diuretic agent Substances 0.000 description 1
- 229940097241 potassium-sparing diuretic Drugs 0.000 description 1
- 238000009597 pregnancy test Methods 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000001536 pro-arrhythmogenic effect Effects 0.000 description 1
- 230000036279 refractory period Effects 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 201000002932 second-degree atrioventricular block Diseases 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000002047 solid lipid nanoparticle Substances 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 229960002370 sotalol Drugs 0.000 description 1
- ZBMZVLHSJCTVON-UHFFFAOYSA-N sotalol Chemical compound CC(C)NCC(O)C1=CC=C(NS(C)(=O)=O)C=C1 ZBMZVLHSJCTVON-UHFFFAOYSA-N 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008174 sterile solution Substances 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 208000011117 substance-related disease Diseases 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 150000003460 sulfonic acids Chemical class 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 201000002931 third-degree atrioventricular block Diseases 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 238000009810 tubal ligation Methods 0.000 description 1
- 238000002562 urinalysis Methods 0.000 description 1
- 210000002700 urine Anatomy 0.000 description 1
- 229960001722 verapamil Drugs 0.000 description 1
- 238000011179 visual inspection Methods 0.000 description 1
- 229960005080 warfarin Drugs 0.000 description 1
- PJVWKTKQMONHTI-UHFFFAOYSA-N warfarin Chemical compound OC=1C2=CC=CC=C2OC(=O)C=1C(CC(=O)C)C1=CC=CC=C1 PJVWKTKQMONHTI-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/439—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom the ring forming part of a bridged ring system, e.g. quinuclidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
- A61K31/4747—Quinolines; Isoquinolines spiro-condensed
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/06—Antiarrhythmics
Definitions
- the present invention relates to the use of 3,7-diazabicyclo[3,3,1]nonane compounds of formula I, preferably of 9,9-alkylene-3,7-diazabicyclo[3,3,1]nonane compounds of formula I, and most preferably to the use of tedisamil, and the physiologically acceptable acid addition salts and/or solvates thereof, in the form of a liquid pharmaceutical preparation for the treatment and/or prophylaxis of arrhythmic events in human patients, preferably in conversion of recent onset of atrial fibrillation (Afib) or flutter to normal sinus rhythm (NSR) in human patients.
- Afib atrial fibrillation
- NSR normal sinus rhythm
- 9,9-Alkylene-3,7-diazabicyclononane compounds of formula I and their pharmacological activities are known from published European Patent No. EP 103,833 and the corresponding U.S. Pat. No. 4,550,112 , and Finnish Patent No. FI 76,338 .
- Compounds are a sub-group of the 9,9-N,N'-tetra-substituted 3,7-diazabicyclo[3.3.1]nonane compounds described in the aforementioned patent specifications and can be prepared by the methods described therein.
- the aforementioned patent specifications disclose that the compounds of formula I have useful cardio-active properties, particularly oxygen-saving effects and effects on the heart rate and heart rhythm in general, and are distinguished by a high physiological tolerance.
- the compounds of formula I show a satisfactory anti-arrhythmic action even at low doses. Moreover, the undesired negative effect on the contractile power of the heart is extremely low; i.e. the compounds of formula I have a particularly favourable ratio of anti-arrhythmic or the refractory period of the heart prolonging activities, to negative inotropic secondary activities.
- WO 9931100 discloses various anti-arrythmic effects of 3,7-diazabicyclo (3.3.1) nonanes which are administered as infusions. A dosing regimen with 6-10 doses is disclosed.
- Atrial fibrillation is the most common sustained cardiac arrhythmia requiring hospitalization, and affects millions of people worldwide.
- AFib is the most common sustained cardiac arrhythmia requiring hospitalization, and affects millions of people worldwide.
- the prevalence of AFib increases with age and in the presence of structural heart disease, it is also slightly more common in men than women.
- AFib is associated with significant mortality and morbidity and impacts significantly on quality of life.
- Restoration of normal sinus rhythm (NSR) in patients with these arrhythmias may improve their hemodynamic condition, relieve symptoms, and probably reduce embolic risk. Although this may be achieved using DC cardioversion, the technique has limitations such as the need for general anesthesia and hospitalization. Accordingly, pharmacological conversion has been proposed as an attractive alternative; a variety of anti-arrhythmic drugs have been tested for this purpose.
- Tedisamil is a novel class III anti-arrhythmic agent that blocks multiple potassium-channels and slows sinus rate. It prolongs both atrial and ventricular action potential duration by blocking the transient outward I to , the ATP-dependent I K-ATP , and the delayed rectifier potassium currents I Kr , I Ks , and I Ku . Tedisamil prolongs action potential duration more strongly in the atria than the ventricles. Unlike other selective potassium-channel blocking drugs, tedisamil does not exhibit reverse rate-dependent effects on atrial refractoriness, which makes it far less likely to be pro-arrhythmic. Finally, tedisamil also possesses significant anti-anginal and anti-ischemic properties. The present prospective, randomized, controlled trial evaluated the efficacy and safety of tedisamil for rapid conversion of AFib or AFlu.
- the objects of the invention are achieved by surprisingly discovering that the known beneficial pharmacological properties of 3.7-diazabicyclo-[3,3,1]-nonane compounds of formula I, preferably of 9,9-alkylene-3,7-diazabicyclo[3,3,1]-nonane compounds, and most preferably tedisamil, and of pharmaceutically acceptable acid addition salts and/or solvates of said compounds of formula I particularly may be further enhanced in the treatment and/or prophylaxis of arrhytmic events in human patients, preferably in conversion of recent onset of atrial fibrillation (Afib) or flutter to normal sinus rhythm (NSR) in human patients, when being administered as a liquid formulation according to a specific administration route or scheme instead of a completely single and/or continuous administration.
- an anti-arrhythmic liquid pharmaceutical formulation comprising an anti-arrhythmic amount effective in human patients of at least one anti-arrhythmic active 3,7-diaza-bicyclo[3,3,1]-nonane compound of formula I as described in the present invention.
- liquid means any fluid form of the 3,7-diaza-bicyclo-[3,3,1]nonane compound of formula I, a physiologically acceptable acid addition salt and/or solvate thereof, suitable for administration to a human patient.
- the liquid formulation may be a solution, an emulsion including micro-emulsion and nano-emulsion, or a suspension including micro- suspension and nano-suspension.
- phasewise indicate that the liquid formulation according to the invention is applied in one step with at least two phases with distinct administration dosage scheme.
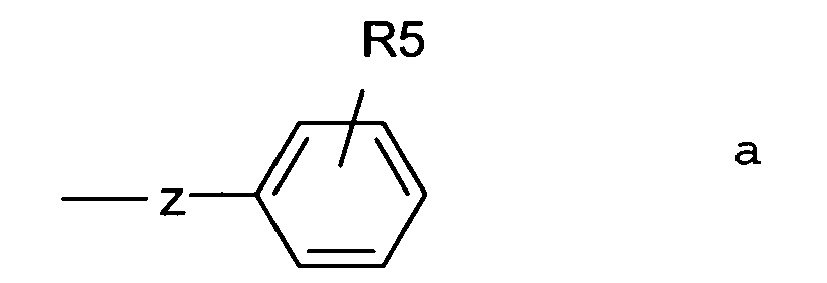
- Particularly suited compounds according to the invention are compounds of formula I, wherein R1 represents an alkyl group containing from 1 to 6 carbon atoms or a cycloalkylalkyl group containing from 4 to 7 carbon atoms.
