US20070065483A1 - In vivo formed matrices including natural biodegradable polysaccharides and uses thereof - Google Patents
In vivo formed matrices including natural biodegradable polysaccharides and uses thereof Download PDFInfo
- Publication number
- US20070065483A1 US20070065483A1 US11/271,238 US27123805A US2007065483A1 US 20070065483 A1 US20070065483 A1 US 20070065483A1 US 27123805 A US27123805 A US 27123805A US 2007065483 A1 US2007065483 A1 US 2007065483A1
- Authority
- US
- United States
- Prior art keywords
- composition
- natural biodegradable
- coating
- biodegradable polysaccharide
- biodegradable
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 150000004676 glycans Chemical class 0.000 title claims abstract description 411
- 229920001282 polysaccharide Polymers 0.000 title claims abstract description 410
- 239000005017 polysaccharide Substances 0.000 title claims abstract description 410
- 238000001727 in vivo Methods 0.000 title claims abstract description 42
- 239000012867 bioactive agent Substances 0.000 claims abstract description 247
- 230000008878 coupling Effects 0.000 claims abstract description 121
- 238000010168 coupling process Methods 0.000 claims abstract description 121
- 238000005859 coupling reaction Methods 0.000 claims abstract description 121
- 239000011159 matrix material Substances 0.000 claims abstract description 99
- 238000000576 coating method Methods 0.000 claims description 325
- 239000011248 coating agent Substances 0.000 claims description 260
- 239000000203 mixture Substances 0.000 claims description 258
- 229920000856 Amylose Polymers 0.000 claims description 129
- 238000000034 method Methods 0.000 claims description 111
- 239000000463 material Substances 0.000 claims description 67
- 229920002774 Maltodextrin Polymers 0.000 claims description 57
- 239000005913 Maltodextrin Substances 0.000 claims description 54
- 229940035034 maltodextrin Drugs 0.000 claims description 54
- 238000006116 polymerization reaction Methods 0.000 claims description 26
- 230000002209 hydrophobic effect Effects 0.000 claims description 25
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 15
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 14
- NIXOWILDQLNWCW-UHFFFAOYSA-M acrylate group Chemical group C(C=C)(=O)[O-] NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 claims description 12
- 239000003505 polymerization initiator Substances 0.000 claims description 11
- 102000039446 nucleic acids Human genes 0.000 claims description 10
- 108020004707 nucleic acids Proteins 0.000 claims description 10
- 150000007523 nucleic acids Chemical class 0.000 claims description 10
- 229910052751 metal Inorganic materials 0.000 claims description 9
- 239000002184 metal Substances 0.000 claims description 9
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical group NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 claims description 8
- 150000002739 metals Chemical class 0.000 claims description 7
- 229920001184 polypeptide Polymers 0.000 claims description 7
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 7
- 102000004316 Oxidoreductases Human genes 0.000 claims description 6
- 108090000854 Oxidoreductases Proteins 0.000 claims description 6
- 235000014113 dietary fatty acids Nutrition 0.000 claims description 6
- 229930195729 fatty acid Natural products 0.000 claims description 6
- 239000000194 fatty acid Substances 0.000 claims description 6
- 150000004665 fatty acids Chemical class 0.000 claims description 6
- FQPSGWSUVKBHSU-UHFFFAOYSA-N methacrylamide Chemical group CC(=C)C(N)=O FQPSGWSUVKBHSU-UHFFFAOYSA-N 0.000 claims description 6
- 150000003839 salts Chemical class 0.000 claims description 6
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 4
- 108091007187 Reductases Proteins 0.000 claims description 3
- 229910044991 metal oxide Inorganic materials 0.000 claims description 3
- 150000004706 metal oxides Chemical class 0.000 claims description 3
- CERQOIWHTDAKMF-UHFFFAOYSA-M methacrylate group Chemical group C(C(=C)C)(=O)[O-] CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 claims description 3
- 150000002978 peroxides Chemical class 0.000 claims description 3
- WROUWQQRXUBECT-UHFFFAOYSA-N 2-ethylacrylic acid Chemical group CCC(=C)C(O)=O WROUWQQRXUBECT-UHFFFAOYSA-N 0.000 claims description 2
- LVHBHZANLOWSRM-UHFFFAOYSA-N itaconic acid Chemical group OC(=O)CC(=C)C(O)=O LVHBHZANLOWSRM-UHFFFAOYSA-N 0.000 claims description 2
- 125000003011 styrenyl group Chemical group [H]\C(*)=C(/[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 claims description 2
- 230000001590 oxidative effect Effects 0.000 claims 1
- 230000001225 therapeutic effect Effects 0.000 abstract description 9
- 239000000243 solution Substances 0.000 description 150
- 239000011859 microparticle Substances 0.000 description 105
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 100
- 239000002953 phosphate buffered saline Substances 0.000 description 100
- 229920000642 polymer Polymers 0.000 description 93
- -1 PDGF Chemical compound 0.000 description 79
- 239000003999 initiator Substances 0.000 description 79
- 239000000565 sealant Substances 0.000 description 75
- 230000015556 catabolic process Effects 0.000 description 51
- 238000006731 degradation reaction Methods 0.000 description 51
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 47
- 230000015572 biosynthetic process Effects 0.000 description 47
- 238000002360 preparation method Methods 0.000 description 46
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 44
- 239000010410 layer Substances 0.000 description 42
- 239000007943 implant Substances 0.000 description 41
- 239000003153 chemical reaction reagent Substances 0.000 description 40
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 39
- 239000008199 coating composition Substances 0.000 description 34
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 33
- 150000001875 compounds Chemical class 0.000 description 30
- 102000004139 alpha-Amylases Human genes 0.000 description 29
- 108090000637 alpha-Amylases Proteins 0.000 description 29
- 210000001519 tissue Anatomy 0.000 description 29
- 229940024171 alpha-amylase Drugs 0.000 description 28
- 229920002988 biodegradable polymer Polymers 0.000 description 28
- 239000004621 biodegradable polymer Substances 0.000 description 28
- 239000003795 chemical substances by application Substances 0.000 description 26
- 230000006870 function Effects 0.000 description 26
- 238000005286 illumination Methods 0.000 description 26
- 239000000047 product Substances 0.000 description 26
- 229920002472 Starch Polymers 0.000 description 25
- 235000019698 starch Nutrition 0.000 description 25
- 239000003638 chemical reducing agent Substances 0.000 description 24
- 239000008107 starch Substances 0.000 description 24
- 239000003814 drug Substances 0.000 description 23
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 22
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 22
- 229940098773 bovine serum albumin Drugs 0.000 description 22
- 239000007800 oxidant agent Substances 0.000 description 22
- 229940079593 drug Drugs 0.000 description 21
- 229920000139 polyethylene terephthalate Polymers 0.000 description 21
- 239000005020 polyethylene terephthalate Substances 0.000 description 21
- 108010089934 carbohydrase Proteins 0.000 description 20
- 108090000623 proteins and genes Proteins 0.000 description 20
- 210000004027 cell Anatomy 0.000 description 19
- 102000004169 proteins and genes Human genes 0.000 description 19
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 18
- 229920002614 Polyether block amide Polymers 0.000 description 18
- 238000006243 chemical reaction Methods 0.000 description 18
- 230000000694 effects Effects 0.000 description 18
- 235000018102 proteins Nutrition 0.000 description 18
- 239000000758 substrate Substances 0.000 description 18
- 238000012360 testing method Methods 0.000 description 18
- 102000004190 Enzymes Human genes 0.000 description 17
- 108090000790 Enzymes Proteins 0.000 description 17
- 239000000306 component Substances 0.000 description 17
- 229940088598 enzyme Drugs 0.000 description 17
- IRLPACMLTUPBCL-KQYNXXCUSA-N 5'-adenylyl sulfate Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](COP(O)(=O)OS(O)(=O)=O)[C@@H](O)[C@H]1O IRLPACMLTUPBCL-KQYNXXCUSA-N 0.000 description 16
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 16
- 230000001186 cumulative effect Effects 0.000 description 16
- 239000000126 substance Substances 0.000 description 16
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 15
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 15
- 230000008901 benefit Effects 0.000 description 15
- 239000003480 eluent Substances 0.000 description 15
- 229920001296 polysiloxane Polymers 0.000 description 15
- 238000003756 stirring Methods 0.000 description 15
- 238000002965 ELISA Methods 0.000 description 13
- 229920001577 copolymer Polymers 0.000 description 13
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 13
- 239000004382 Amylase Substances 0.000 description 12
- 102000013142 Amylases Human genes 0.000 description 12
- 108010065511 Amylases Proteins 0.000 description 12
- 229920001213 Polysorbate 20 Polymers 0.000 description 12
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 12
- 230000003213 activating effect Effects 0.000 description 12
- 235000019418 amylase Nutrition 0.000 description 12
- 239000012620 biological material Substances 0.000 description 12
- 239000008367 deionised water Substances 0.000 description 12
- 229910021641 deionized water Inorganic materials 0.000 description 12
- 238000002156 mixing Methods 0.000 description 12
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 12
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 12
- 239000007787 solid Substances 0.000 description 12
- 238000003786 synthesis reaction Methods 0.000 description 12
- 229920000945 Amylopectin Polymers 0.000 description 11
- 229920002307 Dextran Polymers 0.000 description 11
- 229920002245 Dextrose equivalent Polymers 0.000 description 11
- 241001465754 Metazoa Species 0.000 description 11
- 241000283973 Oryctolagus cuniculus Species 0.000 description 11
- 210000004369 blood Anatomy 0.000 description 11
- 239000008280 blood Substances 0.000 description 11
- 229960002086 dextran Drugs 0.000 description 11
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 description 11
- 229920000136 polysorbate Polymers 0.000 description 11
- 230000003578 releasing effect Effects 0.000 description 11
- 239000012812 sealant material Substances 0.000 description 11
- 239000010935 stainless steel Substances 0.000 description 11
- 229910001220 stainless steel Inorganic materials 0.000 description 11
- 238000011282 treatment Methods 0.000 description 11
- 238000005160 1H NMR spectroscopy Methods 0.000 description 10
- 238000005481 NMR spectroscopy Methods 0.000 description 10
- 238000002835 absorbance Methods 0.000 description 10
- 239000007864 aqueous solution Substances 0.000 description 10
- 230000004888 barrier function Effects 0.000 description 10
- 238000000502 dialysis Methods 0.000 description 10
- 239000000499 gel Substances 0.000 description 10
- 239000011521 glass Substances 0.000 description 10
- 238000010348 incorporation Methods 0.000 description 10
- 230000002792 vascular Effects 0.000 description 10
- 108010035532 Collagen Proteins 0.000 description 9
- 102000008186 Collagen Human genes 0.000 description 9
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 9
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- 150000001412 amines Chemical class 0.000 description 9
- 235000010323 ascorbic acid Nutrition 0.000 description 9
- 229960005070 ascorbic acid Drugs 0.000 description 9
- 239000011668 ascorbic acid Substances 0.000 description 9
- YHASWHZGWUONAO-UHFFFAOYSA-N butanoyl butanoate Chemical compound CCCC(=O)OC(=O)CCC YHASWHZGWUONAO-UHFFFAOYSA-N 0.000 description 9
- 229920001436 collagen Polymers 0.000 description 9
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 9
- 239000002245 particle Substances 0.000 description 9
- 238000010526 radical polymerization reaction Methods 0.000 description 9
- 230000008439 repair process Effects 0.000 description 9
- 229920001059 synthetic polymer Polymers 0.000 description 9
- KKEYFWRCBNTPAC-UHFFFAOYSA-L terephthalate(2-) Chemical compound [O-]C(=O)C1=CC=C(C([O-])=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-L 0.000 description 9
- 230000003612 virological effect Effects 0.000 description 9
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 8
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 8
- 208000007536 Thrombosis Diseases 0.000 description 8
- 239000008351 acetate buffer Substances 0.000 description 8
- 238000012377 drug delivery Methods 0.000 description 8
- 238000001035 drying Methods 0.000 description 8
- 229920000669 heparin Polymers 0.000 description 8
- 230000001976 improved effect Effects 0.000 description 8
- 238000011065 in-situ storage Methods 0.000 description 8
- 230000000977 initiatory effect Effects 0.000 description 8
- 239000007788 liquid Substances 0.000 description 8
- 229920000083 poly(allylamine) Polymers 0.000 description 8
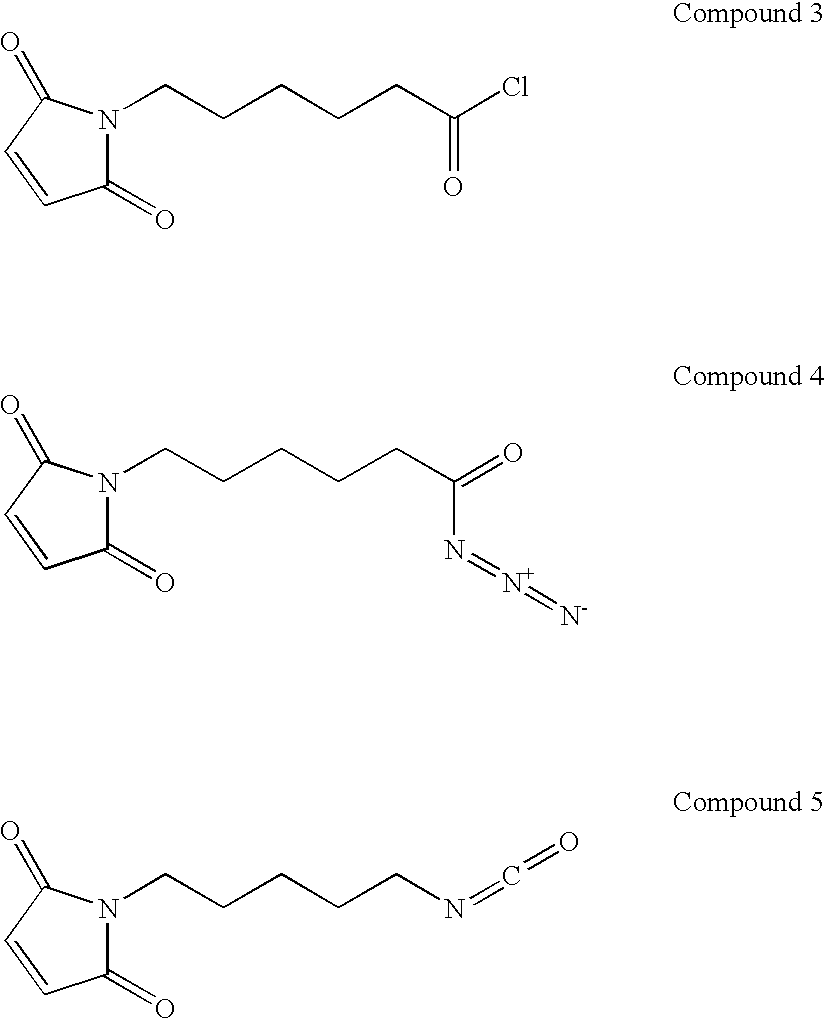
- GNJJTOYUIYQRKH-UHFFFAOYSA-N 6-(2,5-dioxopyrrol-1-yl)hexanoyl azide Chemical compound [N-]=[N+]=NC(=O)CCCCCN1C(=O)C=CC1=O GNJJTOYUIYQRKH-UHFFFAOYSA-N 0.000 description 7
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 7
- 238000004458 analytical method Methods 0.000 description 7
- 210000001124 body fluid Anatomy 0.000 description 7
- 238000007598 dipping method Methods 0.000 description 7
- 239000004744 fabric Substances 0.000 description 7
- 239000012530 fluid Substances 0.000 description 7
- 229960002897 heparin Drugs 0.000 description 7
- FZWBNHMXJMCXLU-BLAUPYHCSA-N isomaltotriose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@@H](OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O)O1 FZWBNHMXJMCXLU-BLAUPYHCSA-N 0.000 description 7
- 230000004048 modification Effects 0.000 description 7
- 238000012986 modification Methods 0.000 description 7
- 230000000399 orthopedic effect Effects 0.000 description 7
- 230000008569 process Effects 0.000 description 7
- 239000011541 reaction mixture Substances 0.000 description 7
- 238000003860 storage Methods 0.000 description 7
- 230000002885 thrombogenetic effect Effects 0.000 description 7
- 230000001732 thrombotic effect Effects 0.000 description 7
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 6
- UFBXHEYCJFUMIX-UHFFFAOYSA-N (3-azido-3-oxopropyl) prop-2-enoate Chemical compound C=CC(=O)OCCC(=O)N=[N+]=[N-] UFBXHEYCJFUMIX-UHFFFAOYSA-N 0.000 description 6
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 6
- 241000283690 Bos taurus Species 0.000 description 6
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical group CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 6
- 229920001661 Chitosan Polymers 0.000 description 6
- 102000003992 Peroxidases Human genes 0.000 description 6
- 239000010839 body fluid Substances 0.000 description 6
- 210000000988 bone and bone Anatomy 0.000 description 6
- 230000000747 cardiac effect Effects 0.000 description 6
- 229960001760 dimethyl sulfoxide Drugs 0.000 description 6
- 239000008103 glucose Substances 0.000 description 6
- 239000003102 growth factor Substances 0.000 description 6
- 229920002674 hyaluronan Polymers 0.000 description 6
- 229960003160 hyaluronic acid Drugs 0.000 description 6
- 230000001900 immune effect Effects 0.000 description 6
- 238000002513 implantation Methods 0.000 description 6
- 238000005259 measurement Methods 0.000 description 6
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 6
- 108040007629 peroxidase activity proteins Proteins 0.000 description 6
- 230000000704 physical effect Effects 0.000 description 6
- 229920000728 polyester Polymers 0.000 description 6
- 229920001223 polyethylene glycol Polymers 0.000 description 6
- 239000011148 porous material Substances 0.000 description 6
- 102000005962 receptors Human genes 0.000 description 6
- 108020003175 receptors Proteins 0.000 description 6
- 238000005096 rolling process Methods 0.000 description 6
- 241000283707 Capra Species 0.000 description 5
- 108020004414 DNA Proteins 0.000 description 5
- 241000196324 Embryophyta Species 0.000 description 5
- PEEHTFAAVSWFBL-UHFFFAOYSA-N Maleimide Chemical compound O=C1NC(=O)C=C1 PEEHTFAAVSWFBL-UHFFFAOYSA-N 0.000 description 5
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 5
- 208000027418 Wounds and injury Diseases 0.000 description 5
- RYULULVJWLRDQH-UHFFFAOYSA-N [4-(bromomethyl)phenyl]-phenylmethanone Chemical compound C1=CC(CBr)=CC=C1C(=O)C1=CC=CC=C1 RYULULVJWLRDQH-UHFFFAOYSA-N 0.000 description 5
- 239000003242 anti bacterial agent Substances 0.000 description 5
- 229940088710 antibiotic agent Drugs 0.000 description 5
- 239000003146 anticoagulant agent Substances 0.000 description 5
- 150000001540 azides Chemical class 0.000 description 5
- 210000001175 cerebrospinal fluid Anatomy 0.000 description 5
- 230000008859 change Effects 0.000 description 5
- 125000003636 chemical group Chemical group 0.000 description 5
- 238000001816 cooling Methods 0.000 description 5
- 238000004132 cross linking Methods 0.000 description 5
- 239000007857 degradation product Substances 0.000 description 5
- 239000000975 dye Substances 0.000 description 5
- 230000007062 hydrolysis Effects 0.000 description 5
- 238000006460 hydrolysis reaction Methods 0.000 description 5
- 239000003112 inhibitor Substances 0.000 description 5
- 229920000747 poly(lactic acid) Polymers 0.000 description 5
- 238000007717 redox polymerization reaction Methods 0.000 description 5
- 238000010992 reflux Methods 0.000 description 5
- 229910000077 silane Inorganic materials 0.000 description 5
- 230000001954 sterilising effect Effects 0.000 description 5
- 238000004659 sterilization and disinfection Methods 0.000 description 5
- 238000010998 test method Methods 0.000 description 5
- 238000003260 vortexing Methods 0.000 description 5
- RFBYLSCVRUTUSB-ZZMNMWMASA-L (2r)-2-[(1s)-1,2-dihydroxyethyl]-3-hydroxy-5-oxo-2h-furan-4-olate;iron(2+) Chemical compound [Fe+2].OC[C@H](O)[C@H]1OC(=O)C(O)=C1[O-].OC[C@H](O)[C@H]1OC(=O)C(O)=C1[O-] RFBYLSCVRUTUSB-ZZMNMWMASA-L 0.000 description 4
- VNQXSTWCDUXYEZ-UHFFFAOYSA-N 1,7,7-trimethylbicyclo[2.2.1]heptane-2,3-dione Chemical compound C1CC2(C)C(=O)C(=O)C1C2(C)C VNQXSTWCDUXYEZ-UHFFFAOYSA-N 0.000 description 4
- WJFKNYWRSNBZNX-UHFFFAOYSA-N 10H-phenothiazine Chemical compound C1=CC=C2NC3=CC=CC=C3SC2=C1 WJFKNYWRSNBZNX-UHFFFAOYSA-N 0.000 description 4
- LRFVTYWOQMYALW-UHFFFAOYSA-N 9H-xanthine Chemical compound O=C1NC(=O)NC2=C1NC=N2 LRFVTYWOQMYALW-UHFFFAOYSA-N 0.000 description 4
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical compound OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- 102000000905 Cadherin Human genes 0.000 description 4
- 108050007957 Cadherin Proteins 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- 102000001706 Immunoglobulin Fab Fragments Human genes 0.000 description 4
- 108010054477 Immunoglobulin Fab Fragments Proteins 0.000 description 4
- 206010061218 Inflammation Diseases 0.000 description 4
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 4
- KFSLWBXXFJQRDL-UHFFFAOYSA-N Peracetic acid Chemical compound CC(=O)OO KFSLWBXXFJQRDL-UHFFFAOYSA-N 0.000 description 4
- AUNGANRZJHBGPY-SCRDCRAPSA-N Riboflavin Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-SCRDCRAPSA-N 0.000 description 4
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 4
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- XNOMWKDAQZCFGK-UHFFFAOYSA-N [4-[(4-benzoylphenyl)methoxymethyl]phenyl]-phenylmethanone Chemical compound C=1C=C(COCC=2C=CC(=CC=2)C(=O)C=2C=CC=CC=2)C=CC=1C(=O)C1=CC=CC=C1 XNOMWKDAQZCFGK-UHFFFAOYSA-N 0.000 description 4
- 229930006711 bornane-2,3-dione Natural products 0.000 description 4
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 230000006242 butyrylation Effects 0.000 description 4
- 238000010514 butyrylation reaction Methods 0.000 description 4
- 150000001720 carbohydrates Chemical group 0.000 description 4
- 210000000845 cartilage Anatomy 0.000 description 4
- 201000010099 disease Diseases 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- 238000000605 extraction Methods 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 239000012634 fragment Substances 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 229920001477 hydrophilic polymer Polymers 0.000 description 4
- 230000006872 improvement Effects 0.000 description 4
- CGIGDMFJXJATDK-UHFFFAOYSA-N indomethacin Chemical compound CC1=C(CC(O)=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(Cl)C=C1 CGIGDMFJXJATDK-UHFFFAOYSA-N 0.000 description 4
- 208000015181 infectious disease Diseases 0.000 description 4
- 230000004054 inflammatory process Effects 0.000 description 4
- 238000001802 infusion Methods 0.000 description 4
- 238000001990 intravenous administration Methods 0.000 description 4
- 238000007726 management method Methods 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- 230000008018 melting Effects 0.000 description 4
- 239000012528 membrane Substances 0.000 description 4
- 230000000926 neurological effect Effects 0.000 description 4
- 210000000056 organ Anatomy 0.000 description 4
- 229950000688 phenothiazine Drugs 0.000 description 4
- 239000004626 polylactic acid Substances 0.000 description 4
- 239000003805 procoagulant Substances 0.000 description 4
- 230000001737 promoting effect Effects 0.000 description 4
- 230000003331 prothrombotic effect Effects 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- 150000003254 radicals Chemical class 0.000 description 4
- 210000002966 serum Anatomy 0.000 description 4
- 238000000527 sonication Methods 0.000 description 4
- 229940032147 starch Drugs 0.000 description 4
- 239000008399 tap water Substances 0.000 description 4
- 235000020679 tap water Nutrition 0.000 description 4
- 150000003512 tertiary amines Chemical class 0.000 description 4
- 239000004753 textile Substances 0.000 description 4
- 230000017423 tissue regeneration Effects 0.000 description 4
- 108010047303 von Willebrand Factor Proteins 0.000 description 4
- 102100036537 von Willebrand factor Human genes 0.000 description 4
- 229960001134 von willebrand factor Drugs 0.000 description 4
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 3
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 3
- CYUZOYPRAQASLN-UHFFFAOYSA-N 3-prop-2-enoyloxypropanoic acid Chemical compound OC(=O)CCOC(=O)C=C CYUZOYPRAQASLN-UHFFFAOYSA-N 0.000 description 3
- SQDAZGGFXASXDW-UHFFFAOYSA-N 5-bromo-2-(trifluoromethoxy)pyridine Chemical compound FC(F)(F)OC1=CC=C(Br)C=N1 SQDAZGGFXASXDW-UHFFFAOYSA-N 0.000 description 3
- SLXKOJJOQWFEFD-UHFFFAOYSA-N 6-aminohexanoic acid Chemical compound NCCCCCC(O)=O SLXKOJJOQWFEFD-UHFFFAOYSA-N 0.000 description 3
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 3
- 206010002329 Aneurysm Diseases 0.000 description 3
- 102000016289 Cell Adhesion Molecules Human genes 0.000 description 3
- 108010067225 Cell Adhesion Molecules Proteins 0.000 description 3
- 229920001287 Chondroitin sulfate Polymers 0.000 description 3
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 3
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 3
- 102100027285 Fanconi anemia group B protein Human genes 0.000 description 3
- 108010049003 Fibrinogen Proteins 0.000 description 3
- 102000008946 Fibrinogen Human genes 0.000 description 3
- 208000010412 Glaucoma Diseases 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 241000124008 Mammalia Species 0.000 description 3
- 241001529936 Murinae Species 0.000 description 3
- 208000008883 Patent Foramen Ovale Diseases 0.000 description 3
- 102100033237 Pro-epidermal growth factor Human genes 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 3
- 229930006000 Sucrose Natural products 0.000 description 3
- 102000009618 Transforming Growth Factors Human genes 0.000 description 3
- 108010009583 Transforming Growth Factors Proteins 0.000 description 3
- 208000018756 Variant Creutzfeldt-Jakob disease Diseases 0.000 description 3
- 208000001910 Ventricular Heart Septal Defects Diseases 0.000 description 3
- 241000700605 Viruses Species 0.000 description 3
- 125000000217 alkyl group Chemical group 0.000 description 3
- ROBVIMPUHSLWNV-UHFFFAOYSA-N aminoglutethimide Chemical compound C=1C=C(N)C=CC=1C1(CC)CCC(=O)NC1=O ROBVIMPUHSLWNV-UHFFFAOYSA-N 0.000 description 3
- 238000002399 angioplasty Methods 0.000 description 3
- 230000003466 anti-cipated effect Effects 0.000 description 3
- 229940121363 anti-inflammatory agent Drugs 0.000 description 3
- 239000002260 anti-inflammatory agent Substances 0.000 description 3
- 230000001028 anti-proliverative effect Effects 0.000 description 3
- 230000002421 anti-septic effect Effects 0.000 description 3
- 230000000890 antigenic effect Effects 0.000 description 3
- 229940064004 antiseptic throat preparations Drugs 0.000 description 3
- 239000003443 antiviral agent Substances 0.000 description 3
- 238000013459 approach Methods 0.000 description 3
- 238000003149 assay kit Methods 0.000 description 3
- 229920001400 block copolymer Polymers 0.000 description 3
- 208000005881 bovine spongiform encephalopathy Diseases 0.000 description 3
- 210000000481 breast Anatomy 0.000 description 3
- 235000014633 carbohydrates Nutrition 0.000 description 3
- 230000017455 cell-cell adhesion Effects 0.000 description 3
- 230000036755 cellular response Effects 0.000 description 3
- 239000001913 cellulose Substances 0.000 description 3
- 229920002678 cellulose Polymers 0.000 description 3
- 239000000919 ceramic Substances 0.000 description 3
- 239000000460 chlorine Substances 0.000 description 3
- 229940059329 chondroitin sulfate Drugs 0.000 description 3
- 239000000356 contaminant Substances 0.000 description 3
- 229920006037 cross link polymer Polymers 0.000 description 3
- 230000000593 degrading effect Effects 0.000 description 3
- 238000006356 dehydrogenation reaction Methods 0.000 description 3
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 3
- 239000012153 distilled water Substances 0.000 description 3
- 239000004222 ferrous gluconate Substances 0.000 description 3
- 235000013924 ferrous gluconate Nutrition 0.000 description 3
- 229960001645 ferrous gluconate Drugs 0.000 description 3
- 239000000835 fiber Substances 0.000 description 3
- 229940012952 fibrinogen Drugs 0.000 description 3
- 238000001415 gene therapy Methods 0.000 description 3
- 230000012010 growth Effects 0.000 description 3
- 238000001631 haemodialysis Methods 0.000 description 3
- 210000002216 heart Anatomy 0.000 description 3
- 230000000322 hemodialysis Effects 0.000 description 3
- 239000003018 immunosuppressive agent Substances 0.000 description 3
- 229940125721 immunosuppressive agent Drugs 0.000 description 3
- 230000001965 increasing effect Effects 0.000 description 3
- VRIVJOXICYMTAG-IYEMJOQQSA-L iron(ii) gluconate Chemical compound [Fe+2].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O VRIVJOXICYMTAG-IYEMJOQQSA-L 0.000 description 3
- 230000033001 locomotion Effects 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 230000001404 mediated effect Effects 0.000 description 3
- 239000003607 modifier Substances 0.000 description 3
- 150000002772 monosaccharides Chemical class 0.000 description 3
- 239000012044 organic layer Substances 0.000 description 3
- 239000003960 organic solvent Substances 0.000 description 3
- 230000002093 peripheral effect Effects 0.000 description 3
- 229920001606 poly(lactic acid-co-glycolic acid) Polymers 0.000 description 3
- 229920001707 polybutylene terephthalate Polymers 0.000 description 3
- 238000010561 standard procedure Methods 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 150000003431 steroids Chemical class 0.000 description 3
- 239000005720 sucrose Substances 0.000 description 3
- 230000008467 tissue growth Effects 0.000 description 3
- 231100000331 toxic Toxicity 0.000 description 3
- 230000002588 toxic effect Effects 0.000 description 3
- 238000012546 transfer Methods 0.000 description 3
- 201000003130 ventricular septal defect Diseases 0.000 description 3
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 description 2
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 2
- CQFPHDPTNCRLJK-UHFFFAOYSA-N (3-chloro-3-oxopropyl) prop-2-enoate Chemical compound ClC(=O)CCOC(=O)C=C CQFPHDPTNCRLJK-UHFFFAOYSA-N 0.000 description 2
- DKBZZUNCOFCQFT-UHFFFAOYSA-L (4-benzoylphenyl)methyl-[2-[(4-benzoylphenyl)methyl-dimethylazaniumyl]ethyl]-dimethylazanium;dibromide Chemical compound [Br-].[Br-].C=1C=C(C(=O)C=2C=CC=CC=2)C=CC=1C[N+](C)(C)CC[N+](C)(C)CC(C=C1)=CC=C1C(=O)C1=CC=CC=C1 DKBZZUNCOFCQFT-UHFFFAOYSA-L 0.000 description 2
- WXPWZZHELZEVPO-UHFFFAOYSA-N (4-methylphenyl)-phenylmethanone Chemical compound C1=CC(C)=CC=C1C(=O)C1=CC=CC=C1 WXPWZZHELZEVPO-UHFFFAOYSA-N 0.000 description 2
- PVPBBTJXIKFICP-UHFFFAOYSA-N (7-aminophenothiazin-3-ylidene)azanium;chloride Chemical compound [Cl-].C1=CC(=[NH2+])C=C2SC3=CC(N)=CC=C3N=C21 PVPBBTJXIKFICP-UHFFFAOYSA-N 0.000 description 2
- IAKHMKGGTNLKSZ-INIZCTEOSA-N (S)-colchicine Chemical compound C1([C@@H](NC(C)=O)CC2)=CC(=O)C(OC)=CC=C1C1=C2C=C(OC)C(OC)=C1OC IAKHMKGGTNLKSZ-INIZCTEOSA-N 0.000 description 2
- SWJPEBQEEAHIGZ-UHFFFAOYSA-N 1,4-dibromobenzene Chemical compound BrC1=CC=C(Br)C=C1 SWJPEBQEEAHIGZ-UHFFFAOYSA-N 0.000 description 2
- MCTWTZJPVLRJOU-UHFFFAOYSA-N 1-methyl-1H-imidazole Chemical compound CN1C=CN=C1 MCTWTZJPVLRJOU-UHFFFAOYSA-N 0.000 description 2
- ZXCIEWBDUAPBJF-MUUNZHRXSA-N 2-O-acetyl-1-O-octadecyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCCCCOC[C@@H](OC(C)=O)COP([O-])(=O)OCC[N+](C)(C)C ZXCIEWBDUAPBJF-MUUNZHRXSA-N 0.000 description 2
- DPNXHTDWGGVXID-UHFFFAOYSA-N 2-isocyanatoethyl prop-2-enoate Chemical compound C=CC(=O)OCCN=C=O DPNXHTDWGGVXID-UHFFFAOYSA-N 0.000 description 2
- OHQFLBMAHZJOJA-WAYWQWQTSA-N 6-[[(z)-3-carboxyprop-2-enoyl]amino]hexanoic acid Chemical compound OC(=O)CCCCCNC(=O)\C=C/C(O)=O OHQFLBMAHZJOJA-WAYWQWQTSA-N 0.000 description 2
- IHTHWBOGGPTYNH-UHFFFAOYSA-N 7-methyl-9-oxothioxanthene-3-carboxylic acid Chemical compound OC(=O)C1=CC=C2C(=O)C3=CC(C)=CC=C3SC2=C1 IHTHWBOGGPTYNH-UHFFFAOYSA-N 0.000 description 2
- GJCOSYZMQJWQCA-UHFFFAOYSA-N 9H-xanthene Chemical compound C1=CC=C2CC3=CC=CC=C3OC2=C1 GJCOSYZMQJWQCA-UHFFFAOYSA-N 0.000 description 2
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 2
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 2
- 108020004491 Antisense DNA Proteins 0.000 description 2
- 108020005544 Antisense RNA Proteins 0.000 description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 2
- 108010049931 Bone Morphogenetic Protein 2 Proteins 0.000 description 2
- 102100024506 Bone morphogenetic protein 2 Human genes 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- DLGOEMSEDOSKAD-UHFFFAOYSA-N Carmustine Chemical compound ClCCNC(=O)N(N=O)CCCl DLGOEMSEDOSKAD-UHFFFAOYSA-N 0.000 description 2
- 229930186147 Cephalosporin Natural products 0.000 description 2
- 102100023804 Coagulation factor VII Human genes 0.000 description 2
- 229920001634 Copolyester Polymers 0.000 description 2
- 208000020406 Creutzfeldt Jacob disease Diseases 0.000 description 2
- 208000003407 Creutzfeldt-Jakob Syndrome Diseases 0.000 description 2
- 208000010859 Creutzfeldt-Jakob disease Diseases 0.000 description 2
- AUNGANRZJHBGPY-UHFFFAOYSA-N D-Lyxoflavin Natural products OCC(O)C(O)C(O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-UHFFFAOYSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- 229920000045 Dermatan sulfate Polymers 0.000 description 2
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 2
- 102000016942 Elastin Human genes 0.000 description 2
- 108010014258 Elastin Proteins 0.000 description 2
- 101800003838 Epidermal growth factor Proteins 0.000 description 2
- ULGZDMOVFRHVEP-RWJQBGPGSA-N Erythromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@@](C)(O)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 ULGZDMOVFRHVEP-RWJQBGPGSA-N 0.000 description 2
- OHCQJHSOBUTRHG-KGGHGJDLSA-N FORSKOLIN Chemical compound O=C([C@@]12O)C[C@](C)(C=C)O[C@]1(C)[C@@H](OC(=O)C)[C@@H](O)[C@@H]1[C@]2(C)[C@@H](O)CCC1(C)C OHCQJHSOBUTRHG-KGGHGJDLSA-N 0.000 description 2
- 108010071289 Factor XIII Proteins 0.000 description 2
- 108010067306 Fibronectins Proteins 0.000 description 2
- 102000016359 Fibronectins Human genes 0.000 description 2
- 108010073178 Glucan 1,4-alpha-Glucosidase Proteins 0.000 description 2
- 239000004366 Glucose oxidase Substances 0.000 description 2
- 108010015776 Glucose oxidase Proteins 0.000 description 2
- 102000003886 Glycoproteins Human genes 0.000 description 2
- 108090000288 Glycoproteins Proteins 0.000 description 2
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 description 2
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 2
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 description 2
- 229920002971 Heparan sulfate Polymers 0.000 description 2
- 108010028750 Integrin-Binding Sialoprotein Proteins 0.000 description 2
- 102000016921 Integrin-Binding Sialoprotein Human genes 0.000 description 2
- 102000014150 Interferons Human genes 0.000 description 2
- 108010050904 Interferons Proteins 0.000 description 2
- 229920001202 Inulin Polymers 0.000 description 2
- 229920000288 Keratan sulfate Polymers 0.000 description 2
- FEWJPZIEWOKRBE-JCYAYHJZSA-L L-tartrate(2-) Chemical compound [O-]C(=O)[C@H](O)[C@@H](O)C([O-])=O FEWJPZIEWOKRBE-JCYAYHJZSA-L 0.000 description 2
- NHTGHBARYWONDQ-JTQLQIEISA-N L-α-methyl-Tyrosine Chemical compound OC(=O)[C@](N)(C)CC1=CC=C(O)C=C1 NHTGHBARYWONDQ-JTQLQIEISA-N 0.000 description 2
- 102000007547 Laminin Human genes 0.000 description 2
- 108010085895 Laminin Proteins 0.000 description 2
- BAPJBEWLBFYGME-UHFFFAOYSA-N Methyl acrylate Chemical compound COC(=O)C=C BAPJBEWLBFYGME-UHFFFAOYSA-N 0.000 description 2
- KWYHDKDOAIKMQN-UHFFFAOYSA-N N,N,N',N'-tetramethylethylenediamine Chemical compound CN(C)CCN(C)C KWYHDKDOAIKMQN-UHFFFAOYSA-N 0.000 description 2
- 108050000637 N-cadherin Proteins 0.000 description 2
- 206010028980 Neoplasm Diseases 0.000 description 2
- 102000004264 Osteopontin Human genes 0.000 description 2
- 108010081689 Osteopontin Proteins 0.000 description 2
- 101710135467 Outer capsid protein sigma-1 Proteins 0.000 description 2
- 241001494479 Pecora Species 0.000 description 2
- 108010003541 Platelet Activating Factor Proteins 0.000 description 2
- 108010038512 Platelet-Derived Growth Factor Proteins 0.000 description 2
- 102000010780 Platelet-Derived Growth Factor Human genes 0.000 description 2
- 239000004373 Pullulan Substances 0.000 description 2
- 229920001218 Pullulan Polymers 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 239000005708 Sodium hypochlorite Substances 0.000 description 2
- 208000006011 Stroke Diseases 0.000 description 2
- 102000007000 Tenascin Human genes 0.000 description 2
- 108010008125 Tenascin Proteins 0.000 description 2
- 108010000499 Thromboplastin Proteins 0.000 description 2
- 102000002262 Thromboplastin Human genes 0.000 description 2
- 108060008245 Thrombospondin Proteins 0.000 description 2
- 102000002938 Thrombospondin Human genes 0.000 description 2
- 102100031372 Thymidine phosphorylase Human genes 0.000 description 2
- 108700023160 Thymidine phosphorylases Proteins 0.000 description 2
- 108090000373 Tissue Plasminogen Activator Proteins 0.000 description 2
- 102000003978 Tissue Plasminogen Activator Human genes 0.000 description 2
- 206010046788 Uterine haemorrhage Diseases 0.000 description 2
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 description 2
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 2
- 108010031318 Vitronectin Proteins 0.000 description 2
- 102100035140 Vitronectin Human genes 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- XQAXGZLFSSPBMK-UHFFFAOYSA-M [7-(dimethylamino)phenothiazin-3-ylidene]-dimethylazanium;chloride;trihydrate Chemical compound O.O.O.[Cl-].C1=CC(=[N+](C)C)C=C2SC3=CC(N(C)C)=CC=C3N=C21 XQAXGZLFSSPBMK-UHFFFAOYSA-M 0.000 description 2
- 229960000446 abciximab Drugs 0.000 description 2
- 208000002223 abdominal aortic aneurysm Diseases 0.000 description 2
- 206010000269 abscess Diseases 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- DPKHZNPWBDQZCN-UHFFFAOYSA-N acridine orange free base Chemical compound C1=CC(N(C)C)=CC2=NC3=CC(N(C)C)=CC=C3C=C21 DPKHZNPWBDQZCN-UHFFFAOYSA-N 0.000 description 2
- RJURFGZVJUQBHK-UHFFFAOYSA-N actinomycin D Natural products CC1OC(=O)C(C(C)C)N(C)C(=O)CN(C)C(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)NC4C(=O)NC(C(N5CCCC5C(=O)N(C)CC(=O)N(C)C(C(C)C)C(=O)OC4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-UHFFFAOYSA-N 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 2
- AVJBPWGFOQAPRH-FWMKGIEWSA-N alpha-L-IdopA-(1->3)-beta-D-GalpNAc4S Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@H](OS(O)(=O)=O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](C(O)=O)O1 AVJBPWGFOQAPRH-FWMKGIEWSA-N 0.000 description 2
- 229910052782 aluminium Inorganic materials 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- 150000003863 ammonium salts Chemical class 0.000 description 2
- 230000003872 anastomosis Effects 0.000 description 2
- 229940035674 anesthetics Drugs 0.000 description 2
- PYKYMHQGRFAEBM-UHFFFAOYSA-N anthraquinone Natural products CCC(=O)c1c(O)c2C(=O)C3C(C=CC=C3O)C(=O)c2cc1CC(=O)OC PYKYMHQGRFAEBM-UHFFFAOYSA-N 0.000 description 2
- 150000004056 anthraquinones Chemical class 0.000 description 2
- 230000001754 anti-pyretic effect Effects 0.000 description 2
- 230000002785 anti-thrombosis Effects 0.000 description 2
- 229940127219 anticoagulant drug Drugs 0.000 description 2
- 239000000427 antigen Substances 0.000 description 2
- 108091007433 antigens Proteins 0.000 description 2
- 102000036639 antigens Human genes 0.000 description 2
- 229940030600 antihypertensive agent Drugs 0.000 description 2
- 239000002220 antihypertensive agent Substances 0.000 description 2
- 239000002221 antipyretic Substances 0.000 description 2
- 229940125716 antipyretic agent Drugs 0.000 description 2
- 239000003816 antisense DNA Substances 0.000 description 2
- 229960004676 antithrombotic agent Drugs 0.000 description 2
- 208000007474 aortic aneurysm Diseases 0.000 description 2
- 125000003118 aryl group Chemical group 0.000 description 2
- 230000003416 augmentation Effects 0.000 description 2
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 2
- 239000012965 benzophenone Substances 0.000 description 2
- DZBUGLKDJFMEHC-UHFFFAOYSA-N benzoquinolinylidene Natural products C1=CC=CC2=CC3=CC=CC=C3N=C21 DZBUGLKDJFMEHC-UHFFFAOYSA-N 0.000 description 2
- 230000005540 biological transmission Effects 0.000 description 2
- 239000012503 blood component Substances 0.000 description 2
- 230000036772 blood pressure Effects 0.000 description 2
- 230000008468 bone growth Effects 0.000 description 2
- 238000012662 bulk polymerization Methods 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 229940124587 cephalosporin Drugs 0.000 description 2
- 150000001780 cephalosporins Chemical class 0.000 description 2
- 238000007385 chemical modification Methods 0.000 description 2
- 239000005482 chemotactic factor Substances 0.000 description 2
- 229940045110 chitosan Drugs 0.000 description 2
- IJOOHPMOJXWVHK-UHFFFAOYSA-N chlorotrimethylsilane Chemical compound C[Si](C)(C)Cl IJOOHPMOJXWVHK-UHFFFAOYSA-N 0.000 description 2
- 239000011247 coating layer Substances 0.000 description 2
- 239000003184 complementary RNA Substances 0.000 description 2
- 238000009833 condensation Methods 0.000 description 2
- 230000005494 condensation Effects 0.000 description 2
- 210000000795 conjunctiva Anatomy 0.000 description 2
- 238000010276 construction Methods 0.000 description 2
- 210000004087 cornea Anatomy 0.000 description 2
- 238000001723 curing Methods 0.000 description 2
- 239000004053 dental implant Substances 0.000 description 2
- 229940051593 dermatan sulfate Drugs 0.000 description 2
- 239000003599 detergent Substances 0.000 description 2
- 229960000633 dextran sulfate Drugs 0.000 description 2
- 238000009792 diffusion process Methods 0.000 description 2
- 150000002016 disaccharides Chemical class 0.000 description 2
- 230000002526 effect on cardiovascular system Effects 0.000 description 2
- 229920002549 elastin Polymers 0.000 description 2
- 230000007831 electrophysiology Effects 0.000 description 2
- 238000002001 electrophysiology Methods 0.000 description 2
- 210000002889 endothelial cell Anatomy 0.000 description 2
- 230000007515 enzymatic degradation Effects 0.000 description 2
- 239000002532 enzyme inhibitor Substances 0.000 description 2
- SEACYXSIPDVVMV-UHFFFAOYSA-L eosin Y Chemical compound [Na+].[Na+].[O-]C(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C([O-])=C(Br)C=C21 SEACYXSIPDVVMV-UHFFFAOYSA-L 0.000 description 2
- 229940116977 epidermal growth factor Drugs 0.000 description 2
- IINNWAYUJNWZRM-UHFFFAOYSA-L erythrosin B Chemical compound [Na+].[Na+].[O-]C(=O)C1=CC=CC=C1C1=C2C=C(I)C(=O)C(I)=C2OC2=C(I)C([O-])=C(I)C=C21 IINNWAYUJNWZRM-UHFFFAOYSA-L 0.000 description 2
- 239000004174 erythrosine Substances 0.000 description 2
- 235000012732 erythrosine Nutrition 0.000 description 2
- 229940011411 erythrosine Drugs 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- UKZQEOHHLOYJLY-UHFFFAOYSA-M ethyl eosin Chemical compound [K+].CCOC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C([O-])=C(Br)C=C21 UKZQEOHHLOYJLY-UHFFFAOYSA-M 0.000 description 2
- 230000007717 exclusion Effects 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 239000003527 fibrinolytic agent Substances 0.000 description 2
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 2
- 229920001002 functional polymer Polymers 0.000 description 2
- 239000003193 general anesthetic agent Substances 0.000 description 2
- 229940116332 glucose oxidase Drugs 0.000 description 2
- 235000019420 glucose oxidase Nutrition 0.000 description 2
- 125000002791 glucosyl group Chemical group C1([C@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO)* 0.000 description 2
- 230000002439 hemostatic effect Effects 0.000 description 2
- 210000003494 hepatocyte Anatomy 0.000 description 2
- WQPDUTSPKFMPDP-OUMQNGNKSA-N hirudin Chemical compound C([C@@H](C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C=CC(OS(O)(=O)=O)=CC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CCCCN)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H]1NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@@H]2CSSC[C@@H](C(=O)N[C@@H](CCC(O)=O)C(=O)NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@H](C(=O)N[C@H](C(NCC(=O)N[C@@H](CCC(N)=O)C(=O)NCC(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)N2)=O)CSSC1)C(C)C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]1NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=2C=CC(O)=CC=2)NC(=O)[C@@H](NC(=O)[C@@H](N)C(C)C)C(C)C)[C@@H](C)O)CSSC1)C(C)C)[C@@H](C)O)[C@@H](C)O)C1=CC=CC=C1 WQPDUTSPKFMPDP-OUMQNGNKSA-N 0.000 description 2
- 229920001519 homopolymer Polymers 0.000 description 2
- 208000010544 human prion disease Diseases 0.000 description 2
- JYGXADMDTFJGBT-VWUMJDOOSA-N hydrocortisone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 JYGXADMDTFJGBT-VWUMJDOOSA-N 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 230000003301 hydrolyzing effect Effects 0.000 description 2
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 description 2
- 239000012216 imaging agent Substances 0.000 description 2
- 230000028993 immune response Effects 0.000 description 2
- 230000002163 immunogen Effects 0.000 description 2
- 229960000905 indomethacin Drugs 0.000 description 2
- 230000001939 inductive effect Effects 0.000 description 2
- 230000008595 infiltration Effects 0.000 description 2
- 238000001764 infiltration Methods 0.000 description 2
- 238000003780 insertion Methods 0.000 description 2
- 230000037431 insertion Effects 0.000 description 2
- 230000010354 integration Effects 0.000 description 2
- 229940029339 inulin Drugs 0.000 description 2
- JYJIGFIDKWBXDU-MNNPPOADSA-N inulin Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)OC[C@]1(OC[C@]2(OC[C@]3(OC[C@]4(OC[C@]5(OC[C@]6(OC[C@]7(OC[C@]8(OC[C@]9(OC[C@]%10(OC[C@]%11(OC[C@]%12(OC[C@]%13(OC[C@]%14(OC[C@]%15(OC[C@]%16(OC[C@]%17(OC[C@]%18(OC[C@]%19(OC[C@]%20(OC[C@]%21(OC[C@]%22(OC[C@]%23(OC[C@]%24(OC[C@]%25(OC[C@]%26(OC[C@]%27(OC[C@]%28(OC[C@]%29(OC[C@]%30(OC[C@]%31(OC[C@]%32(OC[C@]%33(OC[C@]%34(OC[C@]%35(OC[C@]%36(O[C@@H]%37[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O%37)O)[C@H]([C@H](O)[C@@H](CO)O%36)O)[C@H]([C@H](O)[C@@H](CO)O%35)O)[C@H]([C@H](O)[C@@H](CO)O%34)O)[C@H]([C@H](O)[C@@H](CO)O%33)O)[C@H]([C@H](O)[C@@H](CO)O%32)O)[C@H]([C@H](O)[C@@H](CO)O%31)O)[C@H]([C@H](O)[C@@H](CO)O%30)O)[C@H]([C@H](O)[C@@H](CO)O%29)O)[C@H]([C@H](O)[C@@H](CO)O%28)O)[C@H]([C@H](O)[C@@H](CO)O%27)O)[C@H]([C@H](O)[C@@H](CO)O%26)O)[C@H]([C@H](O)[C@@H](CO)O%25)O)[C@H]([C@H](O)[C@@H](CO)O%24)O)[C@H]([C@H](O)[C@@H](CO)O%23)O)[C@H]([C@H](O)[C@@H](CO)O%22)O)[C@H]([C@H](O)[C@@H](CO)O%21)O)[C@H]([C@H](O)[C@@H](CO)O%20)O)[C@H]([C@H](O)[C@@H](CO)O%19)O)[C@H]([C@H](O)[C@@H](CO)O%18)O)[C@H]([C@H](O)[C@@H](CO)O%17)O)[C@H]([C@H](O)[C@@H](CO)O%16)O)[C@H]([C@H](O)[C@@H](CO)O%15)O)[C@H]([C@H](O)[C@@H](CO)O%14)O)[C@H]([C@H](O)[C@@H](CO)O%13)O)[C@H]([C@H](O)[C@@H](CO)O%12)O)[C@H]([C@H](O)[C@@H](CO)O%11)O)[C@H]([C@H](O)[C@@H](CO)O%10)O)[C@H]([C@H](O)[C@@H](CO)O9)O)[C@H]([C@H](O)[C@@H](CO)O8)O)[C@H]([C@H](O)[C@@H](CO)O7)O)[C@H]([C@H](O)[C@@H](CO)O6)O)[C@H]([C@H](O)[C@@H](CO)O5)O)[C@H]([C@H](O)[C@@H](CO)O4)O)[C@H]([C@H](O)[C@@H](CO)O3)O)[C@H]([C@H](O)[C@@H](CO)O2)O)[C@@H](O)[C@H](O)[C@@H](CO)O1 JYJIGFIDKWBXDU-MNNPPOADSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 229910052742 iron Inorganic materials 0.000 description 2
- 239000012948 isocyanate Substances 0.000 description 2
- KXCLCNHUUKTANI-RBIYJLQWSA-N keratan Chemical compound CC(=O)N[C@@H]1[C@@H](O)C[C@@H](COS(O)(=O)=O)O[C@H]1O[C@@H]1[C@@H](O)[C@H](O[C@@H]2[C@H](O[C@@H](O[C@H]3[C@H]([C@@H](COS(O)(=O)=O)O[C@@H](O)[C@@H]3O)O)[C@H](NC(C)=O)[C@H]2O)COS(O)(=O)=O)O[C@H](COS(O)(=O)=O)[C@@H]1O KXCLCNHUUKTANI-RBIYJLQWSA-N 0.000 description 2
- 210000005248 left atrial appendage Anatomy 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 229910052744 lithium Inorganic materials 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 102000006240 membrane receptors Human genes 0.000 description 2
- 108020004084 membrane receptors Proteins 0.000 description 2
- 229910001092 metal group alloy Inorganic materials 0.000 description 2
- 229920000609 methyl cellulose Polymers 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- IYETZZCWLLUHIJ-UHFFFAOYSA-N methyl-(1-phenylpropan-2-yl)-prop-2-ynylazanium;chloride Chemical compound Cl.C#CCN(C)C(C)CC1=CC=CC=C1 IYETZZCWLLUHIJ-UHFFFAOYSA-N 0.000 description 2
- 239000001923 methylcellulose Substances 0.000 description 2
- 235000010981 methylcellulose Nutrition 0.000 description 2
- YYGBVRCTHASBKD-UHFFFAOYSA-M methylene green Chemical compound [Cl-].C1=CC(N(C)C)=C([N+]([O-])=O)C2=[S+]C3=CC(N(C)C)=CC=C3N=C21 YYGBVRCTHASBKD-UHFFFAOYSA-M 0.000 description 2
- 229960000907 methylthioninium chloride Drugs 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- CFCUWKMKBJTWLW-BKHRDMLASA-N mithramycin Chemical compound O([C@@H]1C[C@@H](O[C@H](C)[C@H]1O)OC=1C=C2C=C3C[C@H]([C@@H](C(=O)C3=C(O)C2=C(O)C=1C)O[C@@H]1O[C@H](C)[C@@H](O)[C@H](O[C@@H]2O[C@H](C)[C@H](O)[C@H](O[C@@H]3O[C@H](C)[C@@H](O)[C@@](C)(O)C3)C2)C1)[C@H](OC)C(=O)[C@@H](O)[C@@H](C)O)[C@H]1C[C@@H](O)[C@H](O)[C@@H](C)O1 CFCUWKMKBJTWLW-BKHRDMLASA-N 0.000 description 2
- 210000004115 mitral valve Anatomy 0.000 description 2
- 239000002808 molecular sieve Substances 0.000 description 2
- 239000000178 monomer Substances 0.000 description 2
- XHIRWEVPYCTARV-UHFFFAOYSA-N n-(3-aminopropyl)-2-methylprop-2-enamide;hydrochloride Chemical compound Cl.CC(=C)C(=O)NCCCN XHIRWEVPYCTARV-UHFFFAOYSA-N 0.000 description 2
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 229930014626 natural product Natural products 0.000 description 2
- 210000005036 nerve Anatomy 0.000 description 2
- 210000002569 neuron Anatomy 0.000 description 2
- 229910001000 nickel titanium Inorganic materials 0.000 description 2
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 239000012454 non-polar solvent Substances 0.000 description 2
- 229920001778 nylon Polymers 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- RPQRDASANLAFCM-UHFFFAOYSA-N oxiran-2-ylmethyl prop-2-enoate Chemical compound C=CC(=O)OCC1CO1 RPQRDASANLAFCM-UHFFFAOYSA-N 0.000 description 2
- 229940094443 oxytocics prostaglandins Drugs 0.000 description 2
- 229920001277 pectin Polymers 0.000 description 2
- 239000001814 pectin Substances 0.000 description 2
- 235000010987 pectin Nutrition 0.000 description 2
- 229940043138 pentosan polysulfate Drugs 0.000 description 2
- JRKICGRDRMAZLK-UHFFFAOYSA-L peroxydisulfate Chemical class [O-]S(=O)(=O)OOS([O-])(=O)=O JRKICGRDRMAZLK-UHFFFAOYSA-L 0.000 description 2
- 150000002989 phenols Chemical class 0.000 description 2
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical compound [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 description 2
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 description 2
- 239000013612 plasmid Substances 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 229960003171 plicamycin Drugs 0.000 description 2
- 239000002798 polar solvent Substances 0.000 description 2
- 229920000052 poly(p-xylylene) Polymers 0.000 description 2
- 229920001610 polycaprolactone Polymers 0.000 description 2
- 239000004632 polycaprolactone Substances 0.000 description 2
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 2
- 229920002635 polyurethane Polymers 0.000 description 2
- 239000004814 polyurethane Substances 0.000 description 2
- 238000011321 prophylaxis Methods 0.000 description 2
- 150000003180 prostaglandins Chemical class 0.000 description 2
- 235000019423 pullulan Nutrition 0.000 description 2
- 230000009257 reactivity Effects 0.000 description 2
- 239000012966 redox initiator Substances 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 230000008929 regeneration Effects 0.000 description 2
- 238000011069 regeneration method Methods 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 238000000518 rheometry Methods 0.000 description 2
- 235000019192 riboflavin Nutrition 0.000 description 2
- 239000002151 riboflavin Substances 0.000 description 2
- 229960002477 riboflavin Drugs 0.000 description 2
- AZJPTIGZZTZIDR-UHFFFAOYSA-L rose bengal Chemical compound [K+].[K+].[O-]C(=O)C1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1C1=C2C=C(I)C(=O)C(I)=C2OC2=C(I)C([O-])=C(I)C=C21 AZJPTIGZZTZIDR-UHFFFAOYSA-L 0.000 description 2
- 229930187593 rose bengal Natural products 0.000 description 2
- 229940081623 rose bengal Drugs 0.000 description 2
- STRXNPAVPKGJQR-UHFFFAOYSA-N rose bengal A Natural products O1C(=O)C(C(=CC=C2Cl)Cl)=C2C21C1=CC(I)=C(O)C(I)=C1OC1=C(I)C(O)=C(I)C=C21 STRXNPAVPKGJQR-UHFFFAOYSA-N 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 208000008864 scrapie Diseases 0.000 description 2
- 238000009958 sewing Methods 0.000 description 2
- 239000002356 single layer Substances 0.000 description 2
- QFJCIRLUMZQUOT-HPLJOQBZSA-N sirolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 QFJCIRLUMZQUOT-HPLJOQBZSA-N 0.000 description 2
- 239000002002 slurry Substances 0.000 description 2
- 150000003384 small molecules Chemical class 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 2
- SUKJFIGYRHOWBL-UHFFFAOYSA-N sodium hypochlorite Chemical compound [Na+].Cl[O-] SUKJFIGYRHOWBL-UHFFFAOYSA-N 0.000 description 2
- 229910052938 sodium sulfate Inorganic materials 0.000 description 2
- 235000011152 sodium sulphate Nutrition 0.000 description 2
- 230000003381 solubilizing effect Effects 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- 208000010110 spontaneous platelet aggregation Diseases 0.000 description 2
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 2
- 150000005846 sugar alcohols Polymers 0.000 description 2
- 229920002994 synthetic fiber Polymers 0.000 description 2
- 230000009885 systemic effect Effects 0.000 description 2
- 229960001685 tacrine Drugs 0.000 description 2
- 229940095064 tartrate Drugs 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- 238000002560 therapeutic procedure Methods 0.000 description 2
- 229960000187 tissue plasminogen activator Drugs 0.000 description 2
- 239000002407 tissue scaffold Substances 0.000 description 2
- 229960005267 tositumomab Drugs 0.000 description 2
- MAFQBSQRZKWGGE-UHFFFAOYSA-N trimethoxy-[2-[4-(2-trimethoxysilylethyl)phenyl]ethyl]silane Chemical compound CO[Si](OC)(OC)CCC1=CC=C(CC[Si](OC)(OC)OC)C=C1 MAFQBSQRZKWGGE-UHFFFAOYSA-N 0.000 description 2
- 230000002485 urinary effect Effects 0.000 description 2
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 2
- 229920002554 vinyl polymer Polymers 0.000 description 2
- 229920001285 xanthan gum Polymers 0.000 description 2
- 229940075420 xanthine Drugs 0.000 description 2
- 229910052725 zinc Inorganic materials 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- JIAARYAFYJHUJI-UHFFFAOYSA-L zinc dichloride Chemical compound [Cl-].[Cl-].[Zn+2] JIAARYAFYJHUJI-UHFFFAOYSA-L 0.000 description 2
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 description 2
- ZPEFMSTTZXJOTM-OULXEKPRSA-N (1R,2S)-tranylcypromine hydrochloride Chemical compound Cl.N[C@@H]1C[C@H]1C1=CC=CC=C1 ZPEFMSTTZXJOTM-OULXEKPRSA-N 0.000 description 1
- XDIYNQZUNSSENW-UUBOPVPUSA-N (2R,3S,4R,5R)-2,3,4,5,6-pentahydroxyhexanal Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O XDIYNQZUNSSENW-UUBOPVPUSA-N 0.000 description 1
- RJMIEHBSYVWVIN-LLVKDONJSA-N (2r)-2-[4-(3-oxo-1h-isoindol-2-yl)phenyl]propanoic acid Chemical compound C1=CC([C@H](C(O)=O)C)=CC=C1N1C(=O)C2=CC=CC=C2C1 RJMIEHBSYVWVIN-LLVKDONJSA-N 0.000 description 1
- RDJGLLICXDHJDY-NSHDSACASA-N (2s)-2-(3-phenoxyphenyl)propanoic acid Chemical compound OC(=O)[C@@H](C)C1=CC=CC(OC=2C=CC=CC=2)=C1 RDJGLLICXDHJDY-NSHDSACASA-N 0.000 description 1
- JXXHTBJOHYIVNT-UHFFFAOYSA-N (3-chloro-3-oxopropyl) 3-chloropropanoate Chemical compound ClCCC(=O)OCCC(Cl)=O JXXHTBJOHYIVNT-UHFFFAOYSA-N 0.000 description 1
- ZGGHKIMDNBDHJB-NRFPMOEYSA-M (3R,5S)-fluvastatin sodium Chemical compound [Na+].C12=CC=CC=C2N(C(C)C)C(\C=C\[C@@H](O)C[C@@H](O)CC([O-])=O)=C1C1=CC=C(F)C=C1 ZGGHKIMDNBDHJB-NRFPMOEYSA-M 0.000 description 1
- PUDHBTGHUJUUFI-SCTWWAJVSA-N (4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-n-[(2s,3r)-1-amino-3-hydroxy-1-oxobutan-2-yl]-19-[[(2r)-2-amino-3-naphthalen-2-ylpropanoyl]amino]-16-[(4-hydroxyphenyl)methyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-7-propan-2-yl-1,2-dithia-5,8,11,14,17-p Chemical compound C([C@H]1C(=O)N[C@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](N)CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)=O)C(C)C)C1=CC=C(O)C=C1 PUDHBTGHUJUUFI-SCTWWAJVSA-N 0.000 description 1
- SGKRLCUYIXIAHR-AKNGSSGZSA-N (4s,4ar,5s,5ar,6r,12ar)-4-(dimethylamino)-1,5,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide Chemical compound C1=CC=C2[C@H](C)[C@@H]([C@H](O)[C@@H]3[C@](C(O)=C(C(N)=O)C(=O)[C@H]3N(C)C)(O)C3=O)C3=C(O)C2=C1O SGKRLCUYIXIAHR-AKNGSSGZSA-N 0.000 description 1
- FFTVPQUHLQBXQZ-KVUCHLLUSA-N (4s,4as,5ar,12ar)-4,7-bis(dimethylamino)-1,10,11,12a-tetrahydroxy-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide Chemical compound C1C2=C(N(C)C)C=CC(O)=C2C(O)=C2[C@@H]1C[C@H]1[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]1(O)C2=O FFTVPQUHLQBXQZ-KVUCHLLUSA-N 0.000 description 1
- YEGBEDZBQAKLIO-UHFFFAOYSA-N (7,7-dimethyl-2,3-dioxo-4-bicyclo[2.2.1]heptanyl)methanesulfonic acid;hydrate Chemical compound O.C1CC2(CS(O)(=O)=O)C(=O)C(=O)C1C2(C)C YEGBEDZBQAKLIO-UHFFFAOYSA-N 0.000 description 1
- FPVKHBSQESCIEP-UHFFFAOYSA-N (8S)-3-(2-deoxy-beta-D-erythro-pentofuranosyl)-3,6,7,8-tetrahydroimidazo[4,5-d][1,3]diazepin-8-ol Natural products C1C(O)C(CO)OC1N1C(NC=NCC2O)=C2N=C1 FPVKHBSQESCIEP-UHFFFAOYSA-N 0.000 description 1
- 229920002818 (Hydroxyethyl)methacrylate Polymers 0.000 description 1
- GQGRDYWMOPRROR-ZIFKCHSBSA-N (e)-7-[(1r,2r,3s,5s)-3-hydroxy-5-[(4-phenylphenyl)methoxy]-2-piperidin-1-ylcyclopentyl]hept-4-enoic acid Chemical compound O([C@H]1C[C@@H]([C@@H]([C@H]1CC\C=C\CCC(O)=O)N1CCCCC1)O)CC(C=C1)=CC=C1C1=CC=CC=C1 GQGRDYWMOPRROR-ZIFKCHSBSA-N 0.000 description 1
- BQCIDUSAKPWEOX-UHFFFAOYSA-N 1,1-Difluoroethene Chemical compound FC(F)=C BQCIDUSAKPWEOX-UHFFFAOYSA-N 0.000 description 1
- RKDVKSZUMVYZHH-UHFFFAOYSA-N 1,4-dioxane-2,5-dione Chemical compound O=C1COC(=O)CO1 RKDVKSZUMVYZHH-UHFFFAOYSA-N 0.000 description 1
- PGRNEGLBSNLPNP-UHFFFAOYSA-N 1,6-dichloro-3-methylhex-1-ene Chemical compound ClC=CC(C)CCCCl PGRNEGLBSNLPNP-UHFFFAOYSA-N 0.000 description 1
- CZJMQTZQSNUDNV-UHFFFAOYSA-N 1-(2,3-dichlorophenyl)ethanamine Chemical compound CC(N)C1=CC=CC(Cl)=C1Cl CZJMQTZQSNUDNV-UHFFFAOYSA-N 0.000 description 1
- SCLQRRKBXAGUTF-UHFFFAOYSA-N 1-(5-isocyanatopentyl)pyrrole-2,5-dione Chemical compound O=C=NCCCCCN1C(=O)C=CC1=O SCLQRRKBXAGUTF-UHFFFAOYSA-N 0.000 description 1
- UOTMYNBWXDUBNX-UHFFFAOYSA-N 1-[(3,4-dimethoxyphenyl)methyl]-6,7-dimethoxyisoquinolin-2-ium;chloride Chemical compound Cl.C1=C(OC)C(OC)=CC=C1CC1=NC=CC2=CC(OC)=C(OC)C=C12 UOTMYNBWXDUBNX-UHFFFAOYSA-N 0.000 description 1
- YGEIMSMISRCBFF-UHFFFAOYSA-M 1-[bis(4-chlorophenyl)methyl]-3-[2-(2,4-dichlorophenyl)-2-[(2,4-dichlorophenyl)methoxy]ethyl]imidazol-3-ium;chloride Chemical compound [Cl-].C1=CC(Cl)=CC=C1C([N+]1=CN(CC(OCC=2C(=CC(Cl)=CC=2)Cl)C=2C(=CC(Cl)=CC=2)Cl)C=C1)C1=CC=C(Cl)C=C1 YGEIMSMISRCBFF-UHFFFAOYSA-M 0.000 description 1
- JWYVGKFDLWWQJX-UHFFFAOYSA-N 1-ethenylazepan-2-one Chemical compound C=CN1CCCCCC1=O JWYVGKFDLWWQJX-UHFFFAOYSA-N 0.000 description 1
- VSNHCAURESNICA-NJFSPNSNSA-N 1-oxidanylurea Chemical compound N[14C](=O)NO VSNHCAURESNICA-NJFSPNSNSA-N 0.000 description 1
- FUFLCEKSBBHCMO-UHFFFAOYSA-N 11-dehydrocorticosterone Natural products O=C1CCC2(C)C3C(=O)CC(C)(C(CC4)C(=O)CO)C4C3CCC2=C1 FUFLCEKSBBHCMO-UHFFFAOYSA-N 0.000 description 1
- QKBIEFMTGNXLAZ-UHFFFAOYSA-N 16-[(4-benzoylphenyl)methoxy]hexadecanoic acid Chemical compound C1=CC(COCCCCCCCCCCCCCCCC(=O)O)=CC=C1C(=O)C1=CC=CC=C1 QKBIEFMTGNXLAZ-UHFFFAOYSA-N 0.000 description 1
- QRIMLDXJAPZHJE-UHFFFAOYSA-N 2,3-dihydroxypropyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(O)CO QRIMLDXJAPZHJE-UHFFFAOYSA-N 0.000 description 1
- OWPUOLBODXJOKH-UHFFFAOYSA-N 2,3-dihydroxypropyl prop-2-enoate Chemical compound OCC(O)COC(=O)C=C OWPUOLBODXJOKH-UHFFFAOYSA-N 0.000 description 1
- ZIIUUSVHCHPIQD-UHFFFAOYSA-N 2,4,6-trimethyl-N-[3-(trifluoromethyl)phenyl]benzenesulfonamide Chemical compound CC1=CC(C)=CC(C)=C1S(=O)(=O)NC1=CC=CC(C(F)(F)F)=C1 ZIIUUSVHCHPIQD-UHFFFAOYSA-N 0.000 description 1
- FHIKZROVIDCMJA-UHFFFAOYSA-N 2-(2,2-diphenylpentanoyloxy)ethyl-diethylazanium;chloride Chemical compound Cl.C=1C=CC=CC=1C(C(=O)OCCN(CC)CC)(CCC)C1=CC=CC=C1 FHIKZROVIDCMJA-UHFFFAOYSA-N 0.000 description 1
- OZAIFHULBGXAKX-UHFFFAOYSA-N 2-(2-cyanopropan-2-yldiazenyl)-2-methylpropanenitrile Chemical compound N#CC(C)(C)N=NC(C)(C)C#N OZAIFHULBGXAKX-UHFFFAOYSA-N 0.000 description 1
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 1
- QUCRZIVNKYXKAM-UHFFFAOYSA-N 2-(dimethylamino)-1-phenothiazin-10-ylpropan-1-one;hydron;chloride Chemical compound Cl.C1=CC=C2N(C(=O)C(N(C)C)C)C3=CC=CC=C3SC2=C1 QUCRZIVNKYXKAM-UHFFFAOYSA-N 0.000 description 1
- CBGLNJDCMZWZOQ-UHFFFAOYSA-N 2-[2-(3-benzoylanilino)-2-oxoethyl]hexadecanoic acid Chemical compound CCCCCCCCCCCCCCC(C(O)=O)CC(=O)NC1=CC=CC(C(=O)C=2C=CC=CC=2)=C1 CBGLNJDCMZWZOQ-UHFFFAOYSA-N 0.000 description 1
- AZCMJRSTRGKARS-UHFFFAOYSA-N 2-[2-[2-[(4-benzoylphenyl)methoxy]ethylamino]-2-oxoethyl]hexadecanoic acid Chemical compound C1=CC(COCCNC(=O)CC(CCCCCCCCCCCCCC)C(O)=O)=CC=C1C(=O)C1=CC=CC=C1 AZCMJRSTRGKARS-UHFFFAOYSA-N 0.000 description 1
- LHTYJYAXCBIQEG-UHFFFAOYSA-N 2-[2-[3-[(4-benzoylbenzoyl)amino]propylamino]-2-oxoethyl]hexadecanoic acid Chemical compound C1=CC(C(=O)NCCCNC(=O)CC(CCCCCCCCCCCCCC)C(O)=O)=CC=C1C(=O)C1=CC=CC=C1 LHTYJYAXCBIQEG-UHFFFAOYSA-N 0.000 description 1
- MMSORURMHHPEGC-UHFFFAOYSA-N 2-[2-oxo-2-[12-(2-oxo-1,2-diphenylethoxy)dodecylamino]ethyl]hexadecanoic acid Chemical compound C=1C=CC=CC=1C(OCCCCCCCCCCCCNC(=O)CC(CCCCCCCCCCCCCC)C(O)=O)C(=O)C1=CC=CC=C1 MMSORURMHHPEGC-UHFFFAOYSA-N 0.000 description 1
- RTQWWZBSTRGEAV-PKHIMPSTSA-N 2-[[(2s)-2-[bis(carboxymethyl)amino]-3-[4-(methylcarbamoylamino)phenyl]propyl]-[2-[bis(carboxymethyl)amino]propyl]amino]acetic acid Chemical compound CNC(=O)NC1=CC=C(C[C@@H](CN(CC(C)N(CC(O)=O)CC(O)=O)CC(O)=O)N(CC(O)=O)CC(O)=O)C=C1 RTQWWZBSTRGEAV-PKHIMPSTSA-N 0.000 description 1
- TYIRELDXFZWDBV-UHFFFAOYSA-N 2-amino-1-cyclooctylethanol;hydrochloride Chemical compound Cl.NCC(O)C1CCCCCCC1 TYIRELDXFZWDBV-UHFFFAOYSA-N 0.000 description 1
- OMIGHNLMNHATMP-UHFFFAOYSA-N 2-hydroxyethyl prop-2-enoate Chemical compound OCCOC(=O)C=C OMIGHNLMNHATMP-UHFFFAOYSA-N 0.000 description 1
- MUIJMYIUSKMVOY-UHFFFAOYSA-N 2-methylprop-2-enamide 9H-thioxanthene 10-oxide Chemical compound CC(=C)C(N)=O.C1=CC=C2S(=O)C3=CC=CC=C3CC2=C1 MUIJMYIUSKMVOY-UHFFFAOYSA-N 0.000 description 1
- CTRPRMNBTVRDFH-UHFFFAOYSA-N 2-n-methyl-1,3,5-triazine-2,4,6-triamine Chemical class CNC1=NC(N)=NC(N)=N1 CTRPRMNBTVRDFH-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- APIXJSLKIYYUKG-UHFFFAOYSA-N 3 Isobutyl 1 methylxanthine Chemical compound O=C1N(C)C(=O)N(CC(C)C)C2=C1N=CN2 APIXJSLKIYYUKG-UHFFFAOYSA-N 0.000 description 1
- VDCDWNDTNSWDFJ-UHFFFAOYSA-N 3,5-dinitrocatechol Chemical compound OC1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1O VDCDWNDTNSWDFJ-UHFFFAOYSA-N 0.000 description 1
- BBAZDLONIUABKI-UHFFFAOYSA-N 3-(2,4-dichlorophenoxy)-n-methyl-n-prop-2-ynylpropan-1-amine;hydrochloride Chemical compound Cl.C#CCN(C)CCCOC1=CC=C(Cl)C=C1Cl BBAZDLONIUABKI-UHFFFAOYSA-N 0.000 description 1
- OFKWWALNMPEOSZ-UHFFFAOYSA-N 3-(hydrazinylmethyl)phenol Chemical compound NNCC1=CC=CC(O)=C1 OFKWWALNMPEOSZ-UHFFFAOYSA-N 0.000 description 1
- MEXSALMIYHSYHC-UHFFFAOYSA-N 3-[(3-benzoylphenyl)carbamoyl]heptadecanoic acid Chemical compound CCCCCCCCCCCCCCC(CC(O)=O)C(=O)NC1=CC=CC(C(=O)C=2C=CC=CC=2)=C1 MEXSALMIYHSYHC-UHFFFAOYSA-N 0.000 description 1
- CVUSKSUSMNOCQV-UHFFFAOYSA-N 3-[(4-benzoylphenyl)carbamoyl]heptadecanoic acid Chemical compound C1=CC(NC(=O)C(CC(O)=O)CCCCCCCCCCCCCC)=CC=C1C(=O)C1=CC=CC=C1 CVUSKSUSMNOCQV-UHFFFAOYSA-N 0.000 description 1
- AUXHPKRITNINQO-UHFFFAOYSA-N 3-[12-(2-oxo-1,2-diphenylethoxy)dodecylcarbamoyl]heptadecanoic acid Chemical compound C=1C=CC=CC=1C(OCCCCCCCCCCCCNC(=O)C(CC(O)=O)CCCCCCCCCCCCCC)C(=O)C1=CC=CC=C1 AUXHPKRITNINQO-UHFFFAOYSA-N 0.000 description 1
- PABZPROINNIKKP-UHFFFAOYSA-N 3-[2-[(4-benzoylphenyl)methoxy]ethylcarbamoyl]heptadecanoic acid Chemical compound C1=CC(COCCNC(=O)C(CC(O)=O)CCCCCCCCCCCCCC)=CC=C1C(=O)C1=CC=CC=C1 PABZPROINNIKKP-UHFFFAOYSA-N 0.000 description 1
- ZCNBZFRECRPCKU-UHFFFAOYSA-N 3-[2-[4-[bis(4-fluorophenyl)methylidene]-1-piperidinyl]ethyl]-2-sulfanylidene-1H-quinazolin-4-one Chemical compound C1=CC(F)=CC=C1C(C=1C=CC(F)=CC=1)=C1CCN(CCN2C(C3=CC=CC=C3NC2=S)=O)CC1 ZCNBZFRECRPCKU-UHFFFAOYSA-N 0.000 description 1
- BEVBWJOGIFYRPN-UHFFFAOYSA-N 3-[3-[(4-benzoylbenzoyl)amino]propylcarbamoyl]heptadecanoic acid Chemical compound C1=CC(C(=O)NCCCNC(=O)C(CC(O)=O)CCCCCCCCCCCCCC)=CC=C1C(=O)C1=CC=CC=C1 BEVBWJOGIFYRPN-UHFFFAOYSA-N 0.000 description 1
- NOIIUHRQUVNIDD-UHFFFAOYSA-N 3-[[oxo(pyridin-4-yl)methyl]hydrazo]-N-(phenylmethyl)propanamide Chemical compound C=1C=CC=CC=1CNC(=O)CCNNC(=O)C1=CC=NC=C1 NOIIUHRQUVNIDD-UHFFFAOYSA-N 0.000 description 1
- UQTZMGFTRHFAAM-ZETCQYMHSA-N 3-iodo-L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C(I)=C1 UQTZMGFTRHFAAM-ZETCQYMHSA-N 0.000 description 1
- DEHHYUARFKIUDI-UHFFFAOYSA-N 3-phenylprop-2-yn-1-amine Chemical compound NCC#CC1=CC=CC=C1 DEHHYUARFKIUDI-UHFFFAOYSA-N 0.000 description 1
- BHPSIKROCCEKQR-UHFFFAOYSA-N 3-sulfanylpyrrole-2,5-dione Chemical compound SC1=CC(=O)NC1=O BHPSIKROCCEKQR-UHFFFAOYSA-N 0.000 description 1
- VFXXTYGQYWRHJP-UHFFFAOYSA-N 4,4'-azobis(4-cyanopentanoic acid) Chemical compound OC(=O)CCC(C)(C#N)N=NC(C)(CCC(O)=O)C#N VFXXTYGQYWRHJP-UHFFFAOYSA-N 0.000 description 1
- HTHGAIADRJRJOY-UHFFFAOYSA-N 6-(4-bromophenyl)-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Chemical compound C1=CC(Br)=CC=C1C1N=C2SCCN2C1 HTHGAIADRJRJOY-UHFFFAOYSA-N 0.000 description 1
- WOJKKJKETHYEAC-UHFFFAOYSA-N 6-Maleimidocaproic acid Chemical compound OC(=O)CCCCCN1C(=O)C=CC1=O WOJKKJKETHYEAC-UHFFFAOYSA-N 0.000 description 1
- WYWHKKSPHMUBEB-UHFFFAOYSA-N 6-Mercaptoguanine Natural products N1C(N)=NC(=S)C2=C1N=CN2 WYWHKKSPHMUBEB-UHFFFAOYSA-N 0.000 description 1
- MFVJXLPANKSLLD-UHFFFAOYSA-N 6-[2-[4-[(4-fluorophenyl)-phenylmethylidene]-1-piperidinyl]ethyl]-7-methyl-5-thiazolo[3,2-a]pyrimidinone Chemical compound CC=1N=C2SC=CN2C(=O)C=1CCN(CC1)CCC1=C(C=1C=CC(F)=CC=1)C1=CC=CC=C1 MFVJXLPANKSLLD-UHFFFAOYSA-N 0.000 description 1
- NOOBQTYVTDBXTL-UHFFFAOYSA-N 6-hydroxy-1,3-benzothiazole-2-sulfonamide Chemical compound C1=C(O)C=C2SC(S(=O)(=O)N)=NC2=C1 NOOBQTYVTDBXTL-UHFFFAOYSA-N 0.000 description 1
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 description 1
- PIAMUVPINIPMQS-UHFFFAOYSA-N 7-methyl-n-[3-(2-methylprop-2-enoylamino)propyl]-9-oxothioxanthene-3-carboxamide Chemical compound C1=C(C)C=C2C(=O)C3=CC=C(C(=O)NCCCNC(=O)C(=C)C)C=C3SC2=C1 PIAMUVPINIPMQS-UHFFFAOYSA-N 0.000 description 1
- IADAQXMUWITWNG-UHFFFAOYSA-N 8,9-dichloro-2,3,4,5-tetrahydro-1h-benzo[c]azepine Chemical compound C1CCNCC2=C(Cl)C(Cl)=CC=C21 IADAQXMUWITWNG-UHFFFAOYSA-N 0.000 description 1
- 239000005541 ACE inhibitor Substances 0.000 description 1
- 208000030507 AIDS Diseases 0.000 description 1
