WO2010100028A2 - Enzymatic textile bleach-whitening methods - Google Patents
Enzymatic textile bleach-whitening methods Download PDFInfo
- Publication number
- WO2010100028A2 WO2010100028A2 PCT/EP2010/051953 EP2010051953W WO2010100028A2 WO 2010100028 A2 WO2010100028 A2 WO 2010100028A2 EP 2010051953 W EP2010051953 W EP 2010051953W WO 2010100028 A2 WO2010100028 A2 WO 2010100028A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- acid
- textile
- enzymatic
- enzyme
- Prior art date
Links
- 0 *c1cc(-[n](nc2)nc2-c2ccccc2)ccc1C=Cc(cc1)c(*)cc1-[n](nc1)nc1-c1ccccc1 Chemical compound *c1cc(-[n](nc2)nc2-c2ccccc2)ccc1C=Cc(cc1)c(*)cc1-[n](nc1)nc1-c1ccccc1 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
- C11D3/38636—Preparations containing enzymes, e.g. protease or amylase containing enzymes other than protease, amylase, lipase, cellulase, oxidase or reductase
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3947—Liquid compositions
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/40—Dyes ; Pigments
- C11D3/42—Brightening agents ; Blueing agents
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06L—DRY-CLEANING, WASHING OR BLEACHING FIBRES, FILAMENTS, THREADS, YARNS, FABRICS, FEATHERS OR MADE-UP FIBROUS GOODS; BLEACHING LEATHER OR FURS
- D06L4/00—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs
- D06L4/40—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs using enzymes
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06L—DRY-CLEANING, WASHING OR BLEACHING FIBRES, FILAMENTS, THREADS, YARNS, FABRICS, FEATHERS OR MADE-UP FIBROUS GOODS; BLEACHING LEATHER OR FURS
- D06L4/00—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs
- D06L4/60—Optical bleaching or brightening
Definitions
- the present invention relates to methods for the enzymatic bleach-whitening of synthetic textile material, in particular polyamide or polyamide-containing textile materials.
- a pretreatment or preparation step is typically required to properly prepare the textile materials for further use and in particular for the dyeing, printing and/or finishing stages typically required for commercial goods.
- These textile treatment steps remove impurities and color bodies, either naturally existing or those added by the spinning and weaving steps to the fibers and/or fabrics.
- textile treatments may include a number of varying treatments and stages, the most common include: de-sizing, the removal of sizing agents, such as starches, via enzymatic, alkali or oxidative soaking; scouring, the removal of greases, oils, waxes, pectic substances, protein and fats by contact with a solution of sodium hydroxide at temperatures near boiling; and bleaching, the removal and lightening of color bodies from textiles by commonly using oxidizing agents (such as hydrogen peroxide, hypochlorite, and chlorine dioxide), or by using reducing agents (such as, sulfur dioxide or dithionite salts).
- oxidizing agents such as hydrogen peroxide, hypochlorite, and chlorine dioxide
- reducing agents such as, sulfur dioxide or dithionite salts
- the present invention accordingly relates to a method for bleach-whitening of synthetic textile material, in particular polyamide or polyamide-containing textile material, comprising contacting said textile material with
- the subject matter according to the present invention provides methods for bleaching and whitening of synthetic textile material, in particular polyamide or polyamide-containing textile materials, which are contacted with an enzymatic bleaching composition and a fluorescent whitening agent for a length of time and under conditions enabling a full white effect to be achieved.
- the invention is also suitable for the brightening of pastel shades.
- the invention also provides dyed textiles produced from textiles that have been prepared according to the enzymatic bleaching-whitening methods described herein.
- the enzymatic bleach-whitening process according to the present invention is environmentally friendly and provides synthetic textile material which does not exhibit yellowing problems encountered in subsequent drying when the residual sulfur derivatives are not properly and thoroughly removed as compared to the whitening obtained in conventional reduction bleach.
- the enzymatic bleaching step of the present invention will employ, unless otherwise indicated, conventional techniques of molecular biology (including recombinant techniques), microbiology, cell biology, and biochemistry, which are within the skill of the art. Such techniques are explained fully in the literature, for example, Molecular Cloning: A Laboratory Manual, 2 nd ed., (Sambrook et al., 1989); Oligonucleotide Synthesis (MJ. Gait, ed., 1984); Current Protocols in Molecular Biology (F. M. Ausubel et al., eds., 1994); PCR: The Polymerase Chain Reaction (MuIMs et al., eds., 1994); and Gene Transfer and Expression: A Laboratory Manual (Kriegler, 1990).
- nucleic acids are written left to right in 5' to 3' orientation; amino acid sequences are written left to right in amino to carboxy orientation, respectively.
- bleaching means the process of treating a textile material for a sufficient length of time and under appropriate pH and temperature conditions to produce a lighter color in said textile material by removal, modification or masking of color-causing compounds in the textile material.
- bleaching refers to the treatment of a textile material to effect a brightening of the textile material.
- bleaching agent encompasses any moiety that is capable of bleaching a textile. They may require the presence of a bleach activator. Bleaching may generally be performed using chemical bleaching agent(s) and/or enzymatically generated bleaching agent(s). Examples of suitable chemical bleaching agents useful in the processes - A -
- H 2 O 2 may be considered a chemical bleaching agent when it has been generated enzymatically in situ.
- a "chemical bleaching composition” contains one or more chemical bleaching agent(s).
- an "enzymatic bleaching system” or “enzymatic bleaching composition” as understood in the context of the present invention contains one or more perhydrolase enzymes and one or more ester substrates capable of enzymatically generating a bleaching agent, and a hydrogen peroxide source, for production of a peracid bleaching agent.
- enzymatic bleaching systems are described, for example, in WO 2005/056782.
- synthetic textile material comprises polyamides, polyurethanes, polyacrylics, polyesters, polyolefines, polylactide, and semi-synthetic material such as cellulose acetate.
- the cited textile materials can be in a very wide range of forms of presentations, for example, in the form of fibers, yarns, fabrics, garments, knits, wovens and non-wovens.
- polyamide or polyamide-containing textile materials comprises synthetic polyamide, for example, Nylon 6 (polycaproamide), Nylon 6,6 (polyhexamethylene adipamide), Nylon 7 (polyenanthamide), Nylon 6,12 (polyhexamethylene dodecamide), Nylon 11 , Nylon 12 and copolyamides of Nylon 6,6 or Nylon 6, such as polymers made from hexamethylene diamine, ⁇ -caprolactam and adipic acid and polymers prepared from adipic acid, hexamethylene diamine and isophthalic acid, or from adipic acid, hexamehylene diamine and 2-methyl-pentamethylene diamine or 2-ethyl-tetramethylene diamine.
- synthetic polyamide for example, Nylon 6 (polycaproamide), Nylon 6,6 (polyhexamethylene adipamide), Nylon 7 (polyenanthamide), Nylon 6,12 (polyhexamethylene dodecamide), Nylon 11 ,
- blends for example, blends of polyamide/wool, polyamide/polyacrylonitril, polyamide/cellulose, polyamide/polyester, polyamide/cellulose acetate, polyamide/cellulose triacetate, polyamide/cellulose/wool and all blends indicated above blended with elastane fibers.
- a “perhydrolase” refers to an enzyme that is capable of catalyzing a perhydrolysis reaction that results in the production of a sufficiently high amount of peracid suitable for use in an enzymatic textile bleaching composition according to the method described herein.
- a perhydrolase enzyme used in the methods described herein exhibits a high perhydrolysis to hydrolysis ratio.
- the perhydrolase comprises, consists of, or consists essentially of the Mycobacterium smegmatis perhydrolase amino acid sequence set forth in SEQ ID NO:1 , or a variant or homolog thereof.
- the perhydrolase enzyme comprises acyl transferase activity and catalyzes an aqueous acyl transfer reaction.
- esters substrate in reference to an enzymatic textile bleaching composition according to the invention described herein refers to a perhydrolase substrate that contains an ester linkage.
- Esters comprising aliphatic and/or aromatic carboxylic acids and alcohols may be utilized as substrates with perhydrolase enzymes.
- the ester source is selected from the esters of one or more of the following acids: formic acid, acetic acid, propionic acid, butyric acid, valeric acid, caproic acid, caprylic acid, nonanoic acid, decanoic acid, dodecanoic acid, myristic acid, palmitic acid, stearic acid, and oleic acid.
- the ester source is an acetate ester. In some embodiments, the ester source is selected from one or more of propylene glycol diacetate, ethylene glycol diacetate, glycerol triacetate, ethyl acetate, and glycerol tributyrate.
- perhydrolyzation or “perhydrolyze” or “perhydrolysis” as used herein refer to a reaction wherein a peracid is generated from an ester substrate and a hydrogen peroxide source.
- the perhydrolyzation reaction is catalyzed with a perhydrolase, e.g., acyl transferase or aryl esterase, enzyme.
- -OR * is -OH.
- -OR * is replaced by -NH 2 .
- a peracid is produced by perhydrolysis of a carboxylic acid or amide substrate.
- peracid refers to a molecule derived from a carboxylic acid ester which has been reacted with hydrogen peroxide to form a highly reactive product that is able to transfer one of its oxygen atoms. It is this ability to transfer oxygen atoms that enables a peracid, for example, peracetic acid, to function as a bleaching agent.
- perhydrolysis to hydrolysis ratio refers to the ratio of the amount of enzymatically produced peracid to the amount of enzymatically produced acid by a perhydrolase enzyme from an ester substrate under defined conditions and within a defined time.
- the assays provided in WO 05/056782 are used to determine the amounts of peracid and acid produced by the enzyme.
- an effective amount of perhydrolase enzyme refers to the quantity of perhydrolase enzyme necessary to achieve the enzymatic activity required in the processes or methods described herein. Such effective amounts are readily ascertained by one of ordinary skill in the art and are based on many factors, such as the particular enzyme variant used, the pH used, the temperature used and the like, as well as the results desired (e.g., level of bleaching).
- transferase refers to an enzyme that catalyzes the transfer of a functional group from one substrate to another substrate.
- an acyl transferase may transfer an acyl group from an ester substrate to a hydrogen peroxide substrate to form a peracid.
- acyl refers to an organic group with the general formula RCO-, derived from an organic acid by removal of the -OH group.
- acyl group names end with the suffix "-oyl,” e.g., ethanoyl chloride, CH 3 CO-CI, is the acyl chloride formed from ethanoic acid, CH 3 CO-OH.
- acylation refers to a chemical transformation in which one of the substituents of a molecule is substituted by an acyl group, or the process of introduction of an acyl group into a molecule.
- oxidizing chemical refers to a chemical that has the capability of bleaching a textile.
- the oxidizing chemical is present at an amount, pH and temperature suitable for bleaching.
- the term includes, but is not limited to hydrogen peroxide and peracids.
- the terms “purified” and “isolated” refer to the removal of contaminants from a sample and/or to a material (e.g., a protein, nucleic acid, cell, etc.) that is removed from at least one component with which it is naturally associated.
- a material e.g., a protein, nucleic acid, cell, etc.
- these terms may refer to a material which is substantially or essentially free from components which normally accompany it as found in its native state, such as, for example, an intact biological system.
- polynucleotide refers to a polymeric form of nucleotides of any length and any three-dimensional structure and single- or multi-stranded (e.g., single-stranded, double-stranded, triple-helical, etc.), which contain deoxyribonucleotides, ribonucleotides, and/or analogs or modified forms of deoxyribonucleotides or ribonucleotides, including modified nucleotides or bases or their analogs. Because the genetic code is degenerate, more than one codon may be used to encode a particular amino acid, and the polynucleotides applied applied within the context of the present invention encode a particular amino acid sequence.
- any type of modified nucleotide or nucleotide analog may be used, so long as the polynucleotide retains the desired functionality under conditions of use, including modifications that increase nuclease resistance (e.g., deoxy, 2'-0-Me, phosphorothioates, etc.).
- Labels may also be incorporated for purposes of detection or capture, for example, radioactive or nonradioactive labels or anchors, e.g., biotin.
- polynucleotide also includes peptide nucleic acids (PNA). Polynucleotides may be naturally occurring or non-naturally occurring.
- Polynucleotide and “nucleic acid” and “oligonucleotide” are used herein interchangeably.
- Polynucleotides of the invention may contain RNA, DNA, or both, and/or modified forms and/or analogs thereof.
- a sequence of nucleotides may be interrupted by non-nucleotide components.
- One or more phosphodiester linkages may be replaced by alternative linking groups.
- linking groups include, but are not limited to, embodiments wherein phosphate is replaced by P(O)S ("thioate”), P(S)S ("dithioate”), (O)NR 2 ("amidate"), P(O)R, P(O)OR', CO or CH 2 ("formacetal"), in which each R or R' is independently H or substituted or unsubstituted alkyl (Ci-C 2 o) optionally containing an ether (-O-) linkage, aryl, alkenyl, cycloalkyl, cycloalkenyl or araldyl. Not all linkages in a polynucleotide need be identical. Polynucleotides may be linear or circular or comprise a combination of linear and circular portions. Suitable polynucleotides are described in WO 2005/056782.
- polypeptide refers to any composition comprised of amino acids and recognized as a protein by those of skill in the art.
- the conventional one-letter or three-letter code for amino acid residues is used herein.
- polypeptide and protein are used interchangeably herein to refer to polymers of amino acids of any length.
- the polymer may be linear or branched, it may comprise modified amino acids, and it may be interrupted by non-amino acids.
- the terms also encompass an amino acid polymer that has been modified naturally or by intervention; for example, disulfide bond formation, glycosylation, lipidation, acetylation, phosphorylation, or any other manipulation or modification, such as conjugation with a labeling component.
- polypeptides containing one or more analogs of an amino acid including, for example, unnatural amino acids, etc.
- homologous proteins are familiar to those skilled in the art and are described in more detail in WO 2005/056782 on pages 12, 13 and 50 to 52, which are herein incorporated by reference.
- homologous proteins are engineered to produce enzymes with desired activity(ies).
- the degree of homology between sequences may be determined using any suitable method known in the art.
- PILEUP is a useful program to determine sequence homology levels. PILEUP creates a multiple sequence alignment from a group of related sequences using progressive, pairwise alignments. It can also plot a tree showing the clustering relationships used to create the alignment.
- Another example of a useful algorithm is the BLAST algorithm. Useful methods and programs are referred to in WO 2005/056782 on pages 59 and 60, which are herein incorporated by reference.
- “Surfactant” refers to a substance that reduces surface tension of a liquid.
- Emulsifier refers to a substance that promotes the suspension of one liquid in another.
- Sequestering agent refers to a substance capable of reacting with metallic ions by forming a water-soluble complex in which the metal is held in a non-ionizable form.
- Size refers to compounds used in the textile industry to improve weaving performance by increasing the abrasion resistance and strength of the yarn. Size is usually made of, for example, starch or starch-like compounds.
- desize or “desizing,” as used herein, refer to the process of eliminating size, generally starch, from textiles usually prior to applying special finishes, dyes or bleaches.
- Desizing enzyme(s) refer to enzymes that are used to enzymatically remove the size.
- Exemplary enzymes are amylases, cellulases and mannanases.
- scouring means to remove impurities, for example, much of the impurities (e.g., pectins, proteins, wax, etc.) naturally found in textile materials.
- impurities e.g., pectins, proteins, wax, etc.
- scouring can remove, in some embodiments, residual materials introduced by manufacturing processes, such as spinning, coning or slashing lubricants.
- bleaching may be employed to remove impurities from textiles.
- bioscouring enzyme(s) refers to an enzyme(s) capable of removing at least a portion of the impurities found in textile materials.
