CN107320552B - 口服药物组合物和其用于制备治疗或预防疾病的药物的用途 - Google Patents
口服药物组合物和其用于制备治疗或预防疾病的药物的用途 Download PDFInfo
- Publication number
- CN107320552B CN107320552B CN201710217749.5A CN201710217749A CN107320552B CN 107320552 B CN107320552 B CN 107320552B CN 201710217749 A CN201710217749 A CN 201710217749A CN 107320552 B CN107320552 B CN 107320552B
- Authority
- CN
- China
- Prior art keywords
- vol
- oral pharmaceutical
- pharmaceutical composition
- essential oil
- composition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000008203 oral pharmaceutical composition Substances 0.000 title claims abstract description 141
- 238000011282 treatment Methods 0.000 title claims abstract description 17
- 239000003814 drug Substances 0.000 title claims abstract description 8
- 230000002265 prevention Effects 0.000 title claims description 5
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 title description 23
- 201000010099 disease Diseases 0.000 title description 19
- 239000000341 volatile oil Substances 0.000 claims abstract description 148
- 239000000203 mixture Substances 0.000 claims abstract description 113
- 239000002199 base oil Substances 0.000 claims abstract description 60
- 235000015872 dietary supplement Nutrition 0.000 claims abstract description 56
- 235000014113 dietary fatty acids Nutrition 0.000 claims abstract description 52
- 239000000194 fatty acid Substances 0.000 claims abstract description 52
- 229930195729 fatty acid Natural products 0.000 claims abstract description 52
- 150000004665 fatty acids Chemical class 0.000 claims abstract description 51
- 241000196324 Embryophyta Species 0.000 claims abstract description 29
- 240000007673 Origanum vulgare Species 0.000 claims abstract description 17
- 235000013628 Lantana involucrata Nutrition 0.000 claims abstract description 16
- 235000006677 Monarda citriodora ssp. austromontana Nutrition 0.000 claims abstract description 16
- 244000178870 Lavandula angustifolia Species 0.000 claims abstract description 9
- 235000010663 Lavandula angustifolia Nutrition 0.000 claims abstract description 9
- 239000001102 lavandula vera Substances 0.000 claims abstract description 9
- 235000018219 lavender Nutrition 0.000 claims abstract description 9
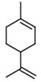
- XMGQYMWWDOXHJM-UHFFFAOYSA-N limonene Chemical compound CC(=C)C1CCC(C)=CC1 XMGQYMWWDOXHJM-UHFFFAOYSA-N 0.000 claims description 26
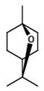
- WEEGYLXZBRQIMU-UHFFFAOYSA-N Eucalyptol Chemical compound C1CC2CCC1(C)OC2(C)C WEEGYLXZBRQIMU-UHFFFAOYSA-N 0.000 claims description 22
- DTOSIQBPPRVQHS-PDBXOOCHSA-N alpha-linolenic acid Chemical compound CC\C=C/C\C=C/C\C=C/CCCCCCCC(O)=O DTOSIQBPPRVQHS-PDBXOOCHSA-N 0.000 claims description 20
- 244000178231 Rosmarinus officinalis Species 0.000 claims description 17
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 claims description 17
- 244000042664 Matricaria chamomilla Species 0.000 claims description 16
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 claims description 15
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 claims description 15
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 claims description 15
- 244000144725 Amygdalus communis Species 0.000 claims description 15
- 235000011437 Amygdalus communis Nutrition 0.000 claims description 15
- 206010067125 Liver injury Diseases 0.000 claims description 15
- 239000005642 Oleic acid Substances 0.000 claims description 15
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 claims description 15
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 claims description 15
- 229920000642 polymer Polymers 0.000 claims description 15
- OYHQOLUKZRVURQ-NTGFUMLPSA-N (9Z,12Z)-9,10,12,13-tetratritiooctadeca-9,12-dienoic acid Chemical compound C(CCCCCCC\C(=C(/C\C(=C(/CCCCC)\[3H])\[3H])\[3H])\[3H])(=O)O OYHQOLUKZRVURQ-NTGFUMLPSA-N 0.000 claims description 14
- 235000001510 limonene Nutrition 0.000 claims description 13
- 229940087305 limonene Drugs 0.000 claims description 13
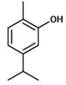
- HHTWOMMSBMNRKP-UHFFFAOYSA-N carvacrol Natural products CC(=C)C1=CC=C(C)C(O)=C1 HHTWOMMSBMNRKP-UHFFFAOYSA-N 0.000 claims description 12
- RECUKUPTGUEGMW-UHFFFAOYSA-N carvacrol Chemical compound CC(C)C1=CC=C(C)C(O)=C1 RECUKUPTGUEGMW-UHFFFAOYSA-N 0.000 claims description 12
- 235000007746 carvacrol Nutrition 0.000 claims description 12
- WYXXLXHHWYNKJF-UHFFFAOYSA-N isocarvacrol Natural products CC(C)C1=CC=C(O)C(C)=C1 WYXXLXHHWYNKJF-UHFFFAOYSA-N 0.000 claims description 12
- 235000013311 vegetables Nutrition 0.000 claims description 12
- VZCCETWTMQHEPK-UHFFFAOYSA-N gamma-Linolensaeure Natural products CCCCCC=CCC=CCC=CCCCCC(O)=O VZCCETWTMQHEPK-UHFFFAOYSA-N 0.000 claims description 11
- 241000721662 Juniperus Species 0.000 claims description 10
- 235000004429 Matricaria chamomilla var recutita Nutrition 0.000 claims description 10
- 244000179970 Monarda didyma Species 0.000 claims description 10
- 235000010672 Monarda didyma Nutrition 0.000 claims description 10
- 235000020661 alpha-linolenic acid Nutrition 0.000 claims description 10
- VZCCETWTMQHEPK-QNEBEIHSSA-N gamma-linolenic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/CCCCC(O)=O VZCCETWTMQHEPK-QNEBEIHSSA-N 0.000 claims description 10
- 229960004488 linolenic acid Drugs 0.000 claims description 10
- 244000000231 Sesamum indicum Species 0.000 claims description 9
- 235000003434 Sesamum indicum Nutrition 0.000 claims description 9
- 235000020664 gamma-linolenic acid Nutrition 0.000 claims description 9
- 229960002733 gamolenic acid Drugs 0.000 claims description 9
- OYHQOLUKZRVURQ-HZJYTTRNSA-N Linoleic acid Chemical compound CCCCC\C=C/C\C=C/CCCCCCCC(O)=O OYHQOLUKZRVURQ-HZJYTTRNSA-N 0.000 claims description 8
- 231100000439 acute liver injury Toxicity 0.000 claims description 7
- 229960002969 oleic acid Drugs 0.000 claims description 7
- 235000021313 oleic acid Nutrition 0.000 claims description 7
