US8841265B2 - Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof - Google Patents
Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof Download PDFInfo
- Publication number
- US8841265B2 US8841265B2 US12/344,682 US34468208A US8841265B2 US 8841265 B2 US8841265 B2 US 8841265B2 US 34468208 A US34468208 A US 34468208A US 8841265 B2 US8841265 B2 US 8841265B2
- Authority
- US
- United States
- Prior art keywords
- group
- angeloyl
- acetyl
- compound
- sugar moiety
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related, expires
Links
- 125000005601 angeloyl group Chemical group 0.000 title claims description 292
- 150000001875 compounds Chemical class 0.000 title claims description 280
- 238000000034 method Methods 0.000 title claims description 50
- 239000000203 mixture Substances 0.000 title abstract description 34
- 125000000524 functional group Chemical group 0.000 title description 7
- 229930182493 triterpene saponin Natural products 0.000 title description 2
- 230000002401 inhibitory effect Effects 0.000 claims abstract description 50
- 206010061535 Ovarian neoplasm Diseases 0.000 claims abstract description 26
- 208000000453 Skin Neoplasms Diseases 0.000 claims abstract description 13
- 235000000346 sugar Nutrition 0.000 claims description 240
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 claims description 188
- 125000002252 acyl group Chemical group 0.000 claims description 173
- SRBFZHDQGSBBOR-UHFFFAOYSA-N beta-D-Pyranose-Lyxose Natural products OC1COC(O)C(O)C1O SRBFZHDQGSBBOR-UHFFFAOYSA-N 0.000 claims description 112
- WQZGKKKJIJFFOK-SVZMEOIVSA-N (+)-Galactose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-SVZMEOIVSA-N 0.000 claims description 100
- IAJILQKETJEXLJ-UHFFFAOYSA-N Galacturonsaeure Natural products O=CC(O)C(O)C(O)C(O)C(O)=O IAJILQKETJEXLJ-UHFFFAOYSA-N 0.000 claims description 92
- -1 alkenoyl Chemical group 0.000 claims description 74
- SHZGCJCMOBCMKK-UHFFFAOYSA-N D-mannomethylose Natural products CC1OC(O)C(O)C(O)C1O SHZGCJCMOBCMKK-UHFFFAOYSA-N 0.000 claims description 69
- 239000002253 acid Substances 0.000 claims description 69
- 125000000217 alkyl group Chemical group 0.000 claims description 67
- CBOIHMRHGLHBPB-UHFFFAOYSA-N hydroxymethyl Chemical compound O[CH2] CBOIHMRHGLHBPB-UHFFFAOYSA-N 0.000 claims description 66
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 claims description 65
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 64
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 claims description 62
- PNNNRSAQSRJVSB-UHFFFAOYSA-N L-rhamnose Natural products CC(O)C(O)C(O)C(O)C=O PNNNRSAQSRJVSB-UHFFFAOYSA-N 0.000 claims description 58
- IAJILQKETJEXLJ-RSJOWCBRSA-N aldehydo-D-galacturonic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-RSJOWCBRSA-N 0.000 claims description 46
- AEMOLEFTQBMNLQ-DTEWXJGMSA-N D-Galacturonic acid Natural products O[C@@H]1O[C@H](C(O)=O)[C@H](O)[C@H](O)[C@H]1O AEMOLEFTQBMNLQ-DTEWXJGMSA-N 0.000 claims description 37
- AEMOLEFTQBMNLQ-WAXACMCWSA-N alpha-D-glucuronic acid Chemical compound O[C@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O AEMOLEFTQBMNLQ-WAXACMCWSA-N 0.000 claims description 37
- AEMOLEFTQBMNLQ-UHFFFAOYSA-N beta-D-galactopyranuronic acid Natural products OC1OC(C(O)=O)C(O)C(O)C1O AEMOLEFTQBMNLQ-UHFFFAOYSA-N 0.000 claims description 37
- PYMYPHUHKUWMLA-UHFFFAOYSA-N arabinose Natural products OCC(O)C(O)C(O)C=O PYMYPHUHKUWMLA-UHFFFAOYSA-N 0.000 claims description 27
- 206010033128 Ovarian cancer Diseases 0.000 claims description 25
- 125000001488 beta-D-galactosyl group Chemical group C1([C@H](O)[C@@H](O)[C@@H](O)[C@H](O1)CO)* 0.000 claims description 25
- SHZGCJCMOBCMKK-JFNONXLTSA-N L-rhamnopyranose Chemical group C[C@@H]1OC(O)[C@H](O)[C@H](O)[C@H]1O SHZGCJCMOBCMKK-JFNONXLTSA-N 0.000 claims description 18
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 claims description 16
- 229930182830 galactose Natural products 0.000 claims description 16
- 239000008103 glucose Substances 0.000 claims description 16
- 150000003839 salts Chemical class 0.000 claims description 16
- PYMYPHUHKUWMLA-WDCZJNDASA-N arabinose Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)C=O PYMYPHUHKUWMLA-WDCZJNDASA-N 0.000 claims description 15
- 229910052739 hydrogen Inorganic materials 0.000 claims description 15
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 15
- 150000002148 esters Chemical class 0.000 claims description 14
- 239000001257 hydrogen Substances 0.000 claims description 13
- 201000000849 skin cancer Diseases 0.000 claims description 12
- 230000010261 cell growth Effects 0.000 claims description 9
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 6
- DCKVNWZUADLDEH-UHFFFAOYSA-N sec-butyl acetate Chemical compound CCC(C)OC(C)=O DCKVNWZUADLDEH-UHFFFAOYSA-N 0.000 claims description 3
- 125000000328 arabinofuranosyl group Chemical group C1([C@@H](O)[C@H](O)[C@H](O1)CO)* 0.000 claims 3
- SRBFZHDQGSBBOR-HWQSCIPKSA-N L-arabinopyranose Chemical compound O[C@H]1COC(O)[C@H](O)[C@H]1O SRBFZHDQGSBBOR-HWQSCIPKSA-N 0.000 claims 1
- 125000005640 glucopyranosyl group Chemical group 0.000 claims 1
- 230000002611 ovarian Effects 0.000 claims 1
- 150000008130 triterpenoid saponins Chemical class 0.000 abstract description 25
- 230000004565 tumor cell growth Effects 0.000 abstract description 10
- 206010029098 Neoplasm skin Diseases 0.000 abstract 1
- 125000005607 tigloyl group Chemical group 0.000 description 165
- 229930182490 saponin Natural products 0.000 description 79
- 235000017709 saponins Nutrition 0.000 description 79
- 150000007949 saponins Chemical class 0.000 description 75
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 65
- SRBFZHDQGSBBOR-IOVATXLUSA-N D-xylopyranose Chemical compound O[C@@H]1COC(O)[C@H](O)[C@H]1O SRBFZHDQGSBBOR-IOVATXLUSA-N 0.000 description 62
- 125000003118 aryl group Chemical group 0.000 description 55
- SRBFZHDQGSBBOR-SOOFDHNKSA-N D-ribopyranose Chemical compound O[C@@H]1COC(O)[C@H](O)[C@@H]1O SRBFZHDQGSBBOR-SOOFDHNKSA-N 0.000 description 54
- 125000000896 monocarboxylic acid group Chemical group 0.000 description 53
- PNNNRSAQSRJVSB-BXKVDMCESA-N aldehydo-L-rhamnose Chemical compound C[C@H](O)[C@H](O)[C@@H](O)[C@@H](O)C=O PNNNRSAQSRJVSB-BXKVDMCESA-N 0.000 description 40
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 36
- 201000003176 Severe Acute Respiratory Syndrome Diseases 0.000 description 34
- 241000725303 Human immunodeficiency virus Species 0.000 description 31
- WURBFLDFSFBTLW-UHFFFAOYSA-N benzil Chemical group C=1C=CC=CC=1C(=O)C(=O)C1=CC=CC=C1 WURBFLDFSFBTLW-UHFFFAOYSA-N 0.000 description 30
- 239000001397 quillaja saponaria molina bark Substances 0.000 description 30
- 206010028980 Neoplasm Diseases 0.000 description 29
- 241001092090 Pittosporum Species 0.000 description 29
- 210000004027 cell Anatomy 0.000 description 29
- 201000011510 cancer Diseases 0.000 description 28
- 230000006870 function Effects 0.000 description 26
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 25
- 229910052799 carbon Inorganic materials 0.000 description 25
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 24
- 229940079593 drug Drugs 0.000 description 22
- 239000003814 drug Substances 0.000 description 22
- 239000000284 extract Substances 0.000 description 22
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 21
- 0 *[C@H]1CC[C@@]2(C)C(CC[C@]3(C)C2CC=C2C4CC(C)(C)[C@@H](C)[C@H](C)[C@]4([4*])[C@H](O)[C@H]([3*])[C@]23C)C1(C)C Chemical compound *[C@H]1CC[C@@]2(C)C(CC[C@]3(C)C2CC=C2C4CC(C)(C)[C@@H](C)[C@H](C)[C@]4([4*])[C@H](O)[C@H]([3*])[C@]23C)C1(C)C 0.000 description 20
- 230000000694 effects Effects 0.000 description 20
- 241001629515 Harpullia Species 0.000 description 18
- 206010022000 influenza Diseases 0.000 description 18
- 206010005003 Bladder cancer Diseases 0.000 description 17
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 description 17
- 201000005112 urinary bladder cancer Diseases 0.000 description 17
- XBZYWSMVVKYHQN-MYPRUECHSA-N (4as,6as,6br,8ar,9r,10s,12ar,12br,14bs)-10-hydroxy-2,2,6a,6b,9,12a-hexamethyl-9-[(sulfooxy)methyl]-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-4a-carboxylic acid Chemical compound C1C[C@H](O)[C@@](C)(COS(O)(=O)=O)[C@@H]2CC[C@@]3(C)[C@]4(C)CC[C@@]5(C(O)=O)CCC(C)(C)C[C@H]5C4=CC[C@@H]3[C@]21C XBZYWSMVVKYHQN-MYPRUECHSA-N 0.000 description 16
- 206010005949 Bone cancer Diseases 0.000 description 16
- 208000018084 Bone neoplasm Diseases 0.000 description 16
- 241001385730 Aesculus assamica Species 0.000 description 15
- 150000008163 sugars Chemical class 0.000 description 14
- 241000157282 Aesculus Species 0.000 description 13
- 241000196324 Embryophyta Species 0.000 description 13
- 241000700605 Viruses Species 0.000 description 13
- 238000012217 deletion Methods 0.000 description 12
- 230000037430 deletion Effects 0.000 description 12
- 238000006467 substitution reaction Methods 0.000 description 12
- 230000003612 virological effect Effects 0.000 description 12
- PNNNRSAQSRJVSB-SLPGGIOYSA-N Fucose Natural products C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C=O PNNNRSAQSRJVSB-SLPGGIOYSA-N 0.000 description 11
- SHZGCJCMOBCMKK-DHVFOXMCSA-N L-fucopyranose Chemical compound C[C@@H]1OC(O)[C@@H](O)[C@H](O)[C@@H]1O SHZGCJCMOBCMKK-DHVFOXMCSA-N 0.000 description 11
- 230000005907 cancer growth Effects 0.000 description 11
- 238000002474 experimental method Methods 0.000 description 11
- 238000011282 treatment Methods 0.000 description 10
- 241001635755 Aesculus flava Species 0.000 description 9
- 241001156402 Allophylus Species 0.000 description 9
- IAJILQKETJEXLJ-QTBDOELSSA-N aldehydo-D-glucuronic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-QTBDOELSSA-N 0.000 description 9
- 229940117173 croton oil Drugs 0.000 description 9
- WQLVFSAGQJTQCK-UHFFFAOYSA-N diosgenin Natural products CC1C(C2(CCC3C4(C)CCC(O)CC4=CCC3C2C2)C)C2OC11CCC(C)CO1 WQLVFSAGQJTQCK-UHFFFAOYSA-N 0.000 description 9
- 229940097043 glucuronic acid Drugs 0.000 description 9
- 230000005764 inhibitory process Effects 0.000 description 9
- NWMIYTWHUDFRPL-UHFFFAOYSA-N sapogenin Natural products COC(=O)C1(CO)C(O)CCC2(C)C1CCC3(C)C2CC=C4C5C(C)(O)C(C)CCC5(CCC34C)C(=O)O NWMIYTWHUDFRPL-UHFFFAOYSA-N 0.000 description 9
- 241001119624 Aesculus chinensis Species 0.000 description 8
- WQZGKKKJIJFFOK-CBPJZXOFSA-N D-Gulose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@H](O)[C@H]1O WQZGKKKJIJFFOK-CBPJZXOFSA-N 0.000 description 8
- WQZGKKKJIJFFOK-WHZQZERISA-N D-aldose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-WHZQZERISA-N 0.000 description 8
- WQZGKKKJIJFFOK-IVMDWMLBSA-N D-allopyranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@H](O)[C@@H]1O WQZGKKKJIJFFOK-IVMDWMLBSA-N 0.000 description 8
- LKDRXBCSQODPBY-JDJSBBGDSA-N D-allulose Chemical compound OCC1(O)OC[C@@H](O)[C@@H](O)[C@H]1O LKDRXBCSQODPBY-JDJSBBGDSA-N 0.000 description 8
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 8
- 229930091371 Fructose Natural products 0.000 description 8
- 239000005715 Fructose Substances 0.000 description 8
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 8
- LKDRXBCSQODPBY-AMVSKUEXSA-N L-(-)-Sorbose Chemical compound OCC1(O)OC[C@H](O)[C@@H](O)[C@@H]1O LKDRXBCSQODPBY-AMVSKUEXSA-N 0.000 description 8
- WQZGKKKJIJFFOK-VSOAQEOCSA-N L-altropyranose Chemical compound OC[C@@H]1OC(O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-VSOAQEOCSA-N 0.000 description 8
- 231100000002 MTT assay Toxicity 0.000 description 8
- 238000000134 MTT assay Methods 0.000 description 8
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 8
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 8
- SRBFZHDQGSBBOR-STGXQOJASA-N alpha-D-lyxopyranose Chemical compound O[C@@H]1CO[C@H](O)[C@@H](O)[C@H]1O SRBFZHDQGSBBOR-STGXQOJASA-N 0.000 description 8
- 230000015556 catabolic process Effects 0.000 description 8
- 125000002951 idosyl group Chemical class C1([C@@H](O)[C@H](O)[C@@H](O)[C@H](O1)CO)* 0.000 description 8
- BJHIKXHVCXFQLS-PQLUHFTBSA-N keto-D-tagatose Chemical compound OC[C@@H](O)[C@H](O)[C@H](O)C(=O)CO BJHIKXHVCXFQLS-PQLUHFTBSA-N 0.000 description 8
- 241000700159 Rattus Species 0.000 description 7
- 230000012010 growth Effects 0.000 description 7
- 239000002207 metabolite Substances 0.000 description 7
- 150000003648 triterpenes Chemical class 0.000 description 7
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- 206010042674 Swelling Diseases 0.000 description 6
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 6
- 238000010521 absorption reaction Methods 0.000 description 6
- 238000011319 anticancer therapy Methods 0.000 description 6
- 230000000975 bioactive effect Effects 0.000 description 6
- 230000008827 biological function Effects 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 235000010418 carrageenan Nutrition 0.000 description 6
- 229920001525 carrageenan Polymers 0.000 description 6
- 238000004128 high performance liquid chromatography Methods 0.000 description 6
- 239000000419 plant extract Substances 0.000 description 6
- 102200082402 rs751610198 Human genes 0.000 description 6
- 230000008961 swelling Effects 0.000 description 6
- 208000024891 symptom Diseases 0.000 description 6
- 201000002282 venous insufficiency Diseases 0.000 description 6
- 206010030124 Oedema peripheral Diseases 0.000 description 5
- 230000000259 anti-tumor effect Effects 0.000 description 5
- 239000013642 negative control Substances 0.000 description 5
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 5
- 241000209507 Camellia Species 0.000 description 4
- 241001119537 Lepisanthes Species 0.000 description 4
- 238000005481 NMR spectroscopy Methods 0.000 description 4
- 241000167562 Pittosporum tobira Species 0.000 description 4
- 241000580938 Sapindus Species 0.000 description 4
- OIPILFWXSMYKGL-UHFFFAOYSA-N acetylcholine Chemical compound CC(=O)OCC[N+](C)(C)C OIPILFWXSMYKGL-UHFFFAOYSA-N 0.000 description 4
- UCTWMZQNUQWSLP-UHFFFAOYSA-N adrenaline Chemical compound CNCC(O)C1=CC=C(O)C(O)=C1 UCTWMZQNUQWSLP-UHFFFAOYSA-N 0.000 description 4
- 230000002490 cerebral effect Effects 0.000 description 4
- 238000004587 chromatography analysis Methods 0.000 description 4
- 235000018597 common camellia Nutrition 0.000 description 4
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N coumarin Chemical compound C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 4
- 239000000287 crude extract Substances 0.000 description 4
- VYFYYTLLBUKUHU-UHFFFAOYSA-N dopamine Chemical compound NCCC1=CC=C(O)C(O)=C1 VYFYYTLLBUKUHU-UHFFFAOYSA-N 0.000 description 4
- 238000010828 elution Methods 0.000 description 4
- 230000002949 hemolytic effect Effects 0.000 description 4
- 125000000623 heterocyclic group Chemical group 0.000 description 4
- 210000000265 leukocyte Anatomy 0.000 description 4
- 239000003960 organic solvent Substances 0.000 description 4
- 239000013641 positive control Substances 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 230000001105 regulatory effect Effects 0.000 description 4
- 239000000523 sample Substances 0.000 description 4
- 230000004614 tumor growth Effects 0.000 description 4
- 239000003643 water by type Substances 0.000 description 4
- 241000208140 Acer Species 0.000 description 3
- 241000954717 Aesculus californica Species 0.000 description 3
- 241001385729 Aesculus chinensis var. wilsonii Species 0.000 description 3
- 241001635754 Aesculus glabra Species 0.000 description 3
- 241001635752 Aesculus glabra var. arguta Species 0.000 description 3
- 241000157280 Aesculus hippocastanum Species 0.000 description 3
- 241001385733 Aesculus indica Species 0.000 description 3
- 241000159211 Aesculus pavia Species 0.000 description 3
- 241001385727 Aesculus polyneura Species 0.000 description 3
- 241001635753 Aesculus sylvatica Species 0.000 description 3
- 241001646648 Aesculus turbinata Species 0.000 description 3
- 241000519336 Amesiodendron Species 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 208000003174 Brain Neoplasms Diseases 0.000 description 3
- 206010048962 Brain oedema Diseases 0.000 description 3
- 101800000414 Corticotropin Proteins 0.000 description 3
- 240000005153 Dodonaea viscosa Species 0.000 description 3
- 241000612162 Maesa Species 0.000 description 3
- 241000781625 Maesa lanceolata Species 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 241001308575 Neglecta Species 0.000 description 3
- 241000557736 Paranephelium Species 0.000 description 3
- 241000903504 Pavieasia Species 0.000 description 3
- 241000781597 Pittosporum viridiflorum Species 0.000 description 3
- 102100027467 Pro-opiomelanocortin Human genes 0.000 description 3
- 206010046996 Varicose vein Diseases 0.000 description 3
- 108010004977 Vasopressins Proteins 0.000 description 3
- 102000002852 Vasopressins Human genes 0.000 description 3
- 244000248162 Xanthoceras sorbifolium Species 0.000 description 3
- 235000009240 Xanthoceras sorbifolium Nutrition 0.000 description 3
- 125000003342 alkenyl group Chemical group 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- KBZOIRJILGZLEJ-LGYYRGKSSA-N argipressin Chemical compound C([C@H]1C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CSSC[C@@H](C(N[C@@H](CC=2C=CC(O)=CC=2)C(=O)N1)=O)N)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)NCC(N)=O)C1=CC=CC=C1 KBZOIRJILGZLEJ-LGYYRGKSSA-N 0.000 description 3
- 210000000988 bone and bone Anatomy 0.000 description 3
- 208000006752 brain edema Diseases 0.000 description 3
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- 208000037998 chronic venous disease Diseases 0.000 description 3
- 201000002816 chronic venous insufficiency Diseases 0.000 description 3
- IDLFZVILOHSSID-OVLDLUHVSA-N corticotropin Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(O)=O)NC(=O)[C@@H](N)CO)C1=CC=C(O)C=C1 IDLFZVILOHSSID-OVLDLUHVSA-N 0.000 description 3
- 229960000258 corticotropin Drugs 0.000 description 3
- 235000013399 edible fruits Nutrition 0.000 description 3
- 208000014617 hemorrhoid Diseases 0.000 description 3
- 235000010181 horse chestnut Nutrition 0.000 description 3
- 239000010903 husk Substances 0.000 description 3
- 208000010729 leg swelling Diseases 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 210000001672 ovary Anatomy 0.000 description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 3
- 230000002980 postoperative effect Effects 0.000 description 3
- 238000001228 spectrum Methods 0.000 description 3
- 208000011580 syndromic disease Diseases 0.000 description 3
- 229960003726 vasopressin Drugs 0.000 description 3
- SFLSHLFXELFNJZ-QMMMGPOBSA-N (-)-norepinephrine Chemical compound NC[C@H](O)C1=CC=C(O)C(O)=C1 SFLSHLFXELFNJZ-QMMMGPOBSA-N 0.000 description 2
- 239000000275 Adrenocorticotropic Hormone Substances 0.000 description 2
- 241001635751 Aesculus parviflora Species 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- 206010006187 Breast cancer Diseases 0.000 description 2
- 208000026310 Breast neoplasm Diseases 0.000 description 2
- 235000014143 Camellia sinensis var assamica Nutrition 0.000 description 2
- 240000008441 Camellia sinensis var. assamica Species 0.000 description 2
- 206010010904 Convulsion Diseases 0.000 description 2
- NZNMSOFKMUBTKW-UHFFFAOYSA-N Cyclohexanecarboxylic acid Natural products OC(=O)C1CCCCC1 NZNMSOFKMUBTKW-UHFFFAOYSA-N 0.000 description 2
- 125000002353 D-glucosyl group Chemical group C1([C@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO)* 0.000 description 2
- 241000613447 Dimocarpus Species 0.000 description 2
- 235000000522 Dimocarpus Nutrition 0.000 description 2
- 240000001008 Dimocarpus longan Species 0.000 description 2
- 241000158421 Dipteronia sinensis Species 0.000 description 2
- 235000009919 Dodonaea viscosa subsp angustifolia Nutrition 0.000 description 2
- 238000002965 ELISA Methods 0.000 description 2
- 208000008967 Enuresis Diseases 0.000 description 2
- 235000000235 Euphoria longan Nutrition 0.000 description 2
- 241001676753 Eurycorymbus Species 0.000 description 2
- 241001119562 Handeliodendron Species 0.000 description 2
- 241000872192 Hypelate Species 0.000 description 2
- 241000167834 Koelreuteria Species 0.000 description 2
- 241001533112 Koelreuteria bipinnata Species 0.000 description 2
- 241000167847 Koelreuteria paniculata Species 0.000 description 2
- 241000536286 Melicoccus Species 0.000 description 2
- 235000017285 Melicoccus Nutrition 0.000 description 2
- 241001119530 Mischocarpus Species 0.000 description 2
- 240000006849 Mischocarpus pentapetalus Species 0.000 description 2
- 241001629494 Nephelium Species 0.000 description 2
- 244000183278 Nephelium litchi Species 0.000 description 2
- 208000002193 Pain Diseases 0.000 description 2
- 206010033425 Pain in extremity Diseases 0.000 description 2
- 208000018262 Peripheral vascular disease Diseases 0.000 description 2
- 241001533116 Pittosporum glabratum Species 0.000 description 2
- 241001573294 Pittosporum illicioides Species 0.000 description 2
- 206010060862 Prostate cancer Diseases 0.000 description 2
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 2
- 208000003251 Pruritus Diseases 0.000 description 2
- 208000028017 Psychotic disease Diseases 0.000 description 2
- 241001523328 Ptelea Species 0.000 description 2
- 239000012980 RPMI-1640 medium Substances 0.000 description 2
- 240000001190 Sapindus rarak Species 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 208000007107 Stomach Ulcer Diseases 0.000 description 2
- 244000269722 Thea sinensis Species 0.000 description 2
- 235000006468 Thea sinensis Nutrition 0.000 description 2
- 208000007536 Thrombosis Diseases 0.000 description 2
- 241000408907 Xanthoceras Species 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 229960004373 acetylcholine Drugs 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- AFVLVVWMAFSXCK-VMPITWQZSA-N alpha-cyano-4-hydroxycinnamic acid Chemical compound OC(=O)C(\C#N)=C\C1=CC=C(O)C=C1 AFVLVVWMAFSXCK-VMPITWQZSA-N 0.000 description 2
- 239000003242 anti bacterial agent Substances 0.000 description 2
- 230000001093 anti-cancer Effects 0.000 description 2
- 230000002402 anti-lipaemic effect Effects 0.000 description 2
- 230000002155 anti-virotic effect Effects 0.000 description 2
- 229940088710 antibiotic agent Drugs 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 210000000481 breast Anatomy 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 210000003679 cervix uteri Anatomy 0.000 description 2
- 210000001072 colon Anatomy 0.000 description 2
- 230000036461 convulsion Effects 0.000 description 2
- 229960000956 coumarin Drugs 0.000 description 2
- 235000001671 coumarin Nutrition 0.000 description 2
- 231100000135 cytotoxicity Toxicity 0.000 description 2
- 230000003013 cytotoxicity Effects 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 239000002934 diuretic Substances 0.000 description 2
- 229960003638 dopamine Drugs 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 238000011067 equilibration Methods 0.000 description 2
- 239000003172 expectorant agent Substances 0.000 description 2
- 230000003419 expectorant effect Effects 0.000 description 2
- 239000012894 fetal calf serum Substances 0.000 description 2
- 238000005194 fractionation Methods 0.000 description 2
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 2
- 239000001963 growth medium Substances 0.000 description 2
- 125000005842 heteroatom Chemical group 0.000 description 2
- 238000003919 heteronuclear multiple bond coherence Methods 0.000 description 2
- 238000003929 heteronuclear multiple quantum coherence Methods 0.000 description 2
- 238000002955 isolation Methods 0.000 description 2
- 210000004185 liver Anatomy 0.000 description 2
- 201000007270 liver cancer Diseases 0.000 description 2
- 208000014018 liver neoplasm Diseases 0.000 description 2
- 210000004072 lung Anatomy 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 238000001840 matrix-assisted laser desorption--ionisation time-of-flight mass spectrometry Methods 0.000 description 2
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 2
- 230000027939 micturition Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- VMGAPWLDMVPYIA-HIDZBRGKSA-N n'-amino-n-iminomethanimidamide Chemical compound N\N=C\N=N VMGAPWLDMVPYIA-HIDZBRGKSA-N 0.000 description 2
- 229960002748 norepinephrine Drugs 0.000 description 2
- SFLSHLFXELFNJZ-UHFFFAOYSA-N norepinephrine Natural products NCC(O)C1=CC=C(O)C(O)=C1 SFLSHLFXELFNJZ-UHFFFAOYSA-N 0.000 description 2
- 235000020030 perry Nutrition 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 238000002953 preparative HPLC Methods 0.000 description 2
- 210000002307 prostate Anatomy 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- BOLDJAUMGUJJKM-LSDHHAIUSA-N renifolin D Natural products CC(=C)[C@@H]1Cc2c(O)c(O)ccc2[C@H]1CC(=O)c3ccc(O)cc3O BOLDJAUMGUJJKM-LSDHHAIUSA-N 0.000 description 2
- 230000000241 respiratory effect Effects 0.000 description 2
- 102220067365 rs143592561 Human genes 0.000 description 2
- 238000012216 screening Methods 0.000 description 2
- QZAYGJVTTNCVMB-UHFFFAOYSA-N serotonin Chemical compound C1=C(O)C=C2C(CCN)=CNC2=C1 QZAYGJVTTNCVMB-UHFFFAOYSA-N 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 230000003595 spectral effect Effects 0.000 description 2