- 108010059616 Activins Proteins 0.000 description 1
- 206010067484 Adverse reaction Diseases 0.000 description 1
- 108010088751 Albumins Proteins 0.000 description 1
- 102000009027 Albumins Human genes 0.000 description 1
- 108020000948 Antisense Oligonucleotides Proteins 0.000 description 1
- IYMAXBFPHPZYIK-BQBZGAKWSA-N Arg-Gly-Asp Chemical compound NC(N)=NCCC[C@H](N)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(O)=O IYMAXBFPHPZYIK-BQBZGAKWSA-N 0.000 description 1
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 1
- XUKUURHRXDUEBC-KAYWLYCHSA-N Atorvastatin Chemical compound C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CC[C@@H](O)C[C@@H](O)CC(O)=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 XUKUURHRXDUEBC-KAYWLYCHSA-N 0.000 description 1
- XUKUURHRXDUEBC-UHFFFAOYSA-N Atorvastatin Natural products C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CCC(O)CC(O)CC(O)=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 XUKUURHRXDUEBC-UHFFFAOYSA-N 0.000 description 1
- XHVAWZZCDCWGBK-WYRLRVFGSA-M Aurothioglucose Chemical compound OC[C@H]1O[C@H](S[Au])[C@H](O)[C@@H](O)[C@@H]1O XHVAWZZCDCWGBK-WYRLRVFGSA-M 0.000 description 1
- 108090001008 Avidin Proteins 0.000 description 1
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical class C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 description 1
- 239000005552 B01AC04 - Clopidogrel Substances 0.000 description 1
- 239000005528 B01AC05 - Ticlopidine Substances 0.000 description 1
- 108010001478 Bacitracin Proteins 0.000 description 1
- 206010004446 Benign prostatic hyperplasia Diseases 0.000 description 1
- 239000004342 Benzoyl peroxide Substances 0.000 description 1
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 description 1
- 102100026189 Beta-galactosidase Human genes 0.000 description 1
- 108010006654 Bleomycin Proteins 0.000 description 1
- 102000015081 Blood Coagulation Factors Human genes 0.000 description 1
- 108010039209 Blood Coagulation Factors Proteins 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- 108090000342 C-Type Lectins Proteins 0.000 description 1
- 102000003930 C-Type Lectins Human genes 0.000 description 1
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 229940127291 Calcium channel antagonist Drugs 0.000 description 1
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 1
- 108090000994 Catalytic RNA Proteins 0.000 description 1
- 102000053642 Catalytic RNA Human genes 0.000 description 1
- UQLLWWBDSUHNEB-CZUORRHYSA-N Cefaprin Chemical compound N([C@H]1[C@@H]2N(C1=O)C(=C(CS2)COC(=O)C)C(O)=O)C(=O)CSC1=CC=NC=C1 UQLLWWBDSUHNEB-CZUORRHYSA-N 0.000 description 1
- JWBOIMRXGHLCPP-UHFFFAOYSA-N Chloditan Chemical compound C=1C=CC=C(Cl)C=1C(C(Cl)Cl)C1=CC=C(Cl)C=C1 JWBOIMRXGHLCPP-UHFFFAOYSA-N 0.000 description 1
- GHXZTYHSJHQHIJ-UHFFFAOYSA-N Chlorhexidine Chemical compound C=1C=C(Cl)C=CC=1NC(N)=NC(N)=NCCCCCCN=C(N)N=C(N)NC1=CC=C(Cl)C=C1 GHXZTYHSJHQHIJ-UHFFFAOYSA-N 0.000 description 1
- PTOAARAWEBMLNO-KVQBGUIXSA-N Cladribine Chemical compound C1=NC=2C(N)=NC(Cl)=NC=2N1[C@H]1C[C@H](O)[C@@H](CO)O1 PTOAARAWEBMLNO-KVQBGUIXSA-N 0.000 description 1
- 102100022641 Coagulation factor IX Human genes 0.000 description 1
- 108050006018 Coagulation factor VII Proteins 0.000 description 1
- 102100026735 Coagulation factor VIII Human genes 0.000 description 1
- 102100029117 Coagulation factor X Human genes 0.000 description 1
- 102100030563 Coagulation factor XI Human genes 0.000 description 1
- 102100030556 Coagulation factor XII Human genes 0.000 description 1
- 206010053567 Coagulopathies Diseases 0.000 description 1
- 229910000684 Cobalt-chrome Inorganic materials 0.000 description 1
- 102000004266 Collagen Type IV Human genes 0.000 description 1
- 108010042086 Collagen Type IV Proteins 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- MFYSYFVPBJMHGN-ZPOLXVRWSA-N Cortisone Chemical compound O=C1CC[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 MFYSYFVPBJMHGN-ZPOLXVRWSA-N 0.000 description 1
- MFYSYFVPBJMHGN-UHFFFAOYSA-N Cortisone Natural products O=C1CCC2(C)C3C(=O)CC(C)(C(CC4)(O)C(=O)CO)C4C3CCC2=C1 MFYSYFVPBJMHGN-UHFFFAOYSA-N 0.000 description 1
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 1
- PMATZTZNYRCHOR-CGLBZJNRSA-N Cyclosporin A Chemical compound CC[C@@H]1NC(=O)[C@H]([C@H](O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C1=O PMATZTZNYRCHOR-CGLBZJNRSA-N 0.000 description 1
- 108010036949 Cyclosporine Proteins 0.000 description 1
- UHDGCWIWMRVCDJ-CCXZUQQUSA-N Cytarabine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 UHDGCWIWMRVCDJ-CCXZUQQUSA-N 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- 108010092160 Dactinomycin Proteins 0.000 description 1
- SUZLHDUTVMZSEV-UHFFFAOYSA-N Deoxycoleonol Natural products C12C(=O)CC(C)(C=C)OC2(C)C(OC(=O)C)C(O)C2C1(C)C(O)CCC2(C)C SUZLHDUTVMZSEV-UHFFFAOYSA-N 0.000 description 1
- 241000702421 Dependoparvovirus Species 0.000 description 1
- 108010001394 Disaccharidases Proteins 0.000 description 1
- 108010056764 Eptifibatide Proteins 0.000 description 1
- 102000003951 Erythropoietin Human genes 0.000 description 1
- 108090000394 Erythropoietin Proteins 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- HKVAMNSJSFKALM-GKUWKFKPSA-N Everolimus Chemical compound C1C[C@@H](OCCO)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 HKVAMNSJSFKALM-GKUWKFKPSA-N 0.000 description 1
- 108010076282 Factor IX Proteins 0.000 description 1
- 108010014172 Factor V Proteins 0.000 description 1
- 108010023321 Factor VII Proteins 0.000 description 1
- 108010014173 Factor X Proteins 0.000 description 1
- 108010074864 Factor XI Proteins 0.000 description 1
- 108010080865 Factor XII Proteins 0.000 description 1
- 108010074860 Factor Xa Proteins 0.000 description 1
- 108010073385 Fibrin Proteins 0.000 description 1
- 102000009123 Fibrin Human genes 0.000 description 1
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 1
- 108050007372 Fibroblast Growth Factor Proteins 0.000 description 1
- 102000018233 Fibroblast Growth Factor Human genes 0.000 description 1
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 description 1
- 229940123457 Free radical scavenger Drugs 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 208000003098 Ganglion Cysts Diseases 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- JRZJKWGQFNTSRN-UHFFFAOYSA-N Geldanamycin Natural products C1C(C)CC(OC)C(O)C(C)C=C(C)C(OC(N)=O)C(OC)CCC=C(C)C(=O)NC2=CC(=O)C(OC)=C1C2=O JRZJKWGQFNTSRN-UHFFFAOYSA-N 0.000 description 1
- CEAZRRDELHUEMR-URQXQFDESA-N Gentamicin Chemical compound O1[C@H](C(C)NC)CC[C@@H](N)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](NC)[C@@](C)(O)CO2)O)[C@H](N)C[C@@H]1N CEAZRRDELHUEMR-URQXQFDESA-N 0.000 description 1
- 229930182566 Gentamicin Natural products 0.000 description 1
- 102000006395 Globulins Human genes 0.000 description 1
- 108010044091 Globulins Proteins 0.000 description 1
- 102100022624 Glucoamylase Human genes 0.000 description 1
- SXRSQZLOMIGNAQ-UHFFFAOYSA-N Glutaraldehyde Chemical compound O=CCCCC=O SXRSQZLOMIGNAQ-UHFFFAOYSA-N 0.000 description 1
- 102000004269 Granulocyte Colony-Stimulating Factor Human genes 0.000 description 1
- 108010017080 Granulocyte Colony-Stimulating Factor Proteins 0.000 description 1
- 229940122853 Growth hormone antagonist Drugs 0.000 description 1
- 208000032843 Hemorrhage Diseases 0.000 description 1
- 206010019909 Hernia Diseases 0.000 description 1
- 102000007625 Hirudins Human genes 0.000 description 1
- 108010007267 Hirudins Proteins 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000911390 Homo sapiens Coagulation factor VIII Proteins 0.000 description 1
- 101000599951 Homo sapiens Insulin-like growth factor I Proteins 0.000 description 1
- 108010001336 Horseradish Peroxidase Proteins 0.000 description 1
- WOBHKFSMXKNTIM-UHFFFAOYSA-N Hydroxyethyl methacrylate Chemical compound CC(=C)C(=O)OCCO WOBHKFSMXKNTIM-UHFFFAOYSA-N 0.000 description 1
- WTDHULULXKLSOZ-UHFFFAOYSA-N Hydroxylamine hydrochloride Chemical compound Cl.ON WTDHULULXKLSOZ-UHFFFAOYSA-N 0.000 description 1
- XQFRJNBWHJMXHO-RRKCRQDMSA-N IDUR Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(I)=C1 XQFRJNBWHJMXHO-RRKCRQDMSA-N 0.000 description 1
- HEFNNWSXXWATRW-UHFFFAOYSA-N Ibuprofen Chemical compound CC(C)CC1=CC=C(C(C)C(O)=O)C=C1 HEFNNWSXXWATRW-UHFFFAOYSA-N 0.000 description 1
- XDXDZDZNSLXDNA-TZNDIEGXSA-N Idarubicin Chemical compound C1[C@H](N)[C@H](O)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2C[C@@](O)(C(C)=O)C1 XDXDZDZNSLXDNA-TZNDIEGXSA-N 0.000 description 1
- XDXDZDZNSLXDNA-UHFFFAOYSA-N Idarubicin Natural products C1C(N)C(O)C(C)OC1OC1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2CC(O)(C(C)=O)C1 XDXDZDZNSLXDNA-UHFFFAOYSA-N 0.000 description 1
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 1
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 1
- 102100026818 Inhibin beta E chain Human genes 0.000 description 1
- 108010004250 Inhibins Proteins 0.000 description 1
- 108090000723 Insulin-Like Growth Factor I Proteins 0.000 description 1
- 102100037852 Insulin-like growth factor I Human genes 0.000 description 1
- 208000035478 Interatrial communication Diseases 0.000 description 1
- 102000000589 Interleukin-1 Human genes 0.000 description 1
- 108010002352 Interleukin-1 Proteins 0.000 description 1
- 102100039068 Interleukin-10 Human genes 0.000 description 1
- 108090000174 Interleukin-10 Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- NYMGNSNKLVNMIA-UHFFFAOYSA-N Iproniazid Chemical compound CC(C)NNC(=O)C1=CC=NC=C1 NYMGNSNKLVNMIA-UHFFFAOYSA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 1
- 108010059881 Lactase Proteins 0.000 description 1
- 108090001090 Lectins Proteins 0.000 description 1
- 102000004856 Lectins Human genes 0.000 description 1
- NNJVILVZKWQKPM-UHFFFAOYSA-N Lidocaine Chemical compound CCN(CC)CC(=O)NC1=C(C)C=CC=C1C NNJVILVZKWQKPM-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- SBDNJUWAMKYJOX-UHFFFAOYSA-N Meclofenamic Acid Chemical compound CC1=CC=C(Cl)C(NC=2C(=CC=CC=2)C(O)=O)=C1Cl SBDNJUWAMKYJOX-UHFFFAOYSA-N 0.000 description 1
- 102000012750 Membrane Glycoproteins Human genes 0.000 description 1
- 108010090054 Membrane Glycoproteins Proteins 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 1
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 1
- 229930192392 Mitomycin Natural products 0.000 description 1
- 229920000881 Modified starch Polymers 0.000 description 1
- 239000004368 Modified starch Substances 0.000 description 1
- PCZOHLXUXFIOCF-UHFFFAOYSA-N Monacolin X Natural products C12C(OC(=O)C(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 PCZOHLXUXFIOCF-UHFFFAOYSA-N 0.000 description 1
- 102000014962 Monocyte Chemoattractant Proteins Human genes 0.000 description 1
- 108010064136 Monocyte Chemoattractant Proteins Proteins 0.000 description 1
- 101710135898 Myc proto-oncogene protein Proteins 0.000 description 1
- 102100038895 Myc proto-oncogene protein Human genes 0.000 description 1
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical compound C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 description 1
- 101001055320 Myxine glutinosa Insulin-like growth factor Proteins 0.000 description 1
- BLXXJMDCKKHMKV-UHFFFAOYSA-N Nabumetone Chemical compound C1=C(CCC(C)=O)C=CC2=CC(OC)=CC=C21 BLXXJMDCKKHMKV-UHFFFAOYSA-N 0.000 description 1
- CMWTZPSULFXXJA-UHFFFAOYSA-N Naproxen Natural products C1=C(C(C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-UHFFFAOYSA-N 0.000 description 1
- 229930193140 Neomycin Natural products 0.000 description 1
- 108010025020 Nerve Growth Factor Proteins 0.000 description 1
- 102000015336 Nerve Growth Factor Human genes 0.000 description 1
- 208000012902 Nervous system disease Diseases 0.000 description 1
- 208000025966 Neurological disease Diseases 0.000 description 1
- MWUXSHHQAYIFBG-UHFFFAOYSA-N Nitric oxide Chemical class O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 1
- 229940123134 Nitric oxide inhibitor Drugs 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 229920000299 Nylon 12 Polymers 0.000 description 1
- 229920002292 Nylon 6 Polymers 0.000 description 1
- 229920002302 Nylon 6,6 Polymers 0.000 description 1
- DQBGMRIIUUTEBH-UHFFFAOYSA-N O=C(Cl)CCCCCN1C(=O)C=CC1=O.O=C=NCCCCCN1C(=O)C=CC1=O.[N-]=[N+]=NC(=O)CCCCCN1C(=O)C=CC1=O Chemical compound O=C(Cl)CCCCCN1C(=O)C=CC1=O.O=C=NCCCCCN1C(=O)C=CC1=O.[N-]=[N+]=NC(=O)CCCCCN1C(=O)C=CC1=O DQBGMRIIUUTEBH-UHFFFAOYSA-N 0.000 description 1
- KCVVGPZDBGAZTL-YSMBQZINSA-N O=C(O)/C=C\C(=O)NCCCCCC(=O)O.O=C(O)CCCCCN1C(=O)C=CC1=O Chemical compound O=C(O)/C=C\C(=O)NCCCCCC(=O)O.O=C(O)CCCCCN1C(=O)C=CC1=O KCVVGPZDBGAZTL-YSMBQZINSA-N 0.000 description 1
- 241000282943 Odocoileus Species 0.000 description 1
- 108010035766 P-Selectin Proteins 0.000 description 1
- 102100023472 P-selectin Human genes 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 229930012538 Paclitaxel Natural products 0.000 description 1
- 102000001746 Pancreatic alpha-Amylases Human genes 0.000 description 1
- 108010029785 Pancreatic alpha-Amylases Proteins 0.000 description 1
- 101000882917 Penaeus paulensis Hemolymph clottable protein Proteins 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 102000015439 Phospholipases Human genes 0.000 description 1
- 108010064785 Phospholipases Proteins 0.000 description 1
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical group OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 108010001014 Plasminogen Activators Proteins 0.000 description 1
- 102000001938 Plasminogen Activators Human genes 0.000 description 1
- 102000023159 Platelet Glycoprotein GPIb-IX Complex Human genes 0.000 description 1
- 108010045766 Platelet Glycoprotein GPIb-IX Complex Proteins 0.000 description 1
- 102100036154 Platelet basic protein Human genes 0.000 description 1
- 101710195957 Platelet basic protein Proteins 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 229920002732 Polyanhydride Polymers 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920000954 Polyglycolide Polymers 0.000 description 1
- 229920000331 Polyhydroxybutyrate Polymers 0.000 description 1
- 229920001710 Polyorthoester Polymers 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 229920003082 Povidone K 90 Polymers 0.000 description 1
- TUZYXOIXSAXUGO-UHFFFAOYSA-N Pravastatin Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(O)C=C21 TUZYXOIXSAXUGO-UHFFFAOYSA-N 0.000 description 1
- 208000024777 Prion disease Diseases 0.000 description 1
- 101710184309 Probable sucrose-6-phosphate hydrolase Proteins 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000004403 Prostatic Hyperplasia Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 108010009736 Protein Hydrolysates Proteins 0.000 description 1
- 102100026827 Protein associated with UVRAG as autophagy enhancer Human genes 0.000 description 1
- 101710102978 Protein associated with UVRAG as autophagy enhancer Proteins 0.000 description 1
- 108010094028 Prothrombin Proteins 0.000 description 1
- 102100027378 Prothrombin Human genes 0.000 description 1
- 241000125945 Protoparvovirus Species 0.000 description 1
- 206010037660 Pyrexia Diseases 0.000 description 1
- 208000017442 Retinal disease Diseases 0.000 description 1
- CZMRCDWAGMRECN-UHFFFAOYSA-N Rohrzucker Natural products OCC1OC(CO)(OC2OC(CO)C(O)C(O)C2O)C(O)C1O CZMRCDWAGMRECN-UHFFFAOYSA-N 0.000 description 1
- RYMZZMVNJRMUDD-UHFFFAOYSA-N SJ000286063 Natural products C12C(OC(=O)C(C)(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 RYMZZMVNJRMUDD-UHFFFAOYSA-N 0.000 description 1
- SKZKKFZAGNVIMN-UHFFFAOYSA-N Salicilamide Chemical compound NC(=O)C1=CC=CC=C1O SKZKKFZAGNVIMN-UHFFFAOYSA-N 0.000 description 1
- 102000013523 Salivary alpha-Amylases Human genes 0.000 description 1
- 108010026386 Salivary alpha-Amylases Proteins 0.000 description 1
- XHQYBDSXTDXSHY-UHFFFAOYSA-N Semicarbazide hydrochloride Chemical compound Cl.NNC(N)=O XHQYBDSXTDXSHY-UHFFFAOYSA-N 0.000 description 1
- 208000012936 Sheep disease Diseases 0.000 description 1
- 229910052581 Si3N4 Inorganic materials 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- 108091027967 Small hairpin RNA Proteins 0.000 description 1
- 108020004459 Small interfering RNA Proteins 0.000 description 1
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- 102000013275 Somatomedins Human genes 0.000 description 1
- 108010090804 Streptavidin Proteins 0.000 description 1
- 108010023197 Streptokinase Proteins 0.000 description 1
- 102400000472 Sucrase Human genes 0.000 description 1
- 101710112652 Sucrose-6-phosphate hydrolase Proteins 0.000 description 1
- 208000005400 Synovial Cyst Diseases 0.000 description 1
- 210000001744 T-lymphocyte Anatomy 0.000 description 1
- QJJXYPPXXYFBGM-LFZNUXCKSA-N Tacrolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1\C=C(/C)[C@@H]1[C@H](C)[C@@H](O)CC(=O)[C@H](CC=C)/C=C(C)/C[C@H](C)C[C@H](OC)[C@H]([C@H](C[C@H]2C)OC)O[C@@]2(O)C(=O)C(=O)N2CCCC[C@H]2C(=O)O1 QJJXYPPXXYFBGM-LFZNUXCKSA-N 0.000 description 1
- 229940123237 Taxane Drugs 0.000 description 1
- 239000004809 Teflon Substances 0.000 description 1
- 229920006362 Teflon® Polymers 0.000 description 1
- 239000004098 Tetracycline Substances 0.000 description 1
- 101000712605 Theromyzon tessulatum Theromin Proteins 0.000 description 1
- 108090000190 Thrombin Proteins 0.000 description 1
- 229940122388 Thrombin inhibitor Drugs 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 101710120037 Toxin CcdB Proteins 0.000 description 1
- 101710150448 Transcriptional regulator Myc Proteins 0.000 description 1
- 102000004887 Transforming Growth Factor beta Human genes 0.000 description 1
- 108090001012 Transforming Growth Factor beta Proteins 0.000 description 1
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 description 1
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 1
- 102000003990 Urokinase-type plasminogen activator Human genes 0.000 description 1
- 108090000435 Urokinase-type plasminogen activator Proteins 0.000 description 1
- 241000700618 Vaccinia virus Species 0.000 description 1
- 108010059993 Vancomycin Proteins 0.000 description 1
- 108010073929 Vascular Endothelial Growth Factor A Proteins 0.000 description 1
- 206010047163 Vasospasm Diseases 0.000 description 1
- OIRDTQYFTABQOQ-UHTZMRCNSA-N Vidarabine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1O OIRDTQYFTABQOQ-UHTZMRCNSA-N 0.000 description 1
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 description 1
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- IUHMWLMDASQDJQ-ZBFHGGJFSA-N [(3ar,8bs)-3,4,8b-trimethyl-2,3a-dihydro-1h-pyrrolo[2,3-b]indol-7-yl] n,n-dimethylcarbamate Chemical compound C12=CC(OC(=O)N(C)C)=CC=C2N(C)[C@@H]2[C@@]1(C)CCN2C IUHMWLMDASQDJQ-ZBFHGGJFSA-N 0.000 description 1
- YYBNDIVPHIWTPK-KYJQVDHRSA-N [(3as,8bs)-3,4,8b-trimethyl-1,2,3,3a-tetrahydropyrrolo[2,3-b]indol-3-ium-7-yl] n-methylcarbamate;sulfate Chemical compound [O-]S([O-])(=O)=O.C12=CC(OC(=O)NC)=CC=C2N(C)[C@@H]2[C@@]1(C)CC[NH+]2C.C12=CC(OC(=O)NC)=CC=C2N(C)[C@@H]2[C@@]1(C)CC[NH+]2C YYBNDIVPHIWTPK-KYJQVDHRSA-N 0.000 description 1
- WAIPAZQMEIHHTJ-UHFFFAOYSA-N [Cr].[Co] Chemical compound [Cr].[Co] WAIPAZQMEIHHTJ-UHFFFAOYSA-N 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- RAUARFJXOYWAHD-JEDNCBNOSA-N acetic acid;(2s)-5-(diaminomethylideneamino)-2-(methylamino)pentanoic acid Chemical compound CC(O)=O.CN[C@H](C(O)=O)CCCNC(N)=N RAUARFJXOYWAHD-JEDNCBNOSA-N 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 229960001138 acetylsalicylic acid Drugs 0.000 description 1
- 238000005903 acid hydrolysis reaction Methods 0.000 description 1
- 229920006397 acrylic thermoplastic Polymers 0.000 description 1
- 231100000764 actin inhibitor Toxicity 0.000 description 1
- RJURFGZVJUQBHK-IIXSONLDSA-N actinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-IIXSONLDSA-N 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 239000000488 activin Substances 0.000 description 1
- 125000002252 acyl group Chemical group 0.000 description 1
- 238000012644 addition polymerization Methods 0.000 description 1
- 230000001464 adherent effect Effects 0.000 description 1
- 239000001361 adipic acid Substances 0.000 description 1
- 235000011037 adipic acid Nutrition 0.000 description 1
- 210000001789 adipocyte Anatomy 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 230000006838 adverse reaction Effects 0.000 description 1
- 108010061456 afaacytin Proteins 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 108010033320 aggregoserpentin Proteins 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 238000007605 air drying Methods 0.000 description 1
- 229940050528 albumin Drugs 0.000 description 1
- 229960000548 alemtuzumab Drugs 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 125000005250 alkyl acrylate group Chemical group 0.000 description 1
- OFCNXPDARWKPPY-UHFFFAOYSA-N allopurinol Chemical compound OC1=NC=NC2=C1C=NN2 OFCNXPDARWKPPY-UHFFFAOYSA-N 0.000 description 1
- 229960003459 allopurinol Drugs 0.000 description 1
- 108010028144 alpha-Glucosidases Proteins 0.000 description 1
- 102000016679 alpha-Glucosidases Human genes 0.000 description 1
- 150000001371 alpha-amino acids Chemical class 0.000 description 1
- 235000008206 alpha-amino acids Nutrition 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- DKNWSYNQZKUICI-UHFFFAOYSA-N amantadine Chemical compound C1C(C2)CC3CC2CC1(N)C3 DKNWSYNQZKUICI-UHFFFAOYSA-N 0.000 description 1
- 229960003805 amantadine Drugs 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 125000004103 aminoalkyl group Chemical group 0.000 description 1
- 229960002684 aminocaproic acid Drugs 0.000 description 1
- 229920000469 amphiphilic block copolymer Polymers 0.000 description 1
- 230000003444 anaesthetic effect Effects 0.000 description 1
- 229940035676 analgesics Drugs 0.000 description 1
- 239000004037 angiogenesis inhibitor Substances 0.000 description 1
- 239000002333 angiotensin II receptor antagonist Substances 0.000 description 1
- 229940125364 angiotensin receptor blocker Drugs 0.000 description 1
- 229940044094 angiotensin-converting-enzyme inhibitor Drugs 0.000 description 1
- 239000000730 antalgic agent Substances 0.000 description 1
- 229940045799 anthracyclines and related substance Drugs 0.000 description 1
- RJGDLRCDCYRQOQ-UHFFFAOYSA-N anthrone Chemical compound C1=CC=C2C(=O)C3=CC=CC=C3CC2=C1 RJGDLRCDCYRQOQ-UHFFFAOYSA-N 0.000 description 1
- 230000001093 anti-cancer Effects 0.000 description 1
- 230000001078 anti-cholinergic effect Effects 0.000 description 1
- 230000001773 anti-convulsant effect Effects 0.000 description 1
- 230000001430 anti-depressive effect Effects 0.000 description 1
- 230000003474 anti-emetic effect Effects 0.000 description 1
- 230000001384 anti-glaucoma Effects 0.000 description 1
- 230000003276 anti-hypertensive effect Effects 0.000 description 1
- 230000000340 anti-metabolite Effects 0.000 description 1
- 230000002927 anti-mitotic effect Effects 0.000 description 1
- 230000003064 anti-oxidating effect Effects 0.000 description 1
- 230000002141 anti-parasite Effects 0.000 description 1
- 230000000648 anti-parkinson Effects 0.000 description 1
- 230000000702 anti-platelet effect Effects 0.000 description 1
- 230000000842 anti-protozoal effect Effects 0.000 description 1
- 230000000561 anti-psychotic effect Effects 0.000 description 1
- 230000002769 anti-restenotic effect Effects 0.000 description 1
- 230000000692 anti-sense effect Effects 0.000 description 1
- 230000002921 anti-spasmodic effect Effects 0.000 description 1
- 229940065524 anticholinergics inhalants for obstructive airway diseases Drugs 0.000 description 1
- 229940125681 anticonvulsant agent Drugs 0.000 description 1
- 239000001961 anticonvulsive agent Substances 0.000 description 1
- 239000000935 antidepressant agent Substances 0.000 description 1
- 229940005513 antidepressants Drugs 0.000 description 1
- 239000002111 antiemetic agent Substances 0.000 description 1
- 229940125683 antiemetic agent Drugs 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 229940125715 antihistaminic agent Drugs 0.000 description 1
- 239000000739 antihistaminic agent Substances 0.000 description 1
- 229940100197 antimetabolite Drugs 0.000 description 1
- 239000002256 antimetabolite Substances 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 239000003080 antimitotic agent Substances 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 229940045719 antineoplastic alkylating agent nitrosoureas Drugs 0.000 description 1
- 239000000939 antiparkinson agent Substances 0.000 description 1
- 239000003904 antiprotozoal agent Substances 0.000 description 1
- 239000000074 antisense oligonucleotide Substances 0.000 description 1
- 238000012230 antisense oligonucleotides Methods 0.000 description 1
- 229940124575 antispasmodic agent Drugs 0.000 description 1
- 239000004019 antithrombin Substances 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 239000000010 aprotic solvent Substances 0.000 description 1
- KXNPVXPOPUZYGB-XYVMCAHJSA-N argatroban Chemical compound OC(=O)[C@H]1C[C@H](C)CCN1C(=O)[C@H](CCCN=C(N)N)NS(=O)(=O)C1=CC=CC2=C1NC[C@H](C)C2 KXNPVXPOPUZYGB-XYVMCAHJSA-N 0.000 description 1
- 229960003856 argatroban Drugs 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 210000001130 astrocyte Anatomy 0.000 description 1
- 229960005370 atorvastatin Drugs 0.000 description 1
- 230000001746 atrial effect Effects 0.000 description 1
- 208000013914 atrial heart septal defect Diseases 0.000 description 1
- 206010003664 atrial septal defect Diseases 0.000 description 1
- 230000002238 attenuated effect Effects 0.000 description 1
- 230000003190 augmentative effect Effects 0.000 description 1
- AUJRCFUBUPVWSZ-XTZHGVARSA-M auranofin Chemical compound CCP(CC)(CC)=[Au]S[C@@H]1O[C@H](COC(C)=O)[C@@H](OC(C)=O)[C@H](OC(C)=O)[C@H]1OC(C)=O AUJRCFUBUPVWSZ-XTZHGVARSA-M 0.000 description 1
- 229960005207 auranofin Drugs 0.000 description 1
- 229960001799 aurothioglucose Drugs 0.000 description 1
- LMEKQMALGUDUQG-UHFFFAOYSA-N azathioprine Chemical compound CN1C=NC([N+]([O-])=O)=C1SC1=NC=NC2=C1NC=N2 LMEKQMALGUDUQG-UHFFFAOYSA-N 0.000 description 1
- 229960002170 azathioprine Drugs 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 229960003071 bacitracin Drugs 0.000 description 1
- 229930184125 bacitracin Natural products 0.000 description 1
- CLKOFPXJLQSYAH-ABRJDSQDSA-N bacitracin A Chemical compound C1SC([C@@H](N)[C@@H](C)CC)=N[C@@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]1C(=O)N[C@H](CCCN)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2N=CNC=2)C(=O)N[C@H](CC(O)=O)C(=O)N[C@@H](CC(N)=O)C(=O)NCCCC1 CLKOFPXJLQSYAH-ABRJDSQDSA-N 0.000 description 1
- 210000003651 basophil Anatomy 0.000 description 1
- 235000015278 beef Nutrition 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- WRUAHXANJKHFIL-UHFFFAOYSA-N benzene-1,3-disulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC(S(O)(=O)=O)=C1 WRUAHXANJKHFIL-UHFFFAOYSA-N 0.000 description 1
- 235000019400 benzoyl peroxide Nutrition 0.000 description 1
- 108010005774 beta-Galactosidase Proteins 0.000 description 1
- 229960002537 betamethasone Drugs 0.000 description 1
- UREBDLICKHMUKA-DVTGEIKXSA-N betamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-DVTGEIKXSA-N 0.000 description 1
- 229960000397 bevacizumab Drugs 0.000 description 1
- 238000002306 biochemical method Methods 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 229960002685 biotin Drugs 0.000 description 1
- 235000020958 biotin Nutrition 0.000 description 1
- 239000011616 biotin Substances 0.000 description 1
- LBSPZZSGTIBOFG-UHFFFAOYSA-N bis[2-(4,5-dihydro-1h-imidazol-2-yl)propan-2-yl]diazene;dihydrochloride Chemical compound Cl.Cl.N=1CCNC=1C(C)(C)N=NC(C)(C)C1=NCCN1 LBSPZZSGTIBOFG-UHFFFAOYSA-N 0.000 description 1
- 238000004061 bleaching Methods 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- OYVAGSVQBOHSSS-UAPAGMARSA-O bleomycin A2 Chemical class N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1N=CNC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C OYVAGSVQBOHSSS-UAPAGMARSA-O 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 230000023555 blood coagulation Effects 0.000 description 1
- 239000003114 blood coagulation factor Substances 0.000 description 1
- 210000001772 blood platelet Anatomy 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 229960002092 busulfan Drugs 0.000 description 1
- ABBZJHFBQXYTLU-UHFFFAOYSA-N but-3-enamide Chemical compound NC(=O)CC=C ABBZJHFBQXYTLU-UHFFFAOYSA-N 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000000480 calcium channel blocker Substances 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 229960004205 carbidopa Drugs 0.000 description 1
- TZFNLOMSOLWIDK-JTQLQIEISA-N carbidopa (anhydrous) Chemical compound NN[C@@](C(O)=O)(C)CC1=CC=C(O)C(O)=C1 TZFNLOMSOLWIDK-JTQLQIEISA-N 0.000 description 1
- 229960001631 carbomer Drugs 0.000 description 1
- 235000011089 carbon dioxide Nutrition 0.000 description 1
- YAYRGNWWLMLWJE-UHFFFAOYSA-L carboplatin Chemical compound O=C1O[Pt](N)(N)OC(=O)C11CCC1 YAYRGNWWLMLWJE-UHFFFAOYSA-L 0.000 description 1
- 229960004562 carboplatin Drugs 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 125000002057 carboxymethyl group Chemical group [H]OC(=O)C([H])([H])[*] 0.000 description 1
- 210000004413 cardiac myocyte Anatomy 0.000 description 1
- 229960005243 carmustine Drugs 0.000 description 1
- 238000005266 casting Methods 0.000 description 1
- QYIYFLOTGYLRGG-GPCCPHFNSA-N cefaclor Chemical compound C1([C@H](C(=O)N[C@@H]2C(N3C(=C(Cl)CS[C@@H]32)C(O)=O)=O)N)=CC=CC=C1 QYIYFLOTGYLRGG-GPCCPHFNSA-N 0.000 description 1
- 229960005361 cefaclor Drugs 0.000 description 1
- 229960004841 cefadroxil Drugs 0.000 description 1
- NBFNMSULHIODTC-CYJZLJNKSA-N cefadroxil monohydrate Chemical compound O.C1([C@@H](N)C(=O)N[C@H]2[C@@H]3N(C2=O)C(=C(CS3)C)C(O)=O)=CC=C(O)C=C1 NBFNMSULHIODTC-CYJZLJNKSA-N 0.000 description 1
- 229960000603 cefalotin Drugs 0.000 description 1
- XIURVHNZVLADCM-IUODEOHRSA-N cefalotin Chemical compound N([C@H]1[C@@H]2N(C1=O)C(=C(CS2)COC(=O)C)C(O)=O)C(=O)CC1=CC=CS1 XIURVHNZVLADCM-IUODEOHRSA-N 0.000 description 1
- 229960003012 cefamandole Drugs 0.000 description 1
- OLVCFLKTBJRLHI-AXAPSJFSSA-N cefamandole Chemical compound CN1N=NN=C1SCC1=C(C(O)=O)N2C(=O)[C@@H](NC(=O)[C@H](O)C=3C=CC=CC=3)[C@H]2SC1 OLVCFLKTBJRLHI-AXAPSJFSSA-N 0.000 description 1
- 229960004350 cefapirin Drugs 0.000 description 1
- 229960001139 cefazolin Drugs 0.000 description 1
- MLYYVTUWGNIJIB-BXKDBHETSA-N cefazolin Chemical compound S1C(C)=NN=C1SCC1=C(C(O)=O)N2C(=O)[C@@H](NC(=O)CN3N=NN=C3)[C@H]2SC1 MLYYVTUWGNIJIB-BXKDBHETSA-N 0.000 description 1
- DYAIAHUQIPBDIP-AXAPSJFSSA-N cefonicid Chemical compound S([C@@H]1[C@@H](C(N1C=1C(O)=O)=O)NC(=O)[C@H](O)C=2C=CC=CC=2)CC=1CSC1=NN=NN1CS(O)(=O)=O DYAIAHUQIPBDIP-AXAPSJFSSA-N 0.000 description 1
- 229960004489 cefonicid Drugs 0.000 description 1
- GCFBRXLSHGKWDP-XCGNWRKASA-N cefoperazone Chemical compound O=C1C(=O)N(CC)CCN1C(=O)N[C@H](C=1C=CC(O)=CC=1)C(=O)N[C@@H]1C(=O)N2C(C(O)=O)=C(CSC=3N(N=NN=3)C)CS[C@@H]21 GCFBRXLSHGKWDP-XCGNWRKASA-N 0.000 description 1
- 229960004682 cefoperazone Drugs 0.000 description 1
- SLAYUXIURFNXPG-CRAIPNDOSA-N ceforanide Chemical compound NCC1=CC=CC=C1CC(=O)N[C@@H]1C(=O)N2C(C(O)=O)=C(CSC=3N(N=NN=3)CC(O)=O)CS[C@@H]21 SLAYUXIURFNXPG-CRAIPNDOSA-N 0.000 description 1
- 229960004292 ceforanide Drugs 0.000 description 1
- 229960004261 cefotaxime Drugs 0.000 description 1
- GPRBEKHLDVQUJE-VINNURBNSA-N cefotaxime Chemical compound N([C@@H]1C(N2C(=C(COC(C)=O)CS[C@@H]21)C(O)=O)=O)C(=O)/C(=N/OC)C1=CSC(N)=N1 GPRBEKHLDVQUJE-VINNURBNSA-N 0.000 description 1
- WZOZEZRFJCJXNZ-ZBFHGGJFSA-N cefoxitin Chemical compound N([C@]1(OC)C(N2C(=C(COC(N)=O)CS[C@@H]21)C(O)=O)=O)C(=O)CC1=CC=CS1 WZOZEZRFJCJXNZ-ZBFHGGJFSA-N 0.000 description 1
- 229960002682 cefoxitin Drugs 0.000 description 1
- 229960002588 cefradine Drugs 0.000 description 1
- 229960001991 ceftizoxime Drugs 0.000 description 1
- NNULBSISHYWZJU-LLKWHZGFSA-N ceftizoxime Chemical compound N([C@@H]1C(N2C(=CCS[C@@H]21)C(O)=O)=O)C(=O)\C(=N/OC)C1=CSC(N)=N1 NNULBSISHYWZJU-LLKWHZGFSA-N 0.000 description 1
- 229960004755 ceftriaxone Drugs 0.000 description 1
- VAAUVRVFOQPIGI-SPQHTLEESA-N ceftriaxone Chemical compound S([C@@H]1[C@@H](C(N1C=1C(O)=O)=O)NC(=O)\C(=N/OC)C=2N=C(N)SC=2)CC=1CSC1=NC(=O)C(=O)NN1C VAAUVRVFOQPIGI-SPQHTLEESA-N 0.000 description 1
- 229960001668 cefuroxime Drugs 0.000 description 1
- JFPVXVDWJQMJEE-IZRZKJBUSA-N cefuroxime Chemical compound N([C@@H]1C(N2C(=C(COC(N)=O)CS[C@@H]21)C(O)=O)=O)C(=O)\C(=N/OC)C1=CC=CO1 JFPVXVDWJQMJEE-IZRZKJBUSA-N 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 102000008373 cell-cell adhesion mediator activity proteins Human genes 0.000 description 1
- 108040002566 cell-cell adhesion mediator activity proteins Proteins 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 229940106164 cephalexin Drugs 0.000 description 1
- ZAIPMKNFIOOWCQ-UEKVPHQBSA-N cephalexin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@@H]3N(C2=O)C(=C(CS3)C)C(O)=O)=CC=CC=C1 ZAIPMKNFIOOWCQ-UEKVPHQBSA-N 0.000 description 1
- RDLPVSKMFDYCOR-UEKVPHQBSA-N cephradine Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@@H]3N(C2=O)C(=C(CS3)C)C(O)=O)=CCC=CC1 RDLPVSKMFDYCOR-UEKVPHQBSA-N 0.000 description 1
- 229960005110 cerivastatin Drugs 0.000 description 1
- SEERZIQQUAZTOL-ANMDKAQQSA-N cerivastatin Chemical compound COCC1=C(C(C)C)N=C(C(C)C)C(\C=C\[C@@H](O)C[C@@H](O)CC(O)=O)=C1C1=CC=C(F)C=C1 SEERZIQQUAZTOL-ANMDKAQQSA-N 0.000 description 1
- 229960005395 cetuximab Drugs 0.000 description 1
- 239000012986 chain transfer agent Substances 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- JCKYGMPEJWAADB-UHFFFAOYSA-N chlorambucil Chemical compound OC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 JCKYGMPEJWAADB-UHFFFAOYSA-N 0.000 description 1
- 229960004630 chlorambucil Drugs 0.000 description 1
- 229960005091 chloramphenicol Drugs 0.000 description 1
- WIIZWVCIJKGZOK-RKDXNWHRSA-N chloramphenicol Chemical compound ClC(Cl)C(=O)N[C@H](CO)[C@H](O)C1=CC=C([N+]([O-])=O)C=C1 WIIZWVCIJKGZOK-RKDXNWHRSA-N 0.000 description 1
- 229960003260 chlorhexidine Drugs 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 239000000812 cholinergic antagonist Substances 0.000 description 1
- 210000001612 chondrocyte Anatomy 0.000 description 1
- 210000003161 choroid Anatomy 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 208000017580 chronic wasting disease Diseases 0.000 description 1
- 229960001265 ciclosporin Drugs 0.000 description 1
- 229960001747 cinchocaine Drugs 0.000 description 1
- PUFQVTATUTYEAL-UHFFFAOYSA-N cinchocaine Chemical compound C1=CC=CC2=NC(OCCCC)=CC(C(=O)NCCN(CC)CC)=C21 PUFQVTATUTYEAL-UHFFFAOYSA-N 0.000 description 1
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 description 1
- 229960004316 cisplatin Drugs 0.000 description 1
- 229960003009 clopidogrel Drugs 0.000 description 1
- GKTWGGQPFAXNFI-HNNXBMFYSA-N clopidogrel Chemical compound C1([C@H](N2CC=3C=CSC=3CC2)C(=O)OC)=CC=CC=C1Cl GKTWGGQPFAXNFI-HNNXBMFYSA-N 0.000 description 1
- 230000035602 clotting Effects 0.000 description 1
- 239000003426 co-catalyst Substances 0.000 description 1
- 230000015271 coagulation Effects 0.000 description 1
- 238000005345 coagulation Methods 0.000 description 1
- 238000013034 coating degradation Methods 0.000 description 1
- 239000010952 cobalt-chrome Substances 0.000 description 1
- 229960001338 colchicine Drugs 0.000 description 1
- OHCQJHSOBUTRHG-UHFFFAOYSA-N colforsin Natural products OC12C(=O)CC(C)(C=C)OC1(C)C(OC(=O)C)C(O)C1C2(C)C(O)CCC1(C)C OHCQJHSOBUTRHG-UHFFFAOYSA-N 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940125782 compound 2 Drugs 0.000 description 1
- 229940126214 compound 3 Drugs 0.000 description 1
- 229940125898 compound 5 Drugs 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 108010089485 convulxin Proteins 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 229940099112 cornstarch Drugs 0.000 description 1
- 238000007887 coronary angioplasty Methods 0.000 description 1
- 210000004351 coronary vessel Anatomy 0.000 description 1
- 229960004544 cortisone Drugs 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 229960004397 cyclophosphamide Drugs 0.000 description 1
- 229930182912 cyclosporin Natural products 0.000 description 1
- 229960000684 cytarabine Drugs 0.000 description 1
- 229940127089 cytotoxic agent Drugs 0.000 description 1
- 229960000640 dactinomycin Drugs 0.000 description 1
- 229960000975 daunorubicin Drugs 0.000 description 1
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000032798 delamination Effects 0.000 description 1
- 210000001787 dendrite Anatomy 0.000 description 1
- CFCUWKMKBJTWLW-UHFFFAOYSA-N deoliosyl-3C-alpha-L-digitoxosyl-MTM Natural products CC=1C(O)=C2C(O)=C3C(=O)C(OC4OC(C)C(O)C(OC5OC(C)C(O)C(OC6OC(C)C(O)C(C)(O)C6)C5)C4)C(C(OC)C(=O)C(O)C(C)O)CC3=CC2=CC=1OC(OC(C)C1O)CC1OC1CC(O)C(O)C(C)O1 CFCUWKMKBJTWLW-UHFFFAOYSA-N 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000000151 deposition Methods 0.000 description 1
- GPRLSGONYQIRFK-DYCDLGHISA-N deuteron Chemical compound [2H+] GPRLSGONYQIRFK-DYCDLGHISA-N 0.000 description 1
- 229960003957 dexamethasone Drugs 0.000 description 1
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 125000000950 dibromo group Chemical group Br* 0.000 description 1
- 229960001259 diclofenac Drugs 0.000 description 1
- DCOPUUMXTXDBNB-UHFFFAOYSA-N diclofenac Chemical compound OC(=O)CC1=CC=CC=C1NC1=C(Cl)C=CC=C1Cl DCOPUUMXTXDBNB-UHFFFAOYSA-N 0.000 description 1
- GJQPMPFPNINLKP-UHFFFAOYSA-N diclofenamide Chemical compound NS(=O)(=O)C1=CC(Cl)=C(Cl)C(S(N)(=O)=O)=C1 GJQPMPFPNINLKP-UHFFFAOYSA-N 0.000 description 1
- 229960005081 diclofenamide Drugs 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- HUPFGZXOMWLGNK-UHFFFAOYSA-N diflunisal Chemical compound C1=C(O)C(C(=O)O)=CC(C=2C(=CC(F)=CC=2)F)=C1 HUPFGZXOMWLGNK-UHFFFAOYSA-N 0.000 description 1
- 229960000616 diflunisal Drugs 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- FLISWPFVWWWNNP-BQYQJAHWSA-N dihydro-3-(1-octenyl)-2,5-furandione Chemical compound CCCCCC\C=C\C1CC(=O)OC1=O FLISWPFVWWWNNP-BQYQJAHWSA-N 0.000 description 1
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 1
- 229960002768 dipyridamole Drugs 0.000 description 1
- IZEKFCXSFNUWAM-UHFFFAOYSA-N dipyridamole Chemical compound C=12N=C(N(CCO)CCO)N=C(N3CCCCC3)C2=NC(N(CCO)CCO)=NC=1N1CCCCC1 IZEKFCXSFNUWAM-UHFFFAOYSA-N 0.000 description 1
- 238000002224 dissection Methods 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 229940052760 dopamine agonists Drugs 0.000 description 1
- 239000003136 dopamine receptor stimulating agent Substances 0.000 description 1
- 229960004679 doxorubicin Drugs 0.000 description 1
- 229960003722 doxycycline Drugs 0.000 description 1
- 108010085662 ecarin Proteins 0.000 description 1
- 229960001776 edrecolomab Drugs 0.000 description 1
- BXKDSDJJOVIHMX-UHFFFAOYSA-N edrophonium chloride Chemical compound [Cl-].CC[N+](C)(C)C1=CC=CC(O)=C1 BXKDSDJJOVIHMX-UHFFFAOYSA-N 0.000 description 1
- 229960002406 edrophonium chloride Drugs 0.000 description 1
- 230000002900 effect on cell Effects 0.000 description 1
- 230000002828 effect on organs or tissue Effects 0.000 description 1
- 230000001700 effect on tissue Effects 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 230000003073 embolic effect Effects 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000006911 enzymatic reaction Methods 0.000 description 1
- 210000003979 eosinophil Anatomy 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- 229960001123 epoprostenol Drugs 0.000 description 1
- KAQKFAOMNZTLHT-VVUHWYTRSA-N epoprostenol Chemical compound O1C(=CCCCC(O)=O)C[C@@H]2[C@@H](/C=C/[C@@H](O)CCCCC)[C@H](O)C[C@@H]21 KAQKFAOMNZTLHT-VVUHWYTRSA-N 0.000 description 1
- GLGOPUHVAZCPRB-LROMGURASA-N eptifibatide Chemical compound N1C(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H](CCCCNC(=N)N)NC(=O)CCSSC[C@@H](C(N)=O)NC(=O)[C@@H]2CCCN2C(=O)[C@@H]1CC1=CN=C2[C]1C=CC=C2 GLGOPUHVAZCPRB-LROMGURASA-N 0.000 description 1
- 229960004468 eptifibatide Drugs 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 229960003276 erythromycin Drugs 0.000 description 1
- 229940105423 erythropoietin Drugs 0.000 description 1
- 230000032050 esterification Effects 0.000 description 1
- 238000005886 esterification reaction Methods 0.000 description 1
- 229940011871 estrogen Drugs 0.000 description 1
- 239000000262 estrogen Substances 0.000 description 1
- 238000006266 etherification reaction Methods 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229960005167 everolimus Drugs 0.000 description 1
- 210000002744 extracellular matrix Anatomy 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 229960001419 fenoprofen Drugs 0.000 description 1
- 229950003499 fibrin Drugs 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 229940126864 fibroblast growth factor Drugs 0.000 description 1
- 230000003328 fibroblastic effect Effects 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 239000012847 fine chemical Substances 0.000 description 1
- ODKNJVUHOIMIIZ-RRKCRQDMSA-N floxuridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(F)=C1 ODKNJVUHOIMIIZ-RRKCRQDMSA-N 0.000 description 1
- 229960000961 floxuridine Drugs 0.000 description 1
- 229960002011 fludrocortisone Drugs 0.000 description 1
- AAXVEMMRQDVLJB-BULBTXNYSA-N fludrocortisone Chemical compound O=C1CC[C@]2(C)[C@@]3(F)[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 AAXVEMMRQDVLJB-BULBTXNYSA-N 0.000 description 1
- 229960002949 fluorouracil Drugs 0.000 description 1
- 229960003765 fluvastatin Drugs 0.000 description 1
- 230000003325 follicular Effects 0.000 description 1
- 235000013373 food additive Nutrition 0.000 description 1
- 239000002778 food additive Substances 0.000 description 1
- 230000037406 food intake Effects 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- QTQAWLPCGQOSGP-GBTDJJJQSA-N geldanamycin Chemical compound N1C(=O)\C(C)=C/C=C\[C@@H](OC)[C@H](OC(N)=O)\C(C)=C/[C@@H](C)[C@@H](O)[C@H](OC)C[C@@H](C)CC2=C(OC)C(=O)C=C1C2=O QTQAWLPCGQOSGP-GBTDJJJQSA-N 0.000 description 1
- 229960000578 gemtuzumab Drugs 0.000 description 1
- 229960003297 gemtuzumab ozogamicin Drugs 0.000 description 1
- 125000005640 glucopyranosyl group Chemical group 0.000 description 1
- 229960000587 glutaral Drugs 0.000 description 1
- 229940125672 glycoprotein IIb/IIIa inhibitor Drugs 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 229940015045 gold sodium thiomalate Drugs 0.000 description 1
- 238000010559 graft polymerization reaction Methods 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 230000003394 haemopoietic effect Effects 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 231100001261 hazardous Toxicity 0.000 description 1
- 210000002837 heart atrium Anatomy 0.000 description 1
- 210000003709 heart valve Anatomy 0.000 description 1
- 208000014951 hematologic disease Diseases 0.000 description 1
- 230000023597 hemostasis Effects 0.000 description 1
- ZFGMDIBRIDKWMY-PASTXAENSA-N heparin Chemical compound CC(O)=N[C@@H]1[C@@H](O)[C@H](O)[C@@H](COS(O)(=O)=O)O[C@@H]1O[C@@H]1[C@@H](C(O)=O)O[C@@H](O[C@H]2[C@@H]([C@@H](OS(O)(=O)=O)[C@@H](O[C@@H]3[C@@H](OC(O)[C@H](OS(O)(=O)=O)[C@H]3O)C(O)=O)O[C@@H]2O)CS(O)(=O)=O)[C@H](O)[C@H]1O ZFGMDIBRIDKWMY-PASTXAENSA-N 0.000 description 1
- 239000002628 heparin derivative Substances 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 229940006607 hirudin Drugs 0.000 description 1
- 210000003630 histaminocyte Anatomy 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 229960002474 hydralazine Drugs 0.000 description 1
- ZUXNZUWOTSUBMN-UHFFFAOYSA-N hydralazine hydrochloride Chemical compound Cl.C1=CC=C2C(NN)=NN=CC2=C1 ZUXNZUWOTSUBMN-UHFFFAOYSA-N 0.000 description 1
- 208000003906 hydrocephalus Diseases 0.000 description 1
- 229960000890 hydrocortisone Drugs 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 239000000413 hydrolysate Substances 0.000 description 1
- ZUFVXZVXEJHHBN-UHFFFAOYSA-N hydron;1,2,3,4-tetrahydroacridin-9-amine;chloride Chemical compound [Cl-].C1=CC=C2C([NH3+])=C(CCCC3)C3=NC2=C1 ZUFVXZVXEJHHBN-UHFFFAOYSA-N 0.000 description 1
- 150000002433 hydrophilic molecules Chemical class 0.000 description 1
- 239000003326 hypnotic agent Substances 0.000 description 1
- 230000000147 hypnotic effect Effects 0.000 description 1
- 229960001001 ibritumomab tiuxetan Drugs 0.000 description 1
- 229960001680 ibuprofen Drugs 0.000 description 1
- 229960000908 idarubicin Drugs 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 230000008105 immune reaction Effects 0.000 description 1
- 230000007235 immunity generation Effects 0.000 description 1
- 238000003018 immunoassay Methods 0.000 description 1
- 239000002596 immunotoxin Substances 0.000 description 1
- 230000002637 immunotoxin Effects 0.000 description 1
- 231100000608 immunotoxin Toxicity 0.000 description 1
- 229940051026 immunotoxin Drugs 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000010952 in-situ formation Methods 0.000 description 1
- 230000000415 inactivating effect Effects 0.000 description 1
- 229960004187 indoprofen Drugs 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 230000028709 inflammatory response Effects 0.000 description 1
- 239000000893 inhibin Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- ZPNFWUPYTFPOJU-LPYSRVMUSA-N iniprol Chemical compound C([C@H]1C(=O)NCC(=O)NCC(=O)N[C@H]2CSSC[C@H]3C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](C(N[C@H](C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC=4C=CC=CC=4)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC=4C=CC=CC=4)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC2=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC=2C=CC=CC=2)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H]2N(CCC2)C(=O)[C@@H](N)CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N2[C@@H](CCC2)C(=O)N2[C@@H](CCC2)C(=O)N[C@@H](CC=2C=CC(O)=CC=2)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N2[C@@H](CCC2)C(=O)N3)C(=O)NCC(=O)NCC(=O)N[C@@H](C)C(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@H](C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@H](C(=O)N1)C(C)C)[C@@H](C)O)[C@@H](C)CC)=O)[C@@H](C)CC)C1=CC=C(O)C=C1 ZPNFWUPYTFPOJU-LPYSRVMUSA-N 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 229940079322 interferon Drugs 0.000 description 1
- 229940047124 interferons Drugs 0.000 description 1
- 229940046732 interleukin inhibitors Drugs 0.000 description 1
- 102000002467 interleukin receptors Human genes 0.000 description 1
- 108010093036 interleukin receptors Proteins 0.000 description 1
- 229940076144 interleukin-10 Drugs 0.000 description 1
- 229940047122 interleukins Drugs 0.000 description 1
- 235000011073 invertase Nutrition 0.000 description 1
- 229940070023 iproniazide Drugs 0.000 description 1
- SURQXAFEQWPFPV-UHFFFAOYSA-L iron(2+) sulfate heptahydrate Chemical compound O.O.O.O.O.O.O.[Fe+2].[O-]S([O-])(=O)=O SURQXAFEQWPFPV-UHFFFAOYSA-L 0.000 description 1
- LNOZJRCUHSPCDZ-UHFFFAOYSA-L iron(ii) acetate Chemical compound [Fe+2].CC([O-])=O.CC([O-])=O LNOZJRCUHSPCDZ-UHFFFAOYSA-L 0.000 description 1
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 1
- 150000002513 isocyanates Chemical class 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 229960000318 kanamycin Drugs 0.000 description 1
- 229930027917 kanamycin Natural products 0.000 description 1
- SBUJHOSQTJFQJX-NOAMYHISSA-N kanamycin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CN)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O2)O)[C@H](N)C[C@@H]1N SBUJHOSQTJFQJX-NOAMYHISSA-N 0.000 description 1
- 229930182823 kanamycin A Natural products 0.000 description 1
- 229960004752 ketorolac Drugs 0.000 description 1
- OZWKMVRBQXNZKK-UHFFFAOYSA-N ketorolac Chemical compound OC(=O)C1CCN2C1=CC=C2C(=O)C1=CC=CC=C1 OZWKMVRBQXNZKK-UHFFFAOYSA-N 0.000 description 1
- 229940116108 lactase Drugs 0.000 description 1
- 150000002596 lactones Chemical class 0.000 description 1
- 108010021336 lanreotide Proteins 0.000 description 1
- 229960002437 lanreotide Drugs 0.000 description 1
- 239000002523 lectin Substances 0.000 description 1
- 210000005240 left ventricle Anatomy 0.000 description 1
- 210000002332 leydig cell Anatomy 0.000 description 1
- 229960004194 lidocaine Drugs 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 239000003589 local anesthetic agent Substances 0.000 description 1
- 229960005015 local anesthetics Drugs 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 229960004844 lovastatin Drugs 0.000 description 1
- PCZOHLXUXFIOCF-BXMDZJJMSA-N lovastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 PCZOHLXUXFIOCF-BXMDZJJMSA-N 0.000 description 1
- QLJODMDSTUBWDW-UHFFFAOYSA-N lovastatin hydroxy acid Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(C)C=C21 QLJODMDSTUBWDW-UHFFFAOYSA-N 0.000 description 1
- 239000003055 low molecular weight heparin Substances 0.000 description 1
- 229940127215 low-molecular weight heparin Drugs 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 229960003646 lysine Drugs 0.000 description 1
- 210000002819 lysozyme secreting cell Anatomy 0.000 description 1
- 229940124302 mTOR inhibitor Drugs 0.000 description 1
- 210000002540 macrophage Anatomy 0.000 description 1
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 1
- 125000005439 maleimidyl group Chemical group C1(C=CC(N1*)=O)=O 0.000 description 1
- 239000000845 maltitol Substances 0.000 description 1
- VQHSOMBJVWLPSR-WUJBLJFYSA-N maltitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O VQHSOMBJVWLPSR-WUJBLJFYSA-N 0.000 description 1
- 235000010449 maltitol Nutrition 0.000 description 1
- 229940035436 maltitol Drugs 0.000 description 1
- 239000003628 mammalian target of rapamycin inhibitor Substances 0.000 description 1
- 238000000691 measurement method Methods 0.000 description 1
- 229960004961 mechlorethamine Drugs 0.000 description 1
- HAWPXGHAZFHHAD-UHFFFAOYSA-N mechlorethamine Chemical compound ClCCN(C)CCCl HAWPXGHAZFHHAD-UHFFFAOYSA-N 0.000 description 1
- 229960003803 meclofenamic acid Drugs 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 229960003464 mefenamic acid Drugs 0.000 description 1
- HYYBABOKPJLUIN-UHFFFAOYSA-N mefenamic acid Chemical compound CC1=CC=CC(NC=2C(=CC=CC=2)C(O)=O)=C1C HYYBABOKPJLUIN-UHFFFAOYSA-N 0.000 description 1
- 210000003593 megakaryocyte Anatomy 0.000 description 1
- 210000002752 melanocyte Anatomy 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- SGDBTWWWUNNDEQ-LBPRGKRZSA-N melphalan Chemical compound OC(=O)[C@@H](N)CC1=CC=C(N(CCCl)CCCl)C=C1 SGDBTWWWUNNDEQ-LBPRGKRZSA-N 0.000 description 1
- 229960001924 melphalan Drugs 0.000 description 1
- 229960000901 mepacrine Drugs 0.000 description 1
- GLVAUDGFNGKCSF-UHFFFAOYSA-N mercaptopurine Chemical compound S=C1NC=NC2=C1NC=N2 GLVAUDGFNGKCSF-UHFFFAOYSA-N 0.000 description 1
- 229960001428 mercaptopurine Drugs 0.000 description 1
- 229960000485 methotrexate Drugs 0.000 description 1
- 229960004584 methylprednisolone Drugs 0.000 description 1
- HPNSFSBZBAHARI-UHFFFAOYSA-N micophenolic acid Natural products OC1=C(CC=C(C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-UHFFFAOYSA-N 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 231100000782 microtubule inhibitor Toxicity 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 229960004023 minocycline Drugs 0.000 description 1
- 230000003547 miosis Effects 0.000 description 1
- 239000003604 miotic agent Substances 0.000 description 1
- 229960004857 mitomycin Drugs 0.000 description 1
- 229960000350 mitotane Drugs 0.000 description 1
- KKZJGLLVHKMTCM-UHFFFAOYSA-N mitoxantrone Chemical compound O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO KKZJGLLVHKMTCM-UHFFFAOYSA-N 0.000 description 1
- 229960001156 mitoxantrone Drugs 0.000 description 1
- 235000019426 modified starch Nutrition 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- 230000000921 morphogenic effect Effects 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 229920006030 multiblock copolymer Polymers 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 229940035363 muscle relaxants Drugs 0.000 description 1
- RTGDFNSFWBGLEC-SYZQJQIISA-N mycophenolate mofetil Chemical compound COC1=C(C)C=2COC(=O)C=2C(O)=C1C\C=C(/C)CCC(=O)OCCN1CCOCC1 RTGDFNSFWBGLEC-SYZQJQIISA-N 0.000 description 1
- 229960004866 mycophenolate mofetil Drugs 0.000 description 1
- 229960000951 mycophenolic acid Drugs 0.000 description 1
- HPNSFSBZBAHARI-RUDMXATFSA-N mycophenolic acid Chemical compound OC1=C(C\C=C(/C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-RUDMXATFSA-N 0.000 description 1
- 239000003158 myorelaxant agent Substances 0.000 description 1
- BCXCABRDBBWWGY-UHFFFAOYSA-N n-benzyl-n-methylprop-2-yn-1-amine;hydrochloride Chemical compound Cl.C#CCN(C)CC1=CC=CC=C1 BCXCABRDBBWWGY-UHFFFAOYSA-N 0.000 description 1
- ZQXSMRAEXCEDJD-UHFFFAOYSA-N n-ethenylformamide Chemical compound C=CNC=O ZQXSMRAEXCEDJD-UHFFFAOYSA-N 0.000 description 1
- 229960004270 nabumetone Drugs 0.000 description 1
- 229960002009 naproxen Drugs 0.000 description 1
- CMWTZPSULFXXJA-VIFPVBQESA-M naproxen(1-) Chemical compound C1=C([C@H](C)C([O-])=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-VIFPVBQESA-M 0.000 description 1
- 229920005615 natural polymer Polymers 0.000 description 1
- 230000014508 negative regulation of coagulation Effects 0.000 description 1
- 229960004927 neomycin Drugs 0.000 description 1
- LULNWZDBKTWDGK-UHFFFAOYSA-M neostigmine bromide Chemical compound [Br-].CN(C)C(=O)OC1=CC=CC([N+](C)(C)C)=C1 LULNWZDBKTWDGK-UHFFFAOYSA-M 0.000 description 1
- 229960001499 neostigmine bromide Drugs 0.000 description 1
- 229940053128 nerve growth factor Drugs 0.000 description 1
- 230000007514 neuronal growth Effects 0.000 description 1
- 239000002581 neurotoxin Substances 0.000 description 1
- 231100000618 neurotoxin Toxicity 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 229960003057 nialamide Drugs 0.000 description 1
- 239000002840 nitric oxide donor Substances 0.000 description 1
- 229910000510 noble metal Inorganic materials 0.000 description 1
- 150000002840 non-reducing disaccharides Chemical class 0.000 description 1
- 239000000041 non-steroidal anti-inflammatory agent Substances 0.000 description 1
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 230000000269 nucleophilic effect Effects 0.000 description 1
- 239000002777 nucleoside Substances 0.000 description 1
- 125000003835 nucleoside group Chemical group 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 125000003729 nucleotide group Chemical group 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 210000004248 oligodendroglia Anatomy 0.000 description 1
- 230000000771 oncological effect Effects 0.000 description 1
- 210000000287 oocyte Anatomy 0.000 description 1
- 229940005483 opioid analgesics Drugs 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 210000000963 osteoblast Anatomy 0.000 description 1
- 210000002997 osteoclast Anatomy 0.000 description 1
- 210000004409 osteocyte Anatomy 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- CZYFDZPOVORIRJ-UHFFFAOYSA-N oxiran-2-amine Chemical compound NC1CO1 CZYFDZPOVORIRJ-UHFFFAOYSA-N 0.000 description 1
- 229960001592 paclitaxel Drugs 0.000 description 1
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 229960001789 papaverine Drugs 0.000 description 1
- 229960005489 paracetamol Drugs 0.000 description 1
- 210000004738 parenchymal cell Anatomy 0.000 description 1
- 229960001779 pargyline Drugs 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 1
- 229960002340 pentostatin Drugs 0.000 description 1
- FPVKHBSQESCIEP-JQCXWYLXSA-N pentostatin Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(N=CNC[C@H]2O)=C2N=C1 FPVKHBSQESCIEP-JQCXWYLXSA-N 0.000 description 1
- 108010011903 peptide receptors Proteins 0.000 description 1
- 102000014187 peptide receptors Human genes 0.000 description 1
- 239000000816 peptidomimetic Substances 0.000 description 1
- 238000013146 percutaneous coronary intervention Methods 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 230000003285 pharmacodynamic effect Effects 0.000 description 1
- 229960002895 phenylbutazone Drugs 0.000 description 1
- VYMDGNCVAMGZFE-UHFFFAOYSA-N phenylbutazonum Chemical compound O=C1C(CCCC)C(=O)N(C=2C=CC=CC=2)N1C1=CC=CC=C1 VYMDGNCVAMGZFE-UHFFFAOYSA-N 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 125000002467 phosphate group Chemical group [H]OP(=O)(O[H])O[*] 0.000 description 1
- XYFCBTPGUUZFHI-UHFFFAOYSA-O phosphonium Chemical compound [PH4+] XYFCBTPGUUZFHI-UHFFFAOYSA-O 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 238000002428 photodynamic therapy Methods 0.000 description 1
- 208000017983 photosensitivity disease Diseases 0.000 description 1
- 231100000434 photosensitization Toxicity 0.000 description 1
- 229960001847 physostigmine sulfate Drugs 0.000 description 1
- 229960002702 piroxicam Drugs 0.000 description 1
- QYSPLQLAKJAUJT-UHFFFAOYSA-N piroxicam Chemical compound OC=1C2=CC=CC=C2S(=O)(=O)N(C)C=1C(=O)NC1=CC=CC=N1 QYSPLQLAKJAUJT-UHFFFAOYSA-N 0.000 description 1
- 229940127126 plasminogen activator Drugs 0.000 description 1
- 210000004180 plasmocyte Anatomy 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 229920001643 poly(ether ketone) Polymers 0.000 description 1
- 239000005015 poly(hydroxybutyrate) Substances 0.000 description 1
- 229940065514 poly(lactide) Drugs 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920002627 poly(phosphazenes) Polymers 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 229920001515 polyalkylene glycol Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 238000012643 polycondensation polymerization Methods 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000004633 polyglycolic acid Substances 0.000 description 1
- 230000000379 polymerizing effect Effects 0.000 description 1
- 108091033319 polynucleotide Proteins 0.000 description 1
- 102000040430 polynucleotide Human genes 0.000 description 1
- 239000002157 polynucleotide Substances 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 150000004804 polysaccharides Polymers 0.000 description 1
- 239000004810 polytetrafluoroethylene Substances 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000003361 porogen Substances 0.000 description 1
- 239000012286 potassium permanganate Substances 0.000 description 1
- OXCMYAYHXIHQOA-UHFFFAOYSA-N potassium;[2-butyl-5-chloro-3-[[4-[2-(1,2,4-triaza-3-azanidacyclopenta-1,4-dien-5-yl)phenyl]phenyl]methyl]imidazol-4-yl]methanol Chemical compound [K+].CCCCC1=NC(Cl)=C(CO)N1CC1=CC=C(C=2C(=CC=CC=2)C2=N[N-]N=N2)C=C1 OXCMYAYHXIHQOA-UHFFFAOYSA-N 0.000 description 1
- 229960002965 pravastatin Drugs 0.000 description 1
- TUZYXOIXSAXUGO-PZAWKZKUSA-N pravastatin Chemical compound C1=C[C@H](C)[C@H](CC[C@@H](O)C[C@@H](O)CC(O)=O)[C@H]2[C@@H](OC(=O)[C@@H](C)CC)C[C@H](O)C=C21 TUZYXOIXSAXUGO-PZAWKZKUSA-N 0.000 description 1
- 229960005205 prednisolone Drugs 0.000 description 1
- OIGNJSKKLXVSLS-VWUMJDOOSA-N prednisolone Chemical compound O=C1C=C[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 OIGNJSKKLXVSLS-VWUMJDOOSA-N 0.000 description 1
- 229960004618 prednisone Drugs 0.000 description 1
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 229960004919 procaine Drugs 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- CPTBDICYNRMXFX-UHFFFAOYSA-N procarbazine Chemical compound CNNCC1=CC=C(C(=O)NC(C)C)C=C1 CPTBDICYNRMXFX-UHFFFAOYSA-N 0.000 description 1
- 229960000624 procarbazine Drugs 0.000 description 1
- 229940002612 prodrug Drugs 0.000 description 1
- 239000000651 prodrug Substances 0.000 description 1
- 230000000750 progressive effect Effects 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 150000003815 prostacyclins Chemical class 0.000 description 1
- 229940039716 prothrombin Drugs 0.000 description 1
- 239000003586 protic polar solvent Substances 0.000 description 1
- 150000003856 quaternary ammonium compounds Chemical class 0.000 description 1
- 125000001453 quaternary ammonium group Chemical group 0.000 description 1
- GPKJTRJOBQGKQK-UHFFFAOYSA-N quinacrine Chemical compound C1=C(OC)C=C2C(NC(C)CCCN(CC)CC)=C(C=CC(Cl)=C3)C3=NC2=C1 GPKJTRJOBQGKQK-UHFFFAOYSA-N 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 150000005838 radical anions Chemical class 0.000 description 1
- 150000005839 radical cations Chemical class 0.000 description 1
- 238000007348 radical reaction Methods 0.000 description 1
- 239000002516 radical scavenger Substances 0.000 description 1
- ZAHRKKWIAAJSAO-UHFFFAOYSA-N rapamycin Natural products COCC(O)C(=C/C(C)C(=O)CC(OC(=O)C1CCCCN1C(=O)C(=O)C2(O)OC(CC(OC)C(=CC=CC=CC(C)CC(C)C(=O)C)C)CCC2C)C(C)CC3CCC(O)C(C3)OC)C ZAHRKKWIAAJSAO-UHFFFAOYSA-N 0.000 description 1
- 229920013730 reactive polymer Polymers 0.000 description 1
- 238000006479 redox reaction Methods 0.000 description 1
- 238000007634 remodeling Methods 0.000 description 1
- 230000003252 repetitive effect Effects 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 239000013557 residual solvent Substances 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 210000001995 reticulocyte Anatomy 0.000 description 1
- 210000001525 retina Anatomy 0.000 description 1
- 230000033764 rhythmic process Effects 0.000 description 1
- 108091092562 ribozyme Proteins 0.000 description 1
- 238000007151 ring opening polymerisation reaction Methods 0.000 description 1
- 238000007142 ring opening reaction Methods 0.000 description 1
- 229960004641 rituximab Drugs 0.000 description 1
- 229960000581 salicylamide Drugs 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 239000010980 sapphire Substances 0.000 description 1
- 229910052594 sapphire Inorganic materials 0.000 description 1
- 210000004116 schwann cell Anatomy 0.000 description 1
- 210000000717 sertoli cell Anatomy 0.000 description 1
- 238000007493 shaping process Methods 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 description 1
- 229910010271 silicon carbide Inorganic materials 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- 229960003600 silver sulfadiazine Drugs 0.000 description 1
- UEJSSZHHYBHCEL-UHFFFAOYSA-N silver(1+) sulfadiazinate Chemical compound [Ag+].C1=CC(N)=CC=C1S(=O)(=O)[N-]C1=NC=CC=N1 UEJSSZHHYBHCEL-UHFFFAOYSA-N 0.000 description 1
- 229960002855 simvastatin Drugs 0.000 description 1
- RYMZZMVNJRMUDD-HGQWONQESA-N simvastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)C(C)(C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 RYMZZMVNJRMUDD-HGQWONQESA-N 0.000 description 1
- 229960002930 sirolimus Drugs 0.000 description 1
- 210000002363 skeletal muscle cell Anatomy 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- 239000004055 small Interfering RNA Substances 0.000 description 1
- 210000000329 smooth muscle myocyte Anatomy 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 1
- UGTZMIPZNRIWHX-UHFFFAOYSA-K sodium trimetaphosphate Chemical compound [Na+].[Na+].[Na+].[O-]P1(=O)OP([O-])(=O)OP([O-])(=O)O1 UGTZMIPZNRIWHX-UHFFFAOYSA-K 0.000 description 1
- 235000019832 sodium triphosphate Nutrition 0.000 description 1
- UHYAQBLOGVNWNT-UHFFFAOYSA-M sodium;2-[1-(4-chlorobenzoyl)-5-methoxy-2-methylindol-3-yl]acetate;trihydrate Chemical compound O.O.O.[Na+].CC1=C(CC([O-])=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(Cl)C=C1 UHYAQBLOGVNWNT-UHFFFAOYSA-M 0.000 description 1
- QPBKUJIATWTGHV-UHFFFAOYSA-M sodium;2-carbamoylphenolate Chemical compound [Na+].NC(=O)C1=CC=CC=C1[O-] QPBKUJIATWTGHV-UHFFFAOYSA-M 0.000 description 1
- AGHLUVOCTHWMJV-UHFFFAOYSA-J sodium;gold(3+);2-sulfanylbutanedioate Chemical compound [Na+].[Au+3].[O-]C(=O)CC(S)C([O-])=O.[O-]C(=O)CC(S)C([O-])=O AGHLUVOCTHWMJV-UHFFFAOYSA-J 0.000 description 1
- 238000000935 solvent evaporation Methods 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 210000005070 sphincter Anatomy 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 210000000130 stem cell Anatomy 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 239000002731 stomach secretion inhibitor Substances 0.000 description 1
- 238000005728 strengthening Methods 0.000 description 1
- 229960005202 streptokinase Drugs 0.000 description 1
- 229960001052 streptozocin Drugs 0.000 description 1
- ZSJLQEPLLKMAKR-GKHCUFPYSA-N streptozocin Chemical compound O=NN(C)C(=O)N[C@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O ZSJLQEPLLKMAKR-GKHCUFPYSA-N 0.000 description 1
- 229940014800 succinic anhydride Drugs 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 125000004089 sulfido group Chemical group [S-]* 0.000 description 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-O sulfonium Chemical compound [SH3+] RWSOTUBLDIXVET-UHFFFAOYSA-O 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid group Chemical class S(O)(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- MLKXDPUZXIRXEP-MFOYZWKCSA-N sulindac Chemical compound CC1=C(CC(O)=O)C2=CC(F)=CC=C2\C1=C/C1=CC=C(S(C)=O)C=C1 MLKXDPUZXIRXEP-MFOYZWKCSA-N 0.000 description 1
- 229960000894 sulindac Drugs 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- YLJREFDVOIBQDA-UHFFFAOYSA-N tacrine Chemical compound C1=CC=C2C(N)=C(CCCC3)C3=NC2=C1 YLJREFDVOIBQDA-UHFFFAOYSA-N 0.000 description 1
- 229960001967 tacrolimus Drugs 0.000 description 1
- QJJXYPPXXYFBGM-SHYZHZOCSA-N tacrolimus Natural products CO[C@H]1C[C@H](CC[C@@H]1O)C=C(C)[C@H]2OC(=O)[C@H]3CCCCN3C(=O)C(=O)[C@@]4(O)O[C@@H]([C@H](C[C@H]4C)OC)[C@@H](C[C@H](C)CC(=C[C@@H](CC=C)C(=O)C[C@H](O)[C@H]2C)C)OC QJJXYPPXXYFBGM-SHYZHZOCSA-N 0.000 description 1
- 229910052715 tantalum Inorganic materials 0.000 description 1
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 1
- DKPFODGZWDEEBT-QFIAKTPHSA-N taxane Chemical class C([C@]1(C)CCC[C@@H](C)[C@H]1C1)C[C@H]2[C@H](C)CC[C@@H]1C2(C)C DKPFODGZWDEEBT-QFIAKTPHSA-N 0.000 description 1
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 1
