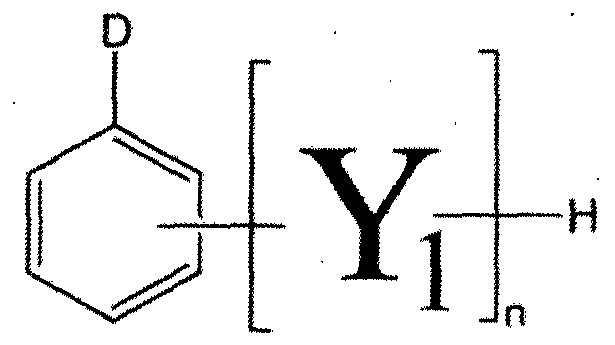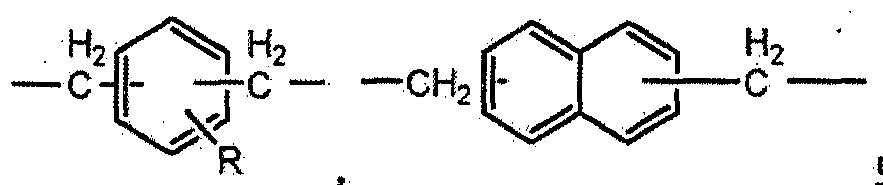WO2015105379A1 - Novel novolac hardener having alkoxysilyl group, method for preparing same, composition containing same, hardened product, and use thereof - Google Patents
Novel novolac hardener having alkoxysilyl group, method for preparing same, composition containing same, hardened product, and use thereof Download PDFInfo
- Publication number
- WO2015105379A1 WO2015105379A1 PCT/KR2015/000243 KR2015000243W WO2015105379A1 WO 2015105379 A1 WO2015105379 A1 WO 2015105379A1 KR 2015000243 W KR2015000243 W KR 2015000243W WO 2015105379 A1 WO2015105379 A1 WO 2015105379A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- composition
- group
- formulas
- curing agent
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 163
- 229920003986 novolac Polymers 0.000 title claims abstract description 132
- 125000005370 alkoxysilyl group Chemical group 0.000 title claims abstract description 81
- 238000000034 method Methods 0.000 title claims abstract description 33
- 239000004848 polyfunctional curative Substances 0.000 title claims abstract description 16
- 239000002131 composite material Substances 0.000 claims abstract description 54
- 239000000126 substance Substances 0.000 claims abstract description 24
- 230000009477 glass transition Effects 0.000 claims abstract description 22
- 239000000945 filler Substances 0.000 claims abstract description 21
- 239000003795 chemical substances by application Substances 0.000 claims description 155
- 239000003822 epoxy resin Substances 0.000 claims description 64
- 229920000647 polyepoxide Polymers 0.000 claims description 64
- 239000000835 fiber Substances 0.000 claims description 62
- 239000002904 solvent Substances 0.000 claims description 55
- -1 alkenyl compound Chemical class 0.000 claims description 48
- 239000010954 inorganic particle Substances 0.000 claims description 48
- 239000003365 glass fiber Substances 0.000 claims description 42
- 239000000047 product Substances 0.000 claims description 38
- 125000000217 alkyl group Chemical group 0.000 claims description 32
- 239000007858 starting material Substances 0.000 claims description 26
- 125000003545 alkoxy group Chemical group 0.000 claims description 25
- 125000004432 carbon atom Chemical group C* 0.000 claims description 25
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 24
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 24
- 239000004065 semiconductor Substances 0.000 claims description 23
- 239000007787 solid Substances 0.000 claims description 21
- 239000003054 catalyst Substances 0.000 claims description 20
- 229910052739 hydrogen Inorganic materials 0.000 claims description 19
- 239000001257 hydrogen Substances 0.000 claims description 19
- 238000004519 manufacturing process Methods 0.000 claims description 17
- 239000000758 substrate Substances 0.000 claims description 16
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 claims description 15
- 229910052751 metal Inorganic materials 0.000 claims description 14
- 239000002184 metal Substances 0.000 claims description 14
- 150000001412 amines Chemical class 0.000 claims description 13
- 239000007809 chemical reaction catalyst Substances 0.000 claims description 13
- 150000001875 compounds Chemical class 0.000 claims description 13
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 claims description 12
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 claims description 12
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 12
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 claims description 10
- 239000013067 intermediate product Substances 0.000 claims description 10
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 9
- 239000000463 material Substances 0.000 claims description 9
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 claims description 8
- 239000004305 biphenyl Substances 0.000 claims description 8
- 235000010290 biphenyl Nutrition 0.000 claims description 8
- 239000012776 electronic material Substances 0.000 claims description 8
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 claims description 8
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 8
- 125000003342 alkenyl group Chemical group 0.000 claims description 7
- 125000003055 glycidyl group Chemical group C(C1CO1)* 0.000 claims description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 7
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 claims description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 6
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims description 6
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims description 6
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 claims description 6
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 6
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 claims description 6
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical compound C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 claims description 6
- 239000012784 inorganic fiber Substances 0.000 claims description 6
- 239000005022 packaging material Substances 0.000 claims description 6
- 229910052697 platinum Inorganic materials 0.000 claims description 6
- 239000000377 silicon dioxide Substances 0.000 claims description 6
- 239000000853 adhesive Substances 0.000 claims description 5
- 230000001070 adhesive effect Effects 0.000 claims description 5
- 239000003973 paint Substances 0.000 claims description 5
- BLDLRWQLBOJPEB-UHFFFAOYSA-N 2-(2-hydroxyphenyl)sulfanylphenol Chemical compound OC1=CC=CC=C1SC1=CC=CC=C1O BLDLRWQLBOJPEB-UHFFFAOYSA-N 0.000 claims description 4
- 229930185605 Bisphenol Natural products 0.000 claims description 4
- 125000001931 aliphatic group Chemical group 0.000 claims description 4
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical compound OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 claims description 4
- PEQHIRFAKIASBK-UHFFFAOYSA-N tetraphenylmethane Chemical compound C1=CC=CC=C1C(C=1C=CC=CC=1)(C=1C=CC=CC=1)C1=CC=CC=C1 PEQHIRFAKIASBK-UHFFFAOYSA-N 0.000 claims description 4
- AAAQKTZKLRYKHR-UHFFFAOYSA-N triphenylmethane Chemical compound C1=CC=CC=C1C(C=1C=CC=CC=1)C1=CC=CC=C1 AAAQKTZKLRYKHR-UHFFFAOYSA-N 0.000 claims description 4
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 claims description 3
- CDAWCLOXVUBKRW-UHFFFAOYSA-N 2-aminophenol Chemical compound NC1=CC=CC=C1O CDAWCLOXVUBKRW-UHFFFAOYSA-N 0.000 claims description 3
- YBRVSVVVWCFQMG-UHFFFAOYSA-N 4,4'-diaminodiphenylmethane Chemical compound C1=CC(N)=CC=C1CC1=CC=C(N)C=C1 YBRVSVVVWCFQMG-UHFFFAOYSA-N 0.000 claims description 3
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 claims description 3
- 239000002033 PVDF binder Substances 0.000 claims description 3
- 239000004695 Polyether sulfone Substances 0.000 claims description 3
- 239000004698 Polyethylene Substances 0.000 claims description 3
- 239000004743 Polypropylene Substances 0.000 claims description 3
- 229910052581 Si3N4 Inorganic materials 0.000 claims description 3
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 claims description 3
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 claims description 3
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 claims description 3
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims description 3
- 229910021529 ammonia Inorganic materials 0.000 claims description 3
- 125000003118 aryl group Chemical group 0.000 claims description 3
- PMHQVHHXPFUNSP-UHFFFAOYSA-M copper(1+);methylsulfanylmethane;bromide Chemical compound Br[Cu].CSC PMHQVHHXPFUNSP-UHFFFAOYSA-M 0.000 claims description 3
- 229910017604 nitric acid Inorganic materials 0.000 claims description 3
- 229920001778 nylon Polymers 0.000 claims description 3
- AFEQENGXSMURHA-UHFFFAOYSA-N oxiran-2-ylmethanamine Chemical compound NCC1CO1 AFEQENGXSMURHA-UHFFFAOYSA-N 0.000 claims description 3
- 229920003207 poly(ethylene-2,6-naphthalate) Polymers 0.000 claims description 3
- 229920002577 polybenzoxazole Polymers 0.000 claims description 3
- 229920000728 polyester Polymers 0.000 claims description 3
- 229920006393 polyether sulfone Polymers 0.000 claims description 3
- 229920000573 polyethylene Polymers 0.000 claims description 3
- 239000011112 polyethylene naphthalate Substances 0.000 claims description 3
- 239000005020 polyethylene terephthalate Substances 0.000 claims description 3
- 229920000139 polyethylene terephthalate Polymers 0.000 claims description 3
- 229920001155 polypropylene Polymers 0.000 claims description 3
- 229920002981 polyvinylidene fluoride Polymers 0.000 claims description 3
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 claims description 3
- 229910052723 transition metal Inorganic materials 0.000 claims description 3
- 125000002947 alkylene group Chemical group 0.000 claims description 2
- 239000004973 liquid crystal related substance Substances 0.000 claims description 2
- 239000010453 quartz Substances 0.000 claims description 2
- 239000004696 Poly ether ether ketone Substances 0.000 claims 1
- 229920002530 polyetherether ketone Polymers 0.000 claims 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 abstract description 14
- 125000003700 epoxy group Chemical group 0.000 abstract description 4
- 230000001747 exhibiting effect Effects 0.000 abstract description 3
- 238000006243 chemical reaction Methods 0.000 description 67
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 42
- 239000004593 Epoxy Substances 0.000 description 30
- 230000015572 biosynthetic process Effects 0.000 description 22
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 21
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 20
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 19
- 238000003786 synthesis reaction Methods 0.000 description 19
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 18
- 229920005989 resin Polymers 0.000 description 16
- 239000011347 resin Substances 0.000 description 16
- 239000002585 base Substances 0.000 description 15
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 12
- 239000010408 film Substances 0.000 description 12
