WO2005019147A2 - Pesticidally active phenol derivatives - Google Patents
Pesticidally active phenol derivatives Download PDFInfo
- Publication number
- WO2005019147A2 WO2005019147A2 PCT/EP2004/009500 EP2004009500W WO2005019147A2 WO 2005019147 A2 WO2005019147 A2 WO 2005019147A2 EP 2004009500 W EP2004009500 W EP 2004009500W WO 2005019147 A2 WO2005019147 A2 WO 2005019147A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- spp
- alkyl
- crc
- formula
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 N**NCC(O)=O Chemical compound N**NCC(O)=O 0.000 description 3
- YGEAAKJVLAZTDG-UHFFFAOYSA-N CCC(COc(cc1C)n[n]1-c1cc([Br]=C)ccc1)=C Chemical compound CCC(COc(cc1C)n[n]1-c1cc([Br]=C)ccc1)=C YGEAAKJVLAZTDG-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/18—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof
- A01N37/20—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof containing the group, wherein Cn means a carbon skeleton not containing a ring; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N31/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic oxygen or sulfur compounds
- A01N31/08—Oxygen or sulfur directly attached to an aromatic ring system
- A01N31/16—Oxygen or sulfur directly attached to an aromatic ring system with two or more oxygen or sulfur atoms directly attached to the same aromatic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N35/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having two bonds to hetero atoms with at the most one bond to halogen, e.g. aldehyde radical
- A01N35/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having two bonds to hetero atoms with at the most one bond to halogen, e.g. aldehyde radical at least one of the bonds to hetero atoms is to nitrogen
- A01N35/10—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having two bonds to hetero atoms with at the most one bond to halogen, e.g. aldehyde radical at least one of the bonds to hetero atoms is to nitrogen containing a carbon-to-nitrogen double bond
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/04—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom
- A01N43/06—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings
- A01N43/10—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings with sulfur as the ring hetero atom
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/54—1,3-Diazines; Hydrogenated 1,3-diazines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/56—1,2-Diazoles; Hydrogenated 1,2-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
- A01N43/653—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/713—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with four or more nitrogen atoms as the only ring hetero atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/80—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,2
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/82—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with three ring hetero atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/84—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms six-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C205/00—Compounds containing nitro groups bound to a carbon skeleton
- C07C205/27—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by etherified hydroxy groups
- C07C205/35—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by etherified hydroxy groups having nitro groups and etherified hydroxy groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C205/36—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by etherified hydroxy groups having nitro groups and etherified hydroxy groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton to carbon atoms of the same non-condensed six-membered aromatic ring or to carbon atoms of six-membered aromatic rings being part of the same condensed ring system
- C07C205/37—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by etherified hydroxy groups having nitro groups and etherified hydroxy groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton to carbon atoms of the same non-condensed six-membered aromatic ring or to carbon atoms of six-membered aromatic rings being part of the same condensed ring system the oxygen atom of at least one of the etherified hydroxy groups being further bound to an acyclic carbon atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C233/00—Carboxylic acid amides
- C07C233/01—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms
- C07C233/16—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms
- C07C233/17—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms with the substituted hydrocarbon radical bound to the nitrogen atom of the carboxamide group by an acyclic carbon atom
- C07C233/22—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms with the substituted hydrocarbon radical bound to the nitrogen atom of the carboxamide group by an acyclic carbon atom having the carbon atom of the carboxamide group bound to an acyclic carbon atom of a carbon skeleton containing six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C251/00—Compounds containing nitrogen atoms doubly-bound to a carbon skeleton
- C07C251/32—Oximes
- C07C251/34—Oximes with oxygen atoms of oxyimino groups bound to hydrogen atoms or to carbon atoms of unsubstituted hydrocarbon radicals
- C07C251/48—Oximes with oxygen atoms of oxyimino groups bound to hydrogen atoms or to carbon atoms of unsubstituted hydrocarbon radicals with the carbon atom of at least one of the oxyimino groups bound to a carbon atom of a six-membered aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/49—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C255/54—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton containing cyano groups and etherified hydroxy groups bound to the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C43/00—Ethers; Compounds having groups, groups or groups
- C07C43/02—Ethers
- C07C43/20—Ethers having an ether-oxygen atom bound to a carbon atom of a six-membered aromatic ring
- C07C43/225—Ethers having an ether-oxygen atom bound to a carbon atom of a six-membered aromatic ring containing halogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C49/00—Ketones; Ketenes; Dimeric ketenes; Ketonic chelates
- C07C49/20—Unsaturated compounds containing keto groups bound to acyclic carbon atoms
- C07C49/255—Unsaturated compounds containing keto groups bound to acyclic carbon atoms containing ether groups, groups, groups, or groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C49/00—Ketones; Ketenes; Dimeric ketenes; Ketonic chelates
- C07C49/76—Ketones containing a keto group bound to a six-membered aromatic ring
- C07C49/84—Ketones containing a keto group bound to a six-membered aromatic ring containing ether groups, groups, groups, or groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/62—Halogen-containing esters
- C07C69/65—Halogen-containing esters of unsaturated acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/44—Radicals substituted by doubly-bound oxygen, sulfur, or nitrogen atoms, or by two such atoms singly-bound to the same carbon atom
- C07D213/53—Nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/62—Oxygen or sulfur atoms
- C07D213/63—One oxygen atom
- C07D213/64—One oxygen atom attached in position 2 or 6
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/14—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D231/18—One oxygen or sulfur atom
- C07D231/20—One oxygen atom attached in position 3 or 5
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/32—One oxygen, sulfur or nitrogen atom
- C07D239/38—One sulfur atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
- C07D249/10—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D249/12—Oxygen or sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D257/00—Heterocyclic compounds containing rings having four nitrogen atoms as the only ring hetero atoms
- C07D257/02—Heterocyclic compounds containing rings having four nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D257/04—Five-membered rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D261/00—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings
- C07D261/02—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings
- C07D261/06—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having two or more double bonds between ring members or between ring members and non-ring members
- C07D261/10—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having two or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D261/18—Carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D271/00—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms
- C07D271/02—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms not condensed with other rings
- C07D271/06—1,2,4-Oxadiazoles; Hydrogenated 1,2,4-oxadiazoles
- C07D271/07—1,2,4-Oxadiazoles; Hydrogenated 1,2,4-oxadiazoles with oxygen, sulfur or nitrogen atoms, directly attached to ring carbon atoms, the nitrogen atoms not forming part of a nitro radical
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/04—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms
- C07D295/08—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms
- C07D295/096—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms with the ring nitrogen atoms and the oxygen or sulfur atoms separated by carbocyclic rings or by carbon chains interrupted by carbocyclic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/06—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring carbon atoms
- C07D333/22—Radicals substituted by doubly bound hetero atoms, or by two hetero atoms other than halogen singly bound to the same carbon atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/081—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te
Definitions
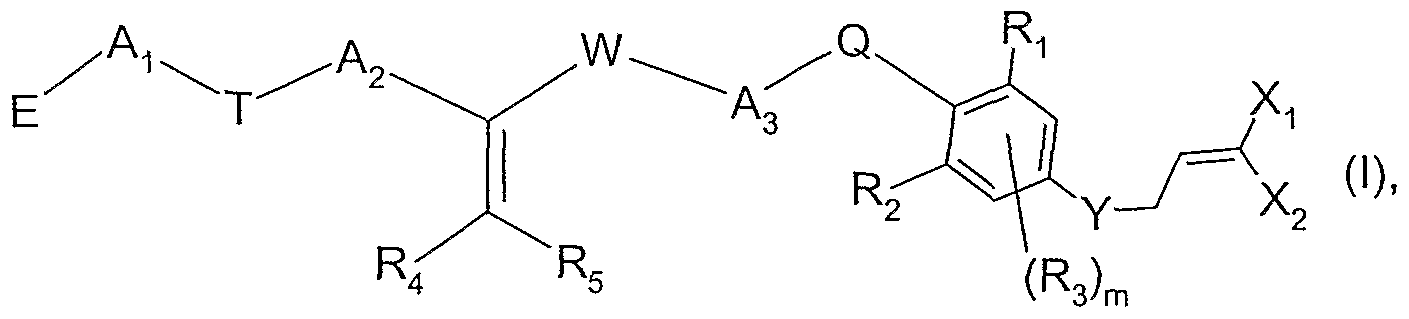
- the present invention relates (1) to a compound of formula
- X T and X 2 are each independently of the other fluorine, chlorine or bromine;
- a T and A 2 are each independently of the other a bond or a CrC 6 alkylene bridge which is unsubstituted or substituted by from one to six identical or different substituents selected from halogen and C 3 -C 8 cycloalkyl;
- a 3 is a d-C ⁇ alkylene bridge which is unsubstituted or substituted by from one to six identical or different substituents selected from halogen and C 3 -C 8 cycloalkyl;
- R-i and R 2 are each independently of the other halogen, OH, SH, CN, nitro, C-,-C 6 alkyl, d-Cehaloalkyl, C-rC 6 alkyl-carbonyl, C 2 -C 6 alkenyl, C 2 -C 6 haloalkenyl, C 2 -C 6 alkynyl, Ci-Cealkoxy
- Certain dihaloallyl derivatives are proposed in the literature as active ingredients in pesticidal compositions.
