WO2018220929A1 - 植物細胞でのタンパク質発現システム及びその使用 - Google Patents
植物細胞でのタンパク質発現システム及びその使用 Download PDFInfo
- Publication number
- WO2018220929A1 WO2018220929A1 PCT/JP2018/008512 JP2018008512W WO2018220929A1 WO 2018220929 A1 WO2018220929 A1 WO 2018220929A1 JP 2018008512 W JP2018008512 W JP 2018008512W WO 2018220929 A1 WO2018220929 A1 WO 2018220929A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- nucleic acid
- acid fragment
- egfp
- vector
- expression
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/82—Vectors or expression systems specially adapted for eukaryotic hosts for plant cells, e.g. plant artificial chromosomes (PACs)
- C12N15/8201—Methods for introducing genetic material into plant cells, e.g. DNA, RNA, stable or transient incorporation, tissue culture methods adapted for transformation
- C12N15/8202—Methods for introducing genetic material into plant cells, e.g. DNA, RNA, stable or transient incorporation, tissue culture methods adapted for transformation by biological means, e.g. cell mediated or natural vector
- C12N15/8205—Agrobacterium mediated transformation
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/82—Vectors or expression systems specially adapted for eukaryotic hosts for plant cells, e.g. plant artificial chromosomes (PACs)
- C12N15/8201—Methods for introducing genetic material into plant cells, e.g. DNA, RNA, stable or transient incorporation, tissue culture methods adapted for transformation
- C12N15/8202—Methods for introducing genetic material into plant cells, e.g. DNA, RNA, stable or transient incorporation, tissue culture methods adapted for transformation by biological means, e.g. cell mediated or natural vector
- C12N15/8203—Virus mediated transformation
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/82—Vectors or expression systems specially adapted for eukaryotic hosts for plant cells, e.g. plant artificial chromosomes (PACs)
- C12N15/8241—Phenotypically and genetically modified plants via recombinant DNA technology
- C12N15/8242—Phenotypically and genetically modified plants via recombinant DNA technology with non-agronomic quality (output) traits, e.g. for industrial processing; Value added, non-agronomic traits
- C12N15/8243—Phenotypically and genetically modified plants via recombinant DNA technology with non-agronomic quality (output) traits, e.g. for industrial processing; Value added, non-agronomic traits involving biosynthetic or metabolic pathways, i.e. metabolic engineering, e.g. nicotine, caffeine
- C12N15/8251—Amino acid content, e.g. synthetic storage proteins, altering amino acid biosynthesis
Definitions
- the present invention relates to an expression system and use thereof. More specifically, the present invention relates to an expression system, a method for producing a target protein, and an expression vector.
- This application claims priority based on Japanese Patent Application No. 2017-107965 filed in Japan on May 31, 2017, the contents of which are incorporated herein by reference.
- Transgenic plants may be used for the purpose of producing recombinant proteins and analyzing protein localization in plants. However, it takes a long time to produce a transgenic plant. Moreover, the expression level of the protein by the transgenic plant tends to be relatively low.
- a transient expression system using a virus-based vector may be able to obtain high expression of the recombinant protein in a short time.
- an expression system called the magnICON system is a viral system based on tobacco mosaic virus, which was developed to achieve a high level of accumulation of recombinant proteins in tobacco leaves (eg, non-patent literature). 1).
- geminivirus has a single-stranded circular DNA genome and replicates the genome with a very high copy number by a rolling circle type DNA replication mechanism. This mechanism has been used to increase the expression of a protein in a transgenic plant or increase the expression level of a recombinant protein in a transient expression system (see, for example, Non-Patent Document 2).
- Marillonnet S., et al. Systemic Agrobacterium tumefaciens-mediated transfection of viral replicons for efficient transient expression in plants., Nat. Biotechnol., 23 (6), 718-723, 2005.
- Moon K. B., et al. Overexpression and self-assembly of virus-like particles in Nicotiana benthamiana by a single-vector DNA replicon system., Appl. Microbiol. Biotechnol., 98 (19), 8281-8290, 2014 .
- Non-Patent Document 1 is limited to plants belonging to the genus Tobacco, and may be difficult to apply to other plants.
- the system described in Non-Patent Document 2 may not have a sufficient protein expression level. Therefore, the present invention can be applied to plants other than the genus Tobacco, and an object thereof is to provide an expression system having a high protein expression level.
- a first comprising a Long Intelligent Region (LIR) derived from a geminivirus, a Small Intelligent Region (SIR) derived from a geminivirus, and an expression cassette of a target protein linked between the LIR and the SIR.
- LIR Long Intelligent Region
- SIR Small Intelligent Region
- An expression system comprising terminators linked in this order.
- the gene silencing inhibitor is gene silencing inhibitor P19 derived from tomato bushy stunt virus.
- the expression system according to [4] or [5], wherein the first nucleic acid fragment, the second nucleic acid fragment, and the third nucleic acid fragment are contained in a single vector.
- a T-DNA right border sequence (RB) and a T-DNA left border sequence (LB) are further included, and the first nucleic acid fragment, the second nucleic acid fragment, and the third nucleic acid fragment are the RB And the LB, the expression system according to [6].
- a method for producing a target protein comprising the step of introducing the expression system according to any one of [1] to [8] into a plant cell.
- a first nucleic acid fragment comprising a geminivirus-derived LIR, a geminivirus-derived SIR, and an expression cassette linked between the LIR and the SIR, wherein the expression cassette comprises a promoter, An expression vector comprising a multicloning site and two or more terminators linked in this order.
- a T-DNA right border sequence (RB) and a T-DNA left border sequence (LB) are further provided, and the first nucleic acid fragment, the second nucleic acid fragment, and the third nucleic acid fragment are the RB
- an expression system that can be applied to plants other than the genus Tobacco and has a high protein expression level can be provided.
- A It is a photograph which shows the state which put about 1.2L Agrobacterium suspension in a 2L glass beaker, and installed in the vacuum desiccator in performing Agroinfiltration.
- B A photograph showing a state in which lettuce is immersed in an Agrobacterium suspension and set to a pressure of 736 mmHg.
- C It is the photograph which shows the state which put the lettuce after agroinfiltration in the bowl.
- D It is a photograph which shows the state which is incubating the lettuce after agroinfiltration.
- A is a schematic diagram of the T-DNA region of the pBYR2fp-EGFP vector.
- (B) is a schematic diagram of the T-DNA region of the pBYR2HS-EGFP vector.
- C is a schematic diagram of the T-DNA region of the pBYR2EE-EGFP vector.
- (D) is a schematic diagram of the T-DNA region of the pBYR2HH-EGFP vector.
- E is a schematic diagram of the T-DNA region of the pBYR2H-EGFP vector.
- (F) is a schematic diagram of the T-DNA region of the pBYR2TN-EGFP vector.
- G is a schematic diagram of the T-DNA region of the pBYR2T-EGFP vector.
- (H) is a schematic diagram of the T-DNA region of the pBYR2HT-EGFP vector.
- (I) is a schematic diagram of the T-DNA region of the pBYR2HTS-EGFP vector.
- (A) to (i) are photographs showing the results of observation of fluorescence of expressed EGFP in Experimental Example 1.
- (A) is a photograph showing the result of subjecting all soluble proteins prepared from the leaves of Bensamiana tobacco in Experimental Example 2 to SDS-PAGE and staining with CBB.
- (B) is a photograph showing the result of transferring the gel of (a) onto a PVDF membrane and performing immunoblot analysis using an anti-GFP antibody.
- (C) is a graph in which the expression level of EGFP in (a) is quantified.
- (A) is a photograph showing the result of subjecting all soluble proteins prepared from lettuce leaves to SDS-PAGE and CBB staining in Experimental Example 2.
- (B) is a photograph showing the result of transferring the gel of (a) onto a PVDF membrane and performing immunoblot analysis using an anti-GFP antibody.
- (C) is a graph in which the expression level of EGFP in (a) is quantified.
- (A) is a photograph showing the result of subjecting all soluble proteins prepared from eggplant leaves to SDS-PAGE and CBB staining in Experimental Example 2.
- (B) is a photograph showing the result of transferring the gel of (a) onto a PVDF membrane and performing immunoblot analysis using an anti-GFP antibody.
- (C) is a graph in which the expression level of EGFP in (a) is quantified.
- (A) is a photograph showing the result of immunoblotting analysis using anti-GFP antibody after transferring all soluble proteins prepared from tomato leaves in Experimental Example 2 to a PVDF membrane after subjecting them to SDS-PAGE.
- (B) is a photograph showing the result of subjecting all soluble proteins prepared from capsicum leaves and rose petals to SDS-PAGE, transferring them to a PVDF membrane, and immunoblotting analysis using an anti-GFP antibody.
- (A) to (e) are photographs showing the results of observation of fluorescence of expressed EGFP in Experimental Example 3.
- (A) is a photograph showing the result of subjecting all soluble proteins prepared from the leaves of Bensamiana tobacco in Experimental Example 4 to SDS-PAGE and staining with CBB.
- (B) is a graph in which the expression level of EGFP in (a) is quantified.
- (A) is a photograph showing the result of subjecting total soluble protein prepared from the leaf of Bensamiana tobacco in Example 5 to SDS-PAGE and staining with CBB.
- (B) is a graph in which the expression level of EGFP in (a) is quantified. It is a schematic diagram which shows the structure of GFP_pICH18711 vector.
- (A) And (b) is a photograph which shows the result of having observed the fluorescence of expressed EGFP or GFP in Experimental example 6.
- FIG. (A) is a photograph showing the result of subjecting all soluble proteins prepared from the leaves of Bensamiana tobacco in Experimental Example 6 to SDS-PAGE and staining with CBB.
- (B) is a graph obtained by quantifying the expression level of EGFP or GFP in (a).
- the present invention provides a first nucleic acid fragment comprising a geminivirus-derived LIR, a geminivirus-derived SIR, and an expression cassette for a target protein linked between the LIR and the SIR; A second nucleic acid fragment containing a Geminivirus-derived Rep / RepA protein expression cassette, wherein the expression cassette for the target protein is linked to a promoter and two or more nucleic acid fragments encoding the target protein.
