WO2007127665A2 - Novel formulations which stabilize and inhibit precipitation of immunogenic compositions - Google Patents
Novel formulations which stabilize and inhibit precipitation of immunogenic compositions Download PDFInfo
- Publication number
- WO2007127665A2 WO2007127665A2 PCT/US2007/066959 US2007066959W WO2007127665A2 WO 2007127665 A2 WO2007127665 A2 WO 2007127665A2 US 2007066959 W US2007066959 W US 2007066959W WO 2007127665 A2 WO2007127665 A2 WO 2007127665A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formulation
- polysaccharide
- polypeptide
- crm
- pneumoniae serotype
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/02—Bacterial antigens
- A61K39/095—Neisseria
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/02—Bacterial antigens
- A61K39/085—Staphylococcus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/02—Bacterial antigens
- A61K39/09—Lactobacillales, e.g. aerococcus, enterococcus, lactobacillus, lactococcus, streptococcus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/02—Bacterial antigens
- A61K39/09—Lactobacillales, e.g. aerococcus, enterococcus, lactobacillus, lactococcus, streptococcus
- A61K39/092—Streptococcus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/02—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/16—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing nitrogen, e.g. nitro-, nitroso-, azo-compounds, nitriles, cyanates
- A61K47/18—Amines; Amides; Ureas; Quaternary ammonium compounds; Amino acids; Oligopeptides having up to five amino acids
- A61K47/183—Amino acids, e.g. glycine, EDTA or aspartame
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/64—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent
- A61K47/646—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent the entire peptide or protein drug conjugate elicits an immune response, e.g. conjugate vaccines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/555—Medicinal preparations containing antigens or antibodies characterised by a specific combination antigen/adjuvant
- A61K2039/55505—Inorganic adjuvants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/555—Medicinal preparations containing antigens or antibodies characterised by a specific combination antigen/adjuvant
- A61K2039/55511—Organic adjuvants
- A61K2039/55583—Polysaccharides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/57—Medicinal preparations containing antigens or antibodies characterised by the type of response, e.g. Th1, Th2
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/60—Medicinal preparations containing antigens or antibodies characteristics by the carrier linked to the antigen
- A61K2039/6018—Lipids, e.g. in lipopeptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/60—Medicinal preparations containing antigens or antibodies characteristics by the carrier linked to the antigen
- A61K2039/6031—Proteins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/60—Medicinal preparations containing antigens or antibodies characteristics by the carrier linked to the antigen
- A61K2039/6031—Proteins
- A61K2039/6037—Bacterial toxins, e.g. diphteria toxoid [DT], tetanus toxoid [TT]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/60—Medicinal preparations containing antigens or antibodies characteristics by the carrier linked to the antigen
- A61K2039/6031—Proteins
- A61K2039/6081—Albumin; Keyhole limpet haemocyanin [KLH]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/70—Multivalent vaccine
Definitions
- the present invention generally relates to the fields of immunology, bacteriology, vaccine formulation, protein stability and process development. More particularly, the invention relates to novel formulations which inhibit precipitation of immunogenic compositions.
- an immunogenic composition e.g., a protein immunogen, a polysaccharide-protein conjugate
- an immunogenic composition must appear fresh, elegant and professional when administered to a patient. Any changes in stability and/or physical appearance of the immunogenic composition, such as color change, clouding or haziness, may cause a patient or consumer to lose confidence in the product.
- uniformity of dose content of the active ingredient e.g., a polysaccharide-protein conjugate
- the immunogenic composition must be active throughout its "expected" shelf life, wherein any breakdown of the immunogenic composition to an inactive or otherwise undesired form (e.g., an aggregate) lowers the total concentration of the product.
- a particular immunogenic composition e.g., a protein immunogen, a polysaccharide- protein conjugate
- Ho et ai, 2001 Ho et ai, 2002; Bolgiano et ai, 2001
- stability analysis of meningococcal C (MenC) polysaccharides and Haemophilus influenzae type b (Hib) polysaccharides, conjugated to either a tetanus toxoid (TT) or a CRM 197 carrier protein revealed different stability profiles dependent on the carrier protein (Ho et al., 2002).
- MenC-CRM 197 conjugates from two different manufacturers were analyzed (Ho et al., 2001 ), wherein the MenC-
- CRM 197 conjugates differed in their conjugation chemistry and length of conjugate polysaccharide (both having the same carrier protein, CRM 197 ).
- conjugation chemistry e.g., reductive amination either directly or via a chemical spacer group
- number of conjugation sites e.g., polysaccharide chain length
- pH, storage buffer, storage temperature(s) and freeze/thaw cycles also influence the stability of an immunogenic composition.
- Such considerations include, but are not limited to, chemical stability of the immunogenic composition (e.g., hydrolysis of saccharides, de-polymerization of polysaccharides, proteolysis or fragmentation of proteins), physical/thermal stability of the immunogenic composition (e.g., aggregation, precipitation, adsorption), compatibility of the immunogenic composition with the container/closure system, interactions between immunogenic composition and inactive ingredients (e.g., buffers, salts, excipients, cryoprotectants), the manufacturing process, the dosage form (e.g., lyophilized, liquid), the environmental conditions encountered during shipping, storage and handling (e.g., temperature, humidity, shear forces), and the length of time between manufacture and usage.
- chemical stability of the immunogenic composition e.g., hydrolysis of saccharides, de-polymerization of polysaccharides, proteolysis or fragmentation of proteins
- physical/thermal stability of the immunogenic composition e.g., aggregation, precipitation, ad
- silicone oil which induces protein secondary and tertiary conformational changes, might be responsible for the aggregation/precipitation seen in certain protein pharmaceutical preparations (Jones et al., 2005).
- several reports in the 1980s implicated the release of silicone oil from disposable plastic syringes as the causative agent in the aggregation of human insulin (Chantelau and Berger, 1985; Chantelau et al., 1986; Chantelau, 1989; Bernstein, 1987; Baldwin, 1988; Collier and Dawson, 1985). Chantelau et al.
- silicone oil is a necessary component of plastic syringes, as it serves to lubricate the rubber plunger and facilitate transfer of the plunger down the syringe barrel (i.e., silicone oil improves the syringeability of the formulation).
- silicone oil is not limited to syringes, as it is used as a coating for glass vials to minimize protein adsorption, as a lubricant to prevent conglomeration of rubber stoppers during filing procedures, as a lubricant critical to the processability/machinability of glass and elastomeric closures and as a lubricant to ease needle penetration of vial rubber stoppers.
- the siliconization of syringes, glass vials, rubber stoppers and the like is not a well controlled nor standardized process, and as such, there is a high degree of variability of the silicone oil content from one lot to another.
- the present invention broadly relates to novel formulations which stabilize and inhibit precipitation of immunogenic compositions. More specifically in certain embodiments, the present invention is directed to novel formulations which inhibit precipitation of immunogenic compositions comprised in container means. In one specific embodiment, the invention is directed to novel formulations which stabilize immunogenic compositions against silicone oil interactions, shear forces, shipping agitation, and the like. Thus, in certain embodiments, the invention is directed to formulations which stabilize a polysaccharide-protein conjugate, the formulation comprising (i) a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 7.5, (ii) a surfactant and (iii) one or more polysaccharide-protein conjugates.
- the polysaccharide-protein conjugate formulation is comprised in a container means.
- the container means is selected from one or more of the group consisting of a vial, a vial stopper, a vial closure, a glass closure, a rubber closure, a plastic closure, a syringe, a syringe stopper, a syringe plunger, a flask, a beaker, a graduated cylinder, a fermentor, a bioreactor, tubing, a pipe, a bag, a jar, an ampoule, a cartridge and a disposable pen.
- the container means is siliconized.
- the pH buffered saline solution of the formulations has a pH of 5.5 to 7.5.
- the buffer is phosphate, succinate, histidine or citrate.
- the buffer is succinate at a final concentration of 1 mM to 10 mM and pH 5.8 to 6.0.
- the final concentration of the succinate buffer is 5 mM.
- the salt in the pH buffered saline solution comprises magnesium chloride, potassium chloride, sodium chloride or a combination thereof.
- the salt in the pH buffered saline solution is sodium chloride.
- the surfactant of the formulations is selected from the group consisting of polysorbate 20 (TweenTM20), polysorbate 40 (TweenTM40), polysorbate 60 (TweenTM60), polysorbate 65 (TweenTM65), polysorbate 80
- the surfactant is polysorbate 80.
- the final concentration of the polysorbate 80 in formulation is at least 0.01 % to 10% polysorbate 80 weight/volume of the formulation.
- the final concentration of the polysorbate 80 in the formulation is 0.01 % polysorbate 80 weight/volume of the formulation. In yet other embodiments, the final concentration of the polysorbate 80 in the formulation is 0.05% polysorbate 80 weight/volume of the formulation. In another embodiment, the final concentration of the polysorbate 80 in the formulation is 0.1 % polysorbate 80 weight/volume of the formulation. In certain other embodiments, the final concentration of the polysorbate 80 in the formulation is 1.0% polysorbate 80 weight/volume of the formulation. In yet other embodiments, the final concentration of the polysorbate 80 in the formulation is 10.0% polysorbate 80 weight/volume of the formulation.
- the polysaccharide-protein conjugate comprises one or more pneumococcal polysaccharides.
- the one or more pneumococcal polysaccharides are a S. pneumoniae serotype 4 polysaccharide, a S. pneumoniae serotype 6B polysaccharide, a S. pneumoniae serotype 9V polysaccharide, a S. pneumoniae serotype 14 polysaccharide, a S. pneumoniae serotype 18C polysaccharide, a S. pneumoniae serotype 19F polysaccharide, a S. pneumoniae serotype 23F polysaccharide, a S. pneumoniae serotype 1 polysaccharide, a S.
- the protein of the polysaccharide-protein conjugate formulation is selected from the group consisting of CRM 197 , a tetanus toxoid, a cholera toxoid, a pertussis toxoid, an E.
- coli heat labile toxoid LT
- pneumolysin toxoid pneumococcal surface protein A
- pneumococcal adhesin protein A PsaA
- C5a peptidase from Streptococcus Haemophilus influenzae
- BSA purified protein derivative of tuberculin
- the polysaccharide-protein conjugate formulation is a 7-valent pneumococcal conjugate (7vPnC) formulation comprising a S. pneumoniae serotype 4 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 6B polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 9V polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 14 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 18C polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 19F polysaccharide conjugated to a CRM 197 polypeptide and a 7-valent pneumococcal conjugate (7vPnC) formulation comprising a S. pneumoniae serotype 4 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumonia
- S. pneumoniae serotype 23F polysaccharide conjugated to a CRM 197 polypeptide S. pneumoniae serotype 23F polysaccharide conjugated to a CRM 197 polypeptide.
- the polysaccharide-protein conjugate formulation is a 13-valent pneumococcal conjugate (13vPnC) formulation comprising a S. pneumoniae serotype 4 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 6B polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 9V polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 14 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 18C polysaccharide conjugated to a CRM 197 polypeptide, a S.
- 13vPnC 13-valent pneumococcal conjugate
- pneumoniae serotype 19F polysaccharide conjugated to a CRM 197 polypeptide a S. pneumoniae serotype 23F polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 1 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 3 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 5 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 6A polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 7F polysaccharide conjugated to a CRM 197 polypeptide and a
- S. pneumoniae serotype 19A polysaccharide conjugated to a CRM 197 polypeptide S. pneumoniae serotype 19A polysaccharide conjugated to a CRM 197 polypeptide.
- the formulation further comprises one or more meningococcal polysaccharides, one or more meningococcal antigenic proteins, or a combination thereof. In yet other embodiments, the formulation further comprises one or more streptococcal polysaccharides, one or more streptococcal antigenic proteins, or a combination thereof.
- the formulation further comprises one or more adjuvants.
- adjuvants are described herein below.
- the invention is directed to formulations which stabilize a streptococcal C5a peptidase (SCP) composition, the formulation comprising (i) a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 6.5,
- SCP formulation is comprised in a container means.
- the container means is selected from one or more of the group consisting of a vial, a vial stopper, a vial closure, a glass closure, a rubber closure, a plastic closure, a syringe, a syringe stopper, a syringe plunger, a flask, a beaker, a graduated cylinder, a fermentor, a bioreactor, tubing, a pipe, a bag, a jar, an ampoule, a cartridge and a disposable pen.
- the pH buffered saline solution of the formulation has a pH of 5.5 to 7.5.
- the buffer is succinate, histidine, phosphate or citrate.
- the buffer is succinate at a final concentration of 1 mM to 10 mM and pH 5.8 to 6.0.
- the final concentration of the succinate buffer is 5 mM.
- the salt in the pH buffered saline solution comprises magnesium chloride, potassium chloride, sodium chloride or a combination thereof.
- the surfactant in the formulations is selected from the group consisting of polysorbate 20 (TweenTM20), polysorbate 40 (TweenTM40), polysorbate 60 (TweenTM60), polysorbate 65 (TweenTM65), polysorbate 80 (TweenTM80), polysorbate 85 (TweenTM85), TritonTM N-101 , TritonTM X-100, oxtoxynol 40, nonoxynol-9, triethanolamine, triethanolamine polypeptide oleate, polyoxyethylene-660 hydroxystearate (PEG-15, Solutol H 15), polyoxyethylene-35- ricinoleate (Cremophor ELTM), soy lecithin and a poloxamer.
- the surfactant is polysorbate 80.
- the final concentration of the polysorbate 80 in formulation is 0.01 % to 10% polysorbate 80 weight/volume of the formulation. In yet other embodiments, the final concentration of the polysorbate 80 in the formulation is 0.01 % polysorbate 80 weight/volume of the formulation. In other embodiments, the final concentration of the polysorbate 80 in the formulation is 0.05% polysorbate 80 weight/volume of the formulation. In yet other embodiments, the final concentration of the polysorbate 80 in the formulation is 0.1 % polysorbate 80 weight/volume of the formulation. In another embodiment, the final concentration of the polysorbate 80 in the formulation is 1.0% polysorbate 80 weight/volume of the formulation. In yet another embodiment, the final concentration of the polysorbate 80 in the formulation is 10.0% polysorbate 80 weight/volume of the formulation.
- the SCP composition further comprises one or more polypeptides selected from the group consisting of a streptococcal polypeptide, a pneumococcal polypeptide, a meningococcal polypeptide and a staphylococcal polypeptide.
- the SCP composition further comprises one or more polysaccharides selected from the group consisting of a streptococcal polysaccharide, a pneumococcal polysaccharide, a meningococcal polysaccharide and a staphylococcal polysaccharide.
- the formulation further comprises one or more adjuvants.
- adjuvants are described herein below.
- the invention is directed to formulations which inhibit silicone induced precipitation of a polysaccharide-protein conjugate comprised in a siliconized container means, the formulation comprising (i) a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 7.5, (ii) an aluminum salt and (iii) one or more polysaccharide-protein conjugates.
- the siliconized container means is selected from one or more of the group consisting of a vial, a vial stopper, a vial closure, a glass closure, a rubber closure, a plastic closure, a syringe, a syringe stopper, a syringe plunger, a flask, a beaker, a graduated cylinder, a fermentor, a bioreactor, tubing, a pipe, a bag, a jar, an ampoule, a cartridge and a disposable pen.
- the pH buffered saline solution in the formulations has a pH of 5.5 to 7.5.
- the buffer in the formulations is phosphate, succinate, histidine or citrate.
- the buffer is succinate at a final concentration of 1 mM to 10 mM and pH 5.8 to 6.0.
- the final concentration of the succinate buffer is 5 mM.
