EP1273671B1 - Dezincification resistant copper-zinc alloy and method for producing the same - Google Patents
Dezincification resistant copper-zinc alloy and method for producing the same Download PDFInfo
- Publication number
- EP1273671B1 EP1273671B1 EP20020015027 EP02015027A EP1273671B1 EP 1273671 B1 EP1273671 B1 EP 1273671B1 EP 20020015027 EP20020015027 EP 20020015027 EP 02015027 A EP02015027 A EP 02015027A EP 1273671 B1 EP1273671 B1 EP 1273671B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- rest
- weight
- alloy
- copper
- dezincification
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22F—CHANGING THE PHYSICAL STRUCTURE OF NON-FERROUS METALS AND NON-FERROUS ALLOYS
- C22F1/00—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working
- C22F1/08—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working of copper or alloys based thereon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C9/00—Alloys based on copper
- C22C9/04—Alloys based on copper with zinc as the next major constituent
Definitions
- the invention relates to a dezincification-resistant copper-zinc alloy, which is particularly suitable for sanitary applications, e.g. suitable for drinking water fittings, drinking water pipes or pipe binders.
- the invention further relates to a manufacturing method for such a copper-zinc alloy.
- Common copper-zinc alloys can occur in two different metallic phases.
- the brass alloy has an fcc (Face-Centered-Cubic) structure, while in the ⁇ -phase it has a Bcc (Body-Centered-Cubic) structure.
- the pure ⁇ -phase occurs at a copper content of more than 62% by weight.
- the brass alloy is present with portions of both the ⁇ and the ⁇ phase. In the presence of additional metallic components, the above-mentioned phase boundaries shift.
- a brass alloy For sanitary applications, a brass alloy must have high corrosion resistance due to the constant water contact. Brass, and also copper, forms cover layers of copper oxide in aqueous, slightly acidic to alkaline electrolyte solutions, which generally provide some protection against general corrosion. In soft, chloride-containing waters, brass alloys may experience a special form of corrosion called dezincification. The zinc is dissolved out of the brass alloy and a porous copper sponge remains behind. Furthermore, the ⁇ -phase is attacked more as the ⁇ -phase, since the former is less electropositive to the latter. To improve the corrosion resistance of a brass alloy, it is therefore known to prevent the formation of a ⁇ -phase as much as possible by means of a heat treatment during production.
- the addition of a single further metallic component can drastically change other characteristics of the brass alloy.
- the additive arsenic although the corrosion protection for the ⁇ -phase, but deteriorates in the presence of ⁇ and ⁇ phase, the corrosion resistance to sulphate waters. Since the composition of drinking water, ie, the type and concentration of ions dissolved therein, is highly dependent on its geographical origin, lengthy research is needed to assemble a brass alloy that will not corrode even after decades of use and contact with water.
- As dezincification and corrosion resistant brass alloy is from the WO 89/08725 a copper-zinc alloy containing up to 0.8% by weight of nickel, between 0.5 and 3% by weight of lead, between 0.3 and 1% by weight of silicon and between 0.07 and 0 , 8 wt .-% iron.
- Arsenic is considered to be in principle disturbing because it promotes intergranular corrosion, which is why its content should be less than 0.02% by weight.
- the object of the invention is to provide a copper-zinc alloy which has a particularly high resistance to dezincification and intercrystalline corrosion, and is particularly suitable for sanitary applications. It is another object of the invention to provide a manufacturing method for such a copper-zinc alloy.
- the first-mentioned object is achieved for a copper-zinc alloy according to the invention in that it contains 50 to 80% by weight of copper, 0 to 5% by weight of lead, 0.01 to 0.1% by weight of arsenic, 0, 03 to less than 0.3% by weight of silicon, 0 to 0.3% by weight of iron, 0 to 0.04% by weight of manganese, the balance being zinc and unavoidable impurities, and that the effective copper Equivalent to 60 to 70 wt .-% is.
- a copper-zinc alloy with the said composition both has a high dezincification resistance and also a very low intergranular corrosion, referred to below as IK shows.
- the invention is based on the recognition that a certain content of arsenic is crucial for the avoidance of intercrystalline corrosion. Furthermore, the invention assumes that iron is not required for the corrosion resistance of the brass alloy, but has a disturbing effect. However, the disturbing influence of iron can be avoided to a limited extent by the addition of silicon.
- the object of the production method for a copper-zinc alloy wherein the starting materials are mixed, melted and cast according to the desired composition into a metallic alloy, and wherein the metallic alloy for forming the ⁇ -phase at 500-650 Is annealed ° C, is inventively achieved in that the annealed metallic alloy is actively cooled by means of coolants.
- Table 1 lists the composition of these 44 experimental alloys in terms of copper, lead, arsenic, iron, silicon, manganese, aluminum and zinc.
- the desired and the actual proportion in wt .-% is reproduced.
- the trial alloys are each designated with four-digit numbers. This term is used below for the respective alloy.
- the experimental alloys listed in Table 1 were prepared as follows: 1. Cast the molten alloy at a temperature of 1050 to 1100 ° C in a steel mold of the dimensions 110 mm x 40 mm 2. Milling the sample to a thickness of 14 mm 3. Rolling the sample to 5 mm, with intermediate annealing for 1 hour at temperatures of about 700 ° C. 4. ⁇ -anneal for 2 hours at a temperature of 550 degrees Celsius Table 1: ⁇ b> Nominal and actual compositions of the experimental alloys in% by weight ⁇ / b> Leg.-Nr.
- a dezincification test was performed according to ISO 6509.
- the degree of dezincification of the respective alloy becomes corroding after a predefined test procedure Liquids determined by measuring a maximum Entzinkungstiefe in microns.
- the maximum depth of dezincification determined in this way is a measure of the susceptibility of the test alloy to be dezincified. The greater the maximum dezincification depth of the particular experimental alloy, the greater its susceptibility to dezincification.
- the dezincification resistance test according to ISO 6509 uses a very aggressive electrolyte solution for dezincification, which is not suitable for detecting IK due to very fine potential differences between grain boundaries and grain area.
