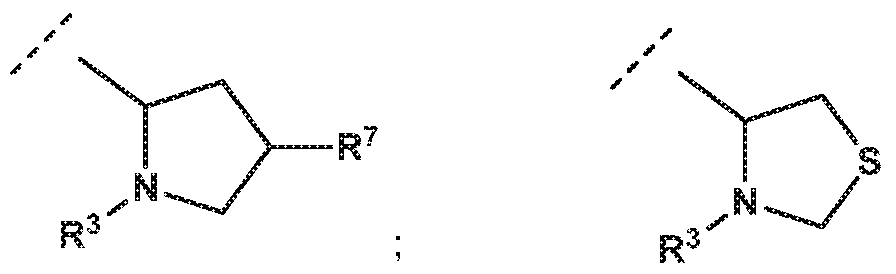WO2014128612A1 - Quinazolin-4-one derivatives - Google Patents
Quinazolin-4-one derivatives Download PDFInfo
- Publication number
- WO2014128612A1 WO2014128612A1 PCT/IB2014/059071 IB2014059071W WO2014128612A1 WO 2014128612 A1 WO2014128612 A1 WO 2014128612A1 IB 2014059071 W IB2014059071 W IB 2014059071W WO 2014128612 A1 WO2014128612 A1 WO 2014128612A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino
- chloro
- oxo
- carbonitrile
- dihydroquinazolin
- Prior art date
Links
- 0 CC1N(*)CSC1 Chemical compound CC1N(*)CSC1 0.000 description 31
- LMQMQSGHBAYOAI-UHFFFAOYSA-N CC(C1C#N)=NC=NC1N Chemical compound CC(C1C#N)=NC=NC1N LMQMQSGHBAYOAI-UHFFFAOYSA-N 0.000 description 1
- SNBKIYGGKKMWGW-UHFFFAOYSA-N CC1N(C)CSC1 Chemical compound CC1N(C)CSC1 SNBKIYGGKKMWGW-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D407/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00
- C07D407/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/02—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6
- C07D473/16—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 two nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/26—Heterocyclic compounds containing purine ring systems with an oxygen, sulphur, or nitrogen atom directly attached in position 2 or 6, but not in both
- C07D473/32—Nitrogen atom
- C07D473/34—Nitrogen atom attached in position 6, e.g. adenine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/04—Ortho-condensed systems
Definitions
- the present invention relates to the preparation and use of new quinazolin-4-one derivatives as drug candidates in free form or in pharmaceutically acceptable salt form with valuable druglike properties, such as e.g. metabolic stability and suitable pharmacokinetics, form for the modulation, notably the inhibition of the activity or function of the phosphoinositide 3' OH kinase family (hereinafter PI3K).
- valuable druglike properties such as e.g. metabolic stability and suitable pharmacokinetics, form for the modulation, notably the inhibition of the activity or function of the phosphoinositide 3' OH kinase family (hereinafter PI3K).
- PI3K phosphoinositide-3 kinase
- PI3Ks have been identified, divided into three main classes (I, II and III) on the basis of their genetic sequence, structure, adapter molecules, expression, mode of activation, and prefered substrate.
- class I family comprising isoforms PI3K ⁇ , ⁇ , ⁇ and ⁇
- IA and IB are further subdivided into subclasses IA and IB.
- Class IA PI3 kinases consist of an 85 kDa regulatory/adapter protein and three 1 10 kDa catalytic subunits (p1 10a, p1 10 ⁇ and p1 105) which are activated in the tyrosine kinase system whilst class IB consists of a single p1 10 ⁇ isoform ( ⁇ 3 ⁇ ) which is activated by G protein-coupled receptors.
- PI3K5 and ⁇ 3 ⁇ are both lipid kinases belonging to the class I PI3K family (PI3K ⁇ , ⁇ , ⁇ and ⁇ ).
- PI3K5 generates second messenger signals downstream of tyrosine kinase- linked receptors while ⁇ 3 ⁇ is primarily activated by G protein-coupled receptors (GPCR).
- GPCR G protein-coupled receptors
- PI3K5 and ⁇ 3 ⁇ are heterodimers composed of an adaptor protein and a p1 105 or p1 10 ⁇ catalytic subunit, respectively, which converts phosphatidylinositol-4,5-bis- phosphate (PtdlnsP2) to phosphatidylinositol-3,4,5-tri-phosphate (PtdlnsP3). Effector proteins interact with PtdlnsP3 and trigger specific signaling pathways involved in cell activation, differentiation, migration, and cell survival. Expression of the p1 105 and p1 10 ⁇ catalytic subunits is preferential to leukocytes.
- p1 10a and p1 10 ⁇ are expressed by all cell types (Marone et al. Biochimica et Biophysica Acta 1784:159 (2008)).
- PI3K5 is associated with B cell development and function (Okkenhaug et al. Science 297:1031 (2002)).
- B cells play also a critical role in the pathogenesis of a number of autoimmune and allergic diseases as well as in the process of transplant rejection (Martin and Chan, Annu. Rev. Immunol. 24:467 (2006)).
- Chemotaxis is involved in many autoimmune or inflammatory diseases, in angiogenesis, invasion/metastasis, neurodegeneration or woud healing (Gerard et al. Nat. Immunol.
- chemokines are fully dependent on PI3K5 and ⁇ 3 ⁇ (Liu et al. Blood 1 10:1 191 (2007)).
- PI3Ka and ⁇ 3 ⁇ play an essential role in maintaining homeostasis and pharmacological inhibition of these molecular targets has been associated with cancer therapy (Maira et al. Expert Opin. Ther. Targets 12:223 (2008)).
- PI3Ka is involved in insulin signaling and cellular growth pathways (Foukas et al. Nature 441 :366 (2006)). PI3K5 and/or ⁇ 3 ⁇ isoform-selective inhibition is expected to avoid potential side effects such as hyperglycemia, and metabolic or growth disregulation.
- Parasitic infections still represent one of the most important causes of morbidity and mortality worldwide.
- the phylum apicomplexa comprises a group of vector-borne parasites that is responsible for a wide variety of serious illnesses including but not limited to malaria, leishmaniasis and trypanosomiasis. Malaria alone infects 5-10% of civilization and causes around two milion deaths per year.
- TLRs Toll-like receptors
- TLR1 functional TLR family members
- the immune system of the infected host responds to infection with the TLR induced production of pro-inflammatory cytokines mainly of the T-helper 1 type (Th1). While adequate amounts of these cytokines are remedial and required to clear the infection an overproduction of these mediators is harmful to the host and associated with immune mediated pathology including neuropathology and tissue damage with severe and often fatal consequences.
- immune mediated pathology includes neuropathology and tissue damage with severe and often fatal consequences.
- CM acute and cerebral malaria
- Malaria is an infectious disease caused by four protozoan parasites: Plasmodium falciparum; Plasmodium vivax; Plasmodium ovale; and Plasmodium malaria. These four parasites are typically transmitted by the bite of an infected female Anopheles mosquito. Malaria is a problem in many parts of the world and over the last few decades the malaria burden has steadily increased. An estimated 1 -3 million people die every year from malaria - mostly children under the age of 5. This increase in malaria mortality is due in part to the fact that Plasmodium falciparum, the deadliest malaria parasite, has acquired resistance against nearly all available antimalarial drugs, with the exception of the artemisinin derivatives.
- Leishmaniasis is caused by one or more than 20 varieties of parasitic protozoa that belong to the genus Leishmania, and is transmitted by the bite of female sand flies. Leishmaniasis is endemic in about 88 countries, including many tropical and sub-tropical areas. There are four main forms of Leishmaniasis. Visceral leishmaniasis, also called kala-azar, is the most serious form and is caused by the parasite Leishmania donovani. Patients who develop visceral leishmaniasis can die within months unless they receive treatment. The two main therapies for visceral leishmaniasis are the antimony derivatives sodium stibogluconate (Pentostam®) and meglumine antimoniate (Glucantim®). Sodium stibogluconate has been used for about 70 years and resistance to this drug is a growing problem. In addition, the treatment is relatively long and painful, and can cause undesirable side effects.
- Pentostam® sodium stibogluconate
- Glucantim® me
- Human African Trypanosomiasis also known as sleeping sickness, is a vector-borne parasitic disease.
- the parasites concerned are protozoa belonging to the Trypanosoma Genus. They are transmitted to humans by tsetse fly (Glossina Genus) bites which have acquired their infection from human beings or from animals harboring the human pathogenic parasites.
- Chagas disease also called American Trypanosomiasis
- the disease is caused by the protozoan parasite Trypanosoma cruzi, which is transmitted to humans by bloodsucking insects.
- the human disease occurs in two stages: the acute stage, which occurs shortly after infection and the chronic stage, which can develop over many years.
- Chronic infections result in various neurological disorders, including dementia, damage to the heart muscle and sometimes dilation of the digestive tract, as well as weight loss. Untreated, the chronic disease is often fatal.
- the drugs currently available for treating Chagas disease are Nifurtimox and benznidazole.
- problems with these current therapies include their diverse side effects, the length of treatment, and the requirement for medical supervision during treatment.
- treatment is really only effective when given during the acute stage of the disease. Resistance to the two frontline drugs has already occurred.
- the antifungal agent Amphotericin b has been proposed as a second-line drug, but this drug is costly and relatively toxic.
- Toxoplasmosis is endemic in many areas globally and can infect a large proportion of the adult population. However, its prevalence differs in different countries. It is estimated to infect at least 10% of adults in northern temperate countries and more than half of
- Toxoplasma gondii the causative pathogen of toxoplamosis
- Toxoplasma gondii the causative pathogen of toxoplamosis
- the course of disease in immunocompetent adults is usually asymptomatic
- the parasite forms latent cysts in the retina and in other organs of the body, which can reactivate years after the initial infection giving rise to acute retinochoroiditis and the formation of new retinochoroidal lesions.
- Neurocysticercosis is the most common parasitic disease of the CNS (incidence ⁇ 2.5 milion worldwide) caused by the larvae of Taenia solium.
- the disease has a long asymptomatic phase in humans characterized by the absence of a detectable inflammatory response surrounding the parasite.
- the overall immune response during the asymptomatic phase is of the Th2 phenotype.
- the destruction of larvae by therapeutic treatment or by normal parasite attrition causes a strong inflammatory response, often consisting of a chronic granulomatous reaction and manifestation of typical symptoms of the disease.
- the immune response in the CNS of symptomatic patients consists of an overt Th1 phenotype or a mixed Th1 , Th2, and Th3 response, depending upon the absence or presence of granulomas.
- the hyperinflammatory response prevailing during the symptomatic phase in the CNS is responsible for the severe neuropathology and mortality associated with neurocysticercosis . [Mishra et al, "TLRs in CNS Parasitic infections", Curr Top Micro Imm 2009]
- compounds of the invention should bind potently to class I PI3 kinases whilst showing little affinity for other receptors and show functional activity as inhibitors. They should be well absorbed from the gastrointestinal tract, be metabolically stable and possess favourable pharmacokinetic properties. When targeted against receptors in the central nervous system they should cross the blood brain barrier freely and when targeted selectively against receptors in the peripheral nervous system they should not cross the blood brain barrier. They should be non-toxic and demonstrate few side-effects. Furthermore, the ideal drug candidate will exist in a physical form that is stable, non-hygroscopic and easily formulated. The compounds of the invention show a certain level of selectivity against the different paralogs PI3K ⁇ , ⁇ , ⁇ and ⁇ . In particular, show a certain level of selectivity for the isoforms PI3K5, ⁇ 3 ⁇ and ⁇ 3 ⁇ over the PI3Ka isoform.
- the compounds of the present invention are therefore potentially useful in the treatment of a wide range of disorders, particularly disorders including but not limited to
- autoimmune disorders autoinflammatory and inflammatory diseases, allergic diseases, disease or infection associated immunopathologies, airway diseases, such as asthma and COPD, transplant rejection, cancers eg of hematopoietic origin or solid tumors.
- airway diseases such as asthma and COPD
- transplant rejection cancers eg of hematopoietic origin or solid tumors.
- the invention provides a pharmaceutical composition
- a pharmaceutical composition comprising a therapeutically effective amount of a compound according to the definition of formula (I), or a pharmaceutically acceptable salt thereof, or subformulae thereof and one or more pharmaceutically acceptable carriers.
- the invention provides a combination, in particular a pharmaceutical combination, comprising a therapeutically effective amount of the compound according to the definition of formula (I), or a pharmaceutically acceptable salt thereof, or subformulae thereof and one or more therapeutically active agent.
- the invention also relates to the treatment, either alone or in combination, with one or more other pharmacologically active compounds, including methods of treating conditions, diseases or disorders in which one or more of the functions of B cells such as antibody production, antigen presentation, cytokine
- lymphoid organogenesis are abnormal or are undesirable including rheumatoid arthritis and related diseases (such as ankylosing spondylarthritis, psoriatic arthritis, juvenile arthritis), pemphigus vulgaris and related diseases, idiopathic
- thrombocytopenia purpura systemic lupus erythematosus, multiple sclerosis
- thrombocytopenic purpura chronic autoimmune urticaria
- allergy atopic dermatitis, contact dermatitis, allergic rhinitis
- goodpasture's syndrome different types of
- glomerulonephritides AMR (antibody-mediated transplant rejection), B cell-mediated hyperacute, acute and chronic transplant rejection and cancers of haematopoietic origin including but not limited to multiple myeloma; acute myelogenous leukemia; chronic myelogenous leukemia; lymphocytic leukemia; myeloid leukemia; non-Hodgkin
- lymphoma lymphomas
- polycythemia vera essential thrombocythemia
- myelofibrosis with myeloid metaplasia Walden stroem disease as well as in disease or infection associated immunopathology.
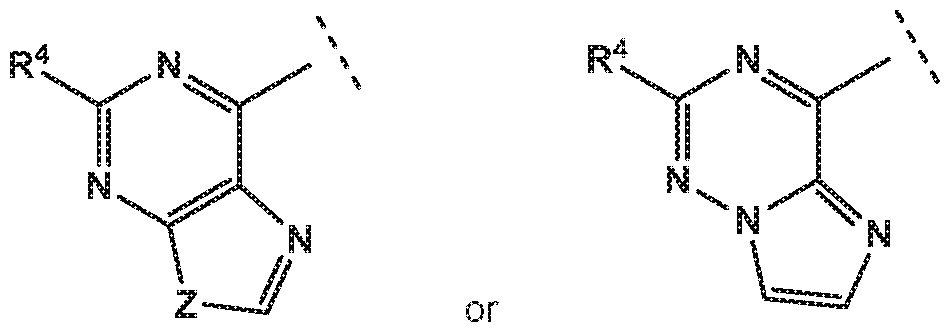
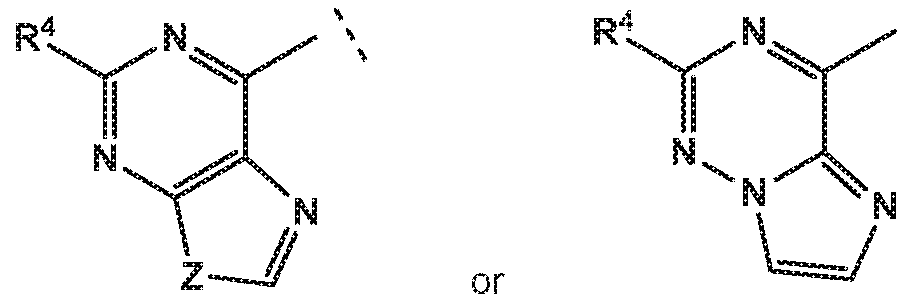
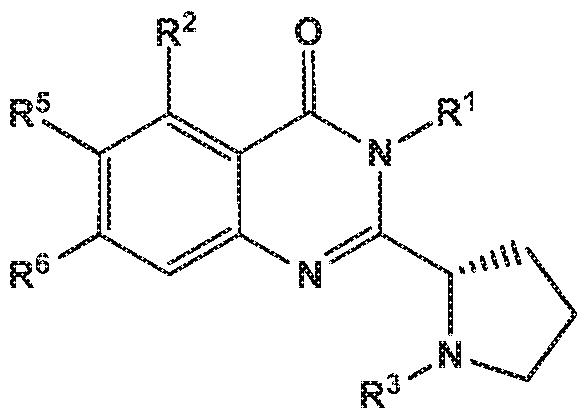
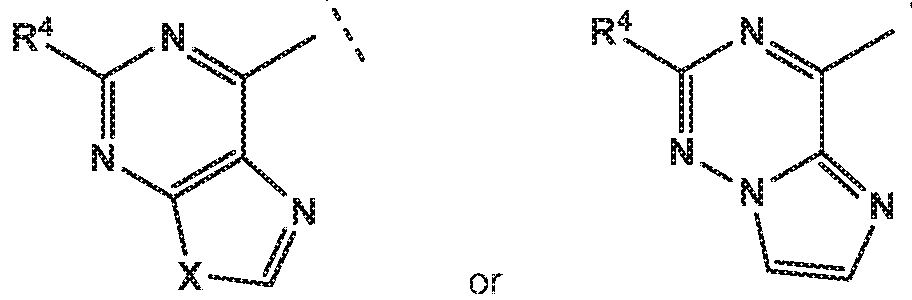
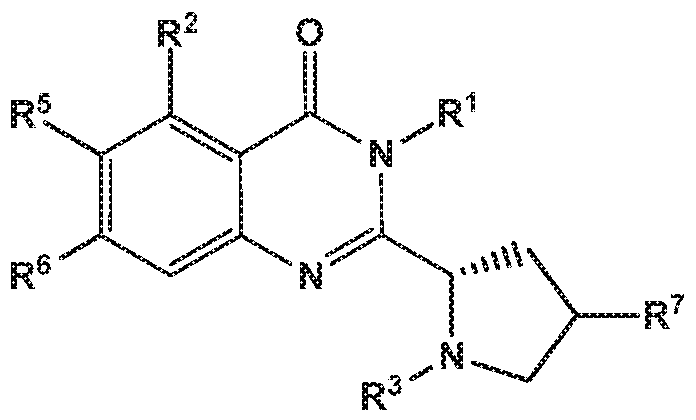
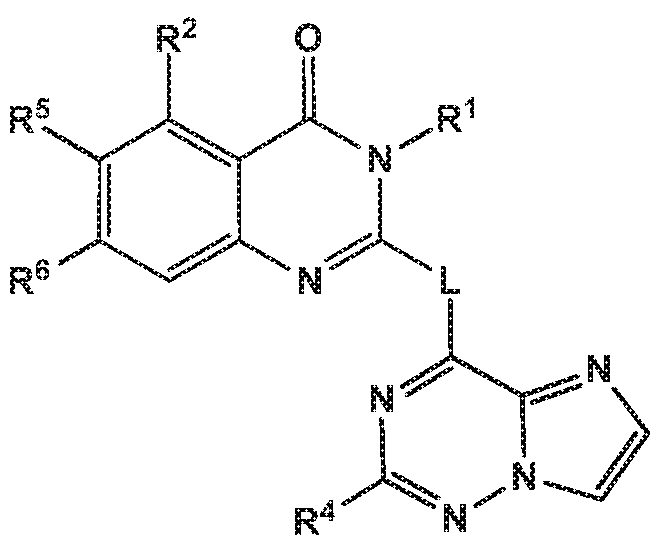
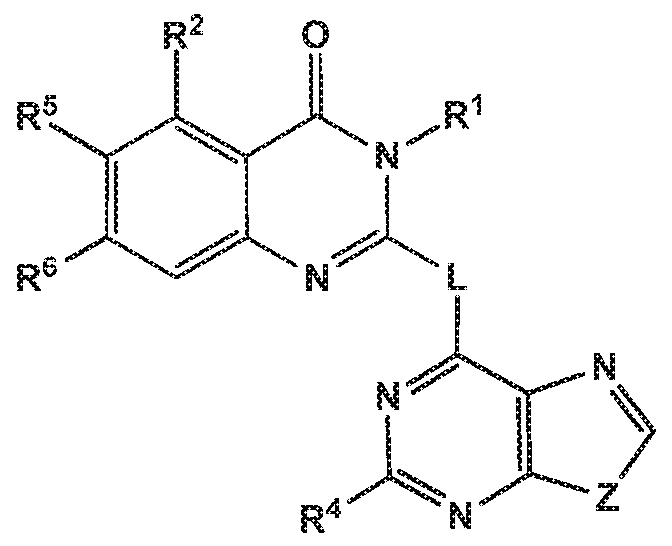
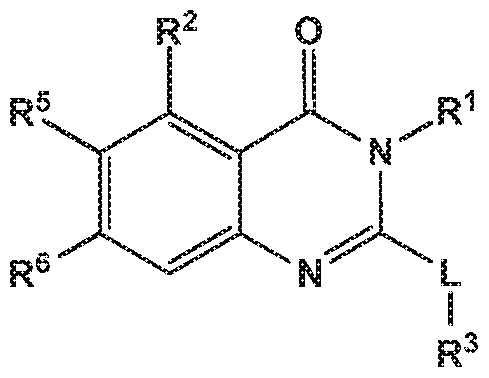
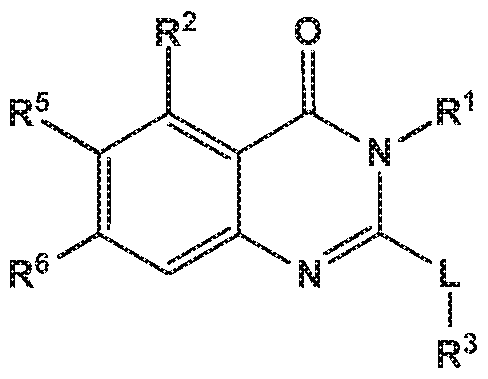
- the invention provides quinazolin-4-one compounds of the formula (I) and/or
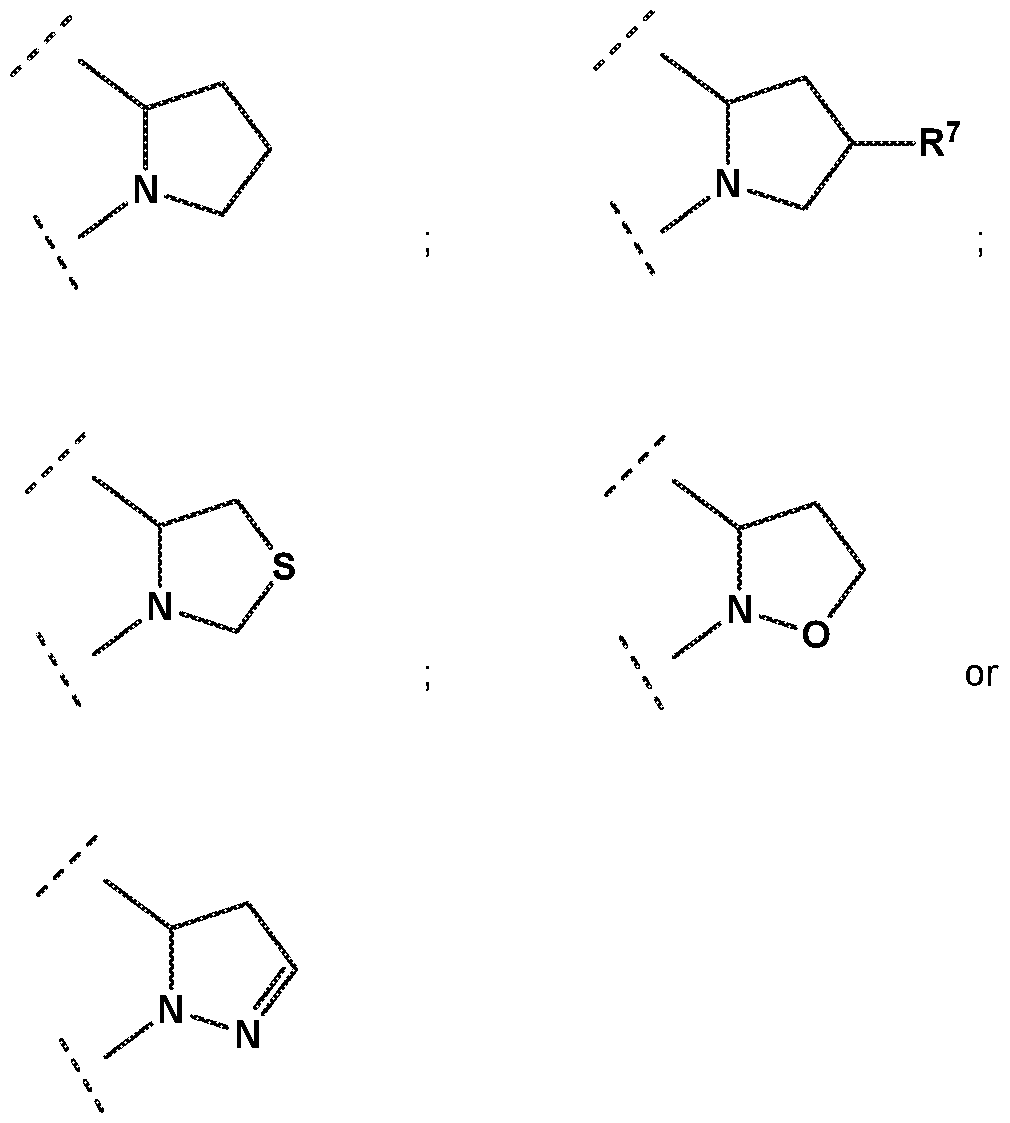
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, trideuteromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
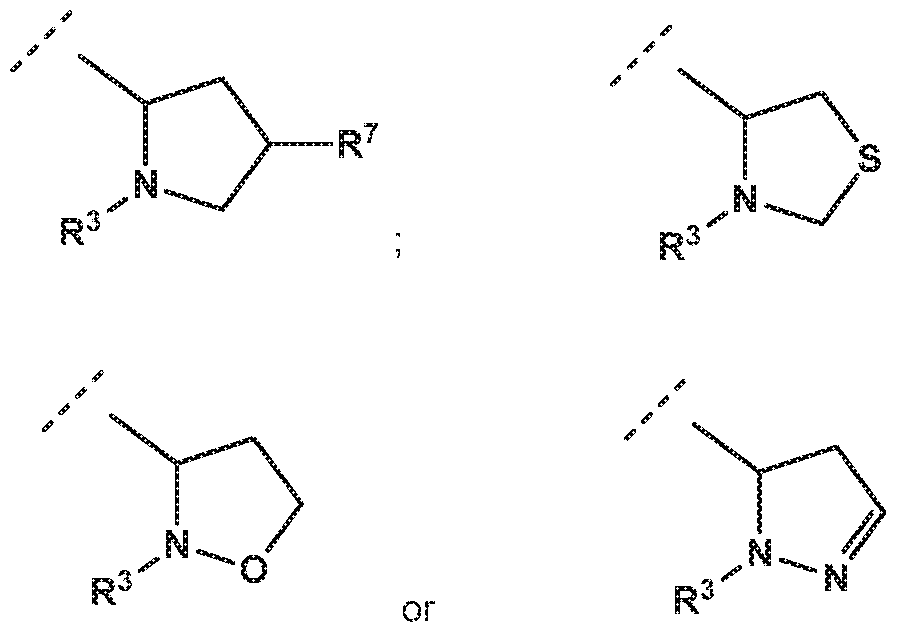
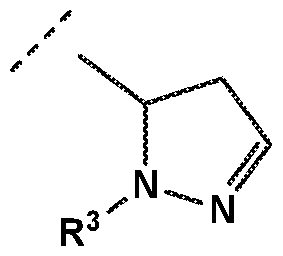
- -L-R 3 is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is independently selected from
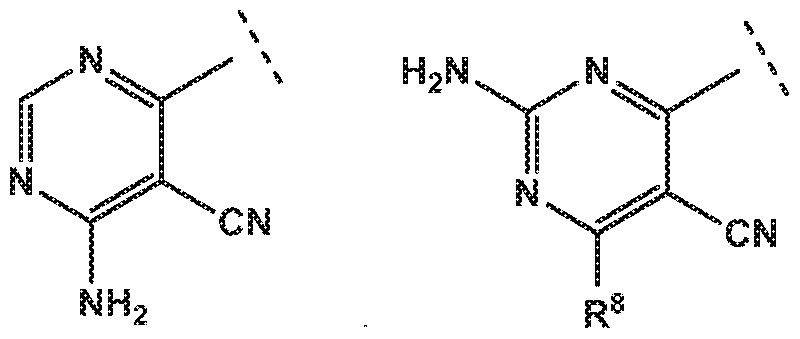
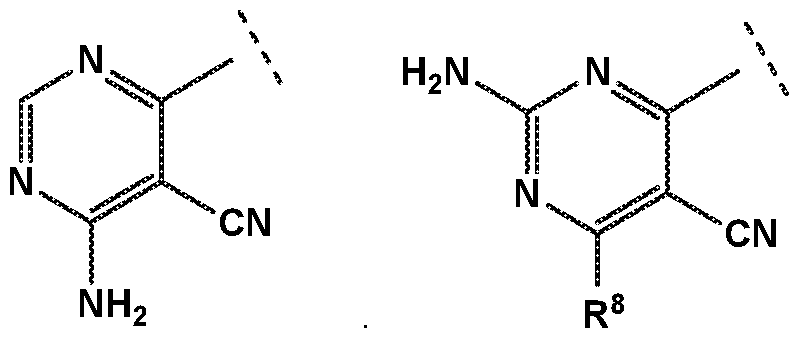
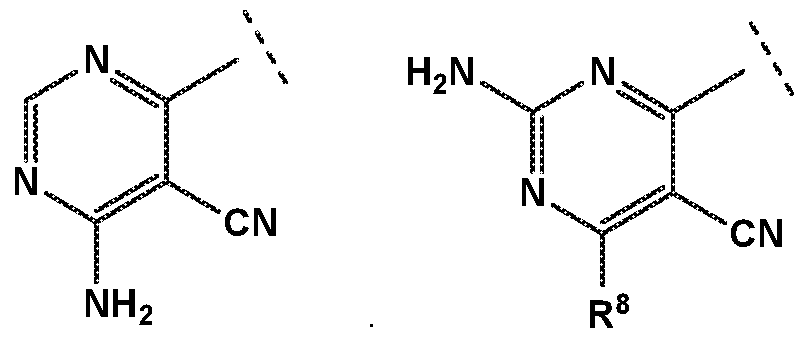
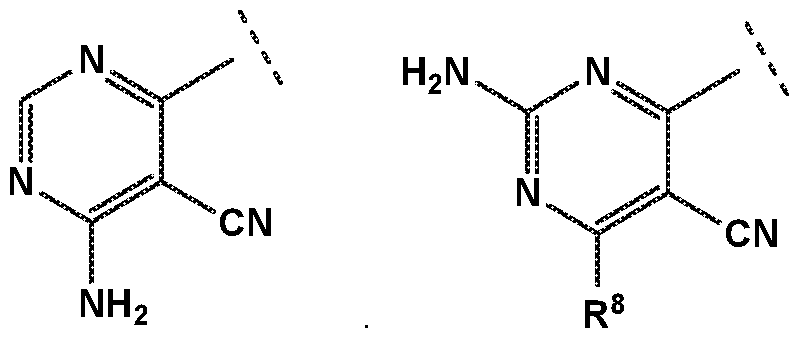
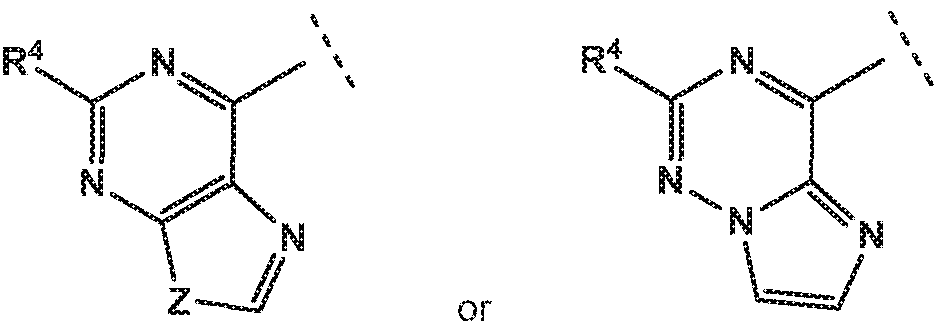
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S;
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro , methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- -L-R 3 is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and Z is independently selected from NMe or S;
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- -L-R 3 is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino.
- the term "compounds of the present invention” refers to compounds of formula (I) and subformulae thereof, salts of the compound, as well as all stereoisomers (including diastereoisomers and enantiomers), rotamers, tautomers and isotopically labeled compounds (including deuterium substitutions, as well as inherently formed moieties). Where compounds of formula (I) are mentioned, this is meant to include also the tautomers and N-oxides of the compounds of formula (I).
- Tautomers such as tautomers between purine forms can be present for example in the R 3 portion of compounds of formula (I).
- Nitrogen containing heterocyclyl and heteroaryl residues may form N-oxides. Where the plural form is used for compounds, salts, and the like, this is taken to mean also a single compound, salt, or the like.
- C 3 -C 6 -cycloalkyl refers to a 3 to 6 membered monocyclic saturated ring.
- examples of C 3 -C 6 -cycloalkyl include cyclopropyl; cyclobutyl;
- examples of C 3 -C 6 -cycloalkyl which is substituted in the 1 position by methyl include 1 -methylcyclopropyl; 1 -methylcyclobutyl; 1 -methylcyclopentyl and 1 - methylcyclohexyl.
- all substituents are written in a way to show the order of functional groups (groups) they are composed of.
- the functional groups are defined herein above.
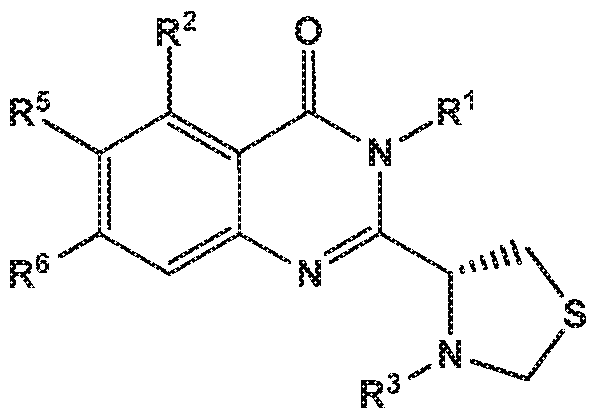
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula ( ⁇ )
- R 1 is selected from
- morpholin-4-yl pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- -L-R 3 is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is independently selected from wherein
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
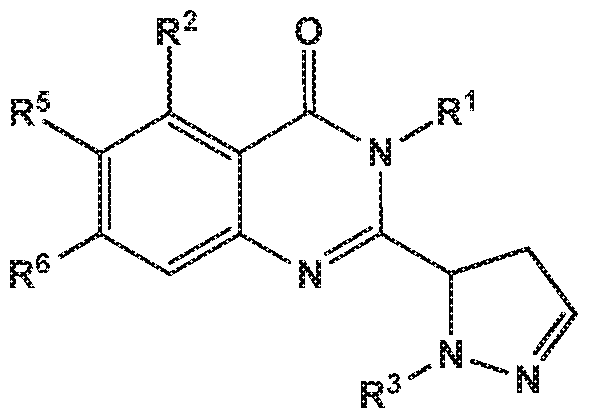
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula ( ⁇ )
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro; -L-R 3 is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy,
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
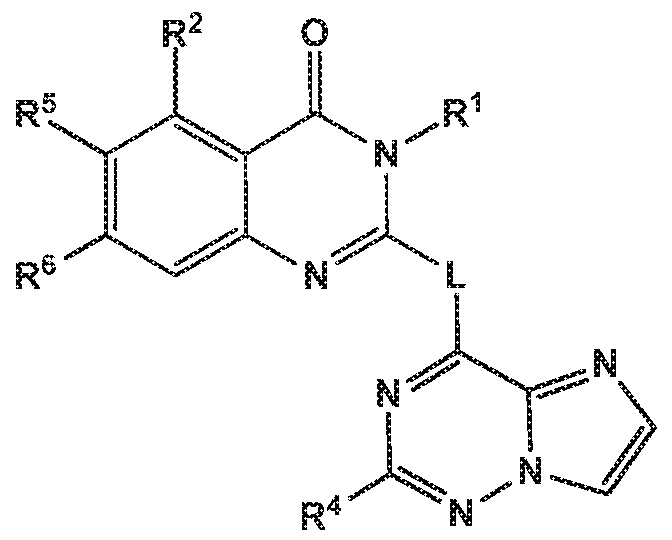
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (I")
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- -L-R 3 is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (I")
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- -L-R 3 is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula ( ⁇ ")
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- -L-R 3 is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula ( ⁇ ")
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- -L-R 3 is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (la')
- R 1 is selected from
- phenyl which is substituted by one substituent in the 2 position which is independently selected from methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (la')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro;
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro;
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine;
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
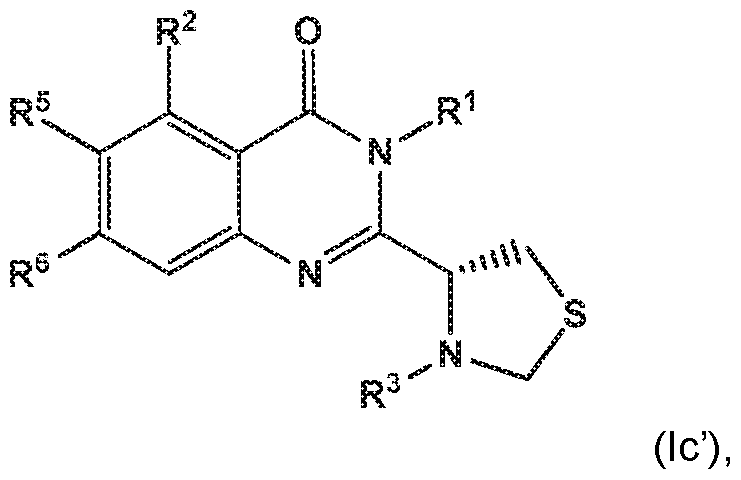
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Ic')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lc')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id')
- R 1 is selected from
- 3-pyridyl which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino
- X is independently selected from NH, NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and X is independently selected from NH, NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le')
- R 1 is selected from
- phenyl which is substituted by one substituent in the 2 position which is independently selected from methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- X is independently selected from NH, NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula ( ⁇ ')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro; -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula ( ⁇ ')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro;
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IB')
- R 1 is selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino,
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IB')
- R 1 is selected from phenyl, which is substituted by one substituent in the 2 position which is independently selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IC)
- R 1 is selected from phenyl, which is substituted by one substituent in the 2 position which is independently selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino
- X is independently selected from NH, NMe or S;
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IC)
- R 1 is selected from phenyl, which is substituted by one substituent in the 2 position which is independently selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino
- X is independently selected from NH, NMe or S; -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ID')
- R 1 is selected from phenyl, which is substituted by one substituent in the 2 position which is independently selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ID')
- R 1 is selected from phenyl, which is substituted by one substituent in the 2 position which is independently selected from
- pyrolidin-3-yl which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (la).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (la).
- R 1 is selected from phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro;
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine;
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lc).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl.chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Ic).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro;
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le).
- R 1 is selected from phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 3 is independently selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IA).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro; -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IA).
- R 1 is selected from phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro; -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IB).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino;
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IB).
- R 1 is selected from phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino;
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IC).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino
- Z is independently selected from NMe or S
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IC).
- R 1 is selected from phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino
- Z is independently selected from NMe or S
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ID).
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ID).
- R 1 is selected from phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino
- -L- is independently selected from
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 3 is attached to L via the trivalent nitrogen atom.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb').
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro;
- R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb').
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine
- R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lc').
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro; and R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Ic')
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id').
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id').
- R is selected from phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le').
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le').
- R 1 is selected from
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
- R 5 and R 6 are independently selected from hydrogen, deuterium or fluoro
- R 4 is independently selected from hydrogen or amino.
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula ( ⁇ )
- -L-R 3 is independently selected from
- R 1 , R 2 , R 3 , R 5 , R 6 and R 7 are as defined above for the compound of formula ( ⁇ ).
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula ( ⁇ )
- R 4 is independently selected from hydrogen or amino
- R 8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
- Z is independently selected from NMe or S; and R , R , R , R and L are as defined above for the compound of formula ( ⁇ ).
- the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (I")
- -L-R 3 is independently selected from
- R 1 , R 2 , R 3 , R 5 , R 6 and R 7 are as defined above for the compound of formula (I").
- the invention provides a compound of the formulae (I) or ( ⁇ ), (la'), (lb'), (IC), (Id'), (le'), ( ⁇ '), ( ⁇ '), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or ( ⁇ "), (lb'"), (Ic'"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
- R 5 is independently selected from hydrogen or fluoro
- R 6 is independently selected from hydrogen.
- the invention provides a compound of the formulae (I) or ( ⁇ ), (la'), (lb'), (IC), (Id'), (le'), ( ⁇ '), ( ⁇ '), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID”) or ( ⁇ "), (lb'"), (IC"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
- R 2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, methylsulfonyl, ethylsulfonyl or cyclopropylsulfonyl.
- the invention provides a compound of the formulae (I) or ( ⁇ ), (la'), (lb'), (IC), (Id'), (le'), ( ⁇ '), ( ⁇ '), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (I'"), (lb'"), (IC"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
- R 2 is independently selected from methyl, trifluoromethyl, difluoromethoxy, fluoro or methylsulfonyl.
- the invention provides a compound of the formulae (I) or ( ⁇ ), (la'), (lb'), (IC), (Id'), (le'), ( ⁇ '), ( ⁇ '), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID”) or ( ⁇ "), (lb'"), (IC"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
- R 5 is independently selected from hydrogen or fluoro
- R 6 is independently selected from hydrogen
- R 2 is independently selected from methyl, trifluoromethyl, difluoromethoxy, fluoro or methylsulfonyl.
- the invention provides a compound of the formulae (I) or ( ⁇ ), (la'), (lb'), (IC), (Id'), (le'), ( ⁇ '), ( ⁇ '), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le”), (IA"), (IB"), (IC"), (ID”) or ( ⁇ "), (lb'"), (IC"), (Id'"), (le'”) and/or a pharmaceutically acceptable salt thereof, wherein
- R 5 is independently selected from fluoro
- R 6 is independently selected from hydrogen
- R 2 is independently selected from methyl or fluoro.
- the invention provides a compound of the formulae (I) or ( ⁇ ), (la'), (lb'), (IC), (Id'), (le'), ( ⁇ '), ( ⁇ '), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID”) or ( ⁇ "), (lb'"), (IC"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
- R 5 is independently selected from hydrogen
- R 6 is independently selected from hydrogen
- R 2 is independently selected from trifluoromethyl, difluoromethoxy or methylsulfonyl.
- the invention provides a compound of the formulae (I) or ( ⁇ ), (la'), (lb'), (Ic'), (Id'), (le'), ( ⁇ '), ( ⁇ '), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID”) or ( ⁇ "), (lb'"), (Ic'"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
- R 5 is independently selected from hydrogen
- R 6 is independently selected from hydrogen
- R 2 is independently selected from trifluoromethyl.
- the invention provides a compound of the formulae ( ⁇ ), (la'), (lb'), (Ic'), (Id') or (le') and/or a pharmaceutically acceptable salt thereof, wherein
- R 3 is selected from
- R 4 is independently selected from hydrogen or amino
- R 8 is methyl
- X is S.
- the invention provides a compound of the formulae (I"), (la"), (lb"), (Ic"), (Id") or (le”) and/or a pharmaceutically acceptable salt thereof, wherein R 3 is selected from wherein
- R 4 is independently selected from hydrogen or amino
- R 8 is methyl
- the invention provides a compound of the formulae ( ⁇ ), (la'), (lb'), (Ic'), (Id') or (le') and/or a pharmaceutically acceptable salt thereof, wherein
- R 3 is selected from
- R 8 is methyl
- the invention provides a compound of the formulae (la") or (lb"), (Ic"), (Id”) or (le”) and/or a pharmaceutically acceptable salt thereof, wherein
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Public Health (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Tropical Medicine & Parasitology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Immunology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention relates to quinazolin-4-one compounds of the formula (I) and/or pharmaceutically acceptable salts and/or solvates thereof, Formula (I) wherein R1, R2, R3, R5, R6 and L are as defined in the description. Such compounds are suitable for the treatment of a disorder or disease which is mediated by the activity of the class I PI3K kinases.
Description
Quinazolin-4-one Derivatives
FIELD OF THE INVENTION
The present invention relates to the preparation and use of new quinazolin-4-one derivatives as drug candidates in free form or in pharmaceutically acceptable salt form with valuable druglike properties, such as e.g. metabolic stability and suitable pharmacokinetics, form for the modulation, notably the inhibition of the activity or function of the phosphoinositide 3' OH kinase family (hereinafter PI3K). BACKGROUND OF THE INVENTION
Members of the phosphoinositide-3 kinase (PI3K) family are involved in cell growth, differentiation, survival, cytoskeletal remodeling and the trafficking of intracellular organelles in many different types of cells (Okkenhaug and Wymann, Nature Rev.
Immunol. 3:317 (2003).
To date, eight mammalian PI3Ks have been identified, divided into three main classes (I, II and III) on the basis of their genetic sequence, structure, adapter molecules, expression, mode of activation, and prefered substrate. The most widely understood class I family (comprising isoforms PI3K α, β, γ and δ) is further subdivided into subclasses IA and IB. Class IA PI3 kinases (isoforms PI3Ka, ΡΙ3Κβ and PI3K5) consist of an 85 kDa regulatory/adapter protein and three 1 10 kDa catalytic subunits (p1 10a, p1 10β and p1 105) which are activated in the tyrosine kinase system whilst class IB consists of a single p1 10γ isoform (ΡΙ3Κγ) which is activated by G protein-coupled receptors.
PI3K5 and ΡΙ3Κγ are both lipid kinases belonging to the class I PI3K family (PI3K α, β, γ and δ). PI3K5 generates second messenger signals downstream of tyrosine kinase- linked receptors while ΡΙ3Κγ is primarily activated by G protein-coupled receptors (GPCR).
