EP0029418B1 - A method of acid pickling iron and iron alloys and a composition for carrying out the method - Google Patents
A method of acid pickling iron and iron alloys and a composition for carrying out the method Download PDFInfo
- Publication number
- EP0029418B1 EP0029418B1 EP80850166A EP80850166A EP0029418B1 EP 0029418 B1 EP0029418 B1 EP 0029418B1 EP 80850166 A EP80850166 A EP 80850166A EP 80850166 A EP80850166 A EP 80850166A EP 0029418 B1 EP0029418 B1 EP 0029418B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- acid
- pickling
- phosphoric acid
- mole per
- mol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23G—CLEANING OR DE-GREASING OF METALLIC MATERIAL BY CHEMICAL METHODS OTHER THAN ELECTROLYSIS
- C23G1/00—Cleaning or pickling metallic material with solutions or molten salts
- C23G1/02—Cleaning or pickling metallic material with solutions or molten salts with acid solutions
- C23G1/08—Iron or steel
- C23G1/083—Iron or steel solutions containing H3PO4
Definitions
- the present invention relates to a method of acid pickling metallic material having an iron content of at least about 80 per cent by weight, while simultaneously inhibiting exposed metal surfaces against acid attack.
- the invention also relates to a pickling composition for carrying out the method containing phosphoric acid and one or both of the mineral acids hydrochloric acid and sulphuric acid and having a total acid concentration 0.5-2 mol/I.
- Iron and steel works and manufacturing industries use acid pickling processes in the surface treatment of articles of iron, steel and other iron alloys.
- pickling is primarily carried out to remove scale and other oxide and hydroxide layers which have formed on the metal surfaces of the articles during production. These layers are formed primarily when the articles are treated thermally, e.g. when annealed and subsequently rolled.
- pickling processes are applied for the purpose of removing rust and other impurities from the surface of the material.
- Rust is formed during the transportation, storage and handling of the metal articles, these articles comprising finished goods from the steel industry or intermediate products from other metal working industries, e.g. from sub-contractors.
- the impurities originate from the same sources and may comprise, for instance, welding, soldering or brazing residues and handprint, although they may also be in the form of oil coatings and the like from processing operations or oil which has been purposely applied to protect the material against corrosion.
- the acid solution used to pickle non-alloyed steels or low-alloy steels is mostly a sulphuric acid solution or a hydrochloric acid solution.
- the acid solution used is mostly a nitric acid solution or a hydrofluoric acid solution, or a mixture of these two acids.
- the acid used for pickling purposes in the manufacturing industry is predominantly sulphuric acid, although hydrochloric acid, nitric acid and hydrofluoric acid may also be used when considered an advantage in view of prevailing circumstances.
- Phosphating - a chemical pre-treatment process in which there is preferably used solutions whose major component is either iron phosphate or zinc phosphate - is often used, however, to obtain a short time corrosion protection prior to finally treating an article, and to provide better adhesion surfaces for paints and varnishes applied to the article, thereby to improve the anti-rust properties of the finished surfaces.
- the pickling time for removing rust is normally from 5 to 20 minutes.
- a common feature of all pickling processes is that as soon as the base metal is exposed, it is attacked by the acid and dissolves, resulting in an unnecessary loss of metal. To avoid this there is normally added an organic inhibitor, which is adsorbed on the exposed surfaces of the metal to block the same, thereby protecting said surfaces against acid attack.
- phosphoric acid primarily for special pickling processes, for intensified pickling processes and for pickling special metals, where the disadvantages associated with the use of phosphoric acid as a pickling agent can be accepted in view of the special application.
- SU-A-152603 teaches the use of an acid pickling solution comprising a mixture of phosphoric acid and hydrochloric acid for the precision- pickling of iron metals, whereat the phosphoric acid content is 50 per cent by weight or more of the total acid content. It is proposed to recover the phosphoric acid content with the use of ion- exchanging resins, which is, of course, a very expensive method.
- US-A-1 279 331 suggests the use of a mixture of phosphoric acid and sulphuric acid when pickling iron and steel.
- the phosphoric acid content is more than 50 mole per cent by weight of the total acid content.
- the stated purpose of the phosphoric acid addition is to avoid subsequent attack by rust.
- the FR-A-2208006 also proposes a pickling solution containing phosphoric acid together with hydrochloric acid and sulphuric acid, whereat contents of 1-99 mole per cent by weight phosphoric acid, 5-99 mole per cent by weight sulphuric acid, and 5-99 mole per cent by weight hydrochloric acid are proposed as a pickling agent for stainless steel, and particularly as an agent to passivate such steels. Also JP-A- No. 53-138928 discloses stainless steel pickling compositions containing phosphoric acid.
- the admixture of minor quantities of phosphoric acid with pure or mixed concentrated mineral acids, such as sulphuric acid, hydrochloric acid and nitric acid, has also been proposed.
- the phosphoric acid is said to have a passivating effect on the acid, and its use is proposed in connection with acids which remain in contact with metal surfaces over very long periods of time.
- the method can be considered as a phosphating method in situ.