- IK susceptibility compared to CuZn36Pb2As in% Maximum dezincification depth to ISO 6509 in ⁇ m 2088 19 13 2089 46 12 2090 100 15 2094 30 6 2102 8th 0 2106 24 12 2131 91 0 2132 25 0 2133 10 0 2134 10 0 2135 65 0 2136 27 0 2156 5 212 2157 7 18 2158 7 0 2159 100 0 2160 100 0 2161 17 0 2162 13 0 2163 10 0 2164 8th 0 2165 23 0 2166 15 0 2167 18 0 2168 18 0 2169 17 0 2170 48 0 2171 100 0 2172 10 0 2173 11 0 2174 10 0 2175 88 50 2176 96 46 2177 100 75 2178 100 45 2179 83 60 2180 42 80 2181 42 95 2182 33 40 2183 58 34 2184 42 38 2185 46 54 2186 21 62 2187 33 108
- FIG. 1 the susceptibility to intergranular corrosion in% is shown in column 3 of Table 2 for test alloys Nos. 2088, 2089, 2090, 2131 and 2132. These alloys differ in otherwise substantially the same composition in their arsenic content. The respective arsenic content in wt .-% is on the abscissa in FIG. 1 applied.
- the illustrated experimental alloys have only a negligible or no share of iron, silicon, manganese and aluminum.
- FIG. 1 a necessary for arsenic content between 0.01 and 0.1 wt .-% for a dezincification and resistant to IK brass alloy. Above a proportion of arsenic of 0.1 wt .-% protection against IK is no longer guaranteed. If the arsenic content is reduced below 0.08 wt .-%, the susceptibility to intergranular corrosion is only less than 60%.
- the experimental alloys Nos. 2180 and 2181 have a value of 0.05% by weight with respect to their arsenic content which is not critical with respect to the IC. In addition, however, these trial alloys show a small amount of iron of 0.03 wt% and 0.06 wt%, respectively.
- a comparison with the non-ferrous experimental alloys Nos. 2088 and 2089 shows that the addition of iron obviously lowers the critical upper limit for the arsenic content.
- susceptibility to intergranular corrosion increases.
- the susceptibility to dezincification increases with the addition of iron.
- the trial alloy No. 2159 having an iron content of 0.082% by weight is used, its susceptibility to intergranular corrosion becomes clear.
- the trial alloy No. 2159 shows a susceptibility to IK of 100% according to Table 2, whereas the trial alloys No. 2088 and No. 2089 show a susceptibility of only 19% and 46%, respectively.
- alloys Nos. 2094, 2102 and 2106. These alloys have iron contents at an arsenic content of 0.07 wt% of 0.14 wt .-%, 0.1 wt .-% and 0.2 wt .-% to.
- these experimental alloys contain silicon in a proportion of 0.08% by weight and 0.2% by weight.
- a look at Table 2 shows that silicon can neutralize the damaging effects of iron. Susceptibility to intergranular corrosion decreases.
- these experimental alloys are generally dezincification.
- alloys Nos. 2135 and 2136 which have a silicon content of about 2% by weight, that they again become susceptible to intergranular corrosion.
- FIG. 2 the susceptibility to intergranular corrosion is shown according to the results of Table 2 for trial alloys Nos. 2168 to 2171. These alloys differ in their composition in terms of their manganese content, which increases from the test alloy No. 2168 to the test alloy 2171 of 0.006 wt .-% to 0.055 wt .-%. It turns out that manganese is a very critical element in intercrystalline corrosion, and can not be neutralized by a proportion of silicon. All trial alloys Nos. 2168 to 2171 have a silicon content of about 0.2% by weight.
- the permissible tolerances for manganese must therefore remain low. At a level of 0.04 wt%, the susceptibility to intergranular corrosion is less than 50%.
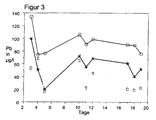
- FIG. 3 is for a trial alloy containing 64.05% copper, 2.7% lead, 0.1% iron, 0.3% aluminum, 0.2% silicon and 0.05% arsenic and the remainder zinc (* symbol) , a CuZn36Pb2.7As0.14 alloy (O symbol) and a CuZn36Pb2.7 alloy ( ⁇ symbol), the lead release to drinking water based on NSF Standard 61, 1994 shown.
- the lead levy is shown in ⁇ g per liter over time.
Description
Die Erfindung betrifft eine entzinkungsbeständige Kupfer-Zink-Legierung, die sich insbesondere für Anwendungen im Sanitärbereich, z.B. für Trinkwasserarmaturen, Trinkwasserrohre oder Rohrbinder eignet. Die Erfindung betrifft weiter ein Herstellungsverfahren für eine derartige Kupfer-Zink-Legierung.The invention relates to a dezincification-resistant copper-zinc alloy, which is particularly suitable for sanitary applications, e.g. suitable for drinking water fittings, drinking water pipes or pipe binders. The invention further relates to a manufacturing method for such a copper-zinc alloy.
Gebräuchliche Kupfer-Zink-Legierungen, auch Messing oder Messinglegierungen genannt, können in zwei verschiedenen metallischen Phasen auftreten. In der sogenannten α-Phase weist die Messinglegierung eine fcc (Face - Centered - Cubic) Struktur auf, während sie in der β-Phase eine Bcc (Body - Centered - Cubic) Struktur besitzt. Im Phasendiagramm einer Messinglegierung tritt die reine α-Phase bei einem Kupferanteil von mehr als 62 Gew.-% auf. Bei einem Anteil von Kupfer zwischen 54 und 62 Gew.-% liegt die Messinglegierung mit Anteilen sowohl der α- als auch der β-Phase vor. Bei Vorhandensein von zusätzlichen metallischen Komponenten verschieben sich die oben genannten Phasengrenzen.Common copper-zinc alloys, also called brass or brass alloys, can occur in two different metallic phases. In the so-called α-phase, the brass alloy has an fcc (Face-Centered-Cubic) structure, while in the β-phase it has a Bcc (Body-Centered-Cubic) structure. In the phase diagram of a brass alloy, the pure α-phase occurs at a copper content of more than 62% by weight. With a proportion of copper between 54 and 62 wt .-%, the brass alloy is present with portions of both the α and the β phase. In the presence of additional metallic components, the above-mentioned phase boundaries shift.