- the biological properties of those known compounds are not entirely satisfactory in the field of pest control, however, for which reason there is a need to provide further compounds having pesticidal properties, especially for controlling insects and members of the order Acarina, that problem surprisingly being solved by the provision of the present compounds of formula (I).
- the compounds of formula (I) and, where applicable, their tautomers are able to form salts, e.g. acid addition salts.
- the latter are formed, for example, with strong inorganic acids, such as mineral acids, e.g.
- organic carboxylic acids such as unsubstituted or substituted, e.g. halo-substituted, d-C 4 alkanecarboxylic acids, for example acetic acid, saturated or unsaturated dicarboxylic acids, e.g. oxalic, malonic, maleic, fumaric or phthalic acid, hydroxycarboxylic acids, e.g. ascorbic, lactic, malic, tartaric or citric acid, or benzoic acid, or with organic sulfonic acids, such as unsubstituted or substituted, e.g.
- salts with bases are, for example, metal salts, such as alkali metal or alkaline earth metal salts, e.g. sodium, potassium or magnesium salts, or salts with ammonia or an organic amine, such as morph- oline, piperidine, pyrrolidine, a mono-, di- or tri-lower alkylamine, e.g.
- Halogen as a group per se and as a structural element of other groups and compounds, such as of haloalkyl, halocycloalkyl, haloalkenyi, haloalkynyl and haloalkoxy, is fluorine, chlorine, bromine or iodine, especially fluorine, chlorine or bromine, more especially fluorine or chlorine, especially chlorine.
- carbon-containing groups and compounds each contain from 1 up to and including 20, preferably from 1 up to and including 18, especially from 1 up to and including 10, more especially from 1 up to and including 6, especially from 1 up to and including 4, more especially from 1 up to and including 3, very especially 1 or 2, carbon atoms; methyl is especially preferred.
- Alkylene is a straight-chain or branched bridging member and is especially -CH 2 -, -CH 2 CH -, -CH 2 -CH 2 -CH 2 -, -CH -CH 2 -CH -CH 2 -, -CH 2 -CH 2 -CH 2 -CH 2 _ , -CH(CH 3 )-, -CH 2 (CH 3 )CH 2 -CH 2 -, -CH(C 2 H 5 )-, -C(CH 3 ) 2 -, -CH(CH 3 )CH 2 -, -CH(CH 3 )CH(CH 3 )- or Alkyl, as a group per se and as a structural element of other groups and compounds, such as of haloalkyl, alkoxy, alkoxyalkyl, haloalkoxy, alkoxycarbonyl, alkylthio, haloalkylthio, alkylsulfonyl and al
- Alkenyl and alkynyl - as groups per se and as structural elements of other groups and compounds, such as of haloalkenyl, haloalkynyl, alkenyloxy, haloalkenyloxy, alkynyloxy or haloalkynyloxy - are straight-chain or branched and each contains two or preferably one unsaturated carbon-carbon bond(s).
- haloalkenyl, haloalkynyl, alkenyloxy, haloalkenyloxy, alkynyloxy or haloalkynyloxy - are straight-chain or branched and each contains two or preferably one unsaturated carbon-carbon bond(s).
- Cycloalkyl - as a group per se and as a structural element of other groups and compound ' s, such as of cycloalkylalkyl - is cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl or cyclooctyl. Cyclopentyl and cyclohexyl, and especially cyclopropyl, are preferred.
- Halo-substituted carbon-containing groups and compounds, such as haloalkyl and haloalkoxy may be partially halogenated or perhalogenated, the halogen substituents in the case of polyhalogenation being the same or different.
- haloalkyl - as a group per se and as a structural element of other groups and compounds, such as of haloalkoxy - are methyl substituted from one to three times by fluorine, chlorine and/or bromine, such as CHF 2 , CF 3 or CH 2 CI; ethyl substituted from one to five times by fluorine, chlorine and/or bromine, such as CH 2 CF 3 , CF 2 CF 3 , CF 2 CCI 3 , CF 2 CHCI 2 , CF 2 CHF 2 , CF 2 CFCI 2 , CH 2 CH 2 CI, CF 2 CHBr 2 , CF 2 CHCIF, CF 2 CHBrF or CCIFCHCIF; propyl or isopropyl substituted from one to seven times by fluorine, chlorine and/or bromine, such as CH 2 CHBrCH 2 Br, CF 2 CHFCF 3 , CH 2 CF 2 CF 3 , CF 2 CF 2 CF 3
- Aryl is especially phenyl or naphthyl, preferably phenyl.
- Heterocyclyl is to be understood as being a five- to seven-membered monocyclic ring containing from one to four hetero atoms selected from the group consisting of N, O and S, or a bicyclic ring system which may contain either in only one ring - such as, for example, in quinolyl, quinoxalinyl, indolinyl, benzothiophenyl or benzofuranyl - or in both rings - such as, for example, in pteridinyl or purinyl - independently of one another, one or more hetero atoms selected from N, O and S.
- Preferred embodiments within the scope of the invention are (2) compounds according to (1) of formula (I) wherein A 3 is a straight-chain alkylene bridge, especially methylene, ethylene, propylene or butylene; more especially methylene; (3) compounds according to (1) or (2) of formula (I) wherein Q is oxygen; (4) compounds according to (1) to (3) of formula (I) wherein Y is oxygen; (5) compounds according to (1) to (4) of formula (I) wherein R ⁇ and R 2 are each independently of the other CN, nitro, d-C 6 alkyl, C ⁇ -C 6 haloalkyl, fluorine, chlorine or bromine; especially chlorine; (6) compounds according to (1) to (5) of formula (I) wherein R 3 is hydrogen; (7) compounds according to (1) to (6) of formula (I) wherein X-i and X 2 are chlorine or bromine, especially chlorine; (8) compounds according to (1) to (7) of formula (I) wherein R 4 and R 5 are H, methyl, fluorine or chlorine; (9) compounds according to
- L 3 is a leaving group, preferably chlorine or bromine, and Hal is chlorine or bromine; or (g 2 ) for the preparation of a compound of formula (I) wherein X ⁇ and X 2 are chlorine or bromine, a compound of formula (IV) is reacted in the presence of a base with a compound of formula
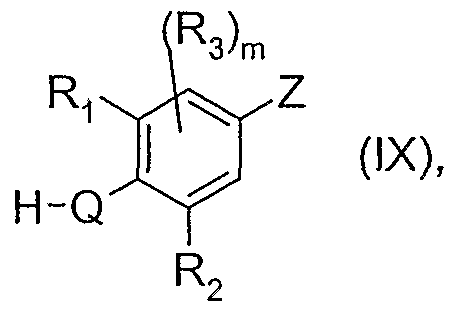
- the invention relates also to (h) a process for the preparation of a compound of formula (I), as defined under (1), and wherein Q is O, NR 7 or S, and R 7 is as defined for formula (I) under (1 ), wherein a compound of formula
- a ⁇ A 2 , A 3 , T, W, R 4 and R 5 are as defined for formula (I) under (1), and L is a leaving group, is reacted in the presence of a base with a compound of formula
- R ⁇ R 2 , R 3 and m are as defined for formula (I) under (1)
- Q is O, NR 7 or S and Z is one of the radicals Z to Z 6 as defined for formulae (II) to (VII) above
- R 7 is as defined for formula (I) under (1)
- the resulting compound of formula wherein E, T, W, Q, A-,, A 2 , A 3 , R 1 t R 2 , R 3 , R 4 , R 5 and m are as defined for formula (I) under (1) and Z is one of the radicals Zi to Z 6 as defined for formulae (II) to (VII) above, is, as necessary, that is to say according to the meaning of the radical Z, reacted further analogously to one or more of process steps (a) to (g).