- a method for pickling soft carbon steel it is proposed in DE-B-1 169251 that the steel is treated with a solution containing sulphuric acid and/or phosphoric acid and hydrogen peroxide.
- the phosphoric acid content is said to be 80-95 per cent by weight of the total acid content.
- Phosphoric acid additions as pickling agents are also proposed in GB-A-854588, whereat the pickling agent, which may be in liquid or paste form, also includes sulphuric acid, hydrochloric acid, oxalic acid or an oxalic acid salt, a chromium salt and a wetting agent.
- the pickling agent which may be in liquid or paste form, also includes sulphuric acid, hydrochloric acid, oxalic acid or an oxalic acid salt, a chromium salt and a wetting agent.
- the method is considered particularly suitable for cleaning metal surfaces which are to be painted. In such applications factors concerning pickling times and the dissolving of metals are not important and have not been discussed in the publication.
- FR-A-1236268 a mixture of hydrochloric acid, phosphoric acid and sodium borate can be used for rapidly pickling iron metals.
- the pickling time is relatively long and the phosphoric acid addition relatively high.
- CH-A-134632 it has also been proposed to use phosphoric acid as a pickling agent at elevated temperatures and pressures, in order to achieve satisfactory pickling times and results.
- the use of phosphoric acid as a pickling agent either alone or in combination with other pickling acids, is well known to the art. It will be noted however, that in all cases the acid has been used despite those problems which it can cause.
- phosphoric acid as a pickling agent can be considered known in extreme cases as an extreme measure, where extreme requirements are placed on the appearance of the surfaces of a workpiece subsequent to pickling and treating the same, and where a residual protective surface is required (phosphating effect).
- the correct pickling time is chosen. If the time chosen is too short, the result may be an incompletely cleansed surface, which may provide a product of poorer quality and render the surface more liable to corrosion later on.
- the pickling time chosen is, on the other hand, too long, the amount of acid and steel consumed is greater than necessary. Moreover, in this latter case the structure of the steel surfaces becomes coarser, the useful lifetime of the bath decreases, and the load on the surroundings resulting from the vapours given off from the bath increases.
- the pickling process could be made less sensitive to variations in pickling times.
- the acid By controlling the process, it would be possible to utilize the acid more effectively, to reduce the energy consumed, to produce a product of more uniform quality, and to reduce the number of interruptions in production caused by bath changes.
- the pickling process can be divided into two stages. In the first stage, a major part of the scale present on the surfaces of the articles is quickly dissolved. The remainder of the scale is dissolved in the second stage, while the dominating part of the process is an attack on the cleansed metal surfaces exposed by the removal of the scale.
- the two stages in the pickling process can be separated specifically, if the decrease in weight per unit of time is determined as a function of the pickling time. During the second stage, it is also possible to observe the generation of hydrogen gas in a quantity corresponding to the amount of pure metal dissolved.
- pickling processes using hydrochloric acid are effected at lower temperatures than those using sulphuric acid.
- scale and pure metal are dissolved more rapidly than in sulphuric acid.
- the pickling rate in a hydrochloric bath is very high, especially when Fe 2+ ions are already found in the bath at the commencement of the process.
- the said second stage of the process is also effected much more slowly in phosphoric acid, which can be attributed to the formation of iron phosphate on the exposed metal surfaces.
- Phosphoric acid differs advantageously from both sulphuric acid and hydrochloric acid in this way.
- a prime object of the present invention is to provide a method of overcoming the aforementioned disadvantages while substantially fulfilling the aforeindicated desiderata. It has been found possible to achieve this object by using a pickling bath comprising novel, specific combinations of pickling acids.
- Another object of the invention is to provide a method in which the oxide layer, in the form of scale on rust, partially or completely covering the surfaces of the article, is rapidly dissolved.
- a further object of the invention is to reduce the extent to which metal is undesirably dissolved with extensive pickling times. This can be achieved by blocking the pure metal surfaces against the attack of hydrogen ions in the acid solution.
- Yet another object is to facilitate further surface treatment of the articles in conjunction with the pickling process, so that said surfaces can, for example, be pre-treated before applying thereto, e.g. a coating of paint or varnish, or some other layer forming organic substance.
- the present invention which relates to a method and an improved pickling agent for pickling articles of iron and iron alloys of before mentioned kind.
- the method of the invention is characterized in that said material is brought into contact with an aqueous solution containing phosphoric acid and one or both of the mineral acids hydrochloric acid and sulphuric acid, whereat the amount of phosphoric acid in the solution is adjusted to at least 0.1 mol/I and at most 1 mol/I, and the total acid concentration is 0.5-2 mol/I.
- the agent for carrying out the method is characterized in that it includes, as an inhibitor, phosphoric acid in an amount of at least 0.01 mol/I and at most 1 mol/I.
- the lower limit for the phosphoric acid content in the pickling solution is 0.01 mol/I. If the content is not maintained in the solution a substantial greater attack on the base metal will occur, which is not desirable. When the phosphoric acid content exceeds 1 mol/I the scale dissolution on the other hand will be so slow that the pickling time would have to be extended to an essential extent. This will decrease the pickling capacity and thus also the pickling economy. It is also essential in the method that the total acid concentration is controlled to be within the range of 0.5-2 mol/I. A lower total acid concentration will also give unacceptable long pickling times which results in serious disadvantages of beforementioned type.
