WO2005010080A1 - 温度応答性ハイドロゲル - Google Patents
温度応答性ハイドロゲル Download PDFInfo
- Publication number
- WO2005010080A1 WO2005010080A1 PCT/JP2004/011006 JP2004011006W WO2005010080A1 WO 2005010080 A1 WO2005010080 A1 WO 2005010080A1 JP 2004011006 W JP2004011006 W JP 2004011006W WO 2005010080 A1 WO2005010080 A1 WO 2005010080A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- temperature
- hyaluronic acid
- poly
- glycol
- equivalents
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08B—POLYSACCHARIDES; DERIVATIVES THEREOF
- C08B37/00—Preparation of polysaccharides not provided for in groups C08B1/00 - C08B35/00; Derivatives thereof
- C08B37/006—Heteroglycans, i.e. polysaccharides having more than one sugar residue in the main chain in either alternating or less regular sequence; Gellans; Succinoglycans; Arabinogalactans; Tragacanth or gum tragacanth or traganth from Astragalus; Gum Karaya from Sterculia urens; Gum Ghatti from Anogeissus latifolia; Derivatives thereof
- C08B37/0063—Glycosaminoglycans or mucopolysaccharides, e.g. keratan sulfate; Derivatives thereof, e.g. fucoidan
- C08B37/0072—Hyaluronic acid, i.e. HA or hyaluronan; Derivatives thereof, e.g. crosslinked hyaluronic acid (hylan) or hyaluronates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
- A61K9/0024—Solid, semi-solid or solidifying implants, which are implanted or injected in body tissue
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/20—Polysaccharides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/52—Hydrogels or hydrocolloids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L71/00—Compositions of polyethers obtained by reactions forming an ether link in the main chain; Compositions of derivatives of such polymers
- C08L71/02—Polyalkylene oxides
Definitions
- the present invention relates to a compound comprising hyaluronic acid and a polyalkylene oxide derivative. More specifically, the present invention relates to a temperature-responsive hide mouth gel comprising hyaluronic acid and a polyalkylene oxide derivative.
- the above-mentioned example requires two incisions for cell harvesting and implanting into the body, which is extremely burdensome for the patient.
- the development of artificial materials suitable for endoscopic surgery is very important.
- the required properties of the artificial material are as follows: 1) the shape can be freely controlled (can be directly injected into the affected area), 2) cells and growth factors can be easily embedded, etc. This material is very suitable for regenerative medicine, and is considered to have a great advantage in regenerative medicine.
- Temperature-responsive hide mouth gel is a type of lower critical solution temperature (LCST) that hydrates at a certain temperature or lower and dehydrates at a certain temperature or higher and causes a volume change in an aqueous environment.
- LCST lower critical solution temperature
- the above can be classified as Upper Critical Solution Temperature (UCST) type, which causes volume change by hydration.
- UCT Upper Critical Solution Temperature
- the type of hide-mouth gel having the property of LCST which is superior in terms of response speed and the like is preferably used in a drug delivery system.
- Hide-type gels of the type having the property of LCST for example, are uniformly dissolved in an aqueous solution below a certain temperature because the interaction between polymer and water takes precedence, but become hydrated above a certain temperature.
- Polymers having the property of LCST in a water-polymer system include N-substituted (meth) acrylamide derivatives such as poly (N-isopropylacrylamide), poly (N-acryloylpyrrolidine), and poly (N-acryloylbiperyl).
- N-containing cyclic polymers such as gin), vinyl group-containing amino acids such as poly (N-acryloyl L-proline) and esters thereof, poly (vinyl methyl ether), poly (ethylene glycol) Z poly (propylene glycol), Polylactic acid-polyglycolic acid-polyethylene oxide copolymers are known.
- poly (N-isopropylacrylamide) copolymer is a typical polymer having a sharp transition and a phase transition temperature suitable for application to biological systems.
- phase transition temperature Various aspects of controlling the phase transition temperature, improving the phase transition temperature, and elucidating the phase transition mechanism Research is being actively pursued from this point.
- thermoresponsiveness can be imparted to a biomatrix material, it is expected that an ideal temperature-responsive hide-mouth gel excellent in bioabsorbability and biocompatibility will be obtained.