- An expression system comprising a terminator in this order is provided.
- Rep / RepA protein which is a replication initiation protein of geminivirus
- Rep / RepA protein which is a replication initiation protein of geminivirus
- the expression cassette of the target protein linked between the LIR and SIR on the first nucleic acid fragment is replicated with a high copy number by the rolling circle type DNA replication mechanism of Geminivirus.
- the target protein is expressed at a high expression level from the expression cassette of the target protein replicated at a high copy number.
- the expression system of this embodiment can achieve a very high expression level of the target protein by including two or more terminators linked to the expression cassette of the target protein. it can.
- the terminator is a base sequence that terminates transcription of DNA into mRNA. Although it does not specifically limit as a terminator, for example, the terminator derived from Arabidopsis heat shock protein 18.2 gene, the terminator of tobacco extensin gene, the 35S terminator of a cauliflower mosaic virus (CaMV), the NOS terminator of CaMV, etc. are mentioned. In the expression system of the present embodiment, two or more terminators linked to each other may be terminators having the same base sequence, or may be terminators having different base sequences.
- SEQ ID NO: 19 shows the base sequence of the terminator derived from the Arabidopsis heat shock protein 18.2 gene.
- SEQ ID NO: 20 shows the base sequence of the terminator of the tobacco extensin gene.
- SEQ ID NO: 21 shows the base sequence of the CaMV 35S terminator.
- SEQ ID NO: 22 shows the base sequence of the CaMV NOS terminator.
- the base sequences of these terminators may have mutations relative to the base sequences of SEQ ID NOs: 19 to 22 as long as they have a function of terminating transcription of DNA into mRNA. A part of the base sequence may be deleted.
- each base sequence is, for example, 70 with respect to the base sequences of SEQ ID NOs: 19 to 22, respectively.
- % Sequence identity preferably 80% or more sequence identity, more preferably 90% or more sequence identity, and 95% or more sequence identity. Is particularly preferred.
- the expression cassette of the target protein preferably includes two terminators.
- the expression level of the target protein tends to be higher when the expression cassette of the target protein includes two or more terminators than when the expression cassette of the target protein includes three or more terminators.
- At least one of the terminators included in the expression cassette of the target protein is a terminator derived from the Arabidopsis heat shock protein 18.2 gene.
- an expression system including a terminator derived from the Arabidopsis heat shock protein 18.2 gene as a terminator tends to have a high expression level of the target protein.
- the expression system of this embodiment can express about 3 mg or more of the target protein per 1 g of fresh weight of the plant body. This expression level is equivalent to the MAGNICICON system currently used commercially.
- magnICON system is limited to plants belonging to the genus Tobacco
- expression system of the present embodiment is applicable not only to plants belonging to the genus Tobacco but also to plants other than the genus Tobacco. Can also achieve a high expression level of the target protein.
- plants other than the genus Tobacco include solanaceous plants such as tomato, eggplant, and pepper, asteraceae plants such as lettuce, cucurbitaceae plants such as melon, orchidaceae plants such as moth orchids, and the like. It is not limited to.
- the first nucleic acid fragment includes a promoter, a nucleic acid fragment encoding the target protein, and two or more terminators linked in this order.
- any promoter can be used as long as it exhibits the transcriptional activity of DNA linked downstream in plant cells of the host.
- cauliflower mosaic virus (CaMV) 35S promoter ubiquitin promoter
- cassava bean mosaic virus (CsVMV) promoter and the like can be mentioned.
- the target protein is not particularly limited, and any protein can be expressed. According to the expression system of the present embodiment, the protein can be expressed at a lower cost than protein expression using animal cells. Moreover, the expression system of this embodiment can be used for the expression of pollen allergens and the like that are difficult to express in an expression system such as Escherichia coli. For this reason, the target protein may be, for example, a pollen allergen.
- the terminator consists of a base sequence involved in specific termination of RNA transcription by RNA polymerase.
- the terminator two or more of one kind of terminator may be connected and used, or two or more kinds of terminators may be used in combination.
- the expression system of this embodiment can achieve a high expression level of the target protein by having two or more terminators linked together.
- the expression level of the target protein tends to be further increased.
- the first nucleic acid fragment may have, for example, a 5'-untranslated region (UTR), a polyadenylation signal, and the like in addition to the promoter, the nucleic acid fragment encoding the target protein, and the terminator.
- UTR 5'-untranslated region
- polyadenylation signal and the like in addition to the promoter, the nucleic acid fragment encoding the target protein, and the terminator.
- 5′-UTR By having 5'-UTR, the expression efficiency of the target protein may further increase.
- 5′-UTR include 5′-UTR of tobacco mosaic virus, 5′-UTR of Arabidopsis alcohol dehydrogenase gene, 5′-UTR of Arabidopsis elongation factor 1 ⁇ -A3 gene, 5′-UTR of rice alcohol dehydrogenase gene, etc. Is mentioned.
- the second nucleic acid fragment contains an expression cassette for Rep / RepA protein derived from geminivirus.
- the Rep / RepA protein expression cassette is not particularly limited as long as the Rep / RepA protein can be expressed in the host plant cell, and includes a promoter, a nucleic acid fragment encoding the Rep / RepA protein, a terminator, 5′-UTR, and polyadenyl. It may have an activating signal or the like.
- a promoter of the Rep / RepA protein in addition to those described above as those that can be used for the first nucleic acid fragment, for example, LIR derived from Geminivirus may also be used because it has promoter activity.
- the “expression system” means a system capable of expressing a target protein by introducing a combination of a first nucleic acid fragment and a second nucleic acid fragment into a plant cell.
- the expression system may be composed of one nucleic acid fragment or a combination of two or more nucleic acid fragments as long as the effect of the present invention is obtained.
- the nucleic acid fragment may be a vector.
- the first nucleic acid fragment and the second nucleic acid fragment may exist separately as independent nucleic acid fragments, or are linked to form one nucleic acid fragment. It may be.
- first nucleic acid fragment and the second nucleic acid fragment are linked, the order of the linkage is not particularly limited, and the first nucleic acid fragment may exist on the 5 ′ side, or the 5 ′ A second nucleic acid fragment may be present on the side.
- LIR, SIR, and Rep / RepA are derived from Geminivirus.
- the geminivirus is not particularly limited as long as it has a rolling circle type DNA replication mechanism.
- geminaviidae mastrevirus genus macular atrophy virus BeYDV
- tomato golden mosaic virus TGMV
- African cassava mosaic A virus ACMV
- RLCV rose cigar virus
- SEQ ID NO: 23 shows the base sequence of LIR derived from Geminivirus.
- SEQ ID NO: 24 shows the base sequence of SIR derived from geminivirus.
- SEQ ID NO: 25 shows the base sequence of Geminivirus-derived Rep / RepA protein (open reading frames C1 and C2 encoding Rep / RepA protein which is a replication initiator protein of BeYDV).
- the LIR base sequence, the SIR base sequence, and the base sequence encoding Rep / RepA protein are expressed by the Rep / RepA protein encoded by the base sequence so that the expression cassette of the target protein linked between LIR and SIR is obtained.
- it may have mutations in the base sequences of SEQ ID NOs: 23, 24, and 25, respectively. It may be deleted.
- each base sequence is
- each base sequence is
- Each of the nucleotide sequences of SEQ ID NOs: 23, 24, and 25 preferably has, for example, 70% or more sequence identity, more preferably 80% or more sequence identity, and more than 90% sequence. It is more preferable to have identity, and it is particularly preferable to have 95% or more sequence identity.
- sequence identity of the target base sequence with respect to the reference base sequence can be determined, for example, according to the above-described formula (1).
- the expression system of the present embodiment may further include a third nucleic acid fragment containing an expression cassette for a gene silencing inhibitor.
- a gene silencing inhibitor include gene silencing inhibitor P19 derived from tomato bushy stunt virus, gene silencing inhibitor 16K derived from tobacco rattle virus, and the like.
- the third nucleic acid fragment may exist separately as a nucleic acid fragment independent of the first nucleic acid fragment and the second nucleic acid fragment described above, or may be arbitrarily combined with the first nucleic acid fragment or the second nucleic acid fragment.
- the first nucleic acid fragment, the second nucleic acid fragment and the third nucleic acid fragment may be linked in any order. That is, the first nucleic acid fragment, the second nucleic acid fragment, and the third nucleic acid fragment may be contained in a single vector.
- the first nucleic acid fragment, the second nucleic acid fragment, and the third nucleic acid fragment are contained in a single vector, and the vector further comprises T-DNA RB and LB.
- the first nucleic acid fragment, the second nucleic acid fragment, and the third nucleic acid fragment may exist between RB and LB of T-DNA.
- T-DNA is a specific region possessed by a Ti plasmid or Ri plasmid found in a pathogenic strain of Agrobacterium, a pathogenic bacterium of crown gall, which is a dicotyledonous tumor.
- Agrobacterium having T-DNA coexists with a plant cell, a nucleic acid fragment existing between RB and LB is transferred into the host plant cell.
- the first nucleic acid fragment, the second nucleic acid fragment and the third nucleic acid fragment can be easily introduced into the host plant cell.
- the vector in which the first nucleic acid fragment, the second nucleic acid fragment, and the third nucleic acid fragment are present between RB and LB is preferably a vector that can be used in the binary vector method.
- the binary vector method is a gene introduction method into a plant using a vir helper Ti plasmid from which the original T-DNA of the Ti plasmid has been removed and a small shuttle vector having an artificial T-DNA.
- the shuttle vector is preferably one that can be maintained by both E. coli and Agrobacterium.
- the vir helper Ti plasmid does not have the original T-DNA, it cannot form a crown gall on the plant. However, the vir helper Ti plasmid has the vir region necessary for introducing T-DNA into the host plant cell.
- T-DNA having a desired nucleic acid fragment into Agrobacterium having a vir helper Ti plasmid and introducing the Agrobacterium into the host plant, the desired nucleic acid fragment can be easily transferred to the host plant cell. Can be introduced in.