- the rate at which the metal is dissolved is low and remains unchanged when the phosphoric acid content is at least 0.2 M, corresponding to 10 mole per cent of the total acid present. When the phosphoric acid content falls below this level, the rate at which the metal dissolves increases.
Abstract
Description
- The present invention relates to a method of acid pickling metallic material having an iron content of at least about 80 per cent by weight, while simultaneously inhibiting exposed metal surfaces against acid attack. The invention also relates to a pickling composition for carrying out the method containing phosphoric acid and one or both of the mineral acids hydrochloric acid and sulphuric acid and having a total acid concentration 0.5-2 mol/I.
- Iron and steel works and manufacturing industries use acid pickling processes in the surface treatment of articles of iron, steel and other iron alloys. In the steel industry, pickling is primarily carried out to remove scale and other oxide and hydroxide layers which have formed on the metal surfaces of the articles during production. These layers are formed primarily when the articles are treated thermally, e.g. when annealed and subsequently rolled.
- In the manufacturing industry, pickling processes are applied for the purpose of removing rust and other impurities from the surface of the material. Rust is formed during the transportation, storage and handling of the metal articles, these articles comprising finished goods from the steel industry or intermediate products from other metal working industries, e.g. from sub-contractors. The impurities originate from the same sources and may comprise, for instance, welding, soldering or brazing residues and handprint, although they may also be in the form of oil coatings and the like from processing operations or oil which has been purposely applied to protect the material against corrosion.
- In the steel industry the acid solution used to pickle non-alloyed steels or low-alloy steels (carbon steel) is mostly a sulphuric acid solution or a hydrochloric acid solution. When pickling stainless steel and other difficultly worked grades of material, the acid solution used is mostly a nitric acid solution or a hydrofluoric acid solution, or a mixture of these two acids.
- The acid used for pickling purposes in the manufacturing industry is predominantly sulphuric acid, although hydrochloric acid, nitric acid and hydrofluoric acid may also be used when considered an advantage in view of prevailing circumstances.
- Although in principle phosphoric acid can be considered equivalent to sulphuric acid and hydrochloric acid when pickling steel, it has a limited use because of longer pickling times and higher costs, since it is necessary to provide some form of regeneration equipment if the process is to be economically viable.
- Phosphating - a chemical pre-treatment process in which there is preferably used solutions whose major component is either iron phosphate or zinc phosphate - is often used, however, to obtain a short time corrosion protection prior to finally treating an article, and to provide better adhesion surfaces for paints and varnishes applied to the article, thereby to improve the anti-rust properties of the finished surfaces.
- When pickling articles of non-alloyed or low- alloyed steel which are heavily coated with scale, the scale is first partially dissolved while the major part is removed mechanically, by the generation of hydrogen gas in the pores and cavities of the scale. Subsequent to this removal of the scale, the exposed surfaces of the base metal of the article will be attacked by the acid, and if the pickling process is not stopped at just the right time excessive material will be lost without any advantage being gained.
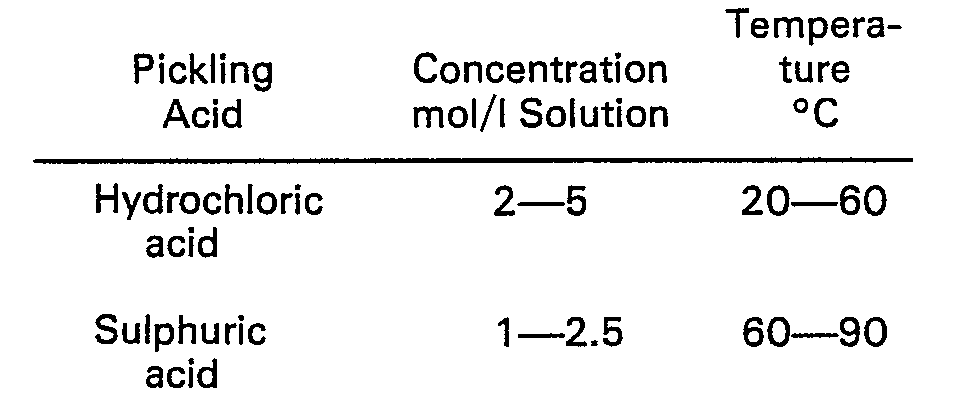
-
- Corresponding conditions when using phosphoric acid - which is seldom used on a technical scale, however - are 0.5-3 mol/I solution and 20-90°C.
- To obtain complete removal of scale in rolling mills, a pickling time of about one minute is normally required.
- In the case of pickling processes for the removal of rust, the rust and other impurities are progressively dissolved on the surface of the steel, and the conditions required are slightly different to those required when de-scaling. Normal concentrations and temperatures are shown in the following table.
- The pickling time for removing rust is normally from 5 to 20 minutes.