- examples of attempts to impart temperature responsiveness to a biomatrix material include chitosan (WO 01 36000) and hyaluronic acid (WO 99Z24070).
- chitosan WO 01 36000
- hyaluronic acid WO 99Z24070
- a main object of the present invention is to provide a temperature-responsive hide mouth gel excellent in bioabsorbability and biocompatibility. More specifically, the present invention provides a temperature-responsive hide-mouth gel that can cope with various response temperature ranges.
- the present invention is as follows.
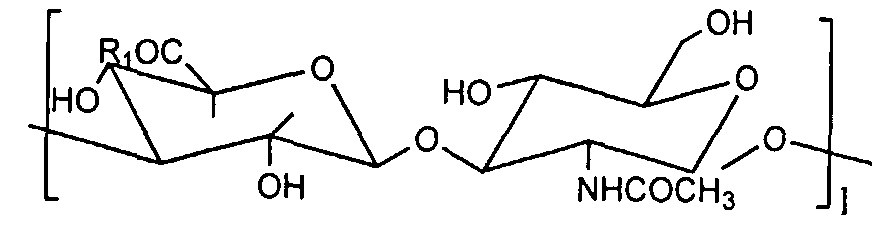
- R2 is NH, 0, R3 is H, CH3, R4 is C2H4, CH2CH (CH3), R5 is any of H, CH3, C2H5, C4H9.
- I is 300-30,000, m is 3 It is an integer up to 140.
- Hydrate gel wherein the content of the carboxyl group of the hyaluronic acid is 100 equivalents of the polyalkylene oxide derivative residue is 5 to 100 equivalents.
- FIG. 1 is a phase transition behavior diagram of a compound obtained by introducing 10 equivalents of hyaluronic acid to 100 equivalents of a carboxyl group of hyaluronic acid.
- FIG. 2 is a phase transition behavior diagram of a compound obtained by introducing 50 equivalents of hyaluronic acid to 100 equivalents of the carboxyl group of hyaluronic acid by using Diephamin (registered trademark) XTJ-507.
- FIG. 3 is a phase transition behavior diagram of a compound obtained by introducing 100 equivalents of hyaluronic acid to 100 equivalents of the carboxyl group of hyaluronic acid.
- Figure 4 shows the phase transition behavior of sodium hyaluronate.
- Figure 5 shows the phase transition behavior of propyl hyaluronate.
- the hyaluronic acid used in the present invention may be either extracted from animal tissues or produced by fermentation.
- the strain used in the fermentation method is a microorganism having the ability to produce hyaluronic acid of the genus Streptococcus, and Streptococcus' equi FM-10 0 (JP-A-63-123392) and Streptococcus' equi FM-300 (JP-A-2-234689). Use those cultured and purified using these mutant strains.
- the molecular weight of hyaluronic acid is preferably about 1 ⁇ 10 5 to 1 ⁇ 10 7 dalton.
- the hyaluronic acid referred to in the present invention also includes alkali metal salts thereof, for example, sodium, potassium and lithium salts.
- the polyalkylene oxide used in the present invention is preferably 1) polypropylene glycol or 2) a copolymer composed of poly (propylene glycol) and poly (ethylene glycol).
- polypropylene glycol or 2) a copolymer composed of poly (propylene glycol) and poly (ethylene glycol).
- Compounds having an amino group at the terminal such as poly (ethylene glycol) butoxid
- 1-polypropylene glycol methoxide When introduced into hyaluronic acid through an ester bond, 1-polypropylene glycol methoxide, 1-polypropylene glycol ethoxide, 1-polypropylene glycol propoxide, 1-polypropylene glycol butoxide are used.
- the molecular weight of the above polyalkylene oxide derivative is preferably 200-6,000. If it is less than 200, the reaction product with hyaluronic acid does not show temperature responsiveness. On the other hand, if it is more than 6,000, a precipitate is formed and a hide mouth gel is not formed.