- Z in compound X/a has the same meanings as Zi in the compound of formula (II)
- Z in compound X/b has the same meanings as Z 2 in formula (III), and so on.
- the invention relates also to (i) a process for the preparation of a compound of formula (I) as defined under (1 ) above, wherein a compound of formula (VIII) as defined above, is reacted analogously to process step (h) with a compound of formula
- R ⁇ R 2 , R 3 , Q, Xi, X 2 , Y and m are as defined for formula (I) under (1).
- D is a halogen atom, especially chlorine
- the radicals Z are as defined above for the compounds X/a to X/f; that is to say, Z in the compound of formula XIII/a has the same meanings as Z-i in the compound of formula (II), and Z in compound Xlll/b has the same meanings as Z 2 in formula (III), and so on.
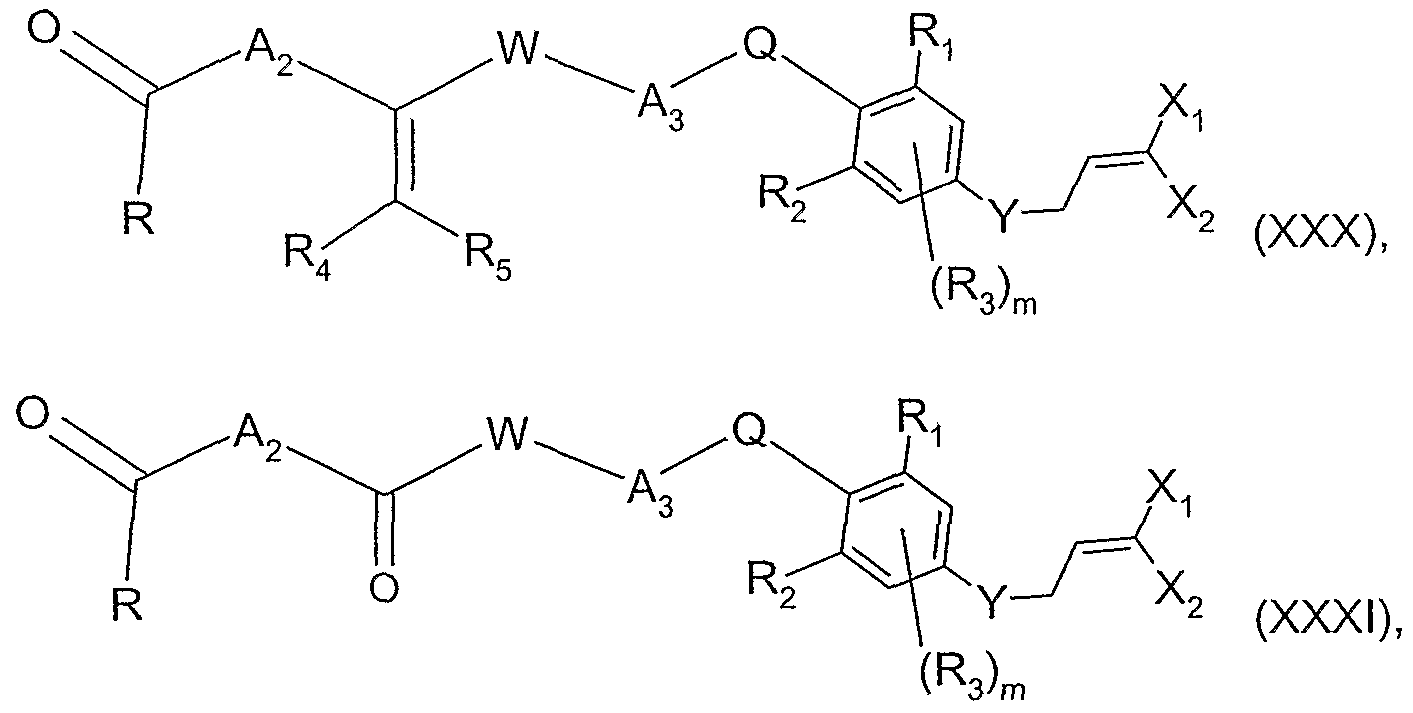
- the invention relates also to (l-i) a process for the preparation of a compound of formula
- E, T, A ⁇ and A 2 are as defined for formula (I) and R 41 and R 51 are CrC 6 alkyl, C C 3 haloalkyl, CrC 6 alkoxy-CrC 6 alkyl, C 3 -C 8 cycloalkyl, or C 3 -Cscycloalkyl-CrC 6 alkyl, wherein a compound of formula
- E, T, A T and A 2 are as defined for formula (I), R 41 and R 42 are as defined above for formula (XII) or are halogen, and R is C C 6 alkyl, wherein a compound of formula wherein E, T, A-,, A 2 and R are as defined for formula (Xlld), is reacted with a tetrahalomethane in the presence of triphenylphosphine or with an alkyltriphenyl- phosphonium salt in the presence of a base.
- the invention relates also to (m-i) a process for the preparation of a compound of formula
- the invention relates also to a process for the preparation of a compound of formula (I), wherein W is a bond and A 3 is CH 2 , wherein a compound of formula (XVIII) is processed further analogously to one of process steps (I-,) to (l 3 ) and steps (a) to (g).
- process steps (a) to (g) and (I-,) to (l 3 ) may be carried out in any desired order, that is to say, for example, there are carried out first two steps analogously to steps (a) and (b), then, for example, step (l 2 ), and subsequently the steps analogously to steps (c) to (g).
- the invention relates also to (n) a process for the preparation of a compound of formula
- compounds of formula (I) wherein the bridging members Q, T and W are O, NR 7 or S can be obtained by reacting a starting compound that contains a leaving group at the appropriate position with another starting compound that contains OH, SH or NHR 7 at the position to be bridged.
- Such starting compounds are, for example, the compounds of formulae (VIII), (XIX), (XXI), (XXII), (XXIII) and (XXIV), some of which are known and some of which are novel. Where novel, the invention relates also to such compounds.
- Further suitable compounds that are also novel, to which the invention also relates are, for example, the compounds of formulae
- a starting material that carries an aldehyde or a keto function can be combined with a starting material that carries a hydroxylamine function at the appropriate position.
- Such preparation processes require, in addition to the above-mentioned compounds of formula (XII), for example, intermediates of the following formulae:
- halogens such as fluorine, chlorine, bromine, iodine,
- Especially preferred as a leaving group are chlorine or bromine, mesylate, triflate, tosylate, especially chlorine; or chloride or bromide, especially chloride.
- oxidising agents there are used, for example, hydrogen peroxide, a peracid, such as peracetic acid, trifluoroperacetic acid, 3-chloroperbenzoic acid or a mixture, such as sodium perborate in acetic acid.
- an inorganic base such as NaOH or KOH
- aminolysis with a primary amine, such as n-butylamine can be carried out in a hydrocarbon, such as toluene or benzene, at temperatures of from 0°C to 150°C, preferably at from 20°C to 80°C.
- the preferred reaction temperature is from 0°C to 120°C, especially from 20°C to 80°C.
- a strong mineral acid for example hydrochloric acid, sulfuric acid or 4- toluenesulfonic acid.
- the reaction is carried out in an inert solvent such as, for example, benzene or toluene, or an ether, such as diethyl ether, diisopropyl ether, dioxane or tetrahydrofuran, at temperatures of from 0°C to 150°C, preferably at from 20°C to 80°C.
- an inert solvent such as, for example, benzene or toluene
- an ether such as diethyl ether, diisopropyl ether, dioxane or tetrahydrofuran
- dichlorovinyl compounds it is also possible for the process to be carried out in dimethylformamide, benzene, toluene, or in an ether, at temperatures of from 0°C to 120°C, preferably from 20°C to 80°C, and in the presence of trichloroacetic acid/ sodium trichloroacetate, then by addition of acetic anhydride, optionally with the addition of base, for example triethylamine, and finally by addition of zinc and acetic acid.
- dimethylformamide, benzene, toluene, or in an ether at temperatures of from 0°C to 120°C, preferably from 20°C to 80°C, and in the presence of trichloroacetic acid/ sodium trichloroacetate, then by addition of acetic anhydride, optionally with the addition of base, for example triethylamine, and finally by addition of zinc and acetic acid.