- A common feature of all pickling processes, however, is that as soon as the base metal is exposed, it is attacked by the acid and dissolves, resulting in an unnecessary loss of metal. To avoid this there is normally added an organic inhibitor, which is adsorbed on the exposed surfaces of the metal to block the same, thereby protecting said surfaces against acid attack.
- As beforementioned, phorphoric' acid is seldom used in pickling solution on a technical commercial scale, because of the aforesaid reasons. It has been proposed, however, to use phosphoric acid primarily for special pickling processes, for intensified pickling processes and for pickling special metals, where the disadvantages associated with the use of phosphoric acid as a pickling agent can be accepted in view of the special application. Thus, SU-A-152603 teaches the use of an acid pickling solution comprising a mixture of phosphoric acid and hydrochloric acid for the precision- pickling of iron metals, whereat the phosphoric acid content is 50 per cent by weight or more of the total acid content. It is proposed to recover the phosphoric acid content with the use of ion- exchanging resins, which is, of course, a very expensive method.
- US-A-1 279 331 (Gravell) suggests the use of a mixture of phosphoric acid and sulphuric acid when pickling iron and steel. In this case, the phosphoric acid content is more than 50 mole per cent by weight of the total acid content. The stated purpose of the phosphoric acid addition is to avoid subsequent attack by rust. The FR-A-2208006 also proposes a pickling solution containing phosphoric acid together with hydrochloric acid and sulphuric acid, whereat contents of 1-99 mole per cent by weight phosphoric acid, 5-99 mole per cent by weight sulphuric acid, and 5-99 mole per cent by weight hydrochloric acid are proposed as a pickling agent for stainless steel, and particularly as an agent to passivate such steels. Also JP-A- No. 53-138928 discloses stainless steel pickling compositions containing phosphoric acid.
- The admixture of minor quantities of phosphoric acid with pure or mixed concentrated mineral acids, such as sulphuric acid, hydrochloric acid and nitric acid, has also been proposed. The phosphoric acid is said to have a passivating effect on the acid, and its use is proposed in connection with acids which remain in contact with metal surfaces over very long periods of time. The method can be considered as a phosphating method in situ. In a method for pickling soft carbon steel, it is proposed in DE-B-1 169251 that the steel is treated with a solution containing sulphuric acid and/or phosphoric acid and hydrogen peroxide. The phosphoric acid content is said to be 80-95 per cent by weight of the total acid content.
- Phosphoric acid additions as pickling agents are also proposed in GB-A-854588, whereat the pickling agent, which may be in liquid or paste form, also includes sulphuric acid, hydrochloric acid, oxalic acid or an oxalic acid salt, a chromium salt and a wetting agent. The method is considered particularly suitable for cleaning metal surfaces which are to be painted. In such applications factors concerning pickling times and the dissolving of metals are not important and have not been discussed in the publication.
- According to DE-B-1185037, the use of a phosphoric acid pickling bath provides no significant phosphating effect on the surfaces of the metal being treated, due to the high content of free acid required for the pickling process. It is also maintained that phosphating methods or anti-rust methods using phosphoric acid and/or phosphate baths cannot be used for pickling purposes, because of the small content of free acid in the bath. To improve these conditions, the said publication proposes that one or more borophosphoric acid compounds be added to the bath. An addition of 0-60 per cent by volume phosphoric acid to hydrochloric acid for chemically polishing stainless steel is proposed in GB-A-1 140856. In US-A-2 337 062, which relates to the chemical treatment of metals and in particular of stainless steel, there is proposed a pickling solution containing a mixture of sulphuric acid, nitric acid and hydrofluoric acid, whereat it is mentioned that phosphoric acid can replace the sulphuric acid, either in full or in part, with only a "slight worsening effect" on the effectiveness of the solution. According to another method proposed in US-A-3 438 799 there is used a pickling solution containing a mineral acid mixture having a phosphoric acid content which is approximately three times as large as the hydrochloric acid content calculated in mole per cent.
- Certain specific mixtures of phosphoric acid and hydrochloric acid are suggested by US-A-2 559 445 (Lotz) in order to avoid pitting when scale pickling steel. Lotz, however, discloses use of a relative high content of phosphoric acid for obtaining optimal results, i.e. 20 per cent by volume of an 85 per cent by weight acid in the mixture.
- According to FR-A-1236268 a mixture of hydrochloric acid, phosphoric acid and sodium borate can be used for rapidly pickling iron metals. The pickling time, however, is relatively long and the phosphoric acid addition relatively high. In CH-A-134632 it has also been proposed to use phosphoric acid as a pickling agent at elevated temperatures and pressures, in order to achieve satisfactory pickling times and results. Thus, the use of phosphoric acid as a pickling agent, either alone or in combination with other pickling acids, is well known to the art. It will be noted however, that in all cases the acid has been used despite those problems which it can cause. Thus, the use of phosphoric acid as a pickling agent can be considered known in extreme cases as an extreme measure, where extreme requirements are placed on the appearance of the surfaces of a workpiece subsequent to pickling and treating the same, and where a residual protective surface is required (phosphating effect).
