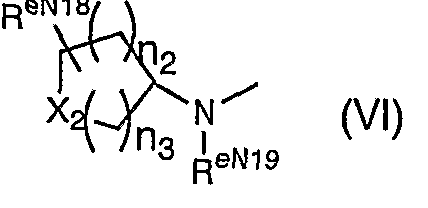WO2004011661A1 - 新規生理活性物質 - Google Patents
新規生理活性物質 Download PDFInfo
- Publication number
- WO2004011661A1 WO2004011661A1 PCT/JP2003/009753 JP0309753W WO2004011661A1 WO 2004011661 A1 WO2004011661 A1 WO 2004011661A1 JP 0309753 W JP0309753 W JP 0309753W WO 2004011661 A1 WO2004011661 A1 WO 2004011661A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- substituent
- membered
- compound
- hydrogen atom
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D407/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00
- C07D407/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00 containing two hetero rings
- C07D407/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00 containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/14—Vasoprotectives; Antihaemorrhoidals; Drugs for varicose therapy; Capillary stabilisers
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D313/00—Heterocyclic compounds containing rings of more than six members having one oxygen atom as the only ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P17/00—Preparation of heterocyclic carbon compounds with only O, N, S, Se or Te as ring hetero atoms
- C12P17/02—Oxygen as only ring hetero atoms
- C12P17/08—Oxygen as only ring hetero atoms containing a hetero ring of at least seven ring members, e.g. zearalenone, macrolide aglycons
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P17/00—Preparation of heterocyclic carbon compounds with only O, N, S, Se or Te as ring hetero atoms
- C12P17/16—Preparation of heterocyclic carbon compounds with only O, N, S, Se or Te as ring hetero atoms containing two or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P17/00—Preparation of heterocyclic carbon compounds with only O, N, S, Se or Te as ring hetero atoms
- C12P17/16—Preparation of heterocyclic carbon compounds with only O, N, S, Se or Te as ring hetero atoms containing two or more hetero rings
- C12P17/162—Heterorings having oxygen atoms as the only ring heteroatoms, e.g. Lasalocid
Definitions
- the present invention relates to a 12-membered ring macrolide compound useful as a medicament, a method for producing the compound, and a use thereof.
- canceration of normal cells is caused by mutation of a gene in the cell and expression of a gene different from normal. Therefore, the present inventors have proposed a cancer cell That the change in gene expression can lead to suppression of the growth of cancer cells, for example, by changing the expression of the cancer gene's tumor suppressor gene, or by changing the expression of genes involved in the cell cycle. Intensive research was conducted based on the inference that it would be possible to suppress the growth of cancer cells.
- the present inventors have developed a compound that causes a change in gene expression, in particular, a compound that suppresses VEGF (Vascular Endothelial Growth Factor) production in hypoxia, suppresses angiogenesis due to cancer,
- VEGF Vascular Endothelial Growth Factor
- fermentation products of various microorganisms and their derivatives were screened using VEGF production of hypoxia-stimulated U251 cells as an index.
- the present inventors have proposed a novel bioactive substance, a 12-membered macrolide system, that suppresses VEGF production under hypoxic conditions in vitro and further suppresses the growth of solid tumor cells in vivo.
- Compound 111 and its analogs were found.
- 6-deoxy111107D in which the hydroxyl group at position 6 of 11107D became hydrogen was used.
- 6-dexoxy 11107D derivatives are found to be stable in aqueous solution, and these derivatives inherit the stable properties of 1U07D
- 6-dexoxy 11107D derivatives inherit the stable properties of 1U07D
- the growth of solid tumor cells was more strongly suppressed in in vivo experiments, and completed the present invention.
- FD-895 In vitro cytotoxic activity has been disclosed for vesicles, L-1210 mouse leukemia cells and HL-60 human leukemia cells (Publication No. 6 ⁇ , Table 2). However, FD-895 had ineffective antitumor activity in in vivo experiments using P388 mouse leukemia cells. (Seki-Asano M. et al, J. Antibiotics, 47, 1395-1401, 1994) 0 Further, as described later, FD-895 is unstable in aqueous solution, Because it is expected that it will be difficult to mix it with an infusion at the time of administration, it cannot be said that it has sufficient qualities as an antitumor agent.
- R 3 , R 7 , R 16 , R 17 , R 2Q , R 21 and R 21 ′ are the same or different
- R C ( Y) ⁇ — [where ⁇ is an oxygen or sulfur atom, R is a) a hydrogen atom,
- i) may have a substituent unsaturated C 2. 22 alkoxy group
- R N1 and R N2 are the same or different
- n represents a 3- to 14-membered non-aromatic heterocyclic group which may have a substituent
- f) may be substituted 5 to terrorist ⁇ reel O dimethylvinylsiloxy groups to 1 4-membered ring, g) may have a substituent group C 7 _ 22 7 Rarukiruokishi group or
- h represents a 5- to 14-membered heteroalkyloxy group which may have a substituent
- W has the meaning described above, and R 3a , R 7a , R 16a , R 17a , R 20a , R 21a and R 21a ′ are the same or different,
- a 5- to 14-membered heteroalkyl group which may have a substituent h) an alkoxy group which may have a substituent, i) may have a substituent unsaturated C 2 - 22 alkoxy group, j) may have a substituent group c 6-14 Ariruokishi group,
- R aN1 and R aN2 are the same or different
- ⁇ may have a substituent ⁇ 6-14 aryl group
- g) may have a substituent group 5 to Teroariru group to 1 4-membered ring, h) which may have a substituent C 7 _ 22 Ararukiru group,
- n represents a 3- to 4-membered non-aromatic heterocyclic group which may have a substituent.
- R 3b , R 7b , R 16b , R 17b , R 20b , R 21b and R 21 ′ b are the same or different,
- b) may have a substituent unsaturated C 2 - 22 alkyl group
- c) may have a substituent group C 7 - 22 Ararukiru group
- d) may have a substituent group 5 to Teroararukiru group to 1 4-membered ring, e) which may have a substituent C 6. 14 Ariruokishi group,
- g represents a 3- to 14-membered non-aromatic heterocyclic group which may have a substituent], 5) R bS1 R bS2 R bS3 S i 0— [where R bS1 , R bS2 and R bS3 are the same or different, a) an alkyl group or
- R bN1 and R bN2 are the same or different
- RbN1 and RbN2 are formed together with the nitrogen atom to be bonded together by a 3- to 14-membered non-aromatic heterocyclic group (the 3- to 14-membered non-aromatic heterocyclic group is substituted) May have a group),
- e represents a 3- to 4-membered non-aromatic heterocyclic group which may have a substituent.
- R 3c , R 7e , R 16c , R I7c , and R 2 . c , R 21e and R 21 ' c are the same or different,
- R cN1 and R eN2 are the same or different
- d) may have a substituent group 3 - 14 cycloalkyl group, or
- e represents a 3- to 4-membered non-aromatic heterocyclic group which may have a substituent.
- 3) may have a substituent unsaturated C 2 - 22 alkoxy group, 3 009753) an optionally substituted C 7 _ 22 aralkyloxy group,
- e) may have a substituent group 5 to Teroariru group to 1 4-membered ring
- f) may have a substituent group C 7 - 22 Ararukiru group
- i) may have a substituent unsaturated C 2 _ 22 alkoxy group
- k represents a 5- to 14-membered heteroaryloxy group which may have a substituent
- e) may have a substituent group 5 to Teroariru group to 1 4-membered ring
- f) may have a substituent group C 7 - 22 Ararukiru group
- g) may have a substituent group 5 to 1 4-membered Teroararukiru group to ring, h) which may have a substituent C 3 - 14 cycloalkyl group,
- R dN1 and R dN2 gar cord Represents a 3 to 14-membered non-aromatic heterocyclic group together with the nitrogen atom to be bonded (the 3- to 14-membered non-aromatic heterocyclic group may have a substituent) ], R 7d and R 21d are the same or different,
- b) may have a substituent group C 6 - 14 7 aryl group
- f) may be substituted 5 to terrorist ⁇ reel O dimethylvinylsiloxy groups to 1 4-membered ring, g) may have a substituent group C 7 - 22 Ararukiruokishi group or
- h represents a 5- to 14-membered heteroalkyloxy group which may have a substituent
- R N1 and R N2 have the above-mentioned meanings]
- R 3e , R 16e and R 21e are the same or different,
- d) may have a substituent group 5 to Teroariru group to 1 4-membered ring, e) which may have a substituent C 7 - 10 Ararukiru group, which may have a 'f) substituent Good 5- to 14-membered heteroaralkyl group, g) optionally substituted 3- to 4-membered non-aromatic heterocyclic group, h) Formula ( ⁇ )
- n is an integer from 0 to 4,
- R eN1 is
- Hydrogen freshness is ii) an alkyl group which may have a substituent
- R eN3 and R eN4 are the same or different
- iii) which may have a substituent unsaturated c 2 _ 10 alkyl group, iv) may have a substituent group C 6 - 14 7 aryl group,
- vi) may have a substituent group C 7 - 10 Ararukiru group
- X e , n, R eN3 , R eN4 and R eN5 represent a group as defined above, and R eN1 and R eN2 together form a 5- to 14-membered non-aromatic heterocyclic group (the 5- to 14-membered non-aromatic heterocyclic group may have a substituent),
- X e , n, R eN2 , R eN4 and R eN5 represent a group as defined above, and R eN1 and R eN3 together form a 5- to 14-membered non-aromatic heterocyclic group (the 5- to 14-membered non-aromatic heterocyclic group may have a substituent) or
- X e , n, R eN1 , R eN4 and R eN5 represent a group as defined above, and R eN2 and R eN3 together form a 5- to 14-membered non-aromatic heterocyclic group (the A 5- to 14-membered non-aromatic heterocyclic group may have a substituent)] or
- R eN8 and R eN9 are the same or different
- v) represents which may have a substituent C 7 _ 10 Ararukiru group or vi) a substituted 5 to may have a group 14-membered ring to Teroararu kill group]]]]]]]
- n is an integer from 0 to 4,
- R eNK and R eN11 are the same or different
- R eN12 and R eN13 are the same or different
- 3) may have a substituent unsaturated c 2-K) alkyl group
- 4) may have a substituent group C 6 - 14 7 aryl group
- a 5- to 14-membered non-aromatic heterocyclic group (the 5- to 14-membered non-aromatic heterocyclic group may have a substituent) or
- a 5- to 14-membered non-aromatic heterocyclic group (representing the 5- to 14-membered non-aromatic heterocyclic group which may have a substituent), or
- NR eN14 [wherein NR eN14 has the meaning described above], or a pharmaceutically acceptable salt or a hydrate of the compound or the pharmaceutically acceptable salt thereof according to 5.
- n is an integer from 0 to 6
- Morpholine-4-yl group which may have a substituent or
- 7) represents a piperazine-11-yl group which may have a substituent]], a compound represented by the formula: or a pharmacologically acceptable salt thereof or a hydrate thereof ;
- n 2 and n 3 are the same or different and are each an integer of 0 to 4,
- X 2 is One CHR eN21- ,
- R eN19 is
- R eN20 is a hydrogen atom
- R 7e and / or R 21e is R e5 CO— 0— [where: R e5 is a group of formula (VII) (VII)
- n 4 is 1 or 2
- R eN25 is
- R 7E and Z or R 21E are R E6 CO— ⁇ [where R E6 is of the formula (VIII)
- n 2 and n 3 are the same or different and are each an integer of 0 to 4,
- R eN31 and R eN32 are the same or different and represent a hydrogen atom or an alkyl group which may have a substituent, or are taken together with a bonding nitrogen atom.
- R eN31 and R eN32 are the same or different and represent a hydrogen atom or an alkyl group which may have a substituent, or are taken together with a bonding nitrogen atom.
- R eN30 is
- R 7e and / or R 21e is R e7 CO— 0— [where R e7 is of the formula (IX)
- ⁇ 5 is an integer of 1 to 3
- R eN33 is 1) amino group
- Morpholine which may have a substituent-representing a 41-yl group]], the compound according to 5. or a pharmaceutically acceptable salt thereof or a hydrate thereof;
- R 7e and / or R 21e is R e8 CO— 0— [where R e8 is of the formula (X)
- n 5 is an integer of 1 to 3
- R eN34 is
- n 5 is an integer of 1 to 3
- R 7E and / or R 21E is R EL0 C ⁇ — O— [where R EL0 is of the formula (XII)
- n 5 is an integer from 1 to 3
- R 7e and / or R 21e is R ell CO— 0— [where R el1 is a group represented by the formula (XIII)
- n 5 represents an integer of 1 to 3, n 5 represents 2 or 3]], or a pharmaceutically acceptable salt thereof or a hydrate thereof;
- R 7e and / or R 21e is R el2 CO—0— [where R el2 may have a substituent on the ring, or
- W is a compound in which R, R 3 and R 21 are hydroxyl groups, R 7 is an acetoxyl group, R 16 , R 17 , R 20 and R 21 ′ are hydrogen atoms; H ⁇ , ⁇
- R 3 R 16, R M and R 21 are hydroxyl
- R 7 is Asetokishi group
- compounds R 17 and R 21 ' is a hydrogen atom
- 7 W is a compound in which / ⁇ , R 3 , R 17 , 6 and R 21 are a hydroxyl group, is an acetyl group, R 20 and R 21 ′ are a hydrogen atom or
- a medicament comprising, as an active ingredient, the compound according to any of 1. to 24., or a pharmacologically acceptable salt thereof or a hydrate thereof;
- a pharmaceutical composition comprising, as an active ingredient, the compound according to any of 1. to 24., or a pharmacologically acceptable salt thereof, or a hydrate thereof;
- the medicament according to 25 as a therapeutic agent for retinal angiogenesis or a therapeutic agent for diabetic retinopathy; 35.
- the medicament according to 25. as a therapeutic agent for an inflammatory disease;
- the medicament according to 25 ⁇ as a therapeutic agent for an inflammatory disease comprising osteoarthritis, rheumatoid arthritis, psoriasis, or a delayed hypersensitivity reaction;
- the medicament according to 25 as a therapeutic agent for atherosclerosis.
- the solid tumor is lung cancer, brain tumor cell, breast cancer, prostate cancer, ovarian cancer cell, colon cancer or melanoma;
- a method for preventing and treating diseases for which gene expression control is effective by administering to a patient a pharmacologically effective amount of the drug described in 4.2.5.
- a method for preventing and treating a disease for which a VEGF production inhibitory effect is effective by administering to a patient a pharmacologically effective amount of the drug described in 25. 5.
- a method for preventing and treating a disease for which angiogenesis inhibitory effect is effective by administering to a patient a pharmacologically effective amount of the drug described in 25.
- 1 W is, Z ⁇ , R 3 and R 21 represents a hydroxyl group, R 7 is Asetokishi group, R 16, R l7, R and R 21 'is hydrogen compound or
- R 21 represents a hydroxyl group
- R 7 is Asetokishi group
- R 21 ' is hydrogen atom compound
- a microorganism having the ability to produce dexamethasone is cultured, and the above-mentioned compound (1) or (2) (hereinafter referred to as the 6-doxyl 11107 compound) is collected from the culture, and the 6-doxyl 11 is characterized in that Method for producing 107 compound;
- R 17, R M and R 21 ' is hydrogen atom (hereinafter, referred to as 6-Dokishi 1 1 10 7 B.) Of 3 W is, ⁇ R 3, R 16 and R '21 is hydroxyl, R 7 is Asetokishi group, R 17, R 20 and R 21' compound is a hydrogen atom,
- R zl and R 21 ' form a connexion Okiso groups together with the carbon bonded
- R 3 R 16 and R 2FL is hydroxyl
- R 7 is Asetokishi group
- compounds R 17 is a hydrogen atom
- 5W is a compound in which Z ⁇ , R 3 , R 16 , R 2Q and R 21 are a hydroxyl group, R 7 is an acetyloxy group, R 17 and R 21 ′ are a hydrogen atom,
- R 7W is a compound in which, R 3 , R 17 , R 16 and R 21 are a hydroxyl group, R 7 is an acetyloxy group, R 20 and R 21 ′ are a hydrogen atom and
- a method comprising:
- microorganism belonging to the bacterium is strain A-1544 (FERM BP-8446) or strain A-1545 (FERM BP-8447).
