WO2017170300A1 - 老化を反映するミトコンドリアバイオマーカー - Google Patents
老化を反映するミトコンドリアバイオマーカー Download PDFInfo
- Publication number
- WO2017170300A1 WO2017170300A1 PCT/JP2017/012205 JP2017012205W WO2017170300A1 WO 2017170300 A1 WO2017170300 A1 WO 2017170300A1 JP 2017012205 W JP2017012205 W JP 2017012205W WO 2017170300 A1 WO2017170300 A1 WO 2017170300A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- uur
- taurine
- trna leu
- mitochondrial
- aging
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/17—Amino acids, peptides or proteins
- A23L33/18—Peptides; Protein hydrolysates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/46—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing sulfur
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/46—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing sulfur
- A61K8/466—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing sulfur containing sulfonic acid derivatives; Salts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/08—Anti-ageing preparations
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6809—Methods for determination or identification of nucleic acids involving differential detection
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6813—Hybridisation assays
- C12Q1/6816—Hybridisation assays characterised by the detection means
- C12Q1/6825—Nucleic acid detection involving sensors
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/106—Pharmacogenomics, i.e. genetic variability in individual responses to drugs and drug metabolism
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/148—Screening for cosmetic compounds
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/156—Polymorphic or mutational markers
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/158—Expression markers
Definitions
- the present invention relates to a mitochondrial biomarker that reflects aging, and relates to the field of visualizing aging and providing an index of healthy life expectancy.
- Mitochondria are intracellular organelles responsible for aerobic metabolism of somatic cells, and produce ATP as energy by hydrogen transmission system and oxidative phosphorylation. This mitochondrial function is thought to be impaired not only by mitochondrial diseases caused by mutations in mitochondrial DNA, which has a replication mechanism independent of the genome, but also by aging. Therefore, early diagnosis of mitochondrial dysfunction and evaluation of the degree of disability not only play an important role in the selection of treatment policy and prognosis determination of patients with mitochondrial disease, but are also attracting attention as aging markers.
- MELAS is the most common mitochondrial disease and exhibits various and characteristic symptoms such as Myopathy, Encephalopathy, Lactic acidosis and Stroke-like episodes. Life expectancy from diagnosis is 5 to 10 years. It is a refractory rare disease with no indications. Since stroke-like seizures progress repeatedly, anticipation of recurrence as soon as possible is awaited. MELAS is caused by a single base substitution (A3243G, T3271C, G3244A, T3258C, T3291C) of the tRNA Leu (UUR) gene coding region of mitochondrial DNA, but its basic pathology has long been unknown. Ota et al.
- Non-patent Document 1 MELAS mutant tRNA Leu
- Non-Patent Document 3 A review on the role of tRNA modification in human diseases has been published (Non-Patent Document 3), and a measurement method on lack of taurine modification of mitochondrial tRNA in human mitochondrial disease (Non-Patent Document 4) and a review have also been published (Non-Patent Document 5). ). In addition, several patent applications have been filed for diagnosis or treatment focusing on mitochondrial tRNA. 1) A method for diagnosing type 2 diabetes by measuring the chemical modification of tRNA including mitochondrial tRNA, specifically, the thiomethyl modification rate of tRNA encoding lysine is known (Patent Document 1).
- Patent Document 2 A method for improving the health of elderly animals by preventing or repairing changes in metabolism accompanying aging, particularly changes associated with mitochondrial dysfunction is known (Patent Document 2).
- Glutathione is a major intracellular antioxidant system that prevents oxidative damage of mitochondrial DNA (mtDNA), but with the aging of glutathione itself, the oxidation of glutathione itself progresses and its antioxidant function decreases.
- oxidative damage of mtDNA can be prevented by supplying a thiol compound such as taurine.
- taurine a thiol compound
- Patent Document 3 A therapeutic agent for mitochondrial disease containing taurine, taurine chloramine, or taurine precursor as an active ingredient is known (Patent Document 3).
- UUR normal mitochondrial tRNA Leu
- the first object of the present invention is to provide a novel biomarker that leads to early detection of mitochondrial dysfunction and evaluation of the degree of damage associated with aging.
- a second object of the present invention is to provide an agent for reducing the rate of taurine modification of mitochondrial tRNA Leu (UUR) accompanying aging, which causes a decrease in the function of mycotondoria.
- UUR mitochondrial tRNA Leu
- lactic acid levels in blood and cerebrospinal fluid which have been used clinically as markers for mitochondrial diseases, and the ratio of lactic acid levels to pyruvate levels are not only increased due to the pathological conditions in which anaerobic metabolism is increased in mitochondrial diseases. It is a low-specificity marker that is elevated by various conditions such as increased glycolytic metabolism due to hypoxia, ie, shock, respiratory failure, disseminated intravascular coagulation syndrome (Kraut JA, Madias NE. Lactic acidosis. N Engl J Med. 2015; 372 (11): 1078-1079).
- the present inventors measured the taurine modification rate of mitochondrial tRNA Leu (UUR) in leukocyte specimens of subjects and intensively studied its usefulness as a marker for mitochondrial disease. Specifically, not only decreased taurine modification rate of mutant mitochondrial tRNA Leu (UUR) in patients with mitochondrial disease, but also decreased taurine modification rate of normal mitochondrial tRNA Leu (UUR).
- the inventors have found that the taurine modification rate of mitochondrial tRNA Leu (UUR) can be an indicator of aging, and have conceived that it can be applied not only to patients with mitochondrial diseases but also to healthy individuals, and have completed the present invention.
- the present invention provides the following.
- a method for measuring the aging of a subject comprising measuring the degree of taurine modification of mitochondrial tRNA Leu (UUR) in a biological sample isolated from the subject by a reverse transcription reaction from a primer using the tRNA Leu (UUR) as a template. Measuring method.
- the primer is an oligonucleotide having a length of 10 to 25 bases complementary to the template, and a difference in the primer extension product due to the presence or absence of taurine modification is detected.
- the primer comprises at least one base sequence selected from the group consisting of the base sequences represented by SEQ ID NOs: 3, 4, and 5.
- U represents the amount of primer extension product from the tRNA Leu (UUR) with taurine modification
- U is indicative of the amount of primer extension product from no taurine-modified tRNA Leu (UUR)
- the method according to any one of [1] to [4], wherein the total taurine modification rate represented is used as an index of aging.
- the subject is suspected of having a mutated mitochondrial DNA having a single base substitution selected from the group consisting of A3243G, T3271C, G3244A, T3258C and T3291C of the tRNA Leu (UUR) gene coding region, Following 1), the following formula (2):
- the mtDNA point mutation rate is represented by the A-position at position 14 (A3243G), the G-position at position 15 (G3244A), the T-position at position 29 (DNA ) encoding tRNA Leu (UUR) represented by SEQ ID NO: 1 Normal taurine modification rate determined by T3258C), T at position 42 (T3271C) and T point at position 62 (showing the highest point mutation rate among point mutations selected from the group consisting of T3291C) as an index of aging The method according to [5]. [7] An agent for determining aging, comprising a primer comprising a base sequence consisting of 10 to 25 bases complementary to mitochondrial tRNA Leu (UUR) .
- the mtDNA point mutation rate is represented by the A-position at position 14 (A3243G), the G-position at position 15 (G3244A), the T-position at position 29 (DNA ) encoding tRNA Leu (UUR) represented by SEQ ID NO: 1 T3258C), showing the highest rate of point mutation among point mutations selected from the group consisting of T at position 42 (T3271C) and T at position 62 (T3291C)).
- the improver according to [11] or [12] which is a medicine or cosmetic.
- the improver according to [11] or [12] which is a health functional food or food additive.
- the taurine modification rate of normal tRNA Leu is a conventional mitochondrial disease marker, the ratio of blood lactate level, blood lactate level and pyruvate level, Cerebrospinal fluid lactic acid levels, cerebrospinal fluid lactic acid levels, and pyruvate levels were found to be correlated with aging. Therefore, the taurine modification rate of normal tRNA Leu (UUR) is a universal marker that can grasp the state of physiological functions of healthy individuals as well as patients with mitochondrial diseases.
- aging means that the biological function is lowered due to the influence of aging or other internal or external factors such as genetic factors and life / environmental factors. Aging is the physical passage of time from birth to death and is synonymous with calendar age. Other internal factors include those related to cellular senescence such as mitochondrial function, peroxide radical concentration, and telomere length. Other external factors include, for example, individual exercise amount, the presence or absence of smoking, dietary habits, and nutritional status. Examples of biological functions include muscle strength, nerve conduction velocity, vital capacity, and resistance to disease. Aging by aging generally begins after reaching the reproductive age, and there are individual differences, but in humans it begins after 20 to 30 years of age.
- Aging is thought to involve environmental factors and genetic predispositions that are intricately intertwined, and the speed of such functional decline is not the same for all humans, and it cannot always be defined only by factors such as aging. Previously there was no clear aging indicator.
- the index of aging provided by the present invention can also be used from the viewpoint of healthy life expectancy. “Aging” of the present invention refers to physical functions associated with so-called aging such as a decrease in height and weight, external changes such as changes in skin and hair, a decrease in motor function, a decrease in sensory function, or a decrease in physiological function.
- aging of the cells that make up the body (cell aging), as well as the aging of mitochondria that perform important functions in respiration and energy metabolism are included.
- UUR mitochondrial tRNA Leu
- the taurine modification rate of normal mitochondrial tRNA Leu can be used as an indicator of mitochondrial aging and functional decline, but it can be used as a living body, organ (organ), tissue, cell, or It is also useful as an indicator of the function of intracellular organelles, in particular, the aging and functional deterioration of organs, tissues, cells, or organelles with high demand for living organisms and energy.
- the above index is useful for selecting a target for taurine therapy for patients with mitochondrial diseases currently under development, for companion diagnosis used for determining the therapeutic effect, and for assisting it.
- healthy life expectancy is defined by the Ministry of Health, Labor and Welfare as the period during which people can live without being restricted due to health problems.
- Each country, including the World Health Organization, aims to increase the healthy life expectancy.
- mitochondrial tRNA Leu is a tRNA that is transcribed from mitochondrial DNA and recognizes the leucine codon UUR (R is A or G).
- Mitochondrial tRNA Leu defines the positions of nucleotides (UAA) corresponding to anticodons as positions 34 to 36, since the bases at positions 17, 20, and 47 increase or decrease depending on the animal species.
- DNA encoding human mitochondrial tRNA Leu (UUR) has been registered with NCBI and published under GenBank accession number AB026838 (version AB026838.1) (SEQ ID NO: 1).
- the mitochondrial tRNA Leu (UUR) transcribed from the DNA published in SEQ ID NO: 1 is represented by SEQ ID NO: 2.
- the uridine (U) at position 36 of SEQ ID NO: 2 may or may not be taurine-modified.
- mitochondrial tRNA Leu (UUR) may be simply abbreviated as “tRNA Leu (UUR) ”.
- mutant tRNA Leu are the DNA encoding tRNA Leu (UUR) represented by SEQ ID NO: 1, A at position 14 (A3243G), G at position 15 (G3244A), T at position 29 (T ( T3258C), T mutation at position 42 (T3271C), point mutation at position T (T3291C): these mutations result in a lack of taurine modification and are mutations found in MELAS patients; G at position 13 (A3242A), 21-position T (T3250C), 25-position C (C3254T), 51-position A (A3280G) point mutations: These mutations have no effect on taurine modification and are found in mitochondrial diseases other than MELAS And the like; For other mutations, reference can be made to MITOMAP (www.mitomap.org/MITOMAP).
- the mutant tRNA Leu (UUR) in the present invention means the transferred tRNA Leu (UUR) from mtDNA with mutations that cause loss of taurine-modified, free of tRNA Leu (UUR) with mutations that do not affect the taurine-modified Make it not exist.