- pectinase denotes a pectinase enzyme defined according to the art where pectinases are a group of enzymes that cleave glycosidic linkages of pectic substances mainly poly(1 ,4-alpha-D-galacturonide) and its derivatives (see Sakai et al. (1993) Advances in Applied Microbiology 39:213-294).
- a pectinase useful herein is a pectinase enzyme which catalyzes the random cleavage of alpha-1 ,4-glycosidic linkages in pectic acid also called polygalacturonic acid by transelimination, such as the enzyme class polygalacturonate lyase (EC 4.2.2.2) (PGL), also known as poly(1 ,4-alpha-D-galacturonide) lyase, also known as pectate lyase.
- PGL enzyme class polygalacturonate lyase
- PGL poly(1 ,4-alpha-D-galacturonide) lyase
- pectic substances denotes pectate, polygalacturonic acid and pectin which may be esterified to a higher or lower degree.
- cutinase refers to as a plant, bacterial or fungal derived enzyme used in textile processing. Cutinases are lipolytic enzymes capable of hydrolyzing the substrate cutin. Cutinases can breakdown fatty acid esters and other oil-based compositions need to be removed in the processing (e.g., the scouring) of textiles. "Cutinase” means an enzyme that has significant plant cutin hydrolysis activity. Specifically, a cutinase will have hydrolytic activity on the biopolyester polymer cutin found on the leaves of plants. Suitable cutinases may be isolated from many different plant, fungal and bacterial sources.
- ⁇ -amylase refers to an enzyme that cleaves the ⁇ (1-4)glycosidic linkages of amylose to yield maltose molecules (disaccharides of ⁇ -glucose).
- Amylases are digestive enzymes found in saliva and are also produced by many plants. Amylases break down long-chain carbohydrates (such as starch) into smaller units.
- An "oxidative stable" ⁇ -amylase is an ⁇ -amylase that is resistive to degradation by oxidative means, when compared to non-oxidative stable ⁇ -amylase, especially when compared to the non-oxidative stable ⁇ -amylase form which the oxidative stable ⁇ -amylase was derived.
- protea means a protein or polypeptide domain of a protein or polypeptide derived from a microorganism, e.g. a fungus, bacterium, or from a plant or animal, and that has the ability to catalyze cleavage of peptide bonds at one or more of various positions of a protein carbohydrate backbone.
- Catalase refers to an enzyme (i.e., a polypeptide having catalytic activity) that catalyzes the decomposition of hydrogen peroxide to water and oxygen.
- Batch process or “batchwise process” or “discontinuous process” refers to processing of textiles as lots or batches in which the whole of each batch is subjected to a process or one stage of a process at a time.
- Exhaust process refers to a batch process in which pretreatment chemicals and/or an enzymatic pretreatment composition and dye are added simultaneously or sequentially in a single textile treatment bath.
- Liquor ratio refers to the ratio of the weight of liquor (liquid) employed in a textile treatment process to the weight of the textile treated.
- A “an” and “the” include plural references unless the context clearly dictates otherwise.
- the enzymatic textile bleach-whitening methods described herein are particularly carried out as a discontinuous process (one-bath two-steps exhaustion method) but can also be carried out as a semi-continuous process like pad-batch or pad-roll.
- the enzymatic bleaching composition used in accordance with the enzymatic textile bleach-whitening method of the present invention contain a perhydrolase enzyme, an ester substrate for the perhydrolase enzyme suitable for production of a peracid upon catalytic reaction of the perhydrolase enzyme on the substrate in the presence of a hydrogen peroxide source and/or hydrogen peroxide, a surfactant and/or an emulsifier, a peroxide stabilizer, a sequestering agent, and a buffer which maintains a pH of about 6 to about 8 during a textile bleaching process.
- the enzymatic bleaching composition may, optionally, further contain a bioscouring agent or enzyme and/or a desizing agent or enzyme.
- One or more perhydrolase enzymes may be used in the compositions according to the methods for enzymatic textile bleach-whitening as described herein.
- the perhydrolase enzyme is naturally-occurring (i.e., a perhydrolase enzyme encoded by a genome of a cell).
- the perhydrolase enzyme comprises, consists of, or consists essentially of an amino acid sequence that is at least about 80%, 85%, 90%, 95%, 97%, 98%, 99%, or 99.5% identical to the amino acid sequence of a naturally-occurring perhydrolase enzyme.
- the perhydrolase enzyme is a naturally-occurring M. smegmatis perhydrolase enzyme.
- the perhydrolase enzyme comprises, consists of, or consists essentially of the amino acid sequence set forth in SEQ ID NO:1 or a variant or homologue thereof.
- the perhydrolase enzyme comprises, consists of, or consists essentially of an amino acid sequence that is at least about 80%, 85%, 90%, 95%, 97%, 98%, 99%, or 99.5% identical to the amino acid sequence set forth in SEQ ID NO:1.
- M. smegmatis perhydrolase The amino acid sequence of M. smegmatis perhydrolase is shown below: MAKRILCFGDSLTWGWVPVEDGAPTERFAPDVRWTGVLAQQLGADFEVIEEGLSARTTNID DPTDPRLNGASYLPSCLATHLPLDLVIIMLGTNDTKAYFRRTPLDIALGMSVLVTQVLTSAGG VGTTYPAPKVLVVSPPPLAPMPHPWFQLIFEGGEQKTTELARVYSALASFMKVPFFDAGSVI STDGVDGIHFTEANNRDLGVALAEQVRSLL (SEQ ID NO:1 ).
- the perhydrolase enzyme comprises one or more substitutions at one or more amino acid positions equivalent to position(s) in the M. smegmatis perhydrolase amino acid sequence set forth in SEQ ID NO:1.
- the perhydrolase enzyme comprises any one or any combination of substitutions of amino acids selected from M1 , K3, R4, 15, L6, C7, D10, S11 , L12, T13, W14, W16, G15, V17, P18, V19, D21 , G22, A23, P24, T25, E26, R27, F28, A29, P30, D31 , V32, R33, W34, T35, G36, L38, Q40, Q41 , D45, L42, G43, A44, F46, E47, V48, I49, E50, E51 , G52, L53, S54, A55, R56, T57, T58, N59, I60, D61 , D62, P63, T
- the perhydrolase enzyme comprises one or more of the following substitutions at one or more amino acid positions equivalent to position(s) in the M. smegmatis perhydrolase amino acid sequence set forth in SEQ ID NO:1 : L12C, Q, or G; T25S, G, or P; L53H, Q, G, or S; S54V, L A, P, T, or R; A55G or T; R67T, Q, N, G, E, L, or F; K97R; V125S, G, R, A, or P; F154Y; F196G.
- the perhydrolase enzyme is the S54V variant of SEQ ID NO:1.
- the perhydrolase enzyme comprises a combination of amino acid substitutions at amino acid positions equivalent to amino acid positions in the M. smegmatis perhydrolase amino acid sequence set forth in SEQ ID NO:1 : L12I S54V; L12M S54T; L12T S54V; L12Q T25S S54V; L53H S54V; S54P V125R; S54V V125G; S54V F196G; S54V K97R V125G; or A55G R67T K97R V125G.
- the perhydrolase enzyme comprises a perhydrolysis to hydrolysis ratio of at least 1. In some embodiments, the perhydrolase enzyme comprises a perhydrolysis to hydrolysis ratio greater than 1.
- the perhydrolase enzyme is provided in the enzymatic textile bleaching composition used according to the textile bleach-whitening method of the present invention at a concentration of about 0.5 to about 2.5 ppm, about 1.5 to about 2.0 ppm, for example, about 1.7 ppm, based on the total weight of the aqueous composition (bath) applied for treatment of the textile material.
- the enzymatic bleaching compositions used in accordance with the method described herein include an ester which serves as a substrate for the perhydrolase enzyme for production of a peracid in the presence of hydrogen peroxide.
- the ester substrate is an ester of an aliphatic and/or aromatic carboxylic acid.
- the ester substrate is an ester of one or more of the following: formic acid, acetic acid, propionic acid, butyric acid, valeric acid, caproic acid, caprylic acid, nonanoic acid, decanoic acid, dodecanoic acid, myristic acid, palmitic acid, stearic acid, and oleic acid.
- glycerol triacetate, glycerol tributyrate, and other esters serve as acyl donors for peracid formation.
- the ester substrate is selected from propylene glycol diacetate, ethylene glycol diacetate, glycerol triacetate, ethyl acetate, and glycerol tributyrate.
- the ester substrate is propylene glycol diacetate, ethylene glycol diacetate, or ethyl acetate.
- the ester substrate is propylene glycol diacetate.
- the ester substrate for example, propylene glycol diacetate
- the ester substrate is provided at a concentration of about 2000 to about 4000 ppm, about 2500 to about 3500 ppm, about 2800 ppm to about 3200 ppm, or about 3000 ppm, based on the total weight of the aqueous composition (bath) applied for treatment of the textile material.
- the enzymatic bleaching compositions used in accordance with the method described herein include a hydrogen peroxide source.
- Hydrogen peroxide can be either added directly in batch, or generated continuously “in situ” by chemical, electro-chemical, and/or enzymatic means.
- the hydrogen peroxide source is hydrogen peroxide.
- the hydrogen peroxide source is a solid compound that generates hydrogen peroxide spontaneously upon addition to water.
- Such compounds include adducts of hydrogen peroxide with various inorganic or organic compounds, of which the most widely employed is sodium carbonate perhydrate, also referred to as sodium percarbonate.
- lnorganic perhydrate salts are one embodiment of hydrogen peroxide source.
- inorganic perhydrate salts include perborate, percarbonate, perphosphate, persulfate and persilicate salts.
- the inorganic perhydrate salts are normally the alkali metal salts.
- hydrogen peroxide adducts useful in the compositions used in accordance with the method described herein include adducts of hydrogen peroxide with zeolites, or urea hydrogen peroxide.
- the hydrogen peroxide source compounds may be included as the crystalline and/or substantially pure solid without additional protection.
- the preferred executions of such granular compositions utilize a coated form of the material which provides better storage stability for the perhydrate salt in the granular product.
- Suitable coatings comprise inorganic salts such as alkali metal silicate, carbonate or borate salts or mixtures thereof, or organic materials such as waxes, oils, or fatty soaps.
- the hydrogen peroxide source is an enzymatic hydrogen peroxide generation system.
- the enzymatic hydrogen peroxide generation system comprises an oxidase and its substrate.
- Suitable oxidase enzymes include, but are not limited to: glucose oxidase, sorbitol oxidase, hexose oxidase, choline oxidase, alcohol oxidase, glycerol oxidase, cholesterol oxidase, pyranose oxidase, carboxyalcohol oxidase, L-amino acid oxidase, glycine oxidase, pyruvate oxidase, glutamate oxidase, sarcosine oxidase, lysine oxidase, lactate oxidase, vanillyl oxidase, glycolate oxidase, galactose oxidase, uri
- any enzyme that generates H 2 O 2 with a suitable substrate may be used in the present invention.
- lactate oxidases from Lactobacillus species which are known to create H 2 O 2 from lactic acid and oxygen may be used.
- acid e.g., gluconic acid in the above example
- One advantage of the enzymatic generation of acid is that this reduces the pH of a basic solution to the pH range in which a peracid is most effective in bleaching (i.e., at or below the pKa).
- enzymes e.g., alcohol oxidase, ethylene glycol oxidase, glycerol oxidase, amino acid oxidase, etc.
- ester substrates in combination with the perhydrolase enzymes of the present invention to generate peracids.
- the hydrogen peroxide generating oxidase is a carbohydrate oxidase.
- Hydrogen peroxide may also be generated electrochemically, for example using a fuel cell fed oxygen and hydrogen gas.
- the hydrogen peroxide source is hydrogen peroxide provided at a concentration of about 1000 to about 3200 ppm, about 1500 to about 2800 ppm, about 2000 ppm to about 2200 ppm, or about 2100 ppm, based on the total weight of the aqueous composition (bath) applied for treatment of the textile material.
- the enzymatic textile bleaching compositions used in accordance with the present method contain one or more, i.e., at least one surfactant and/or at least one emulsifier.
- Surfactants suitable for use in practicing the present invention include, without limitation, nonionic (see, e.g., U.S. Pat. No. 4,565,647, which is herein incorporated by reference); anionic; cationic; and zwitterionic surfactants (see, e.g., U.S. Pat. No. 3,929,678 which is herein incorporated by reference).
- Anionic surfactants include, without limitation, linear alkylbenzenesulfonate, ⁇ -olefinsulfonate, alkyl sulfate (fatty alcohol sulfate), alcohol ethoxysulfate, secondary alkanesulfonate, alpha-sulfo fatty acid methyl ester, alkyl- or alkenylsuccinic acid, and soap.
- Non-ionic surfactants include, without limitation, fatty alcohol ethoxylate, isotridecanol ethoxylate, nonylphenol ethoxylate, alkylpolyglycoside, alkyldimethylamineoxide, ethoxylated fatty acid monoethanolamide, fatty acid monoethanolamide, polyhydroxy alkyl fatty acid amide, and N-acyl N-alkyl derivatives of glucosamine (“glucamides").
- the surfactant and/or emulsifier comprises a non-ionic surfactant.
- the non-ionic surfactant is a fatty alcohol ethoxylate.
- the non-ionic surfactant is isotridecanol ethoxylate.
- the non-ionic surfactant is a fatty alcohol ethoxylate and isotridecanol ethoxylate.
- the composition used in accordance with the present method comprises a surfactant and an emulsifier.
- a surfactant may be present at a concentration of about 300 ppm to about 4800 ppm, about 600 ppm to about 3600 ppm, or about 300 ppm to about 1200 ppm, based on the total weight of the aqueous composition (bath) applied for treatment of the textile material.
- the enzymatic bleaching composition contains isotridecanol ethoxylate at a concentration of about 300 ppm to about 3600 ppm, about 600 ppm to about 3000 ppm, or about 900 ppm to about 2400 ppm, based on the total weight of the aqueous composition (bath) applied for treatment of the textile material.
- the enzymatic bleaching compositions used in accordance with the method described herein contain a peroxide stabilizer.
- peroxide stabilizers include, but are not limited to, sodium silicate, sodium carbonate, acrylic polymers, magnesium salts, and phosphonic acid.
- the peroxide stabilizer is phosphonic acid.
- a peroxide stabilizer may be present in the enzymatic textile bleaching composition at a concentration of about 60 ppm to about 600 ppm, about 60 ppm to about 1200 ppm, or about 120 ppm to about 960 ppm based on the total weight of the aqueous composition (bath) applied for treatment of the textile material.
- the enzymatic bleaching compositions used in accordance with the method described herein include a sequestering agent.
- sequestering agents include, but are not limited to, amino carboxylates, amino phosphonates, polyfunctionally-substituted aromatic chelating agents, polyhydroxy-carboxylic acids, aminopolycarboxylic acids, polyphosphonates, and polyacrylic acids, and mixtures thereof.
- Amino carboxylates useful as sequestering agents include ethylenediaminetetracetates, N-hydroxyethylethylenediaminetriacetates, nitrilotriacetates, ethylenediamine tetraproprionates, and triethylenetetraaminehexacetates.
- Polyfunctionally-substituted aromatic sequestering agents are also useful in the compositions herein (see U.S. Pat. No. 3,812,044).
- Preferred compounds of this type in acid form are dihydroxydisulfobenzenes such as 1 ,2-dihydroxy-3,5-disulfobenzenediethylenetriamine- pentaacetates, and ethanoldiglycines, alkali metal, ammonium, and substituted ammonium salts therein and mixtures thereof.
