CN111328713B - Method for constructing malus asiatica leaf regeneration system - Google Patents
Method for constructing malus asiatica leaf regeneration system Download PDFInfo
- Publication number
- CN111328713B CN111328713B CN202010151839.0A CN202010151839A CN111328713B CN 111328713 B CN111328713 B CN 111328713B CN 202010151839 A CN202010151839 A CN 202010151839A CN 111328713 B CN111328713 B CN 111328713B
- Authority
- CN
- China
- Prior art keywords
- culture medium
- culture
- concentration
- culturing
- rooting
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01H—NEW PLANTS OR NON-TRANSGENIC PROCESSES FOR OBTAINING THEM; PLANT REPRODUCTION BY TISSUE CULTURE TECHNIQUES
- A01H4/00—Plant reproduction by tissue culture techniques ; Tissue culture techniques therefor
- A01H4/001—Culture apparatus for tissue culture
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01H—NEW PLANTS OR NON-TRANSGENIC PROCESSES FOR OBTAINING THEM; PLANT REPRODUCTION BY TISSUE CULTURE TECHNIQUES
- A01H4/00—Plant reproduction by tissue culture techniques ; Tissue culture techniques therefor
- A01H4/008—Methods for regeneration to complete plants
Abstract
The invention discloses a method for constructing a malus asiatica cotyledon regeneration system, which comprises the following steps: firstly, preprocessing; secondly, sterilizing; thirdly, adventitious bud induction; fourthly, elongation and strong bud culture; fifthly, rooting culture; and sixthly, transplanting. The invention uses the safflower leaves as explants to complete in vitro regeneration, and the technical scheme provided by the invention is utilized to carry out in vitro regeneration on the safflower, thereby improving the induced germination rate and the increment coefficient.
Description
Technical Field
The invention belongs to the technical field of plant tissue culture, and particularly relates to a method for constructing a malus asiatica cotyledon regeneration system.
Background
Carthamus tinctorius (Malus asiatica), Rosaceae (Rosaceae), Malus (Malus), and Small deciduous trees. The crab apple, also known as crabapple, is neutral in nature, sweet and sour in taste, is rich in various nutrient substances and has the effects of quenching thirst, promoting the production of body fluid, and promoting digestion to remove food stagnation. Besides fresh eating, the fruit can be processed into dried fruit, sweetend roll or wine. The flower red is not only an ornamental tree species with beautiful flowers and fruits, but also an excellent stock of the apple due to good grafting compatibility with the apple. However, the scarlet is mainly grafted and bred, and serious germplasm resource deficiency caused by the large downcut is caused, and the problem can be solved by establishing a regeneration system.
The in vitro plant regeneration technology is the basis of the researches on the preservation of the germplasm of the saffron, genetic improvement and the like. At present, explants commonly used for establishing a plant regeneration system comprise organs such as leaves, cotyledons, hypocotyls, stem segments and the like, researchers establish the regeneration system by using young embryos and stem segments of the saffron, but the regeneration effect is not particularly ideal. Stem segments are regenerated in vitro, the optimal culture medium multiplication coefficient is 3.8, and rooted seedlings are not obtained; the young embryo is taken as an ectosome, 6 strong tips and bud spots longer than 1cm are formed on the optimal culture medium 30 balance, but the individual body formed by the embryo development possibly has a character separation phenomenon, the quality of the variety is influenced, and the embryo is not suitable for being used as the ectosome of a regeneration system. The stem section with buds is used as an ectosome of an in vitro rapid propagation system, multiplication of sterile seedlings of the saffron can be effectively promoted on an MS culture medium added with 2.0 mg/L6-BA and 0.25mg/L NAA, the saffron is a perennial woody plant, the stem section is easy to lignify, and the cotyledon explant has the advantages of being thick, rich in nutrient substances, easy to obtain materials, easy to operate and the like, and is probably the best choice for establishing a stable regeneration system of the saffron.
The problem that the establishment of a regeneration system is immature, the regeneration difficulty is high, the regeneration difference among different genotypes is large, the number of deformed seedlings is large, the regeneration speed is low, the rooting is difficult and the like exists in the anthurium andraeanum. Therefore, the establishment of a regeneration system of the flower red needs to be solved urgently, and a high-efficiency and stable flower red regeneration system is established, so that technical support is provided for genetic engineering and molecular breeding.