- the chemical formula of the compound according to the present invention is described as a planar chemical formula for convenience, but may include certain isomers derived from the chemical formula.
- the present invention includes all geometric isomers, optical isomers based on asymmetric carbon, rotational isomers, stereoisomers, tautomers, etc., and isomer mixtures present in the structure of the compound
- the present invention is not limited to the description of the chemical formula for convenience, and may include any one of the above isomers and a mixture thereof. Therefore, when the compound of the present invention has an asymmetric carbon atom in the molecule, an optically active compound and a racemic compound exist, and both are included in the present invention.
- polymorphism may exist, but is not limited to one kind of crystal form in the same manner.Either single crystal form or a mixture of plural crystal forms may be used. Good.
- the compound of the formula (I) of the present invention or a salt thereof may be an anhydride or a hydrate, and both are included in the present invention. Metabolites formed by decomposing the compound of the formula (I) of the present invention in vivo and prodrugs of the compound of the formula (I) or a salt thereof of the present invention are also encompassed by the present invention.
- halogen atom means a fluorine atom, a chlorine atom, a bromine atom, an iodine atom, and is preferably, for example, a fluorine atom, a chlorine atom, a bromine atom, etc., and among them, for example, a fluorine atom, a chlorine atom, etc. are preferred. .
- _22 alkyl group refers to a linear or branched alkyl group having 1 to 22 carbon atoms, such as a methyl group, an ethyl group, an n-propyl group, an iso-propyl group.
- saturated C 2 _22 alkyl group refers to a linear or branched alkenyl group having 2 to 22 carbon atoms, or a linear or branched alkenyl group having 2 to 22 carbon atoms.
- Expressor 5 means an aromatic hydrocarbon cyclic group composed of one, monocyclic group, bicyclic group, Also included are condensed rings such as tricyclic groups.
- phenyl, indenyl, 1-naphthyl, 2-naphthyl, azulenyl, heptenylenyl, indacenyl, acenaphthyl, full Examples include an olenyl group, a phenalenyl group, a phenanthrenyl group, an anthracenyl group and the like, and preferably, for example, a phenyl group, a 1-naphthyl group, a 2-naphthyl group and the like.
- 5- to 14-membered heteroaryl group refers to a monocyclic, bicyclic or heterocyclic group containing at least one heteroatom selected from the group consisting of a nitrogen atom, a sulfur atom and an oxygen atom.
- a tricyclic 5- to 14-membered aromatic heterocyclic group refers to a monocyclic, bicyclic or heterocyclic group containing at least one heteroatom selected from the group consisting of a nitrogen atom, a sulfur atom and an oxygen atom.
- Preferable examples include nitrogen-containing aromatic heterocyclic groups such as pyrrolyl, pyridyl, pyridaziel, pyrimidinyl, pyrazinyl, triazolyl, tetrazolyl, benzotriazolyl, pyrazolyl, Imidazolyl group, benzimidazolyl group, indolyl group, isoindolyl group, indolizinyl group, purinyl group, indazolyl group, quinolyl group, isoquinolyl group, quinolidyl group, phthalazyl group, naphthyridinyl group, quinoxalyl group, quinazolinyl group, cinnolinyl group, ptinomilinyl group Zotriazinyl group, pyrazinopyridazinyl group, acridinyl group, phenanthridinyl group, carpazolyl group, carbazolinyl group
- Preferable examples include, for example, aziridinyl group, azetidyl group, pyrrolidini Group, pyrrolyl group, piperidinyl group, piperazinyl group, homopiperidinyl group, homopiperazinyl group, imidazolyl group, birazolidyl group, imidazolidyl group, morpholinyl group, thiomorpholinyl group, imidazolyl group, oxazolinyl group, 2,5-diazabiaza 2.1] heptyl group, 2,5-diazabicyclo [2.2.2] octyl group, 3,8-diazabicyclo [3.2.1] octyl group, 1,4 diazabicyclo [4..3.0] A nonyl group, a quinuclidyl group, a tetrahydrofuranyl group, a tetrahydrothiophenyl group, and the like.
- the non-aromatic heterocyclic group also includes a group derived from a pyridone ring and a non-aromatic condensed ring (for example, a group derived from a phthalimide ring, a succinimide ring, and the like).
- C 6 substituted moiety is defined - means 14 Ariru group "in the substituted group Specific examples thereof include a benzyl group, a phenyl group, a 3-phenylpropyl group, a 4-phenylbutyl group, a 1-naphthylmethyl group, and a 21-naphthylmethyl group, and preferably have 7 to 10 carbon atoms.
- Aralkyl groups such as a benzyl group and a phenethyl group.
- the term "5- to 14-membered heteroalkyl group” refers to an rC ⁇ alkyl group as defined above, wherein the substitutable moiety is a "5- to 14-membered heteroaryl group” as defined above. It means a substituted group, and specifically includes, for example, a phenylmethyl group, a furylmethyl group, a pyridylmethyl group, a pyridazylmethyl group, a pyrimidylmethyl group, a pyrazylmethyl group, and the like, and preferably, for example, a chenylmethyl group, a furylmethyl group And a pyridylmethyl group.
- C 3 _14 cycloalkyl group refers to a cycloalkyl group composed of 3 to 14 carbon atoms, and preferable groups include, for example, a cyclobutylpyr group and a cyclobutyl group. , Cyclopentyl, cyclohexyl, cyclohepti
- Examples include a mouth hexyl group, a cycloheptyl group, and a cyclooctyl group.
- c cycloalkylalkyl group is defined as above.
- a substituted moiety is the above-defined "C 3 _ ir Nkuroaruki And specifically substituted groups such as cyclopropylmethyl group, cyclobutylmethyl group, cyclopentylmethyl group, cyclohexylmethyl group, cycloheptylmethyl group, cyclooctylmethyl group, and the like. Is, for example, a cyclopropylmethyl group, a cyclobutylmethyl group, a cyclopentylmethyl group or the like.
- C 22 alkoxy group used in the specification of the present application means a group in which an oxygen atom is bonded to the terminal of the “alkyl group” defined above, and preferable groups include, for example, a methoxy group, Ethoxy, n-propoxy, iso-propoxy, n-butoxy, iso-butoxy, sec-butoxy, tert-butoxy, n-pentyloxy, iso-pentyloxy, sec-pentyloxy, n- Hexoxy group, iso-hexoxy group, 1,1-dimethylpropyloxy group, 1,2-dimethylpropoxy group, 2,2-dimethylpropyloxy group, 1-ethyl-12-methylpropoxy group, 1, 1,2-trimethylpropoxy group, 1,2,2-trimethylpropoxy group, 1,1-dimethylbutoxy group, 1,2-dimethylbutoxy group, 2,2-dimethylbutoxy group, 2,3- Examples include a dimethylbutyloxy group, a 1,3-di
- unsaturated C 2 _ 22 alkoxy group the “non-saturation C 2 _ 22 alkyl group” defined above, it refers to a group having an oxygen atom attached to its end.
- Suitable groups include, for example, vinyloxy group, aryloxy group, 1-propenyloxy group, isopropenyloxy group, 2-methyl-1-propenyloxy group, 2-methyl-2-propenyloxy group, 1-butenyloxy group , 2-butenyloxy, 3-butenyloxy, 11-pentenyloxy, 1-hexenyloxy, 1,3_hexayne Lord 3 ⁇ 4_, one-
- Examples thereof include a 2-butynyloxy group and the like, preferably an aryloxy group, a propargyloxy group, a 2-butynyloxy group and the like.
- aryl group means a group having an oxygen atom bonded to its terminal, and specifically includes, for example, phenyloxy, indenyloxy, 1-naphthyloxy, 2-naphthyloxy, azulenyloxy, heptylenyloxy, indenyl A senyloxy group, an acenaphthyloxy group, a fluorenyloxy group, a phenalenyloxy group, a phenanthrenyloxy group, an anthracenyloxy group, and the like, preferably a phenyloxy group, And a naphthyloxy group and a 2-naphthyloxy group.
- C 7 one 22 Ararukiruokishi group means a group having an oxygen atom attached to its end, specifically, for example, base Njiruokishi Group, phenethyloxy group, 3-phenylpropyloxy group, 4-phenylbutyloxy group, 11-naphthylmethyloxy group, 2-naphthylmethyloxy group and the like, and preferably benzyloxy group and the like.
- the term "5- to 14-membered heteroalkyloxy group” refers to a group in which an oxygen atom is bonded to the terminal of the "5- to 14-membered heteroalkyl group" defined above. It means, specifically, for example, a chenylmethyloxy group, a furylmethyloxy group, a pyridylmethyloxy group, a pyridazylmethyloxy group, a pyrimidylmethyloxy group, a pyrazylmethyloxy group, and the like. Is, for example, a phenylmethyloxy group, a furylmethyloxy group, a pyridylmethyloxy group or the like.
- ⁇ 5- to 14-membered heteroaryloxy group '' means a group in which an oxygen atom is bonded to the terminal of the ⁇ 5- to 14-membered heteroaryl group '' defined above, Specifically, for example, a pyrrolyloxy group, a pyridyloxy group, a pyridazinyloxy group, a pyrimidinyloxy group, a pyrazinyloxy group, a triazolyloxy group, a tetrazolyloxy group, a benzotriazolyloxy group, a virazolyloxy group, an imidazolyloxy group Xy group, benzimidazolyloxy group, indoloxy group, isoindoloxy group, indolizinyloxy group, prynyloxy group, inlio _ ⁇ 3 ⁇ 4,-key group: ⁇ ⁇ ⁇ "Jj_Leo , Is a main
- Ruokishi group Shin quinolinyl O alkoxy group, flop Teri Gini Ruo alkoxy group, Imidazotori Ajiniruokishi group, pyrazinopyridazinyl pyridazinyloxy Ruo alkoxy group, ⁇ chestnut Gini Ruo alkoxy group, Hue Nantridinyloxy group, carbazolyloxy group, rivazolinyloxy group, perimidinyloxy group, phenanthine linyloxy group, phenacinyloxy group, imidazopyridinyloxy group, imidazopyrimidinyloxy group, pyrazo Mouth pyridinyloxy group, pyrazo-mouth pyridinyloxy group, chenyloxy group, benzochenyloxy group, furyloxy group, pyranyloxy group, cyclopentaviranyloxy group, benzofuryloxy group, isobenzofuryloxy group, thiazolyloxy
- aliphatic c 2 - 22 Ashiru group refers to "Ji ⁇ ⁇ alkyl group” defined above, in the "unsaturated c 2 _ 22 alkyl group” bonded carbonyl group in the terminal group
- Examples include a naphthoyl group, a picolinoyl group, a nicotinol group, an isonicotinoyl group, a furoyl group, and a thiophene liponyl group, and preferred examples include a benzoyl group, a picolinol group, a nicotinol group, and an isonicotinol group.
- c ⁇ alkylsulfonyl group means a sulfonyl le radical of the definition "c i_ 22 alkyl group” is bonded, specifically, for example Mechirusuru Honiru group, Echirusuruhoniru group And n-propylsulfonyl group, iso-propylsulfonyl group and the like, and preferably, a methylsulfonyl group and the like.
- C 6 _ 14 ⁇ reel sulfonyl group means a "C 6_ 14 Ariru group” bonded to sulfonyl Le groups defined above, specifically, for example, benzenesulfonic Ruhoniru group, 1 —Naphthylene sulphonyl group, 21-naphthalene sulphonyl group and the like, preferably benzenesulphonyl group and the like.
- aliphatic C 2 _ 22 Ashirokishi group used herein as “aliphatic C 2 _ 22 Ashirokishi group", “aliphatic C 2 - 22 Ashiru group” defined above in, it means a group having an oxygen atom attached to its end, for example, ⁇ Setokishi group And a propionyloxy group, an acryloxy group and the like, and preferably, for example, an acetoxy group and a propionyloxy group.
- c 2 22 alkoxy Cal Poni Le group Used herein - a "c 2 22 alkoxy Cal Poni Le group", the definition of "c, _ 22 alkoxy group” in means a group Karuponiru group is bonded to the terminal, For example a methoxycarbonyl group, E Ethoxycarbonyl, n-propoxycarbonyl, iso-propoxycarbonyl, n-butoxycarbonyl, iso-butoxycarbonyl, sec-butoxycarbonyl, tert-butoxycarbonyl, etc. , Iso-propoxycarbonyl group, tert-butoxycarbonyl group and the like.
- unsaturated C 3 _ 22 alkoxy Cal Poni Le group used herein "unsaturated C 2 _ 22 alkoxy group" defined refers to a group Karuponiru group is bonded at its ends, for example, vinyl Roxycarbonyl group, aryloxycarbonyl group, 1-propenyloxycarbonyl group, isopropenyloxycarbonyl group, propargyloxycarbonyl group, 2-butynyloxycarbonyl group, and the like are preferable.
- Example 1 Re-mouth-power-return-
- C j- 22 alkylthio group the "_ 22 ⁇ alkyl group” defined above, means a group in which a sulfur atom is bonded at its distal end, for example Mechiruchi O group, Echiruchio Group, n-propylthio group, is 0-propylthio group and the like, Preferred are, for example, a methylthio group, an ethylthio group and the like.
- the ⁇ rc 2 alkylsulfinyl group '' used in the present specification means a group in which a sulfinyl group is bonded to the terminal in the above-mentioned ⁇ c 22 alkyl group '', for example, a methylsulfinyl group, an ethylsulfinyl group, ⁇ -propylsulfinyl group, iso-propylsulfinyl group and the like. And preferably a methylsulfinyl group, an ethylsulfiel group and the like.
- C, tolerance 22 alkylsulfonyloxy group refers to a group in which an oxygen atom is bonded to the terminal of the“ rc ⁇ alkylsulfonyl group ”defined above, for example, a methylsulfonyloxy group And an ethylsulfonyloxy group, an n-propylsulfonyloxy group, an iso-propylsulfonyloxy group and the like, and preferably, a methylsulfonyloxy group and the like.
- cycloalkyl group e.g. cyclopropyl group, cyclobutyl group, consequent opening pentyl group, cyclohexyl group, heptyl group cycloalkyl, Shikurookuchiru group
- alkylsulfinyl group (Eg, methylsulfinyl, ethylsulfinyl, n-propylsulfinyl, iso-propylsulfinyl, etc.),
- (33) include one or more groups selected from formyl group, preferred examples ⁇ amino group, C, - 22 alkyl group, unsaturated C 2 - 22 alkyl group, ⁇ 6 _ 14 Ariru group, from 5 1 4-membered ring to Teroariru group, 3 to 14-membered non-aromatic heterocyclic group, a C 3 _ 14 a cycloalkyl group, etc., among them for example amino groups, C] _ 22 alkyl group, 3 to 14-membered ring non Kaoru aromatic heterocyclic group, it is preferred that such C 3 _ 14 consequent opening alkyl group is substituted two from 1.
- the above-mentioned (9) amino group and (31) carbamoyl group as the substituents in “optionally have a substituent” further include 1 to 2 C 2 alkyl groups, unsaturated C 2 It may be substituted by a 2 to 22 alkyl group or a C 6 -i4 aryl group.
- the compound of the formula (I) inhibits VEGF production under hypoxic conditions, furthermore has an inhibitory effect on the growth of solid tumor cells in vivo, and is used in an amount not accompanied by significant weight loss. Although it works effectively, the compounds of the above formula (I-a) are preferred.