- the salt in the pH buffered saline solution comprises magnesium chloride, potassium chloride, sodium chloride or a combination thereof.
- the salt in the pH buffered saline solution is sodium chloride.
- the aluminum salt is aluminum hydroxide, aluminum phosphate or aluminum sulfate. In one specific embodiment, the aluminum salt is aluminum phosphate.
- the formulation further comprises polysorbate 80 (TweenTM80).
- the final concentration of the polysorbate 80 in formulation is at least 0.01 % to 10% polysorbate 80 weight/volume of the formulation.
- the polysaccharide-protein conjugate comprises one or more pneumococcal polysaccharides.
- the one or more pneumococcal polysaccharides are a S. pneumoniae serotype 4 polysaccharide, a S. pneumoniae serotype 6B polysaccharide, a S. pneumoniae serotype 9V polysaccharide, a S. pneumoniae serotype 14 polysaccharide, a S. pneumoniae serotype 18C polysaccharide, a S. pneumoniae serotype 19F polysaccharide, a S. pneumoniae serotype 23F polysaccharide, a S. pneumoniae serotype 1 polysaccharide, a S.
- the protein of the polysaccharide-protein conjugate formulation is selected from the group consisting of CRM 197 , a tetanus toxoid, a cholera toxoid, a pertussis toxoid, an E.
- coli heat labile toxoid LT
- pneumolysin toxoid pneumococcal surface protein A
- pneumococcal adhesin protein A PsaA
- KLH keyhole limpet haemocyanin
- BSA purified protein derivative of tuberculin
- the polysaccharide-protein conjugate formulation is a 7-valent pneumococcal conjugate (7vPnC) formulation comprising a
- S. pneumoniae serotype 4 polysaccharide conjugated to a CRM 197 polypeptide
- a S. pneumoniae serotype 6B polysaccharide conjugated to a CRM 197 polypeptide a S. pneumoniae serotype 9V polysaccharide conjugated to a CRM 197 polypeptide
- a S. pneumoniae serotype 14 polysaccharide conjugated to a CRM 197 polypeptide
- S. pneumoniae serotype 19F polysaccharide conjugated to a CRM 197 polypeptide
- a S. pneumoniae serotype 23F polysaccharide conjugated to a CRM 197 polypeptide a S. pneumoniae serotype 23F polysaccharide conjugated to a CRM 197 polypeptide.
- the polysaccharide-protein conjugate formulation is a 13-valent pneumococcal conjugate (13vPnC) formulation comprising a S. pneumoniae serotype 4 polysaccharide conjugated to a CRM 197 polypeptide, a
- S. pneumoniae serotype 6B polysaccharide conjugated to a CRM 197 polypeptide a S. pneumoniae serotype 9V polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 14 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 18C polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 19F polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 23F polysaccharide conjugated to a CRM 197 polypeptide, a S.
- the formulation further comprises one or more meningococcal polysaccharides, one or more meningococcal antigenic proteins, or a combination thereof.
- the formulation further comprises one or more streptococcal polysaccharides, one or more streptococcal antigenic proteins, or a combination thereof.
- the formulation further comprises one or more adjuvants.
- adjuvants are described herein below.
- the present invention is directed to formulations which inhibit silicone induced precipitation of a streptococcal C5a peptidase (SCP) composition comprised in a siliconized container means, the formulation comprising
- the container means is selected from one or more of the group consisting of a vial, a vial stopper, a vial closure, a glass closure, a rubber closure, a plastic closure, a syringe, a syringe stopper, a syringe plunger, a flask, a beaker, a graduated cylinder, a fermentor, a bioreactor, tubing, a pipe, a bag, a jar, an ampoule, a cartridge and a disposable pen.
- the pH buffered saline solution of the formulation has a pH of 5.5 to 7.5.
- the buffer is succinate, histidine, phosphate or citrate.
- the buffer is succinate at a final concentration of 1 mM to 10 mM and pH 5.8 to 6.0.
- the salt in the pH buffered saline solution comprises magnesium chloride, potassium chloride, sodium chloride or a combination thereof.
- the formulation further comprises polysorbate
- the final concentration of the polysorbate 80 in the formulation is 0.01 % to 10% polysorbate 80 weight/volume of the formulation.
- the SCP composition further comprises one or more polypeptides selected from the group consisting of a streptococcal polypeptide, a pneumococcal polypeptide, a meningococcal polypeptide and a staphylococcal polypeptide.
- the SCP composition further comprises one or more polysaccharides selected from the group consisting of a streptococcal polysaccharide, a pneumococcal polysaccharide, a meningococcal polysaccharide and a staphylococcal polysaccharide.
- the formulation further comprises one or more adjuvants. Exemplary suitable adjuvants are described herein below.
- the invention is directed to formulations which stabilize a N. meningitidis 2086 protein composition, the formulation comprising (i) a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 6.5, (ii) a surfactant and (iii) a N. meningitidis 2086 protein.
- a N. meningitidis 2086 protein Exemplary N. meningitidis 2086 proteins are described herein below.
- the N. meningitidis 2086 protein formulation is comprised in a container means.
- the container means is selected from one or more of the group consisting of a vial, a vial stopper, a vial closure, a glass closure, a rubber closure, a plastic closure, a syringe, a syringe stopper, a syringe plunger, a flask, a beaker, a graduated cylinder, a fermentor, a bioreactor, tubing, a pipe, a bag, a jar, an ampoule, a cartridge and a disposable pen.
- the pH buffered saline solution of the formulation has a pH of 5.5 to 7.5.
- the buffer is succinate, histidine, phosphate or citrate.
- the buffer is succinate at a final concentration of 1 mM to 10 mM and pH 5.8 to 6.0.
- the final concentration of the succinate buffer is 5 mM.
- the salt in the pH buffered saline solution comprises magnesium chloride, potassium chloride, sodium chloride or a combination thereof.
- the surfactant in the formulations is selected from the group consisting of polysorbate 20 (TweenTM20), polysorbate 40 (TweenTM40), polysorbate 60 (TweenTM60), polysorbate 65 (TweenTM65), polysorbate 80 (TweenTM80), polysorbate 85 (TweenTM85), TritonTM N-101 , TritonTM X-100, oxtoxynol 40, nonoxynol-9, triethanolamine, triethanolamine polypeptide oleate, polyoxyethylene-660 hydroxystearate (PEG-15, Solutol H 15), polyoxyethylene-35- ricinoleate (Cremophor ELTM), soy lecithin and a poloxamer.
- the surfactant is polysorbate 80.
- the final concentration of the polysorbate 80 in the formulation is 0.01 % to 10% polysorbate 80 weight/volume of the formulation. In yet other embodiments, the final concentration of the polysorbate 80 in the formulation is 0.01 % polysorbate 80 weight/volume of the formulation. In other embodiments, the final concentration of the polysorbate 80 in the formulation is 0.05% polysorbate 80 weight/volume of the formulation. In yet other embodiments, the final concentration of the polysorbate 80 in the formulation is 0.1 % polysorbate 80 weight/volume of the formulation. In another embodiment, the final concentration of the polysorbate 80 in the formulation is 1.0% polysorbate 80 weight/volume of the formulation. In yet another embodiment, the final concentration of the polysorbate 80 in the formulation is 10.0% polysorbate 80 weight/volume of the formulation.
- the N. meningitidis 2086 protein composition further comprises one or more polypeptides selected from the group consisting of a streptococcal polypeptide, a pneumococcal polypeptide, a meningococcal polypeptide and a staphylococcal polypeptide.
- the N. meningitidis 2086 protein composition further comprises one or more polysaccharides selected from the group consisting of a streptococcal polysaccharide, a pneumococcal polysaccharide, a meningococcal polysaccharide and a staphylococcal polysaccharide.
- the formulation further comprises one or more adjuvants.
- adjuvants are described herein below.
- the present invention is directed to formulations which inhibit silicone induced precipitation of a N. meningitidis 2086 protein composition comprised in a siliconized container means, the formulation comprising (i) a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 6.5, (ii) an aluminum salt and (iii) a N. meningitidis 2086 protein.
- the container means is selected from one or more of the group consisting of a vial, a vial stopper, a vial closure, a glass closure, a rubber closure, a plastic closure, a syringe, a syringe stopper, a syringe plunger, a flask, a beaker, a graduated cylinder, a fermentor, a bioreactor, tubing, a pipe, a bag, a jar, an ampoule, a cartridge and a disposable pen.
- the pH buffered saline solution of the formulation has a pH of 5.5 to 7.5.
- the buffer is succinate, histidine, phosphate or citrate.
- the buffer is succinate at a final concentration of 1 mM to 10 mM and pH 5.8 to 6.0.
- the salt in the pH buffered saline solution comprises magnesium chloride, potassium chloride, sodium chloride or a combination thereof.
- the formulation further comprises polysorbate
- the final concentration of the polysorbate 80 in the formulation is 0.01 % to 10% polysorbate 80 weight/volume of the formulation.
- the N. meningitidis 2086 protein composition further comprises one or more polypeptides selected from the group consisting of a streptococcal polypeptide, a pneumococcal polypeptide, a meningococcal polypeptide and a staphylococcal polypeptide.
- the N. meningitidis 2086 protein composition further comprises one or more polysaccharides selected from the group consisting of a streptococcal polysaccharide, a pneumococcal polysaccharide, a meningococcal polysaccharide and a staphylococcal polysaccharide.
- the formulation further comprises one or more adjuvants. Exemplary suitable adjuvants are described herein below.
- FIG. 1 shows the stability of Streptococcal C5a peptidase (SCP) formulations (filled in syringes) before and after two days of gentle agitation (60 cpm) on a horizontal orbital shaker.
- SCP Streptococcal C5a peptidase
- FIG. 1A The data presented in FIG. 1A is the two day stability of the SCP formulated without any TweenTM80 (Ae., 0%), whereas the data in FIG. 1 B is the two day stability of the SCP formulated with 0.025% TweenTM80.
- the buffers used in the formulations shown in FIG. 1A and 1 B are succinate buffered saline (SBS), phosphate buffered saline (PBS) and tris(hydroxymethyl)aminomethane (TRIS).
- Figure 2 shows the total antigenicity loss of the 13vPnC formulated with AIPO 4 (0.25 mg/ml) and filled in a BD Hypak syringe, after two hours, eight hours and twenty-four hours of agitation at 500 rpm and 2-8°C.
- Figure 3 shows the total antigenicity loss of the 13vPnC formulated with AIPO 4 (0.25 mg/ml) and filled in an un-siliconized syringe, after two hours, eight hours and twenty-four hours of agitation at 500 rpm and 2-8°C.
- Figure 4 shows the total antigenicity loss of the 13vPnC formulated with
- AIPO 4 (0.25 mg/ml) and filled in a Vetter syringe, after two hours, eight hours and twenty-four hours of agitation at 500 rpm and 2-8°C.
- Figure 5 shows the total antigenicity loss of the 13vPnC formulated with AIPO 4 (0.25 mg/ml) and filled in a Schott TopPac syringe, after two hours, eight hours and twenty-four hours of agitation at 500 rpm and 2-8°C.
- Figure 6 shows the total antigenicity loss of the 13vPnC formulated with (FIG. 6A) and without (FIG. 6B) AIPO 4 (0.25 mg/ml) and filled in a BD Baked syringe, after two hours, eight hours and twenty-four hours of agitation at 500 rpm and 2-8°C.
- Figure 7 shows the total antigenicity loss of the 13vPnC formulated with (FIG. 7A) and without (FIG. 7B) AIPO 4 (0.25 mg/ml) and filled in a B ⁇ nderGlas PS2 syringe, after two hours, eight hours and twenty-four hours of agitation at 500 rpm and 2-8°C.
- the present invention addresses an ongoing need in the art to improve the stability of immunogenic compositions such as polysaccharide-protein conjugates and protein immunogens.
- the present invention broadly relates to novel surfactant formulations and/or novel aluminum salt formulations which stabilize and inhibit precipitation of immunogenic compositions.
- the invention described hereinafter addresses a need in the art for formulations which stabilize and inhibit particulate formation (e.g., aggregation, precipitation) of immunogenic compositions which are processed, developed, formulated, manufactured and/or stored in container means such as fermentors, bioreactors, vials, flasks, bags, syringes, rubber stoppers, tubing and the like.
- immunogenic compositions including, but not limited to, chemical stability of the immunogenic composition, physical/thermal stability of the immunogenic composition, compatibility of the immunogenic composition with the container/closure system, interactions between immunogenic composition and inactive ingredients (e.g., buffers, salts, excipients, cryoprotectants), manufacturing processes, dosage form, environmental conditions encountered during shipping, storage and handling (e.g., temperature, humidity, shear forces), and the length of time between manufacture and usage.
- inactive ingredients e.g., buffers, salts, excipients, cryoprotectants
- manufacturing processes e.g., dosage form, environmental conditions encountered during shipping, storage and handling (e.g., temperature, humidity, shear forces), and the length of time between manufacture and usage.
- an immunogenic composition of the invention is readily determined using standard techniques, which are well known and routine to those of skill in the art.
- an immunogenic composition is assayed for stability, aggregation, immunogenicity, particulate formation, protein (concentration) loss, and the like, by methods including, but not limited to, light scattering, optical density, sedimentation velocity centrifugation, sedimentation equilibrium centrifugation, circular dichroism (CD), Lowry assay, bicinchoninic acid (BCA) assay, antibody binding, and the like.
- the present invention relates to the unexpected and surprising results that formulating an immunogenic composition with a surfactant such as TweenTM80 significantly enhances the stability and inhibits precipitation of an immunogenic composition. For example, it was observed in the present invention
- a second stability study of the 13vPnC further confirmed that the addition of a surfactant to the formulation significantly enhanced the stability of the 13vPnC.
- the stability i.e., assayed by measuring change in 13vPnC antigenicity
- TweenTM80 Table 1
- TweenTM80 0.0%, Table 1
- Table 1 there was a significant decrease in antigenicity of the thirteen serotype polysaccharides (formulated without TweenTM80) within the two hour assay .
- TweenTM80 or 0.05% TweenTM80 could withstand significant shear forces induced via vortexing the formulations for thirty minutes at 2-8°C, with little or no loss in antigenicity (e.g., see Example 2, Table 2).
- an immunogenic streptococcal C5a peptidase (SCP) composition was greatly enhanced when formulated with a surfactant such as TweenTM80.
- a surfactant such as TweenTM80.
- FIG. 1 A after two days of vortexing an SCP (55 ⁇ g/mL) formulated in either a 5 mM succinate buffer (pH 6.0), a 10 mM phosphate buffer (pH 7.0 and 7.4) or a 10 mM Tris buffer (pH 7.5), there was a significant decrease (e.g., greater than 90%) in the SCP concentration.
- FIG. 1 B the addition of 0.025%
- a 13vPnC immunogenic composition of the invention may also be formulated with or without an adjuvant, such as aluminum phosphate (AIPO 4 ).
- an adjuvant such as aluminum phosphate (AIPO 4 ).
- 13vPnC immunogenic compositions were formulated in 5 mM succinate buffer (pH 5.8), 0.85% NaCI and AIPO 4 (0.25 mg aluminum/ml), without the addition of a surfactant (e.g., no TweenTM80 was included in the formulation).
- the 13vPnC immunogenic composition (formulated in the presence of AIPO 4 ) were filled in various siliconized and non-siliconized container means (e.g., see Table 3) and subjected to simulated shipping and handling conditions via agitation at 2-8°C. It was observed in these experiments (Example 4), that the container means with higher silicone content exhibited a higher degree of 13vPnC particulate formation and a higher percent of 13vPnC antigenicity loss.