PI3K5 and ΡΙ3Κγ are heterodimers composed of an adaptor protein and a p1 105 or p1 10γ catalytic subunit, respectively, which converts phosphatidylinositol-4,5-bis- phosphate (PtdlnsP2) to phosphatidylinositol-3,4,5-tri-phosphate (PtdlnsP3). Effector proteins interact with PtdlnsP3 and trigger specific signaling pathways involved in cell activation, differentiation, migration, and cell survival.
Expression of the p1 105 and p1 10γ catalytic subunits is preferential to leukocytes.
Expression is also observed in smooth muscle cells, myocytes and endothelial cells. In contrast, p1 10a and p1 10β are expressed by all cell types (Marone et al. Biochimica et Biophysica Acta 1784:159 (2008)).
PI3K5 is associated with B cell development and function (Okkenhaug et al. Science 297:1031 (2002)).
B cells play also a critical role in the pathogenesis of a number of autoimmune and allergic diseases as well as in the process of transplant rejection (Martin and Chan, Annu. Rev. Immunol. 24:467 (2006)).
A link between PI3Ky and processes such as leukocyte chemotaxis and mast cell degranulation has been shown, thereby generating interest in this target for the treatment of autoimmune and inflammatory disorders (Ghigo et al., Bioessays, 2010, 32, 185-196; Reif et al., J. Immunol., 2004, 173, 2236-2240; Laffargue et al., Immunity, 2002, 16, 441 -451). There are also reports linking ΡΙ3Κγ ίο cancer, diabetes, cardiovascular disease, and Alzheimer's disease.
Chemotaxis is involved in many autoimmune or inflammatory diseases, in angiogenesis, invasion/metastasis, neurodegeneration or woud healing (Gerard et al. Nat. Immunol.
2:108 (2001)). Temporarily distinct events in leukocyte migration in response to
chemokines are fully dependent on PI3K5 and ΡΙ3Κγ (Liu et al. Blood 1 10:1 191 (2007)).
PI3Ka and ΡΙ3Κβ play an essential role in maintaining homeostasis and pharmacological inhibition of these molecular targets has been associated with cancer therapy (Maira et al. Expert Opin. Ther. Targets 12:223 (2008)).
PI3Ka is involved in insulin signaling and cellular growth pathways (Foukas et al. Nature 441 :366 (2006)). PI3K5 and/or ΡΙ3Κγ isoform-selective inhibition is expected to avoid potential side effects such as hyperglycemia, and metabolic or growth disregulation.
Parasitic infections still represent one of the most important causes of morbidity and mortality worldwide. Among the parasites that cause human and animal pathology the phylum apicomplexa comprises a group of vector-borne parasites that is responsible for a wide variety of serious illnesses including but not limited to malaria, leishmaniasis and trypanosomiasis. Malaria alone infects 5-10% of humanity and causes around two milion deaths per year. [Schofield et al, "Immunological processes in malaria pathogenesis", Nat Rev Imm 2005], [Schofiled L, "Intravascular infiltrates and organ-specific inflammation in
malaria pathogenesis], [Mishra et al, "TLRs in CNS Parasitic infections", Curr Top Micro Imm 2009], [Bottieau et al, "Therapy of vector-borne protozoan infections in nonendemic settings", Expert Rev. Anti infect. Ther, 2011]. Toll-like receptors (TLRs) are germ-line encoded, phylogenetically ancient molecules that recognize evolutionary conserved structural relevant molecules (known as pathogen - associated molecular patterns (PAMPs)) within microbial pathogens. Various different cell types including cells of the immune system express TLRs and are thereby able to detect the presence of PAMPs. Sofar 10 functional TLR family members (TLR1 -10) have been described in humans, all of which recognize specific PAMP molecules. Following recognition of these specific PAMPs TLRs induce and orchestrate the immuneresponse of the host to infections with bacteria, viruses, fungi and parasites. [Hedayat et al, "Targeting of TLRs: a decade of progress in combating infectious disease", review, Lancet Infectious disease 2011], [Kwai et al, "TLRs and their crosstalk with other innate receptors in infection and immunity", review, Immunity May-2011]
The immune system of the infected host responds to infection with the TLR induced production of pro-inflammatory cytokines mainly of the T-helper 1 type (Th1). While adequate amounts of these cytokines are benefical and required to clear the infection an overproduction of these mediators is harmful to the host and associated with immune mediated pathology including neuropathology and tissue damage with severe and often fatal consequences. One prominent and highly relevant example of such immune mediated pathology is acute and cerebral malaria (CM) which causes severe clinical symptoms and is often fatal. [Schofield et al, "Immunological processes in malaria pathogenesis", Nat Rev Imm 2005], [Schofiled L, "Intravascular infiltrates and organ-specific inflammation in malaria pathogenesis], [Mishra et al, "TLRs in CNS Parasitic infections", Curr Top Micro Imm 2009], [Bottieau et al, "Therapy of vector-borne protozoan infections in nonendemic settings", Expert Rev. Anti infect. Ther, 2011] [Hedayat et al, "Targeting of TLRs: a decade of progress in combating infectious disease", review, Lancet Infectious disease 2011]. Despite progress made in treatment and eradication of malaria, the mortality rate that is associated with severe malaria, including CM remains unacceptably high. Strategies directed solely at the eradication of the parasite in the host might therefore not be sufficient to prevent neurological complications and death in all cases of CM. Development of new innovative adjunct therapeutic strategies to efficiently reduce the CM-associated mortality and morbidity that is caused, in part, by host-mediated immunopathology remains therefore an urgent medical need. [Higgins et al, "Immunopathogenesis of falciparum malaria: implications for adjunctive therapy in the management of severe and cerebral malaria", Expert Rev. Anti Infect. Ther. 2011]
Recently further evidence has been provided that TLR9 plays a key role in the recognition and response to parasites including but not limited to Plasmodium, Leishmania,
Trypanosoma and Toxoplasma [Gowda et al, "The Nucleosome is the TLR9-specific Immunostimulatory component of Plasmodium falciparum that activates DCs", PLoS ONE, June 2011], [Peixoto-Rangel et al, "Candidate gene analysis of ocular toxoplasmosis in Brazil: evidence for a role for TLR9", Mem Inst Oswaldo Cruz 2009], [Pellegrini et al, "The role of TLRs and adoptive immunity in the development of protective or pathological immune response triggered by the Trypanosoma cruzi protozoan", Future Microbiol 2011] and that interference with the activation of TLRs including TLR9 represents a promising strategy to prevent the deleterious inflammatory responses in severe and cerebral malaria [Franklin et al, "Therapeutical targeting of nucleic acid-sensing TLRs prevents experimental cerebral malaria", PN AS 2011]
Malaria is an infectious disease caused by four protozoan parasites: Plasmodium falciparum; Plasmodium vivax; Plasmodium ovale; and Plasmodium malaria. These four parasites are typically transmitted by the bite of an infected female Anopheles mosquito. Malaria is a problem in many parts of the world and over the last few decades the malaria burden has steadily increased. An estimated 1 -3 million people die every year from malaria - mostly children under the age of 5. This increase in malaria mortality is due in part to the fact that Plasmodium falciparum, the deadliest malaria parasite, has acquired resistance against nearly all available antimalarial drugs, with the exception of the artemisinin derivatives.
Leishmaniasis is caused by one or more than 20 varieties of parasitic protozoa that belong to the genus Leishmania, and is transmitted by the bite of female sand flies. Leishmaniasis is endemic in about 88 countries, including many tropical and sub-tropical areas. There are four main forms of Leishmaniasis. Visceral leishmaniasis, also called kala-azar, is the most serious form and is caused by the parasite Leishmania donovani. Patients who develop visceral leishmaniasis can die within months unless they receive treatment. The two main therapies for visceral leishmaniasis are the antimony derivatives sodium stibogluconate (Pentostam®) and meglumine antimoniate (Glucantim®). Sodium stibogluconate has been used for about 70 years and resistance to this drug is a growing problem. In addition, the treatment is relatively long and painful, and can cause undesirable side effects.
Human African Trypanosomiasis, also known as sleeping sickness, is a vector-borne parasitic disease. The parasites concerned are protozoa belonging to the Trypanosoma Genus. They are transmitted to humans by tsetse fly (Glossina Genus) bites which have
acquired their infection from human beings or from animals harboring the human pathogenic parasites.
Chagas disease (also called American Trypanosomiasis) is another human parasitic disease that is endemic amongst poor populations on the American continent. The disease is caused by the protozoan parasite Trypanosoma cruzi, which is transmitted to humans by bloodsucking insects. The human disease occurs in two stages: the acute stage, which occurs shortly after infection and the chronic stage, which can develop over many years. Chronic infections result in various neurological disorders, including dementia, damage to the heart muscle and sometimes dilation of the digestive tract, as well as weight loss. Untreated, the chronic disease is often fatal. The drugs currently available for treating Chagas disease are Nifurtimox and benznidazole. However, problems with these current therapies include their diverse side effects, the length of treatment, and the requirement for medical supervision during treatment. Furthermore, treatment is really only effective when given during the acute stage of the disease. Resistance to the two frontline drugs has already occurred. The antifungal agent Amphotericin b has been proposed as a second-line drug, but this drug is costly and relatively toxic.
Toxoplasmosis is endemic in many areas globally and can infect a large proportion of the adult population. However, its prevalence differs in different countries. It is estimated to infect at least 10% of adults in northern temperate countries and more than half of
adults in Mediterranean and tropical contries. Toxoplasma gondii, the causative pathogen of toxoplamosis, is a ubiquitous, obligate intracellular protozoan and is considered to be the most common cause of infective retinitis in humans, which depends on a variety of factors, including climate, hygiene, and dietary habits. The course of disease in immunocompetent adults is usually asymptomatic
and self-limiting. As soon as infection has occurred, the parasite forms latent cysts in the retina and in other organs of the body, which can reactivate years after the initial infection giving rise to acute retinochoroiditis and the formation of new retinochoroidal lesions.
[Arevalo et al, "Ocular Toxoplasmosis in the developing world", Internat. Ophthal. Clin 2010]
Neurocysticercosis is the most common parasitic disease of the CNS (incidence ~2.5 milion worldwide) caused by the larvae of Taenia solium. The disease has a long asymptomatic phase in humans characterized by the absence of a detectable inflammatory response surrounding the parasite. The overall immune response during the asymptomatic phase is of the Th2 phenotype. However, the destruction of larvae by therapeutic treatment or by normal parasite attrition causes a strong inflammatory response, often consisting of a chronic
granulomatous reaction and manifestation of typical symptoms of the disease. The immune response in the CNS of symptomatic patients consists of an overt Th1 phenotype or a mixed Th1 , Th2, and Th3 response, depending upon the absence or presence of granulomas. The hyperinflammatory response prevailing during the symptomatic phase in the CNS is responsible for the severe neuropathology and mortality associated with neurocysticercosis . [Mishra et al, "TLRs in CNS Parasitic infections", Curr Top Micro Imm 2009]
SUMMARY OF THE INVENTION
There is a need to provide new class I PI3 kinase inhibitors that are good drug
candidates. In particular, compounds of the invention should bind potently to class I PI3 kinases whilst showing little affinity for other receptors and show functional activity as inhibitors. They should be well absorbed from the gastrointestinal tract, be metabolically stable and possess favourable pharmacokinetic properties. When targeted against receptors in the central nervous system they should cross the blood brain barrier freely and when targeted selectively against receptors in the peripheral nervous system they should not cross the blood brain barrier. They should be non-toxic and demonstrate few side-effects. Furthermore, the ideal drug candidate will exist in a physical form that is stable, non-hygroscopic and easily formulated. The compounds of the invention show a certain level of selectivity against the different paralogs PI3K α, β, γ and δ. In particular, show a certain level of selectivity for the isoforms PI3K5, ΡΙ3Κγ and ΡΙ3Κβ over the PI3Ka isoform.
The compounds of the present invention are therefore potentially useful in the treatment of a wide range of disorders, particularly disorders including but not limited to
autoimmune disorders, autoinflammatory and inflammatory diseases, allergic diseases, disease or infection associated immunopathologies, airway diseases, such as asthma and COPD, transplant rejection, cancers eg of hematopoietic origin or solid tumors. Various embodiments of the invention are described herein.
Within certain aspects, provided herein is a compound of formula (I) or a pharmaceutically acceptable salt thereof:
C - Iri another embodiment, the invention provides a pharmaceutical composition comprising a therapeutically effective amount of a compound according to the definition of formula (I), or a pharmaceutically acceptable salt thereof, or subformulae thereof and one or more pharmaceutically acceptable carriers.
In another embodiment, the invention provides a combination, in particular a pharmaceutical combination, comprising a therapeutically effective amount of the compound according to the definition of formula (I), or a pharmaceutically acceptable salt thereof, or subformulae thereof and one or more therapeutically active agent. In another embodiment, the invention also relates to the treatment, either alone or in combination, with one or more other pharmacologically active compounds, including methods of treating conditions, diseases or disorders in which one or more of the functions of B cells such as antibody production, antigen presentation, cytokine
production or lymphoid organogenesis are abnormal or are undesirable including rheumatoid arthritis and related diseases (such as ankylosing spondylarthritis, psoriatic arthritis, juvenile arthritis), pemphigus vulgaris and related diseases, idiopathic
thrombocytopenia purpura, systemic lupus erythematosus, multiple sclerosis,
myasthenia gravis, Sjogren's syndrome, Graft versus host disease, autoimmune hemolytic anemia, ANCA-associated vasculitides, cryoglobulinemia, thrombotic
thrombocytopenic purpura, chronic autoimmune urticaria, allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, different types of
glomerulonephritides, AMR (antibody-mediated transplant rejection), B cell-mediated hyperacute, acute and chronic transplant rejection and cancers of haematopoietic origin including but not limited to multiple myeloma; acute myelogenous leukemia; chronic myelogenous leukemia; lymphocytic leukemia; myeloid leukemia; non-Hodgkin
lymphoma; lymphomas; polycythemia vera; essential thrombocythemia; myelofibrosis with myeloid metaplasia; and Walden stroem disease as well as in disease or infection associated immunopathology.
DETAILED DESCRIPTION OF THE INVENTION
The invention provides quinazolin-4-one compounds of the formula (I) and/or
pharmaceutically acceptable salts and/or solvates thereof,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, trideuteromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S; or
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro , methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S;
or
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
wherein
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
Unless specified otherwise, the term "compounds of the present invention" refers to compounds of formula (I) and subformulae thereof, salts of the compound, as well as all stereoisomers (including diastereoisomers and enantiomers), rotamers, tautomers and isotopically labeled compounds (including deuterium substitutions, as well as inherently formed moieties). Where compounds of formula (I) are mentioned, this is meant to include also the tautomers and N-oxides of the compounds of formula (I).
The invention may be more fully appreciated by reference to the following description, including the following glossary of terms and the concluding examples. As used herein, the terms "including", "containing" and "comprising" are used herein in their open, non- limiting sense.
Tautomers, such as tautomers between purine forms can be present for example in the R3 portion of compounds of formula (I). Nitrogen containing heterocyclyl and heteroaryl residues may form N-oxides. Where the plural form is used for compounds, salts, and the like, this is taken to mean also a single compound, salt, or the like.
The general terms used hereinbefore and hereinafter preferably have within the context of this disclosure the following meanings, unless otherwise indicated:
As used herein, the term "C3-C6-cycloalkyl" refers to a 3 to 6 membered monocyclic saturated ring.
In the context of R1, examples of C3-C6-cycloalkyl include cyclopropyl; cyclobutyl;
cyclopentyl and cyclohexyl;
In the context of R1, examples of C3-C6-cycloalkyl which is substituted in the 1 position by methyl include 1 -methylcyclopropyl; 1 -methylcyclobutyl; 1 -methylcyclopentyl and 1 - methylcyclohexyl.
As used herein, all substituents are written in a way to show the order of functional groups (groups) they are composed of. The functional groups are defined herein above.
Various embodiments of the invention are described herein. It will be recognized that features specified in each embodiment may be combined with other specified features to provide further embodiments of the present invention. In one embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Γ)
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
wherein
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Γ)
(!')
wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; -L-R3 is independently selected from
trifluoromethoxy, hydroxy or methylsulfonylamine; and R3 is independently selected from
wherein
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (I")
(I")
wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (I")
(I")
wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Γ")
(Γ')
wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
wherein
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Γ")
(!■")
wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
wherein
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is independently selected from
In one embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (la')
(la'),
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (la')
(la'),
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
wherein
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb')
(lb'),
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb')
(lb'),
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Ic')
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lc')
(ic),
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id')
(Id'),
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S. In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id')
(Id'),
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le')
(le'),
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le')
(le'),
wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
wherein
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
In one embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ΙΑ')
wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl,
cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; -L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ΙΑ')
wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IB')
(IB')
wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino,
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IB')
(IB') wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, -L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IC)
(IC) wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino;
X is independently selected from NH, NMe or S;
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IC)
(IC) wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino;
X is independently selected from NH, NMe or S; -L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ID')
(ID') wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino; -L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ID')
(ID') wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino;
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is attached to L via the trivalent nitrogen atom.
In one embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (la")
(la"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (la")
(la"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb")
(lb"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb")
(lb"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lc")
(lc"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl.chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Ic")
(Ic"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id")
(Id"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id")
(Id"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le")
(le"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le")
(le"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
In one embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IA")
OA") wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; -L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IA")
OA") wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; -L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IB")
(IB") wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino;
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IB")
(IB") wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino;
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IC")
(ic") wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino;
Z is independently selected from NMe or S;
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (IC")
(IC") wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino;
Z is independently selected from NMe or S;
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ID")
(ID") wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino;
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is attached to L via the trivalent nitrogen atom.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (ID")
(ID") wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R4 is independently selected from hydrogen or amino;
-L- is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is attached to L via the trivalent nitrogen atom.
In one embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb'")
(lb"'),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and
R4 is independently selected from hydrogen or amino.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lb'")
(lb'"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R4 is independently selected from hydrogen or amino.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (lc'")
(lc-),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; and
R4 is independently selected from hydrogen or amino.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Ic'")
(IC"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; and
R4 is independently selected from hydrogen or amino.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id'")
(Id'"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; and
R4 is independently selected from hydrogen or amino.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Id'")
(Id'"),
wherein
R is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; and
R4 is independently selected from hydrogen or amino.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le'")
(le'"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; and
R4 is independently selected from hydrogen or amino.
In another embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (le'")
(le'"),
wherein
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; and
R4 is independently selected from hydrogen or amino.
In one embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Γ)
-L-R3 is independently selected from
In one embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (Γ)
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S; and R , R , R , R and L are as defined above for the compound of formula (Γ).
In one embodiment, the invention provides a compound of the formula (I) and/or a pharmaceutically acceptable salt thereof, selected from a compound of the formula (I")
wherein,
-L-R3 is independently selected from
In one embodiment, the invention provides a compound of the formulae (I) or (Γ), (la'), (lb'), (IC), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (Γ"), (lb'"), (Ic'"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
R5 is independently selected from hydrogen or fluoro; and
R6 is independently selected from hydrogen.
In one embodiment, the invention provides a compound of the formulae (I) or (Γ), (la'), (lb'), (IC), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (Γ"), (lb'"), (IC"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, methylsulfonyl, ethylsulfonyl or cyclopropylsulfonyl.
In another embodiment, the invention provides a compound of the formulae (I) or (Γ), (la'), (lb'), (IC), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (I'"), (lb'"), (IC"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
R2 is independently selected from methyl, trifluoromethyl, difluoromethoxy, fluoro or methylsulfonyl.
In one embodiment, the invention provides a compound of the formulae (I) or (Γ), (la'), (lb'), (IC), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (Γ"), (lb'"), (IC"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
R5 is independently selected from hydrogen or fluoro;
R6 is independently selected from hydrogen; and
R2 is independently selected from methyl, trifluoromethyl, difluoromethoxy, fluoro or methylsulfonyl. In one embodiment, the invention provides a compound of the formulae (I) or (Γ), (la'), (lb'), (IC), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (Γ"), (lb'"), (IC"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
R5 is independently selected from fluoro;
R6 is independently selected from hydrogen; and
R2 is independently selected from methyl or fluoro.
In one embodiment, the invention provides a compound of the formulae (I) or (Γ), (la'), (lb'), (IC), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (Γ"), (lb'"), (IC"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
R5 is independently selected from hydrogen;
R6 is independently selected from hydrogen; and
R2 is independently selected from trifluoromethyl, difluoromethoxy or methylsulfonyl.
In one embodiment, the invention provides a compound of the formulae (I) or (Γ), (la'), (lb'), (Ic'), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (Ic"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (Γ"), (lb'"), (Ic'"), (Id'"), (le'") and/or a pharmaceutically acceptable salt thereof, wherein
R5 is independently selected from hydrogen;
R6 is independently selected from hydrogen; and
R2 is independently selected from trifluoromethyl.
In one embodiment, the invention provides a compound of the formulae (Γ), (la'), (lb'), (Ic'), (Id') or (le') and/or a pharmaceutically acceptable salt thereof, wherein
R3 is selected from
R4 is independently selected from hydrogen or amino,
R8 is methyl, and
X is S.
In one embodiment embodiment, the invention provides a compound of the formulae (I"), (la"), (lb"), (Ic"), (Id") or (le") and/or a pharmaceutically acceptable salt thereof, wherein R3 is selected from
wherein
R4 is independently selected from hydrogen or amino,
R8 is methyl, and
Z is S.
In one embodiment, the invention provides a compound of the formulae (Γ), (la'), (lb'), (Ic'), (Id') or (le') and/or a pharmaceutically acceptable salt thereof, wherein
R3 is selected from
R8 is methyl.
In one embodiment embodiment, the invention provides a compound of the formulae (la") or (lb"), (Ic"), (Id") or (le") and/or a pharmaceutically acceptable salt thereof, wherein
R8 is methyl.
In one embodiment, the invention provides a compound of the formulae (la') or (lb'), and/or a pharmaceutically acceptable salt thereof, wherein
R3 is selected from
R4 is independently selected from hydrogen or amino, R8 is methyl, and
X is S.
In one embodiment embodiment, the invention provides a compound of the formulae (la") or (lb") and/or a pharmaceutically acceptable salt thereof, wherein
R3 is selected from
R4 is independently selected from hydrogen or amino, R8 is methyl, and
Z is S.
In one embodiment, the invention provides a compound of the formulae (la') or (lb') and/or a pharmaceutically acceptable salt thereof, wherein
R3 is selected from
R8 is methyl. In one embodiment embodiment, the invention provides a compound of the formulae (la") or (lb") and/or a pharmaceutically acceptable salt thereof, wherein
R8 is methyl.
In one embodiment, the invention provides a compound of the formulae (la') or (lb') wherein R7 is hydroxyl, and/or a pharmaceutically acceptable salt thereof, wherein R3 is selected from
R4 is independently selected from hydrogen or amino, R8 is methyl, and
X is S.
In one embodiment, the invention provides a compound of the formulae (la") or (lb") wherein R7 is hydroxyl, and/or a pharmaceutically acceptable salt thereof, wherein R3 is selected from
R4 is independently selected from hydrogen or amino,
R8 is methyl, and
Z is S.
In one embodiment, the invention provides a compound of the formulae (la') or (lb') wherein R7 is hydroxyl, and/or a pharmaceutically acceptable salt thereof, wherein R3 is selected from
R8 is methyl. In one embodiment embodiment, the invention provides a compound of the formulae (la") or (lb") wherein R7 is hydroxyl, and/or a pharmaceutically acceptable salt thereof, wherein
R8 is methyl. In one embodiment, the invention provides a compound of the formulae (la') or (lb') wherein R7 is hydroxyl, and/or a pharmaceutically acceptable salt thereof, wherein R2 is independently selected from methyl, trifluoromethyl, difluoromethoxy, fluoro or methylsulfonyl; and
R3 is selected from
R4 is independently selected from hydrogen or amino, R8 is methyl, and
X is S.
In one embodiment, the invention provides a compound of the formulae (la") or (lb") wherein R7 is hydroxyl, and/or a pharmaceutically acceptable salt thereof, wherein R2 is independently selected from from methyl, trifluoromethyl, difluoromethoxy, fluoro methylsulfonyl; and
R3 is selected from
R4 is independently selected from hydrogen or amino,
R8 is methyl, and
Z is S.
In one embodiment, the invention provides a compound of the formulae (la') or (lb') wherein R7 is hydroxyl, and/or a pharmaceutically acceptable salt thereof, wherein R2 is independently selected from methyl, trifluoromethyl, difluoromethoxy, fluoro or methylsulfonyl; and
R3 is selected from
R8 is methyl.
In one embodiment embodiment, the invention provides a compound of the formulae (la") or (lb") wherein R7 is hydroxyl, and/or a pharmaceutically acceptable salt thereof, wherein
R2 is independently selected from methyl, trifluoromethyl, difluoromethoxy, fluoro or methylsulfonyl; and
R3 is selected from
R8 is methyl.
The invention also provides following enumerated embodiments:
Embodiment 1 A compound of formula (I)
(I)
or a pharmaceutically acceptable salt thereof wherein
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, trideuteromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S; or
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; -L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S;
or
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro; -L-R3 is independently selected from
wherein
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is independently selected from
Embodiment 2 A compound according to embodiment 1 or a pharmaceutically acceptable salt thereof, of the formula (Γ)
wherein,
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
Embodiment 3 A compound according to embodiment 1 or a pharmaceutically acceptable salt thereof, of the formula (Γ)
(I")
wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl or chloro;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy or methylsulfonylamine; and
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
Embodiment 4 A compound according to embodiment 2 or a pharmaceutically acceptable salt thereof,
wherein
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S;
Embodiment 5 A compound according to embodiment 2 or a pharmaceutically acceptable salt thereof
wherein
R3 is selected from
R4 is independently selected from hydrogen or amino, R8 is methyl, and
X is S.
Embodiment 6 A compound according to embodiment 3 or a pharmaceutically acceptable salt thereof, wherein
R3 is selected from
R4 is independently selected from hydrogen or amino,
R8 is methyl, and
Z is S.
Embodiment 7 A compound according to any one of embodiments 1 to 6 or a pharmaceutically acceptable salt thereof, wherein
R5 is independently selected from hydrogen or fluoro;
R6 is independently selected from hydrogen; and
R2 is independently selected from methyl, trifluoromethyl, difluoromethoxy, fluoro or methylsulfonyl.
Embodiment 8 A compound according to embodiment 1 , selected from
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one, (S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclopropylquinazolin-4(3H)-one, (S)-5-Chloro-3-phenyl-2-(1 -(thiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)quinazolin-4(3H)-one, (S)-2-Amino-4-(2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile,
(S)-5-Chloro-2-(1 -(imidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-methoxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin-4(3H)-one, (S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(pyridin-3-yl)quinazolin-4(3H)-one, (S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(6-methoxypyridin-3-yl)quinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(pyridin-3-yl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-3-(6-methoxypyridin-3-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(pyridin-3-yl)quinazolin-4(3H)-one, (S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(6-methoxypyridin-3-yl)quinazolin- 4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(tetrahydro-2H^yran-4-yl)quinazolin-4(3H one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(tetrahydro-2H-pyran-4-yl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin-4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(dimethylamino)quinazolin-4(3H)-on (S)-4-Amino-6-(2-(5-chloro-3-(dimethylamino)-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolid yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(dimethylamino)quinazoli one,
(S)-2-(1-(5-Aminothiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin- 4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(piperidin-1-yl)quinazolin-4(3H)-one,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(piperidin-1 -yl)quinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(piperidin-1 -yl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(1 -methylcyclopropyl)quinazolin-4(3H)-one, (S)-4-Amino-6-(2-(5-chloro-3-(1 -methylcyclopropyl)-4-oxo-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(1 -methylcyclopropyl)quinazolin- 4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclohexylquinazolin-4(3H)-one,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclohexylquinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-3-cyclohexyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclopropylquinazolin- 4(3H)-one,
(S)-2-(1-(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin- 4(3H)-one,
(S)-2-Amino-4-(2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3^henyl-5-(trifluoro
yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3-(o-tolyl)-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolid yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5,7-difluoro-3-phenylquinazolin-4(3H)-one,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5 ,7-difluoro-3-phenylquinazolin-4(3H)-one, (S)-2-Amino-4-(2-(5 ,7-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5,7-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5,6-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5,6-difluoro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-2-methyl-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrroli^ yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-(difluoromethoxy)-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)-6-methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-(difluoromethoxy)-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1^ yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-methyl-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-5-methyl-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-5-methyl-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)-6-methylpyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(R)-2-(3-(2-Amino-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H^
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-phenylquinazolin-4(3H)-one,
(R)-4-Amino-6-(4-(5-chloro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)thiazolidin-^ yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)thiazolidin-3-yl)- pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(2-cyclopropyl-phenyl)-4-oxo-3,4-dihydroquinazolin-2-yl)- thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(2-(difluoromethoxy)phenyl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro
(R)-2-(3-(2-Amino-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3^henylquinazolin-4(3H)-on
(R)-4-Amino-6-(4-(5-chloro-3-(2-fluorophenyl)-4-oxo-3,4-dihydroquinazolin-2-yl)thiazolidi^ yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(2-(difluoromethoxy)phenyl)quinazoli 4(3H)-one,
(R)-2-(3-(2-Amino-9-methyl-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazoli 4(3H)-one,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(2-cyclopropylphenyl)quinazolin-4(3H)- one,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(1 -methyl-1 H-pyrazol-5-yl)quinazolin- 4(3H)-one,
(R)-4-Amino-6-(4-(5-chloro-3-(1 -methyl-1 H-pyrazol-5-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(5-methylthiophen-2-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(5-methylthiophen-2-yl)quinazoli one,
4- Amino-6-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
5- Chloro-2-((2S,4S)-4-methoxy-1 -(9H^urin-6-yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)- one,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-4-methoxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazo yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-methoxy-2-(4-oxo-3-phenyl-5-(triflu^^
yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(R,S)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H^yrazol-5-yl)-5-chloro-3^henylquinazolin-4(3H)- one,
(R,S)-4-Amino-6-(5-(5-chloro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R,S)-2-Amino-4-(5-(5-chloro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(R,S)-4-Amino-6-(5-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R,S)-4-Amino-6-(5-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4,5-dih pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H^yrazol-5-yl)-5-chloro-3^henylquinazolin-4(3H)- one,
(S or R)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H^yrazol-5-yl)-5-chloro-3^henylquinazolin-4(3H)- one,
(S or R)-4-Amino-6-(5-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)-4,5- dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(S or R)-2-Amino-4-methyl-6-(5-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazoli yl)-4,5-dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-4-Amino-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)-4,5- dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-2-Amino-4-methyl-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2^ yl)-4,5-dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(S or R)-4-Amino-6-(5-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-4-Amino-6-(5-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
N-((3S,5S)-5-(5-Chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-1 -(9H-purin-6- yl)pyrrolidin-3-yl)methanesulfonamide,
((S)-4-Amino-6-(3-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
(S)-2-(2-(9H-Purin-6-yl)isoxazolidin-3-yl)-5-chloro-3-phenylquinazolin-4(3H)-one,
(S)-5-Chloro-3^henyl-2-(2-(thiazolo[5,4-d]pyrimidin-7-yl)isoxazolidin-3-yl)quinazolin-4(3H^ one,
(S)-2-(2-(9H-Purin-6-yl)isoxazolidin-3-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(3-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(3-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
5-Chloro-2-((2S,4S)-4-hydroxy-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)- one,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4- Amino-6-((2S,4S)-4-hydroxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
5- Chloro-2-((2S,4S)-4-(difluoromethoxy)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenylquinazolin- 4(3H)-one,
(S)-4-Amino-6-(2-(5-hydroxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin- 4(3H)-one,
(S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-morpholino- quinazolin-4(3H)-one,
(S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro^
tolyl)quinazolin-4(3H)-one,
2-((2S,4S)-1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)-4-methoxypyrrolidin-2-y ^ phenylquinazolin-4(3H)-one,
2-((S)-1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((S)-1 -(methylsulfonyl)pyrroli yl)quinazolin-4(3H)-one,
(S)-Methyl 3-(2-((S)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4-oxoquinazolin-3(4H)- yl)pyrrolidine-1 -carboxylate,
(R)-Methyl 3-(2-((S)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4-oxoquinazolin-3(4H)- yl)pyrrolidine-1 -carboxylate,
2-((S)-1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((R)-1 -(methylsulfonyl)pyrrolidi^ yl)quinazolin-4(3H)-one,
2-((S)-1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chto
yl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile, or
(S)-2-Amino-4-methyl-6-(2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolm yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
or a pharmaceutically acceptable salt thereof.
Embodiment 9 A pharmaceutical composition comprising a therapeutically effective amount of a compound according to any one of embodiments 1 to 8 or a pharmaceutically acceptable salt thereof and one or more pharmaceutically acceptable carriers.
Embodiment 10 A combination comprising a therapeutically effective amount of a compound according to any one of embodiments 1 to 8 or a pharmaceutically acceptable salt thereof and one or more therapeutically active co-agents. Embodiment 1 1 A method of modulating the activity of the class I PI3 kinases, in a subject, wherein the method comprises administering to the subject a therapeutically effective amount of a compound according to any one of embodiments 1 to 8 or a pharmaceutically acceptable salt thereof. Embodiment 12 A method of treating a disorder or a disease selected from rheumatoid arthritis (RA), pemphigus vulgaris (PV), endemic form of Brazilian pemphigus (Fogo selvagem), idiopathic thrombocytopenia purpura (ITP), thrombotic
thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA-associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection, cancers of haematopoietic origin, severe and cerebral malaria, trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis comprising administering to a subject a therapeutically effective amount of a compound according to any one of embodiments 1 to 8 or a pharmaceutically acceptable salt thereof.
Embodiment 13 A compound according to any one of embodiments 1 to 8 or a pharmaceutically acceptable salt thereof, for use as a medicament.
Embodiment 14 A compound according to any one of embodiments 1 to 8 or a pharmaceutically acceptable salt thereof, for use in the treatment of a disorder or a disease selected from rheumatoid arthritis (RA), pemphigus vulgaris (PV), endemic form of Brazilian pemphigus (Fogo selvagem), idiopathic thrombocytopenia purpura (ITP), thrombotic thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA-associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection, cancers of haematopoietic origin, severe and cerebral malaria,
trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis.
Embodiment 15 Use of a compound according to anyone of embodiments 1 to 8 or a pharmeceutically acceptable salt therof in the manufacture of a medicament for the treatment of a disorder or a disease selected from rheumatoid arthritis (RA), pemphigus vulgaris (PV), endemic form of Brazilian pemphigus (Fogo selvagem), idiopathic thrombocytopenia purpura (ITP), thrombotic thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA-associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection, cancers of haematopoietic origin, severe
and cerebral malaria, trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis.
In one embodiment embodiment, the invention provides a compound of the formulae (lb'), (lb") or (lb'") and/or a pharmaceutically acceptable salt thereof, wherein
R7 is hydroxy.
In one embodiment embodiment, the invention provides a compound of the formula (I), selected from
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one, (S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclopropylquinazolin-4(3H)-one, (S)-5-Chloro-3-phenyl-2-(1 -(thiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)quinazolin-4(3H)-one, (S)-2-Amino-4-(2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile,
(S)-5-Chloro-2-(1 -(imidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-methoxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin-4(3H)-one, (S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(pyridin-3-yl)quinazolin-4(3H)-one, (S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(6-methoxypyridin-3-yl)quinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(pyridin-3-yl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-3-(6-methoxypyridin-3-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(pyridin-3-yl)quinazolin-4(3H)-one,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-c^
4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(tetrahydro-2H^yran-4-yl)quinazolin-4(3H one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(tetrahydro-2H-pyran-4-yl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin-4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(dimethylamino)quinazolin-4(3H)-on (S)-4-Amino-6-(2-(5-chloro-3-(dimethylamino)-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolid yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(dimethylamino)quinazoli one,
(S)-2-(1-(5-Aminothiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin- 4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(piperidin-1-yl)quinazolin-4(3H)-one,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(piperidin-1 -yl)quinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(piperidin-1 -yl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(1 -methylcyclopropyl)quinazolin-4(3H)-one, (S)-4-Amino-6-(2-(5-chloro-3-(1 -methylcyclopropyl)-4-oxo-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(1 -methylcyclopropyl)quinazolin- 4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclohexylquinazolin-4(3H)-one,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclohexylquinazolin-4(3H)-one, (S)-4-Amino-6-(2-(5-chloro-3-cyclohexyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclopropylquinazolin- 4(3H)-one,
(S)-2-(1-(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin- 4(3H)-one,
(S)-2-Amino-4-(2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrroli^ yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3-(o-tolyl)-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolid yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5,7-difluoro-3-phenylquinazolin-4(3H)-one,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5 ,7-difluoro-3-phenylquinazolin-4(3H)-one, (S)-2-Amino-4-(2-(5 ,7-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5,7-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5,6-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5,6-difluoro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-2-methyl-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrroli^ yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-(difluoromethoxy)-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)-6-methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-(difluoromethoxy)-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1^ yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-methyl-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-5-methyl-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-5-methyl-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)-6-methylpyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(R)-2-(3-(2-Amino-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazolin-4(^ (R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-phenylquinazolin-4(3H)-one,
(R)-4-Amino-6-(4-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)thiazolidin-3- yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)thiazolidin-3-yl)- pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(2-cyclopropyl-phenyl)-4-oxo-3,4-dihydroquinazolin-2-yl)- thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(2-(difluoromethoxy)phenyl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro
(R)-2-(3-(2-Amino-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3^henylquinazolin-4(3H)-on (R)-4-Amino-6-(4-(5-chloro-3-(2-fluorophenyl)-4-oxo-3,4-dihydroquinazolin-2-yl)thiazolidi^ yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(2-(difluoromethoxy)phenyl)quinazoli 4(3H)-one,
(R)-2-(3-(2-Amino-9-methyl-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazoli 4(3H)-one,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(2-cyclopropylphenyl)quinazolin-4(3H)- one,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(1 -methyl-1 H^yrazol-5-yl)quinazolin 4(3H)-one,
(R)-4-Amino-6-(4-(5-chloro-3-(1 -methyl-1 H-pyrazol-5-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(5-methylthiophen-2-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(5-methylthiophen-2-yl)quinazoN one,
4- Amino-6-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
5- Chloro-2-((2S,4S)-4-methoxy-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)- one,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-4-methoxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-methoxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(R,S)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H-pyrazol-5-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(R,S)-4-Amino-6-(5-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R,S)-2-Amino-4-(5-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(R,S)-4-Amino-6-(5-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R,S)-4-Amino-6-(5-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H-pyrazol-5-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(S or R)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H-pyrazol-5-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(S or R)-4-Amino-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)-4,5- dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(S or R)-2-Amino-4-methyl-6-(5-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)-4,5-dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-4-Amino-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)-4,5- dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-2-Amino-4-methyl-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)-4,5-dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(S or R)-4-Amino-6-(5-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-4-Amino-6-(5-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolm^ H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
N-((3S,5S)-5-(5-Chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-1 -(9H-purin-6- yl)pyrrolidin-3-yl)methanesulfonamide,
((S)-4-Amino-6-(3-(5-chloro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
(S)-2-(2-(9H-Purin-6-yl)isoxazolidin-3-yl)-5-chloro-3-phenylquinazolin-4(3H)-one,
(S)-5-Chloro-3^henyl-2-(2-(thiazolo[5,4-d]pyrimidin-7-yl)isoxazolidin-3-yl)quinazolin one,
(S)-2-(2-(9H-Purin-6-yl)isoxazolidin-3-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(3-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(3-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazol^ yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
5-Chloro-2-((2S,4S)-4-hydroxy-1 -(9H^urin-6-yl)pyrrolidin-2-yl)-3^henylquinazolin-4(3H)- one,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4- Amino-6-((2S,4S)-4-hydroxy-2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
5- Chloro-2-((2S,4S)-4-(difluoromethoxy)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenylquinazolin- 4(3H)-one,
(S)-4-Amino-6-(2-(5-hydroxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin- 4(3H)-one,
(S)-2-(1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-morpholino- quinazolin-4(3H)-one,
(S)-2-(1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-(o- tolyl)quinazolin-4(3H)-one,
2-((2S,4S)-1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)-4-methoxypyrrolidin-2-yl)-5-chloro phenylquinazolin-4(3H)-one,
2-((S)-1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((S)-1 -(methylsulfonyl)pyrrolidin-3- yl)quinazolin-4(3H)-one,
(S)-Methyl 3-(2-((S)-1-(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4-oxoquinazolin-3(4H)- yl)pyrrolidine-1-carboxylate,
(R)-Methyl 3-(2-((S)-1-(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4-oxoquinazolin-3(4H)- yl)pyrrolidine-1 -carboxylate,
2-((S)-1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((R)-1 -(methylsulfonyl)pyrrolidin-3- yl)quinazolin-4(3H)-one,
2-((S)-1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((R)-1-(cyclopropylsulfonyl)pyrrolidin-3- yl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile, or
(S)-2-Amino-4-methyl-6-(2-(5-(methylsulfonyl)-4-oxo-3^henyl-3,4-dihydroquinazolin yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
and/or a pharmaceutically acceptable salt thereof.
In another embodiment embodiment, the invention provides a compound of the formula (I), selected from
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one, (S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclopropylquinazolin-4(3H)-one, (S)-5-Chloro-3-phenyl-2-(1-(thiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)quinazolin-4(3H)-one, (S)-2-Amino-4-(2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-5-Chloro-2-(1 -(imidazo[2,1-f][1 ,2,4]triazin-4^
one,
(S)-4-Amino-6-(2-(5-methoxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin-4(3H^ (S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(pyridin-3-yl)quinazolin-4(3H)-one, (S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(6-methoxypyridin-3-yl)quinazolin-4(3H one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(pyridin-3-yl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-3-(6-methoxypyridin-3-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(pyridin-3-yl)quinazolin-4(3H)-one, (S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(6-methoxypyridin-3-yl)quinazolin- 4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(tetrahydro-2H-pyran-4-yl)quinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(tetrahydro-2H-pyran-4-yl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin-4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(dimethylamino)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-3-(dimethylamino)-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(dimethylamino)quinazolin-4(3H^ one,
(S)-2-(1-(5-Aminothiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin- 4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(piperidin-1-yl)quinazolin-4(3H)-one,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(piperidin-1 -yl)quinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(piperidin-1 -yl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(1 -methylcyclopropyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-3-(1 -methylcyclopropyl)-4-oxo-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(1 -methylcyclopropyl)quinazolin- 4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclohexylquinazolin-4(3H)-one,
(S)-2-(1-(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclohexylquinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-3-cyclohexyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclopropylquinazolin- 4(3H)-one,
(S)-2-(1-(2-Amino-9-methyl-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazoli 4(3H)-one,
(S)-2-Amino-4-(2-(5-chloro-3-morpholino-4-oxo-3,4^
methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrroli^ yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3-(o-tolyl)-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolid yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5,7-difluoro-3-phenylquinazolin-4(3H)-one,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5 ,7-difluoro-3-phenylquinazolin-4(3H)-one, (S)-2-Amino-4-(2-(5 ,7-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5,7-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5,6-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5,6-difluoro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-2-methyl-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrroli^ yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-(difluoromethoxy)-4-oxo-3^
yl)-6-methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-(difluoromethoxy)-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1^ yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-methyl-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-5-methyl-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-5-methyl-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)-6-methylpyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(5-Aminothiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
(S)-2-Amino-4-(2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(S)-2-Amino-4-methyl-6-(2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4-dihydroquin yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(3-(5-fluoropyridin-3-yl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-3-(5-fluoropyridin-3-yl)-5- (trifluoromethyl)quinazolin-4(3H)-one,
(S)-2-Amino-4-methyl-6-(2-(4-oxo-3-(pyrimidin-5-yl)-5-(trifluoromethyl)-3,4-dihydroqu 2-yl) pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-2,4-Diamino-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrroli^ 1 -yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(R)-2-(3-(2-Amino-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazolin-4(^ (R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-phenylquinazolin-4(3H)-one,
(R)-4-Amino-6-(4-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)thiazolidin-3- yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)thiazolidin-3-yl)- pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(2-cyclopropyl-phenyl)-4-oxo-3,4-dihydroquinazolin-2-yl)- thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(2-(difluoromethoxy)phenyl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro
(R)-2-(3-(2-Amino-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3^henylquinazolin-4(3H)-on (R)-4-Amino-6-(4-(5-chloro-3-(2-fluorophenyl)-4-oxo-3,4-dihydroquinazolin-2-yl)thiazolidi^ yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(2-(difluoromethoxy)phenyl)quinazoli 4(3H)-one,
(R)-2-(3-(2-Amino-9-methyl-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazoli 4(3H)-one,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(2-cyclopropylphenyl)quinazolin-4(3H)- one,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(1 -methyl-1 H^yrazol-5-yl)quinazolin 4(3H)-one,
(R)-4-Amino-6-(4-(5-chloro-3-(1 -methyl-1 H-pyrazol-5-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(5-methylthiophen-2-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(5-methylthiophen-2-yl)quinazoN one,
4- Amino-6-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
5- Chloro-2-((2S,4S)-4-methoxy-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)- one,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-4-methoxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-methoxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(R,S)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H-pyrazol-5-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(R,S)-4-Amino-6-(5-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R,S)-2-Amino-4-(5-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(R,S)-4-Amino-6-(5-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R,S)-4-Amino-6-(5-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H-pyrazol-5-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(S or R)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H-pyrazol-5-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(S or R)-4-Amino-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)-4,5- dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(S or R)-2-Amino-4-methyl-6-(5-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)-4,5-dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-4-Amino-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)-4,5- dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-2-Amino-4-methyl-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)-4,5-dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(S or R)-4-Amino-6-(5-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-4-Amino-6-(5-(5-methyl-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)-4,5-dih H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
N-((3S,5S)-5-(5-Chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-1 -(9H-purin-6- yl)pyrrolidin-3-yl)methanesulfonamide,
((S)-4-Amino-6-(3-(5-chloro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
(S)-2-(2-(9H-Purin-6-yl)isoxazolidin-3-yl)-5-chloro-3^henylquinazolin-4(3H)-one,
(S)-5-Chloro-3^henyl-2-(2-(thiazolo[5,4-d]pyrimidin-7-yl)isoxazolidin-3-yl)quinazolin one,
(S)-2-(2-(9H-Purin-6-yl)isoxazolidin-3-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(3-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(3-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazoli yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
5-Chloro-2-((2S,4S)-4-hydroxy-1 -(9H^urin-6-yl)pyrrolidin-2-yl)-3^henylquinazolin-4(3H)- one,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-4-hydroxy-2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-((2S,4S)-1 -(5-Aminothiazolo[5,4-d]pyrimidin-7-yl)-4-hydroxypyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
2-Amino-4-((2S,4S)-2-(5-(difluoromethoxy)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(pyridin-3-yl)-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(3-(3,5-difluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin- 2-yl)-4-hydroxypyrrolidin-1-yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(3-morpholino-4-oxo-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(3-(2-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)-4-hydroxypyrrolidin-1-yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5,6-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(6-fluoro-5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(6-fluoro-5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin-^^ yl)-4-hydroxypyrrolidin-1-yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin yl)-4-hydroxypyrrolidin-1-yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-4-hydroxy-2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1-yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-6-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-6-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-((2S,4S)-4-Hydroxy-1-(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenyl-5-(trifluoromethyl)quinazolin- 4(3H)-one,
2-((2S,4S)-1-(2-Amino-9H-purin-6-yl)-4-hydroxypyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
4-Amino-6-((2S,4S)-2-(5,6-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-cyclopropyl-6-fluoro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
5-Chloro-2-((2S,4S)-4-(difluoromethoxy)-1-(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenylquinazolin- 4(3H)-one,
(S)-4-Amino-6-(2-(5-hydroxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin- 4(3H)-one,
(S)-2-(1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-morpholino- quinazolin-4(3H)-one,
(S)-2-(1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-(o- tolyl)quinazolin-4(3H)-one,
2-((2S,4S)-1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)-4-methoxypyrrolidin-2-yl)-5-chloro phenylquinazolin-4(3H)-one,
2-((2S,4S)-1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)-4-hydroxypyrrolidin-2-yl)-3^henyl-5^ (trifluoromethyl)quinazolin-4(3H)-one,
2-((2S,4S)-1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)-4-hydroxypyrrolidin-2-yl)-5-chloro-3- phenylquinazolin-4(3H)-one,
2-((S)-1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((S)-1 -(methylsulfonyl)pyrrolidin-3- yl)quinazolin-4(3H)-one,
(S)-Methyl 3-(2-((S)-1-(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4-oxoquinazolin-3(4H)- yl)pyrrolidine-1 -carboxylate,
(R)-Methyl 3-(2-((S)-1-(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4-oxoquinazolin-3(4H)- yl)pyrrolidine-1 -carboxylate,
2-((S)-1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((R)-1 -(methylsulfonyl)pyrrolidi yl)quinazolin-4(3H)-one,
2-((S)-1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((R)-1-(cyclopropylsulfonyl)pyrrolidin-3 yl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-methyl-6-(2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-(difluoromethyl)-6-((2S,4S)-4-hydroxy-2-(4-oxo-3^henyl-5-(trifluorom dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
2-Amino-4-((2S,4S)-4-fluoro-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazoli yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,2-((2S,4S)-4-Fluoro-1 -(9H^un yl)pyrrolidin-2-yl)-3^henyl-5-(trifluoromethyl)quinazolin-4(3H)-one,
2-((2S,4S)-1 -(2-Amino-9H-purin-6-yl)-4-fluoropyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
4-Amino-6-((2S,4S)-4-fluoro-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazoli yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-((2S,4S)-1 -(2-Amino-9H^urin-6-yl)-4-fluoropyrrolidin-2-yl)-3-(pyridin-3-yl)-5- (trifluoromethyl)quinazolin-4(3H)-one,
2-Amino-4-((2S,4S)-4-fluoro-2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-((2S,4S)-1 -(2-Amino-9H^urin-6-yl)-4-fluoropyrrolidin-2-yl)-3-(3-fluorophenyl)-5- (trifluoromethyl)quinazolin-4(3H)-one, or
2-Amino-4-((2S,4S)-4-fluoro-2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
and/or a pharmaceutically acceptable salt thereof. In another embodiment individual compounds according to the invention are those listed in the Examples section below.