- the normal tRNA Leu (UUR) refers to the tRNA Leu (UUR) except for the above-mentioned mutant tRNA Leu (UUR).
- tRNA Leu of SEQ ID NO: 2 tRNA having tRNA Leu (UUR) and different base sequence represented by tRNA Leu (UUR) and SEQ ID NO: 2 having the same base sequence (UUR) Leu ( even UUR), but differences in the base sequences include tRNA Leu (UUR) that does not affect the taurine modification tRNA Leu (UUR), the function of the normal tRNA Leu or without taurine-modified (UUR) Does not include items that do not have
- the 14th A, the 15th G, the 29th T, the 42nd T and the 62nd T are conserved in gorillas, cattle, pigs, dogs, cats, etc. in addition to humans.
- the mutation that causes the lack of taurine modification of tRNA Leu is not limited to humans, and will be described later. Mammals such as primates, pets, farm animals and laboratory animals can also be targeted.
- taurine modification of mitochondrial tRNA Leu means that a taurinomethyl group is bound to the 5th position of the uracil base of the first letter of the anticodon (UAA) of tRNA Leu (UUR) (the wobble position at position 34 according to the above definition).
- taurine-modified uridine may be indicated by the abbreviation ⁇ m 5 U.
- mtDNA mitochondrial DNA
- heteroplasmy a state composed of a single mtDNA
- heteroplasmy a state in which normal and mutant mtDNA are mixed
- the degree of heteroplasmy varies from tissue to tissue such as skeletal muscle, blood vessels, and skin, or from cell to cell.
- the degree of taurine modification of mitochondrial tRNA Leu is defined as follows, and subjects who have substantially normal mtDNA (healthy subjects) and subjects who contain mutant mtDNA at a certain ratio The taurine modification rate of tRNA Leu (UUR) is measured separately from (eg, MELAS patient, diabetic patient).
- the total taurine modification ratio in the present invention the taurine-modified in mitochondria tRNA Leu (UUR) in a specimen from a subject tRNA Leu (UUR), taurine-modified tRNA Leu with (UUR) and taurine unmodified tRNA Leu (UUR) The value divided by the sum.
- test subject is a mammal including a human
- mammals include rodents such as mice, rats, hamsters, guinea pigs, and laboratory animals such as rabbits, pigs, cows, goats, horses
- livestock such as sheep and mink
- pets such as dogs and cats
- primates such as humans, monkeys, cynomolgus monkeys, rhesus monkeys, marmosets, orangutans, chimpanzees, and gorillas.
- the subject-derived specimen is not particularly limited as long as it can be collected from a mammal, preferably a human, body fluid sample such as blood, lymph, urine, hair, buccal mucosa, stomach, large intestine, lung, Biopsy tissues such as liver and brain can be mentioned.
- a blood sample is preferred, and white blood cells in the blood sample are more preferred.
- Total taurine modification rate is determined indirectly by measuring the amount of taurine-modified tRNA Leu (UUR) and non-taurine-modified tRNA Leu (UUR ) by measuring the amount of primer extension product by primer-based reverse transcription (primer extension method).
- UUR taurine-modified tRNA Leu
- UUR non-taurine-modified tRNA Leu
- U is tRNA Leu with a taurine-modified (indicates the amount of primer extension product from UUR), U indicates the amount of primer extension product from no taurine-modified tRNA Leu (UUR)) The value calculated by substituting for.
- the mtDNA point mutation rate (%) is the 14th position A (A3243G), 15th position G (G3244A), 29th position in the DNA encoding tRNA Leu (UUR) represented by SEQ ID NO: 1.
- T (T3258C), 42 position T (T3271C) and 62 position T (T3291C) are used to determine the normal taurine modification rate. It is desirable to use as an index of aging.
- DNA sequencing method For the analysis of mtDNA point mutation rate, DNA sequencing method, PCR-RFLP method, PCR-SSCP method, denaturing high-performance liquid chromatography (DHPLC) method, BiPlex Invader method, SnaP shot method, high-resolution melt (HRM)
- TTGE temporal temperature gradient gel electrophoresis
- the total taurine modification rate is theoretically in the range of 0-100%. However, considering the pathology of MELAS patients, if the total taurine modification rate decreases, serious symptoms may develop. On the other hand, the normal taurine modification rate is almost the same as the total taurine modification rate when calculated using the formula [6] because the mtDNA point mutation rate is approximately 0% in healthy individuals. However, the normal taurine modification rate in patients with mitochondrial disease can be calculated to exceed 1OO% due to the mtDNA point mutation rate.
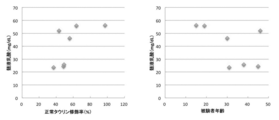
- the normal taurine modification rate of tRNA Leu (UUR) was plotted on the vertical axis and the subject age was plotted on the horizontal axis, and it was found that the normal taurine modification rate decreased as the subject age increased ( Figure 2). It was also revealed that oral taurine modification in MELAS patients recovered the normal taurine modification rate and improved the taurine modification of tRNA Leu (UUR) derived from mutant mtDNA (FIG. 7, Table 1). Administration of an effective amount of taurine can be expected to delay the progression of aging. It can be judged that aging is progressing, so that the normal taurine modification rate is low. That is, using the normal taurine modification rate, the scale of aging can be indicated as “taurine age”. Moreover, in healthy individuals, the total taurine modification rate and the normal taurine modification rate are close to each other. For convenience, the total taurine modification rate can be used to indicate the scale of aging as “taurine age”.
- Measurement method of aging A step of measuring the degree of taurine modification of mitochondrial tRNA Leu (UUR) in a biological sample isolated from a subject by reverse transcription reaction from a primer using the tRNA Leu (UUR) as a template
- biological samples include, but are not limited to, biopsy tissues such as hair, buccal mucosa, stomach, large intestine, lung, liver and brain, and body fluid samples such as blood, lymph and urine. Blood is preferable from the viewpoint of easy collection, and white blood cells obtained from blood are more preferable.
- a method for measuring aging will be described using white blood cells as an example, but other biological samples can be measured in the same manner.
- White blood cells to be used for measurement can be obtained by collecting blood from a subject and collecting the white blood cell fraction by a conventional method.
- Commercially available reagents can be suitably used for this purpose.
- Commercially available reagents include Lymphoprep (registered trademark), Ficoll (registered trademark) -Paque, and the like.
- the leukocyte fraction is obtained by performing density gradient centrifugation according to the instructions of the reagent manufacturer and separating the blood cell layer (peripheral blood mononuclear cells: PBMC) containing lymphocytes and monocytes.
- PBMC peripheral blood mononuclear cells
- the subject is typically a human, but may be a mammal other than a human.
- mammals other than humans include, for example, laboratory animals such as rodents such as mice, rats, hamsters, and guinea pigs, and rabbits, domestic animals such as pigs, cows, goats, horses, sheep and minks, and pets such as dogs and cats.
- Primates such as monkeys, cynomolgus monkeys, rhesus monkeys, marmosets, orangutans, chimpanzees and gorillas.
- RNA can be prepared from leukocytes by precipitating the gene using a method such as that described above.
- a commercially available extraction kit can be suitably used for this operation. For example, Isogen (made by Nippon Gene), Trizol ⁇ (made by Ambion) and the like can be mentioned.
- the obtained RNA can be subjected to a reverse transcription reaction without further purification.
- a primer that specifically hybridizes to the tRNA Leu (UUR) is used.
- the primer is an oligonucleotide complementary to the template.
- the oligonucleotide may be DNA, RNA, or a hybrid of DNA and RNA, but DNA is preferred from the viewpoint of operability.
- the nucleotide constituting the oligonucleotide may be a natural nucleotide or an artificial nucleotide.
- the length of the primer is not particularly limited as long as it can retain a function capable of specifically hybridizing to tRNA Leu (UUR) , but is 8 to 30 bases long, preferably 10 to 25 bases long, more preferably 10 to 22 bases long It is.
- the specific base sequence of the primer is designed so that the length of the primer extension product is different or the length of the primer extension product is different from the base sequence depending on the presence or absence of taurine modification of tRNA Leu (UUR) .
- a suitable base sequence is designed to hybridize in the 3 ′ to 5 ′ direction (from position 56 to position 41 ) of tRNA Leu (UUR) represented by SEQ ID NO: 2, which is shown below: It is DNA containing the base sequence represented by 5′-acctctgactgtaaag-3 ′ (SEQ ID NO: 3).
- the primer may have one or more (for example, two) bases added to the 5 ′ end and / or the 3 ′ end in the base sequence shown in SEQ ID NO: 3.
- one or more (eg, two) bases may be deleted from the 5 ′ end and / or the 3 ′ end.
- the tRNA Leu (UUR) to be measured has a mutated base sequence at the position where it hybridizes with the primer, so as to be complementary to the base sequence of the mutated tRNA Leu (UUR)
- Any base in the base sequence shown in SEQ ID NO: 3 may be substituted with another base.
- the primers designed in this way may be used in combination of not only one type but also two or more types.
- the primer may be directly or indirectly labeled with a labeling substance.
- labeling substance include fluorescent substances (eg, FITC, rhodamine, FAM TM , VIC TM , NED TM , PET TM , the above four are trade names of Applied Biosystems), radioactive materials (eg, 32 P, 35 S). , 14 C, 3 H), enzymes (eg, alkaline phosphatase, peroxidase), colored particles (eg, metal colloid particles, colored latex), biotin and the like.
- it is a fluorescent substance or a radioactive substance
- the site to be labeled is desirably the 5 ′ end of the primer.
- the reverse transcription reaction can be performed by a known method with reference to Molecular Cloning: A Laboratory Manual, Fourth Edition (2012).
- Reverse transcriptase using the above primers and dNTPs (nucleotide mix), the presence of RNase inhibitor necessary, tRNA in the obtained RNA Leu and (UUR) as a template, a reverse transcription reaction is performed.
- a reverse transcriptase is commercially available, and a commercially available enzyme kit can be used as long as it is an enzyme capable of performing a reverse transcription reaction using tRNA Leu (UUR) as a template.
- AMV reverse transcriptase, M-MuLV reverse transcriptase and the like can be mentioned, but mutant reverse transcriptases such as Superscript III (manufactured by Thermo Fisher Scientific Inc.) cannot be used because the reaction does not stop at the taurine modification site.
- As the template RNA about 1 pg to 1 ⁇ g of total RNA is used. About 0.1 to 1 ⁇ g of primer is used.
- dNTP a mixture of four types of nucleotides (eg, dATP, dGTP, dCTP, dTTP) may be used, or 1 to 3 types of nucleotides complementary to the base sequence of the template may be used.
- dNTP is usually used at a concentration of about 1 to 500 ⁇ M per nucleotide.
- the RNase inhibitor can be used without limitation as long as it can inhibit RNase under the conditions of reverse transcription reaction.
- Various RNase inhibitors are commercially available, and examples of commercially available products include RNaseOUT (manufactured by Thermo Fisher Scientific Inc.), RNasin (manufactured by Promega), and the like.
- the composition of the reverse transcription reaction solution can be appropriately set depending on the reverse transcriptase used.
- the reaction buffer attached to a commercially available enzyme kit can be diluted and used.
- the reverse transcription reaction is divided into a template and primer annealing reaction and a subsequent extension reaction from the primer.
- a condition of incubating at about 80 ° C. for 1 to 5 minutes and then allowing to stand at 37 ° C. or less, usually room temperature (around 25 ° C.) for 10 minutes to several hours can be mentioned.
- the primer extension reaction for example, the reaction is carried out at 42 ° C. to 60 ° C. for 15 minutes to 2 hours, and after completion of the reaction, the reverse transcriptase is inactivated by heat treatment at 95 ° C. for 1 to 5 minutes.
- the difference in the primer extension product due to the presence or absence of taurine modification is detected.