- the invention as set forth herein, is directed to novel formulations which stabilize and inhibit aggregation or precipitation of immunogenic compositions such as polysaccharide-protein conjugates (e.g., a 13vPnC) and protein immunogens
- immunogenic compositions such as polysaccharide-protein conjugates (e.g., a 13vPnC) and protein immunogens
- immunogenic compositions e.g., shear forces, shipping agitation, silicone oil interactions, adsorption, manufacturing processes, temperature, humidity, length of time between manufacture and usage, efc.
- the invention is directed to a formulation which stabilizes a polysaccharide-protein conjugate, the formulation comprising a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 7.5, a surfactant and one or more polysaccharide-protein conjugates.
- the polysaccharide-protein conjugate formulation is comprised in a container means.
- the invention is directed to a formulation which stabilizes a streptococcal C5a peptidase (SCP) composition, the formulation comprising a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 6.5, a surfactant and a streptococcal C5a peptidase.
- SCP streptococcal C5a peptidase
- the SCP formulation is comprised in a container means.
- the invention is directed to a formulation which stabilizes a N. meningitidis 2086 protein composition, the formulation comprising a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 7.5, a surfactant and a N.
- meningitidis 2086 protein is comprised in a container means.
- the invention is directed to a formulation which inhibits silicone induced precipitation of a polysaccharide-protein conjugate comprised in a siliconized container means, the formulation comprising a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 7.5, an aluminum salt and one or more polysaccharide-protein conjugates.
- the invention is directed to a formulation which inhibits silicone induced precipitation of a streptococcal C5a peptidase (SCP) composition comprised in a siliconized container means, the formulation comprising a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 6.5, an aluminum salt and a streptococcal C5a peptidase.
- SCP streptococcal C5a peptidase
- the invention is directed to a formulation which inhibits silicone induced precipitation of a N.
- meningitidis 2086 protein composition comprised in a siliconized container means, the formulation comprising a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 7.5, an aluminum salt and a N. meningitidis 2086 protein.
- the invention is directed to a formulation that optimizes antigen stability and binding percentage to an aluminum salt adjuvant (e.g.,
- a N. meningitidis 2086 protein comprising a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 7.5, a surfactant, an aluminum salt, and a N. meningitidis 2086 protein.
- the formulation is in a container means.
- precipitation As defined hereinafter, the terms "precipitation”, “precipitate” “particulate formation”, “clouding” and “aggregation” may be used interchangeably and are meant to refer to any physical interaction or chemical reaction which results in the "aggregation" of a polysaccharide-protein conjugate or a protein (or polypeptide) immunogen.
- aggregation e.g., protein aggregation
- process of aggregation is well known (but not well understood) and described in the art, and is often influenced by numerous physicochemical stresses, including heat, pressure, pH, agitation, shear forces, freeze-thawing, dehydration, heavy metals, phenolic compounds, silicon oil, denaturants and the like.
- a "polysaccharide-protein conjugate”, a “pneumococcal conjugate”, a “7-valent pneumococcal conjugate (7vPnC)", a “13- valent pneumococcal conjugate (13vPnC)”, a “streptococcal C5a peptidase (SCP) immunogenic composition” and a " ⁇ /. meningitidis 2086 protein immunogenic composition” of the invention includes liquid formulations, frozen liquid formulations and solid (e.g., freeze-dried or lyophilized) formulations.
- the invention is directed to formulations which stabilize and inhibit aggregation of immunogenic compositions against the various factors which influence the stability of immunogenic compositions (e.g., shear forces, shipping agitation, silicone oil interactions, adsorption, manufacturing processes, temperature, humidity, length of time between manufacture and usage, efc.).
- the invention is directed to formulations comprising a surfactant.
- a surfactant (or a surface-active agent) is generally defined as (a) a molecule or compound comprising a hydrophilic group or moiety and a lipophilic (hydrophobic) group or moiety and/or (b) a molecule, substance or compound that lowers or reduces surface tension of a solution.
- a "surfactant" of the present invention is any molecule or compound that lowers the surface tension of an immunogenic composition formulation.
- a surfactant used in a formulation of the present invention comprises any surfactant or any combination of surfactants which stabilizes and inhibits aggregation of an immunogenic composition described herein.
- a surfactant of the invention includes, but is not limited to, polysorbate 20 (TweenTM20), polysorbate 40 (TweenTM40), polysorbate 60 (TweenTM60), polysorbate 65 (TweenTM65), polysorbate 80 (TweenTM80), polysorbate 85 (TweenTM85), TritonTM N-101 , TritonTM X-100, oxtoxynol 40, nonoxynol-9, triethanolamine, triethanolamine polypeptide oleate, polyoxyethylene-660 hydroxystearate (PEG-15, Solutol H 15), polyoxyethylene-35- ricinoleate (Cremophor ELTM), soy lecithin, poloxamer, hexadecylamine, octadecylamine
- a person of skill in the art may readily determine a suitable surfactant or surfactant combination by measuring the surface tension of a particular immunogenic composition formulation in the presence and absence of the surfactant(s).
- a surfactant is evaluated qualitatively (e.g., visual inspection of particulate formation) or quantitatively (e.g., light scattering, sedimentation velocity centrif ligation, optical density, antigenicity) for its ability to reduce, inhibit or prevent aggregation of an immunogenic composition.
- the invention is directed to formulations of immunogenic compositions comprised in a container means.
- a "container means" of the present invention includes any composition of matter which is used to "contain”, “hold”, “mix”, “blend”, “dispense”, “inject”, “transfer”, “nebulize”, etc. an immunogenic composition during research, processing, development, formulation, manufacture, storage and/or administration.
- a container means of the present invention includes, but is not limited to, general laboratory glassware, flasks, beakers, graduated cylinders, fermentors, bioreactors, tubings, pipes, bags, jars, vials, vial closures (e.g., a rubber stopper, a screw on cap), ampoules, syringes, syringe stoppers, syringe plungers, rubber closures, plastic closures, glass closures, and the like.
- general laboratory glassware flasks, beakers, graduated cylinders, fermentors, bioreactors, tubings, pipes, bags, jars, vials, vial closures (e.g., a rubber stopper, a screw on cap), ampoules, syringes, syringe stoppers, syringe plungers, rubber closures, plastic closures, glass closures, and the like.
- a container means of the present invention is not limited by material of manufacture, and includes materials such as glass, metals (e.g., steel, stainless steel, aluminum, efc.) and polymers (e.g., thermoplastics, elastomers, thermoplastic-elastomers).
- materials such as glass, metals (e.g., steel, stainless steel, aluminum, efc.) and polymers (e.g., thermoplastics, elastomers, thermoplastic-elastomers).
- container means set forth above are by no means an exhaustive list, but merely serve as guidance to the artisan with respect to the variety of container means which are used to contain, hold, mix, blend, dispense, inject, transfer, nebulize, efc. an immunogen or immunogenic composition during research, processing, development, formulation, manufacture, storage and/or administration of the composition.
- Additional container means contemplated for use in the present invention may be found in published catalogues from laboratory equipment vendors and manufacturers such as United States Plastic Corp. (Lima, OH), VWRTM (West Chester, PA), BD Biosciences (Franklin Lakes, NJ), Fisher Scientific International Inc. (Hampton, NH) and Sigma-Aldrich (St. Louis, MO).
- novel formulations of the present invention are particularly advantageous in that they stabilize and inhibit precipitation of immunogenic formulations comprised in a container means throughout the various stages of research, processing, development, formulation, manufacture, storage and/or administration of the composition.
- the novel formulations of the invention not only stabilize immunogenic compositions against physical/thermal stresses (e.g., temperature, humidity, shear forces, efc.), they also enhance stability and inhibit precipitation of immunogenic compositions against negative factors or influences such as incompatibility of the immunogenic composition with the container/closure system (e.g., a siliconized container means).
- novel formulations of the present invention are particularly useful in stabilizing the immunogen (i.e., a polysaccharide-protein conjugate, a protein or polypeptide antigen) against the silicon oil induced precipitation and precipitation described above.
- immunogen i.e., a polysaccharide-protein conjugate, a protein or polypeptide antigen
- the immunogenic compositions of the invention are further formulated with an adjuvant.
- An adjuvant is a substance that enhances the immune response when administered together with an immunogen or antigen.
- a number of cytokines or lymphokines have been shown to have immune modulating activity, and thus may be used as adjuvants, including, but not limited to, the interleukins 1- ⁇ , 1- ⁇ , 2, 4, 5, 6, 7, 8, 10, 12 (see, e.g., U.S. Patent No. 5,723,127), 13, 14, 15, 16, 17 and 18 (and its mutant forms), the interferons- ⁇ , ⁇ and Y, granulocyte-macrophage colony stimulating factor (GMCSF, see, e.g., U.S. Patent No.
- GMCSF granulocyte-macrophage colony stimulating factor
- chemokines including without limitation, MCP-1 , MIP-1 ⁇ , MIP-1 ⁇ , and RANTES.
- an adjuvant used to enhance an immune response of an immunogenic composition formulation includes, without limitation, MPLTM (3-O- deacylated monophosphoryl lipid A; Corixa, Hamilton, MT), which is described in U.S. Patent No. 4,912,094, which is hereby incorporated by reference.
- MPLTM 3-O- deacylated monophosphoryl lipid A
- Corixa Hamilton, MT
- AGP synthetic lipid A analogs or aminoalkyl glucosamine phosphate compounds
- AGP aminoalkyl glucosamine phosphate compounds
- AGP is 2-[(R)-3- Tetradecanoyloxytetradecanoylamino] ethyl 2-Deoxy-4-O-phosphono-3-O-[(R)-3- tetradecanoyoxytetradecanoyl]-2-[(R)-3-tetradecanoyloxytetradecanoyl-amino]-b-D- glucopyranoside, which is also known as 529 (formerly known as RC529).
- This 529 adjuvant is formulated as an aqueous form or as a stable emulsion (RC529-SE).
- Still other adjuvants include mineral oil and water emulsions, aluminum salts (alum), such as aluminum hydroxide, aluminum phosphate, aluminum sulfate efc., Amphigen, Avridine, L121/squalene, D-lactide-polylactide/glycoside, pluronic polyols, muramyl dipeptide, killed Bordetella, saponins, such as StimulonTM QS-21 (Antigenics, Framingham, MA.), described in U.S. Patent No.
- coli heat-labile toxin particularly LT-K63, LT-R72, PT-K9/G129; see, e.g., International Patent Publication Nos. WO 93/13302 and WO 92/19265, incorporated herein by reference.
- adjuvants and carrier proteins
- cholera toxins and mutants thereof including those described in published International Patent Application number WO 00/18434 (wherein the glutamic acid at amino acid position 29 is replaced by another amino acid (other than aspartic acid), preferably a histidine).
- CT toxins or mutants are described in published International Patent Application number WO 02/098368 (wherein the isoleucine at amino acid position 16 is replaced by another amino acid, either alone or in combination with the replacement of the serine at amino acid position 68 by another amino acid; and/or wherein the valine at amino acid position 72 is replaced by another amino acid).
- Other CT toxins are described in published International Patent Application number WO 02/098369 (wherein the arginine at amino acid position 25 is replaced by another amino acid; and/or an amino acid is inserted at amino acid position 49; and/or two amino acids are inserted at amino acid positions 35 and 36).
- the immunogenic composition formulations comprise a pharmaceutically acceptable diluent, excipient or a pharmaceutically acceptable carrier.
- the pharmaceutically acceptable diluent is sterile water, water for injection, sterile isotonic saline or a biological buffer.
- the polysaccharide- protein conjugates and/or protein immunogens are mixed with such diluents or carriers in a conventional manner.
- pharmaceutically acceptable carrier is intended to include any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like, compatible with administration to humans or other vertebrate hosts.
- the appropriate carrier is evident to those skilled in the art and will depend in large part upon the route of administration.
- excipients that may be present in the immunogenic composition formulation are preservatives, chemical stabilizers and suspending or dispersing agents.
- stabilizers, preservatives and the like are optimized to determine the best formulation for efficacy in the targeted recipient (e.g., a human subject).
- preservatives include chlorobutanol, potassium sorbate, sorbic acid, sulfur dioxide, propyl gallate, the parabens, ethyl vanillin, glycerin, phenol, and parachlorophenol.
- stabilizing ingredients include casamino acids, sucrose, gelatin, phenol red, N-Z amine, monopotassium diphosphate, lactose, lactalbumin hydrolysate, and dried milk.
- an immunogenic composition formulation is prepared for administration to human subjects in the form of, for example, liquids, powders, aerosols, tablets, capsules, enteric-coated tablets or capsules, or suppositories.
- the immunogenic composition formulations may also include, but are not limited to, suspensions, solutions, emulsions in oily or aqueous vehicles, pastes, and implantable sustained-release or biodegradable formulations.
- the immunogenic compositions of the present invention are not limited by the selection of the conventional, physiologically acceptable carriers, diluents and excipients such as solvents, buffers, adjuvants, or other ingredients useful in pharmaceutical preparations of the types described above.
- the preparation of these pharmaceutically acceptable compositions, from the above-described components, having appropriate pH isotonicity, stability and other conventional characteristics is within the skill of the art.
- a polysaccharide-protein conjugate formulation of the invention comprises one or more pneumococcal polysaccharides. In other embodiments, a polysaccharide-protein conjugate formulation of the invention comprises one or more streptococcal polysaccharides. In yet other embodiments, a polysaccharide-protein conjugate formulation of the invention comprises one or more meningococcal polysaccharides.
- a polysaccharide- protein conjugate formulation of the invention comprises a combination of one or more pneumococcal polysaccharides, one or more pneumococcal polypeptides, one or more streptococcal polysaccharides, one or more streptococcal polypeptides, one or more meningococcal polysaccharides, and/or one or more meningococcal polypeptides.
- the term "polysaccharide” is meant to include any antigenic saccharide element (or antigenic unit) commonly used in the immunologic and bacterial vaccine arts, including, but not limited to, a "saccharide", an
- oligosaccharide a "polysaccharide”, a “liposaccharide”, a “lipo-oligosaccharide (LOS)", a “lipopolysaccharide (LPS)", a “glycosylate”, a “glycoconjugate” and the like.
- the one or more pneumococcal polysaccharides are a S. pneumoniae serotype 4 polysaccharide, a S. pneumoniae serotype 6B polysaccharide, a S. pneumoniae serotype 9V polysaccharide, a S. pneumoniae serotype 14 polysaccharide, a S. pneumoniae serotype 18C polysaccharide, a S. pneumoniae serotype 19F polysaccharide, a S. pneumoniae serotype 23F polysaccharide, a S. pneumoniae serotype 1 polysaccharide, a S. pneumoniae serotype 3 polysaccharide, a S.
- a polysaccharide-protein conjugate formulation is a
- 7-valent pneumococcal conjugate (7vPnC) formulation comprising a S. pneumoniae serotype 4 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 6B polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 9V polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 14 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 18C polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 19F polysaccharide conjugated to a CRM 197 polypeptide and a S. pneumoniae serotype 23F polysaccharide conjugated to a CRM 197 polypeptide.
- a polysaccharide-protein conjugate formulation is a 13-valent pneumococcal conjugate (13vPnC) formulation comprising a S. pneumoniae serotype 4 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 6B polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 9V polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 14 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 18C polysaccharide conjugated to a CRM 197 polypeptide, a S.
