WO2000009108A2 - Compositions and uses for vision and memory disorders - Google Patents
Compositions and uses for vision and memory disorders Download PDFInfo
- Publication number
- WO2000009108A2 WO2000009108A2 PCT/US1999/018241 US9918241W WO0009108A2 WO 2000009108 A2 WO2000009108 A2 WO 2000009108A2 US 9918241 W US9918241 W US 9918241W WO 0009108 A2 WO0009108 A2 WO 0009108A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- straight
- branched chain
- alkyl
- alkenyl
- phenyl
- Prior art date
Links
- 0 C*C(CN(C(C)C(**IC)=*)C(C(*)=O)=O)C1CC1 Chemical compound C*C(CN(C(C)C(**IC)=*)C(C(*)=O)=O)C1CC1 0.000 description 3
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/401—Proline; Derivatives thereof, e.g. captopril
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/4025—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil not condensed and containing further heterocyclic rings, e.g. cromakalim
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/426—1,3-Thiazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4439—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. omeprazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/12—Ophthalmic agents for cataracts
Definitions
- This invention relates to pharmaceutical compositions and methods for treating vision loss, preventing vision degeneration, and promoting vision regeneration ("neopsis") using low molecular weight, small molecule derivatives.
- the visual system is composed of the eyes, ocular adnexa and the visual pathways. Dysfunction of the visual system may lead to permanent or temporary visual impairment, i.e. a deviation from normal in one or more functions of the eye. Visual impairment manifests itself in various ways and includes a broad range of visual dysfunctions and disturbances.
- these dysfunctions and disturbances include partial or total loss of vision, the need for correction of visual acuity for objects near and far, loss of visual field, impaired ocular motility without diplopia (double vision) , impaired or skewed color perception, limited adaptation to light and dark, diminished accommodation, metamorphopsic distortion, impaired binocular vision, paresis of accommodation, iridoplegia, entropion, ectropion, epiphora, lagophthalmos , and scarring.
- PDR Physicians ' Desk Reference
- the visual system may be adversely affected by various ophthalmologic disorders, diseases, injuries, and complications, including, without limitation, genetic disorders; [non-genetic disorders;] disorders associated with aging or degenerative diseases; disorders correlating to physical injury to the eye, head, or other parts of the body resulting from external forces; disorders resulting from environmental factors; disorders resulting from a broad range of diseases; and combinations of any of the above .
- the visual system is a complex system composed of numerous components. Visual impairment can involve the entire visual system, any one component, or any combination of components, depending upon the precise nature of the circumstances.
- the eye is composed of a lens, which is suspended in the zonules of Zinn and is focused by the ciliary body.
- the ciliary body also secretes aqueous humor, which fills the posterior chamber, passes through the pupil into the anterior chamber, then drains primarily via the canal of Schlemm.
- the iris regulates the quantity of light entering the eye by adjusting the size of its central opening, the pupil.
- a visual image is focused onto the retina, the fovea centralis being the retinal area of sharpest visual acuity.
- the conjunctiva is the mucus membrane which lines the eyelids and the eyeball, and ends abruptly at the limbus conjunctivae, the edge of the conjunctiva overlapping the cornea.
- the cornea is the clear, transparent anterior portion of the fibrous coat of the eye; it is important in light refraction and is covered with an epithelium that differs in many respects from the conjunctival epithelium.
- the retina is the innermost, light sensitive portion of the eye, containing two types of photoreceptors, cones, which are responsible for color vision in brighter light, and rods, which are essential for vision in dim light but do not perceive colors.
- light After light passes through the cornea, lens system, and the vitreous humor, it enters the retina from the inside; that is, it passes through the ganglion cells and nerve fibers, the inner and outer plexiform layers, the inner and outer nuclear layers, and the internal and external limiting membranes before it finally reaches the layer of photoreceptors located near the outside of the retina, just inside the outermost pigment epithelium layer.
- the cells of the pigment epithelium layer act as an anatomical barrier to liquids and substances located outside of the eye, forming the "blood-retina" barrier, and provide nourishment, oxygen, a source of functionally useful substances like vitamin A, and phagocytosis of decomposition products to photoreceptor cells. There is no anatomical connection between the pigment epithelium and the photoreceptor layer, permitting separation of the layers in some pathological situations.
- rods or cones When rods or cones are excited by light, signals are transmitted through successive neurons in the retina itself, into the optic nerve fibers, and ultimately to the cerebral cortex. Both rods and cones contain molecules that decompose on exposure to light and, in the process, excite the nerve fibers leading from the eye.
- the molecule in rods is rhodopsin.
- the three light-sensitive molecules in cones, collectively called iodopsin have compositions only slightly different from that of rhodopsin and are maximally excited by red, blue, or green light, respectively.
- rods nor cones generate action potentials. Rather, the light -induced membrane hyperpolarization generated in the outer, photosensitive segment of a rod or cone cell is transmitted from the outer segment through the inner segment to the synaptic body by direct conduction of the electrical voltage itself, a process called electrotonic conduction. At the synaptic body, the membrane potential controls the release of an unknown transmitter molecule. In low light, rod and cone cell membranes are depolarized and the rate of transmitter release is greatest. Light-induced hyperpolarization causes a marked decrease in the release of transmitter molecules .
- the transmitters released by rod and cone cells induce signals in the bipolar neurons and horizontal cells.
- the signals in both these cells are also transmitted by electrotonic conduction and not by action potential.
- the rod bipolar neurons connect with as many as 50 rod cells, while the dwarf and diffuse bipolar cells connect with one or several cone cells.
- a depolarizing bipolar cell is stimulated when its connecting rods or cones are exposed to light. The release of transmitter molecules inhibits the depolarizing bipolar cell. Therefore, in the dark, when the rods and cones are secreting large quantities of transmitter molecules, the depolarizing bipolar cells are inhibited. In the light, the decrease in release of transmitter molecules from the rods and cones reduces the inhibition of the bipolar cell, allowing it to become excited. In this manner, both positive and negative signals can be transmitted through different bipolar cells from the rods and cones to the amacrine and ganglion cells.
- horizontal cells project horizontally in the retina, where they may synapse with rods, cones, other horizontal cells, or a combination of cells types.
- the function of horizontal cells is unclear, although some mechanism in the convergence of photoreceptor signaling has been postulated.
- A-type ganglion cells predominately connect with rod bipolar cells
- B-type ganglion cells predominately connect with dwarf and diffuse bipolar cells. It appears that A-type ganglion cells are sensitive to contrast, light intensity, and perception of movement, while B-type ganglion cells appear more concerned with color vision and visual acuity.
- the Amacrine cells horizontally synapse with several to many other cells, in this case bipolar cells, ganglion cells, and other Amacrine cells. The function of Amacrine cells is also unclear.
- the axons of ganglion cells carry signals into the nerve fiber layer of the eye, where the axons converge into fibers which further converge at the optic disc, where they exit the eye as the optic nerve.
- the ganglion cells transmit their signals through the optic nerve fibers to the brain in the form of action potentials. These cells, even when unstimulated, transmit continuous nerve impulses at an average, baseline rate of about 5 per second.
- the visual signal is superimposed onto this baseline level of ganglion cell stimulation. It can be either an excitatory signal, with the number of impulses increasing above the baseline rate, or an inhibitory signal, with the number of nerve impulses decreasing below the baseline rate.
- the eye As part of the central nervous system, the eye is in some ways an extension of the brain; as such, it has a limited capacity for regeneration. This limited regeneration capacity further complicates the challenging task of improving vision, resolving dysfunction of the visual system, and/or treating or preventing ophthalmologic disorders.
- Many disorders of the eye such as retinal photic injury, retinal ischemia-induced eye injury, age-related macular degeneration, free radical -induced eye diseases, as well as numerous other disorders, are considered to be entirely untreatable.
- Other ophthalmologic disorders e.g., disorders causing permanent visual impairment, are corrected only by the use of ophthalmic devices and/or surgery, with varying degrees of success.
- the immunosuppressant drugs FK506, rapamycin, and cyclosporin are well known as potent T-cell specific immunosuppressants, and are effective against autoimmunity, transplant or graft rejection, inflammation, allergic responses, other autoimmune or immune-mediated diseases, and infectious diseases. It has been disclosed that application of Cyclosporin, FK-506, Rapamycin, Buspirone, Spiperone, and/or their derivatives are effective in treating some ophthalmologic disorders of these types.
- Several ophthalmologic disorders or vision problems are known to be associated with autoimmune and immunologically-mediated activities; hence, immunomodulatory compounds are expected to demonstrate efficacy for treating those types of ophthalmologic disorders or vision problems.
- non-immunosuppressant small molecule compounds, and compositions and methods for use of such compounds, that are useful in improving vision; preventing, treating, and/or repairing visual impairment or dysfunction of the visual system; and preventing, treating, and/or resolving ophthalmologic disorders .
- non- immunosuppressive compounds disclosing methods of use for permitting or promoting wound healing (whether from injury or surgery) ; controlling intraocular pressure (often resulting from glaucoma) ; controlling neurodegenerative eye disorders, including damage or injury to retinal neurons, damage or injury to retinal ganglion cells, and macular degeneration; stimulating neurite outgrowth; preventing or reducing oxidative damage caused by free radicals; and treating impaired oxygen and nutrient supply, as well as impaired waste product removal, resulting from low blood flow.
- non- immunosuppressive substances fall into one of two general categories: naturally occurring molecules, such as proteins, glycoproteins, peptides, hormones, and growth factors; and synthetic molecules.
- GDNF glial cell line derived neurotrophic factor
- U.S. Patent No. 5,667,968 discloses the use of a variety of neurotrophic proteins, including brain-derived neurotrophic factor, ciliary neurotrophic factor, neurotrophin-3 or neurotrophin-4 , acidic or basic fibroblast growth factors, interleukin, tumor necrosis factor- , insulin-like growth factor-2 and other growth factors.
- Wong et al . U.S. Patent No. 5,632,984, discloses the use of interferons, especially interferon ⁇ -2a, for treating the symptoms of macular degeneration by reducing hemorrhage and limiting neovascularization.
- Wallace et al . U.S. Patent No. 5,441,937, discloses the use of a lung-derived neurotrophic factor (NTF) to maintain the functionality of ciliary ganglion and parasympathetic neuron cells.
- NTF lung-derived neurotrophic factor
- a key characteristic of factors derived from specific cell lines is their localization to specific cell lines or tissues; systemic treatment with these molecules would run a substantial risk of unintended, and potentially dangerous, effects in cell lines where the genes encoding these molecules are inactive. Similarly, hormones and growth factors often activate a large number of genes in many cell lines; again, non-localized application of these molecules would run a substantial risk of provoking an inappropriate, and potentially dangerous, response.
- synthetic molecules most of the patented compounds are immunosuppressive and disclose uses in treating inflammatory, autoimmune, and allergic responses, as discussed above. A few others are non-immunosuppressive and claim the ability to treat cellular degeneration, and in some cases promote cellular regeneration, most often in the context of their antioxidant properties.
- Tso et al . U.S. Patent No. 5,527,533, discloses the use of astaxanthin, a carotenoid antioxidant, for preventing or reducing photoreceptor damage resulting from the presence of free radicals.
- Babcock et al . U.S. Patent No. 5,252,319 discloses the use of antioxidant aminosteroids for treating eye disease and injury, by increasing resistance to oxidative damage.
- Freeman U.S. Patent No. 5,468,752 discloses the use of the antiviral phosphonylmethoxyalkylcytosines to reduce abnormally increased intraocular pressure.
- these compounds may be differentiated from the non-immunosuppressive compounds used to treat vision disorders by their novel small molecule structure and their lack of general, systemic effects.
- Naturally occurring hormones, growth factors, cytokines, and signaling molecules are generally multifunctional and activate many genes in diverse cell lines.
- the present compounds do not, thus avoiding the unexpected, and potentially dangerous, side effects of systemic use.
- the present compounds also avoid the potential unexpected side effects of introducing cell line-specific molecules into other cell lines were they do not naturally occur.
- the present invention relates to the surprising discovery that non-immunosuppressive immunophilin ligands, i.e. inhibitors or binding agents, may be useful for treating a vision disorder, improving vision, treating memory impairment or enhancing memory performance in an animal . Accordingly, novel compositions and methods of using non- immunosuppressive immunophilin ligands are provided. A preferred feature of the compounds of the present invention is that they do not exert any significant immunosuppressive activity.
- Preferred embodiments of this invention also include methods and compositions wherein the non- immunosuppressive immunophilin ligand has an affinity for FKBP-type immunophilins, and in particular FKBP-12.
