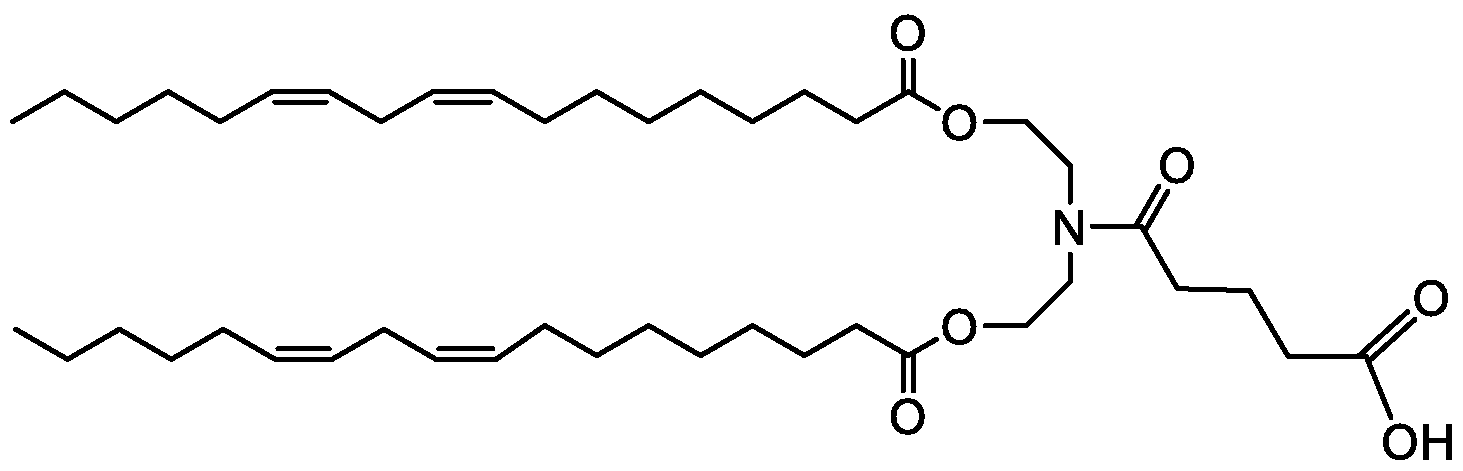
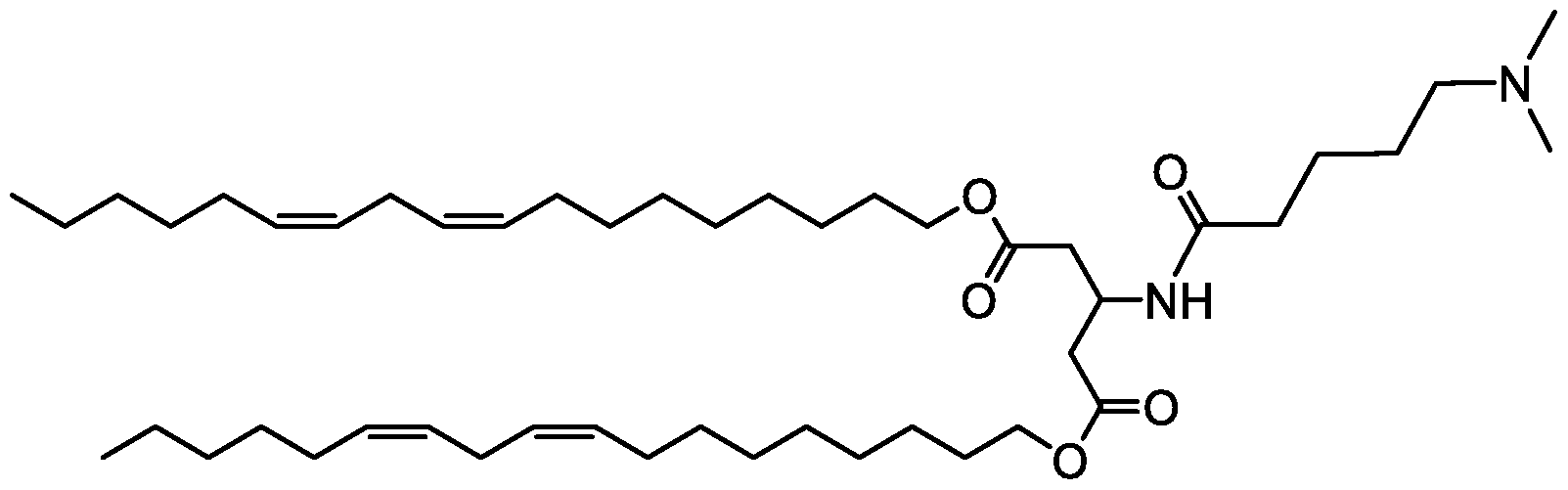
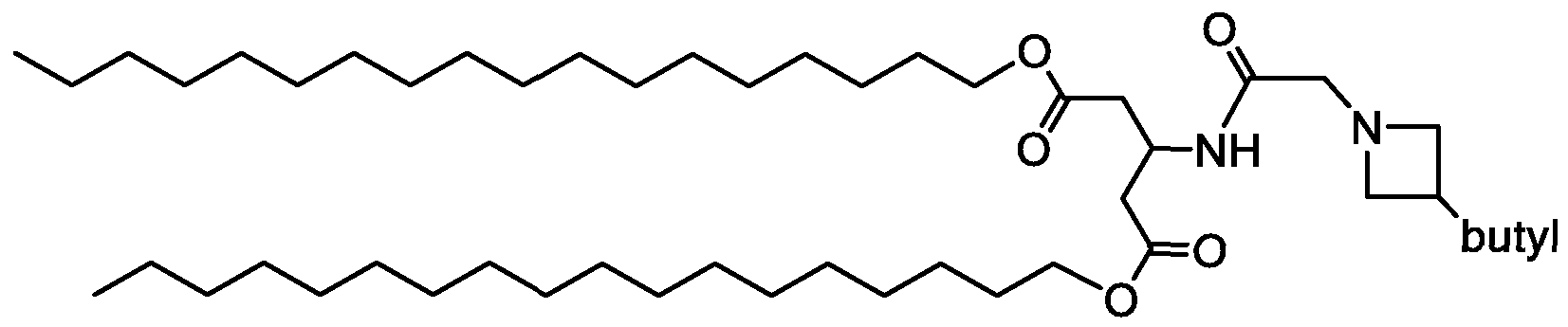
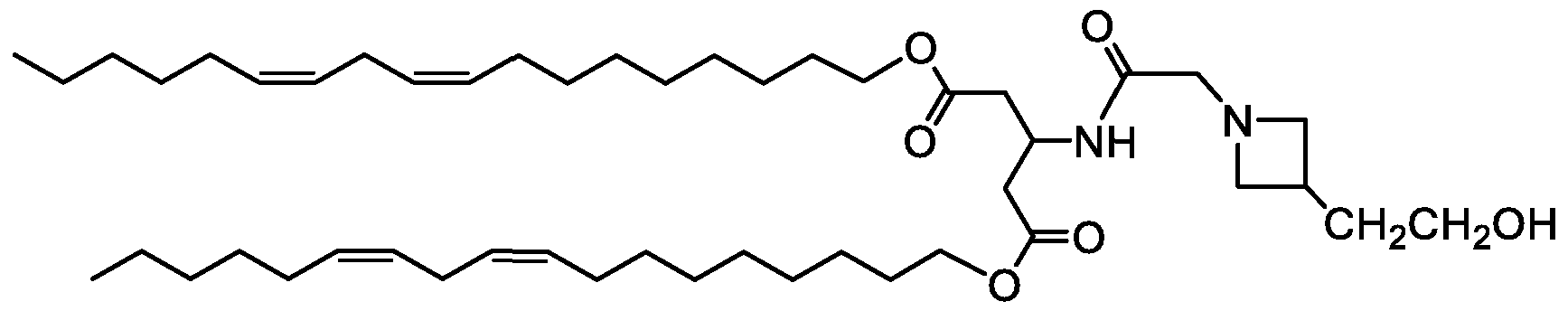
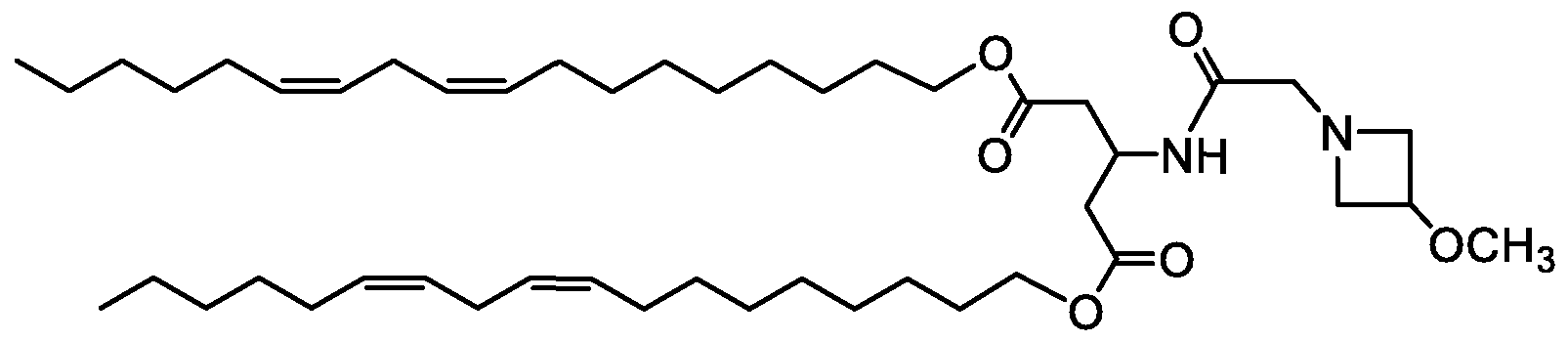
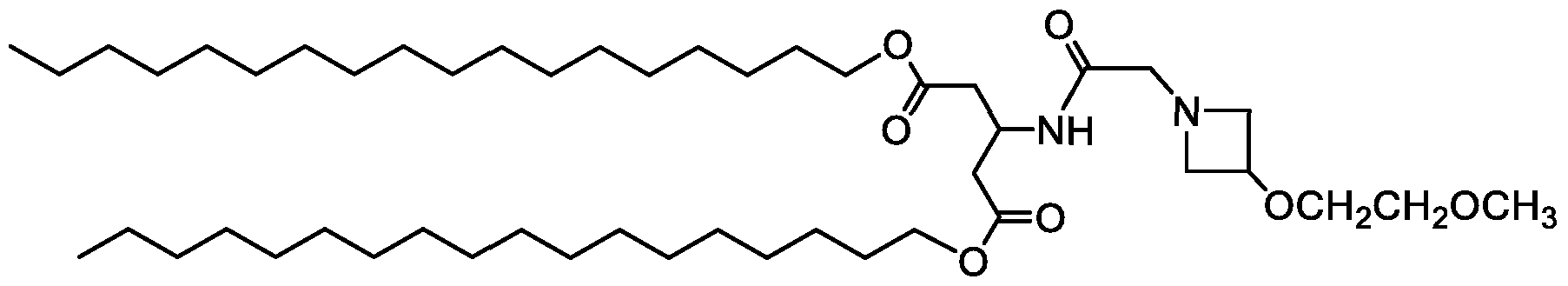
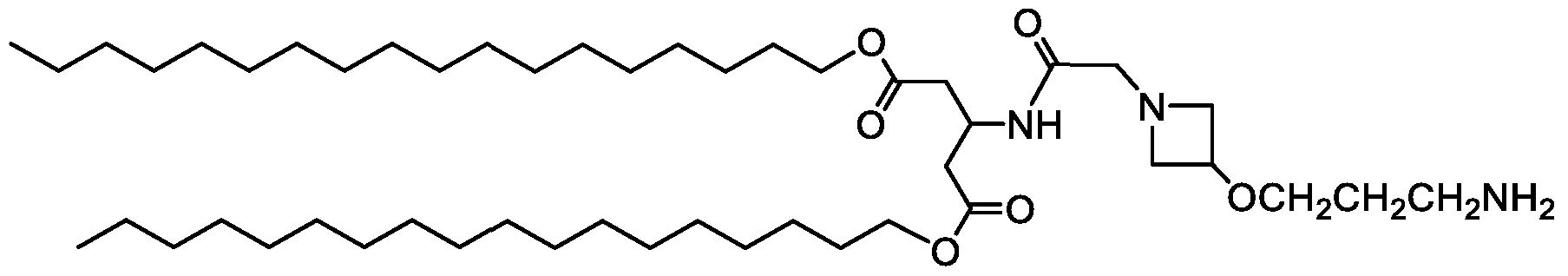
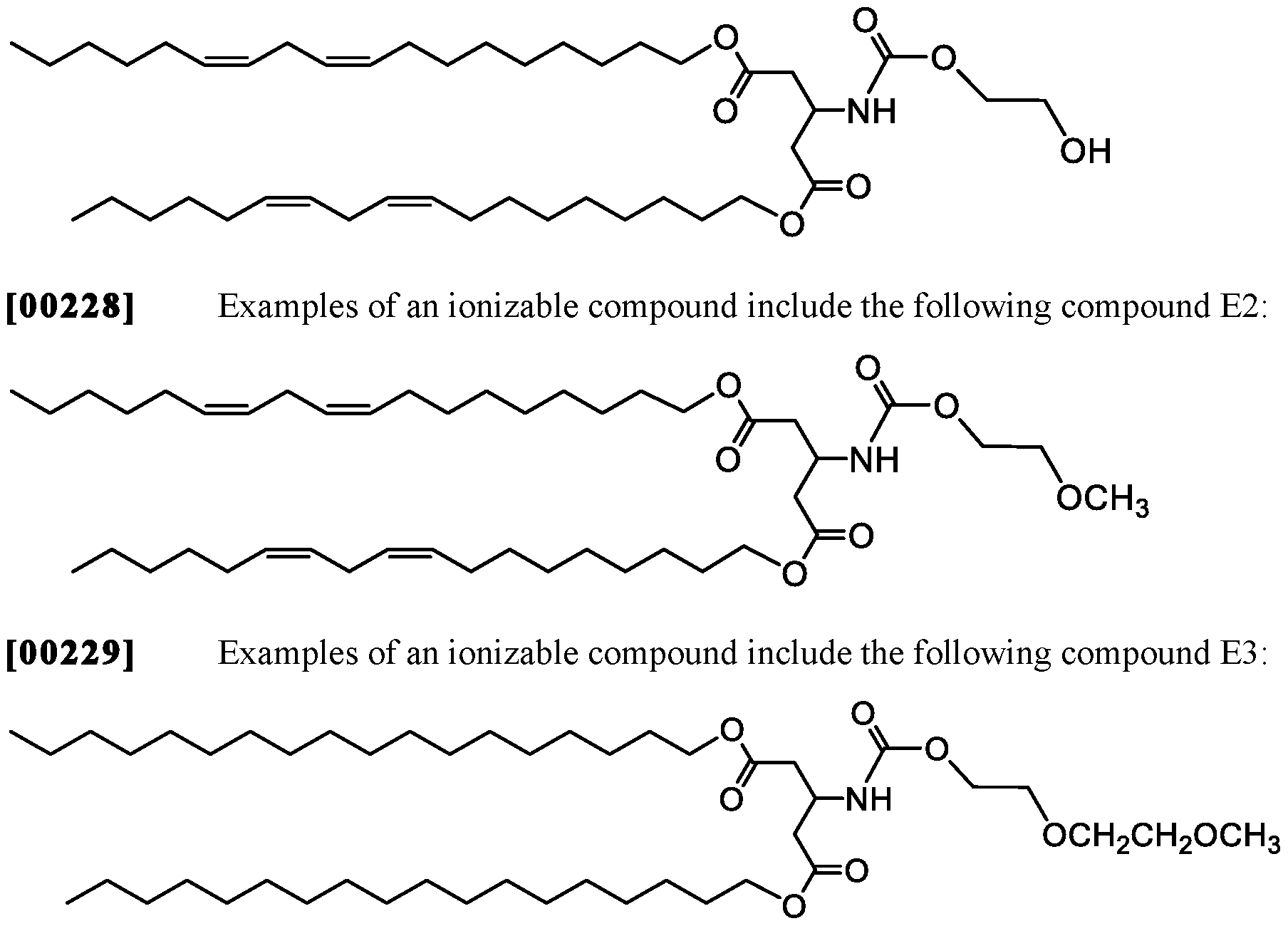
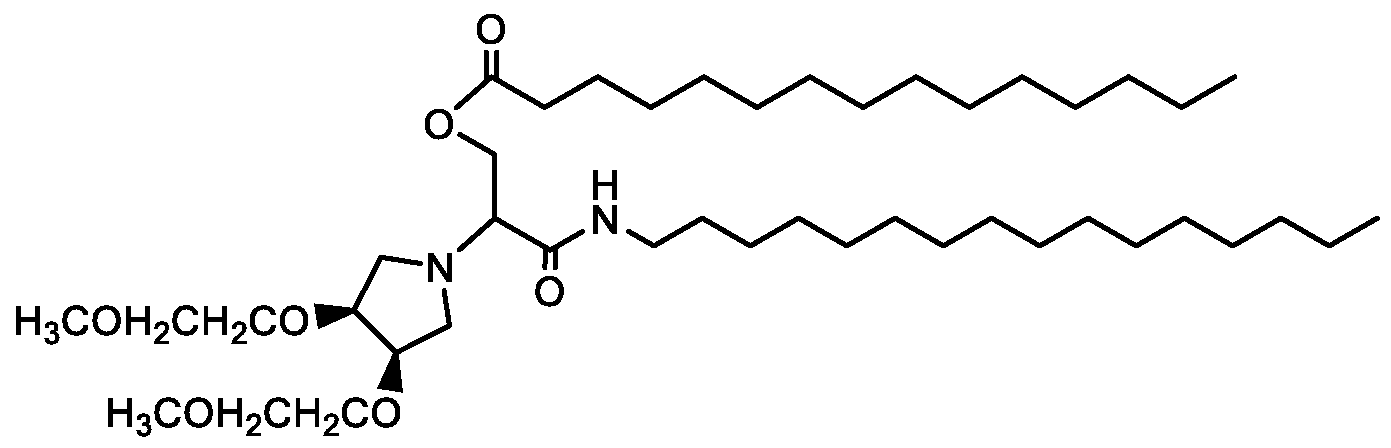
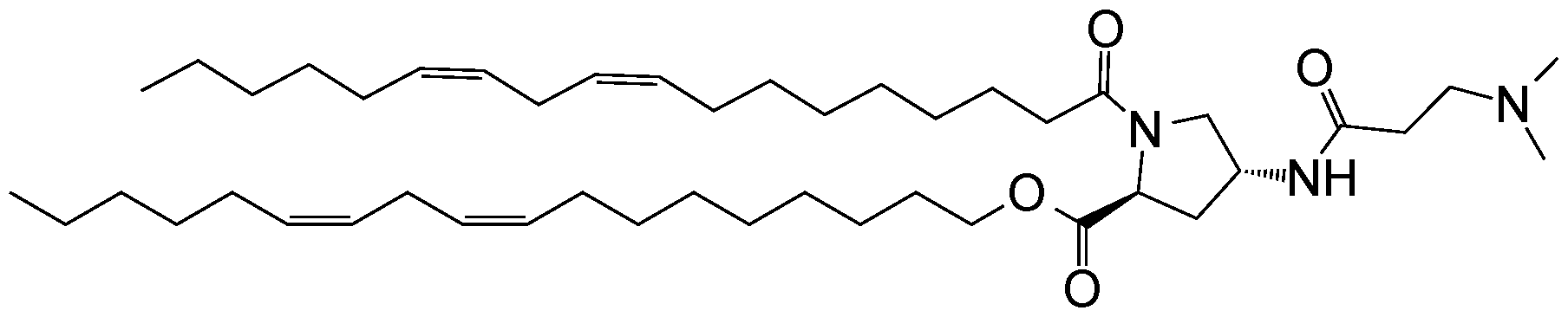
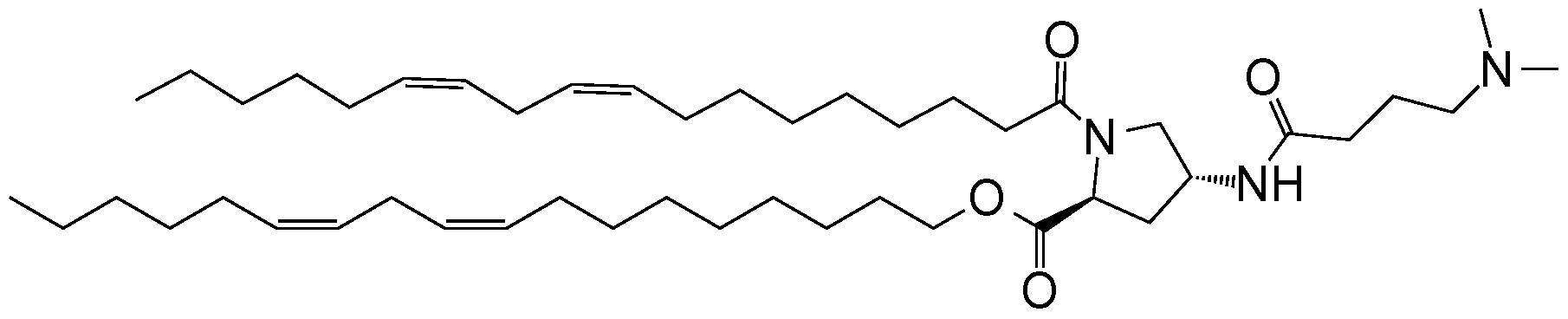
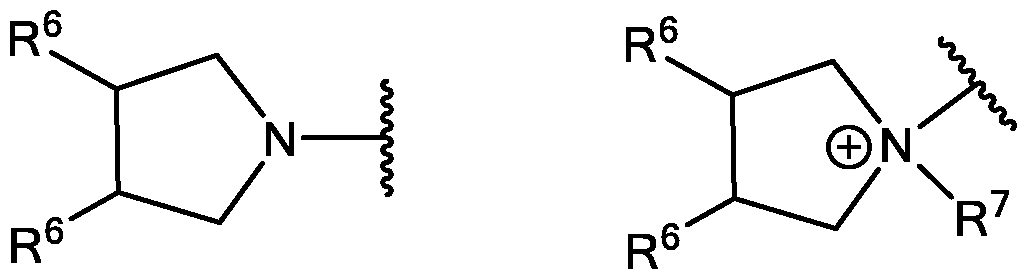
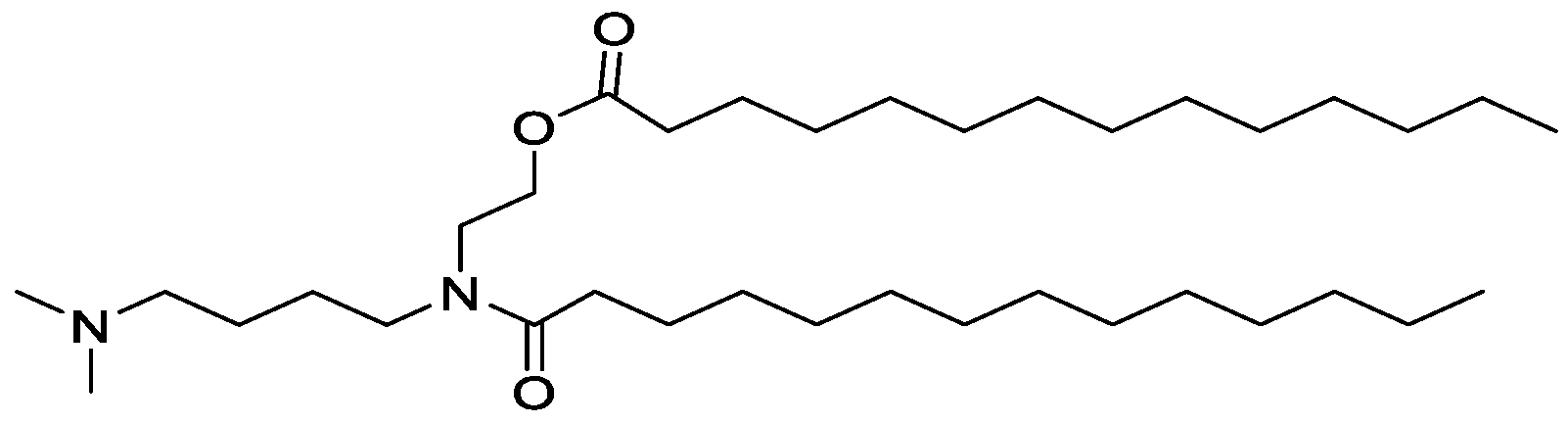
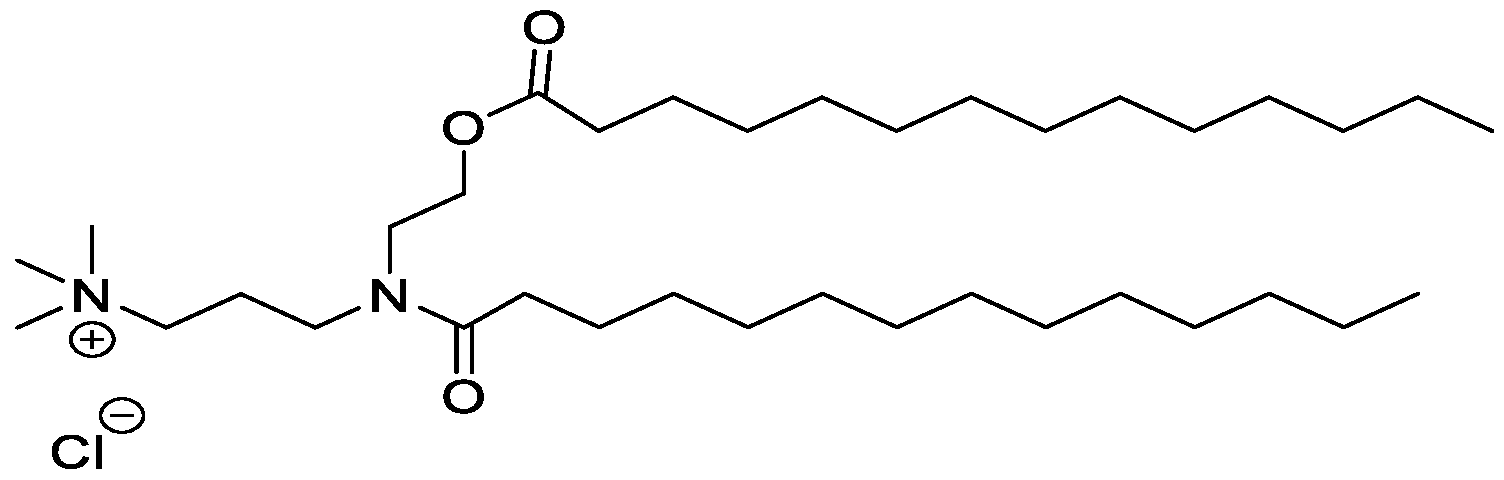
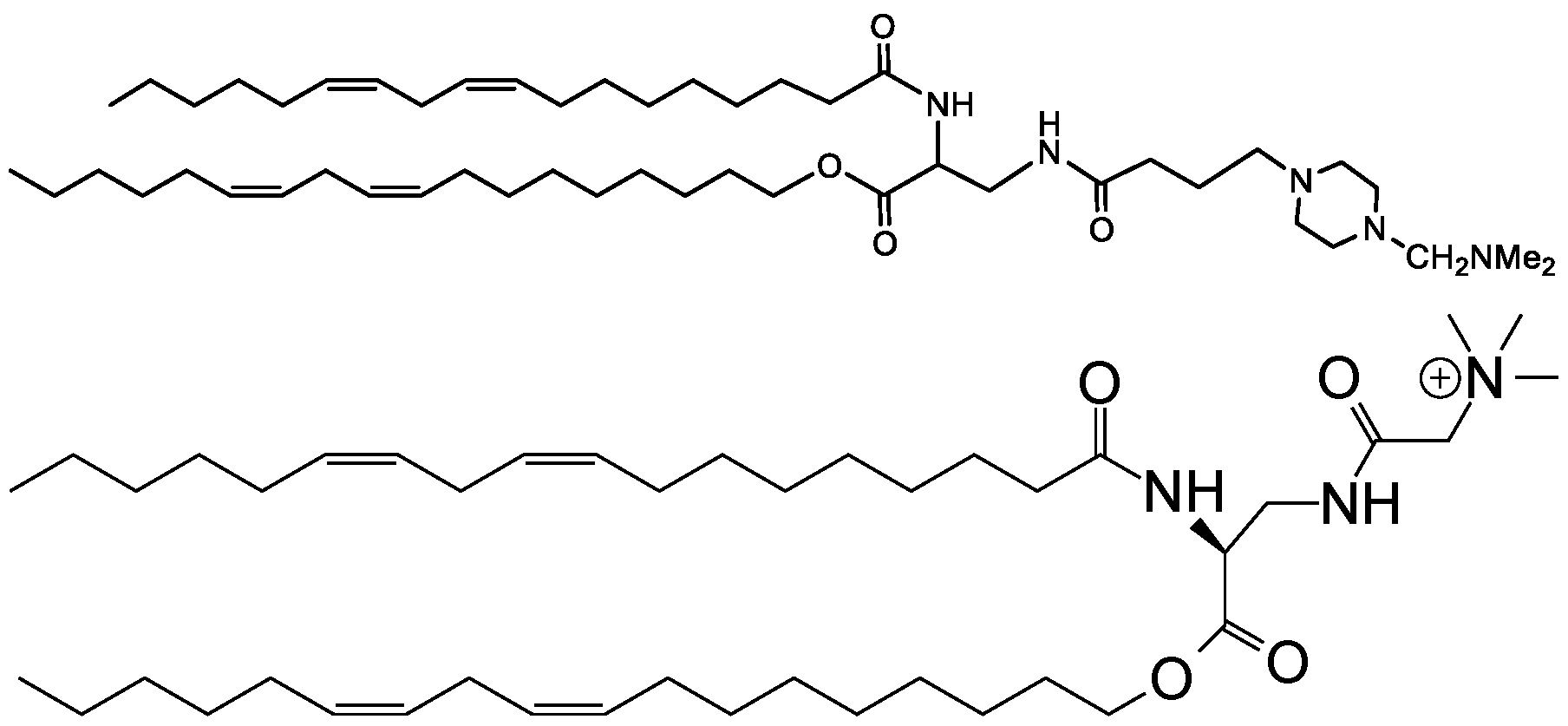
WO2016210190A1 - Ionizable compounds and compositions and uses thereof - Google Patents
Ionizable compounds and compositions and uses thereof Download PDFInfo
- Publication number
- WO2016210190A1 WO2016210190A1 PCT/US2016/039114 US2016039114W WO2016210190A1 WO 2016210190 A1 WO2016210190 A1 WO 2016210190A1 US 2016039114 W US2016039114 W US 2016039114W WO 2016210190 A1 WO2016210190 A1 WO 2016210190A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- compound
- aminoalkyl
- hydroxyalkyl
- independently
- Prior art date
Links
- 150000001875 compounds Chemical class 0.000 title claims abstract description 560
- 239000000203 mixture Substances 0.000 title claims abstract description 327
- 239000013543 active substance Substances 0.000 claims abstract description 41
- 239000002105 nanoparticle Substances 0.000 claims abstract description 27
- 239000003814 drug Substances 0.000 claims abstract description 9
- 125000000217 alkyl group Chemical group 0.000 claims description 336
- 125000003342 alkenyl group Chemical group 0.000 claims description 154
- 125000004103 aminoalkyl group Chemical group 0.000 claims description 146
- 125000002768 hydroxyalkyl group Chemical group 0.000 claims description 137
- 150000002632 lipids Chemical class 0.000 claims description 133
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 63
- 125000003545 alkoxy group Chemical group 0.000 claims description 50
- 125000005083 alkoxyalkoxy group Chemical group 0.000 claims description 49
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 41
- 239000003381 stabilizer Substances 0.000 claims description 29
- 230000005847 immunogenicity Effects 0.000 claims description 26
- 229910052739 hydrogen Inorganic materials 0.000 claims description 14
- 229910052760 oxygen Inorganic materials 0.000 claims description 13
- 108091030071 RNAI Proteins 0.000 claims description 10
- 230000009368 gene silencing by RNA Effects 0.000 claims description 10
- 108091032973 (ribonucleotides)n+m Proteins 0.000 claims description 9
- 102000040650 (ribonucleotides)n+m Human genes 0.000 claims description 9
- 108020004459 Small interfering RNA Proteins 0.000 claims description 9
- 125000004181 carboxyalkyl group Chemical group 0.000 claims description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 7
- 239000003937 drug carrier Substances 0.000 claims description 7
- 239000008194 pharmaceutical composition Substances 0.000 claims description 7
- 101100347612 Arabidopsis thaliana VIII-B gene Proteins 0.000 claims description 6
- 108700011259 MicroRNAs Proteins 0.000 claims description 6
- 108091007412 Piwi-interacting RNA Proteins 0.000 claims description 6
- 108091027967 Small hairpin RNA Proteins 0.000 claims description 6
- 201000010099 disease Diseases 0.000 claims description 5
- 229910052717 sulfur Inorganic materials 0.000 claims description 5
- 150000001732 carboxylic acid derivatives Chemical class 0.000 claims description 4
- 125000004185 ester group Chemical group 0.000 claims description 4
- 125000004183 alkoxy alkyl group Chemical group 0.000 claims description 3
- 125000005081 alkoxyalkoxyalkyl group Chemical group 0.000 claims description 3
- 125000005191 hydroxyalkylamino group Chemical group 0.000 claims description 3
- 239000000758 substrate Substances 0.000 claims description 3
- 230000000630 rising effect Effects 0.000 claims 4
- 238000004519 manufacturing process Methods 0.000 claims 1
- 208000024891 symptom Diseases 0.000 claims 1
- 238000000034 method Methods 0.000 abstract description 11
- 210000000056 organ Anatomy 0.000 abstract description 10
- 229960000074 biopharmaceutical Drugs 0.000 abstract description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N dichloromethane Natural products ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 258
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 222
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 147
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 107
- 239000000047 product Substances 0.000 description 104
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 94
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 90
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 65
- 238000002360 preparation method Methods 0.000 description 64
- 239000000243 solution Substances 0.000 description 57
- 239000011541 reaction mixture Substances 0.000 description 55
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 52
- 239000007788 liquid Substances 0.000 description 49
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 49
- 239000012044 organic layer Substances 0.000 description 48
- 238000000746 purification Methods 0.000 description 46
- 235000019439 ethyl acetate Nutrition 0.000 description 45
- 239000011734 sodium Substances 0.000 description 42
- 238000003818 flash chromatography Methods 0.000 description 39
- 239000000741 silica gel Substances 0.000 description 39
- 229910002027 silica gel Inorganic materials 0.000 description 39
- 239000000306 component Substances 0.000 description 38
- -1 lipid compounds Chemical class 0.000 description 37
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 37
- 239000000377 silicon dioxide Substances 0.000 description 33
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 32
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 29
- 238000009472 formulation Methods 0.000 description 27
- 238000003756 stirring Methods 0.000 description 27
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 26
- CCGKOQOJPYTBIH-UHFFFAOYSA-N ethenone Chemical compound C=C=O CCGKOQOJPYTBIH-UHFFFAOYSA-N 0.000 description 25
- 229910001868 water Inorganic materials 0.000 description 25
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 24
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 24
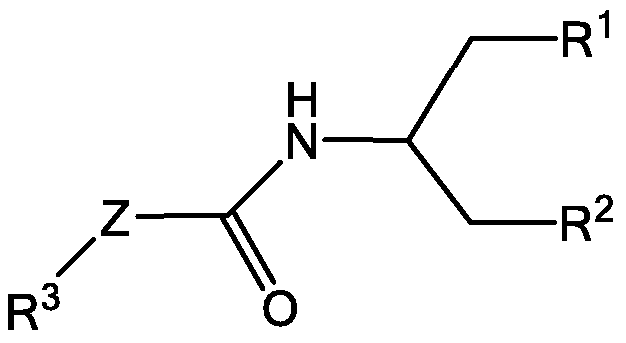
- 0 *CC(N(*)*)=O Chemical compound *CC(N(*)*)=O 0.000 description 21
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 20
- 229910000027 potassium carbonate Inorganic materials 0.000 description 19
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 18
- 229910052786 argon Inorganic materials 0.000 description 16
- 239000007789 gas Substances 0.000 description 16
- 239000007787 solid Substances 0.000 description 15
- DTQVDTLACAAQTR-UHFFFAOYSA-N trifluoroacetic acid Substances OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 15
- OYHQOLUKZRVURQ-HZJYTTRNSA-N Linoleic acid Chemical compound CCCCC\C=C/C\C=C/CCCCCCCC(O)=O OYHQOLUKZRVURQ-HZJYTTRNSA-N 0.000 description 14
- 238000006243 chemical reaction Methods 0.000 description 14
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 14
- OYHQOLUKZRVURQ-IXWMQOLASA-N linoleic acid Natural products CCCCC\C=C/C\C=C\CCCCCCCC(O)=O OYHQOLUKZRVURQ-IXWMQOLASA-N 0.000 description 14
- 235000020778 linoleic acid Nutrition 0.000 description 14
- 239000003921 oil Substances 0.000 description 14
- 235000019198 oils Nutrition 0.000 description 14
- 239000002904 solvent Substances 0.000 description 14
- 108020004707 nucleic acids Proteins 0.000 description 13
- 102000039446 nucleic acids Human genes 0.000 description 13
- 150000007523 nucleic acids Chemical class 0.000 description 13
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 13
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 12
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 12
- 239000007864 aqueous solution Substances 0.000 description 12
- 238000010936 aqueous wash Methods 0.000 description 12
- 238000000605 extraction Methods 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- 125000006355 carbonyl methylene group Chemical group [H]C([H])([*:2])C([*:1])=O 0.000 description 10
- 239000003795 chemical substances by application Substances 0.000 description 10
- 210000002966 serum Anatomy 0.000 description 10
- 235000017557 sodium bicarbonate Nutrition 0.000 description 10
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 10
- QTBSBXVTEAMEQO-UHFFFAOYSA-N acetic acid Substances CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 9
- 235000012000 cholesterol Nutrition 0.000 description 9
- 239000000706 filtrate Substances 0.000 description 9
- CBOIHMRHGLHBPB-UHFFFAOYSA-N hydroxymethyl Chemical compound O[CH2] CBOIHMRHGLHBPB-UHFFFAOYSA-N 0.000 description 9
- 230000002452 interceptive effect Effects 0.000 description 8
- 229920001223 polyethylene glycol Polymers 0.000 description 8
- 150000001721 carbon Chemical group 0.000 description 7
- 229910052799 carbon Inorganic materials 0.000 description 7
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 7
- 229920001427 mPEG Polymers 0.000 description 7
- 239000000463 material Substances 0.000 description 7
- 210000001519 tissue Anatomy 0.000 description 7
- SNKAWJBJQDLSFF-NVKMUCNASA-N 1,2-dioleoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/CCCCCCCC SNKAWJBJQDLSFF-NVKMUCNASA-N 0.000 description 6
- LSTRKXWIZZZYAS-UHFFFAOYSA-N 2-bromoacetyl bromide Chemical compound BrCC(Br)=O LSTRKXWIZZZYAS-UHFFFAOYSA-N 0.000 description 6
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 6
- 125000006364 carbonyl oxy methylene group Chemical group [H]C([H])([*:2])OC([*:1])=O 0.000 description 6
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 6
- 238000005538 encapsulation Methods 0.000 description 6
- 125000004005 formimidoyl group Chemical group [H]\N=C(/[H])* 0.000 description 6
- OYHQOLUKZRVURQ-UHFFFAOYSA-M octadeca-9,12-dienoate Chemical compound CCCCCC=CCC=CCCCCCCCC([O-])=O OYHQOLUKZRVURQ-UHFFFAOYSA-M 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N tetrahydrofuran Substances C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- UOXJNGFFPMOZDM-UHFFFAOYSA-N 2-[di(propan-2-yl)amino]ethylsulfanyl-methylphosphinic acid Chemical compound CC(C)N(C(C)C)CCSP(C)(O)=O UOXJNGFFPMOZDM-UHFFFAOYSA-N 0.000 description 5
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 5
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium on carbon Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 5
- 239000002202 Polyethylene glycol Substances 0.000 description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 5
- 239000003957 anion exchange resin Substances 0.000 description 5
- 210000004027 cell Anatomy 0.000 description 5
- 229920001429 chelating resin Polymers 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- JXNPEDYJTDQORS-HZJYTTRNSA-N (9Z,12Z)-octadecadien-1-ol Chemical compound CCCCC\C=C/C\C=C/CCCCCCCCO JXNPEDYJTDQORS-HZJYTTRNSA-N 0.000 description 4
- JZNWSCPGTDBMEW-UHFFFAOYSA-N Glycerophosphorylethanolamin Natural products NCCOP(O)(=O)OCC(O)CO JZNWSCPGTDBMEW-UHFFFAOYSA-N 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 4
- AQUVQGSNKVDBBF-UHFFFAOYSA-N azetidin-3-ylmethanol;hydrochloride Chemical compound Cl.OCC1CNC1 AQUVQGSNKVDBBF-UHFFFAOYSA-N 0.000 description 4
- 239000012230 colorless oil Substances 0.000 description 4
- 239000005457 ice water Substances 0.000 description 4
- JXNPEDYJTDQORS-UHFFFAOYSA-N linoleyl alcohol Natural products CCCCCC=CCC=CCCCCCCCCO JXNPEDYJTDQORS-UHFFFAOYSA-N 0.000 description 4
- 210000004185 liver Anatomy 0.000 description 4
- 210000004072 lung Anatomy 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Substances [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 4
- 238000001890 transfection Methods 0.000 description 4
- 239000003981 vehicle Substances 0.000 description 4
- OQQOAWVKVDAJOI-VWLOTQADSA-N 1,2-dilauroyl-sn-glycerol Chemical compound CCCCCCCCCCCC(=O)OC[C@H](CO)OC(=O)CCCCCCCCCCC OQQOAWVKVDAJOI-VWLOTQADSA-N 0.000 description 3
- TWJNQYPJQDRXPH-UHFFFAOYSA-N 2-cyanobenzohydrazide Chemical compound NNC(=O)C1=CC=CC=C1C#N TWJNQYPJQDRXPH-UHFFFAOYSA-N 0.000 description 3
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 3
- LWEOFVINMVZGAS-UHFFFAOYSA-N 3-piperazin-1-ylpropan-1-ol Chemical compound OCCCN1CCNCC1 LWEOFVINMVZGAS-UHFFFAOYSA-N 0.000 description 3