Für Sanitäranwendungen muss eine Messinglegierung aufgrund des ständigen Wasserkontakts eine hohe Korrosionsbeständigkeit aufweisen. Messing, und auch Kupfer, bildet in wässrigen, schwach sauren bis alkalischen Elektrolytlösungen Deckschichten aus Kupferoxid, die in der Regel einen gewissen Schutz gegen allgemeine Korrosion bieten. In weichen, chloridhaltigen Wässern kann bei Messinglegierungen eine besondere Korrosionsform auftreten, die als Entzinkung bezeichnet wird. Dabei wird das Zink aus der Messinglegierung herausgelöst, und es bleibt ein poröser Kupferschwamm zurück. Weiter wird die β-Phase stärker angegriffen als die α-Phase, da Erstere gegenüber der Letzteren weniger elektropositiv ist.
Zur Verbesserung der Korrosionsbeständigkeit einer Messinglegierung ist es daher bekannt, durch eine Wärmebehandlung bei der Herstellung die Ausbildung einer β-Phase möglichst zu verhindern. Weiter ist es auch bekannt, einer Messinglegierung zusätzliche metallische Komponenten beizumengen, die die α-Phase vor Entzinkung schützen. Als derartige metallische Komponenten sind aus der
To improve the corrosion resistance of a brass alloy, it is therefore known to prevent the formation of a β-phase as much as possible by means of a heat treatment during production. Furthermore, it is also known to add to a brass alloy additional metallic components which protect the α-phase from dezincification. As such metallic components are from the
Das Hinzufügen einer einzelnen weiteren metallischen Komponente kann jedoch andere charakteristische Merkmale der Messinglegierung drastisch verändern. So verbessert gemäß der
Als entzinkungsarme und korrosionsbeständige Messinglegierung ist aus der
Aufgabe der Erfindung ist es, eine Kupfer-Zink-Legierung anzugeben, die eine besonders hohe Beständigkeit gegenüber einer Entzinkung und einer interkristallinen Korrosion aufweist, und sich insbesondere für Anwendungen im Sanitärbereich eignet. Weiter ist es Aufgabe der Erfindung, ein Herstellungsverfahren für eine derartige Kupfer-Zink-Legierung anzugeben.The object of the invention is to provide a copper-zinc alloy which has a particularly high resistance to dezincification and intercrystalline corrosion, and is particularly suitable for sanitary applications. It is another object of the invention to provide a manufacturing method for such a copper-zinc alloy.
Die erstgenannte Aufgabe wird für eine Kupfer-Zink-Legierung erfindungsgemäß dadurch gelöst, dass diese 50 bis 80 Gew.-% Kupfer, 0 bis 5 Gew.-% Blei, 0,01 bis 0,1 Gew.-% Arsen, 0,03 bis weniger als 0,3 Gew.-% Silizium, 0 bis 0,3 Gew.-% Eisen, 0 bis 0,04 Gew.-% Mangan, als Rest Zink sowie nicht vermeidbare Verunreinigungen umfasst, und dass das wirksame Kupfer-Äquivalent 60 bis 70 Gew.-% beträgt.The first-mentioned object is achieved for a copper-zinc alloy according to the invention in that it contains 50 to 80% by weight of copper, 0 to 5% by weight of lead, 0.01 to 0.1% by weight of arsenic, 0, 03 to less than 0.3% by weight of silicon, 0 to 0.3% by weight of iron, 0 to 0.04% by weight of manganese, the balance being zinc and unavoidable impurities, and that the effective copper Equivalent to 60 to 70 wt .-% is.
Mit dem Kupfer-Äquivalent oder dem wirksamen Kupfergehalt berücksichtigt man die Wirkungen der unterschiedlichen Legierungselemente bei Messinglegierungen bezüglich der Gefügeausbildung. Ist beispielsweise Blei oder Eisen in der Kupfer-Zink-Legierung enthalten, so addieren sich deren Anteile hinsichtlich der Wirksamkeit zu dem tatsächlichen Kupfer-Anteil. In diesem Fall ist der tatsächliche Kupfer-Anteil niedriger als das wirksame Kupfer-Äquivalent. Beträgt das wirksame Kupfer-Äquivalent etwa 60 Gew.-% und ist zusätzlich ein Blei-Anteil von 2 Gew.-% enthalten, so beträgt der tatsächliche Kupfer-Anteil (1-0,02) x 60 Gew.-% = 58,8 Gew.-%.With the copper equivalent or the effective copper content, one considers the effects of the different alloying elements in brass alloys on the microstructure formation. If, for example, lead or iron are contained in the copper-zinc alloy, then their shares add up in terms of effectiveness to the actual copper content. In this case, the actual copper content is lower than the effective copper equivalent. If the effective copper equivalent is about 60% by weight and additionally contains a lead content of 2% by weight, the actual copper content is (1-0.02) × 60% by weight = 58, 8% by weight.
Umfangreiche Untersuchungen haben gezeigt, dass eine Kupfer-Zink-Legierung mit der genannten Zusammensetzung sowohl eine hohe Entzinkungs-Beständigkeit aufweist als auch eine sehr geringe interkristalline Korrosion, im folgenden IK genannt, zeigt. Die Erfindung geht dabei von der Erkenntnis aus, dass ein bestimmter Gehalt an Arsen entscheidend für das Vermeiden der interkristallinen Korrosion ist. Weiter geht die Erfindung davon aus, dass Eisen für die Korrosionsbeständigkeit der Messinglegierung nicht erforderlich ist, sondern störend wirkt. Der störende Einfluss von Eisen läßt sich jedoch in begrenztem Umfang durch die Zugabe von Silizium vermeiden.Extensive studies have shown that a copper-zinc alloy with the said composition both has a high dezincification resistance and also a very low intergranular corrosion, referred to below as IK shows. The invention is based on the recognition that a certain content of arsenic is crucial for the avoidance of intercrystalline corrosion. Furthermore, the invention assumes that iron is not required for the corrosion resistance of the brass alloy, but has a disturbing effect. However, the disturbing influence of iron can be avoided to a limited extent by the addition of silicon.
Weitere vorteilhafte Ausgestaltungen finden sich in den Unteransprüchen 2-5.Further advantageous embodiments can be found in the dependent claims 2-5.