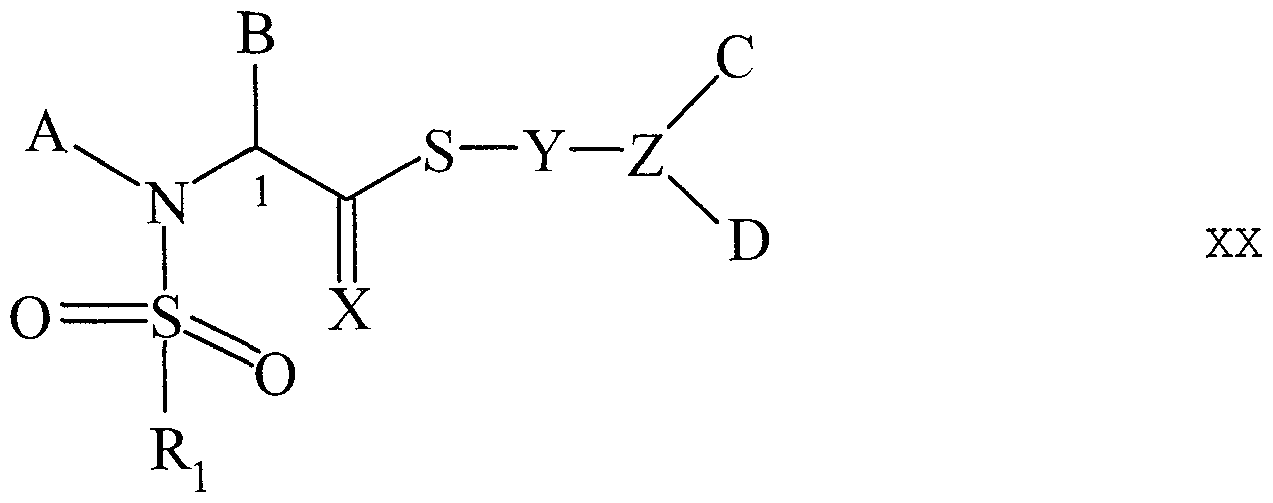
- Preferred FKBP-type non- immunosuppressive immunophilin ligands include without limitation small molecule heterocyclic ring compounds having a first and second substituent group attached thereto wherein the first substituent group comprises i) an acidic moiety or ii) an alkyl, alkenyl, alkylaryl, alkenylaryl or group otherwise exemplified herein which is linked to the heterocyclic ring by an ester, thioester, amide, amine, ketone linkage, or a variation as disclosed herein, and wherein the second substituent group comprises an alkyl, alkenyl, alkylaryl, alkenylaryl, or group otherwise exemplified herein which is linked to the heterocyclic ring by a diketo, thiocarbonyl , carbamate, urea, sulfonyl, or a linkage as exemplified herein.
- Preferred embodiments of the invention include methods and compositions using a compound selected from formula
- Figure 2 shows that GPI 1046 prevents degeneration of optic nerve axons and myelin following retinal ischemia.
- Figure 3 shows that GPI 1046 provides moderate protection against retinal ganglion cell death after optic nerve transection.
- Figure 4 shows that GPI 1046 treatment duration significantly affects the process of optic nerve axonal degeneration after transection.
- Figure 5 shows that GPI 1046 treatment produces a greater effect on optic nerve axons than ganglion cell bodies.
- Figure 6 shows that GPI 1046 treatment for 28 days after optic nerve transection prevents myelin degeneration in the proximal stump.
- Figure 7 shows that FKBP-12 immunohistochemistry labels oligodendroglia (large dark cells with fibrous processes) , the cells which produce myelin, located between the fascicles of optic nerve fibers, and also some optic nerve axons.
- oligodendroglia large dark cells with fibrous processes
- Figure 8 shows GPI 1046 treatment for 28 days after optic nerve transection prevents myelin degeneration in the distal stump .
- Figure 9 shows that 28 day treatment with GPI 1046 treatment beginning 8 weeks after onset of streptozotocin induced diabetes decreases the extent of neovascularization in the inner and outer retina and protects neurons in the inner nuclear layer (INL) and ganglion cell layer (GCL) from degeneration.
- INL inner nuclear layer
- GCL ganglion cell layer
- Eye refers to the anatomical structure responsible for vision in humans and other animals, and encompasses the following anatomical structures, without limitation: lens, vitreous body, ciliary body, posterior chamber, anterior chamber, pupil, cornea, iris, canal of Schlemm, zonules of Zinn, limbus, conjunctiva, choroid, retina, central vessels of the retina, optic nerve, fovea centralis, macula lutea, and sclera.
- GPI 1046 refers to 3- (3-pyridyl) -1-propyl (2s) -1- (3,3- dimethyl-1, 2-dioxopentyl ) -2-pyrrolidinecarboxylate, a compound of formula
- 'GPI 1312 refers to a compound of formula
- 'GPI 1511 refers to a compound of formula
- 'GPI 1234" refers to a compound of formula
- “Isomers” refer to different compounds that have the same molecular formula. “Stereoisomers” are isomers that differ only in the way the atoms are arranged in space. "Enantiomers” are a pair of stereoisomers that are non- superimposable mirror images of each other. “Diastereoisomers” are stereoisomers which are not mirror images of each other. “Racemic mixture” means a mixture containing equal parts of individual enantiomers. “Non- racemic mixture” is a mixture containing unequal parts of individual enantiomers or stereoisomers.
- “Enhancing memory performance” refers to improving or increasing the mental faculty by which to register, retain or recall past experiences, knowledge, ideas, sensations, thoughts or impressions.
- “Memory impairment” refers to a diminished mental registration, retention or recall of past experiences, knowledge, ideas, sensations, thoughts or impressions. Memory impairment may affect short and long-term information retention, facility with spatial relationships, memory (rehearsal) strategies, and verbal retrieval and production. Common causes of memory impairment are age, severe head trauma, brain anoxia or ischemia, alcoholic-nutritional diseases, and drug intoxications. Examples of memory impairment include, without limitation, benign forgetfulness, amnesia and any disorder in which memory deficiency is present, such as Korsakoff ' s amnesic psychosis, dementia and learning disorders.
- Neopsic factors or “neopsics” refers to compounds useful in treating vision loss, preventing vision degeneration, or promoting vision regeneration.
- Neopsis refers to the process of treating vision loss, preventing vision degeneration, or promoting vision regeneration.
- Optological refers to anything about or concerning the eye, without limitation, and is used interchangeably with “ocular,” “ophthalmic,” “ophthalmologic,” and other such terms, without limitation.
- “Pharmaceutically acceptable salt, ester, or solvate” refers to a salt, ester, or solvate of a subject compound which possesses the desired pharmacological activity and which is neither biologically nor otherwise undesirable.
- a salt, ester, or solvate can be formed with inorganic acids such as acetate, adipate, alginate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, citrate, camphorate, camphorsulfonate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, fumarate, glucoheptanoate, gluconate, glycerophosphate , hemisulfate, heptanoate, hexanoate, hydrochloride, hydrobromide, hydroiodide, 2- hydroxyethanesulfonate, lactate, maleate, methanesulfonate, naphthylate, 2-naphthalenesulfonate, nicotinate, oxalate, sulfate, thiocyanate, tosylate and undecanoate .
- base salts, esters, or solvates include ammonium salts; alkali metal salts, such as sodium and potassium salts; alkaline earth metal salts, such as calcium and magnesium salts; salts with organic bases, such as dieyelohexylamine salts; N-methyl-D-glucamine; and salts with amino acids, such as arginine, lysine, and so forth.
- the basic nitrogen- containing groups can be quarternized with such agents as lower alkyl halides, such as methyl, ethyl, propyl , and butyl chlorides, bromides, and iodides; dialkyl sulfates, such as dimethyl, diethyl, dibutyl , and diamyl sulfates; long chain halides, such as decyl , lauryl, myristyl, and stearyl chlorides, bromides, and iodides; aralkyl halides, such as benzyl and phenethyl bromides; and others. Water or oil- soluble or dispersible products are thereby obtained.
- lower alkyl halides such as methyl, ethyl, propyl , and butyl chlorides, bromides, and iodides
- dialkyl sulfates such as dimethyl, diethyl, dibutyl , and diamy
- Preventing vision degeneration refers to the ability to prevent degeneration of vision in patients newly diagnosed as having a degenerative disease affecting vision, or at risk of developing a new degenerative disease affecting vision, and for preventing further degeneration of vision in patients who are already suffering from or have symptoms of a degenerative disease affecting vision.
- “Promoting vision regeneration” refers to maintaining, improving, stimulating or accelerating recovery of, or revitalizing one or more components of the visual system in a manner which improves or enhances vision, either in the presence or absence of any ophthalmologic disorder, disease, or injury.
- Treating refers to: (i) preventing a disease and/or condition from occurring in a subject which may be predisposed to the disease and/or condition but has not yet been diagnosed as having it;
- Vision refers to the ability of humans and other animals to process images, and is used interchangeably with “sight”, “seeing”, and other such terms, without limitation.
- Vision disorder refers to any disorder that affects or involves vision, including without limitation visual impairment, orbital disorders, disorders of the lacrimal apparatus, disorders of the eyelids, disorders of the conjunctiva, disorders of the cornea, cataracts, disorders of the uveal tract, disorders of the retina, disorders of the optic nerve or visual pathways, free radical induced eye disorders and diseases, immunologically-mediated eye disorders and diseases, eye injuries, and symptoms and complications of eye disease, eye disorder, or eye injury.
- Visual impairment refers to any dysfunction in vision including without limitation disturbances or diminution in vision (e.g., binocular, central, peripheral, scotopic) , visual acuity for objects near and far, visual field, ocular motility, color perception, adaptation to light and dark, accommodation, refraction, and lacrimation. See Physician's Desk Reference (PDR) for Ophthalmology, 16 th Edition, 6:47 (1988) .
- PDR Physician's Desk Reference
- the present invention relates to a method of treating a vision disorder, improving vision, treating memory impairment, or enhancing memory performance in an animal, which comprises administering to said animal an effective amount of a derivative.
- inventive methods are particularly useful for treating various eye disorders including but not limited to visual disorders, diseases, injuries, and complications, genetic disorders; disorders associated with aging or degenerative vision diseases; vision disorders correlating to physical injury to the eye, head, or other parts of the body resulting from external forces; vision disorders resulting from environmental factors; vision disorders resulting from a broad range of diseases; and combinations of any of the above .
- compositions and methods of the present invention are useful for improving vision, or correcting, treating, or preventing visual (ocular) impairment or dysfunction of the visual system, including permanent and temporary visual impairment, without limitation.
- the present invention is also useful in preventing and treating ophthalmologic diseases and disorders, treating damaged and injured eyes, and preventing and treating diseases, disorders, and injuries which result in vision deficiency, vision loss, or reduced capacity to see or process images, and the symptoms and complications resulting from same.
- the eye diseases and disorders which may be treated or prevented by the compositions and methods of the present invention are not limited with regard to the cause of said diseases or disorders. Accordingly, said compositions and methods are applicable whether the disease or disorder is caused by genetic or environmental factors, as well as any other influences.
- compositions and methods of the present invention are particularly useful for eye problems or vision loss or deficiency associated with all of the following, without limitation: aging, cellular or physiological degeneration, central nervous system or neurological disorder, vascular defects, muscular defects, and exposure to adverse environmental conditions or substances.
- compositions and methods of the present invention are particularly useful in correcting, treating, or improving visual impairment, without limitation.
- Visual impairment in varying degrees occurs in the presence of a deviation from normal in one or more functions of the eye, including (1) visual acuity for objects at distance and near; (2) visual fields; and (3) ocular motility without diplopia.
- PDR Physicians ' Desk Reference
- Vision is imperfect without the coordinated function of all three. Id .
- compositions and methods of use are also useful in correcting, treating, or improving other ocular functions including, without limitation, color perception, adaptation to light and dark, accommodation, metamorphopsia, and binocular vision.
- the compositions and methods of use are particularly useful in treating, correcting, or preventing ocular disturbances including, without limitation, paresis of accommodation, iridoplegia, entropion, ectropion, epiphora, lagophthalmos , scarring, vitreous opacities, non-reactive pupil, light scattering disturbances of the cornea or other media, and permanent deformities of the orbit.
- compositions and methods of use of the present invention are also highly useful in improving vision and treating vision loss. Vision loss ranging from slight loss to absolute loss may be treated or prevented using said compositions and methods of use. Vision may be improved by the treatment of eye disorders, diseases, and injuries using the compositions and methods of the invention. However, improvements in vision using the compositions and methods of use are not so limited, and may occur in the absence of any such disorder, disease, or injury.
- compositions and methods of the present invention are also useful in the treatment or prevention of the following non-limiting exemplary diseases and disorders, and symptoms and complications resulting therefrom.
