USRE46095E1 - Herbicidal compositions - Google Patents
Herbicidal compositions Download PDFInfo
- Publication number
- USRE46095E1 USRE46095E1 US14/596,041 US201514596041A USRE46095E US RE46095 E1 USRE46095 E1 US RE46095E1 US 201514596041 A US201514596041 A US 201514596041A US RE46095 E USRE46095 E US RE46095E
- Authority
- US
- United States
- Prior art keywords
- plants
- herbicide combination
- herbicide
- compounds
- active compounds
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 0 [1*]C1=NC(N([2*])[3*])=NC(NC2([H])C3=C([8*])C([7*])=C([6*])C([5*])=C3*CC2[4*])=N1 Chemical compound [1*]C1=NC(N([2*])[3*])=NC(NC2([H])C3=C([8*])C([7*])=C([6*])C([5*])=C3*CC2[4*])=N1 0.000 description 6
- YFONKFDEZLYQDH-OPQQBVKSSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC(C)=CC=C3C[C@@H]2C)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC(C)=CC=C3C[C@@H]2C)=N1 YFONKFDEZLYQDH-OPQQBVKSSA-N 0.000 description 4
- AVXMNBKONPAEIC-UHFFFAOYSA-N [H]C1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCOC2=C(C)C(C)=CC=C21 Chemical compound [H]C1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCOC2=C(C)C(C)=CC=C21 AVXMNBKONPAEIC-UHFFFAOYSA-N 0.000 description 3
- YBSFKZDTZUZBSM-UHFFFAOYSA-N [H]C1(NC2=NC(CC)=NC(N)=N2)CCCC2=CC=C(C)C=C21 Chemical compound [H]C1(NC2=NC(CC)=NC(N)=N2)CCCC2=CC=C(C)C=C21 YBSFKZDTZUZBSM-UHFFFAOYSA-N 0.000 description 3
- NEQSSOIGWBJMTE-UHFFFAOYSA-N CC1=CC=C2CCC(NC3=NC(C(C)C)=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CCC(NC3=NC(C(C)C)=NC(N)=N3)C2=C1 NEQSSOIGWBJMTE-UHFFFAOYSA-N 0.000 description 2
- PBYYUKGWLNONHO-UHFFFAOYSA-N CC1=CC=C2OCCC(NC3=NC(C)=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2OCCC(NC3=NC(C)=NC(N)=N3)C2=C1 PBYYUKGWLNONHO-UHFFFAOYSA-N 0.000 description 2
- VRMAJKHLEGDABJ-GFCCVEGCSA-N [H]C([H])(C)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C=C32)=N1 Chemical compound [H]C([H])(C)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C=C32)=N1 VRMAJKHLEGDABJ-GFCCVEGCSA-N 0.000 description 2
- SGWOQYJRAGQVKU-UHFFFAOYSA-N [H]C1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCC2=CC=C(CC)C=C21 Chemical compound [H]C1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCC2=CC=C(CC)C=C21 SGWOQYJRAGQVKU-UHFFFAOYSA-N 0.000 description 2
- OJDRDFIVPJISKX-UHFFFAOYSA-N [H]C1(NC2=NC(C)=NC(N)=N2)CCC2=CC=C(CC)C=C21 Chemical compound [H]C1(NC2=NC(C)=NC(N)=N2)CCC2=CC=C(CC)C=C21 OJDRDFIVPJISKX-UHFFFAOYSA-N 0.000 description 2
- ZOZQHRCMNROJSG-UHFFFAOYSA-N [H]C1(NC2=NC(CC)=NC(N)=N2)CCC2=CC=C(CC)C=C21 Chemical compound [H]C1(NC2=NC(CC)=NC(N)=N2)CCC2=CC=C(CC)C=C21 ZOZQHRCMNROJSG-UHFFFAOYSA-N 0.000 description 2
- BAPPLRJUXSNEGX-UHFFFAOYSA-N [H]C1(NC2=NC(CC)=NC(N)=N2)CCOC2=C(C)C(C)=CC=C21 Chemical compound [H]C1(NC2=NC(CC)=NC(N)=N2)CCOC2=C(C)C(C)=CC=C21 BAPPLRJUXSNEGX-UHFFFAOYSA-N 0.000 description 2
- WZCCTBCFBOFFNH-UHFFFAOYSA-N [H]C1(NC2=NC(CCC)=NC(N)=N2)C2=CC=CC=C2CCC1C Chemical compound [H]C1(NC2=NC(CCC)=NC(N)=N2)C2=CC=CC=C2CCC1C WZCCTBCFBOFFNH-UHFFFAOYSA-N 0.000 description 2
- LUDKSPRHCCBQEM-UHFFFAOYSA-N [H]C1(NC2=NC(CCC)=NC(N)=N2)CCC2=CC=C(CC)C=C21 Chemical compound [H]C1(NC2=NC(CCC)=NC(N)=N2)CCC2=CC=C(CC)C=C21 LUDKSPRHCCBQEM-UHFFFAOYSA-N 0.000 description 2
- PJAIMWYTWFLUIS-UHFFFAOYSA-N [H]C1(NC2=NC(CCC)=NC(N)=N2)CCOC2=C(C)C(C)=CC=C21 Chemical compound [H]C1(NC2=NC(CCC)=NC(N)=N2)CCOC2=C(C)C(C)=CC=C21 PJAIMWYTWFLUIS-UHFFFAOYSA-N 0.000 description 2
- CYQCFCJBHXWBFK-GXFFZTMASA-N [H][C@@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC(C)=C(C)C=C32)=N1 Chemical compound [H][C@@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC(C)=C(C)C=C32)=N1 CYQCFCJBHXWBFK-GXFFZTMASA-N 0.000 description 2
- UYPSSXJUTSZDRJ-GFCCVEGCSA-N [H][C@@]1(NC2=NC=NC(N)=N2)CCCC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC=NC(N)=N2)CCCC2=CC=C(C)C=C21 UYPSSXJUTSZDRJ-GFCCVEGCSA-N 0.000 description 2
- HSYYSQWDHGPRNV-UHFFFAOYSA-N CC(C)(F)C1=NC(N)=NC(NC2CCC3=CC=CC=C32)=N1 Chemical compound CC(C)(F)C1=NC(N)=NC(NC2CCC3=CC=CC=C32)=N1 HSYYSQWDHGPRNV-UHFFFAOYSA-N 0.000 description 1
- AVXMNBKONPAEIC-GFCCVEGCSA-N CC(C)(c1nc(N)nc(N[C@H]2c3ccc(C)c(C)c3OCC2)n1)F Chemical compound CC(C)(c1nc(N)nc(N[C@H]2c3ccc(C)c(C)c3OCC2)n1)F AVXMNBKONPAEIC-GFCCVEGCSA-N 0.000 description 1
- KSTPUSBFAGYIHA-UHFFFAOYSA-N CC(F)C1=NC(N)=NC(NC2C3=CC=CC=C3CC2C)=N1 Chemical compound CC(F)C1=NC(N)=NC(NC2C3=CC=CC=C3CC2C)=N1 KSTPUSBFAGYIHA-UHFFFAOYSA-N 0.000 description 1
- ZTJZCGWUTGCBGC-UHFFFAOYSA-N CC(F)C1=NC(N)=NC(NC2C3=CC=CC=C3CCC2C)=N1 Chemical compound CC(F)C1=NC(N)=NC(NC2C3=CC=CC=C3CCC2C)=N1 ZTJZCGWUTGCBGC-UHFFFAOYSA-N 0.000 description 1
- ZADJLTSOJSBSKK-UHFFFAOYSA-N CC(F)C1=NC(N)=NC(NC2CCC3=CC=C(F)C=C32)=N1 Chemical compound CC(F)C1=NC(N)=NC(NC2CCC3=CC=C(F)C=C32)=N1 ZADJLTSOJSBSKK-UHFFFAOYSA-N 0.000 description 1
- DMHTZMUKTPELHM-UHFFFAOYSA-N CC(F)C1=NC(N)=NC(NC2CCC3=CC=CC=C32)=N1 Chemical compound CC(F)C1=NC(N)=NC(NC2CCC3=CC=CC=C32)=N1 DMHTZMUKTPELHM-UHFFFAOYSA-N 0.000 description 1
- RGJNSAJDECMUHM-UHFFFAOYSA-N CC(F)C1=NC(N)=NC(NC2CCCC3=CC=CC=C32)=N1 Chemical compound CC(F)C1=NC(N)=NC(NC2CCCC3=CC=CC=C32)=N1 RGJNSAJDECMUHM-UHFFFAOYSA-N 0.000 description 1
- ODBBCCGHGWRMDS-UHFFFAOYSA-N CC(F)C1=NC(N)=NC(NC2CCOC3=CC=CC=C32)=N1 Chemical compound CC(F)C1=NC(N)=NC(NC2CCOC3=CC=CC=C32)=N1 ODBBCCGHGWRMDS-UHFFFAOYSA-N 0.000 description 1
- CYQCFCJBHXWBFK-UHFFFAOYSA-N CC1=C(C)C=C2C(=C1)CCC2NC1=NC(C(C)F)=NC(N)=N1 Chemical compound CC1=C(C)C=C2C(=C1)CCC2NC1=NC(C(C)F)=NC(N)=N1 CYQCFCJBHXWBFK-UHFFFAOYSA-N 0.000 description 1
- DTPHPXHVZJXWOC-UHFFFAOYSA-N CC1=C(C)C=C2C(=C1)CCC2NC1=NC=NC(N)=N1 Chemical compound CC1=C(C)C=C2C(=C1)CCC2NC1=NC=NC(N)=N1 DTPHPXHVZJXWOC-UHFFFAOYSA-N 0.000 description 1
- BQJACPPUSXKAHZ-UHFFFAOYSA-N CC1=C(F)C=C2CCC(NC3=NC(C(C)F)=NC(N)=N3)C2=C1 Chemical compound CC1=C(F)C=C2CCC(NC3=NC(C(C)F)=NC(N)=N3)C2=C1 BQJACPPUSXKAHZ-UHFFFAOYSA-N 0.000 description 1
- GZCYDTWEJRABAU-UHFFFAOYSA-N CC1=CC(=O)N(C2=CC(C(=O)CS(=O)(=O)N(C)C(C)C)=C(Cl)C=C2F)C(=O)N1C Chemical compound CC1=CC(=O)N(C2=CC(C(=O)CS(=O)(=O)N(C)C(C)C)=C(Cl)C=C2F)C(=O)N1C GZCYDTWEJRABAU-UHFFFAOYSA-N 0.000 description 1
- DYWRAFBBUFUGMW-UHFFFAOYSA-N CC1=CC(C)=C2CCC(NC3=NC(C)=NC(N)=N3)C2=C1 Chemical compound CC1=CC(C)=C2CCC(NC3=NC(C)=NC(N)=N3)C2=C1 DYWRAFBBUFUGMW-UHFFFAOYSA-N 0.000 description 1
- YXWYFLKWKRSTFP-UHFFFAOYSA-N CC1=CC(C)=C2CCC(NC3=NC=NC(N)=N3)C2=C1 Chemical compound CC1=CC(C)=C2CCC(NC3=NC=NC(N)=N3)C2=C1 YXWYFLKWKRSTFP-UHFFFAOYSA-N 0.000 description 1
- ITNMLCGSHMGFHC-UHFFFAOYSA-N CC1=CC(C)=C2CCCC(NC3=NC(C(C)F)=NC(N)=N3)C2=C1 Chemical compound CC1=CC(C)=C2CCCC(NC3=NC(C(C)F)=NC(N)=N3)C2=C1 ITNMLCGSHMGFHC-UHFFFAOYSA-N 0.000 description 1
- KDLDOSBKSFGNTF-UHFFFAOYSA-N CC1=CC=C2C(=C1C)OCCC2NC1=NC(C(C)F)=NC(N)=N1 Chemical compound CC1=CC=C2C(=C1C)OCCC2NC1=NC(C(C)F)=NC(N)=N1 KDLDOSBKSFGNTF-UHFFFAOYSA-N 0.000 description 1
- HASKBXRBSOROHL-UHFFFAOYSA-N CC1=CC=C2CC(C)C(NC3=NC(C(C)(C)F)=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CC(C)C(NC3=NC(C(C)(C)F)=NC(N)=N3)C2=C1 HASKBXRBSOROHL-UHFFFAOYSA-N 0.000 description 1
- YFONKFDEZLYQDH-UHFFFAOYSA-N CC1=CC=C2CC(C)C(NC3=NC(C(C)F)=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CC(C)C(NC3=NC(C(C)F)=NC(N)=N3)C2=C1 YFONKFDEZLYQDH-UHFFFAOYSA-N 0.000 description 1
- OFVCGBCCPNUKHQ-UHFFFAOYSA-N CC1=CC=C2CC(C)C(NC3=NC(C)=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CC(C)C(NC3=NC(C)=NC(N)=N3)C2=C1 OFVCGBCCPNUKHQ-UHFFFAOYSA-N 0.000 description 1
- ABFOFYVGUAQOCS-UHFFFAOYSA-N CC1=CC=C2CC(C)C(NC3=NC=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CC(C)C(NC3=NC=NC(N)=N3)C2=C1 ABFOFYVGUAQOCS-UHFFFAOYSA-N 0.000 description 1
- VZMXMUUMSCAJLL-UHFFFAOYSA-N CC1=CC=C2CCC(NC3=NC(C(C)(C)F)=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CCC(NC3=NC(C(C)(C)F)=NC(N)=N3)C2=C1 VZMXMUUMSCAJLL-UHFFFAOYSA-N 0.000 description 1
- ODUPNTHHJAJTEA-UHFFFAOYSA-N CC1=CC=C2CCC(NC3=NC(C(C)F)=NC(N)=N3)C2=C1C Chemical compound CC1=CC=C2CCC(NC3=NC(C(C)F)=NC(N)=N3)C2=C1C ODUPNTHHJAJTEA-UHFFFAOYSA-N 0.000 description 1
- UYKRKNNPPJOCGX-UHFFFAOYSA-N CC1=CC=C2CCC(NC3=NC(C)=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CCC(NC3=NC(C)=NC(N)=N3)C2=C1 UYKRKNNPPJOCGX-UHFFFAOYSA-N 0.000 description 1
- RAMCTRCUFFUYAU-UHFFFAOYSA-N CC1=CC=C2CCC(NC3=NC=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CCC(NC3=NC=NC(N)=N3)C2=C1 RAMCTRCUFFUYAU-UHFFFAOYSA-N 0.000 description 1
- YVODLZWNLHIKID-UHFFFAOYSA-N CC1=CC=C2CCCC(NC3=NC(C(C)F)=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CCCC(NC3=NC(C(C)F)=NC(N)=N3)C2=C1 YVODLZWNLHIKID-UHFFFAOYSA-N 0.000 description 1
- UYPSSXJUTSZDRJ-UHFFFAOYSA-N CC1=CC=C2CCCC(NC3=NC=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2CCCC(NC3=NC=NC(N)=N3)C2=C1 UYPSSXJUTSZDRJ-UHFFFAOYSA-N 0.000 description 1
- WGTHLJFCLNVZBF-UHFFFAOYSA-N CC1=CC=C2OCCC(NC3=NC(C(C)(C)F)=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2OCCC(NC3=NC(C(C)(C)F)=NC(N)=N3)C2=C1 WGTHLJFCLNVZBF-UHFFFAOYSA-N 0.000 description 1
- BKTNAIJYAXLLGB-UHFFFAOYSA-N CC1=CC=C2OCCC(NC3=NC=NC(N)=N3)C2=C1 Chemical compound CC1=CC=C2OCCC(NC3=NC=NC(N)=N3)C2=C1 BKTNAIJYAXLLGB-UHFFFAOYSA-N 0.000 description 1
- FJLRHDREFRAVAW-UHFFFAOYSA-N CC1=NC(N)=NC(NC2C3=CC=CC=C3CCC2C)=N1 Chemical compound CC1=NC(N)=NC(NC2C3=CC=CC=C3CCC2C)=N1 FJLRHDREFRAVAW-UHFFFAOYSA-N 0.000 description 1
- ZMQQPNJRNOSBMM-UHFFFAOYSA-N CC1=NC(N)=NC(NC2CCC3=CC(C)=C(C)C=C32)=N1 Chemical compound CC1=NC(N)=NC(NC2CCC3=CC(C)=C(C)C=C32)=N1 ZMQQPNJRNOSBMM-UHFFFAOYSA-N 0.000 description 1
- QNLVIRFXEWWEDT-UHFFFAOYSA-N CC1=NC(N)=NC(NC2CCC3=CC(F)=C(C)C=C32)=N1 Chemical compound CC1=NC(N)=NC(NC2CCC3=CC(F)=C(C)C=C32)=N1 QNLVIRFXEWWEDT-UHFFFAOYSA-N 0.000 description 1
- DJHJQRYZBSINET-UHFFFAOYSA-N CC1=NC(N)=NC(NC2CCC3=CC=CC=C32)=N1 Chemical compound CC1=NC(N)=NC(NC2CCC3=CC=CC=C32)=N1 DJHJQRYZBSINET-UHFFFAOYSA-N 0.000 description 1
- DWSPRBSLSXQIEJ-UHFFFAOYSA-N CC1=NN(C)C(O)=C1C(=O)C1=CC=C(C(F)(F)F)C=C1S(C)(=O)=O Chemical compound CC1=NN(C)C(O)=C1C(=O)C1=CC=C(C(F)(F)F)C=C1S(C)(=O)=O DWSPRBSLSXQIEJ-UHFFFAOYSA-N 0.000 description 1
- BEVCALTXKNSIAZ-UHFFFAOYSA-N CC1CCC2=CC=CC=C2C1NC1=NC(C(C)(C)F)=NC(N)=N1 Chemical compound CC1CCC2=CC=CC=C2C1NC1=NC(C(C)(C)F)=NC(N)=N1 BEVCALTXKNSIAZ-UHFFFAOYSA-N 0.000 description 1
- XHMWUIBRNPCLQF-UHFFFAOYSA-N CCC(F)C1=CC(N)=CC(NC2C3=CC(C)=CC=C3CC2C)=C1 Chemical compound CCC(F)C1=CC(N)=CC(NC2C3=CC(C)=CC=C3CC2C)=C1 XHMWUIBRNPCLQF-UHFFFAOYSA-N 0.000 description 1
- BQWOIXAAQZWCCA-LSLKUGRBSA-N CCC(F)C1=NC(N)=NC(NC2CCCC3=CC=C(C)C=C32)=N1 Chemical compound CCC(F)C1=NC(N)=NC(NC2CCCC3=CC=C(C)C=C32)=N1 BQWOIXAAQZWCCA-LSLKUGRBSA-N 0.000 description 1
- ULWSXRCTIDLNFK-UHFFFAOYSA-N CCC(F)C1=NC(N)=NC(NC2CCCC3=CC=CC=C32)=N1 Chemical compound CCC(F)C1=NC(N)=NC(NC2CCCC3=CC=CC=C32)=N1 ULWSXRCTIDLNFK-UHFFFAOYSA-N 0.000 description 1
- FKECSAQRVODFJX-UHFFFAOYSA-N CCC1=NC(N)=NC(NC2C3=CC(C)=CC=C3CC2C)=N1 Chemical compound CCC1=NC(N)=NC(NC2C3=CC(C)=CC=C3CC2C)=N1 FKECSAQRVODFJX-UHFFFAOYSA-N 0.000 description 1
- BWWQWIUFESVXDF-UHFFFAOYSA-N CCC1=NC(N)=NC(NC2C3=CC=CC=C3CCC2C)=N1 Chemical compound CCC1=NC(N)=NC(NC2C3=CC=CC=C3CCC2C)=N1 BWWQWIUFESVXDF-UHFFFAOYSA-N 0.000 description 1
- ZNOQBAIGFBTSTQ-UHFFFAOYSA-N CCC1=NC(N)=NC(NC2CCC3=C(C)C=C(C)C=C32)=N1 Chemical compound CCC1=NC(N)=NC(NC2CCC3=C(C)C=C(C)C=C32)=N1 ZNOQBAIGFBTSTQ-UHFFFAOYSA-N 0.000 description 1
- FBDFIBNIZBFUBM-UHFFFAOYSA-N CCC1=NC(N)=NC(NC2CCC3=CC(F)=C(C)C=C32)=N1 Chemical compound CCC1=NC(N)=NC(NC2CCC3=CC(F)=C(C)C=C32)=N1 FBDFIBNIZBFUBM-UHFFFAOYSA-N 0.000 description 1
- JJYVYPKELQHLMS-UHFFFAOYSA-N CCC1=NC(N)=NC(NC2CCC3=CC=C(C)C(C)=C32)=N1 Chemical compound CCC1=NC(N)=NC(NC2CCC3=CC=C(C)C(C)=C32)=N1 JJYVYPKELQHLMS-UHFFFAOYSA-N 0.000 description 1
- VRMAJKHLEGDABJ-UHFFFAOYSA-N CCC1=NC(N)=NC(NC2CCC3=CC=C(C)C=C32)=N1 Chemical compound CCC1=NC(N)=NC(NC2CCC3=CC=C(C)C=C32)=N1 VRMAJKHLEGDABJ-UHFFFAOYSA-N 0.000 description 1
- QTUVOQUQNXGNRR-UHFFFAOYSA-N CCC1=NC(N)=NC(NC2CCOC3=CC=C(C)C=C32)=N1 Chemical compound CCC1=NC(N)=NC(NC2CCOC3=CC=C(C)C=C32)=N1 QTUVOQUQNXGNRR-UHFFFAOYSA-N 0.000 description 1
- ATDWACRVTKJURQ-UHFFFAOYSA-N CCCC(F)C1=NC(N)=NC(NC2C3=CC(C)=CC=C3CC2C)=N1 Chemical compound CCCC(F)C1=NC(N)=NC(NC2C3=CC(C)=CC=C3CC2C)=N1 ATDWACRVTKJURQ-UHFFFAOYSA-N 0.000 description 1
- RIHGEWPQEYDNKC-UHFFFAOYSA-N CCCC1=NC(N)=NC(NC2C3=CC(C)=CC=C3CC2C)=N1 Chemical compound CCCC1=NC(N)=NC(NC2C3=CC(C)=CC=C3CC2C)=N1 RIHGEWPQEYDNKC-UHFFFAOYSA-N 0.000 description 1
- GVMXSOSITWGPME-UHFFFAOYSA-N CCCC1=NC(N)=NC(NC2CCC3=CC=C(C)C=C32)=N1 Chemical compound CCCC1=NC(N)=NC(NC2CCC3=CC=C(C)C=C32)=N1 GVMXSOSITWGPME-UHFFFAOYSA-N 0.000 description 1
- HPYFKDLTGXFVSH-UHFFFAOYSA-N CCCC1=NC(N)=NC(NC2CCOC3=CC=C(C)C=C32)=N1 Chemical compound CCCC1=NC(N)=NC(NC2CCOC3=CC=C(C)C=C32)=N1 HPYFKDLTGXFVSH-UHFFFAOYSA-N 0.000 description 1
- VYJQPQIQJQVONN-UHFFFAOYSA-N CCOC(=O)COC1=C(OC2=C(Cl)C=C(F)C(N3C(=O)C=C(C)N(C)C3=O)=C2)C=CC=N1 Chemical compound CCOC(=O)COC1=C(OC2=C(Cl)C=C(F)C(N3C(=O)C=C(C)N(C)C3=O)=C2)C=CC=N1 VYJQPQIQJQVONN-UHFFFAOYSA-N 0.000 description 1
- IRCMYGHHKLLGHV-UHFFFAOYSA-N CCOC1OC2=CC=C(OS(C)(=O)=O)C=C2C1(C)C Chemical compound CCOC1OC2=CC=C(OS(C)(=O)=O)C=C2C1(C)C IRCMYGHHKLLGHV-UHFFFAOYSA-N 0.000 description 1
- CASLETQIYIQFTQ-UHFFFAOYSA-N CN1N=C(C(F)(F)F)C(CS(=O)(=O)C2=NOC(C)(C)C2)=C1OC(F)F Chemical compound CN1N=C(C(F)(F)F)C(CS(=O)(=O)C2=NOC(C)(C)C2)=C1OC(F)F CASLETQIYIQFTQ-UHFFFAOYSA-N 0.000 description 1
- IDYIQDLGRGWZBP-UHFFFAOYSA-N CO(O)SC1=CC=C(C(=O)C2C(=O)CCCC2=O)C(Cl)=C1COCC(F)(F)F Chemical compound CO(O)SC1=CC=C(C(=O)C2C(=O)CCCC2=O)C(Cl)=C1COCC(F)(F)F IDYIQDLGRGWZBP-UHFFFAOYSA-N 0.000 description 1
- WWBGJHXSLPEIBX-UHFFFAOYSA-N COC1=CC(OC)=NC(NC(=O)CS(=O)(=O)C2=C(OCC(F)(F)F)C=CC=N2)=N1 Chemical compound COC1=CC(OC)=NC(NC(=O)CS(=O)(=O)C2=C(OCC(F)(F)F)C=CC=N2)=N1 WWBGJHXSLPEIBX-UHFFFAOYSA-N 0.000 description 1
- AHGJZBKNCSRNOO-UHFFFAOYSA-N COC1=CC(OC)=NC2=NC(CS(=O)(=O)C3=C(C(F)(F)F)C=CC=C3OCC(F)F)=NN12 Chemical compound COC1=CC(OC)=NC2=NC(CS(=O)(=O)C3=C(C(F)(F)F)C=CC=C3OCC(F)F)=NN12 AHGJZBKNCSRNOO-UHFFFAOYSA-N 0.000 description 1
- PLEUAHMFISRJCR-UHFFFAOYSA-N COC1=CC(OC)=NC2=NC(CS(=O)(=O)C3=C(C(F)(F)F)C=CN=C3OC)=NN12 Chemical compound COC1=CC(OC)=NC2=NC(CS(=O)(=O)C3=C(C(F)(F)F)C=CN=C3OC)=NN12 PLEUAHMFISRJCR-UHFFFAOYSA-N 0.000 description 1
- WCBCSPDPXWQIFG-UHFFFAOYSA-N COC1=NN(C(=O)NO(O)SC2=C(C)SC=C2C(=O)O)C(=O)N1C Chemical compound COC1=NN(C(=O)NO(O)SC2=C(C)SC=C2C(=O)O)C(=O)N1C WCBCSPDPXWQIFG-UHFFFAOYSA-N 0.000 description 1
- HUYBEDCQLAEVPD-UHFFFAOYSA-N COCCOCC1=NC(C(F)(F)F)=CC=C1C(=O)C1=C(O)C2CCC(C2)C1=O Chemical compound COCCOCC1=NC(C(F)(F)F)=CC=C1C(=O)C1=C(O)C2CCC(C2)C1=O HUYBEDCQLAEVPD-UHFFFAOYSA-N 0.000 description 1
- SZYMLAKHSJXMEC-MNRXQERYSA-N C[C@@H](Cc1c2cc(C)cc1)[C@H]2Nc1nc([C@@](C)(NC)F)nc(N)n1 Chemical compound C[C@@H](Cc1c2cc(C)cc1)[C@H]2Nc1nc([C@@](C)(NC)F)nc(N)n1 SZYMLAKHSJXMEC-MNRXQERYSA-N 0.000 description 1