- 206010016654 Fibrosis Diseases 0.000 description 3
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 3
- TUNFSRHWOTWDNC-UHFFFAOYSA-N Myristic acid Natural products CCCCCCCCCCCCCC(O)=O TUNFSRHWOTWDNC-UHFFFAOYSA-N 0.000 description 3
- 235000021360 Myristic acid Nutrition 0.000 description 3
- UEEJHVSXFDXPFK-UHFFFAOYSA-N N-dimethylaminoethanol Chemical compound CN(C)CCO UEEJHVSXFDXPFK-UHFFFAOYSA-N 0.000 description 3
- 229930182558 Sterol Natural products 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- 125000001821 azanediyl group Chemical group [H]N(*)* 0.000 description 3
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 3
- 125000002091 cationic group Chemical group 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- 238000011097 chromatography purification Methods 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 230000004761 fibrosis Effects 0.000 description 3
- 239000001257 hydrogen Substances 0.000 description 3
- UEXQBEVWFZKHNB-UHFFFAOYSA-N intermediate 29 Natural products C1=CC(N)=CC=C1NC1=NC=CC=N1 UEXQBEVWFZKHNB-UHFFFAOYSA-N 0.000 description 3
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 239000002502 liposome Substances 0.000 description 3
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 3
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- JUJWROOIHBZHMG-UHFFFAOYSA-O pyridinium Chemical compound C1=CC=[NH+]C=C1 JUJWROOIHBZHMG-UHFFFAOYSA-O 0.000 description 3
- 239000012279 sodium borohydride Substances 0.000 description 3
- 229910000033 sodium borohydride Inorganic materials 0.000 description 3
- 150000003432 sterols Chemical class 0.000 description 3
- 235000003702 sterols Nutrition 0.000 description 3
- 125000001424 substituent group Chemical group 0.000 description 3
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 3
- OILXMJHPFNGGTO-UHFFFAOYSA-N (22E)-(24xi)-24-methylcholesta-5,22-dien-3beta-ol Natural products C1C=C2CC(O)CCC2(C)C2C1C1CCC(C(C)C=CC(C)C(C)C)C1(C)CC2 OILXMJHPFNGGTO-UHFFFAOYSA-N 0.000 description 2
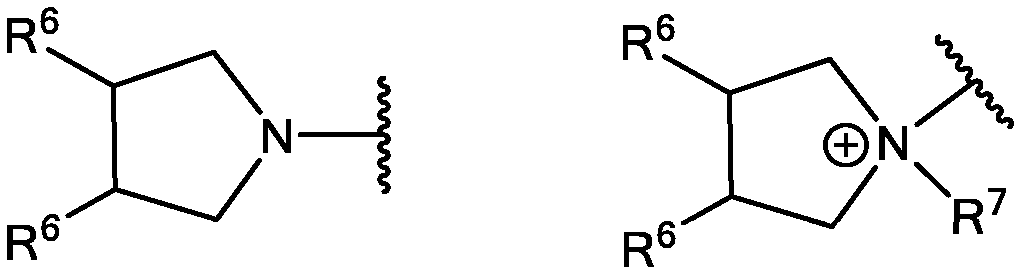
- JCZPOYAMKJFOLA-IMJSIDKUSA-N (3s,4s)-pyrrolidine-3,4-diol Chemical compound O[C@H]1CNC[C@@H]1O JCZPOYAMKJFOLA-IMJSIDKUSA-N 0.000 description 2
- MAUMSNABMVEOGP-UHFFFAOYSA-N (methyl-$l^{2}-azanyl)methane Chemical compound C[N]C MAUMSNABMVEOGP-UHFFFAOYSA-N 0.000 description 2
- KILNVBDSWZSGLL-KXQOOQHDSA-N 1,2-dihexadecanoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCCCCCCCCC KILNVBDSWZSGLL-KXQOOQHDSA-N 0.000 description 2
- UHUSDOQQWJGJQS-QNGWXLTQSA-N 1,2-dioctadecanoyl-sn-glycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](CO)OC(=O)CCCCCCCCCCCCCCCCC UHUSDOQQWJGJQS-QNGWXLTQSA-N 0.000 description 2
- JEJLGIQLPYYGEE-XIFFEERXSA-N 1,2-dipalmitoyl-sn-glycerol Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](CO)OC(=O)CCCCCCCCCCCCCCC JEJLGIQLPYYGEE-XIFFEERXSA-N 0.000 description 2
- NRJAVPSFFCBXDT-HUESYALOSA-N 1,2-distearoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCCCCCCCCCCC NRJAVPSFFCBXDT-HUESYALOSA-N 0.000 description 2
- JFBCSFJKETUREV-LJAQVGFWSA-N 1,2-ditetradecanoyl-sn-glycerol Chemical compound CCCCCCCCCCCCCC(=O)OC[C@H](CO)OC(=O)CCCCCCCCCCCCC JFBCSFJKETUREV-LJAQVGFWSA-N 0.000 description 2
- FJKROLUGYXJWQN-UHFFFAOYSA-N 4-hydroxybenzoic acid Chemical compound OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 2
- OQMZNAMGEHIHNN-UHFFFAOYSA-N 7-Dehydrostigmasterol Natural products C1C(O)CCC2(C)C(CCC3(C(C(C)C=CC(CC)C(C)C)CCC33)C)C3=CC=C21 OQMZNAMGEHIHNN-UHFFFAOYSA-N 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- NSGDYZCDUPSTQT-UHFFFAOYSA-N N-[5-bromo-1-[(4-fluorophenyl)methyl]-4-methyl-2-oxopyridin-3-yl]cycloheptanecarboxamide Chemical compound Cc1c(Br)cn(Cc2ccc(F)cc2)c(=O)c1NC(=O)C1CCCCCC1 NSGDYZCDUPSTQT-UHFFFAOYSA-N 0.000 description 2
- SJDMTGSQPOFVLR-UHFFFAOYSA-N [10,13-dimethyl-17-(6-methylheptan-2-yl)-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3-yl] tetradecanoate Chemical compound C12CCC3(C)C(C(C)CCCC(C)C)CCC3C2CC=C2C1(C)CCC(OC(=O)CCCCCCCCCCCCC)C2 SJDMTGSQPOFVLR-UHFFFAOYSA-N 0.000 description 2
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 125000004429 atom Chemical group 0.000 description 2
- LGJMUZUPVCAVPU-UHFFFAOYSA-N beta-Sitostanol Natural products C1CC2CC(O)CCC2(C)C2C1C1CCC(C(C)CCC(CC)C(C)C)C1(C)CC2 LGJMUZUPVCAVPU-UHFFFAOYSA-N 0.000 description 2
- AOJDZKCUAATBGE-UHFFFAOYSA-N bromomethane Chemical compound Br[CH2] AOJDZKCUAATBGE-UHFFFAOYSA-N 0.000 description 2
- 125000001589 carboacyl group Chemical group 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 150000001841 cholesterols Chemical class 0.000 description 2
- 208000011444 chronic liver failure Diseases 0.000 description 2
- 210000001072 colon Anatomy 0.000 description 2
- 239000010779 crude oil Substances 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- QBSJHOGDIUQWTH-UHFFFAOYSA-N dihydrolanosterol Natural products CC(C)CCCC(C)C1CCC2(C)C3=C(CCC12C)C4(C)CCC(C)(O)C(C)(C)C4CC3 QBSJHOGDIUQWTH-UHFFFAOYSA-N 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- HCUYBXPSSCRKRF-UHFFFAOYSA-N diphosgene Chemical compound ClC(=O)OC(Cl)(Cl)Cl HCUYBXPSSCRKRF-UHFFFAOYSA-N 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 2
- 238000004108 freeze drying Methods 0.000 description 2
- JFCQEDHGNNZCLN-UHFFFAOYSA-L glutarate(2-) Chemical compound [O-]C(=O)CCCC([O-])=O JFCQEDHGNNZCLN-UHFFFAOYSA-L 0.000 description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 210000000496 pancreas Anatomy 0.000 description 2
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 2
- 150000008104 phosphatidylethanolamines Chemical class 0.000 description 2
- 150000003904 phospholipids Chemical class 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- ONIBWKKTOPOVIA-UHFFFAOYSA-M prolinate Chemical compound [O-]C(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-M 0.000 description 2
- 208000005069 pulmonary fibrosis Diseases 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- KMUNFRBJXIEULW-UHFFFAOYSA-N tert-butyl n,n-bis(2-hydroxyethyl)carbamate Chemical compound CC(C)(C)OC(=O)N(CCO)CCO KMUNFRBJXIEULW-UHFFFAOYSA-N 0.000 description 2
- 229940124597 therapeutic agent Drugs 0.000 description 2
- OSELKOCHBMDKEJ-UHFFFAOYSA-N (10R)-3c-Hydroxy-10r.13c-dimethyl-17c-((R)-1-methyl-4-isopropyl-hexen-(4c)-yl)-(8cH.9tH.14tH)-Delta5-tetradecahydro-1H-cyclopenta[a]phenanthren Natural products C1C=C2CC(O)CCC2(C)C2C1C1CCC(C(C)CCC(=CC)C(C)C)C1(C)CC2 OSELKOCHBMDKEJ-UHFFFAOYSA-N 0.000 description 1
- BQPPJGMMIYJVBR-UHFFFAOYSA-N (10S)-3c-Acetoxy-4.4.10r.13c.14t-pentamethyl-17c-((R)-1.5-dimethyl-hexen-(4)-yl)-(5tH)-Delta8-tetradecahydro-1H-cyclopenta[a]phenanthren Natural products CC12CCC(OC(C)=O)C(C)(C)C1CCC1=C2CCC2(C)C(C(CCC=C(C)C)C)CCC21C BQPPJGMMIYJVBR-UHFFFAOYSA-N 0.000 description 1
- MBZYKEVPFYHDOH-UHFFFAOYSA-N (10S)-3c-Hydroxy-4.4.10r.13t.14c-pentamethyl-17t-((R)-1.5-dimethyl-hexyl)-(5tH)-Delta8-tetradecahydro-1H-cyclopenta[a]phenanthren Natural products CC12CCC(O)C(C)(C)C1CCC1=C2CCC2(C)C(C(C)CCCC(C)C)CCC21C MBZYKEVPFYHDOH-UHFFFAOYSA-N 0.000 description 1
- RWTQCZGAMKTBRV-PTHRTHQKSA-N (1s,2r,5s,10s,11s,14r,15r)-2,15-dimethyl-14-[(2r)-6-methylheptan-2-yl]tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-7-en-5-yl pentanoate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCC)C1 RWTQCZGAMKTBRV-PTHRTHQKSA-N 0.000 description 1
- NEZDNQCXEZDCBI-WJOKGBTCSA-N (2-aminoethoxy)[(2r)-2,3-bis(tetradecanoyloxy)propoxy]phosphinic acid Chemical compound CCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OCCN)OC(=O)CCCCCCCCCCCCC NEZDNQCXEZDCBI-WJOKGBTCSA-N 0.000 description 1
- RQOCXCFLRBRBCS-UHFFFAOYSA-N (22E)-cholesta-5,7,22-trien-3beta-ol Natural products C1C(O)CCC2(C)C(CCC3(C(C(C)C=CCC(C)C)CCC33)C)C3=CC=C21 RQOCXCFLRBRBCS-UHFFFAOYSA-N 0.000 description 1
- MCWVPSBQQXUCTB-UHFFFAOYSA-N (24Z)-5alpha-Stigmasta-7,24(28)-dien-3beta-ol Natural products C1C(O)CCC2(C)C(CCC3(C(C(C)CCC(=CC)C(C)C)CCC33)C)C3=CCC21 MCWVPSBQQXUCTB-UHFFFAOYSA-N 0.000 description 1
- RGZSQWQPBWRIAQ-HUUCEWRRSA-N (2r)-6-methyl-2-[(1s)-4-methylcyclohex-3-en-1-yl]hept-5-en-2-ol Chemical compound CC(C)=CCC[C@@](C)(O)[C@H]1CCC(C)=CC1 RGZSQWQPBWRIAQ-HUUCEWRRSA-N 0.000 description 1
- PKAUMAVONPSDRW-IBGZPJMESA-N (2s)-2-(9h-fluoren-9-ylmethoxycarbonylamino)-3-[(2-methylpropan-2-yl)oxycarbonylamino]propanoic acid Chemical compound C1=CC=C2C(COC(=O)N[C@@H](CNC(=O)OC(C)(C)C)C(O)=O)C3=CC=CC=C3C2=C1 PKAUMAVONPSDRW-IBGZPJMESA-N 0.000 description 1
- OTKXCALUHMPIGM-FQEVSTJZSA-N (2s)-2-(9h-fluoren-9-ylmethoxycarbonylamino)-5-[(2-methylpropan-2-yl)oxy]-5-oxopentanoic acid Chemical compound C1=CC=C2C(COC(=O)N[C@@H](CCC(=O)OC(C)(C)C)C(O)=O)C3=CC=CC=C3C2=C1 OTKXCALUHMPIGM-FQEVSTJZSA-N 0.000 description 1
- CHGIKSSZNBCNDW-UHFFFAOYSA-N (3beta,5alpha)-4,4-Dimethylcholesta-8,24-dien-3-ol Natural products CC12CCC(O)C(C)(C)C1CCC1=C2CCC2(C)C(C(CCC=C(C)C)C)CCC21 CHGIKSSZNBCNDW-UHFFFAOYSA-N 0.000 description 1
- NXLNNXIXOYSCMB-UHFFFAOYSA-N (4-nitrophenyl) carbonochloridate Chemical compound [O-][N+](=O)C1=CC=C(OC(Cl)=O)C=C1 NXLNNXIXOYSCMB-UHFFFAOYSA-N 0.000 description 1
- XIIAYQZJNBULGD-UHFFFAOYSA-N (5alpha)-cholestane Natural products C1CC2CCCCC2(C)C2C1C1CCC(C(C)CCCC(C)C)C1(C)CC2 XIIAYQZJNBULGD-UHFFFAOYSA-N 0.000 description 1
- 239000001149 (9Z,12Z)-octadeca-9,12-dienoate Substances 0.000 description 1
- LVNGJLRDBYCPGB-LDLOPFEMSA-N (R)-1,2-distearoylphosphatidylethanolamine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[NH3+])OC(=O)CCCCCCCCCCCCCCCCC LVNGJLRDBYCPGB-LDLOPFEMSA-N 0.000 description 1
- 125000003088 (fluoren-9-ylmethoxy)carbonyl group Chemical group 0.000 description 1
- LZLVZIFMYXDKCN-QJWFYWCHSA-N 1,2-di-O-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC LZLVZIFMYXDKCN-QJWFYWCHSA-N 0.000 description 1
- FVXDQWZBHIXIEJ-LNDKUQBDSA-N 1,2-di-[(9Z,12Z)-octadecadienoyl]-sn-glycero-3-phosphocholine Chemical compound CCCCC\C=C/C\C=C/CCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/C\C=C/CCCCC FVXDQWZBHIXIEJ-LNDKUQBDSA-N 0.000 description 1
- SLKDGVPOSSLUAI-PGUFJCEWSA-N 1,2-dihexadecanoyl-sn-glycero-3-phosphoethanolamine zwitterion Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OCCN)OC(=O)CCCCCCCCCCCCCCC SLKDGVPOSSLUAI-PGUFJCEWSA-N 0.000 description 1
- PORPENFLTBBHSG-MGBGTMOVSA-N 1,2-dihexadecanoyl-sn-glycerol-3-phosphate Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(O)=O)OC(=O)CCCCCCCCCCCCCCC PORPENFLTBBHSG-MGBGTMOVSA-N 0.000 description 1
- UKDDQGWMHWQMBI-UHFFFAOYSA-O 1,2-diphytanoyl-sn-glycero-3-phosphocholine Chemical compound CC(C)CCCC(C)CCCC(C)CCCC(C)CC(=O)OCC(COP(O)(=O)OCC[N+](C)(C)C)OC(=O)CC(C)CCCC(C)CCCC(C)CCCC(C)C UKDDQGWMHWQMBI-UHFFFAOYSA-O 0.000 description 1
- TZCPCKNHXULUIY-RGULYWFUSA-N 1,2-distearoyl-sn-glycero-3-phosphoserine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OC[C@H](N)C(O)=O)OC(=O)CCCCCCCCCCCCCCCCC TZCPCKNHXULUIY-RGULYWFUSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- BIAAQBNMRITRDV-UHFFFAOYSA-N 1-(chloromethoxy)-2-methoxyethane Chemical compound COCCOCCl BIAAQBNMRITRDV-UHFFFAOYSA-N 0.000 description 1
- RYCNUMLMNKHWPZ-SNVBAGLBSA-N 1-acetyl-sn-glycero-3-phosphocholine Chemical compound CC(=O)OC[C@@H](O)COP([O-])(=O)OCC[N+](C)(C)C RYCNUMLMNKHWPZ-SNVBAGLBSA-N 0.000 description 1
- WGCYRFWNGRMRJA-UHFFFAOYSA-N 1-ethylpiperazine Chemical compound CCN1CCNCC1 WGCYRFWNGRMRJA-UHFFFAOYSA-N 0.000 description 1
- WTJKGGKOPKCXLL-VYOBOKEXSA-N 1-hexadecanoyl-2-(9Z-octadecenoyl)-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/CCCCCCCC WTJKGGKOPKCXLL-VYOBOKEXSA-N 0.000 description 1
- XYTLYKGXLMKYMV-UHFFFAOYSA-N 14alpha-methylzymosterol Natural products CC12CCC(O)CC1CCC1=C2CCC2(C)C(C(CCC=C(C)C)C)CCC21C XYTLYKGXLMKYMV-UHFFFAOYSA-N 0.000 description 1
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 1
- FKASAVXZZLJTNX-UHFFFAOYSA-N 2-(dimethylamino)acetic acid;hydrochloride Chemical compound [Cl-].C[NH+](C)CC(O)=O FKASAVXZZLJTNX-UHFFFAOYSA-N 0.000 description 1
- DENMGZODXQRYAR-UHFFFAOYSA-N 2-(dimethylamino)ethanethiol Chemical compound CN(C)CCS DENMGZODXQRYAR-UHFFFAOYSA-N 0.000 description 1
- SFFSGPCYJCMDJM-UHFFFAOYSA-N 2-[2-(3-oxo-1,2-benzoselenazol-2-yl)ethyl]-1,2-benzoselenazol-3-one Chemical compound [se]1C2=CC=CC=C2C(=O)N1CCN1C(=O)C(C=CC=C2)=C2[se]1 SFFSGPCYJCMDJM-UHFFFAOYSA-N 0.000 description 1
- DZXXJBHDASOXEV-KWXKLSQISA-N 2-[3-(dimethylamino)propyl-[(9Z,12Z)-octadeca-9,12-dienoyl]amino]ethyl (9Z,12Z)-octadeca-9,12-dienoate Chemical compound C(CCCCCCC\C=C/C\C=C/CCCCC)(=O)OCCN(C(CCCCCCC\C=C/C\C=C/CCCCC)=O)CCCN(C)C DZXXJBHDASOXEV-KWXKLSQISA-N 0.000 description 1