- NRUKOCRGYNPUPR-QBPJDGROSA-N teniposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@@H](OC[C@H]4O3)C=3SC=CC=3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 NRUKOCRGYNPUPR-QBPJDGROSA-N 0.000 description 1
- 229960001278 teniposide Drugs 0.000 description 1
- 229960002871 tenoxicam Drugs 0.000 description 1
- LZNWYQJJBLGYLT-UHFFFAOYSA-N tenoxicam Chemical compound OC=1C=2SC=CC=2S(=O)(=O)N(C)C=1C(=O)NC1=CC=CC=N1 LZNWYQJJBLGYLT-UHFFFAOYSA-N 0.000 description 1
- ISXSCDLOGDJUNJ-UHFFFAOYSA-N tert-butyl prop-2-enoate Chemical compound CC(C)(C)OC(=O)C=C ISXSCDLOGDJUNJ-UHFFFAOYSA-N 0.000 description 1
- 229960002372 tetracaine Drugs 0.000 description 1
- GKCBAIGFKIBETG-UHFFFAOYSA-N tetracaine Chemical compound CCCCNC1=CC=C(C(=O)OCCN(C)C)C=C1 GKCBAIGFKIBETG-UHFFFAOYSA-N 0.000 description 1
- 229960002180 tetracycline Drugs 0.000 description 1
- 229930101283 tetracycline Natural products 0.000 description 1
- 235000019364 tetracycline Nutrition 0.000 description 1
- 150000003522 tetracyclines Chemical class 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- ZRKFYGHZFMAOKI-QMGMOQQFSA-N tgfbeta Chemical compound C([C@H](NC(=O)[C@H](C(C)C)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCSC)C(C)C)[C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 ZRKFYGHZFMAOKI-QMGMOQQFSA-N 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- YRHRIQCWCFGUEQ-UHFFFAOYSA-N thioxanthen-9-one Chemical compound C1=CC=C2C(=O)C3=CC=CC=C3SC2=C1 YRHRIQCWCFGUEQ-UHFFFAOYSA-N 0.000 description 1
- 210000000115 thoracic cavity Anatomy 0.000 description 1
- 229960004072 thrombin Drugs 0.000 description 1
- 239000003868 thrombin inhibitor Substances 0.000 description 1
- 230000002345 thrombinlike Effects 0.000 description 1
- 229960000103 thrombolytic agent Drugs 0.000 description 1
- RZWIIPASKMUIAC-VQTJNVASSA-N thromboxane Chemical compound CCCCCCCC[C@H]1OCCC[C@@H]1CCCCCCC RZWIIPASKMUIAC-VQTJNVASSA-N 0.000 description 1
- 229960005001 ticlopidine Drugs 0.000 description 1
- PHWBOXQYWZNQIN-UHFFFAOYSA-N ticlopidine Chemical compound ClC1=CC=CC=C1CN1CC(C=CS2)=C2CC1 PHWBOXQYWZNQIN-UHFFFAOYSA-N 0.000 description 1
- MNRILEROXIRVNJ-UHFFFAOYSA-N tioguanine Chemical compound N1C(N)=NC(=S)C2=NC=N[C]21 MNRILEROXIRVNJ-UHFFFAOYSA-N 0.000 description 1
- 229960003087 tioguanine Drugs 0.000 description 1
- 229960003425 tirofiban Drugs 0.000 description 1
- COKMIXFXJJXBQG-NRFANRHFSA-N tirofiban Chemical compound C1=CC(C[C@H](NS(=O)(=O)CCCC)C(O)=O)=CC=C1OCCCCC1CCNCC1 COKMIXFXJJXBQG-NRFANRHFSA-N 0.000 description 1
- 239000003106 tissue adhesive Substances 0.000 description 1
- 229940075469 tissue adhesives Drugs 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 229960001017 tolmetin Drugs 0.000 description 1
- UPSPUYADGBWSHF-UHFFFAOYSA-N tolmetin Chemical compound C1=CC(C)=CC=C1C(=O)C1=CC=C(CC(O)=O)N1C UPSPUYADGBWSHF-UHFFFAOYSA-N 0.000 description 1
- 210000000515 tooth Anatomy 0.000 description 1
- 238000011200 topical administration Methods 0.000 description 1
- 239000003204 tranquilizing agent Substances 0.000 description 1
- 230000002936 tranquilizing effect Effects 0.000 description 1
- 238000005809 transesterification reaction Methods 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 229960003741 tranylcypromine Drugs 0.000 description 1
- 229960000575 trastuzumab Drugs 0.000 description 1
- 229960005294 triamcinolone Drugs 0.000 description 1
- GFNANZIMVAIWHM-OBYCQNJPSA-N triamcinolone Chemical compound O=C1C=C[C@]2(C)[C@@]3(F)[C@@H](O)C[C@](C)([C@@]([C@H](O)C4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 GFNANZIMVAIWHM-OBYCQNJPSA-N 0.000 description 1
- VSQQQLOSPVPRAZ-RRKCRQDMSA-N trifluridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(C(F)(F)F)=C1 VSQQQLOSPVPRAZ-RRKCRQDMSA-N 0.000 description 1
- 229960003962 trifluridine Drugs 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 108010008187 triwaglerin Proteins 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 102000003390 tumor necrosis factor Human genes 0.000 description 1
- 241000701161 unidentified adenovirus Species 0.000 description 1
- 241000701447 unidentified baculovirus Species 0.000 description 1
- 241001529453 unidentified herpesvirus Species 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- 150000003673 urethanes Chemical class 0.000 description 1
- 229960005356 urokinase Drugs 0.000 description 1
- 229960003165 vancomycin Drugs 0.000 description 1
- MYPYJXKWCTUITO-UHFFFAOYSA-N vancomycin Natural products O1C(C(=C2)Cl)=CC=C2C(O)C(C(NC(C2=CC(O)=CC(O)=C2C=2C(O)=CC=C3C=2)C(O)=O)=O)NC(=O)C3NC(=O)C2NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(CC(C)C)NC)C(O)C(C=C3Cl)=CC=C3OC3=CC2=CC1=C3OC1OC(CO)C(O)C(O)C1OC1CC(C)(N)C(O)C(C)O1 MYPYJXKWCTUITO-UHFFFAOYSA-N 0.000 description 1
- MYPYJXKWCTUITO-LYRMYLQWSA-O vancomycin(1+) Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=C2C=C3C=C1OC1=CC=C(C=C1Cl)[C@@H](O)[C@H](C(N[C@@H](CC(N)=O)C(=O)N[C@H]3C(=O)N[C@H]1C(=O)N[C@H](C(N[C@@H](C3=CC(O)=CC(O)=C3C=3C(O)=CC=C1C=3)C([O-])=O)=O)[C@H](O)C1=CC=C(C(=C1)Cl)O2)=O)NC(=O)[C@@H](CC(C)C)[NH2+]C)[C@H]1C[C@](C)([NH3+])[C@H](O)[C@H](C)O1 MYPYJXKWCTUITO-LYRMYLQWSA-O 0.000 description 1
- 229950007952 vapiprost Drugs 0.000 description 1
- 229940124549 vasodilator Drugs 0.000 description 1
- 239000003071 vasodilator agent Substances 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 229960003636 vidarabine Drugs 0.000 description 1
- 229960003048 vinblastine Drugs 0.000 description 1
- JXLYSJRDGCGARV-XQKSVPLYSA-N vincaleukoblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-XQKSVPLYSA-N 0.000 description 1
- 229960004528 vincristine Drugs 0.000 description 1
- OGWKCGZFUXNPDA-XQKSVPLYSA-N vincristine Chemical compound C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](OC(C)=O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-XQKSVPLYSA-N 0.000 description 1
- OGWKCGZFUXNPDA-UHFFFAOYSA-N vincristine Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(OC(C)=O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-UHFFFAOYSA-N 0.000 description 1
- 229960002066 vinorelbine Drugs 0.000 description 1
- GBABOYUKABKIAF-GHYRFKGUSA-N vinorelbine Chemical compound C1N(CC=2C3=CC=CC=C3NC=22)CC(CC)=C[C@H]1C[C@]2(C(=O)OC)C1=CC([C@]23[C@H]([C@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC GBABOYUKABKIAF-GHYRFKGUSA-N 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 239000003039 volatile agent Substances 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
- 235000005074 zinc chloride Nutrition 0.000 description 1
- 239000011592 zinc chloride Substances 0.000 description 1
- CGTADGCBEXYWNE-JUKNQOCSSA-N zotarolimus Chemical compound N1([C@H]2CC[C@@H](C[C@@H](C)[C@H]3OC(=O)[C@@H]4CCCCN4C(=O)C(=O)[C@@]4(O)[C@H](C)CC[C@H](O4)C[C@@H](/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C3)OC)C[C@H]2OC)C=NN=N1 CGTADGCBEXYWNE-JUKNQOCSSA-N 0.000 description 1
- 229950009819 zotarolimus Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/20—Polysaccharides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
- A61K9/0024—Solid, semi-solid or solidifying implants, which are implanted or injected in body tissue
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0048—Eye, e.g. artificial tears
- A61K9/0051—Ocular inserts, ocular implants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/28—Materials for coating prostheses
- A61L27/34—Macromolecular materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/54—Biologically active materials, e.g. therapeutic substances
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/58—Materials at least partially resorbable by the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L29/00—Materials for catheters, medical tubing, cannulae, or endoscopes or for coating catheters
- A61L29/08—Materials for coatings
- A61L29/085—Macromolecular materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L29/00—Materials for catheters, medical tubing, cannulae, or endoscopes or for coating catheters
- A61L29/14—Materials characterised by their function or physical properties, e.g. lubricating compositions
- A61L29/16—Biologically active materials, e.g. therapeutic substances
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/04—Macromolecular materials
- A61L31/042—Polysaccharides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/08—Materials for coatings
- A61L31/10—Macromolecular materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/16—Biologically active materials, e.g. therapeutic substances
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D1/00—Processes for applying liquids or other fluent materials
- B05D1/36—Successively applying liquids or other fluent materials, e.g. without intermediate treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08B—POLYSACCHARIDES; DERIVATIVES THEREOF
- C08B31/00—Preparation of derivatives of starch
- C08B31/003—Crosslinking of starch
- C08B31/006—Crosslinking of derivatives of starch
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08B—POLYSACCHARIDES; DERIVATIVES THEREOF
- C08B31/00—Preparation of derivatives of starch
- C08B31/02—Esters
- C08B31/04—Esters of organic acids, e.g. alkenyl-succinated starch
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08B—POLYSACCHARIDES; DERIVATIVES THEREOF
- C08B33/00—Preparation of derivatives of amylose
- C08B33/02—Esters
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08B—POLYSACCHARIDES; DERIVATIVES THEREOF
- C08B33/00—Preparation of derivatives of amylose
- C08B33/04—Ethers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L3/00—Compositions of starch, amylose or amylopectin or of their derivatives or degradation products
- C08L3/02—Starch; Degradation products thereof, e.g. dextrin
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L3/00—Compositions of starch, amylose or amylopectin or of their derivatives or degradation products
- C08L3/12—Amylose; Amylopectin; Degradation products thereof
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D103/00—Coating compositions based on starch, amylose or amylopectin or on their derivatives or degradation products
- C09D103/02—Starch; Degradation products thereof, e.g. dextrin
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D103/00—Coating compositions based on starch, amylose or amylopectin or on their derivatives or degradation products
- C09D103/14—Amylose derivatives; Amylopectin derivatives
- C09D103/18—Ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/34—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyesters, polyamino acids, polysiloxanes, polyphosphazines, copolymers of polyalkylene glycol or poloxamers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/20—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing organic materials
- A61L2300/23—Carbohydrates
- A61L2300/232—Monosaccharides, disaccharides, polysaccharides, lipopolysaccharides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/20—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing organic materials
- A61L2300/252—Polypeptides, proteins, e.g. glycoproteins, lipoproteins, cytokines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/20—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing organic materials
- A61L2300/258—Genetic materials, DNA, RNA, genes, vectors, e.g. plasmids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/60—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a special physical form
- A61L2300/602—Type of release, e.g. controlled, sustained, slow
- A61L2300/604—Biodegradation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/60—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a special physical form
- A61L2300/606—Coatings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2420/00—Materials or methods for coatings medical devices
- A61L2420/02—Methods for coating medical devices
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D7/00—Processes, other than flocking, specially adapted for applying liquids or other fluent materials to particular surfaces or for applying particular liquids or other fluent materials
- B05D7/50—Multilayers
- B05D7/52—Two layers
Definitions
- the present invention relates to in vivo formed matrices comprising a natural biodegradable polymeric material.
- Bioactive agents can be included in the in vivo formed matrices to provide a therapeutic effect to a patient.
- DES drug-eluting stents
- a bioactive agent can be coupled to the surface of a medical device by surface modification, embedded, and released from within polymeric materials (matrix-type), or surrounded by and released through a carrier (reservoir-type).
- the polymeric materials in such applications should optimally act as a biologically inert barrier and not induce further inflammation within the body.
- the molecular weight, porosity of the polymer, a greater percentage of coating exposed on the medical device, and the thickness of the polymer coating can contribute to adverse reactions to the medical device.
- biodegradable coatings that include polylactic acid have been described in a number of documents, for example, U.S. Pat. No. 6,258,121, there remains a need for improved coatings and coating materials.
- biodegradable materials that degrade into materials that are not typically found in the body, or that are found at particularly low levels in the body. These types of biodegradable materials have the potential to degrade into products that cause unwanted side effects in the body by virtue of their presence or concentration in vivo. These unwanted side effects can include immune reactions, toxic buildup of the degradation products in the liver, or the initiation or provocation of other adverse effects on cells or tissue in the body.
- biodegradable materials may not be obtained at consistent purity due to variations inherent in natural materials. This is relevant at least with regard to biodegradable materials derived from animal sources. Inconsistencies in preparations of biodegradable materials can result in problematic coatings.
- biodegradable drug delivery coatings that are easy to prepare, cost effective, and that also offer a wide range of flexibility with regard to the type and amount of drug or drugs to be delivered from the biodegradable coating.
- Biodegradable sealant compositions have been used on articles having porous surfaces, such as fabrics associated with implantable medical articles.
- the sealant coating initially renders the porous surface impermeable to fluids for a period of time.
- cells involved in tissue repair infiltrate the porous material and replace the sealant materials.
- newly formed tissue replaces the original function of the coated sealant over a period of time.
- the collagen used in sealant technologies is obtained from non-human animal sources, such as bovine sources.
- bovine collagen preparations may contain unwanted contaminants that are undesirable for introduction into a human subject.
- an unwanted contaminant is the prionic particles that cause Bovine Spongiform Encephalopathy (BSE).
- BSE also termed Mad Cow Disease
- TSEs transmissible spongiform encephalopathies
- Various forms of TSE have been reported, including scrapie in sheep and chronic wasting disease in elk and mule deer. It is generally believed that the use of recycled animal parts led to the cross-species contamination of scrapie in sheep to mad cow disease, and the ingestion of contaminated beef and bovine products led to the human variant of this disease, Creutzfeldt-Jakob Disease (CJD).
- CJD Creutzfeldt-Jakob Disease
- preparations from animal sources may provide other unwanted contaminants, such as antigenic factors.
- antigenic factors may promote a localized immune response in the vicinity of the implanted article and foul its function. These factors may also cause infection as well as local inflammation.
- the present invention provides compositions and methods for preparing biodegradable coatings that are particularly useful for coating surfaces of implantable medical devices, such as stents and catheters, and are capable of releasing bioactive agents from the device surface.
- These coating compositions include a natural biodegradable polysaccharide as a component that can be crosslinked to form a matrix from which a therapeutic material such as a drug, a biomolecule, or cells (referred to herein as a “bioactive agents”) can be released or retained.
- a bioactive agent is present in and can be released from the biodegradable matrix; in other embodiments a bioactive agent is present in a biodegradable microparticle, the microparticle being immobilized within the matrix.
- the natural biodegradable polysaccharide is used to prepare an article, such as an article that can be implanted or formed within the body (for example, by in situ formation).
- the article can be amorphous, such as a polymerized mass of natural biodegradable polysaccharides that is formed within or on a portion of the body, by using an in vivo matrix-forming composition.
- the invention provides an article fabricated from natural biodegradable polysaccharide, wherein the article has a defined structure, and wherein the article can be implanted in the body (such as a filament).
- Such articles are referred to herein as “medical implants”.
- a medical implants having a defined structure can be formed by any suitable process, including molding, extruding, shaping, cutting, casting, and the like.
- the article can be used for one or more purposes, such as for releasing or retaining a bioactive agent at a location in the body.
- the article can be a bioactive agent-containing medical implant or depot.
- the article can also provide one or more mechanical or physical properties to a portion the body.
- the natural biodegradable polysaccharides can be included in a composition used for the formation of a biodegradable medical device such as a stent.
- the article such as an in vivo formed matrix
- the article is used in methods for the treatment of any one or more of a variety of medical conditions or indications, including restoring, improving, and/or augmenting tissue growth or function, in particular those for orthopedic, dental, and bone graft applications.
- These functions can be provided by placing a polymerized matrix of biodegradable polysaccharides in contact with a host tissue.
- the matrix can restore or improve tissue growth or function by, for example, promoting or permitting formation of new tissue between and into the matrix.
- the effect on tissue can be caused by the biodegradable polysaccharide itself, or the biodegradable polysaccharide in combination with one or more bioactive agent(s) that can be present in and/or released from the matrix.
- bioactive agents that can affect tissue function include peptides, such as peptides that are involved in tissue repair processes and belonging to the EGF, FGF, PDGF, TGF- ⁇ , VEGF, PD-ECGF or IGF families, and also peptides derived from bone morphogenetic protein 2, or BMP-2.
- the bioactive agent can also be a cell, such as a platelet.
- the coating or article can include a radiopacifying agent.
- a plurality of natural biodegradable polysaccharides are crosslinked to each other via coupling groups that are pendent from the natural biodegradable polysaccharide (i.e., one or more coupling groups are chemically bonded to the polysaccharide).
- the coupling group on the natural biodegradable polysaccharide is a polymerizable group.
- the polymerizable group can crosslink natural biodegradable polysaccharides together in the composition, thereby forming a natural biodegradable polysaccharide matrix, which can be a portion of a coating, an in-vivo formed matrix, or the body member of a medical implant.
- the natural biodegradable polysaccharides described herein are non-synthetic polysaccharides that can be associated with each other to form a matrix, which can be used as a coating or as an article, for example, a medical implant or an in-vivo formed matrix.
- the natural biodegradable polysaccharides can also be enzymatically degraded, but offer the advantage of being generally non-enzymatically hydrolytically stable. This is particularly advantageous for bioactive agent delivery, as in some aspects the invention provides coatings or articles capable of releasing the bioactive agent under conditions of enzyme-mediated degradation, but not by diffusion. Therefore, the kinetics of bioactive agent release from the coatings or articles of the invention are fundamentally different than those of coatings prepared from synthetic biodegradable materials, such as poly(lactides).
- Natural biodegradable polysaccharides include polysaccharide and/or polysaccharide derivatives that are obtained from natural sources, such as plants or animals.
- Exemplary natural biodegradable polysaccharides include amylose, maltodextrin, amylopectin, starch, dextran, hyaluronic acid, heparin, chondroitin sulfate, dermatan sulfate, heparan sulfate, keratan sulfate, dextran sulfate, pentosan polysulfate, and chitosan.
- Preferred polysaccharides are low molecular weight polymers that have little or no branching, such as those that are derived from and/or found in starch preparations, for example, amylose and maltodextrin.
- natural biodegradable polysaccharides are used that have an average molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, or 50,000 Da or less. In some aspects the natural biodegradable polysaccharides have an average molecular weight of 500 Da or greater. In some aspects the natural biodegradable polysaccharides have an average molecular weight in the range of about 1000 Da to about 10,000 Da. Natural biodegradable polysaccharides of particular molecular weights can be obtained commercially or can be prepared, for example, by acid hydrolysis and/or enzymatic degradation of a natural biodegradable polysaccharide preparation, such as starch.
- the decision of using natural biodegradable polysaccharides of a particular size range may depend on factors such as the physical characteristics of the coating composition (e.g., viscosity), the desired rate of degradation of the coating, the presence of other optional moieties in the coating composition (for example, bioactive agents, etc.), etc.
- the natural biodegradable polysaccharides that are used in accordance with the methods and compositions of the invention are readily available at a low cost and/or can be prepared easily using established techniques. This allows for a cost effective method of coating and fabricating medical articles.
- biodegradable polysaccharides such as maltodextrin or amylose
- a coating composition applied to the surface of a medical device, or for the formation of an article, such as one that can be used in vivo.
- Degradation of a natural biodegradable polysaccharide-containing article, or coating from the surface of a medical device can result in the release of, for example, naturally occurring mono- or disaccharides, such as glucose, which are common serum components.
- biodegradable polysaccharides that degrade into common serum components such as glucose
- synthetic biodegradable polysaccharides that degrade into non-natural compounds, or compounds that are found at very low concentrations in the body can be viewed as more acceptable than the use of synthetic biodegradable polysaccharides that degrade into non-natural compounds, or compounds that are found at very low concentrations in the body.
- this advantageous feature is reflected in the use of natural biodegradable polysaccharides which are non-animal derived, such as amylose and maltodextrin, and that degrade into products that present little or no immunogenic or toxic risk to the individual.
- the invention provides improved, cost-efficient, natural biodegradable polysaccharide compositions for articles or coatings that can be used in a variety of medical treatments.
- Another advantage of the invention is that the natural biodegradable polysaccharide-based coatings are more resistant to hydrolytic degradation than other biodegradable polymers, such as poly(lactides).
- Degradation of the natural biodegradable polysaccharides of the invention are primarily enzyme-mediated, with minimal or no hydrolysis of the natural biodegradable polysaccharide occurring when a natural biodegradable polysaccharide-containing coating is prepared under ambient conditions. This allows the natural biodegradable polysaccharide-based coatings to remain substantially stable (for example, resistant to degradation) prior to placing the coated-article in vivo.
- a natural biodegradable polysaccharide coated article can be manipulated in a non-biological, aqueous-based-medium without risk that the coating will prematurely degrade due to non-enzyme-mediatated hydrolysis.
- Other coatings that are based on biodegradable polymers such as poly(lactide) or poly(lactide-co-glycolide) are subject to hydrolysis even at relatively neutral pH ranges (e.g., pH 6.5 to 7.5) and therefore do not offer this advantage.
- the invention includes natural biodegradable polysaccharide-containing compositions, coatings, articles, and methods of preparing such that have the advantage of providing stability in the presence of an aqueous environment.
- the invention provides a shelf-stable composition for preparing a biodegradable coating, the shelf stable composition comprising a natural biodegradable polysaccharide comprising coupling groups.
- shelf stable composition comprising a natural biodegradable polysaccharide comprising coupling groups.
- the invention also provides methods for preparing a biodegradable coating comprising preparing a biodegradable coating composition comprising a natural biodegradable polysaccharide comprising coupling group; storing the coating composition for an amount of time; and then using the coating composition to prepare a biodegradable coating or a biodegradable article.
- the biodegradable article is formed in situ, for example, by promoting the polymerization of the natural biodegradable polysaccharide within the body.
- one or more bioactive agents and/or microparticles can be added before or after storage of the coating composition.
- the invention also provides the advantage of being able to perform methods wherein the natural biodegradable polysaccharide is subject to exposure to an aqueous solution without risking significant degradation of the natural biodegradable polysaccharide.
- the natural biodegradable polysaccharide may be contacted with an aqueous solution in a synthetic or post-synthetic step, including addition synthesis reactions and purification steps, or a coating that includes the natural biodegradable polysaccharide can be contacted with an aqueous solution in, for example, a sterilization step or a step that involves incorporation of a bioactive agent into the biodegradable coating.
- the invention relates to the stability of an article or a coating that is formed on an article.
- the invention provides a method comprising obtaining an article formed from, or having a coating comprising, a natural biodegradable polysaccharide, and then contacting the article with an aqueous solution for a period of time wherein the article or coating remains predominantly stable in the solution.
- the aqueous solution can be, for example, a storage solution, a solution that is used to hydrate the surface of the coated device, or an aqueous sterilization solution.
- Degradation of the natural biodegradable polysaccharide-containing coating or article may commence when placed in contact with a body fluid, which may include natural biodegradable polysaccharide-degrading enzymes, such as carbohydrases.
- the invention also provides a useful way to deliver larger hydrophilic bioactive agents, such as polypeptides, nucleic acids, and polysaccharides, as well as viral particles and cells from the biodegradable article, or biodegradable coating on a surface, such as a medical device or a coating on a surface thereof.
- biodegradable coating on a surface, such as a medical device or a coating on a surface thereof.
- the use of non-degrading drug delivery matrices may not be useful for the delivery of these larger bioactive agents if they are too large to diffuse out of the matrix.
- an article or a coating that includes a matrix of the natural biodegradable polysaccharide having a bioactive agent can be placed or formed in the body, and as the matrix degrades the bioactive agent is gradually released from the matrix.
- the bioactive agent has a molecular weight of about 10,000 Da or greater.
- the invention provides a drug-releasing biodegradable article, coating, or composition
- a drug-releasing biodegradable article, coating, or composition comprising (i) a natural biodegradable polysaccharide, preferably selected from amylose and maltodextrin, comprising an ethylenically unsaturated group, (ii) an initiator, and (iii) a bioactive agent selected from the group of polypeptides, polynucleotides, and polysaccharides.
- a coated surface is prepared on a medical device, such as a stent or catheter.
- the methods include disposing in one or more steps the following reagents on a surface: (a) an initiator, (b) a natural biodegradable polysaccharide, preferably selected from amylose and maltodextrin, comprising an ethylenically unsaturated group, and (c) a bioactive agent.
- the initiator is activated to crosslink a plurality of natural biodegradable polysaccharides comprising ethylenically unsaturated groups that are present in the composition, thereby forming a coating on the surface that includes the bioactive agent.
- the initiator can be first disposed on the surface, followed by disposing the natural biodegradable polysaccharide and bioactive agent on the layer of initiator.
- the initiator, natural biodegradable polysaccharide, and bioactive agent are mixed and disposed together on the surface.
- the invention provides a method for delivery of a bioactive agent, or more than one bioactive agent, to a subject.
- the method comprises the steps of providing a coated article to a subject, the coated article having a biodegradable coating which comprises a plurality of natural biodegradable polysaccharides associated via coupling groups, and bioactive agent.
- the coated article is then exposed to a carbohydrase to promote the degradation of the coating and release of the bioactive agent.
- a biodegradable coating or article including amylose and/or maltodextrin polymers can be exposed to an ⁇ -amylase to promote degradation of the coating and release of the bioactive agent.
- the step of exposing can be performed by placing the biodegradable coating or article in a patient. In the absence of the carbohydrase there is substantially no release of the bioactive agent.
- the bioactive agent comprises a polypeptide, such as an antibody or an antibody fragment.
- the methods of the invention can be used to prepare coatings wherein an amount of bioactive agent in the range of 1% to 17% of the total amount of bioactive agent present in the coating is released from the coating within a period of 2 days, and coatings wherein an amount of bioactive agent in the range of 1% to 20% of the total amount of bioactive agent present in the coating is released from the coating within a period of 8 days.
- the bioactive agent is delivered from a medical implant having a biodegradable body member which comprises a plurality of natural biodegradable polysaccharide associated via pendent coupling groups, the body member also including a bioactive agent.
- the medical implant is then exposed to a carbohydrase to promote the degradation of the implant and release of the bioactive agent.
- the methods of the invention can be used to prepare medical implants wherein an amount of bioactive agent in the range of 1% to 17% of the total amount of bioactive agent present in the medical implant is released within a period of 8 days, medical implants wherein an amount of bioactive agent in the range of 1% to 41% of the total amount of bioactive agent present in the medical implant is released within a period of 14 days, and medical implants wherein an amount of bioactive agent in the range of 1% to 60% of the total amount of bioactive agent present in the medical implant is released within a period of 21 days.
- a carbohydrase can be administered to a subject, or the carbohydrase can be provided to a portion of the article, wherein the carbohydrase is released from the portion and locally causes the degradation of the coating.
- the coatings can also have favorable bioactive agent-releasing properties when the coated article has been placed in the body.
- the present invention provides an overall improvement in terms of providing coatings for implantable medical articles.
- Articles that are fabricated from the biodegradable polysaccharides can have many of the same beneficial surface characteristics as provided by the biodegradable polysaccharide coatings.
- the natural biodegradable polysaccharide is modified with a hydrophobic moiety in order to provide a biodegradable matrix having hydrophobic properties. Therefore, a biodegradable coating or article can be formed from natural biodegradable polysaccharide comprising one or more pendent coupling groups and one or more pendent hydrophobic moieties. Exemplary hydrophobic moieties include fatty acids and derivatives thereof, and C 2 -C 18 alkyl chains.
- modification of the natural biodegradable polysaccharide allows for preparation of coatings or articles that are biodegradable and that can release a hydrophobic bioactive agent.
- the hydrophobic moiety pendent from the natural biodegradable has properties of a bioactive agent. Upon degradation of the matrix, the hydrophobic moiety can be hydrolyzed from the natural biodegradable polymer and released to provide a therapeutic effect.
- a therapeutically useful hydrophobic moiety is butyric acid.
- the invention provides methods and articles for improving the stability of a bioactive agent that is delivered from a coating or an article by utilizing a natural biodegradable non-reducing polysaccharide.
- the non-reducing polysaccharide can provide an inert matrix and thereby improve the stability of sensitive bioactive agents, such as proteins and enzymes.
- the article or coating can include a matrix having a plurality of natural biodegradable non-reducing polysaccharides along with a bioactive agent, such as a polypeptide.
- An exemplary non-reducing polysaccharide comprises polyalditol.
- Biodegradable non-reducing polysaccharides can very useful for formulating coatings or articles that release the bioactive agent over a prolonged period of time.
- coatings or articles that provide desired properties (for example, bioactive agent release, wettability, etc.), their actual preparation can be challenging.
- desired properties for example, bioactive agent release, wettability, etc.
- the use of some polysaccharides for preparing coatings or articles may result in products that are unsuitable for use.
- some polysaccharide-based coatings including those made from starch-based materials, have the potential to be overly brittle and inflexible. While these properties may be suitable for pharmaceutical capsules or tablets they are generally undesirable as properties for coatings or articles, such as bioactive agent releasing or sealant coatings, or medical implants.
- the present invention demonstrates the preparation of articles and coatings that include natural biodegradable polysaccharides that are suitable for in vivo use. These products display excellent physical characteristics and are suitable for use in applications wherein a particular function, such as bioactive agent delivery or a sealant function is desired.
- coatings or articles can be prepared having viscoelastic properties.
- the coating or article has an elastic modulus value in the range of 27 kPa to 30 kPa.
- the coatings of the present invention can have desirable surface properties that include elasticity and wettability, in addition to being biodegradable. Also, it has surprisingly been discovered that the coatings demonstrate excellent lubricity, which can provide distinct advantages for short term and single use devices. Therefore, in one aspect, the invention presents a method for providing lubricity to an article surface comprising the steps of disposing a composition comprising a plurality of natural biodegradable polysaccharides comprising pendent coupling groups and activating the coupling groups to promote association of the plurality of natural biodegradable polysaccharides and formation of a lubricious coating on the article surface.
- the coatings can be formed on the surfaces of medical articles, including those designed for single use or for short-term use.
- a lubricious coating can be formed on a catheter.
- Coatings including natural biodegradable polysaccharides can be prepared to provide a lubricity of 20 g or less, and can also be prepared to provide a lubricity of 15 g or less, or 10 g or less, as based on friction testing.
- the coatings were also shown to be highly durable, as lubricity was maintained during multiple cycles of friction testing. Methods utilizing a photoinitiator have been shown to provide coatings with both excellent lubricity and durability.
- the methods of preparing the compositions for fabrication of articles and/or coated surfaces do not require the use of organic solvents.
- organic solvents can be physically hazardous. Use of organic solvents can potentially destroy the activity of a bioactive agent that can be optionally included in the natural biodegradable polysaccharide-based composition.
- the starting materials in particular the natural biodegradable polysaccharides having pendent coupling groups.
- the natural biodegradable polysaccharides have pendent polymerizable groups, such as ethylenically unsaturated groups.
- the degradable polymerizable polymers are formed by reacting a natural biodegradable polysaccharide with a compound comprising an ethylenically unsaturated group.
- a natural biodegradable polysaccharide is reacted with a compound including an ethylenically unsaturated group and an isocyanate group.
- a natural biodegradable polysaccharide is treated with an oxidizing agent to form a reactive aldehyde species on the polysaccharide and then reacted with a compound comprising an ethylenically unsaturated group and an amine group.
- Polysaccharide macromers were shown to have excellent matrix forming capabilities.
- Synthesis can be carried out to provide the natural biodegradable polysaccharide with a desired quantity of pendent coupling groups. It has been found that use of a natural biodegradable polysaccharide having a predetermined amount of the coupling groups allows for the formation of a coating or an article having desirable physical characteristics (for example, the coatings are not brittle). Therefore, in some aspects, the invention provides natural biodegradable polysaccharides having an amount of pendent coupling groups of about 0.7 ⁇ moles of coupling group per milligram of natural biodegradable polysaccharide.
- the amount of coupling group per natural biodegradable polysaccharide is in the range of about 0.3 ⁇ moles/mg to about 0.7 ⁇ moles/mg.
- amylose or maltodextrin can be subject to a synthesis reaction with a compound having an ethylenically unsaturated group to provide an amylose or maltodextrin macromer having a ethylenically unsaturated group load level in the range of about 0.3 ⁇ moles/mg to about 0.7 ⁇ moles/mg.
- an initiator is used to promote the formation of the natural biodegradable polysaccharide matrix for article or coating formation.
- the initiator can be an independent compound or a pendent chemical group used to activate the coupling group pendent from the natural biodegradable polymer and promote coupling of a plurality of natural biodegradable polymers.
- the coupling group pendent from the natural biodegradable polysaccharide is a polymerizable group
- the initiator can be used in a free radical polymerization reaction to promote crosslinking of the natural biodegradable polysaccharides together in the composition.
- the invention provides a biodegradable coating or article composition
- a biodegradable coating or article composition comprising (i) a natural biodegradable polysaccharide, preferably selected from amylose and maltodextrin, comprising a coupling group, (ii) an initiator, and (iii) a bioactive agent, wherein the coupling group is able to be activated by the initiator and promote crosslinking of a plurality of natural biodegradable polysaccharides.
- the initiator is independent of the natural biodegradable polysaccharide and in other aspects the initiator is pendent from the natural biodegradable polysaccharide.
- the natural biodegradable polysaccharide comprises an ethylenically unsaturated group.
- a photoinitiator is used, such as a photoinitiator that is activated by light wavelengths having no or a minimal effect on the bioactive agent present in the composition.
- the initiator includes an oxidizing agent/reducing agent pair, a “redox pair,” to drive polymerization of the biodegradable polysaccharide.
- a redox pair to drive polymerization of the biodegradable polysaccharide.
- the oxidizing agent and reducing agent are combined in the presence of the biodegradable polysaccharide.
- One benefit of using a redox pair is that, when combined, the oxidizing agent and reducing agent can provide a particularly robust initiation system. This is advantageous as it can promote the formation of a matrix, for example, useful for coating or article preparation, from biodegradable polysaccharide compositions having a relatively low viscosity.
- a low viscosity composition can be passed through a small gauge delivery conduit with relative ease to provide the composition that can polymerize in situ.
- the viscosity of the composition is above about 5 centi Poise (cP), or about 10 cP or greater. In other aspects of the invention the viscosity of the composition is between about 5 cP or 10 cP and about 700 cP, and in some aspects between about 5 cP or 10 cP and about 250 cP. In some aspects the viscosity of the composition is above about 5 cP or 10 cP and the biodegradable polysaccharides in the composition have an average molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, or 50,000 Da or less.
- a method for preparing a coating or article can include the steps of (a) providing a first composition that includes a natural biodegradable polysaccharide comprising a coupling group and a first member of a redox pair (for example, the oxidizing agent) and then (b) mixing the first composition with second composition that includes a second member of the redox pair (for example, the reducing agent).
- the second composition includes a natural biodegradable polysaccharide.
- the first composition can include (a) a natural biodegradable polysaccharide having a coupling group and an oxidizing agent and the second composition can include a (b) natural biodegradable polysaccharide having a coupling group and a reducing agent.
- the final composition when the first composition is combined with the second composition, the final composition can be about 5 cP or greater.
- the oxidizing agent can be selected from inorganic or organic oxidizing agents, including enzymes; the reducing agent can be selected from inorganic or organic reducing agents, including enzymes.
- Exemplary oxidizing agents include peroxides, including hydrogen peroxide, metal oxides, and oxidases, such as glucose oxidase.
- Exemplary reducing agents include salts and derivatives of electropositive elemental metals such as Li, Na, Mg, Fe, Zn, Al, and reductases.
- the reducing agent is present in the composition at a concentration of 2.5 mM or greater when mixed with the oxidizing agent.
- Other reagents, such as metal or ammonium salts of persulfate can be present in the composition to promote polymerization of the biodegradable polysaccharide.
- a coating or article formed using redox polymerization can therefore comprise a plurality of natural biodegradable polysaccharides associated via polymerized groups, a reduced oxidizing agent, and an oxidized reducing agent.
- the biodegradable coated layer is formed article having a first coated layer comprising a synthetic polymer.
- an alternative method for forming a coating includes disposing in two or more steps at least the following reagents on a surface: (a) a natural biodegradable polysaccharide comprising a first coupling group, (b) a natural biodegradable polysaccharide comprising a second coupling group that is reactive with the first coupling group, and (c) a bioactive agent.
- reagents (a) and (b) are reactive with each other and are disposed separately on the surface but can individually include reagent (c).
- reagent (a) is first disposed on the surface and then a mixture comprising reagent (b) and (c) is then disposed on reagent (a).
- Reagent (a) reacts with (b) to link the natural biodegradable polysaccharides together to form a coating that includes reagent (c), the bioactive agent.
- An article can be formed in a similar manner, for example, by a method that includes combining (a) a natural biodegradable polysaccharide comprising a first coupling group with (b) a natural biodegradable polysaccharide comprising a second coupling group that is reactive with the first coupling group, and (c) a bioactive agent.
- the article can be partially or fully formed when reagent (a) reacts with (b) to link the natural biodegradable polysaccharides together to form the article, which includes reagent (c), the bioactive agent.
- the present invention employs the use of biodegradable microparticles that include a bioactive agent and a natural biodegradable polysaccharide, such as amylose and maltodextrin that have pendent coupling groups.
- the microparticles are used in association with the natural biodegradable polysaccharides to prepare a biodegradable, bioactive agent-releasing coating for the surface of medical devices.
- a medical device having a coating that includes a crosslinked matrix of natural biodegradable polysaccharides and biodegradable microparticles having a bioactive agent can be placed in the body, and as the biodegradable microparticles degrade the bioactive agent is gradually released from the coating.
- the natural biodegradable polysaccharide matrix provides the ability to associate the biodegradable microparticles with the surface of the coated device.
- the biodegradable microparticles are dispersed in the natural biodegradable polysaccharide matrix.
- Such coatings can be formed by disposing a mixture of (a) biodegradable microparticles having a bioactive agent and (b) natural biodegradable polysaccharides having pendent coupling groups, disposing the mixture on a surface, and then treating the composition to form a coated layer wherein the biodegradable microparticles are dispersed within the matrix.
- the coating is formed by disposing the biodegradable microparticles independently of the natural biodegradable polysaccharide having pendent coupling groups.
- the biodegradable microparticles can be present predominantly one face of the layer that is formed from the natural biodegradable polysaccharide and a microparticle-matrix interface can be formed.
- the methods include disposing in one or more steps the following components on a surface: (a) an initiator, (b) a natural biodegradable polysaccharide, preferably selected from amylose and maltodextrin, comprising a coupling group, and (c) biodegradable microparticles comprising a bioactive agent.
- the initiator is activated to couple a plurality of natural biodegradable polysaccharide polymers that are present in the composition, thereby forming a natural biodegradable polysaccharide matrix on the surface that is associated with the biodegradable microparticles having the bioactive agent.
- the method includes the steps of (i) disposing a composition comprising (a) a natural biodegradable polysaccharide having a coupling group, (b) an initiator, and (c) biodegradable microparticles comprising a bioactive agent on a surface; and (ii) activating the initiator to provide a coated composition having the natural biodegradable polysaccharide and the biodegradable microparticles having the bioactive agent on the surface.
- the initiator can be disposed independently of the natural biodegradable polysaccharide.
- microparticles having a bioactive agent in the natural biodegradable polysaccharide-containing coating, the invention also provides a way to effectively and efficiently prepare a variety of drug-delivery coatings.
- the use of microparticles offers the ability to easily prepare coatings having one or more bioactive agents present in desired amounts in the coating.
- Such coatings can be prepared by obtaining biodegradable microparticles that have a bioactive agent and then forming a coating that includes the microspheres associated with the natural biodegradable polysaccharide matrix.
- different microparticles having different bioactive agents can be included in the coating in desired amounts to provide a bioactive agent-releasing coating that is able to release a desired combination of bioactive agents in desired amounts. This is a particular advantage when using bioactive agents that are typically not compatible in the same composition (for example, bioactive agents that have different physical properties).
- Microparticles can also be included in articles formed from the natural biodegradable polysaccharide.
- microparticles can be included in an implantable medical article formed from the natural biodegradable polysaccharides of the invention, or can be included in an article that is formed in situ.
- the presence of biodegradable microparticles in articles can offer many of the advantages that are offered by the presence of the microparticles in the coatings.
- the present invention provides compositions and methods for preparing sealant materials that are particularly useful in connection with implantable medical articles having a porous surface, such as grafts, patches, and wound dressings.
- inventive compositions can be used to prepare a sealant coating for implantable medical articles, particularly implantable medical articles that include a porous surface.
- the sealant coating can provide a barrier to the movement of body fluids, such as blood, near the surface of the coated article.
- the natural biodegradable polysaccharide-based sealant coating can provide hemostasis at the article surface by formation of a tight seal.
- the natural biodegradable polysaccharide in the sealant coating degrades and a tissue layer is formed as the sealant coating is replaced by cells and other factors involved in tissue repair.
- natural biodegradable polysaccharide degradation products such as naturally occurring mono- or disaccharides, for example, glucose
- the sealant coating which can be considered an ideal in vivo degradation product because it is commonly found in the body and may also be utilized by the cells involved in tissue repair during the degradation/infiltration process.
- infiltrated tissue growth replaces the function of the natural biodegradable polysaccharide-containing sealant coating.
- Another particular advantage of the invention is that release of glucose reduces the likelihood that the process of natural biodegradable polysaccharide degradation and tissue infiltration will promote a strong inflammatory response. This is because the natural biodegradable polysaccharide-based sealant coating can degrade into materials that are non-antigenic or that have low antigenicity. Another advantage is that the degradation products are free of other materials that may cause disease, such as microbial, viral, or prionic materials potentially present in animal-derived preparations (such as bovine collagen preparations).
- sealant compositions of the invention which include natural biodegradable polysaccharides, such as amylose or maltodextrin polymers, that can be coupled together to form a matrix (at least a portion of the sealant coating) on the medical article, can include a bioactive agent, which can be released as the sealant coating degrades.
- natural biodegradable polysaccharides such as amylose or maltodextrin polymers
- the invention provides a biodegradable sealant composition
- a biodegradable sealant composition comprising (i) a natural biodegradable polysaccharide comprising a coupling group, and (ii) an initiator, wherein the coupling group is able to be activated by the initiator and promote coupling of a plurality of natural biodegradable polysaccharides.
- the natural biodegradable polysaccharide is a polymer such as amylose or maltodextrin.
- the sealant composition can also include a bioactive agent.
- the initiator can be independent of the natural biodegradable polysaccharide, pendent from the natural biodegradable polysaccharide polymer, or both pendent and independent of the natural biodegradable polysaccharide polymer.
- the invention also provides methods for preparing a surface having a sealant coating.
- the sealant coated surface is prepared on a medical article or article having a porous surface.
- the methods include disposing in one or more steps the following reagents on a surface: (a) an initiator, and (b) a natural biodegradable polysaccharide comprising a coupling group.
- a bioactive agent is also disposed on the surface.
- the bioactive agent is a prothrombotic or procoagulant factor.
- the initiator is activated to couple the natural biodegradable polysaccharides that are present in the composition, thereby forming a natural biodegradable polysaccharide coating on the surface that includes the bioactive agent.
- the natural biodegradable polysaccharide is contacted with the initiator and the initiator is activated to promote the coupling of two or more natural biodegradable polysaccharides via their coupling groups.
- the natural biodegradable polysaccharide includes a polymerizable group, such as an ethylenically unsaturated group, and initiator is capable of initiating free radical polymerization of the polymerizable groups.
- the invention also provides alternative methods for preparing a sealant coating on the surface of an article.
- the methods include disposing at least the following reagents on a surface: (a) a natural biodegradable polysaccharide comprising a first coupling group and (b) a natural biodegradable polysaccharide comprising a second coupling group, where in the second coupling group is reactive with the first coupling group.
- reagents (a) and (b) are reactive with each other to couple the natural biodegradable polysaccharide, or (a) and/or (b) can be treated to be made reactive with each other.
- (a) and (b) are disposed separately on the surface to form the sealant coating.
- the natural biodegradable polysaccharide can be the same types of polymers of different types of polymers.
- the first coupling group and second coupling group can be a pair of chemical groups that are reactive with one another, preferably specifically reactive.
- the groups can also become reactive with each other upon addition of a particular agent to the mixture of natural biodegradable polysaccharide having different reactive groups.
- FIG. 1 is a graph of cumulative BSA release from maltodextrin-acrylate filaments treated with amylase, over a period of time.
- FIG. 2 is a graph of cumulative BSA release from maltodextrin-acrylate/photo-PVP-coated PEBAX rod treated with amylase, over a period of time.
- FIG. 3 is a graph of cumulative absorbance values of active and total IgG Fab fragment release from maltodextrin-acrylate filaments treated with amylase, over a period of time.
- FIG. 4 is a graph of cumulative absorbance values of active and total IgG release from maltodextrin-acrylate(redox)/photo-PVP-coated stainless steel rods treated with amylase, over a period of time.
- FIG. 5 is a graph of cumulative absorbance values of active and total IgG release from maltodextrin-acrylate(photoinitiation)/photo-PVP-coated stainless steel rods treated with amylase and percent degradation of the maltodextrin-acrylate coating, over a period of time.
- FIG. 6 is a graph of cumulative absorbance values of active and total IgG release from a maltodextrin-acrylate filament treated with amylase and percent degradation of the filament, over a period of time.
- FIG. 7 is a graph of modulus of a maltodextrin-acrylate matrix formed via REDOX polymerization, over a period of time.
- FIG. 8 is a graph of repetitive force testing of maltodextrin-acrylate coated PEBAX rods versus synthetic polymer-coated PEBAX rods.
- the invention provides methods of preparing biodegradable coatings that release bioactive agents from the surface of medical devices.
- the compositions and methods of the present invention are particularly useful for coating surfaces of implantable medical devices, such as stents and catheters, and that are capable of releasing drugs from the device.
- the invention provides methods of preparing biodegradable articles, such as medical implants or in vivo formed matrices.
- the biodegradable articles can also be used for the release of bioactive agents, and in this manner can function as bioactive agent-releasing implants or depots.
- the biodegradable articles of the invention biodegrade within a period that is acceptable for the desired application.
- the biodegradable article is a medical implant that provides mechanical properties at the implantation site and maintains these mechanical properties until they are no longer needed. After this period of time has elapsed, the medical implant is degraded to an extent that the properties are no longer provided by the medical implant, and the biodegradable components can be absorbed and/or excreted by the body. In some embodiments, the medical implant slowly degrades and transfers stress at the appropriate rate to surrounding tissues as these tissues heal and can accommodate the stress once borne by the medical device.
- the biodegradable coating or article includes a natural biodegradable polysaccharide having a coupling group.
- exemplary natural biodegradable polysaccharides include amylose and maltodextrin.
- the present invention provides biodegradable coatings having excellent surface characteristics and that can provide a suitable vehicle for the delivery of bioactive agents. These biodegradable coatings can be disposed on medical devices having a variety of biomaterial surfaces.
- a coating is formed on a device that includes a biodegradable matrix and biodegradable microparticles, the biodegradable microparticles including one or more bioactive agents.
- the biodegradable material used to form the matrix includes a natural biodegradable polysaccharide as a component.
- natural biodegradable polysaccharides such as amylose and maltodextrin are coupled to each other and the biodegradable microparticles are associated with the matrix.
- a sealant coating is formed on a device.
- the sealant coating includes a biodegradable matrix and optionally one or more bioactive agents, such as prothrombotic agents.
- the sealant coating of the invention can, at least initially, provide a barrier on the porous surface that is not permeable to fluids within the body. Gradually, the sealant coating degrades and its function is replaced by tissue that infiltrates the porous surface. Therefore, the sealant coating has particular properties, such as biodegradability and relative impermeability (i.e., relative to the degradation of the sealant coating).
- the sealant coating can also be compliant and/or conformal, and can have properties such as flexibility, elasticity, and bendability.
- impermeable used in relation to the function of the sealant coating, refers to a significant reduction in the transmission of bulk liquid or fluids through the substrate which the sealant coating is associated with.
- the sealant coating can be impermeable to the transmission of blood.
- the impermeability can be maintained as the natural biodegradable polysaccharide-based sealant coating degrades, and is replaced by tissue.
- a “natural biodegradable polysaccharide” refers to a non-synthetic polysaccharide that is capable of being enzymatically degraded but that is generally non-enzymatically hydrolytically stable.
- Natural biodegradable polysaccharides include polysaccharide and/or polysaccharide derivatives that are obtained from natural sources, such as plants or animals.
- Natural biodegradable polysaccharides include any polysaccharide that has been processed or modified from a natural biodegradable polysaccharide (for example, maltodextrin is a natural biodegradable polysaccharide that is processed from starch).
- Exemplary natural biodegradable polysaccharides include hyaluronic acid, starch, dextran, heparin, chondroitin sulfate, dermatan sulfate, heparan sulfate, keratan sulfate, dextran sulfate, pentosan polysulfate, and chitosan.
- Preferred polysaccharides are low molecular weight polymers that have little or no branching, such as those that are derived from and/or found in starch preparations, for example, amylose and maltodextrin. Therefore, the natural biodegradable polysaccharide can be a substantially non-branched or non-branched poly(glucopyranose) polymer.
- natural biodegradable polysaccharides having an average molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, or 50,000 Da or less. It is also preferred that the natural biodegradable polysaccharides have an average molecular weight of 500 Da or greater.
- a particularly preferred size range for the natural biodegradable polysaccharides is in the range of about 1000 Da to about 10,000 Da. Natural biodegradable polysaccharides of particular molecular weights can be obtained commercially or can be prepared.
- the decision of using natural biodegradable polysaccharides of a particular size range may depend on factors such as the physical characteristics of the coating composition (e.g., viscosity), the desired rate of degradation of the coating, the presence of other optional moieties in the coating composition, for example, bioactive agents, etc.
- amylose or “amylose polymer” refers to a linear polymer having repeating glucopyranose units that are joined by ⁇ -1,4 linkages. Some amylose polymers can have a very small amount of branching via ⁇ -1,6 linkages (about less than 0.5% of the linkages) but still demonstrate the same physical properties as linear (unbranched) amylose polymers do. Generally amylose polymers derived from plant sources have molecular weights of about 1 ⁇ 10 6 Da or less. Amylopectin, comparatively, is a branched polymer having repeating glucopyranose units that are joined by ⁇ -1,4 linkages to form linear portions and the linear portions are linked together via ⁇ -1,6 linkages. The branch point linkages are generally greater than 1% of the total linkages and typically 4%-5% of the total linkages. Generally amylopectin derived from plant sources have molecular weights of 1 ⁇ 10 7 Da or greater.
- Amylose can be obtained from, or is present in, a variety of sources. Typically, amylose is obtained from non-animal sources, such as plant sources. In some aspects, a purified preparation of amylose is used as starting material for the preparation of the amylose polymer having coupling groups. In other aspects, as starting material, amylose can be used in a mixture that includes other polysaccharides.
- starch preparations having a high amylose content, purified amylose, synthetically prepared amylose, or enriched amylose preparations can be used in the preparation of amylose having the coupling groups.
- amylose is typically present along with amylopectin, which is a branched polysaccharide.
- starch preparations having high amylose content, purified amylose, synthetically prepared amylose, or enriched amylose preparations can be used in the preparation of amylose polymer having the coupling groups.
- the composition includes a mixture of polysaccharides including amylose wherein the amylose content in the mixture of polysaccharides is 50% or greater, 60% or greater, 70% or greater, 80% or greater, or 85% or greater by weight.
- the composition includes a mixture of polysaccharides including amylose and amylopectin and wherein the amylopectin content in the mixture of polysaccharides is 30% or less, or 15% or less.
- non-retrograding starches such as waxy starch
- the amount of amylopectin present in a starch may also be reduced by treating the starch with amylopectinase, which cleaves ⁇ -1,6 linkages resulting in the debranching of amylopectin into amylose.
- a synthesis reaction can be carried out to prepare an amylose polymer having pendent coupling groups (for example, amylose with pendent ethylenically unsaturated groups) and steps may be performed before, during, and/or after the synthesis to enrich the amount of amylose, or purify the amylose.
- pendent coupling groups for example, amylose with pendent ethylenically unsaturated groups
- Amylose of a particular size, or a combination of particular sizes can be used.
- the choice of amylose in a particular size range may depend on the application, for example, the type of surface coated or the porosity of the surface.
- amylose having an average molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, 50,000 Da or less, preferably greater than 500 Da, or preferably in the range of about 1000 Da to about 10,000 Da is used.
