WO2005123762A2 - Codon-optimized hpv16 li for salmonella vaccine strains against human papillomavirus type 16 - Google Patents
Codon-optimized hpv16 li for salmonella vaccine strains against human papillomavirus type 16 Download PDFInfo
- Publication number
- WO2005123762A2 WO2005123762A2 PCT/IB2005/001725 IB2005001725W WO2005123762A2 WO 2005123762 A2 WO2005123762 A2 WO 2005123762A2 IB 2005001725 W IB2005001725 W IB 2005001725W WO 2005123762 A2 WO2005123762 A2 WO 2005123762A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- salmonella
- hpv16
- strain
- lis
- enterica serovar
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/005—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/12—Viral antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N7/00—Viruses; Bacteriophages; Compositions thereof; Preparation or purification thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/52—Bacterial cells; Fungal cells; Protozoal cells
- A61K2039/523—Bacterial cells; Fungal cells; Protozoal cells expressing foreign proteins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/525—Virus
- A61K2039/5258—Virus-like particles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/54—Medicinal preparations containing antigens or antibodies characterised by the route of administration
- A61K2039/541—Mucosal route
- A61K2039/542—Mucosal route oral/gastrointestinal
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/54—Medicinal preparations containing antigens or antibodies characterised by the route of administration
- A61K2039/541—Mucosal route
- A61K2039/543—Mucosal route intranasal
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2710/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA dsDNA viruses
- C12N2710/00011—Details
- C12N2710/20011—Papillomaviridae
- C12N2710/20022—New viral proteins or individual genes, new structural or functional aspects of known viral proteins or genes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2710/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA dsDNA viruses
- C12N2710/00011—Details
- C12N2710/20011—Papillomaviridae
- C12N2710/20023—Virus like particles [VLP]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2710/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA dsDNA viruses
- C12N2710/00011—Details
- C12N2710/20011—Papillomaviridae
- C12N2710/20034—Use of virus or viral component as vaccine, e.g. live-attenuated or inactivated virus, VLP, viral protein
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Definitions
- the present invention relates to a novel nucleic acid sequence (HPV16 LIS) encoding antigenic HPV16 LI protein as provided in SEQ ID NO: 1, wherein the said sequence has atleast one modified codon for optimum stability of recombinant plasmid vector when transformed in the prokaryotic micro-organism for improved immunogenicity of the resulting prokaryotic micro-organism.
- the invention also relates to an attenuated strain of a prokaryotic micro-organism transformed with nucleic acid encoding HPV16 (Human Papillomavirus) major capsid protein and expressing the corresponding protein.
- the invention discloses a process of producing a vaccine based on prokaryotic micro-organism for the treatment of papillomavirus infection and associated risk of cancer.
- BACKGROUND AND PRIOR ART REFERENCES Cervical cancer is the second leading cause of cancer deaths in women worldwide, and virtually all of these tumors are attributable to infection with a sub-set of human papillomaviruses (HPVs), of which HPV16 is found most frequently (6, 41). An effective vaccine against these HPVs would therefore be expected to have a dramatic impact on the incidence of this cancer and its precursor lesions, as well as on the less common tumors attributable to these viruses.
- HPVs human papillomaviruses
- the leading candidate is a prophylactic sub-unit HPV viruslike particle (VLP) vaccine (reviewed by (35) and (24)).
- HPV vaccines based on such VLPs and currently tested in clinical trials have proven to be well tolerated, highly immunogenic, and able to prevent the development of HPV16-induced cervical intraepithelial neoplasia (reviewed by [Schiller, 2004 #1431] and [Lowy, 2003 #1397]).
- these expensive vaccines require multiple intramuscular doses to be efficient and since most of cervical cancers occur in developing countries, such vaccines appear to be inaccessible for people needing them most. It is thus of great importance to develop other strategies that have world wide applicability.
- Live attenuated Salmonella strains may be effective antigen delivery systems, as they are able to express foreign antigens and to elicit mucosal as well as systemic immune responses against homologous and heterologous antigens after oral vaccination (reviewed in [Sch ⁇ del, 1992 #245; Curtiss, 1994 #466; Levine, 1997 #1416].
- Figure 1 shows Codon-optimized HPV 16 LIS orf.
- SEQ ID NO: l The nucleotide sequence of LIS is shown with the modified codons underlined and modified nucleotides in bold.
- Figure 2 shows HPV16L1 expression in PhoP LI and PhoP 0 LI S recombinant strains.
- Figure 3 shows LI and LIS-plasmid stability in vitro.
- Figure 4 shows Anti-HPV16 VLP systemic (A) and vaginal (B) antibody titers after nasal and oral vaccination with PhoP 0 LIS.
- Figure 5 shows Anti-HPV16 VLP systemic IgG titers after nasal or oral vaccination with %4989 LIS, x4990 LIS, PhoP" LIS and AroA LIS.
- Figure 6 is a comparison of serum anti-HPV16 VLP antibody titers after oral vaccination with PhoP c kan LIS, Phop " kan LIS and ⁇ Aro A kan LIS.
- Figure 7 shows in vitro stability of the kan LIS plasmid in different Salmonella enterica serovar Typhi strains.
- Figure 8 is comparison of semm anti-HPV16 VLP antibody titers after nasal vaccination with Ty21a kan LIS, Ty800 kan LIS and CVD908htrA kan LIS.