- Vision disorders include but are not limited to the following : visual impairment, such as diminished visual acuity for objects near and far, visual fields, and ocular motility; orbital disorders, such as orbital cellulitis, periorbital cellulitis, cavernous sinus thrombosis, and exophthalmos (proptosis) ; disorders of the lacrimal apparatus, such as dacryostenosis, congenital dacryostenosis, and dacryocystitis (acute or chronic) ; disorders of the eyelids, such as lid edema, blepharitis, ptosis, Bell's palsy, blepharospasm, hordeolum (stye) , external hordeolum, internal hordeolum (meibomian stye) , chalazion, entropion (inversion of the eyelid) , ectropion (eversion of the eyelid) , tumor
- compositions and methods of the present invention are also useful in the treatment of the following non- limiting eye injuries, and symptoms and complications resulting therefrom: conjunctival and corneal foreign body injuries, corneal abrasion, intraocular foreign body injuries, lacerations, lid lacerations, contusions, lid contusions (black eye), trauma to the globe, laceration of the iris, cataract, dislocated lens, glaucoma, vitreous hemorrhage, orbital -floor fractures, retinal hemorrhage or detachment, and rupture of the eyeball, anterior chamber hemorrhage (traumatic hyphema) , burns, eyelid burns, chemical burns, chemical burns of the cornea and conjunctiva, and ultraviolet light burns (sunburn) . See The Merck Manual , Sixteenth Edi tion, 217:2364-2365 (1992).
- compositions and methods of the present invention are also useful in treating and/or preventing the following non-limiting exemplary symptoms and complications of eye disease, eye disorder or eye injury: subconjunctival hemorrhages, vitreous hemorrhages, retinal hemorrhages, floaters, retinal detachments, photophobia, ocular pain, scotomas (negative and positive) , errors of refraction, emmetropia, ametropia, hyperopia (farsightedness) , myopia
- the derivative may be administered in combination with an effective amount of one or more factor (s) useful in treating vision disorder, improving vision, treating memory impairment, or enhancing memory performance.
- the factor (s) to be combined with the derivative is/are selected from the group consisting of immunosuppressants for treating autoimmune, inflammatory, and immunologically-mediated disorders; wound healing agents for treating wounds resulting from injury or surgery; antiglaucomatous medications for treating abnormally elevated intraocular pressure; neurotrophic factors and growth factors for treating neurodegenerative disorders or stimulating neurite outgrowth; compounds effective in limiting or preventing hemorrhage or neovascularization for treating macular degeneration; and antioxidants for treating oxidative damage to eye tissues.
- the present invention also relates to a pharmaceutical composition
- a pharmaceutical composition comprising:
- the derivative may be administered in combination with an effective amount of one or more factor (s) useful in treating vision disorders, improving vision, treating memory impairment, or enhancing memory performance.
- the non- immunosuppressive neuroimmunophilin FKBP ligand used in the method and pharmaceutical composition of the present invention is a low molecular weight, small molecule compound having an affinity for an FKBP-type immunophilin, such as FKBP12.
- FKBP-type immunophilin such as FKBP12.
- FKBP12 FKBP12
- the compound binds to an FKBP-type immunophilin, it has been found to inhibit the prolyl- peptidyl cis- trans isomerase activity, or rotamase, activity of the binding protein.
- the compound is devoid of any significant immunosuppressive activity.
- non- immunosuppressive neuroimmunophilin FKBP ligand examples include a non- immunosuppressive neuroimmunophilin FKBP ligand that may be used in the inventive method and pharmaceutical composition are set forth below.
- the non- immunosuppressive neuroimmunophilin FKBP ligand may be a compound of formula I
- a and B together with the nitrogen and carbon atoms to which they are respectively attached, form a 5-7 membered saturated or unsaturated heterocyclic ring containing one or more heteroatom (s) independently selected from the group consisting of O, S, SO, S0 2 , N, NH, and NR 2 ;
- X is either O or S;
- Z is either S, CH 2 , CHR X or CR 1 R 3 ;
- W and Y are independently 0, S, CH 2 or H 2 ;
- R x and R 3 are independently straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is substituted with one or more substituent (s) independently selected from the group consisting of (Ar -, straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl substituted with
- R 2 is either C x -C 9 straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, or Ar 1; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of C 1 -C 4 straight or branched chain alkyl, C 2 -C 4 straight or branched chain alkenyl, and hydroxy; and
- Ar-, . and Ar 2 are independently an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein said ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxyl, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, - ⁇ - ⁇ alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino; wherein the individual ring size is 5-8 members; and wherein the heterocyclic ring contains 1-6 heteroatom (s) independently selected from the group consisting of 0, N, and S.
- Useful carbo- and heterocyclic rings include without limitation phenyl, benzyl, naphthyl, indenyl , azulenyl, fluorenyl, anthracenyl, indolyl , isoindolyl, indolinyl, benzofuranyl , benzothiophenyl , indazolyl, benzimidazolyl , benzthiazolyl , tetrahydrofuranyl , tetrahydropyranyl, pyridyl, pyrrolyl, pyrrolidinyl , pyridinyl, pyrimidinyl , purinyl , quinolinyl, isoquinolinyl , tetrahydroquinolinyl , quinolizinyl , furyl , thiophenyl , imidazolyl , oxazolyl , benzoxazolyl , thiazo
- the non- immunosuppressive neuroimmunophilin FKBP ligand may also be a compound of formula II
- n 1 or 2
- X is 0 or S
- Z is selected from the group consisting of S, CH 2 , CHR 1; and CR X R 3 ;
- R x and R 3 are independently selected from the group consisting of C- L -C S straight or branched chain alkyl, C 2 -C 5 straight or branched chain alkenyl, and Ar x , wherein said alkyl, alkenyl or Ar x is unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, nitro, Cj-Cg straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, hydroxy, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, amino , and Ar x ;
- R 2 is selected from the group consisting of Ci-Cg straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, and Ar 1 - and
- Ar x is phenyl, benzyl, pyridyl, fluorenyl, thioindolyl or naphthyl, wherein said Ar x is unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, trifluoromethyl, hydroxy, nitro, C- L -Cg straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino.
- substituent independently selected from the group consisting of halo, trifluoromethyl, hydroxy, nitro, C- L -Cg straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy,
- non- immunosuppressive neuroimmunophilin FKBP ligand may be a compound of formula III
- A, B, and C are independently CH 2 , 0, S, SO, S0 2 , NH or NR 2 ;
- X is O or S
- Z is S, CH 2/ CHR- L or CR ⁇ ;
- R- L and R 3 are independently straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is substituted with one or more substituent (s) independently selected from the group consisting of (Ar -, straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl substituted with (Ar -, C 3 -C 8 cycloalkyl, straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl substituted with C 3 -C 8 cycloalkyl, and Ar 2 ; n is 1 or 2;
- R 2 is either straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl or Ar x , wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of C 1 -C 4 straight or branched chain alkyl, C 2 -C 4 straight or branched chain alkenyl, and hydroxyl; and r !
- Ar 2 are independently an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein said ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxyl, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino; wherein the individual ring size is 5-8 members; and wherein the heterocyclic ring contains 1-6 heteroatom (s) independently selected from the group consisting of 0, N, and S.
- substituent independently selected from the group consisting of halo, hydroxyl, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C
- the non - immuno suppre s s ive neuroimmunophilin FKBP ligand may be a compound of formula IV
- A, B, C and D are independently CH 2 , 0, S, SO, S0 2 , NH or NR 2 ;
- Z is S, CH 2 , CHR X or CR X R 3 ;
- R x and R 3 are independently straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is substituted with one or more substituent (s) independently selected from the group consisting of (Ar -, Ci-Cg straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl substituted with (Ar -, C 3 -C 8 cycloalkyl, straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl substituted with C 3 -C 8 cycloalkyl, and Ar 2 ; n is 1 or 2 ;
- R 2 is either straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl or Ar 17 wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 1 -C 4 straight or branched chain alkyl, C 2 -C 4 straight or branched chain alkenyl , and hydroxyl ; and
- Ar x and Ar 2 are independently an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein said ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxyl, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C x -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino; wherein the individual ring size is 5-8 members; and wherein the heterocyclic ring contains 1-6 heteroatom (s) independently selected from the group consisting of 0, N, and S.
- substituent independently selected from the group consisting of halo, hydroxyl, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C x -C
- the non- immunosuppressive neuroimmunophilin FKBP ligand may further be a compound of formula V
- V is C , N, or S ;
- a and B together with V and the carbon atom to which they are respectively attached, form a 5-7 membered saturated or unsaturated heterocyclic ring which may contain, in addition to V, one or more heteroatom (s) independently selected from the group consisting of O, S, SO, S0 2 , N, NH, and NR 4 ;
- R 4 is either straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 9 cycloakyl, C 5 -C 7 cycloalkenyl, or Ar 3 , wherein R 4 is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, haloalkyl , carbonyl, carboxy, hydroxy, nitro, trifluoromethyl, C x -C 6 straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, thioalkyl, alkylthio, sulfhydryl, amino, alkylamino, aminoalkyl, aminocarboxyl , and Ar 4 ;
- Ar 3 and Ar 4 are independently an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring; wherein the individual ring size is 5-8 members; wherein said heterocyclic ring contains 1-6 heteroatom(s) independently selected from the group consisting of 0, N, and S; and
- R 17 R 2 , W, X, Y, and Z are as defined in Formula I above.
- non- immunosuppress ive neuroimmunophilin FKBP ligand may be a compound of formula VI
- a and B together with the nitrogen and carbon atoms to which they are respectively attached, form a 5-7 membered saturated or unsaturated heterocyclic ring which may contain, in addition to the nitrogen atom, one or more heteroatom (s) independently selected from the group consisting of 0, S, SO, S0 2 , N, NH, and NR 1;
- - X is O or S;
- Z is O, NH or NR 1 ;
- W and Y are independently O, S, CH 2 or H 2 ;
- Rj is C- L -Cg straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl, which is substituted with one or more substituent (s) independently selected from the group consisting of (Ar- -, straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl substituted with (Ar -, C 3 -C 8 cycloalkyl, C x -C 6 straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl substituted with C 3 -C 8 cycloalkyl, and Ar 2 ;
- n is 1 or 2;
- R 2 is either straight or branched chain alkyl, C 2 -C 9 straight or branched chain or alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, or Ar 1; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of C 1 -C 4 straight or branched chain alkyl, C 2 -C 4 straight or branched chain alkenyl, and hydroxyl; and Ar x and Ar 2 are independently an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxyl, nitro, trifluoromethyl, C 1 -C 6 straight or branched chain
- Suitable carbo- and heterocyclic rings include without limitation naphthyl, indolyl , furyl , thiazolyl, thienyl, pyridyl, quinolinyl, isoquinolinyl , fluorenyl and phenyl.
- the non- immunosuppressive neuroimmunophilin FKBP ligand may also be a compound of formula VII
- A, B and C are independently CH 2 , 0, S, SO, S0 2 , NH or NR ⁇ ;
- R ⁇ is C- L -C S straight or branched chain alkyl or C 2 -C 5 straight or branched chain alkenyl, which is substituted with one or more substituent (s) independently selected from the group consisting of (Ar x ) n and straight or branched chain alkyl or C 2 -C 3 straight or branched chain alkenyl substituted with (Ar- -; n is 1 or 2;
- R 2 is either C x -C 9 straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, or Ar 1 - and
- Ar x is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxyl, nitro, trifluoromethyl, C j -Cg straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino; wherein the individual ring size is 5-8 members; and wherein the heterocyclic ring contains 1-6 heteroatom (s) independently selected from the group consisting of 0, N, and S.
- the heterocyclic ester or amide is the Compound GPI 1572, of the formula
- A is CH 2 ;
- R- L is selected from the group consisting of 3- phenylpropyl and 3 - (3 -pyridyl ) propyl ; and R 2 is selected from the group consisting of 1,1- dimethvlpropyl , cyclohexyl, and tert-butyl . Specific examples of this embodiment are presented in TABLE D.
- non- immunosuppressive neuroimmunophilin FKBP ligand may be a compound of formula VIII
- A, B, C and D are independently CH 2 , 0, S, SO, S0 2 , NH or NR : ;
- R x is C ⁇ -C 3 straight or branched chain alkyl or C 2 -C 5 straight or branched chain alkenyl, which is substituted with one or more substituent (s) independently selected from the group consisting of (Ar x ) n and straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl substituted with (Ar -; n is 1 or 2;
- R 2 is either straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, or Ar lr - and
- Ar- L is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxyl, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C x -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino; wherein the individual ring size is 5-8 members; and wherein the heterocyclic ring contains 1-6 heteroatom (s) independently selected from the group consisting of 0, N, and S.
- substituent independently selected from the group consisting of halo, hydroxyl, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C x -C 4 alkoxy
- A is CH 2 ;
- B is CH 2 ;
- C is S, 0 or NH
- D is CH 2 ;
- R x is selected from the group consisting of 3- phenylpropyl and (3,4,5 -1rimethoxy) phenylpropyl ; and R 2 is selected from the group consisting of 1,1- dimethylpropyl , cyclohexyl, tert-butyl, phenyl, and 3,4,5- trimethoxyphenyl .
