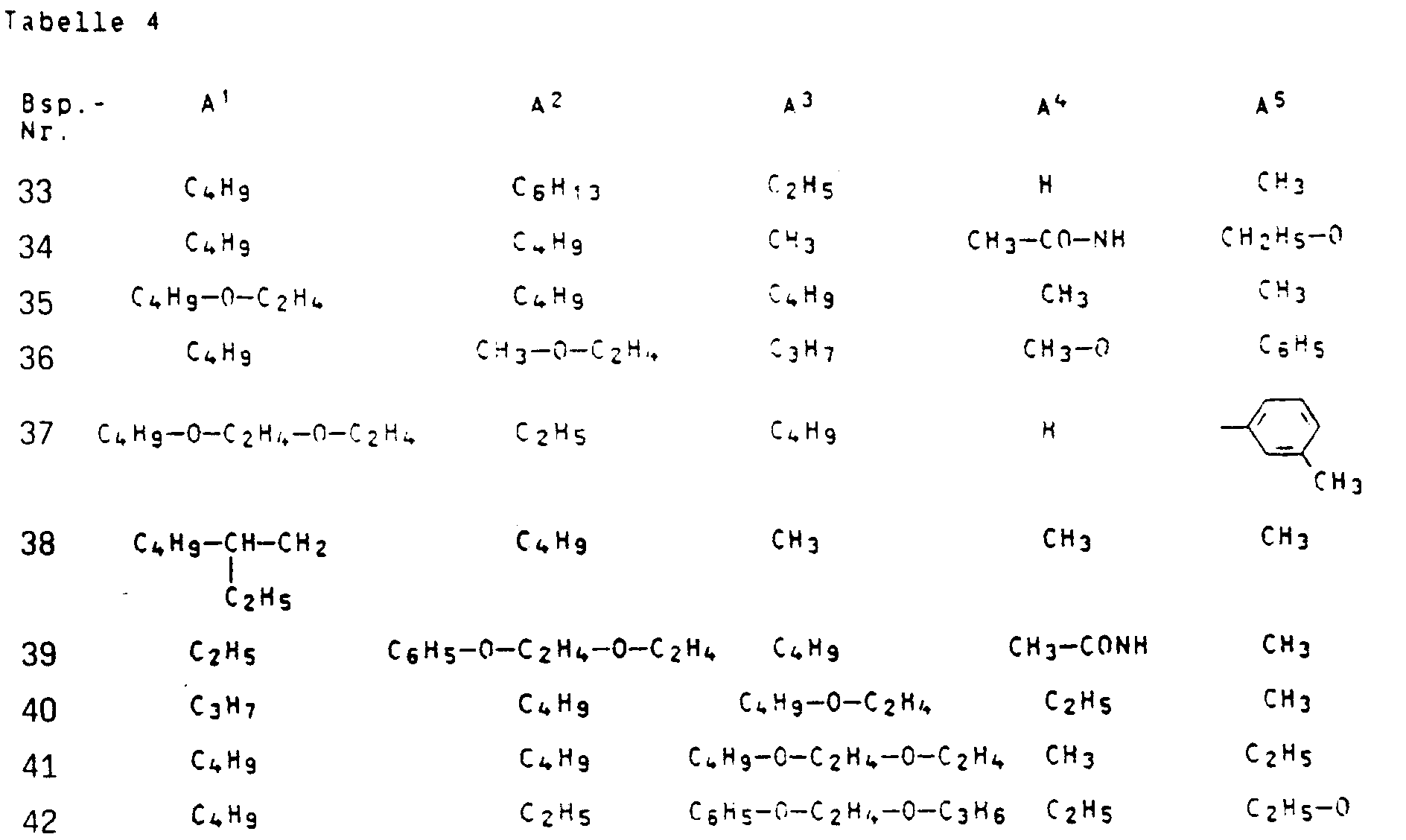EP0344592B2 - Process for azo dye transfer - Google Patents
Process for azo dye transfer Download PDFInfo
- Publication number
- EP0344592B2 EP0344592B2 EP89109327A EP89109327A EP0344592B2 EP 0344592 B2 EP0344592 B2 EP 0344592B2 EP 89109327 A EP89109327 A EP 89109327A EP 89109327 A EP89109327 A EP 89109327A EP 0344592 B2 EP0344592 B2 EP 0344592B2
- Authority
- EP
- European Patent Office
- Prior art keywords
- alkyl
- alkoxy
- phenyl
- cyano
- formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 238000012546 transfer Methods 0.000 title claims description 22
- 238000000034 method Methods 0.000 title claims description 15
- 239000000987 azo dye Substances 0.000 title claims description 13
- 230000008569 process Effects 0.000 title claims description 13
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 27
- -1 cyano, phenyl Chemical group 0.000 claims description 19
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 14
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 14
- 229910052739 hydrogen Inorganic materials 0.000 claims description 14
- 239000001257 hydrogen Substances 0.000 claims description 14
- 125000000217 alkyl group Chemical group 0.000 claims description 13
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 12
- 229910052736 halogen Inorganic materials 0.000 claims description 9
- 150000002367 halogens Chemical class 0.000 claims description 9
- 229910052801 chlorine Inorganic materials 0.000 claims description 8
- 239000000460 chlorine Substances 0.000 claims description 8
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 claims description 7
- 125000001797 benzyl group Chemical class [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 6
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 6
- 125000005041 acyloxyalkyl group Chemical group 0.000 claims description 5
- 125000004432 carbon atom Chemical group C* 0.000 claims description 5
- 125000004400 (C1-C12) alkyl group Chemical group 0.000 claims description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 4
- 125000000027 (C1-C10) alkoxy group Chemical group 0.000 claims description 3
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 claims description 3
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 claims description 3
- 125000005078 alkoxycarbonylalkyl group Chemical group 0.000 claims description 3
- 125000005205 alkoxycarbonyloxyalkyl group Chemical group 0.000 claims description 3
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 3
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 claims description 2
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims description 2
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 2
- 238000009792 diffusion process Methods 0.000 claims description 2
- 229920000642 polymer Polymers 0.000 claims description 2
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims 7
- 125000000051 benzyloxy group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])O* 0.000 claims 1
- 239000000758 substrate Substances 0.000 claims 1
- 238000007651 thermal printing Methods 0.000 claims 1
- 239000000975 dye Substances 0.000 description 40
- 239000011230 binding agent Substances 0.000 description 13
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 8
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 8
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 6
- 239000001856 Ethyl cellulose Substances 0.000 description 6
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 6
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 6
- 229920001249 ethyl cellulose Polymers 0.000 description 6
- 235000019325 ethyl cellulose Nutrition 0.000 description 6
- 150000002431 hydrogen Chemical class 0.000 description 6
- 239000000123 paper Substances 0.000 description 5
- 229920005989 resin Polymers 0.000 description 5
- 239000011347 resin Substances 0.000 description 5
- 125000004642 (C1-C12) alkoxy group Chemical group 0.000 description 4
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 4
- 125000003860 C1-C20 alkoxy group Chemical group 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- 229920000896 Ethulose Polymers 0.000 description 4
- 239000001859 Ethyl hydroxyethyl cellulose Substances 0.000 description 4
- 239000011248 coating agent Substances 0.000 description 4
- 238000000576 coating method Methods 0.000 description 4
- 235000019326 ethyl hydroxyethyl cellulose Nutrition 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- 238000007639 printing Methods 0.000 description 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- 230000008033 biological extinction Effects 0.000 description 3
- 125000000664 diazo group Chemical group [N-]=[N+]=[*] 0.000 description 3
- 230000005012 migration Effects 0.000 description 3
- 238000013508 migration Methods 0.000 description 3
- 239000003960 organic solvent Substances 0.000 description 3
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 3
- 229930192474 thiophene Natural products 0.000 description 3
- 238000010023 transfer printing Methods 0.000 description 3
- 229920002554 vinyl polymer Polymers 0.000 description 3
- VMJNTFXCTXAXTC-UHFFFAOYSA-N 2,2-difluoro-1,3-benzodioxole-5-carbonitrile Chemical group C1=C(C#N)C=C2OC(F)(F)OC2=C1 VMJNTFXCTXAXTC-UHFFFAOYSA-N 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- 241001295925 Gegenes Species 0.000 description 2
- 238000005299 abrasion Methods 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 125000001231 benzoyloxy group Chemical group C(C1=CC=CC=C1)(=O)O* 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 2
- 235000012000 cholesterol Nutrition 0.000 description 2
- 229920001577 copolymer Polymers 0.000 description 2
- ZXEKIIBDNHEJCQ-UHFFFAOYSA-N isobutanol Chemical compound CC(C)CO ZXEKIIBDNHEJCQ-UHFFFAOYSA-N 0.000 description 2
- 229920003023 plastic Polymers 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 239000011092 plastic-coated paper Substances 0.000 description 2
- 229920006267 polyester film Polymers 0.000 description 2
- 239000002861 polymer material Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- 125000004343 1-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- HVYWMOMLDIMFJA-UHFFFAOYSA-N 3-cholesterol Natural products C1C=C2CC(O)CCC2(C)C2C1C1CCC(C(C)CCCC(C)C)C1(C)CC2 HVYWMOMLDIMFJA-UHFFFAOYSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- 241000978776 Senegalia senegal Species 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 238000006887 Ullmann reaction Methods 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 125000001204 arachidyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 239000012876 carrier material Substances 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000008199 coating composition Substances 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000006612 decyloxy group Chemical group 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000004043 dyeing Methods 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 239000011086 glassine Substances 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 125000002960 margaryl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001196 nonadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 239000006259 organic additive Substances 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002958 pentadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000002985 plastic film Substances 0.000 description 1