- 235000020778 linoleic acid Nutrition 0.000 claims description 6
- OYHQOLUKZRVURQ-IXWMQOLASA-N linoleic acid Natural products CCCCC\C=C/C\C=C\CCCCCCCC(O)=O OYHQOLUKZRVURQ-IXWMQOLASA-N 0.000 claims description 6
- 208000022309 Alcoholic Liver disease Diseases 0.000 claims description 5
- 206010014513 Embolism arterial Diseases 0.000 claims description 5
- 206010003827 Autoimmune hepatitis Diseases 0.000 claims description 4
- 239000011248 coating agent Substances 0.000 claims description 4
- 238000000576 coating method Methods 0.000 claims description 4
- 235000020224 almond Nutrition 0.000 claims description 3
- 208000002672 hepatitis B Diseases 0.000 claims description 3
- QXJSBBXBKPUZAA-CMDGGOBGSA-N (e)-octadec-10-enoic acid Chemical compound CCCCCCC\C=C\CCCCCCCCC(O)=O QXJSBBXBKPUZAA-CMDGGOBGSA-N 0.000 claims description 2
- 230000001684 chronic effect Effects 0.000 claims description 2
- 229940079593 drug Drugs 0.000 claims description 2
- 150000004676 glycans Chemical class 0.000 claims description 2
- 229920001282 polysaccharide Polymers 0.000 claims description 2
- 239000005017 polysaccharide Substances 0.000 claims description 2
- 241000218236 Cannabis Species 0.000 claims 2
- 210000004556 brain Anatomy 0.000 claims 1
- -1 gamma-linolenic acid fatty acid Chemical class 0.000 claims 1
- 239000012948 isocyanate Substances 0.000 claims 1
- 150000002513 isocyanates Chemical class 0.000 claims 1
- 235000007232 Matricaria chamomilla Nutrition 0.000 abstract description 7
- 241000207199 Citrus Species 0.000 abstract description 6
- 235000017945 Matricaria Nutrition 0.000 abstract description 6
- 235000020971 citrus fruits Nutrition 0.000 abstract description 6
- 230000006806 disease prevention Effects 0.000 abstract description 5
- 238000002360 preparation method Methods 0.000 abstract description 4
- 244000192528 Chrysanthemum parthenium Species 0.000 abstract 1
- 244000301850 Cupressus sempervirens Species 0.000 abstract 1
- 230000000694 effects Effects 0.000 description 48
- 108010082126 Alanine transaminase Proteins 0.000 description 34
- 102100036475 Alanine aminotransferase 1 Human genes 0.000 description 33
- 241000699670 Mus sp. Species 0.000 description 25
- 238000000034 method Methods 0.000 description 19
- 108010074328 Interferon-gamma Proteins 0.000 description 14
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 14
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 14
- 210000004185 liver Anatomy 0.000 description 14
- 102000003814 Interleukin-10 Human genes 0.000 description 13
- 108090000174 Interleukin-10 Proteins 0.000 description 13
- 229940076144 interleukin-10 Drugs 0.000 description 13
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 description 12
- 102100037850 Interferon gamma Human genes 0.000 description 12
- 239000000463 material Substances 0.000 description 12
- 238000012360 testing method Methods 0.000 description 12
- 239000008168 almond oil Substances 0.000 description 11
- 210000004369 blood Anatomy 0.000 description 11
- 239000008280 blood Substances 0.000 description 11
- 239000003094 microcapsule Substances 0.000 description 11
- 108010003415 Aspartate Aminotransferases Proteins 0.000 description 10
- 102000004625 Aspartate Aminotransferases Human genes 0.000 description 10
- 208000024172 Cardiovascular disease Diseases 0.000 description 10
- 210000005228 liver tissue Anatomy 0.000 description 10
- GMVPRGQOIOIIMI-UHFFFAOYSA-N (8R,11R,12R,13E,15S)-11,15-Dihydroxy-9-oxo-13-prostenoic acid Natural products CCCCCC(O)C=CC1C(O)CC(=O)C1CCCCCCC(O)=O GMVPRGQOIOIIMI-UHFFFAOYSA-N 0.000 description 9
- 244000025254 Cannabis sativa Species 0.000 description 9
- 108010062580 Concanavalin A Proteins 0.000 description 9
- 229960000711 alprostadil Drugs 0.000 description 9
- 235000009120 camo Nutrition 0.000 description 9
- 235000005607 chanvre indien Nutrition 0.000 description 9
- 208000019423 liver disease Diseases 0.000 description 9
- 239000003921 oil Substances 0.000 description 9
- 235000019198 oils Nutrition 0.000 description 9
- GMVPRGQOIOIIMI-DWKJAMRDSA-N prostaglandin E1 Chemical compound CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O GMVPRGQOIOIIMI-DWKJAMRDSA-N 0.000 description 9
- XEYBRNLFEZDVAW-UHFFFAOYSA-N prostaglandin E2 Natural products CCCCCC(O)C=CC1C(O)CC(=O)C1CC=CCCCC(O)=O XEYBRNLFEZDVAW-UHFFFAOYSA-N 0.000 description 9
- 239000000243 solution Substances 0.000 description 9
- 235000012766 Cannabis sativa ssp. sativa var. sativa Nutrition 0.000 description 8
- 235000012765 Cannabis sativa ssp. sativa var. spontanea Nutrition 0.000 description 8
- 239000011487 hemp Substances 0.000 description 8
- 208000006454 hepatitis Diseases 0.000 description 8
- 231100000283 hepatitis Toxicity 0.000 description 8
- 102000004127 Cytokines Human genes 0.000 description 7
- 108090000695 Cytokines Proteins 0.000 description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 7
- 238000010171 animal model Methods 0.000 description 7
- 229960004232 linoleic acid Drugs 0.000 description 7
- 240000004308 marijuana Species 0.000 description 7
- 235000015097 nutrients Nutrition 0.000 description 7
- 235000011803 sesame oil Nutrition 0.000 description 7
- 239000008159 sesame oil Substances 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- 108010001202 Cytochrome P-450 CYP2E1 Proteins 0.000 description 6
- 102100024889 Cytochrome P450 2E1 Human genes 0.000 description 6
- 241000699666 Mus <mouse, genus> Species 0.000 description 6
- 238000012742 biochemical analysis Methods 0.000 description 6
- 230000037396 body weight Effects 0.000 description 6
- 239000000284 extract Substances 0.000 description 6
- 235000013305 food Nutrition 0.000 description 6
- 238000004817 gas chromatography Methods 0.000 description 6
- 231100000753 hepatic injury Toxicity 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 6
- 108010081668 Cytochrome P-450 CYP3A Proteins 0.000 description 5