- 125000001181 organosilyl group Chemical group [SiH3]* 0.000 description 12
- 239000007795 chemical reaction product Substances 0.000 description 11
- 229910010272 inorganic material Inorganic materials 0.000 description 11
- 239000011147 inorganic material Substances 0.000 description 11
- 125000001424 substituent group Chemical group 0.000 description 11
- 0 CC*1=CC=C(C=CC=C2)C2=CC1 Chemical compound CC*1=CC=C(C=CC=C2)C2=CC1 0.000 description 10
- 239000012467 final product Substances 0.000 description 10
- 238000002156 mixing Methods 0.000 description 10
- 238000003756 stirring Methods 0.000 description 10
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 9
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- 229960001760 dimethyl sulfoxide Drugs 0.000 description 9
- 229920001971 elastomer Polymers 0.000 description 9
- 239000002245 particle Substances 0.000 description 9
- 239000005060 rubber Substances 0.000 description 9
- 230000000704 physical effect Effects 0.000 description 8
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 7
- 239000000376 reactant Substances 0.000 description 7
- FRGPKMWIYVTFIQ-UHFFFAOYSA-N triethoxy(3-isocyanatopropyl)silane Chemical compound CCO[Si](OCC)(OCC)CCCN=C=O FRGPKMWIYVTFIQ-UHFFFAOYSA-N 0.000 description 7
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 6
- 239000000654 additive Substances 0.000 description 6
- 230000002411 adverse Effects 0.000 description 6
- 229910052786 argon Inorganic materials 0.000 description 6
- 238000013329 compounding Methods 0.000 description 6
- 150000002431 hydrogen Chemical class 0.000 description 6
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 6
- 239000000543 intermediate Substances 0.000 description 6
- 229910052709 silver Inorganic materials 0.000 description 6
- 239000004332 silver Substances 0.000 description 6
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 5
- 239000012071 phase Substances 0.000 description 5
- 229920003023 plastic Polymers 0.000 description 5
- 239000004033 plastic Substances 0.000 description 5
- 238000002360 preparation method Methods 0.000 description 5
- 230000009257 reactivity Effects 0.000 description 5
- WADSJYLPJPTMLN-UHFFFAOYSA-N 3-(cycloundecen-1-yl)-1,2-diazacycloundec-2-ene Chemical compound C1CCCCCCCCC=C1C1=NNCCCCCCCC1 WADSJYLPJPTMLN-UHFFFAOYSA-N 0.000 description 4
- 150000008064 anhydrides Chemical class 0.000 description 4
- 125000000524 functional group Chemical group 0.000 description 4
- 239000011521 glass Substances 0.000 description 4
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 4
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- 229920005992 thermoplastic resin Polymers 0.000 description 4
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 4
- RUGHUJBHQWALKM-UHFFFAOYSA-N 1,2,2-triphenylethylbenzene Chemical group C1=CC=CC=C1C(C=1C=CC=CC=1)C(C=1C=CC=CC=1)C1=CC=CC=C1 RUGHUJBHQWALKM-UHFFFAOYSA-N 0.000 description 3
- LXBGSDVWAMZHDD-UHFFFAOYSA-N 2-methyl-1h-imidazole Chemical compound CC1=NC=CN1 LXBGSDVWAMZHDD-UHFFFAOYSA-N 0.000 description 3
- QWVGKYWNOKOFNN-UHFFFAOYSA-N Cc(cccc1)c1O Chemical compound Cc(cccc1)c1O QWVGKYWNOKOFNN-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 230000000052 comparative effect Effects 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 3
- 230000035484 reaction time Effects 0.000 description 3
- LTVUCOSIZFEASK-MPXCPUAZSA-N (3ar,4s,7r,7as)-3a-methyl-3a,4,7,7a-tetrahydro-4,7-methano-2-benzofuran-1,3-dione Chemical compound C([C@H]1C=C2)[C@H]2[C@H]2[C@]1(C)C(=O)OC2=O LTVUCOSIZFEASK-MPXCPUAZSA-N 0.000 description 2
- HECLRDQVFMWTQS-RGOKHQFPSA-N 1755-01-7 Chemical compound C1[C@H]2[C@@H]3CC=C[C@@H]3[C@@H]1C=C2 HECLRDQVFMWTQS-RGOKHQFPSA-N 0.000 description 2
- RNFJDJUURJAICM-UHFFFAOYSA-N 2,2,4,4,6,6-hexaphenoxy-1,3,5-triaza-2$l^{5},4$l^{5},6$l^{5}-triphosphacyclohexa-1,3,5-triene Chemical compound N=1P(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP=1(OC=1C=CC=CC=1)OC1=CC=CC=C1 RNFJDJUURJAICM-UHFFFAOYSA-N 0.000 description 2
- YTWBFUCJVWKCCK-UHFFFAOYSA-N 2-heptadecyl-1h-imidazole Chemical compound CCCCCCCCCCCCCCCCCC1=NC=CN1 YTWBFUCJVWKCCK-UHFFFAOYSA-N 0.000 description 2
- KXGFMDJXCMQABM-UHFFFAOYSA-N 2-methoxy-6-methylphenol Chemical compound [CH]OC1=CC=CC([CH])=C1O KXGFMDJXCMQABM-UHFFFAOYSA-N 0.000 description 2
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 2
- RNLHGQLZWXBQNY-UHFFFAOYSA-N 3-(aminomethyl)-3,5,5-trimethylcyclohexan-1-amine Chemical compound CC1(C)CC(N)CC(C)(CN)C1 RNLHGQLZWXBQNY-UHFFFAOYSA-N 0.000 description 2
- WVRNUXJQQFPNMN-VAWYXSNFSA-N 3-[(e)-dodec-1-enyl]oxolane-2,5-dione Chemical compound CCCCCCCCCC\C=C\C1CC(=O)OC1=O WVRNUXJQQFPNMN-VAWYXSNFSA-N 0.000 description 2
- MWSKJDNQKGCKPA-UHFFFAOYSA-N 6-methyl-3a,4,5,7a-tetrahydro-2-benzofuran-1,3-dione Chemical compound C1CC(C)=CC2C(=O)OC(=O)C12 MWSKJDNQKGCKPA-UHFFFAOYSA-N 0.000 description 2
- DLFVBJFMPXGRIB-UHFFFAOYSA-N Acetamide Chemical compound CC(N)=O DLFVBJFMPXGRIB-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- MQJKPEGWNLWLTK-UHFFFAOYSA-N Dapsone Chemical compound C1=CC(N)=CC=C1S(=O)(=O)C1=CC=C(N)C=C1 MQJKPEGWNLWLTK-UHFFFAOYSA-N 0.000 description 2
- RPNUMPOLZDHAAY-UHFFFAOYSA-N Diethylenetriamine Chemical compound NCCNCCN RPNUMPOLZDHAAY-UHFFFAOYSA-N 0.000 description 2
- 239000002841 Lewis acid Substances 0.000 description 2
- 229920000459 Nitrile rubber Polymers 0.000 description 2
- 239000004721 Polyphenylene oxide Substances 0.000 description 2
- QHWKHLYUUZGSCW-UHFFFAOYSA-N Tetrabromophthalic anhydride Chemical compound BrC1=C(Br)C(Br)=C2C(=O)OC(=O)C2=C1Br QHWKHLYUUZGSCW-UHFFFAOYSA-N 0.000 description 2
- FDLQZKYLHJJBHD-UHFFFAOYSA-N [3-(aminomethyl)phenyl]methanamine Chemical compound NCC1=CC=CC(CN)=C1 FDLQZKYLHJJBHD-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 150000008065 acid anhydrides Chemical class 0.000 description 2
- 229920000800 acrylic rubber Polymers 0.000 description 2
- 125000002723 alicyclic group Chemical group 0.000 description 2
- 150000004982 aromatic amines Chemical class 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 229910010293 ceramic material Inorganic materials 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 238000005336 cracking Methods 0.000 description 2
- 229930003836 cresol Natural products 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 2
- XXBDWLFCJWSEKW-UHFFFAOYSA-N dimethylbenzylamine Chemical compound CN(C)CC1=CC=CC=C1 XXBDWLFCJWSEKW-UHFFFAOYSA-N 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 150000002118 epoxides Chemical group 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 239000003063 flame retardant Substances 0.000 description 2
- 239000005350 fused silica glass Substances 0.000 description 2
- 239000004845 glycidylamine epoxy resin Substances 0.000 description 2
- 150000004820 halides Chemical group 0.000 description 2
- 150000004678 hydrides Chemical class 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 150000007517 lewis acids Chemical class 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 239000007769 metal material Substances 0.000 description 2
- 229910044991 metal oxide Inorganic materials 0.000 description 2
- 150000004706 metal oxides Chemical class 0.000 description 2
- RTWNYYOXLSILQN-UHFFFAOYSA-N methanediamine Chemical compound NCN RTWNYYOXLSILQN-UHFFFAOYSA-N 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 239000012044 organic layer Substances 0.000 description 2
- 238000004806 packaging method and process Methods 0.000 description 2
- 229920001568 phenolic resin Polymers 0.000 description 2
- 239000005011 phenolic resin Substances 0.000 description 2
- 229920000058 polyacrylate Polymers 0.000 description 2
- 229920000570 polyether Polymers 0.000 description 2
- CYIDZMCFTVVTJO-UHFFFAOYSA-N pyromellitic acid Chemical compound OC(=O)C1=CC(C(O)=O)=C(C(O)=O)C=C1C(O)=O CYIDZMCFTVVTJO-UHFFFAOYSA-N 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 239000010703 silicon Substances 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- FAGUFWYHJQFNRV-UHFFFAOYSA-N tetraethylenepentamine Chemical compound NCCNCCNCCNCCN FAGUFWYHJQFNRV-UHFFFAOYSA-N 0.000 description 2
- AGGKEGLBGGJEBZ-UHFFFAOYSA-N tetramethylenedisulfotetramine Chemical compound C1N(S2(=O)=O)CN3S(=O)(=O)N1CN2C3 AGGKEGLBGGJEBZ-UHFFFAOYSA-N 0.000 description 2
- 239000010409 thin film Substances 0.000 description 2
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 2
- 229920002554 vinyl polymer Polymers 0.000 description 2
- 125000006274 (C1-C3)alkoxy group Chemical group 0.000 description 1
- CPEOJEBZAIYHGD-UHFFFAOYSA-N 1,2-diazacycloundecene Chemical compound C1CCCCN=NCCCC1 CPEOJEBZAIYHGD-UHFFFAOYSA-N 0.000 description 1
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 description 1
- WZCQRUWWHSTZEM-UHFFFAOYSA-N 1,3-phenylenediamine Chemical compound NC1=CC=CC(N)=C1 WZCQRUWWHSTZEM-UHFFFAOYSA-N 0.000 description 1
- OQURWGJAWSLGQG-UHFFFAOYSA-N 1-isocyanatopropane Chemical compound CCCN=C=O OQURWGJAWSLGQG-UHFFFAOYSA-N 0.000 description 1
- ZDZHCHYQNPQSGG-UHFFFAOYSA-N 1-naphthalen-1-ylnaphthalene Chemical compound C1=CC=C2C(C=3C4=CC=CC=C4C=CC=3)=CC=CC2=C1 ZDZHCHYQNPQSGG-UHFFFAOYSA-N 0.000 description 1
- ICXMPEKOMBEQFH-UHFFFAOYSA-N 2-(2h-pyrazin-1-yl)ethanamine Chemical compound NCCN1CC=NC=C1 ICXMPEKOMBEQFH-UHFFFAOYSA-N 0.000 description 1
- ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 2-phenyl-1h-imidazole Chemical compound C1=CNC(C=2C=CC=CC=2)=N1 ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- LLEASVZEQBICSN-UHFFFAOYSA-N 2-undecyl-1h-imidazole Chemical compound CCCCCCCCCCCC1=NC=CN1 LLEASVZEQBICSN-UHFFFAOYSA-N 0.000 description 1
- XYXBMCIMPXOBLB-UHFFFAOYSA-N 3,4,5-tris(dimethylamino)-2-methylphenol Chemical compound CN(C)C1=CC(O)=C(C)C(N(C)C)=C1N(C)C XYXBMCIMPXOBLB-UHFFFAOYSA-N 0.000 description 1
- ULKLGIFJWFIQFF-UHFFFAOYSA-N 5K8XI641G3 Chemical compound CCC1=NC=C(C)N1 ULKLGIFJWFIQFF-UHFFFAOYSA-N 0.000 description 1
- LCFVJGUPQDGYKZ-UHFFFAOYSA-N Bisphenol A diglycidyl ether Chemical compound C=1C=C(OCC2OC2)C=CC=1C(C)(C)C(C=C1)=CC=C1OCC1CO1 LCFVJGUPQDGYKZ-UHFFFAOYSA-N 0.000 description 1