- APCTXOMENLJELA-UHFFFAOYSA-N 2-[3-(dimethylamino)propyl-tetradecanoylamino]ethyl tetradecanoate Chemical compound C(CCCCCCCCCCCCC)(=O)OCCN(C(CCCCCCCCCCCCC)=O)CCCN(C)C APCTXOMENLJELA-UHFFFAOYSA-N 0.000 description 1
- TUINMNKNCRPPQT-KWXKLSQISA-N 2-[4-(dimethylamino)butyl-[(9Z,12Z)-octadeca-9,12-dienoyl]amino]ethyl (9Z,12Z)-octadeca-9,12-dienoate Chemical compound C(CCCCCCC\C=C/C\C=C/CCCCC)(=O)OCCN(C(CCCCCCC\C=C/C\C=C/CCCCC)=O)CCCCN(C)C TUINMNKNCRPPQT-KWXKLSQISA-N 0.000 description 1
- OFNPZHPBVQKPQL-ZPPAUJSGSA-N 2-[bis[2-[(9Z,12Z)-octadeca-9,12-dienoyl]oxyethyl]carbamoyloxy]ethyl-trimethylazanium Chemical compound C(CCCCCCC\C=C/C\C=C/CCCCC)(=O)OCCN(C(=O)OCC[N+](C)(C)C)CCOC(CCCCCCC\C=C/C\C=C/CCCCC)=O OFNPZHPBVQKPQL-ZPPAUJSGSA-N 0.000 description 1
- OBPOCAKICKFMGM-PGUFJCEWSA-N 2-[di(hexadecanoyl)amino]ethyl [(2R)-2,3-dihydroxypropyl] hydrogen phosphate Chemical compound C(CCCCCCCCCCCCCCC)(=O)N(CCOP(OC[C@@H](CO)O)(=O)O)C(CCCCCCCCCCCCCCC)=O OBPOCAKICKFMGM-PGUFJCEWSA-N 0.000 description 1
- LKADKXJLDMQPTL-UHFFFAOYSA-N 2-[tert-butyl(dimethyl)silyl]acetaldehyde Chemical compound CC(C)(C)[Si](C)(C)CC=O LKADKXJLDMQPTL-UHFFFAOYSA-N 0.000 description 1
- ZLGYVWRJIZPQMM-HHHXNRCGSA-N 2-azaniumylethyl [(2r)-2,3-di(dodecanoyloxy)propyl] phosphate Chemical compound CCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OCCN)OC(=O)CCCCCCCCCCC ZLGYVWRJIZPQMM-HHHXNRCGSA-N 0.000 description 1
- MBZYKEVPFYHDOH-BQNIITSRSA-N 24,25-dihydrolanosterol Chemical compound C([C@@]12C)C[C@H](O)C(C)(C)[C@@H]1CCC1=C2CC[C@]2(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@]21C MBZYKEVPFYHDOH-BQNIITSRSA-N 0.000 description 1
- SURVRMRLLSFWHA-UHFFFAOYSA-N 3-(dimethylazaniumyl)butanoate Chemical compound CN(C)C(C)CC(O)=O SURVRMRLLSFWHA-UHFFFAOYSA-N 0.000 description 1
- XNDWCDKNSZRXQY-MURFETPASA-N 3-[[2-(dimethylamino)acetyl]amino]-5-[(9Z,12Z)-octadeca-9,12-dienoxy]-5-oxopentanoic acid Chemical compound CCCCC/C=C\C/C=C\CCCCCCCCOC(=O)CC(CC(=O)O)NC(=O)CN(C)C XNDWCDKNSZRXQY-MURFETPASA-N 0.000 description 1
- 125000004575 3-pyrrolidinyl group Chemical group [H]N1C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- FPTJELQXIUUCEY-UHFFFAOYSA-N 3beta-Hydroxy-lanostan Natural products C1CC2C(C)(C)C(O)CCC2(C)C2C1C1(C)CCC(C(C)CCCC(C)C)C1(C)CC2 FPTJELQXIUUCEY-UHFFFAOYSA-N 0.000 description 1
- XUGISPSHIFXEHZ-UHFFFAOYSA-N 3beta-acetoxy-cholest-5-ene Natural products C1C=C2CC(OC(C)=O)CCC2(C)C2C1C1CCC(C(C)CCCC(C)C)C1(C)CC2 XUGISPSHIFXEHZ-UHFFFAOYSA-N 0.000 description 1
- YEYCQJVCAMFWCO-UHFFFAOYSA-N 3beta-cholesteryl formate Natural products C1C=C2CC(OC=O)CCC2(C)C2C1C1CCC(C(C)CCCC(C)C)C1(C)CC2 YEYCQJVCAMFWCO-UHFFFAOYSA-N 0.000 description 1
- 229940090248 4-hydroxybenzoic acid Drugs 0.000 description 1
- CHAICOCMBPWCLB-HKBQPEDESA-N 4-oxo-4-[[(2S)-1-oxo-3-tetradecanoyloxy-1-(tetradecylamino)propan-2-yl]amino]butanoic acid Chemical compound O=C(CCC(=O)O)N[C@H](C(NCCCCCCCCCCCCCC)=O)COC(CCCCCCCCCCCCC)=O CHAICOCMBPWCLB-HKBQPEDESA-N 0.000 description 1
- QDDQSSZZYNCVHC-UHFFFAOYSA-N 5-[(4-tert-butylphenoxy)carbonylamino]-2-hydroxybenzoic acid Chemical compound C1=CC(C(C)(C)C)=CC=C1OC(=O)NC1=CC=C(O)C(C(O)=O)=C1 QDDQSSZZYNCVHC-UHFFFAOYSA-N 0.000 description 1
- QETLKNDKQOXZRP-XTGBIJOFSA-N 5alpha-cholest-8-en-3beta-ol Chemical compound C([C@@]12C)C[C@H](O)C[C@@H]1CCC1=C2CC[C@]2(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]21 QETLKNDKQOXZRP-XTGBIJOFSA-N 0.000 description 1
- QETLKNDKQOXZRP-UHFFFAOYSA-N 5alpha-cholest-8-en-3beta-ol Natural products CC12CCC(O)CC1CCC1=C2CCC2(C)C(C(C)CCCC(C)C)CCC21 QETLKNDKQOXZRP-UHFFFAOYSA-N 0.000 description 1
- SFHYNDMGZXWXBU-LIMNOBDPSA-N 6-amino-2-[[(e)-(3-formylphenyl)methylideneamino]carbamoylamino]-1,3-dioxobenzo[de]isoquinoline-5,8-disulfonic acid Chemical compound O=C1C(C2=3)=CC(S(O)(=O)=O)=CC=3C(N)=C(S(O)(=O)=O)C=C2C(=O)N1NC(=O)N\N=C\C1=CC=CC(C=O)=C1 SFHYNDMGZXWXBU-LIMNOBDPSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- OYHQOLUKZRVURQ-HZJYTTRNSA-M 9-cis,12-cis-Octadecadienoate Chemical compound CCCCC\C=C/C\C=C/CCCCCCCC([O-])=O OYHQOLUKZRVURQ-HZJYTTRNSA-M 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- IMXSFYNMSOULQS-UHFFFAOYSA-N Arachidonsaeure-cholesterylester Natural products C12CCC3(C)C(C(C)CCCC(C)C)CCC3C2CC=C2C1(C)CCC(OC(=O)CCCC=CCC=CCC=CCC=CCCCCC)C2 IMXSFYNMSOULQS-UHFFFAOYSA-N 0.000 description 1
- OILXMJHPFNGGTO-NRHJOKMGSA-N Brassicasterol Natural products O[C@@H]1CC=2[C@@](C)([C@@H]3[C@H]([C@H]4[C@](C)([C@H]([C@@H](/C=C/[C@H](C(C)C)C)C)CC4)CC3)CC=2)CC1 OILXMJHPFNGGTO-NRHJOKMGSA-N 0.000 description 1
- CKDZWMVGDHGMFR-UHFFFAOYSA-N Buttersaeure-cholesterylester Natural products C12CCC3(C)C(C(C)CCCC(C)C)CCC3C2CC=C2C1(C)CCC(OC(=O)CCC)C2 CKDZWMVGDHGMFR-UHFFFAOYSA-N 0.000 description 1
- QKVSMSABRNCNRS-UHFFFAOYSA-N CC(C)CN1CCOCC1 Chemical compound CC(C)CN1CCOCC1 QKVSMSABRNCNRS-UHFFFAOYSA-N 0.000 description 1
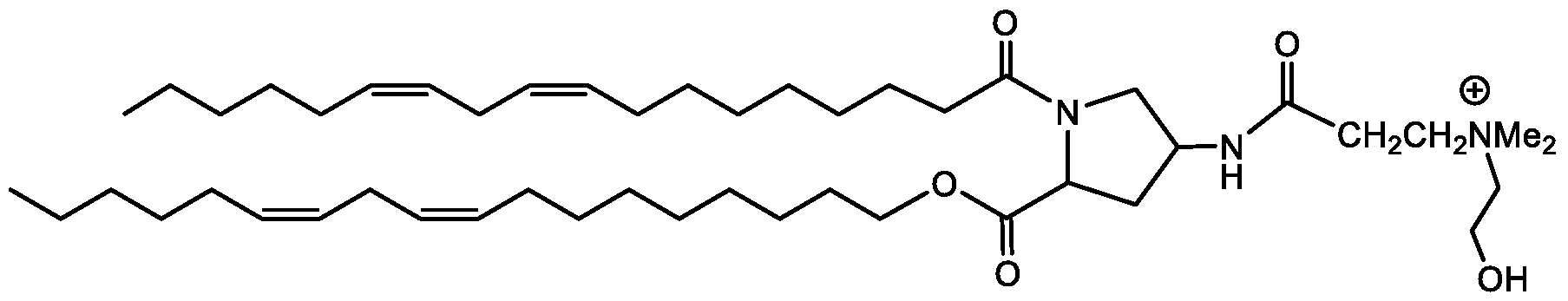
- LCZICHQZWSNXSF-MAZCIEHSSA-N CCCCC/C=C\C/C=C\CCCCCCCC(OCCCC(CCOC(CCCCCCC/C=C\C/C=C\CCCCC)=O)NC(O)OCCO)=O Chemical compound CCCCC/C=C\C/C=C\CCCCCCCC(OCCCC(CCOC(CCCCCCC/C=C\C/C=C\CCCCC)=O)NC(O)OCCO)=O LCZICHQZWSNXSF-MAZCIEHSSA-N 0.000 description 1
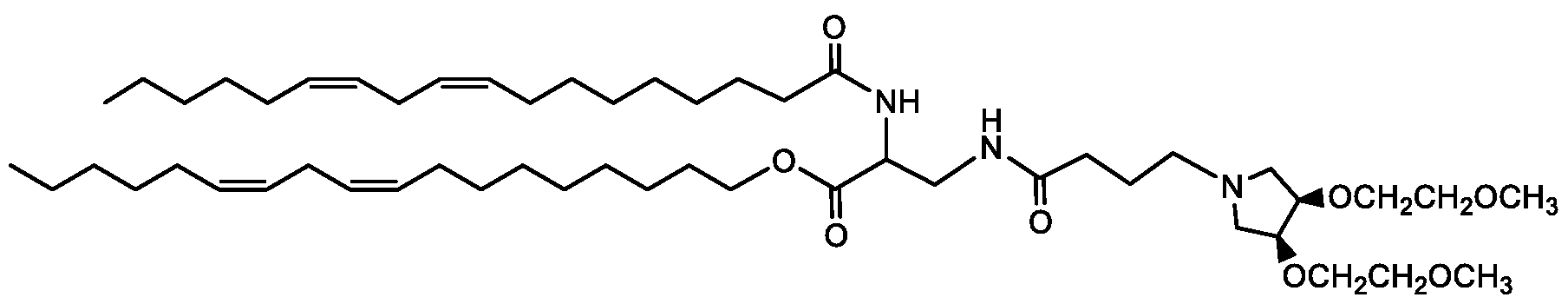
- RQBKDUUZJWNOFT-QYCRHRGJSA-N CCCCC/C=C\C/C=C\CCCCCCCC(OCCCC(CCOC(CCCCCCC/C=C\C/C=C\CCCCC)=O)NC(OCCOC)=O)=O Chemical compound CCCCC/C=C\C/C=C\CCCCCCCC(OCCCC(CCOC(CCCCCCC/C=C\C/C=C\CCCCC)=O)NC(OCCOC)=O)=O RQBKDUUZJWNOFT-QYCRHRGJSA-N 0.000 description 1
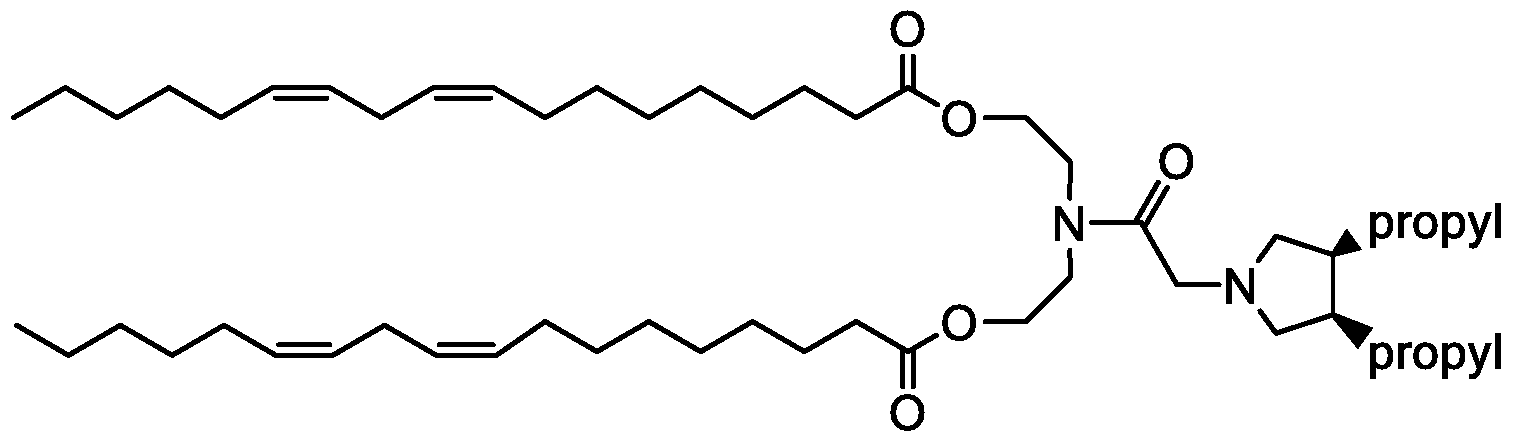
- QDQFKBYRBLDWTM-KWXKLSQISA-N CCCCC/C=C\C/C=C\CCCCCCCCOC(C(CC(C1)OC(CCN2CC(C)CC(C)C2)=O)N1C(CCCCCCC/C=C\C/C=C\CCCCC)=O)=O Chemical compound CCCCC/C=C\C/C=C\CCCCCCCCOC(C(CC(C1)OC(CCN2CC(C)CC(C)C2)=O)N1C(CCCCCCC/C=C\C/C=C\CCCCC)=O)=O QDQFKBYRBLDWTM-KWXKLSQISA-N 0.000 description 1
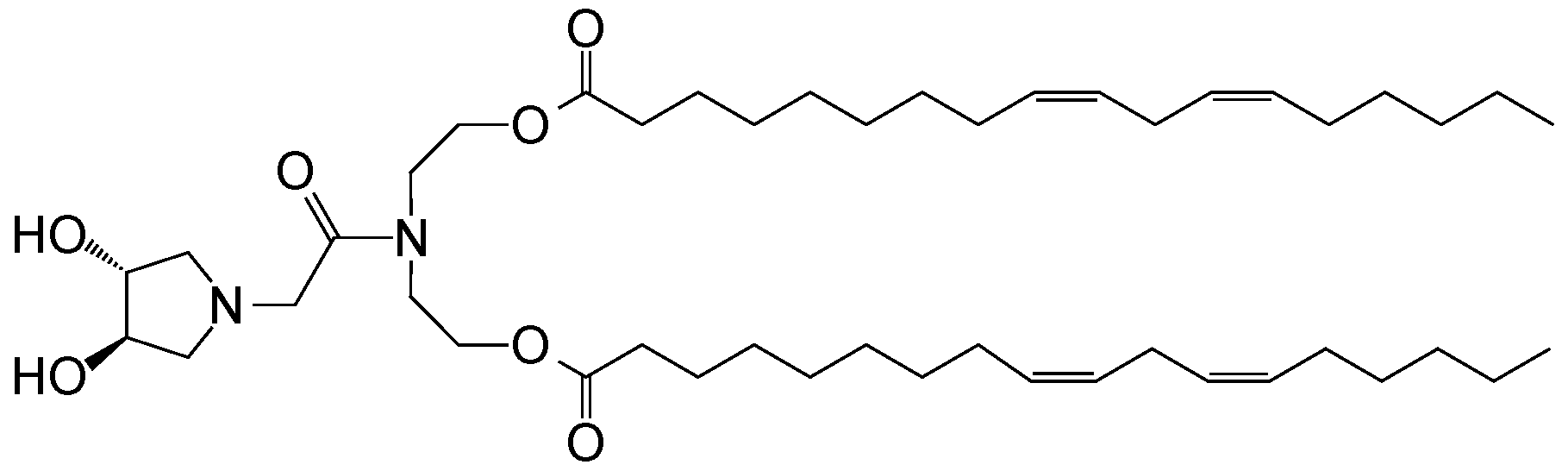
- GUYLNFCIGHADDD-MAZCIEHSSA-N CCCCC/C=C\C/C=C\CCCCCCCCOC(C(CNC(CCCCCCC/C=C\C/C=C\CCCCC)=O)NC(CCN(CC1O)CC1O)=O)=O Chemical compound CCCCC/C=C\C/C=C\CCCCCCCCOC(C(CNC(CCCCCCC/C=C\C/C=C\CCCCC)=O)NC(CCN(CC1O)CC1O)=O)=O GUYLNFCIGHADDD-MAZCIEHSSA-N 0.000 description 1
- MKTFPRNYKDVEFF-KWXKLSQISA-N CCCCC/C=C\C/C=C\CCCCCCCCOC(C(CNC(CCCCN(C)C)=O)NC(CCCCCCC/C=C\C/C=C\CCCCC)=O)=O Chemical compound CCCCC/C=C\C/C=C\CCCCCCCCOC(C(CNC(CCCCN(C)C)=O)NC(CCCCCCC/C=C\C/C=C\CCCCC)=O)=O MKTFPRNYKDVEFF-KWXKLSQISA-N 0.000 description 1
- GGSSKPBNWLWWLZ-MAZCIEHSSA-N CCCCC/C=C\C/C=C\CCCCCCCCOC(C(CNC(CN=O)=O)NC(CCCCCCC/C=C\C/C=C\CCCCC)=O)=O Chemical compound CCCCC/C=C\C/C=C\CCCCCCCCOC(C(CNC(CN=O)=O)NC(CCCCCCC/C=C\C/C=C\CCCCC)=O)=O GGSSKPBNWLWWLZ-MAZCIEHSSA-N 0.000 description 1
- GYDJFZKGYABAIQ-GZEXZTTFSA-N CCCCC/C=C\C/C=C\CCCCCCCCOC(CC(CC(OCCCCCCCC/C=C\C/C=C\CCCCC)=O)NC(CN(C[C@H]1O)C[C@@H]1O)=O)=O Chemical compound CCCCC/C=C\C/C=C\CCCCCCCCOC(CC(CC(OCCCCCCCC/C=C\C/C=C\CCCCC)=O)NC(CN(C[C@H]1O)C[C@@H]1O)=O)=O GYDJFZKGYABAIQ-GZEXZTTFSA-N 0.000 description 1
- ZQHOJWJVTHGFMP-MAZCIEHSSA-N CCCCC/C=C\C/C=C\CCCCCCCCOC(CC(CC(OCCCCCCCC/C=C\C/C=C\CCCCC)=O)NC(CN1CCN(CCO)CC1)=O)=O Chemical compound CCCCC/C=C\C/C=C\CCCCCCCCOC(CC(CC(OCCCCCCCC/C=C\C/C=C\CCCCC)=O)NC(CN1CCN(CCO)CC1)=O)=O ZQHOJWJVTHGFMP-MAZCIEHSSA-N 0.000 description 1
- DNTOSKGWCLUKBB-UHFFFAOYSA-N CCCCCCCCCCCCCCCCCCOC(CC(CC(OCCCCCCCCCCCCCCCCCC)=O)NC(CN(CC(C1)O)CC1O)=O)=O Chemical compound CCCCCCCCCCCCCCCCCCOC(CC(CC(OCCCCCCCCCCCCCCCCCC)=O)NC(CN(CC(C1)O)CC1O)=O)=O DNTOSKGWCLUKBB-UHFFFAOYSA-N 0.000 description 1
- LJGMGXXCKVFFIS-UHFFFAOYSA-N CE(10:0) Chemical compound C12CCC3(C)C(C(C)CCCC(C)C)CCC3C2CC=C2C1(C)CCC(OC(=O)CCCCCCCCC)C2 LJGMGXXCKVFFIS-UHFFFAOYSA-N 0.000 description 1
- SXDPQGRNHSLZPC-ZJNRKIDTSA-N CNC(=O)C[C@@H]1C[C@H]([C@@H](CN1)NC(=O)c1cc(c(Cl)o1)-c1c(Cl)cnn1C)c1ccc(F)c(F)c1 Chemical compound CNC(=O)C[C@@H]1C[C@H]([C@@H](CN1)NC(=O)c1cc(c(Cl)o1)-c1c(Cl)cnn1C)c1ccc(F)c(F)c1 SXDPQGRNHSLZPC-ZJNRKIDTSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- SGNBVLSWZMBQTH-FGAXOLDCSA-N Campesterol Natural products O[C@@H]1CC=2[C@@](C)([C@@H]3[C@H]([C@H]4[C@@](C)([C@H]([C@H](CC[C@H](C(C)C)C)C)CC4)CC3)CC=2)CC1 SGNBVLSWZMBQTH-FGAXOLDCSA-N 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 229920000623 Cellulose acetate phthalate Polymers 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical group [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- VGCXGMAHQTYDJK-UHFFFAOYSA-N Chloroacetyl chloride Chemical compound ClCC(Cl)=O VGCXGMAHQTYDJK-UHFFFAOYSA-N 0.000 description 1
- RMLFYKFCGMSLTB-ZBDFTZOCSA-N Cholesteryl laurate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCCCCCC)C1 RMLFYKFCGMSLTB-ZBDFTZOCSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- UCTLRSWJYQTBFZ-UHFFFAOYSA-N Dehydrocholesterol Natural products C1C(O)CCC2(C)C(CCC3(C(C(C)CCCC(C)C)CCC33)C)C3=CC=C21 UCTLRSWJYQTBFZ-UHFFFAOYSA-N 0.000 description 1
- MCWVPSBQQXUCTB-AMOSEXRZSA-N Delta7-Avenasterol Natural products CC=C(CC[C@@H](C)[C@H]1CC[C@H]2C3=CC[C@@H]4C[C@@H](O)CC[C@]4(C)[C@H]3CC[C@]12C)C(C)C MCWVPSBQQXUCTB-AMOSEXRZSA-N 0.000 description 1
- BDCFUHIWJODVNG-UHFFFAOYSA-N Desmosterol Natural products C1C=C2CC(O)C=CC2(C)C2C1C1CCC(C(C)CCC(CC)C(C)C)C1(C)CC2 BDCFUHIWJODVNG-UHFFFAOYSA-N 0.000 description 1
- GZDFHIJNHHMENY-UHFFFAOYSA-N Dimethyl dicarbonate Chemical compound COC(=O)OC(=O)OC GZDFHIJNHHMENY-UHFFFAOYSA-N 0.000 description 1
- DNVPQKQSNYMLRS-NXVQYWJNSA-N Ergosterol Natural products CC(C)[C@@H](C)C=C[C@H](C)[C@H]1CC[C@H]2C3=CC=C4C[C@@H](O)CC[C@]4(C)[C@@H]3CC[C@]12C DNVPQKQSNYMLRS-NXVQYWJNSA-N 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- BKLIAINBCQPSOV-UHFFFAOYSA-N Gluanol Natural products CC(C)CC=CC(C)C1CCC2(C)C3=C(CCC12C)C4(C)CCC(O)C(C)(C)C4CC3 BKLIAINBCQPSOV-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- ZWZWYGMENQVNFU-UHFFFAOYSA-N Glycerophosphorylserin Natural products OC(=O)C(N)COP(O)(=O)OCC(O)CO ZWZWYGMENQVNFU-UHFFFAOYSA-N 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- BTEISVKTSQLKST-UHFFFAOYSA-N Haliclonasterol Natural products CC(C=CC(C)C(C)(C)C)C1CCC2C3=CC=C4CC(O)CCC4(C)C3CCC12C BTEISVKTSQLKST-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- LOPKHWOTGJIQLC-UHFFFAOYSA-N Lanosterol Natural products CC(CCC=C(C)C)C1CCC2(C)C3=C(CCC12C)C4(C)CCC(C)(O)C(C)(C)C4CC3 LOPKHWOTGJIQLC-UHFFFAOYSA-N 0.000 description 1
- NAACPBBQTFFYQB-UHFFFAOYSA-N Linolsaeure-cholesterylester Natural products C12CCC3(C)C(C(C)CCCC(C)C)CCC3C2CC=C2C1(C)CCC(OC(=O)CCCCCCCC=CCC=CCCCCC)C2 NAACPBBQTFFYQB-UHFFFAOYSA-N 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- JMOXSQYGVIXBBZ-UHFFFAOYSA-N N,N-dimethyl-beta-alanine Chemical compound CN(C)CCC(O)=O JMOXSQYGVIXBBZ-UHFFFAOYSA-N 0.000 description 1
- CAHGCLMLTWQZNJ-UHFFFAOYSA-N Nerifoliol Natural products CC12CCC(O)C(C)(C)C1CCC1=C2CCC2(C)C(C(CCC=C(C)C)C)CCC21C CAHGCLMLTWQZNJ-UHFFFAOYSA-N 0.000 description 1
- RJECHNNFRHZQKU-UHFFFAOYSA-N Oelsaeurecholesterylester Natural products C12CCC3(C)C(C(C)CCCC(C)C)CCC3C2CC=C2C1(C)CCC(OC(=O)CCCCCCCC=CCCCCCCCC)C2 RJECHNNFRHZQKU-UHFFFAOYSA-N 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- BBJQPKLGPMQWBU-UHFFFAOYSA-N Palmitinsaeurecholesterylester Natural products C12CCC3(C)C(C(C)CCCC(C)C)CCC3C2CC=C2C1(C)CCC(OC(=O)CCCCCCCCCCCCCCC)C2 BBJQPKLGPMQWBU-UHFFFAOYSA-N 0.000 description 1
- 235000019483 Peanut oil Nutrition 0.000 description 1
- LGJMUZUPVCAVPU-JFBKYFIKSA-N Sitostanol Natural products O[C@@H]1C[C@H]2[C@@](C)([C@@H]3[C@@H]([C@H]4[C@@](C)([C@@H]([C@@H](CC[C@H](C(C)C)CC)C)CC4)CC3)CC2)CC1 LGJMUZUPVCAVPU-JFBKYFIKSA-N 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- OKJPEAGHQZHRQV-UHFFFAOYSA-N Triiodomethane Natural products IC(I)I OKJPEAGHQZHRQV-UHFFFAOYSA-N 0.000 description 1
- OILXMJHPFNGGTO-ZRUUVFCLSA-N UNPD197407 Natural products C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)C=C[C@H](C)C(C)C)[C@@]1(C)CC2 OILXMJHPFNGGTO-ZRUUVFCLSA-N 0.000 description 1
- UJELMAYUQSGICC-UHFFFAOYSA-N Zymosterol Natural products CC12CCC(O)CC1CCC1=C2CCC2(C)C(C(C)C=CCC(C)C)CCC21 UJELMAYUQSGICC-UHFFFAOYSA-N 0.000 description 1
- CWRILEGKIAOYKP-SSDOTTSWSA-M [(2r)-3-acetyloxy-2-hydroxypropyl] 2-aminoethyl phosphate Chemical compound CC(=O)OC[C@@H](O)COP([O-])(=O)OCCN CWRILEGKIAOYKP-SSDOTTSWSA-M 0.000 description 1
- PUKCXSHDVCMMAN-ATRPORLUSA-N [(3s,8s,9s,10r,13r,14s,17r)-10,13-dimethyl-17-[(2r)-6-methylheptan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3-yl] (e)-tetracos-15-enoate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCCCCCCCC/C=C/CCCCCCCC)C1 PUKCXSHDVCMMAN-ATRPORLUSA-N 0.000 description 1