- Amylose of particular molecular weights can be obtained commercially or can be prepared. For example, synthetic amyloses with average molecular masses of 70, 110, 320, and 1,000 kDa can be obtained from Nakano Vinegar Co., Ltd. (Aichi, Japan).
- amylose of a particular size range may depend on factors such as the physical characteristics of the coating composition (e.g., viscosity), the desired rate of degradation of the coating, the presence of other optional moieties in the coating composition (for example, bioactive agents, etc.), etc.
- Maltodextrin is typically generated by hydrolyzing a starch slurry with heat-stable ⁇ -amylase at temperatures at 85-90° C. until the desired degree of hydrolysis is reached and then inactivating the ⁇ -amylase by a second heat treatment.
- the maltodextrin can be purified by filtration and then spray dried to a final product.
- DE dextrose equivalent
- a starch preparation that has been totally hydrolyzed to dextrose (glucose) has a DE of 100, where as starch has a DE of about zero.
- a DE of greater than 0 but less than 100 characterizes the mean-average molecular weight of a starch hydrolysate, and maltodextrins are considered to have a DE of less than 20.
- Maltodextrins of various molecular weights, for example, in the range of about 500-5000 Da are commercially available (for example, from CarboMer, San Diego, Calif.).
- a non-reducing polysaccharide can provide an inert matrix thereby improving the stability of sensitive bioactive agents, such as proteins and enzymes.
- a non-reducing polysaccharide refers to a polymer of non-reducing disaccharides (two monosaccharides linked through their anomeric centers) such as trehalose ( ⁇ -D-glucopyranosyl ⁇ -D-glucopyranoside) and sucrose ( ⁇ -D-fructofuranosyl ⁇ -D-glucopyranoside).
- An exemplary non-reducing polysaccharide comprises polyalditol which is available from GPC (Muscatine, Iowa).
- the polysaccharide is a glucopyranosyl polymer, such as a polymer that includes repeating (1 ⁇ 3)O- ⁇ -D-glucopyranosyl units.
- the coating compositions can include natural biodegradable polysaccharides that include chemical modifications other than the pendent coupling group.
- modified amylose having esterified hydroxyl groups can be prepared and used in sealant coating compositions in association with the methods of the invention.
- Other natural biodegradable polysaccharides having hydroxyl groups may be modified in the same manner. These types of modifications can change or improve the properties of the natural biodegradable polysaccharide making for a coating composition that is particularly suitable for a desired application.
- Many chemically modified amylose polymers, such as chemically modified starch have at least been considered acceptable food additives.
- modified natural biodegradable polysaccharides refers to chemical modifications to the natural biodegradable polysaccharide that are different than those provided by the coupling group or the initiator group.
- Modified amylose polymers having a coupling group (and/or initiator group) can be used in the compositions and methods of the invention.
- modified amylose is described.
- chemically modifying the hydroxyl groups of the amylose the physical properties of the amylose can be altered.
- the hydroxyl groups of amylose allow for extensive hydrogen bonding between amylose polymers in solution and can result in viscous solutions that are observed upon heating and then cooling amylose-containing compositions such as starch in solution (retrograding).
- the hydroxyl groups of amylose can be modified to reduce or eliminate hydrogen bonding between molecules thereby changing the physical properties of amylose in solution.
- the natural biodegradable polysaccharides can include one or more modifications to the hydroxyl groups wherein the modifications are different than those provided by coupling group.
- Modifications include esterification with acetic anhydride (and adipic acid), succinic anhydride, 1-octenylsuccinic anhydride, phosphoryl chloride, sodium trimetaphosphate, sodium tripolyphosphate, and sodium monophosphate; etherification with propylene oxide, acid modification with hydrochloric acid and sulfuric acids; and bleaching or oxidation with hydrogen peroxide, peracetic acid, potassium permanganate, and sodium hypochlorite.
- modified amylose polymers include carboxymethyl amylose, carboxyethyl amylose, ethyl amylose, methyl amylose, hydroxyethyl amylose, hydroxypropyl amylose, acetyl amylose, amino alkyl amylose, allyl amylose, and oxidized amylose.
- modified amylose polymers include succinate amylose and oxtenyl succinate amylose.
- the natural biodegradable polysaccharide is modified with a hydrophobic moiety in order to provide a biodegradable matrix having hydrophobic properties.
- exemplary hydrophobic moieties include those previously listed, fatty acids and derivatives thereof, and C 2 -C 18 alkyl chains.
- a polysaccharide, such as amylose or maltodextrin, can be modified with a compound having a hydrophobic moiety, such as a fatty acid anhydride.
- the hydroxyl group of a polysaccharide can also cause the ring opening of lactones to provide pendent open-chain hydroxy esters.
- the hydrophilic moiety pendent from the natural biodegradable has properties of a bioactive agent.
- the hydrophilic moiety can be hydrolyzed from the natural biodegradable polymer and released from the matrix to provide a therapeutic effect.
- a therapeutically useful hydrophilic moiety is butyric acid, which has been shown to elicit tumor cell differentiation and apoptosis, and is thought to be useful for the treatment of cancer and other blood diseases.
- the hydrophilic moiety that provides a therapeutic effect can also be a natural compound (such as butyric acid). Therefore, degradation of the matrix having a coupled therapeutic agent can result in all natural degradation products.
- a natural biodegradable polysaccharide that includes a coupling group is used to form an article or a coating on the surface of a medical article.
- Other polysaccharides can also be present in the coating composition.
- the two or more natural biodegradable polysaccharides are used to form an article or a coating on the surface of a medical article.
- examples include amylose and one or more other natural biodegradable polysaccharide(s), and maltodextrin and one or more other natural biodegradable polysaccharide(s); in one aspect the composition includes a mixture of amylose and maltodextrin, optionally with another natural biodegradable polysaccharide.
- amylose or maltodextrin is the primary polysaccharide.
- the composition includes a mixture of polysaccharides including amylose or maltodextrin and the amylose or maltodextrin content in the mixture of polysaccharides is 50% or greater, 60% or greater, 70% or greater, 80% or greater, or 85% or greater by weight.
- Purified or enriched amylose preparations can be obtained commercially or can be prepared using standard biochemical techniques such as chromatography. In some aspects, high-amylose cornstarch can be used.
- “coupling group” can include (1) a chemical group that is able to form a reactive species that can react with the same or similar chemical group to form a bond that is able to couple the natural biodegradable polysaccharides together (for example, wherein the formation of a reactive species can be promoted by an initiator); or (2) a pair of two different chemical groups that are able to specifically react to form a bond that is able to couple the natural biodegradable polysaccharides together.
- the coupling group can be attached to any suitable natural biodegradable polysaccharide, including the amylose and maltodextrin polymers as exemplified herein.
- Contemplated reactive pairs include Reactive Group A and corresponding Reactive Group B as shown in the Table 1 below.
- a reactive group from group A can be selected and coupled to a first set of natural biodegradable polysaccharides and a corresponding reactive group B can be selected and coupled to a second set of natural biodegradable polysaccharides.
- Reactive groups A and B can represent first and second coupling groups, respectively. At least one and preferably two, or more than two reactive groups are coupled to an individual natural biodegradable polysaccharide polymer.
- the first and second sets of natural biodegradable polysaccharides can be combined and reacted, for example, thermochemically, if necessary, to promote the coupling of natural biodegradable polysaccharides and the formation of a natural biodegradable polysaccharide matrix.
- Reactive group A Reactive group B amine, hydroxyl, sulfhydryl N-oxysuccinimide (“NOS”) amine Aldehyde amine Isothiocyanate amine, sulfhydryl Bromoacetyl amine, sulfhydryl Chloroacetyl amine, sulfhydryl Iodoacetyl amine, hydroxyl Anhydride aldehyde Hydrazide amine, hydroxyl, carboxylic acid Isocyanate amine, sulfhydryl Maleimide sulfhydryl Vinylsulfone
- Amine also includes hydrazide (R—NH—NH 2 )
- a suitable coupling pair would be a natural biodegradable polysaccharide having an electrophilic group and a natural biodegradable polysaccharide having a nucleophilic group.
- An example of a suitable electrophilic-nucleophilic pair is N-hydroxysuccinimide-amine pair, respectively.
- Another suitable pair would be an oxirane-amine pair.
- the natural biodegradable polysaccharides of the invention include at least one, and more typically more than one, coupling group per natural biodegradable polysaccharide, allowing for a plurality of natural biodegradable polysaccharides to be coupled in linear and/or branched manner.
- the natural biodegradable polysaccharide includes two or more pendent coupling groups.
- the coupling group on the natural biodegradable polysaccharide is a polymerizable group.
- the polymerizable group can couple natural biodegradable polysaccharides together in the composition, thereby forming a biodegradable natural biodegradable polysaccharide matrix.
- a preferred polymerizable group is an ethylenically unsaturated group.
- Suitable ethylenically unsaturated groups include vinyl groups, acrylate groups, methacrylate groups, ethacrylate groups, 2-phenyl acrylate groups, acrylamide groups, methacrylamide groups, itaconate groups, and styrene groups. Combinations of different ethylenically unsaturated groups can be present on a natural biodegradable polysaccharide, such as amylose or maltodextrin.
- any suitable synthesis procedure can be used. Suitable synthetic schemes typically involve reaction of, for example, hydroxyl groups on the natural biodegradable polysaccharide, such as amylose or maltodextrin. Synthetic procedures can be modified to produce a desired number of coupling groups pendent from the natural biodegradable polysaccharide backbone. For example, the hydroxyl groups can be reacted with a coupling group-containing compound or can be modified to be reactive with a coupling group-containing compound. The number and/or density of acrylate groups can be controlled using the present method, for example, by controlling the relative concentration of reactive moiety to saccharide group content.
- the biodegradable polysaccharides have an amount of pendent coupling groups of about 0.7 ⁇ moles of coupling group per milligram of natural biodegradable polysaccharide.
- the amount of coupling group per natural biodegradable polysaccharide is in the range of about 0.3 ⁇ moles/mg to about 0.7 ⁇ moles/mg.
- amylose or maltodextrin can be reacted with an acrylate groups-containing compound to provide an amylose or maltodextrin macromer having a acrylate group load level in the range of about 0.3 ⁇ moles/mg to about 0.7 ⁇ moles/mg.
- an “initiator” refers to a compound, or more than one compound, that is capable of promoting the formation of a reactive species from the coupling group.
- the initiator can promote a free radical reaction of natural biodegradable polysaccharide having a coupling group.
- the initiator is a photoreactive group (photoinitiator) that is activated by radiation.
- the initiator can be an “initiator polymer” that includes a polymer having a backbone and one or more initiator groups pendent from the backbone of the polymer.
- the initiator is a compound that is light sensitive and that can be activated to promote the coupling of the amylose polymer via a free radical polymerization reaction. These types of initiators are referred to herein as “photoinitiators.” In some aspects it is preferred to use photoinitiators that are activated by light wavelengths that have no or a minimal effect on a bioactive agent if present in the composition. A photoinitiator can be present in a sealant composition independent of the amylose polymer or pendent from the amylose polymer.
- photoinitiation occurs using groups that promote an intra- or intermolecular hydrogen abstraction reaction.
- This initiation system can be used without additional energy transfer acceptor molecules and utilizing nonspecific hydrogen abstraction, but is more commonly used with an energy transfer acceptor, typically a tertiary amine, which results in the formation of both aminoalkyl radicals and ketyl radicals.
- an energy transfer acceptor typically a tertiary amine
- Examples of molecules exhibiting hydrogen abstraction reactivity and useful in a polymeric initiating system include analogs of benzophenone, thioxanthone, and camphorquinone.
- the photoinitiator includes one or more charged groups.
- the presence of charged groups can increase the solubility of the photoinitiator (which can contain photoreactive groups such as aryl ketones) in an aqueous system and therefore provide for an improved coating composition.
- Suitable charged groups include, for example, salts of organic acids, such as sulfonate, phosphonate, carboxylate, and the like, and onium groups, such as quaternary ammonium, sulfonium, phosphonium, protonated amine, and the like.
- a suitable photoinitiator can include, for example, one or more aryl ketone photogroups selected from acetophenone, benzophenone, anthraquinone, anthrone, anthrone-like heterocycles, and derivatives thereof; and one or more charged groups, for example, as described herein. Examples of these types of water-soluble photoinitiators have been described in U.S. Pat. No. 6,077,698.
- the photoinitiator is a compound that is activated by long-wavelength ultraviolet (UV) and visible light wavelengths.
- the initiator includes a photoreducible or photo-oxidizable dye.
- Photoreducible dyes can also be used in conjunction with a compound such as a tertiary amine. The tertiary amine intercepts the induced triplet producing the radical anion of the dye and the radical cation of the tertiary amine.
- Examples of molecules exhibiting photosensitization reactivity and useful as an initiator include acridine orange, camphorquinone, ethyl eosin, eosin Y, erythrosine, fluorescein, methylene green, methylene blue, phloxime, riboflavin, rose bengal, thionine, and xanthine dyes.
- Use of these types of photoinitiators can be particularly advantageous when a light-sensitive bioactive agent is included in the sealant coating.
- the invention provides a coating composition
- a coating composition comprising (i) a natural biodegradable polysaccharide comprising an ethylenically unsaturated group (ii) a photoinitiator selected from the group consisting of acridine orange, camphorquinone, ethyl eosin, eosin Y, erythrosine, fluorescein, methylene green, methylene blue, phloxime, riboflavin, rose bengal, thionine, and xanthine dyes, and (iii) a bioactive agent.
- a photoinitiator selected from the group consisting of acridine orange, camphorquinone, ethyl eosin, eosin Y, erythrosine, fluorescein, methylene green, methylene blue, phloxime, riboflavin, rose bengal, thionine, and xanthin
- Thermally reactive initiators can also be used to promote the polymerization ofnatural biodegradable polymers having pendent coupling groups.
- thermally reactive initiators include 4,4′azobis(4-cyanopentanoic acid), 2,2-azobis[2-(2-imidazolin-2-yl) propane]dihydrochloride, and analogs of benzoyl peroxide.
- Redox initiators can also be used to promote the polymerization of the natural biodegradable polymers having pendent coupling groups.
- combinations of organic and inorganic oxidizers, and organic and inorganic reducing agents are used to generate radicals for polymerization. A description of redox initiation can be found in Principles of Polymerization, 2 nd Edition, Odian G., John Wiley and Sons, pgs 201-204, (1981).
- the initiator can be included in a base coating and the natural biodegradable polysaccharide or composition that includes the natural biodegradable polysaccharide can be disposed on the base coating.
- a coated layer that includes the natural biodegradable polysaccharide can be formed on a coated layer that includes a synthetic polymer.
- the synthetic polymer can be a hydrophilic polymer such as poly(vinylpyrrolidone), poly(acrylamide), or copolymers thereof.
- the synthetic polymer is formed using photoreactive groups, such as photoreactive groups that are pendent from the synthetic polymer, which can be used to covalently bond the synthetic polymer to a surface of the article.
- the polymerization initiator is a polymer that includes an initiator group (herein referred to as an “initiator polymer”).
- the polymeric portion of the initiator polymer can be obtained or prepared to have particular properties or features that are desirable for use with acoating composition, such as a sealant coating composition.
- acoating composition such as a sealant coating composition.
- the polymeric portion of the initiator polymer can have hydrophilic or amphoteric properties, it can include pendent charged groups, or it can have groups that allow it to interact with a particular surface (this can depend on the type of surface to be coated).
- the polymer can change or improve the properties of the coating that is formed by the amylose polymer having coupling groups.
- the initiator polymer can change the elasticity, flexibility, wettability, or softness (or combinations thereof) of the coating formed on the surface.
- Certain polymers, as described herein, are useful as plasticizing agents for coatings that include natural biodegradable polysaccharides. Initiator groups can be added to these plasticizing polymers and used in the compositions and methods of the invention.
- an initiator can be pendent from a natural biodegradable polysaccharide. Therefore, the natural biodegradable polysaccharide is able to promote activation of polymerizable groups that are pendent from other natural biodegradable polysaccharides and promote the formation of a natural biodegradable polysaccharide matrix.
- the polymeric portion of the initiator polymer can include, for example, acrylamide and methacrylamide monomeric units, or derivatives thereof.
- the coating composition includes an initiator polymer having a photoreactive group and a polymeric portion selected from the group of acrylamide and methacrylamide polymers and copolymers.
- the initiator includes an oxidizing agent/reducing agent pair, a “redox pair,” to drive polymerization of the biodegradable polysaccharide.
- polymerization of the biodegradable polysaccharide is carried out upon combining one or more oxidizing agents with one or more reducing agents.
- Other compounds can be included in the composition to promote polymerization of the biodegradable polysaccharides.
- the oxidizing agent and reducing agent can provide a particularly robust initiation system and can drive the formation of a polymerized matrix of polysaccharides from a composition having a low viscosity.
- a polysaccharide composition with a low viscosity may be due to a low concentration of polysaccharide in the composition, a polysaccharide having a low average molecular weight, or combinations thereof.
- Matrix formation from a polysaccharide composition having a low viscosity is particularly advantageous in many applications, especially for in situ polymerization.
- a low viscosity polysaccharide composition is passed through a small gauge delivery conduit, such as a needle, wherein the redox pair causes the polymerization of the polysaccharides in situ.
- the viscosity of the composition is above about 5 cP, or about 10 cP or greater. In other aspects of the invention the viscosity of the composition is between about 5 cP or 10 cP and about 700 cP, or between about 5 cP or 10 cP and about 250 cP.
- the oxidizing agent is added to the reducing agent in the presence of the one or more biodegradable polysaccharides.
- a composition including a biodegradable polysaccharide and a reducing agent is added to a composition including an oxidizing agent, or a composition including a biodegradable polysaccharide and an oxidizing agent is added to a composition containing a reducing agent.
- One desirable method of preparing a matrix is to combine a composition including a biodegradable polysaccharide and an oxidizing agent with a composition including a biodegradable polysaccharide and a reducing agent.
- the terms “first composition” and “second composition” can be used.
- the viscosities of biodegradable polysaccharide in the first and second compositions can be the same or can be different. Generally, though, it has been observed that good mixing and subsequent matrix formation is obtained when the compositions have the same or similar viscosities. In this regard, if the same biodegradable polymer is used in the first and second compositions, the concentration of the biodegradable polymer may be the same or different.
- the oxidizing agent can be selected from inorganic or organic oxidizing agents, including enzymes; the reducing agent can be selected from inorganic or organic reducing agents, including enzymes.
- Exemplary oxidizing agents include peroxides, including hydrogen peroxide, metal oxides, and oxidases, including glucose oxidase.
- Exemplary reducing agents include salts and derivatives of electropositive elemental metals such as Li, Na, Mg, Fe, Zn, Al, and reductases.
- the reducing agent is present at a concentration of about 2.5 mM or greater when the reducing agent is mixed with the oxidizing agent. Prior to mixing, the reducing agent can be present in a composition at a concentration of, for example, 5 mM or greater.
- compositions can be present in the composition to promote polymerization of the biodegradable polysaccharide.
- Other polymerization promoting compounds can be included in the composition, such as metal or ammonium salts of persulfate.
- compositions and methods of the invention can include polymerization accelerants that can improve the efficiency of polymerization.
- useful accelerants include N-vinyl compounds, particularly N-vinyl pyrrolidone and N-vinyl caprolactam.
- Such accelerants can be used, for instance, at a concentration of between about 0.01% and about 5%, and preferably between about 0.05% and about 0.5%, by weight, based on the volume of the coating composition.
- an aqueous composition that includes the natural biodegradable polysaccharide, such as amylose or maltodextrin having pendent coupling groups, and a bioactive agent is obtained and used in the method of coating a surface.
- an aqueous composition is used to form an article. This composition can be prepared by mixing a bioactive agent, such as a water-soluble small molecule, a protein, or a nucleic acid, with the natural biodegradable polysaccharide.
- the natural biodegradable polysaccharide that includes a coupling group is used to form a coating on the surface of a medical device or to form an article.
- Other polysaccharides can also be present in the coating composition.
- the coating can include two different natural biodegradable polysaccharides, or more than two different natural biodegradable polysaccharides.
- the natural biodegradable polysaccharide such as amylose or maltodextrin
- another biodegradable polymer i.e., a secondary polymer
- An additional polymer or polymers can be used to alter the properties of the matrix, or serve as bulk polymers to alter the volume of the matrix.
- other biodegradable polysaccharides can be used in combination with the amylose polymer. These include hyaluronic acid, dextran, starch, amylose (for example, non-derivitized), amylopectin, cellulose, xanthan, pullulan, chitosan, pectin, inulin, alginates, and heparin.
- a composition is disposed on a surface that includes at least the natural biodegradable polysaccharide, such as amylose or maltodextrin having a coupling group and a bioactive agent.
- the composition includes the natural biodegradable polysaccharide, a bioactive agent, and an initiator.
- a coating is formed by disposing the natural biodegradable polysaccharide and disposing the biodegradable microparticles on a surface.
- a composition containing both the natural biodegradable polysaccharide and the biodegradable microparticles having the bioactive agent are disposed on a surface.
- a sealant composition that includes at least the natural biodegradable polysaccharide having a coupling group is disposed on a porous surface.
- the concentration of the natural biodegradable polysaccharide in the composition can be chosen to provide a coating or an article having a desired density of crosslinked natural biodegradable polysaccharide.
- the concentration of natural biodegradable polysaccharide in the composition can depend on the type or nature of the bioactive agent that is included in the composition.
- the natural biodegradable polysaccharide having the coupling groups is present in the coating composition at a concentration in the range of 5-100% (w/v), and 5-50%, and in more specific embodiments in the range of 10-20% and in other embodiments in the range of 20-50% (w/v).
- the concentration of the natural biodegradable polysaccharide may be higher to provide a more structurally rigid implant.
- compositions can change or improve the properties of the coating or article that is formed by the natural biodegradable coating having coupling groups in order to change the elasticity, flexibility, wettability, or adherent properties, (or combinations thereof) of the coating formed on the surface.
- plasticizing agents include glycerol, diethylene glycol, sorbitol, sorbitol esters, maltitol, sucrose, fructose, invert sugars, corn syrup, and mixtures thereof.
- the amount and type of plasticizing agents can be readily determined using known standards and techniques.
- compositions of this invention can be used to coat the surface of a variety of implantable devices.
- the coating of natural biodegradable polysaccharide (with or without bioactive agent) can be applied to a medical device using standard techniques to cover the entire surface of the device, or a portion of the device surface.
- the medical articles on which the biodegradable coating can be formed can be fabricated from any suitable biomaterial or combinations of biomaterials.
- Preferred biomaterials include those formed of synthetic polymers, including oligomers, homopolymers, and copolymers resulting from either addition or condensation polymerizations.
- suitable addition polymers include, but are not limited to, acrylics such as those polymerized from methyl acrylate, methyl methacrylate, hydroxyethyl methacrylate, hydroxyethyl acrylate, acrylic acid, methacrylic acid, glyceryl acrylate, glyceryl methacrylate, methacrylamide, and acrylamide; vinyls such as ethylene, propylene, vinyl chloride, vinyl acetate, vinyl pyrrolidone, and vinylidene difluoride.
- acrylics such as those polymerized from methyl acrylate, methyl methacrylate, hydroxyethyl methacrylate, hydroxyethyl acrylate, acrylic acid, methacrylic acid, glyceryl acrylate, glyceryl methacrylate, methacrylamide, and acrylamide
- vinyls such as ethylene, propylene, vinyl chloride, vinyl acetate, vinyl pyrrolidone, and vinylidene difluoride.
- condensation polymers include, but are not limited to, nylons such as polycaprolactam, polylauryl lactam, polyhexamethylene adipamide, and polyhexamethylene dodecanediamide, and also polyurethanes, polycarbonates, polyamides, polysulfones, poly(ethylene terephthalate), polylactic acid, polyglycolic acid, polydimethylsiloxanes, and polyetherketone.
- biomaterials include metals, metal alloys, and ceramics.
- the metals and metal alloys include, but are not limited to, titanium, Nitinol, stainless steel, tantalum, and cobalt chromium.
- a second class of metals includes the noble metals such as gold, silver, copper, and platinum uridium.
- the ceramics include, but are not limited to, silicon nitride, silicon carbide, zirconia, and alumina, as well as glass, silica, and sapphire. Combinations of ceramics and metals are another class of biomaterials.
- Certain natural materials are also suitable biomaterials, including human tissue such as bone, cartilage, skin and teeth; and other organic materials such as wood, cellulose, compressed carbon, and rubber.
- the surface of such biomaterials can be pretreated (for example, with a Parylene coating composition) in order to alter the surface properties of the biomaterial, when desired.
- the biomaterials as described herein can be used to fabricate a variety of implantable devices on which the biodegradable coating can be formed.
- the medical device can be any device that is introduced temporarily or permanently into a mammal for the prophylaxis or treatment of a medical condition. These devices include any that are introduced subcutaneously, percutaneously or surgically to rest within an organ, tissue, or lumen of an organ, such as arteries, veins, ventricles or atria of the heart.
- the device can be a biostable device, a partially degradable device, or a completely degradable device (for example, stents can be fabricated from biodegradable polymeric materials).
- the natural biodegradable polysaccharide coating can be formed on the surface of virtually any implantable device.
- implantable devices include but are not limited to drug-delivering vascular stents; other vascular devices (e.g., grafts, catheters, valves, artificial hearts, heart assist devices); implantable defibrillators; blood oxygenator devices; surgical devices; tissue-related materials; membranes; cell culture devices; chromatographic support materials; biosensors; shunts for hydrocephalus; wound management devices; endoscopic devices; infection control devices; orthopedic devices; dental devices, urological devices; colostomy bag attachment devices; ophthalmic devices; glaucoma drain shunts; synthetic prostheses; intraocular lenses; respiratory, peripheral cardiovascular, spinal, neurological, dental, and ear/nose/throat devices (e.g., ear drainage tubes); renal devices; and dialysis articles (e.g., tubing, membranes, grafts).
- vascular devices e.g., grafts,
- contemplated devices include self-expanding stents (e.g., made from nitinol), balloon-expanded stents (e.g., prepared from stainless steel), degradable coronary stents, non-degradable coronary stents, peripheral coronary stents, urinary catheters (e.g., surface-coated with antimicrobial agents), penile implants, sphincter devices, urethral devices, bladder devices, renal devices, vascular implants and grafts, intravenous catheters (e.g., treated with antithrombotic agents), small diameter grafts, artificial lung catheters, electrophysiology catheters, anastomosis devices, vertebral disks, bone pins, suture anchors, hemostatic barriers, clamps, surgical staples/sutures/screws/plates/clips, atrial septal defect closures, electro-stimulation leads for cardiac rhythm management (e.g., pacer leads), glucose sensors (long-term and
- compositions are particularly useful for forming biodegradable coatings on the surface of devices that will come in contact with aqueous systems.
- the body fluids typically have enzymes that allow for the degradation of the natural biodegradable polysaccharide-based coating.
- the aqueous system (such as bodily fluids) allows for the degradation of the biodegradable coating and release of the bioactive agent from the device.
- the bioactive agent can diffuse out of the matrix. For example, it has been demostrated that a loosely formed matrix may allow some diffusion of bioactive agents, particularly smaller bioactive agents. More desirably, well-formed matrices having signification polysaccharide association via coupling groups are able to retain bioactive agents. Release of bioactive agents from these matrices is mediated by enzymatic degradation.
- the coatings can also be formed on a biological article.
- a “biological article” refers to any sort of non-synthetic biologically-based article such as a cell or a portion of a cell, a group of cells, tissue, or an organ or a portion of a organ.
- the present reagents can be used in methods for encapsulating cellular material.
- a sealant coating is provided on a porous surface of a medical article.
- the medical article can be any article that is introduced into a mammal for the prophylaxis or treatment of a medical condition, wherein the medical article include a sealant coating (at least initially) and has a sealant function.
- the medical article having the sealant coating can provide one or more functions, including providing a barrier to the movement of body fluids, such as blood.
- the sealant coatings can be formed on the surface of articles that have a porous structure wherein it is desired to seal the porous structure, providing a barrier to the movement of body fluids. In many cases it is desirable to form these artificial barriers to ensure that the implanted article functions as it is intended to in the body. Gradually, however, it is desired to allow the body to maintain the function of the sealant coating by replacing the sealant barrier materials with natural materials from the body.
- the sealant composition can be prepared and/or applied in such a manner as to fill the pores on the surface of the article with the sealant material. This can be achieved by, for example, controlling factors such as the viscosity of the coating composition and the coupling of the natural biodegradable polysaccharides during formation of the coating.
- An article having a “porous surface” refers to any article having a surface with pores on which a natural biodegradable polysaccharide-based sealant coating can be formed.
- the pores are preferably of a physical dimension that permits in-growth of tissue into the pores as the sealant coating degrades.
- the porous surface can be associated with a non-porous surface, such as a scaffold that can provide support to the porous surface.
- the medical article can include porous surfaces that can be provided with a sealant coating and non-porous surfaces that are not coated with the sealant coating, optionally coated with the sealant coating, or coated with a material that is different than the sealant coating. All or a portion of the porous surfaces can be coated with the sealant coating.
- a sealant material that is different than the natural biodegradable polysaccharide-based sealant material can be used in conjunction with the natural biodegradable polysaccharide-based sealant material.
- either the interior or the exterior portions can be coated, or portions of the interior and/or exterior can be coated.
- the portion or portions of the article that are coated can depend on a particular desired application or function of the coated article. For example, in some cases it may be desirable to have a difference in the flow of fluids, such as blood, through porous portions of the medical article. Also, tissue in-growth on selected portions of the article can also be promoted by depositing the sealant coating at desired locations.
- the porous surface of the article can also include a material that is thrombogenic and/or presents surface stasis areas (regions of minimized or no blood flow). Depending on the application, a surface having a desired degree of porosity is obtained. The surface will have a degree of porosity sufficient for proper in-growth of cells and tissue forming factors. Upon tissue in-growth, the surface can provide a barrier that is fluid impermeable.
- the porous surface of the article is a fabric or has fabric-like qualities.
- the porous surface can be formed from textiles, which include woven materials, knitted materials, and braided materials. Particularly useful textile materials are woven materials which can be formed using any suitable weave pattern known in the art.
- the porous surface can be that of a graft, sheath, cover, patch, sleeve, wrap, casing, and the like. These types of articles can function as the medical article itself or be used in conjunction with another part of a medical article (examples of which are described herein).
- the porous surface can include any suitable type of biomaterial.
- Useful biomaterials can be woven into fibers for the preparation of fabrics as described herein.
- Useful materials include synthetic addition or condensation polymers such as polyesters, polypropylenes, polyethylenes, polyurethanes, and polytetrafluoroethylenes.
- Polyethylene terephthalate (PET) is a commonly used polymer in fabrics. Blends of these polymers can also be utilized in the preparation of fibers, such as monofilament or multi-filament fibers, for the construction of fabrics. Commonly used fabrics include those such as nylon, velour, and DACRONTM.
- the fabrics can optionally include stiffening materials to improve the physical properties of the article, for example, to improve the strength of a graft.
- stiffening materials can improve the function of an implanted article.
- strengthening materials can improve the patency of the graft.
- Porous surfaces can also be formed by dipping mandrels in these types of polymers.
- porous surfaces include those of cardiac patches. These can be used to decrease suture line bleeding associated with cardiovascular reconstructions. The patches can be used to seal around the penetrating suture. Common materials used in cardiac patches include PTFE and DACRONTM.
- the thickness of the material used as the porous surface can be chosen depending on the application. However, it is common that these thicknesses are about 1.0 mm or less on average, and typically in the range of about 0.10 mm to about 1.0 mm.
- porous surfaces include grafts, particularly grafts having textured exterior portions.
- textured grafts include those that have velour-textured exteriors, with textured or smooth interiors.
- Grafts constructed from woven textile products are well known in the art and have been described in numerous documents, for example, U.S. Pat. No. 4,047,252; U.S. Pat. No. 5,178,630; U.S. Pat. No. 5,282,848; and U.S. Pat. No. 5,800,514.
- the natural biodegradable polysaccharide can be used to provide a sealant coating to a wide variety of articles.
- article is used in its broadest sense and includes objects such as devices.
- Such articles include, but are not limited to vascular implants and grafts, grafts, surgical devices; synthetic prostheses; vascular prosthesis including endoprosthesis, stent-graft, and endovascular-stent combinations; small diameter grafts, abdominal aortic aneurysm grafts; wound dressings and wound management device; hemostatic barriers; mesh and hernia plugs; patches, including uterine bleeding patches, atrial septic defect (ASD) patches, patent foramen ovale (PFO) patches, ventricular septal defect (VSD) patches, and other generic cardiac patches; ASD, PFO, and VSD closures; percutaneous closure devices, mitral valve repair devices; left atrial appendage filters; valve annuloplasty devices, catheters; central venous access catheters, vascular access catheters,
- the polymeric compositions can be utilized in connection with an ophthalmic article.
- the ophthalmic article can be configured for placement at an external or internal site of the eye.
- Suitable ophthalmic articles in accordance with these aspects can provide bioactive agent to any desired area of the eye.
- the articles can be utilized to deliver bioactive agent to an anterior segment of the eye (in front of the lens), and/or a posterior segment of the eye (behind the lens).
- Suitable ophthalmic devices can also be utilized to provide bioactive agent to tissues in proximity to the eye, when desired.
- the biodegradable polysaccharide compositions can be used either for the formation of a coating on the surface of an ophthalmic article, or in the construction of an ophthalmic article.
- Suitable external articles can be configured for topical administration of bioactive agent.
- Such external devices can reside on an external surface of the eye, such as the cornea (for example, contact lenses) or bulbar conjunctiva.
- suitable external devices can reside in proximity to an external surface of the eye.
- Articles configured for placement at an internal site of the eye can reside within any desired area of the eye.
- the ophthalmic article can be configured for placement at an intraocular site, such as the vitreous.
- Illustrative intraocular devices include, but are not limited to, those described in U.S. Pat. No. 6,719,750 B2 (“Devices for Intraocular Drug Delivery,” Varner et al.) and U.S. Pat. No. 5,466,233 (“Tack for Intraocular Drug Delivery and Method for Inserting and Removing Same,” Weiner et al.); U.S. Publication Nos.
- the biodegradable polysaccharide coating is included on a non-linear intraocular device.
- the biodegradable polysaccharide coating includes a bioactive agent, such as a high molecular weight bioactive agent useful for treating an ocular condition.
- the ophthalmic article can be configured for placement, or can be formed, at a subretinal area within the eye.
- Illustrative ophthalmic devices for subretinal application include, but are not limited to, those described in U.S. Patent Publication No. 2005/0143363 (“Method for Subretinal Administration of Therapeutics Including Steroids; Method for Localizing Pharmacodynamic Action at the Choroid and the Retina; and Related Methods for Treatment and/or Prevention of Retinal Diseases,” de Juan et al.); U.S. application Ser. No. 11/175,850 (“Methods and Devices for the Treatment of Ocular Conditions,” de Juan et al.); and related applications.
- the invention provides a biodegradable implant that is formed from the biodegradable polysaccharide and that includes a bioactive agent, such as a high molecular weight bioactive agent useful for treating an ocular condition.
- a bioactive agent such as a high molecular weight bioactive agent useful for treating an ocular condition.
- the invention provides a method for forming an article from the biodegradable polysaccharide, wherein the method includes polymerizing a composition that includes the biodegradable polysaccharide within the eye, such as in a subretinal area or within the vitreous.
- a low viscosity composition including a natural biodegradable polysaccharide and a redox pair to promote polymerization for in situ matrix formation.
- Ophthalmic articles can also be configured for placement within any desired tissues of the eye.
- ophthalmic devices can be configured for placement at a subconjunctival area of the eye, such as devices positioned extrasclerally but under the conjunctiva, such as glaucoma drainage devices and the like.
- a medical article having a biodegradable coating can also be prepared by assembling an article having two or more “parts” (for example, pieces of a medical article that can be put together to form the article) wherein at least one of the parts has a biodegradable coating. All or a portion of the part of the medical article can have a biodegradable coating.
- the invention also contemplates parts of medical article (for example, not the fully assembled article) that have a natural biodegradable polysaccharide-based coating.
- the natural biodegradable polymer is used to form the body member of a medical implant, wherein the body member has a wet weight of about 10 g or less, or a dry weight of about 2.5 g or less.
- the device can also have a base coating of material.
- the base coating can serve one or more functions, for example, it can provide an improved surface for the natural biodegradable polysaccharide or composition that includes the natural biodegradable polysaccharide.
- the base coating can include a polymeric material, such as a natural or synthetic polymer. Examples of suitable compounds that can be used to pretreat a surface to provide a base coat include Parylene and organosilane compounds.
- Suitable base coatings can include, for example, methacrylate, acrylate, alkylacrylate, acrylamide, vinylpyrrolidinone, vinylacetamide, and vinyl formamide based polymers and copolymers. These polymers can also include latent reactive groups such as photoreactive groups.
- Base coatings can be useful in various coating processes.
- biodegradable microparticles can be first disposed on a base coat and then the natural biodegradable polysaccharide having coupling groups can be disposed on the microparticles.
- the surface can then be treated to form a coating wherein the microparticles are predominantly located between the base layer and the layer formed from the natural biodegradable polysaccharide having coupling groups.
- an initiator can be included in a base coating and the natural biodegradable polysaccharide polymer or composition that includes the natural biodegradable polysaccharide polymer can be disposed on the base coating.
- the base coating can serve one or more functions, for example, it can provide an improved surface for the natural biodegradable polysaccharide or composition that includes the natural biodegradable polysaccharide.
- the natural biodegradable polysaccharide coating or the biodegradable article includes one or more bioactive agents.
- the bioactive agent can be dispersed within the natural biodegradable polysaccharide coating or biodegradable article itself.
- the bioactive agent can be present in microparticles that are associated with the natural biodegradable polysaccharide coating.
- the bioactive agent can be delivered from the coated surface upon degradation of the natural biodegradable polysaccharide and/or biodegradable microparticles.
- bioactive agent refers to a peptide, protein, carbohydrate, nucleic acid, lipid, polysaccharide, synthetic inorganic or organic molecule, viral particle, cell, or combinations thereof, that causes a biological effect when administered in vivo to an animal, including but not limited to birds and mammals, including humans.
- suitable gene therapy agents include (a) therapeutic nucleic acids, including antisense DNA, antisense RNA, and interference RNA, and (b) nucleic acids encoding therapeutic gene products, including plasmid DNA and viral fragments, along with associated promoters and excipients.
- Other molecules that can be incorporated include nucleosides, nucleotides, vitamins, minerals, and steroids.
- the coatings of the invention are particularly useful for delivering bioactive agents that are large hydrophilic molecules, such as polypeptides (including proteins and peptides), nucleic acids (including DNA and RNA), polysaccharides (including heparin), as well as particles, such as viral particles, and cells.
- bioactive agent has a molecular weight of about 10,000 or greater.
- Classes of bioactive agents which can be incorporated into biodegradable coatings (both the natural biodegradable matrix and/or the biodegradable microparticles) of this invention include, but are not limited to: ACE inhibitors, actin inhibitors, analgesics, anesthetics, anti-hypertensives, anti polymerases, antisecretory agents, anti-AIDS substances, antibiotics, anti-cancer substances, anti-cholinergics, anti-coagulants, anti-convulsants, anti-depressants, anti-emetics, antifungals, anti-glaucoma solutes, antihistamines, antihypertensive agents, anti-inflammatory agents (such as NSAIDs), anti metabolites, antimitotics, antioxidizing agents, anti-parasite and/or anti-Parkinson substances, antiproliferatives (including antiangiogenesis agents), anti-protozoal solutes, anti-psychotic substances, anti-pyretics, antiseptics, anti-spasmodics
- Antibiotics are art recognized and are substances which inhibit the growth of or kill microorganisms.
- antibiotics include penicillin, tetracycline, chloramphenicol, minocycline, doxycycline, vancomycin, bacitracin, kanamycin, neomycin, gentamycin, erythromycin, cephalosporins, geldanamycin, and analogs thereof.
- cephalosporins examples include cephalothin, cephapirin, cefazolin, cephalexin, cephradine, cefadroxil, cefamandole, cefoxitin, cefaclor, cefuroxime, cefonicid, ceforanide, cefotaxime, moxalactarn, ceftizoxime, ceftriaxone, and cefoperazone.
- Antiseptics are recognized as substances that prevent or arrest the growth or action of microorganisms, generally in a nonspecific fashion, e.g., by inhibiting their activity or destroying them.
- antiseptics include silver sulfadiazine, chlorhexidine, glutaraldehyde, peracetic acid, sodium hypochlorite, phenols, phenolic compounds, iodophor compounds, quaternary ammonium compounds, and chlorine compounds.
- Anti-viral agents are substances capable of destroying or suppressing the replication of viruses.
- examples of anti-viral agents include ⁇ -methyl-P-adamantane methylamine, hydroxy-ethoxyrnethylguanine, adamantanamine, 5-iodo-2′-deoxyuridine, trifluorothymidine, interferon, and adenine arabinoside.
- Enzyme inhibitors are substances that inhibit an enzymatic reaction.
- enzyme inhibitors include edrophonium chloride, N-methylphysostigmine, neostigmine bromide, physostigmine sulfate, tacrine HCl, tacrine, 1-hydroxymaleate, iodotubercidin, p-bromotetramisole, 10-( ⁇ -diethylaminopropionyl)-phenothiazine hydrochloride, calmidazolium chloride, hemicholinium-3,3,5-dinitrocatechol, diacylglycerol kinase inhibitor I, diacylglycerol kinase inhibitor II, 3-phenylpropargylamine, N-monomethyl-L-arginine acetate, carbidopa, 3-hydroxybenzylhydrazine HCl, hydralazine HCl, clorgyline HCl, deprenyl HCl
- Anti-pyretics are substances capable of relieving or reducing fever.
- Anti-inflammatory agents are substances capable of counteracting or suppressing inflammation. Examples of such agents include aspirin (salicylic acid), indomethacin, sodium indomethacin trihydrate, salicylamide, naproxen, colchicine, fenoprofen, sulindac, diflunisal, diclofenac, indoprofen and sodium salicylamide.
- Local anesthetics are substances that have an anesthetic effect in a localized region. Examples of such anesthetics include procaine, lidocaine, tetracaine and dibucaine.
- Cell response modifiers are chemotactic factors such as platelet-derived growth factor (pDGF).
- Other chemotactic factors include neutrophil-activating protein, monocyte chemoattractant protein, macrophage-inflammatory protein, SIS (small inducible secreted) proteins, platelet factor, platelet basic protein, melanoma growth stimulating activity, epidermal growth factor, transforming growth factor (alpha), fibroblast growth factor, platelet-derived endothelial cell growth factor, insulin-like growth factor, nerve growth factor, and bone growth/cartilage-inducing factor (alpha and beta).
- pDGF platelet-derived growth factor
- Other chemotactic factors include neutrophil-activating protein, monocyte chemoattractant protein, macrophage-inflammatory protein, SIS (small inducible secreted) proteins, platelet factor, platelet basic protein, melanoma growth stimulating activity, epidermal growth factor, transforming growth factor (alpha), fibroblast growth factor, platelet-derived endo
- cell response modifiers are the interleukins, interleukin inhibitors or interleukin receptors, including interleukin 1 through interleukin 10; interferons, including alpha, beta and gamma; hematopoietic factors, including erythropoietin, granulocyte colony stimulating factor, macrophage colony stimulating factor and granulocyte-macrophage colony stimulating factor; tumor necrosis factors, including alpha and beta; transforming growth factors (beta), including beta-1, beta-2, beta-3, inhibin, activin, and DNA that encodes for the production of any of these proteins.
- interleukins interleukin inhibitors or interleukin receptors, including interleukin 1 through interleukin 10
- interferons including alpha, beta and gamma
- hematopoietic factors including erythropoietin, granulocyte colony stimulating factor, macrophage colony stimulating factor and granulocyte-macrophage
- statins examples include lovastatin, pravastatin, simvastatin, fluvastatin, atorvastatin, cerivastatin, rousvastatin, and superstatin.
- Imaging agents are agents capable of imaging a desired site, e.g., tumor, in vivo, can also be included in the coating composition.
- imaging agents include substances having a label which is detectable in vivo, e.g., antibodies attached to fluorescent labels.
- the term antibody includes whole antibodies or fragments thereof.
- Exemplary ligands or receptors include antibodies, antigens, avidin, streptavidin, biotin, heparin, type IV collagen, protein A, and protein G.
- antibiotics include antibiotic peptides.
- the bioactive agent can be selected to improve the compatibility (for example, with blood and/or surrounding tissues) of medical device surfaces.
- biocompatible agents when associated with the medical device surface, can serve to shield the blood from the underlying medical device material.
- Suitable biocompatible agents preferably reduce the likelihood for blood components to adhere to the medical device, thus reducing the formation of thrombus or emboli (blood clots that release and travel downstream).
- the bioactive agent can improve the biocompatibility of the medical article having a coating that includes the natural biodegradable polymer and the biodegradable microparticle.
- the bioactive agent can provide antirestenotic effects, such as antiproliferative, anti-platelet, and/or antithrombotic effects.
- the bioactive agent can include anti-inflammatory agents, immunosuppressive agents, cell attachment factors, receptors, ligands, growth factors, antibiotics, enzymes, nucleic acids, and the like.
- Compounds having antiproliferative effects include, for example, actinomycin D, angiopeptin, c-myc antisense, paclitaxel, taxane, and the like.
- bioactive agents having antithrombotic effects include heparin, heparin derivatives, sodium heparin, low molecular weight heparin, hirudin, lysine, prostaglandins, argatroban, forskolin, vapiprost, prostacyclin and prostacyclin analogs, D-ph-pr-arg-chloromethylketone (synthetic antithrombin), dipyridamole, glycoprotein IIb/IIIa platelet membrane receptor antibody, coprotein IIb/IIIa platelet membrane receptor antibody, recombinant hirudin, thrombin inhibitor (such as commercially available from Biogen), chondroitin sulfate, modified dextran, albumin, streptokinase, tissue plasminogen activator (TPA), urokinase, nitric oxide inhibitors, and the like.
- heparin heparin derivatives, sodium heparin, low molecular weight heparin,
- the bioactive agent can also be an inhibitor of the GPIIb-IIIa platelet receptor complex, which mediates platelet aggregation.
- GPIIb/IIIa inhibitors can include monoclonal antibody Fab fragment c7E3, also know as abciximab (ReoPrOTM), and synthetic peptides or peptidomimetics such as eptifibatide (IntegrilinTM) or tirofiban (AgrastatTM).
- the bioactive agent can be an immunosuppressive agent, for example, cyclosporine, CD-34 antibody, everolimus, mycophenolic acid, sirolimus, tacrolimus, and the like.
- immunosuppressive agent for example, cyclosporine, CD-34 antibody, everolimus, mycophenolic acid, sirolimus, tacrolimus, and the like.
- exemplary therapeutic antibodies include trastuzumab (HerceptinTM), a humanized anti-HER2 monoclonal antibody (moAb); alemtuzumab (CampathTM), a humanized anti-CD52 moAb; gemtuzumab (MylotargTM), a humanized anti-CD33 moAb; rituximab (RituxanTM), a chimeric anti-CD20 moAb; ibritumomab (ZevalinTM), a murine moAb conjugated to a beta-emitting radioisotope; tositumomab (BexxarTM), a murine anti-CD20 moAb; edrecolomab (PanorexTM), a murine anti-epithelial cell adhesion molecule moAb; cetuximab (ErbituxTM), a chimeric anti-EGFR moAb; and bevacizumab (AvastinTM), a humanized anti-VEGF moAb.
- the bioactive agent can be a surface adhesion molecule or cell-cell adhesion molecule.
- Exemplary cell adhesion molecules or attachment proteins such as extracellular matrix proteins including fibronectin, laminin, collagen, elastin, vitronectin, tenascin, fibrinogen, thrombospondin, osteopontin, von Willibrand Factor, bone sialoprotein (and active domains thereof), or a hydrophilic polymer such as hyaluronic acid, chitosan or methyl cellulose, and other proteins, carbohydrates, and fatty acids.
- Exemplary cell-cell adhesion molecules include N-cadherin and P-cadherin and active domains thereof.
- Exemplary growth factors include fibroblastic growth factors, epidermal growth factor, platelet-derived growth factors, transforming growth factors, vascular endothelial growth factor, bone morphogenic proteins and other bone growth factors, and neural growth factors.
- the bioactive agent can be also be selected from mono-2-(carboxymethyl)hexadecanamidopoly(ethylene glycol) 200 mono-4-benzoylbenzyl ether, mono-3-carboxyheptadecanamidopoly(ethylene glycol) 200 mono-4-benzoylbenzyl ether, mono-2-(carboxymethyl)hexadecanamidotetra(ethylene glycol)mono-4-benzoylbenzyl ether, mono-3-carboxyheptadecanamidotetra(ethylene glycol)mono-4-benzoylbenzyl ether, N-[2-(4-benzoylbenzyloxy)ethyl]-2-(carboxymethyl)hexadecanamide, N-[2-(4-benzoylbenzyloxy)ethyl]-3-carboxyheptadecanamide, N-[12-(benzoylbenzyloxy)dodecyl]-2-(carbox
- bioactive agents and/or bioactive agent include analogues of rapamycin (“rapalogs”), ABT-578 from Abbott, dexamethasone, betamethasone, vinblastine, vincristine, vinorelbine, poside, teniposide, daunorubicin, doxorubicin, idarubicin, anthracyclines, mitoxantrone, bleomycins, plicamycin (mithramycin), mitomycin, mechlorethamine, cyclophosphamide and its analogs, melphalan, chlorambucil, ethylenimines and methylmelamines, alkyl sulfonates-busulfan, nitrosoureas, carmustine (BCNU) and analogs, streptozocin, trazenes-dacarbazinine, methotrexate, fluorouracil, floxuridine, cytarabine, mercap
- Viral particles and viruses include those that may be therapeutically useful, such as those used for gene therapy, and also attenuated viral particles and viruses which can promote an immune response and generation of immunity.
- Useful viral particles include both natural and synthetic types.
- Viral particles include, but are not limited to, adenoviruses, baculoviruses, parvoviruses, herpesviruses, poxviruses, adeno-associated viruses, vaccinia viruses, and retroviruses.
- bioactive agents that can be used for altering gene function include plasmids, phages, cosmids, episomes, and integratable DNA fragments, antisense oligonucleotides, antisense DNA and RNA, modified DNA and RNA, iRNA, ribozymes, siRNA, and shRNA.
- bioactive agents include cells such as platelets, stem cells, T lymphocytes, B lymphocytes, acidophils, adipocytes, astrocytes, basophils, hepatocytes, neurons, cardiac muscle cells, chondrocytes, epithelial cells, dendrites, endrocrine cells, endothelial cells, eosinophils, erythrocytes, fibroblasts, follicular cells, ganglion cells, hepatocytes, endothelial cells, Leydig cells, parenchymal cells, lymphocytes, lysozyme-secreting cells, macrophages, mast cells, megakaryocytes, melanocytes, monocytes, myoid cells, neck nerve cells, neutrophils, oligodendrocytes, oocytes, osteoblasts, osteochondroclasts, osteoclasts, osteocytes, plasma cells, spermatocytes, reticulocytes, Schwann cells, Sertoli cells, skeletal
- Additives such as inorganic salts, BSA (bovine serum albumin), and inert organic compounds can be used to alter the profile of bioactive agent release, as known to those skilled in the art.
- BSA bovine serum albumin
- inert organic compounds can be used to alter the profile of bioactive agent release, as known to those skilled in the art.
- the concentration of the bioactive agent or agents dissolved or suspended in the coating mixture can range from about 0.01 to about 90 percent, by weight, based on the weight of the final coated composition.
- bioactive agent or combination of bioactive agents, can be selected depending upon one or more of the following factors: the application of the controlled delivery device, the medical condition to be treated, the anticipated duration of treatment, characteristics of the implantation site, the number and type of bioactive agents to be utilized, and the like.
- any of the polymer compositions described herein can be provided to the surface of the medical article and can include any number of desired bioactive agents, depending upon the final application of the medical device.
- Bioactive agents are commercially available from Sigma Aldrich Fine Chemicals, Milwaukee, Wis.
- the bioactive agent can be used to promote thrombosis in association with the natural biodegradable polysaccharide-based coating, which can be of particular use when a coating having a sealant function is desired.
- a sealant coating including a thrombogenic agent can promote the in-growth of tissue upon degradation of the sealant coating material.
- the degree of thrombosis can be controlled by various factors, including, for example, the presence of one or more thrombosis-promoting bioactive agents on or within the coating. Suitable thrombotic agents are described herein.
- the thrombotic agent can be selected to have an affect on the blood and/or surrounding tissues that are in contact with the article surface.
- the thrombotic agent is chosen for the ability to affect the ability of blood components to adhere to the medical article.
- the thrombotic agent can, in some cases, be chosen to promote thrombus formation at the surface of the coated article. Therefore, in some embodiments, the sealant coating can include a thrombotic agent, such as thrombin, collagen (for example, (synthetic) recombinant human collagen (FibroGen, South San Francisco, Calif.)), ADP, or convulxin to promote thrombosis at the coated surface of the article.
- prothrombotic or procoagulant factors include platelet factors 1-4, platelet activating factor (acetyl glyceryl ether phosphoryl choline); P-selectin and von Willebrand Factor (vWF); tissue factor; plasminogen activator initiator-1; thromboxane; procoagulant thrombin-like enzymes including cerastotin and afaacytin; phospholipase A 2 ; Ca 2+ -dependent lectins (C-type lectin); factors that bind glycoprotein receptors and induce aggregation including aggretin, rhodocytin, aggregoserpentin, triwaglerin, and equinatoxin; glycoprotein Ib agonists including mamushigin and alboaggregin; vWF interacting factors including botrocetin, bitiscetin, cerastotin, and ecarin.
- platelet activating factor acetyl glyceryl ether
- coagulation factors I-XIII for example, fibrinogen, prothrombin, tissue thromboplastin, calcium, proaccelerin (accelerator globulin), proconvertin (serum prothrombin conversion accelerator), antihemophilic factor, plasma thromboplastin component, Stuart factor (autoprothrombin C), plasma thromboplastin antecedent (PTA), Hageman factor, and fibrin-stabilizing factor (FSF, fibrinase, protransglutaminase)).
- coagulation factors I-XIII for example, fibrinogen, prothrombin, tissue thromboplastin, calcium, proaccelerin (accelerator globulin), proconvertin (serum prothrombin conversion accelerator), antihemophilic factor, plasma thromboplastin component, Stuart factor (autoprothrombin C), plasma thromboplastin antecedent (PTA), Hageman factor, and fibrin-stabilizing factor (FSF, fibrinase, protransglutamina
- Some surface adhesion molecule or cell-cell adhesion molecules may also function to promote coagulation or thrombosis.
- Exemplary cell adhesion molecules or attachment proteins include fibronectin, laminin, collagen, elastin, vitronectin, tenascin, fibrinogen, thrombospondin, osteopontin, von Willebrand Factor, bone sialoprotein (and active domains thereof), or a hydrophilic polymer such as hyaluronic acid, chitosan or methyl cellulose, and other proteins, carbohydrates, and fatty acids.
- Exemplary cell-cell adhesion molecules include N-cadherin and P-cadherin and active domains thereof.
- the particular thrombotic agent, or a combination of thrombotic agents with other bioactive agents can be selected depending upon one or more of the following factors: the application of the medical article, the medical condition to be treated, the anticipated duration of treatment, characteristics of the implantation site, the number and type of thrombogenic/bioactive agents to be utilized, the chemical composition of the sealant coating (such as amylose, selected additives, and the like), the extent of coupling in the formed sealant coating, and the like.
- sealant compositions described herein can be provided to the surface of the medical article.
- the sealant coating can include any number of desired thrombogenic/bioactive agents, depending upon the final application of the medical article.
- the coating of sealant material (with or without thrombogenic/bioactive agents) can be applied to the medical article using standard techniques to cover the entire surface of the article, or a portion of the article surface.
- the sealant composition material can be provided as a single coated layer (with or without thrombogenic/bioactive agents), or as multiple coated layers (with or without thrombogenic/bioactive agents). When multiple coated layers are provided on the surface, the materials of each coated layer can be chosen to provide a desired effect.
- a microparticle is used to deliver the bioactive agent from the natural biodegradable polysaccharide-based coating.
- the microparticles of the invention can comprise any three-dimensional structure that can be immobilized on a substrate in association with the matrix formed by the amylose polymer.
- the term “microparticle” is intended to reflect that the three-dimensional structure is very small but not limited to a particular size range, or not limited to a structure that has a particular shape.
- microparticles typically have a size in the range of 5 nm to 100 ⁇ m in diameter.
- microparticles are spherical or somewhat spherical in shape, but can have other shapes as well.
- the biodegradable microparticles have a size in the range of 100 nm to 20 ⁇ m in diameter, and even more preferable in the range of 400 nm to 20 ⁇ m in diameter.
- the microparticle being “biodegradable” refers to the presence of one or more biodegradable materials in the microparticle.
- the biodegradable microparticles include at least a biodegradable material (such as a biodegradable polymer) and a bioactive agent.
- the biodegradable microparticles can gradually decompose and release bioactive agent upon exposure to an aqueous environment, such as body fluids.
- the biodegradable microparticle can also include one or more biodegradable polymers.
- biodegradable polymers that can be included in the biodegradable microparticle include, for example, polylactic acid, poly(lactide-co-glycolide), polycaprolactone, polyphosphazine, polymethylidenemalonate, polyorthoesters, polyhydroxybutyrate, polyalkeneanhydrides, polypeptides, polyanhydrides, and polyesters, and the like.
- Biodegradable polyetherester copolymers can be used.
- the polyetherester copolymers are amphiphilic block copolymers that include hydrophilic (for example, a polyalkylene glycol, such as polyethylene glycol) and hydrophobic blocks (for example, polyethylene terephthalate).
- block copolymers include poly(ethylene glycol)-based and poly(butylene terephthalate)-based blocks (PEG/PBT polymer). Examples of these types of multiblock copolymers are described in, for example, U.S. Pat. No. 5,980,948.
- PEG/PBT polymers are commercially available from Octoplus BV, under the trade designation PolyActiveTM.
- Biodegradable copolymers having a biodegradable, segmented molecular architecture that includes at least two different ester linkages can also be used.
- the biodegradable polymers can be block copolymers (of the AB or ABA type) or segmented (also known as multiblock or random-block) copolymers of the (AB) n type. These copolymers are formed in a two (or more) stage ring opening copolymerization using two (or more) cyclic ester monomers that form linkages in the copolymer with greatly different susceptibilities to transesterification. Examples of these polymers are described in, for example, in U.S. Pat. No. 5,252,701 (Jarrett et al., “Segmented Absorbable Copolymer”).
- biodegradable polymer materials include biodegradable terephthalate copolymers that include a phosphorus-containing linkage.
- Polymers having phosphoester linkages called poly(phosphates), poly(phosphonates) and poly(phosphites), are known. See, for example, Penczek et al., Handbook of Polymer Synthesis, Chapter 17: “Phosphorus-Containing Polymers,” 1077-1132 (Hans R. Kricheldorf ed., 1992), as well as U.S. Pat. Nos. 6,153,212, 6,485,737, 6,322,797, 6,600,010, 6,419,709.
- Biodegradable terephthalate polyesters can also be used that include a phosphoester linkage that is a phosphite. Suitable terephthalate polyester-polyphosphite copolymers are described, for example, in U.S. Pat. No. 6,419,709 (Mao et al., “Biodegradable Terephthalate Polyester-Poly(Phosphite) Compositions, Articles, and Methods of Using the Same). Biodegradable terephthalate polyester can also be used that include a phosphoester linkage that is a phosphonate. Suitable terephthalate polyester-poly(phosphonate)copolymers are described, for example, in U.S. Pat. Nos.
- Biodegradable Terephthalate Polyester-Poly(Phosphonate) Compositions, Articles and Methods of Using the Same Biodegradable terephthalate polyesters can be used that include a phosphoester linkage that is a phosphate. Suitable terephthalate polyester-poly(phosphate)copolymers are described, for example, in U.S. Pat. Nos. 6,322,797 and 6,600,010 (Mao et al., “Biodegradable Terephthalate Polyester-Poly(Phosphate)Polymers, Compositions, Articles, and Methods for Making and Using the Same).
- Biodegradable polyhydric alcohol esters can also be used (See U.S. Pat. No. 6,592,895). This patent describes biodegradable star-shaped polymers that are made by esterifying polyhydric alcohols to provide acyl moieties originating from aliphatic homopolymer or copolymer polyesters.
- the biodegradable polymer can be a three-dimensional crosslinked polymer network containing hydrophobic and hydrophilic components which forms a hydrogel with a crosslinked polymer structure, such as that described in U.S. Pat. No. 6,583,219.
- the hydrophobic component is a hydrophobic macromer with unsaturated group terminated ends
- the hydrophilic polymer is a polysaccharide containing hydroxy groups that are reacted with unsaturated group introducing compounds.
- the components are convertible into a one-phase crosslinked polymer network structure by free radical polymerization.
- the biodegradable polymer can comprise a polymer based upon ⁇ -amino acids (such as elastomeric copolyester amides or copolyester urethanes, as described in U.S. Pat. No. 6,503,538).
- the biodegradable microparticle can include one or more biodegradable polymers obtained from natural sources.
- the biodegradable polymer is selected from hyaluronic acid, dextran, starch, amylose, amylopectin, cellulose, xanthan, pullulan, chitosan, pectin, inulin, alginates, and heparin.
- One, or combinations of more than one of these biodegradable polymers can be used.
- a particular biodegradable polymer can also be selected based on the type of bioactive agent that is present in the microparticle. Therefore, in some aspects of the invention, the biodegradable coating can include a natural biodegradable polysaccharide matrix and a natural biodegradable polysaccharide-containing microparticle.
- the microparticles include a natural biodegradable polysaccharide such as amylose or maltodextrin.
- the natural biodegradable polysaccharide can be the primary biodegradable component in the microparticle.
- both the coating matrix and the microparticle include amylose and/or maltodextrin as components.
- Dextran-based microparticles can be particularly useful for the incorporation of bioactive agents such as proteins, peptides, and nucleic acids. Examples of the preparation of dextran-based microparticles are described in U.S. Pat. No. 6,303,148.
- amylose and other starch-based microparticles have been described in various references, including, for example, U.S. Pat. No. 4,713,249; U.S. Pat. No. 6,692,770; and U.S. Pat. No. 6,703,048.
- Biodegradable polymers and their synthesis have been also been described in various references including Mayer, J. M., and Kaplan, D. L. (1994) Trends in Polymer Science 2: pages 227-235; and Jagur-Grodzinski, J., (1999) Reactive and Functional Polymers: Biomedical Application of Functional Polymers, Vol. 39, pages 99-138.
- the biodegradable microparticle contains a biologically active agent (a “bioactive agent”), such as a pharmaceutical or a prodrug.
- a biologically active agent such as a pharmaceutical or a prodrug.
- Microparticles can be prepared incorporating various bioactive agents by established techniques, for example, by solvent evaporation (see, for example, Wichert, B. and Rohdewald, P. J Microencapsul. (1993) 10:195).
- the bioactive agent can be released from the biodegradable microparticle (the microparticle being present in the natural biodegradable polysaccharide coating) upon degradation of the biodegradable microparticle in vivo.
- Microparticles having bioactive agent can be formulated to release a desired amount of the agent over a predetermined period of time.
- microparticles can also be treated with a porogen, such as salt, sucrose, PEG, or an alcohol, to create pores of a desired size for incorporation of the bioactive agent.
- a porogen such as salt, sucrose, PEG, or an alcohol
- the quantity of bioactive agents provided in the biodegradable microparticle can be adjusted by the user to achieve the desired effect. For example, a particular amount of anti-coagulant drug can be incorporated into the microparticle to provide a certain level of anti-coagulant activity from the biodegradable coating.
- Biologically active compounds can be provided by the microparticles in a range suitable for the application.
- protein molecules can be provided by biodegradable microparticles. For example, the amount of protein molecules present can be in the range of 1-250,000 molecules per 1 ⁇ m diameter microparticle.
- the concentration of the bioactive agent present in the biodegradable microparticles can be chosen based on any one or a combination of a number of factors, including, but not limited to, the release rate from the coating, the type of bioactive agent(s) in the coating, the desired local or systemic concentration of the bioactive agent following release, and the half life of the bioactive agent.
- the concentration of bioactive agent in the microparticle can be about 0.001% or greater, or in the range of about 0.001% to about 50 percent, or greater, by weight, based on the weight of the microparticle.
- bioactive agent to be included in the biodegradable microparticle, or combination of bioactive agents in microparticles can be selected depending upon factors such as the application of the coated device, the medical condition to be treated, the anticipated duration of treatment, characteristics of the implantation site, the number and type of bioactive agents to be utilized, the chemical composition of the microparticle, size of the microparticle, crosslinking, and the like.
- the invention advantageously allows for preparation of surfaces having two, or more than two, different bioactive agents, wherein the bioactive agents are mutually incompatible in a particular environment, for example, as hydrophobic and hydrophilic drugs are incompatible in either a polar or non-polar solvent.
- bioactive agents may also demonstrate incompatibility based on protic/aprotic solvents or ionic/non-ionic solvents.
- the invention allows for the preparation of one set of biodegradable microparticles containing a hydrophobic drug and the preparation of another set of biodegradable microparticles containing a hydrophilic drug; the mixing of the two different sets of microparticles into a polymeric material used to form the matrix; and the disposing of the mixture on the surface of a substrate.
- Both hydrophobic and hydrophilic drugs can be released from the surface of the coated substrate at the same time as the biodegradable microparticles degrade, or the composition of the biodegradable microparticles or the natural biodegradable polysaccharide matrix can be altered so that one bioactive agent is released at a different rate or time than the other one.
- Biodegradable microparticles can be prepared having compositions that are suitable for either hydrophobic or hydrophilic drugs.
- polymers such as polylactide or polycaprolactone can be useful for preparing biodegradable microparticles that include hydrophobic drugs; whereas polymers such as amylose or glycolide can be useful for preparing microparticles that include hydrophilic drugs.
- Traditional coating procedures directed at disposing at least two different types of bioactive agents have often required that the bioactive agents be put down separately.
- Traditional approaches may include the steps of solubilizing a hydrophobic drug in a non-polar solvent, coating the surface of the substrate with the non-polar mixture, drying the non-polar mixture, solubilizing the hydrophilic drug in a polar solvent, coating the layer of the dried non-polar mixture with the polar mixture, and then drying the polar mixture.
- This type of traditional coating process can be inefficient and can also result in undesirable surface properties (e.g., the layering of the drugs will cause one drug to be released before the other one is released).
- the method of preparing surfaces having two, or more than two, different bioactive agents, in particular when the two different bioactive agents are released from the surface of the substrate is a significant improvement over traditional methods of coating substrates and delivering bioactive agents from the surface of the substrates.
- Components of the biodegradable coating can be applied to the medical device using standard techniques to cover the entire surface of the device, or a portion of the device surface. As indicated, the components can be applied to the medical device independently or together, for example, in a composition.
- the coating formed on the device can be a single layer coating, or a multiple layer coating.
- bioactive agents from the coating can influence the delivery of bioactive agents from the coating. These include the concentration of the natural biodegradable polysaccharide and the extent of natural biodegradable polysaccharide coupling in the coating, the amount and location of biodegradable microparticles associated with the coating, the concentration of bioactive agent in the microparticles, and the presence of other coated layers, if included in the overall coating and the like.
- the rate of delivery of the drug can be decreased by increasing the concentration of polymeric material or the relative amount of coupling or crosslinking of the polymeric material in the polymeric matrix or in the microparticle. Based on the description provided herein and the general knowledge in this technical area, one can alter properties of the coating to provide a desired release rate for one or more particular bioactive agents from the coating.
- Portions of the coating can be prepared to degrade at the same or different rates.
- the biodegradable microparticles can be prepared or obtained to have a faster rate of degradation than the natural biodegradable polysaccharide matrix.
- the bioactive agent can be released into the natural biodegradable polysaccharide matrix and/or diffuse out of the natural biodegradable polysaccharide matrix.
- a natural biodegradable polysaccharide-based coating can be prepared by any one of a variety of methods.
- a “coating” as used herein can include one or more “coated layers”, each coated layer including one or more coating materials.
- the coating consists of a single layer of material that includes the natural biodegradable polysaccharide, such as amylose or maltodextrin.
- the coating includes more than one coated layer, at least one of the coated layers including the natural biodegradable polysaccharide. If more than one layer is present in the coating, the layers can be composed of the same or different materials. If multiple polymeric layers are provided on the surface, each individual layer of polymer can be chosen to provide a desired effect. Additionally, multiple layers of various bioactive agents can be deposited onto the medical device surface so that a particular bioactive agent can be presented to or released from the medical device at one time, one or more bioactive agents in each layer, which can be separated by polymeric material.
- a natural biodegradable polysaccharide coated layer can be formed by, for example, dipping, spraying, bushing, or swabbing the coating material on the article to form a layer, and then drying the coated layer. The process can be repeated to provide a coating having multiple coated layers, wherein at least one layer includes the natural biodegradable polysaccharide.
- each coated layer is composed of the same materials.
- one or more of the coated layers is composed of materials that are different from one or more of the other layers.
- multiple layers of various bioactive agents can be deposited onto the medical article surface so that a particular bioactive agent can be presented to or released from the medical article at one time, one or more bioactive agents in each layer, which can be separated by polymeric material.
- an initiator is disposed on a surface of a medical article along with the natural biodegradable polysaccharide having pendent coupling groups.
- a bioactive agent can be disposed if desired.
- the initiator can be disposed in a mixture with the natural biodegradable polysaccharide together, or the initiator can be disposed independently.
- These compounds are generally disposed in a fluid state (for example, suspended or dissolved in an aqueous liquid) and can be disposed on an article surface using any one of number of techniques as described herein.
- the initiator is activated, resulting in the activation of the pendent coupling groups, the coupling of natural biodegradable polysaccharide molecules, and the formation of the coating.
- the steps of disposing and activating can be performed in ways (as described herein and/or known in the art) to precisely control the formation of a coating.
- the thickness and the location of the coating on the article surface can be controlled using techniques described herein and/or known in the art.
- the natural biodegradable polysaccharide is selected from the group of amylose and maltodextrin. In other preferred aspects of the following methods, the natural biodegradable polysaccharide has a molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, or 50,000 Da or less. It is also preferred that the natural biodegradable polysaccharides have an average molecular weight of 500 Da or greater. A particularly preferred size range for the natural biodegradable polysaccharides is in the range of about 1000 Da to about 10,000 Da.
- the method includes the steps of (i) disposing a composition comprising (a) a natural biodegradable polysaccharide having a coupling group, (b) an initiator, and (c) a bioactive agent on a surface; and (ii) activating the initiator to provide a coated composition having the natural biodegradable polysaccharide and the bioactive agent on the surface.
- This method can also be used to form a medical implant wherein the composition is disposed to form an implant of a desired configuration.
- the composition can be disposed in a mold.