Disclosure of Invention
The invention aims to provide a method for constructing a carthamus tinctorius leaf regeneration system by taking carthamus tinctorius leaves as explants and inducing and differentiating roots or buds, which overcomes the defects of the original regeneration system and solves the problems of difficult regeneration, difficult rooting and the like.
The technical scheme adopted by the invention is as follows:
a method for constructing a carthamus tinctorius cotyledon regeneration system, which takes the carthamus tinctorius cotyledon as an explant to complete in vitro regeneration, comprises the following steps:
(1) pretreatment: selecting mature and plump anthus andraeanum seeds, and soaking the seeds in sterile water for 10-12 min;
(2) and (3) sterilization: soaking the pretreated seeds in alcohol for 30-35s, washing with sterile water for 2-3 times, soaking with sodium hypochlorite solution for 8-10min, and washing with sterile water for 3-4 times;
(3) adventitious bud induction: peeling germs from the sterilized seeds, cutting off partial cotyledons at the near shaft end and the far shaft end, inoculating to an adventitious bud induction culture medium, performing dark culture for 7d, and performing light-to-light culture;
(4) and (3) elongation and strong bud culture: transferring the culture material in the step (3) to an elongation bud-strengthening culture medium, and culturing for 30-40d under light;
(5) rooting culture: transferring the material with stem height of 1-2cm cultured in the step (4) to a rooting culture medium, and culturing for 30-40 days under light;
(6) transplanting: and (4) when the root length induced in the step (5) is 2-3cm, washing the root culture medium, transplanting the root culture medium into sterilized nutrient soil, and placing the root culture medium in an indoor environment at 25 ℃ for natural growth.
Preferably, the alcohol concentration in the step (2) is 75%, and the mass fraction of the sodium hypochlorite is 10%.
Further, the adventitious bud induction medium in the step (3) comprises the following components: the MS medium was supplemented with TDZ at a concentration of 1mg/L and NAA at a concentration of 0.05 mg/L.
Further, the components of the medium for elongating and strengthening the bud in the step (4) are as follows: MS culture medium was supplemented with 6-BA at a concentration of 1.5mg/L and NAA at a concentration of 0.3 mg/L.
Further, the rooting medium in the step (5) comprises the following components: MS culture medium was supplemented with IBA at a concentration of 1mg/L and NAA at a concentration of 0.2 mg/L.
Preferably, the culturing conditions under light are as follows: the temperature is kept at 24-26 ℃, the illumination time is 16h/d, the dark culture is 8h/d, and the illumination intensity is 3000-.
Further, the growth conditions of the step (6) are as follows: the illumination time is 16h/d, the dark culture is 8h/d, and the illumination intensity is 4000-.
The invention has the beneficial effects that: compared with the prior art, the method for constructing the carthamus tinctorius regeneration system by adopting the scheme of the invention takes the carthamus tinctorius leaves as explants, and the cotyledon explants have the advantages of being fat, rich in nutrient substances, easily available in materials, easy to operate and the like, can efficiently complete in vitro regeneration, completely retain the genetic characters of the carthamus tinctorius, and are the best choice for establishing the carthamus tinctorius stable regeneration system. An induction culture medium MS +1mg/L TDZ +0.05mg/L NAA is used, the induction rate of the floral safflower cotyledon cluster buds reaches 38.89%, the regenerated buds are more and robust through the elongation bud-strengthening culture, the multiplication coefficient reaches 5.13 +/-0.96 and is higher than that of a stem section used as an explant; the rooting medium comprises MS +1mg/L IBA +0.2mg/LNAA, the rooting rate is 60% after 30-40 days of culture, the average rooting number is 9.67 +/-3.04, the rooting rate can reach 100% along with the extension of the subculture time, the grown root system is thick, the transplanting survival is facilitated, and the rooting condition is obviously superior to the rooting condition of taking the young embryo as an ectosome.
The invention provides a stable and efficient method, and solves the problems that the material taking of a carthamus tinctorius regeneration system is limited, explants are easy to brown and the like.
Drawings
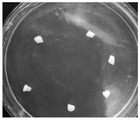
FIG. 1 is a graph showing the leaf peeling of a safflower;
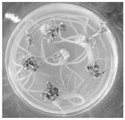
FIG. 2 shows the induction of regenerated shoots from 20d safflower leaves;
FIG. 3 shows the growth of the shoots after 50 days of cultivation;
FIG. 4 shows regenerated shoots grown in culture;
FIG. 5 shows the culture of proliferating strong shoots: the A picture is the proliferation of the regenerated bud, and the B picture is the strong bud of the regenerated bud;
FIG. 6 shows rooting culture.