- Figure 9 is comparison of HPV16 VLP and Flagellin -specific CD4 + T cell proliferations.
- Figure 10 shows HPV16-neutralizing and anti-HPV16 VLP antibodies in serum and vaginal secretions of mice nasally vaccinated with Ty21a kan LIS.
- Figure 11 shows HPV 16 neutralizing and anti-HPV16 VLP antibodies in serum and vaginal secretions of mice nasally vaccinated with Ty21a kan LIS alone or primed with purified VLPs.
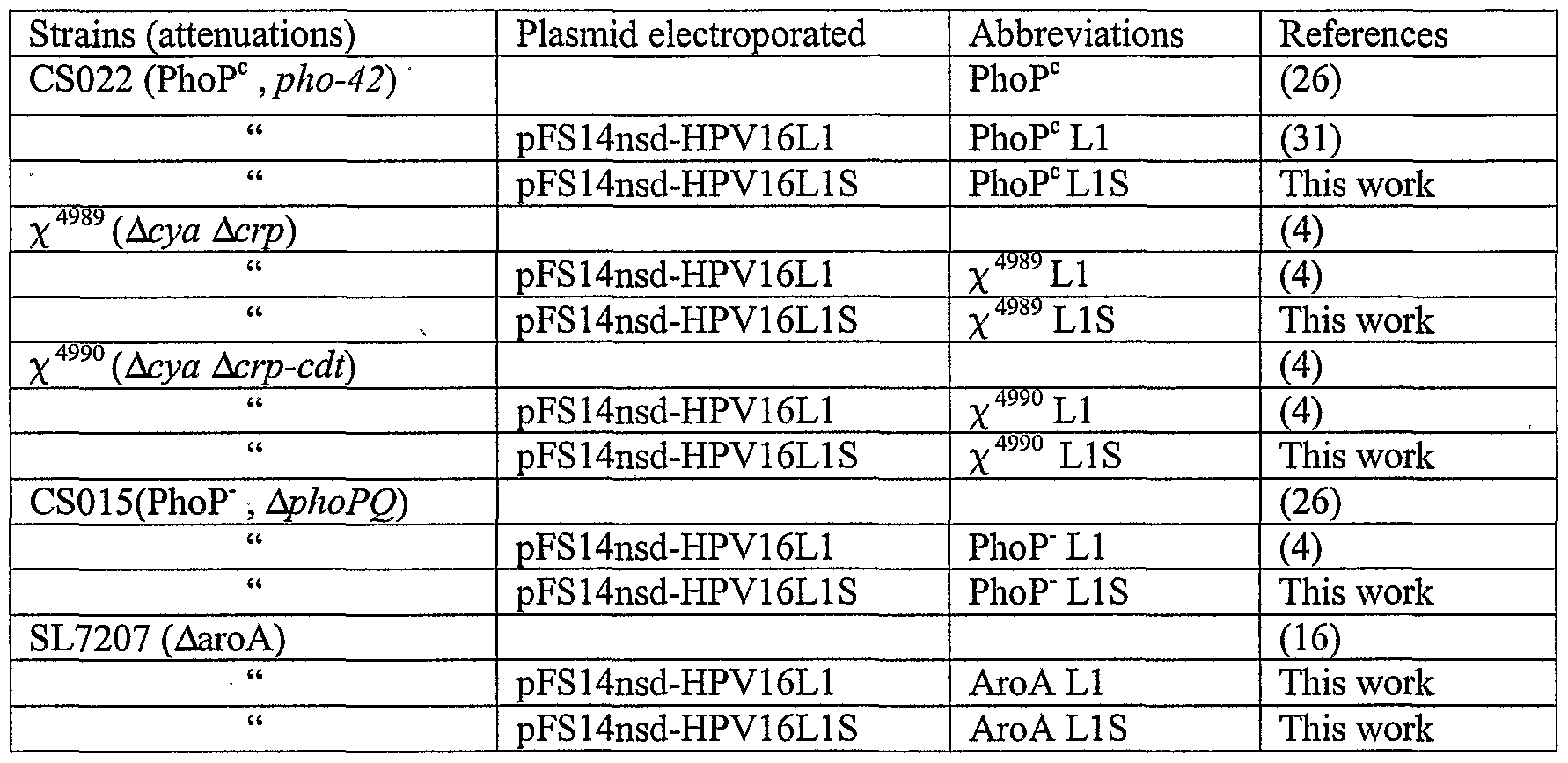
- TABLE 1 & 1A refers to Salmonella strains used in this study
- TABLE 2 refers to recovery of Salmonella PhoP 0 earring LI- or LlS-encoding plasmids two weeks after nasal or oral immunization
- TABLE 2A refers to recovery of Salmonella PhoP 0 LlS-encoding plasmids earring the ampiciline or the kanamycine resistant genes two weeks after oral immunization
- TABLE 3 refers to recovery of different Salmonella enterica serovar Typhi carrying kan LIS plasmids one week after nasal immunization.
- OBJECTS OF THE INVENTION The main object of the present invention is to provide a novel nucleic acid sequence
- HPV16 LIS encoding antigenic HPV16 LI protein as provided in SEQ ID NO: 1, wherein the said sequence has atleast one modified codon for optimum stability of recombinant plasmid vector when transformed in the prokaryotic micro-organism for improved immunogenicity of the resulting prokaryotic micro-organism.