- a primer extension product is developed on an agarose gel or polyacrylamide gel by electrophoresis and then detected by a difference in size.
- the 5 ′ end of the primer is fluorescently or radiolabeled in advance, and after electrophoresis, a signal is taken into the imaging plate and can be detected using an appropriate detection device.
- tRNA Leu with a taurine-modified when carrying out reverse transcription reaction using primers comprising the nucleotide sequence represented by SEQ ID NO: 3, .tau.m 5 U modification acts as a barricade against elongation reaction As a result, the elongation reaction stops at the U position.
- the extension reaction proceeds without steric hindrance to the extension reaction, and dideoxynucleotides (eg, ddA, ddG, ddC, ddT) are allowed to coexist in the reaction solution.
- the extension reaction can be stopped at a desired position. For example, when ddG is used, as shown in FIG. 1, the difference between tRNA Leu (UUR) with taurine modification and tRNA Leu (UUR) without taurine modification is primer extension with different length of one base. Expressed as a product.
- the band may be identified by cutting out the band separated by electrophoresis, determining the base sequence by a conventional method, and measuring the difference in the base sequence of the primer extension product depending on the presence or absence of taurine modification.
- a primer extension product from the tRNA Leu (UUR) having a taurine-modified in step the step of calculating the quantitative ratio between the primer extension products from the no taurine-modified tRNA Leu (UUR), a primer extension product
- the signal captured on the imaging plate is quantified using densitometry such as BAS 2000 Image Analyzer (manufactured by GE Healthcare).
- primer extension product from tRNA Leu (UUR) with taurine modification can be expressed in arbitrary units, or expressed in quantitative ratios. Also good. These values are expressed by the following formula (1):
- U represents the amount of primer extension product from the tRNA Leu (UUR) with taurine modification
- U is indicative of the amount of primer extension product from no taurine-modified tRNA Leu (UUR)
- the mtDNA point mutation rate (%) is the 14th position A (A3243G), 15th position G (G3244A), 29th position in the DNA encoding tRNA Leu (UUR) represented by SEQ ID NO: 1.
- T (T3258C), 42 position T (T3271C) and 62 position T (T3291C) are used to determine the normal taurine modification rate. It is desirable to use as an index of aging.
- the point mutation rate of mtDNA is DNA sequencing method, PCR-RFLP method, PCR-SSCP method, denaturing high-performance liquid chromatography (DHPLC) method, BiPlex Invader method, SnaP shot method, high-resolution melt (HRM) profiling method,
- TTGE temporal temperature gradient gel electrophoresis
- the PCR product amplified using the PCR-RFLP method is used for the presence or absence of a restriction enzyme ApaI cleavage site (a PCR product with a mutation has a cleavage site, and a PCR product without a mutation has The difference in the length of the band obtained by (without a cleavage site) can be quantified, and the point mutation rate of A3243G can be measured.
- the PCR product amplified using the PCR-RFLP method has a cleavage site for the restriction enzyme AflII (a PCR product with a mutation has a cleavage site, and a PCR product without a mutation has a cleavage site)
- AflII a PCR product with a mutation has a cleavage site
- a PCR product without a mutation has a cleavage site
- the total taurine modification rate and the normal taurine modification rate thus obtained can be used as indicators of aging in the present invention.
- a healthy person can use the total taurine modification rate as an index of aging because the total taurine modification rate and the normal taurine modification rate are almost the same.
- the total taurine modification rate of 100% can be determined as young from the viewpoint of taurine age.
- the rate of taurine modification of normal mitochondrial tRNA Leu can be used as an indicator of aging and functional degradation of mitochondria itself, and it can be used as a living body, organ (organ), tissue, cell, or cell. It shows that it is useful as an indicator of aging and functional deterioration of organelles, especially organs, tissues, cells, or organelles with high energy demand.
- the taurine modification rate of normal mitochondrial tRNA Leu is a biomarker used in determining whether a subject is healthy or unaffected, determining whether a subject is in need of life improvement and treatment, and assisting them. Useful.
- the rate of taurine modification of normal mitochondrial tRNA Leu is As an innovative new biomarker, it has been shown to be useful for selecting companion diagnostics for assisting the selection and treatment of taurine therapy for patients with mitochondrial diseases currently under development .
- a primer comprising a base sequence consisting of 10 to 25 bases complementary to mitochondrial tRNA Leu (UUR) , which is included in the aging determination drug of the present invention, is the same as the primer described in "Method for measuring aging”. It is.
- the primer may be contained in a lyophilized state or may be contained in a solution with a pharmaceutically acceptable carrier.
- a pharmaceutically acceptable carrier when preparing the determination drug of the present invention as a liquid, various carriers conventionally used as a preparation material, for example, a diluent, a solvent, a solubilizing agent, a tonicity agent, a buffering agent, etc. May be included.
- the diluent include water and physiological saline.
- the solvent include water, physiological saline, ethanol and the like.
- solubilizers include cyclodextrins.
- the isotonic agent examples include inorganic salts such as sodium chloride and potassium chloride, and carbohydrates such as glycerin, mannitol and sorbitol.
- the buffer examples include phosphate buffer, acetate buffer, borate buffer, carbonate buffer, citrate buffer, Tris buffer, and the like. Those skilled in the art can appropriately determine the mixing ratio of these carriers. It is desirable to store the primer on ice or on ice until just before use.
- the aging determination kit of the present invention contains a primer containing a base sequence consisting of 10 to 25 bases complementary to mitochondrial tRNA Leu (UUR) and a reverse transcriptase.
- the primer containing a base sequence consisting of 10 to 25 bases complementary to mitochondrial tRNA Leu (UUR) which is included in the aging determination kit of the present invention, is the same as the primer described in “Method for measuring aging”.
- the kit of the present invention may further contain nucleoside triphosphate, a buffer for primer extension reaction, and an RNase inhibitor.
- Nucleoside triphosphates are substrates that are incorporated into primers to form primer extension products and are usually used as dNTP mixtures.
- a dNTP mixture typically consists of dATP, dTTP, dCTP and dGTP. If necessary, it may contain at least one dideoxynucleotide (ddATP, ddTTP, ddCTP, ddGTP) for stopping primer extension at a desired site.
- the buffer examples include tris buffer, phosphate buffer, veronal buffer, borate buffer, Good buffer and the like, which are used when performing a normal hybridization reaction.
- the pH is not particularly limited, but is usually preferably in the range of 5 to 9.
- the RNase inhibitor is the same as the RNase inhibitor described in “Measurement method for aging”.
- the kit of the present invention includes a reaction vessel, a buffer for diluting the primer, a positive control (eg, normal human-derived cybrid cell RNA: Proc Natl Acad Sci U S A. 2005; 102 ( 20): 7127-7132), negative control (eg, MELAS patient-derived cybrid cell RNA: Proc Natl Acad Sci U S A. 2005; 102 (20): 7127-7132), instructions describing the protocol, etc. Further, it may be included.
- a positive control eg, normal human-derived cybrid cell RNA: Proc Natl Acad Sci U S A. 2005; 102 ( 20): 7127-7132
- negative control eg, MELAS patient-derived cybrid cell RNA: Proc Natl Acad Sci U S A. 2005; 102 (20): 7127-7132
- instructions describing the protocol etc. Further, it may be included.
- taurine and it Contained composition is used to improve the rate of taurine modification of mitochondrial tRNA Leu (UUR) , to prevent a decrease in the rate of taurine modification of mitochondrial tRNA Leu (UUR) , especially due to aging of normal mitochondrial tRNA Leu (UUR) It is useful as a pharmaceutical, quasi-drug, cosmetic or food for improving the taurine modification rate.
- the improving agent of the present invention contains taurine as an active ingredient.
- Taurine is also referred to as aminoethylsulfonic acid or 2-aminoethanesulfonic acid.
- Taurine may be organically synthesized according to a conventional method, or may be a natural product extracted and isolated from animals and plants.
- Taurine may be in the form of a taurine derivative as long as it acts as taurine in vivo.
- taurine derivatives include N-methyltaurine, N, N-dimethyltaurine, N, N, N-trimethyltaurine, guanidinoethanesulfonic acid, guanidinoethanesulfinic acid, N- (2-acetamido) -2-aminoethanesulfonic acid Piperazino-N, N′-bis (2-ethanesulfonic acid), N- [1′-aza-cycloheptan-2′-yl] -2-aminoethanesulfonic acid, N- [1′-aza-cyclo Pentane-2′-yl] -2-aminoethanesulfonic acid, N- [1′-aza-cycloheptane-2′-yl] -3-aminopropanesulfonic acid, N- [1′-aza-cyclopentane-
- the administration of the improving agent of the present invention to a subject improves the taurine modification rate in the subject as compared to before administration or ingestion.
- the improved taurine modification rate is the total taurine modification rate represented by the above-described formula (1) and / or the normal taurine modification rate represented by the above-described formula (2).
- the improving agent of the present invention is taurine alone or an excipient (for example, lactose, sucrose, starch, cyclodextrin, etc.), and in some cases, a fragrance, a pigment, a seasoning, a stabilizer, a storage Containing tablets, pills, granules, fine granules, powders, pellets, capsules, solutions, emulsions, suspensions, syrups, troches, etc., and pharmaceuticals, quasi drugs, cosmetics or foods ( Preferably, it can be used as a health functional food) or food additive. Further, the improving agent of the present invention can also be used as a research reagent.
- an excipient for example, lactose, sucrose, starch, cyclodextrin, etc.
- a fragrance for example, lactose, sucrose, starch, cyclodextrin, etc.
- a pigment for example, lactose, sucrose, starch, cyclodextr
- the amount of taurine contained in the improving agent of the present invention is not particularly limited as long as the effect of the present invention is exhibited, but is usually 0.0001 to 100% by weight, preferably 0.001 to 99.0% by weight. %.
- the effective amount of taurine can be expressed as the amount of taurine per kg body weight per day.
- the recommended effective amount is 0.01 to 1.0 g / kg body weight / day, preferably 0.02 to 0.5 g / kg body weight / day.
- the improving agent of the present invention when used as a medicine, it preferably contains an effective amount of taurine and a pharmaceutically acceptable carrier.
- the pharmaceutically acceptable carrier examples include excipients (eg, lactose, sucrose, dextrin, hydroxypropylcellulose, polyvinylpyrrolidone, light anhydrous silicic acid, etc.), disintegrating agents (eg, starch, carboxymethylcellulose, etc.), Lubricant (eg, magnesium stearate, talc, etc.), surfactant (eg, polyoxyethylene hydrogenated castor oil, glyceryl monostearate, sodium lauryl sulfate, macrogol, sucrose fatty acid ester, etc.), solvent (eg, , Water, saline, soybean oil, etc.), preservatives (for example, p-hydroxybenzoic acid ester, etc.) and the like, but are not limited thereto.
- excipients eg, lactose, sucrose, dextrin, hydroxypropylcellulose, polyvinylpyrrolidone, light anhydrous silicic acid, etc.
- disintegrating agents
- the improver of the present invention can be safely administered orally or parenterally to animals (eg, mammals, birds, fish, etc.).
- animals eg, mammals, birds, fish, etc.
- the improver of the present invention can be taken as a food or as a food additive.
- Food in the present invention means all foods, but in addition to general foods including so-called health foods, health foods such as foods for specified health use and functional foods for nutrition specified in the health function food system of the Ministry of Health, Labor and Welfare. And may be foods with health claims or health claims, and supplements, feeds and feeds are also included in the foods of the present invention.
- taurine can be used by adding it to general foods (including so-called health foods) such as soft drinks, breads and confectionery.