Detailed Description
The present invention will be explained in further detail with reference to specific embodiments, which are given solely for the purpose of illustrating the invention and are not to be construed as limiting the scope of the invention.
The following examples are carried out in a conventional manner unless otherwise specified.
Materials, reagents and the like used in the following examples are generally commercially available.
The MS culture medium used in the examples of the present invention was purchased from Hangzhou Baisi Biotechnology, Inc.
TDZ: thidiazuron, a cytokinin;
NAA: naphthaleneacetic acid, an auxin;
6-BA: 6-benzylaminopurine, a cytokinin;
IBA: indolebutyric acid, an endogenous auxin.
The culture conditions under light were: keeping the temperature at 24-26 ℃, the illumination time at 16h/d, dark culture at 8h/d, and the illumination intensity at 3000 lux.
Example 1
A method for constructing a carthamus tinctorius leaf regeneration system, which takes carthamus tinctorius leaves as an explant to complete in vitro regeneration, comprises the following steps:
(1) pretreatment: selecting mature and plump anthurium seeds, and soaking the seeds in sterile water for 10 min;
(2) and (3) sterilization: soaking the seeds in 75% alcohol for 30s, washing with sterile water for 2-3 times, soaking in 10% sodium hypochlorite solution for 10min, and washing with sterile water for 3-4 times;
(3) adventitious bud induction: peeling germs from the sterilized seeds, cutting off partial cotyledons at the near shaft end and the far shaft end, inoculating the seeds on an adventitious bud induction culture medium, performing dark culture for 7 days, and performing light-to-light culture;
the adventitious bud induction culture medium comprises the following components: MS +1mg/LTDZ +0.05 mg/LNAA;
(4) and (3) elongation and strong bud culture: transferring the culture material in the step (3) to an elongation bud-strengthening culture medium, and culturing for 30-40d under light;
the components of the culture medium for the elongation and strong bud are as follows: MS +1.5 mg/L6-BA +0.3 mg/LNAA;
(5) rooting culture: transferring the material with stem height of 1-2cm cultured in the step (4) to a rooting culture medium, and culturing for 30-40 days under light;
the rooting medium comprises the following components: MS +1mg/L IBA +0.2 mg/LNAA;
(6) transplanting: and (3) when the root length induced in the step (5) is 2-3cm, washing the root culture medium, transplanting the root culture medium into sterilized nutrient soil, performing dark culture for 8h under the conditions of illumination for 16h and illumination for 6000lux at 25 ℃ for 14 days, and performing natural growth under the conditions of illumination for 6000lux at 25 ℃.
Example 2
A method for constructing a anthurium andraeanum cotyledon regeneration system takes a anthurium andraeanum cotyledon as an explant to complete in-vitro regeneration, and specifically comprises the following steps:
(1) pretreatment: soaking plump seed of Malus asiatica with sterile water for 10 min;
(2) and (3) sterilization: washing with 75% alcohol for 30s, washing with sterile water for 2-3 times, treating with 10% sodium hypochlorite solution for 10min, and washing with sterile water for 3-4 times;
(3) adventitious bud induction: peeling germs from the sterilized seeds, cutting off cotyledons at the end of a proximal shaft and the end of a distal shaft, inoculating the cut cotyledons on an adventitious bud induction culture medium, and culturing under light;
the adventitious bud induction culture medium comprises the following components: MS +1mg/LTDZ +0.05 mg/LNAA;
(4) and (3) elongation and strong bud culture: transferring the culture material in the step (3) to a culture medium for extending strong buds, and culturing for 30-40 days under light;
the components of the culture medium for the elongation and strong bud are as follows: MS +1.5 mg/L6-BA +0.3 mg/LNAA;
(5) rooting culture: transferring the material with stem height of 1-2cm cultured in the step (4) to a rooting culture medium, and culturing for 30-40 days under the light;
the rooting medium comprises the following components: MS +1mg/L IBA +0.2 mg/LNAA;
(6) transplanting: and (3) when the root length induced in the step (5) is 2-3cm, washing the root culture medium, transplanting the root culture medium into sterilized nutrient soil, performing dark culture for 8h under the conditions of illumination for 16h and illumination for 6000lux at 25 ℃ for 14 days, and performing natural growth under the conditions of illumination for 6000lux at 25 ℃.
