WO2013000479A1 - Fouling control coating compositions - Google Patents
Fouling control coating compositions Download PDFInfo
- Publication number
- WO2013000479A1 WO2013000479A1 PCT/DK2012/050228 DK2012050228W WO2013000479A1 WO 2013000479 A1 WO2013000479 A1 WO 2013000479A1 DK 2012050228 W DK2012050228 W DK 2012050228W WO 2013000479 A1 WO2013000479 A1 WO 2013000479A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polysiloxane
- poly
- hydrophilic
- biocides
- polymer moieties
- Prior art date
Links
- 0 CCC(C)(CC*)N(C(C)(C)NCC(C)(C)*)I Chemical compound CCC(C)(CC*)N(C(C)(C)NCC(C)(C)*)I 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D183/00—Coating compositions based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon, with or without sulfur, nitrogen, oxygen, or carbon only; Coating compositions based on derivatives of such polymers
- C09D183/04—Polysiloxanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/16—Antifouling paints; Underwater paints
- C09D5/1656—Antifouling paints; Underwater paints characterised by the film-forming substance
- C09D5/1662—Synthetic film-forming substance
- C09D5/1675—Polyorganosiloxane-containing compositions
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/42—Block-or graft-polymers containing polysiloxane sequences
- C08G77/46—Block-or graft-polymers containing polysiloxane sequences containing polyether sequences
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D183/00—Coating compositions based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon, with or without sulfur, nitrogen, oxygen, or carbon only; Coating compositions based on derivatives of such polymers
- C09D183/10—Block or graft copolymers containing polysiloxane sequences
- C09D183/12—Block or graft copolymers containing polysiloxane sequences containing polyether sequences
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/14—Paints containing biocides, e.g. fungicides, insecticides or pesticides
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/16—Antifouling paints; Underwater paints
- C09D5/1687—Use of special additives
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/14—Polysiloxanes containing silicon bound to oxygen-containing groups
- C08G77/16—Polysiloxanes containing silicon bound to oxygen-containing groups to hydroxyl groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/20—Oxides; Hydroxides
- C08K3/22—Oxides; Hydroxides of metals
- C08K2003/2265—Oxides; Hydroxides of metals of iron
- C08K2003/2272—Ferric oxide (Fe2O3)
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/09—Carboxylic acids; Metal salts thereof; Anhydrides thereof
- C08K5/098—Metal salts of carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/56—Organo-metallic compounds, i.e. organic compounds containing a metal-to-carbon bond
- C08K5/57—Organo-tin compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/02—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group
- C08L2205/025—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group containing two or more polymers of the same hierarchy C08L, and differing only in parameters such as density, comonomer content, molecular weight, structure
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/03—Polymer mixtures characterised by other features containing three or more polymers in a blend
- C08L2205/035—Polymer mixtures characterised by other features containing three or more polymers in a blend containing four or more polymers in a blend
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/31504—Composite [nonstructural laminate]
- Y10T428/31652—Of asbestos
- Y10T428/31663—As siloxane, silicone or silane
Definitions
- the present invention relates to novel fouling control coating compositions.
- silicone formulations rely on physical means, this being mainly a factor of modulus of elasticity and surface tension to create a low bio-fouling surface.
- PDMS polydimethylsiloxane
- WO 2007/053163 discloses an antifouling material that may include one or more of a number of suitable copolymers (e.g. block copolymers, graft copolymers, etc.) which provide biocidal and/or fouling release characteristics.
- the copolymers may include a polysiloxane backbone with one or more polymers grafted onto the polysiloxane backbone. Such grafted polymers may carry biocidal groups.
- WO 2008/132195 discloses an antifouling coating composition
- a curable polymer e.g. an organosiloxane-containing polymer
- an organosilicone polymer e.g. an organosilicone-containing polymer
- WO 2008/132196 discloses a method to physically deter marine fouling which method involves forming on the substrate a coating composition comprising curable polyorgano- siloxane, polyoxyalkylene block copolymer, organosilicon cross-linking agent and/or catalyst.
- the polyoxyalkylene is reacted to the silicone binder by addition reaction
- US 2004/006190 discloses a room-temperature curable organopolysiloxane composition, which includes (A) an organopolysiloxane with the terminals blocked with a hydroxyl group, a hydrolysable group, or both these types of groups, and (B) an organosilicon compound containing a hydrolysable group, a partial hydrolysis-condensation product thereof, or a mixture of the two, and (C) a polysiloxane with at least one oxyalkylene group bonded to a silicon atom via, for example, a C-C-Si linkage.
- WO 2002/088043 discloses a process for coating a siliceous substrate with a silicon- containing layer, wherein in a first step a layer comprising a biocide is applied.
- US 2002/0197490 Al discloses a curable antifouling polysiloxane based composition
- a curable antifouling polysiloxane based composition comprising hydrophobic silica possibly in combination with hydrophilic silica.
- the composition further comprises a silicone oil, e.g. oils comprising poly(ethylene glycol) or poly(propylene glycol) moieties.
- antifouling agents may be used, in particular copper and inorganic copper compounds.
- EP 2 103 655 Al discloses a curable antifouling polysiloxane based composition
- a curable antifouling polysiloxane based composition comprising a reaction curable silicone rubber and a particularly designed organopolysiloxane mixture.
- the composition further comprises a silicone oil, e.g. a polyether- modified silicone oil.
- antifouling agents may be used, in particular copper and inorganic copper compounds.
- US 6,313,193 Bl i.a. discloses a composition comprising a silanol-terminated polydimethyl siloxane, a dimethylethoxy-terminated polydimethyl siloxane, polydiethoxy siloxane, and benzalkonium chloride.
- the polydiethoxy siloxane reacts with the polydimethyl siloxanes so that the polydiethoxy siloxane becomes an integral part of the binder network.
- JP 2006 052283 A discloses a coating composition comprising a polyether-modified silicone oil having polyoxyalkylene side chains, an acrylic type binder system based on a polysiloxane macropolymer and an antifoulant.
- JP 2006 299132 A discloses an antifouling coating composition which is based on a vinyl copolymer binder system which include polysiloxane side chains, and which is modified with certain reactive silanes, and which further comprises, e.g., an poly(oxyalkylene)-modified polysiloxane.
- the composition may also include an antifouling agent.
- JP 05-263022 discloses a hydrolysable coating composition typically based on an acid- anhydride vinyl binder for preventing aquatic organism attachment.
- EP 1 261 318 Al discloses a triphenylboron compound-containing antifouling coating composition stabilised with an amine compound.
- the composition comprises a non-reactive polysiloxane (oil) modified with oxyalkylene groups.
- US 2009/0018276 Al discloses a polysiloxane based coating composition having biocide moieties covalently bound to the binder matrix.
- the silicone based fouling release coatings have demonstrated an advantage over conventional antifouling coatings showing significant lower drag resistance, hence reduced fuel consumption of marine vessels. The difference is especially obvious as long as the silicone coating is free from marine fouling including slime fouling. Many conventional silicone coatings have until now only been able to maintain a slime free surface for a shorter period.
- a few biocide containing antifouling coatings have demonstrated a greater resistance towards marine fouling compared to the silicone based fouling release coatings under e.g. static conditions.
- the surface characteristics of such a coating will however lead to an increased drag resistance compared to silicone coatings even when the surface is fouling free.
- the rationale behind the present invention has been to prolong the slime free period of a silicone based coating by combining the biocidal components from the antifouling coatings with a silicone based fouling release coating. This provides a coating with low drag resistance that will remain fouling free for a longer time than conventional silicone based fouling release coatings.
- paint compositions for preparing new fouling control coatings comprising (i) a polysiloxane-based binder matrix having included as a part thereof hydrophilic oligomer/polymer moieties, and (ii) one or more biocides, which matrix facilitates and controls the leaching of the biocides.
- a polysiloxane-based binder matrix having included as a part thereof hydrophilic oligomer/polymer moieties
- biocides which matrix facilitates and controls the leaching of the biocides.
- the present inventors have realised that the use of a polysiloxane-based binder system having included as a part thereof hydrophilic oligomer/polymer moieties, in particular poly(oxyalkylene) moieties, (see further below) renders it possible to obtain a media for water- and biocidal transport through the cured polysiloxane matrix film.
- the leach rate of the biocide can be controlled amongst others by the amount and the hydrophilicity of the hydrophilic oligomer/polymer moieties.
- the present invention relates to a cured paint coat comprising a polysiloxane-based binder matrix and one or more biocides, said binder matrix having included as a part thereof hydrophilic oligomer/polymer moieties, wherein the weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is in the range 1 : 0.02 to 1 : 20, and wherein more than 50 % by weight of the binder matrix is represented by polysiloxane parts.
- a second aspect of the invention relates to a marine structure comprising on at least a part of the outer surface thereof a paint coat as defined herein.
- the present invention relates to a fouling control coating composition
- a fouling control coating composition comprising a polysiloxane-based binder system, said binder system comprising one or more polysiloxane components modified with hydrophilic oligomer/polymer moieties, and one or more biocides, wherein the weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is in the range 1 : 0.02 to 1 : 20, and wherein more than 50 % by weight of the binder system is represented by polysiloxane parts.
- the present invention relates to a fouling control coating composition
- a fouling control coating composition comprising a polysiloxane-based binder system, said binder system comprising one or more hydrophilic oligomer/polymer moieties which at one or both ends carry -Si(R*) 3 groups, wherein each R* independently is selected from Ci- 4 -alkyl and Ci- 4 -alkoxy, or R* is another condensation curable termination, at least one being Ci- 4 -alkoxy, or at least one being another condensation curable termination, and one or more biocides, wherein the weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is in the range 1 : 0.02 to 1 : 20, and wherein more than 50 % by weight of the binder system is represented by polysiloxane parts.
- a fifth aspect of the invention relates to a fouling control coating composition
- a fouling control coating composition comprising a polysiloxane-based binder system, said binder system comprising a cross-linker of formula
- each R 3 is independently selected from C 2 - 5 -alkylene (e.g. -CH 2 CH 2 -, -CH 2 CH(CH 3 )-, -CH 2 CH 2 CH 2 -, -CH 2 CH 2 CH 2 CH 2 -, -CH 2 CH(CH 2 CH 3 )-), arylene (e.g. 1,4-phenylene) and C 2 - 5 - alkylene substituted with aryl (e.g.
- 1-phenyl ethylene in particular from C 2 - 5 -alkylene such as -CH 2 CH 2 - and -CH 2 CH(CH 3 )-); p is 0-50; a is 0-2; z is 1-3; and one or more biocides, wherein the weight ratio between the poly(oxyalkylene) and the one or more biocides is in the range 1 : 0.02 to 1 : 20, and wherein more than 50 % by weight of the binder system is represented by polysiloxane parts.
- a sixth aspect of the invention relates to the use of the combination of one or more polysiloxane components modified with hydrophilic oligomer/polymer moieties and one or more biocides, wherein the weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is in the range 1 : 0.02 to 1 : 20, for improving the antifouling properties of a polysiloxane based coating composition.
- the present invention provides a cured paint coat comprising a polysiloxane-based binder matrix and one or more biocides, said binder matrix having included as a part thereof hydrophilic oligomer/polymer moieties, wherein the weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is in the range 1 : 0.02 to 1 : 20, such as 1 : 0.05 to 1 : 10, and wherein more than 50 % by weight of the binder system is represented by polysiloxane parts.
- the paint coat is preferably used as the top coat in a coating system comprising two or more paint coat layers (e.g. at least a primer coat and a tie coat in addition to the top coat).
- the function of the polysiloxane-based binder matrix rendered hydrophilic by means of oligomer/polymer moieties is to facilitate the transport and accessibility of the biocide at the surface upon contact with sea-water. Potentially, a hydrated layer formed at the coating-water interphase will also aid in retaining the biocide at the surface, hence allowing the coating to exert its fouling deterrent activity for extended exposure intervals.
- hydrophilic oligomer/polymer moieties forms a part of the polysiloxane-based binder matrix, i.e. that the moieties are covalently incorporated into the binder matrix. It should also be understood that the formed covalent bond(s) preferably are non-hydrolysable.
- the polysiloxane-based binder matrix typically constitutes at least 40 % by dry weight, such as 40-98 % by dry weight, or 45-95 % by dry weight, or 50-95 % by dry weight, in particular 50-90 % by dry weight, or 55-90 % by dry weight, or 60-90 % by dry weight, or 60-80 % by dry weight, of the cured paint coat.
- polysiloxane-based binder matrix is intended to mean that the binder matrix mainly consists of polysiloxane parts, i.e. that more than 50 % by weight, preferably more than 60 % by weight, e.g. more than 70 % by weight, of the binder matrix is represented by polysiloxane parts.
- the polysiloxane parts constitute 50-99.99 % by weight, e.g.
- the binder matrix i.e. the binder components and any cross-linkers.
- the remainder of the binder matrix is preferably made of the hydrophilic oligomer/polymer moieties and any (non-polysiloxane-type) cross-linkers.