- a vector in which the first nucleic acid fragment, the second nucleic acid fragment and the third nucleic acid fragment are present between RB and LB has an origin of replication for E. coli and an origin of replication for Agrobacterium. It may be a shuttle vector that can be maintained by both E. coli and Agrobacterium.
- SEQ ID NO: 26 shows the RB base sequence of T-DNA.
- SEQ ID NO: 27 shows the LB base sequence of T-DNA.
- the base sequences of RB and LB have mutations with respect to the base sequences of SEQ ID NOs: 26 and 27, respectively, as long as they have a function of transferring a nucleic acid fragment existing between RB and LB into a host plant cell.
- a part of the base sequences of SEQ ID NOs: 26 and 27 may be deleted.
- the base sequences of RB and LB have mutations and deletions with respect to the base sequences of SEQ ID NOs: 26 and 27, respectively, the base sequences are respectively compared with the base sequences of SEQ ID NOs: 26 and 27, respectively.
- it preferably has 70% or more sequence identity, more preferably 80% or more sequence identity, more preferably 90% or more sequence identity, and 95% or more sequence identity. It is particularly preferable to have it.
- sequence identity of the target base sequence with respect to the reference base sequence can be determined, for example, according to the above-described formula (1).
- the present invention comprises a first nucleic acid fragment comprising a geminivirus-derived LIR, a geminivirus-derived SIR, and an expression cassette linked between the LIR and the SIR.
- An expression vector is provided in which a cassette comprises a promoter, a multicloning site, and two or more terminators linked in this order.
- the expression vector of this embodiment can be suitably used for the production of the expression system described above. As will be described later in Examples, the expression vector of this embodiment can achieve a very high expression level of the target protein by introducing a gene fragment encoding the target protein into the multicloning site of the expression cassette. . This is due to the fact that two or more expression cassettes are connected to each other.
- the expression cassette preferably contains two terminators.
- the expression level of the target protein tends to be higher when the expression cassette contains two or more terminators than when the expression cassette contains three or more terminators.
- At least one of the terminators included in the expression cassette is a terminator derived from the Arabidopsis heat shock protein 18.2 gene.
- an expression vector containing a terminator derived from the Arabidopsis heat shock protein 18.2 gene as a terminator tends to have a high expression level of the target protein.
- LIR, SIR, promoter, and terminator are the same as those described above. That is, in the expression vector of the present embodiment, the geminivirus may be kidney macular atrophy virus (BeYDV).
- BeYDV kidney macular atrophy virus
- the multi-cloning site is a region in which one or more base sequences recognized by restriction enzymes are arranged. That is, in the multicloning site of the expression vector of the present embodiment, there may be one restriction enzyme site or a plurality of restriction enzyme sites. Since the vector of this embodiment has a multiple cloning site, a nucleic acid fragment encoding the target protein can be easily cloned.
- the expression vector of this embodiment may not contain a nucleic acid fragment encoding the target protein, or a nucleic acid fragment encoding the target protein may be introduced. That is, a vector into which a nucleic acid fragment encoding the target protein has been introduced is also included in the expression vector of this embodiment.
- the vector of the present embodiment may further include a second nucleic acid fragment containing an expression cassette of Rep / RepA protein derived from geminivirus.
- the Rep / RepA protein is the same as described above.
- the vector of the present embodiment may further include a third nucleic acid fragment containing an expression cassette for a gene silencing inhibitor.
- the gene silencing inhibitor is the same as described above, and may be, for example, the gene silencing inhibitor P19 derived from tomato bushy stunt virus.
- the vector of this embodiment further comprises RB and LB of T-DNA, and the first nucleic acid fragment, the second nucleic acid fragment and the third nucleic acid fragment are located between RB and LB of T-DNA. May be present.
- RB and LB are the same as those described above.
- the vector of the present embodiment is preferably a vector that can be used in the binary vector method.
- the present invention provides a method for producing a target protein, the method comprising the step of introducing the expression system described above into a plant cell.
- the expression system may be one in which a gene fragment encoding the target protein is introduced into the multicloning site of the vector described above.
- the production method of the present embodiment can produce a target protein with a high expression level using tobacco plants and plants other than tobacco as hosts.
- Agrobacterium tumefaciens strain GV3101 having a binary vector was added to a YEB medium (6 g / L yeast extract, 5 g / L tryptone) supplemented with antibiotics (100 mg / L kanamycin, 30 mg / L gentamicin, 30 mg / L rifampicin). 5 g / L sucrose, 2 mM magnesium sulfate) and cultured at 28 ° C. for 2 days.
- the culture cultured for 2 days was diluted 100-fold with the same medium as above, MES was added to a final concentration of 10 mM to adjust the pH to 5.6, and acetosyringone was further adjusted to a final concentration of 20 ⁇ M. And then scaled up by culturing at 28 ° C. for 18-24 hours using a rotary shaker set at 140 rpm.
- sucrose is added to a final concentration of 55 g / L
- acetosyringone is further added to a final concentration of 200 ⁇ M, and incubated at 22 ° C. for 1 hour. did.
- red leaf lettuce was washed with distilled water, and water was removed with a paper towel. Subsequently, the base of the washed lettuce was placed on a wet paper towel. Subsequently, the lettuce was covered with a plastic wrap and allowed to stand at 24 degrees for one day. Subsequently, the lettuce was irradiated with blue LED light for 30 minutes or more before vacuum infiltration.
- FIG. 1 (a) about 1.2 L of Agrobacterium suspension was placed in a 2 L glass beaker and placed in a vacuum desiccator. Then, as shown in FIG.1 (b), lettuce was immersed in the Agrobacterium suspension, set to the pressure of 736 mmHg, and left still for 20 minutes. Subsequently, the pressure was returned to atmospheric pressure and the lettuce was washed with water.
- Agrobacterium tumefaciens strain GV3101 having a binary vector was added to L-broth medium supplemented with 10 mM MES (pH 5.6), 20 ⁇ M acetosyringone, 100 mg / L kanamycin, 30 mg / L gentamicin, and 30 mg / L rifampicin.
- the cells were cultured at 28 ° C. until the stationary phase.
- the suspension of Agrobacterium tumefaciens was infiltrated on the back side of a 4-week-old benthamiana tobacco leaf using a 1 mL syringe without a needle.
- N. benthamiana tobacco was immersed in an Agrobacterium suspension, allowed to stand for 20 minutes under a pressure of 736 mmHg, and then infiltrated by returning the pressure to atmospheric pressure.
- the same suspension was also infiltrated into tomato fruits using a 1 mL syringe with a needle.
- FIG. 2 (a) is a schematic diagram of the T-DNA region of the pBYR2fp-EGFP vector (provided by Dr. Mason, Arizona State University, USA).
- the pBYR2fp vector which is a known vector, has a replication system derived from bean macular dwarf virus (BeYDV).
- the pBYR2fp vector has an expression cassette of a gene silencing inhibitory factor P19 derived from tomato bushy stunt virus.
- an enhanced green fluorescence protein (EGFP) gene fragment was PCR amplified using a primer (pBYR2fp-EGFP-F, SEQ ID NO: 1) and a primer (EGFP-pBYR2fp-R, SEQ ID NO: 2). Subsequently, the obtained PCR product was cloned into a pBYR2fp vector cleaved with a restriction enzyme XbaI to prepare a pBYR2fp-EGFP vector.
- FIG. 2 (b) is a schematic diagram of the T-DNA region of the pBYR2HS-EGFP vector.
- An EGFP gene fragment having a 5′-UTR of the alcohol dehydrogenase gene and a terminator of the Arabidopsis heat shock protein 18.2 gene was introduced into the pBYR2fp vector.
- the EGFP gene fragment was PCR amplified using a primer (pRI201-EGFP-F, SEQ ID NO: 3) and a primer (EGFP-pRI201-R, SEQ ID NO: 4).
- PCR product was cloned into pRI201-AN (Takara Bio Inc.) cut with restriction enzymes NdeI and SalI to prepare a pRI201-EGFP vector.
- the primer pBYR2fp-AtADH-F, SEQ ID NO: 5
- the primer pBYR2fp-HSPter-R, SEQ ID NO: 6
- the alcohol dehydrogenase gene 5′-UTR and Arabidopsis thaliana were used.
- the EGFP gene fragment having the heat shock protein 18.2 gene terminator was PCR amplified.
- SEQ ID NO: 28 shows the full-length base sequence of the pBYR2HS-EGFP vector.
- FIG. 2 (c) is a schematic diagram of the T-DNA region of the pBYR2EE-EGFP vector.
- a tobacco extensin gene terminator was PCR amplified using a primer (pBYR2EE-Ext3-F, SEQ ID NO: 7) and a primer (pBYR2EE-Ext3-R, SEQ ID NO: 8).
- the obtained PCR product was cloned into a pBYR2HS-EGFP vector cleaved with restriction enzymes SalI and XbaI to prepare a pBYR2EE-EGFP vector.
- the terminator of the pBYR2EE-EGFP vector was a combination of two tobacco extensin gene terminators.
- FIG. 2 (d) is a schematic diagram of the T-DNA region of the pBYR2HH-EGFP vector.
- PCR amplification was performed using the pBYR2HS-EGFP vector as a template, using the terminator of the Arabidopsis heat shock protein 18.2 gene as a primer (pBYR2H-HSPter-F, SEQ ID NO: 9) and pBYR2H-HSPter-R (SEQ ID NO: 10).
- the PCR product was cloned into pBYR2H-EGFP cut with a restriction enzyme XbaI to prepare a pBYR2HH-EGFP vector.
- the terminator of the pBYR2HH-EGFP vector was a combination of two terminators of the Arabidopsis heat shock protein 18.2 gene.
- FIG. 2 (e) is a schematic diagram of the T-DNA region of the pBYR2H-EGFP vector.
- PCR amplification of SIR-C2 was performed using a primer (HSPter-SIR-F, SEQ ID NO: 11) and a primer (C1-ClaI-C2-R, SEQ ID NO: 12) using the pBYR2HS-EGFP vector as a template.
- FIG. 2 (f) is a schematic diagram of the T-DNA region of the pBYR2TN-EGFP vector.
- pCambia1391Z Marker Gene Technologies, Inc.