- general foods including so-called health foods
- taurine is mixed with excipients (for example, lactose, sucrose, starch, etc.), and in some cases, flavors, pigments, etc., tablets, pills, granules, fine granules, powders, pellets, capsules, solutions, emulsions, It can be formulated into suspensions, syrups, lozenges, etc., and used as health functional foods and supplements such as foods for specified health use and nutritional functional foods.
- the food and food additive of the present invention can also be applied to feed and feed applications, and can be ingested or administered to poultry and livestock in addition to normal feed and feed.
- the intake or dose of the improving agent of the present invention varies depending on the age, weight and health condition of the intake or administration target, and cannot be determined in general.
- it is usually in the form of quasi-drugs such as drinks, foods or cosmetics, while treatment or health for disorders caused by a decreased rate of taurine modification
- taurine is 0.4-40 g per adult, preferably 0.8 g-20 g, more preferably 4.0. It is preferable to take or administer ⁇ 15 g in 1 to several times a day.
- the body weight is 40 kg or less, it is usually taken in an amount of 0.3 to 30 g, preferably 0.6 to 15 g, more preferably 3.0 to 12 g per day divided into once to several times a day. Administration is preferred.
- the administration method of the improving agent (medicine) of the present invention is not particularly limited as long as it provides a preventive and therapeutic effect against a decrease in the taurine modification rate.
- parenteral administration intravenous administration, intramuscular administration, direct administration within tissue, intranasal administration, intradermal administration, transdermal administration, intraperitoneal administration, gastrostomy, tube administration, enteral nutrition administration, etc.
- oral administration and in particular for application of the medicament to humans, it can be administered by intravenous, intramuscular or oral administration.
- the dosage form is not particularly limited, and various dosage forms such as oral preparations (eg, granules, powders, tablets, capsules, syrups, emulsions, suspensions, drinks), injections, It can be administered as an instillation or an external preparation (nasal preparation, transdermal preparation, ointment, etc.). External preparations (transdermal preparations, ointments, etc.) can also be administered as quasi drugs or cosmetics.
- oral preparations eg, granules, powders, tablets, capsules, syrups, emulsions, suspensions, drinks
- injections It can be administered as an instillation or an external preparation (nasal preparation, transdermal preparation, ointment, etc.).
- External preparations transdermal preparations, ointments, etc.
- quasi drugs or cosmetics can also be administered as quasi drugs or cosmetics.
- the agent for improving the rate of taurine modification of mitochondrial tRNA Leu (UUR) of the present invention is a patient who has developed mitochondrial disease, preferably a patient who has developed mitochondrial disease due to a mutation in a mitochondrial tRNA Leu (UUR) taurine modification.
- tRNA Leu (UUR) represented by SEQ ID NO: 1
- a at position 14 A3243G
- G at position 15 G3244A
- T at position 29 T3258C
- T at position 42 Patients with mitochondrial disease caused by any point mutation selected from the group consisting of T3271C) and T at position 62 (T3291C), particularly preferably encoding tRNA Leu (UUR) represented by SEQ ID NO: 1
- Factor Living and environmental factors mitochondrial tRNA Leu with a reduced due state of vital functions due to the effect of such may be used as improving agent of taurine modification rate of (UUR).
- the improving agent of the present invention reduces the rate of taurine modification of mitochondrial tRNA Leu (UUR) due to aging, the state of biological functions due to the influence of genetic factors, living and environmental factors, etc. in these subjects and patients It may be used as a preventive agent.
- improving agents of taurine-modified mitochondrial tRNA Leu (UUR) of the present invention not only the improvement of taurine modification rate of abnormal mitochondrial tRNA Leu (UUR), normal mitochondrial tRNA Leu taurine modification rate is decreased (UUR ) Is particularly useful in that it can improve the taurine modification rate.
- Example 1 Detection of Taurine Modification of Mitochondrial tRNA Leu (UUR) by Reverse Transcription Reaction Basically, the method was performed according to the method of Kirino et al. (Proc Natl Acad Sci US A. 2005; 102 (20): 7127-7132) It was.
- the principle of the primer extension method by Kirino et al. Is shown in FIG.
- the base sequences of the primers used are as follows. 5'-acctctgactgtaaag-3 '(SEQ ID NO: 3)
- the base sequences of the two expected primer extension products are as follows.
- dATP, dTTP, ddGTP (1.5 mM each, manufactured by AmershamPharmacia) was added in the reaction buffer for reverse transcriptase (manufactured by Invitrogen) (final concentration 37.5 ⁇ M), and 1 ⁇ L of M-MuLV Reverse transcriptase (RNase H ⁇ ) ( 40 units / ⁇ L, manufactured by Invitrogen), and a reverse transcription reaction was performed at 42 ° C. for 1 hour.
- the 32 P-labeled primer (mixed primer, 0.1 pmol) represented by SEQ ID NO: 3 and SEQ ID NO: 4 was used.
- the reaction mixture was subjected to 15% polyacrylamide electrophoresis containing 7M urea.
- the band labeled with RI was visualized with a BAS5000 bioimaging analyzer (Fuji Film).
- Anti-codon (first letter U: underlined in Fig. 1) lacks taurine modification ( 5 U 34 ) and reaction stops at AAG, and taurine modification exists ( ⁇ m 5 U 34 ) and reaction stops at AG .
- the RI-labeled reaction product was separated by electrophoresis and quantified with a BAS5000 bioimaging analyzer (Fuji Film) to determine the total taurine modification rate. The results are shown in FIG.
- T3271C mutation For: 5'-taagaagaggaattgaacctctgaccttaa-3 '(SEQ ID NO: 10) and Rev: 5′-aggacaagagaaataaggcc-3 ′ (SEQ ID NO: 11) was used for PCR, and the amplified PCR product was cleaved with the restriction enzyme AflII.
- AflII cleavage site there is no restriction enzyme AflII cleavage site, so there is only one 170 bp band
- AflII cleavage site there is an AflII cleavage site, so two fragments of 140 bp and 30 bp are cleaved. A band appears.
- the PCR product cleaved with the above restriction enzyme was subjected to 15% polyacrylamide electrophoresis, the separated band was stained with ethidium bromide, the intensity of the band was quantified using a densitometer, and the point mutation rate was determined ( Table 1).
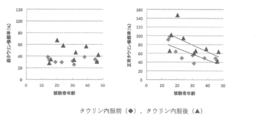
- the normal taurine modification rate of MELAS patients was determined using the total taurine modification rate and point mutation rate. The results are shown in FIG.
- Test Example 1 Measurement of blood and cerebrospinal fluid lactate and pyruvate levels
- the blood and cerebrospinal fluid lactate levels were measured by reacting L-lactic acid with lactate oxidase and converting the resulting hydrogen peroxide to peroxidase.
- the dye produced in the presence of was quantified by colorimetric measurement.
- the pyruvate value was measured by allowing pyruvate oxidase to act on pyruvate and colorimetrically measuring the resulting hydrogen peroxide in the presence of peroxidase.
- Table 1 shows the total taurine modification rate, the normal taurine modification rate, and the point mutation rate of individual MELAS patients after the start of taurine administration and after the end of administration.
- Example 1 and Test Example 2 indicate that the taurine modification rate of normal mitochondrial tRNA Leu (UUR) decreases with aging, and the decreased taurine modification rate increases by taking taurine. Furthermore, the fact that the taurine modification rate of normal tRNA Leu (UUR) exceeds 100% indicates that the taurine modification rate of mutant tRNA Leu (UUR) is also improved by taking taurine. Until now, it was thought that the cause of MELAS was due to the lack of taurine modification of mutant tRNA Leu (UUR) , but this result shows that the rate of taurine modification of normal tRNA Leu (UUR) associated with the onset of MELAS It is shown that the decline of is involved.
- Example 1 the taurine modification rate of normal mitochondrial tRNA Leu (UUR) can be used as an indicator of aging and functional deterioration of mitochondria itself.
- Organ tissue, cell, or organelle function, especially useful as an indicator of aging or functional degradation of organs, tissues, cells, or organelles with high energy demand. It shows that there is.
- the normal taurine modification is performed although the calendar age is relatively high as in the case of the subject age 14 in Test Example 2 and the case where the normal taurine modification rate is low compared to the calendar age and the subject age 46
- the rate of normal taurine modification is simply an aging that accompanies aging of the subject because there is a considerable range in the rate of normal taurine modification in young healthy subjects in Test Example 4 described later.
- an indicator of aging but also as an indicator of aging reflecting the health status of subjects including the presence of diseases such as nutritional status and metabolic abnormalities, or as an indicator of healthy life expectancy, especially healthy life expectancy that does not include a specific cause of death Indicates that it can be used.
- composition is for improvement of taurine modification rate of mitochondrial tRNA Leu (UUR), for the prevention of lowering taurine modification rate of mitochondrial tRNA Leu (UUR), among others, decreased by aging of normal mitochondrial tRNA Leu (UUR) This indicates that it is useful as a pharmaceutical, quasi-drug, cosmetic or food for improving the taurine modification rate.
- Test example 3 Improvement of diabetes marker by administration of taurine to diabetic patients
- improvement of disease state was observed by administration of taurine.
- the therapeutic effect of taurine administration on diabetes mellitus caused by base mutation (A3243G) was confirmed.
- taurine (trade name Taurinsan 98% Taisho, Taisho Pharmaceutical Co., Ltd., manufactured by Taisho Pharmaceutical Co., Ltd.) was diagnosed as diabetes due to a single nucleotide mutation (A3243G) in the mitochondrial gene and was also associated with cardiomyopathy. ) was orally administered daily.
- HbA1c hemoglobin A1c
- MELAS reduces the rate of normal mitochondrial tRNA Leu (UUR) taurine modification due to aging and the effects of genetic factors, living and environmental factors, etc. Since it is not limited and is a universal phenomenon even in healthy individuals, the mechanism of diabetes onset by mitochondrial gene mutation is the same as in MELAS, in addition to abnormal mitochondrial tRNA Leu ( in addition to UUR , normal mitochondrial tRNA Leu presumably due to a decrease in taurine modification rate of (UUR).
- UUR normal mitochondrial tRNA Leu
- Test Example 4 Measurement of normal mitochondrial tRNA Leu (UUR) taurine modification rate in healthy subjects
- the normal mitochondrial tRNA Leu (UUR) taurine modification rate decreases with age
- the rate of taurine modification of normal mitochondrial tRNA Leu (UUR) decreased with aging even in healthy subjects. The result is shown in FIG.
- the normal mitochondrial tRNA Leu (UUR) modification rate is Compared with the average of younger (10s) 56.7 ⁇ 12.6% (Mean ⁇ SE), the elderly (80s) showed a significant decrease with aging, 35.8 ⁇ 3.0%.
- a modification rate is about 80% to about 40%, which is about twice as wide. Therefore, a decrease in the normal mitochondrial tRNA Leu (UUR) taurine modification rate is This is thought to reflect not only aging but also the decline in biological functions due to genetic factors, lifestyle and environmental factors. This result shows that the taurine modification rate of normal mitochondrial tRNA Leu (UUR) can be used as a useful aging biomarker not only in MELAS patients but also in healthy individuals.
- the taurine modification rate of normal mitochondrial tRNA Leu is universally aging regardless of mitochondrial disease patients and healthy individuals due to the effects of aging, genetic factors, living and environmental factors, etc. It shows that it decreases with it. Moreover, since it was confirmed that the decrease in the taurine modification rate was improved by administration of taurine, the taurine modification-improving agent of the mitochondrial tRNA Leu (UUR) of the present invention is suitable for not only healthy subjects but also unaffected subjects and some diseases.
- the taurine modification rate of normal mitochondrial tRNA Leu (UUR) is a biomarker that can be used to select the target of taurine therapy for patients with mitochondrial disease that is currently under development, as well as for companion diagnostic applications used to determine the therapeutic effect It is also useful for assistance.