- CKDZWMVGDHGMFR-GTPODGLVSA-N [(3s,8s,9s,10r,13r,14s,17r)-10,13-dimethyl-17-[(2r)-6-methylheptan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3-yl] butanoate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCC)C1 CKDZWMVGDHGMFR-GTPODGLVSA-N 0.000 description 1
- KXWDMNPRHKRGKB-DYQRUOQXSA-N [(3s,8s,9s,10r,13r,14s,17r)-10,13-dimethyl-17-[(2r)-6-methylheptan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3-yl] heptanoate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCC)C1 KXWDMNPRHKRGKB-DYQRUOQXSA-N 0.000 description 1
- FPBODWXATDKICU-FLFWOSPYSA-N [(3s,8s,9s,10r,13r,14s,17r)-10,13-dimethyl-17-[(2r)-6-methylheptan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3-yl] hexanoate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCC)C1 FPBODWXATDKICU-FLFWOSPYSA-N 0.000 description 1
- SKLBBRQPVZDTNM-SJTWHRLHSA-N [(3s,8s,9s,10r,13r,14s,17r)-10,13-dimethyl-17-[(2r)-6-methylheptan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3-yl] octanoate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCC)C1 SKLBBRQPVZDTNM-SJTWHRLHSA-N 0.000 description 1
- QZENWXOYJAHXJF-LWOXEAPXSA-N [(9Z,12Z)-octadeca-9,12-dienyl] (2S)-3-[[2-(dimethylamino)acetyl]amino]-2-[[(9Z,12Z)-octadeca-9,12-dienoyl]amino]propanoate Chemical compound CN(CC(=O)NC[C@@H](C(=O)OCCCCCCCC\C=C/C\C=C/CCCCC)NC(CCCCCCC\C=C/C\C=C/CCCCC)=O)C QZENWXOYJAHXJF-LWOXEAPXSA-N 0.000 description 1
- NAACPBBQTFFYQB-TVYVBBRWSA-N [10,13-dimethyl-17-(6-methylheptan-2-yl)-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3-yl] (9e,12e)-octadeca-9,12-dienoate Chemical compound C12CCC3(C)C(C(C)CCCC(C)C)CCC3C2CC=C2C1(C)CCC(OC(=O)CCCCCCC/C=C/C/C=C/CCCCC)C2 NAACPBBQTFFYQB-TVYVBBRWSA-N 0.000 description 1
- AOVFSSLGBNEDAD-KWXKLSQISA-O [2-[[1-[(9Z,12Z)-heptadeca-9,12-dienoxy]-5-[(9Z,12Z)-octadeca-9,12-dienoxy]-1,5-dioxopentan-3-yl]amino]-2-oxoethyl]-trimethylazanium Chemical compound C(CCCCCCC\C=C/C\C=C/CCCC)OC(CC(CC(=O)OCCCCCCCC\C=C/C\C=C/CCCCC)NC(C[N+](C)(C)C)=O)=O AOVFSSLGBNEDAD-KWXKLSQISA-O 0.000 description 1
- YKTSYUJCYHOUJP-UHFFFAOYSA-N [O--].[Al+3].[Al+3].[O-][Si]([O-])([O-])[O-] Chemical compound [O--].[Al+3].[Al+3].[O-][Si]([O-])([O-])[O-] YKTSYUJCYHOUJP-UHFFFAOYSA-N 0.000 description 1
- 150000003855 acyl compounds Chemical class 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229910001413 alkali metal ion Inorganic materials 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 150000001441 androstanes Chemical class 0.000 description 1
- 238000005349 anion exchange Methods 0.000 description 1
- 238000000149 argon plasma sintering Methods 0.000 description 1
- 125000003710 aryl alkyl group Chemical group 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- MCWVPSBQQXUCTB-OQTIOYDCSA-N avenasterol Chemical compound C1[C@@H](O)CC[C@]2(C)[C@@H](CC[C@@]3([C@@H]([C@H](C)CC/C(=C/C)C(C)C)CC[C@H]33)C)C3=CC[C@H]21 MCWVPSBQQXUCTB-OQTIOYDCSA-N 0.000 description 1
- GNVWVYIAQBJHGV-UHFFFAOYSA-N azetidin-3-ylmethanol Chemical compound OCC1CNC1 GNVWVYIAQBJHGV-UHFFFAOYSA-N 0.000 description 1
- 229940076810 beta sitosterol Drugs 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- NJKOMDUNNDKEAI-UHFFFAOYSA-N beta-sitosterol Natural products CCC(CCC(C)C1CCC2(C)C3CC=C4CC(O)CCC4C3CCC12C)C(C)C NJKOMDUNNDKEAI-UHFFFAOYSA-N 0.000 description 1
- YLRGSCHIWGSMIK-KWXKLSQISA-N bis[(9Z,12Z)-octadeca-9,12-dienyl] 3-[2-(dimethylamino)ethoxycarbonylamino]pentanedioate Chemical compound CN(CCOC(=O)NC(CC(=O)OCCCCCCCC\C=C/C\C=C/CCCCC)CC(=O)OCCCCCCCC\C=C/C\C=C/CCCCC)C YLRGSCHIWGSMIK-KWXKLSQISA-N 0.000 description 1
- YATJIJSEVWHILF-KWXKLSQISA-N bis[(9Z,12Z)-octadeca-9,12-dienyl] 3-[2-(dimethylamino)ethylsulfanylcarbonylamino]pentanedioate Chemical compound CN(CCSC(=O)NC(CC(=O)OCCCCCCCC\C=C/C\C=C/CCCCC)CC(=O)OCCCCCCCC\C=C/C\C=C/CCCCC)C YATJIJSEVWHILF-KWXKLSQISA-N 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- OILXMJHPFNGGTO-ZAUYPBDWSA-N brassicasterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)/C=C/[C@H](C)C(C)C)[C@@]1(C)CC2 OILXMJHPFNGGTO-ZAUYPBDWSA-N 0.000 description 1
- 235000004420 brassicasterol Nutrition 0.000 description 1
- 239000012267 brine Substances 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- FUFJGUQYACFECW-UHFFFAOYSA-L calcium hydrogenphosphate Chemical compound [Ca+2].OP([O-])([O-])=O FUFJGUQYACFECW-UHFFFAOYSA-L 0.000 description 1
- 150000001700 campestanes Chemical class 0.000 description 1
- SGNBVLSWZMBQTH-PODYLUTMSA-N campesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CC[C@@H](C)C(C)C)[C@@]1(C)CC2 SGNBVLSWZMBQTH-PODYLUTMSA-N 0.000 description 1
- 235000000431 campesterol Nutrition 0.000 description 1
- 125000006358 carbonyl ethylene group Chemical group [H]C([H])([*:2])C([H])([H])C([*:1])=O 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 229940081734 cellulose acetate phthalate Drugs 0.000 description 1
- 229940106189 ceramide Drugs 0.000 description 1
- 150000001829 cholanes Chemical class 0.000 description 1
- XIIAYQZJNBULGD-LDHZKLTISA-N cholestane Chemical compound C1CC2CCCC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 XIIAYQZJNBULGD-LDHZKLTISA-N 0.000 description 1
- 150000001838 cholestanes Chemical class 0.000 description 1
- XHRPOTDGOASDJS-UHFFFAOYSA-N cholesterol n-octadecanoate Natural products C12CCC3(C)C(C(C)CCCC(C)C)CCC3C2CC=C2C1(C)CCC(OC(=O)CCCCCCCCCCCCCCCCC)C2 XHRPOTDGOASDJS-UHFFFAOYSA-N 0.000 description 1
- XUGISPSHIFXEHZ-VEVYEIKRSA-N cholesteryl acetate Chemical compound C1C=C2C[C@@H](OC(C)=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 XUGISPSHIFXEHZ-VEVYEIKRSA-N 0.000 description 1
- WBOQXYUYHINMOC-FTAWAYKBSA-N cholesteryl behenate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCCCCCCCCCCCCCCCC)C1 WBOQXYUYHINMOC-FTAWAYKBSA-N 0.000 description 1
- RJECHNNFRHZQKU-WYIFMRBMSA-N cholesteryl elaidate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCC/C=C/CCCCCCCC)C1 RJECHNNFRHZQKU-WYIFMRBMSA-N 0.000 description 1
- WCLNGBQPTVENHV-MKQVXYPISA-N cholesteryl nonanoate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCCC)C1 WCLNGBQPTVENHV-MKQVXYPISA-N 0.000 description 1
- NAACPBBQTFFYQB-XNTGVSEISA-N cholesteryl octadeca-9,12-dienoate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCCC=CCC=CCCCCC)C1 NAACPBBQTFFYQB-XNTGVSEISA-N 0.000 description 1
- RJECHNNFRHZQKU-RMUVNZEASA-N cholesteryl oleate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCC\C=C/CCCCCCCC)C1 RJECHNNFRHZQKU-RMUVNZEASA-N 0.000 description 1
- BBJQPKLGPMQWBU-JADYGXMDSA-N cholesteryl palmitate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCCCCCCCCCC)C1 BBJQPKLGPMQWBU-JADYGXMDSA-N 0.000 description 1
- XHRPOTDGOASDJS-XNTGVSEISA-N cholesteryl stearate Chemical compound C([C@@H]12)C[C@]3(C)[C@@H]([C@H](C)CCCC(C)C)CC[C@H]3[C@@H]1CC=C1[C@]2(C)CC[C@H](OC(=O)CCCCCCCCCCCCCCCCC)C1 XHRPOTDGOASDJS-XNTGVSEISA-N 0.000 description 1
- 208000019425 cirrhosis of liver Diseases 0.000 description 1
- 239000003240 coconut oil Substances 0.000 description 1
- 235000019864 coconut oil Nutrition 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 150000001928 cycloartanes Chemical class 0.000 description 1
- DEZRYPDIMOWBDS-UHFFFAOYSA-N dcm dichloromethane Chemical compound ClCCl.ClCCl DEZRYPDIMOWBDS-UHFFFAOYSA-N 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- OQMZNAMGEHIHNN-CIFIHVIMSA-N delta7-stigmasterol Chemical compound C1[C@@H](O)CC[C@]2(C)[C@@H](CC[C@@]3([C@@H]([C@H](C)/C=C/[C@@H](CC)C(C)C)CC[C@H]33)C)C3=CC=C21 OQMZNAMGEHIHNN-CIFIHVIMSA-N 0.000 description 1
- AVSXSVCZWQODGV-DPAQBDIFSA-N desmosterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@@H](CCC=C(C)C)C)[C@@]1(C)CC2 AVSXSVCZWQODGV-DPAQBDIFSA-N 0.000 description 1
- 235000019700 dicalcium phosphate Nutrition 0.000 description 1
- IUNMPGNGSSIWFP-UHFFFAOYSA-N dimethylaminopropylamine Chemical compound CN(C)CCCN IUNMPGNGSSIWFP-UHFFFAOYSA-N 0.000 description 1
- CETRZFQIITUQQL-UHFFFAOYSA-N dmso dimethylsulfoxide Chemical compound CS(C)=O.CS(C)=O CETRZFQIITUQQL-UHFFFAOYSA-N 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 150000002133 ergostanes Chemical class 0.000 description 1
- DNVPQKQSNYMLRS-SOWFXMKYSA-N ergosterol Chemical compound C1[C@@H](O)CC[C@]2(C)[C@H](CC[C@]3([C@H]([C@H](C)/C=C/[C@@H](C)C(C)C)CC[C@H]33)C)C3=CC=C21 DNVPQKQSNYMLRS-SOWFXMKYSA-N 0.000 description 1
- 150000002162 estranes Chemical class 0.000 description 1
- OJCSPXHYDFONPU-UHFFFAOYSA-N etoac etoac Chemical compound CCOC(C)=O.CCOC(C)=O OJCSPXHYDFONPU-UHFFFAOYSA-N 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 238000000105 evaporative light scattering detection Methods 0.000 description 1
- 210000001508 eye Anatomy 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- FAQVCWVVIYYWRR-UHFFFAOYSA-N glutaminyl-alanine Chemical compound OC(=O)C(C)NC(=O)C(N)CCC(N)=O FAQVCWVVIYYWRR-UHFFFAOYSA-N 0.000 description 1
- VANNPISTIUFMLH-UHFFFAOYSA-N glutaric anhydride Chemical compound O=C1CCCC(=O)O1 VANNPISTIUFMLH-UHFFFAOYSA-N 0.000 description 1
- JEJLGIQLPYYGEE-UHFFFAOYSA-N glycerol dipalmitate Natural products CCCCCCCCCCCCCCCC(=O)OCC(CO)OC(=O)CCCCCCCCCCCCCCC JEJLGIQLPYYGEE-UHFFFAOYSA-N 0.000 description 1
- 150000003804 gonanes Chemical class 0.000 description 1
- 150000002347 gorgostanes Chemical class 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- IKGLACJFEHSFNN-UHFFFAOYSA-N hydron;triethylazanium;trifluoride Chemical compound F.F.F.CCN(CC)CC IKGLACJFEHSFNN-UHFFFAOYSA-N 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 210000000936 intestine Anatomy 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 229940058690 lanosterol Drugs 0.000 description 1
- CAHGCLMLTWQZNJ-RGEKOYMOSA-N lanosterol Chemical compound C([C@]12C)C[C@@H](O)C(C)(C)[C@H]1CCC1=C2CC[C@]2(C)[C@H]([C@H](CCC=C(C)C)C)CC[C@@]21C CAHGCLMLTWQZNJ-RGEKOYMOSA-N 0.000 description 1
- 125000002669 linoleoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C([H])=C([H])\C([H])([H])/C([H])=C([H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- CSNNHWWHGAXBCP-UHFFFAOYSA-L magnesium sulphate Substances [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 230000007721 medicinal effect Effects 0.000 description 1
- COTNUBDHGSIOTA-UHFFFAOYSA-N meoh methanol Chemical compound OC.OC COTNUBDHGSIOTA-UHFFFAOYSA-N 0.000 description 1
- GDJLJNFNXINTHS-RGMNGODLSA-N methyl (2s)-2-amino-3-[(2-methylpropan-2-yl)oxycarbonylamino]propanoate;hydrochloride Chemical compound Cl.COC(=O)[C@@H](N)CNC(=O)OC(C)(C)C GDJLJNFNXINTHS-RGMNGODLSA-N 0.000 description 1
- 125000006362 methylene amino carbonyl group Chemical group [H]N(C([*:2])=O)C([H])([H])[*:1] 0.000 description 1
- 239000000693 micelle Substances 0.000 description 1
- WOOWBQQQJXZGIE-UHFFFAOYSA-N n-ethyl-n-propan-2-ylpropan-2-amine Chemical compound CCN(C(C)C)C(C)C.CCN(C(C)C)C(C)C WOOWBQQQJXZGIE-UHFFFAOYSA-N 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000000312 peanut oil Substances 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- NKZVILXYMBJHBC-UHFFFAOYSA-J pentacalcium hydroxide phosphate Chemical compound P(=O)([O-])([O-])[O-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[OH-].[Ca+2] NKZVILXYMBJHBC-UHFFFAOYSA-J 0.000 description 1
- 239000005426 pharmaceutical component Substances 0.000 description 1
- 239000008024 pharmaceutical diluent Substances 0.000 description 1
- 150000008105 phosphatidylcholines Chemical class 0.000 description 1
- 150000003905 phosphatidylinositols Chemical class 0.000 description 1
- LFGREXWGYUGZLY-UHFFFAOYSA-N phosphoryl Chemical group [P]=O LFGREXWGYUGZLY-UHFFFAOYSA-N 0.000 description 1
- 229940068065 phytosterols Drugs 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 150000003105 poriferastanes Chemical class 0.000 description 1
- 231100000683 possible toxicity Toxicity 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 150000003128 pregnanes Chemical class 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- CJIVCSBYIRBJSG-UHFFFAOYSA-N pyrrolidine-3,3-diol Chemical compound OC1(O)CCNC1 CJIVCSBYIRBJSG-UHFFFAOYSA-N 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- KZJWDPNRJALLNS-VJSFXXLFSA-N sitosterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CC[C@@H](CC)C(C)C)[C@@]1(C)CC2 KZJWDPNRJALLNS-VJSFXXLFSA-N 0.000 description 1
- 229950005143 sitosterol Drugs 0.000 description 1
- NLQLSVXGSXCXFE-UHFFFAOYSA-N sitosterol Natural products CC=C(/CCC(C)C1CC2C3=CCC4C(C)C(O)CCC4(C)C3CCC2(C)C1)C(C)C NLQLSVXGSXCXFE-UHFFFAOYSA-N 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- 229940126586 small molecule drug Drugs 0.000 description 1
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 1
- 239000004299 sodium benzoate Substances 0.000 description 1