- Amino phosphonates are also suitable for use as sequestering agents in the compositions of the invention when at least low levels of total phosphorus are permitted.
- a biodegradable sequestering agent suitable for use herein is ethylenediamine disuccinate ("EDDS"), especially the [S, S] isomer as described in U.S. Pat. No. 4,704,233.
- EDDS ethylenediamine disuccinate
- the sequestering agent is polyacrylic acid.
- a sequestering agent may be present in the enzymatic textile bleaching composition at a concentration of about 60 ppm to about 1800 ppm, about 300 ppm to about 1200 ppm, or about 180 ppm to about 1200 ppm based on the total weight of the aqueous composition (bath) applied for treatment of the textile material.
- At least one surfactant and/or emulsifier, at least one peroxide stabilizer and at least one sequestering agent are applied as a combination product containing each of at least one surfactant and/or emulsifier, at least one peroxide stabilizer and at least one sequestering agent.
- the said combination product is designated bleaching processor and is commercially available, for example, CLARITE ® LTC (product of Huntsman).
- a surfactant may be present at a concentration of about 5% to about 40%, about 20% to about 30%, or about 5% to about 10%, based on the total weight of the bleaching processor.
- a peroxide stabilizer may be present in the bleaching processor at a concentration of about 1% to about 5%, about 1 % to about 10%, or about 2% to about 8%, based on the total weight of the bleaching processor.
- a sequestering agent may be present in the bleaching processor at a concentration of about 1 % to about 15%, about 5% to about 10%, or about 3% to about 10%, based on the total weight of the bleaching processor.
- the bleaching processor contains isotridecanol ethoxylate at a concentration of about 5% to about 30%, about 10% to about 25%, or about 15% to about 20%, based on the total weight of the bleaching processor.
- the bleaching processor is suitably provided as an aqueous composition comprising the above indicated components.
- the enzymatic bleaching composition contains a buffer that is capable of maintaining the pH of the composition at a pH of about 6 to about 8.
- the buffer is a carbonate buffer, pH 8.
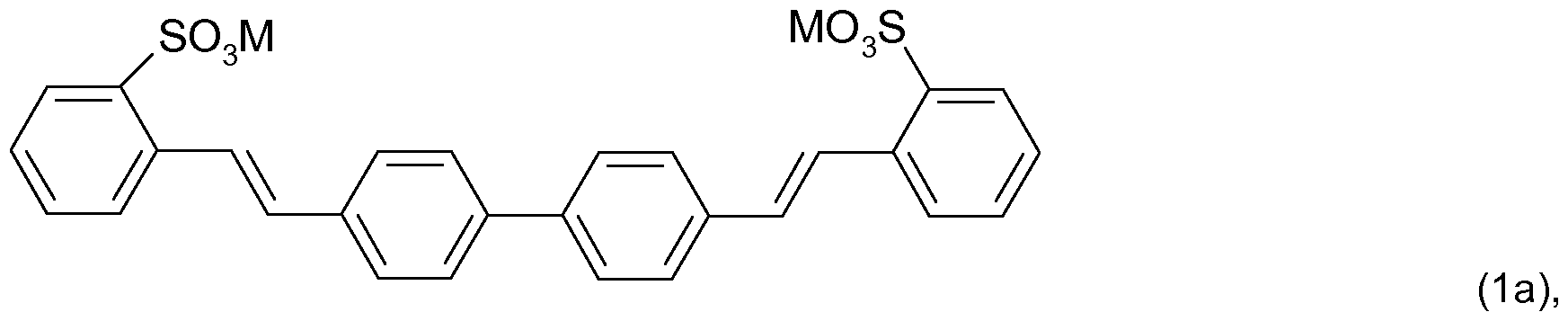
- Suitable fluorescent whitening agents used in accordance with the method described herein are, for example, distyryl biphenyl derivatives as described in U.S. Pat. No. 3,984,399, U.S. Pat. No. 5,969,204 and U.S. Pat. No. 6,096,919 which are herein incorporated by reference.
- distyryl biphenyl derivatives indicated correspond to compounds of the formula in which
- M is hydrogen, lithium, sodium, potassium, ammonium or ammonium that is mono-, di-, tri- or tetra-substituted by C-i-C ⁇ alkyl or C 2 -Cehydroxyalkyl which may be interrupted by an oxygen atom or a mixture thereof, and R 1 and R 2 independently of one another are hydrogen, d-C ⁇ alkyl, d-C ⁇ alkoxy or halogen.
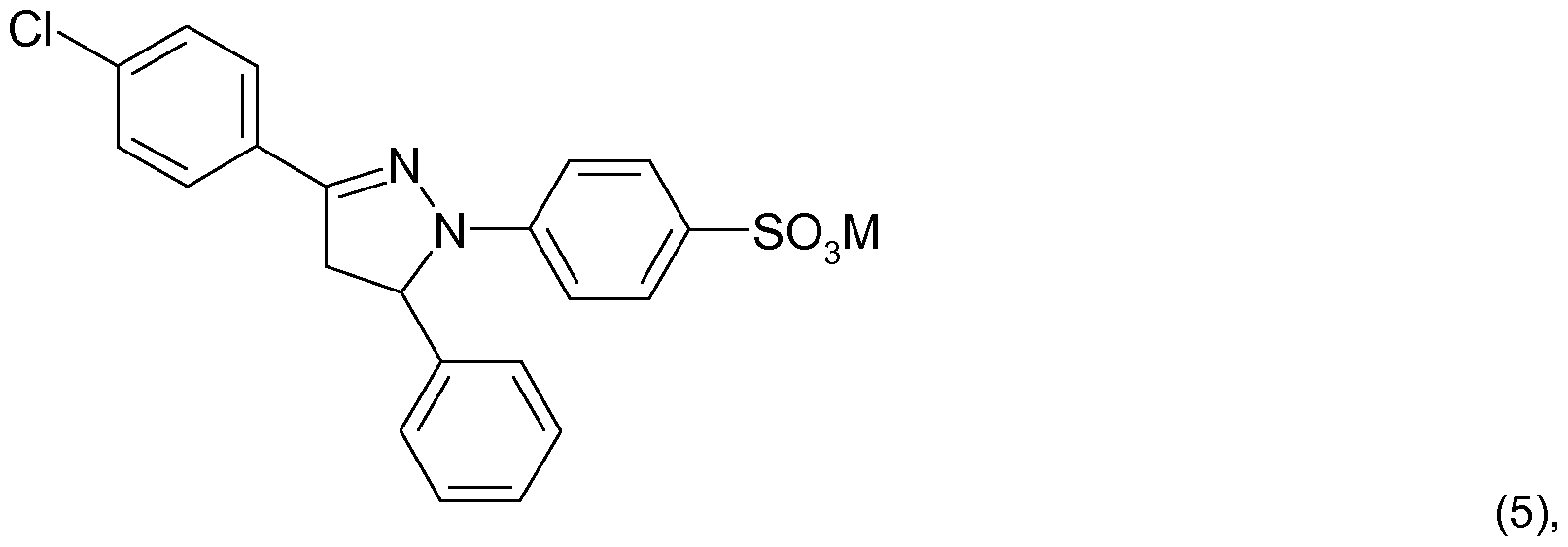
- the compound of the formula (1 ) corresponds to the compound of the formula (1a)
- fluorescent whitening agents are, for example, triazolyl stilbene disulfonic acid derivatives as described in U.S. Pat. No. 3,485,831 , U.S. Pat. No. 3,453,268 and U.S. Pat. No. 3,994,834 which are herein incorporated by reference.
- fluorescent whitening agents which come into consideration for practising the present invention are, for example, diamino stilbene derivatives of the formula
- R 3 and R 4 independently of one another are Ci-C 4 alkoxy, Ci-C 4 alkylthio, substituted or unsubstituted amino or an N-heterocycle which may contain further heteroatoms;
- R 5 and R 6 independently of one another are hydrogen, Ci-C 4 alkyl, Ci-C 4 alkoxy or halogen;
- R 7 and R 8 independently of one another are hydrogen, d-C 4 alkyl, d-C 4 alkoxy or halogen;
- C 2 -C 6 hydroxyalkyl which may be interrupted by an oxygen atom as suitable substituent for M in the meaning of mono-, di-, tri- or tetra-substituted ammonium is suitably typically a radical of formula -CH 2 CH 2 OH, -CH 2 -CH(OH)-CH 3 , -(CH 2 ) 2 -O-(CH 2 ) 2 -OH, -(CH 2 ) 2 -O-CH 2 -CH 2 -OH or -(CH 2 ) 2 -O-CH 2 -CH(CH 3 )-OH.
- CrC ⁇ alkyl as suitable substituent for M in the meaning of mono-, di-, tri- or tetra-substituted ammonium is, for example, methyl, ethyl, propyl, isopropyl, butyl, sec-butyl, tert-butyl, isobutyl, pentyl or hexyl.
- R 1 and R 2 defined as CrC ⁇ alkyl independently of one another are, for example, methyl, ethyl, propyl, isopropyl, butyl, sec-butyl, tert-butyl, isobutyl, pentyl or hexyl and, preferably, Ci-C 4 alkyl, such as ethyl.
- Ri and R 2 defined as Ci-C 6 alkoxy independently of one another are, for example, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, pentyloxy or hexyloxy, preferably Ci-C 4 alkoxy, such as methoxy or ethoxy.
- Ri, R2, Rs, Re, R7 and R 8 defined as halogen independently of one another are, for example, fluorine, chlorine or bromine, preferably chlorine or bromine and in particular chlorine, the definitions and preferences also applying to the following halogen radicals.
- R 3 and R 4 defined as Ci-C 4 alkylthio independently of one another are, for example, methylthio, ethylthio, n-propylthio, isopropylthio or n-butylthio, preferably ethylthio or n-propylthio.
- R 3 , R 4 , R 5 , Re, R7 and R 8 defined as Ci-C 4 alkoxy independently of one another are, for example, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy or isobutoxy, preferably methoxy or ethoxy, the definitions and preferences also applying to the following Ci-C 4 alkoxy radicals.
- R 5 , Re, R7 and R 8 defined as Ci-C 4 alkyl independently of one another are, for example, methyl, ethyl, propyl, isopropyl, butyl, sec-butyl, tert-butyl or isobutyl, the definitions and preferences also applying to the following d-C 4 alkyl radicals.
- R3 and R 4 as substituted or unsubstituted amino independently of one another are, for example, amino which is unsubstituted or substituted on the N-atom by, for example, the following radicals:
- N-mono- or N,N-di-Ci-C 4 alkylamino which includes both the unsubstituted radicals, for example, ethylamino or diethylamino, and the radicals substituted in the alkyl moiety by, for example, N,N-di-Ci-C 4 alkylamino, Ci-C 4 alkoxy, hydroxy, carboxy, sulfo or sulfato; C 5 -C 7 cycloalkylamino, which includes both the unsubstituted radicals and the radicals substituted in the cycloalkyl ring, for example by Ci-C 4 alkyl, in particular methyl; phenylamino or N-Ci-C 4 alkyl-N-phenylamino, which includes both the unsubstituted radicals and the radicals substituted in the phenyl ring, for example by Ci-C 4 alkyl which in turn may be substituted by phenyl, Ci-
- R 3 and R 4 in the meaning of an N-heterocycle which may contain further heteroatoms independently of one another are, for example, morpholino or piperidin-1-yl.
- Carboxy generally means the group -COOM
- Sulfo generally means the group -SO 3 M, in which M is hydrogen, lithium, sodium, potassium, ammonium or ammonium that is mono-, di-, tri- or tetra-substituted by Ci-C 4 alkyl or Ci-C 4 hydroxyalkyl or a mixture thereof.
- At least one of a distyryl biphenyl derivative and a triazolyl stilbene disulfonic acid derivative are used in accordance with the method described herein.
- At least one of a distyryl biphenyl derivative of the formula (1 a) and a triazolyl stilbene disulfonic acid derivative of the formula (2) are used in accordance with the method described herein.
- the fluorescent whitening agent is provided at a concentration of about 0.05 to about 0.8 %, about 0.08 to about 0.5 %, about 0.1 to about 0.4 %, based on the weight of the textile material.
- An organic acid is appropriately used to adjust the pH for the uptake of the fluorescent whitening agent.
- the organic acid is suitably oxalic acid, citric acid and acetic acid.
- the organic acid is acetic acid.
- the organic acid is provided at a concentration of about 100 to about 4000 ppm, about 600 to about 3000 ppm, about 800 ppm to about 1600 ppm, based on the total weight of the aqueous composition (bath) applied for treatment of the textile material.
- the pH is adjusted to pH 4 to 5, for example, pH 4.5.
- the method of the invention utilizes a liquor ratio of about 2:1 to about 50:1 , about 5:1 to about 20:1 , for example, about 20:1 or 10:1.
- the method according to the present invention is performed in a discontinuous process, for example, as a one-bath-two-steps exhaustion method.
- the enzymatic textile bleaching is carried out in the first step and in a subsequent second step whitening is carried out by adding the fluorescent whitening agent into the same bath after adjustment of the bath to pH 4 to 5, for example, pH 4.5, during 20 minutes at about 65°C to about 95°C.
- the method is performed in a semi-continuous process like pad-batch or pad-roll.
- Textiles are contacted with the enzymatic bleaching composition at a temperature of about 55°C to about 75 0 C, about 60 0 C to about 70 0 C, for a processing time of about 20 to about 60 minutes at a pH of about 6 to about 8.
- the bleaching temperature is about 65°C and the processing time is about 50 minutes.
- the temperature of the enzymatic bleaching composition is raised by about 2°C per minute from a starting temperature of about 20 0 C to about 50 0 C, for example, about 20°C to about 40 0 C, until the processing temperature for bleaching is reached.
- the whitening step is carried out in a separate bath after enzymatic textile bleaching, washing-off and neutralization (two-baths-two-steps).
- One or more rinsing steps are performed after treatment of the textile material with the enzymatic bleaching composition, to remove the bleaching composition.
- the textile is rinsed with an aqueous composition (water or a composition containing water).
- the rinsing temperature is about 40 0 C to about 60 0 C, for example, about 50 0 C.
- the aqueous rinsing composition contains a catalase enzyme to catalyze the decomposition of hydrogen peroxide to water and oxygen.
- the textile is rinsed twice with a catalase containing aqueous composition for about 10 minutes for each rinse.
- residual hydrogen peroxide is removed by rinsing twice with an aqueous composition containing catalase at about 50°C.
- a rinsing step as described above is applied in the one-bath-two-steps method after the whitening step is finished.
- the methods for enzymatic textile bleach-whitening described herein include one or more bioscouring enzyme(s).
- One or more bioscouring enzyme(s) may be included in the enzymatic textile bleaching composition, or a textile may be treated with bioscouring enzyme(s) in a subsequent processing step after treatment in the enzymatic textile bleaching composition.
- bioscouring enzymes there come into consideration for practicing the present invention pectinases, hemicellulases, cellulases, enzymes that hydrolyze polyester substrates, for example, cutinases or lipases and other bioscouring enzymes, for example, proteases.
- Suitable pectinases their application as well as assays to determine enzyme activity are described and referred to in more detail in WO 2007/136469 on pages 19 to 21 , which are herein incorporated by reference.
- Suitable cellulases, their application as well as assays to determine enzyme activity are described and referred to in more detail in WO 2007/136469 on pages 22 and 23, which are herein incorporated by reference.
- bioscouring enzymes which come into consideration for practicing the present invention, may be used, for example, proteases, protease variants, and lipases. These enzymes, their application as well as assays to determine enzyme activity are described and referred to in more detail in WO 2007/136469 on pages 23 to 25, which are herein incorporated by reference.
- polyester substrates suitable for use in the present invention may be used, for example, cutinases or lipases. These enzymes, their application as well as assays to determine enzyme activity are described and referred to in more detail in WO 2007/136469 on pages 21 and 22, which are herein incorporated by reference.
- the bioscouring enzyme is a pectinase.
- the methods for enzymatic textile bleach-whitening described herein include one or more desizing enzyme(s).
- One or more desizing enzyme(s) may be included in the enzymatic textile bleaching composition, or a textile may be treated with desizing enzyme(s) in a previous processing step before treatment in the enzymatic textile bleaching composition.
- the desizing enzyme is an amylolytic enzyme. Mannanases and glucoamylases may also be used. In some embodiments, the desizing enzyme is an ⁇ - or ⁇ -amylase and combinations thereof.
- Suitable amylases their application as well as assays to determine enzyme activity are described and referred to in more detail in WO 2007/136469 on pages 18 and 19, which are herein incorporated by reference.
- the following examples are intended to illustrate, but not limit, the invention. Temperatures are in degrees Celsius, parts are parts by weight and the percentage data are percentages by weight, unless noted otherwise. Parts by weight bear the same relation to parts by volume as the kilogram to the litre.
- Enzymatic bleach-whitening carried out as a one-bath-two-steps exhaustion method Nylon 66 fabric, prescoured and heat-set at 175°C, is treated in a bath containing the bleaching processor, soda ash, the ester substrate, hydrogene peroxide and the perhydrolase enzyme in the amounts given in Table 1 using a liquor ratio of 20:1.
- the temperature is raised from ambient temperature to a target temperature of 65°C at a rate of 2°C per minute.
- the bath is then held at 65°C for 30 minutes and afterwards adjusted to pH 4.5 by addition of acetic acid (80%).
- the fluorescent whitening agent in the amount given in Table 1 is added to the bath and treatment is continued at a temperature of 65°C for further 20 minutes.
- After draining the fabric is rinsed twice for 10 minutes each at 50 0 C and then dried at 70 0 C.
- 0.5 g/l of a 25% solution of Catalase T100 available from Genencor
- Nylon 66 fabric prescoured and heat-set at 175°C, is treated in a bath containing the components in the amounts given in Table 2 using a liquor ratio of 20:1. The temperature is raised from ambient temperature to a target temperature of 95°C at a rate of 2°C per minute. The bath is then held at 95°C for 30 minutes. After draining the fabric is rinsed twice for 10 minutes each at 50 0 C and subsequently dried at 70 0 C.
- Table 2 Comparative Examples 1 to 3 - reduction bleach with sodium dithionite
- INVADINE DA or ULTRAVON EL commercial wetting agents available from Huntsman
- Nylon 66 fabric prescoured and heat-set at 175°C, is treated in a bath containing
- the dried fabric obtained according to step (i) is treated using a liquor ratio of 20:1.
- the temperature is raised from ambient temperature to a target temperature of 95°C at a rate of 2°C per minute.
- the bath is then held at 95°C for 30 minutes. After draining the fabric is rinsed twice for 10 minutes each at 50 0 C and subsequently dried at 70 0 C.
- Nylon 6 texturized knitgood, prescoured and heat-set at 175°C, is treated in a bath containing
- T100 (product of Genencor) is included in each rinse.
- Nylon 6 texturized knitgood, prescoured and heat-set at 175°C is treated in a bath containing the components as given in Table 2 for Comparative Example 1 using a liquor ratio of 20:1.
- the temperature is raised from ambient temperature to a target temperature of 95°C at a rate of 2°C per minute.
- the bath is then held at 95°C for 30 minutes. After draining the fabric is rinsed twice for 10 minutes each at 50 0 C and subsequently dried at 70 0 C.
- the degree of whiteness of the fabrics obtained according to the Examples and the Comparative Examples is determined according to the method described by Ganz (Journal of Color and Appearance 1_, No. 5 (1972)). Differences in Whiteness above 5 units Ganz are visually significant. The results are given in Tables 3 to 6.
- the enzymatic bleach at 65°C clearly improves the base white of Nylon 66 fibers, the bleaching effect being more efficient than the bleaching effect achieved by conventional reduction bleach. Superiorfull white effects (with fluorescence) are achieved when the fabric is treated according to the enzymatic bleach-whitening method when compared to a fabric treated in accordance with the conventional bleach-whitening method. One-bath-two-steps bleach-whitening allows to achieve highest full white effects.
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI1013219A BRPI1013219A2 (en) | 2009-03-06 | 2010-02-17 | method for textile enzymatic bleaching-bleaching |
| EP10705848A EP2403990A2 (en) | 2009-03-06 | 2010-02-17 | Enzymatic textile bleach-whitening methods |
| US13/138,455 US20110302724A1 (en) | 2009-03-06 | 2010-02-17 | Enzymatic textile bleach-whitening methods |
| CN2010800096945A CN102333914A (en) | 2009-03-06 | 2010-02-17 | Enzymatic textile bleach-whitening methods |
| MX2011008656A MX2011008656A (en) | 2009-03-06 | 2010-02-17 | Enzymatic textile bleach-whitening methods. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP09154519 | 2009-03-06 | ||
| EP09154519.4 | 2009-03-06 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2010100028A2 true WO2010100028A2 (en) | 2010-09-10 |
| WO2010100028A3 WO2010100028A3 (en) | 2011-01-27 |
Family
ID=42710049
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2010/051953 WO2010100028A2 (en) | 2009-03-06 | 2010-02-17 | Enzymatic textile bleach-whitening methods |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20110302724A1 (en) |
| EP (1) | EP2403990A2 (en) |
| CN (1) | CN102333914A (en) |
| BR (1) | BRPI1013219A2 (en) |
| CO (1) | CO6362055A2 (en) |
| MX (1) | MX2011008656A (en) |
| TW (1) | TW201035407A (en) |
| WO (1) | WO2010100028A2 (en) |
Cited By (224)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013076253A1 (en) | 2011-11-25 | 2013-05-30 | Novozymes A/S | Polypeptides having lysozyme activity and polynucleotides encoding same |
| WO2013098205A2 (en) | 2011-12-29 | 2013-07-04 | Novozymes A/S | Detergent compositions |
| WO2013110766A1 (en) | 2012-01-26 | 2013-08-01 | Novozymes A/S | Use of polypeptides having protease activity in animal feed and detergents |
| WO2013149858A1 (en) | 2012-04-02 | 2013-10-10 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| WO2013167581A1 (en) | 2012-05-07 | 2013-11-14 | Novozymes A/S | Polypeptides having xanthan degrading activity and polynucleotides encoding same |
| WO2013171241A1 (en) | 2012-05-16 | 2013-11-21 | Novozymes A/S | Compositions comprising lipase and methods of use thereof |
| WO2013189972A2 (en) | 2012-06-20 | 2013-12-27 | Novozymes A/S | Use of polypeptides having protease activity in animal feed and detergents |
| WO2014087011A1 (en) | 2012-12-07 | 2014-06-12 | Novozymes A/S | Preventing adhesion of bacteria |
| WO2014096259A1 (en) | 2012-12-21 | 2014-06-26 | Novozymes A/S | Polypeptides having protease activiy and polynucleotides encoding same |
| WO2014147127A1 (en) | 2013-03-21 | 2014-09-25 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2014184164A1 (en) | 2013-05-14 | 2014-11-20 | Novozymes A/S | Detergent compositions |
| WO2014183921A1 (en) | 2013-05-17 | 2014-11-20 | Novozymes A/S | Polypeptides having alpha amylase activity |
| WO2014207227A1 (en) | 2013-06-27 | 2014-12-31 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2014207224A1 (en) | 2013-06-27 | 2014-12-31 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2015001017A2 (en) | 2013-07-04 | 2015-01-08 | Novozymes A/S | Polypeptides having anti-redeposition effect and polynucleotides encoding same |
| WO2015004102A1 (en) | 2013-07-09 | 2015-01-15 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2015049370A1 (en) | 2013-10-03 | 2015-04-09 | Novozymes A/S | Detergent composition and use of detergent composition |
| WO2015109972A1 (en) | 2014-01-22 | 2015-07-30 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2015112341A1 (en) * | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Fabric treatment composition |
| WO2015112340A1 (en) * | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Method of treating textile fabrics |
| WO2015134729A1 (en) | 2014-03-05 | 2015-09-11 | Novozymes A/S | Compositions and methods for improving properties of non-cellulosic textile materials with xyloglucan endotransglycosylase |
| WO2015134737A1 (en) | 2014-03-05 | 2015-09-11 | Novozymes A/S | Compositions and methods for improving properties of cellulosic textile materials with xyloglucan endotransglycosylase |
| WO2015150457A1 (en) | 2014-04-01 | 2015-10-08 | Novozymes A/S | Polypeptides having alpha amylase activity |
| WO2015158237A1 (en) | 2014-04-15 | 2015-10-22 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2015181119A2 (en) | 2014-05-27 | 2015-12-03 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| WO2015189371A1 (en) | 2014-06-12 | 2015-12-17 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2016079305A1 (en) | 2014-11-20 | 2016-05-26 | Novozymes A/S | Alicyclobacillus variants and polynucleotides encoding same |
| WO2016079110A2 (en) | 2014-11-19 | 2016-05-26 | Novozymes A/S | Use of enzyme for cleaning |
| WO2016087401A1 (en) | 2014-12-05 | 2016-06-09 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| WO2016096996A1 (en) | 2014-12-16 | 2016-06-23 | Novozymes A/S | Polypeptides having n-acetyl glucosamine oxidase activity |
| WO2016135351A1 (en) | 2015-06-30 | 2016-09-01 | Novozymes A/S | Laundry detergent composition, method for washing and use of composition |
| WO2016164596A2 (en) | 2015-04-07 | 2016-10-13 | Novozymes A/S | Methods for selecting enzymes having lipase activity |
| WO2016162558A1 (en) | 2015-04-10 | 2016-10-13 | Novozymes A/S | Detergent composition |
| WO2016162556A1 (en) | 2015-04-10 | 2016-10-13 | Novozymes A/S | Laundry method, use of dnase and detergent composition |
| WO2016184944A1 (en) | 2015-05-19 | 2016-11-24 | Novozymes A/S | Odor reduction |
| EP3106508A1 (en) | 2015-06-18 | 2016-12-21 | Henkel AG & Co. KGaA | Detergent composition comprising subtilase variants |
| WO2016202739A1 (en) | 2015-06-16 | 2016-12-22 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2016202785A1 (en) | 2015-06-17 | 2016-12-22 | Novozymes A/S | Container |
| WO2017046232A1 (en) | 2015-09-17 | 2017-03-23 | Henkel Ag & Co. Kgaa | Detergent compositions comprising polypeptides having xanthan degrading activity |
| WO2017046260A1 (en) | 2015-09-17 | 2017-03-23 | Novozymes A/S | Polypeptides having xanthan degrading activity and polynucleotides encoding same |
| WO2017060505A1 (en) | 2015-10-07 | 2017-04-13 | Novozymes A/S | Polypeptides |
| WO2017064253A1 (en) | 2015-10-14 | 2017-04-20 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2017064269A1 (en) | 2015-10-14 | 2017-04-20 | Novozymes A/S | Polypeptide variants |
| WO2017089366A1 (en) | 2015-11-24 | 2017-06-01 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2017093318A1 (en) | 2015-12-01 | 2017-06-08 | Novozymes A/S | Methods for producing lipases |
| WO2017174769A2 (en) | 2016-04-08 | 2017-10-12 | Novozymes A/S | Detergent compositions and uses of the same |
| WO2017186943A1 (en) | 2016-04-29 | 2017-11-02 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2017207762A1 (en) | 2016-06-03 | 2017-12-07 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2017220422A1 (en) | 2016-06-23 | 2017-12-28 | Novozymes A/S | Use of enzymes, composition and method for removing soil |
| WO2018001959A1 (en) | 2016-06-30 | 2018-01-04 | Novozymes A/S | Lipase variants and compositions comprising surfactant and lipase variant |
| WO2018002261A1 (en) | 2016-07-01 | 2018-01-04 | Novozymes A/S | Detergent compositions |
| WO2018007435A1 (en) | 2016-07-05 | 2018-01-11 | Novozymes A/S | Pectate lyase variants and polynucleotides encoding same |
| WO2018007573A1 (en) | 2016-07-08 | 2018-01-11 | Novozymes A/S | Detergent compositions with galactanase |
| WO2018011277A1 (en) | 2016-07-13 | 2018-01-18 | Novozymes A/S | Bacillus cibi dnase variants |
| WO2018015295A1 (en) | 2016-07-18 | 2018-01-25 | Novozymes A/S | Lipase variants, polynucleotides encoding same and the use thereof |
| WO2018037062A1 (en) | 2016-08-24 | 2018-03-01 | Novozymes A/S | Gh9 endoglucanase variants and polynucleotides encoding same |
| WO2018037064A1 (en) | 2016-08-24 | 2018-03-01 | Henkel Ag & Co. Kgaa | Detergent compositions comprising xanthan lyase variants i |
| WO2018037065A1 (en) | 2016-08-24 | 2018-03-01 | Henkel Ag & Co. Kgaa | Detergent composition comprising gh9 endoglucanase variants i |
| WO2018037061A1 (en) | 2016-08-24 | 2018-03-01 | Novozymes A/S | Xanthan lyase variants and polynucleotides encoding same |
| WO2018060216A1 (en) | 2016-09-29 | 2018-04-05 | Novozymes A/S | Use of enzyme for washing, method for washing and warewashing composition |
| EP3309249A1 (en) | 2013-07-29 | 2018-04-18 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| WO2018077938A1 (en) | 2016-10-25 | 2018-05-03 | Novozymes A/S | Detergent compositions |
| WO2018083093A1 (en) | 2016-11-01 | 2018-05-11 | Novozymes A/S | Multi-core granules |
| EP3321360A2 (en) | 2013-01-03 | 2018-05-16 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2018099762A1 (en) | 2016-12-01 | 2018-06-07 | Basf Se | Stabilization of enzymes in compositions |
| WO2018108865A1 (en) | 2016-12-12 | 2018-06-21 | Novozymes A/S | Use of polypeptides |