- 229920006255 plastic film Polymers 0.000 description 1
- 229920003055 poly(ester-imide) Polymers 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 239000004584 polyacrylic acid Substances 0.000 description 1
- 229920006122 polyamide resin Polymers 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920001225 polyester resin Polymers 0.000 description 1
- 239000004645 polyester resin Substances 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 239000004926 polymethyl methacrylate Substances 0.000 description 1
- 229920005749 polyurethane resin Polymers 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 239000011118 polyvinyl acetate Substances 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000005920 sec-butoxy group Chemical group 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 125000002889 tridecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002948 undecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- MWOOGOJBHIARFG-UHFFFAOYSA-N vanillin Chemical compound COC1=CC(C=O)=CC=C1O MWOOGOJBHIARFG-UHFFFAOYSA-N 0.000 description 1
- FGQOOHJZONJGDT-UHFFFAOYSA-N vanillin Natural products COC1=CC(O)=CC(C=O)=C1 FGQOOHJZONJGDT-UHFFFAOYSA-N 0.000 description 1
- 235000012141 vanillin Nutrition 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B41—PRINTING; LINING MACHINES; TYPEWRITERS; STAMPS
- B41M—PRINTING, DUPLICATING, MARKING, OR COPYING PROCESSES; COLOUR PRINTING
- B41M5/00—Duplicating or marking methods; Sheet materials for use therein
- B41M5/26—Thermography ; Marking by high energetic means, e.g. laser otherwise than by burning, and characterised by the material used
- B41M5/382—Contact thermal transfer or sublimation processes
- B41M5/385—Contact thermal transfer or sublimation processes characterised by the transferable dyes or pigments
- B41M5/388—Azo dyes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S428/00—Stock material or miscellaneous articles
- Y10S428/913—Material designed to be responsive to temperature, light, moisture
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S428/00—Stock material or miscellaneous articles
- Y10S428/914—Transfer or decalcomania
Definitions
- the present invention relates to a new process for transferring azo dyes, which have a thiophene-based diazo component, from a support to a plastic-coated paper with the aid of a thermal head.
- a transfer sheet which contains a thermally transferable dye in one or more binders, optionally together with suitable auxiliaries, on a support is heated with a heating head with short heating pulses (duration: fractions of a second) from the back, whereby the dye migrated from the transfer sheet and diffused into the surface coating of a recording medium.
- the main advantage of this method is that it is easy to control the amount of dye to be transferred (and thus the color gradation) by adjusting the energy to be delivered to the heating head.
- EP-A-216 483 and EP-A-258 856 describe azo dyes which have thiophene-based diazo components and aniline-based coupling components.
- disazo dyes based on thiophene and aniline are known from EP-A-218 937 for this purpose.
- the object of the present invention was to provide a process for the transfer of dyes, the dyes being intended to meet the above requirements i) to vii) as well as possible,
- EP-A-302 628 which is state of the art for all designated contracting states in accordance with Art. 54 (3) EPC.
- EP-A-133 011 and EP-A-133 012 deal with the construction of a slave sheet during dye transfer.
- Residues Y in formula I are e.g. Ethylene, 1,2- or 1,3-propylene, 1,2-, 1,3-, 1,4- or 2,3-butylene, pentamethylene, hexamethylene or 2-methylpentamethylene.
- Suitable radicals R 1 , R 2 , R 3 , R 4 and R 6 in formula I are, for example, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl or tert-butyl.
- Residues R 1 , R 2 and R 3 are furthermore, for example, pentyl, isopentyl, neopentyl, tert-pentyl, hexyl, 2-methylpentyl ⁇ heptyl, octyl, 2-ethylhexyl, isooctyl, nonyl, isononyl, decyl or isodecyl.
- R 1 and R 2 are, for example, undecyl, dodecyl. Tridecyl, isotridecyl, tetradecyl, pentadecyl, hexadecyl, heptadecyl, octadecyl, nonadecyl or eicosyl.
- the designations isooctyl, isononyl, isodecyl and isotridecyl are trivial designations and come from the alcohols obtained after oxosynthesis (cf. Ullmanns Enzyklopadie der technical chemistry, 4th edition, volume 7, pages 215 to 217 and volume 11, pages 435 and 436 ).
- R 3 and R 4 are, for example, methoxy, ethoxy, propoxy, isopropoxy, butoxy, isobutoxy or sec-butoxy.
- R 3 radicals are furthermore, for example, pentyloxy, isopentyloxy, neopentyloxy, hexyloxy, heptyloxy, octyloxy, 2-ethylhexyloxy, nonyloxy or decyloxy.
- R 4 radicals are furthermore, for example, methylthio, ethylthio, propylthio, isopropylthio or butylthio.
- R 1 and R 2 are furthermore, for example, benzyl, 1- or 2-phenylethyl,
- a support is used on which there are one or more azo dyes of the formula I in which R 5 is cyano or the radical -CO-OR 1 , in which R 1 is alkyl, alkanoyloxyalkyl or alkyloxycarbonylalkyl, where these radicals can each have up to 12 carbon atoms, or for the radical of the abovementioned formula III, in which n and R 7 each have the abovementioned meaning, or R 7 in particular represents C 1 -C 6 -alkyl.
- the dyes of the formula I are known from EP-A-201 896 or can be obtained by the methods mentioned there.
- the dyes transferred in the process according to the invention are generally distinguished by improved migration properties in the recording medium at room temperature, easier thermal transferability, higher photochemical stability, easier technical accessibility, better resistance to moisture and chemical substances, higher color strength , better solubility and especially from higher color purity.
- the dyes are in a suitable organic solvent, for. B. chlorobenzene, isobutanol, methyl ethyl ketone, methylene chloride, toluene, tetrahydrofuran or mixtures thereof with one or more binders, optionally with the addition of auxiliaries, processed into a printing ink.
