WO1997005190A1 - Block copolymers - Google Patents
Block copolymers Download PDFInfo
- Publication number
- WO1997005190A1 WO1997005190A1 PCT/US1996/012421 US9612421W WO9705190A1 WO 1997005190 A1 WO1997005190 A1 WO 1997005190A1 US 9612421 W US9612421 W US 9612421W WO 9705190 A1 WO9705190 A1 WO 9705190A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- poly
- oar
- diyl
- block copolymer
- oxyethylene
- Prior art date
Links
- 229920001400 block copolymer Polymers 0.000 title claims description 122
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims abstract description 89
- 238000000034 method Methods 0.000 claims abstract description 23
- 230000002209 hydrophobic effect Effects 0.000 claims abstract description 12
- 229920001451 polypropylene glycol Polymers 0.000 claims abstract description 8
- -1 1,4-phenylene, 1,3-phenylene Chemical group 0.000 claims description 108
- 229920000642 polymer Polymers 0.000 claims description 93
- 239000000243 solution Substances 0.000 claims description 67
- 229920001577 copolymer Polymers 0.000 claims description 62
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 56
- 238000005266 casting Methods 0.000 claims description 53
- 229920002492 poly(sulfone) Polymers 0.000 claims description 51
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 claims description 50
- 229920001223 polyethylene glycol Polymers 0.000 claims description 32
- 239000002202 Polyethylene glycol Substances 0.000 claims description 26
- 125000005010 perfluoroalkyl group Chemical group 0.000 claims description 26
- 229920000428 triblock copolymer Polymers 0.000 claims description 25
- 238000005345 coagulation Methods 0.000 claims description 23
- 230000015271 coagulation Effects 0.000 claims description 23
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 23
- 239000000203 mixture Substances 0.000 claims description 22
- 238000000137 annealing Methods 0.000 claims description 18
- 229920001600 hydrophobic polymer Polymers 0.000 claims description 17
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 15
- 229920000359 diblock copolymer Polymers 0.000 claims description 14
- 239000011159 matrix material Substances 0.000 claims description 14
- 239000000654 additive Substances 0.000 claims description 13
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 12
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 12
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 12
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 12
- 125000003118 aryl group Chemical group 0.000 claims description 11
- 235000010290 biphenyl Nutrition 0.000 claims description 11
- 239000004305 biphenyl Substances 0.000 claims description 11
- 125000006267 biphenyl group Chemical group 0.000 claims description 11
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N phenylbenzene Natural products C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 claims description 11
- 239000003880 polar aprotic solvent Substances 0.000 claims description 11
- 229920001169 thermoplastic Polymers 0.000 claims description 11
- 125000004400 (C1-C12) alkyl group Chemical group 0.000 claims description 10
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 10
- 125000002877 alkyl aryl group Chemical group 0.000 claims description 10
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 claims description 10
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 10
- 238000004519 manufacturing process Methods 0.000 claims description 9
- 150000003457 sulfones Chemical class 0.000 claims description 9
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 claims description 8
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 claims description 8
- 239000004698 Polyethylene Substances 0.000 claims description 8
- 238000010438 heat treatment Methods 0.000 claims description 8
- 229920000573 polyethylene Polymers 0.000 claims description 8
- 239000003153 chemical reaction reagent Substances 0.000 claims description 7
- 229920005604 random copolymer Polymers 0.000 claims description 7
- ZDZHCHYQNPQSGG-UHFFFAOYSA-N 1-naphthalen-1-ylnaphthalene Chemical compound C1=CC=C2C(C=3C4=CC=CC=C4C=CC=3)=CC=CC2=C1 ZDZHCHYQNPQSGG-UHFFFAOYSA-N 0.000 claims description 5
- IYDMICQAKLQHLA-UHFFFAOYSA-N 1-phenylnaphthalene Chemical compound C1=CC=CC=C1C1=CC=CC2=CC=CC=C12 IYDMICQAKLQHLA-UHFFFAOYSA-N 0.000 claims description 5
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 claims description 5
- 239000004743 Polypropylene Substances 0.000 claims description 5
- 230000000996 additive effect Effects 0.000 claims description 5
- 125000000217 alkyl group Chemical group 0.000 claims description 5
- 125000002947 alkylene group Chemical group 0.000 claims description 5
- 229920001155 polypropylene Polymers 0.000 claims description 5
- 125000006273 (C1-C3) alkyl group Chemical group 0.000 claims description 4
- 229920005682 EO-PO block copolymer Polymers 0.000 claims description 4
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 4
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 claims description 3
- 239000004952 Polyamide Substances 0.000 claims description 3
- 239000004697 Polyetherimide Substances 0.000 claims description 3
- 239000004642 Polyimide Substances 0.000 claims description 3
- 238000001125 extrusion Methods 0.000 claims description 3
- 229920002239 polyacrylonitrile Polymers 0.000 claims description 3
- 229920002647 polyamide Polymers 0.000 claims description 3
- 229920001601 polyetherimide Polymers 0.000 claims description 3
- 229920001721 polyimide Polymers 0.000 claims description 3
- 229920001291 polyvinyl halide Polymers 0.000 claims description 3
- 229920006214 polyvinylidene halide Polymers 0.000 claims description 3
- YEJRWHAVMIAJKC-UHFFFAOYSA-N 4-Butyrolactone Chemical compound O=C1CCCO1 YEJRWHAVMIAJKC-UHFFFAOYSA-N 0.000 claims description 2
- 125000005362 aryl sulfone group Chemical group 0.000 claims description 2
- 238000001746 injection moulding Methods 0.000 claims description 2
- 229920000469 amphiphilic block copolymer Polymers 0.000 abstract description 2
- 239000012528 membrane Substances 0.000 description 81
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 50
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 30
- 239000002904 solvent Substances 0.000 description 30
- 235000011187 glycerol Nutrition 0.000 description 19
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 13
- 230000015572 biosynthetic process Effects 0.000 description 12
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 12
- 108090000623 proteins and genes Proteins 0.000 description 11
- 102000004169 proteins and genes Human genes 0.000 description 11
- 239000004721 Polyphenylene oxide Substances 0.000 description 10
- 239000000835 fiber Substances 0.000 description 9
- 239000011148 porous material Substances 0.000 description 9
- 238000001179 sorption measurement Methods 0.000 description 9
- 238000011282 treatment Methods 0.000 description 9
- 238000007745 plasma electrolytic oxidation reaction Methods 0.000 description 8
- 238000011084 recovery Methods 0.000 description 8
- 239000004416 thermosoftening plastic Substances 0.000 description 8
- 238000000605 extraction Methods 0.000 description 7
- 239000012510 hollow fiber Substances 0.000 description 7
- 230000035699 permeability Effects 0.000 description 7
- 239000000463 material Substances 0.000 description 6
- 238000002156 mixing Methods 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 5
- 230000007423 decrease Effects 0.000 description 5
- 230000004907 flux Effects 0.000 description 5
- 125000000524 functional group Chemical group 0.000 description 5
- 229920001519 homopolymer Polymers 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- 239000000178 monomer Substances 0.000 description 5
- 229910000027 potassium carbonate Inorganic materials 0.000 description 5
- 235000011181 potassium carbonates Nutrition 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 239000004094 surface-active agent Substances 0.000 description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 4
- 125000004429 atom Chemical group 0.000 description 4
- 229910052799 carbon Inorganic materials 0.000 description 4
- 239000000706 filtrate Substances 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 230000003993 interaction Effects 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- 229920005597 polymer membrane Polymers 0.000 description 4
- 238000001556 precipitation Methods 0.000 description 4
- 239000000376 reactant Substances 0.000 description 4
- 238000010992 reflux Methods 0.000 description 4
- 239000000080 wetting agent Substances 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 3
- 229920002582 Polyethylene Glycol 600 Polymers 0.000 description 3
- MJSNUBOCVAKFIJ-LNTINUHCSA-N chromium;(z)-4-oxoniumylidenepent-2-en-2-olate Chemical compound [Cr].C\C(O)=C\C(C)=O.C\C(O)=C\C(C)=O.C\C(O)=C\C(C)=O MJSNUBOCVAKFIJ-LNTINUHCSA-N 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 238000013461 design Methods 0.000 description 3
- 150000002009 diols Chemical class 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 230000009477 glass transition Effects 0.000 description 3
- 229910052739 hydrogen Inorganic materials 0.000 description 3
- 229920001477 hydrophilic polymer Polymers 0.000 description 3
- 230000005660 hydrophilic surface Effects 0.000 description 3
- 230000008018 melting Effects 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 238000005191 phase separation Methods 0.000 description 3
- 238000006116 polymerization reaction Methods 0.000 description 3
- 238000004626 scanning electron microscopy Methods 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- 238000009736 wetting Methods 0.000 description 3
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 2
- 238000005481 NMR spectroscopy Methods 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 150000001298 alcohols Chemical class 0.000 description 2
- 230000001588 bifunctional effect Effects 0.000 description 2
- 150000001721 carbon Chemical group 0.000 description 2
- 238000012512 characterization method Methods 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 239000000356 contaminant Substances 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 238000001212 derivatisation Methods 0.000 description 2
- 238000000502 dialysis Methods 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 239000004744 fabric Substances 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 230000014509 gene expression Effects 0.000 description 2