- 230000001954 sterilising effect Effects 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 125000003831 tetrazolyl group Chemical group 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 210000002700 urine Anatomy 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- MOMFXATYAINJML-UHFFFAOYSA-N 2-Acetylthiazole Chemical group CC(=O)C1=NC=CS1 MOMFXATYAINJML-UHFFFAOYSA-N 0.000 description 1
- 229940124596 AChE inhibitor Drugs 0.000 description 1
- 206010000087 Abdominal pain upper Diseases 0.000 description 1
- 240000006452 Abelmoschus crinitus Species 0.000 description 1
- 208000000187 Abnormal Reflex Diseases 0.000 description 1
- 241000931526 Acer campestre Species 0.000 description 1
- 241000276920 Acer chienii Species 0.000 description 1
- 241000426233 Acer davidii Species 0.000 description 1
- 241000276927 Acer laxiflorum Species 0.000 description 1
- 241000426239 Acer mandshuricum Species 0.000 description 1
- 241000931515 Acer palmatum Species 0.000 description 1
- 241000219240 Acer pictum subsp. mono Species 0.000 description 1
- 241001121888 Acer sempervirens Species 0.000 description 1
- 241001123290 Acer sinense Species 0.000 description 1
- 241000355242 Acer wilsonii Species 0.000 description 1
- 241001629553 Alectryon Species 0.000 description 1
- 240000005890 Allophylus cobbe Species 0.000 description 1
- 241000730364 Allophylus hirsutus Species 0.000 description 1
- 241000254796 Allophylus longipes Species 0.000 description 1
- 208000024827 Alzheimer disease Diseases 0.000 description 1
- 241000519337 Amesiodendron chinense Species 0.000 description 1
- 206010002383 Angina Pectoris Diseases 0.000 description 1
- 241001043701 Aporrhiza Species 0.000 description 1
- 241000519311 Arfeuillea Species 0.000 description 1
- 206010003210 Arteriosclerosis Diseases 0.000 description 1
- 241001119621 Arytera Species 0.000 description 1
- 241001122819 Arytera littoralis Species 0.000 description 1
- 241001119605 Atalaya Species 0.000 description 1
- 241000519307 Athyana Species 0.000 description 1
- 206010003805 Autism Diseases 0.000 description 1
- 208000020706 Autistic disease Diseases 0.000 description 1
- 241000519309 Averrhoidium Species 0.000 description 1
- 241001326502 Barringtonia Species 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- 241001421500 Blighia Species 0.000 description 1
- 244000212312 Blighia sapida Species 0.000 description 1
- 235000011349 Blighia sapida Nutrition 0.000 description 1
- ODYWCXGTKXWFFT-GLUWXPQDSA-N C/C=C(/C)C(=O)O[C@H]1C(C)O[C@@H](O[C@H]2[C@H](O)[C@@]3(COC(C)=O)C(CC2(C)C)C2=CCC4[C@@]5(C)CC[C@H](O[C@@H]6OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]7O[C@@H](CO)C(O)[C@@H]7O)C6O[C@@H]6OC(CO)[C@H](O)[C@@H](O)C6O)C(C)(C)C5CC[C@@]4(C)[C@]2(C)C[C@H]3O)C(O)[C@H]1OC(=O)/C(C)=C\C Chemical compound C/C=C(/C)C(=O)O[C@H]1C(C)O[C@@H](O[C@H]2[C@H](O)[C@@]3(COC(C)=O)C(CC2(C)C)C2=CCC4[C@@]5(C)CC[C@H](O[C@@H]6OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]7O[C@@H](CO)C(O)[C@@H]7O)C6O[C@@H]6OC(CO)[C@H](O)[C@@H](O)C6O)C(C)(C)C5CC[C@@]4(C)[C@]2(C)C[C@H]3O)C(O)[C@H]1OC(=O)/C(C)=C\C ODYWCXGTKXWFFT-GLUWXPQDSA-N 0.000 description 1
- DISYXZAOMTUARP-GLUWXPQDSA-N C/C=C(/C)C(=O)O[C@H]1C(C)O[C@@H](O[C@H]2[C@H](OC(C)=O)[C@@]3(CO)C(CC2(C)C)C2=CCC4[C@@]5(C)CC[C@H](O[C@@H]6OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]7O[C@@H](CO)C(O)[C@@H]7O)C6O[C@@H]6OC(CO)[C@H](O)[C@@H](O)C6O)C(C)(C)C5CC[C@@]4(C)[C@]2(C)C[C@H]3O)C(O)[C@H]1OC(=O)/C(C)=C\C Chemical compound C/C=C(/C)C(=O)O[C@H]1C(C)O[C@@H](O[C@H]2[C@H](OC(C)=O)[C@@]3(CO)C(CC2(C)C)C2=CCC4[C@@]5(C)CC[C@H](O[C@@H]6OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]7O[C@@H](CO)C(O)[C@@H]7O)C6O[C@@H]6OC(CO)[C@H](O)[C@@H](O)C6O)C(C)(C)C5CC[C@@]4(C)[C@]2(C)C[C@H]3O)C(O)[C@H]1OC(=O)/C(C)=C\C DISYXZAOMTUARP-GLUWXPQDSA-N 0.000 description 1
- GFNXJYIZHWPOMA-BETXXYJASA-N C/C=C(/C)C(=O)O[C@H]1[C@H](OC(=O)/C(C)=C\C)[C@@]2(CO)C(CC1(C)C)C1=CCC3[C@@]4(C)CC[C@H](O[C@@H]5OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]6O[C@@H](CO)C(O)[C@@H]6O)C5O[C@@H]5OC(CO)[C@@H](O)[C@@H](O)C5O)[C@](C)(CO)C4CC[C@@]3(C)[C@]1(C)C[C@H]2O Chemical compound C/C=C(/C)C(=O)O[C@H]1[C@H](OC(=O)/C(C)=C\C)[C@@]2(CO)C(CC1(C)C)C1=CCC3[C@@]4(C)CC[C@H](O[C@@H]5OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]6O[C@@H](CO)C(O)[C@@H]6O)C5O[C@@H]5OC(CO)[C@@H](O)[C@@H](O)C5O)[C@](C)(CO)C4CC[C@@]3(C)[C@]1(C)C[C@H]2O GFNXJYIZHWPOMA-BETXXYJASA-N 0.000 description 1
- QQECVWUIFIPTKP-KUNHZHQASA-N C/C=C(/C)C(=O)O[C@H]1[C@H](OC(=O)/C(C)=C\C)[C@@]2(CO)C(CC1(C)C)C1=CCC3[C@@]4(C)CC[C@H](O[C@@H]5OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]6O[C@@H](CO)C(O)[C@@H]6O)C5O[C@@H]5OC(CO)[C@@H](O)[C@@H](O)C5O)[C@](C)(CO)C4CC[C@@]3(C)[C@]1(C)[C@@H](O)[C@H]2O Chemical compound C/C=C(/C)C(=O)O[C@H]1[C@H](OC(=O)/C(C)=C\C)[C@@]2(CO)C(CC1(C)C)C1=CCC3[C@@]4(C)CC[C@H](O[C@@H]5OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]6O[C@@H](CO)C(O)[C@@H]6O)C5O[C@@H]5OC(CO)[C@@H](O)[C@@H](O)C5O)[C@](C)(CO)C4CC[C@@]3(C)[C@]1(C)[C@@H](O)[C@H]2O QQECVWUIFIPTKP-KUNHZHQASA-N 0.000 description 1
- MBJDXKFGFVRQIV-PHFNXENJSA-N C/C=C(/C)C(=O)O[C@H]1[C@H](OC(=O)/C(C)=C\C)[C@@]2(CO)C(CC1(C)C)C1=CCC3[C@@]4(C)CC[C@H](O[C@@H]5OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]6O[C@@H](CO)C(O)[C@@H]6O)C5O[C@@H]5OC(CO)[C@H](O)[C@@H](O)C5O)C(C)(C)C4CC[C@@]3(C)[C@]1(C)C[C@H]2O Chemical compound C/C=C(/C)C(=O)O[C@H]1[C@H](OC(=O)/C(C)=C\C)[C@@]2(CO)C(CC1(C)C)C1=CCC3[C@@]4(C)CC[C@H](O[C@@H]5OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]6O[C@@H](CO)C(O)[C@@H]6O)C5O[C@@H]5OC(CO)[C@H](O)[C@@H](O)C5O)C(C)(C)C4CC[C@@]3(C)[C@]1(C)C[C@H]2O MBJDXKFGFVRQIV-PHFNXENJSA-N 0.000 description 1
- IKGKAFZGBWHFKI-CFNUSAFTSA-N C/C=C(/C)C(=O)O[C@H]1[C@H](OC(=O)/C(C)=C\C)[C@@]2(CO)C(CC1(C)C)C1=CCC3[C@@]4(C)CC[C@H](O[C@@H]5OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]6O[C@@H](CO)C(O)[C@@H]6O)C5O[C@@H]5OC(CO)[C@H](O)[C@@H](O)C5O)C(C)(C)C4CC[C@@]3(C)[C@]1(C)[C@@H](O)[C@H]2O Chemical compound C/C=C(/C)C(=O)O[C@H]1[C@H](OC(=O)/C(C)=C\C)[C@@]2(CO)C(CC1(C)C)C1=CCC3[C@@]4(C)CC[C@H](O[C@@H]5OC(C(=O)O)[C@@H](O)[C@@H](O[C@@H]6O[C@@H](CO)C(O)[C@@H]6O)C5O[C@@H]5OC(CO)[C@H](O)[C@@H](O)C5O)C(C)(C)C4CC[C@@]3(C)[C@]1(C)[C@@H](O)[C@H]2O IKGKAFZGBWHFKI-CFNUSAFTSA-N 0.000 description 1
- AUUKLXSBJQHPTJ-HOWNBQRTSA-N C/C=C(/C)C(=O)O[C@H]1[C@H](OC(C)=O)C(C)(C)CC2C3=CCC4[C@@]5(C)CC[C@H](O[C@@H]6OC(C(=O)OCCCC)[C@@H](O)[C@@H](O[C@@H]7O[C@@H](CO)C(O)[C@@H]7O)C6O[C@@H]6OC(CO)[C@H](O)[C@@H](O)C6O)C(C)(C)C5CC[C@@]4(C)[C@]3(C)C[C@@H](O)[C@]21CO Chemical compound C/C=C(/C)C(=O)O[C@H]1[C@H](OC(C)=O)C(C)(C)CC2C3=CCC4[C@@]5(C)CC[C@H](O[C@@H]6OC(C(=O)OCCCC)[C@@H](O)[C@@H](O[C@@H]7O[C@@H](CO)C(O)[C@@H]7O)C6O[C@@H]6OC(CO)[C@H](O)[C@@H](O)C6O)C(C)(C)C5CC[C@@]4(C)[C@]3(C)C[C@@H](O)[C@]21CO AUUKLXSBJQHPTJ-HOWNBQRTSA-N 0.000 description 1
- SLXYBYCLCBXBFK-OLZOCXBDSA-N CC(C)([C@H]([C@@H]1O)O)Oc2c1cc(C=CC(O1)=O)c1c2 Chemical compound CC(C)([C@H]([C@@H]1O)O)Oc2c1cc(C=CC(O1)=O)c1c2 SLXYBYCLCBXBFK-OLZOCXBDSA-N 0.000 description 1
- DISYXZAOMTUARP-ZUXKCASVSA-N CC([C@@H](C(C1O)OC(/C(/C)=C\C)=O)OC(/C(/C)=C\C)=O)O[C@H]1O[C@@H]([C@@H]([C@@]12CO)OC(C)=O)C(C)(C)CC1C1=CCC([C@@](C)(CC3)C(CC4)C(C)(C)[C@H]3O[C@@H](C([C@H]([C@@H]3O)O[C@@H](C5O)O[C@@H](CO)C5O)O[C@@H](C(C5O)O)OC(CO)[C@@H]5O)OC3C(O)=O)[C@]4(C)[C@]1(C)C[C@H]2O Chemical compound CC([C@@H](C(C1O)OC(/C(/C)=C\C)=O)OC(/C(/C)=C\C)=O)O[C@H]1O[C@@H]([C@@H]([C@@]12CO)OC(C)=O)C(C)(C)CC1C1=CCC([C@@](C)(CC3)C(CC4)C(C)(C)[C@H]3O[C@@H](C([C@H]([C@@H]3O)O[C@@H](C5O)O[C@@H](CO)C5O)O[C@@H](C(C5O)O)OC(CO)[C@@H]5O)OC3C(O)=O)[C@]4(C)[C@]1(C)C[C@H]2O DISYXZAOMTUARP-ZUXKCASVSA-N 0.000 description 1
- SWECPSYAVWWHLX-UHFFFAOYSA-N CC1C2=C(C=CC3=C2OC(=O)C=C3)OC(C)(C)C1C Chemical compound CC1C2=C(C=CC3=C2OC(=O)C=C3)OC(C)(C)C1C SWECPSYAVWWHLX-UHFFFAOYSA-N 0.000 description 1
- PDCUGWRDEZWWTQ-PSLIRLAXSA-N C[C@@H]1C2=C(C=CC3=C2OC(=O)C=C3)O[C@@H]1C(C)(C)C Chemical compound C[C@@H]1C2=C(C=CC3=C2OC(=O)C=C3)O[C@@H]1C(C)(C)C PDCUGWRDEZWWTQ-PSLIRLAXSA-N 0.000 description 1
- BSCNZVHLNUUZCZ-LMUFZUPESA-N C[C@@H]1CCCC[C@H]1C.C[C@@H]1CCOC[C@@H]1C Chemical compound C[C@@H]1CCCC[C@H]1C.C[C@@H]1CCOC[C@@H]1C BSCNZVHLNUUZCZ-LMUFZUPESA-N 0.000 description 1
- 241000117439 Camellia cordifolia Species 0.000 description 1
- 241001663872 Camellia irrawadiensis Species 0.000 description 1
- 241001343472 Camellia pitardii Species 0.000 description 1
- 241001343463 Camellia tsaii Species 0.000 description 1
- 241000948783 Camellia wardii Species 0.000 description 1
- 241001466828 Camellia yunnanensis Species 0.000 description 1
- 208000020446 Cardiac disease Diseases 0.000 description 1
- 241000871261 Cardiospermum Species 0.000 description 1
- 241000871264 Cardiospermum halicacabum Species 0.000 description 1
- 241001119612 Castanospora Species 0.000 description 1
- 241000261515 Chouxia Species 0.000 description 1
- 241001119596 Chytranthus Species 0.000 description 1
- 241000992930 Conchopetalum Species 0.000 description 1
- 206010010814 Contracted bladder Diseases 0.000 description 1
- OMFXVFTZEKFJBZ-UHFFFAOYSA-N Corticosterone Natural products O=C1CCC2(C)C3C(O)CC(C)(C(CC4)C(=O)CO)C4C3CCC2=C1 OMFXVFTZEKFJBZ-UHFFFAOYSA-N 0.000 description 1
- 241000519322 Cossinia Species 0.000 description 1
- 241000238424 Crustacea Species 0.000 description 1
- 241001119594 Cubilia Species 0.000 description 1
- 241000872245 Cupania Species 0.000 description 1
- 241000723216 Cupaniopsis Species 0.000 description 1
- 241000723214 Cupaniopsis anacardioides Species 0.000 description 1
- 241001119598 Deinbollia Species 0.000 description 1
- 241000316507 Delavaya <gastropod> Species 0.000 description 1
- 241000536357 Delavaya toxocarpa Species 0.000 description 1
- 206010012289 Dementia Diseases 0.000 description 1
- 241000557738 Diatenopteryx Species 0.000 description 1
- 241000519291 Dictyoneura Species 0.000 description 1
- 241000872249 Dilodendron Species 0.000 description 1
- 241001638433 Dimocarpus confinis Species 0.000 description 1
- 235000006448 Dimocarpus confinis Nutrition 0.000 description 1
- 241001471052 Dimocarpus fumatus Species 0.000 description 1
- 235000000525 Dimocarpus longan Nutrition 0.000 description 1
- 241001119602 Diploglottis Species 0.000 description 1
- 241000500419 Diplopeltis <nematode> Species 0.000 description 1
- 241000158424 Dipteronia Species 0.000 description 1
- 241000426258 Dipteronia dyeriana Species 0.000 description 1
- 241001629517 Distichostemon Species 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- 241000158776 Dodonaea Species 0.000 description 1
- 241000733970 Dodonaea microzyga Species 0.000 description 1
- 235000015741 Dodonaea viscosa Nutrition 0.000 description 1
- 241000519287 Doratoxylon Species 0.000 description 1
- 241001119578 Elattostachys Species 0.000 description 1
- 241000519298 Eriocoelum Species 0.000 description 1
- 241001119582 Erioglossum Species 0.000 description 1
- 244000090243 Erioglossum rubiginosum Species 0.000 description 1
- 241000519301 Erythrophysa Species 0.000 description 1
- 241000182892 Erythrophysa alata Species 0.000 description 1
- 241000519296 Euphorianthus Species 0.000 description 1
- 241001676752 Eurycorymbus cavaleriei Species 0.000 description 1
- 241001188925 Exothea Species 0.000 description 1
- 241001119580 Filicium Species 0.000 description 1
- 241001119586 Ganophyllum Species 0.000 description 1
- 241000519534 Glenniea Species 0.000 description 1
- 241001469661 Gloeocarpus Species 0.000 description 1
- 241000519517 Gongrodiscus Species 0.000 description 1
- 240000002045 Guettarda speciosa Species 0.000 description 1
- 235000001287 Guettarda speciosa Nutrition 0.000 description 1
- 241001119584 Guindilia Species 0.000 description 1
- 241000872256 Guioa Species 0.000 description 1
- 241001119566 Haplocoelum Species 0.000 description 1
- 241000221289 Harpullia alata Species 0.000 description 1
- 244000179720 Harpullia arborea Species 0.000 description 1
- 241000057561 Harpullia austrocaledonica Species 0.000 description 1
- 241001009303 Harpullia frutescens Species 0.000 description 1
- 241000733140 Harpullia hillii Species 0.000 description 1
- 241000182306 Harpullia longipetala Species 0.000 description 1
- 241000733141 Harpullia pendula Species 0.000 description 1
- 241001629509 Harpullia ramiflora Species 0.000 description 1
- 241001119563 Harpullia rhyticarpa Species 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- 241001119549 Hippobromus Species 0.000 description 1
- 241000643565 Hornea Species 0.000 description 1
- 241001483208 Houssayanthus Species 0.000 description 1
- 229940122393 Hyaluronidase inhibitor Drugs 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- 241000700189 Hystrix <Rodentia> Species 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 241001119543 Jagera Species 0.000 description 1
- 241001286532 Koelreuteria bipinnata var. integrifoliola Species 0.000 description 1
- 241001671617 Koelreuteria elegans Species 0.000 description 1
- WQZGKKKJIJFFOK-ZZWDRFIYSA-N L-glucose Chemical compound OC[C@@H]1OC(O)[C@@H](O)[C@H](O)[C@H]1O WQZGKKKJIJFFOK-ZZWDRFIYSA-N 0.000 description 1
- 241000519560 Laccodiscus Species 0.000 description 1
- 241001119548 Lecaniodiscus Species 0.000 description 1
- 241001119546 Lepiderema Species 0.000 description 1
- 241001119539 Lepidopetalum Species 0.000 description 1
- 241000519436 Llagunoa Species 0.000 description 1
- 241001483193 Lophostigma Species 0.000 description 1
- 241000519460 Loxodiscus Species 0.000 description 1
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 1
- 241000519468 Macphersonia Species 0.000 description 1
- 241000738807 Maesa japonica Species 0.000 description 1
- 241000475408 Maesa montana Species 0.000 description 1
- 241000633089 Maesa perlaria Species 0.000 description 1
- 241000146438 Maesa tenera Species 0.000 description 1
- 241000519451 Magonia Species 0.000 description 1
- 241000536276 Majidea Species 0.000 description 1
- 241001122817 Matayba Species 0.000 description 1
- 241000536309 Molinaea Species 0.000 description 1
- 244000111261 Mucuna pruriens Species 0.000 description 1
- 235000006161 Mucuna pruriens Nutrition 0.000 description 1
- 241000550096 Myrmecophila Species 0.000 description 1
- KFEUJDWYNGMDBV-LODBTCKLSA-N N-acetyllactosamine Chemical group O[C@@H]1[C@@H](NC(=O)C)[C@H](O)O[C@H](CO)[C@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 KFEUJDWYNGMDBV-LODBTCKLSA-N 0.000 description 1
- 108010057466 NF-kappa B Proteins 0.000 description 1
- 102000003945 NF-kappa B Human genes 0.000 description 1
- 241000536360 Neotina Species 0.000 description 1
- 241001620259 Nephelium chryseum Species 0.000 description 1
- 244000183331 Nephelium lappaceum Species 0.000 description 1
- 235000015738 Nephelium lappaceum Nutrition 0.000 description 1
- 235000015742 Nephelium litchi Nutrition 0.000 description 1
- 208000012902 Nervous system disease Diseases 0.000 description 1
- 206010029216 Nervousness Diseases 0.000 description 1
- 108090000189 Neuropeptides Proteins 0.000 description 1
- 206010030113 Oedema Diseases 0.000 description 1
- 241001119529 Pancovia Species 0.000 description 1
- 241001629489 Pappea Species 0.000 description 1
- 208000018737 Parkinson disease Diseases 0.000 description 1
- 241001119526 Paullinia Species 0.000 description 1
- 235000010242 Paullinia Nutrition 0.000 description 1
- 241000268741 Pax Species 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 241001315608 Pittosporum angustifolium Species 0.000 description 1
- 241001315609 Pittosporum crassifolium Species 0.000 description 1
- 241001671605 Pittosporum pentandrum Species 0.000 description 1
- 241000513347 Pittosporum tenuifolium Species 0.000 description 1
- 241001092086 Pittosporum undulatum Species 0.000 description 1
- 241001085641 Placodiscus <flatworm> Species 0.000 description 1
- 241000872196 Plagioscyphus Species 0.000 description 1
- 241000536454 Podonephelium Species 0.000 description 1
- 241001544342 Polyspora axillaris Species 0.000 description 1
- 241001629485 Pometia Species 0.000 description 1
- 235000017284 Pometia pinnata Nutrition 0.000 description 1
- 240000007653 Pometia tomentosa Species 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 241000536450 Pseudima Species 0.000 description 1
- 241000536469 Pseudopteris Species 0.000 description 1
- 241001190105 Radlkofera Species 0.000 description 1
- 208000003782 Raynaud disease Diseases 0.000 description 1
- 208000012322 Raynaud phenomenon Diseases 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- 241001119521 Rhysotoechia Species 0.000 description 1
- 241000612118 Samolus valerandi Species 0.000 description 1
- 241001093760 Sapindaceae Species 0.000 description 1
- 241000693813 Sapindus delavayi Species 0.000 description 1
- 241000580955 Sapindus mukorossi Species 0.000 description 1
- 241000862929 Sapindus saponaria var. drummondii Species 0.000 description 1
- 241001119523 Sarcopteryx Species 0.000 description 1
- 241001119509 Sarcotoechia Species 0.000 description 1
- 241000536464 Scyphonychium Species 0.000 description 1
- 241000557729 Serjania Species 0.000 description 1
- 241000937377 Sinoradlkofera minor Species 0.000 description 1
- 208000005439 Sleep paralysis Diseases 0.000 description 1
- 241001119493 Smelophyllum Species 0.000 description 1
- 241000536400 Storthocalyx Species 0.000 description 1
- 241001122831 Synima Species 0.000 description 1
- 241000557731 Talisia Species 0.000 description 1
- 241000102556 Thinouia Species 0.000 description 1
- 241000872207 Thouinia Species 0.000 description 1
- 241000911852 Thouinidium Species 0.000 description 1
- 241000536399 Tina Species 0.000 description 1
- 241000536416 Tinopsis Species 0.000 description 1
- 241001122834 Toechima Species 0.000 description 1
- 208000030886 Traumatic Brain injury Diseases 0.000 description 1
- 241001122828 Tristira <angiosperm> Species 0.000 description 1
- 241001122826 Tristiropsis Species 0.000 description 1
- 241000557680 Ungnadia Species 0.000 description 1
- 241000557684 Ungnadia speciosa Species 0.000 description 1
- 206010046543 Urinary incontinence Diseases 0.000 description 1
- 241000536430 Urvillea Species 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- 241000536421 Vouarana Species 0.000 description 1
- 241000937375 Zanha Species 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 230000036626 alertness Effects 0.000 description 1
- 125000002723 alicyclic group Chemical group 0.000 description 1
- 125000005090 alkenylcarbonyl group Chemical group 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 230000001088 anti-asthma Effects 0.000 description 1
- 230000003501 anti-edematous effect Effects 0.000 description 1
- 230000002322 anti-exudative effect Effects 0.000 description 1
- 230000002968 anti-fracture Effects 0.000 description 1
- 230000003602 anti-herpes Effects 0.000 description 1
- 230000001387 anti-histamine Effects 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 230000000767 anti-ulcer Effects 0.000 description 1
- 239000000924 antiasthmatic agent Substances 0.000 description 1
- 239000000739 antihistaminic agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 150000001491 aromatic compounds Chemical class 0.000 description 1
- 208000011775 arteriosclerosis disease Diseases 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 210000005068 bladder tissue Anatomy 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000003327 cancerostatic effect Effects 0.000 description 1
- 125000002837 carbocyclic group Chemical group 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 208000026106 cerebrovascular disease Diseases 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 208000029078 coronary artery disease Diseases 0.000 description 1
- OMFXVFTZEKFJBZ-HJTSIMOOSA-N corticosterone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@H](CC4)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 OMFXVFTZEKFJBZ-HJTSIMOOSA-N 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- WHHGLZMJPXIBIX-UHFFFAOYSA-N decabromodiphenyl ether Chemical compound BrC1=C(Br)C(Br)=C(Br)C(Br)=C1OC1=C(Br)C(Br)=C(Br)C(Br)=C1Br WHHGLZMJPXIBIX-UHFFFAOYSA-N 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 230000001882 diuretic effect Effects 0.000 description 1
- 208000002173 dizziness Diseases 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 230000000855 fungicidal effect Effects 0.000 description 1
- 239000000417 fungicide Substances 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 208000019622 heart disease Diseases 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000002390 heteroarenes Chemical class 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- 125000003104 hexanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 206010020745 hyperreflexia Diseases 0.000 description 1
- 230000035859 hyperreflexia Effects 0.000 description 1
- 201000001881 impotence Diseases 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 238000010829 isocratic elution Methods 0.000 description 1
- 125000000555 isopropenyl group Chemical group [H]\C([H])=C(\*)C([H])([H])[H] 0.000 description 1
- 208000017169 kidney disease Diseases 0.000 description 1
- 201000006370 kidney failure Diseases 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 201000005202 lung cancer Diseases 0.000 description 1
- 208000020816 lung neoplasm Diseases 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 230000001452 natriuretic effect Effects 0.000 description 1
- 230000017095 negative regulation of cell growth Effects 0.000 description 1
- 125000006574 non-aromatic ring group Chemical group 0.000 description 1
- 231100001160 nonlethal Toxicity 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 125000002958 pentadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000000575 pesticide Substances 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 230000001817 pituitary effect Effects 0.000 description 1
- 206010036596 premature ejaculation Diseases 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- OILHVNROWDFZDW-UHFFFAOYSA-N prosapogenin Natural products CC1(C)CCC2(C(O)CC3(C)C(=CCC4C5(C)CCC(OC6OC(CO)C(O)C(O)C6O)C(C)(C=O)C5CCC34C)C2C1)C(=O)O OILHVNROWDFZDW-UHFFFAOYSA-N 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 230000002040 relaxant effect Effects 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 125000006413 ring segment Chemical group 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 239000007790 solid phase Substances 0.000 description 1
- 238000010183 spectrum analysis Methods 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000035882 stress Effects 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 230000003096 thymolvtic effect Effects 0.000 description 1
- 230000017105 transposition Effects 0.000 description 1
- 230000000472 traumatic effect Effects 0.000 description 1
- 125000002889 tridecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002948 undecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000003774 valeryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000002666 vasoprotective effect Effects 0.000 description 1
- 230000001643 venotonic effect Effects 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- AFVLVVWMAFSXCK-UHFFFAOYSA-N α-cyano-4-hydroxycinnamic acid Chemical compound OC(=O)C(C#N)=CC1=CC=C(O)C=C1 AFVLVVWMAFSXCK-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7012—Compounds having a free or esterified carboxyl group attached, directly or through a carbon chain, to a carbon atom of the saccharide radical, e.g. glucuronic acid, neuraminic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/575—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids substituted in position 17 beta by a chain of three or more carbon atoms, e.g. cholane, cholestane, ergosterol, sitosterol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7028—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages
- A61K31/7034—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin
- A61K31/704—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin attached to a condensed carbocyclic ring system, e.g. sennosides, thiocolchicosides, escin, daunorubicin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/77—Sapindaceae (Soapberry family), e.g. lychee or soapberry
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/14—Vasoprotectives; Antihaemorrhoidals; Drugs for varicose therapy; Capillary stabilisers
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H15/00—Compounds containing hydrocarbon or substituted hydrocarbon radicals directly attached to hetero atoms of saccharide radicals
- C07H15/20—Carbocyclic rings
- C07H15/24—Condensed ring systems having three or more rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H15/00—Compounds containing hydrocarbon or substituted hydrocarbon radicals directly attached to hetero atoms of saccharide radicals
- C07H15/20—Carbocyclic rings
- C07H15/24—Condensed ring systems having three or more rings
- C07H15/256—Polyterpene radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J9/00—Normal steroids containing carbon, hydrogen, halogen or oxygen substituted in position 17 beta by a chain of more than two carbon atoms, e.g. cholane, cholestane, coprostane
-
- C07C2103/52—
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/52—Ortho- or ortho- and peri-condensed systems containing five condensed rings
Definitions
- This invention relates to saponins, and compounds with angeloyl groups isolated from plants, their uses and functions, and methods of their preparations.