- 13vPnC 13-valent pneumococcal conjugate
- pneumoniae serotype 19F polysaccharide conjugated to a CRM 197 polypeptide a S. pneumoniae serotype 23F polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 1 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 3 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 5 polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 6A polysaccharide conjugated to a CRM 197 polypeptide, a S. pneumoniae serotype 7F polysaccharide conjugated to a CRM 197 polypeptide and a S. pneumoniae serotype 19A polysaccharide conjugated to a CRM 197 polypeptide.
- Polysaccharides are prepared by standard techniques known to those skilled in the art.
- the capsular polysaccharides set forth in the present invention are prepared from serotypes 1 , 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F and 23F of Streptococcus pneumoniae, wherein each serotype is grown in a soy- based medium and the individual polysaccharides are then purified through centrifugation, precipitation, ultra-filtration, and column chromatography.
- streptococcal polysaccharides e.g., one or more polysaccharides (or oligosaccharides) from a ⁇ -hemolytic Streptococcus such group A Streptococcus, group B Streptococcus, group C Streptococcus and group G Streptococcus
- meningococcal saccharides e.g., an N. meningitidis lipo-oligosaccharide (LOS) or lipo-polysaccharide (LPS)
- LOS meningitidis lipo-oligosaccharide
- LPS lipo-polysaccharide
- each capsular polysaccharide is separately conjugated to a carrier protein (e.g., CRM 197 ) to form a glycoconjugate (or alternatively, each capsular polysaccharide is conjugated to the same carrier protein) and formulated into a single dosage formulation.
- a carrier protein e.g., CRM 197
- Carrier proteins are preferably proteins that are non-toxic and non- reactogenic and obtainable in sufficient amount and purity. Carrier proteins should be amenable to standard conjugation procedures.
- CRM 197 is used as the carrier protein.
- CRM 197 (Wyeth, Sanford, NC) is a non-toxic variant (Ae., toxoid) of diphtheria toxin isolated from cultures of Corynebacterium diphtheria strain C7 ( ⁇ 197) grown in casamino acids and yeast extract-based medium.
- CRM 197 is purified through ultrafiltration, ammonium sulfate precipitation, and ion-exchange chromatography.
- CRM 197 is prepared recombinantly in accordance with U.S. Patent No. 5,614,382, which is hereby incorporated by reference.
- Other diphtheria toxoids are also suitable for use as carrier proteins.
- a carrier protein of the invention is an enzymatically inactive streptococcal C5a peptidase (SCP) (e.g., one or more of the SCP variants described in U.S. Patent 6,951 ,653, U.S. Patent 6,355,255 and U.S. Patent 6,270,775).
- SCP streptococcal C5a peptidase
- carrier proteins include inactivated bacterial toxins such as tetanus toxoid, pertussis toxoid, cholera toxoid (e.g., CT E29H, described in International Patent Application WO2004/083251 ), E. coli LT, E. coli ST, and exotoxin A from Pseudomonas aeruginosa.
- Bacterial outer membrane proteins such as outer membrane complex c (OMPC), porins, transferrin binding proteins, pneumolysin, pneumococcal surface protein A (PspA), pneumococcal adhesin protein (PsaA), or Haemophilus influenzae protein D, can also be used.
- Other proteins such as ovalbumin, keyhole limpet hemocyanin (KLH), bovine serum albumin (BSA) or purified protein derivative of tuberculin (PPD) can also be used as carrier proteins.
- the polysaccharide-protein conjugates are purified (enriched with respect to the amount of polysaccharide-protein conjugate) by a variety of techniques. These techniques include concentration/diafiltration operations, precipitation/elution, column chromatography, and depth filtration.
- the individual glycoconjugates are compounded to formulate the immunogenic composition of the present invention.
- Formulation of the polysaccharide-protein conjugates of the present invention can be accomplished using art-recognized methods.
- the 13 individual pneumococcal conjugates can be formulated with a physiologically acceptable vehicle to prepare the composition.
- physiologically acceptable vehicles include, but are not limited to, water, buffered saline, polyols (e.g., glycerol, propylene glycol, liquid polyethylene glycol) and dextrose solutions.
- the invention is directed to formulations which stabilize a streptococcal C5a peptidase (SCP) immunogenic composition, wherein the formulations comprise a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 6.5, a surfactant and a streptococcal C5a peptidase.
- SCP streptococcal C5a peptidase
- the C5a peptidase is a highly conserved serine protease and is expressed across all ⁇ - hemolytic Streptococci (e.g., streptococcal Groups A, B, C and G).
- an immunogenic composition against infection caused by ⁇ -hemolytic Streptococci comprises a C5a peptidase immunogen (or antigen).
- a C5a peptidase of the invention is an enzymatically inactive streptococcal C5a peptidase (e.g., one or more of the SCP variants described in U.S. Patent 6,951 ,653, U.S.
- the SCP used in the novel immunogenic composition formulations of the invention is cloned from a Group B streptococci.
- the Group B streptococci SCP sequence has been genetically mutated to render it proteolytically inactive (e.g., see U.S. Patents 6,951 ,653; 6,355,255 and 6,270,775) and is expressed as a recombinant protein in E. coli.
- the invention is directed to formulations which stabilize a N.
- meningitidis 2086 protein immunogenic composition wherein the formulations comprise a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 7.5, a surfactant and a N. meningitidis 2086 protein.
- the N. meningitidis 2086 proteins are encoded by a nucleic acid sequence open reading frame (ORF) identified as "ORF 2086" (e.g., see International Publication No. WO
- the invention is directed to formulations that optimize antigen stability and binding percentage to an aluminum salt adjuvant (e.g., AIPO 4 ) of a N.
- an aluminum salt adjuvant e.g., AIPO 4
- meningitidis 2086 protein wherein the formulations comprise a pH buffered saline solution, wherein the buffer has a pKa of about 3.5 to about 7.5, a surfactant, an aluminum salt, and a N. meningitidis 2086 protein.
- All patents and publications cited herein are hereby incorporated by reference.
- IMMUNOGENIC FORMULATIONS COMPRISING 0.001 %-0.05% TWEENTM80 STABILIZE AND PREVENT AGGREGATION OF THE IMMUNOGEN
- the polysaccharide-protein conjugate used in this example was a thirteen- valent pneumococcal polysaccharide conjugate (13vPnC) comprising capsular polysaccharides from S. pneumoniae serotypes 4, 6B, 9V, 18C, 19F, 14, 23F, 1 , 3, 5, 6A, 7F and 19A, each of which were conjugated to CRM 197 .
- the capsular polysaccharides were prepared by standard techniques known to those skilled in the art. Briefly, each pneumococcal polysaccharide serotype was grown in a soy-based medium, the individual polysaccharides were then purified through centrifugation, precipitation, ultra-filtration, and column chromatography.
- CRM 197 (Wyeth, Sanford, NC) is a non-toxic variant (Ae., toxoid) of diphtheria toxin isolated from cultures of Corynebacterium diphtheria strain C7 ( ⁇ 197) grown in casamino acids and yeast extract-based medium.
- CRM 197 is purified through ultra-filtration, ammonium sulfate precipitation, and ion-exchange chromatography.
- the antigenicity experiments described below were performed by mixing the 13vPnC samples with one of thirteen antisera (Ab) specific to the each of the polysaccharide serotypes and detecting the immune complexes via light scattering measurements on an Array® 360 system (Beckman Coulter, Inc.; Fullerton, CA).
- the detected light scattering measurements for each of the thirteen serotypes were then compared to a standard curve and reported as antigenicity ( ⁇ g/mL).
- Polysorbate 80 (TweenTM80) was purchased from JT. Baker (Mallinckrodt Baker, Inc.; Phillipsburg, NJ). Buffered saline was succinate (5 mM) and NaCI (0.85%) at pH 5.8.
- the 13vPnC was formulated (500 ml. total volume) at different surfactant concentrations (TweenTM80; 0.001 %, 0.005%, 0.01 % and 0.05%, weight/volume) as follows: 0.85% saline (150 mM NaCI) was added to a one liter Pyrex® glass beaker, followed by 50 mM succinate buffer (final concentration 5 mM) and the 13vPnC. The final concentration of each serotype conjugate was 4.4 ⁇ g/mL (except for serotype 6B, which was 8.8 ⁇ g/mL).
- the 13vPnC formulation was then divided into five separate glass vials (50 mL per vial), wherein either 0.0%, 0.001 %, 0.005%, 0.01 % or 0.05% TweenTM80 (w/v) was added to one of the five vials and each solution filtered through a 0.22 ⁇ m Durapore® filter (Millipore; Billerica, MA).
- 13vPnC was formulated with and without 0.05% TweenTM80.
- the 13vPnC formulated without TweenTM80 ⁇ i.e., 0.0%) was prepared as follows: 0.85% saline (150 mM NaCI) was added to a one liter Pyrex® glass beaker, followed by 50 mM succinate buffer (final concentration 5 mM) and the 13vPnC, at a total volume of 500 mL.
- TweenTM80 was prepared as follows: 0.85% saline (150 mM NaCI) was added to a one liter Pyrex® glass beaker, followed by 50 mM succinate buffer (final concentration 5 mM), 0.05% TweenTM80 and the 13vPnC, at a total volume of 500 mL. The final concentration of each serotype conjugate in the 500 mL formulations was 4.4 ⁇ g/mL (except for serotype 6B, which was 8.8 ⁇ g/mL). The 500 mL formulations were homogenized via a rotor/stator homogenizer at 6,000 rpm (2-8°C) for 120 minutes. The homogenization process created an air-liquid interface (with air bubbles).
- the 13vPnC/TweenTM80 formulation was further tested for stability against high shear forces.
- a 100 mL 13vPnC composition (4.4 ⁇ g/mL serotypes 1 , 3, 4, 5, 6A, 7F, 9V, 14, 18C, 19A, 19F, 23F and 8.8 ⁇ g/mL serotype 6B, 5 mM succinate buffer, 150 mM NaCI and 0.25 mg/mL AIPO 4 ) was added to three 250 mL glass bottles comprising either 0.0%, 0.01 % or 0.05% TweenTM80. The three bottles were then vortexed for thirty minutes (2-8°C) on a vortexer (Vortex-Genie® 2;
- the 13vPnC formulated with 0.01 % TweenTM80 had a decrease in antigenicity ranging from 2-10% (average 8%) and the 13vPnC formulated with 0.05% TweenTM80 had a decrease in antigenicity ranging from 0-8% (average 3%).
- Table 2 demonstrate that the 13vPnC formulated with either 0.01 % or 0.05% TweenTM80 were significantly stabilized against shear forces, relative to the 13vPnC formulated in the absence of TweenTM80.
- streptococcal C5a peptidase (SCP) used in this example was expressed and purified as follows.
- the SCP was expressed recombinantly in E. coli using an arabinose inducible system. Standard fermentation protocols for E. coli using animal-free defined medium and subsequent cell lysis were followed. Recombinant SCP was purified from the soluble fraction of the cell lysate by saturation to 60%
- the purified SCP (55 ⁇ g/mL) was then formulated with 0.025% TweenTM80 or without TweenTM80 (0.0%) in the following buffers: 5 mM succinate buffer at pH 6.0, 10 mM phosphate buffer at pH 7.0, 10 mM phosphate buffer at 7.4 or 10 mM Tris buffer at pH 7.5 and filled in separate BD Hypak SCFTM syringes.
- the syringes were then placed on an a horizontal orbital shaker at 2-8 0 C, shaken at 180 cpm for two days and the SCP protein concentration determined by the modified Lowry assay.
- the stability of SCP was greatly enhanced when formulated with TweenTM80.
- TweenTM80 For example, after two days on the orbital shaker, the
- the storage stability of the SCP/TweenTM80 (0.025%) formulation was also assessed at 25°C and 37°C for eight weeks and six weeks, respectively (data not shown).
- the SCP 200 ⁇ g was formulated in either succinate buffer or phosphate buffer as follows: succinate buffer (5 mM, pH 6.0) or phosphate buffer (15 mM, pH 7.4), 0.9% NaCI and 0.025% TweenTM80.
- succinate buffer 5 mM, pH 6.0
- phosphate buffer 15 mM, pH 7.4
- SCP/TweenTM80 formulations were assayed by size-exclusion-HPLC, modified Lowry total protein assay and visual inspection for precipitation. It was observed in this study, that the SCP/TweenTM80 formulations (in either buffer) were completely stable at 25°C and 37°C for the entire stability study (Ae., up to eight weeks and six weeks, respectively).
- 13vPnC particulates were readily observable, whereas, in the presence Of AIPO 4 , the 13vPnC particulates were significantly diminished and more difficult to detect.
- container means i.e., container means
- the container means tested comprised syringes, stoppers and vials and are listed below in Table 3.
- the BD and West stoppers listed in Table 3 were siliconized, using either the Huber or Jar process.
- the Huber process of siliconization is more controlled and yielded 30 to 60 ⁇ g/cm2 of silicone on the surface of the stopper, while the Jar process of siliconization resulted in 150 to 300 ⁇ g/cm2 of silicone on the surface of the stopper. Based on theoretical calculations, about 15% of the surface area of the stopper is exposed to the product in the syringe, suggesting that for the Huber and Jar process between 4.5 to 9 ⁇ g and 22.5 to 45 ⁇ g of silicone is extractable from the stoppers, respectively.
- the silicone was Dow Corning® 360 Medical Fluid 1000 centistokes (batch No. 0001846266).
- the 7vPnC was formulated in 5 mM succinate buffer containing 0.85% NaCI and 4.4 ⁇ g/ml of S. pneumoniae serotypes 4, 9, 14, 18C, 19F and 23F and 8.8 ⁇ g/ml of S. pneumoniae serotype 6B, with and without 0.25 mg/ml aluminum phosphate.
- the 13vPnC was formulated in 5 mM succinate buffer containing 0.85% NaCI and 4.4 ⁇ g/ml of S. pneumoniae serotypes 1 , 3, 4, 5, 6A, 7F, 9V, 14, 18C, 19A, 19F and 23F and 8.8 ⁇ g/ml of S.
- Monovalent S. pneumoniae serotype 6B was formulated (5 mM succinate buffer containing 0.85% NaCI, without aluminum phosphate) at a concentration of 61 ⁇ g/ml to simulate the total saccharide concentration of the 13vPnC formulations.
- Methods The 7vPnC and 13vPnC were formulated as described above, and 35 ml of a given formulation was added to a clear 250 ml Nalgene® bottle. Into each Nalgene® bottle, the container means components listed in Table 3 were added. The Nalgene® bottles were then placed on a Labline® Orbit Shaker and swirled overnight at 50 rpm.
- the total protein and protein bound to aluminum was determined by measuring the total protein concentration in the formulated immunogenic composition and the protein associated with the aluminum pellet, respectively (an aliquot of the immunogenic composition was centrifuged and the pellet was re-suspended in saline). Assays were performed using the Pierce Modified Lowry protein assay (catalog # 23240) with bovine serum albumin as a standard. RESULTS
- the monovalent S. pneumoniae serotype 6B was chosen as a model for the 13vPnC and was formulated at 61.6 ⁇ g/ml (without AIPO 4 ) to simulate the total saccharide concentration in the 13vPnC formulation.
- Silicone (Dow Corning 360 Medical Fluid) was added to aliquots of the formulated monovalent 6B, ranging from 2 ppm to 100 ppm. The mixtures were placed on a Labline® Orbit Shaker for 2 hours at 50 rpm. As indicated below in Table 4, fiber-like white particulates were observed at all silicone (Si) concentrations.
- the amount of silicone in 13vPnC formulations was also examined.
- the silicone concentration was determined by DC Plasma Emission Spectroscopy (data not shown). In this method, the content of 25 syringes were pooled and extracted with two 50 ml portions of cyclohexane/isopropyl alcohol mixture. The extracts were combined and evaporated. The residual was solubilized and tested as per existing methods for silicone determination on rubber stoppers.