- 235000010234 sodium benzoate Nutrition 0.000 description 1
- BEOOHQFXGBMRKU-UHFFFAOYSA-N sodium cyanoborohydride Chemical compound [Na+].[B-]C#N BEOOHQFXGBMRKU-UHFFFAOYSA-N 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L sodium sulphate Substances [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 150000003434 stigmastanes Chemical class 0.000 description 1
- LGJMUZUPVCAVPU-HRJGVYIJSA-N stigmastanol Chemical compound C([C@@H]1CC2)[C@@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@H](C)CC[C@@H](CC)C(C)C)[C@@]2(C)CC1 LGJMUZUPVCAVPU-HRJGVYIJSA-N 0.000 description 1
- 229940014800 succinic anhydride Drugs 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 125000001981 tert-butyldimethylsilyl group Chemical group [H]C([H])([H])[Si]([H])(C([H])([H])[H])[*]C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- WHRNULOCNSKMGB-UHFFFAOYSA-N tetrahydrofuran thf Chemical compound C1CCOC1.C1CCOC1 WHRNULOCNSKMGB-UHFFFAOYSA-N 0.000 description 1
- WROMPOXWARCANT-UHFFFAOYSA-N tfa trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.OC(=O)C(F)(F)F WROMPOXWARCANT-UHFFFAOYSA-N 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- VSWJMEOTFCYQNI-ZPPAUJSGSA-N trimethyl-[3-[[(9Z,12Z)-octadeca-9,12-dienoyl]-[2-[(9Z,12Z)-octadeca-9,12-dienoyl]oxyethyl]amino]propyl]azanium Chemical compound C[N+](CCCN(C(CCCCCCC\C=C/C\C=C/CCCCC)=O)CCOC(CCCCCCC\C=C/C\C=C/CCCCC)=O)(C)C VSWJMEOTFCYQNI-ZPPAUJSGSA-N 0.000 description 1
- WBIJWCAGCBRBMS-UHFFFAOYSA-N trimethyl-[3-[tetradecanoyl(2-tetradecanoyloxyethyl)amino]propyl]azanium Chemical compound C[N+](CCCN(C(CCCCCCCCCCCCC)=O)CCOC(CCCCCCCCCCCCC)=O)(C)C WBIJWCAGCBRBMS-UHFFFAOYSA-N 0.000 description 1
- QYSXJUFSXHHAJI-YRZJJWOYSA-N vitamin D3 Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C\C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-YRZJJWOYSA-N 0.000 description 1
- CGSJXLIKVBJVRY-XTGBIJOFSA-N zymosterol Chemical compound C([C@@]12C)C[C@H](O)C[C@@H]1CCC1=C2CC[C@]2(C)[C@@H]([C@@H](CCC=C(C)C)C)CC[C@H]21 CGSJXLIKVBJVRY-XTGBIJOFSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C271/00—Derivatives of carbamic acids, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups
- C07C271/06—Esters of carbamic acids
- C07C271/08—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms
- C07C271/10—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atoms of the carbamate groups bound to hydrogen atoms or to acyclic carbon atoms
- C07C271/16—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atoms of the carbamate groups bound to hydrogen atoms or to acyclic carbon atoms to carbon atoms of hydrocarbon radicals substituted by singly-bound oxygen atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Liposomes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/7105—Natural ribonucleic acids, i.e. containing only riboses attached to adenine, guanine, cytosine or uracil and having 3'-5' phosphodiester links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/16—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing nitrogen, e.g. nitro-, nitroso-, azo-compounds, nitriles, cyanates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/22—Heterocyclic compounds, e.g. ascorbic acid, tocopherol or pyrrolidones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/24—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing atoms other than carbon, hydrogen, oxygen, halogen, nitrogen or sulfur, e.g. cyclomethicone or phospholipids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/51—Nanocapsules; Nanoparticles
- A61K9/5107—Excipients; Inactive ingredients
- A61K9/5123—Organic compounds, e.g. fats, sugars
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C237/00—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups
- C07C237/02—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the carbon atoms of the carboxamide groups bound to acyclic carbon atoms of the carbon skeleton
- C07C237/04—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the carbon atoms of the carboxamide groups bound to acyclic carbon atoms of the carbon skeleton the carbon skeleton being acyclic and saturated
- C07C237/08—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the carbon atoms of the carboxamide groups bound to acyclic carbon atoms of the carbon skeleton the carbon skeleton being acyclic and saturated having the nitrogen atom of at least one of the carboxamide groups bound to an acyclic carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C237/00—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups
- C07C237/02—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the carbon atoms of the carboxamide groups bound to acyclic carbon atoms of the carbon skeleton
- C07C237/16—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the carbon atoms of the carboxamide groups bound to acyclic carbon atoms of the carbon skeleton the carbon skeleton being acyclic and unsaturated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C237/00—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups
- C07C237/50—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the nitrogen atom of at least one of the carboxamide groups quaternised
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D205/00—Heterocyclic compounds containing four-membered rings with one nitrogen atom as the only ring hetero atom
- C07D205/02—Heterocyclic compounds containing four-membered rings with one nitrogen atom as the only ring hetero atom not condensed with other rings
- C07D205/04—Heterocyclic compounds containing four-membered rings with one nitrogen atom as the only ring hetero atom not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/04—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D207/08—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon radicals, substituted by hetero atoms, attached to ring carbon atoms
- C07D207/09—Radicals substituted by nitrogen atoms, not forming part of a nitro radical
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/04—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D207/10—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D207/12—Oxygen or sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/68—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having one double bond between ring members or between a ring member and a non-ring member
- C07D211/72—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having one double bond between ring members or between a ring member and a non-ring member with hetero atoms or with carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D211/74—Oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/80—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D211/82—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/80—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D211/84—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen directly attached to ring carbon atoms
- C07D211/86—Oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/06—Phosphorus compounds without P—C bonds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/06—Phosphorus compounds without P—C bonds
- C07F9/08—Esters of oxyacids of phosphorus
- C07F9/09—Esters of phosphoric acids
- C07F9/10—Phosphatides, e.g. lecithin
- C07F9/106—Adducts, complexes, salts of phosphatides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2300/00—Mixtures or combinations of active ingredients, wherein at least one active ingredient is fully defined in groups A61K31/00 - A61K41/00
Definitions
- This invention relates to the fields of biopharmaceuticals and therapeutics. More particularly, this invention relates to compounds, compositions and methods for providing nanoparticles to deliver and distribute active agents or drug compounds to cells, tissues, organs, and subjects.
- Therapeutic agents such as drug compounds, nucleic acid molecules and other active agents operate by uptake into cells, tissues, and organs of a subject.
- one way to carry out transfection of nucleic acids is to encapsulate the active molecules in a lipid nanoparticle.
- Drawbacks of this methodology include potential toxicity in various modalities of delivery, such as intravenous injection, and low rates of cell penetration.
- nanoparticles for active agents There is a continuing need for lipid-like molecules and compositions for efficient transfection and distribution of nucleic acid molecules and other agents to cells and subjects.
- This invention relates to molecules and compositions thereof for use in biopharmaceuticals and therapeutics. More particularly, this invention relates to compounds, compositions and methods for providing nanoparticles to deliver and distribute active agents or drug compounds to cells, tissues, organs, and subjects.
- This invention provides a range of ionizable compounds.
- the ionizable compounds of this invention can be used to form nanoparticles to deliver and distribute active agents.
- Embodiments of this invention include a broad range of compounds having lipid-like or liposome-forming properties.
- a compound may have the structure shown in
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, and aminoalkyl;
- each R 7 is independently selected from H, alkyl, and hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- Q is O or R 7 ;
- p is from 1 to 4.
- a compound can have the structure shown in
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is a C( 12-20) alkyl group or a C( 12-20) alkenyl group that is substituted with a carboxylic acid or ester group.
- a compound may have the structure shown in
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from alkyl, hydroxyalkyl, alkoxyalkoxy, and carboxyalkyl;
- R 6 is selected from R 7 2 , N + HR 7 2 and N + R 7 3 ;
- R 7 is selected from H, alkyl, hydroxyalkyl.
- a compound of this invention may have the structure shown in Formula
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- Q is O or R 7 ;
- p is from 1 to 4.
- a compound can have the structure shown in
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- Z is S or O
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- p is from 1 to 4.
- a compound may have the structure shown in Formula
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- p is from 1 to 4.
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- a com ound of this invention can have the structure shown in Formula VI
- R 2 and R 4 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from aminoalkyl, quaternary aminoalkyl.
- a compound of this invention can have the structure shown in Formula VII
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from H, alkyl, aminoalkyl, quaternary aminoalkyl, hydroxyalkyl, alkoxyalkyl, alkoxyalkoxy alkyl.
- a compound may have the structure shown in Formula
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from
- R is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- Q is O or R 7 .
- a compound may have the structure shown in Formula
- n and m are each independently from 1 to 2;
- R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- Z is N, O;
- R 3 is selected from
- alkyl hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- p is from 1 to 4.
- a compound can have the structure shown in
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- p is from 1 to 4.
- R' is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- a compound of this disclosure can have the structure shown in Formula X Formula X
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from
- a compound may have the structure shown in
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- Z is O or H
- p is from 1 to 4.
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl; each R is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- Q is O or R 7 .
- compositions containing an ionizable compound above and a pharmaceutically acceptable carrier may contain nanoparticles.
- This disclosure includes pharmaceutical compositions comprising an ionizable compound above, an active agent, and a pharmaceutically acceptable carrier.
- the ionizable compound may be from 15 mol% to 40 mol% of the lipids of the composition.
- the composition may comprise nanoparticles.
- An active agent of this disclosure may be one or more RNAi molecules.