- the copolymerization ratio of poly (propylene daricol) nopoly (ethylene glycol) is 1 / 99-99.9 / 0.9. Power of 1 ⁇ I like it. More preferably, the ratio is from 2080 to 99.9 / 0.1. Outside this range, the reaction product with hyaluronic acid does not exhibit temperature responsiveness.
- the content of the polyalkylene oxide derivative is preferably 5 to 100 equivalents based on 100 equivalents of the carboxyl group of hyaluronic acid. If it is less than 5 equivalents, the reaction product with hyaluronic acid does not show temperature response.
- a typical reaction method of the hyaluronic acid and the polyalkylene oxide derivative includes the following two methods.
- the curves in FIGS. 1 to 3 corresponding to Examples 1 to 3 rise and the temperature at which the phase transition starts is compared. It can be seen that the higher the amount of (oxide derivative), the lower the temperature. In other words, by controlling the amount of ligefamine (polyalkylene oxide derivative) to be introduced, it becomes possible to prepare a hyaluronic acid hydrate mouth gel having a desired phase transition temperature.
- phase transition temperature can be changed depending on the molecular weight of the polyalkylene oxide derivative used and the molecular weight of hyaluronic acid.
- Injectable gel is expected to be a scaffold that is easy to handle because it can be easily mixed with cells and humoral factors in a liquid state at a temperature lower than body temperature. Therefore, a gel that undergoes a phase transition around body temperature cannot be used as an injectable gel.
- the hide mouth gel of the present invention can be used as an injectable gel because it undergoes a phase transition at a temperature close to body temperature.
- the present invention can provide a temperature-responsive hydrid gel comprising hyaluronic acid and a polyalkylene oxide derivative and having excellent bioabsorbability and biocompatibility.
- This temperature-responsive gel is useful as an artificial material in regenerative medicine targeting endoscopic surgery.
- a column ( ⁇ 1.2 XL 20 cm) was packed with an ion exchange resin (DOWEX (registered trademark) 50WX8; Total exchange capacity 1.9 eqZI), and an aqueous solution of tetra-n-phthylammonium bromide (48 gZ100 ml) was added. And replaced. After the replacement, flow ion-exchanged water until ⁇ becomes neutral, then pass an aqueous solution of sodium hyaluronate (2gZ1 OOOml) through the column and freeze-dry to obtain tetra-n-butylammonium hyaluronate.
- DOWEX registered trademark
- 50WX8 Total exchange capacity 1.9 eqZI
- thermoresponsive gel is useful as an artificial material in regenerative medicine targeting endoscopic surgery.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Dermatology (AREA)
- Veterinary Medicine (AREA)
- Transplantation (AREA)
- Organic Chemistry (AREA)
- Polymers & Plastics (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Biomedical Technology (AREA)
- Dispersion Chemistry (AREA)
- Neurosurgery (AREA)
- Pharmacology & Pharmacy (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Materials Engineering (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
- Materials For Medical Uses (AREA)
- Other Resins Obtained By Reactions Not Involving Carbon-To-Carbon Unsaturated Bonds (AREA)
- Cosmetics (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2005512109A JP4500263B2 (ja) | 2003-07-28 | 2004-07-27 | 温度応答性ハイドロゲル |
| US10/565,019 US20060235114A1 (en) | 2003-07-28 | 2004-07-27 | Temperature-responsive hydrogel |