Die Aufgabe hinsichtlich des Herstellungsverfahrens für eine Kupfer-Zink-Legierung, wobei die Ausgangsmaterialien gemäß der gewünschten Zusammensetzung gemischt, geschmolzen und zu einer metallischen Legierung gegossen werden, und wobei die metallische Legierung zur Ausbildung der α-Phase bei 500 - 650 °C geglüht wird, wird erfindungsgemäß dadurch gelöst, dass die geglühte metallische Legierung mittels Kühlmitteln aktiv abgekühlt wird.The object of the production method for a copper-zinc alloy, wherein the starting materials are mixed, melted and cast according to the desired composition into a metallic alloy, and wherein the metallic alloy for forming the α-phase at 500-650 Is annealed ° C, is inventively achieved in that the annealed metallic alloy is actively cooled by means of coolants.
Umfangreiche Untersuchungen haben ergeben, dass sich die Beständigkeit der Legierung gemäß den Merkmalen von Patentanspruch 1 gegen interkristalline Korrosion weiter verbessern lässt, wenn die geglühte Legierung mittels Kühlmitteln aktiv abgekühlt wird. Ein solches aktives Abkühlen zur Beschleunigung des natürlichen Abkühlens, wobei keine zusätzlichen Hilfsmittel eingesetzt werden, kann beispielsweise mittels eines Kühlgebläses geschehen oder aber es kann durch Abschrecken rasch abgekühlt werden. Ein derartiges Abschrecken kann beispielsweise mittels eines Wasserbads erfolgen. Bei Verwendung eines Kühlgebläses kann die Kühlbehandlung als Durchlaufkühlung für größere Stückzahlen oder Mengen ausgebildet werden.Extensive studies have shown that the resistance of the alloy according to the features of claim 1 against intergranular corrosion can be further improved if the annealed alloy is actively cooled by means of coolants. Such an active cooling to accelerate the natural cooling, with no additional aids are used, for example, can be done by means of a cooling fan or it can be cooled rapidly by quenching. Such quenching can be done for example by means of a water bath. When using a cooling fan, the cooling treatment can be designed as a continuous cooling for larger quantities or quantities.
Wird eine derart hergestellte Kupfer-Zink-Legierung durch Kaltverformen weiterbehandelt, so hat es sich gezeigt, dass hierdurch die Beständigkeit gegen eine interkristalline Korrosion abnimmt. Dies lässt sich vorteilhafterweise vermeiden, wenn die kaltverformte Legierung für 10 min bis 2 Stunden bei einer Temperatur zwischen 250 und 600 °C einer Entspannungsglühung unterzogen wird.If such a copper-zinc alloy thus produced is further processed by cold working, it has been found that the resistance to intergranular corrosion is reduced. This can advantageously be avoided if the cold-worked alloy is subjected to flash annealing for 10 minutes to 2 hours at a temperature between 250 and 600 ° C.
Ausführungsbeispiele der Erfindung werden anhand der folgenden Versuche, Zeichnungen und Tabellen näher erläutert. Dabei zeigen:
- Figur 1:
- Die prozentuale Anfälligkeit für interkristalline Korrosion verschiedener Versuchslegierungen in Abhängigkeit vom Arsengehalt,
- Figur 2:
- die prozentuale Anfälligkeit für interkristalline Korrosion verschiedener Versuchslegierungen in Abhängigkeit vom jeweiligen Mangangehalt und
- Figur 3:
- die Abgabe von Blei an Trinkwasser einer Versuchslegierung gegenüber Legierungen des Standes der Technik in Abhängigkeit von der Zeit.
- FIG. 1:
- The percentage susceptibility to intergranular corrosion of various experimental alloys as a function of the arsenic content,
- FIG. 2:
- the percentage susceptibility to intergranular corrosion of various experimental alloys depending on the respective manganese content and
- FIG. 3:
- the delivery of lead to drinking water of a trial alloy compared to alloys of the prior art as a function of time.
Es wurden insgesamt 44 Kupfer-Zink-Legierungen unterschiedlicher Zusammensetzung untersucht. In Tabelle 1 ist die Zusammensetzung dieser 44 Versuchslegierungen hinsichtlich Kupfer, Blei, Arsen, Eisen, Silizium, Mangan, Aluminium und Zink aufgeführt. Für die jeweilige metallische Komponente jeder Legierung ist der Soll- und der Ist-Anteil in Gew.-% wiedergegeben. Die Versuchslegierungen sind jeweils mit vierstelligen Ziffern bezeichnet. Diese Bezeichnung wird im folgenden für die jeweilige Legierung verwendet.A total of 44 copper-zinc alloys of different composition were investigated. Table 1 lists the composition of these 44 experimental alloys in terms of copper, lead, arsenic, iron, silicon, manganese, aluminum and zinc. For the respective metallic component of each alloy, the desired and the actual proportion in wt .-% is reproduced. The trial alloys are each designated with four-digit numbers. This term is used below for the respective alloy.
Die in Tabelle 1 aufgeführten Versuchslegierungen wurden wie folgt hergestellt:
1. Gießen der aufgeschmolzenen Legierung bei einer Temperatur von 1050 bis 1100 °C in eine Stahlkokille der Abmessungen 110 mm x 40 mm
2. Abfräsen der Probe auf eine Dicke von 14 mm
3. Abwalzen der Probe auf 5 mm, mit Zwischenglühungen für jeweils 1 Stunde bei Temperaturen von etwa 700 °C
4. α-Glühen für 2 Stunden bei einer Temperatur von 550 Grad Celsius
1. Cast the molten alloy at a temperature of 1050 to 1100 ° C in a steel mold of the
2. Milling the sample to a thickness of 14 mm
3. Rolling the sample to 5 mm, with intermediate annealing for 1 hour at temperatures of about 700 ° C.
4. α-anneal for 2 hours at a temperature of 550 degrees Celsius
Es wurden folgende Versuche hinsichtlich einer Optimierung der Zusammensetzung durchgeführt:The following experiments were carried out with regard to an optimization of the composition:
Für jede der in Tabelle 1 aufgeführten Versuchslegierungen wurde ein Entzinkungstest gemäß ISO 6509 durchgeführt. Dabei wird der Entzinkungsgrad der jeweiligen Legierung nach einer vordefinierten Versuchsdurchführung in korrodierenden Flüssigkeiten durch Messung einer maximalen Entzinkungstiefe in µm ermittelt. Die auf diese Art und Weise ermittelte maximale Entzinkungstiefe ist ein Maß für die Entzinkungsanfälligkeit der untersuchten Versuchslegierung. Je größer die maximale Entzinkungstiefe der jeweiligen Versuchslegierung ist, um so größer ist deren Anfälligkeit für eine Entzinkung.For each of the experimental alloys listed in Table 1, a dezincification test was performed according to ISO 6509. The degree of dezincification of the respective alloy becomes corroding after a predefined test procedure Liquids determined by measuring a maximum Entzinkungstiefe in microns. The maximum depth of dezincification determined in this way is a measure of the susceptibility of the test alloy to be dezincified. The greater the maximum dezincification depth of the particular experimental alloy, the greater its susceptibility to dezincification.