Example 3
A method for constructing a anthurium andraeanum cotyledon regeneration system takes a anthurium andraeanum cotyledon as an explant to complete in-vitro regeneration, and specifically comprises the following steps:
(1) pretreatment: soaking plump seed of Malus asiatica with sterile water for 10 min;
(2) and (3) sterilization: washing with 75% alcohol for 30s, washing with sterile water for 2-3 times, treating with 10% sodium hypochlorite solution for 10min, and washing with sterile water for 3-4 times;
(3) adventitious bud induction: peeling embryo bud from the sterilized seed, cutting off cotyledon at the part near to shaft end and far from shaft end, inoculating on adventitious bud induction culture medium, dark culturing for 14d, and culturing under light;
the adventitious bud induction culture medium comprises the following components: MS +1mg/LTDZ +0.05 mg/LNAA;
(4) and (3) elongation and strong bud culture: transferring the culture material in the step (3) to a culture medium for extending strong buds, and culturing for 30-40 days under light;
the components of the culture medium for the elongation and strong bud are as follows: MS +1.5 mg/L6-BA +0.3 mg/LNAA;
(5) rooting culture: transferring the material with stem height of 1-2cm cultured in the step (4) to a rooting culture medium, and culturing for 30-40 days under the light;
the rooting medium comprises the following components: MS +1mg/L IBA +0.2 mg/LNAA;
(6) transplanting: and (3) when the root length induced in the step (5) is 2-3cm, washing the root culture medium, transplanting the root culture medium into sterilized nutrient soil, performing dark culture for 8h under the conditions of illumination for 16h and illumination for 6000lux at 25 ℃ for 14 days, and performing natural growth under the conditions of illumination for 6000lux at 25 ℃.
For examples 1-3, medium test groups of different compositions were set, and the adventitious bud regeneration of examples 1-3 was observed and recorded in real time:
TABLE 1 Effect of different media and different dark culture times on adventitious bud Induction
According to the experimental result, in the period of adventitious bud induction, MS +1mg/L TDZ +0.05mg/LNAA culture medium is adopted, cotyledons are cultured in dark for 7 days and then cultured for 2 weeks under light, namely, buds grow out from the cotyledons, the average number of buds is the largest in the same continuous culture time, the regeneration rate of adventitious buds is the highest, the growth quality of the buds is the best, and the method is beneficial to the continuous culture in the later stage.
According to the cultivation method (7 d) of example 1, the adventitious bud induction culture is performed by using the culture medium of TDZ, NAA and IBA combination with different concentrations, the culture is performed for 40 days under illumination of 16h and dark culture of 8h every day at 25 ℃, the callus induction rate and the cluster bud induction rate are counted, and the results are shown in Table 2:
TABLE 2 Effect of different concentrations by mass of TDZ, NAA and IBA combined culture on the regeneration of the cotyledons of Malus asiatica
Through experiments, the following results are obtained: when the concentration of TDZ is 1.0mg/L and the concentration of NAA is 0.05mg/L, the induction rate of the adventitious bud is the highest and is 38.89%, and the average bud number is 11.71.
Transferring the adventitious bud induction proliferation culture material to an elongation bud-strengthening culture medium for culture, setting 6-BA and NAA combinations with different concentrations in the elongation bud-strengthening culture medium, and culturing under the conditions of 25 ℃ and 3000lux of illumination for 16h and 8h in dark every day for 40 days, wherein the results are shown in Table 3.
TABLE 3 Effect of media with different concentrations of 6-BA and NAA combinations on the proliferation of flower red regenerated bud
As can be seen from Table 3, when the concentration of 6-BA was 1.5mg/L and the concentration of NAA was 0.3mg/L, the regenerated shoots were strong and had many regenerated shoots, and the multiplication factor reached 5.13. + -. 0.96.
Transferring the regenerated seedlings with the height of 1-3cm after the elongation bud strengthening culture to a rooting culture medium for inducing rooting, adopting IBA and NAA combined culture media with different concentrations, illuminating for 16h every day and culturing in the dark for 8h under the conditions of 25 ℃ and 3000lux of illumination, culturing for 40 days, and counting the rooting rate, wherein the results are shown in Table 5.
TABLE 4 rooting of safflower seedlings in media with different concentrations of IBA and NAA combinations
As shown in Table 4, when the IBA concentration was 1mg/L and the NAA concentration was 0.2mg/L, the rooting rate of the regenerated seedlings was 60% at the highest.