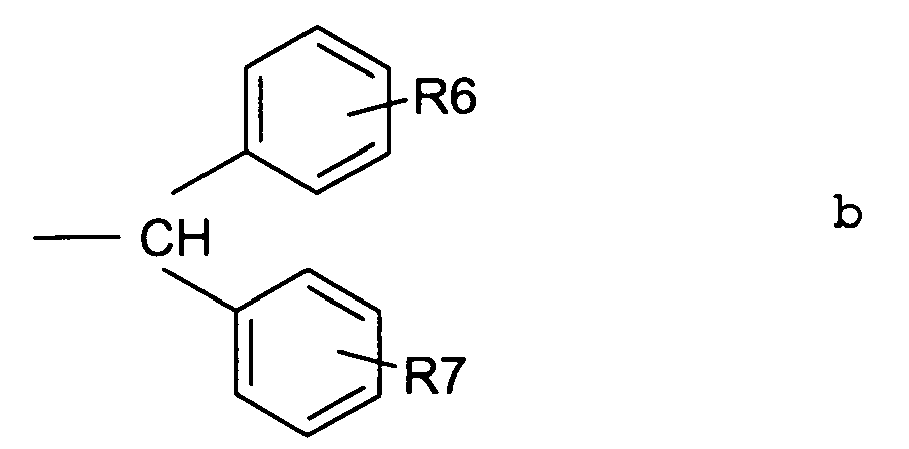
- R4 represents an alkyl group containing from 1 to 6 carbon atoms, a cycloalkylalkyl group containing from 4 to 7 carbon atoms, or a group corresponding to Formula b.
- Preferred compounds of formula I suitable according to the present invention are compounds, wherein R1 represents an alkyl group containing from 3 to 6 carbon atoms or a cycloalkylalkyl group containing from 4 to 7 carbon atoms, and R4 represents an alkyl group containing from 3 to 6 carbon atoms or a cycloalkylalkyl group containing from 4 to 7 carbon atoms.
- Said 3,7-diazabicyclo-[3,3,1]nonane compound of formula I may be a 9,9-alkylene-3,7-diazabicyclo[3,3,1]nonane compound of Formula I wherein R2 and R3 together form an alkylene chain containing from 4 to 5 carbon atoms, and R1 and R4 independently of one another each denote a straight-chain or branched alkyl group of 3-4 carbon atoms or the cyclopropylmethyl group, and physiologically acceptable acid addition salts and/or solvates thereof.
- Preferred salts for this group of compounds of formula I are fumaric acid salts of 9,9-alkylene-3,7-diazabicyclo[3,3,1]nonane compounds containing 1.5 moles of fumaric acid per mole of compound of Formula I.
- suitable compounds of formula I are compounds of formula I selected from the group consisting of N,N'-dicyclopropyl-methyl-9, 9-tetramethylen-3,7-diazabicyclo[3,3,1]nonane (tedisamil), N-isobutyl-N'-isopropyl-9,9-pentamethylen-3,7-diazabicyclo[3,3,1]nonane, and physiologically acceptable acid addition salts and/or solvates thereof.
- Preferred salts for this group of compounds of formula I are fumaric acid salts of N,N'-dicyclopropylmethyl-9, 9-tetramethylene-3,7-diazabicyclo [3,3,1]nonane (tedisamil) or of N-isobutyl-N'-isopropyl-9,9-pentamethylene-3,7-diazabicyclo[3,3,1]nonane containing 1.5 moles of fumaric acid per mole of said 9,9-alkylene-3,7-diazabicyclo[3,3,1]-nonane compound.
- hydrochloride salts are also very suitable in the context of the present invention.
- Particularly preferred 3,7-diazabicyclo[3,3,1]nonane compounds of formula I are the 9,9-alkylene-3,7-diazabicydo[3.3.1]nonane compound tedisamil and the physiologically compatible acid addition salts and/or solvates thereof. These are most preferably used as compounds in the liquid pharmaceutical formulations. If a tedisamil acid addition salt is used, it may preferably be used according to the invention in the form of tedisamil hydrochloride or in the form of tedisamil sesquifumarate. Further pharmacologically compatible acid addition salts of tedisamil are known from European Patent No. EP 103,833 . Thus, salts with inorganic acids, e.g.
- sulfuric acid or hydrohalic acids especially hydrochloric acid
- organic acids for instance lower aliphatic monocarboxylic or dicarboxylic acids such as acetic acid, fumaric acid, tartaric acid, lactic acid, maleic acid, citric acid or salicylic acid
- sulfonic acids for instance lower alkyl sulfonic acids such as methane sulfonic acid, or benzene sulfonic acids optionally substituted in the benzene ring by halogen or lower alkyl, such as p-toluene sulfonic acid, are suitable as physiologically acceptable acid addition salts of the compounds of formula I.
- the 3,7,9, 9-tetra-substituted 3,7-diazabicyclo[3,3,1]nonane compounds corresponding to formula I are distinguished by superior enhanced effects in arrhythmic human patients, in particular in the conversion of recent onset of atrial fibrillation (Afib) or flutter to normal sinus rhythm (NSR) in human patients, in addition to the aforementioned already known general heart-affecting properties, when administered as a liquid pharmaceutical formulation by infusion of a dose consisting of two subsequent phases, such as that in the first phase a first part of the infusion is administered at a first rate, and in the second phase a second part of the infusion is administered at a second, slower rate.
- Afib atrial fibrillation
- NSR normal sinus rhythm
- the use of the liquid pharmaceutical formulation according to the present invention may be containing a first amount of said 3,7-diaza-bicyclo[3,3,1]nonane compound of formula I, a physiologically acceptable acid addition salt and/or solvate thereof, being sufficient for the treatment and/or prophylaxis of arrhythmic events in human patients is administered by infusion of a dose at a first rate over a time period of about 10 ⁇ 2 minutes, preferably of about 10 ⁇ 1 minutes, most preferably of about 10 ⁇ 0.5 minutes, and wherein a liquid pharmaceutical formulation comprising a second amount of said 3,7-diazabicyclo[3,3,1]nonane compound, a physiologically acceptable acid addition salt and/or solvate thereof, being sufficient for continuing the treatment and/or prophylaxis, is foreseen to be administered at a second, slower rate over a second time period of about 20 ⁇ 5 minutes, preferably of about 20 ⁇ 3 minutes, most preferably of about 20 ⁇ 1 minute as claimed.
- a further embodiment of the invention pertains to the use of pharmaceutical products.
- the invention pertains to the use of a pharmaceutical product containing as a medicament in a liquid pharmaceutical formulation at least one 3,7-diazabicyclo[3,3,1]nonane compound of formula I, at least a physiologically acceptable acid addition salt and/or solvate thereof, as defined above, the use thereof being described in a label, leaflet and/or package insert indicating that said 3,7-diazabicyclo[3,3,1]nonane compound of formula I may be administered by two-phase administration of two continuous administration phases of an infusion dose as claimed.
- Such a pharmaceutical product and/or package may be characterized in that the label, leaflet and/or package insert outlines that the liquid pharmaceutical formulation is suited for infusion, preferably for phasewise infusion administration, preferably for two-phase infusion administration of two continuous infusion administration phases of an infusion dose.
- the pharmaceutical product and/or package is characterized in that the label, leaflet and/or package insert outlines that the 3,7-diazabicyclo-[3,3,1]-nonane compound of formula I may be administered according to an administration scheme by infusion of a dose consisting of two subsequent phases in which a liquid pharmaceutical formulation comprising a first amount of said 3,7-diazabicyclo-[3,3,1]nonane compound of formula I, a physiologically acceptable acid addition salt and/or solvate thereof, being sufficient for the treatment and/or prophylaxis of arrhythmic events in human patients, is administered over a first part of the infusion in a first rate of about 10 ⁇ 2 minutes, preferably of about 10 ⁇ 1 minutes, most preferably of about 10 ⁇ 0.5 minutes, and a second amount of said 3,7-diazabicyclo[3,3,1]nonane compound of formula I, a physiologically acceptable acid addition salt and/or solvate thereof, being sufficient for continuing the treatment and/or prophyl
- the pharmaceutical product and/or package is containing as a medicament a amount of at least one 3,7-diazabicyclo[3,3,1]nonane compound of formula I, a physiologically acceptable acid addition salt and/or solvate thereof, as defined above, in the liquid pharmaceutical formulation.
- the embodiments are suited for being administered as a two-phase infusion regimen, wherein over a first time period of about 10 ⁇ 2 minutes, preferably of about 10 ⁇ 1 minutes, most preferably of about 10 ⁇ 0.5 minutes, a first rate -of said 3,7-diazabicyclo[3,3,1]-nonane compound of formula I, a physiologically acceptable acid addition salt and/or solvate thereof, being sufficient for the treatment and/or prophylaxis of an arrhythmic event in a patient, and then subsequently for continuing the treatment and/or prophylaxis, a second rate of said 3,7-diazabicyclo[3,3,1]-nonane compound of formula I, a physiologically acceptable acid addition salt and/or solvate thereof, being sufficient for continuing the treatment and/or prophylaxis is administered over a second time period of about 20 ⁇ 5 minutes, preferably of about 20 ⁇ 3
- the most recommended dosages for the two phases shall be exemplified in the following in some more detail. However, it is clear that the skilled artisan may modify to a certain extend this dosage scheme, if appropriate.
