1. Field of the Invention:
-
The present invention relates to toner for a two-component type developer used for electrophotography. More particularly, the present invention relates to toner, which does not include a charge control agent, suitably used in an electrophotographic image forming apparatus such as an electrostatic copying machine and a laser beam printer.
2. Description of the Related Art:
-
A two-component type developer is used as one of the developers used for developing an electrostatic latent image on a photosensitive body in an electrophotographic image forming apparatus. The two-component type developer includes toner comprising a binder resin and a coloring agent such as carbon black, and magnetic carriers such as iron powder and ferrite particles.
-
An electrostatic latent image is developed by the following steps: the developer forms a magnetic brush shape on a developing roller by a magnetic field thereof and is carried out to the photosensitive body. In this step, the toner is charged by friction with the carrier so as to have a desired charge and polarity of charge. Then, the developer is contacted with the photosensitive body by the developing roller, resulting in attaching the toner onto the electrostatic latent image formed thereon. Generally, the toner includes a charge control agent which controls and stabilizes the charge of the toner so as to attach a constant amount of the toner on the electrostatic latent image and provide a good developed image for a long period of time. Negatively charged toner includes a negative charge control agent such as a dye of a metal complex including a metal ion such as chrome(III) (for example, an azo compound - chrome(III) complex), and an oxycarboxylic acid - metal complex (for example, a salicylic acid - metal complex) (Japanese Laid-Open Patent Publication No. 3-67268). Positively charged toner includes a positive charge control agent such as an oil soluble dye including nigrosine and an amine type charge control agent (Japanese Laid-Open Patent Publication No. 56-106249).
-
Many metal complexes, including a heavy metal ion such as a chrome ion, are used as a conventional charge control agent. They are carefully selected, in terms of environmental safety, so that only those having passed various toxicity tests and safety tests alone are used. Therefore, although they would be safe in themselves or when included in toner, it is more preferable to refrain from using the metal complexes including a heavy metal as the charge control agent. In addition, the charge control agent is expensive as compared with the other materials for toner such as a binder resin and a coloring agent, for example, carbon black. Therefore, although the charge control agent has a content of merely several %, this results in increasing the price of the resultant toner. Accordingly, it is desired to develop toner having no charge control agent of a metal complex.
-
Furthermore, when conventional toner is used for a long period of time, the toner components tend to attach on a surface of the carrier particle. The attached components are called a spent. The spent makes the carrier charge with the same polarity as the toner, resulting in the disadvantages that the toner can be scattered and transfer efficiency of toner image is decreased.
SUMMARY OF THE INVENTION
-
The toner for a two-component type developer of this invention comprises toner particles including a binder resin and magnetic powder dispersed in the binder resin. The binder resin is made of a resin having an anionic polar group, and the magnetic powder is contained in the toner particles in the range of 0.1 to 5 parts by weight based on 100 parts by weight of the binder resin. The toner particles have a volume-based average particle diameter of 5 through 15 µm, and spacer particles with a volume-based average particle diameter of 0.05 through 1.0 µm are attached onto the surfaces of the toner particles.
-
In one embodiment, the spacer particles include first spacer particles having a resistivity between 101 and 10⁵ Ω·cm, and second spacer particles having a resistivity between 10⁸ and 10¹³ Ω·cm.
-
In one embodiment, a stearic acid metallic salt is attached onto the surfaces of at least one of the spacer particles and the toner particles.
-
In one embodiment, the stearic acid metallic salt is at least one selected from the group consisting of zinc stearate and magnesium stearate.
-
In one embodiment, the stearic acid metallic salt is selectively attached onto the surfaces of the spacer particles.
-
In one embodiment, an extracted solution obtained by extracting the toner with methanol has substantially no absorption peak in the range of 280 to 350 nm, and has a substantially zero absorbance in the range of 400 to 700 nm.
-
In one embodiment, the magnetic powder is contained in the range of 0.5 to 3 parts by weight per 100 parts by weight of the binder resin.
-
Thus, the invention described herein makes possible the advantages of (1) providing toner with excellent chargeability including no charge control agent at all; (2) providing toner little scattered in development for realizing a copied image with a high quality; and (3) providing toner in which a spent is not caused even when used for a long period of time, and hence, by which an excellent image quality can be maintained and transfer efficiency can be stabilized.
-
These and other advantages of the present invention will become apparent to those skilled in the art upon reading and understanding the following detailed description with reference to the accompanying figures.
BRIEF DESCRIPTION OF THE DRAWINGS
-
- Figure 1 is a graph showing absorbance of a methanol extracted solution of toner according to the present invention in the range of 200 to 700 nm;
- Figure 2 is a graph showing absorbance of a methanol extracted solution of toner having a dye of an azo compound - chrome complex as a charge control agent in the range of 200 to 700 nm;
- Figure 3 is a graph showing absorbance of a methanol extracted solution of toner having a salicylic acid - metal complex as the charge control agent in the range of 200 to 700 nm;
- Figure 4 is a graph showing absorbance of a methanol extracted solution of carrier in a two-component magnetic developer used for a long time in which toner has a dye of an azo compound - chrome complex as the charge control agent and chargeability of carrier is unstabilized by a spent in the range of 200 to 700 nm;
- Figure 5 is a graph showing a relationship between shaking time and a spent ratio obtained with regard to two kind of a two-component magnetic developer, one comprising toner having a charge control agent and magnetic carrier and another comprising toner having no charge control agent and magnetic carrier;
- Figure 6 is a graph showing a relationship between shaking time and quantity of charge of toner obtained with regard to two kind of a two-component magnetic developer, one comprising toner having a charge control agent and magnetic carrier and another comprising the toner having no charge control agent and magnetic carrier;
- Figure 7 is a graph showing a relationship between an amount of spent of carrier and content of a charge control agent in a toner particle;
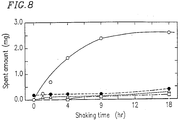
- Figure 8 is a graph showing a relationship between shaking time and amount of spent obtained in the case where each component contained in a toner particle and magnetic carrier are individually mixed and shaken;
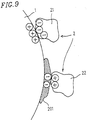
- Figure 9 illustrates a mechanism of a charge failure caused by a spent in a conventional two-component magnetic developer;
- Figure 10 is a schematic diagram of an original used in a copying performance test for observing a white dot in a black solid portion; and
- Figure 11 is a schematic diagram of a resistivity measuring device for measuring the resistivity of a spacer particle used in the invention.
DESCRIPTION OF THE PREFERRED EMBODIMENTS
-
Toner for a two-component type developer accord ing to the present invention has no charge control agent, such as a dye of an azo compound - metal complex and an oxycarboxylic acid - metal complex, at all. Therefore, a spent caused by a charge control agent, which will be described in detail below, scarcely occurs in the present toner, resulting in realizing a high quality copied image for a long period of time. Since the toner of the present invention has no charge control agent, it is impossible to detect any charge control agent, i.e., a dye type compound, from the toner by any chemical or physical method. For example, such a compound cannot be detected in the present toner by any chemical reaction. Alternatively, absorption peaks owing to such a compound cannot be detected in an organic solvent extracted solution of the present toner. For example, when the present toner is extracted with an organic solvent such as methanol, the extracted solution has substantially no absorption peak in the range of 280 to 350 nm, and has substantially zero absorbance in the range of 400 to 700 nm. Herein, "to have substantially no absorption peak" means, in an extracted solution obtained by extracting 0.1 g of the present toner with 50 ml of methanol, absorption peaks are not detected at all, or if detected, values of the absorbance peaks are 0.05 or less. Similarly, "to have substantially zero absorbance" means that values of the absorbance of the extracted solution obtained by extracting 0.1 g of the present toner with 50 ml of methanol are 0.05 or less.
-
In the present toner, instability of the charge of the toner due to a lack of a charge control agent is compensated for as follows. First, a polymer having an anionic group is used as a binder resin of a toner particle; and secondly, magnetic powder is contained in the toner particle at a predetermined proportion. In the present toner, in order to further enhance the function of the toner, two kinds of spacer particles having a predetermined resistivity and/or an attached stearic acid metallic salt are present. Therefore, chargeability of the toner is stabilized and the density of an image is improved.
-
The above-mentioned characteristics of the present toner will be described in detail.
-
Figure 1 shows an UV-visible spectrum of a methanol extracted solution of the present toner in the range of 200 to 700 nm. As is shown in this spectrum, the extracted solution has no peak, which is otherwise formed because of a charge control agent. Specifically, the solution has substantially no absorption peak in the range of 280 to 350 nm, and the absorbance in the range of 400 to 700 nm is substantially zero. To the contrary, in an absorbance curve of a methanol extracted solution of toner having a dye of an azo compound - chrome complex as a charge control agent shown in Figure 2, absorption peaks are found in the range of 400 to 700 nm, in particular, 550 to 570 nm. Further, in the UV-visible spectrum of a methanol extracted solution of toner having a salicylic acid - metal complex as a charge control agent shown in Figure 3, an absorption peak is found in the range of 280 to 350 nm.