- Formulation Example 1 Granule (in one package) Taurine 3000mg Aspartame 9mg Talc 20mg Low-substituted hydroxypropylcellulose 60mg A granule having the above formulation was prepared by a conventional method.
- Formulation Example 2 Liquid for internal use (in 1 drug) Keihi 100mg Royal Jelly 100mg Taurine 5000mg 70% sorbitol solution 1000mg Erythritol 1000mg pH adjuster Add an appropriate amount of purified water to make the total volume 180mL. By an ordinary method, a liquid preparation for internal use comprising the above formulation was prepared.
- Formulation Example 3 Drink (in 1) Vitamin B1 nitrate 10mg Vitamin B2 2mg Carnitine chloride 50mg Taurine 4000mg Sodium pantothenate 10mg Benzoic acid 20mg Butyl paraoxybenzoate 2.5mg Aspartame 6mg Citric acid 100mg Glycine 300mg Add purified water to bring the total volume to 50 mL. A drink comprising the above formulation was prepared by a conventional method.
- the present inventors have found that the taurine modification rate of normal tRNA Leu (UUR) among mitochondrial tRNAs is an indicator of aging.
- the rate of taurine modification of normal tRNA Leu (UUR) is a universal marker that can be used to understand the status of healthy individuals as well as patients with mitochondrial diseases.
- Taurine is a pharmaceutical, quasi-drug, and cosmetic product that improves the rate of taurine modification that has decreased due to the aging of mitochondrial tRNA Leu (UUR) and the effects of life and environmental factors such as strength fatigue and genetic factors. Alternatively, it is useful as a food.
- the present invention is based on Japanese Patent Application No. 2016-062910 (filing date: March 26, 2016) filed in Japan, and all the contents thereof are included in this specification.
Abstract
Description
1)ミトコンドリアtRNAを含むtRNAの化学修飾、具体的にはリジンをコードするtRNAのチオメチル修飾率を測定することで2型糖尿病を診断する方法が知られている(特許文献1)。
2)高齢化に伴う代謝の変化、特にミトコンドリアの機能不全に結びつく変化を予防または修復することによって高齢動物の健康状態を改善する方法が知られている(特許文献2)。グルタチオンはミトコンドリアDNA(mtDNA)の酸化的傷害を防止する主要な細胞内抗酸化システムであるが、高齢化に伴いグルタチオン自体の酸化が進んで抗酸化機能が低下する。これに対して、タウリンなどのチオール化合物を供給することでmtDNAの酸化的傷害を防止できることが記載されている。しかし、tRNALeu(UUR)のタウリン修飾率と老化との関係については記載がない。
3)タウリン、タウリンクロラミンまたはタウリン前駆物質等を有効成分とするミトコンドリア病の治療薬が知られている(特許文献3)。しかし、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率が加齢や、遺伝的要因や生活・環境要因などの影響による老化(生体機能の低下)に伴い低下すること、および、その低下がタウリン投与により改善されることは開示も示唆もされていない。
本発明の第二の目的は、ミコトンドリアの機能低下を惹起する、老化に伴う、ミトコンドリアtRNALeu(UUR)のタウリン修飾率低下の改善剤を提供することにある。
〔1〕 被験者から単離した生体試料におけるミトコンドリアtRNALeu(UUR)のタウリン修飾の程度を、該tRNALeu(UUR)を鋳型としてプライマーからの逆転写反応により測定する工程を含む、被験者の老化の測定方法。
〔2〕 プライマーが鋳型に相補的な10~25塩基長からなるオリゴヌクレオチドであり、タウリン修飾の有無によるプライマー伸長産物の相違を検出する、〔1〕に記載の方法。
〔3〕 プライマーが配列番号3、4および5で表される塩基配列からなる群より選ばれる塩基配列を含む、少なくとも1種である、〔1〕または〔2〕に記載の方法。
〔4〕 タウリン修飾を有するtRNALeu(UUR)からのプライマー伸長産物と、タウリン修飾を有しないtRNALeu(UUR)からのプライマー伸長産物との量比を算出する工程をさらに含む、〔1〕~〔3〕のいずれかに記載の方法。
〔5〕 下記式(1):
〔6〕 被験者が、tRNALeu(UUR)遺伝子コード領域のA3243G、T3271C、G3244A、T3258CおよびT3291Cからなる群より選ばれる一塩基置換を有する変異ミトコンドリアDNAを有すると疑われる者であり、前記式(1)に続いて、下記式(2):
〔7〕 ミトコンドリアtRNALeu(UUR)に相補的な10~25塩基からなる塩基配列を含むプライマーを含有する、老化の判定薬。
〔8〕 プライマーが配列番号3、4および5で表される塩基配列からなる群より選ばれる塩基配列を含む、少なくとも1種である、〔7〕に記載の判定薬。
〔9〕 ミトコンドリアtRNALeu(UUR)に相補的な10~25塩基からなる塩基配列を含むプライマーおよび逆転写酵素を含有する、老化の判定キット。
〔10〕 プライマーが配列番号3、4および5で表される塩基配列からなる群より選ばれる塩基配列を含む、少なくとも1種である、〔9〕に記載のキット。
〔11〕 タウリンを有効成分として含有する、ミトコンドリアtRNALeu(UUR)のタウリン修飾率の改善剤。
〔12〕 ミトコンドリアtRNALeu(UUR)が正常なミトコンドリアtRNALeu(UUR)である、〔11〕に記載の改善剤。
〔13〕 タウリン修飾率の改善が下記式(1):
下記式(2):
〔14〕 医薬または化粧品である、〔11〕または〔12〕に記載の改善剤。
〔15〕 保健機能食品または食品添加物である、〔11〕または〔12〕に記載の改善剤。
〔16〕 ミトコンドリア病を発症した患者、および/または発症リスクを有する対象のミトコンドリアtRNALeu(UUR)のタウリン修飾率の改善用である、〔11〕~〔15〕のいずれかに記載の改善剤。
〔17〕 ミトコンドリア病がミトコンドリアtRNALeu(UUR)のタウリン修飾の欠損遺伝子変異に起因する、〔16〕に記載の改善剤。
〔18〕 ミトコンドリアtRNALeu(UUR)のタウリン修飾の欠損遺伝子変異が、配列番号1で表されるtRNALeu(UUR)をコードするDNAにおいて、14位のA(A3243G)、15位のG(G3244A)、29位のT(T3258C)、42位のT(T3271C)および62位のT(T3291C)からなる群より選ばれる点変異である、〔17〕に記載の改善剤。
〔19〕 ミトコンドリア病が、MELASあるいは糖尿病である、〔16〕~〔18〕のいずれかに記載の改善剤。
本発明において「老化」とは、加齢や、遺伝的要因や生活・環境要因などのその他の内的または外的な要因の影響により生体機能が低下することをいう。加齢とは、生まれてから死までの物理的な時間経過であり、暦年齢と同義である。その他の内的な要因とは、例えば、ミトコンドリア機能、過酸化ラジカル濃度、およびテロメア長などの細胞老化に係わるものなどがあげられる。また、その他の外的な要因とは、例えば、個々の運動量、喫煙の有無、食事習慣、および栄養状態などがあげられる。生体機能とは、例えば筋力、神経伝導速度、肺活量、病気に対する抵抗力などがあげられる。加齢による老化は、一般に生殖年齢に達した後に始まり、個体差があるが、ヒトでは20歳から30歳以降に始まる。老化は、環境要因や遺伝的素因が複雑に絡み合って関与していると考えられ、かかる機能低下の速さはすべてのヒトが同じではなく、必ずしも加齢という要因のみで規定できるものではなく、従来は明確な老化の指標がなかった。本発明が提供する老化の指標は、健康寿命の観点からも指標とすることができる。
本発明の「老化」は、身長や体重の減少、皮膚や頭髪の変化などの外的変化、運動機能の低下、感覚機能の低下、或いは、生理機能の低下などの所謂加齢に伴う身体機能の低下のみでなく、生体を構成する細胞の老化(細胞老化)、さらに呼吸・エネルギー代謝に重要な機能を果たすミトコンドリアの老化が含まれる。老化に伴いミトコンドリアtRNALeu(UUR)のタウリン修飾率が低下すれば、当然、UUGコドン特異的な翻訳能が律速となって、UUGコドンを含むタンパク質の合成が低下する。ひいてはミトコンドリアの機能が低下し、生体やそれを構成する器官等の機能が低下するのは明らかであり、さらに修飾率の低下が進行すれば、例えば、高齢者に認められる呼吸困難や骨格筋の減弱のような重篤な症状を発症するに至る場合もあると考えられる。従って、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率は、ミトコンドリア自体の老化や機能低下の指標として用いることができることは言うに及ばず、生体、器官(臓器)、組織、細胞、或は、細胞内小器官の機能、とりわけ、生体やエネルギー需要の高い器官(臓器)、組織、細胞、或いは、細胞内小器官の老化や機能低下の指標としても有用である。
また、上記指標は、現在開発が進行中のミトコンドリア病患者に対するタウリン療法の対象者の選定や、その治療効果判定に用いるコンパニオン診断用途やその補助にも有用である。
本願明細書においては、「ミトコンドリアtRNALeu(UUR)」を単に「tRNALeu(UUR)」と省略する場合がある。
本発明において変異tRNALeu(UUR)とは、タウリン修飾の欠損を生じさせる変異を有するmtDNAより転写されたtRNALeu(UUR)をいい、タウリン修飾に影響しない変異を有するtRNALeu(UUR)を含まないものとする。
また、正常tRNALeu(UUR)とは、上記変異tRNALeu(UUR)を除くtRNALeu(UUR)をいう。ヒトの場合、配列番号2で表されるtRNALeu(UUR)と同一の塩基配列を有するtRNALeu(UUR)および配列番号2で表されるtRNALeu(UUR)と異なる塩基配列を有するtRNALeu(UUR)であっても、該塩基配列の相違がtRNALeu(UUR)のタウリン修飾に影響しないtRNALeu(UUR)が含まれるが、タウリン修飾の有無にかかわらず正常なtRNALeu(UUR)の機能を有さないものは含まない。
ここで、上記14位のA、15位のG、29位のT、42位のTおよび62位のTは、ヒト以外に、ゴリラ、ウシ、ブタ、イヌ、ネコ等においても保存されており、ラットおよびマウスの場合も、42位がCである以外は同様に保存されているため、tRNALeu(UUR)のタウリン修飾の欠損を生じさせる変異は、ヒトに限定されるものではなく、後述する霊長類、ペット、家畜、実験動物等の哺乳動物も対象となりうる。
細胞あたり数百のミトコンドリアが存在し、そのひとつひとつのミトコンドリアに5~10個のミトコンドリアDNA(mtDNA)が含まれている。単一のmtDNAで構成されている状態を「ホモプラスミー」、正常と変異mtDNAが混在している状態を「ヘテロプラスミー」と呼ぶ。変異mtDNAの比率(ヘテロプラスミーの度合い)が高くなると、ミトコンドリアの呼吸機能に異常を来たし、ミトコンドリア病を発症する。ヘテロプラスミーの度合いは、骨格筋、血管、皮膚などの組織ごと、または細胞ごとに異なる。また、健常者の組織または細胞においても、変異mtDNAが微量に含まれていることが最近報告された(Brendan A.I. Payne et al.: Universal heteroplasmy of human mitochondrial DNA. Hum Mol Gent 22: 384 - 390 (2013))。そこで、本発明においては、ミトコンドリアtRNALeu(UUR)のタウリン修飾の度合いを以下のように定義し、実質的に正常なmtDNAを有する被験者(健常者)と、変異mtDNAを一定の割合で含む被験者(例、MELAS患者、糖尿病患者)とに区別して、tRNALeu(UUR)のタウリン修飾率を測定する。
MELAS患者の細胞のミトコンドリアは、ヘテロプラスミーであるため、変異mtDNAと正常なmtDNAが含まれている。したがって、変異mtDNAより転写されたtRNALeu(UUR)のアンチコドン1番目のウラシルはタウリン修飾が欠損しているとしても、正常なmtDNAより転写されたtRNALeu(UUR)はタウリン修飾されていると考えられる。
MELAS患者を始めとする変異mtDNAを含む被験者由来の細胞に含まれる正常なmtDNAより転写されたtRNALeu(UUR)のタウリン修飾率を「正常タウリン修飾率」と定義し、下記式(1)により求めた「総タウリン修飾率」に基づいて、下記式(2)により求めることができる。