- a coupling reagent for example azodicar- boxylic acid diethyl or diisopropyl ester and triphenylphospine, can be used.
- a halogenated hydrocarbon such as methylene chloride
- an ether such as dioxane or tetrahydrofuran
- the reaction may be carried out, on the one hand, in an alcohol, such as methanol or ethanol, in dimethylformamide, N-methylpyrrolidone or tetrahydrofuran as solvent, in the presence of a base, such as diethylamine, diisopropylamine, piperidine, KOH, NaOH or sodium methylate, at a temperature of from -70°C to the boiling temperature of the solvent; or, on the other hand, for example, the reaction may be carried out in acetic acid at from 20°C to 120°C in the presence of ammonium acetate.
- a base such as diethylamine, diisopropylamine, piperidine, KOH, NaOH or sodium methylate
- an ether such as dioxane, tetrahydrofuran or dimethoxyethane
- aromatic solvent such as toluene or xylene
- an ether such as dioxane, tetrahydrofuran or dimethoxyethane
- an amide such as dimethylformamide, dimethylacet- amide or N-methylpyrrolidone
- aromatic solvent such as toluene or xylene
- a nitrile such as acetonitrile
- a base such as potassium or sodium carbonate.
- the invention relates especially to the preparation processes described in Examples P1 to P11.
- salts of compounds of formula (I) with bases are obtained by treatment of the free compounds with a suitable base or with a suitable ion exchange reagent.
- Salts of compounds of formula (I) can be converted into the free compounds of formula (I) in customary manner, for example by treatment with a suitable acid or with a suitable ion exchange reagent.
- Salts of compounds of formula (I) can be converted in a manner known per se into other salts of a compound of formula (I).
- the compounds of formula (I), in free form or in salt form, may be in the form of one of the possible isomers or in the form of a mixture thereof, for example, depending upon the number of asymmetric carbon atoms occurring in the molecule and their absolute and relative configuration, and/or depending upon the configuration of non-aromatic double bonds occurring in the molecule, in the form of pure isomers, such as enantiomers and/or diastereoisomers, or in the form of mixtures of isomers, such as mixtures of enantiomers, for example racemates, mixtures of diastereoisomers or mixtures of racemates.
- the invention relates both to the pure isomers and to all possible mixtures of isomers and is to be interpreted as such hereinbefore and hereinafter, even if stereochemical details are not mentioned specifically in every case.
- Mixtures of diastereoisomers, mixtures of racemates and mixtures of double bond isomers of compounds of formula (I), in free form or in salt form, that are obtainable by the process according to the invention - depending upon the starting materials and procedures chosen - or by some other method, can be separated into the pure diastereoisomers or racemates in known manner on the basis of the physico-chemical differences between the constituents, for example by means of fractional crystallisation, distillation and/or chromato- graphy.
- enantiomers such as racemates
- Mixtures of enantiomers, such as racemates, that are obtainable in a corresponding manner can be resolved into the enantiomers by known methods, for example by recrystal- lisation from an optically active solvent, by chromatography on chiral adsorbents, for example high pressure liquid chromatography (HPLC) on acetylcellulose, with the aid of suitable microorganisms, by cleavage with specific, immobilised enzymes, via the formation of inclusion compounds, for example using chiral crown ethers, only one enantiomer being complexed, or by conversion into diastereoisomeric salts and separation of the mixture of diastereoisomers so obtained, for example on the basis of their different solubilities by fractional crystallisation, into the diastereoisomers, from which the desired enantiomer can be freed by the action of suitable agents.
- HPLC high pressure liquid chromatography
- the compounds of formula (I), in free form or salt form, can also be obtained in the form of their hydrates and/or may include other solvents, for example solvents which may have been used for the crystallisation of compounds present in solid form.
- the invention relates to all those embodiments of the process according to which a compound obtainable as starting material or intermediate at any stage of the process is used as starting material and some or all of the remaining steps are carried out or a starting material is used in the form of a derivative or salt and/or its racemates or enantiomers or, especially, is formed under the reaction conditions.
- the compounds of formula (1) according to the invention are active ingredients exhibiting valuable preventive and/or curative activity with a very advantageous biocidal spectrum even at low rates of concentration, while being well tolerated by warm-blooded animals, fish and plants.
- the active ingredients according to the invention are effective against all or individual development stages of normally sensitive animal pests, but also of resistant animal pests, such as insects and members of the order Acarina.
- the insecticidal or acaricidal activity of the active ingredients according to the invention may manifest itself directly, i.e.
- Abagrotis spp. Abraxas spp., Acantholeucania spp., Acanthoplusia spp., Acarus spp., Acarus siro, Aceria spp., Aceria sheldoni, Acleris spp., Acoloithus spp., Acompsia spp., Acossus spp., Acria spp., Acrobasis spp., Acrocercops spp., Acrolepia spp., Acrolepiopsis spp., Acronicta spp., Acropolitis spp., Actebia spp., Aculus spp., Aculus pointedendali, Adoxophyes spp., Adoxophyes reticulana, Aedes spp., Aegeria spp., Aethes spp.
- Such pests include, for example, root knot nematodes, cyst- forming nematodes and also stem and leaf nematodes; especially of Heterodera spp., e.g. Heterodera schachtii, Heterodora avenae and Heterodora trifolii; Globodera spp., e.g. Globodera rostochiensis; Meloidogyne spp., e.g. Meloidogyne incognita and Meloidogyne javanica; Radopholus spp., e.g. Radopholus similis; Pratylenchus, e.g.
- Pratylenchus neglectans and Pratylenchus penetrans Pratylenchus penetrans
- Tylenchulus e.g. Tylenchulus semipenetrans
- Longidorus, Trichodorus, Xiphinema, Ditylenchus, Apheen- choides and Anguina especially Meloidogyne, e.g. Meloidogyne incognita, and Heterodera, e.g. Heterodera glycines.
- An especially important aspect of the present invention is the use of the compounds of formula (I) according to the invention in the protection of plants against parasitic feeding pests.
- Suitable additives include, for example, representatives of the following classes of active ingredient: organophosphorus compounds, nitrophenols and derivatives, formamidines, ureas, carba- mates, pyrethroids, chlorinated hydrocarbons, neonicotinoids and Bacillus thuringiensis preparations.
- Examples of especially suitable mixing partners include: azamethiphos; chlorfenvin- phos; cypermethrin, cypermethrin high-cis; cyromazine; diafenthiuron; diazinon; dichlorvos; dicrotophos; dicyclanil; fenoxycarb; fluazuron; furathiocarb; isazofos; iodfenphos; kinoprene; lufenuron; methacriphos; methidathion; monocrotophos; phosphamidon; profenofos; dio- fenolan; a compound obtainable from the Bacillus thuringiensis strain GC91 or from strain NCTC11821 ; pymetrozine; bromopropylate; methoprene; disulfoton; quinalphos; tau- fluvalinate; thiocyclam; thiometon; aldicarb; azin
- the compounds according to the invention can be used to control, i.e. to inhibit or destroy, pests of the mentioned type occurring on plants, especially on useful plants and ornamentals in agriculture, in horticulture and in forestry, or on parts of such plants, such as the fruits, blossoms, leaves, stems, tubers or roots, while in some cases plant parts that grow later are still protected against those pests.
- Target crops include especially cereals, such as wheat, barley, rye, oats, rice, maize and sorghum; beet, such as sugar beet and fodder beet; fruit, e.g. pomes, stone fruit and soft fruit, such as apples, pears, plums, peaches, almonds, cherries and berries, e.g.
- strawberries, raspberries and blackberries leguminous plants, such as beans, lentils, peas and soybeans; oil plants, such as rape, mustard, poppy, olives, sunflowers, coconut, castor oil, cocoa and groundnuts; cucurbitaceae, such as marrows, cucumbers and melons; fibre plants, such as cotton, flax, hemp and jute; citrus fruits, such as oranges, lemons, grapefruit and mandarins; vegetables, such as spinach, lettuce, asparagus, cabbages, carrots, onions, tomatoes, potatoes and paprika; lauraceae, such as avocado, cinnamon and camphor; and tobacco, nuts, coffee, aubergines, sugar cane, tea, pepper, vines, hops, bananas, natural rubber plants and ornamentals.