- the hydrophilic oligomer/polymer moieties preferably makes up 1-30 % by weight, such as 2-20 % by weight, e.g. 1-10 % by weight, of the binder matrix.
- hydrophilic oligomer/polymer moieties which are included in the polysiloxane-based binder matrix are of non-silicon origin.
- oligomer/polymer moieties include all atoms up to, but not including, the silicon atom which is adjacent to the hydrophilic oligomer/polymer moiety.
- the [polysiloxane-0]-Si(Me)2-CH 2 CH 2 CH2-[hydrophilic oligomer]-CH 2 CH 2 CH2-Si(Me)2-[0- polysiloxane] the [polysiloxane-0]-Si(Me) 2 parts are accounted for as silicone parts, whereas the CH2CH 2 CH2-[hydrophilic oligomer]-CH2CH 2 CH2 moiety is accounted for as the hydrophilic oligomer moiety.
- Suitable hydrophilic oligomer/polymer moieties are those selected from poly(vinyl pyrrolidone), poly[N-(2-hydroxypropyl)methacrylamide], poly(N,N- dimethacrylamide), poly(acrylic acid), poly(glycerol), polyHEMA, polysaccharides, poly(vinyl alcohol),
- polyketones poly(aldehyde guluronate), polyvinylamine, polycaprolactones, poly(vinyl acetate), polyoxyalkylenes like poly(ethylene glycol), poly(propylene glycol), poly(2-methyl- 2-oxazoline), etc., including copolymers of the foregoing.
- hydrophilicity is obtained by modification with polyoxyalkylene moieties like poly(ethylene glycol).
- linking group is understood as the product of the reaction of two mutually reaction functional groups; one functional group on the polysiloxane backbone and one on the hydrophilic oligomer/polymer.
- an amine linking group is result of for example - but not exclusively - the reaction of a glycidyl ether with a primary or secondary amine.
- useful linking groups between the hydrophilic oligomer/polymer and the polysiloxane backbone are: amine groups, ether groups, amide groups.
- linking group is the Si-C bond prepared by hydrosilylation reaction catalyzed by platinum where the functional group on the polysiloxane backbone is a hydride and the functional group on the hydrophilic oligomer/polymer is an allyl group.
- the hydrophilic oligomer/polymer moieties provide a permanent hydrophilic contribution to the binder matrix.
- the hydrophilic oligomer/polymer moieties are preferably devoid of any bonds which are hydrolysable in sea-water.
- the hydrophilic oligomer/polymer moieties do not include ester bonds or acid anhydride bonds.
- it is also preferred that the hydrophilic oligomer/polymer moieties provide a permanent hydrophilic contribution to the binder matrix.
- the link between the hydrophilic oligomer/polymer moieties and the silicone is devoid of any bonds which are hydrolysable in sea-water.
- the hydrophilic oligomer/polymer moieties are preferably devoid of any bonds which are hydrolysable in sea-water.
- oligomer/polymer moieties are not attached to the polysiloxane through hydrolysable bonds such as for example ester bonds or acid anhydride bonds.
- hydrophilic moiety may in itself include hydrolysable bonds, such as for example an acetate capped
- oligomer/polymer moieties are understood as those encompassing at least 3 repeating units, such as at least 5 repeating units.
- oligomer/polymer moieties used for modification include 3-1,000 repeating units, such as 3-200, or 5-150, or 5-100, or 10-80, or 5-20, repeating units.
- the hydrophilic oligomer/polymer moieties i.e. oligomeric or polymeric groups incorporated into the binder matrix
- M n number average molecular weight in the range of 100-50,000 g/mol, such as in the range of 100-30,000 g/mol, in particular in the range of 150-20,000 g/mol, or in the range of 200-10,000 g/mol.
- hydrophilic oligomer/polymer moieties hydrophilic polymer moieties
- hydrophilic polymer moieties hydrophilic polymer moieties
- similar are intended to mean that the oligomeric or polymeric moieties, in themselves (i.e. when represented as discrete molecules) have a solubility of at least 1 %(w/w) in demineralized water at 25°C.
- the cured paint coat of the invention is typically prepared from a binder system which - upon curing - forms a cross-linked polysiloxane-based binder matrix which incorporates the biocide(s) as well as other constituents such as additives, pigments, fillers, etc. in the cured paint coat.
- the polysiloxane-based binder matrix is made up of reactive polysiloxane binder components, e.g. functional organopolysiloxanes, cross-linkers, silicates (e.g. ethyl silicate), and the like.
- reactive polysiloxane binder components e.g. functional organopolysiloxanes, cross-linkers, silicates (e.g. ethyl silicate), and the like.
- silicates e.g. ethyl silicate
- the cured paint coat may be formed in various ways, e.g. polymerization/cross-linking by formation of siloxane bonds through a condensation reaction or by the use of their reactive groups such as for example amine/epoxy, carbinol/isocyanate etc. Condensation reaction is preferred.
- the polysiloxane based binder is a functional organopolysiloxane, with terminal and/or pendant functionality. The terminal functionality is preferred.
- the functionality can either be hydrolysable groups, such as for example alkoxy groups, ketoxime groups or the functionality can be silanol groups. A minimum of two reactive groups per molecule is preferred.
- the cross-linker can for example be an alkoxy silane such as methyltrimethoxysilane, but a wide range of useful silanes are available as will be described further on.
- the silane can be used as it is or as hydrolysation-condensation products of same.
- condensation cure is much preferred, the functionality of the organopolysiloxane is not limited to a condensation cure. If so desired, other types of curing can be utilized, for example amine/epoxy either alone or in combination with a condensation reaction. In such cases, the organopolysiloxane can have terminal groups of epoxy or amine and pendant hydrolysable groups, for example with alkoxyfunctionality.
- the polysiloxane-based binder system discussed above is characterized by having included therein, as a part of the binder matrix, one or more polysiloxane components which are modified with hydrophilic oligomer/polymer moieties.
- polysiloxane components will upon reaction with other polysiloxane components and cross-linkers provide hydrophilic properties to the binder system.
- hydrophilic oligomer/polymer moieties functionalized with reactive silanes which enable them to react with the polysiloxane binder, or the hydrophilic oligomer/polymer moieties, and form a non-hydrolysable bond may also be used.
- the polysiloxane components must include silicon-reactive groups such as Si-OH groups, hydrolysable groups such as Si-OR (such as alkoxy, oxime, acetoxy etc.) groups, etc., so as to facilitate reaction with other constituents of the polysiloxane-based binder system.
- silicon-reactive groups such as Si-OH groups
- hydrolysable groups such as Si-OR (such as alkoxy, oxime, acetoxy etc.) groups, etc.
- the hydrophilic oligomer/polymer moiety is a poly(oxyalkylene) moiety.
- the fouling control coating composition including the polysiloxane- based binder system may be a reaction-curable composition or a condensation-curable composition as will be evident for the person skilled in the art.
- Examples hereof are a two- component condensation curing composition based on a silanol-reactive polydiorganosiloxane and a silane with hydrolysable groups, or a one-component condensation-curable
- reaction curable composition based on a polydiorganosiloxane with alkoxy or other hydrolysable reactivity.
- a reaction curable composition based on an epoxy-functional polysiloxane binder and an amine functional polysiloxane curing agent.
- condensation curable compositions are possible, if the binder or the curing agent (or both) includes condensation curable groups, such as alkoxy groups.
- the binder phase comprises (i) a binder and (ii) a cross-linking agent of which the binder (i) should include hydrolysable groups or other reactive groups so as to participate in the formation of the matrix.
- the binder (i) typically constitutes 20-90 % by dry weight of the coating composition, and it should be understood, that the binder (i) may be of different types, e.g. two or more of the types described above, so that the binder (i) may, for example, comprise a non-modified curable polysiloxane and a curable polysiloxane which has been modified with hydrophilic moieties.
- the binder includes a curable diorganopolysiloxane represented by a general formula (1) shown below:
- the binder includes a curable diorganopolysiloxane represented by a general formula (lx) shown below:
- each of A 1 , A 2 , A 3 , A 4 , a and b are as above for formula (1) and wherein each A 5 is independently selected from oxygen or an alkyl group of 2-5 carbon atoms.
- the synthesis is carried out at elevated temperatures, such as 60- 150°C.
- the resulting binder from the reaction (lb) is further modified with a hydrophilic component, for example poly(ethylene glycol) mono allyl ether as outlined in formula ( lc) resulting in a curable, polysiloxane modified with hydrophilic oligomer/polymer moieties.
- a hydrophilic component for example poly(ethylene glycol) mono allyl ether as outlined in formula ( lc) resulting in a curable, polysiloxane modified with hydrophilic oligomer/polymer moieties.
- the binder obtained can be used as is, or in combination with a curable diorganosiloxane (of the generic type presented in formula (1)).
- a curable diorganosiloxane of the generic type presented in formula (1).
- hydrophilic polymers other than poly(ethylene glycol) are also useful for rendering the polysiloxane hydrophilic.
- the cured paint coat comprising a polysiloxane-based binder matrix has included as a part thereof pendant hydrophilic oligomer/polymer moieties.
- pendant means that the hydrophilic oligomer/polymer moieties are attached to the polysiloxane backbone at a non-terminal position and that such moieties are attached at only the one end so that the pendant hydrophilic oligomer/polymer forms a "graft" to the polysiloxane backbone (matrix).
- Pendant hydrophilic oligormers/polymer moieties may in principle carry functional (non- reactive) groups at the free end, e.g. groups exhibiting a biocidal effect, etc.
- the hydrophilic oligomers/polymer moieties are not carrying such functional groups, but are in the form of the native oligomer/polymer, possibly end-capped, like with an alkyl group, or possibly with a hydroxyl group or methoxy terminated.
- binder is an A-B-A copolymer of polysiloxane (A) and a hydrophilic polymer (B), such as poly(oxyalkylene).
- A polysiloxane
- B hydrophilic polymer
- An example of the structure of the polymer is depicted in formula (Id).
- units of a polymer with hydrophilic character, such as polyoxyalkylene are introduced in the backbone of the polysiloxane to form an alternating block copolymer.
- Introducing hydrophilic groups such as oxyalkylene groups in the binder may increase the hydrophilicity of the binder as described in reference WO 2008/132196.
- the binders can be used alone or in combination, and the structure of the copolymer can be A-B- A and B-A-B. In case of B-A-B, a pendant curable functionality is required, since the terminal groups of the silicone portion would be blocked by the hydrophilic polymer.
- Ci- 4 -alkoxy such as for example trimethoxysilane, triethoxysilane or methyldimethoxysilane, in the presence of a hydrosilylation catalyst, such as platinum, yielding a curable poly(oxyalkylene).
- a hydrosilylation catalyst such as platinum
- the reaction is carried out at elevated temperatures, such as 60-150°C.
- the synthesis is outlined in formula (le).
- the polymer has to be used in combination with, e.g., component (i) (formula (1).
- useful silanes include, without being limited to, triethoxysilane, tripropoxysilane, tert-butyldiethoxysilane.
- the cross-linking agent (ii) preferably constitutes 0-10 % by dry weight of the coating composition and is, e.g., an organosilicon compound represented by the general formula (2) shown below, a partial hydrolysis-condensation product thereof, or a mixture of the two:
- each R represents, independently, an unsubstituted or substituted monovalent hydrocarbon group of 1 to 6 carbon atoms or a hydrolysable group
- each X represents, independently, a hydrolysable group
- a represents an integer from 0 to 2, such as from 0 to 1.
- the compound outlined in formula (2) acts as a cross-linker for the binder (i).
- the composition can be formulated as a one component curable RTV (room-temperature vulcanizable) by admixing the binder (i) and the cross-linking agent (ii). If the reactivity on the terminal Si-group of the binder (i) consist of readily hydrolysable groups, such as dimethoxy or trimethoxy, a separate cross-linker is usually not necessary to cure the film.
- RTV room-temperature vulcanizable
- R represents a hydrophilic group such as a poly(oxyalkylene).
- the organopolysiloxane may have oxyalkylene domains.
- the hydrophilicity may be obtained (or added to the hydrophilicity which may have been obtained by incorporating a hydrophilic group to component (i) as outlined in previous section), by using a hydrophilic silane, such as the generic type expressed in formula (2a).
- the hydrophilic silane will react with the silanol or the hydrolysable groups in the binder component (formula (1) or (le)), and thereby incorporate a hydrophilic component.
- each R 3 is independently selected from C 2 - 5 -alkylene (e.g. -CH 2 CH 2 -, -CH 2 CH(CH 3 )-, -CH2CH2CH2-, -CH2CH2CH2CH2-, -CH 2 CH(CH 2 CH 3 )-), arylene (e.g. 1,4-phenylene) and C 2 - 5 - alkylene substituted with aryl (e.g. 1-phenyl ethylene), in particular from C 2 - 5 -alkylene such as -CH2CH2- and -CH 2 CH(CH 3 )-); p is 0-50; a is 0-2; z is 1-3.
- organopolysiloxane will increase the hydrophilicity of the binder, especially when ethyleneoxide type -[CH 2 CH 2 -0]- is used.
- the hydrophilic modification of the binder consists of both A-B-A modifications (as described above) and of pendant hydrophilic oligomer/polymer moieties.
- Preferred cross-linkers are those selected from tetramethoxysilane, tetraethoxysilane;
- tetrapropoxysilane tetra-n-butoxysilane; vinyltris(methylethyloximino)silane; vinyltris- (acetoxime)silane; methyltris(methylethyloximino)silane; methyltris(acetoxime)silane; vinyltrimethoxysilane; methyltrimethoxysilane; vinyltris(isopropenoxy)silane;
- tetraacetoxysilane methyltriacetoxysilane; ethyltriacetoxysilane; vinyltriacetoxysilane; di-t- butoxydiacetoxysilane; methyltris(ethyllactate)silane and vinyltris(ethyllactate)silane as well as hydrolysis-condensation products of the same.
- cross-linkers are those selected from vinyltriethoxysilane, methyltriethoxysilane, ethyltrimethoxysilane, ethyltrimethoxysilane, tetraisopropoxysilane, tetrabutoxysilane as well as hydrolysis-condensation products of the same.
- the polysiloxane-based binder comprises a
- the binder may include fluoro-modifications, e.g.
- fluoroalkyl modified polysiloxane binders such as silanol-terminated poly(trifluoropropyl- methylsiloxane).
- the polysiloxane-based binder system typically constitutes at least 40 % by dry weight, such as 40-98 % by dry weight, or 45-95 % by dry weight, or 50-95 % by dry weight, in particular 50-90 % by dry weight, or 55-90 % by dry weight, or 60-90 % by dry weight, of the coating composition.
- the cured coating may be prepared from a range of different combinations of reactive polysiloxanes and silanes.
- the invention provides a fouling control coating composition
- a fouling control coating composition comprising a polysiloxane-based binder system, said binder system comprising one or more polysiloxane components modified with hydrophilic oligomer/polymer moieties, and one or more biocides, wherein the weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is in the range 1:0.02 to 1:20, in particular 1:0.05 to 1:10, and wherein more than 50 % by weight of the binder system is represented by polysiloxane parts.
- the invention provides a fouling control coating composition
- a fouling control coating composition comprising a polysiloxane-based binder system, said binder system comprising one or more hydrophilic oligomer/polymer moieties which at one or both ends carry -Si(R*) 3 groups, wherein each R* independently is selected from C ⁇ -alkyl and C ⁇ -alkoxy (e.g.
- R* is another condensation curable termination, at least one being Ci-4-alkoxy, or at least one being another curable termination, and one or more biocides, wherein the weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is in the range 1 : 0.02 to 1 : 20, and wherein more than 50 % by weight of the binder system is represented by polysiloxane parts.
- the one or more hydrophilic oligomer/polymer moieties carry at only one end -Si(R*) 3 groups. In this way, it is possible to construct a binder of a polysiloxane chain having grafts of the hydrophilic oligomer/polymer moieties.
- the one or more hydrophilic oligomer/polymer moieties carry at both ends -Si(R*) 3 groups.
- the hydrophilic component will be incorporated in the crosslinked network and constitute a non-hydrolysable hydrophilic moiety.
- the invention provides a fouling control coating composition
- a fouling control coating composition comprising a polysiloxane-based binder system, said binder system comprising a cross-linker of formula (2) above, wherein R represents a poly(oxyalkylene), and one or more biocides, wherein the weight ratio between the poly(oxyalkylene)) and the one or more biocides is in the range 1 : 0.02 to 1 : 20.
- the invention provides a fouling control coating composition
- a fouling control coating composition comprising a polysiloxane-based binder system, said binder system comprising a cross-linker of formula (2a)
- each R 3 is independently selected from C 2 - 5 -alkylene (e.g. -CH 2 CH 2 -, -CH 2 CH(CH 3 )-, -CH2CH2CH2-, -CH2CH2CH2CH2-, -CH 2 CH(CH 2 CH 3 )-), arylene (e.g. 1,4-phenylene) and C 2 - 5 - alkylene substituted with aryl (e.g.
- 1-phenyl ethylene in particular from C 2 - 5 -alkylene such as -CH2CH2- and -CH 2 CH(CH 3 )-); p is 0-50; a is 0-2; z is 1-3; and one or more biocides, wherein the weight ratio between the poly(oxyalkylene) and the one or more biocides is in the range 1 : 0.02 to 1 : 20, and wherein more than 50 % by weight of the binder system is represented by polysiloxane parts.
- the hydrophilic oligomer/polymer moieties are selected from polyvinyl pyrrolidone), poly[N-(2-hydroxypropyl)methacrylamide], poly(acrylic acid), poly(glycerol), polyHEMA, polysaccharides, polyvinyl alcohol), polyketones, poly(aldehyde guluronate), polyvinylamine, polycaprolactones, polyvinyl acetate), polyoxyalkylenes like poly(ethylene glycol), poly(propylene glycol), poly(2-methyl-2- oxazoline), including copolymers of the foregoing, in particular poly(oxyalkylenes), especially from polyoxyethylene, polyoxypropylene and poly(oxyethylene-co-oxypropylene).
- polysiloxane-based binder system is intended to mean that the binder system mainly consists of polysiloxane parts, i.e. that more than 50 % by weight, preferably more than 60 % by weight, e.g. more than 70 % by weight, of the binder system is represented by polysiloxane parts.
- the polysiloxane parts constitute 50-99.99 % by weight, e.g.
- the binder system i.e. the binder components and any cross-linkers.
- the remainder of the binder system is preferably made of the hydrophilic oligomer/polymer moieties and any (non-polysiloxane-type) cross-linkers.
- the hydrophilic oligomer/polymer moieties preferably makes up 1-30 % by weight, such as 2-20 % by weight, e.g. 1-10 % by weight, of the binder system.
- the coating composition used for forming a cured fouling control coat also includes one or more biocides.
- biocide is intended to mean an active substance intended to destroy, deter, render harmless, prevent the action of, or otherwise exert a controlling effect on any harmful organism by chemical or biological means.
- biocides are those selected from metallo-dithiocarbamates such as bis(dimethyldithiocarbamato)zinc, ethylene-bis(dithiocarbamato)zinc, ethylene-bis(dithio- carbamato)manganese, and complexes between these; bis(l-hydroxy-2(lH)-pyridine- thionato-0,S)-copper; copper acrylate; bis(l-hydroxy-2(lH)-pyridinethionato-0,S)-zinc; phenyl(bispyridyl)-bismuth dichloride; metal biocides such as copper(I)oxide, cuprous oxide, metallic copper, copper metal alloys such as copper-nickel alloys; metal salts such as cup
- imidazole containing compound such as medetomidine; 2-(p-chlorophenyl)-3-cyano-4- bromo-5-trifluoromethyl pyrrole and mixtures thereof.
- the biocide does not comprise tin.
- biocides are those selected from the group consisting of 2,4,5,6-tetra- chloroisophtalonitrile (Chlorothalonil), copper thiocyanate (cuprous sulfocyanate), N-dichloro- fluoromethylthio-N',N'-dimethyl-N-phenylsulfamide (Dichlofluanid), 3-(3,4-dichlorophenyl)- 1, 1-dimethylurea (Diuron), N 2 -tert-butyl-N 4 -cyclopropyl-6-methylthio-l,3,5-triazine-2,4- diamine (Cybutryne), 4-bromo-2-(4-chlorophenyl)-5-(trifluoromethyl)-lH-pyrrole-3- carbonitrile, (2-(p-chlorophenyl)-3-cyano-4-bromo-5-trifluoromethyl pyrrole; Tralopyril), ⁇ /
- the biocides are preferably selected among biocides which are effective against soft fouling such as slime and algae.
- biocides are N 2 -tert-butyl-N 4 -cyclopropyl-6-methylthio- l,3,5-triazine-2,4-diamine (Cybutryne), 4,5- dichloro-2-n-octyl-4-isothiazolin-3-one (DCOIT, Sea-Nine ® 211N), bis(l-hydroxy-2(lH)- pyridinethionato-0,S)-(T-4) zinc (zinc pyridinethione; zinc pyrithione), bis(l-hydroxy-2(lH)- pyridinethionato-0,S)-T-4) copper (copper pyridinethione; copper pyrithione) and zinc ethylene- l,2-bis-dithiocarbamate (zinc-ethylene-N-N'-dithiocarbamate
- the biocide is an organic biocide, such as a pyrithione complex, such as zinc pyrithione, or such as copper pyrithione.
- organic biocides are those either fully or in part being of organic origin.
- biocide in those instances in which the biocide is depleted rapidly from the film due to e.g. a high water solubility or a high level of immiscibility with the matrix composition, it can be advantageous to add one or more of the biocide(s) in encapsulated form as a means of controlling the biocide dosage and extending the effective lifetime in the film.
- Encapsulated biocides can also be added if the free biocide alters the properties of the polysiloxane matrix in a way that is detrimental for its use as antifouling coatings (e.g.
- the biocide is encapsulated 4,5-dichloro-2-n-octyl-4- isothiazolin-3-one (Sea-Nine CR2).
- the biocide preferably has a solubility in the range of 0-20 mg/L, such as 0.00001-20 mg/L, in water at 25 °C.
- the biocide typically constitutes 0.1-15 % by dry weight, e.g.0.5-8 % by dry weight, in particular 1-6 % by dry weight, of the coating composition.
- the relative weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is typically in the range of 1:0.02 to 1:20, or 1:0.05 to 1:20, or 1:0.06 to 1:16, or 1:0.08 to 1:12, or 1:0.05 to 1:10, or 1:0.1 to 1:10, even 1:0.15 to 1:6, or 1:0.05 to 1:5, or 1:0.1 to 1:5, or 1:0.2 to 1:4.
- the relative weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is typically in the range of 1:0.02 to 1:20, or 1:0.05 to 1:20, or 1:0.06 to 1:16, or 1:0.08 to 1:14, or 1:0.1 to 1:12, or 1:0.05 to 1:10, even 1:0.15 to 1:10, or 1:0.05 to 1:9, or 1:0.1 to 1:8, or 1:0.2 to 1:7.
- the coating composition used for forming a cured fouling control coat may further comprise a condensation catalyst to accelerate the cross-linking.
- suitable catalysts include organometal- and metal salts of organic carboxylic acids, such as dibutyl tin dilaurate, dibutyl tin diacetate, dibutyl tin dioctoate, dibutyl tin 2-ethylhexanoate, dioctyl tin dilaurate, dioctyl tin diacetate, dioctyl tin dioctoate, dioctyl tin 2-ethylhexanoate, dioctyltin di neodecanoate, tin naphthenate, tin butyrate, tin oleate, tin caprylate, bismuth 2-ethylhexanoate, bismuth octanoate, bismuth neodecan
- tetra(isopropenyloxy)-titanate titanium tetrabutanolate, titanium tatrapropanolate; titanium tetraisopropanolate, zirconium tetrapropanolate, zirconium tetrabutanolate; chelated titanates such as diisopropyl bis(acetylacetonyl)titanate.
- Further catalysts include tertiary amines, such as triethylamine, tetrametylethylenediamine, pentamethyldiethylenetriamine and 1,4-ethylenepiperazine. Further examples include guanidine based catalysts. Even further examples of condensation catalysts are described in WO 2008/132196 and US 2004/006190.
- the catalyst may be used alone or as combination of two or more catalysts.
- the amount of catalyst to be used is depending on the reactivity of the catalyst and the cross-linker(s) and desired drying time.
- the catalyst concentration is between 0.01- 10 %, e.g.0.01-3.0 %, or 5.0-10 %, or 0.1-4.0 %, or 1.0-6.0 %, by weight of the total combined amount of the binder (i) and cross-linking agent (ii).
- a catalyst is not included. Hydrophilic-modified polysiloxane oils
- the composition may further include hydrophilic-modified polysiloxane oils, i.e. constituents which do not form covalent bonds to the polysiloxane-based binder matrix.
- Hydrophilic- modified polysiloxane oils are widely used as surfactants and emulsifiers due to the content of both hydrophilic and lipophilic groups in the same molecule.
- the hydrophilic-modified polysiloxane oils are selected so that they do not contain groups that can react with the binder (or binder components) or the cross-linker (if present), hence the hydrophilic-modified polysiloxane oils are intended to be non-reactive, in particular with respect to the binder components.
- hydrophilic-modified polysiloxane oils are devoid of any silicon-reactive groups such as Si-OH groups, hydrolysable groups such as Si-OR (such as alkoxy, oxime, acetoxy etc.) groups, etc., so as to avoid reaction with constituents of the polysiloxane-based binder system.
- non-reactive hydrophilic-modified polysiloxane oils are typically modified by the addition of non-ionic oligomeric or polymeric groups which can be polar and/or capable of hydrogen bonding, enhancing their interaction with polar solvents, in particular with water, or with other polar oligomeric or polymeric groups.
- these groups include, amides (e.g. poly(vinyl pyrrolidone), poly[N-(2-hydroxypropyl)methacrylamide]), poly(N,N- dimethacrylamide), acids (e.g. poly(acrylic acid)), alcohols (e.g.
- the hydrophilicity is obtained by modification with polyoxyalkylene groups.
- the hydrophilic oligomer/polymer moieties with which the polysiloxane oils are modified are of non-silicon origin.
- the above-mentioned "oligomers” and “polymers” include at least 3 repeating units, such as at least 5 repeating units.
- the oligomers or polymers include 3-1,000 repeating units, such as 3-200, or 5-150, or 5-100 repeating units.
- the hydrophilic groups i.e.
- oligomeric or polymeric groups have a number average molecular weight (M n ) in the range of 100-50,000 g/mol, such as in the range of 100-30,000 g/mol, in particular in the range of 200-20,000 g/mol, or in the range of 200-10,000 g/mol.
- M n number average molecular weight
- hydrophilic-modified in the context of "hydrophilic-modified polysiloxane oil” is intended to mean that the oligomeric or polymeric groups with which the polysiloxane is modified, in themselves (i.e. as discrete molecules) have a solubility of at least 1 %(w/w) in demineralized water at 25°C.
- hydrophilic-modified polysiloxane oils in which the relative weight of the hydrophilic moieties is 1% or more of the total weight (e.g. 1-90%), such as 5% or more (e.g. 5-80%), in particular 10% or more (e.g. 10-70%) of the total weight of the hydrophilic-modified polysiloxane oil.
- the hydrophilic-modified polysiloxane oil (if present) has a number average molecular weight (M n ) in the range of 100-100,000 g/mol, such as in the range of 250-75,000 g/mol, in particular in the range of 500-50,000 g/mol.
- M n number average molecular weight
- hydrophilic-modified polysiloxane oils (if present) have a viscosity in the range of 10-20,000 mPa ⁇ s, such as in the range of 20-10,000 mPa ⁇ s, in particular in the range of 40-5,000 mPa ⁇ s.
- the hydrophilic-modified polysiloxane oils may be utilized to control the leaching of the biocide.
- the hydrophilic-modified polysiloxane oil is a poly(oxyalkylene)-modified polysiloxane.
- the poly(oxyalkylene)-modified polysiloxane oil is a polysiloxane having grafted thereto poly(oxyalkylene) chains.
- An illustrative example of the structure of such hydrophilic-modified polysiloxane oils is formula (A) :
- each R 1 is independently selected from Ci_ 5 -alkyl (including linear or branched hydrocarbon groups) and aryl (e.g . phenyl (-C 6 H 5 )), in particular methyl; each R is independently selected from -H, Ci- 4 -alkyl (e.g . -CH 3 , -CH 2 CH 3 ,
- each R 3 is independently selected from C 2 _ 5 -alkylene (e.g . -CH 2 CH 2 -, -CH 2 CH(CH 3 )-, -CH 2 CH 2 CH 2 -, -CH 2 CH 2 CH 2 CH 2 -, -CH 2 CH(CH 2 CH 3 )-), arylene (e.g. 1,4-phenylene) and C 2 - 5 -alkylene substituted with aryl (e.g . 1-phenyl ethylene), in particular from C 2 - 5 -alkylene such as
- x is 0-2500, y is 1-100 and x+y is 1-2000; and n is 0-50, m is 0-50 and m+ n is 1-50.
- hydrophilic-modified polysiloxane oils of this type are DC5103 (Dow Corning), DC Q2-5097 (Dow Corning), and DC193 (Dow Corning) .
- poly(oxyalkylene)-modified polysiloxane oil is a polysiloxane having incorporated in the backbone thereof poly(oxyalkylene) chains.
- An illustrative example of the structure of such hydrophilic-modified polysiloxane oils is formula (B) : R R R R
- each R 1 is independently selected from Ci_ 5 -alkyl (including linear or branched hydrocarbon groups) and aryl (e.g . phenyl (-C 6 H 5 )), in particular methyl; each R 2 is independently selected from -H, C ⁇ -alkyl (e.g. -CH 3 , -CH 2 CH 3 , -CH 2 CH 2 CH 3 ,
- each R 3 is independently selected from C 2 - 5 -alkylene (e.g. -CH 2 CH 2 -, -CH 2 CH(CH 3 )-, -CH 2 CH 2 CH 2 -,
- arylene e.g. 1,4-phenylene
- C 2-5 - alkylene substituted with aryl e.g . 1-phenyl ethylene
- x is 0-2500
- n is 0-50
- m is 0-50 and m+ n is 1-50.
- hydrophilic-modified polysiloxane oils of this type are DC Q4-3669 (Dow Corning), DC Q4-3667 (Dow Corning) and DC2-8692.
- the poly(oxyalkylene)-modified polysiloxane oil is a polysiloxane having incorporated in the backbone thereof polyoxyalkylene chains and having grafted thereto polyoxyalkylene chains.
- An illustrative example of the structure of such hydrophilic-modified polysiloxane oils is formula (C) :
- each R 1 is independently selected from Ci_ 5 -alkyl (including linear or branched hydrocarbon groups) and aryl (e.g. phenyl (-C 6 H 5 )), in particular methyl; each R 2 is independently selected from -H, d_ 4 -alkyl (e.g. -CH 3 , -CH 2 CH 3 , -CH 2 CH 2 CH 3 ,
- each R 3 is independently selected from C 2 - 5 -alkylene (e.g. -CH 2 CH 2 -, -CH 2 CH(CH 3 )-, -CH 2 CH 2 CH 2 -, -CH 2 CH 2 CH 2 CH 2 -, -CH 2 CH(CH 2 CH 3 )-), arylene (e.g.
- x is 0-2500, y is 1-100 and x+y is 1-2000;
- k is 0-50, I is 0-50 and k+l is 1-50; and
- n is 0-50, m is 0-50 and m+n is 1-50.
- the groups -CH 2 CH(CH 3 )-, -CH 2 CH(CH 2 CH 3 )-, etc. may be present in any of the two possible orientations.
- the segments present x and y times typically are randomly distributed, or distributed as blocks, within the polysiloxane structure.
- the poly(oxyalkylene) is preferably selected from polyoxyethylene, polyoxypropylene and poly(oxyethylene-co-oxypropylene), which sometimes are referred to as poly(ethylene glycol), poly(propylene glycol) and poly(ethylene glycol-co-propylene glycol).
- each R 3 linking two oxygen atoms is preferably selected from -CH 2 CH 2 - and -CH 2 CH(CH 3 )-, whereas each R 3 linking a silicon atom and an oxygen atom preferably is selected from C 2 - 5 -alkyl.
- non-reactive hydrophilic-modified polysiloxane oils if present may be of different types, e.g. two or more of the types described above.
- the one or more hydrophilic-modified polysiloxane oils are included in the coating
- the one or more hydrophilic-modified polysiloxane oils constitutes 0.05-7 % by dry weight, e.g. 0.1-5 % by dry weight, in particular 0.5-3 % by dry weight, of the coating composition. In certain other embodiments, the one or more hydrophilic-modified
- polysiloxane oils constitutes 1-10 % by dry weight, e.g. 2-9 % by dry weight, in particular 2- 7 % by dry weight, or 3-7 % by dry weight, or 3-5 % by dry weight, or 4-8 % by dry weight, of the coating composition.
- the ratio between on the one hand the combined amount of the hydrophilic oligomer/polymer moieties of the binder matrix and the one or more hydrophilic-modified polysiloxane oil(s) and on the other hand the one or more biocides is typically in the range of 1:0.02 to 1:20, or 1:0.05 to 1:20, or 1:0.06 to 1:16, or 1:0.08 to 1:12, or 1:0.1 to 10, even 1:0.15 to 1:6, or 1:0.1 to 1:5, or 1:0.2 to 1:4.
- the ratio between on the one hand the combined amount of the hydrophilic oligomer/polymer moieties of the binder matrix and the one or more hydrophilic-modified polysiloxane oil(s) and on the other hand the one or more biocides is typically in the range of 1:0.02 to 1:20, or 1:0.05 to 1:20, or 1:0.06 to 1:16, or 1:0.08 to 1:14, or 1:0.1 to 12, even 1:0.15 to 1:10, or 1:0.05 to 1:9, or 1:0.1 to 1:8, or 1:0.2 to 1:7.
- the paint coat (and the coating composition) is completely devoid of any hydrophilic-modified polysiloxane oils.
- the paint composition (and the coating composition) is also devoid of any of the non-reactive fluids mentioned under "additives” (i) and any fluorinated oils (cf.
- compositions may be referred to as "oil free”.
- the coating composition may have included therein one or more fluorinated oil(s).
- fluorinated oil is inherently meant that the constituent does not form covalent bonds to the polysiloxane-based binder matrix.
- the fluorinated oil(s) do not contain groups that can react with the binder (or binder components) or the cross-linker (if present), hence the one or more fluorinated oil(s) are intended to be non-reactive, in particular with respect to the binder components.
- the fluorinated oils are devoid of any silicon-reactive groups such as Si-OH groups, hydrolysable groups such as Si-OR (such as alkoxy, oxime, acetoxy etc.) groups, etc., so as to avoid reaction with constituents of the polysiloxane-based binder system.
- the one or more fluorinated oil(s) (if present) have a viscosity in the range of 10-20,000 mPa ⁇ s, such as in the range of 20-10,000 mPa ⁇ s, in particular in the range of 40-5,000 mPa ⁇ s.
- the fluorinated oil(s) (if present) has a number average molecular weight (M n ) in the range of 100-100,000 g/mol, such as in the range of 250- 75,000 g/mol, in particular in the range of 500-50,000 g/mol.
- M n number average molecular weight
- the one or more fluorinated oils may be utilized to control the accessibility of the one or more enzymes and/or to control the leaching of any biocides, as well as to distribute the enzyme in the wet paint.
- the one or more fluorinated oils are selected from fluoroalkyi modified oils, e.g. perfluorinated oils, perfluoroalkyl-modified polysiloxane, perfluoro siloxane, perfluoro polyether, perfluorinated alkanes, or perfluoroalkyl modified polyalkylene oxide, perfluoro polyalkylene oxide.
- fluoroalkyi modified oils e.g. perfluorinated oils, perfluoroalkyl-modified polysiloxane, perfluoro siloxane, perfluoro polyether, perfluorinated alkanes, or perfluoroalkyl modified polyalkylene oxide, perfluoro polyalkylene oxide.
- Lumiflon LF-200 (Fluoroethylene-Alkyl Vinyl Ether) alternating copolymer.
- the one or more fluorinated oils are selected from fluoroalkyi modified polyoxyalkylene polysiloxane oils (e.g. PEG-PDMS).
- fluoroalkyi modified polyoxyalkylene polysiloxane oils e.g. PEG-PDMS.
- These compounds are polysiloxanes which have been modified with a fluoroalkyi group and polyoxyalkylene, and can have a linear or branched/pendant conformation, or a combination of a linear and branched/pendant conformation.
- the polymer will generally have the structure A-B-C where A is a fluoroalkyi group, B is a polysiloxane and C is a polyoxyalkylene. Therefore, it will only have one fluoroalkyi group and one polyoxyalkylene group per polysiloxane molecule.
- the modification of the polysiloxane molecule is in a branched/pendant conformation, in which the fluoroalkyi group(s) and the polyoxyalkylene group(s) are attached to the polysiloxane backbone in non-terminal positions. This allows more than one of each group per polysiloxane molecule. Examples of possible synthetic routes are disclosed in US 5,445,114. This type of fluorinated oils appears to provide certain advantages.
- Fluorosil 2110 a perfluoro nonyl ethyl PEG- 8 dimethicone and C1910, a fluorinated silicone polyether of the same generic structure as Fluorosil 2110.
- the one or more non-reactive fluorinated oil(s), if present, may be of different types, e.g. two or more of the types described above. If present, the one or more fluorinated oils are typically included in the coating composition in an amount of 0.01-20 %, e.g. 0.05-10 %, by dry weight. In certain embodiments, the one or more fluorinated oils constitutes 0.05-7 % by dry weight, e.g. 0.1-5 % by dry weight, in particular 0.5-3 % by dry weight, of the coating composition. In certain other embodiments, the one or more fluorinated oils constitutes 1-10 % by dry weight, e.g. 2-9 % by dry weight, in particular 2-7 % by dry weight, or 3-7 % by dry weight, or 3-5 % by dry weight, or 4-8 % by dry weight, of the coating composition.
- the one or more fluorinated oils and the one or more hydrophilic-modified polysiloxane oils are typically included in the coating composition in a combined amount of 0.01-20 %, e.g. 0.05-10 %, by dry weight.
- the one or more fluorinated oils constitutes 0.05-7 % by dry weight, e.g. 0.1-5 % by dry weight, in particular 0.5-5 % by dry weight, of the coating composition.
- the one or more fluorinated oils constitutes 1-10 % by dry weight, e.g. 2-9 % by dry weight, in particular 2-7 % by dry weight, or 3-7 % by dry weight, or 2-6 % by dry weight, or 3-5 % by dry weight, or 4-8 % by dry weight, of the coating composition.
- the coating composition used for forming a cured fouling control coat may further comprise solvents and additives.
- solvents are aliphatic, cycloaliphatic and aromatic hydrocarbons such as white spirit, cyclohexane, toluene, xylene and naphtha solvent, esters such as methoxypropyl acetate, n-butyl acetate and 2-ethoxyethyl acetate; octamethyltrisiloxane, and mixtures thereof.
- the solvent system may include water or be water-based (>50% water in the solvent system).
- the solvents are selected from aliphatic, cycloaliphatic and aromatic hydrocarbons such as white spirit, cyclohexane, toluene, xylene and naphtha solvent, esters such as methoxypropyl acetate, n-butyl acetate and 2-ethoxyethyl acetate;
- octamethyltrisiloxane and mixtures thereof, preferably those solvents having a boiling point of 110 °C or more.
- additives are:
- non-reactive fluids such as organopolysiloxanes; for example polydimethylsiloxane, methylphenyl polysiloxane; petroleum oils and combinations thereof;
- surfactants such as derivatives of propylene oxide or ethylene oxide such as alkylphenol- ethylene oxide condensates (alkylphenol ethoxylates); ethoxylated monoethanolamides of unsaturated fatty acids such as ethoxylated monoethanolamides of linoleic acid; sodium dodecyl sulfate; and soya lecithin;
- thickeners and anti-settling agents e.g. thixotropic agents
- thixotropic agents such as colloidal silica, hydrated aluminium silicate (bentonite), aluminium tristearate, aluminium monostearate, xanthan gum, chrysotile, pyrogenic silica, hydrogenated castor oil, organo-modified clays, polyamide waxes and polyethylene waxes;
- dyes such as l,4-bis(butylamino)anthraquinone and other anthraquinone derivatives; toluidine dyes, etc. ;
- antioxidants such as bis(tert-butyl) hydroquinone, 2,6-bis(tert-butyl) phenol, resorcinol, 4-tert-butyl catechol, tris(2,4-di-tert-butylphenyl)phosphite, pentaerythritol Tetrakis(3-(3,5- di-tert-butyl-4-hydroxyphenyl)propionate), bis(2,2,6,6,-tetramethyl-4-piperidyl)sebacate, etc.
- Any additives typically constitute 0-30 %, such as 0-15 %, by dry weight of the coating composition.
- the coating composition comprises one or more thickeners and/or anti-settling agents (e.g. thixotropic agents), preferably in an amount of 0.2-10 %, such as 0.5-5 %, e.g. 0.6-4 %, by dry weight of the coating composition.
- thickeners and/or anti-settling agents e.g. thixotropic agents
- the coating composition used for forming a cured fouling control coat may comprise pigments and fillers.
- Pigments and fillers are in the present context viewed in conjunction as constituents that may be added to the coating composition with only limited implications on the adhesion properties. "Pigments” are normally characterised in that they render the final paint coating non-transparent and non-translucent, whereas “fillers” normally are characterised in that they do not render the paint non-translucent and therefore do not contribute significantly to hide any material below the coating.
- pigments are grades of titanium dioxide, red iron oxide, zinc oxide, carbon black, graphite, yellow iron oxide, red molybdate, yellow molybdate, zinc sulfide, antimony oxide, sodium aluminium sulfosilicates, quinacridones, phthalocyanine blue, phthalocyanine green, black iron oxide, indanthrone blue, cobalt aluminium oxide, carbazole dioxazine, chromium oxide, isoindoline orange, bis-acetoacet-o-tolidiole, benzimidazolon, quinaphtalone yellow, isoindoline yellow, tetrachloroisoindolinone, quinophthalone yellow.
- fillers examples include calcium carbonate such as calcite, dolomite, talc, mica, feldspar, barium sulfate, kaolin, nephelin, silica, perlite, magnesium oxide, and quartz flour, etc. Fillers (and pigments) may also be added in the form of nanotubes or fibres, thus, apart from the before-mentioned examples of fillers, the coating composition may also comprise fibres, e.g. those generally and specifically described in WO 00/77102 which is hereby incorporated by reference.
- Any pigments and/or fillers typically constitute 0-60 %, such as 0-50 %, preferably 5-45 %, such as 5-40 %, or 5-35 %, or 0.5-25 %, or 1-20 %, by dry weight of the coating composition. Taking into account the density of any pigments and/or fillers, such constituents typically constitute 0.2-20 %, such as 0.5-15 % by solids volume of the coating composition
- the coating composition typically has a viscosity in the range of 25-25,000 mPa ⁇ s, such as in the range of 150- 15,000 mPa ⁇ s, in particular in the range of 200-4,000 mPa ⁇ s.
- the coating composition may be prepared by any suitable technique that is commonly used within the field of paint production.
- the various constituents may be mixed together utilizing a mixer, a high speed disperser, a ball mill, a pearl mill, a grinder, a three-roll mill etc.
- the coating compositions are typically prepared and shipped as two- or three-component systems that should be combined and thoroughly mixed immediately prior to use.
- the paints according to the invention may be filtrated using bag filters, patron filters, wire gap filters, wedge wire filters, metal edge filters, EGLM turnoclean filters (ex. Cuno), DELTA strain filters (ex. Cuno), and Jenag Strainer filters (ex. Jenag), or by vibration filtration.
- An example of a suitable preparation method is described in the Examples.
- the coating composition to be used in the method of the invention is typically prepared by mixing two or more components e.g. two pre-mixtures, one pre-mixture comprising the one or more reactive polysiloxane binders and one pre-mixture comprising the one or more cross- linking agents. It should be understood that when reference is made to the coating composition, it is the mixed coating composition ready to be applied. Furthermore, all amounts stated as % by dry weight of the coating composition should be understood as % by dry weight of the mixed paint composition ready to be applied, i.e. the weight apart from the solvents (if any).
- the paint coat (preferably a top coat) comprises:
- the binder in the preferred embodiment above mentioned has been pre-reacted in a moisture free environment to form a single component formulation by admixing 100 parts by weight of silanol terminated polydiorganosiloxane with 0.5-30 parts by weight of hydrolysable cross-linker, such as vinyltrimethoxysilane or a vinyloximosilane.
- the remainder of the paint coat (up to 100 % by dry weight) is preferably made up of one or more constituents selected from additives, catalysts, pigments, fillers, and hydrophilic-modified polysiloxane oils (preferably those selected from the poly(oxyalkylene)-modified polysiloxane oils).
- the paint coats according to the above embodiments include additives, pigments and fillers in the amounts specified further above.
- the coating composition of the invention is typically applied to at least a part of the surface of a substrate.
- applying is used in its normal meaning within the paint industry.
- “applying” is conducted by means of any conventional means, e.g. by brush, by roller, by spraying, by dipping, etc.
- the commercially most interesting way of “applying” the coating composition is by spraying.
- the coating composition is preferably sprayable. Spraying is effected by means of conventional spraying equipment known to the person skilled in the art.
- the coating is typically applied in a dry film thickness of 50-600 ⁇ , such as 50-500 pm, e.g. 75- 400 pm, or 20-100 pm.
- the coating composition is preferably such with respect to sag resistance cf. ASTM D 4400-99 (i.e. relating to its ability to be applied in a suitable film thickness to a vertical surface without sagging) that it exhibits sag resistance for a wet film thickness up to at least 70 ⁇ , such as up to at least 200 ⁇ , preferably up to at least 400 pm, and in particular up to at least 600 pm.
- the term "at least a part of the surface of a substrate” refers to the fact that the coating composition may be applied to any fraction of the surface.
- the coating composition is at least applied to the part of the substrate (e.g. a vessel) where the surface (e.g. the ship's hull) may come in contact with water, e.g. sea-water.
- substrate is intended to mean a solid material onto which the coating composition is applied.
- the substrate typically comprises a metal such as steel, iron, aluminium, or glass- fibre reinforced polyester.
- the substrate is a metal substrate, in particular a steel substrate.
- the substrate is a glass-fibre reinforced polyester substrate.
- the substrate is at least a part of the outermost surface of a marine structure.
- the term "surface” is used in its normal sense, and refers to the exterior boundary of an object. Particular examples of such surfaces are the surface of marine structures, such as vessels (including but not limited to boats, yachts, motorboats, motor launches, ocean liners, tugboats, tankers, container ships and other cargo ships, submarines, and naval vessels of all types), pipes, shore and off-shore machinery, constructions and objects of all types such as piers, pilings, bridge substructures, water-power installations and structures, underwater oil well structures, nets and other aquatic culture installations, and buoys, etc.
- the surface of the substrate may either be the "native" surface (e.g. the steel surface).
- the substrate is typically coated, e.g. with an anticorrosive coating and/or a tie coat, so that the surface of the substrate is constituted by such a coating.
- the (anticorrosive and/or tie) coating is typically applied in a total dry film thickness of 100-600 ⁇ , such as 150-450 pm, e.g. 200-400 pm.
- the substrate may carry a paint coat, e.g. a worn-out fouling control paint coat, or similar.
- the substrate is a metal substrate (e.g. a steel substrate) coated with an anticorrosive coating such as an anticorrosive epoxy-based coating, e.g. cured epoxy-based coating, or a shop-primer, e.g. a zinc-rich shop-primer.
- an anticorrosive coating such as an anticorrosive epoxy-based coating, e.g. cured epoxy-based coating, or a shop-primer, e.g. a zinc-rich shop-primer.
- the substrate is a glass-fiber reinforced polyester substrate coated with an epoxy primer coating.
- fouling release (as well as “fouling control”) relates to all types of bio-fouling of a surface (i.e. settlement of organisms on a surface), in particular surfaces exposed to an aqueous environment or to aqueous liquids (e.g. within tanks, pipes, etc.). It is however, believed that the coatings defined herein are particularly relevant for avoiding or reducing marine bio-fouling, i.e. bio-fouling arising in connection with the exposure of a surface to a marine environment, in particular to sea-water.
- the present invention also provides a marine structure comprising on at least a part of the outer surface thereof an outermost fouling control coating prepared from a coating composition as defined hereinabove.
- an outermost fouling control coating prepared from a coating composition as defined hereinabove.
- at least as part of the outer surface carrying the outermost coating is a submerged part of said structure.
- the fouling control coating system of the marine structure may consist of an anticorrosive layer, a tie-coat and the fouling control coating as described herein.
- the fouling control coating composition is applied on top of a used fouling control coating system, e.g. on top of a used polysiloxane-based fouling control coat.
- the anticorrosive layer has a total dry film thickness of 100-600 pm, such as 150-450 ⁇ , e.g. 200-400 pm;
- the tie-coat has a total dry film thickness of 50-500 pm, such as 50-400 pm, e.g. 75-350 pm or 75-300 pm or 75-250 pm;
- the fouling control coating has a total dry film thickness of 20-500 pm, such as 20-400 pm, e.g. 50-300 pm.
- a further embodiment of the marine structure is that where at least a part of the outermost surface of said structure is coated with a paint system comprising a total dry film thickness of 150-400 pm of an anticorrosive layer of an epoxy-based coating established by application of 1-4, such as 2-4, layers;
- the fouling control coating is applied directly on the anticorrosive layer without the use of tie-coat.
- the invention further relates to the use of the combination of one or more polysiloxane components modified with hydrophilic oligomer/polymer moieties and one or more biocides, wherein the weight ratio between the hydrophilic oligomer/polymer moieties and the one or more biocides is in the range 1 : 0.02 to 1 : 20, for improving the antifouling properties of a polysiloxane based coating composition.
- the combination is particularly relevant for improving the antifouling properties against slime and algae.
- the coating compositions defined herein may comprise one, two or more types of the individual constituents.
- the total amount of the respective constituent should correspond to the amount defined above for the individual constituent.
- compound(s) in the expressions: compound(s), polysiloxane(s), agent(s), etc. indicates that one, two or more types of the individual constituents may be present.
- Aerosil R8200 ex. Evonik Industries - Germany, Hexamethyldisilazane treated fumed silica DC550, ex. Dow Corning - USA, non-reactive methylphenyl polysiloxane
- Neostann U-12 ex. Nitto Kasai - Japan, Dibutyltin Dilaurate
- viscosity is measured at 25 °C in accordance with ISO 2555: 1989.
- HMP1 Pendant hvdrophilic modified polvsiloxane
- a pendant, curable poly(ethylene glycol) modified polysiloxane is prepared by mixing 25.0 g (polydimethylsiloxane-methylhydrosiloxane, hydride terminated) dissolved in 50.0 g water free toluene, with 0.013 g of a solution of platinum-divinyltetramethyldisiloxane complex in xylene in a three necked flask with reflux and a continuous flow of dry nitrogen gas to keep the reaction atmosphere moisture free.
- a magnetic stirrer is used in the reaction flask to keep the solution agitated during the synthesis. The solution is heated to 80°C.
- a linear, curable poly(ethylene glycol) modified polysiloxane is prepared by mixing 5.00 g (polyethylene glycol diallyl ether) dissolved in 50.0 g water free toluene, with 0.013 g of a solution of platinum-divinyltetramethyldisiloxane complex in xylene in a three necked flask with reflux and a continuous flow of dry nitrogen gas to keep the reaction atmosphere moisture free.
- a magnetic stirrer is used in the reaction flask to keep the solution agitated during the synthesis.
- the solution is heated to 65°C. To this solution, 65.0 g
- polydimethylsiloxane (hydride terminated) is added dropwise, and allowed to react for 1 h at 80°C. After the completion of the reaction, 35.0 g of vinyltrimethoxysilane is added dropwise at 80°C and allowed to react for 1 h at 80°C.
- the solvent was hereafter partially removed under reduced pressure using a rotary evaporator until the solvent content reached 10% to obtain the final product HMP2.
- the content of PEG-modified PDMS binder in HMP2 is 90 % w/w.
- the amount of PEG in the HMP2 binder is 4.8% w/w calculated on dry weight.
- a pendant, curable poly(ethylene glycol) modified polysiloxane is prepared by mixing 25.0 g polydimethylsiloxane-methylhydrosiloxane, hydride terminated dissolved in 50.0 g water free toluene, with 0.14 g platinum - divinyltetramethyldisiloxane complex in xylene solution. The solution is heated to 80°C. To this solution, 4.0 g of vinyltrimethoxysilane is added dropwise and allowed to react for 1 /2h at 80°C. After the completion of the reaction, 68.5 g
- polyethylene glycol mono allyl ether [A350] is added drop wise, and allowed to react for 3h at 80°C.
- the content of PEG-modified PDMS binder in HMP3 is 66.1 % w/w.
- the amount of PEG in the HMP3 binder is 70.3% w/w calculated on dry weight.
- a linear, curable poly(ethylene glycol) modified polysiloxane is prepared by mixing 100.0 g of hydride terminated polydimethylsiloxane dissolved in 75.0 g water free toluene, with 0.17 g of platinum - divinyltetramethyldisiloxane complex in xylene solution. The solution is heated to 80°C under stirring. To this solution 1.7 g polyethylene glycol di allyl ether [AA300] is added dropwise, and allowed to react for 2h at 80°C. After the completion of the reaction 5.4 g of vinyltrimethoxysilane is added dropwise at 80°C and allowed to react for lh at 80°C.
- the content of PEG-modified PDMS binder in HMP4 is 58.8 % w/w.
- the amount of PEG in the HMP4 binder is 1.6% w/w calculated on dry weight.
- a pendant, curable poly(ethylene glycol) modified polysiloxane is prepared by mixing 25.0 g polydimethylsiloxane-methylhydrosiloxane, hydride terminated dissolved in 50.0 g water free toluene, with 0.14 g platinum - divinyltetramethyldisiloxane complex in xylene solution. The solution is heated to 80°C. To this solution, 4.0 g of vinyltrimethoxysilane is added dropwise and allowed to react for 1 /2h at 80°C. After the completion of the reaction, 90.0 g
- compositions A to Z are added dropwise, and allowed to react for 3h at 80°C.
- the content of PEG-modified PDMS binder in HMP5 is 70.4 % w/w.
- the amount of PEG in the HMP5 binder is 75.6% w/w calculated on dry weight.
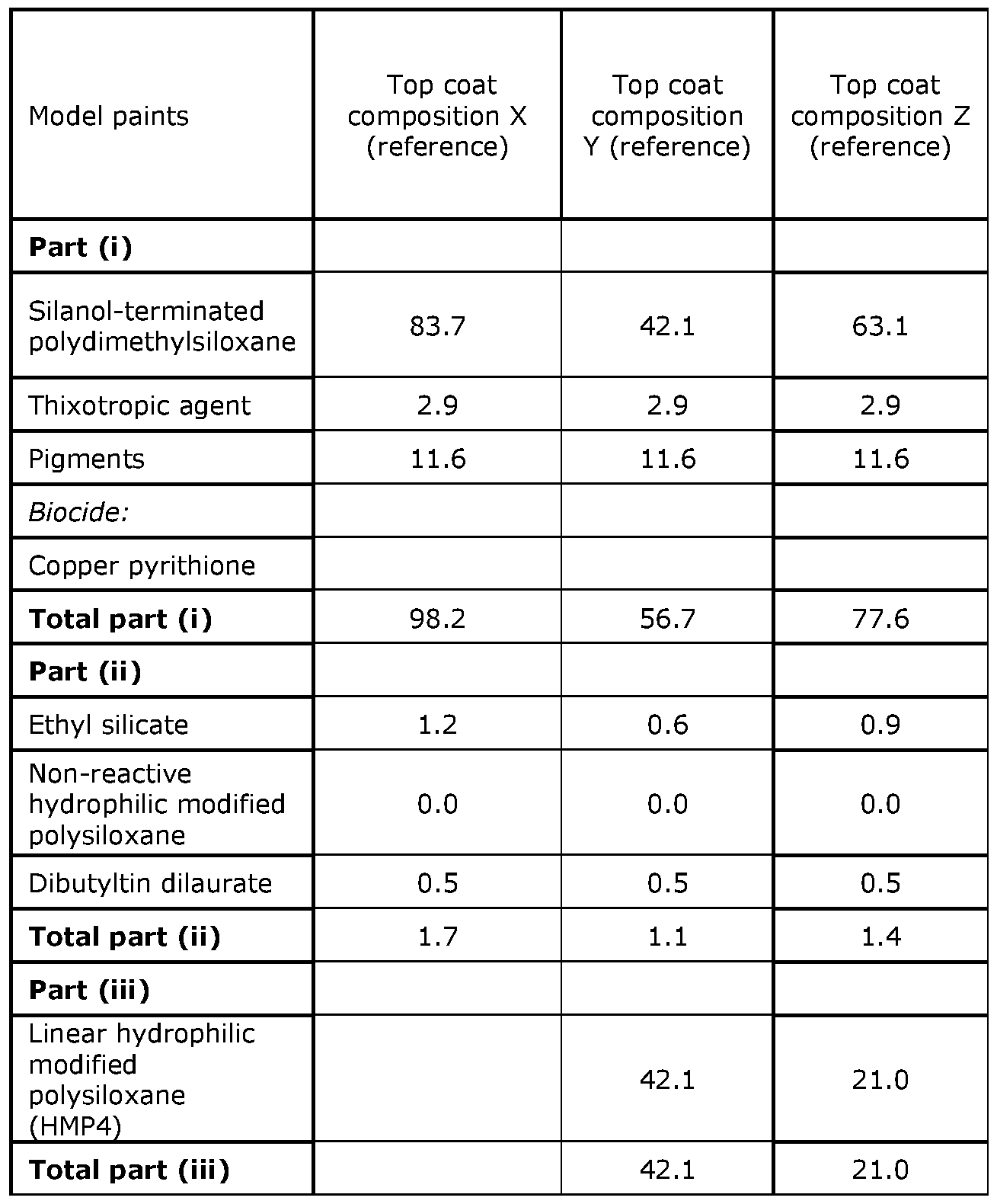
- Part (i) silanol-terminated polydimethylsiloxane, xylene, silica, polyamide wax, red iron oxide, (biocide) are mixed on a Diaf dissolver equipped with an impeller disc (70 mm in diameter) in a 1 L can for 15 minutes at 2000 rpm.
- Part (ii) ethyl silicate, (xylene), catalyst, 2,4-pentanedione, (non-reactive hydrophilic modified polysiloxane oil), (phenyl methyl polysiloxane) are mixed on a Diaf dissolver equipped with an impeller disc (70 mm in diameter) in a 1 L can for 2 minutes at 500 rpm.
- Part (iii) Reactive hydrophilic modified polysiloxane
- the container is closed under dry nitrogen to avoid moisture.
- part (i), part (ii) and part (iii) are mixed to a homogenous mixture.
- the Blister Box test is used to determine the influence of polysiloxanes modified with hydrophilic oligomer/polymer moieties on the stability of the PDMS coating to which they are added to.
- Steel panels (150x75x15 mm) are coated with 100 pm (dry film thickness, DFT) of a commercial epoxy primer (HEMPADUR Quattro 17634) applied by airless spraying. After 12 - 48 hrs of drying at room temperature a silicone tie coat (HEMPASIL Nexus 27302) is applied by doctor blade of 300 pm clearance. After 16-30 hrs of drying the top coat paint
- compositions are applied by doctor blade of 400 pm clearance.
- the panels are dried for 24 hrs before testing in the blister box.
- the panel surface with the coating system is exposed to 40 °C, saturated water vapour, at an angle of 15°/60° to the horizontal.
- the reverse side of the panel is exposed to room temperature. At the selected inspection intervals during and after completion of exposure, adhesion between tie coat/top coat and general condition of the top coat are evaluated.
- Adhesion Ranking Value FAIL/ POOR No adhesion/ poor adhesion
- Panels are exposed for two months and typically checked every week.
- composition A composition B
- the examples A, B illustrate the performance of the biocide containing paint compositions when the hydrophilic polymers have been incorporated in the paint matrix.
- Comments: The examples C, D illustrate the influence of the addition of a hydrophilic modified polysiloxane oil in a composition with hydrophilic oligomer/polymer moieties reacted to the binder.
- Reference example E shows a biocide containing paint formulation without hydrophilic moieties reacted to the binder.
- the coating compositions are provided in terms of the dry weight of the cured film.
- example F and reference example G show the improvement of performance when adding biocide to a hydrophilic modified polysiloxane-based fouling release coating.
- compositions L to O are further examples of the performance of a hydrophilic modified polysiloxane binder. Also shown in the examples L to O are the different ratios between biocide and hydrophilic oligomer/polymer that gives rise to an improved
- composition H
- the example R shows the effect of a different biocide in a hydrophilic modified polysiloxane binder matrix.
- Examples P and Q shows the effect of addition of non-reactive polysiloxane oils in a biocide containing, hydrophilic modified polysiloxane binder.
- composition S composition T composition U composition V (reference)
- the examples S, T, U and V show the performance improvement of adding biocides to an ABA type hydrophilic modified polysiloxane binder.
- Reference examples H and X is an unmodified polysiloxane binder composition with and without biocide, respectively.
- Reference examples Y and Z illustrate the performance of a linear hydrophilic modified polysiloxane without biocides.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Paints Or Removers (AREA)
- Laminated Bodies (AREA)
- Application Of Or Painting With Fluid Materials (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
Claims
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201280031779.2A CN103732698B (en) | 2011-06-30 | 2012-06-29 | Fouling control coating compositions |
| BR112013031646A BR112013031646A2 (en) | 2011-06-30 | 2012-06-29 | dirt accumulation control coating compositions |
| AU2012278334A AU2012278334B2 (en) | 2011-06-30 | 2012-06-29 | Fouling control coating compositions |
| EP12805420.2A EP2726561B1 (en) | 2011-06-30 | 2012-06-29 | Fouling control coating compositions |
| JP2014517465A JP2014524949A (en) | 2011-06-30 | 2012-06-29 | Adhesion suppression coating composition |
| KR1020147001422A KR102078782B1 (en) | 2011-06-30 | 2012-06-29 | Fouling control coating compositions |
| EP19189362.7A EP3617278A1 (en) | 2011-06-30 | 2012-06-29 | Novel fouling control coating compositions |
| NZ618053A NZ618053B2 (en) | 2011-06-30 | 2012-06-29 | Fouling control coating compositions |
| US14/129,796 US20140135422A1 (en) | 2011-06-30 | 2012-06-29 | Fouling control coating compositions |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP11172169 | 2011-06-30 | ||
| EP11172169.2 | 2011-06-30 | ||
| EP11172163.5 | 2011-06-30 | ||
| EP11172163 | 2011-06-30 | ||
| EP11172166 | 2011-06-30 | ||
| EP11172166.8 | 2011-06-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013000479A1 true WO2013000479A1 (en) | 2013-01-03 |
Family
ID=47423436
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/DK2012/050228 WO2013000479A1 (en) | 2011-06-30 | 2012-06-29 | Fouling control coating compositions |
| PCT/DK2012/050227 WO2013000478A1 (en) | 2011-06-30 | 2012-06-29 | Fouling control coating compositions comprising polysiloxane and pendant hydrophilic oligomer/polymer moieties |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/DK2012/050227 WO2013000478A1 (en) | 2011-06-30 | 2012-06-29 | Fouling control coating compositions comprising polysiloxane and pendant hydrophilic oligomer/polymer moieties |
Country Status (11)
| Country | Link |
|---|---|
| US (2) | US20140135422A1 (en) |
| EP (4) | EP2726561B1 (en) |
| JP (2) | JP2014524949A (en) |
| KR (1) | KR102078782B1 (en) |
| CN (3) | CN107936825A (en) |
| AU (2) | AU2012278334B2 (en) |
| BR (1) | BR112013031646A2 (en) |
| PL (3) | PL2726558T5 (en) |
| PT (1) | PT2726561T (en) |
| SG (1) | SG10201605381PA (en) |
| WO (2) | WO2013000479A1 (en) |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014131695A1 (en) | 2013-02-26 | 2014-09-04 | Akzo Nobel Coatings International B.V. | Anti-fouling compositions with a fluorinated oxyalkylene-containing polymer or oligomer |
| US20150353741A1 (en) * | 2013-02-15 | 2015-12-10 | Momentive Performance Materials Inc. | Antifouling system comprising silicone hydrogel |
| JP2016518969A (en) * | 2013-03-20 | 2016-06-30 | ヘンペル エイ/エス | New polysiloxane antifouling coating system |
| JP2016522848A (en) * | 2013-05-03 | 2016-08-04 | ヘンペル エイ/エス | Novel polysiloxane-based fouling release coat |
| WO2017009297A1 (en) | 2015-07-13 | 2017-01-19 | Jotun A/S | Antifouling composition |
| JP2017525786A (en) * | 2014-07-11 | 2017-09-07 | ヘンペル エイ/エス | Novel polysiloxane-based fouling release coat containing poly (oxyalkylene) -modified alcohol |
| US9920216B2 (en) | 2013-03-27 | 2018-03-20 | Hempel A/S | Curing agent for tie-coat composition comprising an amino-silane adduct |
| WO2018134291A1 (en) | 2017-01-19 | 2018-07-26 | Jotun A/S | Antifouling composition |
| EP3077446B1 (en) * | 2013-12-05 | 2018-08-22 | PPG Coatings Europe B.V. | A coating composition |
| WO2019115020A1 (en) | 2017-12-14 | 2019-06-20 | Akzo Nobel Coatings International B.V. | Foul release coating composition, substrate coated with such coating composition, and use of such coating composition |
| WO2019233985A1 (en) | 2018-06-04 | 2019-12-12 | Hempel A/S | Method for establishing a fouling release coating system |
| GB202107159D0 (en) | 2021-03-23 | 2021-06-30 | Jotun As | Monitoring a vessel |
| WO2021180588A2 (en) | 2020-03-09 | 2021-09-16 | Jotun A/S | Hull cleaning robot |
| US11279835B2 (en) | 2016-11-11 | 2022-03-22 | Hempel A/S | Antifouling coating composition comprising novel carbon-based hydrolysable polymers |
| WO2022200430A1 (en) | 2021-03-23 | 2022-09-29 | Jotun A/S | Monitoring the cleanliness of an underwater surface of a stationary object |
| WO2023139212A1 (en) | 2022-01-21 | 2023-07-27 | Danmarks Tekniske Universitet | Curable polysiloxane coating composition comprising polysilazane |
| WO2023170427A1 (en) * | 2022-03-11 | 2023-09-14 | Mark Ieuan Edwards | Anti-fouling polymeric agents |
| CN118500304A (en) * | 2024-07-19 | 2024-08-16 | 南京航空航天大学 | Visual guidance shooting method for measuring concave-convex amount of surface fastener of aviation aircraft |
Families Citing this family (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MY160309A (en) | 2010-10-20 | 2017-02-28 | Valspar Sourcing Inc | Water-based coating system with improved adhesion to a wide range of coated and uncoated substrates including muffler grade stainless steel |
| KR20150082501A (en) * | 2012-11-13 | 2015-07-15 | 주고꾸 도료 가부시키가이샤 | Antifouling coating composition, antifouling coating film, antifouling substrate, and method for improving storage stability of antifouling coating composition |
| US9718920B2 (en) | 2012-12-19 | 2017-08-01 | 3M Innovative Properties Company | Reactive polyoxazolines having a perfluorinated group |
| WO2014099518A2 (en) * | 2012-12-19 | 2014-06-26 | 3M Innovative Properties Company | Polyoxazoline copolymers |
| CN105120973A (en) * | 2012-12-25 | 2015-12-02 | 墨尔本大学 | Materials and methods |
| KR20150136014A (en) * | 2014-05-26 | 2015-12-04 | 닛신 가가꾸 고교 가부시끼가이샤 | Ink composition |
| KR20150136013A (en) * | 2014-05-26 | 2015-12-04 | 닛신 가가꾸 고교 가부시끼가이샤 | Ink composition |
| US20150247042A1 (en) * | 2014-09-10 | 2015-09-03 | N1 Technologies | Antifouling Paint Formulation Incorporating Tungsten Nanotubes |
| AU2015321646B2 (en) * | 2014-09-26 | 2017-11-30 | Alcon Inc. | Polymerizable polysiloxanes with hydrophilic substituents |
| CN106999981B (en) | 2014-12-02 | 2020-12-04 | 中国涂料株式会社 | Method for reinforcing fouling-inhibiting coating film |
| JP2017088653A (en) * | 2015-11-04 | 2017-05-25 | 中国塗料株式会社 | Coating composition, antifouling coated film, antifouling substrate and manufacturing method of antifouling substrate |
| JP6635589B2 (en) * | 2015-12-28 | 2020-01-29 | ライオン株式会社 | Antifungal composition |
| US10167414B1 (en) * | 2016-03-14 | 2019-01-01 | Thomas O'Lenick | Alkyl silicones as pigment coatings |
| EP3489010B1 (en) * | 2016-04-05 | 2020-06-10 | Adaptive Surface Technologies, Inc. | Curable polysiloxane compositions and slippery materials and coatings and articles made therefrom |
| KR102489614B1 (en) * | 2016-05-27 | 2023-01-16 | 삼성전자주식회사 | Oxazoline modified copolymer, composition including oxazoline modified copolymer, and article manufactured from the composition |
| CN109890917B (en) * | 2016-11-01 | 2022-01-04 | 科慕埃弗西有限公司 | Coatings incorporating non-fluorinated cleanability additives |
| JP2020503394A (en) * | 2016-11-01 | 2020-01-30 | ザ ケマーズ カンパニー エフシー リミテッド ライアビリティ カンパニー | Non-fluorinated coatings and finishes |
| WO2018160820A1 (en) * | 2017-03-02 | 2018-09-07 | Ppg Architectural Finishes, Inc. | Sealant composition |
| CN115717337A (en) * | 2017-04-17 | 2023-02-28 | 康奈尔大学 | Fluorine-free oil-proof coating and manufacturing method and application thereof |
| KR101973961B1 (en) * | 2017-08-14 | 2019-04-30 | 주식회사 비앤비 | Coating agent composition for preventing the attachment of marine organisms |
| EP3470475B1 (en) | 2017-10-13 | 2021-01-27 | Evonik Operations GmbH | Curable composition for coatings with anti-adhesive property |
| CN108088792B (en) * | 2017-11-27 | 2021-04-02 | 中国船舶重工集团公司第七二五研究所 | Intermediate scale sample and preparation method and application thereof |
| CN110117365B (en) * | 2018-02-07 | 2021-12-14 | 奇美实业股份有限公司 | Modified polymethylsiloxane, modified high-cis conjugated diene polymer, method for producing same, rubber composition, and tire |
| US11535772B2 (en) | 2018-06-08 | 2022-12-27 | Adaptive Surface Technologies, Inc. | Sidechain functionalized organosiloxanes, coating compositions containing sidechain functionalized organosiloxanes, coated articles, and methods of making and methods of use thereof |
| JP2020200367A (en) * | 2019-06-06 | 2020-12-17 | 日東化成株式会社 | Antifouling coating composition |
| JP7351512B2 (en) * | 2019-10-01 | 2023-09-27 | 日東化成株式会社 | Antifouling paint composition |
| KR102117328B1 (en) * | 2019-11-25 | 2020-06-01 | 전상진 | Passive type dial gear transmission apparatus |
| EP3828240A1 (en) * | 2019-11-29 | 2021-06-02 | Jotun A/S | Fouling release coating |
| EP3974481A1 (en) * | 2020-09-29 | 2022-03-30 | Jotun A/S | Fouling release coating composition |
| CN113537759B (en) * | 2021-07-13 | 2023-08-04 | 上海银行股份有限公司 | Weight self-adaption-based user experience measurement model |
| CN115785803A (en) * | 2021-09-09 | 2023-03-14 | 涂创时代(苏州)科技开发有限公司 | Nontoxic amphiphilic organic silicon polymer-based marine antifouling paint and preparation method and application thereof |
| DE102021125789B3 (en) | 2021-10-05 | 2022-09-15 | Bio-Gate Ag | Biocorrodible solid and method of coating a solid |
| CN114716913B (en) * | 2022-03-17 | 2023-03-03 | 中国船舶重工集团公司第七二五研究所 | Amphiphilic antifouling active agent and preparation method thereof |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1993013179A1 (en) * | 1991-12-20 | 1993-07-08 | Courtaulds Coatings (Holdings) Limited | Coating compositions |
| US6291549B1 (en) * | 1996-10-24 | 2001-09-18 | Bayer Aktiengesellschaft | Antifouling paints |
| WO2001094487A2 (en) * | 2000-06-02 | 2001-12-13 | Microphase Coatings, Inc. | Antifouling coating composition |
| US20080116493A1 (en) * | 2006-11-17 | 2008-05-22 | Adkisson James W | Multi-layer spacer with inhibited recess/undercut and method for fabrication thereof |
| WO2008132195A1 (en) * | 2007-05-01 | 2008-11-06 | Akzo Nobel Coatings International B.V. | Anti-fouling coating compositions containing a carboxyl-functional organosilicone |
| WO2011076856A1 (en) * | 2009-12-22 | 2011-06-30 | Hempel A/S | Novel fouling control coating compositions |
Family Cites Families (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6015457A (en) * | 1983-07-08 | 1985-01-26 | Toray Silicone Co Ltd | Room temperature curing organo-polysiloxane composition |
| GB8803458D0 (en) * | 1988-02-15 | 1988-03-16 | Int Paint Plc | Inhibition of marine fouling |
| JP2952375B2 (en) * | 1990-03-05 | 1999-09-27 | 関西ペイント株式会社 | Non-toxic antifouling paint composition |
| WO1992020664A1 (en) * | 1991-05-10 | 1992-11-26 | Sunkyong Industries Co., Ltd. | A process for preparing 4-isothiazolin-3-one |
| JPH05263022A (en) | 1992-03-17 | 1993-10-12 | Kansai Paint Co Ltd | Coating composition for preventing aquatic organism attachment |
| US5445114A (en) | 1993-12-10 | 1995-08-29 | Walker; Douglas M. | Training harness for use when practicing rock climbing and sport climbing |
| WO1995032862A1 (en) * | 1994-05-31 | 1995-12-07 | The Government Of The United States Of America, Represented By The Secretary Of The Navy | Antifouling and foul-release coatings |
| JP3910228B2 (en) * | 1995-12-26 | 2007-04-25 | 中国塗料株式会社 | Underwater antifouling agent composition, and fishing net and underwater structure antifouling treatment method using the same |
| US6864269B2 (en) | 1996-04-12 | 2005-03-08 | University Of Arkansas | Concentrated, non-foaming solution of quarternary ammonium compounds and methods of use |
| JP3120731B2 (en) * | 1996-04-26 | 2000-12-25 | 信越化学工業株式会社 | Underwater antifouling agent |
| US5919689A (en) * | 1996-10-29 | 1999-07-06 | Selvig; Thomas Allan | Marine antifouling methods and compositions |
| DE69806860T2 (en) † | 1997-06-16 | 2003-03-13 | General Electric Co., Schenectady | RTV anti-fouling silicone coatings and objects coated with them |
| FR2769469B1 (en) * | 1997-10-15 | 1999-11-26 | Rhodia Chimie Sa | BIOCIDE AND POLYETHER SILICONE SYSTEM AND USE THEREOF FOR HARD SURFACE DISINFECTION |
