WO1998047473A1 - Use of unsaturated aldehydes in dying fibers containing keratin - Google Patents
Use of unsaturated aldehydes in dying fibers containing keratin Download PDFInfo
- Publication number
- WO1998047473A1 WO1998047473A1 PCT/EP1998/002243 EP9802243W WO9847473A1 WO 1998047473 A1 WO1998047473 A1 WO 1998047473A1 EP 9802243 W EP9802243 W EP 9802243W WO 9847473 A1 WO9847473 A1 WO 9847473A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino
- acid
- methyl
- hydroxy
- group
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/41—Amines
- A61K8/411—Aromatic amines, i.e. where the amino group is directly linked to the aromatic nucleus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/41—Amines
- A61K8/415—Aminophenols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4906—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom
- A61K8/4913—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom having five membered rings, e.g. pyrrolidone carboxylic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4906—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom
- A61K8/4926—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom having six membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/494—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with more than one nitrogen as the only hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/494—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with more than one nitrogen as the only hetero atom
- A61K8/4953—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with more than one nitrogen as the only hetero atom containing pyrimidine ring derivatives, e.g. minoxidil
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4973—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with oxygen as the only hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/06—Preparations for styling the hair, e.g. by temporary shaping or colouring
- A61Q5/065—Preparations for temporary colouring the hair, e.g. direct dyes
Definitions
- the invention relates to the use of unsaturated aldehydes for dyeing keratin fibers, in particular human hair.
- keratin fibers e.g. B. hair, wool or fur
- direct dyes or oxidation dyes which are formed by oxidative coupling of one or more developer components with one another or with one or more coupler components
- Coupler and developer components are also referred to as oxidation dye precursors.
- Primary aromatic amines with a further free or substituted hydroxy or amino group in the para or ortho position, diaminopyridine derivatives, heterocyclic hydrazones, 4-aminopyrazolone derivatives and 2,4,5,6-tetraaminopyrimidine and their derivatives are usually used as developer components.
- M-Phenylenediamine derivatives, naphthols, resorcinol and resorcinol derivatives, pyrazolones and m-aminophenols are generally used as coupler components.
- Particularly suitable coupler substances are 1-naphthol, pyrogallol, 1, 5-, 2,7- and 1,7-dihydroxynaphthalene, o-aminophenol, 5-amino-2-methylphenol, m-aminophenol, resorcinol, resorcinol monomethyl ether, m-phenylenediamine , 1-phenyl-3-methyl-pyrazolone-5, 2,4-dichloro-3-aminophenol, 1,3-bis (2,4-diaminophenoxy) propane, 4-chlororesorcinol, 2-chloro-6-methyl -3-aminophenol, 2-methylresorcinol, 5-methylresorcinol, 2,5-dimethylresorcinol, 2,6-
- oxidation dyes can be achieved with oxidation dyes, but the development of the color generally takes place under the influence of oxidizing agents such as H 2 O 2 , which in some cases can damage the fibers. Furthermore, some oxidation dye precursors or certain mixtures of oxidation dye precursors can sometimes have a sensitizing effect on people with sensitive skin. Direct dyes are applied under gentler conditions, but their disadvantage is that the dyeings often have inadequate fastness properties.
- German patent application DE 28 30 497 discloses hair colorants which contain salicylaldehyde in combination with oxidation dye precursors, for example 2,5-diaminotoluene, 4-aminophenol or 2,4-diaminotoluene, but which (in combination with the additionally contained hydrogen peroxide) more or less can have great potential for sensitization.
- the German patent application DE 29 32 489 also discloses H 2 O 2 -containing hair colorants which contain benzaldehydes, for example 2-hydroxy-3-methoxybenzaldehyde or 4-hydroxy-3-methoxybenzaldehyde, in combination with oxidation dye precursors. US Pat. Nos.
- 5,034,014 and 5,199,954 describe examples of hair dye formulation which use p-dime contain thylaminobenzaldehyde or p-dimethylamino cinnamaldehyde, for example in combination with the sensitizing p-phenylenediamine.
- the US Pat. No. 4,391,603 has the object of oxidizing agent-free hair colorants which contain substituted benzaldehydes.
- the hair dyes disclosed there represent direct hair dyes with which the color nuances, depths of color and color fastness of hair dyes containing oxidation hair dye precursors cannot be achieved.
- the object of the present invention is to provide colorants for keratin fibers, in particular human hair, which are at least equivalent in quality to the usual oxidation hair colorants in terms of color depth, gray coverage and fastness properties, but without necessarily being dependent on oxidizing agents such as H 2 O 2 .
- the colorants may have no or only a very low sensitization potential.