- the compound of the formula (I) in which R 16 is a hydroxyl group is a compound having particularly excellent stability in an aqueous solution.
- Compounds of formula (I) have functional groups at positions 3, 7, 16 and 21 (eg, For example, a hydroxyl group) is produced using a known conversion reaction, and therefore, the same substituent can be introduced at the 3-, 7-, 16-, and 21-positions. Due to the structural features of the 7- and 21-side chains, a more preferred group of compounds can be defined as compounds of the formula (I-d).
- Compounds of the formula (I) as well as compounds of the formula (I) which form an oxo group together with the carbon atom to which R 21 is bonded, as well as the compounds of the formula (Id) have a good activity. It is.
- more preferred embodiments of the compounds can be exemplified by the compounds of the invention of the above-mentioned "7.” to "20.”
- Preferred examples of the compound of the formula (I) will be described below.
- Preferred examples of the compound group including the compounds of the examples described below, are, for example, (8E, 12E, 14E) -3, 16, 21—Trihydroxy-6, 10, 12, 16, 20—Pentamethyl-7
- the compound of the formula (I) is a physiologically active substance 6-doxy 11107 compound [the formula (I)
- W is H, R 3 and R 21 are hydroxyl groups, R 7 is an acetooxy group, and R 16 , R I7 , R 20 and R 21 ′ are hydrogen atoms (hereinafter referred to as 6-deoxy 1 11 B) )
- R 21 and R 21 ′ form an oxo group together with the carbon to be bonded
- R 3 , R i6 and R 2fl are a hydroxyl group
- R 7 is an acetyl group
- R 17 is a hydrogen atom
- R 21 is a hydroxyl group
- R 7 is an acetyl group
- R ′ 7 and R 21 ′ are hydrogen atoms
- 6W is / /
- R 3 , R 7 , R 16 and R 21 are hydroxyl groups
- R 17 , R and R 21 ′ are hydrogen atoms
- 8 W is, R 2 ′ and; 21 ′ are bonded to the carbon to be bonded to form an oxo group, R 3 and R 16 are a hydroxyl group, R 7 is an acetoxy group, R 17 and R 2fl are a hydrogen atom (Hereinafter referred to as 6-doxy compound) as a key compound, and can be produced by appropriate chemical modification according to a standard method.
- the 6-deoxy111-107 compound (particularly, 6-deoxy111107B), which is a physiologically active substance of the present invention, which is a 6-deoxy111107D compound, is produced by fermentation using a microorganism. be able to.
- the microorganism used to produce the 6-deoxy 11107 compound can be any microorganism that has the ability to produce the 6-deoxy 11107 compound.
- a soil isolate or a known strain capable of producing 11107B analogs A conventional mutagenesis method using a mutagen such as ultraviolet light or N-methyl-N'-nitro-N-2-nitrosoguanidine (NTG) as a mutagen, or gene disruption by homologous recombination, etc. As a result, a strain producing 6-dexoxy 11107B can be obtained.
- the following deposited strains can be exemplified as microorganisms used to produce the 6-deoxy 11107 compound.
- the strain was deposited internationally with the National Institute of Advanced Industrial Science and Technology (AIST), Tsukuba East 1-chome, Ibaraki Prefecture, Japan I have.
- Streptomyces sp. Mer-11107 was designated as FE M P-18144 as of February 19, 2012 by the Institute of Industrial Science and Technology, 1-3-1 Tsukuba East, Ibaraki Prefecture, 305-8566, Japan.
- the technology was transferred to the International Depository FERM BP-7812 at the Joint Research Institute and the Patent Organism Depositary (IP0D).
- a strain for producing the 6-dexoxy 11107 compound belongs to the genus Streptomyces, and a mutant of these strains as long as it has the ability to produce the 6-dexoxy 11107 compound. There is no particular limitation including, but in addition to the above strains, for example,
- Streptomyces sp. A-1543 and the like can be exemplified. This strain was found on July 23, 2004 at 1-1 -1 Higashi, Tsukuba, Ibaraki, 305-8566 Japan, Japan 1 FE RM at the National Institute of Advanced Industrial Science and Technology (AIST) Deposited as P-1 894 42, and as of July 28, 2003 1-5-1, Higashi, Tsukuba, Ibaraki, Japan 1-chome 1 1 1 1 Transferred to International Deposit FE RM BP—8442 at the National Institute of Advanced Industrial Science and Technology, Patent and Biological Depositary Center (IP0D).
- IP0D National Institute of Advanced Industrial Science and Technology
- any strain belonging to the genus Streptomyces and capable of producing a 6-dexoxy 11107 compound is expected to be usable.
- typical strains include the strain numbered by the present inventors as Mer-11107 strain, A-1543 which is a mutant strain of this strain, and the like.
- the bacteriological properties of these strains are as follows.
- This strain elongates spiral aerial hyphae from the basal hyphae. It forms a spore chain consisting of about 10 to 20 cylindrical spores at the tip of a mature aerial hypha. The size of the spores is about 0.7 X 1. The surface of the spores is smooth and no special organs such as spores, sclerotium, and flagella are found.
- This strain grows well and forms aerial hyphae on its surface to form light gray (d) spores.
- the back of the culture is Light me lon ye l low (3ea). Production of soluble dye is not observed.
- This strain is moderately growing and forms a gray spore (Gray; g) with a slight formation of aerial hyphae on its surface.
- the reverse side of the culture is Nude tan (4 gc) or Putty (1 1/2 ec). No production of soluble dye is observed.
- This strain grows well and forms aerial hyphae on its surface to form gray spores (Gray; e).
- the reverse side of the culture is Fawn (4ig) or Gray (g). No production of soluble dye is observed.
- This strain grows well and forms aerial mycelium on its surface to form white spores (White; a).
- the back of the culture is Pead pink (3ca). No production of soluble dye is observed.
- the following shows the growth status of this strain after culturing at 28 ° C and 2 weeks on a medium obtained by adding various carbon sources to Preedham-Gotrib agar medium.
- the 6-deoxy 11107 compound of the present invention can be produced by inoculating the above strain into a nutrient medium and aerobically culturing it.
- the strain producing 6-dexoxy 1 1 1 1 1 0 7 compounds the present invention is not limited to the above strains, as long as it belongs to the genus Streptomyces and has the ability to produce 6-dexoxy 1 1 1 1 1 7 compounds.
- Available to The method for culturing the above-mentioned microorganisms is basically the same as the method for culturing general microorganisms. However, it is usually preferable to carry out the culture under aerobic conditions such as shaking culture by liquid culture and aeration-agitation culture.
- the medium used for the culture may contain a nutrient source that can be used by microorganisms belonging to the genus Streptomyces, and any of various synthetic, semi-synthetic media, and natural media can be used.
- a nutrient source for example, glucose, sucrose, fructose, glycerin, dextrin, starch, molasses, soybean oil and the like can be used alone or in combination as a carbon source.
- a nitrogen source use an organic nitrogen source such as pharma media, peptone, meat extract, soybean powder, casein, amino acid, yeast extract, urea, etc., or use an inorganic nitrogen source such as sodium nitrate, ammonium sulfate alone or in combination. be able to.
- salts such as sodium chloride, potassium chloride, carbonated calcium sulfate, magnesium sulfate, sodium phosphate, potassium phosphate, and cobalt chloride; heavy metal salts; vitamins such as vitamin B and biotin may be added as necessary. it can. If foaming during culturing is remarkable, use various antifoaming agents as appropriate.
- concentration of JlDlL & fc must be such that it does not affect the eyes. For example, the concentration used is preferably 0.05% or less.
- Culture conditions are appropriately selected within a range where the strain can grow well and produce the above-mentioned substances.
- the culture temperature is usually maintained at 20 to 40 ° C, preferably at 23 to 35 ° C.
- the culture period is about 2 to 8 days, usually about 3 to 5 days.
- the various culture conditions described above can be appropriately changed according to the type and characteristics of the microorganism used, external conditions, and the like, and it goes without saying that the optimum conditions can be selected.
- the 6-deoxy 11107 compound of the present invention accumulated in the culture solution can be recovered by ordinary separation means utilizing its properties, such as a solvent extraction method and an adsorption resin method.
- a separation and purification method generally used for isolating a microbial metabolite from the culture solution can be used.
- organic solvent extraction using methanol, ethanol, butanol, ethyl acetate, orifice, etc. various ion exchange chromatography, gel filtration chromatography using Sephadex LH-20, etc., adsorption with activated carbon, silica gel, etc.
- This includes any known method such as adsorption / desorption treatment by mouth chromatography or thin layer chromatography, or high performance liquid chromatography using a reversed phase column or the like.
- the purification method is not particularly limited to the method shown here.
- the 6-deoxy compound of the present invention is obtained by converting the hydrogen atom at the 16-position of the 6-deoxy 11107 compound (particularly, 6-deoxy111107B) obtained as described above by microbial conversion. It can be obtained by hydroxylation.
- the strain has the ability to hydroxylate the hydrogen atom at the 16-position of the product (particularly, 6-deoxy 11107 7) and convert it to the 6-deoxy 11107 D compound of the present invention, Anything can be used.
- the present inventors A-154 strain and A-154 strain isolated from soil can be mentioned. These strains were obtained from the National Institute of Advanced Industrial Science and Technology (AIST) and the Patent Organism Depositary Center, Chuo No. 6, 1-chome, Tsukuba, Ibaraki, 305-8566, Japan on July 23, 1994.
- the 6-deoxy compound is obtained by bringing the 6-deoxy compound (particularly, 6-deoxy) into contact with the cells or cell preparations of the above strains or mutants thereof. It is manufactured by Usually, a culture solution obtained by inoculating the above strain with a nutrient source-containing medium and aerobically culturing is prepared by suspending the washed cells of the strain in an appropriate buffer. The compound is produced by adding a 6-dexoxy 11107 compound (particularly, 6-dexoxy 1107 ⁇ ) as a raw material compound. Production is performed in a culture medium, and the addition of 6-deoxy 1 11 7 compounds (especially 6-deoxy 1 1 1 1 1 0 7 ⁇ ) to the culture medium has elapsed before or after the start of the culture.
- Culturing of the strain for preparing such a culture solution or washed cells and culturing of the strain with the starting compound added can be performed according to a general microorganism culturing method. For example, it is preferable to carry out under aerobic conditions such as shaking culture by liquid culture and aeration and agitation culture.
- the medium used for the culture may be any medium containing a nutrient source that can be used by microorganisms, and any of various synthetic media, semi-synthetic media, and natural media can be used.
- a medium composition as a carbon source, for example, glucose, galactose, sucrose- ⁇ -! ⁇ ,- ⁇ ⁇ - ⁇ , starch, honey-ta-bean-oil-oil, etc. They can be used alone or in combination.
- Nitrogen sources include, for example, organic media such as pharmacy media, peptone, meat extract, soybean meal, fishmeal, dartene meal, casein, dried yeast, amino acids, yeast extract, urea, sodium nitrate, ammonium sulfate
- Inorganic nitrogen sources such as platinum can be used alone or in combination.
- salts such as sodium chloride, potassium chloride, calcium carbonate, magnesium sulfate, sodium phosphate, potassium phosphate, copper sulfate, iron sulfate, manganese chloride, cobalt chloride, heavy metal salts, vitamins such as vitamin B and biotin.
- clathrates such as cyclodextrins can be added and used as necessary. If foaming during culturing is remarkable, various antifoaming agents can be appropriately added to the medium. When adding an antifoaming agent, it is necessary to use a concentration that does not adversely affect the production of the target substance.
- the culturing conditions are appropriately selected within a range where the strain can grow well and convert the 6-dexoxy 11107 compound (particularly, 6-dexoxy 11107 B) to produce the 6-doxy compound. can do.
- the pH of the medium is preferably 5 to 9, usually around neutral.
- the culture temperature is generally maintained at 20 to 40 ° C, preferably 24 to 30 ° C. Culture days :! ⁇ 8 days, usually 2-5 days.
- the various culture conditions described above can be appropriately changed according to the type and characteristics of the microorganism used, external conditions, and the like, and it goes without saying that the optimum conditions can be selected.
- the 6-dexoxy compound accumulated in the culture solution can be recovered by a usual separation method utilizing its properties, for example, a solvent extraction method, an adsorption resin method and the like.
- the 6-deoxy compound can be collected from the culture using the separation and purification methods generally used to isolate microbial metabolites from the culture.
- the method may be performed according to the method for purifying 107 compounds (especially, 6-doxy 11 11 B), and these methods may be used alone or in combination in any order.
- -Deoxy compounds can be isolated and purified.
- a method for producing the compound of the formula (I) other than the 6-deoxy 11107 compound and the 6-deoxy compound will be described.
- a method for producing a sulfonic acid ester derivative I.
- a method for producing an amine derivative J.
- the introduction and removal of a hydroxyl-protecting group as needed depends on the type and stability of the compound involved in the production. For example, the method described in the literature [Protective group-syn-organic- Synthesis (Protecti ve Group sin Organic Synthesis), written by TW Green (TW Gre en e), John John & Sons, Inc., 3rd edition (3rd Edition)] or It can be performed according to a method corresponding thereto.
- the compound of the formula (I) can be produced by appropriately combining a reaction for introducing / removing a hydroxyl-protecting group and the above-mentioned production method.
- R 3 , R 7 , R 16 , R 17 , R 2D and R 21 are the ft-substituted compounds listed in the above 10), and the compound of the urethane derivative
- the method for producing a thiourethane derivative, the method for producing an amide sulfate, or the method for producing an amine conductor, the compounds of the substituents listed in the above 3) to 6) are produced by the method for producing an ether derivative.
- the compound of the substituent listed in the above 7) is used to prepare the compound of the substituent listed in the above 12) or 13) using the method for producing an ester derivative.
- the compound of the substituent listed in the above 11) is a compound of the substituent listed in the above 9) using a method for manufacturing a sulfate ester or an ester of sulfonic acid, and the compound of the substituent is halogen.
- the compounds of the substituents listed in the above 8) can be produced by a method for producing an oxo form by oxidizing a hydroxyl group in the oxo form described in the above 1) by using a reaction for introducing and removing a protecting group for a hydroxyl group. Can be.
- R 3x , R 16x and R 21x are a hydrogen atom or a protecting group (however, R 3x , R I6x and R do not represent a hydrogen atom at the same time), R 3y , R i6y and R 2h ′ are hydrogen atom or a protecting group or the formula R f O-CO-, a group represented by (wherein, R f is a good C 6 which may have a substituent - shows a 14 Ariru group) (wherein, R 3y , R 16y and R 21y show no simultaneously hydrogen atoms), R 3e, R 16e and R 2k is a hydrogen atom or a protecting group if Kuhashiki R N 'R N2 N- CO-, a group represented by (Where RN1 and R'V2 have the meanings described above) (provided that R3e , R ⁇ and R21e do not simultaneously represent a hydrogen atom).