- Afanasev and Malaysheva (J. Appl. Chem. USSR Vol. 37 No. 4 (1964) p. 903-908 and Vol. 41 No. 3 (1968) p. 523-527 and Chem. Abstracts Vol. 67 (1967) p. 7385-8 and Vol. 65 (1966) Abstr. No. 10218e) have investigated acid mixtures of two and three components containing phosphoric acid for formulating an acid pickling paste. The optional total acid concentration was found to be above 5 mol/I, that is for more than would be actual for acid pickling solutions.
- When carrying out a pickling operation in practice, it is important that the correct pickling time is chosen. If the time chosen is too short, the result may be an incompletely cleansed surface, which may provide a product of poorer quality and render the surface more liable to corrosion later on. When the pickling time chosen is, on the other hand, too long, the amount of acid and steel consumed is greater than necessary. Moreover, in this latter case the structure of the steel surfaces becomes coarser, the useful lifetime of the bath decreases, and the load on the surroundings resulting from the vapours given off from the bath increases.
- Thus, it would be an advantage if the pickling process could be made less sensitive to variations in pickling times. By controlling the process, it would be possible to utilize the acid more effectively, to reduce the energy consumed, to produce a product of more uniform quality, and to reduce the number of interruptions in production caused by bath changes.
- When pickling articles in sulphuric acid, and also in other mineral acids, the pickling process can be divided into two stages. In the first stage, a major part of the scale present on the surfaces of the articles is quickly dissolved. The remainder of the scale is dissolved in the second stage, while the dominating part of the process is an attack on the cleansed metal surfaces exposed by the removal of the scale.
- The two stages in the pickling process can be separated specifically, if the decrease in weight per unit of time is determined as a function of the pickling time. During the second stage, it is also possible to observe the generation of hydrogen gas in a quantity corresponding to the amount of pure metal dissolved.
- It is well known to one skilled in this art that pickling is effected more rapidly if there is already a quantity of iron(II) ions present in the bath when the pickling process is started. Thus, the presence of Fe2+ ions increases the rate of dissolution of both the scale and the pure metal.
- When pickling an article in hydrochloric acid, it is not possible to work with as high temperatures as when sulphuric acid is used, since the partial pressure of hydrogen chloride gas in air above the surface of the bath is high, which results in the removal of vapours, which is both expensive and troublesome to the working environment. Moreover, equipment and apparatus in the vicinity of the bath are subjected to damage by corrosion.
- In practice, pickling processes using hydrochloric acid are effected at lower temperatures than those using sulphuric acid. Under comparable conditions, scale and pure metal are dissolved more rapidly than in sulphuric acid. The pickling rate in a hydrochloric bath is very high, especially when Fe2+ ions are already found in the bath at the commencement of the process.
- One great difficulty in acid pickling processes, particularly when using hydrochloric acid, is to control the process in a manner such as to keep the amount of material lost through acid attack on the exposed metal surfaces low. A further difficulty is that caused by the hydrogen gas generated as the metal is dissolved. Among other things, this formation of hydrogen gas causes small droplets of liquid from the bath to fly around in the surroundings.
- As beforementioned, it has been suggested to reduce the acid attack on the pure metal surfaces, by using an organic inhibitor. It is however very doubtful whether such inhibitors are effective in the way as has been suggested and it is in practice almost impossible to control the pickling process in this way. Consequently, in the majority of cases there is chosen in practice longer pickling times and excessive pickling, whereat it is impossible to avoid metal losses, with subsequent difficulties in recovery and deposition.
- As when pickling in hydrochloric acid or sulphuric acid, the pickling process when using phosphoric acid can be separated into two distinct stages. Dissolution of the major part of the scale takes place more slowly than in sulphuric acid, even though the process is carried out at high temperatures.
- The said second stage of the process is also effected much more slowly in phosphoric acid, which can be attributed to the formation of iron phosphate on the exposed metal surfaces. Phosphoric acid differs advantageously from both sulphuric acid and hydrochloric acid in this way.
- A prime object of the present invention is to provide a method of overcoming the aforementioned disadvantages while substantially fulfilling the aforeindicated desiderata. It has been found possible to achieve this object by using a pickling bath comprising novel, specific combinations of pickling acids.
- Another object of the invention is to provide a method in which the oxide layer, in the form of scale on rust, partially or completely covering the surfaces of the article, is rapidly dissolved.
- A further object of the invention is to reduce the extent to which metal is undesirably dissolved with extensive pickling times. This can be achieved by blocking the pure metal surfaces against the attack of hydrogen ions in the acid solution.
- Yet another object is to facilitate further surface treatment of the articles in conjunction with the pickling process, so that said surfaces can, for example, be pre-treated before applying thereto, e.g. a coating of paint or varnish, or some other layer forming organic substance.