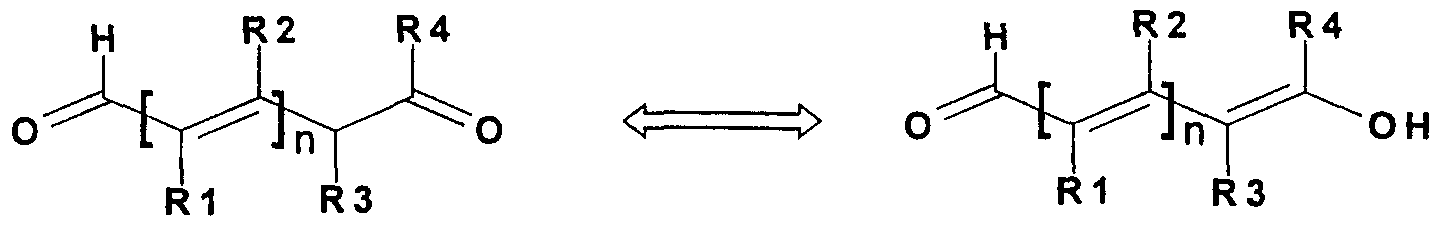
- the invention relates to the use of unsaturated aldehydes described by the tautomeric limit formulas Ia and Ib,
- R 2 , R 3 and R 4 independently of one another represent hydrogen, halogen, a CC 4 alkoxy, CrC alkyl, aryl or CC ⁇ alkoxy-C C ⁇ alkyl radical and n stand for the numbers 1 or 2 , where R 1 and R 2 , R 1 and R 3 , R 2 and R 3 and R 2 and R 4 can each form a 5- to 7-membered ring together with the rest of the molecule if n is 1, and the corresponding Mono-, bis- or ⁇ -alkoxyacetals for dyeing keratin fibers, especially human hair.
- Keratin-containing fibers are wool, furs, feathers and in particular human hair.
- the colorants according to the invention can also be used to dye other natural fibers, such as Cotton, jute, sisal, linen or silk, modified natural fibers, e.g. Regenerated cellulose, nitro, alkyl or hydroxyalkyl or acetyl cellulose and synthetic fibers such as e.g. Polyamide, polyacrylonitrile, polyurethane and polyester fibers can be used.
- the unsaturated aldehydes of the formulas Ia and Ib are preferably selected from the group consisting of glutaconaldehyde, 1-formyl-3-hydroxymethylene-1-cyclohexene, 1-formyl-3-hydroxymethylene-1-cycloheptene and 7-hydroxy-2,4,6 -heptatrienal and its substituted derivatives and the physiologically tolerable salts thereof, in particular the alkali and ammonium salts, the tetrabutylammonium salts and Na salts and mixtures thereof being particularly preferred.
- the aldehydes of the formulas Ia and Ib are contained in the colorants preferably in an amount of 0.03 to 65, in particular 1 to 40 mmol, in each case based on 100 g of the total colorant.
- a single compound with the formula Ia or Ib or a mixture of several compounds can be used. They can be used as direct colorants or in the presence of compounds which enhance the coloring effect of the aldehydes used according to the invention, such as oxidation dye precursors.
- Coloring agents which contain aldehydes of the formulas Ia and Ib alone as the coloring component are preferably used for coloring in the yellow and red range.
- Another object of the present invention is an agent for dyeing keratin fibers, in particular human hair, which A at least one unsaturated aldehyde with the formula la or Ib and B at least one compound with primary or secondary amino group or hydroxy group selected from primary or contains secondary aliphatic or aromatic amines, nitrogen-containing heterocyclic compounds, amino acids, oligopeptides and aromatic hydroxy compounds composed of 2 to 9 amino acids, and / or at least one CH-active compound.
- the above-mentioned compounds of component B can be used in an amount of 0.03 to 65, in particular 1 to 40 mmol, in each case based on 100 g of the total colorant.
- Suitable compounds with primary or secondary amino groups are e.g. primary aromatic amines such as N, N-dimethyl, N, N-diethyl, N- (2-hydroxyethyl) -N-ethyl, N, N-bis (2-hydroxyethyl) -, N- (2-methoxyethyl -), 2,3-, 2,4-, 2,5-dichloro-p-phenylenediamine, 2-chloro-p-phenylenediamine, 2,5-dihydroxy-4-morpholinoaniline dihydrobromide, 2-, 3-, 4th - Aminophenol, 2-aminomethyl-4-aminophenol, 2-hydroxymethyl-4-aminophenol o-, m-, p-phenylenediamine, o-, m-toluenediamine, 2,5-diaminotoluene, -phenol, -phenethol, 4-amino -3-methylphenol, 2- (2,5-di
- R 6 , R 7 , R 8 , R 9 and R 10 represent hydrogen, a hydroxy or an amino group
- C ⁇ alkyl, C M hydroxyalkyl, C ⁇ alkoxy, C ⁇ aminoalkyl or C ⁇ alkoxy-C ⁇ - alkyl groups may be substituted, or represent a sulfonic acid group, and
- the abovementioned compounds can be used both in free form and in the form of their physiologically tolerable salts, in particular as salts of inorganic acids, such as hydrochloric or sulfuric acid.
- Suitable nitrogen-containing heterocyclic compounds are, for example, 2-, 3-, 4-amino, 2-amino-3-hydroxy, 2,6-diamino, 2,5-diamino, 2,3-diamino, 2-dimethylamino -5- amino, 2-methylamino-3-amino-6-methoxy, 2,3-diamino-6-methoxy, 2,6-dimethoxy-3,5-diamino, 2,4,5-triamino-, 2nd , 6-dihydroxy-3,4-dimethylpyridine, 2,4-dihydroxy-5,6-diamino-, 4,5,6-triamino-, 4-hydroxy-2,5,6-triamino-, 2-hydroxy- 4,5,6-triamino-, 2,4,5,6-tetraamino-, 2-methylamino-4,5,6-triamino-, 2,4-, 4,5-diamino-, 2-amino-4 - methoxy-6-
- amino acids are suitable as amino acids, e.g. obtained by hydrolysis from vegetable or animal proteins, e.g. Collagen, keratin, casein, elastin, soy protein, wheat gluten or almond protein, accessible amino acids. Both acidic and alkaline amino acids can be used.