- This method can also be used to form an in situ formed matrix wherein the composition is disposed within a portion of a subject.
- the method includes the steps of (i) disposing an initiator on a surface, (ii) disposing a composition comprising (a) a natural biodegradable polysaccharide having a coupling group and (b) a bioactive agent on the surface; and (iii) activating the initiator to provide a coated composition having the natural biodegradable polysaccharide and the bioactive agent.
- a composition including the natural biodegradable polysaccharide and the bioactive agent are contacted with the initiator and the initiator is activated to promote the crosslinking of two or more natural biodegradable polysaccharides via their coupling groups.
- the natural biodegradable polysaccharide includes a polymerizable group, such as an ethylenically unsaturated group, and initiator is capable of initiating free radical polymerization of the polymerizable groups.
- the invention provides a method for coating a surface, including the steps of (i) disposing a composition comprising (a) a natural biodegradable polysaccharide having a ethylenically unsaturated group, (b) a polymerization initiator, and (c) a bioactive agent on a surface; and (ii) activating the polymerization initiator to cause the polymerization of the amylose compound thereby providing a coated composition having the natural biodegradable polysaccharide and the bioactive agent on the surface.
- These methods can also be used to form medical implants and in situ-formed matrices, wherein the composition is disposed in a mold or in a subject, respectively, rather than on a surface.
- the invention provides a medical device having a coated composition comprising a plurality of coupled natural biodegradable polysaccharide and a bioactive agent.
- the invention provides methods for preparing biodegradable coatings that include (a) a natural biodegradable polysaccharide having a coupling group and (b) biodegradable microparticles having a bioactive agent.
- the coupling group can be activated by an initiator. Therefore, the method can include the steps of (i) disposing an initiator on a surface, (ii) disposing a composition comprising (a) a natural biodegradable polysaccharide having a coupling group and (b) biodegradable microparticles comprising a bioactive agent; and (iii) activating the initiator to provide a biodegradable bioactive agent-releasing coated composition having the natural biodegradable polysaccharide and the biodegradable microparticles having the bioactive agent.
- the natural biodegradable polysaccharide includes a polymerizable group, such as an ethylenically unsaturated group, and initiator is capable of initiating free radical polymerization of the polymerizable groups. Therefore, in another embodiment, the invention provides a method for coating a surface, including the steps of (i) disposing a composition comprising (a) a natural biodegradable polysaccharide having an ethylenically unsaturated group, (b) a polymerization initiator, and (c) biodegradable microparticles having a bioactive agent on a surface; and (ii) activating the polymerization initiator to cause the polymerization of the natural biodegradable polysaccharide thereby providing a coated composition that includes biodegradable microparticles in a natural biodegradable polysaccharide matrix.
- a composition comprising (a) a natural biodegradable polysaccharide having an ethylenically unsaturated group, (b)
- the invention also provides alternative methods for preparing a coated surface that is biodegradable and having microparticles that can release a bioactive agent.
- the methods include disposing in two or more steps at least the following reagents on a surface: (a) a natural biodegradable polysaccharide comprising a first coupling group (b) a natural biodegradable polysaccharide comprising a second coupling group that is reactive with the first coupling group, and (c) biodegradable microparticles that include a bioactive agent.
- reagents (a) and (b) are reactive with each other and are disposed separately on the surface but can individually include (c).
- reagent (a) is first disposed on the surface and then a mixture comprising reagent (b) and (c) is then disposed on reagent (a).
- Reagent (a) reacts with (b) to link the natural biodegradable polysaccharide together to form a coating that includes (c), the biodegradable microparticles.
- the invention also provides methods for preparing biodegradable sealant coatings that include a natural biodegradable polysaccharide having a coupling group; optionally a bioactive agent can be included in the sealant coating.
- the method includes the steps of (i) disposing a sealant composition comprising (a) a natural biodegradable polysaccharide having a coupling group, and (b) an initiator, and (ii) activating the initiator to form a sealant coating.
- This aspect of the invention includes coating methods where a bulk polymerization approach is performed.
- a composition including a polymerization initiator and natural biodegradable polysaccharides having a polymerizable group is disposed on a surface. The initiator is then activated to promote bulk polymerization and coupling of the natural biodegradable polysaccharides in association with the surface.
- the method includes the steps of (i) disposing an initiator on a surface, (ii) disposing a natural biodegradable polysaccharide having a coupling group; and (iii) activating the initiator to provide a coated composition having the amylose polymer.
- the natural biodegradable polysaccharides can be disposed on the surface along with other reagents if desired.
- This aspect of the invention includes coating methods where a graft polymerization approach is performed. For example, in some embodiments, a polymerization initiator is first disposed on a surface and then a natural biodegradable polysaccharide having a polymerizable group is disposed on the surface having the initiator.
- an aqueous composition that includes the natural biodegradable polysaccharide having the coupling group and a bioactive agent is obtained and used in the method of providing a sealant coating to a surface.
- This composition can be prepared by mixing the natural biodegradable polysaccharide with a bioactive agent, for example, a water-soluble small molecule, a protein, or a nucleic acid.
- the bioactive agent is a procoagulant or prothrombotic factor.
- the bioactive agent can be a protein such as recombinant collagen, or other proteins that associate with receptors on platelets to induce platelet aggregation.
- the coating is placed in contact with an aqueous solution, or the materials of the coating composition.
- the coating or coating materials are designed to be stable in the presence of the aqueous solution provided that an enzyme that causes the degradation of the natural biodegradable polysaccharide (or another degrading agent) is not present in an amount sufficient to cause substantial degradation of the materials.
- the invention provides a shelf stable composition comprising a natural biodegradable polysaccharide comprising coupling groups.
- a shelf stable composition comprising a natural biodegradable polysaccharide comprising coupling groups.
- the invention also provides methods for preparing a biodegradable coating comprising preparing a biodegradable coating composition comprising a natural biodegradable polysaccharide comprising coupling group; storing the coating composition for an amount of time; and then using the coating composition to prepare a biodegradable coating.
- one or more bioactive agents and/or microparticles can be added before or after storage of the coating composition.
- the invention also provides the advantage of being able to perform synthetic and post-synthetic procedures wherein the natural biodegradable polysaccharide is contacted with an aqueous composition, and there is minimal risk of degradation of the polysaccharide.
- the natural biodegradable polysaccharide may be contacted with an aqueous solution for purification without risking significant degradation of the natural biodegradable polysaccharide.
- the invention relates to the stability of the coatings that are formed on an article.
- the invention provides a method comprising obtaining an article having a coating comprising a natural biodegradable polysaccharide, and then contacting the article with an aqueous solution.
- the aqueous solution can be, for example, a storage solution, a solution that is used to hydrate the surface of the coated device, or an aqueous sterilization solution.
- the coating can be contacted with an aqueous sterilization solution.
- Medical articles, or parts of medical articles can be prepared having a coating and these articles can be treated to sterilize one or more parts of the article, or the entire medical article. Sterilization can take place prior to using the medical article and/or, in some cases, during implantation of the medical article.
- the invention provides a method for delivering a bioactive agent from a biodegradable coating or a biodegradable article by exposing the coating or article to an enzyme that causes the degradation of the coating.
- a coated article such as an implantable medical device is provided to a subject.
- the coated article has a biodegradable coating comprising a natural biodegradable polysaccharide having pendent coupling groups, wherein the coating is formed on a surface of the article by reaction of the coupling groups to form a crosslinked matrix of a plurality of natural biodegradable polysaccharides, and wherein the coating includes a bioactive agent.
- the coating or article is then exposed to a carbohydrase that can promote the degradation of the biodegradable coating.
- the carbohydrase that contacts the coating or article can specifically degrade the natural biodegradable polysaccharide causing release of the bioactive agent.
- carbohydrases that can specifically degrade natural biodegradable polysaccharide coatings include ⁇ -amylases, such as salivary and pancreatic ⁇ -amylases; disaccharidases, such as maltase, lactase and sucrase; trisaccharidases; and glucoamylase(amyloglucosidase).
- Serum concentrations for amylase are estimated to be in the range of about 50-100 U per liter, and vitreal concentrations also fall within this range (Varela, R. A., and Bossart, G. D. (2005) J Am Vet Med Assoc 226:88-92).
- the carbohydrase can be administered to a subject to increase the local concentration, for example in the serum or the tissue surrounding the implanted device, so that the carbohydrase may promote the degradation of the coating.
- exemplary routes for introducing a carbohydrase include local injection, intravenous (IV) routes, and the like.
- degradation can be promoted by indirectly increasing the concentration of a carbohydrase in the vicinity of the coated article, for example, by a dietary process, or by ingesting or administering a compound that increases the systemic levels of a carbohydrase.
- the carbohydrase can be provided on a portion of the coated article.
- the carbohydrase may be eluted from a portion of the article that does not have the natural biodegradable polymer coating.
- the carbohydrase as the carbohydrase is released it locally acts upon the coating to cause its degradation and promote the release of the bioactive agent.
- the carbohydrase can be present in a microparticle in one or more portions the coating. As the carbohydrase is released from the microparticle, it causes coating degradation and promote the release of the bioactive agent.
- the invention sets forth methods for providing lubricity to an article surface.
- the matrix of biodegradable polysaccharides as described herein can provide a surprisingly lubricious and durable surface when formed, for example, on the surface of a device.
- lubricity refers to a characterization of the frictional force associated with a coating.
- a lubricious coating can reduce the frictional forces present on the surface of the device when another surface is moved against the device surface.
- a catheter having a coating that provides improved lubricity will encounter less frictional resistance when moved within a portion of the body, as compared to an uncoated substrate, or a coating that is not lubricious.
- Lubricity can also be important for devices with inner moving parts in addition to devices that function along with another device, for example, a coronary catheter which guides the insertion of a PTCA catheter. The methods can be used to prepare lubricious coatings for short term use and/or single use devices.
- Improved lubricity can be shown by one or more methods.
- One method of testing lubricity of a coating is by the horizontal sled style friction test method (such as ASTM D-1894; a modified test is described herein).
- the lubricity measurements described herein refer to the kinetic coefficient of friction, which is equal to the average force reading obtained during uniform sliding of the surfaces divided by the sled weight. The measurements are in grams.
- the biodegradable polysaccharide coatings generally show a lubricity of 20 g or less, and coatings having a lubricity of 15 g or less, or 10 g or less can be prepared by the methods described herein.
- a biodegradable polysaccharide matrix is formed using a photoinitiator to promote association of the biodegradable polysaccharides and matrix formation.
- Another method which can be used to demonstrate an improvement in lubricity is the water contact angle measurement method. Reduction of water contact angle is indicative of increased wettability, which associates with an improvement in lubricity.
- the biodegradable coating of the invention demonstrates excellent durability.
- durability refers to the wear resistance of the biodegradable polysaccharide coating. A more durable coating is less easily removed from a substrate by abrasion. Durability of a coating can be assessed by subjecting the device to conditions that simulate use conditions. The durability of the coating compositions is demonstrated by the ability to adhere to the device surface sufficiently to withstand the effect of shear forces encountered during insertion and/or removal of the device, which could otherwise result in delamination of the coating from the body member.
- Improved durability can be shown by one or more methods.
- One preferred method of testing durability, as well as lubricity, is by the horizontal sled style friction test method described herein. In the method multiple push-pull cycles are performed and friction measurements are taken during the measurements. If minimal increase in friction values is seen after multiple push pull cycles, the coating is though to have good durability.
- the biodegradable coatings of the present invention showed excellent durability.
- the lubricity of the fifth push and pull cycle generally was not greater than 10% or more typically not greater than 5% of the lubricity of the first push and pull cycle.
- the lubricity generally was not greater than 20% or more typically not greater than 10% of the lubricity of the first push and pull cycle.
- Amylose having polymerizable vinyl groups was prepared by mixing 0.75 g of amylose (A0512; Aldrich) with 100 mL of methylsulfoxide (J T Baker) in a 250 mL amber vial, with stirring. After one hour, 2 mL of triethylamine (TEA; Aldrich) was added and the mixture was allowed to stir for 5 minutes at room temperature. Subsequently, 2 mL of glycidyl acrylate(Polysciences) was added and the amylose and glycidyl acrylate were allowed to react by stirring overnight at room temperature. The mixture containing the amylose-glycidyl acrylate reaction product was dialyzed for 3 days against DI water using continuous flow dialysis. The resultant acrylated-amylose (0.50 g; 71.4% yield) was then lyophilized and stored desiccated at room temperature with protection from light.
- TEA triethylamine
- a polymerization initiator was prepared by copolymerizing a methacrylamide having a photoreactive group with acrylamide.
- a methacrylamide-oxothioxanthene monomer(N-[3-(7-Methyl-9-oxothioxanthene-3-carboxamido)propyl]methacrylamide (MTA-APMA)) was first prepared.
- N-(3-aminopropyl)methacrylamide hydrochloride (APMA), 4.53 g (25.4 mmol), prepared as described in U.S. Pat. No. 5,858,653, Example 2 was suspended in 100 mL of anhydrous chloroform in a 250 mL round bottom flask equipped with a drying tube.
- 7-methyl-9-oxothioxanthene-3-carboxylic acid (MTA) was prepared as described in U.S. Pat.
- MTA-chloride (MTA-Cl) was made as described in U.S. Pat. No. 6,007,833, Example 1. After cooling the slurry in an ice bath, MTA-Cl (7.69 g; 26.6 mmol) was added as a solid with stirring to the APMA-chloroform suspension. A solution of 7.42 mL (53.2 mmol) of TEA in 20 mL of chloroform was then added over a 1.5 hour time period, followed by a slow warming to room temperature. The mixture was allowed to stir 16 hours at room temperature under a drying tube.
- MTA-APMA was then copolymerized with acrylamide in DMSO in the presence of 2-mercaptoethanol (a chain transfer agent), N,N,N′,N′-tetramethyl-ethylenediamine (a co-catalyst), and 2,2′-azobis(2-methyl-propionitrile) (a free radical initiator) at room temperature.
- 2-mercaptoethanol a chain transfer agent
- N,N,N′,N′-tetramethyl-ethylenediamine a co-catalyst
- 2,2′-azobis(2-methyl-propionitrile) a free radical initiator
- acrylated-amylose as prepared in Example 1 was placed in an 8 mL amber vial.
- MTA-PAAm lyophilized
- 2-NVP N-vinyl-2-pyrrolidone; accelerant (Bimax)
- 1 ⁇ PBS 1 ⁇ phosphate-buffered saline
- the reagents were then mixed for one hour on a shaker at 37° C.
- the mixture in an amount of 50 ⁇ L was placed onto a glass slide (2991FI; Esco) and illuminated for 50 seconds with an EFOS 100 SS illumination system equipped with a 400-500 nm filter (50 mW/cm 2 ). After illumination the polymer was found to form a semi-firm gel having elastomeric properties.
- the product was analyzed by gas chromatography and was found to contain 71% of the desired 4-bromomethylbenzophenone, 8% of the dibromo product, and 20% unreacted 4-methylbenzophenone.
- the reaction mixture was washed with 10 g of sodium bisulfite in 100 mL of water, followed by washing with 3 ⁇ 200 mL of water.
- the product was dried over sodium sulfate and recrystallized twice from 1:3 toluene:hexane. After drying under vacuum, 635 g of 4-bromomethylbenzophenone was isolated, providing a yield of 60%, having a melting point of 112° C.-114° C.
- N,N,N′,N′-Tetramethylethylenediamine (6 g; 51.7 mmol) was dissolved in 225 mL of chloroform with stirring.
- BMBP 29.15 g; 106.0 mmol
- the reaction mixture was stirred at room temperature for 72 hours. After this time, the resulting solid was isolated by filtration and the white solid was rinsed with cold chloroform. The residual solvent was removed under vacuum and 34.4 g of solid was isolated for a 99.7% yield, melting point 218° C.-220° C.
- the PET substrate with the applied amylose mixture was placed in a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 15 cm from the light source, and illuminated for 60 seconds. After illumination, the applied amylose mixture was found to form a semi-firm gel on the PET substrate, with elastomeric properties evident.
- Dymax Lightweld PC-2 illumination system Dymax Corp.; light intensity 6.5 mW/cm 2
- a maleimide functional acid was prepared in the following manner, and was used in Example 8.
- EACA 6-aminocaproic acid
- Maleic anhydride (78.5 g; 0.801 moles)
- Maleic anhydride was dissolved in 200 mL of acetic acid and added to the EACA solution.
- the mixture was stirred one hour while heating on a boiling water bath, resulting in the formation of a white solid. After cooling overnight at room temperature, the solid was collected by filtration and rinsed two times with 50 mL of hexane each rinse.
- Chlorotrimethyl silane (600 mL; 510 g, 4.69 moles) was then added over two hours. The reaction was brought to reflux and was refluxed overnight ( ⁇ 16 hours). The reaction was cooled and added to a mixture of CHCl 3 (500 mL), water (1.0 liters), ice (300 g), and 12 N hydrochloric acid (240 mL) in a 20 liter container over 15 minutes. After 15 minutes of stirring, the aqueous layer was tested to make sure the pH was less than 5. The organic layer was separated, and the aqueous layer was extracted three times with CHCl 3 (700 mL) each extraction. The organic layers were combined and evaporated on a rotary evaporator.
- the N-(6-oxo-6-azidohexyl)maleimide (Compound 4) solution was further dried by gentle swirling over molecular sieves over night.
- the cold azide solution was filtered and added to refluxing CHCl 3 , 5 mL over a 10 minute period.
- the azide solution was refluxed for 2 hours.
- the weight of Mal-C5-NCO (Compound 5) solution obtained was 55.5 g, which was protected from moisture.
- CEA from Example 9 51 g; ⁇ 0.35 mole
- DMF dimethyl formamide
- 0.2 mL 0.26 mmole
- CH 2 Cl 3 100 mL
- the CEA solution was added slowly (over 2 hours) to a stirred solution of oxalyl chloride (53 mL; 0.61 mole), DMF (0.2 mL; 2.6 mmole), anthraquinone (0.5 g; 2.4 mmole), phenothiazine (0.1 g, 0.5 mmole), and CH 2 Cl 3 (75 mL) in a 500 mL round bottom flask in an ice bath at 200 mm pressure.
- oxalyl chloride 53 mL; 0.61 mole
- DMF 0.2 mL; 2.6 mmole
- anthraquinone 0.5 g; 2.4 mmole
- phenothiazine 0.1 g, 0.5 mmole
- CH 2 Cl 3
- CEA-Cl from Example 10 (109.2 g; 0.671 mole) was dissolved in acetone (135 mL). Sodium azide (57.2 g; 0.806 mole) was dissolved in water (135 mL) and chilled. The CEA-Cl solution was then added to the chilled azide solution with vigorous stirring in an ice bath for 1.5 hours. The reaction mixture was extracted two times with 150 mL of CHCl 3 each extraction. The CHCl 3 solution was passed through a silica gel column 40 mm in diameter by 127 mm. The 3-azido-3-oxopropyl acrylate (Compound 8) solution was gently agitated over dried molecular sieves at 4° C. overnight. The dried solution was used in Example 12 without purification.
- Compound 8 3-azido-3-oxopropyl acrylate
- the dried azide solution (from Example 11) was slowly added to refluxing CHCl 3 , 75 mL. After the addition was completed, refluxing was continued 2 hours.
- the EA-NCO (Compound 9) solution (594.3 g) was protected from moisture.
- a sample of the EA-NCO solution (283.4 mg) was mixed with DBB (8.6 mg) and evaporated.
- Maltodextrin (MD; Aldrich; 9.64 g; ⁇ 3.21 mmole; DE (Dextrose Equivalent): 4.0-7.0) was dissolved in dimethylsulfoxide (DMSO) 60 mL. The size of the maltodextrin was calculated to be in the range of 2,000 Da-4,000 Da.
- DMSO dimethylsulfoxide
- a solution of EA-NCO from Example 12 24.73 g; 19.3 mmole was evaporated and dissolved in dried DMSO (7.5 mL). The two DMSO solutions were mixed and heated to 55° C. overnight. The DMSO solution was placed in dialysis tubing (1000 MWCO, 45 mm flat width ⁇ 50 cm long) and dialyzed against water for 3 days.
- the macromer solution was filtered and lyophilized to give 7.91 g white solid.
- a sample of the macromer (49 mg), and DBB (4.84 mg) was dissolved in 0.8 mL DMSO-d 6 : 1 H NMR (DMSO-d 6 , 400 MHz) ⁇ 7.38 (s, 4H; internal std. integral value of 2.7815), 6.50, 6.19, and 5.93 (doublets, 3H; vinyl protons integral value of 3.0696).
- the calculated acrylate load of macromer was 0.616 ⁇ moles/mg of polymer.
- Example 13 A procedure similar to Example 13 was used to make the MD-Mal macromer.
- a solution of Mal-C5-NCO from Example 8 (0.412 g; 1.98 mmole) was evaporated and dissolved in dried DMSO (2 mL).
- MD (0.991 g; 0.33 mmole) was dissolved in DMSO (5 mL).
- the DMSO solutions were combined and stirred at 55° C. for 16 hours. Dialysis and lyophilization gave 0.566 g product.
- a sample of the macromer 44 mg
- DBB (2.74 mg) was dissolved in 00.8 mL DMSO-d 6 : 1 H NMR (DMSO-d 6 , 400 MHz) ⁇ 7.38 (s, 4H; internal std.
- Example 13 250 mg of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 3 mg of MTA-PAAm (lyophilized), 2 ⁇ L of 2-NVP, and 1 mL of 1 ⁇ phosphate-buffered saline (1 ⁇ PBS), providing a composition having MD-Acrylate at 250 mg/mL. The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 50 ⁇ L was placed onto a glass slide and illuminated for 40 seconds with an EFOS 100 SS illumination system equipped with a 400-500 nm filter. After illumination the polymer was found to form a semi-firm gel having elastomeric properties.
- MTA-PAAm lyophilized
- 2-NVP 1 ⁇ phosphate-buffered saline
- 1 ⁇ PBS 1 ⁇ phosphate-buffered saline
- Example 13 250 mg of MD-acrylate as prepared in Example 13 was placed in an 8mL amber vial. To the MD-Acrylate was added 14 mg of camphorquinone-10-sulfonic acid hydrate (Toronto Research Chemicals, Inc.), 3 ⁇ L of 2-NVP, and 1 mL of distilled water. The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 50 ⁇ L was placed onto a glass slide and illuminated for 40 seconds with a SmartliteIQTM LED curing light (Dentsply Caulk). After illumination the polymer was found to form a semi-firm gel having with elastomeric properties.
- camphorquinone-10-sulfonic acid hydrate Toronto Research Chemicals, Inc.
- 2-NVP 2-NVP
- distilled water 1 mL
- the reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 50 ⁇ L was placed onto a glass slide
- Example 14 250 mg of MD-Mal as prepared in Example 14 was placed in an 8 mL amber vial. To the MD-Mal was added 3 mg of MTA-PAAm (lyophilized), 2 ⁇ L of 2-NVP, and 1 mL of 1 ⁇ phosphate-buffered saline (1 ⁇ PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 50 ⁇ L was placed onto a glass slide and illuminated for 40 seconds with an EFOS 100 SS illumination system equipped with a 400-500 nm filter. After illumination the polymer was found to form a semi-firm gel having elastomeric properties.
- MTA-PAAm lyophilized
- 2-NVP 1 ⁇ phosphate-buffered saline
- 1 ⁇ PBS 1 ⁇ phosphate-buffered saline
- photo-PVP photo-derivatized poly(vinylpyrrolidone)
- photo-PVP photo-derivatized poly(vinylpyrrolidone)
- photoinitiator tetrakis (4-benzoylbenzyl ether) of pentaerythritol [“tetra-BBE-PET”] 5 mg
- IPA isopropyl alcohol
- PR01 100 mg photo-derivatized poly(vinylpyrrolidone)
- a 1.2 cm PEBAX® rod (Medical Profiles, Inc) was dipped into the solution for 10 seconds, at a dip rate of 0.1 cm/second, and then removed at the same rate. The rod was allowed to air dry for 5 minutes. The rod was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 30 cm from light source, illuminated for 180 seconds, and then removed.
- Dymax Lightweld PC-2 illumination system Dymax Corp.; light intensity 6.5 mW/cm 2
- MD-Acrylate 300 mg was placed in an 8 mL amber vial.
- MD-Acrylate 4,5-bis(4-benzoylphenylmethyleneoxy)benzene-1,3-disulfonic acid (5 mg) (DBDS), prepared as described in U.S. Pat. No. 6,278,018 (Example 1) and commercially available from SurModics, Inc. (Eden Prairie, Minn.) as DBDS, and 1 mL of 1 ⁇ phosphate-buffered saline (1 ⁇ PBS).
- DBDS 4,5-bis(4-benzoylphenylmethyleneoxy)benzene-1,3-disulfonic acid
- DBDS 4,5-bis(4-benzoylphenylmethyleneoxy)benzene-1,3-disulfonic acid
- DBDS 4,5-bis(4-benzoylphenylmethyleneoxy)benzene-1,3-disulfonic acid
- DBDS 4,5
- the mixture in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR).
- the photo-PVP/tetra-BBE-PET coated PEBAX® rod was dipped into the mixture for 30 seconds, at a dip rate of 0.3 cm/s, and then removed at the same rate.
- the rod was immediately placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 30 cm from light source, and illuminated for 180 seconds and then removed.
- the MD-Acrylate coated rod was examined under Scanning Electron Microscope (SEM; LEO Supra 35 VP); the MD-Acrylate coating thickness varied from 2.1 ⁇ m to 2.5 ⁇ m, with an average coating thickness of 2.3 ⁇ m.
- Example 13 500 mg of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 3 mg of MTA-PAAm (lyophilized), 2 ⁇ L of 2-NVP, and 1 mL of 1 ⁇ phosphate-buffered saline (1 ⁇ PBS). The reagents were then mixed for one hour on a shaker at 37° C. To this mixture was added either 5 mg 70 kD FITC-Dextran or 5 mg 10 kD FITC-Dextran (Sigma) and vortexed for 30 seconds.
- MTA-PAAm lyophilized
- 2-NVP 1 ⁇ phosphate-buffered saline
- 1 ⁇ PBS 1 ⁇ phosphate-buffered saline
- the mixture in an amount of 200 ⁇ L was placed into a Teflon well plate (8 mm diameter, 4 mm deep) and illuminated for 40 seconds with an EFOS 100 SS illumination system equipped with a 400-500 nm filter.
- the formed matrix was loose, and not as well crosslinked as the formed MD-acrylate matrix in Example 17.
- the matrix was transferred to a 12 well plate (Falcon) and placed in a well containing 0.6 mL PBS.
- 150 ⁇ L of PBS was removed from each well and placed into a 96 well plate. The remaining 850 ⁇ L were removed from the samples, and replaced with 1 mL fresh PBS.
- the 96 well plate was analyzed for FITC-Dextran on a spectrophotometer (Shimadzu) at 490 absorbance. Results showed that at least 70% of the detectable 10 kd or 70 kD FITC-Dextran was released from the matrix after 2 days. Visual observation showed that an unquantified amount of 10 kD or 70 kD FITC-Dextran remained within the matrix after 6 days.
- a MD-Acrylate-coated PEBAX rod (from Example 18) was placed in 5 mL of 1 ⁇ phosphate-buffered saline (PBS) containing 24 ⁇ g alpha-Amylase (Sigma; catalog # A6814) for 7 days on a rotating plate at 37° C. After 7 days, the rod was removed from the PBS and washed with distilled water. The rod was then examined under a Scanning Electron Microscope (LEO Supra 35 VP); upon examination, no trace of the MD-Acrylate coating was detected.
- PBS phosphate-buffered saline
- alpha-Amylase Sigma; catalog # A6814
- a MD-Acrylate-coated PEBAX was placed in 1 ⁇ phosphate-buffered saline (PBS) without alpha-Amylase; upon examination, the MD-Acrylate coating was intact and showed no signs of degradation.
- PBS phosphate-buffered saline
- Polyalditol (PA; GPC; 9.64 g; ⁇ 3.21 mmole) was dissolved in dimethylsulfoxide (DMSO) 60 mL. The size of the polyalditol was calculated to be in the range of 2,000 Da-4,000 Da.
- a solution of EA-NCO from Example 12 (24.73 g; 19.3 mmole) was evaporated and dissolved in dried DMSO (7.5 mL). The two DMSO solutions were mixed and heated to 55° C. overnight. The DMSO solution was placed in dialysis tubing (1000 MWCO, 45 mm flat width ⁇ 50 cm long) and dialyzed against water for 3 days.
- the polyalditol macromer solution was filtered and lyophilized to give 7.91 g white solid.
- a sample of the macromer (49 mg), and DBB (4.84 mg) was dissolved in 0.8 mL DMSO-d 6 : 1 H NMR (DMSO-d 6 , 400 MHz) ⁇ 7.38 (s, 4H; internal std. integral value of 2.7815), 6.50, 6.19, and 5.93 (doublets, 3H; vinyl protons integral value of 3.0696).
- the calculated acrylate load of macromer was 0.616 ⁇ moles/mg of polymer.
- Example 13 1,100 milligrams of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 1 mg of a photoinitiator 4,5-bis(4-benzoylphenyl-methyleneoxy)benzene-1,3-disulfonic acid (5 mg) (DBDS) and 1 mL of 1 ⁇ phosphate-buffered saline (PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 uL was injected, using a 23 gauge needle, into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.).
- DBDS photoinitiator 4,5-bis(4-benzoylphenyl-methyleneoxy)benzene-1,3-disulfonic acid
- PBS 1 ⁇ phosphate-buffered saline
- the tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm, which indicated complete polymerization of the MD-Acrylate. No excess liquid was observed. The filament was manipulated with forceps. Maltodextrin filaments were also made from a MD-acrylate solution having concentration of 200 mg/mL. These are physically firm and same as 1,100 mg/ml.
- Example 21 1,500 milligrams of polyalditol-acrylate as prepared in Example 21 was placed in an 8 ml amber vial. To the polyalditol-acrylate was added 1 mg of DBDS (lyophilized), 15 mg Bovine Serum Albumin, and 200 uL of 1 ⁇ phosphate-buffered saline (PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 uL was injected, using a 23 gauge needle, into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.).
- DBDS lyophilized
- Bovine Serum Albumin 1 ⁇ phosphate-buffered saline
- PBS 1 ⁇ phosphate-buffered saline
- the tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm, which indicated complete polymerization of the polyalditol-acrylate. No excess liquid was observed. The filament was manipulated with forceps
- Maltodextrin-acrylate filaments were synthesized using 200 mg/mL and 1100 mg/mL MD-acrylate as described in example 22 and were tested for degradation in Amylase solutions. These filaments were placed in microcentrifuge tubes containing 1 mL of either 1 ⁇ PBS (control), 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL (Sigma; catalog # A6814), or 1 ⁇ PBS containing alpha-Amylase at 24 ⁇ g/mL. The tubes were then placed in an incubator at 37° C.
- the 1100 mg/mL filament After 33 days in the 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL, the 1100 mg/mL filament had lost some of its initial firmness (as noted by the slightly curled appearance of the filament), but was still completely intact. The 1,100 mg/mL filament in the PBS with 24 ug Amylase had completely degraded after 48 hours. The 1,100 mg/ml filament in the PBS showed no signs of degradation.
- MD-Acrylate in an amount of 1,100 milligrams of as prepared in Example 13 was placed in an 8 ml amber vial. To the MD-Acrylate was added 1 mg of DBDS (lyophilized), 15 mg Bovine Serum Albumin (representing the bioactive agent; and 1 mL of 1 ⁇ phosphate-buffered saline (1 ⁇ PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 uL was injected, using a 23 gauge needle, into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.).
- the tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm, which indicated complete polymerization of the MD-Acrylate. No excess liquid was observed.
- Dymax Lightweld PC-2 illumination system Dymax Corp.; light intensity 6.5 mW/cm 2
- the filament was placed in a 1.7 ml microcentrifuge tube with 1 ml 1 ⁇ PBS. At daily intervals for 6 days, 150 ⁇ L of PBS was removed from each well and placed into a 96 well plate for subsequent analysis. The remaining 850 ⁇ L was removed from the sample, and to the tube was added 1 ml of 1 ⁇ PBS. After 6 days, the filament was placed in a 1.7 ml microcentrifuge tube with 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL. At daily intervals for 35 days, 150 ⁇ L of PBS was removed from each well and placed into a 96 well plate for subsequent analysis.
- Polyaldtiol-acrylate in an amount of 1,500 mg of as prepared in Example 21 was placed in an 8 ml amber vial.
- To the PA-Acrylate was added 1 mg of DBDS (lyophilized), 15 mg Bovine Serum Albumin, and 1 mL of 1 ⁇ phosphate-buffered saline (1 ⁇ PBS).
- the reagents were then mixed for one hour on a shaker at 37° C.
- the mixture in an amount of 10 uL was injected, using a 23 gauge needle, into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.).
- the tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm, which indicated complete polymerization of the polyalditol-acrylate. No excess liquid was observed. The filament was manipulated with forceps.
- the filament was placed in a 1.7 ml microcentrifuge tube with 1 ml PBS containing alpha-Amylase at 0.121 ⁇ g/mL. At daily intervals for 15 days, 150 ⁇ l of PBS was removed from each well and placed into a 96 well plate for subsequent analysis. The remaining 850 ⁇ L was removed from the sample, and to the tube was added 1 ml of fresh PBS containing alpha-Amylase at 0.121 ⁇ g/mL. The 96-well plate was analyzed for BSA using the Quanitpro Assay Kit (Sigma).
- Maltodextrin filaments were synthesized using a 1,100 mg/mL solution as described in example 25 using an anti-horseradish peroxidase antibody (P7899; Sigma) instead of BSA.
- the filament contained 800 ug of the anti-horseradish peroxidase antibody.
- the filament was placed in a 1.7 ml microcentrifuge tube containing 1 ml of 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL. At daily intervals for 5 days, 100 ⁇ l of PBS was removed from the sample, placed into a 96 well plate and incubated for 60 minutes at 37° C.
- the remaining 850 ⁇ L was removed from the sample, and replaced with 1 ml fresh 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL. After 1 hour, the plate was washed three times with 1 ml PBS/Tween (Sigma). 150 ul StabilCoatTM Immunoassay Stabilizer (SurModics, Eden Prairie, Minn.) was added to the well and incubated for 30 minutes at room temperature. After 30 minutes, the 96-well plate was washed three times with PBS/Tween. A solution of 0.5 mg/ml Horseradish Peroxidase (Sigma) in 1 ⁇ PBS (100 uL) was added to the well and incubated for 60 minutes.
- the 96-well plate was washed six times with PBS/Tween. A chromogenic assay was then performed. After 15 minutes, the 96 well plate was analyzed for HRP conjugate on a spectrophotometer (Tecan) at 560 nm absorbance. Detectable Antibody was found at each time point.
- MD-Acrylate 1000 mg was placed in an 8 mL amber vial.
- To the MD-Acrylate was added 5 mg DBDS, 1 ml of 1 ⁇ phosphate-buffered saline (PBS), and 100 mg BSA.
- the reagents were then mixed for one hour on a shaker at 37° C.
- the mixture in an amount of 1 ml was placed into a 1.8 ml eppendorf tube (VWR).
- the photo-PVP/tetra-BBE-PET coated PEBAX® rod was dipped into the mixture for 30 seconds, at a dip rate of 0.3 cm/s, and then removed at the same rate.
- the rod was immediately placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 30 cm from light source, and illuminated for 180 seconds and then removed.
- Dymax Lightweld PC-2 illumination system Dymax Corp.; light intensity 6.5 mW/
- the rod was placed in a 1.7 ml microcentrifuge tube with 1 ml 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL.
- 150 ⁇ l of PBS was removed from each well and placed into a 96 well plate.
- the remaining 850 ⁇ l were removed from the samples, and replaced with 1 ml fresh 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL.
- the 96 well-plate was analyzed for BSA release using the Quanitpro Assay Kit (Sigma). BSA was detected at each timepoint. Results are shown in Table 3 and FIG. 2 . TABLE 3 Timepoint Cumulative BSA release (% of Total BSA) 1 7.0 2 15.0 3 19.1 4 22.7 5 25.6 7 28.6 8 31.5 9 34.2
- the MD-Acrylate-coated PEBAX rod (as prepared in Example 28) was placed in 5 ml of 1 ⁇ phosphate-buffered saline (PBS) containing alpha-Amylase at 24 ⁇ g/mL for 7 days on a rotating plate at 37° C. After 7 days, the rod was removed from the PBS and washed with distilled water. The rod was then examined under a Scanning Electron Microscope (LEO Supra 35 VP); upon examination, no trace of the MD-Acrylate coating was detected.
- PBS phosphate-buffered saline
- a circumferential dissection of the anterior segment (cornea, aqueous humour, lens) of porcine eye was performed, and the vitreous was squeezed out from the globe into a 20 mL amber vial; approx 10 mL total was retrieved from a total of four eyes.
- 200 mg/mL and 1100 mg/mL Maltodextrin filaments, formed in example 21, were placed into 2 mL of the vitreous solution, and placed at 37° C. on a rotator plate. The 200 mg/mL filament had completely dissolved after 24 hours. The 1,100 mg/mL filaments completely degraded after 30 days in the vitreous.
- Solution #1 was prepared as follows: 250 mg of MD-acrylate as prepared in example 13 was placed in an 8 mL vial. To the MD-acrylate was added 15 mg ferrous gluconate hydrate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 uL AMPS (Lubrizol) and 1,000 uL deionized water.
- Solution #2 was prepared as follows: 250 mg of MD-acrylate as prepared in example 13 was placed in a second 8 mL vial. To this MD-acrylate was added 30 uL AMPS, 80 uL Hydrogen Peroxide (Sigma) and 890 uL 0.1 M Acetate buffer (pH 5.5).
- Solution #1 Two solutions were prepared, similar to Example 31, but in this Example Solution #1 different concentrations of ferrous gluconate hydrate (Sigma) and ascorbic acid were used.
- Solution #1 was prepared as follows: 250 mg of MD-acrylate (as prepared in example 13) was placed in an 8 mL vial. To the MD-acrylate was added 5 mg ferrous gluconate hydrate (Sigma), 40 mg ascorbic acid (Sigma), 67 uL AMPS (Lubrizol) and 1,000 uL deionized water.
- Solution #2 was prepared as follows: 250 mg of MD-acrylate as prepared in example 7 was placed in a second 8 mL vial. To this MD-acrylate was added 30 uL AMPS, 80 uL Hydrogen Peroxide (Sigma) and 890 uL 0.1 M Acetate buffer (pH 5.5).
- Solution #1 was prepared as follows: 250 mg of MD-acrylate (as prepared in example 13) was placed in an 8 mL vial. To the MD-acrylate was added 15 mg Iron (II) L-Ascorbate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 uL AMPS (Lubrizol) and 1,000 uL deionized water.
- Solution #2 was prepared as follows: 250 mg of MD-acrylate as prepared in example 7 was placed in a second 8 mL vial. To this MD-acrylate was added 30 uL AMPS, 80 uL hydrogen peroxide (Sigma) and 890 uL 0.1 M Acetate buffer (pH 5.5).
- Solution #1 was prepared as follows: 1,000 mg of Polyalditol-acrylate as prepared in Example 21 was placed in an 8 mL vial. To the Polyalditol-acrylate was added 15 mg Ferrous Sulfate Heptahydrate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 uL AMPS (Lubrizol) and 1,000 uL deionized water; Solution #2 was prepared as follows: 1,000 mg of Polyalditol-acrylate as prepared in example was placed in a second 8 mL vial. To this Polyalditol-acrylate was added 30 uL AMPS, 80 uL Hydrogen Peroxide (Sigma) and 890 uL 0.1 M Acetate buffer (pH 5.5).
- Photo-PVP 100 mg
- the photoinitiator tetra-BBE-PET 5 mg
- IPA isopropyl alcohol
- the mixture in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR).
- a 1.2 cm PEBAX® rod (Medical Profiles, Inc) was dipped into the solution for 10 seconds, at a dip rate of 0.1 cm/second, and then removed at the same rate.
- the rod was allowed to air dry for 5 minutes.
- the rod was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 30 cm from light source, illuminated for 180 seconds, and then removed.
- Solution #1 was prepared as follows: 250 mg of MD-acrylate (as prepared in example 13) was placed in an 8 ml vial. To the MD-acrylate was added 15 mg Iron (II) Ascorbate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 ul AMPS (Lubrizol) and 1,000 ul deionized water.
- Solution #2 was prepared as follows: 250 mg of MD-acrylate was placed in a second 8 ml vial. To this MD-acrylate was added 30 ul AMPS, 80 ul Hydrogen Peroxide (Sigma) and 890 ul 0.1 M Acetate buffer (pH 5.5).
- Solution #1 in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR).
- the photo-PVP/tetra-BBE-PET coated PEBAX® rod was dipped into the mixture for 30 seconds, at a dip rate of 0.5 cm/s, and then removed at the same rate.
- the PEBAX rod was allowed to air dry for 10 minutes.
- Solution #2 in an amount of 1 mL was placed into a second 1.8 mL eppendorf tube (VWR).
- the photo-PVP/tetra-BBE-PET and Solution #1 coated PEBAX® rod was dipped into the mixture for 30 seconds, at a dip rate of 0.5 cm/s, and then removed at the same rate.
- the MD-Acrylate coated rod was examined under Scanning Electron Microscope (SEM; LEO Supra 35 VP); the MD-Acrylate coating thickness ranged from 15 to 20 um, with an average thickness of 16.8 um.
- Solution #1 was prepared as follows: 250 mg of MD-acrylate (as prepared in example 13) was placed in an 8 ml vial. To the MD-acrylate was added 15 mg Iron (II) Acetate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 ul AMPS (Lubrizol), 75 mg Bovine Serum Albumin (BSA; representing the bioactive agent) and 1,000 ⁇ L deionized water. Solution #1 was prepared as follows: 250 mg of MD-acrylate was placed in a second 8 ml vial. To this MD-acrylate was added 30 ⁇ L AMPS, 80 ⁇ L Hydrogen Peroxide (Sigma), 75 mg BSA and 890 ⁇ L Acetate buffer (pH 5.5).
- Maltodextrin-acrylate filaments were prepared using the reagents at concentrations as described in Example 31. These filaments were placed in microcentrifuge tubes containing 1 ml either Phosphate Buffered Saline (PBS) or 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL. The tubes were then placed in an incubator at 37° C.
- PBS Phosphate Buffered Saline
- 1 ⁇ PBS containing alpha-Amylase 0.121 ⁇ g/mL.
- Example 13 600 milligrams of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 5 mg of DBDS (lyophilized), 10 mg Rabbit Anti-Goat Fragment Antibody (catalog # 300-007-003; Jackson Immunological Research, West Grove, Pa.) and 1 mL of 1 ⁇ phosphate-buffered saline (PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 ⁇ L was pipetted into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.).
- the tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm and completely crosslinked, with no excess liquid.
- the filament was placed in a 1.7 mL microcentrifuge tube with 0.5 ml 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL (eluent solution). At predetermined intervals for 17 days, 200 ⁇ L of the eluent solution was removed from each tube, and 100 ⁇ L was placed into two 96 well plates.
- the remaining 300 ⁇ L were removed from the samples, and replaced with 0.5 mL fresh 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL.
- the 96 well plates were analyzed for total FAB molecule release and FAB activity using an Enzyme-Linked Immunosorbent Assay (ELISA). Briefly, the 100 ⁇ L eluent solution was incubated at 37° C. for one hour and then washed 3 ⁇ with 2 ml PBS/Tween 20 (Sigma). The wells were blocked with 100 ⁇ L StabilCoatTM for 1 hour at room temperature and then washed 3 ⁇ with 2 mL PBS/Tween 20.
- 316V or 304V stainless steel rods (0.035′′ diameter; Small Parts, Inc.) were cleaned by wiping the rods with isopropyl alcohol (IPA) soaked Alpha 10 clean-wipes (Texwipe; Kernersville, N.C.) and then sonicating the rods in a solution of 10% Valtron SP2200 detergent (Valtech Corp.) in hot tap water for 10 minutes. The rods were then rinsed 3 ⁇ with deionized water followed by a 1 minute sonication in hot tap water; the rinsed and sonication steps were then repeated.
- IPA isopropyl alcohol
- Alpha 10 clean-wipes Texwipe; Kernersville, N.C.
- Valtron SP2200 detergent Valtech Corp.
- a 0.5% 1,4-bis(trimethoxysilylethyl)benzene (B2495.6, UCT, Bristol, Pa.) solution in 89.5% IPA and 10% deionized water was prepared (See Example 1 of U.S. Pat. No. 6,706,408B2) and the cleaned rods were dipped into the silane solution within 2 minutes of coming out of the deionized rinse. The rods were allowed to dwell in the silane solution for 3 minutes and then pulled out at a rate of 1.0 cm/s. They were allowed to air dry for 2 minutes at room temperature and then placed at 110° C. for 5 minutes.
- a solution of photo-PVP (15 mg/mL), DBDS (1 mg/mL), tetra-BBE-PET 0.075 (mg/mL) and PVP K90 (20 mg/ml; BASF) was prepared in 60% IPA/40% water) (see also Example 1 of U.S. Pat. No. 6,706,408B2).
- the silane treated rods were dip coated into the above solution using the following parameters:
- the first solution (#1) was prepared by placing 600 mg of MD-acrylate (as prepared in example 13) into an 8 mL vial and then adding 9 mg iron (II) ascorbate (Sigma), 30 mg ascorbic acid (Sigma), 67 ⁇ L AMPS (Lubrizol), 16 mg Rabbit Anti-HRP antibody (Sigma; catalog # P7899), and 1,000 ⁇ L deionized water.
- the second solution (#2) was prepared by placing 600 mg of MD-acrylate in a second 8 mL vial and then adding 30 ⁇ L AMPS, 16 mg Rabbit Anti-HRP antibody (Sigma; catalog # P7899), 80 ⁇ L hydrogen peroxide (Sigma) and 890 ⁇ L 0.1 M Acetate buffer (pH 5.5).
- Solution #1 in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR).
- the (PV01/K90/DBDS/tetra-BBE-PET)-coated stainless steel rod (20 mm in length) was dipped into the mixture for 20 seconds, at a dip rate of 0.75 cm/s, and then removed at the same rate. The rod was allowed to air dry for 10 minutes.
- Solution #2 in an amount of 1 mL was placed into a second 1.8 mL eppendorf tube (VWR).
- the (PV01/K90/DBDS/tetra-BBE-PET)-coated and Solution #1 coated rod was dipped into the Solution #2 for 20 seconds, at a dip rate of 0.75 cm/s, and then removed at the same rate.
- Contact with Solution #2 initiated the redox reaction and caused formation of an MD-Acrylate coated layer on the PV01/K90/DBDS/tetra-BBE-PET coated layer; the MD-Acrylate coating was cured within 10 seconds
- the coated rod was examined under Optical Inferometer Microscope (Veeco) which revealed that the MD-Acrylate coating layer had a thickness of 70 ⁇ m.
- the MD-Acrylate coated rod was placed in a 0.6 mL microcentrifuge tube with 0.5 ml 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL (eluent solution) to assess release of the bioactive agent (antibody) from the coating.
- eluent solution was removed from the tube, which was divided into two 100 ⁇ L aliquots and placed into two 96 well plates. The remaining 300 ⁇ L was removed from the microcentrifuge tube, and 0.5 mL of fress eluent solution (1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL) was added to the microcentrifuge tube having the MD-Acrylate coated rod.
- ELISA Enzyme-Linked Immunosorbent Assay
- 316V or 304V stainless steel rods (0.035′′ diameter; Small Parts, Inc., Miami Lakers, Fla.) were cleaned by wiping the rods with isopropyl alcohol (IPA)-soaked Alpha 10 clean-wipe (Texwipe) and then sonicating the rods in a solution of 10% Valtron SP2200 detergent in hot tap water for 10 minutes. The rods were then rinsed 3 ⁇ with deionized water followed by a 1 minute sonication in hot tap water; the rinse and sonication step was then repeated.
- IPA isopropyl alcohol
- the cleaned rods were dipped into a 0.5% 1,4-bis(trimethoxysilylethyl)benzene solution (as described in Example 39) within 2 minutes of coming out of the deionized rinse.
- the rods were allowed to dwell in the silane solution for 3 minutes and then pulled out at a rate of 1.0 cm/s.
- the rods were allowed to air dry for 2 minutes at room temperature and then placed at 110° C. for 5 minutes.
- a solution of PV01 (15 mg/mL), DBDS (1 mg/mL), tetra-BBE-PET (0.075 mg/ml) and K90 (20 mg/mL; BASF) was prepared in 60% IPA/40% water.
- the silane treated rods were dip coated into the above solution using the following parameters:
- the coated rods were allowed to air dry at room temperature for 5 minutes and then illuminated in a Dymax lamp UV light chamber, with rotation, for 3 minutes. The dipping procedure was then repeated with a dwell time of 120 seconds (all other dipping parameters the same).
- MD-Acrylate 600 milligrams of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial.
- To the MD-Acrylate was added 5 mg of DBDS (lyophilized), 16 mg Rabbit Anti-HRP antibody (Sigma; catalog # P7899) and 1 ml of 1 ⁇ phosphate-buffered saline (PBS).
- the reagents were then mixed for one hour on a shaker at 37° C.
- the MD-Acrylate solution in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR).
- the (PV01/K90/DBDS/tetra-BBE-PET)-coated stainless steel rod (20 mm in length) was dipped into the mixture for 20 seconds, at a dip rate of 0.75 cm/s, and then removed at the same rate.
- the rod was then immediately illuminated in a Dymax lamp UV light chamber, with rotation, for 3 minutes.
- the rod was allowed to air dry for 5 minutes, and the dipping and illuminating procedures were repeated one more time.
- the MD-Acrylate coated rod was examined under Optical Inferometer Microscope (Veeco); the MD-Acrylate coating layer had a coating thickness of 20 ⁇ m.
- the MD-Acrylate coated rod was placed in a 0.6 mL microcentrifuge tube with 0.5 mL 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL (eluent solution) to assess degradation of the coating and release of the bioactive agent (antibody) from the coating.
- eluent solution 0.5 mL 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL
- the remaining 300 ⁇ L was removed from the microcentrifuge tube, and 0.5 mL of fress eluent solution (1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL) was added to the microcentrifuge tube having the MD-Acrylate coated rod.
- the 96 well plates were analyzed for total Rabbit Antibody molecule release and activity using an Enzyme-Linked Immunosorbent Assay (ELISA). Briefly, the 100 ⁇ L eluent solution was added to the wells and incubated at 37° C. for one hour and then washed 3 ⁇ with 2 mL PBS/Tween 20 (Sigma). The wells were blocked with 100 ⁇ L StabilCoatTM for 1 hour at room temperature and then washed 3 ⁇ with 2 mL PBS/Tween 20.
- ELISA Enzyme-Linked Immunosorbent Assay
- the MD-Acrylate coated rod was weighed at various time points to determine the amount of material of the MD-Acrylate coated layer lost from the rods due to amylase digestion. Results are shown in Table 6 and FIG. 5 .
- Example 13 600 milligrams of MD-Acrylate as prepared in Example 13 was placed in an 8 ml amber vial. To the MD-Acrylate was added 5 mg of DBDS (lyophilized), 16 mg Rabbit Antibody Anti-HRP (Sigma; catalog # P7899) and 1 ml of 1 ⁇ phosphate-buffered saline (PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 ⁇ L was pipetted into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.).
- the tubing was placed into a Dymax Lightweld PC-2 illuminationion system (Dymax Corp.; light intensity 6.5 mW/cm 2 ), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm and completely crosslinked, with no excess liquid.
- Dymax Lightweld PC-2 illumination system Dymax Corp.; light intensity 6.5 mW/cm 2
- the filament was placed in a 1.7 ml microcentrifuge tube with 0.5 ml 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL (eluent solution). At predetermined intervals for 25 days, 200 ⁇ l of the eluent solution was removed from each tube, and 100 ⁇ L was placed into two 96 well plates. The remaining 300 ⁇ l were removed from the samples, and replaced with 0.5 ml fresh 1 ⁇ PBS containing alpha-Amylase at 0.121 ⁇ g/mL. The 96 wellplates were analyzed for total Rabbit Antibody molecule release and activity using an Enzyme-Linked Immunosorbent Assay (ELISA).
- ELISA Enzyme-Linked Immunosorbent Assay
- the 100 ⁇ L eluent solution was added to the wells and incubated at 37 degrees C. for one hour and then washed 3 ⁇ with 2 ml PBS/Tween 20 (Sigma).
- the wells were blocked with 100 ⁇ L StabilCoatTM (SurModics) for 1 hour at room temperature and then washed 3 ⁇ with 2 ml PBS/Tween 20.
- MD-acrylate discs formed via redox polymerization of MD-acrylate coating solutions were tested for mechanical properties.
- a first solution (#1) was prepared by placing 300 mg of MD-acrylate as prepared in Example 13 into an 8 ml vial and then adding 9 mg iron (II) ascorbate (Sigma), 30 mg ascorbic acid (Sigma), 67 ⁇ L AMPS (Lubrizol), and 1,000 ⁇ L deionized water.
- Solution #2 prepared by placing 300 mg of MD-acrylate into a second 8 ml vial and then adding 30 ⁇ L AMPS, 80 ⁇ L if hydrogen peroxide (Sigma) and 890 ⁇ L of 0.1 M Acetate buffer (pH 5.5).
- Viscosity of the first and second solutions were determined on a Brookfield Viscometer. The average viscosity for both solutions was 10.9 cP.
- the modulus of the formed matrix was determined by rheological measurements.
- the first and second solutions were combined on the testing plate in the Rheometer (Rheometric Scientific; model # SR-2000) and the mixture was allowed to polymerize to form a matrix.
- Data recording began before sample was cured in plates. Briefly, 100 ⁇ L of solution #1 and 100 ⁇ L of solution #2 were mixed on the lower testing plate. As the matrix formed, the upper testing plate was lowered to fully contact the mixture of the first and second solutions as the mixture polymerized into matrix. The sample was cured within 15 seconds. This curing method ensured maximum contact between the two testing plates resulting in more accurate testing compared to preformed matrices being placed between the testing plates.
- the resulting MD-acrylate matrix had properties of an elastic solid with an elastic (storage) modulus ranging from 27 kPa to 30 kPa, and a viscous (loss) modulus of only about 1 kPa. Results are shown in Table 8 and FIG. 7 .
- Fricitional testing over the last 40 cycles of a 50 cycle test was evaluated.
- Coated rods were evaluated by a horizontal sled style friction test method (modified ASTM D-1894, as described below).
- Silicone Pads (7 mm diameter) were hydrated and then wrapped around a 200 gram stainless steel sled. The silicone pad was clipped together tightly on the opposite side of the sled.
- the sled with rotatable arm was then attached to a 500 gram Chatillon Digital Force Gauge (DGGHS, 500 ⁇ 0.1) with computer interface.
- the testing surface was mounted on a 22.5 inch positioning rail table with micro-stepper motor control (Compumotor SX6 Indexer/Drive).
- MD-coated rods from example 18 were hydrated in deionized water and clamped onto the test surface 1 inch (or approximately 2.5 cm) apart.
- the hydrated Silicone pad (jaw force set at 500 g) moved at 0.5 cm/sec over a 5 cm section for 50 push/pull cycles, and the final force measurements were taken over the last 40 push/pull cycles.
- the MD-acrylate coated rod As shown in Table 9 and FIG. 8 , compared to the benchmark synthetic coating (see example 1 of U.S. Pat. 6,706,408B2) the MD-acrylate coated rod provided a coating having excellent lubricity and durability; the rods coated with a MD-acrylate formulation containing a photoinitiator, the grams of force remained relatively constant for the last 40 cycles, indicating a durable coating.
- Maltodextrin having pendent butyryl groups were prepared by coupling butyric anhydride at varying molar ratios.
- butyrylated-MD (1 butyl/4 glucose units, 1:4 B/GU)
- Maltodextrin (MD; Aldrich; 11.0 g; 3.67 mmole; DE (Dextrose Equivalent): 4.0-7.0) was dissolved in dimethylsulfoxide (DMSO) 600 mL with stirring. The size of the maltodextrin was calculated to be in the range of 2,000 Da-4,000 Da.
- 1-methylimidazole Aldrich; 2.0 g, 1.9 mls
- butyric anhydride Aldrich; 5.0 g, 5.2 mls
- butyrylated-MD (1:2 B/GU)
- 10.0 g (10.4 mL) butyric anhydride was substituted for the amount of butyric anhydride described above.
- a yield of 96% (10.536 g) was obtained.
- butyrylated-MD-acrylate (1 butyl/4 glucose units, 1:4 B/GU) the following procedure was performed.
- MD-Acrylate (Example 13; 1.1 g; 0.367 mmoles) was dissolved in dimethylsulfoxide (DMSO) 60 mL with stirring.
- DMSO dimethylsulfoxide
- 1-methylimidazole (0.20 g, 0.19 mls)
- butyric anhydride (0.50 g, 0.52 mls) was added with stirring.
- the reaction mixture was stirred for four hours at room temperature. After this time, the reaction mixture was quenched with water and dialyzed against DI water using 1,000 MWCO dialysis tubing.
- the butyrylated starch acrylate was isolated via lyophylization to give 821 mg (75% yield, material lost during isolation).
- NMR confirmed a butyrylation of 1:3 B/GU (2.38 mmoles butyl/g sample).
- butyrylated-MD-acrylate To provide butyrylated-MD-acrylate the following procedure is performed. Butyrylated-MD (Example 43; 1.0 g; 0.333 mmole) is dissolved in dimethylsulfoxide (DMSO) 60 mL with stirring. Once the reaction solution is complete, a solution of EA-NCO from Example 12 (353 mg; 2.50 mmole) is evaporated and dissolved in dried DMSO 1.0 mL). The two DMSO solutions are mixed and heated to 55° C. overnight. The DMSO solution is placed in dialysis tubing (1000 MWCO) and dialyzed against water for 3 days. The macromer solution is filtered and lyophilized to give a white solid.
- DMSO dimethylsulfoxide
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Materials Engineering (AREA)
- Polymers & Plastics (AREA)
- Dermatology (AREA)
- Pharmacology & Pharmacy (AREA)
- Biochemistry (AREA)
- Biomedical Technology (AREA)
- Transplantation (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Heart & Thoracic Surgery (AREA)
- Vascular Medicine (AREA)
- Surgery (AREA)
- Molecular Biology (AREA)
- Wood Science & Technology (AREA)
- Ophthalmology & Optometry (AREA)
- Neurosurgery (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Materials For Medical Uses (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
- Prostheses (AREA)
- Biological Depolymerization Polymers (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Paints Or Removers (AREA)
Abstract
In vivo formed matrices including natural biodegradable polysaccharides are described. The matrix is formed from a plurality of natural biodegradable polysaccharides having pendent coupling groups. The matrix can also include a bioactive agent which can be released to provide a therapeutic effect to a patient.
Description
- The present non-provisional Application claims the benefit of commonly owned provisional Application having Ser. No. 60/719,466, filed on Sep. 21, 2005, and entitled ARTICLES AND COATINGS INCLUDING NATURAL BIODEGRADABLE POLYSACCHARIDES AND USES THEREOF.
- The present invention relates to in vivo formed matrices comprising a natural biodegradable polymeric material. Bioactive agents can be included in the in vivo formed matrices to provide a therapeutic effect to a patient.
- Recently, the use of drug-eluting stents (DES) in percutaneous coronary interventions has received much attention. DES are medical devices that present or release bioactive agent into their surroundings (for example, luminal walls of coronary arteries). Generally speaking, a bioactive agent can be coupled to the surface of a medical device by surface modification, embedded, and released from within polymeric materials (matrix-type), or surrounded by and released through a carrier (reservoir-type). The polymeric materials in such applications should optimally act as a biologically inert barrier and not induce further inflammation within the body. However, the molecular weight, porosity of the polymer, a greater percentage of coating exposed on the medical device, and the thickness of the polymer coating can contribute to adverse reactions to the medical device.
- Another way to deliver bioactive agents from the surface of a medical device is by using a coating that has a biodegradable polymer, such as polylactic acid. As the coating degrades, the bioactive agent is released from the surface of the device. Although biodegradable coatings that include polylactic acid have been described in a number of documents, for example, U.S. Pat. No. 6,258,121, there remains a need for improved coatings and coating materials.
- Some concerns exist that regard the use of biodegradable materials that degrade into materials that are not typically found in the body, or that are found at particularly low levels in the body. These types of biodegradable materials have the potential to degrade into products that cause unwanted side effects in the body by virtue of their presence or concentration in vivo. These unwanted side effects can include immune reactions, toxic buildup of the degradation products in the liver, or the initiation or provocation of other adverse effects on cells or tissue in the body.
- Another problem is that preparations of some biodegradable materials may not be obtained at consistent purity due to variations inherent in natural materials. This is relevant at least with regard to biodegradable materials derived from animal sources. Inconsistencies in preparations of biodegradable materials can result in problematic coatings.
- It is also desirable to provide biodegradable drug delivery coatings that are easy to prepare, cost effective, and that also offer a wide range of flexibility with regard to the type and amount of drug or drugs to be delivered from the biodegradable coating.
- Other aspects of the present invention relate to the use of polymeric coatings for providing a sealant function to medical articles. Biodegradable sealant compositions have been used on articles having porous surfaces, such as fabrics associated with implantable medical articles. The sealant coating initially renders the porous surface impermeable to fluids for a period of time. However, as the sealant materials degrade and are resorbed by the body, cells involved in tissue repair infiltrate the porous material and replace the sealant materials. Thus, newly formed tissue replaces the original function of the coated sealant over a period of time.
- Animal-derived sealant materials such as collagen and gelatin are commonly used to coat textile grafts. These materials can be resorbed in vivo. The blood clotting protein fibrin has also been utilized as a sealant material. Despite their uses, there are drawbacks and concerns with using these types of sealant materials. One particular problem is that it is difficult to produce consistent sealant compositions from these animal sources due to batch-to-batch variations inherent in their production.
- In many cases the collagen used in sealant technologies is obtained from non-human animal sources, such as bovine sources. In these cases there is the possibility that bovine collagen preparations may contain unwanted contaminants that are undesirable for introduction into a human subject. One example of an unwanted contaminant is the prionic particles that cause Bovine Spongiform Encephalopathy (BSE).
- BSE, also termed Mad Cow Disease, is one of a group of progressive neurological diseases called transmissible spongiform encephalopathies, or TSEs (named for deteriorated areas of the brain that look like sponges). Various forms of TSE have been reported, including scrapie in sheep and chronic wasting disease in elk and mule deer. It is generally believed that the use of recycled animal parts led to the cross-species contamination of scrapie in sheep to mad cow disease, and the ingestion of contaminated beef and bovine products led to the human variant of this disease, Creutzfeldt-Jakob Disease (CJD).
- Additional concerns are that preparations from animal sources may provide other unwanted contaminants, such as antigenic factors. These antigenic factors may promote a localized immune response in the vicinity of the implanted article and foul its function. These factors may also cause infection as well as local inflammation.
- While synthetic materials can be used in the preparation of sealant compositions, these synthetic materials have the potential of degrading into non-naturally occurring products. These non-naturally occurring products have the potential to be at least partially toxic to the organism or immunogenic and cause inflammation, as well as infection, at or around the site of implantation.
- In one aspect, the present invention provides compositions and methods for preparing biodegradable coatings that are particularly useful for coating surfaces of implantable medical devices, such as stents and catheters, and are capable of releasing bioactive agents from the device surface. These coating compositions include a natural biodegradable polysaccharide as a component that can be crosslinked to form a matrix from which a therapeutic material such as a drug, a biomolecule, or cells (referred to herein as a “bioactive agents”) can be released or retained. In some embodiments of the invention, a bioactive agent is present in and can be released from the biodegradable matrix; in other embodiments a bioactive agent is present in a biodegradable microparticle, the microparticle being immobilized within the matrix.
- In other aspects of the invention, the natural biodegradable polysaccharide is used to prepare an article, such as an article that can be implanted or formed within the body (for example, by in situ formation). In some aspects, the article can be amorphous, such as a polymerized mass of natural biodegradable polysaccharides that is formed within or on a portion of the body, by using an in vivo matrix-forming composition.
- In other aspects, the invention provides an article fabricated from natural biodegradable polysaccharide, wherein the article has a defined structure, and wherein the article can be implanted in the body (such as a filament). Such articles are referred to herein as “medical implants”. A medical implants having a defined structure can be formed by any suitable process, including molding, extruding, shaping, cutting, casting, and the like.
- The article can be used for one or more purposes, such as for releasing or retaining a bioactive agent at a location in the body. For example, the article can be a bioactive agent-containing medical implant or depot. The article can also provide one or more mechanical or physical properties to a portion the body. For example, the natural biodegradable polysaccharides can be included in a composition used for the formation of a biodegradable medical device such as a stent.
- In some aspects, the article, such as an in vivo formed matrix, is used in methods for the treatment of any one or more of a variety of medical conditions or indications, including restoring, improving, and/or augmenting tissue growth or function, in particular those for orthopedic, dental, and bone graft applications. These functions can be provided by placing a polymerized matrix of biodegradable polysaccharides in contact with a host tissue. The matrix can restore or improve tissue growth or function by, for example, promoting or permitting formation of new tissue between and into the matrix. The effect on tissue can be caused by the biodegradable polysaccharide itself, or the biodegradable polysaccharide in combination with one or more bioactive agent(s) that can be present in and/or released from the matrix. Exemplary bioactive agents that can affect tissue function include peptides, such as peptides that are involved in tissue repair processes and belonging to the EGF, FGF, PDGF, TGF-β, VEGF, PD-ECGF or IGF families, and also peptides derived from bone
morphogenetic protein 2, or BMP-2. The bioactive agent can also be a cell, such as a platelet. - In some aspects, the coating or article can include a radiopacifying agent.
- In preparing the coatings or articles, a plurality of natural biodegradable polysaccharides are crosslinked to each other via coupling groups that are pendent from the natural biodegradable polysaccharide (i.e., one or more coupling groups are chemically bonded to the polysaccharide). In some aspects, the coupling group on the natural biodegradable polysaccharide is a polymerizable group. In a free radical polymerization reaction the polymerizable group can crosslink natural biodegradable polysaccharides together in the composition, thereby forming a natural biodegradable polysaccharide matrix, which can be a portion of a coating, an in-vivo formed matrix, or the body member of a medical implant.
- The natural biodegradable polysaccharides described herein are non-synthetic polysaccharides that can be associated with each other to form a matrix, which can be used as a coating or as an article, for example, a medical implant or an in-vivo formed matrix. The natural biodegradable polysaccharides can also be enzymatically degraded, but offer the advantage of being generally non-enzymatically hydrolytically stable. This is particularly advantageous for bioactive agent delivery, as in some aspects the invention provides coatings or articles capable of releasing the bioactive agent under conditions of enzyme-mediated degradation, but not by diffusion. Therefore, the kinetics of bioactive agent release from the coatings or articles of the invention are fundamentally different than those of coatings prepared from synthetic biodegradable materials, such as poly(lactides).
- Natural biodegradable polysaccharides include polysaccharide and/or polysaccharide derivatives that are obtained from natural sources, such as plants or animals. Exemplary natural biodegradable polysaccharides include amylose, maltodextrin, amylopectin, starch, dextran, hyaluronic acid, heparin, chondroitin sulfate, dermatan sulfate, heparan sulfate, keratan sulfate, dextran sulfate, pentosan polysulfate, and chitosan. Preferred polysaccharides are low molecular weight polymers that have little or no branching, such as those that are derived from and/or found in starch preparations, for example, amylose and maltodextrin.
- Because of the particular utility of the amylose and maltodextrin polymers, in some aspects natural biodegradable polysaccharides are used that have an average molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, or 50,000 Da or less. In some aspects the natural biodegradable polysaccharides have an average molecular weight of 500 Da or greater. In some aspects the natural biodegradable polysaccharides have an average molecular weight in the range of about 1000 Da to about 10,000 Da. Natural biodegradable polysaccharides of particular molecular weights can be obtained commercially or can be prepared, for example, by acid hydrolysis and/or enzymatic degradation of a natural biodegradable polysaccharide preparation, such as starch. The decision of using natural biodegradable polysaccharides of a particular size range may depend on factors such as the physical characteristics of the coating composition (e.g., viscosity), the desired rate of degradation of the coating, the presence of other optional moieties in the coating composition (for example, bioactive agents, etc.), etc.
- The natural biodegradable polysaccharides that are used in accordance with the methods and compositions of the invention are readily available at a low cost and/or can be prepared easily using established techniques. This allows for a cost effective method of coating and fabricating medical articles.
- The use of natural biodegradable polysaccharides, such as maltodextrin or amylose, provides many advantages when used in a coating composition applied to the surface of a medical device, or for the formation of an article, such as one that can be used in vivo. Degradation of a natural biodegradable polysaccharide-containing article, or coating from the surface of a medical device, can result in the release of, for example, naturally occurring mono- or disaccharides, such as glucose, which are common serum components. Furthermore, the use of natural biodegradable polysaccharides that degrade into common serum components, such as glucose, can be viewed as more acceptable than the use of synthetic biodegradable polysaccharides that degrade into non-natural compounds, or compounds that are found at very low concentrations in the body.
- In some aspects of the invention, this advantageous feature is reflected in the use of natural biodegradable polysaccharides which are non-animal derived, such as amylose and maltodextrin, and that degrade into products that present little or no immunogenic or toxic risk to the individual. The invention provides improved, cost-efficient, natural biodegradable polysaccharide compositions for articles or coatings that can be used in a variety of medical treatments.
- Another advantage of the invention is that the natural biodegradable polysaccharide-based coatings are more resistant to hydrolytic degradation than other biodegradable polymers, such as poly(lactides). Degradation of the natural biodegradable polysaccharides of the invention are primarily enzyme-mediated, with minimal or no hydrolysis of the natural biodegradable polysaccharide occurring when a natural biodegradable polysaccharide-containing coating is prepared under ambient conditions. This allows the natural biodegradable polysaccharide-based coatings to remain substantially stable (for example, resistant to degradation) prior to placing the coated-article in vivo. For example, a natural biodegradable polysaccharide coated article can be manipulated in a non-biological, aqueous-based-medium without risk that the coating will prematurely degrade due to non-enzyme-mediatated hydrolysis. Other coatings that are based on biodegradable polymers such as poly(lactide) or poly(lactide-co-glycolide) are subject to hydrolysis even at relatively neutral pH ranges (e.g., pH 6.5 to 7.5) and therefore do not offer this advantage.
- Therefore, the invention includes natural biodegradable polysaccharide-containing compositions, coatings, articles, and methods of preparing such that have the advantage of providing stability in the presence of an aqueous environment.
