WO1987006934A1 - Nitric esters of benzothiazepinones - Google Patents
Nitric esters of benzothiazepinones Download PDFInfo
- Publication number
- WO1987006934A1 WO1987006934A1 PCT/CH1987/000053 CH8700053W WO8706934A1 WO 1987006934 A1 WO1987006934 A1 WO 1987006934A1 CH 8700053 W CH8700053 W CH 8700053W WO 8706934 A1 WO8706934 A1 WO 8706934A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- benzothiazepine
- dimethylamino
- ethyl
- nitric
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D281/00—Heterocyclic compounds containing rings of more than six members having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D281/02—Seven-membered rings
- C07D281/04—Seven-membered rings having the hetero atoms in positions 1 and 4
- C07D281/08—Seven-membered rings having the hetero atoms in positions 1 and 4 condensed with carbocyclic rings or ring systems
- C07D281/10—Seven-membered rings having the hetero atoms in positions 1 and 4 condensed with carbocyclic rings or ring systems condensed with one six-membered ring
Abstract
Nitric esters of benzothiazepines useful in cardiovascular therapy, and their synthesis methods.
Description
Esters nitriques de benzothiazepinones . Benzothiazepinone nitric esters.
Les esters nitriques de l ' invention possèdent des propriétés vasodilatatrices coronariennes utiles pour le traitement de l'angor de différents types , soul agement de l a douleur et amél ioration de l' irrigation sanguine du tissu cardiaque ischémié.The nitric esters of the invention have coronary vasodilatory properties useful for the treatment of angina of various types, relieving pain and improving the blood supply to ischemic heart tissue.
Les dérivés oxy-acyles des benzothiazepines actuellement util isés corme vasodilatateurs, comme l e dut iazem, s'hydrolysent relativement aisément dans le tractus digestif et par métabol isation hépatique, si bien que l a biodisponibilité, par ex. du 2-(4-méthoxyphényl )-3-oxy-acétyl -5(2-diméthylamino-éthyl )-2 ,3 dihydro-1 ,5 benzothiazépine-4(5H)-one n'est que de 10% environ.The oxy-acyl derivatives of benzothiazepines currently in use as vasodilators, such as iazem, hydrolyze relatively easily in the digestive tract and by hepatic metabolism, so that bioavailability, e.g. 2- (4-methoxyphenyl) -3-oxy-acetyl -5 (2-dimethylamino-ethyl) -2, 3 dihydro-1,5 benzothiazepine-4 (5H) -one is only about 10%.
La propriété basique de cette benzothiazépine bien que relativement faible, n' autorise toutefoi s pas une bonne pénétration intra-épidermique tel que de nouvelles préparations galéniques l ' autoriseraient. Les dérivés oxy-nitro de l ' invention tel que: le 2-(4-méthoxyphényl )-3- oxy-nitro-5(2-diméthylamino-éthyl ) 2,3 dihydro-1 ,5 benzothiazépine-4(5H)- one possèdent une grande stabilité dans les mil ieux biologiques et une bonne diffusion à travers l 'épiderme par laquell e ils atteignent l a circulation sanguine de façon prolonguée. Ils peuvent être utilisés par les techniques d'administration trans-épidermiques.The basic property of this benzothiazepine, although relatively weak, does not, however, allow good intraepidermal penetration as new galenic preparations would allow. The oxy-nitro derivatives of the invention such as: 2- (4-methoxyphenyl) -3-oxy-nitro-5 (2-dimethylamino-ethyl) 2,3 dihydro-1,5 benzothiazepine-4 (5H) - they have great stability in biological media and good diffusion through the epidermis through which they reach the bloodstream for a prolonged period. They can be used by trans-epidermal administration techniques.
Exemple :Example:
1.- Cis(+)-3-(4méthoxyphényl)-3-oxy-nitro-5(2-diméthylamino-éthyl)-2,3 dihydro-1,5benzothiazépine-4(5H)-one.1.- Cis (+) - 3- (4methoxyphenyl) -3-oxy-nitro-5 (2-dimethylamino-ethyl) -2,3 dihydro-1,5benzothiazepine-4 (5H) -one.