- This invention provides a compound comprising a triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin, comprising two angeloyl groups, or at least two side groups selected from the group consisting of: angeloyl groups, tigloyl groups and senecioyl groups, wherein the side groups are attached to carbon 21, 22 or/and 28 of triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin backbone.
- This invention provides a composition for inhibiting tumor cell growth, comprising an appropriate amount of a triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin, wherein the triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin comprises two angeloyl groups or any two side groups selected from the group consisting of: angeloyl groups, tigloyl groups and senecioyl groups, wherein the side groups are attached to carbon 21 and 22 of triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin backbone. In an embodiment, the side groups are attached to carbon 21, 22 or/and 28 of triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin backbone.
- This invention provides a composition for inhibiting tumor cell growth, comprising an appropriate amount of a triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin, wherein the triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin comprises two angeloyl groups or any two side groups selected from the group consisting of: angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl, wherein the side groups are attached to carbon 21 and 22 of triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin backbone.
- the side groups are attached to carbon 21, 22 or/and 28 of triterpenoidal saponin, triterpenoid, triter
- the invention provides the methods and uses of triterpenoidal saponins purified and isolated from plants.
- compositions comprising the triterpenoidal saponins or their derivatives for inhibition of tumor growth.
- the compounds comprise angeloyl group(s) or tigloyl group(s) or senecioyl group(s) or combinations thereof which are attached to carbon 21, 22 or/and 28 of their sapongenines.
- the compounds may comprise any two angeloyl groups or tigeloyl groups or senecioyl groups or combinations thereof attached to a sugar moiety which bonds to carbon 21 or 22 of their sapongenines.
- the side groups are attached to carbon 21, 22 or/and 28 of triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin backbone.
- compositions comprising the triterpenoidal saponins or their derivatives for inhibition of tumor growth.
- the compounds comprise angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or combinations thereof which are attached to carbon 21, 22 or/and 28 of their sapongenines.
- the compounds may comprise any two angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or combinations thereof attached to a sugar moiety which bonds to carbon 21 or 22 of their sapongenines.
- the side groups are attached to carbon 21, 22 or/and 28 of triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin backbone.
- the saponin comprises a sugar moiety, wherein the sugar moiety comprises at least one sugar, or D-glucose, or D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid or D-galacturonic acid, or their derivative thereof, or the combination thereof.
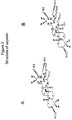
- FIG. 1 shows structure of saponins.
- FIG. 2 shows structure of saponins.
- FIG. 3 shows structure of saponins
- Positions 23-27, 29-30 are attached with CH3 or CH2OH or COOH or acetyl group
- FIG. 4 shows structure of saponins
- FIG. 5 shows a structure of saponins
- positions 23-27, 29-30 are attached with CH3 or CH2OH or COOH or acetyl group
- FIG. 6 shows a structure of saponins
- positions 23-27, 28-30 are attached with CH3 or CH2OH or COOH or acetyl group
- FIG. 7 A shows a structure of saponins
- FIG. 7 B, C, D shows a structure of saponins
- Position 23-27 and 28-30 are attached with CH3 or CH2OH or COOH or CHO
- FIG. 8 shows a structure of saponins:
- FIG. 9 A shows a structure of saponins:
- FIG. 9 B shows a structure of saponins:
- FIG. 10 shows a structure of saponins:
- FIG. 11 A shows a structure of saponins:
- FIG. 11 B shows a structure of saponins:
- FIG. 12 A shows a structure of saponins:
- FIG. 12 B shows a structure of saponins:
- FIG. 13 shows a structure of saponins:
- FIG. 14 shows a structure of saponins:
- FIG. 15 shows a structure of saponins
- FIG. 16 A, 16 B shows a structure of saponins
- FIG. 17 shows a structure of saponins
- FIG. 18 shows a structure of saponins
- FIG. 19 shows a structure of saponins
- FIG. 20 shows a structure of saponins
- FIG. 21 shows a structure of saponins
- Position 23-27, 29, 30 are attached a CH3 or CH2OH or COOH or acetyl group
- Positions 28 CH3 or CH2OH or COOH or acetyl group or angeloyl or tigloyl or senecioly or sugar chains
- FIG. 22 shows a structure of saponins
- S1 sucgar moiety one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid or/and their derivatives.
- Positions 23-27, 29, 30 are attached a CH3 or CH2OH or COOH or acetyl group.
- Position 28 CH3 or CH2OH or COOH or acetyl group or angeloyl or tigloyl or senecioly or sugar chains
- FIG. 23 shows a structure of saponins
- Positions 23-27, 29, 30 are attached a CH3 or CH2OH or COOH or acetyl group
- Position 28 CH3 or CH2OH or COOH or acetyl group or angeloyl or tigloyl or senecioly or sugar chains
- FIG. 24 shows a structure of saponins
- Positions 23-27, 29, 30 are attached a CH3 or CH2OH or COOH or acetyl group
- Position 28 CH3 or CH2OH or COOH or acetyl group or angeloyl or tigloyl or senecioly or sugar chains or sugar chain with angeloyl group
- FIG. 25 shows a structure of saponins
- Positions 23-27, 29, 30 are attached a CH3 or CH2OH or COOH
- Position 28 CH3 or CH2OH or COOH or acetyl group or angeloyl or tigloyl or senecioly or sugar chains or sugar chain with angeloyl group.
- FIG. 26 shows a structure of compounds with angeloyl groups
- FIG. 27 shows a structure of saponins
- FIGS. 28 A and B shows the comparison of potency of Compound Y (saponin with 2 angeloyl groups) and compound X (saponin with 1 angeloyl) in ovarian cancer cells.
- the IC50 for Compound Y in ovary cells is about 1.5 ug/ml while the IC50 for compound X is 30 ug/ml.
- FIG. 28 C shows the inhibition of the purified Compound Y on the growth of skin cancer cell.
- the IC50 is 0.23 ug/ml.
- FIG. 29 shows the inhibition of the purified Compound Y1 and Compound Y2 on the growth of ovarian cancer cells.
- FIG. 30 shows the anticancer activity of Y, Y8, Y9 and Y10 on ovarian cancer cells as determined by MTT assay.
- FIG. 31 shows a compound of the invention without angeloyl groups.
- (B) shows a compound of the invention without sugar moiety.
- FIG. 32 shows a comparison of MTT and Haemolytic activities of saponin compound and Compound Ys of the invention.
- (A) and (B) shows hemolytic activities.
- (C) and (D) show MTT activities.
- FIG. 33 shows the saponin compounds Y, X ACH—Y, AKOH—Y of the invention. These compounds are purified and their structures were verified by NMR and MS. These compounds are then used for cell growth inhibition studies (MTT).
- Removal of sugars from Y (ACH—Y) but retaining the diangeloyl group retains 40% of activity (IC50 9.5 ug/ml).
- AKOH—Y removal of both angeloyl groups from C21 and C22 of Y completely abolishes its activity (no activity even at 120 ug/ml).
- Results indicate that diangeloyl groups in compound Ys are important for anti-tumor activity.
- This invention describes the results of a program of screening the bioactive compounds from natural plants. Most of the plants are in Sapindaceae family, which has 1400-2000 species with 140-150 genera.
- the program of screening for bioactive compounds is based on our purification methods and biological assays including the MTT assay.
- the invention provides methods and uses of saponins including triterpenoidal saponins purified or isolated from plants in the following genus:
- Saponins including triterpenoidal saponins may also be purified or isolated from the following species of plants:
- Acer campestre L. Acer chienii Hu et Cheng, Acer chingii Hu, Acer davidii Franch, Acer laxiflorum Pax, Acer mandshuricum Maxim., Acer mono Maxim., Acer orientale L., Acer palmatum Thunb., Acer sinense Pax, Acer wilsonii Redhd., Acer yui Fang, Aesculus arguta, Aesculus assamica Griff., Aesculus californica (Spach) Nutt., Aesculus chinensis Bunge, Aesculus chinensis var.
- Harpullia alata F. Mueller Harpullia arborea (Blanco) Rdlk., Harpullia austro - calcdonica Baillon, Harpullia camptoneura Radlk., Harpullia cauliflora K. Schum. & Lauterb., Harpullia crustacea Radlk., Harpullia cupanoides Roxb., Harpullia frutescens F. M. Bailey, Harpullia giganteacapsula M. Vente, Harpullia hillii F.
- This invention provides a compound comprising the structures recited in FIGS. 1 to 27 .
- This invention provides a compound comprising a triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin, comprising two angeloyl group or at least two side groups selected from the group consisting of: angeloyl groups, tigloyl groups and senecioyl groups, wherein the side groups are attached to carbon 21 and 22 of triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin backbone.
- the saponin comprising a sugar moiety
- the sugar moiety comprises at least one sugar, or D-glucose, or D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid or D-galacturonic acid, or their derivative thereof, or the combination, thereof.
- a sugar moiety is a segment of molecule comprising one or more sugar group.
- compositions comprising the structures recited in FIGS. 1 to 27 , or a compound comprising a triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin, comprising at least two side groups selected from the group consisting of: angeloyl groups, tigloyl groups and senecioyl groups, wherein the side groups are attached to carbon 21, 22 or 28 of triterpenoidal saponin, triterpenoid, triterpenoidal compound or sapongenin backbone.
- compositions are obtainable from the above-identified plants.
- composition comprising the structures recited in FIGS. 1 to 27 , or a compound comprising two angeloyl groups or at least two side groups selected from the group consisting of: angeloyl groups, tigloyl groups and senecioyl groups,
- This invention provides uses of the saponins isolated from the roots, kernel, leave, bark, stem, husk, seed, seed shell or fruit of the above plants, and methods of their preparations.
- This invention provides a method of preparing the saponins, comprising the steps of:
- HTB-9 (bladder), HeLa-S3 (cervix), DU145 (prostate), H460 (lung), MCF-7 (breast), K562 (leukocytes), HCT116 (colon), HepG2 (liver), U2OS (bone), T98G (brain), SK-MEL-5 (Skin) and OVCAR-3 (ovary).
- Cells were grown in culture medium (HeLa-S3, DU145, MCF-7, Hep-G2 and T98G in MEN (Earle's salts); HTB-9, H460, K562, OVCAR-3 in RPMI-1640; HCT-116, U2OS in McCoy-5A) supplemented with 10% fetal calf serum, glutamine and antibiotics in a 5% CO 2 humidified incubator at 37° C.
- culture medium HeLa-S3, DU145, MCF-7, Hep-G2 and T98G in MEN (Earle's salts); HTB-9, H460, K562, OVCAR-3 in RPMI-1640; HCT-116, U2OS in McCoy-5A
- MTT Assay The procedure for MTT assay followed the method described in (Carmichael et al., 1987) with modifications.
- Cells were seeded into a 96-wells plate at concentrations of 10,000/well (HTB-9, HeLa, H460, HCT116, T98G, OVCAR-3), 15,000/well (DU145, MCF-7, HepG2, U2OS), or 40,000/well (K562), for 24 hours before drug-treatment. Cells were then exposed to drugs for 48 hours (72 hours for HepG2, U2OS, and 96 hours for MCF-7). After the drug-treatment, MTT (0.5 mg/ml) was added to cultures for an hour.
- % G ( TD ⁇ T 0/ TC ⁇ T 0) ⁇ 100 (1), where TC or TD represent O.D. readings of control or drug-treated cells.
- LC cytotoxicity
- This invention provides a composition effective in reducing or inhibiting cancer growth.
- the cancer includes but is not limited to bladder cancer, bone cancer and ovary cancer.
- This invention provides a composition comprising triterpenoidal saponins or their derivatives for inhibiting tumor growth.
- This invention provides a compound selected from a compound of formula (1):
- R1 and R2 represent angeloyl group
- R3 represents H or OH
- R4 represent CH2OR6
- R6 is H
- R5 represents at least one sugar moiety or its derivatives.
- R1 and R2 represent angeloyl group; R3 represents H or OH; R4 represents COOR6 wherein R6 is H.
- R1 represents H;
- R2 represents angeloyl group;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6; wherein R6 is an angeloyl group.
- R4 represents CH2OR6 or COOR6; at least two of R1, R2, and R6 comprise an angeloyl group or acid having five carbons; R3 represents H or OH; and wherein R6 is angeloyl group, H, acetyl group, tigloyl group, senecioly group, or an acid having two to five carbons.
- At least one angeloyl of R1 or R2 is replaced by acetyl group, tigloyl group, senecioly group, or an acid having two to five carbons;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6; and wherein R6 is angeloyl group.
- the R4 represents CH2OR6 or COOR6; and wherein R6 is H or acetyl.
- At least one of R1, R2, and R4 comprises a sugar moiety or is a compound comprises sugar moiety, wherein the sugar moiety comprises at least two angeloyl groups, acetyl group, tigloyl group, senecioly group, or an acid having two to five carbons or combination thereof.
- positions 23, 24, 25, 26, 29, 30 of the compound independently comprise CH3, CH2OH, CHO, COOH, alkyls group, acetyl group or derivative thereof.
- R5 represents sugar moiety comprising glucose, galactose or/and arabinose.
- R5 represents sugar moiety, wherein the sugar moiety comprises two sugars selected from D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, and derivative thereof, and the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises three sugars selected from D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, and derivative thereof, and the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises three sugars selected from D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, and derivative thereof, and the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises at least one sugar, or D-glucose, D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid or D-galacturonic acid, or derivative thereof, or the combination thereof.
- R5 represents a compound capable of performing the function of the sugar moiety.
- the R5 represents H.
- R4 represents H or OH or CH3.
- R1 or/and R2 is a functional group capable of performing the function of the angeloyl.
- R5 represents a compound capable of performing the function of the sugar moiety.
- a sugar moiety is a segment of molecule comprising one or more sugar group. Substitution, deletion and/or addition of any group in the above-described compounds will be apparent to one of ordinary skill in the art based on the teaching of this application. In a further embodiment, the substitution, deletion and/or addition of the group(s) in the compound of the invention does not substantially affect the biological function of the compound. In a further embodiment, the angeloyl groups are in a trans-position on a structure.
- This invention provides a compound selected from a compound of formula (2):
- R1 represents angeloyl group
- R2 represents angeloyl group
- R3 represents OH or H
- Positions 23, 24, 25, 26, 27, 29, of the compound independently comprise CH3, or CH2OH, or CHO, or COOH, alkyls group, or acetyl group, or derivative
- R6 represents Ac or H
- R5 represents sugar moiety, wherein the sugar moiety comprises at least one sugar, or D-glucose, or D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid, or D-galacturonic acid, or their derivative thereof, or the combination thereof.
- R5 represents a compound capable of performing the function of the sugar moiety.
- the sugar moiety comprises L-arabinose, D-glucose and/or D-galactose, or combinations thereof.
- any two of R1, R2 or R6 are angeloyl groups, or any one of R1, R2 or R6 is attached to a sugar moiety in which two angeloyl groups are attached to adjacent carbons of the monosaccharides.
- R1, R2, and R6 comprises angeloyl group, acetyl group, tigloyl group, senecioly group, or an acid with two to five carbons or combination thereof.
- At least one of R1, R2 or R6 is attached a sugar moiety, wherein sugar moiety comprises two angeloyl group, acetyl group, tigloyl group, senecioly group, acid having two to five carbons, or combinations thereof.
- This invention provides a compound selected from a compound of formula (3):
- R1 represents angeloyl group
- R2 represents angeloyl group
- R3 represents OH or H
- R4 represents CH3 or CH2OH or alkyls group or their derivatives
- R6 represents Ac or H
- R5 represents sugar moiety, wherein the sugar moiety comprises at least one sugar, or D-glucose, or D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid, or D-galacturonic acid, or derivative thereof, or the combination thereof.
- R5 represents a compound capable of performing the function of sugar moiety.
- the sugar moiety comprises L-arabinose, or D-glucose, or D-galactose, or combinations thereof.
- at least one of R1, R2 or R6 is attached a sugar moiety or rhamnose, wherein sugar moiety or rhamnose comprises two angeloyl group, acetyl group, tigloyl group, senecioly group, acid having two to five carbons, or combinations thereof.
- This invention provides a compound selected from a compound of formula (4):
- R1 and R2 represent angeloyl group
- R3 represents H or OH
- R4 represent CH2OR6
- R6 is H or acetyl
- R5 represents sugar moiety or D-glucose
- R7 represents a sugar moiety or L-arabinose
- R8 represents sugar moiety or D-galactose
- R9 represent COOH or CH2OH.
- R4 represents COOR6, wherein the R6 is H or acetyl.
- the R5, R7 or/and R8 are H or sugar moiety, wherein the sugar moiety comprises at least one sugar, or glucose, or galactose, or rhamnose, or arabinose, or xylose, or alduronic acid, or glucuronic acid or galacturonic acid, or derivative thereof.
- at least 2 of R1, R2 and R6 are angeloyl group;
- R4 represent CH2OR6 or COOR6, wherein R6, R1 and R2 are angeloyl group, acetyl group, tigloyl group, senecioly group, an acid having two to five carbons or H.
- R1, R2 and R6 are angeloyl group, acetyl group, tigloyl group, senecioly group, or an acid having two to five carbons;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6, where R6 is H or angeloyl group, acetyl group, tigloyl group, senecioly group.
- R1 and R2 represent angeloyl group;
- R3 represents H or OH;
- R4 represents COOR6 wherein R6 is H;
- R1 represents H;
- R2 represents angeloyl group;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6; wherein R6 is an angeloyl group.
- At least two of R1, R2, and R6 comprise an angeloyl group;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6; and wherein R6 is H, angeloyl group, acetyl group, tigloyl group, senecioly group, or an acid having two to five carbons.
- At least one angeloyl of R1 or R2 is replaced by acetyl group, tigloyl group, senecioly group, or an acid having two to five carbons;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6; and wherein R6 is angeloyl group.
- At least one of R1, R2, and R6 is a sugar moiety or rhamnose comprising at least two angeloyl groups, acetyl group, tigloyl group, senecioly group, or an acid having two to five carbons or combination thereof.
- positions 23, 24, 25, 26, 29, 30 of the compound independently comprise CH3, CH2OH, CHO, COOH, alkyls group, acetyl group or derivative thereof.
- R4 represents H or OH or CH3.
- a sugar moiety is a segment of molecule comprising one or more sugar group.
- This invention provides a compound selected from a compound of formula (5):
- R1 and R2 represent angeloyl group
- R3 represents H or OH
- R6 represent H or acetyl
- R9 represents COOH or CH2OH
- R10 represent CH3 or CH2OH or COOH
- R5, R7 and R8 are H or/and sugar moiety, wherein the sugar moiety comprises at least one sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, or D-galacturonic acid, or derivative thereof.
- At least one of R1, R2, and R6 is a sugar moiety or compound comprising at least two angeloyl groups, acetyl group, tigloyl group, senecioly group, or an acid having two to five carbons or combination thereof. In another embodiment, at least two of R1, R2, and R6 comprise an angeloyl group
- This invention provides a compound selected from a compound of formula (6):
- R1 represents angeloyl group
- R2 represents angeloyl group
- R3 represents OH or H
- R4 represents CH3 or CH2OH
- R7 represents H
- R5 represents D-glucose, D-Galactose, L-arabinose or H
- R6 represents D-glucose, D-Galactose, L-arabinose or H.