Depending on the choice of the starting materials and procedures, the compounds can be present in the form of one of the possible isomers or as mixtures thereof, for example as pure optical isomers, or as isomer mixtures, such as racemates and diastereoisomer mixtures, depending on the number of asymmetric carbon atoms. The present invention is meant to include all such possible isomers, including racemic mixtures, diasteriomeric mixtures and optically pure forms. Optically active (R)- and (S)- isomers may be prepared using chiral synthons or chiral reagents, or resolved using conventional techniques. If the compound contains a double bond, the substituent may be E or Z configuration . If the compound contains a disubstituted cycloalkyl, the cycloalkyl substituent may have a cis- or trans-configuration. All tautomeric forms are also intended to be included . As used herein, the terms "salt" or "salts" refers to an acid addition or base addition salt of a compound of the invention . "Salts" include in particular "pharmaceutical acceptable salts". The term "pharmaceutically acceptable salts" refers to salts that retain the
biological effectiveness and properties of the compounds of this invention and, which typically are not biologically or otherwise undesirable. In many cases, the compounds of the present invention are capable of forming acid and/or base salts by virtue of the presence of amino and/or carboxyl groups or groups similar thereto.
Pharmaceutically acceptable acid addition salts can be formed with inorganic acids and organic acids.
Inorganic acids from which salts can be derived include, for example, hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like.
Organic acids from which salts can be derived include, for example, acetic acid, propionic acid, glycolic acid, oxalic acid, maleic acid, malonic acid, succinic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, toluenesulfonic acid, sulfosalicylic acid, and the like.
Pharmaceutically acceptable base addition salts can be formed with inorganic and organic bases.
Inorganic bases from which salts can be derived include, for example, ammonium salts and metals from columns I to XII of the periodic table. In certain embodiments, the salts are derived from sodium, potassium, ammonium, calcium, magnesium, iron, silver, zinc, and copper; particularly suitable salts include ammonium, potassium, sodium, calcium and magnesium salts. Organic bases from which salts can be derived include, for example, primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, basic ion exchange resins, and the like. Certain organic amines include isopropylamine, benzathine, cholinate, diethanolamine, diethylamine, lysine, meglumine, piperazine and tromethamine.
In another aspect, the present invention provides compounds of formula (I) in acetate, ascorbate, adipate, aspartate, benzoate, besylate, bromide/hydrobromide,
bicarbonate/carbonate, bisulfate/sulfate, camphorsulfonate, caprate,
chloride/hydrochloride, chlortheophyllonate, citrate, ethandisulfonate, fumarate, gluceptate, gluconate, glucuronate, glutamate, glutarate, glycolate, hippurate, hydroiodide/iodide, isethionate, lactate, lactobionate, laurylsulfate, malate, maleate, malonate, mandelate, mesylate, methylsulphate, mucate, naphthoate, napsylate,
nicotinate, nitrate, octadecanoate, oleate, oxalate, palmitate, pamoate, phosphate/hydrogen phosphate/dihydrogen phosphate, polygalacturonate, propionate, sebacate, stearate, succinate, subsalicylate, sulfate, tartrate, tosylate trifenatate, trifluoroacetate or xinafoate salt form.
Any formula given herein is also intended to represent unlabeled forms as well as isotopically labeled forms of the compounds. Isotopically labeled compounds have structures depicted by the formulas given herein except that one or more atoms are replaced by an atom having a selected atomic mass or mass number. Examples of isotopes that can be incorporated into compounds of the invention include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorous, fluorine, and chlorine, such as 2H, 3H, 11C, 13C, 14C, 15N, 18F 31P, 32P, 35S, 36CI, 123l, 124l, 125l respectively. The invention includes various isotopically labeled compounds as defined herein, for example those into which radioactive isotopes, such as 3H and 14C, or those into which non-radioactive isotopes, such as 2H and 13C are present. Such isotopically labelled compounds are useful in metabolic studies (with 14C), reaction kinetic studies (with, for example 2H or 3H), detection or imaging techniques, such as positron emission tomography (PET) or single- photon emission computed tomography (SPECT) including drug or substrate tissue distribution assays, or in radioactive treatment of patients. In particular, an 18F or labeled compound may be particularly desirable for PET or SPECT studies. Isotopically-labeled compounds of formula (I) can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described in the accompanying Examples and Preparations using an appropriate isotopically-labeled reagent in place of the non-labeled reagent previously employed.
Further, substitution with heavier isotopes, particularly deuterium (i.e., 2H or D) may afford certain therapeutic advantages resulting from greater metabolic stability, for example increased in vivo half-life or reduced dosage requirements or an improvement in therapeutic index. It is understood that deuterium in this context is regarded as a substituent of a compound of the formula (I). The concentration of such a heavier isotope, specifically deuterium, may be defined by the isotopic enrichment factor. The term "isotopic enrichment factor" as used herein means the ratio between the isotopic abundance and the natural abundance of a specified isotope. If a substituent in a compound of this invention is denoted deuterium, such compound has an isotopic enrichment factor for each designated deuterium atom of at least 3500 (52.5% deuterium incorporation at each designated deuterium atom), at least 4000 (60% deuterium incorporation), at least 4500 (67.5% deuterium incorporation), at least 5000 (75%
deuterium incorporation), at least 5500 (82.5% deuterium incorporation), at least 6000 (90% deuterium incorporation), at least 6333.3 (95% deuterium incorporation), at least 6466.7 (97% deuterium incorporation), at least 6600 (99% deuterium incorporation), or at least 6633.3 (99.5% deuterium incorporation).
Pharmaceutically acceptable solvates in accordance with the invention include those wherein the solvent of crystallization may be isotopically substituted, e.g. D20, de- acetone, DMSO-d6. Compounds of the invention, i.e. compounds of formula (I) that contain groups capable of acting as donors and/or acceptors for hydrogen bonds may be capable of forming co- crystals with suitable co-crystal formers. These co-crystals may be prepared from compounds of formula (I) by known co-crystal forming procedures. Such procedures include grinding, heating, co-subliming, co-melting, or contacting in solution compounds of formula (I) with the co-crystal former under crystallization conditions and isolating co- crystals thereby formed. Suitable co-crystal formers include those described in WO 2004/078163. Hence the invention further provides co-crystals comprising a compound of formula (I). As used herein, the term "pharmaceutically acceptable carrier" includes any and all solvents, dispersion media, coatings, surfactants, antioxidants, preservatives (e.g., antibacterial agents, antifungal agents), isotonic agents, absorption delaying agents, salts, preservatives, drug stabilizers, binders, excipients, disintegration agents, lubricants, sweetening agents, flavoring agents, dyes, and the like and combinations thereof, as would be known to those skilled in the art (see, for example, Remington's Pharmaceutical Sciences, 18th Ed. Mack Printing Company, 1990, pp. 1289- 1329). Except insofar as any conventional carrier is incompatible with the active ingredient, its use in the therapeutic or pharmaceutical compositions is contemplated.
The term "a therapeutically effective amount" of a compound of the present invention refers to an amount of the compound of the present invention that will elicit the biological or medical response of a subject, for example, reduction or inhibition of an enzyme or a protein activity, or ameliorate symptoms, alleviate conditions, slow or delay disease progression, or prevent a disease, etc. In one non-limiting embodiment, the term "a therapeutically effective amount" refers to the amount of the compound of the present invention that, when administered to a subject, is effective to (1) at least partially alleviate, inhibit, prevent and/or ameliorate a condition, or a disorder or a disease (i) mediated by class I PI3 kinases or (ii) associated with class I PI3 kinase activity, or (iii)
characterized by activity (normal or abnormal) of class I PI3 kinases or (2) reduce or inhibit the activity of class I PI3 kinases or (3) reduce or inhibit the expression of class I PI3 kinases. In another non-limiting embodiment, the term "a therapeutically effective amount" refers to the amount of the compound of the present invention that, when administered to a cell, or a tissue, or a non-cellular biological material, or a medium, is effective to at least partially reducing or inhibiting the activity of class I PI3 kinases ; or at least partially reducing or inhibiting the expression of class I PI3 kinases. The meaning of the term "a therapeutically effective amount" as illustrated in the above embodiment for class I PI3 kinases also applies by the same means to any other relevant proteins/peptides/enzymes.
As used herein, the term "subject" refers to an animal. Typically the animal is a mammal. A subject also refers to for example, primates (e.g. , humans, male or female), cows, sheep, goats, horses, dogs, cats, rabbits, rats, mice, fish, birds and the like. In certain embodiments, the subject is a primate. In yet other embodiments, the subject is a human.
As used herein, the term "inhibit", "inhibition" or "inhibiting" refers to the reduction or suppression of a given condition, symptom, or disorder, or disease, or a significant decrease in the baseline activity of a biological activity or process.
As used herein, the term "treat", "treating" or "treatment" of any disease or disorder refers in one embodiment, to ameliorating the disease or disorder (i.e., slowing or arresting or reducing the development of the disease or at least one of the clinical symptoms thereof). In another embodiment "treat", "treating" or "treatment" refers to alleviating or ameliorating at least one physical parameter including those which may not be discernible by the patient. In yet another embodiment, "treat", "treating" or
"treatment" refers to modulating the disease or disorder, either physically, (e.g., stabilization of a discernible symptom), physiologically, (e.g. , stabilization of a physical parameter), or both. In yet another embodiment, "treat", "treating" or "treatment" refers to preventing or delaying the onset or development or progression of the disease or disorder.
As used herein, a subject is "in need of a treatment if such subject would benefit biologically, medically or in quality of life from such treatment.
As used herein, the term "a," "an," "the" and similar terms used in the context of the present invention (especially in the context of the claims) are to be construed to cover
both the singular and plural unless otherwise indicated herein or clearly contradicted by the context.
All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly contradicted by context. The use of any and all examples, or exemplary language (e.g. "such as") provided herein is intended merely to better illuminate the invention and does not pose a limitation on the scope of the invention otherwise claimed. Any asymmetric atom (e.g., carbon or the like) of the compound(s) of the present invention can be present in racemic or enantiomerically enriched, for example the ( ?)-, (S)- or (R,S)- configuration. In certain embodiments, each asymmetric atom has at least 50% enantiomeric excess, at least 60% enantiomeric excess, at least 70% enantiomeric excess, at least 80% enantiomeric excess, at least 90% enantiomeric excess, at least 95% enantiomeric excess, or at least 99% enantiomeric excess in the (R)- or (S)- configuration. Substituents at atoms with unsaturated double bonds may, if possible, be present in cis- (Z)- or trans- (£)- form.
Accordingly, as used herein a compound of the present invention can be in the form of one of the possible isomers, rotamers, atropisomers, tautomers or mixtures thereof, for example, as substantially pure geometric (cis or trans) isomers, diastereomers, optical isomers (antipodes), racemates or mixtures thereof. Diastereomeric atropisomers may be present in certain compounds of formula (I) with respect to hindered rotation around bond N-R1.
Any resulting mixtures of isomers can be separated on the basis of the physicochemical differences of the constituents, into the pure or substantially pure geometric or optical isomers, diastereomers, racemates, for example, by chromatography and/or fractional crystallization.
Any resulting racemates of final products or intermediates can be resolved into the optical antipodes by known methods, e.g. , by separation of the diastereomeric salts thereof, obtained with an optically active acid or base, and liberating the optically active acidic or basic compound. In particular, a basic moiety may thus be employed to resolve the compounds of the present invention into their optical antipodes, e.g. , by fractional crystallization of a salt formed with an optically active acid, e.g., tartaric acid, dibenzoyl tartaric acid, diacetyl tartaric acid, di-0, 0'-p-toluoyl tartaric acid, mandelic acid, malic
acid or camphor-10-sulfonic acid. Racemic products can also be resolved by chiral chromatography, e.g. , high pressure liquid chromatography (HPLC) using a chiral adsorbent. Furthermore, the compounds of the present invention, including their salts, can also be obtained in the form of their hydrates, or include other solvents used for their crystallization. The compounds of the present invention may inherently or by design form solvates with pharmaceutically acceptable solvents (including water); therefore, it is intended that the invention embrace both solvated and unsolvated forms. The term "solvate" refers to a molecular complex of a compound of the present invention (including pharmaceutically acceptable salts thereof) with one or more solvent molecules. Such solvent molecules are those commonly used in the pharmaceutical art, which are known to be innocuous to the recipient, e.g., water, ethanol, and the like. The term "hydrate" refers to the complex where the solvent molecule is water.
The compounds of the present invention, including salts, hydrates and solvates thereof, may inherently or by design form polymorphs.
Typically, the compounds of formula (I) can be prepared according to
the methods provided infra.
In one embodiment, the invention relates to a process for manufacturing a compound of formula (I) according to steps a, a', b, b', c and c' (Scheme A), wherein a', b' and c' denote optional functionalization steps or functional group adjustment steps, as required.
The compound of formula (I), wherein is obtained via the coupling reaction step step c of a coumpound of formula (D),
R1' is R1, as defined above for a compound of formula (I), or a substituent that can be transferred into R1 via functionalization steps or functional group adjustment steps;
R2' is R2, as defined above for a compound of formula (I), or a substituent that can be transferred into R2 via functionalization steps or functional group adjustment steps;
R5' is R5, as defined above for a compound of formula (I), or a substituent that can be transferred into R5 via functionalization steps or functional group adjustment steps;
R6' is R6, as defined above for a compound of formula (I), or a substituent that can be transferred into R6 via functionalization steps or functional group adjustment steps; and
U is L, as defined above for a compound of formula (I), or a substituent that can be transferred into L via functionalization steps or functional group adjustment steps;
with R3'-Hal
wherein
R3' is R3, as defined above for a compound of formula (I), or a substituent that can be transferred into R3 via functionalization steps or functional group adjustment steps; and Hal represents halogen, such as chloro, bromo or iodo;
wherein in one embodiment, the coupling reaction is carried out in the presence of an amine base such as Ν,Ν-diisopropylethylamine. The reaction is carried out in the presence of an organic solvent such as an alcohol under microwave heating or conventional heating in an oil bath at a temperature range from 120-160°C for 30 minutes to 6 days;
Alternatively, the reaction is carried out under customary Buchwald-Hartwig conditions using a suitable Pd catalyst/ligand combination such as Pd2(dba)3/2- (dicyclohexylphosphino)biphenyl or Pd2(dba)3/2-dicyclohexylphosphino-2',4',6'- triisopropyl-biphenyl, Pd2(dba)3/XPhos, Pd2(dba)3/(rac)-BINAP, Pd(OAc)2/(rac)-BINAP or bis(tri-t-butylphosphine)palladium and a suitable base, such as NaOtBu, Cs2C03 or K3P04 and organic solvent such as toluene, dioxane or THF. The reaction is stirred at a temperature of approximately 60-140°C, for example at 100°C to 1 10°C and is optionally performed in a microwave reactor. The reaction is preferably carried out under an inert gas such as nitrogen or argon;
optionally followed by functionalization steps or functional group adjustment steps c'.
The compound of formula (D) is obtained via step b of deprotecting PG from the compound of formula (C),
wherein PG represents a suitable protecting group, such as a Boc group, and the other substituents are as defined above;
wherein in one embodiment, where PG is a Boc group, the deprotection reaction is carried out in an organic solvent such as THF or DCM in the presence of an organic acid, such as trifluoroacetic acid or in the presence of an inorganic acid such as HCI or
H3PO4, optionally in the presence of water. The reaction is stirred at room temperature for approximately 60-140°C, for example at 100°C to 1 10°C;
optionally followed by functionalization steps or functional group adjustment steps b'. The compound of formula (C) is obtained via step a of reacting a compound of formula (A),
with a compound of formula (B),
COOH
PG
(B) wherein the substituents are as defined above;
followed by reaction with R1'-NH2
wherein R1' is as defined above;
wherein in one embodiment, step a is carried out in the presence of pyridine and triphenylphosphite at elevated temperatures, such as 50-100°C for 0.5 to 30 h, for example for 2 to 18 h;
optionally followed by functionalization steps or functional group adjustment steps a'.
The term "protecting group" as used herein relates to a group that protects a functional group which is present in the starting materials and is not intended to take part in the reaction. In additional process steps, carried out as desired, functional groups of the starting compounds which should not take part in the reaction may be present in unprotected form or may be protected for example by one or more protecting groups. The protecting groups are then wholly or partly removed according to one of the known methods. Protecting groups, and the manner in which they are introduced and removed are described, for example, in "Protective Groups in Organic Chemistry", Plenum Press, London, New York 1973, and in "Methoden der organischen Chemie", Houben-Weyl, 4th edition, Vol. 15/1 , Georg-Thieme-Verlag, Stuttgart 1974 and in Theodora W. Greene,
"Protective Groups in Organic Synthesis", John Wiley & Sons, New York 1981 . A characteristic of protecting groups is that they can be removed readily, i.e. without the occurrence of undesired secondary reactions, for example by solvolysis, reduction, photolysis or alternatively under physiological conditions.
Intermediates and final products can be worked up and/or purified according to standard methods, e.g. using chromatographic methods, distribution methods, (re-) crystallization, and the like.
The following applies in general to all processes mentioned herein before and hereinafter.
All the above-mentioned process steps can be carried out under reaction conditions that are known to those skilled in the art, including those mentioned specifically, in the absence or, customarily, in the presence of solvents or diluents, including, for example, solvents or diluents that are inert towards the reagents used and dissolve them, in the absence or presence of catalysts, condensation or neutralizing agents, for example ion exchangers, such as cation exchangers, e.g. in the H+ form, depending on the nature of the reaction and/or of the reactants at reduced, normal or elevated temperature, for example in a temperature range of from about -100 °C to about 190 °C, including, for example, from approximately -80 °C to approximately 150 °C, for example at from -80 to -60 °C, at room temperature, at from -20 to 40 °C or at reflux temperature, under atmospheric pressure or in a closed vessel, where appropriate under pressure, and/or in an inert atmosphere, for example under an argon or nitrogen atmosphere.
At all stages of the reactions, mixtures of isomers that are formed can be separated into the individual isomers, for example diastereoisomers or enantiomers, or into any desired mixtures of isomers, for example racemates or mixtures of diastereoisomers, for example analogously to the methods described herein above.
The solvents from which those solvents that are suitable for any particular reaction may be selected include those mentioned specifically or, for example, water, esters, such as CrCs-alkyl-C Cs-alkanoates, for example ethyl acetate, ethers, such as aliphatic ethers, for example diethyl ether, or cyclic ethers, for example tetrahydrofuran or dioxane, liquid aromatic hydrocarbons, such as benzene or toluene, alcohols, such as methanol, ethanol or 1 - or 2-propanol, nitriles, such as acetonitrile, halogenated hydrocarbons, such as methylene chloride or chloroform, acid amides, such as dimethylformamide or dimethyl acetamide, bases, such as heterocyclic nitrogen bases, for example pyridine or /V-methylpyrrolidin-2-one, carboxylic acid anhydrides, such as C Ce-alkanoic acid anhydrides, for example acetic anhydride, cyclic, linear or branched hydrocarbons, such as cyclohexane, hexane or isopentane, methycyclohexane, or mixtures of those
solvents, for example aqueous solutions, unless otherwise indicated in the description of the processes. Such solvent mixtures may also be used in working up, for example by chromatography or partitioning.
The compounds, including their salts, may also be obtained in the form of hydrates, or their crystals may, for example, include the solvent used for crystallization. Different crystalline forms may be present.
In another aspect, the present invention provides a intermediate of formula (C)
In another aspect, the present invention provides a intermediate of formula (D)
R1' is R1, as defined above for a compound of formula (I), or a substituent that can be transferred into R1 via functionalization steps or functional group adjustment steps;
R2' is R2, as defined above for a compound of formula (I), or a substituent that can be transferred into R2 via functionalization steps or functional group adjustment steps;
R5' is R5, as defined above for a compound of formula (I), or a substituent that can be transferred into R5 via functionalization steps or functional group adjustment steps;
R6' is R6, as defined above for a compound of formula (I), or a substituent that can be transferred into R6 via functionalization steps or functional group adjustment steps; and U is L, as defined above for a compound of formula (I), or a substituent that can be transferred into L via functionalization steps or functional group adjustment steps.
In another aspect, the present invention provides a intermediate of formula (C)
wherein PG represents a suitable protecting group, such as a Boc group, and the other substituents are as defined above.
The invention further includes any variant of the present processes, in which an
intermediate product obtainable at any stage thereof is used as starting material and the remaining steps are carried out, or in which the starting materials are formed in situ under the reaction conditions, or in which the reaction components are used in the form of their salts or optically pure material.
Compounds of the invention and intermediates can also be converted into each other according to methods generally known to those skilled in the art. In another aspect, the present invention provides a pharmaceutical composition comprising a compound of the present invention, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable carrier. In a further embodiment, the composition comprises at least two pharmaceutically acceptable carriers, such as those described herein. For purposes of the present invention, unless designated otherwise, solvates and hydrates are generally considered compositions. Preferably, pharmaceutically acceptable carriers are sterile. The pharmaceutical composition can be formulated for particular routes of administration such as oral administration, parenteral administration, and rectal
administration, etc. In addition, the pharmaceutical compositions of the present invention can be made up in a solid form (including without limitation capsules, tablets, pills, granules, powders or suppositories), or in a liquid form (including without limitation solutions, suspensions or emulsions). The pharmaceutical compositions can be subjected to conventional pharmaceutical operations such as sterilization and/or can contain conventional inert diluents, lubricating agents, or buffering agents, as well as adjuvants, such as preservatives, stabilizers, wetting agents, emulsifiers and buffers, etc.
Typically, the pharmaceutical compositions are tablets or gelatin capsules comprising the active ingredient together with one or more of:
a) diluents, e.g. , lactose, dextrose, sucrose, mannitol, sorbitol, cellulose and/or glycine; b) lubricants, e.g. , silica, talcum, stearic acid, its magnesium or calcium salt and/or polyethyleneglycol; for tablets also
c) binders, e.g. , magnesium aluminum silicate, starch paste, gelatin, tragacanth, methylcellulose, sodium carboxymethylcellulose and/or polyvinylpyrrolidone; if desired d) disintegrants, e.g. , starches, agar, alginic acid or its sodium salt, or effervescent mixtures; and
e) absorbents, colorants, flavors and sweeteners. Tablets may be either film coated or enteric coated according to methods known in the art.
Suitable compositions for oral administration include an effective amount of a compound of the invention in the form of tablets, lozenges, aqueous or oily suspensions, dispersible powders or granules, emulsion, hard or soft capsules, or syrups or elixirs. Compositions intended for oral use are prepared according to any method known in the art for the manufacture of pharmaceutical compositions and such compositions can contain one or more agents selected from the group consisting of sweetening agents, flavoring agents, coloring agents and preserving agents in order to provide pharmaceutically elegant and palatable preparations. Tablets may contain the active ingredient in admixture with nontoxic pharmaceutically acceptable excipients which are suitable for the manufacture of tablets. These excipients are, for example, inert diluents, such as calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate; granulating and disintegrating agents, for example, corn starch, or alginic acid; binding agents, for example, starch, gelatin or acacia; and lubricating agents, for example magnesium stearate, stearic acid or talc. The tablets are uncoated or coated by known techniques to delay disintegration and absorption in the gastrointestinal tract and thereby provide a sustained action over a longer period. For example, a time delay material such as glyceryl monostearate or glyceryl distearate can be employed. Formulations for oral use can be presented as hard gelatin capsules wherein the active ingredient is mixed with an inert solid diluent, for example, calcium carbonate, calcium phosphate or kaolin, or as soft gelatin capsules wherein the active ingredient is mixed with water or an oil medium, for example, peanut oil, liquid paraffin or olive oil.
Certain injectable compositions are aqueous isotonic solutions or suspensions, and suppositories are advantageously prepared from fatty emulsions or suspensions. Said compositions may be sterilized and/or contain adjuvants, such as preserving, stabilizing, wetting or emulsifying agents, solution promoters, salts for regulating the osmotic pressure and/or buffers. In addition, they may also contain other therapeutically valuable substances.
Said compositions are prepared according to conventional mixing, granulating or coating methods, respectively, and contain about 0.1 -75%, or contain about 1 -50%, of the active ingredient. Suitable compositions for transdermal application include an effective amount of a compound of the invention with a suitable carrier. Carriers suitable for transdermal delivery include absorbable pharmacologically acceptable solvents to assist passage through the skin of the host. For example, transdermal devices are in the form of a bandage comprising a backing member, a reservoir containing the compound optionally with carriers, optionally a rate controlling barrier to deliver the compound of the skin of the host at a controlled and predetermined rate over a prolonged period of time, and means to secure the device to the skin.
Suitable compositions for topical application, e.g. , to the skin and eyes, include aqueous solutions, suspensions, ointments, creams, gels or sprayable formulations, e.g. , for delivery by aerosol or the like. Such topical delivery systems will in particular be appropriate for dermal application, e.g. , for the treatment of skin cancer, e.g. , for prophylactic use in sun creams, lotions, sprays and the like. They are thus particularly suited for use in topical, including cosmetic, formulations well-known in the art. Such may contain solubilizers, stabilizers, tonicity enhancing agents, buffers and preservatives.
As used herein a topical application may also pertain to an inhalation or to an intranasal application. They may be conveniently delivered in the form of a dry powder (either alone, as a mixture, for example a dry blend with lactose, or a mixed component particle, for example with phospholipids) from a dry powder inhaler or an aerosol spray presentation from a pressurised container, pump, spray, atomizer or nebuliser, with or without the use of a suitable propellant.
The compounds of formula (I) in free form or in pharmaceutically acceptable salt form, exhibit valuable pharmacological properties, e.g. class I PI3 kinase modulating properties, e.g. as indicated in in vitro and in vivo tests as provided in the next sections, and are therefore indicated for therapy or for use as research chemicals, e.g. as tool compounds.
Compounds of the invention may be useful in the treatment of conditions, diseases or disorders including disease or infection associated immunopathology in which one or more of the functions of B cells such as antibody production, antigen presentation, cytokine production or lymphoid organogenesis are abnormal or are undesirable
including rheumatoid arthritis and related diseases (such as ankylosing spondylarthritis, psoriatic arthritis, juvenile arthritis), pemphigus vulgaris and related diseases, idiopathic thrombocytopenia purpura, systemic lupus erythematosus, multiple sclerosis, myasthenia gravis, Sjogren's syndrome , Graft versus host disease, autoimmune hemolytic anemia, ANCA-associated vasculitides, cryoglobulinemia, thrombotic thrombocytopenic purpura, chronic autoimmune urticaria, allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, different types of glomerulonephritides, AMR (antibody-mediated transplant rejection), B cell-mediated hyperacute, acute and chronic transplant rejection and cancers of haematopoietic origin including but not limited to multiple myeloma; acute myelogenous leukemia; chronic myelogenous leukemia; lymphocytic leukemia; myeloid leukemia; non-Hodgkin lymphoma; lymphomas; polycythemia vera; essential thrombocythemia; myelofibrosis with myeloid metaplasia; and Walden stroem disease. The invention includes methods of treating conditions, diseases or disorders in which one or more of the functions of neutrophils, such as superoxide release, stimulated exocytosis, or chemoatractic migration are abnormal or are undesirable including rheumatoid arthritis, pulmonary or resporatory disorders such as asthma, inflammatory dermatoses such as psoriasis, as well as in disease or infection associated
immunopathology and others.
The invention includes methods of treating conditions, diseases or disorders in which one or more of the functions of basophil and mast cells such as chemoatractic migration or allergen-lgE-mediated degranulation are abnormal or are undesirable including allergic diseases (atopic dermatitis, contact dermatitis, allergic rhinitis, chronic allergic urticaria) as well as other disorders such as COPD, asthma or emphysema.
The invention includes methods of treating conditions, diseases or disorders in which one or more of the functions of T cells such as cytokine production or cell-mediated cytotoxicity abnormal or are undesirable including rheumatoid arthritis, multiple sclerosis, acute or chronic rejection of cell tissue or organ grafts or cancers of haematopoietic origin as well as in disease or infection associated immunopathology.
Further, the invention includes methods of treating neurodegenerative diseases, cardiovascular diseases and platelet aggregation.
Further, the invention includes methods of treating skin diseases such as porphyria cutanea tarda, polymorphous light eruption, dermatomyositis, solar urticaria, oral lichen planus, panniculitis, scleroderma, urticarial vasculitis. Further, the invention includes methods of treating chronic inflammatory diseases such as sarcoidosis, granuloma annulare.
In other embodiments, the condition or disorder (e.g. class I PI3 kinase-mediated) is selected from the group consisting of: polycythemia vera, essential thrombocythemia, myelofibrosis with myeloid metaplasia, asthma, COPD, ARDS, Loffler's syndrome, eosinophilic pneumonia, parasitic (in particular metazoan) infestation (including tropical eosinophilia), bronchopulmonary aspergillosis, polyarteritis nodosa (including Churg- Strauss syndrome), eosinophilic granuloma, eosinophil-related disorders affecting the airways occasioned by drug-reaction, psoriasis, contact dermatitis, atopic dermatitis, alopecia areata, erythema multiforme, dermatitis herpetiformis, scleroderma, vitiligo, hypersensitivity angiitis, urticaria, bullous pemphigoid, lupus erythematosus, pemphigus, epidermolysis bullosa acquisita, autoimmune haematogical disorders (e.g. haemolytic anaemia, aplastic anaemia, pure red cell anaemia and idiopathic thrombocytopenia), systemic lupus erythematosus, polychondritis, scleroderma, Wegener granulomatosis, dermatomyositis, chronic active hepatitis, myasthenia gravis, Steven-Johnson syndrome, idiopathic sprue, autoimmune inflammatory bowel disease (e.g. ulcerative colitis and Crohn's disease), endocrine opthalmopathy, Grave's disease, sarcoidosis, alveolitis, chronic hypersensitivity pneumonitis, multiple sclerosis, primary biliary cirrhosis, uveitis (anterior and posterior), interstitial lung fibrosis, psoriatic arthritis, glomerulonephritis, cardiovascular diseases, atherosclerosis, hypertension, deep venous thrombosis, stroke, myocardial infarction, unstable angina, thromboembolism, pulmonary embolism, thrombolytic diseases, acute arterial ischemia, peripheral thrombotic occlusions, and coronary artery disease, reperfusion injuries, retinopathy, such as diabetic retinopathy or hyperbaric oxygen-induced retinopathy.
In another embodiment, the compounds of the present invention are useful in the treatment, prevention, or amelioration of autoimmune disease and of inflammatory conditions, in particular inflammatory conditions with an aetiology including an autoimmune component such as arthritis (for example rheumatoid arthritis, arthritis chronica progrediente and arthritis deformans) and rheumatic diseases, including inflammatory conditions and rheumatic diseases involving bone loss, inflammatory pain, spondyloarhropathies including ankolsing spondylitis, Reiter syndrome, reactive arthritis,
psoriatic arthritis, and enterophathics arthritis, hypersensitivity (including both airways hypersensitivity and dermal hypersensitivity) and allergies. Specific auto-immune diseases for which compounds of the invention may be employed include autoimmune haematological disorders (including e.g. hemolytic anaemia, aplastic anaemia, pure red cell anaemia and idiopa-thic thrombocytopenia), acquired hemophilia A, cold agglutinin disease, cryoglobulinemia, thrombotic thrombocytopenic purpura, Sjogren's syndrome, systemic lupus erythematosus, inflammatory muscle disorders, polychondritis,
sclerodoma, anti-neutrophil cytoplasmic antibody- associated vasculitis, IgM mediated neuropathy, opsoclonus myoclonus syndrome, Wegener granulomatosis,
dermatomyositis, chronic active hepatitis, myasthenia gravis, psoriasis, Steven-Johnson syndrome, pemphigus vulgaris, pemphigus foliacius, idio-pathic sprue, autoimmune inflammatory bowel disease (including e.g. ulcerative colitis, Crohn's disease and
Irritable Bowel Syndrome), endocrine ophthalmopathy, Graves' disease, sarcoidosis, multiple sclerosis, neuromyelitis optica, primary biliary cirrhosis, juvenile diabetes
(diabetes mellitus type I), uveitis (anterior, intermediate and posterior as well as panuveitis), keratoconjunctivitis sicca and vernal keratoconjunctivitis, interstitial lung fibrosis, psoriatic arthritis, ancylosing spondylitis and glomerulonephritis (with and without nephrotic syndrome, e.g. including idiopathic nephro-tic syndrome or minimal change nephropathy), tumors, inflammatory disease of skin and cornea, myositis, loosening of bone implants, metabolic disorders, such as atherosclerosis, diabetes, and dislipidemia.
In another embodiment, the compounds of the present invention are useful in the treatment of conditions or disorders selected from the group consisting of, primary cutaneous B-cell lymphoma, immunobullous disease, pemphigus vulgaris, pemphigus foliaceus, endemic form of Brazilian pemphigus (Fogo selvagem), paraneoplastic pemphigus, bullous pemphigoid, mucous membrane pemphigoid, epidermolysis bullosa acquisita, chronic graft versus host disease, dermatomyositis, systemic lupus
erythematosus, vasculitis, small vessel vasculitis, hypocomplementemic urticarial vasculitis, antineutrophil cytoplasmic antibody-vasculitis, cryoglobulinemia, Schnitzler syndrome, Waldenstrom's macroglobulinemia, angioedema, vitiligo, systemic lupus erythematosus, idiopathic thrombocytopenic purpura, multiple sclerosis, cold agglutinin disease, autoimmune hemolytic anemia, antineutrophil cytoplasmic antibody—
associated vasculitis, graft versus host disease, cryoglobulinemia and thrombotic thrombocytopenic.
Thus, as a further embodiment, the present invention provides the use of a compound of the formulae (I) or (l'), (la'), (lb'), (Ic'), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (Ic"),
(Id"), (le"), (IA"), (IB"), (IC"), (ID") or (I'"), (lb'"), (lc'"), (Id'"), (le'") in therapy. In a further embodiment, the therapy is selected from a disease which may be treated by inhibition of class I PI3 kinases. In another embodiment, the disease is selected from the aforementioned list, suitably from autoimmune disorders, autoinflammatory and inflammatory diseases, allergic diseases, airway diseases, such as asthma and COPD, transplant rejection; antibody production, antigen presentation, cytokine production or lymphoid organogenesis are abnormal or are undesirable including rheumatoid arthritis and related diseases (such as ankylosing spondylarthritis, psoriatic arthritis, juvenile arthritis), pemphigus vulgaris and related diseases, idiopathic thrombocytopenia purpura, systemic lupus erythematosus, multiple sclerosis, myasthenia gravis, Sjogren's syndrome, Graft versus host disease, autoimmune hemolytic anemia, ANCA-associated vasculitides, cryoglobulinemia, thrombotic thrombocytopenic purpura, chronic autoimmune urticaria, allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, different types of glomerulonephritides, AMR (antibody-mediated transplant rejection), B cell-mediated hyperacute, acute and chronic transplant rejection and cancers of haematopoietic origin including but not limited to multiple myeloma; a leukaemia; acute myelogenous leukemia; chronic myelogenous leukemia; lymphocytic leukemia; myeloid leukemia; non-Hodgkin lymphoma; lymphomas; polycythemia vera; essential thrombocythemia; myelofibrosis with myeloid metaplasia; and Walden stroem disease; more suitably from rheumatoid arthritis (RA), pemphigus vulgaris (PV), idiopathic thrombocytopenia purpura (ITP), thrombotic thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA-associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection and cancers of haematopoietic origin as well as in disease or infection associated immunopathology, for example in severe and cerebral malaria, trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis. Thus, as a further embodiment, the present invention provides a compound of the formulae (I) or (l'), (la'), (lb'), (IC), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (lc"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (I'"), (lb'"), (lc'"), (Id'"), (le'") for use in therapy. In a further embodiment, the therapy is selected from a disease which may be treated by inhibition of class I PI3kinases. In another embodiment, the disease is selected from the afore- mentioned list, suitably from autoimmune disorders, autoinflammatory and inflammatory diseases, allergic diseases, airway diseases, such as asthma and COPD, transplant rejection; antibody production, antigen presentation, cytokine production or lymphoid
organogenesis are abnormal or are undesirable including rheumatoid arthritis and related diseases (such as ankylosing spondylarthritis, psoriatic arthritis, juvenile arthritis), pemphigus vulgaris and related diseases, idiopathic thrombocytopenia purpura, systemic lupus erythematosus, multiple sclerosis, myasthenia gravis, Sjogren's syndrome, Graft versus host disease, autoimmune hemolytic anemia, ANCA-associated vasculitides, cryoglobulinemia, thrombotic thrombocytopenic purpura, chronic autoimmune urticaria, allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, different types of glomerulonephritides, AMR (antibody-mediated transplant rejection), B cell-mediated hyperacute, acute and chronic transplant rejection and cancers of haematopoietic origin including but not limited to multiple myeloma; a leukaemia; acute myelogenous leukemia; chronic myelogenous leukemia; lymphocytic leukemia; myeloid leukemia; non-Hodgkin lymphoma; lymphomas; polycythemia vera; essential thrombocythemia; myelofibrosis with myeloid metaplasia; and Walden stroem disease; more suitably from rheumatoid arthritis (RA), pemphigus vulgaris (PV), idiopathic thrombocytopenia purpura (ITP), thrombotic thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA-associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection and cancers of haematopoietic origin as well as in disease or infection associated immunopathology, for example in severe and cerebral malaria, trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis.
In another embodiment, the invention provides a method of treating a disease which is treated by inhibition of class I PI3 kinases comprising administration of a therapeutically acceptable amount of a compound of the formulae (I) or (Γ), (la'), (lb'), (lc'), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (lc"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (I'"), (lb'"), (lc'"), (Id'"), (le'"). In a further embodiment, the disease is selected from the afore-mentioned list, suitably from autoimmune disorders, autoinflammatory and inflammatory diseases, allergic diseases, airway diseases, such as asthma and COPD, transplant rejection; antibody production, antigen presentation, cytokine production or lymphoid organogenesis are abnormal or are undesirable including rheumatoid arthritis and related diseases (such as ankylosing spondylarthritis, psoriatic arthritis, juvenile arthritis), pemphigus vulgaris and related diseases, idiopathic thrombocytopenia purpura, systemic lupus erythematosus, multiple sclerosis, myasthenia gravis, Sjogren's syndrome, Graft versus host disease, autoimmune hemolytic anemia, ANCA-associated vasculitides, cryoglobulinemia, thrombotic thrombocytopenic purpura, chronic autoimmune urticaria, allergy (atopic dermatitis, contact
dermatitis, allergic rhinitis), goodpasture's syndrome, different types of glomerulonephritides, AMR (antibody-mediated transplant rejection), B cell-mediated hyperacute, acute and chronic transplant rejection and cancers of haematopoietic origin including but not limited to multiple myeloma; a leukaemia; acute myelogenous leukemia; chronic myelogenous leukemia; lymphocytic leukemia; myeloid leukemia; non-Hodgkin lymphoma; lymphomas; polycythemia vera; essential thrombocythemia; myelofibrosis with myeloid metaplasia; and Walden stroem disease; more suitably from rheumatoid arthritis (RA), pemphigus vulgaris (PV), idiopathic thrombocytopenia purpura (ITP), thrombotic thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA-associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection and cancers of haematopoietic origin as well as in disease or infection associated immunopathology, for example in severe and cerebral malaria, trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis.
Thus, as a further embodiment, the present invention provides the use of a compound of the formulae (I) or (l'), (la'), (lb'), (lc'), (Id'), (le'), (ΙΑ'), (ΙΒ'), (IC), (ID') or (I"), (la"), (lb"), (lc"), (Id"), (le"), (IA"), (IB"), (IC"), (ID") or (I'"), (lb"'), (lc"'), (Id"'), (le'") for the manufacture of a medicament. In a further embodiment, the medicament is for treatment of a disease which may be treated inhibition of class I PI3 kinases. In another embodiment, the disease is selected from the afore-mentioned list, suitably from autoimmune disorders,
autoinflammatory and inflammatory diseases, allergic diseases, airway diseases, such as asthma and COPD, transplant rejection; antibody production, antigen presentation, cytokine production or lymphoid organogenesis are abnormal or are undesirable including rheumatoid arthritis arthritis and related diseases (such as ankylosing spondylarthritis, psoriatic arthritis, juvenile arthritis), pemphigus vulgaris and related diseases, idiopathic thrombocytopenia purpura, systemic lupus erythematosus, multiple sclerosis, myasthenia gravis, Sjogren's syndrome, Graft versus host disease, autoimmune hemolytic anemia, ANCA-associated vasculitides, cryoglobulinemia, thrombotic thrombocytopenic purpura, chronic autoimmune urticaria, allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, different types of glomerulonephritides, AMR (antibody-mediated transplant rejection), B cell-mediated hyperacute, acute and chronic transplant rejection and cancers of haematopoietic origin including but not limited to multiple myeloma; a leukaemia; acute myelogenous leukemia; chronic myelogenous leukemia; lymphocytic leukemia; myeloid leukemia; non-Hodgkin lymphoma; lymphomas; polycythemia vera; essential
thrombocythemia; myelofibrosis with myeloid metaplasia; and Walden stroem disease; more
suitably from rheumatoid arthritis (RA), pemphigus vulgaris (PV), idiopathic thrombocytopenia purpura (ITP), thrombotic thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA- associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection and cancers of haematopoietic origin as well as in disease or infection associated immunopathology, for example in severe and cerebral malaria, trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis.
The pharmaceutical composition or combination of the present invention can be in unit dosage of about 1 -1000 mg of active ingredient(s) for a subject of about 50-70 kg, or about 1 -500 mg or about 1 -250 mg or about 1 -150 mg or about 0.5-100 mg of active ingredients. The therapeutically effective dosage of a compound, the pharmaceutical composition, or the combinations thereof, is dependent on the species of the subject, the body weight, age and individual condition, the disorder or disease or the severity thereof being treated. A physician, clinician or veterinarian of ordinary skill can readily
determine the effective amount of each of the active ingredients necessary to prevent, treat or inhibit the progress of the disorder or disease.