Die jeweils gemäß ISO 6509 ermittelten maximalen Entzinkungstiefen sind in Tabelle 2 für die Versuchslegierungen wiedergegeben.The maximum dezincification depths determined according to ISO 6509 are reproduced in Table 2 for the experimental alloys.
Der Test gemäß ISO 6509 zur Feststellung der Entzinkungsbeständigkeit arbeitet mit einer hinsichtlich der Entzinkung sehr aggressiven Elektrolytlösung, die zum Feststellen der IK, die auf sehr feine Potentialunterschiede zwischen den Korngrenzen und der Kornfläche zurückzuführen ist, nicht geeignet ist.The dezincification resistance test according to ISO 6509 uses a very aggressive electrolyte solution for dezincification, which is not suitable for detecting IK due to very fine potential differences between grain boundaries and grain area.
Aus diesem Grund wurden alle Versuchslegierungen solchen Versuchsbedingungen ausgesetzt, von denen bekannt ist, dass diese zur interkristallinen Korrosion führen. Hierzu wurden beispielsweise Versuche mit Ammoniumchlorid-haltigen Lösungen unter definierten Bedingungen durchgeführt. Der Grad der interkristallinen Korrosion wurde in Relation zu dem Grad der interkristallinen Korrosion einer CuZn36 Pb2As-Legierung gesetzt, deren bekannte interkristalline Korrosion als 100 % gesetzt wurde. Das Ergebnis dieser Untersuchungen ist in Spalte 2 der Tabelle 2 aufgeführt.
Es wurde in
Eine Probe einer CuZn36Pb2As-Legierung mit einem Durchmesser von 27 mm wurde nach einem α-Glühen von 500 bzw. von 600 °C jeweils einmal in einem Wasserbad abgeschreckt und einmal im Ofen abgekühlt. Es wurde der Anteil an IK gemessen. Es zeigte sich, dass sich durch eine aktive Abkühlung die Anfälligkeit gegen IK zwischen 27 und 60 % verringern läßt.A sample of 27 mm diameter CuZn36Pb2As alloy was quenched once each in a water bath after an α-annealing of 500 and 600 ° C, respectively, and once cooled in the oven. The proportion of IK was measured. It has been shown that active cooling can reduce susceptibility to IK by between 27 and 60%.
In
Es zeigt sich, dass für eine Kupfer-Zink-Legierung mit steigendem Arsengehalt die Anfälligkeit für interkristalline Korrosion steigt. Bei einem Arsengehalt von 0,14 Gew.-% ist die IK-Anfälligkeit der Vergleichslegierung entsprechend Tabelle 2 erreicht. Die Anfälligkeit für IK nimmt aber für kleine Arsengehalte beträchtlich ab.It turns out that for a copper-zinc alloy with increasing arsenic content the susceptibility to intergranular corrosion increases. At an arsenic content of 0.14 wt .-%, the IK susceptibility of the comparative alloy according to Table 2 is reached. However, susceptibility to IC decreases significantly for small arsenic levels.
Allerdings wird für eine von Arsen freie Legierung wie z.B. Nr. 2156 eine sehr hohe maximale Entzinkungstiefe ersichtlich. Soll also eine Messinglegierung entzinkungsbeständig sein, so muss ein gewisser Gehalt an Arsen vorhanden sein. Unter diesem Gesichtspunkt ergibt sich aus
Die Versuchslegierungen Nr. 2180 und 2181 weisen hinsichtlich ihres Arsengehalts einen bezüglich der IK unkritischen Wert von 0,05 Gew.-% auf. Zusätzlich zeigen diese Versuchslegierungen jedoch einen geringen Anteil an Eisen von 0,03 Gew.-% bzw. 0,06 Gew.-%. Ein Vergleich mit den eisenfreien Versuchslegierungen Nr. 2088 und 2089 zeigt, dass sich durch den Zusatz an Eisen offensichtlich die kritische Obergrenze für den Arsengehalt erniedrigt. Innerhalb der Fehlertoleranzen nimmt die Anfälligkeit für interkristalline Korrosion zu. Ebenso steigt die Anfälligkeit für Entzinkung beim Hinzufügen von Eisen. Wird die Versuchslegierung Nr. 2159 hinzugezogen, die einen Eisengehalt von 0,082 Gew.-% aufweist, so wird deren Anfälligkeit für eine interkristalline Korrosion klar ersichtlich. Die Versuchslegierung Nr. 2159 zeigt entsprechend Tabelle 2 eine Anfälligkeit gegen IK von 100 %, wogegen die Versuchslegierungen Nr. 2088 und Nr. 2089 eine Anfälligkeit von lediglich 19 % bzw. 46 % zeigen.The experimental alloys Nos. 2180 and 2181 have a value of 0.05% by weight with respect to their arsenic content which is not critical with respect to the IC. In addition, however, these trial alloys show a small amount of iron of 0.03 wt% and 0.06 wt%, respectively. A comparison with the non-ferrous experimental alloys Nos. 2088 and 2089 shows that the addition of iron obviously lowers the critical upper limit for the arsenic content. Within fault tolerances, susceptibility to intergranular corrosion increases. Likewise, the susceptibility to dezincification increases with the addition of iron. When the trial alloy No. 2159 having an iron content of 0.082% by weight is used, its susceptibility to intergranular corrosion becomes clear. The trial alloy No. 2159 shows a susceptibility to IK of 100% according to Table 2, whereas the trial alloys No. 2088 and No. 2089 show a susceptibility of only 19% and 46%, respectively.