| WO2018177936A1 (en) | 2017-03-31 | 2018-10-04 | Novozymes A/S | Polypeptides having dnase activity |
| WO2018177938A1 (en) | 2017-03-31 | 2018-10-04 | Novozymes A/S | Polypeptides having dnase activity |
| WO2018178061A1 (en) | 2017-03-31 | 2018-10-04 | Novozymes A/S | Polypeptides having rnase activity |
| EP3385362A1 (en) | 2017-04-05 | 2018-10-10 | Henkel AG & Co. KGaA | Detergent compositions comprising fungal mannanases |
| EP3385361A1 (en) | 2017-04-05 | 2018-10-10 | Henkel AG & Co. KGaA | Detergent compositions comprising bacterial mannanases |
| WO2018184817A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018185285A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018185280A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018185152A1 (en) | 2017-04-04 | 2018-10-11 | Novozymes A/S | Polypeptide compositions and uses thereof |
| WO2018185181A1 (en) | 2017-04-04 | 2018-10-11 | Novozymes A/S | Glycosyl hydrolases |
| WO2018184873A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2018185267A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018185150A1 (en) | 2017-04-04 | 2018-10-11 | Novozymes A/S | Polypeptides |
| WO2018185269A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018184818A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018184816A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018202846A1 (en) | 2017-05-05 | 2018-11-08 | Novozymes A/S | Compositions comprising lipase and sulfite |
| EP3401385A1 (en) | 2017-05-08 | 2018-11-14 | Henkel AG & Co. KGaA | Detergent composition comprising polypeptide comprising carbohydrate-binding domain |
| WO2018206535A1 (en) | 2017-05-08 | 2018-11-15 | Novozymes A/S | Carbohydrate-binding domain and polynucleotides encoding the same |
| WO2019038057A1 (en) | 2017-08-24 | 2019-02-28 | Novozymes A/S | Xanthan lyase variants and polynucleotides encoding same |
| WO2019038059A1 (en) | 2017-08-24 | 2019-02-28 | Henkel Ag & Co. Kgaa | Detergent compositions comprising gh9 endoglucanase variants ii |
| WO2019038058A1 (en) | 2017-08-24 | 2019-02-28 | Novozymes A/S | Gh9 endoglucanase variants and polynucleotides encoding same |
| WO2019038060A1 (en) | 2017-08-24 | 2019-02-28 | Henkel Ag & Co. Kgaa | Detergent composition comprising xanthan lyase variants ii |
| EP3453757A1 (en) | 2013-12-20 | 2019-03-13 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2019057758A1 (en) | 2017-09-20 | 2019-03-28 | Novozymes A/S | Use of enzymes for improving water absorption and/or whiteness |
| WO2019057902A1 (en) | 2017-09-22 | 2019-03-28 | Novozymes A/S | Novel polypeptides |
| WO2019067390A1 (en) | 2017-09-27 | 2019-04-04 | The Procter & Gamble Company | Detergent compositions comprising lipases |
| WO2019063499A1 (en) | 2017-09-27 | 2019-04-04 | Novozymes A/S | Lipase variants and microcapsule compositions comprising such lipase variants |
| WO2019076800A1 (en) | 2017-10-16 | 2019-04-25 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2019076833A1 (en) | 2017-10-16 | 2019-04-25 | Novozymes A/S | Low dusting granules |
| WO2019076834A1 (en) | 2017-10-16 | 2019-04-25 | Novozymes A/S | Low dusting granules |
| DE102017125559A1 (en) | 2017-11-01 | 2019-05-02 | Henkel Ag & Co. Kgaa | CLEANSING COMPOSITIONS CONTAINING DISPERSINE II |
| WO2019084349A1 (en) | 2017-10-27 | 2019-05-02 | The Procter & Gamble Company | Detergent compositions comprising polypeptide variants |
| WO2019081724A1 (en) | 2017-10-27 | 2019-05-02 | Novozymes A/S | Dnase variants |
| DE102017125560A1 (en) | 2017-11-01 | 2019-05-02 | Henkel Ag & Co. Kgaa | CLEANSING COMPOSITIONS CONTAINING DISPERSINE III |
| DE102017125558A1 (en) | 2017-11-01 | 2019-05-02 | Henkel Ag & Co. Kgaa | CLEANING COMPOSITIONS CONTAINING DISPERSINE I |
| WO2019086528A1 (en) | 2017-11-01 | 2019-05-09 | Novozymes A/S | Polypeptides and compositions comprising such polypeptides |
| WO2019086530A1 (en) | 2017-11-01 | 2019-05-09 | Novozymes A/S | Polypeptides and compositions comprising such polypeptides |
| WO2019086532A1 (en) | 2017-11-01 | 2019-05-09 | Novozymes A/S | Methods for cleaning medical devices |
| WO2019105781A1 (en) | 2017-11-29 | 2019-06-06 | Basf Se | Storage-stable enzyme preparations, their production and use |
| WO2019110462A1 (en) | 2017-12-04 | 2019-06-13 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| WO2019121057A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Laundry formulation for removing fatty compounds having a melting temperature>30°c deposited on textiles |
| EP3521434A1 (en) | 2014-03-12 | 2019-08-07 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2019162000A1 (en) | 2018-02-23 | 2019-08-29 | Henkel Ag & Co. Kgaa | Detergent composition comprising xanthan lyase and endoglucanase variants |
| WO2019180111A1 (en) | 2018-03-23 | 2019-09-26 | Novozymes A/S | Subtilase variants and compositions comprising same |
| EP3553172A1 (en) | 2012-08-16 | 2019-10-16 | Novozymes A/S | Method for treating textile with endoglucanase |
| WO2019201636A1 (en) | 2018-04-19 | 2019-10-24 | Basf Se | Compositions and polymers useful for such compositions |
| WO2019201785A1 (en) | 2018-04-19 | 2019-10-24 | Novozymes A/S | Stabilized cellulase variants |
| WO2019201783A1 (en) | 2018-04-19 | 2019-10-24 | Novozymes A/S | Stabilized cellulase variants |
| WO2019201793A1 (en) | 2018-04-17 | 2019-10-24 | Novozymes A/S | Polypeptides comprising carbohydrate binding activity in detergent compositions and their use in reducing wrinkles in textile or fabric. |
| WO2019238761A1 (en) | 2018-06-15 | 2019-12-19 | Basf Se | Water soluble multilayer films containing wash active chemicals and enzymes |
| WO2020002604A1 (en) | 2018-06-28 | 2020-01-02 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2020002608A1 (en) | 2018-06-29 | 2020-01-02 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2020002255A1 (en) | 2018-06-29 | 2020-01-02 | Novozymes A/S | Subtilase variants and compositions comprising same |
| WO2020008024A1 (en) | 2018-07-06 | 2020-01-09 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020007863A1 (en) | 2018-07-02 | 2020-01-09 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020008043A1 (en) | 2018-07-06 | 2020-01-09 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020007875A1 (en) | 2018-07-03 | 2020-01-09 | Novozymes A/S | Cleaning compositions and uses thereof |
| EP3608403A2 (en) | 2014-12-15 | 2020-02-12 | Henkel AG & Co. KGaA | Detergent composition comprising subtilase variants |
| WO2020030623A1 (en) | 2018-08-10 | 2020-02-13 | Basf Se | Packaging unit comprising a detergent composition containing an enzyme and at least one chelating agent |
| EP3611260A1 (en) | 2013-07-29 | 2020-02-19 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| WO2020070199A1 (en) | 2018-10-03 | 2020-04-09 | Novozymes A/S | Polypeptides having alpha-mannan degrading activity and polynucleotides encoding same |
| WO2020069914A1 (en) | 2018-10-05 | 2020-04-09 | Basf Se | Compounds stabilizing amylases in liquids |
| WO2020070014A1 (en) | 2018-10-02 | 2020-04-09 | Novozymes A/S | Cleaning composition comprising anionic surfactant and a polypeptide having rnase activity |
| WO2020070011A1 (en) | 2018-10-02 | 2020-04-09 | Novozymes A/S | Cleaning composition |
| WO2020069915A1 (en) | 2018-10-05 | 2020-04-09 | Basf Se | Compounds stabilizing hydrolases in liquids |
| WO2020070063A2 (en) | 2018-10-01 | 2020-04-09 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2020069913A1 (en) | 2018-10-05 | 2020-04-09 | Basf Se | Compounds stabilizing hydrolases in liquids |
| WO2020070249A1 (en) | 2018-10-03 | 2020-04-09 | Novozymes A/S | Cleaning compositions |
| WO2020070209A1 (en) | 2018-10-02 | 2020-04-09 | Novozymes A/S | Cleaning composition |
| WO2020074499A1 (en) | 2018-10-09 | 2020-04-16 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020074498A1 (en) | 2018-10-09 | 2020-04-16 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020074545A1 (en) | 2018-10-11 | 2020-04-16 | Novozymes A/S | Cleaning compositions and uses thereof |
| EP3647397A1 (en) | 2018-10-31 | 2020-05-06 | Henkel AG & Co. KGaA | Cleaning compositions containing dispersins iv |
| EP3647398A1 (en) | 2018-10-31 | 2020-05-06 | Henkel AG & Co. KGaA | Cleaning compositions containing dispersins v |
| WO2020104231A1 (en) | 2018-11-19 | 2020-05-28 | Basf Se | Powders and granules containing a chelating agent and an enzyme |
| WO2020114965A1 (en) | 2018-12-03 | 2020-06-11 | Novozymes A/S | LOW pH POWDER DETERGENT COMPOSITION |
| WO2020114968A1 (en) | 2018-12-03 | 2020-06-11 | Novozymes A/S | Powder detergent compositions |
| WO2020127775A1 (en) | 2018-12-21 | 2020-06-25 | Novozymes A/S | Detergent pouch comprising metalloproteases |
| WO2020127796A2 (en) | 2018-12-21 | 2020-06-25 | Novozymes A/S | Polypeptides having peptidoglycan degrading activity and polynucleotides encoding same |
| EP3677676A1 (en) | 2019-01-03 | 2020-07-08 | Basf Se | Compounds stabilizing amylases in liquids |
| EP3690037A1 (en) | 2014-12-04 | 2020-08-05 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3702452A1 (en) | 2019-03-01 | 2020-09-02 | Novozymes A/S | Detergent compositions comprising two proteases |
| WO2020182521A1 (en) | 2019-03-08 | 2020-09-17 | Basf Se | Cationic surfactant and its use in laundry detergent compositions |
| WO2020188095A1 (en) | 2019-03-21 | 2020-09-24 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| EP3715442A1 (en) | 2016-03-23 | 2020-09-30 | Novozymes A/S | Use of polypeptide having dnase activity for treating fabrics |
| WO2020201403A1 (en) | 2019-04-03 | 2020-10-08 | Novozymes A/S | Polypeptides having beta-glucanase activity, polynucleotides encoding same and uses thereof in cleaning and detergent compositions |
| EP3722406A1 (en) | 2014-04-11 | 2020-10-14 | Novozymes A/S | Detergent composition |
| WO2020207944A1 (en) | 2019-04-10 | 2020-10-15 | Novozymes A/S | Polypeptide variants |
| WO2020208056A1 (en) | 2019-04-12 | 2020-10-15 | Novozymes A/S | Stabilized glycoside hydrolase variants |
| EP3739029A1 (en) | 2014-07-04 | 2020-11-18 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2020229480A1 (en) | 2019-05-14 | 2020-11-19 | Basf Se | Compounds stabilizing hydrolases in liquids |
| EP3741848A2 (en) | 2014-12-19 | 2020-11-25 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| EP3741849A2 (en) | 2014-12-19 | 2020-11-25 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| WO2021009067A1 (en) | 2019-07-12 | 2021-01-21 | Novozymes A/S | Enzymatic emulsions for detergents |
| EP3786269A1 (en) | 2013-06-06 | 2021-03-03 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2021037878A1 (en) | 2019-08-27 | 2021-03-04 | Novozymes A/S | Composition comprising a lipase |
| WO2021037895A1 (en) | 2019-08-27 | 2021-03-04 | Novozymes A/S | Detergent composition |
| WO2021053127A1 (en) | 2019-09-19 | 2021-03-25 | Novozymes A/S | Detergent composition |
| WO2021064068A1 (en) | 2019-10-03 | 2021-04-08 | Novozymes A/S | Polypeptides comprising at least two carbohydrate binding domains |
| WO2021074430A1 (en) | 2019-10-18 | 2021-04-22 | Basf Se | Storage-stable hydrolase containing liquids |
| WO2021105336A1 (en) | 2019-11-29 | 2021-06-03 | Basf Se | Compositions comprising polymer and enzyme |
| WO2021115912A1 (en) | 2019-12-09 | 2021-06-17 | Basf Se | Formulations comprising a hydrophobically modified polyethyleneimine and one or more enzymes |
| WO2021122118A1 (en) | 2019-12-20 | 2021-06-24 | Henkel Ag & Co. Kgaa | Cleaning compositions comprising dispersins vi |
| WO2021122120A2 (en) | 2019-12-20 | 2021-06-24 | Henkel Ag & Co. Kgaa | Cleaning compositions comprising dispersins viii |
| WO2021122121A1 (en) | 2019-12-20 | 2021-06-24 | Henkel Ag & Co. Kgaa | Cleaning compositions comprising dispersins ix |
| WO2021121394A1 (en) | 2019-12-20 | 2021-06-24 | Novozymes A/S | Stabilized liquid boron-free enzyme compositions |
| WO2021122117A1 (en) | 2019-12-20 | 2021-06-24 | Henkel Ag & Co. Kgaa | Cleaning composition coprising a dispersin and a carbohydrase |
| WO2021123307A2 (en) | 2019-12-20 | 2021-06-24 | Novozymes A/S | Polypeptides having proteolytic activity and use thereof |
| WO2021130167A1 (en) | 2019-12-23 | 2021-07-01 | Novozymes A/S | Enzyme compositions and uses thereof |
| WO2021148364A1 (en) | 2020-01-23 | 2021-07-29 | Novozymes A/S | Enzyme compositions and uses thereof |
| WO2021152123A1 (en) | 2020-01-31 | 2021-08-05 | Novozymes A/S | Mannanase variants and polynucleotides encoding same |
| WO2021152120A1 (en) | 2020-01-31 | 2021-08-05 | Novozymes A/S | Mannanase variants and polynucleotides encoding same |
| EP3872175A1 (en) | 2015-06-18 | 2021-09-01 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3878957A1 (en) | 2014-05-27 | 2021-09-15 | Novozymes A/S | Methods for producing lipases |
| EP3878960A1 (en) | 2014-07-04 | 2021-09-15 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3892708A1 (en) | 2020-04-06 | 2021-10-13 | Henkel AG & Co. KGaA | Cleaning compositions comprising dispersin variants |
| WO2021204838A1 (en) | 2020-04-08 | 2021-10-14 | Novozymes A/S | Carbohydrate binding module variants |
| WO2021214059A1 (en) | 2020-04-21 | 2021-10-28 | Novozymes A/S | Cleaning compositions comprising polypeptides having fructan degrading activity |
| WO2021239818A1 (en) | 2020-05-26 | 2021-12-02 | Novozymes A/S | Subtilase variants and compositions comprising same |
| EP3929285A2 (en) | 2015-07-01 | 2021-12-29 | Novozymes A/S | Methods of reducing odor |
| WO2021259099A1 (en) | 2020-06-24 | 2021-12-30 | Novozymes A/S | Use of cellulases for removing dust mite from textile |
| EP3936593A1 (en) | 2020-07-08 | 2022-01-12 | Henkel AG & Co. KGaA | Cleaning compositions and uses thereof |
| WO2022008732A1 (en) | 2020-07-10 | 2022-01-13 | Basf Se | Enhancing the activity of antimicrobial preservatives |
| EP3950939A2 (en) | 2015-07-06 | 2022-02-09 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| EP3957711A2 (en) | 2015-10-28 | 2022-02-23 | Novozymes A/S | Detergent composition comprising amylase and protease variants |
| WO2022043321A2 (en) | 2020-08-25 | 2022-03-03 | Novozymes A/S | Variants of a family 44 xyloglucanase |
| WO2022043563A1 (en) | 2020-08-28 | 2022-03-03 | Novozymes A/S | Polyester degrading protease variants |
| WO2022063699A1 (en) | 2020-09-22 | 2022-03-31 | Basf Se | Improved combination of protease and protease inhibitor with secondary enzyme |
| WO2022074037A2 (en) | 2020-10-07 | 2022-04-14 | Novozymes A/S | Alpha-amylase variants |
| WO2022084303A2 (en) | 2020-10-20 | 2022-04-28 | Novozymes A/S | Use of polypeptides having dnase activity |
| WO2022090361A2 (en) | 2020-10-29 | 2022-05-05 | Novozymes A/S | Lipase variants and compositions comprising such lipase variants |
| WO2022090320A1 (en) | 2020-10-28 | 2022-05-05 | Novozymes A/S | Use of lipoxygenase |
| WO2022103725A1 (en) | 2020-11-13 | 2022-05-19 | Novozymes A/S | Detergent composition comprising a lipase |
| WO2022106404A1 (en) | 2020-11-18 | 2022-05-27 | Novozymes A/S | Combination of proteases |
| WO2022106400A1 (en) | 2020-11-18 | 2022-05-27 | Novozymes A/S | Combination of immunochemically different proteases |
| EP4032966A1 (en) | 2021-01-22 | 2022-07-27 | Novozymes A/S | Liquid enzyme composition with sulfite scavenger |
| WO2022162043A1 (en) | 2021-01-28 | 2022-08-04 | Novozymes A/S | Lipase with low malodor generation |
| EP4039806A1 (en) | 2021-02-04 | 2022-08-10 | Henkel AG & Co. KGaA | Detergent composition comprising xanthan lyase and endoglucanase variants with im-proved stability |
| WO2022171872A1 (en) | 2021-02-12 | 2022-08-18 | Novozymes A/S | Stabilized biological detergents |
| WO2022171780A2 (en) | 2021-02-12 | 2022-08-18 | Novozymes A/S | Alpha-amylase variants |
| WO2022189521A1 (en) | 2021-03-12 | 2022-09-15 | Novozymes A/S | Polypeptide variants |
| EP4060036A1 (en) | 2021-03-15 | 2022-09-21 | Novozymes A/S | Polypeptide variants |
| WO2022194673A1 (en) | 2021-03-15 | 2022-09-22 | Novozymes A/S | Dnase variants |
| WO2022199418A1 (en) | 2021-03-26 | 2022-09-29 | Novozymes A/S | Detergent composition with reduced polymer content |
| WO2022268885A1 (en) | 2021-06-23 | 2022-12-29 | Novozymes A/S | Alpha-amylase polypeptides |
| WO2023116569A1 (en) | 2021-12-21 | 2023-06-29 | Novozymes A/S | Composition comprising a lipase and a booster |
| EP4206309A1 (en) | 2021-12-30 | 2023-07-05 | Novozymes A/S | Protein particles with improved whiteness |
| EP4234664A1 (en) | 2022-02-24 | 2023-08-30 | Evonik Operations GmbH | Composition comprising glucolipids and enzymes |
| WO2023165950A1 (en) | 2022-03-04 | 2023-09-07 | Novozymes A/S | Dnase variants and compositions |
| WO2023165507A1 (en) | 2022-03-02 | 2023-09-07 | Novozymes A/S | Use of xyloglucanase for improvement of sustainability of detergents |
| WO2023194204A1 (en) | 2022-04-08 | 2023-10-12 | Novozymes A/S | Hexosaminidase variants and compositions |
| WO2023232192A1 (en) | 2022-06-01 | 2023-12-07 | Henkel Ag & Co. Kgaa | Detergent and cleaning agent with improved enzyme stability |
| DE102022205588A1 (en) | 2022-06-01 | 2023-12-07 | Henkel Ag & Co. Kgaa | DETERGENT AND CLEANING AGENTS WITH IMPROVED ENZYME STABILITY |
| WO2023232193A1 (en) | 2022-06-01 | 2023-12-07 | Henkel Ag & Co. Kgaa | Detergents and cleaning agents with an improved enzyme stability |
| WO2023247664A2 (en) | 2022-06-24 | 2023-12-28 | Novozymes A/S | Lipase variants and compositions comprising such lipase variants |
| EP4324900A1 (en) | 2022-08-17 | 2024-02-21 | Henkel AG & Co. KGaA | Detergent composition comprising enzymes |
| EP4339282A2 (en) | 2014-12-04 | 2024-03-20 | Novozymes A/S | Liquid cleaning compositions comprising protease variants |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FI20085345L (en) * | 2008-04-22 | 2009-10-23 | Kemira Oyj | Method for reducing light-induced yellowing in lignin-containing material |
| CN103194890A (en) * | 2013-04-24 | 2013-07-10 | 太仓市其盛化纤厂 | Pretreatment method of cotton fabric |
| CN108914542A (en) * | 2018-05-15 | 2018-11-30 | 周晓东 | A kind of fabric bleaching agent |
| CN110042647B (en) * | 2019-04-02 | 2022-02-18 | 嘉兴学院 | High-whiteness low-temperature bleaching method for cashmere or wool |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3453268A (en) | 1966-01-18 | 1969-07-01 | Bayer Ag | Substituted v-triazolyl-2-stilbenes and their use for brightening materials |
| US3485831A (en) | 1965-05-20 | 1969-12-23 | Bayer Ag | V-triazolyl-substituted stilbenes |
| US3812044A (en) | 1970-12-28 | 1974-05-21 | Procter & Gamble | Detergent composition containing a polyfunctionally-substituted aromatic acid sequestering agent |
| US3929678A (en) | 1974-08-01 | 1975-12-30 | Procter & Gamble | Detergent composition having enhanced particulate soil removal performance |
| US3984399A (en) | 1967-10-03 | 1976-10-05 | Ciba-Geigy Ag | Bis-stilbene compounds |
| US3994834A (en) | 1972-10-05 | 1976-11-30 | Bayer Aktiengesellschaft | Triazolyl stilbene brighteners method of preparing and detergents therewith |
| US4565647A (en) | 1982-04-26 | 1986-01-21 | The Procter & Gamble Company | Foaming surfactant compositions |
| US4704233A (en) | 1986-11-10 | 1987-11-03 | The Procter & Gamble Company | Detergent compositions containing ethylenediamine-N,N'-disuccinic acid |
| US5969204A (en) | 1996-12-20 | 1999-10-19 | Ciba Specialty Chemicals Corporation | Process for the production of distyryl-biphenyl compounds |
| US6096919A (en) | 1998-05-13 | 2000-08-01 | Ciba Specialty Chemicals Corporation | Process for the preparation of sulphonated distyryl-biphenyl compounds |
| WO2005056782A2 (en) | 2003-12-03 | 2005-06-23 | Genencor International, Inc. | Perhydrolase |
| WO2007136469A2 (en) | 2006-04-14 | 2007-11-29 | Genencor International, Inc. | One-step treatment of textiles |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6096098A (en) * | 1996-10-15 | 2000-08-01 | The Procter & Gamble Company | Asymmetrical bleach activators and compositions employing the same |
| US8518675B2 (en) * | 2005-12-13 | 2013-08-27 | E. I. Du Pont De Nemours And Company | Production of peracids using an enzyme having perhydrolysis activity |
| US7964378B2 (en) * | 2005-12-13 | 2011-06-21 | E.I. Du Pont De Nemours And Company | Production of peracids using an enzyme having perhydrolysis activity |
-
2010
- 2010-02-17 EP EP10705848A patent/EP2403990A2/en not_active Withdrawn
- 2010-02-17 BR BRPI1013219A patent/BRPI1013219A2/en not_active IP Right Cessation
- 2010-02-17 US US13/138,455 patent/US20110302724A1/en not_active Abandoned
- 2010-02-17 CN CN2010800096945A patent/CN102333914A/en active Pending
- 2010-02-17 MX MX2011008656A patent/MX2011008656A/en active IP Right Grant
- 2010-02-17 WO PCT/EP2010/051953 patent/WO2010100028A2/en active Application Filing
- 2010-03-04 TW TW099106262A patent/TW201035407A/en unknown
-
2011
- 2011-08-17 CO CO11104425A patent/CO6362055A2/en active IP Right Grant
Patent Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3485831A (en) | 1965-05-20 | 1969-12-23 | Bayer Ag | V-triazolyl-substituted stilbenes |
| US3453268A (en) | 1966-01-18 | 1969-07-01 | Bayer Ag | Substituted v-triazolyl-2-stilbenes and their use for brightening materials |
| US3984399A (en) | 1967-10-03 | 1976-10-05 | Ciba-Geigy Ag | Bis-stilbene compounds |
| US3812044A (en) | 1970-12-28 | 1974-05-21 | Procter & Gamble | Detergent composition containing a polyfunctionally-substituted aromatic acid sequestering agent |
| US3994834A (en) | 1972-10-05 | 1976-11-30 | Bayer Aktiengesellschaft | Triazolyl stilbene brighteners method of preparing and detergents therewith |
| US3929678A (en) | 1974-08-01 | 1975-12-30 | Procter & Gamble | Detergent composition having enhanced particulate soil removal performance |
| US4565647A (en) | 1982-04-26 | 1986-01-21 | The Procter & Gamble Company | Foaming surfactant compositions |
| US4565647B1 (en) | 1982-04-26 | 1994-04-05 | Procter & Gamble | Foaming surfactant compositions |
| US4704233A (en) | 1986-11-10 | 1987-11-03 | The Procter & Gamble Company | Detergent compositions containing ethylenediamine-N,N'-disuccinic acid |
| US5969204A (en) | 1996-12-20 | 1999-10-19 | Ciba Specialty Chemicals Corporation | Process for the production of distyryl-biphenyl compounds |
| US6096919A (en) | 1998-05-13 | 2000-08-01 | Ciba Specialty Chemicals Corporation | Process for the preparation of sulphonated distyryl-biphenyl compounds |
| WO2005056782A2 (en) | 2003-12-03 | 2005-06-23 | Genencor International, Inc. | Perhydrolase |
| WO2007136469A2 (en) | 2006-04-14 | 2007-11-29 | Genencor International, Inc. | One-step treatment of textiles |
Non-Patent Citations (9)
| Title |
|---|
| "Current Protocols in Molecular Biology", 1994 |
| "Oligonucleotide Synthesis", 1996 |
| "PCR: The Polymerase Chain Reaction", 1994 |
| GANZ, JOURNAL OF COLOR AND APPEARANCE, vol. 1, no. 5, 1972 |
| HALE; MARKHAM: "The Harper Collins Dictionary of Biology", 1991, HARPER PERENNIAL |
| KRIEGLER: "Gene Transfer and Expression: A Laboratory Manual", 1990 |
| SAKAI ET AL., ADVANCES IN APPLIED MICROBIOLOGY, vol. 39, 1993, pages 213 - 294 |
| SAMBROOK ET AL.: "Molecular Cloning: A Laboratory Manual", 1989 |
| SINGLETON ET AL.: "Dictionary of Microbiology and Molecular Biology", 1994, JOHN WILEY AND SONS |
Cited By (259)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013076259A2 (en) | 2011-11-25 | 2013-05-30 | Novozymes A/S | Polypeptides having lysozyme activity and polynucleotides encoding same |
| WO2013076253A1 (en) | 2011-11-25 | 2013-05-30 | Novozymes A/S | Polypeptides having lysozyme activity and polynucleotides encoding same |
| EP3382003A1 (en) | 2011-12-29 | 2018-10-03 | Novozymes A/S | Detergent compositions with lipase variants |
| WO2013098205A2 (en) | 2011-12-29 | 2013-07-04 | Novozymes A/S | Detergent compositions |
| WO2013110766A1 (en) | 2012-01-26 | 2013-08-01 | Novozymes A/S | Use of polypeptides having protease activity in animal feed and detergents |
| WO2013149858A1 (en) | 2012-04-02 | 2013-10-10 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| WO2013167581A1 (en) | 2012-05-07 | 2013-11-14 | Novozymes A/S | Polypeptides having xanthan degrading activity and polynucleotides encoding same |
| WO2013171241A1 (en) | 2012-05-16 | 2013-11-21 | Novozymes A/S | Compositions comprising lipase and methods of use thereof |
| WO2013189972A2 (en) | 2012-06-20 | 2013-12-27 | Novozymes A/S | Use of polypeptides having protease activity in animal feed and detergents |
| EP3553172A1 (en) | 2012-08-16 | 2019-10-16 | Novozymes A/S | Method for treating textile with endoglucanase |
| EP3556836A1 (en) | 2012-12-07 | 2019-10-23 | Novozymes A/S | Preventing adhesion of bacteria |
| WO2014087011A1 (en) | 2012-12-07 | 2014-06-12 | Novozymes A/S | Preventing adhesion of bacteria |
| WO2014096259A1 (en) | 2012-12-21 | 2014-06-26 | Novozymes A/S | Polypeptides having protease activiy and polynucleotides encoding same |
| EP3321360A2 (en) | 2013-01-03 | 2018-05-16 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2014147127A1 (en) | 2013-03-21 | 2014-09-25 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2014184164A1 (en) | 2013-05-14 | 2014-11-20 | Novozymes A/S | Detergent compositions |
| WO2014183921A1 (en) | 2013-05-17 | 2014-11-20 | Novozymes A/S | Polypeptides having alpha amylase activity |
| EP3786269A1 (en) | 2013-06-06 | 2021-03-03 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2014207227A1 (en) | 2013-06-27 | 2014-12-31 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2014207224A1 (en) | 2013-06-27 | 2014-12-31 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2015001017A2 (en) | 2013-07-04 | 2015-01-08 | Novozymes A/S | Polypeptides having anti-redeposition effect and polynucleotides encoding same |
| WO2015004102A1 (en) | 2013-07-09 | 2015-01-15 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| EP3613853A1 (en) | 2013-07-29 | 2020-02-26 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| EP3611260A1 (en) | 2013-07-29 | 2020-02-19 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| EP3309249A1 (en) | 2013-07-29 | 2018-04-18 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| WO2015049370A1 (en) | 2013-10-03 | 2015-04-09 | Novozymes A/S | Detergent composition and use of detergent composition |
| EP3453757A1 (en) | 2013-12-20 | 2019-03-13 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2015112341A1 (en) * | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Fabric treatment composition |
| WO2015112340A1 (en) * | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Method of treating textile fabrics |
| WO2015109972A1 (en) | 2014-01-22 | 2015-07-30 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2015134729A1 (en) | 2014-03-05 | 2015-09-11 | Novozymes A/S | Compositions and methods for improving properties of non-cellulosic textile materials with xyloglucan endotransglycosylase |
| WO2015134737A1 (en) | 2014-03-05 | 2015-09-11 | Novozymes A/S | Compositions and methods for improving properties of cellulosic textile materials with xyloglucan endotransglycosylase |
| EP3521434A1 (en) | 2014-03-12 | 2019-08-07 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2015150457A1 (en) | 2014-04-01 | 2015-10-08 | Novozymes A/S | Polypeptides having alpha amylase activity |
| EP3722406A1 (en) | 2014-04-11 | 2020-10-14 | Novozymes A/S | Detergent composition |