The above-cited dosage properties are demonstrable in vitro and in vivo tests using advantageously mammals, e.g., mice, rats, dogs, monkeys or isolated organs, tissues and preparations thereof. The compounds of the present invention can be applied in vitro in the form of solutions, e.g., aqueous solutions, and in vivo either enterally, parenterally, advantageously intravenously, e.g. , as a suspension or in aqueous solution. The dosage in vitro may range between about 10 3 molar and 10~9 molar concentrations. A therapeutically effective amount in vivo may range depending on the route of
administration, between about 0.1 -500 mg/kg, or between about 1 -100 mg/kg. The compound of the present invention may be administered either simultaneously with, or before or after, one or more other therapeutic agent. The compound of the present invention may be administered separately, by the same or different route of administration, or together in the same pharmaceutical composition as the other agents. A therapeutic agent is, for example, a chemical compound, peptide, antibody, antibody fragment or nucleic acid, which is therapeutically active or enhances the therapeutic activity when administered to a patient in combination with a compound of the invention.
In one embodiment, the invention provides a product comprising a compound of formula (I) and at least one other therapeutic agent as a combined preparation for simultaneous, separate or sequential use in therapy. In one embodiment, the therapy is the treatment of a disease or condition mediated by the activity of the class I PI3 kinases. Products provided as a combined preparation include a composition comprising the compound of formula (I) and the other therapeutic agent(s) together in the same pharmaceutical composition, or the compound of formula (I) and the other therapeutic agent(s) in separate form, e.g. in the form of a kit. In one embodiment, the invention provides a pharmaceutical composition comprising a compound of formula (I) and another therapeutic agent(s). Optionally, the pharmaceutical composition may comprise a pharmaceutically acceptable carrier, as described above.
In one embodiment, the invention provides a kit comprising two or more separate pharmaceutical compositions, at least one of which contains a compound of formula (I). In one embodiment, the kit comprises means for separately retaining said compositions, such as a container, divided bottle, or divided foil packet. An example of such a kit is a blister pack, as typically used for the packaging of tablets, capsules and the like. The kit of the invention may be used for administering different dosage forms, for example, oral and parenteral, for administering the separate compositions at different dosage intervals, or for titrating the separate compositions against one another. To assist compliance, the kit of the invention typically comprises directions for administration. In the combination therapies of the invention, the compound of the invention and the other therapeutic agent may be manufactured and/or formulated by the same or different manufacturers. Moreover, the compound of the invention and the other therapeutic may be brought together into a combination therapy: (i) prior to release of the combination product to physicians (e.g. in the case of a kit comprising the compound of the invention and the other therapeutic agent); (ii) by the physician themselves (or under the guidance of the physician) shortly before administration; (iii) in the patient themselves, e.g. during sequential administration of the compound of the invention and the other therapeutic agent.
Accordingly, the invention provides the use of a compound of formula (I) for treating a disease or condition mediated by the activity of the class I PI3kinases, wherein the medicament is prepared for administration with another therapeutic agent. The invention also provides the use of another therapeutic agent for treating a disease or condition mediated by
the activity of the class I PI3kinases, wherein the medicament is administered with a compound of formula (I).
The invention also provides a compound of formula (I) for use in a method of treating a disease or condition mediated by the activity of the class I PI3 kinases enzymes, wherein the compound of formula (I) is prepared for administration with another therapeutic agent. The invention also provides another therapeutic agent for use in a method of treating a disease or condition mediated by the activity of the class I PI3 kinases, wherein the other therapeutic agent is prepared for administration with a compound of formula (I). The invention also provides a compound of formula (I) for use in a method of treating a disease or condition mediated by the activity of the class I PI3 kinases wherein the compound of formula (I) is administered with another therapeutic agent. The invention also provides another therapeutic agent for use in a method of treating a disease or condition mediated by the activity of class I PI3 kinases wherein the other therapeutic agent is administered with a compound of formula (I).
The invention also provides the use of a compound of formula (I) for treating a disease or condition mediated by the activity of the class I PI3 kinases, wherein the patient has previously (e.g. within 24 hours) been treated with another therapeutic agent. The invention also provides the use of another therapeutic agent for treating a disease or condition mediated by the activity of the class I PI3 kinases, wherein the patient has previously (e.g. within 24 hours) been treated with a compound of formula (I).
The compounds of formula (I) may be administered as the sole active ingredient or in conjunction with, e.g. as an adjuvant to, other drugs e.g. immunosuppressive or
immunomodulating agents or other anti-inflammatory agents, e.g. for the treatment or prevention of alio- or xenograft acute or chronic rejection or inflammatory or autoimmune disorders, or a chemotherapeutic agent, e.g a malignant cell anti-proliferative agent. For example, the compounds of formula (I) may be used in combination with a calcineurin inhibitor, e.g. cyclosporin A or FK 506; a mTOR inhibitor, e.g. rapamycin, 40-O-(2- hydroxyethyl)-rapamycin, CCI779, ABT578, AP23573, TAFA-93, biolimus-7 or biolimus- 9; an ascomycin having immuno-suppressive properties, e.g. ABT-281 , ASM981 , etc.;
corticosteroids; cyclophosphamide; azathioprene; methotrexate; leflunomide; mizoribine; mycophenolic acid or salt; mycophenolate mofetil; 15-deoxyspergualine or an
immunosuppressive homologue, analogue or derivative thereof; a PKC inhibitor, e.g. as disclosed in WO 02/38561 or WO 03/82859, e.g. the compound of Example 56 or 70; a JAK3 kinase inhibitor, e.g. N-benzyl-3,4-dihydroxy-benzylidene-cyanoacetamide a-
cyano-(3,4-dihydroxy)-]N-benzylcinnamamide (Tyrphostin AG 490), prodigiosin 25-C (PNU156804), [4-(4'-hydroxyphenyl)-amino-6,7-dimethoxyquinazoline] (WHI-P131), [4- (3'-bromo-4'-hydroxylphenyl)-amino-6,7-dimethoxyquinazoline] (WHI-P154), [4-(3',5'- dibromo-4'-hydroxylphenyl)-amino-6,7-dimethoxyquinazoline] WHI-P97, KRX-21 1 , 3- {(3R,4R)-4-methyl-3-[methyl-(7H-pyrrolo[2,3-d]pyrimidin-4^^
oxo-propionitrile, in free form or in a pharmaceutically acceptable salt form, e.g. mono- citrate (also called CP-690,550), or a compound as disclosed in WO 04/052359 or WO 05/066156; immunosuppressive monoclonal antibodies, e.g., monoclonal antibodies to leukocyte receptors, e.g., MHC, CD2, CD3, CD4, CD7, CD8, CD25, CD28, CD40, CD45, CD52, CD58, CD80, CD86 or their ligands; other immunomodulatory compounds, e.g. a recombinant binding molecule having at least a portion of the extracellular domain of CTLA4 or a mutant thereof, e.g. an at least extracellular portion of CTLA4 or a mutant thereof joined to a non-CTLA4 protein sequence, e.g. CTLA4lg (for ex. designated ATCC 68629) or a mutant thereof, e.g. LEA29Y; adhesion molecule inhibitors, e.g. LFA- 1 antagonists, ICAM-1 or -3 antagonists, VCAM-4 antagonists or VLA-4 antagonists; or antihistamines; or antitussives, or a bronchodilatory agent; or an angiotensin receptor blockers; or an anti-infectious agent.
Where the compounds of formula (I) are administered in conjunction with other immunosuppressive / immunomodulatory, anti-inflammatory, chemotherapeutic or anti- infectious therapy, dosages of the co-administered immunosuppressant,
immunomodulatory, anti-inflammatory, chemotherapeutic or anti-infectious compound will of course vary depending on the type of co-drug employed, e.g. whether it is a steroid or a calcineurin inhibitor, on the specific drug employed, on the condition being treated and so forth.
A compound of the formula (I) may also be used to advantage in combination with each other or in combination with other therapeutic agents, especially other antiproliferative agents. Such antiproliferative agents include, but are not limited to, aromatase inhibitors; antiestrogens; topoisomerase I inhibitors; topoisomerase II inhibitors;
microtubule active agents; alkylating agents; histone deacetylase inhibitors; compounds, which induce cell differentiation processes; cyclooxygenase inhibitors; MMP inhibitors; mTOR inhibitors; antineoplastic antimetabolites; platin compounds; compounds targeting/decreasing a protein or lipid kinase activity and further anti-angiogenic compounds; compounds which target, decrease or inhibit the activity of a protein or lipid phosphatase; gonadorelin agonists; anti-androgens; methionine aminopeptidase inhibitors; bisphosphonates; biological response modifiers; antiproliferative antibodies; heparanase inhibitors; inhibitors of Ras oncogenic isoforms; telomerase inhibitors;
proteasome inhibitors; agents used in the treatment of hematologic malignancies;
compounds which target, decrease or inhibit the activity of Flt-3; Hsp90 inhibitors;
temozolomide (TEMODAL®); and leucovorin.
The term "aromatase inhibitor", as used herein, relates to a compound which inhibits the estrogen production, i.e., the conversion of the substrates androstenedione and testosterone to estrone and estradiol, respectively. The term includes, but is not limited to, steroids, especially atamestane, exemestane and formestane; and, in particular, nonsteroids, especially aminoglutethimide, roglethimide, pyridoglutethimide, trilostane, testolactone, ketoconazole, vorozole, fadrozole, anastrozole and letrozole. Exemestane can be administered, e.g., in the form as it is marketed, e.g., under the trademark
AROMASIN. Formestane can be administered, e.g., in the form as it is marketed, e.g., under the trademark LENTARON. Fadrozole can be administered, e.g., in the form as it is marketed, e.g., under the trademark AFEMA. Anastrozole can be administered, e.g., in the form as it is marketed, e.g., under the trademark ARI Ml DEX. Letrozole can be administered, e.g., in the form as it is marketed, e.g., under the trademark FEMARA or FEMAR. Aminoglutethimide can be administered, e.g., in the form as it is marketed, e.g., under the trademark ORIMETEN. A combination of the invention comprising a chemotherapeutic agent which is an aromatase inhibitor is particularly useful for the treatment of hormone receptor positive tumors, e.g., breast tumors.
The term "anti-estrogen", as used herein, relates to a compound which antagonizes the effect of estrogens at the estrogen receptor level. The term includes, but is not limited to, tamoxifen, fulvestrant, raloxifene and raloxifene hydrochloride. Tamoxifen can be administered, e.g., in the form as it is marketed, e.g., under the trademark NOLVADEX. Raloxifene hydrochloride can be administered, e.g., in the form as it is marketed, e.g., under the trademark EVISTA. Fulvestrant can be formulated as disclosed in U.S. Patent No. 4,659,516 or it can be administered, e.g., in the form as it is marketed, e.g., under the trademark FASLODEX. A combination of the invention comprising a
chemotherapeutic agent which is an antiestrogen is particularly useful for the treatment of estrogen receptor positive tumors, e.g., breast tumors.
The term "anti-androgen", as used herein, relates to any substance which is capable of inhibiting the biological effects of androgenic hormones and includes, but is not limited to, bicalutamide (CASODEX), which can be formulated, e.g., as disclosed in U.S. Patent No. 4,636,505.
The term "gonadorelin agonist", as used herein, includes, but is not limited to, abarelix, goserelin and goserelin acetate. Goserelin is disclosed in U.S. Patent No. 4,100,274 and can be administered, e.g., in the form as it is marketed, e.g., under the trademark ZOLADEX. Abarelix can be formulated, e.g., as disclosed in U.S. Patent No. 5,843,901 .
The term "topoisomerase I inhibitor", as used herein, includes, but is not limited to, topotecan, gimatecan, irinotecan, camptothecian and its analogues, 9-nitrocamptothecin and the macromolecular camptothecin conjugate PNU-166148 (compound A1 in WO 99/17804). Irinotecan can be administered, e.g., in the form as it is marketed, e.g., under the trademark CAMPTOSAR. Topotecan can be administered, e.g., in the form as it is marketed, e.g., under the trademark HYCAMTIN.
The term "topoisomerase II inhibitor", as used herein, includes, but is not limited to, the anthracyclines, such as doxorubicin, including liposomal formulation, e.g., CAELYX; daunorubicin; epirubicin; idarubicin; nemorubicin; the anthraquinones mitoxantrone and losoxantrone; and the podophillotoxines etoposide and teniposide. Etoposide can be administered, e.g., in the form as it is marketed, e.g., under the trademark
ETOPOPHOS. Teniposide can be administered, e.g., in the form as it is marketed, e.g., under the trademark VM 26-BRISTOL. Doxorubicin can be administered, e.g., in the form as it is marketed, e.g., under the trademark ADRIBLASTIN or ADRIAMYCIN.
Epirubicin can be administered, e.g., in the form as it is marketed, e.g., under the trademark FARMORUBICIN. Idarubicin can be administered, e.g., in the form as it is marketed, e.g., under the trademark ZAVEDOS. Mitoxantrone can be administered, e.g., in the form as it is marketed, e.g., under the trademark NOVANTRON.
The term "microtubule active agent" relates to microtubule stabilizing, microtubule destabilizing agents and microtublin polymerization inhibitors including, but not limited to, taxanes, e.g., paclitaxel and docetaxel; vinca alkaloids, e.g., vinblastine, especially vinblastine sulfate; vincristine, especially vincristine sulfate and vinorelbine;
discodermolides; cochicine; and epothilones and derivatives thereof, e.g., epothilone B or D or derivatives thereof. Paclitaxel may be administered, e.g., in the form as it is marketed, e.g., TAXOL. Docetaxel can be administered, e.g., in the form as it is marketed, e.g., under the trademark TAXOTERE. Vinblastine sulfate can be
administered, e.g., in the form as it is marketed, e.g., under the trademark VINBLASTIN R.P. Vincristine sulfate can be administered, e.g., in the form as it is marketed, e.g., under the trademark FARMISTIN. Discodermolide can be obtained, e.g., as disclosed in U.S. Patent No. 5,010,099. Also included are epothilone derivatives which are disclosed in WO 98/10121 , U.S. Patent No. 6,194,181 , WO 98/25929, WO 98/08849,
WO 99/43653, WO 98/22461 and WO 00/31247. Especially preferred are epothilone A and/or B.
The term "alkylating agent", as used herein, includes, but is not limited to,
cyclophosphamide, ifosfamide, melphalan or nitrosourea (BCNU or Gliadel).
Cyclophosphamide can be administered, e.g., in the form as it is marketed, e.g., under
the trademark CYCLOSTIN. Ifosfamide can be administered, e.g., in the form as it is marketed, e.g., under the trademark HOLOXAN.
The term "histone deacetylase inhibitors" or "HDAC inhibitors" relates to compounds which inhibit the histone deacetylase and which possess antiproliferative activity. This includes compounds disclosed in WO 02/22577, especially /V-hydroxy-3-[4-[[(2- hydroxyethyl)[2-(1 /-/-indol-3-yl)ethyl]-amino]methyl]phenyl]-2£-2-propenamide, N- hydroxy-3-[4-[[[2-(2-methyl-1 /-/-indol-3-yl)-ethyl]-amino]methyl]phenyl]-2£-2-propenamide and pharmaceutically acceptable salts thereof. It further especially includes
suberoylanilide hydroxamic acid (SAHA).
The term "antineoplastic antimetabolite" includes, but is not limited to, 5-fluorouracil or 5- FU; capecitabine; gemcitabine; DNA demethylating agents, such as 5-azacytidine and decitabine; methotrexate and edatrexate; and folic acid antagonists, such as
pemetrexed. Capecitabine can be administered, e.g., in the form as it is marketed, e.g., under the trademark XELODA. Gemcitabine can be administered, e.g., in the form as it is marketed, e.g., under the trademark GEMZAR. Also included is the monoclonal antibody trastuzumab which can be administered, e.g., in the form as it is marketed, e.g., under the trademark HERCEPTIN.
The term "platin compound", as used herein, includes, but is not limited to, carboplatin, c/'s-platin, cisplatinum and oxaliplatin. Carboplatin can be administered, e.g., in the form as it is marketed, e.g., under the trademark CARBOPLAT. Oxaliplatin can be administered, e.g., in the form as it is marketed, e.g., under the trademark ELOXATIN. The term "compounds targeting/decreasing a protein or lipid kinase activity; or a protein or lipid phosphatase activity; or further anti-angiogenic compounds", as used herein, includes, but is not limited to, protein tyrosine kinase and/or serine and/or threonine kinase inhibitors or lipid kinase inhibitors, e.g.,
a) compounds targeting, decreasing or inhibiting the activity of the platelet- derived growth factor-receptors (PDGFR), such as compounds which target, decrease or inhibit the activity of PDGFR, especially compounds which inhibit the PDGF receptor, e.g., a /V-phenyl-2-pyrimidine-amine derivative, e.g., imatinib, SU101 , SU6668 and GFB-1 1 1 ;
b) compounds targeting, decreasing or inhibiting the activity of the fibroblast growth factor-receptors (FGFR);
c) compounds targeting, decreasing or inhibiting the activity of the insulin-like growth factor receptor I (IGF-IR), such as compounds which target, decrease or inhibit the activity of IGF-IR, especially compounds which inhibit the IGF-IR receptor, such as those compounds disclosed in WO 02/092599;
d) compounds targeting, decreasing or inhibiting the activity of the Trk receptor tyrosine kinase family;
e) compounds targeting, decreasing or inhibiting the activity of the Axl receptor tyrosine kinase family;
f) compounds targeting, decreasing or inhibiting the activity of the c-Met receptor;
g) compounds targeting, decreasing or inhibiting the activity of the Kit/SCFR receptor tyrosine kinase;
h) compounds targeting, decreasing or inhibiting the activity of the C-kit receptor tyrosine kinases - (part of the PDGFR family), such as compounds which target, decrease or inhibit the activity of the c-Kit receptor tyrosine kinase family, especially compounds which inhibit the c-Kit receptor, e.g., imatinib;
i) compounds targeting, decreasing or inhibiting the activity of members of the c- Abl family and their gene-fusion products, e.g., BCR-Abl kinase, such as compounds which target decrease or inhibit the activity of c-Abl family members and their gene fusion products, e.g., a /V-phenyl-2-pyrimidine-amine derivative, e.g., imatinib, PD180970, AG957, NSC 680410 or PD173955 from ParkeDavis; j) compounds targeting, decreasing or inhibiting the activity of members of the protein kinase C (PKC) and Raf family of serine/threonine kinases, members of the MEK, SRC, JAK, FAK, PDK and Ras/MAPK family members, or Pl(3) kinase family, or of the Pl(3)-kinase-related kinase family, and/or members of the cyclin- dependent kinase family (CDK) and are especially those staurosporine derivatives disclosed in U.S. Patent No. 5,093,330, e.g., midostaurin; examples of further compounds include, e.g., UCN-01 ; safingol; BAY 43-9006; Bryostatin 1 ; Perifosine; llmofosine; RO 318220 and RO 320432; GO 6976; Isis 3521 ;
LY333531 /LY379196; isochinoline compounds, such as those disclosed in WO 00/09495; FTIs; PD184352; or QAN697 (a P13K inhibitor);
k) compounds targeting, decreasing or inhibiting the activity of protein-tyrosine kinase inhibitors, such as compounds which target, decrease or inhibit the activity of protein-tyrosine kinase inhibitors include imatinib mesylate (GLEEVEC) or tyrphostin. A tyrphostin is preferably a low molecular weight (Mr < 1500) compound, or a pharmaceutically acceptable salt thereof, especially a compound selected from the benzylidenemalonitrile class or the S-arylbenzenemalonirile or bisubstrate quinoline class of compounds, more especially any compound selected from the group consisting of Tyrphostin A23/RG-50810, AG 99,
Tyrphostin AG 213, Tyrphostin AG 1748, Tyrphostin AG 490, Tyrphostin B44, Tyrphostin B44 (+) enantiomer, Tyrphostin AG 555, AG 494, Tyrphostin AG 556, AG957 and adaphostin (4-{[(2,5-dihydroxyphenyl)methyl]amino}-benzoic acid adamantyl ester, NSC 680410, adaphostin; and
I) compounds targeting, decreasing or inhibiting the activity of the epidermal growth factor family of receptor tyrosine kinases (EGFR, ErbB2, ErbB3, ErbB4 as homo- or hetero-dimers), such as compounds which target, decrease or inhibit the activity of the epidermal growth factor receptor family are especially compounds, proteins or antibodies which inhibit members of the EGF receptor tyrosine kinase family, e.g., EGF receptor, ErbB2, ErbB3 and ErbB4 or bind to EGF or EGF related ligands, and are in particular those compounds, proteins or monoclonal antibodies generically and specifically disclosed in WO 97/02266, e.g., the compound of Example 39, or in EP 0 564 409; WO 99/03854; EP 0520722; EP 0 566 226; EP 0 787 722; EP 0 837 063; U.S. Patent No.
5,747,498; WO 98/10767; WO 97/30034; WO 97/49688; WO 97/38983 and, especially, WO 96/30347, e.g., compound known as CP 358774; WO 96/33980, e.g., compound ZD 1839; and WO 95/03283, e.g., compound ZM105180, e.g., trastuzumab (HERCEPTIN), cetuximab, Iressa, Tarceva, OSI-774, CI-1033, EKB-569, GW-2016, E1 .1 , E2.4, E2.5, E6.2, E6.4, E2.1 1 , E6.3 or E7.6.3; and 7H-pyrrolo-[2,3-cf]pyrimidine derivatives which are disclosed in WO 03/013541 . Further anti-angiogenic compounds include compounds having another mechanism for their activity, e.g., unrelated to protein or lipid kinase inhibition, e.g., thalidomide (THALOMID) and TNP-470.
Compounds which target, decrease or inhibit the activity of a protein or lipid phosphatase are, e.g., inhibitors of phosphatase 1 , phosphatase 2A, PTEN or CDC25, e.g., okadaic acid or a derivative thereof.
Compounds which induce cell differentiation processes are e.g. retinoic acid, α- γ- or δ-tocopherol or a- γ- or δ-tocotrienol.
The term cyclooxygenase inhibitor, as used herein, includes, but is not limited to, e.g., Cox-2 inhibitors, 5-alkyl substituted 2-arylaminophenylacetic acid and derivatives, such as celecoxib (CELEBREX), rofecoxib (VIOXX), etoricoxib, valdecoxib or a 5-alkyl-2- arylaminophenylacetic acid, e.g., 5-methyl-2-(2'-chloro-6'-fluoroanilino)phenyl acetic acid or lumiracoxib.
The term "bisphosphonates", as used herein, includes, but is not limited to, etridonic, clodronic, tiludronic, pamidronic, alendronic, ibandronic, risedronic and zoledronic acid. "Etridonic acid" can be administered, e.g., in the form as it is marketed, e.g., under the trademark DIDRONEL. "Clodronic acid" can be administered, e.g., in the form as it is
marketed, e.g., under the trademark BONEFOS. "Tiludronic acid" can be administered, e.g., in the form as it is marketed, e.g., under the trademark SKELID. "Pamidronic acid" can be administered, e.g., in the form as it is marketed, e.g., under the trademark AREDIA™. "Alendronic acid" can be administered, e.g., in the form as it is marketed, e.g., under the trademark FOSAMAX. "Ibandronic acid" can be administered, e.g., in the form as it is marketed, e.g., under the trademark BONDRANAT. "Risedronic acid" can be administered, e.g., in the form as it is marketed, e.g., under the trademark ACTONEL. "Zoledronic acid" can be administered, e.g., in the form as it is marketed, e.g., under the trademark ZOMETA.
The term "mTOR inhibitors" relates to compounds which inhibit the mammalian target of rapamycin (mTOR) and which possess antiproliferative activity, such as sirolimus (Rapamune®), everolimus (Certican™), CCI-779 and ABT578.
The term "heparanase inhibitor", as used herein, refers to compounds which target, decrease or inhibit heparin sulphate degradation. The term includes, but is not limited to, PI-88.
The term "biological response modifier", as used herein, refers to a lymphokine or interferons, e.g., interferon γ.
The term "inhibitor of Ras oncogenic isoforms", e.g., H-Ras, K-Ras or N-Ras, as used herein, refers to compounds which target, decrease or inhibit the oncogenic activity of Ras, e.g., a "farnesyl transferase inhibitor", e.g., L-744832, DK8G557 or R1 15777 (Zarnestra).
The term "telomerase inhibitor", as used herein, refers to compounds which target, decrease or inhibit the activity of telomerase. Compounds which target, decrease or inhibit the activity of telomerase are especially compounds which inhibit the telomerase receptor, e.g., telomestatin.
The term "methionine aminopeptidase inhibitor", as used herein, refers to compounds which target, decrease or inhibit the activity of methionine aminopeptidase. Compounds which target, decrease or inhibit the activity of methionine aminopeptidase are, e.g., bengamide or a derivative thereof.
The term "proteasome inhibitor", as used herein, refers to compounds which target, decrease or inhibit the activity of the proteasome. Compounds which target, decrease or inhibit the activity of the proteasome include, e.g., PS-341 and MLN 341 .
The term "matrix metalloproteinase inhibitor" or "MMP inhibitor", as used herein, includes, but is not limited to, collagen peptidomimetic and nonpeptidomimetic inhibitors, tetracycline derivatives, e.g., hydroxamate peptidomimetic inhibitor batimastat and its orally bioavailable analogue marimastat (BB-2516), prinomastat (AG3340), metastat (NSC 683551) BMS-279251 , BAY 12-9566, TAA21 1 , MMI270B or AAJ996.
The term "agents used in the treatment of hematologic malignancies", as used herein, includes, but is not limited to, FMS-like tyrosine kinase inhibitors, e.g., compounds targeting, decreasing or inhibiting the activity of FMS-like tyrosine kinase receptors (Flt- 3R); interferon, 1 -b-D-arabinofuransylcytosine (ara-c) and bisulfan; and ALK inhibitors, e.g., compounds which target, decrease or inhibit anaplastic lymphoma kinase.
Compounds which target, decrease or inhibit the activity of FMS-like tyrosine kinase receptors (Flt-3R) are especially compounds, proteins or antibodies which inhibit members of the Flt-3R receptor kinase family, e.g., PKC412, midostaurin, a
staurosporine derivative, SU1 1248 and MLN518.
The term "HSP90 inhibitors", as used herein, includes, but is not limited to, compounds targeting, decreasing or inhibiting the intrinsic ATPase activity of HSP90; degrading, targeting, decreasing or inhibiting the HSP90 client proteins via the ubiquitin proteasome pathway. Compounds targeting, decreasing or inhibiting the intrinsic ATPase activity of HSP90 are especially compounds, proteins or antibodies which inhibit the ATPase activity of HSP90, e.g., 17-allylamino,17-demethoxygeldanamycin (17AAG), a geldanamycin derivative, other geldanamycin related compounds, radicicol and HDAC inhibitors.
The term "antiproliferative antibodies", as used herein, includes, but is not limited to, trastuzumab (Herceptin™), Trastuzumab-DM1 , erlotinib (Tarceva™), bevacizumab (Avastin™), rituximab (Rituxan®), PR064553 (anti-CD40) and 2C4 antibody. By antibodies is meant, e.g., intact monoclonal antibodies, polyclonal antibodies, multispecific antibodies formed from at least two intact antibodies, and antibodies fragments so long as they exhibit the desired biological activity.
For the treatment of acute myeloid leukemia (AML), compounds of formula (I) can be used in combination with standard leukemia therapies, especially in combination with therapies used for the treatment of AML. In particular, compounds of formula (I) can be administered in combination with, e.g., farnesyl transferase inhibitors and/or other drugs useful for the treatment of AML, such as Daunorubicin, Adriamycin, Ara-C, VP-16, Teniposide, Mitoxantrone, Idarubicin, Carboplatinum and PKC412. A compound of the formula (I) may also be used to advantage in combination with each other or in combination with other therapeutic agents, especially other anti-malarial agents. Such anti-malarial agents include, but are not limited to proguanil,
chlorproguanil, trimethoprim, chloroquine, mefloquine, lumefantrine, atovaquone, pyrimethamine-sulfadoxine, pyrimethamine-dapsone, halofantrine, quinine, quinidine, amodiaquine, amopyroquine, sulphonamides, artemisinin, arteflene, artemether, artesunate, primaquine, inhaled NO, L-arginine, Dipropylenetri-amine NONOate (NO
donor), Rosiglitzone (PPARy agonist), activated charcoal, Erythropoietin, Levamisole, and pyronaridine.
A compound of the formula (I) may also be used to advantage in combination with each other or in combination with other therapeutic agents, such as used for the treatment of Leishmaniosis, Trypanosomiasis, Toxoplasmosis and Neurocysticercosis. Such agents include, but are not limited to chloroquine sulfate, atovaquone-proguanil, artemether- lumefantrine, quinine-sulfate, artesunate, quinine, doxycycline, clindamycin, meglumine antimoniate, sodium stibogluconate, miltefosine, ketoconazole, pentamidine, amphotericin B (AmB), liposomal-AmB, paromomycine, eflornithine, nifurtimox, suramin, melarsoprol, prednisolone, benznidazole, sulfadiazine, pyrimethamine, clindamycin, trimetropim, sulfamethoxazole, azitromycin, atovaquone, dexamethasone, praziquantel, albendazole, beta-lactams, fluoroquinolones, macrolides, aminoglycosides, sulfadiazine and pyrimethamine.
The structure of the active agents identified by code nos., generic or trade names may be taken from the actual edition of the standard compendium "The Merck Index" or from databases, e.g., Patents International, e.g., IMS World Publications.
The above-mentioned compounds, which can be used in combination with a compound of the formula (I), can be prepared and administered as described in the art, such as in the documents cited above.
A compound of the formula (I) may also be used to advantage in combination with known therapeutic processes, e.g., the administration of hormones or especially radiation.
A compound of formula (I) may in particular be used as a radiosensitizer, especially for the treatment of tumors which exhibit poor sensitivity to radiotherapy.
EXAMPLES Experimental details:
The following examples are intended to illustrate the invention and are not to be construed as being limitations thereon. Temperatures are given in degrees Celsius. If not mentioned otherwise, all evaporations are performed under reduced pressure, typically between about 15 mm Hg and 100 mm Hg (= 20-133 mbar). The structure of final products, intermediates and starting materials is confirmed by standard analytical methods, e.g. , microanalysis and spectroscopic characteristics, e.g., MS, IR, NMR. Abbreviations used are those conventional in the art.
All starting materials, building blocks, reagents, acids, bases, dehydrating agents, solvents, and catalysts utilized to synthesis the compounds of the present invention are either commercially available or can be produced by organic synthesis methods known to one of ordinary skill in the art (Houben-Weyl 4th Ed. 1952, Methods of Organic
Synthesis, Thieme, Volume 21). Further, the compounds of the present invention can be produced by organic synthesis methods known to one of ordinary skill in the art as shown in the following examples.
Abbreviations
ACN acetonitrile
aq. aqueous
Boc fe/ -butoxycarbonyl
B0C2O d\-tert- butyl dicarbonate
br s broad singlet
brine sat. aq. NaCI soln,
Cs2C03 cesium carbonate
d day(s)
dd doublet of doublets
DCM dichloromethane
DIPEA diisopropylethylamine
DMF dimethylformamide
DMSO dimethylsulfoxide
DPPF 1 ,1 '-(bisdiphenylphosphino)ferrocen
eq. equivalent(s)
ESI electrospray ionisation
Et3N triethylamine
Et20 diethylether
EtOAc ethyl acetate
EtOH ethanol
FC flash chromatography
h hour(s)
HPLC high performance liquid chromatography
K3PO4 tripotassium phosphate
LCMS liquid chromatography with mass spectrometry
MeOH methanol
m multiplet
min minute(s)
MS mass spectrometry
mw microwave
nBuOH n-butanol
NaOtBu sodium tert-butoxide
NMR nuclear magnetic resonance spectrometry
Pd2(dba)3 tris(dibenzylideneacetone)dipalladium
Pd(OAc)2 palladium diacetate
PL-HCO3 MP SPE Polymer-supported bicarbonate cartridge for acid removal prep. preparative
PPh3 triphenylphosphine
q quartet
(rac)-BINAP racemic 2,2'-bis(diphenylphosphino)-1 ,1 '-binaphthyl
RP reversed phase
Rt retention time
rt room temperature
sat. saturated
SFC supercritical fluid chromatography
soln. solution
t triplet
t-butyl tert-butyl
tic thin layer chromatography
TFA trifluoroacetic acid
THF tetrahydrofuran
UPLC ultra performance liquid chromatography
XPhos 2-dicyclohexylphosphino-2',4',6'-triisopropylbiphenyl
Microwave equipment used is a Biotage Initiator®
All compounds are named using ChemDraw® Ultra software or commercial names have been used. Preparation of Examples - General Procedures
Scheme 1
the other substituents are as defined above.
a) At rt, a dry solution of anthranilic acid derivative XII (1 eq.) and of enantiomerically pure cyclic amino acid derivative II (1 eq.) in pyridine (0.83 M) was treated with triphenylphosphite (2.5 eq.) and the reaction mixture was stirred at 70 C for 2 to 18 h, then treated with an amino or hydrazine derivative III. The reaction mixture was stirred at 70 to 80°C for 2 to 18 h. b) At rt, a dry solution of Boc intermediate IV (1 eq.) in THF or DCM (0.1 to 0.6 M) was treated with TFA (10 eq.), 4M HCI in dioxane or aq. H3P04 soln. (85 wt%) and the reaction mixture was stirred at rt for 1 h to 5 d.
c) At rt, a solution of deprotected intermediate V (1 eq.) and an aryl halogenide VI (1 to 2.5 eq.) in alcohol (nBuOH or EtOH, 0.08M to 0.1 M) was treated with DIPEA (2 to 5 eq.) and irridiated in a microwave reactor or heated in an oil bath at 120-160°C for 0.5 h to 6 d.
Any functional groups present in R2 , R', R1 and R3'l may be further functionalized in additional steps as described in individual examples shown below. Cyclic amino acid derivative II could alternatively be replaced by racemic 1 -(tert-butoxycarbonyl)-4,5-dihydro- 1 H-pyrazole-5-carboxylic acid VIII (see Scheme 2). Purification of intermediates and final products was carried out by flash chromatography or HPLC chromatography. Separation of diastereomeric atropisomers of final products VII or of enantiomers of final products XI or of debocylated intermediates X was performed by HPLC chromatography.
Scheme 2
General chromatography information
LCMS method M1 (RtM1)
HPLC column dimensions: 2.1 x 50 mm
HPLC column type: Acquity UPLC HSS T3, 1 .8 μηι
HPLC eluent: A) water + 0.05 vol% formic acid + 3.75 mM ammonium acetate B) ACN + 0.04 vol% formic acid
HPLC gradient: 2 - 98% B in 1.4 min, 98% B 0.45 min, flow = 1.2 ml / min
HPLC column temperature: 50°C
LCMS method M2 (RtM2)
HPLC column dimensions: 2.1 x 50 mm
HPLC column type: Acquity UPLC HSS T3, 1 .8 μηι
HPLC eluent: A) water + 0.05 vol% formic acid + 3.75 mM ammonium acetate B) ACN + 0.04 vol% formic acid
HPLC gradient: 5 - 98% B in 1.4 min, 98% B 0.4 min, flow = 1.0 ml / min
HPLC column temperature: 60°C
LCMS method M3 (RtM3):
HPLC column dimensions: 4.6 x 30 mm
HPLC column type: Poroshell 120 EC-18, 2.7 μηι
HPLC eluent: A) water + 0.1 vol% formic acid B) ACN
HPLC gradient (t[min]/%B): 0/30, 2.0/95, 5/95, 5.1/30; flow = 1.0 ml / min
HPLC column temperature: 23°C
LCMS method M4 (RtM4) :
HPLC column dimensions: 4.6 x 50 mm
HPLC column type: Zorbax-SB-C18, 3.1 μηι
HPLC eluent: A) water + 0.1 vol% formic acid B) ACN
HPLC gradient (t[min]/%B): 0/30, 2.0/95, 5/95, 5.1/30; flow = 1.0 ml / min
HPLC column temperature: 23°C
LCMS method M5 (RtM5):
HPLC column dimensions: 4.6 x 50 mm
HPLC column type: Zorbax-SB-C18, 3.1 μηι
HPLC eluent: A) 5 mM ammonium acetate in water B) ACN
HPLC gradient (t[min]/%B): 0/10, 0.5/10, 2.0/90, 5/90, 5.1/10; flow = 1 .0 ml / min HPLC column temperature: 23°C
LCMS method M6 (RtM6):
HPLC column dimensions: 4.6 x 30 mm
HPLC column type: Poroshell 120 EC-18, 2.7 μηι
HPLC eluent: A) 5 mM ammonium acetate in water B) ACN
HPLC gradient (t[min]/%B): 0/10, 0.5/10, 2.0/90, 5/90, 5.1/10; flow = 1.0 ml HPLC column temperature 23°C
LCMS method M7 (RtM7):
HPLC column dimensions: 4.6 x 50 mm
HPLC column type: Zorbax-SB-C18, 3.1 μηι
HPLC eluent: A) 5 mM ammonium acetate in water B) ACN
HPLC gradient (t[min]/%B): 0/10, 0.5/10, 2.0/90, 6/90, 6.1/10; flow = 1 .0 ml / min HPLC column temperature 23°C
LCMS method M8 (RtM8):
HPLC column dimensions: 4.6 x 50 mm
HPLC column type: Zorbax-SB-C18, 3.1 μηι
HPLC eluent: A) water + 0.1 vol% formic acid B) ACN
HPLC gradient (t[min]/%B): 0/30, 2.0/95, 4/95, 4.1/30; flow = 1 .0 ml / min HPLC column temperature 23°C
Preparation of examples
Where it is stated that compounds were prepared in the manner described for an earlier example, the skilled person will appreciate that reaction times, number of equivalents of reagents and reaction temperatures may be modified for each specific reaction, and that it may nevertheless be necessary or desirable to employ different work-up or purification conditions. Compounds that show atropisomerism around bond N-R1 may be obtained as mixtures of diastereomeric atropisomers that may be separated by chromatographic conditions. These compounds are exemplified as mixtures and, if applicable, in addition as separated diastereomeric atropisomers, detailing the separation conditions in each case.
Example A1 : (S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(o- tolyl)quinazolin-4(3H)-one
a) (S)-tert-Butyl 2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)pyrrolidine-1 - carboxylate
A dry solution of 2-amino-6-chlorobenzoic acid (CAS registry 2148-56-3) (1 g, 5.83 mmol) and (S)-Boc-proline (CAS registry 15761 -39-4) (1 .254 g, 5.83 mmol) in pyridine (7.00 ml), was treated with triphenylphosphite (CAS registry 101 -02-0) (3.83 ml, 14.57 mmol). The light brown solution was stirred at 70°C for 2 h (orange solution, formation of the intermediate) and then o-toluidine (CAS registry 95-53-4) (0.757 ml, 6.99 mmol) was added dropwise. The reaction mixture was stirred at 70°C for 2 h, concentrated and then diluted with EtOAc, washed with sat. aq. NaHC03 soln. (2x) and brine. The organic phase was dried over MgS04 and concentrated under reduced pressure. The crude product was purified by
chromatography on silica gel (cyclohexane / EtOAc 100% to 70% cyclohexane) to provide the title compound as yellow gummy solid (1 .43 g, 50% yield).
HPLC peakl / peak2 1 .30 min / 1 .34 min; ESIMS: 440 [(M+H)+].
1H NMR (600 MHz, DMSO-c/6): δ 7.17 - 7.85 (m, 7 H), 3.90 - 4.43 (m, 1 H), 3.36 - 3.63 (m, 2 H), 1 .97 - 2.28 (m, 3 H), 1 .56 - 2.00 (m, 4 H) ,1 .08 - 1 .45 (m, 9 H). b) (S)-5-Chloro-2-(pyrrolidin-2-yl)-3-(o-tolyl)quinazolin-4(3H)-one
A solution of (S)-tert-butyl 2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)pyrrolidine- 1 -carboxylate (1 .23 g, 5.52 mmol) in DCM (1 1 ml) was dropwise treated with TFA (2.15 ml), the light brown solution was stirred at rt for 3.5 h. After being cooled down to 0°C with an ice bath, the reaction mixture was basified to pH=8-9 with using sat. aq. NHC03 soln., the resulting biphasic solution was twice extracted with DCM. The combined organic phases were washed with brine, dried over MgS04 and concentrated under reduced pressure. The crude product was purified by chromatography on silica gel (DCM / MeOH, 100% to 90% DCM) to provide the title compound as a colorless solid (0.805 g, 43% yield).
HPLC peakl / peak2 0.68 min / 0.70 min; ESIMS: 340 [(M+H)+].
1H NMR (600 MHz, DMSO-c/6) δ 7.72 - 7.84 (m, 1 H), 7.65 (d, J=8.09 Hz, 1 H), 3.37 - 3.68 (m, 1 H), 2.92 - 3.13 (m, 1 H), 2.54 - 2.69 (m, 1 H), 2.03 (s, 3 H), 1 .80 - 1 .94 (m, 1 H), 1 .69 - 1 .77 (m, 1 H), 1 .65 (br s, 1 H), 1 .47 - 1 .60 (m, 2 H). c) (S)-(3-((4-(6-Methoxy-5-methylpyridin-3-yl)-3,4-dihydro-2H-benzo[b][1 ,4]oxazin-6- yl)oxy)pyrrolidin-1 -yl)(tetrahydro-2H-pyran-4-yl)methanone
A solution of (S)-5-chloro-2-(pyrrolidin-2-yl)-3-(o-tolyl)quinazolin-4(3H)-one (200 mg, 0.589 mmol) and 6-chloro-9H-purin-2-amine (CAS registry 10310-21 -1) (1 10 mg, 0.647 mmol) in nBuOH (3.27 ml) was treated with DIPEA (206 μΙ_, 1 .18 mmol) and heated in a mw oven at 160°C for 1 .2 h under normal absorption. The crude product was purified by prep. RP-HPLC (column SunFire C18, H20 + 0.1 % TFA / ACN + 0.1 % TFA 80:20 to 60:40 in 16 min) to afford the title compounds as colorless solids (separated diastereoisomers). Free bases were obtained by elution through a Varian ipe PL-HC03 MP cartridge.
1H NMR (400 MHz, DMSO-c/6) δ 12.18 (br s, 1 H), 7.60 - 7.80 (m, 2 H), 7.29 - 7.57 (m, 7 H), 5.12 (br s, 2 H), 4.64 (d, J=7.78 Hz, 1 H), 4.29 (br s, 2 H), 2.00 - 2.27 (m, 2 H), 1 .68 - 1 .96 (m, 2 H). CH3 of tolyl moiety under DMSO peak.
1H NMR (400 MHz, DMSO-c/6) 5 12.12 (br s, 1 H), 7.78 - 8.61 (m, 1 H), 7.25 - 7.74 (m, 7 H),
5.29 - 5.94 (m, 2 H), 4.81 (br s, 1 H), 3.66 - 4.46 (m, 2 H), 1 .48 - 2.21 (m, 7 H).
Examples A2 to A57: The compounds listed in Table 1 were prepared by a procedure analogous to that used in Example A1 using (S)-1 -(tert-butoxycarbonyl)pyrrolidine-2- carboxylic acid (intermediate IA1) as precursor.
Table 1
1 .08
A40 478
(M2)
(S)-4-Amino-6-(2-(4-oxo-3-phenyl-5- (trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
Precursors: IB2, IC2, ID4
Deprotection condition: CB1
(S)-4-Amino-6-(2-(4-oxo-3-(o-tolyl)-5-
A41 (trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
Precursors: IB2, IC1 , ID4
Deprotection condition: CB1
1 .14 / 1 .17 a) Diastereoisomer mixture (A41 a) 492
(M2)
b) Peak 1 (A41 b) 1 .14
492 Separation condition: CD2 (M2)
c) Peak 2 (A41 c) 1 .16
492 Separation condition: CD2 (M2)
A55 1 .12 (M2) 456
(S)-2-Amino-4-(2-(6-fluoro-5-methyl-4-oxo-3- phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)-6-methylpyrimidine-5-carbonitrile
Precursors: IB5, IC2, ID5
Deprotection condition: CB1
A56 1 .13 (M2) 510
(S)-2-Amino-4-(2-(6-fluoro-4-oxo-3-phenyl-5- (trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5- carbonitrile
Precursors: IB7, IC2, ID5
Deprotection condition: CB1
0.98
A67 442
(M2)
(S)-2-Amino-4-(2-(5-fluoro-4-oxo-3-phenyl-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile
Precursors: ΙΒ1 1 , IC2, ID5
Deprotection condition: CB1
Example A68: (S)-2,4-Diamino-6-(2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
a) (S)-4-Chloro-2-(methylthio)-6-(2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
The title compound was prepared according to the procedure described for Example 1 starting from precursors IA1 , IB2, IC2 and ID15.
HPLC RtM2=1 .40; ESIMS: 543, 545 [M+H].
1H NMR (400 MHz, DMSO-d6) δ ppm 7.9 (3 H, m) 7.6 (5 H, m) 4.7 (1 H, d) 4.1 (1 H, d) 4.0 (2 H, m) 2.4 (3 H, s) 2.2 (2 H, m) 1 .9 (3 H, m) b) (S)-4-Chloro-2-(methylsulfonyl)-6-(2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
m-CPBA (38.0 mg, 0.22 mmol) was added to a solution of (S)-4-amino-2-(methylthio)-6-(2- (4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5- carbonitrile (60.0 mg, 0.1 1 mmol) in anhydrous DCM (5 ml) at 0 °C. The mixture was warmed to rt and stirred for 3.5 h.The mixture was partitioned bewtenn DCM (20 ml) and sat. aq. NaHC03, the layers were separated and the organic phase was dried (Na2S04), filtered and concentrated under reduced pressure. The residue was purfied by flash chromatography on silica gel eluting with EtOAc/heptanes (0% - 55% EtOAc) to give the title compound as colorless solid (15 mg, 18%).