- 102000004328 Cytochrome P-450 CYP3A Human genes 0.000 description 5
- 238000012449 Kunming mouse Methods 0.000 description 5
- 230000001640 apoptogenic effect Effects 0.000 description 5
- 210000001367 artery Anatomy 0.000 description 5
- 150000001875 compounds Chemical class 0.000 description 5
- 239000002158 endotoxin Substances 0.000 description 5
- 239000010460 hemp oil Substances 0.000 description 5
- 210000003494 hepatocyte Anatomy 0.000 description 5
- 229920006008 lipopolysaccharide Polymers 0.000 description 5
- 238000003305 oral gavage Methods 0.000 description 5
- 230000000770 proinflammatory effect Effects 0.000 description 5
- 230000001681 protective effect Effects 0.000 description 5
- 239000012086 standard solution Substances 0.000 description 5
- 230000001225 therapeutic effect Effects 0.000 description 5
- 238000002965 ELISA Methods 0.000 description 4
- 238000008157 ELISA kit Methods 0.000 description 4
- 101000653005 Homo sapiens Thromboxane-A synthase Proteins 0.000 description 4
- 206010061218 Inflammation Diseases 0.000 description 4
- 244000184734 Pyrus japonica Species 0.000 description 4
- 102100030973 Thromboxane-A synthase Human genes 0.000 description 4
- YZXBAPSDXZZRGB-DOFZRALJSA-N arachidonic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O YZXBAPSDXZZRGB-DOFZRALJSA-N 0.000 description 4
- 235000004251 balanced diet Nutrition 0.000 description 4
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 4
- 210000000805 cytoplasm Anatomy 0.000 description 4
- 230000036541 health Effects 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 230000002440 hepatic effect Effects 0.000 description 4
- 230000004054 inflammatory process Effects 0.000 description 4
- 230000028709 inflammatory response Effects 0.000 description 4
- VNYSSYRCGWBHLG-AMOLWHMGSA-N leukotriene B4 Chemical compound CCCCC\C=C/C[C@@H](O)\C=C\C=C\C=C/[C@@H](O)CCCC(O)=O VNYSSYRCGWBHLG-AMOLWHMGSA-N 0.000 description 4
- GWNVDXQDILPJIG-NXOLIXFESA-N leukotriene C4 Chemical compound CCCCC\C=C/C\C=C/C=C/C=C/[C@H]([C@@H](O)CCCC(O)=O)SC[C@@H](C(=O)NCC(O)=O)NC(=O)CC[C@H](N)C(O)=O GWNVDXQDILPJIG-NXOLIXFESA-N 0.000 description 4
- 150000002617 leukotrienes Chemical class 0.000 description 4
- 229930003658 monoterpene Natural products 0.000 description 4
- 239000003981 vehicle Substances 0.000 description 4
- 201000001320 Atherosclerosis Diseases 0.000 description 3
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 3
- 108020004206 Gamma-glutamyltransferase Proteins 0.000 description 3
- 208000018522 Gastrointestinal disease Diseases 0.000 description 3
- 101000629400 Homo sapiens Mesoderm-specific transcript homolog protein Proteins 0.000 description 3
- 102100026821 Mesoderm-specific transcript homolog protein Human genes 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- 206010028851 Necrosis Diseases 0.000 description 3
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- 210000004027 cell Anatomy 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 230000002950 deficient Effects 0.000 description 3
- 208000035475 disorder Diseases 0.000 description 3
- 235000004626 essential fatty acids Nutrition 0.000 description 3
- 102000006640 gamma-Glutamyltransferase Human genes 0.000 description 3
- 238000007490 hematoxylin and eosin (H&E) staining Methods 0.000 description 3
- 235000021268 hot food Nutrition 0.000 description 3
- 230000002757 inflammatory effect Effects 0.000 description 3
- 239000004615 ingredient Substances 0.000 description 3
- 239000012528 membrane Substances 0.000 description 3
- 150000002773 monoterpene derivatives Chemical class 0.000 description 3
- 230000017074 necrotic cell death Effects 0.000 description 3
- 210000004940 nucleus Anatomy 0.000 description 3
- 210000000056 organ Anatomy 0.000 description 3
- 102000004169 proteins and genes Human genes 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 2
- FPIPGXGPPPQFEQ-UHFFFAOYSA-N 13-cis retinol Natural products OCC=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-UHFFFAOYSA-N 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 2
- 239000001828 Gelatine Substances 0.000 description 2
- WZUVPPKBWHMQCE-UHFFFAOYSA-N Haematoxylin Chemical compound C12=CC(O)=C(O)C=C2CC2(O)C1C1=CC=C(O)C(O)=C1OC2 WZUVPPKBWHMQCE-UHFFFAOYSA-N 0.000 description 2
- 102000008070 Interferon-gamma Human genes 0.000 description 2
- 208000005764 Peripheral Arterial Disease Diseases 0.000 description 2
- 208000030831 Peripheral arterial occlusive disease Diseases 0.000 description 2
- 241000283984 Rodentia Species 0.000 description 2
- 101100545004 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) YSP2 gene Proteins 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 239000006180 TBST buffer Substances 0.000 description 2
- 230000003110 anti-inflammatory effect Effects 0.000 description 2
- 235000021342 arachidonic acid Nutrition 0.000 description 2
- 229940114079 arachidonic acid Drugs 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- 229940098773 bovine serum albumin Drugs 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 235000012000 cholesterol Nutrition 0.000 description 2
- 208000037976 chronic inflammation Diseases 0.000 description 2
- 230000006020 chronic inflammation Effects 0.000 description 2
- 235000020940 control diet Nutrition 0.000 description 2
- 208000029078 coronary artery disease Diseases 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- RRAFCDWBNXTKKO-UHFFFAOYSA-N eugenol Chemical compound COC1=CC(CC=C)=CC=C1O RRAFCDWBNXTKKO-UHFFFAOYSA-N 0.000 description 2
- 239000003205 fragrance Substances 0.000 description 2
- 230000005714 functional activity Effects 0.000 description 2
- 229940098330 gamma linoleic acid Drugs 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- WTEVQBCEXWBHNA-JXMROGBWSA-N geranial Chemical compound CC(C)=CCC\C(C)=C\C=O WTEVQBCEXWBHNA-JXMROGBWSA-N 0.000 description 2
- 231100000234 hepatic damage Toxicity 0.000 description 2