- NIXXQNOQHKNPEJ-UHFFFAOYSA-N NC1=C(Cl)C(C(=O)O)=NC(Cl)=C1 Chemical compound NC1=C(Cl)C(C(=O)O)=NC(Cl)=C1 NIXXQNOQHKNPEJ-UHFFFAOYSA-N 0.000 description 1
- KWAIHLIXESXTJL-UHFFFAOYSA-N NC1=NC(C2CC2)=NC(C(=O)O)=C1Cl Chemical compound NC1=NC(C2CC2)=NC(C(=O)O)=C1Cl KWAIHLIXESXTJL-UHFFFAOYSA-N 0.000 description 1
- XWKYBCPQSOQBTC-UHFFFAOYSA-N [H]C(C)(F)C1=NC(N)=NC(NC2CCC3=CC=C(C)C=C32)=N1 Chemical compound [H]C(C)(F)C1=NC(N)=NC(NC2CCC3=CC=C(C)C=C32)=N1 XWKYBCPQSOQBTC-UHFFFAOYSA-N 0.000 description 1
- YETAYWRZVCIPBZ-UHFFFAOYSA-N [H]C(F)(CC)C1=NC(N)=NC(NC2CCC3=CC=C(C)C=C32)=N1 Chemical compound [H]C(F)(CC)C1=NC(N)=NC(NC2CCC3=CC=C(C)C=C32)=N1 YETAYWRZVCIPBZ-UHFFFAOYSA-N 0.000 description 1
- YETAYWRZVCIPBZ-ZGTCLIOFSA-N [H]C(F)(CC)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C=C32)=N1 Chemical compound [H]C(F)(CC)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C=C32)=N1 YETAYWRZVCIPBZ-ZGTCLIOFSA-N 0.000 description 1
- YFONKFDEZLYQDH-OUJBWJOFSA-N [H][C@@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC(C)=CC=C3C[C@@H]2C)=N1 Chemical compound [H][C@@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC(C)=CC=C3C[C@@H]2C)=N1 YFONKFDEZLYQDH-OUJBWJOFSA-N 0.000 description 1
- YFONKFDEZLYQDH-GBIKHYSHSA-N [H][C@@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC(C)=CC=C3C[C@H]2C)=N1 Chemical compound [H][C@@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC(C)=CC=C3C[C@H]2C)=N1 YFONKFDEZLYQDH-GBIKHYSHSA-N 0.000 description 1
- BQJACPPUSXKAHZ-QPUJVOFHSA-N [H][C@@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC(F)=C(C)C=C32)=N1 Chemical compound [H][C@@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC(F)=C(C)C=C32)=N1 BQJACPPUSXKAHZ-QPUJVOFHSA-N 0.000 description 1
- ODUPNTHHJAJTEA-CMPLNLGQSA-N [H][C@@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C(C)=C32)=N1 Chemical compound [H][C@@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C(C)=C32)=N1 ODUPNTHHJAJTEA-CMPLNLGQSA-N 0.000 description 1
- VZMXMUUMSCAJLL-GFCCVEGCSA-N [H][C@@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCC2=CC=C(C)C=C21 VZMXMUUMSCAJLL-GFCCVEGCSA-N 0.000 description 1
- HSYYSQWDHGPRNV-LLVKDONJSA-N [H][C@@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCC2=CC=CC=C21 Chemical compound [H][C@@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCC2=CC=CC=C21 HSYYSQWDHGPRNV-LLVKDONJSA-N 0.000 description 1
- JTOXQKDXPQABPU-GFCCVEGCSA-N [H][C@@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCCC2=CC=CC=C21 Chemical compound [H][C@@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCCC2=CC=CC=C21 JTOXQKDXPQABPU-GFCCVEGCSA-N 0.000 description 1
- WGTHLJFCLNVZBF-LLVKDONJSA-N [H][C@@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCOC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)CCOC2=CC=C(C)C=C21 WGTHLJFCLNVZBF-LLVKDONJSA-N 0.000 description 1
- NEQSSOIGWBJMTE-CYBMUJFWSA-N [H][C@@]1(NC2=NC(C(C)C)=NC(N)=N2)CCC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(C(C)C)=NC(N)=N2)CCC2=CC=C(C)C=C21 NEQSSOIGWBJMTE-CYBMUJFWSA-N 0.000 description 1
- DMHTZMUKTPELHM-QHDYGNBISA-N [H][C@@]1(NC2=NC(C(C)F)=NC(N)=N2)CCC2=CC=CC=C21 Chemical compound [H][C@@]1(NC2=NC(C(C)F)=NC(N)=N2)CCC2=CC=CC=C21 DMHTZMUKTPELHM-QHDYGNBISA-N 0.000 description 1
- BQWOIXAAQZWCCA-ARLHGKGLSA-N [H][C@@]1(NC2=NC(C(F)CC)=NC(N)=N2)CCCC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(C(F)CC)=NC(N)=N2)CCCC2=CC=C(C)C=C21 BQWOIXAAQZWCCA-ARLHGKGLSA-N 0.000 description 1
- ULWSXRCTIDLNFK-ZGTCLIOFSA-N [H][C@@]1(NC2=NC(C(F)CC)=NC(N)=N2)CCCC2=CC=CC=C21 Chemical compound [H][C@@]1(NC2=NC(C(F)CC)=NC(N)=N2)CCCC2=CC=CC=C21 ULWSXRCTIDLNFK-ZGTCLIOFSA-N 0.000 description 1
- DYWRAFBBUFUGMW-CYBMUJFWSA-N [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=C(C)C=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=C(C)C=C(C)C=C21 DYWRAFBBUFUGMW-CYBMUJFWSA-N 0.000 description 1
- ZMQQPNJRNOSBMM-CYBMUJFWSA-N [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=CC(C)=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=CC(C)=C(C)C=C21 ZMQQPNJRNOSBMM-CYBMUJFWSA-N 0.000 description 1
- QNLVIRFXEWWEDT-GFCCVEGCSA-N [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=CC(F)=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=CC(F)=C(C)C=C21 QNLVIRFXEWWEDT-GFCCVEGCSA-N 0.000 description 1
- UYKRKNNPPJOCGX-GFCCVEGCSA-N [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=CC=C(C)C=C21 UYKRKNNPPJOCGX-GFCCVEGCSA-N 0.000 description 1
- DJHJQRYZBSINET-LLVKDONJSA-N [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=CC=CC=C21 Chemical compound [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCC2=CC=CC=C21 DJHJQRYZBSINET-LLVKDONJSA-N 0.000 description 1
- PBYYUKGWLNONHO-LLVKDONJSA-N [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCOC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(C)=NC(N)=N2)CCOC2=CC=C(C)C=C21 PBYYUKGWLNONHO-LLVKDONJSA-N 0.000 description 1
- ZNOQBAIGFBTSTQ-CYBMUJFWSA-N [H][C@@]1(NC2=NC(CC)=NC(N)=N2)CCC2=C(C)C=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(CC)=NC(N)=N2)CCC2=C(C)C=C(C)C=C21 ZNOQBAIGFBTSTQ-CYBMUJFWSA-N 0.000 description 1
- FBDFIBNIZBFUBM-GFCCVEGCSA-N [H][C@@]1(NC2=NC(CC)=NC(N)=N2)CCC2=CC(F)=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(CC)=NC(N)=N2)CCC2=CC(F)=C(C)C=C21 FBDFIBNIZBFUBM-GFCCVEGCSA-N 0.000 description 1
- JJYVYPKELQHLMS-GFCCVEGCSA-N [H][C@@]1(NC2=NC(CC)=NC(N)=N2)CCC2=CC=C(C)C(C)=C21 Chemical compound [H][C@@]1(NC2=NC(CC)=NC(N)=N2)CCC2=CC=C(C)C(C)=C21 JJYVYPKELQHLMS-GFCCVEGCSA-N 0.000 description 1
- QTUVOQUQNXGNRR-LLVKDONJSA-N [H][C@@]1(NC2=NC(CC)=NC(N)=N2)CCOC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(CC)=NC(N)=N2)CCOC2=CC=C(C)C=C21 QTUVOQUQNXGNRR-LLVKDONJSA-N 0.000 description 1
- GVMXSOSITWGPME-CYBMUJFWSA-N [H][C@@]1(NC2=NC(CCC)=NC(N)=N2)CCC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(CCC)=NC(N)=N2)CCC2=CC=C(C)C=C21 GVMXSOSITWGPME-CYBMUJFWSA-N 0.000 description 1
- HPYFKDLTGXFVSH-GFCCVEGCSA-N [H][C@@]1(NC2=NC(CCC)=NC(N)=N2)CCOC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC(CCC)=NC(N)=N2)CCOC2=CC=C(C)C=C21 HPYFKDLTGXFVSH-GFCCVEGCSA-N 0.000 description 1
- YXWYFLKWKRSTFP-GFCCVEGCSA-N [H][C@@]1(NC2=NC=NC(N)=N2)CCC2=C(C)C=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC=NC(N)=N2)CCC2=C(C)C=C(C)C=C21 YXWYFLKWKRSTFP-GFCCVEGCSA-N 0.000 description 1
- DTPHPXHVZJXWOC-GFCCVEGCSA-N [H][C@@]1(NC2=NC=NC(N)=N2)CCC2=CC(C)=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC=NC(N)=N2)CCC2=CC(C)=C(C)C=C21 DTPHPXHVZJXWOC-GFCCVEGCSA-N 0.000 description 1
- RAMCTRCUFFUYAU-LLVKDONJSA-N [H][C@@]1(NC2=NC=NC(N)=N2)CCC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC=NC(N)=N2)CCC2=CC=C(C)C=C21 RAMCTRCUFFUYAU-LLVKDONJSA-N 0.000 description 1
- BKTNAIJYAXLLGB-SNVBAGLBSA-N [H][C@@]1(NC2=NC=NC(N)=N2)CCOC2=CC=C(C)C=C21 Chemical compound [H][C@@]1(NC2=NC=NC(N)=N2)CCOC2=CC=C(C)C=C21 BKTNAIJYAXLLGB-SNVBAGLBSA-N 0.000 description 1
- YFONKFDEZLYQDH-GIPNMCIBSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC(C)=CC=C3C[C@H]2C)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC(C)=CC=C3C[C@H]2C)=N1 YFONKFDEZLYQDH-GIPNMCIBSA-N 0.000 description 1
- KSTPUSBFAGYIHA-YGOYTEALSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC=CC=C3C[C@@H]2C)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC=CC=C3C[C@@H]2C)=N1 KSTPUSBFAGYIHA-YGOYTEALSA-N 0.000 description 1
- VNLZREKGKDYHSP-YGOYTEALSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC=CC=C3OC[C@@H]2C)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@@]2([H])C3=CC=CC=C3OC[C@@H]2C)=N1 VNLZREKGKDYHSP-YGOYTEALSA-N 0.000 description 1
- CYQCFCJBHXWBFK-ZWNOBZJWSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC(C)=C(C)C=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC(C)=C(C)C=C32)=N1 CYQCFCJBHXWBFK-ZWNOBZJWSA-N 0.000 description 1
- BQJACPPUSXKAHZ-PRHODGIISA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC(F)=C(C)C=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC(F)=C(C)C=C32)=N1 BQJACPPUSXKAHZ-PRHODGIISA-N 0.000 description 1
- ODUPNTHHJAJTEA-ZYHUDNBSSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C(C)=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C(C)=C32)=N1 ODUPNTHHJAJTEA-ZYHUDNBSSA-N 0.000 description 1
- XWKYBCPQSOQBTC-BXKDBHETSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(C)C=C32)=N1 XWKYBCPQSOQBTC-BXKDBHETSA-N 0.000 description 1
- ZADJLTSOJSBSKK-RDDDGLTNSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(F)C=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=C(F)C=C32)=N1 ZADJLTSOJSBSKK-RDDDGLTNSA-N 0.000 description 1
- DMHTZMUKTPELHM-LDYMZIIASA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=CC=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCC3=CC=CC=C32)=N1 DMHTZMUKTPELHM-LDYMZIIASA-N 0.000 description 1
- ITNMLCGSHMGFHC-BXUZGUMPSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCCC3=C(C)C=C(C)C=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCCC3=C(C)C=C(C)C=C32)=N1 ITNMLCGSHMGFHC-BXUZGUMPSA-N 0.000 description 1
- YVODLZWNLHIKID-ZWNOBZJWSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCCC3=CC=C(C)C=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCCC3=CC=C(C)C=C32)=N1 YVODLZWNLHIKID-ZWNOBZJWSA-N 0.000 description 1
- RGJNSAJDECMUHM-BXKDBHETSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCCC3=CC=CC=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCCC3=CC=CC=C32)=N1 RGJNSAJDECMUHM-BXKDBHETSA-N 0.000 description 1
- KDLDOSBKSFGNTF-ZYHUDNBSSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCOC3=C(C)C(C)=CC=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCOC3=C(C)C(C)=CC=C32)=N1 KDLDOSBKSFGNTF-ZYHUDNBSSA-N 0.000 description 1
- SUUVEMDWURGYSD-MWLCHTKSSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCOC3=CC=C(C)C=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCOC3=CC=C(C)C=C32)=N1 SUUVEMDWURGYSD-MWLCHTKSSA-N 0.000 description 1
- ODBBCCGHGWRMDS-PSASIEDQSA-N [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCOC3=CC=CC=C32)=N1 Chemical compound [H][C@](C)(F)C1=NC(N)=NC(N[C@]2([H])CCOC3=CC=CC=C32)=N1 ODBBCCGHGWRMDS-PSASIEDQSA-N 0.000 description 1
- HASKBXRBSOROHL-GXFFZTMASA-N [H][C@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C Chemical compound [H][C@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C HASKBXRBSOROHL-GXFFZTMASA-N 0.000 description 1
- BEVCALTXKNSIAZ-GXFFZTMASA-N [H][C@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)C2=CC=CC=C2CC[C@@H]1C Chemical compound [H][C@]1(NC2=NC(C(C)(C)F)=NC(N)=N2)C2=CC=CC=C2CC[C@@H]1C BEVCALTXKNSIAZ-GXFFZTMASA-N 0.000 description 1
- QIGRKUBXJFPSQH-WOPFXOFLSA-N [H][C@]1(NC2=NC(C(F)CC)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C Chemical compound [H][C@]1(NC2=NC(C(F)CC)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C QIGRKUBXJFPSQH-WOPFXOFLSA-N 0.000 description 1
- ATDWACRVTKJURQ-AXWJTWPOSA-N [H][C@]1(NC2=NC(C(F)CCC)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C Chemical compound [H][C@]1(NC2=NC(C(F)CCC)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C ATDWACRVTKJURQ-AXWJTWPOSA-N 0.000 description 1
- OFVCGBCCPNUKHQ-TVQRCGJNSA-N [H][C@]1(NC2=NC(C)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C Chemical compound [H][C@]1(NC2=NC(C)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C OFVCGBCCPNUKHQ-TVQRCGJNSA-N 0.000 description 1
- FJLRHDREFRAVAW-TVQRCGJNSA-N [H][C@]1(NC2=NC(C)=NC(N)=N2)C2=CC=CC=C2CC[C@@H]1C Chemical compound [H][C@]1(NC2=NC(C)=NC(N)=N2)C2=CC=CC=C2CC[C@@H]1C FJLRHDREFRAVAW-TVQRCGJNSA-N 0.000 description 1
- FKECSAQRVODFJX-IINYFYTJSA-N [H][C@]1(NC2=NC(CC)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C Chemical compound [H][C@]1(NC2=NC(CC)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C FKECSAQRVODFJX-IINYFYTJSA-N 0.000 description 1
- BWWQWIUFESVXDF-IINYFYTJSA-N [H][C@]1(NC2=NC(CC)=NC(N)=N2)C2=CC=CC=C2CC[C@@H]1C Chemical compound [H][C@]1(NC2=NC(CC)=NC(N)=N2)C2=CC=CC=C2CC[C@@H]1C BWWQWIUFESVXDF-IINYFYTJSA-N 0.000 description 1
- RIHGEWPQEYDNKC-XHDPSFHLSA-N [H][C@]1(NC2=NC(CCC)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C Chemical compound [H][C@]1(NC2=NC(CCC)=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C RIHGEWPQEYDNKC-XHDPSFHLSA-N 0.000 description 1
- ABFOFYVGUAQOCS-JOYOIKCWSA-N [H][C@]1(NC2=NC=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C Chemical compound [H][C@]1(NC2=NC=NC(N)=N2)C2=CC(C)=CC=C2C[C@@H]1C ABFOFYVGUAQOCS-JOYOIKCWSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/66—1,3,5-Triazines, not hydrogenated and not substituted at the ring nitrogen atoms
- A01N43/68—1,3,5-Triazines, not hydrogenated and not substituted at the ring nitrogen atoms with two or three nitrogen atoms directly attached to ring carbon atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N41/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a sulfur atom bound to a hetero atom
- A01N41/02—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a sulfur atom bound to a hetero atom containing a sulfur-to-oxygen double bond
- A01N41/10—Sulfones; Sulfoxides
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/04—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom
- A01N43/06—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings
- A01N43/12—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings condensed with a carbocyclic ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/54—1,3-Diazines; Hydrogenated 1,3-diazines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/56—1,2-Diazoles; Hydrogenated 1,2-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/80—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,2
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/28—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N<
- A01N47/36—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N< containing the group >N—CO—N< directly attached to at least one heterocyclic ring; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/28—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N<
- A01N47/38—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N< containing the group >N—CO—N< where at least one nitrogen atom is part of a heterocyclic ring; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N2300/00—Combinations or mixtures of active ingredients covered by classes A01N27/00 - A01N65/48 with other active or formulation relevant ingredients, e.g. specific carrier materials or surfactants, covered by classes A01N25/00 - A01N65/48
Definitions
- One possibility of improving the application profile of a herbicide can consist in combining the active compound with one or more other active compounds which contribute the desired additional properties.