- It has now been surprisingly found that the aforementioned objects can be realized in an efficient and simple manner, whereat there is thus obtained a combined rapid dissolution of the scale and slow attack on exposed metal surfaces, by applying the present invention, which relates to a method and an improved pickling agent for pickling articles of iron and iron alloys of before mentioned kind. Thus, the method of the invention is characterized in that said material is brought into contact with an aqueous solution containing phosphoric acid and one or both of the mineral acids hydrochloric acid and sulphuric acid, whereat the amount of phosphoric acid in the solution is adjusted to at least 0.1 mol/I and at most 1 mol/I, and the total acid concentration is 0.5-2 mol/I. The agent for carrying out the method is characterized in that it includes, as an inhibitor, phosphoric acid in an amount of at least 0.01 mol/I and at most 1 mol/I.
- The lower limit for the phosphoric acid content in the pickling solution is 0.01 mol/I. If the content is not maintained in the solution a substantial greater attack on the base metal will occur, which is not desirable. When the phosphoric acid content exceeds 1 mol/I the scale dissolution on the other hand will be so slow that the pickling time would have to be extended to an essential extent. This will decrease the pickling capacity and thus also the pickling economy. It is also essential in the method that the total acid concentration is controlled to be within the range of 0.5-2 mol/I. A lower total acid concentration will also give unacceptable long pickling times which results in serious disadvantages of beforementioned type. On the other hand acid concentrations above 4 mol/I are not particularly interesting, since the pickling operation will be difficult to control owing to the fact that too rapid reactions will occur. Additionally, the consumption of acid will be unacceptably high unless an expensive acid regenerating operation is introduced.
- Thus, when pickling an article in a mixture of sulphuric acid and phosphoric acid, the major part of the scale is dissolved at approximately the same rate as when pickling in solely sulphuric acid, whereat the percentage of phosphoric acid is below 20 mole per cent of the total acid present. When the amount of phosphoric acid added exceeds this value, the pickling rate, and thus also the pickling effect of the solution, is successively decreased.
- The addition of phosphoric acid to the sulphuric acid inhibits the ability of the sulphuric acid to dissolve metal. This inhibiting effect increases successively with increasing amounts of phosphoric acid, up to an amount of 20 mole per cent of the total amount of acid present, at which level said inhibition would appear to be complete. In practice, this inhibition is observed by the fact that the amount of hydrogen gas generated decreases. Thus, when using additions of phosphoric acid, only very small amounts of hydrogen gas are generated as compared with the use of pure sulphuric acid, where the generation of hydrogen gas is very high.
- When pickling in a mixture of hydrochloric acid and phosphoric acid, the conditions are similar to those when pickling in a mixture of sulphuric acid and phosphoric acid. The scale is dissolved very rapidly, when the phosphoric acid concentration is between 5 and 25 mole per cent of the total acid present. The pickling rate decreases successively with an increase of the phosphoric acid content above this range. Inhibition also begins immediately in the case of hydrochloric acid, when minor quantities of phosphoric acid are present, and increases successively until a phosphoric acid content of about 10 mole per cent of the total amount of acid present is reached. The presence of iron(II) ions in the pickling agent has a positive action on the inhibiting effect of phosphoric acid. Thus, when iron(II) ions are present, less phosphoric acid is required to achieve the same inhibiting effect as that achieved in the absence of said ions.
- The invention will now be further illustrated with reference to a number of examples and with reference to the accompanying drawings, which illustrate the pickling of articles using the novel pickling agent of the invention and known pickling agents. Throughout the examples and drawings M has been used for designating mole per litre (mol/I).
- In one experiment, three parallel pickling tests were carried out, by immersing sheets of carbon steel covered with scale in aqueous solutions which in the first test contained 2.0 M sulphuric acid, in the second test 2.0 M phosphoric acid, and in the third test 0.4 M phosphoric acid and 1.6 M sulphuric acid. Different sample pieces were pickled for differing lengths of time, thereby enabling the decrease in weight to be studied as a function of time. The experiment was carried out at a temperature of 70°C. The result is shown in Figure 1, from which it can be seen that the acid mixture, similar to the pure sulphuric acid, rapidly dissolved the scale, but - similar to the pure phosphoric acid - with considerably less dissolution of the base material.
- In another experiment three parallel pickling tests were carried out in a manner similar to that recited in Example 1, but with the exception that there was added iron(II) sulphate such that the concentration of Fell ions at the beginning of the pickling process was 35 g/I. The result is illustrated in Figure 2, which shows that the scale was dissolved by the acid mixture as rapid as by the pure sulphuric acid. Similar to the phosphoric acid, however, much less base material was dissolved by the acid mixture.
- In a similar experiment, a series of parallel pickling tests were carried out in aqueous solutions containing mixtures of sulphuric acid and phosphoric acid of varying composition, having a total acid content of 2 M. All tests were made at a temperature of 70°C. The rate at which the scale and metal dissolved was measured during the tests.
- The results, which are given in the table below and which are illustrated by Figure 3, show that the rate at which scale is dissolved remains unchanged when the phosphoric acid is increased from 0 to 0.4 M, which corresponds to 20 mole percent of the total acid present, but decreases when the increase exceeds this range. The rate at which metal is dissolved is low and remains unchanged when the phosphoric acid content is at least 0.4 M, i.e. 20 mole per cent of the total acid present. The rate at which the metal dissolves increases, however, when the phosphoric acid content falls below this value.