- 239000003906 humectant Substances 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 2
- 229920001778 nylon Polymers 0.000 description 2
- 239000006259 organic additive Substances 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 230000008520 organization Effects 0.000 description 2
- 230000005298 paramagnetic effect Effects 0.000 description 2
- 229920000090 poly(aryl ether) Polymers 0.000 description 2
- 229920000515 polycarbonate Polymers 0.000 description 2
- 239000004417 polycarbonate Substances 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 230000000717 retained effect Effects 0.000 description 2
- 239000012465 retentate Substances 0.000 description 2
- 150000003839 salts Chemical class 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 238000004611 spectroscopical analysis Methods 0.000 description 2
- 238000009987 spinning Methods 0.000 description 2
- 238000007669 thermal treatment Methods 0.000 description 2
- 238000000108 ultra-filtration Methods 0.000 description 2
- POILWHVDKZOXJZ-ARJAWSKDSA-M (z)-4-oxopent-2-en-2-olate Chemical compound C\C([O-])=C\C(C)=O POILWHVDKZOXJZ-ARJAWSKDSA-M 0.000 description 1
- BHKKSKOHRFHHIN-MRVPVSSYSA-N 1-[[2-[(1R)-1-aminoethyl]-4-chlorophenyl]methyl]-2-sulfanylidene-5H-pyrrolo[3,2-d]pyrimidin-4-one Chemical compound N[C@H](C)C1=C(CN2C(NC(C3=C2C=CN3)=O)=S)C=CC(=C1)Cl BHKKSKOHRFHHIN-MRVPVSSYSA-N 0.000 description 1
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 1
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 1
- ZXVONLUNISGICL-UHFFFAOYSA-N 4,6-dinitro-o-cresol Chemical group CC1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1O ZXVONLUNISGICL-UHFFFAOYSA-N 0.000 description 1
- LFLSATHZMYYIAQ-UHFFFAOYSA-N 4-flourobenzenesulfonamide Chemical group NS(=O)(=O)C1=CC=C(F)C=C1 LFLSATHZMYYIAQ-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 101100465853 Caenorhabditis elegans psf-2 gene Proteins 0.000 description 1
- 102000018832 Cytochromes Human genes 0.000 description 1
- 108010052832 Cytochromes Proteins 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical group C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 101150046368 PSF1 gene Proteins 0.000 description 1
- 229920000463 Poly(ethylene glycol)-block-poly(propylene glycol)-block-poly(ethylene glycol) Polymers 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 229920000265 Polyparaphenylene Polymers 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000000274 adsorptive effect Effects 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 125000001204 arachidyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 239000012888 bovine serum Substances 0.000 description 1
- 229940098773 bovine serum albumin Drugs 0.000 description 1
- 150000003842 bromide salts Chemical class 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 238000003181 co-melting Methods 0.000 description 1
- FDJOLVPMNUYSCM-WZHZPDAFSA-L cobalt(3+);[(2r,3s,4r,5s)-5-(5,6-dimethylbenzimidazol-1-yl)-4-hydroxy-2-(hydroxymethyl)oxolan-3-yl] [(2r)-1-[3-[(1r,2r,3r,4z,7s,9z,12s,13s,14z,17s,18s,19r)-2,13,18-tris(2-amino-2-oxoethyl)-7,12,17-tris(3-amino-3-oxopropyl)-3,5,8,8,13,15,18,19-octamethyl-2 Chemical compound [Co+3].N#[C-].N([C@@H]([C@]1(C)[N-]\C([C@H]([C@@]1(CC(N)=O)C)CCC(N)=O)=C(\C)/C1=N/C([C@H]([C@@]1(CC(N)=O)C)CCC(N)=O)=C\C1=N\C([C@H](C1(C)C)CCC(N)=O)=C/1C)[C@@H]2CC(N)=O)=C\1[C@]2(C)CCC(=O)NC[C@@H](C)OP([O-])(=O)O[C@H]1[C@@H](O)[C@@H](N2C3=CC(C)=C(C)C=C3N=C2)O[C@@H]1CO FDJOLVPMNUYSCM-WZHZPDAFSA-L 0.000 description 1
- 238000002288 cocrystallisation Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 125000006165 cyclic alkyl group Chemical group 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 238000000113 differential scanning calorimetry Methods 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 229940113088 dimethylacetamide Drugs 0.000 description 1
- 238000011038 discontinuous diafiltration by volume reduction Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 238000000909 electrodialysis Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 150000002222 fluorine compounds Chemical class 0.000 description 1
- 239000013505 freshwater Substances 0.000 description 1
- 238000007306 functionalization reaction Methods 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 150000008282 halocarbons Chemical class 0.000 description 1
- 229920013746 hydrophilic polyethylene oxide Polymers 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 150000004694 iodide salts Chemical class 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 125000005647 linker group Chemical group 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 230000004807 localization Effects 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 238000001471 micro-filtration Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000006082 mold release agent Substances 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 125000006353 oxyethylene group Chemical group 0.000 description 1
- 229920006280 packaging film Polymers 0.000 description 1
- 239000012785 packaging film Substances 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 235000015320 potassium carbonate Nutrition 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 102000004196 processed proteins & peptides Human genes 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000001055 reflectance spectroscopy Methods 0.000 description 1
- 239000013557 residual solvent Substances 0.000 description 1
- 238000001223 reverse osmosis Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 238000001878 scanning electron micrograph Methods 0.000 description 1
- 238000002791 soaking Methods 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 238000000371 solid-state nuclear magnetic resonance spectroscopy Methods 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 230000003595 spectral effect Effects 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 229940124530 sulfonamide Drugs 0.000 description 1
- 150000003456 sulfonamides Chemical class 0.000 description 1
- 238000006277 sulfonation reaction Methods 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229920002725 thermoplastic elastomer Polymers 0.000 description 1
- CMQCNTNASCDNGR-UHFFFAOYSA-N toluene;hydrate Chemical compound O.CC1=CC=CC=C1 CMQCNTNASCDNGR-UHFFFAOYSA-N 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- 238000010626 work up procedure Methods 0.000 description 1
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/52—Polyethers
- B01D71/521—Aliphatic polyethers
- B01D71/5211—Polyethylene glycol or polyethyleneoxide
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
- C08J9/28—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof by elimination of a liquid phase from a macromolecular composition or article, e.g. drying of coagulum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/18—Macromolecular materials obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L29/00—Materials for catheters, medical tubing, cannulae, or endoscopes or for coating catheters
- A61L29/04—Macromolecular materials
- A61L29/06—Macromolecular materials obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/52—Polyethers
- B01D71/522—Aromatic polyethers
- B01D71/5223—Polyphenylene oxide, phenyl ether polymers or polyphenylethers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/76—Macromolecular material not specifically provided for in a single one of groups B01D71/08 - B01D71/74
- B01D71/80—Block polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring
- C08G65/32—Polymers modified by chemical after-treatment
- C08G65/329—Polymers modified by chemical after-treatment with organic compounds
- C08G65/334—Polymers modified by chemical after-treatment with organic compounds containing sulfur
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L71/00—Compositions of polyethers obtained by reactions forming an ether link in the main chain; Compositions of derivatives of such polymers
- C08L71/02—Polyalkylene oxides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/52—Polyethers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/66—Polymers having sulfur in the main chain, with or without nitrogen, oxygen or carbon only
- B01D71/68—Polysulfones; Polyethersulfones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2201/00—Foams characterised by the foaming process
- C08J2201/04—Foams characterised by the foaming process characterised by the elimination of a liquid or solid component, e.g. precipitation, leaching out, evaporation
- C08J2201/054—Precipitating the polymer by adding a non-solvent or a different solvent
- C08J2201/0542—Precipitating the polymer by adding a non-solvent or a different solvent from an organic solvent-based polymer composition
- C08J2201/0544—Precipitating the polymer by adding a non-solvent or a different solvent from an organic solvent-based polymer composition the non-solvent being aqueous
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2371/00—Characterised by the use of polyethers obtained by reactions forming an ether link in the main chain; Derivatives of such polymers
- C08J2371/02—Polyalkylene oxides
Definitions
- Desirable separation membranes are strong, thermally-stable, and resistant to oxidative or corrosive elements in the material to be separated such as acids or chloride ions.
- Hydrophobic polymers such as polysulfones often provide the above characteristics.
- hydrophilic surface is also desirable.
- a hydrophilic or wettable surface on a porous polymer promotes uniform filtration, increases recovery of both filtrate and retentate, and decreases adsorption of material such as protein and other solutes (i.e., fouling). It is worth noting that the surface energies associated with small-diameter pores hamper both an initial wetting of a porous surface, and also a re-wetting of the surface after the membrane has been dried.
- hydrophobic polymers In addition to one method which simply blends hydrophobic polymers with a hydrophilic additive or alloy, there are several methods of treating pre-formed polymer membranes to increase surface hydrophilicity. For example, one method relies on hydrophobic bonding between a pre-formed hydrophobic polymer and an
- One aspect of the invention relates to a
- hydrophilic-hydrophobic diblock copolymer or a
- hydrophilic-hydrophobic-hydrophilic triblock copolymer or a combination thereof.
- X is -Z 1 -(OAr 1 OAr 2 ) m - or -(OAr 2 Q) m -, Q being OAr 1 or [OCH 2 E] u , wherein m is between 1 and 500, each E is independently selected from CH 2 and CH(CH 3 ) , and u is between 1 and 10,000.