医師主導治験において、tRNALeu(UUR)の正常タウリン修飾率を縦軸に、被験者年齢を横軸にプロットしたところ、被験者年齢が高くなるに従って正常タウリン修飾率が減少していることを発見した(図2)。また、MELAS患者のタウリン内服により、正常タウリン修飾率が回復し、変異mtDNAに由来するtRNALeu(UUR)のタウリン修飾も改善することが明らかとなった(図7、表1)。有効量のタウリン投与により、老化の進行を遅延させることが期待できる。
正常タウリン修飾率が低いほど、老化が進行していると判断することができる。すなわち、正常タウリン修飾率を用いて、老化の尺度を「タウリン年齢」として示すことができる。また、健常者においては、総タウリン修飾率と正常タウリン修飾率とが近似しており、便宜的に総タウリン修飾率を用いて、老化の尺度を「タウリン年齢」として示すことができる。
(1)被験者から単離した生体試料におけるミトコンドリアtRNALeu(UUR)のタウリン修飾の程度を、該tRNALeu(UUR)を鋳型としてプライマーからの逆転写反応により測定する工程
測定対象の生体試料としては、毛髪、頬粘膜、胃、大腸、肺、肝臓、脳などの生検組織や、血液、リンパ液、尿などの体液試料が限定なくあげられる。採取が容易な点から血液が好ましく、血液より得られる白血球がより好ましい。以下、白血球を例にして、老化の測定方法を説明するが、他の生体試料も同様に測定することができる。
5’-acctctgactgtaaag-3’(配列番号3)で表される塩基配列を含むDNAである。
プライマーは、本発明の目的の範囲内で、配列番号3に示される塩基配列において、5’末端および/または3’末端に1または複数個(例えば、2個)の塩基が付加されていてもよく、5’末端および/または3’末端から1または複数個(例えば、2個)の塩基が欠失してもよい。
さらに、プライマーとハイブリダイズする位置に、測定対象のtRNALeu(UUR)が変異した塩基配列を有することが想定される場合は、変異tRNALeu(UUR)の塩基配列と相補的となるように、配列番号3に示される塩基配列の任意の塩基を他の塩基に置換してもよい。具体的には、下記:
5’-acctctgactgtaagg-3’(配列番号4):3271変異の場合、または
5’-acctctgactgcaaag-3’(配列番号5):3280変異の場合
で表される塩基配列を含むDNAがあげられる。このようにして設計したプライマーは、1種のみならず、2種以上を組み合わせて使用してもよい。
逆転写酵素は市販されており、tRNALeu(UUR)を鋳型として、逆転写反応を行える酵素であれば、市販の酵素キットを使用することができる。例えば、AMV逆転写酵素、M-MuLV逆転写酵素などがあげられるが、SuperscriptIII(Thermo Fisher Scientific Inc. 製)のような変異型の逆転写酵素はタウリン修飾部位で反応が停止しないため使用できない。
鋳型RNAは、総RNAとして1pg~1μg程度を用いる。
プライマーは、0.1~1μg程度を用いる。
dNTPは、4種のヌクレオチド(例、dATP、dGTP、dCTP、dTTP)の混合物を用いてもよく、鋳型の塩基配列に相補的な1~3種のヌクレオチドを用いてもよい。dNTPは、通常、各ヌクレオチドあたり1-500μM程度の濃度で使用する。
RNase阻害剤は、逆転写反応の条件下でRNaseを阻害できる限り限定なく使用することができる。種々のRNase阻害剤が市販されており、市販品としては、RNaseOUT(Thermo Fisher Scientific Inc. 製)、RNasin(Promega製)等があげられる。
逆転写反応溶液の組成は、用いる逆転写酵素により適宜設定することができる。また、市販の酵素キットに添付されている反応緩衝液を希釈して使用することができる。
アニーリング反応の一例として、例えば、約80℃で1~5分間インキュベートした後、37℃以下、通常は室温(25℃前後)にて10分~数時間静置する条件があげられる。
プライマー伸長反応の一例として、例えば、42℃~60℃で15分間~2時間行い、反応終了後は95℃で1~5分間熱処理することにより逆転写酵素を失活させる。
前記工程で、プライマー伸長産物を電気泳動後、イメージングプレートに取り込んだシグナルを、BAS 2000イメージアナライザー (GEヘルスケア製)等のデンシトメトリーを用いて定量する。
例えば、A3243Gを有するMELAS患者の場合、PCR-RFLP法を用いて増幅したPCR産物を、制限酵素ApaIの切断部位の有無(変異のあるPCR産物は切断部位を有し、変異のないPCR産物は切断部位を有しない)により得られるバンドの長さの相違を定量し、A3243Gの点変異率を測定することができる。T3271Cを有するMELAS患者の場合、PCR-RFLP法を用いて増幅したPCR産物を、制限酵素AflIIの切断部位の有無(変異のあるPCR産物は切断部位を有し、変異のないPCR産物は切断部位を有しない)により得られるバンドの長さの相違を定量し、T3271Cの点変異率を測定することができる。
総タウリン修飾率100%は、タウリン年齢の観点からは若年と判断することができる。
加えて、稀に、健常者、MELAS患者のいずれにおいても暦年齢と相関しない正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率の例が認められることから、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率は、単に、被験者の加齢に伴う老化の指標となるだけでなく、栄養状態や代謝異常など疾患の有無なども含めた被験者の健康状態を反映した身体機能全般の老化の指標、或いは、健康寿命、とりわけ、特定死因を含まない健康寿命の指標としても利用できることを示している。
また、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率は、被験者が健常か未病の状態かの判定や被験者の生活改善や治療の要否の判定、それらの補助方法において用いるバイオマーカーとしても有用である。
また、MELAS患者のみでなく、ミトコンドリアtRNALeu(UUR)の一塩基置換(A3243G)を有する糖尿病患者でもタウリン投与の有効性が確認されており、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率が、革新的な新規バイオマーカーとして、現在開発が進行中のこれらミトコンドリア病患者に対するタウリン療法の対象者の選定やその治療効果判定に用いるコンパニオン診断用途やその補助にも有用であることを示している。
本発明の老化の判定薬に含まれる、ミトコンドリアtRNALeu(UUR)に相補的な10~25塩基からなる塩基配列を含むプライマーは、「老化の測定方法」に記載のプライマーと同様である。
医薬上許容されうる担体としては、本発明の判定薬を液剤として調製する場合、製剤素材として慣用されている各種担体、例えば希釈剤、溶剤、溶解補助剤、等張化剤、緩衝剤などを含んでいてもよい。
希釈剤としては、水、生理用食塩水などがあげられる。
溶剤としては、水、生理用食塩水、エタノールなどがあげられる。
溶解補助剤としては、シクロデキストリン類などがあげられる。
等張化剤としては、塩化ナトリウム、塩化カリウムなどの無機塩類、グリセリン、マンニトール、ソルビトールなどの炭水化物などがあげられる。
緩衝剤としては、リン酸緩衝液、酢酸緩衝液、ホウ酸緩衝液、炭酸緩衝液、クエン酸緩衝液、トリス緩衝液などがあげられる。
これらの担体の配合比は、当業者が適宜決定することができる。
プライマーは、使用直前まで凍結または氷上で保管しておくことが望ましい。
本発明の老化の判定キットは、ミトコンドリアtRNALeu(UUR)に相補的な10~25塩基からなる塩基配列を含むプライマーおよび逆転写酵素を含有する。
必要に応じて、プライマー伸長を所望の部位で停止させるためのジデオキシヌクレオチド(ddATP、ddTTP、ddCTP、ddGTP)を少なくとも1種含んでいてもよい。
加齢による正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率の低下がタウリンの投与により改善できることから、試験例2の結果は、タウリンおよびそれを含有する組成物が、ミトコンドリアtRNALeu(UUR)のタウリン修飾率の改善用、ミトコンドリアtRNALeu(UUR)のタウリン修飾率低下の予防用、とりわけ、正常なミトコンドリアtRNALeu(UUR)の加齢により低下したタウリン修飾率の改善用の医薬品、医薬部外品、化粧品、或いは、食品として有用であることを示している。
本発明の改善剤は、タウリンを有効成分として含有する。タウリンは、アミノエチルスルホン酸または2-アミノエタンスルホン酸ともいう。タウリンは、常法に従って有機合成されたものであってもよく、動植物から抽出単離された天然物であってもよい。
治験に参加したMELAS患者から書面によるインフォームドコンセントを得て、患者血液を採取した。採取した血液から、Isogen(ニッポンジーン製)を用いて、製造業者の指示書に従って粗RNAを得た。
患者血液の取り扱いについては、川崎医科大学での倫理規定にも従った。
基本的には、桐野らの方法(Proc Natl Acad Sci U S A. 2005; 102(20): 7127-7132)に準じて行った。桐野らによるプライマー伸長法の原理を図1に示す。
使用したプライマーの塩基配列は、以下の通りである。
5’-acctctgactgtaaag-3’(配列番号3)
予想される2種のプライマー伸長産物の塩基配列は、以下の通りである。
5’-acctctgactgtaaagTTTTAAG-3’(配列番号6:プライマーから伸長した配列を大文字で示す)
5’-acctctgactgtaaagTTTTAG-3’(配列番号7:プライマーから伸長した配列を大文字で示す)
上記プライマーおよびプライマー伸長産物の5’末端は、本実施例においては32Pで標識されている。
被験者白血球から得たRNA(0.2~1μg)を鋳型として、tRNALeu(UUR)のアンチコドンの3'側に相当する32P 標識プライマー(配列番号3、0.1pmol)と80℃で2分間インキュベートした後、室温で1時間静置することにより、プライマーをアニーリングさせた。その後、逆転写酵素用反応緩衝液(Invitrogen製)中、dATP、dTTP、ddGTP(各1.5mM、AmershamPharmacia製)を添加し(最終濃度37.5μM)、M-MuLV Reverse transcriptase(RNase H-)1μL(40 units/μL、Invitrogen製)を混合し、42℃で1時間逆転写反応を行なった。T3271C変異を有するMELAS患者の場合は、配列番号3および配列番号4で示される32P 標識プライマー(混合プライマー、0.1pmol)を用いた。
反応終了後、反応混合物を、7M尿素を含む15%ポリアクリルアミド電気泳動に供した。RIで標識されたバンドを、BAS5000バイオイメージングアナライザー(富士フィルム製)で可視化した。
For: 5’-gcccttcccccgtaaatgatat-3’(配列番号8)および
Rev: 5’-gaagaggaattgaacctctgactg-3’(配列番号9)の2本のプライマーでPCRを行い、増幅したPCR産物を制限酵素のApaIで切断した。正常mtDNAでは、制限酵素ApaIの切断部位がないため136 bpのバンドが1本であるのに対し、A3243G変異では、ApaIの切断部位があるため、切断されて84 bp、52 bpの2本のバンドが現れる。T3271C変異については、
For: 5’-taagaagaggaattgaacctctgaccttaa-3’ (配列番号10)および
Rev: 5’-aggacaagagaaataaggcc-3’(配列番号11)の2本のプライマーでPCRを行い、増幅したPCR産物を制限酵素のAflIIで切断した。正常mtDNAでは、制限酵素AflIIの切断部位がないため170 bpのバンドが1本であるのに対し、T3271C変異では、AflIIの切断部位があるため、切断されて140 bp、30 bpの2本のバンドが現れる。
上記制限酵素で切断したPCR産物を、15%ポリアクリルアミド電気泳動に供し、分離したバンドを臭化エチジウムで染色し、デンシトメーターを用いてバンドの強度を定量し、点変異率を求めた(表1)。
血液中および髄液中の乳酸値の測定は、L-乳酸に乳酸オキシダーゼを作用させ、生じた過酸化水素を、パーオキシダーゼの存在下で生じる色素を比色測定することにより定量した。
ピルビン酸値の測定は、ピルビン酸にピルビン酸オキシダーゼを作用させ、生じた過酸化水素を、パーオキシダーゼの存在下で生じる色素を比色測定することにより定量した。
治験参加MELAS患者の血中白血球におけるミトコンドリアtRNALeu(UUR)のタウリン修飾率を測定し、被験者の年齢を横軸にプロットしてグラフ化した。その結果、総タウリン修飾率と年齢とは相関がなかったのに対し、正常タウリン修飾率は、年齢依存性に低下した(図2)。一方、正常タウリン修飾率は、従来のミトコンドリア病マーカーとされる、血中および髄液乳酸値、乳酸/ピルビン酸比とは相関しなかったし、被験者の年齢と従来のミトコンドリア病マーカーとも相関しなかった(図3-6)。
上記医師主導治験において、MELAS患者にタウリンを投与した。投与量は、体重40kg以上の被験者の場合12g/日、25kg以上40kg未満の場合9g/日で、投薬期間は52週(1年間)であった。タウリン投与終了日(52週)に被験者から血液を採取し、実施例1に記載の方法に従って総タウリン修飾率および正常タウリン修飾率を測定した。結果を実施例1で求めた投与開始日(開始前0週)のデータと併せて図7に示す。また、個々のMELAS患者の、タウリン投与開始日および投与終了後の総タウリン修飾率、正常タウリン修飾率および点変異率をまとめて表1に示す。総タウリン修飾率をタウリン投与前と投与後とで比較した結果、有意差傾向があった(P=0.0546)。タウリン内服により、正常タウリン修飾率が回復し、変異mtDNAに由来するtRNALeu(UUR)のタウリン修飾も改善することが明らかとなった。
これまで、MELASの発症原因は変異tRNALeu(UUR)のタウリン修飾の欠損にあると考えられてきたが、この結果は、MELASの発症に加齢に伴う正常tRNALeu(UUR)のタウリン修飾率の低下が関与していることを示している。
周知のようにMELAS患者においてtRNALeu(UUR)のタウリン修飾機能自体に欠損はない。
実施例1で認められた現象は、MELAS患者の解析結果ではあるが、健常者でも同様に発生する現象を反映していると言える。つまりtRNALeu(UUR)のタウリン修飾率はミトコンドリアの老化のバイオマーカーとして用いることができることを示している。