- In a similar experiment a series of parallel pickling tests were carried out in a manner similar to Example 3, but with an addition of iron(II) sulphate such that the concentration of Fe2+ ions at the commencement of the pickling period was 35 g/I. The rate at which scale and metal were dissolved was measured in the tests. The results are given in the table below and illustrated by Figure 4.
- The results from this experiment show that the presence of iron(II) ions affects the dissolution rate of the metal as a function of mole percent phosphoric acid, such that a smaller quantity of phosphoric acid is required for the same inhibiting effect as in the case when no iron(II) ions are present. Thus, the amount of phosphoric acid required to achieve maximum inhibition of the dissolution of the metal is only about 5-10 mole per cent. It is also evident that a phosphoric acid content above 0.01 M must be present to achieve required low metal dissolving rate.
- In a similar experiment, three parallel tests were carried out in aqueous solutions containing in the first test 2.0 M hydrochloric acid; in the second test 2.0 M phosphoric acid; and in the third test a mixture of 0.2 M phosphoric acid and 1.8 M hydrochloric acid. Each of the tests was carried out at a temperature of 70°C. The result is illustrated by Figure 5, which shows that the acid mixture, similar to the pure hydrochloric acid caused the scale to dissolve rapidly, but that, as with the pure phosphoric acid, far less base material was dissolved.
- In a similar experiment, a series of parallel pickling tests were conducted in aqueous solutions containing mixtures of hydrochloric acid and phosphoric acid of varying composition but all having a total acid content of 2 M. All the tests were carried out at a temperature of 70°C. The rate at which scale and metal were dissolved was measured in the tests. The result is given in the following table, which shows that the rate at which scale is dissolved depends upon the amount of phosphoric acid present. When the phosphoric acid content, however, lies between 0 and 0.5 M, corresponding to between 0 and 25 mole per cent of the total acid present, the rate of dissolution is of the same order as that obtained with 2 M sulphuric acid at the same temperature. A further increase will decrease the rate of dissolution. The rate at which the metal is dissolved is low and remains unchanged when the phosphoric acid content is at least 0.2 M, corresponding to 10 mole per cent of the total acid present. When the phosphoric acid content falls below this level, the rate at which the metal dissolves increases.
Claims (3)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT80850166T ATE10016T1 (en) | 1979-11-06 | 1980-11-05 | METHOD OF ACID PICKLING IRON AND FERROUS ALLOYS AND COMPOSITION FOR CARRYING OUT THE PROCESS. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SE7909187 | 1979-11-06 | ||
| SE7909187 | 1979-11-06 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0029418A1 EP0029418A1 (en) | 1981-05-27 |
| EP0029418B1 true EP0029418B1 (en) | 1984-10-24 |
Family
ID=20339264
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP80850166A Expired EP0029418B1 (en) | 1979-11-06 | 1980-11-05 | A method of acid pickling iron and iron alloys and a composition for carrying out the method |
Country Status (8)
| Country | Link |
|---|---|
| EP (1) | EP0029418B1 (en) |
| JP (1) | JPS56501490A (en) |
| AT (1) | ATE10016T1 (en) |
| DE (1) | DE3069514D1 (en) |
| DK (1) | DK289081A (en) |
| FI (1) | FI68865C (en) |
| NO (1) | NO812196L (en) |
| WO (1) | WO1981001298A1 (en) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2288190A (en) * | 1994-04-08 | 1995-10-11 | Wonder Wheels | Cleaning composition containing hydrochloric acid |
| US6420178B1 (en) * | 2000-09-20 | 2002-07-16 | General Electric Company | High throughput screening method, array assembly and system |
| US10450668B2 (en) | 2017-04-11 | 2019-10-22 | Savannah River Nuclear Solutions, Llc | Development of a passivated stainless steel surface |
| CN108930043A (en) * | 2018-08-15 | 2018-12-04 | 东莞市同盛电子科技有限公司 | A kind of cleaning agent and preparation method thereof removing metal oxide layer and application method |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SU152603A3 (en) * | 1900-01-01 | |||
| US1279331A (en) * | 1918-05-11 | 1918-09-17 | James H Gravell | Pickling iron and steel. |
| US2559445A (en) * | 1946-12-12 | 1951-07-03 | Union Switch & Signal Co | Method for removing scale from steel |
| FR1148779A (en) * | 1956-02-01 | 1957-12-13 | Process for protecting metals against corrosion by strong acids and compositions for the implementation of this process | |
| US2923608A (en) * | 1956-04-13 | 1960-02-02 | Fmc Corp | Method of improving the bonding properties of steel surfaces |
| BE579543A (en) * | 1958-06-19 | |||
| US3438799A (en) * | 1965-06-22 | 1969-04-15 | Chem Eng Ltd | Method for the surface treatment of metal articles |
| US3457107A (en) * | 1965-07-20 | 1969-07-22 | Diversey Corp | Method and composition for chemically polishing metals |
| BE740608A (en) * | 1968-10-22 | 1970-04-01 | Metal cleaning and protection process | |
| BE790538A (en) * | 1971-10-26 | 1973-02-15 | Ericsson Telefon Ab L M | ALUMINUM CHEMICAL CLEANING BATH |