- KSSJBGNOJJETTC-UHFFFAOYSA-N COC1=C(C=CC=C1)N(C1=CC=2C3(C4=CC(=CC=C4C=2C=C1)N(C1=CC=C(C=C1)OC)C1=C(C=CC=C1)OC)C1=CC(=CC=C1C=1C=CC(=CC=13)N(C1=CC=C(C=C1)OC)C1=C(C=CC=C1)OC)N(C1=CC=C(C=C1)OC)C1=C(C=CC=C1)OC)C1=CC=C(C=C1)OC Chemical compound COC1=C(C=CC=C1)N(C1=CC=2C3(C4=CC(=CC=C4C=2C=C1)N(C1=CC=C(C=C1)OC)C1=C(C=CC=C1)OC)C1=CC(=CC=C1C=1C=CC(=CC=13)N(C1=CC=C(C=C1)OC)C1=C(C=CC=C1)OC)N(C1=CC=C(C=C1)OC)C1=C(C=CC=C1)OC)C1=CC=C(C=C1)OC KSSJBGNOJJETTC-UHFFFAOYSA-N 0.000 description 1
- RKIRQSVYPLZQLD-ZETCQYMHSA-N C[C@@H](CC=C1)C=C1OC Chemical compound C[C@@H](CC=C1)C=C1OC RKIRQSVYPLZQLD-ZETCQYMHSA-N 0.000 description 1
- HRSLYNJTMYIRHM-UHFFFAOYSA-N Cc1cc(-c(cc2C)cc(C)c2OCC2OC2)cc(C)c1OCC1OC1 Chemical compound Cc1cc(-c(cc2C)cc(C)c2OCC2OC2)cc(C)c1OCC1OC1 HRSLYNJTMYIRHM-UHFFFAOYSA-N 0.000 description 1
- 241000238557 Decapoda Species 0.000 description 1
- OHKXVJWXIQQZCI-ZCFIWIBFSA-N NO[C@@H]1C=CC=CC1 Chemical compound NO[C@@H]1C=CC=CC1 OHKXVJWXIQQZCI-ZCFIWIBFSA-N 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 229920002732 Polyanhydride Polymers 0.000 description 1
- 239000005062 Polybutadiene Substances 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- UKLDJPRMSDWDSL-UHFFFAOYSA-L [dibutyl(dodecanoyloxy)stannyl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCC UKLDJPRMSDWDSL-UHFFFAOYSA-L 0.000 description 1
- DPRMFUAMSRXGDE-UHFFFAOYSA-N ac1o530g Chemical compound NCCN.NCCN DPRMFUAMSRXGDE-UHFFFAOYSA-N 0.000 description 1
- 239000011354 acetal resin Substances 0.000 description 1
- DHKHKXVYLBGOIT-UHFFFAOYSA-N acetaldehyde Diethyl Acetal Natural products CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 1
- 125000002777 acetyl group Chemical class [H]C([H])([H])C(*)=O 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 125000004450 alkenylene group Chemical group 0.000 description 1
- BHELZAPQIKSEDF-UHFFFAOYSA-N allyl bromide Chemical compound BrCC=C BHELZAPQIKSEDF-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 239000000010 aprotic solvent Substances 0.000 description 1
- MZTZYHFBBOZYFC-UHFFFAOYSA-N benzene;9h-fluorene Chemical compound C1=CC=CC=C1.C1=CC=C2CC3=CC=CC=C3C2=C1 MZTZYHFBBOZYFC-UHFFFAOYSA-N 0.000 description 1
- 239000012965 benzophenone Substances 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000000536 complexating effect Effects 0.000 description 1
- 238000010668 complexation reaction Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 239000011889 copper foil Substances 0.000 description 1
- 239000011258 core-shell material Substances 0.000 description 1
- 239000007822 coupling agent Substances 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 229910002026 crystalline silica Inorganic materials 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 239000012975 dibutyltin dilaurate Substances 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- GLUUGHFHXGJENI-UHFFFAOYSA-N diethylenediamine Natural products C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- KZTYYGOKRVBIMI-UHFFFAOYSA-N diphenyl sulfone Chemical compound C=1C=CC=CC=1S(=O)(=O)C1=CC=CC=C1 KZTYYGOKRVBIMI-UHFFFAOYSA-N 0.000 description 1
- MGHPNCMVUAKAIE-UHFFFAOYSA-N diphenylmethanamine Chemical compound C=1C=CC=CC=1C(N)C1=CC=CC=C1 MGHPNCMVUAKAIE-UHFFFAOYSA-N 0.000 description 1
- ZZTCPWRAHWXWCH-UHFFFAOYSA-N diphenylmethanediamine Chemical compound C=1C=CC=CC=1C(N)(N)C1=CC=CC=C1 ZZTCPWRAHWXWCH-UHFFFAOYSA-N 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 239000008393 encapsulating agent Substances 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- ANSXAPJVJOKRDJ-UHFFFAOYSA-N furo[3,4-f][2]benzofuran-1,3,5,7-tetrone Chemical compound C1=C2C(=O)OC(=O)C2=CC2=C1C(=O)OC2=O ANSXAPJVJOKRDJ-UHFFFAOYSA-N 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- FLBJFXNAEMSXGL-UHFFFAOYSA-N het anhydride Chemical compound O=C1OC(=O)C2C1C1(Cl)C(Cl)=C(Cl)C2(Cl)C1(Cl)Cl FLBJFXNAEMSXGL-UHFFFAOYSA-N 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 239000011256 inorganic filler Substances 0.000 description 1
- 229910003475 inorganic filler Inorganic materials 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- XLSZMDLNRCVEIJ-UHFFFAOYSA-N methylimidazole Natural products CC1=CNC=N1 XLSZMDLNRCVEIJ-UHFFFAOYSA-N 0.000 description 1
- 239000003094 microcapsule Substances 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 239000012778 molding material Substances 0.000 description 1
- ZCIJAGHWGVCOHJ-UHFFFAOYSA-N naphthalene phenol Chemical compound C1(=CC=CC=C1)O.C1(=CC=CC=C1)O.C1=CC=CC2=CC=CC=C12.C1(=CC=CC=C1)O ZCIJAGHWGVCOHJ-UHFFFAOYSA-N 0.000 description 1
- BDJRBEYXGGNYIS-UHFFFAOYSA-N nonanedioic acid Chemical compound OC(=O)CCCCCCCC(O)=O BDJRBEYXGGNYIS-UHFFFAOYSA-N 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- WWZKQHOCKIZLMA-UHFFFAOYSA-N octanoic acid Chemical class CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- MUMZUERVLWJKNR-UHFFFAOYSA-N oxoplatinum Chemical compound [Pt]=O MUMZUERVLWJKNR-UHFFFAOYSA-N 0.000 description 1
- KSOCVFUBQIXVDC-FMQUCBEESA-N p-azophenyltrimethylammonium Chemical compound C1=CC([N+](C)(C)C)=CC=C1\N=N\C1=CC=C([N+](C)(C)C)C=C1 KSOCVFUBQIXVDC-FMQUCBEESA-N 0.000 description 1
- 239000013034 phenoxy resin Substances 0.000 description 1
- 229920006287 phenoxy resin Polymers 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229910003446 platinum oxide Inorganic materials 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 229920002857 polybutadiene Polymers 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 239000009719 polyimide resin Substances 0.000 description 1
- 229920006254 polymer film Polymers 0.000 description 1
- 229920006324 polyoxymethylene Polymers 0.000 description 1
- 150000003141 primary amines Chemical group 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 125000001453 quaternary ammonium group Chemical group 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 230000002787 reinforcement Effects 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 239000003566 sealing material Substances 0.000 description 1
- 239000007790 solid phase Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 229920003048 styrene butadiene rubber Polymers 0.000 description 1
- 239000012756 surface treatment agent Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 150000003512 tertiary amines Chemical class 0.000 description 1
- JRMUNVKIHCOMHV-UHFFFAOYSA-M tetrabutylammonium bromide Chemical compound [Br-].CCCC[N+](CCCC)(CCCC)CCCC JRMUNVKIHCOMHV-UHFFFAOYSA-M 0.000 description 1
- 230000000930 thermomechanical effect Effects 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 229920001187 thermosetting polymer Polymers 0.000 description 1
- 239000004416 thermosoftening plastic Substances 0.000 description 1
- AYEKOFBPNLCAJY-UHFFFAOYSA-O thiamine pyrophosphate Chemical compound CC1=C(CCOP(O)(=O)OP(O)(O)=O)SC=[N+]1CC1=CN=C(C)N=C1N AYEKOFBPNLCAJY-UHFFFAOYSA-O 0.000 description 1
- SRPWOOOHEPICQU-UHFFFAOYSA-N trimellitic anhydride Chemical compound OC(=O)C1=CC=C2C(=O)OC(=O)C2=C1 SRPWOOOHEPICQU-UHFFFAOYSA-N 0.000 description 1
- 239000004034 viscosity adjusting agent Substances 0.000 description 1
- 239000011800 void material Substances 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
Definitions
- New novolac curing agents having alkoxysilyl groups preparation methods thereof, compositions comprising the same, cured products and uses thereof
- the present invention is a novel novolac curing agent having an alkoxysilyl group exhibiting excellent heat resistance in the composite (hereinafter also referred to as 'new novolac curing agent' or 'novolak curing agent of the present invention'), a method for preparing the same, a composition comprising the same, It relates to a cured product thereof and its use.
- the present invention has in the composite material, an excellent heat resistance, specifically, the low thermal expansion characteristics, (also which includes a Tg less does not exhibit a glass transition temperature) Glass jeonyieun lift, and a group, alkoxysilyl group that exhibits good flame retardant New novolac curing agents, methods for preparing the same, compositions comprising the same, cured products thereof and uses thereof.
- the low thermal expansion characteristics also which includes a Tg less does not exhibit a glass transition temperature
- Glass jeonyieun lift and a group, alkoxysilyl group that exhibits good flame retardant New novolac curing agents, methods for preparing the same, compositions comprising the same, cured products thereof and uses thereof.
- the thermal expansion coefficient of epoxy resin is about 50 ⁇ 80 ppm / ° C, and the thermal expansion coefficient of ceramic material and metal material which are inorganic particles (for example, the thermal expansion coefficient of silicon is 3 ⁇ 5 ppm / ° C, the thermal expansion coefficient of copper is 17ppm / ° C.)
- the coefficient of thermal expansion is very large, several times to several ten times.
- the coefficient of thermal expansion significantly limits the design and processing of parts.
- new epoxy resins generally have (1) complexing epoxy resins with inorganic particles (inorganic fillers) and / or glass fibers or (2) reduced CTEs.
- a method of designing has been used.
- inorganic particles as epoxy resin and filler In the case of complexation, the use of a large amount of non-subsidiary silica particles of about 2-30 ⁇ size can reduce CTE.
- the improved heat resistance in the composite material specifically, there is provided a new novolak curing agent having an alkoxysilyl group representing a low CTE and a high glass transition temperature and excellent flame retardancy.
- the improved composites specifically low CTE, high glass transition temperature and good flame retardancy A method for producing a new novolac curing agent having an alkoxysilyl group is provided.
- composition comprising a new novolac curing agent having an alkoxysilyl group which exhibits improved heat resistance in the composite, specifically low CTE, high glass transition temperature and good flame retardancy.
- a cured product of a composition comprising a new novolac curing agent having an alkoxysilyl group exhibiting improved heat resistance properties, in particular low CTE, high glass transition temperature and excellent flame retardancy in the composite.
- curing agent which has an alkoxylsilyl group is provided.