- Y is hydroxy, -(OAr 3 O)-Z 2 - [BCH 2 O] p -R 1 , or -(OAr 4 O)-[BCH 2 O] p -R 1 , wherein each B is independently selected from CH 2 and CH(CH 3 ), and p is between 1 and 10,000.
- R 1 is H, C 1-20 alkyl, C 7-20 alkylaryl, C 7-20 arylalkyl, or c i- 20 perfluoroalkyl.
- Each Ar 1 and each Ar 3 is
- diphenylalkylene-4,4'-diyl wherein alkylene is -(CH 2 ) q -, q being 1, 3, 5, 7 or 9, p-terphenyl-4,4'-diyl, and
- diphenylsulfoxide-4,4'-diyl diphenylsulfone-4,4'-diyl, diphenyl ketone-4,4'-diyl, and bivalent radicals of diphenyl-C 1- 12 alkyl phosphine oxide and diphenyl-C 6-20 aryl phosphine oxide.
- Another aspect of the invention features a block copolymer, which includes a central block segment
- polyethylene oxide block and a polyethylene oxide-polypropylene oxide copolymer, wherein the segment is linked between two polyarylsulfone block segments by ether linkages; and a first endgroup and a second
- each endgroup selected independently from polyethylene oxide
- Another aspect of the invention relates to a porous polymer article with a covalently-bonded
- the polymer article includes (i) a hydrophilic-hydrophobic block copolymer (such as a hydrophilic-hydrophobic diblock copolymer, or a hydrophilic-hydrophobic-hydrophilic triblock copolymer, or a combination thereof, e.g., formula (1) above, or formulae (2) or (3), below), and (ii) a hydrophobic polymer.
- the polymer matrix of the article is formed by the hydrophobic polymer mixing with hydrophobic block segments of a disclosed block copolymer of formula (1).
- a hydrophilic moiety includes a partial length or portion of a hydrophilic block segment of the block copolymer.
- Still another aspect of the invention features a method of making a porous polymer article with enhanced hydrophilicity including three steps.
- the first step is providing a casting solution which includes (i) 0.1 - 50 weight percent of a block copolymer of formula (1), (2) or (3), (ii) 0 - 40 weight percent of a hydrophilic polymer, and (iii) 40 - 95 weight percent of a polar aprotic solvent.
- the second step is contacting the casting solution with a nonsolvent coagulation bath until a porous polymer article forms.
- the third step thermally annealing the article in the presence of water to enhance the hydrophilicity of the article.
- active surface means the total surface area, including pores and channels, exposed to a
- the invention features block copolymers designed to form polymer articles such as membranes which spontaneously undergo a phase inversion to form an active surface (e.g., a hydrophilic surface).
- an active surface e.g., a hydrophilic surface
- the active surface generally extends across the exterior membrane faces and generally extends through the interior pore surfaces and channels.
- the active surface of an article is therefore generally greater than the macroscopic or planar dimension.
- a 4 inch by 4 inch membrane has an active surface greater than 16 square inches.
- active surface block segment refers to a block segment of a copolymer which is localized at least in part on the active surface of a polymer article during membrane formation (e.g., poly(oxyethylene) block segment in Example 1). Such localization or presentation results from careful
- An active surface block segment is covalently-bound to an anchor segment of the same block copolymer.
- An anchor segment e.g., polysulfone block segment in Example 1
- a matrix-forming polymer e.g., hydrophobic polymer
- the bond between an anchor segment and an active surface segment may be literally a single covalent bond, or may be a linking moiety, such as an ether linkage (-O-), an amide, ester, or sulfonamide linkage, a monomer or any other organic moiety which does not adversely affect either solvent quality of the casting solution or
- active surface segment moiety means a portion of a block copolymer segment which is present on, or extends above, the active surface of a polymer
- An active surface segment moiety may be any active surface segment moiety.
- An active surface segment moiety has a property different from the bulk or matrix polymer, such as hydrophilicity, chirality, or a specific reactivity or affinity. Active surface segment moieties and presentation thereof are further addressed in the Detailed Discussion section below.
- alkyl includes straight-chain groups such as methyl, n-hexyl, nonyl, tetradecyl, and icosyl; branched groups such as isopropyl, isopentyl, neopentyl, tert-pentyl, 4-ethyl-5-methyloctyl, and 4-isopropyl-5-propyl-nonyl; and cyclic alkyl groups such as
- bivalent radical means a structural moiety with two free valences.
- the bivalent radical is bonded to two other moieties at the sites of the two valences.
- X can be -Z 1 -(OAr 1 OAr 2 ) m -, wherein Z 1 is -N(R 2 )-(SO 2 )-C 6 H 4 -.
- the left-terminal atom of X i.e., the nitrogen atom of Z 1
- perfluoroalkyl means fully-fluorinated straight-chain carbon chains, such as trifluoromethyl and pentafluoroethyl. Perfluoroalkyls can be generalized by
- hydrophilic as in “hydrophilic active surface” means the same as wettable, or capable of forming at least some hydrogen bonds with water by means of polar atoms or groups of atoms. Hydrophilicity can be measured macroscopically by measuring, e.g., water contact angle, permeability, solvent flux, and solute rejection. Even after being dried, a spontaneously wettable surface requires little or no additional
- Certain polymer articles which include the disclosed block copolymers are spontaneously wettable (see, e.g., Example 3).
- weight percent as used above to describe a casting solution means, for example, the weight of a component (e.g., block copolymer, solvent, matrix polymer, or additive, if any) divided by the total weight of the casting solution and then multiplied by 100.
- a component e.g., block copolymer, solvent, matrix polymer, or additive, if any
- the polymer articles disclosed herein are useful in aqueous separation processes such as microfiltration, ultrafiltration, dialysis, osmosis, reverse osmosis, ion exchange, or electrodialysis, or a combination thereof.
- Applications include water treatment, cell culturing, artificial organs, and food processing.
- One aspect of the invention features a block copolymer used to make a porous polymer article having a plurality of active surface block segment moieties on the active surface of the article, wherein the active surface block segments are covalently bound to, and yet have properties different from, the interior anchor block segments of the article.
- the invention includes the porous polymer article, and a method of making it.
- each Q moiety is OAr 1 .
- One example is a block copolymer of formula (2):
- R (OCH 2 CH 2 ) n -X-Y (2)
- R is C 1-20 alkyl (e.g., C 6-20 alkyl), C 7-20 alkylaryl,
- n is between 20 and 500
- X is -Z 1 -(OAr 1 OAr 2 ) m - or -(OAr 2 OAr 1 ) m -.
- Y is hydroxy, -(OAr 3 O)-Z 2 -(CH 2 CH 2 O) p -R 1 , or -(OAr 4 O)-(CH 2 CH 2 O) p -R 1 .
- Z 1 is
- R 2 being C 1-12 alkyl or C 6-20 aryl.
- R 1 is C 1- 20 alkyl, C 7-20 alkylaryl, C 7-20 aryl-alkyl, or C 1- 20 perfluoroalkyl.
- each of R 1 and R is selected from lauryl, myristyl, palmityl, stearyl, cetyl, methyl, phenyl, octylphenyl, nonylphenyl, and perfluoroalkyl, or any of possible combinations thereof.
- R and R 1 are the same.
- Each of Ar 1 and Ar 3 is independently selected from 1,4-phenylene, 1,3-phenylene, naphthyl-1,4-diyl, naphthyl-1,5-diyl, 4,4'-biphenylene, diphenyl ether-4,4'-diyl, diphenylthioether-4,4'-diyl, diphenylisopropyl-idene-4,4'-diyl,
- alkylene is -(CH 2 ) -, q being 1, 3, 5, 7 or 9, p-terphenyl-4,4'-diyl, and bivalent radicals of binaphthalene, anthracene, and phenylnaph-thalene.
- Ar 1 is
- Ar 1 and Ar 3 are the same.
- Each of Ar 2 and Ar 4 is independently selected from diphenylsulfoxide-4,4'-diyl, diphenylsulfone-4,4'-diyl, diphenyl ketone-4,4'-diyl, and bivalent radicals of diphenyl-C 1-12 alkyl phosphine oxide and diphenyl-C 6-20 aryl phosphine oxide.
- Ar 2 is preferably diphenylsulfone-4,4'-diyl or diphenyl sulfoxide-4,4'-diyl.
- Ar 2 and Ar 4 are the same.
- the value of m is between 1 and 250 (e.g., between 1 and 200, or between 10 and 250).
- the value of p is between 20 and 500 (e.g., between 20 and 400, or between 20 and 300).
- Z 1 is -N(R 2 )-(S0 2 )-C 6 H 4 - and Z 2 is -C 6 H 4 -(SO 2 )-N(R 2 )-.
- the weight ratio of (OCH 2 CH 2 ):(OAr 1 OAr 2 ) + (OAr 2 OAr 1 ) is between 5:1 and 1:20, e.g., between 3:1 and 1:10.
- R is not H; or Y is not OH; or R is not H and Y is not OH.