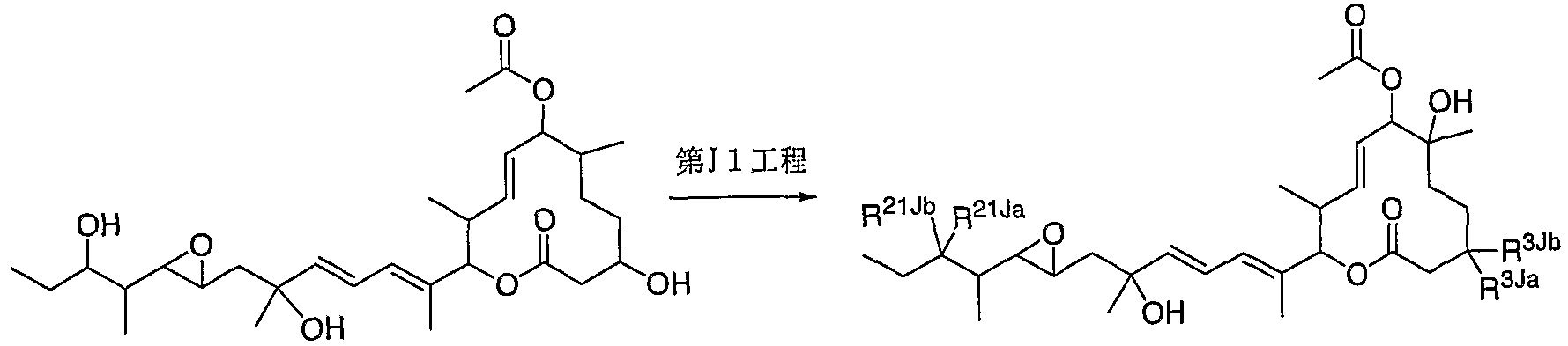
- Step A1 is a step of producing a compound of the formula (IA). This step is attained by protecting the hydroxyl group of 6-doxy 1 11 0 7 D (compound (3) above).
- the reaction for protecting the hydroxyl group depends on the type of the protecting group, but is carried out by a method well known in synthetic organic chemistry.
- each hydroxyl group such as 1-ethoxyhexyl, tetrahydropyranyl, 1-methoxycyclohexyl, 4-methoxytetrahydropyranyl, 4-methoxytetrahydrothiopyranyl, 4-methoxytetrahydrothiopyranyl S, S-dioxide, etc.
- the protected derivative can be synthesized by treating a corresponding vinyl ether, such as ethyl vinyl ether or dihydropyran, with 6-deoxy111107D in the presence of an acid.
- common acids include organic acids such as pyridinium p-toluenesulfonic acid (PPTS), p-toluenesulfonic acid, camphorsulfonic acid, acetic acid, trifluoroacetic acid, and methanesulfonic acid, such as hydrogen chloride, nitric acid, hydrochloric acid, and the like.
- organic acids such as pyridinium p-toluenesulfonic acid (PPTS), p-toluenesulfonic acid, camphorsulfonic acid, acetic acid, trifluoroacetic acid, and methanesulfonic acid, such as hydrogen chloride, nitric acid, hydrochloric acid, and the like.
- Inorganic acids such as sulfuric acid and the like, preferably, for example; -pyridinium toluenesulfonate (PPTS), p-toluenesulfonic acid, camphorsulfonic acid and
- the solvent used in the reaction is not particularly limited, but is preferably an inert solvent that does not easily react with the raw materials, for example, ethers such as tetrahydrofuran, dimethyl ether, diisopropyl ether, dioxane, and dimethyloxetane, for example, dichloromethane, Halogenated hydrocarbons such as chloroform, carbon tetrachloride, and 1,2-dichloroethane, for example, hexane, benzene, toluene, and other hydrocarbons; for example, acetone, methylethyl ketone, and other ketones; Nitriles such as acetonitrile, for example, N, N-dimethylformamide, N, N-dimethylacetoamide, N-methyl-2-pyridone, hexamethylphosphorylamide
- ethers such as tetrahydrofuran, dimethyl ether, diisopropyl ether, dioxan
- dichloromethane, chloroform, tetrahydrofuran and the like are used.
- the reaction time is 10 minutes to 5 days, preferably 1 day to 2 days.
- the reaction temperature ranges from 178 ° C. to hot reflux, preferably room temperature.
- Vinyl ether used for the reaction And acid, respectively, against 6-doxy 1 11 7 D To 200 equivalents and 0.05 to 2 equivalents, preferably 30 to 50 equivalents and 0.1 to 0.3 equivalents, respectively.
- protecting groups include, for example, methoxymethyl, methylthiomethyl, methoxyethoxymethyl, trichloroethoxymethyl, trimethylsilylethyl, trimethylsilylethoxymethyl, tert-butyldimethylsilyl, triethylsilyl, trimethylsilyl, getylisopropylsilyl, Examples include triisopropylsilyl, tert-butyldimethylsilyl, diphenylmethylsilyl, benzyl, p-methoxybenzyl, p-methylpentyl, p-methylbenzyl, p-chlorobenzyl, triphenylmethyl, and the like.
- hydroxyl-protected derivatives can be synthesized by reacting a chloro, bromo or trifluoromethanesulfonyl derivative of each protective group in the presence of a base.
- a base include general organic bases and inorganic bases.
- organic base include imidazole and 4- (N, N-dimethylamino) pyridine
- aromatic bases such as pyridine, 2,6-lutidine and collidine
- N-methylbiperidine Tertiary amines such as N-methylpyrrolidine, triethylamine, trimethylamine, di-isopropylpropylamine, cyclohexyldimethylamine, N-methylmorpholine, 1,8-bis (dimethylamino) naphthalene
- secondary amines such as di-iso-butylamine and dicyclohexylamine
- alkyllithiums such as methyllithium and butyllithium
- metal alkoxides such as sodium methoxide and sodium ethoxide
- inorganic bases such as hydrogen Sodium hydride, potassium hydride, etc.
- Alkali metal hydrides for example, alkaline earth metal hydrides such as calcium hydride, etc., hydroxides of sodium hydroxide, alkali metal such as hydroxide hydroxide, eg potassium carbonate, monocarbonate
- carbonates of LJ metal fittings such as, for example, hydrogen carbonate of sodium metal such as sodium hydrogen carbonate.
- Preferred bases used when protecting the hydroxyl group with a silyl protecting group include, for example, aromatic bases such as imidazole and 4-dimethylaminopyridine, and tertiary amines such as triethylamine. Min.
- the solvent used in the reaction is not particularly limited, but is preferably one that does not easily react with the raw materials.
- the inert solvent examples include, for example, tetrahydrofuran, dichloromethane, N, N-dimethylformamide and the like.
- the reaction time is 10 minutes to 3 days, preferably 1 to 2 days.
- the reaction temperature is from 178 ° C. to the reflux temperature, preferably from 10 to 50 ° C.
- the chloro form, bromo form or trifluoromethanesulfonyl form and the base to be used in the reaction are preferably 1 to 20 equivalents and 0.5 to 30 equivalents, and more preferably 6 to 10-Doxy 111 D, respectively. Is 1 to 15 equivalents and 0.5 to 20 equivalents, respectively.
- reagent used for protecting the hydroxyl group and its equivalent selective protection of the hydroxyl group of 6-dexoxy 11107 D can be achieved.
- selective protection of the hydroxyl group of 6-dexoxy 11107 D can be achieved.
- dichloromethane using chlorotriethylsilane, triethylamine, or 4-dimethylaminopyridine, or in N, N-dimethylformamide using tert-butylchlorodimethylsilane or imidazole at room temperature.
- a compound in which the hydroxyl groups at the 3-, 21-, and 21-positions are selectively protected can be obtained.
- Step A2 is a step for producing a compound of the formula (IIA). This step is achieved by treating the acetoxyl group of the compound of the formula (IA) with a base in an inert solvent and converting it into a hydroxyl group.
- the base used is, for example, an inorganic base such as sodium hydride, potassium hydride or the like, an alkali metal hydride such as calcium hydride, for example, lithium hydroxide, sodium hydroxide, or water.
- Alkali metal water such as lium oxide, etc.-Oxidized L, sprinkled;
- Alkali metal carbonates such as lithium carbonate, sodium carbonate, carbonated lime, etc.
- Alkali metal carbonates such as sodium hydrogen carbonate
- Bicarbonates include, for example, metal alkoxides such as lithium methoxide, sodium methoxide, sodium ethoxide, potassium tert-butoxide and the like.
- Bases such as monia are exemplified.
- Preferred bases include potassium carbonate, guanidine and the like.
- the inert solvent used is, for example, an alcoholic solvent such as methanol, ethanol, isopropanol, or tert-butanol, or water, in addition to the above-mentioned inert solvent. These solvents may be used as a mixture. it can. Preferred solvents include alcoholic solvents and mixtures of alcohols and halogenated solvents.
- the reaction time is 10 minutes to 5 days, preferably 30 minutes to 1 day.
- the reaction temperature is from 1 78 to the reflux temperature, preferably room temperature.
- the base used in the reaction is 1 to 10 equivalents, preferably 2 to 5 equivalents, relative to the compound of the formula (IA).
- Collection A3 is a process for producing a compound of the formula (IIIA). This step is achieved by treating the hydroxyl group of the compound of the formula (IIA) with a chromate formate derivative or heptonyl diimidazole in the presence of a base.
- a chromate formate derivative or heptonyl diimidazole examples include, but are not limited to, 412-trophenylchloroformate, phenylchloroformate, 4-chlorophenylchromate, 4-bromophenylchromate, and 2,4-dinitrophenylchromate. No.
- the base examples include the above-mentioned organic bases and inorganic bases, and preferably, for example, diisopropylethylamine, 4-dimethylaminopyridine, triethylamine, pyridine, 2,6-lilletidine, sodium hydride and the like are used.
- the solvent used in the reaction is not particularly limited, but is preferably one that does not easily react with the starting materials.
- the inert solvent include, for example, tetrahydrofuran, dichloromethane, N, N-dimethylformamide and the like.
- the chromate formate derivative and the base used in the reaction are 1 to 10 equivalents and 1 to 20 equivalents, preferably 1 to 5 equivalents and 1 to 10 equivalents, respectively, relative to the compound of the formula (IIA). It is.
- the reaction time is 10 minutes to 30 hours, preferably 1 to 4 hours.
- the reaction temperature is from 1 78 ° C to the temperature of heating and refluxing, and is preferably ⁇ 1—0— ° & Kara ”5 ⁇ 0 G ⁇ A ⁇ ⁇ —— ⁇
- the hydroxyl group of the compound (IA) is esterified in the step A3. It is also possible to convert to a group. That is, it is converted to a carbonate group. By treating with an equivalent amount of a base corresponding to the number of hydroxyl groups and a formaldehyde derivative, other hydroxyl groups of compound (IA) can be converted to carbonate groups as well as the hydroxyl group at position 7. Can be.
- Step A4 is a step for producing a compound of the formula (IVA).
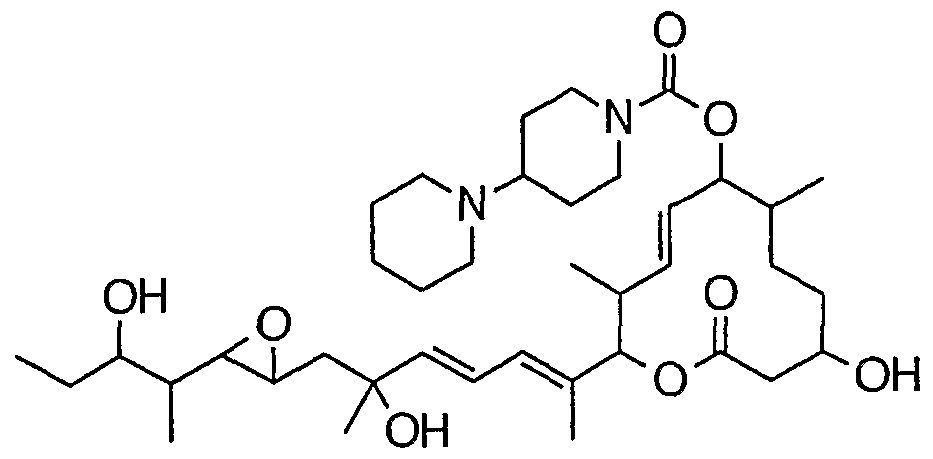
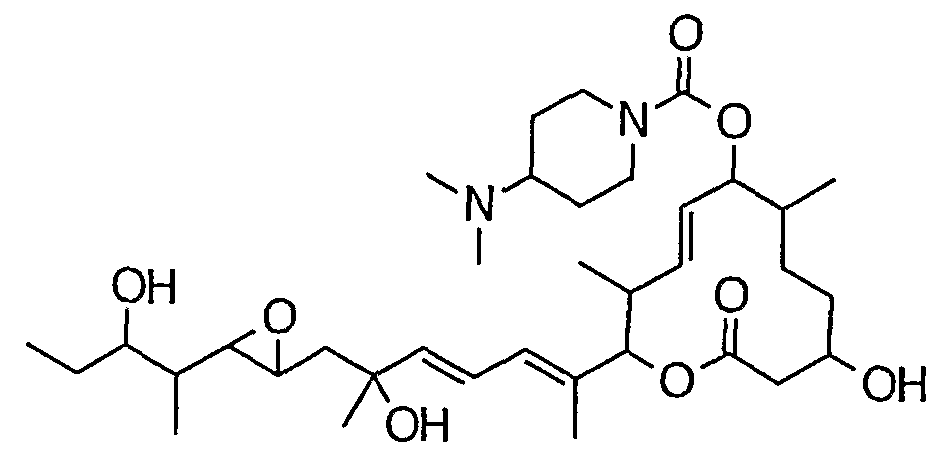
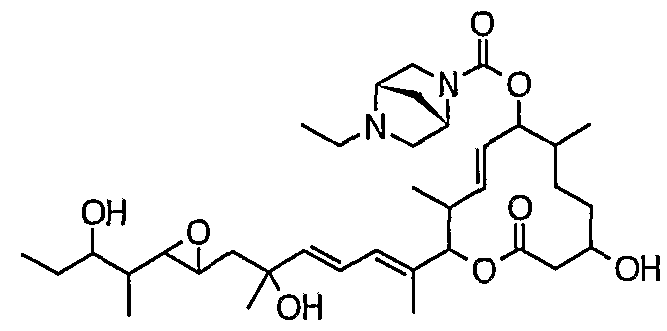
- the carbonate of the formula (IIIA) is treated with an amine capable of forming a desired compound of the formula (I) (R N1 R N2 H) or an amine alone in an inert solvent in the presence of a base. This is achieved by:
- the amines used are, for example, methylamine, ethylamine, propylamine, butylamine, octylamine, decylamine, cyclopropylamine, cyclopentylamine, cyclohexylamine, dimethylamine, getylamine, ethylmethylamine, ethylenediamine, 1,3-propanediamine.
- Giroux N ⁇ '-dimethyl-1,3-propanediamine, morpholine, thiomorpholine, thiomorpholine-S-oxide, thiomorpholine-S, S-dioxide, pyrrolidine, pyridin, piperazine, homopirazine, 4 -Hydroxypi Lysine, 4-methoxypiperidine, 1-methylbiperazine, 1-ethylbiperazine, 1-propylpiperazine, 1-butylbiperazine, 1-isopropylpiperazine, 1-cyclobutylpiperazine, 1-cyclopentylbiperazine, 1 —Cyclohexyl piperazine, 1-cycloheptyl biperazine, 1-cyclooctyl piperazine, 1- (cyclopropylmethyl) piperazine, 1-benzyl piperazine, 1-methyl homo piperazine, 1-ethyl homo piperazine, 1
- the base examples include the above-mentioned organic bases and inorganic bases, and preferably, for example, disopropylethylamine, dimethylaminopyridine, triethylamine, pyridine, 2,6-lutidine, sodium hydride and the like are used.
- the solvent used in the reaction is not particularly limited, but is preferably one that does not easily react with the raw material, and includes the above-mentioned inert solvent, and preferably, for example, tetrahydrofuran, dichloromethane, N, N-dimethylformamide and the like are used.
- the amine and the base used in the reaction are 110 equivalents and 220 equivalents, preferably 1.55 equivalents and 210 equivalents, respectively, based on the compound of the formula (IIIA).
- the reaction time is 10 minutes to 30 hours, preferably 1 to 2 hours.
- the reaction temperature is from ⁇ 78 to the reflux temperature, and preferably from ⁇ 10 to 50.
- the compound of the formula (IVA) can also be obtained by treating the compound of the formula (IIA) with an isocyanate in an inert solvent in the presence of a base and a salt of dibasic or cuprous chloride.