- the total amount of the 3,7-diazabicyclo[3,3,1]-nonane compound of formula I, preferably tedisamil or one of its salts, based on the quantity of the base compound may range from about 0.16 mg/kg to about 0.64 mg/kg.
- Examples of specific amounts to be administered by the preferred a two phase infusion regimen may amount within this range to 0.16 mg/kg, 0.24 mg/kg, 0.32 mg/kg, 0.48 mg/kg or 0.64 mg/kg.
- About half e.g.
- 50 ⁇ 5 % by wt.) of this total amount (100 % by wt.) is administered in the first time period of e.g. about 10 ⁇ 2 minutes, and about the second half (e.g. the remaining 50 ⁇ 5 % by wt. up to 100 % by wt.) is administered over a second time period of e.g. about 20 ⁇ 5 minutes.
- 0.32 mg/kg tedisamil base was used, infusing half the dose over 10 minutes and half the dose over 20 minutes.
- the present invention is applicable to patients of any age and/or both genders.
- the findings of the present invention are particularly suitable also for the treatment and/or prophylaxis of arrhythmic events in male patients, preferably in conversion of recent onset of atrial fibrillation (Afib) or flutter to normal sinus rhythm (NSR) in male patients.
- the invention also pertains to a method of treatment and/or prophylaxis of arrhythmic events in male patients, preferably in conversion of recent onset of atrial fibrillation (Afib) or flutter to normal sinus rhythm (NSR) in male patients, wherein a liquid pharmaceutical formulation comprising at least one 3,7-diaza-bicyclo-[3,3,1]-nonane compound of formula I, or a physiologically acceptable acid addition salt and/or solvate thereof, is administered to said male patients, by a phasewise infusion administration route, but most preferably when administered as a two-phase administration of two administration phases.
- the invention can also be used in liquid pharmaceutical formulations, which are suited for phasewise, preferably suited for administration as a two-phase administration of two administration phases of the 3,7-diaza-bicyclo-[3,3,1]-nonane compounds of formula I, e.g. preferably of tedisamil and its acid addition salts, to a male patient.
- a further aspect of this embodiment of the invention related to the treating of male patients is directed to the use in a pharmaceutical product and/or package containing as a medicament in a liquid pharmaceutical formulation at least one 3,7-diazabicyclo[3,3,1]-nonane compound of formula I, at least a physiologically acceptable acid addition salt and/or solvate thereof, and the use of a label, leaflet and/or package insert indicating that said 3,7-diazabicyclo[3,3,1]-nonane compound of formula I may be administered phase-wisely, as a two-phase administration of two administration phase as claimed, preferably to a male patient.
- the details described in the context of the above general embodiments of invention fully apply also to this particular embodiment related to the treatment and/or prophylaxis of arrhythmic events in male patients.
- the superior anti-arrhythmic effect of the compounds of Formula I in human patients can be demonstrated by PK/PD modelling and is confirmed by clinical study data which prove the surprising enhanced suitability and efficacy of 3,7-diazabicyclo-[3,3,1]-nonane compounds of formula I, e.g. of tedisamil and its acid addition salts, for the treatment and/or prophylaxis of arrhythmic effects in human patients, preferably for conversion of recent onset of atrial fibrillation (Afib) or flutter to normal sinus rhythm (NSR) in human patients, when administered as a liquid pharmaceutical formulation by a phasewise administration scheme according to the present invention.
- Afib atrial fibrillation
- NSR normal sinus rhythm
- Tedisamil is a new class III antiarrhythmic agent, blocking various potassium outward currents such as IKur, Ito, IKr, IKs, and IKATP, under development for the rapid conversion of recent onset atrial fibrillation (AF) and flutter (AFL) into normal sinus rhythm (NSR).
- a two phase infusion regimen was developed using a PK/PD model to enhance drug concentration/exposure at the effect site. The PK/PD model was based on the data from a single infusion study.
- a three-compartment infusion PK model and an effect compartment model with linear link gave the best fit.
- the model for the PD effect was a simple linear model.
- the modeling results were validated in a double-blind, placebo-controlled, randomized, two period cross-over study in 18 healthy male subjects, in which a two phase infusion regimen of 0.32 mg/kg TEDI base was used, infusing half the dose over 10 minutes and half the dose over 20 minutes.
- Aim The objectives of the study were to assess safety and tolerability and the correlation between TEDI plasma concentrations and QT, QTc, and heart rate, and compare these results with the simulation data.
- TEDI was safe and well tolerated.
- the expected PD action i.e. a prolongation of QT and QTc intervals and a distinct decrease in heart rate, was observed over two hours.
- the PK and PD results were in close agreement with the simulations based on the single infusion PK/PD model: the simulation based on single infusion estimates lied within the 95 % confidence interval around the predicted population mean of the study.
- the two phase infusion regimen is of benefit in the administration of 3,7-diazabicyclo[3,3,1]nonane compounds of formula I, e.g. of tedisamil and its acid addition salts, for the treatment and/or prophylaxis of arrhythmic effects, preferably for conversion of recent onset of atrial fibrillation (Afib) or flutter to normal sinus rhythm (NSR), when administered as a liquid pharmaceutical formulation by a phasewise administration scheme, thereby increasing the time of exposure at the physiologically effect site.
- 3,7-diazabicyclo[3,3,1]nonane compounds of formula I e.g. of tedisamil and its acid addition salts
- arrhythmic effects preferably for conversion of recent onset of atrial fibrillation (Afib) or flutter to normal sinus rhythm (NSR)
- Afib atrial fibrillation
- NSR normal sinus rhythm
- a double-blind, placebo-controlled, randomized, single iv dose, two periods cross-over study to assess the safety, tolerability, pharmacodynamics and pharmacokinetics during and following a two phase infusion of Tedisamil in healthy male subjects was performed with the following drug and dosages: Tedisamil dihydrochloride / 0.4 mg/kg body weight infused over 30 minutes.
- Test product, dose and mode of administration Tedisamil dihydrochloride; Strength: 2 mg/ml; Dose: 0.4 mg/kg body weight infused over 30 minutes, with half of the dose infused over 10 minutes and half of the dose infused over the remaining 20 minutes.
- Route intravenous.
- Screening assessments included physical examination, ECG, HIV / Hepatitis screening, blood pressure and heart rate measurements, clinical laboratory determinations, urine drug screen, alcohol breath test, and medical history.
- Follow-up assessments included physical examination, ECG, blood pressure and heart rate measurements, clinical laboratory determinations, and questions for adverse events and concomitant medication.
- ECG ECG
- blood pressure and heart rate measurements ECG
- clinical laboratory determinations ECG
- questions for adverse events and concomitant medication included physical examination, ECG, blood pressure and heart rate measurements, clinical laboratory determinations, and questions for adverse events and concomitant medication.
- Tedisamil blood pressure, heart rate, and ECGs were recorded. Adverse events and concomitant medication were monitored throughout the study.
- Descriptive statistics were performed for the ECG intervals QT, QTc, RR, PR, QRS and for heart rate by treatment (tedisamil, placebo and "tedisamil - placebo") and timepoint.
- Derived parameters i.e. maximum value and maximum increase for QTc and QT, and minimum value and maximum decrease for heart rate were submitted to separate analyses of (co)variance (covariance only for maximum and minimum values) including subject, treatment, sequence, and period as class effects (and the individual pre-dose value as covariate). Point estimates and 95% confidence intervals were constructed for the difference "tedisamil - placebo" using the residual variance. Model assumptions were checked by visual inspection of residual plots. Additionally, descriptive statistics were calculated for the derived parameters per treatment and for the difference "tedisamil-placebo".
- Descriptive statistics for the placebo corrected AUC(0-24) of QTc, QT and heart rate and the placebo corrected maximum effect (maximum for QTc and QT and minimum for heart rate) were calculated.