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 2
- 206010020718 hyperplasia Diseases 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 229960003130 interferon gamma Drugs 0.000 description 2
- CDOSHBSSFJOMGT-UHFFFAOYSA-N linalool Chemical compound CC(C)=CCCC(C)(O)C=C CDOSHBSSFJOMGT-UHFFFAOYSA-N 0.000 description 2
- UWKAYLJWKGQEPM-LBPRGKRZSA-N linalyl acetate Chemical compound CC(C)=CCC[C@](C)(C=C)OC(C)=O UWKAYLJWKGQEPM-LBPRGKRZSA-N 0.000 description 2
- 230000008818 liver damage Effects 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 235000002577 monoterpenes Nutrition 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 235000015639 rosmarinus officinalis Nutrition 0.000 description 2
- 235000010413 sodium alginate Nutrition 0.000 description 2
- 239000000661 sodium alginate Substances 0.000 description 2
- 229940005550 sodium alginate Drugs 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 238000007619 statistical method Methods 0.000 description 2
- 229940124597 therapeutic agent Drugs 0.000 description 2
- 230000009261 transgenic effect Effects 0.000 description 2
- 210000003462 vein Anatomy 0.000 description 2
- 230000003442 weekly effect Effects 0.000 description 2
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 2
- GRWFGVWFFZKLTI-IUCAKERBSA-N (-)-α-pinene Chemical compound CC1=CC[C@@H]2C(C)(C)[C@H]1C2 GRWFGVWFFZKLTI-IUCAKERBSA-N 0.000 description 1
- 239000001490 (3R)-3,7-dimethylocta-1,6-dien-3-ol Substances 0.000 description 1
- CDOSHBSSFJOMGT-JTQLQIEISA-N (R)-linalool Natural products CC(C)=CCC[C@@](C)(O)C=C CDOSHBSSFJOMGT-JTQLQIEISA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- 208000020154 Acnes Diseases 0.000 description 1
- 235000019489 Almond oil Nutrition 0.000 description 1
- 235000003840 Amygdalus nana Nutrition 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- 206010002329 Aneurysm Diseases 0.000 description 1
- 208000023275 Autoimmune disease Diseases 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- 235000008697 Cannabis sativa Nutrition 0.000 description 1
- NPBVQXIMTZKSBA-UHFFFAOYSA-N Chavibetol Natural products COC1=CC=C(CC=C)C=C1O NPBVQXIMTZKSBA-UHFFFAOYSA-N 0.000 description 1
- 108010077544 Chromatin Proteins 0.000 description 1
- 208000017667 Chronic Disease Diseases 0.000 description 1
- WTEVQBCEXWBHNA-UHFFFAOYSA-N Citral Natural products CC(C)=CCCC(C)=CC=O WTEVQBCEXWBHNA-UHFFFAOYSA-N 0.000 description 1
- 241000468081 Citrus bergamia Species 0.000 description 1
- 241000675108 Citrus tangerina Species 0.000 description 1
- 241000186216 Corynebacterium Species 0.000 description 1
- 241000186427 Cutibacterium acnes Species 0.000 description 1
- 201000004624 Dermatitis Diseases 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- 208000005189 Embolism Diseases 0.000 description 1
- 239000005770 Eugenol Substances 0.000 description 1
- 208000007882 Gastritis Diseases 0.000 description 1
- 208000005577 Gastroenteritis Diseases 0.000 description 1
- 241000134874 Geraniales Species 0.000 description 1
- 208000000857 Hepatic Insufficiency Diseases 0.000 description 1
- 208000027761 Hepatic autoimmune disease Diseases 0.000 description 1
- 206010019663 Hepatic failure Diseases 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 208000026350 Inborn Genetic disease Diseases 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 241000592238 Juniperus communis Species 0.000 description 1
- 108010028554 LDL Cholesterol Proteins 0.000 description 1
- 108010007622 LDL Lipoproteins Proteins 0.000 description 1
- 102000007330 LDL Lipoproteins Human genes 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 241000219925 Oenothera Species 0.000 description 1
- 235000004496 Oenothera biennis Nutrition 0.000 description 1
- 235000010677 Origanum vulgare Nutrition 0.000 description 1
- 239000002033 PVDF binder Substances 0.000 description 1
- 235000021314 Palmitic acid Nutrition 0.000 description 1
- 235000019483 Peanut oil Nutrition 0.000 description 1
- 208000008469 Peptic Ulcer Diseases 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
- 241000220299 Prunus Species 0.000 description 1
- 235000011432 Prunus Nutrition 0.000 description 1
- UVMRYBDEERADNV-UHFFFAOYSA-N Pseudoeugenol Natural products COC1=CC(C(C)=C)=CC=C1O UVMRYBDEERADNV-UHFFFAOYSA-N 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 208000007107 Stomach Ulcer Diseases 0.000 description 1
- 208000007536 Thrombosis Diseases 0.000 description 1
- 102000003929 Transaminases Human genes 0.000 description 1
- FPIPGXGPPPQFEQ-BOOMUCAASA-N Vitamin A Natural products OC/C=C(/C)\C=C\C=C(\C)/C=C/C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-BOOMUCAASA-N 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 208000026594 alcoholic fatty liver disease Diseases 0.000 description 1
- 150000001338 aliphatic hydrocarbons Chemical group 0.000 description 1
- 229940087168 alpha tocopherol Drugs 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 230000002429 anti-coagulating effect Effects 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 238000000222 aromatherapy Methods 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 244000213578 camo Species 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000002038 chemiluminescence detection Methods 0.000 description 1
- BHQCQFFYRZLCQQ-OELDTZBJSA-N cholic acid Chemical compound C([C@H]1C[C@H]2O)[C@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@@H](CCC(O)=O)C)[C@@]2(C)[C@@H](O)C1 BHQCQFFYRZLCQQ-OELDTZBJSA-N 0.000 description 1
- 210000003483 chromatin Anatomy 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 208000019425 cirrhosis of liver Diseases 0.000 description 1
- 206010009887 colitis Diseases 0.000 description 1
- 239000007891 compressed tablet Substances 0.000 description 1
- 238000002591 computed tomography Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 208000010643 digestive system disease Diseases 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 208000010227 enterocolitis Diseases 0.000 description 1