- a printing ink contains the dye preferably in a molecularly disperse dissolved form.
- the printing ink is applied to the inert carrier using a doctor blade and the dyeing is air-dried.
- All resins or polymer materials which are soluble in organic solvents and are capable of binding the dye to the inert support in an abrasion-resistant manner are suitable as binders. All resins or polymer materials which are soluble in organic solvents and are capable of binding the dye to the inert support in an abrasion-resistant manner are suitable as binders. Preference is given to binders which, after the printing ink has dried in air, absorb the dye in the form of a clear, transparent film without the dye crystallizing out visibly.
- binders examples include cellulose derivatives, e.g. B. methyl cellulose, ethyl cellulose, ethyl hydroxyethyl cellulose, hydroxypropyl cellulose, cellulose acetate or cellulose acetobutyrate, starch, alginates, alkyl resin, vinyl resin, polyvinyl alcohol, polyvinyl acetate, polyvinyl butyrate or polyvinyl pyrrolidone.
- Polymers and copolymers of acrylates or their derivatives, such as polyacrylic acid, polymethyl methacrylate or styrene acrylate copolymers, polyester resin, polyamide resin, polyurethane resin or natural CH resin, such as gum arabic are also suitable as binders. Further suitable binders are described in DE-A-3 524 519.
- Preferred binders are ethyl cellulose or ethyl hydroxyethyl cellulose of medium to small viscosity settings.
- the ratio of binder to dye preferably varies between 5: 1 and 1: 1.
- auxiliaries such as those specified in EP-A-227 092, EP-A-192 435 or the patent applications cited therein, are also suitable as auxiliaries, in addition, in particular organic additives which cause the transfer dyes to crystallize out during storage and when the ink ribbon is heated prevent. e.g. B. cholesterol or vanillin.
- Inert carriers are e.g. B. tissue, blotting paper or glassine paper or plastic films with good heat resistance, e.g. B. optionally metal-coated polyester, polyamide or polyimide.
- the inert carrier is optionally additionally coated with a slip layer on the side facing the thermal head in order to prevent the thermal head from sticking to the carrier material.
- Suitable lubricants are e.g. B. described in EP-A-216 483 or EP-A-227 095.
- the thickness of the dye carrier is generally 3 to 30 microns, preferably 5 to 10 microns.
- thermostable plastic layers with affinity for the dyes to be transferred can be used as the dye-receiving layer.
- Your glass transition temperature should be below 150 ° C.
- Modified polycarbonates or polyesters may be mentioned as examples.
- Suitable formulations for the receiver layer composition are e.g. B. in EP-A-227 094, EP-A-133 012, EP-A-133 011, EP-A-111 004, JP-A-199 997/1986, JP-A-283 595/1986, JP-A-237 694/1986 or JP-A-127 392/1986 described in detail.
- the transfer takes place by means of a thermal head, which must be able to be heated to a temperature of ⁇ 300 ° C. so that the dye transfer can take place in the time range t: 0 ⁇ t ⁇ 15 msec.
- the dye migrates from the transfer sheet and diffuses into the surface coating of the recording medium.
Landscapes
- Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Thermal Transfer Or Thermal Recording In General (AREA)
- Printing Methods (AREA)
- Coloring (AREA)
- Plural Heterocyclic Compounds (AREA)
Description
Die vorliegende Erfindung betrifft ein neues Verfahren zur Übertragung von Azofarbstoffen, die eine Diazokomponente auf Thiophenbasis aufweisen, von einem Träger auf ein mit Kunststoff beschichtetes Papier mit Hilfe eines Thermokopfes.The present invention relates to a new process for transferring azo dyes, which have a thiophene-based diazo component, from a support to a plastic-coated paper with the aid of a thermal head.
Beim Thermotransferdruckverfahren wird ein Transferblatt, welches einen thermisch transferierbaren Farbstoff in einem oder mehreren Bindemitteln, gegebenenfalls zusammen mit geeigneten Hilfsmitteln, auf einem Träger enthält, mit einem Heizkopf mit kurzen Heizimpulsen (Dauer: Bruchteile einer Sekunde) von der Rückseite her erhitzt, wodurch der Farbstoff aus dem Transferblatt migriert und in die Oberflächenbeschichtung eines Aufnahmemediums hineindiffundiert. Der wesentliche Vorteil dieses Verfahrens besteht darin, daß die Steuerung der zur übertragenden Farbstoffmenge (und damit die Farbabstufung) durch Einstellung der an den Heizkopf abzugebenden Energie leicht möglich ist.In the thermal transfer printing process, a transfer sheet, which contains a thermally transferable dye in one or more binders, optionally together with suitable auxiliaries, on a support is heated with a heating head with short heating pulses (duration: fractions of a second) from the back, whereby the dye migrated from the transfer sheet and diffused into the surface coating of a recording medium. The main advantage of this method is that it is easy to control the amount of dye to be transferred (and thus the color gradation) by adjusting the energy to be delivered to the heating head.
Allgemein wird die Farbaufzeichnung unter Verwendung der drei subtraktiven Grundfarben Gelb, Magenta und Cyan (und gegebenenfalls Schwarz) durchgeführt. Um eine optimale Farbaufzeichnung zu ermöglichen, müssen die Farbstoffe folgende Eigenschaften besitzen:
- i) leichte thermische Transferierbarkeit,
- ii) geringe Migration innerhalb oder aus der Oberflächenbeschichtung des Aufnahmemediums bei Raumtemperatur,
- iii) hohe thermische und photochemische Stabilität sowie Resistenz gegen Feuchtigkeit und chemische Stoffe,
- iv) für substraktive Farbmischung die geeigneten Farbtöne aufweisen,
- v) einen hohen molaren Absorptionskoeffizienten aufweisen,
- vi) bei Lagerung des Transferblattes nicht auskristallisieren,
- vii) technisch leicht zugänglich sein.
- i) easy thermal transferability,
- ii) low migration within or from the surface coating of the recording medium at room temperature,
- iii) high thermal and photochemical stability and resistance to moisture and chemical substances,
- iv) have suitable shades for subtractive color mixing,
- v) have a high molar absorption coefficient,
- vi) do not crystallize out when the transfer sheet is stored,
- vii) be easily accessible technically.
Die Forderungen i), iii), vii), und insbesondere iv) und v) sind erfahrungsgemäß bei den Cyanfarbstoffen besonders schwierig zu erfüllen.Experience has shown that requirements i), iii), vii), and in particular iv) and v), are particularly difficult to meet with cyan dyes.
Daher entsprechen die meisten der bekannten, für den thermischen Transferdruck verwendeten Cyanfarbstoffe nicht dem geforderten Anforderungsprofil.Therefore, most of the known cyan dyes used for thermal transfer printing do not meet the required profile.
Aus dem Stand der Technik sind bereits Farbstoffe bekannt, die in Thermotransferdruckverfahren zur Anwendung kommen. So sind beispielsweise in der EP-A-216 483 und EP-A-258 856 Azofarbstoffe beschrieben, die Diazokomponenten auf Thiophenbasis und Kupplungskomponenten auf Anilinbasis aufweisen.Dyes which are used in thermal transfer printing processes are already known from the prior art. For example, EP-A-216 483 and EP-A-258 856 describe azo dyes which have thiophene-based diazo components and aniline-based coupling components.
Weiterhin sind aus der EP-A- 218 937 für diesen Zweck Disazofarbstoffe auf Thiophen- und Anilinbasis bekannt.Furthermore, disazo dyes based on thiophene and aniline are known from EP-A-218 937 for this purpose.