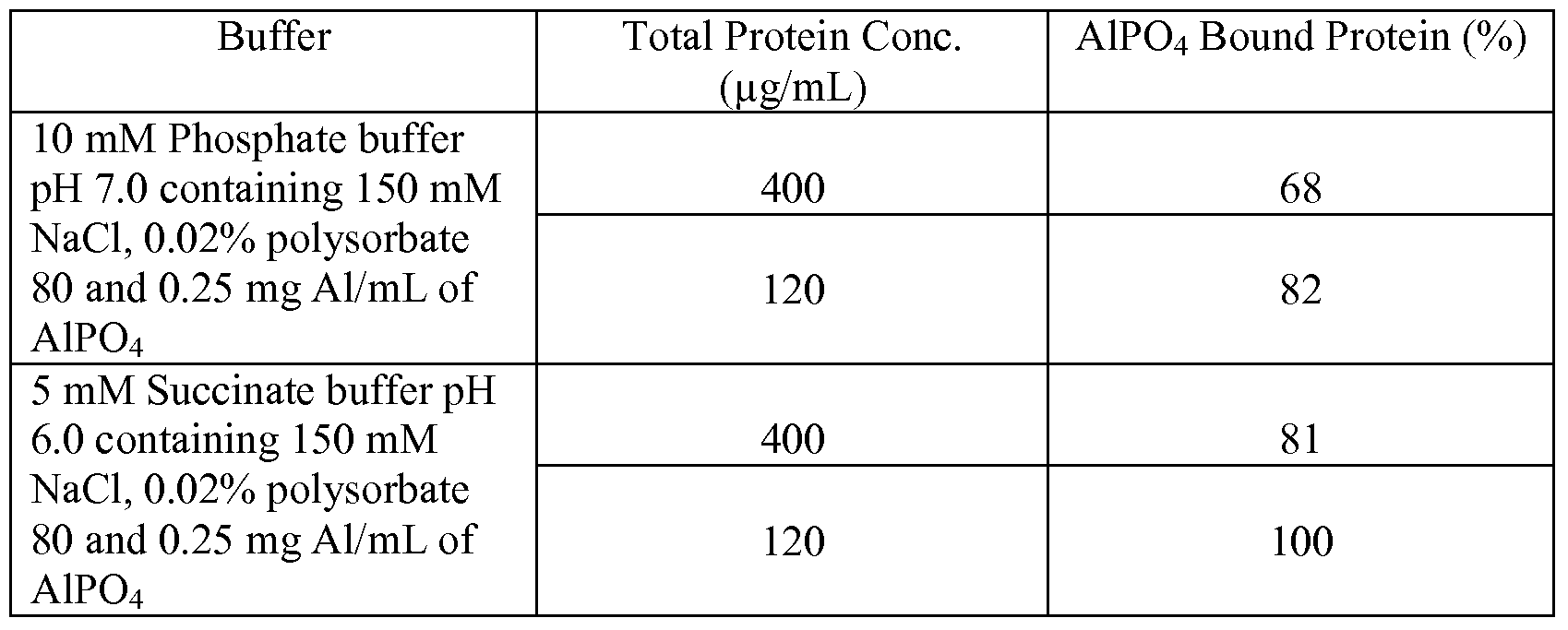
- AIPO 4 was 100% bound to the AIPO 4 (data not shown).
- a 13vPnC immunogenic composition is a liquid formulation comprising 4.4 ⁇ g/mL of S. pneumoniae serotypes 1 , 3, 4, 5, 6A, 7F, 9V, 14, 18C, 19A, 19F, 23F and 8.8 ⁇ g/mL of type 6B in 5 mM succinate buffer
- 13vPnC (pH 5.8) and 0.85% NaCI, which may also be formulated with or without an adjuvant (e.g., an aluminum adjuvant).
- the 13vPnC may also be formulated with or without an adjuvant, such as 0.25 mg aluminum/ml aluminum phosphate (AIPO 4 ). It was observed in Example 3, that 13vPnC formulated without AIPO 4 and filled in BD Hypak SCFTM syringes (capped with Hypak plungers) failed visual inspection due to the observation of particulates, wherein further studies revealed that the particulates were in part a result of protein-polysaccharide interactions with silicone. In the following example, syringes (and plungers) from various vendors were evaluated with 13vPnC formulations, wherein shipping and handling conditions were simulated via agitation (described below).
- the 13vPnC was formulated in 5 mM succinate buffer containing 0.85% NaCI and 4.4 ⁇ g/ml of S. pneumoniae serotypes 1 , 3, 4, 5, 6A, 7F, 9V, 14, 18C, 19A, 19F and 23F and 8.8 ⁇ g/ml of S. pneumoniae serotype 6B, with and without 0.25 mg/ml aluminum phosphate.
- the container means tested are listed below in Table 6.
- VWR® signature Digital Multitube vortexer (Catalog No. 14005-826) was used to agitate the samples.
- the syringes filled with 13vPnC were placed horizontal and fixed by the two support plates of the vortexer. Samples were held at horizontal position and agitated at 500 rpm pause mode at 2-8°C for twenty-four hours.
- the agitation conditions were optimized based on the antigenicity loss of the two controls: (1 ) the worst-case control (positive control, high silicone; FIG. 2) and (2) the best-case control (negative control, no silicone; FIG. 3). The conditions were then optimized such that the antigenicity loss was low in positive control, yet detectable in the negative control. This was to ensure that the agitation was neither too weak to produce precipitation in the syringes; nor too strong, such that the precipitation might be caused by factors other than the silicone interaction (e.g., by shear forces). Thus, agitation at 500 rpm (pause mode) for twenty-four hours was chosen as the most suitable agitation condition, while a temperature of 2-
- BD Hypak syringe control 1
- the BD baked syringe syringe 3; 0.1 mg silicone
- the BD high viscosity syringe 5
- the B ⁇ nderGlas PS4 syringe syringe 8, 0.14 mg silicone
- the smallest antigenicity losses of the 13vPnC formulated with AIPO 4 occurred in the syringes with lower silicone levels.
- the Vetter syringes (FIG. 4) and the Schott TopPac plastic syringes were most similar to un-siliconized syringes (FIG. 2), both demonstrating minor antigenicity losses for 13vPnC formulated with AIPO 4 .
- AIPO 4 (FIG. 7B) had antigenicity losses ranging from about 5% to about 30% (depending on serotype).
- the recombinant lipidated N. meningitidis 2086 protein (rl_P2086) used in this example was expressed and purified as follows.
- the rl_P2086 was expressed recombinantly in E. coli utilizing a native leader sequence. Standard fermentation protocols for E. coli using animal-free defined medium and subsequent cell lysis were followed.
- Recombinant lipidated N. meningitidis 2086 protein was purified from the membrane pellet with 5OmM Tris-HCI/5mM EDTA/1 % sarcosyl pH 8.
- This sarcosyl extract was adjusted to 1 % Zwittergent 3-14 (Z3-14) and dialyzed twice against a 30 fold excess of 5OmM Tris-HCI/5mM EDTA/1 % Z3-14.
- the dialyzed rl_P2086 extract was precipitated with 90% ethanol to remove remaining sarcosyl, and solubilized with 5OmM Tris-HCI/5mM EDTA/1 % Z3-14 pH 8. Insoluble material was removed by centrifugation, the supernatant was passed over an anion exchange chromatography column, and rl_P2086 was collected in the unbound fraction.
- the unbound material was then dialyzed twice against a 30-fold excess of 25mM NaAc/1 % Z3-14 pH 4.5, and passed over a cation exchange chromatography column.
- the rl_P2086 was eluted with a 0-0.3M NaCI gradient and stored frozen (-25 0 C).
- the purified rl_P2086 was then formulated with 150 mM NaCI, 0.020% TweenTM80, 0.25 mg AI/mL Of AIPO 4 , and in the following buffers: 10 mM phosphate buffer at pH 7.0 and 5 mM succinate buffer at pH 6.0. Table 8 compares protein binding percentage to the AIPO 4 adjuvant.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Molecular Biology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biochemistry (AREA)
- Virology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Inorganic Chemistry (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Medicinal Preparation (AREA)
- Medical Preparation Storing Or Oral Administration Devices (AREA)
- Infusion, Injection, And Reservoir Apparatuses (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
Claims
Priority Applications (41)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EA200870480A EA017436B1 (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| PL13185292.3T PL2676679T5 (en) | 2006-04-26 | 2007-04-19 | Formulations which stabilize and inhibit precipitation of immunogenic compositions |
| KR1020227013826A KR20220061256A (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| PL13186454T PL2679245T3 (en) | 2006-04-26 | 2007-04-19 | Formulations which stabilize and inhibit precipitation of immunogenic compositions |
| KR1020177031015A KR20170122857A (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| PL07760907T PL2010219T3 (en) | 2006-04-26 | 2007-04-19 | Formulations which stabilize and inhibit precipitation of immunogenic compositions |
| EP13186453.0A EP2679244B1 (en) | 2006-04-26 | 2007-04-19 | Formulations which stabilize and inhibit precipitation of immunogenic compositions |
| KR20147030833A KR20140132779A (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| AU2007243019A AU2007243019B2 (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| MX2014003400A MX359741B (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions. |
| SI200731706T SI2010219T1 (en) | 2006-04-26 | 2007-04-19 | Formulations which stabilize and inhibit precipitation of immunogenic compositions |
| EP13186454.8A EP2679245B1 (en) | 2006-04-26 | 2007-04-19 | Formulations which stabilize and inhibit precipitation of immunogenic compositions |
| KR1020137033305A KR101514913B1 (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| MX2008013572A MX2008013572A (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions. |
| KR1020187034525A KR102093269B1 (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| KR1020167011184A KR20160049063A (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| BRPI0710918A BRPI0710918B8 (en) | 2006-04-26 | 2007-04-19 | formulation that stabilizes a polysaccharide-protein conjugate |
| CN2007800147343A CN101500612B (en) | 2006-04-26 | 2007-04-19 | Novel formulations that stabilize and inhibit precipitation of immunogenic compositions |
| RU2008141680/15A RU2482878C2 (en) | 2006-04-26 | 2007-04-19 | New compositions stabilising immunogenic compositions and inhibiting deposition thereof |
| CA2650056A CA2650056C (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| DK07760907.1T DK2010219T3 (en) | 2006-04-26 | 2007-04-19 | Formulations that stabilize and prevent precipitation of immunogenic compositions |
| JP2009507900A JP5612305B2 (en) | 2006-04-26 | 2007-04-19 | New formulations that stabilize immunogenic compositions and inhibit precipitation |
| HRP20151203TT HRP20151203T1 (en) | 2006-04-26 | 2007-04-19 | Formulations that stabilize and inhibit the deposition of immunogenic preparations |
| KR1020207007952A KR20200032268A (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| EP13185292.3A EP2676679B2 (en) | 2006-04-26 | 2007-04-19 | Use of formulations to stabilize and inhibit precipitation of immunogenic compositions |
| KR1020217027284A KR20210111326A (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| ES07760907.1T ES2557504T3 (en) | 2006-04-26 | 2007-04-19 | Formulations that stabilize and inhibit the precipitation of immunogenic compositions |
| BR122020000285-9A BR122020000285B1 (en) | 2006-04-26 | 2007-04-19 | FORMULATION THAT INHIBITES SILICONE-INDUCED AGGREGATION OF A POLYSACCHARIDE-PROTEIN CONJUGATE COMPRISED IN A SILICONIZED CONTAINER MEDIUM |
| EP07760907.1A EP2010219B1 (en) | 2006-04-26 | 2007-04-19 | Formulations which stabilize and inhibit precipitation of immunogenic compositions |
| PL13186453T PL2679244T3 (en) | 2006-04-26 | 2007-04-19 | Formulations which stabilize and inhibit precipitation of immunogenic compositions |
| EP19155604.2A EP3517129A1 (en) | 2006-04-26 | 2007-04-19 | Immunogenic compositions |
| KR1020087028865A KR101514847B1 (en) | 2006-04-26 | 2007-04-19 | A new formulation for stabilizing the immunogenic composition and inhibiting its precipitation |
| IL194753A IL194753A (en) | 2006-04-26 | 2008-10-22 | Formulations which stabilize and inhibit precipitation of immunogenic compositions |
| AU2012216628A AU2012216628B9 (en) | 2006-04-26 | 2012-09-04 | Novel Formulations which Stabilize and Inhibit Precipitation of Immunogenic Compositions |
| IL239788A IL239788A0 (en) | 2006-04-26 | 2015-07-05 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| IL239978A IL239978B (en) | 2006-04-26 | 2015-07-16 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| CY20191100130T CY1121251T1 (en) | 2006-04-26 | 2019-01-30 | PREPARATIONS THAT STABILIZE AND INHERE THE DEPRECIATION OF IMMUNOGRAPHIC COMPOSITIONS |
| LTPA2022509C LTC2676679I2 (en) | 2006-04-26 | 2022-06-08 | |
| FR22C1027C FR22C1027I2 (en) | 2006-04-26 | 2022-06-09 | Use of formulations stabilizing and inhibiting the precipitation of immunogenic compositions |
| LU00267C LUC00267I2 (en) | 2006-04-26 | 2022-06-10 | |
| CY2022016C CY2022016I2 (en) | 2006-04-26 | 2022-06-10 | PREPARATIONS WHICH STABILIZING AND INHIBITING THE SEDITION OF IMMUNE COMPOSITIONS |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US79526106P | 2006-04-26 | 2006-04-26 | |
| US60/795,261 | 2006-04-26 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2007127665A2 true WO2007127665A2 (en) | 2007-11-08 |
| WO2007127665A3 WO2007127665A3 (en) | 2008-11-13 |
Family
ID=38441477
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2007/066959 Ceased WO2007127665A2 (en) | 2006-04-26 | 2007-04-19 | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
Country Status (31)
| Country | Link |
|---|---|
| US (3) | US7935787B2 (en) |
| EP (5) | EP2676679B2 (en) |
| JP (7) | JP5612305B2 (en) |
| KR (9) | KR20160049063A (en) |
| CN (4) | CN104857524B (en) |
| AR (2) | AR060665A1 (en) |
| AU (1) | AU2007243019B2 (en) |
| BR (2) | BRPI0710918B8 (en) |
| CA (3) | CA2873346C (en) |
| CL (1) | CL2007001185A1 (en) |
| CR (1) | CR10405A (en) |
| CY (2) | CY1116966T1 (en) |
| DK (4) | DK2010219T3 (en) |
| EA (2) | EA017436B1 (en) |
| ES (4) | ES2885762T3 (en) |
| FI (2) | FI2676679T4 (en) |
| FR (1) | FR22C1027I2 (en) |
| HR (3) | HRP20190198T4 (en) |
| HU (5) | HUE058394T2 (en) |
| IL (3) | IL194753A (en) |
| LT (2) | LT2676679T (en) |
| LU (1) | LUC00267I2 (en) |
| MX (3) | MX2008013572A (en) |
| PL (4) | PL2676679T5 (en) |
| PT (4) | PT2010219E (en) |
| RU (1) | RU2482878C2 (en) |
| SI (4) | SI2679244T1 (en) |
| TR (1) | TR201902402T4 (en) |
| TW (1) | TW200806315A (en) |
| WO (1) | WO2007127665A2 (en) |
| ZA (1) | ZA200809167B (en) |
Cited By (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010109323A1 (en) | 2009-03-24 | 2010-09-30 | Novartis Ag | Adjuvanting meningococcal factor h binding protein |
| WO2010109324A1 (en) * | 2009-03-24 | 2010-09-30 | Novartis Ag | Combinations of meningococcal factor h binding protein and pneumococcal saccharide conjugates |
| WO2011161653A1 (en) | 2010-06-25 | 2011-12-29 | Novartis Ag | Combinations of meningococcal factor h binding proteins |
| WO2013098589A1 (en) | 2011-12-29 | 2013-07-04 | Novartis Ag | Adjuvanted combinations of meningococcal factor h binding proteins |
| US8840907B2 (en) | 2001-09-06 | 2014-09-23 | Novartis Vaccines And Diagnostics S.R.L. | Isolated protein and compositions comprising the protein |
| US8980286B2 (en) | 2002-11-22 | 2015-03-17 | Novartis Ag | Multiple variants of meningococcal protein NBM1870 |
| US9067987B2 (en) | 1999-10-29 | 2015-06-30 | Glaxosmithkline Biologicals Sa | Neisserial antigenic peptides |
| USRE45587E1 (en) | 2001-07-26 | 2015-06-30 | Glaxosmithkline Biologicals Sa | Vaccines comprising aluminum adjuvants and histidine |