- An active agent may be one or more RNAi molecules selected from small interfering RNAs (siRNA), double stranded RNAs (dsRNA) that are Dicer substrates, microRNAs (miRNA), short hairpin RNAs (shRNA), DNA-directed RNAs (ddRNA), Piwi- interacting RNAs (piRNA), repeat associated siRNAs (rasiRNA), and modified forms thereof.
- siRNA small interfering RNAs
- dsRNA double stranded RNAs
- shRNA short hairpin RNAs
- ddRNA DNA-directed RNAs
- piRNA Piwi- interacting RNAs
- rasiRNA repeat associated siRNAs
- An active agent of this disclosure may be one or more active
- this invention includes compositions for use in distributing an active agent for treating a condition or disease in a subject, the composition comprising an ionizable compound above, a structural lipid, a stabilizer lipid, and a lipid for reducing immunogenicity of the composition.
- the active agent can be one or more RNAi molecules and the composition may comprise nanoparticles that encapsulate the RNAi molecules.
- compositions containing an ionizable compound and one or more pharmaceutically acceptable excipients.
- a composition of this invention can be a nanoparticle composed, at least in part, of an ionizable compound.
- Compounds of this invention can be used to make compositions for use in distributing an active agent in a subject, where the composition includes an ionizable compound.
- a composition of this invention can be used in distributing an active agent for treating a condition or disease in a subject.
- a composition for use in distributing an active agent for treating a condition or disease in a subject can include an ionizable compound, a structural lipid, and a lipid for reducing immunogenicity of the composition.
- FIG. 1 shows a scheme for the preparation of Compound A6.
- FIG. 2 shows a scheme for the preparation of Compound AB.
- FIG. 3 shows a scheme for the preparation of Compound A4.
- FIG. 4 shows a scheme for the preparation of Compound B8.
- FIG. 5 shows a scheme for the preparation of Compound A9.
- FIG. 6 shows a scheme for the preparation of Compound AA.
- FIG. 7 shows a scheme for the preparation of Compound A5.
- FIG. 8 shows a scheme for the preparation of Compound Al .
- FIG. 9 shows a scheme for the preparation of Compound D22.
- FIG. 10 shows a scheme for the preparation of Compounds A7 and A8.
- FIG. 11 shows a scheme for the preparation of Compounds C3 and C2.
- FIG. 12 shows a scheme for the preparation of Compound DD.
- FIG. 13 shows a scheme for the preparation of Compound E4.
- FIG. 14 shows a scheme for the preparation of Compound CA.
- FIG. 15 shows a scheme for the preparation of Compound Dl .
- FIG. 16 shows a scheme for the preparation of Compound D7.
- FIG. 17 shows a scheme for the preparation of Compound F6.
- FIG. 18 shows a scheme for the preparation of Compounds F5 and F7.
- FIG. 19 shows a scheme for the preparation of Compounds F8 and F9.
- FIG. 20 shows a scheme for the preparation of Compounds C25 and C24.
- FIG. 21 shows a scheme for the preparation of Compound D16.
- FIG. 22 shows a scheme for the preparation of Compound D17.
- FIG. 23 shows a scheme for the preparation of Compound D18.
- FIG. 24 shows a scheme for the preparation of Compound D19.
- FIG. 25 shows a scheme for the preparation of Compound D20.
- FIG. 26 shows a scheme for the preparation of Compound D21.
- FIG. 27 shows a scheme for the preparation of Compound E37.
- FIG. 28 shows a scheme for the preparation of Compounds E38 and E39.
- FIG. 29 shows a scheme for the preparation of Compound E40.
- FIG. 30 shows a scheme for the preparation of Compound A23.
- FIG. 31 shows a scheme for the preparation of Compound A24.
- FIG. 32 shows a scheme for the preparation of Compound A25.
- This invention provides a range of ionizable molecules that are amphiphiles with lipid-like properties.
- the ionizable compounds of this invention can be used in delivering therapeutic agents to cells, tissues or organs, organisms, and subjects.
- this invention provides compounds for forming lipid nanoparticles for encapsulating and delivering active agents such as nucleic acid molecules to cells and subjects.
- This invention can provide a composition for use in distributing an active agent in cells, tissues or organs, organisms, and subjects, where the composition includes one or more of the ionizable molecules of this invention.
- compositions of this invention may include one or more of the ionizable molecules, along with a structural lipid, and one or more lipids for reducing
- An ionizable molecule of this invention can be any mol% of a composition of this invention.
- compositions of this invention may include one or more of the ionizable molecules, along with a structural lipid, and one or more lipids for reducing
- compositions of this invention may include one or more of the ionizable molecules, along with a structural lipid, one or more stabilizer lipids, and one or more lipids for reducing immunogenicity of the composition.
- a component of a formulation can be a single compound, or can be a combination of one or more suitable lipid compounds.
- a stabilizer lipid can refer to a single stabilizer lipid, or to a combination of one or more suitable stabilizer lipids.
- the ionizable compounds of a composition of this invention can be from
- the ionizable molecules of a composition can be from 55 mol% to 65 mol% of the lipid components of the composition. In further embodiments, the ionizable molecules of a composition can be about 60 mol% of the lipid components of the composition.
- the structural lipid of a composition of this invention can be from 20 mol% to 50 mol% of the lipid components of the composition. In certain embodiments, the structural lipid of a composition can be from 35 mol% to 45 mol% of the lipid components of the composition.
- the one or more lipids for reducing immunogenicity of the composition can be from a total of 1 mol% to 8 mol% of the lipid components of the composition. In certain embodiments, the one or more lipids for reducing immunogenicity of the composition can be from a total of 1 mol% to 5 mol% of the lipid components of the composition.
- a composition of this invention can further include a cationic lipid, which can be from 5 mol% to 25 mol% of the lipid components of the composition.
- a composition of this invention can further include a cationic lipid, which can be from 5 mol% to 15 mol% of the lipid components of the composition.
- the molar ratio of the concentrations of the cationic lipid to the ionizable molecules of a composition of this invention can be from 5:80 to 25:50.
- the entirety of the lipid components may include one or more of the ionizable compound molecular components, a structural lipid, and one or more lipids for reducing immunogenicity of the composition.
- the ionizable molecules of a composition of this invention can be from 15 mol% to 40 mol% of the lipid components of the composition. In certain embodiments, the ionizable molecules of a composition can be from 20 mol% to 35 mol% of the lipid components of the composition. In further embodiments, the ionizable molecules of a composition can be from 25 mol% to 30 mol% of the lipid components of the
- the structural lipid of a composition of this invention can be from 25 mol% to 40 mol% of the lipid components of the composition. In certain embodiments, the structural lipid of a composition can be from 30 mol% to 35 mol% of the lipid components of the composition.
- the sum of the stabilizer lipids of a composition of this invention can be from 25 mol% to 40% mol% of the lipid components of the composition. In certain embodiments, the sum of the stabilizer lipids of a composition can be from 30 mol% to 40 mol% of the lipid components of the composition.
- a composition of this invention can include two or more stabilizer lipids, where each of the stabilizer lipids individually can be from 5 mol% to 35 mol% of the lipid components of the composition. In certain embodiments, a composition of this invention can include two or more stabilizer lipids, where each of the stabilizer lipids individually can be from 10 mol% to 30 mol% of the lipid components of the composition.
- the sum of the one or more stabilizer lipids can be from 25 mol% to 40 mol% of the lipids of the composition, wherein each of the stabilizer lipids individually can be from 5 mol% to 35% mol%.
- the sum of the one or more stabilizer lipids can be from 30 mol% to 40 mol% of the lipids of the composition, wherein each of the stabilizer lipids individually can be from 10 mol% to 30% mol%.
- the one or more lipids for reducing immunogenicity of the composition can be from a total of 1 mol% to 8 mol% of the lipid components of the composition. In certain embodiments, the one or more lipids for reducing immunogenicity of the composition can be from a total of 1 mol% to 5 mol% of the lipid components of the composition.
- a composition of this invention can further include a cationic lipid, which can be from 5 mol% to 25 mol% of the lipid components of the composition.
- a composition of this invention can further include a cationic lipid, which can be from 5 mol% to 15 mol% of the lipid components of the composition.
- the molar ratio of the concentrations of the cationic lipid to the ionizable molecules of a composition of this invention can be from 5:35 to 25: 15.
- the entirety of the lipid components of a composition may include one or more of the ionizable compound molecular components, a structural lipid, one or more lipids for reducing immunogenicity of the composition, and one or more stabilizer lipids.
- three lipid-like components i.e. one or more ionizable molecules, a structural lipid, and one or more lipids for reducing
- immunogenicity of the composition can be 100% of the lipid components of the composition.
- a cationic lipid can be included.
- compositions of this invention are shown in Table 1.
- Table 1 Compositions of lipid components (each in mol% of total)
- lipid-like components i.e. one or more ionizable molecules, a structural lipid, and one or more lipids for reducing
- immunogenicity of the composition and one or more stabilizer lipids can be 100% of the lipid components of the composition.
- compositions of this invention are shown in Table 2.
- Table 2 Compositions of lipid components (each in mol% of total)
- aspects of this invention can provide a range of compositions for use in distributing an active agent to various organs or tissues of a subject.
- compositions of this invention can contain an ionizable lipid, a structural lipid, and a lipid for reducing immunogenicity of the composition.
- compositions of this invention can contain an ionizable lipid, a structural lipid, one or more stabilizer lipids, and a lipid for reducing immunogenicity of the composition.
- compositions of this invention may provide a surprisingly selective biodistribution of the active agent to a particular organ or tissue.
- composition of this invention can provide a surprisingly selective biodistribution of the active agent to the lung of a subject.
- composition of this invention can provide a surprisingly selective biodistribution of the active agent to the liver of a subject.
- a composition of this invention can provide a surprisingly selective biodistribution of the active agent to the colon of a subject. [00100] In some embodiments, a composition of this invention can provide a surprisingly selective biodistribution of the active agent to the pancreas of a subject.
- the ratio of the distribution of the active agent to the lung over the distribution of the active agent to the subject's liver can be at least 1.5.
- the ratio of the distribution of the active agent to the lung to the distribution of the active agent to the subject's liver can be at least 5.
- the ionizable compounds of this invention can have lipid-like properties, for example, as amphiphiles.
- Examples of an ionizable molecule include compounds having the structure shown in Formula I Formula I
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from 1-azetidines, 1 -pyrrolidines, 1-piperidines, 4-morpholines, and 1 4-piperazines wherein the rings can be substituted at any carbon atom position,
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, and aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl;
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- Q is O or R 7 ;
- p is from 1 to 4.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- p is 1, 2, 3 or 4.
- q is 0, 1, 2, 3 or 4.