- Another object of the present invention is to construct recombinant vectors P FS14nsdHPV16Ll and P FS14nsdHPV16 kan LIS harboring SEQ ID NO: 1, wherein the former carries Ampicillin and the latter, Kanamycin as a selection marker.
- Yet another object of the invention provides an attenuated strain of a prokaryotic micro-organism transformed with nucleic acid encoding HPV16 (Human Papillomavirus) major capsid protein and expressing the corresponding protein.
- One more object of the invention is to provide a process of producing a vaccine based on prokaryotic micro-organism for the treatment of papillomavirus infection and associated risk of cancer.
- the present invention provides a novel nucleic acid sequence (HPV 16 LIS) as provided in SEQ ID NO: 1, based on Human Papilloma Virus Type 16 (HPV 16) encoding antigenic HPV 16 LI protein, wherein the said sequence has atleast one modified codon for optimum stability of recombinant plasmid vector when transformed in the prokaryotic micro-organism for improved immunogenicity of the resulting prokaryotic micro-organism, preferably the strains belonging to Salmonella species.
- HPV 16 LIS novel nucleic acid sequence
- SEQ ID NO: 1 based on Human Papilloma Virus Type 16 (HPV 16) encoding antigenic HPV 16 LI protein
- the said sequence has atleast one modified codon for optimum stability of recombinant plasmid vector when transformed in the prokaryotic micro-organism for improved immunogenicity of the resulting prokaryotic micro-organism, preferably the strains belonging to Salmonella species.
- the invention further relates to constructing recombinant vectors pFS14nsdHPV16Ll and pFS14nsdHPV16 kan LIS harboring SEQ ID NO: 1, wherein the former carries Ampicillin and the latter, Kanamycin as a selection marker.
- the invention further provides an attenuated strain belonging to Salmonella species transformed with pFS14nsdHPV16Ll or pFS14nsdHPV16 kan LIS harboring SEQ ID NO: 1.
- the invention discloses a process for producing a Salmonella based vaccine for the treatment of papillomavirus infection and associated risk of cancer.
- LIS sequence is the improved stability of the LIS-expressing plasmid in vitro and in vivo in absence of antibiotic selection. This may contribute to the higher immunogenicity of the recombinant Salmonella as it results in a longer persistence of the VLP antigen carried by the bacteria.
- Such explanation is in agreement with the idea that a longer persistence of antigens in the mucosa-associated lymphoid tissues is a key mechanism that underlies the immune responses elicited by Salmonella vaccine strains (34) and contrasts with the other suggestion that the initial amount of antigen that prime the mucosal lymphoid tissue is the critical point for inducing efficient immune responses (8, 10).
- Different approaches have been used to improve plasmid stability in bacterial carriers (reviewed in (13, 25)).
- a single nasal immunization with PhoP 0 LIS induced similar serum and vaginal anti-HVP16 VLPs IgG titers than three sub-cutaneous injections with 1 ⁇ g purified HPV 16 VLPs, or three nasal/aerosol immunizations with 5 ⁇ g VLP doses together with the mucosal adjuvant cholera toxin, including induction of specific IgA in vaginal washes for the mucosal protocols (2, 29).
- nasal vaccination with recombinant Salmonella can be highly efficient at low doses and without concomitant lung inflammation (28), there are still safety concerns for using such a route of immunization in human.
- the kanamycin selectable marker will not give the organisms any selectable advantage outside the laboratory and this phenotype is already quite ubiquitous in nature.
- the widespread bacterial resistance to kanamycine has limited the use of this antibiotic in human medicine and the high substrate specificity of the enzyme warranty the absence of development of resistance or interference against modern antibiotic therapies.
- the presence of the kanamycin resistance gene has further improved the stability of the LIS expressing plasmid in Phop° in vivo, but the immune responses induced by oral immunization with the three attenuated Salmonella enterica serovar Typhimurium harboring the kan LIS plasmid were similar to those obtained with the bacteria harboring the ampicilin LIS plasmid [Baud, 2004 #1439].
- Salmonella enterica serovar Typhimurium PhoP- strain that was tested as a recombinant vaccine in human [Angelakopoulos, 2000 #1194]
- Salmonella-based vaccines are attenuated Salmonella enterica serovar Typhi strains that were first developed as vaccines against typho ⁇ d fever (reviewed in [Levine, 2001 #1428]).
- Attenuated Salmonella enterica serovar Typhi intranasally administered at high doses in mice elicit an array of immune responses similar to those observed in volunteers given the attenuated strains orally (reviewed in [Pasetti, 2003 #1404]).
- the kan LIS instability in CVC908 htrA and in Ty800 may relate to the low immunogenicity of these bacteria against HVP16 VLPs.
- Our data clearly show that only the recombinant Ty21a kan LIS is able to induce high anti-HPV16 VLP antibodies and VLP-specific CD4+ T cell responses in the nasal murine mode.
- both CVD908 htrA and Ty800 induced higher anti- LPS and anti-Flagellin antibodies, as well as flagellin-specific CD4T cells responses (for Ty800) than Ty21a. This finding is in agreement with previous studies were CVD908 htrA and/or Ty800 and Ty21a were compared in the murine nasal model [Wang, 2001 #1533 or in human.