- PCR amplification was performed as a template.
- the NOS terminator was PCR-amplified using pRI201-AN (Takara Bio Inc.) as a template using a primer (35Ster-NOSter-F, SEQ ID NO: 15) and a primer (pBYR2TN-NOSter-R, SEQ ID NO: 16).
- the PCR product was cloned into the pBYR2H-EGFP vector cut with restriction enzymes SalI and XbaI to prepare pBYR2TN-EGFP.
- the terminator of the pBYR2TN-EGFP vector was a cauliflower mosaic virus (CaMV) 35S terminator and one NOS terminator linked together.
- CaMV cauliflower mosaic virus
- FIG. 2 (g) is a schematic diagram of the T-DNA region of the pBYR2T-EGFP vector.
- the cauliflower mosaic virus (CaMV) 35S terminator was PCR-amplified using pCambia1391Z as a template using a primer (pBYR2T-35Ster-F, SEQ ID NO: 13) and a primer (pBYR2HS-35Ster-R, SEQ ID NO: 17).
- the PCR product was cloned into a pBYR2H-EGFP vector cut with restriction enzymes SalI and XbaI to prepare a pBYR2T-EGFP vector.
- the terminator of the pBYR2T-EGFP vector had one 35S terminator of cauliflower mosaic virus (CaMV).
- FIG. 2 (h) is a schematic diagram of the T-DNA region of the pBYR2HT-EGFP vector.
- the cauliflower mosaic virus (CaMV) 35S terminator was PCR-amplified using pCambia1391Z as a template, using a primer (pBYR2HS-35Ster-F, SEQ ID NO: 18) and a primer (pBYR2HS-35Ster-R, SEQ ID NO: 17).
- the PCR product was cloned into the pBYR2H-EGFP vector cleaved with the restriction enzyme XbaI to prepare a pBYR2HT-EGFP vector.
- the terminator of the pBYR2HT-EGFP vector was an Arabidopsis heat shock protein 18.2 gene terminator and a cauliflower mosaic virus (CaMV) 35S terminator, one by one.
- FIG. 2 (i) is a schematic diagram of the T-DNA region of the pBYR2HTS-EGFP vector.
- the cauliflower mosaic virus (CaMV) 35S terminator was PCR-amplified using pCambia1391Z as a template, using a primer (pBYR2HS-35Ster-F, SEQ ID NO: 18) and a primer (pBYR2HS-35Ster-R, SEQ ID NO: 17).
- the PCR product was cloned into the pBYR2HS-EGFP vector cleaved with the restriction enzyme XbaI to prepare a pBYR2HTS-EGFP vector.
- the terminator of the pBYR2HTS-EGFP vector was an Arabidopsis heat shock protein 18.2 gene terminator, a cauliflower mosaic virus (CaMV) 35S terminator, and a tobacco extensin gene terminator.
- 35S-p ⁇ 2 means the 35S promoter of cauliflower mosaic virus (CaMV) having two enhancement elements
- TMV ⁇ is the 5′-UTR of tobacco mosaic virus.
- AtADH5 ′ means 5′-UTR of the Arabidopsis alcohol dehydrogenase gene
- EGFP means enhanced green fluorescence protein
- Ext3 ′ means the terminator of the tobacco extensin gene
- HSPter means the terminator of the Arabidopsis heat shock protein 18.2 gene
- 35Ster means the 35S terminator of cauliflower mosaic virus (CaMV)
- Nos-t means the NOS terminator.
- LIR Long Intelligent Region of the kidney bean dwarf virus (BeYDV) genome
- SIR Short Intelligent Region of the BeYDV genome
- C1 and C2 are replicas of BeYDV. It means open reading frames C1 and C2 encoding Rep / RepA protein which is the starting protein
- LB and “RB” mean the left border sequence and the right border sequence of T-DNA, respectively
- Nos-p Means the NOS promoter
- p19 means the gene encoding the gene silencing inhibitor P19 derived from tomato bushy stunt virus.
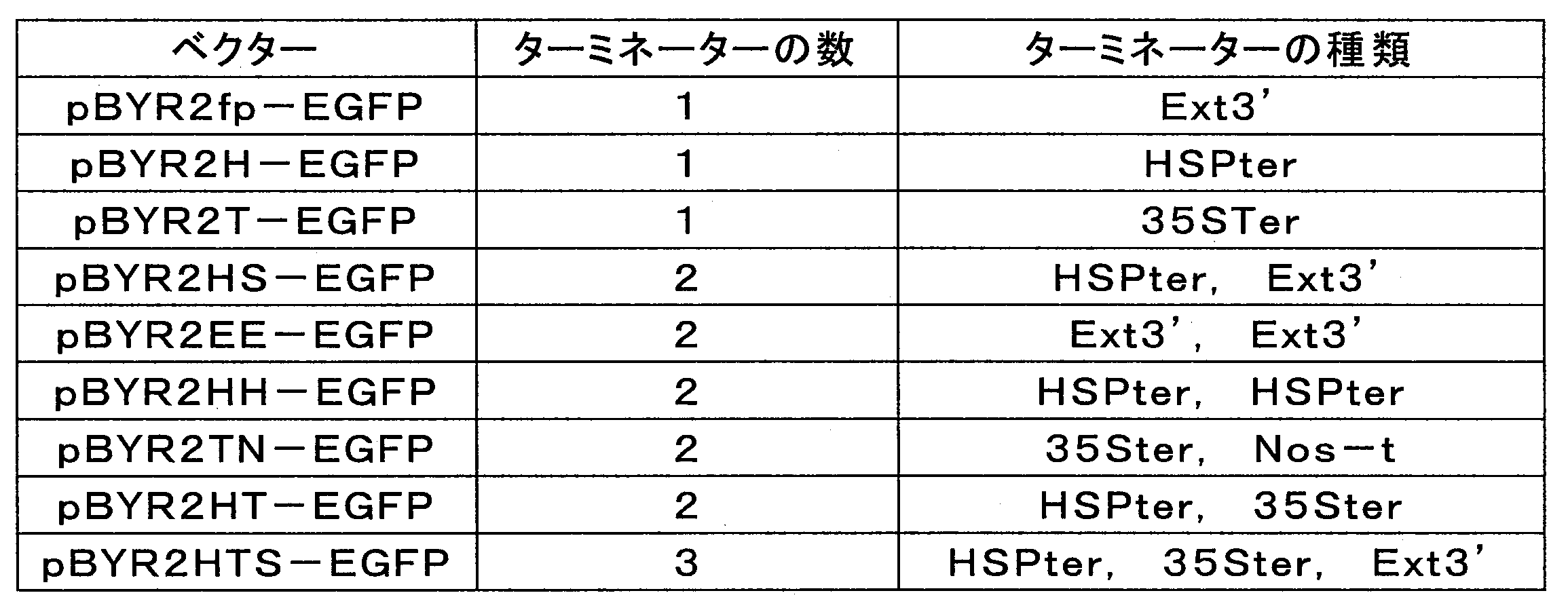
- Table 1 below shows an outline of the pBYR2HS-EGFP vector and the pBYR2fp-EGFP vector.
- HSPter means the terminator of the Arabidopsis heat shock protein 18.2 gene
- Ext3 ′ means the terminator of the tobacco extensin gene.
- pBYR2HS-EGFP vector and pBYR2fp-EGFP vector were introduced into Agrobacterium tumefaciens GV3101 strain, respectively, benthamiana tobacco leaf, lettuce leaf, eggplant leaf, tomato fruit, tomato leaf, capsicum leaf, It was introduced into melon leaves, rose petals and moth orchids, respectively.
- each plant was incubated for 3 days to express EGFP transiently. Subsequently, each plant was irradiated with a blue LED, and EGFP fluorescence was observed using an ultraviolet absorption filter (model “SC-52”, Fujifilm).
- FIGS. 3A to 3I are photographs showing the results of observation of the fluorescence of the expressed EGFP.
- the scale bar indicates 1 cm.
- FIG. 3 (a) shows the results for Bensamiana tobacco leaves
- FIG. 3 (b) shows the results for lettuce leaves
- FIG. 3 (c) shows the results for eggplant leaves
- FIG. 3 (d) Is the result of tomato fruit
- FIG. 3 (e) is the result of tomato leaf
- FIG. 3 (f) is the result of pepper leaf
- FIG. 3 (g) is the result of melon leaf
- FIG. 3 (h) shows the result of rose petals
- FIG. 3 (i) shows the result of petals of moth orchid.
- the left side shows the result of introducing the pBYR2HS-EGFP vector
- the right side shows the result of introducing the pBYR2fp-EGFP vector.
- the pBYR2HS-EGFP vector having two terminators, the terminator derived from the Arabidopsis heat shock protein 18.2 gene and the terminator derived from the Arabidopsis heat shock protein, rather than the pBYR2fp-EGFP vector having one terminator of the tobacco extensin gene. It was revealed that the expression level of EGFP was significantly higher.
- the prepared protein was subjected to SDS-polyacrylamide gel electrophoresis (SDS-PAGE), and the total soluble protein was detected by Coomassie Brilliant Blue (CBB) staining.
- SDS-PAGE SDS-polyacrylamide gel electrophoresis
- CBB Coomassie Brilliant Blue
- SDS-PAGE gel was transferred to a PVDF membrane, and EGFP protein was detected by immunoblot analysis using an anti-GFP antibody.
- FIG. 4 (a) is a photograph showing the result of subjecting all soluble proteins prepared from the leaves of Bensamiana tobacco to SDS-PAGE and staining with CBB.
- the arrowhead indicates the EGFP protein.
- NT means total soluble protein prepared from the leaves of Bensamiana tobacco into which no vector has been introduced.
- GFP means a commercially available purified GFP protein (Vector Laboratories, Inc.).
- FIG. 4 (b) is a photograph showing the result of immunoblotting analysis using an anti-GFP antibody after transferring the gel of FIG. 4 (a) to a PVDF membrane.
- FIG. 4C is a graph in which the expression level of EGFP in FIG. The numerical value of the graph of FIG.4 (c) is shown by the average value +/- standard deviation.