- examples of an ionizable molecule include compounds havin the structure shown in Formula I Formula I
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- Q is O or R 7 .
- each of the alkenyl groups can have from one to two double bonds.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- examples of an ionizable molecule include compounds havin the structure shown in Formula I Formula I
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl
- Q is O or R 7 .
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group. [00118] In some embodiments, R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- n and m can be each independently from 3 to 6.
- examples of an ionizable molecule include compounds havin the structure shown in Formula I Formula I
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- examples of an ionizable molecule include compounds havin the structure shown in Formula I Formula I
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from
- each R 6 is independently hydroxyl
- each R 7 is independently selected from H, alkyl.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a
- Examples of an ionizable compound include the following compound Al :
- Examples of an ionizable compound include the following compound A2:
- Examples of an ionizable compound include the following compound A3 :
- Examples of an ionizable compound include the following compound A4:
- Examples of an ionizable compound include the following compound A5:
- an ionizable compound examples include the following compound A6:
- Examples of an ionizable compound include the following compound A7:
- Examples of an ionizable compound include the following compound A8:
- Examples of an ionizable compound include the following compound A9:
- an ionizable compound examples include the following compound AA:
- Examples of an ionizable compound include the following compound AB:
- Examples of an ionizable compound include the following compound AC:
- Examples of an ionizable compound include the following compound AD:
- Examples of an ionizable compound include the following compound AE:
- Examples of an ionizable compound include the following compound AF:
- Examples of an ionizable compound include the following compound Bl :
- Examples of an ionizable compound include the following compound B2:
- Examples of an ionizable compound include the following compound B3 :
- Examples of an ionizable compound include the following compound B4:
- Examples of an ionizable compound include the following compound B5 :
- Examples of an ionizable compound include the following compound B6: 00149] Examples of an ionizable compound include the following compound B7:
- Examples of an ionizable molecule include compounds having the structure shown in Formula II Formula II
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is a C( 12-20) alkyl group or a C( 12-20) alkenyl group that is substituted with a carboxylic acid or ester group.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- R 3 is a CQ4-18) alkyl group, or a C(14-18) alkenyl group that is substituted with a carboxylic acid or ester group.
- Examples of an ionizable compound include the following compound B8:
- Examples of an ionizable compound include the following compound B9:
- Examples of an ionizable compound include the following compound BA:
- Examples of an ionizable compound include the following compound BB:
- examples of an ionizable compound include compounds having the structure shown in Formula III Formula III
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from alkyl, hydroxyalkyl, alkoxyalkoxy, and carboxyalkyl;
- R 6 is selected from R 7 2 , N + HR 7 2 and N + R 7 3 ;
- R 7 is selected from H, alkyl, hydroxyalkyl, and aminoalkyl.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include the following compound BC:
- Examples of an ionizable compound include the following compound BD:
- Examples of an ionizable compound include the following compound BE :
- each of the alkenyl groups can have from one to two double bonds.
- Examples of an ionizable compound include compounds having the structure shown in Formula IV
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from 1-azetidines, 1 -pyrrolidines, 1-piperidines, 4-morpholines, and 1 4-piperazines wherein the rings can be substituted at any carbon atom position,
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl;
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- Q is O or R 7 ;
- p is from 1 to 4.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- p is 1, 2, 3 or 4.
- q is 0, 1, 2, 3 or 4.
- Examples of an ionizable compound include compounds having the structure shown in Formula IV Formula IV
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R J is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- p is from 1 to 4.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- p is 1, 2, 3 or 4.
- q is 0, 1, 2, 3 or 4.
- Examples of an ionizable compound include compounds having the structure shown in Formula IV
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include compounds having the structure shown in Formula IV
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from
- each R 6 is independently selected from H, hydroxyl, hydroxyalkyl, aminoalkyl;
- each R 7 is independently selected from H, alkyl
- p is from 1 to 4.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include the following compound BF:
- Examples of an ionizable compound include the following compound CI :
- Examples of an ionizable compound include the following compound C3 :
- Examples of an ionizable compound include the following compound C4:
- Examples of an ionizable compound include the following compound C5:
- Examples of an ionizable compound include the following compound C6:
- Examples of an ionizable compound include the following compound C7:
- Examples of an ionizable compound include the following compound C8:
- Examples of an ionizable compound include the following compound C9:
- Examples of an ionizable compound include the following compound CA:
- Examples of an ionizable compound include the following compound CB:
- Examples of an ionizable compound include the following compound CC:
- Examples of an ionizable compound include the following compound CD:
- Examples of an ionizable compound include the following compound CE:
- Examples of an ionizable compound include the following compound Dl :
- Examples of an ionizable compound include the following compound D2:
- ionizable compound include the following compound D3 :
- Examples of an ionizable compound include the following compound D4:
- n ionizable compound include the following compound D5:
- Examples of an ionizable compound include the following compound D6: 00212] Examples of an ionizable compound include the following compound D7:
- Examples of an ionizable compound include compounds having the structure shown in Formula IV-B
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- Z is S or O
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl;
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- p is 1, 2, 3 or 4.
- Examples of an ionizable compound include the following compound D8:
- Examples of an ionizable compound include the following compound D9:
- Examples of an ionizable compound include the following compound DA: 00222] Examples of an ionizable compound include the following compound DB:
- Examples of an ionizable compound include the following compound DD:
- Examples of an ionizable compound include the following compound DE:
- Examples of an ionizable compound include the following compound DF:
- Examples of an ionizable compound include the following compound E4:
- Embodiments of this invention include compounds having the structure shown in Formula V Formula V
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- p is from 1 to 4.
- R 3 is selected from 1-azetidines, 1 -pyrrolidines, 1-piperidines, 4-morpholines, and 1 4-piperazines wherein the rings can be substituted at any carbon atom position,
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- Q is O or R 7 .
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a C(14-18) alkyl group, or a C(14-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- p is 1, 2, 3 or 4.
- q is 0, 1, 2, 3 or 4.
- Embodiments of this invention include compounds having the stmcture
- R 4 and R 5 are independently for occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- p is from 1 to 4.
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl;
- Q is O or R 7 .
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a C(14-18) alkyl group, or a C(14-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include the following compound E5:
- Examples of an ionizable compound include the following compound E7:
- Examples of an ionizable compound include the following compound E8:
- Examples of an ionizable compound include the following compound E9:
- Examples of an ionizable compound include the following compound EA:
- Examples of an ionizable compound include the following compound EB:
- Examples of an ionizable compound include the following compound EC:
- Examples of an ionizable compound include the following compound ED:
- Examples of an ionizable compound include the following compound EE:
- Examples of an ionizable compound include the following compound EF:
- Examples of an ionizable compound include the following compound Fl :
- Examples of an ionizable compound include the following compound F2:
- Examples of an ionizable compound include compounds having the structure shown in Formula VI
- R 2 and R 4 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from aminoalkyl, quaternary aminoalkyl.
- each of the alkenyl groups can have from one to two double bonds.
- R 2 and R 4 are independently for each occurrence a C(14-18) alkyl group, or a C(14-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include the following compound F5:
- Examples of an ionizable compound include the following compound F6:
- Examples of an ionizable compound include the following compound F7:
- Examples of an ionizable compound include the following compound F8:
- Examples of an ionizable compound include the following compound F9:
- Examples of an ionizable compound include compounds having the structure shown in Formula VII
- n and m are each independently from 1 to 2; and R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- R 3 is selected from H, alkyl, aminoalkyl, quaternary aminoalkyl, hydroxyalkyl, alkoxyalkyl, alkoxyalkoxy alkyl.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include the following compound FA:
- Examples of an ionizable compound include the following compound FB:
- Examples of an ionizable compound include the following compound FC:
- each of the alkenyl groups can have from one to two double bonds.
- a compound can have the structure shown in
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R' is selected from
- R 10 is selected from 1-azetidines, 1 -pyrrolidines, 1 -piped dines, 4-morpholines, and 1 4-piperazines wherein the rings can be substituted at any carbon atom position,
- R 9 is selected from
- alkyl hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl;
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- p is from 1 to 4.
- Q is O or R 7 .
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- p is 1, 2, 3 or 4.
- q is 0, 1, 2, 3 or 4.
- a compound in certain embodiments, can have the structure shown in
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R is selected from
- R 9 is selected from
- alkyl hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- p is from 1 to 4.
- q is from zero to four;
- Q is O or R 7 .
- a compound can have the structure shown in Formula VIII
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from
- R 10 is selected from
- R 9 is selected from
- alkyl hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- p is from 1 to 4.
- q is from zero to four;
- Q is O or R 7 .
- a compound can have the structure shown in Formula VIII
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group; wherein R is selected from
- R is selected from
- R 9 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- Q is O or R 7 .
- a compound can have the structure shown in Formula VIII
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from
- R 10 is selected from
- R 9 is selected from
- alkyl hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- Q is O or R 7 .
- Examples of an ionizable compound include the following compound FD:
- Examples of an ionizable compound include the following compound FE:
- Examples of an ionizable compound include the following compound FF:
- Examples of an ionizable compound include the following compound Al l :
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound A14:
- Examples of an ionizable compound include the following compound A15:
- Examples of an ionizable compound include the following compound A16: [00295] Examples of an ionizable compound include the following compound A18:
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound A20:
- Examples of an ionizable compound include the following compound A21 : [00299] Examples of an ionizable compound include the following compound A22:
- Examples of an ionizable compound include the following compound A23 :
- Examples of an ionizable compound include the following compound A24:
- Examples of an ionizable compound include the following compound A25:
- each of the alkenyl groups can have from one to two double bonds.
- a compound can have the structure shown in Formula VIII-B
- n and m are each independently from 1 to 2;
- R 4 and R 5 are independently for each occurrence a C(12-20) alkyl group, or a C(12-20) alkenyl group;
- Z is N, O;
- R 3 is selected from
- alk l hydroxyalkyl, alkoxy, alkox alkoxy, aminoalkyl; independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl;
- p is from 1 to 4.
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include the following compound Bl l :
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound B14:
- a compound can have the stmcture shown in Formula IX
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- p is from 1 to 4.
- R 3 is selected from 1-azetidines, 1 -pyrrolidines, 1-piperidines, 4-morpholines, and 1 4-piperazines wherein the rings can be substituted at any carbon atom position,
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl;
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- Q is O or R 7 .
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- p is 1, 2, 3 or 4.
- q is 0, 1, 2, 3 or 4.
- a compound can have the structure shown in Formula IX
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- p is from 1 to 4.
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- a compound can have the structure shown in Formula IX
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- p is from 1 to 4.
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include the following compound Cl l :
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound C20:
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound C25:
- a compound can have the structure shown in Formula X Formula X
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- R 3 is selected from
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a C(14-18) alkyl group, or a CQ4-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include the following compound Dl l :
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound D14:
- Embodiments of this invention include compounds having the structure shown in Formula XI Formula XI
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- Z is O or H
- p is from 1 to 4.
- R 3 is selected from 1-azetidines, 1 -pyrrolidines, 1-piperidines, 4-morpholines, and 1 4-piperazines wherein the rings can be substituted at any carbon atom position,
- n also be selected from amino and aminoalkyl groups which can be substituted
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxyalkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl;
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- q is from zero to four;
- Q is O or R 7 .
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a CQ4-18) alkyl group, or a C(14-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- R 4 and R 5 are independently for each occurrence a C(16-18) alkyl group, or a C(16-18) alkenyl group.
- p is 1, 2, 3 or 4.
- q is 0, 1, 2, 3 or 4.
- Embodiments of this invention include compounds having the stmcture
- R 4 and R 5 are independently for each occurrence a C( 12-20) alkyl group, or a C( 12-20) alkenyl group;
- Z is O or H
- p is from 1 to 4.
- R 3 is selected from
- each R 6 is independently selected from H, alkyl, hydroxyl, hydroxyalkyl, alkoxy, alkoxy alkoxy, aminoalkyl;
- each R 7 is independently selected from H, alkyl
- each R 8 is independently selected from H, alkyl, hydroxyalkyl, and aminoalkyl, and any two R 8 may form a ring;
- each of the alkenyl groups can have from one to two double bonds.
- R 4 and R 5 are independently for each occurrence a C(14-18) alkyl group, or a C(14-18) alkenyl group.
- alkyl, hydroxyalkyl, and aminoalkyl refer to C(l-6)alkyl, hydroxyl[C(l-6)alkyl], and amino[C(l-6)alkyl].
- Examples of an ionizable compound include the following compound El l :
- Examples of an ionizable compound include the following compound E12:
- Examples of an ionizable compound include the following compound E13 :
- Examples of an ionizable compound include the following compound E14:
- Examples of an ionizable compound include the following compound E15 :
- Examples of an ionizable compound include the following compound E16:
- Examples of an ionizable compound include the following compound E17:
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound E22:
- Examples of an ionizable compound include the following compound E23 :
- Examples of an ionizable compound include the following compound E24
- Examples of an ionizable compound include the following compound E25
- Examples of an ionizable compound include the following compound E26: Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound E37
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of an ionizable compound include the following compound
- Examples of structural lipids include cholesterols, sterols, and steroids.
- Examples of structural lipids include cholanes, cholestanes, ergostanes, campestanes, poriferastanes, stigmastanes, gorgostanes, lanostanes, gonanes, estranes, androstanes, pregnanes, and cycloartanes.
- Examples of structural lipids include sterols and zoosterols such as cholesterol, lanosterol, zymosterol, zymostenol, desmosterol, stigmastanol,
- Examples of structural lipids include pegylated cholesterols, and cholestane 3-oxo-(Cl-22)acyl compounds, for example, cholesteryl acetate, cholesteryl arachidonate, cholesteryl butyrate, cholesteryl hexanoate, cholesteryl myristate, cholesteryl palmitate, cholesteryl behenate, cholesteryl stearate, cholesteryl caprylate, cholesteryl n-decanoate, cholesteryl dodecanoate, cholesteryl nervonate, cholesteryl pelargonate, cholesteryl n-valerate, cholesteryl oleate, cholesteryl elaidate, cholesteryl erucate, cholesteryl heptanoate, cholesteryl linolelaidate, and cholesteryl linoleate.
- cholesteryl acetate cholesteryl arachidonate, cholesteryl but
- Examples of structural lipids include sterols such as phytosterols, beta- sitosterol, campesterol, ergosterol, brassicasterol, delta-7-stigmasterol, and delta-7- avenasterol.
- sterols such as phytosterols, beta- sitosterol, campesterol, ergosterol, brassicasterol, delta-7-stigmasterol, and delta-7- avenasterol.
- Stabilizer lipids such as phytosterols, beta- sitosterol, campesterol, ergosterol, brassicasterol, delta-7-stigmasterol, and delta-7- avenasterol.
- Examples of stabilizer lipids include zwitterionic lipids.
- stabilizer lipids include compounds such as phospholipids.
- Examples of phospholipids include phosphatidylcholine,
- phosphatidylethanolamine phosphatidylserine, phosphatidylinositol, phosphatidic acid, palmitoyloleoyl phosphatidylcholine, lysophosphatidylcholine,
- stabilizer lipids include phosphatidyl ethanolamine compounds and phosphatidyl choline compounds.
- stabilizer lipids include l,2-dioleoyl-s «-Glycero-3- Phosphocholine (DOPC).
- stabilizer lipids include diphytanoyl phosphatidyl ethanolamine (DPhPE) and l,2-Diphytanoyl-s «-Glycero-3-Phosphocholine (DPhPC).
- DPhPE diphytanoyl phosphatidyl ethanolamine
- DPhPC l,2-Diphytanoyl-s «-Glycero-3-Phosphocholine
- stabilizer lipids include l,2-distearoyl-sn-glycero-3- phosphocholine (DSPC), l,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC), 1,2- dipalmitoyl-sn-glycero-3-phosphoethanolamine (DPPE), and l,2-dioleoyl-sn-glycero-3- phosphoethanolamine (DOPE).
- Examples of stabilizer lipids include 1,2-dilauroyl-sn-glycerol (DLG); 1,2-dimyristoyl-sn-glycerol (DMG); 1,2-dipalmitoyl-sn-glycerol (DPG); 1,2-distearoyl- sn-glycerol (DSG); l,2-diarachidoyl-sn-glycero-3-phosphocholine (DAPC);
- DMG 1,2-dimyristoyl-sn-glycerol
- DPG 1,2-dipalmitoyl-sn-glycerol
- DSG 1,2-distearoyl- sn-glycerol
- DAPC l,2-diarachidoyl-sn-glycero-3-phosphocholine
- DPePC l,2-dilauroyl-sn-glycero-3-phosphoethanolamine
- DLPE 1,2-dimyristoyl-sn- glycero-3-phosphoethanolamine
- DMPE 1,2-dimyristoyl-sn- glycero-3-phosphoethanolamine
- DSPE l,2-distearoyl-sn-glycero-3- phosphoethanolamine
- POPC palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine
- P-Lyso-PC l-palmitoyl-2-lyso-sn-glycero- 3-phosphocholine
- S- Lyso-PC l-Stearoyl-2-lyso-sn-glycero-3-phosphocholine
- Examples of lipids for reducing immunogenicity include polymeric compounds and polymer-lipid conjugates.
- Examples of lipids for reducing immunogenicity include pegylated lipids having polyethyleneglycol (PEG) regions.
- PEG regions can be of any molecular mass.
- a PEG region can have a molecular mass of 200, 300, 350, 400, 500, 550, 750, 1000, 1500, 2000, 3000, 3500, 4000 or 5000 Da.
- Examples of lipids for reducing immunogenicity include compounds having a methoxypolyethyleneglycol region.