- the invention therefore relates also to pesticidal compositions, such as emulsifiable concentrates, suspension concentrates, directly sprayable or dilutable solutions, spreadable pastes, dilute emulsions, wettable powders, soluble powders, dispersible powders, wettable powders, dusts, granules and encapsulations of polymer substances, that comprise at least one of the compounds according to the invention, the choice of formulation being made in accordance with the intended objectives and the prevailing circumstances.
- the active ingredient is used in those compositions in pure form, a solid active ingredient, for example, in a specific particle size, or preferably together with at least one of the adjuvants customary in formulation technology, such as extenders, e.g.
- Solvents are, for example: non-hydrogenated or partly hydrogenated aromatic hydrocarbons, preferably fractions C 8 to C 12 of alkylbenzenes, such as xylene mixtures, alkylated naphthalenes or tetrahydronaphthalene, aliphatic or cycloaliphatic hydrocarbons, such as paraffins or cyclohexane, alcohols, such as ethanol, propanol or butanol, glycols and ethers and esters thereof, such as propylene glycol, dipropyiene glycol ether, ethylene glycol or ethylene glycol monomethyl or -ethyl ether, ketones, such as cyclohexanone, isophorone or diacetone alcohol, strongly polar solvents, such as N-
- the solid carriers used are as a rule natural rock powders, such as calcite, talc, kaolin, montmorillonite or attapulgite. Highly disperse silicic acids or highly disperse absorbent polymers can also be added to improve the physical properties.
- Granular adsorptive granule carriers are porous types, such as pumice, crushed brick, sepiolite or bentonite, and non-sorbent carrier materials are calcite or sand. A large number of granular materials of inorganic or organic nature can furthermore be used, in particular dolomite or comminuted plant residues.
- Surface-active compounds are, depending on the nature of the active compound to be formulated, nonionic, cationic and/or anionic surfactants or surfactant mixtures with good emulsifying, dispersing and wetting properties.
- the surfactants listed below are to be regarded only as examples; many other surfactants which are customary in formulation technology and are suitable according to the invention are described in the relevant literature.
- Nonionic surfactants are, in particular, polyglycol ether derivatives of aliphatic or cyclo- aliphatic alcohols, saturated or unsaturated fatty acids and alkylphenols, which can contain 3 to 30 glycol ether groups and 8 to 20 carbon atoms in the (aliphatic) hydrocarbon radical and 6 to 18 carbon atoms in the alkyl radical of the alkylphenols.
- Substances which are furthermore suitable are water-soluble polyethylene oxide adducts, containing 20 to 250 ethylene glycol ether and 10 to 100 propylene glycol ether groups, on propylene glycol, ethylene diaminopolypropylene glycol and alkyl polypropylene glycol having 1 to 10 carbon atoms in the alkyl chain.
- the compounds mentioned usually contain 1 to 5 ethylene glycol units per propylene glycol unit.
- examples are nonylphenol-polyethoxyethanols, castor oil polyglycol ethers, polypropylene-polyethylene oxide adducts, tributylphenoxypoly- ethoxyethanol, polyethylene glycol and octylphenoxypolyethoxyethanol.
- Other substances are fatty acid esters of polyoxyethylene sorbitan, such as polyoxyethylene sorbitan trioleate.
- the cationic surfactants are, in particular, quaternary ammonium salts which contain, as substituents, at least one alkyl radical having 8 to 22 C atoms and, as further substituents, lower, non-halogenated or halogenated alkyl, benzyl or lower hydroxyalkyl radicals.
- the salts are preferably in the form of halides, methyl-sulfates or ethyl-sulfates. Examples are stearyl-trimethyl-ammoniurn chloride and benzyl-di-(2-chloroethyl)-ethyl- ammonium bromide.
- Suitable anionic surfactants can be both water-soluble soaps and water-soluble synthetic surface-active compounds.
- Suitable soaps are the alkali metal, alkaline earth metal and substituted or unsubstituted ammonium salts of higher fatty acids (C 10 -C 22 ), such as the sodium or potassium salts of oleic or stearic acid, or of naturally occurring fatty acid mixtures, which can be obtained, for example, from coconut oil or tall oil; and furthermore also the fatty acid methyl-taurine salts.
- synthetic surfactants are more frequently used, in particular fatty sulfonates, fatty sulfates, sulfonated benzimidazole derivatives or alkylarylsulfonates.
- the fatty sulfonates and sulfates are as a rule in the form of alkali metal, alkaline earth metal or substituted or unsubstituted ammonium salts and in general have an alkyl radical of 8 to 22 C atoms, alkyl also including the alkyl moiety of acyl radicals; examples are the sodium or calcium salt of ligninsulfonic acid, of dodecylsulfuric acid ester or of a fatty alcohol sulfate mixture prepared from naturally occurring fatty acids. These also include the salts of sulfuric acid esters and sulfonic acids of fatty alcohol-ethylene oxide adducts.
- the sulfonated benzimidazole derivatives preferably contain 2 sulfonic acid groups and a fatty acid radical having about 8 to 22 C atoms.
- Alkylarylsulfonates are, for example, the sodium, calcium or triethanolammonium salts of dodecylbenzenesulfonic acid, of dibutylnaphthalenesulfonic acid or of a naphthalenesulfonic acid-formaldehyde condensation product.
- Corresponding phosphates such as salts of the phosphoric acid ester of a p-nonylphenol-(4-14)-ethylene oxide adduct or phospholipids, can further also be used.
- Emulsifiable concentrates active ingredient: 1 to 90%, preferably 5 to 20% surfactant: 1 to 30%, preferably 10 to 20% solvent: 5 to 98%, preferably 70 to 85%
- Dusts active ingredient: 0.1 to 10%, preferably 0.1 to 1% solid carrier: 99.9 to 90%, preferably 99.9 to 99%
- Suspension concentrates active ingredient: 5 to 75%, preferably 10 to 50% water: 94 to 24%, preferably 88 to 30% surfactant: 1 to 40%, preferably 2 to 30% Wettable powders: active ingredient: 0.5 to 90%, preferably 1 to 80% surfactant: 0.5 to 20%, preferably 1 to 15% solid carrier: 5 to 99%, preferably 15 to 98%
- Granules active ingredient: 0.5 to 30%, preferably 3 to 15% solid carrier: 99.5 to 70%, preferably 97 to 85%
- compositions according to the invention may also comprise further solid or liquid adjuvants, such as stabilisers, e.g. vegetable oils or epoxidised vegetable oils (e.g. epoxi- dised coconut oil, rapeseed oil or soybean oil), antifoams, e.g. silicone oil, preservatives, viscosity regulators, binders and/or tackifiers as well as fertilisers or other active ingredients for obtaining special effects, e.g. acaricides, bactericides, fungicides, nematicides, mollus- cicides or selective herbicides.
- the crop protection products according to the invention are prepared in known manner, in the absence of adjuvants, e.g.
- the invention relates likewise to those processes for the preparation of the compositions according to the invention and to the use of the compounds of formula (I) in the preparation of those compositions.
- the invention relates also to the methods of application of the crop protection products, i.e.
- Typical rates of concentration are from 0.1 to 1000 ppm, preferably from 0.1 to 500 ppm, of active ingredient.
- the rates of application per hectare are generally from 1 to 2000 g of active ingredient per hectare, especially from 10 to 1000 g/ha, preferably from 20 to 600 g/ha.
- a preferred method of application in the area of crop protection is application to the foliage of the plants (foliar application), the frequency and the rate of application being dependent upon the risk of infestation by the pest in question.
- the active ingredient can also penetrate the plants through the roots (systemic action) when the locus of the plants is impregnated with a liquid formulation or when the active ingredient is incorporated in solid form into the locus of the plants, for example into the soil, e.g. in granular form (soil application). In the case of paddy rice crops, such granules may be applied in metered amounts to the flooded rice field.
- the crop protection products according to the invention are also suitable for protecting plant propagation material, e.g. seed, such as fruits, tubers or grains, or plant cuttings, against animal pests.
- the propagation material can be treated with the composition before planting: seed, for example, can be dressed before being sown.
- the active ingredients according to the invention can also be applied to grains (coating), either by impregnating the seeds in a liquid formulation or by coating them with a solid formulation.
- the composition can also be applied to the planting site when the propagation material is being planted, for example to the seed furrow during sowing.
- compositions according to the invention are also suitable for protecting plant propagation material, including genetically modified propagation material, e.g. seed, such as fruits, tubers or grains, or plant seedlings, against animal pests.
- the propagation material can be treated with the composition before being planted: seed, for example, can be dressed before being sown.