| EP04748165A EP1659143A4 (en) | 2003-07-28 | 2004-07-27 | HYDROGEL SENSITIVE AT TEMPERATURE |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003280757 | 2003-07-28 | ||
| JP2003-280757 | 2003-07-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2005010080A1 true WO2005010080A1 (ja) | 2005-02-03 |
Family
ID=34100893
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2004/011006 WO2005010080A1 (ja) | 2003-07-28 | 2004-07-27 | 温度応答性ハイドロゲル |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20060235114A1 (ja) |
| EP (1) | EP1659143A4 (ja) |
| JP (1) | JP4500263B2 (ja) |
| TW (1) | TW200510012A (ja) |
| WO (1) | WO2005010080A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010046442A (ja) * | 2008-08-25 | 2010-03-04 | Olympus Corp | 膨隆液組成物および内視鏡装置 |
| JP2010539271A (ja) * | 2007-09-11 | 2010-12-16 | サントル、ナショナール、ド、ラ、ルシェルシュ、シアンティフィク、(セーエヌエルエス) | キトサンの官能化に使用できる感熱性((ポリエチレンオキシド)ポリ(プロピレンオキシド))誘導体の製造方法 |
| JP2016141769A (ja) * | 2015-02-04 | 2016-08-08 | 帝人株式会社 | ヒアルロン酸誘導体および医療用製剤 |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2005299748B2 (en) * | 2004-10-21 | 2009-04-09 | Tgel Bio Co., Ltd | In situ controlled release drug delivery system |
| US20090227689A1 (en) * | 2007-03-05 | 2009-09-10 | Bennett Steven L | Low-Swelling Biocompatible Hydrogels |
| US20090227981A1 (en) * | 2007-03-05 | 2009-09-10 | Bennett Steven L | Low-Swelling Biocompatible Hydrogels |
| AU2008339362B2 (en) | 2007-12-17 | 2013-01-10 | Teijin Limited | Cellulose derivative and hydrogel thereof |
| CN103087333B (zh) * | 2012-11-13 | 2014-08-06 | 西安交通大学 | 一种可快速解离型温敏性透明质酸水凝胶的制备方法 |
| FR3023485B1 (fr) * | 2014-07-11 | 2016-07-01 | Celenys | Procede de modification de polysaccharides par greffage de polyetheramines, polysaccharides ainsi modifies et preparations les comportant et presentant des proprietes rheologiques thermosensibles |
| EP3250163B1 (en) | 2015-01-27 | 2023-07-12 | Medivance Incorporated | Medical pad for thermotherapy |
| CN111065359A (zh) * | 2017-06-16 | 2020-04-24 | 埃斯库莱泰克股份有限公司 | 热反应性聚合物及其用途 |
| EP4118147A1 (en) * | 2020-03-11 | 2023-01-18 | Solvcor Technologies, LLC | Liquid-liquid phase transition compositions and processes |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07278203A (ja) * | 1993-11-03 | 1995-10-24 | Collagn Corp | グリコサミノグリカン−合成ポリマー結合体 |
| WO2002044276A2 (en) * | 2000-11-28 | 2002-06-06 | Focal, Inc. | Polyalkylene glycol viscosity-enhancing polymeric formulations |
| WO2003028781A1 (fr) * | 2001-08-21 | 2003-04-10 | Japan Science And Technology Agency | Complexe polycation-glycosaminoglycane reticule par un agent de reticulation polyfonctionnel et procede de production associe |
Family Cites Families (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5258041A (en) * | 1982-09-29 | 1993-11-02 | Bio-Metric Systems, Inc. | Method of biomolecule attachment to hydrophobic surfaces |
| US5162430A (en) * | 1988-11-21 | 1992-11-10 | Collagen Corporation | Collagen-polymer conjugates |
| US5003022A (en) * | 1989-02-10 | 1991-03-26 | Penford Products Company | Starch graft polymers |
| US5270421A (en) * | 1990-05-08 | 1993-12-14 | University Of Iowa Research Foundation | Sugar-based polymers |
| EP0516026A1 (en) * | 1991-05-28 | 1992-12-02 | Takeda Chemical Industries, Ltd. | Hydrogel and method of producing same |
| US5550189A (en) * | 1992-04-17 | 1996-08-27 | Kimberly-Clark Corporation | Modified polysaccharides having improved absorbent properties and process for the preparation thereof |