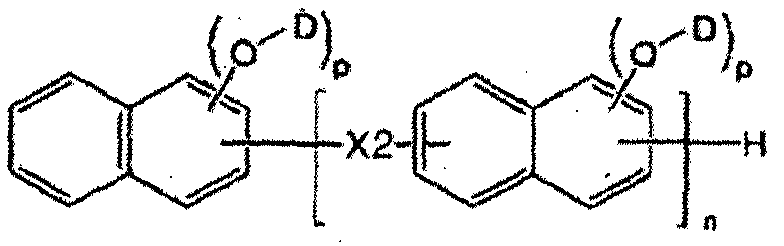
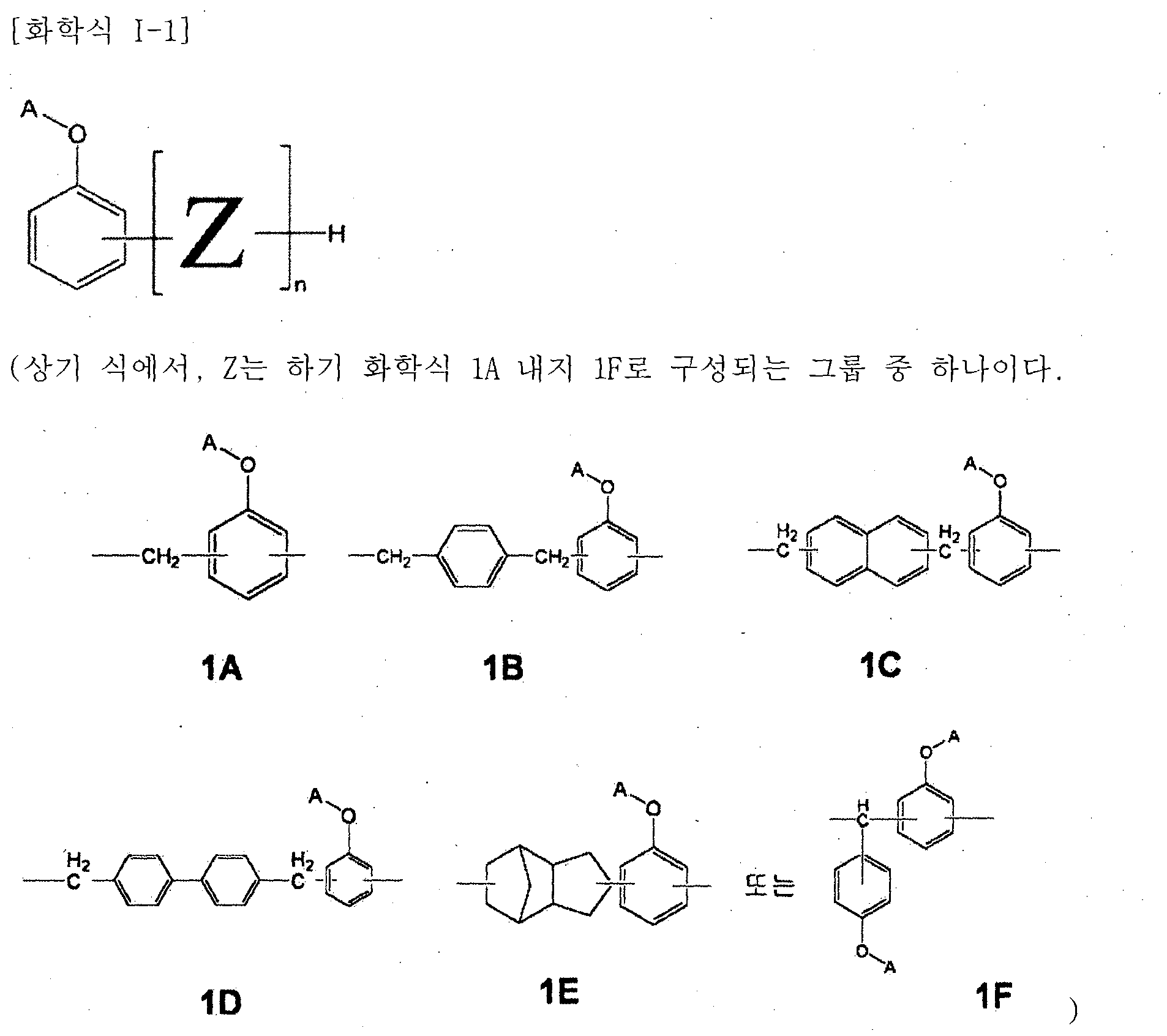
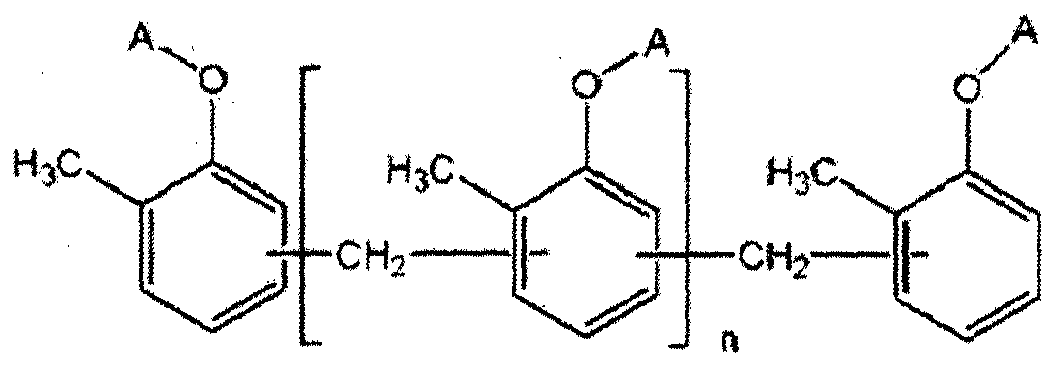
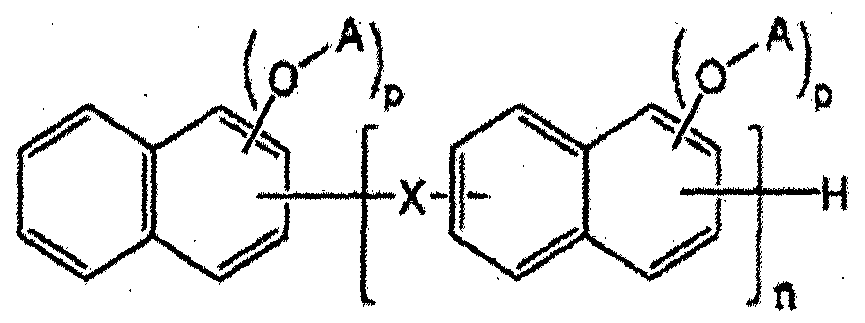
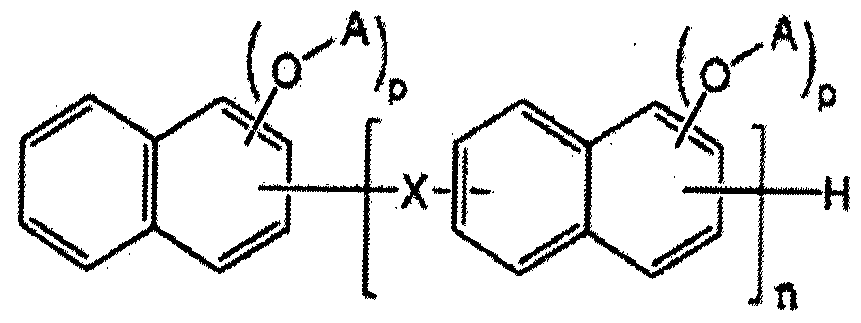
- a novolak curing agent having at least one alkoxysilyl group selected from the group consisting of the following formulas (1-1) to (1-4). [Formula 1-1]
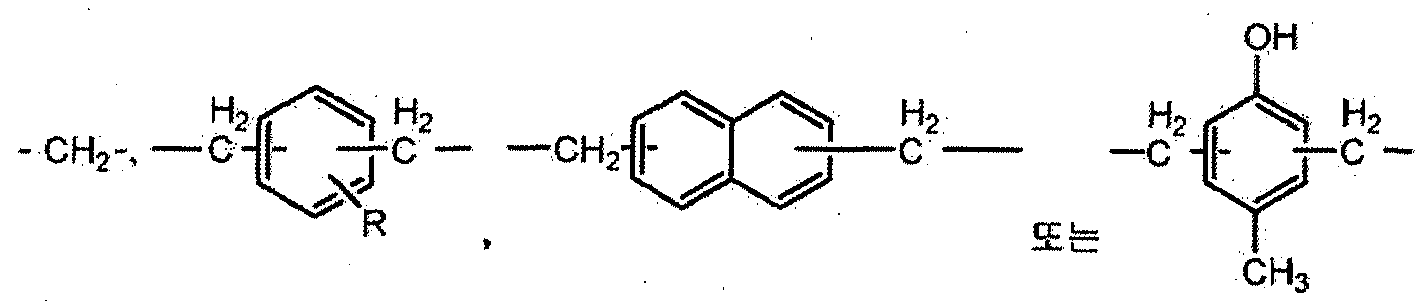
- Z is one of the groups consisting of the following Chemical Formulas 1A to 1F.
- X-cr And R is a C1-C10 straight or branched alkyl group.
- At least one A is of formula A2 or A3, if at least one is A2, the remaining A is of formula B2 or hydrogen, if at least one is A3, the remaining A is hydrogen, and n is an integer of at least 1 .
- R 3 is a straight or branched chain alkoxy group having 1 to 5 carbon atoms, the remainder is a straight chain black or branched alkyl group having 1 to 10 carbon atoms, m is an integer of 3 to 10 to be.
- X is one group group consisting of the following formulas 2A to 2F.
- R is a C1-C10 linear or branched alkyl group.
- D may be represented by Formula B2, and the remainder may be hydrogen.
- N is an integer of 1 or more. to be.
- X is CI, Br, I,-S0 2 ⁇ CH 3 ,-S0 2 _CF 3 , or -0-S0 2 -C 6 — CH 3 )
- R a to R c is a straight chain or molecular chain C1-C5 alkoxy group, and the rest is a straight chain black branched CI—C10 alkyl group.
- X is And Where R is a C1-C10 straight or branched alkyl group.
- R 3 is a linear or branched alkoxy group having 1 to 5 carbon atoms, the remainder is a straight chain black or branched alkyl group having 1 to 10 carbon atoms, and m is an integer of 3 to 10.
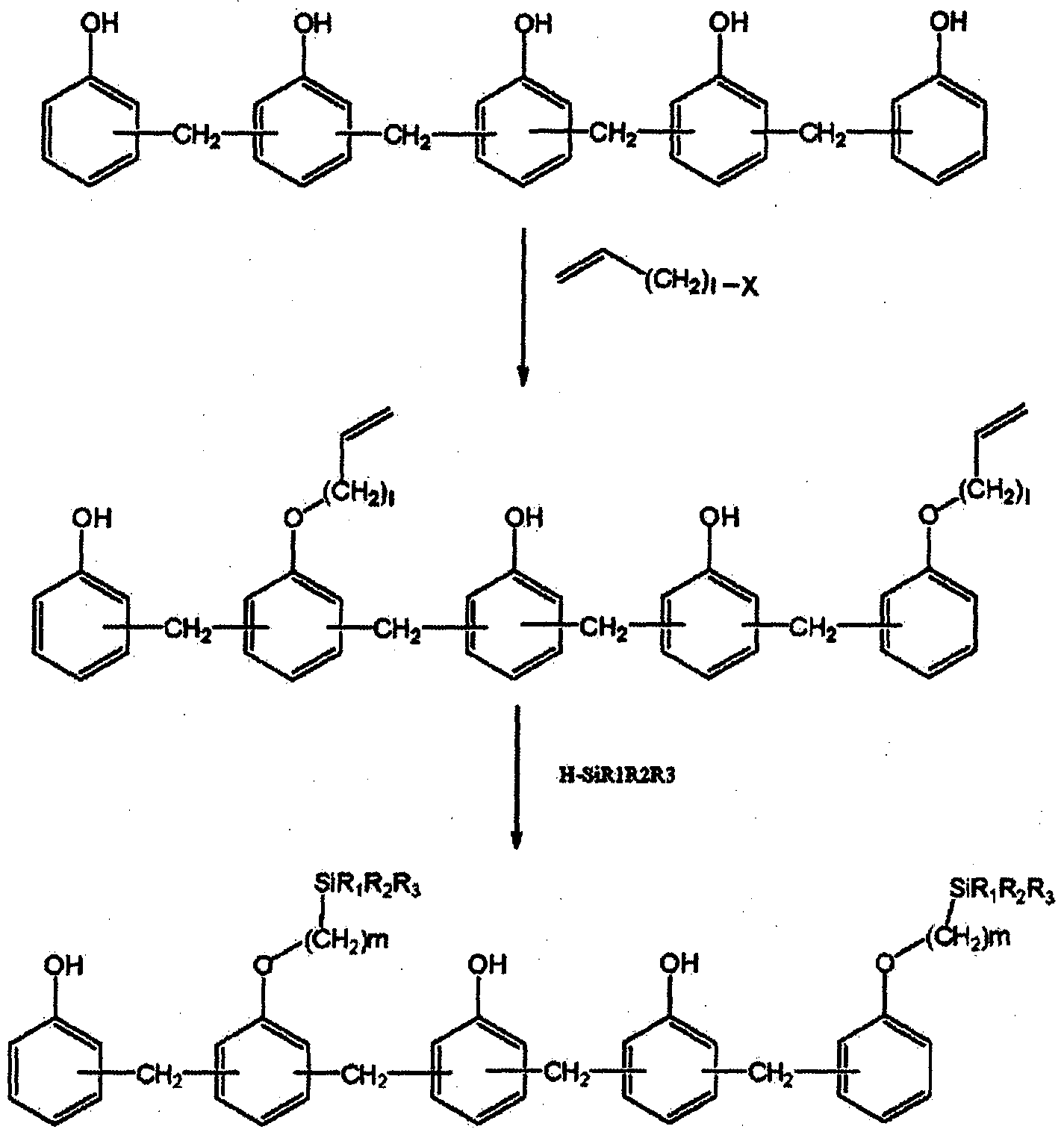
- the first step corresponds to one equivalent of the hydroxy group of the starting material of one of Formulas IA-1 to IA-4.