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 1
- 230000002327 eosinophilic effect Effects 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 235000020774 essential nutrients Nutrition 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 229960002217 eugenol Drugs 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 208000010706 fatty liver disease Diseases 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 201000005917 gastric ulcer Diseases 0.000 description 1
- 208000018685 gastrointestinal system disease Diseases 0.000 description 1
- 238000001502 gel electrophoresis Methods 0.000 description 1
- 208000016361 genetic disease Diseases 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- ZFGMDIBRIDKWMY-PASTXAENSA-N heparin Chemical compound CC(O)=N[C@@H]1[C@@H](O)[C@H](O)[C@@H](COS(O)(=O)=O)O[C@@H]1O[C@@H]1[C@@H](C(O)=O)O[C@@H](O[C@H]2[C@@H]([C@@H](OS(O)(=O)=O)[C@@H](O[C@@H]3[C@@H](OC(O)[C@H](OS(O)(=O)=O)[C@H]3O)C(O)=O)O[C@@H]2O)CS(O)(=O)=O)[C@H](O)[C@H]1O ZFGMDIBRIDKWMY-PASTXAENSA-N 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 238000003119 immunoblot Methods 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 210000004969 inflammatory cell Anatomy 0.000 description 1
- 208000027866 inflammatory disease Diseases 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 210000001865 kupffer cell Anatomy 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 229930007744 linalool Natural products 0.000 description 1
- UWKAYLJWKGQEPM-UHFFFAOYSA-N linalool acetate Natural products CC(C)=CCCC(C)(C=C)OC(C)=O UWKAYLJWKGQEPM-UHFFFAOYSA-N 0.000 description 1
- 201000007270 liver cancer Diseases 0.000 description 1
- 210000005229 liver cell Anatomy 0.000 description 1
- 208000014018 liver neoplasm Diseases 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000003760 magnetic stirring Methods 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 208000031225 myocardial ischemia Diseases 0.000 description 1
- WQEPLUUGTLDZJY-UHFFFAOYSA-N n-Pentadecanoic acid Natural products CCCCCCCCCCCCCCC(O)=O WQEPLUUGTLDZJY-UHFFFAOYSA-N 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 231100000915 pathological change Toxicity 0.000 description 1
- 230000036285 pathological change Effects 0.000 description 1
- 239000000312 peanut oil Substances 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 208000011906 peptic ulcer disease Diseases 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229920000747 poly(lactic acid) Polymers 0.000 description 1
- 229920001606 poly(lactic acid-co-glycolic acid) Polymers 0.000 description 1
- 239000004626 polylactic acid Substances 0.000 description 1
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 235000014774 prunus Nutrition 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 208000037803 restenosis Diseases 0.000 description 1
- 229960003471 retinol Drugs 0.000 description 1
- 235000020944 retinol Nutrition 0.000 description 1
- 239000011607 retinol Substances 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 235000003441 saturated fatty acids Nutrition 0.000 description 1
- 150000004671 saturated fatty acids Chemical class 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 235000020183 skimmed milk Nutrition 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 229960000984 tocofersolan Drugs 0.000 description 1
- 238000003325 tomography Methods 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 description 1
- 235000021122 unsaturated fatty acids Nutrition 0.000 description 1
- 150000004670 unsaturated fatty acids Chemical class 0.000 description 1
- 210000003934 vacuole Anatomy 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 235000019155 vitamin A Nutrition 0.000 description 1
- 239000011719 vitamin A Substances 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
- 229940045997 vitamin a Drugs 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
- 239000002076 α-tocopherol Substances 0.000 description 1
- 235000004835 α-tocopherol Nutrition 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/75—Rutaceae (Rue family)
- A61K36/752—Citrus, e.g. lime, orange or lemon
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/53—Lamiaceae or Labiatae (Mint family), e.g. thyme, rosemary or lavender
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/115—Fatty acids or derivatives thereof; Fats or oils
- A23L33/12—Fatty acids or derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/01—Hydrocarbons
- A61K31/015—Hydrocarbons carbocyclic
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/20—Carboxylic acids, e.g. valproic acid having a carboxyl group bound to a chain of seven or more carbon atoms, e.g. stearic, palmitic, arachidic acids
- A61K31/201—Carboxylic acids, e.g. valproic acid having a carboxyl group bound to a chain of seven or more carbon atoms, e.g. stearic, palmitic, arachidic acids having one or two double bonds, e.g. oleic, linoleic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/13—Coniferophyta (gymnosperms)
- A61K36/14—Cupressaceae (Cypress family), e.g. juniper or cypress
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/28—Asteraceae or Compositae (Aster or Sunflower family), e.g. chamomile, feverfew, yarrow or echinacea
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/28—Asteraceae or Compositae (Aster or Sunflower family), e.g. chamomile, feverfew, yarrow or echinacea
- A61K36/287—Chrysanthemum, e.g. daisy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/02—Antithrombotic agents; Anticoagulants; Platelet aggregation inhibitors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
Landscapes
- Health & Medical Sciences (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Mycology (AREA)
- Botany (AREA)
- Alternative & Traditional Medicine (AREA)
- Microbiology (AREA)
- Medical Informatics (AREA)