Aufgabe der vorliegenden Erfindung war es, ein Verfahren zur Übertragung von Farbstoffen bereitzustellen, wobei die Farbstoffe die obengenannten Forderungen i) bis vii) möglichst gut erfüllen sollten,The object of the present invention was to provide a process for the transfer of dyes, the dyes being intended to meet the above requirements i) to vii) as well as possible,
Es wurde nun gefunden, daß die Übertragung von Azofarbstoffen von einem Träger auf ein mit Kunststoff beschichtetes Papier durch Diffusion mit Hilfe eines Thermokopfes vorteilhaft gelingt, wenn man einen Träger verwendet, auf dem sich ein oder mehrere Azofarbstoffe der Formel I
- R1 und R2
- gleich oder verschieden sind und unabhängig voneinander jeweils Alkyl, Alkanoyloxyalkyl, Alkoxycarbonyloxyalkyl oder Alkoxycarbonylalkyl, wobei die Reste jeweils bis zu 20 Kohlenstoffatomen aufweisen und durch Phenyl, C1-C4-Alkylphenyl, C1-C4-Alkoxyphenyl, Benzoyloxy, C1-C4-Alkylbenzyloxy, C1-C4-Alkoxybenzyloxy, Halogen, Hydroxy oder Cyano substituiert sein können, Wasserstoff, gegebenenfalls durch C1-C20-Alkoxy oder Halogen substituiertes Phenyl, gegebenenfalls durch C1-C20-Alkyl. C1-C20-Alkoxy oder Halogen substituiertes Benzyl oder einen Rest der Formel II
- Y
- für C2-C6-Alkylen
- m
- für 1, 2, 3, 4, 5 oder 6 und
- R6
- für C1-C4-Alkyl oder gegebenenfalls durch C1-C4-Alkyl oder C1-C4-Alkoxy substituiertes Phenyl stehen,
- R3
- Wasserstoff, C1-C10-Alkyl, C1-C10-Alkoxy oder den Rest -NH-CO-R1, wobei R1 die obengenannte Bedeutung besitzt,
- R4
- Wasserstoff, Chlor, C1-C4-Alkyl, C1-C4-Alkoxy, C1-C4-Alkylthio oder gegebenenfalls durch C1-C4-Alkyl, C1-C4-Alkoxy oder Halogen substituiertes Phenyl und
- R5
- Cyano oder den Rest -CO-OR1, -CO-NHR1 oder -CO-NR1R2, wobei R1 und R2 jeweils die obengenannte Bedeutung besitzen, bedeuten,
- R 1 and R 2
- are identical or different and are each independently of the other alkyl, alkanoyloxyalkyl, alkoxycarbonyloxyalkyl or alkoxycarbonylalkyl, where the radicals each have up to 20 carbon atoms and are substituted by phenyl, C 1 -C 4 -alkylphenyl, C 1 -C 4 -alkoxyphenyl, benzoyloxy, C 1 - C 4 alkylbenzyloxy, C 1 -C 4 alkoxybenzyloxy, halogen, hydroxy or cyano may be substituted, hydrogen, phenyl optionally substituted by C 1 -C 20 alkoxy or halogen, optionally substituted by C 1 -C 20 alkyl. C 1 -C 20 alkoxy or halogen substituted benzyl or a radical of the formula II
- Y
- for C 2 -C 6 alkylene
- m
- for 1, 2, 3, 4, 5 or 6 and
- R 6
- represent C 1 -C 4 alkyl or phenyl optionally substituted by C 1 -C 4 alkyl or C 1 -C 4 alkoxy,
- R 3
- Hydrogen, C 1 -C 10 alkyl, C 1 -C 10 alkoxy or the radical -NH-CO-R 1 , where R 1 has the meaning given above,
- R 4
- Hydrogen, chlorine, C 1 -C 4 alkyl, C 1 -C 4 alkoxy, C 1 -C 4 alkylthio or phenyl optionally substituted by C 1 -C 4 alkyl, C 1 -C 4 alkoxy or halogen and
- R 5
- Cyano or the radical -CO-OR 1 , -CO-NHR 1 or -CO-NR 1 R 2 , where R 1 and R 2 each have the abovementioned meaning,
Die oben ausgenommenen Verbindungen sind in der EP-A- 302 628 beschrieben, die für alle benannten Vertragsstaaten Stand der Technik gemäß Art. 54 (3) EPÜ ist.The compounds excluded above are described in EP-A-302 628, which is state of the art for all designated contracting states in accordance with Art. 54 (3) EPC.
Die EP-A- 133 011 und die EP-A- 133 012 behandeln den Aufbau eines Nehmerblatts beim Farbstofftransfer.EP-A-133 011 and EP-A-133 012 deal with the construction of a slave sheet during dye transfer.
Alle in der obengenannten Formel I auftretenden Alkylreste können sowohl geradkettig als auch verzweigt sein.All alkyl radicals occurring in the above formula I can be both straight-chain and branched.
Reste Y in Formel I sind z.B. Ethylen, 1,2- oder 1,3-Propylen, 1,2-, 1,3-, 1,4- oder 2,3-Butylen, Pentamethylen, Hexamethylen oder 2-Methylpentamethylen.Residues Y in formula I are e.g. Ethylene, 1,2- or 1,3-propylene, 1,2-, 1,3-, 1,4- or 2,3-butylene, pentamethylene, hexamethylene or 2-methylpentamethylene.
Geeignete Reste R1, R2, R3, R4 und R6 in Formel I sind z.B. Methyl, Ethyl, Propyl, Isopropyl, Butyl, Isobutyl, sec-Butyl oder tert-Butyl.Suitable radicals R 1 , R 2 , R 3 , R 4 and R 6 in formula I are, for example, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl or tert-butyl.
Reste R1, R2 und R3 sind weiterhin z.B. Pentyl, Isopentyl, Neopentyl, tert-Pentyl, Hexyl, 2-Methylpentyl` Heptyl, Octyl, 2-Ethylhexyl, Isooctyl, Nonyl, Isononyl, Decyl oder Isodecyl.Residues R 1 , R 2 and R 3 are furthermore, for example, pentyl, isopentyl, neopentyl, tert-pentyl, hexyl, 2-methylpentyl` heptyl, octyl, 2-ethylhexyl, isooctyl, nonyl, isononyl, decyl or isodecyl.
Reste R1 und R2 sind weiterhin z.B. Undecyl, Dodecyl. Tridecyl, Isotridecyl, Tetradecyl, Pentadecyl, Hexadecyl, Heptadecyl, Octadecyl, Nonadecyl oder Eicosyl. (Die Bezeichnungen Isooctyl, Isononyl, Isodecyl und Isotridecyl sind Trivialbezeichnungen und stammen von den nach der Oxosynthese erhaltenen Alkoholen (vgl. dazu Ullmanns Enzyklopadie der technischen Chemie, 4. Auflage, Band 7, Seiten 215 bis 217 sowie Band 11, Seiten 435 und 436).)R 1 and R 2 are, for example, undecyl, dodecyl. Tridecyl, isotridecyl, tetradecyl, pentadecyl, hexadecyl, heptadecyl, octadecyl, nonadecyl or eicosyl. (The designations isooctyl, isononyl, isodecyl and isotridecyl are trivial designations and come from the alcohols obtained after oxosynthesis (cf. Ullmanns Enzyklopadie der technical chemistry, 4th edition, volume 7, pages 215 to 217 and volume 11, pages 435 and 436 ).)
Reste R3 und R4 sind weiterhin beispielsweise Methoxy, Ethoxy, Propoxy, Isopropoxy, Butoxy, Isobutoxy oder sec-Butoxy.R 3 and R 4 are, for example, methoxy, ethoxy, propoxy, isopropoxy, butoxy, isobutoxy or sec-butoxy.