HPLC RtM2=1 .1 ; ESIMS: 575 [M+H]. c) (S)-2,4-Diamino-6-(2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
A 2 ml microwave vial was charged with (S)-4-amino-2-(methylsulfonyl)-6-(2-(4-oxo-3- phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile (1 1 .0 mg, 0.019 mmol), 32% aq. NH3 solution (0.5 ml) and THF (0.5 ml). The vial was sealed and the mixture irradiated at 160 °C for 30 min in a microwave oven. The reaction mixture was concentrated under reduced pressure and the residue purified by SFC eluting with MeOH/C02 (23% - 28% MeOH in 6 min; Princeton 250X30 mm, 5 μπτι, 60 A Si02 column). The title compound was obtained as colorless solid (2.4 mg, 25%).
HPLC .02; ESIMS: 493 [M+H].
Examples B1 to B17: The compounds listed in Table 2 were prepared by a procedure analogous to that used in Example A1 , using (R)-3-(tert-butoxycarbonyl)-thiazolidine-4- carboxylic acid (intermediate IA2) as precursor.
Table 2
HPLC Rt MS
Compound /
Example [min] [m/z;
Reaction Conditions
(method) (M+1 )+]
Deprotection condition: CB1
Examples C1 to C11 : The compounds listed in Table 3 were prepared bv a procedure analogous to that used in Example A1 , using (2S,4S)-1 -(tert-butoxycarbonyl)-4- methoxypyrrolidine-2-carboxylic acid (intermediate IA3) as precursor.
Table 3
Deprotection condition: CB1
Examples D1 to D13: The compounds listed in Table 4 were prepared by a procedure analogous to that used in Example A1 , using 1 -(tert-butoxycarbonyl)-4,5-dihydro-1 H- pyrazole-5-carboxylic acid (intermediate IA4) as precursor.
Table 4
HPLC Rt MS
Compound /
Example [min] [m/z;
Reaction Conditions
(method) (M+1 )+]
(M2)
NH2
D13 423
(R or S)-4-Amino-6-(5-(5-methyl-4-oxo-3-phenyl- 16.076
3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- (CD1 1)
pyrazol-1 -yl)pyrimidine-5-carbonitrile
Precursors: IB3, IC2, ID4
Deprotection condition: CB1
Separation condition for debocylated
intermediate: CD1 1
Example E: The compound listed in Table 5 was prepared by a procedure analogous to that used in Example A1 , using (2S,4S)-1 -(tert-butoxycarbonyl)-4-(methylsulfonamido)- pyrrolidine-2-carboxylic acid (intermediate IA5) as precursor.
Table 5
HPLC Rt MS
Compound /
Example [min] [m/z;
Reaction Conditions
(method) (M+1 )+]
Deprotection condition: CB1
Examples F1 to F6: The compounds listed in Table 6 were prepared bv a procedure analogous to that used in Example A1 , using (S)-2-(tert-butoxycarbonyl)isoxazolidine-3-carboxylic acid (intermediate IA6) as precursor
Table 6
HPLC Rt MS
Compound /
Example [min] [m/z;
Reaction Conditions
(method) (M+1 )+]
CI O
0.91
F1 446
ΝΗ2 (M1 )
((S)-4-Amino-6-(3-(5-chloro-4-oxo-3-phenyl-3,4- dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-ca rbonitrile
Precursors: IB1 , IC2, ID4
Deprotection condition: CB1
0.73/074
475 (M1)
(S)-4-Amino-6-(3-(5-chloro-4-oxo-3-(o-tolyl)-3,4- dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile
Precursors: IB1, IC1, ID2
Deprotection condition: CB1
Diastereoisomeric mixture 1/1
Example G1 : 2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile
A solution of (2S,4S)-1 -(tert-butoxycarbonyl)-4-hydroxypyrrolidine-2-carboxylic acid (CAS registry 87691 -27-8) (592 mg, 2.56 mmol) and imidazole (CAS registry 288-32-4) (871 mg, 12.80 mmol) were combined in DCM / DMF (12/2.4 ml) and treated with TBDMSCI (CAS registry 18162-48-6) (849 mg, 5.63 mmol) at rt. The resulting mixture was stirred at rt for 6 h, diluted with EtOAc, washed with sat. aqu. NH4CI soln, dried over MgS04 and concentrated under reduced pressure to afford a colorless oil (1 .49 g, crude).
HPLC F¾MI =1 .20 min; ESIMS: 346 [(M+H)+].
1H NMR (400 MHz, DMSO-d6): 12.12 (br s, 1 H), 4.45-4.31 (m, 1 H), 4.28-4.05 (m, 1 H), 3.65- 3.44 (m, 1 H), 3.13-2.99 (m, 1 H), 2.43-2.24 (m, 1 H), 1 .75-1 .84 (m, 1 H), 1 .38-1 .33 (m, 9H), 0.89-0.81 (m, 9H), 0.02 (m, 6H). b) (2S,4S)-tert-Butyl 4-((tert-butyldimethylsilyl)oxy)-2-(4-oxo-3-phenyl-5- (trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolidine-1 -carboxylate
A solution of 2-amino-6- (trifluoromethyl)benzoic acid (CAS registry 314-46-5) (531 mg, 2.59 mmol) and (2S,4S)-1 -(tert-butoxycarbonyl)-4-((tert-butyldimethylsilyl)oxy)pyrrolidine-2- carboxylic acid (1 .49 g, 2.59 mmol) in pyridine (3.1 1 ml) was treated with triphenylphosphite (CAS registry 101 -02-0) (1 .7 ml, 6.48 mmol). The light brown solution was stirred at 70°C for 18 h, then aniline (CAS registry 62-53-3) (0.28 ml, 3.1 1 mmol) was added dropwise. The reaction mixture was stirred at 70°C for 3 h. The reaction mixture was diluted with EtOAc, washed with sat. aq. CuS04 soln. and brine. The organic phase was dried over MgS04 and concentrated in vacuo to afford an orange oil . The crude product was purified by flash chromatography on silica gel (cyclohexane / EtOAc 100:0 to 85:15) to provide a yellow oil (870 mg, 51 % yield).
HPLC RtM2 =1 .60 min; ESIMS: 590 [(M+H)+].
1H NMR (400 MHz, DMSO-d6): δ 8.01 -6.42 (m, 12 H) 4.38 - 4.19 (m, 2 H) 3.74 - 3.60 (m, 1 H) 3.25 - 3.10 (m, 1 H) 2.17 - 2.04 (m, 1 H) 2.01 - 1 .81 (m, 1 H) 1 .36 - 1 .10 (m, 10 H) 0.74 (s, 9 H) -0.03 (d, J=6.85 Hz, 6 H). c) 2-((2S,4S)-4-Hydroxypyrrolidin-2-yl)-3-phenyl-5-(trifluoromethyl)quinazolin-4(3H)- one
At rt, a solution of (2S,4S)-tert-butyl 4-((tert-butyldimethylsilyl)oxy)-2-(4-oxo-3-phenyl-5- (trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolidine-1 -carboxylate (0.87 g, 1 .31 mmol) in THF (2 ml) was treated with aq. H3P04 soln. (85 wt% purchased from Aldrich) (3 ml). The mixture was stirred for 5 d at rt. Water was added and the mixture was basified with a sat. aq. NaHC03 soln. to pH~9. The mixture was extracted with EtOAc (four times), the combined organic layers were dried over MgS04 and concentrated in vacuo to afford a beige oil (552 mg). The oil was triturated with Et20, dried under high vacuum to afford the title compound as a white solid (290 mg, 59% yield).
HPLC RtM2 =0.63 min; ESIMS: 376 [(M+H)+]. d) 2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile
A solution of 2-((2S,4S)-4-hydroxypyrrolidin-2-yl)-3-phenyl-5-trifluoromethyl)quinazolin-4(3H)- one (290 mg, 0.77 mmol) and 2-amino-4-chloro-6-methylpyrimidine-5-carbonitrile (CAS registry 99586-66-0) (130 mg, 0.77 mmol) in nBuOH (7.7 ml) was treated with DIPEA (0.540 ml, 3.09 mmol) and was stirred at 90°C for 18 h in an oil bath. The reaction mixture was cooled down, and evaporated under reduced pressure. The oil was taken up in DCM and washed with water, the organic layer was dried over MgS04 and concentrated in vacuo (500 mg). The crude was purified over SFC (column NH2, 250 x 30 mm, 60A, 5μηι, Princeton, flow at 100 ml/min; gradient of MeOH in supercritical C02, from 17% to 22% in 1 1 min) and triturated with pentane to afford the title compound as a beige solid (258 mg, 66% yield). HPLC RtM2 =0.94 min; ESIMS: 508 [(M+H)+].
1H NMR (400 MHz, DMSO-d6): δ 7.98 - 7.80 (m, 4H), 7.65-7.47 (m, 4H), 7.13-6.52 (m, 2H), 5.25 (br s, 1 H), 4.65 (br s, 1 H), 4.17 (d, 2H), 3.79 - 3.60 (m, 1 H), 2.27 (s, 3H), 2.07-1 .96 (m, 1 H), 1 .96-1 .84 (m, 1 H).
Examples G2 to G8: The compounds listed in Table 7 were prepared by a procedure analogous to that used in Example G1 , using (2S,4S)-1 -(tert-butoxycarbonyl)-4-((tert-butyldi- methylsilyl)oxy)pyrrolidine-2-carboxylic acid (intermediate IA7) as precursor.
Example G31 : 2-Amino-4-((2S,4S)-2-(5-cyclopropyl-6-fluoro-4-oxo-3-phenyl-3,4- dihydroquinazolin-2-yl)-4-hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile
A solution of 2-amino-4-((2S,4S)-2-(5-bromo-6-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin- 2-yl)-4-hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile (31 mg, 0.058 mmol, reference example G28) in dioxane (0.5 ml) and water (0.1 ml) was treated with
cyclopropylboronic acid (CAS registry 41 1235-57-9) (14.9 mg, 0.173 mmol),
tricyclohexylphosphine (CAS registry 2622-14-2) (3.24 mg, 0.012 mmol) and potassium phosphate tribasic (CAS registry 7778-53-2) (36.8mg, 0.173 mmol). The reaction mixture was degassed with argon for 5 min and treated with diacetoxypalladium (CAS registry 3375- 31 -3) (1 .3 mg, 0.0058 mmol). The reaction mixture was stirred in a sealed vessel under argon atmosphere at 100°C for 1 h, then diluted with DCM and washed with sat. aq. NaHC03 soln., filtered through a Biotage Universal Phase Separator cartridge and the combined organic phases were concentrated under reduced pressure. The crude product was purified by prep. RP-HPLC (column SunFire C18, H20 + 0.1 % TFA / ACN + 0.1 % TFA 79:21 to 59:41 in 16 min) to afford the title compound as colorless solid. The free base was obtained by elution through a Varian ipe PL-HC03 MP cartridge (7.4 mg, 26%).
HPLC RtM2 =0.99 min; ESIMS: 498 [(M+H)+] ;
1H NMR (400 MHz, CHLOROFORM-d): 7.70-7.49 (m, 4H), 7.44-7.32 (m, 2H), 7.23-7.16 (m, 1 H), 5.85 (d, 1 H), 4.99 (br. S, 2H), 4.83-4.72 (m, 1 H), 4.61 -4.47 (m, 2H), 4.1 1 (dd, 1 H), 2.79- 2.69 (m, 1 H), 2.44 (s, 3H), 2.25-2.09 (m, 2H), 1 .09-1 .02 (m, 2H), 0.96-0.90 (m, 2H). Example H: 5-Chloro-2-((2S,4S)-4-(difluoromethoxy)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-3- phenylquinazolin-4(3H)-one
a) 5-Chloro-2-((2S,4S)-4-hydroxy-1 -(9-(tetrahydro-2H-pyran-2-yl)-9H-purin-6- yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)-one
A solution of 5-chloro-2-((2S,4S)-4-hydroxypyrrolidin-2-yl)-3-phenylquinazolin-4(3H)-one (prepared as described in example G01) (400 mg, 1 .17 mmol) and 6-bromo-9-(tetrahydro- 2H-pyran-2-yl)-9H-purine (CAS registry 381246-26-0) (331 mg, 1 .17 mmol) in nBuOH (1 1 ml) was treated with DIPEA (0.41 ml, 2.34 mmol) and stirred at 90°C for 7 h in a dry-syn alu block. The reaction mixture was cooled down and evaporated to dryness to afford an orange oil. The crude product was purified by flash chromatography on silica gel (cyclohexane / EtOAc 100:0 to 60:40, then cyclohexane / EtOAc 100:0 to 0:100) to provide the title compound as a colorless solid (215 mg, 34% yield).
HPLC RtM2 =1 .04 min; ESIMS: 544 [(M+H)+]. b) 5-Chloro-2-((2S,4S)-4-(difluoromethoxy)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-3- phenylquinazolin-4(3H)-one
A suspension of 5-chloro-2-((2S,4S)-4-hydroxy-1 -(9-(tetrahydro-2H-pyran-2-yl)-9H-purin-6- yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)-one (400 mg, 1 .17 mmol) and Cul (7.7 mg, 0.04 mmol) in ACN (14 ml) at 45°C was treated with 2,2-difluoro-2-(fluorosulfonyl)acetic acid (CAS registry 1717-59-5) (42 uL, 0.40 mmol) in ACN (0.4 ml) over 3 h. The resulting yellow solution was stirred at 45°C for 18 h, diluted with EtOAc, washed with brine, dried over
MgS04 and concentrated under reduced pressure to afford an orange residue. MeOH was added and the mixture was filtered off, the filtrate was purified over SFC (column silica 250 x 30 mm, 60A, 5μηι, Princeton, flow at 100 ml/min; gradient of MeOH in supercritical C02, from 22% to 27% in 1 1 min) and triturated with pentane to afford the title compound as a colorless solid (1 1 .8 mg, 1 1 % yield).
HPLC RtM2 =0.99 min; ESIMS: 510 [(M+H)+].
1H NMR (400 MHz, 120°C, DMSO-d6): δ 8.20 (s, 1 H), 8.00 (s, 1 H), 7.94-7.70 (m, 1 H), 7.66- 7.50 (m, 5H), 7.40 (m, 2H), 7.30 (dd, 1 H), 6.86-6.38 (t, 1 H), 5.27-5.09 (m, 1 H), 4.92-4.76 (m, 2H), 4.1 1 -3.95 (m, 1 H), 2.50-2.38 (m, 1 H), 2.32-2.26 (m, 1 H).
Example I: (S)-4-Amino-6-(2-(5-hydroxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
a) (S)-tert-Butyl 2-(5-methoxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidine-1 - carboxylate
A solution of 2-amino-6-methoxybenzoic acid (CAS registry 53600-33-2) (500 mg, 2.99 mmol) and (S)-1 -(tert-butoxycarbonyl)pyrrolidine-2-carboxylic acid (CAS registry 15761 -39-4) (644 mg, 2.99 mmol) in pyridine (3.6 ml), was treated with triphenylphosphite (CAS registry 101 -02-0) (1 .9 ml, 7.48 mmol). The yellow solution was stirred at 70°C for 2 h and then aniline (CAS registry 62-53-3) (0.3 ml, 7.48 mmol) was added dropwise. The reaction mixture was stirred at 70°C for 1 .5 h. The reaction mixture was diluted with EtOAc, washed with sat. aq. NaHC03 soln. and brine. The organic phase was dried over MgS04 and concentrated in vacuo to afford a yellow oil (pyridine trapped). The crude product was purified by flash chromatography on silica gel (cyclohexane / EtOAc 100:0 to 0:100) to provide the title compound as a colorless solid (760 mg, 60% yield).
HPLC RtM2 =1 .1 1 min; ESIMS: 422 [(M+H)+].
1H NMR (400 MHz, DMSO-d6): δ 7.74-7.65 (m, 1 H), 7.65-7.48 (m, 3H), 7.48-7.38 (m, 1 H), 7.29 (d, 1 H), 7.16-7.07 (m, 1 H), 7.06-6.89 (m,1 H), 4.23-4.10 (m, 1 H), 3.82 (s, 3H), 3.58-3.46 (m, 1 H), 3.32-3.17 (m, 1 H), 2.09-1 .95 (m, 1 H), 1 .95-1 .73 (m, 2H), 1 .73-1 .60 (m, 1 H), 1 .34- 1 .02 (d, 10H). b) (S)-5-Hydroxy-3-phenyl-2-(pyrrolidin-2-yl)quinazolin-4(3H)-one
A colorless stirred solution of (S)-tert-butyl 2-(5-methoxy-4-oxo-3-phenyl-3,4- dihydroquinazolin-2-yl)pyrrolidine-1 -carboxylate (0.7 g, 1 .80 mmol) in DCM (9 ml) was treated with BBr3 (1 M in DCM) (9 ml, 9.02 mmol) with rapid stirring at 0°C, the yellow suspension was stirred for 2 h at rt, then poured into crushed ice. The reaction mixture was basified until pH = 9 using sat. aq. NaHC03 soln. and the organic layer was separated and dried by elution through a phase separating cartridge and concentrated under reduced pressure to afford the title compound as a yellow oil (558 mg, crude, rotamers).
HPLC RtM2 =0.62 min; ESIMS: 308 [(M+H)+].
1H NMR (400 MHz, 100°C, DMSO-d6): δ 1 1 .50 (br s, 1 H), 7.78-7.68 (m, 1 H), 7.65-7.40 (m, 5H), 7.18 (dd, 1 H), 6.94-6.85 (m, 1 H), 3.80-3.76 (m, 0.5H), 3.78-3.03 (m, 1 .5H), 2.77-2.63 (m, 0.5H), 2.27 (m, 0.5H), 2.01 -1 .56 (m, 4H). c) (S)-4-Amino-6-(2-(5-hydroxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile
A solution of (S)-5-hydroxy-3-phenyl-2-(pyrrolidin-2-yl)quinazolin-4(3H)-one (100 mg, 0.32 mmol) and 4-amino-6-chloropyrimidine-5-carbonitrile (CAS registry 60025-09-4) (55.3 mg, 0.32 mmol) in nBuOH (2 ml) was treated with DIPEA (0.1 14 ml, 0.65 mmol) and heated in a mw oven at 160°C for 1 h under 2 bars and normal absorption. The formed precipitate was filtered off, washed with MeOH, Et20, pentane, dried in high vacum to afford the title compound as an off-white solid (34 mg, 24% yield).
HPLC RtM2 =1 .02 min; ESIMS: 426 [(M+H)+].
1H NMR (400 MHz, DMSO-d6): δ 1 1 .55 (s, 1 H), 7.98 (s, 1 H), 7.72-7.52 (m, 6H), 7.41 (m, 2H),
7.03 (d, 1 H), 6.85 (d, 1 H), 4.68-4.54 (m, 1 H), 4.08-3.96 (m, 1 H), 3.95-3.79 (m, 1 H), 2.24-1 .76
(m, 4H).
Example J1 : (S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro- 3-phenylquinazolin-4(3H)-one
A dry solution of 2-amino-6-chlorobenzoic acid (CAS registry 2148-56-3) (2 g, 1 1 .66 mmol) and (S)-Boc-proline (CAS registry 1 5761 -39-4) (2.509 g, 1 1 .66 mmol) in pyridine (14.0 ml) was treated with triphenylphosphite (CAS registry 101 -02-0) (7.66 ml, 29.1 mmol). The light brown solution was stirred at 70°C for 2 h (orange solution, formation of the intermediate) and then aniline (CAS registry 62-53-3) (1 .47 ml, 14.0 mmol) was dropwise added. The
reaction mixture was stirred at 70°C for 2 h. The reaction mixture was concentrated, then diluted with EtOAc, washed with sat. aq. NaHC03 soln. (2x) and brine. The organic phase was dried over MgS04 and concentrated under reduced pressure. The crude product was purified by chromatography on silica gel (cyclohexane / EtOAc 100:0 to 50:50) to provide the title compound as yellow gummy solid (4.52 g, 86% yield).
HPLC RtMi = 1 .24 min; ESIMS: 426 [(M+H)+].
1H NMR (600 MHz, DMSO-c/6): δ 7.14 - 7.88 (m, 8 H),. 4.03 - 4.36 (m, 1 H), 3.38 - 3.66 (m, 1 H), 3.19 - 3.37 (m, 1 H), 1 .55 - 2.14 (m, 4 H), 1 .04 - 1 .50 (m, 9 H). b) (S)-5-Chloro-3-phenyl-2-(pyrrolidin-2-yl)quinazolin-4(3H)-one
A solution of (S)-tert-butyl 2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidine- 1 -carboxylate (3.82 g, 8.97 mmol.) in DCM (36 ml) was treated dropwise with TFA (6.91 ml), the light brown solution was stirred at rt for 15 h. After being cooled down to 0°C with an ice bath, the mixture was basified until pH=8-9 using a sat. aq. NHC03 soln, the organic phase was dried by elution through a separating phase cartridge. The organic phase was concentrated under reduced pressure to afford the title compound as a colorless solid (1 .86 g, 64% yield).
HPLC RtMi = 0.64 min; ESIMS: 326 [(M+H)+].
1H NMR (600 MHz, DMSO-c/6): δ 7.67 - 7.83 (m, 1 H),.7.28 - 7.65 (m, 7 H), 3.42 - 3.68 (m, 1 H), 2.90- 3.14 (m, 1 H), 2.55 - 2.73 (m, 1 H), 1 .70 - 1 .96 (m, 1 H), 1 .31 - 1 .70 (m, 3 H). c) (S)-5-Chloro-2-(1 -(2-(methylthio)imidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-3- phenylquinazolin-4(3H)-one
A solution of (S)-5-chloro-3-phenyl-2-(pyrrolidin-2-yl)quinazolin-4(3H)-one (93 mg, 0.284 mmol) and 4-chloro-2-(methylthio)imidazo[2,1 -f][1 ,2,4]triazine (ID7) (1 10 mg, 0.647 mmol) in
EtOH (2 ml) was treated with DIPEA (100 μΙ_, 0.568 mmol) and stirred at 120°C for 16.5 h.
The reaction mixture was cooled down to rt, then diluted with DCM and washed with a sat. aq. NaHC03 soln., then water. The aq. phases were backextracted with DCM. The combined organic phases were dried over Na2S04 and concentrated under reduced pressure. The crude product was purified by chromatography on silica gel (heptane / EtOAc 88:12 to 0:100) to provide the title compound as yellow compound (128 mg, 92% yield).
HPLC RtMi = 1 .29 min; ESIMS: 490 [(M+H)+].
1H NMR (400 MHz, CDCI3) δ 7.17 - 8.31 (m, 10 H), 5.73 - 5.97 (m, 0.5 H), 4.79 - 5.00 (m, 1 ,5 H), 4.46 - 4.73 (m, 0.5 H), 3.85 - 4.02 (m, 0.5 H), 2.36 - 2.70 (m, 3 H), 1 .90 - 2.25 (m, 4 H). d) (S)-5-Chloro-2-(1 -(2-(methylsulfonyl)imidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)- 3-phenylquinazolin-4(3H)-one
A solution of (S)-5-chloro-2-(1 -(2-(methylthio)imidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)- 3-phenylquinazolin-4(3H)-one (125 mg, 0.255 mmol.) in dioxane / H20 (10 / 3.5 ml) was treated with oxone (CAS registry 70693-62-8) (549 mg, 0.893 mmol.). The reaction mixture was stirred at rt for 24 h. The reaction mixture was diluted with EtOAc and washed successively with H20 and a sat. aq. NaHC03 soln. The combined aq. phases were back- extracted with EtOAc, and the combined organic phases were dried with Na2S04 and concentrated under pressure to afford the title compound (131 mg, 98% yield).
HPLC 1 .08 min; ESIMS: 522 [(M+H)+]. e) (S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3- phenylquinazolin-4(3H)-one
A solution of (S)-5-chloro-2-(1 -(2-(methylsulfonyl)imidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2- yl)quinazolin-4(3H)-one (46.54 mg, 0.089 mmol) in DMSO (1 .27 ml) was treated with a 32% aq. NH4OH soln. (325 μΙ, 2.67 mmol.). The reaction mixture was stirred in a closed vial at 140°C for 6 d. The reaction mixture was diluted with H20 and DCM, the aq. phase was extracted with DCM (2x) and the combined organic phases were dried over an 1ST separating phase cartridge, then concentrated under reduced pressure to afford an orange oil. The crude product was purified by SFC (column Silica 250x30 mm, 5um, Princeton, gradient of MeOH in supercritical C02: 21 -26-% MeOH in 6 min) to afford the tiltle compound as a colorless solid (diastereoisomer mixture, 2.88 mg, 6% yield).
HPLC RtM2= 1 .04 min; ESIMS: 459 [(M+H)+].
1H NMR (600 MHz, DMSO-c/6) δ 8.13 (d, J=8.05 Hz, 0.5 H) 7.92 (d, J=7.68 Hz, 0.5 H) 7.26 - 7.77 (m, 8 H) 5.65 - 5.96 (m, 2 H) 5.58 (d, J=6.59 Hz, 0.5 H) 4.64 - 4.75 (m, 0.5 H) 4.32 - 4.50 (m, 1 H) 3.70 - 3.99 (m, 1 H) 2.30 (br s, 0.5 H) 2.18 (d, J=6.95 Hz, 1 H) 1 .76 - 2.05 (m, 2.5 H)
Examples J2 to J4: The compounds listed in Table 8 were prepared by a procedure analogous to that used in Example G1 .
Table 8
HPLC Rt MS
Compound /
Example [min] [m/z;
Reaction Conditions
0.97
J2 468
(S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4- (M2)
yl)pyrrolidin-2-yl)-5-chloro-3-morpholino- quinazolin-4(3H)-one
Precursors: IB1 , IC8, ID8
Deprotection condition: CB1
The two last step
Oxidation condition CE
Amination conditon CF
1 .09-1 .12
J3 (S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4- 473
(M2)
yl)pyrrolidin-2-yl)-5-chloro-3-(o-tolyl)quinazolin- 4(3H)-one
Precursors: IB1 , IC1 , ID7
Deprotection condition: CB1
Two last steps:
Oxidation condition: CE
Amination condition: CF
Diastereoisomeric mixture
Example K1 : 2-((S)-1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((S)-1 - (methylsulfonyl)pyrrolidin-3-yl)quinazolin-4(3H)-one
a) (S)-tert-Butyl 2-(3-((S)-1 -((benzyloxy)carbonyl)pyrrolidin-3-yl)-5-chloro-4-oxo-3,4- dihydroquinazolin-2-yl)pyrrolidine-1 -carboxylate
A stirred solution of 2-amino-chlorobenzoic acid (CAS registry 2148-56-3) (0.61 g, 3.49 mmol) and Boc-L-proline (CAS registry 15761 -39-4) (0.76 g, 3.49 mmol) in pyridine (5.6 ml) was treated dropwise with P(OPh)3 (2.20 ml, 8.38 mmol) at rt. After 2.5 h at 70°C (S)-benzyl- 3-aminopyrrolidine-1 -carboxylate (CAS registry 122536-72-5) was added and the solution was stirred at 80°C for 18 h. After cooling to rt, the reaction mixture was dissolved with
EtOAc and washed with sat. NaHC03-soln. and brine. The organics were dried (Na2S04) and concentrated under reduced pressure. Purification by flash chromatography on silica gel (hexanes/EtOAc 100:0 to 70:30 then hexane/EtOAc 50:50) provided the title compound (290 mg, 14% yield) as a colorless foam.
ESIMS: 553 [(M+H)+];
1H NMR (600 MHz, DMSO, rotamers): 7.73-7.68 (m, 1 H), 7.55-7.30 (m, 7H), 2.25-5.05 (m, 4H), 3.86-3.72 (m, 2H), 3.71 -3.56 (m, 2H), 3.55-3.42 (m, 2H), 2.68-2.40 (m, 2H), 2.22-1 .85 (m, 4H), 1 .10/1 .07 (2, 9H). b) (S)-Benzyl 3-(5-chloro-4-oxo-2-((S)-pyrrolidin-2-yl)quinazolin-3(4H)-yl)pyrrolidine-1 - carboxylate
A stirred solution of (S)-tert-butyl 2-(3-((S)-1 -((benzyloxy)carbonyl)pyrrolidin-3-yl)-5-chloro-4- oxo-3, 4-dihydroquinazolin-2-yl)pyrrolidine-1 -carboxylate (290 mg. 0.52 mmol) in dioxane (5 ml) was treated with a 4M solution of HCI in dioxane (3.28 ml, 13.1 1 mmol) and stirred at rt. After 18 h solvents were removed under reduced pressure and the residue (255 mg, 99% yield) used for the next step without further purification.
ESIMS: 453 [(M+H)+].
c) (S)-Benzyl 3-(2-((S)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4-oxoquinazolin-3(4H)- yl)pyrrolidine-1 -carboxylate
A solution of (S)-benzyl 3-(5-chloro-4-oxo-2-((S)-pyrrolidin-2-yl)quinazolin-3(4H)- yl)pyrrolidine-1 -carboxylate (255 mg, 0.52 mmol), DIPEA (0.27 ml, 1 .56 mmol) and 6-chloro- purine (CAS registry 87-42-3) (81 mg, 0.52 mmol) in BuOH (3 ml) was irradiated at 160°C in a mw reactor for 1 h. Then, solvent was removed under reduced pressure and the residue purified by flash chromatography (DCM/MeOH 100:0 to 96:4) to yield the title compound as a colorless solid (215 mg, 72% yield).
ESIMS: 571 [(M+H)+];
1H NMR (400 MHz, DMSO, rotamers): 8.77/8.69 (br. S, 1 H), 8.25 (s, 1 H), 7.87/7.71 (s, 1 H), 7.62-7.51 (m, 1 H), 7.49-7.21 (m, 7H), 6.35-6.26 (m, 1 H), 5.52-5.41 (m, 1 H), 5.14 (br. d, 2H), 4.21/4.1 1 (tr., 1 H), 4.03-3.76 (m, 4H), 3.62-3.49 (m, 1 H), 2.73-2.54 (m, 2H), 2.31 -1 .97 (m, 4H). d) 2-((S)-1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((S)-pyrrolidin-3-yl)quinazolin- 4(3H)-one hydrobromide
A suspension of (S)-benzyl 3-(2-((S)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4- oxoquinazolin-3(4H)-yl)pyrrolidine-1 -carboxylate (215 mg, 0.38 mmol) in 33% HBr in HOAc (4 ml) was stirred at rt. After 16 h solvents were removed under reduced pressure. The sticky off-white residue was taken up in MeOH (1 ml), Et20 was added until the solution became turbid and stirring continued until an off-white precipitate was formed. Solvents were decanted, precipitate washed with Et20 and dried under reduced pressure to yield the title compound as off-white solid (131 mg, 67% yield) which was used for the next step without further purification.
ESIMS: 435 [(M+H)+]. e) 2-((S)-1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((S)-1 -(methylsulfonyl)pyrrolidin-3- yl)quinazolin-4(3H)-one
A suspension of 2-((S)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((S)-pyrrolidin-3- yl)quinazolin-4(3H)-one hydrobromide (65 mg, 0.13 mmol) in CH2CI2 (4 ml) was treated with
DIPEA (49 mg, 0.38 mmol) and stirred at rt for 10 min until it was partially dissolved. Then methanesulfonyl chloride (17 mg, 015 mmol) dissolved in CH2CI2 (0.15 ml) was slowly added and stirring was continued. After 1 h the reaction mixture was poured on sat. NaHC03 soln. and CH2CI2 and the phases separated. The aq. phase was twice extracted with DCM, the combined organics dried (Na2S04) and solvents removed under reduced pressure.
Purification by flash chromatography (DCM/MeOH 100:0 to 95:5) on silica gel yielded the title compound as colorless solid (35 mg, 51 % yield).
ESIMS: 513 [(M+H)+];
1M NMR (400 MHz, DMSO, 100°C, rotamers): 12.58 (br. S, 1H), 8.18 (br. S, 1H), 7.86 (br. S, 1H), 7.55 (t, 1H), 7.41 (d, 1H), 7.25 (d, 1H), 6.16 (br. S, 1H), 5.54-5.40 (m, 1H), 4.23-3.97 (m, 2H), 3.91-3.74 (m, 2H), 3.71-3.48 (m, 2H), 3.04/3.02 (s, 2H), 2.77-2.05 (m, 6H).
Examples K2 to K5: The compounds listed in Table 9 were prepared by a procedure analogous to that used in Example K1.
Table 9
K5 0.90 (M2) 542
2- ((S)-1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-
3- ((R)-1 -(cyclopropylsulfonyl)pyrrolidin-3- yl)quinazolin-4(3H)-one
Precursors: IB1 , IC18, ID1
Deprotection condition: CB2
Example L1 : (S)-4-Amino-6-(2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
A dry solution of 2-amino-6-bromobenzoic acid (CAS registry 20776-48-1) (612 mg, 2.83 mmol) and (S)-Boc-proline (CAS registry 15761 -39-4) (610 mg, 2.83 mmol) in pyridine (4.00 ml), was treated with triphenylphosphite (CAS registry 101 -02-0) (1 .86 ml, 7.08 mmol). The light brown solution was stirred at 70°C for 1 h (orange solution, formation of the intermediate) and then aniline (CAS registry 62-53-3) (0.31 ml, 3.40 mmol) was added dropwise. The reaction mixture was stirred at 70°C for 18 h, concentrated and then diluted with EtOAc, washed with sat. aq. NaHC03 soln. (2x) and brine. The organic phase was dried over Na2S04 and concentrated under reduced pressure. The crude product was purified by
chromatography on silica gel (heptane / EtOAc 100% to 50% heptane) to provide the title compound as white gummy solid (1 .19 g, 76 % yield).
HPLC RtM2= 1 .27 min; ESIMS: 472 [(M+H)+].
1H NMR (400 MHz, DMSO-c/6): δ 7.43 - 7.83 (m, 7 H), 7.21 - 7.38 (m, 1 H), 4.05 - 4.27 (m, 1 H), 3.47 - 3.57 (m, 1 H), 3.25 - 3.38 (m, 1 H), 1 .56 - 2.10 (m, 4 H), 1 .14 - 1 .36 (m, 9 H).
b) (S)-tert-Butyl 2-(5-(methylthio)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidine-1 -carboxylate
A solution of (S)-tert-butyl 2-(5-bromo-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidine- 1 -carboxylate (206 mg, 0.438 mmol) in toluene (2 ml) was treated with sodium thiomethoxide (CAS registry 5188-07-8) (33.8 mg, 0.482 mmol), 4,5-bis(diphenylphosphino)-9,9- dimethylxanthene (CAS registry 161265-03-8) (50.7 mg, 0.088 mmol), and the mixture was bubbled 5 min with argon, then tris(dibenzylideneacetone)dipalladium(0) (CAS registry 51364-51 -3) (40.1 mg, 0.044 mmol) was added. The reaction mixture was floated with argon, and stirred in a sealed tube at 100°C for 18 h, then diluted with EtOAc, washed with sat. aq. NaHC03 soln. (2x) and brine. The organic phase was dried over Na2S04 and concentrated under reduced pressure. The crude product was purified by chromatography on silica gel (heptane / EtOAc 48% to 40% heptane) to provide the title compound as white gummy solid (164 mg, 77 % yield).
HPLC RtM2= 1 .24 min; ESIMS: 438 [(M+H)+]. c) (S)-tert-Butyl 2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidine-1 -carboxylate
An orange solution of (S)-tert-butyl 2-(5-(methylthio)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidine-1 -carboxylate (164 mg, 0.32 mmol) in THF (1 .5 ml) and water (0.5 ml) was treated with potassium peroxymonosulfate (Oxone, CAS registry 70693-62-8) (588 mg, 0.96 mmol). The reaction mixture was stirred at rt for 18 h, then diluted with EtOAc andwashed with sat. aq. NaHC03 soln.. The organic phase was dried over Na2S04 and concentrated under reduced pressure. The crude product was purified by chromatography on silica gel (heptane / EtOAc 100% to 0% heptane) to provide the title compound as clear gummy solid (1 12 mg, 75 % yield).
HPLC RtM2= 1 .09 min; ESIMS: 470 [(M+H)+].
1H NMR (400 MHz, DMSO-c/6): δ 8.27 (t, 1 H), 7.90 - 8.09 (m, 2 H), 7.48 - 7.71 (m, 4 H), 7.33 (dd, 1 H), 4.20 - 4.31 (m, 1 H), 3.51 - 3.60 (m, 1 H), 3.48 (s, 3 H), 3.27 - 3.38 (m, 1 H), 1 .65 - 2.17 (m, 4 H), 1 .26 (d, 9 H). d) (S)-5-(Methylsulfonyl)-3-phenyl-2-(pyrrolidin-2-yl)quinazolin-4(3H)-one
A solution of (S)-tert-butyl 2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidine-1 -carboxylate (150 mg, 0.32 mmol) in DCM (3 ml) was dropwise treated with TFA (0.25 ml, 3.19 mmol), the light brown solution was stirred at rt for 18 h. After being cooled down to 0°C with an ice bath, the reaction mixture was basified to pH=8-9 with using sat. aq. NHC03 soln., the resulting biphasic solution was twice extracted with DCM. The combined organic phases were dried over Na2S04 and concentrated under reduced pressure to provide the title compound as a brown solid (1 14 mg, 97 % yield, crude).
HPLC RtM2= 0.50 min; ESIMS: 370 [(M+H)+]. e) (S)-4-Amino-6-(2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
A solution of (S)-5-(methylsulfonyl)-3-phenyl-2-(pyrrolidin-2-yl)quinazolin-4(3H)-one (38 mg, 0.10 mmol) and 4-amino-6-chloropyrimidine-5-carbonitrile (CAS registry 60025-09-4) (15.9 mg, 0.10 mmol) in EtOH (1 ml) was treated with DIPEA (0.045 ml, 0.26 mmol) and was stirred at 120°C for 45 min in mw. The reaction mixture was evaporated under reduced pressure. The oil was taken up in DCM and washed with sat. aq. NHC03 soln., the organic layer was dried over Na2S04 and concentrated in vacuo. The crude was purified over SFC (column PPU, 250 x 30 mm, 60A, 5μηι, Princeton, flow at 100 ml/min; gradient of MeOH in supercritical C02, from 18 % to 23 % in 1 1 min) to afford the title compound as a beige solid (33 mg, 63 % yield).
HPLC RtM2 =0.87 min; ESIMS: 488 [(M+H)+].
1 H NMR (400 MHz, DMSO-c/6): δ 8.24 (dd, 1 H), 7.85 - 8.05 (m, 3 H), 7.50 - 7.70 (m, 5 H), 7.24 (br s, 2 H), 4.50 - 4.75 (m, 1 H), 3.99 - 4.16 (m, 1 H), 3.80 - 3.97 (m, 1 H), 3.48 (s, 3 H), 2.18 - 2.31 (m, 1 H), 2.05 - 2.18 (m, 1 H), 1 .78 - 2.03 (m, 2 H).
Example L2: The compound listed in Table 10 was prepared by a procedure analogous to that used in Example L1 .
Table 10
HPLC Rt MS
Compound /
Example [min] [m/z;
Reaction Conditions
Deprotection condition: CB1
Example M: 2-Amino-4-(difluoromethyl)-6-((2S,4S)-4-hydroxy-2-(4-oxo-3-phenyl-5- (trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitn
a) (2S,4S)-tert-Butyl 4-((tert-butyldimethylsilyl)oxy)-2-(4-oxo-3-phenyl-5- (trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolidine-1 -carboxylate
A solution of 2-amino-6- (trifluoromethyl)benzoic acid (CAS registry 314-46-5) (1 .07 g, 5.20 mmol) and intermediate IA7 (2.64 g, 5.20 mmol) in pyridine (6.24 ml) was treated with triphenylphosphite (CAS registry 101 -02-0) (3.42 ml, 13.00 mmol). The light brown solution was stirred at 70°C for 18 h, then aniline (CAS registry 62-53-3) (0.57 ml, 6.24 mmol) was added dropwise. The reaction mixture was stirred at 70°C for 3 h. The reaction mixture was diluted with EtOAc, washed with sat. aq. CuS04 soln. and brine. The organic phase was dried over MgS04 and concentrated in vacuo to afford an orange oil. The crude product was
purified by flash chromatography on silica gel (cyclohexane / EtOAc 100:0 to 85:15) to provide a white solid (1 .7 g, 55% yield).
HPLC RtM2 =1 .59 min; ESIMS: 590 [(M+H)+].
1H NMR (400 MHz, DMSO-d6) δ ppm 7.89 - 8.03 (m, 3 H) 7.51 - 7.69 (m, 3 H) 7.41 - 7.51 (m, 1 H) 7.24 (d, 1 H) 4.16 - 4.37 (m, 2 H) 3.59 - 3.75 (m, 1 H) 3.22 (br. s., 1 H) 2.00 - 2.18 (m, 1 H) 1 .78 - 1 .92 (m, 1 H) 1 .34 (s, 3 H) 1 .16 (s, 7 H) 0.74 (s, 9 H) -0.03 (d, 6 H). b) 2-((2S,4S)-4-Hydroxypyrrolidin-2-yl)-3-phenyl-5-(trifluoromethyl)quinazolin-4(3H)- one
At rt, a solution of (2S,4S)-tert-butyl 4-((tert-butyldimethylsilyl)oxy)-2-(4-oxo-3-phenyl-5- (trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolidine-1 -carboxylate (1 .70 g, 2.88 mmol) in THF (10.7 ml) was treated with aq. H3P04 soln. (85 wt% purchased from Aldrich) (12 ml). The mixture was stirred for 18 h at rt. Water was added and the mixture was basified with a sat. aq. NaHC03 soln. to pH~9. The mixture was extracted with EtOAc (twice), the combined organic layers were dried over MgS04 and concentrated in vacuo to afford a white solid (1 g). The solid was triturated with Et20, dried under high vacuum to afford the title compound as a white solid (810 mg, 75% yield).
1H NMR (400 MHz, DMSO-d6) δ ppm 7.91 - 8.06 (m, 3 H) 7.41 - 7.61 (m, 5 H) 4.66 (d, 1 H) 4.03 (br. s., 1 H) 3.50 - 3.67 (m, 1 H) 3.17 (br. s., 1 H) 2.85 (d, 1 H) 1 .48 - 1 .76 (m, 2 H). c) 2-Amino-6-(difluoromethyl)pyrimidin-4(3H)-one
A mixture of guanidine (CAS registry 1 13-00-8) (1 .2 g, 20.35 mmol) and ethyl 4,4-difluoro-3- oxobutanoate (CAS registry 352-24-9) (1 .61 ml, 15.65 mmol) was treated dropwise with NaOMe soln. (30% in MeOH purchased from Aldrich) (3.88 ml, 20.35 mmol). The suspension was diluted with MeOH (4 ml) and was stirred at rt for 48 h. The reaction mixture was evaporated under reduced pressure and the residue was taken up in water , then the suspension was acidifed with acetic acid, the solid was filtered off, washed with little water, dried to afford a brown solid (986 mg, 39% yield).
HPLC RtM2 =0.34 min; ESIMS: 162 [(M+H)+].
1H NMR (400 MHz, DMSO-d6) δ ppm 1 1 .92 (br. s., 1 H) 7.09 (br. s., 1 H) 6.13 - 6.73 (m, 1 H) 5.69 (s, 1 H). d) 2-Amino-5-bromo-6-(difluoromethyl)pyrimidin-4(3H)-one
A suspension 2-amino-6-(difluoromethyl)pyrimidin-4(3H)-one (986 mg, 6.12 mmol) in AcOH (10 ml) was treated with Br2 (CAS registry 7726-95-6) (0.35 ml, 6.73 mmol) at rt, the orange solution was stirred at 90 °C for 18 h. Br2 (20 ul) was added and the mixture stirred for another 24 h. The mixture was cooled to rt and the suspension was poured into a sat. aq.
NaHC03 soln. and extracted with EtOAc, the organic layer was dried over MgS04 and concentrated in vacuo to afford a beige solid (760 mg, 52% yield).
HPLC RtM2 =0.47 min; ESIMS: 240-242 [(M+H)+].
1H NMR (400 MHz, DMSO-d6) δ ppm 1 1 .69 (br. s., 1 H) 6.61 - 7.28 (m, 3 H). e) 5-Bromo-4-chloro-6-(difluoromethyl)pyrimidin-2 -amine
A suspension of 2-amino-5-bromo-6-(difluoromethyl)pyrimidin-4(3H)-one (100 mg, 0.42 mmol) and POCI3 (0.50 ml, 5.42 mmol) was refluxed for 30 min. The formed solution was cooled to rt and poured into crushed ice. The mixture was basified and extracted with EtOAc, the organic layer was dried over MgS04 and concentrated in vacuo to afford an orange solid (76 mg, 71 % yield).
1H NMR (400 MHz, DMSO-d6) δ ppm 7.70 (br. s., 2 H) 6.76 - 7.1 1 (m, 1 H). f) 2-((2S,4S)-1 -(2-Amino-5-bromo-6-(difluoromethyl)pyrimidin-4-yl)-4-hydroxypyrrolidin- 2-yl)-3-phenyl-5-(trifluoromethyl)quinazolin-4(3H)-one
A solution of 2-((2S,4S)-4-hydroxypyrrolidin-2-yl)-3-phenyl-5-trifluoromethyl)quinazolin-4(3H)- one (1 10 mg, 0.29 mmol) and 5-bromo-4-chloro-6-(difluoromethyl)pyrimidin-2-amine (76 mg, 0.29 mmol) in n-BuOH (2.9 ml) was treated with DIPEA (0.102 ml, 0.59 mmol) and was stirred at 90 °C for 2 h. The reaction mixture was cooled, and evaporated under reduced pressure. The residue was taken up in DCM and washed with water, the organic layer was dried over MgS04 and concentrated in vacuo. The crude product was purified by flash chromatography on silica gel (cyclohexane / EtOAc 100:0 to 0:100) to provide a solid (100 mg, 57% yield).
HPLC RtM2 =1 .05 min; ESIMS: 597-599 [(M+H)+].