| WO2015158237A1 (en) | 2014-04-15 | 2015-10-22 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| EP3878957A1 (en) | 2014-05-27 | 2021-09-15 | Novozymes A/S | Methods for producing lipases |
| EP3760713A2 (en) | 2014-05-27 | 2021-01-06 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| WO2015181119A2 (en) | 2014-05-27 | 2015-12-03 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| WO2015189371A1 (en) | 2014-06-12 | 2015-12-17 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| EP3878960A1 (en) | 2014-07-04 | 2021-09-15 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3739029A1 (en) | 2014-07-04 | 2020-11-18 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2016079110A2 (en) | 2014-11-19 | 2016-05-26 | Novozymes A/S | Use of enzyme for cleaning |
| WO2016079305A1 (en) | 2014-11-20 | 2016-05-26 | Novozymes A/S | Alicyclobacillus variants and polynucleotides encoding same |
| EP3690037A1 (en) | 2014-12-04 | 2020-08-05 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP4339282A2 (en) | 2014-12-04 | 2024-03-20 | Novozymes A/S | Liquid cleaning compositions comprising protease variants |
| WO2016087401A1 (en) | 2014-12-05 | 2016-06-09 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| EP4067485A2 (en) | 2014-12-05 | 2022-10-05 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| EP3608403A2 (en) | 2014-12-15 | 2020-02-12 | Henkel AG & Co. KGaA | Detergent composition comprising subtilase variants |
| US10760036B2 (en) | 2014-12-15 | 2020-09-01 | Henkel Ag & Co. Kgaa | Detergent composition comprising subtilase variants |
| WO2016096996A1 (en) | 2014-12-16 | 2016-06-23 | Novozymes A/S | Polypeptides having n-acetyl glucosamine oxidase activity |
| EP3741848A2 (en) | 2014-12-19 | 2020-11-25 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| EP3741849A2 (en) | 2014-12-19 | 2020-11-25 | Novozymes A/S | Protease variants and polynucleotides encoding same |
| WO2016164596A2 (en) | 2015-04-07 | 2016-10-13 | Novozymes A/S | Methods for selecting enzymes having lipase activity |
| WO2016162556A1 (en) | 2015-04-10 | 2016-10-13 | Novozymes A/S | Laundry method, use of dnase and detergent composition |
| WO2016162558A1 (en) | 2015-04-10 | 2016-10-13 | Novozymes A/S | Detergent composition |
| WO2016184944A1 (en) | 2015-05-19 | 2016-11-24 | Novozymes A/S | Odor reduction |
| WO2016202739A1 (en) | 2015-06-16 | 2016-12-22 | Novozymes A/S | Polypeptides with lipase activity and polynucleotides encoding same |
| WO2016202785A1 (en) | 2015-06-17 | 2016-12-22 | Novozymes A/S | Container |
| EP3872175A1 (en) | 2015-06-18 | 2021-09-01 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP4071244A1 (en) | 2015-06-18 | 2022-10-12 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| EP3106508A1 (en) | 2015-06-18 | 2016-12-21 | Henkel AG & Co. KGaA | Detergent composition comprising subtilase variants |
| WO2016135351A1 (en) | 2015-06-30 | 2016-09-01 | Novozymes A/S | Laundry detergent composition, method for washing and use of composition |
| EP3929285A2 (en) | 2015-07-01 | 2021-12-29 | Novozymes A/S | Methods of reducing odor |
| EP3950939A2 (en) | 2015-07-06 | 2022-02-09 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| WO2017046232A1 (en) | 2015-09-17 | 2017-03-23 | Henkel Ag & Co. Kgaa | Detergent compositions comprising polypeptides having xanthan degrading activity |
| WO2017046260A1 (en) | 2015-09-17 | 2017-03-23 | Novozymes A/S | Polypeptides having xanthan degrading activity and polynucleotides encoding same |
| EP3708660A2 (en) | 2015-10-07 | 2020-09-16 | Novozymes A/S | Polypeptides |
| WO2017060505A1 (en) | 2015-10-07 | 2017-04-13 | Novozymes A/S | Polypeptides |
| WO2017064253A1 (en) | 2015-10-14 | 2017-04-20 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2017064269A1 (en) | 2015-10-14 | 2017-04-20 | Novozymes A/S | Polypeptide variants |
| EP4324919A2 (en) | 2015-10-14 | 2024-02-21 | Novozymes A/S | Polypeptide variants |
| EP3957711A2 (en) | 2015-10-28 | 2022-02-23 | Novozymes A/S | Detergent composition comprising amylase and protease variants |
| WO2017089366A1 (en) | 2015-11-24 | 2017-06-01 | Novozymes A/S | Polypeptides having protease activity and polynucleotides encoding same |
| WO2017093318A1 (en) | 2015-12-01 | 2017-06-08 | Novozymes A/S | Methods for producing lipases |
| EP3715442A1 (en) | 2016-03-23 | 2020-09-30 | Novozymes A/S | Use of polypeptide having dnase activity for treating fabrics |
| WO2017174769A2 (en) | 2016-04-08 | 2017-10-12 | Novozymes A/S | Detergent compositions and uses of the same |
| EP3693449A1 (en) | 2016-04-29 | 2020-08-12 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2017186943A1 (en) | 2016-04-29 | 2017-11-02 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2017207762A1 (en) | 2016-06-03 | 2017-12-07 | Novozymes A/S | Subtilase variants and polynucleotides encoding same |
| WO2017220422A1 (en) | 2016-06-23 | 2017-12-28 | Novozymes A/S | Use of enzymes, composition and method for removing soil |
| WO2018001959A1 (en) | 2016-06-30 | 2018-01-04 | Novozymes A/S | Lipase variants and compositions comprising surfactant and lipase variant |
| WO2018002261A1 (en) | 2016-07-01 | 2018-01-04 | Novozymes A/S | Detergent compositions |
| WO2018007435A1 (en) | 2016-07-05 | 2018-01-11 | Novozymes A/S | Pectate lyase variants and polynucleotides encoding same |
| WO2018007573A1 (en) | 2016-07-08 | 2018-01-11 | Novozymes A/S | Detergent compositions with galactanase |
| WO2018011277A1 (en) | 2016-07-13 | 2018-01-18 | Novozymes A/S | Bacillus cibi dnase variants |
| WO2018011276A1 (en) | 2016-07-13 | 2018-01-18 | The Procter & Gamble Company | Bacillus cibi dnase variants and uses thereof |
| EP3950941A2 (en) | 2016-07-13 | 2022-02-09 | Novozymes A/S | Dnase polypeptide variants |
| WO2018015295A1 (en) | 2016-07-18 | 2018-01-25 | Novozymes A/S | Lipase variants, polynucleotides encoding same and the use thereof |
| WO2018037062A1 (en) | 2016-08-24 | 2018-03-01 | Novozymes A/S | Gh9 endoglucanase variants and polynucleotides encoding same |
| WO2018037064A1 (en) | 2016-08-24 | 2018-03-01 | Henkel Ag & Co. Kgaa | Detergent compositions comprising xanthan lyase variants i |
| WO2018037065A1 (en) | 2016-08-24 | 2018-03-01 | Henkel Ag & Co. Kgaa | Detergent composition comprising gh9 endoglucanase variants i |
| WO2018037061A1 (en) | 2016-08-24 | 2018-03-01 | Novozymes A/S | Xanthan lyase variants and polynucleotides encoding same |
| WO2018060216A1 (en) | 2016-09-29 | 2018-04-05 | Novozymes A/S | Use of enzyme for washing, method for washing and warewashing composition |
| WO2018077938A1 (en) | 2016-10-25 | 2018-05-03 | Novozymes A/S | Detergent compositions |
| WO2018083093A1 (en) | 2016-11-01 | 2018-05-11 | Novozymes A/S | Multi-core granules |
| WO2018099762A1 (en) | 2016-12-01 | 2018-06-07 | Basf Se | Stabilization of enzymes in compositions |
| WO2018108865A1 (en) | 2016-12-12 | 2018-06-21 | Novozymes A/S | Use of polypeptides |
| WO2018177936A1 (en) | 2017-03-31 | 2018-10-04 | Novozymes A/S | Polypeptides having dnase activity |
| WO2018177938A1 (en) | 2017-03-31 | 2018-10-04 | Novozymes A/S | Polypeptides having dnase activity |
| WO2018178061A1 (en) | 2017-03-31 | 2018-10-04 | Novozymes A/S | Polypeptides having rnase activity |
| WO2018185150A1 (en) | 2017-04-04 | 2018-10-11 | Novozymes A/S | Polypeptides |
| WO2018185152A1 (en) | 2017-04-04 | 2018-10-11 | Novozymes A/S | Polypeptide compositions and uses thereof |
| WO2018185181A1 (en) | 2017-04-04 | 2018-10-11 | Novozymes A/S | Glycosyl hydrolases |
| EP3385362A1 (en) | 2017-04-05 | 2018-10-10 | Henkel AG & Co. KGaA | Detergent compositions comprising fungal mannanases |
| EP3385361A1 (en) | 2017-04-05 | 2018-10-10 | Henkel AG & Co. KGaA | Detergent compositions comprising bacterial mannanases |
| WO2018184767A1 (en) | 2017-04-05 | 2018-10-11 | Henkel Ag & Co. Kgaa | Detergent compositions comprising bacterial mannanases |
| WO2018185285A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018184818A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018184817A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018185280A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018184873A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2018185267A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018185269A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| EP3626809A1 (en) | 2017-04-06 | 2020-03-25 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2018184816A1 (en) | 2017-04-06 | 2018-10-11 | Novozymes A/S | Cleaning compositions and uses thereof |
| EP3967756A1 (en) | 2017-04-06 | 2022-03-16 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2018202846A1 (en) | 2017-05-05 | 2018-11-08 | Novozymes A/S | Compositions comprising lipase and sulfite |
| EP3401385A1 (en) | 2017-05-08 | 2018-11-14 | Henkel AG & Co. KGaA | Detergent composition comprising polypeptide comprising carbohydrate-binding domain |
| WO2018206535A1 (en) | 2017-05-08 | 2018-11-15 | Novozymes A/S | Carbohydrate-binding domain and polynucleotides encoding the same |
| WO2018206178A1 (en) | 2017-05-08 | 2018-11-15 | Henkel Ag & Co. Kgaa | Detergent composition comprising polypeptide comprising carbohydrate-binding domain |
| WO2019038057A1 (en) | 2017-08-24 | 2019-02-28 | Novozymes A/S | Xanthan lyase variants and polynucleotides encoding same |
| WO2019038058A1 (en) | 2017-08-24 | 2019-02-28 | Novozymes A/S | Gh9 endoglucanase variants and polynucleotides encoding same |
| WO2019038059A1 (en) | 2017-08-24 | 2019-02-28 | Henkel Ag & Co. Kgaa | Detergent compositions comprising gh9 endoglucanase variants ii |
| WO2019038060A1 (en) | 2017-08-24 | 2019-02-28 | Henkel Ag & Co. Kgaa | Detergent composition comprising xanthan lyase variants ii |
| WO2019057758A1 (en) | 2017-09-20 | 2019-03-28 | Novozymes A/S | Use of enzymes for improving water absorption and/or whiteness |
| WO2019057902A1 (en) | 2017-09-22 | 2019-03-28 | Novozymes A/S | Novel polypeptides |
| WO2019067390A1 (en) | 2017-09-27 | 2019-04-04 | The Procter & Gamble Company | Detergent compositions comprising lipases |
| WO2019063499A1 (en) | 2017-09-27 | 2019-04-04 | Novozymes A/S | Lipase variants and microcapsule compositions comprising such lipase variants |
| WO2019076800A1 (en) | 2017-10-16 | 2019-04-25 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2019076833A1 (en) | 2017-10-16 | 2019-04-25 | Novozymes A/S | Low dusting granules |
| WO2019076834A1 (en) | 2017-10-16 | 2019-04-25 | Novozymes A/S | Low dusting granules |
| WO2019081721A1 (en) | 2017-10-27 | 2019-05-02 | Novozymes A/S | Dnase variants |
| WO2019084350A1 (en) | 2017-10-27 | 2019-05-02 | The Procter & Gamble Company | Detergent compositions comprising polypeptide variants |
| WO2019084349A1 (en) | 2017-10-27 | 2019-05-02 | The Procter & Gamble Company | Detergent compositions comprising polypeptide variants |
| WO2019081724A1 (en) | 2017-10-27 | 2019-05-02 | Novozymes A/S | Dnase variants |
| DE102017125560A1 (en) | 2017-11-01 | 2019-05-02 | Henkel Ag & Co. Kgaa | CLEANSING COMPOSITIONS CONTAINING DISPERSINE III |
| WO2019086520A1 (en) | 2017-11-01 | 2019-05-09 | Henkel Ag & Co. Kgaa | Cleaning compositions containing dispersins i |
| WO2019086528A1 (en) | 2017-11-01 | 2019-05-09 | Novozymes A/S | Polypeptides and compositions comprising such polypeptides |
| WO2019086526A1 (en) | 2017-11-01 | 2019-05-09 | Henkel Ag & Co. Kgaa | Cleaning compositions containing dispersins iii |
| WO2019086521A1 (en) | 2017-11-01 | 2019-05-09 | Henkel Ag & Co. Kgaa | Cleaning compositions containing dispersins ii |
| WO2019086532A1 (en) | 2017-11-01 | 2019-05-09 | Novozymes A/S | Methods for cleaning medical devices |
| WO2019086530A1 (en) | 2017-11-01 | 2019-05-09 | Novozymes A/S | Polypeptides and compositions comprising such polypeptides |
| DE102017125559A1 (en) | 2017-11-01 | 2019-05-02 | Henkel Ag & Co. Kgaa | CLEANSING COMPOSITIONS CONTAINING DISPERSINE II |
| DE102017125558A1 (en) | 2017-11-01 | 2019-05-02 | Henkel Ag & Co. Kgaa | CLEANING COMPOSITIONS CONTAINING DISPERSINE I |
| WO2019105780A1 (en) | 2017-11-29 | 2019-06-06 | Basf Se | Compositions, their manufacture and use |
| WO2019105781A1 (en) | 2017-11-29 | 2019-06-06 | Basf Se | Storage-stable enzyme preparations, their production and use |
| WO2019110462A1 (en) | 2017-12-04 | 2019-06-13 | Novozymes A/S | Lipase variants and polynucleotides encoding same |
| WO2019121057A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Laundry formulation for removing fatty compounds having a melting temperature>30°c deposited on textiles |
| WO2019162000A1 (en) | 2018-02-23 | 2019-08-29 | Henkel Ag & Co. Kgaa | Detergent composition comprising xanthan lyase and endoglucanase variants |
| WO2019180111A1 (en) | 2018-03-23 | 2019-09-26 | Novozymes A/S | Subtilase variants and compositions comprising same |
| WO2019201793A1 (en) | 2018-04-17 | 2019-10-24 | Novozymes A/S | Polypeptides comprising carbohydrate binding activity in detergent compositions and their use in reducing wrinkles in textile or fabric. |
| WO2019201636A1 (en) | 2018-04-19 | 2019-10-24 | Basf Se | Compositions and polymers useful for such compositions |
| WO2019201785A1 (en) | 2018-04-19 | 2019-10-24 | Novozymes A/S | Stabilized cellulase variants |
| WO2019201783A1 (en) | 2018-04-19 | 2019-10-24 | Novozymes A/S | Stabilized cellulase variants |
| WO2019238761A1 (en) | 2018-06-15 | 2019-12-19 | Basf Se | Water soluble multilayer films containing wash active chemicals and enzymes |
| WO2020002604A1 (en) | 2018-06-28 | 2020-01-02 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2020002608A1 (en) | 2018-06-29 | 2020-01-02 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2020002255A1 (en) | 2018-06-29 | 2020-01-02 | Novozymes A/S | Subtilase variants and compositions comprising same |