- the isocyanate is not limited, but is, for example, ethyl isocyanate, methyl isocyanate, phenyl isocyanate and the like.
- the base include the above-mentioned organic bases and inorganic bases.
- diisopropylethylamine, dimethylaminopyridine, triethylamine, pyridine, 2,6-lutidine, sodium hydride and the like are used.
- the solvent used in the reaction is not particularly limited, but is preferably one that does not easily react with the starting materials.
- the inert solvent include, for example, tetrahydrofuran, dichloromethane, N, N-dimethylformamide and the like.
- the base and the isocyanate used in the reaction are each 31,000 equivalents, 1 to 20 equivalents, and preferably 5 to 20 equivalents, respectively, based on the compound of the formula (IIIA).
- the amount of cuprous monochloride is the total amount of I-II, and one equivalent is 16 equivalents.
- the reaction time is 10 minutes to 30 hours, preferably 1 to 2 hours.
- the reaction temperature is from 180 ° C. to the reflux temperature, preferably from 110 ° C. to 50 °.
- the OR 3X at the A 1 step one or compounds of two of the unprotected hydroxyl group of OR I6X and OR 21 ⁇ chi is, after converting them hydroxyl into carbonic ester groups in the A 3 step, By converting to a carpamoyloxy group in step A4, it is also possible to convert to a derivative having a plurality of urethane structures.
- Step A5 is a step of producing a compound of the formula (VA). This step is achieved by subjecting a urethane derivative of the compound of the formula (IVA) to deprotection treatment as described below in an inert solvent.
- the reaction of deprotecting the hydroxyl-protecting group varies depending on the type of the protecting group, but is carried out by a method well known in organic synthetic chemistry.
- each hydroxyl group such as 1-ethoxyhexyl, tetrahydroviranyl, 1-methoxycyclohexyl, 4-methoxytetrahydroviranyl, 4-methoxytetrahydrothiopyranyl, 4-methoxytetrahydrothiovilanyl S, S-dioxide Protection is facilitated by acid treatment in an inert solvent.
- the acid include the above-mentioned organic acids and inorganic acids, and preferably include, for example, pyridinium p-toluenesulfonic acid, p-toluenesulfonic acid, camphorsulfonic acid and the like.
- the solvent used in the reaction is not particularly limited, but is preferably one that does not easily react with the raw material, and is preferably an alcoholic solvent such as methanol, ethanol, isopropanol, tert-butanol and the like. An inert solvent may be used as a mixture.
- the acid used in the reaction is 0.5 to 5 equivalents, preferably 1 to 3 equivalents, relative to the compound of the formula (IVA).
- the reaction time is 10 minutes to 10 days, preferably 1 day to 4 days.
- the reaction temperature is from ⁇ 78 ° C. to the reflux temperature, preferably from 10 ° C. to 50 ° C.
- Examples of the monofluorine anion include tetrabutylammonium fluoride, hydrogen fluoride, potassium fluoride, and pyridinium hydrogen fluoride.
- the acid is the above-mentioned organic acid, inorganic acid, or the like. , Formic acid, trifluoroacetic acid, p-toluene Pyridinium sulfonic acid, camphorsulfonic acid and the like.
- the solvent used in the reaction is not particularly limited, but is preferably one that does not easily react with the raw material, and includes the above-mentioned inactive laurel solvents.
- tetrahydrofuran, getyl ether, water and the like are used.
- the fluorine anion and the acid used in the reaction are 1 to 5 equivalents and 0.5 to 5 equivalents, preferably 1 to 4 equivalents and 0.5 to 3 equivalents, respectively, relative to the compound of the formula (IVA). Is equivalent.
- the reaction time is 10 minutes to 30 hours, preferably 1 to 2 hours.
- the reaction temperature is from 178 to the reflux temperature, preferably from 110 ° C to 50 ° C.
- each of the 3- or 21-position hydroxyl groups can be selectively derivatized to a urethane derivative. It is possible to do.
- the method for selectively modifying the hydroxyl group at the 3-position or 21-position performed by variously combining protection and deprotection can also be used for other modification methods described below.
- R N1 R N2 N—CS— An elementary atom or a protecting group or a group represented by the formula R N1 R N2 N—CS— (where “and R have the above-mentioned meanings) (provided that R 3e , R 16e and R 21e are R 3d , R 16d and R 2id are a hydrogen atom or a formula R NI R N2 N-CS- (wherein R N1 and R N2 have the above-mentioned meanings).
- R N1 R N2 N—CS— An elementary atom or a protecting group or a group represented by the formula R N1 R N2 N—CS— (where “and R have the above-mentioned meanings) (provided that R 3e , R 16e and R 21e are R 3d , R 16d and R 2id are a hydrogen atom or a formula R NI R N2 N-CS- (wherein R N1 and R N2 have the above-mentioned meanings).
- Step B1 is a step of synthesizing a compound of the formula (IB) by using thioisocyanate or thiocarbamoyl chloride instead of isocyanate. This step is accomplished by treating the compound of formula (IIA) with isothiocynate or thiocarbamoyl chloride in an inert solvent in the presence of a base or bis (triptyltin) oxide.
- the isothiocyanate to be used is not limited.
- Ethyl isothiocyanate 2- (N, N-ethylamino) ethyl isothiocyanate, 3- (N, N-dimethylamino) propyl isothiocyanate, 3- (N, N-ethylamino) propyl isothiocyanate 2- (Morpholine-41-yl) ethyl isothiocyanate, 2- (Piperidine-111-yl) ethyl isothiocyanate, 2-((Pyrrolidine-1-1-yl) ethyl Isothiosinate and the like.
- the thiocarbamoyl chloride used is not limited, but for example, N, N-dimethylthiocarbamoyl chloride, N-phenyl-2-methylthio carbamoyl chloride, (morpholine-4-yl) thiocarbamoyl chloride, (4-1) Methyl piperazine (11-yl) thiocarpamoyl chloride, (4-methyl homopyrazine-11-yl) thiocarbamoyl chloride, and the like.
- the base examples include the above-mentioned organic bases and inorganic bases, and preferably, for example, diisopropylethylamine, 4-dimethylaminopyridine, triethylamine, pyridine, 2,6-lutidine, hydrogenated sodium, and the like are used.
- the solvent used in the reaction is not particularly limited, but is preferably a solvent that does not easily react with the raw materials, and includes the above-mentioned inert solvents.
- tetrahydrofuran, dichloromethane, N, N-dimethylformamide, toluene -Raru 7 translation For f-Salt-Group + S- (Met-y ⁇ "Run-)-O-I-I-I- and isothiocynate or thiocarbamoyl chloride are of the formula (IIA)
- the amount is 1 to 5 equivalents and 1 to 10 equivalents, preferably 1 to 3 equivalents and 2 to 5 equivalents, respectively, based on the compound.
- the reaction time is 10 minutes to 72 hours, preferably 1 to 24 hours.
- the reaction temperature is from 180 ° C. to the reflux temperature, preferably from 110 ° C. to 70 ° C.
- step A5 the protecting group for the hydroxyl group is removed in step A5, whereby the thiourene derivative of the formula (IIB) can be synthesized.
- R 3e , R 16e and R 21e are a hydrogen atom or a protecting group (R 3e , R 16e and R 21e do not simultaneously represent a hydrogen atom, and at least one represents a hydrogen atom), R 3f , R 16f and R 21f are a C alkyl group which may have a protecting group or a substituent, R 3g , R 16 g and R 21 g are a hydrogen atom, an alkyl group or a carpamoyl group, and R 7g is acetyl Or a rubamoyl group. ]
- reaction is performed in the same manner as the reaction corresponding to one step. However, one or two hydroxyl groups are protected.
- Step C2 is a step of synthesizing a compound of the formula (IIC). This step is achieved by alkylating a hydroxyl group which is not protected by the compound of (IC).
- Alkylation can be achieved by treatment with an alkylating agent of the formula IT-X in the presence of a base.
- X represents a leaving group, and the leaving group includes, for example, a chloro group, a bromo group, an odo group, a trifluoromethanesulfonyl group and the like.
- the base examples include the above-mentioned organic bases and inorganic bases, and preferably, for example, sodium hydride, lithium bis (trimethylsilyl) amide, lithium diisopropylamide, lithium dicyclohexylamide, potassium carbonate, cesium carbonate, 1,8 —Bis (N, N-dimethylamino) naphthene and the like.
- the solvent used in the reaction is not particularly limited, but is preferably one that does not easily react with the starting materials.
- the inert solvent include, for example, getyl ether, tetrahydrofuran, dimethoxyethane, and toluene.
- the alkylating agent and the base used in the reaction are 3 to 20 equivalents and 5 to 30 equivalents, preferably 3 to 5 equivalents and 5 to 10 equivalents, respectively, based on the compound of the formula (IC). .
- the reaction time is 10 minutes to 48 hours, preferably 1 to 24 hours.
- the reaction temperature is from 180 ° C. to the reflux temperature, preferably from 110 ° C. to 70 ° C.
- Step C3 is a step of synthesizing a compound represented by the general formula (IIIC). In this step, if desired, by carrying out Step A2, Step A3, Step A4 and Step A5, it is possible to obtain a compound modified with both a carbamoyl group and an alkyl group. -Ta ⁇ A-One 5 presentations are standing ⁇ — Orayonomi -Gaa -Ooh- ⁇ 1 ⁇ 4 things. Step C3 can be performed by applying the reaction conditions of Step A2, Step A3, Step A4, and Step A5. , OR 3x
- R 3X , R 16X and R 21X have the meaning described above, and R 3F , R 7F , R 16F "and R 21f each represent a hydrogen atom or an alkyl group.]
- the compound of formula (IIA) is subjected to the second step C2 in the same manner as described above, and then to the fifth step A5, wherein the hydroxyl group at the 7-position is alkylated.
- An ether derivative can be obtained.
- the compound of the formula (IIIC) is subjected to Step B1 and then to Step A5 to introduce a thiocarbamoyl group into the hydroxyl group at the 7-position, 21 A derivative having an alkyl group introduced into one or two of the hydroxyl groups at position 1 can also be obtained.
- the step D1 is a step of converting an unprotected hydroxyl group into an ester group using the compound of the formula (I C) synthesized in the step C1 as a raw material.
- the esterification reaction is carried out by, for example, an acid anhydride and a base, an acid halide and a base, a carboxylic acid and a condensing agent, or a Mitsunobu reaction.
- the acid anhydride various carboxylic anhydrides are used.
- a mixed acid anhydride composed of acetic acid, propionic acid, butyric acid, valeric acid, benzoic acid, etc.
- symmetrical acid anhydride succinic anhydride, dartart anhydride Acids, cyclic acid anhydrides such as adipic anhydride, etc., and preferably acetic anhydride, propionic anhydride, butyric anhydride, benzoic anhydride and the like.
- the acid halide for example, various acid chlorides and acid bromides are used, and preferably, for example, acetyl chloride, propionyl chloride, benzoyl chloride, benzoyl bromide and the like are used.
- the base include the above-mentioned organic bases and inorganic bases, and preferably, for example, imidazole, 4-dimethylaminopyridine, pyridine, sodium hydride and the like.
- carboxylic acid various carboxylic acids are used, and preferably, for example, acetic acid, propionic acid, etc., and as the condensing agent, preferably, for example, dicyclohexylcarbodiimide, trifluoroacetic anhydride, carboxylic acid imidazo And N, N-diisopropylcarbodiimide, 1-ethyl-3- (3-dimethylaminopropyl) carbodiimide, and the like.
- various carboxylic acids can be substituted in the presence of triphenylphosphine and getyl azodicarboxylate or diisopropyl azodicarboxylate.
- the solvent used in each reaction is not particularly limited, but is preferably one that does not easily react with the raw material.
- the inert solvent include, for example, dichloromethane, chloroform, tetrahydrofuran and the like.
- the reaction one acid group, 7 acid pulp, one compound
- the reaction time is 10 minutes to 30 hours, preferably 1 to 2 hours.
- the reaction temperature is from ⁇ 78 to the reflux temperature, preferably from ⁇ 10 ° C. to 50 ° C.
- R 3x , R 16x and R 21x have the above-mentioned meaning, and R 3h ′, R 7h ′, R 16h ′ and R 21h ′ are represented by a hydrogen atom or a formula R c ° CO— (Where, represents the same group as defined above). ]
- Step D1 the compound represented by Formula (ID ′) in which the hydroxyl group at the 7-position is esterified is obtained.
- Step D5 the compound represented by Formula (ID ′) in which the hydroxyl group at the 7-position is esterified is obtained.
- R 3 R 16x and R 21x have the meaning described above, and R 3i , R 7i , R 16i and R 21i
- R N30 represents a group represented by the formula (where R ′ Nl , RN2 and RN3 have the above-mentioned meanings). ]
- a hydroxyl group is phosphorylated with the compound of the formula (IIA) as a raw material, This is a step of amidating phosphoric acid esterification.
- the phosphoric esterification reaction is performed, for example, using a phosphoric acid halide and a base.
- Various phosphoric acid halides are used. Examples thereof include dialkoxyphosphoryl chloride, diphenyloxyphosphoryl chloride, alkoxy (N, N-disubstituted amino) phosphoryl chloride, aryloxy (N, N-disubstituted amino) phosphoryl chloride, Alkoxy (N-substituted amino) phosphoryl chloride, and aryloxy (N-substituted amino) phosphoryl chloride.
- Examples of the base include the above-mentioned organic bases and inorganic bases, and preferably, for example, pyridine, 4-dimethylaminopyridine, trieduramine, ethyldiisopropylamine, sodium hydride, n-butyllithium, lithium carbonate, carbonic acid And sodium.
- the solvent used in each reaction is not particularly limited, but is preferably one that does not easily react with the starting materials.
- the inert solvent include the above-mentioned inert solvents. Preferably, for example, dichloromethane, chloroform, tetrahydrofuran, N, N-dimethyl Ruformamide and the like are used.
- the reaction time is 10 minutes to 72 hours, preferably 1 to 24 hours.
- the phosphoric acid halide and the base used in the reaction are 1 to 10 equivalents and 2 to 20 equivalents, preferably 1 to 5 equivalents and 2 to 10 equivalents, respectively, relative to the compound of the formula (IIA). It is.
- the reaction temperature is from 178 to the reflux temperature, preferably from 110 to 50 ° C.
- the thus-prepared phosphoric ester derivative is subjected to the same reaction as in the step A5 to remove the protecting group for the hydroxyl group, thereby producing the compound of the formula (IE).
- R 3x , R 16x and R 21x have the meaning described above, and R 3 R 7 ⁇ and R 21 are a hydrogen atom, R N1 R ′ V2 N—S 0 2 — or R ′ 4 ⁇ A group represented by one S ⁇ 2 — Te, R ⁇ R N2 and R M represents shown) the same groups as previously defined.
- Step F1 is a step of sulfate-forming a hydroxyl group using the compound of the formula (IIA) as a raw material.
- the sulfuric acid esterification reaction is carried out using a sulfuric acid halide and a base.
- Various sulfate halides are used, for example, alkoxysulfonyl chloride, N, N-disubstituted sulfamoyl chloride, and the like.
- the base include the above-mentioned organic bases and inorganic bases, and preferably, for example, pyridine, 4-dimethylethylaminopyridine, triethylamine, ethyldiisopropylamine, sodium hydride, n-butyllithium, potassium carbonate, And sodium carbonate.
- the solvent used in each reaction is not particularly limited, but is preferably one that does not easily react with the starting materials, and examples thereof include the above-mentioned inert solvents.
- inert solvents for example, dichloromethane, chloroform, tetrahydrofuran, N, N-dimethyl Ruformamide and the like are used.