- In one aspect, the invention provides a shelf-stable composition for preparing a biodegradable coating, the shelf stable composition comprising a natural biodegradable polysaccharide comprising coupling groups. These compositions could be obtained or prepared, according to the details provided herein, and then stored for a period of time before the composition is used to form a biodegradable coating or article, without significant degradation of the natural biodegradable polysaccharide occurring during storage. Accordingly, the invention also provides methods for preparing a biodegradable coating comprising preparing a biodegradable coating composition comprising a natural biodegradable polysaccharide comprising coupling group; storing the coating composition for an amount of time; and then using the coating composition to prepare a biodegradable coating or a biodegradable article. In some aspects, the biodegradable article is formed in situ, for example, by promoting the polymerization of the natural biodegradable polysaccharide within the body. Optionally, one or more bioactive agents and/or microparticles can be added before or after storage of the coating composition.
- In a related aspect, the invention also provides the advantage of being able to perform methods wherein the natural biodegradable polysaccharide is subject to exposure to an aqueous solution without risking significant degradation of the natural biodegradable polysaccharide. For example, the natural biodegradable polysaccharide may be contacted with an aqueous solution in a synthetic or post-synthetic step, including addition synthesis reactions and purification steps, or a coating that includes the natural biodegradable polysaccharide can be contacted with an aqueous solution in, for example, a sterilization step or a step that involves incorporation of a bioactive agent into the biodegradable coating.
- In yet another aspect, the invention relates to the stability of an article or a coating that is formed on an article. The invention provides a method comprising obtaining an article formed from, or having a coating comprising, a natural biodegradable polysaccharide, and then contacting the article with an aqueous solution for a period of time wherein the article or coating remains predominantly stable in the solution. The aqueous solution can be, for example, a storage solution, a solution that is used to hydrate the surface of the coated device, or an aqueous sterilization solution.
- Degradation of the natural biodegradable polysaccharide-containing coating or article may commence when placed in contact with a body fluid, which may include natural biodegradable polysaccharide-degrading enzymes, such as carbohydrases.
- The invention also provides a useful way to deliver larger hydrophilic bioactive agents, such as polypeptides, nucleic acids, and polysaccharides, as well as viral particles and cells from the biodegradable article, or biodegradable coating on a surface, such as a medical device or a coating on a surface thereof. Comparatively, the use of non-degrading drug delivery matrices may not be useful for the delivery of these larger bioactive agents if they are too large to diffuse out of the matrix. However, according to some aspects of the invention, an article or a coating that includes a matrix of the natural biodegradable polysaccharide having a bioactive agent can be placed or formed in the body, and as the matrix degrades the bioactive agent is gradually released from the matrix. In one aspect of the invention, the bioactive agent has a molecular weight of about 10,000 Da or greater.
- In some aspects, the invention provides a drug-releasing biodegradable article, coating, or composition comprising (i) a natural biodegradable polysaccharide, preferably selected from amylose and maltodextrin, comprising an ethylenically unsaturated group, (ii) an initiator, and (iii) a bioactive agent selected from the group of polypeptides, polynucleotides, and polysaccharides.
- In another aspect, a coated surface is prepared on a medical device, such as a stent or catheter. The methods include disposing in one or more steps the following reagents on a surface: (a) an initiator, (b) a natural biodegradable polysaccharide, preferably selected from amylose and maltodextrin, comprising an ethylenically unsaturated group, and (c) a bioactive agent. After the components have been disposed on the surface, the initiator is activated to crosslink a plurality of natural biodegradable polysaccharides comprising ethylenically unsaturated groups that are present in the composition, thereby forming a coating on the surface that includes the bioactive agent.
- Depending on the application, the initiator can be first disposed on the surface, followed by disposing the natural biodegradable polysaccharide and bioactive agent on the layer of initiator. Alternatively, the initiator, natural biodegradable polysaccharide, and bioactive agent are mixed and disposed together on the surface.
- Therefore, in some aspects, the invention provides a method for delivery of a bioactive agent, or more than one bioactive agent, to a subject. The method comprises the steps of providing a coated article to a subject, the coated article having a biodegradable coating which comprises a plurality of natural biodegradable polysaccharides associated via coupling groups, and bioactive agent. The coated article is then exposed to a carbohydrase to promote the degradation of the coating and release of the bioactive agent. For example, a biodegradable coating or article including amylose and/or maltodextrin polymers can be exposed to an α-amylase to promote degradation of the coating and release of the bioactive agent. The step of exposing can be performed by placing the biodegradable coating or article in a patient. In the absence of the carbohydrase there is substantially no release of the bioactive agent. In some aspects the bioactive agent comprises a polypeptide, such as an antibody or an antibody fragment.
- In some aspects, the methods of the invention can be used to prepare coatings wherein an amount of bioactive agent in the range of 1% to 17% of the total amount of bioactive agent present in the coating is released from the coating within a period of 2 days, and coatings wherein an amount of bioactive agent in the range of 1% to 20% of the total amount of bioactive agent present in the coating is released from the coating within a period of 8 days.
- In other aspects, the bioactive agent is delivered from a medical implant having a biodegradable body member which comprises a plurality of natural biodegradable polysaccharide associated via pendent coupling groups, the body member also including a bioactive agent. The medical implant is then exposed to a carbohydrase to promote the degradation of the implant and release of the bioactive agent.
- In some aspects, the methods of the invention can be used to prepare medical implants wherein an amount of bioactive agent in the range of 1% to 17% of the total amount of bioactive agent present in the medical implant is released within a period of 8 days, medical implants wherein an amount of bioactive agent in the range of 1% to 41% of the total amount of bioactive agent present in the medical implant is released within a period of 14 days, and medical implants wherein an amount of bioactive agent in the range of 1% to 60% of the total amount of bioactive agent present in the medical implant is released within a period of 21 days.
- Alternatively a carbohydrase can be administered to a subject, or the carbohydrase can be provided to a portion of the article, wherein the carbohydrase is released from the portion and locally causes the degradation of the coating.
- The coatings can also have favorable bioactive agent-releasing properties when the coated article has been placed in the body. In this regard, the present invention provides an overall improvement in terms of providing coatings for implantable medical articles. Articles that are fabricated from the biodegradable polysaccharides can have many of the same beneficial surface characteristics as provided by the biodegradable polysaccharide coatings.
- In another aspect of the invention, the natural biodegradable polysaccharide is modified with a hydrophobic moiety in order to provide a biodegradable matrix having hydrophobic properties. Therefore, a biodegradable coating or article can be formed from natural biodegradable polysaccharide comprising one or more pendent coupling groups and one or more pendent hydrophobic moieties. Exemplary hydrophobic moieties include fatty acids and derivatives thereof, and C2-C18 alkyl chains.
- Therefore, in some aspects of the invention, modification of the natural biodegradable polysaccharide allows for preparation of coatings or articles that are biodegradable and that can release a hydrophobic bioactive agent.
- In other aspects, the hydrophobic moiety pendent from the natural biodegradable has properties of a bioactive agent. Upon degradation of the matrix, the hydrophobic moiety can be hydrolyzed from the natural biodegradable polymer and released to provide a therapeutic effect. One example of a therapeutically useful hydrophobic moiety is butyric acid.
- In yet another aspect, the invention provides methods and articles for improving the stability of a bioactive agent that is delivered from a coating or an article by utilizing a natural biodegradable non-reducing polysaccharide. The non-reducing polysaccharide can provide an inert matrix and thereby improve the stability of sensitive bioactive agents, such as proteins and enzymes. The article or coating can include a matrix having a plurality of natural biodegradable non-reducing polysaccharides along with a bioactive agent, such as a polypeptide. An exemplary non-reducing polysaccharide comprises polyalditol. Biodegradable non-reducing polysaccharides can very useful for formulating coatings or articles that release the bioactive agent over a prolonged period of time.
- While it is desirable to make coatings or articles that provide desired properties (for example, bioactive agent release, wettability, etc.), their actual preparation can be challenging. In particular, the use of some polysaccharides for preparing coatings or articles may result in products that are unsuitable for use. For example, some polysaccharide-based coatings, including those made from starch-based materials, have the potential to be overly brittle and inflexible. While these properties may be suitable for pharmaceutical capsules or tablets they are generally undesirable as properties for coatings or articles, such as bioactive agent releasing or sealant coatings, or medical implants.
- Despite this, the present invention demonstrates the preparation of articles and coatings that include natural biodegradable polysaccharides that are suitable for in vivo use. These products display excellent physical characteristics and are suitable for use in applications wherein a particular function, such as bioactive agent delivery or a sealant function is desired. For example, coatings or articles can be prepared having viscoelastic properties. In one aspect of the invention, the coating or article has an elastic modulus value in the range of 27 kPa to 30 kPa.
- The coatings of the present invention can have desirable surface properties that include elasticity and wettability, in addition to being biodegradable. Also, it has surprisingly been discovered that the coatings demonstrate excellent lubricity, which can provide distinct advantages for short term and single use devices. Therefore, in one aspect, the invention presents a method for providing lubricity to an article surface comprising the steps of disposing a composition comprising a plurality of natural biodegradable polysaccharides comprising pendent coupling groups and activating the coupling groups to promote association of the plurality of natural biodegradable polysaccharides and formation of a lubricious coating on the article surface.
- The coatings can be formed on the surfaces of medical articles, including those designed for single use or for short-term use. For example, a lubricious coating can be formed on a catheter.
- Coatings including natural biodegradable polysaccharides can be prepared to provide a lubricity of 20 g or less, and can also be prepared to provide a lubricity of 15 g or less, or 10 g or less, as based on friction testing. The coatings were also shown to be highly durable, as lubricity was maintained during multiple cycles of friction testing. Methods utilizing a photoinitiator have been shown to provide coatings with both excellent lubricity and durability.
- In some embodiments of the invention, the methods of preparing the compositions for fabrication of articles and/or coated surfaces do not require the use of organic solvents. The use of organic solvents can be physically hazardous. Use of organic solvents can potentially destroy the activity of a bioactive agent that can be optionally included in the natural biodegradable polysaccharide-based composition.
- Many of the advantageous features of the present natural biodegradable polysaccharide-containing coatings and articles are thought to be provided by the starting materials, in particular the natural biodegradable polysaccharides having pendent coupling groups. In some aspects the natural biodegradable polysaccharides have pendent polymerizable groups, such as ethylenically unsaturated groups. In a preferred aspect, the degradable polymerizable polymers (macromers) are formed by reacting a natural biodegradable polysaccharide with a compound comprising an ethylenically unsaturated group. For example, in some cases, a natural biodegradable polysaccharide is reacted with a compound including an ethylenically unsaturated group and an isocyanate group. In another example of synthesis, a natural biodegradable polysaccharide is treated with an oxidizing agent to form a reactive aldehyde species on the polysaccharide and then reacted with a compound comprising an ethylenically unsaturated group and an amine group. Polysaccharide macromers were shown to have excellent matrix forming capabilities.
- Synthesis can be carried out to provide the natural biodegradable polysaccharide with a desired quantity of pendent coupling groups. It has been found that use of a natural biodegradable polysaccharide having a predetermined amount of the coupling groups allows for the formation of a coating or an article having desirable physical characteristics (for example, the coatings are not brittle). Therefore, in some aspects, the invention provides natural biodegradable polysaccharides having an amount of pendent coupling groups of about 0.7 μmoles of coupling group per milligram of natural biodegradable polysaccharide. Preferably the amount of coupling group per natural biodegradable polysaccharide is in the range of about 0.3 μmoles/mg to about 0.7 μmoles/mg. For example, amylose or maltodextrin can be subject to a synthesis reaction with a compound having an ethylenically unsaturated group to provide an amylose or maltodextrin macromer having a ethylenically unsaturated group load level in the range of about 0.3 μmoles/mg to about 0.7 μmoles/mg.
- In some aspects of the invention an initiator is used to promote the formation of the natural biodegradable polysaccharide matrix for article or coating formation. The initiator can be an independent compound or a pendent chemical group used to activate the coupling group pendent from the natural biodegradable polymer and promote coupling of a plurality of natural biodegradable polymers. When the coupling group pendent from the natural biodegradable polysaccharide is a polymerizable group, the initiator can be used in a free radical polymerization reaction to promote crosslinking of the natural biodegradable polysaccharides together in the composition.
- Therefore, in one aspect, the invention provides a biodegradable coating or article composition comprising (i) a natural biodegradable polysaccharide, preferably selected from amylose and maltodextrin, comprising a coupling group, (ii) an initiator, and (iii) a bioactive agent, wherein the coupling group is able to be activated by the initiator and promote crosslinking of a plurality of natural biodegradable polysaccharides. In some aspects of the invention the initiator is independent of the natural biodegradable polysaccharide and in other aspects the initiator is pendent from the natural biodegradable polysaccharide. Preferably, the natural biodegradable polysaccharide comprises an ethylenically unsaturated group. In some aspects a photoinitiator is used, such as a photoinitiator that is activated by light wavelengths having no or a minimal effect on the bioactive agent present in the composition.
- In another aspect, the initiator includes an oxidizing agent/reducing agent pair, a “redox pair,” to drive polymerization of the biodegradable polysaccharide. In preparing the biodegradable coating or article the oxidizing agent and reducing agent are combined in the presence of the biodegradable polysaccharide. One benefit of using a redox pair is that, when combined, the oxidizing agent and reducing agent can provide a particularly robust initiation system. This is advantageous as it can promote the formation of a matrix, for example, useful for coating or article preparation, from biodegradable polysaccharide compositions having a relatively low viscosity. This can be particularly useful in many applications, especially when the biodegradable polysaccharide composition is used for the formation of an in situ polymerized article. For example, a low viscosity composition can be passed through a small gauge delivery conduit with relative ease to provide the composition that can polymerize in situ.
- In some aspects of the invention, the viscosity of the composition is above about 5 centi Poise (cP), or about 10 cP or greater. In other aspects of the invention the viscosity of the composition is between about 5 cP or 10 cP and about 700 cP, and in some aspects between about 5 cP or 10 cP and about 250 cP. In some aspects the viscosity of the composition is above about 5 cP or 10 cP and the biodegradable polysaccharides in the composition have an average molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, or 50,000 Da or less.
- A method for preparing a coating or article can include the steps of (a) providing a first composition that includes a natural biodegradable polysaccharide comprising a coupling group and a first member of a redox pair (for example, the oxidizing agent) and then (b) mixing the first composition with second composition that includes a second member of the redox pair (for example, the reducing agent). In some aspects the second composition includes a natural biodegradable polysaccharide. For example, the first composition can include (a) a natural biodegradable polysaccharide having a coupling group and an oxidizing agent and the second composition can include a (b) natural biodegradable polysaccharide having a coupling group and a reducing agent. In some aspects, when the first composition is combined with the second composition, the final composition can be about 5 cP or greater.
- The oxidizing agent can be selected from inorganic or organic oxidizing agents, including enzymes; the reducing agent can be selected from inorganic or organic reducing agents, including enzymes. Exemplary oxidizing agents include peroxides, including hydrogen peroxide, metal oxides, and oxidases, such as glucose oxidase. Exemplary reducing agents include salts and derivatives of electropositive elemental metals such as Li, Na, Mg, Fe, Zn, Al, and reductases. In one aspect, the reducing agent is present in the composition at a concentration of 2.5 mM or greater when mixed with the oxidizing agent. Other reagents, such as metal or ammonium salts of persulfate, can be present in the composition to promote polymerization of the biodegradable polysaccharide.
- A coating or article formed using redox polymerization can therefore comprise a plurality of natural biodegradable polysaccharides associated via polymerized groups, a reduced oxidizing agent, and an oxidized reducing agent. In one preferred aspect, the biodegradable coated layer is formed article having a first coated layer comprising a synthetic polymer.
- The invention also provides alternative methods for preparing a coated surface or an article that is biodegradable and that can release a bioactive agent. For example, an alternative method for forming a coating includes disposing in two or more steps at least the following reagents on a surface: (a) a natural biodegradable polysaccharide comprising a first coupling group, (b) a natural biodegradable polysaccharide comprising a second coupling group that is reactive with the first coupling group, and (c) a bioactive agent. According to this method reagents (a) and (b) are reactive with each other and are disposed separately on the surface but can individually include reagent (c). For example, reagent (a) is first disposed on the surface and then a mixture comprising reagent (b) and (c) is then disposed on reagent (a). Reagent (a) reacts with (b) to link the natural biodegradable polysaccharides together to form a coating that includes reagent (c), the bioactive agent. An article can be formed in a similar manner, for example, by a method that includes combining (a) a natural biodegradable polysaccharide comprising a first coupling group with (b) a natural biodegradable polysaccharide comprising a second coupling group that is reactive with the first coupling group, and (c) a bioactive agent. The article can be partially or fully formed when reagent (a) reacts with (b) to link the natural biodegradable polysaccharides together to form the article, which includes reagent (c), the bioactive agent.
- In some aspects, the present invention employs the use of biodegradable microparticles that include a bioactive agent and a natural biodegradable polysaccharide, such as amylose and maltodextrin that have pendent coupling groups. The microparticles are used in association with the natural biodegradable polysaccharides to prepare a biodegradable, bioactive agent-releasing coating for the surface of medical devices.
- According to this aspect of the invention, a medical device having a coating that includes a crosslinked matrix of natural biodegradable polysaccharides and biodegradable microparticles having a bioactive agent can be placed in the body, and as the biodegradable microparticles degrade the bioactive agent is gradually released from the coating.
- The natural biodegradable polysaccharide matrix provides the ability to associate the biodegradable microparticles with the surface of the coated device. In some arrangements, the biodegradable microparticles are dispersed in the natural biodegradable polysaccharide matrix. Such coatings can be formed by disposing a mixture of (a) biodegradable microparticles having a bioactive agent and (b) natural biodegradable polysaccharides having pendent coupling groups, disposing the mixture on a surface, and then treating the composition to form a coated layer wherein the biodegradable microparticles are dispersed within the matrix.
- In other arrangements, the coating is formed by disposing the biodegradable microparticles independently of the natural biodegradable polysaccharide having pendent coupling groups. In these arrangements the biodegradable microparticles can be present predominantly one face of the layer that is formed from the natural biodegradable polysaccharide and a microparticle-matrix interface can be formed.
- The methods include disposing in one or more steps the following components on a surface: (a) an initiator, (b) a natural biodegradable polysaccharide, preferably selected from amylose and maltodextrin, comprising a coupling group, and (c) biodegradable microparticles comprising a bioactive agent. After the components have been disposed on the surface, the initiator is activated to couple a plurality of natural biodegradable polysaccharide polymers that are present in the composition, thereby forming a natural biodegradable polysaccharide matrix on the surface that is associated with the biodegradable microparticles having the bioactive agent.
- In these aspects, the method includes the steps of (i) disposing a composition comprising (a) a natural biodegradable polysaccharide having a coupling group, (b) an initiator, and (c) biodegradable microparticles comprising a bioactive agent on a surface; and (ii) activating the initiator to provide a coated composition having the natural biodegradable polysaccharide and the biodegradable microparticles having the bioactive agent on the surface. Alternatively, the initiator can be disposed independently of the natural biodegradable polysaccharide.
- By including microparticles having a bioactive agent in the natural biodegradable polysaccharide-containing coating, the invention also provides a way to effectively and efficiently prepare a variety of drug-delivery coatings. The use of microparticles offers the ability to easily prepare coatings having one or more bioactive agents present in desired amounts in the coating. Such coatings can be prepared by obtaining biodegradable microparticles that have a bioactive agent and then forming a coating that includes the microspheres associated with the natural biodegradable polysaccharide matrix. In some aspects, different microparticles having different bioactive agents can be included in the coating in desired amounts to provide a bioactive agent-releasing coating that is able to release a desired combination of bioactive agents in desired amounts. This is a particular advantage when using bioactive agents that are typically not compatible in the same composition (for example, bioactive agents that have different physical properties).
- Microparticles can also be included in articles formed from the natural biodegradable polysaccharide. For example, microparticles can be included in an implantable medical article formed from the natural biodegradable polysaccharides of the invention, or can be included in an article that is formed in situ. In these aspects, the presence of biodegradable microparticles in articles can offer many of the advantages that are offered by the presence of the microparticles in the coatings.
- In another aspect, the present invention provides compositions and methods for preparing sealant materials that are particularly useful in connection with implantable medical articles having a porous surface, such as grafts, patches, and wound dressings. In preferred aspects, the inventive compositions can be used to prepare a sealant coating for implantable medical articles, particularly implantable medical articles that include a porous surface.
- The sealant coating can provide a barrier to the movement of body fluids, such as blood, near the surface of the coated article. For example, the natural biodegradable polysaccharide-based sealant coating can provide hemostasis at the article surface by formation of a tight seal. Gradually, the natural biodegradable polysaccharide in the sealant coating degrades and a tissue layer is formed as the sealant coating is replaced by cells and other factors involved in tissue repair. During the process of degradation, natural biodegradable polysaccharide degradation products, such as naturally occurring mono- or disaccharides, for example, glucose, are released from the sealant coating, which can be considered an ideal in vivo degradation product because it is commonly found in the body and may also be utilized by the cells involved in tissue repair during the degradation/infiltration process. Gradually, infiltrated tissue growth replaces the function of the natural biodegradable polysaccharide-containing sealant coating.
- Another particular advantage of the invention is that release of glucose reduces the likelihood that the process of natural biodegradable polysaccharide degradation and tissue infiltration will promote a strong inflammatory response. This is because the natural biodegradable polysaccharide-based sealant coating can degrade into materials that are non-antigenic or that have low antigenicity. Another advantage is that the degradation products are free of other materials that may cause disease, such as microbial, viral, or prionic materials potentially present in animal-derived preparations (such as bovine collagen preparations).
- The sealant compositions of the invention, which include natural biodegradable polysaccharides, such as amylose or maltodextrin polymers, that can be coupled together to form a matrix (at least a portion of the sealant coating) on the medical article, can include a bioactive agent, which can be released as the sealant coating degrades.
- In some aspects, the invention provides a biodegradable sealant composition comprising (i) a natural biodegradable polysaccharide comprising a coupling group, and (ii) an initiator, wherein the coupling group is able to be activated by the initiator and promote coupling of a plurality of natural biodegradable polysaccharides. Preferably the natural biodegradable polysaccharide is a polymer such as amylose or maltodextrin. In some aspects the sealant composition can also include a bioactive agent. The initiator can be independent of the natural biodegradable polysaccharide, pendent from the natural biodegradable polysaccharide polymer, or both pendent and independent of the natural biodegradable polysaccharide polymer.
- Accordingly, the invention also provides methods for preparing a surface having a sealant coating. The sealant coated surface is prepared on a medical article or article having a porous surface. The methods include disposing in one or more steps the following reagents on a surface: (a) an initiator, and (b) a natural biodegradable polysaccharide comprising a coupling group. In some aspects a bioactive agent is also disposed on the surface. In one preferred aspect, the bioactive agent is a prothrombotic or procoagulant factor. In these aspects, after the components have been disposed on the surface, the initiator is activated to couple the natural biodegradable polysaccharides that are present in the composition, thereby forming a natural biodegradable polysaccharide coating on the surface that includes the bioactive agent.
- During the step of activating, the natural biodegradable polysaccharide is contacted with the initiator and the initiator is activated to promote the coupling of two or more natural biodegradable polysaccharides via their coupling groups. In preferred aspects, the natural biodegradable polysaccharide includes a polymerizable group, such as an ethylenically unsaturated group, and initiator is capable of initiating free radical polymerization of the polymerizable groups.
- The invention also provides alternative methods for preparing a sealant coating on the surface of an article. The methods include disposing at least the following reagents on a surface: (a) a natural biodegradable polysaccharide comprising a first coupling group and (b) a natural biodegradable polysaccharide comprising a second coupling group, where in the second coupling group is reactive with the first coupling group. According to this method, reagents (a) and (b) are reactive with each other to couple the natural biodegradable polysaccharide, or (a) and/or (b) can be treated to be made reactive with each other. In some aspects (a) and (b) are disposed separately on the surface to form the sealant coating. The natural biodegradable polysaccharide can be the same types of polymers of different types of polymers.
- The first coupling group and second coupling group can be a pair of chemical groups that are reactive with one another, preferably specifically reactive. The groups can also become reactive with each other upon addition of a particular agent to the mixture of natural biodegradable polysaccharide having different reactive groups.
-
FIG. 1 is a graph of cumulative BSA release from maltodextrin-acrylate filaments treated with amylase, over a period of time. -
FIG. 2 is a graph of cumulative BSA release from maltodextrin-acrylate/photo-PVP-coated PEBAX rod treated with amylase, over a period of time. -
FIG. 3 is a graph of cumulative absorbance values of active and total IgG Fab fragment release from maltodextrin-acrylate filaments treated with amylase, over a period of time. -
FIG. 4 is a graph of cumulative absorbance values of active and total IgG release from maltodextrin-acrylate(redox)/photo-PVP-coated stainless steel rods treated with amylase, over a period of time. -
FIG. 5 is a graph of cumulative absorbance values of active and total IgG release from maltodextrin-acrylate(photoinitiation)/photo-PVP-coated stainless steel rods treated with amylase and percent degradation of the maltodextrin-acrylate coating, over a period of time. -
FIG. 6 is a graph of cumulative absorbance values of active and total IgG release from a maltodextrin-acrylate filament treated with amylase and percent degradation of the filament, over a period of time. -
FIG. 7 is a graph of modulus of a maltodextrin-acrylate matrix formed via REDOX polymerization, over a period of time. -
FIG. 8 is a graph of repetitive force testing of maltodextrin-acrylate coated PEBAX rods versus synthetic polymer-coated PEBAX rods. - The embodiments of the present invention described herein are not intended to be exhaustive or to limit the invention to the precise forms disclosed in the following detailed description. Rather, the embodiments are chosen and described so that others skilled in the art can appreciate and understand the principles and practices of the present invention.
- All publications and patents mentioned herein are hereby incorporated by reference. The publications and patents disclosed herein are provided solely for their disclosure. Nothing herein is to be construed as an admission that the inventors are not entitled to antedate any publication and/or patent, including any publication and/or patent cited herein.
- In one aspect, the invention provides methods of preparing biodegradable coatings that release bioactive agents from the surface of medical devices. The compositions and methods of the present invention are particularly useful for coating surfaces of implantable medical devices, such as stents and catheters, and that are capable of releasing drugs from the device.
- In another aspect, the invention provides methods of preparing biodegradable articles, such as medical implants or in vivo formed matrices. The biodegradable articles can also be used for the release of bioactive agents, and in this manner can function as bioactive agent-releasing implants or depots. In some aspects, the biodegradable articles of the invention biodegrade within a period that is acceptable for the desired application.
- In some aspects, the biodegradable article is a medical implant that provides mechanical properties at the implantation site and maintains these mechanical properties until they are no longer needed. After this period of time has elapsed, the medical implant is degraded to an extent that the properties are no longer provided by the medical implant, and the biodegradable components can be absorbed and/or excreted by the body. In some embodiments, the medical implant slowly degrades and transfers stress at the appropriate rate to surrounding tissues as these tissues heal and can accommodate the stress once borne by the medical device.
- The biodegradable coating or article includes a natural biodegradable polysaccharide having a coupling group. Exemplary natural biodegradable polysaccharides include amylose and maltodextrin. In some aspects, the present invention provides biodegradable coatings having excellent surface characteristics and that can provide a suitable vehicle for the delivery of bioactive agents. These biodegradable coatings can be disposed on medical devices having a variety of biomaterial surfaces.
- In some embodiments of the invention, a coating is formed on a device that includes a biodegradable matrix and biodegradable microparticles, the biodegradable microparticles including one or more bioactive agents. The biodegradable material used to form the matrix includes a natural biodegradable polysaccharide as a component. In the matrix, natural biodegradable polysaccharides such as amylose and maltodextrin are coupled to each other and the biodegradable microparticles are associated with the matrix.
- In yet other embodiments of the invention, a sealant coating is formed on a device. The sealant coating includes a biodegradable matrix and optionally one or more bioactive agents, such as prothrombotic agents.
- The sealant coating of the invention can, at least initially, provide a barrier on the porous surface that is not permeable to fluids within the body. Gradually, the sealant coating degrades and its function is replaced by tissue that infiltrates the porous surface. Therefore, the sealant coating has particular properties, such as biodegradability and relative impermeability (i.e., relative to the degradation of the sealant coating). The sealant coating can also be compliant and/or conformal, and can have properties such as flexibility, elasticity, and bendability.
- As used herein, impermeable, used in relation to the function of the sealant coating, refers to a significant reduction in the transmission of bulk liquid or fluids through the substrate which the sealant coating is associated with. For example, the sealant coating can be impermeable to the transmission of blood. The impermeability can be maintained as the natural biodegradable polysaccharide-based sealant coating degrades, and is replaced by tissue.
- As referred to herein, a “natural biodegradable polysaccharide” refers to a non-synthetic polysaccharide that is capable of being enzymatically degraded but that is generally non-enzymatically hydrolytically stable. Natural biodegradable polysaccharides include polysaccharide and/or polysaccharide derivatives that are obtained from natural sources, such as plants or animals. Natural biodegradable polysaccharides include any polysaccharide that has been processed or modified from a natural biodegradable polysaccharide (for example, maltodextrin is a natural biodegradable polysaccharide that is processed from starch). Exemplary natural biodegradable polysaccharides include hyaluronic acid, starch, dextran, heparin, chondroitin sulfate, dermatan sulfate, heparan sulfate, keratan sulfate, dextran sulfate, pentosan polysulfate, and chitosan. Preferred polysaccharides are low molecular weight polymers that have little or no branching, such as those that are derived from and/or found in starch preparations, for example, amylose and maltodextrin. Therefore, the natural biodegradable polysaccharide can be a substantially non-branched or non-branched poly(glucopyranose) polymer.
- Because of the particular utility of the amylose and maltodextrin polymers, it is preferred that natural biodegradable polysaccharides having an average molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, or 50,000 Da or less. It is also preferred that the natural biodegradable polysaccharides have an average molecular weight of 500 Da or greater. A particularly preferred size range for the natural biodegradable polysaccharides is in the range of about 1000 Da to about 10,000 Da. Natural biodegradable polysaccharides of particular molecular weights can be obtained commercially or can be prepared. The decision of using natural biodegradable polysaccharides of a particular size range may depend on factors such as the physical characteristics of the coating composition (e.g., viscosity), the desired rate of degradation of the coating, the presence of other optional moieties in the coating composition, for example, bioactive agents, etc.
- As used herein, “amylose” or “amylose polymer” refers to a linear polymer having repeating glucopyranose units that are joined by α-1,4 linkages. Some amylose polymers can have a very small amount of branching via α-1,6 linkages (about less than 0.5% of the linkages) but still demonstrate the same physical properties as linear (unbranched) amylose polymers do. Generally amylose polymers derived from plant sources have molecular weights of about 1×106 Da or less. Amylopectin, comparatively, is a branched polymer having repeating glucopyranose units that are joined by α-1,4 linkages to form linear portions and the linear portions are linked together via α-1,6 linkages. The branch point linkages are generally greater than 1% of the total linkages and typically 4%-5% of the total linkages. Generally amylopectin derived from plant sources have molecular weights of 1×107 Da or greater.
- Amylose can be obtained from, or is present in, a variety of sources. Typically, amylose is obtained from non-animal sources, such as plant sources. In some aspects, a purified preparation of amylose is used as starting material for the preparation of the amylose polymer having coupling groups. In other aspects, as starting material, amylose can be used in a mixture that includes other polysaccharides.
- For example, in some aspects, starch preparations having a high amylose content, purified amylose, synthetically prepared amylose, or enriched amylose preparations can be used in the preparation of amylose having the coupling groups. In starch sources, amylose is typically present along with amylopectin, which is a branched polysaccharide. According to the invention, it is preferred to use coating compositions that include amylose, wherein the amylose is present in the composition in an amount greater than amylopectin, if present in the composition. For example, in some aspects, starch preparations having high amylose content, purified amylose, synthetically prepared amylose, or enriched amylose preparations can be used in the preparation of amylose polymer having the coupling groups. In some embodiments the composition includes a mixture of polysaccharides including amylose wherein the amylose content in the mixture of polysaccharides is 50% or greater, 60% or greater, 70% or greater, 80% or greater, or 85% or greater by weight. In other embodiments the composition includes a mixture of polysaccharides including amylose and amylopectin and wherein the amylopectin content in the mixture of polysaccharides is 30% or less, or 15% or less. In some cases it may be desirable to use non-retrograding starches, such as waxy starch, in the current invention. The amount of amylopectin present in a starch may also be reduced by treating the starch with amylopectinase, which cleaves α-1,6 linkages resulting in the debranching of amylopectin into amylose.
- In some cases a synthesis reaction can be carried out to prepare an amylose polymer having pendent coupling groups (for example, amylose with pendent ethylenically unsaturated groups) and steps may be performed before, during, and/or after the synthesis to enrich the amount of amylose, or purify the amylose.
- Amylose of a particular size, or a combination of particular sizes can be used. The choice of amylose in a particular size range may depend on the application, for example, the type of surface coated or the porosity of the surface. In some embodiments amylose having an average molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, 50,000 Da or less, preferably greater than 500 Da, or preferably in the range of about 1000 Da to about 10,000 Da is used. Amylose of particular molecular weights can be obtained commercially or can be prepared. For example, synthetic amyloses with average molecular masses of 70, 110, 320, and 1,000 kDa can be obtained from Nakano Vinegar Co., Ltd. (Aichi, Japan). The decision of using amylose of a particular size range may depend on factors such as the physical characteristics of the coating composition (e.g., viscosity), the desired rate of degradation of the coating, the presence of other optional moieties in the coating composition (for example, bioactive agents, etc.), etc.
- Maltodextrin is typically generated by hydrolyzing a starch slurry with heat-stable α-amylase at temperatures at 85-90° C. until the desired degree of hydrolysis is reached and then inactivating the α-amylase by a second heat treatment. The maltodextrin can be purified by filtration and then spray dried to a final product. Maltodextrins are typically characterized by their dextrose equivalent (DE) value, which is related to the degree of hydrolysis defined as: DE=MW dextrose/number-averaged MW starch hydrolysate×100.
- A starch preparation that has been totally hydrolyzed to dextrose (glucose) has a DE of 100, where as starch has a DE of about zero. A DE of greater than 0 but less than 100 characterizes the mean-average molecular weight of a starch hydrolysate, and maltodextrins are considered to have a DE of less than 20. Maltodextrins of various molecular weights, for example, in the range of about 500-5000 Da are commercially available (for example, from CarboMer, San Diego, Calif.).
- Another contemplated class of natural biodegradable polysaccharides is natural biodegradable non-reducing polysaccharides. A non-reducing polysaccharide can provide an inert matrix thereby improving the stability of sensitive bioactive agents, such as proteins and enzymes. A non-reducing polysaccharide refers to a polymer of non-reducing disaccharides (two monosaccharides linked through their anomeric centers) such as trehalose (α-D-glucopyranosyl α-D-glucopyranoside) and sucrose (β-D-fructofuranosyl α-D-glucopyranoside). An exemplary non-reducing polysaccharide comprises polyalditol which is available from GPC (Muscatine, Iowa). In another aspect, the polysaccharide is a glucopyranosyl polymer, such as a polymer that includes repeating (1→3)O-β-D-glucopyranosyl units.
- In some aspects, the coating compositions can include natural biodegradable polysaccharides that include chemical modifications other than the pendent coupling group. To exemplify this aspect, modified amylose having esterified hydroxyl groups can be prepared and used in sealant coating compositions in association with the methods of the invention. Other natural biodegradable polysaccharides having hydroxyl groups may be modified in the same manner. These types of modifications can change or improve the properties of the natural biodegradable polysaccharide making for a coating composition that is particularly suitable for a desired application. Many chemically modified amylose polymers, such as chemically modified starch, have at least been considered acceptable food additives.
- As used herein, “modified natural biodegradable polysaccharides” refers to chemical modifications to the natural biodegradable polysaccharide that are different than those provided by the coupling group or the initiator group. Modified amylose polymers having a coupling group (and/or initiator group) can be used in the compositions and methods of the invention.
- To exemplify this aspect, modified amylose is described. By chemically modifying the hydroxyl groups of the amylose, the physical properties of the amylose can be altered. The hydroxyl groups of amylose allow for extensive hydrogen bonding between amylose polymers in solution and can result in viscous solutions that are observed upon heating and then cooling amylose-containing compositions such as starch in solution (retrograding). The hydroxyl groups of amylose can be modified to reduce or eliminate hydrogen bonding between molecules thereby changing the physical properties of amylose in solution.
- Therefore, in some embodiments the natural biodegradable polysaccharides, such as amylose, can include one or more modifications to the hydroxyl groups wherein the modifications are different than those provided by coupling group. Modifications include esterification with acetic anhydride (and adipic acid), succinic anhydride, 1-octenylsuccinic anhydride, phosphoryl chloride, sodium trimetaphosphate, sodium tripolyphosphate, and sodium monophosphate; etherification with propylene oxide, acid modification with hydrochloric acid and sulfuric acids; and bleaching or oxidation with hydrogen peroxide, peracetic acid, potassium permanganate, and sodium hypochlorite.
- Examples of modified amylose polymers include carboxymethyl amylose, carboxyethyl amylose, ethyl amylose, methyl amylose, hydroxyethyl amylose, hydroxypropyl amylose, acetyl amylose, amino alkyl amylose, allyl amylose, and oxidized amylose. Other modified amylose polymers include succinate amylose and oxtenyl succinate amylose.
- In another aspect of the invention, the natural biodegradable polysaccharide is modified with a hydrophobic moiety in order to provide a biodegradable matrix having hydrophobic properties. Exemplary hydrophobic moieties include those previously listed, fatty acids and derivatives thereof, and C2-C18 alkyl chains. A polysaccharide, such as amylose or maltodextrin, can be modified with a compound having a hydrophobic moiety, such as a fatty acid anhydride. The hydroxyl group of a polysaccharide can also cause the ring opening of lactones to provide pendent open-chain hydroxy esters.
- In some aspects, the hydrophilic moiety pendent from the natural biodegradable has properties of a bioactive agent. The hydrophilic moiety can be hydrolyzed from the natural biodegradable polymer and released from the matrix to provide a therapeutic effect. One example of a therapeutically useful hydrophilic moiety is butyric acid, which has been shown to elicit tumor cell differentiation and apoptosis, and is thought to be useful for the treatment of cancer and other blood diseases. The hydrophilic moiety that provides a therapeutic effect can also be a natural compound (such as butyric acid). Therefore, degradation of the matrix having a coupled therapeutic agent can result in all natural degradation products.
- According to the invention, a natural biodegradable polysaccharide that includes a coupling group is used to form an article or a coating on the surface of a medical article. Other polysaccharides can also be present in the coating composition. For example, the two or more natural biodegradable polysaccharides are used to form an article or a coating on the surface of a medical article. Examples include amylose and one or more other natural biodegradable polysaccharide(s), and maltodextrin and one or more other natural biodegradable polysaccharide(s); in one aspect the composition includes a mixture of amylose and maltodextrin, optionally with another natural biodegradable polysaccharide.
- In one preferred embodiment, amylose or maltodextrin is the primary polysaccharide. In some embodiments, the composition includes a mixture of polysaccharides including amylose or maltodextrin and the amylose or maltodextrin content in the mixture of polysaccharides is 50% or greater, 60% or greater, 70% or greater, 80% or greater, or 85% or greater by weight.
- Purified or enriched amylose preparations can be obtained commercially or can be prepared using standard biochemical techniques such as chromatography. In some aspects, high-amylose cornstarch can be used.
- As used herein, “coupling group” can include (1) a chemical group that is able to form a reactive species that can react with the same or similar chemical group to form a bond that is able to couple the natural biodegradable polysaccharides together (for example, wherein the formation of a reactive species can be promoted by an initiator); or (2) a pair of two different chemical groups that are able to specifically react to form a bond that is able to couple the natural biodegradable polysaccharides together. The coupling group can be attached to any suitable natural biodegradable polysaccharide, including the amylose and maltodextrin polymers as exemplified herein.
- Contemplated reactive pairs include Reactive Group A and corresponding Reactive Group B as shown in the Table 1 below. For the preparation of a coating composition, a reactive group from group A can be selected and coupled to a first set of natural biodegradable polysaccharides and a corresponding reactive group B can be selected and coupled to a second set of natural biodegradable polysaccharides. Reactive groups A and B can represent first and second coupling groups, respectively. At least one and preferably two, or more than two reactive groups are coupled to an individual natural biodegradable polysaccharide polymer. The first and second sets of natural biodegradable polysaccharides can be combined and reacted, for example, thermochemically, if necessary, to promote the coupling of natural biodegradable polysaccharides and the formation of a natural biodegradable polysaccharide matrix.
TABLE 1 Reactive group A Reactive group B amine, hydroxyl, sulfhydryl N-oxysuccinimide (“NOS”) amine Aldehyde amine Isothiocyanate amine, sulfhydryl Bromoacetyl amine, sulfhydryl Chloroacetyl amine, sulfhydryl Iodoacetyl amine, hydroxyl Anhydride aldehyde Hydrazide amine, hydroxyl, carboxylic acid Isocyanate amine, sulfhydryl Maleimide sulfhydryl Vinylsulfone - Amine also includes hydrazide (R—NH—NH2)
- For example, a suitable coupling pair would be a natural biodegradable polysaccharide having an electrophilic group and a natural biodegradable polysaccharide having a nucleophilic group. An example of a suitable electrophilic-nucleophilic pair is N-hydroxysuccinimide-amine pair, respectively. Another suitable pair would be an oxirane-amine pair.
- In some aspects, the natural biodegradable polysaccharides of the invention include at least one, and more typically more than one, coupling group per natural biodegradable polysaccharide, allowing for a plurality of natural biodegradable polysaccharides to be coupled in linear and/or branched manner. In some preferred embodiments, the natural biodegradable polysaccharide includes two or more pendent coupling groups.
- In some aspects, the coupling group on the natural biodegradable polysaccharide is a polymerizable group. In a free radical polymerization reaction the polymerizable group can couple natural biodegradable polysaccharides together in the composition, thereby forming a biodegradable natural biodegradable polysaccharide matrix.
- A preferred polymerizable group is an ethylenically unsaturated group. Suitable ethylenically unsaturated groups include vinyl groups, acrylate groups, methacrylate groups, ethacrylate groups, 2-phenyl acrylate groups, acrylamide groups, methacrylamide groups, itaconate groups, and styrene groups. Combinations of different ethylenically unsaturated groups can be present on a natural biodegradable polysaccharide, such as amylose or maltodextrin.
- In preparing the natural biodegradable polysaccharide having pendent coupling groups any suitable synthesis procedure can be used. Suitable synthetic schemes typically involve reaction of, for example, hydroxyl groups on the natural biodegradable polysaccharide, such as amylose or maltodextrin. Synthetic procedures can be modified to produce a desired number of coupling groups pendent from the natural biodegradable polysaccharide backbone. For example, the hydroxyl groups can be reacted with a coupling group-containing compound or can be modified to be reactive with a coupling group-containing compound. The number and/or density of acrylate groups can be controlled using the present method, for example, by controlling the relative concentration of reactive moiety to saccharide group content.
- In some modes of practice, the biodegradable polysaccharides have an amount of pendent coupling groups of about 0.7 μmoles of coupling group per milligram of natural biodegradable polysaccharide. In a preferred aspect, the amount of coupling group per natural biodegradable polysaccharide is in the range of about 0.3 μmoles/mg to about 0.7 μmoles/mg. For example, amylose or maltodextrin can be reacted with an acrylate groups-containing compound to provide an amylose or maltodextrin macromer having a acrylate group load level in the range of about 0.3 μmoles/mg to about 0.7 μmoles/mg.
- As used herein, an “initiator” refers to a compound, or more than one compound, that is capable of promoting the formation of a reactive species from the coupling group. For example, the initiator can promote a free radical reaction of natural biodegradable polysaccharide having a coupling group. In one embodiment the initiator is a photoreactive group (photoinitiator) that is activated by radiation. In some embodiments, the initiator can be an “initiator polymer” that includes a polymer having a backbone and one or more initiator groups pendent from the backbone of the polymer.
- In some aspects the initiator is a compound that is light sensitive and that can be activated to promote the coupling of the amylose polymer via a free radical polymerization reaction. These types of initiators are referred to herein as “photoinitiators.” In some aspects it is preferred to use photoinitiators that are activated by light wavelengths that have no or a minimal effect on a bioactive agent if present in the composition. A photoinitiator can be present in a sealant composition independent of the amylose polymer or pendent from the amylose polymer.
- In some embodiments, photoinitiation occurs using groups that promote an intra- or intermolecular hydrogen abstraction reaction. This initiation system can be used without additional energy transfer acceptor molecules and utilizing nonspecific hydrogen abstraction, but is more commonly used with an energy transfer acceptor, typically a tertiary amine, which results in the formation of both aminoalkyl radicals and ketyl radicals. Examples of molecules exhibiting hydrogen abstraction reactivity and useful in a polymeric initiating system, include analogs of benzophenone, thioxanthone, and camphorquinone.
- In some preferred embodiments the photoinitiator includes one or more charged groups. The presence of charged groups can increase the solubility of the photoinitiator (which can contain photoreactive groups such as aryl ketones) in an aqueous system and therefore provide for an improved coating composition. Suitable charged groups include, for example, salts of organic acids, such as sulfonate, phosphonate, carboxylate, and the like, and onium groups, such as quaternary ammonium, sulfonium, phosphonium, protonated amine, and the like. According to this embodiment, a suitable photoinitiator can include, for example, one or more aryl ketone photogroups selected from acetophenone, benzophenone, anthraquinone, anthrone, anthrone-like heterocycles, and derivatives thereof; and one or more charged groups, for example, as described herein. Examples of these types of water-soluble photoinitiators have been described in U.S. Pat. No. 6,077,698.
- In some aspects the photoinitiator is a compound that is activated by long-wavelength ultraviolet (UV) and visible light wavelengths. For example, the initiator includes a photoreducible or photo-oxidizable dye. Photoreducible dyes can also be used in conjunction with a compound such as a tertiary amine. The tertiary amine intercepts the induced triplet producing the radical anion of the dye and the radical cation of the tertiary amine. Examples of molecules exhibiting photosensitization reactivity and useful as an initiator include acridine orange, camphorquinone, ethyl eosin, eosin Y, erythrosine, fluorescein, methylene green, methylene blue, phloxime, riboflavin, rose bengal, thionine, and xanthine dyes. Use of these types of photoinitiators can be particularly advantageous when a light-sensitive bioactive agent is included in the sealant coating.
- Therefore, in yet another aspect, the invention provides a coating composition comprising (i) a natural biodegradable polysaccharide comprising an ethylenically unsaturated group (ii) a photoinitiator selected from the group consisting of acridine orange, camphorquinone, ethyl eosin, eosin Y, erythrosine, fluorescein, methylene green, methylene blue, phloxime, riboflavin, rose bengal, thionine, and xanthine dyes, and (iii) a bioactive agent.
- Thermally reactive initiators can also be used to promote the polymerization ofnatural biodegradable polymers having pendent coupling groups. Examples of thermally reactive initiators include 4,4′azobis(4-cyanopentanoic acid), 2,2-azobis[2-(2-imidazolin-2-yl) propane]dihydrochloride, and analogs of benzoyl peroxide. Redox initiators can also be used to promote the polymerization of the natural biodegradable polymers having pendent coupling groups. In general, combinations of organic and inorganic oxidizers, and organic and inorganic reducing agents are used to generate radicals for polymerization. A description of redox initiation can be found in Principles of Polymerization, 2nd Edition, Odian G., John Wiley and Sons, pgs 201-204, (1981).
- In some cases, the initiator can be included in a base coating and the natural biodegradable polysaccharide or composition that includes the natural biodegradable polysaccharide can be disposed on the base coating. For example, a coated layer that includes the natural biodegradable polysaccharide can be formed on a coated layer that includes a synthetic polymer. The synthetic polymer can be a hydrophilic polymer such as poly(vinylpyrrolidone), poly(acrylamide), or copolymers thereof. In some aspects the synthetic polymer is formed using photoreactive groups, such as photoreactive groups that are pendent from the synthetic polymer, which can be used to covalently bond the synthetic polymer to a surface of the article.
- In some aspects the polymerization initiator is a polymer that includes an initiator group (herein referred to as an “initiator polymer”). The polymeric portion of the initiator polymer can be obtained or prepared to have particular properties or features that are desirable for use with acoating composition, such as a sealant coating composition. For example, the polymeric portion of the initiator polymer can have hydrophilic or amphoteric properties, it can include pendent charged groups, or it can have groups that allow it to interact with a particular surface (this can depend on the type of surface to be coated). Optionally, or additionally, the polymer can change or improve the properties of the coating that is formed by the amylose polymer having coupling groups. For example, the initiator polymer can change the elasticity, flexibility, wettability, or softness (or combinations thereof) of the coating formed on the surface. Certain polymers, as described herein, are useful as plasticizing agents for coatings that include natural biodegradable polysaccharides. Initiator groups can be added to these plasticizing polymers and used in the compositions and methods of the invention.
- For example, in some aspects an initiator can be pendent from a natural biodegradable polysaccharide. Therefore, the natural biodegradable polysaccharide is able to promote activation of polymerizable groups that are pendent from other natural biodegradable polysaccharides and promote the formation of a natural biodegradable polysaccharide matrix.
- In other cases, the polymeric portion of the initiator polymer can include, for example, acrylamide and methacrylamide monomeric units, or derivatives thereof. In some embodiments, the coating composition includes an initiator polymer having a photoreactive group and a polymeric portion selected from the group of acrylamide and methacrylamide polymers and copolymers.
- In some aspects, the initiator includes an oxidizing agent/reducing agent pair, a “redox pair,” to drive polymerization of the biodegradable polysaccharide. In this case, polymerization of the biodegradable polysaccharide is carried out upon combining one or more oxidizing agents with one or more reducing agents. Other compounds can be included in the composition to promote polymerization of the biodegradable polysaccharides.
- When combined, the oxidizing agent and reducing agent can provide a particularly robust initiation system and can drive the formation of a polymerized matrix of polysaccharides from a composition having a low viscosity. A polysaccharide composition with a low viscosity may be due to a low concentration of polysaccharide in the composition, a polysaccharide having a low average molecular weight, or combinations thereof. Matrix formation from a polysaccharide composition having a low viscosity is particularly advantageous in many applications, especially for in situ polymerization. In some aspects of the invention, a low viscosity polysaccharide composition is passed through a small gauge delivery conduit, such as a needle, wherein the redox pair causes the polymerization of the polysaccharides in situ.
- In some aspects of the invention, the viscosity of the composition is above about 5 cP, or about 10 cP or greater. In other aspects of the invention the viscosity of the composition is between about 5 cP or 10 cP and about 700 cP, or between about 5 cP or 10 cP and about 250 cP.
- In order to promote polymerization of the biodegradable polysaccharides in a composition to form a matrix, the oxidizing agent is added to the reducing agent in the presence of the one or more biodegradable polysaccharides. For example, a composition including a biodegradable polysaccharide and a reducing agent is added to a composition including an oxidizing agent, or a composition including a biodegradable polysaccharide and an oxidizing agent is added to a composition containing a reducing agent. One desirable method of preparing a matrix is to combine a composition including a biodegradable polysaccharide and an oxidizing agent with a composition including a biodegradable polysaccharide and a reducing agent. For purposes of describing this method, the terms “first composition” and “second composition” can be used.
- The viscosities of biodegradable polysaccharide in the first and second compositions can be the same or can be different. Generally, though, it has been observed that good mixing and subsequent matrix formation is obtained when the compositions have the same or similar viscosities. In this regard, if the same biodegradable polymer is used in the first and second compositions, the concentration of the biodegradable polymer may be the same or different.
- The oxidizing agent can be selected from inorganic or organic oxidizing agents, including enzymes; the reducing agent can be selected from inorganic or organic reducing agents, including enzymes. Exemplary oxidizing agents include peroxides, including hydrogen peroxide, metal oxides, and oxidases, including glucose oxidase. Exemplary reducing agents include salts and derivatives of electropositive elemental metals such as Li, Na, Mg, Fe, Zn, Al, and reductases. In one mode of practice, the reducing agent is present at a concentration of about 2.5 mM or greater when the reducing agent is mixed with the oxidizing agent. Prior to mixing, the reducing agent can be present in a composition at a concentration of, for example, 5 mM or greater.
- Other reagents can be present in the composition to promote polymerization of the biodegradable polysaccharide. Other polymerization promoting compounds can be included in the composition, such as metal or ammonium salts of persulfate.
- Optionally, the compositions and methods of the invention can include polymerization accelerants that can improve the efficiency of polymerization. Examples of useful accelerants include N-vinyl compounds, particularly N-vinyl pyrrolidone and N-vinyl caprolactam. Such accelerants can be used, for instance, at a concentration of between about 0.01% and about 5%, and preferably between about 0.05% and about 0.5%, by weight, based on the volume of the coating composition.
- In some aspects, an aqueous composition that includes the natural biodegradable polysaccharide, such as amylose or maltodextrin having pendent coupling groups, and a bioactive agent is obtained and used in the method of coating a surface. In another aspect, an aqueous composition is used to form an article. This composition can be prepared by mixing a bioactive agent, such as a water-soluble small molecule, a protein, or a nucleic acid, with the natural biodegradable polysaccharide.
- According to the invention, the natural biodegradable polysaccharide that includes a coupling group is used to form a coating on the surface of a medical device or to form an article. Other polysaccharides can also be present in the coating composition. For example, the coating can include two different natural biodegradable polysaccharides, or more than two different natural biodegradable polysaccharides. For example, in some cases the natural biodegradable polysaccharide (such as amylose or maltodextrin) can be present in the coating or article composition along with another biodegradable polymer (i.e., a secondary polymer), or more than one other biodegradable polymer. An additional polymer or polymers can be used to alter the properties of the matrix, or serve as bulk polymers to alter the volume of the matrix. For example, other biodegradable polysaccharides can be used in combination with the amylose polymer. These include hyaluronic acid, dextran, starch, amylose (for example, non-derivitized), amylopectin, cellulose, xanthan, pullulan, chitosan, pectin, inulin, alginates, and heparin.
- In some aspects of the invention, a composition is disposed on a surface that includes at least the natural biodegradable polysaccharide, such as amylose or maltodextrin having a coupling group and a bioactive agent. In some embodiments the composition includes the natural biodegradable polysaccharide, a bioactive agent, and an initiator. In other embodiments, a coating is formed by disposing the natural biodegradable polysaccharide and disposing the biodegradable microparticles on a surface. In some embodiments a composition containing both the natural biodegradable polysaccharide and the biodegradable microparticles having the bioactive agent are disposed on a surface. In yet other embodiments of the invention, a sealant composition that includes at least the natural biodegradable polysaccharide having a coupling group is disposed on a porous surface.
- The concentration of the natural biodegradable polysaccharide in the composition can be chosen to provide a coating or an article having a desired density of crosslinked natural biodegradable polysaccharide. In some embodiments, the concentration of natural biodegradable polysaccharide in the composition can depend on the type or nature of the bioactive agent that is included in the composition. In some embodiments the natural biodegradable polysaccharide having the coupling groups is present in the coating composition at a concentration in the range of 5-100% (w/v), and 5-50%, and in more specific embodiments in the range of 10-20% and in other embodiments in the range of 20-50% (w/v).
- For example, in forming a medical implant, the concentration of the natural biodegradable polysaccharide may be higher to provide a more structurally rigid implant.
- Other polymers or non-polymeric compounds can be included in the composition that can change or improve the properties of the coating or article that is formed by the natural biodegradable coating having coupling groups in order to change the elasticity, flexibility, wettability, or adherent properties, (or combinations thereof) of the coating formed on the surface.
- For example, in order to improve the properties of a coating, such as a sealant coating when formed, it is possible to include in the mixture one or a combination of plasticizing agents. Suitable plasticizing agents include glycerol, diethylene glycol, sorbitol, sorbitol esters, maltitol, sucrose, fructose, invert sugars, corn syrup, and mixtures thereof. The amount and type of plasticizing agents can be readily determined using known standards and techniques.
- Compositions of this invention can be used to coat the surface of a variety of implantable devices. The coating of natural biodegradable polysaccharide (with or without bioactive agent) can be applied to a medical device using standard techniques to cover the entire surface of the device, or a portion of the device surface.
- The medical articles on which the biodegradable coating can be formed can be fabricated from any suitable biomaterial or combinations of biomaterials. Preferred biomaterials include those formed of synthetic polymers, including oligomers, homopolymers, and copolymers resulting from either addition or condensation polymerizations.
- Examples of suitable addition polymers include, but are not limited to, acrylics such as those polymerized from methyl acrylate, methyl methacrylate, hydroxyethyl methacrylate, hydroxyethyl acrylate, acrylic acid, methacrylic acid, glyceryl acrylate, glyceryl methacrylate, methacrylamide, and acrylamide; vinyls such as ethylene, propylene, vinyl chloride, vinyl acetate, vinyl pyrrolidone, and vinylidene difluoride.
- Examples of condensation polymers include, but are not limited to, nylons such as polycaprolactam, polylauryl lactam, polyhexamethylene adipamide, and polyhexamethylene dodecanediamide, and also polyurethanes, polycarbonates, polyamides, polysulfones, poly(ethylene terephthalate), polylactic acid, polyglycolic acid, polydimethylsiloxanes, and polyetherketone.
- Other suitable biomaterials include metals, metal alloys, and ceramics. The metals and metal alloys include, but are not limited to, titanium, Nitinol, stainless steel, tantalum, and cobalt chromium. A second class of metals includes the noble metals such as gold, silver, copper, and platinum uridium. The ceramics include, but are not limited to, silicon nitride, silicon carbide, zirconia, and alumina, as well as glass, silica, and sapphire. Combinations of ceramics and metals are another class of biomaterials.
- Certain natural materials are also suitable biomaterials, including human tissue such as bone, cartilage, skin and teeth; and other organic materials such as wood, cellulose, compressed carbon, and rubber.
- The surface of such biomaterials can be pretreated (for example, with a Parylene coating composition) in order to alter the surface properties of the biomaterial, when desired.
- The biomaterials as described herein can be used to fabricate a variety of implantable devices on which the biodegradable coating can be formed. The medical device can be any device that is introduced temporarily or permanently into a mammal for the prophylaxis or treatment of a medical condition. These devices include any that are introduced subcutaneously, percutaneously or surgically to rest within an organ, tissue, or lumen of an organ, such as arteries, veins, ventricles or atria of the heart. The device can be a biostable device, a partially degradable device, or a completely degradable device (for example, stents can be fabricated from biodegradable polymeric materials).
- The natural biodegradable polysaccharide coating (in some embodiments including biodegradable microparticles) can be formed on the surface of virtually any implantable device. Exemplary implantable devices include but are not limited to drug-delivering vascular stents; other vascular devices (e.g., grafts, catheters, valves, artificial hearts, heart assist devices); implantable defibrillators; blood oxygenator devices; surgical devices; tissue-related materials; membranes; cell culture devices; chromatographic support materials; biosensors; shunts for hydrocephalus; wound management devices; endoscopic devices; infection control devices; orthopedic devices; dental devices, urological devices; colostomy bag attachment devices; ophthalmic devices; glaucoma drain shunts; synthetic prostheses; intraocular lenses; respiratory, peripheral cardiovascular, spinal, neurological, dental, and ear/nose/throat devices (e.g., ear drainage tubes); renal devices; and dialysis articles (e.g., tubing, membranes, grafts).
- Other contemplated devices include self-expanding stents (e.g., made from nitinol), balloon-expanded stents (e.g., prepared from stainless steel), degradable coronary stents, non-degradable coronary stents, peripheral coronary stents, urinary catheters (e.g., surface-coated with antimicrobial agents), penile implants, sphincter devices, urethral devices, bladder devices, renal devices, vascular implants and grafts, intravenous catheters (e.g., treated with antithrombotic agents), small diameter grafts, artificial lung catheters, electrophysiology catheters, anastomosis devices, vertebral disks, bone pins, suture anchors, hemostatic barriers, clamps, surgical staples/sutures/screws/plates/clips, atrial septal defect closures, electro-stimulation leads for cardiac rhythm management (e.g., pacer leads), glucose sensors (long-term and short-term), blood pressure and stent graft catheters, blood oxygenator tubing, blood oxygenator membranes, blood bags, birth control devices, breast implants, benign prostatic hyperplasia and prostate cancer implants, bone repair/augmentation devices, breast implants, cartilage repair devices, orthopedic joint implants, orthopedic fracture repairs, tissue adhesives, tissue sealants, tissue scaffolds, cerebral spinal fluid (CSF) shunts, dental implants, dental fracture repair devices, implanted drug infusion tubes, intravitreal drug delivery devices, nerve regeneration conduits, oncological implants, electrostimulation leads, pain management implants, spinal/orthopedic repair devices, wound dressings, embolic protection filters, abdominal aortic aneurysm grafts, heart valves (e.g., mechanical, polymeric, tissue, percutaneous, carbon, sewing cuff), valve annuloplasty devices, mitral valve repair devices, vascular intervention devices, left ventricle assist devices, neuro aneurysm treatment coils, neurological catheters, left atrial appendage filters, central venous access catheters, hemodialysis devices, catheter cuffs, anastomotic closures, vascular access catheters, cardiac sensors, uterine bleeding patches, urological catheters/stents/implants, in vitro diagnostics, aneurysm exclusion devices, neuropatches, Vena cava filters, urinary dialators, endoscopic surgical tissue extractors, atherectomy catheters, clot extraction catheters, percutaneous transluminal angioplasty (PTA) catheters, percutaneous transluminal coronary angioplasty (PTCA) catheters, stylets (vascular and non-vascular), coronary guidewires, drug infusion catheters, esophageal stents, circulatory support systems, angiographic catheters, transition sheaths and dialators, coronary and peripheral guidewires, hemodialysis catheters, neurovascular balloon catheters, tympanostomy vent tubes, cerebro-spinal fluid shunts, defibrillator leads, percutaneous closure devices, drainage tubes, thoracic cavity suction drainage catheters, electrophysiology catheters, stroke therapy catheters, abscess drainage catheters, biliary drainage products, dialysis catheters, central venous access catheters, and parental feeding catheters.
- The compositions are particularly useful for forming biodegradable coatings on the surface of devices that will come in contact with aqueous systems. The body fluids typically have enzymes that allow for the degradation of the natural biodegradable polysaccharide-based coating. The aqueous system (such as bodily fluids) allows for the degradation of the biodegradable coating and release of the bioactive agent from the device. In some cases, depending on the bioactive agent and the matrix, the bioactive agent can diffuse out of the matrix. For example, it has been demostrated that a loosely formed matrix may allow some diffusion of bioactive agents, particularly smaller bioactive agents. More desirably, well-formed matrices having signification polysaccharide association via coupling groups are able to retain bioactive agents. Release of bioactive agents from these matrices is mediated by enzymatic degradation.
- The coatings can also be formed on a biological article. A “biological article” refers to any sort of non-synthetic biologically-based article such as a cell or a portion of a cell, a group of cells, tissue, or an organ or a portion of a organ. The present reagents can be used in methods for encapsulating cellular material.
- In some aspects of the invention, a sealant coating is provided on a porous surface of a medical article. The medical article can be any article that is introduced into a mammal for the prophylaxis or treatment of a medical condition, wherein the medical article include a sealant coating (at least initially) and has a sealant function. The medical article having the sealant coating can provide one or more functions, including providing a barrier to the movement of body fluids, such as blood.
- The sealant coatings can be formed on the surface of articles that have a porous structure wherein it is desired to seal the porous structure, providing a barrier to the movement of body fluids. In many cases it is desirable to form these artificial barriers to ensure that the implanted article functions as it is intended to in the body. Gradually, however, it is desired to allow the body to maintain the function of the sealant coating by replacing the sealant barrier materials with natural materials from the body.
- The sealant composition can be prepared and/or applied in such a manner as to fill the pores on the surface of the article with the sealant material. This can be achieved by, for example, controlling factors such as the viscosity of the coating composition and the coupling of the natural biodegradable polysaccharides during formation of the coating.
- An article having a “porous surface” refers to any article having a surface with pores on which a natural biodegradable polysaccharide-based sealant coating can be formed. The pores are preferably of a physical dimension that permits in-growth of tissue into the pores as the sealant coating degrades. The porous surface can be associated with a non-porous surface, such as a scaffold that can provide support to the porous surface.
- The medical article can include porous surfaces that can be provided with a sealant coating and non-porous surfaces that are not coated with the sealant coating, optionally coated with the sealant coating, or coated with a material that is different than the sealant coating. All or a portion of the porous surfaces can be coated with the sealant coating. In some cases a sealant material that is different than the natural biodegradable polysaccharide-based sealant material can be used in conjunction with the natural biodegradable polysaccharide-based sealant material.
- For articles that have interior and exterior porous surfaces, either the interior or the exterior portions can be coated, or portions of the interior and/or exterior can be coated. The portion or portions of the article that are coated can depend on a particular desired application or function of the coated article. For example, in some cases it may be desirable to have a difference in the flow of fluids, such as blood, through porous portions of the medical article. Also, tissue in-growth on selected portions of the article can also be promoted by depositing the sealant coating at desired locations.
- The porous surface of the article can also include a material that is thrombogenic and/or presents surface stasis areas (regions of minimized or no blood flow). Depending on the application, a surface having a desired degree of porosity is obtained. The surface will have a degree of porosity sufficient for proper in-growth of cells and tissue forming factors. Upon tissue in-growth, the surface can provide a barrier that is fluid impermeable.
- In many cases the porous surface of the article is a fabric or has fabric-like qualities. The porous surface can be formed from textiles, which include woven materials, knitted materials, and braided materials. Particularly useful textile materials are woven materials which can be formed using any suitable weave pattern known in the art.
- The porous surface can be that of a graft, sheath, cover, patch, sleeve, wrap, casing, and the like. These types of articles can function as the medical article itself or be used in conjunction with another part of a medical article (examples of which are described herein).
- The porous surface can include any suitable type of biomaterial. Useful biomaterials can be woven into fibers for the preparation of fabrics as described herein. Useful materials include synthetic addition or condensation polymers such as polyesters, polypropylenes, polyethylenes, polyurethanes, and polytetrafluoroethylenes. Polyethylene terephthalate (PET) is a commonly used polymer in fabrics. Blends of these polymers can also be utilized in the preparation of fibers, such as monofilament or multi-filament fibers, for the construction of fabrics. Commonly used fabrics include those such as nylon, velour, and DACRON™.
- The fabrics can optionally include stiffening materials to improve the physical properties of the article, for example, to improve the strength of a graft. Such materials can improve the function of an implanted article. For example, strengthening materials can improve the patency of the graft.
- Porous surfaces can also be formed by dipping mandrels in these types of polymers.
- Other particular contemplated porous surfaces include those of cardiac patches. These can be used to decrease suture line bleeding associated with cardiovascular reconstructions. The patches can be used to seal around the penetrating suture. Common materials used in cardiac patches include PTFE and DACRON™.
- The thickness of the material used as the porous surface can be chosen depending on the application. However, it is common that these thicknesses are about 1.0 mm or less on average, and typically in the range of about 0.10 mm to about 1.0 mm.
- Other particular contemplated porous surfaces include grafts, particularly grafts having textured exterior portions. Examples of textured grafts include those that have velour-textured exteriors, with textured or smooth interiors. Grafts constructed from woven textile products are well known in the art and have been described in numerous documents, for example, U.S. Pat. No. 4,047,252; U.S. Pat. No. 5,178,630; U.S. Pat. No. 5,282,848; and U.S. Pat. No. 5,800,514.
- The natural biodegradable polysaccharide can be used to provide a sealant coating to a wide variety of articles. As used herein, “article” is used in its broadest sense and includes objects such as devices. Such articles include, but are not limited to vascular implants and grafts, grafts, surgical devices; synthetic prostheses; vascular prosthesis including endoprosthesis, stent-graft, and endovascular-stent combinations; small diameter grafts, abdominal aortic aneurysm grafts; wound dressings and wound management device; hemostatic barriers; mesh and hernia plugs; patches, including uterine bleeding patches, atrial septic defect (ASD) patches, patent foramen ovale (PFO) patches, ventricular septal defect (VSD) patches, and other generic cardiac patches; ASD, PFO, and VSD closures; percutaneous closure devices, mitral valve repair devices; left atrial appendage filters; valve annuloplasty devices, catheters; central venous access catheters, vascular access catheters, abscess drainage catheters, drug infusion catheters, parental feeding catheters, intravenous catheters (e.g., treated with antithrombotic agents), stroke therapy catheters, blood pressure and stent graft catheters; anastomosis devices and anastomotic closures; aneurysm exclusion devices; biosensors including glucose sensors; birth control devices; breast implants; cardiac sensors; infection control devices; membranes; tissue scaffolds; tissue-related materials; shunts including cerebral spinal fluid (CSF) shunts, glaucoma drain shunts; dental devices and dental implants; ear devices such as ear drainage tubes, tympanostomy vent tubes; ophthalmic devices; cuffs and cuff portions of devices including drainage tube cuffs, implanted drug infusion tube cuffs, catheter cuff, sewing cuff; spinal and neurological devices; nerve regeneration conduits; neurological catheters; neuropatches; orthopedic devices such as orthopedic joint implants, bone repair/augmentation devices, cartilage repair devices; urological devices and urethral devices such as urological implants, bladder devices, renal devices and hemodialysis devices, colostomy bag attachment devices; biliary drainage products.
- In some aspects, the polymeric compositions can be utilized in connection with an ophthalmic article. The ophthalmic article can be configured for placement at an external or internal site of the eye. Suitable ophthalmic articles in accordance with these aspects can provide bioactive agent to any desired area of the eye. In some aspects, the articles can be utilized to deliver bioactive agent to an anterior segment of the eye (in front of the lens), and/or a posterior segment of the eye (behind the lens). Suitable ophthalmic devices can also be utilized to provide bioactive agent to tissues in proximity to the eye, when desired. The biodegradable polysaccharide compositions can be used either for the formation of a coating on the surface of an ophthalmic article, or in the construction of an ophthalmic article.
- Suitable external articles can be configured for topical administration of bioactive agent. Such external devices can reside on an external surface of the eye, such as the cornea (for example, contact lenses) or bulbar conjunctiva. In some embodiments, suitable external devices can reside in proximity to an external surface of the eye.
- Articles configured for placement at an internal site of the eye can reside within any desired area of the eye. In some aspects, the ophthalmic article can be configured for placement at an intraocular site, such as the vitreous. Illustrative intraocular devices include, but are not limited to, those described in U.S. Pat. No. 6,719,750 B2 (“Devices for Intraocular Drug Delivery,” Varner et al.) and U.S. Pat. No. 5,466,233 (“Tack for Intraocular Drug Delivery and Method for Inserting and Removing Same,” Weiner et al.); U.S. Publication Nos. 2005/0019371 A1 (“Controlled Release Bioactive Agent Delivery Device,” Anderson et al.), 2004/0133155 A1 (“Devices for Intraocular Drug Delivery,” Varner et al.), 2005/0059956 A1 (“Devices for Intraocular Drug Delivery,” Varner et al.), and U.S. application Ser. No. 11/204,195 (filed Aug. 15, 2005, Anderson et al.), Ser. No. 11/204,271 (filed Aug. 15, 2005, Anderson et al.), Ser. No. 11/203,981 (filed Aug. 15, 2005, Anderson et al.), Ser. No. 11/203,879 (filed Aug. 15, 2005, Anderson et al.), Ser. No. 11/203,931 (filed Aug. 15, 2005, Anderson et al.); and related applications.