- the combined use of a plurality of active compounds frequently causes phenomena of physical and biological incompatibility, for example a lack of stability in a coformulation, decomposition of an active compound, or antagonism of the active compounds.
- What is desired are, in contrast, combinations of active compounds having an advantageous activity profile, high stability and, if possible, an unexpected synergistically improved action, which allows the application rate to be reduced in comparison with the individual application of the active compounds to be combined.
- the synergistic effects allow the application rate to be reduced further, a broader spectrum of broad-leaved weeds and weed grasses to be controlled, a more rapid onset of the herbicidal action, a longer persistency, a better control of the harmful plants with only one or a few applications and a widening of the application period possible.
- the amount of harmful ingredients, such as nitrogen or oleic acid, and their introduction into the soil are also likewise reduced.
- the lower carbon skeletons for example those having 1 to 6 carbon atoms or, in the case of unsaturated groups, having 2 to 6 carbon atoms, are preferred for the hydrocarbon radicals, such as alkyl, alkenyl and alkynyl radicals, including in composite radicals.
- Preferred components (A) also include mixtures of the optically active compounds (A) mentioned above, preferably those having the same chemical constitution, which differ only in the stereochemical configuration. Preference is given, for example, to optically active mixtures of the compounds (A9)+(A11), (A21)+(A22), (A23)+(A24), (A28)+(A29), (A32)+(A33), where the ratios may be varied within a wide range.
- the application rate is preferably in the range of from 0.1 to 500 g of AS/ha, in particular in the range of from 0.2 to 250 g/ha.
- the application rate is preferably in the range of from 0.1 to 1000 g of AS/ha, in particular in the range of from 0.2 to 750 g/ha.
- the application rate is preferably in the range of from 0.1 to 500 g of AS/ha, in particular in the range of from 0.2 to 200 g/ha.
- the Table 2 below states preferred and especially preferred application rates for the individual active compounds (B).
- the combinations according to the invention can furthermore be used together with other active compounds, for example from the group of the herbicides, safeners, fungicides, insecticides and plant growth regulators, or from the group of the formulation auxiliaries and additives customary in crop protection.
- Emulsions for example oil-in-water emulsions (EW)
- EW oil-in-water emulsions
- Crop plants were grown in the open on plots under natural outdoor conditions, and seeds or rhizome pieces of typical harmful plants were laid out or the natural weed growth was utilized. Treatment with the compositions according to the invention was carried out after the harmful plants had emerged and the crop plants were, generally, at the 2- to 4-leaf stage; in some cases (as stated), application of individual active compounds or active compound combinations was carried out pre-emergence or as a sequential treatment partly pre-emergence and/or post-emergence.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Furan Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Pyridine Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Catching Or Destruction (AREA)
Abstract
- R1 is H or a group of the formula CZ1Z2Z3, where Z1, Z2 and Z3 are as defined in claim 1, R2 and R3 are each H, alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl having in each case up to 4 carbon atoms or acyl, R4 is H, (C1-C6)-alkyl or (C1-C6)-alkoxy;
- R5, R6, R7 and R8 are each H, (C1-C4)-alkyl, (C1-C3)-haloalkyl, halogen, (C1-C3)-alkoxy, (C1-C3)-haloalkoxy or cyano;
- A is CH2 or O or a direct bond, and
- the component (B) is one or more herbicides from the group of compounds consisting of
- (B1) thiencarbazone, tembotrione, SYN-523, pyroxsulam, penoxsulam, SYN-449,
- (B2) pyrasulfotole, trifloxysulfuron, saflufenacil, aminopyralid, ethofumesate, aminocyclopyrachlor and
- (B3) pyroxasulfone (KIH-485)
are suitable for controlling harmful plants or for regulating the growth of plants.
Description
-
- in which
- R1 is H or a group of the formula CZ1Z2Z3, where
- Z1 is H, halogen, (C1-C6)-alkyl, (C1-C6)-haloalkyl, [(C1-C4)-alkoxy]-(C1-C6)-alkyl, (C3-C6)-cycloalkyl which is unsubstituted or substituted by one or more radicals selected from the group consisting of halogen, (C1-C4)-alkyl and (C1-C4)-haloalkyl, or is (C2-C6)-alkenyl, (C2-C6)-alkynyl, (C2-C6)-haloalkenyl, (C4-C6)-cyclo-alkenyl, (C4-C6)-halocycloalkenyl, (C1-C6)-alkoxy or (C1-C6)-halo-alkoxy,
- Z2 is H, halogen, (C1-C6)-alkyl or (C1-C4)-alkoxy; or
- Z1 and Z2 together with the (shown) carbon atom of the group CZ1Z2Z3 are a (C3-C6)-cycloalkyl radical or (C4-C6)-cycloalkenyl radical, where each of the two last-mentioned radicals is unsubstituted or substituted by one or more radicals selected from the group consisting of (C1-C4)-alkyl, and
- Z3 is H, (C1-C6)-alkyl, (C1-C4)-alkoxy or halogen,
- R2 and R3 are each independently of one another H, (C1-C4)-alkyl, (C1-C4)-haloalkyl, (C3-C4)-alkenyl, (C3-C4)-haloalkenyl, (C3-C4)-alkynyl, (C3-C4)-haloalkynyl or an acyl radical.
- R4 is H, (C1-C6)-alkyl or (C1-C6)-alkoxy,
- R5, R6, R7 and R8 are each independently of one another H, (C1-C4)-alkyl, (C1-C3)-haloalkyl, halogen, (C1-C3)-alkoxy, (C1-C3)-haloalkoxy or cyano and
- A is a divalent group of the formula CH2 or O or a direct bond, and
- component (B) is one or more herbicidally active compounds (B) selected from the group consisting of the herbicidally active compounds (B1), (B2) and (B3), where the herbicidally active compounds are
- (B1) herbicidally active compounds particularly suitable for post-emergence application against monocotyledonous or dicotyledonous harmful plants, selected from the group consisting of
- (B1.1) thiencarbazone and its esters and salts,
- (B1.2) tembotrione and its salts,
- (B1.3) ethyl [[3-[2-chloro-5-[3,6-dihydro-3-methyl-2,6-dioxo-4-(trifluoromethyl)-1(2H)-pyrimidinyl]-4-fluorophenoxy]-2-pyridinyl]oxy]acetate (SYN-523),
- (B1.4) pyroxsulam and its salts,
- (B1.5) penoxsulam and its salts,
- (B1.6) 4-hydroxy-3-[[2-[(2-methoxyethoxy)methyl]-6-trifluoromethyl-3-pyridinyl]carbonyl]bicyclo[3.2.1]oct-3-en-2-one (SYN-449) and its salts,
- (B2) herbicidally active compounds particularly suitable for post-emergence application against dicotyledonous harmful plants, selected from the group consisting of
- (B2.1) pyrasulfotole and its salts,
- (B2.2) trifloxysulfuron and its salts,
- (B2.3) saflufenacil and its salts,
- (B2.4) aminopyralid and its salts,
- (B2.5) ethofumesate,
- (B2.6) aminocyclopyrachlor and its salts and esters and
- (B3) herbicidally active compounds particularly suitable for pre-emergence application against monocotyledonous or dicotyledonous harmful plants, selected from the group consisting of
- (B3.1) pyroxasulfone (KIH-485).
- R1 is H or a group of the formula CZ1Z2Z3 in which
- Z1 is H, halogen, (C1-C4)-alkyl, (C1-C4)-haloalkyl, [(C1-C4)-alkoxy]-(C1-C6)-alkyl or (C3-C6)-cycloalkyl which is unsubstituted or substituted by one or more radicals selected from the group consisting of (C1-C4)-alkyl, or is (C2-C4)-alkenyl, (C2-C4)-alkynyl, (C1-C4)-alkoxy or (C1-C4)-haloalkoxy;
- Z2 is H, halogen, (C1-C4)-alkyl or
- Z1 and Z2 together with the carbon atom attached to the radicals are a (C3-C6)-cycloalkyl radical and
- Z3 is H, (C1-C4)-alkyl, (C1-C2)-alkoxy or halogen,
- R2 is H, (C1-C4)-alkyl, (C1-C4)-haloalkyl, (C3-C4)-alkenyl, (C3-C4)-haloalkenyl, (C3-C4)-alkynyl, (C3-C4)-haloalkynyl or an acyl radical having 1 to 12 carbon atoms, wherein an acyl radical preferably is a acyl radical selected from the group consisting of formyl, phenylcarbonyl, phenoxycarbonyl, where phenyl in the two last-mentioned radicals is unsubstituted or substituted by one or more radicals from the group consisting of halogen, (C1-C2)-alkyl, (C1-C2)-haloalkyl, (C1-C2)-alkoxy, (C1-C2)-haloalkoxy and nitro, and (C1-C6)-alkyl-carbonyl, (C1-C6)-alkoxy-carbonyl and (C1-C6)-alkyl-sulfonyl,
- R3 is H, (C1-C4)-alkyl or (C1-C4)-haloalkyl,
- R4 is H, (C1-C3)-alkyl or (C1-C3)-alkoxy,
- R5, R6, R7 and R8 are each independently of one another H, (C1-C3)-alkyl, halogen, (C1-C3)-alkoxy, and
- A is a divalent group of the formula CH2 or O or a direct bond, preferably CH2 or a direct bond, in particular a direct bond.
- 1. priority is given to substituted NH; 2. priority is given to the bond to the phenyl ring; 3. priority is given to the other carbon ring atom, 4. priority is given to the hydrogen atom.
| TABLE 1 |
| Compounds of the formula (I) (herbicide (A)): |
| Com- | |
| pound | |
| No. | Chemical formula or chemical name |
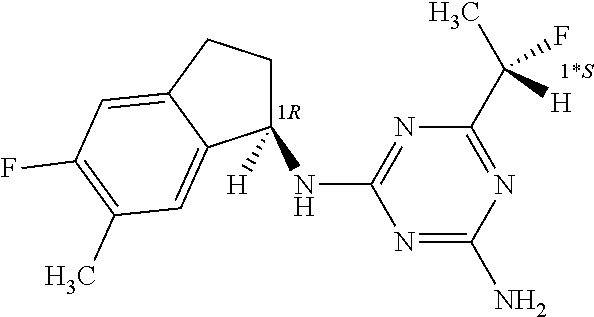
| A1 |
|
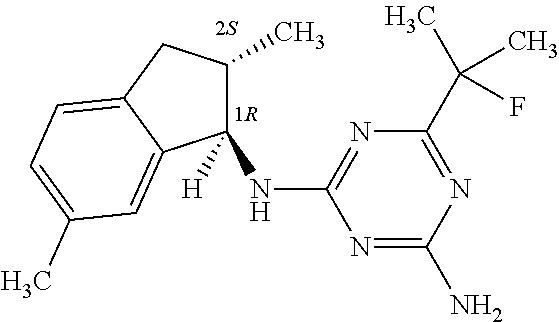
| A2 |
|
| A3 |
|
| A4 |
|
| A5 |
|
| A6 |
|
| A7 |
|
| A8 |
|
| A9 |
|
| A10 |
|
| A11 |
|
| A12 |
|
| A13 |
|
| A14 |
|
| A15 |
|
| A16 |
|
| A17 |
|
| A18 |
|
| A19 |
|
| A20 |
|
| A21 |
|
| A22 |
|
| A23 |
|
| A24 |
|
| A25 |
|
| A26 |
|
| A27 |
|
| A28 |
|
| A29 |
|
| A30 |
|
| A31 |
|
| A32 |
|
| A33 |
|
| A34 |
|
| A35 |
|
| A36 |
|
| A37 |
|
| A38 |
|
| A39 |
|
| A40 |
|
| A41 |
|
| A42 |
|
| A43 |
|
| A44 |
|
| A45 |
|
| A46 |
|
| A47 |
|
| A48 |
|
| A49 |
|
| A50 |
|
| A51 |
|
| A52 |
|
| A53 |
|
| A54 |
|
| A55 |
|
| A56 |
|
| A57 |
|
| A58 |
|
| A59 |
|
| A60 |
|
| A61 |
|
| A62 |
|
| A63 |
|
| A64 |
|
| A65 |
|
| A66 |
|
| A67 |
|
| A68 |
|
| A69 |
|
| A70 |
|
| A71 |
|
| A72 |
|
| A73 |
|
| A74 |
|
| A75 |
|
| A76 |
|
| A77 |
|
| A78 |
|
| A79 |
|
| A80 |
|
| A81 |
|
| A82 |
|
| A83 |
|
| A84 |
|
| A85 |
|
| A86 |
|
| A87 |
|
| A88 |
|
| A89 |
|
| A90 |
|
| A91 |
|
| A92 |
|
| A93 |
|
| A94 |
|
| A95 |
|
| A96 |
|
| A97 |
|
| A98 |
|
| A99 |
|
| A100 |
|
| A101 |
|
| A102 |
|
| A103 |
|
| A104 |
|
| A105 |
|
| A106 |
|
| A107 |
|
| A108 |
|
| A109 |
|
| A110 |
|
| A111 |
|
| A112 |
|
| A113 |
|
| A114 |
|
| A115 |
|
| A116 |
|
| A117 |
|
| A118 |
|
| A119 |
|
| A120 |
|
| A121 |
|
| A122 |
|
| A123 |
|
| A124 |
|
| A125 |
|
| A126 |
|
| A127 |
|
- (B1) herbicidally active compounds which are particularly suitable for post-emergence application against monocotyledonous or dicotyledonous harmful plants, selected from the group consisting of
- (B1.1) thiencarbazone and its esters and salts (WO 01/05788), in particular
- (B1.1.1) thiencarbazone, i.e. the chemical compound 4-[(4,5-dihydro-3-methoxy-4-methyl-5-oxo-1H-1,2,4-triazol-1-yl)carbonyl-sulfamoyl]-5-methylthiophene-3-carboxylic acid of the formula:
- (B1.1) thiencarbazone and its esters and salts (WO 01/05788), in particular
-
-
- (B1.1.2) thiencarbazone-methyl, i.e. the chemical compound methyl 4-[(4,5-dihydro-3-methoxy-4-methyl-5-oxo-1H-1,2,4-triazol-1-yl)carbonylsulfamoyl]-5-methylthiophene-3-carboxylate [CAS-Reg. 317815-83-1] or
- (B1.1.3) thiencarbazone-methyl sodium salt, i.e. the compound methyl 4-[(4,5-dihydro-3-methoxy-4-methyl-5-oxo-1H-1,2,4-triazol-1-yl)carbonylsulfamoyl]-5-methylthiophene-3-carboxylate sodium, i.e. the acidic hydrogen atom at the sulfonamide group in thiencarbazone-methyl has been replaced by a sodium atom,
- (B1.2) tembotrione and its salts, for example its alkali metal salts, such as sodium or potassium salts (see WO-A-00/21924), in particular
- (B1.2.1) tembotrione, i.e. the chemical compound 2-{2-chloro-4-mesyl-3-[(2,2,2-trifluoroethoxy)methyl]benzoyl}cyclohexane-1,3-dione [CAS-Reg. 335104-84-2] of the formula (the formula only shows the triketo form, which is generally in an equilibrium with a plurality of possible enol forms):
-
-
- (B1.3) ethyl [[3-[2-chloro-5-[3,6-dihydro-3-methyl-2,6-dioxo-4-(trifluoromethyl)-1(2H)-pyrimidinyl]-4-fluorophenoxy]-2-pyridinyl]oxy]acetate (herein also referred to as SYN-523) (WO 2006/061562, EP 1122244) of the formula
-
- (B1.4) pyroxsulam and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
- (B1.4.1) pyroxsulam, i.e. the chemical compound N-(5,7-dimethoxy[1,2,4]triazolo[1,5-a]pyrimidin-2-yl)-2-methoxy-4-(trifluoromethyl)pyridine-3-sulfonamide [CAS-Reg. 422556-08-9] of the formula:
- (B1.4) pyroxsulam and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
-
- (B1.5) penoxsulam and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
- (B1.5.1) penoxsulam, i.e. the chemical compound 2-(2,2-difluoroethoxy)-N-(5,8-dimethoxy[1,2,4]triazolo [1,5-c]pyrimidin-2-yl)-6-(trifluoromethyl)benzene-sulfonamide [CAS-Reg. 219714-96-2] of the formula:
- (B1.5) penoxsulam and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
-
-
- and
- (B1.6) 4-hydroxy-3-[[2-[(2-methoxyethoxy)methyl]-6-trifluoromethyl-3-pyridinyl]carbonyl]bicyclo[3.2.1]oct-3-en-2-one and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
- (B1.6.1) SYN-449, i.e. the chemical compound 4-hydroxy-3-[[2-[(2-methoxyethoxy)methyl]-6-trifluoromethyl-3-pyridinyl]carbonyl]-bicyclo[3.2.1]oct-3-en-2-one (CAS RN 352010-68-5, SYN-449, WO-A-2006/097322, WO-A-01/94339) of the formula (the formula shows only one of the possible enol forms which are generally in an equilibrium with one another and with the keto form=the triketone):
-
- (B2) herbicidally active compounds which are particularly suitable for post-emergence application against dicotyledonous harmful plants, selected from the group consisting of
- (B2.1) pyrasulfotole and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
- (B2.1.1) pyrasulfotole, i.e. the chemical compound (5-hydroxy-1,3-dimethyl-1H-pyrazol-4-yl)-[2-(methylsulfonyl)-4-(trifluoromethyl)-phenyl]methanone [CAS-Reg. 365400-11-9] of the formula:
- (B2.1) pyrasulfotole and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
-
- (B2.2) trifloxysulfuron and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
- (B2.2.1) trifloxysulfuron, i.e. the chemical compound 1-(4,6-dimethoxypyrimidin-2-yl)-3-[3-(2,2,2-trifluoroethoxy)-2-pyridylsulfonyl]urea of the formula:
- (B2.2) trifloxysulfuron and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
-
-
- or, preferably.
- (B2.2.2) trifloxysulfuron-sodium, i.e. the sodium salt of trifloxysulfuron in which the acidic hydrogen atom at the sulfonamide group has been replaced by a sodium atom;
- (B2.3) saflufenacil and its salts, for example its alkali metal salts, such as sodium or potassium salts, in particular
- (B2.3.1) saflufenacil (herein also referred to as BAS-H800), i.e. the chemical compound 2-chloro-5-[3,6-dihydro-3-methyl-2,6-dioxo-4-(trifluoromethyl)-1 (2H)-pyrimidinyl]-4-fluoro-N-[[methyl(1-methylethyl)amino]sulfonyl]benzamide [CAS-Reg. 372137-35-4] (WO 2001/083459) of the formula:
-
-
- (B2.4) aminopyralid and its salts, for example its alkali metal salts, such as sodium or potassium salts, or ammonium salts or else acid addition salts (for example hydrochlorides), in particular
- (B2.4.1) aminopyralid, i.e. the chemical compound 4-amino-3,6-dichloropyridine-2-carboxylic acid [CAS-Reg. 150114-71-9] of the formula:
- (B2.4) aminopyralid and its salts, for example its alkali metal salts, such as sodium or potassium salts, or ammonium salts or else acid addition salts (for example hydrochlorides), in particular
-
-
- and
- (B2.5) ethofumesate, i.e. the chemical compound (RS)—O-(2-ethoxy-2,3-dihydro-3,3-dimethylbenzofuran-5-yl)methanesulfonate [CAS-Reg. 26225-79-6] of the formula:
-
-
- (B2.6) aminocyclopyrachlor and its salts and esters, for example its alkali metal salts, such as sodium or potassium salts, and its alkyl esters, such as (C1-C4)-alkyl ester, preferably
- (B2.6.1) aminocyclopyrachlor, i.e. the chemical compound 6-amino-5-chloro-2-cyclopropylpyrimidine-4-carboxylic acid [CAS-Reg. 858958-08-8] of the formula:
- (B2.6) aminocyclopyrachlor and its salts and esters, for example its alkali metal salts, such as sodium or potassium salts, and its alkyl esters, such as (C1-C4)-alkyl ester, preferably
-
-
- or preferably also
- (B2.6.2) aminocyclopyrachlor potassium salt or
- (B2.6.3) aminocyclopyrachlor sodium salt or
- (B2.6.4) aminocyclopyrachlor methyl ester and
- (B3) herbicidally active compounds which are particularly suitable for pre-emergence application against monocotyledonous or dicotyledonous harmful plants, selected from the group consisting of
- (B3.1) pyroxasulfone (KIH-485), i.e. the chemical compound 3-[5-(difluoromethoxy)-1-methyl-3-(trifluoromethyl)pyrazol-4-ylmethylsulfonyl]-4,5-dihydro-5,5-dimethyl-1,2-oxazole [CAS-Reg. 447399-55-5] of the formula:
-
- (B1.1) thiencarbazone and its esters and salts belong to the class of chemical compounds of the triazolone herbicides and are inhibitors of acetolactate synthase in plants (ALS inhibitors).
- (B1.2) tembotrione and its salts belong to the class of chemical compounds of the benzoylcyclohexanediones and are inhibitors of hydroxyphenylpyruvate dioxygenase in plants (HPPD inhibitors).
- (B1.3) SYN-523 belongs to the class of chemical compounds of the uracils (pyrimidinediones) and is an inhibitor of protoporphyrinogen oxidase in plants (PPO inhibitor).
- (B1.4) pyroxsulam and (B1.5) penoxsulam and their salts belong to the class of chemical compounds of the triazolopyrimidines and are inhibitors of acetolactate synthase in plants (ALS inhibitors).
- (B1.6) SYN-449 and its salts belong to the class of chemical compounds of the (bridged) benzoylcyclohexanediones and are inhibitors of hydroxyphenylpyruvate dioxygenase in plants (HPPD inhibitors).