- R5 or/and R6 are H or sugar moiety comprises at least one sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, or D-galacturonic acid, or derivative thereof.
- a sugar moiety is a segment of molecule comprising one or more sugar group. Substitution, deletion and/or addition of any group in the above-described compounds will be apparent to one of ordinary skill in the art based on the teaching of this application. In a further embodiment, the substitution, deletion and/or addition of the group(s) in the compound of the invention does not substantially affect the biological function of the compound.
- This invention provides a compound selected from a compound of formula (1A):
- R1 and R2 independently represent angeloyl group
- R3 represents H or OH
- R4 represent CH2OR6
- R6 is H
- R5 represents at least one sugar moiety or its derivatives.
- R1 and R2 independently represent angeloyl group; R3 represents H or OH; R4 represents COOR6 wherein R6 is H; R5 represents at least one sugar moiety or its derivatives.
- R1 represents H;
- R2 represents angeloyl group;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6; wherein R6 is an angeloyl group; and
- R5 represents at least one sugar moiety or its derivatives.
- R3 represents H or OH
- R4 represents CH2OR6 or COOR6
- R6 is angeloyl group, H, acetyl group, tigloyl group, senecioyl group, or an acid having two to five carbons
- at least two of R1, R2, and R6 comprise an angeloyl group or acid having five carbons
- R5 represents at least one sugar moiety or its derivatives.
- At least one angeloyl from R1 or R2 is replaced by acetyl group, tigloyl group, senecioyl group, or an acid having two to five carbons;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6 wherein R6 is angeloyl group;
- R5 represents at least one sugar moiety or its derivatives.
- R4 represents CH2OR6 or COOR6; at least one of R1, R2, and R6 is a sugar moiety comprising at least two angeloyl groups, acetyl group, tigloyl group, senecioyl group, or an acid having two to five carbons or combination thereof.
- position 24 of the compound comprises CH3 or CH2OH.
- positions 23, 24, 25, 26, 29, 30 of the compound independently comprise CH3 or CH2OH.
- positions 23, 24, 25, 26, 29, 30 of the compound independently comprise CH3, CH2OH, CHO, COOH, COOalkyl, COOaryl, COO-heterocyclic, COO-heteroaryl, CH 2 Oaryl, CH 2 O-heterocyclic, CH 2 O-heteroaryl, alkyls group, acetyl group or derivative thereof.
- R5 represents sugar moiety comprising glucose, galactose and arabinose.
- R5 represents sugar moiety, wherein the sugar moiety comprises at least one sugar, or D-glucose, D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid or D-galacturonic acid, or derivative thereof, or the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises two sugars comprising D-glucose, or D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid or D-galacturonic acid, or derivative thereof, or the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises at least three sugars selected from D-glucose, or D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid or D-galacturonic acid, or derivative thereof, or the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises at least one sugar, glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid or galacturonic acid, or derivative thereof, or the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises three sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid and galacturonic acid, and derivative thereof, and the combination thereof.
- sugar moiety comprises three sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid and galacturonic acid, and derivative thereof, and the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises three sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, and derivative thereof, and the combination thereof.
- R5 represents a compound capable of performing the function of the sugar moiety.
- the R5 represents H.
- R4 represents H or OH or CH3.
- R1 or/and R2 is a functional group capable of performing the function of the angeloyl.
- R5 represents a compound capable of performing the function of the sugar moiety.
- R1 and R2 are selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl.
- R1 and R2 comprise angeloyl, tigloyl, senecioyl, benzoyl or alkenoyl.
- R4 represents CH2OR6; at least two of R1, R2 and R6 are selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl.
- R4 represents CH2OR6; at least two of R1, R2 and R6 are comprise angeloyl, tigloyl, senecioyl, benzoyl or alkenoyl.
- R4 represents CH2OR6; at least two of R1, R2 and R6 are comprise angeloyl, benzoyl or alkenoyl.
- R1 and R2 are selected from H, angeloyl, acetyl, tigloyl, senecioyl, alkyl, acyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, heterocylic or heteroraryl;
- R4 represents CH2OR6 or COOR6; wherein R6 is selected from H, COCH3, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 or CH2COOR6; at least two of R1, R2 and R6 are selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- At least two of R1, R2 and R4 are comprising angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- At least two of R1, R2 and R4 comprise angeloyl, acetyl, tigloyl, senecioyl, benzoyl, alkenoyl, or derivative thereof.
- At least two of R1, R2 and R4 comprise angeloyl, tigloyl, senecioyl, benzoyl, alkenoyl, or derivative thereof.
- At least two of R1, R2 and R4 comprise a compound capable of performing the function of angeloyl.
- At least two of R1, R2 and R4 comprise a compound capable of performing the function of benzoyl.
- R4 represents CH2OR6, COOR6 or CH2COOR6; at least two of R1, R2 and R6 are comprise angeloyl, tigloyl, senecioyl, benzoyl, alkenoyl, benzoyl or derivative thereof.
- R4 represents CH2OR6, COOR6 or CH2COOR6;
- R1, R2 and/or R6 is/are sugar moiety, which comprise compounds selected from H, angeloyl, acetyl, tigloyl, senecioly, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 or CH2COOR6;
- R1, R2 and/or R6 is/are sugar moiety, which comprise at least 2 compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 or CH2COOR6;
- R1, R2 and/or R6 is/are sugar moiety, which comprise at least 2 compounds selected from angeloyl, tigloyl, senecioyl, benzoyl, alkenoyl or derivative thereof.
- R4 represents CH2OR6, COOR6 or CH2COOR6;
- R1, R2 and/or R6 is/are sugar moiety, which comprise at least 2 compounds selected from angeloyl, benzoyl, alkenoyl or derivative thereof.
- a compound selected from formula (1A) comprises at least 2 compounds selected from angeloyl, acetyl, tigloyl, senecioyl, or derivative thereof or a compound capable performing the function of angeloyl.
- a compound selected from formula (1A) comprise at least 2 compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- a compound selected from formula (1A) comprises a sugar moiety or a compound capable of performing function of sugar moiety and at least 2 compounds selected from angeloyl, acetyl, tigloyl, senecioyl, or derivative thereof or a compound capable performing the function of angeloyl.
- a compound selected from formula (1A) comprise a sugar moiety or a compound capable of performing the function of sugar moiety and at least 2 compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 is a compound comprising CH2OCCH3, CH2COOalkyl, CH2OH, COOH, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- a sugar moiety is a segment of molecule comprising one or more sugar group. Substitution, deletion and/or addition of any group in the above-described compounds will be apparent to one of ordinary skill in the art based on the teaching of this application. In a further embodiment, the substitution, deletion and/or addition of the group(s) in the compound of the invention does not substantially affect the biological function of the compound.
- This invention provides a compound selected from a compound of formula (1B):
- R1 comprise compound selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, dibenzoyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, heterocylic or heteroraryl or derivative thereof;
- R2 comprise compound selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R4 represents CH2OR6, COOR6 wherein R6 is selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, dibenzoyl
- R1 represent a sugar moiety comprises at least two compound selected from, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R2 represent a sugar moiety comprises at least one compound selected from, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R2 represent a sugar moiety or a compound comprises at least two compound selected from, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 wherein R6 is a sugar moiety which comprises at least one compound selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R6 is a sugar moiety which comprises at least one compound selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 wherein R6 is a sugar moiety which comprises at least two compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R6 is a sugar moiety which comprises at least two compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 wherein R6 is a sugar moiety which comprises at least two compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl,
- R4 represents CH2OR6, COOR6 wherein R6 is a sugar moiety which comprises at least two compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, dibenzoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R6 is a sugar moiety which comprises at least two compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, dibenzoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 of formula (1B), at least two of R1, R2 and R6 comprise the compound selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 of formula (1B), at least two of R1, R2 and R6 comprise angeloyl, benzoyl, alkenoyl, or derivative thereof.
- R4 is a compound comprising CH2OCCH3, CH2COOalkyl, CH2OH, COOH, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises at least one sugar, glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid or galacturonic acid, or derivative thereof, or the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises two sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid, galacturonic acid, and derivative thereof, and the combination thereof.
- the sugar moiety comprises two sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid, galacturonic acid, and derivative thereof, and the combination thereof
- R5 represents sugar moiety, wherein the sugar moiety comprises three sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid, galacturonic acid, and derivative thereof, and the combination thereof.
- sugar moiety comprises three sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid, galacturonic acid, and derivative thereof, and the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises at least four sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid, galacturonic acid, and derivative thereof, and the combination thereof.
- sugar moiety comprises at least four sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid, galacturonic acid, and derivative thereof, and
- R5 represents sugar moiety, wherein the sugar moiety comprises three sugars selected from glucose, galactose, rhamnose, arabinose, xylose, fucose, and derivative thereof, and the combination thereof.
- R5 represents sugar moiety or a compound capable of performing the function of the sugar moiety.
- the R5 represents H.
- R4 represents H or OH or CH3.
- positions 23, 24, 25, 26, 29, 30 of the compound independently comprise CH3, CH2OH, CHO, COOH, COOalkyl, COOaryl, COO-heterocyclic, COO-heteroaryl, CH 2 Oaryl, CH 2 O-heterocyclic, CH 2 O-heteroaryl, alkyls group, acetyl group or derivative thereof.
- R5 represents sugar moiety selected from D-glucose, D-galactose, L-rhamnose and L-arabinose and their combination thereof.
- R5 represents sugar moiety comprising D-glucose, D-galactose and L-arabinose or their combination thereof.
- R5 represents sugar moiety comprising D-glucose, D-galactose, L-rhamnose, D-xylose or L-arabinose or their combination thereof or derivative thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises at least two sugar selected from D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid and derivative thereof, and the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises three sugars selected from D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid and derivative thereof, and the combination thereof.
- R5 represents sugar moiety, wherein the sugar moiety comprises at least three sugars selected from D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid and derivative thereof, and the combination thereof.
- R1 and R2 independently comprise an angeloyl group.
- R1 is a sugar moiety or a compound which comprise two angeloyl groups.
- R1 and R2 independently comprise a benzoyl group.
- R1 is a sugar moiety which comprises two benzoyl groups.
- R3 represents H or OH.
- a sugar moiety is a segment of molecule comprising one or more sugar group. Substitution, deletion and/or addition of any group in the above-described compounds will be apparent to one of ordinary skill in the art based on the teaching of this application. In a further embodiment, the substitution, deletion and/or addition of the group(s) in the compound of the invention does not substantially affect the biological function of the compound.
- This invention provides a compound selected from a compound of formula (1C):
- R1 and R2 independently represent angeloyl group
- R3 represents H or OH
- R4 represent CH2OR6 wherein R6 is H
- R5 represents sugar moiety or D-glucose, D-galactose or its derivatives.
- R7 represent COOH
- R1 and R2 independently represent angeloyl group;
- R3 represents H or OH;
- R4 represents COOR6 wherein R6 is H;
- R5 represents sugar moiety or D-glucose, D-galactose or its derivatives.
- R7 represent COOH.
- R1 represents H;
- R2 represents angeloyl group;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6; wherein R6 is an angeloyl group or acetyl group.
- At least two of R1, R2, and R6 comprise an angeloyl group or acid having five carbons;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6; and wherein R6 is angeloyl group, H, acetyl group, tigloyl group, senecioyl group, or an acid having two to five carbons.
- At least one angeloyl from R1 or R2 is replaced by acetyl group, tigloyl group, senecioyl group, or an acid having two to five carbons;
- R3 represents H or OH;
- R4 represents CH2OR6 or COOR6 wherein R6 is angeloyl group.
- R4 represents CH2OR6 or COOR6; at least one of R1, R2, and R6 is a sugar moiety comprising at least two angeloyl groups, acetyl group, tigloyl group, senecioyl group, or an acid having two to five carbons or combination thereof.
- positions 24 of the compound comprise CH3 or CH2OH.
- R7 represent COOH, CH2OH or CH3.
- R7 represent CH3, CH2OH, CHO, COOH, COOalkyl.
- R7 represent CH3, CH2OH, CHO; COOH, COOalkyl, COOaryl, COO-heterocyclic, COO-heteroaryl, CH 2 Oaryl, CH 2 O-heterocyclic, CH 2 O-heteroaryl, alkyls group, acetyl group or derivative thereof.
- positions 24 of the compound comprise CH3 or CH2OH.
- positions 23, 24, 25, 26, 29, 30 of the compound independently comprise CH3, CH2OH.
- positions 23, 24, 25, 26, 29, 30 of the compound independently comprise CH3, CH2OH, CHO, COOH, COOalkyl, COOaryl, COO-heterocyclic, COO-heteroaryl, CH 2 Oaryl, CH 2 O-heterocyclic, CH 2 O-heteroaryl, alkyls group, acetyl group or derivative thereof.
- R5 represents sugar moiety comprising glucose or galactose.
- R5 represents sugar moiety, or D-glucose, D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid or D-galacturonic acid, or derivative thereof, or the combination thereof.
- R5 represents sugar moiety, glucose, galactose, rhamnose, arabinose, xylose, fucose or derivative thereof.
- R5 represents sugar moiety, glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid or galacturonic acid, or derivative thereof, or the combination thereof.
- the R5 represents H.
- R4 represents CH2OR6 wherein R6 represent H or acetyl group.
- R4 represents H or OH or CH3.
- R1 or/and R2 is a functional group capable of performing the function of the angeloyl.
- R5 represents a compound capable of performing the function of the sugar moiety.
- R1 and R2 are selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R1 or/and R2 is a sugar moiety comprise two of compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R1 and R2 are selected from H, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl;
- R4 represents CH2OR6 or COOR6; wherein R6 is selected from H, COCH3, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 or CH2COOR6; at least two of R1, R2 and R6 are selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 or CH2COOR6;
- R1, R2 and/or R6 is/are sugar moiety, which comprise compounds selected from H angeloyl, acetyl, tigloyl, senecioly, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 represents CH2OR6, COOR6 or CH2COOR6;
- R1, R2 and/or R6 is/are sugar moiety, which comprise at least 2 compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- a compound selected from formula (1C) comprises at least 2 compounds selected from angeloyl, acetyl, tigloyl, senecioyl, or derivative thereof or a compound capable performing the function of angeloyl.
- a compound selected from formula (1C) comprise at least 2 compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R4 is a compound comprising CH2OCCH3, CH2COOalkyl, CH2OH, COOH, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- a sugar moiety is a segment of molecule comprising one or more sugar group.
- substitution, deletion and/or addition of any group in the above-described compounds will be apparent to one of ordinary skill in the art based on the teaching of this application.
- substitution, deletion and/or addition of the group(s) in the compound of the invention does not substantially affect the biological function of the compound.
- This invention provides a compound selected from a compound of formula (1D):
- R1 comprise compound selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R2 comprise compound selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R4 represents CH2OR6, COOR6 wherein R6 is selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl,
- R7 represent CH3, CH2OH, COOH, COOalkyl
- R7 represent CH3, CH2OH, CHO, COOH, COOalkyl, COOaryl, COO-heterocyclic, COO-heteroaryl, CH 2 Oaryl, CH 2 O-heterocyclic, CH 2 O-heteroaryl, alkyls group, acetyl group or derivative thereof.
- R1 represent a compound comprising a sugar moiety comprises at least two compound selected from, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R1 represent a compound comprising a sugar moiety comprises at least one compound selected from, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R2 represent a compound comprising a sugar moiety comprises at least one compound selected from, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R2 represent a compound comprising a sugar moiety or a compound which comprises at least two compound selected from, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R4 represents CH2OR6, COOR6 wherein R6 is a sugar moiety which comprises at least one compound selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R4 represents CH2OR6, COOR6 wherein R6 is a sugar moiety which comprises at least two compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R4 represents CH2OR6, COOR6 wherein R6 is a sugar moiety which comprises at least two compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl,
- R4 represents CH2OR6, COOR6 wherein R6 is a sugar moiety which comprises at least two compounds selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, dibenzoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R4 represents CH2OR6, COOR6 wherein at least two of R1, R2 and R6 comprise the compound selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof;
- R4 is a compound comprising CH2OCCH3, CH2COOalkyl, CH2OH, COOH, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof.
- R5 represents sugar moiety, glucose, galactose, rhamnose, arabinose, xylose, fucose, allose, altrose, gulose, idose, lyxose, mannose, psicose, ribose, sorbose, tagatose, talose, fructose, alduronic acid, glucuronic acid or galacturonic acid, or derivative thereof, or the combination thereof.
- R5 represents a compound capable of performing the function of the sugar moiety.
- the R5 represents H.
- R4 represents H or OH or CH3.
- position 24 of the compound comprise CH3 or CH2OH
- positions 23, 24, 25, 26, 29, 30 of the compound independently comprise CH3, CH2OH, CHO, COOH, COOalkyl, COOaryl, COO-heterocyclic, COO-heteroaryl, CH 2 Oaryl, CH 2 O-heterocyclic, CH 2 O-heteroaryl, alkyls group, acetyl group or derivative thereof.
- R5 represents sugar moiety comprising L-glucose, D-galactose, L-rhamnose, or/and L-arabinose.
- R1 and R2 independently represent angeloyl group; In a further embodiment, R1 is a sugar moiety or rhamnose which comprise two angeloyl groups.
- R3 represents H or OH
- a sugar moiety is a segment of molecule comprising one or more sugar group. Substitution, deletion and/or addition of any group in the above-described compounds will be apparent to one of ordinary skill in the art based on the teaching of this application. In a further embodiment, the substitution, deletion and/or addition of the group(s) in the compound of the invention does not substantially affect the biological function of the compound.
- This invention provides a method of inhibiting tumor cell growth comprising administering to a subject, in need thereof, an appropriate amount of triterpenoidal saponins comprising two or more angeloyl groups or comprising the structure of FIGS. 1-27 .
- This invention provides a composition comprising the compounds as described above effective in reducing or inhibiting cancer growth.
- the cancer includes but is not limited to bladder cancer, bone cancer, skin cancer and ovarian cancer.
- the saponins isolated from Xanthoceras sorbifolia with the characteristic structure described in the present invention can be used for anti-cancer therapy.
- the cancer includes but is not limited to bladder cancer, bone cancer, skin cancer and ovarian cancer.
- This invention provides a composition comprising the above described compounds or their derivatives for treating human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS), flu disease or inhibits virus activities.
- HIV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- flu disease or inhibits virus activities.
- the biologically active triterpenoid saponins structures are shown in FIG. 1
- Triterpenoid saponins comprises the formula as following:
- This invention provides a composition comprising the compounds as described above effective in reducing or inhibiting cancer growth.
- the cancer includes but is not limited to bladder cancer, bone, cancer, skin cancer and ovarian cancer.
- composition for reducing or inhibiting cancer growth comprising any of compound selected from following:
- composition for reducing or inhibiting cancer growth comprising any of compound selected from following:
- Triterpenoid saponins with the characteristic structure mentioned above in this invention can be used to reduce or inhibit cancer growth.
- the cancer includes but is not limited to bladder cancer, bone cancer, skin cancer and ovarian cancer.
- This invention also provides a composition comprising the above described compounds or their derivatives capable of inhibiting human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities.
- HIV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- flu disease or capable of inhibiting viral activities.
- the saponins isolated from Harpullia austro - calcdonica with the characteristic structure described in the present invention can be used for anti-cancer therapy.
- the cancer includes but is not limited to bladder cancer, bone cancer and ovarian cancer.
- This invention provides a composition comprising the above described compounds or their derivatives for inhibiting human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS), flu disease or inhibits virus activities.
- HAV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- FIG. 11A The biologically active triterpenoid saponins structures are shown in FIG. 11A . See also Phytochemistry 59 (2002) 825-832, Triterpenoid saponins and acylated prosapogenins from Harpullia austro - calcdonica.
- R3 CH2OH or CH3 or CHO.
- the biologically active triterpenoid saponins structures are shown in FIG. 15 . See also Phytochemistry 66 (2005) 825-835, Haemolytic acylated triterpenoid saponins from Harpullia austro - caledonica.
- Triterpenoid saponins isolated from seeds of Aesculus chinensis having the characteristic structure(s) as disclosed in the present invention can be used in anti-cancer therapy.
- the cancer that triterpenoid saponins is effective against includes but is not limited to bladder cancer, bone cancer and ovarian cancer.
- This invention provides a composition comprising the above-described compounds and their derivatives for inhibiting cancer, Severe Acute Respiratory Syndrome (SARS), flu disease or inhibits virus activities.
- SARS Severe Acute Respiratory Syndrome
- Triterpenoidal saponins comprise the structures shown or described in FIG. 7A .
- R1 R2 R3 1 Tigloyl Acetyl H 2 Angeloyl Acetyl H 3 Tigloyl H H 4 Angeloyl H Acetyl 5 H Tigloyl Acetyl 6 H Angeloyl Acetyl 7 H H Tigloyl 8 H H Angeloyl
- Aesculus glabra Willd. Aesculus hippocastanum, Aesculus indica, Aesculus lantsangensis Hu et Fang, wangii Aesculus megaphylla Hu et Fang, chinensis Aesculus neglecta, Aesculus octandra Marsh., Aesculus parvifiora, Aesculus pavia, Aesculus polyneura Hu et Fang, Aesculus tsianguii Hu et Fang, Aesculus sylvatica, Aesculus turbinata, Aesculus wangii Hu, Aesculus wangii var.
- ruticola Hu et Fang or Aesculus wilsonii having the characteristic structure(s) as disclosed in the present invention can be used in anti-cancer therapy.
- the cancer that triterpenoid saponin is effective against includes but is not limited to bladder cancer, bone cancer and ovarian cancer.
- This invention provides a composition comprising the above-described compounds and their derivatives for inhibiting cancer, Severe Acute Respiratory Syndrome (SARS), flu disease or inhibits virus activities.
- SARS Severe Acute Respiratory Syndrome
- Triterpenoid saponins comprise the structures shown or described in FIG. 7A , 7 B, 7 C, 7 D.
- Position 23-27 and 28-30 are attached with CH3 or CH2OH or COOH or CHO
- Aesculus glabra Willd. Aesculus hippocastanum, Aesculus indica, Aesculus lantsangensis Hu et Fang, wangii Aesculus megaphylla Hu et Fang, chinensis Aesculus neglecta, Aesculus octandra Marsh., Aesculus parviflora, Aesculus pavia, Aesculus polyneura Hu et Fang, Aesculus tsianguii Hu et Fang, Aesculus sylvatica, Aesculus turbinata, Aesculus wangii Hu, Aesculus wangii var.
- ruticola Hu et Fang or Aesculus wilsonii having the characteristic structure(s) as disclosed in the present invention can be used in anti-cancer therapy.
- the cancer that triterpenoid saponin is effective against includes but is not limited to bladder cancer, bone cancer, skin cancer and ovarian cancer.
- This invention provides a composition comprising the above-described compounds and their derivatives for inhibiting cancer, Severe Acute Respiratory Syndrome (SARS), flu disease or inhibits virus activities.
- SARS Severe Acute Respiratory Syndrome
- Triterpenoidal saponins comprise the structures shown or described in FIG. 16A , 16 B.
- Triterpenoid saponins isolated from the plants described in this invention with the characteristic structure mentioned in this invention can be used to reduce or inhibit cancer growth.
- the cancer includes but is not limited to bladder cancer, bone cancer, skin cancer and ovarian cancer.
- This invention also provides a composition comprising the above described compounds or their derivatives capable of inhibiting human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities.
- HIV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- flu disease or capable of inhibiting viral activities.
- Triterpenoid saponins isolated from roots of Camellia sinensis var. assamica , showed in FIG. 13 with the characteristic structure mentioned in this invention is effective in inhibiting or reducing cancer growth.
- the cancer includes but is not limited to bladder cancer, bone cancer, skin cancer and ovarian cancer.
- This invention provides a composition comprising the above described compounds and their derivatives for inhibiting human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS), flu disease or inhibits virus activities. See also Phytochemistry 53 (2000) 941-946 Triterpenoid saponins from the roots of tea plant ( Camellia sinensis var. assamica ).
- Triterpenoid saponins isolated from Pittosporum viridiflorum with the characteristic structure mentioned in this invention can be used to reduce or inhibit cancer growth.
- the cancer includes but is not limited to bladder cancer, bone cancer, skin cancer and ovarian cancer.
- This invention also provides a composition comprising the above described compounds or their derivatives capable of inhibiting human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities.
- HIV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- flu disease or capable of inhibiting viral activities.
- the triterpenoid saponins isolated from Pittosporum tobira with the characteristic structure mentioned in this invention can be used for anti-cancer therapy.
- the cancer includes but is not limited to bladder cancer, bone cancer and ovary cancer.
- This invention also provides a composition comprising the above described compounds and their derivatives capable of inhibiting human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities.
- HIV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- the triterpenoid saponins isolated from Maesa lanceolata with the characterized structure mentioned in this invention can be used to reduce or inhibit cancer growth.
- the cancer includes but is not limited to bladder cancer, bone cancer and ovarian cancer.
- This invention also provides a composition comprising the above described compounds or their derivatives capable of inhibiting human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities.
- HIV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- flu disease or capable of inhibiting viral activities.
- the triterpenoid saponins isolated from Xanthoceras sobifolia with the characteristic structure mentioned in this invention can be used to reduce or inhibit cancer growth.
- the cancer includes but is not limited to bladder cancer, bone cancer and ovarian cancer.
- This invention also provides a composition comprising the above described compounds or their derivatives capable of inhibiting human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities.
- HIV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- This invention provides a method for inhibiting tumor cell growth, human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities, comprising contacting an effective amount of the compounds in FIG. 1 to FIG. 27 .
- the above described compound comprising at least two angeloyl groups at carbon 21, 22 and 28.