- An embodiment of the present invention relates to synthesis of novel nucleic acid sequences based on HPV (Human Papilloma Virus) such as HPV 16, HPV 18, 45 31 etc., major capsid proteins by modifying their codons for optimum stability of the recombinant plasmid vector when transformed in the prokaryotic micro-organism for improved immunogenicity of the resulting prokaryotic micro-organism.
- HPV Human Papilloma Virus
- the prokaryotic micro- organisms are preferably selected from a group consisting of Salmonella sps, Escherichia coli, Shigella yersinia, Lactobacillus, Mycobacteria, or Lysteria, preferably Salmonella sps.
- the attenuated strain of Salmonella sps is selected from a group consisting of Salmonella enterica serovar Typhimurium, Salmonella typhi, Salmonella typhimurium or Salmonella dublin.
- the Salmonella strain is preferably selected from a group consisting of Salmonella enterica serovar Typhimurium PhoP (CS022), Salmonella enterica serovar Typhimurium PhoP " (CS015), Salmonella enterica serovar Typhimurium ⁇ 4989 ( ⁇ cya ⁇ crp-cdt and ⁇ aroA (SL7207)), Salmonella enterica serovar Typhi, Ty21a, CVD908 htrA, Ty800.
- Yet another embodiment of the present invention relates to a method for improving the immunogenicity of a prokaryotic micro-organism against Human Papillomavirus Type 16 (HPV16) comprising steps of: a. synthesizing a novel nucleic acid as shown in SEQ ID NO: 1 encoding antigenic HPV16 LI protein by modifying codon usage, b. constructing recombinant pFS14nsdHPV16LlS vector harboring SEQ ID No: 1 by replacing the original HPV16L1 gene in the plasmid vector pFS14nsdHPV16Ll or pFS14nsdHPV16 kan LIS, c.
- Another embodiment relates to an attenuated strain of a prokaryotic microorganism transformed with codon modified nucleic acid encoding HPV (Human Papillomavirus) major capsid protein and expressing the corresponding protein, wherein atleast one codon is modified.
- HPV Human Papillomavirus
- the HPV major capsid protein is selected from a group consisting of HPV 16, HPV18, HPV 31 and HPV 45 preferably HPV 16 LI.
- the above said strain harbors a novel nucleic acid sequence SEQ ID NO: 1.
- Yet another embodiment of the present invention relates to a novel nucleic acid having one or more modified codons for optimum stability of recombinant plasmid vector in Salmonella strains for improved immunogenicity of the resulting Salmonella strain.
- Yet another embodiment of the present invention provides a recombinant plasmid vector containing DNA sequences as shown in SEQ ID NO: 1 wherein the said recombinant plasmid vector is either pFS14nsd-HPV16Ll S or pFSnsd-HPV16 kan LIS.
- Another embodiment of the present invention relates to a mode of administration of the Salmonella based vaccine against HPV 16 which may be selected from group consisting of oral, intra-nasal, vaginal or rectal.
- One more embodiment of the invention provides the use of attenuated strain of a prokaryotic microorganism in the preparation of a medicament for the prophylactic or therapeutic treatment of papillomavirus infection and associated risk of cancer.
- Preferred embodiments of the present invention are now described by way of example and not limitation with reference to the accompanying drawings. Embodiments of the invention EXAMPLES Example 1
- the resulting plasmid, pFS14nsd HPV16-L1S was introduced by electroporation (37) into the attenuated Salmonella enterica serovar Typhimurium strains PhoP 0 , (CS022 (27)) and PhoP" (CSO 15(26)), both a kind gift from John Mekalanos, Boston, USA, x 4 989 ⁇ Acya Acrp, (4)), x4990 (Acya Acrp-cdt, (4)) and AaroA (SL7207 (16)), a kind gift from Irene Corthesy- Theulaz, Lausanne, CH.
- HPV16 LI and VLP analysis Expression of LI in Salmonella ly sates was analyzed by Western blot as previously described 15 (31) using the anti-HPV16 LI mAb, CAMVIR-1 (Anawa). Data were normalized to the content in bacteria as measured by the OD600 of the cultures.
- the HPV16 VLP content was measured by a sandwich ELISA as previsouly described (4) using two monoclonal antibodies that recognize conformational epitopes on HPV16 VLPs, H16E70 and HI 6 V5, (9).
- 20 Immunization of mice, analysis of the immune response and recovery of S. typhimurium Six-week old female BALB/c mice from Iffa Credo, France were used in all experiments.
- Tewnty JLLl of bacterial inoculum were administered orally (10 8 ⁇ 9 CFU) or intranasally (10 67 CFU) under anesthesia as previously described (17, 31).
- Recovery of Salmonella enterica serovar Typhimurium was determined in organs from euthanized mice as previously described (31).
- the LI orf was then replaced in plasmid pFS14nsd-HPV16 LI (31) by the new LIS orf, yielding pFS 14nsd-HPVl 6 LIS.
- the new plasmid was first introduced in the attenuated Salmonella enterica serovar Typhimurium strain PhoP 0 (27) to generate the recombinant strain called PhoP 0 LIS hereafter.
- PhoP 0 LIS the recombinant attenuated Salmonella strains were subsequently produced (see here below) and Table 1 summarizes the different strains and abbreviations used in this study.
- the stability of the LlS-encoding plasmid was further examined in vivo after nasal and oral immunization of mice (see Table 2).
- the LIS encoding plasmid was completely stable for at least two weeks in the organs close to the sites of infection/entry.
