WO2012138102A2 - Anticorps humains spécifiques de dlk1 et compositions pharmaceutiques les contenant - Google Patents
Anticorps humains spécifiques de dlk1 et compositions pharmaceutiques les contenant Download PDFInfo
- Publication number
- WO2012138102A2 WO2012138102A2 PCT/KR2012/002496 KR2012002496W WO2012138102A2 WO 2012138102 A2 WO2012138102 A2 WO 2012138102A2 KR 2012002496 W KR2012002496 W KR 2012002496W WO 2012138102 A2 WO2012138102 A2 WO 2012138102A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- dlk1
- set out
- heavy chain
- light chain
- Prior art date
Links
- 239000008194 pharmaceutical composition Substances 0.000 title claims abstract description 26
- 101150102995 dlk-1 gene Proteins 0.000 title description 3
- 230000027455 binding Effects 0.000 claims abstract description 125
- 239000012634 fragment Substances 0.000 claims abstract description 66
- 239000000427 antigen Substances 0.000 claims abstract description 52
- 108091007433 antigens Proteins 0.000 claims abstract description 52
- 102000036639 antigens Human genes 0.000 claims abstract description 52
- 238000000034 method Methods 0.000 claims abstract description 51
- 239000013598 vector Substances 0.000 claims abstract description 41
- 102000040430 polynucleotide Human genes 0.000 claims abstract description 23
- 108091033319 polynucleotide Proteins 0.000 claims abstract description 23
- 239000002157 polynucleotide Substances 0.000 claims abstract description 23
- 201000010099 disease Diseases 0.000 claims abstract description 13
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 13
- 208000026278 immune system disease Diseases 0.000 claims abstract description 9
- 208000019423 liver disease Diseases 0.000 claims abstract description 9
- 208000030159 metabolic disease Diseases 0.000 claims abstract description 9
- 201000007270 liver cancer Diseases 0.000 claims abstract description 7
- 208000014018 liver neoplasm Diseases 0.000 claims abstract description 7
- 238000009007 Diagnostic Kit Methods 0.000 claims abstract description 5
- 108010057453 activin receptor type II-B Proteins 0.000 claims abstract 5
- 102100027647 Activin receptor type-2B Human genes 0.000 claims description 111
- 101710191689 Activin receptor type-2B Proteins 0.000 claims description 98
- 206010028980 Neoplasm Diseases 0.000 claims description 58
- 201000011510 cancer Diseases 0.000 claims description 48
- 102100035360 Cerebellar degeneration-related antigen 1 Human genes 0.000 claims description 42
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 39
- 206010027476 Metastases Diseases 0.000 claims description 30
- 230000009401 metastasis Effects 0.000 claims description 30
- 108010059616 Activins Proteins 0.000 claims description 24
- 239000000488 activin Substances 0.000 claims description 24
- 206010061902 Pancreatic neoplasm Diseases 0.000 claims description 22
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 claims description 22
- 201000002528 pancreatic cancer Diseases 0.000 claims description 22
- 208000008443 pancreatic carcinoma Diseases 0.000 claims description 22
- 125000003275 alpha amino acid group Chemical group 0.000 claims description 20
- 230000002401 inhibitory effect Effects 0.000 claims description 20
- 238000003018 immunoassay Methods 0.000 claims description 14
- 102000004190 Enzymes Human genes 0.000 claims description 13
- 108090000790 Enzymes Proteins 0.000 claims description 13
- 239000000203 mixture Substances 0.000 claims description 13
- 206010021143 Hypoxia Diseases 0.000 claims description 12
- 108020001507 fusion proteins Proteins 0.000 claims description 8
- 102000037865 fusion proteins Human genes 0.000 claims description 8
- 208000002495 Uterine Neoplasms Diseases 0.000 claims description 7
- 206010046766 uterine cancer Diseases 0.000 claims description 7
- 238000012258 culturing Methods 0.000 claims description 6
- 208000008839 Kidney Neoplasms Diseases 0.000 claims description 5
- 206010038389 Renal cancer Diseases 0.000 claims description 5
- 208000016097 disease of metabolism Diseases 0.000 claims description 5
- 201000010982 kidney cancer Diseases 0.000 claims description 5
- 230000001146 hypoxic effect Effects 0.000 claims description 4
- 230000008595 infiltration Effects 0.000 claims description 4
- 238000001764 infiltration Methods 0.000 claims description 4
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims description 3
- 206010006187 Breast cancer Diseases 0.000 claims description 3
- 208000026310 Breast neoplasm Diseases 0.000 claims description 3
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 3
- 208000000453 Skin Neoplasms Diseases 0.000 claims description 3
- 208000005718 Stomach Neoplasms Diseases 0.000 claims description 3
- 206010017758 gastric cancer Diseases 0.000 claims description 3
- 230000004068 intracellular signaling Effects 0.000 claims description 3
- 201000005202 lung cancer Diseases 0.000 claims description 3
- 208000020816 lung neoplasm Diseases 0.000 claims description 3
- 201000000849 skin cancer Diseases 0.000 claims description 3
- 201000011549 stomach cancer Diseases 0.000 claims description 3
- 230000001131 transforming effect Effects 0.000 claims description 3
- 208000007788 Acute Liver Failure Diseases 0.000 claims description 2
- 206010000804 Acute hepatic failure Diseases 0.000 claims description 2
- 208000023275 Autoimmune disease Diseases 0.000 claims description 2
- 206010008909 Chronic Hepatitis Diseases 0.000 claims description 2
- 206010009208 Cirrhosis alcoholic Diseases 0.000 claims description 2
- 206010009944 Colon cancer Diseases 0.000 claims description 2
- 208000004930 Fatty Liver Diseases 0.000 claims description 2
- 206010019708 Hepatic steatosis Diseases 0.000 claims description 2
- 208000008589 Obesity Diseases 0.000 claims description 2
- 206010033128 Ovarian cancer Diseases 0.000 claims description 2
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 2
- 231100000836 acute liver failure Toxicity 0.000 claims description 2
- 208000010002 alcoholic liver cirrhosis Diseases 0.000 claims description 2
- 208000029742 colonic neoplasm Diseases 0.000 claims description 2
- 206010012601 diabetes mellitus Diseases 0.000 claims description 2
- 208000010706 fatty liver disease Diseases 0.000 claims description 2
- 208000006454 hepatitis Diseases 0.000 claims description 2
- 235000020824 obesity Nutrition 0.000 claims description 2
- 206010039073 rheumatoid arthritis Diseases 0.000 claims description 2
- 238000003118 sandwich ELISA Methods 0.000 claims description 2
- 231100000240 steatosis hepatitis Toxicity 0.000 claims description 2
- 102000005606 Activins Human genes 0.000 claims 2
- 238000002965 ELISA Methods 0.000 claims 1
- 230000019491 signal transduction Effects 0.000 abstract description 7
- 230000002265 prevention Effects 0.000 abstract description 5
- 238000004519 manufacturing process Methods 0.000 abstract description 4
- 101000928535 Homo sapiens Protein delta homolog 1 Proteins 0.000 abstract 3
- 102100036467 Protein delta homolog 1 Human genes 0.000 abstract 3
- 230000000259 anti-tumor effect Effects 0.000 abstract 1
- 210000004027 cell Anatomy 0.000 description 149
- 230000000694 effects Effects 0.000 description 44
- 101100112922 Candida albicans CDR3 gene Proteins 0.000 description 27
- 108090000623 proteins and genes Proteins 0.000 description 27
- 102100035361 Cerebellar degeneration-related protein 2 Human genes 0.000 description 26
- 101000737793 Homo sapiens Cerebellar degeneration-related antigen 1 Proteins 0.000 description 26
- 101000737796 Homo sapiens Cerebellar degeneration-related protein 2 Proteins 0.000 description 26
- 230000011664 signaling Effects 0.000 description 24
- 102100026818 Inhibin beta E chain Human genes 0.000 description 22
- 108010023082 activin A Proteins 0.000 description 18
- 229920001213 Polysorbate 20 Polymers 0.000 description 15
- 238000002474 experimental method Methods 0.000 description 15
- 230000014509 gene expression Effects 0.000 description 15
- 239000002609 medium Substances 0.000 description 15
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 15
- 108020004414 DNA Proteins 0.000 description 14
- 102000053602 DNA Human genes 0.000 description 14
- 230000001965 increasing effect Effects 0.000 description 14
- 229920001817 Agar Polymers 0.000 description 13
- 101000937269 Homo sapiens Activin receptor type-2B Proteins 0.000 description 13
- 239000008272 agar Substances 0.000 description 13
- 239000012679 serum free medium Substances 0.000 description 13
- 238000001262 western blot Methods 0.000 description 13
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 12
- 102000004169 proteins and genes Human genes 0.000 description 12
- 230000002829 reductive effect Effects 0.000 description 12
- 238000004458 analytical method Methods 0.000 description 11
- 238000006243 chemical reaction Methods 0.000 description 10
- 230000012010 growth Effects 0.000 description 10
- 238000005406 washing Methods 0.000 description 10
- 230000001093 anti-cancer Effects 0.000 description 9
- 239000003446 ligand Substances 0.000 description 9
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 9
- 239000000243 solution Substances 0.000 description 9
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 8
- 238000010171 animal model Methods 0.000 description 8
- 239000000872 buffer Substances 0.000 description 8
- 230000003247 decreasing effect Effects 0.000 description 8
- 230000007954 hypoxia Effects 0.000 description 8
- 238000002198 surface plasmon resonance spectroscopy Methods 0.000 description 8
- 238000002054 transplantation Methods 0.000 description 8
- 108010076504 Protein Sorting Signals Proteins 0.000 description 7
- 238000012790 confirmation Methods 0.000 description 7
- 238000004091 panning Methods 0.000 description 7
- 238000002360 preparation method Methods 0.000 description 7
- 102000005962 receptors Human genes 0.000 description 7
- 108020003175 receptors Proteins 0.000 description 7
- 210000004102 animal cell Anatomy 0.000 description 6
- 239000011248 coating agent Substances 0.000 description 6
- 238000000576 coating method Methods 0.000 description 6
- UQLDLKMNUJERMK-UHFFFAOYSA-L di(octadecanoyloxy)lead Chemical compound [Pb+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O UQLDLKMNUJERMK-UHFFFAOYSA-L 0.000 description 6
- 238000011065 in-situ storage Methods 0.000 description 6
- 230000005764 inhibitory process Effects 0.000 description 6
- 239000012528 membrane Substances 0.000 description 6
- 230000002441 reversible effect Effects 0.000 description 6
- 239000011780 sodium chloride Substances 0.000 description 6
- 239000006228 supernatant Substances 0.000 description 6
- 238000001890 transfection Methods 0.000 description 6
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 5
- 241001465754 Metazoa Species 0.000 description 5
- 230000002860 competitive effect Effects 0.000 description 5
- 210000004748 cultured cell Anatomy 0.000 description 5
- 238000001035 drying Methods 0.000 description 5
- 238000000684 flow cytometry Methods 0.000 description 5
- 239000008103 glucose Substances 0.000 description 5
- 238000011534 incubation Methods 0.000 description 5
- 230000007246 mechanism Effects 0.000 description 5
- 238000002823 phage display Methods 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 239000000725 suspension Substances 0.000 description 5
- 230000029663 wound healing Effects 0.000 description 5
- 108010047041 Complementarity Determining Regions Proteins 0.000 description 4
- 239000012979 RPMI medium Substances 0.000 description 4
- 239000006180 TBST buffer Substances 0.000 description 4
- 206010052428 Wound Diseases 0.000 description 4
- 208000027418 Wounds and injury Diseases 0.000 description 4
- 238000003556 assay Methods 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 229910002092 carbon dioxide Inorganic materials 0.000 description 4
- 238000005119 centrifugation Methods 0.000 description 4
- 210000000349 chromosome Anatomy 0.000 description 4
- 230000007423 decrease Effects 0.000 description 4
- 239000012153 distilled water Substances 0.000 description 4
- 239000000839 emulsion Substances 0.000 description 4
- 238000011503 in vivo imaging Methods 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 230000001404 mediated effect Effects 0.000 description 4
- 230000002018 overexpression Effects 0.000 description 4
- 210000000496 pancreas Anatomy 0.000 description 4
- 108090000765 processed proteins & peptides Proteins 0.000 description 4
- 230000035755 proliferation Effects 0.000 description 4
- 239000002096 quantum dot Substances 0.000 description 4
- 238000000926 separation method Methods 0.000 description 4
- 235000020183 skimmed milk Nutrition 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- 238000012795 verification Methods 0.000 description 4
- OZFAFGSSMRRTDW-UHFFFAOYSA-N (2,4-dichlorophenyl) benzenesulfonate Chemical compound ClC1=CC(Cl)=CC=C1OS(=O)(=O)C1=CC=CC=C1 OZFAFGSSMRRTDW-UHFFFAOYSA-N 0.000 description 3
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 3
- 208000003174 Brain Neoplasms Diseases 0.000 description 3
- 108091026890 Coding region Proteins 0.000 description 3
- 229920000742 Cotton Polymers 0.000 description 3
- 239000012591 Dulbecco’s Phosphate Buffered Saline Substances 0.000 description 3
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 239000012083 RIPA buffer Substances 0.000 description 3
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 3
- 235000014680 Saccharomyces cerevisiae Nutrition 0.000 description 3
- 108020004459 Small interfering RNA Proteins 0.000 description 3
- 102000004142 Trypsin Human genes 0.000 description 3
- 108090000631 Trypsin Proteins 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 210000003719 b-lymphocyte Anatomy 0.000 description 3
- 230000000903 blocking effect Effects 0.000 description 3
- 239000001569 carbon dioxide Substances 0.000 description 3
- 239000002975 chemoattractant Substances 0.000 description 3
- 238000010276 construction Methods 0.000 description 3
- 238000002224 dissection Methods 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 238000004520 electroporation Methods 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 238000009472 formulation Methods 0.000 description 3
- BRZYSWJRSDMWLG-CAXSIQPQSA-N geneticin Natural products O1C[C@@](O)(C)[C@H](NC)[C@@H](O)[C@H]1O[C@@H]1[C@@H](O)[C@H](O[C@@H]2[C@@H]([C@@H](O)[C@H](O)[C@@H](C(C)O)O2)N)[C@@H](N)C[C@H]1N BRZYSWJRSDMWLG-CAXSIQPQSA-N 0.000 description 3
- 230000009545 invasion Effects 0.000 description 3
- BPHPUYQFMNQIOC-NXRLNHOXSA-N isopropyl beta-D-thiogalactopyranoside Chemical compound CC(C)S[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O BPHPUYQFMNQIOC-NXRLNHOXSA-N 0.000 description 3
- 239000006166 lysate Substances 0.000 description 3
- 238000010232 migration assay Methods 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 239000008188 pellet Substances 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 239000011148 porous material Substances 0.000 description 3
- 229920002477 rna polymer Polymers 0.000 description 3
- 238000013207 serial dilution Methods 0.000 description 3
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000000829 suppository Substances 0.000 description 3
- 238000001356 surgical procedure Methods 0.000 description 3
- 239000012588 trypsin Substances 0.000 description 3
- PMUNIMVZCACZBB-UHFFFAOYSA-N 2-hydroxyethylazanium;chloride Chemical compound Cl.NCCO PMUNIMVZCACZBB-UHFFFAOYSA-N 0.000 description 2
- 102000043279 ADAM17 Human genes 0.000 description 2
- 108091007505 ADAM17 Proteins 0.000 description 2
- 102100022900 Actin, cytoplasmic 1 Human genes 0.000 description 2
- 108010085238 Actins Proteins 0.000 description 2
- 108010052946 Activin Receptors Proteins 0.000 description 2
- 102000018918 Activin Receptors Human genes 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- 101710132601 Capsid protein Proteins 0.000 description 2
- 101710094648 Coat protein Proteins 0.000 description 2
- 108020004705 Codon Proteins 0.000 description 2
- 241000724791 Filamentous phage Species 0.000 description 2
- 102100021181 Golgi phosphoprotein 3 Human genes 0.000 description 2
- 206010019909 Hernia Diseases 0.000 description 2
- 229920000209 Hexadimethrine bromide Polymers 0.000 description 2
- 108060003951 Immunoglobulin Proteins 0.000 description 2
- 108010054477 Immunoglobulin Fab Fragments Proteins 0.000 description 2
- 102000001706 Immunoglobulin Fab Fragments Human genes 0.000 description 2
- 101710125418 Major capsid protein Proteins 0.000 description 2
- 102100030608 Mothers against decapentaplegic homolog 7 Human genes 0.000 description 2
- 241000699660 Mus musculus Species 0.000 description 2
- 101710141454 Nucleoprotein Proteins 0.000 description 2
- 241001494479 Pecora Species 0.000 description 2
- 101710083689 Probable capsid protein Proteins 0.000 description 2
- 229940124158 Protease/peptidase inhibitor Drugs 0.000 description 2
- 108091027981 Response element Proteins 0.000 description 2
- 101700026522 SMAD7 Proteins 0.000 description 2
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 2
- 206010041660 Splenomegaly Diseases 0.000 description 2
- 101710120037 Toxin CcdB Proteins 0.000 description 2
- 102100023935 Transmembrane glycoprotein NMB Human genes 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 230000000735 allogeneic effect Effects 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000003125 aqueous solvent Substances 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- 235000014121 butter Nutrition 0.000 description 2
- 230000036952 cancer formation Effects 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 239000013592 cell lysate Substances 0.000 description 2
- 230000012292 cell migration Effects 0.000 description 2
- 230000005754 cellular signaling Effects 0.000 description 2
- 238000003776 cleavage reaction Methods 0.000 description 2
- 238000000749 co-immunoprecipitation Methods 0.000 description 2
- 230000009137 competitive binding Effects 0.000 description 2
- 230000034994 death Effects 0.000 description 2
- 231100000517 death Toxicity 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 238000010494 dissociation reaction Methods 0.000 description 2
- 230000005593 dissociations Effects 0.000 description 2
- 239000003623 enhancer Substances 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 210000004408 hybridoma Anatomy 0.000 description 2
- 102000018358 immunoglobulin Human genes 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- 238000003780 insertion Methods 0.000 description 2
- 230000037431 insertion Effects 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical group N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 230000003834 intracellular effect Effects 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- 238000013508 migration Methods 0.000 description 2
- 102000039446 nucleic acids Human genes 0.000 description 2
- 108020004707 nucleic acids Proteins 0.000 description 2
- 150000007523 nucleic acids Chemical class 0.000 description 2
- 239000002773 nucleotide Substances 0.000 description 2
- 125000003729 nucleotide group Chemical group 0.000 description 2
- 238000011580 nude mouse model Methods 0.000 description 2
- 239000000137 peptide hydrolase inhibitor Substances 0.000 description 2
- 239000006187 pill Substances 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 229920001184 polypeptide Polymers 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 102000004196 processed proteins & peptides Human genes 0.000 description 2
- 238000001959 radiotherapy Methods 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 238000003757 reverse transcription PCR Methods 0.000 description 2
- 239000012723 sample buffer Substances 0.000 description 2
- 230000007017 scission Effects 0.000 description 2
- 230000028327 secretion Effects 0.000 description 2
- 238000012163 sequencing technique Methods 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 239000001632 sodium acetate Substances 0.000 description 2
- 235000017281 sodium acetate Nutrition 0.000 description 2
- RPENMORRBUTCPR-UHFFFAOYSA-M sodium;1-hydroxy-2,5-dioxopyrrolidine-3-sulfonate Chemical compound [Na+].ON1C(=O)CC(S([O-])(=O)=O)C1=O RPENMORRBUTCPR-UHFFFAOYSA-M 0.000 description 2
- 230000009870 specific binding Effects 0.000 description 2
- 210000002784 stomach Anatomy 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 230000002195 synergetic effect Effects 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 230000008685 targeting Effects 0.000 description 2
- 108091007466 transmembrane glycoproteins Proteins 0.000 description 2
- 238000010200 validation analysis Methods 0.000 description 2
- 239000000080 wetting agent Substances 0.000 description 2
- XXZCIYUJYUESMD-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]-3-(morpholin-4-ylmethyl)pyrazol-1-yl]-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C=1C(=NN(C=1)CC(=O)N1CC2=C(CC1)NN=N2)CN1CCOCC1 XXZCIYUJYUESMD-UHFFFAOYSA-N 0.000 description 1
- BFSVOASYOCHEOV-UHFFFAOYSA-N 2-diethylaminoethanol Chemical compound CCN(CC)CCO BFSVOASYOCHEOV-UHFFFAOYSA-N 0.000 description 1
- 101150031865 ACVR2B gene Proteins 0.000 description 1
- 102100034111 Activin receptor type-1 Human genes 0.000 description 1
- 102100034134 Activin receptor type-1B Human genes 0.000 description 1
- 102000000132 Alpha tubulin Human genes 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 241000593245 Bionia Species 0.000 description 1
- 102100022525 Bone morphogenetic protein 6 Human genes 0.000 description 1
- 102100022544 Bone morphogenetic protein 7 Human genes 0.000 description 1
- 101100283604 Caenorhabditis elegans pigk-1 gene Proteins 0.000 description 1
- 208000005623 Carcinogenesis Diseases 0.000 description 1
- 208000017667 Chronic Disease Diseases 0.000 description 1
- 108010014303 DNA-directed DNA polymerase Proteins 0.000 description 1
- 102000016928 DNA-directed DNA polymerase Human genes 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 102100038132 Endogenous retrovirus group K member 6 Pro protein Human genes 0.000 description 1
- 101710091045 Envelope protein Proteins 0.000 description 1
- 102400001368 Epidermal growth factor Human genes 0.000 description 1
- 101800003838 Epidermal growth factor Proteins 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 208000000461 Esophageal Neoplasms Diseases 0.000 description 1
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 208000032612 Glial tumor Diseases 0.000 description 1
- 206010018338 Glioma Diseases 0.000 description 1
- 102100040898 Growth/differentiation factor 11 Human genes 0.000 description 1
- 102100035379 Growth/differentiation factor 5 Human genes 0.000 description 1
- 241000238631 Hexapoda Species 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000799140 Homo sapiens Activin receptor type-1 Proteins 0.000 description 1
- 101000799189 Homo sapiens Activin receptor type-1B Proteins 0.000 description 1
- 101000899390 Homo sapiens Bone morphogenetic protein 6 Proteins 0.000 description 1
- 101000899361 Homo sapiens Bone morphogenetic protein 7 Proteins 0.000 description 1
- 101000893545 Homo sapiens Growth/differentiation factor 11 Proteins 0.000 description 1
- 101001023988 Homo sapiens Growth/differentiation factor 5 Proteins 0.000 description 1
- 108010067060 Immunoglobulin Variable Region Proteins 0.000 description 1
- 102000017727 Immunoglobulin Variable Region Human genes 0.000 description 1
- 108020005350 Initiator Codon Proteins 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 102000006992 Interferon-alpha Human genes 0.000 description 1
- 108010047761 Interferon-alpha Proteins 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 102000003960 Ligases Human genes 0.000 description 1
- 108090000364 Ligases Proteins 0.000 description 1
- 108060001084 Luciferase Proteins 0.000 description 1
- 102000019149 MAP kinase activity proteins Human genes 0.000 description 1
- 108040008097 MAP kinase activity proteins Proteins 0.000 description 1
- 102000018697 Membrane Proteins Human genes 0.000 description 1
- 108010052285 Membrane Proteins Proteins 0.000 description 1
- 102100025751 Mothers against decapentaplegic homolog 2 Human genes 0.000 description 1
- 101710143123 Mothers against decapentaplegic homolog 2 Proteins 0.000 description 1
- 102100025725 Mothers against decapentaplegic homolog 4 Human genes 0.000 description 1
- 101710143112 Mothers against decapentaplegic homolog 4 Proteins 0.000 description 1
- 208000003445 Mouth Neoplasms Diseases 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 230000004988 N-glycosylation Effects 0.000 description 1
- 108091028043 Nucleic acid sequence Proteins 0.000 description 1
- 230000004989 O-glycosylation Effects 0.000 description 1
- 206010030155 Oesophageal carcinoma Diseases 0.000 description 1
- 108700020796 Oncogene Proteins 0.000 description 1
- 239000008118 PEG 6000 Substances 0.000 description 1
- 239000002033 PVDF binder Substances 0.000 description 1
- 108010002747 Pfu DNA polymerase Proteins 0.000 description 1
- 229940122907 Phosphatase inhibitor Drugs 0.000 description 1
- 229920002584 Polyethylene Glycol 6000 Polymers 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 101710188315 Protein X Proteins 0.000 description 1
- 208000035415 Reinfection Diseases 0.000 description 1
- 108700008625 Reporter Genes Proteins 0.000 description 1
- 101150014136 SUC2 gene Proteins 0.000 description 1
- 238000012300 Sequence Analysis Methods 0.000 description 1
- 102000011842 Serrate-Jagged Proteins Human genes 0.000 description 1
- 108010036039 Serrate-Jagged Proteins Proteins 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- 108020005038 Terminator Codon Proteins 0.000 description 1
- 244000299461 Theobroma cacao Species 0.000 description 1
- 235000005764 Theobroma cacao ssp. cacao Nutrition 0.000 description 1
- 235000005767 Theobroma cacao ssp. sphaerocarpum Nutrition 0.000 description 1
- 108090000704 Tubulin Proteins 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 108010023079 activin B Proteins 0.000 description 1
- 230000006978 adaptation Effects 0.000 description 1
- 239000011543 agarose gel Substances 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 239000013602 bacteriophage vector Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 235000001046 cacaotero Nutrition 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 229940041514 candida albicans extract Drugs 0.000 description 1
- 231100000504 carcinogenesis Toxicity 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 230000007910 cell fusion Effects 0.000 description 1
- 230000010261 cell growth Effects 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 238000002659 cell therapy Methods 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 239000012829 chemotherapy agent Substances 0.000 description 1
- 229960005091 chloramphenicol Drugs 0.000 description 1
- WIIZWVCIJKGZOK-RKDXNWHRSA-N chloramphenicol Chemical compound ClC(Cl)C(=O)N[C@H](CO)[C@H](O)C1=CC=C([N+]([O-])=O)C=C1 WIIZWVCIJKGZOK-RKDXNWHRSA-N 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 239000013601 cosmid vector Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 230000001934 delay Effects 0.000 description 1
- 238000012217 deletion Methods 0.000 description 1
- 230000037430 deletion Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 210000000188 diaphragm Anatomy 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 238000002651 drug therapy Methods 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 230000003028 elevating effect Effects 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 201000004101 esophageal cancer Diseases 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- -1 ethyl oleate Chemical class 0.000 description 1
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 1
- 229940093471 ethyl oleate Drugs 0.000 description 1
- 210000003527 eukaryotic cell Anatomy 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- MHMNJMPURVTYEJ-UHFFFAOYSA-N fluorescein-5-isothiocyanate Chemical compound O1C(=O)C2=CC(N=C=S)=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 MHMNJMPURVTYEJ-UHFFFAOYSA-N 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 238000010353 genetic engineering Methods 0.000 description 1
- 210000004602 germ cell Anatomy 0.000 description 1
- 230000009036 growth inhibition Effects 0.000 description 1
- 230000010005 growth-factor like effect Effects 0.000 description 1
- 238000003306 harvesting Methods 0.000 description 1
- 201000005787 hematologic cancer Diseases 0.000 description 1
- 208000024200 hematopoietic and lymphoid system neoplasm Diseases 0.000 description 1
- 210000003494 hepatocyte Anatomy 0.000 description 1
- 238000001794 hormone therapy Methods 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 230000016784 immunoglobulin production Effects 0.000 description 1
- 229940072221 immunoglobulins Drugs 0.000 description 1
- 239000000367 immunologic factor Substances 0.000 description 1
- 238000001114 immunoprecipitation Methods 0.000 description 1
- 238000012744 immunostaining Methods 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000001427 incoherent neutron scattering Methods 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 108010067471 inhibin A Proteins 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 239000007928 intraperitoneal injection Substances 0.000 description 1
- 229930027917 kanamycin Natural products 0.000 description 1
- 229960000318 kanamycin Drugs 0.000 description 1
- SBUJHOSQTJFQJX-NOAMYHISSA-N kanamycin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CN)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O2)O)[C@H](N)C[C@@H]1N SBUJHOSQTJFQJX-NOAMYHISSA-N 0.000 description 1
- 229930182823 kanamycin A Natural products 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- VMPHSYLJUKZBJJ-UHFFFAOYSA-N lauric acid triglyceride Natural products CCCCCCCCCCCC(=O)OCC(OC(=O)CCCCCCCCCCC)COC(=O)CCCCCCCCCCC VMPHSYLJUKZBJJ-UHFFFAOYSA-N 0.000 description 1
- 208000012987 lip and oral cavity carcinoma Diseases 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- 239000006193 liquid solution Substances 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 230000033001 locomotion Effects 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 230000002934 lysing effect Effects 0.000 description 1
- 229960003511 macrogol Drugs 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 108010082117 matrigel Proteins 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 238000000520 microinjection Methods 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 239000012457 nonaqueous media Substances 0.000 description 1
- 230000009871 nonspecific binding Effects 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000013600 plasmid vector Substances 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 230000008488 polyadenylation Effects 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000002685 pulmonary effect Effects 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 210000003705 ribosome Anatomy 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 230000008054 signal transmission Effects 0.000 description 1
- 230000007727 signaling mechanism Effects 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 230000005062 synaptic transmission Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 230000005030 transcription termination Effects 0.000 description 1
- 230000002103 transcriptional effect Effects 0.000 description 1
- 238000010361 transduction Methods 0.000 description 1
- 230000026683 transduction Effects 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000009261 transgenic effect Effects 0.000 description 1
- 238000003146 transient transfection Methods 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 239000012137 tryptone Substances 0.000 description 1
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 239000013603 viral vector Substances 0.000 description 1
- 210000005253 yeast cell Anatomy 0.000 description 1
- 239000012138 yeast extract Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/475—Growth factors; Growth regulators
- C07K14/485—Epidermal growth factor [EGF], i.e. urogastrone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
Definitions
- the present invention provides a fragment comprising a human antibody or antigen-binding site thereof that specifically binds to the extracellular domain water-soluble domain of a delta-like 1 homolog (DLK1), a polynucleotide encoding the human antibody or fragment thereof, the polynucleotide Recombinant vector comprising a, transformants transformed with the recombinant vector, a method for producing the human antibody or fragment thereof, pharmaceutical composition and diagnostic kit for preventing or treating a disease comprising the human antibody or fragment thereof It is about.
- DLK1 delta-like 1 homolog
- Cancer is characterized by "uncontrolled cell growth,” which results in the formation of cell masses called tumors that penetrate into surrounding tissues and, in severe cases, metastasize to other organs in the body. These cancers are intractable chronic diseases that, even if treated with surgery, radiation, and chemotherapy, in many cases fail to cure the underlying condition and cause pain to the patient and ultimately lead to death.
- Cancer varies from country to country, but developed countries and Korea account for more than 20% of all deaths. Cancer is classified into blood cancer and solid cancer, and it occurs in almost every part of the body such as lung cancer, stomach cancer, breast cancer, oral cancer, liver cancer, uterine cancer, esophageal cancer, and skin cancer.
- chemotherapy agents except surgery or radiation therapy, are collectively called anticancer agents, and most of them show anticancer activity by inhibiting the synthesis of nucleic acids.
- the delta-like 1 homolog (DLK1) belonging to the notch / delta / serrate family is a transmembrane glycoprotein encoded by the dlk1 gene located on chromosome 14q32. It consists of.
- the protein is divided into 280 extracellular regions, 24 membrane transmembrane regions, and 56 intracellular regions, and has six epidermal growth factor like repeat domains outside the double membrane, and three It has N-glycosylation and seven O-glycosylation sites.
- DLK1 is a membrane protein, it is well known as a protein that sheds the outer part of the cell membrane by a tumor necrosis factor alpha converting enzyme (TACE) and functions separately.
- TACE tumor necrosis factor alpha converting enzyme
- DLK1 and cancer studies on the relationship between DLK1 and cancer include overexpression of DLK1 expression in brain cancer cells (glioma), and overexpression of cDNA of DLK1 in brain cancer cells increases the proliferation of brain cancer cells, leading to increased migration.
- glioma brain cancer cells
- cDNA of DLK1 in brain cancer cells increases the proliferation of brain cancer cells, leading to increased migration.
- the expression of DLK1 in liver cancer is higher than that of normal hepatocytes, and the tumor size is reduced when the expression of DLK1 is reduced by siRNA experiment. It has been reported to decrease (Huang J et al., Carcinogenesis, 28 (5): 1094-1103, 2007).
- the inventors of the present invention first identified that the DLK1-Fc fusion protein conjugated with the extracellular domain water-soluble domain of DLK1 and the Fc domain of the IgG antibody in the Republic of Korea Patent No. 10-0982170 can exhibit an anticancer effect. .
- the anticancer action of the extracellular domain soluble domain of DLK1 is caused.
- the present inventors have made diligent efforts to elucidate the anticancer mechanism of the extracellular domain soluble domain of DLK1.
- the extracellular domain soluble domain of DLK1 binds to activin type II B competitively with activin.
- the present invention was found to exhibit anticancer effects by blocking activin signaling, thereby completing the present invention.
- human antibodies that specifically bind to the extracellular domain soluble domain of DLK1 not only inhibit cancer cell metastasis, but also significantly increase the binding ability of DLK1 to the activin receptor type 2B of the extracellular domain soluble domain. It was confirmed that the present invention was completed, and the present invention was completed.
- Another object of the present invention is to provide a polynucleotide encoding a fragment comprising the human antibody or antigen-binding site thereof.
- Another object of the present invention is to provide a recombinant vector comprising the polynucleotide.
- Still another object of the present invention is to provide a transformant transformed with the recombinant vector.
- Still another object of the present invention is to provide a method for preparing a fragment comprising the human antibody or antigen-binding site thereof.
- Another object of the present invention to provide a pharmaceutical composition for preventing or treating cancer, comprising a fragment comprising the human antibody or antigen-binding site thereof.
- the present invention provides a fragment comprising a human antibody or antigen-binding site thereof that specifically binds to the extracellular domain water-soluble domain of DLK1 (delta-like 1 homolog).
- delta-like 1 homolog refers to a transmembrane glycoprotein consisting of 383 amino acids encoded in the dlk1 gene located on chromosome 14q32.
- extracellular domain water-soluble domain of DLK1 refers to the water-soluble domain of the extracellular domain of DLK1 protein divided into extracellular, transmembrane and intracellular regions, and anticancer of the extracellular domain of the DLK1 extracellular domain. The effect was first identified by the inventors.
- extracellular domain water-soluble domain of DLK1 may be expressed in combination with water-soluble DLK1.
- the water-soluble DLK1 of the present invention may be composed of 200 to 300 amino acids having water-soluble DLK1 activity, more preferably may be composed of the amino acid sequence described in SEQ ID NO: 1, the amino acid sequence showing water-soluble DLK1 activity is not limited May be included.
- human antibody refers to a molecule derived from human immunoglobulin, in which all of the amino acid sequences constituting the antibody including complementarity determining regions and structural regions are composed of human immunoglobulins.
- one antibody molecule has two heavy chains and two light chains, each of which has a variable region at its N-terminus.
- Each variable region consists of three complementarity determining regions (CDRs) and four framework regions (FRs), which determine the antigen binding specificity of the antibody and determine the structure of the variable region.
- CDRs complementarity determining regions
- FRs framework regions
- the fragment comprising the human antibody or antigen-binding site thereof of the present invention has the activity of specifically binding to water-soluble DLK1.
- the fragment comprising the human antibody or antigen-binding site thereof of the present invention may have an activity of specifically binding water-soluble DLK1, thereby ultimately increasing the binding capacity of the water-soluble DLK1 to activin receptor type 2B.
- fragments comprising the human antibody or antigen-binding site thereof of the present invention can competitively bind DLK1 to activin receptor type 2B by elevating the binding ability of the extracellular domain soluble domain of DLK1 to activin receptor type 2B. Signaling can be inhibited by blocking the binding of other ligands.
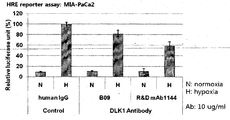
- the fragment comprising the human antibody or antigen-binding site thereof of the present invention can inhibit intracellular signaling in a hypoxic state.
- the human antibody of the present invention specifically binds to the water-soluble DLK1 by confirming that the B09 antibody is typically capable of binding to the water-soluble DLK1 of the human antibodies of the present invention ( 21, 22 and 27), when the human antibody of the present invention was treated with water-soluble DLK1 it was confirmed that the binding capacity of water-soluble DLK1 to activin receptor type 2B significantly increased (Figs. 25, 27 and 29).
- the human antibody of the present invention can react the cells in the hypoxic state It was confirmed that it can be suppressed (FIG. 28).
- the fragment comprising the human antibody or antigen-binding portion thereof of the present invention is a part of the heavy chain variable region, SEQ ID NO: 2 (SYAMN), SEQ ID NO: 8 (DYAIH), SEQ ID NO: 14 (EHAMH), SEQ ID NO: 20 ( DYAMH), SEQ ID NO: 26 (LYGMS) and SEQ ID NO: 32 (DYYMS), any one heavy chain CDR1 sequence selected from the group consisting of; SEQ ID NO: 3 (TITATSGKTYYADSVKG), SEQ ID NO: 9 (WINPGSGNTKYSHNFEG), SEQ ID NO: 15 (GINWNSGKTGYADSVKG), SEQ ID NO: 21 (GISWNSGSIGYADSVKG), SEQ ID NO: 27 (SIPGSGTRTHYADSVKG), and SEQ ID NO: 33 (one of the groups selected from YISGSGITTYYADSVKG) CDR2 sequence; Or any one selected from the group consisting of SEQ ID NO: 4 (GESCSGG
- Human antibody comprising a heavy chain CDR3 sequence or a fragment comprising an antigen binding site thereof, and also as part of a light chain variable region, SEQ ID NO: 5 (TGTSSDIGRYNRVS), SEQ ID NO: 11 (QASQDISNYLN), SEQ ID NO: 17 (IGTSSNIGVGYDVH), Any one light chain CDR1 sequence selected from the group consisting of SEQ ID NO: 23 (RASQRISSWLA), SEQ ID NO: 29 (RASQSIRHYLA) and SEQ ID NO: 35 (RASQSILTYLN); Any one light chain selected from the group consisting of SEQ ID NO: 6 (DVTTRPS), SEQ ID NO: 12 (STSNLQS), SEQ ID NO: 18 (GNNNRPS), SEQ ID NO: 24 (SASTLHN), SEQ ID NO: 30 (GASSRAT), and SEQ ID NO: 36 (AASSLQR) CDR2 sequence; Or any one selected from the group consisting of SEQ ID NO: 7 (GSYAGS
- the fragment comprising the human antibody or antigen-binding portion thereof of the present invention comprises a heavy chain variable region, the heavy chain CDR1 of SEQ ID NO: 2, the heavy chain CDR2 of SEQ ID NO: 3, and the heavy chain CDR3 of SEQ ID NO: 4 Heavy chain variable region;
- a heavy chain variable region comprising a heavy chain CDR1 as set out in SEQ ID NO: 8, a heavy chain CDR2 as set out in SEQ ID NO: 9, and a heavy chain CDR3 as set out in SEQ ID NO: 10;
- a heavy chain variable region comprising a heavy chain CDR1 as set out in SEQ ID NO: 14, a heavy chain CDR2 as set out in SEQ ID NO: 15, and a heavy chain CDR3 as set out in SEQ ID NO: 16;
- a heavy chain variable region comprising a heavy chain CDR1 as set out in SEQ ID NO: 20, a heavy chain CDR2 as set out in SEQ ID NO: 21, and a heavy chain CDR3 as set out in SEQ ID
- a light chain variable region comprising a light chain CDR1 as set out in SEQ ID NO: 5, a light chain CDR2 as set out in SEQ ID NO: 6 and a light chain CDR3 as set out in SEQ ID NO: 7;
- a light chain variable region comprising a light chain CDR1 as set out in SEQ ID NO: 11, a light chain CDR2 as set out in SEQ ID NO: 12, and a light chain CDR3 as set out in SEQ ID NO: 13;
- a light chain variable region comprising a light chain CDR1 as set out in SEQ ID NO: 17, a light chain CDR2 as set out in SEQ ID NO: 18, and a light chain CDR3 as set out in SEQ ID NO: 19;
- a light chain variable region comprising a light chain CDR1 as set out in SEQ ID NO: 23, a light chain CDR2 as set out in SEQ ID NO: 24, and a light chain CDR3 as set out in SEQ ID NO: 25;
- a light chain variable region
- the fragment comprising the human antibody or antigen-binding portion thereof of the present invention is a heavy chain variable region sequence, SEQ ID NO: 38, SEQ ID NO: 40, SEQ ID NO: 42, SEQ ID NO: 44, SEQ ID NO: 46 or SEQ ID NO: 48 It may be a human antibody comprising the amino acid sequence described or a fragment comprising an antigen-binding portion thereof, and as the light chain variable region sequence, SEQ ID NO: 39, SEQ ID NO: 41, SEQ ID NO: 43, SEQ ID NO: 45, SEQ ID NO: 47 or SEQ ID NO: 49 It may be a human antibody or a fragment comprising an antigen-binding site thereof comprising the amino acid sequence described.
- the heavy and light chains may be used individually or together depending on the purpose, and a combination of a plurality of CDR sequences and light and heavy chains is possible according to conventional genetic engineering methods as desired by those skilled in the art.
- fragment comprising the antigen-binding site of a human antibody refers to a fragment having immunological activity of the human antibody molecule of the present invention that can achieve antigen (water-soluble DLK1) -antibody binding and possesses antigen-binding function. It means the piece being done.
- fragments include (i) a Fab fragment consisting of the variable region (VL) of the light chain and the variable region (VH) of the heavy chain, the constant region of the light chain (CL) and the first constant region of the heavy chain (CH1); (ii) a Fd fragment consisting of the VH and CH1 domains; (iii) a Fv fragment consisting of the VL and VH domains of a monoclonal antibody; (iv) dAb fragment consisting of the VH domain (Ward ES et al., Nature, 341: 544-546,1989); (v) isolated CDR regions; (vi) a F (ab ') 2 fragment that is a bivalent fragment comprising two linked Fab fragments; (vii) a single chain Fv molecule (scFv) bound by a peptide linker that binds the VH domain and the VL domain to form an antigen binding site; (viii) bispecific single-chain Fv dimers (PCT
- Human antibodies of the present invention can be easily prepared by known monoclonal antibody production techniques.
- methods for preparing monoclonal antibodies can be performed by preparing hybridomas using B lymphocytes obtained from immunized animals, or by using phage display technology, but are not limited thereto. It doesn't happen.
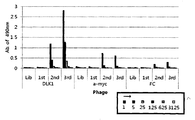
- the human antibody of the present invention may be a human antibody produced from phage display.
- the antibody library using phage display technology is a method of expressing an antibody on a phage surface by obtaining an antibody gene from B lymphocytes without producing hybridomas.
- Phage display technology can overcome many of the existing difficulties associated with generating monoclonal antibodies by B-cell immortalization.
- Typical phage display techniques of antibodies include the steps of: 1) inserting a sequence comprising an antibody variable region of a random sequence into a gene region corresponding to the coat protein p (or p) N-terminus of the phage; 2) expressing a portion of the native coat protein and a fusion protein of the antibody variable region encoded by the random sequence; 3) treating the antigen capable of binding with the antibody fusion protein library; 4) eluting antibody-phage particles bound to the antigen using low pH or binding competitive molecules; 5) amplifying the eluted phage in host cells; 6) repeating the method to obtain the desired amount; And 7) determining the sequence of the active antibody variable region from the DNA sequences of the phage clones selected by panning.
- Phage that can be used to build antibody libraries in this step include fd, M13, f1, If1, Ike, Zj / Z, Ff, Xf, Pf1 or Pf3 phage, for example as filamentous phage.
- the kind of phage that can be used in the present invention is not limited.
- examples of vectors that can be used for expression of heterologous genes on the surface of the filamentous phage include phage vectors such as fUSE5, fAFF1, fd-CAT1 or fdtetDOG or pHEN1, pComb3, pComb8, pKRIBB-Fab or pSEX. Phagemid vectors of, but are not limited thereto.
- helper phage that may be used to provide the wild type envelope protein required for successful reinfection of recombinant phage for amplification include, for example, M13K07 or VSCM13, but is not limited thereto.
- the present invention provides a polynucleotide encoding a fragment (hereinafter, fragment thereof) comprising a human antibody or antigen-binding portion thereof according to the present invention and a recombinant vector comprising the polynucleotide.
- the polynucleotide of the present invention is a polymer of nucleotides in which nucleotide monomers are long chained by covalent bonds, and are strands of DNA (deoxyribonucleic acid) or RNA (ribonucleic acid) having a predetermined length or more, and according to the present invention, Polynucleotides encoding an antibody or fragment thereof.
- the polynucleotide of the present invention may be a polynucleotide comprising a heavy chain variable region of SEQ ID NO: 50 and a light chain variable region of SEQ ID NO: 51.
- the polynucleotide encoding the human antibody or fragment thereof of the present invention is a human expressed from a coding region due to the degeneracy of the codon or in view of the codons preferred in the organism in which the human antibody or fragment thereof is to be expressed.
- Various modifications may be made to the coding region within the range of not changing the amino acid sequence of the antibody or fragment thereof, and various modifications or modifications may be made to the region except for the coding region without affecting the expression of the gene. Those skilled in the art will appreciate that such modified genes are also within the scope of the present invention.
- nucleic acid bases may be mutated by substitution, deletion, insertion, or a combination thereof, which are also included in the scope of the present invention.
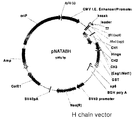
- the recombinant vector of the present invention is a means for introducing a DNA into a host cell to express a human antibody or fragment thereof of the present invention in a microorganism.
- expression control sequences such as promoters, terminators, enhancers, etc., sequences for membrane targeting or secretion, etc. may be appropriately selected and variously combined according to the purpose.
- Recombinant vectors of the invention include, but are not limited to, plasmid vectors, cosmid vectors, bacteriophage vectors, viral vectors, and the like.
- Suitable recombinant vectors include signal sequences or leader sequences for membrane targeting or secretion in addition to expression control elements such as promoters, operators, initiation codons, termination codons, polyadenylation signals and enhancers and can be prepared in various ways depending on the purpose.
- the promoter of the recombinant vector may be constitutive or inducible.
- As the signal sequence, MF ⁇ signal sequence, SUC2 signal sequence, etc. may be used when the host is a yeast, and insulin signal sequence, ⁇ -interferon signal sequence, antibody molecular signal sequence, etc.
- the recombinant vector may include a selection marker for selecting a host cell containing the vector, and may include the origin of replication in the case of a replicable recombinant vector.
- the present invention provides a method for producing a recombinant vector comprising a transformant transformed with a recombinant vector of the present invention and (a) a polynucleotide encoding a human antibody or fragment thereof of the present invention; (b) introducing the recombinant vector into a host cell and transforming the transformant with a transformant; And (c) provides a method for producing a human antibody or fragment thereof according to the present invention comprising the step of culturing the transformant.
- the recombinant vector of the present invention may be transformed into an appropriate host cell, for example, a yeast cell, an animal cell, or the like, followed by culturing the transformed host cell, thereby producing a large amount of the human antibody or fragment thereof of the present invention.
- an appropriate host cell for example, a yeast cell, an animal cell, or the like
- the term "transformation” refers to introducing a gene into a host cell so that the gene can be expressed in the host cell, and if the transformed gene can be expressed in the host cell, the insertion into the chromosome of the host cell or Anything located outside the chromosome is included without limitation.
- the gene also includes DNA and RNA as polynucleotides capable of encoding a polypeptide.
- the gene may be introduced in any form as long as it can be expressed by being introduced into a host cell.
- the gene may be introduced into a host cell in the form of an expression cassette, which is a polynucleotide construct containing all the elements necessary for its expression.
- the expression cassette typically includes a promoter, transcription termination signal, ribosomal binding site, and translation termination signal operably linked to the gene.
- the method of transforming by introducing a recombinant vector of the present invention into a host cell is a method known in the art, such as, but not limited to, a transgenic vector comprising the DNA of the present invention, transient transfection, Microinjection, transduction, cell fusion, calcium phosphate precipitation, liposome-mediated transfection, DEAE Dextran-mediated transfection, polybrene-mediated
- the cell can be transformed by introducing into a host cell by a known method such as polybrene-mediated transfection or electroporation.
- the host cell may be a eukaryotic cell derived from yeast, insect cells, plant cells, animal cells, such as Saccharomyces cerevisiae, preferably the animal cell may be an autologous or allogeneic animal cell. have. Transformants prepared by introducing into autologous or allogeneic animal cells may be used in cell therapies for administration to a subject to treat cancer.
- the method of culturing the transformant of the present invention may be appropriately selected and used in any method known in the art for producing the human antibody or fragment thereof of the present invention.
- the human antibody or fragment comprising the antigen-binding site thereof of the present invention specifically binds to the extracellular water soluble domain of DLK1, thereby activating the activin receptor type 2B of the extracellular domain water soluble domain of DLK1. It is useful for preventing or treating diseases related to the activin receptor type 2B signaling system by blocking the binding of other ligands capable of competitively binding DLK1 to activin receptor type 2B, thereby increasing the binding capacity. Can be used.
- Ligands that bind to activin receptor type 2B include activin (activin A, activin B and inhibin A) (Derynck, Zhang et al. 1998), nodal (Oh and Li 2002), BMP6 (Ebisawa, Tada et al. 1999), BMP7 (Yamashita, ten Dijke et al. 1995), GDF5 (Nishitoh, Ichijo et al. 1996), GDF11 (Oh, Yeo et al. 2002) and the like.
- activin activin A, activin B and inhibin A
- nodal Oh and Li 2002
- BMP6 Bisawa, Tada et al. 1999
- BMP7 Yamashita, ten Dijke et al. 1995
- GDF5 Neishitoh, Ichijo et al. 1996)
- GDF11 Oh, Yeo et al. 2002
- ACVR2B is phosphorylated to activate type I receptors.
- Activated type I receptors activate Smad, a transcriptional regulator, to carry out cell signal transduction (Derynck, Zhang et al. 1998). These ligands perform a wide variety of signal transduction mechanisms through ACVR2B, and it is known that overregulation of signal transduction, particularly by overexpression of ligands, indicates various abnormalities if the signal transduction is misregulated.
- the present invention provides a pharmaceutical composition for preventing or treating a disease, comprising a fragment comprising a human antibody or antigen-binding site thereof according to the present invention.
- the present invention provides a pharmaceutical composition for preventing or treating cancer, metabolic disease, immune system disease or liver disease, comprising a fragment comprising a human antibody or antigen-binding portion thereof according to the present invention.
- the present invention provides a method of treating cancer, comprising administering a pharmaceutical composition according to the present invention to a subject having or likely to develop a cancer, metabolic disease, immune system disease, liver disease, or the like. To provide.
- prevention means any action that inhibits or delays the onset of ACVR2B-related diseases including cancer by administration of the pharmaceutical composition of the present invention
- treatment means the pharmaceutical composition of the present invention.
- the term "individual” means any animal, including humans, who may develop or invent ACVR2B related diseases, including cancer.
- the pharmaceutical composition of the present invention may improve or advantageously alter the symptoms caused by cancer through cancer metastasis or invasion inhibiting activity.
- the pharmaceutical composition of the present invention may further comprise a water-soluble DLK1, the water-soluble DLK1 may exhibit an anticancer effect by binding to activin receptor type 2B competitively with activin to block activin signaling.
- the activity of water-soluble DLK1 that inhibits the binding between activin receptor type 2B and activin can be significantly elevated by the human antibody of the present invention, which can effectively treat ACVR2B related diseases including cancer.
- composition of the present invention may further comprise a DLK1-Fc fusion protein conjugated with the water-soluble DLK1 and the antibody Fc region, preferably the antibody Fc region may be a human antibody Fc.
- the pharmaceutical composition of the present invention exhibits anti-cancer effects by inhibiting the binding between activin and activin receptor type 2B to block activin signaling, and thus may be useful for the prevention and treatment of cancer.
- the pharmaceutical composition of the present invention can be used without limitation as long as the activin is overexpressed during the onset or progression of the cancer, or the cancer is grown or metastasized by activin / activin receptor type 2B signaling.
- the metabolic disease may include diabetes or obesity
- the immune system disease may include an autoimmune disease or rheumatoid arthritis
- the liver disease is chronic hepatitis, alcoholic cirrhosis, acute liver failure, liver cancer or fatty liver It may include, but is not limited to diseases associated with signal transduction mechanisms due to the binding of the ligand and ACVR2B.
- the route of administration of the pharmaceutical composition of the present invention may be administered via any general route as long as it can reach the desired tissue or cell.
- the pharmaceutical composition of the present invention may be administered intraperitoneally, intravenously, intramuscularly, subcutaneously, intradermally, orally, pulmonary, rectally, intracellularly or indirectly, as desired.
- the pharmaceutical compositions of the present invention may be administered by any device in which the active substance may migrate to the target cell.
- the pharmaceutical composition of the present invention may comprise an acceptable carrier.
- the pharmaceutical composition comprising a pharmaceutically acceptable carrier may be in various oral or parenteral formulations. When formulated, diluents or excipients such as fillers, extenders, binders, wetting agents, disintegrating agents, and surfactants are usually used. Solid form preparations for oral administration include tablet pills, powders, granules, capsules, and the like, which form at least one excipient such as starch, calcium carbonate, sucrose or lactose in one or more compounds. ) And gelatin.
- Liquid preparations for oral administration include suspensions, solution solutions, emulsions, and syrups, and various excipients, such as wetting agents, sweeteners, fragrances, and preservatives, in addition to commonly used simple diluents such as water and liquid paraffin, may be included.
- Formulations for parenteral administration include sterile aqueous solutions, non-aqueous solvents, suspensions, emulsions, lyophilized preparations, suppositories.
- non-aqueous solvent and the suspension solvent propylene glycol, polyethylene glycol, vegetable oil such as olive oil, injectable ester such as ethyl oleate, and the like can be used.
- As the base of the suppository witepsol, macrogol, tween 61, cacao butter, laurin butter, glycerogelatin and the like can be used.
- the pharmaceutical composition of the present invention is selected from the group consisting of tablets, pills, powders, granules, capsules, suspensions, liquid solutions, emulsions, syrups, sterile aqueous solutions, non-aqueous solutions, suspensions, emulsions, lyophilized preparations and suppositories It can have any one formulation.
- the pharmaceutical composition of the present invention is administered in a pharmaceutically effective amount.
- pharmaceutically effective amount means an amount sufficient to treat a disease at a reasonable benefit / risk ratio applicable to medical treatment, and an effective dose level is determined by the type and severity of the subject, age, sex, activity of the drug, drug Sensitivity, time of administration, route of administration and rate of release, duration of treatment, factors including concurrent use of drugs, and other factors well known in the medical arts.
- composition of the present invention can be used alone or in combination with methods using surgery, hormonal therapy, drug therapy and biological response modifiers for the treatment of cancer.
- the present invention comprises a human antibody or a fragment comprising an antigen-binding site thereof that specifically binds to the extracellular domain water-soluble domain of the delta-like 1 homolog (DLK1), cancer, metabolic diseases ,
- DLK1 delta-like 1 homolog
- An immune system disease or liver disease diagnostic kit is provided.
- the kit may include an enzyme immunoassay (ELISA) kit or a sandwich ELISA kit, but is not limited thereto.
- ELISA enzyme immunoassay
- a fragment of a human antibody or antigen-binding portion thereof that specifically binds to the extracellular domain water-soluble domain of DLK1 of the present invention exhibits anti-cancer effects, as well as the ability of DLK1 to bind to activin receptor type 2B of the extracellular domain water-soluble domain. Increase significantly.
- FIG. 1 is a photograph and a graph showing a metastasis inhibitory effect in pancreatic cancer cell line (MIA-PaCa-2) by soluble DLK1.
- Figure 2 is a photograph and graph showing the effect of inhibiting metastasis in uterine cancer cell line (HeLa) by water-soluble DLK1.
- FIG. 3 is a photograph and graph showing the effect of inhibiting growth independent adhesion in pancreatic cancer cell line (MIA-PaCa-2) by water-soluble DLK1.
- Figure 4 is a photograph and graph showing the effect of inhibiting growth independent adhesion in uterine cancer cell line (HeLa) by water-soluble DLK1.
- FIG. 5 is a photograph showing the wound healing inhibitory effect in pancreatic cancer cell line (MIA-PaCa-2) and kidney cancer cell line (786-O) by water-soluble DLK1.
- Figure 6 is a photograph showing the Western blot results to determine the effect of water-soluble DLK1 on the signaling of real cells.
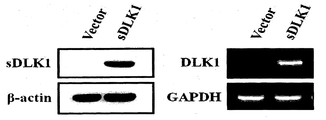
- FIG. 7 is a Western blot and RT-PCR photograph showing that soluble DLK1 was overexpressed in a cell line overexpressing soluble DLK1.
- FIG. 8 is a photograph showing the effects of inhibiting metastasis, invasion, adhesion independence growth, and wound healing by water-soluble DLK1 using cell lines overexpressing water-soluble DLK1.
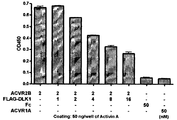
- Figure 10 shows the results by enzyme immunoassay showing that water-soluble DLK1 specifically binds to activin receptor type 2B.
- FIG. 11 shows the results by enzyme immunoassay showing that water soluble DLK1 does not bind to activin A.
- Figure 12 shows the results by a competitive enzyme immunoassay showing that water soluble DLK1 binds to activin receptor type 2B competitively with activin.
- FIG. 13 shows the results by surface plasmon resonance showing that water-soluble DLK1 specifically binds to activin receptor type 2B in various activin receptor families.
- FIG. 14 is a Western blot photograph showing the activin receptor type 2B expression of cells used in flow cytometry migration assay.
- FIG. 15 shows flow cytometry migration assay results showing that binding between water soluble DLK1 and activin receptor type 2B is occurring on real cells.
- Figure 17 shows the results confirmed by fingerprinting the diversity of monophageclonal antibodies to water-soluble DLK1.
- Figure 18 shows the results of analyzing the polypeptides in the CDRs of monophageclonal antibodies against water soluble DLK1.
- Figure 21 shows the flow cytometry analysis results (top) and fluorescence immunostaining results (bottom) showing that the B09 antibody specifically binds to the water-soluble portion of DLK1.
- Figure 22 shows the results of measuring the binding force between the water-soluble DLK1 and B09 antibody by the surface plasmon resonance method.
- Figure 23 shows the results of the binding force measurement between activin A and activin receptor type 2B by surface plasmon resonance method.
- Fig. 24 shows the result of binding force measurement between water-soluble DLK1 and activin receptor type 2B by surface plasmon resonance method.
- Fig. 25 shows the results of measurement of the binding force between the water-soluble DLK1 and the activin receptor type 2B when the B09 antibody and the water-soluble DLK1 were treated together by the surface plasmon resonance method.
- Fig. 26 is a schematic diagram showing the effect of the B09 antibody on the binding strength between water-soluble DLK1 and activin receptor type 2B.
- FIG. 27 shows the results by competition enzyme immunoassay showing the effect of antibodies on binding between water soluble DLK1 and activin receptor type 2B.
- FIG. 28 shows the results by competition enzyme immunoassay showing the effect of antibodies on the binding between activin A and activin receptor type 2B.
- FIG. 29 shows the results by competitive enzyme immunoassay showing the effect of water soluble DLK1, DLK1 antibodies and activin receptor type 2B antibodies on binding between activin A and activin receptor type 2B.
- Figure 31 shows the results of SEAP reporter analysis showing the effect of water-soluble DLK1 on Smad signaling.
- Figure 32 shows the results of SEAP reporter analysis showing the effect of the B09 antibody on Smad signaling.
- HRE hypoxia response element
- Figure 34 shows the metastasis inhibitory effect of activin receptor type 2B antibody and DLK1 antibody.
- 35 shows the effects of activin receptor type 2B antibody and DLK1 antibody on the metastasis inhibitory effect of water-soluble DLK1.
- Figure 40 shows that the non-large band of the DLK1-Fc treatment group in pancreatic cancer in situ transplantation metastasis animal model experiments.
- FIG. 41 shows in vivo images of pancreatic cancer in situ transplantation metastasis animal model.
- the extracellular region water soluble domain (hereinafter referred to as water soluble DLK1) of DLK1 and the DLK1-Fc fusion protein (hereinafter referred to as DLK1-Fc) in which the water soluble DLK1 and the human antibody Fc region are conjugated as used in the Examples of the present specification are Prepared according to the method disclosed in 0982170.
- a migration assay of cancer cell lines was performed using the method of Reference (Chen HC, Methods in molecular biology. 294: 15-22, 2005).
- cells MIA-PaCa-2, HeLa cell line
- the cells were changed to serum-free medium, and after 24 hours, the cells were detached through trypsin treatment, and the number of cells was measured. 100 ⁇ l of cells, serum-free medium and proteins to be treated were combined and incubated at 37 ° C. for 1 hour.
- Soft agar assay was performed to determine the effect of water-soluble DLK1 on anchorage independence growth.
- RPMI medium containing 10% FBS and 1% agar were mixed 1: 1 and placed on a 60 mm culture dish to form 0.5% agar, followed by 6 x 10 3 cells (MIA-PaCa-2 , HeLa cell line) with 5 ⁇ g / ml of DLK1-Fc or Fc, and then mixed with RPMI medium containing 10% FBS and 1% agar, the concentration of the final agar was 0.3%, followed by 0.5% agar. Laid on top. It was incubated in a 37 ° C.
- Pancreatic cancer cell line MIA-PaCa-2 or kidney cancer cell line 786-O was grown in 6-well plates and then changed to serum-free medium for 16 hours when 90% of the cells grew. Thereafter, the cells were treated with DLK1-Fc or Fc at a concentration of 5 to 25 ⁇ g / ml for 1 hour, after which the cells were scratched and 5% FBS was added, and the 786-O cell line was 16 hours for MIA. The PaCa-2 cell line was cultured for 48 hours, and the wound was observed.
- Harvested cells were prepared in RIPA buffer (50 mM TrisHCl pH 7.4, 150 mM NaCl, 2 mM EDTA, 1% NP-40), protease inhibitor (Roche), phosphotase inhibitor cocktail I, II (phosphotase inhibitor cocktail I Cells were lysed using a buffer added with II, Sigma), and the concentration of lysed cell lysate was determined using a BCA quantitative kit (Thermo). 30 ⁇ g of the lysate on SDS-PAGE was denatured using 5 ⁇ sample buffer, electrophoresed, and transferred to NC membrane (Watman).
- Western blot was performed using p-FAK, FAK, p-AKT, AKT, p-eNOS, eNOS, p-ERK1 / 2 and ERK1 / 2 antibody (Cell signaling).
- beta-actin antibody Sigma
- the blot was fluorescently developed using an ECL solution (Intron) to develop a film, as shown in FIG. 6.
- a cell line overexpressing soluble DLK1 was constructed using the pancreatic cancer cell line MIA-PaCa-2 cell line.
- the cell line was constructed by incorporating a DNA fragment encoding the extracellular domain region of DLK1 into a pcDNA 3.1 vector.
- the cell line was screened at 500 ⁇ g / ml G418 (Geneticin, GIBCO / BRL), RT-PCR was performed to confirm that the cell line was well established, and the cultured cell was grown 10-fold using Centricon 15 (Millpore). After Western blot, it was confirmed whether the water-soluble DLK1 was well expressed.
- Western blot used the DLK1 monoclonal antibody (R & D) product. In the comparison group, only the vector without the insert was used by transfection.
- the cell line constructed in Example 5 was used to analyze metastasis and infiltration mechanism. First, the cells were changed to serum-free medium when the level was about 50%, and after 24 hours, the cells were detached through trypsin treatment, and the number of cells was measured. Place 5-10% FBS with 1 ml of chemo-attractant in a 24-well plate, place a transwell (Corning # 3422) with 8.0 ⁇ m pore on it, and then place 5 ⁇ 10 4 Cells were added and then cultured in a 37 ° C. carbon dioxide incubator for 24 to 48 hours.
- the transwell medium was removed, the cells were stained with Diff Quick solution (Sysmex, Japan), and the cells that did not pass through the transwell were completely removed using a cotton swab. Then, after completely drying the transwell, the cells passing through the transwell were observed at a magnification of 400 times and photographed.
- the experiment was performed after coating 100 ⁇ l of Matrigel (BD Science) at a concentration of 1 mg / ml on the lower side of the transwell.
- Matrigel BD Science
- RPMI medium containing 10% FBS and 1% agar were mixed 1: 1 and placed in a 60 mm culture dish to obtain 0.5% agar composition, followed by 6 ⁇ 10 3 cells in 10% FBS. After mixing 1% agar with RPMI medium containing the final concentration of agar to 0.3% and then laid on 0.5% agar. Incubated for 3 weeks in a 37 °C CO2 incubator, medium was added every 3 days to prevent agar from drying. After colonies were produced, photographs were taken to observe the results.
- Wound healing assays were grown in 6-well plates and then changed to serum-free medium when 90% of the cells were grown. After culturing in serum-free medium for 16 hours, the cells were scraped and wounded, and the wound was filled after incubation for 48 hours with 5% FBS.
- Co-immunoprecipitation was performed to identify receptors of water-soluble DLK1.
- soluble DLK1 and ACVR2B were overexpressed in 293E cells by transfection using PEI (Polyscience) method.
- Cells were washed three times with cold DPBS and lysed in modified RIPA buffer (50 mM TrisHCl pH7.4, 150 mM NaCl, 2 mM EDTA, 1% NP-40). This lysate was pre-cleared using human IgG (Jackson Lab) and protein G beads (GE). Immunoprecipitation was performed using human antibodies and protein G beads produced in the laboratory, and samples were separated on SDS-PAGE and Western blots were shown in FIG. 9.
- Enzyme immunoassay confirmed the binding between ACVR2B and soluble DLK1.
- water-soluble DLK1 tagged with 1 ⁇ g / ml FLAG was coated on immunoplate (Nunc) at 4 ° C. for 16 hours.
- BSA was used as a control for measuring nonspecific binding.
- the coated plate was washed three times with PBST (0.05% Tween 20) and then blocked with 5% skim milk PBST.
- ACVR2B-Fc with Fc attached to the extracellular part of ACVR2B was subjected to serial dilution and allowed to react at room temperature for 1 hour.
- 293E was also prepared.
- the harvested cells were washed twice with cold DPBS and 1 ⁇ g of DLK1-Fc-FITC was added and reacted at 4 ° C. for 2 hours.
- DLK1-Fc-FITC used for the reaction was prepared using the Pierce protein FITC labeling kit. After the reaction, washing was performed using 1% PBA (1% BSA in DPBS) buffer, and the degree of binding was determined by flow cytometry.
- the cells used above confirmed the expression pattern of ACVR2B via Western blot (FIG. 14).
- Example 2 30 ⁇ g of the purified DLK1-Fc obtained in Example 1 was added to an Immunosorb tube (Nunc 470319) in 4 ml of a coating buffer; 1.59 g of Na 2 CO 3 (sigma, S7795), 2.93 g of NaHCO 3 (sigma, S8875), NaN 3 ( sigma, S2002), 0.2 g], coated with a rotator at 4 ° C. for about 16 hours, followed by room temperature. It was dissolved in PBS for 2 hours at and blocked in immunotubes using skim milk [(BD, 232100) -4% in 1XPBS].
- the cultured cells were centrifuged (4500 rpm, 15 min, 4 ° C.) and the supernatant (panned poly scFv-phage) was transferred to a new tube.
- 96-well immune plates (NUNC 439454) were coated with antigen at 100 ng per well with coating buffer for 16 hours at 4 ° C., and then each well was blocked using skim milk (4%) dissolved in PBS. Wash each well with 0.2 ml of PBS-tween20 (0.05%), and add 100 ⁇ l of the diluted solution of panned poly scFV-phage and 5, 25, 125, 625, and 3125 times to each well. Reacted for a while.
- the secondary antibody anti-M13-HRP (Amersham 27-9421-01) was diluted 1: 2000 and reacted at room temperature for 1 hour. It was.
- Colonies obtained from the polyclonal phage antibody group having high binding capacity were incubated for 16 hours at 37 ° C. in a 96-deep well plate (Bionia 90030) containing 2 ml of YTCM, 2% glucose, and 1 ml of 5 mM MgCl 2 medium. It was. Take 100-200 ⁇ l of the cultured cells so that the OD600 value is 0.1 and dilute in 1 ml 2 ⁇ YTCM, 2% glucose, 5 mM MgCl 2 medium, and then measure the OD600 value at 37 ° C. in a 96-deep well plate. Incubated for 2-3 hours so as to be 0.5-0.7.
- M1 helper phages were infected with a MOI of 1:20, and then incubated for 16 hours at a temperature of 30 ° C. in 2 ⁇ YTCMK, 5 mM MgCl 2 , and 1 mM IPTG medium. After culturing the cells were centrifuged (4500 rpm, 15 minutes, 4 °C), the supernatant was taken, dissolved by adding 4% PEG 6000 and 3% NaCl, and reacted for 1 hour on ice.
- each well was washed with 0.2 ml of PBS-tween20 (0.05%), and 100 ⁇ l of monoclonal scFv-phage (each 100 scFv-phage) obtained by the above method was added to each well and allowed to react at room temperature for 2 hours. .
- each well was washed four times with 0.2 ml of PBS-tween20 (0.05%), and the second antibody, anti-M13-HRP, was diluted to 1/2000 and reacted at room temperature for 1 hour. After washing with 0.2 ml of PBS-tween20 (0.05%), color development was measured at 490 nm. As a result, as shown in Table 2, 27 single phage clones having a binding capacity of 2 or more antigens were selected.
- the colony PCR product was identified on 1% agarose gel (Seakem LE, CAMERES 50004), and 0.2 ⁇ l of BstNI (Roche11288075001, 10 U / ⁇ l) was added and reacted at 37 ° C. for 2-3 hours.
- the reaction conditions are shown in Table 4 below.
- the cleaved product was identified on an 8% DNA polyacrylamide gel.
- the heavy chain was 1 ⁇ l of monoclonal DNA, 10 pmole / ⁇ l of heavy chain forward and reverse primers in Table 6, 5 ⁇ l of 10 ⁇ buffer, 1 ⁇ l of 10 mM dNTP mix.
- Colony PCR iCycler iQ, BIO-RAD was performed by mixing 0.5 ⁇ l of pfu DNA polymerase (solgent, 2.5 U / ⁇ l) and distilled water.
- Light Chain Colony PCR was performed in the same manner using the light chain forward and reverse primers in Table 6.
- the heavy chain gene obtained by PCR was purified by DNA-gel extraction kit (Qiagen), and then 1 ⁇ l (10 ng) of pNATAB H vector (FIG. 19), 15 ⁇ l of heavy chain (100-200 ng), 2 ⁇ l of 10 ⁇ buffer, 1 ⁇ l of ligase (1 U / ⁇ l) and distilled water were mixed and left at room temperature for 1-2 hours to connect with the vector.
- the vector was allowed to stand on ice with transformed cells (XL1-blue) for 30 minutes, and then transduced with heat shock at 42 ° C. for 90 seconds. After standing again on ice for 5 minutes, 1 ml of LB medium was injected and incubated at 37 ° C. for 1 hour.
- the plate was plated on LB Amp solid medium, and then cultured at 37 ° C. for 16 hours. Single colonies were inoculated in 5 ml of LB Amp liquid medium and incubated at 37 ° C. for 16 hours. DNA-prep. DNA was extracted using the kit (Nuclogen). In addition, the light chain was extracted DNA in the same manner as above using the pNATAB L vector (Fig. 20).
- the obtained DNA was requested for sequencing using CMV-proF primer (SEQ ID NO: 54: AAA TGG GCG GTA GGC GTG) (solgent). As a result, it was confirmed that the sequences of the heavy and light chains of the six clone phages for DLK1-Fc converted to total IgG match the sequences of the phage antibody.
- the surface plasmon resonance method was performed using ProteOn XPR36 of Bio-Rad, and the method is summarized as follows.
- the sensor chip was activated by flowing 0.1 M EDC and 0.025 M sulfo-NHS for 60 seconds, and the protein to be coated was added to 10 mM sodium acetate (pH 5.0) at a rate of 30 ⁇ l / min for 240 seconds to attach to the chip.
- the channel was blocked with 1 M ethanolamine-HCl (pH 8.5) for 200 seconds and then washed with DPBST (PBS with 0.005% tween 20).
- DPBST DPBST
- the serial dilution was performed twice, and the binding force was measured by flowing for 120 seconds at a rate of 30 ⁇ l / min.
- a dissociation phase was taken for 240 seconds.
- the binding force of the B09 antibody to the water-soluble DLK1 was measured with a Kd value of 1.90E-11 (FIG. 22).
- the binding force of activin A to ACVR2B was measured with a Kd value of 1.96E-09 (FIG. 23)
- the binding force of water-soluble DLK1 to ACVR2B was measured with a Kd value of 1.31E-09 (FIG. 24).
- Pancreatic cancer cell line, MIA-PaCa-2 was grown to about 70% in culture vessels, exchanged with serum-free medium and incubated for 16 hours.
- the DLK1-Fc treatment group was treated with 10 ⁇ g / ml DLK1-Fc, and 0.5 nM activin A was added and harvested 30 minutes later.
- Harvested cells were prepared using RIPA buffer (50 mM TrisHCl pH 7.4, 150 mM NaCl, 2 mM EDTA, 1% NP-40) with protease inhibitor (Roche) phosphatase inhibitor cocktail I, II (Sigma). After lysing the cells, the concentration of the lysed cell lysate was determined using a BCA quantitative kit (Thermo).
- SEAP reporter analysis was performed to determine the effect of water-soluble DLK1 on Smad neurotransmission.
- the vector used for SEAP reporter analysis has a Smad binding element (SBE) that is increased by activin signaling in front of the SEAP gene. have.
- SBE Smad binding element
- the vector was introduced by electroporation into the MIA-PaCa-2 cell line. The next day the transfected cell lines were transferred to 96-well plates, incubated for 24 hours and further incubated for 16 hours with serum free medium. Cells were pretreated with Fc or DLK1-Fc for 1 hour, treated with 0.5 nM of activin A, incubated for 48 hours, and cell culture was collected for reporter analysis. The analysis was performed using ABI's Phospha-Light system. In addition, the same experiment was performed using the B09 antibody.
- a reporter assay was performed to determine the effect of B09 antibodies that specifically bind to the water-soluble portion of DLK1 on signal transduction in hypoxia conditions in which DLK1 is overexpressed.
- the vector used for reporter analysis has a hypoxia response element (HRE) in front of the luciferase gene, and thus has a system in which the reporter gene is expressed when transcription control by hypoxia is performed in the cell.
- HRE hypoxia response element
- the vector was introduced into the MIA-PaCa-2 cell line by electroporation (Invitrogen). The next day the transfected cell lines were transferred to 96-well plates, incubated for 24 hours and further incubated for 16 hours with serum free medium.
- CoCl 2 (Sigma) was treated to cause artificial hypoxia.
- Cells were pretreated with DLK1 antibody for 1 hour and subjected to reporter analysis. Analysis was performed using ABI's Phospha-Light system.
- metastasis experiments were performed using MIA-PaCa-2 cell line.
- the cells were changed to serum-free medium when the level was about 70%, and after 24 hours, the cells were detached through trypsin treatment, and the number of cells was measured.
- Cells, serum-free medium and each protein to be treated were combined to make 100 ⁇ l and incubated at 37 ° C. for 1 hour.
- Example 19 orthotopic animal model of MIA-Paca-2 pancreatic cancer
- MIA-Paca-2 pancreatic cancer In situ transplantation animal model experiment of MIA-Paca-2 pancreatic cancer was performed to confirm the ability of DLK1-Fc to metastasize cancer and inhibit the proliferation of cancer cells.
- Nude mice used in the experiment were Can Cg-Foxnl-nu / CrljBgi, and 6-week-old females were purchased from Orient Co., Ltd.
- 5 ⁇ 10 6 MIA-Paca-2 cell lines which are pancreatic cancer cell lines, were orally transplanted into the pancreas. Divided into two groups, one group injected DLK1-Fc at 5 mpk, the other group received 15 mpk, and PBS was used as a control. Protein was injected by injection twice a week via intraperitoneal injection. Five weeks after orthodontic transplantation, the results were observed through dissection.
- SEQ. ID. NO. 1 extracellular water-soluble domain of DLK1: ECFPACNPQN GFCEDDNVCR CQPGWQGPLC DQCVTSPGCL HGLCGEPGQC ICTDGWDGEL CDRDVRACSS APCANNGTCV SLDDGLYECS CAPGYSGKDC QKKDGPCVIN GSPCQHGGTC VDDEGRASHA SCLCPPGFSG NFCEIVANSC TPNPCENDGV CTDIGGDFRC RCPAGFIDKT CSRPVTNCAS SPCQNGGTCL QHTQVSYECL CKPEFTGLTC VKKRALSPQQ VTRLPSGYGL AYRLTPGVHE LPVQQPEHRI LKVSMKELNK KTPLLTEG
- SEQ. ID. NO. 2 (CDR1 of heavy chain): SYAMN
- SEQ. ID. NO. 3 (CDR2 of heavy chain): TITATSGKTY YADSVKG
- SEQ. ID. NO. 7 (CDR3 of light chain): GSYAGSYTY
- SEQ. ID. NO. 9 (CDR2 of heavy chain): WINPGSGNTK YSHNFEG
- SEQ. ID. NO. 10 (CDR3 of heavy chain): SVSAYGSNYF DP
- SEQ. ID. NO. 16 (CDR3 of heavy chain): SGGYGGNTNW YFDL
- SEQ. ID. NO. 18 (CDR2 of light chain): GNNNRPS
- SEQ. ID. NO. 19 (CDR3 of light chain): QSYDSRLGV
- SEQ. ID. NO. 21 (CDR2 of heavy chian): GISWNSGSIG YADSVKG
- SEQ. ID. NO. 22 (CDR3 of heavy chain): GPGLATGKGY ADY
- SEQ. ID. NO. 27 (CDR2 of heavy chain): SIPGSGTRTH YADSVKG
- SEQ. ID. NO. 28 (CDR3 of heavy chain): STAYLFDY
- SEQ. ID. NO. 33 (CDR2 of heavy chain): YISGSGITTY YADSVKG
- SEQ. ID. NO. 34 (CDR3 of heavy chain): LQGHCSGGAC SNWFDA
- SEQ. ID. NO. 38 (heavy chain VR of B09): QMQLVESGGR LVRPGGSLRL SCAASGFPFT SYAMNWVRQT PGKGLEWVST ITATSGKTYY ADSVKGRFTI SRDNSRNTLF LQMNSLRAED TAVYYCVRGE SCSGGACSDF DYWGQGALVT VSS
- SEQ. ID. NO. 39 (light chain VR of B09): QLVLTQPASV SGSPGQSVTI SCTGTSSDIG RYNRVSWYQH HPGKAPKLIL NDVTTRPSGF SNRFSGSKSG NTASLTISGL QAEDEADYSC GSYAGSYTYV FGTGTKVTVL GGG
- SEQ. ID. NO. 41 (light chain VR of A04): DIQMTQSPSS LSASVGDRVT ITCQASQDIS NYLNWYQQKP GKAPKLLIYS TSNLQSGVPS RFSGSGSGTD FTLTISSLQP EDFATYYCQQ LNSYPLTFGG GTKVDIKRGG
- SEQ. ID. NO. 42 (heavy chain VR of A05): QMQLVESGGG LVQPGRSLRL SCAASGFTFD EHAMHWVRQA PGKGLEWVSG INWNSGKTGY ADSVKGRFTI SRDNGKNSLY LQMNSLRAED TAVYYCAKSG GYGGNTNWYF DLWGRGTLVT VSS
- SEQ. ID. NO. 43 (light chain VR of A05): QFVLTQPPSV SGAPGQNVTI SCIGTSSNIG VGYDVHWYQQ VPGTAPKLLV YGNNNRPSGV PDRFSGSKSG TSASLAITGL QAEDEADYYC QSYDSRLGVV FGGGTKLTVL GGG
- SEQ. ID. NO. 44 (heavy chain VR of A10): QVQLVESGGG LVQPGRSLRL SCAASGFTFD DYAMHWVRQA PGKGLEWVSG ISWNSGSIGY ADSVKGRFTI SRDNAKNSLY LQMNSLRAED TAVYYCARGP GLATGKGYAD YWGQGTLVTV SS
- SEQ. ID. NO. 45 (light chain VR of A10): DIQMTQSPSS VSASVGDRVT ITCRASQRIS SWLAWYQQKP GRAPKLLIHS ASTLHNGVPS RFSGSASGTD FTLTISSLQP EDYAIYYCQQ GHSFPYTFGQ GTKLEIKRGG
- SEQ. ID. NO. 46 (heavy chain VR of H06): QVQLVQSGGG LIQPGGSLRL SCAASGFTFS LYGMSWVRQA PGKGLEWVSS IPGSGTRTHY ADSVKGRFTI SRDNSKNTLF LQMNSLRAED TAVYYCAKST AYLFDYWGPG TLVTVSS
- SEQ. ID. NO. 47 (light chain VR of H06): DIQMTQSPAT LSLSPGERAT LSCRASQSIR HYLAWYQQKP GQAPRLLIHG ASSRATGIPD RFSGSGSGTD FTLTISRLEP EDFAVYYCQH YGSPLHSFGP GTKVEIKRGG
- SEQ. ID. NO. 48 (heavy chain VR of H12): QVQLVQSGGG LVKPGGSLRL SCAASEFTFS DYYMSWVRQA PGKGLEWLSY ISGSGITTYY ADSVKGRFTI SRDNGKKSLY LEMNSLRAED TAVYYCARLQ GHCSGGACSN WFDAWGQGTL ITVSS
- SEQ. ID. NO. 49 (light chain VR of H12): DIQMTQSPSS LSASVGDRLT ITCRASQSIL TYLNWYQQKP GKAPKLLIYA ASSLQRGVPS RFSGSGTD FTLTISGLQP EDFATYYCQQ GYGTPYTFGQ GTKVDIKRGG
- SEQ. ID. NO. 50 polynucleotide sequence coding heavy chain of B09 antibody: atgggatgga gctatatcat cctctttttg gtggccacag cggccgatgt ccactcgcag atgcagctgg tggagtctgg gggacgctta gtgcggcctg gggggtcctg gagactctcc tgtgcagcct ct cttaccagt tatgccatga actgggtccg ccagactcca gggaaggggc tggaatgggt ctctactatc actgctacta gtggtaagac atactacgca gactccgtgaaaggc tggaatgggt ctctactatc actgctacta gtggtaagac atactacg
- SEQ. ID. NO. 51 polynucleotide sequence coding light chain of B09 antibody: atgggatgga gctatatcat cctctttttg gtggccacag cggccgatgt ccactcgcag ctcgtgctga ctcagcctgc ctccgtgtctg gacagtcggt caccatctcc tgcactggaa ccagcagtga cattggtcgt tataaccgtg tctcctggta ccaacaccac ccggcaagg ccccaaact cattctttaat gatgtcacta ctcggccctc agggttttct aatcgcttctccaa gtctggcaacggcggcccaaact cattcttaat gatgtcacta c
- SEQ. ID. NO. 52 forward primer: ctagataacg agggcaaatc atg
- SEQ. ID. NO. 56 (NATVH7-1 primer): ttggtggcca cagcggccga tgtccactcg cagatgcagc tggtggagtc
- SEQ. ID. NO. 58 (NATJH-ALL primer): gaggaggcta gctgaggaga cggtga
- SEQ. ID. NO. 60 (NATVL13 primer): ttggtggcca cagcggccga tgtccactcg cagttcgtgc tgactcagcc
- SEQ. ID. NO. 61 (NATVL10 primer): ttggtggcca cagcggccga tgtccactcg cagctcgtgc tgactcagcc
- SEQ. ID. NO. 62 (NATJK-R7 primer): gaggagagat cttttgatat ccaccttggt
- SEQ. ID. NO. 63 (NATJL2-R primer): gaggagagat cttaggacgg tcagcttggt ccc
- SEQ. ID. NO. 64 (NATJK-R5 primer): gaggagagat cttttgattt ccagcttggt
- SEQ. ID. NO. 65 (NATJL1-R primer): gaggagagat cttaggacgg tgaccttggt ccc
- SEQ. ID. NO. 66 (NATJK-R4 primer): gaggagagat cttttgattt ccaccttggt
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Gastroenterology & Hepatology (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Molecular Biology (AREA)
- Immunology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Toxicology (AREA)
- Zoology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Peptides Or Proteins (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
La présente invention concerne les anticorps humains qui se lient spécifiquement au domaine soluble dans l'eau extracellulaire de DLK1 et des fragments de ceux-ci qui comprennent des sites de liaison à un antigène ; un polynucléotide qui code pour les anticorps humains ou des fragments de ceux-ci ; un vecteur recombinant contenant le polynucléotide ; des transformants transformés par le vecteur recombinant ; un procédé de fabrication des anticorps humains ou des fragments de ceux-ci ; une composition pharmaceutique, pour la prévention ou le traitement d'une maladie, qui comprend les anticorps humains ou un fragment de ceux-ci ; des nécessaires de diagnostic. Les anticorps humains et des fragments qui comprennent des sites de liaison à un antigène qui se lient de façon spécifique au domaine soluble dans l'eau extracellulaire de DLK1 de la présente invention présentent une activité antinéoplasique. En augmentant de façon significative la capacité de liaison du domaine soluble dans l'eau extracellulaire de DLK1 au récepteur de type II B de l'Activine (ACVR2B), son utilité est démontrée pour la prévention et le traitement de maladies associées à la transduction de signal par ACVR2B, comprenant des troubles métaboliques associés, une maladie immunitaire, les maladies hépatiques et le cancer.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2011-0030871 | 2011-04-04 | ||
| KR20110030871 | 2011-04-04 | ||
| KR1020120001457A KR101438265B1 (ko) | 2011-04-04 | 2012-01-05 | Dlk1 특이적 인간 항체 및 이를 포함하는 약학적 조성물 |
| KR10-2012-0001457 | 2012-01-05 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2012138102A2 true WO2012138102A2 (fr) | 2012-10-11 |
| WO2012138102A3 WO2012138102A3 (fr) | 2013-01-10 |
Family
ID=46969663
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2012/002496 WO2012138102A2 (fr) | 2011-04-04 | 2012-04-03 | Anticorps humains spécifiques de dlk1 et compositions pharmaceutiques les contenant |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2012138102A2 (fr) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3657176A1 (fr) * | 2018-11-23 | 2020-05-27 | Syddansk Universitet | Dlk1 comme marqueur prédictif pour la résistance à l'insuline et/ou le diabète de type 2 et/ou un syndrome métabolique |
| CN111278860A (zh) * | 2017-09-08 | 2020-06-12 | Y生物股份有限公司 | 抗人dlk1的抗体及其用途 |
| CN112135638A (zh) * | 2019-03-06 | 2020-12-25 | 乐高化学生物科学股份有限公司 | 包含抗人dlk1抗体的抗体-药物偶联物及其用途 |
| EP3936150A4 (fr) * | 2019-03-06 | 2023-03-29 | LegoChem Biosciences, Inc. | Conjugués anticorps-médicament comprenant un anticorps contre dlk1 humain et utilisation associée |
| US11827703B2 (en) | 2018-05-09 | 2023-11-28 | Legochem Biosciences, Inc. | Compositions and methods related to anti-CD19 antibody drug conjugates |
| US11975076B2 (en) | 2015-11-25 | 2024-05-07 | Legochem Biosciences, Inc. | Antibody-drug conjugates comprising branched linkers and methods related thereto |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1702982A1 (fr) * | 2003-11-28 | 2006-09-20 | Kanagawa Academy Of Science And Technology | Methode de detection, diagnostic et remede contre le cancer du foie |
| EP1394263B1 (fr) * | 2001-05-16 | 2008-10-08 | Kanagawa Academy Of Science And Technology | Procede pour detecter et isoler des cellules hepatiques indifferenciees au moyen de dlk |
| KR20090088893A (ko) * | 2006-11-10 | 2009-08-20 | 가부시키가이샤 리부텍쿠 | in vivo 에서 항종양 활성을 갖는 항인간 Dlk-1 항체 |
| KR100982170B1 (ko) * | 2010-03-16 | 2010-09-15 | 한국생명공학연구원 | DLK1―Fc 융합 단백질을 유효성분으로 함유하는 암 전이 억제용 조성물 |
-
2012
- 2012-04-03 WO PCT/KR2012/002496 patent/WO2012138102A2/fr active Application Filing
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1394263B1 (fr) * | 2001-05-16 | 2008-10-08 | Kanagawa Academy Of Science And Technology | Procede pour detecter et isoler des cellules hepatiques indifferenciees au moyen de dlk |
| EP1702982A1 (fr) * | 2003-11-28 | 2006-09-20 | Kanagawa Academy Of Science And Technology | Methode de detection, diagnostic et remede contre le cancer du foie |
| KR20090088893A (ko) * | 2006-11-10 | 2009-08-20 | 가부시키가이샤 리부텍쿠 | in vivo 에서 항종양 활성을 갖는 항인간 Dlk-1 항체 |
| KR100982170B1 (ko) * | 2010-03-16 | 2010-09-15 | 한국생명공학연구원 | DLK1―Fc 융합 단백질을 유효성분으로 함유하는 암 전이 억제용 조성물 |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11975076B2 (en) | 2015-11-25 | 2024-05-07 | Legochem Biosciences, Inc. | Antibody-drug conjugates comprising branched linkers and methods related thereto |
| CN111278860A (zh) * | 2017-09-08 | 2020-06-12 | Y生物股份有限公司 | 抗人dlk1的抗体及其用途 |
| EP3683236A4 (fr) * | 2017-09-08 | 2021-10-13 | Y-Biologics Inc. | Anticorps dirigé contre dlk1 humain et utilisation associée |
| US11306141B2 (en) | 2017-09-08 | 2022-04-19 | Y-Biologics Inc. | Antibody against human DLK1 and use thereof |
| CN111278860B (zh) * | 2017-09-08 | 2024-03-29 | Y生物股份有限公司 | 抗人dlk1的抗体及其用途 |
| US11827703B2 (en) | 2018-05-09 | 2023-11-28 | Legochem Biosciences, Inc. | Compositions and methods related to anti-CD19 antibody drug conjugates |
| EP3657176A1 (fr) * | 2018-11-23 | 2020-05-27 | Syddansk Universitet | Dlk1 comme marqueur prédictif pour la résistance à l'insuline et/ou le diabète de type 2 et/ou un syndrome métabolique |
| CN112135638A (zh) * | 2019-03-06 | 2020-12-25 | 乐高化学生物科学股份有限公司 | 包含抗人dlk1抗体的抗体-药物偶联物及其用途 |
| EP3936150A4 (fr) * | 2019-03-06 | 2023-03-29 | LegoChem Biosciences, Inc. | Conjugués anticorps-médicament comprenant un anticorps contre dlk1 humain et utilisation associée |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2012138102A3 (fr) | 2013-01-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101438265B1 (ko) | Dlk1 특이적 인간 항체 및 이를 포함하는 약학적 조성물 | |
| WO2012138102A2 (fr) | Anticorps humains spécifiques de dlk1 et compositions pharmaceutiques les contenant | |
| WO2020096381A1 (fr) | Anticorps humain présentant une affinité élevée vis-à-vis du récepteur alpha d'il -4 humain, et son utilisation | |
| AU2019386549B2 (en) | Anti-4-1BB antibody and use thereof | |
| WO2019098682A1 (fr) | Anticorps anti-her2 ou fragment de liaison à l'antigène de celui-ci, et récepteur antigénique chimérique le comprenant | |
| WO2019225777A1 (fr) | Anticorps anti-ror1 et son utilisation | |
| WO2016137108A1 (fr) | Nouvel anticorps se liant à la tfpi et composition le comprenant | |
| WO2014077648A1 (fr) | Anticorps se liant spécifiquement à la protéine l1cam humaine et murine, et son utilisation | |
| WO2016114567A1 (fr) | Procédé de préparation d'une nouvelle bibliothèque d'anticorps et bibliothèque ainsi préparée | |
| WO2014030780A1 (fr) | Procédé de criblage et de fabrication de domaines variables d'immunoglobulines super-stables et applications associées | |
| WO2014090053A1 (fr) | Anticorps monoclonal pour l'antagonisation et l'inhibition de la liaison du facteur de croissance cellulaire endothélial vasculaire et de son récepteur, et sa séquence codante et son utilisation | |
| WO2015058573A1 (fr) | Anticorps monoclonal pour l'antagonisme et l'inhibition de la liaison de mort programmée (pd-1) à son ligand et sa séquence codante et son utilisation | |
| WO2022039490A1 (fr) | Anticorps bispécifiques anti-b7-h4/anti-4-1bb et leurs utilisations | |
| WO2015133882A1 (fr) | Banque d'anticorps scfv, son procédé de préparation et procédé de criblage d'anticorps scfv l'utilisant | |
| WO2016013870A1 (fr) | Procédé pour positionner, dans le cytoplasme, un anticorps au format immunoglobuline complète par pénétration de l'anticorps à travers la membrane cellulaire, et son utilisation | |
| WO2020080908A1 (fr) | Anticorps anti-l1cam ou fragment de liaison à l'antigène de celui-ci, et récepteur antigénique chimérique le comprenant | |
| WO2022177394A1 (fr) | Anticorps bispécifique à domaine unique dirigé contre pd-l1 et cd47 et son utilisation | |
| WO2020251316A1 (fr) | ANTICORPS BISPÉCIFIQUE DIRIGÉ CONTRE α-SYN/IGF1R ET UTILISATION ASSOCIÉE | |
| WO2021210939A1 (fr) | Afficorps anti-her2 et récepteur antigénique chimérique commutable utilisant celui-ci en tant que molécule de commutation | |
| WO2022103245A1 (fr) | Anticorps à domaine unique dirigé contre sars-cov-2 et son utilisation | |
| WO2022149837A1 (fr) | Anticorps anti-fgfr3 et son utilisation | |
| WO2016013871A1 (fr) | Procédé d'inhibition de la protéine ras activée dans une cellule utilisant un anticorps ayant la capacité de pénétrer dans le cytoplasme et se présentant sous la forme d'une immunoglobuline complète, et son utilisation | |
| WO2022124866A1 (fr) | Anticorps anti-pd-1 et ses utilisations | |
| WO2021101346A1 (fr) | Anticorps bispécifiques anti-ror1/anti-4-1bb et leurs utilisations | |
| WO2022085905A1 (fr) | Anticorps se liant spécifiquement à la protéine de spicule du sars-cov-2 et utilisation associée |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12768656 Country of ref document: EP Kind code of ref document: A2 |
|
| NENP | Non-entry into the national phase in: |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12768656 Country of ref document: EP Kind code of ref document: A2 |