- the above described compound comprising at least two angeloyl groups, tigloyl groups, senecioyl groups or acetyl group or their combinations.
- This invention provides a method for inhibiting tumor cell growth, human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities, comprising contacting an effective amount of the compounds in the FIGS. 3A and B wherein the compound comprises two angeloyl groups at any two of R1, R2 and R4; or the compounds in FIGS. 5A , 5 B, 6 A and 6 B wherein the compound comprises two angeloyl groups at any two of R1, R2, R3 and R4; or the compounds in FIGS. 7A , 7 B, 7 C and 7 D wherein the compound comprises two angeloyl groups at any two of R1, R2 and R3; or the compounds in FIG.
- HCV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- the compound comprises two angeloyl groups at any two of R1, R2 and R3; or the compounds in FIGS. 9A , 9 B and 10 wherein the compound comprises two angeloyl groups at any two of R1, R2 and R3; or the compounds in FIGS. 11 , 12 and 13 wherein the compound comprises angeloyl groups at R1 and R2; or the compounds in FIG. 14 wherein the compound comprises two angeloyl groups at any two of R1, R2 and R3; or the compounds in FIG. 15 wherein the compound comprises two angeloyl groups; or the compounds in FIG. 16 wherein the compound comprises two angeloyl groups at any two of R1, R2 and R6; or the compounds in FIG. 16-25 wherein the compound comprises two angeloyl groups.
- This invention provides a method for inhibiting tumor cell growth, human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities, comprising contacting an effective amount of the above described compounds.
- the compound is a triterpenoidal saponin or a sapongenin comprising two angeloyl groups attached to carbon 21 and 22 of its sapogenin.
- the sapongenin comprising any two of angeloyl groups, tigloyl groups or senecioyl groups, or their combinations thereof attached to carbon 21 and 22 of its sapogenin.
- the compound is a triterpenoidal saponin or a sapongenin comprising any two of angeloyl groups, tigeloyl groups or senecioyl groups, or their combinations thereof attached to a sugar moiety which bonds to carbon 21 or 22.
- the compound is a triterpenoidal saponin or sapongenin comprising at least any one of angeloyl group, tigloyl group, or senecioyl group, or their combinations thereof attached to carbon 21 and/or 22 of its sapogenin.
- the compound is a triterpenoidal saponin or a sapongenin comprising at least two of angeloyl group or tigloyl group or senecioyl group, or their combinations thereof attached to a sugar moiety which bonds to carbon 21 or 22.
- the compound is a triterpenoidal saponin or a sapongenin comprising at least two angeloyl groups attached to carbon 21, 22 or 28 of its sapogenin.
- the compound is a triterpenoidal saponin or a sapongenin comprising any two of angeloyl groups, tigeloyl groups or senecioyl groups, or their combinations thereof attached to a sugar moiety which bonds to carbon 21, 22 or 28.
- the compound is a triterpenoidal saponin or a sapongenin comprising a sugar moiety comprises at least two angeloyl groups attached to carbon 21, 22 or 28 of its sapogenin.
- the compound is a five ring triterpene comprising at least two angeloyl group, attached to the side chains at one end of the five rings of its sapogenin and a sugar moiety is attached to the side chains of the ring at the other end of the triterpene.
- the compound comprising at least any two of angeloyl group, tigloyl group, or senecioyl group, or their combinations thereof.
- the compound is a sapogenin or triterpene comprising at least two angeloyl group, attached to the side chains of its sapogenin and a sugar moiety is attached to the side chains of the triterpene or sapogenin.
- the compound comprising at least any two of angeloyl group, tigloyl group, or senecioyl group, or their combinations thereof.
- the compound comprises at least two angeloyl group, attached to the side chains of a compound and a sugar moiety is attached to a side chain of the compound.
- the compound comprising at least any two of angeloyl group, tigloyl group, or senecioyl group, or their combinations thereof.
- the angeloyl can be replaced by a function group which has the function as angeloyl group.
- a sugar moiety or chain with one or more sugar such as D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid or D-galacturonic acid, or their combinations thereof, or their derivatives thereof is attached to carbon 3.
- the compound is a triterpene or sapongenin comprising at least any one of angeloyl group, tigloyl group or senecioyl group, or their combinations thereof attached to it.
- the compound is a triterpene or sapongenin comprising at least one of angeloyl group, tigloyl group or senecioyl group, or their combinations thereof attached to a sugar moiety which bonds to it.
- At least one sugar moiety with one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, or alduronic acid, D-glucuronic acid or D-galacturonic acid, or their combinations thereof, or their derivatives thereof is attached to the triterpene.
- bonds 23-30 are attached with CH3 or CH2OH or COOH or acetyl group.
- the activities of a saponin compound for regulating or inhibiting tumor cell growth are based on or attributed to its structure that comprises functional group(s) such as angeloyl group, tigloyl group, senecioyl group or acetyl group, or their combinations thereof.
- the Compound Y1 and Compound Y2 which comprise with two angeloyl groups show the inhibition on the growth of ovarian cancer cells. See FIG. 29 .
- the Compound Y, Y8, Y9 and Y10 which comprise with two angeloyl groups show the inhibition on ovarian cancer cells as determined by MTT assay. See FIG. 30 .
- the compound with single angeloyl group shows weaker anticancer activity than a compound with two angeloyl groups. See FIG. 28 .
- the compound with two angeloyl groups is more potency than the one with on angeloyl for inhibiting human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS), flu disease or inhibits virus activities.
- HAV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- flu disease flu disease or inhibits virus activities.
- composition comprising the compounds comprises the structure of:
- R1 and R2 are angeloyl group.
- R1 and R2 are angeloyl groups, tigloyl groups, senecioyl groups or acetyl group or their combinations.
- R1 and R2 comprise compound selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, heterocylic or heteroraryl or derivative thereof.
- the above compounds can be used for inhibiting tumor cell growth, human immunodeficiency virus (HIV), Severe Acute Respiratory Syndrome (SARS) or flu disease, or capable of inhibiting viral activities, comprising contacting an effective amount of the above described compounds.
- HIV human immunodeficiency virus
- SARS Severe Acute Respiratory Syndrome
- flu disease or capable of inhibiting viral activities, comprising contacting an effective amount of the above described compounds.
- This invention provides a method of inhibiting the growth of ovarian cancer, breast cancer, brain cancer, bladder cancer, prostate cancer, bone cancer, skin cancer, leukocyte cancer, liver cancer or leukemia in a subject, comprising administering to a subject, in need thereof, an effective amount of the compound which comprises any of the above structures to said subject.
- This invention provides a method for inhibiting tumor cell growth, regulating cell growth, reducing inflammation, in a subject, comprising administering to a subject, in need thereof, an effective amount of the compound which comprises any of the above structures to said subject.
- This invention provides a method for reducing leg swelling, reducing the symptom of chronic venous insufficiency, peripheral edema, antilipemic, chronic venous disease, varicose vein disease, varicose syndrome, venous stasis, Expectorant, peripheral vascular disorders, cerebro-organic convulsion, cerebral circulation disorder, cerebral edema, psychoses, dysmenorrheal, hemorrhoids, episiotomies, haemonhoids, peripheral oedema formation or postoperative swelling; for reducing symptoms of leg pain; for treating pruritis, lower leg volume, for reducing symptoms of pain; thrombosis, thromophlebitis; for preventing gastric ulcers antispasmotic comprising administering to a subject, in need thereof, an effective amount of the composition of this invention.
- This invention provides a composition comprising the compounds of the invention for treating enuresis and frequency micturition, and for improving the functions of the central nervous system including signaling the bladder to wake up from deep sleep or to relax the bladder so that it can store more urine.
- the compounds of the invention can be used to relax the detrusor tension caused by aging, stress, nervousness, over-activity, instability, hyper-reflexia, and uninhibited bladder.
- the compounds may be used for relaxing the contracted bladder tissue induced by acetylcholine (Ach).
- the compounds identified and isolated from extract of this invention may be used as acetylcolinesterase, an AChE inhibitor, for regulating Antidiuretic hormone (ADH), which reduces the volume of urine, and as an anti-inflammatory agent.
- the compounds of the invention can be used for accelerating the growth of bladder, for suppressing deep sleep, for increasing alertness in a sleeping subject, for modulating the release, breakdown and uptake of Antidieuretic hormone (ADH) and its receptors, for modulating the secretion, breakdown and uptake of Adrenocorticotropic hormone (ACTH) and its receptors, for modulating the release, breakdown and uptake of 5-hydroxytryptamine and its receptors, for modulating the release, breakdown and uptake of Acetylcholine (Ach) and its receptors, for modulating the release, breakdown and uptake of Adrenaline (AD) and its receptors, for modulating the release, breakdown and uptake of Dopamine (DA) and its receptors, for modulating the release, breakdown and uptake of Norepinephrine (NE) and its receptors, for preventing sleep paralysis, for modulating the formation, release, breakdown and activity of neuropeptides and their receptors.
- ADH Antidieuretic hormone
- ACTH Adrenocorticotropic
- This invention provides a composition comprising the compounds of the invention for treating cancers; for inhibiting virus; for preventing cerebral aging; for improving memory; improving cerebral functions, for curing enuresis, frequent micturition, urinary incontinence, dementia, Alzheimer's disease, autism, brain trauma, Parkinson's disease or other diseases caused by cerebral dysfunctions; for treating arthritis, rheumatism, poor circulation, arteriosclerosis, Raynaud's syndrome, angina pectoris, cardiac disorder, coronary heart disease, headache, dizziness, kidney disorder; cerebrovascular disease; inhibiting NF-Kappa B activation; for treating brain edema, sever acute respiratory syndrome, respiratory viral diseases, chronic venous insufficiency, hypertension, chronic venous disease, anti-oedematous, anti inflammatory, haemonhoids, peripheral oedema formation, varicose vein disease, flu, post traumatic edema and postoperative swelling; for inhibiting ethanol absorption; for lowering blood sugar; for regulating the a
- This invention provides a composition for treating chronic venous insufficiency, peripheral edema, antilipemic, chronic venous disease, varicose vein disease, varicose syndrome, venous stasis, Expectorant, peripheral vascular disorders, cerebro-organic convulsion, cerebral circulation disorder, cerebral edema, psychoses, dysmenorrheal, hemorrhoids, episiotomies, haemonhoids, peripheral oedema formation or postoperative swelling; for reducing symptoms of pain; for reducing symptoms of stomach pain; for reducing symptoms of leg pain; for treating pruritis, lower leg volume, thrombosis, thromophlebitis; for preventing gastric ulcers antispasmotic.
- This invention provides a composition for AntiMS, antianeurysm, antiasthmatic, antibradykinic, anticapillarihemorrhagic, anticephalagic, anticervicobrachialgic, antieclamptic, antiedemic, antiencaphalitic, antiepiglottitic, antiexudative, antiflu, antifracture, antigingivitic, antihematomic, antiherpetic, antihistaminic, antihydrathritic, antimeningitic, antioxidant, antiperiodontic, antiphlebitic, antipleuritic, antiraucedo, antirhinitic, antitonsilitic, antiulcer, antivaricose, antivertiginous, cancerostatic, corticosterogenic, diuretic, fungicide, hemolytic, hyaluronidase inhibitor, lymphagogue, natriuretic, pesticide, pituitary stimulant, thymolytic, vasoprotective, venotonic treatment,
- an angeloyl group combined with a coumarin shows strong anti-tumor activities.
- This invention provides a compound capable of reducing or inhibiting cancer cell growth, comprising the following structure:
- Angeloyl or tigloyl or senecioyl or acetyl in the above compound is replaced with hydroxyl group, the anti-tumor activities will be lost.
- the replacement of Angeloyl group with tigloyl group also reduces the anti-tumor activities. If we replace the acetyl group with Angeloyl group, the anti-tumor activities is increased.
- Angeloyl group combined with a coumarin shows activities.
- the structure is shown below:
- the above described compounds can be used for inhibiting cancer, wherein the cancer is not limited to breast cancer, leukocyte cancer, liver cancer, ovarian cancer, bladder cancer, prostate cancer, bone cancer, skin cancer, lung cancer, brain cancer, cervix cancer, KB cancer or brain cancer.
- a sugar moiety is a segment of molecule comprising one or more sugar group.
- Alkenyl means, unsaturated linear or branched structures and combinations thereof, having 1-7 carbon atoms, one or more double bonds therein.
- alkenyl groups include vinyl, propenyl, isopropenyl, butenyl, s- and t-butenyl, pentenyl, hexenyl, butadienyl, pentadienyl, hexadienyl.
- An aryl is a functional group of organic molecule derived from an aromatic compound such as benzene, a 6-14 membered carbocyclic aromatic ring system comprising 1-3 benzene ring s. If two or more aromatic rings are present, then the rings are fused together, so that adjacent rings share a common bond. Examples include phenyl and naphthyl.
- the aryl group may be substituted with one or more substitutes independently selected from halogen, alkyl or alkoxy.
- Acyl is a function group obtained from an organic acid by the removal of the carboxyl.
- Acyl groups can be written as having the general formula —COR, where there is a double bond between the carbon and oxygen.
- the names of acyl groups typically end in -yl, such as formyl, acetyl, propionyl, butyryl, benzoyl.
- Benzoyl is one of acyls, C6H5.COR, obtained from benzoic acid by the removal of the carboxyl.
- heterocyclic compound a compound containing a heterocyclic ring which refers to a non-aromatic ring having 1-4 heteroatoms said ring being isolated or fused to a second ring selected from 3- to 7-membered alicyclic ring containing 0-4 heteroatoms, aryl and heteroaryl, wherein said heterocyclic include pyrrolidinyl, piperazinyl, morpholinyl, trahydrofuranyl, imidazolinyl thiomorpholinyl, and the like.
- heterocyclyl groups derived from heteroarenes by removal of a hydrogen atom from any ring atom
- alkanoyl is the general name for an organic functional group R.CO—, where R represents hydrogen nor an alkyl group.
- R represents hydrogen nor an alkyl group.
- alkanoyl is selected from acetyl, propionoyl, butyryl, isobutyryl, pentanoyl and hexanoyl.
- Alkenoyl is alkenylcarbonyl in which alkenyl is defined above. Examples are pentenoyl(tigloyl) and hexenoyl(angeloyl).
- Alkyl is a radical containing only carbon and hydrogen atoms arranged in a chain, branched, cyclic or bicyclic structure or their combinations, having 1-18 carbon atoms. Examples included but not limited to methyl, ethyl, propyl isopropyl, butyl, s- and t-butyl, pentyl, hexyl, heptyl, octyl, nonyl, undecyl, dodecyl, tridecyl, tetradecyl, pentadecyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl.
- Absorption maximum Three absorption maximum were identified for plant extract; 207 nm, 278 nm and 500 nm.
- HTB-9 bladedder
- HeLa-S3 cervix
- DU145 prostate
- H460 lung
- MCF-7 breast
- K562 leukocytes
- HCT116 colon
- HepG2 liver
- U2OS bone
- T98G skin
- Skin OVCAR-3 (ovary).
- Cells were grown in culture medium (HeLa-S3, DU145, MCF-7, Hep-G2 and T98G in MEN (Earle's salts); HTB-9, H460, K562, OVCAR-3 in RPMI-1640; HCT-116, U2OS in McCoy-5A) supplemented with 10% fetal calf serum, glutamine and antibiotics in a 5% CO2 humidified incubator at 37° C.
- culture medium HeLa-S3, DU145, MCF-7, Hep-G2 and T98G in MEN (Earle's salts); HTB-9, H460, K562, OVCAR-3 in RPMI-1640; HCT-116, U2OS in McCoy-5A
- MTT assay The procedure for MTT assay followed the method described in (Carmichael et al., 1987) with only minor modifications.
- Cells were seeded into a 96-wells plate at concentrations of 10,000/well (HTB-9, HeLa, H460, HCT116, T98G, OVCAR-3), 15,000/well (DU145, MCF-7, HepG2, U2OS), or 40,000/well (K562), for 24 hours before drug-treatment. Cells were then exposed to drugs for 48 hours (72 hours for HepG2, U2OS, and 96 hours for MCF-7). After the drug-treatment, MTT (0.5 mg/ml) was added to cultures for an hour.
- the response of the cell lines to the tested extract can be categorized into four groups: Most sensitive, Sensitive; Semi-sensitive; and least sensitive.
- the plant extract was fractionated.
- Sample loading 1-2 ml, concentration: 100 mg/ml in 10% acetonitrile/TFA.
- Fraction Collector 5 ml/fractions (collect from 10% to 72% acetonitrile)
- Instrument AKTA-FPLC, P920 pump; Monitor UPC-900; Frac-900.
- NMR analysis The pure compound Y of Xanthoceras sorbifolia was dissolved in pyridine-D5 with 0.05% v/v TMS. All NMR spectra were acquired using a Bruker Avance 600 MHz NMR spectrometer with a QXI probe (1H/13C/15N/31P) at 298 K. The numbers of scans for 1D 1H spectra were 16 to 128, depending on the sample concentration. 2D HMQC spectra were recorded with spectral widths of 6000 ⁇ 24,000 Hz and data points of 2024 ⁇ 256 for t2 and t1 dimensions, respectively. The number of scans was 4 to 128.
- 2D HMBC were acquired with spectral widths of 6000 ⁇ 30,000 Hz and data points of 2024 ⁇ 512 for t2 and t 1 dimensions, respectively.
- the number of scans was 64.
- the 2D data were zero-filled in t 1 dimension to double the data points, multiplied by cosine-square-bell window functions in both t 1 and t 2 dimensions, and Fourier-transformed using software XWIN-NMR.
- the final real matrix sizes of these 2D spectra are 2048 ⁇ 256 and 2048 ⁇ 512 data points (F2 ⁇ F1) for HMQC and HMBC, respectively.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Epidemiology (AREA)
- Biotechnology (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- Microbiology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Alternative & Traditional Medicine (AREA)
- Mycology (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medical Informatics (AREA)
- Botany (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Vascular Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Steroid Compounds (AREA)
- Medicines Containing Plant Substances (AREA)
Abstract
Description
- R5=Bor C or S1 (see note 1)
- R1=A or B or C
- R2=A or B or C
- R4=A or B or C
- A=angeloyl or Tigloyl or Senecioyl
- B=acetyl
- C=H
- S1=sugar moiety comprising one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, or their derivatives.
- R5=B or C or S1 (see note 1)
- R1=A or B or C
- R2=A or B or C
- R3=A or B or C
- R4=A or B or C
- A=angeloyl or Tigloyl or Senecioyl
- B=acetyl
- C=H
- S1=sugar moiety comprising one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, or their derivatives.
- R5=B or C or S1 (see note 1)
- R1=A or B or C
- R2=A or B or C
- R3=A or B or C
- R4=A or B or C
- A=angeloyl or Tigloyl or Senecioyl
- B=acetyl
- C=H
- S1=sugar moiety comprising one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, or their derivatives.
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R6=H or OH
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R4=OH or H
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=Acetyl or H.
- R8=H or OH
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=Acetyl or H.
- R4=CH3 or CH2OH or COOH
- R6=CH3 or CH2OH or COOH
- R8=H or OH
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=Acetyl or H.
- R4=COOH OR COOMe or CH2OH
- R5=α-L-araf and R6=α-L-arap and R7=β-D-glup; or R5, R6, and R7 is a sugar moiety, or D-glucose, or D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid, or D-galacturonic acid, or their derivatives.
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=CH2OH or CH3 or CHO
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=CH2OH or CH3 or CHO
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=CH2OH or CH3 or CHO or COOCH3
- R4=S1=sugar moiety comprising one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic-acid, D-glucuronic acid, D-galacturonic acid, or/and their derivatives.
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=CH2OH or CH3 or CHO or COOCH3
- R4=sugar moiety comprising one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, or/and their derivatives.
- Wherein R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R3=angeloyl group or tigloyl group or senecioyl group or acetyl group or H.
- R4=CH2OH or CH3 or CHO or COOCH3
- R1=sugar moiety comprising one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, or/and their derivatives.
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or propanoyl or butanoly or acetyl group or H.
- R2=angeloyl group or tigloyl group or senecioyl group or propanoyl or butanoly or acetyl group or H.
- R3=angeloyl group or tigloyl group or senecioyl group or propanoyl or butanoly or acetyl group or H.
- R4=sugar moiety comprising one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, or/and their derivatives.
- R1=angeloyl or Tigloyl or Senecioyl or acetyl or H
- R2=angeloyl or Tigloyl or Senecioyl or acetyl or H
- R6=angeloyl or Tigloyl or Senecioyl or acetyl or H
- R3=H or OH
- R10=CH3 or CH2OH or CHO
- R5=D-glucose or D-galactose or L-rhamnose or L-arabinose or, D-xylose or alduronic acid or D-glucuronic acid or D-galacturonic acid or H
- R7=D-glucose or D-galactose or L-rhamnose or L-arabinose or, D-xylose or alduronic acid or D-glucuronic acid or D-galacturonic acid or H
- R8=D-glucose or D-galactose or L-rhamnose or L-arabinose or, D-xylose or alduronic acid or D-glucuronic acid or D-galacturonic acid or H
- R9=COOH or CH2OH
- R1=angeloyl or tigloyl or senecioly or acetyl group or H
- R2=angeloyl or tigloyl or senecioly or acetyl group or H
- R3=angeloyl or tigloyl or senecioly or acetyl group or H
- R6=H
- R4=CH3 or CH2OH or COOH
- R1=angeloyl or tigloyl or senecioly or acetyl group or H
- R2=angeloyl or tigloyl or senecioly or acetyl group or H
- R3=angeloyl or tigloyl or senecioly or acetyl group or H
- R6=angeloyl or tigloyl or senecioly or acetyl group or H
- R4=CH3 or CH2OH or COOH
- R5=acetyl or H or S1 (see note 1)
- R1=angeloyl or tigloyl or senecioly or acetyl group or H
- R2=angeloyl or tigloyl or senecioly or acetyl group or H
- R3=angeloyl or tigloyl or senecioly or acetyl group or H
- R6=angeloyl or tigloyl or senecioly or acetyl group or H
- R4=CH3 or CH2OH or COOH
- R5=acetyl or H or S1 (see note 1)
- S1=sugar moiety comprises one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, or/and their derivatives.
- R2, R3, R5, R6, R8, R9=angeloyl or tigloyl or senecioly or acetyl or H
- R4=CH3 or CH2OH or COOH
- S1=Sugar moiety comprises one or more sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, D-galacturonic acid, or/and their derivatives.
- R2, R3, R5, R6, R8, R9=angeloyl or tigloyl or senecioly or acetyl or H
- R4, R7=CH3 or CH2OH or COOH
- (a) extracting roots, kernel, leave, bark, stem, husk, seed, seed shell or fruit or combinations thereof of the above plant with organic solvents such as ethanol or methanol to obtain a organic extract;
- (b) collecting the organic extract;
- (c) refluxing the organic extract to obtain a second extract;
- (d) removing the organic solvent from the second extract to obtain a third extract;
- (e) drying and sterilizing the third extract to obtain a crude extract powder;
- (f) fractionating the crude extract powder into fractions or components. Fractionation may be achieved by HPLC and FPLC chromatography with silica gel, C18 or other equivalent solid phase materials;
- (g) monitoring the fractions. If using HPLC or FPLC, absorption wavelength at 207 nm to 500 nm may be used;
- (h) identifying the bioactive components of the crude extract;
- (i) purifying one or more bioactive components of the crude extract with chromatographic techniques that employ FPLC to obtain one or more fraction of the bioactive component; and
- (j) isolating the bioactive components with chromatographic techniques that employ preparative columns and HPLC.