- Some instability of the LIS plasmid was, however, observed in more distant organs such as the spleen, where ca. 10 % of the bacteria were still harboring the LIS plasmid but none were detected harboring the LI plasmid.
- PhoP 0 LIS strain The anti-VLP antibody titers measured in serum and vaginal secretions of the mice 4 to 6, 8 and 24 weeks after a single immunization are shown in Fig. 4.
- These antibody titers are similar to those induced after a double nasal vaccination with the original PhoP 0 LI strain, but a major improvement is that they are one to two orders of magnitude higher than those achieved with a single nasal vaccination with the original PhoP 0 LI strain (4, 31).
- PhoP 0 LIS oral vaccination with PhoP 0 LIS was also highly immunogenic, although less than nasal vaccination, while even a double oral vaccination with the original PhoP 0 strain was inefficient (31).
- Administration of two nasal or three oral doses of PhoP 0 LIS did not increase the immune responses.
- Anti-HPV16 VLP systemic antibodies following nasal or oral vaccination with differently attenuated Salmonella enterica serovar Typhimurium strains expressing the HPV16 LIS gene Inventor have previously shown that nasal vaccination of mice with differently attenuated Salmonella enterica serovar Typhimurium strains expressing the original LI encoding plasmid induced only low or no anti-HPV16 VLPs antibodies (4). Given the high immunogenicity observed with the PhoP 0 strain expressing the LIS encoding plasmid, we further introduced this plasmid in different strains including %4989, ⁇ 4990, PhoP" and AroA (see Table 1 for precise attenuations, abbreviations and references).
- the anti-HVP16 VLP IgG titers measured in mice 7 weeks after a single nasal or oral vaccination with these new recombinant strains are shown in Fig. 5.
- all the new recombinant strains induced consistent anti-HPV16 VLP humoral responses after a single nasal vaccination; although the titers are about one order of magnitude lower than those achieved with the PhoP 0 LIS strain (see Fig. 4).
- oral vaccination was less immunogenic, with the exception of the AroA LIS strain, which induced similar anti-HPVl 6 VLP IgG titers after both routes of vaccination.
- the primers used were a 25-mer located 54 nucleotides upstream form the first ATG of kanamycin and containing a SacII restriction site (underlined): 5'- GGGCCGCGGTGGTC ATGAAC AATAA-3 ' .
- the new plasmid was introduced by electroporation [Sch ⁇ del, 1990b #242] into the attenuated Salmonella enterica serovar Typhimurium strains PhoP 0 , (CS022 [Miller, 1990 #181]) and PhoP " (CSO 15 [Miller, 1989 #178]), and ⁇ aroA (SL7207 [Hoiseth, 1981 #123]), as well as into the attenuated Salmonella enterica serovar Typhi Ty800 [Hohmann, 1996 #596] CVD908htrA [Tacket, 1997 #643], and Ty21a ([Germanier, 1975 #103], Berna biotech, Switzerland).
- HPV16 LI and VLP analysis Expression of LI in Salmonella lysates was analyzed by Western blot as previously described [Nardellihaefliger, 1997 #742] using the anti-HPV16 LI mAb, CAMVIR-1 (Anawa). Data were normalized to the content in bacteria.
- the HPV16 VLP content was measured by a sandwich ELISA as previously described [Benyacoub, 1999 #1050] using two monoclonal antibodies that recognize conformational epitopes on HPV16 VLPs, H16E70 or H161A and H16 V5, [Christensen, 1996 #1053].
- mice Immunization of mice, analysis of anti-HPV16 VLP antibodies and recovery of Salmonella Six-week old female BALB/c mice from Iffa Credo, France were used in all experiments. Tewnty ⁇ l of bacterial inoculum were administered orally (10 9 CFU) or intranasally (10 7"9 CFU) under anesthesia as previously described [Hopkins, 1995 #381; Nardellihaefliger, 1997 #742]. Sampling of blood and vaginal washes as well as determination of anti-HPVl 6 VLP antibody titers by ELISA were performed as reported earlier [Hopkins, 1995 #381; Nardellihaefliger, 1997 #742]. Recovery of Salmonella enterica serovar Typhimurium or Typhi was determined in organs from euthanized mice as previously described [Nardellihaefliger, 1997 #742].
- Neutralization assays were performed with secreted alkaline phosphatase (SEAP) HPV16 pseudoviruses as described in detail in [Pastrana DV, 2004 #1429]. Briefly, optiprep-purified SEAP HPV 16 pseudoviruses diluted 2000-fold were incubated on ice for 1 h with two-fold serial serum dilutions, and the pseudorivus-antibody mixtures were used to infect 293TT cells for 3 days. The SEAP content in lO ⁇ l of clarified cell supernatant was determined using the Great ESCAPE SEAP Chemiluminescence Kit (BD Clontech). Neutralization titers were defined as the reciprocal of the highest serum dilution that caused at least a 50% reduction in SEAP activity (100% SEAP activity ranging from 50 to 100 relative light units).
- CD4 + T cells were purified by magnetic antibody cell sorting
- CD4 T cells form na ⁇ ve or immunized mice were incubated for three days with medium alone or Concavalin A (2.5 ⁇ g/ml, ) as negative and positive controls as well as with HPV16 VLPs (2 ⁇ g/ml, GMP preparation) and Flagellin (lO ⁇ g/ml, purified from Ty21a) then 0.5 ⁇ Curie of 3H thimidine was added over night and incorporation was measured in cpm.