- Biotechnology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Nutrition Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Food Science & Technology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Polymers & Plastics (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Diabetes (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Hematology (AREA)
- Gastroenterology & Hepatology (AREA)
- Physiology (AREA)
- Medicines Containing Plant Substances (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
| 组分 | 基于组合物中的脂肪酸的总量的量 |
| 棕榈酸 | 6.2% |
| 硬脂酸 | 2.7% |
| 油酸 | 42% |
| 异油酸 | 1.5% |
| 亚油酸 | 34% |
| α-亚油酸 | 3.5% |
| γ-亚油酸 | 1.7% |
| 维生素A (视黄醇) | <100 IU/kg |
| 维生素E (α-生育酚) | 193 ppm |
| 组分 | 基于口服药物组合物的总量的量 |
| a-蒎烯 | 5.8% |
| 柠檬烯 | 9.66% |
| 1,8-桉树脑 | 9.34% |
| 沉香醇 | 4.60% |
| 橙花醛 | 4.74% |
| 乙酸芳樟酯 | 6.23% |
| 香叶醛 | 7.17% |
| 香芹酚 | 8.05% |
| 丁香酚 | 5.63% |
| 反式-β-femesene | 4.41% |
| 组 | ALT (mU/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 1.444±0.499 | 100.0 | 42.7 |
| 模型 | 3.382±1.137## | 234.2 | 100.0 |
| Ah | 1.971±0.861* | 136.5 | 58.3 |
| Am | 1.620±0.691** | 112.2 | 47.9 |
| Al | 2.982±1.068 | 206.5 | 88.2 |
| 组 | LTB4 (ng/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 0.275±0.046 | 100.0 | 51.6 |
| 模型 | 0.532±0.655## | 193.5 | 100.0 |
| Ah | 0.266±0.957** | 136.5 | 50.0 |
| Am | 0.315±0.976** | 96.7 | 59.2 |
| Al | 0.347±0.156** | 126.2 | 65.2 |
| 组 | LTC4 (ng/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 1.380±0.221 | 100.0 | 75.9 |
| 模型 | 1.817±0.102## | 131.7 | 100.0 |
| Ah | 1.472±0.094** | 106.7 | 81.0 |
| Am | 1.531±0.215* | 110.9 | 84.3 |
| Al | 1.526±0.198* | 110.6 | 84.0 |
| 组 | ALT (mU/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 1.437±0.074 | ------ | 18.53 |
| 模型 | 7.756±0.319 ## | 539.7 | ------ |
| Ah | 3.425±0.108 ** | 238.3 | 44.16 |
| Am | 3.086±0.203 ** | 214.8 | 39.79 |
| Al | 2.731±0.169 ** | 190.0 | 35.21 |
| A100h | 6.668±0.074 ** | 464.0 | 85.97 |
| A100m | 6.495±0.154 ** | 452.0 | 83.74 |
| A100l | 5.603±0.127 ** | 389.9 | 72.24 |
| 组 | IL-10 (pg/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 103.80±7.12 | ------ | 108.54 |
| 模型 | 95.63±5.82 | 92.13 | ------ |
| Ah | 112.69±3.15* | 108.56 | 117.84 |
| Am | 111.04±7.30* | 106.97 | 116.11 |
| Al | 111.15±1.29* | 107.08 | 116.23 |
| A100h | 98.06±1.94 | 94.47 | 102.54 |
| A100m | 99.50±6.79 | 95.86 | 104.05 |
| A100l | 102.06±3.43 | 98.32 | 106.72 |
| 组 | IFN- γ (pg/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 32.95±2.49 | ------ | 40.39 |
| 模型 | 81.58±2.52## | 247.58 | ------ |
| Ah | 43.90±4.97** | 133.23 | 53.81 |
| Am | 62.34±3.27** | 189.19 | 76.42 |
| Al | 54.24±10.89** | 164.61 | 66.49 |
| A100h | 62.29±4.19** | 189.04 | 76.35 |
| A100m | 73.56±10.22 | 223.25 | 90.17 |
| A100l | 77.13±8.15 | 234.08 | 94.54 |
| 组 | TNF-α (pg/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 317.93±24.37 | ------ | 61.83 |
| 模型 | 514.16±21.94## | 161.72 | ------ |
| Ah | 379.77±25.43** | 119.45 | 73.86 |
| Am | 477.76±7.77* | 150.28 | 92.92 |
| Al | 502.12±14.62 | 157.94 | 97.66 |
| A100h | 467.09±8.33** | 146.92 | 90.84 |
| A100m | 484.53±25.08 | 152.40 | 94.23 |
| A100l | 480.48±23.90 | 151.13 | 93.45 |
| 组 | TG (mmol/L) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 1.34±0.10 | ------ | 79.25 |
| 模型 | 1.69±0.15## | 126.18 | ------ |
| Ah | 1.44±0.06** | 107.98 | 85.57 |
| Am | 1.38±0.10** | 103.49 | 82.02 |
| Al | 1.50±0.06* | 112.47 | 88.34 |
| A100h | 1.45±0.04 | 108.48 | 89.13 |
| A100m | 1.66±0.13 | 120.20 | 85.97 |
| A100l | 1.56±0.08 | 116.96 | 95.26 |
| 组 | TC (mmol/L) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 2.94±0.29 | ------ | 60.19 |
| 模型 | 4.89±0.30## | 166.32 | ------ |
| Ah | 3.63±0.42** | 123.47 | 74.23 |
| Am | 4.13±0.07** | 140.48 | 84.46 |
| Al | 4.32±0.19* | 146.94 | 88.34 |
| A100h | 4.93±0.07 | 167.80 | 100.89 |
| A100m | 4.77±0.56 | 162.47 | 97.68 |
| A100l | 5.08±0.11 | 172.90 | 103.95 |
| 组 | PGE1 (pg/L)) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 803.28±82.84 | ------ | 61.16 |
| 模型 | 1313.36±101.77## | 163.50 | ------ |
| Ah | 920.73±16.52** | 114.62 | 70.11 |
| Am | 1022.93±57.61** | 127.34 | 77.89 |
| Al | 1079.04±54.24** | 134.33 | 82.16 |
| A100h | 1324.86±66.37 | 164.93 | 100.87 |
| A100m | 1293.30±47.69 | 161.00 | 98.47 |
| A100l | 1342.67±75.40 | 167.15 | 102.23 |
| 组 | ALT (mU/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 1.503±0.116 | ------ | 11.53 |
| 模型 | 13.03±0.204## | 866.9 | ------ |
| Ah | 10.83±0.249** | 720.6 | 83.12 |
| Am | 5.501±0.412** | 366.0 | 42.22 |
| Al | 4.856±0.363** | 323.1 | 37.27 |
| A100h | 12.80±0.262 | 851.6 | 98.23 |
| A100m | 12.21±0.634 | 812.4 | 93.71 |
| A100l | 10.30±0.578** | 685.3 | 79.05 |
| 组 | IL-10 (pg/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 100.38±2.93 | ------ | 583.27 |
| 模型 | 17.21±4.70## | 17.14 | ------ |
| Ah | 80.06±2.72** | 79.75 | 465.19 |
| Am | 55.93±9.50** | 55.72 | 324,99 |
| Al | 51.94±3.17** | 51.75 | 301.80 |
| A100h | 19.25±1.64 | 19.17 | 111.85 |
| A100m | 48.92±10.66** | 48.73 | 284.25 |
| A100l | 25.38±6.88* | 25.28 | 147.47 |
| 组 | IFN- γ (pg/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 34.59±5.10 | ------ | 40.00 |
| 模型 | 86.47±5.46## | 249.99 | ------ |
| Ah | 39.64±6.24** | 114.60 | 45.84 |
| Am | 40.47±2.96** | 117.00 | 46.80 |
| Al | 82.80±3.54** | 239.38 | 95.76 |
| A100h | 80.79±13.41 | 233.56 | 93.43 |
| A100m | 77.49±3.39* | 224.02 | 89.61 |
| A100l | 80.04±6.44 | 231.40 | 92.56 |
| 组 | TNF-α (pg/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 320.17±17.67 | ------ | 28.43 |
| 模型 | 1126.17±121.69## | 351.74 | ------ |
| Ah | 750.99±72.96** | 234.56 | 66.69 |
| Am | 769.86±57.98** | 240.45 | 68.36 |
| Al | 749.27±12.00** | 234.02 | 66.53 |
| A100h | 949.53±191.44 | 296.59 | 84.32 |
| A100m | 985.94±84.43 | 307.94 | 87.54 |
| A100l | 1108.54±66.40 | 346.23 | 98.43 |
| 组 | TG (mmol/L) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 1.40±0.07 | ------ | 101.95 |
| 模型 | 1.37±0.08 | 98.09 | ------ |
| Ah | 1.40±0.03 | 100.00 | 101.95 |