% G=(TD−T0/TC−T0)×100 (1),
where TC or TD represent O.D. readings of control or drug-treated cells. When T0>TD, then the cytotoxicity (LC) expressed as % of the control is calculated as:
% LC=(TD−T0/T0)×100 (2)
or a salt, ester, metabolite or derivative thereof, wherein R1 and R2 represent angeloyl group; R3 represents H or OH; R4 represent CH2OR6; and wherein R6 is H; R5 represents at least one sugar moiety or its derivatives.
or a salt, ester or derivative thereof, wherein R1 represents angeloyl group; R2 represents angeloyl group; R3 represents OH or H; Positions 23, 24, 25, 26, 27, 29, of the compound independently comprise CH3, or CH2OH, or CHO, or COOH, alkyls group, or acetyl group, or derivative; R6 represents Ac or H; and R5 represents sugar moiety, wherein the sugar moiety comprises at least one sugar, or D-glucose, or D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid, or D-galacturonic acid, or their derivative thereof, or the combination thereof. In an embodiment, R5 represents a compound capable of performing the function of the sugar moiety. In another embodiment the sugar moiety comprises L-arabinose, D-glucose and/or D-galactose, or combinations thereof. In a further embodiment, any two of R1, R2 or R6 are angeloyl groups, or any one of R1, R2 or R6 is attached to a sugar moiety in which two angeloyl groups are attached to adjacent carbons of the monosaccharides. In a further embodiment, R1, R2, and R6 comprises angeloyl group, acetyl group, tigloyl group, senecioly group, or an acid with two to five carbons or combination thereof. In a further embodiment, at least one of R1, R2 or R6 is attached a sugar moiety, wherein sugar moiety comprises two angeloyl group, acetyl group, tigloyl group, senecioly group, acid having two to five carbons, or combinations thereof.
or a salt, ester or derivative thereof, wherein R1 represents angeloyl group; R2 represents angeloyl group; R3 represents OH or H; R4 represents CH3 or CH2OH or alkyls group or their derivatives; R6 represents Ac or H and R5 represents sugar moiety, wherein the sugar moiety comprises at least one sugar, or D-glucose, or D-galactose, or L-rhamnose, or L-arabinose, or D-xylose, or alduronic acid, or D-glucuronic acid, or D-galacturonic acid, or derivative thereof, or the combination thereof. In an embodiment, R5 represents a compound capable of performing the function of sugar moiety. In another embodiment the sugar moiety comprises L-arabinose, or D-glucose, or D-galactose, or combinations thereof. In a further embodiment, at least one of R1, R2 or R6 is attached a sugar moiety or rhamnose, wherein sugar moiety or rhamnose comprises two angeloyl group, acetyl group, tigloyl group, senecioly group, acid having two to five carbons, or combinations thereof.
or a salt, ester, metabolite or derivative thereof, wherein R1 and R2 represent angeloyl group; R3 represents H or OH; R4 represent CH2OR6; and wherein R6 is H or acetyl; R5 represents sugar moiety or D-glucose; R7 represents a sugar moiety or L-arabinose; R8 represents sugar moiety or D-galactose; R9 represent COOH or CH2OH.
or a salt, ester, metabolite or derivative thereof, wherein R1 and R2 represent angeloyl group; R3 represents H or OH; R6 represent H or acetyl; R9 represents COOH or CH2OH; R10 represent CH3 or CH2OH or COOH; R5, R7 and R8 are H or/and sugar moiety, wherein the sugar moiety comprises at least one sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, or D-galacturonic acid, or derivative thereof. In an embodiment, at least one of R1, R2, and R6 is a sugar moiety or compound comprising at least two angeloyl groups, acetyl group, tigloyl group, senecioly group, or an acid having two to five carbons or combination thereof. In another embodiment, at least two of R1, R2, and R6 comprise an angeloyl group
or a salt, ester or derivative thereof, wherein R1 represents angeloyl group; R2 represents angeloyl group; R3 represents OH or H; R4 represents CH3 or CH2OH; R7 represents H; and R5 represents D-glucose, D-Galactose, L-arabinose or H; and R6 represents D-glucose, D-Galactose, L-arabinose or H. In an embodiment, R5 or/and R6 are H or sugar moiety comprises at least one sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid, or D-galacturonic acid, or derivative thereof.
or a salt, ester, metabolite or derivative thereof, wherein R1 and R2 independently represent angeloyl group; R3 represents H or OH; R4 represent CH2OR6; and wherein R6 is H; R5 represents at least one sugar moiety or its derivatives.
or a salt, ester, metabolite or derivative thereof, wherein R1 comprise compound selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, dibenzoyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, heterocylic or heteroraryl or derivative thereof; R2 comprise compound selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof; R4 represents CH2OR6, COOR6 wherein R6 is selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof; R3 is H or OH; R5 represents sugar moiety, wherein the sugar moiety comprises at least one sugar, D-glucose, D-galactose, L-rhamnose, L-arabinose, D-xylose, alduronic acid, D-glucuronic acid or D-galacturonic acid or derivative thereof, or the combination thereof.
or a salt, ester, metabolite or derivative thereof, wherein R1 and R2 independently represent angeloyl group; R3 represents H or OH; R4 represent CH2OR6 wherein R6 is H; R5 represents sugar moiety or D-glucose, D-galactose or its derivatives. R7 represent COOH
or a salt, ester, metabolite or derivative thereof, wherein R1 comprise compound selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof; R2 comprise compound selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof; R4 represents CH2OR6, COOR6 wherein R6 is selected from hydrogen, angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, acyl, heterocylic or heteroraryl or derivative thereof; R3 is H or OH; R5 represents sugar moiety, or D-glucose or D-galactose; R7 represent COOH
- 3-O-{[β-D-galactopyranosyl(1→2)]-[α-L-arabinofuranosyl(1→3)]-β-D-glucuronopyranoside butyl ester}-21-O-acetyl-22-O-angeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene.
- 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21,22-O-diangeloyl-3β, 15α, 16α, 21β, 22α, 28-hexahydroxyolean-12-ene,
- 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21-O-(3,4-diangeloyl)-α-L-rhamnophyranosyl-22-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene,
- 3-O-[β-D-glucopyranosyl-(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21,22-O-diangeloyl-3β, 15α, 16α, 21β, 22α, 24β, 28-heptahydroxyolean-12-ene,
- 3-O-[β-glucopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21,22-O-diangeloyl-3β, 16α, 21β, 22α, 24β, 28-hexahydroxyolean-12-ene,
- 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21-O-(3,4-diangeloyl)-α-rhamnopyranosyl-28-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene,
- 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21,22-O-diangeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene,
- A) 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21,22-O-diangeloyl-3β, 15α, 16α, 21β, 22α, 28-hexahydroxyolean-12-ene,
- B) 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21-O-(3,4-diangeloyl)-α-L-rhamnophyranosyl-22-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- C) 3-O-[β-D-glucopyranosyl-(1→2)]-α-L-arabinofuranosyl(1→3)-(-D-glucuronopyranosyl-21,22-O-diangeloyl-3β, 15α, 16α, 21β, 22α, 24β, 28-heptahydroxyolean-12-ene
- D) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21,22-O-diangeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- E) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21-O-(3,4-diangeloyl)-α-rhamnopyranosyl-28-O-acetyl-3β, 16α,21β, 22α, 28-pentahydroxyolean-12-ene
- F) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21,22-O-diangeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- G) 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21,22-O-dibenzoyl-3β, 15α, 16α, 21β, 22α, 28-hexahydroxyolean-12-ene,
- H) 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21-O-(3,4-dibenzoyl)-α-L-rhamnophyranosyl-22-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- I) 3-O-[β-D-glucopyranosyl-(1→2)]-α-L-arabinofuranosyl(1→3)-(-D-glucuronopyranosyl-21,22-O-dibenzoyl-3β, 15α, 16α, 21β, 22α, 24β, 28-heptahydroxyolean-12-ene
- J) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21,22-O-dibenzoyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- K) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21-O-(3,4-dibenzoyl)-α-rhamnopyranosyl-28-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- L) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21,22-O-dibenzoyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- M) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21,22-O-dibenzoyl-3β, 15α, 16α, 21β, 22α, 28-hexahydroxyolean-12-ene,
- N) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21-O-(3,4-dibenzoyl)-α-L-rhamnophyranosyl-22-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- O) 3-O-[β-D-glucopyranosyl-(1→2)]-β-D-xyopyranosyl(1→3)-(-D-glucuronopyranosyl-21,22-O-dibenzoyl-3β, 15α, 16α, 21β, 22α, 24β, 28-heptahydroxyolean-12-ene
- P) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21,22-O-dibenzoyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- Q) 3-O-[β-galactopyranosyl(1→2)]-β-xyopyranosyl(1→3)-β-glucuronopyranosyl-21-O-(3,4-dibenzoyl)-α-rhamnopyranosyl-28-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- R) 3-O-[β-galactopyranosyl(1→2)]-β-xyopyranosyl(1→3)-β-glucuronopyranosyl-21,22-O-dibenzoyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- S) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21,22-O-diangeloyl-3β, 15α, 16α, 21β, 22α, 28-hexahydroxyolean-12-ene,
- T) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21-O-(3,4-diangeloyl)-α-L-rhamnophyranosyl-22-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- U) 3-O-[β-D-glucopyranosyl-(1→2)]-β-D-xyopyranosyl(1→3)-(-D-glucuronopyranosyl-21,22-O-diangeloyl-3β, 15α, 16α, 21β, 22α, 24β, 28-heptahydroxyolean-12-ene
- V) 3-O-[β-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-glucuronopyranosyl-21,22-O-diangeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- W) 3-O-[β-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-glucuronopyranosyl-21-O-(3,4-diangeloyl)-α-rhamnopyranosyl-28-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- X) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21,22-O-diangeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- A) 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21-O-angeloyl,22-O-benzoyl-3β, 15α, 16α, 21β, 22α, 28-hexahydroxyolean-12-ene,
- B) 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21-O-(3-angeloyl,4-benzoyl)-α-L-rhamnophyranosyl-22-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- C) 3-O-[β-D-glucopyranosyl-(1→2)]-α-L-arabinofuranosyl(1→3)-(-D-glucuronopyranosyl-21-O-angeloyl,22-O-benzoyl-3β, 15α, 16α, 21β, 22α, 24β, 28-heptahydroxyolean-12-ene
- D) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21-O-angeloyl,22-benzoyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- E) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21-O—F) (3-angeloyl,4-benzoyl)-α-rhamnopyranosyl-28-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- F) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21-O-angeloyl,22-O-benzoyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- G) 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21-O-benzoyl,22-O-angeloyl-3β, 15α, 16α, 21β, 22α, 28-hexahydroxyolean-12-ene,
- H) 3-O-[β-D-galactopyranosyl(1→2)]-α-L-arabinofuranosyl(1→3)-β-D-glucuronopyranosyl-21-O-(3-benzoyl,4-angeloyl)-α-L-rhamnophyranosyl-22-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- I) 3-O-[β-D-glucopyranosyl-(1→2)]-α-L-arabinofuranosyl(1→3)-(-D-glucuronopyranosyl-21-O-benzoyl,22-O-angeloyl-3β, 15α, 16α, 21β, 22α, 24β, 28-heptahydroxyolean-12-ene
- J) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21-O-benzoyl,22-O-angeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- K) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21-O-(3-benzoyl,4-angeloyl)-α-rhamnopyranosyl-28-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- L) 3-O-[β-galactopyranosyl(1→2)]-α-arabinofuranosyl(1→3)-β-glucuronopyranosyl-21-O-benzoyl,22-O-angeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- M) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21-O-angeloyl,22-O-benzoyl-3β, 15α, 16α, 21β, 22α, 28-hexahydroxyolean-12-ene,
- N) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21-O-(3-angeloyl,4-dibenzoyl)-α-L-rhamnophyranosyl-22-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- O) 3-O-[β-D-glucopyranosyl-(1→2)]-β-D-xyopyranosyl(1→3)-(-D-glucuronopyranosyl-21-O-21-O-angeloyl,22-O-benzoyl-3β, 15α, 16α, 21β, 22α, 24β, 28-heptahydroxyolean-12-ene
- P) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-2121-O-angeloyl,22-O-benzoyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- Q) 3-O-[β-galactopyranosyl(1→2)]-β-xyopyranosyl(1→3)-β-glucuronopyranosyl-21-O-(3-angeloyl,4-dibenzoyl)-α-rhamnopyranosyl-28-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- R) 3-O-[β-galactopyranosyl(1→2)]-β-xyopyranosyl(1→3)-β-glucuronopyranosyl-angeloyl,22-O-benzoyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- S) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21-O-benzoyl,22-O-angeloyl-3β, 15α, 16α, 21β, 22α, 28-hexahydroxyolean-12-ene,
- T) 3-O-[β-D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21-O-(3-benzoyl,4-angeloyl)-α-L-rhamnophyranosyl-22-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- U) 3-O-[β-D-glucopyranosyl-(1→2)]-β-D-xyopyranosyl(1→3)-(-D-glucuronopyranosyl-21-O-benzoyl,22-O-angeloyl-3β, 15α, 16α, 21β, 22α, 24β, 28-heptahydroxyolean-12-ene
- V) 3-O-[β-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-glucuronopyranosyl-21-O-benzoyl,22-O-angeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- W) 3-O-[β-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-glucuronopyranosyl-21-O-(3-benzoyl,4-angeloyl)-α-rhamnopyranosyl-28-O-acetyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
- X) 3-O-[D-galactopyranosyl(1→2)]-β-D-xyopyranosyl(1→3)-β-D-glucuronopyranosyl-21-O-benzoyl,22-O-angeloyl-3β, 16α, 21β, 22α, 28-pentahydroxyolean-12-ene
| Wherein | | R2 | R3 | |
| 1 | | Acetyl | H | |
| 2 | | Acetyl | H | |
| 3 | | H | H | |
| 4 | Angeloyl | H | Acetyl | |
| 5 | H | Tigloyl | Acetyl | |
| 6 | H | Angeloyl | Acetyl | |
| 7 | H | H | Tigloyl | |
| 8 | H | H | Angeloyl | |
- Wherein R1=angeloyl group or tigloyl group or senecioyl group or acetyl or H.
- R2=angeloyl group or tigloyl group or senecioyl group or acetyl group or acetyl or H.
- R3=angeloyl group or tigloyl group or senecioyl group or acetyl group or acetyl or H.
- R6=H or OH
- Wherein R1=angeloyl or Tigloyl or Senecioyl or acetyl or H
- R2=angeloyl or Tigloyl or Senecioyl or acetyl or H
- R6=angeloyl or Tigloyl or Senecioyl or acetyl or H
- R3=H or OH
- R10=CH3 or CH2OH or CHO
- R5=sugar moiety or D-glucose or D-galactose or L-rhamnose or L-arabinose or, D-xylose or alduronic acid or D-glucuronic acid or D-galacturonic acid or H
- R7=sugar moiety or D-glucose or D-galactose or L-rhamnose or L-arabinose or, D-xylose or alduronic acid or D-glucuronic acid or D-galacturonic acid or H
- R8=sugar moiety or D-glucose or D-galactose or L-rhamnose or L-arabinose or, D-xylose or alduronic acid or D-glucuronic acid or D-galacturonic acid or H
- R9=COOH or CH2OH
- Wherein R1=angeloyl group.
- R2=senecioyl group.
| R1 | | R3 | R4 | ||
| 1. | 2-acetoxy-2- | acetyl | | COOH | ||
| methybutanoyl | ||||||
| 2. | Angeloyl | | H | COOH | ||
| 3. | Angeloyl | | acetyl | COOH | ||
| 4. | Angeloyl | | H | COOH | ||
| 5. | H | | H | COOH | ||
| 7. | H | | H | COOMe | ||
| 8. | H | H | H | CH2OH | ||
| R5 = α-L-araf | ||||||
| R6 = α-L-arap | ||||||
| R7 = β-D-glup | ||||||
| compound | | R2 | R3 | |
| 2 | acetyl | H | angeloyl |
| 3 | H | acetyl | angeloyl |
| 5 | H | propanoyl | angeloyl |
| 7 | H | butanoyl | angeloyl |
| 8 | H | angeloyl | angeloyl |
| 4 | acetyl | acetyl | angeloyl |
| 6 | acetyl | propanoyl | angeloyl |
| 9 | acetyl | butanoyl | angeloyl |
| 10 | acetyl | angeloyl | angeloyl |
wherein R1 and R2 are angeloyl group. In embodiment, R1 and R2 are angeloyl groups, tigloyl groups, senecioyl groups or acetyl group or their combinations. In embodiment, R1 and R2 comprise compound selected from angeloyl, acetyl, tigloyl, senecioyl, alkyl, benzoyl, dibenzoyl, alkanoyl, alkenoyl, benzoyl alkyl substituted alkanoyl, aryl, heterocylic or heteroraryl or derivative thereof.
- Wherein the R1, R2=Angeloyl, or tigloyl, or senecioyl, or acetyl group.
- Wherein the R1, R2=Angeloyl or tigloyl or senecioyl or acetyl group.
- Wherein the R1, R2=Angeloyl or tigloyl or senecioyl or actyl group.
% G=(TD−T0/TC−T0)×100 (1)
where TC or TD represent O.D. readings of control or drug-treated cells. When T0>TD, then the cytotoxicity (LC) expressed as % of the control is calculated as:
% LC=(TD−T0/T0)×100. (2)
Results
- 2. Mix treated virus with cells.
- 3. Measure Virus production.
- 4. Negative control: no virus in cell.
- 5. Positive control—untreated virus mixed with cell.
- Result: The virus growth is inhibited after treatments of compound of this invention.
- 1) 5 groups of rats:
- 2) 3 dose groups (low, mid, high), 1 positive control (no drug, with croton oil), 1 negative control (no drug, no croton Oil)
- 3) Give drug (Compound Y) for 7 days in 3 dose groups (low, mid, high) of rats.
- 4) On
day 7, apply croton oil to the Give-Drug-groups after giving drug. - 5) At the 24th hour after applying croton oil, isolate the recto-anus tissue (10 mm) of rats.
- 6) Apply croton oil to control group. At the 24th hour after applying croton oil, isolate the recto-anus tissue (10 mm) of rats.
- 7) Compare the swelling of give-drug-groups with positive control (no drug, with croton oil) and negative control (no drug, no croton oil).
- 8) The result shows that Compound Y inhibiting the swelling of recto-annus induced by croton oil.
- 1) 5 groups of rats:
- 2) 3 dose groups (low, mid, high), 1 positive control (no drug, with Carrageenin), 1 negative control (no drug, no Carrageenin)
- 3) Give drug (Compound Y) for 7 days in 3 dose groups (low, mid, high) of rats.
- 4) On
day 7, apply Carrageenin to the Give-Drug-groups after givingdrug 10 min. - 5) Measure the swell volume of the paw of rats at 0.5, 1, 2, 4, 6 hours after applying the Carrageenin.
- 6) Compare the swelling of give-drug-groups with positive control (no drug, with Carrageenin) and negative control (no drug, no Carrageenin).
- 7) The result shows that Compound Y inhibiting the leg swelling.
Claims (23)
Priority Applications (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/344,682 US8841265B2 (en) | 2003-10-09 | 2008-12-29 | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| AU2009226063A AU2009226063A1 (en) | 2008-02-15 | 2009-02-13 | Blocking the metastasis of cancer cells and the uses of new compounds thereof |
| CA2715501A CA2715501A1 (en) | 2008-02-15 | 2009-02-13 | Blocking the metastasis of cancer cells and the uses of new compounds thereof |
| PCT/US2009/034115 WO2009117196A1 (en) | 2008-02-15 | 2009-02-13 | Blocking the metastasis of cancer cells and the uses of new compounds thereof |
| EP09721583A EP2254899A4 (en) | 2008-02-15 | 2009-02-13 | Blocking the metastasis of cancer cells and the uses of new compounds thereof |
| CN200980112951.5A CN101998964B (en) | 2008-02-15 | 2009-02-13 | Blocking the metastasis of cancer cells and the uses of new compounds thereof |
| US12/541,713 US8735558B2 (en) | 2005-02-14 | 2009-08-14 | Blocking the migration or metastasis of cancer cells by affecting adhesion proteins and the uses of new compounds thereof |
| US12/856,322 US8586719B2 (en) | 2005-04-27 | 2010-08-13 | Triterpenes for modulating gene expression and cell membrane, and as antiprotozoal agents |
| US13/841,053 US9382285B2 (en) | 2004-09-07 | 2013-03-15 | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US15/181,631 US10213451B2 (en) | 2004-09-07 | 2016-06-14 | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US16/285,634 US11046724B2 (en) | 2004-09-07 | 2019-02-26 | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US17/362,028 US20220024961A1 (en) | 2004-09-07 | 2021-06-29 | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
Applications Claiming Priority (16)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US50985103P | 2003-10-09 | 2003-10-09 | |
| US53210103P | 2003-12-23 | 2003-12-23 | |
| US60785804P | 2004-09-07 | 2004-09-07 | |
| US61381104P | 2004-09-27 | 2004-09-27 | |
| US61737904P | 2004-10-08 | 2004-10-08 | |
| PCT/US2004/033359 WO2005037200A2 (en) | 2003-10-09 | 2004-10-08 | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| PCT/US2004/043465 WO2005063273A1 (en) | 2003-12-23 | 2004-12-23 | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US10/906,303 US7524824B2 (en) | 2003-09-04 | 2005-02-14 | Composition comprising Xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US67528205P | 2005-04-27 | 2005-04-27 | |
| US67528405P | 2005-04-27 | 2005-04-27 | |
| US11/117,760 US7727561B2 (en) | 2001-08-31 | 2005-04-27 | Composition comprising Xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US11/131,551 US7262285B2 (en) | 2003-10-09 | 2005-05-17 | Anticancer biangeloyl saponins |
| PCT/US2005/031900 WO2006029221A2 (en) | 2004-09-07 | 2005-09-07 | Anti-tumor compounds with angeloyl groups |
| US11/267,523 US20060111310A1 (en) | 2003-10-09 | 2005-11-04 | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| US11/289,142 US7488753B2 (en) | 2003-10-09 | 2005-11-28 | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| US12/344,682 US8841265B2 (en) | 2003-10-09 | 2008-12-29 | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
Related Parent Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/289,142 Continuation US7488753B2 (en) | 2003-10-09 | 2005-11-28 | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| PCT/US2008/002086 Continuation-In-Part WO2008133766A1 (en) | 2004-09-07 | 2008-02-15 | Blocking the migration or metastasis of cancer cells by affecting adhesion proteins and the uses of new compounds thereof |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/412,659 Continuation-In-Part US20060263458A1 (en) | 2003-10-09 | 2006-04-27 | Composition comprising growth factor of xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| PCT/US2009/034115 Continuation-In-Part WO2009117196A1 (en) | 2004-09-07 | 2009-02-13 | Blocking the metastasis of cancer cells and the uses of new compounds thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20090156515A1 US20090156515A1 (en) | 2009-06-18 |
| US8841265B2 true US8841265B2 (en) | 2014-09-23 |
Family
ID=41017015
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/289,142 Expired - Fee Related US7488753B2 (en) | 2003-10-09 | 2005-11-28 | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| US12/344,682 Expired - Fee Related US8841265B2 (en) | 2003-10-09 | 2008-12-29 | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/289,142 Expired - Fee Related US7488753B2 (en) | 2003-10-09 | 2005-11-28 | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
Country Status (3)
| Country | Link |
|---|---|
| US (2) | US7488753B2 (en) |
| EP (1) | EP1876896A4 (en) |
| WO (1) | WO2006116656A2 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130190261A1 (en) * | 2003-10-09 | 2013-07-25 | Pacific Arrow Limited | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US20130338089A1 (en) * | 2004-09-07 | 2013-12-19 | Pacific Arrow Limited | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US9434677B2 (en) | 2009-07-16 | 2016-09-06 | Pacific Arrow Limited | Natural and synthetic compounds for treating cancer and other diseases |
| US9499577B2 (en) | 2009-07-16 | 2016-11-22 | Pacific Arrow Limited | Natural and synthetic compounds for treating cancer and other diseases |
| US10213451B2 (en) * | 2004-09-07 | 2019-02-26 | Pacific Arrow Limited | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US20190256543A1 (en) * | 2004-09-07 | 2019-08-22 | Pacific Arrow Limited | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7727561B2 (en) * | 2001-08-31 | 2010-06-01 | Pacific Arrow Limited | Composition comprising Xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US8614197B2 (en) * | 2003-10-09 | 2013-12-24 | Pacific Arrow Limited | Anti-tumor compounds with angeloyl groups |
| US7488753B2 (en) * | 2003-10-09 | 2009-02-10 | Pacific Arrow Limited | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| US7262285B2 (en) * | 2003-10-09 | 2007-08-28 | Pacific Arrow Limited | Anticancer biangeloyl saponins |
| US20060111310A1 (en) * | 2003-10-09 | 2006-05-25 | Chan Pui-Kwong | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| JP4815558B2 (en) * | 2003-10-09 | 2011-11-16 | パシフィック アロー リミテッド | COMPOSITION COMPRISING XANTHOCERASSORBIFOLIA EXTRACT, COMPOUND ISOLATED FROM THE EXTRACT, METHOD FOR PREPARING THEM, AND USE |
| US7514412B2 (en) * | 2003-10-09 | 2009-04-07 | Pacific Arrow Limited | Anticancer biangeloyl saponins |
| WO2008133766A1 (en) * | 2007-02-16 | 2008-11-06 | Pacific Arrow Limited | Blocking the migration or metastasis of cancer cells by affecting adhesion proteins and the uses of new compounds thereof |
| US8735558B2 (en) * | 2005-02-14 | 2014-05-27 | Pacific Arrow Limited | Blocking the migration or metastasis of cancer cells by affecting adhesion proteins and the uses of new compounds thereof |
| US20220024961A1 (en) * | 2004-09-07 | 2022-01-27 | Pacific Arrow Limited | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US8586719B2 (en) | 2005-04-27 | 2013-11-19 | Pacific Arrow Limited | Triterpenes for modulating gene expression and cell membrane, and as antiprotozoal agents |
| US20070082071A1 (en) * | 2005-10-07 | 2007-04-12 | Willimann John A | Composition for controlling the respiratory effects of inhaled pollutants & allergens |
| CN101553497B (en) * | 2006-09-01 | 2013-03-27 | 太平洋艾瑞有限公司 | Anti-tumor compounds for inhibiting cancer growth |
| DE102007044094A1 (en) * | 2007-09-14 | 2009-04-02 | Klaus-Otto von Yeppoon Gliszczynski | Production of leaf extracts of Pittosporum phillyraeoides and their use in medicine |
| US20120277308A1 (en) | 2010-07-16 | 2012-11-01 | Pacific Arrow Limited | compounds for treating cancer and other diseases |
| CN115197293A (en) * | 2010-07-16 | 2022-10-18 | 太平洋艾瑞有限公司 | Novel compounds useful for the treatment of cancer and other diseases |
| KR101873607B1 (en) * | 2010-07-16 | 2018-07-02 | 퍼시픽 애로우 리미티드 | New compounds for treating cancer and other diseases |
| US8632827B2 (en) * | 2011-12-13 | 2014-01-21 | Avon Products, Inc | Modulation of thymosin beta-4 in skin |
| CN102924618B (en) * | 2012-10-30 | 2014-10-08 | 中国人民解放军第四军医大学 | Triterpenoid saponin anti-myocardial ischemia compound |
| EP3061765B1 (en) | 2015-02-27 | 2018-01-31 | Warszawski Uniwersytet Medyczny | Protoescigenin derivative, process of its preparation, use of said compound and pharmaceutical composition comprising that compound |
| CN105203693A (en) * | 2015-09-18 | 2015-12-30 | 成都中医药大学 | Thin layer detection method for glabrousleaf pittosporum seeds |
| TWI612899B (en) * | 2017-04-27 | 2018-02-01 | 行政院農業委員會農業藥物毒物試驗所 | Camellia seed extract and biopesticides with the same |
Citations (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS617285A (en) | 1984-06-22 | 1986-01-13 | Kishimoto Sangyo Kk | Extraction of purified saponin |
| JPS61130232A (en) | 1984-11-29 | 1986-06-18 | Shiseido Co Ltd | Shikonin-containing composition |
| JPH0587400A (en) | 1991-09-27 | 1993-04-06 | Corona Kogyo Kk | Hot water circulating device for bath with boiler |
| WO2000038700A1 (en) | 1998-12-22 | 2000-07-06 | Janssen Pharmaceutica N.V. | Antiprotozoal saponins |
| US6197306B1 (en) | 1998-03-23 | 2001-03-06 | Dalmia Centre For Biotechnology | Herbal composition for the treatment of chronic obstructive pulmonary disease, bronchitis and respiratory disorders and a process for preparing the same |
| US6231859B1 (en) | 1996-12-02 | 2001-05-15 | Aquila Biopharmaceuticals, Inc. | Saponin adjuvant compositions |
| JP2002515430A (en) | 1998-05-19 | 2002-05-28 | リサーチ ディベロップメント ファンデーション | Triterpene composition and use thereof |
| JP2002247196A (en) | 2001-02-22 | 2002-08-30 | We'll Corporation:Kk | Connection aid service system for telephone station |
| CN1413724A (en) | 2001-08-31 | 2003-04-30 | 银泉科技有限公司 | Brain active effect matter for prevention and curing encephalopathy and developing intelligence and its preparation method |
| CA2541425A1 (en) | 2003-10-09 | 2005-04-28 | Pacific Arrow Limited | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US20050209445A1 (en) | 2004-11-30 | 2005-09-22 | Gokaraju Ganga R | Process for producing enriched fractions containing upto 100% of bacoside a and bacoside b from plant materials of bacopa species |
| US20050277601A1 (en) | 2003-10-09 | 2005-12-15 | Chan Pui-Kwong | Anticancer biangeloyl saponins |
| JP2006070018A (en) | 2004-08-05 | 2006-03-16 | Nippon Yakuyo Shokuhin Kenkyusho:Kk | Ingredient of tea blossom and its use |
| US20060183687A1 (en) | 1997-09-17 | 2006-08-17 | Suzanne Cory | Novel therapeutic molecules |
| US20070161580A1 (en) * | 2003-10-09 | 2007-07-12 | Chan Pui-Kwong | Anti-Tumor Compounds With Angeloyl Groups |
| US20070196517A1 (en) | 2005-07-13 | 2007-08-23 | Dictuc S.A. | Modified Saponin Molluscicide |
| US20070212329A1 (en) | 2003-10-13 | 2007-09-13 | Bruck Claudine E M | Vaccine Compositions Comprising An Interleukin 18 And Saponin Adjuvant System |
| US20070243269A1 (en) | 2006-04-18 | 2007-10-18 | Sartec Corporation | Saponin and preservative compositions and methods |
| US20070245470A1 (en) | 2004-08-04 | 2007-10-25 | Reckitt Benckiser Inc. | Dispensing Device |
| US20070249711A1 (en) | 2003-10-10 | 2007-10-25 | Wonrack Choi | Triterpene Compounds which are Effective on Improvement of Brain Function |
| US20070254847A1 (en) | 2004-09-30 | 2007-11-01 | Chengdu Di'ao Pharmaceutical Group Co., Ltd. | Pharmaceutical Composition Containing Steroidal Saponins, the Preparation Method and Use Thereof |
| WO2008028060A2 (en) | 2006-09-01 | 2008-03-06 | Pacific Arrow Limited | Anti-tumor compounds for inhibiting cancer growth |
| US20080058273A1 (en) | 2004-07-07 | 2008-03-06 | Baizhen Yang | Compound Extracted from Husk and Fruit Stem of Xanthoceras Sobifolia and Its Extracting Method and Use Thereof |
| US20080064762A1 (en) | 2004-07-13 | 2008-03-13 | Hendrik Fuchs | Composition Comprising a Pharmacologically Active Agent Coupled to a Target Cell Specific Component, and a Saponin |
| US20080096938A1 (en) | 2006-08-01 | 2008-04-24 | Praecis Pharmaceuticals Incorporated | Chemical compounds |
| US20080112925A1 (en) | 2000-06-22 | 2008-05-15 | Hancock Gerald E | Qs-21 and il-12 as an adjuvant combination |
| US20080119420A1 (en) | 2006-11-17 | 2008-05-22 | Liang Liu | Saponins Derived from Ilex Pubescens and Method of Purifying the same |
| WO2008133766A1 (en) | 2007-02-16 | 2008-11-06 | Pacific Arrow Limited | Blocking the migration or metastasis of cancer cells by affecting adhesion proteins and the uses of new compounds thereof |
| US7488753B2 (en) * | 2003-10-09 | 2009-02-10 | Pacific Arrow Limited | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| AU2009200988A1 (en) | 2003-10-09 | 2009-04-02 | Pacific Arrow Limited | Anti-tumor compounds for inhibiting cancer growth |
| US7514412B2 (en) * | 2003-10-09 | 2009-04-07 | Pacific Arrow Limited | Anticancer biangeloyl saponins |
| US7524824B2 (en) * | 2003-09-04 | 2009-04-28 | Pacific Arrow Limited | Composition comprising Xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US20100004190A1 (en) * | 2005-02-14 | 2010-01-07 | Pacific Arrow Limited | Blocking the migration or metastasis of cancer cells by affecting adhesion proteins and the uses of new compounds thereof |
| US7727561B2 (en) * | 2001-08-31 | 2010-06-01 | Pacific Arrow Limited | Composition comprising Xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US7780974B2 (en) | 2004-09-20 | 2010-08-24 | Research Development Foundation | Avicin coated stents |
| US20100317606A1 (en) * | 2005-04-27 | 2010-12-16 | Pacific Arrow Limited | Novel triterpenes for modulating gene expression and cell membrane, and as antiprotozoal agents |
| WO2011009032A1 (en) | 2009-07-16 | 2011-01-20 | Pacific Arrow Limited | Inhibiting the invasion and metastasis of cancer cells |
| NZ554037A (en) | 2004-09-07 | 2011-04-29 | Pacific Arrow Ltd | Pentacyclic anti-tumour compounds with angeloyl groups and optional sugar moieties |
| JP4880479B2 (en) | 2003-12-23 | 2012-02-22 | パシフィック アロー リミテッド | Composition comprising Xantoceros sorbifolia extract, compound isolated from said extract, method of preparing it, and method of use thereof |
| JP5087400B2 (en) | 2004-09-07 | 2012-12-05 | パシフィック アロー リミテッド | Antitumor compounds having an angeloyl group |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1052636C (en) | 1993-03-29 | 2000-05-24 | 中国科学院沈阳应用生态研究所 | Medicine for raising brains function and its preparing method |
| CN1092992A (en) | 1993-03-29 | 1994-10-05 | 中国科学院沈阳应用生态研究所 | Improve medicine of brain function and preparation method thereof |
| ATE380439T1 (en) | 1999-07-13 | 2007-12-15 | Sun Microsystems Inc | METHOD AND DEVICE FOR SELECTING MULTIPLE SENT IP DATA WHICH ARE TRANSMITTED WITHIN A BROADCAST STREAM |
| CN1350001A (en) | 2001-10-31 | 2002-05-22 | 杨柏珍 | Can be used for extracting xanthoceras sorbifolia Bunge total saponin, crude fat, crude protein, and sugar |
| CN100346805C (en) | 2001-10-31 | 2007-11-07 | 杨柏珍 | Xanthoceras sorbifolia bunge shell extract for treating cerebral hypofunction and anti-diabetic activity |
| US20060111310A1 (en) | 2003-10-09 | 2006-05-25 | Chan Pui-Kwong | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| US20060263458A1 (en) | 2003-10-09 | 2006-11-23 | Mak May S | Composition comprising growth factor of xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| JP2007108159A (en) * | 2005-09-15 | 2007-04-26 | Auto Network Gijutsu Kenkyusho:Kk | Driving support apparatus |
| US7679632B2 (en) * | 2006-05-31 | 2010-03-16 | Toshiba Tec Kabushiki Kaisha | Thermal printer and method of controlling the same |
| DE102006031975A1 (en) * | 2006-07-11 | 2008-01-17 | Bayer Cropscience Ag | Drug combinations with insecticidal and acaricidal properties |
-
2005
- 2005-11-28 US US11/289,142 patent/US7488753B2/en not_active Expired - Fee Related
-
2006
- 2006-04-27 EP EP06751723A patent/EP1876896A4/en not_active Withdrawn
- 2006-04-27 WO PCT/US2006/016158 patent/WO2006116656A2/en active Application Filing
-
2008
- 2008-12-29 US US12/344,682 patent/US8841265B2/en not_active Expired - Fee Related
Patent Citations (57)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS617285A (en) | 1984-06-22 | 1986-01-13 | Kishimoto Sangyo Kk | Extraction of purified saponin |
| JPS61130232A (en) | 1984-11-29 | 1986-06-18 | Shiseido Co Ltd | Shikonin-containing composition |
| JPH0587400A (en) | 1991-09-27 | 1993-04-06 | Corona Kogyo Kk | Hot water circulating device for bath with boiler |
| US6231859B1 (en) | 1996-12-02 | 2001-05-15 | Aquila Biopharmaceuticals, Inc. | Saponin adjuvant compositions |
| US20060183687A1 (en) | 1997-09-17 | 2006-08-17 | Suzanne Cory | Novel therapeutic molecules |
| US6197306B1 (en) | 1998-03-23 | 2001-03-06 | Dalmia Centre For Biotechnology | Herbal composition for the treatment of chronic obstructive pulmonary disease, bronchitis and respiratory disorders and a process for preparing the same |
| US6689398B2 (en) | 1998-05-19 | 2004-02-10 | Research Development Foundation | Triterpene compositions and methods for use thereof |
| JP2002515430A (en) | 1998-05-19 | 2002-05-28 | リサーチ ディベロップメント ファンデーション | Triterpene composition and use thereof |
| US6444233B1 (en) | 1998-05-19 | 2002-09-03 | Research Development Foundation | Triterpene compositions and methods for use thereof |
| US7105186B2 (en) | 1998-05-19 | 2006-09-12 | Research Development Foundation | Triterpene compositions and methods for use thereof |
| US7670632B2 (en) | 1998-05-19 | 2010-03-02 | Research Development Foundation | Triterpene compositions and methods for use thereof |
| US6746696B2 (en) | 1998-05-19 | 2004-06-08 | Research Development Foundation | Triterpene compositions and methods for use thereof |
| US6962720B2 (en) | 1998-05-19 | 2005-11-08 | Research Development Foundation | Triterpene compositions and methods for use thereof |
| US20040138151A1 (en) | 1998-12-22 | 2004-07-15 | Janssen Pharmaceutica N.V. | Antiprotozoal saponins |
| WO2000038700A1 (en) | 1998-12-22 | 2000-07-06 | Janssen Pharmaceutica N.V. | Antiprotozoal saponins |
| US20080112925A1 (en) | 2000-06-22 | 2008-05-15 | Hancock Gerald E | Qs-21 and il-12 as an adjuvant combination |
| JP2002247196A (en) | 2001-02-22 | 2002-08-30 | We'll Corporation:Kk | Connection aid service system for telephone station |
| TWI330087B (en) | 2001-08-31 | 2010-09-11 | Wang Yun | The use of wenguanguo (xanthoceras sorbifolia) saponins for the manufacture of medicament for activating brain function, mehods for preparing saponins and medical compositions thereof |
| US20100204169A1 (en) * | 2001-08-31 | 2010-08-12 | Pacific Arrow Limited | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US7727561B2 (en) * | 2001-08-31 | 2010-06-01 | Pacific Arrow Limited | Composition comprising Xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| CN1413724A (en) | 2001-08-31 | 2003-04-30 | 银泉科技有限公司 | Brain active effect matter for prevention and curing encephalopathy and developing intelligence and its preparation method |
| US8334269B2 (en) | 2003-09-04 | 2012-12-18 | Pacific Arrow Limited | Composition comprising Xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US7524824B2 (en) * | 2003-09-04 | 2009-04-28 | Pacific Arrow Limited | Composition comprising Xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| NZ546138A (en) | 2003-10-09 | 2009-12-24 | Pacific Arrow Ltd | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US7262285B2 (en) * | 2003-10-09 | 2007-08-28 | Pacific Arrow Limited | Anticancer biangeloyl saponins |
| JP4815558B2 (en) | 2003-10-09 | 2011-11-16 | パシフィック アロー リミテッド | COMPOSITION COMPRISING XANTHOCERASSORBIFOLIA EXTRACT, COMPOUND ISOLATED FROM THE EXTRACT, METHOD FOR PREPARING THEM, AND USE |
| AU2004281707A1 (en) | 2003-10-09 | 2005-04-28 | Pacific Arrow Limited | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US20090041877A1 (en) | 2003-10-09 | 2009-02-12 | Mak May Sung | Composition comprising growth factor of xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US20050277601A1 (en) | 2003-10-09 | 2005-12-15 | Chan Pui-Kwong | Anticancer biangeloyl saponins |
| CA2541425A1 (en) | 2003-10-09 | 2005-04-28 | Pacific Arrow Limited | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US20070161580A1 (en) * | 2003-10-09 | 2007-07-12 | Chan Pui-Kwong | Anti-Tumor Compounds With Angeloyl Groups |
| US7488753B2 (en) * | 2003-10-09 | 2009-02-10 | Pacific Arrow Limited | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof |
| US7514412B2 (en) * | 2003-10-09 | 2009-04-07 | Pacific Arrow Limited | Anticancer biangeloyl saponins |
| AU2009200988A1 (en) | 2003-10-09 | 2009-04-02 | Pacific Arrow Limited | Anti-tumor compounds for inhibiting cancer growth |
| KR101135824B1 (en) | 2003-10-09 | 2012-04-16 | 퍼시픽 애로우 리미티드 | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US20070249711A1 (en) | 2003-10-10 | 2007-10-25 | Wonrack Choi | Triterpene Compounds which are Effective on Improvement of Brain Function |
| US20070212329A1 (en) | 2003-10-13 | 2007-09-13 | Bruck Claudine E M | Vaccine Compositions Comprising An Interleukin 18 And Saponin Adjuvant System |
| JP4880479B2 (en) | 2003-12-23 | 2012-02-22 | パシフィック アロー リミテッド | Composition comprising Xantoceros sorbifolia extract, compound isolated from said extract, method of preparing it, and method of use thereof |
| US20080058273A1 (en) | 2004-07-07 | 2008-03-06 | Baizhen Yang | Compound Extracted from Husk and Fruit Stem of Xanthoceras Sobifolia and Its Extracting Method and Use Thereof |
| US20080064762A1 (en) | 2004-07-13 | 2008-03-13 | Hendrik Fuchs | Composition Comprising a Pharmacologically Active Agent Coupled to a Target Cell Specific Component, and a Saponin |
| US20070245470A1 (en) | 2004-08-04 | 2007-10-25 | Reckitt Benckiser Inc. | Dispensing Device |
| JP2006070018A (en) | 2004-08-05 | 2006-03-16 | Nippon Yakuyo Shokuhin Kenkyusho:Kk | Ingredient of tea blossom and its use |
| JP5087400B2 (en) | 2004-09-07 | 2012-12-05 | パシフィック アロー リミテッド | Antitumor compounds having an angeloyl group |
| NZ554037A (en) | 2004-09-07 | 2011-04-29 | Pacific Arrow Ltd | Pentacyclic anti-tumour compounds with angeloyl groups and optional sugar moieties |
| US7780974B2 (en) | 2004-09-20 | 2010-08-24 | Research Development Foundation | Avicin coated stents |
| US20070254847A1 (en) | 2004-09-30 | 2007-11-01 | Chengdu Di'ao Pharmaceutical Group Co., Ltd. | Pharmaceutical Composition Containing Steroidal Saponins, the Preparation Method and Use Thereof |
| US20050209445A1 (en) | 2004-11-30 | 2005-09-22 | Gokaraju Ganga R | Process for producing enriched fractions containing upto 100% of bacoside a and bacoside b from plant materials of bacopa species |
| US20100004190A1 (en) * | 2005-02-14 | 2010-01-07 | Pacific Arrow Limited | Blocking the migration or metastasis of cancer cells by affecting adhesion proteins and the uses of new compounds thereof |
| US20100317606A1 (en) * | 2005-04-27 | 2010-12-16 | Pacific Arrow Limited | Novel triterpenes for modulating gene expression and cell membrane, and as antiprotozoal agents |
| US8586719B2 (en) | 2005-04-27 | 2013-11-19 | Pacific Arrow Limited | Triterpenes for modulating gene expression and cell membrane, and as antiprotozoal agents |
| US20070196517A1 (en) | 2005-07-13 | 2007-08-23 | Dictuc S.A. | Modified Saponin Molluscicide |
| US20070243269A1 (en) | 2006-04-18 | 2007-10-18 | Sartec Corporation | Saponin and preservative compositions and methods |
| US20080096938A1 (en) | 2006-08-01 | 2008-04-24 | Praecis Pharmaceuticals Incorporated | Chemical compounds |
| WO2008028060A2 (en) | 2006-09-01 | 2008-03-06 | Pacific Arrow Limited | Anti-tumor compounds for inhibiting cancer growth |
| US20080119420A1 (en) | 2006-11-17 | 2008-05-22 | Liang Liu | Saponins Derived from Ilex Pubescens and Method of Purifying the same |
| WO2008133766A1 (en) | 2007-02-16 | 2008-11-06 | Pacific Arrow Limited | Blocking the migration or metastasis of cancer cells by affecting adhesion proteins and the uses of new compounds thereof |
| WO2011009032A1 (en) | 2009-07-16 | 2011-01-20 | Pacific Arrow Limited | Inhibiting the invasion and metastasis of cancer cells |
Non-Patent Citations (167)
| Title |
|---|
| "Cancer". Wikipedia, The Free Encyclopedia. Wikimedia Foundation, Inc. Jul. 22, 2004. Web Aug. 11, 2011. |
| 2006 Chemical Abstracts Catalog, published 2006 by Chemical Abstracts Service, p. 52. |
| Ahmad V U et al., "The Sapogenins from Dodonaea viscosa", Fitoterapia, vol. 58, No. 5, 1987, pp. 361-362. |
| Akihisa et al, 2006, "Cancer Chemopreventive Effects and Cytotoxic Activities of the Triterpene Acids from the Resin of Boswellia carteri", Biological & Pharmaceutical Bulletin, vol. 29(9):1976-1979. |
| Apers Sandra et al., "Antiviral, haemolytic and moliuscicidal activities of triterpenoid saponins. from Maesa lanceolata: Establishment of structure-activity relationships", Planta Medica, vol. 67, No. 6, Aug. 2001, pp. 528-532. |
| Australian Letters Patent No. 2002348988, Nov. 8, 2007, Wang, Yun, "Composition comprising Wenguanguo Extracts, Methods for Preparing Same and Uses Thereof". |
| Australian Office Action for Australian Patent No. 2004281707, Feb. 19, 2010, Pacific Arrow Limited. |
| Australian Office Action, Apr. 16, 2013 for Pacific Arrow Limited, Australian App'l No. 2009226063, filed Sep. 6, 2010. |
| Australian Office Action, Feb. 6, 2012 for Pacific Arrow Limited, Australian App'l No. 2005282437, filed Mar. 19, 2007. |
| Australian Office Action, Feb. 6, 2012 for Pacific Arrow Limited, Australian App'l No. 2009200988, filed Mar. 10, 2009. |
| Australian Office Action, Feb. 6, 2012 for Pacific Arrow Limited, Australian App'l No. 2009208069, filed Aug. 7, 2009. |
| Australian Office Action, Jun. 21, 2012 for Pacific Arrow Limited, Australian App'l No. 2008244648, filed Aug. 21, 2009. |
| Australian Office Action, Mar. 18, 2011 for Pacific Arrow Limited et al., Australian Patent Application No. 2004281707, filed Oct. 8, 2004. |
| Bang. et al., 2007. "Facile Synthesis of Trisaccharide Moiety Corresponding to Antitumor Activity in Triterpenoid Saponins Isolated from Pullsatilla Roots", Chemical & Pharmaceutical Bulletin, vol. 55(12): 1734-1739. |
| Canadian Notice of Allowance, Oct. 5, 2011, for Pacific Arrow Limited et al, Canadian App'l No. 2,541,425, filed Oct. 8, 2004. |
| Canadian Office Action for Canadian Application No. 2,451,740 , May 26, 2000, Fountain Silver Limited, "Composition Comprising Wenguanoguo Extracts, Methods for Perparing Same and Uses Thereof". |
| Canadian Office Action for Canadian Application No. 2,451,740 , Nov. 7, 2008, Fountain Silver Limited, "Composition Comprising Wenguanoguo Extracts, Methods for Perparing Same and Uses Thereof". |
| Canadian Office Action, Feb. 26, 2013, for Pacific Arrow Limited, Canadian App'l No. 2,579,231, filed Mar. 6, 2007. |
| Canadian Office Action, Jan. 31, 2012, for Pacific Arrow Limited, Canadian Application No. 2,579,231, filed Mar. 6, 2007. |
| Canadian Office Action, Jul. 5, 2012, for Fountain Silver Limited et al., Canadian App'l No. 2,451,740, filed Dec. 18, 2003. |
| Canadian Office Action, May 21, 2013, for Fountain Silver Limited et al., Canadian App'l No. 2,451,740, filed Dec. 18, 2003. |
| Canadian Office Action, Sep. 8, 2011, for Fountain Silver Limited et al., Canadian App'l No. 2,451,740, filed Dec. 18, 2003. |
| Chang et al, 2007, "Biologically Active Triterpenoid Saponins from Ardisia japonica", Journal of Natural Products, vol. 70(2): 179-187. |
| Chemical Abstracts Service, Columbus, Ohio, US, Germonprez N. et al., "Antileishmanial saponins from Maesa", Tap Chi Hoa Hoc, 41(spec.), 125-130, (2003). |
| Cheng, et al. "Two new sterols in the husk of Xanthoceras sorbifolia." Chinese Traditional and Herbal Drugs (2001), 32(3), 199-201. |
| China Office Action for Pacific Arrow Limited, et al., China App'l No. 2004800367617, Dated Jan. 15, 2010. |
| Chinese Notice of Allowance, Feb. 1, 2011, for Pacific Arrow Limited, Chinese app'l No. 200580037524.7, filed Apr. 30, 2007. |
| Chinese Office Action for Wang, Yun, Chinese Publication No. CN 1236792C, filed Jan. 18, 2006, Dated Aug. 27, 2004. |
| Chinese Office Action for Wang, Yun, Chinese Publication No. CN 1236792C, filed Jan. 18, 2006, Dated May 27, 2005. |
| Chinese Office Action, Apr. 21, 2010 for Pacific Arrow Limited, Chinese application No. 2005800375247, filed Jul. 26, 2007. |
| Chinese Office Action, Apr. 9, 2012, for Pacific Arrow Limited, Chinese App'l No. 200780040744.4, filed Apr. 30, 2009. |
| Chinese Office Action, Jun. 14, 2011, for Pacific Arrow Limited, Chinese App'l No. 200480036761.7, filed Jun. 8, 2006. |
| Chinese Office Action, Jun. 3, 2011 for Pacific Arrow Limited, Chinese application No. 200880012065.0, filed Oct. 14, 2009. |