Reste R3 sind weiterhin z.B. Pentyloxy, Isopentyloxy, Neopentyloxy, Hexyloxy, Heptyloxy, Octyloxy, 2-Ethylhexyloxy, Nonyloxy oder Decyloxy.R 3 radicals are furthermore, for example, pentyloxy, isopentyloxy, neopentyloxy, hexyloxy, heptyloxy, octyloxy, 2-ethylhexyloxy, nonyloxy or decyloxy.
Reste R4 sind weiterhin z.B. Methylthio, Ethylthio, Propylthio, Isopropylthio oder Butylthio.R 4 radicals are furthermore, for example, methylthio, ethylthio, propylthio, isopropylthio or butylthio.
Reste R1 und R2 sind weiterhin z.B. Benzyl, 1- oder 2-Phenylethyl,
Vorzugsweise verwendet man im erfindungsgemäßen Verfahren einen Träger, auf dem sich ein oder mehrere Azofarbstoffe der Formel I befinden, in der
- R1 und R2
- unabhängig voneinander Alkyl, Alkanoloxyalkyl oder Alkyloxycarbonylalkyl, wobei diese Reste jeweils bis zu 12 Kohlenstoffatome aufweisen und durch Phenyl, C1-C4-Alkylphenyl, C1-C4-Alkoxyphenyl, Hydroxy oder Cyano substituiert sein können, gegebenenfalls durch C1-C12-Alkyl oder C1-C12-Alkoxy substituiertes Phenyl, gegebenenfalls durch C1-C12-Alkyl oder C1-C12-Alkoxy substituiertes Benzyl oder einen Rest der Formel II
- Y
- für C2-C4-Alkylen,
- m
- für 1, 2, 3 oder 4 und
- R6
- für C1-C4-Alkyl oder gegebenenfalls durch C1-C4-Alkyl oder C1-C4-Alkoxy substituiertes Phenyl stehen,
- R3
- Wasserstoff, C1-C6-Alkyl, C1-C6-Alkoxy oder den Rest -NH-CO-R1, wobei R1 die zuletztgenannte obige Bedeutung besitzt,
- R4
- Wasserstoff, Chlor, C1-C4-Alkyl, C1-C4-Alkoxy oder Phenyl und
- R5
- Cyano oder den Rest -CO-OR1-CO-NHR1 oder -CO-NR1R2, wobei R1 und R2 jeweils die zuletztgenannte obige Bedeutung besitzen, bedeuten.
- R 1 and R 2
- independently of one another alkyl, alkanoloxyalkyl or alkyloxycarbonylalkyl, where these radicals each have up to 12 carbon atoms and can be substituted by phenyl, C 1 -C 4 -alkylphenyl, C 1 -C 4 -alkoxyphenyl, hydroxy or cyano, optionally by C 1 -C 12 alkyl or C 1 -C 12 alkoxy substituted phenyl, optionally substituted by C 1 -C 12 alkyl or C 1 -C 12 alkoxy benzyl or a radical of formula II
- Y
- for C 2 -C 4 alkylene,
- m
- for 1, 2, 3 or 4 and
- R 6
- represent C 1 -C 4 alkyl or phenyl optionally substituted by C 1 -C 4 alkyl or C 1 -C 4 alkoxy,
- R 3
- Hydrogen, C 1 -C 6 alkyl, C 1 -C 6 alkoxy or the radical -NH-CO-R 1 , where R 1 has the last-mentioned meaning given above,
- R 4
- Hydrogen, chlorine, C 1 -C 4 alkyl, C 1 -C 4 alkoxy or phenyl and
- R 5
- Cyano or the radical -CO-OR 1 -CO-NHR 1 or -CO-NR 1 R 2 , where R 1 and R 2 each have the last-mentioned meaning given above.
Insbesondere verwendet man im neuen Verfahren einen Träger, auf dem sich ein oder mehrere Azofarbstoffe der Formel I befinden, in der
- R1 und R2
- unabhängig voneinander C1-C12-Alkyl, das gegebenenfalls durch Cyano, Phenyl, C1-C4-Alkylphenyl oder C1-C4-Alkoxyphenyl substituiert ist, oder einen Rest der Formel III
- n
- für 1, 2, 3 oder 4 und
- R7
- für C1-C4-Alkyl oder Phenyl stehen,
- R3
- Wasserstoff, Methyl, Methoxy oder Acetylamino,
- R4
- Chlor und
- R5
- den Rest -CO-OR1, -CO-NHR1 oder -CO-NR1 R2, wobei R1 und R2 jeweils die letztgenannte obige Bedeutung besitzen, bedeuten.
- R 1 and R 2
- independently of one another C 1 -C 12 alkyl which is optionally substituted by cyano, phenyl, C 1 -C 4 alkylphenyl or C 1 -C 4 alkoxyphenyl, or a radical of the formula III
- n
- for 1, 2, 3 or 4 and
- R 7
- represent C 1 -C 4 alkyl or phenyl,
- R 3
- Hydrogen, methyl, methoxy or acetylamino,
- R 4
- Chlorine and
- R 5
- the radical -CO-OR 1 , -CO-NHR 1 or -CO-NR 1 R 2 , where R 1 and R 2 each have the latter meaning given above.
Besonders gute Ergebnisse erzielt man, wenn man einen Träger verwendet, auf dem sich ein oder mehrere Azofarbstoffe der Formel I befinden, in der R2 für C1-C6 -Alkyl steht und R1 die zuletztgenannte obige Bedeutung besitzt oder insbesondere ebenfalls C1-C6-Alkyl bedeutet.Particularly good results are obtained if a support is used on which there are one or more azo dyes of the formula I in which R 2 is C 1 -C 6 -alkyl and R 1 has the last-mentioned meaning mentioned above or in particular also C 1 -C 6 alkyl.
Man erzielt weiterhin besonders günstige Ergebnisse, wenn man einen Träger verwendet, auf dem sich ein oder mehrere Azofarbstoffe der Formel I befinden, in der R5 Cyano oder den Rest -CO-OR1 bedeutet, worin R1 für Alkyl, Alkanoyloxyalkyl oder Alkyloxycarbonylalkyl, wobei diese Reste jeweils bis zu 12 Kohlenstoffatomen aufweisen können, oder für den Rest der obengenannten Formel III, in der n und R7 jeweils die obengenannte Bedeutung besitzen, oder R7 insbesondere für C1-C6-Alkyl steht.Particularly favorable results are also achieved if a support is used on which there are one or more azo dyes of the formula I in which R 5 is cyano or the radical -CO-OR 1 , in which R 1 is alkyl, alkanoyloxyalkyl or alkyloxycarbonylalkyl, where these radicals can each have up to 12 carbon atoms, or for the radical of the abovementioned formula III, in which n and R 7 each have the abovementioned meaning, or R 7 in particular represents C 1 -C 6 -alkyl.
Die Farbstoffe der Formel I sind aus der EP-A- 201 896 bekannt oder können nach den dort genannten Methoden erhalten werden.The dyes of the formula I are known from EP-A-201 896 or can be obtained by the methods mentioned there.
Im Vergleich zu den bei den bekannten Verfahren verwendeten Farbstoffen zeichnen sich die beim erfindungsgemäßen Verfahren übertragenen Farbstoffe im allgemeinen durch verbesserte Migrationseigenschaften im Aufnahmemedium bei Raumtemperatur, leichtere thermische Transferierbarkeit, höhere photochemische Stabilität, leichtere technische Zugänglichkeit, bessere Resistenz gegen Feuchtigkeit und chemische Stoffe, höhere Farbstärke, bessere Löslichkeit und insbesondere durch höhere Farbtonreinheit aus.Compared to the dyes used in the known processes, the dyes transferred in the process according to the invention are generally distinguished by improved migration properties in the recording medium at room temperature, easier thermal transferability, higher photochemical stability, easier technical accessibility, better resistance to moisture and chemical substances, higher color strength , better solubility and especially from higher color purity.