- HPV16L1S and plasmid stability in a kanamycin resistant PhoP c strain A kanamycin resistant plamid expressing HPV 16 LIS was constructed by replacing the ampicillin resistant gene in the original pFSnsdHPVl ⁇ LIS [Baud, 2004 #1439] by a kanamycin selectable gene. An inverse PCR strategy was used to amplify the entire plasmid flanking the ampicillin resistant gene sequence and the resulting fragment was ligated to a kanamycine resistance encoding sequence (see material and method for details).
- the resulting plasmid was designated pFSnsd-kan LIS and electroporated in PhoP 0 to yield PhoP°-kanLlS (see Table 1A) for abbreviations and references of the strains used in this study).
- PhoP°-LlS ca 10 ⁇ gVLPs/10 u CFU, [Baud, 2004 #1439].
- Similar growth rates were also observed between the two strains, with about 7 hours to reach mid-log phase and plasmid stability I was very high with almost 100% of the bacteria still harboring the plasmid after four consecutive over-night cultures in absence of antibiotic.
- the stability of the kan LIS plasmid was increased in vivo, as compared to the ampicillin plasmid, with all the bacteria harborig the kan LIS plasmid two weeks after oral immunization and this, in all organs examined (see Table 2A).
- mice Three groups of 5 to 10 mice were immunized once by the oral route with the three recombinant Salmonella enterica serovar Typhimurium strains and the HPV16 VLPs specific antibody responses in serum and vaginal washes are compared at 6 weeks post-immunization (see Fig. 6).
- the anti-VLP antibody titers induced by the AroA kan LIS strain appear as high as those induced by PhoP 0 kan LIS and by the former PhoP 0 LIS strain [Baud, 2004 #1439]. These antibody titers were stable for at least four months (data not shown).
- kan LIS plasmid stability in the three vaccine strains in absence of antibiotics was first examined in vitro (see Fig.7).
- a lower stability of the kan LIS plasmid was observed in the vaccine strains.
- a slow loss of plasmid occurred after the second over-night, but after five consecutive overnights, the plasmid was still retained in 9, 7 and 18 % of the bacteria in Ty21a kan LIS, Ty800 kan LIS and CVD908-htrA kan LIS, respectively.
- Salmonella enterica serovar Typhz vaccines strains will only undergo limited rounds or replication in human and they are not invasive by the oral route in mice.
- mice can transiently be infected if the bacteria are administered at high doses by the nasal route as shown with recombinant CVD908htrA [Pickett, 2000 #1158] [Pasetti, 2000 #1494].
- CVD908htrA Panet, 2000 #1158
- Pasetti, 2000 #1494 We have thus examined the stability of the kan LIS plasmid in Salmonella recovered from the lung and spleen of mice one week after intranasal immunization with 10 9 CFU of CVD908-htrA kan LIS and Ty21a kan LIS or 10 7 CFU of Ty800 kan LIS.
- mice Eight weeks after immunization, the mice were killed and antigen specific proliferation was measured with CD4 T cells purified from the spleen (see Fig.9).
- HPV16 VLP stimulation of CD4 + T cells was only significantly induced after two intranasal vaccination with Ty21a kan LIS (p ⁇ 0.001) in agreement with the induction of anti- HPV16 VLPs titers.
- a significant flagellin specific stimulation of CD4 + T cells was only measured after vaccination with Ty800 kan LI S (p ⁇ 0.001).
- HPV16-neutralizing titers are induced in serum and genital secretions of mice immunized with Ty21a kan LIS alone or primed with a subcutaneous VLP dose.
- mice received two nasal doses of Ty21 kan LIS (ca.lO 9 CFU) at week 0 and 4 and sampling of blood and vaginal secretions was performed at week 8.
- Anti-HPV16 VLP and HPV16-neutralizing antibody titers were determined by ELISA and the SEAP HPV 16 pseudovirion neutralization assay, respectively, in both serum and vaginal secretions (see Fig 10 A).
- the HPV16-neutralizing titers were slightly lower than the anti-VLP ELISA titers [Baud, 2004 #1439].
- a b means (log, 0 ) of CFU/organ ⁇ SEM; data after nasal immunization with PhoP c LI are taken from [Benyacoub, 1999#1050].; c Not detectable
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Virology (AREA)
- Animal Behavior & Ethology (AREA)
- Genetics & Genomics (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Immunology (AREA)
- Wood Science & Technology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biochemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Zoology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Microbiology (AREA)
- Biophysics (AREA)
- Biotechnology (AREA)
- Biomedical Technology (AREA)
- Oncology (AREA)
- General Engineering & Computer Science (AREA)
- Gastroenterology & Hepatology (AREA)
- Communicable Diseases (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Mycology (AREA)
- Epidemiology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2005800278872A CN101115766B (en) | 2004-06-18 | 2005-06-20 | Codon-optimized HPV16LI for salmonella vaccine strains against human papillomavirus type 16 |
| JP2007516066A JP4769247B2 (en) | 2004-06-18 | 2005-06-20 | Codon optimized HPV16L1 for Salmonella vaccine strain against human papillomavirus type 16 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH2004000373 | 2004-06-18 | ||
| CHPCT/CH2004/000373 | 2004-06-18 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO2005123762A2 true WO2005123762A2 (en) | 2005-12-29 |
| WO2005123762A3 WO2005123762A3 (en) | 2006-04-27 |
| WO2005123762B1 WO2005123762B1 (en) | 2006-06-08 |
Family
ID=35510322
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2005/001725 WO2005123762A2 (en) | 2004-06-18 | 2005-06-20 | Codon-optimized hpv16 li for salmonella vaccine strains against human papillomavirus type 16 |
Country Status (3)
| Country | Link |
|---|---|
| JP (1) | JP4769247B2 (en) |
| CN (1) | CN101115766B (en) |
| WO (1) | WO2005123762A2 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100532548C (en) * | 2007-02-14 | 2009-08-26 | 马润林 | Method of increasing yield of human papilloma virus L1 albumen pronucleus expression |
| JP2010500013A (en) * | 2006-08-10 | 2010-01-07 | インターナショナル インベストメント アンド パテンツ ソシエテ アノニム | Plasmid with immunological action |
| EP2801364A1 (en) * | 2013-05-07 | 2014-11-12 | Centre Hospitalier Universitaire Vaudois (CHUV) | Salmonella strain for use in the treatment and/or prevention of cancer |
| RU2628693C1 (en) * | 2016-11-10 | 2017-08-21 | федеральное государственное бюджетное учреждение "Федеральный научно-исследовательский центр эпидемиологии и микробиологии имени почетного академика Н.Ф. Гамалеи" Министерства здравоохранения Российской Федерации (ФГБУ "ФНИЦЭМ им. Н.Ф. Гамалеи" Минздрава России) | Recombinant l1hpv16 gene, recombinant pqe-l1/16 plasmid, l1hpv16 protein and their application |
| CN113528544A (en) * | 2021-06-02 | 2021-10-22 | 郑州大学 | Gene for coding soluble HPV23L1 protein and construction and application of recombinant plasmid thereof |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102178944A (en) * | 2010-09-17 | 2011-09-14 | 大连雅立峰生物制药有限公司 | Expression and application of human papilloma viruses type 16 and 18 L1 proteins in inset cells |
| LT2988762T (en) * | 2013-04-25 | 2018-09-25 | Vaximm Ag | Salmonella-based vectors for cancer immunotherapy targeting wilms` tumor gene wt1 |
| CN112280793B (en) * | 2013-09-29 | 2022-06-24 | 上海泽润生物科技有限公司 | Human papilloma virus gene, vector, strain and expression method |
| MA40624A (en) * | 2014-09-11 | 2016-03-17 | Cadila Healthcare Ltd | Superior human papilloma virus antigens with superior immunological properties and vaccine containing it |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020025328A1 (en) * | 1996-10-09 | 2002-02-28 | Denise Nardelli Haefliger | Attenuated microorganism strains expressing hpv proteins |
| WO2004084831A2 (en) * | 2003-03-24 | 2004-10-07 | Merck & Co. Inc. | Optimized expression of hpv 31 l1 in yeast |
-
2005
- 2005-06-20 CN CN2005800278872A patent/CN101115766B/en not_active Expired - Fee Related
- 2005-06-20 JP JP2007516066A patent/JP4769247B2/en not_active Expired - Fee Related
- 2005-06-20 WO PCT/IB2005/001725 patent/WO2005123762A2/en active Search and Examination
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020025328A1 (en) * | 1996-10-09 | 2002-02-28 | Denise Nardelli Haefliger | Attenuated microorganism strains expressing hpv proteins |
| WO2004084831A2 (en) * | 2003-03-24 | 2004-10-07 | Merck & Co. Inc. | Optimized expression of hpv 31 l1 in yeast |
Non-Patent Citations (5)
| Title |
|---|
| BAUD DAVID ET AL: "Immunogenicity against human papillomavirus type 16 virus-like particles is strongly enhanced by the PhoPc phenotype in Salmonella enterica serovar typhimurium." INFECTION AND IMMUNITY, vol. 72, no. 2, February 2004 (2004-02), pages 750-756, XP002364716 ISSN: 0019-9567 cited in the application * |
| BAUD DAVID ET AL: "Improved efficiency of a Salmonella-based vaccine against human papillomavirus type 16 virus-like particles achieved by using a codon-optimized version of L1" JOURNAL OF VIROLOGY, vol. 78, no. 23, December 2004 (2004-12), pages 12901-12909, XP002364715 ISSN: 0022-538X * |
| LEDER C ET AL: "Enhancement of capsid gene expression: preparing the human papillomavirus type 16 major structural gene L1 for DNA vaccination purposes" JOURNAL OF VIROLOGY, THE AMERICAN SOCIETY FOR MICROBIOLOGY, US, vol. 75, no. 19, October 2001 (2001-10), pages 9201-9209, XP002202893 ISSN: 0022-538X cited in the application * |
| SEEGERS J F M L: "Lactobacilli as live vaccine delivery vectors: progress and prospects" TRENDS IN BIOTECHNOLOGY, ELSEVIER PUBLICATIONS, CAMBRIDGE, GB, vol. 20, no. 12, 1 December 2002 (2002-12-01), pages 508-515, XP004393459 ISSN: 0167-7799 * |