| Chinese Office Action, Mar. 23, 2011, for Pacific Arrow Limited, Chinese App'l No. 200780040744.4, filed Apr. 30, 2009. |
| Chinese Office Action, Mar. 27, 2009 for Pacific Arrow Limited, Chinese application No. 2005800375247, filed Jul. 26, 2007. |
| Chinese Office Action, Oct. 28, 2011, for Pacific Arrow Limited, Chinese App'l No. 200480038698.0, filed Jun. 23, 2006. |
| Chinese Office Action, Sep. 28, 2011, for Pacific Arrow Limited, Chinese App'l No. 200480036761.7, filed Jun. 8, 2006. |
| D'Acquarica et al., "Isolation and structure elucidation of four new triterpenoid estersaponins from fruits of Pittosporum tobira AIT" Tetrahedron (2002) vol. 58 pp. 10127-10136. * |
| Dan Peer, et al. "Nanocarriers as an emerging platform for cancer therapy." Nature Publishing Group (2007), 751-760. |
| Declaration of Interference #105,982, Dec. 9, 2013, Pui-Kwong Chan, May Sung Mak and Yun Wang (Junior Party) v. Baizhen Yang, Songjiang Wang and Congfu Zhao (Senior Party). |
| Dizes C et al., "Harpuloside a triterpenoid saponin from Harpullia ramiflora", Phytochemistry, Pergamon Press, GB, vol. 48, No. 7, Aug. 1, 1998, pp. 1229-1232. |
| European Office Communication for Mak May Sun et al., European App'l No. EP 0581026.3-2123, Dated Dec, 29, 2009. |
| European Office Communication for Mak May Sung, et al., European App'l No. EP 05810263.3-2123, Dated Apr. 19, 2010. |
| European Office Communication for Pacific Arrow Limited, et al., European App'l No. EP 04809909.7-2123, Dated Apr. 19, 2010. |
| European Office Communication for Pacific Arrow Limited, et al., European App'l No. EP 04815530.3-2123, Dated Apr. 19, 2010. |
| European Office Communication for Pacific Arrow Limited, et al., European App'l No. EP 06751723.5-2123, Dated Jan. 15, 2010. |
| European Office Communication for Pacific Arrow Limited, et al., European App'l No. EP 07841638.5-2123, Dated Apr. 19, 2010. |
| European Office Communication, Apr. 26, 2012 for Pacific Arrow Limited, European App'l No. EP 09721583.4-2123, filed Sep. 7, 2010. |
| European Office Communication, Feb. 13, 2012 for Pacific Arrow Limited, European App'l No. EP 05810263.3-2123, filed Mar. 30, 2007. |
| European Office Communication, Feb. 13, 2012, for Pacific Arrow Limited, European App'l No. EP 04815530.3-2123, filed Jul. 19, 2006. |
| European Office Communication, Feb. 13, 2012, for Pacific Arrow Limited, European App'l No. EP 07841638.5-2123, filed Mar. 27, 2009. |
| European Office Communication, Jun. 26, 2013, for Pacific Arrow Limited, European App'l No. EP 04815530.3-1464, filed Jul. 19, 2006. |
| European Office Communication, Jun. 4, 2012 for Pacific Arrow Limited, European App'l No. EP 02781502.6-2112, filed Feb. 25, 2004. |
| European Office Communication, Mar. 5, 2012 for Pacific Arrow Limited, European App'l No. EP 04809909.7-2123, filed Mar. 27, 2006. |
| European Office Communication, May 13, 2013 for Pacific Arrow Limited, European App'l No. EP 10800596.8-1462, filed Mar. 30, 2012. |
| Fujioka, et al., 2006, "Antiproliferative Constituents from Umbelliferae Plants. New Triterpenoid Glycosides from the Fruits of Bupleurum rotundifolium", Chemical & Pharmaceutical Bulletin, vol. 54 (12):1694-1704. |
| Germonprez N. et al. "In vitro and in vivo anti-leishmanial activity of triterpenoid saponins isolated from Maesa balansae and some chemical derivatives", Journal of Medicinal Chemistry, vol. 48 No. 1 (Jan. 13, 2005), p. 32-37. |
| Japan Final Office Action, Feb. 23, 2009, for Fountain Silver Limited et al., Japan App'l No. 2003-522442, filed Feb. 5, 2004. |
| Japan Office Action for Japan Patent Application No. 2003-522442, Nov. 4, 2008, International No. PCT/IB02/04750, filed Aug. 28, 2002, Fountain Silver Limited, "Composition Comprising Wenguanoguo Extracts, Methods for Perparing Same and Uses Thereof". |
| Japan Office Action, Feb. 2, 2011, for Pacific Arrow Limited et al., Japan app'l No. 2006-534419, filed Mar. 22, 2006. |
| Japan Office Action, Jan. 14, 2011, for Pacific Arrow Limited et al., Japan App'l No. 2006-534419, filed Mar. 22, 2006. |
| Japan Office Action, Jun. 3, 2011 for Pacific Arrow Limited et al., Japan App'l No. 2006-534419, filed Mar. 22, 2006. |
| Japan Office Action, Mar. 18, 2011, for Pacific Arrow Limited et al., Japan app'l No. 2006-547422, filed Jun. 16, 2006. |
| Japanese Notice of Allowance, Nov. 15, 2011, for Pacific Arrow Limited, Japanese app'l No. 2006-547422, filed Jun. 16, 2006. |
| Japanese Office Action, May 8, 2012, for Pacific Arrow Limited, Japanese app'l No. 2007-530484, filed Mar. 2, 2007. |
| Japanese Office Action, Nov. 21, 2011, for Pacific Arrow Limited, Japanese app'l No. 2007-530484, filed Mar. 2, 2007. |
| Korean Office Action for Korean Application No. 10-2004-7002889, Nov. 21, 2008, International Application No. PCT/IB02/04750, filed Aug. 28, 2002, Fountain Silver Limited, "Composition Comprising Wenguanoguo Extracts, Methods for Perparing Same and Uses Thereof". |
| Korean Office Action, Jun. 22, 2012 for Pacific Arrow Limited, Korean App'l No. 10-2007-7007902, filed Apr. 6, 2007 (w/English Translation). |
| Korean Office Action, Jun. 3, 2011 for Pacific Arrow Limited et al., Korean App'l No. 10-2006-7008896, filed May 8, 2006. |
| Liang, et al., 2006, "Triterpenoid Saponins from Lysimachia davurica", Chemical & Pharmaceutical Bulletin, vol. 54 (10):1380-1383. |
| Maes, et al. "In vitro and in vivo activities of a triterpenoid saponin extract (px-6518) from the plant Maesa balansae against visceral leishmania species." Antimicrobial agents and chemotherapy, Jan. 2004, p. 130-136. |
| Mahato et al. Tetrahedron 1991 (47) 5215-5230. |
| Murakami, et al. "New hypoglycemic constituents in "gymnemic acid" from gymnema sylvestre." Chem, Pharm. Bull. 44(2) 469-471 (1996 ). |
| Na, et al. "Protein tyroshine phosphatase 1B inhibitory activity of triterpenes isolated from Astilbe koreana." Bioorg Med Chem Lett. Jun. 15, 2006:16(12): 3273-6. |
| New Zealand Letters Patent No. 530449, Oct. 11, 2007, Wang, Yun, "Composition comprising Wenguanguo Extracts, Methods for Preparing Same and Uses Thereof". |
| New Zealand Letters Patent No. 546138, Apr. 8, 2010, Pacific Arrow Limited, et al. "Composition Comprising Xanthoceras sorbifolia Extracts, Compounds Isolated From Same, Methods for Perparing Same and Uses Thereof". |
| New Zealand Office Action, Apr. 12, 2011 for Pacific Arrow Limited et al., New Zealand App'l No. 554037, filed Mar. 19, 2007. |
| New Zealand Office Action, Aug. 12, 2009, New Zealand Application No. 546138, filed Mar. 22, 2007. |
| New Zealand Office Action, Jan. 11, 2012, for Pacific Arrow Limited, New Zealand App'l No. 579194, filed Aug. 21, 2009. |
| New Zealand Office Action, Mar. 26, 2012, for Pacific Arrow Limited, New Zealand App'l No. 598934, filed Mar. 21, 2012. |
| New Zealand Office Action, Mar. 7, 2011 for Pacific Arrow Limited et al., New Zealand App'l No. 587973, filed Sep. 14, 2010. |
| New Zealand Office Action, May 8, 2009, for New Zealand Application No. 554037, filed Sep. 7, 2005. |
| New Zealand Office Action, Sep. 22, 2009, New Zealand Application No. 546138, filed Mar. 27, 2006. |
| New Zealand Office Action, Sep. 24, 2010, for Pacific Arrow Limited, New Zealand App'l No. 579194, filed Aug. 21, 2009. |
| Notice of Acceptance for Pacific Arrow Limited et al., Australian Patent App'l No. 2004281707, filed Mar. 23, 2006, Dated May 26, 2011. |
| Notice of Allowability for Chan et al., U.S. Appl. No. 10/906,303, filed Feb. 14, 2005, dated Nov. 26, 2008. |
| Notice of Allowability for Chan et al., U.S. Appl. No. 11/117,745, filed Apr. 27, 2005, dated Dec. 2, 2008. |
| Notice of Allowability for Chan et al., U.S. Appl. No. 11/683,198, filed Mar. 7, 2007, Dated May 5, 2011. |
| Notice of Allowability for Chan et al., U.S. Appl. No. 12/541,713, filed Aug. 14, 2009, Dated Sep. 12, 2012. |
| Notice of Allowability for Chan et al., U.S. Appl. No. 14/020,099, filed Sep. 6, 2013, Dated Mar. 3, 2014. |
| Notice of Allowability, Aug. 15, 2012, for Chan et al., U.S. Appl. No. 12/392,795, filed Feb. 25, 2009. |
| Oda, K. et al., "Adjuvant and Haemolytic Activities of 47 Saponins Derived from Medicinal and Food Plants" Biol. Chem. (2000) vol. 381, pp. 67-74. |
| Office Action for Mak, et al., U.S. Appl. No. 11/412,659, filed Apr. 27, 2006, Dated Feb. 20, 2008. |
| Ohtsuki et al. "Acylated triterpenoid saponins from Schima noronhae and their cell growth inhibitory activity", Journal of Natural Products, vol. 71, No. 5, Mar. 20, 2008, pp. 918-921, XP002694762. |
| PCT International Preliminary Report on Patentability for Pacific Arrow Limited et al., Intl App'l No. PCT/US2004/033359, filed Oct. 8, 2004, Dated Apr. 11, 2006. |
| PCT International Preliminary Report on Patentability, Jun. 25, 2013, for Pacific Arrow Limited, Int'l App'l No. PCT/US2011/044233, filed Jul. 15, 2011. |
| PCT International Preliminary Report on Patentability, Mar. 12, 2009, International App'l No. PCT/US2007/077273, filed Aug. 30, 2007. |
| PCT International Search Report Dec. 6, 2011, for Pacific Arrow Limited, Int'l App'l No. PCT/US2011/044233, filed Jul. 15, 2011. |
| PCT International Search Report for Pacific Arrow Limited et al,, International App'l No. PCT/US07/77273, filed Aug. 30, 2007, Dated Aug. 4, 2008. |
| PCT International Search Report for PCT/US10/42240, filed Jul. 16, 2010 for Pacific Arrow Limited et al., dated Sep. 2, 2010. |
| PCT International Search Report for PCT/US2009/034115, filed Feb. 13, 2009 for Pacific Arrow Limited et al., dated Jun. 2, 2009. |
| PCT International Search Report issued on Jul. 7, 2008 for Pacific Arrow Limited et al., Int'l App'l No. PCT/US2008/002086. |
| PCT International Search Report, Oct. 2, 2012, for Pacific Arrow Limited, Int'l App'l No. PCT/US12/46716, filed Jul. 13, 2012. |
| PCT Notification Concerning Transmittal of International Preliminary Report on Patentability, Jul. 4, 2013, for Pacific Arrow Limited, Int'l App'l No. PCT/US2011/044233, filed Jul. 15, 2011. |
| PCT Notification of Transmittal of International Preliminary Examination Report for PCT/IB02/04750, filed Aug. 28, 2002 for Fountain Silver Limited et al., dated Jun. 3, 2003. |
| PCT Notification of Transmittal of the International Search Report and the Written Opinion of the International Searching Authority, or the Declaration for PCT/US09/34115, filed Feb. 13, 2009 for Pacific Arrow Limited et al., dated Jun. 2, 2009. |
| PCT Preliminary Report on Patentability for PCT/US2009/034115, filed Feb. 13, 2009 for Pacific Arrow Limited et al., dated Aug. 26, 2010. |
| PCT Written Opinion of the International Searching Authority for Pacific Arrow Limited et al., International App'l No. PCT/US07/77273, filed Aug. 30, 2007, Dated Aug. 4, 2008. |
| PCT Written Opinion of the International Searching Authority for PCT/US09/34115, filed Feb. 13, 2009 for Pacific Arrow Limited at al., dated Jun. 2, 2009. |
| PCT Written Opinion of the International Searching Authority for PCT/US10/42220, filed Jul. 16, 2010 for Pacific Arrow Limited et al., dated Sep. 2, 2010. |
| PCT Written Opinion of the International Searching Authority issued on Jul. 7, 2008 for Pacific Arrow Limited et al., Int'l App'l No. PCT/US2008/002086. |
| PCT Written Opinion of the International Searching Authority, Aug. 4, 2008, International App'l No. PCT/US07/77273, filed Aug. 30, 2007. |
| PCT Written Opinion of the International Searching Authority, Dec. 6, 2011, for Pacific Arrow Limited, Int'l App'l No. PCT/US2011/044233, filed Jul. 15, 2011. |
| PCT Written Opinion of the International Searching Authority, Jun. 2, 2009, International App'l No. PCT/US09/34115, filed Feb. 13, 2009. |
| PCT Written Opinion of the International Searching Authority, Oct. 2, 2012, for Pacific Arrow Limited, Int'l App'l No. PCT/US12/46716, filed Jul. 13, 2012. |
| Puiffe, et al., 2007, "Characterization of Ovarian Cancer Ascites on Cell Invasion, Proliferation, Spheroid Formation, and Gene Expression in an In Vitro Model of Epithelial Ovarian Cancer" Neoplasia, vol. 9(10):820-829. |
| Rabi, et al., 2007; "Novel triterpenoid 25-hydroxy-3-oxoolean-12-en-28-oic acid induces growth arrest and apoptosis in breast cancer cells", Breast Cancer Research & Treatment, vol. 101:27-36. |
| Ricciardelli, et al., 2006, "Extracellular Matrix of Ovarian Tumors", Seminars in Reproductive Medicine, vol. 24(4): 270-282. |
| Sharma et al. "Lanthadenes and their esters as potential antitumor agents", Journal of Natural Products, vol. 71, No. 7, Jun. 14, 2008, pp. 1222-1227, XP002694763. |
| Sheng-Xiang et al. Phytochemistry (1993), 34(5), 1385-1387. |
| Siroti, C., "Aescin: Pharmacology, Pharmacokinetic Profile" Pharmacological Research(2001) vol. 44, No. 3, pp. 183-193. |
| Sporn, et al., 2007, "Platforms and Networks in Triterpenoid Pharmacology", Drug Development Research, vol. 68:174-182 (2007). |
| Supplementary European Search Report issued on Oct. 13, 2009 for Pacific Arrow Limited et al., European Patent Application No. 04809909.7. |
| Supplementary European Search Report issued on Oct. 13, 2009 for Pacific Arrow Limited et al., European Patent Application No. 04815530.3. |
| Supplementary European Search Report issued on Oct. 22, 2009 for Mak et al., European Patent Application No. 05810263.3, PCT/US2005031900. |
| Taiwan Office Action for Wang, Yun, Taiwan Application No. 091119471, filed Aug. 28, 2002, Dated Apr. 26, 2005. |
| Taiwan Office Action for Wang, Yun, Taiwan Application No. 091119471, filed Aug. 28, 2002, Dated Sep. 14, 2004. |
| Taiwan Office Action, Jan. 18, 2012 for Pacific Arrow Limited, Taiwan App'l No. 094130519, filed Sep. 6, 2005. |
| Taiwan Office Action, Mar. 12, 2010 for Pacific Arrow Limited, Taiwan App'l No. 093140030, filed Dec. 22, 2004. |
| Taiwan Office Action, Mar. 3, 2011 for Pacific Arrow Limited, Taiwan App'l No. 093140030, filed Dec. 22, 2004. |
| Talmadge, James E., 2008, "Follistatin as an Inhibitor of Experimental Metastasis", Clinical Cancer Research, vol. 14(3) 624-626. |
| U.S. Appl. No. 12/392,795, Mar. 20, 2009, Chan et al. |
| U.S. Appl. No. 12/856,322, Aug. 13, 2010, Chan et al. |
| U.S. Appl. No. 60/795,417, Apr. 27, 2006, Mak et al. |
| U.S. Appl. No. 60/890,380, Feb. 16, 2007, Chan et al. |
| U.S. Appl. No. 60/947,705. Jul. 3, 2007, Chan et al. |
| U.S. Notice of Allowance , Jan. 30, 2012, for Chan et al., U.S. Appl. No. 11/683,198, filed Mar. 7, 2007. |
| U.S. Office Action , Apr. 11, 2013, for Chan et al., U.S. Appl. No. 11/683,198, filed Mar. 7, 2007. |
| U.S. Office Action for Chan, et al., U.S. Appl. No. 10/906,303, filed Feb. 14, 2005, Dated Feb. 12, 2007. |
| U.S. Office Action for Chan, et al., U.S. Appl. No. 10/906,303, filed Feb. 14, 2005, Dated Jul. 27, 2007. |
| U.S. Office Action for Chan, et al., U.S. Appl. No. 11/117,760, filed Apr. 27, 2005, Dated Mar. 18, 2009. |
| U.S. Office Action for Chan, et al., U.S. Appl. No. 11/683,198, filed Mar. 7, 2007, Dated Feb. 18, 2010. |
| U.S. Office Action for Chan, et al., U.S. Appl. No. 11/683,198, filed Mar. 7, 2007, Dated May 1, 2009. |
| U.S. Office Action for Chan, et al., U.S. Appl. No. 11/683,198, filed Mar. 7, 2007, Dated Sep. 10, 2008. |
| U.S. Office Action for Mak, et al., U.S. Appl. No. 11/412,659, filed Apr. 27, 2006, Dated Mar. 8, 2007. |
| U.S. Office Action for Mak, et al., U.S. Appl. No. 12/195,112, filed Aug. 20, 2008, Dated Feb. 18, 2010. |
| U.S. Office Action for Mak, et al., U.S. Appl. No. 12/195,112, filed Aug. 20, 2008, Dated May 19, 2009. |
| U.S. Office Action, Apr. 17, 2012, for Chan et al., U.S. Appl. No. 12/392,795, filed Feb. 25, 2009. |
| U.S. Office Action, Dec. 21, 2011, for Chan et al., U.S. Appl. No. 12/541,713, filed Aug. 14, 2009. |
| U.S. Office Action, Dec. 28, 2011, for Chan et al., U.S. Appl. No. 12/714,598, filed Mar. 1, 2010. |
| U.S. Office Action, Feb. 1, 2013, for Chan et al., U.S. Appl. No. 13/259,480, filed Sep. 23, 2011. |
| U.S. Office Action, Jun. 26, 2012, for Chan et al., U.S. Appl. No. 12/541,713, filed Aug. 14, 2009. |
| U.S. Office Action, Jun. 6, 2013, for Chan et al., U.S. Appl. No. 13/259,480, filed Sep. 23, 2011. |
| U.S. Office Action, Mar. 21, 2013, for Chan et al., U.S. Appl. No. 12/856,322, filed Aug. 13, 2010. |
| U.S. Office Action, May 20, 2011, for Chan et al., U.S. Appl. No. 12/392,795, filed Feb, 25, 2009. |
| U.S. Office Action, Nov. 21, 2013, for Chan et al., U.S. Appl. No. 13/718,575, filed Dec. 18, 2012. |
| U.S. Office Action, Oct. 15, 2012, for Chan et al., U.S. Appl. No. 13/259,480, filed Sep. 23, 2011. |
| U.S. Office Communication for Chan, et al., U.S. Appl. No. 11/117,760, filed Apr. 27, 2005, Dated Feb. 8, 2008. |
| US Office Action, May 12, 2011, for Chan et al., U.S. Appl. No. 12/714,598, filed Mar. 1, 2010. |
| Ushijima et al, 2008, "Triterpene Glycosides from the Roots of Codonopsis lanceolata", Chemical & Pharmaceutical Bulletin, vol. 56(3) 308-314. |
| Voutquenne et al. "Structure-Activity Relationships of Haemolytic Saponins" Pharmaceutical Biology (2002), vol. 40, No. 4, pp. 253-262. |
| Wang et al., 2008, "Bioactive Triterpene Saponins from the Roots of Phytolacca Americana", Journal of Natural Products, vol. 71(1): 35-40. |
| Wei, et al., 2004, "Anti-inflammatory Triterpenoid Saponins from the Seeds of Aesculus chinensis", Chemical & Pharmaceutical Bulletin, vol. 52(10): 1246-1248. |
| Yadava et al., 2008, "New antibacterial triterpenoid saponin from Lactuca scariola", Fitoterapia, vol. 1:1-5. |
| Yang et al. "The Influence of aquaporin-1 and microvessel density on ovarian carcinogenesis and ascites formation", International Journal of Gynecological Cancer, vol. 16, No. S1, Feb. 1, 2006, pp. 400-405. |
| Zhou, et al. "The first naturally occurring tie2 kinase inhibitor." Org Lett. Dec. 13, 2001;3(25): 4047-9. |
| Zhu et al, "Preliminary test of chemical constituents of wenguanguo and its multipurpose utilization", Research of Land and Natural Resources (1): 69-71, 1997 Arda, et al. "Saniculoside N from Sanicula europaea L." Journal of Natural Products (1997), 60(11), 1170-1173. |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130190261A1 (en) * | 2003-10-09 | 2013-07-25 | Pacific Arrow Limited | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof |
| US20130338089A1 (en) * | 2004-09-07 | 2013-12-19 | Pacific Arrow Limited | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US9382285B2 (en) * | 2004-09-07 | 2016-07-05 | Pacific Arrow Limited | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US10213451B2 (en) * | 2004-09-07 | 2019-02-26 | Pacific Arrow Limited | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US20190256543A1 (en) * | 2004-09-07 | 2019-08-22 | Pacific Arrow Limited | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US11046724B2 (en) * | 2004-09-07 | 2021-06-29 | Pacific Arrow Limited | Methods and compounds for modulating the secretion or expression of adhesion proteins or angiopoietins of cells |
| US9434677B2 (en) | 2009-07-16 | 2016-09-06 | Pacific Arrow Limited | Natural and synthetic compounds for treating cancer and other diseases |
| US9499577B2 (en) | 2009-07-16 | 2016-11-22 | Pacific Arrow Limited | Natural and synthetic compounds for treating cancer and other diseases |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1876896A4 (en) | 2009-11-25 |
| WO2006116656A3 (en) | 2007-02-15 |
| US7488753B2 (en) | 2009-02-10 |
| WO2006116656A2 (en) | 2006-11-02 |
| EP1876896A2 (en) | 2008-01-16 |
| US20090156515A1 (en) | 2009-06-18 |
| US20060122129A1 (en) | 2006-06-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8841265B2 (en) | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof | |
| US20060111310A1 (en) | Composition comprising triterpene saponins and compounds with angeloyl functional group, methods for preparing same and uses thereof | |
| JP5087400B2 (en) | Antitumor compounds having an angeloyl group | |
| AU2009200988B2 (en) | Anti-tumor compounds for inhibiting cancer growth | |
| US8614197B2 (en) | Anti-tumor compounds with angeloyl groups | |
| US7262285B2 (en) | Anticancer biangeloyl saponins | |
| US7514412B2 (en) | Anticancer biangeloyl saponins | |
| Zhao et al. | Novel dammarane-type sapogenins from Panax ginseng berry and their biological activities | |
| US20130190261A1 (en) | Composition comprising xanthoceras sorbifolia extracts, compounds isolated from same, methods for preparing same and uses thereof | |
| WO2008028060A2 (en) | Anti-tumor compounds for inhibiting cancer growth | |
| Hamed et al. | Oleanane glycosides from the roots of Alhagi maurorum | |
| Yue et al. | Cucurbitane triterpenoids from the fruit of Momordica charantia L. and their anti-hepatic fibrosis and anti-hepatoma activities | |
| Yadav et al. | Antioxidant furofuran lignans from Premna integrifolia | |
| Akter et al. | A new cytotoxic steroidal glycoalkaloid from the methanol extract of Blumea lacera leaves | |
| Simo et al. | Benjaminamide: A new ceramide and other compounds from the twigs of Ficus benjamina (Moraceae) | |
| Fawzy et al. | Bioactivity and chemical characterization of Acalypha fruticosa Forssk. growing in Saudi Arabia | |
| Mai et al. | Steroidal saponins from Datura metel | |
| El-Mesallamy et al. | Reno-protective effect of methanolic extract of Stevia rebaudiana Bertoni and bioactive phenolic compounds in type-1-diabetes | |
| Liu et al. | Chemical constituents of Anchusa italica Retz. and their protective effects on cardiomyocytes injured by hypoxia/reoxygenation | |
| Lee et al. | The effect of curculigoside on the expression of matrix metalloproteinase-1 in cultured human skin fibroblasts | |
| Ahmed et al. | Triterpenoid antioxidant agents found in Holoptelea integrifolia (Roxb) Planch | |
| Aliyu et al. | Phytochemical Analysis and Isolation of Secondary Metabolites from Persea Declinata | |
| Al-Mahweety et al. | PHYTOCHEMICAL PURIFICATION OF ACTIVE CONSTITUENTS ISOLATED FROM ROOT OF THE MEDICINAL HERB, CARALLUMA QUADRANGULA | |
| Van Mai Do et al. | Polysciosides J and K, two new oleanane-type triterpenoid saponins from the leaves of Polyscias fruticosa (L.) harms. cultivating in An Giang Province, Viet Nam | |
| Jin et al. | Cytotoxic anthraquinones and stilbenes from Reynoutria sachalinensis (Fr. Schm.) Nakai |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: PACIFIC ARROW LIMITED, HONG KONG Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:MAK, MAY SUNG;WANG, YUN;REEL/FRAME:022341/0051;SIGNING DATES FROM 20051225 TO 20060201 Owner name: BAYLOR COLLEGE OF MEDICINE, TEXAS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:CHAN, PUI-KWONG;REEL/FRAME:022341/0313 Effective date: 20051221 Owner name: PACIFIC ARROW LIMITED, HONG KONG Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:BAYLOR COLLEGE OF MEDICINE;REEL/FRAME:022341/0374 Effective date: 20051221 Owner name: PACIFIC ARROW LIMITED, HONG KONG Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:MAK, MAY SUNG;WANG, YUN;SIGNING DATES FROM 20051225 TO 20060201;REEL/FRAME:022341/0051 |
|
| AS | Assignment |
Owner name: PACIFIC ARROW LIMITED, CHINA Free format text: CHANGE OF ASSIGNEE ADDRESS;ASSIGNOR:PACIFIC ARROW LIMITED;REEL/FRAME:032845/0074 Effective date: 20140424 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YR, SMALL ENTITY (ORIGINAL EVENT CODE: M2551) Year of fee payment: 4 |
|
| FEPP | Fee payment procedure |
Free format text: MAINTENANCE FEE REMINDER MAILED (ORIGINAL EVENT CODE: REM.); ENTITY STATUS OF PATENT OWNER: SMALL ENTITY |
|
| LAPS | Lapse for failure to pay maintenance fees |
Free format text: PATENT EXPIRED FOR FAILURE TO PAY MAINTENANCE FEES (ORIGINAL EVENT CODE: EXP.); ENTITY STATUS OF PATENT OWNER: SMALL ENTITY |
|
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| FP | Lapsed due to failure to pay maintenance fee |
Effective date: 20220923 |