- a process for producing a novolak curing agent having an alkoxysilyl group which is carried out by reacting an alkylene group of the alkenyl compound of the formula [pi] to 0.01 to 10 equivalents.
- the first step is a method for preparing a novolak curing agent having an alkoxysilyl group, which is performed by reacting for 120 hours at -20 ° C to 100 ° C for 1 hour.
- the second step is an alkoxysilane equivalent to 1 equivalent of the alkenyl group of one of the intermediates of Formulas IB-1 to IB-4.
- a process for producing a novolac curing agent having an alkoxysilyl group which is carried out by reacting to 0.1 equivalents to 5 equivalents.
- the second step is a method for preparing a noblock curing agent having an alkoxysilyl group, which is carried out by reacting at 20 ° C. to 12 CTC for 1 to 72 hours.
- a starting material of any one of the following formulas IA-1 to IA-4 is reacted with an alkoxysilane of the following formula ⁇ to have a structure of formula A3.
- X is one of the groups consisting of the following Formulas 2A to 2F.
- n is an integer of 1 or more.
- R 3 is a straight or branched chain alkoxy group having 1 to 5 carbon atoms, the rest is a straight or branched chain alkyl group having 1 to 10 carbon atoms, and m is an integer of 3 to 10.
- X is And R is a C1-C10 straight or branched alkyl group
- the alkoxysilylation step is performed so that the alkoxysilane is 0.1 to 5 equivalents to 1 equivalent of the hydroxy group of the starting material of one of Formulas I A-1 to IA-4. the production method of the novolac curing agent having an "alkoxysilyl group provided that banung was performed.
- the alkoxysilylation step provides a method for preparing a novolac curing agent having an alkoxysilyl group, which is performed by reacting at -20 ° C to 120 ° C for 1 to 72 hours.
- a composition comprising an epoxy resin and a novolac curing agent having at least one alkoxysilyl group selected from the group consisting of the following formulas 1-1 to 1-4.
- X is And R is a straight or branched chain alkyl group of Cl-ClO.
- At least one A is of formula A2 or A3 and at least one is A2 Remaining A is the following formula B2 or hydrogen, when at least one is A3, the remaining A is hydrogen, n is an integer of 1 or more.
- R 3 is a straight or branched chain alkoxy group having 1 to 5 carbon atoms, the rest is a straight or branched chain alkyl group having 1 to 10 carbon atoms, and m is an integer of 3 to 10. .
- the epoxy resin is a glycidyl ether epoxy resin, glycidyl epoxy resin, glycidylamine epoxy resin And at least one composition selected from the group consisting of glycidyl ester epoxy resins.
- the epoxy resin has a core structure of bisphenol, biphenyl, naphthalene, benzene, thiodiphenol, fluorene, anthracene, isocyanurate, and triphenylmethane.
- a composition having 1,1,2,2-tetraphenylethane, tetraphenylmethane, 4,4'-diaminodiphenylmethane, aminophenol, alicyclic, aliphatic, or novolak unit is provided.
- the epoxy resin is provided with a composition comprising an epoxy resin with or without an alkoxysilyl group.
- a composition further comprising inorganic particles or / and fibers as a filler is provided.
- the inorganic particles are at least one composition selected from the group consisting of silica, zirconia, titania, alumina, silicon nitride, aluminum nitride, and silsesquioxane.
- the inorganic particles are provided with a composition containing 5 wt% to 95% based on the total weight of solids of the composition.
- the inorganic particles A composition is provided that includes 30 wt% to 95% based on the total weight of solids.
- the inorganic particles are provided in a range of 5 wt% to 6 (1 ⁇ 2 «based on the total weight of solids of the composition.
- the fiber is glass fiber selected from the group consisting of E glass fiber, T glass fiber, S glass fiber, NE glass fiber, H glass fiber, and quartz; and liquid crystal polyester fiber, polyethylene terephthalate fiber, wholly aromatic fiber, polybenzoxazole fiber, nylon fiber, polyethylene naphthalate fiber, polypropylene fiber, polyethersulfone fiber, polyvinylidene fluoride fiber, polyethylene sulfide fibers, ⁇ and a polyether selected from the group consisting of ketone fiber At least one composition selected from the group consisting of organic fibers is provided.
- the fiber was provided with the composition which is E glass fiber .
- the composition is the T glass fiber in 19th aspect.
- a composition is provided which comprises from 10 wt% to 90 3 ⁇ 4 with respect to the total weight of solids.
- a composition further comprising inorganic particles is provided.
- a composition further comprising an alkoxysilyl group semi-aqueous catalyst is provided.
- the alkoxysilyl group reaction catalyst is selected from nitric acid, sulfuric acid, hydrochloric acid, acetic acid, phosphoric acid, ammonia, K0H, NH 4 0H, amine, transition metal alkoxide, and tin (t in) compound.
- At least one composition selected from the group consisting of is provided.
- the composition is provided in which the semi-aqueous catalyst is contained in an amount of 0.01 phr to 10 phr relative to the epoxy resin having an alkoxysilyl group.
- a composition further comprising water is provided.
- the electronic material containing the composition in any one of 10th-27th aspect is provided. According to the 29th opinion, the 10th to 27th opinions
- a substrate comprising. According to the thirtieth opinion, tenth to twenty-seventh opinion
- a film comprising is provided.
- a laminate comprising a metal worm on a base layer made of the composition of any one of the tenth to twenty-seventh aspects.
- a printed wiring board including a laminated board is provided.
- a semiconductor device including a printed wiring board is provided.
- a semiconductor packaging material comprising the composition of any one of the tenth to twenty-seventh aspects.
- a semiconductor device including a semiconductor packaging material is provided.
- an adhesive comprising the composition of any one of the tenth to twenty seventh aspects.
- a paint comprising the composition of any one of the crabs 10 to 27.
- a composite material comprising the composition of any one of the tenth to twenty seventh aspects.
- a prepreg comprising the composition of any one of the tenth to twenty-seventh symptoms.
- positioned at the prepreg is provided.
- the cured product of any one of the tenth to twenty-seventh symptom compositions is provided.
- a cured product of the composition having a thermal expansion coefficient of 60 ppm / ° C. or less is provided.
- the glass transition temperature does not represent a higher or a glass transition temperature greater than 100 ° C is provided in the.
- composition comprising a new novolac curing agent having an alkoxysilyl group according to the present invention is used in the composite and / or cured product to form an interfacial bond between an alkoxysilyl group and a filler (fibers and / or inorganic particles) and to react an alkoxysilyl group with an epoxy and a curing agent.
- the formation of further chemical bonds by means of improved heat resistance. That is, the CTE of the epoxy composite is reduced and the glass transition degree is increased or the glass transition temperature is not exhibited (hereinafter referred to as 'Tg lease').
- curing agent which has the alkoxy silyl group by this invention shows the outstanding flame retardance by introduction of the alkoxy silyl group.
- the novel novolak curing agent having an alkoxysilyl group according to the present invention is easy to be solidified (sol id) to an epoxy resin having an alkoxysilyl group, so that it is easy to be applied to a process requiring a solid phase sample, and brittleness of the cured product. (br it leness) and adhesion are improved.
- the composition according to the present invention to a metal film of a substrate, the chemical film of the functional group on the surface of the metal film and the alkoxysilyl group is applied to the metal film. Excellent adhesion.
- FIG. 1 is a result of measuring the length change with the temperature of the glass fiber composite body by Example 1 and the comparative example 1.
- FIG. 1 is a result of measuring the length change with the temperature of the glass fiber composite body by Example 1 and the comparative example 1.
- the present invention has improved heat resistance properties when forming a composite using a composition comprising a curing agent having an alkoxysilyl group, specifically having a low CTE and a high Tg (including Tg lease) and / or having an alkoxysilyl group having excellent flame retardancy in the cured product. It is to provide a new novolac curing agent, a method for preparing the same, a curing agent composition and a cured product comprising the same, and uses thereof.
- composite or epoxy composite refers to a cured product of a composition comprising an epoxy resin curing agent and a filler (fibers and / or inorganic particles).
- the term "cured or epoxy cured product” refers to a cured product of a composition comprising an epoxy resin and a curing agent.
- cured material of a composition is said.
- the cured product may include a semi-cargo.
- inorganic particles And / or a cured product reinforced with fibers so that the cured product has a broader meaning than the composite, but an inorganic particle. And / or a cured product reinforced with fibers may be understood to have the same meaning as the composite.
- the new novolac curing agent according to the present invention forms an interfacial bond with the surface of the filler (fibers and / or inorganic particles) and / or an alkoxysilyl group, an epoxide group and a phenol group and a chemical bond when the complex is formed by curing. Therefore, it shows low CTE and high glass transition temperature synergistic effect or Tg-less (less). Therefore, the dimensional stability is improved.
- the cured product including the novolac curing agent according to the present invention exhibits excellent flame retardancy.
- composition according to the present invention is applied to a chemically treated metal film, for example, copper foil, and the like, and the alkoxysilyl group of the curing agent is applied to the metal surface.
- a chemically treated metal film for example, copper foil, and the like
- the alkoxysilyl group of the curing agent is applied to the metal surface.
- it is chemically bonded with -0H group on the metal surface by treatment, it shows excellent adhesion with metal film.
- a novolak curing agent having any one alkoxysilyl group selected from the group consisting of the following formulas I '1 to 1-4. [Formula I ⁇ 1]
- Z is one of the groups consisting of the following Chemical Formulas 1A to 1F.
- X is And R is a straight or branched alkyl group of CI—CIO.
- At least one of the plurality of A is of formula A2 or A3; At least when one of A2, and when the remaining A are hydrogen or the formula B2, at least one of which is A3, and the remaining A are hydrogen.
- n is 1 or more, for example, the integer of 1-1000.
- R 3 and R 3 are an alkoxy group having 1 to 5 carbon atoms, preferably an alkoxy group having 3 to 3 carbon atoms. More preferably, it is a methoxy group or a special group. The remainder is a C1-C10 alkyl group, m is an integer of 3-10, Preferably it is an integer of 3-6.
- the alkoxy group and the alkyl group may be straight chain black or branched chain.
- an "alkoxy group” is a monovalent group which is —OR (R is an alkyl group), which may be linear or branched, In this specification, the branched chain and the branched chain may be used interchangeably.
- an "alkyl group” refers to a monovalent ent hydrocarbon group, which may be linear or branched, and preferably 1 It has a carbon number of 10 to 10, More preferably, it has a carbon number of 1 to 5. Furthermore, the alkoxysilyl group by one Embodiment of the said invention is mentioned.
- the novolac curing agent having exhibits low CTE and high glass transition temperature or Tg-less when forming a composite of the composition comprising the same.
- the novolac curing agent having an alkoxysilyl group represented by Chemical Formulas 1-1 to 1-4 may be synthesized by the following method.
- the manufacturing method of the novolak hardening agent which has an alkoxy silyl group mentioned later is simple and simple compared with the manufacturing method of the epoxy resin which has an alkoxy silyl group.
- a reaction of a hydroxy group in the starting material by reaction of a starting material of one of the following formulas IA-1 to ⁇ -4 (hereinafter, also referred to as 'starting material' or 'starting material IA') and alkenyl of the formula
- a starting material of one of the following formulas IA-1 to ⁇ -4 hereinafter, also referred to as 'starting material' or 'starting material IA'
- alkenyl of the formula At least one of the alkenylated intermediates of any one of formulas IB ′ 1 to IB-4 (hereinafter referred to as “intermediate IB”).
- X in Formula IA ′ 1 is one of a group consisting of Formulas 2A to 2F.
- R is a C1-C10 linear or branched alkyl group.
- n is an integer of 1 or more.
- n is an integer of 1 or more, for example, an integer of 1 to 1000, which includes all integers in these ranges and any other smaller range of integers. It means.
- p is 1 or 2.
- ⁇ is selected from the group consisting of the following formulas 3A to 3F One.
- R is a C1-C10 linear or branched alkyl group.
- At least one of a plurality of D may be represented by Formula B2, and the rest may be hydrogen.
- n is an integer of 1 or more.
- p is 1 or 2.
- 1 is an integer of 1-8, Preferably it is an integer of 1-4.
- Alkenylation the reaction of the first step, is carried out by reacting the starting material of one of the above formulas IA-1 to IA-4 with the alkenyl compound of the formula ⁇ , which reaction is carried out in the presence of a base and an optional solvent.