- Another embodiment of the invention is a block copolymer of formula (1), wherein each of R and R 1 is H or C 1- 10 alkyl, n is between 40 and 8,000, X is -[OAr 2 Q] m , m is between 4 and 250, Y is -OAr 4 O-[BCH 2 O] p -R 1 , and p is between 40 and 8,000, provided that one Q moiety is
- This embodiment has the formula (3):
- 80% - 100% of E moieties is CH 2 in one Q, and 80% - 100% of A moieties and B moieties combined is CH 2 ; at least two Q moieties are [OCH 2 E] u ; each of R and R 1 is independently H or C 1-3 alkyl, e.g., independently H or methyl, or independently C 1-3 alkyl; 80% - 100% of Ar 2 and Ar 4 is diphenylsulfone-4,4'-diyl; or 80% - 100% of Ar 1 and Ar 3 is
- copolymer is polyethylene oxide homopolymer, or a
- polyethylene oxide-polypropylene oxide copolymer e.g., up to 20%, between 10% and 80%, between 10% and 60%, between 20% and 50%, or between 40 and 80%.
- central block segment -[OAr 2 Q] m - includes a polyethylene oxide block polymer or an ethylene oxide-propylene oxide block copolymer; or -[OAr 2 Q] m - includes a moiety having a molecular weight between 2,000 Da and 200,000 Da, this moiety being a polyethylene oxide block, an ethylene oxide-propylene oxide block copolymer, or a polyethylene oxide-polypropylene oxide-polyethylene oxide block copolymer.
- n, u and p are each independently between 1 and 10,000 (e.g., between 40 and 8,000, 40 (or 100 or 2,000) and 5,000; and m is between 1 and 500 (e.g., between 3 (or 4, 10, 25, or 50) and 500, 50 and 500, 125 and 200, and 100 and 250).
- n is between 100 and 5,000
- u is between 100 and 5,000
- p is between 100 and 5,000
- m is between 125 and 200.
- the number of PEO repeating units in a given block copolymer formed with a 10,000 g/mole PEO-diol is greater than the number of PEO repeating units in a block copolymer formed with a 35,000 g/mole PEO-diol.
- the run length of PSF repeating units will increase as the molecular wegiht of the PEO-diol
- the disclosed block copolymers (e.g., formulae (1) (2)) are first described generally by their relationship and function in a method of making porous polymer
- These articles are formed by a wet-casting method.
- a casting solution includes a block copolymer (e.g., A-b-B), a matrix-forming (or bulk) polymer (B'), and a solvent.
- diblock copolymer A-b-B includes a first block segment A which has a greater affinity for the coagulation medium than for the casting solution solvent, and a second block segment B which can mix with a desired matrix-forming polymer B'.
- the coagulation medium is a nonsolvent with respect to the matrix-forming polymer, i.e., the
- coagulation medium does not dissolve the matrix-forming polymer.
- B and B' have an affinity for each other that results in mixing. Mixing includes physical
- portions of the first block segment A (which has a greater affinity for the coagulation medium) localize at the interface between the coagulation medium and the casting solution solvent.
- the second block segment B mixes with the matrix-forming polymer B' to form a polymer matrix.
- portions of the first block segment are localized on the active surface of the polymer article, the first and second block segments remain covalently-bonded to each other.
- the first block segment is covalently bound to the second block segment which, in turn, is mixed with or anchored to the matrix-forming polymer.
- the resulting polymer article therefore, has an organized structure with portions of active surface segments present across the active surface of the article.
- Any portion or moiety of a block segment may be present on the active surface of an article; surface presentation of the entire segment is not required.
- a PEO block segment moiety could be 50%, 35%, or 10%, or more, or less, of the 200-monomer length. It is possible that a relatively long single block segment gives rise to two or more active surface segment moieties which are connected to each other by a portion of the block segment that is not presented at the
- the design of the disclosed block copolymers favors presentation of one active surface segment moiety per block segment.
- the active surface segment moieties are uniformly present across the active surface of the article. The uniformity (i.e., more or less even or average distribution of active surface segments) can be measured by methods such as X-ray photoelectron
- the density of active surface segment moieties is sufficient to impart macroscopically-detectable hydrophilicity to the active surface (e.g., low water contact angle).
- a plurality of active surface segments e.g., membrane-integrated or covalently-bonded hydrophilic moieties means two or more active surface segments to create an active surface with the desired property or properties, such as degree of hydrophilicity.
- block copolymers and membranes formed therefrom wherein the block copolymer is a diblock (A-b-B), a triblock (e.g., A-b-B-b-A or A-b-B-b-C, where C has a higher affinity for A and the coagulation medium, than for B and the casting solution solvent), a tetrablock, etc., or combinations thereof.
- A-b-B diblock
- a triblock e.g., A-b-B-b-A or A-b-B-b-C, where C has a higher affinity for A and the coagulation medium, than for B and the casting solution solvent
- a tetrablock etc., or combinations thereof.
- One of the disclosed casting solutions includes a poly(ethylene oxide)-b-polysulfone-b-poly(ethylene oxide) triblock copolymer,
- hydrophobic (matrix-forming) polysulfone polymer a hydrophobic (matrix-forming) polysulfone polymer, and N-methylpyrrolidinone as a casting solution solvent.
- This triblock copolymer includes (1) a hydrophobic polysulfone (PSF) block segment which is miscible with a hydrophobic matrix-forming polymer which may be the same (PSF) or different, between (2) two terminal hydrophilic poly(ethylene oxide) (PEO) block segments which have a greater affinity for an aqueous coagulation medium containing water than for N-methyl-pyrrolidinone, the casting solution solvent.
- PSF polysulfone
- PEO poly(ethylene oxide)
- hydrophilic active surface PEO moieties are presented at the active surface of the membrane, and yet the PEO block remains covalently bonded to the PSF membrane matrix.
- the active surface moiety (here, the PEO) is integral to the block copolymer, and is incorporated, via the casting solution, into the membrane during formation.
- the surface of a polymer article made according to the invention requires neither reactive functional groups nor subsequently derivatized functional groups to increase surface hydrophilicity.
- a polymer article containing one or more disclosed block copolymers has improved performance.
- addition of a PEO-b-PSF-b-PEO triblock copolymer improves performance by increasing water permeability and protein recovery (resistance to protein adsorption), and
- One of the unexpected results of the disclosed block copolymers is the superior performance of membranes made therefrom when treated with only a fraction (e.g., a 5% solution of glycerol) of the usual amount of surfactant or humectant.
- hydrophilic active surface segments are permanently integrated into the polymer during polymer formation. Therefore, no further functionalization or derivatization is required, which shortens the
- hydrophilic surface results from membrane-integrated active surface segments which are covalently-bonded to the polymer matrix during membrane formation.
- both the retentate and filtrate are therefore free of certain contaminants. Examples of contaminants include chemical residues associated with derivatized membranes
- One aspect of the invention relates to a block copolymer having the formula (1) or (2) as described in the Summary. According to the invention, a block copolymer having the formula (1) or (2) as described in the Summary. According to the invention, a block copolymer having the formula (1) or (2) as described in the Summary. According to the invention, a block copolymer having the formula (1) or (2) as described in the Summary. According to the invention, a block copolymer having the formula (1) or (2) as described in the Summary. According to the invention, a block
- an amphiphilic block copolymer generally includes two block segments with different properties.
- an amphiphilic block copolymer has a hydrophilic block segment and a hydrophobic block segment.
- Hydrophobic and hydrophilic are relative terms well-known to those in the art.
- Hydrophilic polymers include peptides,
- dialkylacrylamides and preferably poly(ethylene oxide) (PEO).
- PEO poly(ethylene oxide)
- PEO or PPO is commercially available in a variety of molecular weights.
- PEO or PEG is available in a range between 0.4 kD to 1,000 kD, such as 1.0 kD to 600 kD, or 4 kD to 200 kD.
- Block copolymers having lower molecular weight PEO segments generally decrease the glass transition
- Block copolymers having higher molecular weight PEO segments have a greater tendency to undergo phase separation, and are therefore suitable to prepare thermoplastic elastomers.
- Certain embodiments include PEO in the higher available molecular weight ranges.
- Hydrophobic polymers include
- polyalkylmethacrylates polyphenylene, and preferably polyacrylonitrile, polyvinyl halides, polyvinylidene halides, polyimide, polyamide, polyetherimide, and even more preferably poly(arylethers) such as homopolymers of -(OAr 1 OAr 2 )- (e.g., wherein Ar 2 is polyether sulfone or polysulfone) .
- Halides include bromides, iodides, and preferably fluorides and chlorides.
- the invention contemplates the use of combinations of diblock and triblock copolymers, and combinations of different block copolymers, e.g., nonylphenyl
- Examples of pairs of diblock and corresponding triblock copolymers include methyl
- perfluoroalkyl poly(oxyethylene)-b-poly(aryl ether sulfone)-b-poly(ethyleneoxy)perfluoroalkyl.
- X is -Z 1 -(OAr 1 OAr 2 ) m - or -(OAr 2 OAr 1 ) m -.
- An alternative expression for this block is -(OAr 2 i OAr 1 i ) i -, where i is 1 to m. Similar expressions exist for the other block segments involving n and p.
- the methods of making a block copolymer permit wide variation, such that Ar 1 and Ar 2 in one -(OAr 2 OAr 1 )- unit may differ from the Ar 1 and Ar 2 in another (e.g., adjacent) -(OAr 2 OAr 1 )- unit.
- each of Ar 1 1 and Ar 1 2 is independently selected.
- the term "each of Ar 1 and Ar 3 is independently selected from” is therefore understood to encompass such "mixed" blocks. However, it is preferred that 90% of the m units are the same.
- the overall weight percent of PEO could be limited by the need for sufficient polysulfone to precipitate the polymer product in water.
- the product can be isolated by other means, such as dialyzing the reaction mixture against water to remove smaller molecular weight impurities and evaporating the remaining water; by using solvent mixtures which are less polar than water to cause precipitation.
- the wt % of PEO can also be increased by decreasing, or even omitting, bisphenol A and linking the bifunctional segments by bischlorophenyl sulfone
- one or more internal segments of the block copolymer is PEO or
- PEO/propylene oxide copolymers Such internal segments are generally formed with the corresponding difunctional PEG, which is commercially available in a wide variety of intermediate and high molecular weights (kD) such as l, 2, 4, 8, 10, 12, 18.5, 35, 50, 80, and 100. Random difunctional PEO/propylene oxide copolymers, and diblock and triblock copolymers are commercially available in many molecular weights between 1,000 and 1,000,000 Da. Copolymers and block copolymers are preferably between 2,000 and 20,000 Da.
- Combinations of the above include: two or more different molecular weight PEGs (Example 9); random PEO/PPO (Example 10); and the same or different molecular weight PEGs (internal bifunctional) and PEOs (chain terminating) (see Examples 11 and 12).
- the total weight percent of polyethylene oxide block and polyethylene oxide- polypropylene oxide random copolymer is up to 80 weight percent (e.g., between 10 and 60 weight percent of the block copolymer, or between 10 and 40% weight percent).
- a given total weight percent of PEO block and PEO/PPO random copolymer such as 40 weight percent
- contributions of PEO block and PEO/PPO random copolymer respectively, include: 0% and 40%, 10% and 30%, 40% and 0%, 25% and 15%, 20% and 20%, and so on.
- other pairs or multi- component combinations from among PEO block, PPO block, PEO/PPO random copolymer, and PEO-PPO-PEO triblock can constitute up to 80 weight percent of a disclosed block copolymer.
- thermoplastic applications such film formation, melt- extrusion, and molding.
- Sampled block copolymers of formula (3) had melting points of 150°C - 170°C and degraded at temperatures around 300°C. Based on melting point, DSC, and glass transition measurements, the processing windows for block copolymers of formula (3) are appropriate for a range of applications, especially as interfacial adhesion agents, and as dispersion, or adsorption agents which improve mechanical strength.
- Block copolymers of formula (3) are particularly suitable for making thermoplastic polymeric articles.
- the method of making these articles includes (a) blending and/or co-melting a block copolymer of formula (3) with a compatible (e.g., high-melting) thermoplastic such as polysulfones, polyaryl ethers, polycarbonates, and nylons; or (b) co-mixing a block copolymer of formula (3) with an incompatible polymer or with an organic additive or inorganic such as titania or silica.
- a compatible thermoplastic such as polysulfones, polyaryl ethers, polycarbonates, and nylons
- co-mixing a block copolymer of formula (3) with an incompatible polymer or with an organic additive or inorganic such as titania or silica.
- the blended, co-melted, or co-mixed material is then injection molded, extruded, or thermo-formed under pressure, and cooled to a rigid or semi-rigid state.
- Polymers having 30 wt% (Example 11), 50 wt% (Example 12), and 80 wt% (not shown) have been made.
- Thermoplastic elastic properties can be easily measured by methods known to those in the art, such as melting point, DSC, and glass transition temperatures.
- the invention is based, in part, on the design of a block copolymer consistent with the principles
- poly(oxyethylene)-b-polysulfone-b-poly(ethyleneoxy) triblock copolymer by condensation.
- a PEO chain-terminating monomer is reacted with a reactive 4-fluorophenyl sulfonamide end group.
- the PEO chain terminating monomer is reacted with 4,4'-isopropylidene diphenol and bischlorophenyl sulfone.
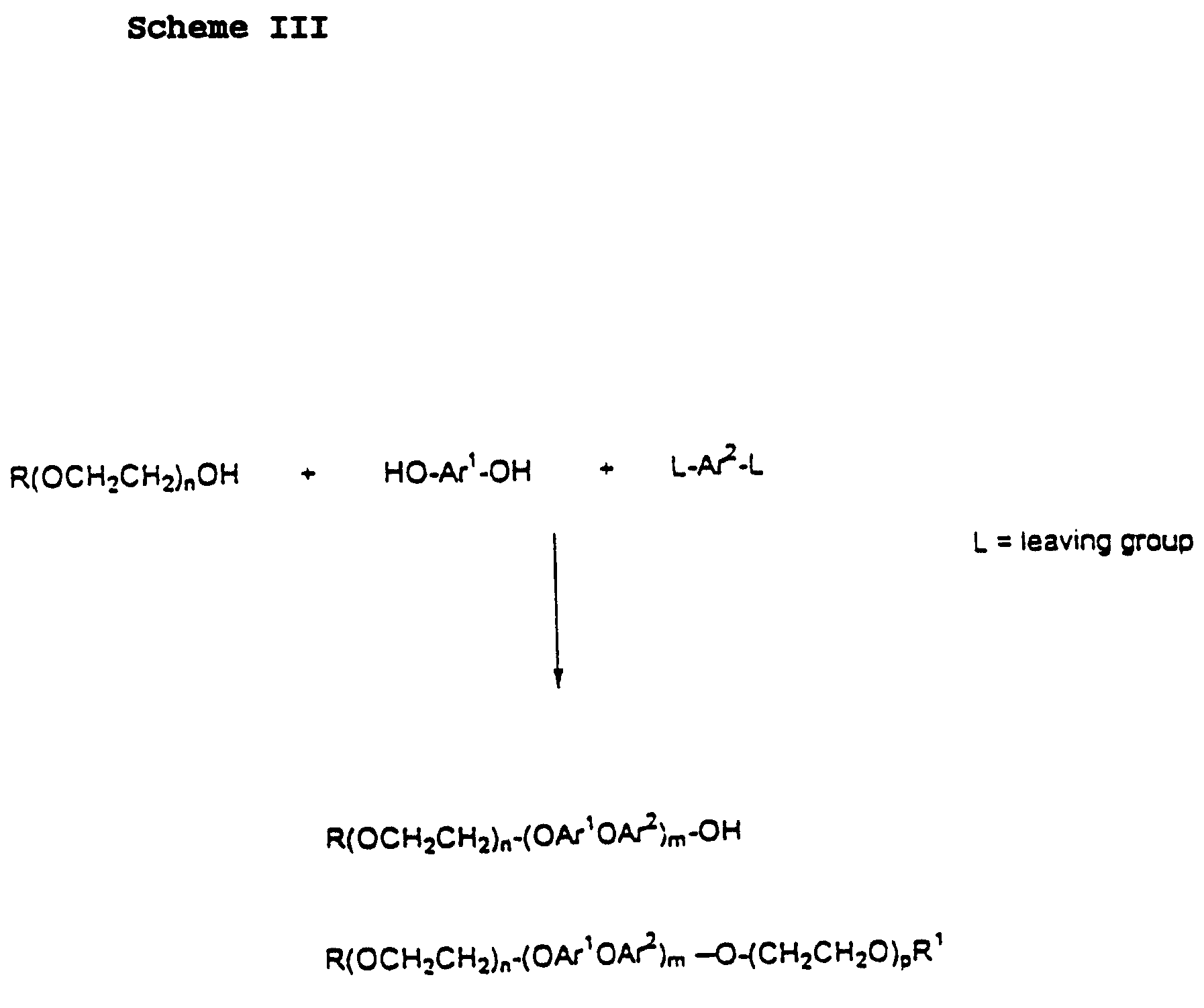
- Scheme III illustrates an alternative preparation, wherein a PEO-block alcohol, an aryl diol, and a reactive difunctionalized (e.g., dihalo, dinitro, or halo-nitro) aryl reagent (e.g., bischlorophenyl sulfone) are reacted to form a combination of diblock and triblock copolymers. Reacting the three reactants in stoichiometric
- reacting equal amounts of the difunctional reactants e.g., diol and sulfone
- some amount of endcapping reactant e.g., polyethyleneoxy or polyethylene glycol
- reacting equal amounts of the difunctional reactants e.g., diol and sulfone
- endcapping reactant e.g., polyethyleneoxy or polyethylene glycol
- One embodiment includes 10 - 80 weight percent; another includes 15 - 40 weight percent of PEO. The latter embodiment is
- porous polymer article with a covalently-bonded hydrophilic moiety present on its active surface as described in the summary section.
- the porous polymer article includes (i) a hydrophilic-hydrophobic diblock copolymer, a hydrophilic- hydrophobic-hydrophilic triblock copolymer, or a
- the polymer article has up to 80 weight percent of one or more block copolymers (e.g., in weight percents, between 0.01 and 80, between 5 and 50, and between 15 and 40) .
- the article includes a diblock copolymer (i.e., Y is hydroxy); in another embodiment, the article includes a triblock copolymer (Y is disclosed or undisclosed moiety that is not hydroxy).
- polymer articles e.g., flat sheet or hollow fiber membranes
- a disclosed block copolymer is adapted from methods known in the art, provided that the casting solution solvent, the coagulation bath medium, the temperature, and other conditions known and understood in the art are selected in a manner consistent with the invention as described above.
- One aspect of the invention relates to a method of making a porous polymer article with enhanced hydrophilicity including three steps as described in the Summary section.
- the invention is also based, in part, on the discovery that after a polymer article is formed, thermally annealing the article enhances the already-superior or desirable performance properties.
- the first step in preparing a polymer article is providing a casting solution.
- the disclosed block copolymers are used to prepare a casting solution which includes a block copolymer, a matrix-forming polymer, and a polar aprotic organic solvent.
- a casting solution includes up to 80% solids, i.e., the sum of block copolymer and matrix polymer is less than or equal to 80% (e.g., less than or equal to 50, 40%, or 35%; or between 5% and 25%).
- a person of ordinary skill in the art can easily determine whether, and how much, heating is necessary and appropriate to form an acceptably homogenous casting solution.
- a casting solution may include either a single block copolymer or a combination of two or more block copolymers in any desirable proportion.
- a casting solution includes between 0.1 and 50 weight percent
- a hydrophobic matrix-forming polymer is a rigid block polymer known in the art, or a nonblock polymer such as a homopolymer of bischlorophenyl sulfone.
- the weight percent of the hydrophobic polymer is 0.1 - 30 weight percent (e.g., 5 - 20 weight percent) of the casting solution.
- hydrophobic polymers include the group consisting of polyacrylonitrile, polyvinyl halide, polyvinylidene halide, polyimide, polyetherimide, and polyamide, and polymers of - (OAr 1 OAr 2 )-, or a copolymer thereof.
- the hydrophobic polymer is a polymer or copolymer of - (OAr 1 OAr 2 )-, wherein Ar 2 is, e.g., diphenylsulfone-4,4'- diyl.
- the casting solution includes between 40 and 95 weight percent (e.g., between 50 and 90 weight percent, or between 65 and 90 weight percent) of a solvent or solvent system.
- Suitable polar aprotic organic solvents include N-methylpyrrolidinone (NMP), dimethylformamide, dimethyl-acetamide, 4-butyrolactone, and a solvent system thereof.
- NMP N-methylpyrrolidinone
- dimethylformamide dimethyl-acetamide
- 4-butyrolactone 4-butyrolactone
- a solvent system thereof may include toluene or water, provided the solvent quality is
- the polar aprotic solvent is N-methyl pyrrolidone.
- other additives may be included, provided that the adjusted solvent quality is not so adversely affected that precipitation occurs.
- solvent quality-adjusting additives include a hydroxyl-containing reagent such as polyethylene glycol (commercially available in molecular weights such as 300, 400, 600, 900, 1000, 1,500, 2,000, 4,000, 8,000, 10,000, 12,000, and
- the casting solution may further include 5 - 30 weight percent (e . g. , 10 - 28 weight percent) of a hydroxyl-containing reagent selected from polyethylene glycol (e.g., molecular weight of 300, 400, or 600), water, and C 1-8 alkanols.
- a hydroxyl-containing reagent selected from polyethylene glycol (e.g., molecular weight of 300, 400, or 600), water, and C 1-8 alkanols.
- the block copolymer contains higher molecular weight blocks (e.g., high molecular weight PEG )
- higher molecular weight PEG or alkanols as a casting solution additives can be used without causing phase separation.
- Additives also include those additives well-known in the art such as small amounts of colorants. Coagulation temperatures range from 18 - 90°C (e.g.,
- Disclosed casting solutions include (in weight percents): 10 weight percent block copolymer, 10 weight percent polysulfone, and 80 weight percent polar aprotic solvent; 10 weight percent block copolymer, 15 weight percent polysulfone, 25 weight percent
- polyethylene glycol and 50 weight percent polar aprotic solvent; or 15 weight percent block copolymer, 5 weight percent polysulfone, and 80 weight percent polar aprotic solvent.
- Some casting solutions consist of disclosed block copolymers, polysulfone, NMP, and a soluble salt such as lithium chloride.
- a suitable nonsolvent coagulation bath medium based on the block copolymer, particularly the block segment to be presented on the active surface, the casting solution solvent, the matrix polymer, and the desired pore structure of the polymer article.
- the solvent and solvent additives should be soluble in the coagulation medium, and the hydrophilic matrix polymer should precipitate in the coagulation medium.
- coagulation media include of NMP and water in combinations such as 0% NMP-100% water, 50% NMP-50% water, 70% NMP- water, and 80% NMP-20% water.
- a casting solution was spread onto sheets of nonwoven polypropylene fabric taped to glass plate supports and immersed in a coagulation medium of reverse osmosis- processed water at room temperature.
- the membrane including the integrated support sheet was placed in a second water bath to remove residual solvent (NMP) and pore former (e.g., NMP) and pore former (e.g., NMP) and pore former (e.g., NMP) and pore former (e.g., NMP) and pore former (e.g., NMP) and pore former (e.g., NMP) and pore former (e.g., NMP) and pore former (e.g., NMP) and pore former (e.g., NMP)
- the rinse water was
- the membranes were placed in 70% v/v glycerol solution for 24 hours, placed between two sheets of blotting paper, dried at room temperature, and stored in air-tight bags.
- thermal annealing is not necessary to produce a porous, polymer article which includes a block copolymer of the invention, such treatment is preferable, since it was found to enhance the hydrophilic or wetting properties of polymer articles of the invention to an unexpected degree in an unexpected way.
- autoclaving increases the membrane molecular weight cut-off.
- membranes of the invention were thermally annealed, the molecular weight cut-off was found to stay the same or even decrease. For example, it is believed that in membranes containing PEO, thermal annealing in the presence of water enhances the degree of PEO presentation, and the overall distribution of PEO active surface segments.
- thermal annealing generally occurs in the presence of water (e.g., a water bath or an autoclave).
- the thermal annealing step includes heating the article in an
- the thermal annealing step includes heating the article at a temperature between 40 and 90°C (e.g., between 45 and 75°C) .
- the annealing temperature and annealing time generally have an inverse relationship.
- thermal annealing included heating the article at about 120°C for approximately 30 minutes.
- thermal annealing was performed at about 50°C for
- Polymer articles were characterized by several methods known to those in the art. Such methods include characterization by Fourier-transformed infrared
- FTIR spectroscopy
- solid state proton and 13 C NMR with a paramagnetic relaxation agent such as Cr(acac) 3 to determine organization of the block copolymer at the membrane interface
- SEM scanning electron microscopy
- thermoplastic e.g., extruded, molded, or moldable
- polymer article which has been molded, extruded, or otherwise formed from a composition which includes (i) one or more block copolymers of formula (1) or (3), and (ii) an inorganic additive such as titania or silica; a thermoplastic polymer, such as a polyaryl ether, a polycarbonate, or a nylon; or other organic additives; or a combination thereof.
- the thermoplastic article has at least one surface capable of phase-separation, surface adhesion, or interfacial interaction as a result of the block copolymer of formula (1) or (3).
- the invention therefore features a polymeric article formed by either the direct extrusion or the injection molding of a composition containing a block copolymer of formula (3), e.g., wherein the composition further contains an inorganic additive such as titania or silica; a thermoplastic polymer, such as a polyaryl ether,
- thermoplastic films and molded forms can be formed with block copolymers having generally higher molecular weight percents of PEO relative to the cast polymers.
- Solvents include polar aprotic solvents and other organic solvents.
- hydrophilicity and lubricity and therefore the disclosed block copolymers can act as internal mold release agents.
- Hydrophilicity is desirable for biocompatibility or hemocompatibility, including improved lubricity for manufacturing, demonstrated by ease of release of an injection molded article from the mold.
- Lubricity or hydrophilicity are desirable for biomedical devices, particularly invasive or insertable devices such as urogenital and ocular devices, such as contact lenses, catheters, and other prosthetic devices.
- hydrophilic packaging films are more resistant to
- a 50% PEO/50% PSF block copolymer (5 kD) was synthesized as follows. Reflux apparatus was purged with argon. Methyl poly(oxyethylene) with a terminal hydroxyl (200 g, 0.04 moles), bisphenyl disopropylidene-4,4'-diol (Bisphenol A, 114.145 g, 0.5 moles), and bischlorophenylsulfone (149.328 g, 0.52 mole) were added to 600 mL N-methylpyrrolidinone and 150 mL toluene and potassium carbonate (143.5 g, 1.04 moles). The reaction mixture was slowly heated over 0.5 hours until an azeotrope reflux was reached (about 160°C).
- polyethylene glycol M.W. 400
- 75.0 weight percent N-methyl pyrrolidinone was prepared.
- the solution was prepared in a glass jar and placed on a roll mixer for 24 - 48 hours until a single phase solution was obtained.
- Flat sheet membranes were prepared by casting a 0.015 inch coating of the casting solution onto a glass plate or a nonwoven polypropylene support (Freudenberg). The coated support was immersed in a coagulation water bath until precipitation. After additional extraction with water, the membranes were immersed in a glycerol/water solution for a minimum of 1 hour. One group of membranes were dried in ambient conditions.
- a second group of membranes after drying in ambient conditions, were immersed in a water bath and thermally annealed in an autoclave at 121°C for 30 minutes and allowed to dry at ambient conditions.
- a third group of membranes was simply removed from the extraction bath and allowed to dry at ambient conditions. The resulting membranes were cut into discs having a 47 mm diameter and evaluated by filtering 20 ml of a 0.1% Cytochrome-C solution
- a casting solution containing 10.0 weight percent PEO-PSF-PEO (a PE0:PSF::30:70 block copolymer), 10.0 weight percent PSF, 5.0 weight percent polyethylene glycol, and 75.0 weight percent N-methyl pyrrolidinone was prepared.
- Membranes were prepared according to Example 2 except the above casting solution was used.
- the block copolymer membranes of Example 3 were also tested for water permeability and filtration. After extraction with water, the membranes were thermally annealed by heating in a water bath at 121°C for 30 minutes in an autoclave. The membranes were then dried at room temperature, and compared with two polysulfone membranes made of 15% PSF, 10% PEG MW 400 and 75% NMP; one dried with glycerol (PSF 1) and one dried without glycerol or surfactant (PSF 2) . The data indicate that the disclosed membranes, even when dry, spontaneously wetted with water (see Table I) . In contrast, to impart wettability to the conventional polysulfone membranes, treatment with a surfactant was required (e.g.
- a casting solution containing 15.0 weight percent PEO-PSF-PEO (a PEO:PSF::30:70 block copolymer), 5.0 weight percent of PSF, 80.0 weight percent of N-methyl pyrrolidinone was prepared.
- Membranes were prepared according to Example 2 except that the above casting solution was used and the mixture was coagulated in a 3 : 1: :NMP:water bath. After coagulation, the membranes were placed in a secondary extraction bath of pure water for a minimum of 12 hours and dried under ambient
- the NMR results suggest the organization of the block copolymer at the membrane surface.
- the distinct relaxation times for PEO and PSF indicate two distinct physical domains within the membrane.
- the relaxation associated with PEO was suppressed while the relaxation associated with PSF was unaffected. This is consistent with PEO being localized in domains accessible to the Cr(acac) 3 solution at the membrane surface, and PSF being localized in the interior membrane matrix.
- a casting solution for hollow fiber membranes containing 10.0 weight percent PEO-PSF-PEO (a PEO:PSF::30:70 block copolymer), 15.0 weight percent PSF, 25.0 weight percent polyethylene glycol (M n 600) and 50.0 weight percent N-methyl pyrrolidinone was prepared.
- Hollow fibers were formed from the casting solution by spinning the solution at 55°C at 100 feet per minute with nominal inner diameter of 200 ⁇ m and nominal outer diameter of 300 ⁇ m.
- the nominal wall thickness was 50 ⁇ m.
- the core solution was either pure water or a mixture of NMP and water. After collection on the take-up wheel, the fiber was extracted repeatedly in water. Post-treatment included soaking in various glycerol/water solutions ranging from 0 to 80% glycerol/water by weight, or thermally annealing the fiber in water for about 30 minutes at 120°C (autoclave). The fibers were then air dried at room temperature overnight.
- Membranes obtained in Example 5 were characterized via SEM and were further characterized by measurement of the pre-foul NaCl and Vitamin B-12 permeability and the bovine serum ultrafiltration coefficient, K uf .
- the wettability of fibers made from casting solutions with and without triblock copolymer present is in Table III. Spinning conditions for the casting solution without the triblock copolymer were identical to those given above. The performance of the fiber soaked in the 80% glycerin solution represents the maximum obtainable for a fully-wetted membrane in both cases. For membranes made from casting solution containing only PSF and PEG 600, the wettability is lost or the performance is reduced at all other glycerin concentrations tested.
- the fiber collapsed when dried overnight. Performance therefore could not be measured.
- the fiber made from casting solution containing PSF, PEG 600 and the PEO-PSF-PEO triblock copolymer maintained wettability or performance for all glycerin concentrations, including 5%.
- the autoclaved fiber containing no wetting agent also maintained wettability. Only the fiber that was air dried overnight with no wetting agent or
- Membranes from Example 5 are contrasted to a PSF membrane made of 22% polysulfone, PEG 600 and 48% NMP (see Table III).
- the membrane of the present invention retains remarkable performance when treated with a low
- a survey of casting solution compositions incorporating PEO-b-PSF and polysulfone (Udel 3500) was made (see Table V).
- N-methyl pyrrolidinone was used as the solvent for all the formulations and a coagulation bath consisting of H 2 O at 50°C.
- PEO-b-PSF can be used to formulate membranes whose molecular weight cutoff ranges from > 2,000 kD (i.e. 5% PMX30-5B13/5% PSF) to 20 kD (i.e. 10% PMX30-5B13/15% PSF).
- An asterisk (*) indicates that the membrane was tested at 55 psi, and the reported flux was normalized to 10 psi.
- BLDex Blue Dextran
- Mn ⁇ 2,000 kD
- BSA Bovine Serum Albumin
- Mn 67 kD
- Cyt-C Cytochrome C
- Mn 12.4 kD.
- Solvent (NMP) formed the balance of each formulation. All were cast (8 mil) on nonwoven polypropylene fabric (Freudenberg) and
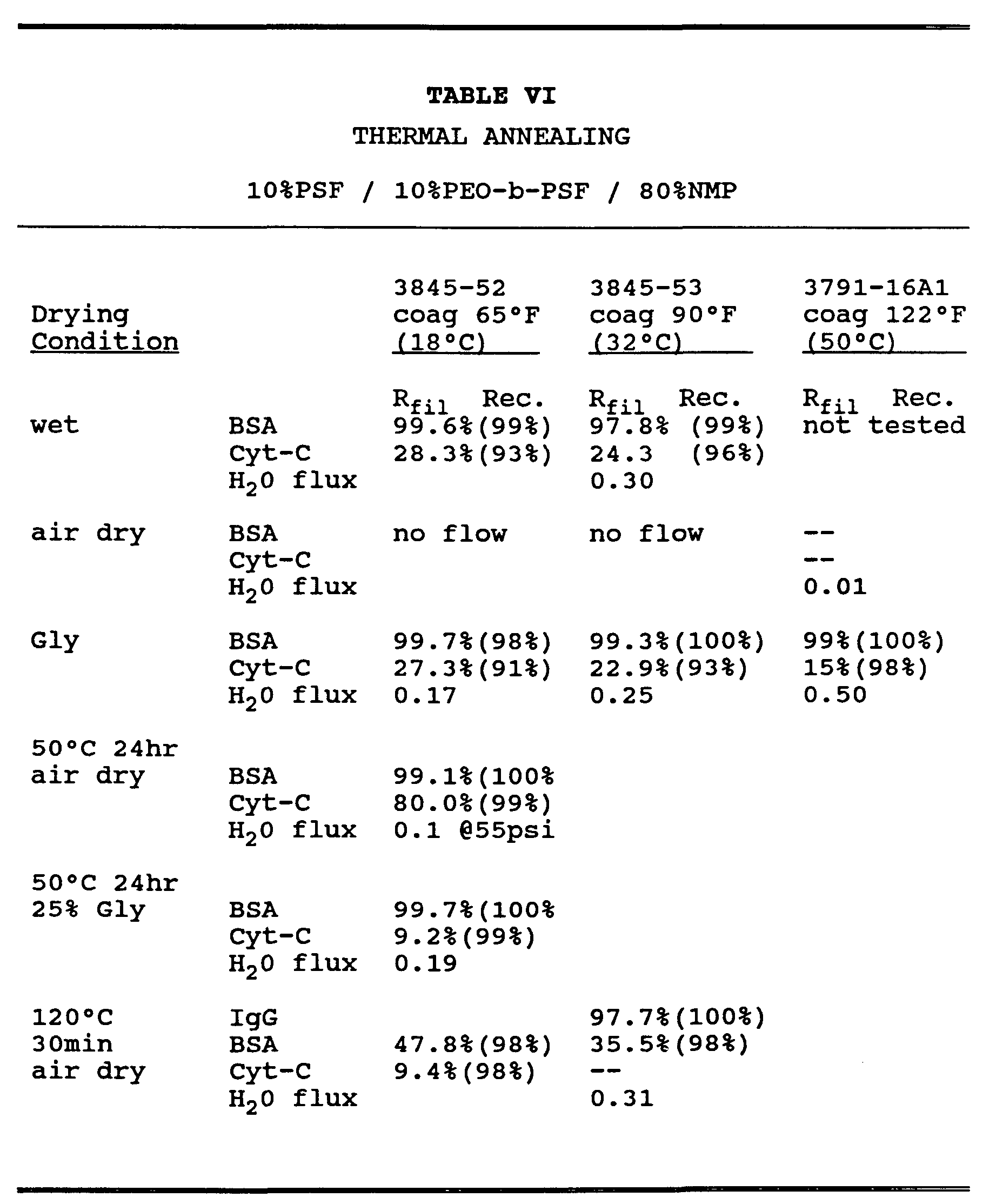
- Membranes were also thermally treated after fabrication to improve the PEO surface enrichment (column 1, membrane 3845-52). Membranes which were placed in a water bath at 50°C for 24 hour, and then either air dried or dried from glycerin had a markedly improved Cyt-C recovery compared to the membrane which received no thermal post- treatment. This information supports the enhanced surface enrichment of PEO after thermal
- Reflectance spectroscopy provided a complementary measure of Cyt-C adsorption to the membrane (see Table VII). Increasing spectral absorbance indicated
- Example 9 Four additional block polymers were prepared as Example 9, substituting for polyethylene glycol of average molecular weight 10,000 Da, polyethylene glycols of 4000 Da, 8000 Da, 12000 Da, and 18500 Da,
- a polymer was prepared as in Example 9, with the following stoichiometry.
- a weight fraction of 40% of a random two-component PEO/PPO (5:1 ratio, MW 8750, Polyscience, Warrington, PA) was used.
- the two-component PEO/PPO (0.006 mole) was reacted with 0.2754 mole
- a polymer was prepared as in Example 9, with the following stoichiometry.
- a polymer was prepared as in Example 9, with the following stoichiometry.
- a tetrablock or pentablock copolymer including poly(ethyleneoxy) and a hydrophobic block segment such as but not limited to those described herein is also within the scope of the claims.
- a non-porous dense film may be made with the recited block copolymers.
- poly (ethyleneoxy) may be replaced by a
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- Polymers & Plastics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Dermatology (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Transplantation (AREA)
- Polyethers (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/US1996/012421 WO1997005190A1 (en) | 1995-07-27 | 1996-07-29 | Block copolymers |
| AU66825/96A AU6682596A (en) | 1995-07-27 | 1996-07-29 | Block copolymers |
| JP50783697A JP4067564B2 (en) | 1995-07-27 | 1996-07-29 | Block copolymer |
| EP96926798A EP0840759A4 (en) | 1995-07-27 | 1996-07-29 | Block copolymers |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/508,172 | 1995-07-27 | ||
| PCT/US1996/012421 WO1997005190A1 (en) | 1995-07-27 | 1996-07-29 | Block copolymers |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1997005190A1 true WO1997005190A1 (en) | 1997-02-13 |
Family
ID=22255525
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1996/012421 WO1997005190A1 (en) | 1995-07-27 | 1996-07-29 | Block copolymers |
Country Status (2)
| Country | Link |
|---|---|
| AU (1) | AU6682596A (en) |
| WO (1) | WO1997005190A1 (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007020010A1 (en) * | 2005-08-12 | 2007-02-22 | Novartis Ag | Method for making contact lenses |
| EP2554564A1 (en) * | 2011-08-05 | 2013-02-06 | Basf Se | Process for preparing blockcopolymers |
| WO2014195234A1 (en) * | 2013-06-03 | 2014-12-11 | Basf Se | Improved membranes |
| WO2014177638A3 (en) * | 2013-05-02 | 2014-12-24 | Basf Se | Block copolymers |
| US8952109B2 (en) | 2011-08-05 | 2015-02-10 | Basf Se | Process for preparing a block copolymer |
| WO2015132173A1 (en) * | 2014-03-06 | 2015-09-11 | Basf Se | New copolymers suitable for making membranes |
| WO2016023765A1 (en) * | 2014-08-12 | 2016-02-18 | Basf Se | Process for making membranes |
| US9623614B2 (en) | 2010-11-10 | 2017-04-18 | Novartis Ag | Method for making contact lenses |
| US9758634B2 (en) | 2013-05-02 | 2017-09-12 | Basf Se | Polyarylethersulfone copolymers |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4812269A (en) * | 1985-03-07 | 1989-03-14 | Gambro Dialysatoren Kg | Process for producing hollow fiber semi-permeable membranes |
| US5006256A (en) * | 1988-01-14 | 1991-04-09 | The Standard Oil Company | Affinity membranes having pendant hydroxy groups and processes for the preparation and use thereof |
-
1996
- 1996-07-29 AU AU66825/96A patent/AU6682596A/en not_active Abandoned
- 1996-07-29 WO PCT/US1996/012421 patent/WO1997005190A1/en not_active Application Discontinuation
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4812269A (en) * | 1985-03-07 | 1989-03-14 | Gambro Dialysatoren Kg | Process for producing hollow fiber semi-permeable membranes |
| US5006256A (en) * | 1988-01-14 | 1991-04-09 | The Standard Oil Company | Affinity membranes having pendant hydroxy groups and processes for the preparation and use thereof |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007020010A1 (en) * | 2005-08-12 | 2007-02-22 | Novartis Ag | Method for making contact lenses |
| US9623614B2 (en) | 2010-11-10 | 2017-04-18 | Novartis Ag | Method for making contact lenses |
| US8952109B2 (en) | 2011-08-05 | 2015-02-10 | Basf Se | Process for preparing a block copolymer |
| EP2554564A1 (en) * | 2011-08-05 | 2013-02-06 | Basf Se | Process for preparing blockcopolymers |
| WO2013020871A1 (en) * | 2011-08-05 | 2013-02-14 | Basf Se | Method for producing a block copolymer |
| CN105377952A (en) * | 2013-05-02 | 2016-03-02 | 巴斯夫欧洲公司 | Block copolymers |
| US10184040B2 (en) | 2013-05-02 | 2019-01-22 | Basf Se | Polyarylethersulfone copolymers |
| US10144807B2 (en) | 2013-05-02 | 2018-12-04 | Basf Se | Block copolymers |
| WO2014177638A3 (en) * | 2013-05-02 | 2014-12-24 | Basf Se | Block copolymers |
| US9758634B2 (en) | 2013-05-02 | 2017-09-12 | Basf Se | Polyarylethersulfone copolymers |
| JP2016522849A (en) * | 2013-05-02 | 2016-08-04 | ビーエーエスエフ ソシエタス・ヨーロピアBasf Se | Block copolymer |
| WO2014195234A1 (en) * | 2013-06-03 | 2014-12-11 | Basf Se | Improved membranes |
| CN105451867A (en) * | 2013-06-03 | 2016-03-30 | 巴斯夫欧洲公司 | Improved membranes |
| US10058823B2 (en) | 2013-06-03 | 2018-08-28 | Basf Se | Membranes |
| US10759908B2 (en) | 2013-06-03 | 2020-09-01 | Basf Se | Polymers for membranes |
| WO2015132173A1 (en) * | 2014-03-06 | 2015-09-11 | Basf Se | New copolymers suitable for making membranes |
| US10189943B2 (en) | 2014-03-06 | 2019-01-29 | Basf Se | Copolymers suitable for making membranes |
| WO2016023765A1 (en) * | 2014-08-12 | 2016-02-18 | Basf Se | Process for making membranes |
| US10441925B2 (en) | 2014-08-12 | 2019-10-15 | Basf Se | Process for making membranes |
Also Published As
| Publication number | Publication date |
|---|---|
| AU6682596A (en) | 1997-02-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0840759A1 (en) | Block copolymers | |
| JP4067564B2 (en) | Block copolymer | |
| Rana et al. | Novel hydrophilic surface modifying macromolecules for polymeric membranes: Polyurethane ends capped by hydroxy group | |
| KR100525604B1 (en) | Application of sulfone, ketone and ester containing polyalkyl ether units to medical materials | |
| CA2306538C (en) | Medical material containing fluorinated polysulfone having excellent antithrombotic activity | |
| EP1141059B1 (en) | Highly branched block copolymers | |
| US5798437A (en) | Thermoplastic block copolymers | |
| JP2017196613A (en) | Dialysis membrane and manufacturing method thereof | |
| JP7100017B2 (en) | Compositions comprising aromatic and fluorinated polymers, and their use | |
| KR0159772B1 (en) | Semipermeable membrane made from a homogeneously miscible polymer blend | |
| US11499016B2 (en) | Polysulfone-urethane copolymer, membranes and products incorporating same, and methods for making and using same | |
| JP2017125204A (en) | Hydrophilic block copolymers and membranes prepared therefrom (i) | |
| JP2023166376A (en) | Purification methods comprising use of membranes obtained from bio-based sulfone polymers | |
| WO1997005190A1 (en) | Block copolymers | |
| Ren et al. | mPEG-b-PES-b-mPEG-based candidate hemodialysis membrane with enhanced performance in sieving, flux, and hemocompatibility | |
| Huang et al. | Preparation of antifouling ultrafiltration membranes from copolymers of polysulfone and zwitterionic poly (arylene ether sulfone) s | |
| KR20200034759A (en) | Hydrophilic copolymers and membranes | |
| Dai et al. | Acrylonitrile-based copolymer membranes containing reactive groups: Fabrication dual-layer biomimetic membranes by the immobilization of biomacromolecules | |
| JP3948993B2 (en) | Polysulfone copolymer | |
| CN114173915B (en) | Film and polymer for producing same | |
| JP3872173B2 (en) | POLYHYDROXY ETHER RESIN AND METHOD FOR SYNTHESIZING THE SAME, POLYHYDROXY ETHER MEMBRANE AND METHOD FOR PRODUCING THE SAME | |
| Sun et al. | Low‐fouling polysulfone ultrafiltration membranes with amphiphilic sulfobetaine polyamide as additive | |
| JPS62168503A (en) | Separation membrane | |
| JP6137562B6 (en) | Hydrophilic block copolymers and membranes produced therefrom (I) | |
| US20200165392A1 (en) | Aromatic sulfone polymers comprising (per)fluoropolyether segments |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AL AM AT AU AZ BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GE HU IL IS JP KE KG KP KR KZ LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK TJ TM TR TT UA UG UZ VN AM AZ BY KG KZ MD RU TJ TM |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): KE LS MW SD SZ UG AT BE CH DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| ENP | Entry into the national phase |
Ref document number: 2227998 Country of ref document: CA Ref country code: CA Ref document number: 2227998 Kind code of ref document: A Format of ref document f/p: F |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1996926798 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1996926798 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1996926798 Country of ref document: EP |