Weiterhin ist überraschend, daß die Farbstoffe der Formel I gut transferierbar sind, obwohl sie ein relativ hohes Molekulargewicht besitzen.It is also surprising that the dyes of the formula I are readily transferable, even though they have a relatively high molecular weight.
Zur Herstellung der für das neue Verfahren benötigten Farbstoffträger werden die Farbstoffe in einem geeigneten organischen Lösungsmittel, z. B. Chlorbenzol, Isobutanol, Methylethylketon` Methylenchlorid, Toluol, Tetrahydrofuran oder deren Mischungen mit einem oder mehreren Bindemitteln, gegebenenfalls unter Zugabe von Hilfsmitteln, zu einer Druckfarbe verarbeitet. Diese enthält den Farbstoff vorzugsweise in molekular-dispers gelöster Form. Die Druckfarbe wird mittels einer Rakel auf den inerten Träger aufgetragen und die Färbung an der Luft getrocknet.To prepare the dye carrier required for the new process, the dyes are in a suitable organic solvent, for. B. chlorobenzene, isobutanol, methyl ethyl ketone, methylene chloride, toluene, tetrahydrofuran or mixtures thereof with one or more binders, optionally with the addition of auxiliaries, processed into a printing ink. This contains the dye preferably in a molecularly disperse dissolved form. The printing ink is applied to the inert carrier using a doctor blade and the dyeing is air-dried.
Als Bindemittel kommen alle Resins oder Polymermaterialien in Betracht, welche in organischen Lösungsmitteln löslich sind und den Farbstoff an den inerten Träger abriebfest zu binden vermögen. Dabei werden solcheAls Bindemittel kommen alle Resins oder Polymermaterialien in Betracht, welche in organischen Lösungsmitteln löslich sind und den Farbstoff an den inerten Träger abriebfest zu binden vermögen. Dabei werden solche Bindemittel bevorzugt, welche den Farbstoff nach Trocknung der Druckfarbe an der Luft in Form eines klaren, transparenten Films aufnehmen, ohne daß dabei eine sichtbare Auskristallisation des Farbstoffes auftritt.All resins or polymer materials which are soluble in organic solvents and are capable of binding the dye to the inert support in an abrasion-resistant manner are suitable as binders. All resins or polymer materials which are soluble in organic solvents and are capable of binding the dye to the inert support in an abrasion-resistant manner are suitable as binders. Preference is given to binders which, after the printing ink has dried in air, absorb the dye in the form of a clear, transparent film without the dye crystallizing out visibly.
Beispiele für solche Bindemittel sind Cellulosederivate, z. B. Methylcellulose, Ethylcellulose, Ethylhydroxyethylcellulose, Hydroxypropylcellulose, Celluloseacetat oder Celluloseacetobutyrat, Stärke, Alginate, Alkylresins, Vinylresins, Polyvinylalkohol, Polyvinylacetat, Polyvinylbutyrat oder Polyvinylpyrrolidone. Weiterhin kommen Polymere und Copolymere von Acrylaten oder deren Derivate, wie Polyacrylsäure, Polymethylmethacrylat oder Styrolacrylatcopolymere, Polyesterresins, Polyamidresins, Polyurethanresins oder natürliche CH-Resins, wie Gummi Arabicum, als Bindemittel in Betracht. Weitere geeignete Bindemittel sind in der DE-A- 3 524 519 beschrieben.Examples of such binders are cellulose derivatives, e.g. B. methyl cellulose, ethyl cellulose, ethyl hydroxyethyl cellulose, hydroxypropyl cellulose, cellulose acetate or cellulose acetobutyrate, starch, alginates, alkyl resin, vinyl resin, polyvinyl alcohol, polyvinyl acetate, polyvinyl butyrate or polyvinyl pyrrolidone. Polymers and copolymers of acrylates or their derivatives, such as polyacrylic acid, polymethyl methacrylate or styrene acrylate copolymers, polyester resin, polyamide resin, polyurethane resin or natural CH resin, such as gum arabic, are also suitable as binders. Further suitable binders are described in DE-A-3 524 519.
Bevorzugte Bindemittel sind Ethylcellulose oder Ethylhydroxyethylcellulose mittlerer bis kleiner Viskositätseinstellungen.Preferred binders are ethyl cellulose or ethyl hydroxyethyl cellulose of medium to small viscosity settings.
Das Verhältnis Bindemittel zu Farbstoff variiert vorzugsweise zwischen 5:1 und 1:1.The ratio of binder to dye preferably varies between 5: 1 and 1: 1.
Als Hilfsmittel kommen Trennmittel in Betracht, wie sie in der EP-A-227 092, EP-A-192 435 oder den dort zitierten Patentanmeldungen spezifiziert sind, darüber hinaus besonders organische Additive, welche das Auskristallisieren der Transferfarbstoffe bei Lagerung und beim Erhitzen des Farbbandes verhindern. z. B. Chlolesterin oder Vanillin.Auxiliaries, such as those specified in EP-A-227 092, EP-A-192 435 or the patent applications cited therein, are also suitable as auxiliaries, in addition, in particular organic additives which cause the transfer dyes to crystallize out during storage and when the ink ribbon is heated prevent. e.g. B. cholesterol or vanillin.
Inerte Träger sind z. B. Seiden-, Lösch- oder Pergaminpapier oder Kunststoffolien mit guter Wärmebeständigkeit, z. B. gegebenenfalls metallbeschichteter Polyester, Polyamid oder Polyimid. Der inerte Träger wird auf der dem Thermokopf zugewandten Seite gegebenenfalls zusätzlich mit einer Gleitmittelschicht (Slipping layer) beschichtet, um ein Verkleben des Thermokopfes mit dem Trägermaterial zu verhindern. Geeignete Gleitmittel werden z. B. in der EP-A-216 483 oder EP-A-227 095 beschrieben. Die Dicke des Farbstoff-Trägers beträgt im allgemeinen 3 bis 30 µm, vorzugsweise 5 bis 10 µm.Inert carriers are e.g. B. tissue, blotting paper or glassine paper or plastic films with good heat resistance, e.g. B. optionally metal-coated polyester, polyamide or polyimide. The inert carrier is optionally additionally coated with a slip layer on the side facing the thermal head in order to prevent the thermal head from sticking to the carrier material. Suitable lubricants are e.g. B. described in EP-A-216 483 or EP-A-227 095. The thickness of the dye carrier is generally 3 to 30 microns, preferably 5 to 10 microns.
Als Farbstoffnehmerschicht kommen prinzipiell alle temperaturstabilen Kunststoffschichten mit Affinität zu den zu transferierenden Farbstoffen in Betracht. Ihre Glasumwandlungstemperatur sollte unter 150° C liegen. Beispielhaft sind modifizierte Polycarbonate oder Polyester zu nennen. Geeignete Rezepturen für die Nehmerschichtzusammensetzung werden z. B. in der EP-A-227 094, EP-A-133 012, EP-A-133 011, EP-A-111 004, JP-A- 199 997/1986, JP-A- 283 595/1986, JP-A- 237 694/1986 oder JP-A- 127 392/1986 ausführlich beschrieben.In principle, all temperature-stable plastic layers with affinity for the dyes to be transferred can be used as the dye-receiving layer. Your glass transition temperature should be below 150 ° C. Modified polycarbonates or polyesters may be mentioned as examples. Suitable formulations for the receiver layer composition are e.g. B. in EP-A-227 094, EP-A-133 012, EP-A-133 011, EP-A-111 004, JP-A-199 997/1986, JP-A-283 595/1986, JP-A-237 694/1986 or JP-A-127 392/1986 described in detail.