- 95% confidence intervals were derived based on normally distributed outcomes.
- Treatment emergent adverse events defined as any adverse event present at baseline that worsened or any event that began after treatment with study medication, were summarized by COSTART term for each treatment group. Each adverse event was evaluated for frequency of occurrence, duration, intensity, action taken, outcome, and relationship to administration of the study medication. Adverse events were listed for each subject.
- Geometric means and corresponding 90% confidence intervals were calculated for the plasma concentration of tedisamil at each time-point, for AUC, AUC(0-t), CL, and C max .
- Arithmetic means and corresponding 90% confidence intervals were calculated for the other variables (V ss , ⁇ z , t 1/2 , MRT). T max was described using minimum, median, maximum, and frequency counts. Individual and mean plasma-concentration versus time curves were plotted using both linear and semi-logarithmic scale.
- Non-linear mixed effect models were used to characterize the pharmacokinetics and pharmacodynamics of tedisamil after two-phase infusion.
- Population mean prediction profiles and confidence limits over time were obtained from the predicted PK and PD profiles generated for each subject.
- PK/PD compartment models The PK model for plasma concentration, Cp, was a three compartmental constant infusion model. To model observed PD effects on ECG parameters, a sequential PK/PD effect-compartment linear link model was used.
- NONMEM V was used for all pharmacokinetic modelling.
- the PK/PD linear-link model was fitted using using the SAS macro NLINMIX for non-linear mixed modelling.
- Table A Descriptive statistics for derived parameters of QT, QTc interval and heart rate are summarized in the Table A below.
- Table A Descriptive statistics for derived parameters Parameter Derived Parameter Tedisamil Arith. Mean (SD) Placebo Arith. Mean (SD) QT Interval Maximum 474.2 (39.2) 416.9 (19.4) (ms) Max. Increase 76.0 (22.4) 18.0 (11.5) QTc Interval Maximum 455.9 (30.0) 421.9 (21.5) (ms) Max. Increase 61.1 (30.3) 27.0 (13.9) Heart Rate Minimum 49.2(7.0) 52.7(4.8) (bpm) Max. Decrease 10.6(3.8) 6.4(3.4)
- Table B Summary of Analyses for the derived parameters of QT, QTc interval and heart rate for the comparison of tedisamil-placebo are given in the table below: Table B: Summary of Analyses for derived parameters Parameter Derived Parameter Estimate (Ted-Pla) 95% C. I. lower upper QT Interval Maximum 58.4 46.4 70.4 (ms) Max. Increase 57.9 44.7 71.1 QTc Interval Maximum 34.0 16.7 51.2 (ms) Max. Increase 34.1 16.1 52.1 Heart Rate Minimum - 4.01 - 5.96 - 2.06 (bpm) Max. Decrease 4.22 2.04 6.39
- Tedisamil has a strong effect on QT, QTc and heart in the first two hours post dosing. Thus in the considered AUC(0-24) the Tedisamil effect was masked by the chosen interval.
- Table D Descriptive statistics of the main pharmacokinetic parameters are given in the following Table D.
- Table D Descriptive statistics of the main pharmacokinetic parameters Parameter Geom. Mean 90% C. I. (lower, upper) AUC(0-inf), (ng*h/ml) 1715 (1554, 2088) AUC(0-t), (ng*h/ml) 1670 (1512, 2042) Cmax (ng/ml) 905 (826, 1086) t1/2 (h) 5.5* (4.88, 6.04) CL (ml/min) 315 (284, 386) * Arithmetic mean
- Time of peak plasma concentration ranged between 5 and 75 minutes after start of infusion and showed a median value of 12.5 minutes.
- the best-fitting pharmacokinetic model was the three-compartment constant infusion model.
- the PK modeling and the PK/PD modeling of this study were in close agreement with the simulations based on the PK/PD model of the single infusion study.
- the effect-compartment PK/PD model for infusion administration confers substantial extra predictive power for QT and QTc: correlations between the predicted tedisamil concentration in the effect compartment and QT and QTc (0.93 and 0.807 respectively) were higher than the correlations between observed plasma concentration and unadjusted QT and QTc (0.695 and 0.513 respectively).
- Tedisamil was safe and well tolerated following a two phase infusion of 0.4 mg Tedisamil dihydrochloride per kg body weight in healthy male subjects.
- Tedisamil i.e. a marked prolongation of QT and QTc interval and a distinct decrease in heart rate, was observed over a time period of two hours post dosing.
- the PK analysis has validated the three-compartment constant infusion model for describing both single and two-phase infusion pharmacokinetics, while the PK/PD analysis has validated the effect-compartment link model for describing both single and two-phase infusion pharmacodynamics. Simulation studies based on these models have a good predictive value.
- This multicenter trial was a double-blind, randomized, placebo-controlled, ascending dose groups study comparing the efficacy and safety of intravenous tedisamil treatment versus placebo in symptomatic patients with atrial fibrillation or flutter (> 3 hours and ⁇ 48 hours).
- the primary efficacy endpoint of this study was the termination of atrial fibrillation or flutter (for at least 60 seconds) within 2.5 hours after start of the infusion.
- patients were randomized to tedisamil or placebo with a sequential dose escalation approach.
- patients received 0.4 mg tedisamil//kg body weight or placebo over a 30 minutes infusion period, with half the dose infused during the first 10 minutes and half the dose infused over the remaining 20 minutes.
- patients received 0.6 mg tedisamil/kg body weight or placebo, with the same infusion regimen.
- the patient's heart rhythm was continuously monitored by telemetry for 24 hours. Digitalis and/or ß-blockers or diltiazem were allowed for heart rate control during the study.
- Antiarrhythmic drugs were allowed only after 24 hours after start of infusion to avoid any potential drug interactions with tedisamil.
- FIG. 1 A study flowchart is given in Fig. 1 .
- the primary objective of the study was to demonstrate the superiority of any dose of tedisamil to placebo in the termination of atrial fibrillation/flutter as measured by the percentage of patients converted to normal sinus rhythm (NSR) (for at least 60 seconds) at any time within 2.5 hours after the start of infusion.
- NSR normal sinus rhythm
- the safety objective was: To determine the safety and tolerability of tedisamil versus placebo.
- Class-I or Class-III antiarrhythmic medications were to be discontinued more than five elimination half-lives prior to randomization.
- Amiodarone was to be discontinued for three months prior to randomization.
- verapamil and any potentially PR prolonging drug were not allowed. Allowed medications included anticoagulants, warfarin and other coumarin derivates, heparin, antihypertensive agents including potassium sparing diuretics (alone or combination with other diuretics), ß-blockers (except Sotalol), dihydropyridine calcium channel blockers and diltiazem, aspirin, digoxin and digitoxin.
- Tedisamil dihydrochloride 0.4 mg/kg body weight, 0.6.mg/kg body weight was administered intravenously.
- the total infusion time was 30 minutes, with half the dose infused within 10 minutes, and half the dose infused within the remaining 20 minutes.
- Matching placebo was given intravenously.
- the percentage of patients converted to normal sinus rhythm (for at least 60 seconds) at any time within 2.5 hours after the initiation of the infusion of study drug.
- the intent-to-treat patient sample was analyzed for efficacy. All tests were two-sided and a p-value ⁇ 0.05 was considered significant. Differences in percentages of conversion were compared among the treatment groups using the Pearson chi-square statistic. Safety analysis was conducted on the safety patient sample. Numbers and percentages of patients with adverse events were calculated for each treatment group, using MedDRA preferred terms and body systems. Descriptive statistics were summarized by treatment group for changes from baseline in QT, heart rate and blood pressure.
- the treatment comparison versus placebo was statistically significant for both treatment groups (p ⁇ 0.001).
- the percentage of patients converted to normal sinus rhythm (for at least 60 seconds) at any time within 2.5 hours after the initiation of the infusion of study drug for patients with atrial flutter was 0% in the placebo group, and 11% and 27% in the tedisamil 0.4 mg and 0.6 mg group respectively.