- the starting material and the alkenyl compound may be reacted in the presence of a base and an optional solvent such that the alkenyl compound corresponds to 1 equivalent of the hydroxy group of the starting material and the alkenyl group is from 0.1 to 10 equivalents.
- Get IB The reaction product is reacted with a stoichiometric equivalence ratio, and in consideration of this, the reaction product is reacted with the equivalence ratio to obtain a desired intermediate product IB.
- the reaction temperature and reaction time of the first step reaction are dependent on the type of reactant, but, for example, the intermediate product IB is obtained by reacting the reaction at 20 ° C. to 100 ° C. for 1 hour to 120 hours.
- bases examples include, but are not limited to, for example, KOH, NaOH, 2 C0 3 , Na 2 C0 3 , KHC0 3 , NaHC0 3) NaH, triethylamine, diisopropylethyl Amines. These bases may be used alone or in combination of two or more of them.
- the base may be used in an amount of 0.01 to 5 equivalents based on 1 equivalent of the hydroxy group of the starting material IA in terms of reaction efficiency.
- the solvent may optionally be used as necessary. For example, the solvent may not be used if the viscosity of the reactants at the reaction temperature is suitable for the reaction to proceed without reaction in the first step reaction.
- the reaction product can be dissolved well, and any organic solvent can be used as long as it can be easily removed after the reaction without adversely affecting the reaction.
- acetonitrile, tetrahydrofuran (THF), methyl ethyl ketone (MEK), dimethyl formamicle (DMF), dimethyl sul foxide (DMMS0), methylene chloride (MC), 3 ⁇ 40, alcohols and roluene can be used. have.
- the intermediate product IB is obtained by alkoxysilylation.
- curing agent of Formula 1-1 to 1-4 which has substituent A2 by one embodiment of the invention is obtained.
- This reaction can be carried out in the presence of a catalyst and any solvent, in which case a platinum catalyst is preferred. In the second stage reaction.
- the alkoxysilane of the general formula ⁇ is 0 to 1 equivalent of the alkenyl group of the intermediate product IB.
- Intermediate IB is reacted with an alkoxysilane of the formula
- R a to R c is a C1-C5 alkoxy group, preferably a C1-C3 alkoxy group, more preferably a mesophilic or ethoxy group and the remainder is an ⁇ - ⁇ alkyl group, the alkoxy group And the alkyl group may be straight chain black branched chain.
- the reaction temperature and reaction time of the second stage reaction vary depending on the reactant, but for example, a novolak curing agent of the formulas 1-1 to 1-4 having a substituent A2 by reacting for 1 hour to 72 hours at 2CTC to 120 ° C. Is obtained.
- the platinum catalyst that can be used in the second reaction reaction is not limited thereto.
- a platinum catalyst of Pt3 ⁇ 4 or H 2 PtCl 6 may be used.
- the platinum catalyst is preferably used in an amount of lxlO— 4 to 0.05 equivalents based on 1 equivalent of the alkenyl group of the intermediate product IB in terms of reaction efficiency.
- the solvent in the second stage reaction can optionally be used as needed.
- a separate water banung viscosity at the reaction temperature without a solvent, in the "second reaction step suitable for the reaction proceeds may not use a solvent.
- a separate solvent is not required, which can be easily determined by those skilled in the art.
- the reaction product can be dissolved well, and any aprotic solvent can be used as long as it can be easily removed after the reaction without any adverse effect on the reaction.
- any aprotic solvent can be used as long as it can be easily removed after the reaction without any adverse effect on the reaction.
- toluene, acetonitrile, tetrahydrofuran (THF), methyl ethyl ketone ( ⁇ ), dimethyl formamide (DMF), dimethyl sul foxide (DMSO), methylene chloride (MC), and the like Can be used.
- solvents may be used alone or in combination of two or more.
- the amount of the solvent used is not particularly limited, and may be used in a suitable amount and / or concentration within a range in which the reactants are sufficiently dissolved and do not adversely affect the reaction, and those skilled in the art may select appropriately in consideration of this. have.
- An exemplary reaction method of the above-described preparation method (1) of Formula 1-1, wherein Z is IA, is as follows.
- At least one of the plurality of substituents A of Formulas 1-1 to 1-4 is a structure of Formula A2 (ie, an alkoxysilyl group), and the rest May be hydrogen.
- the number of alkoxysilyl groups in the formula (1-1) to (1-4) is the first of the reaction 1
- the equivalence and reaction of the alkenyl compound can be varied by controlling the number of hydroxy groups to be alkenylated by controlling the degree. This can be appropriately adjusted by those skilled in the art in view of the reaction from the above disclosure.
- the starting material is an alkoxysilylation of one of the formulas IA-1 to IA-4 to obtain a novolak curing agent of one of formulas 1-1 to 1-4 having a substituent of formula A3.
- the alkoxysilylation step may be carried out in the presence of any base and any solvent.
- the alkoxysilane having the isocyanate functional group of the starting material IA and the following formula ⁇ is 0.1 to 5 equivalents of the alkoxysilane having the isocyanate functional group of the formula ⁇ relative to 1 equivalent of the hydroxy group of the starting material IA.
- At least one of to is an alkoxy group having 1 to 5 carbon atoms, preferably an alkoxy group having 1 to 3 carbon atoms, more preferably a methoxy group or an hydroxy group to be.
- the hydroxy group is preferable in view of high reactivity, and the ethoxy value is preferable in view of sample stability and good reaction properties.
- the remainder is an alkyl group having 1 to 10 carbon atoms, wherein the alkoxy group and the alkyl group may be linear or branched, m is an integer of 3 to 10, preferably an integer of 3 to 6.
- the alkoxysilane having the starting material IA and the isocyanate functional group reacts with the hydroxy group of the starting material IA and the alkoxysilane in an equivalent ratio according to the stoichiometry, and thus taking into account the hydroxy group 1 of the starting material IA
- the alkoxysilane having the starting material IA and the isocyanate functional group is reacted such that the equivalent weight of the alkoxysilane is from 0.01 equivalents to 5 equivalents.
- the reaction temperature and reaction time of the alkoxysilylation step depend on the reaction water, but, for example, by reacting for 1 to 72 hours at 20 ° C. to 120 ° C.
- a novolac curing agent of any one of formulas I ′ 1 to 1-4 having substituents of ⁇ ((3 ⁇ 4) ⁇ ⁇ ⁇ 3 ⁇ 43 ⁇ 43 ⁇ 4 is obtained.
- the reaction of the alkoxysilylation step may be carried out in the presence of a base if necessary.
- the use of a separate base may result in a faster reaction reaction, but examples of the base that can be used include, but are not limited to, for example, K 2 CO 3 , Na 2 CO 3 , KHCOs, NaHC0 3 , triethylamine
- bases may be used alone or in combination of two or more.In the case of using a base, the base is 0.1 to 1 equivalent of hydroxy group of the starting material IA.
- the solvent may be optionally used in the reaction of the alkoxysilylation step, for example, without the need for a separate solvent in the alkoxysilylation step. If the viscosity of the reactants in silver is suitable for the reaction to proceed, the solvent may not be used, i.e. if the viscosity of the reactants is low enough that the mixing and stirring of the reaction product can proceed smoothly without solvent, then no additional solvent is required. In the case of using a solvent, the possible solvent can dissolve the reaction product well, and any aproticity can be easily removed after the reaction without any adverse effect on the reaction.
- a solvent may be used, including but not limited to, for example, toluene, acetonitrile, tetrahydrofuran (THF), methyl ethyl ketone (MEK), dimethyl formaniide (DMF), DMS0 (dimethyl sul foxide), Methylene chloride (MC) and the like may be used. These solvents may be used alone or in combination of two or more black.
- the amount of the solvent used is not particularly limited and may be used in a suitable amount within a range in which the reaction product is sufficiently dissolved and does not adversely affect the reaction, and those skilled in the art may appropriately select it.
- At least one of the plurality of substituents A of Formulas 1-1 to 1-4 is a structure of Formula A3 (ie, an alkoxysilyl group) and the rest is hydrogen Can be.
- the number of alkoxysilyl groups in Chemical Formulas 1-1 to 1-4 refers to the equivalent of alkoxysilane and reaction temperature in the alkoxysilylation step. It can be varied by controlling the number of hydroxy groups to be epoxidized by controlling. This can be appropriately adjusted by those skilled in the art in view of reactivity from the above disclosure.
- composition comprising an epoxy resin and at least one new novolac curing agent selected from the group consisting of the above formulas 1-1 to 1-4.
- compositions provided in the present invention are used for electronic materials, for example, but not limited to, for example, semiconductor substrates such as IC substrates, build-up films, encapsulation materials (packaging materials), printed wiring boards, and the like. It can be used for various uses such as parts, adhesives, paints and composite materials.
- any composition provided in the present invention may be a curable composition comprising an inorganic material.
- composition according to any of the above and later embodiments of the present invention comprises as a curing agent at least one new novolac curing agent selected from the group consisting of Formulas 1—1 to 1-4 according to embodiments of the present invention.
- a curing agent at least one new novolac curing agent selected from the group consisting of Formulas 1—1 to 1-4 according to embodiments of the present invention.
- any kind and / or combination of compositions known in the art are included, and the epoxy resin constituting the composition, the curing agent of the present invention and conventional curing agents, curing accelerators (catalysts), inorganic materials (fillers) (Eg inorganic particles and / or Fiber) and other additives, and the mixing ratio is not limited.
- the epoxy resin may be any conventional epoxy resin known in the art, but is not particularly limited, for example, glycidyl ether-based epoxy resin, glycidyl-based epoxy resin, glycidylamine-based It may be at least one selected from the group consisting of an epoxy resin and a glycidyl ester epoxy resin.
- the epoxy resin has a core structure of bisphenol, biphenyl, naphthalene, benzene, thiodiphenol, fluorene (fhiorene), anthracene, isocyanurate, triphenylmethane, 1,1,2,2-tetraphenyl Ethane, tetraphenylmethane, 4,4'-diaminodiphenyl methane, aminophenol alicyclic, aliphatic, or glycidyl ether epoxy resin having a novolak unit, glycidyl epoxy resin, glycidylamine epoxy At least one selected from the group consisting of a resin and a glycidyl ester epoxy resin.
- the conventional epoxy resin is bisphenol, biphenyl, naphthalene, fluorene benzene, thiodiphenol, fluorene, anthracene as a core structure.
- the existing and epoxy resins may or may not have an alkoxysilyl group.
- Any conventional curing agent generally known as a curing agent for resins, may be added, and as the existing curing agent, there is no particular limitation, for example, a novolac curing agent (a conventional novolak having no alkoxylsilyl group). Curing agents), amines, acid anhydrides and the like can be used. More specifically.
- amine curing agent aliphatic amines, alicyclic amines, aromatic amines, other amines and modified polyamines can be used, and amine compounds containing two or more primary amine groups can be used.
- amine curing agent examples include 4,4 ' ⁇ dimethylaniline (diamino diphenyl methane) (4 > 4'-Dimethylani 1 ine (di amino diphenyl methane, DAM or DDM), diamino diphenylsulfone (diamino di phenyl sul fone, DDS), m_ lung alkenylene di ⁇ min (m -phenyl ene diamine) aromatic amine, at least one selected from the group consisting of diethylene triamine (dieihylene tri amine, DETA), diethylene tetraamine ( diethylene tetramine, tri iethy lene tetramine (TETA), m-xylene diamine (MXDA), methane diamine (MDA), ⁇ , ⁇ '- Diethylenediamine (N, N '-di et hy 1 ened i amine, ⁇ , ⁇ ' -DEDA), t
- phenol curing agent examples include, but are not limited to, phenol novolac resins, cresol novolac resins, bisphenol A novolac resins, xylene novolac resins, triphenyl novolac resins, biphenyl novolac resins and dicyclopentadiene novolacs. Resins, phenol P-xylene resins, phenol 4, 4'-dimethylbiphenylene resins, naphthalene phenol novolac resins and the like.
- acid anhydride-based curing agents include, but are not limited to, dodecenyl succinic anhydride (DDSA), aliphatic acid anhydrides such as poly azelaic poly anhydride, and nucleohydrophthalic anhydrides.