| US20040138329A1 (en) * | 1992-04-20 | 2004-07-15 | Board Of Regents Of The University Of Texas System | Gels for encapsulation of biological materials |
| JPH0733682A (ja) * | 1993-07-26 | 1995-02-03 | Shiseido Co Ltd | 新規複合体、それを担体として利用する徐放性製剤 |
| US6080194A (en) * | 1995-02-10 | 2000-06-27 | The Hospital For Joint Disease Orthopaedic Institute | Multi-stage collagen-based template or implant for use in the repair of cartilage lesions |
| CZ293968B6 (cs) * | 1995-08-29 | 2004-08-18 | Fidiaáadvancedábiopolymersźásrl | Složený biomateriál pro zabránění chirurgických adhezí tkáněŹ jeho použití a způsob |
| US6833408B2 (en) * | 1995-12-18 | 2004-12-21 | Cohesion Technologies, Inc. | Methods for tissue repair using adhesive materials |
| EP0907721A1 (en) * | 1996-05-28 | 1999-04-14 | Brown University Research Foundation | Hyaluronan based biodegradable scaffolds for tissue repair |
| US5914381A (en) * | 1996-09-12 | 1999-06-22 | Mitsui Chemicals, Inc. | Degradable polymer and preparation process of the same |
| AU4648697A (en) * | 1996-09-23 | 1998-04-14 | Chandrashekar Pathak | Methods and devices for preparing protein concentrates |
| US5932539A (en) * | 1996-10-15 | 1999-08-03 | The Board Of Trustees Of The University Of Illinois | Biodegradable polymer matrix for tissue repair |
| IT1294797B1 (it) * | 1997-07-28 | 1999-04-15 | Fidia Advanced Biopolymers Srl | Uso dei derivati dell'acido ialuronico nella preparazione di biomateriali aventi attivita' emostatica fisica e tamponante |
| US6824793B1 (en) * | 1998-06-01 | 2004-11-30 | Chiron Corporation | Use of hyaluronic acid polymers for mucosal delivery of vaccine antigens and adjuvants |
| US6191196B1 (en) * | 1999-04-12 | 2001-02-20 | The United States Of America As Represented By The Secretary Of Agriculture | Biodegradable polymer compositions, methods for making same and articles therefrom |
| US7638464B2 (en) * | 1999-04-26 | 2009-12-29 | Biocept, Inc. | Three dimensional format biochips |
| WO2001042367A1 (fr) * | 1999-12-08 | 2001-06-14 | National Institute Of Advanced Industrial Science And Technology | Compositions a base de resine biodegradable |
| US6517933B1 (en) * | 2000-01-18 | 2003-02-11 | Nano-Tex, Llc | Hybrid polymer materials |
| KR100721752B1 (ko) * | 2000-01-24 | 2007-05-25 | 쿠라레 메디카루 가부시키가이샤 | 수팽윤성 고분자 겔 및 그 제조법 |
| JP4753525B2 (ja) * | 2000-07-03 | 2011-08-24 | 伸彦 由井 | 組織再生用基材、移植用材料及びその製法 |
| JP3958536B2 (ja) * | 2000-07-12 | 2007-08-15 | 大日精化工業株式会社 | 水性溶液組成物および物品の表面改質方法 |
| US6586493B1 (en) * | 2001-03-07 | 2003-07-01 | Arizona Board Of Regents Arizona State University | Polysaccharide-based hydrogels and pre-gel blends for the same |
| US7265085B2 (en) * | 2001-10-10 | 2007-09-04 | Neose Technologies, Inc. | Glycoconjugation methods and proteins/peptides produced by the methods |
| AU2003205278B2 (en) * | 2002-01-18 | 2008-08-07 | Psivida Us Inc. | Polymeric gel system for the controlled delivery of codrugs |
| US7138105B2 (en) * | 2002-02-27 | 2006-11-21 | Pharmain | Compositions for delivery of therapeutics and other materials, and methods of making and using the same |
| KR100507545B1 (ko) * | 2002-09-03 | 2005-08-09 | 주식회사 엘지생명과학 | 히알루론산 유도체 및 그의 제조방법 |
| US7090745B2 (en) * | 2002-09-13 | 2006-08-15 | University Of Pittsburgh | Method for increasing the strength of a cellulosic product |
| US6800712B2 (en) * | 2002-10-07 | 2004-10-05 | Steven William Doane | Starch graft copolymers and method of making and using starch graft copolymers for agriculture |