| US9150898B2 (en) | 2000-02-28 | 2015-10-06 | Glaxosmithkline Biologicals Sa | Heterologous expression of Neisserial proteins |
| US9156894B2 (en) | 2005-11-25 | 2015-10-13 | Glaxosmithkline Biologicals Sa | Chimeric, hybrid and tandem polypeptides of meningococcal NMB1870 |
| US9205143B2 (en) | 2009-04-30 | 2015-12-08 | Coley Pharmaceutical Group Inc. | Pneumococcal vaccine and uses thereof |
| US9249196B2 (en) | 1998-05-01 | 2016-02-02 | Glaxosmithkline Biologicals Sa | Neisseria meningitidis antigens and compositions |
| US9364528B1 (en) | 1999-05-19 | 2016-06-14 | Glaxosmithkline Biologicals Sa | Combination neisserial compositions |
| US9468673B2 (en) | 2008-02-21 | 2016-10-18 | Glaxosmithkline Biologicals Sa | Meningococcal fHBP polypeptides |
| US9556240B2 (en) | 2010-08-23 | 2017-01-31 | Wyeth Llc | Stable formulations of Neisseria meningitidis rLP2086 antigens |
| US9561269B2 (en) | 2012-03-09 | 2017-02-07 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US9623101B2 (en) | 2001-10-11 | 2017-04-18 | Wyeth Holdings Llc | Immunogenic compositions for the prevention and treatment of meningococcal disease |
| US9757443B2 (en) | 2010-09-10 | 2017-09-12 | Wyeth Llc | Non-lipidated variants of Neisseria meningitidis ORF2086 antigens |
| US9802987B2 (en) | 2013-03-08 | 2017-10-31 | Pfizer Inc. | Immunogenic fusion polypeptides |
| US9822150B2 (en) | 2013-09-08 | 2017-11-21 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US10183070B2 (en) | 2017-01-31 | 2019-01-22 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US10195264B2 (en) | 2004-04-22 | 2019-02-05 | Glaxosmithkline Biologicals S.A. | Immunising against meningococcal serogroup Y using proteins |
| US10196429B2 (en) | 2012-03-09 | 2019-02-05 | Pfizer Inc. | Neisseria meningitidis composition and methods thereof |
| US10376573B2 (en) | 2012-06-14 | 2019-08-13 | Glaxosmithkline Biologicals Sa | Vaccines for serogroup X meningococcus |
| EP3585427A4 (en) * | 2017-02-24 | 2020-12-30 | Merck Sharp & Dohme Corp. | PROCESS FOR IMPROVING THE FILTERABILITY OF POLYSACCHARIDE-PROTEIN CONJUGATE REACTIONS |
| US10888611B2 (en) | 2015-02-19 | 2021-01-12 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US11090374B2 (en) | 2017-02-24 | 2021-08-17 | Merck Sharp & Dohme Corp. | Enhancing immunogenicity of Streptococcus pneumoniae polysaccharide-protein conjugates |
| US11116828B2 (en) | 2017-12-06 | 2021-09-14 | Merck Sharp & Dohme Corp. | Compositions comprising Streptococcus pneumoniae polysaccharide-protein conjugates and methods of use thereof |
| US11197921B2 (en) | 2017-01-31 | 2021-12-14 | Merck Sharp & Dohme Corp. | Methods for making polysaccharide-protein conjugates |
| US11389540B2 (en) | 2017-09-07 | 2022-07-19 | Merck Sharp & Dohme Llc | Pneumococcal polysaccharides and their use in immunogenic polysaccharide-carrier protein conjugates |
| US11395849B2 (en) | 2017-09-07 | 2022-07-26 | Merck Sharp & Dohme Llc | Pneumococcal polysaccharides and their use in immunogenic polysaccharide-carrier protein conjugates |
| US11642406B2 (en) | 2018-12-19 | 2023-05-09 | Merck Sharp & Dohme Llc | Compositions comprising Streptococcus pneumoniae polysaccharide-protein conjugates and methods of use thereof |
| US11896656B2 (en) | 2018-04-30 | 2024-02-13 | Merck Sharp & Dohme Llc | Methods for providing a homogenous solution of lyophilized mutant diptheria toxin in dimethylsulfoxide |
| US11992521B2 (en) | 2018-04-30 | 2024-05-28 | Merck Sharp & Dohme Llc | Methods for producing Streptococcus pneumoniae capsular polysaccharide carrier protein conjugates |
| US12144855B2 (en) | 2018-04-30 | 2024-11-19 | Merck Sharp & Dohme Llc | Methods for producing streptococcus pneumoniae capsular polysaccharide carrier protein conjugates from lyospheres |
| US12383611B2 (en) | 2019-09-27 | 2025-08-12 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
Families Citing this family (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0419408D0 (en) * | 2004-09-01 | 2004-10-06 | Chiron Srl | 741 chimeric polypeptides |
| US7955605B2 (en) * | 2005-04-08 | 2011-06-07 | Wyeth Llc | Multivalent pneumococcal polysaccharide-protein conjugate composition |
| TW200806315A (en) | 2006-04-26 | 2008-02-01 | Wyeth Corp | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| US8808707B1 (en) | 2006-05-08 | 2014-08-19 | Wyeth Llc | Pneumococcal dosing regimen |
| US8883146B2 (en) | 2007-11-30 | 2014-11-11 | Abbvie Inc. | Protein formulations and methods of making same |
| EP2234637A2 (en) * | 2007-12-21 | 2010-10-06 | GlaxoSmithKline Biologicals S.A. | Vaccines for malaria |
| ES2614807T3 (en) * | 2010-06-04 | 2017-06-02 | Wyeth Llc | Vaccine Formulations |
| US8802603B2 (en) | 2010-06-17 | 2014-08-12 | Becton, Dickinson And Company | Medical components having coated surfaces exhibiting low friction and low reactivity |
| FR2966044B1 (en) * | 2010-10-18 | 2012-11-02 | Sanofi Pasteur | METHOD FOR CONDITIONING A VACCINE CONTAINING AN ALUMINUM ADJUVANT |
| JP5919606B2 (en) | 2010-11-11 | 2016-05-18 | アッヴィ バイオテクノロジー リミテッド | Improved high concentration anti-TNF alpha antibody liquid formulation |
| CN103391714A (en) * | 2010-12-10 | 2013-11-13 | 默沙东公司 | Novel formulations which mitigate agitation-induced aggregation of immunogenic compositions |
| ES2983239T3 (en) | 2012-06-26 | 2024-10-22 | Lg Electronics Inc | Video decoding apparatus and video encoding apparatus |
| JOP20200175A1 (en) * | 2012-07-03 | 2017-06-16 | Novartis Ag | Syringe |
| LT3060256T (en) * | 2013-10-25 | 2019-07-10 | Bayer Pharma Aktiengesellschaft | A novel stable formulation |
| NZ721943A (en) * | 2014-01-21 | 2022-07-01 | Pfizer | Immunogenic compositions comprising conjugated capsular saccharide antigens and uses thereof |
| US11160855B2 (en) | 2014-01-21 | 2021-11-02 | Pfizer Inc. | Immunogenic compositions comprising conjugated capsular saccharide antigens and uses thereof |
| PE20180460A1 (en) * | 2015-06-08 | 2018-03-06 | Serum Inst Of India Private Ltd | METHODS TO IMPROVE THE ADSORPTION OF POLYSACCHARIDE-PROTEIN CONJUGATES AND A MULTIVALENT VACCINE FORMULATION OBTAINED THROUGH THE SAME |
| TWI756893B (en) * | 2015-07-21 | 2022-03-01 | 美商輝瑞股份有限公司 | Immunogenic compositions comprising conjugated capsular saccharide antigens, kits comprising the same and uses thereof |
| KR102429287B1 (en) * | 2015-12-10 | 2022-08-05 | 가부시끼가이샤 메니콘 | Peptide composition |
| LT3506935T (en) | 2016-09-02 | 2024-04-25 | Sanofi Pasteur, Inc. | Neisseria meningitidis vaccine |
| US11951165B2 (en) | 2016-12-30 | 2024-04-09 | Vaxcyte, Inc. | Conjugated vaccine carrier proteins |
| MX2019007910A (en) | 2016-12-30 | 2019-12-05 | Sutrovax Inc | POLYPEPTIDE-ANTIGEN CONJUGATES WITH NON-NATURAL AMINO ACIDS. |
| CN106620681A (en) * | 2017-01-12 | 2017-05-10 | 南京佰泰克生物技术有限公司 | Cell lysis solution, kit and application of cell lysis solution to preparation of tumor whole cell antigen loaded DC tumor vaccine |
| US20200222550A1 (en) | 2017-01-31 | 2020-07-16 | Merck Sharp & Dohme Corp. | Methods for production of capsular polysaccharide protein conjugates from streptococcus pneumoniae serotype 19f |
| US20200054740A1 (en) | 2017-02-24 | 2020-02-20 | Merck Sharp & Dohme Corp. | Pneumococcal conjugate vaccine formulations |
| EP3700565A4 (en) * | 2017-10-25 | 2021-09-29 | Merck Sharp & Dohme Corp. | VACCINES WITH ADJUVANT |
| US11260119B2 (en) | 2018-08-24 | 2022-03-01 | Pfizer Inc. | Escherichia coli compositions and methods thereof |
| JP2022502493A (en) * | 2018-09-24 | 2022-01-11 | バイオヴァクシーズ インコーポレイテッド | (Ii) Haptenized self-vaccine and its use |
| US12138302B2 (en) | 2020-10-27 | 2024-11-12 | Pfizer Inc. | Escherichia coli compositions and methods thereof |
| US12357681B2 (en) | 2020-12-23 | 2025-07-15 | Pfizer Inc. | E. coli FimH mutants and uses thereof |
| US20240230545A9 (en) | 2021-02-26 | 2024-07-11 | Biological E Limited | Activated-quenched polysaccharide and improved methods for quantification of polysaccharide in a vaccine composition |
| KR20230175284A (en) * | 2021-05-28 | 2023-12-29 | 화이자 인코포레이티드 | Immunogenic compositions comprising conjugated capsular saccharide antigens and uses thereof |
Family Cites Families (73)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1189340A (en) * | 1967-09-13 | 1970-04-22 | American Cyanamid Co | Antigen-Adjuvant Composition |
| US4902506A (en) | 1983-07-05 | 1990-02-20 | The University Of Rochester | Immunogenic conjugates |
| US4673574A (en) | 1981-08-31 | 1987-06-16 | Anderson Porter W | Immunogenic conjugates |
| SE8405493D0 (en) | 1984-11-01 | 1984-11-01 | Bror Morein | IMMUNOGENT COMPLEX AND KITCHEN FOR PREPARING IT AND USING IT AS IMMUNOSTIMENTING AGENTS |
| US5078996A (en) | 1985-08-16 | 1992-01-07 | Immunex Corporation | Activation of macrophage tumoricidal activity by granulocyte-macrophage colony stimulating factor |
| US5057540A (en) | 1987-05-29 | 1991-10-15 | Cambridge Biotech Corporation | Saponin adjuvant |
| FR2630115B1 (en) * | 1988-04-14 | 1994-10-28 | Merieux Inst | PROCESS FOR STABILIZING HUMAN ALBUMIN SOLUTIONS AND SOLUTION OBTAINED |
| US4912094B1 (en) | 1988-06-29 | 1994-02-15 | Ribi Immunochem Research Inc. | Modified lipopolysaccharides and process of preparation |
| CA2017507C (en) | 1989-05-25 | 1996-11-12 | Gary Van Nest | Adjuvant formulation comprising a submicron oil droplet emulsion |
| ATE129906T1 (en) * | 1990-07-19 | 1995-11-15 | Nardino Righi | SINGLE USE SAFETY SYRINGE. |
| CA2059692C (en) * | 1991-01-28 | 2004-11-16 | Peter J. Kniskern | Pneumoccoccal polysaccharide conjugate vaccine |
| IL101715A (en) | 1991-05-02 | 2005-06-19 | Amgen Inc | Recombinant dna-derived cholera toxin subunit analogs |
| US5769047A (en) | 1991-12-23 | 1998-06-23 | Zoche; Michael | Engine with oil separator |
| IT1253009B (en) | 1991-12-31 | 1995-07-10 | Sclavo Ricerca S R L | DETOXIFIED IMMUNOGENIC MUTANTS OF COLERIC TOXIN AND TOXIN LT, THEIR PREPARATION AND USE FOR THE PREPARATION OF VACCINES |
| EP0671948B1 (en) | 1992-06-25 | 1997-08-13 | SMITHKLINE BEECHAM BIOLOGICALS s.a. | Vaccine composition containing adjuvants |
| ES2231770T3 (en) | 1993-03-05 | 2005-05-16 | Wyeth Holdings Corporation | NEW PLASMIDS FOR THE PRODUCTION OF CRM PROTEIN AND DIFTERIC TOXIN. |
| GB9326253D0 (en) | 1993-12-23 | 1994-02-23 | Smithkline Beecham Biolog | Vaccines |
| JP3696267B2 (en) | 1994-02-28 | 2005-09-14 | 天野エンザイム株式会社 | Method for stabilizing bioactive protein |
| DE4407489A1 (en) * | 1994-03-07 | 1995-09-14 | Bayer Ag | Vaccine for the prevention of respiratory and reproductive diseases of the pig |
| US5571515A (en) | 1994-04-18 | 1996-11-05 | The Wistar Institute Of Anatomy & Biology | Compositions and methods for use of IL-12 as an adjuvant |
| US6207646B1 (en) | 1994-07-15 | 2001-03-27 | University Of Iowa Research Foundation | Immunostimulatory nucleic acid molecules |
| UA56132C2 (en) | 1995-04-25 | 2003-05-15 | Смітклайн Бічем Байолоджікалс С.А. | Vaccine composition (variants), method for stabilizing qs21 providing resistance against hydrolysis (variants), method for manufacturing vaccine |
| US6355255B1 (en) | 1998-12-07 | 2002-03-12 | Regents Of The University Of Minnesota | Streptococcal C5a peptidase vaccine |
| US5846547A (en) * | 1996-01-22 | 1998-12-08 | Regents Of The University Of Minnesota | Streptococcal C5a peptidase vaccine |
| US6403098B1 (en) | 1996-09-26 | 2002-06-11 | Merck & Co., Inc. | Rotavirus vaccine formulations |
| US5932223A (en) | 1996-09-26 | 1999-08-03 | Merck & Co., Inc. | Rotavirus vaccine formulations |
| RU2098109C1 (en) | 1996-09-26 | 1997-12-10 | Александр Григорьевич Чучалин | Antiallergic, antiinflammatory agent, method of its preparing, curative and cosmetic agent and its use |
| JP4078406B2 (en) * | 1997-01-22 | 2008-04-23 | 有限会社コーキ・エンジニアリング | Method for manufacturing syringe cylinder |
| SK138199A3 (en) * | 1997-04-08 | 2000-06-12 | Merck & Co Inc | Human papillomavirus antigen formulation and method for stabilizing a population of virus-like purified particles, derived from protein l1 or protein l1 and l2 of human papillomavirus |
| US6113918A (en) | 1997-05-08 | 2000-09-05 | Ribi Immunochem Research, Inc. | Aminoalkyl glucosamine phosphate compounds and their use as adjuvants and immunoeffectors |
| FR2763244B1 (en) * | 1997-05-14 | 2003-08-01 | Pasteur Merieux Serums Vacc | MULTIVALENT VACCINE COMPOSITION WITH MIXED CARRIER |
| US6224880B1 (en) * | 1997-09-24 | 2001-05-01 | Merck & Co., Inc. | Immunization against Streptococcus pneumoniae using conjugated and unconjugated pneumoccocal polysaccharide vaccines |