Wird der Arsengehalt erhöht, so wird die Anfälligkeit gegen eine interkristalline Korrosion durch das Hinzufügen von Eisen nicht mehr wesentlich verschlechtert. Dies wird durch Vergleich der Versuchslegierungen Nr.2175 bis 2178, die bei einem Arsengehalt von 0,08 Gew.-% einen steigenden Eisen-Gehalt aufweisen, mit den Versuchslegierungen Nr. 2089 oder Nr. 2090 ersichtlich.If the arsenic content is increased, the susceptibility to intergranular corrosion by the addition of iron is no longer significantly deteriorated. This can be seen by comparing Experimental alloys Nos. 2175 to 2178, which show an increasing iron content at an arsenic content of 0.08% by weight, with the experimental alloys No. 2089 or No. 2090.
Durch geringe Eisen-Zusätze wird demnach die kritische Obergrenze für den Arsen erniedrigt, so dass letztendlich durch den Einfluss von Eisen auch bei "sicheren" Arsengehalten eine interkristalline Korrosion auftreten kann. Eisen stellt demnach ein kritisches Element dar. Eisen ist demnach unerwünscht, lässt sich jedoch aus Kostengründen nicht vermeiden, da es bei Verwendung von billigem Messingschrott als Verunreinigung wieder in den Herstellungsprozess einfließt. Das Herstellen einer eisenfreien Messinglegierung aus reinen bzw. eisenfreien Ausgangssubstanzen ist erheblich teurer.Low iron additions therefore lower the critical upper limit for the arsenic so that intergranular corrosion can ultimately occur due to the influence of iron even with "safe" arsenic contents. Iron is thus a critical element. Iron is therefore undesirable, but can not be avoided for cost reasons, since it flows when using cheap brass scrap as an impurity back into the manufacturing process. The production of an iron-free brass alloy from pure or iron-free starting substances is considerably more expensive.
Es hat sich jedoch überraschend weiter gezeigt, dass durch das Hinzufügen von Silizium das Eisen offensichtlich gebunden werden kann, und damit sein schädlicher Einfluss auf die Korrosionsbeständigkeit einer Messinglegierung unterdrückt werden kann. Dies wird aus den Legierungen Nr. 2094, 2102 und 2106 ersichtlich. Diese Legierungen weisen bei einem Arsengehalt von 0,07 Gew.-% Eisenanteile von 0,14 Gew.-%, 0,1 Gew.-% bzw. 0,2 Gew.-% auf. Gleichzeitig enthalten diese Versuchslegierungen Silizium mit einem Anteil von 0,08 Gew.-% und 0,2 Gew.-%. Ein Blick in die Tabelle 2 belegt, dass Silizium die schädlichen Wirkungen von Eisen aufheben kann. Die Anfälligkeit gegen interkristalline Korrosion sinkt. Ebenso sind diese Versuchslegierungen generell entzinkungsarm.However, it has surprisingly been found that by adding silicon, the iron can obviously be bound and thus its detrimental impact on the corrosion resistance of a brass alloy can be suppressed. This can be seen in alloys Nos. 2094, 2102 and 2106. These alloys have iron contents at an arsenic content of 0.07 wt% of 0.14 wt .-%, 0.1 wt .-% and 0.2 wt .-% to. At the same time, these experimental alloys contain silicon in a proportion of 0.08% by weight and 0.2% by weight. A look at Table 2 shows that silicon can neutralize the damaging effects of iron. Susceptibility to intergranular corrosion decreases. Likewise, these experimental alloys are generally dezincification.
Weiter wird aus den Versuchslegierungen Nr. 2160-2167 der Einfluss von Silizium auf die Korrosionsbeständigkeit der Versuchslegierungen ersichtlich. Bei sonst gleichen Anteilen an Blei, Arsen und Eisen wird von der Versuchslegierung Nr. 2160 bis zur Versuchslegierung Nr. 2167 der Siliziumgehalt gesteigert. Während die generelle Korrosionsbeständigkeit gleichbleibend gut bleibt, wird wiederum ersichtlich, dass ein Hinzufügen von Silizium in geringen Mengen die schädlichen Wirkungen von kleinen Mengen Eisen verhindern kann. Ab etwa 0,3 Gew.-% tritt jedoch wieder vermehrt eine Anfälligkeit gegen interkristalline Korrosion auf. Sollen also in einer Messinglegierung kleine Anteile von Eisen toleriert werden, um günstige Herstellungskosten zu erhalten, so können diese durch die Zugabe von kleinen Mengen an Silizium unschädlich gemacht werden. Bei tolerierbaren Anteilen von Eisen bis zu 0,3 Gew.-% sollte daher Silizium zwischen 0,03 und 0,3 Gew.-% enthalten sein.Further, from the experimental alloys No. 2160-2167, the influence of silicon on the corrosion resistance of the trial alloys becomes apparent. With otherwise equal amounts of lead, arsenic and iron, the silicon content is increased from test alloy No. 2160 to test alloy No. 2167. Again, while general corrosion resistance remains consistently good, it can be seen that adding silicon in small amounts can prevent the deleterious effects of small amounts of iron. However, from about 0.3% by weight, susceptibility to intercrystalline corrosion increases again. If small amounts of iron are to be tolerated in a brass alloy in order to obtain favorable production costs, these can be rendered harmless by the addition of small amounts of silicon. At tolerable levels of iron up to 0.3% by weight, therefore, silicon should be included between 0.03 and 0.3% by weight.
Aus den Legierungen Nr. 2135 und 2136, die einen Siliziumgehalt von etwa 2 Gew.-% haben, wird ersichtlich, dass diese wieder anfällig gegen eine interkristalline Korrosion werden.It is apparent from alloys Nos. 2135 and 2136, which have a silicon content of about 2% by weight, that they again become susceptible to intergranular corrosion.
In
Die zulässigen Toleranzen für Mangan müssen daher niedrig bleiben. Unter einem Anteil von 0,04 Gew.-% ist die Anfälligkeit gegen interkristalline Korrosion bei unter 50 %.The permissible tolerances for manganese must therefore remain low. At a level of 0.04 wt%, the susceptibility to intergranular corrosion is less than 50%.