- FIG. 5 (a) is a photograph showing the result of subjecting all soluble proteins prepared from lettuce leaves to SDS-PAGE and CBB staining.
- the arrowhead indicates the EGFP protein.
- “NT” means total soluble protein prepared from lettuce leaves into which no vector has been introduced.
- FIG. 5 (b) is a photograph showing the result of immunoblotting analysis using an anti-GFP antibody after transferring the gel of FIG. 5 (a) to a PVDF membrane.
- FIG. 5C is a graph in which the expression level of EGFP in FIG. The numerical value of the graph of FIG.5 (c) is shown by the average value +/- standard deviation.
- FIG. 6 (a) is a photograph showing the result of subjecting all soluble proteins prepared from eggplant leaves to SDS-PAGE and staining with CBB.
- the arrowhead indicates the EGFP protein.
- “NT” means total soluble protein prepared from eggplant leaves into which no vector has been introduced.
- FIG. 6B is a photograph showing the result of immunoblotting analysis using an anti-GFP antibody after transferring the gel of FIG. 6A to a PVDF membrane.
- FIG.6 (c) is the graph which digitized the expression level of EGFP in Fig.6 (a). The numerical value of the graph of FIG.6 (c) is shown by the average value +/- standard deviation.
- FIG. 7 (a) is a photograph showing the result of subjecting all soluble proteins prepared from tomato leaves to SDS-PAGE, transferring them to a PVDF membrane, and immunoblotting analysis using an anti-GFP antibody.
- “NT” means the total soluble protein prepared from tomato leaves into which no vector has been introduced.
- FIG. 7 (b) is a photograph showing the result of immunoblotting analysis using an anti-GFP antibody after transferring all soluble proteins prepared from capsicum leaves and rose petals to SDS-PAGE and then transferring them to a PVDF membrane. is there.
- “NT” means the total soluble protein prepared from the pepper leaf into which no vector has been introduced.
- pBYR2HS-EGFP having two terminators, that is, a tobacco extensin gene terminator and an Arabidopsis heat shock protein 18.2 gene terminator, rather than a pBYR2fp-EGFP vector having one tobacco extensin gene terminator. It was revealed that the expression level of EGFP was significantly higher in the vector. It was also revealed that this expression system functions not only in tobacco but also in a plurality of species of plants other than the genus Tobacco.
- Example 2 The outline of each vector is shown in Table 2 below.
- Example 3 ′ means the terminator of tobacco extensin gene
- HSPter means the terminator of Arabidopsis heat shock protein 18.2 gene
- 35Ster is the 35S terminator of cauliflower mosaic virus (CaMV).
- Nos-t means NOS terminator.
- each plant was incubated for 3 days to express EGFP transiently. Subsequently, each plant was irradiated with excitation light, and the fluorescence of EGFP was observed using an ultraviolet absorption filter (type “SC-52”, Fujifilm).
- FIGS. 8 (a) to (e) are photographs showing the results of observing the fluorescence of the expressed EGFP.
- the introduced vectors are shown in the upper part of FIGS. 8 (a) to (e), respectively.
- the vector, pBYR2TN-EGFP vector, pBYR2HT-EGFP vector, and pBYR2HTS-EGFP vector tended to have a higher expression level of EGFP.
- the prepared protein corresponding to 0.2 mg fresh weight (FW) was subjected to SDS-polyacrylamide gel electrophoresis (SDS-PAGE), and the total soluble protein was detected by Coomassie Brilliant Blue (CBB) staining.
- SDS-PAGE SDS-polyacrylamide gel electrophoresis
- CBB Coomassie Brilliant Blue
- FIG. 9 (a) is a photograph showing the result of subjecting all soluble proteins prepared from the leaves of Bensamiana tobacco to SDS-PAGE and CBB staining.
- the arrowhead indicates the EGFP protein.
- “NT” means total soluble protein prepared from the leaves of Bensamiana tobacco into which no vector has been introduced.
- FIG. 9B is a graph in which the expression level of EGFP in FIG. The numerical values in the graph of FIG. 9B are shown as an average value ⁇ standard deviation.
- the prepared protein corresponding to 0.2 mg fresh weight (FW) was subjected to SDS-polyacrylamide gel electrophoresis (SDS-PAGE), and the total soluble protein was detected by Coomassie Brilliant Blue (CBB) staining.
- SDS-PAGE SDS-polyacrylamide gel electrophoresis
- CBB Coomassie Brilliant Blue
- FIG. 10 (a) is a photograph showing the result of subjecting all soluble proteins prepared from the leaves of Bensamiana tobacco to SDS-PAGE and CBB staining.
- the arrowhead indicates the EGFP protein.
- “NT” means total soluble protein prepared from the leaves of Bensamiana tobacco into which no vector has been introduced.
- FIG. 10B is a graph in which the expression level of EGFP in FIG. The numerical value of the graph of FIG.10 (b) is shown by the average value +/- standard deviation.
- Table 3 shows the outline of each vector used in this experimental example and the expression level of EGFP.
- “Ext3 ′” means the terminator of tobacco extensin gene
- “HSPter” means the terminator of Arabidopsis heat shock protein 18.2 gene
- “35Ster” is the 35S terminator of cauliflower mosaic virus (CaMV).
- “Nos-t” means NOS terminator.
- the expression level of EGFP is expressed as an average value ⁇ standard deviation.
- EGFP high expression of EGFP was observed in any of the vectors. Specifically, in any of the vectors, about 3 mg or more of EGFP was expressed per 1 g of fresh weight of the plant body. In addition, it was recognized that the expression level of EGFP was higher in the vector having 2 terminators than in the vector having 3 terminators. Moreover, the vector which contains the terminator of Arabidopsis heat shock protein 18.2 gene in the terminator tended to have a particularly high expression level of EGFP. In particular, in the pBYR2HS-EGFP vector, about 4 mg of EGFP was expressed per 1 g of fresh weight of the plant body.
- magnICON system is an expression system currently used commercially, and it is known that about 3 mg or more of a target protein can be expressed per 1 g fresh weight of a plant body. Therefore, the expression levels of the proteins by the above-mentioned pBYR2HS-EGFP vector and the magnICON system were compared.
- FIG. 11 is a schematic diagram showing a structure of a GFP_pICH18711 vector (provided by Dr. Klimyuuk of Icon Genetics), which is an expression vector using the magnICON system.
- RB and “LB” mean the right and left border sequences of T-DNA
- Act2 means the Act2 promoter derived from Arabidopsis thaliana
- ⁇ is derived from tobacco mosaic virus.
- RdRp means RNA-dependent RNA polymerase derived from tobamovirus (Turnip bain clearing virus)
- MP means movement protein
- GFP green Fluorescence protein
- NTR means 3′-UTR derived from cr-TMV (Cruciferae infectious tobamovirus)
- Nos-t means NOS terminator.
- a region indicated by a box with “RdRp” and “MP” dots means an intron.
- the pBYR2HS-EGFP vector and the GFP_pICH18711 vector were introduced into the Agrobacterium tumefaciens GV3101 strain, respectively, and introduced into the leaves of 4-week-old or 5-week-old benthamiana tobacco.
- each plant was incubated for 3 days to express EGFP or GFP transiently. Subsequently, each plant was irradiated with a blue LED, and fluorescence of EGFP or GFP was observed using an ultraviolet absorption filter (model “SC-52”, Fujifilm).
- FIGS. 12 (a) and 12 (b) are photographs showing the results of observing the fluorescence of expressed EGFP or GFP. 12A and 12B, the scale bar indicates 1 cm.
- FIG. 12 (a) shows the results for 4-week-old benthamiana tobacco
- FIG. 12 (b) shows the results for 5-week-old benthamiana tobacco leaves.
- the introduced vectors are shown in the upper part of FIGS. 12 (a) and 12 (b), respectively.
- “4wo” indicates that the result is 4 weeks old
- “5wo” indicates that the result is 5 weeks old.
- the expression level of EGFP or GFP is greater when the pBYR2HS-EGFP vector is introduced than when the GFP_pICH18711 vector is introduced, regardless of whether 4-week-old or 5-week-old benthamiana tobacco leaves are used. A high tendency was observed.
- FIG. 13 (a) is a photograph showing the result of subjecting all soluble proteins prepared from the leaves of each Bensamiana tobacco to SDS-PAGE and staining with CBB.
- the introduced vectors are shown in the upper part of FIG.
- the arrowhead indicates EGFP or GFP protein
- “4wo” indicates a result of 4 weeks of age
- “5wo” indicates a result of 5 weeks of age.
- “NT” means total soluble protein prepared from the leaves of Bensamiana tobacco into which no vector has been introduced.
- FIG. 13 (b) is a graph in which the expression level of EGFP or GFP in FIG. 13 (a) is quantified.
- the numerical values in the graph of FIG. 13 (b) are shown as an average value ⁇ standard deviation of the expression level of EGFP or GFP in 4-week-old and 5-week-old benthamiana tobacco.
- “*” indicates that there is a significant difference at P ⁇ 0.05 as a result of Student's t-test.