- Addi t i ona l l y , t he non - immuno suppre s s ive neuroimmunophilin FKBP ligand may be a compound of formula IX
- V is C, N, or S
- a and B together with V and the carbon atom to which they are respectively attached, form a 5-7 membered saturated or unsaturated heterocyclic ring which may contain, in addition to V, one or more heteroatom (s) independently selected from the group consisting of 0, S, SO, S0 2 , N, NH, and NR;
- R is either C x -C 9 straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 9 cycloakyl, C 5 -C 7 cycloalkenyl, or Ar 3 , wherein R is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, haloalkyl, carbonyl, carboxy, hydroxy, nitro, trifluoromethyl, Cj-Cg straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, thioalkyl, alkylthio, sulfhydryl, amino, alkylamino, aminoalkyl, aminocarboxyl , and Ar 4 ;
- Ar 3 and Ar 4 are independently an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring; wherein the individual ring size is 5-8 members; wherein said heterocyclic ring contains 1-6 heteroatom (s) independently selected from the group consisting of O, N, and S; and
- R l r R 2 , W, X, Y, and Z are as defined in Formula VI above .
- the non- immunosuppressive neuroimmunophilin FKBP ligand may further be a compound of formula X
- a and B together with the nitrogen and carbon atoms to which they are respectively attached, form a 5-7 membered saturated or unsaturated heterocyclic ring containing one or more heteroatom (s) independently selected from the group consisting of CH, CH 2 , O, S, SO, S0 2 , N, NH, and NR 1; - W i s 0 , S , CH 2 , or H 2 ;
- R is C- L -Cg straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, or Ar 1# which is optionally substituted with one or more substituent (s) independently selected from the group consisting of C- L - ⁇ alkyl, C 2 -C 4 alkenyl, hydroxy, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, and Ar 2 ;
- Ar ! and Ar 2 are independently selected from the group consisting of 1-napthyl, 2-napthyl, 1-indolyl, 2-indolyl, 2- furyl , 3-furyl, 2-thienyl, 3-thienyl, 2 -pyridyl, 3-pyridyl, 4-pyridyl and phenyl, having one or more substituent (s) independently selected from the group consisting of hydrogen, halo, hydroxy, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino;
- X is O, NH, NR lf S, CH, CR X , or CR X R 3 ;
- Y is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl, cycloalkenyl, or Ar is optionally substituted with C x -C 4 alkyl, C 2 -C 4 alkenyl, hydroxy, or carbonyl oxygen; wherein any carbon atom of said alkyl, alkenyl, cycloalkyl, cycloalkenyl, or Ar is optionally replaced with 0, NH, NR 2 , S, SO, or S
- R 2 is selected from the group consisting of hydrogen, C,_- C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, and bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Z is an aromatic amine or a tertiary amine oxidized to a corresponding N-oxide; said aromatic amine is selected from the group consisting of pyridyl, pyrimidyl , quinolinyl, or isoquinolinyl, which is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxy, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 3 straight or branched chain alken
- Ar is selected from the group consisting of pyrrolidinyl, pyridyl, pyrimidyl, pyrazyl, pyridazyl, quinolinyl, and isoquinolinyl;
- R- L and R 3 are independently hydrogen, C x -C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, or Y-Z.
- non- immunosuppressive neuroimmunophilin FKBP ligand may be a compound of formula XI
- E, F, G and J are independently CH 2 , 0, S, SO, S0 2 , NH or NR 17 -
- W is O, S, CH 2 , or H 2 ;
- R is C- L -Cg straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, or Ar x , which is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 1 -C 4 alkyl, C 2 -C 4 alkenyl, hydroxy, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, and Ar l; -
- Ar x is selected from the group consisting of 1-napthyl, 2-napthyl, 1-indolyl, 2-indolyl, 2-furyl, 3-furyl, 2-thienyl, 3-thienyl, 2 -pyridyl, 3-pyridyl, 4 -pyridyl, and phenyl, having one or more substituent (s) independently selected from the group consisting of hydrogen, halo, hydroxy, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 5 straight or branched chain alkenyl, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino;
- X is 0, NH, NR X , S, CH, CR X , or CR X R 3 ;
- Y is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C- L -Cg straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl, cycloalkenyl, or Ar is optionally substituted with C x -C 4 alkyl, C 2 -C 4 alkenyl, hydroxy, or carbonyl oxygen; wherein any carbon atom of said alkyl, alkenyl, cycloalkyl, cycloalkenyl, or Ar is optionally replaced with 0, NH, NR 2
- Z is an aromatic amine or a tertiary amine oxidized to a corresponding N-oxide; said aromatic amine is pyridyl, pyrimidyl, quinolinyl, and isoquinolinyl, which is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxy, nitro, trifluoromethyl, C- L -Cg straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino; said tertiary amine is NR 4 R 5 R 6 , wherein R 4 , R 5 , and R 6 are independently selected from the group consisting of straight or branched chain alkyl and C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkeny
- any carbon atom of said alkyl, alkenyl, cycloalkyl, cycloalkenyl, or Ar is optionally replaced with 0, NH, NR 1( S, SO, or S0 2 ;
- Ar is selected from the group consisting of pyrrolidinyl , pyridyl, pyrimidyl, pyrazyl, pyridazyl, quinolinyl, and isoquinolinyl; and
- R x and R 3 are independently hydrogen, C 1 -C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, or Y-Z.
- non- immunosuppressive neuroimmunophilin FKBP ligand may be a compound of formula XII
- E, F, and G are independently CH 2 , 0, S, SO, S0 2 , NH or NR ⁇ ;
- W is O, S, CH 2 , or H 2 ;
- R is C- L -C J straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, or Ar : , which is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 1 -C 4 alkyl, C 2 -C 4 alkenyl, hydroxy, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, and Ar x ;
- Ar x is selected from the group consisting of 1-napthyl, 2-napthyl, 1-indolyl, 2-indolyl, 2-furyl, 3-furyl, 2-thienyl, 3-thienyl, 2-pyridyl, 3-pyridyl, 4-pyridyl and phenyl , having one or more substituent (s) independently selected from the group consisting of hydrogen, halo, hydroxy, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino;
- X is 0, NH, NR 1# S, CH, CR X , or CR X R 3 ;
- Y is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl, cycloalkenyl, or Ar is optionally substituted with C x -C 4 alkyl, C 2 -C 4 alkenyl, hydroxy, or carbonyl oxygen; wherein any carbon atom of said alkyl, alkenyl, cycloalkyl, cycloalkenyl, or Ar is optionally replaced with 0, NH, NR 2 , S, SO, or S
- Z is an aromatic amine or a tertiary amine oxidized to a corresponding N-oxide; said aromatic amine is pyridyl, pyrimidyl, quinolinyl, or isoquinolinyl, which is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxy, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C x -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino; said tertiary amine is NR 4 R 5 R 6 , wherein R 4 , R 5 , and R 6 are independently selected from the group consisting of straight or branched chain alkyl and C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted
- R x and R 3 are independently hydrogen, C 1 -C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, or Y-Z.
- the non-immunosuppressive neuroimmunophilin FKBP ligand may also be a compound of formula XIII
- n 1, 2, or 3 , forming a 5-7 member heterocyclic ring
- W is O, S, CH 2 , or H 2 ;
- R is C- L -Cg straight or branched chain alkyl, C 2 -C 5 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, or Ar 1; which is optionally substituted with one or more substituent (s) independently selected from the group consisting of C x -C 4 alkyl, C 2 -C 4 alkenyl, hydroxy, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, and Ar 1( -
- Ar x is selected from the group consisting of 1-napthyl, 2-napthyl, 1-mdolyl, 2-mdolyl, 2-furyl, 3-furyl, 2-thienyl, 3-thienyl, 2-pyridyl, 3-pyridyl, 4-pyridyl and phenyl , having one or more substituent (s) independently selected from the group consisting of hydrogen, halo, hydroxy, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and ammo;
- X is O, NH, NR 1 S, CH, CR ⁇ , or CR J R J ;
- Y is a direct bond, C j ⁇ -Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl, cycloalkenyl, or Ar is optionally substituted with C x -C 4 alkyl, C 2 -C 4 alkenyl, hydroxy, or carbonyl oxygen; wherein any carbon atom of said alkyl, alkenyl, cycloalkyl, cycloalkenyl, or Ar is optionally replaced with O, NH, NR
- R 2 is selected from the group consisting of hydrogen, C ⁇ - C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl , and C 1 -C 4 bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Z is an aromatic amine or a tertiary amme oxidized to a corresponding N-oxide; said aromatic amme is pyridyl, pyrimidyl, quinolinyl, or isoquinolinyl, which is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, hydroxy, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino; said tertiary amine is NR 4 R 5 R 6 , wherein R 4 , R 5 , and R 6 are independently selected from the group consisting of straight or branched chain alkyl and C 2 -C 3 straight or branched chain alkenyl ; wherein said alkyl or alkenyl is optionally substituted
- Ar is selected from the group consisting of pyrrolidinyl, pyridyl, pyrimidyl, pyrazyl , pyridazyl , quinolinyl, and isoquinolinyl;
- R x and R 3 hydrogen, C x -C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, or Y-Z.
- Examples of the compounds of formula XIII when W is 0 are presented in TABLE VI .
- Preferred compounds of formula XIII may be selected from the group consisting of:
- Add i t i ona l l y , t he non - i mmuno s uppre s s i e neuroimmunophilin FKBP l igand may be a compound of formula XIV
- V is C, N, or S;
- a and B together with V and the carbon atom to which they are respectively attached, form a 5-7 membered saturated or unsaturated heterocyclic ring which may contain, in addition to V, one or more heteroatom (s) independently selected from the group consisting of O, S, SO, S0 2 , N, NH, and NR 7 ;
- R 7 is either C-Cg straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 9 cycloalkyl, C 5 -C 7 cycloalkenyl, or Ar 3 , wherein R 7 is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of halo, haloalkyl , carbonyl, carboxy, hydroxy, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, thioalkyl, alkylthio, sulfhydryl, amino, alkylamino, aminoalkyl, aminocarboxyl , and Ar 4 ;
- Ar 3 and Ar 4 are independently an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring; wherein the individual ring size is 5-8 members; wherein said heterocyclic ring contains 1-6 heteroatom(s) independently selected from the group consisting of O, N, and S; and
- R, W, X, Y, and Z are as defined in Formula X above.
- the non- immunosuppressive neuroimmunophilin FKBP ligand may further be a compound of formula XV
- a and B together with the nitrogen and carbon atoms to which they are respectively attached, form a 5-7 membered saturated or unsaturated heterocyclic ring which may contain, in addition to the nitrogen atom, one or more additional heteroatom (s) independently selected from the group consisting of 0, S, SO, S0 2 , N, NH, and NR 3 ;
- X is either 0 or S;
- Y is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl , sulfhydryl, thioalkyl , sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3 , S, SO, or S0 2 ; R 3 is selected from the group consisting of hydrogen, C 1 - C 6 straight or branched chain alkyl, C 3 -C 6 straight or branched chain alkenyl or alkynyl , and
- Ar is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of alkylamino, amido, amino, aminoalkyl, azo, benzyloxy, C 1 -C 9 straight or branched chain alkyl, alkoxy, C 2 -C 9 alkenyloxy, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, carbonyl, carboxy, cyano, diazo, ester, formanilido, halo, haloalkyl, hydroxy, imino, isocyano, isonitrilo, nitrilo, nitro, nitroso, phenoxy, sulfhydryl, sulfonylsulf
- Z is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3 , S, SO, or S0 2 ;
- C and D are independently hydrogen, Ar, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 3 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is optionally substituted with C-Cg alkyl, C 2 -C 5 alkenyl, hydroxy, amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, or sulfonyl; wherein any carbon atom of said al
- Useful carbo- and heterocyclic rings include without limitation phenyl, benzyl, naphthyl, indenyl , azulenyl, fluorenyl, anthracenyl , indolyl , isoindolyl, indolinyl, benzofuranyl , benzothiophenyl , indazolyl, benzimidazolyl , benzthiazolyl , tetrahydrofuranyl, tetrahydropyranyl , pyridyl, pyrrolyl , pyrrolidinyl , pyridinyl , pyrimidinyl, purinyl , quinol inyl , isoquinolinyl , tetrahydroquinolinyl , quinolizinyl , furyl , thiophenyl , imidazolyl, oxazolyl, benzoxazolyl ,
- Ar is selected from the group consisting of phenyl, benzyl, naphthyl, indolyl, pyridyl, pyrrolyl , pyrrolidinyl , pyridinyl , pyrimidinyl, purinyl , quinolinyl, isoquinolinyl, furyl , fluorenyl, thiophenyl, imidazolyl, oxazolyl, thiazolyl, pyrazolyl, and thienyl.
- non-immunosuppressive neuroimmunophilin FKBP ligand may be a compound of formula XVI
- E, F, G and J are independently CH 2 , 0, S, SO, S0 2 , NH, or NR 3 ;
- X is either 0 or S ;
- Y is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3 , S, SO, or S0 2 ;
- R 3 is selected from the group consisting of hydrogen, C x - C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, and C x -C 4 bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Ar is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of alkylamino, amido, amino, aminoalkyl, azo, benzyloxy, C- L -CC, straight or branched chain alkyl, C 1 -C 9 alkoxy, C 2 -C 9 alkenyloxy, C 2 -C
- Z is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3 , S, SO, or S0 2 ;
- C and D are independently hydrogen, Ar, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is optionally substituted with alkyl, C 2 -C 6 alkenyl, hydroxy, amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, or sulfonyl; wherein any carbon atom of said alkyl or al
- U is either O or N, provided that: when U is O, then R x is a lone pair of electrons and R 2 is selected from the group consisting of Ar, C 3 -C 8 cycloalkyl, straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of Ar and C 3 -C 8 cycloalkyl; and when U is N, then R 1 and R 2 are independently selected from the group consisting of hydrogen, Ar, C 3 - C 8 cycloalkyl, straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of Ar and C 3 -C 8 cycloalkyl; or R x and R 2 are taken
- Useful carbo- and heterocyclic rings include without limitation phenyl, benzyl, naphthyl, indenyl , azulenyl, fluorenyl, anthracenyl , indolyl, isoindolyl, indolinyl, benzofuranyl , benzothiophenyl , indazolyl, benzimidazolyl , benzthiazolyl, tetrahydrofuranyl , tetrahydropyranyl, pyridyl, pyrrolyl, pyrrolidinyl , pyridinyl , pyrimidinyl, purinyl , quinolinyl, isoquinolinyl, tetrahydroquinolinyl , quinolizinyl , furyl , thiophenyl, imidazolyl, oxazolyl, benzoxazolyl , thiazolyl, iso
- Ar is selected from the group consisting of phenyl, benzyl, naphthyl, pyrrolyl, pyrrolidinyl , pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, furyl , thiophenyl, imidazolyl, oxazolyl, thiazolyl, pyrazolyl, and thienyl.
- the non- immunosuppressive neuroimmunophilin FKBP ligand may also be a compound of formula XVII
- E, F, and G are independently CH 2 , 0, S, SO, S0 2 , NH, and NR 3 ;
- X is either 0 or S
- Y is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3 , S, SO, or S0 2 ;
- R 3 is selected from the group consisting of hydrogen, C x - C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, and Ci- j bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Ar is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of alkylamino, amido, amino, aminoalkyl, azo, benzyloxy, straight or branched chain alkyl, alkoxy, C 2 -C 9 alkenyloxy, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, carbonyl, carboxy, cyano, diazo, ester, formanilido, halo, haloalkyl, hydroxy, imino, isocyano, isonitrilo, nitrilo, nitro, nitroso, phenoxy, sulfhydryl, sulfonylsulfoxy, thio,
- Z is a direct bond, C- L -Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with O, NH, NR 3 , S, SO, or S0 2 ;
- C and D are independently hydrogen, Ar, Ci-Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is optionally substituted with alkyl, C 2 -C 6 alkenyl, hydroxy, amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, or sulfonyl; wherein any carbon atom of said al
- U is either 0 or N, provided that: when U is 0, then R x is a lone pair of electrons and R 2 is selected from the group consisting of Ar, C 3 ⁇ C 8 cycloalkyl, straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of Ar and C 3 -C 8 cycloalkyl; and when U is N, then R 1 and R 2 are independently selected from the group consisting of hydrogen, Ar, C 3 - C 8 cycloalkyl, straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of Ar and C 3 -C 8 cycloalkyl; or R x and R 2 are taken
- Useful carbo- and heterocyclic rings include without limitation phenyl, benzyl, naphthyl, indenyl , azulenyl, fluorenyl, anthracenyl , indolyl, isoindolyl, indolinyl, benzofuranyl , benzothiophenyl , indazolyl, benzimidazolyl , benzthiazolyl , tetrahydrofuranyl , tetrahydropyranyl , pyridyl , pyrrolyl, pyrrolidinyl , pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, tetrahydroquinolinyl , quinolizinyl , furyl , thiophenyl, imidazolyl, oxazolyl, benzoxazolyl , thiazolyl, is
- Ar is selected from the group consisting of phenyl, benzyl, naphthyl, pyrrolyl, pyrrolidinyl , pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, furyl , thiophenyl, imidazolyl, oxazolyl, thiazolyl, pyrazolyl, and thienyl.
- the non- immunosuppressive neuroimmunophilin FKBP ligand may further be a compound of formula XVIII
- n 1, 2 or 3 ;
- X is either O or S;
- Y is a direct bond, C 1 -C 6 straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with O, NH, NR 3 , S, SO, or S0 2 ;
- R 3 is selected from the group consisting of hydrogen, C x -
- Ar is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of alkylamino, amido, amino, aminoalkyl, azo, benzyloxy, straight or branched chain alkyl, C- L -Cg alkoxy, C 2 -C 9 alkenyloxy, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, carbonyl, carboxy, cyano, diazo, ester, formanilido, halo, haloalkyl, hydroxy, imino, isocyano, isonitrilo, nitrilo, nitro, nitroso, phenoxy, sulfhydryl, sulfonylsul
- Z is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with O, NH, NR 3 , S, SO, or S0 2 ;
- C and D are independently hydrogen, Ar, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is optionally substituted with C ⁇ Cg alkyl, C 2 -C 3 alkenyl, hydroxy, amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, or sulfonyl; wherein any carbon atom of said al
- Useful carbo- and heterocyclic rings include without limitation phenyl, benzyl, naphthyl, indenyl , azulenyl, fluorenyl, anthracenyl , indolyl, isoindolyl, indolinyl, benzofuranyl , benzothiophenyl , indazolyl, benzimidazolyl , benzthiazolyl, tetrahydrofuranyl , tetrahydropyranyl, pyridyl, pyrrolyl, pyrrolidinyl , pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, tetrahydroquinolinyl , quinolizinyl , furyl , thiophenyl, imidazolyl, oxazolyl, benzoxazolyl , thiazolyl, isoxazo
- Ar is selected from the group consisting of phenyl, benzyl, naphthyl, pyrrolyl, pyrrolidinyl, pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, furyl , thiophenyl, imidazolyl, oxazolyl, thiazolyl, pyrazolyl, and thienyl.
- the most preferred compounds of formula XVIII are selected from the group consisting of : 3- (3-Pyridyl) -l-propyl-2S-l- [ (2-methylbutyl) carbamoyl] pyrrolidine-2 -carboxylate ;
- FORMULA XIX Add i t i ona l l y , t he non - i mmuno s uppre s s i ve neuroimmunophilin FKBP ligand may be a compound of formula XIX
- V is C, N, or S
- Y is a direct bond, C x -C 3 straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3 , S, SO, or S0 2 ;
- R 3 is selected from the group consisting of hydrogen, C 1 - C s straight or branched chain alkyl, C 3 -C 6 straight or branched chain alkenyl or alkynyl, and C 1 -C 4 bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Ar is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) ; wherein the individual ring size is 5-8 members; wherein said heterocyclic ring contains 1-6 heteroatom (s) independently selected from the group consisting of 0, N, and S; and wherein said aromatic or tertiary alkyl amine is optionally oxidized to a corresponding N-oxide;
- Z is a direct bond, C 1 -C 6 straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3 , S, SO, or S0 2 ;
- C and D are independently hydrogen, Ar, C x -Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is optionally substituted with alkyl, C 2 -C 6 alkenyl, hydroxy, amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, or sulfonyl; wherein any carbon atom
- A, B, R x , R 2 , U, W, and X are as otherwise defined in formula XV.
- the non- immunosuppressive neuroimmunophilin FKBP ligand may further be a compound of formula XX
- a and B together with the nitrogen and carbon atoms to which they are respectively attached, form a 5-7 membered saturated or unsaturated heterocyclic ring which may contain, in addition to the nitrogen atom, one or more heteroatom (s) independently selected from the group consisting of 0, S, SO, S0 2 , N, NH, and NR 2 ;
- X is either 0 or S
- Y is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3 , S, SO, or S0 2 ;
- R 2 is selected from the group consisting of hydrogen, C x - C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, and C x -C 4 bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Ar is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) ; wherein the individual ring size is 5-8 members; wherein the heterocyclic ring contains 1-6 heteroatom (s) independently selected from the group consisting of 0, N, and S ; wherein an aromatic or tertiary alkyl amine is optionally oxidized to a corresponding N-oxide;
- Z is a direct bond, C- L -Cg straight or branched chain alkyl, or C 2 -C 3 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 2 , S, SO, or S0 2 ;
- C and D are independently hydrogen, Ar, C j. -Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is optionally substituted with alkyl, C 2 -C 6 alkenyl, hydroxy, amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, or sulfonyl; wherein any carbon
- R- L is selected from the group consisting of Ar, C 3 -C 8 cycloalkyl, straight or branched chain alkyl, and C 2 -C 5 straight or branched chain alkenyl, wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of Ar, C 3 -C 8 cycloalkyl, amino, halo, haloalkyl, hydroxy, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, carbonyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, and sulfonyl, wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3
- Useful carbo- and heterocyclic rings include without limitation phenyl, benzyl, naphthyl, indenyl , azulenyl, fluorenyl, anthracenyl , indolyl , isoindolyl, indolinyl, benzofuranyl, benzothiophenyl , indazolyl , benzimidazolyl , benzthiazolyl , tetrahydrofuranyl , tetrahydropyranyl , pyridyl , pyrrolyl, pyrrolidinyl, pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, tetrahydroquinolinyl , quinolizinyl , furyl , thiophenyl, imidazolyl, oxazolyl, benzoxazolyl , thiazolyl, is
- Ar is selected from the group consisting of phenyl, benzyl, naphthyl, indolyl , pyridyl, pyrrolyl, pyrrolidinyl, pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, furyl , fluorenyl, thiophenyl, imidazolyl, oxazolyl, thiazolyl, pyrazolyl, and thienyl.
- a and B together with the nitrogen and carbon atoms to which they are respectfully attached, form a 6 membered saturated or unsaturated heterocyclic ring; and R 2 is C 4 -C 7 branched chain alkyl, C 4 -C 7 cycloalkyl, phenyl, or 3 , 4 , 5-trimethoxyphenyl .
- the compound is selected from the group consisting of:
- non- immunosuppressive neuroimmunophilin FKBP ligand may be a compound of formula XXI
- E, F, G and J are independently CH 2 , 0, S, SO, S0 2 , NH or NR 2 ;
- X is either O or S ;
- Y is a direct bond, C x -C 6 straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with O, NH, NR 3 , S, SO, or S0 2 ;
- R 2 is selected from the group consisting of hydrogen, C ⁇ - C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, and C 1 -C 4 bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Z is a direct bond, straight or branched chain alkyl, or C 2 -C 5 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 2 , S, SO, or S0 2 ;
- Ar is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) ; wherein the individual ring size is 5-8 members; wherein the heterocyclic ring contains 1-6 heteroatom(s) independently selected from the group consisting of O, N, and S; wherein an aromatic or tertiary alkyl amine is optionally oxidized to a corresponding N-oxide;
- C and D are independently hydrogen, Ar, C ⁇ Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl; wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is optionally substituted with alkyl, C 2 -C 5 alkenyl, hydroxy, amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, or sulfonyl; wherein any carbon atom of said al
- R- L is selected from the group consisting of Ar, C 3 -C 8 cycloalkyl, straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of Ar, C 3 -C 8 cycloalkyl, amino, halo, haloalkyl, hydroxy, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, carbonyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, and sulfonyl, wherein any carbon atom of said alkyl or alkenyl is optionally replaced with O, NH, NR 3
- Useful carbo- and heterocyclic rings include without limitation phenyl, benzyl, naphthyl, indenyl , azulenyl, fluorenyl, anthracenyl, indolyl , isoindolyl, indolinyl, benzofuranyl , benzothiophenyl , indazolyl, benzimidazolyl , benzthiazolyl, tetrahydrofuranyl , tetrahydropyranyl, pyridyl, pyrrolyl, pyrrolidinyl, pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, tetrahydroquinolinyl , quinolizinyl , furyl , thiophenyl, imidazolyl, oxazolyl, benzoxazolyl , thiazolyl, isoxazoly
- Ar is selected from the group consisting of phenyl, benzyl, naphthyl, indolyl, pyridyl, pyrrolyl, pyrrolidinyl, pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, furyl , fluorenyl, thiophenyl, imidazolyl, oxazolyl, thiazolyl, pyrazolyl, and thienyl.
- the non- immunosuppressive neuroimmunophilin FKBP ligand may also be a compound of formula XXII
- E, F, and G are independently CH 2 , 0, S, SO, S0 2 , NH or NR 2 ;
- X is either 0 or S
- Y is a direct bond, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 2 , S, SO, or S0 2 ;
- R 2 is selected from the group consisting of hydrogen, C x - C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, and C 1 -C 4 bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Ar is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) ; wherein the individual ring size is 5-8 members; wherein the heterocyclic ring contains 1-6 heteroatom(s) independently selected from the group consisting of 0, N, and S ; wherein an aromatic or tertiary alkyl amine is optionally oxidized to a corresponding N-oxide;
- Z is a direct bond, C x -C 6 straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 2 , S, SO, or S0 2 ;
- R 2 is selected from the group consisting of hydrogen, C x - C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, and C 1 -C 4 bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- C and D are independently hydrogen, Ar, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is optionally substituted with C x -C 4 alkyl, C 2 -C 4 alkenyl, or hydroxy; wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 2 , S, SO, or S0 2 ; and
- R- L is selected from the group consisting of Ar, C 3 -C 8 cycloalkyl, straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of Ar, C 3 -C 8 cycloalkyl, amino, halo, haloalkyl, hydroxy, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, carbonyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, and sulfonyl, wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 3
- Useful carbo- and heterocyclic rings include without limitation phenyl, benzyl, naphthyl, indenyl , azulenyl, fluorenyl, anthracenyl , indolyl, isoindolyl, indolinyl, benzofuranyl , benzothiophenyl , indazolyl, benzimidazolyl , benzthiazolyl, tetrahydrofuranyl , tetrahydropyranyl, pyridyl, pyrrolyl, pyrrolidinyl, pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, tetrahydroquinolinyl , quinolizinyl , furyl , thiophenyl, imidazolyl, oxazolyl, benzoxazolyl , thiazolyl, isoxazoly
- Ar is selected from the group consisting of phenyl, benzyl, naphthyl, indolyl, pyridyl, pyrrolyl, pyrrolidinyl, pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, furyl, fluorenyl, thiophenyl, imidazolyl, oxazolyl, thiazolyl, pyrazolyl, and thienyl.
- Add i t i ona l l y , t he non - i mmuno s upp re s s i ve neuroimmunophilin FKBP ligand may be a compound of formula XXI I I
- n 1, 2 or 3;
- X is either 0 or S
- Y is a direct bond, Ci-Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with 0, NH, NR 2 , S, SO, or S0 2 ;
- R 2 is selected from the group consisting of hydrogen, C ⁇ - C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, and C x -C 4 bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Z is a direct bond, C x -C 6 straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with amino, halo, haloalkyl, thiocarbonyl, ester, thioester, alkoxy, alkenoxy, cyano, nitro, imino, alkylamino, aminoalkyl, sulfhydryl, thioalkyl, sulfonyl, or oxygen to form a carbonyl, or wherein any atom of said alkyl or alkenyl is optionally replaced with O , NH , NR 2 , S , SO , or S0 2 ;
- R 2 is selected from the group consisting of hydrogen, C x - C 4 straight or branched chain alkyl, C 3 -C 4 straight or branched chain alkenyl or alkynyl, and C ⁇ bridging alkyl wherein a bridge is formed between the nitrogen and a carbon atom of said alkyl or alkenyl chain containing said heteroatom to form a ring, wherein said ring is optionally fused to an Ar group;
- Ar is an alicyclic or aromatic, mono-, bi- or tricyclic, carbo- or heterocyclic ring, wherein the ring is either unsubstituted or substituted with one or more substituent (s) ; wherein the individual ring size is 5-8 members; wherein the heterocyclic ring contains 1-6 heteroatom(s) independently selected from the group consisting of O, N, and S; wherein an aromatic or tertiary alkyl amine is optionally oxidized to a corresponding N-oxide;
- C and D are independently hydrogen, Ar, straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein said alkyl or alkenyl is optionally substituted with one or more substituent (s) independently selected from the group consisting of C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, carbonyl oxygen, and Ar; wherein said alkyl, alkenyl, cycloalkyl or cycloalkenyl is optionally substituted with C x -C 4 alkyl, C 2 -C 4 alkenyl, or hydroxy; wherein any carbon atom of said alkyl or alkenyl is optionally substituted in one or more position (s) with oxygen to form a carbonyl, or wherein any carbon atom of said alkyl or alkenyl is optionally replaced with O, NH, NR 2 , S, SO, or S0 2 ; and R ⁇ is selected from
- Useful carbo- and heterocyclic rings include without limitation phenyl, benzyl, naphthyl, indenyl, azulenyl, fluorenyl, anthracenyl , indolyl, isoindolyl, indolinyl, benzofuranyl , benzothiophenyl , indazolyl , benzimidazolyl , benzthiazolyl , tetrahydrofuranyl , tetrahydropyranyl , pyridyl , pyrrolyl, pyrrolidinyl, pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, tetrahydroquinolinyl , quinolizinyl, furyl , thiophenyl, imidazolyl, oxazolyl, benzoxazolyl , thiazolyl, isox
- Ar is selected from the group consisting of phenyl, benzyl, naphthyl, indolyl, pyridyl, pyrrolyl, pyrrolidinyl, pyridinyl, pyrimidinyl, purinyl, quinolinyl, isoquinolinyl, furyl , fluorenyl, thiophenyl, imidazolyl, oxazolyl, thiazolyl, pyrazolyl, and thienyl.
- non-immunosuppressive neuroimmunophilin FKBP ligand may be a compound of formula XXIV
- V is C, N, or S
- A, B, C, D, R l t X, Y, and Z are as defined in formula XX above .
- the non-immunosupressive neuroimmunophilin FKBP ligand may also be a compound of formula XXV
- R 1 is C-C 9 straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl or Ar 1# wherein said R 1 is unsubstituted or substituted with one or more substituents independently selected from the group consisting of alkyl, C 2 -C 6 alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, and Ar 2 ;
- Ar- L and Ar 2 are independently selected from the group consisting of 1 -naphthyl, 2-napthyl, 2 -indolyl, 3 -indolyl, 2- furyl , 3-furyl, 2-thienyl, 3-thienyl, 2-pyridyl, 3-pyridyl, 4-pyridyl and phenyl, wherein said Ar x is unsub
- Z is C- L -Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein said Z is substituted with one or more substituent (s) independently selected from the group consisting of Ar l7 C 3 -C 8 cycloalkyl, and C- L -Cg straight or branched chain alkyl or C 2 -C 3 straight or branched chain alkenyl substituted with C 3 -C 8 cycloalkyl; or Z is fragment o
- R 3 is straight or branched chain alkyl which is unsubstituted or substituted with C 3 -C 8 cycloalkyl or Ar x ;
- X 2 is 0 or NR 5 , wherein R 5 is selected from the group consisting of hydrogen, straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl; and R 4 is selected from the group consisting of phenyl, benzyl, -Cg straight or branched chain alkyl, C 2 -C 5 straight or branched chain alkenyl, straight or branched chain alkyl substituted with phenyl, and C 2 -C 5 straight or branched chain alkenyl substituted with phenyl .
- Z and R x are lipophilic .
- the compound is selected from the group consisting of: 3 -phenyl-1-propyl (2S) -1- (3 , 3-dimethyl-l, 2 -dioxopentyl) -
- Add i t i ona l l y , t h e no n - i mmuno s upp re s s i ve neuroimmunophilin FKBP l igand may be a compound of formula XXVI
- R x is straight or branched chain alkyl, C 2 -C 9 straight or branched chain alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl or Ar 1# wherein said R is unsubstituted or substituted with one or more substituents independently selected from the group consisting of alkyl, C 2 -C 6 alkenyl, C 3 -C 8 cycloalkyl, C 5 -C 7 cycloalkenyl, hydroxy, and Ar 2 ;
- Ar ! and Ar 2 are independently selected from the group consisting of 1 -naphthyl, 2-napthyl, 2 -indolyl, 3 -indolyl, 2- furyl, 3-furyl, 2-thienyl, 3-thienyl, 2-pyridyl, 3-pyridyl, 4-pyridyl and phenyl, wherein said Ar x is unsubstituted or substituted with one or more substituent (s) independently selected from the group consisting of hydrogen, halo, hydroxy, nitro, trifluoromethyl, straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 1 -C 4 alkoxy, C 2 -C 4 alkenyloxy, phenoxy, benzyloxy, and amino;
- Z is C- L -Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein said Z is substituted with one or more substituent (s) independently selected from the group consisting of Ar x , C 3 -C 8 cycloalkyl, and C-,-C e straight or branched chain alkyl or C 2 -C 6 straight or branched chain alkenyl substituted with C 3 -C 8 cycloalkyl; or Z is fragment o
- R 3 is C- L -Cg straight or branched chain alkyl which is unsubstituted or substituted with C 3 -C 8 cycloalkyl or Ar 17 -
- X 2 is 0 or NR 5 , wherein R s is selected from the group consisting of hydrogen, C x -Cg straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl; and
- R 4 is selected from the group consisting of phenyl, benzyl, C 1 -C 5 straight or branched chain alkyl, C 2 -C 5 straight or branched chain alkenyl, C ⁇ C s straight or branched chain alkyl substituted with phenyl, and C 2 -C 5 straight or branched chain alkenyl substituted with phenyl.
- R x is selected from the group consisting of straight or branched chain alkyl, 2 -cyclohexyl , 4 -cyclohexyl , 2 -furanyl, 2-thienyl, 2 -thiazolyl, and 4-hydroxybutyl .
- Z and R x are lipophilic.
- the non-immunosuppressive neuroimmunophilin FKBP ligand may also be a compound of formula XXVII
- R 3 is Ci-Cg straight or branched chain alkyl or unsubstituted Ar l; wherein said alkyl is unsubstituted or substituted with C 3 -C 8 cycloalkyl or Ar 17 -
- X 2 is 0 or NR 5 , wherein R 5 is selected from the group consisting of hydrogen, straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl; R 4 is selected from the group consisting of phenyl, benzyl, C 1 -C 5 straight or branched chain alkyl, C 2 -C 5 straight or branched chain alkenyl, straight or branched chain alkyl substituted with phenyl, and C 2 -C 5 straight or branched chain alkenyl substituted with phenyl; and Ar- L is as defined in formula XXVI.
- Z' is lipophilic .
- the non- immunosuppressive neuroimmunophilin FKBP ligand may also be a compound of formula XXVIII
- R ⁇ is straight or branched chain alkyl, C 2 -C 6 straight or branched chain alkenyl, C 3 -C 3 cycloalkyl or Ar l; wherein said alkyl or alkenyl is unsubstituted or substituted with C 3 -C 6 cycloalkyl or Ar 2 ;
- Ar- L and Ar 2 are independently selected from the group consisting of 2-furyl, 2-thienyl, and phenyl; X is 0, S, CH 2 or H 2 ; Y is oxygen;
- Z is straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein said Z is substituted with one or more substituent (s) independently selected from the group consisting of 2-furyl, 2-thienyl, C 3 - C 6 cycloalkyl, pyridyl, and phenyl, each having one or more substituent (s) independently selected from the group consisting of hydrogen and C x -C 4 alkoxy.
- Z and R are lipophilic.
- the compound is selected from the group consisting of:
- the compound is selected from the group consisting of: 3- (3-pyridyl) -1-propyl (2S) - 1- (3 , 3 -dimethyl-1 , 2 - dioxopentyl ) -2 -pyrrolidinecarboxylate ;
- the compound is 3- (3-pyridyl) -1-propyl (2S) -1- (3 , 3-dimethyl-l , 2- dioxopentyl) -2-pyrrolidinecarboxylate, and pharmaceutically acceptable salts, esters, and solvates thereof.
- FORMULA XXIX Add i t i ona l l y , t he non - i mmuno supp re s s ive neuroimmunophilin FKBP l igand may be a compound of formula XXIX
- Y is a direct bond, 0, or NR 2 , wherein R 2 is hydrogen or Ci-Cg alkyl;
- Z is C x -Cg straight or branched chain alkyl, or C 2 -C 6 straight or branched chain alkenyl, wherein said Z is substituted with one or more substituent (s) independently selected from the group consisting of Ar 1 C 3 -C 8 cycloalkyl, and straight or branched chain alkyl or C 2 -C 5 straight or branched chain alkenyl substituted with C 3 -C 8 cycloalkyl; or Z is fragment o
- R 3 is C x -Cg straight or branched chain alkyl which is unsubstituted or substituted with C 3 -C 8 cycloalkyl or Ar 1 -
- X 2 is O or NR 5 , wherein R 5 is selected from the group consisting of hydrogen, C x -Cg straight or branched chain alkyl, and C 2 -C 6 straight or branched chain alkenyl; and R 4 is selected from the group consisting of phenyl, benzyl, C 1 -C 5 straight or branched chain alkyl, C 2 -C 5 straight or branched chain alkenyl, C 1 -C 5 straight or branched chain alkyl substituted with phenyl, and C 2 -C 5 straight or branched chain alkenyl substituted with phenyl.