1H NMR (400 MHz, , DMSO-d6) δ ppm 7.75 - 7.97 (m, 4 H) 7.47 - 7.65 (m, 4 H) 6.63 - 7.07 (m, 1 H) 6.48 (br. s., 2 H) 5.25 (d, 1 H) 4.66 - 4.78 (m,1 H) 3.95 - 4.13 (m, 4 H) 3.61 - 3.84 (m, 1 H) 1 .92 - 2.05 (m, 2 H) 1 .73 - 1 .89 (m, 1 H). g) 2-Amino-4-(difluoromethyl)-6-((2S,4S)-4-hydroxy-2-(4-oxo-3-phenyl-5- (trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile
A solution of 2-((2S,4S)-1 -(2-amino-5-bromo-6-(difluoromethyl)pyrimidin-4-yl)-4- hydroxypyrrolidin-2-yl)-3-phenyl-5-(trifluoromethyl)quinazolin-4(3H)-one (100 mg, 0.67 mmol) in DMF (1 .7 ml) was treated with Zn(CN)2 (13.76 mg, 0.12 mmol), Pd2(dba)3 (13.80 mg, 0.01 mmol), and DPPF (6.50 mg, 0.01 mmol). The mixtue was stirred at 120°C for 18 h. The reaction mixture was cooled to rt, diluted with EtOAc and washed with water and brine. The organic layer was dried over MgS04 and concentrated in vacuo. The residue was purified over SFC (column NH2, 250 x 30 mm, 60A, 5μηι, Princeton, flow at 100 ml/min; gradient of
MeOH in supercritical C02, from 13% to 18% in 1 1 min) and triturated with pentane to afford the title compound as a yellow solid (61 mg, 67% yield).
HPLC RtM2 =1 .00 min; ESIMS: 544 [(M+H)+].
1H NMR (400 MHz, DMSO-d6) δ ppm 7.79 - 7.98 (m, 4 H) 7.44 - 7.65 (m, 5 H) 7.10 (br. s., 1 H) 6.56 - 6.91 (m, 1 H) 5.13 - 5.37 (m, 1 H) 4.67 (br. s., 1 H) 4.10 - 4.28 (m, 2 H) 3.70 (br. s., 1 H) 1 .83 - 2.08 (m, 2 H).
Examples N1 to N8: The compounds listed in Table 1 1 were prepared by a procedure analogous to that used in Example A1 , using (2S,4S)-1 -(tert-butoxycarbonyl)-4- fluoropyrrolidine-2-carboxylic acid (intermediate IA8) as precursor.
Table 1 1
General procedures used for preparation of table examples:
CA) Cyclization, using POPh3
At rt, a dry solution of amino acid derivative (1 eq.) and anthranilic acid derivative (1 eq.) in pyridine (0.83 M) was treated with triphenylphosphite (2.5 eq.) and the reaction mixture was stirred at 70 C for 2 to 18 h, then treated with the amino or hydrazine derivative. The reaction mixture was stirred at 70°C for 2 to 18 h.
CB) Deprotection conditions
CB1 ) Using TFA
At rt, a dry solution of Boc intermediate (1 eq.) in DCM (0.1 to 0.25 M) was treated with TFA (10 eq.) and the reaction mixture was stirred at rt for 1 to 18 h.
CB2) Using HCI
At rt, a dry solution of Boc derivative (1 eq.) in THF or DCM (0.25 to 0.55 M) was treated with a 4M HCI in dioxane soln.. The reaction mixture was stirred at rt for 2.5 h to 3 d.
CB3) Using H3P04
At rt, a dry solution of amino acid derivative (1 eq.) in THF (0.6M) was treated with aq. H3P04 soln. (85 wt% purchased from Aldrich) (same volume than solvent). The reaction mixture was stirred at rt for 24 h to 5 d.
CC) Coupling of R3 moiety
At rt, a solution of deprotected intermediate (1 eq.) and an aryl halogenide (1 to 2.5 eq.) in alcohol (nBuOH or EtOH, 0.08M to 0.1 M) was treated with DIPEA (2 to 5 eq.) and irridiated in a microwave reactor or oil bath at 120-160°C for 0.5 h to 6 d.
CD) Chromatographic conditions for separation of diastereomeric atropisomers
Method# Column Eluent UV Detection
CD1 Waters Sunfire Solvent A: Water + 0.1 % 216 nm
C18, 5 urn, 30x100 TFA; Solvent B: Acetonitrile
mm + 0.1 % TFA, gradient (%B):
20-40% in 16 min
CD2 Waters Sunfire Solvent A: Water + 0.1 % 216 nm
C18, 5 urn, 30x100 TFA; Solvent B:
mm Acetonitrile, gradient (%B):
30-50% in 16 min
CD3 Waters Sunfire Solvent A: Water + 0.1 % 216 nm
C18, 5 urn, 30x100 TFA; Solvent B:
mm Acetonitrile, gradient (%B):
15-35% in 16 min
CD4 Sunfire ACN + 0.1 % TFA / H2O + 254 nm
100x30 mm, 5μηι 0.1 % TFA, 20 to 50% from
ACN over 20 min
CD5 Chiralcel OZ-H Heptane/EtOH/MeOH 220 nm
50/25/25
CD6 Phenomenex Luna A: 0.1 % TFA in H20, B: 200-400 nm
C-18(2) 250x21.2 ACN; gradient (t[min]/%B):
mm, 5μηι : 0/50, 10/90, 30/90,
30.1/50
CD7 Phenomenex Luna A: 0.1 % TFA in H20, B: 200-400 nm
C-18(2) 250x21.2 ACN; gradient (t[min]/%B):
mm, 5 μηι : 0/30, 10/70, 15/90, 30/90,
30.1/30
CD8 Phenomenex Luna A: 0.1 % TFA in H20, B: 200-400 nm
C-18(2) 250x21.2 ACN gradient (t[min]/%B): :
mm, 5μηι 0/70, 10/70, 15/90, 30/90,
30.1/70
CD9 Phenomenex Luna A: 0.1 % TFA in H20, B: 200-400 nm
C-18(2) 250x21.2 ACN; gradient (t[min]/%B):
mm, 5μηι : 0/40, 10/70, 15/90, 30/90,
30.1/40
CD10 Chiralpak As-H, 20 Heptane / iPrOH 60/40 210 nm
um 50x 5 cm
CD1 1 Chiralpak AD, 20 Heptane / EtOH50/50 210 nm
um 76.5 x 393 mm
CE) Oxidation:
A solution of methylthio derivative (1 eq.) in dioxane / H20 (3 :1 , 0.02M) was treated with oxone (2 eq to 3.5 eq.). The reaction mixture was stirred at rt 18 h.
CF) Amination:
A solution of sulfone derivative (1 eq.) in DMSO (0.08 M) was treated with a 32% aq. NH4OH soln.(14 eq.). The reaction mixture was stirred in a sealed vial at 140°C for 18 h to 6 d. CG) Alkylation:
A suspension of the corresponding amino-purine derivative (1 eq.) in dry DMF (0.05 M) was treated with K2C03 (3 eq.). Then MS 4A and iodomethane (1 .5 eq.) were added and the suspension was stirred for 1 .5 h at rt.
Intermediates for table examples
IA) Amino Acid Derivatives
Intermediate # Structure Name CAS # or comment on synthesis
IA1 (S)-1 -(tert- 15761 -39-4
Butoxycarbonyl)p
yrrolidine-2-
Boc carboxylic acid
IA2 (R)-3-(tert- 51077-16-8
ΗΟΟΟ^,1( Butoxycarbonyl)- thiazolidine-4-
R ,N
Boc J5 carboxylic acid
IA3 (2S,4S)-1 -(tert- 83623-93-2
HOOCL .v. Butoxycarbonyl)-
4- Boc methoxypyrrolidin
e-2-carboxylic
acid
IA4 (rac)-1 -(tert-
Hoo _ Butoxycarbonyl)-
2 ste s from CAS 4,5-dihydro-1 H- 96-33-3
pyrazole-5- carboxylic acid
IA5 (2S,4S)-1 -(tert- 2 steps from CAS
HOGCL Butoxycarbonyl)- 121 148-01 -4
-NHS02 e 4-(methylsulfon-
Boc' amido)pyrrolidine- 2-carboxylic acid
IA6 (S)-2-(tert- 1372202-46-4
Butoxycarbonyl)is
IA7 (2S,4S)-1 -(tert- 401564-17-8
HOOCL Butoxycarbonyl)-
! >-«'OTBDMS 4-((tert-
Boc butyldimethylsilyl)
oxy)pyrrolidine-2- carboxylic acid
IA8 (2S,4S)-1 -(tert- 203866-13-1
HOCX 5l butoxycarbonyl)-
4- Boc fluoropyrrolidine- 2-carboxylic acid
Intermediate IA4: 1 -(tert-Butoxycarbonyl)-4,5-dihydro-1 H-pyrazole-5-carboxylic acid a) 1 -tert-butyl 5-methyl 4,5-dihydro-1 H-pyrazole-1 ,5-dicarboxylate
A solution of methyl acrylate (CAS registry 96-33-3) (1 g, 1 1 .62 mmol) in hexane/toluene (29 ml / 29 ml) was treated with 2M trimethylsilyldiazomethane solution in diethyl ether (8.71 ml, 17.42 mmol) . The resulting yellow solution was stirred at 23°C for 18 h.
The mixture was concentrated in vacuo (water bath below 40°C) then diluted with DCM (0.1 M, 29 ml) and treated with TFA (2.68 ml, 34.8 mmol) at 0°C, the resulting orange solution was stirred at 0°C for 2 h, then evaporated under reduced pressure (water bath below 40°C). The resulting orange oil was dissolved in DCM (40 ml), treated with Et3N (6.48 ml, 46.5 mmol) at 0°C, then with DMAP (71 mg, 0.58 mmol) and Boc20 (3.04 g, 13.94 mmol). The orange solution was stirred at rt for 18 h, then Et3N (1 ml) and Boc20 (1 g) were added and the solution was stirred for another 24 h. The reaction was quenched with water and extracted with DCM, the organic layer was dried by passing through a phase separating cartridge and evaporated in vacuo to afford an orange oil. The crude product was purified by chromatography on silica gel, (cyclohexane / DCM 55:45 to 0:100) to provide the title compound as a yellow oil (573 mg, 38% yield).
HPLC RtM2= 0.73 min; ESIMS: 229 [(M+H)+].
1H NMR (400 MHz, DMSO-d6): δ 6.97 (s, 1 H) 4.62 (dd, 1 H) 3.66 (s, 3 H) 3.23 (dd, 1 H) 2.93 (m, 1 H) 1 .39 (s 9 H).
b) 1 -(tert-Butoxycarbonyl)-4,5-dihydro-1 H-pyrazole-5-carboxylic acid
A colorless solution of 1 -tert- butyl 5-methyl 4,5-dihydro-1 H-pyrazole-1 ,5-dicarboxylate (573 mg, 2.51 mmol) in ACN (6.38 ml) was treated with a LiOH soln. (1 M in water, 1 eq.) and stirred at rt for 1 h. The reaction mixture was evaporated under reduced pressure (water bath below 40°C) and slowly acidified until pH=3 with an aq. 1 N HCI soln. and extracted with EtOAc, the organic layer was dried over MgS04, concentrated under reduced pressure to afford the title compound as a yellow oil (EtOAc not fully removed, product may be volatile) (250 mg, 97% yield, crude).
HPLC RtM2= 0.50 min; ESIMS: 215 [(M+H)+].
1H NMR (400 MHz, DMSO-d6): δ 12.92 (br s, 1 H) 6.94 (s, 1 H) 4.49 (dd, 1 H) 3.30 - 2.81 (m, 2H) 1 .40 (m, 9H).
Intermediate IA5: (2S,4S)-1 -(tert-Butoxycarbonyl)-4-(methylsulfonamido)pyrrolidine-2- carboxylic acid
a) (2S,4S)-1 -tert-butyl 2-methyl 4-(methylsulfonamido)pyrrolidine-1 ,2-dicarboxylate
A mixture of (2S,4S)-1 -tert-butyl 2-methyl 4-aminopyrrolidine-1 ,2-dicarboxylate (CAS registry 121 148-01 -4) (250 mg, 1 .02 mmol) in DMF (5 ml) was dropwise treated with Et3N (285 uL, 2.05 mmol) at 0°C. The resulting mixture was stirred for 5 min at 0°C then methanesu!fonyl chloride (CAS registry 124-63-0) (80 uL, 1 .02 mmol) was added. The solution became orange and the temperature was allowed to reach rt. Stirring was continued at rt for 4 d. The reaction mixture was washed with sat. aq.NaHC03 soln. and extracted with DCM, the organic layer was dried by passing it through a phase separating cartridge and was evaporated in vacuo to afford a yellow oil (282 mg, 85% yield, crude).
1H NMR (400 MHz, DMSO-d6): δ 7.42 - 7.32 (m, 1 H) 4.15 (t, 1 H) 3.95 - 3.80 (m, 1 H) 3.71 (dd, 1 H) 3.676 - 3.56 (m, 3 H) 3.12 - 3.00 (m, 1 H) 2.60 - 2.53 (m, 1 H) 1 .84 - 1 .66 (m, 1 H) 1 .42 - 1 .26 (m, 9 H). b) (2S,4S)-1 -(tert-Butoxycarbonyl)-4-(methylsulfonamido)pyrrolidine-2-carboxylic acid A colorless solution of (2S,4S)-1 -tert-butyl 2-methyl 4-(methylsulfonamido)pyrrolidine-1 ,2- dicarboxylate (282 mg, 0.88 mmol) in ACN (2.5 ml) was treated with a LiOH soln. (1 M in water, 1 .3 eq.) and stirred at rt for 18 h. The reaction mixture was slowly acidified until pH=3 with an aq. 1 N HCI soln. (1 ml) and extracted with EtOAc, the organic layer was dried over MgS04, concentrated under reduced pressure to afford the title compound as a colorless oil (218 mg, 81 %, crude).
1H NMR (400 MHz, DMSO-d6): δ 12.64 (br s, 1 H) 7.38 (br s, 1 H) 3.84 (br s, 1 H) 3.76 - 3.56 (m, 1 H) 3.12 - 2.98 (m, 1 H), 2.93 (s, 3H) 1 .83 - 1 .62 (m, 1 H) 1 .46 - 1 .26 (m, 9H).
B) Anthranilic Acid Derivatives
Intermediate IB7: 6-Amino-3-fluoro-2-(trifluoromethyl)benzoic acid
a) 3-Fluoro-6-nitro-2-(trifluoromethyl)benzoic acid
At 0°C, a solution of 3-fluoro-2-(trifluoromethyl)benzoic acid (CAS registry 261951 -80-8) (800 mg, 3.77 mmol) in concentrated sulfuric acid (95 %) (2.6 ml, 49 mmol) was dropwise treated with fuming nitric acid (90 %) (0.23 ml, 4.52 mmol) over 15 min. The reaction mixture was stirred at 0°C for 4 h.
The mixture was poured onto ice water and stirred for 10 min. The resulting mixture was then diluted with water and DCM. The organic layer was dried by passing through a phase
separating cartridge and evaporated in vacuo to afford a white solid (904 mg, 94 % yield, crude).
HPLC RtM2= 0.35 min; ESIMS: 252 [(M-H)-].
1H NMR (400 MHz, DMSO-d6): δ 14.76 (br s, 1 H), 8.60 (dd, 1 H), 7.89 (t, 1 H). b) 6-Amino-3-fluoro-2-(trifluoromethyl)benzoic acid
A pale yellow solution of 3-fluoro-6-nitro-2-(trifluoromethyl)benzoic acid (900 mg, 3.56 mmol) in THF (20 ml) was successively degassed and purged with nitrogen (5x), then the solution was treated with 10 % palladium on carbon (200 mg, 0.19 mmol). The black mixture was purged, then backfilled with hydrogen gas and allowed to stir at rt for 18 h.
The reaction mixture was filtered through hyflo and the pad was rinsed with EtOAc (2x). The collected filtrates were evaporated in vacuo to afford a pale brown solid (847 mg, 91 % yield, crude).
HPLC RtM2= 0.56 min; ESIMS: 222 [(M-H)-].
1H NMR (400 MHz, DMSO-d6): δ 7.23 (t, 1 H), 7.01 (dd, 1 H).
Intermediate IB8: 2-Amino-6-(difluoromethoxy)benzoic acid
a) Methyl 2-hydroxy-6-nitrobenzoate
A yellow stirred solution of methyl 2-methoxy-6-nitrobenzoate (CAS registry 77901 -52-1) (650 mg, 2.99 mmol) in DCM (15 ml) at 0°C was dropwise treated with BBr3 (1 M in DCM ) (12 ml_, 1 1 .94 mmol). The resulting mixture was stirred for 1 h at rt. It was poured into crushed ice, the organic layer was separated and the aqueous layer was extracted with EtOAc and dried over MgS04, evaporated under reduced pressure to afford a black residue (750 mg).The crude product was purified by chromatography on silica gel (cyclohexane/ EtOAc, 0% to 60% ) to provide the title compound as a red residue (0.250 g, 42% yield). HPLC RtM2= 0.75 min ; ESIMS: 196 [(M-H)+].
1H NMR (400 MHz, DMSO-c/6): δ 10.99 (s, 1 H), 7.63 (dd, 1 H), 7.53 (t, 1 H), 7.33 (dd, 1 H), 3.81 (s, 3H). b) Methyl 2-(difluoromethoxy)-6-nitrobenzoate
An orange solution of methyl 2-hydroxy-6-nitrobenzoate (224 mg, 1 .14 mmol) and KOH (1 .27 g, 22.72 mmol) in ACN/Water (5.7 ml/5.7 mL) at 0°C was treated with diethyl
(bromodifluoromethyl)phosphonate (CAS registry 65094-22-6) (607 mg, 2.27 mmol) and the biphasic mixture was allowed to reach rt for 15 min. The reaction mixture was diluted with Et20. The layers were separated and the aqueous layer was further extracted with Et20. The combined extracts were dried by passing through a phase separating cartridge, and evaporated under reduced pressure to afford a green oil (290mg, crude).
HPLC Rt 2= 0.95 min
1H NMR (400 MHz, DMSO-d6): δ 8.13 (dd, 1 H), 7.84 (d, 1 H), 7.80 - 7.89 (m, 1 H), 7.39 (t, 1 H), 3.88 (s, 3 H). c) 2-(Difluoromethoxy)-6-nitrobenzoic acid
A yellow solution of methyl 2-(difluoromethoxy)-6-nitrobenzoate (294 mg, 1 .17 mmol) in ACN (3ml_) was treated with LiOH (1 M in water) (1 eq.) and the mixture was stirred at rt for 1 h. Then MeOH (10 mL) and 4N aq. NaOH soln. (5ml_) were added, the mixture was stirred at rt for 2 d. It was evaporated to dryness and then slowly acidified with aq. HC1 1 N soln. until pH 1 and extracted with EtOAc, the organic layer was dried over MgS04, concentrated under reduced pressure to afford an orange oil (374 mg). The crude product was purified by chromatography on silica gel (DCM/MeOH, 0% to 20%) to provide the title compound as a yellow oil (290 mg, 80% yield).
HPLC RtM2= 0.38 min c) 2-amino-6-(difluoromethoxy)benzoic acid
A yellow solution of 2-(difluoromethoxy)-6-nitrobenzoic acid (210 mg, 0.90 mmol) in MeOH (5 mL) was treated with Pd/C 10% (36 mg) and the mixture was hydrogenated at rt for 0.5 h at 0.1 bar. The mixture was filtrated through hyflo and the pad was rinsed with MeOH, concentration under reduced pressure afforded an orange residue (165 mg, crude).
HPLC RtM2= 0.64 min ; ESIMS: 202 [(M-H)+].
IC) Amines or Hydrazines
Intermediate # Structure Name CAS # or comment on synthesis
IC3 1 -Methyl-1 H-pyrazol-5- 1 192-21 -8
IC6 2-Cyclopropylaniline 3158-73-4
IC8 Morpholin-4-amine 4319-49-7
IC9 Pyridin-3-amine 462-08-8
IC10 6-Methoxypyridin-3-amine 6628-77-9
IC11 1 -Methylcyclopropan- 22936-83-0 amine
H2N^^
IC12 Cyclopropanamine 765-30-0
ID) R3 Precursors
Intermediate # Structure Name CAS # or comment on synthesis
ID1 6-Chloro-9H-purine 87-42-3
N
H *N— /■*/
ID2 6-Chloro-9H-purin-2- 10310-21 -1
ID3 6-Chloro-9-methyl-9H- 3035-73-2
ID4 4-Amino-6- 60025-09-4
Av01 chloropyrimidine-5- carbonitrile
NH2
ID5 2-Amino-4-chloro-6- 99586-66-0
methylpyrimdine-5- carbonitrile
ID6 7-Chlorothiazolo[5,4- 13316-12-6
ID7 7-Chloro-5- 1028332-36-6
CI
(methylthio)thiazolo[5,4- d]pyrimidine
Synthesis of 4-Chloro-2-(methylthio)imidazor2,1 -flM ,2,41triazine
a) Ethyl 1 -(3-benzoylthioureido)-1 H-imidazole-2-carboxylate
A solution of ethyl 1 -amino-1 H-imidazole-2-carboxylate (CAS registry 1008130-15-1 ) (180 mg, 1 .16 mmol) in THF (4 ml) was treated with a solution of benzoyl isothiocyanate (CAS registry 532-55-8) (0.16 ml, 1 .16 mmol) in THF (1 ml). The reaction mixture was stirred at rt for 3 d. The reaction mixture was concentrated under reduced pressure, the resulting residue was taken up in Et20 (1 .5 ml), then stirred for 0.5 h. The formed solid was filtrered off, washed with a mixture of heptane / Et20 9:1 and dried under high vacuum to afford the title compound (379 mg, 100% yield).
HPLC 0.82 min; ESIMS: 319 [(M+H)+].
1H NMR (400 MHz, DMSO-c/6) δ 13.02 (s, 1 H), 12.05 (s, 1 H), 7.87 - 8.10 (m, 2 H), 7.67 (s, 1 H), 7.47 - 7.58 (m, 3 H), 7.14 (d, J=1 .00 Hz, 1 H), 7.14 (d, J=1 .00 Hz, 1 H), 4.21 (q, J=7.03 Hz, 2 H), 1 .21 (t, J=7.03 Hz, 3 H). b) 2-Thioxo-2,3-dihydroimidazo[2,1 -f][1 ,2,4]triazin-4(1 H)-one
A solution of ethyl 1 -(3-benzoylthioureido)-1 H-imidazole-2-carboxylate (373 mg, 1 .17 mmol) in 2M aq. NaOH soln. was stirrred at 85°C for 75 min. The reaction mixture was allowed to cool down to rt and was treated with EtOH (0.93 ml), then cooled down to 0°C and treated with acetic acid (283 μΙ_, 4.9 mmol.). The formed precipitate was stirred at 0°C for 0.5 h, filtered off, washed with cold EtOH and dried under high vacuum to afford the title compound (124 mg, 63% yield).
HPLC 0.21 min; ESIMS: 169 [(M+H)+].
1H NMR (400 MHz, DMSO-c/6) δ 1 1 .31 (br s, 1 H), 7.65 (d, J=0.75 Hz, 1 H), 7.52 (s, 1 H). c) 2-(Methylthio)imidazo[2,1 -f][1 ,2,4]triazin-4(3H)-one
A suspension of 2-thioxo-2,3-dihydroimidazo[2,1 -f][1 ,2,4]triazin-4(1 H)-one (123 mg, 0.731 mmol) in THF (3.5 ml) was treated with iodomethane (59 μΙ_, 0.951 mmol). The reaction mixture was stirred at 45°C for 1 .5 h. The reaction was concentrated under reduced pressure, the colorless residue was taken up with H20 (3.5 ml) and a sat. aq. NaHC03 soln. (3.5 ml), stirred for 0.5 h and lyophilized. The colorless residue was taken in a small amount of H20, filtrered off, washed with water and dried under high vacuum to afford the title compound as a colorless solid (94 mg, 70% yield).
HPLC 0.47 min; ESIMS: 183 [(M+H)+].
1H NMR (400 MHz, DMSO-c/6) δ 7.45 (s, 1 H), 7.09 (s, 1 H), 2.32 (s, 3 H). d) 4-Chloro-2-(methylthio)imidazo[2,1 -f][1 ,2,4]triazine
A solution of 2-(methylthio)imidazo[2,1 -f][1 ,2,4]triazin-4(3H)-one (91 mg, 0.5 mmol) in POCI3 was stirred at 100°C for 21 h. The reaction mixture was concentrated under reduced pressure. Ice water and EtOAc were added to the residue, the organic phase was washed with a sat. aq. NaHC03 soln. and brine. The combined aq. phases were back-extracted with EtOAc, the combined organic phases were dried over Na2S04, concentrated under reduced pressure to afford the tiltle compound as brown solid (59 mg, 59% yield).
HPLC RtMi = 0.76 min; ESIMS: 201 [(M+H)+].
1H NMR (400 MHz, DMSO-c/6) δ 8.50 (d, J=1 .00 Hz, 1 H), 7.94 (d, J=1 .00 Hz, 1 H), 2.57 (s, 3 H).
Biological evaluation
The activity of a compound according to the present invention can be assessed by the following in vitro & in vivo methods.
Biological assays
1 Determination of enzymatic PI3K alpha, PI3Kbeta, PI3Kgamma and PI3K delta isoform inhibition
1.1 Biochemical assays for PI3Kalpha, PI3Kbeta
The efficacy of the compounds of examples 1 -1 17 as PI3 kinase inhibitors can be demonstrated as follows:
The kinase reaction is performed in a final volume of 50 μΙ per well of a half area
COSTAR, 96 well plate. The final concentrations of ATP and phosphatidyl inositol in the assay are 5 μΜ and 6 μg/mL, respectively. The reaction is started by the addition of PI3 kinase, e.g. PI3 kinase δ.
ρ110δ. The components of the assay are added per well as follows:
· 10 μΙ test compound in 5% DMSO per well in columns 2-1 .
• Total activity is determined by addition 10 μΙ of 5% vol/vol DMSO in the first 4 wells of column 1 and the last 4 wells of column 12.
• The background is determined by addition of 10 μΜ control compound to the last 4 wells of column 1 and the first 4 wells of column 12.
· 2 mL 'Assay mix' are prepared per plate:
1 .912 mL of HEPES assay buffer
8.33 μΙ of 3 mM stock of ATP giving a final concentration of 5 μΜ per well
1 μΙ of [33P]ATP on the activity date giving 0.05 μθί per well
30 μΙ of 1 mg/mL PI stock giving a final concentration of 6 μg/mL per well 5 μΙ of 1 M stock MgCI2 giving a final concentration of 1 mM per well
• 20 μΙ of the assay mix are added per well.
• 2 mL 'Enzyme mix' are prepared per plate (x* μΙ PI3 kinase p1 10β in 2 mL of kinase buffer). The 'Enzyme mix' is kept on ice during addition to the assay plates.
20 μΙ 'Enzyme mix' are added/well to start the reaction.
The plate is then incubated at room temperature for 90 minutes.
The reaction is terminated by the addition of 50 μΙ WGA-SPA bead (wheat germ agglutinin-coated Scintillation Proximity Assay beads) suspension per well.
The assay plate is sealed using TopSeal-S (heat seal for polystyrene microplates, PerkinElmer LAS [Deutschland] GmbH, Rodgau, Germany) and incubated at room temperature for at least 60 minutes.
The assay plate is then centrifuged at 1500 rpm for 2 minutes using the Jouan bench top centrifuge (Jouan Inc., Nantes, France).
The assay plate is counted using a Packard TopCount, each well being counted for 20 seconds.
* The volume of enzyme is dependent on the enzymatic activity of the batch in use.
In a more preferred assay, the kinase reaction is performed in a final volume of 10 μΙ per well of a low volume non-binding CORNING, 384 well black plate (Cat. No. #3676). The final concentrations of ATP and phosphatidyl inositol (PI) in the assay are 1 μΜ and 10 μg/mL, respectively. The reaction is started by the addition of ATP.
The components of the assay are added per well as follows:
50 nl test compounds in 90% DMSO per well, in columns 1 -20, 8 concentrations (1/3 and 1/3.33 serial dilution step) in single.
• Low control: 50 nl of 90% DMSO in half the wells of columns 23-24 (0.45% in final).
High control: 50 nl of reference compound (e.g. compound of Example 7 in WO 2006/122806) in the other half of columns 23-24 (2.5 μΜ in final).
Standard: 50 nl of reference compound as just mentioned diluted as the test compounds in columns 21 -22.
20 mL 'buffer' are prepared per assay :
200 μΙ of 1 M TRIS HCI pH7.5 (10 mM in final)
60 μΙ of 1 M MgCI2 (3 mM in final)
500 μΙ of 2M NaCI (50 mM in final)
100 μΙ of 10% CHAPS (0.05% in final)
200 μΙ of 10OmM DTT (1 mM in final)
18.94 mL of nanopure water
10 mL 'PI' are prepared per assay :
200 μΙ of 1 mg/mL l-alpha-Phosphatidylinositol (Liver Bovine, Avanti Polar Lipids Cat. No. 840042C MW=909.12) prepared in 3% OctylGlucoside (10 μg/mL in final)
9.8 mL of 'buffer'
10 mL 'ATP' are prepared per assay :
6.7 μΙ of 3 mM stock of ATP giving a final concentration of 1 μΜ per well 10 mL of 'buffer'
2.5 mL of each PI3K construct are prepared per assay in 'PI' with the following final concentration :
10 nM PI3K alfa EMV B1075
25 nM beta EMV BV949
10 nM delta EMV BV1060
150 nM gamma EMV BV950 · 5 μΙ of 'PI/PI3K' are added per well.
5 μΙ 'ATP' are added per well to start the reaction.
The plates are then incubated at room temperature for 60 minutes (alfa, beta, delta) or 120 minutes (gamma).
The reaction is terminated by the addition of 10 μΙ Kinase-Glo (Promega Cat. No. #6714).
The assay plates are read after 10 minutes in Synergy 2 reader (BioTek, Vermont USA) with an integration time of 100 milliseconds and sensitivity set to 191 .
Output : The High control is around 60Ό00 counts and the Low control is 30Ό00 or lower
· This luminescence assay gives a useful Z' ratio between 0.4 and 0.7
The Z' value is a universal measurement of the robustness of an assay. A Z' between 0.5 and 1 .0 is considered an excellent assay.
For this assay, the PI3K constructs mentioned are prepared as follows:
1.2 Generation of gene constructs
Two different constructs, BV 1052 and BV 1075, are used to generate the PI3 Kinase a proteins for compound screening. PI3Ka BV-1052 p85(iSH2)-Glv Iinker-p1 10a(D20aa)-C-term His tag
PCR products for the inter SH2 domain (iSH2) of the p85 subunit and for the p1 10-a subunit (with a deletion of the first 20 amino acids) are generated and fused by overlapping PCR.
The iSH2 PCR product is generated from first strand cDNA using initially primers gwG130-p01 (5'-CGAGAATATGATAGATTATATGAAGAAT-3') (SEQ ID NO: 1) and gwG130-p02 (5'-TGGTTT-AATGCTGTTCATACGTTTGTCAAT-3') (SEQ ID NO: 2). Subsequently in a secondary PCR reaction, Gateway (Invitrogen AG, Basel,
Switzerland) recombination AttB1 sites and linker sequences are added at the 5'end and 3'end of the p85 iSH2 fragment respectively, using primers
gwG130-p03 (5'-
GGGACAAGTTTGTACAAAAAAGCAGGCTACGAAGGAGATATACATAT- GCGAGAATATGATAGATTATATGAAGAAT -3') (SEQ ID NO: 3) and
gwG152-p04 (5'- TACCATAATTCCACCACCACCACCGGAAATTCCCCCTGGTTT- AATGCTGTTCATACGTTTGTCAAT-3') (SEQ ID NO: 4).
The p1 10-a fragment is also generated from first strand cDNA, initially using primers gwG152-p01 (5'- CTAGTGGAATGTTTACTACCAAATGG-3') (SEQ ID NO: 5) and gwG152-p02 (5'- GTTCAATG-CATGCTGTTTAATTGTGT -3') (SEQ ID NO: 6).
In a subsequent PCR reaction, linker sequence and a Histidine tag are added at the 5'end and 3'end of the p1 10-a fragment respectively, using primers
gw152-p03 (5'-GGGGGAATTTCCGGTGGTGGTGGTGGAATTATGGTAC- TAGTGGAATGTTTACTACC-AAATGGA-3') (SEQ ID NO: 7) and
gwG152-p06 (5'-AGCTCCGTGATGGTGATGGTGATGTGCTCCGTTCAATG- C ATG CTGTTTAATTGTGT- 3 ') (SEQ ID NO: 8).
The p85-iSH2/p1 10-a fusion protein is assembled in a third PCR reaction by the overlapping linkers at the 3'end of the iSH2 fragment and the 5'end of the p1 10-a fragment, using the above mentioned gwG130-p03 primer and a primer containing an overlapping Histidine tag and the AttB2 recombination sequences
(5 -
GGGACCACTTTGTACAAGAAAGCTGGGTTTAAGCTCCGTGATGGTGATGGTGAT- GTGCTCC-3') (SEQ ID NO: 9).
This final product is recombined in a (Invitrogen) OR reaction into the donor vector pDONR201 to generate the ORF318 entry clone. This clone is verified by sequencing and used in a Gateway LR reaction to transfer the insert into the Gateway adapted pBlueBac4.5 (Invitrogen) vector for generation of the baculovirus expression vector LR410.
PI3Ka BV-1075 p85(iSH2)-12 XGIv Iinker-p1 10a(D20aa)-C-term His tag
The construct for Baculovirus BV-1075 is generated by a three-part ligation comprised of a p85 fragment and a p1 10-a fragment cloned into vector pBlueBac4.5. The p85 fragment is derived from plasmid p1661 -2 digested with Nhe/Spe. The p1 10-a fragment derived from LR410 (see above) as a Spel/Hindlll fragment. The cloning vector pBlueBac4.5 (Invitrogen) is digested with Nhe/Hindlll. This results in the construct PED 153.8
The p85 component (iSH2) is generated by PCR using ORF 318 (described above) as a template and one forward primer
KAC1028 (5'- GCTAGCATGCGAGAATATGATAGATTATATGAAGAATATACC) (SEQ ID NO: 10) and two reverse primers,
KAC1029 (5'- GCCTCCACCACCTCCGCCTGGTTTAATGCTGTTCATACGTTTGTC) (SEQ ID NO: 1 1) and
KAC1039 (5'-TACTAGTCCGCCTCCACCACCTCCGCCTCCACCACCTCCGCC) (SEQ ID NO: 12).
The two reverse primers overlap and incorporate the 12x Gly linker and the N-terminal sequence of the p1 10a gene to the Spel site. The 12x Gly linker replaces the linker in the BV1052 construct. The PCR fragment is cloned into pCR2.1 TOPO (Invitrogen). Of the resulting clones, p1661 -2 is determined to be correct. This plasmid is digested with Nhe and Spel and the resulting fragment is gel-isolated and purified for sub-cloning. The p1 10-a cloning fragment is generated by enzymatic digest of clone LR410 (see above) with Spe I and Hind III. The Spel site is in the coding region of the p1 10a gene. The resulting fragment is gel-isolated and purified for sub-cloning.
The cloning vector, pBlueBac4.5 (Invitrogen) is prepared by enzymatic digestion with Nhe and Hind 111. The cut vector is purified with Qiagen (Quiagen N.V, Venlo,
Netherlands) column and then dephosphorylated with Calf Intestine alkaline
phosphatase (CIP) (New England BioLabs, Ipswich, MA). After completion of the CIP reaction the cut vector is again column purified to generate the final vector. A 3 part ligation is performed using Roche Rapid ligase and the vendor specifications. PI3KB BV-949 p85(iSH2)-Glv Iinker-p1 10b(full-lenqth)-C-term His tag
PCR products for the inter SH2 domain (iSH2) of the p85 subunit and for the full-length p1 10-b subunit are generated and fused by overlapping PCR.
The iSH2 PCR product is generated from first strand cDNA initially using primers gwG130-p01 (5'-CGAGAATATGATAGATTATATGAAGAAT-3') (SEQ ID NO: 1) and gwG130-p02 (5'-TGGTTT-AATGCTGTTCATACGTTTGTCAAT-3') (SEQ ID NO: 2). Subsequently, in a secondary PCR reaction Gateway (Invitrogen) recombination AttB1
sites and linker sequences are added at the 5'end and 3'end of the p85 iSH2 fragment respectively, using primers
gwG130-p03 (5'- GGGACAAGTTTGTACAAAAAAGCAGGCTACGAAGGAGATA- TACATATGCGAGAATATGATAGATTATATGAAGAAT -3') (SEQ ID NO: 3) and gwG130-p05 (5'-ACTGAAGCATCCTCCTCCTCCTCCTCCTGGTTTAAT- GCTGTTCATACGTTTGTC-3') (SEQ ID NO: 13).
The p1 10-b fragment is also generated from first strand cDNA initially using primers gwG130-p04 (5'-
ATTAAACCAGGAGGAGGAGGAGGAGGATGCTTCAGTTTCATAATGCC-TCCTGCT - 3') (SEQ ID NO: 4)
which contains linker sequences and the 5'end of p1 10-b and
gwG130-p06 (5'-AGCTCCGTGATGGTGATGGTGATGTGCTCCAGATCTGTAGTCTTT- CCGAACTGTGTG -3') (SEQ ID NO: 14)
which contains sequences of the 3'end of p1 10-b fused to a Histidine tag.
The p85-iSH2/p1 10-b fusion protein is assembled by an overlapping PCR a reaction of the linkers at the 3'end of the iSH2 fragment and the 5'end of the p1 10-b fragment, using the above mentioned gwG130-p03 primer and a primer containing an overlapping Histidine tag and the AttB2 recombination sequences (5 - GG G AC C ACTTTGTAC AAG AAAG CTGG GTTT- AAGCTCCGTGATGGTGATGGTGATGTGCTCC-3') (SEQ ID NO: 15).
This final product is recombined in a Gateway (Invitrogen) OR reaction into the donor vector pDONR201 to generate the ORF253 entry clone. This clone is verified by sequencing and used in a Gateway LR reaction to transfer the insert into the Gateway adapted pBlueBac4.5 (Invitrogen) vector for generation of the baculovirus expression vector LR280.
PI3K6 BV-1060 p85(iSH2)-Glv Iinker-p1 10d(full-lenqth)-C-term His tag
PCR products for the inter SH2 domain (iSH2) of the p85 subunit and for the full-length p1 10-d subunit are generated and fused by overlapping PCR.
The iSH2 PCR product is generated from first strand cDNA using initially primers gwG130-p01 (5'-CGAGAATATGATAGATTATATGAAGAAT-3') (SEQ ID NO: 1) and gwG130-p02 (5'-TGGTTT-AATGCTGTTCATACGTTTGTCAAT-3') (SEQ ID NO: 2). Subsequently, in a secondary PCR reaction Gateway (Invitrogen) recombination AttB1 sites and linker sequences are added at the 5'end and 3'end of the p85 iSH2 fragment respectively, using primers
gwG130-p03 (5'- GGGACAAGTTTGTACAAAAAAGCAGGCTACGAAGGAGATATACAT- ATGCGAGAATATGATAGATTATATGAAGAAT -3') (SEQ ID NO: 3) and
gwG154-p04 (5'- TCCTCCTCCTCCTCCTCCTGGTTTAATGCTGTTCATACGTTTGTC - 3') (SEQ ID NO: 16).
The p1 10-a fragment is also generated from first strand cDNA using initially primers gwG154-p01 (5'- ATGCCCCCTGGGGTGGACTGCCCCAT -3') (SEQ ID NO: 17) and gwG154-p02 (5'- CTACTG-CCTGTTGTCTTTGGACACGT -3') (SEQ ID NO: 18).
In a subsequent PCR reaction linker sequences and a Histidine tag is added at the 5'end and 3'end of the p1 10-d fragment respectively, using primers
gw154-p03 (5'- ATTAAACCAGGAGGAGGAGGAGGAGGACCCCCTGGGGTGGAC- TGCCCCATGGA -3') (SEQ ID NO: 19) and gwG154-p06 (5'-AGCTCCGTGATGGTGAT- GGTGATGTGCT-CCCTGCCTGTTGTCTTTGGACACGTTGT -3') (SEQ ID NO: 20). The p85-iSH2/p1 10-d fusion protein is assembled in a third PCR reaction by the overlapping linkers at the 3'end of the iSH2 fragment and the 5'end of the p1 10-d fragment, using the above mentioned gwG130-p03 primer and a primer containing an overlapping Histidine tag and the Gateway (Invitrogen) AttB2 recombination sequences (5'-GGGACCACTTTGTA-CAAGAAAGCTGGGTTT-
AAGCTCCGTGATGGTGATGGTGATGTGCTCC-3') (SEQ ID NO: 21 ).
This final product is recombined in a Gateway (Invitrogen) OR reaction into the donor vector pDONR201 to generate the ORF319 entry clone. This clone is verified by sequencing and used in a Gateway LR reaction to transfer the insert into the Gateway adapted pBlueBac4.5 (Invitrogen) vector for generation of the baculovirus expression vector LR415.
PI3Kv BV-950 p1 10a(D144aaVC-term His tag
This construct is obtained from Roger Williams lab, MRC Laboratory of Molecular Biology, Cambridge, UK (November, 2003). Description of the construct in: Pacold M. E. et al. (2000) Cell 103, 931 -943.
1.3 Protein expression and purification
Methods to generate recombinant baculovirus and protein for PI3K isoforms:
The pBlue-Bac4.5 (for a, b, and d isoforms) or pVL1393 (for g) plasmids containing the different PI3 kinase genes are co-transfected with BaculoGold WT genomic DNA (BD Biosciences, Franklin Lakes, NJ, USA) using methods recommended by the vendor. Subsequently, the recombinant baculovirus obtained from the transfection is plaque- purified on Sf9 insect cells to yield several isolates expressing recombinant protein. Positive clones are selected by anti-HIS or anti-isoform antibody western. For PI3K alpha and delta isoforms, a secondary plaque-purification is performed on the first clonal virus stocks of PI3K. Amplification of all baculovirus isolates is performed at low
multiplicity of infection (moi) to generate high-titer, low passage stock for protein production. The baculoviruses are designated BV1052 (a) and BV1075 (a), BV949 (β), BV1060 (δ) and BV950 (γ).
Protein production involves infection (passage 3 or lower) of suspended Tn5
(Trichoplusia ni) or TiniPro (Expression Systems, LLC, Woodland, CA, USA) cells in protein-free media at moi of 2-10 for 39-48 hours in 2 I glass Erlenmyer flasks (1 10 rpm) or wave-bioreactors (22-25 rpm). Initially, 10 I working volume wave-bioreactors are seeded at a density of 3e5 cells/mL at half capacity (5L). The reactor is rocked at 15 rpm during the cell growth phase for 72 hours, supplemented with 5% oxygen mixed with air (0.2 I per minute). Immediately prior to infection, the wave-reactor cultures are analyzed for density, viability and diluted to approximately 1 .5e6 cell/mL. 100-500 mL of high titer, low passage virus is added following 2-4 hours of additional culture. Oxygen is increased to 35% for the 39-48 hour infection period and rocking platform rpm increased to 25. During infection, cells are monitored by Vicell viability analyzer (Beckman Coulter, Inc, Fullerton, CA, USA) bioprocess for viability, diameter and density. Nova Bioanalyzer (NOVA Biomedical Corp., Waltham, MA, USA) readings of various parameters and metabolites (pH, 02 saturation, glucose, etc.) are taken every 12-18 hours until harvest. The wave-bioreactor cells are collected within 40 hours post infection. Cells are collected by centrifugation (4 degrees C at 1500 rpm), and subsequently maintained on ice during pooling of pellets for lysis and purification. Pellet pools are made with small amounts of cold, un-supplemented Grace's media (w/o protease inhibitors).
PI3K alpha Purification Protocol For HTS (BV1052)
PI3K alpha is purified in three chromatographic steps: immobilized metal affinity chromatography on a Ni Sepharose resin (GE Healthcare, belonging to General Electric Company, Fairfield, CT, USA), gel filtration utilizing a Superdex 200 26/60 column (GE Healthcare), and finally a cation exchange step on a SP-XL column (GE Healthcare). All buffers are chilled to 4°C and lysis is performed chilled on ice. Column fractionation is performed rapidly at room temperature.
Typically frozen insect cells are lysed in a hypertonic lysis buffer and applied to a prepared IMAC column. The resin is washed with 3-5 column volumes of lysis buffer, followed by 3-5 column volumes wash buffer containing 45 mM imidazole, and the target protein is then eluted with a buffer containing 250 mM imidazole. Fractions are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing target protein are pooled and applied to a prepared GFC column. Fractions from the GFC column are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing target protein are pooled. The pool from the GFC column is diluted into a low salt buffer and applied to
a prepared SP-XL column. The column is washed with low salt buffer until a stable A280 baseline absorbance is achieved, and eluted using a 20 column volume gradient from 0 mM NaCI to 500 mM NaCI. Again, fractions from the SP-XL column are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing the target protein are pooled. The final pool is dialyzed into a storage buffer containing 50% glycerol and stored at -20°C. The final pool is assayed for activity in a phosphoinosititol kinase assay.
PI3K beta Purification Protocol For HTS (BV949)
PI3K beta is purified in two chromatographic steps: immobilized metal affinity chromatography (IMAC) on a Ni Sepharose resin (GE Healthcare) and gel filtration
(GFC) utilizing a Superdex 200 26/60 column (GE Healthcare). All buffers are chilled to 4°C and lysis is performed chilled on ice. Column fractionation is performed rapidly at room temperature.