また、老化に伴いミトコンドリアtRNALeu(UUR)のタウリン修飾率が低下すれば、当然、UUGコドン特異的な翻訳能が律速と成って、UUGコドンを含むタンパク質全般の合成が低下する。それに伴い、呼吸・エネルギー代謝に重要な機能を果たすミトコンドリアの機能が低下し、生体やそれを構成する器官等の機能が低下するのは明らかであり、さらに修飾率の低下が進行すれば、例えば、高齢者に認められる呼吸困難や骨格筋の減弱のような重篤な症状を発症するに至る場合もあると考えられる。
従って、実施例1および試験例の結果は、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率は、ミトコンドリア自体の老化や機能低下の指標として用いることができることは言うに及ばず、生体、器官(臓器)、組織、細胞、或いは、細胞内小器官の機能、とりわけ、生体やエネルギー需要の高い器官(臓器)、組織、細胞、或いは、細胞内小器官の老化や機能低下の指標としても有用であることを示している。加えて、試験例2の被験者年齢14の患者のように暦年齢に比して正常タウリン修飾率の低い例や被験者年齢46の患者のように暦年齢が比較的高いにも関わらず正常タウリン修飾率が比較的高い例が認められることや、後述の試験例4の若年健常者の正常タウリン修飾率にかなりの幅があることから、正常タウリン修飾率は、単に、被験者の加齢に伴う老化の指標となるだけでなく、栄養状態や代謝異常など疾患の有無なども含めた被験者の健康状態を反映した老化の指標、或いは、健康寿命、とりわけ、特定死因を含まない健康寿命の指標としても利用できることを示している。
さらに、加齢などによる老化に伴う正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率の低下がタウリンの投与により改善できることから、試験例2および後述の試験例3の結果は、タウリンおよびそれを含有する組成物が、ミトコンドリアtRNALeu(UUR)のタウリン修飾率の改善用、ミトコンドリアtRNALeu(UUR)のタウリン修飾率低下の予防用、とりわけ、正常なミトコンドリアtRNALeu(UUR)の加齢により低下したタウリン修飾率の改善用の医薬品、医薬部外品、化粧品、或いは、食品として有用であることを示している。
試験例2では、ミトコンドリア遺伝子の一塩基変異(A3243G)によるMELAS患者において、タウリン投与により病状の改善が認められたので、同じミトコンドリア遺伝子の一塩基変異(A3243G)に起因する糖尿病に対するタウリン投与の治療効果を確認した。具体的には、ミトコンドリア遺伝子の一塩基変異(A3243G)による糖尿病と診断され、心筋症を併発した患者(49歳 女性)に、タウリン(商品名 タウリン散98%「大正」、大正製薬株式会社製)を、毎日経口投与した。投与は、タウリンとして4g/回、3回/日(12g/日)で、投薬期間は約1年間であった。当該患者より、タウリン投与開始から終了まで経時的に血液を採取し、糖尿病マーカーであるヘモグロビンA1c(HbA1c)の値を測定したところ、開始時には7.7%あった値が、投与開始23週目(約5カ月目)には6.9%に低下し、投与終了時には6.8%になった。上記結果が示すように、当該患者のHbA1cの値を、タウリン投与期間中を通じて、糖尿病の合併症を予防するための目標値である7%未満にコントロールした。さらに、タウリン投与開始時には12単位必要であったインスリン投与を、投与終了時には9単位にまで低減することができ、タウリン内服により、ミトコンドリア遺伝子変異による糖尿病患者の症状改善が出来ることを確認した。
後述の試験例4から明らかなように、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率の加齢や、遺伝的要因や生活・環境要因などの影響による生体機能の状態による低下は、MELASに限定されるものではなく健常者においても普遍的な現象であるので、ミトコンドリア遺伝子変異による糖尿病発症のメカニズムは、MELASの場合と同様に、異常ミトコンドリアtRNALeu(UURに加えて、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率の低下によるものと考えられる。従って、当該糖尿病の場合でも、タウリンの投与による異常ミトコンドリアtRNALeu(UUR)のタウリン修飾率の改善に加えて、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率が改善したことにより、臨床症状が改善したものと考えることができる。
試験例2において、MELAS患者で確認された正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率が加齢に伴い低下することが確認されたので、健常者でも同様に、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率が加齢に伴い低下することを、試験例2と同様の方法により確認した。その結果を図8に示す。実施例1に記載の方法に従った、若年(10代)および老年(80代)健常人各々3名の末梢血白血球検体を用いた解析では、正常なミトコンドリアtRNALeu(UUR)修飾率は、若年者(10代)平均56.7±12.6% (Mean±SE)と比較して、高齢者(80代)では35.8±3.0%と、加齢による有意な低下が認められた。また、若年者間で比較した場合、かかる修飾率は約80%~約40%と約2倍程度の幅が認められることから、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率の低下は、加齢のみでなく、遺伝的要因や生活・環境要因などの影響による生体機能の低下をも反映していると考えられる。この結果は、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率は、MELAS患者のみでなく健常者においても、有用な老化のバイオマーカーとして用いることができることを示している。
また、前記タウリン修飾率の低下が、タウリン投与により改善することが確認されたため、本発明のミトコンドリアtRNALeu(UUR)のタウリン修飾改善剤は、健常者はもとより、未病の対象や何らかの疾患を発症するリスクを有する対象、さらには、何らかの疾患を有する患者、とりわけ、ミトコンドリア病患者やその発症リスクを有する対象における、老化に伴うミトコンドリアtRNALeu(UUR)のタウリン修飾率低下、特に、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率低下の改善や予防に有用である。
さらに、正常なミトコンドリアtRNALeu(UUR)のタウリン修飾率は、バイオマーカーとして、現在開発が進行中のミトコンドリア病患者に対するタウリン療法の対象者の選定や、その治療効果判定に用いるコンパニオン診断用途やその補助にも有用である。
タウリン 3000mg
アスパルテーム 9mg
タルク 20mg
低置換ヒドロキシプロピルセルロース 60mg
常法により、上記処方からなる顆粒剤を調製した。
ケイヒ 100mg
ローヤルゼリー 100mg
タウリン 5000mg
70%ソルビトール液 1000mg
エリスリトール 1000mg
pH調整剤 適量
精製水を加えて全量を180mLとする。
常法により、上記処方からなる内服用の液剤を調製した。
ビタミンB1硝酸塩 10mg
ビタミンB2 2mg
塩化カルニチン 50mg
タウリン 4000mg
パントテン酸ナトリウム 10mg
安息香酸 20mg
パラオキシ安息香酸ブチル 2.5mg
アスパルテーム 6mg
クエン酸 100mg
グリシン 300mg
精製水を加え全量を50mLとする。
常法により、上記処方からなるドリンク剤を調製した。
Claims (19)
- 被験者から単離した生体試料におけるミトコンドリアtRNALeu(UUR)のタウリン修飾の程度を、該tRNALeu(UUR)を鋳型としてプライマーからの逆転写反応により測定する工程を含む、被験者の老化の測定方法。
- プライマーが鋳型に相補的な10~25塩基長からなるオリゴヌクレオチドであり、タウリン修飾の有無によるプライマー伸長産物の相違を検出する、請求項1に記載の方法。
- プライマーが配列番号3、4および5で表される塩基配列からなる群より選ばれる塩基配列を含む、少なくとも1種である、請求項1または2に記載の方法。
- タウリン修飾を有するtRNALeu(UUR)からのプライマー伸長産物と、タウリン修飾を有しないtRNALeu(UUR)からのプライマー伸長産物との量比を算出する工程をさらに含む、請求項1~3のいずれか1項に記載の方法。
- 被験者が、tRNALeu(UUR)遺伝子コード領域のA3243G、T3271C、G3244A、T3258CおよびT3291Cからなる群より選ばれる一塩基置換を有する変異ミトコンドリアDNAを有すると疑われる者であり、前記式(1)に続いて、下記式(2):
- ミトコンドリアtRNALeu(UUR)に相補的な10~25塩基からなる塩基配列を含むプライマーを含有する、老化の判定薬。
- プライマーが配列番号3、4および5で表される塩基配列からなる群より選ばれる塩基配列を含む、少なくとも1種である、請求項7に記載の判定薬。
- ミトコンドリアtRNALeu(UUR)に相補的な10~25塩基からなる塩基配列を含むプライマーおよび逆転写酵素を含有する、老化の判定キット。
- プライマーが配列番号3、4および5で表される塩基配列からなる群より選ばれる塩基配列を含む、少なくとも1種である、請求項9に記載のキット。
- タウリンを有効成分として含有する、ミトコンドリアtRNALeu(UUR)のタウリン修飾率の改善剤。
- ミトコンドリアtRNALeu(UUR)が正常なミトコンドリアtRNALeu(UUR)である、請求項11に記載の改善剤。
- タウリン修飾率の改善が下記式(1):
下記式(2):
- 医薬または化粧品である、請求項11または12に記載の改善剤。
- 保健機能食品または食品添加物である、請求項11または12に記載の改善剤。
- ミトコンドリア病を発症した患者、および/または発症リスクを有する対象のミトコンドリアtRNALeu(UUR)のタウリン修飾率の改善用である、請求項11~15のいずれか1項に記載の改善剤。
- ミトコンドリア病がミトコンドリアtRNALeu(UUR)のタウリン修飾の欠損遺伝子変異に起因する、請求項16に記載の改善剤。
- ミトコンドリアtRNALeu(UUR)のタウリン修飾の欠損遺伝子変異が、配列番号1で表されるtRNALeu(UUR)をコードするDNAにおいて、14位のA(A3243G)、15位のG(G3244A)、29位のT(T3258C)、42位のT(T3271C)および62位のT(T3291C)からなる群より選ばれる点変異である、請求項17に記載の改善剤。
- ミトコンドリア病が、MELASあるいは糖尿病である、請求項16~18のいずれか1項に記載の改善剤。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018509283A JP6966784B2 (ja) | 2016-03-26 | 2017-03-25 | 老化を反映するミトコンドリアバイオマーカー |
| EP17774837.3A EP3434783A4 (en) | 2016-03-26 | 2017-03-25 | BIOMARKER MITOCHONDRIAL REFLECTING AGING |
| US16/088,596 US20190112656A1 (en) | 2016-03-26 | 2017-03-25 | Mitochondrial biomarker reflecting aging |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016062910 | 2016-03-26 | ||
| JP2016-062910 | 2016-03-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017170300A1 true WO2017170300A1 (ja) | 2017-10-05 |
Family
ID=59964418
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/012205 WO2017170300A1 (ja) | 2016-03-26 | 2017-03-25 | 老化を反映するミトコンドリアバイオマーカー |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20190112656A1 (ja) |
| EP (1) | EP3434783A4 (ja) |
| JP (1) | JP6966784B2 (ja) |
| WO (1) | WO2017170300A1 (ja) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003048829A (ja) | 2001-08-02 | 2003-02-21 | Taisho Pharmaceut Co Ltd | ミトコンドリア病の予防又は治療薬 |
| JP2004519241A (ja) | 2001-03-09 | 2004-07-02 | ソシエテ デ プロデユイ ネツスル ソシエテ アノニム | 高齢化に伴う生理的障害を改善し寿命を延ばす組成物 |
| WO2014136870A1 (ja) | 2013-03-08 | 2014-09-12 | 国立大学法人熊本大学 | Rna修飾の簡易検出法、及び該検出法を用いた2型糖尿病の検査方法 |
| JP2016062910A (ja) | 2014-09-12 | 2016-04-25 | 株式会社東芝 | 半導体装置 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20150361450A1 (en) * | 2014-06-13 | 2015-12-17 | Universite De Strasbourg | Vector for therapy of mitochondrial disease |
-
2017
- 2017-03-25 US US16/088,596 patent/US20190112656A1/en not_active Abandoned
- 2017-03-25 WO PCT/JP2017/012205 patent/WO2017170300A1/ja active Application Filing
- 2017-03-25 EP EP17774837.3A patent/EP3434783A4/en not_active Withdrawn
- 2017-03-25 JP JP2018509283A patent/JP6966784B2/ja active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004519241A (ja) | 2001-03-09 | 2004-07-02 | ソシエテ デ プロデユイ ネツスル ソシエテ アノニム | 高齢化に伴う生理的障害を改善し寿命を延ばす組成物 |