Referring to fig. 2 to 6, which are photographs of cotyledon-induced regenerated shoots and regenerated shoots elongating, strengthening and rooting in example 1 of the present invention, it can be seen that the method can induce saffron regenerated shoots more efficiently, and the construction of a saffron regeneration system by the method is more suitable and efficient than the original method.
The invention selects the Guizhou native safflower cotyledon as the explant to obtain the adventitious bud of the safflower, screens the adventitious bud induction culture medium formula and constructs the regeneration system of the safflower.
The embodiments described above are only a part of the embodiments of the present invention, and not all of the embodiments. All other embodiments, which can be obtained by a person skilled in the art without making any creative effort based on the embodiments in the present invention, shall fall within the protection scope of the present invention.
Claims (3)
1. A method for constructing a carthamus tinctorius leaf regeneration system is characterized by comprising the following steps: the method takes the safflower cotyledon as an explant to complete in vitro regeneration and comprises the following steps:
(1) pretreatment: selecting mature and plump anthus andraeanum seeds, and soaking the seeds in sterile water for 10-12 min;
(2) and (3) sterilization: soaking the pretreated seeds in alcohol for 30-35s, washing with sterile water for 2-3 times, soaking with sodium hypochlorite solution for 8-10min, and washing with sterile water for 3-4 times; the alcohol concentration is 75%, the mass fraction of the sodium hypochlorite is 10%, the germ of the sterilized seed is stripped, and partial cotyledons at the near shaft end and the far shaft end are cut off for later use;
(3) adventitious bud induction: inoculating the standby material into an adventitious bud induction culture medium, culturing in dark for 7d, and then culturing under light, wherein the adventitious bud induction culture medium comprises the following components: adding TDZ with the concentration of 1mg/L and NAA with the concentration of 0.05mg/L into the MS culture medium;
(4) And (3) elongation and strong bud culture: transferring the culture material in the step (3) to a culture medium for growing strong buds, and culturing for 30-40 days under light, wherein the culture medium for growing strong buds comprises the following components: adding 6-BA with the concentration of 1.5mg/L and NAA with the concentration of 0.3mg/L into an MS culture medium;
(5) rooting culture: transferring the material with the stem height of 1-2cm cultured in the step (4) to a rooting medium, and culturing for 30-40 days under light, wherein the rooting medium comprises the following components: adding IBA with the concentration of 1mg/L and NAA with the concentration of 0.2mg/L into the MS culture medium;
(6) transplanting: and (4) when the root length induced in the step (5) is 2-3cm, washing the root culture medium, transplanting the root culture medium into sterilized nutrient soil, and placing the root culture medium in an indoor environment at 25 ℃ for natural growth.
2. The method for constructing a anthurium cotyledon regeneration system according to claim 1, wherein the culturing conditions under light are as follows: the temperature is kept at 24-26 ℃, the illumination time is 16h/d, the dark culture is 8h/d, and the illumination intensity is 3000-.
3. A method for constructing a malus asiatica cotyledon regeneration system according to claim 1, wherein the growth conditions of step (6) are as follows: the illumination time is 16h/d, the dark culture is 8h/d, and the illumination intensity is 4000-.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010151839.0A CN111328713B (en) | 2020-03-06 | 2020-03-06 | Method for constructing malus asiatica leaf regeneration system |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010151839.0A CN111328713B (en) | 2020-03-06 | 2020-03-06 | Method for constructing malus asiatica leaf regeneration system |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111328713A CN111328713A (en) | 2020-06-26 |
| CN111328713B true CN111328713B (en) | 2022-05-24 |
Family
ID=71174292
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010151839.0A Active CN111328713B (en) | 2020-03-06 | 2020-03-06 | Method for constructing malus asiatica leaf regeneration system |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111328713B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111183900A (en) * | 2020-01-19 | 2020-05-22 | 贵州大学 | Method for efficiently obtaining aseptic safflower seedlings |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2397032A1 (en) * | 2009-02-13 | 2011-12-21 | Incorporated National University Iwate University | Method for accelerating flowering of rose family fruit tree using apple latent spherical virus vector |