| CA2264970A1 (en) * | 1998-03-10 | 1999-09-10 | American Cyanamid Company | Antigenic conjugates of conserved lipolysaccharides of gram negative bacteria |
| PT1117435E (en) | 1998-09-30 | 2008-02-21 | Us Gov Univ Health Sciences | Mutant cholera holotoxin as an adjuvant |
| IL145043A0 (en) | 1999-03-19 | 2002-06-30 | Smithkline Beecham Biolog | Vaccine |
| GB9909077D0 (en) | 1999-04-20 | 1999-06-16 | Smithkline Beecham Biolog | Novel compositions |
| GB9928196D0 (en) * | 1999-11-29 | 2000-01-26 | Chiron Spa | Combinations of B, C and other antigens |
| JP4948731B2 (en) * | 1999-12-02 | 2012-06-06 | ノバルティス バクシンズ アンド ダイアグノスティックス,インコーポレーテッド | Compositions and methods for stabilizing biological molecules during lyophilization |
| DE10012370A1 (en) * | 2000-03-14 | 2001-09-27 | Chiron Behring Gmbh & Co | Use of oil-in-water emulsion as vaccine adjuvant, particularly for influenza and pneumococcal vaccines, administered at different site from the vaccine |
| HU228264B1 (en) | 2000-06-08 | 2013-02-28 | Intercell Ag | Immunostimulatory oligodeoxynucleotides |
| GB0108364D0 (en) | 2001-04-03 | 2001-05-23 | Glaxosmithkline Biolog Sa | Vaccine composition |
| HU227893B1 (en) * | 2000-06-29 | 2012-05-29 | Glaxosmithkline Biolog Sa | Vaccine compositions |
| KR100385711B1 (en) * | 2000-07-05 | 2003-05-27 | 녹십자백신 주식회사 | The quadrivalent combination vaccine including diphtheria toxoid, tetanus toxoid, whole cell pertussis and hepatitis b surface antigen and the preparation thereof |
| CA2416224C (en) * | 2000-07-10 | 2015-08-25 | Edwin W. Ades | Multiple antigenic peptides immunogenic against streptococcus pneumoniae |
| GB0025414D0 (en) * | 2000-10-16 | 2000-11-29 | Consejo Superior Investigacion | Nanoparticles |
| AT410635B (en) | 2000-10-18 | 2003-06-25 | Cistem Biotechnologies Gmbh | VACCINE COMPOSITION |
| JP2004525921A (en) * | 2001-03-19 | 2004-08-26 | イオマイ コーポレイシヨン | Transdermal immunization patch |
| BR0210216A (en) | 2001-06-07 | 2004-06-08 | Wyeth Corp | Immunogenic cholera holotoxin, mutant (ct-crm), immunogenic composition, methods to increase the immune response of a vertebrate host to an antigen, and to produce an immunogenic, mutant cholera holotoxin, isolated and purified nucleic acid molecule, sequence host cell, and use of a mutant cholera holotoxin |
| CN1977971A (en) | 2001-06-07 | 2007-06-13 | 惠氏控股有限公司 | Mutant forms of cholera holotoxin as an adjuvant |
| GB0115176D0 (en) * | 2001-06-20 | 2001-08-15 | Chiron Spa | Capular polysaccharide solubilisation and combination vaccines |
| AU2002330681C1 (en) * | 2001-07-26 | 2015-04-02 | Glaxosmithkline Biologicals S.A. | Vaccines comprising aluminium adjuvants and histidine |
| MX339524B (en) * | 2001-10-11 | 2016-05-30 | Wyeth Corp | NEW IMMUNOGENIC COMPOSITIONS FOR THE PREVENTION AND TREATMENT OF MENINGOCOCICAL DISEASE. |
| ATE556591T1 (en) * | 2001-11-08 | 2012-05-15 | Abbott Biotherapeutics Corp | STABLE PHARMACEUTICAL LIQUID FORMULATION OF IGG ANTIBODIES |
| EP1590459A2 (en) | 2003-01-15 | 2005-11-02 | Wyeth Holdings Corporation | Methods for increasing neisseria protein expression and compositions thereof |
| ES2411080T3 (en) * | 2003-01-30 | 2013-07-04 | Novartis Ag | Injectable vaccines against multiple serogroups of meningococci |
| EP1592440A4 (en) * | 2003-02-10 | 2007-07-11 | Elan Pharm Inc | Immunoglobulin formulation and method of preparation thereof |
| CA2519511A1 (en) | 2003-03-17 | 2004-09-30 | Wyeth Holdings Corporation | Mutant cholera holotoxin as an adjuvant and an antigen carrier protein |
| JP2006525330A (en) * | 2003-04-16 | 2006-11-09 | ワイエス・ホールディングス・コーポレーション | Novel immunogenic composition for prevention and treatment of meningococcal disease |
| WO2005000246A2 (en) * | 2003-06-25 | 2005-01-06 | Elan Pharmaceuticals, Inc. | Methods and compositions for treating rheumatoid arthritis |
| UA67144A (en) | 2003-07-29 | 2004-06-15 | Inst Exprm & Clinical Veterina | Method for production of inactivated vaccine against salmonellosis and escherichiosis of animals |
| CN102151331B (en) | 2003-08-06 | 2013-10-30 | 美国政府健康及人类服务部 | Polysaccharide-protein conjugate vaccines |
| DE10348550A1 (en) * | 2003-10-20 | 2005-06-16 | Hexal Biotech Forschungsgmbh | Stable aqueous G-CSF-containing compositions |
| RU37462U1 (en) | 2004-01-16 | 2004-04-27 | Бычкова Ольга Владимировна | TEST |
| JP2008518953A (en) * | 2004-11-01 | 2008-06-05 | ザ ブリガム アンド ウィメンズ ホスピタル インコーポレイテッド | Streptococcus modified polysaccharides and their use |
| JP2008528638A (en) * | 2005-01-28 | 2008-07-31 | ワイス | Polypeptide stabilized liquid formulations |
| US7709001B2 (en) | 2005-04-08 | 2010-05-04 | Wyeth Llc | Multivalent pneumococcal polysaccharide-protein conjugate composition |
| US7955605B2 (en) | 2005-04-08 | 2011-06-07 | Wyeth Llc | Multivalent pneumococcal polysaccharide-protein conjugate composition |
| US20070184072A1 (en) | 2005-04-08 | 2007-08-09 | Wyeth | Multivalent pneumococcal polysaccharide-protein conjugate composition |
| CN102716480B (en) * | 2005-04-08 | 2023-03-21 | 惠氏有限责任公司 | Multivalent pneumococcal polysaccharide-protein conjugate composition |
| GB0524066D0 (en) * | 2005-11-25 | 2006-01-04 | Chiron Srl | 741 ii |
| TW200806315A (en) | 2006-04-26 | 2008-02-01 | Wyeth Corp | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions |
| WO2007127668A2 (en) | 2006-04-26 | 2007-11-08 | Wyeth | Novel processes for coating container means which inhibit precipitation of polysaccharide-protein conjugate formulations |
| ES2615390T3 (en) | 2008-03-05 | 2017-06-06 | Sanofi Pasteur | Process to stabilize a vaccine composition containing adjuvant |
-
2007
- 2007-04-12 TW TW096112884A patent/TW200806315A/en unknown
- 2007-04-19 PT PT77609071T patent/PT2010219E/en unknown
- 2007-04-19 HR HRP20190198TT patent/HRP20190198T4/en unknown
- 2007-04-19 PL PL13185292.3T patent/PL2676679T5/en unknown
- 2007-04-19 EP EP13185292.3A patent/EP2676679B2/en active Active
- 2007-04-19 JP JP2009507900A patent/JP5612305B2/en active Active
- 2007-04-19 KR KR1020167011184A patent/KR20160049063A/en not_active Ceased
- 2007-04-19 DK DK07760907.1T patent/DK2010219T3/en active
- 2007-04-19 RU RU2008141680/15A patent/RU2482878C2/en active
- 2007-04-19 KR KR20147030833A patent/KR20140132779A/en not_active Ceased
- 2007-04-19 MX MX2008013572A patent/MX2008013572A/en active IP Right Grant
- 2007-04-19 PT PT131864548T patent/PT2679245T/en unknown
- 2007-04-19 ES ES13186454T patent/ES2885762T3/en active Active
- 2007-04-19 EP EP13186454.8A patent/EP2679245B1/en active Active
- 2007-04-19 PL PL13186454T patent/PL2679245T3/en unknown
- 2007-04-19 BR BRPI0710918A patent/BRPI0710918B8/en active IP Right Grant
- 2007-04-19 US US11/737,674 patent/US7935787B2/en active Active
- 2007-04-19 CA CA2873346A patent/CA2873346C/en active Active
- 2007-04-19 CN CN201510239424.8A patent/CN104857524B/en active Active
- 2007-04-19 KR KR1020207007952A patent/KR20200032268A/en not_active Ceased
- 2007-04-19 PT PT131864530T patent/PT2679244T/en unknown
- 2007-04-19 HU HUE13186453A patent/HUE058394T2/en unknown
- 2007-04-19 TR TR2019/02402T patent/TR201902402T4/en unknown
- 2007-04-19 EP EP13186453.0A patent/EP2679244B1/en active Active
- 2007-04-19 MX MX2014003400A patent/MX359741B/en unknown
- 2007-04-19 PL PL13186453T patent/PL2679244T3/en unknown
- 2007-04-19 HU HUE07760907A patent/HUE028137T2/en unknown
- 2007-04-19 CA CA2650056A patent/CA2650056C/en active Active
- 2007-04-19 HR HRP20151203TT patent/HRP20151203T1/en unknown
- 2007-04-19 AU AU2007243019A patent/AU2007243019B2/en active Active
- 2007-04-19 EA EA200870480A patent/EA017436B1/en active Protection Beyond IP Right Term
- 2007-04-19 EP EP19155604.2A patent/EP3517129A1/en active Pending
- 2007-04-19 ES ES07760907.1T patent/ES2557504T3/en active Active
- 2007-04-19 ES ES13186453T patent/ES2910432T3/en active Active
- 2007-04-19 SI SI200732187T patent/SI2679244T1/en unknown
- 2007-04-19 LT LTEP13185292.3T patent/LT2676679T/en unknown
- 2007-04-19 FI FIEP13185292.3T patent/FI2676679T4/en active
- 2007-04-19 SI SI200731706T patent/SI2010219T1/en unknown
- 2007-04-19 SI SI200732093T patent/SI2676679T2/en unknown
- 2007-04-19 PL PL07760907T patent/PL2010219T3/en unknown
- 2007-04-19 HU HUE13186454A patent/HUE055682T2/en unknown
- 2007-04-19 KR KR1020187034525A patent/KR102093269B1/en active Active
- 2007-04-19 DK DK13186453.0T patent/DK2679244T3/en active
- 2007-04-19 KR KR1020217027284A patent/KR20210111326A/en not_active Ceased
- 2007-04-19 CN CN201610269964.5A patent/CN105879021B/en active Active
- 2007-04-19 HU HUE13185292A patent/HUE042923T2/en unknown
- 2007-04-19 WO PCT/US2007/066959 patent/WO2007127665A2/en not_active Ceased
- 2007-04-19 KR KR1020137033305A patent/KR101514913B1/en active Active
- 2007-04-19 CA CA2803111A patent/CA2803111C/en active Active
- 2007-04-19 KR KR1020177031015A patent/KR20170122857A/en not_active Ceased
- 2007-04-19 DK DK13185292.3T patent/DK2676679T4/en active
- 2007-04-19 DK DK13186454.8T patent/DK2679245T3/en active
- 2007-04-19 KR KR1020087028865A patent/KR101514847B1/en not_active Ceased
- 2007-04-19 ES ES13185292T patent/ES2712956T5/en active Active
- 2007-04-19 SI SI200732180T patent/SI2679245T1/en unknown
- 2007-04-19 PT PT13185292T patent/PT2676679T/en unknown
- 2007-04-19 BR BR122020000285-9A patent/BR122020000285B1/en active IP Right Grant
- 2007-04-19 EA EA201200929A patent/EA023470B1/en active Protection Beyond IP Right Term
- 2007-04-19 CN CN201310587301.4A patent/CN103768615B/en active Active
- 2007-04-19 EP EP07760907.1A patent/EP2010219B1/en active Active
- 2007-04-19 CN CN2007800147343A patent/CN101500612B/en active Active
- 2007-04-19 KR KR1020227013826A patent/KR20220061256A/en not_active Ceased
- 2007-04-25 CL CL2007001185A patent/CL2007001185A1/en unknown
- 2007-04-26 AR ARP070101814A patent/AR060665A1/en not_active Application Discontinuation
-
2008
- 2008-10-22 IL IL194753A patent/IL194753A/en active IP Right Grant
- 2008-10-22 MX MX2018012171A patent/MX2018012171A/en unknown
- 2008-10-24 ZA ZA200809167A patent/ZA200809167B/en unknown
- 2008-10-28 CR CR10405A patent/CR10405A/en unknown
-
2011
- 2011-03-24 US US13/070,664 patent/US20110172393A1/en not_active Abandoned
-
2012
- 2012-09-28 US US13/631,573 patent/US8562999B2/en active Active
-
2013
- 2013-11-19 JP JP2013239284A patent/JP2014055169A/en not_active Withdrawn
-
2014
- 2014-07-14 JP JP2014144436A patent/JP6192115B2/en active Active
-
2015
- 2015-07-05 IL IL239788A patent/IL239788A0/en active IP Right Grant
- 2015-07-16 IL IL239978A patent/IL239978B/en active IP Right Grant
- 2015-11-26 CY CY20151101066T patent/CY1116966T1/en unknown
-
2016
- 2016-07-27 JP JP2016147591A patent/JP6321094B2/en active Active
-
2018
- 2018-01-05 AR ARP180100033A patent/AR110731A2/en not_active Application Discontinuation
- 2018-04-03 JP JP2018071339A patent/JP2018123145A/en not_active Withdrawn
-
2019
- 2019-01-30 CY CY20191100130T patent/CY1121251T1/en unknown
- 2019-07-10 JP JP2019128073A patent/JP2019194240A/en active Pending
-
2021
- 2021-08-03 JP JP2021127528A patent/JP2021176910A/en active Pending
- 2021-09-07 HR HRP20211417TT patent/HRP20211417T1/en unknown
-
2022
- 2022-06-08 LT LTPA2022509C patent/LTC2676679I2/lt unknown
- 2022-06-09 HU HUS2200028C patent/HUS2200028I1/en unknown
- 2022-06-09 FR FR22C1027C patent/FR22C1027I2/en active Active
- 2022-06-10 LU LU00267C patent/LUC00267I2/fr unknown
- 2022-06-13 FI FIC20220022C patent/FIC20220022I1/en unknown
Cited By (83)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9266929B2 (en) | 1998-05-01 | 2016-02-23 | Glaxosmithkline Biologicals Sa | Neisseria meningitidis antigens and compositions |
| US9249198B2 (en) | 1998-05-01 | 2016-02-02 | Glaxosmithkline Biologicals Sa | Neisseria meningitidis antigens and compositions |
| US9249196B2 (en) | 1998-05-01 | 2016-02-02 | Glaxosmithkline Biologicals Sa | Neisseria meningitidis antigens and compositions |
| US9364528B1 (en) | 1999-05-19 | 2016-06-14 | Glaxosmithkline Biologicals Sa | Combination neisserial compositions |
| US9610342B2 (en) | 1999-05-19 | 2017-04-04 | Glaxosmithkline Biologicals Sa | Combination neisserial compositions |
| US9067987B2 (en) | 1999-10-29 | 2015-06-30 | Glaxosmithkline Biologicals Sa | Neisserial antigenic peptides |
| US9267163B2 (en) | 2000-02-28 | 2016-02-23 | Glaxosmithkline Biologicals Sa | Hybrid expression of neisserial proteins |
| US9150898B2 (en) | 2000-02-28 | 2015-10-06 | Glaxosmithkline Biologicals Sa | Heterologous expression of Neisserial proteins |
| USRE45587E1 (en) | 2001-07-26 | 2015-06-30 | Glaxosmithkline Biologicals Sa | Vaccines comprising aluminum adjuvants and histidine |