- Table 1 Patients with atrial fibrillation Parameter Tedisamil Placebo 0.4 mg/kg 0.6 mg/kg Conversion to NSR at any time within 2.5 hrs after the start of infusion 24 out of 52 (46 %) 24 out of 42 (57 %) 4 out of 46 (9 %) p-value, Pearson ChiSq (vs placebo) ⁇ 0.001 ⁇ 0.001 Conversion to NSR at any time within 2.5 hrs and remaining in NSR until 2.5 hrs after the start of infusion 23 out of 52 (44%) 24 out of 42 (57 %) 4 out of 46 (9 %) p-value, Pearson ChiSq (vs placebo) ⁇ 0.001 ⁇ 0.001 Conversion to NSR at any time within 24 hrs after the start of infusion 26 out of 42 (62 %) 26 out of 37 (70 %) 12 out of 34(35%) p-value, Pearson ChiSq (vs placebo) 0.021 0.003 Conversion to NSR
- the results according to the present invention demonstrate that intravenously administered tedisamil is effective in the acute conversion of patients with atrial fibrillation or flutter of up to 48 hours duration. There was a conversion rate of 46% observed with 0.4 mg/kg tedisamil and of 57% at a dose 0.6 mg/kg. There was a rapid onset of action with an average time to cardioversion of 35 minutes after the start of tedisamil administration.
- the results for the primary and secondary efficacy parameters indicated a dose dependent and statistically significant difference between placebo and both, tedisamil 0.4 mg/kg and tedisamil 0.6 mg/kg in the rapid conversion of atrial fibrillation into normal sinus rhythm.
- the results for patients with atrial flutter were statistically significant for the high dose group.
- tedisamil given as a two-phase infusion regimen has shown to be effective in conversion of atrial fibrillation and flutter to NSR.
- Tedisamil was well tolerated, no sustained polymorphic VT were observed, no patient required DC cardioversion for treatment of VTs.
- a two phase infusion regimen for rapid conversion of recent onset atrial fibrillation/flutter to normal sinus rhythm may be in particular beneficial in male patients.
- This first clinical study was a Multi-Center, Double-Blind, Randomized, Placebo-Controlled, Sequential Ascending Dose Groups Study to evaluate the Efficacy and Safety of Intravenous Tedisamil in the Rapid Conversion to Normal Sinus Rhythm in Patients with Atrial Fibrillation or Flutter.
- the active ingredient used was tedisamil dihydrochloride.
- the study was designed as a phase II study and executed in 35 to 40 centers in 3 to 4 countries. Study duration: Screening: up to 48 hours; treatment (in-patient): single 30-minute infusion; safety follow up: 24 hours (in-patient) with continuous telemetry and 28 day safety follow up.
- the primary efficacy objective of the study was to demonstrate the superiority of any dose of tedisamil to placebo in the termination of atrial fibrillation/flutter as measured by the percentage of patients converted to normal sinus rhythm (NSR) (for at least 60 seconds) at any time within 2.5 hours after the start of infusion.
- NSR normal sinus rhythm
- Secondary efficacy objectives were to determine the percentage of patients remaining in sinus rhythm at 2.5 hours after initiation of the intravenous infusion of tedisamil versus placebo; to determine the percentage of patients remaining in sinus rhythm at 24 hours after initiation of the intravenous infusion of tedisamil versus placebo; to determine the time to conversion after the start of the infusion of tedisamil versus placebo; and to determine the dose- and plasma concentration-response relationships of tedisamil versus placebo.
- Safety objective determining the safety and tolerability of tedisamil versus placebo.
- a multi-center, double-blind, randomized, placebo-controlled, sequential ascending dose groups study to evaluate the efficacy and safety of intravenous tedisamil versus placebo.
- the study drug was infused over 30 minutes, receiving half the dose within 10 minutes and half the dose within the remaining 20 minutes
- the first patient group received 0.4 mg/kg bodyweight (bw), infused as 0.2 mg/kg bw within 10 minutes, continued with 0.2 mg/kg bw infused within 20 minutes.
- the next higher dose 0.6 mg/kg bw (0.3 mg/kg bw infused within 10 min, continued with 0.3 mg/kg bw infused within 20 minutes) was only administered after the initial dose has been assessed (blinded) and found to be safe.
- a third stage may be added with a higher dose. Tedisamil blood concentrations were assessed during the infusion (at 10 and 30 minutes), at conversion to normal sinus rhythm, at recurrence and 24 hours after start of infusion.
- Planned number of subjects 330 randomized (110 patients per dose group)
- Atrial fibrillation or flutter with a duration of > 3 hours and ⁇ 48 hours, occurring as a first or recurrent episode.
- Placebo (vehicle) administered as a 30-minute intravenous infusion.
- the total infusion time is 30 minutes, with half the dose infused within 10 minutes, and half the dose infused within the remaining 20 minutes.
- Interim analyses for efficacy was performed halfway the first and second stages by an external statistician. The purpose is to terminate a stage when the tedisamil dose used in that stage is inefficacious.
- each interim analysis was a predictive power calculation for the comparison of tedisamil and placebo with respect to the primary efficacy variable.
- Blinded safety reviews were performed at the same time as the efficacy analyses and, in addition, at the end of the each stage (if applicable).
- the analysis of a second study confirmed the finding of the first study, e.g. that there is a gender difference related to efficacy, e.g. that male patients show a higher conversion rate compared to women when treated with tedisamil.
- This second clinical study was a Multi-Center, Double-Blind, Randomized, Placebo-Controlled, Parallel Design Study to evaluate the Efficacy and Safety of Intravenous Tedisamil Sesquifumarate in the Rapid Conversion to Normal Sinus Rhythm in Subjects with Recent Onset Atrial Fibrillation or Flutter.
- the active ingredient used was tedisamil sesqifumarate.
- the study was designed as a phase II study and executed in 30 to 40 centers in 5 countries. Study duration: Screening: up to 48 hours; treatment (in-patient): single 30-minute infusion; safety follow up: 24 hours (in-patient) with continuous telemetry and 28 day safety follow up.
- the primary efficacy objective of the study was to demonstrate the superiority of any dose of tedisamil sesquifumarate to placebo in the rapid conversion to normal sinus rhythm (for at least 60 seconds), as measured by the percentage of subjects converted at any time within 2.5 hours after the start of infusion.
- Secondary efficacy objectives were to determine the percentage of subjects converting to normal sinus rhythm at any time within 2.5 hours after start of the intravenous infusion and in normal sinus rhythm at 2.5 hours after initiation of the infusion of tedisamil sesquifumarate versus placebo; to determine the percentage of subjects converting to normal sinus rhythm at any time within 2.5 hours after start of the intravenous infusion and in normal sinus rhythm at 24 hours after initiation of the infusion of tedisamil sesquifumarate versus placebo; to determine the percentage of subjects converting to normal sinus rhythm at any time within 2.5 hours after start of the intravenous infusion and in normal sinus rhythm at hospital discharge; to determine the time to conversion to normal sinus rhythm after the start of the infusion of tedisamil sesquifumarate versus placebo; to determine the dose- and concentration-response relationships of tedisamil sesquifumarate versus placebo; and to determine the energy required for DC cardioversion of tedisamil sesquifumarate versus placebo.
- Safety objective determining the
- Atrial fibrillation or flutter with a duration of > 3 hours and ⁇ 45 days, occurring as a first or recurrent episode.
- the dosages will be given as an intravenous regimen.
- Tedisamil doses in the protocol refer to tedisamil free base.
- Placebo (vehicle) administered as a 30-minute intravenous infusion similar to tedisamil infusion.
- the total infusion time is 30 minutes, with half the dose infused within 10 minutes, and half the dose infused within the remaining 20 minutes.
- Primary efficacy the percentage of subjects converted to normal sinus rhythm (for at least 60 seconds) at any time within 2.5 hours after the initiation of the infusion of study drug.
- Secondary efficacy percentage of subjects in normal sinus rhythm at any time within 2.5 hours and at 24 hours after start of infusion, as well as at hospital discharge, time to conversion, dose- and concentration-response relationships and DC cardioversion energy.
- Percentages of conversion will be compared among treatment groups using the (Pearson) chi-square statistics. Times to conversion will be compared among treatment groups using the log-rank test. Dose-response and concentration-response relationships and the energy required for DC conversion will be examined using descriptive statistics. Subjects with atrial fibrillation and atrial flutter will be separately analyzed. In addition, both populations will be pooled for analysis. All analyses involving the atrial flutter subject population will be considered as exploratory. Table I: Conversion of recent onset of atrial fibrillation (Afib) to NSR Conversion to NSR at any time within 2.5 hrs after start of infusion. ITT patient sample; patients with DC cardioversion are excluded.
- 3,7-diazabicyclo-[3,3,1]nonane compounds of formula I preferably of 9,9-alkylene-3,7-diazabicyclo[3,3,1]-nonane compounds, and most preferably of tedisamil, and of pharmaceutically acceptable acid addition salts and/or solvates, may be contained together with any conventional and/or newly developed pharmaceutical galenical ingredient suitable for the use in i.v. injections and infusions such like excipient, liquid carrier, auxiliary, and/or adjuvant in the liquid pharmaceutical formulations.
- liquid formulations are such as solutions, suspensions or emulsions, liposomes, solid lipid nano-particles and any other conventional and/or novel drug delivery system suitable for i.v. injection or infusion of in particular 3,7-diaza-bicyclo-[3,3,1]nonane compounds of formula I, preferably of 9,9-alkylene-3,7-diazabicyclo[3,3,1]-nonane compounds, and most preferably of tedisamil, and of pharmaceutically acceptable acid addition salts and/or solvates thereof.
- Infusions are e.g. sterile, aqueous solutions or emulsions with water as the continuous phase; they are usually made isotonic with blood. They are principally intended for administration in large volume.
- the liquid formulations may contain usual diluents such as water, oils and/or lipids, organic solvent or co-solvents and, suspension agents.
- auxiliaries suitable for the use in i.v. injections and in particular for infusions, may additionally be added, such as preservatives, stabilizers, anti-oxidants, pH adjustments, buffers, adjuvants for adjustment of e.g. tonicity or adjustment of viscosity, antibacterial and/or antifungal agents, e.g. parabene, chlorobutanol, phenol, and sorbic acid.
- the liquid pharmaceutical formulations are suitable for infusion regimens, e.g. for intravenous infusion.
- the ingredients, techniques and the procedures required to manufacture liquid pharmaceutical formulations of an active agent are well known to the skilled person.
- the 3,7-diazabicyclo-[3,3,1]nonane compounds of formula I preferably of 9,9-alkylene-3,7-diazabicyclo[3,3,1]-nonane compounds, and most preferably tedisamil, and pharmaceutically acceptable acid addition salts and/or solvates thereof, can be mixed and formulated with the pharmaceutical excipients, auxiliaries liquid carriers and/or adjuvants in known manner.
- the ingredients like the active compound and the required liquid pharmaceutical excipients or carriers and the desired auxiliaries and adjuvants may be mixed together, optionally lyophilized, and e.g. a resulting solution, emulsion, suspension or liposomes may be filled into suitable primary packing materials, e.g.
- infusion solutions must be prepared with enhanced care; in particular the solution must be isotonic or at least approximately isotonic.
- the adjustment of pH is usually arranged by addition of hydro-chloric acid or sodium hydroxide.
- Infusions should be isotonic or approximately isotonic, e.g. iso-osmotic or approximately iso-osmotic.
- sodium chloride glucose or mannitol may be added to arrange for iso-osmotic conditions.
- Stabilisation and or sterilisation may be suitable.
- measures are taken to ensure a suitable and controlled particle size with regard to the intended use.
- the volume of the infusion in the container is sufficient to permit the withdrawal and administration of the nominal dose using a normal technique.
- Suitable methods for the preparation of infusions, the ingredients and conditions to be met are well known to the skilled artisan, e.g. from monographs and/or pharmacopoeias such as the European pharmacopoeia or the US pharmacopoeia.
- the liquid pharmaceutical is suited for administration by infusion, in particular by intravenous infusion.
- infusions may be prepared from concentrates for injections or infusions. Concentrates for injections or infusions are sterile solutions intended for injection or infusion after dilution. They are diluted to a prescribed volume with a prescribed liquid before administration. After dilution, they comply with the requirements for injections or for infusions. Infusions may be also prepared from solid powders for injections or infusions which are solved in the required amount of a suitable sterile solvent just before the administration to the patient.
- Powders for injections or infusions are solid, sterile substances distributed in their final containers and which, when shaken with the prescribed volume of a prescribed sterile liquid, rapidly form either clear and practically particle-free solutions or uniform suspensions. After dissolution or suspension, they comply with the requirements for injections or for infusions. Freeze-dried products for parenteral use usually are considered as powders for injections or infusions.
- Intravenous administration allows rapid administration of larger volumes.
- the advantage of infusion is e.g. rapid onset of the pharmaceutically effect of the active agent, in particular in emergency situations.
- the infusion may be administered as a rapid infusion during the first phase of the administration, and then followed by drop-wise intravenous infusion by which over the period of administration constant blood levels of the drug may be achieved and/or maintained.
- Administration may be also by dual chamber systems, e.g. to avoid drug instability prior administration.
- Example 2 describes a liquid pharmaceutical formulation which contains an active substance of Formula I according to the invention, and also the production of such pharmaceutical preparations.
- the following example explains the production of a liquid pharmaceutical preparation containing tedisamil dihydrochloride. Liquid pharmaceutical preparations containing tedisamil sesquifumarate may be obtained in an analogous manner.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Medicinal Preparation (AREA)
Claims (10)
- Verwendung wenigstens einer 3,7-Diazabicyclo[3.3.1]nonan Verbindung der Formel I,
- Verwendung einer flüssigen pharmazeutischen Formulierung gemäß Anspruch 1, worin R1 eine Alkylgruppe enthaltend 1 bis 6 Kohlenstoffatome oder eine Cycloalkylalkylgruppe enthaltend 4 bis 7 Kohlenstoffatome bedeutet.
- Verwendung einer flüssigen pharmazeutischen Formulierung gemäß einem der vorhergehenden Ansprüche, worin R4 eine Alkylgruppe enthaltend 1 bis 6 Kohlenstoffatome, eine Cycloalkylalkylgruppe enthaltend 4 bis 7 Kohlenstoffatome, oder eine Gruppe entsprechend der Formel b bedeutet.
- Verwendung einer flüssigen pharmazeutischen Formulierung gemäß einem der vorhergehenden Ansprüche, worin R1 eine Alkylgruppe enthaltend 3 bis 6 Kohlenstoffatome oder eine Cycloalkylalkylgruppe enthaltend 4 bis 7 Kohlenstoffatome, und R4 eine Alkylgruppe enthaltend 3 bis 6 Kohlenstoffatome oder eine Cycloalkylalkylgruppe enthaltend 4 bis 7 Kohlenstoffatome, bedeutet.
- Verwendung einer flüssigen pharmazeutischen Formulierung gemäß einem der vorhergehenden Ansprüche, worin besagte 3,7-Diazabicyclo[3.3.1]nonan Verbindung eine 9,9-Alkylene-3,7-diazabicyclo[3.3.1]nonan Verbindung der Formel I ist, worin R2 und R3 zusammen eine Alkylenkette enthaltend 4 bis 5 Kohlenstoffatome bilden, und R1 und R4 unabhängig voneinander jeweils eine geradkettige oder verzweigte Alkylgruppe mit 3 - 4 Kohlenstoffatome oder die Cyclopropylmethylgruppe, und physiologisch akzeptable Säureadditionssalze und/oder Solvate davon bezeichnen.
- Verwendung einer flüssigen pharmazeutischen Formulierung gemäß Anspruch 5, worin besagte 9,9-Alkylene-3,7-diazabicyclo[3.3.1]nonan Verbindung der Formel I ein Fumarsäuresalz der besagten 9,9-Alkylene-3,7-diazabicyclo[3.3.1]nonan Verbindung der Formel I wie in Anspruch 5 definiert ist, enthaltend 1.5 Mol Fumarsäure pro Mol der besagten 9,9-Alkylene-3,7-diazabicyclo[3.3.1]-nonan Verbindung der Formel I.
- Verwendung einer flüssigen pharmazeutischen Formulierung gemäß einem der Ansprüche 1 bis 6, worin besagte 3,7-Diazabicyclo[3.3.1]nonan Verbindung ausgewählt ist aus der Gruppe bestehend aus N,N'-Dicyclopropylmethyl-9,9-tetramethylen-3,7-diazabicyclo[3.3.1]nonan, N-Isobutyl-N'-isopropyl-9,9-pentamethylen-3,7-diazabicyclo[3.3.1]nonan, und physiologisch akzeptable Säureadditionssalze und/oder Solvate davon.
- Verwendung einer flüssigen pharmazeutischen Formulierung gemäß Anspruch 7, worin besagte 3,7-Diazabicyclo[3.3.1]nonan Verbindung ein Fumarsäuresalz von N,N'-Dicyclopropylmethyl-9,9-tetramethylen-3,7-diazabicyclo[3.3.1]nonan oder von N-Isobutyl-N'-isopropyl-9,9-pentamethylen-3,7-diazabicyclo[3.3.1]nonan ist, enthaltend 1.5 Mol Fumarsäure pro Mol der besagten 9,9-Alkylene-3,7-diazabicyclo[3.3.1]-nonan Verbindung.
- Verwendung einer flüssigen pharmazeutischen Formulierung gemäß einem der Ansprüche 1 to 5, und 8, worin besagte 3,7-Diazabicyclo[3.3.1]nonan Verbindung ein Hydrochloridsalz ist.
- Wenigstens eine der 3,7-Diazabicyclo[3.3.1]nonan Verbindungen der Formel I,
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US42679502P | 2002-11-18 | 2002-11-18 | |
| US426795P | 2002-11-18 | ||
| PCT/EP2003/012844 WO2004045591A2 (en) | 2002-11-18 | 2003-11-17 | Liquid pharmaceutical formulations containing 3,7-diazabicyclo`3,3,1! nonanes for treating anti-arrhythmic events |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1565239A2 EP1565239A2 (de) | 2005-08-24 |
| EP1565239B1 true EP1565239B1 (de) | 2011-03-16 |
| EP1565239B8 EP1565239B8 (de) | 2011-06-22 |
Family
ID=32326429
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP03811379A Expired - Lifetime EP1565239B8 (de) | 2002-11-18 | 2003-11-17 | Flüssige zusammensetzungen mit 3,7-diazabicyclo[3,3,1] nonanen zur behandlung von anti-arrhythmischen ereignissen |
Country Status (20)
| Country | Link |
|---|---|
| US (2) | US7087620B2 (de) |
| EP (1) | EP1565239B8 (de) |
| JP (1) | JP2006510613A (de) |
| KR (1) | KR101059330B1 (de) |
| CN (1) | CN1726062A (de) |
| AR (1) | AR042199A1 (de) |
| AT (1) | ATE501758T1 (de) |
| AU (1) | AU2003296580B8 (de) |
| BR (1) | BR0316383A (de) |
| CA (1) | CA2506083A1 (de) |
| DE (1) | DE60336417D1 (de) |
| IL (1) | IL168630A (de) |
| MX (1) | MXPA05005344A (de) |
| NO (1) | NO20053010L (de) |
| RU (1) | RU2358732C2 (de) |
| SA (1) | SA04250023B1 (de) |
| TW (1) | TW200418468A (de) |
| UA (1) | UA85546C2 (de) |
| WO (1) | WO2004045591A2 (de) |
| ZA (1) | ZA200504049B (de) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW200418468A (en) * | 2002-11-18 | 2004-10-01 | Solvay Pharm Gmbh | Liquid pharmaceutical formulations containing 3,7-diazabicyclo[3,3,1]nonane compounds and method of treatments relating to anti-arrhytmic events |
| US20050054667A1 (en) * | 2003-07-21 | 2005-03-10 | Solvay Pharmaceuticals Gmbh | Method of treating or inhibiting anti-arrhythmic events in male human patients |
| CA2533492A1 (en) * | 2003-07-21 | 2005-02-10 | Solvay Pharmaceuticals Gmbh | Use of 3,7-diazabicyclo[3,3,1]nonane compounds for the treatment and/or prophylaxis of anti-arrhythmic events in male human patients |
| EP1917979A4 (de) * | 2005-08-23 | 2009-06-03 | Astellas Pharma Inc | Therapeutikum für vorhofflimmern |
| EP2149370A1 (de) * | 2008-07-31 | 2010-02-03 | Neuroscienze Pharmaness S.C. A R.L. | Diazabizyclischen Verbindungen und Mikroemulsionen, dieselben enthaltend |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3361244D1 (de) * | 1982-01-29 | 1986-01-02 | Ici Plc | Alkanolamine derivatives |
| DE3234697A1 (de) * | 1982-09-18 | 1984-03-22 | Kali-Chemie Pharma Gmbh, 3000 Hannover | Neue diazabicyclo-(3,3,1)-nonane |
| PT88381B (pt) * | 1987-09-09 | 1995-07-06 | Kali Chemie Pharma Gmbh | Processo para a preparacao de novos compostos 3,7-diazabiciclo{3,3,1} nonano, e de composicoes farmaceuticas que contem estes compostos |
| DE4019080A1 (de) * | 1990-06-15 | 1991-12-19 | Kali Chemie Pharma Gmbh | Neue 3,7-diazabicycolo (3,3,1)nonan-verbindungen enthaltende arzneimittel |
| DE4139763A1 (de) * | 1991-12-03 | 1993-06-09 | Kali-Chemie Pharma Gmbh, 3000 Hannover, De | Kristalline fumarsaure salze von 9,9-alkylen-3,7-diazabicyclononan-verbindungen und diese verbindungen enthaltende arzneimittel |
| DE4402933A1 (de) * | 1994-02-01 | 1995-08-03 | Kali Chemie Pharma Gmbh | Neue 3-Benzoyl-3,7-diazabicyclo[3,3,1] nonan-Verbindungen enthaltende Arzneimittel |
| DE4402931A1 (de) * | 1994-02-01 | 1995-08-03 | Kali Chemie Pharma Gmbh | Neue 3-Phenylsulfonyl-3,7-diazabicyclo[3,3,1] nonan-Verbindungen enthaltende Arzneimittel |
| US5576349A (en) * | 1995-11-30 | 1996-11-19 | The General Hospital Corporation | Retionoic acid treatment of cardiac arrhythmia |
| US6026817A (en) * | 1996-11-14 | 2000-02-22 | Clemens; Anton H. | Method of treating the syndrome of coronary heart disease risk factors in humans |
| SE9704709D0 (sv) * | 1997-12-17 | 1997-12-17 | Astra Ab | Pharmaceutically active compounds |
| US6790463B2 (en) * | 2001-03-30 | 2004-09-14 | Robert F. Hofmann | Uses of targeted oxidative therapeutic formulation in arteriosclerosis |
| DE10131217A1 (de) * | 2001-06-28 | 2003-01-09 | Solvay Pharm Gmbh | 3-Phenyl-3,7-diazabicyclo 3,3,1 nonan-Verbindungen sowie Verfahren zu ihrer Herstellung und diese Verbindungen enthaltende Arzneimittel |
| TW200418468A (en) * | 2002-11-18 | 2004-10-01 | Solvay Pharm Gmbh | Liquid pharmaceutical formulations containing 3,7-diazabicyclo[3,3,1]nonane compounds and method of treatments relating to anti-arrhytmic events |
-
2003
- 2003-11-13 TW TW092131744A patent/TW200418468A/zh unknown
- 2003-11-17 BR BR0316383-0A patent/BR0316383A/pt not_active IP Right Cessation
- 2003-11-17 MX MXPA05005344A patent/MXPA05005344A/es unknown
- 2003-11-17 CN CNA2003801061229A patent/CN1726062A/zh active Pending
- 2003-11-17 RU RU2005119280/15A patent/RU2358732C2/ru not_active IP Right Cessation
- 2003-11-17 AT AT03811379T patent/ATE501758T1/de not_active IP Right Cessation
- 2003-11-17 WO PCT/EP2003/012844 patent/WO2004045591A2/en not_active Ceased
- 2003-11-17 UA UAA200505967A patent/UA85546C2/ru unknown
- 2003-11-17 AU AU2003296580A patent/AU2003296580B8/en not_active Ceased
- 2003-11-17 JP JP2004552627A patent/JP2006510613A/ja not_active Ceased
- 2003-11-17 EP EP03811379A patent/EP1565239B8/de not_active Expired - Lifetime
- 2003-11-17 DE DE60336417T patent/DE60336417D1/de not_active Expired - Lifetime
- 2003-11-17 KR KR1020057008973A patent/KR101059330B1/ko not_active Expired - Fee Related
- 2003-11-17 AR ARP030104235A patent/AR042199A1/es not_active Application Discontinuation
- 2003-11-17 CA CA002506083A patent/CA2506083A1/en not_active Abandoned
- 2003-11-18 US US10/715,130 patent/US7087620B2/en not_active Expired - Fee Related
-
2004
- 2004-03-03 SA SA04250023A patent/SA04250023B1/ar unknown
-
2005
- 2005-05-17 IL IL168630A patent/IL168630A/en not_active IP Right Cessation
- 2005-05-19 ZA ZA200504049A patent/ZA200504049B/en unknown
- 2005-06-20 NO NO20053010A patent/NO20053010L/no not_active Application Discontinuation
- 2005-07-13 US US11/179,635 patent/US20050261300A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| MXPA05005344A (es) | 2005-08-03 |
| KR101059330B1 (ko) | 2011-08-24 |
| BR0316383A (pt) | 2005-10-04 |