| Am | 1.33±0.10 | 94.99 | 96.83 |
| Al | 1.34±0.04 | 95.70 | 97.57 |
| A100h | 1.35±0.06 | 96.90 | 98.78 |
| A100m | 1.39±0.03 | 99.76 | 101.70 |
| A100l | 1.45±0.07 | 103.58 | 105.60 |
| 组 | TC (mmol/L) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 2.98±0.03 | ------ | 100.00 |
| 模型 | 2.98±0.11 | 100.00 | ------ |
| Ah | 3.05±0.04 | 102.35 | 102.35 |
| Am | 3.00±0.11 | 100.67 | 100.67 |
| Al | 2.89±0.10 | 96.87 | 96.87 |
| A100h | 3.00±0.03 | 97.20 | 97.20 |
| A100m | 2.94±0.03 | 98.55 | 98.55 |
| A100l | 3.02±0.04 | 101.45 | 101.45 |
| 组 | PGE1 (pg/L)) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 820.69±104.35 | ------ | 56.23 |
| 模型 | 1459.54±86.97## | 177.84 | ------ |
| Ah | 1004.04±88.59** | 122.34 | 68.79 |
| Am | 1003.29±66.77** | 122.25 | 68.74 |
| Al | 1249.37±44.75* | 152.23 | 85.60 |
| A100h | 1438.69±51.44 | 175.30 | 98.57 |
| A100m | 1452.69±121.76 | 177.01 | 99.53 |
| A100l | 1448.37±124.83 | 176.48 | 99.23 |
| 组 | ALT (mU/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 1.421±0.134 | ------ | 32.58 |
| 模型 | 4.361±0.198## | 306.9 | ------ |
| Ah | 1.958±0.223** | 137.8 | 44.90 |
| Am | 1.974±0.092** | 138.9 | 45.26 |
| Al | 1.883±0.479** | 132.5 | 43.18 |
| A100h | 4.023±0.229 | 283.1 | 92.25 |
| A100m | 3.126±0.175** | 220.0 | 71.68 |
| A100l | 3.075±0.162** | 216.4 | 70.51 |
| 组 | IL-10 (pg/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 98.29±8.57 | ------ | 521.98 |
| 模型 | 18.83±6.89## | 19.16 | ------ |
| Ah | 69.56±6.67** | 70.77 | 369.41 |
| Am | 56.04±2.91** | 57.01 | 297.61 |
| Al | 58.33±6.66** | 59.34 | 309.77 |
| A100h | 16.19±2.67 | 16.47 | 85.97 |
| A100m | 19.48±3.65 | 19.82 | 103.45 |
| A100l | 19.98±1.76 | 20.32 | 106.10 |
| 组 | IFN- γ (pg/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 33.01±3.97 | ------ | 25.25 |
| 模型 | 130.71±10.00## | 395.97 | ------ |
| Ah | 60.83±8.10** | 184.28 | 46.54 |
| Am | 72.89±5.42** | 220.81 | 55.76 |
| Al | 74.22±3.22** | 224.84 | 56.78 |
| A100h | 118.18±5.97 | 358.01 | 90.41 |
| A100m | 98.61±6.25* | 298.73 | 75.44 |
| A100l | 109.38±12.09 | 331.35 | 83.68 |
| 组 | TNF-α (pg/mL) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 316.20±20.20 | ------ | |
| 模型 | 447.73±25.50## | 141.59 | ------ |
| Ah | 360.49±19.12** | 114.01 | 80.51 |
| Am | 356.73±8.74** | 131.14 | 79.90 |
| Al | 416.47±4.44 | 131.71 | 93.02 |
| A100h | 407.79±14.04* | 128.97 | 91.08 |
| A100m | 458.83±19.34 | 145.11 | 102.48 |
| A100l | 433.71±23.45 | 137.17 | 96.87 |
| 组 | TG (mmol/L) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 1.39±0.02 | ------ | 74.06 |
| 模型 | 1.88±0.10## | 135.01 | ------ |
| Ah | 1.42±0.10** | 102.64 | 76.02 |
| Am | 1.59±0.06** | 114.87 | 85.08 |
| Al | 1.55±0.06 | 111.51 | 82.59 |
| A100h | 1.66±0.07 | 119.66 | 88.63 |
| A100m | 1.92±0.04 | 137.89 | 102.13 |
| A100l | 1.86±0.02 | 134.05 | 99.29 |
| 组 | TC (mmol/L) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 2.92±0.13 | ------ | 75.25 |
| 模型 | 3.88±0.10## | 132.88 | ------ |
| Ah | 2.97±0.06** | 101.60 | 76.46 |
| Am | 2.88±0.03** | 98.74 | 74.31 |
| Al | 3.66±0.10 | 125.34 | 94.33 |
| A100h | 3.76±0.18 | 128.65 | 96.82 |
| A100m | 3.88±0.03 | 133.00 | 100.09 |
| A100l | 3.91±0.08 | 133.79 | 100.69 |
| 组 | PGE1 (pg/L)) | vs. 对照 (%) | vs. 模型 (%) |
| 对照 | 759.29±70.83 | ------ | 57.70 |
| 模型 | 1315.80±102.72## | 173.29 | ------ |
| Ah | 876.96±33.81** | 115.50 | 66.65 |
| Am | 804.47±33.80** | 105.95 | 61.14 |
| Al | 944.31±74.56** | 124.37 | 71.77 |
| A100h | 1388.65±120.17 | 182.89 | 105.54 |
| A100m | 1261.96±251.81 | 166.20 | 95.91 |
| A100l | 1083.78±62.48* | 142.74 | 82.37 |
Claims (18)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| HK16103839.1 | 2016-04-05 | ||
| HK16103839.1A HK1221598A2 (zh) | 2016-04-05 | 2016-04-05 | 口服药物组合物以及治疗或预防疾病的方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN107320552A CN107320552A (zh) | 2017-11-07 |
| CN107320552B true CN107320552B (zh) | 2022-08-09 |
Family
ID=58773037
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201710217749.5A Active CN107320552B (zh) | 2016-04-05 | 2017-04-05 | 口服药物组合物和其用于制备治疗或预防疾病的药物的用途 |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP3441075B1 (zh) |
| CN (1) | CN107320552B (zh) |
| HK (1) | HK1221598A2 (zh) |
| TW (1) | TWI733792B (zh) |
| WO (1) | WO2017173993A1 (zh) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112569219B (zh) * | 2019-09-30 | 2023-05-02 | 中国科学院上海药物研究所 | 用于治疗动脉相关疾病的药物及其用途 |
| CN116261462B (zh) * | 2020-07-14 | 2024-12-13 | 科然药业有限公司 | 组合物及其用途 |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1475204A (zh) * | 2002-06-30 | 2004-02-18 | 丛繁滋 | 经口、鼻给药的组合物、干粉吸入剂、喷雾剂、喷雾剂辅助制剂及其制备方法 |
| CN101420848A (zh) * | 2006-02-15 | 2009-04-29 | 博塔诺凯普有限公司 | 微胶囊化精油的应用 |
| CN102058892A (zh) * | 2009-11-13 | 2011-05-18 | 玛丽安娜·布朗德伦德 | 以挥发油混合物为基质的物质混合物及其用途 |
| CN102481324A (zh) * | 2009-05-29 | 2012-05-30 | 新纪元公司 | 调节脂质成分的组合物和方法 |
| DE102011078432A1 (de) * | 2011-06-30 | 2013-01-03 | Barbara Engels-Wehr | Verwendung von Monoterpen-Estern zur Behandlung von hyperkinetischen Störungen |
| CN103316108A (zh) * | 2013-05-20 | 2013-09-25 | 钟小强 | 治疗肝炎的复方精油 |
| CN103463549A (zh) * | 2013-09-10 | 2013-12-25 | 季正红 | 一种保护男性心脏的复方精油 |
| WO2015014497A1 (en) * | 2013-08-02 | 2015-02-05 | Schweitzer-Mauduit International, Inc. | Edible product comprising reconstituted plant material |
| EP2878303A1 (de) * | 2013-11-29 | 2015-06-03 | Doris Hefti | Zusammensetzung für die Aromatherapie, Aromaheilkunde und Aromatologie |
| ES2507915B1 (es) * | 2013-04-15 | 2015-06-30 | Can Rich De Buscastell, S.L. | Licor de hierbas al café y su procedimiento de elaboración |
-
2016
- 2016-04-05 HK HK16103839.1A patent/HK1221598A2/zh not_active IP Right Cessation
-
2017
- 2017-04-05 CN CN201710217749.5A patent/CN107320552B/zh active Active
- 2017-04-05 WO PCT/CN2017/079501 patent/WO2017173993A1/zh not_active Ceased
- 2017-04-05 EP EP17778657.1A patent/EP3441075B1/en active Active