| CN106332780A (en) * | 2016-08-31 | 2017-01-18 | 李军 | Construction method for in-vitro regeneration system of flos carthami |
| CN106386491A (en) * | 2016-09-14 | 2017-02-15 | 上海辰山植物园 | In vitro regeneration method of Begonia wallichiana Lehm |
| CN106879476A (en) * | 2017-04-28 | 2017-06-23 | 李茂兰 | A kind of tissue culture method of strawberry |
| CN108040885A (en) * | 2018-01-29 | 2018-05-18 | 宝鸡松良农业科技有限公司 | A kind of method that tissue cultures are carried out using cherry stem section |
| CN110367124A (en) * | 2019-08-29 | 2019-10-25 | 淮北师范大学 | A method of building peanut cotylcdon regenerating system |
-
2020
- 2020-03-06 CN CN202010151839.0A patent/CN111328713B/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2397032A1 (en) * | 2009-02-13 | 2011-12-21 | Incorporated National University Iwate University | Method for accelerating flowering of rose family fruit tree using apple latent spherical virus vector |
| CN106332780A (en) * | 2016-08-31 | 2017-01-18 | 李军 | Construction method for in-vitro regeneration system of flos carthami |
| CN106386491A (en) * | 2016-09-14 | 2017-02-15 | 上海辰山植物园 | In vitro regeneration method of Begonia wallichiana Lehm |
| CN106879476A (en) * | 2017-04-28 | 2017-06-23 | 李茂兰 | A kind of tissue culture method of strawberry |
| CN108040885A (en) * | 2018-01-29 | 2018-05-18 | 宝鸡松良农业科技有限公司 | A kind of method that tissue cultures are carried out using cherry stem section |
| CN110367124A (en) * | 2019-08-29 | 2019-10-25 | 淮北师范大学 | A method of building peanut cotylcdon regenerating system |
Non-Patent Citations (2)
| Title |
|---|
| Influence of plant growth regulators and sucrose on adventitious shoot formation from cotyledonary explants of apple;An, Hyun-Joo等;《Han"guk Wonye Hakhoechi》;20000430;第41卷;第173页摘要 * |
| 矮花红幼胚诱导成苗及组培快繁;蒋启林等;《四川果树》;19940515(第2期);第10页右栏第3-4段 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111328713A (en) | 2020-06-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5712672B2 (en) | Blueberry cultivation method | |
| Khan et al. | Thidiazuron induced somatic embryogenesis and plant regeneration in Capsicum annuum | |
| CN101057556A (en) | Method for high frequency plant regenerating of tallow tree tissue culture adventitious bud | |
| Srivastava et al. | In vitro seed germination and plant regeneration of an epiphytic orchid Aerides ringens (Lindl.) Fischer | |
| CN105265317B (en) | A kind of For-carrying green onions rapid propagation method | |
| CN111328713B (en) | Method for constructing malus asiatica leaf regeneration system | |
| CN111587688B (en) | In-vitro preservation and breeding method of kiwi fruit resources | |
| CN112273238A (en) | Tissue culture and rapid propagation seedling raising method for Daiyanlu plants | |
| CN108064699A (en) | A kind of tissue Propagation In Vitro method of pueraria lobata plant | |
| CN115885855A (en) | Method for establishing regeneration system by taking hypocotyl of Zikui tea tree as explant | |
| CN111512962B (en) | Prunus humilis tissue culture method | |
| Roy et al. | In vitro propagation and establishment of a new cultivar of jackfruit (Artocarpus heterophyllus) bearing fruits twice yearly | |
| CN109906939B (en) | Pepper in-vitro regeneration method and culture medium used by same | |
| CN111771729A (en) | Tissue culture and rapid propagation method for pitaya | |
| Hoque et al. | Overcoming phenolic accumulation during callus induction and in vitro organogenesis in water chestnut (Trapa japonica Flerov) | |
| Khatun et al. | In vitro plant regeneration from cotyledon and internodes derived callus in watermelon (Citrulus lanatus Thumb.) | |
| CN112005882A (en) | Method for saving actinidia arguta distant hybridization embryos | |
| CN115968785B (en) | Method for establishing Chinese cherry regeneration system | |
| CN114403003B (en) | In-vitro regeneration method for green cherry blossom plants | |
| CN103766220A (en) | Method for establishing efficient regeneration system of Chahu date | |
| CN116569842B (en) | Method for rapidly obtaining regeneration seedlings of Wucai by utilizing embryo tip tissues | |
| CN117502246B (en) | Construction method of lotus rapid propagation system | |
| CN112493126B (en) | Method for induction of lagerstroemia indica somatic embryo and plant regeneration | |
| CN112493127B (en) | Method for inducing somatic embryos and regenerating plants of Dutch beans | |
| CN112400692B (en) | Tissue culture and rapid propagation method of Chinese flowering crabapple |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |