WO2009024541A2 - Isoxazoline compositions and their use as antiparasitics - Google Patents
Isoxazoline compositions and their use as antiparasitics Download PDFInfo
- Publication number
- WO2009024541A2 WO2009024541A2 PCT/EP2008/060732 EP2008060732W WO2009024541A2 WO 2009024541 A2 WO2009024541 A2 WO 2009024541A2 EP 2008060732 W EP2008060732 W EP 2008060732W WO 2009024541 A2 WO2009024541 A2 WO 2009024541A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- hydrogen
- isoxazoline
- methyl
- halogen
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 77
- WEQPBCSPRXFQQS-UHFFFAOYSA-N 4,5-dihydro-1,2-oxazole Chemical compound C1CC=NO1 WEQPBCSPRXFQQS-UHFFFAOYSA-N 0.000 title claims description 171
- 239000003096 antiparasitic agent Substances 0.000 title description 5
- 229940125687 antiparasitic agent Drugs 0.000 title description 4
- 230000002141 anti-parasite Effects 0.000 title description 2
- 241001465754 Metazoa Species 0.000 claims abstract description 98
- 238000000034 method Methods 0.000 claims abstract description 71
- 206010061217 Infestation Diseases 0.000 claims abstract description 36
- 239000003814 drug Substances 0.000 claims abstract description 16
- 230000003071 parasitic effect Effects 0.000 claims abstract description 4
- -1 halomethoxymethyl Chemical group 0.000 claims description 518
- 239000001257 hydrogen Substances 0.000 claims description 172
- 229910052739 hydrogen Inorganic materials 0.000 claims description 172
- 150000002431 hydrogen Chemical class 0.000 claims description 156
- 125000001424 substituent group Chemical group 0.000 claims description 82
- 150000003839 salts Chemical class 0.000 claims description 68
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 claims description 55
- 229910052736 halogen Inorganic materials 0.000 claims description 54
- 150000002367 halogens Chemical class 0.000 claims description 54
- 125000004970 halomethyl group Chemical group 0.000 claims description 53
- 125000005745 ethoxymethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])* 0.000 claims description 46
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 46
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 claims description 46
- 238000011282 treatment Methods 0.000 claims description 45
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 44
- 125000004672 ethylcarbonyl group Chemical group [H]C([H])([H])C([H])([H])C(*)=O 0.000 claims description 42
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 claims description 41
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 claims description 40
- 125000004674 methylcarbonyl group Chemical group CC(=O)* 0.000 claims description 39
- 239000012453 solvate Substances 0.000 claims description 35
- 125000006255 cyclopropyl carbonyl group Chemical group [H]C1([H])C([H])([H])C1([H])C(*)=O 0.000 claims description 34
- 125000001309 chloro group Chemical group Cl* 0.000 claims description 26
- 125000005767 propoxymethyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])[#8]C([H])([H])* 0.000 claims description 26
- 125000004673 propylcarbonyl group Chemical group 0.000 claims description 25
- 239000004480 active ingredient Substances 0.000 claims description 24
- 125000004969 haloethyl group Chemical group 0.000 claims description 23
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 23
- 125000001246 bromo group Chemical group Br* 0.000 claims description 19
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 claims description 17
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 16
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 15
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 15
- 229920005555 halobutyl Polymers 0.000 claims description 13
- 125000004968 halobutyl group Chemical group 0.000 claims description 13
- 229910052760 oxygen Inorganic materials 0.000 claims description 10
- 229910052717 sulfur Inorganic materials 0.000 claims description 10
- 206010014143 Ectoparasitic Infestations Diseases 0.000 claims description 8
- 230000000590 parasiticidal effect Effects 0.000 claims description 7
- 125000002346 iodo group Chemical group I* 0.000 claims description 6
- 241000282326 Felis catus Species 0.000 claims description 5
- 125000001153 fluoro group Chemical group F* 0.000 claims description 5
- 208000000292 ehrlichiosis Diseases 0.000 claims description 4
- 230000001225 therapeutic effect Effects 0.000 claims description 4
- 201000004624 Dermatitis Diseases 0.000 claims description 3
- 206010020751 Hypersensitivity Diseases 0.000 claims description 3
- 208000016604 Lyme disease Diseases 0.000 claims description 3
- 206010039207 Rocky Mountain Spotted Fever Diseases 0.000 claims description 3
- 208000026935 allergic disease Diseases 0.000 claims description 3
- 230000007815 allergy Effects 0.000 claims description 3
- 208000007502 anemia Diseases 0.000 claims description 3
- 208000006004 Flea Infestations Diseases 0.000 claims description 2
- 201000001064 tick infestation Diseases 0.000 claims description 2
- 150000002547 isoxazolines Chemical class 0.000 abstract description 70
- 244000045947 parasite Species 0.000 abstract description 30
- SWQGVHNDAKEOQN-UHFFFAOYSA-N 4-(4,5-dihydro-1,2-oxazol-3-yl)benzamide Chemical class C1=CC(C(=O)N)=CC=C1C1=NOCC1 SWQGVHNDAKEOQN-UHFFFAOYSA-N 0.000 abstract description 3
- JCZOHARKHROBPF-UHFFFAOYSA-N 4-(4,5-dihydro-1,2-oxazol-3-yl)benzenecarbothioamide Chemical class C1=CC(C(=S)N)=CC=C1C1=NOCC1 JCZOHARKHROBPF-UHFFFAOYSA-N 0.000 abstract description 3
- 229940054066 benzamide antipsychotics Drugs 0.000 abstract description 2
- 150000001875 compounds Chemical class 0.000 description 81
- 235000002639 sodium chloride Nutrition 0.000 description 48
- 241000238876 Acari Species 0.000 description 37
- 230000037396 body weight Effects 0.000 description 32
- 241000258242 Siphonaptera Species 0.000 description 28
- 241000699670 Mus sp. Species 0.000 description 25
- 230000000694 effects Effects 0.000 description 20
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical class CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 19
- 238000009472 formulation Methods 0.000 description 19
- 238000001727 in vivo Methods 0.000 description 19
- 241000282472 Canis lupus familiaris Species 0.000 description 18
- 244000078703 ectoparasite Species 0.000 description 18
- 241000258924 Ctenocephalides felis Species 0.000 description 16
- 238000002360 preparation method Methods 0.000 description 16
- 239000000243 solution Substances 0.000 description 16
- 239000002904 solvent Substances 0.000 description 16
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 15
- ZOCSXAVNDGMNBV-UHFFFAOYSA-N 5-amino-1-[2,6-dichloro-4-(trifluoromethyl)phenyl]-4-[(trifluoromethyl)sulfinyl]-1H-pyrazole-3-carbonitrile Chemical compound NC1=C(S(=O)C(F)(F)F)C(C#N)=NN1C1=C(Cl)C=C(C(F)(F)F)C=C1Cl ZOCSXAVNDGMNBV-UHFFFAOYSA-N 0.000 description 14
- 239000005899 Fipronil Substances 0.000 description 14
- 229940013764 fipronil Drugs 0.000 description 14
- 239000013642 negative control Substances 0.000 description 14
- 241000700198 Cavia Species 0.000 description 13
- 230000005764 inhibitory process Effects 0.000 description 13
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- 210000003491 skin Anatomy 0.000 description 11
- 230000009286 beneficial effect Effects 0.000 description 10
- 241000124008 Mammalia Species 0.000 description 9
- 241001481696 Rhipicephalus sanguineus Species 0.000 description 9
- 240000000528 Ricinus communis Species 0.000 description 9
- 239000002253 acid Substances 0.000 description 9
- 239000002585 base Substances 0.000 description 9
- 238000002474 experimental method Methods 0.000 description 9
- 239000004544 spot-on Substances 0.000 description 9
- 239000005660 Abamectin Substances 0.000 description 8
- 241000699666 Mus <mouse, genus> Species 0.000 description 8
- 238000007920 subcutaneous administration Methods 0.000 description 8
- 210000001519 tissue Anatomy 0.000 description 8
- 241001414835 Cimicidae Species 0.000 description 7
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 7
- 239000003795 chemical substances by application Substances 0.000 description 7
- 150000002148 esters Chemical class 0.000 description 7
- 229920001223 polyethylene glycol Polymers 0.000 description 7
- 239000000651 prodrug Substances 0.000 description 7
- 229940002612 prodrug Drugs 0.000 description 7
- 230000000699 topical effect Effects 0.000 description 7
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- 206010004194 Bed bug infestation Diseases 0.000 description 6
- RRZXIRBKKLTSOM-XPNPUAGNSA-N avermectin B1a Chemical class C1=C[C@H](C)[C@@H]([C@@H](C)CC)O[C@]11O[C@H](C\C=C(C)\[C@@H](O[C@@H]2O[C@@H](C)[C@H](O[C@@H]3O[C@@H](C)[C@H](O)[C@@H](OC)C3)[C@@H](OC)C2)[C@@H](C)\C=C\C=C/2[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\2)O)C[C@H]4C1 RRZXIRBKKLTSOM-XPNPUAGNSA-N 0.000 description 6
- 210000004369 blood Anatomy 0.000 description 6
- 239000008280 blood Substances 0.000 description 6
- 238000002648 combination therapy Methods 0.000 description 6
- 201000010099 disease Diseases 0.000 description 6
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 6
- 235000019441 ethanol Nutrition 0.000 description 6
- LZCLXQDLBQLTDK-UHFFFAOYSA-N ethyl 2-hydroxypropanoate Chemical compound CCOC(=O)C(C)O LZCLXQDLBQLTDK-UHFFFAOYSA-N 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 231100000957 no side effect Toxicity 0.000 description 6
- 244000052769 pathogen Species 0.000 description 6
- 0 *c1cnc(*)nc1 Chemical compound *c1cnc(*)nc1 0.000 description 5
- AZSNMRSAGSSBNP-UHFFFAOYSA-N 22,23-dihydroavermectin B1a Natural products C1CC(C)C(C(C)CC)OC21OC(CC=C(C)C(OC1OC(C)C(OC3OC(C)C(O)C(OC)C3)C(OC)C1)C(C)C=CC=C1C3(C(C(=O)O4)C=C(C)C(O)C3OC1)O)CC4C2 AZSNMRSAGSSBNP-UHFFFAOYSA-N 0.000 description 5
- SPBDXSGPUHCETR-JFUDTMANSA-N 8883yp2r6d Chemical compound O1[C@@H](C)[C@H](O)[C@@H](OC)C[C@@H]1O[C@@H]1[C@@H](OC)C[C@H](O[C@@H]2C(=C/C[C@@H]3C[C@@H](C[C@@]4(O[C@@H]([C@@H](C)CC4)C(C)C)O3)OC(=O)[C@@H]3C=C(C)[C@@H](O)[C@H]4OC\C([C@@]34O)=C/C=C/[C@@H]2C)/C)O[C@H]1C.C1C[C@H](C)[C@@H]([C@@H](C)CC)O[C@@]21O[C@H](C\C=C(C)\[C@@H](O[C@@H]1O[C@@H](C)[C@H](O[C@@H]3O[C@@H](C)[C@H](O)[C@@H](OC)C3)[C@@H](OC)C1)[C@@H](C)\C=C\C=C/1[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\1)O)C[C@H]4C2 SPBDXSGPUHCETR-JFUDTMANSA-N 0.000 description 5
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 5
- 239000002202 Polyethylene glycol Substances 0.000 description 5
- 230000000895 acaricidal effect Effects 0.000 description 5
- 239000000872 buffer Substances 0.000 description 5
- 235000014113 dietary fatty acids Nutrition 0.000 description 5
- 239000000194 fatty acid Substances 0.000 description 5
- 229930195729 fatty acid Natural products 0.000 description 5
- 229960002418 ivermectin Drugs 0.000 description 5
- 238000002156 mixing Methods 0.000 description 5
- 239000003921 oil Substances 0.000 description 5
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 5
- 239000013641 positive control Substances 0.000 description 5
- 239000004540 pour-on Substances 0.000 description 5
- 238000007910 systemic administration Methods 0.000 description 5
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 4
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 4
- 241000283690 Bos taurus Species 0.000 description 4
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 4
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 4
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- 229920002125 Sokalan® Polymers 0.000 description 4
- 238000010521 absorption reaction Methods 0.000 description 4
- 230000000507 anthelmentic effect Effects 0.000 description 4
- 229940124339 anthelmintic agent Drugs 0.000 description 4
- 239000000921 anthelmintic agent Substances 0.000 description 4
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid group Chemical group C(C1=CC=CC=C1)(=O)O WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 4
- 239000001768 carboxy methyl cellulose Substances 0.000 description 4
- 239000002552 dosage form Substances 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- GCKZANITAMOIAR-XWVCPFKXSA-N dsstox_cid_14566 Chemical compound [O-]C(=O)C1=CC=CC=C1.C1=C[C@H](C)[C@@H]([C@@H](C)CC)O[C@]11O[C@H](C\C=C(C)\[C@@H](O[C@@H]2O[C@@H](C)[C@H](O[C@@H]3O[C@@H](C)[C@H]([NH2+]C)[C@@H](OC)C3)[C@@H](OC)C2)[C@@H](C)\C=C\C=C/2[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\2)O)C[C@H]4C1 GCKZANITAMOIAR-XWVCPFKXSA-N 0.000 description 4
- 235000013601 eggs Nutrition 0.000 description 4
- 239000007943 implant Substances 0.000 description 4
- 150000007524 organic acids Chemical class 0.000 description 4
- 239000002297 parasiticide Substances 0.000 description 4
- 239000008194 pharmaceutical composition Substances 0.000 description 4
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 4
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 239000000018 receptor agonist Substances 0.000 description 4
- 229940044601 receptor agonist Drugs 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- 239000003381 stabilizer Substances 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 239000004094 surface-active agent Substances 0.000 description 4
- 239000000725 suspension Substances 0.000 description 4
- 150000003626 triacylglycerols Chemical class 0.000 description 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- HIQIXEFWDLTDED-UHFFFAOYSA-N 4-hydroxy-1-piperidin-4-ylpyrrolidin-2-one Chemical compound O=C1CC(O)CN1C1CCNCC1 HIQIXEFWDLTDED-UHFFFAOYSA-N 0.000 description 3
- FJKROLUGYXJWQN-UHFFFAOYSA-N 4-hydroxybenzoic acid Chemical compound OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 3
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical compound OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 3
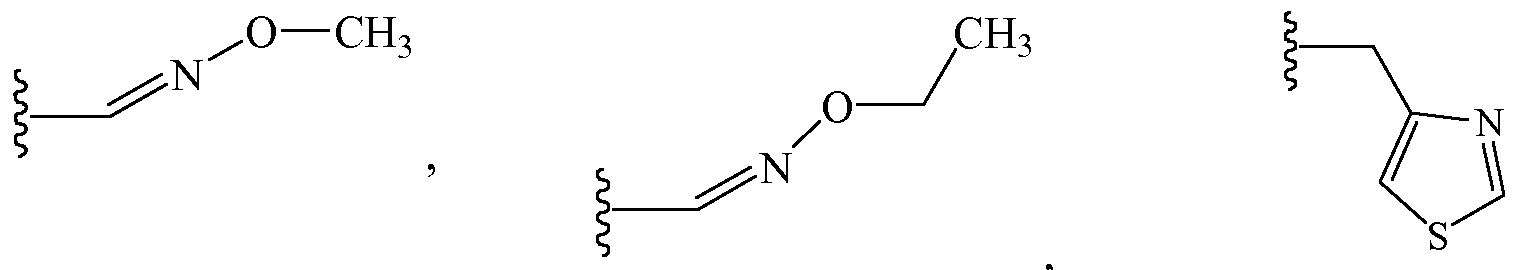
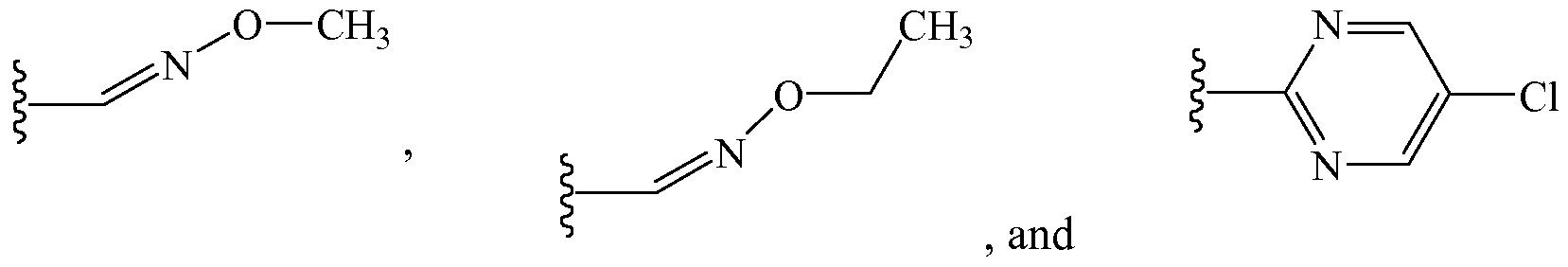
- URSPAOPYZOAFJO-WAYWQWQTSA-N CCO/N=C\C=C Chemical compound CCO/N=C\C=C URSPAOPYZOAFJO-WAYWQWQTSA-N 0.000 description 3
- CGOVTZGCKPMLMN-UHFFFAOYSA-N CCc1c[s]cn1 Chemical compound CCc1c[s]cn1 CGOVTZGCKPMLMN-UHFFFAOYSA-N 0.000 description 3
- 241001327638 Cimex lectularius Species 0.000 description 3
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 3
- 241000255925 Diptera Species 0.000 description 3
- 239000005894 Emamectin Substances 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 241000282324 Felis Species 0.000 description 3
- 241000238631 Hexapoda Species 0.000 description 3
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 3
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 3
- 229930195725 Mannitol Natural products 0.000 description 3
- 241001674048 Phthiraptera Species 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 3
- 241000607479 Yersinia pestis Species 0.000 description 3
- 239000000642 acaricide Substances 0.000 description 3
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- 125000001931 aliphatic group Chemical group 0.000 description 3
- 229940121363 anti-inflammatory agent Drugs 0.000 description 3
- 239000002260 anti-inflammatory agent Substances 0.000 description 3
- 230000008901 benefit Effects 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 210000001124 body fluid Anatomy 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical class OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 238000013270 controlled release Methods 0.000 description 3
- 239000006071 cream Substances 0.000 description 3
- 239000007884 disintegrant Substances 0.000 description 3
- 239000003623 enhancer Substances 0.000 description 3
- 229940116333 ethyl lactate Drugs 0.000 description 3
- 150000004665 fatty acids Chemical class 0.000 description 3
- 150000002191 fatty alcohols Chemical class 0.000 description 3
- NYPJDWWKZLNGGM-UHFFFAOYSA-N fenvalerate Aalpha Natural products C=1C=C(Cl)C=CC=1C(C(C)C)C(=O)OC(C#N)C(C=1)=CC=CC=1OC1=CC=CC=C1 NYPJDWWKZLNGGM-UHFFFAOYSA-N 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 239000008187 granular material Substances 0.000 description 3
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 3
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 3
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 3
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 3
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 3
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 3
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 3
- 229940071676 hydroxypropylcellulose Drugs 0.000 description 3
- 239000004615 ingredient Substances 0.000 description 3
- 230000000749 insecticidal effect Effects 0.000 description 3
- 239000002917 insecticide Substances 0.000 description 3
- 238000002955 isolation Methods 0.000 description 3
- 230000000366 juvenile effect Effects 0.000 description 3
- 239000002949 juvenile hormone Substances 0.000 description 3
- 150000002596 lactones Chemical class 0.000 description 3
- 239000000314 lubricant Substances 0.000 description 3
- 235000010355 mannitol Nutrition 0.000 description 3
- 239000000594 mannitol Substances 0.000 description 3
- 229920000609 methyl cellulose Polymers 0.000 description 3
- 239000001923 methylcellulose Substances 0.000 description 3
- 235000010981 methylcellulose Nutrition 0.000 description 3
- FXWHFKOXMBTCMP-WMEDONTMSA-N milbemycin Natural products COC1C2OCC3=C/C=C/C(C)CC(=CCC4CC(CC5(O4)OC(C)C(C)C(OC(=O)C(C)CC(C)C)C5O)OC(=O)C(C=C1C)C23O)C FXWHFKOXMBTCMP-WMEDONTMSA-N 0.000 description 3
- 201000002266 mite infestation Diseases 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 3
- 235000019198 oils Nutrition 0.000 description 3
- 235000005985 organic acids Nutrition 0.000 description 3
- 229940069328 povidone Drugs 0.000 description 3
- 239000003755 preservative agent Substances 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 229910052708 sodium Inorganic materials 0.000 description 3
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 3
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 3
- 241000894007 species Species 0.000 description 3
- 230000009885 systemic effect Effects 0.000 description 3
- 239000003826 tablet Substances 0.000 description 3
- 238000011200 topical administration Methods 0.000 description 3
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 2
- NFGXHKASABOEEW-UHFFFAOYSA-N 1-methylethyl 11-methoxy-3,7,11-trimethyl-2,4-dodecadienoate Chemical compound COC(C)(C)CCCC(C)CC=CC(C)=CC(=O)OC(C)C NFGXHKASABOEEW-UHFFFAOYSA-N 0.000 description 2
- LZILFXHGPBJADI-UHFFFAOYSA-N 2-(2-hydroxypropoxy)propan-1-ol;nonanoic acid Chemical compound CC(O)COC(C)CO.CCCCCCCCC(O)=O LZILFXHGPBJADI-UHFFFAOYSA-N 0.000 description 2
- WOXFMYVTSLAQMO-UHFFFAOYSA-N 2-Pyridinemethanamine Chemical class NCC1=CC=CC=N1 WOXFMYVTSLAQMO-UHFFFAOYSA-N 0.000 description 2
- ZDOOQPFIGYHZFV-UHFFFAOYSA-N 2-ethyl-4-[(4-phenoxyphenoxy)methyl]-1,3-dioxolane Chemical compound O1C(CC)OCC1COC(C=C1)=CC=C1OC1=CC=CC=C1 ZDOOQPFIGYHZFV-UHFFFAOYSA-N 0.000 description 2
- WRMNZCZEMHIOCP-UHFFFAOYSA-N 2-phenylethanol Chemical compound OCCC1=CC=CC=C1 WRMNZCZEMHIOCP-UHFFFAOYSA-N 0.000 description 2
- 229940090248 4-hydroxybenzoic acid Drugs 0.000 description 2
- IBSREHMXUMOFBB-JFUDTMANSA-N 5u8924t11h Chemical compound O1[C@@H](C)[C@H](O)[C@@H](OC)C[C@@H]1O[C@@H]1[C@@H](OC)C[C@H](O[C@@H]2C(=C/C[C@@H]3C[C@@H](C[C@@]4(O3)C=C[C@H](C)[C@@H](C(C)C)O4)OC(=O)[C@@H]3C=C(C)[C@@H](O)[C@H]4OC\C([C@@]34O)=C/C=C/[C@@H]2C)/C)O[C@H]1C.C1=C[C@H](C)[C@@H]([C@@H](C)CC)O[C@]11O[C@H](C\C=C(C)\[C@@H](O[C@@H]2O[C@@H](C)[C@H](O[C@@H]3O[C@@H](C)[C@H](O)[C@@H](OC)C3)[C@@H](OC)C2)[C@@H](C)\C=C\C=C/2[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\2)O)C[C@H]4C1 IBSREHMXUMOFBB-JFUDTMANSA-N 0.000 description 2
- 241000272517 Anseriformes Species 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 241000416162 Astragalus gummifer Species 0.000 description 2
- 239000005711 Benzoic acid Substances 0.000 description 2
- 239000005885 Buprofezin Substances 0.000 description 2
- YTJDRDYXXWKGGH-UHFFFAOYSA-N CCOCN(C(c(c(I)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O)C(OC)=O Chemical compound CCOCN(C(c(c(I)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O)C(OC)=O YTJDRDYXXWKGGH-UHFFFAOYSA-N 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- 241000282465 Canis Species 0.000 description 2
- 241000700199 Cavia porcellus Species 0.000 description 2
- 241000202814 Cochliomyia hominivorax Species 0.000 description 2
- 241000490513 Ctenocephalides canis Species 0.000 description 2
- 241000692095 Cuterebra Species 0.000 description 2
- 239000005891 Cyromazine Substances 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- 241001481695 Dermanyssus gallinae Species 0.000 description 2
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 239000005893 Diflubenzuron Substances 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- 239000005898 Fenoxycarb Substances 0.000 description 2
- 241000257324 Glossina <genus> Species 0.000 description 2
- 229920002907 Guar gum Polymers 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 241000257176 Hypoderma <fly> Species 0.000 description 2
- 241001480843 Ixodes ricinus Species 0.000 description 2
- 240000007472 Leucaena leucocephala Species 0.000 description 2
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 2
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 2
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- 241000819999 Nymphes Species 0.000 description 2
- 241000238890 Ornithodoros moubata Species 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- 241000718000 Pulex irritans Species 0.000 description 2
- 239000005927 Pyriproxyfen Substances 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- 229920002472 Starch Polymers 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 241000255628 Tabanidae Species 0.000 description 2
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 2
- 239000005937 Tebufenozide Substances 0.000 description 2
- 229920001615 Tragacanth Polymers 0.000 description 2
- 241001210412 Triatominae Species 0.000 description 2
- 239000005942 Triflumuron Substances 0.000 description 2
- 241000331598 Trombiculidae Species 0.000 description 2
- 241000353223 Xenopsylla cheopis Species 0.000 description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 2
- 229950008167 abamectin Drugs 0.000 description 2
- 150000005215 alkyl ethers Chemical class 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- SNAAJJQQZSMGQD-UHFFFAOYSA-N aluminum magnesium Chemical compound [Mg].[Al] SNAAJJQQZSMGQD-UHFFFAOYSA-N 0.000 description 2
- RWZYAGGXGHYGMB-UHFFFAOYSA-N anthranilic acid Chemical compound NC1=CC=CC=C1C(O)=O RWZYAGGXGHYGMB-UHFFFAOYSA-N 0.000 description 2
- 230000003110 anti-inflammatory effect Effects 0.000 description 2
- 229940125715 antihistaminic agent Drugs 0.000 description 2
- 239000000739 antihistaminic agent Substances 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 235000010233 benzoic acid Nutrition 0.000 description 2
- SESFRYSPDFLNCH-UHFFFAOYSA-N benzyl benzoate Chemical compound C=1C=CC=CC=1C(=O)OCC1=CC=CC=C1 SESFRYSPDFLNCH-UHFFFAOYSA-N 0.000 description 2
- 239000010839 body fluid Substances 0.000 description 2
- 150000001649 bromium compounds Chemical class 0.000 description 2
- PRLVTUNWOQKEAI-VKAVYKQESA-N buprofezin Chemical compound O=C1N(C(C)C)\C(=N\C(C)(C)C)SCN1C1=CC=CC=C1 PRLVTUNWOQKEAI-VKAVYKQESA-N 0.000 description 2
- 235000019437 butane-1,3-diol Nutrition 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 229910000019 calcium carbonate Inorganic materials 0.000 description 2
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 2
- 229960001631 carbomer Drugs 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- UISUNVFOGSJSKD-UHFFFAOYSA-N chlorfluazuron Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC(C=C1Cl)=CC(Cl)=C1OC1=NC=C(C(F)(F)F)C=C1Cl UISUNVFOGSJSKD-UHFFFAOYSA-N 0.000 description 2
- 229960004926 chlorobutanol Drugs 0.000 description 2
- 239000002285 corn oil Substances 0.000 description 2
- 235000005687 corn oil Nutrition 0.000 description 2
- LVQDKIWDGQRHTE-UHFFFAOYSA-N cyromazine Chemical compound NC1=NC(N)=NC(NC2CC2)=N1 LVQDKIWDGQRHTE-UHFFFAOYSA-N 0.000 description 2
- 229950000775 cyromazine Drugs 0.000 description 2
- SASYSVUEVMOWPL-NXVVXOECSA-N decyl oleate Chemical compound CCCCCCCCCCOC(=O)CCCCCCC\C=C/CCCCCCCC SASYSVUEVMOWPL-NXVVXOECSA-N 0.000 description 2
- 230000002939 deleterious effect Effects 0.000 description 2
- DOIRQSBPFJWKBE-UHFFFAOYSA-N dibutyl phthalate Chemical compound CCCCOC(=O)C1=CC=CC=C1C(=O)OCCCC DOIRQSBPFJWKBE-UHFFFAOYSA-N 0.000 description 2
- 235000005911 diet Nutrition 0.000 description 2
- 230000037213 diet Effects 0.000 description 2
- QQQYTWIFVNKMRW-UHFFFAOYSA-N diflubenzuron Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC1=CC=C(Cl)C=C1 QQQYTWIFVNKMRW-UHFFFAOYSA-N 0.000 description 2
- 229940019503 diflubenzuron Drugs 0.000 description 2
- 229940113088 dimethylacetamide Drugs 0.000 description 2
- 239000002270 dispersing agent Substances 0.000 description 2
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 2
- 239000003651 drinking water Substances 0.000 description 2
- 235000020188 drinking water Nutrition 0.000 description 2
- 239000000975 dye Substances 0.000 description 2
- 230000001804 emulsifying effect Effects 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- HJUFTIJOISQSKQ-UHFFFAOYSA-N fenoxycarb Chemical compound C1=CC(OCCNC(=O)OCC)=CC=C1OC1=CC=CC=C1 HJUFTIJOISQSKQ-UHFFFAOYSA-N 0.000 description 2
- 238000000855 fermentation Methods 0.000 description 2
- 230000004151 fermentation Effects 0.000 description 2
- 239000000945 filler Substances 0.000 description 2
- RYLHNOVXKPXDIP-UHFFFAOYSA-N flufenoxuron Chemical compound C=1C=C(NC(=O)NC(=O)C=2C(=CC=CC=2F)F)C(F)=CC=1OC1=CC=C(C(F)(F)F)C=C1Cl RYLHNOVXKPXDIP-UHFFFAOYSA-N 0.000 description 2
- 235000003599 food sweetener Nutrition 0.000 description 2
- ZXQYGBMAQZUVMI-GCMPRSNUSA-N gamma-cyhalothrin Chemical compound CC1(C)[C@@H](\C=C(/Cl)C(F)(F)F)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 ZXQYGBMAQZUVMI-GCMPRSNUSA-N 0.000 description 2
- 150000002334 glycols Chemical class 0.000 description 2
- 235000010417 guar gum Nutrition 0.000 description 2
- 239000000665 guar gum Substances 0.000 description 2
- 229960002154 guar gum Drugs 0.000 description 2
- RGNPBRKPHBKNKX-UHFFFAOYSA-N hexaflumuron Chemical compound C1=C(Cl)C(OC(F)(F)C(F)F)=C(Cl)C=C1NC(=O)NC(=O)C1=C(F)C=CC=C1F RGNPBRKPHBKNKX-UHFFFAOYSA-N 0.000 description 2
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid group Chemical group C(CCCCC)(=O)O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 150000007529 inorganic bases Chemical class 0.000 description 2
- 125000003971 isoxazolinyl group Chemical group 0.000 description 2
- 230000002147 killing effect Effects 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- 229910052744 lithium Inorganic materials 0.000 description 2
- 244000144972 livestock Species 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 229930002897 methoprene Natural products 0.000 description 2
- 229950003442 methoprene Drugs 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 239000004530 micro-emulsion Substances 0.000 description 2
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 2
- 239000008108 microcrystalline cellulose Substances 0.000 description 2
- 229940016286 microcrystalline cellulose Drugs 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- KRTSDMXIXPKRQR-AATRIKPKSA-N monocrotophos Chemical compound CNC(=O)\C=C(/C)OP(=O)(OC)OC KRTSDMXIXPKRQR-AATRIKPKSA-N 0.000 description 2
- YZBLFMPOMVTDJY-CBYMMZEQSA-N moxidectin Chemical compound O1[C@H](C(\C)=C\C(C)C)[C@@H](C)C(=N/OC)\C[C@@]11O[C@H](C\C=C(C)\C[C@@H](C)\C=C\C=C/2[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\2)O)C[C@H]4C1 YZBLFMPOMVTDJY-CBYMMZEQSA-N 0.000 description 2
- 229960004816 moxidectin Drugs 0.000 description 2
- 239000000041 non-steroidal anti-inflammatory agent Substances 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- BARWIPMJPCRCTP-UHFFFAOYSA-N oleic acid oleyl ester Natural products CCCCCCCCC=CCCCCCCCCOC(=O)CCCCCCCC=CCCCCCCCC BARWIPMJPCRCTP-UHFFFAOYSA-N 0.000 description 2
- BARWIPMJPCRCTP-CLFAGFIQSA-N oleyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCCOC(=O)CCCCCCC\C=C/CCCCCCCC BARWIPMJPCRCTP-CLFAGFIQSA-N 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 239000006186 oral dosage form Substances 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- 230000001717 pathogenic effect Effects 0.000 description 2
- 230000001575 pathological effect Effects 0.000 description 2
- 239000000575 pesticide Substances 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- 239000012071 phase Substances 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 239000000049 pigment Substances 0.000 description 2
- 239000006187 pill Substances 0.000 description 2
- 230000036470 plasma concentration Effects 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 235000013772 propylene glycol Nutrition 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 239000003223 protective agent Substances 0.000 description 2
- 239000008213 purified water Substances 0.000 description 2
- NHDHVHZZCFYRSB-UHFFFAOYSA-N pyriproxyfen Chemical compound C=1C=CC=NC=1OC(C)COC(C=C1)=CC=C1OC1=CC=CC=C1 NHDHVHZZCFYRSB-UHFFFAOYSA-N 0.000 description 2
- HNJBEVLQSNELDL-UHFFFAOYSA-N pyrrolidin-2-one Chemical compound O=C1CCCN1 HNJBEVLQSNELDL-UHFFFAOYSA-N 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 229920002545 silicone oil Polymers 0.000 description 2
- 239000007909 solid dosage form Substances 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 229940032147 starch Drugs 0.000 description 2
- 235000019698 starch Nutrition 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 239000003765 sweetening agent Substances 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 239000011975 tartaric acid Substances 0.000 description 2
- 235000002906 tartaric acid Nutrition 0.000 description 2
- QYPNKSZPJQQLRK-UHFFFAOYSA-N tebufenozide Chemical compound C1=CC(CC)=CC=C1C(=O)NN(C(C)(C)C)C(=O)C1=CC(C)=CC(C)=C1 QYPNKSZPJQQLRK-UHFFFAOYSA-N 0.000 description 2
- XLNZEKHULJKQBA-UHFFFAOYSA-N terbufos Chemical compound CCOP(=S)(OCC)SCSC(C)(C)C XLNZEKHULJKQBA-UHFFFAOYSA-N 0.000 description 2
- 239000012049 topical pharmaceutical composition Substances 0.000 description 2
- 231100000027 toxicology Toxicity 0.000 description 2
- 235000010487 tragacanth Nutrition 0.000 description 2
- 239000000196 tragacanth Substances 0.000 description 2
- 229940116362 tragacanth Drugs 0.000 description 2
- XAIPTRIXGHTTNT-UHFFFAOYSA-N triflumuron Chemical compound C1=CC(OC(F)(F)F)=CC=C1NC(=O)NC(=O)C1=CC=CC=C1Cl XAIPTRIXGHTTNT-UHFFFAOYSA-N 0.000 description 2
- ZDPHROOEEOARMN-UHFFFAOYSA-N undecanoic acid Chemical compound CCCCCCCCCCC(O)=O ZDPHROOEEOARMN-UHFFFAOYSA-N 0.000 description 2
- 239000013598 vector Substances 0.000 description 2
- 235000015112 vegetable and seed oil Nutrition 0.000 description 2
- 239000008158 vegetable oil Substances 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- 235000010493 xanthan gum Nutrition 0.000 description 2
- 239000000230 xanthan gum Substances 0.000 description 2
- 229920001285 xanthan gum Polymers 0.000 description 2
- 229940082509 xanthan gum Drugs 0.000 description 2
- 229910052725 zinc Inorganic materials 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- AGMMRUPNXPWLGF-AATRIKPKSA-N (2,3,5,6-tetrafluoro-4-methylphenyl)methyl 2,2-dimethyl-3-[(e)-prop-1-enyl]cyclopropane-1-carboxylate Chemical compound CC1(C)C(/C=C/C)C1C(=O)OCC1=C(F)C(F)=C(C)C(F)=C1F AGMMRUPNXPWLGF-AATRIKPKSA-N 0.000 description 1
- LZTIMERBDGGAJD-SNAWJCMRSA-N (2e)-2-(nitromethylidene)-1,3-thiazinane Chemical compound [O-][N+](=O)\C=C1/NCCCS1 LZTIMERBDGGAJD-SNAWJCMRSA-N 0.000 description 1
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Polymers OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 1
- MCKJPJYRCPANCC-XLXYOEISSA-N (8s,9s,10r,11s,13s,14s,17r)-11,17-dihydroxy-10,13-dimethyl-3-oxo-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthrene-17-carboxylic acid Chemical compound O=C1C=C[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(O)=O)[C@@H]4[C@@H]3CCC2=C1 MCKJPJYRCPANCC-XLXYOEISSA-N 0.000 description 1
- ALSTYHKOOCGGFT-KTKRTIGZSA-N (9Z)-octadecen-1-ol Chemical compound CCCCCCCC\C=C/CCCCCCCCO ALSTYHKOOCGGFT-KTKRTIGZSA-N 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- WCXDHFDTOYPNIE-RIYZIHGNSA-N (E)-acetamiprid Chemical compound N#C/N=C(\C)N(C)CC1=CC=C(Cl)N=C1 WCXDHFDTOYPNIE-RIYZIHGNSA-N 0.000 description 1
- PGOOBECODWQEAB-UHFFFAOYSA-N (E)-clothianidin Chemical compound [O-][N+](=O)\N=C(/NC)NCC1=CN=C(Cl)S1 PGOOBECODWQEAB-UHFFFAOYSA-N 0.000 description 1
- CFRPSFYHXJZSBI-DHZHZOJOSA-N (E)-nitenpyram Chemical compound [O-][N+](=O)/C=C(\NC)N(CC)CC1=CC=C(Cl)N=C1 CFRPSFYHXJZSBI-DHZHZOJOSA-N 0.000 description 1
- FZRBKIRIBLNOAM-UHFFFAOYSA-N (E,E)-2-propynyl 3,7,11-trimethyl-2,4-dodecadienoate Chemical compound CC(C)CCCC(C)CC=CC(C)=CC(=O)OCC#C FZRBKIRIBLNOAM-UHFFFAOYSA-N 0.000 description 1
- IQVNEKKDSLOHHK-FNCQTZNRSA-N (E,E)-hydramethylnon Chemical compound N1CC(C)(C)CNC1=NN=C(/C=C/C=1C=CC(=CC=1)C(F)(F)F)\C=C\C1=CC=C(C(F)(F)F)C=C1 IQVNEKKDSLOHHK-FNCQTZNRSA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UWTATZPHSA-M (R)-lactate Chemical compound C[C@@H](O)C([O-])=O JVTAAEKCZFNVCJ-UWTATZPHSA-M 0.000 description 1
- ZFHGXWPMULPQSE-SZGBIDFHSA-N (Z)-(1S)-cis-tefluthrin Chemical compound FC1=C(F)C(C)=C(F)C(F)=C1COC(=O)[C@@H]1C(C)(C)[C@@H]1\C=C(/Cl)C(F)(F)F ZFHGXWPMULPQSE-SZGBIDFHSA-N 0.000 description 1
- HOKKPVIRMVDYPB-UVTDQMKNSA-N (Z)-thiacloprid Chemical compound C1=NC(Cl)=CC=C1CN1C(=N/C#N)/SCC1 HOKKPVIRMVDYPB-UVTDQMKNSA-N 0.000 description 1
- IAKOZHOLGAGEJT-UHFFFAOYSA-N 1,1,1-trichloro-2,2-bis(p-methoxyphenyl)-Ethane Chemical compound C1=CC(OC)=CC=C1C(C(Cl)(Cl)Cl)C1=CC=C(OC)C=C1 IAKOZHOLGAGEJT-UHFFFAOYSA-N 0.000 description 1
- JCIIKRHCWVHVFF-UHFFFAOYSA-N 1,2,4-thiadiazol-5-amine;hydrochloride Chemical compound Cl.NC1=NC=NS1 JCIIKRHCWVHVFF-UHFFFAOYSA-N 0.000 description 1
- VFWCMGCRMGJXDK-UHFFFAOYSA-N 1-chlorobutane Chemical class CCCCCl VFWCMGCRMGJXDK-UHFFFAOYSA-N 0.000 description 1
- VUQPJRPDRDVQMN-UHFFFAOYSA-N 1-chlorooctadecane Chemical class CCCCCCCCCCCCCCCCCCCl VUQPJRPDRDVQMN-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- JNYAEWCLZODPBN-UHFFFAOYSA-N 2-(1,2-dihydroxyethyl)oxolane-3,4-diol Polymers OCC(O)C1OCC(O)C1O JNYAEWCLZODPBN-UHFFFAOYSA-N 0.000 description 1
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 1
- FSVJFNAIGNNGKK-UHFFFAOYSA-N 2-[cyclohexyl(oxo)methyl]-3,6,7,11b-tetrahydro-1H-pyrazino[2,1-a]isoquinolin-4-one Chemical compound C1C(C2=CC=CC=C2CC2)N2C(=O)CN1C(=O)C1CCCCC1 FSVJFNAIGNNGKK-UHFFFAOYSA-N 0.000 description 1
- AWSZRJQNBMEZOI-UHFFFAOYSA-N 2-methoxyethyl 2-(4-tert-butylphenyl)-2-cyano-3-oxo-3-[2-(trifluoromethyl)phenyl]propanoate Chemical compound C=1C=C(C(C)(C)C)C=CC=1C(C#N)(C(=O)OCCOC)C(=O)C1=CC=CC=C1C(F)(F)F AWSZRJQNBMEZOI-UHFFFAOYSA-N 0.000 description 1
- LEACJMVNYZDSKR-UHFFFAOYSA-N 2-octyldodecan-1-ol Chemical compound CCCCCCCCCCC(CO)CCCCCCCC LEACJMVNYZDSKR-UHFFFAOYSA-N 0.000 description 1
- QCDWFXQBSFUVSP-UHFFFAOYSA-N 2-phenoxyethanol Chemical compound OCCOC1=CC=CC=C1 QCDWFXQBSFUVSP-UHFFFAOYSA-N 0.000 description 1
- YUSXTIMBFYGITE-UHFFFAOYSA-N 2-phenyl-3-(1h-pyrrol-2-yl)prop-2-enenitrile Chemical class C=1C=CC=CC=1C(C#N)=CC1=CC=CN1 YUSXTIMBFYGITE-UHFFFAOYSA-N 0.000 description 1
- WMPPDTMATNBGJN-UHFFFAOYSA-N 2-phenylethylbromide Chemical compound BrCCC1=CC=CC=C1 WMPPDTMATNBGJN-UHFFFAOYSA-N 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- UMZCLZPXPCNKML-UHFFFAOYSA-N 2h-imidazo[4,5-d][1,3]thiazole Chemical class C1=NC2=NCSC2=N1 UMZCLZPXPCNKML-UHFFFAOYSA-N 0.000 description 1
- QCAHUFWKIQLBNB-UHFFFAOYSA-N 3-(3-methoxypropoxy)propan-1-ol Chemical compound COCCCOCCCO QCAHUFWKIQLBNB-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- XMIIGOLPHOKFCH-UHFFFAOYSA-N 3-phenylpropionic acid Chemical compound OC(=O)CCC1=CC=CC=C1 XMIIGOLPHOKFCH-UHFFFAOYSA-N 0.000 description 1
- XKTYXVDYIKIYJP-UHFFFAOYSA-N 3h-dioxole Chemical compound C1OOC=C1 XKTYXVDYIKIYJP-UHFFFAOYSA-N 0.000 description 1
- LABQLTFAPITERI-UHFFFAOYSA-N 4-(1-but-2-ynoxyethyl)-1,2-dimethoxybenzene Chemical compound COC1=CC=C(C(C)OCC#CC)C=C1OC LABQLTFAPITERI-UHFFFAOYSA-N 0.000 description 1
- QDFVXXBCJYNKKC-UHFFFAOYSA-N 4-[4-(4-chlorophenyl)-4-cyclopropylbutyl]-1-fluoro-2-phenoxybenzene Chemical compound C1=C(OC=2C=CC=CC=2)C(F)=CC=C1CCCC(C=1C=CC(Cl)=CC=1)C1CC1 QDFVXXBCJYNKKC-UHFFFAOYSA-N 0.000 description 1
- XJFIKRXIJXAJGH-UHFFFAOYSA-N 5-chloro-1,3-dihydroimidazo[4,5-b]pyridin-2-one Chemical group ClC1=CC=C2NC(=O)NC2=N1 XJFIKRXIJXAJGH-UHFFFAOYSA-N 0.000 description 1
- NQPDXQQQCQDHHW-UHFFFAOYSA-N 6-chloro-5-(2,3-dichlorophenoxy)-2-(methylthio)-1H-benzimidazole Chemical compound ClC=1C=C2NC(SC)=NC2=CC=1OC1=CC=CC(Cl)=C1Cl NQPDXQQQCQDHHW-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 208000004998 Abdominal Pain Diseases 0.000 description 1
- 239000005875 Acetamiprid Substances 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- 241000256111 Aedes <genus> Species 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 241000238682 Amblyomma americanum Species 0.000 description 1
- 241000256186 Anopheles <genus> Species 0.000 description 1
- 241000232553 Archaeopsylla erinacei Species 0.000 description 1
- 241000238888 Argasidae Species 0.000 description 1
- 241000238421 Arthropoda Species 0.000 description 1
- 241000972773 Aulopiformes Species 0.000 description 1
- 241000271566 Aves Species 0.000 description 1
- 239000005878 Azadirachtin Substances 0.000 description 1
- GOPGLVITNIXEFO-UHFFFAOYSA-N Bc1cnc(N(C(C)=O)C(c(cc2)c(C)cc2C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)=O)nc1 Chemical compound Bc1cnc(N(C(C)=O)C(c(cc2)c(C)cc2C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)=O)nc1 GOPGLVITNIXEFO-UHFFFAOYSA-N 0.000 description 1
- KUVIULQEHSCUHY-XYWKZLDCSA-N Beclometasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(Cl)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)COC(=O)CC)(OC(=O)CC)[C@@]1(C)C[C@@H]2O KUVIULQEHSCUHY-XYWKZLDCSA-N 0.000 description 1
- 239000005884 Beta-Cyfluthrin Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 239000005653 Bifenazate Substances 0.000 description 1
- 239000005874 Bifenthrin Substances 0.000 description 1
- 241000322475 Bovicola Species 0.000 description 1
- 241000195940 Bryophyta Species 0.000 description 1
- VOVIALXJUBGFJZ-KWVAZRHASA-N Budesonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@@H]2[C@@H]1[C@@H]1C[C@H]3OC(CCC)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O VOVIALXJUBGFJZ-KWVAZRHASA-N 0.000 description 1
- 239000004358 Butane-1, 3-diol Substances 0.000 description 1
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 1
- FIPWRIJSWJWJAI-UHFFFAOYSA-N Butyl carbitol 6-propylpiperonyl ether Chemical compound C1=C(CCC)C(COCCOCCOCCCC)=CC2=C1OCO2 FIPWRIJSWJWJAI-UHFFFAOYSA-N 0.000 description 1
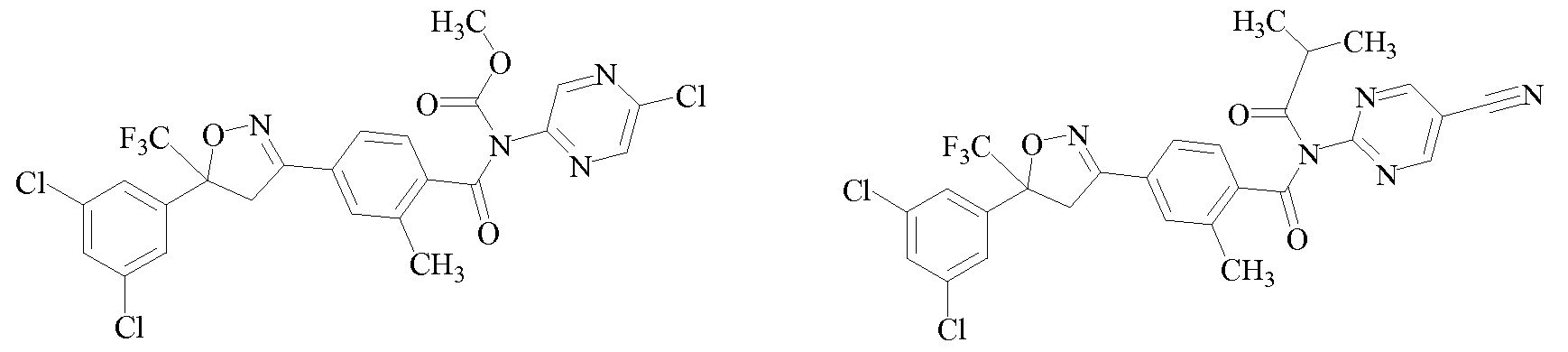
- VTPKOQVDDDEZKA-UHFFFAOYSA-N CC(C(NCC(F)(F)F)=O)NC(c(cc1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(C(F)(F)F)cc(C(F)(F)F)c1)=O Chemical compound CC(C(NCC(F)(F)F)=O)NC(c(cc1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(C(F)(F)F)cc(C(F)(F)F)c1)=O VTPKOQVDDDEZKA-UHFFFAOYSA-N 0.000 description 1
- XFRJYXYXKNEUMW-UHFFFAOYSA-N CC(C)C(N(C(c(c(C)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O)C(N(C1)CC1=C1)=NC=C1Cl)=O Chemical compound CC(C)C(N(C(c(c(C)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O)C(N(C1)CC1=C1)=NC=C1Cl)=O XFRJYXYXKNEUMW-UHFFFAOYSA-N 0.000 description 1
- SQVQOCWOZHALQO-UHFFFAOYSA-N CC(N(C(c(c(C)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O)N(C)c1ccccc1)=O Chemical compound CC(N(C(c(c(C)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O)N(C)c1ccccc1)=O SQVQOCWOZHALQO-UHFFFAOYSA-N 0.000 description 1
- IUPIFBIPFCUTQC-YCGHPLDRSA-N CC/C=C(\C=C/C(C(N(CC)c(nc1)ncc1Cl)=O)=C)/C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1 Chemical compound CC/C=C(\C=C/C(C(N(CC)c(nc1)ncc1Cl)=O)=C)/C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1 IUPIFBIPFCUTQC-YCGHPLDRSA-N 0.000 description 1
- DCLKAVVTKLCBRD-UHFFFAOYSA-O CC=C([NH3+])OC=[IH] Chemical compound CC=C([NH3+])OC=[IH] DCLKAVVTKLCBRD-UHFFFAOYSA-O 0.000 description 1
- CDPRHCUJRDHERO-UHFFFAOYSA-N CCO/N=C\NC(c(c(C)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O Chemical compound CCO/N=C\NC(c(c(C)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O CDPRHCUJRDHERO-UHFFFAOYSA-N 0.000 description 1
- WYIQMHNHYCCQIR-UHFFFAOYSA-N CCOCN(C(c(c(C)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O)C(OC)=O Chemical compound CCOCN(C(c(c(C)c1)ccc1C(C1)=NOC1(C(F)(F)F)c1cc(Cl)cc(Cl)c1)=O)C(OC)=O WYIQMHNHYCCQIR-UHFFFAOYSA-N 0.000 description 1
- ULRXVLGIEWVHEW-UHFFFAOYSA-N CCOCNC(c(cc1)ccc1C(C1)=NOC1(C(F)(F)F)c(cc1Cl)cc(Cl)c1Cl)=O Chemical compound CCOCNC(c(cc1)ccc1C(C1)=NOC1(C(F)(F)F)c(cc1Cl)cc(Cl)c1Cl)=O ULRXVLGIEWVHEW-UHFFFAOYSA-N 0.000 description 1
- JFLRKDZMHNBDQS-UCQUSYKYSA-N CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C(=C[C@H]3[C@@H]2CC(=O)O1)C)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C.CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C=C[C@H]3C2CC(=O)O1)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C Chemical compound CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C(=C[C@H]3[C@@H]2CC(=O)O1)C)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C.CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C=C[C@H]3C2CC(=O)O1)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C JFLRKDZMHNBDQS-UCQUSYKYSA-N 0.000 description 1
- 241000257161 Calliphoridae Species 0.000 description 1
- 241000282421 Canidae Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- UISREOKYJBBYSC-UHFFFAOYSA-N Cc(nc1)ncc1Cl Chemical compound Cc(nc1)ncc1Cl UISREOKYJBBYSC-UHFFFAOYSA-N 0.000 description 1
- YCEAGLOJPQNEEB-UHFFFAOYSA-N Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(C(F)(F)F)cc(C(F)(F)F)c2)ccc1C(N/C=N\OC)=O Chemical compound Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(C(F)(F)F)cc(C(F)(F)F)c2)ccc1C(N/C=N\OC)=O YCEAGLOJPQNEEB-UHFFFAOYSA-N 0.000 description 1
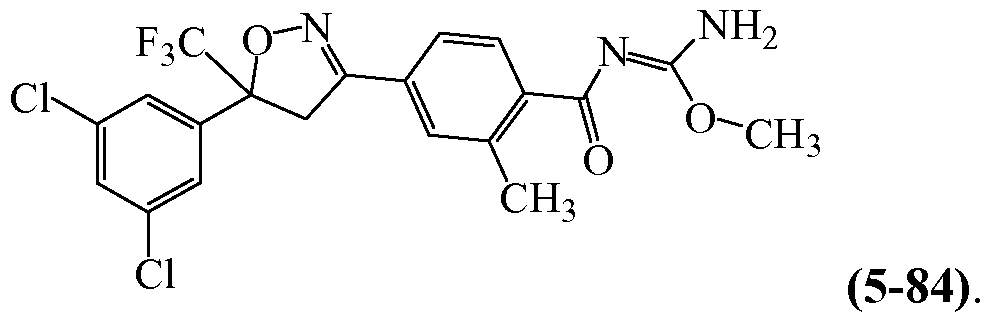
- GCURIORCWWFOGV-UHFFFAOYSA-N Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(/N=C(/N)\OC)=O Chemical compound Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(/N=C(/N)\OC)=O GCURIORCWWFOGV-UHFFFAOYSA-N 0.000 description 1
- SNDCYSAMOSJDPF-UHFFFAOYSA-N Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(N(C(C1CC1)=O)C(N(C1)CC1=C1)=NC=C1Cl)=O Chemical compound Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(N(C(C1CC1)=O)C(N(C1)CC1=C1)=NC=C1Cl)=O SNDCYSAMOSJDPF-UHFFFAOYSA-N 0.000 description 1
- UDFLJGZEMMNXEH-UHFFFAOYSA-N Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(N(C(COC)=O)c(nc1)ncc1Cl)=O Chemical compound Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(N(C(COC)=O)c(nc1)ncc1Cl)=O UDFLJGZEMMNXEH-UHFFFAOYSA-N 0.000 description 1
- BPFUIWLQXNPZHI-UHFFFAOYSA-N Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(N/C=N\OC)=O Chemical compound Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(N/C=N\OC)=O BPFUIWLQXNPZHI-UHFFFAOYSA-N 0.000 description 1
- NNHSBYOJJNPYQT-UHFFFAOYSA-N Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(Nc1cncnc1)=O Chemical compound Cc1cc(C(C2)=NOC2(C(F)(F)F)c2cc(Cl)cc(Cl)c2)ccc1C(Nc1cncnc1)=O NNHSBYOJJNPYQT-UHFFFAOYSA-N 0.000 description 1
- RHOOLJLEYYXKTK-UHFFFAOYSA-N Cc1cnc(C)nc1 Chemical compound Cc1cnc(C)nc1 RHOOLJLEYYXKTK-UHFFFAOYSA-N 0.000 description 1
- TWGNOYAGHYUFFR-UHFFFAOYSA-N Cc1cncnc1 Chemical compound Cc1cncnc1 TWGNOYAGHYUFFR-UHFFFAOYSA-N 0.000 description 1
- 241001609899 Ceratophyllus gallinae Species 0.000 description 1
- 241000282994 Cervidae Species 0.000 description 1
- 241000700114 Chinchillidae Species 0.000 description 1
- 239000005886 Chlorantraniliprole Substances 0.000 description 1
- 239000005944 Chlorpyrifos Substances 0.000 description 1
- 239000005945 Chlorpyrifos-methyl Substances 0.000 description 1
- 241001307956 Chorioptes bovis Species 0.000 description 1
- 239000005887 Chromafenozide Substances 0.000 description 1
- 241001124179 Chrysops Species 0.000 description 1
- LUKZNWIVRBCLON-GXOBDPJESA-N Ciclesonide Chemical compound C1([C@H]2O[C@@]3([C@H](O2)C[C@@H]2[C@@]3(C[C@H](O)[C@@H]3[C@@]4(C)C=CC(=O)C=C4CC[C@H]32)C)C(=O)COC(=O)C(C)C)CCCCC1 LUKZNWIVRBCLON-GXOBDPJESA-N 0.000 description 1
- 239000005888 Clothianidin Substances 0.000 description 1
- 241000748698 Colias elis Species 0.000 description 1
- 208000002881 Colic Diseases 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 241000258922 Ctenocephalides Species 0.000 description 1
- 241000256054 Culex <genus> Species 0.000 description 1
- 239000005655 Cyflumetofen Substances 0.000 description 1
- 239000005946 Cypermethrin Substances 0.000 description 1
- 241000252233 Cyprinus carpio Species 0.000 description 1
- ZZZCUOFIHGPKAK-UHFFFAOYSA-N D-erythro-ascorbic acid Natural products OCC1OC(=O)C(O)=C1O ZZZCUOFIHGPKAK-UHFFFAOYSA-N 0.000 description 1
- 239000005892 Deltamethrin Substances 0.000 description 1
- 241001128004 Demodex Species 0.000 description 1
- 241001480824 Dermacentor Species 0.000 description 1
- 241001480793 Dermacentor variabilis Species 0.000 description 1
- XTJFFFGAUHQWII-UHFFFAOYSA-N Dibutyl adipate Chemical compound CCCCOC(=O)CCCCC(=O)OCCCC XTJFFFGAUHQWII-UHFFFAOYSA-N 0.000 description 1
- ZDQWESQEGGJUCH-UHFFFAOYSA-N Diisopropyl adipate Chemical compound CC(C)OC(=O)CCCCC(=O)OC(C)C ZDQWESQEGGJUCH-UHFFFAOYSA-N 0.000 description 1
- 239000005947 Dimethoate Substances 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 241000283073 Equus caballus Species 0.000 description 1
- 239000005895 Esfenvalerate Substances 0.000 description 1
- FNELVJVBIYMIMC-UHFFFAOYSA-N Ethiprole Chemical compound N1=C(C#N)C(S(=O)CC)=C(N)N1C1=C(Cl)C=C(C(F)(F)F)C=C1Cl FNELVJVBIYMIMC-UHFFFAOYSA-N 0.000 description 1
- 239000001856 Ethyl cellulose Substances 0.000 description 1
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 1
- HMIBKHHNXANVHR-UHFFFAOYSA-N Fenothiocarb Chemical compound CN(C)C(=O)SCCCCOC1=CC=CC=C1 HMIBKHHNXANVHR-UHFFFAOYSA-N 0.000 description 1
- 239000005900 Flonicamid Substances 0.000 description 1
- 239000005901 Flubendiamide Substances 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 239000005903 Gamma-cyhalothrin Substances 0.000 description 1
- 241001660203 Gasterophilus Species 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 244000060234 Gmelina philippensis Species 0.000 description 1
- 241001480796 Haemaphysalis Species 0.000 description 1
- 241000257224 Haematobia Species 0.000 description 1
- 241000257232 Haematobia irritans Species 0.000 description 1
- 241000790933 Haematopinus Species 0.000 description 1
- 241000258937 Hemiptera Species 0.000 description 1
- CMBYOWLFQAFZCP-UHFFFAOYSA-N Hexyl dodecanoate Chemical compound CCCCCCCCCCCC(=O)OCCCCCC CMBYOWLFQAFZCP-UHFFFAOYSA-N 0.000 description 1
- 241001608644 Hippoboscidae Species 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 239000005906 Imidacloprid Substances 0.000 description 1
- 239000005907 Indoxacarb Substances 0.000 description 1
- 241000238889 Ixodidae Species 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-Lysine Natural products NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- 235000019766 L-Lysine Nutrition 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 239000004166 Lanolin Substances 0.000 description 1
- 239000005639 Lauric acid Substances 0.000 description 1
- 241001232872 Leptopsyllidae Species 0.000 description 1
- HLFSDGLLUJUHTE-SNVBAGLBSA-N Levamisole Chemical compound C1([C@H]2CN3CCSC3=N2)=CC=CC=C1 HLFSDGLLUJUHTE-SNVBAGLBSA-N 0.000 description 1
- 241001113970 Linognathus Species 0.000 description 1
- 241000257162 Lucilia <blowfly> Species 0.000 description 1
- 239000005912 Lufenuron Substances 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- WLLGXSLBOPFWQV-UHFFFAOYSA-N MGK 264 Chemical compound C1=CC2CC1C1C2C(=O)N(CC(CC)CCCC)C1=O WLLGXSLBOPFWQV-UHFFFAOYSA-N 0.000 description 1
- 239000005949 Malathion Substances 0.000 description 1
- 241000771994 Melophagus ovinus Species 0.000 description 1
- 241000035436 Menopon Species 0.000 description 1
- 241000699696 Meriones Species 0.000 description 1
- 239000005914 Metaflumizone Substances 0.000 description 1
- MIFOMMKAVSCNKQ-HWIUFGAZSA-N Metaflumizone Chemical compound C1=CC(OC(F)(F)F)=CC=C1NC(=O)N\N=C(C=1C=C(C=CC=1)C(F)(F)F)\CC1=CC=C(C#N)C=C1 MIFOMMKAVSCNKQ-HWIUFGAZSA-N 0.000 description 1
- 239000005956 Metaldehyde Substances 0.000 description 1
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical class OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 1
- 239000005916 Methomyl Substances 0.000 description 1
- 239000005917 Methoxyfenozide Substances 0.000 description 1
- FQISKWAFAHGMGT-SGJOWKDISA-M Methylprednisolone sodium succinate Chemical compound [Na+].C([C@@]12C)=CC(=O)C=C1[C@@H](C)C[C@@H]1[C@@H]2[C@@H](O)C[C@]2(C)[C@@](O)(C(=O)COC(=O)CCC([O-])=O)CC[C@H]21 FQISKWAFAHGMGT-SGJOWKDISA-M 0.000 description 1
- 229920000881 Modified starch Polymers 0.000 description 1
- 241000257226 Muscidae Species 0.000 description 1
- 241000282339 Mustela Species 0.000 description 1
- 241001373736 Myocoptes musculinus Species 0.000 description 1
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-dimethylformamide Substances CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 1
- JMPFSEBWVLAJKM-UHFFFAOYSA-N N-{5-chloro-4-[(4-chlorophenyl)(cyano)methyl]-2-methylphenyl}-2-hydroxy-3,5-diiodobenzamide Chemical compound ClC=1C=C(NC(=O)C=2C(=C(I)C=C(I)C=2)O)C(C)=CC=1C(C#N)C1=CC=C(Cl)C=C1 JMPFSEBWVLAJKM-UHFFFAOYSA-N 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- 241000244206 Nematoda Species 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- SUZOKZNGTCKFNO-UHFFFAOYSA-N O=C(c(cc1)ccc1C(C1)=NOC1(C(F)(F)F)c(cc1Cl)cc(Cl)c1Cl)NCOCC(F)(F)F Chemical compound O=C(c(cc1)ccc1C(C1)=NOC1(C(F)(F)F)c(cc1Cl)cc(Cl)c1Cl)NCOCC(F)(F)F SUZOKZNGTCKFNO-UHFFFAOYSA-N 0.000 description 1
- 241000257191 Oestridae Species 0.000 description 1
- 241000543819 Oestrus ovis Species 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 241000238887 Ornithodoros Species 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 208000030852 Parasitic disease Diseases 0.000 description 1
- 235000019483 Peanut oil Nutrition 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 241000286209 Phasianidae Species 0.000 description 1
- 239000005921 Phosmet Substances 0.000 description 1
- 239000005923 Pirimicarb Substances 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 229920002565 Polyethylene Glycol 400 Polymers 0.000 description 1
- 229920001214 Polysorbate 60 Polymers 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Chemical class OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 208000003251 Pruritus Diseases 0.000 description 1
- 241000287530 Psittaciformes Species 0.000 description 1
- 241001649230 Psoroptes ovis Species 0.000 description 1
- 241001675082 Pulex Species 0.000 description 1
- 239000005925 Pymetrozine Substances 0.000 description 1
- VQXSOUPNOZTNAI-UHFFFAOYSA-N Pyrethrin I Natural products CC(=CC1CC1C(=O)OC2CC(=O)C(=C2C)CC=C/C=C)C VQXSOUPNOZTNAI-UHFFFAOYSA-N 0.000 description 1
- 239000005926 Pyridalyl Substances 0.000 description 1
- MWMQNVGAHVXSPE-UHFFFAOYSA-N Pyriprole Chemical compound ClC=1C=C(C(F)(F)F)C=C(Cl)C=1N1N=C(C#N)C(SC(F)F)=C1NCC1=CC=CC=N1 MWMQNVGAHVXSPE-UHFFFAOYSA-N 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 241001481703 Rhipicephalus <genus> Species 0.000 description 1
- 241000722249 Rhodnius prolixus Species 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 229930001406 Ryanodine Natural products 0.000 description 1
- 241000277331 Salmonidae Species 0.000 description 1
- 206010039509 Scab Diseases 0.000 description 1
- 241000256103 Simuliidae Species 0.000 description 1
- 241000256108 Simulium <genus> Species 0.000 description 1
- 241000044136 Solenopotes Species 0.000 description 1
- 239000005929 Spinetoram Substances 0.000 description 1
- GOENIMGKWNZVDA-OAMCMWGQSA-N Spinetoram Chemical compound CO[C@@H]1[C@H](OCC)[C@@H](OC)[C@H](C)O[C@H]1OC1C[C@H]2[C@@H]3C=C4C(=O)[C@H](C)[C@@H](O[C@@H]5O[C@H](C)[C@H](CC5)N(C)C)CCC[C@H](CC)OC(=O)CC4[C@@H]3CC[C@@H]2C1 GOENIMGKWNZVDA-OAMCMWGQSA-N 0.000 description 1
- 239000005930 Spinosad Substances 0.000 description 1
- 239000005664 Spirodiclofen Substances 0.000 description 1
- 239000005665 Spiromesifen Substances 0.000 description 1
- 239000005931 Spirotetramat Substances 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 241001494115 Stomoxys calcitrans Species 0.000 description 1
- 241000187747 Streptomyces Species 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 241000282898 Sus scrofa Species 0.000 description 1
- 241000255626 Tabanus <genus> Species 0.000 description 1
- 239000005938 Teflubenzuron Substances 0.000 description 1
- 239000005939 Tefluthrin Substances 0.000 description 1
- 239000005940 Thiacloprid Substances 0.000 description 1
- 239000005941 Thiamethoxam Substances 0.000 description 1
- 241001414833 Triatoma Species 0.000 description 1
- 229930003268 Vitamin C Natural products 0.000 description 1
- 241000353224 Xenopsylla Species 0.000 description 1
- QQODLKZGRKWIFG-RUTXASTPSA-N [(R)-cyano-(4-fluoro-3-phenoxyphenyl)methyl] (1S)-3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropane-1-carboxylate Chemical compound CC1(C)C(C=C(Cl)Cl)[C@@H]1C(=O)O[C@@H](C#N)C1=CC=C(F)C(OC=2C=CC=CC=2)=C1 QQODLKZGRKWIFG-RUTXASTPSA-N 0.000 description 1
- KVIZNNVXXNFLMU-AIIUZBJTSA-N [2,3,5,6-tetrafluoro-4-(methoxymethyl)phenyl]methyl (1r,3r)-2,2-dimethyl-3-[(e)-prop-1-enyl]cyclopropane-1-carboxylate Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)[C@H]1C(C)(C)[C@@H]1\C=C\C KVIZNNVXXNFLMU-AIIUZBJTSA-N 0.000 description 1
- OOWCJRMYMAMSOH-UHFFFAOYSA-N [2,3,5,6-tetrafluoro-4-(methoxymethyl)phenyl]methyl 2,2-dimethyl-3-(2-methylprop-1-enyl)cyclopropane-1-carboxylate Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)C1C(C)(C)C1C=C(C)C OOWCJRMYMAMSOH-UHFFFAOYSA-N 0.000 description 1
- 230000037374 absorbed through the skin Effects 0.000 description 1
- YASYVMFAVPKPKE-UHFFFAOYSA-N acephate Chemical compound COP(=O)(SC)NC(C)=O YASYVMFAVPKPKE-UHFFFAOYSA-N 0.000 description 1
- GDZNYEZGJAFIKA-UHFFFAOYSA-N acetoprole Chemical compound NC1=C(S(C)=O)C(C(=O)C)=NN1C1=C(Cl)C=C(C(F)(F)F)C=C1Cl GDZNYEZGJAFIKA-UHFFFAOYSA-N 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 230000002506 adulticidal effect Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 229960002669 albendazole Drugs 0.000 description 1
- HXHWSAZORRCQMX-UHFFFAOYSA-N albendazole Chemical compound CCCSC1=CC=C2NC(NC(=O)OC)=NC2=C1 HXHWSAZORRCQMX-UHFFFAOYSA-N 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 150000001346 alkyl aryl ethers Chemical class 0.000 description 1
- 125000005907 alkyl ester group Chemical group 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 150000008361 aminoacetonitriles Chemical class 0.000 description 1
- 229960002587 amitraz Drugs 0.000 description 1
- QXAITBQSYVNQDR-ZIOPAAQOSA-N amitraz Chemical compound C=1C=C(C)C=C(C)C=1/N=C/N(C)\C=N\C1=CC=C(C)C=C1C QXAITBQSYVNQDR-ZIOPAAQOSA-N 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 239000003674 animal food additive Substances 0.000 description 1
- 239000010775 animal oil Substances 0.000 description 1
- 230000002924 anti-infective effect Effects 0.000 description 1
- 229960005475 antiinfective agent Drugs 0.000 description 1
- 229940111133 antiinflammatory and antirheumatic drug oxicams Drugs 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 229960003093 antiseptics and disinfectants Drugs 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 239000000823 artificial membrane Substances 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- VEHPJKVTJQSSKL-UHFFFAOYSA-N azadirachtin Natural products O1C2(C)C(C3(C=COC3O3)O)CC3C21C1(C)C(O)C(OCC2(OC(C)=O)C(CC3OC(=O)C(C)=CC)OC(C)=O)C2C32COC(C(=O)OC)(O)C12 VEHPJKVTJQSSKL-UHFFFAOYSA-N 0.000 description 1
- FTNJWQUOZFUQQJ-IRYYUVNJSA-N azadirachtin A Natural products C([C@@H]([C@]1(C=CO[C@H]1O1)O)[C@]2(C)O3)[C@H]1[C@]23[C@]1(C)[C@H](O)[C@H](OC[C@@]2([C@@H](C[C@@H]3OC(=O)C(\C)=C/C)OC(C)=O)C(=O)OC)[C@@H]2[C@]32CO[C@@](C(=O)OC)(O)[C@@H]12 FTNJWQUOZFUQQJ-IRYYUVNJSA-N 0.000 description 1
- FTNJWQUOZFUQQJ-NDAWSKJSSA-N azadirachtin A Chemical compound C([C@@H]([C@]1(C=CO[C@H]1O1)O)[C@]2(C)O3)[C@H]1[C@]23[C@]1(C)[C@H](O)[C@H](OC[C@@]2([C@@H](C[C@@H]3OC(=O)C(\C)=C\C)OC(C)=O)C(=O)OC)[C@@H]2[C@]32CO[C@@](C(=O)OC)(O)[C@@H]12 FTNJWQUOZFUQQJ-NDAWSKJSSA-N 0.000 description 1
- CJJOSEISRRTUQB-UHFFFAOYSA-N azinphos-methyl Chemical group C1=CC=C2C(=O)N(CSP(=S)(OC)OC)N=NC2=C1 CJJOSEISRRTUQB-UHFFFAOYSA-N 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- 238000003287 bathing Methods 0.000 description 1
- 229950000210 beclometasone dipropionate Drugs 0.000 description 1
- 230000006399 behavior Effects 0.000 description 1
- 229960000686 benzalkonium chloride Drugs 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical class OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 1
- 229940092714 benzenesulfonic acid Drugs 0.000 description 1
- 150000001556 benzimidazoles Chemical class 0.000 description 1
- 150000001558 benzoic acid derivatives Chemical class 0.000 description 1
- CKNWWFLJDPCURW-UHFFFAOYSA-N benzoic acid;2-phenylphenol Chemical compound OC(=O)C1=CC=CC=C1.OC1=CC=CC=C1C1=CC=CC=C1 CKNWWFLJDPCURW-UHFFFAOYSA-N 0.000 description 1
- 229960002903 benzyl benzoate Drugs 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 229960002537 betamethasone Drugs 0.000 description 1
- UREBDLICKHMUKA-DVTGEIKXSA-N betamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-DVTGEIKXSA-N 0.000 description 1
- SNHRLVCMMWUAJD-SUYDQAKGSA-N betamethasone valerate Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(OC(=O)CCCC)[C@@]1(C)C[C@@H]2O SNHRLVCMMWUAJD-SUYDQAKGSA-N 0.000 description 1
- 229960004311 betamethasone valerate Drugs 0.000 description 1
- VHLKTXFWDRXILV-UHFFFAOYSA-N bifenazate Chemical compound C1=C(NNC(=O)OC(C)C)C(OC)=CC=C1C1=CC=CC=C1 VHLKTXFWDRXILV-UHFFFAOYSA-N 0.000 description 1
- OMFRMAHOUUJSGP-IRHGGOMRSA-N bifenthrin Chemical compound C1=CC=C(C=2C=CC=CC=2)C(C)=C1COC(=O)[C@@H]1[C@H](\C=C(/Cl)C(F)(F)F)C1(C)C OMFRMAHOUUJSGP-IRHGGOMRSA-N 0.000 description 1
- 229960004436 budesonide Drugs 0.000 description 1
- 229940043232 butyl acetate Drugs 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- FUFJGUQYACFECW-UHFFFAOYSA-L calcium hydrogenphosphate Chemical compound [Ca+2].OP([O-])([O-])=O FUFJGUQYACFECW-UHFFFAOYSA-L 0.000 description 1
- DUEPRVBVGDRKAG-UHFFFAOYSA-N carbofuran Chemical compound CNC(=O)OC1=CC=CC2=C1OC(C)(C)C2 DUEPRVBVGDRKAG-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 235000010418 carrageenan Nutrition 0.000 description 1
- 239000000679 carrageenan Substances 0.000 description 1
- 229920001525 carrageenan Polymers 0.000 description 1
- 229940113118 carrageenan Drugs 0.000 description 1
- 239000012876 carrier material Substances 0.000 description 1
- IRUJZVNXZWPBMU-UHFFFAOYSA-N cartap Chemical compound NC(=O)SCC(N(C)C)CSC(N)=O IRUJZVNXZWPBMU-UHFFFAOYSA-N 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 229920003086 cellulose ether Polymers 0.000 description 1
- 229960001927 cetylpyridinium chloride Drugs 0.000 description 1
- YMKDRGPMQRFJGP-UHFFFAOYSA-M cetylpyridinium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCC[N+]1=CC=CC=C1 YMKDRGPMQRFJGP-UHFFFAOYSA-M 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 239000007910 chewable tablet Substances 0.000 description 1
- 229940068682 chewable tablet Drugs 0.000 description 1
- 235000013330 chicken meat Nutrition 0.000 description 1
- PSOVNZZNOMJUBI-UHFFFAOYSA-N chlorantraniliprole Chemical compound CNC(=O)C1=CC(Cl)=CC(C)=C1NC(=O)C1=CC(Br)=NN1C1=NC=CC=C1Cl PSOVNZZNOMJUBI-UHFFFAOYSA-N 0.000 description 1
- CWFOCCVIPCEQCK-UHFFFAOYSA-N chlorfenapyr Chemical compound BrC1=C(C(F)(F)F)N(COCC)C(C=2C=CC(Cl)=CC=2)=C1C#N CWFOCCVIPCEQCK-UHFFFAOYSA-N 0.000 description 1
- SBPBAQFWLVIOKP-UHFFFAOYSA-N chlorpyrifos Chemical compound CCOP(=S)(OCC)OC1=NC(Cl)=C(Cl)C=C1Cl SBPBAQFWLVIOKP-UHFFFAOYSA-N 0.000 description 1
- HPNSNYBUADCFDR-UHFFFAOYSA-N chromafenozide Chemical compound CC1=CC(C)=CC(C(=O)N(NC(=O)C=2C(=C3CCCOC3=CC=2)C)C(C)(C)C)=C1 HPNSNYBUADCFDR-UHFFFAOYSA-N 0.000 description 1
- 229960003728 ciclesonide Drugs 0.000 description 1
- 229950004178 closantel Drugs 0.000 description 1
- 229940110456 cocoa butter Drugs 0.000 description 1
- 235000019868 cocoa butter Nutrition 0.000 description 1
- 229940075614 colloidal silicon dioxide Drugs 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000002301 combined effect Effects 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 239000003246 corticosteroid Substances 0.000 description 1
- 229960001334 corticosteroids Drugs 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 239000002385 cottonseed oil Substances 0.000 description 1
- 229940111134 coxibs Drugs 0.000 description 1
- 229960000913 crospovidone Drugs 0.000 description 1
- 235000010947 crosslinked sodium carboxy methyl cellulose Nutrition 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- WZHCOOQXZCIUNC-UHFFFAOYSA-N cyclandelate Chemical compound C1C(C)(C)CC(C)CC1OC(=O)C(O)C1=CC=CC=C1 WZHCOOQXZCIUNC-UHFFFAOYSA-N 0.000 description 1
- 239000003255 cyclooxygenase 2 inhibitor Substances 0.000 description 1
- ZXQYGBMAQZUVMI-UNOMPAQXSA-N cyhalothrin Chemical compound CC1(C)C(\C=C(/Cl)C(F)(F)F)C1C(=O)OC(C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 ZXQYGBMAQZUVMI-UNOMPAQXSA-N 0.000 description 1
- 229960005424 cypermethrin Drugs 0.000 description 1
- KAATUXNTWXVJKI-UHFFFAOYSA-N cypermethrin Chemical compound CC1(C)C(C=C(Cl)Cl)C1C(=O)OC(C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 KAATUXNTWXVJKI-UHFFFAOYSA-N 0.000 description 1
- 229960002483 decamethrin Drugs 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229960001145 deflazacort Drugs 0.000 description 1
- FBHSPRKOSMHSIF-GRMWVWQJSA-N deflazacort Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@@H]2[C@@H]1[C@@H]1C[C@H]3OC(C)=N[C@@]3(C(=O)COC(=O)C)[C@@]1(C)C[C@@H]2O FBHSPRKOSMHSIF-GRMWVWQJSA-N 0.000 description 1
- OWZREIFADZCYQD-NSHGMRRFSA-N deltamethrin Chemical compound CC1(C)[C@@H](C=C(Br)Br)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 OWZREIFADZCYQD-NSHGMRRFSA-N 0.000 description 1
- 239000007933 dermal patch Substances 0.000 description 1
- 231100000223 dermal penetration Toxicity 0.000 description 1
- 229940000033 dermatological agent Drugs 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 229960003957 dexamethasone Drugs 0.000 description 1
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- WOWBFOBYOAGEEA-UHFFFAOYSA-N diafenthiuron Chemical compound CC(C)C1=C(NC(=S)NC(C)(C)C)C(C(C)C)=CC(OC=2C=CC=CC=2)=C1 WOWBFOBYOAGEEA-UHFFFAOYSA-N 0.000 description 1
- 150000008050 dialkyl sulfates Chemical class 0.000 description 1
- FHIVAFMUCKRCQO-UHFFFAOYSA-N diazinon Chemical compound CCOP(=S)(OCC)OC1=CC(C)=NC(C(C)C)=N1 FHIVAFMUCKRCQO-UHFFFAOYSA-N 0.000 description 1
- 235000019700 dicalcium phosphate Nutrition 0.000 description 1
- 229940095079 dicalcium phosphate anhydrous Drugs 0.000 description 1
- 150000001991 dicarboxylic acids Chemical class 0.000 description 1
- DFBKLUNHFCTMDC-PICURKEMSA-N dieldrin Chemical compound C([C@H]1[C@H]2[C@@]3(Cl)C(Cl)=C([C@]([C@H]22)(Cl)C3(Cl)Cl)Cl)[C@H]2[C@@H]2[C@H]1O2 DFBKLUNHFCTMDC-PICURKEMSA-N 0.000 description 1
- 229950006824 dieldrin Drugs 0.000 description 1
- NGPMUTDCEIKKFM-UHFFFAOYSA-N dieldrin Natural products CC1=C(Cl)C2(Cl)C3C4CC(C5OC45)C3C1(Cl)C2(Cl)Cl NGPMUTDCEIKKFM-UHFFFAOYSA-N 0.000 description 1
- JXSJBGJIGXNWCI-UHFFFAOYSA-N diethyl 2-[(dimethoxyphosphorothioyl)thio]succinate Chemical compound CCOC(=O)CC(SP(=S)(OC)OC)C(=O)OCC JXSJBGJIGXNWCI-UHFFFAOYSA-N 0.000 description 1
- 125000004177 diethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229940028356 diethylene glycol monobutyl ether Drugs 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- MCWXGJITAZMZEV-UHFFFAOYSA-N dimethoate Chemical compound CNC(=O)CSP(=S)(OC)OC MCWXGJITAZMZEV-UHFFFAOYSA-N 0.000 description 1
- APMCZEMFQVQTHY-AGACNZRVSA-N dimethyl (1S,4S,5R,6S,7S,8R,11S,12R,14S,15R)-12-acetyloxy-4,7-dihydroxy-6-[(1S,2S,6S,8S,9R,11S)-2-hydroxy-11-methyl-5,7,10-trioxatetracyclo[6.3.1.02,6.09,11]dodecan-9-yl]-6-methyl-14-(2-methylbutanoyloxy)-3,9-dioxatetracyclo[6.6.1.01,5.011,15]pentadecane-4,11-dicarboxylate Chemical compound C([C@@H]([C@]1(CCO[C@H]1O1)O)[C@]2(C)O3)[C@H]1[C@]23[C@](C)([C@@H]1O)[C@@H]2[C@@](O)(C(=O)OC)OC[C@@]32[C@H]2[C@H]1OC[C@]2(C(=O)OC)[C@H](OC(C)=O)C[C@@H]3OC(=O)C(C)CC APMCZEMFQVQTHY-AGACNZRVSA-N 0.000 description 1
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 1
- YKBZOVFACRVRJN-UHFFFAOYSA-N dinotefuran Chemical compound [O-][N+](=O)\N=C(/NC)NCC1CCOC1 YKBZOVFACRVRJN-UHFFFAOYSA-N 0.000 description 1
- GAFRWLVTHPVQGK-UHFFFAOYSA-N dipentyl sulfate Chemical compound CCCCCOS(=O)(=O)OCCCCC GAFRWLVTHPVQGK-UHFFFAOYSA-N 0.000 description 1
- 238000007598 dipping method Methods 0.000 description 1
- QQJKLNZDWULQGR-UHFFFAOYSA-N dipropan-2-yl benzene-1,3-dicarboxylate Chemical compound CC(C)OC(=O)C1=CC=CC(C(=O)OC(C)C)=C1 QQJKLNZDWULQGR-UHFFFAOYSA-N 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- 229940110377 dl- arginine Drugs 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- QLFZZSKTJWDQOS-YDBLARSUSA-N doramectin Chemical compound O1[C@@H](C)[C@H](O)[C@@H](OC)C[C@@H]1O[C@@H]1[C@@H](OC)C[C@H](O[C@@H]2C(=C/C[C@@H]3C[C@@H](C[C@@]4(O3)C=C[C@H](C)[C@@H](C3CCCCC3)O4)OC(=O)[C@@H]3C=C(C)[C@@H](O)[C@H]4OC\C([C@@]34O)=C/C=C/[C@@H]2C)/C)O[C@H]1C QLFZZSKTJWDQOS-YDBLARSUSA-N 0.000 description 1
- 229960003997 doramectin Drugs 0.000 description 1
- 238000010410 dusting Methods 0.000 description 1
- ZMQMTKVVAMWKNY-YSXLEBCMSA-N emodepside Chemical compound C([C@@H]1C(=O)N(C)[C@@H](CC(C)C)C(=O)O[C@H](C)C(=O)N(C)[C@H](C(O[C@H](CC=2C=CC(=CC=2)N2CCOCC2)C(=O)N(C)[C@@H](CC(C)C)C(=O)O[C@H](C)C(=O)N(C)[C@@H](CC(C)C)C(=O)O1)=O)CC(C)C)C(C=C1)=CC=C1N1CCOCC1 ZMQMTKVVAMWKNY-YSXLEBCMSA-N 0.000 description 1
- 229960001575 emodepside Drugs 0.000 description 1
- 108010056417 emodepside Proteins 0.000 description 1
- 238000003821 enantio-separation Methods 0.000 description 1
- 230000002323 endectocidal effect Effects 0.000 description 1
- 244000079386 endoparasite Species 0.000 description 1
- RDYMFSUJUZBWLH-SVWSLYAFSA-N endosulfan Chemical compound C([C@@H]12)OS(=O)OC[C@@H]1[C@]1(Cl)C(Cl)=C(Cl)[C@@]2(Cl)C1(Cl)Cl RDYMFSUJUZBWLH-SVWSLYAFSA-N 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 239000002702 enteric coating Substances 0.000 description 1
- 238000009505 enteric coating Methods 0.000 description 1
- WPNHOHPRXXCPRA-TVXIRPTOSA-N eprinomectin Chemical compound O1[C@@H](C)[C@@H](NC(C)=O)[C@H](OC)C[C@@H]1O[C@H]1[C@@H](OC)C[C@H](O[C@@H]2C(=C/C[C@@H]3C[C@@H](C[C@@]4(O3)C=C[C@H](C)[C@@H](C(C)C)O4)OC(=O)[C@@H]3C=C(C)[C@@H](O)[C@H]4OC\C([C@@]34O)=C\C=C/[C@@H]2C)\C)O[C@H]1C WPNHOHPRXXCPRA-TVXIRPTOSA-N 0.000 description 1
- 229960002346 eprinomectin Drugs 0.000 description 1
- 230000003628 erosive effect Effects 0.000 description 1
- NYPJDWWKZLNGGM-RPWUZVMVSA-N esfenvalerate Chemical compound C=1C([C@@H](C#N)OC(=O)[C@@H](C(C)C)C=2C=CC(Cl)=CC=2)=CC=CC=1OC1=CC=CC=C1 NYPJDWWKZLNGGM-RPWUZVMVSA-N 0.000 description 1
- 229950009769 etabonate Drugs 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 229940093499 ethyl acetate Drugs 0.000 description 1
- 235000019325 ethyl cellulose Nutrition 0.000 description 1
- 229920001249 ethyl cellulose Polymers 0.000 description 1
- 229960004667 ethyl cellulose Drugs 0.000 description 1
- HDDSHPAODJUKPD-UHFFFAOYSA-N fenbendazole Chemical compound C1=C2NC(NC(=O)OC)=NC2=CC=C1SC1=CC=CC=C1 HDDSHPAODJUKPD-UHFFFAOYSA-N 0.000 description 1
- 229960005473 fenbendazole Drugs 0.000 description 1
- XQUXKZZNEFRCAW-UHFFFAOYSA-N fenpropathrin Chemical compound CC1(C)C(C)(C)C1C(=O)OC(C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 XQUXKZZNEFRCAW-UHFFFAOYSA-N 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- 235000019688 fish Nutrition 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- RLQJEEJISHYWON-UHFFFAOYSA-N flonicamid Chemical compound FC(F)(F)C1=CC=NC=C1C(=O)NCC#N RLQJEEJISHYWON-UHFFFAOYSA-N 0.000 description 1
- YOWNVPAUWYHLQX-UHFFFAOYSA-N fluazuron Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC1=CC=C(Cl)C(OC=2C(=CC(=CN=2)C(F)(F)F)Cl)=C1 YOWNVPAUWYHLQX-UHFFFAOYSA-N 0.000 description 1
- 229950006719 fluazuron Drugs 0.000 description 1
- ZGNITFSDLCMLGI-UHFFFAOYSA-N flubendiamide Chemical compound CC1=CC(C(F)(C(F)(F)F)C(F)(F)F)=CC=C1NC(=O)C1=CC=CC(I)=C1C(=O)NC(C)(C)CS(C)(=O)=O ZGNITFSDLCMLGI-UHFFFAOYSA-N 0.000 description 1
- GBIHOLCMZGAKNG-CGAIIQECSA-N flucythrinate Chemical compound O=C([C@@H](C(C)C)C=1C=CC(OC(F)F)=CC=1)OC(C#N)C(C=1)=CC=CC=1OC1=CC=CC=C1 GBIHOLCMZGAKNG-CGAIIQECSA-N 0.000 description 1
- GJEREQYJIQASAW-UHFFFAOYSA-N flufenerim Chemical compound CC(F)C1=NC=NC(NCCC=2C=CC(OC(F)(F)F)=CC=2)=C1Cl GJEREQYJIQASAW-UHFFFAOYSA-N 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 229960001347 fluocinolone acetonide Drugs 0.000 description 1
- FEBLZLNTKCEFIT-VSXGLTOVSA-N fluocinolone acetonide Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]2(C)C[C@@H]1O FEBLZLNTKCEFIT-VSXGLTOVSA-N 0.000 description 1
- MLBZKOGAMRTSKP-UHFFFAOYSA-N fluralaner Chemical compound C1=C(C(=O)NCC(=O)NCC(F)(F)F)C(C)=CC(C=2CC(ON=2)(C=2C=C(Cl)C=C(Cl)C=2)C(F)(F)F)=C1 MLBZKOGAMRTSKP-UHFFFAOYSA-N 0.000 description 1
- 229960002714 fluticasone Drugs 0.000 description 1
- MGNNYOODZCAHBA-GQKYHHCASA-N fluticasone Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@@H](C)[C@@](C(=O)SCF)(O)[C@@]2(C)C[C@@H]1O MGNNYOODZCAHBA-GQKYHHCASA-N 0.000 description 1
- 229960001469 fluticasone furoate Drugs 0.000 description 1
- XTULMSXFIHGYFS-VLSRWLAYSA-N fluticasone furoate Chemical compound O([C@]1([C@@]2(C)C[C@H](O)[C@]3(F)[C@@]4(C)C=CC(=O)C=C4[C@@H](F)C[C@H]3[C@@H]2C[C@H]1C)C(=O)SCF)C(=O)C1=CC=CO1 XTULMSXFIHGYFS-VLSRWLAYSA-N 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- KVGLBTYUCJYMND-UHFFFAOYSA-N fonofos Chemical compound CCOP(=S)(CC)SC1=CC=CC=C1 KVGLBTYUCJYMND-UHFFFAOYSA-N 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- CNKHSLKYRMDDNQ-UHFFFAOYSA-N halofenozide Chemical compound C=1C=CC=CC=1C(=O)N(C(C)(C)C)NC(=O)C1=CC=C(Cl)C=C1 CNKHSLKYRMDDNQ-UHFFFAOYSA-N 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- 229910001385 heavy metal Inorganic materials 0.000 description 1
- MNWFXJYAOYHMED-UHFFFAOYSA-N heptanoic acid group Chemical group C(CCCCCC)(=O)O MNWFXJYAOYHMED-UHFFFAOYSA-N 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 150000002400 hexanoic acid esters Chemical class 0.000 description 1
- 229940100463 hexyl laurate Drugs 0.000 description 1
- 231100000086 high toxicity Toxicity 0.000 description 1
- 210000003630 histaminocyte Anatomy 0.000 description 1
- 229920001519 homopolymer Polymers 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 239000008309 hydrophilic cream Substances 0.000 description 1
- FYQGBXGJFWXIPP-UHFFFAOYSA-N hydroprene Chemical compound CCOC(=O)C=C(C)C=CCC(C)CCCC(C)C FYQGBXGJFWXIPP-UHFFFAOYSA-N 0.000 description 1
- 229930000073 hydroprene Natural products 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 229940071826 hydroxyethyl cellulose Drugs 0.000 description 1
- 229940056881 imidacloprid Drugs 0.000 description 1
- YWTYJOPNNQFBPC-UHFFFAOYSA-N imidacloprid Chemical compound [O-][N+](=O)\N=C1/NCCN1CC1=CC=C(Cl)N=C1 YWTYJOPNNQFBPC-UHFFFAOYSA-N 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- VBCVPMMZEGZULK-NRFANRHFSA-N indoxacarb Chemical compound C([C@@]1(OC2)C(=O)OC)C3=CC(Cl)=CC=C3C1=NN2C(=O)N(C(=O)OC)C1=CC=C(OC(F)(F)F)C=C1 VBCVPMMZEGZULK-NRFANRHFSA-N 0.000 description 1
- 229940060367 inert ingredients Drugs 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 229940102223 injectable solution Drugs 0.000 description 1
- 230000000266 injurious effect Effects 0.000 description 1
- 238000010255 intramuscular injection Methods 0.000 description 1
- 239000007927 intramuscular injection Substances 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 150000002505 iron Chemical class 0.000 description 1
- HOQADATXFBOEGG-UHFFFAOYSA-N isofenphos Chemical compound CCOP(=S)(NC(C)C)OC1=CC=CC=C1C(=O)OC(C)C HOQADATXFBOEGG-UHFFFAOYSA-N 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- XUGNVMKQXJXZCD-UHFFFAOYSA-N isopropyl palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC(C)C XUGNVMKQXJXZCD-UHFFFAOYSA-N 0.000 description 1
- 229930014550 juvenile hormone Natural products 0.000 description 1
- 150000003633 juvenile hormone derivatives Chemical class 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 229930001540 kinoprene Natural products 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 239000005910 lambda-Cyhalothrin Substances 0.000 description 1
- 229940039717 lanolin Drugs 0.000 description 1
- 235000019388 lanolin Nutrition 0.000 description 1
- 229940094522 laponite Drugs 0.000 description 1
- 230000000974 larvacidal effect Effects 0.000 description 1
- 230000002045 lasting effect Effects 0.000 description 1
- 229960001614 levamisole Drugs 0.000 description 1
- UAWXGRJVZSAUSZ-UHFFFAOYSA-N licofelone Chemical compound OC(=O)CC=1N2CC(C)(C)CC2=C(C=2C=CC=CC=2)C=1C1=CC=C(Cl)C=C1 UAWXGRJVZSAUSZ-UHFFFAOYSA-N 0.000 description 1
- 229950003488 licofelone Drugs 0.000 description 1
- 239000012669 liquid formulation Substances 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- XCOBTUNSZUJCDH-UHFFFAOYSA-B lithium magnesium sodium silicate Chemical compound [Li+].[Li+].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[Na+].[Na+].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3 XCOBTUNSZUJCDH-UHFFFAOYSA-B 0.000 description 1
- 229960001798 loteprednol Drugs 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- PWPJGUXAGUPAHP-UHFFFAOYSA-N lufenuron Chemical compound C1=C(Cl)C(OC(F)(F)C(C(F)(F)F)F)=CC(Cl)=C1NC(=O)NC(=O)C1=C(F)C=CC=C1F PWPJGUXAGUPAHP-UHFFFAOYSA-N 0.000 description 1
- 229960000521 lufenuron Drugs 0.000 description 1
- 210000002751 lymph Anatomy 0.000 description 1
- 210000004880 lymph fluid Anatomy 0.000 description 1
- 150000002680 magnesium Chemical class 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 229960000453 malathion Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical class OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Chemical class 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- GKKDCARASOJPNG-UHFFFAOYSA-N metaldehyde Chemical compound CC1OC(C)OC(C)OC(C)O1 GKKDCARASOJPNG-UHFFFAOYSA-N 0.000 description 1
- NNKVPIKMPCQWCG-UHFFFAOYSA-N methamidophos Chemical compound COP(N)(=O)SC NNKVPIKMPCQWCG-UHFFFAOYSA-N 0.000 description 1
- MEBQXILRKZHVCX-UHFFFAOYSA-N methidathion Chemical compound COC1=NN(CSP(=S)(OC)OC)C(=O)S1 MEBQXILRKZHVCX-UHFFFAOYSA-N 0.000 description 1
- UHXUZOCRWCRNSJ-QPJJXVBHSA-N methomyl Chemical compound CNC(=O)O\N=C(/C)SC UHXUZOCRWCRNSJ-QPJJXVBHSA-N 0.000 description 1
- QCAWEPFNJXQPAN-UHFFFAOYSA-N methoxyfenozide Chemical compound COC1=CC=CC(C(=O)NN(C(=O)C=2C=C(C)C=C(C)C=2)C(C)(C)C)=C1C QCAWEPFNJXQPAN-UHFFFAOYSA-N 0.000 description 1
- KBHDSWIXRODKSZ-UHFFFAOYSA-N methyl 5-chloro-2-(trifluoromethylsulfonylamino)benzoate Chemical compound COC(=O)C1=CC(Cl)=CC=C1NS(=O)(=O)C(F)(F)F KBHDSWIXRODKSZ-UHFFFAOYSA-N 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 229960002900 methylcellulose Drugs 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- 229960004584 methylprednisolone Drugs 0.000 description 1
- 229960001952 metrifonate Drugs 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 235000013336 milk Nutrition 0.000 description 1
- 239000008267 milk Substances 0.000 description 1
- 210000004080 milk Anatomy 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 239000003595 mist Substances 0.000 description 1
- WLLGXSLBOPFWQV-OTHKPKEBSA-N molport-035-783-878 Chemical compound C([C@H]1C=C2)[C@H]2C2C1C(=O)N(CC(CC)CCCC)C2=O WLLGXSLBOPFWQV-OTHKPKEBSA-N 0.000 description 1
- 229960001664 mometasone Drugs 0.000 description 1
- QLIIKPVHVRXHRI-CXSFZGCWSA-N mometasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(Cl)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CCl)(O)[C@@]1(C)C[C@@H]2O QLIIKPVHVRXHRI-CXSFZGCWSA-N 0.000 description 1
- 229960002744 mometasone furoate Drugs 0.000 description 1
- WOFMFGQZHJDGCX-ZULDAHANSA-N mometasone furoate Chemical compound O([C@]1([C@@]2(C)C[C@H](O)[C@]3(Cl)[C@@]4(C)C=CC(=O)C=C4CC[C@H]3[C@@H]2C[C@H]1C)C(=O)CCl)C(=O)C1=CC=CO1 WOFMFGQZHJDGCX-ZULDAHANSA-N 0.000 description 1
- 150000002763 monocarboxylic acids Chemical class 0.000 description 1
- 235000011929 mousse Nutrition 0.000 description 1
- 210000004877 mucosa Anatomy 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- YNKFZRGTXAPYFD-UHFFFAOYSA-N n-[[2-chloro-3,5-bis(trifluoromethyl)phenyl]carbamoyl]-2,6-difluorobenzamide Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC1=CC(C(F)(F)F)=CC(C(F)(F)F)=C1Cl YNKFZRGTXAPYFD-UHFFFAOYSA-N 0.000 description 1
- 229920001206 natural gum Polymers 0.000 description 1
- 229940079888 nitenpyram Drugs 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- UEHLXXJAWYWUGI-UHFFFAOYSA-M nitromersol Chemical compound CC1=CC=C([N+]([O-])=O)C2=C1O[Hg]2 UEHLXXJAWYWUGI-UHFFFAOYSA-M 0.000 description 1
- 229940118238 nitromersol Drugs 0.000 description 1
- SGKGVABHDAQAJO-UHFFFAOYSA-N nitroxynil Chemical compound OC1=C(I)C=C(C#N)C=C1[N+]([O-])=O SGKGVABHDAQAJO-UHFFFAOYSA-N 0.000 description 1
- 229930187416 nodulisporic acid Natural products 0.000 description 1
- 231100000344 non-irritating Toxicity 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 239000000346 nonvolatile oil Substances 0.000 description 1
- NJPPVKZQTLUDBO-UHFFFAOYSA-N novaluron Chemical compound C1=C(Cl)C(OC(F)(F)C(OC(F)(F)F)F)=CC=C1NC(=O)NC(=O)C1=C(F)C=CC=C1F NJPPVKZQTLUDBO-UHFFFAOYSA-N 0.000 description 1
- YTYGAJLZOJPJGH-UHFFFAOYSA-N noviflumuron Chemical compound FC1=C(Cl)C(OC(F)(F)C(C(F)(F)F)F)=C(Cl)C=C1NC(=O)NC(=O)C1=C(F)C=CC=C1F YTYGAJLZOJPJGH-UHFFFAOYSA-N 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 239000003883 ointment base Substances 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 229940055577 oleyl alcohol Drugs 0.000 description 1
- XMLQWXUVTXCDDL-UHFFFAOYSA-N oleyl alcohol Natural products CCCCCCC=CCCCCCCCCCCO XMLQWXUVTXCDDL-UHFFFAOYSA-N 0.000 description 1
- 231100000194 ovacidal Toxicity 0.000 description 1
- 230000003151 ovacidal effect Effects 0.000 description 1
- JCGNDDUYTRNOFT-UHFFFAOYSA-N oxolane-2,4-dione Chemical compound O=C1COC(=O)C1 JCGNDDUYTRNOFT-UHFFFAOYSA-N 0.000 description 1
- JYWIYHUXVMAGLG-UHFFFAOYSA-N oxyclozanide Chemical compound OC1=C(Cl)C=C(Cl)C=C1NC(=O)C1=C(O)C(Cl)=CC(Cl)=C1Cl JYWIYHUXVMAGLG-UHFFFAOYSA-N 0.000 description 1
- 229950003126 oxyclozanide Drugs 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- LCCNCVORNKJIRZ-UHFFFAOYSA-N parathion Chemical compound CCOP(=S)(OCC)OC1=CC=C([N+]([O-])=O)C=C1 LCCNCVORNKJIRZ-UHFFFAOYSA-N 0.000 description 1
- 239000006201 parenteral dosage form Substances 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000000312 peanut oil Substances 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 230000037368 penetrate the skin Effects 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 229960000490 permethrin Drugs 0.000 description 1
- RLLPVAHGXHCWKJ-UHFFFAOYSA-N permethrin Chemical compound CC1(C)C(C=C(Cl)Cl)C1C(=O)OCC1=CC=CC(OC=2C=CC=CC=2)=C1 RLLPVAHGXHCWKJ-UHFFFAOYSA-N 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 229940127557 pharmaceutical product Drugs 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- 229960005323 phenoxyethanol Drugs 0.000 description 1
- WLJVXDMOQOGPHL-UHFFFAOYSA-N phenylacetic acid Chemical compound OC(=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-UHFFFAOYSA-N 0.000 description 1
- 229940096826 phenylmercuric acetate Drugs 0.000 description 1
- 125000003356 phenylsulfanyl group Chemical group [*]SC1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- BULVZWIRKLYCBC-UHFFFAOYSA-N phorate Chemical compound CCOP(=S)(OCC)SCSCC BULVZWIRKLYCBC-UHFFFAOYSA-N 0.000 description 1
- IOUNQDKNJZEDEP-UHFFFAOYSA-N phosalone Chemical compound C1=C(Cl)C=C2OC(=O)N(CSP(=S)(OCC)OCC)C2=C1 IOUNQDKNJZEDEP-UHFFFAOYSA-N 0.000 description 1
- LMNZTLDVJIUSHT-UHFFFAOYSA-N phosmet Chemical compound C1=CC=C2C(=O)N(CSP(=S)(OC)OC)C(=O)C2=C1 LMNZTLDVJIUSHT-UHFFFAOYSA-N 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- OXNIZHLAWKMVMX-UHFFFAOYSA-N picric acid Chemical compound OC1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O OXNIZHLAWKMVMX-UHFFFAOYSA-N 0.000 description 1
- 229940023488 pill Drugs 0.000 description 1
- YFGYUFNIOHWBOB-UHFFFAOYSA-N pirimicarb Chemical compound CN(C)C(=O)OC1=NC(N(C)C)=NC(C)=C1C YFGYUFNIOHWBOB-UHFFFAOYSA-N 0.000 description 1
- IUGYQRQAERSCNH-UHFFFAOYSA-N pivalic acid Chemical compound CC(C)(C)C(O)=O IUGYQRQAERSCNH-UHFFFAOYSA-N 0.000 description 1
- 210000002381 plasma Anatomy 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 229920000523 polyvinylpolypyrrolidone Polymers 0.000 description 1
- 235000013809 polyvinylpolypyrrolidone Nutrition 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 150000003109 potassium Chemical class 0.000 description 1
- 229960002957 praziquantel Drugs 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 229960005205 prednisolone Drugs 0.000 description 1
- OIGNJSKKLXVSLS-VWUMJDOOSA-N prednisolone Chemical compound O=C1C=C[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 OIGNJSKKLXVSLS-VWUMJDOOSA-N 0.000 description 1
- 229960004618 prednisone Drugs 0.000 description 1
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 description 1
- QYMMJNLHFKGANY-UHFFFAOYSA-N profenofos Chemical compound CCCSP(=O)(OCC)OC1=CC=C(Br)C=C1Cl QYMMJNLHFKGANY-UHFFFAOYSA-N 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- QHMTXANCGGJZRX-WUXMJOGZSA-N pymetrozine Chemical compound C1C(C)=NNC(=O)N1\N=C\C1=CC=CN=C1 QHMTXANCGGJZRX-WUXMJOGZSA-N 0.000 description 1
- DDIQWGKUSJOETH-UHFFFAOYSA-N pyrafluprole Chemical compound ClC=1C=C(C(F)(F)F)C=C(Cl)C=1N1N=C(C#N)C(SCF)=C1NCC1=CN=CC=N1 DDIQWGKUSJOETH-UHFFFAOYSA-N 0.000 description 1
- 229960005134 pyrantel Drugs 0.000 description 1
- YSAUAVHXTIETRK-AATRIKPKSA-N pyrantel Chemical compound CN1CCCN=C1\C=C\C1=CC=CS1 YSAUAVHXTIETRK-AATRIKPKSA-N 0.000 description 1
- 150000003218 pyrazolidines Chemical class 0.000 description 1
- HYJYGLGUBUDSLJ-UHFFFAOYSA-N pyrethrin Natural products CCC(=O)OC1CC(=C)C2CC3OC3(C)C2C2OC(=O)C(=C)C12 HYJYGLGUBUDSLJ-UHFFFAOYSA-N 0.000 description 1
- VJFUPGQZSXIULQ-XIGJTORUSA-N pyrethrin II Chemical compound CC1(C)[C@H](/C=C(\C)C(=O)OC)[C@H]1C(=O)O[C@@H]1C(C)=C(C\C=C/C=C)C(=O)C1 VJFUPGQZSXIULQ-XIGJTORUSA-N 0.000 description 1
- AEHJMNVBLRLZKK-UHFFFAOYSA-N pyridalyl Chemical group N1=CC(C(F)(F)F)=CC=C1OCCCOC1=C(Cl)C=C(OCC=C(Cl)Cl)C=C1Cl AEHJMNVBLRLZKK-UHFFFAOYSA-N 0.000 description 1
- 150000004892 pyridazines Chemical class 0.000 description 1
- MIOBBYRMXGNORL-UHFFFAOYSA-N pyrifluquinazon Chemical compound C1C2=CC(C(F)(C(F)(F)F)C(F)(F)F)=CC=C2N(C(=O)C)C(=O)N1NCC1=CC=CN=C1 MIOBBYRMXGNORL-UHFFFAOYSA-N 0.000 description 1
- 150000003230 pyrimidines Chemical class 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 230000001846 repelling effect Effects 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 229950004432 rofleponide Drugs 0.000 description 1
- IXTCZMJQGGONPY-XJAYAHQCSA-N rofleponide Chemical compound C1([C@@H](F)C2)=CC(=O)CC[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@H]3O[C@@H](CCC)O[C@@]3(C(=O)CO)[C@@]2(C)C[C@@H]1O IXTCZMJQGGONPY-XJAYAHQCSA-N 0.000 description 1
- 229940080817 rotenone Drugs 0.000 description 1
- JUVIOZPCNVVQFO-UHFFFAOYSA-N rotenone Natural products O1C2=C3CC(C(C)=C)OC3=CC=C2C(=O)C2C1COC1=C2C=C(OC)C(OC)=C1 JUVIOZPCNVVQFO-UHFFFAOYSA-N 0.000 description 1
- JJSYXNQGLHBRRK-SFEDZAPPSA-N ryanodine Chemical compound O([C@@H]1[C@]([C@@]2([C@]3(O)[C@]45O[C@@]2(O)C[C@]([C@]4(CC[C@H](C)[C@H]5O)O)(C)[C@@]31O)C)(O)C(C)C)C(=O)C1=CC=CN1 JJSYXNQGLHBRRK-SFEDZAPPSA-N 0.000 description 1
- 150000003873 salicylate salts Chemical class 0.000 description 1
- 235000019515 salmon Nutrition 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- AFJYYKSVHJGXSN-KAJWKRCWSA-N selamectin Chemical compound O1[C@@H](C)[C@H](O)[C@@H](OC)C[C@@H]1O[C@@H]1C(/C)=C/C[C@@H](O[C@]2(O[C@@H]([C@@H](C)CC2)C2CCCCC2)C2)C[C@@H]2OC(=O)[C@@H]([C@]23O)C=C(C)C(=N\O)/[C@H]3OC\C2=C/C=C/[C@@H]1C AFJYYKSVHJGXSN-KAJWKRCWSA-N 0.000 description 1
- 229960002245 selamectin Drugs 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 239000002453 shampoo Substances 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 235000017550 sodium carbonate Nutrition 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 235000011008 sodium phosphates Nutrition 0.000 description 1
- 229920003109 sodium starch glycolate Polymers 0.000 description 1
- 239000008109 sodium starch glycolate Substances 0.000 description 1
- 229940079832 sodium starch glycolate Drugs 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- WSWCOQWTEOXDQX-MQQKCMAXSA-N sorbic acid group Chemical group C(\C=C\C=C\C)(=O)O WSWCOQWTEOXDQX-MQQKCMAXSA-N 0.000 description 1
- JNYAEWCLZODPBN-CTQIIAAMSA-N sorbitan Polymers OCC(O)C1OCC(O)[C@@H]1O JNYAEWCLZODPBN-CTQIIAAMSA-N 0.000 description 1
- 229940014213 spinosad Drugs 0.000 description 1
- DTDSAWVUFPGDMX-UHFFFAOYSA-N spirodiclofen Chemical compound CCC(C)(C)C(=O)OC1=C(C=2C(=CC(Cl)=CC=2)Cl)C(=O)OC11CCCCC1 DTDSAWVUFPGDMX-UHFFFAOYSA-N 0.000 description 1
- GOLXNESZZPUPJE-UHFFFAOYSA-N spiromesifen Chemical compound CC1=CC(C)=CC(C)=C1C(C(O1)=O)=C(OC(=O)CC(C)(C)C)C11CCCC1 GOLXNESZZPUPJE-UHFFFAOYSA-N 0.000 description 1
- CLSVJBIHYWPGQY-GGYDESQDSA-N spirotetramat Chemical compound CCOC(=O)OC1=C(C=2C(=CC=C(C)C=2)C)C(=O)N[C@@]11CC[C@H](OC)CC1 CLSVJBIHYWPGQY-GGYDESQDSA-N 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 239000004551 spreading oil Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- JXHJNEJVUNHLKO-UHFFFAOYSA-N sulprofos Chemical compound CCCSP(=S)(OCC)OC1=CC=C(SC)C=C1 JXHJNEJVUNHLKO-UHFFFAOYSA-N 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 238000004885 tandem mass spectrometry Methods 0.000 description 1
- 239000008399 tap water Substances 0.000 description 1
- 235000020679 tap water Nutrition 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- 239000005936 tau-Fluvalinate Substances 0.000 description 1
- INISTDXBRIBGOC-XMMISQBUSA-N tau-fluvalinate Chemical compound N([C@H](C(C)C)C(=O)OC(C#N)C=1C=C(OC=2C=CC=CC=2)C=CC=1)C1=CC=C(C(F)(F)F)C=C1Cl INISTDXBRIBGOC-XMMISQBUSA-N 0.000 description 1
- CJDWRQLODFKPEL-UHFFFAOYSA-N teflubenzuron Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC1=CC(Cl)=C(F)C(Cl)=C1F CJDWRQLODFKPEL-UHFFFAOYSA-N 0.000 description 1
- UBCKGWBNUIFUST-YHYXMXQVSA-N tetrachlorvinphos Chemical compound COP(=O)(OC)O\C(=C/Cl)C1=CC(Cl)=C(Cl)C=C1Cl UBCKGWBNUIFUST-YHYXMXQVSA-N 0.000 description 1
- 150000005326 tetrahydropyrimidines Chemical class 0.000 description 1
- NWWZPOKUUAIXIW-FLIBITNWSA-N thiamethoxam Chemical compound [O-][N+](=O)\N=C/1N(C)COCN\1CC1=CN=C(Cl)S1 NWWZPOKUUAIXIW-FLIBITNWSA-N 0.000 description 1
- RTKIYNMVFMVABJ-UHFFFAOYSA-L thimerosal Chemical compound [Na+].CC[Hg]SC1=CC=CC=C1C([O-])=O RTKIYNMVFMVABJ-UHFFFAOYSA-L 0.000 description 1
- 229940033663 thimerosal Drugs 0.000 description 1
- ZMZDMBWJUHKJPS-UHFFFAOYSA-N thiocyanic acid Chemical compound SC#N ZMZDMBWJUHKJPS-UHFFFAOYSA-N 0.000 description 1
- BAKXBZPQTXCKRR-UHFFFAOYSA-N thiodicarb Chemical compound CSC(C)=NOC(=O)NSNC(=O)ON=C(C)SC BAKXBZPQTXCKRR-UHFFFAOYSA-N 0.000 description 1
- QSOHVSNIQHGFJU-UHFFFAOYSA-L thiosultap disodium Chemical compound [Na+].[Na+].[O-]S(=O)(=O)SCC(N(C)C)CSS([O-])(=O)=O QSOHVSNIQHGFJU-UHFFFAOYSA-L 0.000 description 1
- WPALTCMYPARVNV-UHFFFAOYSA-N tolfenpyrad Chemical compound CCC1=NN(C)C(C(=O)NCC=2C=CC(OC=3C=CC(C)=CC=3)=CC=2)=C1Cl WPALTCMYPARVNV-UHFFFAOYSA-N 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- YWSCPYYRJXKUDB-KAKFPZCNSA-N tralomethrin Chemical compound CC1(C)[C@@H](C(Br)C(Br)(Br)Br)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 YWSCPYYRJXKUDB-KAKFPZCNSA-N 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Chemical class OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 229960002117 triamcinolone acetonide Drugs 0.000 description 1
- YNDXUCZADRHECN-JNQJZLCISA-N triamcinolone acetonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O YNDXUCZADRHECN-JNQJZLCISA-N 0.000 description 1
- NKNFWVNSBIXGLL-UHFFFAOYSA-N triazamate Chemical compound CCOC(=O)CSC1=NC(C(C)(C)C)=NN1C(=O)N(C)C NKNFWVNSBIXGLL-UHFFFAOYSA-N 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 235000019731 tricalcium phosphate Nutrition 0.000 description 1
- NFACJZMKEDPNKN-UHFFFAOYSA-N trichlorfon Chemical compound COP(=O)(OC)C(O)C(Cl)(Cl)Cl NFACJZMKEDPNKN-UHFFFAOYSA-N 0.000 description 1
- 229960000323 triclabendazole Drugs 0.000 description 1
- XZZNDPSIHUTMOC-UHFFFAOYSA-N triphenyl phosphate Chemical compound C=1C=CC=CC=1OP(OC=1C=CC=CC=1)(=O)OC1=CC=CC=C1 XZZNDPSIHUTMOC-UHFFFAOYSA-N 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 238000000825 ultraviolet detection Methods 0.000 description 1
- 229960005486 vaccine Drugs 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 239000011718 vitamin C Substances 0.000 description 1
- 235000019154 vitamin C Nutrition 0.000 description 1
- 235000012431 wafers Nutrition 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- UHVMMEOXYDMDKI-JKYCWFKZSA-L zinc;1-(5-cyanopyridin-2-yl)-3-[(1s,2s)-2-(6-fluoro-2-hydroxy-3-propanoylphenyl)cyclopropyl]urea;diacetate Chemical compound [Zn+2].CC([O-])=O.CC([O-])=O.CCC(=O)C1=CC=C(F)C([C@H]2[C@H](C2)NC(=O)NC=2N=CC(=CC=2)C#N)=C1O UHVMMEOXYDMDKI-JKYCWFKZSA-L 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/80—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D261/00—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings
- C07D261/02—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings
- C07D261/04—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4439—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. omeprazole
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
- A01N47/12—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof containing a —O—CO—N< group, or a thio analogue thereof, neither directly attached to a ring nor the nitrogen atom being a member of a heterocyclic ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
- A01N47/18—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof containing a —O—CO—N< group, or a thio analogue thereof, directly attached to a heterocyclic or cycloaliphatic ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N53/00—Biocides, pest repellants or attractants, or plant growth regulators containing cyclopropane carboxylic acids or derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/42—Oxazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/14—Ectoparasiticides, e.g. scabicides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/06—Antianaemics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Definitions
- This invention relates to methods for controlling parasitic infestations of animals and their environments, and, more particularly, to methods using isoxazolines to control parasites in or on animals or in their environments, as well as treat parasitoses of animals.
- the isoxazolines include 4-(isoxazolinyl)-benzamides (specifically, substituted A- (5-(halomethyl)-5-phenyl-isoxazolin-3-yl)-benzamides) and 4-(isoxazolinyl)- benzothioamides (specifically, substituted 4-(5-(halomethyl)-5-phenyl-isoxazolin-3-yl)- benzothioamides).
- This invention also relates to compositions comprising the isoxazolines for use in such methods, the use of the isoxazolines to make medicaments for use in such methods, and kits comprising the isoxazolines for carrying out such methods.
- This invention further relates to the use of the isoxazolines as medicaments, particularly medicaments that can be used in the above-referenced methods.
- a number of pests and parasites are known to infest warm-blooded animals. These pests and parasites can be great nuisances to both the animals and their owners. For example, virtually all companion and livestock animals can be affected by ectoparasites, such as ticks, mites, lice, and fleas. Ectoparasites tend to irritate the animals, and also can cause clinical disease and adverse sub-clinical conditions, either by themselves or by carrying vector-transmitted pathogens. To date, various treatments have been developed to control ectoparasites on warm-blooded animals.
- compositions that are bioavailable, can provide contact or systemic activity, are potently efficacious, have a quick onset of activity, have a long duration of activity, and/or are safe to the animal recipients and/or their human owners.
- This invention addresses this need.
- this invention is generally directed to isoxazoline compositions (particularly 4-(isoxazolinyl)-benzamide compositions (also known as, for example, “4- (4,5-dihydroisoxazole-3-yl)-benzoic acid amide” compositions) and 4-(isoxazolinyl)- benzothioamide compositions (also known as, for example, "4-(4,5-dihydroisoxazole-3- yl)-benzothioamide” compositions)), and their use to control ectoparasites in or on warmblooded animals.
- these compositions generally show desirable bioavailability, and can provide contact and/or systemic activity.
- compositions also provide desirable safety profiles toward the warm-blooded animal recipients and/or their owners.
- a single administration of such compositions generally provides potent activity against one or more ectoparasites, while also tending to provide fast onset of activity, long duration of activity, and/or desirable safety profiles.
- This invention is directed, in part, to a method for controlling ectoparasitic infestation of an animal.
- the method comprises administering isoxazoline, a salt of the isoxazoline, or a solvate of the isoxazoline or salt to the animal.
- the isoxazoline corresponds in structure to Formula(I):
- One of A and A is selected from the group consisting of halogen and halomethyl.
- One of A 1 and A 2 is selected from the group consisting of hydrogen, halogen, and halomethyl.
- a 3 is selected from the group consisting of hydrogen, halogen, and halomethyl.
- R is halomethyl.
- X is selected from the group consisting of hydrogen, halogen, methyl, halomethyl, ethyl, and haloethyl.
- Z 1 and Z 2 are independent substituents. In these embodiments:
- Z 1 is independently selected from the group consisting of hydrogen, methyl, haloethyl, halopropyl, halobutyl, methoxymethyl, halomethoxymethyl, ethoxymethyl, haloethoxymethyl, propoxymethyl, ethylaminocarbonylmethyl, ethylaminocarbonylethyl, dimethoxyethyl, propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, haloethylaminocarbonylmethyl, haloethylaminocarbonylethyl, tetrahydrofuryl, methylaminocarbonylmethyl, (N,N-dimethylamino)- carbonylmethyl, propylaminocarbonylmethyl, cyclopropylaminocarbonylmethyl, propenylamino carbonylmethyl, haloethylaminocarbonylcyclopropyl,
- Z 2 is selected from the group consisting of hydrogen, ethyl, methoxymethyl, halomethoxymethyl, ethoxymethyl, haloethoxymethyl, propoxymethyl, methylcarbonyl, ethylcarbonyl, propylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, methoxymethylcarbonyl, aminocarbonyl, ethylaminocarbonylmethyl, ethylaminocarbonylethyl, dimethoxyethyl, propynylaminocarbonylmethyl, haloethylaminocarbonylmethyl, and haloethylaminocarbonylethyl.
- Z 2 is selected from the group consisting of methylcarbonyl, ethylcarbonyl, propylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, methoxymethylcarbonyl, and aminocarbonyl.
- Z 1 and Z 2 together form a substituent selected from the group consisting of:
- Z is selected from the group consisting of O and S.
- Z A is selected from the group consisting of hydrogen, halogen, and cyano.
- This invention also is directed, in part, to a method for treating parasitoses of an animal. This method comprises administering an above-described isoxazoline, salt, or solvate to the animal.
- This invention also is directed, in part, to a method for controlling a ectoparasitic infestation in an environment that is occupied (periodically or continuously) by an animal (e.g., a companion animal, such as a cat or dog).
- This method comprises administering an above-described isoxazoline, salt, or solvate to the animal.
- This invention also is directed, in part, to a use of an above-described isoxazoline, salt, or solvate as a medicament.
- This invention also is directed, in part, to a use of an above-described isoxazoline, salt, or solvate to make a medicament for the treatment of parasitoses of an animal.
- This invention also is directed, in part, an above-described isoxazoline, salt, or solvate to treat parasitoses of an animal.
- This invention also is directed, in part, to a parasiticidal composition for use with an animal.
- the composition comprises an above-described isoxazoline, salt, or solvate in an amount that is effective to control an ectoparasitic infestation when the composition is administered to the animal.
- the composition also comprises an excipient (i.e., it comprises at least one excipient).
- This invention also is directed, in part, to a therapeutic kit.
- the kit comprises an above-described isoxazoline, salt, or solvate and an additional component.
- the additional component may be, for example, a diagnostic tool, instructions for administration, an apparatus for administration, an excipient or other active ingredient, or a memory aid.
- Figure 1 shows the mean plasma concentration of Compound 11-1 during the study in Example 6, which assesses the efficacy of Compound 11-1 against cat fleas (Ctenocephalides felis) and brown dog ticks (Rhipicephalus sanguineus) in dogs.
- cat fleas Ctenocephalides felis
- brown dog ticks Rhipicephalus sanguineus
- the "PO” data refers to Group A (I mg/kg body weight Compound 11-1 in the form of a tablet for oral administration); the “SC” data refers to Group B (1 mg/kg body weight Compound 11-1 in the form of an injectable solution for subcutaneous administration); the "TOP W/ ENH” data refers to Group C (I mg/kg body weight Compound 11-1 in the form of a topical spot-on solution containing an absorption enhancer); the "TOP W/ ENH & SPREAD” data refers to Group D (1 mg/kg body weight Compound 11-1 in the form of a topical spot-on solution containing an absorption enhancer and spreading agent); and the "TOP W/ ETHYL LACTATE” data refers to Group E (I mg/kg body weight Compound 11-1 in the form of a topical spot-on solution containing ethyl lactate as a solvent).
- the isoxazolines used in accordance with this invention generally include compounds of Formula (I):
- Preferred substituents in Formula (I) include the following:
- a 1 and A 2 are selected from the group consisting of halogen and halomethyl.
- the other of A 1 and A 2 is selected from the group consisting of hydrogen, halogen, and halomethyl.
- a 1 is halogen. In some such embodiments, A 1 is bromo. In other embodiments, A 1 is chloro.
- a 1 is halomethyl. In some such embodiments, A 1 is trifluoromethyl.
- a 2 is hydrogen
- a 2 is halogen. In some such embodiments, A 2 is fluoro. In other embodiments, A 2 is chloro.
- a 3 is selected from the group consisting of hydrogen, halogen, and halomethyl. [32] In some embodiments, A 3 is hydrogen.
- a 3 is halogen. In some such embodiments, A 3 is chloro. In other embodiments, A 3 is bromo. [34] In some embodiments, A 3 is halomethyl. In some such embodiments, A 3 is trifluoromethyl.
- R is halomethyl. In some embodiments, R is monochloromethyl. In other embodiments, R is trifluoromethyl. In still other embodiments, R is monochloro-difluoro- methyl.
- X is selected from the group consisting of hydrogen, halogen, methyl, halomethyl, ethyl, and haloethyl.
- X is hydrogen.
- X is bromo.
- X is iodo.
- X is chloro.
- X is methyl.
- X is ethyl.
- X is trifluoromethyl.
- Z 1 and Z 2 are independent substituents.