- In some aspects of the invention, the biodegradable polysaccharide coating is included on a non-linear intraocular device. In some aspects of the invention, the biodegradable polysaccharide coating includes a bioactive agent, such as a high molecular weight bioactive agent useful for treating an ocular condition.
- In some aspects, the ophthalmic article can be configured for placement, or can be formed, at a subretinal area within the eye. Illustrative ophthalmic devices for subretinal application include, but are not limited to, those described in U.S. Patent Publication No. 2005/0143363 (“Method for Subretinal Administration of Therapeutics Including Steroids; Method for Localizing Pharmacodynamic Action at the Choroid and the Retina; and Related Methods for Treatment and/or Prevention of Retinal Diseases,” de Juan et al.); U.S. application Ser. No. 11/175,850 (“Methods and Devices for the Treatment of Ocular Conditions,” de Juan et al.); and related applications.
- In some aspects, the invention provides a biodegradable implant that is formed from the biodegradable polysaccharide and that includes a bioactive agent, such as a high molecular weight bioactive agent useful for treating an ocular condition.
- In some aspects, the invention provides a method for forming an article from the biodegradable polysaccharide, wherein the method includes polymerizing a composition that includes the biodegradable polysaccharide within the eye, such as in a subretinal area or within the vitreous. For example, a low viscosity composition including a natural biodegradable polysaccharide and a redox pair to promote polymerization for in situ matrix formation.
- Ophthalmic articles can also be configured for placement within any desired tissues of the eye. For example, ophthalmic devices can be configured for placement at a subconjunctival area of the eye, such as devices positioned extrasclerally but under the conjunctiva, such as glaucoma drainage devices and the like.
- A medical article having a biodegradable coating can also be prepared by assembling an article having two or more “parts” (for example, pieces of a medical article that can be put together to form the article) wherein at least one of the parts has a biodegradable coating. All or a portion of the part of the medical article can have a biodegradable coating. In this regard, the invention also contemplates parts of medical article (for example, not the fully assembled article) that have a natural biodegradable polysaccharide-based coating.
- In some aspects of the invention the natural biodegradable polymer is used to form the body member of a medical implant, wherein the body member has a wet weight of about 10 g or less, or a dry weight of about 2.5 g or less.
- The device can also have a base coating of material. The base coating can serve one or more functions, for example, it can provide an improved surface for the natural biodegradable polysaccharide or composition that includes the natural biodegradable polysaccharide. The base coating can include a polymeric material, such as a natural or synthetic polymer. Examples of suitable compounds that can be used to pretreat a surface to provide a base coat include Parylene and organosilane compounds. Suitable base coatings can include, for example, methacrylate, acrylate, alkylacrylate, acrylamide, vinylpyrrolidinone, vinylacetamide, and vinyl formamide based polymers and copolymers. These polymers can also include latent reactive groups such as photoreactive groups.
- Base coatings can be useful in various coating processes. For example, in some aspects, biodegradable microparticles can be first disposed on a base coat and then the natural biodegradable polysaccharide having coupling groups can be disposed on the microparticles. The surface can then be treated to form a coating wherein the microparticles are predominantly located between the base layer and the layer formed from the natural biodegradable polysaccharide having coupling groups. If desired, an initiator can be included in a base coating and the natural biodegradable polysaccharide polymer or composition that includes the natural biodegradable polysaccharide polymer can be disposed on the base coating. The base coating can serve one or more functions, for example, it can provide an improved surface for the natural biodegradable polysaccharide or composition that includes the natural biodegradable polysaccharide.
- In many aspects of the invention, the natural biodegradable polysaccharide coating or the biodegradable article includes one or more bioactive agents. The bioactive agent can be dispersed within the natural biodegradable polysaccharide coating or biodegradable article itself. Alternatively, the bioactive agent can be present in microparticles that are associated with the natural biodegradable polysaccharide coating. The bioactive agent can be delivered from the coated surface upon degradation of the natural biodegradable polysaccharide and/or biodegradable microparticles.
- The term “bioactive agent” refers to a peptide, protein, carbohydrate, nucleic acid, lipid, polysaccharide, synthetic inorganic or organic molecule, viral particle, cell, or combinations thereof, that causes a biological effect when administered in vivo to an animal, including but not limited to birds and mammals, including humans. Nonlimiting examples are antigens, enzymes, hormones, receptors, peptides, and gene therapy agents. Examples of suitable gene therapy agents include (a) therapeutic nucleic acids, including antisense DNA, antisense RNA, and interference RNA, and (b) nucleic acids encoding therapeutic gene products, including plasmid DNA and viral fragments, along with associated promoters and excipients. Examples of other molecules that can be incorporated include nucleosides, nucleotides, vitamins, minerals, and steroids.
- Although not limited to such, the coatings of the invention are particularly useful for delivering bioactive agents that are large hydrophilic molecules, such as polypeptides (including proteins and peptides), nucleic acids (including DNA and RNA), polysaccharides (including heparin), as well as particles, such as viral particles, and cells. In one aspect, the bioactive agent has a molecular weight of about 10,000 or greater.
- Classes of bioactive agents which can be incorporated into biodegradable coatings (both the natural biodegradable matrix and/or the biodegradable microparticles) of this invention include, but are not limited to: ACE inhibitors, actin inhibitors, analgesics, anesthetics, anti-hypertensives, anti polymerases, antisecretory agents, anti-AIDS substances, antibiotics, anti-cancer substances, anti-cholinergics, anti-coagulants, anti-convulsants, anti-depressants, anti-emetics, antifungals, anti-glaucoma solutes, antihistamines, antihypertensive agents, anti-inflammatory agents (such as NSAIDs), anti metabolites, antimitotics, antioxidizing agents, anti-parasite and/or anti-Parkinson substances, antiproliferatives (including antiangiogenesis agents), anti-protozoal solutes, anti-psychotic substances, anti-pyretics, antiseptics, anti-spasmodics, antiviral agents, calcium channel blockers, cell response modifiers, chelators, chemotherapeutic agents, dopamine agonists, extracellular matrix components, fibrinolytic agents, free radical scavengers, growth hormone antagonists, hypnotics, immunosuppressive agents, immunotoxins, inhibitors of surface glycoprotein receptors, microtubule inhibitors, miotics, muscle contractants, muscle relaxants, neurotoxins, neurotransmitters, opioids, photodynamic therapy agents, prostaglandins, remodeling inhibitors, statins, steroids, thrombolytic agents, tranquilizers, vasodilators, and vasospasm inhibitors.
- Antibiotics are art recognized and are substances which inhibit the growth of or kill microorganisms. Examples of antibiotics include penicillin, tetracycline, chloramphenicol, minocycline, doxycycline, vancomycin, bacitracin, kanamycin, neomycin, gentamycin, erythromycin, cephalosporins, geldanamycin, and analogs thereof. Examples of cephalosporins include cephalothin, cephapirin, cefazolin, cephalexin, cephradine, cefadroxil, cefamandole, cefoxitin, cefaclor, cefuroxime, cefonicid, ceforanide, cefotaxime, moxalactarn, ceftizoxime, ceftriaxone, and cefoperazone.
- Antiseptics are recognized as substances that prevent or arrest the growth or action of microorganisms, generally in a nonspecific fashion, e.g., by inhibiting their activity or destroying them. Examples of antiseptics include silver sulfadiazine, chlorhexidine, glutaraldehyde, peracetic acid, sodium hypochlorite, phenols, phenolic compounds, iodophor compounds, quaternary ammonium compounds, and chlorine compounds.
- Anti-viral agents are substances capable of destroying or suppressing the replication of viruses. Examples of anti-viral agents include α-methyl-P-adamantane methylamine, hydroxy-ethoxyrnethylguanine, adamantanamine, 5-iodo-2′-deoxyuridine, trifluorothymidine, interferon, and adenine arabinoside.
- Enzyme inhibitors are substances that inhibit an enzymatic reaction. Examples of enzyme inhibitors include edrophonium chloride, N-methylphysostigmine, neostigmine bromide, physostigmine sulfate, tacrine HCl, tacrine, 1-hydroxymaleate, iodotubercidin, p-bromotetramisole, 10-(α-diethylaminopropionyl)-phenothiazine hydrochloride, calmidazolium chloride, hemicholinium-3,3,5-dinitrocatechol, diacylglycerol kinase inhibitor I, diacylglycerol kinase inhibitor II, 3-phenylpropargylamine, N-monomethyl-L-arginine acetate, carbidopa, 3-hydroxybenzylhydrazine HCl, hydralazine HCl, clorgyline HCl, deprenyl HCl, L(−), deprenyl HCl, D(+), hydroxylamine HCl, iproniazid phosphate, 6-MeO-tetrahydro-9H-pyrido-indole, nialamide, pargyline HCl, quinacrine HCl, semicarbazide HCl, tranylcypromine HCl, N,N-diethylaminoethyl-2,2-diphenylvalerate hydrochloride, 3-isobutyl-1-methylxanthine, papaverine HCl, indomethacin, 2-cyclooctyl-2-hydroxyethylamine hydrochloride, 2,3-dichloro-α-methylbenzylamine (DCMB), 8,9-dichloro-2,3,4,5-tetrahydro-1H-2-benzazepine hydrochloride, p-aminoglutethimide, p-aminoglutethimide tartrate, R(+), p-aminoglutethimide tartrate, S(−), 3-iodotyrosine, alpha-methyltyrosine, L(−) alpha-methyltyrosine, D L(−), cetazolamide, dichlorphenamide, 6-hydroxy-2-benzothiazolesulfonamide, and allopurinol.
- Anti-pyretics are substances capable of relieving or reducing fever. Anti-inflammatory agents are substances capable of counteracting or suppressing inflammation. Examples of such agents include aspirin (salicylic acid), indomethacin, sodium indomethacin trihydrate, salicylamide, naproxen, colchicine, fenoprofen, sulindac, diflunisal, diclofenac, indoprofen and sodium salicylamide. Local anesthetics are substances that have an anesthetic effect in a localized region. Examples of such anesthetics include procaine, lidocaine, tetracaine and dibucaine.
- Cell response modifiers are chemotactic factors such as platelet-derived growth factor (pDGF). Other chemotactic factors include neutrophil-activating protein, monocyte chemoattractant protein, macrophage-inflammatory protein, SIS (small inducible secreted) proteins, platelet factor, platelet basic protein, melanoma growth stimulating activity, epidermal growth factor, transforming growth factor (alpha), fibroblast growth factor, platelet-derived endothelial cell growth factor, insulin-like growth factor, nerve growth factor, and bone growth/cartilage-inducing factor (alpha and beta). Other cell response modifiers are the interleukins, interleukin inhibitors or interleukin receptors, including
interleukin 1 throughinterleukin 10; interferons, including alpha, beta and gamma; hematopoietic factors, including erythropoietin, granulocyte colony stimulating factor, macrophage colony stimulating factor and granulocyte-macrophage colony stimulating factor; tumor necrosis factors, including alpha and beta; transforming growth factors (beta), including beta-1, beta-2, beta-3, inhibin, activin, and DNA that encodes for the production of any of these proteins. - Examples of statins include lovastatin, pravastatin, simvastatin, fluvastatin, atorvastatin, cerivastatin, rousvastatin, and superstatin.
- Imaging agents are agents capable of imaging a desired site, e.g., tumor, in vivo, can also be included in the coating composition. Examples of imaging agents include substances having a label which is detectable in vivo, e.g., antibodies attached to fluorescent labels. The term antibody includes whole antibodies or fragments thereof.
- Exemplary ligands or receptors include antibodies, antigens, avidin, streptavidin, biotin, heparin, type IV collagen, protein A, and protein G.
- Exemplary antibiotics include antibiotic peptides.
- In some aspects the bioactive agent can be selected to improve the compatibility (for example, with blood and/or surrounding tissues) of medical device surfaces. These agents, referred to herein as “biocompatible agents,” when associated with the medical device surface, can serve to shield the blood from the underlying medical device material. Suitable biocompatible agents preferably reduce the likelihood for blood components to adhere to the medical device, thus reducing the formation of thrombus or emboli (blood clots that release and travel downstream).
- The bioactive agent can improve the biocompatibility of the medical article having a coating that includes the natural biodegradable polymer and the biodegradable microparticle. The bioactive agent can provide antirestenotic effects, such as antiproliferative, anti-platelet, and/or antithrombotic effects. In some embodiments, the bioactive agent can include anti-inflammatory agents, immunosuppressive agents, cell attachment factors, receptors, ligands, growth factors, antibiotics, enzymes, nucleic acids, and the like. Compounds having antiproliferative effects include, for example, actinomycin D, angiopeptin, c-myc antisense, paclitaxel, taxane, and the like.
- Representative examples of bioactive agents having antithrombotic effects include heparin, heparin derivatives, sodium heparin, low molecular weight heparin, hirudin, lysine, prostaglandins, argatroban, forskolin, vapiprost, prostacyclin and prostacyclin analogs, D-ph-pr-arg-chloromethylketone (synthetic antithrombin), dipyridamole, glycoprotein IIb/IIIa platelet membrane receptor antibody, coprotein IIb/IIIa platelet membrane receptor antibody, recombinant hirudin, thrombin inhibitor (such as commercially available from Biogen), chondroitin sulfate, modified dextran, albumin, streptokinase, tissue plasminogen activator (TPA), urokinase, nitric oxide inhibitors, and the like.
- The bioactive agent can also be an inhibitor of the GPIIb-IIIa platelet receptor complex, which mediates platelet aggregation. GPIIb/IIIa inhibitors can include monoclonal antibody Fab fragment c7E3, also know as abciximab (ReoPrO™), and synthetic peptides or peptidomimetics such as eptifibatide (Integrilin™) or tirofiban (Agrastat™).
- The bioactive agent can be an immunosuppressive agent, for example, cyclosporine, CD-34 antibody, everolimus, mycophenolic acid, sirolimus, tacrolimus, and the like.
- Other exemplary therapeutic antibodies include trastuzumab (Herceptin™), a humanized anti-HER2 monoclonal antibody (moAb); alemtuzumab (Campath™), a humanized anti-CD52 moAb; gemtuzumab (Mylotarg™), a humanized anti-CD33 moAb; rituximab (Rituxan™), a chimeric anti-CD20 moAb; ibritumomab (Zevalin™), a murine moAb conjugated to a beta-emitting radioisotope; tositumomab (Bexxar™), a murine anti-CD20 moAb; edrecolomab (Panorex™), a murine anti-epithelial cell adhesion molecule moAb; cetuximab (Erbitux™), a chimeric anti-EGFR moAb; and bevacizumab (Avastin™), a humanized anti-VEGF moAb.
- Additionally, the bioactive agent can be a surface adhesion molecule or cell-cell adhesion molecule. Exemplary cell adhesion molecules or attachment proteins (such as extracellular matrix proteins including fibronectin, laminin, collagen, elastin, vitronectin, tenascin, fibrinogen, thrombospondin, osteopontin, von Willibrand Factor, bone sialoprotein (and active domains thereof), or a hydrophilic polymer such as hyaluronic acid, chitosan or methyl cellulose, and other proteins, carbohydrates, and fatty acids. Exemplary cell-cell adhesion molecules include N-cadherin and P-cadherin and active domains thereof.
- Exemplary growth factors include fibroblastic growth factors, epidermal growth factor, platelet-derived growth factors, transforming growth factors, vascular endothelial growth factor, bone morphogenic proteins and other bone growth factors, and neural growth factors.
- The bioactive agent can be also be selected from mono-2-(carboxymethyl)hexadecanamidopoly(ethylene glycol)200mono-4-benzoylbenzyl ether, mono-3-carboxyheptadecanamidopoly(ethylene glycol)200mono-4-benzoylbenzyl ether, mono-2-(carboxymethyl)hexadecanamidotetra(ethylene glycol)mono-4-benzoylbenzyl ether, mono-3-carboxyheptadecanamidotetra(ethylene glycol)mono-4-benzoylbenzyl ether, N-[2-(4-benzoylbenzyloxy)ethyl]-2-(carboxymethyl)hexadecanamide, N-[2-(4-benzoylbenzyloxy)ethyl]-3-carboxyheptadecanamide, N-[12-(benzoylbenzyloxy)dodecyl]-2-(carboxymethyl)hexadecanamide, N-[12-(benzoylbenzyloxy)dodecyl]-3-carboxy-heptadecanamide, N-[3-(4-benzoylbenzamido)propyl]-2-(carboxymethyl)hexadecanamide, N-[3-(4-benzoylbenzamido)propyl]-3-carboxyheptadecanamide, N-(3-benzoylphenyl)-2-(carboxymethyl)hexadecanamide, N-(3-benzoylphenyl)-3-carboxyheptadecanamide, N-(4-benzoylphenyl)-2-(carboxymethyl)hexadecanamide, poly(ethylene glycol)200mono-15-carboxypentadecyl mono-4-benzoylbenzyl ether, and mono-15-carboxypentadecanamidopoly(ethylene glycol)200mono-4-benzoylbenzyl ether.
- Additional examples of contemplated bioactive agents and/or bioactive agent include analogues of rapamycin (“rapalogs”), ABT-578 from Abbott, dexamethasone, betamethasone, vinblastine, vincristine, vinorelbine, poside, teniposide, daunorubicin, doxorubicin, idarubicin, anthracyclines, mitoxantrone, bleomycins, plicamycin (mithramycin), mitomycin, mechlorethamine, cyclophosphamide and its analogs, melphalan, chlorambucil, ethylenimines and methylmelamines, alkyl sulfonates-busulfan, nitrosoureas, carmustine (BCNU) and analogs, streptozocin, trazenes-dacarbazinine, methotrexate, fluorouracil, floxuridine, cytarabine, mercaptopurine, thioguanine, pentostatin, 2-chlorodeoxyadenosine, cisplatin, carboplatin, procarbazine, hydroxyurea, mitotane, estrogen, ticlopidine, clopidogrel, abciximab, breveldin, cortisol, cortisone, fludrocortisone, prednisone, prednisolone, 6U-methylprednisolone, triamcinolone, acetaminophen, etodalac, tolmetin, ketorolac, ibuprofen and derivatives, mefenamic acid, meclofenamic acid, piroxicam, tenoxicam, phenylbutazone, oxyphenthatrazone, nabumetone, auranofin, aurothioglucose, gold sodium thiomalate, azathioprine, mycophenolate mofetil; angiotensin receptor blockers; nitric oxide donors; and mTOR inhibitors.
- Viral particles and viruses include those that may be therapeutically useful, such as those used for gene therapy, and also attenuated viral particles and viruses which can promote an immune response and generation of immunity. Useful viral particles include both natural and synthetic types. Viral particles include, but are not limited to, adenoviruses, baculoviruses, parvoviruses, herpesviruses, poxviruses, adeno-associated viruses, vaccinia viruses, and retroviruses.
- Other bioactive agents that can be used for altering gene function include plasmids, phages, cosmids, episomes, and integratable DNA fragments, antisense oligonucleotides, antisense DNA and RNA, modified DNA and RNA, iRNA, ribozymes, siRNA, and shRNA.
- Other bioactive agents include cells such as platelets, stem cells, T lymphocytes, B lymphocytes, acidophils, adipocytes, astrocytes, basophils, hepatocytes, neurons, cardiac muscle cells, chondrocytes, epithelial cells, dendrites, endrocrine cells, endothelial cells, eosinophils, erythrocytes, fibroblasts, follicular cells, ganglion cells, hepatocytes, endothelial cells, Leydig cells, parenchymal cells, lymphocytes, lysozyme-secreting cells, macrophages, mast cells, megakaryocytes, melanocytes, monocytes, myoid cells, neck nerve cells, neutrophils, oligodendrocytes, oocytes, osteoblasts, osteochondroclasts, osteoclasts, osteocytes, plasma cells, spermatocytes, reticulocytes, Schwann cells, Sertoli cells, skeletal muscle cells, and smooth muscle cells. Bioactive agents can also include genetically modified, recombinant, hybrid, mutated cells, and cells with other alterations.
- Additives such as inorganic salts, BSA (bovine serum albumin), and inert organic compounds can be used to alter the profile of bioactive agent release, as known to those skilled in the art.
- The concentration of the bioactive agent or agents dissolved or suspended in the coating mixture can range from about 0.01 to about 90 percent, by weight, based on the weight of the final coated composition.
- The particular bioactive agent, or combination of bioactive agents, can be selected depending upon one or more of the following factors: the application of the controlled delivery device, the medical condition to be treated, the anticipated duration of treatment, characteristics of the implantation site, the number and type of bioactive agents to be utilized, and the like.
- Any of the polymer compositions described herein can be provided to the surface of the medical article and can include any number of desired bioactive agents, depending upon the final application of the medical device.
- A comprehensive listing of bioactive agents can be found in The Merck Index, Thirteenth Edition, Merck & Co. (2001). Bioactive agents are commercially available from Sigma Aldrich Fine Chemicals, Milwaukee, Wis.
- In some aspects of the invention, the bioactive agent can be used to promote thrombosis in association with the natural biodegradable polysaccharide-based coating, which can be of particular use when a coating having a sealant function is desired. A sealant coating including a thrombogenic agent can promote the in-growth of tissue upon degradation of the sealant coating material. The degree of thrombosis can be controlled by various factors, including, for example, the presence of one or more thrombosis-promoting bioactive agents on or within the coating. Suitable thrombotic agents are described herein.
- In some aspects the thrombotic agent can be selected to have an affect on the blood and/or surrounding tissues that are in contact with the article surface. In some cases the thrombotic agent is chosen for the ability to affect the ability of blood components to adhere to the medical article. The thrombotic agent can, in some cases, be chosen to promote thrombus formation at the surface of the coated article. Therefore, in some embodiments, the sealant coating can include a thrombotic agent, such as thrombin, collagen (for example, (synthetic) recombinant human collagen (FibroGen, South San Francisco, Calif.)), ADP, or convulxin to promote thrombosis at the coated surface of the article.
- Other prothrombotic or procoagulant factors include platelet factors 1-4, platelet activating factor (acetyl glyceryl ether phosphoryl choline); P-selectin and von Willebrand Factor (vWF); tissue factor; plasminogen activator initiator-1; thromboxane; procoagulant thrombin-like enzymes including cerastotin and afaacytin; phospholipase A2; Ca2+-dependent lectins (C-type lectin); factors that bind glycoprotein receptors and induce aggregation including aggretin, rhodocytin, aggregoserpentin, triwaglerin, and equinatoxin; glycoprotein Ib agonists including mamushigin and alboaggregin; vWF interacting factors including botrocetin, bitiscetin, cerastotin, and ecarin.
- Other factors, including protein factors, that are involved in the clotting cascade include coagulation factors I-XIII (for example, fibrinogen, prothrombin, tissue thromboplastin, calcium, proaccelerin (accelerator globulin), proconvertin (serum prothrombin conversion accelerator), antihemophilic factor, plasma thromboplastin component, Stuart factor (autoprothrombin C), plasma thromboplastin antecedent (PTA), Hageman factor, and fibrin-stabilizing factor (FSF, fibrinase, protransglutaminase)).
- Some surface adhesion molecule or cell-cell adhesion molecules may also function to promote coagulation or thrombosis. Exemplary cell adhesion molecules or attachment proteins (such as extracellular matrix proteins) include fibronectin, laminin, collagen, elastin, vitronectin, tenascin, fibrinogen, thrombospondin, osteopontin, von Willebrand Factor, bone sialoprotein (and active domains thereof), or a hydrophilic polymer such as hyaluronic acid, chitosan or methyl cellulose, and other proteins, carbohydrates, and fatty acids. Exemplary cell-cell adhesion molecules include N-cadherin and P-cadherin and active domains thereof.
- The particular thrombotic agent, or a combination of thrombotic agents with other bioactive agents, can be selected depending upon one or more of the following factors: the application of the medical article, the medical condition to be treated, the anticipated duration of treatment, characteristics of the implantation site, the number and type of thrombogenic/bioactive agents to be utilized, the chemical composition of the sealant coating (such as amylose, selected additives, and the like), the extent of coupling in the formed sealant coating, and the like.
- Any of the sealant compositions described herein can be provided to the surface of the medical article. In some embodiments the sealant coating can include any number of desired thrombogenic/bioactive agents, depending upon the final application of the medical article. The coating of sealant material (with or without thrombogenic/bioactive agents) can be applied to the medical article using standard techniques to cover the entire surface of the article, or a portion of the article surface. Further, the sealant composition material can be provided as a single coated layer (with or without thrombogenic/bioactive agents), or as multiple coated layers (with or without thrombogenic/bioactive agents). When multiple coated layers are provided on the surface, the materials of each coated layer can be chosen to provide a desired effect.
- In some aspects of the invention, a microparticle is used to deliver the bioactive agent from the natural biodegradable polysaccharide-based coating. The microparticles of the invention can comprise any three-dimensional structure that can be immobilized on a substrate in association with the matrix formed by the amylose polymer. The term “microparticle” is intended to reflect that the three-dimensional structure is very small but not limited to a particular size range, or not limited to a structure that has a particular shape. According to the invention, microparticles typically have a size in the range of 5 nm to 100 μm in diameter. Generally microparticles are spherical or somewhat spherical in shape, but can have other shapes as well. In preferred embodiments of the invention, the biodegradable microparticles have a size in the range of 100 nm to 20 μm in diameter, and even more preferable in the range of 400 nm to 20 μm in diameter.
- The microparticle being “biodegradable” refers to the presence of one or more biodegradable materials in the microparticle. The biodegradable microparticles include at least a biodegradable material (such as a biodegradable polymer) and a bioactive agent. The biodegradable microparticles can gradually decompose and release bioactive agent upon exposure to an aqueous environment, such as body fluids.
- The biodegradable microparticle can also include one or more biodegradable polymers. Examples of biodegradable polymers that can be included in the biodegradable microparticle include, for example, polylactic acid, poly(lactide-co-glycolide), polycaprolactone, polyphosphazine, polymethylidenemalonate, polyorthoesters, polyhydroxybutyrate, polyalkeneanhydrides, polypeptides, polyanhydrides, and polyesters, and the like.
- Biodegradable polyetherester copolymers can be used. Generally speaking, the polyetherester copolymers are amphiphilic block copolymers that include hydrophilic (for example, a polyalkylene glycol, such as polyethylene glycol) and hydrophobic blocks (for example, polyethylene terephthalate). Examples of block copolymers include poly(ethylene glycol)-based and poly(butylene terephthalate)-based blocks (PEG/PBT polymer). Examples of these types of multiblock copolymers are described in, for example, U.S. Pat. No. 5,980,948. PEG/PBT polymers are commercially available from Octoplus BV, under the trade designation PolyActive™.
- Biodegradable copolymers having a biodegradable, segmented molecular architecture that includes at least two different ester linkages can also be used. The biodegradable polymers can be block copolymers (of the AB or ABA type) or segmented (also known as multiblock or random-block) copolymers of the (AB)n type. These copolymers are formed in a two (or more) stage ring opening copolymerization using two (or more) cyclic ester monomers that form linkages in the copolymer with greatly different susceptibilities to transesterification. Examples of these polymers are described in, for example, in U.S. Pat. No. 5,252,701 (Jarrett et al., “Segmented Absorbable Copolymer”).
- Other suitable biodegradable polymer materials include biodegradable terephthalate copolymers that include a phosphorus-containing linkage. Polymers having phosphoester linkages, called poly(phosphates), poly(phosphonates) and poly(phosphites), are known. See, for example, Penczek et al., Handbook of Polymer Synthesis, Chapter 17: “Phosphorus-Containing Polymers,” 1077-1132 (Hans R. Kricheldorf ed., 1992), as well as U.S. Pat. Nos. 6,153,212, 6,485,737, 6,322,797, 6,600,010, 6,419,709. Biodegradable terephthalate polyesters can also be used that include a phosphoester linkage that is a phosphite. Suitable terephthalate polyester-polyphosphite copolymers are described, for example, in U.S. Pat. No. 6,419,709 (Mao et al., “Biodegradable Terephthalate Polyester-Poly(Phosphite) Compositions, Articles, and Methods of Using the Same). Biodegradable terephthalate polyester can also be used that include a phosphoester linkage that is a phosphonate. Suitable terephthalate polyester-poly(phosphonate)copolymers are described, for example, in U.S. Pat. Nos. 6,485,737 and 6,153,212 (Mao et al., “Biodegradable Terephthalate Polyester-Poly(Phosphonate) Compositions, Articles and Methods of Using the Same). Biodegradable terephthalate polyesters can be used that include a phosphoester linkage that is a phosphate. Suitable terephthalate polyester-poly(phosphate)copolymers are described, for example, in U.S. Pat. Nos. 6,322,797 and 6,600,010 (Mao et al., “Biodegradable Terephthalate Polyester-Poly(Phosphate)Polymers, Compositions, Articles, and Methods for Making and Using the Same).
- Biodegradable polyhydric alcohol esters can also be used (See U.S. Pat. No. 6,592,895). This patent describes biodegradable star-shaped polymers that are made by esterifying polyhydric alcohols to provide acyl moieties originating from aliphatic homopolymer or copolymer polyesters. The biodegradable polymer can be a three-dimensional crosslinked polymer network containing hydrophobic and hydrophilic components which forms a hydrogel with a crosslinked polymer structure, such as that described in U.S. Pat. No. 6,583,219. The hydrophobic component is a hydrophobic macromer with unsaturated group terminated ends, and the hydrophilic polymer is a polysaccharide containing hydroxy groups that are reacted with unsaturated group introducing compounds. The components are convertible into a one-phase crosslinked polymer network structure by free radical polymerization. In yet further embodiments, the biodegradable polymer can comprise a polymer based upon α-amino acids (such as elastomeric copolyester amides or copolyester urethanes, as described in U.S. Pat. No. 6,503,538).
- The biodegradable microparticle can include one or more biodegradable polymers obtained from natural sources. In some preferred aspects the biodegradable polymer is selected from hyaluronic acid, dextran, starch, amylose, amylopectin, cellulose, xanthan, pullulan, chitosan, pectin, inulin, alginates, and heparin. One, or combinations of more than one of these biodegradable polymers, can be used. A particular biodegradable polymer can also be selected based on the type of bioactive agent that is present in the microparticle. Therefore, in some aspects of the invention, the biodegradable coating can include a natural biodegradable polysaccharide matrix and a natural biodegradable polysaccharide-containing microparticle.
- Therefore, in some embodiments, the microparticles include a natural biodegradable polysaccharide such as amylose or maltodextrin. In some embodiments the natural biodegradable polysaccharide can be the primary biodegradable component in the microparticle. In some embodiments, both the coating matrix and the microparticle include amylose and/or maltodextrin as components.
- Dextran-based microparticles can be particularly useful for the incorporation of bioactive agents such as proteins, peptides, and nucleic acids. Examples of the preparation of dextran-based microparticles are described in U.S. Pat. No. 6,303,148.
- The preparation of amylose and other starch-based microparticles have been described in various references, including, for example, U.S. Pat. No. 4,713,249; U.S. Pat. No. 6,692,770; and U.S. Pat. No. 6,703,048. Biodegradable polymers and their synthesis have been also been described in various references including Mayer, J. M., and Kaplan, D. L. (1994) Trends in Polymer Science 2: pages 227-235; and Jagur-Grodzinski, J., (1999) Reactive and Functional Polymers: Biomedical Application of Functional Polymers, Vol. 39, pages 99-138.
- In some aspects of the invention, the biodegradable microparticle contains a biologically active agent (a “bioactive agent”), such as a pharmaceutical or a prodrug. Microparticles can be prepared incorporating various bioactive agents by established techniques, for example, by solvent evaporation (see, for example, Wichert, B. and Rohdewald, P. J Microencapsul. (1993) 10:195). The bioactive agent can be released from the biodegradable microparticle (the microparticle being present in the natural biodegradable polysaccharide coating) upon degradation of the biodegradable microparticle in vivo. Microparticles having bioactive agent can be formulated to release a desired amount of the agent over a predetermined period of time. It is understood that factors affecting the release of the bioactive agent and the amount released can be altered by the size of the microparticle, the amount of bioactive agent incorporated into the microparticle, the type of degradable material used in fabricating the microparticle, the amount of biodegradable microparticles immobilized per unit area on the substrate, etc.
- The microparticles can also be treated with a porogen, such as salt, sucrose, PEG, or an alcohol, to create pores of a desired size for incorporation of the bioactive agent.
- The quantity of bioactive agents provided in the biodegradable microparticle can be adjusted by the user to achieve the desired effect. For example, a particular amount of anti-coagulant drug can be incorporated into the microparticle to provide a certain level of anti-coagulant activity from the biodegradable coating. Biologically active compounds can be provided by the microparticles in a range suitable for the application. In another example, protein molecules can be provided by biodegradable microparticles. For example, the amount of protein molecules present can be in the range of 1-250,000 molecules per 1 μm diameter microparticle.
- Generally, the concentration of the bioactive agent present in the biodegradable microparticles can be chosen based on any one or a combination of a number of factors, including, but not limited to, the release rate from the coating, the type of bioactive agent(s) in the coating, the desired local or systemic concentration of the bioactive agent following release, and the half life of the bioactive agent. In some cases the concentration of bioactive agent in the microparticle can be about 0.001% or greater, or in the range of about 0.001% to about 50 percent, or greater, by weight, based on the weight of the microparticle.
- The particular bioactive agent to be included in the biodegradable microparticle, or combination of bioactive agents in microparticles, can be selected depending upon factors such as the application of the coated device, the medical condition to be treated, the anticipated duration of treatment, characteristics of the implantation site, the number and type of bioactive agents to be utilized, the chemical composition of the microparticle, size of the microparticle, crosslinking, and the like.
- In one embodiment, the invention advantageously allows for preparation of surfaces having two, or more than two, different bioactive agents, wherein the bioactive agents are mutually incompatible in a particular environment, for example, as hydrophobic and hydrophilic drugs are incompatible in either a polar or non-polar solvent. Different bioactive agents may also demonstrate incompatibility based on protic/aprotic solvents or ionic/non-ionic solvents. For example, the invention allows for the preparation of one set of biodegradable microparticles containing a hydrophobic drug and the preparation of another set of biodegradable microparticles containing a hydrophilic drug; the mixing of the two different sets of microparticles into a polymeric material used to form the matrix; and the disposing of the mixture on the surface of a substrate. Both hydrophobic and hydrophilic drugs can be released from the surface of the coated substrate at the same time as the biodegradable microparticles degrade, or the composition of the biodegradable microparticles or the natural biodegradable polysaccharide matrix can be altered so that one bioactive agent is released at a different rate or time than the other one.
- Biodegradable microparticles can be prepared having compositions that are suitable for either hydrophobic or hydrophilic drugs. For example, polymers such as polylactide or polycaprolactone can be useful for preparing biodegradable microparticles that include hydrophobic drugs; whereas polymers such as amylose or glycolide can be useful for preparing microparticles that include hydrophilic drugs.
- Traditional coating procedures directed at disposing at least two different types of bioactive agents have often required that the bioactive agents be put down separately. Traditional approaches may include the steps of solubilizing a hydrophobic drug in a non-polar solvent, coating the surface of the substrate with the non-polar mixture, drying the non-polar mixture, solubilizing the hydrophilic drug in a polar solvent, coating the layer of the dried non-polar mixture with the polar mixture, and then drying the polar mixture. This type of traditional coating process can be inefficient and can also result in undesirable surface properties (e.g., the layering of the drugs will cause one drug to be released before the other one is released). According to this aspect of the invention, the method of preparing surfaces having two, or more than two, different bioactive agents, in particular when the two different bioactive agents are released from the surface of the substrate, is a significant improvement over traditional methods of coating substrates and delivering bioactive agents from the surface of the substrates.
- Components of the biodegradable coating can be applied to the medical device using standard techniques to cover the entire surface of the device, or a portion of the device surface. As indicated, the components can be applied to the medical device independently or together, for example, in a composition. The coating formed on the device can be a single layer coating, or a multiple layer coating.
- Various factors can influence the delivery of bioactive agents from the coating. These include the concentration of the natural biodegradable polysaccharide and the extent of natural biodegradable polysaccharide coupling in the coating, the amount and location of biodegradable microparticles associated with the coating, the concentration of bioactive agent in the microparticles, and the presence of other coated layers, if included in the overall coating and the like. For example, the rate of delivery of the drug can be decreased by increasing the concentration of polymeric material or the relative amount of coupling or crosslinking of the polymeric material in the polymeric matrix or in the microparticle. Based on the description provided herein and the general knowledge in this technical area, one can alter properties of the coating to provide a desired release rate for one or more particular bioactive agents from the coating.
- Portions of the coating can be prepared to degrade at the same or different rates. For example, the biodegradable microparticles can be prepared or obtained to have a faster rate of degradation than the natural biodegradable polysaccharide matrix. In this case, the bioactive agent can be released into the natural biodegradable polysaccharide matrix and/or diffuse out of the natural biodegradable polysaccharide matrix.
- A natural biodegradable polysaccharide-based coating can be prepared by any one of a variety of methods. A “coating” as used herein can include one or more “coated layers”, each coated layer including one or more coating materials. In many cases, the coating consists of a single layer of material that includes the natural biodegradable polysaccharide, such as amylose or maltodextrin. In other cases, the coating includes more than one coated layer, at least one of the coated layers including the natural biodegradable polysaccharide. If more than one layer is present in the coating, the layers can be composed of the same or different materials. If multiple polymeric layers are provided on the surface, each individual layer of polymer can be chosen to provide a desired effect. Additionally, multiple layers of various bioactive agents can be deposited onto the medical device surface so that a particular bioactive agent can be presented to or released from the medical device at one time, one or more bioactive agents in each layer, which can be separated by polymeric material.
- If more than one coated layer is applied to a surface, it is typically applied successively. For example, a natural biodegradable polysaccharide coated layer can be formed by, for example, dipping, spraying, bushing, or swabbing the coating material on the article to form a layer, and then drying the coated layer. The process can be repeated to provide a coating having multiple coated layers, wherein at least one layer includes the natural biodegradable polysaccharide.
- Thus, in some embodiments wherein multiple coated layers are prepared, each coated layer is composed of the same materials. Alternatively, one or more of the coated layers is composed of materials that are different from one or more of the other layers. Additionally, multiple layers of various bioactive agents can be deposited onto the medical article surface so that a particular bioactive agent can be presented to or released from the medical article at one time, one or more bioactive agents in each layer, which can be separated by polymeric material.
- The invention also provides the advantage of maintaining excellent control over the formation of a coating on the surface of an article. To exemplify this aspect of the invention, an initiator is disposed on a surface of a medical article along with the natural biodegradable polysaccharide having pendent coupling groups. A bioactive agent can be disposed if desired. The initiator can be disposed in a mixture with the natural biodegradable polysaccharide together, or the initiator can be disposed independently. These compounds are generally disposed in a fluid state (for example, suspended or dissolved in an aqueous liquid) and can be disposed on an article surface using any one of number of techniques as described herein. After the initiator and natural biodegradable polysaccharide are both disposed, the initiator is activated, resulting in the activation of the pendent coupling groups, the coupling of natural biodegradable polysaccharide molecules, and the formation of the coating. The steps of disposing and activating can be performed in ways (as described herein and/or known in the art) to precisely control the formation of a coating. For example, the thickness and the location of the coating on the article surface can be controlled using techniques described herein and/or known in the art.
- In preferred aspects of the following methods, the natural biodegradable polysaccharide is selected from the group of amylose and maltodextrin. In other preferred aspects of the following methods, the natural biodegradable polysaccharide has a molecular weight of 500,000 Da or less, 250,000 Da or less, 100,000 Da or less, or 50,000 Da or less. It is also preferred that the natural biodegradable polysaccharides have an average molecular weight of 500 Da or greater. A particularly preferred size range for the natural biodegradable polysaccharides is in the range of about 1000 Da to about 10,000 Da.
- For example, in some aspects the method includes the steps of (i) disposing a composition comprising (a) a natural biodegradable polysaccharide having a coupling group, (b) an initiator, and (c) a bioactive agent on a surface; and (ii) activating the initiator to provide a coated composition having the natural biodegradable polysaccharide and the bioactive agent on the surface. This method can also be used to form a medical implant wherein the composition is disposed to form an implant of a desired configuration. For example, the composition can be disposed in a mold. This method can also be used to form an in situ formed matrix wherein the composition is disposed within a portion of a subject.
- In other aspects, the method includes the steps of (i) disposing an initiator on a surface, (ii) disposing a composition comprising (a) a natural biodegradable polysaccharide having a coupling group and (b) a bioactive agent on the surface; and (iii) activating the initiator to provide a coated composition having the natural biodegradable polysaccharide and the bioactive agent.
- During the step of activating, a composition including the natural biodegradable polysaccharide and the bioactive agent are contacted with the initiator and the initiator is activated to promote the crosslinking of two or more natural biodegradable polysaccharides via their coupling groups. In preferred aspects the natural biodegradable polysaccharide includes a polymerizable group, such as an ethylenically unsaturated group, and initiator is capable of initiating free radical polymerization of the polymerizable groups. Therefore, in another embodiment, the invention provides a method for coating a surface, including the steps of (i) disposing a composition comprising (a) a natural biodegradable polysaccharide having a ethylenically unsaturated group, (b) a polymerization initiator, and (c) a bioactive agent on a surface; and (ii) activating the polymerization initiator to cause the polymerization of the amylose compound thereby providing a coated composition having the natural biodegradable polysaccharide and the bioactive agent on the surface. These methods can also be used to form medical implants and in situ-formed matrices, wherein the composition is disposed in a mold or in a subject, respectively, rather than on a surface.
- In yet another aspect the invention provides a medical device having a coated composition comprising a plurality of coupled natural biodegradable polysaccharide and a bioactive agent.
- In some embodiments, the invention provides methods for preparing biodegradable coatings that include (a) a natural biodegradable polysaccharide having a coupling group and (b) biodegradable microparticles having a bioactive agent.
- In some embodiments the coupling group can be activated by an initiator. Therefore, the method can include the steps of (i) disposing an initiator on a surface, (ii) disposing a composition comprising (a) a natural biodegradable polysaccharide having a coupling group and (b) biodegradable microparticles comprising a bioactive agent; and (iii) activating the initiator to provide a biodegradable bioactive agent-releasing coated composition having the natural biodegradable polysaccharide and the biodegradable microparticles having the bioactive agent.
- In preferred aspects the natural biodegradable polysaccharide includes a polymerizable group, such as an ethylenically unsaturated group, and initiator is capable of initiating free radical polymerization of the polymerizable groups. Therefore, in another embodiment, the invention provides a method for coating a surface, including the steps of (i) disposing a composition comprising (a) a natural biodegradable polysaccharide having an ethylenically unsaturated group, (b) a polymerization initiator, and (c) biodegradable microparticles having a bioactive agent on a surface; and (ii) activating the polymerization initiator to cause the polymerization of the natural biodegradable polysaccharide thereby providing a coated composition that includes biodegradable microparticles in a natural biodegradable polysaccharide matrix. The invention also provides alternative methods for preparing a coated surface that is biodegradable and having microparticles that can release a bioactive agent. The methods include disposing in two or more steps at least the following reagents on a surface: (a) a natural biodegradable polysaccharide comprising a first coupling group (b) a natural biodegradable polysaccharide comprising a second coupling group that is reactive with the first coupling group, and (c) biodegradable microparticles that include a bioactive agent. According to this method reagents (a) and (b) are reactive with each other and are disposed separately on the surface but can individually include (c). For example, reagent (a) is first disposed on the surface and then a mixture comprising reagent (b) and (c) is then disposed on reagent (a). Reagent (a) reacts with (b) to link the natural biodegradable polysaccharide together to form a coating that includes (c), the biodegradable microparticles.
- The invention also provides methods for preparing biodegradable sealant coatings that include a natural biodegradable polysaccharide having a coupling group; optionally a bioactive agent can be included in the sealant coating.
- In some embodiments, the method includes the steps of (i) disposing a sealant composition comprising (a) a natural biodegradable polysaccharide having a coupling group, and (b) an initiator, and (ii) activating the initiator to form a sealant coating. This aspect of the invention includes coating methods where a bulk polymerization approach is performed. For example, in some embodiments, a composition including a polymerization initiator and natural biodegradable polysaccharides having a polymerizable group is disposed on a surface. The initiator is then activated to promote bulk polymerization and coupling of the natural biodegradable polysaccharides in association with the surface.
- In other aspects, the method includes the steps of (i) disposing an initiator on a surface, (ii) disposing a natural biodegradable polysaccharide having a coupling group; and (iii) activating the initiator to provide a coated composition having the amylose polymer. The natural biodegradable polysaccharides can be disposed on the surface along with other reagents if desired. This aspect of the invention includes coating methods where a graft polymerization approach is performed. For example, in some embodiments, a polymerization initiator is first disposed on a surface and then a natural biodegradable polysaccharide having a polymerizable group is disposed on the surface having the initiator. The initiator is activated to promote free radical polymerization, and coupling of the natural biodegradable polysaccharides from the surface. In other embodiments of the invention, an aqueous composition that includes the natural biodegradable polysaccharide having the coupling group and a bioactive agent is obtained and used in the method of providing a sealant coating to a surface. This composition can be prepared by mixing the natural biodegradable polysaccharide with a bioactive agent, for example, a water-soluble small molecule, a protein, or a nucleic acid. In one preferred aspect of the invention, the bioactive agent is a procoagulant or prothrombotic factor. For example, the bioactive agent can be a protein such as recombinant collagen, or other proteins that associate with receptors on platelets to induce platelet aggregation.
- In some aspects of the invention, the coating is placed in contact with an aqueous solution, or the materials of the coating composition. The coating or coating materials are designed to be stable in the presence of the aqueous solution provided that an enzyme that causes the degradation of the natural biodegradable polysaccharide (or another degrading agent) is not present in an amount sufficient to cause substantial degradation of the materials.
- For example, the invention provides a shelf stable composition comprising a natural biodegradable polysaccharide comprising coupling groups. These compositions could be obtained or prepared, according to the details provided herein, and then stored for a period of time before the composition is used to form a biodegradable coating, without the significant degradation of the natural biodegradable polysaccharide occurring during storage.
- Accordingly, the invention also provides methods for preparing a biodegradable coating comprising preparing a biodegradable coating composition comprising a natural biodegradable polysaccharide comprising coupling group; storing the coating composition for an amount of time; and then using the coating composition to prepare a biodegradable coating. Optionally, one or more bioactive agents and/or microparticles can be added before or after storage of the coating composition.
- In a related aspect, the invention also provides the advantage of being able to perform synthetic and post-synthetic procedures wherein the natural biodegradable polysaccharide is contacted with an aqueous composition, and there is minimal risk of degradation of the polysaccharide. For example, the natural biodegradable polysaccharide may be contacted with an aqueous solution for purification without risking significant degradation of the natural biodegradable polysaccharide.
- In yet another aspect, the invention relates to the stability of the coatings that are formed on an article. The invention provides a method comprising obtaining an article having a coating comprising a natural biodegradable polysaccharide, and then contacting the article with an aqueous solution. The aqueous solution can be, for example, a storage solution, a solution that is used to hydrate the surface of the coated device, or an aqueous sterilization solution.
- In some aspects the coating can be contacted with an aqueous sterilization solution. Medical articles, or parts of medical articles, can be prepared having a coating and these articles can be treated to sterilize one or more parts of the article, or the entire medical article. Sterilization can take place prior to using the medical article and/or, in some cases, during implantation of the medical article.
- In some aspects, the invention provides a method for delivering a bioactive agent from a biodegradable coating or a biodegradable article by exposing the coating or article to an enzyme that causes the degradation of the coating. In performing this method a coated article, such as an implantable medical device is provided to a subject. The coated article has a biodegradable coating comprising a natural biodegradable polysaccharide having pendent coupling groups, wherein the coating is formed on a surface of the article by reaction of the coupling groups to form a crosslinked matrix of a plurality of natural biodegradable polysaccharides, and wherein the coating includes a bioactive agent. The coating or article is then exposed to a carbohydrase that can promote the degradation of the biodegradable coating.
- The carbohydrase that contacts the coating or article can specifically degrade the natural biodegradable polysaccharide causing release of the bioactive agent. Examples of carbohydrases that can specifically degrade natural biodegradable polysaccharide coatings include α-amylases, such as salivary and pancreatic α-amylases; disaccharidases, such as maltase, lactase and sucrase; trisaccharidases; and glucoamylase(amyloglucosidase).
- Serum concentrations for amylase are estimated to be in the range of about 50-100 U per liter, and vitreal concentrations also fall within this range (Varela, R. A., and Bossart, G. D. (2005) J Am Vet Med Assoc 226:88-92).
- In some aspects, the carbohydrase can be administered to a subject to increase the local concentration, for example in the serum or the tissue surrounding the implanted device, so that the carbohydrase may promote the degradation of the coating. Exemplary routes for introducing a carbohydrase include local injection, intravenous (IV) routes, and the like. Alternatively, degradation can be promoted by indirectly increasing the concentration of a carbohydrase in the vicinity of the coated article, for example, by a dietary process, or by ingesting or administering a compound that increases the systemic levels of a carbohydrase.
- In other cases, the carbohydrase can be provided on a portion of the coated article. For example the carbohydrase may be eluted from a portion of the article that does not have the natural biodegradable polymer coating. In this aspect, as the carbohydrase is released it locally acts upon the coating to cause its degradation and promote the release of the bioactive agent. Alternatively, the carbohydrase can be present in a microparticle in one or more portions the coating. As the carbohydrase is released from the microparticle, it causes coating degradation and promote the release of the bioactive agent.
- In another aspect, the invention sets forth methods for providing lubricity to an article surface. The matrix of biodegradable polysaccharides as described herein can provide a surprisingly lubricious and durable surface when formed, for example, on the surface of a device.
- As used herein, the term “lubricity” refers to a characterization of the frictional force associated with a coating. A lubricious coating can reduce the frictional forces present on the surface of the device when another surface is moved against the device surface. For example, a catheter having a coating that provides improved lubricity will encounter less frictional resistance when moved within a portion of the body, as compared to an uncoated substrate, or a coating that is not lubricious. Lubricity can also be important for devices with inner moving parts in addition to devices that function along with another device, for example, a coronary catheter which guides the insertion of a PTCA catheter. The methods can be used to prepare lubricious coatings for short term use and/or single use devices.
- Improved lubricity can be shown by one or more methods. One method of testing lubricity of a coating is by the horizontal sled style friction test method (such as ASTM D-1894; a modified test is described herein). The lubricity measurements described herein refer to the kinetic coefficient of friction, which is equal to the average force reading obtained during uniform sliding of the surfaces divided by the sled weight. The measurements are in grams.
- As shown herein, the biodegradable polysaccharide coatings generally show a lubricity of 20 g or less, and coatings having a lubricity of 15 g or less, or 10 g or less can be prepared by the methods described herein. In one desirable method for preparing the coating, a biodegradable polysaccharide matrix is formed using a photoinitiator to promote association of the biodegradable polysaccharides and matrix formation.
- Another method which can be used to demonstrate an improvement in lubricity is the water contact angle measurement method. Reduction of water contact angle is indicative of increased wettability, which associates with an improvement in lubricity.
- Also, in many aspects, the biodegradable coating of the invention demonstrates excellent durability. As used herein, the term “durability” refers to the wear resistance of the biodegradable polysaccharide coating. A more durable coating is less easily removed from a substrate by abrasion. Durability of a coating can be assessed by subjecting the device to conditions that simulate use conditions. The durability of the coating compositions is demonstrated by the ability to adhere to the device surface sufficiently to withstand the effect of shear forces encountered during insertion and/or removal of the device, which could otherwise result in delamination of the coating from the body member.
- Improved durability can be shown by one or more methods. One preferred method of testing durability, as well as lubricity, is by the horizontal sled style friction test method described herein. In the method multiple push-pull cycles are performed and friction measurements are taken during the measurements. If minimal increase in friction values is seen after multiple push pull cycles, the coating is though to have good durability.
- The biodegradable coatings of the present invention showed excellent durability. For example, the lubricity of the fifth push and pull cycle generally was not greater than 10% or more typically not greater than 5% of the lubricity of the first push and pull cycle. At later points, for example, at the fortieth push and pull cycle, the lubricity generally was not greater than 20% or more typically not greater than 10% of the lubricity of the first push and pull cycle.
- The invention will be further described with reference to the following non-limiting Examples. It will be apparent to those skilled in the art that many changes can be made in the embodiments described without departing from the scope of the present invention. Thus the scope of the present invention should not be limited to the embodiments described in this application, but only by embodiments described by the language of the claims and the equivalents of those embodiments. Unless otherwise indicated, all percentages are by weight.
- Amylose having polymerizable vinyl groups was prepared by mixing 0.75 g of amylose (A0512; Aldrich) with 100 mL of methylsulfoxide (J T Baker) in a 250 mL amber vial, with stirring. After one hour, 2 mL of triethylamine (TEA; Aldrich) was added and the mixture was allowed to stir for 5 minutes at room temperature. Subsequently, 2 mL of glycidyl acrylate(Polysciences) was added and the amylose and glycidyl acrylate were allowed to react by stirring overnight at room temperature. The mixture containing the amylose-glycidyl acrylate reaction product was dialyzed for 3 days against DI water using continuous flow dialysis. The resultant acrylated-amylose (0.50 g; 71.4% yield) was then lyophilized and stored desiccated at room temperature with protection from light.
- A polymerization initiator was prepared by copolymerizing a methacrylamide having a photoreactive group with acrylamide.
- A methacrylamide-oxothioxanthene monomer(N-[3-(7-Methyl-9-oxothioxanthene-3-carboxamido)propyl]methacrylamide (MTA-APMA)) was first prepared. N-(3-aminopropyl)methacrylamide hydrochloride (APMA), 4.53 g (25.4 mmol), prepared as described in U.S. Pat. No. 5,858,653, Example 2, was suspended in 100 mL of anhydrous chloroform in a 250 mL round bottom flask equipped with a drying tube. 7-methyl-9-oxothioxanthene-3-carboxylic acid (MTA) was prepared as described in U.S. Pat. No. 4,506,083, Example D. MTA-chloride (MTA-Cl) was made as described in U.S. Pat. No. 6,007,833, Example 1. After cooling the slurry in an ice bath, MTA-Cl (7.69 g; 26.6 mmol) was added as a solid with stirring to the APMA-chloroform suspension. A solution of 7.42 mL (53.2 mmol) of TEA in 20 mL of chloroform was then added over a 1.5 hour time period, followed by a slow warming to room temperature. The mixture was allowed to stir 16 hours at room temperature under a drying tube. After this time, the reaction was washed with 0.1 N HCl and the solvent was removed under vacuum after adding a small amount of phenothiazine as an inhibitor. The resulting product was recrystallized from tetrahydrofuran (THF)/toluene (3/1) and gave 8.87 g (88.7% yield) of product after air drying. The structure of MTA-APMA was confirmed by NMR analysis.
- MTA-APMA was then copolymerized with acrylamide in DMSO in the presence of 2-mercaptoethanol (a chain transfer agent), N,N,N′,N′-tetramethyl-ethylenediamine (a co-catalyst), and 2,2′-azobis(2-methyl-propionitrile) (a free radical initiator) at room temperature. The solution was sparged with nitrogen for 20 minutes, sealed tightly, and incubated at 55° C. for 20 hours. The solution was dialyzed for 3 days against DI water using continuous flow dialysis. The resultant MTA-PAAm was lyophilized, stored desiccated, and protected from light at room temperature.
- 100 mg of acrylated-amylose as prepared in Example 1 was placed in an 8 mL amber vial. To the acrylated-amylose was added 3 mg of MTA-PAAm (lyophilized), 2 μL of 2-NVP (N-vinyl-2-pyrrolidone; accelerant (Bimax)) and 1 mL of 1× phosphate-buffered saline (1×PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 50 μL was placed onto a glass slide (2991FI; Esco) and illuminated for 50 seconds with an
EFOS 100 SS illumination system equipped with a 400-500 nm filter (50 mW/cm2). After illumination the polymer was found to form a semi-firm gel having elastomeric properties. - 4-Methylbenzophenone (750 g; 3.82 moles) was added to a 5 liter Morton flask equipped with an overhead stirrer and dissolved in 2850 mL of benzene. The solution was then heated to reflux, followed by the dropwise addition of 610 g (3.82 moles) of bromine in 330 mL of benzene. The addition rate was approximately 1.5 mL/min and the flask was illuminated with a 90 watt (90 joule/sec) halogen spotlight to initiate the reaction. A timer was used with the lamp to provide a 10% duty cycle (on 5 seconds, off 40 seconds), followed in one hour by a 20% duty cycle (on 10 seconds, off 40 seconds). At the end of the addition, the product was analyzed by gas chromatography and was found to contain 71% of the desired 4-bromomethylbenzophenone, 8% of the dibromo product, and 20% unreacted 4-methylbenzophenone. After cooling, the reaction mixture was washed with 10 g of sodium bisulfite in 100 mL of water, followed by washing with 3×200 mL of water. The product was dried over sodium sulfate and recrystallized twice from 1:3 toluene:hexane. After drying under vacuum, 635 g of 4-bromomethylbenzophenone was isolated, providing a yield of 60%, having a melting point of 112° C.-114° C. Nuclear magnetic resonance (“NMR”) analysis (1H NMR (CDCl3)) was consistent with the desired product: aromatic protons 7.20-7.80 (m, 9H) and methylene protons 4.48 (s, 2H). All chemical shift values are in ppm downfield from a tetramethylsilane internal standard.
- N,N,N′,N′-Tetramethylethylenediamine (6 g; 51.7 mmol) was dissolved in 225 mL of chloroform with stirring. BMBP (29.15 g; 106.0 mmol), as described in Example 4, was added as a solid and the reaction mixture was stirred at room temperature for 72 hours. After this time, the resulting solid was isolated by filtration and the white solid was rinsed with cold chloroform. The residual solvent was removed under vacuum and 34.4 g of solid was isolated for a 99.7% yield, melting point 218° C.-220° C. Analysis on an NMR spectrometer was consistent with the desired product: 1H NMR (DMSO-d6) aromatic protons 7.20-7.80 (m, 18H), benzylic methylenes 4.80 (br. s, 4H), amine methylenes 4.15 (br. s, 4H), and methyls 3.15 (br. s, 12H).
- Acrylated-amylose (100 mg), as described in Example 1, was placed in an 8 mL amber vial. Ethylenebis(4-benzoylbenzyldimethylammonium)dibromide (3 mg), as described in Example 5, 2 μl of 2-NVP, and 1 mL of 1× phosphate buffered saline (1×PBS) was added to the acrylated-amylose and mixed for two hours on a shaker at 37° C. The mixture (250 μl) was spread onto a 3 cm×2 cm polyethylene terephthalate (PET) mesh substrate (41 μm monofil diameter; Goodfellow Cambridge Ltd., UK). The PET substrate with the applied amylose mixture was placed in a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 15 cm from the light source, and illuminated for 60 seconds. After illumination, the applied amylose mixture was found to form a semi-firm gel on the PET substrate, with elastomeric properties evident.
- A maleimide functional acid was prepared in the following manner, and was used in Example 8. EACA (6-aminocaproic acid), (100 g; 0.762 moles), was dissolved in 300 mL of acetic acid in a three-neck, three liter flask equipped with an overhead stirrer and drying tube. Maleic anhydride, (78.5 g; 0.801 moles), was dissolved in 200 mL of acetic acid and added to the EACA solution. The mixture was stirred one hour while heating on a boiling water bath, resulting in the formation of a white solid. After cooling overnight at room temperature, the solid was collected by filtration and rinsed two times with 50 mL of hexane each rinse. After drying, the yield of the (z)-4-oxo-5-aza-undec-2-endioic acid (Compound 1) was in the range of 158-165 g (90-95%) with a melting point of 160-165° C. Analysis on an NMR spectrometer was consistent with the desired product: 1H NMR (DMSO-d6, 400 MHz) δ 6.41, 6.24 (d, 2H, J=12.6 Hz; vinyl protons), 3.6-3.2 (b, 1H; amide proton), 3.20-3.14 (m, 2H: methylene adjacent to nitrogen), 2.20 (t, 2H, J=7.3; methylene adjacent to carbonyl), 1.53-1.44 (m, 4H; methylenes adjacent to the central methylene), and 1.32-1.26 (m, 2H; the central methylene).
- (z)-4-oxo-5-aza-undec-2-endioic acid, (160 g; 0.698 moles), zinc chloride, 280 g (2.05 moles), and phenothiazine, 0.15 g were added to a two liter round bottom flask fitted with an overhead stirrer, condenser, thermocouple, addition funnel, an inert gas inlet, and heating mantle. Chloroform (CHCl3), 320 mL was added to the 2 liter reaction flask, and stirring of the mixture was started. Triethylamine (480 mL; 348 g, 3.44 moles (TEA)) was added over one hour. Chlorotrimethyl silane (600 mL; 510 g, 4.69 moles) was then added over two hours. The reaction was brought to reflux and was refluxed overnight (˜16 hours). The reaction was cooled and added to a mixture of CHCl3 (500 mL), water (1.0 liters), ice (300 g), and 12 N hydrochloric acid (240 mL) in a 20 liter container over 15 minutes. After 15 minutes of stirring, the aqueous layer was tested to make sure the pH was less than 5. The organic layer was separated, and the aqueous layer was extracted three times with CHCl3 (700 mL) each extraction. The organic layers were combined and evaporated on a rotary evaporator. The residue was then placed in a 20 liter container. A solution of sodium bicarbonate (192 g) in water (2.4 liters) was added to the residue. The bicarbonate solution was stirred until the solids were dissolved. The bicarbonate solution was treated with a solution of hydrochloric acid, (26 liters of 1.1 N) over 5 minutes to a pH of below 2. The acidified mixture was then extracted with two portions of CHCl3, (1.2 liters and 0.8 liters) each extraction. The combined extracts were dried over sodium sulfate and evaporated. The residue was recrystallized from toluene and hexane. The crystalline product was then isolated by filtration and dried which produced 85.6 g of white N-(6-oxo-6-hydroxyhexyl)maleimide (Mal-EACA; Compound 2). Analysis on an NMR spectrometer was consistent with the desired product: 1H NMR (CDCl3, 400 MHz) δ 6.72 (s, 2H; maleimide protons), 3.52 (t, 2H, J=7.2 Hz; methylene next to maleimide), 2.35 (t, 2H, J=7.4; methylene next to carbonyl), 1.69-1.57 (m, 4H; methylenes adjacent to central methylene), and 1.39-1.30 (m, 2H; the central methylene). The product had a DSC (differential scanning calorimator) melting point peak at 89.9° C.
- Mal-EACA from Example 7 (5.0 g; 23.5 mmole) and CHCl3 (25 mL) were placed in a 100 mL round bottom flask and stirred using a magnetic bar with cooling in an ice bath. Oxalyl chloride (10.3 mL; ˜15 g; 118 mmole) was added and the reaction was brought to room temperature with stirring overnight. The volatiles were removed on a rotary evaporator, and the residue was azetroped with three times with 10 mL CHCl3 each time. The intermediate Mal-EAC-Cl [N-(6-oxo-6-chlorohexyl)maleimide] (Compound 3) was dissolved in acetone (10 mL) and added to a cold (ice bath) stirred solution of sodium azide (2.23 g; 34.3 mmole) in water (10 mL). The mixture was stirred one hour using an ice bath. The organic layer was set aside in an ice bath, and the aqueous layer was extracted three times with 10 mL CHCl3. All operations of the acylazide were done at ice bath temperatures. The combined organic solutions of the azide reaction were dried for an hour over anhydrous sodium sulfate. The N-(6-oxo-6-azidohexyl)maleimide (Compound 4) solution was further dried by gentle swirling over molecular sieves over night. The cold azide solution was filtered and added to refluxing CHCl3, 5 mL over a 10 minute period. The azide solution was refluxed for 2 hours. The weight of Mal-C5-NCO (Compound 5) solution obtained was 55.5 g, which was protected from moisture. A sample of the isocyanate solution, 136 mg was evaporated and treated with DBB (1,4-dibromobenzene), 7.54 mg and chloroform-d, 0.9 mL: 1H NMR (CDCl3, 400MHz) δ 6.72 (s,2H), 3.55 (t, 2H, J=7.2 Hz), 3.32 (t, 2H, J=6.6 Hz), 1.70-1.59 (m, 4H), 1.44-1.35 (m, 2H). The NMR spectra was consistent with desired product. The DBB internal standard δ at 7.38 (integral value was 2.0, 4H; per mole of product) was used to estimate the moles of Mal-C5-NCO in solution. The calculated amount of product in solution was 23.2 mmole for a yield of 98% of theory. NCO reagent (concentration was 0.42 mmole/g) was used to prepare a macromer in Example 14.
- Acrylic acid (100 g; 1.39 mole) and phenothiazine (0.1 g) were placed in a 500 mL round bottom flask. The reaction was stirred at 92° C. for 14 hours. The excess acrylic acid was removed on a rotary evaporator at 25° C. using a mechanical vacuum pump. The amount of residue obtained was 51.3 g. The CEA (Compound 6) was used in Example 10 without purification.
- CEA from Example 9 (51 g; ˜0.35 mole) and dimethyl formamide (DMF; 0.2 mL; 0.26 mmole) were dissolved in CH2Cl3 (100 mL). The CEA solution was added slowly (over 2 hours) to a stirred solution of oxalyl chloride (53 mL; 0.61 mole), DMF (0.2 mL; 2.6 mmole), anthraquinone (0.5 g; 2.4 mmole), phenothiazine (0.1 g, 0.5 mmole), and CH2Cl3 (75 mL) in a 500 mL round bottom flask in an ice bath at 200 mm pressure. A dry ice condenser was used to retain the CH2Cl3 in the reaction flask. After the addition was complete the reaction was stirred at room temperature overnight. The weight of reaction solution was 369 g. A sample of the CEA-Cl (Compound 7) reaction solution (124 mg) was treated with 1,4-dibromobenzene (DBB, 6.85 mg) evaporated and dissolved in CDCl3: 1H NMR (CDCl3, 400 MHz) δ 7.38 (s, 4H; DBB internal std.), 6.45 (d, 1H, J=17.4 Hz), 6.13 (dd, 1H, J=17.4, 10.4 Hz), 5.90 (d, 1H, J=10.4 Hz), 4.47 (t, 2H, J=5.9 Hz), 3.28 (t, 2H, J=5.9). The spectra was consistent with the desired product. There was 0.394 mole DBB for 1.0 mole CEA-Cl by integration, which gave a calculated yield of 61%. Commercially available CEA (426 g; Aldrich) was reacted with oxalyl chloride (532 mL) in a procedure similar to the one listed above. The residue CEA-Cl (490 g) was distilled using an oil bath at 140° C. at a pressure of 18 mm Hg. The distillate temperature reached 98° C. and 150 g of distillate was collected. The distillate was redistilled at 18 mm Hg at a maximum bath temperature of 120° C. The temperature range for the distillate was 30° C. to 70° C. which gave 11 g of material. The distillate appeared to be 3-chloro-3-oxopropyl 3-chloropropanoate. The residue of the second distillation (125 g; 26% of theory) was used in Example 11.
- CEA-Cl from Example 10 (109.2 g; 0.671 mole) was dissolved in acetone (135 mL). Sodium azide (57.2 g; 0.806 mole) was dissolved in water (135 mL) and chilled. The CEA-Cl solution was then added to the chilled azide solution with vigorous stirring in an ice bath for 1.5 hours. The reaction mixture was extracted two times with 150 mL of CHCl3 each extraction. The CHCl3 solution was passed through a
silica gel column 40 mm in diameter by 127 mm. The 3-azido-3-oxopropyl acrylate (Compound 8) solution was gently agitated over dried molecular sieves at 4° C. overnight. The dried solution was used in Example 12 without purification. - The dried azide solution (from Example 11) was slowly added to refluxing CHCl3, 75 mL. After the addition was completed, refluxing was continued 2 hours. The EA-NCO (Compound 9) solution (594.3 g) was protected from moisture. A sample of the EA-NCO solution (283.4 mg) was mixed with DBB (8.6 mg) and evaporated. The residue was dissolved in CDCl3: 1H NMR (CDCl3, 400 MHz) δ 7.38 (s, 4H; DBB internal std.), 6.50 (d, 1H, J=17.3 Hz), 6.19 (dd, 1H, J=17.3, 10.5 Hz), 5.93 (d, 1H, J=10.5 Hz), 4.32 (t, 2H, J=5.3 Hz), 3.59 (t, 2H, J=5.3). The spectra was consistent with the desired EA-NCO. There was 0.165 mole DBB for 1.0 mole EA-NCO by integration, which gave a calculated concentration of 110 mg EA-NCO/g of solution. The EA-NCO solution was used to prepare a macromer in Example 13.
- Maltodextrin (MD; Aldrich; 9.64 g; ˜3.21 mmole; DE (Dextrose Equivalent): 4.0-7.0) was dissolved in dimethylsulfoxide (DMSO) 60 mL. The size of the maltodextrin was calculated to be in the range of 2,000 Da-4,000 Da. A solution of EA-NCO from Example 12 (24.73 g; 19.3 mmole) was evaporated and dissolved in dried DMSO (7.5 mL). The two DMSO solutions were mixed and heated to 55° C. overnight. The DMSO solution was placed in dialysis tubing (1000 MWCO, 45 mm flat width×50 cm long) and dialyzed against water for 3 days. The macromer solution was filtered and lyophilized to give 7.91 g white solid. A sample of the macromer (49 mg), and DBB (4.84 mg) was dissolved in 0.8 mL DMSO-d6: 1H NMR (DMSO-d6, 400 MHz) δ 7.38 (s, 4H; internal std. integral value of 2.7815), 6.50, 6.19, and 5.93 (doublets, 3H; vinyl protons integral value of 3.0696). The calculated acrylate load of macromer was 0.616 μmoles/mg of polymer.