-
It is because the charge control agent is present on the surfaces of the toner particles at a rather high concentration that the methanol extracted solution of the toner having the charge control agent has absorption peaks due to the charge control agent.
-
A carrier included in a developer which has insufficient chargeability owing to occurrence of a spent is extracted with methanol, and then the UV-visible spectrum of the extracted solution is measured to find absorption peaks in the range of 400 to 700 nm derived from a charge control agent. For example, the developer comprising the toner having a dye of an azo compound - chrome complex, whose UV-visible spectrum is shown in Figure 2, was used for a long period of time to cause a spent therein. Then, UV-visible spectrum of a methanol extracted solution of the carrier in this developer was measured to give the spectrum shown in Figure 4. As is shown in Figure 4, absorption peaks are found at the same position as the spectrum in Figure 2. It is conventionally understood that a spent is caused because a binder resin in the toner is attached to the surface of a carrier particle to form a resin film. The comparison between the absorbance curves in Figures 2 and 4, however, reveals that one of the major causes of a spent is the transfer of the charge control agent from the toner particles to the carrier particles.
-
The present inventors conducted the following experiments in order to find out more about the relationship between a charge control agent and a spent. First, toner comprising toner particles containing 1.5 wt% of the dye of the azo compound - chrome complex was mixed with a carrier to obtain a developer. The toner and the carrier was shaken for a predetermined period of time. Figure 5 shows a relationship between the shaking time and amount of an attachment on the surfaces of the carrier particles. In Figure 5, the amount of attachment is indicated as a spent ratio, that is, a percentage based on a total weight of the carrier particles bearing the attachment. Furthermore, Figure 6 shows the relationship between the shaking time and the amount of charge of the toner. The same procedure was repeated with regard to a developer comprising toner having no charge control agent and carrier. The experimental results of this developer are also shown in Figures 5 and 6, wherein the results obtained by the developer including the toner having the charge control agent are plotted with black circles, and those by the developer including the toner having no charge control agent are plotted with white circles. It is apparent from Figures 5 and 6 that a larger amount of attachment is formed on the carrier particles as the spent and the charge amount of the toner has a greater decrease in the developer including the toner particle having the charge control agent than in the developer including the toner particle having no charge control agent.
-
Next, the weight of toner components attached on the surfaces of the carrier particles as the spent was measured with time. The results are shown in a graph of Figure 7, wherein the abscissa indicates a measured amount of the spent and the ordinate indicates the content of the charge control agent in the toner particle. The broken line in Figure 7 indicates the amount of the charge control agent calculated in assuming that the toner components attached as the spent are identical to the components in the toner particles. Figure 7 reveals that a large amount of the charge control agent is deposited to be attached on the surfaces of the carrier particles at the initial stage. In Figure 7, as amount of the spent increases, the measured values approximate the calculated values. This is because they are experimental results obtained in a close system having no supply of fresh toner. Therefore, when toner is exchanged as in a copying machine, the difference between the measured values and the calculated values would be much larger.
-
Furthermore, the present inventors measured the weight of the attachment on the surfaces of the carrier particles resulting from mixing the carrier with each of the toner components, that is, a charge control agent, a binder resin, carbon black as a coloring agent and wax, so as to find out the relationships between the respective toner components and the spent. The results are shown in Figure 8 as a variation with time in the amount of the attachment (i.e., amount of the spent), wherein the results obtained from the mixture with the charge control agent is plotted with white circles, those from the carbon black with black circles, those from the binder resin with squares, and those from the wax with triangles. It is apparent from Figure 8 that the charge control agent causes the largest amount of attachment due to the spent.
-
Based on the above-mentioned facts, the charge failure caused by the spent in a conventional two-component magnetic developer is explained as follows referring to Figure 9. In the initial stage of the usage of a developer, a carrier particle 1 is positively charged and a toner particle 2 is negatively charged as is shown in an upper portion of Figure 9. In this case, the toner particle works as a negative toner particle 21. When this developer is continued to be used, a component including the charge control agent as a main component in the toner particle is attached on the surface of the carrier particle 1. Attachment 201, which is the spent, is negatively charged. The negatively charged attachment 201 leads to the formation of a toner particle having positive charge, that is, a reversely charged toner particle 22. The reversely charged toner particle 22 is formed on the surface of the carrier particle 1 as is shown in a lower portion of Figure 9, resulting in scattering of the toner and decreasing the transfer efficiency of the toner.
-
As described above, preferably, the toner does not have a charge control agent not only because the agent can include a heavy metal but also because the agent is the main cause of the spent, scatter of the toner and of a decrease in the transfer efficiency of the toner. Accordingly, the present toner has no charge control agent at all.
-
The instability of charge of the toner due to the lack of the charge control agent, in particular, the insufficiency in charge amount of the toner is compensated by using a binder resin having an anionic group as mentioned above. The insufficiency in charge amount of the toner particles can be supplemented because the binder resin has a negative charge in itself owing to the anionic group included therein. Since the anionic group is bonded to the main chain of the binder resin, it would never move onto the surface of the carrier particle as the charge control agent does, and hence it never causes the spent. On the contrary, charge around the surface of the toner particle caused by the anionic group of the binder resin is not so large that the electrostatic attraction between the toner particle and the carrier particle owing to the Coulomb force is insufficient when they are conveyed as a magnetic brush for development. Therefore, in a rapid copying operation, the toner cannot be sufficiently prevented from scattering because of insufficient coupling with the carrier particles. The scattered toner stains the inner wall of the copying machine, and can cause so-called a fog on a copied image.
-
In order to overcome such disadvantages, the present toner includes magnetic powder at a predetermined proportion, that is, 0.1 to 5 parts by weight on the basis of 100 parts by weight of the binder resin. The insufficiency in the charge amount of the toner particles can be thus compensated for. The magnetic powder contained in the toner particle causes magnetic attraction between the toner particle and the carrier particle. This magnetic attraction between the toner particle and the carrier particle together with electrostatic attraction prevents the toner from scattering. Moreover, since the number of the toner particles to be attached onto an electrostatic latent image is increased as the charge amount of one toner particle is smaller, apparent development sensitivity is increased.
-
The content of the magnetic powder in the toner particles is in the range of 0.1 to 5 parts by weight per 100 parts by weight of the binder resin as described above. When the content is less than 0.1 parts by weight, the charge amount of the toner particle is insufficient, resulting in insufficient coupling with the carrier particle and causing toner scattering. In this case, a fog can be disadvantageously formed on a copied image. Furthermore, the density of the copied image is low because of the insufficient charge amount. When the contents exceeds 5 parts by weight, the magnetic attraction between the carrier particle and the toner particle becomes so strong that the toner is not sufficiently attached onto an electrostatic latent image, resulting in decreasing the density of the copied image.
-
Several attempts have been made to improve the resolution of a copied image and the like by including (inclusively adding) magnetic powder as a toner component. For example, Japanese Laid-Open Patent Publication No. 56-106249 discloses a toner particle including 10 wt% of ferrite, and Japanese Laid-Open Patent Publication No. 59-162563 discloses a toner particle including 5 through 35 wt% of a magnetic fine particle. In either case, however, the content of the magnetic powder is excessive, and hence, the density of the copied image is low. Japanese Laid-Open Patent Publication No. 3-67268 discloses toner to which 0.05 to 2 wt% of magnetic powder is externally added. In this case, since the magnetic powder is not included in the toner particle, the powder is likely to be ununiformly attached onto the surface of the toner particle, resulting in insufficient magnetic attraction between the toner particle and the carrier particle. Furthermore, in either of the above-mentioned toners, the spent can be disadvantageously caused because a charge control agent is contained therein.
-
In the present invention, it is preferable that spacer particles having a particle diameter of 0.05 through 1.0 µm are attached onto the surfaces of the toner particles in order to increase the transfer efficiency for a toner image. The spacer particles can work as a fluidity enhancer for the toner particles, and in addition, form a gap between the photosensitive body and the toner particles when the toner is attached onto an electrostatic latent image formed on the photosensitive body. Therefore, the toner can be transferred from the photosensitive body onto transfer paper with ease even when the toner attains a large charge amount due to a long copying operation, resulting in a high transfer efficiency.
-
Furthermore, it is preferable that first spacer particles with a resistivity between 10¹ and 10⁵ Ω·cm, and second spacer particles with a resistivity between 10⁸ and 10¹³ Ω·cm are added to the present toner as the spacer particles. When these two kinds of spacer particles having different resistivity are attached onto the surfaces of the toner particles, the transfer efficiency of the toner from the photosensitive body to transfer paper is improved. In addition, the surface of a toner particle is divided into two types of areas having different surface resistivity. As a result, these two types of areas work separately for charging and discharging the toner. Therefore, it is possible to attain a desired charge amount with ease. This results in improving the charge stability of the resultant toner and satisfactorily stabilizing the density of a copied image.