| DE60336417D1 (de) | 2011-04-28 |
| EP1565239B8 (de) | 2011-06-22 |
| UA85546C2 (en) | 2009-02-10 |
| RU2358732C2 (ru) | 2009-06-20 |
| ATE501758T1 (de) | 2011-04-15 |
| AU2003296580B8 (en) | 2010-03-18 |
| AU2003296580B2 (en) | 2010-02-18 |
| AU2003296580A1 (en) | 2004-06-15 |
| US20040152716A1 (en) | 2004-08-05 |
| AR042199A1 (es) | 2005-06-15 |
| IL168630A (en) | 2013-08-29 |
| ZA200504049B (en) | 2006-07-26 |
| NO20053010L (no) | 2005-06-20 |
| SA04250023B1 (ar) | 2008-12-23 |
| US20050261300A1 (en) | 2005-11-24 |
| RU2005119280A (ru) | 2006-03-20 |
| CN1726062A (zh) | 2006-01-25 |
| KR20050116125A (ko) | 2005-12-09 |
| US7087620B2 (en) | 2006-08-08 |
| WO2004045591A3 (en) | 2004-07-22 |
| CA2506083A1 (en) | 2004-06-03 |
| TW200418468A (en) | 2004-10-01 |
| EP1565239A2 (de) | 2005-08-24 |
| WO2004045591A2 (en) | 2004-06-03 |
| JP2006510613A (ja) | 2006-03-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3661514B1 (de) | Mavacamten zur verwendung bei der behandlung der hypertrophen kardiomyopathie | |
| CZ292002B6 (cs) | Léčivo pro snížení úmrtnosti způsobené městnavým selháním srdce u savců | |
| EP2153830A1 (de) | Verwendung von Dronedaron zur Herstellung eines Medikaments zur Prävention von Schlaganfällen oder transienten ischämischen Anfällen | |
| US20260041679A1 (en) | Method of treating hypertension with the aldosterone synthase inhibitor baxdrostat | |
| EP1565239B1 (de) | Flüssige zusammensetzungen mit 3,7-diazabicyclo[3,3,1] nonanen zur behandlung von anti-arrhythmischen ereignissen | |
| Kojima et al. | First case report of anamorelin-induced fatal arrhythmia complicated by sinus arrest and refractory ventricular tachycardia | |
| JP7620758B2 (ja) | 肺動脈性肺高血圧症の治療方法 | |
| US20160303113A1 (en) | Methods of self-administration of vanoxerine for terminating acute episodes of cardiac arrhythmia in mammals | |
| JP2018135278A (ja) | 循環器疾患及び/又はミトコンドリア病の改善用医薬 | |
| WO2005011690A1 (en) | Use of 3,7-diazabicyclo`3, 3,1 !nonane compounds for the treatment and/or prophylaxis of anti-arrhythmic events in male human patients | |
| US20050054667A1 (en) | Method of treating or inhibiting anti-arrhythmic events in male human patients | |
| JP3947582B2 (ja) | 不安神経症治療剤 | |
| EP0813878A1 (de) | Mittel zur Beschleunigung des Tränenflusses enthaltend ein Serotonin-Liganden, insbesondere Aminoalkoxybibenzyle | |
| US20160051542A1 (en) | Methods of administration of single doses of vanoxerine to terminate acute episodes of cardiac arrhythmia | |
| KR20250110354A (ko) | 심장 박동에 유의미한 영향을 미치지 않는 쿠싱 증후군에 대한 치료 | |
| HK40125990A (zh) | 对心律无显着影响的库欣综合征的治疗 | |
| CN119522109A (zh) | 用于钙调磷酸酶抑制剂的间歇性给药的组合物 | |
| US20240058350A1 (en) | Methods of Treatment | |
| US20160304477A1 (en) | Novel metabolites of vanoxerine compounds, pharmaceutical compositions containing the same and methods of terminating acute episodes of cardiac arrhythmia, restoring normal sinus rhythm, preventing recurrence of cardiac arrhythmia and maintaining normal sinus rhythm in mammals | |
| HK40026724B (en) | Mavacamten for use in the treatment of hypertrophic cardiomyopathy | |
| HK40026724A (en) | Mavacamten for use in the treatment of hypertrophic cardiomyopathy | |
| MXPA97006042A (en) | Use of carbazol compounds for the treatment of cardiac congest insufficiency |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20050620 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK |
|
| DAX | Request for extension of the european patent (deleted) | ||
| 17Q | First examination report despatched |
Effective date: 20051216 |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: JANSEN, JOHANNES, WILHELMUS, CATHARINE, MARIA Inventor name: CAUTREELS, WERNER Inventor name: THE OTHER INVENTORS HAVE AGREED TO WAIVE THEIR ENT Inventor name: DE VRIES, MICHIEL, HENRICUS |
|
| RTI1 | Title (correction) |
Free format text: LIQUID PHARMACEUTICAL FORMULATIONS CONTAINING 3,7-DIAZABICYCLO(3,3,1) NONANES FOR TREATING ANTI-ARRHYTHMIC EVENTS |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: ABBOTT PRODUCTS GMBH |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 60336417 Country of ref document: DE Date of ref document: 20110428 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 60336417 Country of ref document: DE Effective date: 20110428 |
|
| RIN2 | Information on inventor provided after grant (corrected) |
Inventor name: JANSEN, JOHANNES, WILHELMUS, CATHARINE, MARIA Inventor name: STEINBORN, CLAUS, RUDOLF Inventor name: CAUTREELS, WERNER Inventor name: DE VRIES, MICHIEL, HENRICUS Inventor name: STRAUB, MATTHIAS |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20110316 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110627 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110616 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: ISLER & PEDRAZZINI AG Ref country code: RO Ref legal event code: EPE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110718 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: T3 Ref document number: E 10194 Country of ref document: SK |
|
| REG | Reference to a national code |
Ref country code: HU Ref legal event code: AG4A Ref document number: E011896 Country of ref document: HU |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20111219 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 60336417 Country of ref document: DE Effective date: 20111219 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: LU Payment date: 20121113 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: HU Payment date: 20121026 Year of fee payment: 10 Ref country code: FR Payment date: 20121113 Year of fee payment: 10 Ref country code: IE Payment date: 20121030 Year of fee payment: 10 Ref country code: CH Payment date: 20121026 Year of fee payment: 10 Ref country code: DE Payment date: 20121130 Year of fee payment: 10 Ref country code: MC Payment date: 20121029 Year of fee payment: 10 Ref country code: CZ Payment date: 20121109 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20121025 Year of fee payment: 10 Ref country code: SK Payment date: 20121025 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20131117 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131130 Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131202 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131130 |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: MM4A Ref document number: E 10194 Country of ref document: SK Effective date: 20131117 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20140731 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131117 Ref country code: RO Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131117 Ref country code: CZ Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131117 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110316 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140603 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 60336417 Country of ref document: DE Effective date: 20140603 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131117 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131118 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131117 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131202 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131117 |