| WYSZYNSKA A ET AL: "Oral immunization of chickens with avirulent Salmonella vaccine strain carrying C. jejuni 72Dz/92 cjaA gene elicits specific humoral immune response associated with protection against challenge with wild-type Campylobacter" VACCINE, BUTTERWORTH SCIENTIFIC. GUILDFORD, GB, vol. 22, no. 11-12, 29 March 2004 (2004-03-29), pages 1379-1389, XP004500381 ISSN: 0264-410X * |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010500013A (en) * | 2006-08-10 | 2010-01-07 | インターナショナル インベストメント アンド パテンツ ソシエテ アノニム | Plasmid with immunological action |
| US8691959B2 (en) | 2006-08-10 | 2014-04-08 | Cosmo Bio-Technologies Srl | Plasmids with immunological action |
| CN100532548C (en) * | 2007-02-14 | 2009-08-26 | 马润林 | Method of increasing yield of human papilloma virus L1 albumen pronucleus expression |
| EP2801364A1 (en) * | 2013-05-07 | 2014-11-12 | Centre Hospitalier Universitaire Vaudois (CHUV) | Salmonella strain for use in the treatment and/or prevention of cancer |
| US9795641B2 (en) | 2013-05-07 | 2017-10-24 | Centre Hospitalier Universitaire Vaudois (C.H.U.V.) | Salmonella strains for use in the treatment and/or prevention of cancer |
| RU2628693C1 (en) * | 2016-11-10 | 2017-08-21 | федеральное государственное бюджетное учреждение "Федеральный научно-исследовательский центр эпидемиологии и микробиологии имени почетного академика Н.Ф. Гамалеи" Министерства здравоохранения Российской Федерации (ФГБУ "ФНИЦЭМ им. Н.Ф. Гамалеи" Минздрава России) | Recombinant l1hpv16 gene, recombinant pqe-l1/16 plasmid, l1hpv16 protein and their application |
| CN113528544A (en) * | 2021-06-02 | 2021-10-22 | 郑州大学 | Gene for coding soluble HPV23L1 protein and construction and application of recombinant plasmid thereof |
| CN113528544B (en) * | 2021-06-02 | 2022-07-08 | 郑州大学 | Gene for coding soluble HPV23L1 protein and construction and application of recombinant plasmid thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2005123762A3 (en) | 2006-04-27 |
| WO2005123762B1 (en) | 2006-06-08 |
| CN101115766A (en) | 2008-01-30 |
| JP2008504020A (en) | 2008-02-14 |
| CN101115766B (en) | 2013-05-08 |
| JP4769247B2 (en) | 2011-09-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6458368B1 (en) | Attenuated microorganism strains expressing HPV proteins | |
| WO2005123762A2 (en) | Codon-optimized hpv16 li for salmonella vaccine strains against human papillomavirus type 16 | |
| Stanley et al. | Immunobiology of human papillomavirus infection and vaccination-implications for second generation vaccines | |
| Baud et al. | Improved efficiency of a Salmonella-based vaccine against human papillomavirus type 16 virus-like particles achieved by using a codon-optimized version of L1 | |
| EP2802349B1 (en) | Immunogenic hpv l2-containing vlps and related compositions and methods | |
| Revaz et al. | Mucosal vaccination with a recombinant Salmonella typhimurium expressing human papillomavirus type 16 (HPV16) L1 virus-like particles (VLPs) or HPV16 VLPs purified from insect cells inhibits the growth of HPV16-expressing tumor cells in mice | |
| Fraillery et al. | Salmonella enterica serovar Typhi Ty21a expressing human papillomavirus type 16 L1 as a potential live vaccine against cervical cancer and typhoid fever | |
| CN110156896B (en) | Recombinant foot-and-mouth disease virus-like particle and preparation method and application thereof | |
| Schiller et al. | Second generation HPV vaccines to prevent cervical cancer | |
| Palmer et al. | Recombinant vaccines for the prevention of human papillomavirus infection and cervical cancer | |
| MX2011001429A (en) | Veterinary pharmaceutical formulation comprising an rna recombinant particle encoding a cu/zn superoxide dismutase protein of ruminant pathogenic bacteria and at least one rna alphavirus belonging to the semliki forest virus family. | |
| US8282936B2 (en) | Human papillomavirus vaccine for oral administration | |
| US9533057B2 (en) | Immunogenic HPV L2-containing VLPs and related compositions, constructs, and therapeutic methods | |
| US9717783B2 (en) | Immunogenic HPV L2-containing VLPs and related compositions, constructs, and therapeutic methods | |
| Mustafa et al. | Listeria monocytogenes delivery of HPV-16 major capsid protein L1 induces systemic and mucosal cell-mediated CD4+ and CD8+ T-cell responses after oral immunization | |
| JP5930716B2 (en) | Method for oral / transmucosal vaccination with recombinant yeast | |
| Curtiss 3rd | Antigen delivery system II: development of live attenuated bacterial vectors | |
| Baud et al. | Improved Efficiency of | |
| Zhai | A broadly protective thermostable next generation HPV vaccine based on a concatemer peptide and a consensus peptide of L2 displayed on bacteriophage virus-like particles | |
| MXPA99003286A (en) | Attenuated microorganism strains expressing hpv proteins |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KM KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NG NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| B | Later publication of amended claims |
Effective date: 20060411 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 12006502512 Country of ref document: PH |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007516066 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 131/CHENP/2007 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580027887.2 Country of ref document: CN |
|
| 122 | Ep: pct application non-entry in european phase | ||
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) |