- the sulfate halide and the base used in the reaction are based on the compound of formula (IIA), respectively: 110 equivalents and 2-20 equivalents, preferably 1-5 equivalents and 2-10 equivalents, respectively.
- the reaction time is 10 minutes to 72 hours, preferably 1 to 24 hours.
- the reaction temperature is from 178 ° C to a reflux temperature, preferably from 110 ° C to 50.
- the sulfate group derivative thus synthesized is subjected to the same reaction as in the step A5 to remove the protecting group for the hydroxyl group, whereby the compound of the formula (IF) can be synthesized.
- Step Gl is a step of converting a hydroxyl group into a halogen using the compound of the formula (IA) as a raw material.
- This halogenation reaction is carried out, for example, by treating with carbon tetrabromide, bromine, phosphorus tribromide, iodine or carbon tetrachloride in the presence of a base such as getylaminosulfur trifluoride (DAST) or triphenylphosphine.
- a base such as getylaminosulfur trifluoride (DAST) or triphenylphosphine.
- DAST getylaminosulfur trifluoride
- triphenylphosphine triphenylphosphine.
- the base include general organic bases and inorganic bases, for example, diisopropylethylamine, dimethylaminopyridine, triethylamine, pyridine, 2,6-glutidine, hydrogenated sodium and the like.
- the solvent used for the reaction is not particularly limited, but is preferably one that does not easily react with the raw material, and examples thereof include tetrahydrofuran, dichloromethane, and N, N-dimethylformamide. Of these, a fluorination reaction with getylaminosulfur trifluoride is preferred, and getylaminosulfur trifluoride used for the reaction is preferred.
- (DAST) is 1 to 5 equivalents, preferably 1 to 3 equivalents, to the compound of the formula (IA), respectively.
- the reaction time is between 10 minutes and 10 hours.
- the reaction temperature is from -78 ° C to room temperature.
- the octogen derivative thus synthesized is subjected to removal of the hydroxyl-protecting group from the step A5, whereby a compound of the formula (IG) can be synthesized.
- R 3x , R 16x and R 21x have the meaning described above, and R 3I , R 71 , R 161 and R 211 represent a hydrogen atom or a group represented by R N5 S ⁇ 2 — In the formula , R N5 represents the same group as defined above. ]
- the sulfonylation reaction is carried out in the presence of a base, for example! ) Sulfonyl chlorides such as toluenesulfonyl chloride, methyl sulfonyl chloride and benzenesulfonyl chloride It can be done by processing.
- a base for example!
- Sulfonyl chlorides such as toluenesulfonyl chloride, methyl sulfonyl chloride and benzenesulfonyl chloride It can be done by processing.
- the base include general organic bases and inorganic bases, for example, diisopropylethylamine, dimethylaminopyridine, triethylamine, pyridine, 2,6-lutidine, sodium borohydride and the like.
- the solvent used in the reaction is not particularly limited, but is preferably one that does not easily react with the raw material, and examples thereof include tetrahydrofuran, dichloromethane, N, N-dimethylformamide and the like.
- the sulfonyl chloride and the base used in the reaction are 1 to 5 equivalents and 2 to 10 equivalents, preferably 1 to 3 equivalents and 2 to 6 equivalents, respectively, based on the compound of the formula (IIA). Reaction times range from 10 minutes to 30 hours.
- the reaction temperature ranges from 178 ° C to the reflux temperature.
- the sulfonic acid ester derivative thus synthesized is subjected to the step A5 to remove the protecting group of the hydroxyl group, whereby a derivative represented by the formula (IH) in which the 7-hydroxyl group is sulfonylated can be synthesized. .
- R 3x , R 16x and R 21x have the meaning described above, and R 3m , 7m , R 16m and R 21m are a hydroxyl group or a formula R N1 R N2 N _ (where R N1 and R N2 has the meaning described above). ]
- the hydroxyl group is converted directly to an amine or converted to a group having good elimination property using the compound of (IIA) as a raw material, then converted to an azide, and then reduced to form an amine by reduction.
- DPPA diphenylphosphoryl azide
- DBU Pendaker 7-Den
- TMAD tetramethyl azodicarboxamide
- tributylphosphine 5) sodium azide in the presence of base it can.
- the base examples include the above-mentioned organic bases and inorganic bases, and preferably, for example, diisopropylethylamine, dimethylaminoviridine, triethylamine, pyridine, 2,6-lutidine, sodium hydride and the like are used. It is also possible to convert to azide by treating with sodium azide in the presence of a palladium catalyst.
- the palladium catalyst include Pd (PPh 3 ) 4 and the like.
- the solvent used in the reaction is not particularly limited, but is preferably a solvent that does not easily react with the starting materials, and examples thereof include tetrahydrofuran, dichloromethane, ⁇ , ⁇ -dimethylformamide, toluene, and benzene. Reaction times range from 10 minutes to 30 hours. The reaction temperature is from ⁇ 78 ° C. to the reflux temperature.
- the reduction of azide to amine can be performed by using, for example, triphenylphosphine, lithium aluminum hydride, or the like. Further, reduction to amine can be carried out in a hydrogen atmosphere by using a catalyst such as palladium carbon or Lindlar catalyst.
- the solvent used for the reaction is not particularly limited, but is preferably a solvent that does not easily react with the raw material, and examples thereof include tetrahydrofuran, getyl ether, and ethanol. Reaction times range from 10 minutes to 30 hours. The reaction temperature ranges from 178 ° C to the reflux temperature.
- the conversion of the hydroxyl group to a group having a good elimination property can be carried out according to the above-mentioned step G1 (octalogenation) or step H1 (sulfonyluidani).
- the group which can be easily removed include a chloro group, a bromo group, an aldehyde group, a methanesulfonyl group and a p-toluenesulfonyl group.
- the compound converted to a leaving group is treated with an amine in an inert solvent in the presence of a base to synthesize a compound having a hydroxyl group converted to an amino group or an amino group having a substituent. be able to.
- —Usage—Ru Example: One-methyl-: ⁇ Ru: ⁇ — ⁇ 3 ⁇ 4 ⁇ ik ⁇ , —ethylamine, etc.
- the base include the above-mentioned organic bases and inorganic bases. Preferred are, for example, diisopropylethylamine, dimethylaminopyridine, triethylamine, pyridine, 2,6-lutidine, sodium hydride and the like.
- the solvent used in the reaction is not particularly limited, but preferably does not easily react with the raw material, and examples thereof include the inert solvent. Preferably, for example, tetrahydrofuran, dichloromethane, N, N-dimethylformamide and the like are used.
- the reaction time is 10 minutes to 30 hours, preferably 1 to 2 hours.
- the reaction temperature is from 178 ° C to the reflux temperature, preferably from 110 ° C to 50 ° C.
- the amide derivative thus synthesized is subjected to the same reaction as in Step A5 to remove the protecting group for the hydroxyl group, whereby the compound of the formula (I ⁇ ) can be produced.
- amino group of the compound of the formula (I ⁇ ) is subjected to alkylation, acylation, sulfamoylation or sulfonylation by a method well-known in organic synthetic chemistry and the above-mentioned method, respectively.
- the compound of (I) can be produced.
- one of R and R 3ia represents a hydroxyl group and the other represents a hydrogen atom, or the oxo group together with the carbon atom to which R 3ia and R 3Ia bind.
- one of R 21Ja and R 21ia represents a hydroxyl group and the other represents a hydrogen atom, or represents an oxo group together with a carbon atom to which R 2 ′′ a and R bind.
- the J1 step is a step of synthesizing a ketone compound represented by the formula (IIJ) by performing an oxidation reaction of a hydroxyl group using 6-deoxy111107D as a raw material.
- the solvent used in the reaction is not particularly limited, but is preferably one that does not easily react with the starting compound, such as tetrahydrofuran, dichloromethane, Loroform, toluene and the like.
- the reaction is carried out at a temperature of 178 ° C or under reflux. Reaction times range from 10 minutes to 78 hours.
- a reaction using a Dess-Martin reagent, manganese dioxide, or a reagent under Swern oxidation conditions is preferable, and a reaction using a Dess-Martin reagent is particularly preferable.
- a solvent for the oxidation reaction using the Dess-Martin reagent dichloromethane and chloroform are particularly preferable, and the oxidizing agent used is 1 to 20 equivalents based on the compound (6-deoxy111107D). It is preferably 1 to 5 equivalents.
- the reaction temperature is preferably 0 ° C. to room temperature, and the reaction time is 30 minutes to 24 hours, preferably 1 to 8 hours.
- a compound previously derivatized into a urethane derivative, a thiourethane derivative, an ester derivative, an alkyl derivative, or the like by the above method is used in place of 6-dexoxy 11107D.
- the hydroxyl groups at the 3-, 7-, and Z- or 21-positions can each be selectively turned into an oxo structure.
- the compound previously converted into an oxo structure is subjected to urethane derivatization, thiourethane derivatization, ester derivatization or alkyl derivation according to the above-described method, so that the corresponding modification and oxo can be obtained.
- Compounds modified by both chemical formulas can also be synthesized.
- the compound represented by the formula (I) can be synthesized by appropriately combining the reactions A to J described above with the protection and deprotection of a hydroxyl group.
- the target substance of each reaction is collected from the reaction mixture according to a conventional method. For example, if insolubles are present, they are filtered off as appropriate and the solvent is distilled off under reduced pressure. ⁇ It can be obtained by drying the ink with magnesium sulfate-free water and then distilling off the solvent.If necessary, use a conventional method such as column chromatography, thin-layer chromatography, high-performance liquid chromatography, etc. It can be purified.
- the compound of the formula (I) can be synthesized from isolated and purified 6-deoxyd 1111 D and 6-dox 11 D compounds using common organic synthesis means. . Representative: A. urethane derivatives, B. thiourethane derivatives, C. ether derivatives, D.
- the H. sulfonic acid ester derivative, the I. amine derivative and the J. keto compound can be produced by the above-mentioned method by changing the conditions of the above-mentioned step 1A of protecting the hydroxyl group.
- a representative compound of the compound of the formula (I) of the present invention was used to inhibit VEGF transcription, inhibit proliferation of WiDr human colon cancer cells, inhibit solid tumor growth. The effects and weight loss (acute toxicity) and stability in aqueous solution were measured.
- the VEGF promoter-night sequence was cloned and inserted into a secreted alkaline phosphatase (PLAP) vector to construct a repo overnight vector.
- PLAP secreted alkaline phosphatase
- VEGF genomic was cloned from a phage library. Based on VEGF cDNA (GenBank access ion number: X62568), PCR primers having the sequences of SEQ ID NO: 1 and SEQ ID NO: 2 were designed and PCR was performed to obtain a fragment of about 340 bp. Using this as a probe, a human genomic library (Clontech) was screened to obtain PUC18-VEGFA containing a VEGF 5 'flanking region of about 5.4 kb.
- This pUC18-VEGFA was digested with KpnI / NheI, and the obtained about 0.3 kb VEGF promoter region was secreted, and the secretory form of the plasmid was expressed as (PtA-P-)-po-" ⁇ -. ⁇ (-Ratsu Fuji-0-1
- VEGF-PLAP vector was constructed by insertion between the multiple cloning sites KpnI / NheI.
- VEGF-PLAP vector was introduced into J251 cells, and cultured in the presence of 1 mg / mL G418 (Merck) to establish stable G418-resistant clones (OJ251 / 1-8 cells).
- U251 / 1-8 cells were prepared by placing PLAP in the medium under hypoxia (2% O 2 incubator), as reported by Minchenko et al. (Cell. Mol. Biol. Res., 40, 35-39, 1994). It was secreted and confirmed to be a repo overnight system reflecting transcription from VEGF promoter overnight. Hereinafter, a compound that suppresses VEGF production induced by hypoxia stimulation was screened using this clone.
- the U251 / 1-8 cells were washed twice with a sufficient amount of PBS (Phosphate buf fered saline) to remove the effects of alkaline phosphatase in serum, 65.
- PBS Phosphate buf fered saline
- C Diluted in DMEM medium containing 10% serum in which alkaline phosphatase in serum was inactivated by treatment for 20 minutes, and seeded on a 96-well plate with 4 ⁇ 10 4 cels I 180 L each.
- C0 2 incubator one (5% C0 2) was cultured overnight at 37 ° C, after adding the broth 20 containing a test compound diluted with 3-fold serial, hypoxic (2% 0 2) incubator Incubated for 18 hours.
- PLAP activity in the culture supernatant of 50 L 0. 28 M Na 2 C0 3 - NaHC0 3 buffer solution (. ⁇ ⁇ 0, 8. 0 DiM MgS0 4) was added to the culture supernatant to 10 ⁇ L, Finally, an alkaline phosphatase substrate (Lumistin, Genome Science Inc.) was added. After the reaction for 1 hour, the PLAP activity was measured by chemiluminescence using a microplate reader (PerkinElmer) to measure the activity of the lipophilic phosphatase.
- the compound of formula (I) showed strong VEGF transcription repressing activity.
- WiDr human colon cancer cells cultured in Dulbecco's modified Eagle's medium (MEM SIGMA) containing fetal serum, benicillin (100 units / mL), and streptomycin (100 g / mL) were cultured in 2X cells. 10 3 cel ls / well was seeded on a 96-well plate. C0 2 incubator base - After overnight incubation at coater, and cultured by adding the culture broth of 20 zL containing the test compound in 3-fold dilution series.
- MEM SIGMA Dulbecco's modified Eagle's medium
- Table 2 shows the IC 5Q values of the representative compounds.
- the compound of the formula (I) exhibited a strong WiDr human colon cancer cell growth inhibitory action.
- WiDr human colon cancer cells were implanted subcutaneously into the body of nude mice, and when the tumor volume reached about 100 band 3, each group The groups were divided so that the average of the tumor volumes was uniform, and the control group was 10 animals and the 6-deoxy 11107D derivative administration group was 5 animals.
- the administration group was intravenously administered for 5 days so as to be any of 0.625 mg, 1.25 mg, 2.5 mg, 5 mg, and lOmg / kg / day, and the vehicle was administered to the control group.
- the tumor volume on the 15th day was measured, and the relative tumor weight ratio (T / C »was determined, taking the tumor weight of the control group as 100%.
- the T / « of a representative compound of the formula (I.) was calculated as The results are shown in Table 3.
- the body weight was measured on the first day of administration, the fifth day, the eighth day, the 12th day, and the 15th (or 16th) day. Relative body weight variation was examined for representative compounds, and the relative body weight ratio on the day when body weight was the lowest was shown as the minimum relative body weight ratio in Table 3.
- the compound of formula (I) is dissolved in DMS0 at a concentration of 10-20,
- the compound of the formula (I) of the present invention can be used for, for example, a therapeutic agent for cancer, particularly It is expected to be used as a cancer metastasis inhibitor, a therapeutic agent for diabetic retinopathy, a therapeutic agent for rheumatoid arthritis, and a therapeutic agent for hematomas.
- a therapeutic agent for cancer particularly It is expected to be used as a cancer metastasis inhibitor, a therapeutic agent for diabetic retinopathy, a therapeutic agent for rheumatoid arthritis, and a therapeutic agent for hematomas.
- the dose of the test mouse without significant weight loss has an inhibitory effect on the growth of WiDr human colon cancer cells. Therefore, it is effective as a preventive and therapeutic agent for diseases in which gene expression control is effective, diseases in which VEGF production inhibitory effect is effective, and diseases in which angiogenesis inhibitory effect is effective.
- “Prevention or treatment” refers to prevention or treatment or both.
- the compound of the formula (I) of the present invention is more specifically effective as an anticancer agent, particularly as an anticancer agent / cancer metastasis inhibitor for solid tumors.