| JP2003048829A (ja) | 2001-08-02 | 2003-02-21 | Taisho Pharmaceut Co Ltd | ミトコンドリア病の予防又は治療薬 |
| WO2014136870A1 (ja) | 2013-03-08 | 2014-09-12 | 国立大学法人熊本大学 | Rna修飾の簡易検出法、及び該検出法を用いた2型糖尿病の検査方法 |
| JP2016062910A (ja) | 2014-09-12 | 2016-04-25 | 株式会社東芝 | 半導体装置 |
Non-Patent Citations (20)
| Title |
|---|
| "Biological Nomenclature", EUR. J. BIOCHEM., vol. 138, 1984, pages 9 |
| "GenBank", Database accession no. AB026838 |
| "Molecular Cloning: A Laboratory Manual", 2012 |
| ADRIAN GABRIEL TORRES; EDUARD BATLLE; LLUIS RIBAS DE POULANA, TRENDS IN MOLECULAR MEDICINE, vol. 20, no. 6, 2014, pages 306 - 314 |
| BRENDAN A.I. PAYNE ET AL.: "Universal heteroplasmy of human mitochondrial DNA", HUM MOL GENT, vol. 22, 2013, pages 384 - 390 |
| GOTO Y. ET AL.: "A new mtDNA mutation associated with mitochondrial myopathy, encephalopathy, lactic acidosis and stroke-like episodes (MELAS", BIOCHIMICA ET BIOPHYSICA ACTA, vol. 1097, 1991, pages 238 - 240, XP023554735, DOI: doi:10.1016/0925-4439(91)90042-8 |
| KIRINO ET AL., PROC NATL ACAD SCI U S A., vol. 102, no. 20, 2005, pages 7127 - 7132 |
| KIRINO Y. ET AL.: "Specific correlation between the wobble modification deficiency in mutant tRNAs and the clinical features of a human mitochondrial disease", PNAS, vol. 102, no. 20, 17 May 2005 (2005-05-17), pages 7127 - 7132, XP055280643, ISSN: 1091-6490 * |
| KIRINO Y; GOTO Y; CAMPOS Y; ARENAS J; SUZUKIT, PROC NATL ACAD SCI U S A., vol. 102, no. 20, 2005, pages 7127 - 7132 |
| KRAUT JA; MADIAS NE: "Lactic acidosis", N ENGL J MED, vol. 372, no. 11, 2015, pages 1078 - 1079 |
| PROC NATL ACAD SCI U S A., vol. 102, no. 20, 2005, pages 7127 - 7132 |
| RIKIMARU M. ET AL.: "Taurine Ameliorates Impaired the Mitochondrial Function and Prevents Stroke-like Episodes in Patients with MELAS", INTERNAL MEDICINE, vol. 51, 2012, pages 3351 - 3357, XP055556731, ISSN: 0918-2918 * |
| RIKIMARU M; OHSAWA Y; WOLF AM; NISHIMAKI K; ICHIMIYA H; KAMIMURA N; NISHIMATSU S; OHTA S; SUNADA Y, INTERN MED, vol. 51, no. 24, 2012, pages 3351 - 3357 |
| See also references of EP3434783A4 |
| SHIGEO OTA ET AL.: "Mitochondrial tRNA diseases: Defect of modification in anticodon in mutant tRNA molecules", PROTEIN, NUCLEIC ACID AND ENZYME, vol. 48, no. 4, 2003, pages 493 - 500, XP009512591, ISSN: 0039-9450 * |
| SHIGEO OTA: "Mitochondria Ijosho no Chiryo Senryaku", JAPANESE JOURNAL FOR INHERITED METABOLIC DISEASES, vol. 21, no. 1, 2005, pages 52 - 61, XP9516012, ISSN: 0912-0122 * |
| SUZUKI T. ET AL.: "Taurine as a constituent of mitochondrial tRNAs: new insights into the functions of taurine and human mitochondrial diseases", THE EMBO JOURNAL, vol. 21, no. 23, 2002, pages 6581 - 6589, XP055556741, ISSN: 2460-2075 * |
| TSUTOMU SUZUKI; ASUTEKA NAGAO; TAKEO SUZUKI, WIRES RNA, vol. 2, 2011, pages 376 - 386 |
| YASUKAWA T; SUZUKI T; UEDA T; OHTA S; WATANABE K, J BIOL CHEM., vol. 275, no. 6, 2000, pages 4251 - 4257 |
| YOSHIHIDE SUNADA ET AL.: "Taurine supplemental therapy for MELAS", JAPANESE JOURNAL OF TAURIN RESEARCH, vol. 1, no. 1, 2015, pages 30 - 32, XP009514949, ISSN: 2189-6232 * |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3434783A1 (en) | 2019-01-30 |
| EP3434783A4 (en) | 2019-11-06 |
| JPWO2017170300A1 (ja) | 2019-02-07 |
| JP6966784B2 (ja) | 2021-11-17 |
| US20190112656A1 (en) | 2019-04-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Reichold et al. | Glycine amidinotransferase (GATM), renal Fanconi syndrome, and kidney failure | |
| Manganelli et al. | Glucose-6-phosphate dehydrogenase deficiency: disadvantages and possible benefits | |
| JP2018023392A (ja) | 肝線維症に関連する遺伝的多型、その検出方法および使用 | |
| Rivera et al. | Marked mitochondrial DNA depletion associated with a novel SUCLG1 gene mutation resulting in lethal neonatal acidosis, multi-organ failure, and interrupted aortic arch | |
| EP3124034B1 (en) | Imidazole dipeptide for treating dementia due to aging or cerebral atrophy | |
| JP2009521905A (ja) | 冠動脈心疾患に関連する遺伝的多型、その検出方法および使用 | |
| Gesing et al. | Expression of key regulators of mitochondrial biogenesis in growth hormone receptor knockout (GHRKO) mice is enhanced but is not further improved by other potential life-extending interventions | |
| AU2014248949A1 (en) | Assays and methods for selecting a treatment regimen for a subject with depression | |
| KR20190138874A (ko) | 높은 안정성을 가진 망가니즈 수퍼옥시드 디스뮤타제의 용도 | |
| Andersen et al. | Effect of dietary copper deficiency on iron metabolism in the pregnant rat | |
| Priyadarshini et al. | Gestational Insulin Resistance Is Mediated by the Gut Microbiome–Indoleamine 2, 3-Dioxygenase Axis | |
| Kahre | Cystic fibrosis in Estonia | |
| WO2017170300A1 (ja) | 老化を反映するミトコンドリアバイオマーカー | |
| WO2006117945A1 (en) | Method for detecting lipid metabolism disorder, and diagnostic agent for use therein | |
| JP2009192383A (ja) | エルゴチオネインを利用したクローン病の診断および治療 | |
| Sokol et al. | Mitochondrial hepatopathies | |
| EP3466446B1 (en) | Pharmaceutical composition or food composition, and method for assessing effect of active ingredient in vivo | |
| Ng et al. | Two adult galactosaemia females with normal ovarian function and identical GALT mutations (Q188R/R333G) | |
| Yew et al. | ACAD10 is not required for metformin's metabolic actions or for maintenance of whole‐body metabolism in C57BL/6J mice | |
| González et al. | Intestinal inflammation marker calprotectin regulates epithelial intestinal zinc metabolism and proliferation in mouse jejunal organoids | |
| Lee et al. | eIF2B Activator Rescues Neonatal Lethality of an eIF2Bα Sugar Phosphate Binding Mutation Associated with Vanishing White Matter Disease | |
| PADELKAR | COMPARATIVE MOLECULAR INTERACTION OF THE OPIOID RECEPTOR (MOR) WITH THE BOVINE A1 & A2 TYPE β-CASEIN VARIANTS AND ASSOCIATED HEALTH (GASTROINTESTINAL) EFFECTS | |
| US20170037471A1 (en) | Methods for diagnosing & treating copper-dependent diseases | |
| US7354712B2 (en) | Estrogen receptor alleles that are predictive of increased susceptibility to bone fracture | |
| Ribeiro et al. | INTRASTRIATAL QUINOLINIC ACID ADMINISTRATION INHIBITS CRITICAL ENZYME ACTIVITIES OF ENERGY METABOLISM IN RAT STRIATUM |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2018509283 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2017774837 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2017774837 Country of ref document: EP Effective date: 20181026 |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17774837 Country of ref document: EP Kind code of ref document: A1 |