- Preferred amino acids are arginine, histidine, tyrosine, phenylalanine, DOPA (dihydroxyphenylalanine), ornithine, lysine and tryptophan.
- the oligopeptides can be naturally occurring or synthetic oligopeptides, but also the oligopeptides contained in polypeptide or protein hydrolyzates, provided that they have sufficient water solubility for use in the colorants according to the invention.
- examples are e.g. Glutathione or the oligopeptides contained in the hydrolyzates of collagen, keratin, casein, elastin, soy protein, wheat gluten or almond protein.
- Use together with compounds having a primary or secondary amino group or with aromatic hydroxy compounds is preferred.
- Suitable aromatic hydroxy compounds are, for example, 2-, 4-, 5-methylresorcinol, 2,5-dimethylresorcinol, resorcinol, 3-methoxyphenol, pyrocatechol, hydroquinone, pyrogallol, phloroglucinol, hydroxyhydroquinone, 2-, 3-, 4-methoxy, 3- Dimethylamino, 2- (2-hydroxyethyl) -, 3,4-methylenedioxyphenol, 2,4-, 3,4-dihydroxybenzoic acid, phenylacetic acid, gallic acid, 2,4,6-trihydroxybenzoic acid, acetophenone, 2-, 4-chlororesorcinol, 1-naphthol, 1, 5-, 2,3-, 2,7-dihydroxynaphthalene, 6-dimethylamino-4-hydroxy- 2-naphthalenesulfonic acid, 3,6-dihydroxy-2,7-naphthalenesulfonic acid.
- CH-active compounds which may be mentioned are 1, 2,3,3-tetramethyl-3H-indium iodide, 1, 2,3,3-tetramethylindolium-p-toluenesulfonate, 1, 2,3,3-tetramethyl-3H-indolium methanesulfonate, 2,3-dimethyl-benzothiazolium iodide, 2,3-dimethyl-benzothiazolium-p-toluenesulfonate, rhodanine, rhodanine-3-acetic acid, 1-ethyl-2-quinal dinium iodide, 1-methyl-2-quinal dinium iodide barbituric acid, Thiobarbituric acid, 1, 3-dimethylthiobarbituric acid, diethylthiobarbituric acid, oxindole, 3-indoxy acetate, coumaranone and 1-methyl-3-phenyl-2-pyrazolinone.
- the compounds of component B are particularly preferably selected from the group consisting of N- (2-hydroxyethyl) -N-ethyl-, 2-chloro-p-phenylenediamine, N, N-bis (2-hydroxyethyl) -p-phenylenediamine, 4-aminophenol, p-phenylenediamine, 2- (2,5-diaminophenyl) ethanol, 2,5-diaminotoluene, 3,4-methylenedioxyaniline, 3-amino-2,4-dichloro-, 2-methyl-5-amino -, 3-methyl-4-amino-, 2-methyl-5- (2-hydroxyethylamino) -, 2-methyl-5-amino-4-chloro, 6-methyl-3-amino-2-chloro, 2-aminomethyl-4-aminophenol, 2-hydroxymethyl-4-aminophenol, 3,4-methylenedioxyphenol, 3,4-diaminobenzoic acid, 2,5-d
- oxidizing agents for example H 2 O 2
- Oxidizing agents are generally used in an amount of 0.01 to 6% by weight, based on the application solution.
- a preferred oxidizing agent for human hair is H 2 O 2 .
- the colorants according to the invention result in a wide range of color shades in the range from yellow-orange to brown-black; the fastness properties are excellent, the sensitization potential is very low.
- the colorants according to the invention contain, in addition to the compounds contained according to the invention and possibly further oxidation dye precursors, customary direct dyes, e.g. from the group of nitrophenylenediamines, nitroaminophenols, azo dyes, anthraquinones or indophenols.
- Preferred direct dyes are those under the international names or trade names HC Yeliow 2, HC Yellow 4, HC Yellow 5, HC Yellow 6, Basic Yellow 57, Disperse Orange 3, HC Red 3, HC Red BN, Basic Red 76, HC Blue 2, HC Blue 12, Disperse Blue 3, Basic Blue 99, HC Violet 1, Disperse Violet 1, Disperse Violet 4, Disperse Black 9, Basic Brown 16 and Basic Brown 17 known compounds and 4-amino-2-nitrodiphenylamine-2 ' -carboxylic acid, 6-nitro-1, 2,3,4-tetrahydroquinoxaline, hydroxyethyl-2-nitro-toluidine, picramic acid, 2-amino-6-chloro-4-nitrophenol 4-ethylamino-3-nitrobenzoic acid and 2- Chloro-6-ethylamino-1-hydroxy-4-nitrobenzene.
- the agents according to the invention in accordance with this embodiment preferably contain the substantive dyes in an amount of 0.01 to 20% by weight, based on the total colorant
- preparations according to the invention can also contain naturally occurring dyes such as, for example, henna red, henna neutral, henna black, chamomile flowers, sandalwood, black tea, rotten bark, sage, blue wood, madder root, catechu, sedre and alkanna root.