| US9056075B2 (en) | 2001-09-06 | 2015-06-16 | Glaxosmithkline Biologicals Sa | Methods of inducing an immune response with compositions comprising a Neisseria meningitidis 741 protein |
| US9011869B2 (en) | 2001-09-06 | 2015-04-21 | Glaxosmithkline Biologicals Sa | Hybrid and tandem expression of Neisserial proteins |
| US8980277B2 (en) | 2001-09-06 | 2015-03-17 | Novartis Ag | Hybrid and tandem expression of neisserial proteins |
| US8840907B2 (en) | 2001-09-06 | 2014-09-23 | Novartis Vaccines And Diagnostics S.R.L. | Isolated protein and compositions comprising the protein |
| US10300122B2 (en) | 2001-10-11 | 2019-05-28 | Wyeth Holdings Llc | Immunogenic compositions for the prevention and treatment of meningococcal disease |
| US11116829B2 (en) | 2001-10-11 | 2021-09-14 | Wyeth Holdings Llc | Immunogenic compositions for the prevention and treatment of meningococcal disease |
| US9623101B2 (en) | 2001-10-11 | 2017-04-18 | Wyeth Holdings Llc | Immunogenic compositions for the prevention and treatment of meningococcal disease |
| US9757444B2 (en) | 2001-10-11 | 2017-09-12 | Wyeth Holdings Llc | Immunogenic compositions for the prevention and treatment of meningococcal disease |
| US9550814B2 (en) | 2002-11-22 | 2017-01-24 | Glaxosmithkline Biologicals Sa | Multiple variants of meningococcal protein NMB1870 |
| US10328142B2 (en) | 2002-11-22 | 2019-06-25 | Glaxosmithkline Biologicals Sa | Multiple variants of meningococcal protein NMB1870 |
| US8980286B2 (en) | 2002-11-22 | 2015-03-17 | Novartis Ag | Multiple variants of meningococcal protein NBM1870 |
| US10195264B2 (en) | 2004-04-22 | 2019-02-05 | Glaxosmithkline Biologicals S.A. | Immunising against meningococcal serogroup Y using proteins |
| US9156894B2 (en) | 2005-11-25 | 2015-10-13 | Glaxosmithkline Biologicals Sa | Chimeric, hybrid and tandem polypeptides of meningococcal NMB1870 |
| US9579372B2 (en) | 2008-02-21 | 2017-02-28 | Glaxosmithkline Biologicals Sa | Meningococcal fHBP polypeptides |
| US9468673B2 (en) | 2008-02-21 | 2016-10-18 | Glaxosmithkline Biologicals Sa | Meningococcal fHBP polypeptides |
| US10245311B2 (en) | 2009-03-24 | 2019-04-02 | Glaxosmithkline Biologicals Sa | Adjuvanting meningococcal factor H binding protein |
| US10568953B2 (en) | 2009-03-24 | 2020-02-25 | Glaxosmithkline Biologicals Sa | Adjuvanting meningococcal factor H binding protein |
| WO2010109324A1 (en) * | 2009-03-24 | 2010-09-30 | Novartis Ag | Combinations of meningococcal factor h binding protein and pneumococcal saccharide conjugates |
| EP3017826A1 (en) * | 2009-03-24 | 2016-05-11 | Novartis AG | Combinations of meningococcal factor h binding protein and pneumococcal saccharide conjugates |
| US9572884B2 (en) | 2009-03-24 | 2017-02-21 | Glaxosmithkline Biologicals Sa | Adjuvanting meningococcal factor H binding protein |
| EP2411048B1 (en) | 2009-03-24 | 2020-05-06 | GlaxoSmithKline Biologicals SA | Adjuvanting meningococcal factor h binding protein |
| AU2010227220B2 (en) * | 2009-03-24 | 2014-11-13 | Glaxosmithkline Biologicals S.A. | Combinations of meningococcal factor H binding protein and pneumococcal saccharide conjugates |
| US8834888B2 (en) | 2009-03-24 | 2014-09-16 | Novartis Ag | Adjuvanting meningococcal factor H binding protein |
| US20120064103A1 (en) * | 2009-03-24 | 2012-03-15 | Novartis Ag | Combinations of meningococcal factor h binding protein and pneumococcal saccharide conjugates |
| WO2010109323A1 (en) | 2009-03-24 | 2010-09-30 | Novartis Ag | Adjuvanting meningococcal factor h binding protein |
| CN102438649A (en) * | 2009-03-24 | 2012-05-02 | 诺华有限公司 | Meningococcal factor H binding proteins and pneumococcal binding saccharides |
| US9205143B2 (en) | 2009-04-30 | 2015-12-08 | Coley Pharmaceutical Group Inc. | Pneumococcal vaccine and uses thereof |
| US10478483B2 (en) | 2010-06-25 | 2019-11-19 | Glaxosmithkline Biologicals Sa | Combinations of meningococcal factor H binding proteins |
| WO2011161653A1 (en) | 2010-06-25 | 2011-12-29 | Novartis Ag | Combinations of meningococcal factor h binding proteins |
| EP3246044B1 (en) | 2010-08-23 | 2020-12-30 | Wyeth LLC | Stable formulations of neisseria meningitidis rlp2086 antigens |
| US9556240B2 (en) | 2010-08-23 | 2017-01-31 | Wyeth Llc | Stable formulations of Neisseria meningitidis rLP2086 antigens |
| EP2608805B1 (en) | 2010-08-23 | 2017-07-05 | Wyeth LLC | STABLE FORMULATIONS OF NEISSERIA MENINGITIDIS rLP2086 ANTIGENS |
| US9757443B2 (en) | 2010-09-10 | 2017-09-12 | Wyeth Llc | Non-lipidated variants of Neisseria meningitidis ORF2086 antigens |
| US11077180B2 (en) | 2010-09-10 | 2021-08-03 | Wyeth Llc | Non-lipidated variants of Neisseria meningitidis ORF2086 antigens |
| US10512681B2 (en) | 2010-09-10 | 2019-12-24 | Wyeth Llc | Non-lipidated variants of Neisseria meningitidis ORF2086 antigens |
| US10596246B2 (en) | 2011-12-29 | 2020-03-24 | Glaxosmithkline Biological Sa | Adjuvanted combinations of meningococcal factor H binding proteins |
| WO2013098589A1 (en) | 2011-12-29 | 2013-07-04 | Novartis Ag | Adjuvanted combinations of meningococcal factor h binding proteins |
| US9724402B2 (en) | 2012-03-09 | 2017-08-08 | Pfizer Inc. | Neisseria meningitidis composition and methods thereof |
| US11472850B2 (en) | 2012-03-09 | 2022-10-18 | Pfizer Inc. | Neisseria meningitidis composition and methods thereof |
| US10550159B2 (en) | 2012-03-09 | 2020-02-04 | Pfizer Inc. | Neisseria meningitidis composition and methods thereof |
| US10196429B2 (en) | 2012-03-09 | 2019-02-05 | Pfizer Inc. | Neisseria meningitidis composition and methods thereof |
| US9561269B2 (en) | 2012-03-09 | 2017-02-07 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US10829521B2 (en) | 2012-03-09 | 2020-11-10 | Pfizer Inc. | Neisseria meningitidis composition and methods thereof |
| US10376573B2 (en) | 2012-06-14 | 2019-08-13 | Glaxosmithkline Biologicals Sa | Vaccines for serogroup X meningococcus |
| US9802987B2 (en) | 2013-03-08 | 2017-10-31 | Pfizer Inc. | Immunogenic fusion polypeptides |
| US11680087B2 (en) | 2013-09-08 | 2023-06-20 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US9822150B2 (en) | 2013-09-08 | 2017-11-21 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US10899802B2 (en) | 2013-09-08 | 2021-01-26 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US10888611B2 (en) | 2015-02-19 | 2021-01-12 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US10813989B2 (en) | 2017-01-31 | 2020-10-27 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US12059461B2 (en) | 2017-01-31 | 2024-08-13 | Merck Sharp & Dohme Corp. | Methods for making polysaccharide-protein conjugates |
| US11730800B2 (en) | 2017-01-31 | 2023-08-22 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US10183070B2 (en) | 2017-01-31 | 2019-01-22 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US11197921B2 (en) | 2017-01-31 | 2021-12-14 | Merck Sharp & Dohme Corp. | Methods for making polysaccharide-protein conjugates |
| US10543267B2 (en) | 2017-01-31 | 2020-01-28 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
| US12295998B2 (en) | 2017-02-24 | 2025-05-13 | Merck Sharp & Dohme Llc | Enhancing immunogenicity of Streptococcus pneumoniae polysaccharide-protein conjugates |
| US11090374B2 (en) | 2017-02-24 | 2021-08-17 | Merck Sharp & Dohme Corp. | Enhancing immunogenicity of Streptococcus pneumoniae polysaccharide-protein conjugates |
| EP3585427A4 (en) * | 2017-02-24 | 2020-12-30 | Merck Sharp & Dohme Corp. | PROCESS FOR IMPROVING THE FILTERABILITY OF POLYSACCHARIDE-PROTEIN CONJUGATE REACTIONS |
| US11964023B2 (en) | 2017-09-07 | 2024-04-23 | Merck Sharp & Dohme Llc | Pneumococcal polysaccharides and their use in immunogenic polysaccharide-carrier protein conjugates |
| US11389540B2 (en) | 2017-09-07 | 2022-07-19 | Merck Sharp & Dohme Llc | Pneumococcal polysaccharides and their use in immunogenic polysaccharide-carrier protein conjugates |
| US11759523B2 (en) | 2017-09-07 | 2023-09-19 | Merck Sharp & Dohme Llc | Pneumococcal polysaccharides and their use in immunogenic polysaccharide-carrier protein conjugates |
| US11759510B2 (en) | 2017-09-07 | 2023-09-19 | Merck, Sharp & Dohme LLC | Pneumococcal polysaccharides and their use in immunogenic polysaccharide-carrier protein conjugates |
| US11759511B2 (en) | 2017-09-07 | 2023-09-19 | Merck Sharp & Dohme Llc | Pneumococcal polysaccharides and their use in immunogenic polysaccharide-carrier protein conjugates |
| US11395849B2 (en) | 2017-09-07 | 2022-07-26 | Merck Sharp & Dohme Llc | Pneumococcal polysaccharides and their use in immunogenic polysaccharide-carrier protein conjugates |
| US11850278B2 (en) | 2017-12-06 | 2023-12-26 | Merck Sharp & Dohme Llc | Compositions comprising Streptococcus pneumoniae polysaccharide-protein conjugates and methods of use thereof |
| US11116828B2 (en) | 2017-12-06 | 2021-09-14 | Merck Sharp & Dohme Corp. | Compositions comprising Streptococcus pneumoniae polysaccharide-protein conjugates and methods of use thereof |
| US12097250B2 (en) | 2017-12-06 | 2024-09-24 | Merck Sharp & Dohme Llc | Compositions comprising Streptococcus pneumoniae polysaccharide-protein conjugates and methods of use thereof |
| US11896656B2 (en) | 2018-04-30 | 2024-02-13 | Merck Sharp & Dohme Llc | Methods for providing a homogenous solution of lyophilized mutant diptheria toxin in dimethylsulfoxide |
| US11992521B2 (en) | 2018-04-30 | 2024-05-28 | Merck Sharp & Dohme Llc | Methods for producing Streptococcus pneumoniae capsular polysaccharide carrier protein conjugates |
| US12144855B2 (en) | 2018-04-30 | 2024-11-19 | Merck Sharp & Dohme Llc | Methods for producing streptococcus pneumoniae capsular polysaccharide carrier protein conjugates from lyospheres |
| US12419943B2 (en) | 2018-04-30 | 2025-09-23 | Merck Sharp & Dohme Llc | Methods for producing Streptococcus pneumoniae capsular polysaccharide carrier protein conjugates |
| US12016914B2 (en) | 2018-12-19 | 2024-06-25 | Merck Sharp & Dohme Llc | Compositions comprising Streptococcus pneumoniae polysaccharide-protein conjugates and methods of use thereof |
| US11642406B2 (en) | 2018-12-19 | 2023-05-09 | Merck Sharp & Dohme Llc | Compositions comprising Streptococcus pneumoniae polysaccharide-protein conjugates and methods of use thereof |
| US12383611B2 (en) | 2019-09-27 | 2025-08-12 | Pfizer Inc. | Neisseria meningitidis compositions and methods thereof |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7935787B2 (en) | Formulations which stabilize and inhibit precipitation of immunogenic compositions | |
| JP2009535353A5 (en) | ||
| AU2019202132B2 (en) | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions | |
| AU2022241552A1 (en) | Novel formulations which stabilize and inhibit precipitation of immunogenic compositions | |
| AU2012216628B2 (en) | Novel Formulations which Stabilize and Inhibit Precipitation of Immunogenic Compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780014734.3 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07760907 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2650056 Country of ref document: CA Ref document number: 2007760907 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 194753 Country of ref document: IL Ref document number: MX/A/2008/013572 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009507900 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12008502380 Country of ref document: PH |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: CR2008-010405 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008101753 Country of ref document: EG Ref document number: 572360 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007243019 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 9394/DELNP/2008 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15163062 Country of ref document: CO Ref document number: 08125423 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008141680 Country of ref document: RU |
|
| ENP | Entry into the national phase |
Ref document number: 2007243019 Country of ref document: AU Date of ref document: 20070419 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200870480 Country of ref document: EA |
|
| ENP | Entry into the national phase |
Ref document number: PI0710918 Country of ref document: BR Kind code of ref document: A2 Effective date: 20081024 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020137033305 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020147030833 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 239788 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 239978 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020167011184 Country of ref document: KR |