Die Übertragung erfolgt mittels eines Thermokopfes, der auf eine Temperatur von ≧ 300 ° C aufheizbar sein muß, damit der Farbstofftransfer im Zeitbereich t: 0 < t < 15 msec erfolgen kann. Dabei migriert der Farbstoff aus dem Transferblatt und diffundiert in die Oberflächenbeschichtung des Aufnahmemediums.The transfer takes place by means of a thermal head, which must be able to be heated to a temperature of ≧ 300 ° C. so that the dye transfer can take place in the time range t: 0 <t <15 msec. The dye migrates from the transfer sheet and diffuses into the surface coating of the recording medium.
Einzelheiten der Herstellung können den Beispielen entnommen werden, in denen sich Angaben über Prozente, sofern nicht anders vermerkt, auf das Gewicht beziehen.Details of the production can be found in the examples, in which information on percentages, unless stated otherwise, relates to the weight.
Um das Transferverhalten der Farbstoffe quantitativ und in einfacher Weise prüfen zu können, wurde der Thermotransfer mit großflächigen Heizbacken statt eines Thermokopfes durchgeführt, wobei die Transfertemperatur im Bereich 70 ° C < T < 120 ° C variierte und die Transferzeit auf 2 Minuten festgelegt wurde.
- A) Allgemeines Rezept für die Beschichtung der Träger mit Farbstoff:
1 g Bindemittel wurde in 8 ml Toluol/Ethanol (8:2 v/v) bei 40 bis 50 ° C gelöst. Dazu wurde eine Lösung aus 0,25 g Farbstoff (und gegebenenfalls Hilfsmittel) in 5 ml Tetrahydrofuran eingerührt. Die so erhaltene Druckpaste wurde mit einer 80 µm Rakel auf eine Polyesterfolie (Dicke: 6 bis 10 µm) abgezogen und mit einem Fön getrocknet. - B) Prüfung auf thermische Transferierbarkeit
Die verwendeten Farbstoffe wurden in der folgenden Weise geprüft:
Die den zu prüfenden Farbstoff in der Beschichtungsmasse (Vorderseite) enthaltende Polyesterfolie (Geber) wurde mit der Vorderseite auf kommerziell erhältliches Hitachi Color Video Print Paper (Nehmer) gelegt und aufgedrückt. Geber/Nehmer wurden dann mit Aluminiumfolie umwickelt und zwischen zwei beheizten Platten bei verschiedener Temperatur T (im Temperaturintervall 70° C < T < 120° C) erhitzt. Die in die glänzende Kunststoffschicht des Nehmens diffundierte Farbstoffmenge ist proportional der optischen Dichte (= Extinktion A). Letztere wurde photometrisch bestimmt. Trägt man den Logarithmus der im Temperaturintervall zwischen 80 und 110° C gemessenen Extinktion A der angefärbten Nehmerpapiere gegen die zugehörige reziproke absolute Temperatur auf, so erhält man Geraden, aus deren Steigung die Aktivierungsenergie ΔET für das Transferexperiment berechnet wird:
- A) General recipe for coating the carrier with dye:
1 g of binder was dissolved in 8 ml of toluene / ethanol (8: 2 v / v) at 40 to 50 ° C. For this purpose, a solution of 0.25 g of dye (and, if appropriate, auxiliary) was stirred into 5 ml of tetrahydrofuran. The printing paste thus obtained was drawn off onto a polyester film (thickness: 6 to 10 μm) using an 80 μm doctor blade and dried using a hair dryer. - B) Testing for thermal transferability
The dyes used were tested in the following way:
The polyester film (donor) containing the dye to be tested in the coating composition (front side) was placed with the front side on commercially available Hitachi Color Video Print Paper (receiver) and pressed on. The encoder / receiver was then wrapped with aluminum foil and heated between two heated plates at different temperatures T (in the temperature interval 70 ° C <T <120 ° C). The amount of dye diffused into the shiny plastic layer of the take is proportional to the optical density (= extinction A). The latter was determined photometrically. If one plots the logarithm of the extinction A of the colored receiving papers measured in the temperature interval between 80 and 110 ° C against the associated reciprocal absolute temperature, straight lines are obtained, from the slope of which the activation energy ΔE T for the transfer experiment is calculated:
Die in folgenden Tabellen genannten Farbstoffe wurden nach A) verarbeitet und die erhaltenen, mit Farbstoff beschichteten Träger nach B) auf das Transferverhalten geprüft. In der Tabelle sind jeweils die Thermotransferparameter T* und ΔET, die Absorptionsmaxima der Farbstoffe λmax (gemessen in Methylenchlorid), die verwendeten Bindemittel und die Hilfsmittel aufgeführt.The dyes mentioned in the following tables were processed according to A) and the resulting dye-coated supports according to B) were tested for the transfer behavior. In the table are the Thermal transfer parameters T * and ΔE T , the absorption maxima of the dyes λ max (measured in methylene chloride), the binders used and the auxiliaries are listed.
Dabei gelten folgende Abkürzungen:
- B =
- Bindemittel (EC = Ethylcellulose, EHEC = Ethylhydroxyethylcellulose,
- MS =
- Mischung aus Polyvinylbutyrat und Ethylcellulose im Gewichtsverhältnis 2:1)
- F =
- Farbstoff
- HM =
- Hilfsmittel (Chol = Cholesterin)
- B =
- Binder (EC = ethyl cellulose, EHEC = ethyl hydroxyethyl cellulose,
- MS =
- Mixture of polyvinyl butyrate and ethyl cellulose in a weight ratio of 2: 1)
- F =
- dye
- HM =
- Auxiliaries (Chol = cholesterol)
In analoger Weise können die In der folgenden Tabelle 4 aufgeführten Farbstoffe der Formel
Claims (3)
- A process for transferring azo dyes from a substrate to a polymer-coated paper by diffusion with the aid of a thermal printing head, which comprises using a transfer comprising one or more azo dyes of the formula I
- A process as claimed in claim 1, wherein the transfer used comprises one or more azo dyes of the formula I whereR1 and R2 are each independently of the other alkyl, alkanoyloxyalkyl or alkyloxycarbonylalkyl, each of which may have up to 12 carbon atoms and be substituted by phenyl, C1-C4-alkylphenyl, C1-C4-alkoxyphenyl, hydroxyl or cyano, or are each independently of the other unsubstituted or C1-C12-alkyl- or C1-C12-alkoxy-substituted phenyl, unsubstituted or C1-C12-alkyl- or C1-C12-alkoxy-substituted benzyl or a radical of the formula II
- A process as claimed in claim 1, wherein the transfer used comprises one or more azo dyes of the formula I whereR1 and R2 are each independently of the other C1-C12-alkyl which may be substituted by cyano, phenyl, C1-C4-alkylphenyl or C1-C4-alkoxyphenyl, or a radical of the formula III
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE3818404 | 1988-05-31 | ||
| DE3818404A DE3818404A1 (en) | 1988-05-31 | 1988-05-31 | METHOD FOR TRANSMITTING AZO DYES |
Publications (4)
| Publication Number | Publication Date |
|---|---|
| EP0344592A2 EP0344592A2 (en) | 1989-12-06 |
| EP0344592A3 EP0344592A3 (en) | 1990-04-04 |
| EP0344592B1 EP0344592B1 (en) | 1993-09-01 |
| EP0344592B2 true EP0344592B2 (en) | 1997-10-15 |
Family
ID=6355459
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP89109327A Expired - Lifetime EP0344592B2 (en) | 1988-05-31 | 1989-05-24 | Process for azo dye transfer |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US5037798A (en) |
| EP (1) | EP0344592B2 (en) |
| JP (1) | JP2746656B2 (en) |