- the active ingredients according to the invention can also be applied to grains (coating), either by impregnating the seeds in a liquid formulation or by coating them with a solid formulation.
- the composition can also be applied to the planting site when the propagation material is being planted, for example to the seed furrow during sowing.
- the invention relates also to such methods of treating plant propagation material and to the plant propagation material so treated.
- the following Examples serve to illustrate the invention. They do not limit the invention.
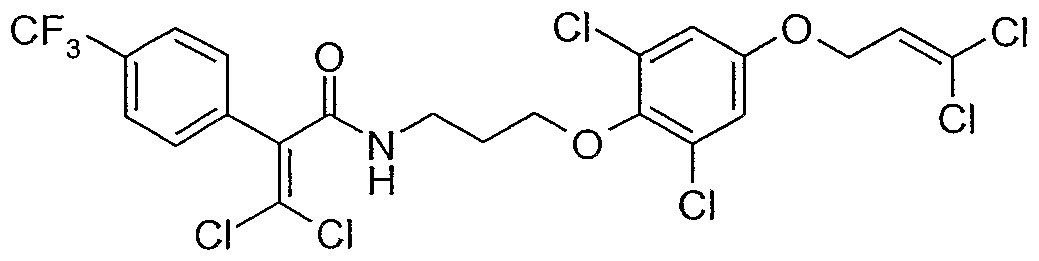
- Example P1 Preparation of 3,3-dichloro-N- ⁇ 3-[2,6-dichloro-4-(3,3-dichloro-allyloxy)- phenoxy]-propyl ⁇ -2-(4-trifluoromethyl-phenyl)-acrylamide
- P1.1 680 mg of oxo-(4-trifluoromethyl-phenyl)-acetic acid ethyl ester and 2.2 g of tri- phenylphosphine are introduced into 8 ml of acetonitrile.
- reaction mixture is cooled to 50°C and a solution of 500 mg of 1-[2,6-dichloro-4-(3,3-dichloro-allyloxy)-phenoxy]-3-(4-tri- fluoromethyl-phenoxy)-propan-2-one in 5 ml of toluene is added in the course of 5 minutes. After 2 hours at 80°C, the reaction mixture is filtered, diluted with ethyl acetate and washed with water. After concentration of the organic phase and purification over silica gel, the title compound is obtained.
- Example P4 Preparation of 1 ,3-dichloro-5-(3,3-dichloro-allyloxy)-2-[3,3-dichloro-2- (4-trifiuoromethyl-phenoxymethyl)-allyloxy]-benzene
- Example P5 Preparation of 1 ,3-dichloro-5-(3,3-dichloro-allyloxy)-2-[4-(4-fluoro- phenyl)-pent-4-enyloxy]-benzene P5.1) 2.0 g of 2-(3-chloro-propyl)-2-(4-fluoro-phenyl)-[1 ,3]dioxolane are added to a suspension of 2.36 g of 2,6-dichioro-4-(3,3-dichloro-allyloxy)-phenol, 1.7 g of potassium carbonate and 120 mg of potassium iodide in 50 ml of acetone. The mixture is then stirred for 48 hours at 60°C.
- reaction mixture is concentrated and the residue is taken up in water and extracted with ethyl acetate. After concentration of the organic phase and purification over silica gel, 2- ⁇ 3-[2,6-dichloro-4-(3,3-dichloro-allyloxy)-phenoxy]-propyl ⁇ -2-(4- fluoro-phenyl)-[1 ,3]dioxolane is obtained.
- Example P6 Preparation of 2-(4-fluoro-phenyl)-3-methyl-but-2-enoic acid ⁇ 3-[2,6-di- chloro-4-(3,3-dichloro-allyloxy)-phenoxy]-propyl ⁇ -amide
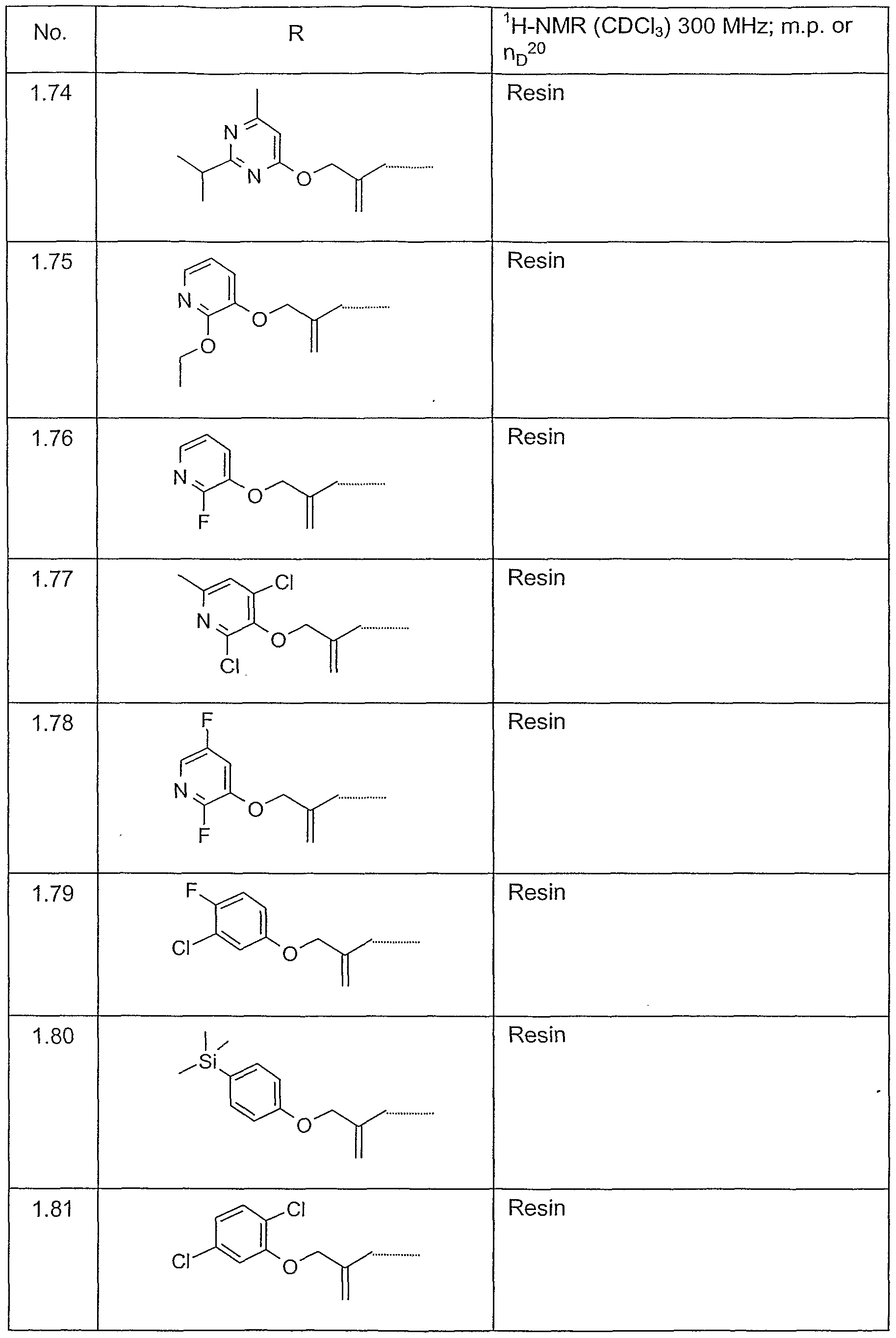
- Example P7 The further Examples in the following Tables can also be prepared in a manner analogous to that described above.