- an expression system that can be applied to plants other than the genus Tobacco and has a high protein expression level can be provided.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Chemical & Material Sciences (AREA)
- Zoology (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Plant Pathology (AREA)
- Biophysics (AREA)
- Microbiology (AREA)
- General Health & Medical Sciences (AREA)
- Physics & Mathematics (AREA)
- Cell Biology (AREA)
- Biochemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Nutrition Science (AREA)
- Virology (AREA)
- Breeding Of Plants And Reproduction By Means Of Culturing (AREA)
- Peptides Or Proteins (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Abstract
Description
[1]ジェミニウイルス由来のLong Intergenic Region(LIR)と、ジェミニウイルス由来のSmall Intergenic Region(SIR)と、前記LIRと前記SIRとの間に連結された目的タンパク質の発現カセットとを含む第1の核酸断片と、ジェミニウイルス由来のRep/RepAタンパク質の発現カセットを含む第2の核酸断片と、を備え、前記目的タンパク質の発現カセットが、プロモーターと、前記目的タンパク質をコードする核酸断片と、2個以上連結されたターミネーターとをこの順に含む、発現システム。
[2]前記ターミネーターが2個連結されている、請求項1に記載の発現システム。
[3]前記ターミネーターの少なくとも1つが、シロイヌナズナ熱ショックタンパク質18.2遺伝子由来のターミネーターである、請求項1又は2に記載の発現システム。
[4]遺伝子サイレンシング阻害因子の発現カセットを含む第3の核酸断片を更に備える、[1]~[3]のいずれかに記載の発現システム。
[5]前記遺伝子サイレンシング阻害因子が、トマトブッシースタントウイルスに由来する遺伝子サイレンシング阻害因子P19である、[4]に記載の発現システム。
[6]前記第1の核酸断片、前記第2の核酸断片及び前記第3の核酸断片が、単一のベクターに含まれている、[4]又は[5]に記載の発現システム。
[7]T-DNA右側ボーダー配列(RB)及びT-DNA左側ボーダー配列(LB)を更に備え、前記第1の核酸断片、前記第2の核酸断片及び前記第3の核酸断片が、前記RBと前記LBとの間に存在する、[6]に記載の発現システム。
[8]前記ジェミニウイルスが、インゲン黄斑萎縮ウイルスである、[1]~[7]のいずれかに記載の発現システム。
[9]目的タンパク質の製造方法であって、植物細胞に[1]~[8]のいずれかに記載の発現システムを導入する工程を備える、製造方法。
[10]ジェミニウイルス由来のLIRと、ジェミニウイルス由来のSIRと、前記LIRと前記SIRとの間に連結された発現カセットとを含む第1の核酸断片を含み、前記発現カセットが、プロモーターと、マルチクローニングサイトと、2個以上連結されたターミネーターとをこの順に含む、発現ベクター。
[11]前記ターミネーターが2個連結されている、[10]に記載の発現ベクター。
[12]前記ターミネーターの少なくとも1つが、シロイヌナズナ熱ショックタンパク質18.2遺伝子由来のターミネーターである、[10]又は[11]に記載の発現ベクター。
[13]ジェミニウイルス由来のRep/RepAタンパク質の発現カセットを含む第2の核酸断片を更に含む、[10]~[12]のいずれかに記載の発現ベクター。
[14]遺伝子サイレンシング阻害因子の発現カセットを含む第3の核酸断片を更に含む、[10]~[13]のいずれかに記載の発現ベクター。
[15]前記遺伝子サイレンシング阻害因子が、トマトブッシースタントウイルスに由来する遺伝子サイレンシング阻害因子P19である、[14]に記載の発現ベクター。
[16]T-DNA右側ボーダー配列(RB)及びT-DNA左側ボーダー配列(LB)を更に備え、前記第1の核酸断片、前記第2の核酸断片及び前記第3の核酸断片が、前記RBと前記LBとの間に存在する、[10]~[15]のいずれか一項に記載の発現ベクター。
[17]前記ジェミニウイルスが、インゲン黄斑萎縮ウイルスである、[10]~[16]のいずれかに記載の発現ベクター。
1実施形態において、本発明は、ジェミニウイルス由来のLIRと、ジェミニウイルス由来のSIRと、前記LIRと前記SIRとの間に連結された目的タンパク質の発現カセットとを含む第1の核酸断片と、ジェミニウイルス由来のRep/RepAタンパク質の発現カセットを含む第2の核酸断片と、を備え、前記目的タンパク質の発現カセットが、プロモーターと、前記目的タンパク質をコードする核酸断片と、2個以上連結されたターミネーターとをこの順に含む、発現システムを提供する。
配列同一性(%)=一致した塩基数/対象塩基配列の総塩基数×100 (1)
1実施形態において、本発明は、ジェミニウイルス由来のLIRと、ジェミニウイルス由来のSIRと、前記LIRと前記SIRとの間に連結された発現カセットとを含む第1の核酸断片を含み、前記発現カセットが、プロモーターと、マルチクローニングサイトと、2個以上連結されたターミネーターとをこの順に含む、発現ベクターを提供する。
1実施形態において、本発明は、目的タンパク質の製造方法であって、植物細胞に上述した発現システムを導入する工程を備える、製造方法を提供する。ここで、発現システムは、上述したベクターのマルチクローニングサイトに目的タンパク質をコードする遺伝子断片が導入されたものであってもよい。実施例において後述するように、本実施形態の製造方法により、タバコ属植物及びタバコ属以外の植物を宿主として、高い発現量で目的タンパク質を製造することができる。
(レタスにおける一過性発現)
まず、バイナリーベクターを有するアグロバクテリウム・ツメファシエンスGV3101株を、抗生物質(100mg/Lカナマイシン、30mg/Lゲンタマイシン、30mg/Lリファンピシン)を添加したYEB培地(6g/Lイーストエキストラクト、5g/Lトリプトン、5g/Lスクロース、2mM硫酸マグネシウム)中、28℃で2日間培養した。
まず、バイナリーベクターを有するアグロバクテリウム・ツメファシエンスGV3101株を、10mM MES(pH5.6)、20μMアセトシリンゴン、100mg/Lカナマイシン、30mg/Lゲンタマイシン、30mg/Lリファンピシンを添加したL-ブロス培地中、28℃で静止期まで培養した。
《pBYR2fp-EGFPベクターの作製》
図2(a)はpBYR2fp-EGFPベクター(米国アリゾナ州立大学Mason博士より分与された。)のT-DNA領域の模式図である。既知のベクターであるpBYR2fpベクターは、インゲン黄斑萎縮ウイルス(BeYDV)由来の複製システムを有している。また、pBYR2fpベクターは、トマトブッシースタントウイルスに由来する遺伝子サイレンシング阻害因子P19の発現カセットを有している。
図2(b)はpBYR2HS-EGFPベクターのT-DNA領域の模式図である。pBYR2fpベクターに、アルコールデヒドロゲナーゼ遺伝子の5’-UTR及びシロイヌナズナ熱ショックタンパク質18.2遺伝子のターミネーターを有するEGFP遺伝子断片を導入した。
図2(c)はpBYR2EE-EGFPベクターのT-DNA領域の模式図である。pBYR2HS-EGFPベクターを鋳型として、プライマー(pBYR2EE-Ext3-F、配列番号7)及びプライマー(pBYR2EE-Ext3-R、配列番号8)を用いて、タバコエクステンシン遺伝子ターミネーターをPCR増幅した。得られたPCR産物を、制限酵素SalI及びXbaIで切断したpBYR2HS-EGFPベクターにクローニングし、pBYR2EE-EGFPベクターを作製した。pBYR2EE-EGFPベクターのターミネーターは、タバコエクステンシン遺伝子のターミネーターが2個連結したものであった。
図2(d)はpBYR2HH-EGFPベクターのT-DNA領域の模式図である。シロイヌナズナ熱ショックタンパク質18.2遺伝子のターミネーターをプライマー(pBYR2H-HSPter-F、配列番号9)及びpBYR2H-HSPter-R、配列番号10)を用いて、pBYR2HS-EGFPベクターを鋳型としてPCR増幅した。PCR産物を、制限酵素XbaIで切断したpBYR2H-EGFPにクローニングし、pBYR2HH-EGFPベクターを作製した。pBYR2HH-EGFPベクターのターミネーターは、シロイヌナズナ熱ショックタンパク質18.2遺伝子のターミネーターが2個連結したものであった。
図2(e)はpBYR2H-EGFPベクターのT-DNA領域の模式図である。pBYR2HS-EGFPからタバコエクステンシン遺伝子のターミネーターを除去するため、制限酵素XmaI及びClaIで切断した。SIR-C2をプライマー(HSPter-SIR-F、配列番号11)及びプライマー(C1-ClaI-C2-R、配列番号12)を用いて、pBYR2HS-EGFPベクターを鋳型として、PCR増幅した。上記XmaI及びClaIで切断したpBYR2HS-EGFPにクローニングし、pBYR2H-EGFPベクターを作製した。pBYR2H-EGFPベクターのターミネーターは、シロイヌナズナ熱ショックタンパク質18.2遺伝子のターミネーターを1個有するものであった。
図2(f)はpBYR2TN-EGFPベクターのT-DNA領域の模式図である。カリフラワーモザイクウイルス(CaMV)の35Sターミネーターはプライマー(pBYR2T-35Ster-F、配列番号13)及びプライマー(35Ster-NOSter-R、配列番号14)を用いて、pCambia1391Z(Marker Gene Technologies,Inc.社)を鋳型として、PCR増幅した。NOSターミネーターはプライマー(35Ster-NOSter-F、配列番号15)及びプライマー(pBYR2TN-NOSter-R、配列番号16)を用いて、pRI201-AN(タカラバイオ社)を鋳型として、PCR増幅した。PCR産物を、制限酵素SalI及びXbaIで切断したpBYR2H-EGFPベクターにクローニングし、pBYR2TN-EGFPを作製した。pBYR2TN-EGFPベクターのターミネーターは、カリフラワーモザイクウイルス(CaMV)の35Sターミネーター及びNOSターミネーターが、それぞれ1個ずつ連結したものであった。
図2(g)はpBYR2T-EGFPベクターのT-DNA領域の模式図である。カリフラワーモザイクウイルス(CaMV)の35Sターミネーターはプライマー(pBYR2T-35Ster-F、配列番号13)及びプライマー(pBYR2HS-35Ster-R、配列番号17)を用いて、pCambia1391Zを鋳型として、PCR増幅した。PCR産物を、制限酵素SalI及びXbaIで切断したpBYR2H-EGFPベクターにクローニングし、pBYR2T-EGFPベクターを作製した。pBYR2T-EGFPベクターのターミネーターは、カリフラワーモザイクウイルス(CaMV)の35Sターミネーターを1個有するものであった。
図2(h)はpBYR2HT-EGFPベクターのT-DNA領域の模式図である。カリフラワーモザイクウイルス(CaMV)の35Sターミネーターはプライマー(pBYR2HS-35Ster-F、配列番号18)及びプライマー(pBYR2HS-35Ster-R、配列番号17)を用いて、pCambia1391Zを鋳型として、PCR増幅した。PCR産物を、制限酵素XbaIで切断したpBYR2H-EGFPベクターにクローニングし、pBYR2HT-EGFPベクターを作製した。pBYR2HT-EGFPベクターのターミネーターは、シロイヌナズナ熱ショックタンパク質18.2遺伝子のターミネーター及びカリフラワーモザイクウイルス(CaMV)の35Sターミネーターが、それぞれ1個ずつ連結したものであった。
図2(i)はpBYR2HTS-EGFPベクターのT-DNA領域の模式図である。カリフラワーモザイクウイルス(CaMV)の35Sターミネーターはプライマー(pBYR2HS-35Ster-F、配列番号18)及びプライマー(pBYR2HS-35Ster-R、配列番号17)を用いて、pCambia1391Zを鋳型として、PCR増幅した。PCR産物を、制限酵素XbaIで切断したpBYR2HS-EGFPベクターにクローニングし、pBYR2HTS-EGFPベクターを作製した。pBYR2HTS-EGFPベクターのターミネーターは、シロイヌナズナ熱ショックタンパク質18.2遺伝子のターミネーター、カリフラワーモザイクウイルス(CaMV)の35Sターミネーター及び、タバコエクステンシン遺伝子のターミネーターが、それぞれ1個ずつ連結したものであった。
(pBYR2HS-EGFPベクター及びpBYR2fp-EGFPベクターによるEGFPの発現レベルの比較1)
pBYR2HS-EGFPベクターは、pBYR2fp-EGFPベクターのTMVΩがシロイヌナズナアルコールデヒドロゲナーゼ遺伝子の5’-UTRに置き換えられ、更にシロイヌナズナ熱ショックタンパク質18.2遺伝子のターミネーターが挿入された、ターミネーターを2つ有するベクターであった。
(pBYR2HS-EGFPベクター及びpBYR2fp-EGFPベクターによるEGFPの発現レベルの比較2)
pBYR2HS-EGFPベクター及びpBYR2fp-EGFPベクターを、それぞれアグロバクテリウム・ツメファシエンスGV3101株に導入し、アグロインフィルトレーションにより、ベンサミアナタバコの葉、レタスの葉、ナスの葉、トマトの葉、トウガラシの葉及びバラの花弁にそれぞれ導入した。続いて、各植物を3日間インキュベートしてEGFPを一過性に発現させた。
(様々なターミネーターを有するベクターによるEGFPの発現レベルの比較1)