- (B2.1) pyrasulfotole and its salts belong to the class of chemical compounds of the benzoylpyrazoles, which are related to the benzoylcyclohexanediones, and are inhibitors of hydroxyphenylpyruvate dioxygenase in plants (HPPD inhibitors).
- (B2.2) trifloxysulfuron and its salts belong to the class of chemical compounds of the pyridylsulfonylureas and are inhibitors of acetolactate synthase in plants (ALS inhibitors).
- (B2.3) saflufenacil and its salts belong to the class of chemical compounds of the uracils (pyrimidinediones) and are inhibitors of protoporphyrinogen oxidase in plants (PPO inhibitors).
- (B2.4) aminopyralid and its salts belong to the class of chemical compounds of the pyridylcarboxylic acids (picolinic acids) and are synthetic auxins in plants.
- (B2.5) ethofumesate belongs to the class of chemical compounds of the benzofuranylmethanesulfonates and is an inhibitor of lipid synthesis in plants.
- (B2.6) aminocyclopyrachlor and its salts or esters belong to the chemical class of the pyrimidinecarboxylic acids and are synthetic auxins in plants.
- (B3.1) pyroxasulfone belongs to the class of chemical compounds of the oxazolinesulfonylmethylpyrazoles and is an inhibitor of the synthesis of long-chain fatty acids (inhibitor of very long chain fatty acid (VLCFA) elongases) in plants.
- (A):(B1) is preferably in the range of from 1:50000 to 2000:1, in particular from 1:13000 to 750:1;
- (A):(B2) is preferably in the range of from 1:100000 to 2000:1, in particular from 1:40000 to 750:1;
- (A):(B3) is preferably in the range of from 1:50000 to 2000:1, in particular from 1:18000 to 75:1.
| TABLE 2 |
| Codes b for the herbicide component (B), preferred application rates |
| with respect to (B) and preferred ratios (A):(B) |
| Application | ||||
| Code | Comp. | rate | Weight ratio | |
| b | (B) | Short name | [g of AS/ha] | (A):(B) |
| 1 | (B1.1) | thiencarbazone | 0.1 to 200 | 1:20000 to 2000:1 |
| and its esters and | (0.2 to 100) | (1:10000 to 750:1) | ||
| salts | ||||
| 2 | (B1.1.1) | thiencarbazone | 0.1 to 200 | 1:20000 to 2000:1 |
| (0.2 to 100) | (1:10000 to 750:1) | |||
| 3 | (B1.1.2) | thiencarbazone- | 0.1 to 200 | 1:20000 to 2000:1 |
| methyl | (0.2 to 100) | (1:10000 to 750:1) | ||
| 4 | (B1.1.3) | thiencarbazone- | 0.1 to 200 | 1:20000 to 2000:1 |
| methyl-sodium | (0.2 to 100) | (1:10000 to 750:1) | ||
| 5 | (B1.2) | tembotrione and | 0.1 to 500 | 1:50000 to 200:1 |
| its salts | (2 to 200) | (1:10000 to 75:1) | ||
| 6 | (B1.2.1) | tembotrione | 0.1 to 500 | 1:50000 to 200:1 |
| (2 to 200) | (1:10000 to 75:1) | |||
| 7 | (B1.3) | SYN-523 | 0.1 to 500 | 1:50000 to 20000:1 |
| (2 to 200) | (1:13000 to 750:1) | |||
| 8 | (B1.4) | pyroxsulam and | 1 to 200 | 1:20000 to 200:1 |
| its salts | (2 to 150) | (1:7500 to 75:1) | ||
| 9 | (B1.4.1) | pyroxsulam | 1 to 200 | 1:20000 to 200:1 |
| (2 to 150) | (1:7500 to 75:1) | |||
| 10 | (B1.5) | penoxsulam and | 1 to 200 | 1:20000 to 200:1 |
| its salts | (2 to 150) | (1:7500 to 75:1) | ||
| 11 | (B1.5.1) | penoxsulam | 1 to 200 | 1:20000 to 200:1 |
| (2 to 150) | (1:7500 to 75:1) | |||
| 12 | (B1.6) | SYN-449 and its | 1 to 200 | 1:20000 to 200:1 |
| salts | (2 to 150) | (1:7500 to 75:1) | ||
| 13 | (B1.6.1) | SYN-449 | 1 to 200 | 1:20000 to 200:1 |
| (2 to 150) | (1:7500 to 75:1) | |||
| 14 | (B2.1) | pyrasulfotole and | 1 to 300 | 1:30000 to 200:1 |
| its salts | (2 to 150) | (1:10000 to 30:1) | ||
| 15 | (B2.1.1) | pyrasulfotole | 1 to 300 | 1:30000 to 200:1 |
| (2 to 150) | (1:10000 to 30:1) | |||
| 16 | (B2.2) | trifloxysulfuron | 0.1 to 300 | 1:30000 to 2000:1 |
| and its salts | (0.2 to 150) | (1:7500 to 300:1) | ||
| 17 | (B2.2.1) | trifloxysulfuron | 0.1 to 300 | 1:30000 to 2000:1 |
| (0.2 to 150) | (1:7500 to 300:1) | |||
| 18 | (B2.2.2) | trifloxysulfuron- | 0.1 to 300 | 1:30000 to 2000:1 |
| sodium salt | (0.2 to 150) | (1:7500 to 300:1) | ||
| 19 | (B2.3) | saflufenacil and | 0.1 to 300 | 1:30000 to 2000:1 |
| its salts | (0.2 to 250) | (1:12500 to 750:1) | ||
| 20 | (B2.3.1) | saflufenacil | 0.1 to 300 | 1:30000 to 2000:1 |
| (0.2 to 250) | (1:12500 to 750:1) | |||
| 21 | (B2.4) | aminopyralid and | 0.1 to 300 | 1:30000 to 2000:1 |
| its salts | (0.2 to 250) | (1:12500 to 750:1) | ||
| 22 | (B2.4.1) | aminopyralid | 0.1 to 300 | 1:30000 to 2000:1 |
| (0.2 to 250) | (1:12500 to 750:1) | |||
| 23 | (B2.5) | ethofumesate | 1 to 1000 | 1:100000 to 200:1 |
| (2 to 750) | (1:37500 to 75:1) | |||
| 24 | (B2.6) | aminocyclopyra- | 1 to 1000 | 1:75000 to 200:1 |
| chlor and its salts | (2 to 350) | (1:25000 to 75:1) | ||
| and esters | ||||
| 25 | (B2.6.1) | aminocyclopyra- | 1 to 1000 | 1:75000 to 200:1 |
| chlor | (2 to 350) | (1:25000 to 75:1) | ||
| 26 | (B2.6.2) | aminocyclopyra- | 1 to 1000 | 1:75000 to 200:1 |
| chlor potassium | (2 to 350) | (1:25000 to 75:1) | ||
| salt | ||||
| 27 | (B2.6.3) | aminocyclopyra- | 1 to 1000 | 1:75000 to 200:1 |
| chlor sodium salt | (2 to 350) | (1:25000 to 75:1) | ||
| 28 | (B2.6.4) | aminocyclopyra- | 1 to 1000 | 1:75000 to 200:1 |
| chlo rmethyl ester | (2 to 350) | (1:25000 to 75:1) | ||
| 29 | (B3.1) | pyroxasulfone | 0.1 to 500 | 1:50000 to 200:1 |
| (0.2 to 200) | (1:17500 to 75:1) | |||
| Abbreviations for Table 2: | ||||
| Comp. = component, active compound | ||||
| AS = active substance (based on 100% active compound) | ||||
| (A):(B) = ratio of the active compounds (A):(B) [(A) = Comp. (I) or preferably according to Tab. 1; (B) = Comp. according to Tab. 2] | ||||
| The columns “Application rates” and “Weight ratios” in Table 2 contain in each case preferred and particularly preferred application rates (the latter in brackets) and “Weight ratios (A):(B)” based on the active compound group (B) or the active compound according to code b. | ||||
| TABLE 3 |
| Binary herbicide combinations of the active compounds (A) and (B) |
| K1.1, K1.2, K1.3, K1.4, K1.5, K1.6, K1.7, K1.8, K1.9, K1.10, K1.11, |
| K1.12, K1.13, K1.14, K1.15, K1.16, K1.17, K1.18, K1.19, K1.20, K1.21, |
| K1.22, K1.23, K1.24, K1.25, K1.26, K1.27, K1.28, K1.29, |
| K2.1, K2.2, K2.3, K2.4, K2.5, K2.6, K2.7, K2.8, K2.9, K2.10, K2.11, |
| K2.12, K2.13, K2.14, K2.15, K2.16, K2.17, K2.18, K2.19, K2.20, K2.21, |
| K2.22, K2.23, K2.24, K2.25, K2.26, K2.27, K2.28, K2.29, |
| K3.1, K3.2, K3.3, K3.4, K3.5, K3.6, K3.7, K3.8, K3.9, K3.10, K3.11, |
| K3.12, K3.13, K3.14, K3.15, K3.16, K3.17, K3.18, K3.19, K3.20, K3.21, |
| K3.22, K3.23, K3.24, K3.25, K3.26, K3.27, K3.28, K3.29, |
| K4.1, K4.2, K4.3, K4.4, K4.5, K4.6, K4.7, K4.8, K4.9, K4.10, K4.11, |
| K4.12, K4.13, K4.14, K4.15, K4.16, K4.17, K4.18, K4.19, K4.20, K4.21, |
| K4.22, K4.23, K4.24, K4.25, K4.26, K4.27, K4.28, K4.29, |
| K5.1, K5.2, K5.3, K5.4, K5.5, K5.6, K5.7, K5.8, K5.9, K5.10, K5.11, |
| K5.12, K5.13, K5.14, K5.15, K5.16, K5.17, K5.18, K5.19, K5.20, K5.21, |
| K5.22, K5.23, K5.24, K5.25, K5.26, K5.27, K5.28, K5.29, |
| K6.1, K6.2, K6.3, K6.4, K6.5, K6.6, K6.7, K6.8, K6.9, K6.10, K6.11, |
| K6.12, K6.13, K6.14, K6.15, K6.16, K6.17, K6.18, K6.19, K6.20, K6.21, |
| K6.22, K6.23, K6.24, K6.25, K6.26, K6.27, K6.28, K6.29, |
| K7.1, K7.2, K7.3, K7.4, K7.5, K7.6, K7.7, K7.8, K7.9, K7.10, K7.11, |
| K7.12, K7.13, K7.14, K7.15, K7.16, K7.17, K7.18, K7.19, K7.20, K7.21, |
| K7.22, K7.23, K7.24, K7.25, K7.26, K7.27, K7.28, K7.29, |
| K8.1, K8.2, K8.3, K8.4, K8.5, K8.6, K8.7, K8.8, K8.9, K8.10, K8.11, |
| K8.12, K8.13, K8.14, K8.15, K8.16, K8.17, K8.18, K8.19, K8.20, K8.21, |
| K8.22, K8.23, K8.24, K8.25, K8.26, K8.27, K8.28, K8.29, |
| K9.1, K9.2, K9.3, K9.4, K9.5, K9.6, K9.7, K9.8, K9.9, K9.10, K9.11, |
| K9.12, K9.13, K9.14, K9.15, K9.16, K9.17, K9.18, K9.19, K9.20, K9.21, |
| K9.22, K9.23, K9.24, K9.25, K9.26, K9.27, K9.28, K9.29, |
| K10.1, K10.2, K10.3, K10.4, K10.5, K10.6, K10.7, K10.8, K10.9, K10.10, |
| K10.11, K10.12, K10.13, K10.14, K10.15, K10.16, K10.17, K10.18, |
| K10.19, K10.20, K10.21, K10.22, K10.23, K10.24, K10.25, K10.26, |
| K10.27, K10.28, K10.29, |
| K11.1, K11.2, K11.3, K11.4, K11.5, K11.6, K11.7, K11.8, K11.9, K11.10, |
| K11.11, K11.12, K11.13, K11.14, K11.15, K11.16, K11.17, K11.18, |
| K11.19, K11.20, K11.21, K11.22, K11.23, K11.24, K11.25, K11.26, |
| K11.27, K11.28, K11.29, |
| K12.1, K12.2, K12.3, K12.4, K12.5, K12.6, K12.7, K12.8, K12.9, K12.10, |
| K12.11, K12.12, K12.13, K12.14, K12.15, K12.16, K12.17, K12.18, |
| K12.19, K12.20, K12.21, K12.22, K12.23, K12.24, K12.25, K12.26, |
| K12.27, K12.28, K12.29, |
| K13.1, K13.2, K13.3, K13.4, K13.5, K13.6, K13.7, K13.8, K13.9, K13.10, |
| K13.11, K13.12, K13.13, K13.14, K13.15, K13.16, K13.17, K13.18, |
| K13.19, K13.20, K13.21, K13.22, K13.23, K13.24, K13.25, K13.26, |
| K13.27, K13.28, K13.29, |
| K14.1, K14.2, K14.3, K14.4, K14.5, K14.6, K14.7, K14.8, K14.9, K14.10, |
| K14.11, K14.12, K14.13, K14.14, K14.15, K14.16, K14.17, K14.18, |
| K14.19, K14.20, K14.21, K14.22, K14.23, K14.24, K14.25, K14.26, |
| K14.27, K14.28, K14.29, |
| K15.1, K15.2, K15.3, K15.4, K15.5, K15.6, K15.7, K15.8, K15.9, K15.10, |
| K15.11, K15.12, K15.13, K15.14, K15.15, K15.16, K15.17, K15.18, |
| K15.19, K15.20, K15.21, K15.22, K15.23, K15.24, K15.25, K15.26, |
| K15.27, K15.28, K15.29, |
| K16.1, K16.2, K16.3, K16.4, K16.5, K16.6, K16.7, K16.8, K16.9, K16.10, |
| K16.11, K16.12, K16.13, K16.14, K16.15, K16.16, K16.17, K16.18, |
| K16.19, K16.20, K16.21, K16.22, K16.23, K16.24, K16.25, K16.26, |
| K16.27, K16.28, K16.29, |
| K17.1, K17.2, K17.3, K17.4, K17.5, K17.6, K17.7, K17.8, K17.9, K17.10, |
| K17.11, K17.12, K17.13, K17.14, K17.15, K17.16, K17.17, K17.18, |
| K17.19, K17.20, K17.21, K17.22, K17.23, K17.24, K17.25, K17.26, |
| K17.27, K17.28, K17.29, |
| K18.1, K18.2, K18.3, K18.4, K18.5, K18.6, K18.7, K18.8, K18.9, K18.10, |
| K18.11, K18.12, K18.13, K18.14, K18.15, K18.16, K18.17, K18.18, |
| K18.19, K18.20, K18.21, K18.22, K18.23, K18.24, K18.25, K18.26, |
| K18.27, K18.28, K18.29, |
| K19.1, K19.2, K19.3, K19.4, K19.5, K19.6, K19.7, K19.8, K19.9, K19.10, |
| K19.11, K19.12, K19.13, K19.14, K19.15, K19.16, K19.17, K19.18, |
| K19.19, K19.20, K19.21, K19.22, K19.23, K19.24, K19.25, K19.26, |
| K19.27, K19.28, K19.29, |
| K20.1, K20.2, K20.3, K20.4, K20.5, K20.6, K20.7, K20.8, K20.9, K20.10, |
| K20.11, K20.12, K20.13, K20.14, K20.15, K20.16, K20.17, K20.18, |
| K20.19, K20.20, K20.21, K20.22, K20.23, K20.24, K20.25, K20.26, |
| K20.27, K20.28, K20.29, |
| K21.1, K21.2, K21.3, K21.4, K21.5, K21.6, K21.7, K21.8, K21.9, K21.10, |
| K21.11, K21.12, K21.13, K21.14, K21.15, K21.16, K21.17, K21.18, |
| K21.19, K21.20, K21.21, K21.22, K21.23, K21.24, K21.25, K21.26, |
| K21.27, K21.28, K21.29, |
| K22.1, K22.2, K22.3, K22.4, K22.5, K22.6, K22.7, K22.8, K22.9, K22.10, |
| K22.11, K22.12, K22.13, K22.14, K22.15, K22.16, K22.17, K22.18, |
| K22.19, K22.20, K22.21, K22.22, K22.23, K22.24, K22.25, K22.26, |
| K22.27, K22.28, K22.29, |
| K23.1, K23.2, K23.3, K23.4, K23.5, K23.6, K23.7, K23.8, K23.9, K23.10, |
| K23.11, K23.12, K23.13, K23.14, K23.15, K23.16, K23.17, K23.18, |
| K23.19, K23.20, K23.21, K23.22, K23.23, K23.24, K23.25, K23.26, |
| K23.27, K23.28, K23.29, |
| K24.1, K24.2, K24.3, K24.4, K24.5, K24.6, K24.7, K24.8, K24.9, K24.10, |
| K24.11, K24.12, K24.13, K24.14, K24.15, K24.16, K24.17, K24.18, |
| K24.19, K24.20, K24.21, K24.22, K24.23, K24.24, K24.25, K24.26, |
| K24.27, K24.28, K24.29, |
| K25.1, K25.2, K25.3, K25.4, K25.5, K25.6, K25.7, K25.8, K25.9, K25.10, |
| K25.11, K25.12, K25.13, K25.14, K25.15, K25.16, K25.17, K25.18, |
| K25.19, K25.20, K25.21, K25.22, K25.23, K25.24, K25.25, K25.26, |
| K25.27, K25.28, K25.29, |
| K26.1, K26.2, K26.3, K26.4, K26.5, K26.6, K26.7, K26.8, K26.9, K26.10, |
| K26.11, K26.12, K26.13, K26.14, K26.15, K26.16, K26.17, K26.18, |
| K26.19, K26.20, K26.21, K26.22, K26.23, K26.24, K26.25, K26.26, |
| K26.27, K26.28, K26.29, |
| K27.1, K27.2, K27.3, K27.4, K27.5, K27.6, K27.7, K27.8, K27.9, K27.10, |
| K27.11, K27.12, K27.13, K27.14, K27.15, K27.16, K27.17, K27.18, |
| K27.19, K27.20, K27.21, K27.22, K27.23, K27.24, K27.25, K27.26, |
| K27.27, K27.28, K27.29, |
| K28.1, K28.2, K28.3, K28.4, K28.5, K28.6, K28.7, K28.8, K28.9, K28.10, |
| K28.11, K28.12, K28.13, K28.14, K28.15, K28.16, K28.17, K28.18, |
| K28.19, K28.20, K28.21, K28.22, K28.23, K28.24, K28.25, K28.26, |
| K28.27, K28.28, K28.29, |
| K29.1, K29.2, K29.3, K29.4, K29.5, K29.6, K29.7, K29.8, K29.9, K29.10, |
| K29.11, K29.12, K29.13, K29.14, K29.15, K29.16, K29.17, K29.18, |
| K29.19, K29.20, K29.21, K29.22, K29.23, K29.24, K29.25, K29.26, |
| K29.27, K29.28, K29.29, |
| K30.1, K30.2, K30.3, K30.4, K30.5, K30.6, K30.7, K30.8, K30.9, K30.10, |
| K30.11, K30.12, K30.13, K30.14, K30.15, K30.16, K30.17, K30.18, |
| K30.19, K30.20, K30.21, K30.22, K30.23, K30.24, K30.25, K30.26, |
| K30.27, K30.28, K30.29, |
| K31.1, K31.2, K31.3, K31.4, K31.5, K31.6, K31.7, K31.8, K31.9, K31.10, |
| K31.11, K31.12, K31.13, K31.14, K31.15, K31.16, K31.17, K31.18, |
| K31.19, K31.20, K31.21, K31.22, K31.23, K31.24, K31.25, K31.26, |
| K31.27, K31.28, K31.29, |
| K32.1, K32.2, K32.3, K32.4, K32.5, K32.6, K32.7, K32.8, K32.9, K32.10, |
| K32.11, K32.12, K32.13, K32.14, K32.15, K32.16, K32.17, K32.18, |
| K32.19, K32.20, K32.21, K32.22, K32.23, K32.24, K32.25, K32.26, |
| K32.27, K32.28, K32.29, |
| K33.1, K33.2, K33.3, K33.4, K33.5, K33.6, K33.7, K33.8, K33.9, K33.10, |
| K33.11, K33.12, K33.13, K33.14, K33.15, K33.16, K33.17, K33.18, |
| K33.19, K33.20, K33.21, K33.22, K33.23, K33.24, K33.25, K33.26, |
| K33.27, K33.28, K33.29, |
| K34.1, K34.2, K34.3, K34.4, K34.5, K34.6, K34.7, K34.8, K34.9, K34.10, |
| K34.11, K34.12, K34.13, K34.14, K34.15, K34.16, K34.17, K34.18, |
| K34.19, K34.20, K34.21, K34.22, K34.23, K34.24, K34.25, K34.26, |
| K34.27, K34.28, K34.29, |
| K35.1, K35.2, K35.3, K35.4, K35.5, K35.6, K35.7, K35.8, K35.9, K35.10, |
| K35.11, K35.12, K35.13, K35.14, K35.15, K35.16, K35.17, K35.18, |
| K35.19, K35.20, K35.21, K35.22, K35.23, K35.24, K35.25, K35.26, |
| K35.27, K35.28, K35.29, |
| K36.1, K36.2, K36.3, K36.4, K36.5, K36.6, K36.7, K36.8, K36.9, K36.10, |
| K36.11, K36.12, K36.13, K36.14, K36.15, K36.16, K36.17, K36.18, |
| K36.19, K36.20, K36.21, K36.22, K36.23, K36.24, K36.25, K36.26, |
| K36.27, K36.28, K36.29, |
| K37.1, K37.2, K37.3, K37.4, K37.5, K37.6, K37.7, K37.8, K37.9, K37.10, |
| K37.11, K37.12, K37.13, K37.14, K37.15, K37.16, K37.17, K37.18, |
| K37.19, K37.20, K37.21, K37.22, K37.23, K37.24, K37.25, K37.26, |
| K37.27, K37.28, K37.29, |
| K38.1, K38.2, K38.3, K38.4, K38.5, K38.6, K38.7, K38.8, K38.9, K38.10, |
| K38.11, K38.12, K38.13, K38.14, K38.15, K38.16, K38.17, K38.18, |
| K38.19, K38.20, K38.21, K38.22, K38.23, K38.24, K38.25, K38.26, |
| K38.27, K38.28, K38.29, |