| FR2208006A1 (en) * | 1972-11-24 | 1974-06-21 | Produits Ind Cie Fse | De-passivation of stainless steel - prior to plating or coating with copper, nickel, chromium, zinc, manganese, oxalate or phosphate |
-
1980
- 1980-11-05 WO PCT/SE1980/000273 patent/WO1981001298A1/en active IP Right Grant
- 1980-11-05 EP EP80850166A patent/EP0029418B1/en not_active Expired
- 1980-11-05 JP JP50259280A patent/JPS56501490A/ja active Pending
- 1980-11-05 DE DE8080850166T patent/DE3069514D1/en not_active Expired
- 1980-11-05 AT AT80850166T patent/ATE10016T1/en not_active IP Right Cessation
-
1981
- 1981-06-26 NO NO812196A patent/NO812196L/en unknown
- 1981-06-30 DK DK289081A patent/DK289081A/en not_active Application Discontinuation
- 1981-07-03 FI FI812107A patent/FI68865C/en not_active IP Right Cessation
Also Published As
| Publication number | Publication date |
|---|---|
| ATE10016T1 (en) | 1984-11-15 |
| EP0029418A1 (en) | 1981-05-27 |
| FI68865B (en) | 1985-07-31 |
| FI812107L (en) | 1981-07-03 |
| DK289081A (en) | 1981-06-30 |
| FI68865C (en) | 1985-11-11 |
| WO1981001298A1 (en) | 1981-05-14 |
| JPS56501490A (en) | 1981-10-15 |
| NO812196L (en) | 1981-06-26 |
| DE3069514D1 (en) | 1984-11-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0180908B1 (en) | Aluminum surface cleaning agent | |
| JPH0320476A (en) | Formation of zinc phosphate film on metallic surface | |
| US2564549A (en) | Pickling treatment | |
| US3114657A (en) | Composition and method for cleaning and stripping metals | |
| JPH06104906B2 (en) | Method for improving surface coating of zinc | |
| CA1330515C (en) | Process of forming phosphate coatings on metals | |
| US5614028A (en) | Method of cleaning and passivating a metal surface with acidic system and ethoxylated tertiary dodecyl mercaptan | |
| JP2000516999A (en) | Aqueous solution and method for phosphating metal surfaces | |
| EP0029418B1 (en) | A method of acid pickling iron and iron alloys and a composition for carrying out the method | |
| EP0617144B1 (en) | Use of an aqueous acidic cleaning solution for aluminum and aluminum alloys and process for cleaning the same | |
| JPH0465151B2 (en) | ||
| US2835616A (en) | Procedure for the manufacture of oxalate coatings on metals | |
| US3338755A (en) | Production of phosphate coatings on metals | |
| US3450578A (en) | Process and composition for the production of protective coatings | |
| US2511988A (en) | Pickling process | |
| US2288995A (en) | Surface treatment of magnesium and its alloys | |
| US1428087A (en) | Rust-resisting steel and method of producing same | |
| Hudson | Pickling and descaling | |
| US3288655A (en) | Phosphating a steel strip prior to anealing and temper rolling | |
| Harris | metal cleaning bibliographical abstracts | |
| US2209291A (en) | Rust removing composition | |
| US3046165A (en) | Method of and solution for forming phosphate coatings on zinc surfaces | |
| US2104667A (en) | Treatment of rustless iron | |
| EP0064295B1 (en) | Method of improving the corrosion resistance of chemical conversion coated aluminum | |
| RU2061101C1 (en) | Solution fo simultaneous pickling and degreasing of metals |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR GB IT LU NL SE |
|
| 17P | Request for examination filed |
Effective date: 19811126 |
|
| ITF | It: translation for a ep patent filed |
Owner name: UFFICIO BREVETTI RICCARDI & C. |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR GB IT LI LU NL SE |
|
| REF | Corresponds to: |
Ref document number: 10016 Country of ref document: AT Date of ref document: 19841115 Kind code of ref document: T |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19841108 Year of fee payment: 5 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 19841112 Year of fee payment: 5 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19841116 Year of fee payment: 5 |
|
| REF | Corresponds to: |
Ref document number: 3069514 Country of ref document: DE Date of ref document: 19841129 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19841130 |
|
| ET | Fr: translation filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 19841231 Year of fee payment: 5 Ref country code: BE Payment date: 19841231 Year of fee payment: 5 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 19851114 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19851130 Year of fee payment: 6 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Effective date: 19861105 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Effective date: 19861106 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Effective date: 19861130 Ref country code: CH Effective date: 19861130 Ref country code: BE Effective date: 19861130 |
|
| BERE | Be: lapsed |
Owner name: BOLIDEN A.B. Effective date: 19861130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19870601 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee | ||
| GBPC | Gb: european patent ceased through non-payment of renewal fee | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19870731 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Effective date: 19870801 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Effective date: 19881118 |
|
| EUG | Se: european patent has lapsed |
Ref document number: 80850166.2 Effective date: 19870901 |