-
Although fine particles having a particle diameter of approximately 0.015 µm are added as a fluidity enhancer in a conventional toner, such small particles cannot form a gap with a sufficient size between the photosensitive body and the toner particles. Therefore, they do not work as spacer particles for attaining the aforementioned purposes.
-
In the invention, a stearic acid metallic salt can be attached onto the surfaces of the toner particles and/or the spacer particles. This enhances the releasing property between the toner and the surface of the photosensitive body, and hence, the toner is unlikely to be attached onto the surface of the photosensitive body. In particular, when the stearic acid metallic salt is selectively attached onto the surfaces of the spacer particles alone, the stearic acid metallic salt effectively comes in contact with the surface of the photosensitive body, thereby more effectively preventing the toner and the spacer particles from attaching onto the photosensitive body.
-
When the stearic acid metallic salt is attached onto the surfaces of the toner particles, the resistance of the toner is appropriately decreased, resulting in a copied image with a high density. Furthermore, since the melt viscosity of the toner is decreased by the stearic acid metallic salt, the toner can be well penetrated into transfer paper in the thermal fixing process by a heat roller, resulting in improving the fixability of a copied image onto the transfer paper. Moreover, since the melting temperature of the stearic acid metallic salt is lower than that of the binder resin, the surface of a copied image formed by fixing a transfer image is more smooth, resulting in the formation of a glossy copied image.
-
Now, preferable resins to be used as the binder resin in the present toner will be described. Herein, a "lower alkyl group" indicates alkyl having 1 to 5 carbon atoms.
(Binder resin)
-
The binder resin contained in the toner particles of the present toner comprises a composition including a polymer having an anionic group. Such a binder resin is obtained by polymerizing a monomer having an anionic group or a mixture of the monomer having an anionic group with other monomers. The obtained resin can be a homopolymer or a copolymer.
-
The binder resin used in the present toner is preferably a copolymer, such as a random copolymer, a block copolymer and a grafted copolymer, obtained from a monomer having an anionic group and other monomers.
-
Examples of the monomer having an anionic group include monomers having a carboxylic acid group, a sulfonic acid group or a phosphoric acid group, and a monomer having a carboxylic acid group is generally used. Examples of the monomer having a carboxylic acid group include ethylenically unsaturated carboxylic acids such as acrylic acid, methacrylic acid, crotonic acid, maleic acid and fumaric acid; monomers that can form a carboxylic acid group such as maleic anhydride; and lower alkyl halfester of dicarboxylic acid such as maleic acid and fumaric acid. Examples of the monomer having a sulfonicacid group include styrene sulfonic acid and 2-acrylamido-2-methylpropane sulfonic acid. Examples of the monomer having a phosphoric acid group include 2-phosphono(oxy)propylmethacrylate, 2-phosphono(oxy) ethylmethacrylate, 3-chloro-2-phosphono(oxy) propylmethacrylate.
-
Such a monomer having an anionic group can be a free acid, a salt of an alkaline metal such as sodium and potassium, a salt of an alkaline earth metal such as calcium and magnesium, and a salt such as zinc.
-
The monomer having no anionic group used to prepare the binder resin is selected so that the resultant binder resin has a sufficient fixability and chargeability required of toner, and is one or a combination of an ethylenically unsaturated monomer. Examples of such a monomer include ethylenically unsaturated carboxylic acid ester, monovinyl arene, vinyl ester, vinyl ether, diolefin and monoolefin.
-
The ethylenically unsaturated carboxylic acid esters are represented by the following Formula (I):
wherein R¹ is a hydrogen atom or a lower alkyl group; and R is a hydrocarbon group having 11 or less carbon atoms or a hydroxyalkyl group having 11 or less carbon atoms.
-
Examples of such ethylenically unsaturated carboxylic acid esters include methyl acrylate, ethyl acrylate, butyl acrylate, 2-ethylhexyl acrylate, cyclohexyl acrylate, phenyl acrylate, methyl methacrylate, hexyl methacrylate, 2-ethylhexyl methacrylate, β-hydroxyethylacrylate, γ-hydroxypropylacrylate, δ-hydroxybutylacrylate and β-hydroxyethylmethacrylate.
-
The monovinyl arenes are represented by the following Formula (II):
wherein R³ is a hydrogen atom, a lower alkyl group or a halogen atom; R⁴ is a hydrogen atom, a lower alkyl group, a halogen atom, an alkoxy group, an amino group or a nitro group; and φ is a phenylene group.
-
Examples of such monovinyl arene include styrene, α-methylstyrene, vinyltoluene, α-chlorostyrene, o-chlorostyrene, m-chlorostyrene, p-chlorostyrene and p-ethylstyrene.
-
The vinyl esters are represented by the following Formula (III):
wherein R⁵ is a hydrogen atom or a lower alkyl group.
-
Examples of such vinyl esters include vinyl formate, vinyl acetate and vinyl propionate.
-
The vinyl ethers are represented by the following Formula (IV):
CH₂=CH-O-R⁶ (IV)
wherein R⁶ is a monovalent hydrocarbon group having 11 or less carbon atoms.
-
Examples of such vinyl ethers include vinyl methyl ether, vinyl ethyl ether, vinyl n-butyl ether, vinyl phenyl ether and vinyl cyclohexyl ether.
-
The diolefins are represented by the following Formula (V):
wherein R⁷, R⁸ and R⁹ are independently a hydrogen atom, a lower alkyl group or a halogen atom.
-
Examples of such diolefins include butadiene, isoprene and chloroprene.
-
The monoolefins are represented by the following Formula (VI):
wherein R¹⁰ and R¹¹ are independently a hydrogen atom or a lower alkyl group.
-
Examples of such monoolefins include ethylene, propylene, isobutylene, 1-butene, 1-pentene and 4-methyl-1-pentene.
-
Specific examples of the polymer having an anionic group, that is, a (co)polymer obtained through the polymerization of the aforementioned monomers, include styrene-acrylic acid copolymers, styrene-maleic acid copolymers and ionomer resins. Furthermore, a polyester resin having an anionic group can be also used. The polymer having an anionic group preferably includes the anionic group at a proportion for attaining an acid value of 2 through 30, and preferably 5 through 15, when the anionic group is present as a free acid. When part or the entire anionic group is neutralized, the anionic group is preferably contained at such a proportion that the acid value would be in the aforementioned range in assuming that it is present as a free acid. When the acid value, i.e., the concentration of the anionic group, of the polymer or the composition is below the aforementioned range, the chargeability of the resultant toner is insufficient. When it exceeds the range, the resultant toner disadvantageously has a hygroscopic property. A preferable binder resin is a copolymer obtained from the monomer having an anionic group and at least one of the ethylenically unsaturated carboxylic acid ester represented by Formula (I) as an indispensable components, and any of the monomers represented by Formulae (II) through (VI) as an optional component to be used if necessary. One or a combination of two or more of the aforementioned monomers is used for preparing the binder resin.
-
The composition can further comprise a polymer having no anionic group. In this case, the content of the anionic group in the entire composition is preferably within the aforementioned range.
(Magnetic powder)
-
The magnetic powder contained in (inclusively added to) the toner particles can be any magnetic powder used in a conventional one-component type developer. Examples of the material for the magnetic powder include triiron tetroxide (Fe₃O₄), maghemite (γ-Fe₂O₃), zinc iron oxide (ZnFe₂O₄), yttrium iron oxide (Y₃Fe₅O₁₂), cadmium iron oxide (CdFe₂O₄), gadolinium iron oxide (Gd₃Fe₅O₁₂), copper iron oxide (CuFe₂O₄), lead iron oxide (PbFe₁₂O₁₉), nickel iron oxide (NiFe₂O₄), neodyum iron oxide (NdFeO₃), barium iron oxide (BaFe₁₂O₁₉), magnesium iron oxide (MgFe₂O₄), manganese iron oxide (MnFe₂O₄), lanthanum iron oxide (LaFeO₃), iron (Fe), cobalt (Co) and Nickel (Ni). Particularly preferable magnetic powder is made from triiron tetroxide (magnetite) in the shape of fine particles. The particle of preferable magnetite is in the shape of a regular octahedron with a particle diameter of 0.05 through 1.0 µm. Such a magnetite particle can be subjected to a surface treatment with a silane coupling agent or a titanium coupling agent. The particle diameter of the magnetic powder contained in the toner particle is generally 1.0 µm or smaller, and preferably in the range between 0.05 and 1.0 µm.