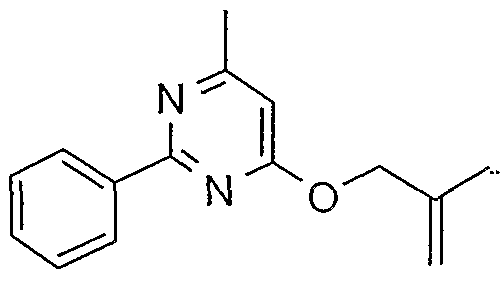
- Table 1 Compounds of formula
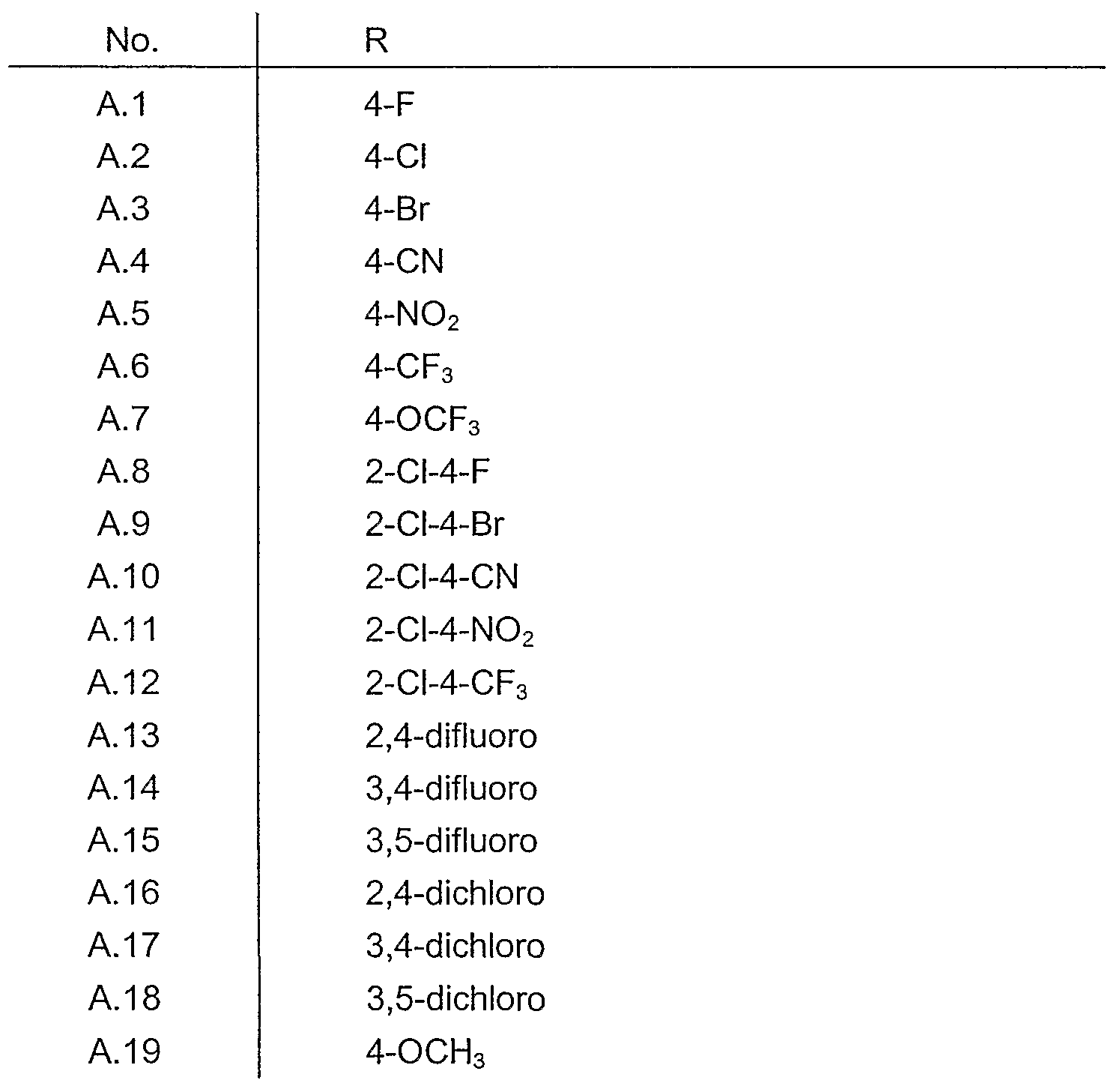
- Table 2 A compound of general formula X-, and X 2 are chlorine and the substituent R for each compound corresponds to A.146 of Table A.
- Table 3 A compound of general formula X-i and X 2 are chlorine and the substituent R for each compound corresponds to A.146 of Table A.
- Table 4 A compound of general formula Xi and X 2 are chlorine and the substituent R for each compound corresponds 1 to A.146 of Table A.
- Table 5 A compound of general formula X-i and X 2 are chlorine and the substituent R for each compound corresponds 1 to A.146 of Table A.
- Table 6 A compound of general formula X and X 2 are chlorine and the substituent R for each compound corresponds 1 to A.146 of Table A.
- Table 7 A compound of general formula X 1 and X 2 are bromine and the substituent R for each compound corresponds 1 to A.146 of Table A.
- Table 8 A compound of general formula X ⁇ and X 2 are bromine and the substituent R for each compound corresponds 1 to A.146 of Table A.
- Table 9 A compound of general formula X T and X 2 are bromine and the substituent R for each compound corresponds 1 to A.146 of Table A.
- Table 10 A compound of general formula (Id) wherein X n and X 2 are bromine and the substituent R for each compound corresponds to a line A.1 to A.146 of Table A.
- Table 11 A compound of general formula (le) wherein X ⁇ and X 2 are bromine and the substituent R for each compound corresponds to a line A.1 to A.146 of Table A.
- Table B Compounds of formulae
- Table 12 A compound of general formula (If) wherein X-i and X 2 are chlorine and the substituent R for each compound corresponds to a line B.1 to B.16 of Table B.
- Table 13 A compound of general formula (Ig) wherein and X 2 are chlorine and the substituent R for each compound corresponds to a line B.1 to B.16 of Table B.
- Table 14 A compound of general formula (If) wherein Xi and X 2 are bromine and the substituent R for each compound corresponds to a line B.1 to B.16 of Table B.
- Table 15 A compound of general formula (Ig) wherein and X 2 are bromine and the substituent R for each compound corresponds to a line B.1 to B.16 of Table B.
- Example F1 Emulsifiable concentrates a) b) c) active ingredient 25% 40% 50% calcium dodecylbenzenesulfonate 5% 8% 6% castor oil polyethylene glycol ether (36 mol EO) 5% - - tributylphenol polyethylene glycol ether (30 mol EO) - 12% 4% cyclohexanone - 15% 20% xylene mixture 65% 25% 20%
- Example F2 Solutions a) b) c) d) active ingredient 80% 10% 5% 95% ethylene glycol monomethyl ether 20% - - - polyethylene glycol (MW 400) - 70% - -
- Example F3 Granules a) b) c) d) active ingredient 5% 10% 8% 21 % kaolin 94% - 79% 54% highly dispersed silicic acid 1 % - 13% 7% attapulgite _ 90% - 18%
- the active ingredient is dissolved in dichloromethane, the solution is sprayed onto the carrier mixture and the solvent is evaporated off in vacuo.
- Example B1 Action against Heliothis virescens caterpillars
- the soybean plants are populated with 10 caterpillars of Heliothis virescens in the first stage and placed in a plastics container. Evaluation is made 6 days later. The percentage reduction in population and the percentage reduction in feeding damage (% activity) are determined by comparing the number of dead caterpillars and the feeding damage on the treated plants with that on the untreated plants.
- the compounds of the Tables exhibit good activity against Heliothis virescens in this test.
- the compounds 1.2 to 1.6, 1.8 to 1.9, 1.11 , 1.13 to 1.14, 1.18 to 1.19, 1.32 to 1.37, 1.41 to 1.44 and 1.46 to 1.115 are more than 80 % effective.
- Example B2 Action against Plutella xylostella caterpillars
- Young cabbage plants are sprayed with an aqueous emulsion spray mixture comprising 400 ppm of test compound. After the spray-coating has dried, the cabbage plants are populated with 10 caterpillars of Plutella xylostella in the third stage and placed in a plastics container. Evaluation is made 3 days later. The percentage reduction in population and the percentage reduction in feeding damage (% activity) are determined by comparing the number of dead caterpillars and the feeding damage on the treated plants with that on the untreated plants.
- the compounds of the Tables exhibit good activity against Plutella xylostella in this test.
- the compounds 1.2 to 1.6, 1.8 to 1.9, 1.11 , 1.13 to 1.14, 1.18 to 1.19, 1.32 to 1.37, 1.41 to 1.44 and 1.46 to 1.115 are more than 80 % effective.
- Example B3 Action against Spodoptera littoralis
- Young soybean plants are sprayed with an aqueous emulsion spray mixture comprising 400 ppm of test compound and, after the spray-coating has dried, the plants are populated with 10 caterpillars of Spodoptera littoralis in the first stage and then placed in a plastics container. 3 days later, the percentage reduction in population and the percentage reduction in feeding damage (% activity) are determined by comparing the number of dead caterpillars and the feeding damage on the treated plants with that on untreated plants.