ターミネーターを1個しか有していない、pBYR2fp-EGFPベクター、pBYR2H-EGFPベクター、pBYR2T-EGFPベクター、ターミネーターを2個有する、pBYR2HS-EGFPベクター、pBYR2EE-EGFPベクター、pBYR2HH-EGFPベクター、pBYR2TN-EGFPベクター、pBYR2HT-EGFPベクター、及び、ターミネーターを3個有する、pBYR2HTS-EGFPベクターを、それぞれアグロバクテリウム・ツメファシエンスGV3101株に導入し、ベンサミアナタバコの葉にそれぞれ導入した。
(様々なターミネーターを有するベクターによるEGFPの発現レベルの比較2)
ターミネーターを1個しか有していない、pBYR2fp-EGFPベクター、pBYR2H-EGFPベクター、及び、ターミネーターを2個有する、pBYR2HS-EGFPベクターを、それぞれアグロバクテリウム・ツメファシエンスGV3101株に導入し、アグロインフィルトレーションにより、ベンサミアナタバコの葉にそれぞれ導入した。続いて、各植物を3日間インキュベートしてEGFPを一過性に発現させた。続いて、ベクターを導入したベンサミアナタバコの葉から、全可溶性タンパク質をそれぞれ調製した。
(様々なターミネーターを有するベクターによるEGFPの発現レベルの比較3)
ターミネーターを2個有する、pBYR2HS-EGFPベクター、pBYR2HH-EGFPベクター、pBYR2EE-EGFPベクター、pBYR2TN-EGFPベクター、pBYR2HT-EGFPベクター、及び、ターミネーターを3個有するpBYR2HTS-EGFPベクターを、それぞれアグロバクテリウム・ツメファシエンスGV3101株に導入し、アグロインフィルトレーションにより、ベンサミアナタバコの葉にそれぞれ導入した。続いて、各植物を3日間インキュベートしてEGFPを一過性に発現させた。続いて、ベクターを導入したベンサミアナタバコの葉から、全可溶性タンパク質をそれぞれ調製した。
(pBYR2HS-EGFPベクター及びmagnICONシステムによるタンパク質の発現レベルの比較)
上述したように、magnICONシステムは、現在商用で用いられている発現システムであり、植物体1g新鮮重量あたり3mg程度以上の目的タンパク質を発現させることができることが知られている。そこで、上述したpBYR2HS-EGFPベクター及びmagnICONシステムによるタンパク質の発現レベルを比較した。
Claims (17)
- ジェミニウイルス由来のLong Intergenic Region(LIR)と、ジェミニウイルス由来のSmall Intergenic Region(SIR)と、前記LIRと前記SIRとの間に連結された目的タンパク質の発現カセットとを含む第1の核酸断片と、
ジェミニウイルス由来のRep/RepAタンパク質の発現カセットを含む第2の核酸断片と、を備え、
前記目的タンパク質の発現カセットが、プロモーターと、前記目的タンパク質をコードする核酸断片と、2個以上連結されたターミネーターとをこの順に含む、発現システム。 - 前記ターミネーターが2個連結されている、請求項1に記載の発現システム。
- 前記ターミネーターの少なくとも1つが、シロイヌナズナ熱ショックタンパク質18.2遺伝子由来のターミネーターである、請求項1又は2に記載の発現システム。
- 遺伝子サイレンシング阻害因子の発現カセットを含む第3の核酸断片を更に備える、請求項1~3のいずれか一項に記載の発現システム。
- 前記遺伝子サイレンシング阻害因子が、トマトブッシースタントウイルスに由来する遺伝子サイレンシング阻害因子P19である、請求項4に記載の発現システム。
- 前記第1の核酸断片、前記第2の核酸断片及び前記第3の核酸断片が、単一のベクターに含まれている、請求項4又は5に記載の発現システム。
- T-DNA右側ボーダー配列(RB)及びT-DNA左側ボーダー配列(LB)を更に備え、前記第1の核酸断片、前記第2の核酸断片及び前記第3の核酸断片が、前記RBと前記LBとの間に存在する、請求項6に記載の発現システム。
- 前記ジェミニウイルスが、インゲン黄斑萎縮ウイルスである、請求項1~7のいずれか一項に記載の発現システム。
- 目的タンパク質の製造方法であって、植物細胞に請求項1~8のいずれか一項に記載の発現システムを導入する工程を備える、製造方法。
- ジェミニウイルス由来のLIRと、ジェミニウイルス由来のSIRと、前記LIRと前記SIRとの間に連結された発現カセットとを含む第1の核酸断片を含み、前記発現カセットが、プロモーターと、マルチクローニングサイトと、2個以上連結されたターミネーターとをこの順に含む、発現ベクター。
- 前記ターミネーターが2個連結されている、請求項10に記載の発現ベクター。
- 前記ターミネーターの少なくとも1つが、シロイヌナズナ熱ショックタンパク質18.2遺伝子由来のターミネーターである、請求項10又は11に記載の発現ベクター。
- ジェミニウイルス由来のRep/RepAタンパク質の発現カセットを含む第2の核酸断片を更に含む、請求項10~12のいずれか一項に記載の発現ベクター。
- 遺伝子サイレンシング阻害因子の発現カセットを含む第3の核酸断片を更に含む、請求項10~13のいずれか一項に記載の発現ベクター。
- 前記遺伝子サイレンシング阻害因子が、トマトブッシースタントウイルスに由来する遺伝子サイレンシング阻害因子P19である、請求項14に記載の発現ベクター。
- T-DNA右側ボーダー配列(RB)及びT-DNA左側ボーダー配列(LB)を更に備え、前記第1の核酸断片、前記第2の核酸断片及び前記第3の核酸断片が、前記RBと前記LBとの間に存在する、請求項10~15のいずれか一項に記載の発現ベクター。
- 前記ジェミニウイルスが、インゲン黄斑萎縮ウイルスである、請求項10~16のいずれか一項に記載の発現ベクター。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA3059025A CA3059025C (en) | 2017-05-31 | 2018-03-06 | Protein expression system in plant cell and use thereof |
| US16/603,741 US20210108218A1 (en) | 2017-05-31 | 2018-03-06 | Protein expression system in plant cell and use thereof |
| JP2019521960A JP6850041B2 (ja) | 2017-05-31 | 2018-03-06 | 植物細胞でのタンパク質発現システム及びその使用 |
| EP18810050.7A EP3604546A4 (en) | 2017-05-31 | 2018-03-06 | SYSTEM OF PROTEIN EXPRESSION IN A PLANT CELL AND ASSOCIATED USE |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017-107965 | 2017-05-31 | ||
| JP2017107965 | 2017-05-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018220929A1 true WO2018220929A1 (ja) | 2018-12-06 |
Family
ID=64456049
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2018/008512 WO2018220929A1 (ja) | 2017-05-31 | 2018-03-06 | 植物細胞でのタンパク質発現システム及びその使用 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20210108218A1 (ja) |
| EP (1) | EP3604546A4 (ja) |
| JP (1) | JP6850041B2 (ja) |
| CA (1) | CA3059025C (ja) |
| WO (1) | WO2018220929A1 (ja) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10894812B1 (en) | 2020-09-30 | 2021-01-19 | Alpine Roads, Inc. | Recombinant milk proteins |
| WO2021020421A1 (ja) | 2019-08-01 | 2021-02-04 | 国立大学法人筑波大学 | 細胞死抑制剤及び細胞死抑制方法 |
| US10947552B1 (en) | 2020-09-30 | 2021-03-16 | Alpine Roads, Inc. | Recombinant fusion proteins for producing milk proteins in plants |
| US11840717B2 (en) | 2020-09-30 | 2023-12-12 | Nobell Foods, Inc. | Host cells comprising a recombinant casein protein and a recombinant kinase protein |
| WO2023218388A3 (en) * | 2022-05-11 | 2023-12-21 | Università Degli Studi Di Bari Aldo Moro | Process for the production of irisin, its formulations and its administration routes |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB202107598D0 (en) * | 2021-05-27 | 2021-07-14 | Leaf Expression Systems Ltd | Recombinant protein production |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016182045A (ja) * | 2015-03-25 | 2016-10-20 | 国立研究開発法人農業・食品産業技術総合研究機構 | 葯特異的にプロモーター活性を有するdna、及びその利用 |
| JP2017107965A (ja) | 2015-12-09 | 2017-06-15 | 日亜化学工業株式会社 | パッケージの製造方法及び発光装置の製造方法、並びにパッケージ及び発光装置 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BR112014031891A2 (pt) * | 2012-06-19 | 2017-08-01 | Univ Minnesota | direcionamento genético nas plantas utilizando vírus de dna |
-
2018
- 2018-03-06 JP JP2019521960A patent/JP6850041B2/ja active Active
- 2018-03-06 WO PCT/JP2018/008512 patent/WO2018220929A1/ja unknown
- 2018-03-06 CA CA3059025A patent/CA3059025C/en active Active
- 2018-03-06 EP EP18810050.7A patent/EP3604546A4/en active Pending
- 2018-03-06 US US16/603,741 patent/US20210108218A1/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016182045A (ja) * | 2015-03-25 | 2016-10-20 | 国立研究開発法人農業・食品産業技術総合研究機構 | 葯特異的にプロモーター活性を有するdna、及びその利用 |
| JP2017107965A (ja) | 2015-12-09 | 2017-06-15 | 日亜化学工業株式会社 | パッケージの製造方法及び発光装置の製造方法、並びにパッケージ及び発光装置 |
Non-Patent Citations (11)
| Title |
|---|
| BEYENE, G. ET AL.: "Unprecedented enhancement of transient gene expression from minimal cassettes using a double terminator", PLANT CELL REP., vol. 30, no. 1, 22 October 2010 (2010-10-22), pages 13 - 25, XP019869854, DOI: 10.1007/s00299-010-0936-3 * |
| DIAMOS, A.G. ET AL.: "5' and 3' Untranslated Regions Strongly Enhance Performance of Geminiviral Replicons in Nicotiana benthamiana Leaves.", FRONT. PLANT. SCI., vol. 7, 200, 24 February 2016 (2016-02-24), XP055503953, DOI: 10.3389/fpls.2016.00200 * |
| LUO, Z; CHEN, Z.: "Improperly Terminated, Unpolyadenylated mRNA of Sense Transgenes Is Targeted by RDR6-Mediated RNA Silencing in Arabidopsis", THE PLANT CELL, vol. 19, no. 3, March 2007 (2007-03-01), pages 943 - 958, XP055634979, DOI: 10.1105/tpc.106.045724 * |
| MARILLONNET S. ET AL.: "Systemic Agrobacterium tumefaciens-mediated transfection of viral replicons for efficient transient expression in plants", NAT. BIOTECHNOL., vol. 23, no. 6, 2005, pages 718 - 723, XP002407902, DOI: 10.1038/nbt1094 |