- All the compounds of Formulas I -XXIX possess asymmetric centers and thus can be produced as mixtures of stereoisomers or as individual R- and S- stereoisomers.
- the individual stereoisomers may be obtained by using an optically active starting material, by resolving a racemic or non-racemic mixture of an intermediate at some appropriate stage of the synthesis, or by resolving the compounds of Formulas I -XXIX.
- stereoisomers (racemic and non-racemic) of stereoisomers.
- S- stereoisomers are used in the pharmaceutical compositions and methods of the present invention.
- Isocyanates (R'NCO) or isothiocyanates (R'NCS) 4 may be conveniently prepared from the corresponding readily available amines by reaction with phosgene or thiophosgene, as depicted in Scheme II.
- R-SH may be conveniently prepared from the corresponding readily available alcohols or halides via a two step replacement of halide by sulfur, as described in Scheme III. Halides may be reacted with thiourea, and the corresponding alkyl thiouronium salts hydrolyzed to provide thiols RSH. If alcohols are used as the starting materials, they may be first converted to the corresponding halides by standard methods.
- the compounds of formulas XX to XXIV may be readily prepared by standard techniques of organic chemistry, utilizing the general synthetic pathway depicted below. As described by Scheme IV, cyclic amino acids 1 protected by suitable blocking groups P on the amino acid nitrogen may be reacted with thiols RSH to generate thioesters 2. After removal of the protecting group, the free amine 3 may be reacted with various sulfonyl chlorides 4 to provide final products 5 in good to excellent yield.
- Thiols R-SH may be conveniently prepared from the corresponding readily available alcohols or halides via a two step replacement of halogen by sulfur, as described in Scheme V. Halides may be reacted with thiourea, and the corresponding alkyl thiouronium salts hydrolyzed to provide thiols RSH. If alcohols are used as the starting materials, they may be first converted to the corresponding halides by standard methods .
- the compounds of formulas XXV to XXIX may be prepared by a variety of synthetic sequences that utilize established chemical transformations.
- the general pathway to the present compounds is described in Scheme VI.
- N-glyoxylproline derivatives may be prepared by reacting L-proline methyl ester with methyl oxalyl chloride as shown in Scheme VI .
- the resulting oxamates may be reacted with a variety of carbon nucleophiles to obtain intermediates compounds. These intermediates are then reacted with a variety of alcohols, amides, or protected amino acid residues to obtain the propyl esters and amides of the invention.
- alkyl or aryl aldehydes may be homologated to phenyl propanols by reaction with methyl (triphenyl-phosphoranylidene) acetate to provide a variety of trans-cinnamates; these latter may be reduced to the saturated alcohols by reaction with excess lithium aluminum hydride, or sequentially by reduction of the double bond by catalytic hydrogenation and reduction of the saturated ester by appropriate reducing agents.
- the trans-cinnamates may be reduced to (E) - allylic alcohols by the use of diisobutylaluminum hydride.
- Ph 3 P CHCOOCH 3 hydride R-CHO » ⁇ / ⁇ COOCH 3
- Longer chain alcohols may be prepared by homologation of benzylic and higher aldehydes.
- these aldehydes may be prepared by conversion of the corresponding phenylacetic and higher acids, and phenethyl and higher alcohols .
- the compounds used in the inventive methods and pharmaceutical compositions have an affinity for the FK506 binding protein, particularly FKBP12.
- the inhibition of the prolyl peptidyl cis - trans isomerase activity of FKBP may be measured as an indicator of this affinity.
- the cis - trans isomerization of an alanine-proline bond in a model substrate, ⁇ -succinyl-Ala-Ala-Pro-Phe-p- nitroanilide is monitored spectrophotometrically in a chymotrypsin-coupled assay, which releases para-nitroanilide from the trans form of the substrate.
- the inhibition of this reaction caused by the addition of different concentrations of inhibitor is determined, and the data is analyzed as a change in first -order rate constant as a function of inhibitor concentration to yield the apparent K. values.
- a plastic cuvette In a plastic cuvette are added 950 mL of ice cold assay buffer (25 mM HEPES, pH 7.8, 100 mM NaCl) , 10 mL of FKBP (2.5 mM in 10 mM Tris-Cl pH 7.5, 100 mM NaCl, 1 mM dithiothreitol) , 25 mL of chymotrypsin (50 mg/ml in 1 mM HCl) and 10 mL of test compound at various concentrations in dimethyl sulfoxide.
- ice cold assay buffer 25 mM HEPES, pH 7.8, 100 mM NaCl
- FKBP 2.5 mM in 10 mM Tris-Cl pH 7.5, 100 mM NaCl, 1 mM dithiothreitol
- 25 mL of chymotrypsin 50 mg/ml in 1 mM HCl
- 10 mL of test compound at various concentrations
- the reaction is initiated by the addition of 5 mL of substrate (succinyl-Ala-Phe-Pro-Phe-para- nitroanilide, 5 mg/mL in 2.35 mM LiCl in trifluoroethanol) .
- substrate succinyl-Ala-Phe-Pro-Phe-para- nitroanilide, 5 mg/mL in 2.35 mM LiCl in trifluoroethanol
- the absorbance at 390 nm versus time is monitored for 90 seconds using a spectrophotometer and the rate constants are determined from the absorbance versus time data files.
- Relative potencies of compounds are ranked according to the following scale: ++++ denotes K. or ED50 ⁇ 1 nM; +++ denotes K, or ED50 of 1-50 nM; ++ denotes K, or ED 50 of 51- 200 nM; + denotes K 1 or ED of 201-500 nM.
- Relative potencies of compounds are ranked according to the following scale: ++++ denotes K x or ED50 ⁇ 1 nM; +++ denotes K ⁇ or ED50 of 1-50 nM; ++ denotes K x or ED 50 of 51- 200 nM; + denotes K ⁇ or ED of 201-500 nM.
- the compounds used in the inventive methods and pharmaceutical compositions must readily affect the targeted areas .
- Other routes of administration known in the pharmaceutical art are also contemplated by this invention.
- Dosage levels on the order of about 0.1 mg to about 10,000 mg of the active ingredient compound are useful in the treatment of the above conditions, with preferred levels of about 0.1 mg to about 1,000 mg .
- the specific dose level for any particular patient will vary depending upon a variety of factors, including the activity of the specific compound employed; the age, body weight, general health, sex and diet of the patient; the time of administration; the rate of excretion; drug combination; the severity of the particular disease being treated; and the form of administration.
- vi tro dosage-effect results provide useful guidance on the proper doses for patient administration. Studies in animal models are also helpful . The considerations for determining the proper dose levels are well known in the art.
- the compounds can be administered with other agents for treating vision loss, preventing vision degeneration, or promoting vision regeneration. Specific dose levels for such other agents will depend upon the factors previously stated and the effectiveness of the drug combination.
- Example 5 1, 2 -dioxopentyl) -2- (4-thiazolidine) carboxylate (81)
- the compound of Example 5 was prepared according to the procedure of Example 4, using 3- (3-pyridyl) -1-propanol in the final step, to yield 3 - (3 -pyridyl) -1-propyl (2S) -1- (3 , 3- dimethyl-1 , 2 -dioxopentyl) -2- (4-thiazolidine) carboxylate .
- the concentrated filtrate was purified on a flash column
- 1,5 -Diphenyl -3 -pentylmercaptyl N- ( p a ra - toluenesulfonyl) pipecolate (18) was prepared from 1,5- diphenyl -3 -pentylmercaptyl pyrrolidine-2 -carboxylate and para-toluenesulfonyl chloride as described for Example 12, in 65% yield.
- Methyl (25) -1- ( 1 , 2 -dioxo- 3 , 3-dimethylpentyl ) -2 - pyrrolidinecarboxylate A solution of methyl (25) -1- (1 , 2 -dioxo-2 -methoxyethyl) - 2-pyrrolidinecarboxylate (2.35 g; 10.90 mmol) in 30 mL of tetrahydrofuran (THF) was cooled to -78°C and treated with 14.2 mL of a 1.0 M solution of 1 , 1-dimethylpropylmagnesium chloride in THF.
- THF tetrahydrofuran
- Example 15 The method of Example 14 was utilized to prepare the following illustrative compounds.
- Compound 140 3- (3 , 4 , 5-trimethoxyphenyl) -l-prop-2- (E) -enyl ( 2 S) - 1 - (3,3 -dimethyl -1,2 -dioxopentyl ) -2 -pyrrolidine carboxylate, 66%.
- Compound 150 3- (2 , 5-dimethoxyphenyl) -1-propyl (25)-l-(3,3- dimethyl-1, 2-dioxopentyl) -2 -pyrrolidine- carboxylate, 99%.
- GPI 1046 protects retinal ganglion cells against degeneration following retinal ischemia.
- Retinal ganglion cells were retrogradely labeled in adult rats by bilateral injection of fluorogold in their lateral geniculate nuclei. Labeled ganglion cells in the normal rat retina appear as white profiles against the dark background ( Figure 1A) .
- Complete retinal ischemia was produced by infusing normal saline solution into the retinal vitreous cavity of each eye until the intraocular pressure exceeded arterial blood pressure. 28 days after the ischemic episode extensive degeneration of retinal ganglion cell was evidenced by massive reduction in the density of fluorogold labeled cells (Figure IB) .
- GPI 1046 prevents degeneration of optic nerve axons and myielin following retinal ischemia
- FIG. 3 GPI 1046 provides moderate protection against retinal ganglion cell death after optic nerve transection.
- Complete transection of the optic nerve 5 mm from the eyeball produces massive degeneration of retinal ganglion cells, representing loss of >87% of the normal ganglion cell population 90 days after the injury (Table 1) .
- Few spared fluorogold pre labeled ganglion cells are present in vehicle treated cases (large white figures) among a population of small microglia that digest the debris of the degenerating cells and take up the fluorogold label ( Figure 3A) .
- GPI 1046 treatment duration significantly affects the process of optic nerve axonal degeneration after transection.
- 90 days after transection extensive degeneration of myelin is evident in vehicle treated cases, characterized by the loss of fascicular organization and the appearance of numerous large dense degenerating myelin figures ( Figure 6B, upper right) .
- Treatment with GPI 1046 for the first 14 days after optic nerve transection did not alter the pattern of myelin degeneration (Figure 6C, lower left panel) , and yielded an insignificant 1.6% quantitative recovery in myelin density (Table 1) .
- FIG. 7 FKBP-12 immunohistochemistry labels oligodendroglia (large dark cells with fibrous processes) , the cells which produce myelin, located between the fascicles of optic nerve fibers, and also some optic nerve axons.
- Figure 9 28 day treatment with GPI 1046 treatment beginning 8 weeks after onset of streptozotocin induced diabetes decreases the extent of neovascularization in the inner and outer retina and protects neurons in the inner nuclear layer (INL) and ganglion cell layer (GCL) from degeneration. Negative images of cresyl violet stained tangential retinal sections reveals perikarya in the three cellular layers ( Figure 9A) .
- the retinae of streptozotocin treated animals administered only vehicle (Figure 9B) exhibited loss of cells from the ONL and INL, decreased thickness of the Outer plexiform layer (the dark area between ONL and INL) and a dramatic increase in the size and density of retinal blood vessels (large black circular outlines) in the INL, OPL, ONL and the photoreceptor layer (PR, the gray fuzzy area above the ONL) .
- GPI 1046 treatment reduced neovascularization (i.e. prevented the proliferation of blood vessels) in the PR, ONL, OPL and INL. Although GPI 1046 did not appear to protect against neuronal loss in the ONL, it appeared to decrease the loss of neurons in both the INL and GCL compared to streptozotocin/vehicle treated controls.
- Example 20 Jn Vivo Retinal Ganglion Cell and Optic Nerve Axon Tests The extent of degeneration reduction or prevention in retinal ganglion cells and optic nerve axons was determined in a vision loss model utilizing surgical optic nerve transection to simulate mechanical damage to the optic nerve.
- mice were divided into six experimental groups of six rats (12 eyes) per group.
- One group received a neuroimmunophilin FKBP ligand (10 milligrams per kg per day sc in PEG vehicle (20 percent propylene glycol, 20 percent ethanol, and 60 percent saline)) for 14 days.
- a second group received the same neuroimmunophilin FKBP ligand dose for 28 days.
- Each treated group had a corresponding sham/surgery and transection control group which received corresponding 14 or 28 day dosing with the vehicle only.
- Retinas were removed from eyes and prepared for wholemount analysis. For each group, five eyes with dense and intense FG labeling were selected for quantitative analysis using a 20 power objective. Digital images were obtained from five fields in the central retina (3-4 millimeters radial to optic nerve head) . FG labeled Large (>18 ⁇ m) , medium (12-16 ⁇ m) , and small ( ⁇ 10 ⁇ m) ganglion cells and microglia were counted in five 400 ⁇ m by 400 ⁇ m fields per case, 5 cases per group. Examination of Optic Nerves
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Ophthalmology & Optometry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Hospice & Palliative Care (AREA)
- Psychiatry (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002335815A CA2335815A1 (en) | 1998-08-14 | 1999-08-12 | Compositions and uses for vision and memory disorders |
| AU55556/99A AU5555699A (en) | 1998-08-14 | 1999-08-12 | Compositions and uses for vision and memory disorders |
| JP2000564611A JP4503180B2 (en) | 1998-08-14 | 1999-08-12 | Compositions and uses for vision and memory impairment |
| EP99942108A EP1105112A2 (en) | 1998-08-14 | 1999-08-12 | Compositions and uses for vision and memory disorders |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13442298A | 1998-08-14 | 1998-08-14 | |
| US09/134,422 | 1998-08-14 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2000009108A2 true WO2000009108A2 (en) | 2000-02-24 |
| WO2000009108A3 WO2000009108A3 (en) | 2000-05-18 |
Family
ID=22463332
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1999/018241 WO2000009108A2 (en) | 1998-08-14 | 1999-08-12 | Compositions and uses for vision and memory disorders |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP1105112A2 (en) |
| JP (1) | JP4503180B2 (en) |
| AU (1) | AU5555699A (en) |
| CA (1) | CA2335815A1 (en) |
| WO (1) | WO2000009108A2 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001008685A1 (en) * | 1999-07-30 | 2001-02-08 | Vertex Pharmaceuticals Incorporated | Acyclic and cyclic amine derivatives |
| WO2001035948A2 (en) * | 1999-11-12 | 2001-05-25 | Alcon Universal Ltd. | Neurophilin ligands for treating ocular conditions |
| WO2003048150A1 (en) * | 2001-12-06 | 2003-06-12 | Institute Of Pharmacology And Toxicology Academy Of Military Medical Sciences P.L.A. | Substituted 5-membered n-heterocyclic compounds and their uses for treating or preventing neurodegenerative diseases |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6218424B1 (en) * | 1996-09-25 | 2001-04-17 | Gpi Nil Holdings, Inc. | Heterocyclic ketone and thioester compounds and uses |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1992000278A1 (en) * | 1990-07-02 | 1992-01-09 | Vertex Pharmaceuticals Incorporated | Novel immunosuppressive compounds |
| WO1992019593A1 (en) * | 1991-05-09 | 1992-11-12 | Vertex Pharmaceuticals Incorporated | Novel immunosuppressive compounds |
| WO1996040633A1 (en) * | 1995-06-07 | 1996-12-19 | Guilford Pharmaceuticals Inc. | Small molecule inhibitors of rotamase enzyme activity |
| WO1996040140A1 (en) * | 1995-06-07 | 1996-12-19 | Guilford Pharmaceuticals Inc. | Inhibitors of rotamase enzyme activity |
| WO1996041609A2 (en) * | 1995-06-08 | 1996-12-27 | Vertex Pharmaceuticals Incorporated | Methods and compositions for stimulating neurite growth |
| WO1997016190A1 (en) * | 1995-10-31 | 1997-05-09 | Guilford Pharmaceuticals Inc. | Rotamase enzyme activity inhibitors |
| WO1997031020A1 (en) * | 1996-02-22 | 1997-08-28 | The General Hospital Corporation | METHODS AND COMPOSITIONS FOR ENHANCING CELLULAR RESPONSE TO TGF-β LIGANDS |
| WO1998013343A1 (en) * | 1996-09-25 | 1998-04-02 | Guilford Pharmaceuticals Inc. | Heterocyclic thioesters and ketones |
| WO1998013355A1 (en) * | 1996-09-25 | 1998-04-02 | Guilford Pharmaceuticals Inc. | Heterocyclic esters and amides |
| WO1998037885A1 (en) * | 1997-02-28 | 1998-09-03 | Guilford Pharmaceuticals Inc. | N-oxides of heterocyclic esters, amides, thioesters, and ketones |
| WO1998048784A2 (en) * | 1997-04-28 | 1998-11-05 | The University Of British Columbia | Method and composition for modulating amyloidosis |
| EP0947506A1 (en) * | 1998-03-24 | 1999-10-06 | Pfizer Products Inc. | 2-Aza-bicyclo[2.2.1]heptane derivatives and their use as rotamase inhibitors |
-
1999
- 1999-08-12 EP EP99942108A patent/EP1105112A2/en not_active Withdrawn
- 1999-08-12 JP JP2000564611A patent/JP4503180B2/en not_active Expired - Fee Related
- 1999-08-12 WO PCT/US1999/018241 patent/WO2000009108A2/en not_active Application Discontinuation
- 1999-08-12 AU AU55556/99A patent/AU5555699A/en not_active Abandoned
- 1999-08-12 CA CA002335815A patent/CA2335815A1/en not_active Abandoned
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1992000278A1 (en) * | 1990-07-02 | 1992-01-09 | Vertex Pharmaceuticals Incorporated | Novel immunosuppressive compounds |
| WO1992019593A1 (en) * | 1991-05-09 | 1992-11-12 | Vertex Pharmaceuticals Incorporated | Novel immunosuppressive compounds |
| WO1996040633A1 (en) * | 1995-06-07 | 1996-12-19 | Guilford Pharmaceuticals Inc. | Small molecule inhibitors of rotamase enzyme activity |
| WO1996040140A1 (en) * | 1995-06-07 | 1996-12-19 | Guilford Pharmaceuticals Inc. | Inhibitors of rotamase enzyme activity |
| WO1996041609A2 (en) * | 1995-06-08 | 1996-12-27 | Vertex Pharmaceuticals Incorporated | Methods and compositions for stimulating neurite growth |
| WO1997016190A1 (en) * | 1995-10-31 | 1997-05-09 | Guilford Pharmaceuticals Inc. | Rotamase enzyme activity inhibitors |
| WO1997031020A1 (en) * | 1996-02-22 | 1997-08-28 | The General Hospital Corporation | METHODS AND COMPOSITIONS FOR ENHANCING CELLULAR RESPONSE TO TGF-β LIGANDS |
| WO1998013343A1 (en) * | 1996-09-25 | 1998-04-02 | Guilford Pharmaceuticals Inc. | Heterocyclic thioesters and ketones |
| WO1998013355A1 (en) * | 1996-09-25 | 1998-04-02 | Guilford Pharmaceuticals Inc. | Heterocyclic esters and amides |
| WO1998037885A1 (en) * | 1997-02-28 | 1998-09-03 | Guilford Pharmaceuticals Inc. | N-oxides of heterocyclic esters, amides, thioesters, and ketones |
| WO1998048784A2 (en) * | 1997-04-28 | 1998-11-05 | The University Of British Columbia | Method and composition for modulating amyloidosis |
| EP0947506A1 (en) * | 1998-03-24 | 1999-10-06 | Pfizer Products Inc. | 2-Aza-bicyclo[2.2.1]heptane derivatives and their use as rotamase inhibitors |
Non-Patent Citations (5)
| Title |
|---|
| H. GUO ET AL.: "The novel small molecule immunophilin ligand GPI 1046 stimulates cholinergic reinnervation of dearrerented hippocampal regions after fimbria-fornix transection" SOCIETY FOR NEUROSCIENCE ABSTRACTS, vol. 23, no. 1-2, 1997, page 1729 XP000878535 * |
| J.P. STEINER ET AL.: "Neurotrophic immunophilin ligands stimulate structural and functional recovery in neurodegenerative animal models" PROC. NATL. ACAD. SCI., vol. 94, no. 5, 1997, pages 2019-2024, XP000876505 * |
| PESSAH, I. N. (1): "Role for the major T-cell immunophilin FKBP12 in mediating neurotoxicity of non-coplanar PCBs." NEUROTOXICOLOGY AND TERATOLOGY, (MAY-JUNE, 1998) VOL. 20, NO. 3, PP. 350. MEETING INFO.: TWENTY-SECOND ANNUAL MEETING OF THE NEUROBEHAVIORAL TERATOLOGY SOCIETY SAN DIEGO, CALIFORNIA, USA JUNE 20-24, 1998 NEUROBEHAVIORAL TERATOLOGY SOCIETY. , XP000878599 * |
| ROSS, D. T. ET AL: "The FKBP neuroimmunophilin ligand GPI - 1046 enhances retinal ganglion cell survival and arrests axonal dying back following optic nerve transection." SOCIETY FOR NEUROSCIENCE ABSTRACTS, (1998) VOL. 24, NO. 1-2, PP. 814. MEETING INFO.: 28TH ANNUAL MEETING OF THE SOCIETY FOR NEUROSCIENCE, PART 1 LOS ANGELES, CALIFORNIA, USA NOVEMBER 7-12, 1998 SOCIETY FOR NEUROSCIENCE. , XP000878539 * |
| SAUER, H. ET AL: "Effects of chronic GPI - 1046 administration on spatial learning and memory in aged rodents." SOCIETY FOR NEUROSCIENCE ABSTRACTS, (1998) VOL. 24, NO. 1-2, PP. 686. MEETING INFO.: 28TH ANNUAL MEETING OF THE SOCIETY FOR NEUROSCIENCE, PART 1 LOS ANGELES, CALIFORNIA, USA NOVEMBER 7-12, 1998 SOCIETY FOR NEUROSCIENCE. , XP000878538 * |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001008685A1 (en) * | 1999-07-30 | 2001-02-08 | Vertex Pharmaceuticals Incorporated | Acyclic and cyclic amine derivatives |
| WO2001035948A2 (en) * | 1999-11-12 | 2001-05-25 | Alcon Universal Ltd. | Neurophilin ligands for treating ocular conditions |
| WO2001035948A3 (en) * | 1999-11-12 | 2002-01-17 | Alcon Universal Ltd | Neurophilin ligands for treating ocular conditions |
| JP2003514015A (en) * | 1999-11-12 | 2003-04-15 | アルコン,インコーポレイテッド | Neurophilin ligand for treating ocular conditions |
| WO2003048150A1 (en) * | 2001-12-06 | 2003-06-12 | Institute Of Pharmacology And Toxicology Academy Of Military Medical Sciences P.L.A. | Substituted 5-membered n-heterocyclic compounds and their uses for treating or preventing neurodegenerative diseases |
| US7262183B2 (en) | 2001-12-06 | 2007-08-28 | Instititute of Pharmacology and Toxicology | Substituted 5-membered n-heterocyclic compounds and their uses for treating or preventing neurodegenerative diseases |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2002522484A (en) | 2002-07-23 |
| CA2335815A1 (en) | 2000-02-24 |
| EP1105112A2 (en) | 2001-06-13 |
| WO2000009108A3 (en) | 2000-05-18 |
| AU5555699A (en) | 2000-03-06 |
| JP4503180B2 (en) | 2010-07-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7410995B1 (en) | N-linked sulfonamide of heterocyclic thioesters for vision and memory disorders | |
| CA2344520A1 (en) | Pipecolic acid derivatives for vision and memory disorders | |
| US6218423B1 (en) | Pyrrolidine derivatives for vision and memory disorders | |
| WO2000009112A2 (en) | Heterocyclic esters or amides for vision and memory disorders | |
| US6384056B1 (en) | Heterocyclic thioesters or ketones for vision and memory disorders | |
| US6335348B1 (en) | Nitrogen-containing linear and azepinyl/ compositions and uses for vision and memory disorders | |
| EP1105128A2 (en) | Carboxylic acids and isosteres of n-heterocyclic compounds for vision and memory disorders | |
| US6506788B1 (en) | N-linked urea or carbamate of heterocyclic thioesters for vision and memory disorders | |
| US6395758B1 (en) | Small molecule carbamates or ureas for vision and memory disorders | |
| WO2000009108A2 (en) | Compositions and uses for vision and memory disorders | |
| US6333340B1 (en) | Small molecule sulfonamides for vision and memory disorders | |
| US6337340B1 (en) | Carboxylic acids and isosteres of heterocyclic ring compounds having multiple heteroatoms for vision and memory disorders |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AL AM AT AU AZ BA BB BG BR BY CA CH CN CR CU CZ DE DK DM EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW SD SL SZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AL AM AT AU AZ BA BB BG BR BY CA CH CN CR CU CZ DE DK DM EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW SD SL SZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| ENP | Entry into the national phase in: |
Ref document number: 2335815 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1999942108 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 55556/99 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2001/000196 Country of ref document: MX |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: 1999942108 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1999942108 Country of ref document: EP |