- Solid tumors include, for example, gut cancer, stomach cancer, colon cancer, breast cancer, prostate cancer, lung cancer, kidney cancer, brain tumor, head and neck cancer, esophageal cancer, skin cancer, liver cancer, uterine cancer, cervical cancer, bladder cancer, Examples include thyroid cancer, testicular tumor, choriocarcinoma, osteosarcoma, soft tissue sarcoma, and ovarian cancer.
- Particularly preferred are cancers such as colon cancer, breast cancer, prostate cancer, lung cancer, head and neck cancer, and ovarian cancer.
- a pH adjuster, buffer, stabilizer, solubilizing agent, etc. may be added to the main drug, if necessary, and then subcutaneously, intramuscularly, intraarticularly, or intravenously by a conventional method. Agent.
- the compound When the compound is administered as a therapeutic or prophylactic agent for various diseases, it may be administered orally as tablets, powders, granules, capsules, syrups, etc., or as sprays, suppositories, injections, It may be administered parenterally as an external preparation or infusion.
- the dosage varies significantly depending on the severity of symptoms, age, type of liver disease, etc. Usually, about lnig to 100 mg per adult per day is administered in one to several doses.
- formulation it is manufactured by a usual method using an ordinary formulation carrier. That is, when preparing a solid dosage form for oral use, excipients and, if necessary, binders, disintegrants, lubricants, coloring agents, flavoring agents, etc. are added to the active ingredient, and then tablets are prepared in the usual manner. , Coated tablets, granules, powders, capsules, etc. These tablets and granules may be sugar-coated, gelatin-coated, or of course coated as needed.
- the compound of formula (I) of the present invention inhibits H VEGF-production-inhibiting rr angiogenesis-inhibiting-inhibiting-r-i-irvi-TO-solid -In addition to having excellent antitumor effects in tumor models, it also has stability in aqueous solution, for example, cancer therapeutics, especially solid cancer therapeutics, cancer metastasis inhibitors, diabetic retina And a therapeutic agent for rheumatoid arthritis, a therapeutic agent for hematoma, and the like.
- cancer therapeutics especially solid cancer therapeutics, cancer metastasis inhibitors, diabetic retina And a therapeutic agent for rheumatoid arthritis, a therapeutic agent for hematoma, and the like.
- Examples 1 to 4 A method for producing 6-dexoxy 11107B (Examples 1 to 4), a reaction for converting microorganisms from 6-dexoxy 11107B to 6-dexoxy 11107D (Examples 5 to 11), and analogs of 6-dexoxy 11107D are described below.
- Examples 12 to: L 7 Examples (Examples 12 to: L 7) and the 6-dexoxy 11107D derivative (Examples 18 to 60) will be described in more detail with reference to Examples and Reference Examples. It is not limited only to the embodiment.
- the present invention relates to a method for preparing Streptomyces esperiium. 11107 (Streptomycess p. Mer- 11107, accession number FERM BP-7812) into Tris' maleic acid buffer (pH 6.0). Mutation treatment (100 g / mL, 28, 1 hour) with N_methyl-N'-nitroso-N-nitrosoguanidine was performed in yeast and then spread on yeast-malt agar medium to form spores . The obtained spores were collected, a part thereof was diluted and spread on yeast-malt agar medium to form colonies.
- Cells are collected from each colony, and 2 mL of seed culture medium (Dalcose 2%, soybean flour (Essan Meat, Ajinomoto Co.) 1%), yeast extract (Orien Yuru Yeast Co., Ltd.) 0.5% , Sodium chloride 0.25% and calcium carbonate 0.32%, pH 6.8) inoculated in a 15 mL test tube, and shaken at 25 ° C for 2 days.
- seed culture medium Dalcose 2%, soybean flour (Essan Meat, Ajinomoto Co.) 1%
- yeast extract Ole Yuru Yeast Co., Ltd.
- Sodium chloride 0.25% and calcium carbonate 0.32%, pH 6.8 inoculated in a 15 mL test tube, and shaken at 25 ° C for 2 days.
- AIST National Institute of Advanced Industrial Science and Technology
- Example 3 Purification of 6-deoxy 11107B and 6-deoxy 11107BI
- the culture solution (1.2L) prepared by the method of Example 2 was separated into filtrate and cells using a centrifuge. separated. The supernatant was extracted with ethyl acetate (1.2 L). The cells were extracted with acetone (500 mL) and filtered to obtain an acetone extract. After acetone was distilled off from the acetone extract under reduced pressure, the mixture was extracted with ethyl acetate (1 L).
- 07B was determined to be a structure represented by the formula avr ').
- Solubility Soluble in dimethyl sulfoxide, pyridine, methanol and acetone, poorly soluble in water
- 6-deoxy 1 1 107 B I The physicochemical properties of 6-deoxy 1 1 107 B I are shown below. 6-Doxy 107 B I was determined to have the structure shown below. Molecular weight: 504, ES I—MS 527 (M + Na) +, ES I—MS 503 (M-H)-'
- slant medium yeast / malt agar medium
- seed culture medium soluble starch 2.4%, glucose 0.1%, soybean flour (Essan Meat, Ajinomoto Co.) 0.5%, Beef extract (Diffco) 0.3%, Yeast extract (Diffco) 0.5%, Tryptone / Peptone (Diffco good)
- seed culture medium soluble starch 2.4%, glucose 0.1%, soybean flour (Essan Meat, Ajinomoto Co.) 0.5%, Beef extract (Diffco) 0.3%, Yeast extract (Diffco) 0.5%, Tryptone / Peptone (Diffco good)
- the seed culture was obtained by culturing on a press. Further, 0.6 mL of the seed culture was added to 60 mL of the production medium (potato starch 2%, glucose 2%, soybean powder (Essan Meat, Ajinomoto Co.) 2%, yeast extract 0.5%, sodium chloride 0.25%, calcium carbonate 0.32%, copper sulfate 0.0005%, manganese chloride 0.0005%, and zinc sulfate 0.0005%; pH 7.4) into a 500 mL Erlenmeyer flask containing The cells were cultured on a shaking incubator at 28 ° C for 4 days.
- the production medium potato starch 2%, glucose 2%, soybean powder (Essan Meat, Ajinomoto Co.) 2%, yeast extract 0.5%, sodium chloride 0.25%, calcium carbonate 0.32%, copper sulfate 0.0005%, manganese chloride 0.0005%, and zinc sulfate 0.0005%; pH 7.4
- the frozen seed mother was thawed, and 0.25 mL of the frozen mother mother was added to 25 mL of seed mother medium (potato starch 2%, glucose 2%, soybean powder (Essan Meat, Ajinomoto Co.) 2%, yeast extract 0.5%, chloride Inoculate a 25 OmL Erlenmeyer flask containing 0.25% sodium, 0.32% calcium carbonate, 0.0005% copper sulfate, 0.0005% manganese chloride and 0.0005% zinc sulfate; pH 7.4).
- seed mother medium potato starch 2%, glucose 2%, soybean powder (Essan Meat, Ajinomoto Co.) 2%, yeast extract 0.5%, chloride Inoculate a 25 OmL Erlenmeyer flask containing 0.25% sodium, 0.32% calcium carbonate, 0.0005% copper sulfate, 0.0005% manganese chloride and 0.0005% zinc sulfate; pH 7.4
- the seed culture was obtained by cul
- a production medium potato starch 2%, glucose 2%, soybean powder (Essan Meat, Ajinomoto Co., Inc.) 2%, yeast extract 5%, sodium salt sodium 0.25) %, Calcium carbonate 0.32%, copper sulfate 0.0005%, manganese chloride 0.0005% and zinc sulfate 0.0005%; pH 7.4.
- C. The cells were cultured on a shaking incubator for 4 days.
- Example 7 Obtained by the method of Example 7 A- 1544 strain culture solution (25 ml / 25 OmL Erlenmeyer Raranu _3 ⁇ _3 - sigma present -) - So - is not one Q-Q-0 one r - The cells were collected by P - m , 1- 1 Q-minute-minute-centrifugation-separation, and suspended in 25 mL of 50 mM phosphate buffer (pH 6.0). Next, 6-deoxy 11107B as a substrate was prepared as a 5 Omg mL dimethyl sulfoxide solution, and 0.25 mL of each was added. After addition, shake at 28 ° C for 22 hours, A hydroxylation reaction was performed.
- the filtrate and the cells were separated by centrifugation at 5000 rpm for 20 minutes.
- the supernatant was extracted with ethyl acetate (750 mL).
- the cells were extracted with acetone (500 mL) and filtered to obtain an acetone extract.
- the acetone was distilled off from the acetone extract under reduced pressure, and then extracted with ethyl acetate (750 mL).
- the respective ethyl acetate layers were washed with water, dehydrated and dried over anhydrous sodium sulfate, and then combined under reduced pressure to obtain 365 mg of a crude active fraction.
- the crude active fraction was subjected to silica gel force chromatography (Kiese 1 gel 60, 35 g), washed with toluene (7 OmL), and treated with toluene and acetone (4: 1; v / v). The mixture was eluted with a mixture (50 OmL) and a mixture of toluene and acetone (2: 1; v / v) (30 OmL), and 139 mg of crude active fraction containing 6-deoxy 11107 D and 6-deoxy 11107 BP was added. 78 mg of a crude active fraction containing 6-deoxy 11107D 20-OH and 78 mg of a crude active fraction containing 6-deoxy 11107 F were obtained.
- the crude active fraction containing 6-deoxy 11107 D and 6-deoxy 11107 BP obtained was subjected to preparative high performance liquid chromatography (HPLC) under the following preparative conditions (B 1) to elute 6-dexoxy. After obtaining 11107 D and 6-deoxy 11107 BP fractions, the solvent was distilled off to obtain 6-deoxy 1110 BP.
- HPLC high performance liquid chromatography
- the crude active fraction containing 6-deoxy 11107 AV was subjected to the above-mentioned fractionation conditions (A1), and the crude active fraction containing 6-deoxy 11107D 20-0H and 6-dexoxy 11107F was subjected to the following fractionation conditions (A1).
- the solvent was distilled off to obtain 6-dexoxy 11107 AV (9.3 mg), 6-dexoxy 11107D 20 -0H (19.9 mg) and 6-dexoxy. 11107 F (22. lmg) was obtained.
- This frozen seed mother was thawed, and 0.25 mL of the mother seed was dissolved in 25 mL of seed mother medium (potato starch 2%, glucose 2%, soybean powder (Essan Meat, Ajinomoto Co.) 2%, yeast extract 0.5%, chloride Inoculate a 25 OmL Erlenmeyer flask containing 0.25% sodium, 0.32% calcium carbonate, 0.0005% copper sulfate, 0.0005% manganese chloride and 0.0005% zinc sulfate; pH 7.4). The mixture was shaken at 28 ° C. for 2 days.
- seed culture was added to 25 mL of production medium (potato starch 2%, glucose 2%, soybean powder (Essan Meat, Ajinomoto Co.) 2%, yeast extract 5%, sodium chloride 0.25%, Transfer to a 25 OmL Erlenmeyer flask containing 0.32% of calcium carbonate, 0.0005% of copper sulfate, 0.0005% of manganese chloride and 0.0005% of zinc sulfate; pH 7.4) at 25 ° C.
- production medium potato starch 2%, glucose 2%, soybean powder (Essan Meat, Ajinomoto Co.) 2%, yeast extract 5%, sodium chloride 0.25%, Transfer to a 25 OmL Erlenmeyer flask containing 0.32% of calcium carbonate, 0.0005% of copper sulfate, 0.0005% of manganese chloride and 0.0005% of zinc sulfate; pH 7.4
- A-1545 strain culture solution (25 mLZ 25 OmL Erlenmeyer flask, 40 tubes) obtained by the method of Example 9 was collected by centrifugation at 3000 rpm for 10 minutes, and 5 OmM phosphorus was collected.
- the cells were suspended in 25 mL of an acid buffer (pH 6.0).