Au mélange de 2,74 g, d'anhydride acétique et de 1,77 g. de NO3H à 100%, refroidi à -5 -0° (préparé par addition gouttes à gouttes de l'acide nitrique dans l'anhydride acétique refroidi à -5º) on ajoute en plusieurs
portions 3,72 g. de 2-(4-méthoxyohényl)-3-oxy-5(2-di.méthylamino-éthyl)-2,3 dihydro-1,5 benzothiazénine-4(5H)-one.To the mixture of 2.74 g, acetic anhydride and 1.77 g. 100% NO 3 H, cooled to -5 -0 ° (prepared by adding dropwise drops of nitric acid in acetic anhydride cooled to -5º) added in several servings 3.72 g. of 2- (4-methoxyohényl) -3-oxy-5 (2-di . methylamino-ethyl) -2,3 dihydro-1,5 benzothiazenine-4 (5H) -one.
Anrès 60 minutes, on retire le milieu de réaction du mélange glace-sel et la réaction se noursuit sous agitation magnétique jusqu'à ce que le milieu devienne homogène.After 60 minutes, the reaction medium is removed from the ice-salt mixture and the reaction continues with magnetic stirring until the medium becomes homogeneous.
La solution dans 50 ml de chlorure de méthylène du milieu est extraite 2 fois par 100 ml de CO3HNa à 5% puis 2 fois par 100 ml d'eau. Le résidu d'évaporation de la solution organique est repris par de l'acétate d'éthyle. Cette solution est extraite 2 fois par 150 ml d'eau. La solution organique est séchée sur du sulfate de magnésium et le solvant évaporé sous pression réduite.The solution in 50 ml of methylene chloride from the medium is extracted twice with 100 ml of CO 3 HNa at 5% and then twice with 100 ml of water. The evaporation residue from the organic solution is taken up in ethyl acetate. This solution is extracted twice with 150 ml of water. The organic solution is dried over magnesium sulfate and the solvent evaporated under reduced pressure.
Le résidu amorphe est cristallisé dans de l'eau, F. 76-77°, recrist. dans l'éther à -18°, F. 86-87°, recristallisation dans EtOH à -18°, F.86-87°.The amorphous residue is crystallized from water, mp 76-77 °, recrystallized. in ether at -18 °, F. 86-87 °, recrystallization in EtOH at -18 °, F.86-87 °.
ccm silice G-60 F 254: Rf Solvantsccm silica G-60 F 254 : Rf Solvents
0,85 n-butanol-H-O/ac. acétiπue0.85 n-butanol-H-O / ac. acétiπue
(5,1 2,5 vol.)(5.1 2.5 vol.)
0,70 acétate d'éthyle0.70 ethyl acetate
0,50 acétate d'éthyle/n-hentane0.50 ethyl acetate / n-hentane
(3,1 vol.)(3.1 vol.)
0,18 acétate d'éthyle/n-heptane0.18 ethyl acetate / n-heptane
(1,1 vol.)(1.1 vol.)
a) 1R (nujol) 1670 (F), 1640 (F), 1610 (m), 1560 (f), 1525 (F), 1400 (f), 1280 (F), 1180 (F), 1090 (m), 1040 (m), 1020 (F), 850 (F), 765 (F), 727 (f) cm-1 a) 1R (nujol) 1670 (F), 1640 (F), 1610 (m), 1560 (f), 1525 (F), 1400 (f), 1280 (F), 1180 (F), 1090 (m) , 1040 (m), 1020 (F), 850 (F), 765 (F), 727 (f) cm -1
intensité : F = forte f = faible m = moyenne b)intensity: F = strong f = weak m = medium b)
Claims
RevendicationsClaims
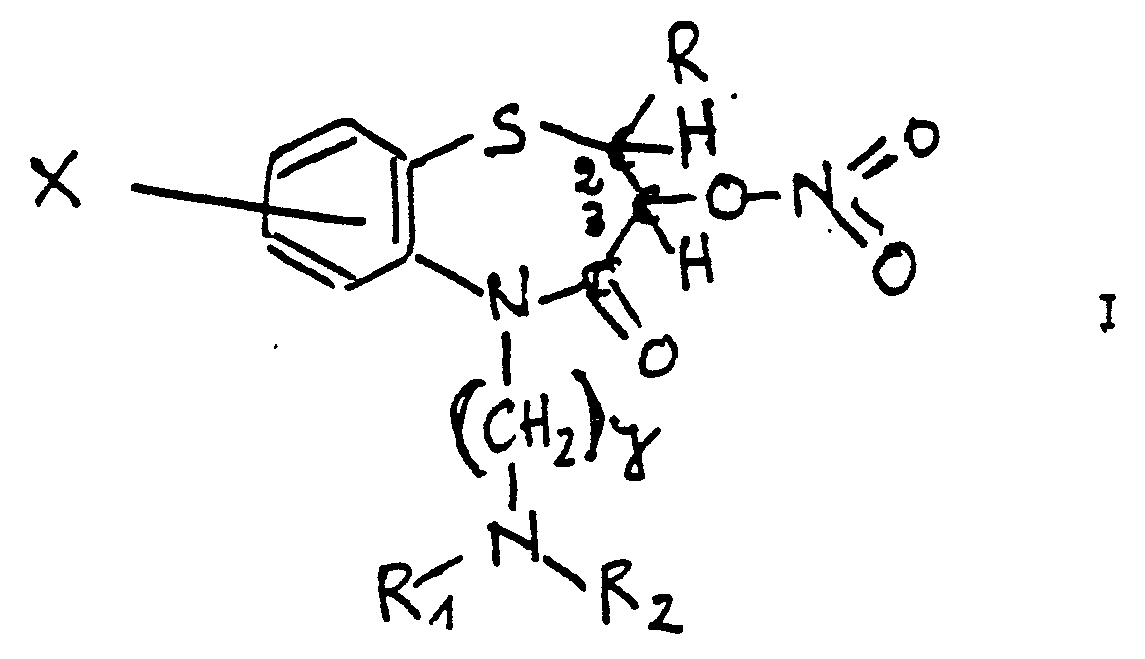
1.- Esters nitrinues thérapeutinuement actifs de formule I1.- Therapeutically active nitrinue esters of formula I
dans laquelle, R représente un reste phényl non substitué ou substitué par un groune alkyl de 1 à 3 atomes ou par plusieurs groupes alkyl inférieurs ou par un grounement alkoxy ou par plusieurs grouoes alkoxy ou par un atome halogène ou plusieurs atomes d'halogènes dans laquelle R1 et R2 sont des groupes alkyl inférieurs, Y est un groune alkylène de 2 à 3 atomes de carbones, X est un atome hydrogène ou un atome halogène. in which, R represents a phenyl residue which is unsubstituted or substituted by an alkyl group of 1 to 3 atoms or by several lower alkyl groups or by an alkoxy group or by several alkoxy groups or by a halogen atom or several halogen atoms in which R 1 and R 2 are lower alkyl groups, Y is an alkylene group of 2 to 3 carbon atoms, X is a hydrogen atom or a halogen atom.
La configuration des carbones 2 et 3 peut être 2S,3R 2R,3S 2S,3S ou 2R,3R ou les racémates.The configuration of carbons 2 and 3 can be 2S, 3R 2R, 3S 2S, 3S or 2R, 3R or the racemates.
La propriété bien que faiblement basique de ces composés permet la formation de sels d'addition d'acides pharmaceutiquement acceptables.The property, although weakly basic, of these compounds allows the formation of pharmaceutically acceptable acid addition salts.
Les actions thérapeutioues des dérivés de benzothiazepinesde la formule I sont particulièrement notables dans le domaine de la vasodilation coronarienne ou en qualité d'anti-dépressant, ceci particulièrement pour les dérivés suivants :The therapeutic actions of the benzothiazepine derivatives of formula I are particularly notable in the field of coronary vasodilation or as an anti-depressant, this particularly for the following derivatives:
a) 2-(4-méthylphényl)-3 oxy nitro-5(2-diméthylamino-éthyl)-2,3 dihydro1,5 benzothiazénine-4(5H)-one,
b) 2-(4-méthoyphényl)-3-oxynitro-5-(2-diméthylamino-éthyl)-2,3-dihydro- 1,5-benzothiazépine-4(5H)-one. c) 2-(4-chlorophényl)-3-oxynitro-5-(2-diméthylamino-éthyl)-7-chloro-2.3- dihydo-1,5-benzothiazépine-4(5H)-one, d) 2-(4-méthoxyphényl)-3-oxynitro-5-(2-diméthylamino-éthyl)-7-chloro-2,3- dihydro-1,5-benzothiazépine-4(5H)-one, d) 2-(4-méthoxyphényl)-3-oxynitro-5-(3-diméthylamino-propyl)-7-chloro-2,3- dihydro-1,5-benzothiazépine-4(5H)-one.a) 2- (4-methylphenyl) -3 oxy nitro-5 (2-dimethylamino-ethyl) -2,3 dihydro1,5 benzothiazenine-4 (5H) -one, b) 2- (4-methoyphenyl) -3-oxynitro-5- (2-dimethylamino-ethyl) -2,3-dihydro-1,5-benzothiazepine-4 (5H) -one. c) 2- (4-chlorophenyl) -3-oxynitro-5- (2-dimethylamino-ethyl) -7-chloro-2.3-dihydo-1,5-benzothiazepine-4 (5H) -one, d) 2- ( 4-methoxyphenyl) -3-oxynitro-5- (2-dimethylamino-ethyl) -7-chloro-2,3-dihydro-1,5-benzothiazepine-4 (5H) -one, d) 2- (4-methoxyphenyl ) -3-oxynitro-5- (3-dimethylamino-propyl) -7-chloro-2,3-dihydro-1,5-benzothiazepine-4 (5H) -one.
Z.- Esters nitriques thérapeitiquement actifs de la formule II ou Intermédiaire de synthèse des composés de la formule I,Z.- Therapically active nitric esters of formula II or Intermediate for the synthesis of compounds of formula I,
dans laquelle R et X ont la même signification que dans le formule I. 3.- Procédé de préparation des esers nitriques de benzothiazépines de la formule in which R and X have the same meaning as in formula I. 3.- Process for the preparation of the nitric benzothiazepine esers of the formula
caractérisé par la réaction du produit provenant du mélange de l'anhydride acétique et de l'acide nitrique à 100 % ou à 96 %,préparé extemporané ment,avec un alcool correspondant es benzothiazépines de la formule I. Le rapport molaire des réactifs peut être de 1/1 ou 2/1 ou supérieur. La température de réaction est initialement comprise entre -5°et 0°,puis à la température ambiantejusqu'à ce que le milieu soit homogène.La solution dans le chlorure de méthylène du milieu de réaction est extraite par une solution aqueuse de CO3HNa,puis lavée àl'eaule résidu d'évaption est
dissout dans de l'acétate d'éthyle,cette solution estextriate par de l'eau séchée et le solvant évaporé sous pression réduite.Le résidu cristallise dans de l'eau comme base del ester nitrique dune benzothiazépine de la formule I.characterized by the reaction of the product from the mixture of acetic anhydride and 100% or 96% nitric acid, prepared extemporaneously, with a corresponding alcohol of the benzothiazepines of formula I. The molar ratio of the reactants can be 1/1 or 2/1 or higher. The reaction temperature is initially between -5 ° and 0 °, then at room temperature until the medium is homogeneous. The solution in methylene chloride of the reaction medium is extracted with an aqueous solution of CO 3 HNa , then washed with evaporation residue is dissolved in ethyl acetate, this solution is extracted with dried water and the solvent evaporated under reduced pressure. The residue crystallizes in water as the base of the nitric ester of a benzothiazepine of formula I.
Les esters nitriques des composés de la formule I peuvent si cela est nécessaire être purifiés par absorption sur silice G 60230à 400 mesh. puis élués par de l'acétate d'éthyle.The nitric esters of the compounds of formula I can, if necessary, be purified by absorption on silica G 60230 at 400 mesh. then eluted with ethyl acetate.
4.- Procédé de préparation des ester nitrioues de benzozhiazépines de la formule II,caractérisé par la réactiondu produit réactionnel de l'anhydride acétique et de l'acide nitrique de 100 à 96 % ,préparé extemporanément,avec l'alcool correspondant d'une benzothiazépine de la formule II.4.- Process for the preparation of the benzrihiazepine nitriou ester of formula II, characterized by the reaction of the reaction product of acetic anhydride and nitric acid from 100 to 96%, prepared immediately, with the corresponding alcohol of benzothiazepine of formula II.
5.- Propriété thérapeutique des benzothiazénines de la formule I,caractérisée par une action vaso-dilatatrice,notamment coronarienne.5.- Therapeutic property of the benzothiazenines of formula I, characterized by a vasodilatory action, in particular coronary.
6.- Propriété thérapeutique des benzothiazénines de la formules I,caractérisée par une action anti-dépressante.6.- Therapeutic property of the benzothiazenines of formula I, characterized by an anti-depressant action.
7.- Propriété thérapeutique des benzothiazepines de la formule I,caractérisée par une administration trans épidermique.
7.- Therapeutic property of the benzothiazepines of formula I, characterized by trans epidermal administration.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH1895/86-0 | 1986-05-09 | ||
| CH189586 | 1986-05-09 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1987006934A1 true WO1987006934A1 (en) | 1987-11-19 |
Family
ID=4221108
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CH1987/000053 WO1987006934A1 (en) | 1986-05-09 | 1987-05-08 | Nitric esters of benzothiazepinones |
Country Status (2)
| Country | Link |
|---|---|
| EP (1) | EP0267924A1 (en) |
| WO (1) | WO1987006934A1 (en) |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2034493A1 (en) * | 1969-01-25 | 1970-12-11 | Tanabe Seiyaku Co | |
| US4065488A (en) * | 1977-02-24 | 1977-12-27 | American Home Products Corporation | Process for preparing 1,4:3,6-dianhydro-D-glucitol 2-nitrate |
-
1987
- 1987-05-08 WO PCT/CH1987/000053 patent/WO1987006934A1/en unknown
- 1987-05-08 EP EP19870902370 patent/EP0267924A1/en not_active Withdrawn
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2034493A1 (en) * | 1969-01-25 | 1970-12-11 | Tanabe Seiyaku Co | |
| US4065488A (en) * | 1977-02-24 | 1977-12-27 | American Home Products Corporation | Process for preparing 1,4:3,6-dianhydro-D-glucitol 2-nitrate |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0267924A1 (en) | 1988-05-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0325540A1 (en) | Aromatic esters and thioesters, process for their preparation and their use in human or animal therapeutics and in cosmetics | |
| CA1167047A (en) | 1,4-benzodioxane-2-methoxypropanolamines and their preparation and medical applications | |
| EP0874812B1 (en) | Novel 2,3,5-trimethyl-4-hydroxy anilide derivatives, preparation thereof and therapeutical use thereof | |
| EP0469984B1 (en) | N-sulfonyl-indoline derivatives, their preparation and pharmaceutical compositions containing them | |
| JPH0576939B2 (en) | ||
| EP0068998B1 (en) | (z)-1-aryl-2-aminomethyl-cyclopropane-carboxylates, their preparation and their use as medicines in the treatment of various disorders | |
| EP0461039B1 (en) | Benzimidazole derivatives, process and intermediates for their preparation, their use as medicaments and pharmaceutical compositions containing them | |
| EP0304665B1 (en) | 2,4,6-triamino-n-3-oxide-pyrimidine urea derivatives, their preparation and their cosmetical and dermopharmaceutical use | |
| EP0122827B1 (en) | 1-(2-ethoxycarbonyl-4-phenylalkylamido)-3-amino-2-propanols, their preparations and their medicinal uses | |
| EP0223674A1 (en) | 7-Acyl-benzoxazinones and derivatives thereof, process for their preparation and pharmaceutical compositions containing them | |
| WO1987006934A1 (en) | Nitric esters of benzothiazepinones | |
| EP0487423A1 (en) | Benzoselenazolinone derivatives, their preparation and pharmaceutical compositions containing them | |
| EP0143016B1 (en) | Derivatives of 4-(3-alkynyloxy-2-hydroxy-propyl)-piperazin-1-yl-n-phenyl acetamide, their preparation and their therapeutical use | |
| EP0015214A1 (en) | Derivatives of substituted 2-benzoyl glycinanilides, their preparation and their use in anxiolytic medicaments | |
| FR2483408A1 (en) | ESTERS OF ALCOXYACYLIC DERIVATIVES OF CARNITINE, PROCESS FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING SAME | |
| EP0201400A1 (en) | 4-Phenyl propyl indole derivatives and their salts, process for their preparation, their use as medicines and compositions containing them | |
| EP0294258A1 (en) | Hydrazine derivatives, process for obtaining them and pharmaceutical composition containing them | |
| EP0395526B1 (en) | Benzothiazolinone derivatives, process for their preparation and pharmaceutical compositions containing them | |
| EP0187096A1 (en) | Arylalcoyloxymethyl-2 morpholines, their preparation and their use as therapeutic agents in the treatment of central nervous system diseases | |
| CA1139754A (en) | Ortho chloro 2-benzoyl 4-chloro glycylanilide derivatives, manufacturing process and use as drugs | |
| KR870007911A (en) | Method for preparing hydroxyindole ester | |
| CH643548A5 (en) | 1,3-BENZOXAZINE-2,4-DIONE DERIVATIVES, THEIR PREPARATION AND MEDICAMENTS CONTAINING THEM | |
| FR2504925A1 (en) | N-2-amino 3-alkoxy or phenoxy-propyl urea and thiourea derivs. - are cardiovascular agents esp. used to treat arrhythmia | |
| EP0871611B1 (en) | New derivatives of glycylanilides, preparation and therapeutical application | |
| LU86259A1 (en) | AROMATIC AMIDES, THEIR PREPARATION PROCESS AND THEIR USE IN HUMAN OR VETERINARY MEDICINE AND IN COSMETICS |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE FR GB IT LU NL SE |