| K39.1, K39.2, K39.3, K39.4, K39.5, K39.6, K39.7, K39.8, K39.9, K39.10, |
| K39.11, K39.12, K39.13, K39.14, K39.15, K39.16, K39.17, K39.18, |
| K39.19, K39.20, K39.21, K39.22, K39.23, K39.24, K39.25, K39.26, |
| K39.27, K39.28, K39.29, |
| K40.1, K40.2, K40.3, K40.4, K40.5, K40.6, K40.7, K40.8, K40.9, K40.10, |
| K40.11, K40.12, K40.13, K40.14, K40.15, K40.16, K40.17, K40.18, |
| K40.19, K40.20, K40.21, K40.22, K40.23, K40.24, K40.25, K40.26, |
| K40.27, K40.28, K40.29, |
| K41.1, K41.2, K41.3, K41.4, K41.5, K41.6, K41.7, K41.8, K41.9, K41.10, |
| K41.11, K41.12, K41.13, K41.14, K41.15, K41.16, K41.17, K41.18, |
| K41.19, K41.20, K41.21, K41.22, K41.23, K41.24, K41.25, K41.26, |
| K41.27, K41.28, K41.29, |
| K42.1, K42.2, K42.3, K42.4, K42.5, K42.6, K42.7, K42.8, K42.9, K42.10, |
| K42.11, K42.12, K42.13, K42.14, K42.15, K42.16, K42.17, K42.18, |
| K42.19, K42.20, K42.21, K42.22, K42.23, K42.24, K42.25, K42.26, |
| K42.27, K42.28, K42.29, |
| K43.1, K43.2, K43.3, K43.4, K43.5, K43.6, K43.7, K43.8, K43.9, K43.10, |
| K43.11, K43.12, K43.13, K43.14, K43.15, K43.16, K43.17, K43.18, |
| K43.19, K43.20, K43.21, K43.22, K43.23, K43.24, K43.25, K43.26, |
| K43.27, K43.28, K43.29, |
| K44.1, K44.2, K44.3, K44.4, K44.5, K44.6, K44.7, K44.8, K44.9, K44.10, |
| K44.11, K44.12, K44.13, K44.14, K44.15, K44.16, K44.17, K44.18, |
| K44.19, K44.20, K44.21, K44.22, K44.23, K44.24, K44.25, K44.26, |
| K44.27, K44.28, K44.29, |
| K45.1, K45.2, K45.3, K45.4, K45.5, K45.6, K45.7, K45.8, K45.9, K45.10, |
| K45.11, K45.12, K45.13, K45.14, K45.15, K45.16, K45.17, K45.18, |
| K45.19, K45.20, K45.21, K45.22, K45.23, K45.24, K45.25, K45.26, |
| K45.27, K45.28, K45.29, |
| K46.1, K46.2, K46.3, K46.4, K46.5, K46.6, K46.7, K46.8, K46.9, K46.10, |
| K46.11, K46.12, K46.13, K46.14, K46.15, K46.16, K46.17, K46.18, |
| K46.19, K46.20, K46.21, K46.22, K46.23, K46.24, K46.25, K46.26, |
| K46.27, K46.28, K46.29, |
| K47.1, K47.2, K47.3, K47.4, K47.5, K47.6, K47.7, K47.8, K47.9, K47.10, |
| K47.11, K47.12, K47.13, K47.14, K47.15, K47.16, K47.17, K47.18, |
| K47.19, K47.20, K47.21, K47.22, K47.23, K47.24, K47.25, K47.26, |
| K47.27, K47.28, K47.29, |
| K48.1, K48.2, K48.3, K48.4, K48.5, K48.6, K48.7, K48.8, K48.9, K48.10, |
| K48.11, K48.12, K48.13, K48.14, K48.15, K48.16, K48.17, K48.18, |
| K48.19, K48.20, K48.21, K48.22, K48.23, K48.24, K48.25, K48.26, |
| K48.27, K48.28, K48.29, |
| K49.1, K49.2, K49.3, K49.4, K49.5, K49.6, K49.7, K49.8, K49.9, K49.10, |
| K49.11, K49.12, K49.13, K49.14, K49.15, K49.16, K49.17, K49.18, |
| K49.19, K49.20, K49.21, K49.22, K49.23, K49.24, K49.25, K49.26, |
| K49.27, K49.28, K49.29, |
| K50.1, K50.2, K50.3, K50.4, K50.5, K50.6, K50.7, K50.8, K50.9, K50.10, |
| K50.11, K50.12, K50.13, K50.14, K50.15, K50.16, K50.17, K50.18, |
| K50.19, K50.20, K50.21, K50.22, K50.23, K50.24, K50.25, K50.26, |
| K50.27, K50.28, K50.29, |
| K51.1, K51.2, K51.3, K51.4, K51.5, K51.6, K51.7, K51.8, K51.9, K51.10, |
| K51.11, K51.12, K51.13, K51.14, K51.15, K51.16, K51.17, K51.18, |
| K51.19, K51.20, K51.21, K51.22, K51.23, K51.24, K51.25, K51.26, |
| K51.27, K51.28, K51.29, |
| K52.1, K52.2, K52.3, K52.4, K52.5, K52.6, K52.7, K52.8, K52.9, K52.10, |
| K52.11, K52.12, K52.13, K52.14, K52.15, K52.16, K52.17, K52.18, |
| K52.19, K52.20, K52.21, K52.22, K52.23, K52.24, K52.25, K52.26, |
| K52.27, K52.28, K52.29, |
| K53.1, K53.2, K53.3, K53.4, K53.5, K53.6, K53.7, K53.8, K53.9, K53.10, |
| K53.11, K53.12, K53.13, K53.14, K53.15, K53.16, K53.17, K53.18, |
| K53.19, K53.20, K53.21, K53.22, K53.23, K53.24, K53.25, K53.26, |
| K53.27, K53.28, K53.29, |
| K54.1, K54.2, K54.3, K54.4, K54.5, K54.6, K54.7, K54.8, K54.9, K54.10, |
| K54.11, K54.12, K54.13, K54.14, K54.15, K54.16, K54.17, K54.18, |
| K54.19, K54.20, K54.21, K54.22, K54.23, K54.24, K54.25, K54.26, |
| K54.27, K54.28, K54.29, |
| K55.1, K55.2, K55.3, K55.4, K55.5, K55.6, K55.7, K55.8, K55.9, K55.10, |
| K55.11, K55.12, K55.13, K55.14, K55.15, K55.16, K55.17, K55.18, |
| K55.19, K55.20, K55.21, K55.22, K55.23, K55.24, K55.25, K55.26, |
| K55.27, K55.28, K55.29, |
| K56.1, K56.2, K56.3, K56.4, K56.5, K56.6, K56.7, K56.8, K56.9, K56.10, |
| K56.11, K56.12, K56.13, K56.14, K56.15, K56.16, K56.17, K56.18, |
| K56.19, K56.20, K56.21, K56.22, K56.23, K56.24, K56.25, K56.26, |
| K56.27, K56.28, K56.29, |
| K57.1, K57.2, K57.3, K57.4, K57.5, K57.6, K57.7, K57.8, K57.9, K57.10, |
| K57.11, K57.12, K57.13, K57.14, K57.15, K57.16, K57.17, K57.18, |
| K57.19, K57.20, K57.21, K57.22, K57.23, K57.24, K57.25, K57.26, |
| K57.27, K57.28, K57.29, |
| K58.1, K58.2, K58.3, K58.4, K58.5, K58.6, K58.7, K58.8, K58.9, K58.10, |
| K58.11, K58.12, K58.13, K58.14, K58.15, K58.16, K58.17, K58.18, |
| K58.19, K58.20, K58.21, K58.22, K58.23, K58.24, K58.25, K58.26, |
| K58.27, K58.28, K58.29, |
| K59.1, K59.2, K59.3, K59.4, K59.5, K59.6, K59.7, K59.8, K59.9, K59.10, |
| K59.11, K59.12, K59.13, K59.14, K59.15, K59.16, K59.17, K59.18, |
| K59.19, K59.20, K59.21, K59.22, K59.23, K59.24, K59.25, K59.26, |
| K59.27, K59.28, K59.29, |
| K60.1, K60.2, K60.3, K60.4, K60.5, K60.6, K60.7, K60.8, K60.9, K60.10, |
| K60.11, K60.12, K60.13, K60.14, K60.15, K60.16, K60.17, K60.18, |
| K60.19, K60.20, K60.21, K60.22, K60.23, K60.24, K60.25, K60.26, |
| K60.27, K60.28, K60.29, |
| K61.1, K61.2, K61.3, K61.4, K61.5, K61.6, K61.7, K61.8, K61.9, K61.10, |
| K61.11, K61.22, K61.23, K61.24, K61.25, K61.26, K61.27, K61.28, |
| K61.29, |
| K62.1, K62.2, K62.3, K62.4, K62.5, K62.6, K62.7, K62.8, K62.9, K62.10, |
| K62.11, K62.12, K62.13, K62.14, K62.15, K62.16, K62.17, K62.18, |
| K62.19, K62.20, K62.21, K62.22, K62.23, K62.24, K62.25, K62.26, |
| K62.27, K62.28, K62.29, |
| K63.1, K63.2, K63.3, K63.4, K63.5, K63.6, K63.7, K63.8, K63.9, K63.10, |
| K63.11, K63.12, K63.13, K63.14, K63.15, K63.16, K63.17, K63.18, |
| K63.19, K63.20, K63.21, K63.22, K63.23, K63.24, K63.25, K63.26, |
| K63.27, K63.28, K63.29, |
| K64.1, K64.2, K64.3, K64.4, K64.5, K64.6, K64.7, K64.8, K64.9, K64.10, |
| K64.11, K64.12, K64.13, K64.14, K64.15, K64.16, K64.17, K64.18, |
| K64.19, K64.20, K64.21, K64.22, K64.23, K64.24, K64.25, K64.26, |
| K64.27, K64.28, K64.29, |
| K65.1, K65.2, K65.3, K65.4, K65.5, K65.6, K65.7, K65.8, K65.9, K65.10, |
| K65.11, K65.12, K65.13, K65.14, K65.15, K65.16, K65.17, K65.18, |
| K65.19, K65.20, K65.21, K65.22, K65.23, K65.24, K65.25, K65.26, |
| K65.27, K65.28, K65.29, |
| K66.1, K66.2, K66.3, K66.4, K66.5, K66.6, K66.7, K66.8, K66.9, K66.10, |
| K66.11, K66.12, K66.13, K66.14, K66.15, K66.16, K66.17, K66.18, |
| K66.19, K66.20, K66.21, K66.22, K66.23, K66.24, K66.25, K66.26, |
| K66.27, K66.28, K66.29, |
| K67.1, K67.2, K67.3, K67.4, K67.5, K67.6, K67.7, K67.8, K67.9, K67.10, |
| K67.11, K67.12, K67.13, K67.14, K67.15, K67.16, K67.17, K67.18, |
| K67.19, K67.20, K67.21, K67.22, K67.23, K67.24, K67.25, K67.26, |
| K67.27, K67.28, K67.29, |
| K68.1, K68.2, K68.3, K68.4, K68.5, K68.6, K68.7, K68.8, K68.9, K68.10, |
| K68.11, K68.12, K68.13, K68.14, K68.15, K68.16, K68.17, K68.18, |
| K68.19, K68.20, K68.21, K68.22, K68.23, K68.24, K68.25, K68.26, |
| K68.27, K68.28, K68.29, |
| K69.1, K69.2, K69.3, K69.4, K69.5, K69.6, K69.7, K69.8, K69.9, K69.10, |
| K69.11, K69.12, K69.13, K69.14, K69.15, K69.16, K69.17, K69.18, |
| K69.19, K69.20, K69.21, K69.22, K69.23, K69.24, K69.25, K69.26, |
| K69.27, K69.28, K69.29, |
| K70.1, K70.2, K70.3, K70.4, K70.5, K70.6, K70.7, K70.8, K70.9, K70.10, |
| K70.11, K70.12, K70.13, K70.14, K70.15, K70.16, K70.17, K70.18, |
| K70.19, K70.20, K70.21, K70.22, K70.23, K70.24, K70.25, K70.26, |
| K70.27, K70.28, K70.29, |
| K71.1, K71.2, K71.3, K71.4, K71.5, K71.6, K71.7, K71.8, K71.9, K71.10, |
| K71.11, K71.12, K71.13, K71.14, K71.15, K71.16, K71.17, K71.18, |
| K71.19, K71.20, K71.21, K71.22, K71.23, K71.24, K71.25, K71.26, |
| K71.27, K71.28, K71.29, |
| K72.1, K72.2, K72.3, K72.4, K72.5, K72.6, K72.7, K72.8, K72.9, K72.10, |
| K72.11, K72.12, K72.13, K72.14, K72.15, K72.16, K72.17, K72.18, |
| K72.19, K72.20, K72.21, K72.22, K72.23, K72.24, K72.25, K72.26, |
| K72.27, K72.28, K72.29, |
| K73.1, K73.2, K73.3, K73.4, K73.5, K73.6, K73.7, K73.8, K73.9, K73.10, |
| K73.11, K73.12, K73.13, K73.14, K73.15, K73.16, K73.17, K73.18, |
| K73.19, K73.20, K73.21, K73.22, K73.23, K73.24, K73.25, K73.26, |
| K73.27, K73.28, K73.29, |
| K74.1, K74.2, K74.3, K74.4, K74.5, K74.6, K74.7, K74.8, K74.9, K74.10, |
| K74.11, K74.12, K74.13, K74.14, K74.15, K74.16, K74.17, K74.18, |
| K74.19, K74.20, K74.21, K74.22, K74.23, K74.24, K74.25, K74.26, |
| K74.27, K74.28, K74.29, |
| K75.1, K75.2, K75.3, K75.4, K75.5, K75.6, K75.7, K75.8, K75.9, K75.10, |
| K75.11, K75.12, K75.13, K75.14, K75.15, K75.16, K75.17, K75.18, |
| K75.19, K75.20, K75.21, K75.22, K75.23, K75.24, K75.25, K75.26, |
| K75.27, K75.28, K75.29, |
| K76.1, K76.2, K76.3, K76.4, K76.5, K76.6, K76.7, K76.8, K76.9, K76.10, |
| K76.11, K76.12, K76.13, K76.14, K76.15, K76.16, K76.17, K76.18, |
| K76.19, K76.20, K76.21, K76.22, K76.23, K76.24, K76.25, K76.26, |
| K76.27, K76.28, K76.29, |
| K77.1, K77.2, K77.3, K77.4, K77.5, K77.6, K77.7, K77.8, K77.9, K77.10, |
| K77.11, K77.12, K77.13, K77.14, K77.15, K77.16, K77.17, K77.18, |
| K77.19, K77.20, K77.21, K77.22, K77.23, K77.24, K77.25, K77.26, |
| K77.27, K77.28, K77.29, |
| K78.1, K78.2, K78.3, K78.4, K78.5, K78.6, K78.7, K78.8, K78.9, K78.10, |
| K78.11, K78.12, K78.13, K78.14, K78.15, K78.16, K78.17, K78.18, |
| K78.19, K78.20, K78.21, K78.22, K78.23, K78.24, K78.25, K78.26, |
| K78.27, K78.28, K78.29, |
| K79.1, K79.2, K79.3, K79.4, K79.5, K79.6, K79.7, K79.8, K79.9, K79.10, |
| K79.11, K79.12, K79.13, K79.14, K79.15, K79.16, K79.17, K79.18, |
| K79.19, K79.20, K79.21, K79.22, K79.23, K79.24, K79.25, K79.26, |
| K79.27, K79.28, K79.29, |
| K80.1, K80.2, K80.3, K80.4, K80.5, K80.6, K80.7, K80.8, K80.9, K80.10, |
| K80.11, K80.12, K80.13, K80.14, K80.15, K80.16, K80.17, K80.18, |
| K80.19, K80.20, K80.21, K80.22, K80.23, K80.24, K80.25, K80.26, |
| K80.27, K80.28, K80.29, |
| K81.1, K81.2, K81.3, K81.4, K81.5, K81.6, K81.7, K81.8, K81.9, K81.10, |
| K81.11, K81.12, K81.13, K81.14, K81.15, K81.16, K81.17, K81.18, |
| K81.19, K81.20, K81.21, K81.22, K81.23, K81.24, K81.25, K81.26, |
| K81.27, K81.28, K81.29, |
| K82.1, K82.2, K82.3, K82.4, K82.5, K82.6, K82.7, K82.8, K82.9, K82.10, |
| K82.11, K82.12, K82.13, K82.14, K82.15, K82.16, K82.17, K82.18, |
| K82.19, K82.20, K82.21, K82.22, K82.23, K82.24, K82.25, K82.26, |
| K82.27, K82.28, K82.29, |
| K83.1, K83.2, K83.3, K83.4, K83.5, K83.6, K83.7, K83.8, K83.9, K83.10, |
| K83.11, K83.12, K83.13, K83.14, K83.15, K83.16, K83.17, K83.18, |
| K83.19, K83.20, K83.21, K83.22, K83.23, K83.24, K83.25, K83.26, |
| K83.27, K83.28, K83.29, |
| K84.1, K84.2, K84.3, K84.4, K84.5, K84.6, K84.7, K84.8, K84.9, K84.10, |
| K84.11, K84.12, K84.13, K84.14, K84.15, K84.16, K84.17, K84.18, |
| K84.19, K84.20, K84.21, K84.22, K84.23, K84.24, K84.25, K84.26, |
| K84.27, K84.28, K84.29, |
| K85.1, K85.2, K85.3, K85.4, K85.5, K85.6, K85.7, K85.8, K85.9, K85.10, |
| K85.11, K85.12, K85.13, K85.14, K85.15, K85.16, K85.17, K85.18, |
| K85.19, K85.20, K85.21, K85.22, K85.23, K85.24, K85.25, K85.26, |
| K85.27, K85.28, K85.29, |
| K86.1, K86.2, K86.3, K86.4, K86.5, K86.6, K86.7, K86.8, K86.9, K86.10, |
| K86.11, K86.12, K86.13, K86.14, K86.15, K86.16, K86.17, K86.18, |
| K86.19, K86.20, K86.21, K86.22, K86.23, K86.24, K86.25, K86.26, |
| K86.27, K86.28, K86.29, |
| K87.1, K87.2, K87.3, K87.4, K87.5, K87.6, K87.7, K87.8, K87.9, K87.10, |
| K87.11, K87.12, K87.13, K87.14, K87.15, K87.16, K87.17, K87.18, |
| K87.19, K87.20, K87.21, K87.22, K87.23, K87.24, K87.25, K87.26, |
| K87.27, K87.28, K87.29, |
| K88.1, K88.2, K88.3, K88.4, K88.5, K88.6, K88.7, K88.8, K88.9, K88.10, |
| K88.11, K88.12, K88.13, K88.14, K88.15, K88.16, K88.17, K88.18, |
| K88.19, K88.20, K88.21, K88.22, K88.23, K88.24, K88.25, K88.26, |
| K88.27, K88.28, K88.29, |
| K89.1, K89.2, K89.3, K89.4, K89.5, K89.6, K89.7, K89.8, K89.9, K89.10, |
| K89.11, K89.12, K89.13, K89.14, K89.15, K89.16, K89.17, K89.18, |
| K89.19, K89.20, K89.21, K89.22, K89.23, K89.24, K89.25, K89.26, |
| K89.27, K89.28, K89.29, |
| K90.1, K90.2, K90.3, K90.4, K90.5, K90.6, K90.7, K90.8, K90.9, K90.10, |
| K90.11, K90.12, K90.13, K90.14, K90.15, K90.16, K90.17, K90.18, |
| K90.19, K90.20, K90.21, K90.22, K90.23, K90.24, K90.25, K90.26, |
| K90.27, K90.28, K90.29, |
| K91.1, K91.2, K91.3, K91.4, K91.5, K91.6, K91.7, K91.8, K91.9, K91.10, |
| K91.11, K91.12, K91.13, K91.14, K91.15, K91.16, K91.17, K91.18, |
| K91.19, K91.20, K91.21, K91.22, K91.23, K91.24, K91.25, K91.26, |
| K91.27, K91.28, K91.29, |
| K92.1, K92.2, K92.3, K92.4, K92.5, K92.6, K92.7, K92.8, K92.9, K92.10, |
| K92.11, K92.12, K92.13, K92.14, K92.15, K92.16, K92.17, K92.18, |
| K92.19, K92.20, K92.21, K92.22, K92.23, K92.24, K92.25, K92.26, |
| K92.27, K92.28, K92.29, |
| K93.1, K93.2, K93.3, K93.4, K93.5, K93.6, K93.7, K93.8, K93.9, K93.10, |
| K93.11, K93.12, K93.13, K93.14, K93.15, K93.16, K93.17, K93.18, |
| K93.19, K93.20, K93.21, K93.22, K93.23, K93.24, K93.25, K93.26, |
| K93.27, K93.28, K93.29, |
| K94.1, K94.2, K94.3, K94.4, K94.5, K94.6, K94.7, K94.8, K94.9, K94.10, |
| K94.11, K94.12, K94.13, K94.14, K94.15, K94.16, K94.17, K94.18, |
| K94.19, K94.20, K94.21, K94.22, K94.23, K94.24, K94.25, K94.26, |
| K94.27, K94.28, K94.29, |
| K95.1, K95.2, K95.3, K95.4, K95.5, K95.6, K95.7, K95.8, K95.9, K95.10, |
| K95.11, K95.12, K95.13, K95.14, K95.15, K95.16, K95.17, K95.18, |
| K95.19, K95.20, K95.21, K95.22, K95.23, K95.24, K95.25, K95.26, |
| K95.27, K95.28, K95.29, |
| K96.1, K96.2, K96.3, K96.4, K96.5, K96.6, K96.7, K96.8, K96.9, K96.10, |
| K96.11, K96.12, K96.13, K96.14, K96.15, K96.16, K96.17, K96.18, |
| K96.19, K96.20, K96.21, K96.22, K96.23, K96.24, K96.25, K96.26, |
| K96.27, K96.28, K96.29, |
| K97.1, K97.2, K97.3, K97.4, K97.5, K97.6, K97.7, K97.8, K97.9, K97.10, |
| K97.11, K97.12, K97.13, K97.14, K97.15, K97.16, K97.17, K97.18, |
| K97.19, K97.20, K97.21, K97.22, K97.23, K97.24, K97.25, K97.26, |
| K97.27, K97.28, K97.29, |
| K98.1, K98.2, K98.3, K98.4, K98.5, K98.6, K98.7, K98.8, K98.9, K98.10, |
| K98.11, K98.12, K98.13, K98.14, K98.15, K98.16, K98.17, K98.18, |
| K98.19, K98.20, K98.21, K98.22, K98.23, K98.24, K98.25, K98.26, |
| K98.27, K98.28, K98.29, |
| K99.1, K99.2, K99.3, K99.4, K99.5, K99.6, K99.7, K99.8, K99.9, K99.10, |
| K99.11, K99.12, K99.13, K99.14, K99.15, K99.16, K99.17, K99.18, |
| K99.19, K99.20, K99.21, K99.22, K99.23, K99.24, K99.25, K99.26, |
| K99.27, K99.28, K99.29, |
| K100.1, K100.2, K100.3, K100.4, K100.5, K100.6, K100.7, K100.8, |
| K100.9, K100.10, K100.11, K100.12, K100.13, K100.14, K100.15, |
| K100.16, K100.17, K100.18, K100.19, K100.20, K100.21, K100.22, |
| K100.23, K100.24, K100.25, K100.26, K100.27, K100.28, K100.29, |
| K101.1, K101.2, K101.3, K101.4, K101.5, K101.6, K101.7, K101.8, |
| K101.9, K101.10, K101.11, K101.12, K101.13, K101.14, K101.15, |
| K101.16, K101.17, K101.18, K101.19, K101.20, K101.21, K101.22, |
| K101.23, K101.24, K101.25, K101.26, K101.27, K101.28, K101.29, |
| K102.1, K102.2, K102.3, K102.4, K102.5, K102.6, K102.7, K102.8, |
| K102.9, K102.10, K102.11, K102.12, K102.13, K102.14, K102.15, |
| K102.16, K102.17, K102.18, K102.19, K102.20, K102.21, K102.22, |
| K102.23, K102.24, K102.25, K102.26, K102.27, K102.28, K102.29, |
| K103.1, K103.2, K103.3, K103.4, K103.5, K103.6, K103.7, K103.8, |
| K103.9, K103.10, K103.11, K103.12, K103.13, K103.14, K103.15, |
| K103.16, K103.17, K103.18, K103.19, K103.20, K103.21, K103.22, |
| K103.23, K103.24, K103.25, K103.26, K103.27, K103.28, K103.29, |
| K104.1, K104.2, K104.3, K104.4, K104.5, K104.6, K104.7, K104.8, |
| K104.9, K104.10, K104.11, K104.12, K104.13, K104.14, K104.15, |
| K104.16, K104.17, K104.18, K104.19, K104.20, K104.21, K104.22, |
| K104.23, K104.24, K104.25, K104.26, K104.27, K104.28, K104.29, |
| K105.1, K105.2, K105.3, K105.4, K105.5, K105.6, K105.7, K105.8, |
| K105.9, K105.10, K105.11, K105.12, K105.13, K105.14, K105.15, |
| K105.16, K105.17, K105.18, K105.19, K105.20, K105.21, K105.22, |
| K105.23, K105.24, K105.25, K105.26, K105.27, K105.28, K105.29, |
| K106.1, K106.2, K106.3, K106.4, K106.5, K106.6, K106.7, K106.8, |
| K106.9, K106.10, K106.11, K106.12, K106.13, K106.14, K106.15, |
| K106.16, K106.17, K106.18, K106.19, K106.20, K106.21, K106.22, |
| K106.23, K106.24, K106.25, K106.26, K106.27, K106.28, K106.29, |
| K107.1, K107.2, K107.3, K107.4, K107.5, K107.6, K107.7, K107.8, |
| K107.9, K107.10, K107.11, K107.12, K107.13, K107.14, K107.15, |
| K107.16, K107.17, K107.18, K107.19, K107.20, K107.21, K107.22, |
| K107.23, K107.24, K107.25, K107.26, K107.27, K107.28, K107.29, |
| K108.1, K108.2, K108.3, K108.4, K108.5, K108.6, K108.7, K108.8, |
| K108.9, K108.10, K108.11, K108.12, K108.13, K108.14, K108.15, |
| K108.16, K108.17, K108.18, K108.19, K108.20, K108.21, K108.22, |
| K108.23, K108.24, K108.25, K108.26, K108.27, K108.28, K108.29, |
| K109.1, K109.2, K109.3, K109.4, K109.5, K109.6, K109.7, K109.8, |
| K109.9, K109.10, K109.11, K109.12, K109.13, K109.14, K109.15, |
| K109.16, K109.17, K109.18, K109.19, K109.20, K109.21, K109.22, |
| K109.23, K109.24, K109.25, K109.26, K109.27, K109.28, K109.29, |
| K110.1, K110.2, K110.3, K110.4, K110.5, K110.6, K110.7, K110.8, |
| K110.9, K110.10, K110.11, K110.12, K110.13, K110.14, K110.15, |
| K110.16, K110.17, K110.18, K110.19, K110.20, K110.21, K110.22, |
| K110.23, K110.24, K110.25, K110.26, K110.27, K110.28, K110.29, |
| K111.1, K111.2, K111.3, K111.4, K111.5, K111.6, K111.7, K111.8, |
| K111.9, K111.10, K111.11, K111.12, K111.13, K111.14, K111.15, |
| K111.16, K111.17, K111.18, K111.19, K111.20, K111.21, K111.22, |
| K111.23, K111.24, K111.25, K111.26, K111.27, K111.28, K111.29, |
| K112.1, K112.2, K112.3, K112.4, K112.5, K112.6, K112.7, K112.8, |
| K112.9, K112.10, K112.11, K112.12, K112.13, K112.14, K112.15, |
| K112.16, K112.17, K112.18, K112.19, K112.20, K112.21, K112.22, |
| K112.23, K112.24, K112.25, K112.26, K112.27, K112.28, K112.29, |
| K113.1, K113.2, K113.3, K113.4, K113.5, K113.6, K113.7, K113.8, |
| K113.9, K113.10, K113.11, K113.12, K113.13, K113.14, K113.15, |
| K113.16, K113.17, K113.18, K113.19, K113.20, K113.21, K113.22, |
| K113.23, K113.24, K113.25, K113.26, K113.27, K113.28, K113.29, |
| K114.1, K114.2, K114.3, K114.4, K114.5, K114.6, K114.7, K114.8, |
| K114.9, K114.10, K114.11, K114.12, K114.13, K114.14, K114.15, |
| K114.16, k114.17, K114.18, K114.19, K114.20, K114.21, K114.22, |
| K114.23, K114.24, K114.25, K114.26, K114.27, K114.28, K114.29, |
| K115.1, K115.2, K115.3, K115.4, K115.5, K115.6, K115.7, K115.8, |
| K115.9, K115.10, K115.11, K115.12, K115.13, K115.14, K115.15, |
| K115.16, K115.17, K115.18, K115.19, K115.20, K115.21, K115.22, |
| K115.23, K115.24, K115.25, K115.26, K115.27, K115.28, K115.29, |
| K116.1, K116.2, K116.3, K116.4, K116.5, K116.6, K116.7, K116.8, |
| K116.9, K116.10, K116.11, K116.12, K116.13, K116.14, K116.15, |
| K116.16, K116.17, K116.18, K116.19, K116.20, K116.21, K116.22, |
| K116.23, K116.24, K116.25, K116.26, K116.27, K116.28, K116.29, |
| K117.1, K117.2, K117.3, K117.4, K117.5, K117.6, K117.7, K117.8, |
| K117.9, K117.10, K117.11, K117.12, K117.13, K117.14, K117.15, |
| K117.16, K117.17, K117.18, K117.19, K117.20, K117.21, K117.22, |
| K117.23, K117.24, K117.25, K117.26, K117.27, K117.28, K117.29, |
| K118.1, K118.2, K118.3, K118.4, K118.5, K118.6, K118.7, K118.8, |
| K118.9, K118.10, K118.11, K118.12, K118.13, K118.14, K118.15, |
| K118.16, K118.17, K118.18, K118.19, K118.20, K118.21, K118.22, |
| K118.23, K118.24, K118.25, K118.26, K118.27, K118.28, K118.29, |
| K119.1, K119.2, K119.3, K119.4, K119.5, K119.6, K119.7, K119.8, |
| K119.9, K119.10, K119.11, K119.12, K119.13, K119.14, K119.15, |
| K119.16, K119.17, K119.18, K119.19, K119.20, K119.21, K119.22, |
| K119.23, K119.24, K119.25, K119.26, K119.27, K119.28, K119.29, |
| K120.1, K120.2, K120.3, K120.4, K120.5, K120.6, K120.7, K120.8, |
| K120.9, K120.10, K120.11, K120.12, K120.13, K120.14, K120.15, |
| K120.16, K120.17, K120.18, K120.19, K120.20, K120.21, K120.22, |
| K120.23, K120.24, K120.25, K120.26, K120.27, K120.28, K120.29, |
| K121.1, K121.2, K121.3, K121.4, K121.5, K121.6, K121.7, K121.8, |
| K121.9, K121.10, K121.11, K121.12, K121.13, K121.14, K121.15, |
| K121.16, K121.17, K121.18, K121.19, K121.20, K121.21, K121.22, |
| K121.23, K121.24, K121.25, K121.26, K121.27, K121.28, K121.29, |
| K122.1, K122.2, K122.3, K122.4, K122.5, K122.6, K122.7, K122.8, |
| K122.9, K122.10, K122.11, K122.12, K122.13, K122.14, K122.15, |
| K122.16, K122.17, K122.18, K122.19, K122.20, K122.21, K122.22, |
| K122.23, K122.24, K122.25, K122.26, K122.27, K122.28, K122.29, |
| K123.1, K123.2, K123.3, K123.4, K123.5, K123.6, K123.7, K123.8, |
| K123.9, K123.10, K123.11, K123.12, K123.13, K123.14, K123.15, |
| K123.16, K123.17, K123.18, K123.19, K123.20, K123.21, K123.22, |
| K123.23, K123.24, K123.25, K123.26, K123.27, K123.28, K123.29, |
| K124.1, K124.2, K124.3, K124.4, K124.5, K124.6, K124.7, K124.8, |
| K124.9, K124.10, K124.11, K124.12, K124.13, K124.14, K124.15, |
| K124.16, K124.17, K124.18, K124.19, K124.20, K124.21, K124.22, |
| K124.23, K124.24, K124.25, K124.26, K124.27, K124.28, K124.29, |
| K125.1, K125.2, K125.3, K125.4, K125.5, K125.6, K125.7, K125.8, |
| K125.9, K125.10, K125.11, K125.12, K125.13, K125.14, K125.15, |
| K125.16, K125.17, K125.18, K125.19, K125.20, K125.21, K125.22, |
| K125.23, K125.24, K125.25, K125.26, K125.27, K125.28, K125.29, |
| K126.1, K126.2, K126.3, K126.4, K126.5, K126.6, K126.7, K126.8, |
| K126.9, K126.10, K126.11, K126.12, K126.13, K126.14, K126.15, |
| K126.16, K126.17, K126.18, K126.19, K126.20, K126.21, K126.22, |
| K126.23, K126.24, K126.25, K126.26, K126.27, K126.28, K126.29, |
| K127.1, K127.2, K127.3, K127.4, K127.5, K127.6, K127.7, K127.8, |
| K127.9, K127.10, K127.11, K127.12, K127.13, K127.14, K127.15, |
| K127.16, K127.17, K127.18, K127.19, K127.20, K127.21, K127.22, |
| K127.23, K127.24, K127.25, K127.26, K127.27, K127.28, K127.29. |
| Abbreviations for Table 3: |
| 1, See explanation prior to Table 2 for the abbreviation “Ka.b” and the codes “a” and “b” |
| 2, Example: in the short notation of Table 3, the combination of the herbicides (A5) and (B2.3.1) is thus stated as K5.20. |
- (A)+(B1)+(B2), (A)+(B1)+(B3), (A)+(B2)+(B3) or (A)+(B1)+(B2)+(B3).
- (A)+(B1)+(C), (A)+(B2)+(C), (A)+(B3)+(C), (A)+(B1)+(B2)+(C), (A)+(B1)+(B3)+(C), (A)+(B2)+(B3)+(C) or (A)+(B1)+(B2)+(B3)+(C).
- (B1.1.2) thiencarbazone-methyl, (B1.2.1) tembotrione, (B1.3) SYN-523, (B1.4.1) pyroxsulam, (B1.5.1) penoxsulam, (B1.6.1) SYN-449, (B2.1.1) pyrasulfotole,
- (B2.2.2) trifloxysulfuron sodium salt, (B2.3.1) saflufenacil, (B2.4.1) aminopyralid, (B2.5) ethofumesate, (B2.6) aminocyclopyrachlor and its salts and esters, in particular (B2.6.1) aminocyclopyrachlor, and (B3.1) pyroxasulfone.
- (B1.1.2) thiencarbazone-methyl, (B1.2.1) tembotrione, (B2.1.1) pyrasulfotole and (B2.5) ethofumesate.
- (A)+(B1.1.2)+(B1.2.1), (A)+(B1.1.2)+(B1.3), (A)+(B1.1.2)+(B1.4.1), (A)+(B1.1.2)+(B1.5.1), (A)+(B1.1.2)+(B1.6.1), (A)+(B1.1.2)+(B2.1.1), (A)+(B1.1.2)+(B2.2.2), (A)+(B1.1.2)+(B2.3.1), (A)+(B1.1.2)+(B2.4.1), (A)+(B1.1.2)+(B2.5), (A)+(B1.1.2)+(B2.6.1), (A)+(B1.1.2)+(B2.6.2), (A)+(B1.1.2)+(B2.6.4), (A)+(B1.1.2)+(B3.1),
- (A)+(B1.2.1)+(B1.3), (A)+(B1.2.1)+(B1.4.1), (A)+(B1.2.1)+(B1.5.1), (A)+(B1.2.1)+(B1.6.1), (A)+(B1.2.1)+(B2.1.1), (A)+(B1.2.1)+(B2.2.2), (A)+(B1.2.1)+(B2.3.1), (A)+(B1.2.1)+(B2.4.1), (A)+(B1.2.1)+(B2.5), (A)+(B1.2.1)+(B2.6.1), (A)+(B1.2.1)+(B2.6.2), (A)+(B1.2.1)+(B2.6.4), (A)+(B1.2.1)+(B3.1),
- (A)+(B1.3)+(B1.4.1), (A)+(B1.3)+(B1.5.1), (A)+(B1.3)+(B1.6.1), (A)+(B1.3)+(B2.1.1), (A)+(B1.3)+(B2.2.2), (A)+(B1.3)+(B2.3.1), (A)+(B1.3)+(B2.4.1), (A)+(B1.3)+(B2.5), (A)+(B1.3)+(B2.6.1), (A)+(B1.3)+(B2.6.2), (A)+(B1.3)+(B2.6.4), (A)+(B1.3)+(B3.1),
- (A)+(B1.4.1)+(B1.5.1), (A)+(B1.4.1)+(B1.6.1), (A)+(B1.4.1)+(B2.1.1), (A)+(B1.4.1)+(B2.2.2), (A)+(B1.4.1)+(B2.3.1), (A)+(B1.4.1)+(B2.4.1), (A)+(B1.4.1)+(B2.5), (A)+(B1.4.1)+(B2.6.1), (A)+(B1.4.1)+(B2.6.2), (A)+(B1.4.1)+(B2.6.4), (A)+(B1.4.1)+(B3.1),
- (A)+(B1.5.1)+(B1.6.1), (A)+(B1.5.1)+(B2.1.1), (A)+(B1.5.1)+(B2.2.2), (A)+(B1.5.1)+(B2.3.1), (A)+(B1.5.1)+(B2.4.1), (A)+(B1.5.1)+(B2.5), (A)+(B1.5.1)+(B2.6.1), (A)+(B1.5.1)+(B2.6.2), (A)+(B1.5.1)+(B2.6.4), (A)+(B1.5.1)+(B3.1),
- (A)+(B1.6.1)+(B2.1.1), (A)+(B1.6.1)+(B2.2.2), (A)+(B1.6.1)+(B2.3.1), (A)+(B1.6.1)+(B2.4.1), (A)+(B1.6.1)+(B2.5), (A)+(B1.6.1)+(B2.6.1), (A)+(B1.6.1)+(B2.6.2), (A)+(B1.6.1)+(B2.6.4), (A)+(B1.6.1)+(B3.1),
- (A)+(B2.1.1)+(B2.2.2), (A)+(B2.1.1)+(B2.3.1), (A)+(B2.1.1)+(B2.4.1), (A)+(B2.1.1)+(B2.5), (A)+(B2.1.1)+(B2.6.1), (A)+(B2.1.1)+(B2.6.2), (A)+(B2.1.1)+(B2.6.4), (A)+(B2.1.1)+(B3.1),
- (A)+(B2.2.2)+(B2.3.1), (A)+(B2.2.2)+(B2.4.1), (A)+(B2.2.2)+(B2.5), (A)+(B2.2.2)+(B2.6.1), (A)+(B2.2.2)+(B2.6.2), (A)+(B2.2.2)+(B2.6.4), (A)+(B2.2.2)+(B3.1),
- (A)+(B2.3.1)+(B2.4.1), (A)+(B2.3.1)+(B2.5), (A)+(B2.3.1)+(B2.6.1), (A)+(B2.3.1)+(B2.6.2), (A)+(B2.3.1)+(B2.6.4), (A)+(B2.3.1)+(B3.1),
- (A)+(B2.4.1)+(B2.5), (A)+(B2.4.1)+(B2.6.1), (A)+(B2.4.1)+(B2.6.2), (A)+(B2.4.1)+(B2.6.4), (A)+(B2.4.1)+(B3.1),
- (A)+(B2.5)+(B2.6.1), (A)+(B2.5)+(B2.6.2), (A)+(B2.5)+(B2.6.4), (A)+(B2.5)+(B3.1).
- (A)+(B2.6.1)+(B2.6.2), (A)+(B2.6.1)+(B2.6.4), (A)+(B2.6.1)+(B3.1), (A)+(B2.6.2)+(B2.6.4), (A)+(B2.6.2)+(B3.1) and
- (A)+(B2.6.4)+(B3.1).
- 2,4-D, acetochlor, acifluorfen, acifluorfen-sodium, aclonifen, alachlor, alloxydim, alloxydim-sodium, ametryn, amicarbazone, amidosulfuron, amitrole, anilofos, asulam, atrazine, azafenidin, azimsulfuron, beflubutamid, benazolin, benazolin-ethyl, benfuresate, bensulfuron-methyl, bentazone, benzfendizone, benzobicyclon, benzofenap, bifenox, bilanafos, bispyribac-sodium, bromacil, bromobutide, bromofenoxim, bromoxynil, butachlor, butafenacil, butenachlor, butralin, butroxydim, butylate, cafenstrole, carbetamide, carfentrazone-ethyl, chlomethoxyfen, chloridazon, chlorimuron-ethyl, chlornitrofen, chlorotoluron, chlorsulfuron, cinidon-ethyl, cinmethylin, cinosulfuron, clefoxydim, clethodim, clodinafop-propargyl, clomazone, clomeprop, clopyralid, cloransulam-methyl, cumyluron, cyanazine, cyclosulfamuron, cycloxydim, cyhalofop-butyl, desmedipham, dicamba, dichlobenil, dichlorprop, dichlorprop-P, diclofop-methyl, diclosulam, difenzoquat, diflufenican, diflufenzopyr, dikegulac-sodium, dimefuron, dimepiperate, dimethachlor, dimethametryn, dimethenamid, Triaziflam, diquatdibromide, dithiopyr, diuron, dymron, EPTC, esprocarb, ethalfluralin, ethametsulfuron-methyl, ethoxyfen, ethoxysulfuron, etobenzanid, fenoxaprop-ethyl, fenoxaprop-P-ethyl, fentrazamide, flamprop-M-isopropyl, flamprop-M-methyl, flazasulfuron, florasulam, fluazifop, fluazifop-butyl, fluazifop-P-butyl, fluazolate, flucarbazone-sodium, flucetosulfuron, fluchloralin, flufenacet, flufenpyr, flumetsulam, flumiclorac-pentyl, flumioxazin, fluometuron, fluorochloridone, fluoroglycofen-ethyl, flupoxam, flupyrsulfuron-methyl-sodium, fluridone, fluoroxypyr, fluoroxypyr-butoxypropyl, fluoroxypyr-meptyl, flurprimidol, flurtamone, fluthiacet-methyl, fomesafen, foramsulfuron, glufosinate, glufosinate-ammonium, glyphosate, halosulfuron-methyl, haloxyfop, haloxyfop-ethoxyethyl, haloxyfop-methyl, haloxyfop-P-methyl, hexazinone, imazamethabenz-methyl, imazamox, imazapic, imazapyr, imazaquin, imazethapyr, imazosulfuron, indanofan, iodosulfuron-methyl-sodium, ioxynil, isoproturon, isouron, isoxaben, isoxachlortole, isoxaflutole, ketospiradox, lactofen, lenacil, linuron, MCPA, mecoprop, mecoprop-P, mefenacet, mesosulfuron-methyl, mesotrione, metamifop, metamitron, metazachlor, methabenzthiazuron, methyldymron, metobromuron, metolachlor, metosulam, metoxuron, metribuzin, metsulfuron-methyl, molinate, monolinuron, naproanilide, napropamide, neburon, nico-sulfuron, norflurazon, orbencarb, oryzalin, oxadiargyl, oxadiazon, oxasulfuron, oxaziclomefone, oxyfluorfen, paraquat, pelargonic acid, pendimethalin, pendralin, pentoxazone, pethoxamid, phenmedipham, picloram, picolinafen, pinoxaden, piperophos, pretilachlor, primisulfuron-methyl, profluazol, profoxydim, prometryn, propachlor, propanil, propaquizafop, propisochlor, propoxycarbazone-sodium, propyzamide, prosulfocarb, prosulfuron, pyraclonil, pyraflufen-ethyl, pyrazolate, pyrazosulfuron-ethyl, pyrazoxyfen, pyribenzoxim, pyributicarb, pyridafol, pyridate, pyriftalid, pyriminobac-methyl, pyrithiobac-sodium, quinclorac, quinmerac, quinoclamine, quizalofop-ethyl, quizalofop-P-ethyl, quizalofop-P-tefuryl, rimsulfuron, sethoxydim, simazine, simetryn, S-metolachlor, sulcotrione, sulfentrazone, sulfometuron-methyl, sulfosate, sulfosulfuron, tebuthiuron, tepraloxydim, terbuthylazine, terbutryn, thenylchlor, thiazopyr, thifensulfuron-methyl, thiobencarb, tiocarbazil, tralkoxydim, triallate, triasulfuron, tribenuron-methyl, triclopyr, tridiphane, trifloxysulfuron, trifluralin, triflusulfuron-methyl, tritosulfuron, WL 110547, i.e. 5-phenoxy-1-[3-(trifluoromethyl) phenyl]-1H-tetrazole; HOK-201, HOK-202, UBH-509; D-489; LS 82-556; KPP-300; NC-324; NC-330; KH-218; DPX-N8189; SC-0774; TH-547, DOWCO-535; DK-8910; V-53482; PP-600; MBH-001; KIH-9201; ET-751; KIH-6127; KIH-2023 and KIH5996. If the respective name (common name) includes more than one form of the active compound, the name preferably defines the commercially available form.
- benoxacor, cloquintocet(-mexyl), cyometrinil, cyprosulfamide, dichlormid, fenchlorazole(-ethyl), fenclorim, flurazole, fluxofenim, furilazole, isoxadifen(-ethyl), mefenpyr (-diethyl), naphthalic anhydride, oxabetrinil, “AD-67” (=“MON 4660”=3-dichloroacetyl-1-oxa-3-azaspiro[4,5] decane), “TI-35” (=1-dichloroacetylazepane), dimepiperate, daimuron, cumyluron, are of particular interest.
- 2-phenylphenol; 8-hydroxyquinoline sulfate; acibenzolar-S-methyl; actinovate; aldimorph; amidoflumet; ampropyl-fos; ampropylfos-potassium; andoprim; anilazine; azaconazole; azoxystrobin; benalaxyl; benodanil; benomyl; benthiavalicarb-isopropyl; benzamacril; benzamacril-isobutyl; binapacryl; biphenyl; bitertanol; blasticidin-S; boscalid; bromuconazole; bupirimate; buthiobate; butylamine; calcium polysulfide; capsimycin; captafol; captan; carbendazim; carboxin; carpropamid; carvone; chinomethionat; chlobenthiazone; chlorfenazole; chloroneb; chlorothalonil; chlozolinate; cis-1-(4-chlorophenyl)-2-(1H-1,2,4-triazol-1-yl)cycloheptanol; clozylacon; cyazofamid; cyflufenamid; cymoxanil; cyproconazole; cyprodinil; cyprofuram; Dagger G; debacarb; dichlofluanid; dichlone; dichlorophen; diclocymet; diclomezine; dicloran; diethofencarb; difenoconazole; diflumetorim; dimethirimol; dimethomorph; dimoxystrobin; diniconazole; diniconazole-M; dinocap; diphenylamine; dipyrithione; ditalimfos; dithianon; dodine; drazoxolon; edifenphos; epoxiconazole; ethaboxam; ethirimol; etridiazole; famoxadone; fenamidone; fenapanil; fenarimol; fenbuconazole; fenfuram; fenhexamid; fenitropan; fenoxanil; fenpiclonil; fenpropidin; fenpropimorph; ferbam; fluazinam; flubenzimine; fludioxonil; flumetover; flumorph; fluoromide; fluoxastrobin; fluquinconazole; flurprimidol; flusilazole; flusulfamide; flutolanil; flutriafol; folpet; fos-etyl-AI; fosetyl-sodium; fuberidazole; furalaxyl; furametpyr; furcarbanil; furmecyclox; guazatine; hexachlorobenzene; hexaconazole; hymexazol; imazalil; imibenconazole; iminoctadine triacetate; iminoctadine tris (albesilate); iodocarb; ipconazole; iprobenfos; iprodione; iprovalicarb; irumamycin; isoprothiolane; isovaledione; kasugamycin; kresoxim-methyl; mancozeb; maneb; meferimzone; mepanipyrim; mepronil; metalaxyl; metalaxyl-M; metconazole; methasulfocarb; methfuroxam; methyl 1-(2,3-dihydro-2,2-dimethyl-1H-inden-1-yl)-1H-imidazole-5-carboxylate; methyl 2-[[[cyclopropyl[(4-methoxyphenyl)imino]methyl]thio]methyl]-.alpha.-(methoxymethylene) benzeneacetate; methyl 2-[2-[3-(4-chlorophenyl)-1-methyl-allylideneaminooxymethyl]phenyl]-3-methoxyacrylate; metiram; metominostrobin; metrafenone; metsulfovax; mildiomycin; monopotassium carbonate; myclobutanil; myclozolin; nabam, N-(3-ethyl-3,5,5-trimethylcyclohexyl)-3-formylamino-2-hydroxy-benzamide; N-(6-methoxy-3-pyridinyl)cyclopropanecarboxamide; N-butyl-8-(1,1-dimethylethyl)-1-oxaspiro[4.5]decan-3-amine; natamycin; nitrothal-isopropyl; noviflumuron; nuarimol; ofurace; orysastrobin; oxadixyl; oxolinic acid; oxpoconazole; oxycarboxin; oxyfenthiin; paclobutrazol; pefurazoate: penconazole; pencycuron; penthiopyrad; phosdiphen; phthalide; picobenzamid; picoxystrobin; piperalin; polyoxins; polyoxorim; probenazole; prochloraz; procymidone; propamocarb; propanosine-sodium; propiconazole; propineb; proquinazid; prothioconazole; pyraclostrobin; pyrazophos; pyrifenox; pyrimethanil; pyroquilon; pyroxyfur; pyrrolnitrine; quinconazole; quinoxyfen; quintozene; silthiofam; simeconazole; sodium tetrathiocarbonate; spiroxamine; sulfur; tebuconazole; tecloftalam; tecnazene; tetcyclacis; tetraconazole; thiabendazole; thicyofen; thifluzamide; thiophanate-methyl; thiram; tiadinil; tioxymid; tolclofos-methyl; tolylfluanid; triadimefon; triadimenol; triazbutil; triazoxide; tricyclamide; tricyclazole; tridemorph; trifloxystrobin; triflumizole; triforine; triticonazole; uniconazole; validamycin A; vinclozolin; zineb; ziram; zoxamide; (2S)—N-[2-[4-[[3-(4-chlorophenyl)-2-propynyl]oxy]-3-methoxyphenyl]ethyl]-3-methyl-2-[(methylsulfonyl) amino]-butanamide; 1-(1-naphthalenyl)-1H-pyrrole-2,5-dione; 2,3,5,6-tetrachloro-4-(methylsulfonyl)pyridine; 2,4-dihydro-5-methoxy-2-methyl-4-[[[[1-[3-(trifluoromethyl)phenyl]ethylidene]amino]oxy]methyl]phenyl]-3H-1,2,3-triazol-3-one; 2-amino-4-methyl-N-phenyl-5-thiazolecarboxamide; 2-chloro-N-(2,3-dihydro-1,1,3-trimethyl-1H-inden-4-yl)-3-pyridinecarboxamide; 3,4,5-trichloro-2,6-pyridinedicarbonitrile; 3-[(3-bromo-6-fluoro-2-methyl-1H-indol-1-yl)sulfonyl]-N,N-dimethyl-1H-1,2,4-triazole-1-sulfonamide; copper salts and copper preparations, such as Bordeaux mixture; copper hydroxide; copper naphthenate; copper oxychloride; copper sulfate; cufraneb; copper(I) oxide; mancopper; oxine-copper.
- alanycarb, aldicarb, aldoxycarb, allyxycarb, aminocarb, bendiocarb, benfuracarb, bufencarb, butacarb, butocarboxim, butoxycarboxim, carbaryl, carbofuran, carbosulfan, cloethocarb, dimetilan, ethiofencarb, fenobucarb, fenothiocarb, formetanate, furathiocarb, isoprocarb, metam-sodium, methiocarb, methomyl, metolcarb, oxamyl, pirimicarb, promecarb, propoxur, thiodicarb, thiofanox, trimethacarb, XMC, xylylcarb, acephate, azamethiphos, azinphos (-methyl, -ethyl), bromophos-ethyl, bromfenvinfos (-methyl), butathiofos, cadusafos, carbophenothion, chlorethoxyfos, chlorfenvinphos, chlormephos, chlorpyrifos (-methyl/-ethyl), coumaphos, cyanofenphos, cyanophos, chlorfenvinphos, demeton-S-methyl, demeton-S-methylsulphon, dialifos, diazinon, dichlofenthion, dichlorvos/DDVP, dicrotophos, dimethoate, dimethylvinphos, dioxabenzofos, disulfoton, EPN, ethion, ethoprophos, etrimfos, famphur, fenamiphos, fenitrothion, fensulfothion, fenthion, flupyrazofos, fonofos, formothion, fosmethilan, fosthiazate, heptenophos, iodofenphos, iprobenfos, isazofos, isofenphos, isopropyl O-salicylate, isoxathion, malathion, mecarbam, methacrifos, methamidophos, methidathion, mevinphos, monocrotophos, naled, omethoate, oxydemeton-methyl, parathion (-methyl/-ethyl), phenthoate, phorate, phosalone, phosmet, phosphamidon, phosphocarb, phoxim, pirimiphos (-methyl/-ethyl), profenofos, propaphos, propetamphos, prothiofos, prothoate, pyraclofos, pyridaphenthion, pyridathion, quinalphos, sebufos, sulfotep, sulprofos, tebupirimfos, temephos, terbufos, tetrachlorvinphos, thiometon, triazophos, triclorfon, vamidothion, acrinathrin, allethrin (d-cis-trans, d-trans), beta-cyfluthrin, bifenthrin, bioallethrin, bioallethrin-S-cyclopentyl-isomer, bioethanomethrin, biopermethrin, bioresmethrin, chlovaporthrin, cis-cypermethrin cis-resmethrin, cis-permethrin, clocythrin, cycloprothrin, cyfluthrin, cyhalothrin, cypermethrin (alpha-, beta-, theta-, zeta-), cyphenothrin, deltamethrin, empenthrin (1R-isomer), esfenvalerate, etofenprox, fenfluthrin, fenpropathrin, fenpyrithrin, fenvalerate, flubrocythrinate, flucythrinate, flufenprox, flumethrin, fluvalinate, fubfenprox, gamma-cyhalothrin, imiprothrin, kadethrin, lambda-cyhalothrin, metofluthrin, permethrin (cis-, trans-), phenothrin (1R-trans isomer), prallethrin, profluthrin, protrifenbute, pyresmethrin, resmethrin, RU 15525, silafluofen, tau-fluvalinate, tefluthrin, terallethrin, tetramethrin (-1R-isomer), tralomethrin, transfluthrin, ZXI 8901, pyrethrins (pyrethrum), DDT, indoxacarb, acetamiprid, clothianidin, dinotefuran, imidacloprid, nitenpyram, nithiazine, thiacloprid, thiamethoxam, nicotine, bensultap, cartap, camphechlor, chlordane, endosulfan, gamma-HCH, HCH, heptachlor, lindane, methoxychlor spinosad, acetoprole, ethiprole, fipronil, vaniliprole, avermectin, emamectin, emamectin-benzoate, ivermectin, milbemycin, diofenolan, epofenonane, fenoxycarb, hydroprene, kinoprene, methoprene, pyriproxifen, triprene, chromafenozide, halofenozide, methoxyfenozide, tebufenozide, bistrifluoron, chlofluazuron, diflubenzuron, fluazuron, flucycloxuron, flufenoxuron, hexaflumuron, lufenuron, novaluron, noviflumuron, penfluoron, teflubenzuron, triflumuron, buprofezin, cyromazine, diafenthiuron, azocyclotin, cyhexatin, fenbutatin-oxide, chlorfenapyr, binapacyrl, dinobuton, dinocap, DNOC, fenazaquin, fenpyroximate, pyrimidifen, pyridaben, tebufenpyrad, tolfenpyrad, hydramethylnon, dicofol, rotenone, acequinocyl, fluacrypyrim, Bacillus thuringiensis strains, spirodiclofen, spiromesifen, 3-(2,5-dimethylphenyl)-8-methoxy-2-oxo-1-azaspiro[4.5]dec-3-en-4-yl ethyl carbonate (alias: carbonic acid, 3-(2,5-dimethylphenyl)-8-methoxy-2-oxo-1-azaspiro[4.5]dec-3-en-4-yl ethyl ester, CAS-Reg.-No.: 382608-10-8) and carbonic acid, cis-3-(2,5-dimethylphenyl)-8-methoxy-2-oxo-1-azaspiro[4.5]dec-3-en-4-yl ethyl ester (CAS-Reg.-No.: 203313-25-1), flonicamid, amitraz, propargite, N2-[1,1-dimethyl-2-(methylsulfonyl)ethyl]-3-iodo-N1-[2-methyl-4-[1,2,2,2-tetrafluoro-1-(trifluoromethyl)ethyl]phenyl]-1,2-benzenedicarboxamide (CAS-Reg.-No.: 272451-65-7), thiocyclam hydrogen oxalate, thiosultap-sodium, azadirachtin, Bacillus spec., Beauveria spec., codlemone, Metarrhizium spec., Paecilomyces spec., thuringiensin, Verticillium spec., aluminum phosphide, methyl bromide, sulfuryl fluoride, cryolite, flonicamid, pymetrozine, clofentezine, etoxazole, hexythiazox, amidoflumet, benclothiaz, benzoximate, bifenazate, bromopropylate, buprofezin, chinomethionat, chlordimeform, chlorobenzilate, chloropicrin, clothiazoben, cycloprene, dicyclanil, fenoxacrim, fentrifanil, flubenzimine, flufenerim, flutenzin, gossyplure, hydramethylnone, japonilure, metoxadiazone, petroleum, piperonyl butoxide, potassium oleate, pyridalyl, sulfluramid, tetradifon, tetrasul, triarathene, verbutin.
- acetamiprid, acrinathrin, aldicarb, amitraz, acinphos-methyl, cyfluthrin, carbaryl, cypermethrin, deltamethrin, endosulfan, ethoprophos, fenamiphos, fenthion, fipronil, imidacloprid, methamidophos, methiocarb, niclosamide, oxydemeton-methyl, prothiophos, silafluofen, thiacloprid, thiodicarb, tralomethrin, triazophos, trichlorfon, triflumuron, terbufos, fonofos, phorate, chlorpyriphos, carbofuran, tefluthrin.
-
- recombinant modifications of crop plants for the purpose of modifying the starch synthesized in the plants (for example WO 92/11376, WO 92/14827, WO 91/19806),
- transgenic crop plants which are resistant to other herbicides, for example to sulfonylureas (EP-A-0257993, U.S. Pat. No. 5,013,659),
- transgenic crop plants, with the capability of producing Bacillus thuringiensis toxins (Bt toxins) which make the plants resistant to certain pests (EP-A-0142924, EP-A-0193259),
- transgenic crop plants having a modified fatty acid composition (WO 91/13972).
- genetically modified crop plants having novel constituents or secondary compounds, for example novel phytoalexins providing increased resistance to disease (EPA 309862, EPA0464461)
- genetically modified plants having reduced photorespiration, which provide higher yields and have higher stress tolerance (EPA 0305398)
- transgenic crop plants producing pharmaceutically or diagnostically important proteins (“molecular pharming”)
- transgenic crop plants distinguished by higher yields or better quality
- transgenic crop plants distinguished by a combination, for example of the novel properties mentioned above (“gene stacking”).
- a) A dust is obtained by mixing 10 parts by weight of an active compound (A) or (B) or of an active compound mixture (A)+(B) (and if appropriate further active compound components) and/or salts thereof and 90 parts by weight of talc as inert material and comminuting the mixture in a hammer mill.
- b) A wettable powder which is readily dispersible in water is obtained by mixing 25 parts by weight of an active compound/active compound mixture, 64 parts by weight of kaolin-containing quartz as inert material, 10 parts by weight of potassium lignosulfonate and 1 part by weight of sodium oleoylmethyltaurinate as wetter and dispersant, and grinding the mixture in a pinned-disk mill.
- c) A dispersion concentrate which is readily dispersible in water is obtained by mixing 20 parts by weight of an active compound/active compound mixture with 6 parts by weight of alkylphenol polyglycol ether (®Triton X 207), 3 parts by weight of isotridecanol polyglycol ether (8 EO) and 71 parts by weight of paraffinic mineral oil (boiling range for example approx. 255 to 277° C.), and grinding the mixture in a ball mill to a fineness of below 5 microns.
- d) An emulsifiable concentrate is obtained from 15 parts by weight of an active compound/active compound mixture, 75 parts by weight of cyclohexanone as solvent and 10 parts by weight of oxethylated nonylphenol as emulsifier.
- e) Water-dispersible granules are obtained by mixing
- 75 parts by weight of an active compound/active compound mixture,
- 10 parts by weight of calcium lignosulfonate,
- 5 parts by weight of sodium lauryl sulfate,
- 3 parts by weight of polyvinyl alcohol and
- 7 parts by weight of kaolin,
- grinding the mixture on a pinned-disk mill and granulating the powder in a fluidized bed by spraying on water as granulation liquid.
- f) Water-dispersible granules are also obtained by homogenizing and precomminuting, in a colloid mill,
- 25 parts by weight of an active compound/active compound mixture,
- 5 parts by weight of sodium 2,2′-dinaphthylmethane-6,6′-disulfonate,
- 2 parts by weight of sodium oleoylmethyltaurinate,
- 1 part by weight of polyvinyl alcohol,
- 17 parts by weight of calcium carbonate and
- 50 parts by weight of water,
- subsequently grinding the mixture in a bead mill and atomizing and drying the resulting suspension in a spray tower by means of a single-substance nozzle.
E=A+B−(A·B/100)
-
- A=action of active compound (A) in % at an application rate of a g of AS/ha;
- B=action of active compound (B) in % at an application rate of b g of AS/ha;
- E=expected value of the action of the combination (A)+(B) in % at the combined application rate a+b g of AS/ha.
- g of AS/ha=grams of active substance (=100% active compound) per hectare;
- the total of the activities of the individual applications is given as EA;
- the expected values according to Colby are in each case given as EC;
Example 5.1
| TABLE 4 | ||
| Active | Application rate | Herbicidal action [%] against |
| compound(s) | [g of AS/ha] | Ipomoea hederifolia |
| (A21) | 0.5 | 45 |
| 0.2 | 0 | |
| (B1.1.2) | 2.0 | 50 |
| 0.5 | 5 | |
| (A21) + (B1.1.2) | 0.2 + 0.5 | 67 |
| (EA = 0 + 5, EC = 0 + 5) | ||
| Abbreviations and conditions in Table 4: | ||
| Conditions: pot trials/greenhouse, application by the pre-emergence method, evaluation 27 days after treatment | ||
| AS = active substance (based on 100% of active compound) | ||
| (A21) = compound (A21) from Table 1 = 2-amino-4-[(1R,2S)-2,6-dimethyl-indan-1-ylamino]-6-[(1R)-1-fluoroethyl]-1,3,5-triazine | ||
| (B1.1.2) = thiencarbazone-methyl = methyl 4-[(4,5-dihydro-3-methoxy-4-methyl-5-oxo-1H-1,2,4-triazol-1-yl)carbonylsulfamoyl]-5-methylthiophene-3-carboxylate | ||
Example 5.2
| TABLE 5 | ||
| Active | Application rate | Herbicidal action [%] against |
| compound(s) | [g of AS/ha] | Erodium cicutarium |
| (A21) | 0.8 | 25 |
| 0.2 | 0 | |
| (B1.2.1) | 50 | 30 |
| (A21) + (B1.2.1) | 0.2 + 50 | 75 |
| (EA = 0 + 30, EC = 0 + 30) | ||
| (B1.3) | 15 | 65 |
| (A21) + (B1.3) | 0.2 + 15 | 75 |
| (EA = 0 + 65, EC = 0 + 65) | ||
| (B1.4.1) | 5 | 50 |
| (A21) + (B1.4.1) | 0.2 + 5 | 68 |
| (EA = 0 + 50, EC = 0 + 50) | ||
| (B1.6.1) | 30 | 55 |
| (A21) + (B1.6.1) | 0.2 + 30 | 75 |
| (EA = 0 + 55, EC = 0 + 55) | ||
| Abbreviations and conditions in Table 5: Conditions: in each case pot trials/greenhouse, application by the pre-emergence method, evaluation 28 days after treatment | ||
| AS = active substance (based on 100% active compound)(A21) = compound (A21) from Table 1 = 2-amino-4-[(1R,2S)-2,6-dimethyl-indan-1-ylamino]-6-[(1R)-1-fluoroethyl]-1,3,5-triazine | ||
| (B1.2.1) = tembotrione = 2-{2-chloro-4-mesyl-3-[(2,2,2-trifluoroethoxy)methyl]-benzoyl}cyclohexane-1,3-dione | ||
| (B1.3) = ethyl [[3-[2-chloro-5-[3,6 dihydro-3-methyl-2,6-dioxo-4-(trifluoromethyl)-1-(2H)-pyrimidinyl]-4-fluorophenoxy]-2-pyridinyl]oxy]acetate | ||
| (B1.4.1) = N-(5,7-dimethoxy[1,2,4]triazolo[1,5-a]pyrimidin-2-yl)-2-methoxy-4-(trifluoromethyl)pyridine-3-sulfonamide | ||
| (B1.6.1) = 4-hydroxy-3-[[2-[(2-methoxyethoxy)methyl]-6-trifluoromethyl-3-pyridinyl]carbonyl]bicyclo[3.2.1]oct-3-en-2-one | ||
Example 5.3
| TABLE 6 | ||
| Active | Application rate | Herbicidal action [%] against |
| compound(s) | [g of AS/ha] | Geranium dissectum |
| (A21) | 1.2 | 20 |
| 0.3 | 5 | |
| (B2.1.1) | 30 | 20 |
| (A21) + (B2.1.1) | 0.3 + 30 | 65 |
| (EAA = 5 + 20, EC = 24) | ||
| (B2.2.2) | 25 | 45 |
| (A21) + (B2.2.2) | 0.3 + 25 | 60 |
| (EA = 5 + 45, EC = 47.8) | ||
| (B2.3.1) | 50 | 65 |
| (A21) + (B2.3.1) | 0.3 + 50 | 80 |
| (EA 5 + 65, EC = 66.8) | ||
| (B2.4.1) | 75 | 50 |
| (A21) + (B2.4.1) | 0.3 + 75 | 85 |
| (EA = 5 + 50, EC = 52.5) | ||
| (B2.5) | 300 | 35 |
| (A21) + (B2.5) | 0.3 + 300 | 55 |
| (EA = 5 + 35, EC = 38.3) | ||
| (B2.6.1) | 100 | 65 |
| (A21) + (B2.6.1) | 0.3 + 100 | 95 |
| (EA = 5 + 65, EC = 66.8) | ||
| (B3.1) | 50 | 50 |
| (A21) + (B3.1) | 0.3 + 50 | 75 |
| (EA = 5 + 50, EC = 52.5) | ||
| Abbreviations and conditions in Table 6: | ||
| Conditions: in each case pot trial/greenhouse, application by the pre-emergence method, evaluation 28 days after treatment | ||
| AS = active substance (based on 100% active compound) | ||
| (A21) = compound (A21) from Table 1 = 2-amino-4-[(1R,2S)-2, 6-dimethyl-indan-1-ylamino]6-[(1R)-1-fluoroethyl]-1,3,5-triazine | ||
| (B2.1.1) = pyrasulfotole = (5-hydroxy-1,3-dimethyl-1H-pyrazol-4-yl)-[2-(methylsulfonyl)-4-(trifiuoromethyl)phenyl]methanone | ||
| (B2.2.2) = trifloxysulfuron sodium salt = 1-(4,6-dimethoxypyrimidin-2-yl)-3-[3-(2,2,2-trifluoroethoxy)-2-pyridylsulfonyl]urea sodium salt | ||
| (B2.3.1) = saflufenacil = 2-chloro-5-[3,6-dihydro-3-methyl-2,6-dioxo-4-(trifluoromethyl)-1(2H)-pyrimidinyl]-4-fluoro-N-[[methyl(1-methylethyl)amino]sulfonyl]benzamide | ||
| (B2.4.1) = aminopyralid = 4-amino-3,6-dichloropyridine-2-carboxylic acid | ||
| (B2.5) = ethofumesate = (RS)—O-(2-ethoxy-2,3-dihydro-3,3-dimethylbenzofuran-5-yl) methanesulfonate | ||
| (B2.6.1) = aminocyclopyrachlor = 6-amino-5-chloro-2-cyclopropylpyrimidine-4-carboxylic acid | ||
| (B3.1) = pyroxasulfone = 3-[5-(difluoromethoxy)-1-methyl-3-(trifluoromethyl)-pyrazol-4-ylmethylsulfonyl]-4,5-dihydro-5,5-dimethyl-1,2-oxazole | ||
Claims (25)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/596,041 USRE46095E1 (en) | 2008-02-21 | 2015-01-13 | Herbicidal compositions |
| US15/084,041 USRE47666E1 (en) | 2008-02-21 | 2016-03-29 | Herbicidal compositions |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP08003156A EP2092825A1 (en) | 2008-02-21 | 2008-02-21 | Herbicidal combinations comprising a herbicide of the class of the diamino-s-triazines |
| EP08003156 | 2008-02-21 | ||
| US12/388,682 US8586503B2 (en) | 2008-02-21 | 2009-02-19 | Herbicidal compositions |
| US14/596,041 USRE46095E1 (en) | 2008-02-21 | 2015-01-13 | Herbicidal compositions |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/388,682 Reissue US8586503B2 (en) | 2008-02-21 | 2009-02-19 | Herbicidal compositions |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/388,682 Continuation US8586503B2 (en) | 2008-02-21 | 2009-02-19 | Herbicidal compositions |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USRE46095E1 true USRE46095E1 (en) | 2016-08-09 |
Family
ID=39688938
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/388,682 Ceased US8586503B2 (en) | 2008-02-21 | 2009-02-19 | Herbicidal compositions |
| US14/596,041 Active USRE46095E1 (en) | 2008-02-21 | 2015-01-13 | Herbicidal compositions |
| US15/084,041 Active USRE47666E1 (en) | 2008-02-21 | 2016-03-29 | Herbicidal compositions |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/388,682 Ceased US8586503B2 (en) | 2008-02-21 | 2009-02-19 | Herbicidal compositions |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/084,041 Active USRE47666E1 (en) | 2008-02-21 | 2016-03-29 | Herbicidal compositions |
Country Status (25)
| Country | Link |
|---|---|
| US (3) | US8586503B2 (en) |
| EP (1) | EP2092825A1 (en) |
| JP (1) | JP5577261B2 (en) |
| KR (1) | KR20100119866A (en) |
| CN (2) | CN105248428B (en) |
| AP (2) | AP2972A (en) |
| AR (1) | AR079087A1 (en) |
| AU (1) | AU2009216995B2 (en) |
| BR (1) | BRPI0907939A2 (en) |
| CA (1) | CA2716262C (en) |
| CL (1) | CL2009000396A1 (en) |
| CO (1) | CO6280581A2 (en) |
| CR (1) | CR11607A (en) |
| EA (1) | EA018407B9 (en) |
| IL (1) | IL207598A (en) |
| MA (1) | MA32092B1 (en) |
| MX (1) | MX2010009242A (en) |
| MY (1) | MY157222A (en) |
| NI (1) | NI201000118A (en) |
| NZ (1) | NZ587447A (en) |
| PE (1) | PE20091487A1 (en) |
| TW (1) | TWI468111B (en) |
| UA (1) | UA103016C2 (en) |
| WO (1) | WO2009103451A1 (en) |
| ZA (1) | ZA201004930B (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9822105B2 (en) | 2014-04-23 | 2017-11-21 | Basf Se | Diaminotriazine compounds as herbicides |
| US11800870B2 (en) | 2018-11-29 | 2023-10-31 | Discovery Purchaser Corporation | Herbicidal compositions for animal grazelands and methods for applying the same |
Families Citing this family (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2013312913B2 (en) | 2012-09-04 | 2016-12-08 | Corteva Agriscience Llc | Synergistic weed control from applications of aminocyclopyrachlor and clopyralid |
| CN104812248A (en) | 2012-09-28 | 2015-07-29 | 美国陶氏益农公司 | Synergistic weed control resulting from the application of cyprolacrim and clopyroxy |
| DE112013004795T5 (en) | 2012-09-28 | 2015-07-16 | Dow Agrosciences Llc | Synergistic weed control by applications of aminocyclopyrachlor and aminopyralid |
| WO2014052795A1 (en) | 2012-09-28 | 2014-04-03 | Dow Agrosciences Llc | Synergistic weed control from applications of aminocyclopyrachlor and fluroxypyr |
| US9426991B2 (en) | 2012-12-12 | 2016-08-30 | Dow Agrosciences Llc | Synergistic weed control from applications of aminocyclopyrachlor and picloram |
| US9149037B2 (en) | 2012-12-12 | 2015-10-06 | Dow Agrosciences Llc | Synergistic weed control from applications of aminocyclopyrachlor and 2,4 dichlorophenoxyacetic acid (2,4-D) |
| US10412964B2 (en) | 2012-12-14 | 2019-09-17 | Dow Agrosciences Llc | Synergistic weed control from applications of aminopyralid and clopyralid |
| AU2013360195B2 (en) | 2012-12-14 | 2017-05-04 | Corteva Agriscience Llc | Synergistic weed control from applications of aminopyralid and clopyralid |
| BR112015015055A2 (en) | 2012-12-21 | 2017-10-03 | Pioneer Hi Bred Int | METHOD FOR DETOXIFYING AN AUXIN ANALOG HERBICIDE, METHOD FOR CONTROLLING AT LEAST ONE WEED IN A GROWING AREA, METHOD FOR TESTING A PLANT RESPONSE TO ONE OR MORE COMPOUNDS |
| MX2015011043A (en) | 2013-02-25 | 2015-10-22 | Dow Agrosciences Llc | Methods of weed control in pineapple. |
| AU2014236162A1 (en) | 2013-03-14 | 2015-09-17 | Arzeda Corp. | Compositions having dicamba decarboxylase activity and methods of use |
| BR112015023272A2 (en) | 2013-03-14 | 2017-07-18 | Pioneer Hi Bred Int | plant cell, plant, plant explant, transgenic seed, method for producing a plant cell having a heterologous polynucleotide encoding a polypeptide having dicamba decarboxylase activity, method for controlling weeds in a field containing a crop and method for controlling weeds in a field containing a culture |
| US9999222B2 (en) * | 2014-04-23 | 2018-06-19 | Basf Se | Herbicidal combination comprising azines |
| CN104886082A (en) * | 2015-06-04 | 2015-09-09 | 广东中迅农科股份有限公司 | Weeding composition containing tembotrione and penoxsulam |
| AU2017301976B2 (en) | 2016-07-29 | 2022-04-07 | Basf Se | Method for controlling PPO resistant weeds |
| CA3030354A1 (en) * | 2016-07-29 | 2018-02-01 | Basf Se | Method for controlling ppo resistant weeds |
| JP6923838B2 (en) * | 2016-10-20 | 2021-08-25 | 日産化学株式会社 | Stabilized pesticide composition |
| US20200207734A1 (en) | 2017-08-03 | 2020-07-02 | Takeda Pharmaceutical Company Limited | Heterocyclic compound and use thereof |
| PY2047858A (en) * | 2019-08-26 | 2021-06-17 | Adama Agan Ltd | NOVEL SOLUBLE LIQUID COMPOSITIONS OF SAFLUFENACIL, METHOD OF PREPARATION AND USE OF THESE |
| CN114788525B (en) * | 2022-06-23 | 2022-09-16 | 山东康惠植物保护有限公司 | Composition containing Ephrifenacil and penoxsulam and application thereof |
| CN116439253A (en) * | 2023-03-28 | 2023-07-18 | 广东碧然美景观艺术有限公司 | Warm-season lawn weed control agent and application thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997031904A1 (en) | 1996-02-28 | 1997-09-04 | Hoechst Schering Agrevo Gmbh | 2-amino-4-bicycloamino-1,3,5-triazines as herbicides and plant growth regulators |
| WO2000016627A1 (en) | 1998-09-18 | 2000-03-30 | Aventis Cropscience Gmbh | Synergistic active substance combinations for controlling harmful plants |
| US20060014642A1 (en) * | 2004-07-17 | 2006-01-19 | Bayer Cropscience Gmbh | Herbicidal compositions |
| US8426342B2 (en) * | 2008-07-21 | 2013-04-23 | Bayer Cropscience Ag | Method for weed control in lawn |
Family Cites Families (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CH76704A (en) | 1917-06-23 | 1918-02-01 | Siemens Schuckertwerke Gmbh | Support frame for the effective parts of electricity meters, especially of induction meters |
| ATE103902T1 (en) | 1982-05-07 | 1994-04-15 | Ciba Geigy Ag | USE OF QUINOLINE DERIVATIVES TO PROTECT CROPS. |
| EP0131624B1 (en) | 1983-01-17 | 1992-09-16 | Monsanto Company | Plasmids for transforming plant cells |
| BR8404834A (en) | 1983-09-26 | 1985-08-13 | Agrigenetics Res Ass | METHOD TO GENETICALLY MODIFY A PLANT CELL |
| BR8600161A (en) | 1985-01-18 | 1986-09-23 | Plant Genetic Systems Nv | CHEMICAL GENE, HYBRID, INTERMEDIATE PLASMIDIO VECTORS, PROCESS TO CONTROL INSECTS IN AGRICULTURE OR HORTICULTURE, INSECTICIDE COMPOSITION, PROCESS TO TRANSFORM PLANT CELLS TO EXPRESS A PLANTINIDE TOXIN, PRODUCED BY CULTURES, UNITED BY BACILLA |
| ATE80182T1 (en) | 1985-10-25 | 1992-09-15 | Monsanto Co | PLANT VECTORS. |
| US5013659A (en) | 1987-07-27 | 1991-05-07 | E. I. Du Pont De Nemours And Company | Nucleic acid fragment encoding herbicide resistant plant acetolactate synthase |
| IL83348A (en) | 1986-08-26 | 1995-12-08 | Du Pont | Nucleic acid fragment encoding herbicide resistant plant acetolactate synthase |
| ATE241007T1 (en) | 1990-03-16 | 2003-06-15 | Calgene Llc | DNAS CODING FOR PLANT DESATURASES AND THEIR APPLICATIONS |
| EP0536293B1 (en) | 1990-06-18 | 2002-01-30 | Monsanto Technology LLC | Increased starch content in plants |
| SE467358B (en) | 1990-12-21 | 1992-07-06 | Amylogene Hb | GENETIC CHANGE OF POTATISE BEFORE EDUCATION OF AMYLOPECT TYPE STARCH |
| DE4104782B4 (en) | 1991-02-13 | 2006-05-11 | Bayer Cropscience Gmbh | Novel plasmids containing DNA sequences that cause changes in carbohydrate concentration and carbohydrate composition in plants, as well as plants and plant cells containing these plasmids |
| JPH08198712A (en) * | 1995-01-26 | 1996-08-06 | Idemitsu Kosan Co Ltd | Herbicide composition |
| US6071856A (en) | 1997-03-04 | 2000-06-06 | Zeneca Limited | Herbicidal compositions for acetochlor in rice |
| AU7527898A (en) | 1997-04-23 | 1998-11-13 | Novartis Ag | Herbicide |
| DE19727410A1 (en) | 1997-06-27 | 1999-01-07 | Hoechst Schering Agrevo Gmbh | 3- (5-tetrazolylcarbonyl) -2-quinolones and crop protection agents containing them |
| DE19742951A1 (en) | 1997-09-29 | 1999-04-15 | Hoechst Schering Agrevo Gmbh | Acylsulfamoylbenzoic acid amides, crop protection agents containing them and process for their preparation |
| DE19846792A1 (en) | 1998-10-10 | 2000-04-13 | Hoechst Schering Agrevo Gmbh | New benzoyl-cycloalkanone and benzoyl-cycloalkanedione derivatives useful as herbicides, especially for selective weed control in crops, and plant growth regulators |
| DE19933260A1 (en) | 1999-07-15 | 2001-01-18 | Bayer Ag | Substituted thien-3-yl-sulfonylamino (thio) carbonyl-triazolin (thi) one |
| IL167958A (en) | 2000-02-04 | 2010-11-30 | Sumitomo Chemical Co | 2-thio 3-hydroxypyridine derivatives |
| HRP20020200B1 (en) | 2000-05-04 | 2013-06-30 | Basf Aktiengesellschaft | PHENYL SULFAMOIL CARBOXAMIDES SUBSTITUTED BY URACIL |
| GT200100103A (en) | 2000-06-09 | 2002-02-21 | NEW HERBICIDES | |
| RU15525U1 (en) | 2000-07-04 | 2000-10-20 | Федеральный научно-производственный центр закрытое акционерное общество "Научно-производственный концерн (объединение) "ЭНЕРГИЯ" | DC MOTOR |
| AU2002242701B2 (en) | 2001-01-31 | 2008-04-03 | Bayer Intellectual Property Gmbh | Herbicide-safener combination based on isoxozoline carboxylate safeners |
| DE10145019A1 (en) | 2001-09-13 | 2003-04-03 | Bayer Cropscience Gmbh | Combinations of herbicides and safeners |
| UA78071C2 (en) | 2002-08-07 | 2007-02-15 | Kumiai Chemical Industry Co | Herbicidal composition |
| CN1761395A (en) * | 2003-03-13 | 2006-04-19 | 巴斯福股份公司 | Synergistic herbicidal mixture |
| EP1689227B1 (en) | 2003-12-04 | 2014-01-15 | Syngenta Participations AG | Herbicidal composition |
| AU2005237241B2 (en) | 2004-04-30 | 2011-04-07 | Syngenta Limited | Herbicidal composition |
| TW200621157A (en) | 2004-12-06 | 2006-07-01 | Syngenta Participations Ag | Herbicidal composition |
| GB0505645D0 (en) | 2005-03-18 | 2005-04-27 | Syngenta Participations Ag | Herbicidal composition |
| JP2007186460A (en) * | 2006-01-13 | 2007-07-26 | Bayer Cropscience Kk | Herbicidal composition for rice field |
-
2008
- 2008-02-21 EP EP08003156A patent/EP2092825A1/en not_active Withdrawn
-
2009
- 2009-02-12 MX MX2010009242A patent/MX2010009242A/en active IP Right Grant
- 2009-02-12 CN CN201510398311.2A patent/CN105248428B/en active Active
- 2009-02-12 BR BRPI0907939-4A patent/BRPI0907939A2/en not_active Application Discontinuation
- 2009-02-12 WO PCT/EP2009/000962 patent/WO2009103451A1/en not_active Ceased
- 2009-02-12 NZ NZ587447A patent/NZ587447A/en unknown
- 2009-02-12 CN CN2009801060632A patent/CN101951774A/en active Pending
- 2009-02-12 MY MYPI2010003308A patent/MY157222A/en unknown
- 2009-02-12 KR KR1020107018131A patent/KR20100119866A/en not_active Abandoned
- 2009-02-12 EA EA201001177A patent/EA018407B9/en not_active IP Right Cessation
- 2009-02-12 AP AP2010005352A patent/AP2972A/en active
- 2009-02-12 AU AU2009216995A patent/AU2009216995B2/en active Active
- 2009-02-12 JP JP2010547084A patent/JP5577261B2/en active Active
- 2009-02-12 AP AP2013007162A patent/AP3262A/en active
- 2009-02-12 CA CA2716262A patent/CA2716262C/en active Active
- 2009-02-19 AR ARP090100578A patent/AR079087A1/en unknown
- 2009-02-19 TW TW98105274A patent/TWI468111B/en active
- 2009-02-19 US US12/388,682 patent/US8586503B2/en not_active Ceased
- 2009-02-20 CL CL2009000396A patent/CL2009000396A1/en unknown
- 2009-02-20 PE PE2009000252A patent/PE20091487A1/en not_active Application Discontinuation
- 2009-12-02 UA UAA201010264A patent/UA103016C2/en unknown
-
2010
- 2010-07-12 NI NI201000118A patent/NI201000118A/en unknown
- 2010-07-13 ZA ZA2010/04930A patent/ZA201004930B/en unknown
- 2010-07-28 CR CR11607A patent/CR11607A/en not_active Application Discontinuation
- 2010-08-12 IL IL207598A patent/IL207598A/en active IP Right Grant
- 2010-08-19 MA MA33103A patent/MA32092B1/en unknown
- 2010-08-19 CO CO10102310A patent/CO6280581A2/en not_active Application Discontinuation
-
2015
- 2015-01-13 US US14/596,041 patent/USRE46095E1/en active Active
-
2016
- 2016-03-29 US US15/084,041 patent/USRE47666E1/en active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997031904A1 (en) | 1996-02-28 | 1997-09-04 | Hoechst Schering Agrevo Gmbh | 2-amino-4-bicycloamino-1,3,5-triazines as herbicides and plant growth regulators |
| WO2000016627A1 (en) | 1998-09-18 | 2000-03-30 | Aventis Cropscience Gmbh | Synergistic active substance combinations for controlling harmful plants |
| CA2344394A1 (en) | 1998-09-18 | 2000-03-30 | Aventis Cropscience Gmbh | Synergistic active compound combinations for controlling harmful plants |
| US20060014642A1 (en) * | 2004-07-17 | 2006-01-19 | Bayer Cropscience Gmbh | Herbicidal compositions |
| WO2006007947A1 (en) | 2004-07-17 | 2006-01-26 | Bayer Cropscience Gmbh | Herbicidal agents |
| US20060019829A1 (en) | 2004-07-17 | 2006-01-26 | Erwin Hacker | Herbicidal compositions |
| US8426342B2 (en) * | 2008-07-21 | 2013-04-23 | Bayer Cropscience Ag | Method for weed control in lawn |
Non-Patent Citations (1)
| Title |
|---|
| Mannschreck et al. J. of Chemical Education v.84(12), 2007, 2012-2018. * |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9822105B2 (en) | 2014-04-23 | 2017-11-21 | Basf Se | Diaminotriazine compounds as herbicides |
| US11800870B2 (en) | 2018-11-29 | 2023-10-31 | Discovery Purchaser Corporation | Herbicidal compositions for animal grazelands and methods for applying the same |
| US11849730B2 (en) | 2018-11-29 | 2023-12-26 | Discovery Purchaser Corporation | Herbicidal compositions for animal grazelands and methods for applying the same |
| US11896011B2 (en) | 2018-11-29 | 2024-02-13 | Discovery Purchaser Corporation | Herbicidal compositions for animal grazelands and methods for applying the same |
| US11917996B2 (en) | 2018-11-29 | 2024-03-05 | Discovery Purchaser Corporation | Herbicidal compositions for animal grazelands and methods for applying the same |
| US12310367B2 (en) | 2018-11-29 | 2025-05-27 | Discovery Purchaser Corporation | Herbicidal compositions for animal grazelands and methods for applying the same |
| US12310366B2 (en) | 2018-11-29 | 2025-05-27 | Discovery Purchaser Corporation | Herbicidal compositions for animal grazelands and methods for applying the same |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| USRE47666E1 (en) | Herbicidal compositions | |
| US8716184B2 (en) | Herbicidal composition comprising the hydrates of saflufenacil and glyphosate or glufosinate | |
| US8236729B2 (en) | Herbicidal compositions | |
| AU2014201131B2 (en) | Herbicide combinations containing a herbicide from the class of diamino-s-triazines |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| AS | Assignment |
Owner name: BAYER CROPSCIENCE AKTIENGESELLSCHAFT, GERMANY Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:BAYER INTELLECTUAL PROPERTY GMBH;REEL/FRAME:049359/0698 Effective date: 20181214 Owner name: BAYER CROPSCIENCE AG, GERMANY Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:HACKER, ERWIN;HESS, MARTIN;HILLS, MARTIN JEFFREY;AND OTHERS;SIGNING DATES FROM 20090305 TO 20090324;REEL/FRAME:047967/0064 Owner name: BAYER INTELLECTUAL PROPERTY GMBH, GERMANY Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:BAYER CROPSCIENCE AG;REEL/FRAME:047967/0117 Effective date: 20130722 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 8TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1552); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 8 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 12TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1553); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 12 |