- DDSA dodecenyl succinic anhydride
- aliphatic acid anhydrides such as poly azelaic poly anhydride
- nucleohydrophthalic anhydrides examples include, but are not limited to, dodecenyl succinic anhydride (DDSA), aliphatic acid anhydrides such as poly azelaic poly anhydride, and nucleohydrophthalic anhydrides.
- Alicyclics such as hydrhydroph halic anhydride (HHPA), methyl tetrahydrophthalic anhydride (MeTHPA) and methylnadic anhydride (MNA) Anhydride, trimellitic not hydride (Trimellitic Anhydride, TMA), pyromellitic dianhydride hydride (pyromellitic acid di anhydr ide, PMDA), benzophenone tetracarboxylic carboxy acid dianhydride hydride '(benzophenonetetracarboxyl ic di anhydr i de, BTDA Aromatic acid anhydrides, such as a), tetrabromophthalic anhydride (TBPA), and halogen-based acid anhydride compounds such as chlorendic anhydride.
- HHPA hydrhydroph halic anhydride
- MeTHPA methyl tetrahydrophthalic anhydride
- MNA methylnadic anhydride
- TMA trimellitic not hydride
- the degree of curing of the epoxy composite can be adjusted to the degree of reaction between the curing agent and the epoxy resin, and the content of the curing agent can be adjusted based on the concentration of the epoxy group of the epoxy resin according to the desired degree of curing.
- the equivalent reaction of the curing agent and the epoxy group is such that the epoxy equivalent / curing agent equivalent ratio is 0.5 to 2.0, and, for example, It is preferable to adjust and use a hardening
- Any curing agent that can be used to cure the epoxy resins not otherwise described herein also depends on the concentration of the total epoxy groups in the composition of the present invention depending on the desired degree of cure and based on the concentration of the epoxy functional groups and the semi-aromatic functional groups of the curing agent. Suitable amounts can be used in combination, as is common in the art.
- composition comprising the new novolac curing agent may further comprise an inorganic material (filler), for example inorganic particles and / or fibers, as needed, and for convenience the composition comprising the inorganic material " Composite compositions', as long as the composite composition comprises an epoxy resin, at least one new novolac curing agent and filler selected from the group consisting of Formulas 1-1 to 1-4.
- iller for example inorganic particles and / or fibers
- Composite compositions' as long as the composite composition comprises an epoxy resin, at least one new novolac curing agent and filler selected from the group consisting of Formulas 1-1 to 1-4.
- any type and / or the combination is understood to include a composition, the epoxy resin constituting the composition, the curing agent of the present invention, a conventional curing agent, a curing accelerator (catalyst) which, weapon material (filler) (e.g., Inorganic particles and / or fibers) and other additives are not limited to the type and compounding ratio.
- a curing accelerator catalyst
- weapon material e.g., Inorganic particles and / or fibers
- other additives are not limited to the type and compounding ratio.
- any inorganic particles known to be used for reinforcement may be used, including but not limited to, consisting of silica (including, for example, fused silica and crystalline silica), zirconia, titania, alumina, silicon nitride and aluminum nitride At least one kind selected from the group consisting of at least one metal oxide selected from the group and silsesquioxane may be used, and the inorganic particles may be used alone or as a mixture with two or more kinds. In this case, it is preferable to use fused silica, although either crushed or spherical can be used.
- the inorganic particles are not limited thereto, but inorganic particles having a particle size of 0.5 nm to several tens (for example, 50 to 100) may be used in consideration of the use of the composite, specifically, the dispersibility of the inorganic particles. Can be. Since the inorganic particles are dispersed in the epoxy resin, the inorganic particles of the above sizes are preferably used together due to the difference in dispersibility according to the particle size.
- the inorganic particles may be suitably added to the epoxy resin according to the appropriate viscosity and use required for reducing and applying CTE of the epoxy composite.
- the content of the inorganic particles is based on the total weight of solids of the composition, while in the case of epoxy cured products, based on the total weight of the epoxy cured product, 5 wt% to 95 wt 3 ⁇ 4, for example 5 wt% to 90 wt%, for example 10 wt% to 90wt3 ⁇ 4>, for example 30 wt% to 95wt, for example 30wt% to 90wt%, for example 5 wt% to 60wt%, for example 10wt to 50%.
- the composition of the present invention is a semiconductor encapsulant
- the content of the inorganic particles is, for example, relative to the total increase in the solid content of the composition, while in the case of epoxy cured products the total weight of the epoxy cured product. 30 wt% to 95 wt%, for example, 30 wt% to 90%.
- the content of the inorganic particles in consideration of the CTE value and strength of the substrate for example, relative to the weight of the total solids of the composition, on the other hand In the case of 5 wt% to 60 wt%, for example 10 wt% to 50%, based on the total weight of the epoxy cured product.
- the fiber is used as an inorganic material, any kind and dimension of fiber generally used in the art may be used.
- the fiber is not limited thereto, and any fiber generally used for improving physical properties of the thermosetting resin and cured material may be used. Specifically glass fiber, organic fiber or a mixture of middles can be used.
- glass fibers include glass fibers such as E glass fiber, T glass fiber, S glass fiber, NE glass fiber, D glass fiber, quartz glass fiber, and the like. Or T glass fibers.
- Liquid crystalline polyester fiber polyethylene terephthalate fiber, Selected from the group consisting of wholly aromatic fibers, polybenzoxazole fibers, nylon heart oil, polyethylene naphthalate fibers, polypropylene fibers, polyether sulfone fibers, polyvinylidene fluoride fibers, polyethylene sulfide fibers, polymoetheretherketone fibers At least one kind alone or more than two kinds of black may be used together.
- Composition comprising any new novolak light "agent of the present invention, for example, the content of fibers in the glass fiber composite composition 10wt3 ⁇ 4> to about 90wt% based on the total weight of the composition solids, for example, 30wt % To 70 wt%, and for example, 35 wt% to 65%.
- the fiber content is 10 wt% to 90 wt%, for example 30 wt% to 70 wt%, and for example 35 wt%, based on the total weight of the cured product. % To 65%.
- the resin content is lp w t% to 90 wt%, for example.
- the content of the fiber is in the above range in terms of heat resistance improvement and processability.
- the solid part except a fiber among the total solid content is normally called resin component, and in the composition containing a fiber, the quantity other than fiber is resin content (res in content).
- any of the compositions of the present invention comprising the fibers may further comprise inorganic particles, if necessary.
- the inorganic particles are in the range of lwt3 ⁇ 4 »to 70wt% based on the increase of the resin content in consideration of the improvement of physical properties and fairness. It can be formulated in amounts.
- the type of inorganic particles that can be used is not particularly limited, any inorganic particles known in the art can be used, for example, the type of the inorganic particles can be used.
- the composition according to any of the above and below embodiments of the present invention may further comprise an alkoxysilyl group reaction catalyst (hereinafter referred to as “banung catalyst”) as needed.
- the composition containing the alkoxysilyl group reaction catalyst is called “reaction catalyst containing composition” for convenience.
- the reaction catalyst-containing composition also includes at least one new novolac curing agent selected from the group consisting of Formulas 1-1 to 1-4, and the reaction catalyst, and an epoxy resin constituting the composition, the curing agent of the present invention.
- the alkoxysilyl group reaction catalyst may be added for improved processability (eg, fast curing rate and / or low curing temperature).
- the alkoxysilyl group semicoagulant is not limited thereto, but may include, for example, nitric acid, sulfuric acid, hydrochloric acid, acetic acid and phosphoric acid.
- At least one inorganic acid selected from the group, ammonia 4OH, NH 4 0H, amines and transition metal alkoxides, metal oxides, metal organic acid salts and halides e.g.
- dibutyltin Dilaurate (di t butyl in di l aurate), tin salts of octylate, tin (II) ethyl 2- nuclear Sano benzoate) is used at least one kinds selected from the group consisting of "Tin (n) compound), such as Can be. These may be used alone or in combination of two or more kinds.
- the blending amount of the alkoxysilyl group reaction catalyst is not particularly limited, but considering the reaction properties, the alkoxysilyl group semi-amplification catalyst is 0.01 phr to 10 to the epoxy resin of the present invention. can be used as phr.
- water may be further included in the composition including the alkoxysilyl group reaction catalyst.
- the blending amount of water is not particularly limited, but may be 0.01 equivalent to 20 equivalents of water with respect to 1 equivalent of alkoxysilyl group in consideration of efficiency and reaction properties as a catalyst.
- Any curing accelerator (curing catalyst) may be further included as necessary to promote the curing reaction. Also, in the case of the novolak curing agent, to improve the reactivity with the epoxy resin.
- Hardening accelerators are usually used, and in view of the common technical knowledge of the art, it will be apparent to those skilled in the art that any of the above-mentioned and later described compositions may include a curing accelerator depending on the degree of curing reaction.
- a curing accelerator any catalyst known in the art to be generally used for curing the composition can be used, and thus although, for example, a curing accelerator such as imidazole, tertiary amine, quaternary ammonium, organic acid salt, Lewis acid, phosphorus compound, etc. may be used.
- curing accelerators may be used as latent in their microcapsule coating and complex salt formation. These may be used independently and may use 2 or more types together according to hardening conditions.
- the compounding quantity of the said hardening accelerator is not specifically limited, It can mix
- the epoxy resin may be 0.1 to 10 phr (parts per hundred resin, parts by weight per 100 parts by weight of epoxy resin), for example, 0.2 to 5 phr.
- the curing accelerator is preferably used in the above content in terms of curing reaction promoting effect and curing reaction rate control.
- the curing accelerator in the compounding amount in the above range By using it, the curing progresses rapidly and the throughput can be expected to be improved.
- the composition is a release agent, surface treatment agent, flame retardant which is conventionally formulated for controlling the physical properties of the composition in a range that does not impair the physical properties of the composition.
- Other additives such as antifoams, colorants, stabilizers, coupling agents, viscosity modifiers, diluents, rubbers, thermoplastics, etc. may also be blended as needed.
- a composition according to the present invention is formed into a thin film, a composition having insufficient flexibility may cause bridging to cause cracking. This is the case, for example, when any composition of the present invention contains a large amount of inorganic particles.
- rubber and / or thermoplastic resins may be added to compositions comprising any new novolac curing agent of the present invention to improve solubility in the composition.
- Thermoplastic resins and rubber-modified epoxy resins can be used that are generally known in the art.
- any rubber known in the art may be used as long as it is not dissolved in the solvent used in the composition and remains dispersed in the composition.
- an acrylonitrile butadiene rubber for example. Butadiene rubber, acrylic rubber, core-shell rubber particles, cross-linked acrylonitrile butadiene rubber particles, cross-linked styrene butadiene rubber particles, acrylic rubber indenters and the like.
- the average particle diameter is preferably in the range of 005 to 1 / im in terms of physical properties, and 0.2 to The range of 0.6 is more preferable. Rubber particles, see the above .
- Thermoplastic resins include, but are not limited to, phenoxy resins, polyvinyl acetal resins. Polyimide resin, polyamideimide resin, polyether sulone resin, polysulfone resin and the like. These may be used alone or in combination of two or more.
- the thermoplastic resin may be blended in an amount of, for example, 0.5 to 60% by weight, preferably 3 to 50% by weight, based on the weight of the solid content of the composition of the present invention.
- composition comprising a composition, a composition of the present invention or a new novolak curing agent” refers to an epoxy resin and the novolak curing agent of the present invention as well as other components constituting the composition as needed, for example,
- optional curing accelerators catalogsts
- inorganic materials fillers
- solvents may contain other additives formulated as needed in the art.
- the solvent in the composition may optionally be used to appropriately adjust the solid content and / or viscosity of the composition in consideration of the processability of the composition and the like.
- total weight of the solid content of the composition refers to the total weight of the components excluding the solvent among the components constituting the composition described above. Any of the compositions of the present invention provided in any of the above embodiments of the present invention may be used for electronic materials.