| MIURA, KENJI ET AL.: "P-103: Improvement of transient expression system for production of recombinant proteins", 35TH ANNUAL MEETING OF THE JAPANESE SOCIETY FOR PLANT CELL MOLECULAR BIOLOGY (SAITAMA) ; 2017/08/29-31, vol. 35, 20 August 2017 (2017-08-20), pages 180, XP009517311 * |
| MIURA, KENJI: "23. Study for expression of valuable proteins in a mass using plants", PROCEEDINGS OF THE 2015 SEMINAR ON GRANT-SUPPORTED RESEARCH FINDINGS, vol. 2016, 2016, JAPAN, pages 46 - 47, XP009518324 * |
| MIURA, KENJI: "ROMBUNNO.25: Study for expression of valuable proteins in a mass using plants", vol. 2016, 2016, pages 1 - 6, XP009518326, Retrieved from the Internet <URL:https://jglobal.jst.go.jp/en/detail?JGLOBAL_ID=201702238944424921> * |
| MOON K. B. ET AL.: "Overexpression and self-assembly of virus-like particles in Nicotiana benthamiana by a single-vector DNA replicon system", APPL. MICROBIOL. BIOTECHNOL., vol. 98, no. 19, 2014, pages 8281 - 8290 |
| NAGAYA, S. ET AL.: "The HSP terminator of Arabidopsis Thaliana increases gene expression in plant cells", PLANT CELL PHYSIOL., vol. 51, no. 2, February 2010 (2010-02-01), pages 328 - 332, XP055140803, DOI: 10.1093/pcp/pcp188 * |
| See also references of EP3604546A4 |
| YAMAMOTO, T. ET AL.: "Improvement of the transient expression system for production of recombinant proteins in plants", SCIENTIFIC REPORTS, vol. 8, 4755, 19 March 2018 (2018-03-19), pages 1 - 10, XP055635026, DOI: 10.1038/s41598-018-23024-y * |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021020421A1 (ja) | 2019-08-01 | 2021-02-04 | 国立大学法人筑波大学 | 細胞死抑制剤及び細胞死抑制方法 |
| US10894812B1 (en) | 2020-09-30 | 2021-01-19 | Alpine Roads, Inc. | Recombinant milk proteins |
| US10947552B1 (en) | 2020-09-30 | 2021-03-16 | Alpine Roads, Inc. | Recombinant fusion proteins for producing milk proteins in plants |
| US10988521B1 (en) | 2020-09-30 | 2021-04-27 | Alpine Roads, Inc. | Recombinant milk proteins |
| US11034743B1 (en) | 2020-09-30 | 2021-06-15 | Alpine Roads, Inc. | Recombinant milk proteins |
| US11072797B1 (en) | 2020-09-30 | 2021-07-27 | Alpine Roads, Inc. | Recombinant fusion proteins for producing milk proteins in plants |
| US11142555B1 (en) | 2020-09-30 | 2021-10-12 | Nobell Foods, Inc. | Recombinant milk proteins |
| US11401526B2 (en) | 2020-09-30 | 2022-08-02 | Nobell Foods, Inc. | Recombinant fusion proteins for producing milk proteins in plants |
| US11685928B2 (en) | 2020-09-30 | 2023-06-27 | Nobell Foods, Inc. | Recombinant fusion proteins for producing milk proteins in plants |
| US11840717B2 (en) | 2020-09-30 | 2023-12-12 | Nobell Foods, Inc. | Host cells comprising a recombinant casein protein and a recombinant kinase protein |
| US11952606B2 (en) | 2020-09-30 | 2024-04-09 | Nobell Foods, Inc. | Food compositions comprising recombinant milk proteins |
| WO2023218388A3 (en) * | 2022-05-11 | 2023-12-21 | Università Degli Studi Di Bari Aldo Moro | Process for the production of irisin, its formulations and its administration routes |
Also Published As
| Publication number | Publication date |
|---|---|
| US20210108218A1 (en) | 2021-04-15 |
| CA3059025C (en) | 2023-03-21 |
| EP3604546A4 (en) | 2020-12-30 |
| JP6850041B2 (ja) | 2021-03-31 |
| CA3059025A1 (en) | 2018-12-06 |
| EP3604546A1 (en) | 2020-02-05 |
| JPWO2018220929A1 (ja) | 2020-04-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2018220929A1 (ja) | 植物細胞でのタンパク質発現システム及びその使用 | |
| Norkunas et al. | Improving agroinfiltration-based transient gene expression in Nicotiana benthamiana | |
| Bouton et al. | Foxtail mosaic virus: a viral vector for protein expression in cereals | |
| Mardanova et al. | Efficient transient expression of recombinant proteins in plants by the novel pEff vector based on the genome of potato virus X | |
| Marillonnet et al. | Systemic Agrobacterium tumefaciens–mediated transfection of viral replicons for efficient transient expression in plants | |
| Zhang et al. | Bean yellow dwarf virus replicons for high‐level transgene expression in transgenic plants and cell cultures | |
| Ghosh et al. | Establishment of embryogenic cell suspension cultures and Agrobacterium-mediated transformation in an important Cavendish banana cv. Robusta (AAA) | |
| EP2044204A2 (en) | Plant expression constructs and methods of utilizing same | |
| AU2001242516A1 (en) | Cestrum yellow leaf curling virus promoters | |
| WO2001073087A1 (en) | Cestrum yellow leaf curling virus promoters | |
| US8222488B2 (en) | Enhancing expression of value-added genes by transgenic expression of tombusvirus-based P19 gene mutants | |
| Azhakanandam et al. | Amplicon-plus targeting technology (APTT) for rapid production of a highly unstable vaccine protein in tobacco plants | |
| Yoon et al. | Agrobacterium-mediated infection of whole plants by yellow dwarf viruses | |
| Sainsbury et al. | Cowpea mosaic virus-based systems for the expression of antigens and antibodies in plants | |
| JP6350995B2 (ja) | 植物において外来遺伝子を発現させるための核酸分子及び方法 | |
| US11649465B2 (en) | Methods and compositions for increasing expression of genes of interest in a plant by co-expression with p21 | |
| JP5230608B2 (ja) | P15ヘアピン構造及びその使用方法 | |
| US20220235362A1 (en) | Geminiviral vectors that reduce cell death and enhance expression of biopharmaceutical proteins | |
| FR2798139A1 (fr) | Vecteurs synthetiques propres, plasmides, plantes et parties de plantes transgeniques les contenant, et leurs methodes d'obtention | |
| CA2329509C (en) | Improved methods and materials for transformation | |
| Kopertekh et al. | Utilization of PVX-Cre expression vector in potato | |
| Mitiouchkina et al. | Molecular biology approach for improving chrysanthemum resistance to virus B | |
| CN112390866B (zh) | OsARF12基因在提高水稻对水稻矮缩病毒抗性中的应用 | |
| Ellison | Development of RNA Viral Vectors for Plant Genome Engineering | |
| Xie et al. | Expressing activity of promoter elements of large intergenic region from cotton leaf curl virus in host plant |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18810050 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2019521960 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 3059025 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2018810050 Country of ref document: EP Effective date: 20191024 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |