WO2011105568A1 - Dried plant tissue and plant tissue extract for ameliorating central nervous system degenerative diseases accompanied by learning/memory disorders, movement disorders and the like, and pharmaceutical agent and food or beverage each comprising the dried plant tissue and the plant tissue extract - Google Patents
Dried plant tissue and plant tissue extract for ameliorating central nervous system degenerative diseases accompanied by learning/memory disorders, movement disorders and the like, and pharmaceutical agent and food or beverage each comprising the dried plant tissue and the plant tissue extract Download PDFInfo
- Publication number
- WO2011105568A1 WO2011105568A1 PCT/JP2011/054358 JP2011054358W WO2011105568A1 WO 2011105568 A1 WO2011105568 A1 WO 2011105568A1 JP 2011054358 W JP2011054358 W JP 2011054358W WO 2011105568 A1 WO2011105568 A1 WO 2011105568A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- plant tissue
- extract
- weight
- nobiletin
- disease
- Prior art date
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/75—Rutaceae (Rue family)
- A61K36/752—Citrus, e.g. lime, orange or lemon
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/35—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom
- A61K31/352—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom condensed with carbocyclic rings, e.g. methantheline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7048—Compounds having saccharide radicals and heterocyclic rings having oxygen as a ring hetero atom, e.g. leucoglucosan, hesperidin, erythromycin, nystatin, digitoxin or digoxin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- the present invention relates to a dried plant tissue and a plant tissue extract that improve central neurodegenerative diseases associated with learning / memory disorders and movement disorders.
- the present invention also relates to pharmaceuticals and foods containing the dried plant tissue or plant tissue extract.
- Non-Patent Document 1 N-methyl-D-aspartate (NMDA) receptor function, which is important for learning / memory Neurodegeneration due to polymerization and accumulation of amyloid ⁇ peptide is considered (see Non-Patent Document 1).
- NMDA N-methyl-D-aspartate
- memory In general, memory consists of three processes: memory acquisition, retention, and recall, and the mechanisms of these three processes are considered different. It is understood that learning and memory impairment, which is a core symptom of Alzheimer's disease, is particularly markedly impaired in memory retention and recall processes.
- the formula (I) See Patent Document 1 or Non-Patent Document 1 or 2), and nobiletin is also known to have a neurite outgrowth action on nerve cells (Patent Document 1). 2).
- Nobiletin is contained in the skin of various citrus plants, but is usually contained in a very small amount.
- Chen which is one of the herbal medicines derived from the peels of citrus plants, usually exhibits a gastric action, expectorant action, antitussive action, etc., and it is considered that nobiletin is not involved in these actions.
- Parkinson's disease one of the central neurodegenerative diseases, is characterized by four major motor signs such as tremor, slow movement, muscle rigidity, and postural reflex disorder, and the dopaminergic nervous system degenerates due to some factor. This is because of motor dysfunction.
- dopamine replacement therapy is currently performed. Many drugs that increase the transcriptional activity of the enzyme in the dopamine biosynthetic pathway, that is, the expression level of its mRNA, are known, but few drugs that increase the production of dopamine itself are known.
- a dried citrus plant tissue exhibiting a dopamine synthesis ability promoting action and a dopamine secretion promoting action and an extract thereof are not known.
- the present inventors have found a dried citrus plant tissue and a plant tissue extract that have a remarkable effect of improving central nervous degenerative diseases.
- the present inventors have found dried plant tissues and plant tissue extracts of citrus fruits that can be applied as a fundamental therapeutic agent for Alzheimer's disease and Parkinson's disease among central neurodegenerative diseases.
- the inventors have further discovered dried citrus plant tissues and plant tissue extracts that enhance not only memory recall but also memory acquisition and retention in memory impairment, a core symptom of Alzheimer's disease.
- the present invention includes the following embodiments: (1) Dry plant tissue of citrus peel for improving central neurodegenerative diseases, containing 0.4% by weight or more of nobiletin with respect to 100% by weight of dry plant tissue; (2) Dry plant tissue of citrus leaves for improving central neurodegenerative diseases, containing 0.3% by weight or more of nobiletin with respect to 100% by weight of dry plant tissue; (3) The dry plant tissue according to any one of (1) and (2), wherein the weight ratio of nobiletin / nariltin is 1.0 or more; (4) The dried plant tissue according to any one of (1) and (2), wherein the central neurodegenerative disease is Alzheimer's disease and / or Parkinson's disease; (5) Improvement of central neurodegenerative diseases is increased cAMP response element (CRE) -dependent transcriptional activity, memory acquisition, retention and enhanced recall ability, tyrosine hydroxylase (TH) transcriptional activity, TH expression level The dried plant tissue according to any one of (1) and (2), which is brought about by an increase, promotion of dopamine synthesis ability, or promotion
- Citrus plant tissue extract for improving central neurodegenerative diseases containing 0.6% by weight or more of nobiletin with respect to 100% by weight of plant tissue extract; (7) The plant tissue extract according to (6), wherein the content weight ratio of nobiletin / nariltin is 2.0 or more; (8) The plant tissue extract according to (6) obtained by water extraction from the dried plant tissue according to any of (1) or (2), preferably 60 to 100 ° C water extraction; (9) The plant tissue extract according to (6), wherein the central neurodegenerative disease is Alzheimer's disease and / or Parkinson's disease; (10) Improvement of central neurodegenerative diseases may increase CRE-dependent transcriptional activity, acquire memory, enhance retention and recall ability, promote TH transcriptional activity, increase TH expression, promote dopamine synthesis, or dopamine secretion The plant tissue extract according to (6), which is caused by the promotion of
- 10% to 100% by weight of the dry plant tissue according to (1) or (2) or 10% to 90% by weight of the plant tissue extract according to (6) is effective with respect to 100% by weight of the pharmaceutical product A drug for treating Alzheimer's disease as an ingredient
- 10% to 100% by weight of the dry plant tissue according to (1) or (2) or 10% to 90% by weight of the plant tissue extract according to (6) is effective with respect to 100% by weight of the pharmaceutical product A drug for treating Parkinson's disease as an ingredient
- a therapeutic drug for treating a central neurodegenerative disease preferably comprising an extract obtained by water extraction at 60 to 100 ° C.
- the content of nobiletin is 0.3 to 2.0% by weight with respect to 100% by weight of the dry plant tissue, and the content weight ratio of nobiletin / nariltin is 1.0 to 23.0.
- Dry plant tissue of (24) The dried plant tissue according to (4), which is obtained by heating and drying citrus plant tissue at 50 to 100 ° C. for 1 to 3 hours, and the yield is 20 to 50%; (25) The content of nobiletin is 0.6 to 3.0% by weight with respect to 100% by weight of the plant tissue extract, and the content weight ratio of nobiletin / nariltin is 2.0 to 14.0.
- Plant tissue extracts as described; (26) The pharmaceutical according to (19), which is in the form of a fine granule, tea, decoction, capsule, tablet, granule, jelly, powder, liquid, syrup or extract preparation.
- the citrus dried plant tissue and plant tissue extract of the present invention show a significant central neurodegenerative disease improving effect as compared with the conventional citrus dried plant tissue.
- the dried plant tissue and plant tissue extract of the present invention also have the effect of activating PKA / ERK / CREB signaling and improving learning / memory impairment, which is one of the symptoms of Alzheimer's disease.
- the dried plant tissue and plant tissue extract of the present invention particularly show the effect of acquiring, maintaining and recalling memory among the learning / memory impairment improving effects.
- the dried plant tissue and plant tissue extract of the present invention further have a strong phosphorylation promoting effect and a PKA / ERK signal transduction promoting effect as compared with nobiletin alone having the same amount of nobiletin contained therein.
- the dried plant tissue and plant tissue extract of the present invention exhibit an improvement effect on Parkinson's disease by CRE transcription activity and TH transcription activity, TH expression level increasing action, dopamine synthesis ability promoting action or dopamine secretion promoting action. .
- the dried plant tissue and plant tissue extract of the present invention further has a central neurodegenerative disease ameliorating effect exceeding that predicted from its nobiletin-containing concentration, in particular, an effect of increasing CRE-dependent transcriptional activity, memory acquisition, retention and recall ability.
- a central neurodegenerative disease ameliorating effect exceeding that predicted from its nobiletin-containing concentration in particular, an effect of increasing CRE-dependent transcriptional activity, memory acquisition, retention and recall ability.
- the effect of promoting TH transcriptional activity and the effect of increasing the TH expression level the effect of promoting dopamine biosynthesis and the effect of promoting dopamine secretion are shown. These effects are considered to be due to a synergistic effect of nobiletin and other components contained in the dried plant tissue and plant tissue extract.
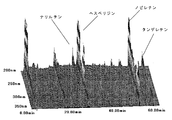
- FIG. 1A shows a three-dimensional chromatogram of flavonoids contained in the skin of the present invention.
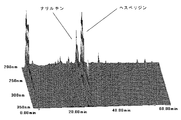
- FIG. 1B shows a three-dimensional chromatogram of flavonoids contained in a conventional skin.
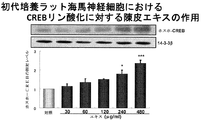
- FIG. 2A shows CREB phosphorylation promoting activity of a skin extract in hippocampal neurons.
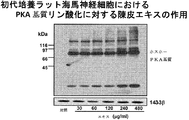
- FIG. 2B shows the PKA substrate phosphorylation promoting activity of the skin extract in hippocampal neurons.
- FIG. 2C shows the ERK1 / 2 phosphorylation promoting activity of the skin extract in hippocampal neurons.
- FIG. 2D shows the PKA signal transduction promoting effect of the skin extract in hippocampal neurons.
- FIG. 2E shows the ERK signal transduction promoting effect of the skin extract in hippocampal neurons.
- FIG. 1A shows CREB phosphorylation promoting activity of a skin extract in hippocampal neurons.
- FIG. 2B shows the PKA substrate phosphorylation promoting activity of the skin extract in hippocampal
- FIG. 2F shows the CRE-dependent transcriptional activity of extracts from the present skin 1 and conventional skin 7 and nobiletin in hippocampal neurons.
- FIG. 2G shows the CRE-dependent transcriptional activity of extracts from the skin 2-4 of the present invention and the conventional skin 8-10 and nobiletin in hippocampal neurons.
- FIG. 3A shows the effect of improving the learning / memory impairment of crustacean extract, that is, the effect of acquiring, retaining, and recalling memory.
- FIG. 3B shows the effect of nobiletin on MK801-induced learning / memory impairment.
- FIG. 4A shows the TH transcriptional activity promoting effect of the skin extract in PC12D cells.
- FIG. 4B shows the effect of increasing the TH expression level of the crust extract in PC12D cells.
- FIG. 4C shows the effect of increasing the dopamine content of the skin extract in PC12D cells.
- FIG. 5A shows CRE transcriptional activity of Tachibana peel extract in hippocampal neurons.
- FIG. 5B shows the improvement effect of learning impairment of Tachibana peel extract.
- Two-way ANOVA including post Bonferroni test for all pairs of learning-promoting effects. Numerical values in the figure show mean ⁇ standard error; n 5 for all tests. ** p ⁇ 0.01 compared to control ( ⁇ ), *** p ⁇ 0.001, ### p ⁇ 0.01 compared to MK-801 ( ⁇ ), ⁇ p ⁇ 0.05.
- FIG. 5C shows the effect of improving the memory impairment of Tachibana peel extract.
- One-way ANOVA including post Tukey test for all pairs of memory improvement effects.
- FIG. 5D shows CRE transcriptional activity of Tachibana peel extract in PC12D cells.
- FIG. 5E shows TH transcriptional activity of Tachibana peel extract in PC12D cells.
- FIG. 5F shows the GCH I expression level increasing effect of Tachibana bark extract in PC12D cells.
- FIG. 6A shows CRE transcriptional activity of the extract derived from the leaf of Tachibana in hippocampal neurons.
- FIG. 6B shows CRE transcriptional activity of Tachibana leaf-derived extract in PC12D cells.
- FIG. 7A shows the CRE transcriptional activity of the extract derived from citrus pericarp in hippocampal neurons.
- FIG. 7B shows the CRE transcriptional activity of the extract derived from the citrus pericarp in PC12D cells.
- FIG. 7C shows the TH transcriptional activity of the extract derived from the citrus pericarp in PC12D cells.
- the citrus fruits used in the present invention are: Citrus tachibana, C. nipponokoreana, Hanayu, Shikitsu, Pheasant, Daidai, Mediterranean Mandarin, Dancy Tangerine, Obenimikan, Kobenimikan, Nucleus Kishu, Fukurimuta, Ponkan, Hirakishu, Sankitsu, Cleopatra, Koji, Giri Mikan, Ichan Lemon, Unshu Mikan (Citrus unshiu Markovich), Citrus reticulata Blanco (Rutaceae), Sikhwasa, Yuzu, Bungtan, Hyuganatsu, Ponkan, Natsumikan, Bunyo Among plants selected from the group consisting of those having a high nobiletin content, preferably Citrus reticulata Blanco (Rutaceae), Japanese tachibana (Citrus tachibana), Kourai tachibana (C. nip ponokoreana) or Gi
- the yield of the citrus peel or leaf is 20 to 50% in the case of the peel and 20 to 50% in the case of the leaf. It means a substance dried under such conditions.
- the heating and drying conditions include a temperature of 50 to 100 ° C. for 1 to 3 hours, and a temperature of 60 ° C. for 2 hours.
- Preferred dry plant tissues of the present invention are Chen, Tachibana and Tachibana leaves.
- Tachibana peel refers to the mature pericarp of Japanese Tachibana or Tachibana, particularly Tachibana.
- the leaf means a leaf of Tachibana such as Japanese Tachibana or Kourai Tachibana.
- the plant tissue extract of the present invention is an extract from the above-mentioned dry plant tissue, preferably an extract obtained by water extraction.
- the preferred water extraction herein is extraction with water at 60-100 ° C.
- components contained in the dried plant tissue and plant tissue extract of the present invention include flavonoids such as nobiletin, nariltin, tangeretin, and hesperidin.
- flavonoids such as nobiletin, nariltin, tangeretin, and hesperidin.
- the activity of the dried plant tissue and plant tissue extract of the present invention is remarkable as compared with the medicinal effect of nobiletin alone due to the synergistic effect due to the content and content ratio of the components contained therein.
- the dry plant tissue of the present invention comprises 0.3 or 0.4% by weight or more, preferably 0.3 to 2.0% by weight, more preferably 0.4 to 2.0% by weight of nobiletin based on the total weight. 0.7% by weight or less, preferably 0.4% by weight or less of nariltin, 0.1 to 0.8% by weight of tangeretin and 0.4 to 12% by weight of hesperidin.
- the dry plant tissue of the present invention also has a nobiletin / nariltin content weight ratio of 1.0 or more, preferably 1.5 to 23.0, more preferably 2.0 to 15.0, still more preferably 5.0 to 15.0.
- the dry plant tissue of the present invention can be derived from any plant as long as the proportion of each component contained in the dry tissue is within the above range, that is, the drug effect of the present invention, that is, the central neurodegenerative disease improving effect (anti Alzheimer's disease activity, anti-Parkinson disease activity, etc., learning / memory impairment improvement effect, phosphorylation promotion effect and PKA / ERK signaling promotion effect, CRE transcription activity and TH transcription activity, TH expression level increase effect, dopamine synthesis ability promotion effect Or show dopamine secretion promoting action.
- the drug effect of the present invention that is, the central neurodegenerative disease improving effect (anti Alzheimer's disease activity, anti-Parkinson disease activity, etc., learning / memory impairment improvement effect, phosphorylation promotion effect and PKA / ERK signaling promotion effect, CRE transcription activity and TH transcription activity, TH expression level increase effect, dopamine synthesis ability promotion effect Or show dopamine secretion promoting action.
- the plant tissue extract of the present invention is 0.6 to 2.0% by weight or more, preferably 0.6 to 3.0% by weight of nobiletin, 0.7% by weight or less, preferably 0. Contains no more than 4% by weight nariltin, 0.1-1.0% by weight tangeretin and 1.8-6.0% by weight hesperidin.
- the plant tissue extract of the present invention also has a nobiletin / nariltin content weight ratio of 2.0 or more, preferably 2.0 to 14.0, more preferably 2.5 to 10.0.
- the plant tissue extract of the present invention can be derived from any plant as long as the ratio of each component contained therein is within the above-mentioned range.
- central neurodegenerative disease includes Alzheimer's disease, particularly learning / memory impairment, and Parkinson's disease.
- Examples of the anti-Alzheimer's disease activity and / or anti-Parkinson's disease activity of the dried plant tissue and plant tissue extract of the present invention include improvement of learning / memory impairment related to the onset mechanism of central neurodegenerative diseases or promotion of dopamine synthesis. .
- the dried plant tissue and plant tissue extract of the present invention may exhibit different activities depending on the type of plant tissue.
- the skin or skin extract may exhibit a dopamine content increasing activity
- the tachibana skin or citrus skin extract may exhibit a dopamine secretion promoting activity.
- learning / memory failure is a failure that reduces the following three abilities in the memory process: memory acquisition (encoding), retention (retention), and recall (recall) abilities.
- acquisition refers to capturing information into memory
- retention refers to storing captured information
- recalling refers to recalling stored information.
- therapeutic agents for central neurodegenerative diseases include anti-Alzheimer's disease agents and / or anti-Parkinson's disease agents, that is, pharmaceuticals having anti-Alzheimer's disease activity and / or anti-Parkinson disease activity.
- the term “pharmaceutical” means a substance for diagnosis, treatment or prevention of diseases in humans and animals, and includes pharmaceuticals including herbal medicines, such as herbal medicines and herbal medicines.
- a preferred pharmaceutical is an oral dosage form, more preferably a fine granule, tea, decoction, capsule, tablet, granule, jelly, powder, liquid, syrup or extract.
- the term “herbal medicine” is a general term for substances used for improving the constitution of a subject without purifying active ingredients from naturally occurring products having medicinal effects, and includes one or more herbal ingredients.
- the term “Chinese medicine” means a medicine prescribed on the basis of Chinese medicine theory.
- the term “food” refers to so-called health foods and health functional foods such as foods for specified health use and functional nutritional foods that are distinguished by the presence or absence of permission from the authorities and the purpose and function of the food. Including. Preferred foods herein are fine granules, jelly sauce, peel, jam or tea, more preferably fine granules or tea.
- the blending amount of each component in the pharmaceutical and food in the present specification can be appropriately adjusted according to the intended purpose of use, sex, symptoms, etc., for example, 10 to 100% by weight of the dried plant tissue of the present invention or plant 10-90% by weight of tissue extract and 0-90% by weight of other carriers or additives.
- the daily intake amount of the dried plant tissue or plant tissue extract of the present invention per adult is preferably 3 to 50 g or 1 to 20 g, and more preferably 5 to 30 g or 2 to 10 g.
- compositions herein include those commonly used in the art, such as pharmaceutically or food hygienically acceptable herbal ingredients, ascorbic acid, aspartame, apple flavor, orange flavor, carrageenan. , Caramel, carnauba wax, carmellose, carmellose calcium, reduced maltose liquid sugar, reduced maltose starch syrup, hydrous silicon dioxide, xylitol, citric acid, trisodium citrate, granulated sugar, light anhydrous silicic acid, gelling agent (FG-2266, Nitta Gelatin Co., Ltd.), synthetic aluminum silicate, hydroxypropyl starch, crystalline cellulose, beeswax, titanium oxide, salt, sucrose fatty acid ester, magnesium stearate, cellulose, talc, dextrin, corn starch, lactose, honey, hydro Xylpropylmethylcellulose, fine silicon dioxide, pullulan, pectin, maltose, magnesium aluminate metasilicate,
- the pharmacologically or food hygienically acceptable herbal medicine components in this specification include those commonly used in the art.
- Chen 1 to 6, Tachibana 1 to 6, Tachibana leaves 1 to 3 and Obenimikan 1 are the dry plant tissue of the present invention, and Chen 7 to 12 are conventional.
- the plant tissue of the same degree can be obtained from Kotaro Hanpo Pharmaceutical Co., Ltd. from the viewpoint of the samples and the components used in the following examples.
- Example 1 Production of dried plant tissue
- the peels and leaves collected from various citrus fruits were shade-dried, sun-dried or heat-dried to obtain dried bodies of the peel, tachibana peels, tachibana leaves, and the fruit of periwinkle.
- Heat drying is carried out until the yield (ratio of mass after drying / mass before drying) is 20-50% for Chen and Tachibana peels, 20-50% for Tachibana leaves, and 20-50% for Prunus pericarp. Specifically, it was dried by heating at a temperature of 60 ° C. for 2 hours.
- Example 2 Production of plant tissue extract 400 mL of pure water was added to about 10 g of the cut skin, tachibana bark, leaves of tachibana or persimmon bark obtained in Example 1 and heated (tachibana bark) For 6), 1000 mL of pure water was added to about 20 g of the minced and heated. After the mixture boiled, it was extracted at 100 ° C. for 1 hour. Subsequently, it filtered through gauze (2 sheets), and the filtrate was freeze-dried to obtain a plant tissue extract. The production results of the plant tissue extract are shown in Table 1 below.
- Example 3 Analysis of contained components The contained components of the dried plant tissues and plant tissue extracts obtained in Examples 1 and 2 were measured as follows. Below, the analysis method about a crust and a crust extract is shown.
- a standard solution used for quantification of nobiletin and tangeretin was prepared as follows. Nobiletin (Wako Pure Chemical Industries, Ltd.) was dried with a desiccator (silica gel) for 24 hours or more. About 5 mg of dried nobiletin was dissolved in a methanol / water mixture (7: 3) to make 100 mL, and used as a nobiletin standard solution. Further, tangeretin (Wako Pure Chemical Industries, Ltd.) was dried with a desiccator (silica gel) for 24 hours or more. About 3 mg of dry tangeretin was dissolved in a methanol / water mixture (7: 3) to make 100 mL, and used as a tangeretin standard solution.
- a standard solution used for quantification of hesperidin and nariltin was prepared as follows. Hesperidin (Wako Pure Chemical Industries, Ltd.) was dried with a desiccator (silica gel) for 24 hours or more. About 10 mg of dry hesperidin was dissolved in 50% methanol to make 50 mL, and used as a hesperidin standard solution. Also, Nariltin (Wako Pure Chemical Industries, Ltd.) was dried with a desiccator (silica gel) for 24 hours or more. About 10 mg of dry narilutin was dissolved in 50% methanol to make 500 mL, which was used as a narilutin standard solution.
- a skin sample solution As a skin sample solution, 30 mL of methanol was added to about 0.1 g of skin powder. The mixture was irradiated with ultrasonic waves for 20 minutes (UT-305HS, Sharp Corporation), extracted, and then centrifuged (KUBOTA KN-70, Kubota Corporation) to collect the supernatant. 20 mL of methanol was added to the residue, and extraction was performed by irradiating with ultrasonic waves for 20 minutes, followed by centrifugation. This was combined with the collected supernatant to obtain a skin sample solution.
- the peel extract As a sample solution of the peel extract, about 0.1 g of the peel extract was placed in a 50 mL stoppered centrifugal sedimentation tube, and 50 mL of a methanol / water mixture (1: 1) was added thereto. The mixture was extracted with ultrasonic waves (UT-305HS, Sharp Corporation) and then centrifuged (KUBOTA KN-70, Kubota Corporation). Then, it filtered and it was set as the sample solution of the crust extract.
- ultrasonic waves UT-305HS, Sharp Corporation
- KUBOTA KN-70 centrifuged
- FIGS. 1A and 1B show three-dimensional chromatograms of flavonoids contained in the skin of the present invention and the conventional skin, respectively.
- each component may vary depending on the sampling location and timing of the citrus plants used.
- the blue peel of tachibana has some nobiletin content of about 1.60% by weight, but about 1.1% by weight of yellow peel.
- Test Example 1 The general test procedure used in Test Example 1 below is as follows. (Culture of rat fetal primary hippocampal neurons) Pregnant Sprague-Dawley (SD) rats were bred by feeding and watering in a 12-hour light / dark cycle. The uterus was aseptically removed from the rat (E18) on day 18 of gestation by a midline abdominal incision under deep ether anesthesia. Under a stereomicroscope, remove the fetal hippocampus in ice-cold phosphate buffered saline (PBS), disperse the tissue with papain enzyme (SUMILON), and centrifuge at 1000 rpm for 4 minutes. Removed.
- PBS ice-cold phosphate buffered saline
- SUMILON ice-cold phosphate buffered saline
- the cell pellet was dispersed in a dispersion liquid (SUMILON), and then the removal liquid (SUMILON) was added to the cells sufficiently dispersed by pipetting, followed by centrifugation at 900 rpm for 5 minutes, and then the supernatant was removed. .
- the pellet is suspended using Neurobasal Medium (Neurobasal Medium 500 mL / Phenol Red-free, 50 x B27 Supplement 10 ml, 0.5 mM L-glutamine, 0.005% penicillin-streptomycin) and added with poly-L-lysine. Seeded on coated dishes or plates. One day after the culture, the medium was changed, and then half of the medium was changed every 3 to 4 days.
- Neurobasal Medium Neurobasal Medium 500 mL / Phenol Red-free, 50 x B27 Supplement 10 ml, 0.5 mM L-glutamine, 0.005% penicillin-streptomycin
- the medium was cultured in a medium containing 10 ⁇ M AraC at 37 ° C. in a 5% CO 2 incubator for 14 days.
- Neurobasal Medium containing 10 ⁇ M AraC was used as a test medium for drug treatment experiments.
- Rat fetal primary hippocampal neurons were seeded at 1 ⁇ 10 6 cells / dish in a 35 mm dish, cultured in a growth medium for 14 days in a 5% CO 2 incubator, and then treated with the drug for 10 minutes. Each cell was treated with drugs and then washed with ice-cold PBS.
- Cell lysate (1 mM EDTA, 1% SDS, 10 mM NaF, 10 nM caliculin, 320 nM okadaic acid, 1 mM sodium orthovanadate, 1 mM p-APMSF, 10 ⁇ g / mL pepstatin A, 10 ⁇ g / mL Antipine, 10 ⁇ g / mL leupeptin, 10 ⁇ g / mL chymostatin, 10 ⁇ g / mL phosphoramidon, 10 mM HEPES, pH 7.5). Thereafter, it was immediately boiled at 95 ° C. for 5 minutes to denature the protein, and then the DNA was sonicated to prepare a sample for SDS-PAGE.
- the PVDF membrane is placed in a stripping buffer (62.5 mm Tris-HCl, 2% SDS, 100 mm mM ⁇ -mercaptoethanol, pH 7.4) pre-warmed to 50 ° C and incubated for 30 minutes at 50 ° C. Was removed and washed with TBST. Thereafter, the internal standard protein was detected by reprobing.
- a stripping buffer (62.5 mm Tris-HCl, 2% SDS, 100 mm mM ⁇ -mercaptoethanol, pH 7.4) pre-warmed to 50 ° C and incubated for 30 minutes at 50 ° C. Was removed and washed with TBST. Thereafter, the internal standard protein was detected by reprobing.
- 7.2 g of the skin extract from the skin 1 was weighed and placed in a 50 ml centrifuge tube, and dimethyl sulfoxide (DMSO) was added thereto to make 30 mL. Then, it was sonicated for 30 minutes and centrifuged at 3000 rpm for 10 minutes, and the supernatant was collected and used as a skin extract.
- DMSO dimethyl sulfoxide
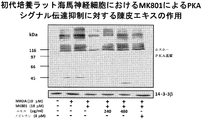
- Extracellular signal-regulated kinase (ERK) 1/2 phosphorylation-promoting effect of skin extract in primary cultured hippocampal neurons About the effect of various concentrations of skin extract or nobiletin on ERK1 / 2 phosphorylation Blotting was used (contains nobiletin 1 ⁇ M in 60 ⁇ g / ml of skin extract). E18 rat hippocampal neurons grown at a density of 1 ⁇ 10 6 cells / 35-mm plastic dish were cultured for 14 days. Cells were treated for 10 minutes with crushed extract (30, 60, 120, 240 and 480 ⁇ g / ml) or nobiletin (4, 8 and 30 ⁇ M).
- crushed extract (30, 60, 120, 240 and 480 ⁇ g / ml) or nobiletin (4, 8 and 30 ⁇ M).
- rat hippocampal neurons were primarily cultured and then a reporter gene assay was performed.
- Hippocampal neurons were seeded in a 48-well plate at 8 ⁇ 10 4 cells / well and cultured in Neurobasal medium for 10-14 days.
- Reporter plasmid (0.1 ⁇ g / well) and Renilla pRG-TK plasmid (0.01 ⁇ g / well) were transfected by lipofection and cultured for 16 hours. It was diluted with Neurobasal medium without AraC (containing B-27 Supplement, L-glutamine, penicillin-streptomycin) and treated with a crust extract at a concentration of 300 ⁇ g / ml for 8 hours.
- the skin extract was a 150 mg / ml DMSO solution as a stock solution, dispensed, and stored at -20 ° C.
- Transcriptional activity was measured using Promega's Dual-Luciferase (registered trademark) Reporter Assay System. Hippocampal neurons were solubilized with Passive Lysis Buffer (Promega), then mixed with Luciferase Assay Regent II (Promega) and Stop & Glo (registered trademark) Reagent (Promega), and the fluorescence value was measured with a luminometer.
- the reporter gene assay and RT-PCR results were evaluated using one-way ANOVA (Tukey). The significance level was tested at 5% on both sides, and p ⁇ 0.05 was considered significant.
- the concentration of nobiletin in the skin extract used in this test example is 2.5 ⁇ M, and its CRE-dependent transcriptional activity is 1 / 12th that of 30 ⁇ M nobiletin. More than 30 ⁇ M nobiletin. That is, it was found that in the multi-component system of the skin extract of the present invention, the presence of components other than nobiletin significantly and synergistically increases the CRE-dependent transcription promoting activity of nobiletin than the activity expected from nobiletin alone. In addition, the skin extract of the present invention showed a CRE-dependent transcriptional activity stronger than that of conventional skin extracts, so it showed an action to improve learning and memory impairment and more effectively improved the recognition function of Alzheimer's disease. It has been shown.
- mice were divided into 4 groups, each with a skin extract of 1.48 g / kg or 3.69 g / kg (nobiletin content: 10 mg / kg or 25 mg / kg, respectively), and 2 groups with physiological saline for 7 consecutive days Orally. On the 7th day, 90 minutes after administration of these, MK801 (0.08 mg / kg) or physiological saline was administered subcutaneously. Thirty minutes later, a fear conditioning learning test was conducted. In the horror learning test, mice were placed in a transparent box and allowed to explore freely for 2 minutes, followed by 0.7 mA, 2 s electrical stimulation.
- Comparative Test 1 Examination of learning / memory impairment improving effect of nobiletin
- Comparative Example 1-1 a comparative test was conducted by replacing oral administration of crust extract with intraperitoneal administration of nobiletin (10, 50 mg / kg). It was. The results are shown in FIG. 3B.
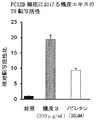
- the freezing action was significantly reduced by the treatment with MK801.
- the administration of the crustacean extract showed an improvement effect on the reduction in a dose-dependent manner in both the learning trial and the confirmation trial. It was.
- the mice administered with the skin extract of the present invention received the MK801 treatment, and the learning / memory function was hindered and easily forgotten.
- the ratio was about 1.5 times that of the control mouse and about 4.4 times that of the MK801-treated mouse skin extract-untreated mouse (FIG. 3A).
- Such a result was not recognized by administration of nobiletin (Comparative Test 1, FIG. 3B). That is, it has been found that the skin extract of the present invention has an effect of acquiring memory and enhancing retention ability, which is not recognized by administration of nobiletin alone.
- Test Example 4 The general test procedure used in Test Example 4 below is as follows. (PC12D cell culture) Dulbecco's modified Eagle medium (DMEM) (9.5 g) was dissolved in MilliQ water (800 mL), and glucose (3.5 g) was added thereto to make 1 L with MilliQ water. 830 mL of the product sterilized by autoclave was taken, and 17 mL of 10% aqueous sodium hydrogen carbonate solution and 16.5 mL of 3% L-glutamine aqueous solution were added. Next, equine serum (HS) to 10% and fetal calf serum (FCS) to 5% were prepared as growth media and cultured in a 5% CO 2 incubator at 37 ° C. . The test medium used was hDMEM containing 2% HS and 1% FCS.
- DMEM Dulbecco's modified Eagle medium
- PC12D cells were seeded on a 48-well plate at 8 ⁇ 10 4 cells / well and cultured in a growth medium for 24 hours, followed by lipofection with 0.1 ⁇ g / well of pRTH plasmid and 0.01 ⁇ g / well of Renilla pRG-TK plasmid.
- the medium was replaced with a test medium in which the compound was dissolved 19 hours after transfection. After incubation for 24 hours, the medium was aspirated and collected with Passive Lysis Buffer. Firefly shiitake and Renilla luciferase were measured with a luminometer using Dual-Luciferase Reporter Assay System (Promega).
- PC12D cells were seeded at 0.67 ⁇ 10 6 cells / dish in a 35 mm dish, cultured in a growth medium for 2 days in a 5% CO 2 incubator, and then treated with the drug for 24 hours. Each cell was treated and then washed with ice-cold PBS.
- Cell lysate (1 mM EDTA, 1% SDS, 10 mM NaF, 10 nM caliculin, 320 nM okadaic acid, 1 mM sodium orthovanadate, 1 mM p-APMSF, 10 ⁇ g / mL pepstatin A, 10 ⁇ g / mL Antipine, 10 ⁇ g / mL leupeptin, 10 ⁇ g / mL chymostatin, 10 ⁇ g / mL phosphoramidon, 10 mM HEPES, pH 7.5). Thereafter, the mixture was immediately boiled at 95 ° C. for 5 minutes to denature the protein, and then the DNA was sonicated and the centrifuged supernatant was prepared as a sample for SDS-PAGE.
- TBST containing 5% skim milk (10 mm Tris-HCl, 100 mm mM NaCl) , 0.05% Tween-20, pH 7.4; hereinafter referred to as blocking buffer) for 1 hour at room temperature. Samples were then washed with TBST and incubated overnight at 4 ° C. with primary antibody diluted 1000-fold with blocking buffer. Further, the sample was washed with TBST, incubated with an HRP-labeled IgG antibody diluted 2000 times with a blocking buffer at room temperature for 1 hour, and washed with TBST.
- the ECL method was used to detect antibody-positive bands.
- the antibody was peeled off using a stripping buffer (62.5 mM Tris-HCl, SDS 2% SDS, 100 mM mM ⁇ -mercaptoethanol, pH 7.4), the blot was washed with TBST, and the internal standard protein was detected.
- PC12D cells were seeded at 1.0 ⁇ 10 6 cells / dish in a 35 mm dish and cultured in a 5% CO 2 incubator on a growth medium for 24 hours, and then the cell culture solution supplemented with skin 1 and skin 7 was further added for 24 hours. Cultured.
- a low K + buffer 140 mM NaCl, 4.7 mM KCl, 1.2 mM KH 2 PO 4 , 2.5 mM CaCl 2 , 1.2 mM MgSO 4 , 11 mM glucose and 15 mM HEPES-Tris, pH 7 heated to 37 ° C.
- the cells were washed in .4), a perchloric acid solution was added so that the final concentration was 0.4 N, and the cells were disrupted with ultrasonic waves for 1 minute.
- the obtained solution was centrifuged, and dopamine was separated and quantified by high performance liquid chromatography using an electrochemical detector.
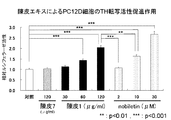
- the skin 1 extract of the present invention increased the dopamine content at both 24 and 48 hours treatment compared to the control and conventional skin 7 extract.
- the skin extract of the present invention increases the TH transcriptional activity, TH expression level and dopamine content in PC12D cells in a concentration-dependent manner, and its action is unexpected and exceptional compared to nobiletin at an equal concentration. It was powerful. That is, it was revealed that the skin extract of the present invention exhibits a dopamine synthesis promoting activity more markedly than nobiletin. Since the activity is not recognized in the conventional extract derived from dermis, it is considered that the dermis extract of the present invention exhibited a dopamine synthesis promoting activity due to a synergistic effect of a high content of nobiletin and other components other than nobiletin. As described above, the skin extract of the present invention is expected to have a medicinal effect as a therapeutic agent for Parkinson's disease.
- Test Example 5 CRE Transcriptional Activity of Tachibana Skin Extract in Hippocampal Nerve Cells Using the extract extracted from Tachibana Skin 3 obtained in Example 2 by the method described herein, CRE transcriptional activity in hippocampal neurons is controlled And compared with nobiletin.
- the test procedure was the same as in Test Example 2. The result is shown in FIG. 5A.
- the Tachibana peel 3 extract 300 ⁇ g / mL used in this test example contained about 9.4 ⁇ M nobiletin.
- the extract of Tachibana peel 3 showed significant CRE transcriptional activity in hippocampal neurons compared to control and nobiletin. From this, the tachibana peel extract of the present invention is expected to have a medicinal effect as a therapeutic agent for Alzheimer's disease.
- Tachibana peel extract learning / memory impairment improving effect (in vivo test) Using the extract extracted from Tachibana bark 6 obtained in Example 2 by the method described in this specification, the effect of the Tachibana bark extract of the present invention on learning / memory impairment was examined. The test procedure was the same as in Test Example 3. The results are shown in FIGS. 5B and 5C. Tachibana peel extract 1.87 g / kg or 3.73 g / kg (nobiletin content: 25 mg / kg or 50 mg / kg, respectively) was orally administered to each group continuously for 7 days. As shown in FIG. 5B and FIG. 5C, in addition to significantly improving learning / memory impairment by chronic administration of the tachibana peel extract of the present invention, it is possible to enhance the ability to acquire and retain memory that is not observed with nobiletin. It became clear.
- the tachibana peel extract of the present invention is expected to have a medicinal effect as a therapeutic agent for Alzheimer's disease.
- Test Example 8 TH transcription activity of Tachibana peel extract in PC12D cells Using the extract extracted from Tachibana peel 3 obtained in Example 2 by the method described in this specification, the TH transcription activity in PC12D cells was compared with control and nobiletin. And compared. The test procedure was the same as in Test Example 4-1. The result is shown in FIG. 5E. As shown in FIG. 5E, Tachibana peel 3 extract showed significant TH transcriptional activity in PC12D cells compared to control and nobiletin.
- GTP cyclohydrolase I (GCH I) level-increasing action of Tachibana bark extract in PC12D cells
- GPC I level-increasing action was examined in PC12D cells.
- GCH I is a rate-limiting enzyme in the biosynthesis of the coenzyme tetrahydrobiopterin (BH 4 ) essential for TH.
- PC12D cells were treated with Tachibana peel extract, Chen peel extract and DMSO (control) at a concentration of 120 ⁇ g / mL for 48 hours, washed with PBS, and cell lysate (1 mM EDTA, 1% SDS, 10 mM HEPES, 10 mM NaF) , 1 mM p-APMSF, 1 mM sodium orthovanadate, 10 nM calyculin, 320 ⁇ M okadaic acid, 10 ⁇ g / mL pepstatin, 10 ⁇ g / mL antipine, 10 ⁇ g / mL leupeptin, 10 ⁇ g / mL chymostatin, 10 ⁇ g / mL phosphoramidon, 240 pM cypermethrin).
- the Tachibana peel extract 120 ⁇ g / mL used in this test example contained about 3.76 ⁇ M nobiletin, and the cuticle extract 120 ⁇ g / mL contained about 2.02 ⁇ M nobiletin.
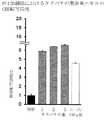
- Tachibana peel extract and Chen peel extract significantly increased the expression level of GCH I compared to the control.
- Tachibana peel extract significantly increased the expression level of GCH I compared to Chen peel extract.
- Coenzyme BH4 which is essential for the expression of TH activity, increases the amount of TH protein, increases the TH enzyme activity, and promotes dopamine synthesis when the intracellular BH4 content is higher than the amount indicated by the Michaelis constant. .
- the Tachibana peel extract of the present invention significantly increases the expression level of GCH I, and therefore increases the amount of GCH I, which is the rate-determining enzyme of coenzyme BH4 synthesis that is essential for the expression of TH activity.
- GCH I is the rate-determining enzyme of coenzyme BH4 synthesis that is essential for the expression of TH activity.
- TH activity is increased, and as a result, production and secretion of dopamine are presumed to be promoted, and a medicinal effect as a Parkinson's disease therapeutic agent is expected.
- Test Example 12 CRE Transcriptional Activity of Citrus Citrus Skin Extract in Hippocampal Nerve Cells Using the extract extracted from Citrus mandarin 1 obtained in Example 2 by the method described herein, CRE transcriptional activity in hippocampal neurons is controlled And compared with nobiletin. The test procedure was the same as in Test Example 2. The results are shown in FIG. 7A. In addition, 100 ⁇ g / mL of the extract derived from 1 pericarp used in this test example contained about 3.4 ⁇ M nobiletin, and about 10.1 ⁇ M nobiletin in 300 ⁇ g / mL. As shown in FIG. 7A, the extract derived from the citrus pericarp showed significant CRE transcriptional activity in hippocampal neurons as compared to the control and nobiletin.
- the extract derived from the fruit peel of the present invention shows a remarkable CRE transcriptional activity exceeding the activity predicted from the content of nobiletin, and is expected to be effective as a therapeutic agent for Alzheimer's disease.
- Fine granules A A total of 200 to 200 g in proportions of 3.0 g of leather, 3.0 g of touki, 3.0 g of butterfly, 3.0 g of cucumber, 4.0 g of peanuts, 4.0 g of bakuryo, 2.0 g of psycho, 1.5 g of licorice and 5.0 g of hanger Herbal medicine was blended to 800 kg and extracted (extracted can) with water 2000-8000 L at 60-100 ° C for 30-180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 5000 rpm, solid-liquid separation was performed, followed by concentration to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator).
- the concentrated solution was spray-dried (spray dryer) at a rotational speed of 10000-20000 rpm, a supply temperature of 130-180 ° C, and an exhaust temperature of 60-120 ° C to obtain an extract formulation (Yokukansanka-Chanka summer extract). .
- a total amount of 50-400 kg is obtained at a ratio of 2.9 g of an additive consisting of magnesium stearate, corn starch, lactose, pullulan and magnesium aluminate metasilicate to 6.1 g of the obtained yokukansan kachu semi-summer extract.
- the resulting fine granule A 9.0 g contains 6.1 g of Yokukansan Kashide semi-summer extract containing 3.0 g of skin and other herbal medicines. Take 2 to 3 times a day.
- additive consisting of magnesium stearate, corn starch, lactose, pullulan and magnesium aluminate metasilicate
- the resulting fine granule B 6.0 g contains 3.7 ⁇ g Kumisoto extract containing 3.0 tachibana peel and other herbal medicines.
- the fine granule B 6.0 g per day Take 2 to 3 times.
- Chotosan extract obtained in a ratio of 1.52 g of additive consisting of hydrous silicon dioxide, magnesium stearate and corn starch in a total amount of 50-400 kg, 4 rpm for 20 minutes Mix (container rotating type mixer), mold at roll pressure of 490-2500 Pa, adjust particle size (dry granulator), classify particles between sieves 30-50 (cassette screen), fine granules C was obtained.
- the obtained fine granule C 6.0 g contains 4.48 g Chotosan extract containing 2.4 g Chen and other herbal medicines.
- the fine granule C 6.0 g 3 times a day for adults Take in divided doses.
- the ratio of 9.2 g of additive consisting of hydrous silicon dioxide, magnesium stearate and corn starch to 10 g of the crude or powdered raw material of cinnamon bark is blended in a total amount of 50 to 400 kg, and the number of rotations Mix at 4 rpm for 20 minutes (container rotation type mixer), mold at roll pressure 490-2500 Pa, adjust size (dry granulator), and classify particles between sieves 30-42 (cassette screen) As a result, a fine granule D was obtained.
- the resulting fine granule D (19.2 g) contains 10 g of crusted or tachibana peel, and as a guideline, the fine granule D (19.2 to 38.4 g) should be taken 2 to 3 times a day for adults.
- Decoction 10-30 g of decoction or tachibana peel was decocted with 200-600 mL of 20 times the amount of water at 100 ° C. for 1 hour, and the water was halved, followed by filtration to obtain a decoction.
- the resulting decoction (100-300 mL) contains 10-30 g of decoction or tachibana peel.
- 100-300 mL of decoction is taken in three divided doses per day for adults.
- the resulting fine granule E 9.0 g contains 7.2 g of cinnamon extract or citrus peel extract equivalent to 20 g of cinnamon or tachibana peel. Take 3 divided doses.
- the 13 capsules obtained (470 to 480 mg / piece) contain 3.6 kg of crust or extract of tachibana, equivalent to 10 g of crust or tachibana, and 13 to 26 capsules as a guide. Take 2 to 3 times a day for adults.
- Coated tablets 50 kg of peel or tachibana peel was extracted (extracted can) with 500 to 2000 L of water at 60 to 100 ° C for 30 to 180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 2500 rpm, solid-liquid separation was performed, and the solution was concentrated to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator). The concentrate was spray-dried (spray dryer) at a rotational speed of 10000 to 20000 rpm, an air supply temperature of 130 to 180 ° C., and an exhaust temperature of 60 to 120 ° C. to obtain an extract of crust or tachibana peel.
- 10 g of the resulting extract is blended in a ratio of 9.1 g of an additive consisting of maltose, lactose, magnesium aluminate metasilicate and magnesium stearate in a total amount in the range of 50 to 400 kg, with a rotation speed of 4 rpm, Mix for 20 minutes (container rotation type mixer), tablet at a rotation speed of 20-55 rpm, primary compression 0.6 mm or more, secondary compression 0.4 mm or more (high-speed rotary tableting machine), 1 tablet 350 mg I got a tablet.
- an additive consisting of maltose, lactose, magnesium aluminate metasilicate and magnesium stearate in a total amount in the range of 50 to 400 kg, with a rotation speed of 4 rpm, Mix for 20 minutes (container rotation type mixer), tablet at a rotation speed of 20-55 rpm, primary compression 0.6 mm or more, secondary compression 0.4 mm or more (high-speed rotary tableting machine), 1 tablet 350 mg
- the obtained 20 coated tablets contain 3.6 g of peel or tachibana peel equivalent to 10 g of peel or tachibana, and as a guideline, 20 to 40 coated tablets 2-3 times a day for adults Take in divided doses.
- Fine granule F 50 kg of peel or tachibana peel was extracted (extraction can) at 500 to 2000 L of water at 60 to 100 ° C. for 30 to 180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 2500 rpm, solid-liquid separation was performed, and the solution was concentrated to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator). The concentrate was spray-dried (spray dryer) at a rotational speed of 10000 to 20000 rpm, an air supply temperature of 130 to 180 ° C., and an exhaust temperature of 60 to 120 ° C. to obtain an extract of crust or tachibana peel.
- 10 g of the resulting extract is blended in a ratio of 15 g of an additive consisting of corn starch, maltose, hydrous silicon dioxide and magnesium stearate in a total amount in the range of 50 to 400 kg, rotating at 4 rpm, 20 Mix for a minute (container rotation type mixer), mold at roll pressure of 490-2500 Pa, adjust the size (dry granulator), classify particles between sieves 30-42 (cassette screen), fine particles Agent F was obtained.
- the obtained fine granule F 4.5 g contains 1.8 g of the skin extract or tachibana peel equivalent to 5 g of crust or tachibana skin, and as a guideline, the fine granule F 4.5 ⁇ 9.0 g per day for adults Take 2 to 3 times.
- 10 g of the resulting extract is blended in a ratio of 40 g of an additive consisting of calcium carmellose, hydrous silicon dioxide, magnesium aluminate metasilicate and magnesium stearate in a total amount of 50 to 400 kg and rotated.
- an additive consisting of calcium carmellose, hydrous silicon dioxide, magnesium aluminate metasilicate and magnesium stearate in a total amount of 50 to 400 kg and rotated.
- Mix at several 4 rpm for 20 minutes (container rotating type mixer), mold at roll pressure 490-2500 Pa, adjust particle size (dry granulator), and classify particles between sieves 12-42 (cassette screen) ) To obtain granules.
- the obtained granule (18 g) contains 3.6 ⁇ g of peel or citrus peel extract equivalent to 10 g of cinnamon or tachibana peel.
- 18 to 36 g of granule (2 to 3 times a day for adults) Take in divided doses.
- 10 g of the resulting extract is added in a ratio of 40 g of additives consisting of carrageenan, locust bean gum, reduced maltose liquid sugar, xylitol, apple fragrance, lecithin and citric acid in a total amount of 10-30 kg
- the mixture was mixed at a rotational speed of 50 to 150 rpm (mixing stirrer), and an equal amount of hot water of 80 ° C. or higher was added with stirring and cooled to 15 ° C. or lower to obtain a jelly agent.
- the obtained jelly preparation (18 g) contains 3.6 g of cinnamon extract or tachibana peel extract equivalent to 10 g of peel or tachibana peel.
- 18 to 36 g of jelly preparation is used 2-3 times per day Take in divided doses.
- the resulting extract (3.6 g) is blended at a ratio of 0.9 g of additive consisting of finely divided silicon dioxide and sucrose fatty acid ester in a total amount of 50 to 400 kg and mixed for 20 minutes at 4 rpm.
- Rotating mixer molded at a roll pressure of 490-2500 Pa, sized (dry granulator), classified between sieves 30-42 (cassette screen) to obtain fine granules.
- the obtained 4.5g of fine grain contains 3.6g of Chen or Tachibana bark extract equivalent to 10g of Chen or Tachibana, and as a guideline, take 4.5g of fine per day for adults.
- Ratio of 15.9 g of additive consisting of lecithin, reduced maltose starch syrup, aspartame, ascorbic acid, trisodium citrate, gelling agent (FG-2266, Nitta Gelatin Co., Ltd.) and orange flavor to 10 g of the obtained extract Then, the total amount was blended in the range of 50 to 400 kg, and mixed at a rotational speed of 4 rpm for 20 minutes (container rotating type mixer) to obtain a jelly base.
- gelling agent FG-2266, Nitta Gelatin Co., Ltd.
- the resulting jelly base of 9.3 g contains 3.6 g of cinnamon extract or citrus peel extract equivalent to 10 g of crust or tachibana, and as a guideline, ingest 9.3 to 18.6 g of jelly per day for adults .
- the dried plant tissue or plant tissue extract of the present invention is useful as a pharmaceutical or food for improving central neurodegenerative diseases such as Alzheimer's disease and Parkinson's disease, and is also used as a component of a promising fundamental therapeutic agent for the disease Is possible.
- the dried plant tissue or plant tissue extract of the present invention is particularly useful for memory acquisition, retention and enhanced recall ability.
Abstract
Description
(1)乾燥植物組織100重量%に対してノビレチンを0.4重量%以上含有する、中枢神経変性疾患の改善のための柑橘類の果皮の乾燥植物組織;
(2)乾燥植物組織100重量%に対してノビレチンを0.3重量%以上含有する、中枢神経変性疾患の改善のための柑橘類の葉の乾燥植物組織;
(3)ノビレチン/ナリルチンの含有重量比が1.0以上である、(1)または(2)のいずれか記載の乾燥植物組織;
(4)中枢神経変性疾患がアルツハイマー病および/またはパーキンソン病である、(1)または(2)のいずれか記載の乾燥植物組織;
(5)中枢神経変性疾患の改善がcAMP応答配列(CRE)依存的転写活性の増大、記憶の獲得、保持および想起能力の増強、チロシン・ヒドロキシラーゼ(TH)転写活性の促進、TH発現量の上昇、ドーパミン合成能の促進、またはドーパミン分泌の促進によりもたらされる、(1)または(2)のいずれか記載の乾燥植物組織; Specifically, the present invention includes the following embodiments:
(1) Dry plant tissue of citrus peel for improving central neurodegenerative diseases, containing 0.4% by weight or more of nobiletin with respect to 100% by weight of dry plant tissue;
(2) Dry plant tissue of citrus leaves for improving central neurodegenerative diseases, containing 0.3% by weight or more of nobiletin with respect to 100% by weight of dry plant tissue;
(3) The dry plant tissue according to any one of (1) and (2), wherein the weight ratio of nobiletin / nariltin is 1.0 or more;
(4) The dried plant tissue according to any one of (1) and (2), wherein the central neurodegenerative disease is Alzheimer's disease and / or Parkinson's disease;
(5) Improvement of central neurodegenerative diseases is increased cAMP response element (CRE) -dependent transcriptional activity, memory acquisition, retention and enhanced recall ability, tyrosine hydroxylase (TH) transcriptional activity, TH expression level The dried plant tissue according to any one of (1) and (2), which is brought about by an increase, promotion of dopamine synthesis ability, or promotion of dopamine secretion;
(7)ノビレチン/ナリルチンの含有重量比が2.0以上である、(6)記載の植物組織抽出物;
(8)(1)または(2)のいずれか記載の乾燥植物組織から水抽出、好ましくは60~100℃の水抽出により得られる、(6)記載の植物組織抽出物;
(9)中枢神経変性疾患がアルツハイマー病および/またはパーキンソン病である、(6)記載の植物組織抽出物;
(10)中枢神経変性疾患の改善がCRE依存的転写活性の増大、記憶の獲得、保持および想起能力の増強、TH転写活性の促進、TH発現量の上昇、ドーパミン合成能の促進、またはドーパミン分泌の促進によりもたらされる、(6)記載の植物組織抽出物; (6) Citrus plant tissue extract for improving central neurodegenerative diseases, containing 0.6% by weight or more of nobiletin with respect to 100% by weight of plant tissue extract;
(7) The plant tissue extract according to (6), wherein the content weight ratio of nobiletin / nariltin is 2.0 or more;
(8) The plant tissue extract according to (6) obtained by water extraction from the dried plant tissue according to any of (1) or (2), preferably 60 to 100 ° C water extraction;
(9) The plant tissue extract according to (6), wherein the central neurodegenerative disease is Alzheimer's disease and / or Parkinson's disease;
(10) Improvement of central neurodegenerative diseases may increase CRE-dependent transcriptional activity, acquire memory, enhance retention and recall ability, promote TH transcriptional activity, increase TH expression, promote dopamine synthesis, or dopamine secretion The plant tissue extract according to (6), which is caused by the promotion of
(12)柑橘類がCitrus reticulata Blanco (Rutaceae)である、(6)記載の植物組織抽出物;
(13)柑橘類がタチバナ、好ましくはニホンタチバナである、(1)または(2)のいずれか記載の乾燥植物組織;
(14)柑橘類がタチバナ、好ましくはニホンタチバナである、(6)記載の植物組織抽出物; (11) The dried plant tissue according to any one of (1) and (2), wherein the citrus fruit is Citrus reticulata Blanco (Rutaceae);
(12) The plant tissue extract according to (6), wherein the citrus fruit is Citrus reticulata Blanco (Rutaceae);
(13) The dried plant tissue according to any one of (1) and (2), wherein the citrus fruit is Tachibana, preferably Japanese Tachibana;
(14) The plant tissue extract according to (6), wherein the citrus fruit is Tachibana, preferably Japanese Tachibana;
(16)柑橘類がオオベニミカンである、(6)記載の植物組織抽出物; (15) The dried plant tissue according to any one of (1) and (2), wherein the citrus fruit is a giant orange.
(16) The plant tissue extract according to (6), wherein the citrus fruit is Obenimikan;
(18)医薬品100重量%に対して、10~100重量%の(1)もしくは(2)のいずれか記載の乾燥植物組織または10~90重量%の(6)記載の植物組織抽出物を有効成分とする、パーキンソン病治療用医薬品;
(19)(1)または(2)のいずれか記載の乾燥植物組織と医薬的に許容される生薬成分とを1:5~1:10の重量比率で配合して得られる配合物から水抽出、好ましくは60~100℃の水抽出により得られる抽出物を有効成分とする、中枢神経変性疾患の治療用医薬品;
(20)食品100重量%に対して、10~100重量%の(1)もしくは(2)のいずれか記載の乾燥植物組織または10~90重量%の(6)記載の植物組織抽出物を含有する、中枢神経変性疾患の改善のための食品;
(21)中枢神経変性疾患がアルツハイマー病および/またはパーキンソン病である、(20)記載の食品;
(22)中枢神経変性疾患の改善が記憶の獲得、保持および想起能力の増強メカニズムによりもたらされる、(20)記載の食品; (17) 10% to 100% by weight of the dry plant tissue according to (1) or (2) or 10% to 90% by weight of the plant tissue extract according to (6) is effective with respect to 100% by weight of the pharmaceutical product A drug for treating Alzheimer's disease as an ingredient;
(18) 10% to 100% by weight of the dry plant tissue according to (1) or (2) or 10% to 90% by weight of the plant tissue extract according to (6) is effective with respect to 100% by weight of the pharmaceutical product A drug for treating Parkinson's disease as an ingredient;
(19) Extraction with water from a blend obtained by blending the dry plant tissue according to any one of (1) and (2) and a pharmaceutically acceptable crude drug component in a weight ratio of 1: 5 to 1:10. A therapeutic drug for treating a central neurodegenerative disease, preferably comprising an extract obtained by water extraction at 60 to 100 ° C. as an active ingredient;
(20) 10 to 100% by weight of the dry plant tissue according to (1) or (2) or 10 to 90% by weight of the plant tissue extract according to (6) with respect to 100% by weight of food Food for ameliorating central neurodegenerative diseases;
(21) The food according to (20), wherein the central neurodegenerative disease is Alzheimer's disease and / or Parkinson's disease;
(22) The food according to (20), wherein the improvement of the central neurodegenerative disease is brought about by an enhancement mechanism of memory acquisition, retention and recall ability;
(24)柑橘類の植物組織を50~100℃にて1~3時間加熱乾燥することにより得られ、歩留まりが20~50%である、(4)記載の乾燥植物組織;
(25)植物組織抽出物100重量%に対してノビレチンを0.6~3.0重量%含有し、かつ、ノビレチン/ナリルチンの含有重量比が2.0~14.0である、(9)記載の植物組織抽出物;
(26)細粒剤、茶剤、煎剤、カプセル剤、錠剤、顆粒剤、ゼリー剤、散剤、液剤、シロップ剤またはエキス製剤の形態である、(19)記載の医薬品。 (23) The content of nobiletin is 0.3 to 2.0% by weight with respect to 100% by weight of the dry plant tissue, and the content weight ratio of nobiletin / nariltin is 1.0 to 23.0. Dry plant tissue of;
(24) The dried plant tissue according to (4), which is obtained by heating and drying citrus plant tissue at 50 to 100 ° C. for 1 to 3 hours, and the yield is 20 to 50%;
(25) The content of nobiletin is 0.6 to 3.0% by weight with respect to 100% by weight of the plant tissue extract, and the content weight ratio of nobiletin / nariltin is 2.0 to 14.0. (9) Plant tissue extracts as described;
(26) The pharmaceutical according to (19), which is in the form of a fine granule, tea, decoction, capsule, tablet, granule, jelly, powder, liquid, syrup or extract preparation.
本発明の乾燥植物組織は、これに含まれる各成分の割合が上記範囲内であれば、いずれの植物に由来するものであっても、本発明の薬効、すなわち中枢神経変性疾患改善効果(抗アルツハイマー病活性、抗パーキンソン病活性など)、学習・記憶障害改善効果、リン酸化促進作用およびPKA/ERKシグナル伝達促進効果、CRE転写活性およびTH転写活性、TH発現量上昇作用、ドーパミン合成能促進作用、またはドーパミン分泌促進作用を示す。 The dry plant tissue of the present invention comprises 0.3 or 0.4% by weight or more, preferably 0.3 to 2.0% by weight, more preferably 0.4 to 2.0% by weight of nobiletin based on the total weight. 0.7% by weight or less, preferably 0.4% by weight or less of nariltin, 0.1 to 0.8% by weight of tangeretin and 0.4 to 12% by weight of hesperidin. The dry plant tissue of the present invention also has a nobiletin / nariltin content weight ratio of 1.0 or more, preferably 1.5 to 23.0, more preferably 2.0 to 15.0, still more preferably 5.0 to 15.0.
The dry plant tissue of the present invention can be derived from any plant as long as the proportion of each component contained in the dry tissue is within the above range, that is, the drug effect of the present invention, that is, the central neurodegenerative disease improving effect (anti Alzheimer's disease activity, anti-Parkinson disease activity, etc., learning / memory impairment improvement effect, phosphorylation promotion effect and PKA / ERK signaling promotion effect, CRE transcription activity and TH transcription activity, TH expression level increase effect, dopamine synthesis ability promotion effect Or show dopamine secretion promoting action.
本発明の植物組織抽出物は、これに含まれる各成分の割合が上記範囲内であれば、いずれの植物に由来するものであっても、本発明の薬効、すなわち中枢神経変性疾患改善効果(抗アルツハイマー病活性、抗パーキンソン病活性など)、学習・記憶障害改善効果、リン酸化促進作用およびPKA/ERKシグナル伝達促進効果、CRE転写活性およびTH転写活性、TH発現量上昇作用、ドーパミン合成能促進作用、またはドーパミン分泌促進作用を示す。 The plant tissue extract of the present invention is 0.6 to 2.0% by weight or more, preferably 0.6 to 3.0% by weight of nobiletin, 0.7% by weight or less, preferably 0. Contains no more than 4% by weight nariltin, 0.1-1.0% by weight tangeretin and 1.8-6.0% by weight hesperidin. The plant tissue extract of the present invention also has a nobiletin / nariltin content weight ratio of 2.0 or more, preferably 2.0 to 14.0, more preferably 2.5 to 10.0.
The plant tissue extract of the present invention can be derived from any plant as long as the ratio of each component contained therein is within the above-mentioned range. Anti-Alzheimer's disease activity, anti-Parkinson's disease activity, etc., learning / memory impairment improvement effect, phosphorylation promotion effect and PKA / ERK signaling promotion effect, CRE transcription activity and TH transcription activity, TH expression level increasing effect, dopamine synthesis ability promotion An action or a dopamine secretion promoting action is shown.
より詳細には、本発明の乾燥植物組織および植物組織抽出物は、植物組織の種類に応じて異なる活性を示してもよい。例えば、ドーパミンが関連する活性について、陳皮または陳皮エキスはドーパミン含量上昇活性を示し得、橘皮または橘皮エキスはドーパミン分泌促進活性を示し得る。 Examples of the anti-Alzheimer's disease activity and / or anti-Parkinson's disease activity of the dried plant tissue and plant tissue extract of the present invention include improvement of learning / memory impairment related to the onset mechanism of central neurodegenerative diseases or promotion of dopamine synthesis. . Specifically, synaptic plasticity promoting activity and CRE (cAMP responsive element) -dependent transcription activity, or tyrosine hydroxylase (TH) transcription activity promoting activity, TH expression level increasing activity, dopamine content increasing activity, dopamine secretion promoting activity, etc. It is.
More specifically, the dried plant tissue and plant tissue extract of the present invention may exhibit different activities depending on the type of plant tissue. For example, with regard to the activity associated with dopamine, the skin or skin extract may exhibit a dopamine content increasing activity, and the tachibana skin or citrus skin extract may exhibit a dopamine secretion promoting activity.
本実施例において、種々の柑橘類から採取した果皮および葉を陰干し、日干しまたは加熱乾燥することにより陳皮、橘皮、タチバナの葉およびオオベニミカンの果皮の乾燥体を製造した。加熱乾燥は、歩留まり(乾燥後質量/乾燥前質量の割合)が陳皮および橘皮について20~50%、タチバナの葉について20~50%、オオベニミカンの果皮について20~50%となるまで行い、具体的には温度60℃にて2時間加熱乾燥した。 [Example 1] Production of dried plant tissue In this example, the peels and leaves collected from various citrus fruits were shade-dried, sun-dried or heat-dried to obtain dried bodies of the peel, tachibana peels, tachibana leaves, and the fruit of periwinkle. Manufactured. Heat drying is carried out until the yield (ratio of mass after drying / mass before drying) is 20-50% for Chen and Tachibana peels, 20-50% for Tachibana leaves, and 20-50% for Prunus pericarp. Specifically, it was dried by heating at a temperature of 60 ° C. for 2 hours.
実施例1で得られた陳皮、橘皮、タチバナの葉またはオオベニミカンの果皮を細切りしたもの約10 gに純水400 mLを加えて加熱した(橘皮6については、細切りしたもの約20 gに純水1000 mLを加えて加熱した)。混合物が沸騰した後、100℃にて1時間かけて抽出した。次いで、ガーゼ(2枚)を通してろ過し、ろ液を凍結乾燥して植物組織抽出物を得た。植物組織抽出物の製造結果を以下の表1に示す。 [Example 2] Production of plant tissue extract 400 mL of pure water was added to about 10 g of the cut skin, tachibana bark, leaves of tachibana or persimmon bark obtained in Example 1 and heated (tachibana bark) For 6), 1000 mL of pure water was added to about 20 g of the minced and heated. After the mixture boiled, it was extracted at 100 ° C. for 1 hour. Subsequently, it filtered through gauze (2 sheets), and the filtrate was freeze-dried to obtain a plant tissue extract. The production results of the plant tissue extract are shown in Table 1 below.
実施例1および2で得られた乾燥植物組織および植物組織抽出物の含有成分を以下のようにして測定した。以下に、陳皮および陳皮エキスについての分析方法を示す。 [Example 3] Analysis of contained components The contained components of the dried plant tissues and plant tissue extracts obtained in Examples 1 and 2 were measured as follows. Below, the analysis method about a crust and a crust extract is shown.
ノビレチンおよびタンゲレチンの定量に用いた標準溶液を以下のように調製した。ノビレチン(和光純薬工業株式会社)をデシケーター(シリカゲル)で24時間以上乾燥した。乾燥ノビレチン約5 mgをメタノール/水混液(7:3)に溶解して100 mLとし、ノビレチン標準溶液とした。また、タンゲレチン(和光純薬工業株式会社)をデシケーター(シリカゲル)で24時間以上乾燥した。乾燥タンゲレチン約3 mgをメタノール/水混液(7:3)に溶解して100 mLとし、タンゲレチン標準溶液とした。 (Quantification of nobiletin and tangeretin)
A standard solution used for quantification of nobiletin and tangeretin was prepared as follows. Nobiletin (Wako Pure Chemical Industries, Ltd.) was dried with a desiccator (silica gel) for 24 hours or more. About 5 mg of dried nobiletin was dissolved in a methanol / water mixture (7: 3) to make 100 mL, and used as a nobiletin standard solution. Further, tangeretin (Wako Pure Chemical Industries, Ltd.) was dried with a desiccator (silica gel) for 24 hours or more. About 3 mg of dry tangeretin was dissolved in a methanol / water mixture (7: 3) to make 100 mL, and used as a tangeretin standard solution.
陳皮エキスの試料溶液として、陳皮エキス約0.3 gを50 mLの共栓遠心沈殿管に入れ、これにメタノール/水混液(7:3)50 mLを加えた。混合物に超音波を照射して(UT-305HS、シャープ株式会社)抽出を行った後、遠心分離した(KUBOTA KN-70、株式会社久保田製作所)。その後ろ過し、陳皮エキスの試料溶液とした。 As a sample solution of crust, about 0.3 g of crust powder was placed in a 50 mL stoppered centrifuge tube, and 50 mL of methanol was added thereto. The mixture was extracted with ultrasonic waves (UT-305HS, Sharp Corporation) and then centrifuged (KUBOTA KN-70, Kubota Corporation). Thereafter, it was filtered to obtain a sample solution for skin.
As a sample solution of the peel extract, about 0.3 g of the peel extract was placed in a 50 mL stoppered centrifugal tube, and 50 mL of a methanol / water mixture (7: 3) was added thereto. The mixture was extracted with ultrasonic waves (UT-305HS, Sharp Corporation) and then centrifuged (KUBOTA KN-70, Kubota Corporation). Then, it filtered and it was set as the sample solution of the crust extract.
検出器: 紫外吸光光度計(測定波長: 338 nm)
カラム: Mightysil RP-18 4.6 mm x 15 cm(関東化学株式会社)
移動相: 水/アセトニトリル混液(3:2)
流速: 1.0 mL/分(ノビレチンの保持時間を約10分とした)
カラム温度: 40℃
注入量: 10μL The standard solution and sample solution prepared as described above were quantified under the following analytical conditions.
Detector: UV absorptiometer (measurement wavelength: 338 nm)
Column: Mightysil RP-18 4.6 mm x 15 cm (Kanto Chemical Co., Inc.)
Mobile phase: Water / acetonitrile mixture (3: 2)
Flow rate: 1.0 mL / min (Nobiletin retention time was approximately 10 minutes)
Column temperature: 40 ° C
Injection volume: 10μL
ヘスペリジンおよびナリルチンの定量に用いた標準溶液を以下のように調製した。ヘスペリジン(和光純薬工業株式会社)をデシケーター(シリカゲル)で24時間以上乾燥した。乾燥ヘスペリジン約10 mgを50%メタノールに溶解して50 mLとし、ヘスペリジン標準溶液とした。また、ナリルチン(和光純薬工業株式会社)をデシケーター(シリカゲル)で24時間以上乾燥した。乾燥ナリルチン約10 mgを50%メタノールに溶解して500 mLとし、ナリルチン標準溶液とした。 (Quantitative determination of hesperidin and nariltin)
A standard solution used for quantification of hesperidin and nariltin was prepared as follows. Hesperidin (Wako Pure Chemical Industries, Ltd.) was dried with a desiccator (silica gel) for 24 hours or more. About 10 mg of dry hesperidin was dissolved in 50% methanol to make 50 mL, and used as a hesperidin standard solution. Also, Nariltin (Wako Pure Chemical Industries, Ltd.) was dried with a desiccator (silica gel) for 24 hours or more. About 10 mg of dry narilutin was dissolved in 50% methanol to make 500 mL, which was used as a narilutin standard solution.
陳皮エキスの試料溶液として、陳皮エキス約0.1 gを50 mLの共栓遠心沈殿管に入れ、これにメタノール/水混液(1:1)50 mLを加えた。混合物に超音波を照射して(UT-305HS、シャープ株式会社)抽出を行った後、遠心分離した(KUBOTA KN-70、株式会社久保田製作所)。その後ろ過し、陳皮エキスの試料溶液とした。 As a skin sample solution, 30 mL of methanol was added to about 0.1 g of skin powder. The mixture was irradiated with ultrasonic waves for 20 minutes (UT-305HS, Sharp Corporation), extracted, and then centrifuged (KUBOTA KN-70, Kubota Corporation) to collect the supernatant. 20 mL of methanol was added to the residue, and extraction was performed by irradiating with ultrasonic waves for 20 minutes, followed by centrifugation. This was combined with the collected supernatant to obtain a skin sample solution.
As a sample solution of the peel extract, about 0.1 g of the peel extract was placed in a 50 mL stoppered centrifugal sedimentation tube, and 50 mL of a methanol / water mixture (1: 1) was added thereto. The mixture was extracted with ultrasonic waves (UT-305HS, Sharp Corporation) and then centrifuged (KUBOTA KN-70, Kubota Corporation). Then, it filtered and it was set as the sample solution of the crust extract.
検出器: 紫外吸光光度計(測定波長: 285 nm)
カラム: L-column2 ODS 4.6 mm x 25 cm(化学物質評価研究機構)
移動相: 水/アセトニトリル/酢酸混液(40:10:1)
流速: 0.8 mL/分(ヘスペリジンの保持時間を約15分とした)
カラム温度: 30℃
注入量: 10μL The standard solution and sample solution prepared as described above were quantified under the following analytical conditions.
Detector: UV spectrophotometer (measurement wavelength: 285 nm)
Column: L-column2 ODS 4.6 mm x 25 cm (Research Organization for Chemical Evaluation)
Mobile phase: Water / acetonitrile / acetic acid mixture (40: 10: 1)
Flow rate: 0.8 mL / min (Hesperidin retention time was about 15 minutes)
Column temperature: 30 ° C
Injection volume: 10μL
上記実施例で得られた陳皮エキスについて以下のインビトロおよびインビボ試験を行った。 [Test example]
The following in vitro and in vivo tests were conducted on the skin extract obtained in the above examples.
以下の試験例1で用いた一般的試験手順は以下のとおりである。
(ラット胎仔初代海馬神経細胞の培養)
妊娠Sprague-Dawley(SD)ラットを12時間周期の明暗サイクルにて給餌・給水して飼育した。妊娠18日目のラット(E18)からエーテル深麻酔下にて腹部正中切開により無菌的に子宮を摘出した。実体顕微鏡下、氷冷リン酸緩衝生理食塩水(PBS)中にて胎仔の海馬を摘出し、パパイン酵素(SUMILON)で組織を分散させ、1000 rpmにて4分間遠心分離した後、上清を除去した。次いで、細胞ペレットを分散液(SUMILON)中にて分散させ、さらにピペッティングにより十分分散させた細胞に除去液(SUMILON)を加えて900 rpmにて5分間遠心分離した後、上清を除去した。
次に、ペレットをNeurobasal Medium(Neurobasal Medium 500 mL/フェノール・レッド不含, 50 x B27 Supplement 10 ml, 0.5 mM L-グルタミン, 0.005%ペニシリン-ストレプトマイシン)を用いて懸濁し、ポリ-L-リジンでコーティングした皿またはプレートに播種した。培養1日後に培地交換し、その後3~4日おきに培地を半量交換し、10μM AraCを含有する培地中37℃にて5% CO2インキュベーター内で14日間培養した。
なお、薬物処置実験用試験培地は、10μM AraCを含有するNeurobasal Mediumを用いた。 [General test procedure 1]
The general test procedure used in Test Example 1 below is as follows.
(Culture of rat fetal primary hippocampal neurons)
Pregnant Sprague-Dawley (SD) rats were bred by feeding and watering in a 12-hour light / dark cycle. The uterus was aseptically removed from the rat (E18) on
Next, the pellet is suspended using Neurobasal Medium (
In addition, Neurobasal Medium containing 10 μM AraC was used as a test medium for drug treatment experiments.
ラット胎仔初代海馬神経細胞を35 mm皿に1×106細胞/皿にて播種し、成長培地中14日間5% CO2インキュベーターで培養した後、薬物を10分間処置した。
各細胞を薬物処置した後、氷冷したPBSで洗浄した。次いで、細胞可溶化液(1 mM EDTA, 1% SDS, 10 mM NaF, 10 nMカリキュリン, 320 nMオカダ酸, 1 mMオルトバナジウム酸ナトリウム, 1 mM p-APMSF, 10μg/mLペプスタチンA, 10μg/mLアンチパイン, 10μg/mLロイペプチン, 10μg/mLキモスタチン, 10μg/mLホスホラミドン, 10 mM HEPES, pH7.5)で回収した。その後すみやかに95℃にて5分間煮沸し、タンパク質を変性させた後、DNAを超音波破砕してSDS-PAGE用サンプルを調製した。 (SDS-PAGE and Western blot analysis 1)
Rat fetal primary hippocampal neurons were seeded at 1 × 10 6 cells / dish in a 35 mm dish, cultured in a growth medium for 14 days in a 5% CO 2 incubator, and then treated with the drug for 10 minutes.
Each cell was treated with drugs and then washed with ice-cold PBS. Cell lysate (1 mM EDTA, 1% SDS, 10 mM NaF, 10 nM caliculin, 320 nM okadaic acid, 1 mM sodium orthovanadate, 1 mM p-APMSF, 10 μg / mL pepstatin A, 10 μg / mL Antipine, 10 μg / mL leupeptin, 10 μg / mL chymostatin, 10 μg / mL phosphoramidon, 10 mM HEPES, pH 7.5). Thereafter, it was immediately boiled at 95 ° C. for 5 minutes to denature the protein, and then the DNA was sonicated to prepare a sample for SDS-PAGE.
実験結果はワンウェイANOVA(Student-Newman-Keuls)を用いて評価した。有意水準を両側5%として検定し、p<0.05を有意とした。 (Statistical analysis 1)
The experimental results were evaluated using a one-way ANOVA (Student-Newman-Keuls). The significance level was tested at 5% on both sides, and p <0.05 was considered significant.
本試験例において、学習・記憶障害に対する本発明の陳皮エキスの作用を検討した。インビトロ試験において、陳皮エキスの海馬ニューロンにおける核内タンパク質CREBのリン酸化促進作用を検討し、初代培養海馬神経細胞におけるNMDA受容体遮断薬(MK801)によるPKA/ERKシグナル伝達抑制への陳皮エキスの影響を検討した。
なお、本明細書において用いるMK801は、グルタミン酸受容体のサブタイプの一つであるNMDA受容体の非選択的アンタゴニストであり、ジゾシルピン(dizocilpine)とも称される。
本試験例で用いた陳皮エキスは以下のように調製した。すなわち、インビトロ試験では、陳皮1からの陳皮エキス7.2 gを秤量し、50 ml遠心チューブに入れ、これにジメチルスルホキシド(DMSO)を加えて30 mLとした。その後、30分間超音波処理し、3000 rpmで10分間遠心分離し、上清を回収して陳皮エキスとして用いた。 [Test Example 1] Improvement of learning and memory impairment by the skin extract (in vitro test)
In this test example, the effect of the skin extract of the present invention on learning / memory impairment was examined. In vitro study, we investigated the phosphorylation-promoting action of nuclear protein CREB in hippocampal neurons, and the effect of skin extract on PKA / ERK signaling suppression by NMDA receptor blocker (MK801) in primary cultured hippocampal neurons It was investigated.
MK801 used in the present specification is a non-selective antagonist of NMDA receptor, which is one of the subtypes of glutamate receptor, and is also referred to as dizocilpine.
The skin extract used in this test example was prepared as follows. That is, in the in vitro test, 7.2 g of the skin extract from the
種々の濃度の陳皮エキスによるCREBリン酸化に対する効果についてウェスタン・ブロット法を用いて検討した。1×106細胞/35-mmプラスチックシャーレの密度にて増殖させたE18ラット海馬神経細胞を14日間培養した。細胞を陳皮エキス(30, 60, 120, 240および480μg/ml)で10分間処理した。抗リン酸化CREB抗体を用いてウェスタン・ブロット法を行った。次いで、ブロットを剥ぎ取り、抗14-3-3-β抗体について再度試験し、等量のタンパク質が各レーンにて電気泳動されたことを確認した。
図2Aに示されるように、陳皮エキスは濃度依存的にCREBリン酸化を促進した。 [Test Example 1-1] CREB phosphorylation-promoting effect of skin extract in primary cultured hippocampal neurons The effect of various concentrations of skin extract on CREB phosphorylation was examined using Western blotting. E18 rat hippocampal neurons grown at a density of 1 × 10 6 cells / 35-mm plastic dish were cultured for 14 days. Cells were treated with crust extract (30, 60, 120, 240 and 480 μg / ml) for 10 minutes. Western blotting was performed using anti-phosphorylated CREB antibody. The blot was then stripped and tested again for anti-14-3-3-β antibody to confirm that an equal amount of protein was electrophoresed in each lane.
As shown in FIG. 2A, the peel extract promoted CREB phosphorylation in a concentration-dependent manner.
種々の濃度の陳皮エキスによるPKA基質リン酸化に対する効果についてウェスタン・ブロット法を用いて検討した。1×106細胞/35-mmプラスチックシャーレの密度にて播種したE18ラット海馬神経細胞を14日間培養した。細胞を異なる濃度の陳皮エキスで10分間処理した。抗リン酸化PKA基質抗体を用いてウェスタン・ブロット法を行った。次いで、ブロットを剥ぎ取り、抗14-3-3-β抗体について再度試験し、等量のタンパク質が各レーンにて電気泳動されたことを確認した。
図2Bに示されるように、陳皮エキスは濃度依存的にPKA基質リン酸化を促進した。 [Test Example 1-2] PKA substrate phosphorylation-promoting effect of skin extract in primary cultured hippocampal neurons The effect of various concentrations of skin extract on PKA substrate phosphorylation was examined using Western blotting. E18 rat hippocampal neurons seeded at a density of 1 × 10 6 cells / 35-mm plastic dish were cultured for 14 days. Cells were treated with different concentrations of crust extract for 10 minutes. Western blotting was performed using anti-phosphorylated PKA substrate antibody. The blot was then stripped and tested again for anti-14-3-3-β antibody to confirm that an equal amount of protein was electrophoresed in each lane.
As shown in FIG. 2B, the peel extract promoted PKA substrate phosphorylation in a concentration-dependent manner.
種々の濃度の陳皮エキスまたはノビレチンのERK1/2リン酸化に対する効果についてウェスタン・ブロット法を用いて検討した(陳皮エキス60μg/ml中にノビレチン1μMを含有)。1×106細胞/35-mmプラスチックシャーレの密度にて増殖させたE18ラット海馬神経細胞を14日間培養した。細胞を陳皮エキス(30, 60, 120, 240および480μg/ml)またはノビレチン(4, 8および30μM)で10分間処理した。抗リン酸化ERK1/2抗体を用いてウェスタン・ブロット法を行った。次いで、ブロットを剥ぎ取り、抗ERK1/2抗体について再度試験し、等量のタンパク質が各レーンにて電気泳動されたことを確認した。
図2Cに示されるように、陳皮エキスは濃度依存的にERK1/2リン酸化を促進した。また、陳皮エキスはノビレチンと比較して、強い作用を示した。すなわち、ノビレチン4μMを含有する本発明の陳皮エキス240μg/mlのリン酸化促進効果は、ブロットの大きさによりノビレチン4μMの効果より強いことが示唆された。 [Test Example 1-3] Extracellular signal-regulated kinase (ERK) 1/2 phosphorylation-promoting effect of skin extract in primary cultured hippocampal neurons About the effect of various concentrations of skin extract or nobiletin on ERK1 / 2 phosphorylation Blotting was used (contains
As shown in FIG. 2C, the peel extract promoted ERK1 / 2 phosphorylation in a concentration-dependent manner. Moreover, Chen extract showed a strong effect compared with nobiletin. That is, it was suggested that the phosphorylation promoting effect of the
初代培養ラット海馬神経細胞においてMK801によるPKA/ERKシグナル伝達抑制に及ぼす陳皮エキスの影響を検討した。初代培養ラット海馬神経細胞に、陳皮エキスまたはノビレチンを1時間前処置した後、MK801を30分間処置し、さらに15分間N-メチル-D-アスパラギン酸(NMDA)で処置した。抗リン酸化PKA基質抗体を用いてウェスタン・ブロット法を行った。次いで、ブロットを剥ぎ取り、抗14-3-3-β抗体について再度試験し、等量のタンパク質が各レーンにて電気泳動されたことを確認した(図2Dを参照のこと)。また、抗リン酸化ERK1/2抗体を用いてウェスタン・ブロット法を行った。次いで、ブロットを剥ぎ取り、抗ERK1/2抗体について再度試験し、等量のタンパク質が各レーンにて電気泳動されたことを確認した(図2Eを参照のこと)。
その結果、MK801はNMDAによるPKA/ERKシグナル伝達を抑制した。しかし、陳皮エキスは濃度依存的にMK801によるシグナル伝達抑制に対して拮抗作用を示し、陳皮エキス(陳皮エキス480μg/ml中にノビレチン8μMを含有)の作用はノビレチン8μMのものよりも強力であった。 [Test Example 1-4] PKA / ERK signal transduction promoting effect of skin extract in primary cultured hippocampal neurons The effect of skin extract on PKA / ERK signal transmission suppression by MK801 in primary cultured rat hippocampal neurons was examined. Primary cultured rat hippocampal neurons were pretreated for 1 hour with skin extract or nobiletin, then with MK801 for 30 minutes, and then with N-methyl-D-aspartic acid (NMDA) for 15 minutes. Western blotting was performed using anti-phosphorylated PKA substrate antibody. The blot was then stripped and retested for anti-14-3-3-β antibody to confirm that equal amounts of protein were electrophoresed in each lane (see FIG. 2D). Further, Western blotting was performed using an anti-phosphorylated ERK1 / 2 antibody. The blot was then stripped and retested for anti-ERK1 / 2 antibodies to confirm that equal amounts of protein were electrophoresed in each lane (see FIG. 2E).
As a result, MK801 suppressed PKA / ERK signaling by NMDA. However, the skin extract showed an antagonistic effect on the signal transduction suppression by MK801 in a concentration-dependent manner, and the effect of the skin extract (containing 8 μM nobiletin in the
上記試験例の結果から、シナプス形成および長期記憶の形成に深く関与することが知られている核内タンパク質であるCREB、さらにその上流にあるPKA基質およびERK1/2のリン酸化において、本発明の陳皮エキスは有意なリン酸化促進作用を有することが明らかとなった。本発明の陳皮エキスはまた、ERK1/2においてノビレチンよりも強いリン酸化促進作用を示した。
さらに、本発明の陳皮エキスは、MK801によるPKA/ERKシグナル伝達抑制に対して有意に拮抗作用を示し、その作用はノビレチンよりも強力であった。これにより本発明の陳皮エキスは、これに含まれるノビレチンとノビレチン以外の成分との相乗的作用を有することが示された。 (Discussion 1)
From the results of the above test examples, CREB, a nuclear protein known to be deeply involved in synapse formation and long-term memory formation, as well as phosphorylation of PKA substrate and ERK1 / 2 upstream thereof, It was revealed that the peel extract has a significant phosphorylation promoting action. The skin extract of the present invention also exhibited a stronger phosphorylation promoting effect than nobiletin at ERK1 / 2.
Furthermore, the skin extract of the present invention exhibited a significant antagonistic effect on the suppression of PKA / ERK signaling by MK801, and the effect was stronger than nobiletin. Thereby, it was shown that the skin extract of this invention has a synergistic effect | action with components other than nobiletin contained therein and nobiletin.
本試験例において、本発明の陳皮エキスのCRE依存的転写活性作用を従来の陳皮エキスおよびノビレチンと比較した。 [Test Example 2] CRE-dependent transcriptional activity of a crust extract in hippocampal neurons (in vitro test)
In this test example, the CRE-dependent transcriptional activity of the skin extract of the present invention was compared with the conventional skin extract and nobiletin.
統計学的解析のために、レポータージーンアッセイ、RT-PCRで得られた結果はone-way ANOVA(Tukey)を用いて評価した。有意水準を両側5%として検定し、p<0.05を有意とした。 As described in
For statistical analysis, the reporter gene assay and RT-PCR results were evaluated using one-way ANOVA (Tukey). The significance level was tested at 5% on both sides, and p <0.05 was considered significant.
図2Fおよび2Gに示されるように、本試験例で用いた陳皮エキスにおけるノビレチン濃度は2.5μMであり、30μMノビレチンの12分の1の濃度であるにもかかわらず、そのCRE依存的転写活性は30μMノビレチンを超えるものであった。すなわち、多成分系の本発明の陳皮エキスでは、ノビレチン以外の成分の存在によりノビレチンのCRE依存的転写促進活性がノビレチン単体から想定される活性よりも顕著かつ相乗的に増大することが判明した。さらに、本発明の陳皮エキスは、従来の陳皮エキスよりも強いCRE依存的転写活性が認められたことから、学習・記憶障害改善作用を示し、より効果的にアルツハイマー病の認識機能を改善することが示された。 (Discussion 2)
As shown in FIGS. 2F and 2G, the concentration of nobiletin in the skin extract used in this test example is 2.5 μM, and its CRE-dependent transcriptional activity is 1 / 12th that of 30 μM nobiletin. More than 30 μM nobiletin. That is, it was found that in the multi-component system of the skin extract of the present invention, the presence of components other than nobiletin significantly and synergistically increases the CRE-dependent transcription promoting activity of nobiletin than the activity expected from nobiletin alone. In addition, the skin extract of the present invention showed a CRE-dependent transcriptional activity stronger than that of conventional skin extracts, so it showed an action to improve learning and memory impairment and more effectively improved the recognition function of Alzheimer's disease. It has been shown.
次に、インビボ試験において、MK801誘発性記憶障害モデルマウスを用いて、行動薬理学的に学習・記憶障害に対する本発明の陳皮エキスの作用を検討した。
本試験例で用いた陳皮エキスは以下のように調製した。すなわち、陳皮1からの陳皮エキス5.54 gを秤量し、50 ml遠心チューブに入れ、これに生理食塩水を加えて30 mLとした。その後、30分間超音波処理した。
マウスへの陳皮エキスの投与用量は、ノビレチン含量を基に調製した。ここで、ノビレチン含量は、予めノビレチン標準物質を用いたHPLC法の絶対検量線法により測定した。 [Test Example 3] Learning and memory impairment improving action of the skin extract (in vivo test)
Next, in the in vivo test, the action of the skin extract of the present invention on behavioral pharmacological learning / memory impairment was examined using MK801-induced memory impairment model mice.
The skin extract used in this test example was prepared as follows. That is, 5.54 g of skin extract from
The administration dose of the skin extract to mice was prepared based on the nobiletin content. Here, the nobiletin content was measured in advance by the absolute calibration curve method of the HPLC method using a nobiletin standard substance.
恐怖付け学習試験において、マウスを透明なボックスに入れ、2分間自由に探索させた後、0.7 mA、2 sの電気刺激を与えた。これを3回繰り返して学習試行を行い、記憶の獲得および保持能力を評価した。24時間後に、マウスを再び透明なボックスに入れ、マウスのすくみ行動、すなわち呼吸以外のすべての動作が停止した状態を指標とした確認試行を行い、学習・記憶行動として5分間の測定により保持および想起能力を評価した。結果を図3Aに示す。 We investigated the effects of chronic administration of Chen extract on MK801-induced learning and memory impairment. Mice were divided into 4 groups, each with a skin extract of 1.48 g / kg or 3.69 g / kg (nobiletin content: 10 mg / kg or 25 mg / kg, respectively), and 2 groups with physiological saline for 7 consecutive days Orally. On the 7th day, 90 minutes after administration of these, MK801 (0.08 mg / kg) or physiological saline was administered subcutaneously. Thirty minutes later, a fear conditioning learning test was conducted.
In the horror learning test, mice were placed in a transparent box and allowed to explore freely for 2 minutes, followed by 0.7 mA, 2 s electrical stimulation. This was repeated three times and learning trials were performed to evaluate memory acquisition and retention ability. After 24 hours, place the mouse in the transparent box again, perform a confirmation test using the mouse's freezing action, i.e., all movements other than breathing as an index, and keep it as a learning / memory action by measuring for 5 minutes. Recall ability was assessed. The results are shown in FIG. 3A.
上記試験例1-1にて、陳皮エキスの経口投与をノビレチン(10, 50 mg/kg)の腹腔内投与に代えて比較試験を行った。結果を図3Bに示す。 [Comparative Test 1] Examination of learning / memory impairment improving effect of nobiletin In Comparative Example 1-1, a comparative test was conducted by replacing oral administration of crust extract with intraperitoneal administration of nobiletin (10, 50 mg / kg). It was. The results are shown in FIG. 3B.
上記試験例の結果から、インビボにおいても、本発明の陳皮エキスの慢性投与により学習・記憶障害を有意に改善することに加え、ノビレチンでは認められない記憶の獲得および保持能力を増強することが明らかとなった。
以上より、本発明の陳皮エキスは、シナプス可塑性促進作用およびPKA/ERK保護作用により、学習・記憶障害を改善し、さらに記憶の獲得能力を増強することが示された。すなわち、本発明の陳皮エキスは、ノビレチンよりもアルツハイマー病の中核症状である学習・記憶障害を改善し、特にノビレチンには認められない記憶の獲得および保持能力を増強および/または改善することが示された。 (Discussion 3)
From the results of the above test examples, it is clear that, in vivo, chronic administration of the skin extract of the present invention significantly improves learning and memory impairment, and also enhances the ability to acquire and retain memory that is not observed with nobiletin. It became.
From the above, it was shown that the skin extract of the present invention improves learning / memory impairment and further enhances the ability to acquire memory by promoting synaptic plasticity and protecting PKA / ERK. That is, it is shown that the skin extract of the present invention improves learning / memory impairment, which is a core symptom of Alzheimer's disease, and enhances and / or improves the ability to acquire and retain memory, which is not found in nobiletin, more than nobiletin. It was done.
以下の試験例4で用いた一般的試験手順は以下のとおりである。
(PC12D細胞の培養)
ダルベッコ改変イーグル培地(DMEM)9.5 gをMilliQ水800 mLに溶解し、これにグルコース3.5 gを加えMilliQ水で1 Lにした。オートクレーブで滅菌したものを830 mLとり、10%炭酸水素ナトリウム水溶液17 mLおよび3% L-グルタミン水溶液16.5 mLを加えた。次いで、10%となるようにウマ血清(HS)、5%となるようにウシ胎仔血清(FCS)を加えて調製したものを成長培地とし、37℃にて5% CO2インキュベーター内で培養した。
試験培地は、2% HS, 1% FCSを含有するhDMEMを使用した。 [General test procedure 2]
The general test procedure used in Test Example 4 below is as follows.
(PC12D cell culture)
Dulbecco's modified Eagle medium (DMEM) (9.5 g) was dissolved in MilliQ water (800 mL), and glucose (3.5 g) was added thereto to make 1 L with MilliQ water. 830 mL of the product sterilized by autoclave was taken, and 17 mL of 10% aqueous sodium hydrogen carbonate solution and 16.5 mL of 3% L-glutamine aqueous solution were added. Next, equine serum (HS) to 10% and fetal calf serum (FCS) to 5% were prepared as growth media and cultured in a 5% CO 2 incubator at 37 ° C. .
The test medium used was hDMEM containing 2% HS and 1% FCS.
PC12D細胞を48ウェルプレートに8×104細胞/ウェルで播種し、成長培地にて24時間培養した後、pRTHプラスミドを0.1μg/ウェル、ウミシイタケpRG-TKプラスミドを0.01μg/ウェルリポフェクションした。トランスフェクション19時間後に化合物を溶解した試験培地で培地交換した。24時間インキュベートした後、培地を吸引し、Passive Lysis Bufferにて回収した。
ホタルシイタケおよびウミシイタケルシフェラーゼはDual-Luciferase Reporter Assay System(Promega)を用い、ルミノメーターで測定した。 (Plasmid introduction and reporter gene assay)
PC12D cells were seeded on a 48-well plate at 8 × 10 4 cells / well and cultured in a growth medium for 24 hours, followed by lipofection with 0.1 μg / well of pRTH plasmid and 0.01 μg / well of Renilla pRG-TK plasmid. The medium was replaced with a test medium in which the compound was dissolved 19 hours after transfection. After incubation for 24 hours, the medium was aspirated and collected with Passive Lysis Buffer.
Firefly shiitake and Renilla luciferase were measured with a luminometer using Dual-Luciferase Reporter Assay System (Promega).
PC12D細胞を35 mm皿に0.67×106細胞/皿にて播種し、成長培地中2日間5% CO2インキュベーターで培養した後、薬物を24時間処置した。
各細胞を処置した後、氷冷したPBSで洗浄した。次いで、細胞可溶化液(1 mM EDTA, 1% SDS, 10 mM NaF, 10 nMカリキュリン, 320 nMオカダ酸, 1 mMオルトバナジウム酸ナトリウム, 1 mM p-APMSF, 10μg/mLペプスタチンA, 10μg/mLアンチパイン, 10μg/mLロイペプチン, 10μg/mLキモスタチン, 10μg/mLホスホラミドン, 10 mM HEPES, pH7.5)で回収した。その後すみやかに95℃にて5分間煮沸し、タンパク質を変性させた後、DNAを超音波破砕して遠心上清をSDS-PAGE用サンプルとして調製した。 (SDS-PAGE and Western blot analysis 2)
PC12D cells were seeded at 0.67 × 10 6 cells / dish in a 35 mm dish, cultured in a growth medium for 2 days in a 5% CO 2 incubator, and then treated with the drug for 24 hours.
Each cell was treated and then washed with ice-cold PBS. Cell lysate (1 mM EDTA, 1% SDS, 10 mM NaF, 10 nM caliculin, 320 nM okadaic acid, 1 mM sodium orthovanadate, 1 mM p-APMSF, 10 μg / mL pepstatin A, 10 μg / mL Antipine, 10 μg / mL leupeptin, 10 μg / mL chymostatin, 10 μg / mL phosphoramidon, 10 mM HEPES, pH 7.5). Thereafter, the mixture was immediately boiled at 95 ° C. for 5 minutes to denature the protein, and then the DNA was sonicated and the centrifuged supernatant was prepared as a sample for SDS-PAGE.
実験結果はワンウェイANOVA(Bonferroni)を用いて評価した。有意水準を両側5%として検定し、p<0.05を有意とした。 (Statistical analysis 2)
The experimental results were evaluated using a one-way ANOVA (Bonferroni). The significance level was tested at 5% on both sides, and p <0.05 was considered significant.
本試験例において、本発明の陳皮エキスのパーキンソン病の症状改善作用として、ドーパミン生合成系への影響を指標にして検討した。具体的には、陳皮エキスによるチロシン・ヒドロキシラーゼ(TH)転写活性への影響についてドーパミン産生細胞であるPC12D細胞において、TH遺伝子プロモーター領域DNAレポータージーンを導入して、ルシフェラーゼ活性を測定して検討した。また、THタンパク質への影響を検討するために陳皮エキスで処置したPC12D細胞におけるTHタンパク質の発現をウェスタン・ブロット法で検討した。さらに、パーキンソン病モデルマウスにおいて黒質-線条体ドーパミン神経の変性による線条体でのドーパミン含有量の低下に対する陳皮エキスまたはノビレチンの薬効を検討するため、陳皮エキスまたはノビレチンの投与後にドーパミン含量を測定した。 [Test Example 4] Dopamine synthesis promoting effect of skin extract In this test example, the effect of the skin extract of the present invention on improving the symptoms of Parkinson's disease was examined using the effect on the dopamine biosynthesis system as an index. Specifically, the influence of the cinnamon extract on tyrosine hydroxylase (TH) transcriptional activity was examined by introducing a TH gene promoter region DNA reporter gene and measuring luciferase activity in PC12D cells, which are dopaminergic cells. In order to examine the influence on TH protein, the expression of TH protein in PC12D cells treated with crushed extract was examined by Western blotting. Furthermore, in order to investigate the medicinal effects of crustacean extract or nobiletin on the reduction of dopamine content in the striatum due to degeneration of substantia nigra-striatal dopamine nerve in Parkinson's disease model mice, It was measured.
8×104細胞/48ウェルプレートの密度にて増殖させたPC12D細胞を24時間培養した。細胞をルシフェラーゼレポーター構築体で19時間トランスフェクションし、次いで対照、陳皮1からの本発明の陳皮エキス(30, 60および120μg/ml)および従来の陳皮7のエキス(120μg/ml)またはノビレチン(2, 10および30μM)で24時間処理した。
図4Aに示されるように、PC12D細胞において、本発明の陳皮1のエキスは対照および従来の陳皮7のエキスと比較して、TH転写活性を顕著に上昇させた。また、ノビレチン換算2μMの陳皮1のエキス120μg/mlは、2μMおよび10μMのノビレチンよりも強力にTH転写活性を促進することが示された。 [Test Example 4-1] Promotion of TH transcription activity of skin extract in PC12D cells PC12D cells grown at a density of 8 × 10 4 cells / 48-well plate were cultured for 24 hours. Cells were transfected with a luciferase reporter construct for 19 hours, then control, a peel extract of the present invention (30, 60 and 120 μg / ml) from
As shown in FIG. 4A, in PC12D cells, the
PC12D細胞において、THタンパク質の発現の促進作用を検討した。0.67×106細胞/皿の密度にて増殖させたPC12D細胞を48時間培養した。細胞を対照または陳皮1からの陳皮エキス(60, 120, 240および480μg/ml)で24時間処理した。抗THおよび抗PKAα抗体を用いてウェスタン・ブロット分析を行った。ブロットは、最初に抗TH抗体について調べた後、剥ぎ取り、抗PKAα抗体で再度試験して等量のタンパク質が各レーンにて電気泳動されたことを確認した。
図4Bに示されるように、PC12D細胞において、陳皮エキスはTHタンパク質の発現を濃度依存的に上昇させた。 [Test Example 4-2] Effect of increasing the TH expression level of the peel extract in PC12D cells The effect of promoting the expression of TH protein in PC12D cells was examined. PC12D cells grown at a density of 0.67 × 10 6 cells / dish were cultured for 48 hours. Cells were treated with control or skin extract from skin 1 (60, 120, 240 and 480 μg / ml) for 24 hours. Western blot analysis was performed using anti-TH and anti-PKAα antibodies. The blot was first examined for anti-TH antibody, then stripped and retested with anti-PKAα antibody to confirm that an equal amount of protein was electrophoresed in each lane.
As shown in FIG. 4B, in PC12D cells, the crust extract increased the expression of TH protein in a concentration-dependent manner.
THがドーパミン合成の律速酵素であり、PC12D細胞がドーパミン合成分泌細胞であることから、ドーパミンの上昇作用を検討した。
PC12D細胞を35 mm皿に1.0×106細胞/皿で播種し、増殖培地上5%CO2インキュベーター中にて24時間培養した後、陳皮1および陳皮7を添加した細胞培養液をさらに24時間培養した。その後、37℃に加温した低K+緩衝液(140 mM NaCl, 4.7 mM KCl, 1.2 mM KH2PO4, 2.5 mM CaCl2, 1.2 mM MgSO4, 11 mMグルコースおよび15 mM HEPES-Tris, pH7.4)にて細胞を洗浄し、最終濃度が0.4Nになるように過塩素酸溶液を加え、1分間超音波にて細胞を破砕した。得られた溶液を遠心分離し、電気化学検出器を用いた高速液体クロマトグラフィー法によりドーパミンを分離定量した。
図4Cに示されるように、本発明の陳皮1のエキスは対照および従来の陳皮7のエキスと比較して、24時間および48時間のいずれの処置においてもドーパミン含有量を増加させた。 [Test Example 4-3] Dopamine content increasing action of skin extract in PC12D cells Since TH is a rate-determining enzyme for dopamine synthesis and PC12D cells are dopamine synthetic secreting cells, the increasing action of dopamine was examined.
PC12D cells were seeded at 1.0 × 10 6 cells / dish in a 35 mm dish and cultured in a 5% CO 2 incubator on a growth medium for 24 hours, and then the cell culture solution supplemented with
As shown in FIG. 4C, the
上記試験例において、本発明の陳皮エキスはPC12D細胞にてTH転写活性、TH発現量およびドーパミン含量をいずれも濃度依存的に上昇させ、その作用は等濃度のノビレチンと比較して予想外かつ格別に強力であった。
すなわち、本発明の陳皮エキスは、ノビレチンより顕著にドーパミン合成促進活性を示すことが明らかとなった。従来の陳皮由来のエキスでは当該活性は認められていないことから、本発明の陳皮エキスは高含有量のノビレチンとノビレチン以外の他の成分との相乗効果によりドーパミン合成促進活性を示したと考えられる。以上により本発明の陳皮エキスは、パーキンソン病治療薬としての薬効が期待される。 (Discussion 4)
In the above test examples, the skin extract of the present invention increases the TH transcriptional activity, TH expression level and dopamine content in PC12D cells in a concentration-dependent manner, and its action is unexpected and exceptional compared to nobiletin at an equal concentration. It was powerful.
That is, it was revealed that the skin extract of the present invention exhibits a dopamine synthesis promoting activity more markedly than nobiletin. Since the activity is not recognized in the conventional extract derived from dermis, it is considered that the dermis extract of the present invention exhibited a dopamine synthesis promoting activity due to a synergistic effect of a high content of nobiletin and other components other than nobiletin. As described above, the skin extract of the present invention is expected to have a medicinal effect as a therapeutic agent for Parkinson's disease.
実施例2で得られた橘皮3から本明細書記載の方法により抽出したエキスを用いて、海馬神経細胞におけるCRE転写活性を対照およびノビレチンと比較検討した。試験手順は試験例2と同様とした。結果を図5Aに示す。
なお、本試験例で用いた橘皮3エキス300μg/mLには、ノビレチンが約9.4μM含まれていた。
図5Aに示されるように、橘皮3のエキスは対照およびノビレチンと比較して、海馬神経細胞において顕著なCRE転写活性を示した。このことから、本発明の橘皮エキスは、アルツハイマー病治療薬としての薬効が期待される。 Test Example 5 CRE Transcriptional Activity of Tachibana Skin Extract in Hippocampal Nerve Cells Using the extract extracted from
The
As shown in FIG. 5A, the extract of
実施例2で得られた橘皮6から本明細書記載の方法により抽出したエキスを用いて、学習・記憶障害に対する本発明の橘皮エキスの作用を検討した。試験手順は試験例3と同様とした。結果を図5Bおよび図5Cに示す。
なお、橘皮エキス1.87 g/kgまたは3.73 g/kg(ノビレチン含量: それぞれ25 mg/kgまたは50 mg/kg)を1群ずつに、7日間連続的に経口投与した。
図5Bおよび図5Cに示されるように、本発明の橘皮エキスの慢性投与により学習・記憶障害を有意に改善することに加え、ノビレチンでは認められない記憶の獲得および保持能力を増強することが明らかとなった。 [Test Example 6] Tachibana peel extract learning / memory impairment improving effect (in vivo test)
Using the extract extracted from
Tachibana peel extract 1.87 g / kg or 3.73 g / kg (nobiletin content: 25 mg / kg or 50 mg / kg, respectively) was orally administered to each group continuously for 7 days.
As shown in FIG. 5B and FIG. 5C, in addition to significantly improving learning / memory impairment by chronic administration of the tachibana peel extract of the present invention, it is possible to enhance the ability to acquire and retain memory that is not observed with nobiletin. It became clear.
上記試験例の結果から、本発明の橘皮エキスは、アルツハイマー病治療薬としての薬効が期待される。 (Discussion 5)
From the results of the above test examples, the tachibana peel extract of the present invention is expected to have a medicinal effect as a therapeutic agent for Alzheimer's disease.
実施例2で得られた橘皮3から本明細書記載の方法により抽出したエキスを用いて、PC12D細胞におけるCRE転写活性を対照およびノビレチンと比較検討した。試験手順は試験例2と同様とした。結果を図5Dに示す。
なお、本試験例で用いた橘皮3エキス300μg/mLには、ノビレチンが約9.4μM含まれていた。
図5Dに示されるように、橘皮3のエキスは対照およびノビレチンと比較して、PC12D細胞において顕著なCRE転写活性を示した。 [Test Example 7] CRE transcription activity of Tachibana peel extract in PC12D cells Using the extract extracted from
The
As shown in FIG. 5D, the extract of
実施例2で得られた橘皮3から本明細書記載の方法により抽出したエキスを用いて、PC12D細胞におけるTH転写活性を対照およびノビレチンと比較検討した。試験手順は試験例4-1と同様とした。結果を図5Eに示す。
図5Eに示されるように、橘皮3のエキスは対照およびノビレチンと比較して、PC12D細胞において顕著なTH転写活性を示した。 [Test Example 8] TH transcription activity of Tachibana peel extract in PC12D cells Using the extract extracted from
As shown in FIG. 5E,
PC12D細胞において、GCH I量上昇作用を検討した。GCH Iは、THに必須の補酵素テトラヒドロビオプテリン(BH4)の生合成における律速酵素である。
PC12D細胞は120 μg/mL濃度の橘皮エキス、陳皮エキスおよびDMSO(対照)で48時間処理後、PBSで洗浄し、細胞溶解液(1 mM EDTA, 1% SDS, 10 mM HEPES, 10 mM NaF, 1 mM p-APMSF, 1 mMオルトバナジウム酸ナトリウム, 10 nMカリキュリン(calyculin), 320 μMオカダ酸, 10 μg/mLペプスタチン(pepstatin), 10 μg/mLアンチパイン, 10 μg/mLロイペプチン, 10 μg/mLキモスタチン(chymostatin), 10 μg/mLホスホラミドン, 240 pMシペルメトリン)中で溶解した。その後5分間95℃で煮沸し、ソニケート後、10000 x gで10分間遠心を行い、上清を用いてSDS-PAGEを行った。10 μgサンプルをゲル濃度12.5%のゲルにアプライした。その後PVDFメンブレンに36Vで2時間転写を行い、メンブレンは5%スキムミルクで1時間ブロッキングした。その後GCH I抗体(ABBIOTEC、1000倍希釈)を用い、それぞれ4℃で一晩反応させ、洗浄後さらにペルオキシダーゼ標識抗ウサギ2次抗体(CST、2000倍希釈)と反応させ、ECL法でバンドを検出した。結果を陳皮エキスと比較して図5Fに示す。
なお、本試験例で用いた橘皮エキス120μg/mLにはノビレチンが約3.76μM含まれており、陳皮エキス120μg/mLにはノビレチンが約2.02μM含まれていた。
図5Fに示されるように、PC12D細胞において、橘皮エキスおよび陳皮エキスは対照に比べてGCH Iの発現量を有意に上昇させた。さらに、橘皮エキスは陳皮エキスに比べてもGCH Iの発現量を有意に上昇させた。 [Test Example 9] GTP cyclohydrolase I (GCH I) level-increasing action of Tachibana bark extract in PC12D cells GPC I level-increasing action was examined in PC12D cells. GCH I is a rate-limiting enzyme in the biosynthesis of the coenzyme tetrahydrobiopterin (BH 4 ) essential for TH.
PC12D cells were treated with Tachibana peel extract, Chen peel extract and DMSO (control) at a concentration of 120 μg / mL for 48 hours, washed with PBS, and cell lysate (1 mM EDTA, 1% SDS, 10 mM HEPES, 10 mM NaF) , 1 mM p-APMSF, 1 mM sodium orthovanadate, 10 nM calyculin, 320 μM okadaic acid, 10 μg / mL pepstatin, 10 μg / mL antipine, 10 μg / mL leupeptin, 10 μg / mL chymostatin, 10 μg / mL phosphoramidon, 240 pM cypermethrin). Thereafter, the mixture was boiled at 95 ° C. for 5 minutes, sonicated, centrifuged at 10000 × g for 10 minutes, and SDS-PAGE was performed using the supernatant. A 10 μg sample was applied to a gel with a gel concentration of 12.5%. Thereafter, transcription was performed on a PVDF membrane at 36 V for 2 hours, and the membrane was blocked with 5% skim milk for 1 hour. After that, GCH I antibody (ABBIOTEC, diluted 1000 times) was used to react overnight at 4 ° C, washed and further reacted with peroxidase-labeled anti-rabbit secondary antibody (CST, diluted 2000 times), and the band was detected by ECL method. did. The results are shown in FIG.
The Tachibana peel extract 120 μg / mL used in this test example contained about 3.76 μM nobiletin, and the
As shown in FIG. 5F, in PC12D cells, Tachibana peel extract and Chen peel extract significantly increased the expression level of GCH I compared to the control. Furthermore, Tachibana peel extract significantly increased the expression level of GCH I compared to Chen peel extract.
THの活性発現に必須な補酵素BH4は、ミカエリス定数が示す量より細胞内のBH4量が高くなると、THタンパク量が上昇して、THの酵素活性が増大し、ドーパミンの合成が促進される。
上記試験例の結果から、本発明の橘皮エキスは、GCH Iの発現量を有意に上昇させることから、THの活性発現に必須な補酵素BH4合成の律速酵素であるGCH I量を増加させることで、TH活性の増大を引き起こし、その結果として、ドーパミンの産生および分泌の促進が推測され、パーキンソン病治療薬としての薬効が期待される。 (Discussion 6)
Coenzyme BH4, which is essential for the expression of TH activity, increases the amount of TH protein, increases the TH enzyme activity, and promotes dopamine synthesis when the intracellular BH4 content is higher than the amount indicated by the Michaelis constant. .
From the results of the above test examples, the Tachibana peel extract of the present invention significantly increases the expression level of GCH I, and therefore increases the amount of GCH I, which is the rate-determining enzyme of coenzyme BH4 synthesis that is essential for the expression of TH activity. As a result, TH activity is increased, and as a result, production and secretion of dopamine are presumed to be promoted, and a medicinal effect as a Parkinson's disease therapeutic agent is expected.
実施例2で得られたタチバナの葉1~3から本明細書記載の方法により抽出したエキスを用いて、海馬神経細胞におけるCRE転写活性を対照およびノビレチンと比較検討した。試験手順は試験例2と同様とした。結果を図6Aに示す。
なお、本試験例で用いたタチバナの葉1由来エキス300μg/mLにはノビレチンが約4.9μM、タチバナの葉2由来エキス300μg/mLにはノビレチンが約8.5μM、タチバナの葉3由来エキス300μg/mLにはノビレチンが約6.9μM含まれていた。
図6Aに示されるように、タチバナの葉由来エキスは対照およびノビレチンと比較して、海馬神経細胞において顕著なCRE転写活性を示した。 [Test Example 10] CRE transcriptional activity of extracts derived from leaves of Tachibana in hippocampal neurons In extracts of Tachibana leaves 1 to 3 obtained in Example 2 by the method described in this specification, CRE transcriptional activity was compared with control and nobiletin. The test procedure was the same as in Test Example 2. The results are shown in FIG. 6A.
In addition, nobiletin is about 4.9 μM for
As shown in FIG. 6A, Tachibana leaf-derived extract showed significant CRE transcriptional activity in hippocampal neurons compared to control and nobiletin.
実施例2で得られたタチバナの葉1~3から本明細書記載の方法により抽出したエキスを用いて、PC12D細胞におけるCRE転写活性を対照およびノビレチンと比較検討した。試験手順は試験例2と同様とした。結果を図6Bに示す。
なお、本試験例で用いたタチバナの葉1由来エキス300μg/mLにはノビレチンが約4.9μM、タチバナの葉2由来エキス300μg/mLにはノビレチンが約8.5μM、タチバナの葉3由来エキス300μg/mLにはノビレチンが約6.9μM含まれていた。
図6Bに示されるように、タチバナの葉由来エキスは対照およびノビレチンと比較して、PC12D細胞において顕著なCRE転写活性を示した。 [Test Example 11] CRE transcriptional activity of Tachibana leaf-derived extract in PC12D cells CRE transcription in PC12D cells using extracts extracted from Tachibana leaves 1 to 3 obtained in Example 2 by the method described herein The activity was compared with the control and nobiletin. The test procedure was the same as in Test Example 2. The result is shown in FIG. 6B.
In addition, nobiletin is about 4.9 μM for
As shown in FIG. 6B, Tachibana leaf-derived extract showed significant CRE transcriptional activity in PC12D cells compared to control and nobiletin.
上記試験例の結果から、本発明のタチバナの葉由来エキスは、ノビレチン含有量から予測される活性を超えた顕著なCRE転写活性を示しており、アルツハイマー病治療薬およびパーキンソン病治療薬としての薬効が期待される。 (Discussion 7)
From the results of the above test examples, the extract derived from the leaf of Tachibana of the present invention shows a remarkable CRE transcriptional activity that exceeds the activity predicted from the content of nobiletin, and is effective as a therapeutic agent for Alzheimer's disease and Parkinson's disease. There is expected.
実施例2で得られたオオベニミカン1から本明細書記載の方法により抽出したエキスを用いて、海馬神経細胞におけるCRE転写活性を対照およびノビレチンと比較検討した。試験手順は試験例2と同様とした。結果を図7Aに示す。
なお、本試験例で用いたオオベニミカン1果皮由来エキス100μg/mLにはノビレチンが約3.4μM、300μg/mLにはノビレチンが約10.1μM含まれていた。
図7Aに示されるように、オオベニミカン果皮由来エキスは対照およびノビレチンと比較して、海馬神経細胞において顕著なCRE転写活性を示した。 Test Example 12 CRE Transcriptional Activity of Citrus Citrus Skin Extract in Hippocampal Nerve Cells Using the extract extracted from
In addition, 100 μg / mL of the extract derived from 1 pericarp used in this test example contained about 3.4 μM nobiletin, and about 10.1 μM nobiletin in 300 μg / mL.
As shown in FIG. 7A, the extract derived from the citrus pericarp showed significant CRE transcriptional activity in hippocampal neurons as compared to the control and nobiletin.
上記試験例の結果から、本発明のオオベニミカン果皮由来エキスは、ノビレチン含有量から予測される活性を超えた顕著なCRE転写活性を示しており、アルツハイマー病治療薬としての薬効が期待される。 (Discussion 6)
From the results of the above-mentioned test examples, the extract derived from the fruit peel of the present invention shows a remarkable CRE transcriptional activity exceeding the activity predicted from the content of nobiletin, and is expected to be effective as a therapeutic agent for Alzheimer's disease.
実施例2で得られたオオベニミカン1から本明細書記載の方法により抽出したエキスを用いて、PC12D細胞におけるCRE転写活性を対照およびノビレチンと比較検討した。試験手順は試験例2と同様とした。結果を図7Bに示す。
なお、本試験例で用いたオオベニミカン果皮由来エキス100μg/mLにはノビレチンが約3.4μM含まれていた。
図7Bに示されるように、オオベニミカン果皮由来エキスは対照およびノビレチンと比較して、PC12D細胞において顕著なCRE転写活性を示した。 [Test Example 13] CRE transcriptional activity of the extract derived from Citrus unshiu peel in PC12D cells Using the extract extracted from
In addition, about 3.4 μM of nobiletin was contained in 100 μg / mL of the extract derived from the fruit cuttlefish used in this test example.
As shown in FIG. 7B, the extract derived from the citrus pericarp showed significant CRE transcriptional activity in PC12D cells compared to the control and nobiletin.
実施例2で得られたオオベニミカン1から本明細書記載の方法により抽出したエキスを用いて、PC12D細胞におけるTH転写活性を対照およびノビレチンと比較検討した。試験手順は試験例4-1と同様とした。結果を図7Cに示す。
なお、本試験例で用いたオオベニミカン果皮由来エキス100μg/mLにはノビレチンが約3.4μM含まれていた。
図7Cに示されるように、オオベニミカン果皮由来エキスは対照およびノビレチンと比較して、PC12D細胞において顕著なTH転写活性を示した。 [Test Example 14] TH transcriptional activity of Citrus pericarp peel-derived extract in PC12D cells Using the extract extracted from
In addition, about 3.4 μM of nobiletin was contained in 100 μg / mL of the extract derived from the fruit cuttlefish used in this test example.
As shown in FIG. 7C, the extract derived from the citrus pericarp showed significant TH transcriptional activity in PC12D cells compared to the control and nobiletin.
上記試験例の結果から、本発明のオオベニミカン果皮由来エキスは、ノビレチン含有量から予測される活性を超えた顕著なCRE転写活性およびTH転写活性を示しており、パーキンソン病治療薬としての薬効が期待される。 (Discussion 7)
From the results of the above-mentioned test examples, the extract derived from Papilio mandarin peel of the present invention exhibits remarkable CRE transcription activity and TH transcription activity exceeding the activity predicted from the content of nobiletin, and is expected to be effective as a treatment for Parkinson's disease. Is done.
以下の手順により、本発明の陳皮、橘皮またはタチバナの葉を含有する医薬品または食品を製造した。 [Production Example] Production of Drug and Food of the Present Invention A drug or food containing the skin, tachibana peel or tachibana leaf of the present invention was produced by the following procedure.
陳皮3.0 g、トウキ3.0 g、チョウトウコウ3.0 g、センキュウ3.0 g、ビャクジュツ4.0 g、ブクリョウ4.0 g、サイコ2.0 g、カンゾウ1.5 gおよびハンゲ5.0 g(合計28.5 g)の比率で、合計量が200~800 kgになるよう生薬を配合し、水2000~8000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~5000 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、エキス製剤(抑肝散加陳皮半夏エキス)を得た。得られた抑肝散加陳皮半夏エキス6.1 gに対し、ステアリン酸マグネシウム、トウモロコシデンプン、乳糖、プルランおよびメタケイ酸アルミン酸マグネシウムからなる添加物2.9 gの比率で、合計量が50~400 kgの範囲で配合し、4 rpm、20分間混合(容器回転型混合機)し、ロール圧490~2500 Paで成型して整粒(乾式造粒装置)し、篩30号~50号間にて粒子分級(カセットスクリーン)し、細粒剤Aを得た。 [Production Example 1] Fine granules A
A total of 200 to 200 g in proportions of 3.0 g of leather, 3.0 g of touki, 3.0 g of butterfly, 3.0 g of cucumber, 4.0 g of peanuts, 4.0 g of bakuryo, 2.0 g of psycho, 1.5 g of licorice and 5.0 g of hanger Herbal medicine was blended to 800 kg and extracted (extracted can) with water 2000-8000 L at 60-100 ° C for 30-180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 5000 rpm, solid-liquid separation was performed, followed by concentration to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator). The concentrated solution was spray-dried (spray dryer) at a rotational speed of 10000-20000 rpm, a supply temperature of 130-180 ° C, and an exhaust temperature of 60-120 ° C to obtain an extract formulation (Yokukansanka-Chanka summer extract). . A total amount of 50-400 kg is obtained at a ratio of 2.9 g of an additive consisting of magnesium stearate, corn starch, lactose, pullulan and magnesium aluminate metasilicate to 6.1 g of the obtained yokukansan kachu semi-summer extract. Mix in the range, mix at 4 rpm for 20 minutes (container rotation type mixer), mold at roll pressure 490-2500 Pa, size control (dry granulator), particles between sieves 30-50 Classification (cassette screen) was performed to obtain fine granules A.
橘皮3.0 g、ビンロウジ4.0 g、コウボク3.0 g、ケイヒ3.0 g、ソヨウ1.5 g、カンゾウ1.0 g、ダイオウ1.0 g、ショウキョウ1.0 g、モッコウ1.0 g、ゴシュユ1.0 gおよびブクリョウ3.0 g(合計22.5 g)の比率で、合計量が200~800 kgになるよう生薬を配合し、水2000~8000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~5000 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、エキス製剤(九味檳榔湯エキス)を得た。得られた九味檳榔湯エキス3.7 gに対し、ステアリン酸マグネシウム、トウモロコシデンプン、乳糖、プルランおよびメタケイ酸アルミン酸マグネシウムからなる添加物2.3 gの比率で、合計量が50~400 kgの範囲で配合し、4 rpm、20分間混合(容器回転型混合機)し、ロール圧490~2500 Paで成型して整粒(乾式造粒装置)し、篩30号~50号間にて粒子分級(カセットスクリーン)し、細粒剤Bを得た。 [Production Example 2] Fine granule B
Tachibana bark 3.0 g, Betel loaf 4.0 g, Koboku 3.0 g, Keihi 3.0 g, Soyo 1.5 g, Licorice 1.0 g, Daio 1.0 g, Pepper 1.0 g, Mokko 1.0 g, Goshuyu 1.0 g and Bukkyo 3.0 g (total 22.5 g) Herbal medicines were blended so that the total amount would be 200 to 800 kg, and extracted (extracted can) with 2000 to 8000 L of water at 60 to 100 ° C. for 30 to 180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 5000 rpm, solid-liquid separation was performed, followed by concentration to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator). The concentrated solution was spray-dried (spray dryer) at a rotational speed of 10,000 to 20000 rpm, an air supply temperature of 130 to 180 ° C., and an exhaust temperature of 60 to 120 ° C. to obtain an extract formulation (Kumisoto extract). Formulated in a range of 50-400 kg in total with a ratio of 2.3 g of additive consisting of magnesium stearate, corn starch, lactose, pullulan and magnesium aluminate metasilicate to 3.7 g of the resulting Kumi miso extract Mix at 4 rpm for 20 minutes (container rotation type mixer), mold at roll pressure of 490-2500 Pa, adjust particle size (dry granulator), and classify particles between sieves 30-50 (cassette) Screen) to obtain fine granule B.
陳皮2.4 g、チョウトウコウ2.4 g、ハンゲ2.4 g、バクモンドウ2.4 g、ブクリョウ2.4 g、ニンジン1.6 g、ボウフウ1.6 g、キクカ1.6 g、カンゾウ0.8 g、ショウキョウ0.8 gおよびセッコウ4.0 g(合計22.4 g)の比率で、合計量が200~800 kgになるよう生薬を配合し、水2000~8000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~5000 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、エキス製剤(釣藤散エキス)を得た。得られた釣藤散エキス4.48 gに対し、含水二酸化ケイ素、ステアリン酸マグネシウムおよびトウモロコシデンプンからなる添加物1.52 gの比率で、合計量が50~400 kgの範囲で配合し、4 rpm、20分間混合(容器回転型混合機)し、ロール圧490~2500 Paで成型して整粒(乾式造粒装置)し、篩30号~50号間にて粒子分級(カセットスクリーン)し、細粒剤Cを得た。 [Production Example 3] Fine granule C
Chestnut 2.4 g, Butterflyfish 2.4 g, Hange 2.4 g, Bakumondou 2.4 g, Bukkuri 2.4 g, Carrot 1.6 g, Bow Fu 1.6 g, Kikuka 1.6 g, Licorice 0.8 g, Pepper 0.8 g and Gypsum 4.0 g (total 22.4 g) Herbal medicines were blended so that the total amount would be 200 to 800 kg, and extracted (extracted can) with 2000 to 8000 L of water at 60 to 100 ° C. for 30 to 180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 5000 rpm, solid-liquid separation was performed, followed by concentration to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator). The concentrated solution was spray-dried (spray dryer) at a rotational speed of 10000 to 20000 rpm, an air supply temperature of 130 to 180 ° C., and an exhaust temperature of 60 to 120 ° C. to obtain an extract formulation (Chitosan extract). 4.48 g of Chotosan extract obtained in a ratio of 1.52 g of additive consisting of hydrous silicon dioxide, magnesium stearate and corn starch in a total amount of 50-400 kg, 4 rpm for 20 minutes Mix (container rotating type mixer), mold at roll pressure of 490-2500 Pa, adjust particle size (dry granulator), classify particles between sieves 30-50 (cassette screen), fine granules C was obtained.
陳皮または橘皮を850~4750μm角に切断し、切断した陳皮または橘皮3~7 gを紙または布の袋に入れて茶剤とした。
得られた茶剤を成人1日あたり3回(1袋/回)で浸出して服用する。 [Production Example 4] Tea preparation The skin or tachibana peel was cut into 850 to 4750 μm squares, and 3 to 7 g of the cut peel or tachibana peel was placed in a paper or cloth bag to give a tea preparation.
The obtained tea is leached 3 times a day (1 bag / time) for adults.
陳皮または橘皮を回転数2000~3500 rpm、目開き0.5~3.0 mmで粗砕(粉砕機)し、さらに回転数5000~8000 rpm、目開き150μmで粉砕(製粉機)し、生薬末とした。得られた陳皮または橘皮の生薬末10 gに対し、含水二酸化ケイ素、ステアリン酸マグネシウムおよびトウモロコシデンプンからなる添加物9.2 gの比率で、合計量が50~400 kgの範囲で配合し、回転数4 rpm、20分間混合(容器回転型混合機)し、ロール圧490~2500 Paで成型して整粒(乾式造粒装置)し、篩30号~42号間にて粒子分級(カセットスクリーン)し、細粒剤Dを得た。 [Production Example 5] Herbal powder-fine granules D
Crushed crust or tachibana peel at a rotational speed of 2000-3500 rpm and an aperture of 0.5-3.0 mm (pulverizer), and further pulverized at a rotational speed of 5000-8000 rpm and an aperture of 150 μm (milling machine) to obtain a crude drug powder. . The ratio of 9.2 g of additive consisting of hydrous silicon dioxide, magnesium stearate and corn starch to 10 g of the crude or powdered raw material of cinnamon bark is blended in a total amount of 50 to 400 kg, and the number of rotations Mix at 4 rpm for 20 minutes (container rotation type mixer), mold at roll pressure 490-2500 Pa, adjust size (dry granulator), and classify particles between sieves 30-42 (cassette screen) As a result, a fine granule D was obtained.
陳皮または橘皮10~30 gを20倍量の水200~600 mLで、100℃1時間煎じて水を半量にし、ろ過して煎剤とした。
得られた煎剤100~300 mLには、陳皮または橘皮10~30 gが含有されており、目安として煎剤100~300 mLを成人1日あたり3回に分けて服用する。 [Production Example 6] Decoction 10-30 g of decoction or tachibana peel was decocted with 200-600 mL of 20 times the amount of water at 100 ° C. for 1 hour, and the water was halved, followed by filtration to obtain a decoction.
The resulting decoction (100-300 mL) contains 10-30 g of decoction or tachibana peel. As a guideline, 100-300 mL of decoction is taken in three divided doses per day for adults.
陳皮または橘皮50 kgを水500~2000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~2500 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、陳皮または橘皮のエキスを得た。得られたエキス7.2 gに対し、微粒二酸化ケイ素およびショ糖脂肪酸エステルからなる添加物1.8 gの比率で、合計量が50~400 kgの範囲で配合し、回転数4 rpm、20分間混合(容器回転型混合機)し、ロール圧490~2500 Paで成型して整粒(乾式造粒装置)し、篩30号~42号間にて粒子分級(カセットスクリーン)し、細粒剤Eを得た。 [Production Example 7] Fine granule E
50 kg of peel or tachibana peel was extracted (extraction can) at 500 to 2000 L of water at 60 to 100 ° C. for 30 to 180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 2500 rpm, solid-liquid separation was performed, and the solution was concentrated to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator). The concentrate was spray-dried (spray dryer) at a rotational speed of 10000 to 20000 rpm, an air supply temperature of 130 to 180 ° C., and an exhaust temperature of 60 to 120 ° C. to obtain an extract of crust or tachibana peel. To 7.2 g of the obtained extract, blended in a ratio of 1.8 g of additive consisting of fine silicon dioxide and sucrose fatty acid ester in a total amount of 50 to 400 kg, and mixed for 20 minutes at 4 rpm Rotating type mixer), molded at roll pressure of 490-2500 Pa, sized (dry granulator), classified between sieves 30-42 (cassette screen), and obtained fine granule E It was.
陳皮または橘皮50 kgを水500~2000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~2500 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、陳皮または橘皮のエキスを得た。得られたエキス10 gに対し、メタケイ酸アルミン酸マグネシウム、合成ケイ酸アルミニウム・ヒドロキシプロピルスターチ・結晶セルロース、トウモロコシデンプン、軽質無水ケイ酸、ステアリン酸マグネシウムおよびカルメロ-スカルシウムからなる添加物4.75 gの比率で、合計量が50~400 kgの範囲で配合し、回転数4 rpm、20分間混合(容器回転型混合機)し、ロール圧490~2500 Paで成型して整粒(乾式造粒装置)し、篩30号~42号間にて粒子分級(カセットスクリーン)し、細粒を得た。この細粒400 mgを当分野にて通常用いられる市販のカプセルに充填(カプセル充填機)し、カプセル剤を得た。 [Production Example 8]
陳皮または橘皮50 kgを水500~2000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~2500 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、陳皮または橘皮のエキスを得た。得られたエキス10 gに対し、マルトース、乳糖、メタケイ酸アルミン酸マグネシウムおよびステアリン酸マグネシウムからなる添加物9.1 gの比率で、合計量が50~400 kgの範囲で配合し、回転数4 rpm、20分間混合(容器回転型混合機)し、回転数20~55 rpm、一次圧縮0.6 mm以上、二次圧縮0.4 mm以上で打錠(高速回転式打錠機)し、1錠350 mgの素錠を得た。ヒドロキシプロピルメチルセルロース、タルク、酸化チタンおよびカラメルからなるコーティング剤1.1 gを水/エタノール混液(3:7~10:0)に溶解または懸濁し、排気温40~55℃、流量200~400 g/mL、回転数4~6 rpmでコーティング(コーティング機)し、微量のカルナウバロウとサラシミツロウを用いて回転数1 rpm、排気温20~30℃で艶出し(コーティング機)を行い、1錠370 mgのコート錠剤とした。 [Production Example 9]
陳皮または橘皮50 kgを水500~2000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~2500 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、陳皮または橘皮のエキスを得た。得られたエキス10 gに対し、トウモロコシデンプン、マルトース、含水二酸化ケイ素およびステアリン酸マグネシウムからなる添加物15 gの比率で、合計量が50~400 kgの範囲で配合し、回転数4 rpm、20分間混合(容器回転型混合機)し、ロール圧490~2500 Paで成型して整粒(乾式造粒装置)し、篩30号~42号間にて粒子分級(カセットスクリーン)し、細粒剤Fを得た。 [Production Example 10] Fine granule F
50 kg of peel or tachibana peel was extracted (extraction can) at 500 to 2000 L of water at 60 to 100 ° C. for 30 to 180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 2500 rpm, solid-liquid separation was performed, and the solution was concentrated to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator). The concentrate was spray-dried (spray dryer) at a rotational speed of 10000 to 20000 rpm, an air supply temperature of 130 to 180 ° C., and an exhaust temperature of 60 to 120 ° C. to obtain an extract of crust or tachibana peel. 10 g of the resulting extract is blended in a ratio of 15 g of an additive consisting of corn starch, maltose, hydrous silicon dioxide and magnesium stearate in a total amount in the range of 50 to 400 kg, rotating at 4 rpm, 20 Mix for a minute (container rotation type mixer), mold at roll pressure of 490-2500 Pa, adjust the size (dry granulator), classify particles between sieves 30-42 (cassette screen), fine particles Agent F was obtained.
陳皮または橘皮50 kgを水500~2000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~2500 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、陳皮または橘皮のエキスを得た。得られたエキス10 gに対し、カルメロースカルシウム、含水二酸化ケイ素、メタケイ酸アルミン酸マグネシウムおよびステアリン酸マグネシウムからなる添加物40 gの比率で、合計量が50~400 kgの範囲で配合し、回転数4 rpm、20分間混合(容器回転型混合機)し、ロール圧490~2500 Paで成型して整粒(乾式造粒装置)し、篩12号~42号間にて粒子分級(カセットスクリーン)し、顆粒剤を得た。 [Production Example 11]
陳皮または橘皮50 kgを水500~2000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~2500 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、陳皮または橘皮のエキスを得た。得られたエキス10 gに対し、カラギナン、ローカストビーンガム、還元麦芽糖液糖、キシリトール、アップル香料、レシチンおよびクエン酸からなる添加物40 gの比率で、合計量が10~30 kgの範囲で配合し、回転数50~150 rpmで混合(混合攪拌機)し、等量の80℃以上の温水を攪拌しながら加え、15℃以下に冷却してゼリー剤とした。 [Production Example 12]
陳皮または橘皮50 kgを水500~2000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~2500 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、陳皮または橘皮のエキスを得た。得られたエキス3.6 gに対し、微粒二酸化ケイ素およびショ糖脂肪酸エステルからなる添加物0.9 gの比率で、合計量が50~400 kgの範囲で配合し、回転数4 rpm、20分間混合(容器回転型混合機)し、ロール圧490~2500 Paで成型して整粒(乾式造粒装置)し、篩30号~42号間にて粒子分級(カセットスクリーン)し、細粒を得た。 [Production Example 13] 50 kg of fine grained peel or tachibana peel was extracted (extracted can) with 500 to 2000 L of water at 60 to 100 ° C for 30 to 180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 2500 rpm, solid-liquid separation was performed, and the solution was concentrated to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator). The concentrate was spray-dried (spray dryer) at a rotational speed of 10000 to 20000 rpm, an air supply temperature of 130 to 180 ° C., and an exhaust temperature of 60 to 120 ° C. to obtain an extract of crust or tachibana peel. The resulting extract (3.6 g) is blended at a ratio of 0.9 g of additive consisting of finely divided silicon dioxide and sucrose fatty acid ester in a total amount of 50 to 400 kg and mixed for 20 minutes at 4 rpm. Rotating mixer), molded at a roll pressure of 490-2500 Pa, sized (dry granulator), classified between sieves 30-42 (cassette screen) to obtain fine granules.
陳皮または橘皮50 kgを水500~2000 Lにて、60~100℃、30~180分間抽出(抽出缶)した。回転数1000~2500 rpmでろ過(遠心分離機)して固液分離した後、8 kPa以下の減圧下で濃度約10~40%まで濃縮(コイル回転型濃縮機)した。濃縮液を、回転数10000~20000 rpm、給気温度130~180℃、排気温度60~120℃で噴霧乾燥(噴霧乾燥機)し、陳皮または橘皮のエキスを得た。得られたエキス10 gに対し、レシチン、還元麦芽糖水飴、アスパルテーム、アスコルビン酸、クエン酸三ナトリウム、ゲル化剤(FG-2266、新田ゼラチン株式会社)およびオレンジ香料からなる添加物15.9 gの比率で、合計量が50~400 kgの範囲で配合し、回転数4 rpm、20分間混合(容器回転型混合機)してゼリーの素とした。 [Production Example 14] 50 kg of jelly skin or tachibana peel was extracted (extraction can) at 500 to 2000 L of water at 60 to 100 ° C for 30 to 180 minutes. After filtration (centrifugation) at a rotational speed of 1000 to 2500 rpm, solid-liquid separation was performed, and the solution was concentrated to a concentration of about 10 to 40% under reduced pressure of 8 kPa or less (coil rotation type concentrator). The concentrate was spray-dried (spray dryer) at a rotational speed of 10000 to 20000 rpm, an air supply temperature of 130 to 180 ° C., and an exhaust temperature of 60 to 120 ° C. to obtain an extract of crust or tachibana peel. Ratio of 15.9 g of additive consisting of lecithin, reduced maltose starch syrup, aspartame, ascorbic acid, trisodium citrate, gelling agent (FG-2266, Nitta Gelatin Co., Ltd.) and orange flavor to 10 g of the obtained extract Then, the total amount was blended in the range of 50 to 400 kg, and mixed at a rotational speed of 4 rpm for 20 minutes (container rotating type mixer) to obtain a jelly base.
タチバナの葉を850~4750μm角に切断し、切断したタチバナの葉3~7 gを紙または布の袋に入れるか、またはそのままの状態で茶とした。
目安として、得られた茶を成人1日あたり3回~5回(1袋または3~7 g/回)にてお湯で淹れて摂取する。 [Production Example 15] Tea Leaves of tachibana were cut into 850 to 4750 μm squares, and 3 to 7 g of the cut leaves of tachibana were put in a paper or cloth bag or used as tea as it was.
As a guideline, drink the tea obtained in
陳皮または橘皮200 gおよび食塩2 gを十分に浸る程度の水に入れ、蓋をして15分間沸騰させた後、お湯を捨て、冷水にくぐらせた。グラニュー糖200 gおよび水100 mLを混ぜ合わせて沸騰させた混合溶液に、水分をきった前記陳皮または橘皮を入れ、時々混ぜながら皮に透明感が出てくるまで煮た。蓋をせず、ごく弱火で40~50分間焦げ付かないように、時々混ぜながら煮詰めた。煮汁を除いて水分をよくきった後、熱いうちに取り出して、広げて乾かした。得られた皮にグラニュー糖20 gをまぶしてピールとした。
得られたピール21.1 gには、陳皮または橘皮10 gが含有されており、目安として成人1日あたりピール21.1~42.2 gを摂取する。 [Production Example 16] Peel 200 g of peel or tachibana peel and 2 g of salt were placed in water that was sufficiently immersed, covered and boiled for 15 minutes, then the hot water was discarded and cold water was passed through. Into a mixed solution in which 200 g of granulated sugar and 100 mL of water were mixed and boiled, the moistened skin or tachibana bark was added and boiled with occasional mixing until the skin became clear. It was boiled with occasional mixing so as not to burn for 40-50 minutes on a very low heat without a lid. After removing the broth and thoroughly draining the water, it was taken out while it was hot, spread and dried. The obtained skin was coated with 20 g of granulated sugar to make a peel.
The obtained peel 21.1 g contains 10 g of cinnamon or tachibana peel, and 21.1-42.2 g of peel per adult is taken as a guideline.
陳皮または橘皮100 gに100 mLのお湯(100℃)をかけ、一晩容器に入れて蒸らし、ミキサーで粉砕した。1 Lの水を加え、強火で1時間30分煮込んだ。グラニュー糖25~30 gを加え、中火で10~15分間煮込んだ後、弱火にし、グラニュー糖25~30 gを加えて溶解させた。さらに、グラニュー糖25~30 gを加えて溶解させ、70℃以下まで冷却した後、ハチミツ60 g、ペクチン1.7 gおよびアスコルビン酸1.7 gを加え攪拌してジャムとした。
得られたジャム100 gには、陳皮または橘皮20 gが含有されており、目安として成人1日あたりジャム25~50 gを摂取する。 [Production Example 17] 100 mL hot water (100 ° C) was applied to 100 g jam jam or tachibana peel, steamed in a container overnight, and pulverized with a mixer. 1 L of water was added and simmered on high heat for 1
100 g of the obtained jam contains 20 g of Chen or Tachibana, and as a guideline, take 25-50 g of jam per day for adults.
タチバナの葉30 kgを水300 Lにて、60~100℃、5~30分間抽出(抽出缶)した。回転数1000~2500 rpmでろ過(遠心分離機)して固液分離し、ろ液に0.2 g/Lのアスコルビン酸を添加し、茶とした。
得られた茶500 mLには、タチバナの葉5 gが含有されており、目安として成人1日あたり茶500 mLを摂取する。 [Production Example 18]
The obtained
Claims (21)
- 乾燥植物組織100重量%に対してノビレチンを0.4重量%以上含有する、中枢神経変性疾患の改善のための柑橘類の果皮の乾燥植物組織。 Dry plant tissue of citrus peel for improvement of central neurodegenerative diseases, containing 0.4% by weight or more of nobiletin to 100% by weight of dry plant tissue.
- 乾燥植物組織100重量%に対してノビレチンを0.3重量%以上含有する、中枢神経変性疾患の改善のための柑橘類の葉の乾燥植物組織。 Dry plant tissue of citrus leaves for improving central neurodegenerative diseases, containing 0.3% by weight or more of nobiletin with respect to 100% by weight of dry plant tissue.
- ノビレチン/ナリルチンの含有重量比が1.0以上である、請求項1または2のいずれか記載の乾燥植物組織。 The dried plant tissue according to claim 1 or 2, wherein the weight ratio of nobiletin / nariltin is 1.0 or more.
- 中枢神経変性疾患がアルツハイマー病および/またはパーキンソン病である、請求項1または2のいずれか記載の乾燥植物組織。 The dry plant tissue according to claim 1 or 2, wherein the central neurodegenerative disease is Alzheimer's disease and / or Parkinson's disease.
- 乾燥植物組織100重量%に対してノビレチンを0.3~2.0重量%含有し、かつ、ノビレチン/ナリルチンの含有重量比が1.0~23.0である、請求項4記載の乾燥植物組織。 The dried plant according to claim 4, wherein 0.3 to 2.0% by weight of nobiletin is contained with respect to 100% by weight of the dried plant tissue, and the weight ratio of nobiletin / nariltin is 1.0 to 23.0. Organization.
- 中枢神経変性疾患の改善がcAMP応答配列依存的転写活性の増大、記憶の獲得、保持および想起能力の増強、チロシン・ヒドロキシラーゼ転写活性の促進、チロシン・ヒドロキシラーゼ発現量の上昇、ドーパミン合成能の促進、またはドーパミン分泌の促進によりもたらされる、請求項1または2のいずれか記載の乾燥植物組織。 Improvement of central neurodegenerative diseases increases cAMP responsive element-dependent transcriptional activity, acquisition of memory, enhancement of retention and recall ability, promotion of tyrosine hydroxylase transcriptional activity, increase of tyrosine hydroxylase expression level, ability of dopamine synthesis The dried plant tissue according to claim 1 or 2 brought about by promotion or promotion of dopamine secretion.
- 植物組織抽出物100重量%に対してノビレチンを0.6重量%以上含有する、中枢神経変性疾患の改善のための柑橘類の植物組織抽出物。 Citrus plant tissue extract for improving central neurodegenerative diseases, containing 0.6% by weight or more of nobiletin with respect to 100% by weight of plant tissue extract.
- ノビレチン/ナリルチンの含有重量比が2.0以上である、請求項7記載の植物組織抽出物。 The plant tissue extract according to claim 7, wherein the weight ratio of nobiletin / nariltin is 2.0 or more.
- 請求項1または2のいずれか記載の乾燥植物組織から水抽出により得られる、請求項7記載の植物組織抽出物。 The plant tissue extract according to claim 7, which is obtained by water extraction from the dried plant tissue according to any one of claims 1 and 2.
- 中枢神経変性疾患がアルツハイマー病および/またはパーキンソン病である、請求項7記載の植物組織抽出物。 The plant tissue extract according to claim 7, wherein the central neurodegenerative disease is Alzheimer's disease and / or Parkinson's disease.
- 植物組織抽出物100重量%に対してノビレチンを0.6~3.0重量%含有し、かつ、ノビレチン/ナリルチンの含有重量比が2.0~14.0である、請求項10記載の植物組織抽出物。 The plant according to claim 10, which contains 0.6 to 3.0% by weight of nobiletin and 100 to 14.0% by weight of nobiletin / nariltin based on 100% by weight of the plant tissue extract. Tissue extract.
- 中枢神経変性疾患の改善がcAMP応答配列依存的転写活性の増大、記憶の獲得、保持および想起能力の増強、チロシン・ヒドロキシラーゼ転写活性の促進、チロシン・ヒドロキシラーゼ発現量の上昇、ドーパミン合成能の促進、またはドーパミン分泌の促進によりもたらされる、請求項7記載の植物組織抽出物。 Improvement of central neurodegenerative diseases increases cAMP responsive element-dependent transcriptional activity, acquisition of memory, enhancement of retention and recall ability, promotion of tyrosine hydroxylase transcriptional activity, increase of tyrosine hydroxylase expression level, ability of dopamine synthesis The plant tissue extract according to claim 7, which is brought about by promotion or promotion of dopamine secretion.
- 柑橘類がCitrus reticulata Blanco(Rutaceae)、タチバナまたはオオベニミカンである、請求項1または2のいずれか記載の乾燥植物組織。 The dried plant tissue according to any one of claims 1 and 2, wherein the citrus fruit is Citrus reticulata Blanco (Rutaceae), Tachibana or Giant orange.
- 柑橘類がCitrus reticulata Blanco(Rutaceae)、タチバナまたはオオベニミカンである、請求項7記載の植物組織抽出物。 The plant tissue extract according to claim 7, wherein the citrus fruit is Citrus reticulata Blanco (Rutaceae), Tachibana or Giant orange.
- 医薬品100重量%に対して、10~100重量%の請求項1もしくは2のいずれか記載の乾燥植物組織または10~90重量%の請求項7記載の植物組織抽出物を有効成分とする、アルツハイマー病治療用医薬品。 10. Alzheimer comprising as an active ingredient 10 to 100% by weight of the dry plant tissue according to claim 1 or 2 or 10 to 90% by weight of the plant tissue extract according to claim 7 with respect to 100% by weight of the pharmaceutical product. Drugs for disease treatment.
- 医薬品100重量%に対して、10~100重量%の請求項1もしくは2のいずれか記載の乾燥植物組織または10~90重量%の請求項7記載の植物組織抽出物を有効成分とする、パーキンソン病治療用医薬品。 Parkinson comprising 10 to 100% by weight of the dry plant tissue according to any one of claims 1 or 2 or 10 to 90% by weight of the plant tissue extract according to claim 7 as an active ingredient with respect to 100% by weight of the pharmaceutical product. Drugs for disease treatment.
- 請求項1または2のいずれか記載の乾燥植物組織と医薬的に許容される生薬成分とを1:5~1:10の重量比率で配合して得られる配合物から水抽出により得られる抽出物を有効成分とする、中枢神経変性疾患の治療用医薬品。 3. An extract obtained by water extraction from a blend obtained by blending the dried plant tissue according to claim 1 and a pharmaceutically acceptable herbal medicine component in a weight ratio of 1: 5 to 1:10. A pharmaceutical product for the treatment of central neurodegenerative diseases, comprising
- 細粒剤、茶剤、煎剤、カプセル剤、錠剤、顆粒剤、ゼリー剤、散剤、液剤、シロップ剤またはエキス製剤の形態である、請求項17記載の医薬品。 18. The pharmaceutical product according to claim 17, which is in the form of fine granules, tea, decoction, capsule, tablet, granule, jelly, powder, liquid, syrup or extract.
- 食品100重量%に対して、10~100重量%の請求項1もしくは2のいずれか記載の乾燥植物組織または10~90重量%の請求項7記載の植物組織抽出物を含有する、中枢神経変性疾患の改善のための食品。 Central neurodegeneration containing 10 to 100% by weight of dried plant tissue according to claim 1 or 10% to 90% by weight of plant tissue extract according to claim 7 with respect to 100% by weight of food. Food for improvement of disease.
- 中枢神経変性疾患がアルツハイマー病および/またはパーキンソン病である、請求項19記載の食品。 The food according to claim 19, wherein the central neurodegenerative disease is Alzheimer's disease and / or Parkinson's disease.
- 中枢神経変性疾患の改善が記憶の獲得、保持および想起能力の増強メカニズムによりもたらされる、請求項19記載の食品。 The food according to claim 19, wherein the improvement of the central neurodegenerative disease is brought about by a mechanism for enhancing memory acquisition, retention and recall ability.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012501889A JP5503726B2 (en) | 2010-02-26 | 2011-02-25 | Dried plant tissue and plant tissue extract for improving central nervous degenerative diseases accompanied by learning / memory disorders and movement disorders, and pharmaceuticals and foods containing them |
| CN201180011122.5A CN102883716B (en) | 2010-02-26 | 2011-02-25 | Dried plant tissue and plant tissue extract for ameliorating central nervous system degenerative diseases accompanied by learning/memory disorders, movement disorders and like, and pharmaceutical agent and food or beverage each comprising same |
| HK13105841.5A HK1178792A1 (en) | 2010-02-26 | 2013-05-16 | Dried plant tissue and plant tissue extract for ameliorating central nervous system degenerative diseases accompanied by learning memory disorders, movement disorders and the like, and pharmaceutical agent and food or beverage each comprising the dried plant tissue and the plant tissue extract |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010042819 | 2010-02-26 | ||
| JP2010-042819 | 2010-02-26 | ||
| JP2010-126717 | 2010-06-02 | ||
| JP2010126717 | 2010-06-02 | ||
| JP2010270578 | 2010-12-03 | ||
| JP2010-270578 | 2010-12-03 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011105568A1 true WO2011105568A1 (en) | 2011-09-01 |
Family
ID=44506965
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2011/054358 WO2011105568A1 (en) | 2010-02-26 | 2011-02-25 | Dried plant tissue and plant tissue extract for ameliorating central nervous system degenerative diseases accompanied by learning/memory disorders, movement disorders and the like, and pharmaceutical agent and food or beverage each comprising the dried plant tissue and the plant tissue extract |
Country Status (4)
| Country | Link |
|---|---|
| JP (2) | JP5503726B2 (en) |
| CN (1) | CN102883716B (en) |
| HK (1) | HK1178792A1 (en) |
| WO (1) | WO2011105568A1 (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013191236A1 (en) * | 2012-06-21 | 2013-12-27 | 小太郎漢方製薬株式会社 | Composition for improving and/or treating central nervous system degenerative disease |
| JP2014230509A (en) * | 2013-05-29 | 2014-12-11 | 要 有持 | Production method, processing method, processed product, and tea |
| JP2015053939A (en) * | 2013-09-10 | 2015-03-23 | 久子 松野 | Processed food of skin of orange |
| WO2015199118A1 (en) * | 2014-06-26 | 2015-12-30 | 日本ゼオン株式会社 | Method for promoting phosphorylation of erk or akt in cultured cell, cell culture method, and phosphorylation promoter |
| WO2016075960A1 (en) * | 2014-11-12 | 2016-05-19 | 株式会社沖縄リサーチセンター | Medicinal composition for preventing or improving dysuria, antagonist against dysuria-related receptor, and method for preventing or improving dysuria using medicinal composition or antagonist |
| WO2017208868A1 (en) * | 2016-06-01 | 2017-12-07 | 株式会社三協ホールディングス | Pharmaceutical composition and food |
| WO2019102602A1 (en) * | 2017-11-27 | 2019-05-31 | 康 大泉 | Screening method |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103479743B (en) * | 2013-01-28 | 2015-07-08 | 钱昌美 | Pharmaceutical composition for treating senile dementia and preparation method thereof |
| JP2016056149A (en) * | 2014-09-11 | 2016-04-21 | 日油株式会社 | Skin cosmetic |
| JP6898628B2 (en) * | 2016-06-15 | 2021-07-07 | 国立大学法人広島大学 | Neurodegenerative disease therapeutic agent |
| CN107648349A (en) * | 2017-10-30 | 2018-02-02 | 江西中医药大学 | Bitter orange flower is preparing the application in improving sleep-disorder or improving failure of memory or treat Alzheimer disease drugs or health food |
| KR101894491B1 (en) * | 2018-04-30 | 2018-09-04 | 주식회사 뉴트라팜텍 | A composition for improving memory or cognitive function comprising a aurantii fructus immaturus supercritical extract as an active ingredient |
| CN112402409A (en) * | 2020-12-11 | 2021-02-26 | 江汉大学 | Application of hesperetin in preparation of drugs for inhibiting beta-secretase activity |
| CN115886258A (en) * | 2022-12-30 | 2023-04-04 | 上海交大昂立股份有限公司 | Composite probiotic composition for improving physiological memory attenuation |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002053152A1 (en) * | 2001-01-08 | 2002-07-11 | Imperial College Of Science, Technology & Medicine | Composition containing flavonoids for treatment brain disorders |
| JP2008127325A (en) * | 2006-11-20 | 2008-06-05 | Tokyoto Koreisha Kenkyu Fukushi Shinko Zaidan | Phosphorylated mbp production promoter |
-
2011
- 2011-02-25 CN CN201180011122.5A patent/CN102883716B/en active Active
- 2011-02-25 WO PCT/JP2011/054358 patent/WO2011105568A1/en active Application Filing
- 2011-02-25 JP JP2012501889A patent/JP5503726B2/en active Active
-
2013
- 2013-05-16 HK HK13105841.5A patent/HK1178792A1/en unknown
-
2014
- 2014-03-14 JP JP2014052426A patent/JP5906267B2/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002053152A1 (en) * | 2001-01-08 | 2002-07-11 | Imperial College Of Science, Technology & Medicine | Composition containing flavonoids for treatment brain disorders |
| JP2008127325A (en) * | 2006-11-20 | 2008-06-05 | Tokyoto Koreisha Kenkyu Fukushi Shinko Zaidan | Phosphorylated mbp production promoter |
Non-Patent Citations (9)
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2013191236A1 (en) * | 2012-06-21 | 2016-05-26 | 小太郎漢方製薬株式会社 | Composition for improvement and / or treatment of central neurodegenerative diseases |
| WO2013191236A1 (en) * | 2012-06-21 | 2013-12-27 | 小太郎漢方製薬株式会社 | Composition for improving and/or treating central nervous system degenerative disease |
| JP2014230509A (en) * | 2013-05-29 | 2014-12-11 | 要 有持 | Production method, processing method, processed product, and tea |
| JP2015053939A (en) * | 2013-09-10 | 2015-03-23 | 久子 松野 | Processed food of skin of orange |
| JP5958658B2 (en) * | 2014-06-26 | 2016-08-02 | 日本ゼオン株式会社 | Method for enhancing phosphorylation of ERK or AKT in cultured cells, cell culture method, and phosphorylation enhancer |
| WO2015199118A1 (en) * | 2014-06-26 | 2015-12-30 | 日本ゼオン株式会社 | Method for promoting phosphorylation of erk or akt in cultured cell, cell culture method, and phosphorylation promoter |
| JP2016146819A (en) * | 2014-06-26 | 2016-08-18 | 日本ゼオン株式会社 | Phosphorylation enhancing method of erk or akt within culturing cells, and cell culture method |
| JPWO2015199118A1 (en) * | 2014-06-26 | 2017-04-20 | 日本ゼオン株式会社 | Method for enhancing phosphorylation of ERK or AKT in cultured cells, cell culture method, and phosphorylation enhancer |
| WO2016075960A1 (en) * | 2014-11-12 | 2016-05-19 | 株式会社沖縄リサーチセンター | Medicinal composition for preventing or improving dysuria, antagonist against dysuria-related receptor, and method for preventing or improving dysuria using medicinal composition or antagonist |
| WO2017208868A1 (en) * | 2016-06-01 | 2017-12-07 | 株式会社三協ホールディングス | Pharmaceutical composition and food |
| JP2017214330A (en) * | 2016-06-01 | 2017-12-07 | 株式会社三協ホールディングス | Pharmaceutical composition and food product |
| EP3466436A4 (en) * | 2016-06-01 | 2020-01-01 | Sankyo Holdings Co., Ltd. | Pharmaceutical composition and food |
| WO2019102602A1 (en) * | 2017-11-27 | 2019-05-31 | 康 大泉 | Screening method |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2011105568A1 (en) | 2013-06-20 |
| JP5906267B2 (en) | 2016-04-20 |
| CN102883716A (en) | 2013-01-16 |
| JP5503726B2 (en) | 2014-05-28 |
| JP2014111664A (en) | 2014-06-19 |
| CN102883716B (en) | 2014-05-07 |
| HK1178792A1 (en) | 2013-09-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5906267B2 (en) | Dried plant tissue and plant tissue extract for improving central nervous degenerative diseases accompanied by learning / memory disorders and movement disorders, and pharmaceuticals and foods containing them | |
| Gupta et al. | Neuroprotective potential of ellagic acid: a critical review | |
| JP6033863B2 (en) | Composition for improvement and / or treatment of central neurodegenerative diseases | |
| Mondal et al. | Central-stimulating and analgesic activity of the ethanolic extract of Alternanthera sessilis in mice | |
| CN108261547A (en) | For adjusting the composition of metabolic pathway and method | |
| US20040247715A1 (en) | Water soluble extract from plant of solanum genus and the preparation process thereof, and pharmaceutical composition containing the water soluble extract | |
| US20210128664A1 (en) | Agent for Activating Astrocyte Glucose Metabolism | |
| KR102041847B1 (en) | Composition for preventing, improving or treating depression and anxiety disorders comprising Fraxinus rhynchophylla extract as effective component | |
| CN112294821B (en) | Application of 5-methyltetrahydrofolic acid and composition thereof | |
| CN108420890B (en) | Composition with blood fat reducing effect and preparation method thereof | |
| DK2845624T3 (en) | Devil's mucoadhesive extracts (harpagophytum procumbens) and their applications | |
| Nafiu et al. | Ameliorative effect of Lecaniodiscus cupanioides (Sapindaceae) aqueous root extract in loperamide-induced constipated rats | |
| WO2022262372A1 (en) | Health-care composition for increasing hdl-c | |
| Dimpfel et al. | Zynamite®(Mangifera indica Leaf Extract) and Caffeine Act in a Synergistic Manner on Electrophysiological Parameters of Rat Central Nervous System | |
| EP2995311A1 (en) | Compositions for the treatment of age related disorders | |
| US20180193401A1 (en) | Anti-obesity composition comprising natural complex | |
| KR20110095765A (en) | Anti-allergic composition containing scrophularia buergeriana extract | |
| KR101509056B1 (en) | Composition having brain function and congnition enhancing activity comprising ginseng mixed herbal extracts, ginsenoside Rg2 and ginsenoside F2 | |
| Moektiwardoyo et al. | Immunomodulatory activity of Plectranthus scutellarioides (L.) R. Br. leaves ethanolic extract and its fraction on rat using carbon clearance method. | |
| CN112043789A (en) | Composition for preventing or treating nicotine addiction comprising extract of liriope platyphylla as active ingredient | |
| US8124138B2 (en) | Composition for prevention and/or treatment of pruritus containing acacia bark derivative | |
| JPWO2005082391A1 (en) | Human β3 adrenergic receptor agonist | |
| CN111246849A (en) | Composition for enhancing learning and memory ability | |
| Onyeso et al. | Coadministration of Caffeine and Hydromethanolic Fraction of Citrullus lanatus Seeds Extract improved Heamatological and Lipid Profile of Wistar Albino Rats | |
| JP2018177748A (en) | Agent for activating glucose metabolism of astrocyte |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201180011122.5 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11747524 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012501889 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 11747524 Country of ref document: EP Kind code of ref document: A1 |