- the electronic material is not limited to this, but, for example, not only a substrate build-up film for semiconductors, a prepreg, or a laminated plate on which a metal film is disposed on a substrate made of the composition of the present invention, but also a sealing material (packaging material), as well as printed wiring Electronic components such as substrates. It can also be applied to various applications such as adhesives, paints and composite materials. According to still another embodiment of the present invention, there is provided an electronic material comprising or consisting of any composition comprising the novolac curing agent of the present invention. Furthermore, there is also provided a semiconductor device comprising, consisting essentially of, or consisting of the electronic material.
- the semiconductor device includes a semiconductor device and / or a semiconductor packaging material including (eg, mounting a semiconductor device) a printed wiring board comprising, or consisting essentially of, a composition comprising the novolac curing agent of the present invention. It may be a semiconductor device including a. Also provided are cured products, adhesives, paints, or composites comprising, consisting essentially of, or consisting of any of the compositions of the present invention provided in any of the embodiments of the present invention. According to another embodiment of the present invention, there is provided a cured product comprising, consisting essentially of, or consisting of the composition of the invention provided in any of the embodiments of the invention described above.
- composition provided in any of the above-described embodiments of the present invention is used as a cured product when it is actually applied, for example, as an electronic material, and in the art, an epoxy resin and an inorganic component
- the cured product of the composition comprising the filler is generally referred to as a composite.
- the novel novolac curing agent provided in one embodiment of the present invention described above exhibits excellent heat resistance and excellent flame retardancy in the composite.
- the composite has a low CTE, for example, 15 ppm / ° C. or less, for example 12 ppm / ° C. or less.
- a CTE of 10 ppm / ° C or less for example, 8 ppm / ° C or less, for example, 6 ppm / ° C or less, for example, 4 ppmni / ° C or less.
- the resin content of 30wt% to 60wt% (may have include an inorganic particles-resin content, that may not be included) in the composite are, for example, 10ppm / ° C or less, for example, 8ppm / ° C or less, e. For example, 6 ppm / ° C. or less, for example, 4 ppm / ° C. or less CTE.
- any novel novolac curing agent according to the present invention as a curing agent a composite containing 60 to 80wt%, for example, 70 to 80wt% of inorganic particles, for example, silica particles as an inorganic material, 20ppmrc or less, For example, 15 ppm / ° C or less, for example, 10 ppm / ° C or less, for example, 8 ppm / ° C or less, for example, 6 ppm / ° C
- CTE of 4 ppm / ° C or less is shown.
- the composite according to the present invention may have a T g higher than locrc, for example, 130 ° C or more, for example, 250 ° C or more, or Tg-less.
- the Tg value is excellent in Kk-Tock physical properties and does not particularly limit the upper limit of Tg.
- the value indicated by the range means that each includes not only the lower limit value and the upper limit value of the range, but also any lower range between the ranges and all the numbers belonging to the range.
- C1 to C10 are CI, C2, C3, C4. It is understood to include all of C5, C6, C7, C8, C9, C10.
- the lower limit value or the upper limit value of the numerical range is not specified, and the smaller or larger the value is, the more preferable. It is understood to include any value.
- a CTE below 4 ppm / ° C is understood to include all values between 4, 3.5, 3, 2.7, 2, 1.4, 1, 0.5 ppm / ° C, and the like.
- the NMR data of the obtained final product is as follows.
- Synthesis Example 5 Synthesis of Silylated Noble Curing Agent (Biphenyl Aralkyl Type Phenolic Resin) 15 g of biphenyl novolac curing agent (Formula 3, Meiwa Plastic Ind., Trade name MEH-7851SS), 4.57 g of trioxysilyl propyl isocyanate, and 150 nil of tetrahydrofuran (THF) were added to the flask, followed by mixing. Stirred at room temperature for 15 hours. After the reaction.
- Biphenyl Aralkyl Type Phenolic Resin Silylated Noble Curing Agent
- biphenyl novolac curing agent Forma 3, Meiwa Plastic Ind., Trade name MEH-7851SS
- THF tetrahydrofuran
- the NMR data of the obtained final product is as follows.
- Synthesis Example 7 Synthesized silylated novolac hardener synthesis (polyfunctional phenolic resin) 15 g of a polyfunctional novolac curing agent (Formula 5, Meiwa Plastic Ind., MEH-7500-3S), 7.22 g of triethoxysilyl propyl isocyanate, and 150 ml of tetrahydrofuran (THF) were mixed, and then filled with argon. , and the mixture was stirred at 15 for phase time status.
- a polyfunctional novolac curing agent Forma 5, Meiwa Plastic Ind., MEH-7500-3S
- THF tetrahydrofuran
- epoxy glass fiber composite (cured product) With the composition shown in Table 1, epoxy compound, and silica slurry (solid content of 70wt%, methyl ethyl ketone solvent, average silica size 1 / ⁇ ) of ⁇ , ⁇ -dimethyl Dissolve in acetamide to 40% solids. After mixing this mixed liquid at the speed of 1500 rpm for 1 hour, the hardening
- An epoxy glass fiber composite containing a silylated curing agent was prepared by immersing glass fiber (N-Ttobo, T-glass) in this binder solution. The composite was then placed in a vacuum oven heated to 100 ° C to remove the solvent and then preheated to 12 CTC. The glass fiber composite was obtained by curing at a hot press for 2 hours at 120 ° C., 2 hours at 180 ° C. and 2 hours at a temperature of 200 ° C.-230 ° C.
- Epoxy Filler Composite (Hardened) According to the composition shown in Table 2 below, epoxy compound, silica slurry (dispersion solution: methyl ethyl ketone solvent, silica average size 1) and polyvinyl acetal ⁇ , ⁇ ⁇ dimethylacet Dissolve the amide to 40% solids. The mixture was mixed for 1 hour and 30 minutes at a speed of 1500 rpm, followed by addition of a hardening agent and further mixing for 30 minutes.
- thermo-mechanical analyzer Therm mechanical analysizer
- Table 1 shows the results obtained in Example 1 and Comparative Example 1 (Table 1).
- the specimen of the glass fiber composite film is 4X 16X0.
- the specimen of the composite filler was prepared by the size of 5X5X3 ( ⁇ 3).
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016545968A JP6282352B2 (en) | 2014-01-09 | 2015-01-09 | Novel novolak curing agent having alkoxysilyl group, method for producing the same, composition containing the same, cured product, and use thereof |
| EP15735456.4A EP3093304B1 (en) | 2014-01-09 | 2015-01-09 | Novel novolac curing agent with alkoxysilyl group, method for preparing the same, composition containing the same, the cured product, and the use thereof |
| US15/110,677 US10519274B2 (en) | 2014-01-09 | 2015-01-09 | Novolac curing agent with alkoxysilyl group, method for preparing the same, composition containing the same, the cured product, and use thereof |
| CN201580012579.6A CN106103532B (en) | 2014-01-09 | 2015-01-09 | Novolac hardeners and preparation method thereof containing alkoxysilyl, the composition containing curing agent, hardening product and its application |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2014-0003070 | 2014-01-09 | ||
| KR20140003070 | 2014-01-09 | ||
| KR10-2015-0002675 | 2015-01-08 | ||
| KR1020150002675A KR101664948B1 (en) | 2014-01-09 | 2015-01-08 | New novolac curing agent having alkoxysilyl group, preparing method thereof, composition comprising thereof, cured product thereof, and uses thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015105379A1 true WO2015105379A1 (en) | 2015-07-16 |
Family
ID=53524144
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2015/000243 WO2015105379A1 (en) | 2014-01-09 | 2015-01-09 | Novel novolac hardener having alkoxysilyl group, method for preparing same, composition containing same, hardened product, and use thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2015105379A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2020522532A (en) * | 2017-06-05 | 2020-07-30 | コリア インスティチュート オブ インダストリアル テクノロジー | Compound having alkoxysilyl group and active ester group, method for producing the same, composition containing the same and use |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5177157A (en) * | 1989-08-31 | 1993-01-05 | Dow Corning Toray Silicone Co., Ltd. | Process for preparing a silicone resin-modified phenolic resin |
| KR20010071125A (en) * | 1999-01-29 | 2001-07-28 | 이시베 슈헤이 | Hardener for epoxy resin, epoxy resin composition, and process for producing silane-modified phenolic resin |
| US6875807B2 (en) * | 2003-05-28 | 2005-04-05 | Indspec Chemical Corporation | Silane-modified phenolic resins and applications thereof |
| KR20070114164A (en) * | 2005-02-18 | 2007-11-29 | 히다치 가세고교 가부시끼가이샤 | Novel curable resin, method for producing same, epoxy resin composition and electronic component device |
| US7365135B2 (en) * | 2004-06-14 | 2008-04-29 | Chung-Shan Institute Of Science & Technology | Method for preparing a novolac phenolic resin/silica hybrid organic-inorganic nanocomposite |
-
2015
- 2015-01-09 WO PCT/KR2015/000243 patent/WO2015105379A1/en active Application Filing
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5177157A (en) * | 1989-08-31 | 1993-01-05 | Dow Corning Toray Silicone Co., Ltd. | Process for preparing a silicone resin-modified phenolic resin |
| KR20010071125A (en) * | 1999-01-29 | 2001-07-28 | 이시베 슈헤이 | Hardener for epoxy resin, epoxy resin composition, and process for producing silane-modified phenolic resin |
| US6875807B2 (en) * | 2003-05-28 | 2005-04-05 | Indspec Chemical Corporation | Silane-modified phenolic resins and applications thereof |
| US7365135B2 (en) * | 2004-06-14 | 2008-04-29 | Chung-Shan Institute Of Science & Technology | Method for preparing a novolac phenolic resin/silica hybrid organic-inorganic nanocomposite |
| KR20070114164A (en) * | 2005-02-18 | 2007-11-29 | 히다치 가세고교 가부시끼가이샤 | Novel curable resin, method for producing same, epoxy resin composition and electronic component device |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2020522532A (en) * | 2017-06-05 | 2020-07-30 | コリア インスティチュート オブ インダストリアル テクノロジー | Compound having alkoxysilyl group and active ester group, method for producing the same, composition containing the same and use |
| US11214583B2 (en) | 2017-06-05 | 2022-01-04 | Korea Institute Of Industrial Technology | Compound having alkoxysilyl group and active ester group, method for preparing same, composition comprising same, and use |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6282352B2 (en) | Novel novolak curing agent having alkoxysilyl group, method for producing the same, composition containing the same, cured product, and use thereof | |
| CN105073760B (en) | Epoxide with alkoxysilyl groups, preparation method include its composition and cured product, and application thereof | |
| KR101898526B1 (en) | Epoxy compound having alkoxysilyl group, composition, cured product thereof, use thereof and preparing method of epoxy compound having alkoxysilyl group | |
| KR101863111B1 (en) | Novolac-based epoxy compound, preparing method thereof, composition, cured product thereof, and use thereof | |
| JP6423443B2 (en) | Novel epoxy compound, mixture containing the same, composition, cured product, method for producing the same, and use thereof | |
| JP2012524828A (en) | New epoxy resin and epoxy resin composition containing the same | |
| JP2018500309A (en) | Thermosetting alkoxysilyl compound having two or more alkoxysilyl groups, composition containing the same, cured product, use thereof, and method for producing alkoxysilyl compound | |
| JP6852039B2 (en) | A method for producing an epoxy compound having an alkoxysilyl group, an epoxy compound having an alkoxysilyl group, a composition containing the same, and its use. | |
| KR101749551B1 (en) | New epoxy compound having alkoxysilyl group, preparing method thereof, composition, cured product thereof, and uses thereof | |
| WO2013066078A1 (en) | Isocyanurate epoxy compound having alkoxysilyl group, method of preparing same, composition including same, cured product of the composition, and use of the composition | |
| WO2015105379A1 (en) | Novel novolac hardener having alkoxysilyl group, method for preparing same, composition containing same, hardened product, and use thereof | |
| KR101157566B1 (en) | Epoxy Resin Having Side Functional Group and Thermosetting Polymer Composite Comprising the Same | |
| WO2015126143A1 (en) | Novel epoxy compound, mixture, composition, and cured product comprising same, method for preparing same, and use thereof | |
| JP2001114865A (en) | Epoxy resin composition and epoxy resin composition for semiconductor sealing | |
| WO2015133840A2 (en) | Epoxy compound having alkoxysilyl group, method for preparing same, composition comprising same, cured product, and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15735456 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016545968 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15110677 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015735456 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015735456 Country of ref document: EP |