| AU2003294258B2 (en) * | 2003-01-16 | 2009-05-28 | Cornell Research Foundation, Inc. | Partially biodegradable temperature and pH sensitive hydrogel |
| JP2007519756A (ja) * | 2004-01-30 | 2007-07-19 | アンジオテック インターナショナル アーゲー | 拘縮を治療するための組成物および方法 |
-
2004
- 2004-07-27 EP EP04748165A patent/EP1659143A4/en not_active Withdrawn
- 2004-07-27 WO PCT/JP2004/011006 patent/WO2005010080A1/ja active Application Filing
- 2004-07-27 US US10/565,019 patent/US20060235114A1/en not_active Abandoned
- 2004-07-27 JP JP2005512109A patent/JP4500263B2/ja not_active Expired - Fee Related
- 2004-07-27 TW TW093122445A patent/TW200510012A/zh unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07278203A (ja) * | 1993-11-03 | 1995-10-24 | Collagn Corp | グリコサミノグリカン−合成ポリマー結合体 |
| WO2002044276A2 (en) * | 2000-11-28 | 2002-06-06 | Focal, Inc. | Polyalkylene glycol viscosity-enhancing polymeric formulations |
| WO2003028781A1 (fr) * | 2001-08-21 | 2003-04-10 | Japan Science And Technology Agency | Complexe polycation-glycosaminoglycane reticule par un agent de reticulation polyfonctionnel et procede de production associe |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP1659143A4 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010539271A (ja) * | 2007-09-11 | 2010-12-16 | サントル、ナショナール、ド、ラ、ルシェルシュ、シアンティフィク、(セーエヌエルエス) | キトサンの官能化に使用できる感熱性((ポリエチレンオキシド)ポリ(プロピレンオキシド))誘導体の製造方法 |
| JP2010046442A (ja) * | 2008-08-25 | 2010-03-04 | Olympus Corp | 膨隆液組成物および内視鏡装置 |
| JP2016141769A (ja) * | 2015-02-04 | 2016-08-08 | 帝人株式会社 | ヒアルロン酸誘導体および医療用製剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1659143A4 (en) | 2008-10-08 |
| TW200510012A (en) | 2005-03-16 |
| JPWO2005010080A1 (ja) | 2006-09-07 |
| JP4500263B2 (ja) | 2010-07-14 |
| EP1659143A1 (en) | 2006-05-24 |
| US20060235114A1 (en) | 2006-10-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20220008612A1 (en) | Preparation and/or formulation of proteins cross-linked with polysaccharides | |
| US7226972B2 (en) | Process for cross-linking hyaluronic acid to polymers | |
| KR101597333B1 (ko) | 생분해성 단일-상 점착성 히드로겔 | |
| Tang et al. | A review on recent advances of Protein-Polymer hydrogels | |
| EP1773399B1 (en) | Hydrogels of hyaluronic acid and alpha, beta-polyaspartylhydrazide and their biomedical and pharmaceutical uses | |
| WO2005010080A1 (ja) | 温度応答性ハイドロゲル | |
| WO2012120138A1 (en) | Implantable swellable bio-resorbable polymer | |
| US20050020719A1 (en) | Environment responsive gelling copolymer | |
| WO2010119994A1 (ja) | 多糖類誘導体およびそのハイドロゲル | |
| JP2016515821A (ja) | ヒアルロン酸の製造方法及び前記製造方法で製造されたヒアルロン酸を含む癒着防止用組成物 | |
| CA2402546C (en) | Environment responsive gelling copolymer | |
| KR20040069051A (ko) | 온도 감응성을 갖는 다당류 공중합체 및 이의 제조방법 | |
| JP2007002063A (ja) | カルボキシメチルセルロース化合物 | |
| CN113906055B (zh) | 官能化的透明质酸的交联的聚合物及其在炎症状态的治疗中的用途 | |
| EP3986491B1 (en) | Crosslinkable hydrogel compositions | |
| JP2005239860A (ja) | 温度応答性ハイドロゲル | |
| Farhan et al. | Synthesis and characterization of cellulose grafted maleic anhydride and substituted it with amoxicillin | |
| Garner et al. | Types and Chemistry of Synthetic Hydrogels | |
| KR20220067417A (ko) | 가교 폴리감마글루탐산 하이드로겔의 제조방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005512109 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006235114 Country of ref document: US Ref document number: 10565019 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004748165 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004748165 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 10565019 Country of ref document: US |