Ist Silizium in einer Messinglegierung enthalten, so hat es sich ferner gezeigt, dass sich der Zunder dieser Messinge leicht ablösen kann. Dies führt dazu, dass durch den umherfliegenden Zunder Produktionsanlagen verunreinigt werden. Dieses Problem kann durch Hinzufügen von Aluminium behoben werden. Glühversuche (Dauer: 2 Stunden, Temperatur: 750 Grad Celsius) an der Versuchslegierung Nr. 2102, die beständig gegen interkristalline Korrosion ist, bestätigten, dass sich deren Zunderschicht leicht ablöst. Durch einen Zusatz von Aluminium mit einem Anteil von etwa 0,2 Gew.-% konnte erreicht werden, dass sich die Haftung der Zunderschicht verbessert.If silicon is contained in a brass alloy, then it has also been found that the scale of these brass rings can easily come off. This leads to the fact that the circulating tinder contaminates production plants. This problem can be solved by adding aluminum. Annealing tests (duration: 2 hours, temperature: 750 degrees Celsius) on the experimental alloy No. 2102, which is resistant to intergranular corrosion, confirmed that its scale layer easily peels off. By adding aluminum in a proportion of about 0.2 wt .-% could be achieved that improves the adhesion of the scale layer.
In
Es zeigt sich überraschend, dass eine gegen interkristalline Korrosion beständige Legierung, nämlich die Versuchslegierung, eine niedrigere Bleiabgabe zeigt, als die entzinkungsbeständige Messinglegierung CuZn36Pb2,7As0,14 gemäß Stand der Technik.It is surprisingly found that an alloy resistant to intergranular corrosion, namely the trial alloy, exhibits a lower lead output than the dezincification resistant brass alloy CuZn36Pb2.7As0.14 according to the prior art.
Claims (6)
- Copper-zinc alloy resistant to dezincification, in particular for use in the sanitary sector, characterized in that it comprises
50 to 80% by weight Cu,
0 to 5% by weight Pb,
0.01 to 0.10% by weight As,
0.03 to less than 0.3% by weight Si,
0 to 0.3% by weight Fe,
0 to 0.04% by weight Mn,
and optionally 0.15 to 0.3% by weight Al,
remainder Zn and also inevitable impurities, and in that the effective Cu equivalent is 60 to 70% by weight. - Copper-zinc alloy according to Claim 1, characterized in that 0 to 0.02% by weight Mn are present.
- Copper-zinc alloy according to either of the preceding claims, characterized in that 0.02 to 0.08% by weight As are present.
- Copper-zinc alloy according to one of the preceding claims, characterized in that up to 0.2% by weight Fe and 0.15 to 0.25% by weight Si are present.
- Process for producing a copper-zinc alloy according to one of the preceding claims, wherein the starting materials are mixed in the weight ratio indicated, melted and cast to form a metallic alloy, and wherein the metallic alloy is annealed to form the α-phase at 500-650°C, characterized in that the annealed metallic alloy is actively cooled by means of coolants.
- Process according to Claim 5, characterized in that the metallic alloy is subjected to stress-relief annealing after a further treatment by cold forming at 250 to 600°C for between 10 min and 2 h.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE10132055 | 2001-07-05 | ||
| DE2001132055 DE10132055C2 (en) | 2001-07-05 | 2001-07-05 | Dezincification-resistant copper-zinc alloy and process for its production |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1273671A1 EP1273671A1 (en) | 2003-01-08 |
| EP1273671B1 true EP1273671B1 (en) | 2012-11-28 |
Family
ID=7690352
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP20020015027 Expired - Lifetime EP1273671B1 (en) | 2001-07-05 | 2002-07-05 | Dezincification resistant copper-zinc alloy and method for producing the same |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP1273671B1 (en) |
| DE (1) | DE10132055C2 (en) |
| ES (1) | ES2399234T3 (en) |
| PT (1) | PT1273671E (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3665312B1 (en) * | 2017-08-11 | 2024-03-06 | LIXIL Corporation | Copper alloy, use of a copper alloy, sanitary fitting and method for producing a sanitary fitting |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE10301552B3 (en) * | 2003-01-16 | 2004-06-24 | Rehau Ag + Co. | Use of a brass alloy for corrosion resistant drinking water molded parts, especially coupling parts, angular parts, angular bent parts, T-pieces, distribution parts and fittings |
| DE102007055446A1 (en) * | 2007-11-12 | 2009-05-14 | Hansgrohe Ag | Provision of water-bearing components from brass alloys with reduced metal ion release |
| CN101451204B (en) * | 2008-12-15 | 2010-10-13 | 无锡吉泉五金机械有限公司 | Guide ball for automobile air conditioner and preparation method thereof |
| CN101440445B (en) | 2008-12-23 | 2010-07-07 | 路达(厦门)工业有限公司 | Leadless free-cutting aluminum yellow brass alloy and manufacturing method thereof |
| CL2010000655A1 (en) * | 2010-06-18 | 2010-10-22 | Com Kraviva Spa | Copper alloy horseshoes with bactericidal and fungicidal properties comprising copper, zinc, aluminum, iron and optionally manganese. |
| DE102013003817A1 (en) * | 2013-03-07 | 2014-09-11 | Grohe Ag | Copper-zinc alloy for a sanitary fitting and method for its production |
| DE102013004081B4 (en) * | 2013-03-11 | 2023-06-07 | Hansa Metallwerke Ag | Sanitary fitting body |
| DE102013004383B4 (en) * | 2013-03-12 | 2015-06-03 | Diehl Metall Stiftung & Co. Kg | Use of a copper-zinc alloy |
| CN104087782A (en) * | 2013-04-01 | 2014-10-08 | 浙江艾迪西流体控制股份有限公司 | Low-lead brass alloy and preparation method thereof |
| SE1450094A1 (en) * | 2014-01-30 | 2015-07-31 | Arsenic-free brass with improved zinc toughness and cutability |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BE757349A (en) * | 1969-10-10 | 1971-03-16 | A H Andersson & Co A B | TANK AND ZINC ALLOY SPECIFICALLY SUITABLE FOR HOT PRESSURE FORMING |
| US3963526A (en) * | 1972-08-22 | 1976-06-15 | Aktieselskabet Nordiske Kabel-Og Traadfabriker | Method of imparting increased dezincification resistance to brass |
| FR2356733A1 (en) * | 1976-06-30 | 1978-01-27 | Toyo Valve Co Ltd | Copper based alloy for mfr. of valves, stop cocks etc. - has excellent corrosion resistance, and machinability |
| FR2506334B1 (en) * | 1981-05-21 | 1986-01-10 | Champagne Ardennes Affinage | BIPHASE BRASS ALLOY FOR FOUNDRY, CONTAINING ARSENIC, AND HAVING IMPROVED IMPLEMENTING PROPERTIES AND A VERY GOOD RESISTANCE OF THE CAST PIECES, TO DEZINCIFICATION |
| SE8800931D0 (en) * | 1988-03-16 | 1988-03-16 | Tour & Andersson Ab | BRASS ALLOY AND SET TO MAKE IT SAME |
| DE19722827A1 (en) * | 1997-05-30 | 1998-12-03 | Diehl Stiftung & Co | Cold formable lead-containing brass for sanitary piping |
| SE511680C2 (en) * | 1998-03-06 | 1999-11-08 | Tour & Andersson Hydronics Ab | Brass alloy with zinc leaching resistance, suitable for hot pressing |
| JP3917304B2 (en) * | 1998-10-09 | 2007-05-23 | 三宝伸銅工業株式会社 | Free-cutting copper alloy |
| SE514752C2 (en) * | 1999-08-26 | 2001-04-09 | Tour & Andersson Hydronics Ab | Zinc-resistant brass alloy for die-casting |
-
2001
- 2001-07-05 DE DE2001132055 patent/DE10132055C2/en not_active Revoked
-
2002
- 2002-07-05 EP EP20020015027 patent/EP1273671B1/en not_active Expired - Lifetime
- 2002-07-05 PT PT2015027T patent/PT1273671E/en unknown
- 2002-07-05 ES ES02015027T patent/ES2399234T3/en not_active Expired - Lifetime
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3665312B1 (en) * | 2017-08-11 | 2024-03-06 | LIXIL Corporation | Copper alloy, use of a copper alloy, sanitary fitting and method for producing a sanitary fitting |
Also Published As
| Publication number | Publication date |
|---|---|
| DE10132055A1 (en) | 2003-01-23 |
| PT1273671E (en) | 2013-02-27 |
| EP1273671A1 (en) | 2003-01-08 |
| ES2399234T3 (en) | 2013-03-26 |
| DE10132055C2 (en) | 2003-12-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE3631119C2 (en) | ||
| DE69828818T2 (en) | AUTOMATED ALLOY ON COPPER BASE | |
| DE102005002763B4 (en) | Copper alloy with high strength and high conductivity | |
| EP1683882B2 (en) | Aluminium alloy with low quench sensitivity and process for the manufacture of a semi-finished product of this alloy | |
| EP2162558B1 (en) | Iron-nickel-chromium-silicon alloy | |
| EP2115179B1 (en) | Iron-nickel-chromium- silicon alloy | |
| EP2964798B1 (en) | Copper-zinc alloy for a plumbing fitting and method for the production thereof | |
| DE112004002639T5 (en) | Lead-free free-cutting copper-antimony alloys | |
| EP2742161B1 (en) | Copper zinc alloy | |
| DE112016005830B4 (en) | Metal gasket and process for its manufacture | |
| EP1273671B1 (en) | Dezincification resistant copper-zinc alloy and method for producing the same | |
| EP1798298A1 (en) | Use of a low-migration copper alloy and parts made of such alloy | |
| DE60206464T2 (en) | Ni alloy with improved oxidation resistance, hot strength and hot workability | |
| EP1439238B1 (en) | Corrosion resistant brass alloy for parts suitable for use in drinking water service | |
| DE69732386T2 (en) | Stainless steel for water with added ozone and its manufacturing process | |
| EP0264463B1 (en) | Copper-chromium-titanium-silicon alloy, process for its production and its use | |
| DE2714712A1 (en) | NICKEL ALLOY AND METHOD FOR MANUFACTURING IT | |
| DE19735361B4 (en) | Austenitic stainless steel | |
| CH669211A5 (en) | COPPER-CHROME-TITANIUM-SILICONE ALLOY AND THEIR USE. | |
| DE4143075C2 (en) | Use of a still cold-formable electromagnetic stainless steel as material for electronically controlled fuel injection systems | |
| DE2629838C3 (en) | Aluminum sheet for fins in heat exchangers and process for its production | |
| DE2033003B2 (en) | PROCESS FOR THE FORMING AND HEAT TREATMENT OF A CALMED, LOW ALLOY STEEL | |
| DE4233668C2 (en) | Corrosion-resistant copper-based alloy | |
| DE69814657T2 (en) | COPPER BASED ALLOY, CHARACTERIZED BY DECAY CURING AND CURING IN SOLID CONDITION | |
| DE60311803T2 (en) | Copper alloy having excellent corrosion resistance and dezincification resistance, and a method of producing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20021025 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| AKX | Designation fees paid |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| 17Q | First examination report despatched |
Effective date: 20090925 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 586230 Country of ref document: AT Kind code of ref document: T Effective date: 20121215 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 50215658 Country of ref document: DE Effective date: 20130124 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Free format text: AVAILABILITY OF NATIONAL TRANSLATION Effective date: 20130219 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2399234 Country of ref document: ES Kind code of ref document: T3 Effective date: 20130326 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20121128 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20121128 |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: EP Ref document number: 20130400439 Country of ref document: GR Effective date: 20130327 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20121128 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130228 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20121128 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20121128 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20121128 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20121128 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20130829 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 50215658 Country of ref document: DE Effective date: 20130829 |
|
| BERE | Be: lapsed |
Owner name: DIEHL METALL STIFTUNG & CO. K.G. Effective date: 20130731 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20121128 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130731 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130705 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130705 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 15 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 16 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CZ Payment date: 20190703 Year of fee payment: 18 Ref country code: PT Payment date: 20190703 Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GR Payment date: 20190716 Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 20190722 Year of fee payment: 18 Ref country code: GB Payment date: 20190719 Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20190719 Year of fee payment: 18 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20201126 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CZ Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200705 Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20190706 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 586230 Country of ref document: AT Kind code of ref document: T Effective date: 20200705 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20200705 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200731 Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20210210 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200731 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200705 Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20210208 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200705 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20210727 Year of fee payment: 20 Ref country code: FR Payment date: 20210727 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20210721 Year of fee payment: 20 Ref country code: TR Payment date: 20210701 Year of fee payment: 20 Ref country code: DE Payment date: 20210915 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R071 Ref document number: 50215658 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: EUG |