- Z 1 is selected from the group consisting of hydrogen, methyl, haloethyl, halopropyl, halobutyl, methoxymethyl, halomethoxymethyl, ethoxymethyl, haloethoxymethyl, propoxymethyl, ethylaminocarbonylmethyl, ethylaminocarbonylethyl, dimethoxyethyl, propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, haloethylaminocarbonylmethyl, haloethylaminocarbonylethyl, tetrahydrofuryl, methylaminocarbonylmethyl, (N,N-dimethylamino)-carbonylmethyl, propylamino carbonylmethyl, cyclopropylaminocarbonylmethyl, propenylaminocarbonylmethyl, hal
- Z is selected from the group consisting of hydrogen, methyl, 2,2,2-trifluoroethyl, methoxymethyl, ethoxymethyl, (2,2,2- trifluoroethoxy)-methyl, isopropoxymethyl, ethylaminocarbonylmethyl, 2,2- dimethoxyethyl, 2-propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, (2,2,2- trifluoroethyl)-aminocarbonylmethyl, l-[(2,2,2-trifluoroethyl)-aminocarbonyl]ethyl, tetrahydrofuryl, methylamino carbonylmethyl, (N,N-dimethylamino)-carbonylmethyl, isopropylaminocarbonylmethyl, cyclopropylamino carbonylmethyl, (2-propenyl)- aminocarbonylmethyl, (2,2,2-trifluoroeth
- Z 1 is selected from the group consisting of hydrogen, methyl, haloethyl, halopropyl, halobutyl, methoxymethyl, halomethoxymethyl, ethoxymethyl, haloethoxymethyl, propoxymethyl, ethylaminocarbonylmethyl, ethylaminocarbonylethyl, dimethoxyethyl, propynylaminocarbonylmethyl, N-phenyl-N- methyl-amino, haloethylamino carbonylmethyl, haloethylaminocarbonylethyl, tetrahydrofuryl, methylamino carbonylmethyl, (N,N-dimethylamino)-carbonylmethyl, propylamino carbonylmethyl, cyclopropylaminocarbonylmethyl, propenylaminocarbonylmethyl, haloethylaminocarbonylcyclopropyl
- Z 1 is selected from the group consisting of hydrogen, methyl, 2,2,2-trifluoroethyl, methoxymethyl, ethoxymethyl, (2,2,2- trifluoroethoxy)-methyl, isopropoxymethyl, ethylaminocarbonylmethyl, 2,2- dimethoxyethyl, 2-propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, (2,2,2- trifluoroethyl)-aminocarbonylmethyl, l-[(2,2,2-trifluoroethyl)-aminocarbonyl]ethyl, tetrahydrofuryl, methylamino carbonylmethyl, (N,N-dimethylamino)-carbonylmethyl, isopropylaminocarbonylmethyl, cy clopropylamino carbonylmethyl, (2-propenyl)- aminocarbonylmethyl, (2,2,2-trifluoro
- Z 1 is selected from the group consisting of hydrogen, methyl, 2,2,2-trifluoroethyl, methoxymethyl, ethoxymethyl, (2,2,2- trifluoroethoxy)-methyl, isopropoxymethyl, ethylaminocarbonylmethyl, 2,2- dimethoxyethyl, 2-propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, (2,2,2- trifluoroethyl)-aminocarbonylmethyl, l-[(2,2,2-trifluoroethyl)-aminocarbonyl]ethyl, tetrahydrofuryl,
- Z 1 is selected from the group consisting of hydrogen, methyl, methoxymethyl, ethoxymethyl, (2,2,2-trifluoroethoxy)-methyl, isopropoxymethyl, ethylaminocarbonylmethyl, 2,2-dimethoxyethyl, 2- propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, (2,2,2-trifluoroethyl)- aminocarbonylmethyl, 1 -[(2,2,2-trifluoroethyl)-aminocarbonyl]ethyl, tetrahydrofuryl,
- Z 1 is selected from the group consisting of hydrogen, methoxymethyl, ethoxymethyl, (2,2,2-trifluoroethoxy)-methyl, isopropoxymethyl, ethylaminocarbonylmethyl, 2-propynylaminocarbonylmethyl, (2,2,2- trifluoroethyl)-aminocarbonylmethyl, tetrahydrofuryl,
- Z 1 is selected from the group consisting of hydrogen, methoxymethyl, ethoxymethyl, (2,2,2-trifluoroethoxy)-methyl, ethylaminocarbonylmethyl, 2,2-dimethoxyethyl, 2-propynylaminocarbonylmethyl, N- phenyl-N-methyl-amino, (2,2,2-trifluoroethyl)-aminocarbonylmethyl, 1 -[(2,2,2- trifluoroethyl)-aminocarbony 1] ethyl, tetrahydrofuryl,
- Z 1 is selected from the group consisting of N- phenyl-N-methyl-amino
- Z 1 is selected from the group consisting of hydrogen, (2,2,2-trifluoroethoxy)-methyl,
- Z 1 is selected from the group consisting of hydrogen, (2,2,2-trifluoroethoxy)-methyl, ethylaminocarbonylmethyl, 2- propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, (2,2,2-trifluoroethyl)- aminocarbonylmethyl, 1 -[(2,2,2-trifluoroethyl)-aminocarbonyl]ethyl, tetrahydrofuryl,
- Z 1 is selected from the group consisting of 1 -[(2,2,2- trifluoroethyl)-aminocarbonyl] -ethyl, methylamino carbonylmethyl, (N,N-dimethylamino)- carbonylmethyl, isopropylaminocarbonylmethyl, cyclopropylaminocarbonylmethyl, (2- propenyl)-aminocarbonylmethyl, (2,2,2-trifluoroethyl)-aminocarbonylcyclopropyl, (2- fluoroethyl)-aminocarbonylmethyl, (2-chloroethyl)-aminocarbonylmethyl, 1 - (ethylaminocarbonyl)-ethyl,
- Z 1 is selected from the group consisting of ethylaminocarbonylmethyl, 2-propynylaminocarbonylmethyl, (2,2,2-trifluoroethyl)- aminocarbonylmethyl, tetrahydro furyl,
- Z 1 is selected from the group consisting of methoxymethyl, ethoxymethyl, ethylaminocarbonylmethyl, 2- propynylamino carbonylmethyl, (2,2,2-trifluoroethyl)-aminocarbonylmethyl, tetrahydro furyl,
- Z 1 is selected from the group consisting of methoxymethyl, ethoxymethyl, and tetrahydro furyl. [52] In other such embodiments, Z 1 is:
- Z 1 is haloethylaminocarbonylmethyl. In some such embodiments, Z 1 is (2,2,2-trifluoroethyl)-aminocarbonylmethyl.
- Z 2 is selected from the group consisting of methylcarbonyl, ethylcarbonyl, propylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, methoxymethylcarbonyl, and amino carbonyl. In some such embodiments, Z 2 is methoxycarbonyl. In other embodiments, Z 2 is aminocarbonyl.
- Z 2 is selected from the group consisting of hydrogen, ethyl, methoxymethyl, halomethoxymethyl, ethoxymethyl, haloethoxymethyl, propoxymethyl, methylcarbonyl, ethylcarbonyl, propylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, methoxymethylcarbonyl, aminocarbonyl, ethylaminocarbonylmethyl, ethylaminocarbonylethyl, dimethoxyethyl, propynylaminocarbonylmethyl, haloethylaminocarbonylmethyl, and haloethylaminocarbonylethyl.
- Z 2 is selected from the group consisting of hydrogen, ethyl, methoxymethyl, halomethoxymethyl, ethoxymethyl, haloethoxymethyl, propoxymethyl, methylcarbonyl, ethylcarbonyl, propylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, methoxymethylcarbonyl, aminocarbonyl, ethylaminocarbonylmethyl, ethylaminocarbonylethyl, dimethoxyethyl, propynylaminocarbonylmethyl, haloethylaminocarbonylmethyl, and haloethylaminocarbonylethyl.
- Z 2 is hydrogen
- Z 1 is an independent substituent other than hydrogen
- Z 2 is ethyl
- Z 1 when Z 1 is an independent substituent other than hydrogen, Z 2 is methoxymethyl. [60] In some embodiments when Z 1 is an independent substituent other than hydrogen, Z 2 is methylcarbonyl.
- Z 1 is an independent substituent other than hydrogen
- Z 2 is ethylcarbonyl
- Z 2 is isopropylcarbonyl.
- Z 1 when Z 1 is an independent substituent other than hydrogen, Z 2 is cyclopropylcarbonyl. [64] In some embodiments when Z 1 is an independent substituent other than hydrogen, Z 2 is methoxycarbonyl.
- Z 2 is methoxymethylcarbonyl
- Z 2 is selected from the group consisting of hydrogen, methylcarbonyl, and ethylcarbonyl.
- Z 1 and Z 2 together form a single substituent rather than being independent substituents.
- Z 1 and Z 2 together form the following structure:
- the isoxazoline corresponds in structure to:
- Z 1 and Z 2 together form a single substituent rather than be ;iinngg iinnddeeppeennddient substituents. In these embodiments, Z 1 and Z 2 together form the following structure:
- the isoxazoline corresponds in structure to:
- Z 3 is O (i.e., oxygen). In other embodiments, Z 3 is S (i.e., sulfur).
- Z A is selected from the group consisting of hydrogen, halogen, and cyano. In some such embodiments, Z A is hydrogen. In other embodiments, Z A is bromo. In other embodiments, Z A is chloro. In other embodiments, Z A is cyano.
- a 1 and A 3 are independently selected from the group consisting of halogen and halomethyl, and A 2 is hydrogen.
- each of A 1 and A 3 is chloro such that the isoxazoline corresponds in structure to the following formula:
- Z 1 and Z 2 are either independent substituents or together form a single substituent as follows:
- Z 1 is independently selected from the group consisting of hydrogen, methyl, haloethyl, halopropyl, halobutyl, methoxymethyl, halomethoxymethyl, ethoxymethyl, haloethoxymethyl, propoxymethyl, ethylaminocarbonylmethyl, ethylaminocarbonylethyl, dimethoxyethyl, propynylaminocarbonylmethyl, N-phenyl- N-methyl-amino, haloethylaminocarbonylmethyl, haloethylaminocarbonylethyl, tetrahydrofuryl,
- Z 1 is hydrogen
- Z 2 is selected from the group consisting of methylcarbonyl, ethylcarbonyl, propylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, methoxymethylcarbonyl, and amino carbonyl.
- Z 1 is other than hydrogen
- Z 2 is independently selected from the group consisting of hydrogen, ethyl, methoxymethyl, halomethoxymethyl, ethoxymethyl, haloethoxymethyl, propoxymethyl, methylcarbonyl, ethylcarbonyl, propylcarbonyl,
- a 1 is selected from the group consisting of chloro, bromo, and trifluoromethyl.
- a 2 is selected from the group consisting of hydrogen, chloro, and fluoro.
- a 3 is selected from the group consisting of chloro, bromo, and trifluoromethyl .
- R is selected from the group consisting of monochloromethyl, trifluoromethyl, and monochloro-difluoro-methyl.
- X is selected from the group consisting of hydrogen, bromo, iodo, chloro, methyl, ethyl, and trifluoromethyl.
- Z 3 is O.
- Z A is selected from the group consisting of hydrogen, bromo, chloro, and cyano.
- Z 1 and Z 2 are independent substituents such that: Z 1 is selected from the group consisting of hydrogen, methyl, 2,2,2- trifluoroethyl, methoxymethyl, ethoxymethyl, (2,2,2-trifluoroethoxy)- methyl, isopropoxymethyl, ethylaminocarbonylmethyl, 2,2- dimethoxyethyl, 2-propynylaminocarbonylmethyl, N-phenyl-N-methyl- amino, (2,2,2-trifluoroethyl)-aminocarbonylmethyl, 1 -[(2,2,2- trifluoroethyl)-aminocarbonyl] ethyl, tetrahydrofuryl,
- Z 2 is selected from the group consisting of methoxycarbonyl and aminocarbonyl.
- Z 2 is selected from the group consisting of hydrogen, ethyl, methoxymethyl, methylcarbonyl, ethylcarbonyl, isopropylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, and methoxymethylcarbonyl.
- Z 1 and Z 2 together form:
- Z 1 and Z 2 are either independent substituents or together form a single substituent as follows:
- Z 1 is selected from the group consisting of hydrogen, methyl, 2,2,2-trifluoroethyl, methoxymethyl, ethoxymethyl,
- (2,2,2-trifluoroethoxy)-methyl isopropoxymethyl, ethylaminocarbonylmethyl, 2,2-dimethoxyethyl, 2- propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, (2,2,2-trifluoroethyl)-aminocarbonylmethyl, 1 -[(2,2,2- trifluoroethyl)-aminocarbonyl] ethyl, tetrahydrofuryl,
- Z 2 is selected from the group consisting of methoxycarbonyl and amino carbonyl.
- Z 1 is other than hydrogen
- Z 2 is selected from the group consisting of hydrogen, ethyl, methoxymethyl, methylcarbonyl, ethylcarbonyl, isopropylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, and methoxymethy lcarbony 1.
- Z 1 and Z 2 together form a single substituent, they form:
- the isoxazoline of Formula (I) is defined as follows: [87] The isoxazoline corresponds in structure to:
- a 1 is selected from the group consisting of chloro and bromo.
- a 3 is selected from the group consisting of chloro and bromo.
- X is selected from the group consisting of hydrogen, bromo, iodo, chloro, methyl, ethyl, and trifluoromethyl.
- Z 1 and Z 2 are independent substituents such that:
- Z 1 is selected from the group consisting of hydrogen, methyl, 2,2,2- trifluoroethyl, methoxymethyl, ethoxymethyl, (2,2,2-trifluoroethoxy)- methyl, isopropoxymethyl, ethylaminocarbonylmethyl, 2,2- dimethoxyethyl, 2-propynylaminocarbonylmethyl, N-phenyl-N-methyl- amino, (2,2,2-trifluoroethyl)-aminocarbonylmethyl, 1 -[(2,2,2- trifluoroethyl)-aminocarbony 1] ethyl, tetrahydrofuryl,
- Z 2 is selected from the group consisting of methoxycarbonyl and aminocarbonyl.
- Z 1 is other than hydrogen
- Z 2 is selected from the group consisting of hydrogen, ethyl, methoxymethyl, methylcarbonyl, ethylcarbonyl, isopropylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, and methoxymethylcarbonyl.
- Z 1 and Z 2 together form:
- substituents of Formula (I) are defined as follows: [95] A 1 and A 3 are independently is selected from the group consisting of chloro, bromo, and trifluoromethyl.
- a 2 is selected from the group consisting of hydrogen, chloro, and fluoro.
- R is selected from the group consisting of trifluoromethyl and monochloro- difluoro-methyl.
- X is selected from the group consisting of hydrogen, bromo, iodo, chloro, methyl, and trifluoromethyl.
- Z A is selected from the group consisting of bromo, chloro, and cyano.
- Z 1 and Z 2 are independent substituents such that:
- Z 1 is selected from the group consisting of hydrogen, methyl, methoxymethyl, ethoxymethyl, (2,2,2-trifluoroethoxy)-methyl, isopropoxymethyl, ethylaminocarbonylmethyl, 2,2-dimethoxyethyl, 2- propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, (2,2,2- trifluoroethyl)-aminocarbonylmethyl, 1 -[(2,2,2-trifluoroethyl)- aminocarbonyl] ethyl, tetrahydrofuryl,
- Z 1 When Z 1 is hydrogen, Z 2 is aminocarbonyl.
- Z 1 When Z 1 is other than hydrogen, Z 2 is selected from the group consisting of hydrogen, ethyl, methoxymethyl, methylcarbonyl, ethylcarbonyl, isopropylcarbonyl, cyclopropylcarbonyl, methoxycarbonyl, and methoxymethylcarbonyl.
- Z 1 When Z 1 is hydrogen, Z 2 is amino carbonyl. [102] In other such embodiments, Z 1 and Z 2 together form:
- isoxazolines of these embodiments tend to exhibit particularly beneficial in vivo flea inhibition within 24 and 48 hours after infestation. See, e.g., Example 3 below.
- the isoxazoline of Formula (I) is defined as follows:
- the isoxazoline corresponds in structure to:
- X is selected from the group consisting of chloro and methyl.
- Z 1 is selected from the group consisting of N-phenyl-N-methyl-amino,
- Z is selected from the group consisting of hydrogen, ethylcarbonyl, methoxycarbonyl, and methoxymethylcarbonyl.
- isoxazolines of these embodiments tend to exhibit particularly beneficial in vivo flea inhibition within 24 and 48 hours after infestation. See, e.g., Example 3 below.
- isoxazo lines encompassed by these embodiments include the following:
- the isoxazoline corresponds in structure to:
- R is selected from the group consisting of trifluoromethyl and monochloro- difluoro-methyl.
- X is selected from the group consisting of iodo and methyl.
- Z 1 is selected from the group consisting of hydrogen, methoxymethyl, ethoxymethyl, (2,2,2-trifluoroethoxy)-methyl, ethylaminocarbonylmethyl, 2,2- dimethoxyethyl, 2-propynylaminocarbonylmethyl, N-phenyl-N-methyl-amino, (2,2,2- trifluoroethyl)-aminocarbonylmethyl, l-[(2,2,2-trifluoroethyl)-aminocarbonyl]ethyl, tetrahydrofuryl,
- Z 2 is selected from the group consisting of hydrogen, methylcarbonyl, ethylcarbonyl, and methoxycarbonyl.
- isoxazo lines of these embodiments tend to exhibit particularly beneficial in vivo results with respect to flea inhibition within 24 and 48 hours after infestation, and ticks for at least 8 days. See, e.g., Examples 3 and 4 below.
- the isoxazoline corresponds in structure to:
- X is selected from the group consisting of iodo and methyl.
- Z 1 is selected from the group consisting of hydrogen, (2,2,2- trifluoroethoxy)-methyl, ethylaminocarbonylmethyl, 2-propynylaminocarbonylmethyl, N- phenyl-N-methyl-amino, (2,2,2-trifluoroethyl)-aminocarbonylmethyl, 1 -[(2,2,2- trifluoroethyl)-aminocarbonyl] ethyl, tetrahydrofuryl,
- Z 1 is hydrogen
- Z 2 is aminocarbonyl
- Z 2 is selected from the group consisting of hydrogen and ethylcarbonyl.
- isoxazo lines of these embodiments also tend to exhibit particularly beneficial in vivo results with respect to both flea inhibition within 24 and 48 hours, and ticks for at least 8 days. See, e.g., Examples 3 and 4 below. HlO. Illustrative Substituent Combination #10 [128]
- the isoxazoline of Formula (I) is defined as follows:
- the isoxazoline corresponds in structure to:
- X is selected from the group consisting of iodo and methyl.
- Z 1 is selected from the group consisting of hydrogen, (2,2,2- trifluoroethoxy)-methyl,
- Z 1 is hydrogen
- Z 2 is aminocarbonyl
- Z 2 is selected from the group consisting of hydrogen, methylcarbonyl, and ethylcarbonyl.
- isoxazo lines of these embodiments tend to exhibit particularly beneficial in vivo results with respect to flea inhibition within 24 and 48 hours after infestation, and ticks for at least 8 days. See, e.g., Examples 3 and 4 below.
- the isoxazoline corresponds in structure to:
- Z 1 is selected from the group consisting of methoxymethyl, ethoxymethyl, and tetrahydro furyl.
- Z 2 is selected from the group consisting of hydrogen and methoxycarbonyl.
- the isoxazoline corresponds in structure to:
- Z 1 is selected from the group consisting of ethylaminocarbonylmethyl, 2- propynylaminocarbonylmethyl, (2,2,2-trifluoroethyl)-aminocarbonylmethyl, tetrahydrofuryl,
- isoxazo lines of these embodiments tend to exhibit particularly beneficial in vivo results with respect to flea inhibition within 1, 24, and 48 hours after infestation, and ticks for at least 8 days. See, e.g., Examples 3 and 4 below.
- isoxazo lines encompassed by these embodiments include the following:
- This isoxazo line is 4-[5-(3,5-dichlorophenyl)-5-trifluoromethyl-4,5- dihydroisoxazol-3-yl]-2-methyl- ⁇ /-[(2,2,2-trifluoro-ethylcarbamoyl)-methyl]-benzamide. It can be found in, for example, CAS RN [864731-61-3]. It has been discovered in accordance with this invention that Compound 11-1 exhibits particularly beneficial in vivo results with respect to duration of flea inhibition using one of various routes of administration, including topical, oral, or subcutaneous. See, e.g., Example 5 below.
- R is selected from the group consisting of monochloromethyl and trifluoromethyl.
- X is selected from the group consisting of iodo and methyl.
- Z 1 is selected from the group consisting of hydrogen, methoxymethyl, ethoxymethyl, (2,2,2-trifluoroethoxy)-methyl, isopropoxymethyl, ethylaminocarbonylmethyl, 2-propynylaminocarbonylmethyl, (2,2,2-trifluoroethyl)- aminocarbonylmethyl, tetrahydro furyl,
- Z 1 is hydrogen
- Z 2 is aminocarbonyl
- Z 1 is other than hydrogen
- Z 2 is selected from the group consisting of hydrogen, methylcarbonyl, ethylcarbonyl, and methoxycarbonyl, except that: Hl 4.
- Illustrative Substituent Combination #14 [156] In some embodiments, the isoxazoline of Formula (I) is defined as follows:
- the isoxazoline corresponds in structure to:
- Z 1 is selected from the group consisting of methoxymethyl, ethoxymethyl, ethylaminocarbonylmethyl, 2-propynylaminocarbonylmethyl, (2,2,2-trifluoroethyl)- aminocarbonylmethyl, tetrahydro furyl,
- [159] Z is selected from the group consisting of hydrogen and methoxycarbonyl.
- the isoxazoline corresponds in structure to:
- Z 1 is selected from the group consisting of ethylaminocarbonylmethyl, 2- propynylaminocarbonylmethyl, (2,2,2-trifluoroethyl)-aminocarbonylmethyl, tetrahydrofuryl,
- isoxazolines of these embodiments tend to exhibit particularly beneficial in vivo results with respect to fleas inhibition within 1, 24, and 48 hours after infestation. See, e.g., Example 3.
- the isoxazoline corresponds in structure to:
- Z 1 and Z 2 are either independent substituents or together form a single substituents.
- Z 1 is selected from the group consisting of l-[(2,2,2-trifluoroethyl)- aminocarbonyl] -ethyl, methylaminocarbonylmethyl, (N,N-dimethylamino)- carbonylmethyl, isopropylaminocarbonylmethyl, cyclopropylaminocarbonylmethyl, (2-propenyl)-aminocarbonylmethyl, (2,2,2-trifluoroethyl)-aminocarbonylcyclopropyl, (2-fluoroethyl)- aminocarbonylmethyl, (2-chloroethyl)-aminocarbonylmethyl, 1- (ethylaminocarbonyl)-ethyl,
- Z 2 is selected from the group consisting of hydrogen, methylcarbonyl, and ethylcarbonyl.
- the isoxazoline corresponds in structure to:
- the isoxazoline corresponds in structure to:
- the isoxazo lines used in this invention generally can have two or more conformational structures. At minimum, for example, all the isoxazo lines comprise a chiral (or asymmetric) carbon at the 5-position of the isoxazo line ring. In some embodiments, for example, the chiral carbon has a left-handed (or "S" or "sinister”) configuration.
- Such isoxazo lines include those having the following structure:
- the isoxazo lines correspond in structure to:
- the chiral carbon has a right-handed (or "R” or “rectus”) configuration.
- isoxazo lines include those corresponding to the following structure:
- the isoxazo lines may additionally have other conformational isomers, such as, for example, substituents with a cis or trans double bond.
- a specific isomer often can be isolated from the corresponding racemic mixture (or a salt thereof) using, for example, chiral high performance liquid chromatography (HPLC) techniques. Such a technique is illustrated in Example 7 below for isolating the R and S enantiomers of racemic Compound 11-1.
- HPLC high performance liquid chromatography
- a more-easily-isolatable derivative of the isomer is isolated from the corresponding derivative racemic mixture (or a salt thereof), and then converted to the isomer.
- a specific isomer often can be directly synthesized from, for example, an optically pure starting material.
- the ratio of one enantiomer (e.g., Compound 17-1) to another enantiomer (e.g., Compound 11-lR) in the pharmaceutical composition used with this invention is greater than 1 :1. In some instances, for example, the ratio is greater than about 70:30, greater than about 85:15, greater than about 90:10, greater than about 95:5, greater than about 98:2, or greater than about 99:1.
- the concentration of one enantiomer e.g.,
- Compound 17-1) in the composition is greater than about 50% (by weight).
- the concentration is greater than about 70% (by weight), greater than about 85% (by weight), greater than about 90% (by weight), greater than about 95% (by weight), greater than about 98% (by weight), greater than about 99% (by weight), or greater than about 99.5% (by weight).
- a isoxazoline structure that does not indicate a particular conformation is intended to encompass compositions of all the possible conformational isomers of the isoxazoline, as well as compositions comprising fewer than all (e.g., just one of) the possible conformational isomers.
- isoxazolines used with this invention may be in the form of a salt.
- a salt may be advantageous due to one or more of its physical properties, such as pharmaceutical stability in differing temperatures and humidities; crystalline properties; and/or a desirable solubility in water, oil, or other solvents.
- Acid and base salts typically can be formed by, for example, mixing a compound with an acid or base, respectively, using various known methods in the art.
- the salt when the salt is intended to be administered in vivo (i.e., to an animal) for a therapeutic benefit, the salt preferably is pharmaceutically acceptable.
- a base addition salt of a isoxazoline of Formula (I) can be prepared by reacting the isoxazoline with an approximately stoichiometric amount of an inorganic or organic base, typically a strong inorganic or organic base.
- base addition salts may include, for example, metallic salts, and organic salts.
- Metallic salts include alkali metal (group Ia, e.g., lithium, sodium, or potassium) salts, alkaline earth metal (group Ha, e.g., barium, calcium, and magnesium) salts, heavy metal (e.g., zinc and iron) salts, and other physiologically acceptable metal salts.
- Such salts may be made from calcium, lithium, magnesium, potassium, sodium, and zinc.
- a free acid isoxazoline may be mixed with sodium hydroxide to form such a base addition salt.
- an acid addition salt of a isoxazoline of Formula (I) can be prepared by reacting the isoxazoline with an approximately stoichiometric amount of an inorganic or organic acid.
- contemplated inorganic acids for making pharmaceutically acceptable salts include hydrochloric, hydrobromic, hydroiodic, nitric, carbonic, sulfuric, and phosphoric acid.
- organic acids for making pharmaceutically acceptable salts generally include, for example, aliphatic, cycloaliphatic, aromatic, araliphatic, heterocyclic, carboxylic, and sulfonic classes of organic acids.
- organic acids include cholic, sorbic, lauric, acetic, trifluoroacetic, formic, propionic, succinic, gly colic, gluconic, digluconic, lactic, malic, tartaric acid, citric, ascorbic, glucuronic, maleic, fumaric, pyruvic, aspartic, glutamic, aryl carboxylic acid (e.g., benzoic), anthranilic acid, mesylic, stearic, salicylic, p- hydroxybenzoic, phenylacetic, mandelic, embonic (pamoic), alkylsulfonic (e.g., ethanesulfonic), arylsulfonic (e.g., benzenesulfonic), pantothenic, 2- hydroxyethanesulfonic, sulfanilic, cyclohexylaminosulfonic, ⁇ -hydroxybutyric, galactaric
- an organic salt of a isoxazoline of Formula (I) may be made by, for example, quaternizing a basic nitrogen-containing group on the isoxazoline with an agent such as a Ci-C ⁇ -alkyl halide (e.g., methyl, ethyl, propyl, and butyl chlorides, bromides, or iodide), dialkyl sulfate (e.g., dimethyl, diethyl, dibuytl, or diamyl sulfate), long chain halide (e.g., decyl, lauryl, myristyl, and stearyl chlorides, bromides, and iodide), arylalkyl halide (e.g., benzyl and phenethyl bromide), and the like.
- an agent such as a Ci-C ⁇ -alkyl halide (e.g., methyl, ethyl, propyl, and but
- an acid or base salt may, in some instances, be optically active (e.g., D-lactate and L-lysine salts) or racemic (e.g., DL- tartrate and DL-arginine salts).
- optically active e.g., D-lactate and L-lysine salts
- racemic e.g., DL- tartrate and DL-arginine salts
- the isoxazolines of Formula (I) are in the form of stable complexes with solvent molecules that remain intact after the non-complexed solvent molecules are removed from the compounds. These complexes generally are referred to as "solvates.” In some instances, the solvate will be capable of isolation, for example when one or more solvent molecules are incorporated into the crystal lattice of the crystalline solid.
- a “solvate” encompasses both solution-phase and isolatable solvates. Examples of suitable solvates include ethanolates, methanolates, and the like.
- a "hydrate” is a solvate wherein the solvent molecule is water.
- a solvate intended to be used in vivo preferably is pharmaceutically acceptable.
- prodrugs of the isoxazolines of Formula (I) may be used with methods of this invention.
- the term "prodrug” is a compound that is convertible in use (e.g., in vivo) by metabolic means or another processes (e.g., hydrolysis) to a isoxazoline of Formula (I).
- delivery of the isoxazoline in a prodrug form achieves improved delivery of the isoxazoline by improving its physicochemical or pharmacokinetic properties by, for example, enhancing systemic absorption, delaying clearance, or delaying breakdown in vivo.
- US2007/0066617 discusses the preparation of Compound 11-1 at Example 21 on page 72, the preparation of Compound 5-19 at Example 28 on pages 74-75, the preparation of Compound 5-61 at Example 30 on pages 76-77, and the preparation of Compound 5-64 at Example 34 on pages 77-78;
- Int'l Patent Publ. No. WO2007/026965 discusses the preparation of Compound 5-16 at Example 21 on page 317, the preparation of Compound 5-25 at Example 22 on page 317-318, the preparation of Compound 5-49 at Example 12 on page 312, the preparation of Compound 5-60 at Example 11 on page 311-312, and the preparation of Compound 10-1 at Example 32 on page 321-322; and Japanese Patent Appl. Publ. No.
- JP2007/308471 discusses the preparation of Compound 5-45 at Example 38 on page 381.
- methods for preparing various isoxazo lines also are discussed in, for example, Int'l Patent Publ. No. WO 2005/085216 (incorporated by reference into this patent), Int'l Patent Appl. No.
- the isoxazo lines of Formula (I) generally may be used to control ectoparasites on animals, and, in turn, diseases directly caused by such ectoparasites and/or diseases caused by pathogens carried by such ectoparasites. It is contemplated that the composition may be used to treat a range of animals, especially warm-blooded animals. Such warm-blooded animals include, for example, mammals. Mammals include, for example, humans.
- Other mammals include, for example, farm or livestock mammals ⁇ e.g., swine, bovines, sheep, goats, etc.), laboratory mammals ⁇ e.g., mice, rats, jirds, etc.), companion mammals ⁇ e.g., dogs, cats, equines, etc.), fur-bearing animals ⁇ e.g., minks, foxes, chinchillas, rabbits, etc.), and wild and zoo mammals ⁇ e.g., buffalo, deer, etc.).
- farm or livestock mammals ⁇ e.g., swine, bovines, sheep, goats, etc.
- laboratory mammals e.g., mice, rats, jirds, etc.
- companion mammals e.g., dogs, cats, equines, etc.
- fur-bearing animals e.g., minks, foxes, chinchillas, rabbits, etc.
- wild and zoo mammals
- the compositions are used to treat canines ⁇ e.g., dogs, such as, for example, pure-bred and/or mongrel companion dogs, show dogs, working dogs, herding dogs, hunting dogs, guard dogs, police dogs, racing dogs, and/or laboratory dogs).
- the compositions are used to treat felines ⁇ e.g., domestic cats). It is contemplated that the compositions also are suitable to treat non-mammals, such as birds ⁇ e.g., turkeys, chickens, geese, ducks, parrots, etc.). It is also contemplated that such compositions may be useful to treat cold-blooded animals as well, such as, for example, fish (e.g., salmon, trout, koi, etc.).
- fish e.g., salmon, trout, koi, etc.
- the isoxazo lines of Formula (I) are generally of particular value for controlling ectoparasites, i.e., arthropods that are injurious to, or spread or act as vectors of diseases in, warmblooded animals.
- the isoxazo lines are generally beneficial for controlling various lifecycle stages of parasites, including egg, nymph, larvae, juvenile, and adult stages.
- Ectoparasites generally insect and acarid pests
- biting insects include, for example, migrating diperous larvae, such as, for example, Hypoderma sp. in cattle, Gastrophilus in horses, and Cuter ebra sp. in rodents; biting flies, such as, for example, bloodsucking adult flies (e.g., the horn fly (Haematobia ir ⁇ tans), horse flies (Tabanus spp.), stable flies (Stomoxys calcitrans), black flies (Simulium spp.), deer flies (Chrysops spp.), louse flies (Melophagus ovinus), tsetse flies (Glossina spp.)); parasitic fly maggots, such as, for example, bot flies (Oestrus ovis and Cuterebra spp.), the blow flies (Phaenicia spp.), screwworms (Cochl
- Astigm ⁇ t ⁇ spp. such as itch or scab mites, which include S ⁇ rcoptid ⁇ e spp. (e.g., S ⁇ rcoptes sc ⁇ biei); and mange mites, which include Psoroptid ⁇ e spp. (e.g., Chorioptes bovis and Psoroptes ovis).
- Prostigm ⁇ t ⁇ spp such as chiggers, which include Trombiculid ⁇ e spp. (e.g.,
- C. Ticks These include, for example, soft-bodied ticks, such as Arg ⁇ sid ⁇ e spp. (e.g., Arg ⁇ s spp. and Ornithodoros spp.); and hard-bodied ticks, such as Ixodid ⁇ e spp. ( e -g- > Ixodes ricinus, Rhipiceph ⁇ lus sanguineus, Haemaphysalis spp, Dermacentor reticulates, Dermacentor variabilis, Amblyomma americanum, and Boophilus spp.).
- D. Lice These include, for example, chewing lice, such as Menopon spp. and Bovicola spp.; and sucking lice, such as Haematopinus spp., Linognathus spp., and Solenopotes spp.
- E. Fleas include, for example, Ctenocephalides spp., such as dog fleas ⁇ Ctenocephalides canis) and cat fleas ⁇ Ctenocephalides felis); Xenopsylla spp., such as oriental rat fleas ⁇ Xenopsylla cheopis); Pulex spp., such as human fleas ⁇ Pulex irritans); hedgehog fleas ⁇ Archaeopsylla erinacei); and bird fleas ⁇ Ceratophyllus gallinae).