| DE (2) | DE3818404A1 (en) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE4004600A1 (en) * | 1990-02-15 | 1991-08-22 | Basf Ag | METHOD FOR TRANSMITTING AZO DYES |
| DE4132074A1 (en) * | 1991-09-26 | 1993-04-01 | Basf Ag | AZO DYES WITH A CHINOLINE SERIES CLUTCH COMPONENT |
| US5518983A (en) * | 1992-10-21 | 1996-05-21 | Imperial Chemical Industries Plc | Dye diffusion thermal transfer printing |
| EP0665117A1 (en) * | 1994-01-31 | 1995-08-02 | Agfa-Gevaert N.V. | Thermal dye transfer image with improved light-fastness |
| US7518715B1 (en) * | 2008-06-24 | 2009-04-14 | International Business Machines Corporation | Method for determination of efficient lighting use |
| US8274649B2 (en) | 2008-06-24 | 2012-09-25 | International Business Machines Corporation | Failure detection in lighting systems |
| CN109574880B (en) * | 2017-09-29 | 2022-06-17 | 纳莹(上海)生物科技有限公司 | Fluorescent probe and preparation method and application thereof |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS58215397A (en) * | 1982-06-08 | 1983-12-14 | Sony Corp | Volatile coloring matter composition |
| DE3427200A1 (en) * | 1984-07-24 | 1986-01-30 | Basf Ag, 6700 Ludwigshafen | METHINE DYES |
| JPS61127392A (en) * | 1984-11-28 | 1986-06-14 | Matsushita Electric Ind Co Ltd | Image receiver for sublimable transfer |
| JPH0725218B2 (en) * | 1985-04-15 | 1995-03-22 | 大日本印刷株式会社 | Heat transfer sheet |
| IN167384B (en) * | 1985-05-14 | 1990-10-20 | Basf Ag | |
| JPH0714665B2 (en) * | 1985-06-10 | 1995-02-22 | 大日本印刷株式会社 | Heat transfer sheet |
| DE3630279A1 (en) * | 1986-09-05 | 1988-03-17 | Basf Ag | METHOD FOR TRANSMITTING DYES |
| GB8718431D0 (en) * | 1987-08-04 | 1987-09-09 | Ici Plc | Thermal transfer printing |
-
1988
- 1988-05-31 DE DE3818404A patent/DE3818404A1/en not_active Withdrawn
-
1989
- 1989-05-24 DE DE89109327T patent/DE58905420D1/en not_active Expired - Lifetime
- 1989-05-24 EP EP89109327A patent/EP0344592B2/en not_active Expired - Lifetime
- 1989-05-30 US US07/358,142 patent/US5037798A/en not_active Expired - Fee Related
- 1989-05-30 JP JP1134837A patent/JP2746656B2/en not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| US5037798A (en) | 1991-08-06 |
| EP0344592A2 (en) | 1989-12-06 |
| DE58905420D1 (en) | 1993-10-07 |
| DE3818404A1 (en) | 1989-12-07 |
| JP2746656B2 (en) | 1998-05-06 |
| EP0344592A3 (en) | 1990-04-04 |
| EP0344592B1 (en) | 1993-09-01 |
| JPH0225384A (en) | 1990-01-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0416434B1 (en) | Triazole pyridine dyes and a thermal transfer process of methine dyes | |
| EP0346729B1 (en) | Azo dye transfer process with a pyridine coupling component | |
| EP0441282B1 (en) | Bichromophore methine and azamethine dyestuffs and a process for their transfer | |
| EP0415203B1 (en) | Thiazole dyes of the merocyanine type and a thermal transfer process using these dyes | |
| EP0439200B1 (en) | Transfer process using methine dyes containing bichromophoric cyano-groups | |
| EP0344592B2 (en) | Process for azo dye transfer | |
| EP0442360B1 (en) | Process for transfer of azo dyes | |
| EP0437282B1 (en) | Bichromophore methine dyes containing cyano groups and a process for their transfer | |
| EP0441208B1 (en) | Use of azo-dyes for thermal transfer printing | |
| EP0479076B1 (en) | Process for transfer of indoaniline dyestuffs | |
| EP0413229B1 (en) | Phenonazo compounds and process for the thermal transfer of these dyes | |
| DE3812053A1 (en) | METHOD FOR TRANSMITTING DYES | |
| EP0509302B1 (en) | Process for transfer of methine dyes | |
| EP0480278B1 (en) | Use of anthraquinone dyestuffs for thermal transfer printing | |
| EP0479068B1 (en) | Quinoline methin dyes and process for their thermal transfer | |
| EP0490225B1 (en) | Use of anthraquinone dyerstuffs for thermal transfer printing | |
| EP0420036B1 (en) | Use of azo dyes in thermal transfer printing | |
| EP0567829B1 (en) | Process for transfer of anthraquinone dyes | |
| DE4010269A1 (en) | INDONAPHTHOL DYES AND A METHOD FOR THEIR THERMAL TRANSFER | |
| DE4019419A1 (en) | USE OF AZO DYES FOR THERMAL TRANSFER PRINT |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): CH DE FR GB IT LI |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): CH DE FR GB IT LI |
|
| 17P | Request for examination filed |
Effective date: 19900220 |
|
| RHK1 | Main classification (correction) |
Ipc: B41M 5/38 |
|
| 17Q | First examination report despatched |
Effective date: 19911220 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): CH DE FR GB IT LI |
|
| REF | Corresponds to: |
Ref document number: 58905420 Country of ref document: DE Date of ref document: 19931007 |
|
| ITF | It: translation for a ep patent filed | ||
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19930920 |
|
| ET | Fr: translation filed | ||
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| 26 | Opposition filed |
Opponent name: ZENECA LIMITED Effective date: 19940526 |
|
| PLAB | Opposition data, opponent's data or that of the opponent's representative modified |
Free format text: ORIGINAL CODE: 0009299OPPO |
|
| R26 | Opposition filed (corrected) |
Opponent name: ZENECA LIMITED Effective date: 19940526 |
|
| PLBQ | Unpublished change to opponent data |
Free format text: ORIGINAL CODE: EPIDOS OPPO |
|
| PLAB | Opposition data, opponent's data or that of the opponent's representative modified |
Free format text: ORIGINAL CODE: 0009299OPPO |
|
| R26 | Opposition filed (corrected) |
Opponent name: ZENECA LIMITED Effective date: 19940526 |
|
| PLAW | Interlocutory decision in opposition |
Free format text: ORIGINAL CODE: EPIDOS IDOP |
|
| PLAW | Interlocutory decision in opposition |
Free format text: ORIGINAL CODE: EPIDOS IDOP |
|
| PUAH | Patent maintained in amended form |
Free format text: ORIGINAL CODE: 0009272 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT MAINTAINED AS AMENDED |
|
| 27A | Patent maintained in amended form |
Effective date: 19971015 |
|
| AK | Designated contracting states |
Kind code of ref document: B2 Designated state(s): CH DE FR GB IT LI |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: AEN Free format text: AUFRECHTERHALTUNG DES PATENTES IN GEAENDERTER FORM |
|
| GBTA | Gb: translation of amended ep patent filed (gb section 77(6)(b)/1977) | ||
| ITF | It: translation for a ep patent filed | ||
| ET3 | Fr: translation filed ** decision concerning opposition | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20010423 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20010425 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20010517 Year of fee payment: 13 Ref country code: DE Payment date: 20010517 Year of fee payment: 13 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF THE APPLICANT RENOUNCES Effective date: 20020518 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020524 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020531 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020531 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20020524 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20030131 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20050524 |