- 2017-04-05 TW TW106111615A patent/TWI733792B/zh active
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1475204A (zh) * | 2002-06-30 | 2004-02-18 | 丛繁滋 | 经口、鼻给药的组合物、干粉吸入剂、喷雾剂、喷雾剂辅助制剂及其制备方法 |
| CN101420848A (zh) * | 2006-02-15 | 2009-04-29 | 博塔诺凯普有限公司 | 微胶囊化精油的应用 |
| CN102481324A (zh) * | 2009-05-29 | 2012-05-30 | 新纪元公司 | 调节脂质成分的组合物和方法 |
| CN102058892A (zh) * | 2009-11-13 | 2011-05-18 | 玛丽安娜·布朗德伦德 | 以挥发油混合物为基质的物质混合物及其用途 |
| DE102011078432A1 (de) * | 2011-06-30 | 2013-01-03 | Barbara Engels-Wehr | Verwendung von Monoterpen-Estern zur Behandlung von hyperkinetischen Störungen |
| ES2507915B1 (es) * | 2013-04-15 | 2015-06-30 | Can Rich De Buscastell, S.L. | Licor de hierbas al café y su procedimiento de elaboración |
| CN103316108A (zh) * | 2013-05-20 | 2013-09-25 | 钟小强 | 治疗肝炎的复方精油 |
| WO2015014497A1 (en) * | 2013-08-02 | 2015-02-05 | Schweitzer-Mauduit International, Inc. | Edible product comprising reconstituted plant material |
| CN103463549A (zh) * | 2013-09-10 | 2013-12-25 | 季正红 | 一种保护男性心脏的复方精油 |
| EP2878303A1 (de) * | 2013-11-29 | 2015-06-03 | Doris Hefti | Zusammensetzung für die Aromatherapie, Aromaheilkunde und Aromatologie |
Non-Patent Citations (5)
| Title |
|---|
| Dietary Juniper Berry Oil Minimizes Hepatic Reperfusion Injury in the Rat;SHANNON M. JONES等;《HEPATOLOGY》;19981031;第28卷(第4期);第1042-1050页,尤其是第1042页第1段、第1049页右栏第2段 * |
| ESSENTIAL OILS FOR DETOXING THE BODY;Beyond the bite life;《http://www.beyondthebite4life.com/2016/02/essential-oils-for-detoxing-the-body.html》;20160222;第1-5页,尤其是第3页倒数第1行、第4页第1-4行 * |
| Evaluation of hepatoprotective activity of Bergamot orange in rats;Mehmet Karaca等;《Eastern Journal of Medicine》;20050131;第10卷;第1-4页,尤其是第1页摘要 * |
| Hepatitis C Treatment and Cure;Mohel(Hemet,Ca);《https://www.earthclinic.com/cures/hep_c.html?toc=Oregano+Oil&ratingvalue=5&sortby=latest》;20150106;第1页第1-10行 * |
| 迷迭香口服精油;百科用户;《https://baike.sogou.com/historylemma?lId=68766585&cId=102727279》;20150807;第1页第3、14-15行 * |
Also Published As
| Publication number | Publication date |
|---|---|
| HK1221598A2 (zh) | 2017-06-02 |
| EP3441075A4 (en) | 2020-01-01 |
| TWI733792B (zh) | 2021-07-21 |
| HK1246639A1 (zh) | 2018-09-14 |
| CN107320552A (zh) | 2017-11-07 |
| EP3441075B1 (en) | 2021-09-01 |
| TW201834679A (zh) | 2018-10-01 |
| WO2017173993A1 (zh) | 2017-10-12 |
| EP3441075A1 (en) | 2019-02-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Behl et al. | Expatiating the pharmacological and nanotechnological aspects of the alkaloidal drug berberine: current and future trends | |
| Memarzia et al. | Experimental and clinical reports on anti‐inflammatory, antioxidant, and immunomodulatory effects of Curcuma longa and curcumin, an updated and comprehensive review | |
| Ramanunny et al. | Treatment strategies against psoriasis: principle, perspectives and practices | |
| Mohanty et al. | Naringin as sustained delivery nanoparticles ameliorates the anti-inflammatory activity in a Freund’s complete adjuvant-induced arthritis model | |
| D’Souza et al. | Nanostructured lipid carriers (NLCs) for drug delivery: Role of liquid lipid (oil) | |
| US9492402B2 (en) | Formulation of curcuminoids with enhanced bioavailability of curcumin, demethoxycurcumin, bisdemethoxycurcumin and method of preparation and uses thereof | |
| WO2009012718A1 (en) | A composite emulsifier, an emulsion prepared from it and the preparation method thereof | |
| CN104188905B (zh) | 一种稳定的氟比洛芬酯微纳米乳液及其制备方法 | |
| JP2019528297A (ja) | 体重減少に用いられる皮下注射製剤およびその用途 | |
| AU2011282200B2 (en) | Anti-viral properties of Aloe vera and Acquired Immune Deficiency Syndrome (AIDS) treatment | |
| CN102405046A (zh) | 表现出抗炎性质的药物组合物 | |
| CN104187627A (zh) | 一种降血脂组合物及其在制备具有降血脂功能的保健食品中的应用 | |
| Maio et al. | The significant role of nutraceutical compounds in ulcerative colitis treatment | |
| Luo et al. | Construction of phlorotannin-based nanoparticles for alleviating acute liver injury | |
| CN107320552B (zh) | 口服药物组合物和其用于制备治疗或预防疾病的药物的用途 | |
| ZANGENEH et al. | Evaluation of the anti-anemic potential of Glycyrrhiza glabra aqueous extract in Phenylhydrazine-treated rats | |
| CN102511821A (zh) | 番茄红素葡萄籽提取物复合降脂软胶囊制剂及其生产方法 | |
| Trushna et al. | Nutraceuticals with anti-inflammatory and anti-oxidant properties as an intervention for reducing the health effects of fine particulate matter: potential and prospects | |
| CN110151815A (zh) | 含光皮树提取物的制剂及其应用 | |
| KR102476373B1 (ko) | 커큐민과 오메가 3를 포함한 항염증용 조성물 및 그 제조방법 | |
| US20170042833A1 (en) | Formulation of Curcuminoids with Enhanced Bioavailability of Curcumin, Demethoxycurcumin, Bisdemethoxycurcumin and method of preparation and uses thereof | |
| CN102949415B (zh) | 一种蜂胶脂肪乳制剂及其制备方法 | |
| WO2017021974A2 (en) | Novel and synergistic composition of lecithin and lysolecithin for improving bioavailability and solubility of hydrophobic compounds and extracts | |
| HK1246639B (zh) | 口服药物组合物和其用於制备治疗或预防疾病的药物的用途 | |
| CN105267187A (zh) | 治疗脂肪肝的医药组合物及其制备方法与用途 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1246639 Country of ref document: HK |
|
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CP02 | Change in the address of a patent holder |
Address after: Room 17-18, 18/F, Star House, 3 Salisbury Road, Tsim Sha Tsui, Kowloon, Hong Kong, China Patentee after: Lai Dongni Address before: 93-107 Lockhart Road, Wan Chai, Hongkong, China Patentee before: Lai Dongni |
|
| CP02 | Change in the address of a patent holder | ||
| CB03 | Change of inventor or designer information | ||
| CB03 | Change of inventor or designer information |
Inventor after: Lai Dongqing Inventor before: Lai Dongni |
|
| CP01 | Change in the name or title of a patent holder |
Address after: Room 17-18, 18/F, Star House, 3 Salisbury Road, Tsim Sha Tsui, Kowloon, Hong Kong, China Patentee after: Lai Dongqing Address before: Room 17-18, 18/F, Star House, 3 Salisbury Road, Tsim Sha Tsui, Kowloon, Hong Kong, China Patentee before: Lai Dongni |
|
| CP01 | Change in the name or title of a patent holder |