- Ctenocephalides spp. such as dog fleas ⁇ Ctenocephalides canis
- Xenopsylla spp. such as oriental rat fleas ⁇ Xenopsylla cheopis
- Pulex spp. such as human fleas ⁇ Pulex irritans
- F. True bugs include, for example, Cimicidae or the common bed bug ⁇ Cimex lectularius); and Triatominae spp., such as triatomid bugs (also known as kissing bugs) ⁇ e.g., Rhodnius prolixus and Triatoma spp.).
- infestation refers to the presence of parasites in numbers that pose a risk of nuisance or harm to humans or animals.
- the presence can be in the environment ⁇ e.g., in animal bedding), on the skin or fur of an animal, etc.
- infestation is intended to be synonymous with the term, "infection,” as that term is generally understood in the art.
- control of ectoparasite infestation means to reduce or eradicate parasite numbers in and/or on an animal, and/or to partially or completely inhibit the development of parasite infestation in and/or on an animal. This may be achieved by, for example, killing, repelling, expelling, incapacitating, deterring, eliminating, alleviating, or minimizing the parasite.
- the control of ectoparasites can be insecticidal and/or acaricidal.
- the effect of the isoxazoline can be, for example, ovicidal, larvicidal, nymphicidal, adulticidal, or a combination thereof.
- the effect can manifest itself directly by killing the parasites either immediately or after some time has elapsed ⁇ e.g., when molting occurs or by destroying eggs).
- the effect alternatively (or additionally) can manifest itself indirectly by, for example, reducing the number of eggs laid and/or the hatch rate.
- an amount of a isoxazoline that is sufficient to "control" or be “effective” against a target parasite is an amount that is sufficient to reduce or eradicate parasite numbers in and/or on an animal, and/or to partially or completely inhibit the development of parasite infestation in and/or on an animal.
- an effective amount generally constitutes an amount that results in tissue and/or blood concentrations generally toxic when ingested by a target parasite.
- a dose is considered effective for controlling a target parasite when the dose is sufficient to cause an existing or potential target parasite count to be reduced by at least about 5%.
- the dose is considered effective when the dose is sufficient to cause an existing or potential parasite count to be reduced by at least about 10% (or at least about 30%, at least about 50%, at least about 60%, at least about 75%, at least about 90%, at least about 95%, or at least about 99%).
- the optimum dosage generally depends on multiple factors, including, for example, the particular isoxazoline; the identity of any other active ingredient(s) being administered to the animal recipient; the route of administration; the type and severity of the target condition and pathogen; the type (e.g., species and breed), age, size, sex, diet, activity, and condition of the intended animal recipient; and pharmacological considerations, such as the activity, efficacy, pharmacokinetic, and toxicology profiles of the isoxazoline and other active ingredient(s) being administered to the recipient animal.
- the amount of each ingredient that constitutes an "effective amount" is an amount that, when combined with the other active ingredients, causes the desired effect.
- the isoxazo lines of Formula (I) may be administered multiple times for a single treatment. Such multiple-dosage treatments are contemplated to include multiple doses per day for one or more days, daily doses for multiple days, and/or doses administered two or more days apart. In some embodiments of this invention, however, a single dose is administered to effectively control a target parasite for a longer duration, such as, for example, at least about one week. In some such embodiments, for example, the single dose is effective to control a target parasite for at least about 2 weeks, at least about 3 weeks, at least about 1 month, at least about 2 months, at least about 3 months, at least about 4 months, or at least about 6 months.
- the frequency of treatments may be, for example, weekly, bi-weekly, monthly, bi-monthly, quarterly, half yearly, or even longer (e.g., yearly).
- the durations over which the isoxazo lines of Formula (I) tend to be effective against various ectoparasites by systemic administration is surprising. This is particularly true, given that such long activities may, in many instances, be obtained using low doses that are non-toxic to the animal recipients without requiring the use of a controlled-release means. Without being limited to any particular theory, it is hypothesized that this long duration of activity stems from the isoxazo lines having particularly high toxicity when ingested by the target parasite.
- the isoxazoline dose and formulation are chosen to maintain a isoxazoline serum level of at least about 1 ng/ml (e.g., 1 to 50 ng/ml).
- the amount of isoxazoline administered to the animal recipient is from about 0.001 to about 200 mg/kg body weight. In some embodiments, for example, from about 0.01 to about 200 mg/kg body weight is administered. In other embodiments, for example, from about 0.001 to about 100 mg/kg body weight is administered. In some such embodiments, for example, from about 0.01 to about 100 mg/kg body weight is administered. In other such embodiments, from about 1 to about 30 mg/kg body weight is administered. Greater dosages tend to provide for greater duration of activity.
- the duration of activity of a isoxazoline can be extended even further (or made more consistent) by using a controlled-release formulation or dosage form.
- the isoxazoline can be administered in microspheres, granules, or implants (e.g., a subcutaneous implant) that release the isoxazoline by, example, diffusion and/or erosion.
- Use of such a dosage form containing from about 1 and about 50 mg/kg body weight (or from about 10 to about 30 mg/kg body weight, such as about 20 mg/kg of body weight) of the isoxazoline may allow for consistent activity lasting over several months or longer (e.g., a year).
- a isoxazoline of Formula (I) is administered to treat parasitoses of an animal (or make a medicament to treat parasitoses of an animal).
- parasitoses includes pathologic conditions and diseases associated with or caused by one or more ectoparasites directly, such as, for example, anemia and flea allergy dermatitis. It also includes pathologic conditions or diseases associated with caused by one or more vector-transmitted pathogens, such as, for example, Lyme disease, ehrlichiosis (particularly canine ehrlichiosis), and Rocky Mountain spotted fever from vector ticks.
- treatment of parasitoses means to partially or completely inhibit the development of parasitoses of an animal susceptible to parasitoses, reduce or completely eliminate the symptoms of parasitoses of an animal having parasitoses, and/or partially or completely cure parasitoses of an animal having parasitoses.
- the treatment of parasitoses is achieved by administering a isoxazoline of Formula (I) to control an ectoparasite infestation.
- This invention also relates to treatment methods wherein at least an ancillary goal of controlling ectoparasites in and/or on an animal is to control an ectoparasitic infestation in an environment that is occupied (periodically or continuously) by the animal.
- the animal is a companion animal (e.g., a cat or dog).
- the environment may be, for example, a house or other shelter; a room; a pen, a stall, or other confinement means; bedding; etc.
- administer refers to the delivery of a isoxazoline of Formula (I), salt of the isoxazoline, solvate of the isoxazoline or salt, or prodrug of the isoxazoline.
- systemic administration is desirable.
- Systemic administration is an administration at a site remote from a site wherein at least a portion of the target parasites reside. With systemic administration, at least a portion of the isoxazoline reaches the target parasite via the animal recipient's bloodstream, other body fluids (lymph fluids), and/or tissues (e.g., skin or fat tissue).
- the parasite ingests the isoxazoline along with the animal recipient's blood, other body fluids, and/or tissue.
- Systemic administration may be achieved in several forms.
- the isoxazoline composition is systemically administered via an oral route in a unit dosage form, such as, for example, a soft or hard capsule, a pill, a powder, granules, a tablet (e.g., a chewable tablet), a paste, a solution, a suspension (aqueous or non-aqueous), an emulsion (oil-in-water or water-in-oil), an elixir, a syrup, a bolus, a drench, or via the animal recipient's feed or drinking water.
- a unit dosage form such as, for example, a soft or hard capsule, a pill, a powder, granules, a tablet (e.g., a chewable tablet), a paste, a solution, a suspension (aqueous or non
- composition When the composition is administered via an animal's feed, it may, for example, be fed as a discrete feed or as a chewable treat. Alternatively (or additionally), it may, for example, be intimately dispersed in the animal recipient's regular feed, used as a top dressing, or in the form of pellets or liquid that is added to the finished feed.
- a feed additive When the composition is administered as a feed additive, it may be convenient to prepare a "premix" in which the composition is dispersed in a liquid or solid carrier. This "premix" is, in turn, dispersed in the animal's feed using, for example, a conventional mixer.
- a solution or suspension formulation This formulation can be, for example, a concentrated suspension that is mixed with water or a dry preparation that is mixed and suspended in the water. In both instances, it is preferable to have the isoxazoline in a finely-pulverized form.
- the isoxazoline composition alternatively (or additionally) may be systemically administered topically using a transdermal formulation (i.e., a formulation that passes through the skin). Alternatively (or additionally), the composition may be systemically administered topically via the mucosa.
- Typical formulations for transdermal and mucosal administration include, for example, pour-ons, spot-ons, dips, sprays, mousses, shampoos, powders, gels, hydrogels, lotions, solutions, creams, ointments, dusting powders, dressings, foams, films, skin patches, limb bands, collars, ear tags, wafers, sponges, fibers, bandages, and microemulsions.
- the pour-on or spot-on methods comprise applying the isoxazoline composition to a specific location of the skin or coat, such as on the neck or backbone of the animal. This may be achieved by, for example, applying a swab or drop of the pour-on or spot-on formulation to a relatively small area of the recipient animal's skin or coat (i.e., generally no greater than about 10% of the animal recipient's skin or coat).
- the isoxazoline is dispersed from the application site to wide areas of the fur due to the spreading nature of the components in the formulation and the animal's movements while, in parallel, being absorbed through the skin and distributed via the animal recipient's fluids and/or tissues.
- the isoxazoline composition alternatively (or additionally) may be systemically administered parenterally, such as via intramuscular injection, intravenous injection, subcutaneous injection, implant (e.g., subcutaneous implant), infusion, bolus, etc.
- the parenteral dosage form provides the animal recipient with from about 0.01 to about 200 mg/kg body weight of the isoxazoline.
- Other contemplated modes of administration include, for example, rectal, vaginal, and via inhalation (e.g., via a mist or aerosol).
- compositions comprising a isoxazoline of Formula (I), salt of the isoxazoline, solvate of the isoxazoline or salt, or prodrug of the isoxazoline.
- the compositions also may (and generally will) comprise one or more pharmaceutically-acceptable excipients.
- Pharmaceutical compositions of the present invention may be manufactured by, for example, processes known in the art. These processes include, for example, a variety of known mixing, dissolving, granulating, emulsifying, encapsulating, entrapping, and lyophilizing processes. Optimal formulation depends on, for example, the route of administration.
- Solid dosage forms may be prepared by, for example, intimately and uniformly mixing the isoxazoline with fillers, binders, lubricants, glidants, disintegrants, flavoring agents (e.g., sweeteners), buffers, preservatives, pharmaceutical- grade dyes or pigments, and controlled release agents.
- Oral dosage forms other than solids may be prepared by mixing the isoxazoline with, for example, one or more solvents, viscosity-enhancing agents, surfactants, preservatives, stabilizers, resins, fillers, binders, lubricants, glidants, disintegrants, co-solvents, sweeteners, flavorings, perfuming agents, buffers, suspending agents, and pharmaceutical-grade dyes or pigments.
- Contemplated binders include, for example, gelatin, acacia, and carboxymethyl cellulose.
- Contemplated lubricants include, for example, magnesium stearate, stearic acid, and talc.
- Contemplated disintegrants include, for example, corn starch, alginic acid, sodium carboxymethylcellulose, and sodium croscarmellose.
- Contemplated buffers include, for example, sodium citrate, and magnesium and calcium carbonate and bicarbonate.
- Contemplated solvents include, for example, water, petroleum, animal oils, vegetable oils, mineral oil, and synthetic oil.
- Physiological saline solution or glycols e.g., ethylene glycol, propylene glycol, or polyethylene glycol
- the solvent preferably has sufficient chemical properties and quantity to keep the isoxazoline solubilized at temperatures in which the composition is stored and used.
- Contemplated viscosity-enhancing agents include, for example, polyethylene, methylcellulose, sodium carboxymethylcellulose, hydroxypropyl- methylcellulose, hydroxypropylcellulose, sodium alginate, carbomer, povidone, acacia, guar gum, xanthan gum, tragacanth, methylcellulose, carbomer, xanthan gum, guar gum, povidone, sodium carboxymethylcellulose, magnesium aluminum silicate, carboxyvinyl polymers, carrageenan, hydroxyethyl cellulose, laponite, water-soluble salts of cellulose ethers, natural gums, colloidal magnesium aluminum silicateor finely divided silica, homopolymers of acrylic acid crosslinked with an alkyl ether of pentaerythritol or an alkyl ether of sucrose, and carbomers.
- Contemplated surfactants include, for example, polyoxyethylene sorbitan fatty acid esters; polyoxyethylene monoalkyl ethers; sucrose monoesters; lanolin esters and ethers; alkyl sulfate salts; and sodium, potassium, and ammonium salts of fatty acids.
- Contemplated preservatives include, for example, phenol, alkyl esters of parahydroxybenzoic acid (e.g., methyl /?-hydroxybenzoate (or “methylparaben”) and propyl /?-hydroxybenzoate (or “propylparaben”)), sorbic acid, o-phenylphenol benzoic acid and the salts thereof, chlorobutanol, benzyl alcohol, thimerosal, phenylmercuric acetate and nitrate, nitromersol, benzalkonium chloride, and cetylpyridinium chloride.
- parahydroxybenzoic acid e.g., methyl /?-hydroxybenzoate (or "methylparaben” and propyl /?-hydroxybenzoate (or “propylparaben”
- sorbic acid e.g., methyl /?-hydroxybenzoate (or "methylparaben”) and propyl /?-hydroxybenzoate (or “
- Contemplated stabilizers include, for example, chelating agents and antioxidants.
- Solid dosage forms also may comprise, for example, one or more excipients to control the release of the isoxazoline.
- the isoxazoline may be dispersed in, for example, hydroxypropylmethyl cellulose.
- Some oral dosage forms e.g., tablets and pills
- enteric coatings e.g., enteric coatings.
- Topical administration may be achieved using, for example, a concentrated solution, suspension (aqueous or non-aqueous), emulsion (water-in-oil or oil-in-water), or microemulsion comprising a isoxazoline dissolved, suspended, or emulgated in a pharmaceutically-acceptable liquid vehicle.
- a crystallization inhibitor optionally may generally be present.
- a liquid formulation When a liquid formulation is used topically on skin, it can be administered by, for example, pouring on, spreading, rubbing, atomizing, spraying, dipping, bathing, or washing.
- a pour-on or spot-on formulation for example, can be poured or atomized onto a limited spot on the skin (typically no greater than about 10% of the skin).
- the formulation allows or facilitates the isoxazoline to penetrate the skin and act on other parts body (e.g., the entire body).
- Such a pour-on or spot-on formulation can be prepared by dissolving, suspending, or emulsifying the isoxazoline in a suitable skin-fitted solvent or solvent mixture.
- Contemplated solvents include, for example, water, alkanol, glycol, polyethylene glycol, polypropylene glycol, glycerin, benzyl alcohol, phenylethanol, phenoxyethanol, ethyl acetate, butyl acetate, benzyl benzoate, dipropylene glycol monomethyl ether, diethylene glycol monobutyl ether, acetone, methyl ethyl ketone, aromatic and/or aliphatic hydrocarbons, vegetable or synthetic oil, DMF, liquid paraffin, silicone, dimethylacetamide, N- methylpyrrolidone, or 2,2-dimethyl-4-oxy-methylene-l,3-dioxolane.
- Contemplated solvents include, for example, water, alkanol, glycol, polyethylene glycol, polypropylene glycol, glycerin, benzyl alcohol, phenylethanol, phenoxyethanol, ethyl acetate, butyl acetate
- a topical formulation (particularly a pour-on or spot- on formulation) comprises a carrier that promotes the absorption or penetration of the isoxazoline through the skin into the blood stream, other bodily fluids (lymph), and/or body tissue (fat tissue).
- Contemplated examples of dermal penetration enhancers include, for example, dimethylsulfoxide, isopropyl myristate, dipropylene glycol pelargonate, silicone oil, aliphatic esters, triglycerides, and fatty alcohols.
- Topical formulations also (or alternatively) may comprise, for example, one or more spreading agents.
- These substances act as carriers that assist in distributing an active ingredient over the animal recipient's coat or skin. They may include, for example, isopropyl myristate, dipropylene glycol pelargonate, silicone oils, fatty acid esters, triglycerides, and/or fatty alcohols.
- Various spreading oil/solvent combinations also may be suitable, such as, for example, oily solutions, alcoholic and isopropanolic solutions (e.g., solutions of 2-octyl dodecanol or oleyl alcohol), solutions of esters of monocarboxylic acids (e.g., isopropyl myristate, isopropyl palmitate, lauric acid oxalic ester, oleic acid oleyl ester, oleic acid decyl ester, hexyl laurate, oleyl oleate, decyl oleate, and caproic acid esters of saturated fatty alcohols having a carbon chain of 12 to 18 carbons), solutions of esters of dicarboxylic acids (e.g., dibutyl phthalate, diisopropyl isophthalate, adipic acid diisopropyl ester, and di-n-butyl adipate), or solutions of esters of alipha
- the formulation comprises a spreading agent
- a dispersant such as, for example, pyrrolidin-2-one, N- alkylpyrrolidin-2-one, acetone, polyethylene glycol or an ether or ester thereof, propylene glycol, or synthetic triglycerides.
- the isoxazoline may be mixed with, for example, either a paraffmic or a water-miscible ointment base.
- the isoxazoline may be formulated with, for example, an oil-in-water cream base.
- the aqueous phase of the cream base includes, for example at least about 30% (w/w) of a polyhydric alcohol, such as propylene glycol, butane- 1,3-diol, mannitol, sorbitol, glycerol, polyethylene glycol, or a mixture thereof.
- Injectable preparations may be formulated according to, for example, the known art using suitable solvents, solubilizing agents, protecting agents, dispersing agents, wetting agents, and/or suspending agents.
- Contemplated carrier materials include, for example, water, ethanol, butanol, benzyl alcohol, glycerin, 1,3-butanediol, Ringer's solution, isotonic sodium chloride solution, bland fixed oils (e.g., synthetic mono- or diglycerides), vegetable oil (e.g., corn oil), dextrose, mannitol, fatty acids (e.g., oleic acid), dimethyl acetamide, surfactants (e.g., ionic and non- ionic detergents), N- methylpyrrolidone, propylene glycol, and/or polyethylene glycols (e.g., PEG 400).
- Contemplated solubilizing agents include, for example, polyvinyl pyrrolidone, polyoxyethylated castor oil, polyoxyethylated sorbitan ester, and the like.
- Contemplated protecting agents include, for example, benzyl alcohol, trichlorobutanol, p- hydroxybenzoic acid ester, n-butanol, and the like.
- a parenteral formulation is, for example, prepared from sterile powders or granules having one or more of the carriers materials discussed above for other formulations.
- the isoxazoline is, for example, dissolved or suspended in a liquid comprising water, polyethylene glycol, propylene glycol, ethanol, corn oil, cottonseed oil, peanut oil, sesame oil, benzyl alcohol, sodium chloride, and/or various buffers.
- the pH generally may be adjusted, if necessary, with a suitable acid, base, or buffer.
- a suppository may be used for rectal administration.
- the suppository may be prepared by, for example, mixing a isoxazoline with a suitable non- irritating excipient that is solid at ordinary temperatures, but liquid at the rectal temperature, and will, therefore, melt in the rectum to release the drug.
- Contemplated excipients include, for example, such as cocoa butter; synthetic mono-, di-, or triglycerides; fatty acids; and/or polyethylene glycols.
- inert ingredients may generally be added to the composition as desired.
- these may include, for example, lactose, mannitol, sorbitol, calcium carbonate, sodium carbonate, tribasic calcium phosphate, dibasic calcium phosphate, sodium phosphate, kaolin, compressible sugar, starch, calcium sulfate, dextro or microcrystalline cellulose, colloidal silicon dioxide, starch, sodium starch glycolate, crospovidone, microcrystalline cellulose, tragacanth, hydroxypropylcellulose, pregelatinized starch, povidone, ethylcellulose, hydroxypropylcellulose, hydroxypropylmethylcellulose, and methylcellulose.
- the concentration of the isoxazoline of Formula (I) (or any salt of the isoxazoline, solvate of the isoxazoline or salt, or prodrug of the isoxazoline) in the composition may vary widely depending on, for example, the mode of administration. In general, the concentration is from about 1 to about 70% (by weight). In some such embodiments, for example, the concentration is from about 1 to about 50% (by weight), or from about 10 to about 50% (by weight). In other embodiments, the concentration is from about 35 to about 65% (by weight), from about 40 to about 60% (by weight), from about 45 to about 55% (by weight), or about 50% (by weight).
- the methods of this invention encompass methods wherein a isoxazoline is the sole active ingredient administered to the recipient animal. It is contemplated, however, that the methods also encompass combination therapies wherein a isoxazoline is administered in combination with one or more other active ingredients.
- the other active ingredient(s) may be, for example, one or more other isoxazo lines. Alternatively (or additionally), the other active ingredient(s) may be one or more compounds that are not isoxazo lines.
- the other active ingredient(s) may target the same and/or different pathogens and conditions.
- Contemplated active ingredient(s) that may be administered in combination with the isoxazoline include, for example, anthelmintics, insecticides and acaricides, insect growth regulators and juvenile hormone analogues, anti- inflammatories, anti- infectives, hormones, dermato logical preparations ⁇ e.g., antiseptics and disinfectants), and immunobiologicals ⁇ e.g., vaccines and antisera) for disease prevention.
- Anthelmintics include, for example, avermectins ⁇ e.g., ivermectin, moxidectin, and milbemycin), benzimidazoles ⁇ e.g., fenbendazole, albendazole, and triclabendazole), salicylanilides ⁇ e.g., closantel and oxyclozanide), substituted phenols ⁇ e.g., nitroxynil), pyrimidines ⁇ e.g., pyrantel), imidazothiazoles ⁇ e.g., levamisole), cyclooctadepsipeptide (e.g., Emodepside), and tetrahydropyrimidines (e.g., praziquantel).
- avermectins ⁇ e.g., ivermectin, moxidectin, and milbemycin
- benzimidazoles ⁇ e.
- Anthelmintics also include, for example, amino acetonitrile derivatives, such as, for example, those discussed in Kaminsky, R., et al, "A new class of anthelmintics effective against drug-resistant nematodes," Nature, vol. 452, pp. 176-180 (March 13, 2008); and Int'l Patent Publ. Nos. WO2006/050887 and WO2005/044784.
- the isoxazoline is administered in combination with (and, in some instances, in the same composition with) one or more macrocyclic lactone endectocidal parasiticides.
- macrocyclic lactone endectocidal parasiticides tend to be useful against, for example, a broad spectrum of endoparasites and ectoparasites in mammals.
- One particularly contemplated macrocyclic lactone parasiticide is ivermectin.
- Ivermectin is a semi- synthetic derivative of avermectin, and is generally produced as a mixture of at least 80% 22,23-dihydroavermectin Bl a and less than 20% 22,23-dihydroavermectin BI b .
- Ivermectin is disclosed in US Patent 4,199,569. Ivermectin has been used as an antiparasitic agent to treat various parasitic diseases since the mid- 1980's.
- Other macrocyclic lactone parasiticides include, for example: A. Abamectin. This compound is, for example, identified as avermectin Bl a /Blb in
- Abamectin contains at least 80% of avermectin Bl a , and not more than 20% of avermectin BIb.
- E. Milbemycin This compound also is known as B41. It is isolated from the fermentation broth of a Milbemycin-producing strain of Streptomyces . The microorganism, fermentation conditions, and isolation procedures are discussed in, for example, US Patents 3,950,360 and 3,984,564.
- F. Emamectin This compound also is known as 4"-deoxy-4"-epi- methylaminoavermectin B 1 . Its preparation is discussed in, for example, US Patent Nos. 5,288,710 and 5,399,717.
- salt of emamectin are commonly used. Non- limiting examples of such salts are those discussed in US Patent 5,288,710, which include salts derived from benzoic acid, substituted benzoic acid, benzenesulfonic acid, citric acid, phosphoric acid, tartaric acid, and maleic acid. A particularly contemplated salt is emamectin benzoate.
- Eprinomectin This compound is known as 4"-epi-acetylamino-4"-deoxy- avermectin B 1 . It was developed for use in all cattle classes and age groups. It was the first avermectin to generally show broad-spectrum activity against both endo- and ecto-parasites, while also leaving minimal residues in meat and milk. It generally has an additional advantage of being highly potent when delivered topically.
- Insecticides and acaricides include, for example, acephate, acetamiprid, acetoprole, amitraz, amidoflumet, avermectin, azadirachtin, azinphos-methyl, bifenthrin, bifenazate, buprofezin, bistrifluron, buprofezin, carbofuran, cartap, chlorfenapyr, chlorfluazuron, chlorantraniliprole), chlorpyrifos, chlorpyrifos-methyl, chromafenozide, clothianidin, cyflumetofen, cyfluthiin, ⁇ -cyfluthrin, cyhalothrin, ⁇ -cyhalothrin ⁇ - cyhalothrin, cypermethrin, cyromazine, deltamethrin, diafenthiuron, diazinon,
- the isoxazoline is administered with pyridylmethylamine derivatives, such as, for example, pyridylmethylamine derivatives discussed in European Patent Appl. EP0539588 or Int'l Patent Appl. Publ. WO2007/115643.
- the isoxazoline is administered with nodulisporic acids and derivatives thereof, such as, for example, compounds discussed in US Patent 5,399,582; 5,945,317; 5,962,499; 5,834,260; 6,221,894; or 5,595,991; or Int'l Patent Appl. Publ. 1996/29073.
- Insect growth regulators include, for example, agridyne, diofenolan, fenoxycarb, hydroprene, kinoprene, methoprene, pyriproxyfen, tetrahydroazadirachtin, chlorfluazuron, cyromazine, diflubenzuron, fluazuron, fluey cloxuron, flufenoxuron, hexaflumuron, ifenuron, tebufenozide, and triflumuron. These compounds tend to provide both initial and sustained control of parasites at all stages of insect development, including eggs, on the animal subject, as well as within the environment of the animal subject.
- antiparasitic compounds contemplated to be useful in combination therapies with the isoxazoline include, for example, imidazo[l,2-b] pyridazine compounds discussed in US Patent Appl. Publ. No. 2005-0182059; l-(4-Mono and di- halomethylsulphonylphenyl)-2-acylamino-3-fluoropropanol compounds discussed US Patent 7,361,689; trifluoromethanesulfonanilide oxime ether compounds discussed in US Patent 7,312,248; n-[(phenyloxy)phenyl]-l,l,l-trifluoromethanesulfonamide and n-
- Anti-inflammatory agents include, for example, corticosteroids, which, in turn, include, for example, beclomethasone dipropionate, betamethasone diproprionate, betamethasone valerate, budesonide, ciclesonide, deflazacort, dexamethasone, fluocinolone acetonide, fluticasone, propionate, fluticasone furoate, loteprednol, etabonate, mometasone, and mometasone furoate, methylprednisolone, prednisolone, prednisone, rofleponide, and triamcinolone acetonide.
- Anti-inflammatory agents also include, for example, one or more non-steroidal anti- inflammatory drugs ("NSAIDs").
- NSAIDs include, for example, salicylates, arylalkanoic acids, 2-arylpropionic acids (or “profens”), N-arylanthranilic acids, pyrazolidine derivatives, oxicams, COX-2 inhibitors, sulphonanilides, and licofelone.
- Anti- inflammatory ingredients also may include, for example, antihistamines.
- Antihistamines include, for example, Hi -receptor agonists, H 2 - receptor agonists, H 3 -receptor agonists, H 4 -receptor agonists, mast cell stabilizers, and vitamin C.
- the isoxazoline of Formula (I) may be administered before, simultaneously, and/or after the other active ingredient(s).
- the isoxazoline may be administered in the same composition as the other active ingredient(s) and/or in a separate compositions from the other active ingredient(s).
- the isoxazoline and other active ingredient(s) may be administered via the same and/or different routes of administration.
- the weight ratio of the active ingredients may vary widely.
- Factors influencing this ratio include, for example, the particular isoxazoline; the identity of the other active ingredient(s) be administered in the combination therapy; the mode(s) of administration of the isoxazoline and other active ingredient(s); the target condition and pathogen; the type (e.g., species and breed), age, size, sex, diet, activity, and condition of the intended recipient; and pharmacological considerations, such as the activity, efficacy, pharmacokinetic, and toxicology profiles of the isoxazoline and other active ingredient(s).
- the weight radio of the isoxazoline to the other active ingredient(s) is, for example, is from about 1:3000 to about 3000:1. In some such instances, the weight ratio is from about 1 :300 to about 300: 1. In other such instances, the weight ratio is from about 1 :30 and about 30:1.
- the isoxazoline may be administered with one or more other compounds that beneficially affects (e.g., enhances or prolongs) the activity (or other characteristic, such as safety) of the isoxazoline.
- the isoxazoline may be administered with one or more synergists, such as, for example, piperonyl butoxide (PBO) and triphenyl phosphate (TPP).
- PBO piperonyl butoxide
- TPP triphenyl phosphate
- synergists include, for example, N ⁇ (2-cihySSiexySj-( ⁇ ,9 JO- trinorborn-5-ene ⁇ 2,3 ⁇ dicarboxamide (also known as “ENT 8184" or “MGK 264”) and Verbutin (also known as "MB-599”).
- ENT 8184 or "MGK 264”
- Verbutin also known as "MB-599”
- a discussion relating to insecticidal synergists may be found in, for example, The Pesticide Manual, 13th Edition, cited above. VI. Therapeutic kits
- kits that are, for example, suitable for use in performing the treatment methods described above.
- a kit will comprise a therapeutically effective amount of a isoxazoline of Formula (I), and an additional component(s).
- the additional component(s) may be, for example, one or more of the following: a diagnostic tool, instructions for administering the composition, an apparatus for administering the composition, a container comprising an excipient or other active ingredient to be mixed or administered in combination with the composition, or a memory aid ⁇ e.g., a stamp to adhere to a calendar to remind an animal owner of a time to administer a subsequent dose of the composition).
- Example 1 Efficacy of Compounds 10-1 and 11-1 against cat fleas ⁇ Ctenocephalides felis).
- Adult fleas (20-30) were fed on artificial membranes with blood spiked with Compound 10-1, Compound 11-1, or a positive control (fipronil) at a concentration of 100, 10, or 1 ppm.
- Flea efficacy was assessed after 48 hr of continuous feeding by comparing the number of killed and damaged fleas with the number of fed fleas.
- Flea efficacy was 100% for Compound 10-1, Compound 11-1, and fipronil at 100, 10, and 1 ppm.
- mice were divided into groups of three, and treated topically with 100 ppm body weight, orally with 10 mg/kg bodyweight, or subcutaneously with 10 mg/kg bodyweight of Compound 11-1, f ⁇ pronil (positive control), or nothing (negative control). These mice were sedated and infested with adult fleas (C.felis) 1, 3, 6, and 24 hr after treatment. Fleas were recovered from the mice after approximately 30 minutes of feeding. The assessment of flea inhibition (% of dead and damaged fleas) was conducted 1 and 24 hr after each infestation. The results are shown in Table 1.
- mice used in the study had a present mite infestation with M. musculinus consisting of all stages of the parasite with at least a medium (“++") infestation rate.
- the mice were divided into groups of three, and treated topically with 100 ppm body weight, orally with 10 mg/kg body weight, or subcutaneously with 10 mg/kg body weight of Compound 11-1, f ⁇ pronil (positive control), or nothing (negative control). This treatment was then repeated 7 days later.
- ticks and bed bugs died within 1 to 5 hr following infestation up to Day 29, and within 8 to 24 hr after day 29.
- ticks and bed bugs died within 1 to 7 hr after infestation up to Day 29, and within 8 to 24 hr after day 29.
- the efficacy results with respect to ticks are shown in Table 3.
- Guinea pigs were divided into groups of three. Each group was subjected to one of the following treatments with either Compound 11-1, fipronil (positive control), or neither (negative control):
- mice were randomly assigned to a treatment group or a negative control (untreated) group. Each group consisted of three mice. The mice in the treatment groups were orally administered 20 mg/kg bodyweight of various isoxazolines dissolved in 7% DMF-premix and 93% purified water (aqua ad injectabilia). The application volume of these treatments was 0.01 mL/g bodyweight.