-
The content of the magnetic powder in the toner particle is in the range of 0.1 to 5 parts by weight, more preferably 0.5 to 4 parts by weight, and most preferably 0.5 to 3 parts by weight per 100 parts by weight of the binder resin. When the content is too small, the toner can be scattered during the development and the transfer efficiency of the toner can be decreased as described above.
(Inner additives in the toner particles)
-
The toner particle contains, as described above, the binder resin and the magnetic powder as indispensable components, and can optionally include some inner additive generally used for a toner, if necessary.
-
Examples of such additives include a coloring agent and a release agent.
-
As the coloring agent, the following pigments can be used:
- Black pigment:
carbon black, acetylene black, lampblack, aniline black; - Extender:
barite powder, barium carbonate, clay, silica, white carbon, talc, alumina white.
-
Such a pigment is contained in the toner particle in the range of 2 to 20 parts by weight, and preferably 5 to 15 parts by weight per 100 parts by weight of the binder resin.
-
As the release agent, various wax and olefin resins can be used as in a conventional toner. Examples of the olefin resin include polypropylene, polyethylene, and propylene-ethylene copolymers, and polypropylene is particularly preferred.
(Preparation of the toner)
-
The toner particles in the present toner can be produced by any ordinary method for toner particles such as crushing and classification, fusing granulation, spray granulation and polymerization, and are generally produced by the crushing and classification method.
-
For example, the components for the toner particles are previously mixed in a mixer such as a Henschel mixer, kneaded with a kneader such as a biaxial extruder, and then cooled. The resultant is crushed and classified to give toner particles. The particle diameter of the toner particle is generally in the range between 5 and 15 µm and preferably between 7 and 12 µm in the volume-base averaged particle diameter (a medium size measured with a Coulter counter).
(Spacer particles)
-
In the present toner for a two-component type developer, the spacer particles are attached onto the surfaces of the toner particles to improve the transfer efficiency for a toner image. As the spacer particles, any of organic and inorganic inactive particles with a particle diameter of 0.05 through 1.0 µm, more preferably 0.07 through 0.5 µm can be used. Examples of material for such inactive particles include silica, alumina, titanium oxide, magnesium carbonate, an acrylic resin, a styrene resin and magnetic materials. The spacer particle can not only function as a fluidity enhancer but also increase the transfer efficiency as described above. The content of the spacer particles is 10 wt% or less, more preferably in the range between 0.1 and 10 wt%, and most preferably 0.1 and 5 wt% on the basis of the total weight of the toner. When the spacer particles are excessively included, the concentration of a copied image is insufficient.
-
In particular, it is preferable that two types of spacer particles with different resistivity as described below are attached onto the surfaces of the toner particles.
-
As first spacer particles, organic or inorganic inactive particles with a volume-based average particle diameter of 0.05 through 1.0 µm, preferably 0.07 through 0.5 µm, and a resistivity between 10¹ and 10⁵ Ω·cm, preferably between 10³ and 10⁵ Ω·cm, are used. Examples of material for such first spacer particles include titanium oxide and magnetite having been subjected to a surface treatment with a tin dope.
-
As second spacer particles, organic or inorganic inactive particles with a volume-based average particle diameter of 0.05 through 1.0 µm, more preferably 0.07 through 0.5 µm, and a resistivity between 10⁸ and 10¹³ Ω·cm, more preferably between 10⁹ and 10¹ Ω·cm, are used. Examples of material for such second spacer particles include silica, alumina, titanium oxide, zinc oxide, barium sulfate, magnesium carbonate, an acrylic resin, a styrene resin and a styrene-acrylic copolymer.
-
The resistivity of the spacer particle can be measured, for example, with a
resistivity measuring device 4 having a structure as is shown in Figure
11. The measuring method with the
resistivity measuring device 4 will now be described. First, 8 g of
spacer particles 203, as a sample, are put in a
tube 41 made of vinyl chloride having an inner diameter H₁ of 1 inch, and being sandwiched between
steel electrodes 421. The outer surfaces of the
electrodes 421 are provided with
Teflon plates 43 with a thickness of 2 mm. While applying a pressure Pr of 200 kg/cm to one of the
Teflon plates 43 with a hydraulic press, a resistance r(Ω) is measured at 500 V with an insulation resistance tester
42 (manufact ured by Toa Electronics Ltd.; SM-5). Then, a length H₂ (cm) between both of the ends of the
electrodes 421 as shown in Figure
11 is measured. Based on a difference between the measured length H₂ and 11.35 cm, that is, a length H₂ without containing a sample in the
tube 41, the thickness of the sample is calculated. Then, the resistivity R (Ω·cm) of the sample is calculated using the following equation:
-
The total content of the spacer particles is 10 wt% or less, preferably in the range between 0.1 and 10 wt%, and most preferably between 0.1 and 5 wt% based on the total weight of the resultant toner. When they are excessively included, the density of a copied image is insufficient.
-
Although the ratio of the first spacer particles to the second spacer particles contained in the present toner is not herein specifically limited, the weight ratio is preferably in the range from 1:7 to 7:1, and more preferably from 1:4 to 4:1. When these spacer particles are contained at such a ratio, it is possible to have the surface of a toner particle to work separately for charging and discharging the toner, thereby enhancing the effect to attain a desired charge amount.
(Stearic acid metallic salt)
-
Examples of a stearic acid metallic salt, which can be added to the toner particles bearing the aforementioned spacer particles, include zinc stearate, magnesium stearate, aluminum stearate, potassium stearate, chromium stearate, mercury stearate, cerium stearate, iron (II) stearate, sodium stearate, lead stearate and barium stearate. One or a mixture of two or more of the saits can be used. At least one selected from the group consisting of zinc stearate and magnesium stearate is preferably used, and more preferably, zinc stearate is singly used. Such a stearic acid metallic salt can be replaced with amide stearate.
-
The stearic acid metallic salt can be added to the toner particles bearing the spacer particles in the range of 0.001 to 3 wt%, preferably 0.003 to 1 wt%, and most preferably 0.005 to 0.2 wt% based on the total weight of the toner. The stearic acid metallic salt can be attached onto the surfaces of both the toner particles and the spacer particles. It is preferable, however, that the stearic acid metallic salt is attached selectively onto the surfaces of the spacer particles attached onto the toner particles. "To be attached selectively onto the surfaces of the spacer particles" herein means that the stearic acid metallic salt is not substantially attached onto the surface of a toner particle but attached onto the surface of a spacer particle alone. In order to selectively attach the stearic acid metallic salt onto the surface of a spacer particle, the stearic acid metallic salt is previously mixed with the spacer particles, and the resultant mixture is then added to the toner particles as described below.
(Other additives)
-
The present toner includes, as described above, the spacer particles with a predetermined particle diameter on the surfaces of the toner particles. If necessary, the spacer particles can be two types with different resistivity, or the stearic acid metallic salt can be attached onto the surfaces of the toner particles and/or the spacer particles. It is possible to further improve the fluidity of the resultant toner by attaching a fluidity enhancer, other than the stearic acid metallic salt, having a smaller particle diameter than that of the spacer particles, such as hydrophobic vapor depositioned silica particles.
-
The primary particle diameter of the fluidity enhancer such as the silica particles is generally approximately 0.015 µm. The fluidity enhancer can be added to the toner in an amount of 0.1 to 2.0 wt% based on the weight of the entire toner, i.e., the total weight of the toner particles and the additives (such as the spacer particles and the fluidity enhancer).
(Preparation of toner)
-
To the toner particles prepared as described above are added spacer particles, and a stearic acid metallic salt, a fluidity enhancer and the like if necessary, in the following manner. For example, the spacer particles (preferably, the two types of spacer particles), and a stearic acid metallic salt, a fluidity enhancer and the like, if necessary, are sufficiently mixed. Then the obtained mixture is added to the toner particles, and the resultant is sufficiently unbound. Thus, the spacer particles can be uniformly attached onto the surfaces of the toner particles. When a stearic acid metallic salt is added in this manner, the stearic acid metallic salt is attached onto the surfaces of both the toner particles and the spacer particles.
-
Alternatively, the toner particles are previously sufficiently mixed with a stearic acid metallic salt. Then, the spacer particles, and a fluidity enhancer, if necessary, are attached onto the surfaces of the toner particles already bearing the stearic acid metallic salt. Thus, in the resultant toner, the stearic acid metallic salt is not attached onto the surfaces of the spacer particles but onto the surfaces of the toner particles alone.
-
To "be attached" herein means both to be held in contact with the surface of a particle and to be partly embedded in the particle.
-
In this manner, the present toner is prepared.
(Carrier particle)
-
In the present invention, generally used magnetite or ferrite can be used as a carrier for the two-component type developer. In such a compound, the electrical resistance is stable and varies very little with time or by the change of the environment, and hence, it can provide the resultant developer with a stable chargeability. Further, such a compound is formed into a soft spicated shape in the developing apparatus when a magnetic field is applied. This prevents the turbulence of a toner image formed on the photosensitive body, thereby suppressing the formation of a white stripe in a copied image. The ferrite can be preferably used.
-
The carrier particle in the carrier used in the present invention is more preferably formed from a particle having a two-layered structure including a core particle and a coating layer over the core particle. The core particle comprises a magnetic material represented by the following Formula (A):
MOFe₂O₃ (A)
wherein M is at least one metal selected from the group consisting of Cu, Zn, Fe, Ba, Ni, Mg, Mn, Al and Co.
-
The compound represented by Formula (A) is magnetite (wherein M is Fe) or ferrite (wherein M is one of the metals other than Fe), and ferrite, wherein M is Cu, Zn, Mn, Ni or Mg, is preferably used. Change of the electrical resistance of such magnetite and ferrite is little for a long time, and the magnetite and ferrite can be formed into a soft spicated shape in the developing apparatus when a magnetic field is applied. The core particle comprising such a magnetic material has a particle diameter between 30 and 200 µm, and preferably between 50 and 150 µm. The core particles are obtained by granulating the fine particles of the magnetic material by spray granulation and the like, and then heating the resultant particles. The core particle has a volume specific resistivity between 10⁵ and 10⁹ Ω·cm, and preferably between 10⁶ and 10⁸ Ω·cm. The saturation magnetization of the core particle is in the range of 30 to 70 emu/g, and preferably between 45 and 65 emu/g.
-
The resin having a cationic group included in the resin composition, which forms the coating layer of the carrier particle, can be a thermoplastic resin and a thermosetting resin, and is preferably a thermosetting resin or a mixture of a thermosetting resin and a thermoplastic resin in terms of the heat resistance and the durability. Examples of the cationic group include a basic nitrogen containing group such as primary, secondary and tertiary amino groups, a quaternary ammonium group, an amido group, an imino group, an imido group, a hydrazino group, a guanidino group and an amidino group, among which an amino group and a quaternary ammonium group are particularly preferred.
-
Examples of the thermoplastic resin having a cationic group include thermoplastic acrylic resins, thermoplastic styrene-acrylic resins, polyester resins, polyamide resins and olefin copolymer, each of which includes a cationic group. Examples of the thermosetting resin include modified and unmodified silicone resins, thermosetting acrylic resins, thermosetting styreneacrylic resins, phenol resins, urethane resins, thermosetting polyester resins, epoxy resins and amino resins, each of which includes a cationic group. Such a resin including a cationic group is obtained by polymerizing a monomer having a cationic group or a mixture containing the monomer having a cationic group. Alternatively, such a resin is obtained by linking a compound having a cationic group with a resin having no cationic group. Further alternatively, a monomer having a cationic group and/or another monomer are (co)polymerized by using a polymerization initiator having a cationic group, thereby introducing the cationic group into the resultant resin.
-
When a resin prepared from alkoxysilane or alkoxytitanium is used, it is possible to produce the resin having a cationic group by allowing a silane coupling agent having a cationic group to react with the resin during or after the preparation of the resin. Examples of the silane coupling agent include N-(2-aminoethyl)-3-aminopropyltrimethoxysilane, N-(2-aminoethyl)-3-aminopropylmethyldimethoxysilane, γ-aminopropyltriethoxysilane and N-phenyl-3-aminopropyltrimethoxysilane. This type of silane coupling agent can be linked onto the surface of the core particle via a hydroxyl group generally present on the surface of the core particle. Therefore, such a silane coupling agent can form the coating layer by itself. Examples of the polymerization initiator having a cationic group include amidine type compound, e.g., azobis compounds.
-
The resin having a cationic group for forming the coating layer is used singly or together with any other of the aforementioned resins, or together with another resin having no cationic group.
-
The content of the cationic group in the resin having a cationic group is generally in the range of 0.1 to 2000 mmole, and preferably of 0.5 to 1,500 mmole per 100 g of the resin. When the resin having a cationic group is used with a resin having no cationic group, the cationic group is preferably contained in the entire resins forming the coating layer of the carrier particle at a proportion in the aforementioned range.
-
The resin composition forming the coating layer of the carrier particle includes at least one of the above-mentioned resins having a cationic group, together with another resin having no cationic group, if necessary. Examples of a mixture of the resin having a cationic group and the resin having no cationic group include a mixture of an alkylated melamine resin and a styrene-acrylic copolymer, and a mixture of an alkylated melamine resin and an acryl-modified silicone resin. The resin composition can further comprise an additive such as silica, alumina, carbon black, fatty acid metal salt, a silane coupling agent and silicone oil. These additives work for regulating physical properties of the coating layer.
(Preparation of the carrier)
-
The resin composition including a cationic group is applied to the surface of the core particle by a known method to form the coating layer. For example, the core particle is coated with a solution or a dispersion of the resin composition and dried, thereby forming the coating layer. Alternatively, when a thermosetting resin or a reactive resin oligomer is used, the core particle is coated with an uncured resin, or a solution or a dispersion of the oligomer, and then heated to cure the resin. The coating layer can be formed by any of the generally used methods such as immersion, spray, a fluidized bed method, a moving bed method and a tumbling layer method. As a solvent used to dissolve or disperse the resin composition, any of the ordinary organic solvents can be used. Examples of the solvent include aromatic hydrocarbons such as toluene and xylene; ketones such as acetone, methyl ethyl ketone, methyl isobutyl ketone and cyclohexanone; cyclic ethers such as tetrahydrofuran and dioxane; alcohols such as ethanol, propanol and butanol; cellosolves such as ethyl cellosolve and butyl cellosolve; esters such as ethyl acetate and butyl acetate; and amide type solvents such as dimethylformamide and dimethylacetoamide. The solvent is appropriately selected in accordance with the chemical properties of the resin such as the solubility.
-
The particle diameter of the thus obtained carrier particle is in the range of 30 to 200 µm, and preferably 50 to 150 µm. The weight ratio of the coating layer on the carrier particle is in the range of 0.001 to 2.5 parts by weight, and preferably 0.005 to 2.0 parts by weight per 100 parts by weight of the core particle. The obtained carrier particle has a volume specific resistivity in the range between 10⁵ and 10¹³ Ω·cm, and preferably 10⁷ and 10¹ Ω·cm, and a saturation magnetization in the range between 30 and 70 emu/g, and preferably 45 and 65 emu/g.
(Preparation of a developer)
-
A two-component type developer is prepared by mixing the above-mentioned toner and carrier. The mixing ratio of the carrier and the toner is generally 98:2 through 90:10, and preferably 97:3 through 94:6, by weight.
-
A copying operation is conducted using the present toner by a general electrophotographic method. Specifically, for example, a photoconductive layer on a photosensitive body is uniformly charged, and an image is exposed to form an electrostatic latent image thereon. Then, a magnetic brush made of the two-component magnetic developer is allowed to come in contact with the photosensitive body, thereby developing the electrostatic latent image with ease into a toner image. The thus obtained toner image is transferred onto transfer paper to form a transfer image, which is then applied with heat and pressure by a heat roller to fix the image thereon.
Examples
-
The present invention will now be described by way of examples. It is noted that the invention is not limited to these examples.
(Example 1.1)
<Preparation of toner>
-
-
The above listed components were fused and kneaded with a biaxial extruder, and the resultant was crushed with a jet mill, and classified with a pneumatic classifier to give toner particles with an average particle diameter of 10.0 µm.
-
To the obtained toner particles were added 0.3 parts by weight of hydrophobic silica fine powder with an average particle diameter of 0.015 µm as a fluidity enhancer and 0.5 parts by weight of acrylic resin particles with an average particle diameter of 0.15 µm as spacer particles, on the basis of 100 parts by weight of the toner particles.
<Preparation of a developer>
-
The thus obtained toner and a ferrite carrier with an average particle diameter of 100 µm were homogeneously mixed to give a two-component type developer having a toner concentration of 3.5 wt%.
(Example 1.2)
-
The same procedure was repeated as in Example 1.1 except that magnetite particles with an average particle diameter of 0.4 µm were added as the spacer particles, thereby preparing a developer.
(Comparative Example 1)
-
The same procedure was repeated as in Example 1.1 except that no spacer particles were used, thereby preparing a developer.
(Example 2.1)
<Preparation of toner>
-
-
The above listed components were fused and kneaded with a biaxial extruder, and the resultant was crushed with a jet mill, and classified with a pneumatic classifier to give toner particles with an average particle diameter of 10.0 µm.
-
To the obtained toner particles were added 0.3 parts by weight of hydrophobic silica fine powder with an average particle diameter of 0.015 µm as a fluidity enhancer on the basis of 100 parts by weight of the toner particles. Further, titanium oxide particles having been subjected to a surface treatment by a tin dope and having a volume-based average particle diameter of 0.3 µm and resistivity of 2 x 10³ Ω·cm were added as the first spacer particles at a proportion of 0.5 part by weight, and alumina particles having a volume-based average particle diameter of 0.3 µm and resistivity of 10¹⁰ Ω·cm were added as the second spacer particles at a proportion of 0.5 parts by weight, on the basis of 100 parts by weight of the toner particles. The resultant mixture was mixed with a Henschel mixer for 2 minutes to give toner including the toner particles on which the aforementioned particles were attached.
<Preparation of a developer>
-
The thus obtained toner and a ferrite carrier with an average particle diameter of 100 µm were homogeneously mixed to give a two-component type developer having a toner concentration of 3.5 wt%.
(Example 2.2)
-
The same procedure was repeated as in Example 2.1 except that the first spacer particles were added at a proportion of 1 part by weight on the basis of 100 parts by weight of the toner particles and that the second spacer particles were not added, thereby preparing a developer.
(Example 2.3)
-
The same procedure was repeated as in Example 2.1 except that the second spacer particles were added at a proportion of 1 part by weight on the basis of 100 parts by weight of the toner particles and that the first spacer particles were not added, thereby preparing a developer.
(Example 2.4)
-
The same procedure was repeated as in Example 2.1 except that the first and second spacer particles were not added, and that titanium oxide particles having a volume-based average particle diameter of 0.3 µm and resistivity of 10⁶ Ω·cm were added at a proportion of 1 part by weight on the basis of 100 parts by weight of the toner particles, thereby preparing a developer.
(Example 3.1)
<Preparation of toner>
-
Toner was prepared in the same manner as in Example 2.1.
<Preparation of a carrier>
-
Spherical ferrite particles with an average particle diameter of 100 µm were used as the magnetic core particles. To 1000 parts by weight of the ferrite particles was added a coating agent with components as listed in Table 1, and the resultant was mixed with a thermal stirrer. The solvent was removed from the resultant mixture, and the residue was subjected to a heat treatment at a temperature of 200°C for 1 hour to give carrier particles each having a coating layer.
<Preparation of a developer>
-
The thus obtained toner and carrier were homogeneously mixed to give a two-component type developer having a toner concentration of 3.5 wt%.
(Example 3.2)
-
The same procedure was repeated as in Example 3.1 except that the components of a used coating agent were as listed in Table 1, thereby preparing a developer.
(Example 3.3)
-
The same procedure was repeated as in Example 3.1 except that the components of a used coating agent were as listed in Table 1, thereby preparing a developer.
(Example 3.4)
-
The same procedure was repeated as in Example 3.1 except that a coating layer was not formed without using any coating agent, thereby preparing a developer.
Table 1 | Coating Agents of Examples 3.1-3.4 |
| component | Example 3.1 | Example 3.2 | Example 3.3 | Example 4.4 |
| Resin 1 | Acryl-modified silicone | Methylphenyl silicone | Styrene-acrylic polymer | none |
| parts by weight | 2.5 | 4.8 | 3.5 | |
| |
| Resin 2 | Methylated melamine | γ-aminopropyltriethoxysilene | Methylated melamine | none |
| parts by weight | 2.5 | 0.2 | 1.5 | |
| |
| Solvent: toluene (parts by weight) | 200 | 200 | 200 | none |
(Example 4.1)
<Preparation of toner>
-
-
The above listed components were fused and kneaded with a biaxial extruder, and the resultant was crushed with a jet mill, and classified with a pneumatic classifier to give toner particles with an average particle diameter of 10.0 µm.
-
To the obtained toner particles were added 0.3 parts by weight of hydrophobic silica fine powder with an average particle diameter of 0.015 µm as a fluidity enhancer, and 0.5 parts by weight of magnesium carbonate particles with an average particle diameter of 0.15 µm as spacer particles, on the basis of 100 parts by weight of the toner particles. Further, zinc stearate was added as the stearic acid metallic salt at a proportion of 0.05 parts by weight based on 100 parts by weight of the toner particles. The resultant mixture was mixed with a Henschel mixer for 2 minutes to give toner.
<Preparation of a developer>
-
The thus obtained toner and a ferrite carrier with an average particle diameter of 100 µm were homogeneously mixed to give a two-component type developer having a toner concentration of 3.5 wt%.
(Example 4.2)
<Preparation of toner>
-
First, 10 parts by weight of magnesium carbonate with an average particle diameter of 0.15 µm and 1 part by weight of zinc stearate as the stearic acid metallic salt were mixed with a Henschel mixer for 10 minutes.
-
The resultant mixture was added to the toner particles prepared in Example 4.1 at a proportion of 0.55 part by weight (i.e., 0.5 part by weight of magnesium carbonate and 0.05 part by weight of zinc stearate) on the basis of 100 parts by weight of the toner particles. Further, hydrophobic silica fine particles with an average particle diameter of 0.015 µm were added as a fluidity enhancer at a proportion of 0.3 part by weight on the basis of 100 parts by weight of the toner particles. The resultant mixture was mixed with a Henschel mixer for 2 minutes to give toner.
<Preparation of a developer>
-
The thus obtained toner and a ferrite carrier with an average particle diameter of 100 µm were homogeneously mixed to give a two-component type developer having a toner concentration of 3.5 wt%.
(Example 4.3)
<Preparation of toner>
-
The same procedure was repeated as in Example 4.2 except that 1 part by weight of zinc stearate was replaced with 2 parts by weight of magnesium stearate, and that the mixture of magnesium carbonate and magnesium stearate was added to the toner particles at a proportion of 0.52 parts by weight (i.e., 0.5 parts by weight of magnesium carbonate and 0.02 parts by weight of magnesium stearate), thereby preparing toner.
<Preparation of a developer>
-
The thus obtained toner and a ferrite carrier with an average particle diameter of 100 µm were homogeneously mixed to give a two-component type developer having a toner concentration of 3.5 wt%.
(Example 4.4)
-
The same procedure was repeated as in Example 4.1 except that zinc stearate was not added, thereby preparing a developer.
(Example 5.1)
<Preparation of toner>
-
Toner was prepared in the same manner as in Example 4.1.
<Preparation of a carrier>
-
Spherical ferrite particles with an average particle diameter of 100 µm were used as the magnetic core particles. To 1000 parts by weight of the ferrite particles was added a coating agent with components as listed in Table 2, and the resultant was mixed with a thermal stirrer. The solvent was removed from the resultant mixture, and the residue was subjected to a heat treatment at a temperature of 200°C for 1 hour to give carrier particles each having a coating layer.
<Preparation of a developer>
-
The thus obtained toner and carrier were homogeneously mixed to give a two-component type developer having a toner concentration of 3.5 wt%.
(Example 5.2)
-
The same procedure was repeated as in Example 5.1 except that the components of a used coating agent were as listed in Table 2, thereby preparing a developer.
(Example 5.3)
-
The same procedure was repeated as in Example 5.1 except that the components of a used coating agent were as listed in Table 2, thereby preparing a developer.
(Example 5.4)
-
The same procedure was repeated as in Example 5.1 except that a coating layer was not formed without using any coating agent, thereby preparing a developer.
Table 2 | Coating Agent of Examples 5.1-5.4 |
| component | Example 5.1 | Example 5.2 | Example 5.3 | Example 5.4 |
| Resin 1 | Acryl-modified silicone | Metylphenyl silicone | Styrene-acrylic polymer | none |
| parts by weight | 2.5 | 4.8 | 3.5 | |
| |
| Resin 2 | Metylated melamine | γ-aminoproyltriethoxysilane | Methylated melamine | none |
| parts by weight | 2.5 | 0.2 | 1.5 | |
| |
| Solvent: toluene (parts by weight) | 200 | 200 | 200 | none |
(Example 6.1)
<Preparation of toner>
-
Toner was prepared in the same manner as in Example 4.2.
<Preparation of a carrier>
-
Spherical ferrite particles with an average particle diameter of 100 µm were used as the magnetic core particles. To 1000 parts by weight of the ferrite particles was added a coating agent with components as listed in Table 3, and the resultant was mixed with a thermal stirrer. The solvent was removed from the resultant mixture, and the residue was subjected to a heat treatment at a temperature of 200°C for 1 hour to give carrier particles each having a coating layer.
<Preparation of a developer>
-
The thus obtained toner and carrier were homogeneously mixed to give a two-component type developer having a toner concentration of 3.5 wt%.
(Example 6.2)
-
The same procedure was repeated as in Example 6.1 except that the components of a used coating agent were as listed in Table 3, thereby preparing a developer.
(Example 6.3)
-
The same procedure was repeated as in Example 6.1 except that the components of a used coating agent were as listed in Table 3, thereby preparing a developer.
(Example 6.4)
-
The same procedure was repeated as in Example 6.1 except that a coating layer was not formed without using any coating agent, thereby preparing a developer.
Table 3 | Coating Agents of Examples 6.1-6.4 |
| component | Example 6.1 | Example 6.2 | Example 6.3 | Example 6.4 |
| Resin 1 | Acryl-modified silicone | Metylphenyl silicone | Styrene-acrylic polymer | none |
| parts by weight | 2.5 | 4.8 | 3.5 | |
| |
| Resin 2 | Metylated melamine | γ-aminopropyltriethoxysilane | Methylated melamine | none |
| parts by weight | 2.5 | 0.2 | 1.5 | |
| |
| Solvent: toluene (parts by weight) | 200 | 200 | 200 | none |
[Evaluation of the developers]
-
The developers obtained in the above described examples and comparative examples were evaluated with regard to the following items. An electric copying machine (manufactured by Mita Industrial Co., Ltd.; brand name: DC-4685) was modified so as to make easier evaluation sampling, and the modified copying machine was used in the evaluation.
- (a) Transfer efficiency:
The amount of toner in a toner hopper in the copying machine was measured at first, and a predetermined number of copies were made. Then, the amount of the toner left in the toner hopper was measured. From a difference between the amounts of the toner before and after the copying operation, a consumed amount of the toner was calculated. At the same time, the amount of the toner collected in a cleaning process during the copying operation was also measured as a collected amount. Based on these amounts, the transfer efficiency of the toner was calculated by using Equation (i) below. An original used in the copying operation bore characters with a black area ratio of 8%. This evaluation was conducted to perform various evaluation tests described in the following items (b) through (i).
- (b) Image density (I.D.):
A copying operation was continued by using an original bearing characters with a black area ratio of 8% until 100,000 copies were made with regard to the developers of Examples 1.1, 1.2 and Comparative Example 1, until 50,000 copies were made with regard to the developers of Examples 2.1 through 2.4 and 4.1 through 4.4, and until the transfer efficiency became less than 70% with regard to the other developers. The density of a black portion in a copied image on every 5000 copies was measured by a reflection densitometer (manufactured by Tokyo Denshoku Co., Ltd.; TC-6D), and the average density was taken as an image density (I.D.). An original used for sampling every 5000 copies had a black area ratio of 15% including a black solid portion. The results obtained from the developers of Examples 1.1 and 1.2 and Comparative Example 1 are listed in Table 4, those of Examples 2.1 through 2.4 in Table 5, those of Examples 3.1 through 3.4 in Table 6, those of Examples 4.1 through 4.4 in Table 7, those of Examples 5.1 through 5.4 in Table 8, and those of Examples 6.1 through 6.4 in Table 9.
- (c) Fog density (F.D.):
A copying operation was continued by using an original bearing characters with a black area ratio of 8% until 100,000 copies were made with regard to the developers of Examples 1.1 and 1.2 and Comparative Example 1, until 50,000 copies were made with regard to the developers of Examples 2.1 through 2.4 and 4.1 through 4.4, and until the transfer efficiency became less than 70% with regard to the other developers. The density of a white portion in a copied image on every 5000 copies was measured by a reflection densitometer (manufactured by Tokyo Denshoku Co., Ltd.; TC-6D). A difference between the thus measured density and the density of paper to be used for the copying operation (base paper) measured by the reflection densitometer was calculated, and the maximum difference was taken as a fog density (F.D.). An original used for sampling every 5000 copies had a black area ratio of 15% including a black solid portion. The results obtained from the developers of Examples 1.1 and 1.2 and Comparative Example 1 are listed in Table 4, those of Examples 2.1 through 2.4 in Table 5, those of Examples 3.1 through 3.4 in Table 6, those of Examples 4.1 through 4.4 in Table 7, those of Examples 5.1 through 5.4 in Table 8, and those of Examples 6.1 through 6.4 in Table 9.
- (d) Resolution:
A copying operation was conducted by using an original bearing characters with a black area ratio of 8%. When 40,000 copies were made with regard to the developers of Examples 1.1 and 1.2 and Comparative Example 1, and when 50,000 copies were made with regard to the other developers (in the case where the transfer efficiency became less than 70% before making 40,000 or 50,000 copies, at that time), a normal chart original (an original bearing a plurality of patterns in each of which a predetermined number of parallel lines are drawn per 1 mm) was copied, and the obtained copied image was visually evaluated. The results obtained from the developers of Examples 1.1 and 1.2 and Comparative Example 1 are listed in Table 4, those of Examples 2.1 through 2.4 in Table 5, those of Examples 3.1 through 3.4 in Table 6, those of Examples 4.1 through 4.4 in Table 7, those of Examples 5.1 through 5.4 in Table 8, and those of Examples 6.1 through 6.4 in Table 9.
- (e) Charge amount:
A copying operation was continued by using an original bearing characters with a black area ratio of 8% until 100,000 copies were made with regard to the developers of Examples 1.1 and 1.2 and Comparative Example 1, until 50,000 copies were made with regard to the developers of Examples 2.1 through 2.4 and 4.1 through 4.4, and until the transfer efficiency became less than 70% with regard to the other developers. During this copying operation, after making every 5,000 copies, the charge amount of 200 mg of the developer was measured by a blowoff type powder charge amount measuring device (manufactured by Toshiba Chemical Co., Ltd.), and the average of the charge amount per 1 g of the toner was calculated based on the measured value. The results obtained from the developers of Examples 1.1 and 1.2 and Comparative Example 1 are listed in Table 4, those of Examples 2.1 through 2.4 in Table 5, those of Examples 3.1 through 3.4 in Table 6, those of Examples 4.1 through 4.4 in Table 7, those of Examples 5.1 through 5.4 in Table 8, and those of Examples 6.1 through 6.4 in Table 9.
- (f) Toner scattering:
A copying operation was continued by using an original bearing characters with a black area ratio of 8% until 100,000 copies were made with regard to the developers of Examples 1.1 and 1.2 and Comparative Example 1, until 50,000 copies were made with regard to the developers of Examples 2.1 through 2.4 and 4.1 through 4.4, and until the transfer efficiency became less than 70% with regard to the other developers. Then, the toner scattering state in the copying machine was visually observed and evaluated. The results obtained from the developers of Examples 1.1 and 1.2 and Comparative Example 1 are listed in Table 4, those of Examples 2.1 through 2.4 in Table 5, those of Examples 3.1 through 3.4 in Table 6, those of Examples 4.1 through 4.4 in Table 7, those of Examples 5.1 through 5.4 in Table 8, and those of Examples 6.1 through 6.4 in Table 9. In these tables, ○ indicates that the toner was not scattered; and × indicates that the toner was scattered.
- (g) Durability:
After making every 10,000 copies, the transfer efficiency was calculated based on the consumed amount and the collected amount of the toner to find the number of copies that had been made before the transfer efficiency became less than 70%. The number was taken as an indicator for the durability of the developer. The results obtained from the developers of Examples 3.1 through 3.4 are listed in Table 6, those of Examples 5.1 through 5.4 in Table 8, and those of Examples 6.1 through 6.4 in Table 9.
- (h) Amount of attachment on the surface of the carrier particle due to the spent:
A copying operation was conducted by using an original bearing characters with a black area ratio of 8%. After making 40,000 copies with regard to the developers of Examples 1.1 and 1.2 and Comparative Example 1, 50,000 copies with regard to the other developers (in the case where the transfer efficiency became less than 70% before making 40,000 or 50,000 copies, at that time), the developer was tested as follows. The developer was placed on a screen of 400 mesh, and sucked from below with a blower, thereby separating the toner and the carrier. Five grams of the carrier remained on the screen and was charged in a beaker, to which toluene was added. Thus, the toner component attached onto the surfaces of the carrier particles due to the spent was dissolved. Then, the toluene solvent was discarded with the carrier attracted upon the bottom of the beaker with a magnet. This procedure was repeated several times until the resultant toluene solution became transparent. Then, the resultant carrier was heated with an oven to evaporate the toluene remaining thereto, and the weight of the obtained residue was measured. A difference between the weight of the carrier charged in the beaker at first (i.e., 5 g in this case) and the weight of the residue after evaporating the toluene was taken as the amount of the toner components attached onto the surfaces of the carrier particles due to the spent (i.e., the spent amount). The spent amount is indicated as the weight in mg of the toner components attached to 1 g of the carrier. The results obtained from the developers of Examples 1.1 and 1.2 and Comparative Example 1 are listed in Table 4, those of Examples 2.1 through 2.4 in Table 5, those of Examples 3.1 through 3.4 in Table 6, those of Examples 4.1 through 4.4 in Table 7, those of Examples 5.1 through 5.4 in Table 8, and those of Examples 6.1 through 6.4 in Table 9.
- (i) Presence of a white dot in a black solid portion:
By using an original 3 with a size of 210 mm x 297 mm bearing three black solid portions 31 each with a size of 50 mm x 50 mm as is shown in Figure 10, 500 copies were continuously made and the copied images were fixed with the heat rollers. The respective copied images were fed to the heat roller in the direction Pa as shown with a white arrow in Figure 10. The copied image at the 50,000 sheets was visually observed to determine whether or not a white dot was present in the three black solid portions. The results are listed in Table 7.
Table 4 | Toner component and Evaluation of Example 1.1, Example 1.2 and Comparative Example 1. |
| | Example 1.1 | Example 1.2 | Comparative Example 1 |
| Toner component (parts by weight) | | | |
| Binder resin having carboxyl groups | 100 | 100 | 100 |
| Carbon black | 7 | 7 | 7 |
| Magnetic powder | 2 | 2 | 2 |
| External additive | | | |
| ;Silica | 0.3 | 0.3 | 0.3 |
| ;Acrylic polymer particle | 0.5 | none | none |
| ;Magnetite | none | 0.5 | none |
| |
| Evaluation | | | |
| I.D. | 1.369 | 1.379 | 1.342 |
| F.D. | 0.002 | 0.001 | 0.003 |
| Resolution | 5.6 | 5.0 | 5.6 |
| Transfer efficiency (%) | 85.2 | 86.5 | 81.5 |
| Charge amount (µC/g) | -18.6 | -17.6 | -22.5 |
| Spent amount (mg) | 0.22 | 0.19 | 0.28 |
| Toner scattering | ○ | ○ | ○ |
Table 5 | Toner component and Evaluation of Example 2.1-2.4. |
| | Example 2.1 | Example 2.2 | Example 2.3 | Example 2.4 |
| Toner component (parts by weight) | | | | |
| Binder resin | 100 | 100 | 100 | 100 |
| Carbon black | 10 | 10 | 10 | 10 |
| Magnetic powder | 2 | 2 | 2 | 2 |
| Charge control agent | none | none | none | none |
| External additive (silica, 0.015 µm) | 0.3 | 0.3 | 0.3 | 0.3 |
| Spacer particle 1 | 0.5 | 1.0 | none | 1.0 |
| ;type | titanim oxide | titanim oxide | | titanim oxide |
| ;diameter (µm) | 0.3 | 0.3 | | 0.3 |
| ;resistivity (Ω·cm) | 2E1 | 2E1 | | 1E6 |
| Spacer particle |
| 2 | 0.5 | none | 1.0 | none |
| ;type | almina | | almina | |
| ;diameter (µm) | 0.3 | | 0.3 | |
| ;resistivity (Ω·cm) | 1E10 | | 1E10 | |
| |
| Evaluation | | | | |
| I.D. | 1.395 | 1.405 | 1.352 | 1.362 |
| F.D. | 0.001 | 0.005 | 0.003 | 0.003 |
| Resolution | 5 | 4.5 | 5 | 5 |
| Spent amount (mg) | 0.63 | 0.69 | 0.67 | 0.65 |
| Toner scattering | ○ | ○ | ○ | ○ |
| Transfer efficiency (%) | 84.2 | 73.1 | 78.5 | 74.4 |
| Charge amount (µC/g) | | | | |
| at 1 copy | -21.5 | -22.2 | -21.6 | -21.4 |
| at 10, 000 copies | -21.8 | -20.5 | -23.2 | -18.5 |
| at 30, 000 copies | -22.1 | -18.3 | -24.3 | -22.1 |
| at 50, 000 copies | -22.0 | -16.2 | -26.4 | -23.2 |
Table 6 | Evaluation of Examples 3.1-3.4 |
| | Example 3.1 | Example 3.2 | Example 3.3 | Example 3.4 |
| I.D. | 1.402 | 1.362 | 1.401 | 1.388 |
| F.D. | 0.003 | 0.003 | 0.003 | 0.004 |
| Resolution | 5 | 5 | 5 | 5 |
| Charge amount (µC/g) | -23.1 | -24.4 | -24.5 | -22.5 |
| Toner scattering | ○ | ○ | ○ | ○ |
| Durability (copies) | 140, 000 | 140, 000 | 130, 000 | 70, 000 |
| Spent amount (mg) at 50, 000 copies | 0.31 | 0.31 | 0.34 | 0.57 |
Table 7 | Toner component and Evaluation of Example 4.1-4.4. |
| | Example 4.1 | Example 4.2 | Example 4.3 | Example 4.4 |
| Toner component (parts by weight) | | | | |
| Binder resin | 100 | 100 | 100 | 100 |
| Carbon black | 10 | 10 | 10 | 10 |
| Magnetic powder | 2 | 2 | 2 | 2 |
| Charge control agent | none | none | none | none |
| Stearic acid metallic salt | 0.05 | 0.05 | 0.02 | none |
| ;metal | zinc | zinc | magnesium | |
| ;adding method | mixed with toner | mixed with MgCo₃ | mixed with MgCo₃ | |
| |
| External additive | | | | |
| ;silica (0.015 µm) | 0.3 | 0.3 | 0.3 | 0.3 |
| ;MgC0₃ (0.15 µm) | 0.5 | 0.5 | 0.5 | 0.5 |
| |
| Evaluation | | | | |
| I.D. | 1.420 | 1.409 | 1.401 | 1.368 |
| F.D. | 0.003 | 0.002 | 0.002 | 0.003 |
| Resolution | 5 | 5 | 5 | 5 |
| Charge amount (µC/g) | -18.6 | -19.3 | -19.8 | -23.2 |
| Toner scattering | ○ | ○ | ○ | ○ |
| Spent amount (mg) | 0.64 | 0.63 | 0.65 | 0.65 |
| White dot in the black portion | none | none | none | present |
Table 8 | Evaluation of Examples 5.1-5.4 |
| | Example 5.1 | Example 5.2 | Example 5.3 | Example 5.4 |
| I.D. | 1.404 | 1.358 | 1.409 | 1.398 |
| F.D. | 0.003 | 0.002 | 0.003 | 0.004 |
| Resolution | 5 | 5 | 5 | 5 |
| Charge amount (µC/g) | -20.3 | -22.7 | -22.0 | -19.5 |
| Toner scattering | ○ | ○ | ○ | ○ |
| Durability (copies) | 140, 000 | 140, 000 | 130, 000 | 70, 000 |
| Spent amount (mg) at 50, 000 copies | 0.32 | 0.32 | 0.34 | 0.57 |
Table 9 | Evaluation of Examples 6.1-6.4. |
| | Example 6.1 | Example 6.2 | Example 6.3 | Example 6.4 |
| I.D. | 1.405 | 1.355 | 1.411 | 1.393 |
| F.D. | 0.002 | 0.002 | 0.003 | 0.004 |
| Resolution | 5 | 5 | 5 | 5 |
| Charge amount (µC/g) | -20.6 | -23.2 | -21.4 | -18.7 |
| Toner scattering | ○ | ○ | ○ | ○ |
| Durability (copies) | 150, 000 | 150, 000 | 140, 000 | 70, 000 |
| Spent amount (mg) at 50, 000 copies | 0.29 | 0.30 | 0.33 | 0.56 |
[Review of the evaluation]
-
The developers produced in Examples 1.1 and 1.2 were more stable in the charge amount and had a smaller spent amount, a lower fog density and higher transfer efficiency as compared with the developer having no spacer particles produced in Comparative Example 1.
-
The developers produced in Examples 2.2 through 2.4 containing the toner including one kind of desired spacer particles were excellent in the fog density, prevention of the toner scattering, the transfer efficiency and the charge stability. The developer produced in Example 2.1 containing the toner including the two kinds of spacer particles was further improved in the transfer efficiency and the charge stability.
-
Although the developer produced in Example 3.4 including the carrier having no coating layer was excellent in the fog density, the charge amount and prevention of the toner scattering, the developers produced in Examples 3.1 through 3.3 including the carrier having a coating layer had a further smaller spent amount and a further higher durability.
-
Although the developer produced in Example 4.4 including the toner having no stearic acid metallic salt was excellent in the fog density, prevention of the toner scattering, the charge amount and the anti-spent property, the developers produced in Examples 4.1 through 4.3 including the toner having a stearic acid metallic salt were further improved in suppressing appearance of a white dot in a black solid portion.
-
Although the developers produced in Examples 5.4 and 6.4 containing the carrier having no coating layer were excellent in the fog density, the charge amount and prevention of the toner scattering, the developers produced in Examples 5.1 through 5.3 and 6.1 through 6.3 containing the carrier with a coating layer were further improved in the spent amount and the durability.
-
Various other modifications will be apparent to and can be readily made by those skilled in the art without departing from the scope and spirit of this invention. Accordingly, it is not intended that the scope of the claims appended hereto be limited to the description as set forth herein, but rather that the claims be broadly construed.