- 6-deoxy 11107B as a substrate was prepared as a 5 Omg / mL dimethyl sulfoxide solution, and 0.25 mL of each was added. After the addition, the mixture was shaken at 28 ° C for 22 hours to carry out a hydroxylation reaction. After the reaction, centrifugation at 5000 rpm for 20 minutes was performed to remove the filtrate and the cells- ⁇ b "ta ⁇ b-cleared from vinegar (-1-L-extracted-
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Genetics & Genomics (AREA)
- General Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Cardiology (AREA)
- Diabetes (AREA)
- Heart & Thoracic Surgery (AREA)
- Rheumatology (AREA)
- Hematology (AREA)
- Oncology (AREA)
- Vascular Medicine (AREA)
- Emergency Medicine (AREA)
- Obesity (AREA)
- Endocrinology (AREA)
- Pain & Pain Management (AREA)
- Ophthalmology & Optometry (AREA)
- Urology & Nephrology (AREA)
- Immunology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Physical Education & Sports Medicine (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Description
Claims
Priority Applications (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN038183129A CN1671856B (zh) | 2002-07-31 | 2003-07-31 | 生理活性物质 |
| BR0313039-8A BR0313039A (pt) | 2002-07-31 | 2003-07-31 | Composto, medicamento, composição farmacêutica, método para prevenir ou tratar uma doença contra a qual um controle da expressão do gene é eficaz, método para prevenir ou tratar uma doença contra a qual a supressão da produção de vegf é eficaz, método para prevenir ou tratar uma doença contra a qual uma inibição da angiogênese é eficaz, uso do composto, método para produzir um composto 9-desoxi 1107, linhagem streptomyces sp. a-1543 (ferm bp-8442), método de produzir um composto 6- desoxi, e linhagem a-1544 (ferm bp-8446) ou linhagem a-1545 (ferm bp-8447) |
| AU2003252445A AU2003252445B2 (en) | 2002-07-31 | 2003-07-31 | Novel physiologically active substances |
| EP20030771459 EP1548121B1 (en) | 2002-07-31 | 2003-07-31 | Physiologically active substances |
| CA2494536A CA2494536C (en) | 2002-07-31 | 2003-07-31 | A 12-membered ring macrolide active substance |
| MXPA05001104A MXPA05001104A (es) | 2002-07-31 | 2003-07-31 | Nuevas sustancias fisiologicamente activas. |
| KR1020057001794A KR101285695B1 (ko) | 2002-07-31 | 2003-07-31 | 신규한 생리 활성 물질 |
| JP2004524326A JP4328293B2 (ja) | 2002-07-31 | 2003-07-31 | 新規生理活性物質 |
| US10/522,731 US7256178B2 (en) | 2002-07-31 | 2003-07-31 | Physiologically active substances |
| NZ537830A NZ537830A (en) | 2002-07-31 | 2003-07-31 | Novel physiologically active substances |
| IL16623005A IL166230A0 (en) | 2002-07-31 | 2005-01-11 | Macrolide compounds pharmaceutical compositions containing the same and methods for the preparation thereof |
| NO20050921A NO20050921L (no) | 2002-07-31 | 2005-02-21 | Nye fysiologisk aktive substanser |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2002-224105 | 2002-07-31 | ||
| JP2002224105 | 2002-07-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004011661A1 true WO2004011661A1 (ja) | 2004-02-05 |
Family
ID=31184996
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2003/009753 WO2004011661A1 (ja) | 2002-07-31 | 2003-07-31 | 新規生理活性物質 |
Country Status (13)
| Country | Link |
|---|---|
| US (1) | US7256178B2 (ja) |
| EP (1) | EP1548121B1 (ja) |
| JP (1) | JP4328293B2 (ja) |
| KR (1) | KR101285695B1 (ja) |
| CN (1) | CN1671856B (ja) |
| AU (1) | AU2003252445B2 (ja) |
| BR (1) | BR0313039A (ja) |
| CA (1) | CA2494536C (ja) |
| MX (1) | MXPA05001104A (ja) |
| NO (1) | NO20050921L (ja) |
| NZ (1) | NZ537830A (ja) |
| RU (1) | RU2338741C2 (ja) |
| WO (1) | WO2004011661A1 (ja) |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005052152A1 (ja) * | 2003-11-27 | 2005-06-09 | Mercian Corporation | マクロライド系化合物の水酸化に関与するdna |
| WO2005075681A1 (ja) * | 2004-02-06 | 2005-08-18 | Eisai R & D Management Co., Ltd. | 抗癌剤に対する癌細胞の感受性を検定する方法 |
| WO2006009276A1 (ja) * | 2004-07-20 | 2006-01-26 | Eisai R & D Management Co., Ltd. | プラジエノライドの生合成に関与するポリペプチドをコードするdna |
| WO2006126723A1 (ja) * | 2005-05-26 | 2006-11-30 | Eisai R & D Management Co., Ltd. | 遺伝子組換え微生物およびそれらの微生物を用いるマクロライド系化合物の製造方法 |
| WO2008016187A1 (fr) | 2006-08-02 | 2008-02-07 | Eisai R & D Management Co., Ltd. | Molécule cible de pladiénolide, composé capable de se lier à la molécule cible et procédé de criblage pour rechercher le composé |
| US7816401B2 (en) | 2005-10-13 | 2010-10-19 | Eisai R&D Management Co., Ltd. | Process for total synthesis of pladienolide B and pladienolide D |
| WO2015175594A1 (en) | 2014-05-15 | 2015-11-19 | Eisai R&D Management Co., Ltd. | Pladienolide pyridine compounds and methods of use |
| WO2017040526A2 (en) | 2015-09-01 | 2017-03-09 | Eisai R&D Management Co., Ltd. | Splice variants associated with neomorphic sf3b1 mutants |
| WO2017087667A1 (en) | 2015-11-18 | 2017-05-26 | Eisai R&D Management Co., Ltd. | A solid state form of pladienolide pyridine compounds and methods of use |
| WO2018170129A1 (en) | 2017-03-15 | 2018-09-20 | Eisai Co., Ltd | Spliceosome mutations and uses thereof |
| WO2019089641A1 (en) | 2017-10-31 | 2019-05-09 | Eisai R&D Management Co., Ltd. | Combination comprising at least one spliceosome modulator and at least one inhibitor chosen from bcl2 inhibitors, bcl2/bclxl inhibitors, and bclxl inhibitors and methods of use |
| WO2019200100A1 (en) | 2018-04-12 | 2019-10-17 | Andrew Cook | Pladienolide derivatives as spliceosome targeting agents for treating cancer |
| WO2019199667A2 (en) | 2018-04-09 | 2019-10-17 | Keaney Gregg F | Certain pladienolide compounds and methods of use |
| WO2019232433A2 (en) | 2018-06-01 | 2019-12-05 | Eisai R&D Management Co., Ltd. | Methods of using splicing modulators |
| JP2021532063A (ja) * | 2018-06-01 | 2021-11-25 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | スプライシング調節薬抗体−薬物コンジュゲート及びその使用方法 |
| WO2022098712A1 (en) | 2020-11-04 | 2022-05-12 | Eisai R&D Management Co., Ltd. | Biomarkers for myelodysplastic syndrome (mds) and methods of using the same |
| WO2023131866A1 (en) | 2022-01-05 | 2023-07-13 | Eisai R&D Management Co., Ltd. | Biomarkers for myelodysplastic syndrome (mds) and methods of using the same |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI312681B (en) * | 2001-02-01 | 2009-08-01 | Novel physiologically active substance | |
| TWI334866B (en) * | 2002-05-29 | 2010-12-21 | Mercian Corp | Novel physiologically active substances |
| CN1717493A (zh) * | 2002-11-29 | 2006-01-04 | 美露香株式会社 | 大环内酯化合物的产生方法 |
| WO2007110705A2 (en) * | 2005-09-28 | 2007-10-04 | Novimmune Sa | Macrolide as inhibitors of mhc class ii |
| AU2008211950A1 (en) * | 2007-01-29 | 2008-08-07 | Eisai R & D Management Co., Ltd. | Solid of macrolide compound, method for production thereof, and pharmaceutical composition comprising the same |
| US20080312317A1 (en) * | 2007-04-12 | 2008-12-18 | Eisai R&D Management Co., Ltd. | 12 membered-ring macrolactam derivatives |
| GB0821539D0 (en) * | 2008-11-25 | 2008-12-31 | Merlion Pharmaceuticals Pte Ltd | Therapeutic compounds and their use |
| CN111973589A (zh) * | 2019-05-21 | 2020-11-24 | 中国科学院近代物理研究所 | pladienolide B在增强宫颈癌细胞辐射敏感性中的应用 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04352783A (ja) * | 1991-05-27 | 1992-12-07 | Taisho Pharmaceut Co Ltd | 12員環マクロライド系化合物 |
| WO2002060890A1 (fr) | 2001-02-01 | 2002-08-08 | Mercian Corporation | Nouvelles substances physiologiquement actives |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SG49853A1 (en) * | 1993-08-16 | 2001-07-24 | Novartis Ag | Novel macrolides and the use thereof |
| US6441186B1 (en) * | 1996-12-13 | 2002-08-27 | The Scripps Research Institute | Epothilone analogs |
| US6380395B1 (en) * | 1998-04-21 | 2002-04-30 | Bristol-Myers Squibb Company | 12, 13-cyclopropane epothilone derivatives |
| CA2360952A1 (en) * | 1999-02-11 | 2000-08-17 | Schering Aktiengesellschaft | Epothilon derivatives, method for the production and the use thereof as pharmaceuticals |
| TWI233361B (en) * | 2001-04-13 | 2005-06-01 | Gen Hospital Corp | Methods of preventing UVB-induced skin damage |
-
2003
- 2003-07-31 BR BR0313039-8A patent/BR0313039A/pt not_active IP Right Cessation
- 2003-07-31 AU AU2003252445A patent/AU2003252445B2/en not_active Ceased
- 2003-07-31 CA CA2494536A patent/CA2494536C/en not_active Expired - Fee Related
- 2003-07-31 WO PCT/JP2003/009753 patent/WO2004011661A1/ja active Application Filing
- 2003-07-31 KR KR1020057001794A patent/KR101285695B1/ko not_active IP Right Cessation
- 2003-07-31 CN CN038183129A patent/CN1671856B/zh not_active Expired - Fee Related
- 2003-07-31 NZ NZ537830A patent/NZ537830A/en not_active IP Right Cessation
- 2003-07-31 JP JP2004524326A patent/JP4328293B2/ja not_active Expired - Fee Related
- 2003-07-31 EP EP20030771459 patent/EP1548121B1/en not_active Expired - Lifetime
- 2003-07-31 US US10/522,731 patent/US7256178B2/en not_active Expired - Fee Related
- 2003-07-31 RU RU2005105312/04A patent/RU2338741C2/ru not_active IP Right Cessation
- 2003-07-31 MX MXPA05001104A patent/MXPA05001104A/es active IP Right Grant
-
2005
- 2005-02-21 NO NO20050921A patent/NO20050921L/no not_active Application Discontinuation
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04352783A (ja) * | 1991-05-27 | 1992-12-07 | Taisho Pharmaceut Co Ltd | 12員環マクロライド系化合物 |
| WO2002060890A1 (fr) | 2001-02-01 | 2002-08-08 | Mercian Corporation | Nouvelles substances physiologiquement actives |
Non-Patent Citations (4)
| Title |
|---|
| AKIFUMI OKUDA ET AL.: "Shinki ko-shuyo kassei busshitsu pladienolide ni kansuru kenkyu(2)-VEGF sansei yokusei kassei o shihyo to shita pladienolide-rui no kozo kassei sokan", JAPAN SOCIETY FOR BIOSCIENCE, BIOTECHNOLOGY AND AGROCHEMISTRY, 2003 NENDO (HEISEI 15 NENDO), TAIKAI KOEN YOSHISHU (2E03P06), March 2003 (2003-03-01), pages 124, XP002972297 * |
| KEIJI MIZUI ET AL.: "Shinki ko-shuyo kassei busshitsu pladienolide ni kansuru kenkyu(3)-pladienolide-rui no yakuri kassei (in vitro, in vivo)", JAPAN SOCIETY FOR BIOSCIENCE, BIOTECHNOLOGY AND AGROCHEMISTRY, 2003 NENDO (HEISEI 15 NENDO), TAIKAI KOEN YOSHISHU (2E03P07), March 2003 (2003-03-01), pages 124, XP002972298 * |
| See also references of EP1548121A4 |
| TAKASHI SAKAI ET AL.: "Shinki ko-shuyo kassei busshitsu pladienolide ni kansuru kenkyu(1)-shinki 12-inkan macrolide pladienolide B no tanri to kozo", JAPAN SOCIETY FOR BIOSCIENCE, BIOTECHNOLOGY AND AGROCHEMISTRY, 2003 NENDO (HEISEI 15 NEN), TAIKAI KOEN YOSHISHU (2E03P05), March 2003 (2003-03-01), pages 123, XP002972296 * |
Cited By (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7932083B2 (en) | 2003-11-27 | 2011-04-26 | Mercian Corporation | DNA participating in hydroxylation of macrolide compound |
| WO2005052152A1 (ja) * | 2003-11-27 | 2005-06-09 | Mercian Corporation | マクロライド系化合物の水酸化に関与するdna |
| WO2005075681A1 (ja) * | 2004-02-06 | 2005-08-18 | Eisai R & D Management Co., Ltd. | 抗癌剤に対する癌細胞の感受性を検定する方法 |
| AU2005265443B2 (en) * | 2004-07-20 | 2008-09-04 | Eisai R & D Management Co., Ltd | DNA coding for polypeptide participating in biosynthesis of pladienolide |
| JPWO2006009276A1 (ja) * | 2004-07-20 | 2008-05-01 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | プラジエノライドの生合成に関与するポリペプチドをコードするdna |
| JP4599357B2 (ja) * | 2004-07-20 | 2010-12-15 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | プラジエノライドの生合成に関与するポリペプチドをコードするdna |
| US8008049B2 (en) | 2004-07-20 | 2011-08-30 | Eisai R&D Management Co., Ltd. | DNA coding for polypeptide participating in biosynthesis of pladienolide |
| WO2006009276A1 (ja) * | 2004-07-20 | 2006-01-26 | Eisai R & D Management Co., Ltd. | プラジエノライドの生合成に関与するポリペプチドをコードするdna |
| WO2006126723A1 (ja) * | 2005-05-26 | 2006-11-30 | Eisai R & D Management Co., Ltd. | 遺伝子組換え微生物およびそれらの微生物を用いるマクロライド系化合物の製造方法 |
| US7816401B2 (en) | 2005-10-13 | 2010-10-19 | Eisai R&D Management Co., Ltd. | Process for total synthesis of pladienolide B and pladienolide D |
| WO2008016187A1 (fr) | 2006-08-02 | 2008-02-07 | Eisai R & D Management Co., Ltd. | Molécule cible de pladiénolide, composé capable de se lier à la molécule cible et procédé de criblage pour rechercher le composé |
| EP3514154A1 (en) | 2014-05-15 | 2019-07-24 | Eisai R&D Management Co., Ltd. | Pladienolide pyridine compounds and methods of use |
| WO2015175594A1 (en) | 2014-05-15 | 2015-11-19 | Eisai R&D Management Co., Ltd. | Pladienolide pyridine compounds and methods of use |
| WO2017040526A2 (en) | 2015-09-01 | 2017-03-09 | Eisai R&D Management Co., Ltd. | Splice variants associated with neomorphic sf3b1 mutants |
| EP3910073A1 (en) | 2015-09-01 | 2021-11-17 | Eisai R&D Management Co., Ltd. | Splice variants associated with neomorphic sf3b1 mutants |
| US11761045B2 (en) | 2015-09-01 | 2023-09-19 | Eisai R&D Management Co., Ltd. | Splice variants associated with neomorphic SF3B1 mutants |
| US10889866B2 (en) | 2015-09-01 | 2021-01-12 | Eisai R&D Management Co., Ltd. | Splice variants associated with neomorphic SF3B1 mutants |
| WO2017087667A1 (en) | 2015-11-18 | 2017-05-26 | Eisai R&D Management Co., Ltd. | A solid state form of pladienolide pyridine compounds and methods of use |
| WO2018170129A1 (en) | 2017-03-15 | 2018-09-20 | Eisai Co., Ltd | Spliceosome mutations and uses thereof |
| WO2019089641A1 (en) | 2017-10-31 | 2019-05-09 | Eisai R&D Management Co., Ltd. | Combination comprising at least one spliceosome modulator and at least one inhibitor chosen from bcl2 inhibitors, bcl2/bclxl inhibitors, and bclxl inhibitors and methods of use |
| WO2019199667A2 (en) | 2018-04-09 | 2019-10-17 | Keaney Gregg F | Certain pladienolide compounds and methods of use |
| WO2019200100A1 (en) | 2018-04-12 | 2019-10-17 | Andrew Cook | Pladienolide derivatives as spliceosome targeting agents for treating cancer |
| JP2021532063A (ja) * | 2018-06-01 | 2021-11-25 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | スプライシング調節薬抗体−薬物コンジュゲート及びその使用方法 |
| WO2019232433A2 (en) | 2018-06-01 | 2019-12-05 | Eisai R&D Management Co., Ltd. | Methods of using splicing modulators |
| JP7390315B2 (ja) | 2018-06-01 | 2023-12-01 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | スプライシング調節薬抗体-薬物コンジュゲート及びその使用方法 |
| WO2022098712A1 (en) | 2020-11-04 | 2022-05-12 | Eisai R&D Management Co., Ltd. | Biomarkers for myelodysplastic syndrome (mds) and methods of using the same |
| WO2023131866A1 (en) | 2022-01-05 | 2023-07-13 | Eisai R&D Management Co., Ltd. | Biomarkers for myelodysplastic syndrome (mds) and methods of using the same |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101285695B1 (ko) | 2013-07-12 |
| MXPA05001104A (es) | 2005-09-08 |
| US20050245514A1 (en) | 2005-11-03 |
| RU2338741C2 (ru) | 2008-11-20 |
| EP1548121B1 (en) | 2015-04-29 |
| AU2003252445B2 (en) | 2009-09-17 |
| CA2494536A1 (en) | 2004-02-05 |
| CA2494536C (en) | 2011-10-04 |
| JPWO2004011661A1 (ja) | 2005-11-24 |
| EP1548121A1 (en) | 2005-06-29 |
| NO20050921L (no) | 2005-04-28 |
| RU2005105312A (ru) | 2005-11-20 |
| NZ537830A (en) | 2007-12-21 |
| US7256178B2 (en) | 2007-08-14 |
| CN1671856B (zh) | 2010-05-12 |
| JP4328293B2 (ja) | 2009-09-09 |
| BR0313039A (pt) | 2005-08-09 |
| CN1671856A (zh) | 2005-09-21 |
| KR20050044898A (ko) | 2005-05-13 |
| AU2003252445A1 (en) | 2004-02-16 |
| EP1548121A4 (en) | 2010-11-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2004011661A1 (ja) | 新規生理活性物質 | |
| JP4491341B2 (ja) | 新規生理活性物質 | |
| JP4459051B2 (ja) | 新規生理活性物質 | |
| CA2436667C (en) | Novel bioactive substance | |
| AU2004292634A1 (en) | DNA participating in hydroxylation of macrolide compound |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2003252445 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 537830 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2494536 Country of ref document: CA Ref document number: 296/DELNP/2005 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003771459 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2005/001104 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004524326 Country of ref document: JP Ref document number: 1-2005-500189 Country of ref document: PH Ref document number: 10522731 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 20038183129 Country of ref document: CN Ref document number: 1020057001794 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1200500229 Country of ref document: VN |
|
| ENP | Entry into the national phase |
Ref document number: 2005105312 Country of ref document: RU Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020057001794 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003771459 Country of ref document: EP |