| WO2020007863A1 (en) | 2018-07-02 | 2020-01-09 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020007875A1 (en) | 2018-07-03 | 2020-01-09 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020008024A1 (en) | 2018-07-06 | 2020-01-09 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020008043A1 (en) | 2018-07-06 | 2020-01-09 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020030623A1 (en) | 2018-08-10 | 2020-02-13 | Basf Se | Packaging unit comprising a detergent composition containing an enzyme and at least one chelating agent |
| WO2020070063A2 (en) | 2018-10-01 | 2020-04-09 | Novozymes A/S | Detergent compositions and uses thereof |
| WO2020070011A1 (en) | 2018-10-02 | 2020-04-09 | Novozymes A/S | Cleaning composition |
| WO2020070209A1 (en) | 2018-10-02 | 2020-04-09 | Novozymes A/S | Cleaning composition |
| WO2020070014A1 (en) | 2018-10-02 | 2020-04-09 | Novozymes A/S | Cleaning composition comprising anionic surfactant and a polypeptide having rnase activity |
| WO2020070199A1 (en) | 2018-10-03 | 2020-04-09 | Novozymes A/S | Polypeptides having alpha-mannan degrading activity and polynucleotides encoding same |
| WO2020070249A1 (en) | 2018-10-03 | 2020-04-09 | Novozymes A/S | Cleaning compositions |
| WO2020069914A1 (en) | 2018-10-05 | 2020-04-09 | Basf Se | Compounds stabilizing amylases in liquids |
| WO2020069915A1 (en) | 2018-10-05 | 2020-04-09 | Basf Se | Compounds stabilizing hydrolases in liquids |
| WO2020069913A1 (en) | 2018-10-05 | 2020-04-09 | Basf Se | Compounds stabilizing hydrolases in liquids |
| WO2020074498A1 (en) | 2018-10-09 | 2020-04-16 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020074499A1 (en) | 2018-10-09 | 2020-04-16 | Novozymes A/S | Cleaning compositions and uses thereof |
| WO2020074545A1 (en) | 2018-10-11 | 2020-04-16 | Novozymes A/S | Cleaning compositions and uses thereof |
| EP3647397A1 (en) | 2018-10-31 | 2020-05-06 | Henkel AG & Co. KGaA | Cleaning compositions containing dispersins iv |
| EP3647398A1 (en) | 2018-10-31 | 2020-05-06 | Henkel AG & Co. KGaA | Cleaning compositions containing dispersins v |
| WO2020088957A1 (en) | 2018-10-31 | 2020-05-07 | Henkel Ag & Co. Kgaa | Cleaning compositions containing dispersins iv |
| WO2020088958A1 (en) | 2018-10-31 | 2020-05-07 | Henkel Ag & Co. Kgaa | Cleaning compositions containing dispersins v |
| WO2020104231A1 (en) | 2018-11-19 | 2020-05-28 | Basf Se | Powders and granules containing a chelating agent and an enzyme |
| WO2020114965A1 (en) | 2018-12-03 | 2020-06-11 | Novozymes A/S | LOW pH POWDER DETERGENT COMPOSITION |
| WO2020114968A1 (en) | 2018-12-03 | 2020-06-11 | Novozymes A/S | Powder detergent compositions |
| WO2020127775A1 (en) | 2018-12-21 | 2020-06-25 | Novozymes A/S | Detergent pouch comprising metalloproteases |
| WO2020127796A2 (en) | 2018-12-21 | 2020-06-25 | Novozymes A/S | Polypeptides having peptidoglycan degrading activity and polynucleotides encoding same |
| EP3677676A1 (en) | 2019-01-03 | 2020-07-08 | Basf Se | Compounds stabilizing amylases in liquids |
| EP3702452A1 (en) | 2019-03-01 | 2020-09-02 | Novozymes A/S | Detergent compositions comprising two proteases |
| WO2020178102A1 (en) | 2019-03-01 | 2020-09-10 | Novozymes A/S | Detergent compositions comprising two proteases |
| WO2020182521A1 (en) | 2019-03-08 | 2020-09-17 | Basf Se | Cationic surfactant and its use in laundry detergent compositions |
| WO2020188095A1 (en) | 2019-03-21 | 2020-09-24 | Novozymes A/S | Alpha-amylase variants and polynucleotides encoding same |
| WO2020201403A1 (en) | 2019-04-03 | 2020-10-08 | Novozymes A/S | Polypeptides having beta-glucanase activity, polynucleotides encoding same and uses thereof in cleaning and detergent compositions |
| WO2020207944A1 (en) | 2019-04-10 | 2020-10-15 | Novozymes A/S | Polypeptide variants |
| WO2020208056A1 (en) | 2019-04-12 | 2020-10-15 | Novozymes A/S | Stabilized glycoside hydrolase variants |
| WO2020229480A1 (en) | 2019-05-14 | 2020-11-19 | Basf Se | Compounds stabilizing hydrolases in liquids |
| WO2021009067A1 (en) | 2019-07-12 | 2021-01-21 | Novozymes A/S | Enzymatic emulsions for detergents |
| WO2021037895A1 (en) | 2019-08-27 | 2021-03-04 | Novozymes A/S | Detergent composition |
| WO2021037878A1 (en) | 2019-08-27 | 2021-03-04 | Novozymes A/S | Composition comprising a lipase |
| WO2021053127A1 (en) | 2019-09-19 | 2021-03-25 | Novozymes A/S | Detergent composition |
| WO2021064068A1 (en) | 2019-10-03 | 2021-04-08 | Novozymes A/S | Polypeptides comprising at least two carbohydrate binding domains |
| WO2021074430A1 (en) | 2019-10-18 | 2021-04-22 | Basf Se | Storage-stable hydrolase containing liquids |
| WO2021105336A1 (en) | 2019-11-29 | 2021-06-03 | Basf Se | Compositions comprising polymer and enzyme |
| WO2021115912A1 (en) | 2019-12-09 | 2021-06-17 | Basf Se | Formulations comprising a hydrophobically modified polyethyleneimine and one or more enzymes |
| WO2021123307A2 (en) | 2019-12-20 | 2021-06-24 | Novozymes A/S | Polypeptides having proteolytic activity and use thereof |
| WO2021122121A1 (en) | 2019-12-20 | 2021-06-24 | Henkel Ag & Co. Kgaa | Cleaning compositions comprising dispersins ix |
| WO2021122120A2 (en) | 2019-12-20 | 2021-06-24 | Henkel Ag & Co. Kgaa | Cleaning compositions comprising dispersins viii |
| WO2021122118A1 (en) | 2019-12-20 | 2021-06-24 | Henkel Ag & Co. Kgaa | Cleaning compositions comprising dispersins vi |
| WO2021121394A1 (en) | 2019-12-20 | 2021-06-24 | Novozymes A/S | Stabilized liquid boron-free enzyme compositions |
| WO2021122117A1 (en) | 2019-12-20 | 2021-06-24 | Henkel Ag & Co. Kgaa | Cleaning composition coprising a dispersin and a carbohydrase |
| WO2021130167A1 (en) | 2019-12-23 | 2021-07-01 | Novozymes A/S | Enzyme compositions and uses thereof |
| WO2021148364A1 (en) | 2020-01-23 | 2021-07-29 | Novozymes A/S | Enzyme compositions and uses thereof |
| WO2021152120A1 (en) | 2020-01-31 | 2021-08-05 | Novozymes A/S | Mannanase variants and polynucleotides encoding same |
| WO2021152123A1 (en) | 2020-01-31 | 2021-08-05 | Novozymes A/S | Mannanase variants and polynucleotides encoding same |
| EP3892708A1 (en) | 2020-04-06 | 2021-10-13 | Henkel AG & Co. KGaA | Cleaning compositions comprising dispersin variants |
| WO2021204838A1 (en) | 2020-04-08 | 2021-10-14 | Novozymes A/S | Carbohydrate binding module variants |
| WO2021214059A1 (en) | 2020-04-21 | 2021-10-28 | Novozymes A/S | Cleaning compositions comprising polypeptides having fructan degrading activity |
| WO2021239818A1 (en) | 2020-05-26 | 2021-12-02 | Novozymes A/S | Subtilase variants and compositions comprising same |
| WO2021259099A1 (en) | 2020-06-24 | 2021-12-30 | Novozymes A/S | Use of cellulases for removing dust mite from textile |
| WO2022008387A1 (en) | 2020-07-08 | 2022-01-13 | Henkel Ag & Co. Kgaa | Cleaning compositions and uses thereof |
| EP3936593A1 (en) | 2020-07-08 | 2022-01-12 | Henkel AG & Co. KGaA | Cleaning compositions and uses thereof |
| WO2022008732A1 (en) | 2020-07-10 | 2022-01-13 | Basf Se | Enhancing the activity of antimicrobial preservatives |
| WO2022043321A2 (en) | 2020-08-25 | 2022-03-03 | Novozymes A/S | Variants of a family 44 xyloglucanase |
| WO2022043563A1 (en) | 2020-08-28 | 2022-03-03 | Novozymes A/S | Polyester degrading protease variants |
| WO2022043547A1 (en) | 2020-08-28 | 2022-03-03 | Novozymes A/S | Protease variants with improved solubility |
| WO2022063699A1 (en) | 2020-09-22 | 2022-03-31 | Basf Se | Improved combination of protease and protease inhibitor with secondary enzyme |
| WO2022074037A2 (en) | 2020-10-07 | 2022-04-14 | Novozymes A/S | Alpha-amylase variants |
| WO2022084303A2 (en) | 2020-10-20 | 2022-04-28 | Novozymes A/S | Use of polypeptides having dnase activity |
| WO2022090320A1 (en) | 2020-10-28 | 2022-05-05 | Novozymes A/S | Use of lipoxygenase |
| WO2022090361A2 (en) | 2020-10-29 | 2022-05-05 | Novozymes A/S | Lipase variants and compositions comprising such lipase variants |
| WO2022103725A1 (en) | 2020-11-13 | 2022-05-19 | Novozymes A/S | Detergent composition comprising a lipase |
| WO2022106404A1 (en) | 2020-11-18 | 2022-05-27 | Novozymes A/S | Combination of proteases |
| WO2022106400A1 (en) | 2020-11-18 | 2022-05-27 | Novozymes A/S | Combination of immunochemically different proteases |
| EP4032966A1 (en) | 2021-01-22 | 2022-07-27 | Novozymes A/S | Liquid enzyme composition with sulfite scavenger |
| WO2022157311A1 (en) | 2021-01-22 | 2022-07-28 | Novozymes A/S | Liquid enzyme composition with sulfite scavenger |
| WO2022162043A1 (en) | 2021-01-28 | 2022-08-04 | Novozymes A/S | Lipase with low malodor generation |
| WO2022167251A1 (en) | 2021-02-04 | 2022-08-11 | Henkel Ag & Co. Kgaa | Detergent composition comprising xanthan lyase and endoglucanase variants with improved stability |
| EP4039806A1 (en) | 2021-02-04 | 2022-08-10 | Henkel AG & Co. KGaA | Detergent composition comprising xanthan lyase and endoglucanase variants with im-proved stability |
| WO2022171872A1 (en) | 2021-02-12 | 2022-08-18 | Novozymes A/S | Stabilized biological detergents |
| WO2022171780A2 (en) | 2021-02-12 | 2022-08-18 | Novozymes A/S | Alpha-amylase variants |
| WO2022189521A1 (en) | 2021-03-12 | 2022-09-15 | Novozymes A/S | Polypeptide variants |
| WO2022194673A1 (en) | 2021-03-15 | 2022-09-22 | Novozymes A/S | Dnase variants |
| WO2022194668A1 (en) | 2021-03-15 | 2022-09-22 | Novozymes A/S | Polypeptide variants |
| EP4060036A1 (en) | 2021-03-15 | 2022-09-21 | Novozymes A/S | Polypeptide variants |
| WO2022199418A1 (en) | 2021-03-26 | 2022-09-29 | Novozymes A/S | Detergent composition with reduced polymer content |
| WO2022268885A1 (en) | 2021-06-23 | 2022-12-29 | Novozymes A/S | Alpha-amylase polypeptides |
| WO2023116569A1 (en) | 2021-12-21 | 2023-06-29 | Novozymes A/S | Composition comprising a lipase and a booster |
| EP4206309A1 (en) | 2021-12-30 | 2023-07-05 | Novozymes A/S | Protein particles with improved whiteness |
| WO2023126254A1 (en) | 2021-12-30 | 2023-07-06 | Novozymes A/S | Protein particles with improved whiteness |
| EP4234664A1 (en) | 2022-02-24 | 2023-08-30 | Evonik Operations GmbH | Composition comprising glucolipids and enzymes |
| WO2023165507A1 (en) | 2022-03-02 | 2023-09-07 | Novozymes A/S | Use of xyloglucanase for improvement of sustainability of detergents |
| WO2023165950A1 (en) | 2022-03-04 | 2023-09-07 | Novozymes A/S | Dnase variants and compositions |
| WO2023194204A1 (en) | 2022-04-08 | 2023-10-12 | Novozymes A/S | Hexosaminidase variants and compositions |
| DE102022205588A1 (en) | 2022-06-01 | 2023-12-07 | Henkel Ag & Co. Kgaa | DETERGENT AND CLEANING AGENTS WITH IMPROVED ENZYME STABILITY |
| WO2023232194A1 (en) | 2022-06-01 | 2023-12-07 | Henkel Ag & Co. Kgaa | Detergents and cleaning agents with an improved enzyme stability |
| WO2023232193A1 (en) | 2022-06-01 | 2023-12-07 | Henkel Ag & Co. Kgaa | Detergents and cleaning agents with an improved enzyme stability |
| DE102022205591A1 (en) | 2022-06-01 | 2023-12-07 | Henkel Ag & Co. Kgaa | DETERGENT AND CLEANING AGENTS WITH IMPROVED ENZYME STABILITY |
| DE102022205593A1 (en) | 2022-06-01 | 2023-12-07 | Henkel Ag & Co. Kgaa | DETERGENT AND CLEANING AGENTS WITH IMPROVED ENZYME STABILITY |
| WO2023232192A1 (en) | 2022-06-01 | 2023-12-07 | Henkel Ag & Co. Kgaa | Detergent and cleaning agent with improved enzyme stability |
| WO2023247664A2 (en) | 2022-06-24 | 2023-12-28 | Novozymes A/S | Lipase variants and compositions comprising such lipase variants |
| EP4324900A1 (en) | 2022-08-17 | 2024-02-21 | Henkel AG & Co. KGaA | Detergent composition comprising enzymes |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201035407A (en) | 2010-10-01 |
| CN102333914A (en) | 2012-01-25 |
| CO6362055A2 (en) | 2012-01-20 |
| MX2011008656A (en) | 2011-09-06 |
| US20110302724A1 (en) | 2011-12-15 |
| WO2010100028A3 (en) | 2011-01-27 |
| BRPI1013219A2 (en) | 2016-03-29 |
| EP2403990A2 (en) | 2012-01-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20110302724A1 (en) | Enzymatic textile bleach-whitening methods | |
| AU2010255863B2 (en) | Enzymatic textile colour modification | |
| AU2009291795B2 (en) | Enzymatic textile bleaching compositions and methods of use thereof | |
| EP2064385B1 (en) | Enzymatic treatment of textiles using a pectate lyase from bacillus subtilis | |
| CA2150563A1 (en) | Enhancement of enzyme reactions | |
| BRPI0709978A2 (en) | one-step treatment of textile products | |
| US20140007356A1 (en) | Color modification of sized fabric | |
| EP0996785A1 (en) | A method of treating polyester fabrics | |
| US20120149269A1 (en) | Combined Textile Abrading And Color Modification | |
| EP2403809B1 (en) | Method of oxidative decolorization of dyes with enzymatically generated peracid | |
| EP2553160B1 (en) | Treatment of keratinous fibers with an enzyme having perhydrolase activity | |
| EP3668960B1 (en) | Method of treating fabrics | |
| Tavčer | Biotechnology in textiles–an opportunity of saving water |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080009694.5 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010705848 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11104425 Country of ref document: CO Ref document number: MX/A/2011/008656 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13138455 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10705848 Country of ref document: EP Kind code of ref document: A2 |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: PI1013219 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: PI1013219 Country of ref document: BR Kind code of ref document: A2 Effective date: 20110905 |