Typically frozen insect cells are lysed in a hypertonic lysis buffer and applied to a prepared IMAC column. The resin is washed with 3-5 column volumes of lysis buffer, followed by 3-5 column volumes wash buffer containing 45 mM imidazole, and the target protein is then eluted with a buffer containing 250 mM imidazole. Fractions are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing target protein are pooled and applied to a prepared GFC column. Fractions from the GFC column are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing target protein are pooled. The final pool is dialyzed into a storage buffer containing 50% glycerol and stored at -20°C. The final pool is assayed for activity in the phosphoinostitol kinase assay. PI3K gamma Purification Protocol For HTS (BV950)
PI3K gamma is purified in two chromatographic steps: immobilized metal affinity chromatography (IMAC) on a Ni Sepharose resin (GE Healthcare) and gel filtration (GFC) utilizing a Superdex 200 26/60 column (GE Healthcare). All buffers are chilled to 4°C and lysis is performed chilled on ice. Column fractionation is performed rapidly at room temperature. Typically frozen insect cells are lysed in a hypertonic lysis buffer and applied to a prepared IMAC column. The resin is washed with 3-5 column volumes of lysis buffer, followed by 3-5 column volumes wash buffer containing 45 mM imidazole, and the target protein is then eluted with a buffer containing 250 mM imidazole.
Fractions are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing target protein are pooled and applied to a prepared GFC column. Fractions from the GFC column are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing target protein are pooled. The final pool is dialyzed into a storage buffer
containing 50% glycerol and stored at -20°C. The final pool is assayed for activity in the phosphoinostitol kinase assay.
PI3K delta Purification Protocol For HTS (BV1060)
PI3K delta is purified in three chromatographic steps: immobilized metal affinity chromatography on a Ni Sepharose resin (GE Healthcare), gel filtration utilizing a Superdex 200 26/60 column (GE Healthcare), and finally a anion exchange step on a Q- HP column (GE Healthcare). All buffers are chilled to 4°C and lysis is performed chilled on ice. Column fractionation is performed rapidly at room temperature. Typically frozen insect cells are lysed in a hypertonic lysis buffer and applied to a prepared IMAC column. The resin is washed with 3-5 column volumes of lysis buffer, followed by 3-5 column volumes wash buffer containing 45 mM imidazole, and the target protein is then eluted with a buffer containing 250 mM imidazole. Fractions are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing the target protein are pooled and applied to a prepared GFC column. Fractions from the GFC column are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing the target protein are pooled. The pool from the GFC column is diluted into a low salt buffer and applied to a prepared Q-HP column. The column is washed with low salt buffer until a stable A280 baseline absorbance is achieved, and eluted using a 20 column volume gradient from 0 mM NaCI to 500 mM NaCI. Again, fractions from the Q-HP column are analyzed by Coomassie stained SDS-PAGE gels, and fractions containing the target protein are pooled. The final pool is dialyzed into a storage buffer containing 50% glycerol and stored at -20°C. The final pool is assayed for activity in the phosphoinostitol kinase assay.
IC50 is determined by a four parameter curve fitting routine that comes along with "excel fit". A four parameter logistic equation is used to calculate IC50 values (IDBS XLfit) of the percentage inhibition of each compound at 8 concentrations (usually 10, 3.0, 1 .0, 0.3, 0.1 , 0.030, 0.010 and 0.003 μΜ). Alternatively, IC50 values are calculated using idbsXLfit model 204, which is a 4 parameter logistic model.
Yet alternatively, for an ATP depletion assay, compounds of the formula (I) to be tested are dissolved in DMSO and directly distributed into a white 384-well plate at 0.5 μΙ per well. To start the reaction, 10 μΙ of 10 nM PI3 kinase and 5 μg/mL 1 -alpha- phosphatidylinositol (PI) are added into each well followed by 10 μΙ of 2 μΜ ATP. The reaction is performed until approx 50% of the ATP is depleted, and then stopped by the addition of 20 μΙ of Kinase-Glo solution (Promega Corp., Madison, Wl, USA). The
stopped reaction is incubated for 5 minutes and the remaining ATP is then detected via luminescence. IC50 values are then determined.
1.4 Biochemical assays for PI3Kdelta, PI3Kgamma
The TR-FRET Adapta™ Universal Kinase Assay Kit was purchased from Invitrogen Corporation (Carlsbad/CA, USA) (Cat. No. PV5099). The kit contains the following reagents: Adapta Eu-anti-ADP Antibody (Europium labeled anti-ADP antibody in HEPES buffered saline, Cat. No. PV5097), Alexa Fluor® 647-labeled ADP tracer (Alexa Fluor® 647-labeled ADP tracer in HEPES buffered saline, Cat. No. PV5098), proprietary TR-FRET dilution buffer pH 7.5 (Cat. No. PV3574).
PIK3CD substrate Phosphatidylinositol was obtained from Invitrogen (vesicules consisting of 2 mM PI in 50mM HEPES pH7.5; Cat. No. PV5371 ). PIK3CG substrate
Phosphatidylinositol-4,5-bisphosphate (PIP(4,5)2 was obtained from Invitrogen (PIP2:PS large unilamellar vesicules consisting of 1 mM PIP2: 19mM PS in 50mM HEPES pH7.5, 3mM MgCI2, 1 mM EGTA; Cat. No. PV5100).
Time-Resolved Fluorescence Resonance Energy Transfer (TR-FRET) is a technology based on energy transfer between two adjacent dyes, from an excited electron in one dye (the donor) to an electron of an adjacent dye (the acceptor) through resonance, then released as a photon. This energy transfer is detected by an increase in the fluorescence emission of the acceptor, and a decrease in the fluorescence emission of the donor. TR- FRET assays for protein kinases use a long-lifetime lanthanide Terbium or Europium chelates as the donor species which overcome interference from compound
autofluorescence or light scatter from precipitated compounds, by introducing a delay after excitation by a flashlamp excitation source. Results are often expressed as a ratio of the intensities of the acceptor and donor fluorophores. The ratiometric nature of such a value corrects for differences in assay volumes between wells, as well as corrects for quenching effects due to colored compounds. The Adapta™ assay can be divided into two phases : a kinase reaction phase and an ADP detection phase. In the kinase reaction phase, all kinase reaction components are added to the well and the reaction is allowed to incubate for a set period of time specific for each kinase. After the reaction, a detection solution of Eu-labeled anti-ADP antibody, Alexa Fluor® 647-labeled ADP tracer, and EDTA (to stop the kinase reaction) are added to the assay well. ADP formed by the kinase reaction will displace the Alexa Fluor® 647-labeled ADP tracer from the antibody, resulting in a decrease in TR- FRET signal. In the presence of an inhibitor, the amount of ADP formed by the kinase reaction is reduced, and the resulting intact antibody-tracer interaction maintains a high
TR-FRET signal. In the Adapta™ assay, the donor (Europium-anti-ADP antibody) is excited
at 340nm and will transfer its energy to the acceptor (Alexa Fluor® 647-labeled ADP tracer). The emission from the Alexa Fluor® 647 can be monitored with a filter centered at 665 nm because it is located between the emission peaks of the donor, which is measured at 615/620 nm.
50 nl_ of compound dilutions were dispensed onto white 384-well small volume polystyrene plate as described in section 2.2. Then 5 μΙ_ of PI3Kgamma and PI3Kdelta and lipid substrate (PI or PIP2:PS) followed by 5 μΙ_ of ATP (final assay volume 10 μΙ_) are incubated at RT. The standard reaction buffer for the Adapta™ TR-FRET assay contained 10mM Tris- HCI pH 7.5, 3mM MgCI2, 50mM NaCI, 1 mM DTT, 0.05% CHAPS. Reactions were stopped with 5 μΙ_ of a mixture of EDTA containing the Eu-labeled anti-ADP antibody and the Alexa Fluor® 647-labeled ADP tracer in TR-FRET dilution buffer (proprietary to IVG). Plates are read 15 to 60 mins later in a Synergy2 reader using an integration time of 0.4 seconds and a delay of 0.05 seconds. Control for the 100% inhibition of the kinase reaction was performed by replacing the PI3K by the standard reaction buffer. The control for the 0% inhibition was given by the solvent vehicle of the compounds (90% DMSO in H20).
Data are analyzed using Excel fit software or Graphpad Prism. EC50 values were derived by fitting a sigmoidal dose-response curve to a plot of assay readout over inhibitor
concentration. All fits were performed with the program XLfit4 (ID Business Solutions, Guildford, UK). Determination of EC50 values of the percentage inhibition of each compound at 8 concentrations (usually 10, 3.0, 1 .0, 0.3, 0.1 , 0.030, 0.010 and 0.003 μΜ) n=2 were derived by fitting a sigmoidal dose-response curve to a plot of assay readout over inhibitor concentration. All fits were performed with the program XLfit4 (ID Business Solutions, Guildford, UK). In one embodiment of the present invention, the class I PI3 kinase inhibitor, wherein said inhibitor has an inhibitory action on the PI3K isoform delta, wherein the range of activity, expressed as IC50, in the enzymatic PI3K delta assay is between 1 nM and 500 nM.
In another embodiment of the present invention, the class I PI3 kinase inhibitor, wherein said inhibitor has an inhibitory action on the PI3K isoform delta, wherein the range of activity, expressed as IC50, in the enzymatic PI3K delta assay is between 1 nM and 100 nM.
In another embodiment of the present invention, the class I PI3 kinase inhibitor, wherein said inhibitor has an inhibitory action on the PI3K isoform delta, wherein the range of activity, expressed as IC50, in the enzymatic PI3K delta assay is between 0.5nM and 10 nM.
In one embodiment of the present invention, the class I PI3 kinase inhibitor, wherein said inhibitor has an inhibitory action on the PI3K isoform gamma, wherein the range of activity, expressed as IC50, in the enzymatic PI3K gamma assay is between 1 nM and 500 nM. In another embodiment of the present invention, the class I PI3 kinase inhibitor, wherein said inhibitor has an inhibitory action on the PI3K isoform gamma, wherein the range of activity, expressed as IC50, in the enzymatic PI3K gamma assay is between 1 nM and 100 nM.
In one embodiment of the present invention, the class I PI3 kinase inhibitor, wherein said inhibitor has an inhibitory action on the PI3K isoform delta, wherein the range of activity, expressed as IC50, in the cellular PI3K delta assay is between 1 nM and 1000 nM.
In another embodiment of the present invention, the class I PI3 kinase inhibitor, wherein said inhibitor has an inhibitory action on the PI3K isoform delta, wherein the range of activity, expressed as IC50, in the cellular PI3K delta assay is between 1 nM and 500 nM.
In one embodiment of the present invention, the class I PI3 kinase inhibitor, wherein said inhibitor has an inhibitory action on the PI3K isoform gamma, wherein the range of activity, expressed as IC50, in the cellular PI3K gamma assay is between 1 nM and 1000 nM.
In another embodiment of the present invention, the class I PI3 kinase inhibitor, wherein said inhibitor has an inhibitory action on the PI3K isoform gamma, wherein the range of activity, expressed as IC50, in the cellular PI3K delta gamma is between 1 nM and 500 nM.
In another embodiment of the present invention, the class I PI3 kinase inhibitor shows an at least 10 fold selectivity over the PI3K isoform alpha.
In another embodiment of the present invention, the class I PI3 kinase inhibitor shows an at least 20 fold selectivity over the PI3K isoform alpha.
2. Cellular assays
2.1 Phosphoinositide-3 kinase (PI3K)-mediated Akt 1/2 (S473) phosphorylation in Rat-1 cells
Rat-1 cells stably overexpressing a myristoylated form of the catalytic subunit of human
phosphoinositide-3 kinase (PI3K) alpha, beta or delta were plated in 384-well plates at a density of 7500 (PI3K alpha), 6200 (PI3K beta), or 4000 (PI3K delta) cells in 30ul complete growth medium (Dulbecco's modified Eagle's medium (DMEM high glucose) supplemented with 10% (v/v) fetal bovine serum, 1 % (v/v) MEM non essential amino acids, 10mM HEPES, 2mM L-glutamine, 10 μg/mL puromycin and 1 % (v/v)
Penicillin/Streptomycin) and were incubated at 37%C / 5%C02 / 95% humidity for 24h. Compounds were diluted in 384-well compound plates to obtain 8-point serial dilutions for 40 test compounds in 90% DMSO, as well as 4 reference compounds plus 16 high controls and 16 low (inhibited) controls. Predilution plates were prepared by dispensing pipetting 250 nl of compound solutions into 384-well polypropylen plates using a
Hummingwell nanoliter dispensor . Compounds were prediluted by the addition of 49.75 ul complete growth medium. 10ul of prediluted compound solution were transferred to the cell plate using a 384-well pipettor, resulting in a final DMSO concentration of 0.1 1 %. Cells were incubated for 1 h at 37%C / 5%C02 / 95% humidity. The supernatant was removed, the cells were lysed in 20ul of lysis buffer for AlphaScreen® SureFire® detection.
For detection of p-AKT(Ser473), the SureFire® p-Akt 1 /2 (Ser473) Assay Kit
(PerkinElmer, U.S.A) was used. 5ul of cell lysate was transferred to 384-well low volume Proxiplates for detection using a 384-well pipettor. Addition of AlphaScreen® SureFire® reagents was done according to the manufacturer's protocol. First, 5ul of reaction buffer plus activation buffer mix containing AlphaScreen® acceptor beads was added, the plate was sealed, and incubated on a plate shaker for 2 hours at room temperature. Second, 2ul of dilution buffer containing AlphaScreen® donor beads was added, and the plate was incubated on plate shaker as above for a further 2 hours. The plate was read on an AlphaScreen® compatible plate reader, using standard AlphaScreen® settings.
2.2 Determination of murine B cell activation
PI3K5 has been recognized to modulate B cell function when cells are stimulated through the B cell receptor (BCR) (Okkenhaug et al. Science 297:1031 (2002). For assessing the inhibitory property of compounds on B cell activation, the upregulation of activation markers CD86 and CD69 on murine B cells derived from mouse spleen antibody is measured after stimulation with anti-lgM. CD69 is a well known activation marker for B and T cells (Sancho et al. Trends Immunol. 26:136 (2005). CD86 (also known as B7-2) is primarily expressed on antigen-presenting cells, including B cells . Resting B cells express CD86 at low levels, but upregulate it following stimulation of e.g. the BCR or IL-4 receptor. CD86 on a B cell interacts with CD28 on T cells. This
interaction is required for optimal T cell activation and for the generation of an optimal lgG1 response (Carreno et al. Annu Rev Immunol. 20:29 (2002)).
Spleens from Balb/c mice are collected, splenocytes are isolated and washed twice with RPMI containing 10% foetal bovine serum (FBS), 10 mM HEPES, 100 Units/mL penicilline/streptomycine. RPMI supplemented in this way is subsequently referred to as medium. The cells are adjusted to 2.5 X 106 cells/mL in medium and 200 μΙ cell suspension (5 x106cells) are added to the appropriate wells of 96 well plates.
Then the cells are stimulated by adding 50 μΙ anti-lgM mAb in medium (final concentration: 30 μg/mL). After incubation for 24 hours at 37°C, the cells are stained with the following antibody cocktails: anti-mouse CD86-FITC, anti-mouse CD69-PerCP- Cy5.5, anti-mouse CD19-PerCP for the assessment of B cells, and anti-mouse CD3- FITC, anti-mouse CD69-PE for the assessment of T cells (2 μΙ of each antibody/well). After one hour at room temperature (rt) in the dark the cells are transferred to 96 Deepwell plates. The cells are washed once with 1 ml_ PBS containing 2% FBS and after re-suspension in 200 μΙ the samples are analyzed on a FACS Calibur flow cytometer. Lymphocytes are gated in the FSC/SSC dot plot according to size and granularity and further analyzed for expression of CD19, CD3 and activation markers (CD86, CD69). Data are calculated from dot blots as percentage of cells positively stained for activation markers within the CD19+ or CD3+ population using BD CellQest Software.
For assessing the inhibitory property of compounds, compounds are first dissolved and diluted in DMSO followed by a 1 :50 dilution in medium. Splenocytes from Balb/c mice are isolated, re-suspended and transfered to 96 well plates as described above (200 μΙ/well). The diluted compounds or solvent are added to the plates (25 μΙ) and incubated at 37°C for 1 hour. Then the cultures are stimulated with 25 μΙ anti-lgM mAb/well (final concentration 30 μg/mL) for 24 hours at 37°C and stained with anti-mouse CD86-FITC and anti-mouse CD19-PerCP (2 μΙ of each antibody/well). CD86 expression on CD19 positive B cells is quantified by flow cytometry as described above. 2.3 Determination of rat B cell activation
PI3K5 has been recognized to modulate B cell function when cells are stimulated through the B cell receptor (BCR) (Okkenhaug et al. Science 297:1031 (2002). For assessing the inhibitory property of compounds on B cell activation, the upregulation of activation markers CD86 on rat B cells derived from whole blood is measured after stimulation with anti-lgM and recombinant IL-4. The CD86 molecule (also known as B7-
2) is primarily expressed on antigen-presenting cells, including B cells. Resting B cells express CD86 at low levels, but upregulate it following stimulation of e.g. the BCR or IL-4 receptor. CD86 on a B cell interacts with CD28 on T cells. This interaction is required for optimal T cell activation and for the generation of an optimal lgG1 response (Carreno et al. Annu Rev Immunol. 20:29 (2002)).
Collection of rat blood
Whole blood was collected from the abdominal aorta adult male Lewis rats (LEW/HanHsd) oby using a 10 ml syringe with hypodermic needle pre-coated with sodium heparin. Blood was transferred into 50 ml Falcon tubes and the anticoagulant concentration was adjusted to 100 U/ml.
Stimulation of rat B cells and treatment with specific inhibitor
For assessment of the in vitro effects of immunosuppressive drugs, heparinized blood was prediluted to 50% with medium. As medium served DMEM high glucose (Animed cat# 1 - 26F01 -I) supplemented with 100 U/ml penicillin, 100 mg/ml streptomycin, 2 mM L-glutamin, 50 mg/ml dextran 40 and 5% fetal calf serum (FCS, Fetaclone I, Gibco #10270-106). Then, 190 μΙ prediluted blood was spiked with 10 μΙ of pre-diluted test compound in 96 well U- bottomed microtiter plates (Nunc) resulting in a 3-fold serial dilution with a concentration range from 20 to 0.0003 μΜ. Control wells were pretreated with DMSO to obtain a final concentration of 0.5% DMSO. Cultures were set up in duplicates, mixed well by agitation on a plate shaker (Heidolph Titramax 101 ; 30 sec, speed 900), pipetting up and down and agitated on the plate shaker again. Cultures were incubated at 37°C, 5% C02 for 1 hr. Then, 20 μΙ of polyclonal goat anti-rat IgM Ab (Serotec, cat# 302001 ) and 10 μΙ of diluted recombinant rlL-4 (Immunotools # 340085) were added to obtain final concentrations of 30 μg/ml and 5 ng/ml, respectively. Plates were mixed by agitation on a plate shaker as above and incubated for 24 hrs at 37°C, 5% C02.
Determination of B cell activation by flow cytometry
After incubation, 15 μΙ of a 25 mM EDTA solution was added per well and shaken for 15 min to detach adherent cells. For analysis of surface activation markers, samples were then stained with PE-Cy5-labeled anti-ratCD45RA (BD cat# 557015) to allow gating on B cells in FACS analysis. In addition, samples were stained with PE-labeled anti-rat CD86 (BD cat# 551396). All staining procedures were performed at rt for 30 min in the dark. After incubation, samples were transferred to 96-deep well V-bottomed microtiter plates (Corning # 396096) containing 2 ml/well of BD Lysing Solution (BD # 349202). After lysis of erythrocytes samples were washed with 2 ml of CellWASH (BD # 349524). Data was acquired on an LSRII or FACScalibur flow cytometer (BD Biosciences) using Cellquest Plus or DIVA (version 6.1 .1) software, respectively. Lymphocytes were gated in the FSC/SSC dot blot according to size and granularity and further analyzed for expression of CD45RA and activation markers. Data
were calculated from dot blots or histograms as percentage of cells positively stained for activation markers within the CD45RA+ population.
Statistical evaluation
The percentage inhibition of B cell activation after exposure to drug was calculated by the following formula:
% Inhibition = 100 x stimulation without drug -stimulation with drug
stimulation without drug - unstimulated
ORIGIN 7 software (OriginLab Corporation, Northampton, MA) was used for non-linear regression curve fitting. The drug concentration resulting in 50% inhibition (IC50) was obtained by fitting the Hill equation to inhibition data.
2.4 Cellular U937 AKT assay for PI 3-kinase gamma
The U937 monocyte cell line is maintained in a basal medium of RPMI 1640 supplemented with 10% heat inactivated FCS, 100U/ml Penicillin, 100ug/ml streptomycin and 2mM L- glutamine (Invitrogen). U937 suspension culture is maintained by seeding cells at a density of 0.125x106 cells per ml in fresh medium every three or four days. Cells are incubated at 37°C, 5% C02. Three or four days prior to assay, cells are seeded at a density of 0.25x106 cells per ml in a total volume of 40 ml in a T175 culture flask.
Before beginning the cell manipulations described below, the MSD (Meso Scale Discovery) assay plate is blocked by addition of 150ul/well blocking buffer supplied and incubated with shaking for a minimum of one hour at room temperature. All steps of the assay must be performed quickly, with accurately timed incubation periods and observing temperature controls where indicated.
Cells seeded at 0.25x106/ml 3 or 4 days prior to the assay are aspirated, transferred to a 50ml
Falcon tube, counted and centrifuged for eight minutes at 300g at room temperature.
Supernatant is aspirated, the cell pellet resuspended and washed once in HBSS (Hank's Balanced Salt Solution) by centrifugation for eight minutes at 300g at room temperature. The cell pellet is resuspended in HBSS to a concentration of 4x106 per ml, and 100μΙ_ of cell suspension added to each well of a flat-bottomed 96-well tissue culture plate. Assay plates are incubated for 1 .5 hours at 37°C, 5% C02 to allow background AKT phosphorylation to reduce before the compound stimulation step.
A 5mM stock concentration of compound is prepared in 100% DMSO; from this a 1 in 125 dilution is made in HBSS giving a top compound concentration of 40μΜ, 0.8% DMSO. Compound titrations are prepared in a fresh flat-bottomed, 96-well plate, by 10-fold serial dilution of 40uM into HBSS 0.8% DMSO; pipette tips are replaced after each dilution is made. Compound concentrations at this stage are 4-times the final concentration required in
the assay plate. Cells are stimulated with compound or HBSS 0.8% DMSO by direct transfer of 50ul/well from the compound dilution plate. The assay plate containing compound-treated cells is then incubated for 30 minutes at 37°C. A standard plate layout is used for all experiments.
Compound-treated cells, in addition to positive control wells ("max MIP1 a"), are stimulated with 50μΙ_ per well of 40ng/ml MIP1 a (R&D Systems catalogue number 270-LD, lyophilized stock reconstituted to 50μg/ml with PBS 0.1 % BSA). Negative control wells ("min HBSS"), are stimulated with 50μΙ/ννβΙΙ of HBSS in the absence of MIP1 a. Final compound
concentrations are now diluted 4-fold giving a top concentration of 10μΜ; where added, the final concentration of MIP1 a is 10ng/ml. Cells are incubated with MIP1 a for 3 minutes, at 37°C, 5% C02. After the three minute stimulation period, the assay plate is kept ice cold at all times. Assay plates are centrifuged for 2 minutes at 300g, 4°C and supernatant is removed by gently inverting, and then blotting the plate on tissue. Cells are then washed by gentle addition of 150μΙ_Λ/νβΙΙ of ice cold HBSS and centrifugation at 300g, for 5 minutes at 4°C. Supernatant is
aspirated and the plate blotted as described above. The plate is placed on ice and cells are immediately treated with 35μΙ_ per well of ice cold lysis buffer, prepared according to the kit instructions (per assay plate, to 5ml of Tris lysis buffer add 10ΟμΙ of 50x protease inhibitor solution and 50μΙ of each 100x phosphatase inhibitor solutions I and II). Plates are incubated on ice for 20 minutes before centrifugation at 841 g for 5 minutes, 4°C.
Block buffer is aspirated from the MSD plate, and the plate washed four times with
300μΙ/ννβΙΙ Tris wash buffer. 25μΙ_ of cell lysate is then transferred from the assay plate to the washed MSD plate which is sealed and incubated at room temperature for one hour with shaking. The plate is washed four times with 300μΙ_ per well of Tris wash buffer before addition of 25μΙ_ per well of sulfo-tag anti-total AKT/pAKT detection antibody (60μΙ of 50x antibody stock is diluted in 1 ml block buffer mixed with 2ml wash buffer) and incubated at room temperature for one hour with shaking. The plate is washed four times with 300μΙ per well of Tris wash buffer and 150μΙ per well of Read buffer is added, taking care to avoid the introduction of bubbles. The plate is immediately read using an MSD SECTOR Imager 6000. Results are exported in Excel and the percentage of phosphorylated AKT is calculated using the equation: % Phosphoprotein = ((2* Phospho signal) / (Phospho signal + Total signal))* 100. Compound-mediated inhibition of AKT phosphorylation is analysed using Prizm V Graphpad software.
2.5 Determination of TLR9-induced IL-6 in mouse splenocytes
Preparation of single cell suspension from mouse spleen
Spleens were dissected from C57BL/6 mice immediately following euthanasia. Excess fat was trimmed from the spleens prior to mashing the spleen through a 0.4 μΜ cell strainer using a plunger from a 5 ml syringe. A single cell suspension was prepared and the volume was adjusted to 15 ml in a 50 ml Falcon tube using cold PBS. Cells were centrifuged at 1500 rpm for 5 minutes at 4°C degrees prior to removal of supernatant and re-suspension in 5 ml of red blood cell lysis buffer per spleen and incubation for 5 minutes at room temperature. Ice cold PBS (30 ml) was added to the cells prior to centrifugation at 1500 rpm for 5 minutes at 4°C. The supernatant was removed and the cells were washed twice with 40 ml of murine splenocyte culture media (MSCM). MSCM consisted of RPMI supplemented with 100 units/ml Penicillin and 100 μg/ml Streptomycin, 1 x nonessential amino acids, 1 mM Sodium Pyruvate, 0.05 mM β-mercaptoethanol, and 10% heatinactivated Fetal Bovine Serum (FBS). Cells were re-suspended in 10-20 ml of MSCM and counted using a Countess cell counter. Approximately 60x106 splenocytes were obtained from a single C57BL/6 mouse spleen. Stimulation of murine splenocytes and treatment with specific inhibitor
Splenocytes were plated at a final density of 2x105 cells/well in a volume of 100 μΙ in 96 well flat bottomed plates and incubated in a humidified 37°C incubator for 2-4 hours. Afterwards, compounds to be tested were dispensed using an automated liquid handling machine using previously prepared compound stock plates. Stock plates consisted of compounds (in 90%/10% DMSO/ddH20) arrayed in 8-10 point using 2- or 3-fold dilutions. The liquid handling machine dispensed 1 μΙ of each dilution from the previously prepared compound source plate into the appropriate destination well in the 96-well plate. The final starting concentration of the compounds in the cell culture was 10 μΜ. The final concentration of DMSO in the cell cultures was 0.5%. Cells were incubated with compounds for 1 hour prior to addition of TLR ligand. Then, a 10x EC80 concentration of CpG1826 was added in a volume of 20 μΙ (for a final culture volume of 200 μΙ) whereupon cultures were incubated overnight in a humidified 37°C incubator.
Determination of lnterleukin-6 by ELISA
After overnight culture, plates were centrifugated at 2000 rpm for 5 minutes at room temperature. Subsequently 150 μΙ of each culture was transferred to 96-well V-bottomed plates and IL-6 levels were measured using commercially available mouse IL-6 sandwich ELISA kit. Briefly, plates were coated overnight with the capture antibody prior to blocking for 1 hour with PBS/0.1 % BSA. Samples and standards were added in a volume of 50 μΙ and the plate was incubated for 2 hours at room temperature. After removal of the
standards/samples, the plate was washed using PBS/0.05% Tween prior to addition of 50 μΙ
of the biotinylated detection antibody whereupon the plate was incubated for 2 hours at room temperature with agitation. Plates were washed again prior to addition of 50 μΙ streptavidin- horseradish peroxidase per well for 20 minutes. Following additional plate washes 50 μΙ TMB substrate was added to each well and plates were incubated for 20 minutes prior addition of 25 μΙ/well stop solution. IL-6 levels were measured using a SpectraMax 190 Plate Reader (450 nm) and analyzed using SoftMax Pro and GraphPad Prism software.
2.6 Determination of TLR9-induced IFNalpha in human peripheral blood
mononuclear cells (PBMC)
Preparation of PBMC from fresh human blood
Human blood (ca. 75 ml) was collected in 10 S-Monovette tubes containing Heparin (S- Monovette 7.5 mL NH Heparin 16 lU/mL blood; Starstedt). Leucosep™ tubes (30 ml_ #227290; Greiner Bio-one) were prepared by addition of 15 ml lymphocyte separation medium LSM1077™ per tube (#J15-004; PAA Laboratories) and centrifugation for 30 sec at 1000g. Some 25 ml blood was transferred to Leucosep™ tubes following dilution with equal parts of PBS (without Ca2+/Mg2+; #14190-094). Samples were centrifuged at 800g for 20 min at 22 °C using an Eppendorf 581 OR centrifuge without brake. The PBMC layer was carefully removed from plasma:separation medium interface and transferred into clean 50 ml tube. Cells were washed once by addition of PBS (up to 45 ml) and centrifuged (1400rpm, 10 min at 22 °C) with brake (set at speed 9) using an Eppendorf 581 OR. Pelleted cells were carefully resuspended in Media (RPMI 1640+GlutaMAX-l, 0.05 mM 2-mercaptoethanol, 10 mM HEPES and 5% v/v FCS) and samples pooled. The medium components 2- mercaptoethanol (#31350-010; 50 mM), Hepes (#15630-056, 1 M) and RPMI 1640 (1 x) + GlutaMAX-l (#61870-010) were obtained from Gibco. FCS (#2-01 F36-1) was obtained from Amimed. The PBMC were counted using a Countess® Automated cell counter (sample was pre-diluted 1 :10 in Media, prior to the addition of equal volume (10 μΙ) of Trypan Blue). Cells were diluted to 4 x 106 cells/ml and seeded in 384-well plates (#353962; Becton Dickinson AG) to give a final volume of 25 μΙ (i.e. 1 x 105 cells/well).
Stimulation of PBMC and treatment with specific inhibitor
Compounds were pre-diluted in 100% v/v DMSO (#41640-1 OOmL; Sigma-Aldrich), followed by transfer in Media (to achieve a final DMSO concentration of 0.25%). Cells were treated with appropriate compound dilution (5 μΙ) or vehicle control (5 μΙ) and incubated for 30 min at 37 °C in a humidified incubator in air with 5% (v/v) C02. Cells were stimulated with CpG2216 (0.3 μΜ; #tlrl-hodna; Invivogen) or vehicle control (10 μΙ/well) and incubated for 20 h. Plates were briefly centrifuged (200 x g for 2 min at 22 °C) and supernatant samples (30 μΙ) removed for quantification of IFNa levels.
Quantification of IFNa using AlphaLisa technology
For quantification of IFNalpha the human interferon AlphaLISA Kit (#AL264F) from
PerkinElmer was used. An antibody mix containing anti-IFNa acceptor beads (5 μg/ml final) and biotinylated antibody anti-IFNa (0.5 nM final) is prepared fresh and dispensed (5 μΙ) into 384-well Optiplates™ (#6007299; PerkinElmer). Dilution of known IFNa standards (human IFNa B (2b)) were prepared and together with cell supernatants (5 μΙ) were added to plates above. Plates were briefly centrifuged (pulse at 200g), covered with adhesive sealing film, vortexed and incubated 1 h at room temperature in the dark. Streptavidin-coated donor beads (20 μg/ml final) was prepared and added to each well (5 μΙ) in a dark lit area (light sensitive mix). Plates were incubated 30 min at room temperature (Pates must not be centrifuged or covered). After incubation, the plates were read with an EnVision™ multiplate reader equipped with the ALPHA option using the instrument's own "AlphaScreen standard settings" (e.g. total measurement time: 550 ms, Laser 680 nm excitation time: 180 ms, mirror: D640 as, emission filter: M570w, center wavelength 570 nm, bandwidth 100 nm, transmittance 75%). Data were collected for analysis and quantification of IFNa levels.
Data evaluation and analysis
Data were analysed using Excel XL fit 4.0 (Microsoft) with XLfit add-in (IDBS; version 4.3.2). Specific IFNa concentrations were determined following extrapolation to standard curves using human IFNa B (2b). Individual IC50 values of compounds were determined by nonlinear regression after fitting of curves to the experimental data.
3 Determination of antibody production to sheep red blood cells (SRBC).
In brief, OFA rats were injected i.v. with sheep erythrocytes on dO and treated orally on four consecutive days (dO to d3) with the compounds under investigation. Spleen cell suspensions were prepared on d4 and lymphocytes were plated onto soft agar in presence of indicator cells (SRBC) and complement. Lysis of the indicator cells due to secretion of SRBC-specific antibody (predominantly of the IgM subclass) and presence of complement yielded plaques. The number of plaques per plate were counted and expressed as number of plaques per spleen. Immunization: Groups of five female OFA rats were immunized on day 0 with 2x108/ml SRBC (obtained from Laboratory Animal Services LAS, Novartis Pharma AG) in a volume of 0.5ml per rat by i.v. injection.
Compound treatment: Animals were treated with compound suspended in 0.5% CMC, 0.5%Tween80 in for 4 consecutive days (days 0, 1 , 2 and 3) starting on the day of immunization. Compound was administered orally twice daily with 12 hours intervalls between doses in an application volume of 5 ml/kg body weight.
Preparation of spleen cell suspensions:
On day 4, animals were euthanized with C02. Spleens were removed, weighed, and deposited in plastic tubes containing 10 ml of cold (4 °C) Hank's balanced salt solution (HBSS; Gibco, pH 7.3, containing 1 mg Phenolred/100ml) for each rat spleen. Spleens were homogenized with a glass potter, left on ice for 5 minutes and 1 ml supernatant was transferred into a new tube. Cells were washed once in 4 ml HBSS then supernatants were discarded and pellets re-suspended in 1 ml of HBSS. Lymphocyte numbers per spleen were determined by automated cell counter and spleen cell suspensions were adjusted to a cell concentration of 30x106/ml.
Plaque forming assay:
Soft agar petri dishes were prepared with 0.7% agarose (SERVA) in HBSS.
In addition, one ml of 0.7% agarose was prepared in plastic tubes and kept at 48°C in a water bath. Some 50 μΙ of a 30x106/ml spleen cell suspension and 50 μΙ of SRBC at 40 x 108/ml were added, mixed rapidly (Vortex) and poured onto the prepared agarose dishes. Petri dishes were slightly tilted to achieve even distribution of cell mixture on agarose layer. The dishes were left at room temperature for 15 minutes and were then incubated at 37°C for 60 minutes. Then, 1 .4ml guinea pig complement (Harlan; 10%) was added and the incubation continued for another 60 minutes at 37 °C. SRBC- specific antibodies released by the plated-out B cells bound to the antigen (SRBC) in their vicinity. These antigen-antibody complexes activated complement and led to the lysis of the SRBC leaving a bright spot (plaque) within the red erythrocyte layer. Plaques were counted with a microscope.
The following formula for determination of inhibition of plaque formation was used: %lnhibition = C*100/V-100
with: V= mean number of plaques/spleen for vehicle group; C= mean number of plaques/spleen for compound treated group
References:
N.K. Jerne & A.A. Nordin (1963) Plaque formation in agar by single antibody-producing cells. Science 140:405.
N.K. Jerne, A.A. Nordin & C. Henry (1963) The agar plaque technique for recognizing antibody-producing cells. In: "Cell Bound Antibodies", B. Amos & H. Koprowski, Eds., Wistar Inst. Press, Philadelphia pp.109-125.
Biological data Enzymatic Assay
PI3K alpha PI3K delta PI3K gamma
Example ID IC50 IC50 IC50
[umol 1-1] [umol 1-1] [umol 1-1]
A01a 0.816 0.008 0.011
A01b 1.014 0.013 0.043
A02a 0.78 0.007 0.021
A02b 0.871 0.005 0.014
A02c 0.78 0.009 0.039
A03 5.208 < 0.003 0.070
A04 0.393 < 0.003 0.024
A05 3.7 0.052 0.305
A06 3.6 0.085 0.110
A07 5.1 0.004 0.081
A08 0.072 0.007 0.022
A09 4.667 0.034 0.103
A10 >10 0.076 5.20
A11 0.56 0.007 0.025
A12 4.6 0.042 0.063
A13 3.3 0.020 0.170
A14 >10 0.660 1.9
A15 7.7 0.014 0.150
A16 >10 0.240 0.460
A17 1.5 0.008 0.024
A18 8.4 0.270 0.220
A19 1.6 0.011 0.200
A20 4.55 0.059 0.295
A21 4.7 0.008 0.280
A22 2.8 0.035 0.140
A23 2.0 0.089 0.320
A24 1.1 0.021 0.190
A25 1.2 0.110 0.780
A26 0.35 0.010 0.019
A27 1 .8 0.010 0.200
A28 2.3 0.012 0.130
A29 >10 0.120 3.2
A30 0.580 0.007 0.490
A31 0.590 < 0.003 0.019
A32 1 .03 0.026 0.058
A33 >10 0.012 0.170
A34 1 .78 0.024 0.096
A35 >10 0.016 1 .1
A36 0.930 0.031 0.1 10
A37 5.95 1 .430 0.445
A38 5.25 2.500 0.505
A39 0.790 0.018 0.071
A40 2.8 0.012 0.14
A41 a 1 .6 0.010 0.300
A41 b 2.1 0.025 0.290
A41 c 1 .24 0.007 0.395
A42 >10 0.120 >10
A43 >10 0.150 4.3
A44 5.6 0.073 3.1
A45 >10 0.043 >10
A46 2.80 0.067 0.093
A47 5.55 0.090 2.30
A48 0.60 0.014 0.041
A49 >10 0.043 >10
A50 3.10 0.023 0.230
A51 0.34 0.009 0.070
A52 2.40 0.012 0.590
A53 0.46 0.005 0.046
A54 0.70 0.01 1 0.120
A55 0.37 0.017 0.049
A56 0.39 0.018 0.030
A57 4.20 0.013 0.50
A58 1 .70 0.048 0.087
A59 0.89 0.017 0.082
A60 0.54 0.024 0.18
A61 2.70 0.075 0.95
A62 5.60 0.019 0.66
A63 4.50 0.050 0.24
A64 >10 0.16 0.32
A65 4.20 0.10 0.23
A66 3.00 0.019 0.65
A67 0.33 0.021 0.064
A68 0.29 0.046 0.052
B01 3.22 0.014 0.081
B02a 2.067 0.004 0.012
B02b 3.321 0.007 0.053
B02c 1 .601 < 0.003 0.010
B03 2.944 0.018 0.202
B04 2.34 0.005 0.129
B05 6.816 0.01 1 0.148
B06a 2.2 0.005 1 .2
B06b 2.4 0.007 0.230
B06c 8.6 0.010 1 .3
B07a > 9.1 0.003 0.610
B07b > 9.1 0.049 3.5
B07c 0.560 0.017 0.350
B08a 3.6 0.019 0.190
B08b 5.0 0.004 0.230
B09 2.3 0.01 1 0.023
B10a 6.7 0.009 0.250
B10b 3.7 0.027 0.310
B10c 4.4 0.012 0.640
B1 1 a > 9.1 0.019 0.330
B1 1 b > 9.1 0.200 2.7
B1 1 c > 9.1 0.035 1 .4
B12a > 9.1 0.076 0.12
B12b > 9.1 0.1 10 0.17
B12c 3.6 0.062 0.098
B13 7.9 0.021 0.34
B14 > 9.1 0.056 0.42
B15 >10 7.2 >10
B16 >10 0.082 1 .1
B17 >10 0.140 0.820
C01 1 .3 0.004 0.035
C02 1 .0 0.01 1 0.027
C03 0.565 0.053 0.030
C04 2.8 0.128 0.417
C05 1 .317 0.104 0.220
C06 4.0 0.066 0.380
C07 1 .23 0.023 0.053
C08a 0.34 0.01 1 0.009
C08b 0.26 0.008 0.025
C09 7.6 0.018 0.460
C10 >10 0.053 1 .9
C1 1 2.1 0.024 0.107
D01 >10 0.035 0.140
D02 0.310 0.004 0.019
D03 0.200 0.018
D04 0.510 0.014 0.133
D05 2.2 0.022 0.605
D06 >10 7.7 >10
D07 >10 0.041 0.67
D08 2.90 0.003 0.180
D09 0.27 0.084 0.013
D10 >10 0.130 0.9
D1 1 2.30 - 0.076
D12 0.45 <0.003 0.087
D13 >10 0.044 -
E 0.3 0.004 0.017
F01 2.0 < 0.003 0.044
F02 > 9.1 0.150 0.980
F03 1 .5 0.007 0.081
F04 >10 0.100 1 .2
F05 0.77 0.003 0.039
F06 7.6 0.1200 0.330
G01 0.78 < 0.003 0.005
G02 1.4 0.005 0.019
G03 0.36 0.012 0.018
G04 0.698 0.221 0.085
G05 0.3 0.005 0.012
G06 1.0 0.004 0.050
G07 0.16 < 0.003 < 0.003
G08 1.80 0.008 0.066
G09 2.50 0.027 0.037
G10 0.45 0.075 0.14
G11 6.60 0.39 0.32
G12 1.00 0.012 0.086
G13 3.10 0.097 0.89
G14 2.00 0.066 0.10
G15 0.53 0.026 0.053
G16 0.70 0.043 0.097
G17 0.27 0.014 0.024
G18 2.20 0.010 0.37
G19 1.70 0.024 0.080
G20 9.30 0.082 0.72
G21 1.80 0.96 0.22
G22 3.80 0.084 1.30
G23 0.28 0.013 0.014
G24 2.80 0.047 0.99
G25 1.40 0.016 0.21
G26 0.51 0.011 0.027
G27 4.00 0.096 1.30
G29 >10 0.082 2.10
G30 1.30 0.018 0.090
G31 0.11 0.004 0.011
H 0.62 0.005 0.034
I 1.433 0.003 0.110
J01 4.4 0.021 0.041
J02 2.6 0.022 0.190
J03 3.8 0.079 0.300
J04 7.2 0.140 0.078
J05 3.30 0.026 0.031
J06 1.00 0.015 0.031
K01 >10 0.360 0.370
K02 9.3 0.440 >10
K03 >10 0.200 >10
K04 >10 0.210 3.7
K05 >10 0.510 5.6
L01 5.10 0.007 1.20
L02 1.10 0.013 0.075
L03 4.50 0.084 7.20
L04 5.50 0.038 1.10
M >10 0.22 0.058
N01 0.59 0.010 0.047
N02 3.60 <0.003 0.068
N03 1.30 0.018 0.009
N04 2.40 0.008 0.055
N05 4.40 0.064 0.048
N06 1.30 0.026 0.035
N07 2.50 0.026 0.038
N08 0.91 0.014 0.042
Cellular Assays
Cell Cell Cell
PI3K alpha PI3K delta PI3K gamma RWB / IC50
IC50 IC50 IC50 CD86
Example ID [umol 1-1 ] [umol 1-1 ] [umol 1-1 ] [nmol 1-1 ]
A01 a 2.83 0.008 0.019 25.5
A01 b 5.52 0.009 0.035 15.5
A02a 2.22 0.008 0.035
A02b 2.46 0.006 0.080
A02c 4.61 0.008 0.046 26.0
A03 2.2 0.013 0.027 8.5
A04 1 .75 0.007 0.074 8.2
A05 >10 0.1 10 0.216 183.0
A06 8.12 0.349 0.886
A07 4.69 0.051 0.215 9.0
A08 1 .107 0.005 21 .0
A09 5.15 0.056
A10 >10 >10 0.382
A1 1 4.02 0.043 0.074 33
A12 >10 0.065 1 .821
A13 >10 0.084
A14 >10 1 .4
A15 4.59 0.012
A16 >10 0.152
A17 >10 0.077 0.675 22.0
A18 >10 0.651
A19 >10 0.020
A20 >10 0.179
A21 >10 0.036
A22 >10 0.080
A23 7.68 0.109
A24 3.53 0.026
A25 2.86 0.128
A26 >10 < 0.003 0.059
A27 >10 0.198
A28 8.58 0.291
A29 >10 0.278
A30 2.15 0.007
A31 4.23 0.005 0.037
A32 3.3 0.032
A33 >10 0.121
A34 >10 0.149
A35 >10 0.013
A36 1 .16 0.061
A37 5.46 0.894
A38 > 6.5 0.476 2.365
A39 1 .79 0.015 0.027 16.5
A40 >10 0.062 0.261 22.0
A41 a > 6.5 0.056 0.554
A41 b 9.2 0.024 0.264
A41 c 2.78 0.014 1 .522
A42 >10 0.733
A43 >10 0.318
A44 >10 0.148
A45 >10 0.292
A46 >10 8.04 0.312
A47 >10 <0.003
A48 >10 <0.003
A49 >10 >10
A50 >10 <0.003
A51 >10 0.077
A52 >10 0.166
A53 0.210 0.037
A54 7.16 0.025
A55 2.09 0.042
A56 0.522
A57 0.21 1
A58 >10 0.1 1 0.040
A59 4.44 0.037 0.164
A60 3.84 0.027 0.082
A61 5.93 0.098 0.934
A62 9.95 0.038 0.718
A63 8.74 0.056 0.092
A64 >10 0.164 0.324
A65 >10 0.295 0.306
A66 >10 <0.003 0.148
A67 2.87 0.009 0.115
A68 2.37 0.006 0.035
B01 6.19 0.071 0.195
B02a >10 0.035
B02b >10 0.103
B02c 7.47 0.039 0.310
B03 >10 0.057
B04 8.12 0.014 >10 10.4
B05 4.47 0.038
B06a 9.54 0.046
B06b 5.4 0.022 85.5
B07a 5.96 0.034
B07b 7.51 0.530
B07c 3.48 0.059
B08a >10 0.295
B08b >10 0.072
B09 6.63 0.046 0.119
B10a 9.16 0.035
B10b >10 0.097
B10c 3.68 0.024
B11a 4.06 0.073
B11b >10 1.380
B11c 6.89 0.112
B12a 7.04 0.500
B12b 8.94 0.805
B12c 7.07 0.391
B13 7.14 0.142
B14 >10 0.222
B15 >10 >10
B16 >10 0.262
B17 >10 0.509
C01 4.04 0.010 0.209 21.0
C02 5.48 0.043 0.352 47.0
C03 1.32 0.006 0.038 19.0
C04 3.23 0.026 0.473
C05 3.54 0.073
C06 >10 0.032
C07 >10 0.029 35.5
C08a 1.08 0.005 17.0
C08b 0.866 0.008 10.0
C09 >6.5 0.035 0.671
C10 >10 0.117 1.888
C11 >6.5 0.182 0.227
D01 >10 0.154
D02 >6.5 < 0.003 0.048 2.78
D03 2.461 < 0.003 0.013 3.655
D04 >6.5 < 0.003 0.085
D05 > 3.0 0.071
D06 >10 >10 9.163
D07 >10 0.119 2.455
D08 >10 0.034 0.160
D09 2.30 0.005
D10 >10 0.377 3.766
D11 >10 0.103 0.101
D12 0.062
E >10 0.076 0.148 25.5
F01 3.69 0.014 0.144 15.5
F02 >10 1.81
F03 5.99 0.057
F04 >10 2.480
F05 4.26 0.011 0.199 3.9
F06 >10 4.040
G01 >3 0.008 0.059 17.0
G02 4.31 0.021 0.178 25.0
G03 1.038 0.004 0.005 1.4
G04 4.23 0.086 0.219 46.5
G05 1.3 < 0.003 0.035
G06 2.85 < 0.003 0.268 56.5
G07 0.601 < 0.003 0.035
G08 >10 0.003
G09 5.31 0.089 0.055
G10 4.67 0.045 0.070
G1 1 >10 0.141 0.1 13
G12 6.24 0.009 0.024
G13 8.84 0.042 0.060
G14 4.59 0.092 0.083
G15 2.50 0.018 0.037
G16 7.13 0.030 0.033
G17 1 .09 0.007 0.018
G18 2.72 0.007 0.331
G19 6.57 0.016 0.010
G20 >10 0.029 0.146
G21 >10 0.067 0.206
G22 3.86 0.086 0.163
G23 1 .95 0.010 0.007
G24 8.02 0.027 0.026
G25 7.22 0.091 0.034
G26 7.54 0.087 0.038
G27 >10 0.045 1 .59
G29 >10 0.023 0.709
G30 6.60 0.024 0.1 14
G31 2.37 0.006 0.035
H >10 0.017 0.1 17 33.0
I >10 0.015 0.494 13.0
J01 >10 0.031 0.162
J02 >10 0.130 0.399
J03 >10 0.013 0.350
J04 >10 0.204 0.793
J05 >10 0.090 0.031
J06
K01 >10 0.939 >10
K02 >10 1 .150
K03 >10 2.350
K04 >10 > 3
K05 >10 > 3
L01 4.60 0.038 0.821
L02 4.60 0.042 0.343
L03 >10 0.065 >10
L04 >1 0 0.103 1 .58
M >1 0 0.517 0.187
N01 2.06 0.012 0.022
N02 5.23 0.033 0.088
N03 3.30 0.029 0.045
N04 0.766 0.010 0.031
N05 >10 0.240 0.076
N06 4.20 0.023 0.053
N07 8.25 0.050 0.071
N08 3.41 0.014 0.042
Claims
1 . A compound of formula (I)
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3-pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4-pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, trideuteromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro,
methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S; or
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S;
or
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
wherein
2. A compound according to claim 1 or a pharmaceutically acceptable salt thereof, of the formula (Γ)
R1 is selected from
phenyl, which is substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
3- pyridyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
4- pyridyl, which is unsubstituted or substituted by one substituent in the 2 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
1 -methylpyrazol-5-yl;
2-methylthiophen-5-yl;
C3-C6-cycloalkyl, which is unsubstituted or substituted in the 1 position by methyl;
tetrahydropyran-4-yl;
piperidin-1 -yl;
morpholin-4-yl;
pyrolidin-3-yl, which is unsubstituted or substituted in the 1 position by a substituent which is independently selected from methoxycarbonyl, methylsulfonyl, methyl or methylcarbonyl; or
dimethylamine;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
wherein
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
X is independently selected from NH, NMe or S.