- Each mouse was sedated and generally infested (whole body) with 30 vital, adult fleas (C. felis). To achieve this, the sedated mice were placed into an infestation-jar, and the fleas were placed directly onto the fur. After approximately 30 minutes of feeding, fleas were recovered from the mice.
- Example 4 Efficacy of various isoxazolines against brown dog ticks (Rhipicephalus sanguineus) on Guinea pigs.
- Guinea pigs were randomly assigned to a treatment group or a negative control (untreated) group. Each group consisted of three Guinea pigs. On Day Zero, each Guinea pig was infested with 100 vital nymphs of R. sanguineus. On Day 1, the Guinea pigs in the treatment groups were orally administered 10 mg/kg bodyweight of various isoxazolines dissolved in 7% DMF-premix and 93% purified water (aqua ad injectabilia).
- Example 5 Efficacy of Compound 11-1 against cat fleas (C. felts) and brown dog ticks (R. sanguineus) on dogs.
- one group of 4 beagles was treated orally with gelatin capsules containing 20 mg/kg body weight of Compound 11-1.
- Another group of 4 beagles was treated topically by washing with 2 L of solution containing 200 ppm body weight of Compound 11-1 dissolved in DMF-premix/tap water (1:100, based on volume). Finally, a group of 3 beagles remained untreated as the negative control. Two days before treatment on Day Zero, all the beagles were each infested with approximately 80 unfed adult fleas (C.
- Compound 11-1 exhibited a flea and tick efficacy of 100% after oral and topical treatment. No side effects were observed during this study.
- Example 6 Further efficacy study of Compound 11-1 against cat fleas (C felis) and brown dog ticks (R. sanguineus) on dogs.
- the dogs were infested with approximately 80 fleas (C.felis) and 60 ticks (R. sanguineus) on Days -2, 7, 14, 21, 28, 35, 42, and 49. Fleas and ticks were counted on Day 2 (approximately 48 h after treatment), as well as Days 9, 16, 23, 30, 37, 44, and 51 (approximately 48 hr after each post-treatment re-infestation) to evaluate the insecticidal and acaricidal activity in the treated groups. In Group B, an additional flea and tick infestation was conducted on Day 56, with a respective flea and tick count on Day 58.
- FIG. 1 The observed mean plasma concentration of Compound 11-1 over the duration of the study is shown in Figure 1.
- the "PO” data refers to Group A
- the "SC” data refers to Group B
- the "TOP W/ ENH” data refers to Group C
- the “TOP W/ ENH & SPREAD” data refers to Group D
- the “TOP W/ ETHYL LACTATE” data refers to Group E.
- the data for Group F was not included in Figure 1, given that there was no Compound 11-1 administered to that group.
- the term "pharmaceutically acceptable” is used adjectivally to mean that the modified noun is appropriate for use in a pharmaceutical product.
- pharmaceutically acceptable when it is used, for example, to describe a salt, excipient, or solvate, it characterizes the salt, excipient, or solvate as being compatible with the other ingredients of the composition, and not deleterious to the intended recipient animal to the extent that the deleterious effect(s) outweighs the benef ⁇ t(s) of the salt, excipient, or solvate.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Veterinary Medicine (AREA)
- Plant Pathology (AREA)
- Agronomy & Crop Science (AREA)
- Environmental Sciences (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Dentistry (AREA)
- Pest Control & Pesticides (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Tropical Medicine & Parasitology (AREA)
- Hematology (AREA)
- Diabetes (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Plural Heterocyclic Compounds (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
Claims
Priority Applications (27)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MX2010001907A MX2010001907A (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics. |
| KR1020177009149A KR101792547B1 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
| PL12188400.1T PL2545777T5 (en) | 2007-08-17 | 2008-08-15 | Use of an isoxazoline composition as antiparasitic |
| BR122020005235-0A BR122020005235B1 (en) | 2007-08-17 | 2008-08-15 | PARASITICIDAL PHARMACEUTICAL COMPOSITION AND USE OF FLURALANER |
| NZ583045A NZ583045A (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
| DK08803041.6T DK2190289T3 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compounds and their use as antiparasitics |
| JP2010520592A JP5411135B2 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitic agents |
| CA2694919A CA2694919C (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
| CN200880103027A CN101778566A (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and as the purposes of antiparasitic agent |
| EP12188400.1A EP2545777B2 (en) | 2007-08-17 | 2008-08-15 | Use of an isoxazoline composition as antiparasitic |
| EP08803041.6A EP2190289B1 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
| ES08803041T ES2417157T3 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitic |
| KR1020167006583A KR101725320B1 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
| US12/673,722 US20110059988A1 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
| MX2013009474A MX351346B (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics. |
| EP18205103.7A EP3473101B1 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics for felines |
| PL08803041T PL2190289T3 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
| AU2008290581A AU2008290581C1 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
| RU2010109901/15A RU2481837C2 (en) | 2007-08-17 | 2008-08-15 | Isoxasoline compositions and their application as anti-parasite medications |
| BRPI0815227-6A BRPI0815227B1 (en) | 2007-08-17 | 2008-08-15 | isoxazoline use |
| HK10109524.4A HK1143037A1 (en) | 2007-08-17 | 2010-10-06 | Isoxazoline compositions and their use as antiparasitics |
| US14/579,387 US20150111936A1 (en) | 2007-08-17 | 2014-12-22 | Isoxazoline compositions and their use as antiparasitics |
| US15/222,171 US20160332975A1 (en) | 2007-08-17 | 2016-07-28 | Isoxazoline compositions and their use as antiparasitics |
| NO2016018C NO2016018I1 (en) | 2007-08-17 | 2016-10-05 | Fluralaner |
| US15/880,770 US20180155301A1 (en) | 2007-08-17 | 2018-01-26 | Isoxazoline compositions and their use as antiparasitics |
| US17/021,698 US20210061775A1 (en) | 2007-08-17 | 2020-09-15 | Isoxazoline compositions and their use as antiparasitics |
| US18/416,336 US20240239759A1 (en) | 2007-08-17 | 2024-01-18 | Isoxazoline compositions and their use as antiparasitics |
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US95644807P | 2007-08-17 | 2007-08-17 | |
| US60/956,448 | 2007-08-17 | ||
| EP07016152 | 2007-08-17 | ||
| EP07016152.6 | 2007-08-17 | ||
| EP07150309 | 2007-12-21 | ||
| EP07150309.8 | 2007-12-21 | ||
| US8044408P | 2008-07-14 | 2008-07-14 | |
| US61/080,444 | 2008-07-14 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/673,722 A-371-Of-International US20110059988A1 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
| US14/579,387 Continuation US20150111936A1 (en) | 2007-08-17 | 2014-12-22 | Isoxazoline compositions and their use as antiparasitics |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2009024541A2 true WO2009024541A2 (en) | 2009-02-26 |
| WO2009024541A3 WO2009024541A3 (en) | 2010-04-01 |
Family
ID=40152741
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2008/060732 WO2009024541A2 (en) | 2007-08-17 | 2008-08-15 | Isoxazoline compositions and their use as antiparasitics |
Country Status (20)
| Country | Link |
|---|---|
| US (6) | US20110059988A1 (en) |
| EP (3) | EP3473101B1 (en) |
| JP (4) | JP5411135B2 (en) |
| KR (3) | KR20100057066A (en) |
| CN (2) | CN101778566A (en) |
| AU (2) | AU2008290581C1 (en) |
| BR (2) | BR122020005235B1 (en) |
| CA (1) | CA2694919C (en) |
| DK (2) | DK2190289T3 (en) |
| ES (2) | ES2785104T5 (en) |
| HK (1) | HK1143037A1 (en) |
| MX (2) | MX351346B (en) |
| NO (1) | NO2016018I1 (en) |
| NZ (2) | NZ583045A (en) |
| PL (2) | PL2545777T5 (en) |
| PT (1) | PT2190289E (en) |
| RU (3) | RU2481837C2 (en) |
| TW (1) | TWI556741B (en) |
| WO (1) | WO2009024541A2 (en) |
| ZA (1) | ZA201203080B (en) |
Cited By (73)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010005048A1 (en) | 2008-07-09 | 2010-01-14 | 日産化学工業株式会社 | Process for production of isoxazoline-substituted benzoic acid amide compound |
| WO2010065852A1 (en) | 2008-12-04 | 2010-06-10 | Merial Limited | Dimeric avermectin and milbemycin derivatives |
| WO2011075591A1 (en) | 2009-12-17 | 2011-06-23 | Merial Limited | Anti parasitic dihydroazole compounds and compositions comprising same |
| WO2011123773A1 (en) | 2010-04-02 | 2011-10-06 | Merial Limited | Parasiticidal compositions comprising multiple active agents, methods and uses thereof |
| WO2011154555A1 (en) * | 2010-06-11 | 2011-12-15 | Syngenta Participations Ag | Process for the preparation of dihydropyrrole derivatives |
| WO2012007426A1 (en) | 2010-07-13 | 2012-01-19 | Basf Se | Azoline substituted isoxazoline benzamide compounds for combating animal pests |
| US8119671B2 (en) * | 2007-03-07 | 2012-02-21 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and pest control agent |
| WO2012060317A1 (en) * | 2010-11-04 | 2012-05-10 | 日本曹達株式会社 | Ectoparasite control agent for animals |
| DE102010063691A1 (en) | 2010-12-21 | 2012-06-21 | Bayer Animal Health Gmbh | Ectoparasiticidal drug combinations |
| WO2012089622A2 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation |
| WO2012089623A1 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| WO2013003505A1 (en) | 2011-06-27 | 2013-01-03 | Merial Limited | Amido-pyridyl ether compounds and compositions and their use against parasites |
| EP2550962A2 (en) | 2008-11-19 | 2013-01-30 | Merial Limited | Compositions comprising an aryl pyrazole and/or a formamidine, methods and uses thereof |
| WO2013039948A1 (en) | 2011-09-12 | 2013-03-21 | Merial Limited | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| WO2013074892A1 (en) | 2011-11-17 | 2013-05-23 | Merial Limited | Compositions comprising an aryl pyrazole and a substituted imidazole, methods and uses thereof |
| WO2013078071A1 (en) * | 2011-11-21 | 2013-05-30 | Eli Lilly And Company | Isoxazoline derivatives used in the control of ectoparasites |
| WO2013082373A1 (en) | 2011-12-02 | 2013-06-06 | Merial Limited | Long-acting injectable moxidectin formulations and novel moxidectin crystal forms |
| WO2013150055A1 (en) | 2012-04-04 | 2013-10-10 | Intervet International B.V. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| WO2013158894A1 (en) | 2012-04-20 | 2013-10-24 | Merial Limited | Parasiticidal compositions comprising benzimidazole derivatives, methods and uses thereof |
| US8648081B2 (en) | 2011-05-19 | 2014-02-11 | Eli Lilly And Company | Parasiticidal dihydroisoxazole compounds |
| US8686014B2 (en) | 2008-09-04 | 2014-04-01 | Syngenta Limited | Insecticidal compounds |
| US8735362B2 (en) | 2009-12-01 | 2014-05-27 | Syngenta Crop Protection, Llc | Insecticidal compounds based on isoxazoline derivatives |
| WO2014081697A2 (en) | 2012-11-20 | 2014-05-30 | Merial Limited | Anthelmintic compounds and compositions and method of using thereof |
| WO2014079825A1 (en) * | 2012-11-20 | 2014-05-30 | Intervet International B.V. | Manufacturing of semi-plastic pharmaceutical dosage units |
| GB2509361A (en) * | 2012-11-01 | 2014-07-02 | Sumitomo Chemical Co | Isoxazoline compound for use in controlling an animal ectoparasite |
| WO2014122083A1 (en) | 2013-02-06 | 2014-08-14 | Bayer Cropscience Ag | Halogen-substituted pyrazol derivatives as pest-control agents |
| US8822502B2 (en) | 2008-08-22 | 2014-09-02 | Syngenta Crop Protection Llc | Insecticidal compounds |
| US8853410B2 (en) | 2009-04-30 | 2014-10-07 | Basf Se | Process for preparing substituted isoxazoline compounds and their precursors |
| EP2785341A1 (en) * | 2011-11-29 | 2014-10-08 | Novartis AG | Aryl derivatives for controlling ectoparasites |
| JP2015057399A (en) * | 2007-06-27 | 2015-03-26 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニーE.I.Du Pont De Nemours And Company | Method of controlling animal pest |
| US9006447B2 (en) | 2010-11-03 | 2015-04-14 | Basf Se | Method for preparing substituted isoxazoline compounds and their precursors 4-chloro, 4-bromo- or 4-iodobenzaldehyde oximes |
| WO2015066277A1 (en) | 2013-11-01 | 2015-05-07 | Merial Limited | Antiparisitic and pesticidal isoxazoline compounds |
| WO2015086551A1 (en) | 2013-12-10 | 2015-06-18 | Intervet International B.V. | Antiparasitic use of isoxazoline compounds |
| WO2015091900A1 (en) | 2013-12-20 | 2015-06-25 | Intervet International B.V. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| WO2015101622A1 (en) | 2014-01-03 | 2015-07-09 | Bayer Cropscience Ag | Novel pyrazolyl-heteroarylamides as pesticides |
| US9085541B2 (en) | 2010-06-23 | 2015-07-21 | Basf Se | Process for producing imine compounds for combating invertebrate pests |
| WO2015179414A1 (en) | 2014-05-19 | 2015-11-26 | Merial, Inc. | Anthelmintic compounds |
| US9216973B2 (en) | 2008-08-22 | 2015-12-22 | Syngenta Participations Ag | Insecticidal compounds |
| WO2015196014A1 (en) | 2014-06-19 | 2015-12-23 | Merial, Inc. | Parasiticidal compositions comprising indole derivatives, methods and uses thereof |
| WO2016008830A1 (en) | 2014-07-15 | 2016-01-21 | Bayer Cropscience Aktiengesellschaft | Aryl-triazolyl pyridines as pest control agents |
| US9314478B2 (en) | 2013-03-15 | 2016-04-19 | Argenta Manufacturing Limited | Method of making an anhydrous, fat soluble, chewable drug delivery formulation |
| WO2016069983A1 (en) | 2014-10-31 | 2016-05-06 | Merial, Inc. | Parasiticidal composition comprising fipronil |
| WO2016102437A1 (en) * | 2014-12-22 | 2016-06-30 | Intervet International B.V. | Use of isoxazoline compounds for treating demodicosis |
| US20160317439A1 (en) * | 2013-12-20 | 2016-11-03 | Intervet Inc. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| EP2306837B1 (en) | 2008-07-09 | 2017-04-26 | Basf Se | Pesticidal active mixtures comprising isoxazoline compounds i |
| WO2017147352A1 (en) | 2016-02-24 | 2017-08-31 | Merial, Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| WO2018039508A1 (en) | 2016-08-25 | 2018-03-01 | Merial, Inc. | Method for reducing unwanted effects in parasiticidal treatments |
| US10045969B2 (en) | 2004-03-05 | 2018-08-14 | Nissan Chemical Industries, Inc. | Isoxazoline-substituted benzamide compound and pesticide |
| WO2019091940A1 (en) | 2017-11-07 | 2019-05-16 | Intervet International B.V. | Process for preparing large size isoxazoline particles |
| WO2019091936A1 (en) | 2017-11-07 | 2019-05-16 | Intervet International B.V. | Injectable isoxazoline pharmaceutical compositions and their use against parasite infestation |
| EP3498696A1 (en) | 2008-11-14 | 2019-06-19 | Merial, Inc. | Enantiomerically enriched aryloazol-2-yl cyanoethylamino parasiticidal compounds |
| WO2019115492A1 (en) | 2017-12-12 | 2019-06-20 | Intervet International B.V. | Implantable isoxazoline pharmaceutical compositions and uses thereof |
| WO2019122324A1 (en) | 2017-12-21 | 2019-06-27 | Intervet International B.V. | Antiparasitic pour-on compositions |
| WO2019157241A1 (en) | 2018-02-08 | 2019-08-15 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising eprinomectin and praziquantel, methods and uses thereof |
| US20190343808A1 (en) * | 2015-06-23 | 2019-11-14 | Intervet Inc. | Isoxazoline solution containing vitamin e for use with sanitized drinking water |
| US10561641B2 (en) | 2015-02-26 | 2020-02-18 | Boehringer Ingelheim Animal Health USA Inc. | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| EP2545777B1 (en) | 2007-08-17 | 2020-03-11 | Intervet International B.V. | Use of an isoxazoline composition as antiparasitic |
| WO2020150032A1 (en) | 2019-01-16 | 2020-07-23 | Boehringer Ingelheim Animal Health USA Inc. | Topical compositions comprising a neonicotinoid and a macrocyclic lactone, methods and uses thereof |
| WO2020180635A1 (en) | 2019-03-01 | 2020-09-10 | Boehringer Ingelheim Animal Health USA Inc. | Injectable clorsulon compositions, methods and uses thereof |
| WO2020202111A1 (en) * | 2019-04-04 | 2020-10-08 | Tarsus Pharmaceuticals, Inc. | Systemic isoxazoline parasiticides for vector-borne and viral disease treatment or prophylaxis |
| WO2020225143A1 (en) | 2019-05-03 | 2020-11-12 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| US10835517B2 (en) | 2017-12-15 | 2020-11-17 | Tarsus Pharmaceuticals, Inc. | Methods for treating ocular demodex using isoxazoline parasiticide formulations |
| WO2021013825A1 (en) | 2019-07-22 | 2021-01-28 | Intervet International B.V. | Soft chewable veterinary dosage form |
| EP3061454B1 (en) | 2012-02-06 | 2021-03-10 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| WO2021122513A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Composition for lice control |
| WO2021122521A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Composition for lice control in poultry |
| WO2021122515A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Parasite control in ruminants |
| WO2021233967A1 (en) | 2020-05-20 | 2021-11-25 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| WO2021242481A1 (en) | 2020-05-28 | 2021-12-02 | Boehringer Ingelheim Animal Health USA Inc. | Bi-modal release intra-ruminal capsule device and methods of use thereof |
| WO2022140728A1 (en) | 2020-12-21 | 2022-06-30 | Boehringer Ingelheim Animam Health Usa Inc. | Parasiticidal collar comprising isoxazoline compounds |
| US20220242835A1 (en) * | 2019-07-01 | 2022-08-04 | Shenyang University Of Chemical Technology | Isoxazoline compounds and use thereof |
| WO2022226660A1 (en) * | 2021-04-30 | 2022-11-03 | Ohalloran John | Use of isoxazoline for protection against parasitic pests in fish |
| WO2022269042A1 (en) | 2021-06-25 | 2022-12-29 | Intervet International B.V. | Palatable veterinary compositions |
Families Citing this family (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW200803740A (en) * | 2005-12-16 | 2008-01-16 | Du Pont | 5-aryl isoxazolines for controlling invertebrate pests |
| TWI412322B (en) | 2005-12-30 | 2013-10-21 | Du Pont | Isoxazolines for controlling invertebrate pests |
| MX2009013469A (en) | 2007-06-13 | 2010-01-20 | Du Pont | Isoxazoline insecticides. |
| TWI600639B (en) | 2007-08-17 | 2017-10-01 | 杜邦股份有限公司 | Compound for preparing 5-haloalkyl-4,5-dihydroisoxazole derivatives |
| TW201444787A (en) | 2008-04-09 | 2014-12-01 | Du Pont | Method for preparing 3-trifluoromethyl chalcones |
| KR20130124458A (en) | 2010-05-27 | 2013-11-14 | 이 아이 듀폰 디 네모아 앤드 캄파니 | Crystalline form of 4-[5-[3-chloro-5-(trifluoromethyl)phenyl]-4,5-dihydro-5-(trifluoromethyl)-3-isoxazolyl]-n-[2-0x0-2-[(2,2,2-trifluoroethyl)amino]ethyl]-1-naphthalenecarboxamide |
| KR102193090B1 (en) * | 2010-12-27 | 2020-12-21 | 인터벳 인터내셔널 비.브이. | Topical localized isoxazoline formulation |
| JO3626B1 (en) | 2012-02-23 | 2020-08-27 | Merial Inc | Topical compositions comprising fipronil and permethrin and methods of use |
| CA2917815C (en) | 2013-07-29 | 2022-08-30 | Attillaps Holdings | Organophosphates for treating afflictions of the skin |
| US11446241B2 (en) | 2013-07-29 | 2022-09-20 | Attillaps Holdings Inc. | Treatment of ophthalmological conditions with acetylcholinesterase inhibitors |
| US9371293B2 (en) | 2013-10-25 | 2016-06-21 | Sumitomo Chemical Company, Limited | Isoxazoline compound composition |
| NZ726251A (en) | 2014-04-17 | 2017-11-24 | Merial Inc | Use of malononitrile compounds for protecting animals from parasites |
| AU2015277038B2 (en) | 2014-06-19 | 2020-09-17 | Attillaps Holdings | Acetylcholinesterase inhibitors for treatment of dermatological conditions |
| CN108055825B (en) * | 2015-04-08 | 2021-03-30 | 勃林格殷格翰动物保健美国公司 | Extended release injectable formulations comprising isoxazoline active agents, methods and uses thereof |
| DK3298027T3 (en) | 2015-05-20 | 2021-08-09 | Boehringer Ingelheim Animal Health Usa Inc | ANTHELMINTIC DEPSIPEPTID COMPOUNDS |
| EP3440062B1 (en) | 2016-04-06 | 2021-10-06 | Boehringer Ingelheim Animal Health USA Inc. | Process for the preparation of enantiomerically enriched isoxazoline compounds - crystalline toluene solvate of (s)-afoxolaner |
| US10653144B2 (en) | 2016-10-14 | 2020-05-19 | Boehringer Ingelheim Animal Health USA Inc. | Pesticidal and parasiticidal vinyl isoxazoline compounds |
| BR112019009977A2 (en) | 2016-11-16 | 2019-08-27 | Boehringer Ingelheim Animal Health Usa Inc | anthelmintic depsipeptide compounds |
| CN107535503A (en) * | 2017-07-21 | 2018-01-05 | 陕西康禾立丰生物科技药业有限公司 | A kind of Pesticidal combination containing Fu oxazoles acid amides and anabasine |
| WO2019036407A1 (en) | 2017-08-14 | 2019-02-21 | Merial, Inc. | Pesticidal and parasiticidal pyrazole-isoxazoline compounds |
| CN107593723A (en) * | 2017-09-29 | 2018-01-19 | 北京科发伟业农药技术中心 | A kind of composition containing fluxametamide |
| CN112996788B (en) | 2018-07-09 | 2024-07-16 | 勃林格殷格翰动物保健美国公司 | Anthelmintic heterocyclic compounds |
| EP3883648A1 (en) | 2018-11-20 | 2021-09-29 | Boehringer Ingelheim Animal Health USA Inc. | Indazolylcyanoethylamino compound, compositions of same, method of making, and methods of using thereof |
| WO2020191091A1 (en) | 2019-03-19 | 2020-09-24 | Boehringer Ingelheim Animal Health USA Inc. | Anthelmintic aza-benzothiophene and aza-benzofuran compounds |
| WO2021242581A1 (en) | 2020-05-29 | 2021-12-02 | Boehringer Ingelheim Animal Health USA Inc. | Anthelmintic heterocyclic compounds |
| CN111657292A (en) * | 2020-06-03 | 2020-09-15 | 浙江中山化工集团股份有限公司 | Insecticidal composition containing fluxapyroxad and beta-cypermethrin |
| CN111909143B (en) * | 2020-07-30 | 2021-10-29 | 山东省联合农药工业有限公司 | Isoxazoline substituted benzamide derivative and preparation method and application thereof |
| CA3201608A1 (en) | 2020-12-18 | 2022-06-23 | Boehringer Ingelheim Animal Health USA Inc. | Boron containing pyrazole compounds, compositions comprising them, methods and uses thereof |
| US20240116854A1 (en) | 2021-01-27 | 2024-04-11 | Intervet Inc. | Cyclopropylamide compounds against parasites in fish |
| CA3209562A1 (en) | 2021-01-27 | 2022-08-04 | Intervet International B.V. | Cyclopropylamide compounds against parasites in fish |
| EP4304356A1 (en) * | 2021-03-11 | 2024-01-17 | In the Bowl Animal Health, Inc. | Oral canine feed and methods for controlling flea infestations in a canine |
| JP2024511956A (en) * | 2021-03-11 | 2024-03-18 | イン ザ ボウル アニマル ヘルス,インコーポレーテッド | Oral canine feed and methods of controlling tick infestations in canids |
| CN113563322B (en) * | 2021-07-26 | 2023-07-07 | 海利尔药业集团股份有限公司 | Substituted benzamide isoxazoline derivative or salt acceptable as pesticide, composition and application thereof |
| KR102660238B1 (en) * | 2021-12-20 | 2024-04-25 | 주식회사경농 | Dihydroisoxazole derivative compound and pesticide composition comprising same |
| WO2023156938A1 (en) | 2022-02-17 | 2023-08-24 | Boehringer Ingelheim Vetmedica Gmbh | Method and system for providing a fluid product mailer |
| CN117126116A (en) * | 2022-05-20 | 2023-11-28 | 天津瑞普生物技术股份有限公司 | Derivative of isoxazoline compound and application thereof |
| CN117122571A (en) * | 2022-05-20 | 2023-11-28 | 天津瑞普生物技术股份有限公司 | Oral medicinal preparation for resisting parasitic infection, and preparation method and application thereof |
| KR20240002746A (en) * | 2022-06-30 | 2024-01-08 | 주식회사경농 | Isoxazoline derivative compound and pesticide composition comprising the same |
| CN115785017B (en) * | 2022-12-06 | 2023-12-08 | 海利尔药业集团股份有限公司 | Substituted benzamide isoxazoline derivative or salt, composition and application thereof as pesticide acceptable |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070066617A1 (en) * | 2004-03-05 | 2007-03-22 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and pesticide |
Family Cites Families (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3950360A (en) | 1972-06-08 | 1976-04-13 | Sankyo Company Limited | Antibiotic substances |
| US3984564A (en) | 1972-06-08 | 1976-10-05 | Sankyo Company Limited | Antibiotic substances B-41, their production and their use as insecticides and acaricides |
| SE434277B (en) | 1976-04-19 | 1984-07-16 | Merck & Co Inc | SET TO MAKE NEW ANTIHELMINTICALLY EFFECTIVE ASSOCIATIONS BY CULTIVATING STREPTOMYCS AVERMITILIS |
| US4199569A (en) | 1977-10-03 | 1980-04-22 | Merck & Co., Inc. | Selective hydrogenation products of C-076 compounds and derivatives thereof |
| ES8800986A1 (en) | 1985-07-27 | 1987-12-01 | Pfizer | Antiparasitic avermectin and milbemycin derivatives and process for their preparation. |
| US4916154A (en) | 1986-09-12 | 1990-04-10 | American Cyanamid Company | 23-Imino derivatives of LL-F28249 compounds |
| IL98599A (en) | 1990-06-28 | 1995-06-29 | Merck & Co Inc | Stable salts of 4"-deoxy-4"-epi-methylamino avermectin b1a/b1b and insecticidal compositions containing them |
| WO1992000964A1 (en) | 1990-07-05 | 1992-01-23 | Nippon Soda Co., Ltd. | Amine derivative |
| GB9205007D0 (en) * | 1992-03-07 | 1992-04-22 | Pfizer Ltd | Antiparasitic agents |
| US5399717A (en) | 1993-09-29 | 1995-03-21 | Merck & Co., Inc. | Glycosidation route to 4"-epi-methylamino-4"-deoxyavermectin B1 |
| US5399582A (en) | 1993-11-01 | 1995-03-21 | Merck & Co., Inc. | Antiparasitic agents |
| US6221894B1 (en) | 1995-03-20 | 2001-04-24 | Merck & Co., Inc. | Nodulisporic acid derivatives |
| SK284840B6 (en) | 1995-03-20 | 2005-12-01 | Merck & Co., Inc. | Nodulisporic acid derivatives and pharmaceutical composition containing them |
| US5595991A (en) | 1995-03-20 | 1997-01-21 | Merck & Co., Inc. | Anthelmintic use of nodulisporic acid and analogs thereof |
| US5962499A (en) | 1995-03-20 | 1999-10-05 | Merck & Co., Inc. | Nodulisporic acid derivatives |
| FR2750861B1 (en) | 1996-07-11 | 1998-12-24 | Rhone Merieux | PROCESSES FOR REMOVING PARASITES, ESPECIALLY VERTEBRATE ECTOPARASITES, ESPECIALLY MAMMALS AND COMPOSITIONS FOR CARRYING OUT THIS PROCESS |
| US5834260A (en) | 1996-08-30 | 1998-11-10 | Merck & Co., Inc. | Antiparasitic agents |
| US20050192319A1 (en) | 1996-09-19 | 2005-09-01 | Albert Boeckh | Spot-on formulations for combating parasites |
| FR2753377B1 (en) | 1996-09-19 | 1999-09-24 | Rhone Merieux | NOVEL PARASITICIDE ASSOCIATION BASED ON 1-N-PHENYLPYRAZOLES AND ENDECTOCIDAL MACROCYCLIC LACTONES |
| FR2761232B1 (en) † | 1997-03-26 | 2000-03-10 | Rhone Merieux | PROCESS AND MEANS FOR ERADICATION OF CHIPS IN PREMISES LIVED BY SMALL MAMMALS |
| RU2129430C1 (en) * | 1998-03-17 | 1999-04-27 | Общество С Ограниченной Ответственностью "Вик - Здоровье Животных" | Preparation "santomectin" for ecto- and endoparasites control in animals |
| US6265384B1 (en) * | 1999-01-26 | 2001-07-24 | Dale L. Pearlman | Methods and kits for removing, treating, or preventing lice with driable pediculostatic agents |
| GB9916052D0 (en) | 1999-07-08 | 1999-09-08 | Pfizer Ltd | Anthelmintic compositions |
| RU2149007C1 (en) * | 1999-09-28 | 2000-05-20 | Закрытое акционерное общество "Агроветсервис" | Antiparasitic, antibacterial, anti-inflammatory ointment showing prolonged effect |
| ES2310567T3 (en) * | 2000-12-08 | 2009-01-16 | THE GOVERNMENT OF THE USA, AS REPRESENTED BY THE SECRETARY, DEPARTMENT OF HEALTH & HUMAN SERVICES | COMPOUNDS TO COMBAT PESTS. |
| EP1671941A4 (en) | 2003-09-30 | 2007-12-12 | Nissan Chemical Ind Ltd | Substituted benzanilide compound and pest control agent |
| GB0402677D0 (en) | 2003-11-06 | 2004-03-10 | Novartis Ag | Organic compounds |
| KR20060110344A (en) | 2003-12-31 | 2006-10-24 | 쉐링-프라우 리미티드 | Control of parasites in animals by the use of imidazo[1,2-b]pyridazine derivatives |
| WO2005066119A2 (en) | 2003-12-31 | 2005-07-21 | Schering-Plough Ltd. | Antibacterial 1-(4-mono-and di-halomethylsulphonylphenyl)-2-acylamino-3-fluoropropanols and preparation thereof |
| ATE406348T1 (en) | 2004-09-23 | 2008-09-15 | Schering Plough Ltd | FIGHTING PARASITES IN ANIMALS BY USING NEW TRIFLUOROMETHANE SULFONANILIDOXIMETHER DERIVATIVES |
| CA2580247C (en) | 2004-11-09 | 2014-06-17 | Novartis Ag | Process for the preparation of enantiomers of amidoacetonitrile compounds from their racemates |
| WO2006055565A2 (en) | 2004-11-19 | 2006-05-26 | Schering-Plough Ltd. | Control of parasites in animals by the use of parasiticidal 2-phenyl-3-(1h-pyrrol-2-yl) acrylonitrile derivatives |
| ATE378789T1 (en) | 2004-12-15 | 2007-11-15 | Matsushita Electric Ind Co Ltd | SUPPORT FOR GUARANTEED BIT RATE TRAFFIC FOR UPLINK TRANSFERS |
| CA2609574A1 (en) | 2005-06-09 | 2006-12-21 | Schering-Plough Ltd. | Control of parasites in animals by n-[(phenyloxy)phenyl]-1,1,1-trifluoromethanesulfonamide and n-[(phenylsulfanyl)phenyl]-1,1,1-trifluoromethanesulfonamide derivatives |
| US20070010465A1 (en) * | 2005-07-06 | 2007-01-11 | Branimir Sikic | Zosuquidar, daunorubicin, and cytarabine for the treatment of cancer |