- Examples of lipids for reducing immunogenicity include compounds having a carbonyl-methoxypolyethyleneglycol region.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Biomedical Technology (AREA)
- Nanotechnology (AREA)
- Optics & Photonics (AREA)
- Physics & Mathematics (AREA)
- Biophysics (AREA)
- Dispersion Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Medicinal Preparation (AREA)
- Hydrogenated Pyridines (AREA)
- Plural Heterocyclic Compounds (AREA)
- Pyrrole Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
Claims
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| RU2018102536A RU2776478C2 (en) | 2015-06-24 | 2016-06-23 | Ionized compounds and compositions and their use |
| EP20163284.1A EP3686184A3 (en) | 2015-06-24 | 2016-06-23 | Ionizable compounds and compositions and uses thereof |
| CA2990668A CA2990668A1 (en) | 2015-06-24 | 2016-06-23 | Ionizable compounds and compositions and uses therefof |
| ES16815348T ES2828717T3 (en) | 2015-06-24 | 2016-06-23 | Ionizable Compounds and Compositions and Uses Thereof |
| EP16815348.4A EP3313449B1 (en) | 2015-06-24 | 2016-06-23 | Ionizable compounds and compositions and uses thereof |
| KR1020187002177A KR102659774B1 (en) | 2015-06-24 | 2016-06-23 | Ionizable compounds and compositions and uses thereof |
| CN201680037092.8A CN107708739A (en) | 2015-06-24 | 2016-06-23 | Ionizable compound, composition and their purposes |
| JP2017566796A JP6480025B2 (en) | 2015-06-24 | 2016-06-23 | Ionic compounds and compositions and uses thereof |
| AU2016281685A AU2016281685B2 (en) | 2015-06-24 | 2016-06-23 | Ionizable compounds and compositions and uses thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201562184188P | 2015-06-24 | 2015-06-24 | |
| US62/184,188 | 2015-06-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016210190A1 true WO2016210190A1 (en) | 2016-12-29 |
Family
ID=57585619
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2016/039114 WO2016210190A1 (en) | 2015-06-24 | 2016-06-23 | Ionizable compounds and compositions and uses thereof |
Country Status (12)
| Country | Link |
|---|---|
| US (3) | US10167253B2 (en) |
| EP (2) | EP3313449B1 (en) |
| JP (1) | JP6480025B2 (en) |
| KR (1) | KR102659774B1 (en) |
| CN (1) | CN107708739A (en) |
| AU (1) | AU2016281685B2 (en) |
| CA (1) | CA2990668A1 (en) |
| ES (1) | ES2828717T3 (en) |
| HK (1) | HK1253031A1 (en) |
| PT (1) | PT3313449T (en) |
| TW (1) | TWI746442B (en) |
| WO (1) | WO2016210190A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020039631A1 (en) * | 2018-08-21 | 2020-02-27 | 株式会社 東芝 | Biodegradable compound, lipid particles, composition containing lipid particles, and kit |
| WO2021204175A1 (en) * | 2020-04-09 | 2021-10-14 | Suzhou Abogen Biosciences Co., Ltd. | Lipid nanoparticle composition |
| EP3880212A4 (en) * | 2018-11-16 | 2022-08-10 | Nitto Denko Corporation | Rna interference delivery formulation and methods for malignant tumors |
| US11510977B2 (en) | 2020-04-09 | 2022-11-29 | Suzhou Abogen Biosciences Co., Ltd. | Nucleic acid vaccines for coronavirus |
| WO2023126404A1 (en) * | 2021-12-28 | 2023-07-06 | BioNTech SE | Lipid-based formulations for administration of rna |
| TWI840449B (en) | 2018-11-16 | 2024-05-01 | 日商日東電工股份有限公司 | Rna interference delivery formulation and methods for malignant tumors |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PT3313449T (en) | 2015-06-24 | 2020-10-30 | Nitto Denko Corp | Ionizable compounds and compositions and uses thereof |
| US20220049251A1 (en) * | 2020-05-20 | 2022-02-17 | Nitto Denko Corporation | dsRNA Directed to Coronavirus Proteins |
| BR112023000568A2 (en) * | 2020-07-17 | 2023-01-31 | Sanofi Pasteur | CLEAVABLE LIPID COMPOUNDS, COMPOSITIONS CONTAINING THEM, AND USES THEREOF |
| CN115466269A (en) * | 2021-06-11 | 2022-12-13 | 复旦大学 | Choline carbonate prodrug and preparation method and application thereof |
| WO2023116804A1 (en) * | 2021-12-23 | 2023-06-29 | 苏州艾博生物科技有限公司 | Lipid compound and lipid nanoparticle composition |
| WO2024012272A1 (en) * | 2022-07-11 | 2024-01-18 | 传信生物医药(苏州)有限公司 | Lipid compound and use thereof |
| CN117285438A (en) * | 2022-08-12 | 2023-12-26 | 上海蓝鹊生物医药有限公司 | Nitrogen-containing chain compound, preparation method thereof, composition containing nitrogen-containing chain compound and application of nitrogen-containing chain compound |
Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4115548A (en) * | 1974-01-18 | 1978-09-19 | The Procter & Gamble Company | Detergent compositions comprising modified proteins |
| US4554100A (en) * | 1981-08-07 | 1985-11-19 | Spofa, Spojene Podniky Pro Zdravotnickou Vyrobu | Alkylamides of carboxyalkanoyl peptides and method for preparation thereof |
| US5314786A (en) * | 1991-04-20 | 1994-05-24 | Hoechst Aktiengesellschaft | Positive-working radiation sensitive mixture comprising sulfonic acid esters of 2,4,6-tris-(2-hydroxyethoxy)-[1,3,5]triazine, and recording material containing these esters |
| US5370877A (en) * | 1988-03-03 | 1994-12-06 | Knoll Ag | Method for improving delivery and reducing toxicity of biologically active substances using novel α-amino dicarboxylic acid derivatives |
| WO2002028438A1 (en) * | 2000-10-05 | 2002-04-11 | King's College London | Lipopeptides as absorption enhancers for bioactive compounds |
| US6372254B1 (en) * | 1998-04-02 | 2002-04-16 | Impax Pharmaceuticals Inc. | Press coated, pulsatile drug delivery system suitable for oral administration |
| WO2002030922A2 (en) * | 2000-10-13 | 2002-04-18 | Shire Biochem Inc. | Dioxolane analogs for improved inter-cellular delivery |
| US6399629B1 (en) * | 1998-06-01 | 2002-06-04 | Microcide Pharmaceuticals, Inc. | Efflux pump inhibitors |
| US20020187915A1 (en) * | 2001-03-28 | 2002-12-12 | Kao Corporation | Surfactant composition |
| US20080132500A1 (en) * | 2006-10-20 | 2008-06-05 | Kun Liu | Antibiotic compounds |
| US20100105899A1 (en) * | 2007-03-01 | 2010-04-29 | Neumann William L | Integrated Photoactive Small Molecules and Uses Thereof |
| US20110052673A1 (en) * | 2008-01-29 | 2011-03-03 | Arthur Tzianabos | Therapeutic compositions |
| WO2012099755A1 (en) * | 2011-01-11 | 2012-07-26 | Alnylam Pharmaceuticals, Inc. | Pegylated lipids and their use for drug delivery |
| US20130115274A1 (en) | 2011-11-04 | 2013-05-09 | Nitto Denko Corporation | Method of producing lipid nanoparticles for drug delivery |
| US20130330401A1 (en) | 2012-06-08 | 2013-12-12 | Nitto Denko Corporation | Lipids for therapeutic agent delivery formulations |
| US20140294934A1 (en) * | 2011-06-08 | 2014-10-02 | Nitto Denko Corporation | Cationic lipids for therapeutic agent delivery formulations |
| US20140364642A1 (en) * | 2011-12-01 | 2014-12-11 | The University Of Tokyo | Compound having read-through activity |
| US8916576B2 (en) * | 2005-03-25 | 2014-12-23 | Surface Logix, Inc. | Pharmacokinetically improved compounds |
| US20150112051A1 (en) * | 2008-07-11 | 2015-04-23 | Steffen Panzner | Construction of cell penetrating nucleic acids |
Family Cites Families (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE198757T1 (en) | 1989-08-29 | 2001-02-15 | Univ Virginia Commonwealth | CYTOPROTECTIVE SUBSTANCES WITH UNSATURATED HYDROCARBON RESIDUE OF 16 TO 20 CARBON ATOMS |
| US5082683A (en) | 1990-08-01 | 1992-01-21 | Nabisco Brands, Inc. | Amide/amine ester derivatives as low calorie fat mimetics |
| JP2575235B2 (en) | 1991-04-11 | 1997-01-22 | 株式会社トクヤマ | Method for producing ion-sensitive membrane |
| MY111077A (en) | 1992-11-13 | 1999-08-30 | Kao Corp | Agricultural chemical composition |
| JP2994898B2 (en) | 1992-12-28 | 1999-12-27 | 花王株式会社 | Liquid soft finish |
| JPH06336466A (en) | 1993-05-26 | 1994-12-06 | Lion Corp | New amido ester amine compound, its intermediate, production thereof and softening agent |
| JPH0718570A (en) | 1993-06-29 | 1995-01-20 | Lion Corp | Softening agent composition |
| JPH0718569A (en) | 1993-06-29 | 1995-01-20 | Lion Corp | Softening agent composition |
| JPH08295657A (en) | 1995-04-27 | 1996-11-12 | Kanebo Ltd | Intermediate for surfactant |
| DE19605175A1 (en) * | 1996-02-13 | 1997-08-14 | Sourovoi Andrej Dr | Lipid compounds and their use |
| NZ334613A (en) | 1996-08-12 | 2002-02-01 | Welfide Corp | Pharmaceutical agents comprising Rho kinase inhibitor |
| DE19637043A1 (en) | 1996-09-12 | 1998-03-19 | Boehringer Mannheim Gmbh | Novel amino alcohol derivatives, processes for their preparation and medicaments and reagents containing these compounds |
| US5958894A (en) | 1997-04-04 | 1999-09-28 | Megabios Corporation | Amphiphilic biguanide derivatives |
| AU739998B2 (en) | 1997-06-12 | 2001-10-25 | Transgene S.A. | Novel lipid complexes for transferring at least one therapeutically active substance, in particular a polynucleotide, into a target cell, and use in gene therapy |
| JP2001247532A (en) | 2000-03-08 | 2001-09-11 | Chukyo Yushi Kk | Fatty acid amide compound and use thereof |
| JP2004250343A (en) | 2003-02-18 | 2004-09-09 | Fuji Photo Film Co Ltd | Iodoaryl group-having amino acid derivative |
| WO2005054486A1 (en) * | 2003-12-05 | 2005-06-16 | Fukuoka Prefectural Government | Method of preparing gene transfer reagent |
| JP2006254877A (en) * | 2005-03-18 | 2006-09-28 | Fukuoka Prefecture | Carrier containing glycolipid and method for transferring gene using the same |
| US7915230B2 (en) | 2005-05-17 | 2011-03-29 | Molecular Transfer, Inc. | Reagents for transfection of eukaryotic cells |
| EP2604255B1 (en) | 2006-05-05 | 2017-10-25 | Molecular Transfer, Inc. | Novel reagents for transfection of eukaryotic cells |
| GB0720486D0 (en) | 2007-10-19 | 2007-11-28 | Univ Edinburgh | Cationic lipids |
| JP5062042B2 (en) | 2008-05-28 | 2012-10-31 | コニカミノルタビジネステクノロジーズ株式会社 | Toner for developing electrostatic image and image forming method |
| US8722082B2 (en) | 2008-11-10 | 2014-05-13 | Tekmira Pharmaceuticals Corporation | Lipids and compositions for the delivery of therapeutics |
| JP5515360B2 (en) | 2009-03-30 | 2014-06-11 | 日油株式会社 | Lipid derivatives |
| CN101648121B (en) | 2009-08-21 | 2012-08-29 | 江苏钟山化工有限公司 | Water-soluble thickener, preparation method thereof and application thereof in pesticide formulation |
| AU2011245987B2 (en) * | 2010-04-28 | 2016-12-15 | Kyowa Hakko Kirin Co., Ltd | Cationic lipid |
| US20160015656A2 (en) * | 2010-08-05 | 2016-01-21 | Nitto Denko Corporation | Composition for regenerating normal tissue from fibrotic tissue |
| JP5733647B2 (en) | 2011-03-30 | 2015-06-10 | 福岡県 | Gene transfer aid and gene transfer method using the same |
| AU2012242455A1 (en) | 2011-04-15 | 2013-11-28 | Molecular Transfer, Inc. | Agents for improved delivery of nucleic acids to eukaryotic cells |
| WO2013158127A1 (en) | 2012-04-16 | 2013-10-24 | Molecular Transfer, Inc. | Agents for improved delivery of nucleic acids to eukaryotic cells |
| WO2012162210A1 (en) | 2011-05-26 | 2012-11-29 | Merck Sharp & Dohme Corp. | Ring constrained cationic lipids for oligonucleotide delivery |
| KR102029657B1 (en) * | 2011-06-08 | 2019-10-08 | 닛토덴코 가부시키가이샤 | COMPOUNDS FOR TARGETING DRUG DELIVERY AND ENHANCING siRNA ACTIVITY |
| JP2013095755A (en) * | 2011-11-02 | 2013-05-20 | Kyowa Hakko Kirin Co Ltd | Cationic lipid |
| WO2013155487A1 (en) | 2012-04-12 | 2013-10-17 | Yale University | Vehicles for controlled delivery of different pharmaceutical agents |
| US10264976B2 (en) | 2014-12-26 | 2019-04-23 | The University Of Akron | Biocompatible flavonoid compounds for organelle and cell imaging |
| WO2016134146A2 (en) | 2015-02-19 | 2016-08-25 | Nitto Denko Corporation | Rna interference therapeutics against ebola virus |
| PT3313449T (en) | 2015-06-24 | 2020-10-30 | Nitto Denko Corp | Ionizable compounds and compositions and uses thereof |
-
2016
- 2016-06-23 PT PT168153484T patent/PT3313449T/en unknown
- 2016-06-23 JP JP2017566796A patent/JP6480025B2/en active Active
- 2016-06-23 EP EP16815348.4A patent/EP3313449B1/en active Active
- 2016-06-23 KR KR1020187002177A patent/KR102659774B1/en active IP Right Grant
- 2016-06-23 CN CN201680037092.8A patent/CN107708739A/en active Pending
- 2016-06-23 WO PCT/US2016/039114 patent/WO2016210190A1/en active Application Filing
- 2016-06-23 EP EP20163284.1A patent/EP3686184A3/en active Pending
- 2016-06-23 ES ES16815348T patent/ES2828717T3/en active Active
- 2016-06-23 US US15/191,511 patent/US10167253B2/en active Active
- 2016-06-23 CA CA2990668A patent/CA2990668A1/en active Pending
- 2016-06-23 AU AU2016281685A patent/AU2016281685B2/en active Active
- 2016-06-24 TW TW105120027A patent/TWI746442B/en active
-
2018
- 2018-09-27 HK HK18112410.7A patent/HK1253031A1/en unknown
- 2018-12-20 US US16/228,054 patent/US11384051B2/en active Active
-
2022
- 2022-04-05 US US17/658,075 patent/US20220242820A1/en active Pending
Patent Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4115548A (en) * | 1974-01-18 | 1978-09-19 | The Procter & Gamble Company | Detergent compositions comprising modified proteins |
| US4554100A (en) * | 1981-08-07 | 1985-11-19 | Spofa, Spojene Podniky Pro Zdravotnickou Vyrobu | Alkylamides of carboxyalkanoyl peptides and method for preparation thereof |
| US5370877A (en) * | 1988-03-03 | 1994-12-06 | Knoll Ag | Method for improving delivery and reducing toxicity of biologically active substances using novel α-amino dicarboxylic acid derivatives |
| US5314786A (en) * | 1991-04-20 | 1994-05-24 | Hoechst Aktiengesellschaft | Positive-working radiation sensitive mixture comprising sulfonic acid esters of 2,4,6-tris-(2-hydroxyethoxy)-[1,3,5]triazine, and recording material containing these esters |
| US6372254B1 (en) * | 1998-04-02 | 2002-04-16 | Impax Pharmaceuticals Inc. | Press coated, pulsatile drug delivery system suitable for oral administration |
| US6399629B1 (en) * | 1998-06-01 | 2002-06-04 | Microcide Pharmaceuticals, Inc. | Efflux pump inhibitors |
| WO2002028438A1 (en) * | 2000-10-05 | 2002-04-11 | King's College London | Lipopeptides as absorption enhancers for bioactive compounds |
| WO2002030922A2 (en) * | 2000-10-13 | 2002-04-18 | Shire Biochem Inc. | Dioxolane analogs for improved inter-cellular delivery |
| US20020187915A1 (en) * | 2001-03-28 | 2002-12-12 | Kao Corporation | Surfactant composition |
| US8916576B2 (en) * | 2005-03-25 | 2014-12-23 | Surface Logix, Inc. | Pharmacokinetically improved compounds |
| US20080132500A1 (en) * | 2006-10-20 | 2008-06-05 | Kun Liu | Antibiotic compounds |
| US20100105899A1 (en) * | 2007-03-01 | 2010-04-29 | Neumann William L | Integrated Photoactive Small Molecules and Uses Thereof |
| US20110052673A1 (en) * | 2008-01-29 | 2011-03-03 | Arthur Tzianabos | Therapeutic compositions |
| US20150112051A1 (en) * | 2008-07-11 | 2015-04-23 | Steffen Panzner | Construction of cell penetrating nucleic acids |
| WO2012099755A1 (en) * | 2011-01-11 | 2012-07-26 | Alnylam Pharmaceuticals, Inc. | Pegylated lipids and their use for drug delivery |
| US20140294934A1 (en) * | 2011-06-08 | 2014-10-02 | Nitto Denko Corporation | Cationic lipids for therapeutic agent delivery formulations |
| US20130115274A1 (en) | 2011-11-04 | 2013-05-09 | Nitto Denko Corporation | Method of producing lipid nanoparticles for drug delivery |
| US20140364642A1 (en) * | 2011-12-01 | 2014-12-11 | The University Of Tokyo | Compound having read-through activity |
| US20130330401A1 (en) | 2012-06-08 | 2013-12-12 | Nitto Denko Corporation | Lipids for therapeutic agent delivery formulations |
Non-Patent Citations (6)
| Title |
|---|
| "Remington's Pharmaceutical Sciences", 1990, MACK PUBLISHING CO. |
| ARTHUR, JC ET AL.: "Microbial genomic analysis reveals the essential role of inflammation in bacteria-induced colorectal cancer.", NATURE COMMUNICATIONS, vol. 5, 3 September 2014 (2014-09-03), pages 1 - 24, XP055340908 * |
| BURKE, B ET AL.: "Macrophages in gene therapy: cellular delivery vehicles and in vivo targets.", JOURNAL OF LEUKOCYTE BIOLOGY, vol. 72, September 2002 (2002-09-01), pages 417 - 428, XP009003789 * |
| DONALD VOETJUDITH VOET: "Biochemistry", 2005, pages: 383 |
| MICHAEL B. SMITHJERRY MARCH: "March's Advanced Organic Chemistry", 2001 |
| SCHULZE, U ET AL.: "Synthesis of Novel Cationic Poly(Ethylene Glycol) Containing Lipids.", BIOCONJUGATE CHEMISTRY, vol. 10, 1999, pages 548 - 552, XP055282546 * |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111164068B (en) * | 2018-08-21 | 2023-11-14 | 株式会社东芝 | Biodegradable compound, lipid particle, composition containing lipid particle, and kit |
| US11548857B2 (en) | 2018-08-21 | 2023-01-10 | Kabushiki Kaisha Toshiba | Biodegradable compound, lipid particle, composition comprising lipid particle, and kit |
| JP2023016905A (en) * | 2018-08-21 | 2023-02-02 | 株式会社東芝 | Biodegradable compound, lipid particles, kit comprising lipid particles, activator delivery method employing lipid particles, and cell production method |
| US11952351B2 (en) | 2018-08-21 | 2024-04-09 | Kabushiki Kaisha Toshiba | Lipid particle, composition comprising lipid particle, and method for delivering activators to cell |
| CN111164068A (en) * | 2018-08-21 | 2020-05-15 | 株式会社东芝 | Biodegradable compound, lipid particle, composition containing lipid particle, and kit |
| JP7431929B2 (en) | 2018-08-21 | 2024-02-15 | 株式会社東芝 | Biodegradable compounds, lipid particles, kits containing lipid particles, active agent delivery methods using lipid particles, and cell production methods |
| WO2020039631A1 (en) * | 2018-08-21 | 2020-02-27 | 株式会社 東芝 | Biodegradable compound, lipid particles, composition containing lipid particles, and kit |
| TWI840449B (en) | 2018-11-16 | 2024-05-01 | 日商日東電工股份有限公司 | Rna interference delivery formulation and methods for malignant tumors |
| US11708575B2 (en) | 2018-11-16 | 2023-07-25 | Nitto Denko Corporation | RNA interference delivery formulation and methods for malignant tumors |
| EP3880212A4 (en) * | 2018-11-16 | 2022-08-10 | Nitto Denko Corporation | Rna interference delivery formulation and methods for malignant tumors |
| US11472766B2 (en) | 2020-04-09 | 2022-10-18 | Suzhou Abogen Biosciences Co., Ltd. | Lipid nanoparticle composition |
| WO2021204175A1 (en) * | 2020-04-09 | 2021-10-14 | Suzhou Abogen Biosciences Co., Ltd. | Lipid nanoparticle composition |
| AU2021254312B2 (en) * | 2020-04-09 | 2024-01-11 | Suzhou Abogen Biosciences Co., Ltd. | Lipid nanoparticle composition |
| US11510977B2 (en) | 2020-04-09 | 2022-11-29 | Suzhou Abogen Biosciences Co., Ltd. | Nucleic acid vaccines for coronavirus |
| CN114206827A (en) * | 2020-04-09 | 2022-03-18 | 苏州艾博生物科技有限公司 | Lipid nanoparticle compositions |
| WO2023126053A1 (en) * | 2021-12-28 | 2023-07-06 | BioNTech SE | Lipid-based formulations for administration of rna |
| WO2023126404A1 (en) * | 2021-12-28 | 2023-07-06 | BioNTech SE | Lipid-based formulations for administration of rna |
Also Published As
| Publication number | Publication date |
|---|---|
| KR102659774B1 (en) | 2024-04-23 |
| RU2018102536A3 (en) | 2019-11-08 |
| TWI746442B (en) | 2021-11-21 |
| EP3686184A2 (en) | 2020-07-29 |
| CN107708739A (en) | 2018-02-16 |
| RU2018102536A (en) | 2019-07-24 |
| US20190127318A1 (en) | 2019-05-02 |
| US10167253B2 (en) | 2019-01-01 |
| ES2828717T3 (en) | 2021-05-27 |
| US20220242820A1 (en) | 2022-08-04 |
| HK1253031A1 (en) | 2019-06-06 |
| EP3313449A4 (en) | 2018-11-07 |
| PT3313449T (en) | 2020-10-30 |
| JP6480025B2 (en) | 2019-03-06 |
| AU2016281685A1 (en) | 2018-01-18 |
| JP2018524330A (en) | 2018-08-30 |
| CA2990668A1 (en) | 2016-12-29 |
| EP3313449B1 (en) | 2020-09-23 |
| EP3313449A1 (en) | 2018-05-02 |
| EP3686184A3 (en) | 2020-08-26 |
| US20160376229A1 (en) | 2016-12-29 |
| TW201713614A (en) | 2017-04-16 |
| US11384051B2 (en) | 2022-07-12 |
| KR20180021115A (en) | 2018-02-28 |
| AU2016281685B2 (en) | 2021-08-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2016281685B2 (en) | Ionizable compounds and compositions and uses thereof | |
| EP2567951B1 (en) | Cationic lipid | |
| JP6226476B2 (en) | Cationic lipid | |
| CA3071968A1 (en) | Nucleic acid-containing lipid nanoparticle | |
| EP3733647B1 (en) | Cationic lipid | |
| EP3162794B1 (en) | Alkyl substituted azetidines as cationic lipid | |
| JP6495408B2 (en) | Cationic lipid | |
| EP3153172A1 (en) | Ckap5-gene-silencing rnai pharmaceutical composition | |
| RU2776478C2 (en) | Ionized compounds and compositions and their use | |
| EP3521270B1 (en) | Cationic lipid compound | |
| BR112017027458B1 (en) | COMPOUND, AND, COMPOSITION | |
| WO2023086514A1 (en) | Ionizable cationic lipids for rna delivery | |
| CN117756653A (en) | Cationic lipid compound, composition and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16815348 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2990668 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2017566796 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2016281685 Country of ref document: AU Date of ref document: 20160623 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20187002177 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2016815348 Country of ref document: EP Ref document number: 2018102536 Country of ref document: RU |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112017027458 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112017027458 Country of ref document: BR Kind code of ref document: A2 Effective date: 20171219 |