3. A compound according to claim 1 or a pharmaceutically acceptable salt thereof, of the formula (I")
(I")
wherein,
R1 is selected from
phenyl, which is unsubstituted or substituted by one substituent in the 4 position which is independently selected from
methyl, ethyl, difluoromethyl, methoxy, difluoromethoxy, cyclopropyl, chloro or fluoro;
R2 is independently selected from methyl, ethyl, difluoromethyl, trifluoromethyl, methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, cyano, fluoro, methylsulfonyl, ethylsulfonyl, cyclopropylsulfonyl, chloro, methylcarbonyl, 1 -hydroxyethyl, methylcarbonyl-amino, amino, methylamino, methylsulfonyl- amino or cyclopropyl;
R5 and R6 are independently selected from hydrogen, deuterium or fluoro;
-L-R3 is independently selected from
R7 is independently selected from methoxy, difluoromethoxy, trifluoromethoxy, hydroxy, methylsulfonylamine or fluoro; and R3 is independently selected from
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S.
4. A compound according to claim 2 or a pharmaceutically acceptable salt thereof, wherein
R3 is independently selected from
R4 is independently selected from hydrogen or amino,
R8 is independently selected from hydrogen, methyl, fluoromethyl, difluoromethyl, trideuteromethyl or amino, and
Z is independently selected from NMe or S;
5. A compound according to claim 2 or a pharmaceutically acceptable salt thereof wherein
R3 is selected from
R4 is independently selected from hydrogen or amino,
R8 is methyl, and
X is S.
6. A compound according to claim 3 or a pharmaceutically acceptable salt thereof, wherein
R3 is selected from
R4 is independently selected from hydrogen or amino,
R8 is methyl, and
Z is S.
7. A compound according to any one of claims 1 to 6 or a pharmaceutically
acceptable salt thereof, wherein
R5 is independently selected from hydrogen or fluoro;
R6 is independently selected from hydrogen; and
R2 is independently selected from methyl, trifluoromethyl, difluoromethoxy, fluoro methylsulfonyl.
8. A compound according to claim 1 , selected from
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one, (S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclopropylquinazolin-4(3H)-one, (S)-5-Chloro-3-phenyl-2-(1-(thiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)quinazolin-4(3H)-one, (S)-2-Amino-4-(2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-5-Chloro-2-(1 -(imidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-methoxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin-4(3H)-one, (S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(pyridin-3-yl)quinazolin-4(3H)-one, (S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(6-methoxypyridin-3-yl)quinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(pyridin-3-yl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-chloro-3-(6-methoxypyridin-3-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(pyridin-3-yl)quinazolin-4(3H)-one, (S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(6-methoxypyridin-3-yl)quinazolin- 4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(tetrahydro-2H^yran-4-yl)quinazolin-4(3H one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(tetrahydro-2H-pyran-4-yl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin-4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(dimethylamino)quinazolin-4(3H)-on (S)-4-Amino-6-(2-(5-chloro-3-(dimethylamino)-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolid yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(dimethylamino)quinazoli one,
(S)-2-(1-(5-Aminothiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin- 4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(piperidin-1-yl)quinazolin-4(3H)-one,
(S)-2-(1 -(2-Amino-9H^urin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(piperidin-1 -yl)quinazolin-4(3H)- one,
(S)-4-Amino-6-(2-(5-chloro-4-oxo-3-(piperidin-1 -yl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(1 -methylcyclopropyl)quinazolin-4(3H)-one, (S)-4-Amino-6-(2-(5-chloro-3-(1 -methylcyclopropyl)-4-oxo-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-(1 -methylcyclopropyl)quinazolin- 4(3H)-one,
(S)-2-(1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclohexylquinazolin-4(3H)-one,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclohexylquinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-chloro-3-cyclohexyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-cyclopropylquinazolin- 4(3H)-one,
(S)-2-(1-(2-Amino-9-methyl-9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-morpholinoquinazolin- 4(3H)-one,
(S)-2-Amino-4-(2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)py yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3-(o-tolyl)-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolid yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(9H-Purin-6-yl)pyrrolidin-2-yl)-5,7-difluoro-3-phenylquinazolin-4(3H)-one,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-5 ,7-difluoro-3-phenylquinazolin-4(3H)-one, (S)-2-Amino-4-(2-(5 ,7-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5,7-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(2-Aminoimidazo[2,1-f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5,6-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5,6-difluoro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-4-Amino-2-methyl-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrroli^ yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-(difluoromethoxy)-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)-6-methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-(difluoromethoxy)-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1^ yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-methyl-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-5-methyl-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-5-methyl-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1- yl)-6-methylpyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-(1-(5-Aminothiazolo[5,4-d]pyrimidin-7-yl)pyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
(S)-2-Amino-4-(2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin-2^ yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(S)-2-Amino-4-methyl-6-(2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4-dihydroquin yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(3-(5-fluoropyridin-3-yl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(S)-2-(1-(2-Amino-9H-purin-6-yl)pyrrolidin-2-yl)-3-(5-fluoropyridin-3-yl)-5- (trifluoromethyl)quinazolin-4(3H)-one,
(S)-2-Amino-4-methyl-6-(2-(4-oxo-3-(pyrimidin-5-yl)-5-(trifluoromethyl)-3,4-dihydroqu 2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(5-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(5-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6- methylpyrimidine-5-carbonitrile,
(S)-2,4-Diamino-6-(2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrroli^ 1 -yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(R)-2-(3-(2-Amino-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H^ (R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-phenylquinazolin-4(3H)-one,
(R)-4-Amino-6-(4-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)thiazolidin-3- yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)thiazolidin-3-yl)- pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(2-cyclopropyl-phenyl)-4-oxo-3,4-dihydroquinazolin-2-yl)- thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(2-(difluoromethoxy)phenyl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(2-fluorophenyl)quinazolin-4(3H)-^ (R)-2-(3-(2-Amino-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3^henylquinazolin-4(3H)-on (R)-4-Amino-6-(4-(5-chloro-3-(2-fluorophenyl)-4-oxo-3,4-dihydroquinazolin-2-yl)thiazolidi^ yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(2-(difluoromethoxy)phenyl)quinazoli 4(3H)-one,
(R)-2-(3-(2-Amino-9-methyl-9H^urin-6-yl)thiazolidin-4-yl)-5-chloro-3-(o-tolyl)qui
4(3H)-one,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(2-cyclopropylphenyl)quinazolin-4(3H)- one,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(1 -methyl-1 H-pyrazol-5-yl)quinazolin- 4(3H)-one,
(R)-4-Amino-6-(4-(5-chloro-3-(1 -methyl-1 H-pyrazol-5-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-4-Amino-6-(4-(5-chloro-3-(5-methylthiophen-2-yl)-4-oxo-3,4-dihydroquinazolin-2- yl)thiazolidin-3-yl)pyrimidine-5-carbonitrile,
(R)-2-(3-(9H-Purin-6-yl)thiazolidin-4-yl)-5-chloro-3-(5-methylthiophen-2-yl)quinazolin one,
4-Amino-6-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
5-Chloro-2-((2S,4S)-4-methoxy-1 -(9H^urin-6-yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)- one,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4- methoxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-4-methoxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-^^ yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-methoxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-^^ yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(R,S)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H-pyrazol-5-yl)-5-chloro-3-phenylquinazolin-4(3H)- one,
(R,S)-4-Amino-6-(5-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R,S)-2-Amino-4-(5-(5-chloro-4-oxo-3^henyl-3,4-dihydroquinazolin-2-yl)-4,5-dih
pyrazol-1 -yl)-6-methylpyrimidine-5-carbonitrile,
(R,S)-4-Amino-6-(5-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R,S)-4-Amino-6-(5-(5-chloro-3-morpholino-4-oxo-3,4-dihydroquinazolin-2-yl)-4,5-dih pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H^yrazol-5-yl)-5-chloro-3^henylquinazolin-4(3H)- one,
(S or R)-2-(1 -(9H-Purin-6-yl)-4,5-dihydro-1 H^yrazol-5-yl)-5-chloro-3^henylquinazolin-4(3H)- one,
(S or R)-4-Amino-6-(5-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)-4,5- dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(S or R)-2-Amino-4-methyl-6-(5-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazoli yl)-4,5-dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-4-Amino-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)-4,5- dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-2-Amino-4-methyl-6-(5-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2^ yl)-4,5-dihydro-1 H-pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(S or R)-4-Amino-6-(5-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
(R or S)-4-Amino-6-(5-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4,5-dihydro-1 H- pyrazol-1 -yl)pyrimidine-5-carbonitrile,
N-((3S,5S)-5-(5-Chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-1 -(9H-purin-6- yl)pyrrolidin-3-yl)methanesulfonamide,
((S)-4-Amino-6-(3-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
(S)-2-(2-(9H-Purin-6-yl)isoxazolidin-3-yl)-5-chloro-3-phenylquinazolin-4(3H)-one,
(S)-5-Chloro-3-phenyl-2-(2-(thiazolo[5,4-d]pyrimidin-7-yl)isoxazolidin-3-yl)quinazolin-4(^ one,
(S)-2-(2-(9H-Purin-6-yl)isoxazolidin-3-yl)-5-chloro-3-(o-tolyl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(3-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(3-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)isoxazolidin-2- yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
5-Chloro-2-((2S,4S)-4-hydroxy-1 -(9H-purin-6-yl)pyrrolidm^^
one,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-3-cyclopropyl-4-oxo-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(o-tolyl)-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-4-hydroxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-((2S,4S)-1 -(5-Aminothiazolo[5,4-d]pyrimidin-7-yl)-4-hydroxypyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
2-Amino-4-((2S,4S)-2-(5-(difluoromethoxy)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3^henyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-4-oxo-3-(pyridin-3-yl)-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(3-(3,5-difluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin
2-yl)-4-hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(3-morpholino-4-oxo-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(3-(2-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)-4-hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5,6-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(6-fluoro-5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(6-fluoro-5-methyl-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin-2- yl)-4-hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4-dihydroquinazolin yl)-4-hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-4-hydroxy-2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-4-hydroxy-2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-chloro-6-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-chloro-6-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-((2S,4S)-4-Hydroxy-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenyl-5-(trifluoromethyl)quinazolin- 4(3H)-one,
2-((2S,4S)-1 -(2-Amino-9H-purin-6-yl)-4-hydroxypyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
4-Amino-6-((2S,4S)-2-(5,6-difluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
4-Amino-6-((2S,4S)-2-(5-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-((2S,4S)-2-(5-cyclopropyl-6-fluoro-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)-4- hydroxypyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile, 5-Chloro-2-((2S,4S)-4- (difluoromethoxy)-1 -(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenylquinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-hydroxy-4-oxo-3-phenyl-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 - yl)pyrimidine-5-carbonitrile,
(S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-phenylquinazolin- 4(3H)-one,
(S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-morpholino- quinazolin-4(3H)-one,
(S)-2-(1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)pyrrolidin-2-yl)-5-chloro-3-(o- tolyl)quinazolin-4(3H)-one,
2-((2S,4S)-1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)-4-methoxypyrrolidin-2-yl)-5-chloro-3- phenylquinazolin-4(3H)-one,
2-((2S,4S)-1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)-4-hydroxypyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
2-((2S,4S)-1 -(2-Aminoimidazo[2,1 -f][1 ,2,4]triazin-4-yl)-4-hydroxypyrrolidin-2-yl)-5-chloro-3- phenylquinazolin-4(3H)-one,
2-((S)-1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((S)-1 -(methylsulfonyl)pyrrolid yl)quinazolin-4(3H)-one,
(S)-Methyl 3-(2-((S)-1-(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4-oxoquinazolin-3(4H)- yl)pyrrolidine-1 -carboxylate,
(R)-Methyl 3-(2-((S)-1-(9H-purin-6-yl)pyrrolidin-2-yl)-5-chloro-4-oxoquinazolin-3(4H)- yl)pyrrolidine-1 -carboxylate,
2-((S)-1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((R)-1 -(methylsulfonyl)pyrrolidi yl)quinazolin-4(3H)-one,
2-((S)-1-(9H-Purin-6-yl)pyrrolidin-2-yl)-5-chloro-3-((R)-1-(cyclopropylsulfonyl)pyrrolidin-3 yl)quinazolin-4(3H)-one,
(S)-4-Amino-6-(2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-methyl-6-(2-(5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-4-Amino-6-(2-(6-fluoro-5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
(S)-2-Amino-4-(2-(6-fluoro-5-(methylsulfonyl)-4-oxo-3-phenyl-3,4-dihydroquinazolin-2- yl)pyrrolidin-1 -yl)-6-methylpyrimidine-5-carbonitrile,
2-Amino-4-(difluoromethyl)-6-((2S,4S)-4-hydroxy-2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,^ dihydroquinazolin-2-yl)pyrrolidin-1-yl)pyrimidm^
(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin-2-yl)pyrrolidin-1 -yl)-6- methylpyrimidine-5-carbonitrile,
2-((2S,4S)-4-Fluoro-1-(9H-purin-6-yl)pyrrolidin-2-yl)-3-phenyl-5-(trifluoromethyl)quinazol^ 4(3H)-one,
2-((2S,4S)-1-(2-Amino-9H-purin-6-yl)-4-fluoropyrrolidin-2-yl)-3-phenyl-5- (trifluoromethyl)quinazolin-4(3H)-one,
4-Amino-6-((2S,4S)-4-fluoro-2-(4-oxo-3-phenyl-5-(trifluoromethyl)-3,4-dihydroquinazolin yl)pyrrolidin-1 -yl)pyrimidine-5-carbonitrile,
2-((2S,4S)-1-(2-Amino-9H-purin-6-yl)-4-fluoropyrrolidin-2-yl)-3-(pyridin-3-yl)-5- (trifluoromethyl)quinazolin-4(3H)-one,
2-Amino-4-((2S,4S)-4-fluoro-2-(4-oxo-3-(pyridin-3-yl)-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6-methylpyrimidine-5-carbonitrile,
2-((2S,4S)-1-(2-Amino-9H-purin-6-yl)-4-fluoropyrrolidin-2-yl)-3-(3-fluorophenyl)-5-
(trifluoromethyl)quinazolin-4(3H)-one, or
2-Amino-4-((2S,4S)-4-fluoro-2-(3-(3-fluorophenyl)-4-oxo-5-(trifluoromethyl)-3,4- dihydroquinazolin-2-yl)pyrrolidin-1-yl)-6-methylpyrimidine-5-carbonitrile,
or a pharmaceutically acceptable salt thereof.
9. A pharmaceutical composition comprising a therapeutically effective amount of a compound according to any one of claims 1 to 8 or a pharmaceutically acceptable salt thereof and one or more pharmaceutically acceptable carriers.
10. A combination comprising a therapeutically effective amount of a compound according to any one of claims 1 to 8 or a pharmaceutically acceptable salt thereof and one or more therapeutically active co-agents.
1 1 . A method of modulating the activity of the class I PI3 kinases, in a subject, wherein the method comprises administering to the subject a therapeutically effective amount of a compound according to any one of claims 1 to 8 or a pharmaceutically acceptable salt thereof.
12. A method of treating a disorder or a disease selected from rheumatoid arthritis (RA), pemphigus vulgaris (PV), endemic form of Brazilian pemphigus (Fogo selvagem), idiopathic thrombocytopenia purpura (ITP), thrombotic thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA-associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection, cancers of
haematopoietic origin, severe and cerebral malaria, trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis comprising administering to a subject a therapeutically effective amount of a compound according to any one of claims 1 to 8 or a pharmaceutically acceptable salt thereof.
13. A compound according to any one of claims 1 to 8 or a pharmaceutically acceptable salt thereof, for use as a medicament.
14. A compound according to any one of claims 1 to 8 or a pharmaceutically acceptable salt thereof, for use in the treatment of a disorder or a disease selected from rheumatoid arthritis (RA), pemphigus vulgaris (PV), endemic form of Brazilian pemphigus (Fogo selvagem), idiopathic thrombocytopenia purpura (ITP), thrombotic thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired
hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA-associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection, cancers of haematopoietic origin, severe and cerebral malaria, trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis.
15. Use of a compound according to anyone of claims 1 to 8 or a pharmeceutically acceptable salt therof in the manufacture of a medicament for the treatment of a disorder or a disease selected from rheumatoid arthritis (RA), pemphigus vulgaris (PV), endemic form of Brazilian pemphigus (Fogo selvagem), idiopathic thrombocytopenia purpura (ITP), thrombotic thrombocytopenic purpura (TTP), autoimmune hemolytic anemia (AIHA), acquired hemophilia type A (AHA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), myasthenia gravis (MG), Sjogren's syndrome (SS), ANCA- associated vasculitides, cryoglobulinemia, chronic autoimmune urticaria (CAU), allergy (atopic dermatitis, contact dermatitis, allergic rhinitis), goodpasture's syndrome, transplant rejection, cancers of haematopoietic origin, severe and cerebral malaria, trypanosomiasis, leishmaniasis, toxoplasmosis and neurocysticercosis.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361766920P | 2013-02-20 | 2013-02-20 | |
| US61/766,920 | 2013-02-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014128612A1 true WO2014128612A1 (en) | 2014-08-28 |
Family
ID=50236229
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2014/059071 WO2014128612A1 (en) | 2013-02-20 | 2014-02-18 | Quinazolin-4-one derivatives |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2014128612A1 (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015010641A1 (en) * | 2013-07-24 | 2015-01-29 | Novartis Ag | Substituted quinazolin-4-one derivatives |
| WO2015191743A1 (en) * | 2014-06-13 | 2015-12-17 | Gilead Sciences, Inc. | Quinazolinone derivatives as phosphatidylinositol 3-kinase inhibitors |
| CN106459008A (en) * | 2014-06-24 | 2017-02-22 | 吉利德科学公司 | Phosphatidylinositol 3-kinase inhibitors |
| US9637488B2 (en) | 2015-01-29 | 2017-05-02 | Fuqiang Ruan | Heterocyclic compounds as inhibitors of class I PI3KS |
| US9944639B2 (en) | 2014-07-04 | 2018-04-17 | Lupin Limited | Quinolizinone derivatives as PI3K inhibitors |
| US10092563B2 (en) | 2014-06-13 | 2018-10-09 | Gilead Sciences, Inc. | Phosphatidylinositol 3-kinase inhibitors |
| US10221197B2 (en) | 2014-06-13 | 2019-03-05 | Gilead Sciences, Inc. | Phosphatidylinositol 3-kinase inhibitors |
| US10751339B2 (en) | 2018-01-20 | 2020-08-25 | Sunshine Lake Pharma Co., Ltd. | Substituted aminopyrimidine compounds and methods of use |
| JP2020529418A (en) * | 2017-07-31 | 2020-10-08 | ザ トラスティーズ オブ コロンビア ユニバーシティ イン ザ シティ オブ ニューヨークThe Trustees Of Columbia University In The City Of New York | Compounds, Compositions and Methods for Treating T-Cell Acute Lymphoblastic Leukemia |
| US11021467B2 (en) | 2014-06-13 | 2021-06-01 | Gilead Sciences, Inc. | Phosphatidylinositol 3-kinase inhibitors |
| CN113512042A (en) * | 2020-04-09 | 2021-10-19 | 成都赜灵生物医药科技有限公司 | Substituted quinazoline-4-ketone compound and preparation method and application thereof |
| US11969428B2 (en) | 2020-11-17 | 2024-04-30 | Thomas Diacovo | Methods and pharmaceutical compositions for treating lymphoid malignancy |
Citations (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4100274A (en) | 1976-05-11 | 1978-07-11 | Imperial Chemical Industries Limited | Polypeptide |
| US4636505A (en) | 1982-07-23 | 1987-01-13 | Imperial Chemical Industries Plc | Amide derivatives |
| US4659516A (en) | 1983-10-12 | 1987-04-21 | Imperial Chemical Industries Plc | Steroid derivatives |
| US5010099A (en) | 1989-08-11 | 1991-04-23 | Harbor Branch Oceanographic Institution, Inc. | Discodermolide compounds, compositions containing same and method of preparation and use |
| US5093330A (en) | 1987-06-15 | 1992-03-03 | Ciba-Geigy Corporation | Staurosporine derivatives substituted at methylamino nitrogen |
| EP0520722A1 (en) | 1991-06-28 | 1992-12-30 | Zeneca Limited | Therapeutic preparations containing quinazoline derivatives |
| EP0564409A1 (en) | 1992-04-03 | 1993-10-06 | Ciba-Geigy Ag | Pyrimidin derivatives and process for their preparation |
| EP0566226A1 (en) | 1992-01-20 | 1993-10-20 | Zeneca Limited | Quinazoline derivatives |
| WO1995003283A1 (en) | 1993-07-19 | 1995-02-02 | Zeneca Limited | Quinazoline derivatives and their use as anti-cancer agents |
| WO1996030347A1 (en) | 1995-03-30 | 1996-10-03 | Pfizer Inc. | Quinazoline derivatives |
| WO1996033980A1 (en) | 1995-04-27 | 1996-10-31 | Zeneca Limited | Quinazoline derivatives |
| WO1997002266A1 (en) | 1995-07-06 | 1997-01-23 | Novartis Ag | Pyrrolopyrimidines and processes for the preparation thereof |
| EP0787722A1 (en) | 1996-02-05 | 1997-08-06 | American Cyanamid Company | Substituted quinazoline derivatives |
| WO1997030034A1 (en) | 1996-02-14 | 1997-08-21 | Zeneca Limited | Quinazoline derivatives as antitumor agents |
| WO1997038983A1 (en) | 1996-04-12 | 1997-10-23 | Warner-Lambert Company | Irreversible inhibitors of tyrosine kinases |
| WO1997049688A1 (en) | 1996-06-24 | 1997-12-31 | Pfizer Inc. | Phenylamino-substituted tricyclic derivatives for treatment of hyperproliferative diseases |
| WO1998008849A1 (en) | 1996-08-30 | 1998-03-05 | Novartis Aktiengesellschaft | Method for producing epothilones, and intermediate products obtained during the production process |
| WO1998010121A1 (en) | 1996-09-06 | 1998-03-12 | Obducat Ab | Method for anisotropic etching of structures in conducting materials |
| WO1998010767A2 (en) | 1996-09-13 | 1998-03-19 | Sugen, Inc. | Use of quinazoline derivatives for the manufacture of a medicament in the treatment of hyperproliferative skin disorders |
| EP0837063A1 (en) | 1996-10-17 | 1998-04-22 | Pfizer Inc. | 4-Aminoquinazoline derivatives |
| US5747498A (en) | 1996-05-28 | 1998-05-05 | Pfizer Inc. | Alkynyl and azido-substituted 4-anilinoquinazolines |
| WO1998022461A1 (en) | 1996-11-18 | 1998-05-28 | GESELLSCHAFT FüR BIOTECHNOLOGISCHE FORSCHUNG MBH (GBF) | Epothilone c, d, e and f, production process, and their use as cytostatic as well as phytosanitary agents |
| WO1998025929A1 (en) | 1996-12-13 | 1998-06-18 | Novartis Ag | Epothilone analogs |
| US5843901A (en) | 1995-06-07 | 1998-12-01 | Advanced Research & Technology Institute | LHRH antagonist peptides |
| WO1999003854A1 (en) | 1997-07-18 | 1999-01-28 | Novartis Ag | Crystal modification of a n-phenyl-2-pyrimidineamine derivative, processes for its manufacture and its use |
| WO1999017804A1 (en) | 1997-10-03 | 1999-04-15 | Pharmacia & Upjohn S.P.A. | Polymeric derivatives of camptothecins |
| WO1999043653A1 (en) | 1998-02-25 | 1999-09-02 | Sloan-Kettering Institute For Cancer Research | Synthesis of epothilones, intermediates thereto and analogues therof |
| WO2000009495A1 (en) | 1998-08-11 | 2000-02-24 | Novartis Ag | Isoquinoline derivatives with angiogenesis inhibiting activity |
| WO2000031247A2 (en) | 1998-11-20 | 2000-06-02 | Kosan Biosciences, Inc. | Recombinant methods and materials for producing epothilone and epothilone derivatives |
| US6194181B1 (en) | 1998-02-19 | 2001-02-27 | Novartis Ag | Fermentative preparation process for and crystal forms of cytostatics |
| WO2002022577A2 (en) | 2000-09-01 | 2002-03-21 | Novartis Ag | Hydroxamate derivatives useful as deacetylase inhibitors |
| WO2002038561A1 (en) | 2000-11-07 | 2002-05-16 | Novartis Ag | Indolylmaleimide derivatives as protein kinase c inhibitors |
| WO2002092599A1 (en) | 2001-05-14 | 2002-11-21 | Novartis Ag | 4-amino-5-phenyl-7-cyclobutyl-pyrrolo (2,3-d) pyrimidine derivatives |
| WO2003013541A1 (en) | 2001-08-07 | 2003-02-20 | Novartis Ag | 4-amino-6-phenyl-pyrrolo[2,3-d]pyrimidine derivatives |
| WO2003082859A1 (en) | 2002-04-03 | 2003-10-09 | Novartis Ag | Indolylmaleimide derivatives |
| WO2004052359A1 (en) | 2002-12-09 | 2004-06-24 | The Board Of Regents Of The University Of Texas System | Methods for selectively inhibiting janus tyrosine kinase 3 (jak3) |
| WO2004078163A2 (en) | 2003-02-28 | 2004-09-16 | Transform Pharmaceuticals, Inc. | Pharmaceutical co-crystal compositions of drugs such as carbamazepine, celecoxib, olanzapine, itraconazole, topiramate, modafinil, 5-fluorouracil, hydrochlorothiazide, acetaminophen, aspirin, flurbiprofen, phenytoin and ibuprofen |
| WO2005066156A1 (en) | 2004-01-12 | 2005-07-21 | Cytopia Research Pty Ltd | Selective kinase inhibitors |
| WO2005113556A1 (en) * | 2004-05-13 | 2005-12-01 | Icos Corporation | Quinazolinones as inhibitors of human phosphatidylinositol 3-kinase delta |
| WO2006122806A2 (en) | 2005-05-20 | 2006-11-23 | Novartis Ag | 1,3-dihydro-imidazo [4,5-c] quinolin-2-ones as lipid kinase inhibitors |
| WO2010123931A1 (en) * | 2009-04-20 | 2010-10-28 | Calistoga Pharmaceuticals Inc. | Methods of treatment for solid tumors |
| WO2011011550A1 (en) * | 2009-07-21 | 2011-01-27 | Calistoga Pharmaceuticals Inc. | Treatment of liver disorders with pi3k inhibitors |
-
2014
- 2014-02-18 WO PCT/IB2014/059071 patent/WO2014128612A1/en active Application Filing
Patent Citations (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4100274A (en) | 1976-05-11 | 1978-07-11 | Imperial Chemical Industries Limited | Polypeptide |
| US4636505A (en) | 1982-07-23 | 1987-01-13 | Imperial Chemical Industries Plc | Amide derivatives |
| US4659516A (en) | 1983-10-12 | 1987-04-21 | Imperial Chemical Industries Plc | Steroid derivatives |
| US5093330A (en) | 1987-06-15 | 1992-03-03 | Ciba-Geigy Corporation | Staurosporine derivatives substituted at methylamino nitrogen |
| US5010099A (en) | 1989-08-11 | 1991-04-23 | Harbor Branch Oceanographic Institution, Inc. | Discodermolide compounds, compositions containing same and method of preparation and use |
| EP0520722A1 (en) | 1991-06-28 | 1992-12-30 | Zeneca Limited | Therapeutic preparations containing quinazoline derivatives |
| EP0566226A1 (en) | 1992-01-20 | 1993-10-20 | Zeneca Limited | Quinazoline derivatives |
| EP0564409A1 (en) | 1992-04-03 | 1993-10-06 | Ciba-Geigy Ag | Pyrimidin derivatives and process for their preparation |
| WO1995003283A1 (en) | 1993-07-19 | 1995-02-02 | Zeneca Limited | Quinazoline derivatives and their use as anti-cancer agents |
| WO1996030347A1 (en) | 1995-03-30 | 1996-10-03 | Pfizer Inc. | Quinazoline derivatives |
| WO1996033980A1 (en) | 1995-04-27 | 1996-10-31 | Zeneca Limited | Quinazoline derivatives |
| US5843901A (en) | 1995-06-07 | 1998-12-01 | Advanced Research & Technology Institute | LHRH antagonist peptides |
| WO1997002266A1 (en) | 1995-07-06 | 1997-01-23 | Novartis Ag | Pyrrolopyrimidines and processes for the preparation thereof |
| EP0787722A1 (en) | 1996-02-05 | 1997-08-06 | American Cyanamid Company | Substituted quinazoline derivatives |
| WO1997030034A1 (en) | 1996-02-14 | 1997-08-21 | Zeneca Limited | Quinazoline derivatives as antitumor agents |
| WO1997038983A1 (en) | 1996-04-12 | 1997-10-23 | Warner-Lambert Company | Irreversible inhibitors of tyrosine kinases |
| US5747498A (en) | 1996-05-28 | 1998-05-05 | Pfizer Inc. | Alkynyl and azido-substituted 4-anilinoquinazolines |
| WO1997049688A1 (en) | 1996-06-24 | 1997-12-31 | Pfizer Inc. | Phenylamino-substituted tricyclic derivatives for treatment of hyperproliferative diseases |
| WO1998008849A1 (en) | 1996-08-30 | 1998-03-05 | Novartis Aktiengesellschaft | Method for producing epothilones, and intermediate products obtained during the production process |
| WO1998010121A1 (en) | 1996-09-06 | 1998-03-12 | Obducat Ab | Method for anisotropic etching of structures in conducting materials |
| WO1998010767A2 (en) | 1996-09-13 | 1998-03-19 | Sugen, Inc. | Use of quinazoline derivatives for the manufacture of a medicament in the treatment of hyperproliferative skin disorders |
| EP0837063A1 (en) | 1996-10-17 | 1998-04-22 | Pfizer Inc. | 4-Aminoquinazoline derivatives |
| WO1998022461A1 (en) | 1996-11-18 | 1998-05-28 | GESELLSCHAFT FüR BIOTECHNOLOGISCHE FORSCHUNG MBH (GBF) | Epothilone c, d, e and f, production process, and their use as cytostatic as well as phytosanitary agents |
| WO1998025929A1 (en) | 1996-12-13 | 1998-06-18 | Novartis Ag | Epothilone analogs |
| WO1999003854A1 (en) | 1997-07-18 | 1999-01-28 | Novartis Ag | Crystal modification of a n-phenyl-2-pyrimidineamine derivative, processes for its manufacture and its use |
| WO1999017804A1 (en) | 1997-10-03 | 1999-04-15 | Pharmacia & Upjohn S.P.A. | Polymeric derivatives of camptothecins |
| US6194181B1 (en) | 1998-02-19 | 2001-02-27 | Novartis Ag | Fermentative preparation process for and crystal forms of cytostatics |
| WO1999043653A1 (en) | 1998-02-25 | 1999-09-02 | Sloan-Kettering Institute For Cancer Research | Synthesis of epothilones, intermediates thereto and analogues therof |
| WO2000009495A1 (en) | 1998-08-11 | 2000-02-24 | Novartis Ag | Isoquinoline derivatives with angiogenesis inhibiting activity |
| WO2000031247A2 (en) | 1998-11-20 | 2000-06-02 | Kosan Biosciences, Inc. | Recombinant methods and materials for producing epothilone and epothilone derivatives |
| WO2002022577A2 (en) | 2000-09-01 | 2002-03-21 | Novartis Ag | Hydroxamate derivatives useful as deacetylase inhibitors |
| WO2002038561A1 (en) | 2000-11-07 | 2002-05-16 | Novartis Ag | Indolylmaleimide derivatives as protein kinase c inhibitors |
| WO2002092599A1 (en) | 2001-05-14 | 2002-11-21 | Novartis Ag | 4-amino-5-phenyl-7-cyclobutyl-pyrrolo (2,3-d) pyrimidine derivatives |
| WO2003013541A1 (en) | 2001-08-07 | 2003-02-20 | Novartis Ag | 4-amino-6-phenyl-pyrrolo[2,3-d]pyrimidine derivatives |
| WO2003082859A1 (en) | 2002-04-03 | 2003-10-09 | Novartis Ag | Indolylmaleimide derivatives |
| WO2004052359A1 (en) | 2002-12-09 | 2004-06-24 | The Board Of Regents Of The University Of Texas System | Methods for selectively inhibiting janus tyrosine kinase 3 (jak3) |
| WO2004078163A2 (en) | 2003-02-28 | 2004-09-16 | Transform Pharmaceuticals, Inc. | Pharmaceutical co-crystal compositions of drugs such as carbamazepine, celecoxib, olanzapine, itraconazole, topiramate, modafinil, 5-fluorouracil, hydrochlorothiazide, acetaminophen, aspirin, flurbiprofen, phenytoin and ibuprofen |
| WO2005066156A1 (en) | 2004-01-12 | 2005-07-21 | Cytopia Research Pty Ltd | Selective kinase inhibitors |
| WO2005113556A1 (en) * | 2004-05-13 | 2005-12-01 | Icos Corporation | Quinazolinones as inhibitors of human phosphatidylinositol 3-kinase delta |
| WO2006122806A2 (en) | 2005-05-20 | 2006-11-23 | Novartis Ag | 1,3-dihydro-imidazo [4,5-c] quinolin-2-ones as lipid kinase inhibitors |
| WO2010123931A1 (en) * | 2009-04-20 | 2010-10-28 | Calistoga Pharmaceuticals Inc. | Methods of treatment for solid tumors |
| WO2011011550A1 (en) * | 2009-07-21 | 2011-01-27 | Calistoga Pharmaceuticals Inc. | Treatment of liver disorders with pi3k inhibitors |
Non-Patent Citations (33)
| Title |
|---|
| "MRC Laboratory of Molecular Biology", November 2003, ROGER WILLIAMS LAB |
| "Protective Groups in Organic Chemistry", 1973, PLENUM PRESS |
| "Remington's Pharmaceutical Sciences", 1990, MACK PRINTING COMPANY, pages: 1289 - 1329 |
| AREVALO ET AL.: "Ocular Toxoplasmosis in the developing world", INTERNAT. OPHTHAL. CLIN, 2010 |
| BOTTIEAU ET AL.: "Therapy of vector-bome protozoan infections in nonendemic settings", EXPERT REV. ANTI INFECT. THER., 2011 |
| BOTTIEAU ET AL.: "Therapy of vector-borne protozoan infections in nonendemic settings", EXPERT REV. ANTI INFECT. THER., 2011 |
| CARRENO ET AL., ANNU REV IMMUNOL., vol. 20, 2002, pages 29 |
| FOUKAS ET AL., NATURE, vol. 441, 2006, pages 366 |
| FRANKLIN ET AL.: "Therapeutical targeting of nucleic acid-sensing TLRs prevents experimental cerebral malaria", PNAS, 2011 |
| GERARD ET AL., NAT. IMMUNOL., vol. 2, 2001, pages 108 |
| GHIGO ET AL., BIOESSAYS, vol. 32, 2010, pages 185 - 196 |
| GOWDA ET AL.: "The Nucleosome is the TLR9-specific Immunostimulatory component of plasmodium falciparum that activates DCs", PLOS ONE, June 2011 (2011-06-01) |
| HEDAYAT ET AL.: "Targeting of TLRs: a decade of progress in combating infectious disease", LANCET INFECTIOUS DISEASE, 2011 |
| HIGGINS ET AL.: "Immunopathogenesis of falciparum malaria: implications for adjunctive therapy in the management of severe and cerebral malaria", EXPERT REV. ANTI INFECT. THER., 2011 |
| HOUBEN-WEYL: "Methoden der organischen Chemie", vol. 15/1, 1974, GEORG-THIEME-VERLAG |
| HOUBEN-WEYL: "Methods of Organic Synthesis", vol. 21, 1952, THIEME |
| KWAI ET AL.: "TLRs and their crosstalk with other innate receptors in infection and immunity", IMMUNITY, May 2011 (2011-05-01) |
| LAFFARGUE ET AL., IMMUNITY, vol. 16, 2002, pages 441 - 451 |
| LIU ET AL., BLOOD, vol. 110, 2007, pages 1191 |
| MAIRA ET AL., EXPERT OPIN. THER. TARGETS, vol. 12, 2008, pages 223 |
| MARONE ET AL., BIOCHIMICA ET BIOPHYSICA ACTA, vol. 1784, 2008, pages 159 |
| MARTIN; CHAN, ANNU. REV. IMMUNOL., vol. 24, 2006, pages 467 |
| MISHRA ET AL.: "TLRs in CNS Parasitic infections", CURR TOP MICRO IMM, 2009 |
| OKKENHAUG ET AL., SCIENCE, vol. 297, 2002, pages 1031 |
| OKKENHAUG; WYMANN, NATURE REV. IMMUNOL., vol. 3, 2003, pages 317 |
| PACOLD M. E. ET AL., CELL, vol. 103, 2000, pages 931 - 943 |
| PEIXOTO-RANGEL ET AL.: "Candidate gene analysis of ocular toxoplasmosis in Brazil: evidence for a role for TLR9", MEM INST OSWALDO CRUZ, 2009 |
| PELLEGRINI ET AL., THE ROLE OF TLRS AND ADOPTIVE IMMUNITY IN THE DEVELOPMENT OF PROTECTIVE OR PATHOLOGICAL IMMUNE RESPONSE TRIGGERED BY THE TRYPANOSOMA CRUZI PROTOZOAN FUTURE MICROBIOL, 2011 |
| REIF ET AL., J. IMMUNOL., vol. 173, 2004, pages 2236 - 2240 |
| SANCHO ET AL., TRENDS IMMUNOL., vol. 26, 2005, pages 136 |
| SCHOFIELD ET AL.: "Immunological processes in malaria pathogenesis", NAT REV IMM, 2005 |
| SCHOFILED L, INTRAVASCULAR INFILTRATES AND ORGAN-SPECIFIC INFLAMMATION IN MALARIA PATHOGENESIS |
| THEODORA W. GREENE: "Protective Groups in Organic Synthesis", 1981, JOHN WILEY & SONS |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9840498B2 (en) | 2013-07-24 | 2017-12-12 | Novartis Ag | Substituted quinazolin-4-one derivatives |
| EA031430B1 (en) * | 2013-07-24 | 2018-12-28 | Новартис Аг | Substituted quinazolin-4-one derivatives |
| WO2015010641A1 (en) * | 2013-07-24 | 2015-01-29 | Novartis Ag | Substituted quinazolin-4-one derivatives |
| US10092563B2 (en) | 2014-06-13 | 2018-10-09 | Gilead Sciences, Inc. | Phosphatidylinositol 3-kinase inhibitors |
| US11021467B2 (en) | 2014-06-13 | 2021-06-01 | Gilead Sciences, Inc. | Phosphatidylinositol 3-kinase inhibitors |
| JP2017521380A (en) * | 2014-06-13 | 2017-08-03 | ギリアード サイエンシーズ, インコーポレイテッド | Quinazolinone derivatives as phosphatidylinositol 3-kinase inhibitors |
| CN106459012A (en) * | 2014-06-13 | 2017-02-22 | 吉利德科学公司 | Quinazolinone derivatives as phosphatidylinositol 3-kinase inhibitors |
| EP3154958B1 (en) * | 2014-06-13 | 2020-10-07 | Gilead Sciences, Inc. | Phosphatidylinositol 3-kinase inhibitors |
| WO2015191743A1 (en) * | 2014-06-13 | 2015-12-17 | Gilead Sciences, Inc. | Quinazolinone derivatives as phosphatidylinositol 3-kinase inhibitors |
| US10221197B2 (en) | 2014-06-13 | 2019-03-05 | Gilead Sciences, Inc. | Phosphatidylinositol 3-kinase inhibitors |
| US10308639B2 (en) | 2014-06-13 | 2019-06-04 | Gilead Sciences, Inc. | Phosphatidylinositol 3-kinase inhibitors |
| US9765060B2 (en) | 2014-06-24 | 2017-09-19 | Gilead Sciences, Inc. | Phosphatidylinositol 3-kinase inhibitors |
| CN106459008A (en) * | 2014-06-24 | 2017-02-22 | 吉利德科学公司 | Phosphatidylinositol 3-kinase inhibitors |
| US9944639B2 (en) | 2014-07-04 | 2018-04-17 | Lupin Limited | Quinolizinone derivatives as PI3K inhibitors |
| US9637488B2 (en) | 2015-01-29 | 2017-05-02 | Fuqiang Ruan | Heterocyclic compounds as inhibitors of class I PI3KS |
| JP2020529418A (en) * | 2017-07-31 | 2020-10-08 | ザ トラスティーズ オブ コロンビア ユニバーシティ イン ザ シティ オブ ニューヨークThe Trustees Of Columbia University In The City Of New York | Compounds, Compositions and Methods for Treating T-Cell Acute Lymphoblastic Leukemia |
| US10751339B2 (en) | 2018-01-20 | 2020-08-25 | Sunshine Lake Pharma Co., Ltd. | Substituted aminopyrimidine compounds and methods of use |
| CN113512042A (en) * | 2020-04-09 | 2021-10-19 | 成都赜灵生物医药科技有限公司 | Substituted quinazoline-4-ketone compound and preparation method and application thereof |
| US11969428B2 (en) | 2020-11-17 | 2024-04-30 | Thomas Diacovo | Methods and pharmaceutical compositions for treating lymphoid malignancy |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2014295538B2 (en) | Substituted quinazolin-4-one derivatives | |
| WO2014128612A1 (en) | Quinazolin-4-one derivatives | |
| US9763952B2 (en) | Dihydro-benzo-oxazine and dihydro-pyrido-oxazine derivatives | |
| WO2012004299A1 (en) | Tetrahydro-pyrido-pyrimidine derivatives | |
| KR102184069B1 (en) | Solid form of dihydro-pyrido-oxazine derivative | |
| WO2013093850A1 (en) | Quinoline derivatives | |
| NZ624872B2 (en) | Dihydro-benzo-oxazine and dihydro-pyrido-oxazine derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14708331 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 14708331 Country of ref document: EP Kind code of ref document: A1 |