- naturally occurring dyes such as, for example, henna red, henna neutral, henna black, chamomile flowers, sandalwood, black tea, rotten bark, sage, blue wood, madder root, catechu, sedre and alkanna root.
- Further dye components contained in the colorants according to the invention can also be indoles and indolines, and also their physiologically tolerable salts.
- Preferred examples are 5,6-dihydroxyindole, N-methyl-5,6-dihydroxyindole, N-ethyl-5,6-dihydroxyindole, N-propyl-5,6-dihydroxyindole, N-butyl-5,6-dihydroxyindole, 6 - hydroxyindole, 6-aminoindole and 4-aminoindole.
- the compounds of component B and possibly further oxidation dye precursors or the optional direct dyes each represent uniform compounds. Rather, in the colorants according to the invention, due to the production processes for the individual dyes, further components may also be present in minor amounts, provided that these do not adversely affect the coloring result or for other reasons, e.g. toxicological, must be excluded.
- the colorants according to the invention produce intensive colorations even at physiologically tolerable temperatures of below 45 ° C. They are therefore particularly suitable for dyeing human hair.
- the colorants can usually be incorporated into a water-containing cosmetic carrier.
- Suitable water-containing cosmetic carriers are e.g. Creams, emulsions, gels or also surfactant-containing foaming solutions such as Shampoos or other preparations that are suitable for use on the keratin fibers. If necessary, it is also possible to incorporate the colorants into anhydrous carriers.
- the colorants according to the invention can contain all active substances, additives and auxiliaries known in such preparations.
- the colorants contain at least one surfactant, both anionic and zwitterionic, ampholytic, nonionic and cationic surfactants being suitable in principle.
- Suitable anionic surfactants in preparations according to the invention are all anionic surface-active substances suitable for use on the human body. These are characterized by a water-solubilizing, anionic group such as. B.
- the molecule can contain glycol or polyglycol ether groups, ester, ether and amide groups and hydroxyl groups.
- suitable anionic surfactants are, in each case in the form of the sodium, potassium and ammonium as well as the mono-, di- and trialkanol ammonium salts with 2 or 3 carbon atoms in the alkanol group,
- Ether carboxylic acids of the formula RO- (CH 2 -CH 2 O) x -CH 2 -COOH, in which R is a linear alkyl group with 10 to 22 C atoms and x 0 or 1 to 16, acyl sarcosides with 10 to 18 C- Atoms in the acyl group, acyl taurides with 10 to 18 C atoms in the acyl group, acyl isethionates with 10 to 18 C atoms in the acyl group, sulfosuccinic acid and dialkyl esters with 8 to 18 C atoms in the alkyl group and sulfosuccinic acid mono-alkyl polyoxyethyl esters with 8 to 18 carbon atoms in the alkyl group and 1 to 6 oxyethyl groups, linear alkane sulfonates with 12 to 18 carbon atoms, linear alpha olefin sulfonates with 12 to 18 carbon atoms, methyl alpha
- esters of tartaric acid and citric acid with alcohols which are adducts of about 2-15 molecules of ethylene oxide and / or propylene oxide with fatty alcohols with 8 to 22 carbon atoms.
- Preferred anionic surfactants are alkyl sulfates, alkyl polyglycol ether sulfates and ether carboxylic acids with 10 to 18 carbon atoms in the alkyl group and up to 12 glycol ether groups in the molecule, and in particular salts of saturated and in particular unsaturated C 8 -C 22 carboxylic acids, such as oleic acid, stearic acid, isostearic acid and palmitic acid.
- Zwitterionic surfactants are surface-active compounds that contain at least one quaternary ammonium group and at least one - COO ⁇ or -SO ⁇ group in the molecule.
- Particularly suitable zwitterionic surfactants are the so-called betaines such as the N-alkyl-N, N-dimethylammonium glycinate, for example the coconut alkyl dimethylammonium glycinate, N-acylaminopropyl-N, N-dimethylammonium glycinate, for example the coconut acylaminopropyl dimethylammonium glycinate, and 2 -Alkyl-3-carboxymethyl-3-hydroxyethyl-imidazolines each having 8 to 18 carbon atoms in the alkyl or acyl group and the cocoacylaminoethylhydroxyethylcarboxymethyiglycinate.
- a preferred zwitterionic surfactant is the fatty acid amide deh
- Ampholytic surfactants are understood to mean those surface-active compounds which, in addition to a C 8 . 18 -alkyl or -acyl group in the molecule contain at least one free amino group and at least one -COOH or -SO 3 H group and are capable of forming internal salts.
- ampholytic surfactants are N-alkylglycine, N-alkylpropionic acid, N-alkylaminobutyric acid, N-alkyliminodipropionic acid, N-hydroxyethyl-N-alkylamidopropylglycine, N-alkyltaurine, N-alkylsarcosine, 2-alkylaminopropionic acid and alkylaminoacetic acid each with about 8 to 18 C. Atoms in the alkyl group.
- Particularly preferred ampholytic surfactants are N-cocoalkylaminopropionate, cocoacylaminoethylamino propionate and C 12 . 18 acyl sarcosine.
- Nonionic surfactants contain z as a hydrophilic group.
- B a polyol group, a polyalkylene glycol ether group or a combination of polyol and polyglycol ether group.
- Such connections are, for example Addition products of 2 to 30 mol of ethylene oxide and / or 0 to 5 mol of propylene oxide with linear fatty alcohols with 8 to 22 carbon atoms, with fatty acids with 12 to
- Examples of the cationic surfactants which can be used in the hair treatment compositions according to the invention are, in particular, quaternary ammonium compounds.
- Ammonium halides such as alkyltrimethylammonium chlorides, dialkyldimethylammonium chlorides and trialkylmethylammonium chlorides, e.g. B. cetyltrimethylammonium chloride, stearyltrimethylammonium chloride, distearyldimethylammonium chloride, lauryldimethylammonium chloride, lauryldimethylbenzylammonium chloride and tricetylmethylammonium chloride.
- the quaternized protein hydrolyzates are further cationic surfactants which can be used according to the invention.
- cationic silicone oils such as, for example, the commercially available products Q2-7224 (manufacturer: Dow Coming; a stabilized trimethylsilylamodimethicone), Dow Corning 929 emulsion (containing a hydroxylamino-modified silicone, which is also referred to as amodimethicone), SM -2059 (manufacturer: General Electric), SLM-55067 (manufacturer: Wacker) and Abil®-Quat 3270 and 3272 (manufacturer: Th. Goldschmidt; diquaternary polydimethylsiloxanes, Quaterium-80).
- alkylamidoamines especially fatty acid amidoamines such as the stearylamidopropyldimethylamine available under the name Tego Amid®S 18, are characterized in particular by their good biodegradability. Also very good biodegradability are quaternary Estereducatingen, so-called “esterquats”, kylammoniummethosulfate such as those sold under the trademark Stepantex ® and methyl dialkyl ammonium methosulfates-hydroxyalkyldialkoyloxyalkyl.
- quaternary sugar derivative that can be used as a cationic surfactant is the commercial product Glucquat®100, according to the CTFA nomenclature a "lauryl methyl gluceth-10 hydroxypropyl dimonium chloride".
- the compounds with alkyl groups used as surfactants can each be uniform substances. However, it is generally preferred to start from natural vegetable or animal raw materials in the production of these substances, so that substance mixtures with different alkyl chain lengths depending on the respective raw material are obtained.
- both products with a "normal” homolog distribution and those with a narrowed homolog distribution can be used.
- “Normal” homolog distribution is understood to mean mixtures of homologs which are obtained as catalysts from the reaction of fatty alcohol and alkylene oxide using alkali metals, alkali metal hydroxides or alkali metal alcoholates.
- narrow homolog distributions are obtained if, for example, hydrotalcites, alkaline earth metal salts of ether carboxylic acids, alkaline earth metal oxides,
- hydroxides or alcoholates can be used as catalysts.
- the use of products with a narrow homolog distribution can be preferred.
- nonionic polymers such as vinylpyrrolidone / vinyl acrylate copolymers, polyvinylpyrrolidone and vinylpyrrolidone / vinyl acetate copolymers and
- Thickeners such as agar agar, guar gum, alginates, xanthan gum, gum arabic, karaya gum, locust bean gum, linseed gums, dextrans,
- Cellulose derivatives e.g. B. methyl cellulose, hydroxyalkyl cellulose and carboxymethyl cellulose, starch fractions and derivatives such as amylose, amylopectin and
- Dextrins clays such as e.g. B. bentonite or fully synthetic hydrocolloids such as e.g.
- Structurants such as glucose and maleic acid, hair-conditioning compounds such as phospholipids, for example soy lecithin, egg lecithin and cephalins, and silicone oils,
- Protein hydrolyzates especially elastin, collagen, keratin, milk protein,
- Solubilizers such as ethanol, isopropanol, ethylene glycol, propylene glycol,
- Anti-dandruff agents such as piroctone olamine and zinc omadine, other substances for adjusting the pH value,
- Active ingredients such as panthenol, pantothenic acid, allantoin, pyrrolidone carboxylic acids and their salts, plant extracts and vitamins,
- Consistency enhancers such as sugar esters, polyol esters or polyol alkyl ethers, Fats and waxes such as whale, beeswax, montan wax, paraffins, fatty alcohols and fatty acid esters, fatty acid alkanolamides,
- Swelling and penetration substances such as glycerol, propylene glycol monoethyl ether, carbonates, hydrogen carbonates, guanidines, ureas and primary, secondary and tertiary phosphates, imidazoles, tannins, pyrrole, opacifiers such as latex,
- Pearlescent agents such as ethylene glycol mono- and distearate
- Blowing agents such as propane-butane mixtures, N 2 0, dimethyl ether, CO 2 and air as well as antioxidants.
- constituents of the water-containing carrier are used to produce the colorants according to the invention in amounts customary for this purpose; e.g. emulsifiers are used in concentrations of 0.5 to 30% by weight and thickeners in concentrations of 0.1 to 25% by weight of the total colorant.
- Suitable metal salts are e.g. Formates, carbonates, halides, sulfates, butyrates, valerates, capronates, acetates, lactates, glycolates, tartrates, citrates, gluconates, propionates, phosphates and phosphonates of alkaline earth metals, such as potassium, sodium or lithium, alkaline earth metals, such as magnesium, calcium, strontium or Barium, or of aluminum, manganese, iron, cobalt, copper or zinc, with sodium acetate, lithium bromide, calcium bromide, calcium gluconate, zinc chloride, zinc sulfate, magnesium chloride, magnesium sulfate, ammonium carbonate, chloride and acetate being preferred.
- These salts are preferably present in an amount of 0.03 to 65, in particular 1 to 40 mmol, based on 100 g of the total colorant.
- the pH of the ready-to-use coloring preparations is usually between 2 and 11, preferably between 5 and 9.
- the colorants are generally in the form of the water-containing cosmetic carrier in an amount of 100 g applied to the hair, left there for about 30 minutes and then rinsed out or washed out with a commercially available hair shampoo.
- the aldehydes of the formulas Ia and Ib and the compounds of component B can either be applied to the hair at the same time or else in succession, it being irrelevant which of the two components is applied first.
- the optionally contained ammonium or metal salts can be added to the first or the second component. There may be an interval of up to 30 minutes between the application of the first and the second component. Pretreatment of the fibers with the saline solution is also possible.
- the aldehydes of the formulas Ia and Ib and the compounds of component B can either be stored separately or together, either in a liquid to pasty preparation (aqueous or anhydrous) or as a dry powder. If the components are stored together in a liquid preparation, this should be largely anhydrous to reduce a reaction of the components. When stored separately, the reactive components are only intimately mixed with one another immediately before use. In dry storage, a defined amount of warm (50 to 80 ° C) water is usually added before use and a homogeneous mixture is produced.
- a slurry of 10 mmol of an aldehyde with the formula Ia or Ib, 10 mmol of a reactant, 10 mmol of sodium acetate and a drop of a 20% fatty alkyl ether sulfate solution in 100 ml of water were prepared.
- the slurry was briefly heated to about 80 ° C and filtered after cooling, the pH was then adjusted to 6.
- a strand of 90% gray, untreated human hair was placed in this dyeing solution at 30 ° C. for 30 minutes.
- the colored streak was then rinsed for 30 seconds with lukewarm water, dried in a warm (30-40 ° C.) air stream and then combed out. Then the colorations are assessed visually in daylight.
- the color depth was rated on the following scale:
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP98922727A EP0977546A1 (en) | 1997-04-24 | 1998-04-16 | Use of unsaturated aldehydes in dying fibers containing keratin |
| JP54497798A JP2001524091A (en) | 1997-04-24 | 1998-04-16 | Use of unsaturated aldehydes for dyeing keratin-containing fibers |
| PL98336070A PL336070A1 (en) | 1997-04-24 | 1998-04-16 | Application of unsaturated aldehydes in dyeing creatine-containing fibres |
| SK1455-99A SK145599A3 (en) | 1997-04-24 | 1998-04-16 | Use of unsaturated aldehydes in dying fibers containing keratin |
| BR9809417-3A BR9809417A (en) | 1997-04-24 | 1998-04-16 | Application of unsaturated villagers to dye keratinous fibers |
| CA002288055A CA2288055A1 (en) | 1997-04-24 | 1998-04-16 | Use of unsaturated aldehydes in dying fibers containing keratin |
| AU75264/98A AU726113B2 (en) | 1997-04-24 | 1998-04-16 | Use of unsaturated aldehydes in dying fibres containing keratin |
| NO995157A NO995157L (en) | 1997-04-24 | 1999-10-22 | Use of unsaturated aldehydes in drawing fibers containing keratin |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19717224.5 | 1997-04-24 | ||
| DE19717224A DE19717224A1 (en) | 1997-04-24 | 1997-04-24 | Use of unsaturated aldehydes for dyeing keratin fibers |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1998047473A1 true WO1998047473A1 (en) | 1998-10-29 |
Family
ID=7827552
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP1998/002243 WO1998047473A1 (en) | 1997-04-24 | 1998-04-16 | Use of unsaturated aldehydes in dying fibers containing keratin |
Country Status (12)
| Country | Link |
|---|---|
| EP (1) | EP0977546A1 (en) |
| JP (1) | JP2001524091A (en) |
| CN (1) | CN1252707A (en) |
| AU (1) | AU726113B2 (en) |
| BR (1) | BR9809417A (en) |
| CA (1) | CA2288055A1 (en) |
| DE (1) | DE19717224A1 (en) |
| HU (1) | HUP0002707A3 (en) |
| NO (1) | NO995157L (en) |
| PL (1) | PL336070A1 (en) |
| SK (1) | SK145599A3 (en) |
| WO (1) | WO1998047473A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002533371A (en) * | 1998-12-23 | 2002-10-08 | ロレアル | Dyeing method using a specific cationic derivative and a compound selected from a specific aldehyde, a specific ketone, a quinone, and a di-imino-isoindoline or 3-amino-isoindolone derivative |
| JP2002533370A (en) * | 1998-12-23 | 2002-10-08 | ロレアル | Dyeing method using a specific active methylene compound and a compound selected from a specific aldehyde, a specific ketone, a quinone, and a di-imino-isoindoline or 3-amino-isoindolone derivative |
| US6537330B1 (en) * | 1998-06-23 | 2003-03-25 | Henkel Kommanditgesellschaft Auf Aktien | Colorants |
| WO2003030848A1 (en) | 2001-10-02 | 2003-04-17 | Henkel Kommanditgesellschaft Auf Aktien | Method for colouring keratin fibres using carbonyl compounds to improve the colour stability of hair coloration |
| US6790239B1 (en) | 1999-10-23 | 2004-09-14 | Henkel Kommanditgesellschaft Auf Aktien | Agent for coloring keratin containing fibers |
| WO2008028861A1 (en) * | 2006-09-05 | 2008-03-13 | Henkel Ag & Co. Kgaa | Substances for dyeing keratinous fibers |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19820894A1 (en) * | 1998-05-09 | 1999-11-11 | Wella Ag | Two-component fiber dyeing agent, especially for use on hair |
| ES2214055T3 (en) | 1998-12-07 | 2004-09-01 | Wella Aktiengesellschaft | PRODUCT FOR DYING FIBERS. |
| WO2000052100A1 (en) * | 1999-02-27 | 2000-09-08 | Wella Aktiengesellschaft | Agents for colouring fibres |
| DE19916030A1 (en) * | 1999-04-09 | 2000-10-19 | Henkel Kgaa | Colorants and uses |
| DE19949033A1 (en) * | 1999-10-12 | 2001-04-19 | Henkel Kgaa | Hair coloring process |
| DE19950404B4 (en) | 1999-10-20 | 2004-07-15 | Wella Ag | Means and processes for dyeing hair and a multi-component kit for dyeing and later removing hair |
| DE10045856A1 (en) | 2000-09-14 | 2002-03-28 | Henkel Kgaa | Colorant for keratin fibers, useful for coloring fur, wool, feathers and especially human hair, contains benzo(b)furan-3-one or benzo(b)thiophen-3-one derivative(s) and reactive carbonyl or methine-active compound(s) |
| JP4500050B2 (en) | 2002-01-15 | 2010-07-14 | チバ ホールディング インコーポレーテッド | Yellow cationic dye for dyeing organic materials |
| DE10218588A1 (en) | 2002-04-26 | 2003-11-06 | Wella Ag | Agent for the oxidative dyeing of keratin fibers |
| JP3675776B2 (en) * | 2002-05-02 | 2005-07-27 | 倉敷紡績株式会社 | How to control the color of fibers |
| DE10260881A1 (en) * | 2002-12-23 | 2004-07-08 | Henkel Kgaa | Agent for dyeing keratin fibers |
| EP2075036B1 (en) | 2004-04-08 | 2013-11-06 | Basf Se | Composition for dyeing keratin containing fibres comprising disulfide dyes and method |
| ES2433131T3 (en) | 2005-08-30 | 2013-12-09 | Basf Se | Dyes containing a thiol group |
| WO2006136617A2 (en) | 2005-10-11 | 2006-12-28 | Ciba Specialty Chemicals Holding Inc. | Mixture of sulfide dyes |
| MX2008015854A (en) | 2006-06-13 | 2009-02-23 | Ciba Holding Inc | Tricationic dyes. |
| US8337569B2 (en) | 2009-07-15 | 2012-12-25 | Basf Se | Polymeric hair dyes |
| EP2606095B1 (en) | 2010-08-17 | 2016-10-12 | Basf Se | Disulfide or thiol polymeric hair dyes |
| US8992633B2 (en) | 2011-05-03 | 2015-03-31 | Basf Se | Disulfide dyes |
| RU2680068C2 (en) | 2013-09-02 | 2019-02-14 | Л'Ореаль | Method for dyeing keratin fibres using cationic styryl disulphide dyes, and composition including said dyes |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1160919A (en) * | 1965-07-29 | 1969-08-06 | Oreal | Process for Treating Hair and Compositions therefor |
| US3871818A (en) * | 1972-10-30 | 1975-03-18 | Avon Prod Inc | Promoting color change in human hair with a dialdehyde compound and a nitrogen containing compound |
| DE19501304A1 (en) * | 1995-01-18 | 1996-07-25 | Henkel Kgaa | Diketo compounds for dyeing keratin fibers |
-
1997
- 1997-04-24 DE DE19717224A patent/DE19717224A1/en not_active Withdrawn
-
1998
- 1998-04-16 EP EP98922727A patent/EP0977546A1/en not_active Withdrawn
- 1998-04-16 BR BR9809417-3A patent/BR9809417A/en not_active IP Right Cessation
- 1998-04-16 AU AU75264/98A patent/AU726113B2/en not_active Ceased
- 1998-04-16 WO PCT/EP1998/002243 patent/WO1998047473A1/en not_active Application Discontinuation
- 1998-04-16 CN CN98804359.9A patent/CN1252707A/en active Pending
- 1998-04-16 HU HU0002707A patent/HUP0002707A3/en unknown
- 1998-04-16 SK SK1455-99A patent/SK145599A3/en unknown
- 1998-04-16 CA CA002288055A patent/CA2288055A1/en not_active Abandoned
- 1998-04-16 JP JP54497798A patent/JP2001524091A/en active Pending
- 1998-04-16 PL PL98336070A patent/PL336070A1/en unknown
-
1999
- 1999-10-22 NO NO995157A patent/NO995157L/en not_active Application Discontinuation
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1160919A (en) * | 1965-07-29 | 1969-08-06 | Oreal | Process for Treating Hair and Compositions therefor |
| US3871818A (en) * | 1972-10-30 | 1975-03-18 | Avon Prod Inc | Promoting color change in human hair with a dialdehyde compound and a nitrogen containing compound |
| DE19501304A1 (en) * | 1995-01-18 | 1996-07-25 | Henkel Kgaa | Diketo compounds for dyeing keratin fibers |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6537330B1 (en) * | 1998-06-23 | 2003-03-25 | Henkel Kommanditgesellschaft Auf Aktien | Colorants |
| JP2002533371A (en) * | 1998-12-23 | 2002-10-08 | ロレアル | Dyeing method using a specific cationic derivative and a compound selected from a specific aldehyde, a specific ketone, a quinone, and a di-imino-isoindoline or 3-amino-isoindolone derivative |
| JP2002533370A (en) * | 1998-12-23 | 2002-10-08 | ロレアル | Dyeing method using a specific active methylene compound and a compound selected from a specific aldehyde, a specific ketone, a quinone, and a di-imino-isoindoline or 3-amino-isoindolone derivative |
| US6790239B1 (en) | 1999-10-23 | 2004-09-14 | Henkel Kommanditgesellschaft Auf Aktien | Agent for coloring keratin containing fibers |
| WO2003030848A1 (en) | 2001-10-02 | 2003-04-17 | Henkel Kommanditgesellschaft Auf Aktien | Method for colouring keratin fibres using carbonyl compounds to improve the colour stability of hair coloration |
| WO2008028861A1 (en) * | 2006-09-05 | 2008-03-13 | Henkel Ag & Co. Kgaa | Substances for dyeing keratinous fibers |
Also Published As
| Publication number | Publication date |
|---|---|
| AU726113B2 (en) | 2000-11-02 |
| BR9809417A (en) | 2000-06-13 |
| NO995157D0 (en) | 1999-10-22 |
| DE19717224A1 (en) | 1998-10-29 |
| AU7526498A (en) | 1998-11-13 |
| CN1252707A (en) | 2000-05-10 |
| JP2001524091A (en) | 2001-11-27 |
| HUP0002707A2 (en) | 2000-12-28 |
| SK145599A3 (en) | 2000-05-16 |
| CA2288055A1 (en) | 1998-10-29 |
| NO995157L (en) | 1999-10-22 |
| PL336070A1 (en) | 2000-06-05 |
| HUP0002707A3 (en) | 2002-11-28 |
| EP0977546A1 (en) | 2000-02-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1037586A1 (en) | Utilization of onium aldehydes and onium ketones for dying fibers containing keratin | |
| WO2001003651A2 (en) | Agent for dyeing fibres containing keratin | |
| WO2000038636A1 (en) | Agent for dying keratin fibers | |
| EP0873745A2 (en) | Dyestuff for keratinous fibers | |
| EP0977546A1 (en) | Use of unsaturated aldehydes in dying fibers containing keratin | |
| EP0873744A2 (en) | Use of heterocyclic carbonyl compounds for dying keratinous fibers | |
| EP0977545B1 (en) | The use of 1-substituted isatins to dye fibers containing keratin | |
| WO2000038632A1 (en) | Method for dying keratin fibers using 2-oxocarboxylic acid derivatives in conjunction with nucleophiles | |
| EP1113779A1 (en) | Method for dyeing fibres containing keratin using stable diazonium salts | |
| EP1028694A1 (en) | Application of indanones for dying fibers containing keratin | |
| WO2000076466A1 (en) | Xanthene derivatives for dying keratin fibers | |
| EP1139988B1 (en) | Agent for dying keratin fibers | |
| EP0873746A2 (en) | Dyestuff for keratinous fibers | |
| EP0873743A2 (en) | Use of benzylidene-ketones for dyeing keratinous fibers | |
| EP1028695A1 (en) | Agent for dyeing fibers containing keratin with a concentration of dehydroascorbic acid content | |
| EP1139990B1 (en) | Agent for dying keratin fibers | |
| WO1999019558A2 (en) | Use of malonaldehyde derivatives for dyeing fibres containing keratin | |
| EP0820760A2 (en) | Use of aldehydes for dyeing keratinic fibres | |
| EP1194116B1 (en) | Agent for dyeing fibres containing keratin | |
| WO2000038634A1 (en) | Agent for dying keratin fibers | |
| WO2001005359A2 (en) | Agent for coloring keratin fibers that contains at least one quaternary heterocyclic aldehyde or one ketone | |
| WO2000015183A1 (en) | Agents for dyeing fibres containing keratin | |
| WO2001003661A1 (en) | Agent for dyeing keratin fibres containing at least one aromatic nitroso compound | |
| WO1999004754A1 (en) | Use of di- and oligoacylaromats for colouring fibres containing keratin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 98804359.9 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AU BR CA CN CZ HU JP NO PL RU SK US VN |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 1998922727 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 145599 Country of ref document: SK Ref document number: 09403531 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2288055 Country of ref document: CA Ref document number: 2288055 Country of ref document: CA Kind code of ref document: A Ref document number: 1998 544977 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PV1999-3740 Country of ref document: CZ Ref document number: 1199900891 Country of ref document: VN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 75264/98 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 1998922727 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: PV1999-3740 Country of ref document: CZ |
|
| WWG | Wipo information: grant in national office |
Ref document number: 75264/98 Country of ref document: AU |
|
| WWR | Wipo information: refused in national office |
Ref document number: PV1999-3740 Country of ref document: CZ |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1998922727 Country of ref document: EP |