| WO2007026965A1 (en) | 2005-09-02 | 2007-03-08 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and harmful organism-controlling agent |
| JP4479917B2 (en) | 2005-09-02 | 2010-06-09 | 日産化学工業株式会社 | Isoxazoline-substituted benzamide compounds and pest control agents |
| JP5342239B2 (en) | 2005-12-06 | 2013-11-13 | ゾエティス・ダブリュー・エルエルシー | Benzidiimidazole non-aqueous composition |
| KR20080080168A (en) | 2005-12-14 | 2008-09-02 | 이 아이 듀폰 디 네모아 앤드 캄파니 | Isoxazolines for controlling invertebrate pests |
| TW200803740A (en) | 2005-12-16 | 2008-01-16 | Du Pont | 5-aryl isoxazolines for controlling invertebrate pests |
| TWI412322B (en) | 2005-12-30 | 2013-10-21 | Du Pont | Isoxazolines for controlling invertebrate pests |
| DE102006015470A1 (en) | 2006-03-31 | 2007-10-04 | Bayer Cropscience Ag | New cyclic enamine ketone derivatives useful for controlling pests, especially insects |
| UA110598C2 (en) | 2006-11-10 | 2016-01-25 | Басф Се | Method of receiving crystalline modification of fipronil |
| JP2008133242A (en) | 2006-11-29 | 2008-06-12 | Nissan Chem Ind Ltd | Method for producing substituted phenyllithium compound |
| CN105640952A (en) * | 2007-06-27 | 2016-06-08 | 杜邦公司 | Animal pest control method |
| TWI556741B (en) * | 2007-08-17 | 2016-11-11 | 英特威特國際股份有限公司 | Isoxazoline compositions and their use as antiparasitics |
| US8044409B2 (en) | 2008-08-11 | 2011-10-25 | Taiwan Semiconductor Manufacturing Company, Ltd. | III-nitride based semiconductor structure with multiple conductive tunneling layer |
-
2008
- 2008-08-04 TW TW097129550A patent/TWI556741B/en active
- 2008-08-15 PT PT88030416T patent/PT2190289E/en unknown
- 2008-08-15 NZ NZ583045A patent/NZ583045A/en unknown
- 2008-08-15 DK DK08803041.6T patent/DK2190289T3/en active
- 2008-08-15 PL PL12188400.1T patent/PL2545777T5/en unknown
- 2008-08-15 KR KR1020107005770A patent/KR20100057066A/en active IP Right Grant
- 2008-08-15 MX MX2013009474A patent/MX351346B/en unknown
- 2008-08-15 AU AU2008290581A patent/AU2008290581C1/en active Active
- 2008-08-15 CA CA2694919A patent/CA2694919C/en active Active
- 2008-08-15 PL PL08803041T patent/PL2190289T3/en unknown
- 2008-08-15 NZ NZ599797A patent/NZ599797A/en unknown
- 2008-08-15 EP EP18205103.7A patent/EP3473101B1/en active Active
- 2008-08-15 US US12/673,722 patent/US20110059988A1/en not_active Abandoned
- 2008-08-15 ES ES12188400T patent/ES2785104T5/en active Active
- 2008-08-15 DK DK18205103.7T patent/DK3473101T3/en active
- 2008-08-15 CN CN200880103027A patent/CN101778566A/en active Pending
- 2008-08-15 CN CN201410011468.0A patent/CN103705509A/en active Pending
- 2008-08-15 BR BR122020005235-0A patent/BR122020005235B1/en active IP Right Grant
- 2008-08-15 KR KR1020167006583A patent/KR101725320B1/en active IP Right Grant
- 2008-08-15 ES ES08803041T patent/ES2417157T3/en active Active
- 2008-08-15 BR BRPI0815227-6A patent/BRPI0815227B1/en active IP Right Grant
- 2008-08-15 RU RU2010109901/15A patent/RU2481837C2/en active
- 2008-08-15 MX MX2010001907A patent/MX2010001907A/en active IP Right Grant
- 2008-08-15 EP EP08803041.6A patent/EP2190289B1/en active Active
- 2008-08-15 KR KR1020177009149A patent/KR101792547B1/en active IP Right Grant
- 2008-08-15 JP JP2010520592A patent/JP5411135B2/en active Active
- 2008-08-15 WO PCT/EP2008/060732 patent/WO2009024541A2/en active Application Filing
- 2008-08-15 EP EP12188400.1A patent/EP2545777B2/en active Active
-
2010
- 2010-10-06 HK HK10109524.4A patent/HK1143037A1/en unknown
-
2012
- 2012-04-26 ZA ZA2012/03080A patent/ZA201203080B/en unknown
-
2013
- 2013-02-18 RU RU2013107094/15A patent/RU2602308C2/en active
- 2013-07-19 AU AU2013207639A patent/AU2013207639B2/en active Active
- 2013-11-06 JP JP2013230009A patent/JP6014006B2/en active Active
-
2014
- 2014-12-22 US US14/579,387 patent/US20150111936A1/en not_active Abandoned
-
2016
- 2016-03-16 JP JP2016052782A patent/JP6251308B2/en active Active
- 2016-07-28 US US15/222,171 patent/US20160332975A1/en not_active Abandoned
- 2016-08-11 RU RU2016133168A patent/RU2715051C2/en active
- 2016-10-05 NO NO2016018C patent/NO2016018I1/en unknown
-
2017
- 2017-07-21 JP JP2017141729A patent/JP6458095B2/en active Active
-
2018
- 2018-01-26 US US15/880,770 patent/US20180155301A1/en not_active Abandoned
-
2020
- 2020-09-15 US US17/021,698 patent/US20210061775A1/en not_active Abandoned
-
2024
- 2024-01-18 US US18/416,336 patent/US20240239759A1/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070066617A1 (en) * | 2004-03-05 | 2007-03-22 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and pesticide |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2190289A2 * |
Cited By (174)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10874645B2 (en) | 2004-03-05 | 2020-12-29 | Nissan Chemical Corporation | Isoxazoline-substituted benzamide compound and pesticide |
| US10045969B2 (en) | 2004-03-05 | 2018-08-14 | Nissan Chemical Industries, Inc. | Isoxazoline-substituted benzamide compound and pesticide |
| US10596157B2 (en) | 2004-03-05 | 2020-03-24 | Nissan Chemical Corporation | Isoxazoline-substituted benzamide compound and pesticide |
| US8119671B2 (en) * | 2007-03-07 | 2012-02-21 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and pest control agent |
| JP2017075152A (en) * | 2007-06-27 | 2017-04-20 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニーE.I.Du Pont De Nemours And Company | Animal pest control method |
| EP2957284B1 (en) | 2007-06-27 | 2018-01-17 | E. I. du Pont de Nemours and Company | Animal pest control method |
| EP2957284A1 (en) * | 2007-06-27 | 2015-12-23 | E. I. du Pont de Nemours and Company | Animal pest control method |
| EP2182945B1 (en) | 2007-06-27 | 2015-08-05 | E. I. du Pont de Nemours and Company | Animal pest control method |
| US11278533B2 (en) | 2007-06-27 | 2022-03-22 | E.I. Du Pont De Nemours And Company | Animal pest control method |
| EP3351243A1 (en) * | 2007-06-27 | 2018-07-25 | E. I. du Pont de Nemours and Company | Animal pest control method |
| JP2015057399A (en) * | 2007-06-27 | 2015-03-26 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニーE.I.Du Pont De Nemours And Company | Method of controlling animal pest |
| EP2545777B2 (en) † | 2007-08-17 | 2023-01-18 | Intervet International B.V. | Use of an isoxazoline composition as antiparasitic |
| EP2545777B1 (en) | 2007-08-17 | 2020-03-11 | Intervet International B.V. | Use of an isoxazoline composition as antiparasitic |
| US9770029B2 (en) | 2008-07-09 | 2017-09-26 | Basf Se | Pesticidal active mixtures comprising isoxazoline compounds I |
| EP2306837B1 (en) | 2008-07-09 | 2017-04-26 | Basf Se | Pesticidal active mixtures comprising isoxazoline compounds i |
| WO2010005048A1 (en) | 2008-07-09 | 2010-01-14 | 日産化学工業株式会社 | Process for production of isoxazoline-substituted benzoic acid amide compound |
| US10231455B2 (en) | 2008-07-09 | 2019-03-19 | Basf Se | Pesticidal active mixtures comprising isoxazoline compounds I |
| US10888094B2 (en) | 2008-07-09 | 2021-01-12 | Basf Se | Pesticidal active mixtures comprising isoxazoline compounds I |
| US9216973B2 (en) | 2008-08-22 | 2015-12-22 | Syngenta Participations Ag | Insecticidal compounds |
| US8822502B2 (en) | 2008-08-22 | 2014-09-02 | Syngenta Crop Protection Llc | Insecticidal compounds |
| US9282742B2 (en) | 2008-09-04 | 2016-03-15 | Syngenta Participations Ag | Insecticidal compounds |
| US8686014B2 (en) | 2008-09-04 | 2014-04-01 | Syngenta Limited | Insecticidal compounds |
| EP3498696A1 (en) | 2008-11-14 | 2019-06-19 | Merial, Inc. | Enantiomerically enriched aryloazol-2-yl cyanoethylamino parasiticidal compounds |
| EP2550962A2 (en) | 2008-11-19 | 2013-01-30 | Merial Limited | Compositions comprising an aryl pyrazole and/or a formamidine, methods and uses thereof |
| WO2010065852A1 (en) | 2008-12-04 | 2010-06-10 | Merial Limited | Dimeric avermectin and milbemycin derivatives |
| US8853410B2 (en) | 2009-04-30 | 2014-10-07 | Basf Se | Process for preparing substituted isoxazoline compounds and their precursors |
| US10750745B2 (en) | 2009-12-01 | 2020-08-25 | Syngenta Crop Protection, Llc | Insecticidal compounds based on isoxazoline derivatives |
| US10206400B2 (en) | 2009-12-01 | 2019-02-19 | Syngenta Participations Ag | Insecticidal compounds based on isoxazoline derivatives |
| US11357231B2 (en) | 2009-12-01 | 2022-06-14 | Syngenta Crop Protection Llc | Insecticidal compounds based on isoxazoline derivatives |
| US8735362B2 (en) | 2009-12-01 | 2014-05-27 | Syngenta Crop Protection, Llc | Insecticidal compounds based on isoxazoline derivatives |
| US9609869B2 (en) | 2009-12-01 | 2017-04-04 | Syngenta Crop Protection, Llc | Insecticidal compounds based on isoxazoline derivatives |
| US9376434B2 (en) | 2009-12-17 | 2016-06-28 | Merial Inc. | Antiparisitic dihydroazole compounds and compositions comprising same |
| US9776999B2 (en) | 2009-12-17 | 2017-10-03 | Merial Inc. | Antiparisitic dihydroazole compounds and compositions comprising same |
| WO2011075591A1 (en) | 2009-12-17 | 2011-06-23 | Merial Limited | Anti parasitic dihydroazole compounds and compositions comprising same |
| EP3560923A1 (en) | 2009-12-17 | 2019-10-30 | Boehringer Ingelheim Animal Health USA Inc. | Anti parasitic dihydroazole compounds and compositions comprising same |
| EP3078664A1 (en) | 2009-12-17 | 2016-10-12 | Merial Inc. | Antiparasitic dihydroazole compositions |
| US8618126B2 (en) | 2009-12-17 | 2013-12-31 | Merial Limited | Antiparisitic dihydroazole compounds and compositions comprising same |
| WO2011123773A1 (en) | 2010-04-02 | 2011-10-06 | Merial Limited | Parasiticidal compositions comprising multiple active agents, methods and uses thereof |
| WO2011154555A1 (en) * | 2010-06-11 | 2011-12-15 | Syngenta Participations Ag | Process for the preparation of dihydropyrrole derivatives |
| JP2013529594A (en) * | 2010-06-11 | 2013-07-22 | シンジェンタ パーティシペーションズ アクチェンゲゼルシャフト | Process for the preparation of dihydropyrrole derivatives |
| CN102939288A (en) * | 2010-06-11 | 2013-02-20 | 先正达参股股份有限公司 | Process for the preparation of dihydropyrrole derivatives |
| US9085541B2 (en) | 2010-06-23 | 2015-07-21 | Basf Se | Process for producing imine compounds for combating invertebrate pests |
| WO2012007426A1 (en) | 2010-07-13 | 2012-01-19 | Basf Se | Azoline substituted isoxazoline benzamide compounds for combating animal pests |
| US9006447B2 (en) | 2010-11-03 | 2015-04-14 | Basf Se | Method for preparing substituted isoxazoline compounds and their precursors 4-chloro, 4-bromo- or 4-iodobenzaldehyde oximes |
| WO2012060317A1 (en) * | 2010-11-04 | 2012-05-10 | 日本曹達株式会社 | Ectoparasite control agent for animals |
| DE102010063691A1 (en) | 2010-12-21 | 2012-06-21 | Bayer Animal Health Gmbh | Ectoparasiticidal drug combinations |
| US9066945B2 (en) | 2010-12-21 | 2015-06-30 | Bayer Intellectual Property Gmbh | Ectoparasiticidal active substance combinations |
| WO2012084852A2 (en) | 2010-12-21 | 2012-06-28 | Bayer Animal Health Gmbh | Ectoparasiticidal active substance combinations |
| EP2658541B1 (en) | 2010-12-27 | 2022-01-26 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| CN105616406A (en) * | 2010-12-27 | 2016-06-01 | 英特维特国际股份有限公司 | Topical localized isoxazoline formulation |
| US9173870B2 (en) | 2010-12-27 | 2015-11-03 | Intervet Inc. | Topical localized isoxazoline formulation |
| WO2012089622A2 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation |
| WO2012089623A1 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| RU2633061C2 (en) * | 2010-12-27 | 2017-10-11 | Интервет Интернэшнл Б.В. | Neprilysin inhibitors |
| WO2012089622A3 (en) * | 2010-12-27 | 2012-11-15 | Intervet International B.V. | Topical localized isoxazoline formulation |
| US10864195B2 (en) | 2010-12-27 | 2020-12-15 | Intervet Inc. | Topical localized isoxazoline formulation |
| AU2016201954B2 (en) * | 2010-12-27 | 2017-11-23 | Intervet International B.V. | Topical localized isoxazoline formulation |
| AU2011351579B2 (en) * | 2010-12-27 | 2016-04-21 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| AU2011351578A1 (en) * | 2010-12-27 | 2013-06-20 | Intervet International B.V. | Topical localized isoxazoline formulation |
| EP2658542B1 (en) | 2010-12-27 | 2022-01-26 | Intervet International B.V. | Topical localized isoxazoline formulation |
| US9532978B2 (en) | 2010-12-27 | 2017-01-03 | Intervet Inc. | Topical localized isoxazoline formulation |
| RU2749733C2 (en) * | 2010-12-27 | 2021-06-16 | Интервет Интернэшнл Б.В. | Composition of isoxazoline for topical and external use |
| AU2017254818B2 (en) * | 2010-12-27 | 2019-08-01 | Intervet International B.V. | Topical localized isoxazoline formulation |
| RU2602189C2 (en) * | 2010-12-27 | 2016-11-10 | Интервет Интернэшнл Б.В. | Topical localized isoxazoline formulation comprising glycofurol |
| US8889710B2 (en) | 2011-05-19 | 2014-11-18 | Eli Lilly And Company | Parasiticidal dihydroisoxazole compounds |
| US8648081B2 (en) | 2011-05-19 | 2014-02-11 | Eli Lilly And Company | Parasiticidal dihydroisoxazole compounds |
| US9255111B2 (en) | 2011-05-19 | 2016-02-09 | Eli Lilly And Company | Parasiticidal dihydroisoxazole compounds |
| WO2013003505A1 (en) | 2011-06-27 | 2013-01-03 | Merial Limited | Amido-pyridyl ether compounds and compositions and their use against parasites |
| EP3788874A1 (en) | 2011-09-12 | 2021-03-10 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising an isoxazoline active agent, method and uses thereof |
| WO2013039948A1 (en) | 2011-09-12 | 2013-03-21 | Merial Limited | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| WO2013074892A1 (en) | 2011-11-17 | 2013-05-23 | Merial Limited | Compositions comprising an aryl pyrazole and a substituted imidazole, methods and uses thereof |
| WO2013078071A1 (en) * | 2011-11-21 | 2013-05-30 | Eli Lilly And Company | Isoxazoline derivatives used in the control of ectoparasites |
| US8546357B2 (en) | 2011-11-21 | 2013-10-01 | Eli Lilly And Company | Boron-containing small molecules for controlling ectoparasites |
| AU2012327171B2 (en) * | 2011-11-21 | 2016-03-31 | Anacor Pharmaceuticals, Inc. | Boron-containing molecules |
| EP2785341A1 (en) * | 2011-11-29 | 2014-10-08 | Novartis AG | Aryl derivatives for controlling ectoparasites |
| WO2013082373A1 (en) | 2011-12-02 | 2013-06-06 | Merial Limited | Long-acting injectable moxidectin formulations and novel moxidectin crystal forms |
| EP3351546A1 (en) | 2011-12-02 | 2018-07-25 | Merial, Inc. | Long-acting injectable moxidectin formulations |
| EP3061454B1 (en) | 2012-02-06 | 2021-03-10 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| US9770440B2 (en) | 2012-04-04 | 2017-09-26 | Intervet Inc. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| US11285101B2 (en) | 2012-04-04 | 2022-03-29 | Intervet Inc. | Soft chewable pharmaceutical products |
| RU2765231C2 (en) * | 2012-04-04 | 2022-01-26 | Интервет Интернэшнл Б.В. | Soft chewable pharmaceutical products |
| WO2013150055A1 (en) | 2012-04-04 | 2013-10-10 | Intervet International B.V. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| AU2013245011B2 (en) * | 2012-04-04 | 2017-11-23 | Intervet International B.V. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| EP2833867B1 (en) | 2012-04-04 | 2017-05-17 | Intervet International B.V. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| US11337917B2 (en) | 2012-04-04 | 2022-05-24 | Intervet Inc. | Soft chewable pharmaceutical products |
| WO2013150052A1 (en) | 2012-04-04 | 2013-10-10 | Intervet International B.V. | Soft chewable pharmaceutical products |
| EP2833866B1 (en) | 2012-04-04 | 2018-11-21 | Intervet International B.V. | Soft chewable pharmaceutical products |
| US11712416B2 (en) | 2012-04-04 | 2023-08-01 | Intervet Inc. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| AU2019246812B2 (en) * | 2012-04-04 | 2021-08-12 | Intervet International B.V. | Soft chewable pharmaceutical products |
| EP3763706A1 (en) | 2012-04-20 | 2021-01-13 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising benzimidazole derivatives, methods and uses thereof |
| WO2013158894A1 (en) | 2012-04-20 | 2013-10-24 | Merial Limited | Parasiticidal compositions comprising benzimidazole derivatives, methods and uses thereof |
| EP3453702A2 (en) | 2012-04-20 | 2019-03-13 | Merial, Inc. | Parasiticidal compositions comprising benzimidazole derivatives, methods and uses thereof |
| GB2509361A (en) * | 2012-11-01 | 2014-07-02 | Sumitomo Chemical Co | Isoxazoline compound for use in controlling an animal ectoparasite |
| US20180263905A1 (en) * | 2012-11-20 | 2018-09-20 | Intervet Inc. | Manufacturing of Semi-Plastic Pharmaceutical Dosage Units |
| EP2922526B1 (en) | 2012-11-20 | 2021-10-27 | Intervet International B.V. | Manufacturing of semi-plastic pharmaceutical dosage units |
| WO2014079825A1 (en) * | 2012-11-20 | 2014-05-30 | Intervet International B.V. | Manufacturing of semi-plastic pharmaceutical dosage units |
| US9532946B2 (en) | 2012-11-20 | 2017-01-03 | Intervet Inc. | Manufacturing of semi-plastic pharmaceutical dosage units |
| EP3428162A1 (en) | 2012-11-20 | 2019-01-16 | Merial Inc. | Anthelmintic compounds and compositions and method of using thereof |
| US10973759B2 (en) | 2012-11-20 | 2021-04-13 | Intervet Inc. | Manufacturing of semi-plastic pharmaceutical dosage units |
| US20190336442A1 (en) * | 2012-11-20 | 2019-11-07 | Intervet Inc. | Manufacturing of Semi-Plastic Pharmaceutical Dosage Units |
| EP3964198A1 (en) | 2012-11-20 | 2022-03-09 | Intervet International B.V. | Manufacturing of semi-plastic pharmaceutical dosage units |
| WO2014081697A2 (en) | 2012-11-20 | 2014-05-30 | Merial Limited | Anthelmintic compounds and compositions and method of using thereof |
| US11911503B2 (en) | 2012-11-20 | 2024-02-27 | Intervet Inc. | Manufacturing of semi-plastic pharmaceutical dosage units |
| AU2018220125B2 (en) * | 2012-11-20 | 2020-04-02 | Intervet International B.V. | Manufacturing of semi-plastic pharmaceutical dosage units |
| WO2014122083A1 (en) | 2013-02-06 | 2014-08-14 | Bayer Cropscience Ag | Halogen-substituted pyrazol derivatives as pest-control agents |
| US9314478B2 (en) | 2013-03-15 | 2016-04-19 | Argenta Manufacturing Limited | Method of making an anhydrous, fat soluble, chewable drug delivery formulation |
| EP3733664A1 (en) | 2013-11-01 | 2020-11-04 | Boehringer Ingelheim Animal Health USA Inc. | Antiparisitic and pesticidal isoxazoline compounds |
| WO2015066277A1 (en) | 2013-11-01 | 2015-05-07 | Merial Limited | Antiparisitic and pesticidal isoxazoline compounds |
| WO2015086551A1 (en) | 2013-12-10 | 2015-06-18 | Intervet International B.V. | Antiparasitic use of isoxazoline compounds |
| US9655884B2 (en) | 2013-12-10 | 2017-05-23 | Intervet Inc. | Antiparasitic use of isoxazoline compounds |
| EP3079474B1 (en) | 2013-12-10 | 2019-07-03 | Intervet International B.V. | Antiparasitic use of isoxazoline compounds |
| AU2014363695B2 (en) * | 2013-12-10 | 2018-12-13 | Intervet International B.V | Antiparasitic use of isoxazoline compounds |
| RU2673723C1 (en) * | 2013-12-10 | 2018-11-29 | Интервет Интернэшнл Б.В. | Antiparasitic use of isoxazoline compounds |
| WO2015091900A1 (en) | 2013-12-20 | 2015-06-25 | Intervet International B.V. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| US11304934B2 (en) | 2013-12-20 | 2022-04-19 | Intervet Inc. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| US10272071B2 (en) | 2013-12-20 | 2019-04-30 | Intervet Inc. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| EP3878281A1 (en) | 2013-12-20 | 2021-09-15 | Intervet International B.V. | Use of isoxazoline compounds in poultry |
| US11883530B2 (en) | 2013-12-20 | 2024-01-30 | Intervet Inc. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| US20160317439A1 (en) * | 2013-12-20 | 2016-11-03 | Intervet Inc. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| RU2688919C1 (en) * | 2013-12-20 | 2019-05-23 | Интервет Интернэшнл Б.В. | Isoxazoline compositions |
| US20190192486A1 (en) * | 2013-12-20 | 2019-06-27 | Intervet Inc. | Use of Isoxazoline Derivatives for the Treatment or Prevention of Arthropod Infestations in Poultry |
| EP4306168A2 (en) | 2013-12-20 | 2024-01-17 | Intervet International B.V. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| US10653675B2 (en) | 2013-12-20 | 2020-05-19 | Intervet Inc. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| US10456358B2 (en) | 2013-12-20 | 2019-10-29 | Intervet Inc. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| WO2015101622A1 (en) | 2014-01-03 | 2015-07-09 | Bayer Cropscience Ag | Novel pyrazolyl-heteroarylamides as pesticides |
| WO2015179414A1 (en) | 2014-05-19 | 2015-11-26 | Merial, Inc. | Anthelmintic compounds |
| WO2015196014A1 (en) | 2014-06-19 | 2015-12-23 | Merial, Inc. | Parasiticidal compositions comprising indole derivatives, methods and uses thereof |
| EP3517524A1 (en) | 2014-06-19 | 2019-07-31 | Merial Inc. | Parasiticidal compositions comprising indole derivatives, methods and uses thereof |
| WO2016008830A1 (en) | 2014-07-15 | 2016-01-21 | Bayer Cropscience Aktiengesellschaft | Aryl-triazolyl pyridines as pest control agents |
| WO2016069983A1 (en) | 2014-10-31 | 2016-05-06 | Merial, Inc. | Parasiticidal composition comprising fipronil |
| WO2016102437A1 (en) * | 2014-12-22 | 2016-06-30 | Intervet International B.V. | Use of isoxazoline compounds for treating demodicosis |
| EP4008329A1 (en) | 2014-12-22 | 2022-06-08 | Intervet International B.V. | Isoxazoline compounds for use in treating demodicosis |
| RU2709198C2 (en) * | 2014-12-22 | 2019-12-17 | Интервет Интернэшнл Б.В. | Use of isoxazoline compounds for demodecosis treatment |
| AU2015371175B2 (en) * | 2014-12-22 | 2021-03-04 | Intervet International B.V. | Use of isoxazoline compounds for treating demodicosis |
| US10799483B2 (en) | 2014-12-22 | 2020-10-13 | Intervet Inc. | Use of isoxazoline compounds for treating demodicosis |
| US20180318265A1 (en) * | 2014-12-22 | 2018-11-08 | Intervet Inc. | Use of isoxazoline compounds for treating demodicosis |
| US10561641B2 (en) | 2015-02-26 | 2020-02-18 | Boehringer Ingelheim Animal Health USA Inc. | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| US11484528B2 (en) | 2015-02-26 | 2022-11-01 | Boehringer Ingelheim Animal Health USA Inc. | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| US11179372B2 (en) | 2015-06-23 | 2021-11-23 | Intervet Inc. | Isoxazoline solution containing vitamin E for use with sanitized drinking water |
| US20190343808A1 (en) * | 2015-06-23 | 2019-11-14 | Intervet Inc. | Isoxazoline solution containing vitamin e for use with sanitized drinking water |
| US11878006B2 (en) | 2015-06-23 | 2024-01-23 | Intervet Inc. | Isoxazoline solution containing vitamin E for use with sanitized drinking water |
| US20210354283A1 (en) * | 2015-06-23 | 2021-11-18 | Intervet Inc. | Isoxazoline solution containing vitamin e for use with sanitized drinking water |
| US11497732B2 (en) | 2016-02-24 | 2022-11-15 | Boehringer Ingelheim Animal Health USA Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| WO2017147352A1 (en) | 2016-02-24 | 2017-08-31 | Merial, Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| EA036716B1 (en) * | 2016-02-24 | 2020-12-11 | БЁРИНГЕР ИНГЕЛЬХАЙМ ЭНИМАЛ ХЕЛТ ЮЭсЭй ИНК. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| EP3763211A1 (en) | 2016-02-24 | 2021-01-13 | Boehringer Ingelheim Animal Health USA Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| WO2018039508A1 (en) | 2016-08-25 | 2018-03-01 | Merial, Inc. | Method for reducing unwanted effects in parasiticidal treatments |
| US11858904B2 (en) | 2017-11-07 | 2024-01-02 | Intervet Inc. | Process for preparing large size isoxazoline particles |
| WO2019091936A1 (en) | 2017-11-07 | 2019-05-16 | Intervet International B.V. | Injectable isoxazoline pharmaceutical compositions and their use against parasite infestation |
| WO2019091940A1 (en) | 2017-11-07 | 2019-05-16 | Intervet International B.V. | Process for preparing large size isoxazoline particles |
| EP4316522A2 (en) | 2017-11-07 | 2024-02-07 | Intervet International B.V. | Injectable isoxazoline pharmaceutical compositions and their use against parasite infestation |
| WO2019115492A1 (en) | 2017-12-12 | 2019-06-20 | Intervet International B.V. | Implantable isoxazoline pharmaceutical compositions and uses thereof |
| US11648238B2 (en) | 2017-12-12 | 2023-05-16 | Intervet Inc. | Implantable isoxazoline pharmaceutical compositions and uses thereof |
| US11690827B2 (en) | 2017-12-15 | 2023-07-04 | Tarsus Pharmaceuticals, Inc. | Methods for treating ocular Demodex using lotilaner formulations |
| US11752137B2 (en) | 2017-12-15 | 2023-09-12 | Tarsus Pharmaceuticals, Inc. | Ophthalmic compositions for treating ocular Demodex using lotilaner formulations |
| US11690826B2 (en) | 2017-12-15 | 2023-07-04 | Tarsus Pharmaceuticals, Inc. | Methods for treating demodex blepharitis using lotilaner formulations |
| US11197847B2 (en) | 2017-12-15 | 2021-12-14 | Tarsus Pharmaceuticals, Inc. | Isoxazoline parasiticide formulations and methods for treating blepharitis |
| US10835517B2 (en) | 2017-12-15 | 2020-11-17 | Tarsus Pharmaceuticals, Inc. | Methods for treating ocular demodex using isoxazoline parasiticide formulations |
| WO2019122324A1 (en) | 2017-12-21 | 2019-06-27 | Intervet International B.V. | Antiparasitic pour-on compositions |
| WO2019157241A1 (en) | 2018-02-08 | 2019-08-15 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising eprinomectin and praziquantel, methods and uses thereof |
| WO2020150032A1 (en) | 2019-01-16 | 2020-07-23 | Boehringer Ingelheim Animal Health USA Inc. | Topical compositions comprising a neonicotinoid and a macrocyclic lactone, methods and uses thereof |
| WO2020180635A1 (en) | 2019-03-01 | 2020-09-10 | Boehringer Ingelheim Animal Health USA Inc. | Injectable clorsulon compositions, methods and uses thereof |
| WO2020202111A1 (en) * | 2019-04-04 | 2020-10-08 | Tarsus Pharmaceuticals, Inc. | Systemic isoxazoline parasiticides for vector-borne and viral disease treatment or prophylaxis |
| WO2020225143A1 (en) | 2019-05-03 | 2020-11-12 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| US20220242835A1 (en) * | 2019-07-01 | 2022-08-04 | Shenyang University Of Chemical Technology | Isoxazoline compounds and use thereof |
| WO2021013825A1 (en) | 2019-07-22 | 2021-01-28 | Intervet International B.V. | Soft chewable veterinary dosage form |
| WO2021122513A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Composition for lice control |
| WO2021122521A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Composition for lice control in poultry |
| WO2021122515A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Parasite control in ruminants |
| WO2021233967A1 (en) | 2020-05-20 | 2021-11-25 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| WO2021242481A1 (en) | 2020-05-28 | 2021-12-02 | Boehringer Ingelheim Animal Health USA Inc. | Bi-modal release intra-ruminal capsule device and methods of use thereof |
| WO2022140728A1 (en) | 2020-12-21 | 2022-06-30 | Boehringer Ingelheim Animam Health Usa Inc. | Parasiticidal collar comprising isoxazoline compounds |
| WO2022226660A1 (en) * | 2021-04-30 | 2022-11-03 | Ohalloran John | Use of isoxazoline for protection against parasitic pests in fish |
| WO2022269042A1 (en) | 2021-06-25 | 2022-12-29 | Intervet International B.V. | Palatable veterinary compositions |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20240239759A1 (en) | Isoxazoline compositions and their use as antiparasitics | |
| AU2022203370B2 (en) | Isoxazoline compositions and their use as antiparasitics | |
| RU2816320C2 (en) | Isoxazoline compositions and use thereof as antiparasitic agents | |
| AU2018203263A1 (en) | Isoxazoline compositions and their use as antiparasitics | |
| BR122020024991B1 (en) | USE OF ISOXAZOLINE |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200880103027.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 08803041 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008803041 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008290581 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2694919 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 583045 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12673722 Country of ref document: US Ref document number: 2010520592 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2010/001907 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2008290581 Country of ref document: AU Date of ref document: 20080815 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20107005770 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010109901 Country of ref document: RU |
|
| ENP | Entry into the national phase |
Ref document number: PI0815227 Country of ref document: BR Kind code of ref document: A2 Effective date: 20100211 |