- the compounds of the Tables exhibit good activity against Spodoptera littoralis in this test.
- the compounds 1.2 to 1.6, 1.8 to 1.9, 1.11 , 1.13 to 1.14, 1.18 to 1.19, 1.32 to .37, 1.41 to 1.44 and 1.46 to 1.115 are more than 80 % effective.
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Environmental Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Plural Heterocyclic Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Nitrogen- Or Sulfur-Containing Heterocyclic Ring Compounds With Rings Of Six Or More Members (AREA)
- Pyridine Compounds (AREA)
- Pyrane Compounds (AREA)
- Heterocyclic Compounds Containing Sulfur Atoms (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP04764476A EP1659863A2 (en) | 2003-08-26 | 2004-08-25 | Pesticidally active phenol derivatives |
| JP2006524326A JP2007503416A (en) | 2003-08-26 | 2004-08-25 | Insecticide active phenol derivatives |
| US10/568,993 US7638514B2 (en) | 2003-08-26 | 2004-08-25 | Pesticidally active phenol derivatives |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH14542003 | 2003-08-26 | ||
| CH01454/03 | 2003-08-26 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2005019147A2 true WO2005019147A2 (en) | 2005-03-03 |
| WO2005019147A3 WO2005019147A3 (en) | 2005-04-07 |
Family
ID=34200996
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2004/009500 Ceased WO2005019147A2 (en) | 2003-08-26 | 2004-08-25 | Pesticidally active phenol derivatives |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US7638514B2 (en) |
| EP (1) | EP1659863A2 (en) |
| JP (1) | JP2007503416A (en) |
| WO (1) | WO2005019147A2 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010060379A1 (en) * | 2008-11-28 | 2010-06-03 | 中国中化集团公司 | Ether compounds with nitrogen-containing 5-member heterocycle and the uses thereof |
| EP1755613A4 (en) * | 2004-05-18 | 2010-08-25 | Bayer Cropscience Ag | Substituted cyclic urea derivatives |
| WO2012130137A1 (en) * | 2011-03-30 | 2012-10-04 | 中国中化股份有限公司 | Aryloxy dihalopropenyl ether compound and use thereof |
| CN105693523A (en) * | 2016-03-14 | 2016-06-22 | 贵州大学 | Chalcone derivate with 1,1-dichloropropene and preparation method and application thereof |
| CN111838158A (en) * | 2020-08-04 | 2020-10-30 | 华南师范大学 | Application of a compound in the preparation of pesticides |
| CN111972419A (en) * | 2020-08-04 | 2020-11-24 | 华南师范大学 | Application of compound in preparation of insecticide |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW200406370A (en) | 2002-06-28 | 2004-05-01 | Syngenta Participations Ag | 4-(3,3-Dihalo-allyloxy)phenoxy alkyl derivatives |
| CN102464621B (en) * | 2010-11-19 | 2014-09-03 | 中国中化股份有限公司 | Ether compound containing pyrimidine rings and application thereof |
| CN105766916B (en) * | 2014-12-23 | 2018-12-14 | 沈阳中化农药化工研发有限公司 | A kind of insecticidal preparation and its application |
| CN111454204B (en) * | 2019-01-22 | 2023-10-17 | 华中师范大学 | Cyanopyridine-containing dichloropropylene ether compounds, preparation methods and applications thereof, and a pesticide |
| CN116925007B (en) * | 2023-07-20 | 2025-12-02 | 南通慧源塑胶有限公司 | A method for preparing and using a 1,3,4-oxadiazole compound containing a chloropropene unit. |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI241889B (en) | 2000-04-28 | 2005-10-21 | Sumitomo Chemical Co | Insecticidal and acaricidal composition |
| JP4945854B2 (en) * | 2000-04-28 | 2012-06-06 | 住友化学株式会社 | Insecticide and acaricide composition |
| TW200406370A (en) | 2002-06-28 | 2004-05-01 | Syngenta Participations Ag | 4-(3,3-Dihalo-allyloxy)phenoxy alkyl derivatives |
| TW200410632A (en) | 2002-12-11 | 2004-07-01 | Syngenta Participations Ag | Dihalo-allyloxy-phenol derivatives having pesticidal activity |
-
2004
- 2004-08-25 EP EP04764476A patent/EP1659863A2/en not_active Withdrawn
- 2004-08-25 WO PCT/EP2004/009500 patent/WO2005019147A2/en not_active Ceased
- 2004-08-25 JP JP2006524326A patent/JP2007503416A/en active Pending
- 2004-08-25 US US10/568,993 patent/US7638514B2/en not_active Expired - Fee Related
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1755613A4 (en) * | 2004-05-18 | 2010-08-25 | Bayer Cropscience Ag | Substituted cyclic urea derivatives |
| CN102216294B (en) * | 2008-11-28 | 2013-08-28 | 中国中化股份有限公司 | Ether compound with nitrogen-containing five-membered heterocycle and application thereof |
| CN102216294A (en) * | 2008-11-28 | 2011-10-12 | 中国中化股份有限公司 | Ether compound with nitrogen-containing five-membered heterocycle and application thereof |
| US8222280B2 (en) | 2008-11-28 | 2012-07-17 | Sinochem Corporation | Ether compounds with nitrogen-containing 5-member heterocycle and uses thereof |
| WO2010060379A1 (en) * | 2008-11-28 | 2010-06-03 | 中国中化集团公司 | Ether compounds with nitrogen-containing 5-member heterocycle and the uses thereof |
| WO2012130137A1 (en) * | 2011-03-30 | 2012-10-04 | 中国中化股份有限公司 | Aryloxy dihalopropenyl ether compound and use thereof |
| CN103201266A (en) * | 2011-03-30 | 2013-07-10 | 中国中化股份有限公司 | Aryloxydihalopropene ether compounds and their applications |
| US8969332B2 (en) | 2011-03-30 | 2015-03-03 | Sinochem Corporation | Aryloxy dihalopropenyl ether compounds and uses thereof |
| CN103201266B (en) * | 2011-03-30 | 2016-04-27 | 沈阳中化农药化工研发有限公司 | Aryloxydihalopropene ether compounds and their applications |
| CN105693523A (en) * | 2016-03-14 | 2016-06-22 | 贵州大学 | Chalcone derivate with 1,1-dichloropropene and preparation method and application thereof |
| CN105693523B (en) * | 2016-03-14 | 2018-11-13 | 贵州大学 | A kind of chalcone derivative, the preparation method and use of the dichloropropylene containing 1,1- |
| CN111838158A (en) * | 2020-08-04 | 2020-10-30 | 华南师范大学 | Application of a compound in the preparation of pesticides |
| CN111972419A (en) * | 2020-08-04 | 2020-11-24 | 华南师范大学 | Application of compound in preparation of insecticide |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2005019147A3 (en) | 2005-04-07 |
| US7638514B2 (en) | 2009-12-29 |
| US20070142229A1 (en) | 2007-06-21 |
| JP2007503416A (en) | 2007-02-22 |
| EP1659863A2 (en) | 2006-05-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20100016383A1 (en) | 4-(3,3-dihalo-allyloxy)phenol derivatives having pesticidal properties | |
| US7414064B2 (en) | 4-(3,3-dihalo-allyloxy) phenoxy alkyl derivatives | |
| US7638514B2 (en) | Pesticidally active phenol derivatives | |
| EP1613639B1 (en) | Avermectins substituted in the 4'' and 4´-positions having pesticidal properties | |
| US7569727B2 (en) | Pesticidally active ketone and oxime derivatives | |
| EP1572612B1 (en) | Dihalo-allyloxy-phenol derivatives having pesticidal activity | |
| US20060063820A1 (en) | 2,6-dihalo-4-(3,3-dichloro-allyloxy)-benzylalcohole derivatives having insecticidal and acaricidal properties | |
| US7632820B2 (en) | Avermectin and avemectin monosaccharide derivatives substituted in the 4″- or 4′-position having pesticidal properties | |
| US7521429B2 (en) | Avermectin B1 monosaccharide derivatives | |
| KR20050016949A (en) | 4-(3,3-Dihalo-allyloxy)phenoxy alkyl derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2004764476 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006524326 Country of ref document: JP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004764476 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007142229 Country of ref document: US Ref document number: 10568993 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 10568993 Country of ref document: US |