| JP3952099B2 (en) * | 1998-03-24 | 2007-08-01 | 信越化学工業株式会社 | Underwater antifouling agent |
| WO2000077102A1 (en) | 1999-06-11 | 2000-12-21 | J.C. Hempel's Skibsfarve-Fabrik A/S | Self-polishing marine antifouling paint composition comprising silicon-containing co-polymers and fibres |
| JP4902037B2 (en) * | 1999-07-08 | 2012-03-21 | 中国塗料株式会社 | Antifouling agent composition, antifouling treatment substrate, and antifouling treatment method for substrate |
| US6451437B1 (en) | 1999-10-13 | 2002-09-17 | Chugoku Marine Paints, Ltd. | Curable composition, coating composition, paint, antifouling paint, cured product thereof and method of rendering base material antifouling |
| US6313193B1 (en) | 2000-06-02 | 2001-11-06 | Microphase Coatings Inc. | Antifouling coating composition |
| US6559201B2 (en) * | 2000-06-02 | 2003-05-06 | Microphase Coatings, Inc. | Antifouling coating composition |
| JP2002212505A (en) | 2001-01-23 | 2002-07-31 | Hitachi Chem Co Ltd | Optical semiconductor metal-organic substance mixture, optical semiconductor metal-containing composition, method for producing photocatalyst coating film and photocatalyst member |
| KR100883736B1 (en) * | 2001-03-21 | 2009-02-12 | 아크조 노벨 엔.브이. | Anti-fouling compositions with a fluorinated alkyl- or alkoxy-containing polymer or oligomer |
| US6635305B2 (en) | 2001-04-26 | 2003-10-21 | Ict Coating N.V. | Process for coating a siliceous substrate with a silicon containing layer |
| JP3835796B2 (en) * | 2002-07-03 | 2006-10-18 | 信越化学工業株式会社 | Room temperature curable organopolysiloxane composition |
| US20080038241A1 (en) * | 2004-07-01 | 2008-02-14 | Biolocus A/S | Self-Polishing Anti-Fouling coating Compositions Comprising An Enzyme |
| JP2006052283A (en) | 2004-08-11 | 2006-02-23 | Kansai Paint Co Ltd | Method for producing aqueous dispersion of organopolysiloxane |
| JP2008527144A (en) | 2005-01-19 | 2008-07-24 | エヌディーエスユー リサーチ ファウンデーション | Polysiloxane with antifouling activity |
| CN101155647B (en) * | 2005-04-05 | 2012-09-05 | 中国涂料株式会社 | Tie coat for organopolysiloxane antifouling coat, composite coats, and ships and underwater structures covered with the composite coats |
| JP2006299132A (en) * | 2005-04-22 | 2006-11-02 | Kansai Paint Co Ltd | Antifouling coating composition |
| US7771833B2 (en) † | 2005-05-09 | 2010-08-10 | Ndsu Research Foundation | Anti-fouling materials containing cationic polysiloxanes |
| EP1922372A4 (en) * | 2005-09-06 | 2010-11-10 | Novus Int Inc | Marine antifouling coating compositions |
| DE102005060302A1 (en) * | 2005-12-16 | 2007-06-28 | Basf Coatings Ag | Aqueous coating material, useful for preparing e.g. thermoplastic and duroplastic materials, comprises an olefin saturated grafted ionically or non-ionically stabilized polyurethane, a surfactant or dispersion agent and an organic solvent |
| BRPI0700482B1 (en) | 2006-03-16 | 2017-04-04 | Rohm & Haas | coating composition |
| JP5153649B2 (en) * | 2006-12-25 | 2013-02-27 | 中国塗料株式会社 | Curable composition, antifouling coating composition, antifouling coating film, substrate with antifouling coating, and method for antifouling substrate |
| US20080166493A1 (en) * | 2007-01-09 | 2008-07-10 | Inframat Corporation | Coating compositions for marine applications and methods of making and using the same |
| DE102007011553A1 (en) * | 2007-03-09 | 2008-09-11 | Chemetall Gmbh | A method of coating metallic surfaces with an aqueous polymer-containing composition, the aqueous composition, and the use of the coated substrates |
| NZ580734A (en) | 2007-05-01 | 2011-04-29 | Akzo Nobel Coatings Int Bv | Antifouling coating composition based on curable polyorganosiloxane polyoxyalkylene copolymers |
| GB0708347D0 (en) * | 2007-05-01 | 2007-06-06 | Dow Corning | Polymer compositions |
| US8053535B2 (en) | 2007-07-11 | 2011-11-08 | Ndsu Research Foundation | Polysiloxanes with anti-fouling activity |
| US9744527B2 (en) * | 2008-05-29 | 2017-08-29 | Bluestar Silicones France Sas | Article having antifouling properties for aquatic and particularly sea use |
| JP4798396B2 (en) * | 2008-07-07 | 2011-10-19 | 信越化学工業株式会社 | Underwater biological adhesion prevention coating composition and underwater structure using the same |
| JP4952952B2 (en) * | 2008-09-12 | 2012-06-13 | 信越化学工業株式会社 | Method for producing room temperature curable organopolysiloxane composition |
| CN101397817B (en) * | 2008-11-07 | 2010-09-29 | 广东同步化工股份有限公司 | Coating method of underwater curing anti-corrosive antifouling paint for sea facilities |
| KR20120101516A (en) † | 2009-12-04 | 2012-09-13 | 엔디에스유 리서치 파운데이션 | Amphiphilic fouling release coatings |
| WO2013052181A2 (en) * | 2011-06-17 | 2013-04-11 | Ndsu Research Foundation | Functionalized silicones with polyalkylene oxide side chains |
-
2012
- 2012-06-29 WO PCT/DK2012/050228 patent/WO2013000479A1/en active Application Filing
- 2012-06-29 CN CN201711259553.9A patent/CN107936825A/en active Pending
- 2012-06-29 JP JP2014517465A patent/JP2014524949A/en active Pending
- 2012-06-29 AU AU2012278334A patent/AU2012278334B2/en active Active
- 2012-06-29 PL PL12805023T patent/PL2726558T5/en unknown
- 2012-06-29 US US14/129,796 patent/US20140135422A1/en not_active Abandoned
- 2012-06-29 PL PL12805177T patent/PL2726559T3/en unknown
- 2012-06-29 EP EP12805420.2A patent/EP2726561B1/en active Active
- 2012-06-29 KR KR1020147001422A patent/KR102078782B1/en active IP Right Grant
- 2012-06-29 EP EP12805023.4A patent/EP2726558B2/en active Active
- 2012-06-29 BR BR112013031646A patent/BR112013031646A2/en not_active Application Discontinuation
- 2012-06-29 CN CN201280031779.2A patent/CN103732698B/en active Active
- 2012-06-29 PT PT128054202T patent/PT2726561T/en unknown
- 2012-06-29 AU AU2012278333A patent/AU2012278333B2/en active Active
- 2012-06-29 PL PL12805420T patent/PL2726561T3/en unknown
- 2012-06-29 EP EP19189362.7A patent/EP3617278A1/en active Pending
- 2012-06-29 EP EP16180746.6A patent/EP3109284A1/en active Pending
- 2012-06-29 SG SG10201605381PA patent/SG10201605381PA/en unknown
- 2012-06-29 CN CN201280031777.3A patent/CN103717682B/en active Active
- 2012-06-29 WO PCT/DK2012/050227 patent/WO2013000478A1/en active Application Filing
- 2012-06-29 US US14/127,485 patent/US20140170426A1/en not_active Abandoned
-
2017
- 2017-06-05 JP JP2017110696A patent/JP6677675B2/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1993013179A1 (en) * | 1991-12-20 | 1993-07-08 | Courtaulds Coatings (Holdings) Limited | Coating compositions |
| US6291549B1 (en) * | 1996-10-24 | 2001-09-18 | Bayer Aktiengesellschaft | Antifouling paints |
| WO2001094487A2 (en) * | 2000-06-02 | 2001-12-13 | Microphase Coatings, Inc. | Antifouling coating composition |
| US20080116493A1 (en) * | 2006-11-17 | 2008-05-22 | Adkisson James W | Multi-layer spacer with inhibited recess/undercut and method for fabrication thereof |
| WO2008132195A1 (en) * | 2007-05-01 | 2008-11-06 | Akzo Nobel Coatings International B.V. | Anti-fouling coating compositions containing a carboxyl-functional organosilicone |
| WO2011076856A1 (en) * | 2009-12-22 | 2011-06-30 | Hempel A/S | Novel fouling control coating compositions |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2726561A1 * |
Cited By (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9988497B2 (en) * | 2013-02-15 | 2018-06-05 | Momentive Performance Materials Inc. | Antifouling system comprising silicone hydrogel |
| US20150353741A1 (en) * | 2013-02-15 | 2015-12-10 | Momentive Performance Materials Inc. | Antifouling system comprising silicone hydrogel |
| WO2014131695A1 (en) | 2013-02-26 | 2014-09-04 | Akzo Nobel Coatings International B.V. | Anti-fouling compositions with a fluorinated oxyalkylene-containing polymer or oligomer |
| EP3323861A1 (en) | 2013-03-20 | 2018-05-23 | Hempel A/S | Novel polysiloxane-based fouling control coating systems |
| US11787953B2 (en) | 2013-03-20 | 2023-10-17 | Hempel A/S | Polysiloxane-based fouling control coating systems |
| JP2016518969A (en) * | 2013-03-20 | 2016-06-30 | ヘンペル エイ/エス | New polysiloxane antifouling coating system |
| US9920216B2 (en) | 2013-03-27 | 2018-03-20 | Hempel A/S | Curing agent for tie-coat composition comprising an amino-silane adduct |
| EP2992056A4 (en) * | 2013-05-03 | 2017-12-27 | Hempel A/S | Novel polysiloxane-based fouling-release coats |
| JP2016522848A (en) * | 2013-05-03 | 2016-08-04 | ヘンペル エイ/エス | Novel polysiloxane-based fouling release coat |
| EP3077446B1 (en) * | 2013-12-05 | 2018-08-22 | PPG Coatings Europe B.V. | A coating composition |
| JP2017525786A (en) * | 2014-07-11 | 2017-09-07 | ヘンペル エイ/エス | Novel polysiloxane-based fouling release coat containing poly (oxyalkylene) -modified alcohol |
| WO2017009297A1 (en) | 2015-07-13 | 2017-01-19 | Jotun A/S | Antifouling composition |
| US10954395B2 (en) | 2015-07-13 | 2021-03-23 | Jotun A/S | Antifouling composition |
| US11279835B2 (en) | 2016-11-11 | 2022-03-22 | Hempel A/S | Antifouling coating composition comprising novel carbon-based hydrolysable polymers |
| WO2018134291A1 (en) | 2017-01-19 | 2018-07-26 | Jotun A/S | Antifouling composition |
| DE112018000434T5 (en) | 2017-01-19 | 2019-10-10 | Jotun A/S | antifouling composition |
| DE112018000434B4 (en) | 2017-01-19 | 2024-05-29 | Jotun A/S | Fouling release coating composition and method for protecting an object from fouling |
| WO2019115020A1 (en) | 2017-12-14 | 2019-06-20 | Akzo Nobel Coatings International B.V. | Foul release coating composition, substrate coated with such coating composition, and use of such coating composition |
| WO2019115021A1 (en) | 2017-12-14 | 2019-06-20 | Akzo Nobel Coatings International B.V. | Substrate coated with a multi-layer coating system and a process for controlling aquatic biofouling on man-made objects using such multi-layer coating system |
| WO2019233985A1 (en) | 2018-06-04 | 2019-12-12 | Hempel A/S | Method for establishing a fouling release coating system |
| WO2021180588A2 (en) | 2020-03-09 | 2021-09-16 | Jotun A/S | Hull cleaning robot |
| GB202107159D0 (en) | 2021-03-23 | 2021-06-30 | Jotun As | Monitoring a vessel |
| WO2022200430A1 (en) | 2021-03-23 | 2022-09-29 | Jotun A/S | Monitoring the cleanliness of an underwater surface of a stationary object |
| WO2022200427A1 (en) | 2021-03-23 | 2022-09-29 | Jotun A/S | Monitoring a vessel |
| WO2023139212A1 (en) | 2022-01-21 | 2023-07-27 | Danmarks Tekniske Universitet | Curable polysiloxane coating composition comprising polysilazane |
| WO2023170427A1 (en) * | 2022-03-11 | 2023-09-14 | Mark Ieuan Edwards | Anti-fouling polymeric agents |
| CN118500304A (en) * | 2024-07-19 | 2024-08-16 | 南京航空航天大学 | Visual guidance shooting method for measuring concave-convex amount of surface fastener of aviation aircraft |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2726561B1 (en) | Fouling control coating compositions | |
| US11787953B2 (en) | Polysiloxane-based fouling control coating systems | |
| EP2899240B1 (en) | Novel polysiloxane-based fouling release coatings including biocide(s) | |
| EP2516559B1 (en) | Novel fouling control coating compositions | |
| AU2012278334A1 (en) | Fouling control coating compositions | |
| AU2012278332A1 (en) | Polysiloxane-based fouling release coats including enzymes | |
| EP3802709B1 (en) | Method for establishing a fouling release coating system | |
| NZ618055B2 (en) | Fouling control coating compositions comprising polysiloxane and pendant hydrophilic oligomer/polymer moieties | |
| NZ618053B2 (en) | Fouling control coating compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12805420 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2012278334 Country of ref document: AU Date of ref document: 20120629 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2014517465 Country of ref document: JP Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012805420 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012805420 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14129796 Country of ref document: US |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013031646 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 20147001422 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 112013031646 Country of ref document: BR Kind code of ref document: A2 Effective date: 20131209 |