- A procedure similar to Example 13 was used to make the MD-Mal macromer. A solution of Mal-C5-NCO from Example 8 (0.412 g; 1.98 mmole) was evaporated and dissolved in dried DMSO (2 mL). MD (0.991 g; 0.33 mmole) was dissolved in DMSO (5 mL). The DMSO solutions were combined and stirred at 55° C. for 16 hours. Dialysis and lyophilization gave 0.566 g product. A sample of the macromer (44 mg), and DBB (2.74 mg) was dissolved in 00.8 mL DMSO-d6: 1H NMR (DMSO-d6, 400 MHz) δ 7.38 (s, 4H; internal std. integral value of 2.3832), 6.9 (s, 2H; Maleimide protons integral value of 1.000). The calculated acrylate load of macromer was 0.222 μmoles/mg of polymer. The macromer was tested for its ability to make a matrix (see Example 17)
- 250 mg of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 3 mg of MTA-PAAm (lyophilized), 2 μL of 2-NVP, and 1 mL of 1× phosphate-buffered saline (1×PBS), providing a composition having MD-Acrylate at 250 mg/mL. The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 50 μL was placed onto a glass slide and illuminated for 40 seconds with an
EFOS 100 SS illumination system equipped with a 400-500 nm filter. After illumination the polymer was found to form a semi-firm gel having elastomeric properties. - 250 mg of MD-acrylate as prepared in Example 13 was placed in an 8mL amber vial. To the MD-Acrylate was added 14 mg of camphorquinone-10-sulfonic acid hydrate (Toronto Research Chemicals, Inc.), 3 μL of 2-NVP, and 1 mL of distilled water. The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 50 μL was placed onto a glass slide and illuminated for 40 seconds with a SmartliteIQ™ LED curing light (Dentsply Caulk). After illumination the polymer was found to form a semi-firm gel having with elastomeric properties.
- 250 mg of MD-Mal as prepared in Example 14 was placed in an 8 mL amber vial. To the MD-Mal was added 3 mg of MTA-PAAm (lyophilized), 2 μL of 2-NVP, and 1 mL of 1× phosphate-buffered saline (1×PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 50 μL was placed onto a glass slide and illuminated for 40 seconds with an
EFOS 100 SS illumination system equipped with a 400-500 nm filter. After illumination the polymer was found to form a semi-firm gel having elastomeric properties. - 100 mg photo-derivatized poly(vinylpyrrolidone) (photo-PVP) as prepared as described in U.S. Pat. No 5,637,460, and the photoinitiator tetrakis (4-benzoylbenzyl ether) of pentaerythritol [“tetra-BBE-PET”] (5 mg), prepared as described in U.S. Pat. No. 5,414,075 (Example 1) and commercially available from SurModics, Inc. (Eden Prairie, Minn.) as PR01, were mixed with 10 mL isopropyl alcohol (IPA; Fisher) for 1 minute. The mixture in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR). A 1.2 cm PEBAX® rod (Medical Profiles, Inc) was dipped into the solution for 10 seconds, at a dip rate of 0.1 cm/second, and then removed at the same rate. The rod was allowed to air dry for 5 minutes. The rod was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 30 cm from light source, illuminated for 180 seconds, and then removed.
- 300 mg of MD-Acrylate, as prepared in Example 13, was placed in an 8 mL amber vial. To the MD-Acrylate was added 4,5-bis(4-benzoylphenylmethyleneoxy)benzene-1,3-disulfonic acid (5 mg) (DBDS), prepared as described in U.S. Pat. No. 6,278,018 (Example 1) and commercially available from SurModics, Inc. (Eden Prairie, Minn.) as DBDS, and 1 mL of 1× phosphate-buffered saline (1×PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR). The photo-PVP/tetra-BBE-PET coated PEBAX® rod was dipped into the mixture for 30 seconds, at a dip rate of 0.3 cm/s, and then removed at the same rate. The rod was immediately placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 30 cm from light source, and illuminated for 180 seconds and then removed.
- The MD-Acrylate coated rod was examined under Scanning Electron Microscope (SEM;
LEO Supra 35 VP); the MD-Acrylate coating thickness varied from 2.1 μm to 2.5 μm, with an average coating thickness of 2.3 μm. - 500 mg of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 3 mg of MTA-PAAm (lyophilized), 2 μL of 2-NVP, and 1 mL of 1× phosphate-buffered saline (1×PBS). The reagents were then mixed for one hour on a shaker at 37° C. To this mixture was added either 5 mg 70 kD FITC-Dextran or 5
mg 10 kD FITC-Dextran (Sigma) and vortexed for 30 seconds. The mixture in an amount of 200 μL was placed into a Teflon well plate (8 mm diameter, 4 mm deep) and illuminated for 40 seconds with anEFOS 100 SS illumination system equipped with a 400-500 nm filter. The formed matrix was loose, and not as well crosslinked as the formed MD-acrylate matrix in Example 17. After illumination, the matrix was transferred to a 12 well plate (Falcon) and placed in a well containing 0.6 mL PBS. At daily intervals for 6 days, 150 μL of PBS was removed from each well and placed into a 96 well plate. The remaining 850 μL were removed from the samples, and replaced with 1 mL fresh PBS. The 96 well plate was analyzed for FITC-Dextran on a spectrophotometer (Shimadzu) at 490 absorbance. Results showed that at least 70% of the detectable 10 kd or 70 kD FITC-Dextran was released from the matrix after 2 days. Visual observation showed that an unquantified amount of 10 kD or 70 kD FITC-Dextran remained within the matrix after 6 days. - A MD-Acrylate-coated PEBAX rod (from Example 18) was placed in 5 mL of 1× phosphate-buffered saline (PBS) containing 24 μg alpha-Amylase (Sigma; catalog # A6814) for 7 days on a rotating plate at 37° C. After 7 days, the rod was removed from the PBS and washed with distilled water. The rod was then examined under a Scanning Electron Microscope (
LEO Supra 35 VP); upon examination, no trace of the MD-Acrylate coating was detected. As a control, a MD-Acrylate-coated PEBAX was placed in 1× phosphate-buffered saline (PBS) without alpha-Amylase; upon examination, the MD-Acrylate coating was intact and showed no signs of degradation. - Polyalditol (PA; GPC; 9.64 g; ˜3.21 mmole) was dissolved in dimethylsulfoxide (DMSO) 60 mL. The size of the polyalditol was calculated to be in the range of 2,000 Da-4,000 Da. A solution of EA-NCO from Example 12 (24.73 g; 19.3 mmole) was evaporated and dissolved in dried DMSO (7.5 mL). The two DMSO solutions were mixed and heated to 55° C. overnight. The DMSO solution was placed in dialysis tubing (1000 MWCO, 45 mm flat width×50 cm long) and dialyzed against water for 3 days. The polyalditol macromer solution was filtered and lyophilized to give 7.91 g white solid. A sample of the macromer (49 mg), and DBB (4.84 mg) was dissolved in 0.8 mL DMSO-d6: 1H NMR (DMSO-d6, 400 MHz) δ 7.38 (s, 4H; internal std. integral value of 2.7815), 6.50, 6.19, and 5.93 (doublets, 3H; vinyl protons integral value of 3.0696). The calculated acrylate load of macromer was 0.616 μmoles/mg of polymer.
- 1,100 milligrams of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 1 mg of a
photoinitiator 4,5-bis(4-benzoylphenyl-methyleneoxy)benzene-1,3-disulfonic acid (5 mg) (DBDS) and 1 mL of 1× phosphate-buffered saline (PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 uL was injected, using a 23 gauge needle, into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.). The tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm, which indicated complete polymerization of the MD-Acrylate. No excess liquid was observed. The filament was manipulated with forceps. Maltodextrin filaments were also made from a MD-acrylate solution having concentration of 200 mg/mL. These are physically firm and same as 1,100 mg/ml. - 1,500 milligrams of polyalditol-acrylate as prepared in Example 21 was placed in an 8 ml amber vial. To the polyalditol-acrylate was added 1 mg of DBDS (lyophilized), 15 mg Bovine Serum Albumin, and 200 uL of 1× phosphate-buffered saline (PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 uL was injected, using a 23 gauge needle, into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.). The tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm, which indicated complete polymerization of the polyalditol-acrylate. No excess liquid was observed. The filament was manipulated with forceps
- Maltodextrin-acrylate filaments were synthesized using 200 mg/mL and 1100 mg/mL MD-acrylate as described in example 22 and were tested for degradation in Amylase solutions. These filaments were placed in microcentrifuge tubes containing 1 mL of either 1×PBS (control), 1×PBS containing alpha-Amylase at 0.121 μg/mL (Sigma; catalog # A6814), or 1×PBS containing alpha-Amylase at 24 μg/mL. The tubes were then placed in an incubator at 37° C.
- After 2 days in the PBS with the 0.121 μg/mL alpha-Amylase solution the 200 mg/mL filament was completely degraded, and no trace of the filament was observable. The 200 mg/mL filament in PBS (control) showed no signs of degradation.
- After 33 days in the 1×PBS containing alpha-Amylase at 0.121 μg/mL, the 1100 mg/mL filament had lost some of its initial firmness (as noted by the slightly curled appearance of the filament), but was still completely intact. The 1,100 mg/mL filament in the PBS with 24 ug Amylase had completely degraded after 48 hours. The 1,100 mg/ml filament in the PBS showed no signs of degradation.
- MD-Acrylate in an amount of 1,100 milligrams of as prepared in Example 13 was placed in an 8 ml amber vial. To the MD-Acrylate was added 1 mg of DBDS (lyophilized), 15 mg Bovine Serum Albumin (representing the bioactive agent; and 1 mL of 1× phosphate-buffered saline (1×PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 uL was injected, using a 23 gauge needle, into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.). The tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm, which indicated complete polymerization of the MD-Acrylate. No excess liquid was observed.
- The filament was placed in a 1.7 ml microcentrifuge tube with 1
ml 1×PBS. At daily intervals for 6 days, 150 μL of PBS was removed from each well and placed into a 96 well plate for subsequent analysis. The remaining 850 μL was removed from the sample, and to the tube was added 1 ml of 1×PBS. After 6 days, the filament was placed in a 1.7 ml microcentrifuge tube with 1×PBS containing alpha-Amylase at 0.121 μg/mL. At daily intervals for 35 days, 150 μL of PBS was removed from each well and placed into a 96 well plate for subsequent analysis. The remaining 850 μL was removed from the sample, and to the tube was added 1 ml of fresh 1×PBS containing alpha-Amylase at 0.121 μg/mL. The 96-well plate was analyzed for BSA using the Quanitpro Assay Kit (Sigma). For the first 6 days, there was an initial burst of BSA, followed by a very slow release. After the addition of PBS+Amylase, the rate of BSA release significantly increase, and was relatively constant over the next 35 days. Results are shown in Table 2 andFIG. 1 .TABLE 2 Cumulative BSA release (% Timepoint of Total BSA) 1 4.8 2 5.35 3 5.7 4 5.98 5 6.19 6 6.36 7 9.46 8 10.7 9 11.82 10 12.94 11 14.01 12 15.06 13 16.11 14 17.23 15 18.11 16 19.04 17 19.92 18 21.26 19 22.15 20 23.04 21 24.06 22 25.35 23 26.31 24 26.91 25 27.51 26 28.63 27 29.19 28 29.75 29 30.44 30 31.11 31 31.43 32 31.63 33 31.83 34 32.07 35 32.31 36 32.72 37 32.95 38 33.27 39 33.83 40 34.15 41 34.43 42 34.71 - Polyaldtiol-acrylate in an amount of 1,500 mg of as prepared in Example 21 was placed in an 8 ml amber vial. To the PA-Acrylate was added 1 mg of DBDS (lyophilized), 15 mg Bovine Serum Albumin, and 1 mL of 1× phosphate-buffered saline (1×PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 uL was injected, using a 23 gauge needle, into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.). The tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 15 cm from light source, illuminated for 270 seconds, and then removed. After ilumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm, which indicated complete polymerization of the polyalditol-acrylate. No excess liquid was observed. The filament was manipulated with forceps.
- The filament was placed in a 1.7 ml microcentrifuge tube with 1 ml PBS containing alpha-Amylase at 0.121 μg/mL. At daily intervals for 15 days, 150 μl of PBS was removed from each well and placed into a 96 well plate for subsequent analysis. The remaining 850 μL was removed from the sample, and to the tube was added 1 ml of fresh PBS containing alpha-Amylase at 0.121 μg/mL. The 96-well plate was analyzed for BSA using the Quanitpro Assay Kit (Sigma).
- Maltodextrin filaments were synthesized using a 1,100 mg/mL solution as described in example 25 using an anti-horseradish peroxidase antibody (P7899; Sigma) instead of BSA. The filament contained 800 ug of the anti-horseradish peroxidase antibody. The filament was placed in a 1.7 ml microcentrifuge tube containing 1 ml of 1×PBS containing alpha-Amylase at 0.121 μg/mL. At daily intervals for 5 days, 100 μl of PBS was removed from the sample, placed into a 96 well plate and incubated for 60 minutes at 37° C. The remaining 850 μL was removed from the sample, and replaced with 1 ml fresh 1×PBS containing alpha-Amylase at 0.121 μg/mL. After 1 hour, the plate was washed three times with 1 ml PBS/Tween (Sigma). 150 ul StabilCoat™ Immunoassay Stabilizer (SurModics, Eden Prairie, Minn.) was added to the well and incubated for 30 minutes at room temperature. After 30 minutes, the 96-well plate was washed three times with PBS/Tween. A solution of 0.5 mg/ml Horseradish Peroxidase (Sigma) in 1×PBS (100 uL) was added to the well and incubated for 60 minutes. After 60 minutes, the 96-well plate was washed six times with PBS/Tween. A chromogenic assay was then performed. After 15 minutes, the 96 well plate was analyzed for HRP conjugate on a spectrophotometer (Tecan) at 560 nm absorbance. Detectable Antibody was found at each time point.
- 100 mg photo-PVP and 5 mg the photoinitiator tetra-BBE-PET were mixed with 10 ml Isopropyl alcohol (IPA; Fisher) for 1 minute. The mixture in an amount of 1 ml was placed into a 1.7 ml eppendorf tube (VWR). A 1.2 cm PEBAX® rod (Medical Profiles, Inc) was dipped into the solution for 10 seconds, at a dip rate of 0.75 cm/second, and then removed at the same rate. The rod was allowed to air dry for 10 minutes. The rod was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 30 cm from light source, illuminated for 180 seconds, and then removed.
- 1000 mg of MD-Acrylate, as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 5 mg DBDS, 1 ml of 1× phosphate-buffered saline (PBS), and 100 mg BSA. The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 1 ml was placed into a 1.8 ml eppendorf tube (VWR). The photo-PVP/tetra-BBE-PET coated PEBAX® rod was dipped into the mixture for 30 seconds, at a dip rate of 0.3 cm/s, and then removed at the same rate. The rod was immediately placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 30 cm from light source, and illuminated for 180 seconds and then removed.
- The rod was placed in a 1.7 ml microcentrifuge tube with 1
ml 1×PBS containing alpha-Amylase at 0.121 μg/mL. At intervals for 9 days, 150 μl of PBS was removed from each well and placed into a 96 well plate. The remaining 850 μl were removed from the samples, and replaced with 1 ml fresh 1×PBS containing alpha-Amylase at 0.121 μg/mL. The 96 well-plate was analyzed for BSA release using the Quanitpro Assay Kit (Sigma). BSA was detected at each timepoint. Results are shown in Table 3 andFIG. 2 .TABLE 3 Timepoint Cumulative BSA release (% of Total BSA) 1 7.0 2 15.0 3 19.1 4 22.7 5 25.6 7 28.6 8 31.5 9 34.2 - The MD-Acrylate-coated PEBAX rod (as prepared in Example 28) was placed in 5 ml of 1× phosphate-buffered saline (PBS) containing alpha-Amylase at 24 μg/mL for 7 days on a rotating plate at 37° C. After 7 days, the rod was removed from the PBS and washed with distilled water. The rod was then examined under a Scanning Electron Microscope (
LEO Supra 35 VP); upon examination, no trace of the MD-Acrylate coating was detected. - A circumferential dissection of the anterior segment (cornea, aqueous humour, lens) of porcine eye was performed, and the vitreous was squeezed out from the globe into a 20 mL amber vial; approx 10 mL total was retrieved from a total of four eyes. 200 mg/mL and 1100 mg/mL Maltodextrin filaments, formed in example 21, were placed into 2 mL of the vitreous solution, and placed at 37° C. on a rotator plate. The 200 mg/mL filament had completely dissolved after 24 hours. The 1,100 mg/mL filaments completely degraded after 30 days in the vitreous.
- Two solutions were prepared.
Solution # 1 was prepared as follows: 250 mg of MD-acrylate as prepared in example 13 was placed in an 8 mL vial. To the MD-acrylate was added 15 mg ferrous gluconate hydrate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 uL AMPS (Lubrizol) and 1,000 uL deionized water.Solution # 2 was prepared as follows: 250 mg of MD-acrylate as prepared in example 13 was placed in a second 8 mL vial. To this MD-acrylate was added 30 uL AMPS, 80 uL Hydrogen Peroxide (Sigma) and 890 uL 0.1 M Acetate buffer (pH 5.5). - 50 uL of
Solution # 1 was added to a glass slide. 50 uL ofsolution # 2 was added toSolution # 1 with slight vortexing. After mixing for 2 seconds, the mixture polymerized and formed a semi-firm gel having elastomeric properties. - Two solutions were prepared, similar to Example 31, but in this
Example Solution # 1 different concentrations of ferrous gluconate hydrate (Sigma) and ascorbic acid were used.Solution # 1 was prepared as follows: 250 mg of MD-acrylate (as prepared in example 13) was placed in an 8 mL vial. To the MD-acrylate was added 5 mg ferrous gluconate hydrate (Sigma), 40 mg ascorbic acid (Sigma), 67 uL AMPS (Lubrizol) and 1,000 uL deionized water.Solution # 2 was prepared as follows: 250 mg of MD-acrylate as prepared in example 7 was placed in a second 8 mL vial. To this MD-acrylate was added 30 uL AMPS, 80 uL Hydrogen Peroxide (Sigma) and 890 uL 0.1 M Acetate buffer (pH 5.5). - 50 uL of
Solution # 1 was added to a glass slide. 50 uL ofsolution # 2 was added toSolution # 1 with slight vortexing. After mixing for 8 seconds, the mixture polymerized and formed a semi-firm gel having elastomeric properties. - Two solutions were prepared.
Solution # 1 was prepared as follows: 250 mg of MD-acrylate (as prepared in example 13) was placed in an 8 mL vial. To the MD-acrylate was added 15 mg Iron (II) L-Ascorbate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 uL AMPS (Lubrizol) and 1,000 uL deionized water.Solution # 2 was prepared as follows: 250 mg of MD-acrylate as prepared in example 7 was placed in a second 8 mL vial. To this MD-acrylate was added 30 uL AMPS, 80 uL hydrogen peroxide (Sigma) and 890 uL 0.1 M Acetate buffer (pH 5.5). - 50 uL of
Solution # 1 was added to a glass slide. 50 uL ofsolution # 2 was added toSolution # 1 with slight vortexing. After mixing for 2 seconds, the mixture polymerized and formed a semi-firm gel having elastomeric properties. - Two solutions were prepared.
Solution # 1 was prepared as follows: 1,000 mg of Polyalditol-acrylate as prepared in Example 21 was placed in an 8 mL vial. To the Polyalditol-acrylate was added 15 mg Ferrous Sulfate Heptahydrate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 uL AMPS (Lubrizol) and 1,000 uL deionized water;Solution # 2 was prepared as follows: 1,000 mg of Polyalditol-acrylate as prepared in example was placed in a second 8 mL vial. To this Polyalditol-acrylate was added 30 uL AMPS, 80 uL Hydrogen Peroxide (Sigma) and 890 uL 0.1 M Acetate buffer (pH 5.5). - 50 uL of
Solution # 1 was added to a glass slide. 50 uL ofsolution # 2 was added toSolution # 1 with slight vortexing. After mixing for 2 seconds, the mixture polymerized and formed a semi-firm gel having elastomeric properties. - Photo-PVP (100 mg) and the photoinitiator tetra-BBE-PET (5 mg), were mixed with 10 mL isopropyl alcohol (IPA; Fisher) for 1 minute. The mixture in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR). A 1.2 cm PEBAX® rod (Medical Profiles, Inc) was dipped into the solution for 10 seconds, at a dip rate of 0.1 cm/second, and then removed at the same rate. The rod was allowed to air dry for 5 minutes. The rod was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 30 cm from light source, illuminated for 180 seconds, and then removed.
- Two solutions were prepared.
Solution # 1 was prepared as follows: 250 mg of MD-acrylate (as prepared in example 13) was placed in an 8 ml vial. To the MD-acrylate was added 15 mg Iron (II) Ascorbate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 ul AMPS (Lubrizol) and 1,000 ul deionized water.Solution # 2 was prepared as follows: 250 mg of MD-acrylate was placed in a second 8 ml vial. To this MD-acrylate was added 30 ul AMPS, 80 ul Hydrogen Peroxide (Sigma) and 890 ul 0.1 M Acetate buffer (pH 5.5). -
Solution # 1 in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR). The photo-PVP/tetra-BBE-PET coated PEBAX® rod was dipped into the mixture for 30 seconds, at a dip rate of 0.5 cm/s, and then removed at the same rate. The PEBAX rod was allowed to air dry for 10 minutes.Solution # 2 in an amount of 1 mL was placed into a second 1.8 mL eppendorf tube (VWR). The photo-PVP/tetra-BBE-PET andSolution # 1 coated PEBAX® rod was dipped into the mixture for 30 seconds, at a dip rate of 0.5 cm/s, and then removed at the same rate. - The MD-Acrylate coated rod was examined under Scanning Electron Microscope (SEM;
LEO Supra 35 VP); the MD-Acrylate coating thickness ranged from 15 to 20 um, with an average thickness of 16.8 um. - Two solutions were prepared.
Solution # 1 was prepared as follows: 250 mg of MD-acrylate (as prepared in example 13) was placed in an 8 ml vial. To the MD-acrylate was added 15 mg Iron (II) Acetate (Sigma), 30 mg Ascorbic Acid (Sigma), 67 ul AMPS (Lubrizol), 75 mg Bovine Serum Albumin (BSA; representing the bioactive agent) and 1,000 μL deionized water.Solution # 1 was prepared as follows: 250 mg of MD-acrylate was placed in a second 8 ml vial. To this MD-acrylate was added 30 μL AMPS, 80 μL Hydrogen Peroxide (Sigma), 75 mg BSA and 890 μL Acetate buffer (pH 5.5). - 50 μL of
Solution # 1 was added to a glass slide. 50 μL ofsolution # 2 was added toSolution # 1 with slight vortexing. After mixing for 2 seconds, the mixture polymerized and formed a semi-firm gel having elastomeric properties. - Maltodextrin-acrylate filaments were prepared using the reagents at concentrations as described in Example 31. These filaments were placed in microcentrifuge tubes containing 1 ml either Phosphate Buffered Saline (PBS) or 1×PBS containing alpha-Amylase at 0.121 μg/mL. The tubes were then placed in an incubator at 37° C.
- After 4 days in the 1×PBS containing alpha-Amylase at 0.121 μg/mL, the 250 mg/mL filament had completely degraded, leaving no trace of the matrix. The matrix in PBS showed no signs of degradation.
- 600 milligrams of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 5 mg of DBDS (lyophilized), 10 mg Rabbit Anti-Goat Fragment Antibody (catalog # 300-007-003; Jackson Immunological Research, West Grove, Pa.) and 1 mL of 1× phosphate-buffered saline (PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 μL was pipetted into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.). The tubing was placed into a Dymax Lightweld PC-2 illumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm and completely crosslinked, with no excess liquid. The filament was placed in a 1.7 mL microcentrifuge tube with 0.5
ml 1×PBS containing alpha-Amylase at 0.121 μg/mL (eluent solution). At predetermined intervals for 17 days, 200 μL of the eluent solution was removed from each tube, and 100 μL was placed into two 96 well plates. The remaining 300 μL were removed from the samples, and replaced with 0.5 mL fresh 1×PBS containing alpha-Amylase at 0.121 μg/mL. The 96 well plates were analyzed for total FAB molecule release and FAB activity using an Enzyme-Linked Immunosorbent Assay (ELISA). Briefly, the 100 μL eluent solution was incubated at 37° C. for one hour and then washed 3× with 2 ml PBS/Tween 20 (Sigma). The wells were blocked with 100 μL StabilCoat™ for 1 hour at room temperature and then washed 3× with 2 mL PBS/Tween 20. 100 uL of either 0.1 ug/mL (in PBS/Tween) HRP-labeled Goat IgG (Jackson Immunological; catalog #005-030-003) for molecule activity or 0.08 ug/mL (in PBS/Tween) HRP-labeled Goat anti-Rabbit IgG (Jackson Immunological; catalog #111-305-003) was incubated for 1 hour at 37° C. The wells were washed 6× with 2 mL PBS/Tween 20. 100 μL of TMB Microwell Peroxidase Substrate System (KPL, Catalog #50-76-00; Gaithersburg, Md.) as added to each well. After 15 minutes, the 96 well plate was analyzed for HRP conjugate on a spectrophotometer (Tecan) at 650 nm absorbance. Detectable Antibody was found at each timepoin. Results are shown in Table 4 andFIG. 3 .TABLE 4 Fab Fragment release ABS values Cumulative Active FAB Abs Cumulative Total Fab Timepoint (Day) at 650 nm Abs at 650 nm 1 1.37 1.97 3 3.12 4.07 4 4.54 5.87 6 5.69 7.54 7 6.12 8.60 8 6.53 9.01 10 6.94 9.79 13 7.34 10.64 15 7.54 11.18 17 7.71 11.62 19 7.81 11.92 21 7.90 12.28 23 8.00 12.68 26 8.09 13.11 - 316V or 304V stainless steel rods (0.035″ diameter; Small Parts, Inc.) were cleaned by wiping the rods with isopropyl alcohol (IPA) soaked
Alpha 10 clean-wipes (Texwipe; Kernersville, N.C.) and then sonicating the rods in a solution of 10% Valtron SP2200 detergent (Valtech Corp.) in hot tap water for 10 minutes. The rods were then rinsed 3× with deionized water followed by a 1 minute sonication in hot tap water; the rinsed and sonication steps were then repeated. - A 0.5% 1,4-bis(trimethoxysilylethyl)benzene (B2495.6, UCT, Bristol, Pa.) solution in 89.5% IPA and 10% deionized water was prepared (See Example 1 of U.S. Pat. No. 6,706,408B2) and the cleaned rods were dipped into the silane solution within 2 minutes of coming out of the deionized rinse. The rods were allowed to dwell in the silane solution for 3 minutes and then pulled out at a rate of 1.0 cm/s. They were allowed to air dry for 2 minutes at room temperature and then placed at 110° C. for 5 minutes.
- A solution of photo-PVP (15 mg/mL), DBDS (1 mg/mL), tetra-BBE-PET 0.075 (mg/mL) and PVP K90 (20 mg/ml; BASF) was prepared in 60% IPA/40% water) (see also Example 1 of U.S. Pat. No. 6,706,408B2). The silane treated rods were dip coated into the above solution using the following parameters:
- Velocity in=2.0 cm/s; dwell time=60 seconds; velocity out=0.1 cm/s The coated rods were allowed to air dry at room temperature for 5 minutes and then illuminated in a Dymax lamp UV light chamber, with rotation, for 3 minutes. The dipping procedure was then repeated with a dwell time of 120 seconds (all other dipping parameters the same).
- Two solutions were prepared. The first solution (#1) was prepared by placing 600 mg of MD-acrylate (as prepared in example 13) into an 8 mL vial and then adding 9 mg iron (II) ascorbate (Sigma), 30 mg ascorbic acid (Sigma), 67 μL AMPS (Lubrizol), 16 mg Rabbit Anti-HRP antibody (Sigma; catalog # P7899), and 1,000 μL deionized water. The second solution (#2) was prepared by placing 600 mg of MD-acrylate in a second 8 mL vial and then adding 30 μL AMPS, 16 mg Rabbit Anti-HRP antibody (Sigma; catalog # P7899), 80 μL hydrogen peroxide (Sigma) and 890 μL 0.1 M Acetate buffer (pH 5.5).
-
Solution # 1 in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR). The (PV01/K90/DBDS/tetra-BBE-PET)-coated stainless steel rod (20 mm in length) was dipped into the mixture for 20 seconds, at a dip rate of 0.75 cm/s, and then removed at the same rate. The rod was allowed to air dry for 10 minutes.Solution # 2 in an amount of 1 mL was placed into a second 1.8 mL eppendorf tube (VWR). The (PV01/K90/DBDS/tetra-BBE-PET)-coated andSolution # 1 coated rod was dipped into theSolution # 2 for 20 seconds, at a dip rate of 0.75 cm/s, and then removed at the same rate. Contact withSolution # 2 initiated the redox reaction and caused formation of an MD-Acrylate coated layer on the PV01/K90/DBDS/tetra-BBE-PET coated layer; the MD-Acrylate coating was cured within 10 seconds - The coated rod was examined under Optical Inferometer Microscope (Veeco) which revealed that the MD-Acrylate coating layer had a thickness of 70 μm.
- The MD-Acrylate coated rod was placed in a 0.6 mL microcentrifuge tube with 0.5
ml 1×PBS containing alpha-Amylase at 0.121 μg/mL (eluent solution) to assess release of the bioactive agent (antibody) from the coating. - At predetermined intervals for 19 days, 200 μL of the eluent solution was removed from the tube, which was divided into two 100 μL aliquots and placed into two 96 well plates. The remaining 300 μL was removed from the microcentrifuge tube, and 0.5 mL of fress eluent solution (1×PBS containing alpha-Amylase at 0.121 μg/mL) was added to the microcentrifuge tube having the MD-Acrylate coated rod.
- The eluent samples in 96 well plates were analyzed for total Rabbit Antibody molecule release and activity using an Enzyme-Linked Immunosorbent Assay (ELISA). Briefly, the 100 μL of eluent solution was incubated at 37° C. for one hour and then washed 3× with 2 ml PBS/Tween 20 (Sigma). The wells were blocked with 100 μL StabilCoat™ (SurModics, Eden Prairie, Minn.) for 1 hour at room temperature and then washed 3× with 2 ml PBS/
Tween 20. 100 μL of either 0.1 ug/mL (in PBS/Tween) HRP (Sigma; catalog # P8375) for molecule activity or 0.08 ug/mL (in PBS/Tween) HRP-labeled Goat anti-Rabbit IgG (Jackson Immunological; catalog # 111-305-003) was incubated for 1 hour at 37° C. The wells were washed 6× with 2 ml PBS/Tween 20. 100 μL of TMB Microwell Peroxidase Substrate System (KPL, Catalog # 50-76-00; Gaithersburg, Md.) was added to each well. After 15 minutes, the 96 well plate was analyzed for HRP conjugate on a spectrophotometer (Tecan) at 650 nm absorbance. Detectable antibody was found in the eluate samples at each timepoint. Results are shown in Table 5 andFIG. 4 .TABLE 5 Cumulative Active IgG Abs at Cumulative Total IgG Timepoint (Day) 650 nm Abs at 650 nm 1 1.08 2.01 2 1.82 3.78 4 2.01 5.11 6 2.41 5.57 8 2.48 5.75 10 2.54 5.88 12 2.59 5.97 19 2.87 7.88 - 316V or 304V stainless steel rods (0.035″ diameter; Small Parts, Inc., Miami Lakers, Fla.) were cleaned by wiping the rods with isopropyl alcohol (IPA)-soaked
Alpha 10 clean-wipe (Texwipe) and then sonicating the rods in a solution of 10% Valtron SP2200 detergent in hot tap water for 10 minutes. The rods were then rinsed 3× with deionized water followed by a 1 minute sonication in hot tap water; the rinse and sonication step was then repeated. - The cleaned rods were dipped into a 0.5% 1,4-bis(trimethoxysilylethyl)benzene solution (as described in Example 39) within 2 minutes of coming out of the deionized rinse. The rods were allowed to dwell in the silane solution for 3 minutes and then pulled out at a rate of 1.0 cm/s. The rods were allowed to air dry for 2 minutes at room temperature and then placed at 110° C. for 5 minutes.
- A solution of PV01 (15 mg/mL), DBDS (1 mg/mL), tetra-BBE-PET (0.075 mg/ml) and K90 (20 mg/mL; BASF) was prepared in 60% IPA/40% water. The silane treated rods were dip coated into the above solution using the following parameters:
- Velocity in=2.0 cm/s; dwell time=60 seconds; velocity out=0.1 cm/s. The coated rods were allowed to air dry at room temperature for 5 minutes and then illuminated in a Dymax lamp UV light chamber, with rotation, for 3 minutes. The dipping procedure was then repeated with a dwell time of 120 seconds (all other dipping parameters the same).
- 600 milligrams of MD-Acrylate as prepared in Example 13 was placed in an 8 mL amber vial. To the MD-Acrylate was added 5 mg of DBDS (lyophilized), 16 mg Rabbit Anti-HRP antibody (Sigma; catalog # P7899) and 1 ml of 1× phosphate-buffered saline (PBS). The reagents were then mixed for one hour on a shaker at 37° C. The MD-Acrylate solution in an amount of 1 mL was placed into a 1.8 mL eppendorf tube (VWR). The (PV01/K90/DBDS/tetra-BBE-PET)-coated stainless steel rod (20 mm in length) was dipped into the mixture for 20 seconds, at a dip rate of 0.75 cm/s, and then removed at the same rate. The rod was then immediately illuminated in a Dymax lamp UV light chamber, with rotation, for 3 minutes. The rod was allowed to air dry for 5 minutes, and the dipping and illuminating procedures were repeated one more time.
- The MD-Acrylate coated rod was examined under Optical Inferometer Microscope (Veeco); the MD-Acrylate coating layer had a coating thickness of 20 μm.
- The MD-Acrylate coated rod was placed in a 0.6 mL microcentrifuge tube with 0.5
mL 1×PBS containing alpha-Amylase at 0.121 μg/mL (eluent solution) to assess degradation of the coating and release of the bioactive agent (antibody) from the coating. At predetermined intervals for 25 days, 200 μL of the eluent solution was removed from each tube, which was divided into two 100 μL aliquots and placed into two 96 well plates. The remaining 300 μL was removed from the microcentrifuge tube, and 0.5 mL of fress eluent solution (1×PBS containing alpha-Amylase at 0.121 μg/mL) was added to the microcentrifuge tube having the MD-Acrylate coated rod. - The 96 well plates were analyzed for total Rabbit Antibody molecule release and activity using an Enzyme-Linked Immunosorbent Assay (ELISA). Briefly, the 100 μL eluent solution was added to the wells and incubated at 37° C. for one hour and then washed 3× with 2 mL PBS/Tween 20 (Sigma). The wells were blocked with 100 μL StabilCoat™ for 1 hour at room temperature and then washed 3× with 2 mL PBS/
Tween 20. 100 μL of either 0.1 ug/mL (in PBS/Tween) HRP (Sigma; catalog # P8375 ) for molecule activity or 0.08 ug/mL (in PBS/Tween) HRP-labeled Goat anti-Rabbit IgG (Jackson Immunological; catalog # 111-305-003) was incubated for 1 hour at 37° C. The well were washed 6× with 2 mL PBS/Tween 20. 100 μL of TMB Microwell Peroxidase Substrate System (KPL, Catalog # 50-76-00; Gaithersburg, Md.) was added to each well. After 15 minutes, the 96 well plate was analyzed for HRP conjugate on a spectrophotometer (Tecan) at 650 nm absorbance. Detectable antibody was found in the eluant samples at each time point. - Based on the initial weight of the coating before degradation, a theoretical maximum concentration of the antibody was calculated. In addition to measurements performed by ELISA, the MD-Acrylate coated rod was weighed at various time points to determine the amount of material of the MD-Acrylate coated layer lost from the rods due to amylase digestion. Results are shown in Table 6 and
FIG. 5 .TABLE 6 Cumulative Cumulative Active IgG Total IgG MD-acrylate Maximum Timepoint release (%) release (%) coating theoretical total (Day) (ELISA) (ELISA) remaining (%) IgG release (%) 1 7.14 9.29 2 8.14 10.14 83 17 4 8.49 10.29 6 8.93 10.49 7 80 20 8 9.27 10.76 10 9.63 10.90 12 10.06 11.19 14 10.19 11.36 80 20 17 10.65 11.91 19 11.19 12.68 22 12.62 13.38 25 14.76 14.35 - 600 milligrams of MD-Acrylate as prepared in Example 13 was placed in an 8 ml amber vial. To the MD-Acrylate was added 5 mg of DBDS (lyophilized), 16 mg Rabbit Antibody Anti-HRP (Sigma; catalog # P7899) and 1 ml of 1× phosphate-buffered saline (PBS). The reagents were then mixed for one hour on a shaker at 37° C. The mixture in an amount of 10 μL was pipetted into a 22 mm length opaque silicone tube (P/N 10-447-01; Helix Medical, Carpinteria, Calif.). The tubing was placed into a Dymax Lightweld PC-2 ilumination system (Dymax Corp.; light intensity 6.5 mW/cm2), 15 cm from light source, illuminated for 270 seconds, and then removed. After illumination, the filament was removed from the silicone tubing by rolling a pencil over the tubing, starting from the back. The filament was firm and completely crosslinked, with no excess liquid.
- The filament was placed in a 1.7 ml microcentrifuge tube with 0.5
ml 1×PBS containing alpha-Amylase at 0.121 μg/mL (eluent solution). At predetermined intervals for 25 days, 200 μl of the eluent solution was removed from each tube, and 100 μL was placed into two 96 well plates. The remaining 300 μl were removed from the samples, and replaced with 0.5 ml fresh 1×PBS containing alpha-Amylase at 0.121 μg/mL. The 96 wellplates were analyzed for total Rabbit Antibody molecule release and activity using an Enzyme-Linked Immunosorbent Assay (ELISA). Briefly, the 100 μL eluent solution was added to the wells and incubated at 37 degrees C. for one hour and then washed 3× with 2 ml PBS/Tween 20 (Sigma). The wells were blocked with 100 μL StabilCoat™ (SurModics) for 1 hour at room temperature and then washed 3× with 2 ml PBS/Tween 20. 100 μL of either 0.1 ug/ml (in PBS/Tween) HRP (Sigma; catalog # P8375 ) for molecule activity or 0.08 ug/ml (in PBS/Tween) HRP-labeled Goat anti-Rabbit IgG (Jackson Immunological; catalog # 111-305-003) was incubated for 1 hour at 37 degrees C. The wells were washed 6× with 2 ml PBS/Tween 20. 100 μL of TMB Microwell Peroxidase Substrate System (KPL, Catalog # 50-76-00; Gaithersburg, Md.) was added to each well. After 15 minutes, the 96 well plate was analyzed for HRP conjugate on a spectrophotometer (Tecan) at 650 nm absorbance. Detectable Antibody was found at each time point. - Results are shown in Table 7 and
FIG. 6 .TABLE 7 Cumulative Cumulative MD-acrylate Active Total IgG coating Maximum Timepoint release (%) IgG release (%) remaining theoretical total (Day) (ELISA) (ELISA) (%) IgG release (%) 1 5.56 5.31 2 12.13 11.94 4 18.38 19.13 6 27.75 22.88 7 83 17 8 33.50 25.44 10 37.63 27.44 12 39.50 28.31 14 40.75 28.57 59 31 17 41.75 28.76 19 42.75 28.98 21 40 60 22 43.44 29.67 25 44.31 30.67 - MD-acrylate discs formed via redox polymerization of MD-acrylate coating solutions were tested for mechanical properties.
- A first solution (#1) was prepared by placing 300 mg of MD-acrylate as prepared in Example 13 into an 8 ml vial and then adding 9 mg iron (II) ascorbate (Sigma), 30 mg ascorbic acid (Sigma), 67 μL AMPS (Lubrizol), and 1,000 μL deionized water.
Solution # 2 prepared by placing 300 mg of MD-acrylate into a second 8 ml vial and then adding 30 μL AMPS, 80 μL if hydrogen peroxide (Sigma) and 890 μL of 0.1 M Acetate buffer (pH 5.5). - Viscosity of the first and second solutions were determined on a Brookfield Viscometer. The average viscosity for both solutions was 10.9 cP.
- The modulus of the formed matrix was determined by rheological measurements. In order to perform rheological measurements, the first and second solutions were combined on the testing plate in the Rheometer (Rheometric Scientific; model # SR-2000) and the mixture was allowed to polymerize to form a matrix. Data recording began before sample was cured in plates. Briefly, 100 μL of
solution # solution # 2 were mixed on the lower testing plate. As the matrix formed, the upper testing plate was lowered to fully contact the mixture of the first and second solutions as the mixture polymerized into matrix. The sample was cured within 15 seconds. This curing method ensured maximum contact between the two testing plates resulting in more accurate testing compared to preformed matrices being placed between the testing plates. The resulting MD-acrylate matrix had properties of an elastic solid with an elastic (storage) modulus ranging from 27 kPa to 30 kPa, and a viscous (loss) modulus of only about 1 kPa. Results are shown in Table 8 andFIG. 7 .TABLE 8 (Testing Conditions: Stress: 433 Pa; strain 1.6%; frequency: 1 radian/sec) G′ (Elastic G″ (Storage G* (Loss Time (seconds) Modulus; Pa) Modulus; Pa) Modulus; Pa) 247 26820.2 1300.5 26851.7 261 26908.5 1294.55 26939.6 274 26872 1299.28 26903.4 288 26943.8 1343.69 26977.3 301 27376.6 1380.43 27411.4 315 27327.7 1373.31 27362.2 329 27319.8 1376.27 27354.5 342 27274.8 1362.35 27308.8 356 27246.6 1369.38 27281 369 27180.6 1373.6 27215.3 383 27174.4 1371.61 27209 397 27119.4 1366.76 27153.9 410 27105.4 1360.49 27139.5 424 27064.1 1358.45 27098.2 437 27019.9 1355.9 27053.9 451 27019.8 1355.39 27053.8 465 26972.3 1355.85 27006.4 478 26956.2 1361.11 26990.5 492 26918.2 1352.58 26952.2 505 26880.7 1355.85 26914.9 519 26840.4 1360.47 26874.9 - To assess lubricity and tenacity of MD-acrylate coated parts, frictional force testing was performed.
- Fricitional testing over the last 40 cycles of a 50 cycle test was evaluated. Coated rods were evaluated by a horizontal sled style friction test method (modified ASTM D-1894, as described below). Silicone Pads (7 mm diameter) were hydrated and then wrapped around a 200 gram stainless steel sled. The silicone pad was clipped together tightly on the opposite side of the sled. The sled with rotatable arm was then attached to a 500 gram Chatillon Digital Force Gauge (DGGHS, 500×0.1) with computer interface. The testing surface was mounted on a 22.5 inch positioning rail table with micro-stepper motor control (Compumotor SX6 Indexer/Drive).
- MD-coated rods (from example 18) were hydrated in deionized water and clamped onto the
test surface 1 inch (or approximately 2.5 cm) apart. The hydrated Silicone pad (jaw force set at 500 g) moved at 0.5 cm/sec over a 5 cm section for 50 push/pull cycles, and the final force measurements were taken over the last 40 push/pull cycles. - As shown in Table 9 and
FIG. 8 , compared to the benchmark synthetic coating (see example 1 of U.S. Pat. 6,706,408B2) the MD-acrylate coated rod provided a coating having excellent lubricity and durability; the rods coated with a MD-acrylate formulation containing a photoinitiator, the grams of force remained relatively constant for the last 40 cycles, indicating a durable coating.TABLE 9 MD-acrylate photo coated rod Synthetic 1 9.67 12.53 2 9.53 12.43 3 9.45 12.26 4 9.18 12.40 5 9.12 12.33 6 9.13 12.17 7 9.11 12.15 8 9.06 12.12 9 8.92 12.25 10 8.96 12.19 11 8.91 12.11 12 8.98 12.02 13 8.95 12.02 14 8.82 12.25 15 8.79 12.10 16 8.84 12.08 17 8.85 12.02 18 8.89 11.92 19 8.76 12.10 20 8.81 12.08 21 8.85 11.93 22 8.85 11.86 23 8.88 11.83 24 8.81 12.01 25 8.90 11.94 26 8.96 11.94 27 9.04 11.87 28 9.04 11.71 29 8.97 12.00 30 9.03 11.94 31 9.05 11.79 32 9.20 11.83 33 9.32 11.75 34 9.23 11.90 35 9.22 11.82 36 9.39 11.79 37 9.46 11.68 38 9.52 11.74 39 9.53 11.91 40 9.56 11.87 - Maltodextrin having pendent butyryl groups were prepared by coupling butyric anhydride at varying molar ratios.
- To provide butyrylated-MD (1 butyl/4 glucose units, 1:4 B/GU) the following procedure was performed. Maltodextrin (MD; Aldrich; 11.0 g; 3.67 mmole; DE (Dextrose Equivalent): 4.0-7.0) was dissolved in dimethylsulfoxide (DMSO) 600 mL with stirring. The size of the maltodextrin was calculated to be in the range of 2,000 Da-4,000 Da. Once the reaction solution was complete, 1-methylimidazole (Aldrich; 2.0 g, 1.9 mls) and butyric anhydride (Aldrich; 5.0 g, 5.2 mls) was added with stirring. The reaction mixture was stirred for four hours at room temperature. After this time, the reaction mixture was quenched with water and dialyzed against DI water using 1,000 MWCO dialysis tubing. The butyrylated starch was isolated via lyophylization to give 9.315 g (85% yield). NMR confirmed a butyrylation of 1:3 B/GU (1.99 mmoles butyl/g sample).
- To provide butyrylated-MD (1:8 B/GU), 2.5 g (2.6 mL) butyric anhydride was substituted for the amount of butyric anhydride described above. A yield of 79% (8.741 g) was obtained. NMR confirmed a butyrylation of 1:5 B/GU (1.31 mmoles butyl/g sample).
- To provide butyrylated-MD (1:2 B/GU), 10.0 g (10.4 mL) butyric anhydride was substituted for the amount of butyric anhydride described above. A yield of 96% (10.536 g) was obtained. NMR confirmed a butyrylation of 1:2 B/GU (3.42 mmoles butyl/g sample).
- Preparation of an acylated maltodextrin macromer having pendent butyryl and acrylate groups prepared by coupling butyric anhydride at varying molar ratios.
- To provide butyrylated-MD-acrylate (1 butyl/4 glucose units, 1:4 B/GU) the following procedure was performed. MD-Acrylate (Example 13; 1.1 g; 0.367 mmoles) was dissolved in dimethylsulfoxide (DMSO) 60 mL with stirring. Once the reaction solution was complete, 1-methylimidazole (0.20 g, 0.19 mls) and butyric anhydride (0.50 g, 0.52 mls) was added with stirring. The reaction mixture was stirred for four hours at room temperature. After this time, the reaction mixture was quenched with water and dialyzed against DI water using 1,000 MWCO dialysis tubing. The butyrylated starch acrylate was isolated via lyophylization to give 821 mg (75% yield, material lost during isolation). NMR confirmed a butyrylation of 1:3 B/GU (2.38 mmoles butyl/g sample).
- Maltodextrin having pendent butyryl and acrylate groups prepared by coupling butyric anhydride at varying molar ratios.
- To provide butyrylated-MD-acrylate the following procedure is performed. Butyrylated-MD (Example 43; 1.0 g; 0.333 mmole) is dissolved in dimethylsulfoxide (DMSO) 60 mL with stirring. Once the reaction solution is complete, a solution of EA-NCO from Example 12 (353 mg; 2.50 mmole) is evaporated and dissolved in dried DMSO 1.0 mL). The two DMSO solutions are mixed and heated to 55° C. overnight. The DMSO solution is placed in dialysis tubing (1000 MWCO) and dialyzed against water for 3 days. The macromer solution is filtered and lyophilized to give a white solid.
Claims (28)
1. An in vivo matrix-forming composition comprising a natural biodegradable polysaccharide comprising a pendent coupling group, and having a molecular weight of 100,000 Da or less.
2. The in vivo matrix-forming composition of claim 1 wherein the natural biodegradable polysaccharide has a molecular weight of 50,000 Da or less.
3. The in vivo matrix-forming composition of claim 2 wherein the natural biodegradable polysaccharide has a molecular weight in the range of 1000 Da to 10,000 Da.
4. The in vivo matrix-forming composition of claim 1 wherein biodegradable polysaccharide is selected from the group consisting of amylose and maltodextrin.
5. The in vivo matrix-forming composition of claim 1 wherein biodegradable polysaccharide comprises a non-reducing natural biodegradable polysaccharide.
6. The in vivo matrix-forming composition of claim 5 wherein biodegradable polysaccharide is selected from the group consisting of polyalditol.
7. The in vivo matrix-forming composition of claim 1 wherein the coupling group is present on the natural biodegradable polysaccharide in an amount of 0.7 mmoles or less of coupling group per milligram of natural biodegradable polysaccharide.
8. The in vivo matrix-forming composition of claim 1 wherein the coupling group is a polymerizable group.
9. The in vivo matrix-forming composition of claim 8 wherein the polymerizable group is selected from vinyl groups, acrylate groups, methacrylate groups, ethacrylate groups, 2-phenyl acrylate groups, acrylamide groups, methacrylamide groups, itaconate groups, and styrene groups.
10. The in vivo matrix-forming composition of claim 9 further comprising a polymerization initiator.
11. The in vivo matrix-forming composition of claim 10 wherein the polymerization initiator comprises a photoreactive group.
12. The in vivo matrix-forming composition of claim 10 wherein the polymerization initiator comprises a redox pair.
13. The in vivo matrix-forming composition of claim 1 wherein the coating comprises a bioactive agent.
14. The in vivo matrix-forming composition of claim 13 wherein the bioactive agent is selected from the group comprising polypeptides, nucleic acids, and polysaccharides.
15. The in vivo matrix-forming composition of claim 13 wherein the bioactive agent has a molecular weight of 10,000 Da or greater.
16. The in vivo matrix-forming composition of claim 15 wherein the bioactive agent comprises a cell.
17. The in vivo matrix-forming composition of claim 1 , wherein the natural biodegradable polysaccharide further comprises a pendent hydrophobic moiety.
18. The in vivo matrix-forming composition of claim 17 , wherein the pendent hydrophobic moiety comprises a fatty acid or derivative thereof.
19. The in vivo matrix-forming composition of claim 18 , wherein the pendent hydrophobic moiety comprises a butyryl group.
20. A kit for preparing an in vivo matrix-forming composition, the kit comprising:
a natural biodegradable polysaccharide comprising a pendent polymerizable group,
a first member of a redox pair, and
a second member of a redox pair.
21. The kit of claim 20 comprising
a first composition comprising the natural biodegradable polysaccharide and the first member of a redox pair, and
a second member of a redox pair.
22. The kit of claim 21 comprising
a second composition comprising a natural biodegradable polysaccharide and the second member of a redox pair.
23. The kit of claim 22 wherein the natural biodegradable polysaccharide of the first composition is the same as the natural biodegradable polysaccharide of the second composition.
24. The kit of claim 22 wherein the first composition, the second composition, or a mixture of the first and second composition has a viscosity of 5 cP or greater.
25. The kit of claim 21 wherein the redox pair comprises an oxidizing member selected from the group consisting of peroxides, metal oxides, and oxidases, and a reducing member selected from the group consisting of salts and derivatives of electropositive elemental metals and reductases.
26. The kit of claim 21 comprising a composition comprising the reducing member at a concentration of 5 mM or greater.
27. A method for forming a matrix of polymerized material in vivo, comprising the steps of
providing a first composition comprising
a natural biodegradable polysaccharide comprising a pendent polymerizable group, and
a first member of a redox pair;
providing a second composition comprising
a natural biodegradable polysaccharide comprising a pendent polymerizable group, and
a second member of a redox pair;
contacting the first composition with the second composition comprising a second member of the redox pair where, in the step of contacting, the redox pair initiates polymerization of the natural biodegradable polysaccharides; and
disposing the first composition, the second composition, or a mixture of the first and second composition in vivo.
28. The method of claim 27 comprising the sequential steps of:
contacting the first composition with the second composition; and then
disposing the mixture of the first and second composition in vivo.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/271,238 US20070065483A1 (en) | 2005-09-21 | 2005-11-11 | In vivo formed matrices including natural biodegradable polysaccharides and uses thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US71946605P | 2005-09-21 | 2005-09-21 | |
| US11/271,238 US20070065483A1 (en) | 2005-09-21 | 2005-11-11 | In vivo formed matrices including natural biodegradable polysaccharides and uses thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20070065483A1 true US20070065483A1 (en) | 2007-03-22 |
Family
ID=36958998
Family Applications (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/271,213 Expired - Fee Related US8512736B2 (en) | 2005-09-21 | 2005-11-11 | Coatings including natural biodegradable polysaccharides and uses thereof |
| US11/271,238 Abandoned US20070065483A1 (en) | 2005-09-21 | 2005-11-11 | In vivo formed matrices including natural biodegradable polysaccharides and uses thereof |
| US11/271,237 Expired - Fee Related US8241656B2 (en) | 2005-09-21 | 2005-11-11 | Articles including natural biodegradable polysaccharides and uses thereof |
| US11/524,926 Abandoned US20100226960A1 (en) | 2005-09-21 | 2006-09-21 | Natural biodegradable matrices and uses thereof |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/271,213 Expired - Fee Related US8512736B2 (en) | 2005-09-21 | 2005-11-11 | Coatings including natural biodegradable polysaccharides and uses thereof |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/271,237 Expired - Fee Related US8241656B2 (en) | 2005-09-21 | 2005-11-11 | Articles including natural biodegradable polysaccharides and uses thereof |
| US11/524,926 Abandoned US20100226960A1 (en) | 2005-09-21 | 2006-09-21 | Natural biodegradable matrices and uses thereof |
Country Status (7)
| Country | Link |
|---|---|
| US (4) | US8512736B2 (en) |
| EP (2) | EP1926509B1 (en) |
| JP (2) | JP5139301B2 (en) |
| CN (3) | CN101309709A (en) |
| AT (2) | ATE540705T1 (en) |
| CA (2) | CA2621595A1 (en) |
| WO (2) | WO2007040557A1 (en) |
Cited By (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050255142A1 (en) * | 2004-05-12 | 2005-11-17 | Surmodics, Inc. | Coatings for medical articles including natural biodegradable polysaccharides |
| US20070065482A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | Articles including natural biodegradable polysaccharides and uses thereof |
| US20070065484A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | In situ occlusion using natural biodegradable polysaccharides |
| US20080114096A1 (en) * | 2006-11-09 | 2008-05-15 | Hydromer, Inc. | Lubricious biopolymeric network compositions and methods of making same |
| US20090060973A1 (en) * | 2002-05-24 | 2009-03-05 | Angiotech International Ag | Compositions and methods for coating medical implants |
| US20090269407A1 (en) * | 2008-04-28 | 2009-10-29 | Surmodics, Inc. | Poly-alpha(1-4)glucopyranose-based matrices with hydrazide crosslinking |
| US20100068170A1 (en) * | 2008-09-15 | 2010-03-18 | Michal Eugene T | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| WO2010136191A1 (en) * | 2009-05-27 | 2010-12-02 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. | Process for preparing polysaccharide esters or polysaccharide mixed esters |
| US20110129513A1 (en) * | 2002-05-24 | 2011-06-02 | Angiotech International Ag | Compositions and methods for coating medical implants |
| US8114429B2 (en) | 2008-09-15 | 2012-02-14 | Cv Ingenuity Corp. | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US8128951B2 (en) | 2008-09-15 | 2012-03-06 | Cv Ingenuity Corp. | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US20120058351A1 (en) * | 2006-03-30 | 2012-03-08 | Becton, Dickinson And Company | Coating System, Articles and Assembly Using the Same and Methods of Reducing Sticktion |
| US8257722B2 (en) | 2008-09-15 | 2012-09-04 | Cv Ingenuity Corp. | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| GB2493263A (en) * | 2011-07-19 | 2013-01-30 | Neoss Ltd | Implantable medical device with a surface coating |
| US8603638B2 (en) | 2006-03-30 | 2013-12-10 | Becton, Dickinson And Company | Sealing members, articles using the same and methods of reducing sticktion |
| US8901092B2 (en) | 2010-12-29 | 2014-12-02 | Surmodics, Inc. | Functionalized polysaccharides for active agent delivery |
| US8936811B2 (en) | 2008-05-07 | 2015-01-20 | Surmodics, Inc. | Device coated with glycogen particles comprising nucleic acid complexes |
| US9910020B1 (en) | 2005-03-30 | 2018-03-06 | Copilot Ventures Fund Iii Llc | Methods and articles for identifying objects using encapsulated perfluorocarbon tracers |
| US9956385B2 (en) | 2012-06-28 | 2018-05-01 | The Spectranetics Corporation | Post-processing of a medical device to control morphology and mechanical properties |
| US10525171B2 (en) | 2014-01-24 | 2020-01-07 | The Spectranetics Corporation | Coatings for medical devices |
| US20200215238A1 (en) * | 2017-05-17 | 2020-07-09 | Phenox Gmbh | Coating for medical devices |
Families Citing this family (133)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6555543B2 (en) | 2000-08-04 | 2003-04-29 | Dmi Biosciences, Inc. | Method of using diketopiperazines and composition containing them |
| US7431710B2 (en) | 2002-04-08 | 2008-10-07 | Glaukos Corporation | Ocular implants with anchors and methods thereof |
| KR20060022659A (en) | 2003-05-15 | 2006-03-10 | 디엠아이 바이오사이언시스, 인크 | Method of treating T-cell mediated disease |
| US9452001B2 (en) * | 2005-02-22 | 2016-09-27 | Tecres S.P.A. | Disposable device for treatment of infections of human limbs |
| WO2007059144A1 (en) * | 2005-11-15 | 2007-05-24 | Surmodics, Inc. | Ultrasonic nozzles for applying two-component coatings |
| US8518414B2 (en) * | 2005-11-17 | 2013-08-27 | Revance Therapeutics, Inc. | Compositions and methods of topical application and transdermal delivery of botulinum toxins with reduced non-toxin proteins |
| US8486135B2 (en) * | 2006-06-01 | 2013-07-16 | Abbott Cardiovascular Systems Inc. | Implantable medical devices fabricated from branched polymers |
| EP2037882B1 (en) * | 2006-06-28 | 2014-12-10 | SurModics, Inc. | Combination degradable and non-degradable matrices for active agent delivery |
| WO2008082694A2 (en) * | 2006-07-14 | 2008-07-10 | Akermin, Inc. | Organelles in bioanodes, biocathodes, and biofuel cells |
| US8067485B2 (en) * | 2006-07-28 | 2011-11-29 | Biograde (Hong Kong) Pty Ltd | Masterbatch and polymer composition |
| WO2008022258A2 (en) * | 2006-08-16 | 2008-02-21 | Surmodics, Inc. | Methods and materials for increasing the adhesion of elution control matrices to substrates |
| WO2008039749A2 (en) * | 2006-09-25 | 2008-04-03 | Surmodics, Inc. | Multi-layered coatings and methods for controlling elution of active agents |
| US20080075779A1 (en) * | 2006-09-27 | 2008-03-27 | Chappa Ralph A | Additives And Methods For Enhancing Active Agent Elution Kinetics |
| JP5241517B2 (en) * | 2007-02-06 | 2013-07-17 | 久光製薬株式会社 | Microneedle device for allergy diagnosis |
| US20090022805A1 (en) * | 2007-06-28 | 2009-01-22 | Joram Slager | Polypeptide microparticles having sustained release characteristics, methods and uses |
| US20110137243A1 (en) * | 2007-09-06 | 2011-06-09 | Abbott Cardiovascular Systems Inc. | Coating On A Balloon Device |
| WO2009038783A1 (en) * | 2007-09-19 | 2009-03-26 | Surmodics, Inc. | Biocompatible foams, systems, and methods |
| CN101809446B (en) | 2007-09-27 | 2013-11-27 | 株式会社三养生物制药 | Sol-gel phase reversible hydrogel template and its application |
| US20090214601A1 (en) * | 2007-09-28 | 2009-08-27 | Chappa Ralph A | Porous Drug Delivery Devices and Related Methods |
| EP2214748B1 (en) * | 2007-10-19 | 2016-12-07 | Interface Biologics Inc. | Self-eliminating coatings |
| WO2009064957A1 (en) * | 2007-11-14 | 2009-05-22 | Osteosphere, Llc | Development of a human colloidal bone graft material |
| WO2009064917A2 (en) * | 2007-11-14 | 2009-05-22 | Osteosphere, Llc | Generation of an hla-negative osteogenic precursor cell line |
| EP2067494A1 (en) * | 2007-12-04 | 2009-06-10 | Charité-Universitätsmedizin Berlin | Sheet or tubular structure consisting of elastic biocompatible material and its use |
| US20090186059A1 (en) * | 2008-01-14 | 2009-07-23 | Johnson Elizabeth E | Devices and methods for elution of nucleic acid delivery complexes |
| US8109908B1 (en) | 2008-01-22 | 2012-02-07 | Greatbatch Ltd. | Biodegradable shroud for a dilator/sheath assembly |
| US8173222B2 (en) * | 2008-03-14 | 2012-05-08 | Spencer Brody | Controlled degradability of organic materials |
| US20090263449A1 (en) * | 2008-04-09 | 2009-10-22 | Surmodics, Inc. | Delivery of nucleic acid complexes from materials including negatively charged groups |
| DE102008021894A1 (en) * | 2008-05-02 | 2009-11-05 | Biotronik Vi Patent Ag | Implant comprising a surface with reduced thrombogenicity |
| JP5856843B2 (en) | 2008-05-27 | 2016-02-10 | アンピオ ファーマシューティカルズ,インコーポレイテッド | Pharmaceutical composition using diketopiperazine |
| JP5340823B2 (en) * | 2008-06-24 | 2013-11-13 | ナガセ医薬品株式会社 | Medical lubricant |
| EP2306929A1 (en) * | 2008-07-14 | 2011-04-13 | SurModics, Inc. | Medical devices and methods for delivery of nucleic acids |
| US20100124568A1 (en) * | 2008-11-20 | 2010-05-20 | Med-Eez, Inc | Pharmaceutical articles coated with lubricious coatings |
| CA2758260A1 (en) * | 2009-04-08 | 2010-10-14 | Surmodics, Inc. | Controlled release devices and methods for delivery of nucleic acids |
| WO2010118213A2 (en) | 2009-04-08 | 2010-10-14 | Surmodics, Inc. | Particles for delivery of nucleic acids and related devices and methods |
| US9155815B2 (en) * | 2009-04-17 | 2015-10-13 | Tenaxis Medical, Inc. | Biocompatible phase invertible proteinaceous compositions and methods for making and using the same |
| US20100280594A1 (en) * | 2009-05-01 | 2010-11-04 | Medi-Solve, Llc | Antithrombotic Neurovascular Device Containing a Glycoprotein IIB/IIIA Receptor Inhibitor for The Treatment of Brain Aneurysms and/or Acute Ischemic Stroke, and Methods Related Thereto |
| US8246576B2 (en) | 2009-05-18 | 2012-08-21 | Surmodics, Inc. | Method and apparatus for delivery of a therapeutic agent with an expandable medical device |
| US20120078362A1 (en) | 2009-05-18 | 2012-03-29 | Dose Medical Corporation | Drug eluting ocular implant |
| US10206813B2 (en) | 2009-05-18 | 2019-02-19 | Dose Medical Corporation | Implants with controlled drug delivery features and methods of using same |
| US20100303878A1 (en) * | 2009-06-02 | 2010-12-02 | Joram Slager | Biodegradable bioactive agent releasing matrices with particulates |
| US8586731B2 (en) * | 2009-06-11 | 2013-11-19 | Surmodics, Inc. | Hydrophobic polysaccharides with diester- or carbonate ester-containing linkages having enhanced degradation |
| US8986377B2 (en) | 2009-07-21 | 2015-03-24 | Lifecell Corporation | Graft materials for surgical breast procedures |
| US20110046721A1 (en) * | 2009-08-10 | 2011-02-24 | Surmodics, Inc. | Biodegradable Metal-Polymer Composite Constructs For Implantable Medical Devices |
| DK2301595T3 (en) | 2009-09-23 | 2014-04-07 | Dentsply Ih Ab | Translucent catheter and method for making such a catheter |
| WO2011041517A1 (en) * | 2009-09-30 | 2011-04-07 | Surmodics, Inc. | Hydrophobic polysaccharides with silyl ether linkages having enhanced degradation and medical articles made therefrom |
| CA2784689A1 (en) | 2009-12-18 | 2011-06-23 | Interface Biologics, Inc. | Local delivery of drugs from self assembled coatings |
| US8568760B2 (en) * | 2009-12-30 | 2013-10-29 | Surmodics, Inc. | Hydrophobic polysaccharides with pendent groups having terminal reactive functionalities and medical uses thereof |
| US8697105B2 (en) | 2010-03-29 | 2014-04-15 | Surmodics, Inc. | Injectable drug delivery formulation |
| JP5945534B2 (en) | 2010-05-10 | 2016-07-05 | サーモディクス,インコーポレイテッド | Glycerol ester activator delivery system and method |
| US20110319473A1 (en) | 2010-06-29 | 2011-12-29 | Surmodics, Inc. | Compositions and methods for enhancement of nucleic acid delivery |
| EP2588157B1 (en) * | 2010-06-30 | 2020-03-18 | SurModics, Inc. | Lipid coating for medical devices delivering bioactive agent |
| US9295663B2 (en) | 2010-07-14 | 2016-03-29 | Abbott Cardiovascular Systems Inc. | Drug coated balloon with in-situ formed drug containing microspheres |
| US20120100187A1 (en) | 2010-10-26 | 2012-04-26 | Surmodics, Inc. | Coatings and methods for controlled elution of hydrophilic active agents |
| PL2646066T3 (en) * | 2010-12-04 | 2018-10-31 | Aachen Scientific International Pte. Ltd. | Coating and coating method for the balloon of a balloon catheter, and balloon catheter with coated balloon |
| US20120171769A1 (en) * | 2010-12-30 | 2012-07-05 | Mcgonigle Joseph S | Cell attachment coatings and methods |
| US10213529B2 (en) | 2011-05-20 | 2019-02-26 | Surmodics, Inc. | Delivery of coated hydrophobic active agent particles |
| US9861727B2 (en) | 2011-05-20 | 2018-01-09 | Surmodics, Inc. | Delivery of hydrophobic active agent particles |
| US10245178B1 (en) | 2011-06-07 | 2019-04-02 | Glaukos Corporation | Anterior chamber drug-eluting ocular implant |
| BR112014007657A2 (en) | 2011-10-10 | 2017-04-11 | Ampio Pharmaceuticals Inc | Implantable medical devices with improved immune tolerance and methods for production and implantation |
| WO2013055734A1 (en) | 2011-10-10 | 2013-04-18 | Ampio Pharmaceuticals, Inc. | Treatment of degenerative joint disease |
| CA2846394A1 (en) | 2011-10-28 | 2013-05-02 | Ampio Pharmaceuticals, Inc. | Treatment of rhinitis |
| EP2802288B1 (en) | 2012-01-13 | 2016-07-27 | Lifecell Corporation | Breast prostheses and methods of manufacturing breast prostheses |
| JP6104232B2 (en) | 2012-03-27 | 2017-03-29 | テルモ株式会社 | Coating composition and medical device |
| US9827401B2 (en) | 2012-06-01 | 2017-11-28 | Surmodics, Inc. | Apparatus and methods for coating medical devices |
| DE102012010800A1 (en) * | 2012-06-01 | 2013-12-05 | Alexander Rübben | Coating of balloon catheters |
| MX351261B (en) | 2012-06-01 | 2017-10-06 | Surmodics Inc | Apparatus and method for coating balloon catheters. |
| WO2014004997A1 (en) | 2012-06-29 | 2014-01-03 | Surmodics, Inc. | Cell attachment coatings and methods using phosphorous-containing photoreagent |
| US9708600B2 (en) * | 2012-07-13 | 2017-07-18 | Diomics Corporation | Biologic sample collection devices and methods of production and use thereof |
| US8759075B2 (en) * | 2012-07-13 | 2014-06-24 | Diomics Corporation | Biologic sample collection devices and methods of production and use thereof |
| US10519434B2 (en) | 2012-07-13 | 2019-12-31 | Diomics Corporation | Biologic sample collection devices and methods of production and use thereof |
| CN102827770A (en) * | 2012-08-20 | 2012-12-19 | 三峡大学 | Color developing method for screening amylase producing strain |
| US20140105956A1 (en) * | 2012-10-11 | 2014-04-17 | Rupak BANERJEE | Biodegradable polymer based microimplant for ocular drug delivery |
| EP2908870B1 (en) | 2012-10-16 | 2018-05-23 | SurModics, Inc. | Wound packing device and methods |
| US11090468B2 (en) | 2012-10-25 | 2021-08-17 | Surmodics, Inc. | Apparatus and methods for coating medical devices |
| US11246963B2 (en) | 2012-11-05 | 2022-02-15 | Surmodics, Inc. | Compositions and methods for delivery of hydrophobic active agents |
| US9555119B2 (en) | 2012-11-05 | 2017-01-31 | Surmodics, Inc. | Composition and method for delivery of hydrophobic active agents |
| US9925355B2 (en) | 2012-11-12 | 2018-03-27 | Hollister Incorporated | Intermittent catheter assembly and kit |
| HUE041859T2 (en) | 2012-11-14 | 2019-06-28 | Hollister Inc | Disposable catheter with selectively degradable inner core |
| NL2010040C2 (en) * | 2012-12-21 | 2014-06-24 | Internat Inst For Diagnostic And Analitical Affairs B V | Cleavable coating material having microbial functionality. |
| AU2014232728B2 (en) | 2013-03-15 | 2019-02-21 | Ampio Pharmaceuticals, Inc. | Compositions for the mobilization, homing, expansion and differentiation of stem cells and methods of using the same |
| GB2514592A (en) * | 2013-05-30 | 2014-12-03 | Medtrade Products Ltd | Degradable haemostat composition |
| US9446431B2 (en) | 2013-08-15 | 2016-09-20 | Cook Medical Technologies Llc | Hydrophilic coatings and methods for coating medical devices with hydrophilic coatings |
| HUE063905T2 (en) | 2013-11-08 | 2024-02-28 | Hollister Inc | Oleophilic lubricated catheters |
| WO2015089197A2 (en) | 2013-12-12 | 2015-06-18 | Hollister Incorporated | Flushable catheters |
| WO2015089178A1 (en) | 2013-12-12 | 2015-06-18 | Hollister Incorporated | Flushable disintegration catheter |
| ES2792976T3 (en) | 2013-12-12 | 2020-11-12 | Hollister Inc | Disposable toilet catheters |
| DK3620198T3 (en) | 2013-12-12 | 2021-04-12 | Hollister Inc | EXCLUSIVE CATHETRES |
| EP3166726A4 (en) | 2014-05-01 | 2018-03-07 | Diomics Corporation | Devices and kits for collection, storage and analysis of samples and methods of production and use thereof |
| CA2950187A1 (en) | 2014-05-29 | 2015-12-03 | Glaukos Corporation | Implants with controlled drug delivery features and methods of using same |
| CA2952962C (en) * | 2014-06-19 | 2022-08-23 | Daniel SIPPLE | Biomarker detection and identification system and apparatus |
| WO2016014455A1 (en) | 2014-07-22 | 2016-01-28 | Diomics Corporation | Airborne agent collectors, methods, systems and devices for monitoring airborne agents |
| US10201457B2 (en) | 2014-08-01 | 2019-02-12 | Surmodics, Inc. | Wound packing device with nanotextured surface |
| WO2016025021A1 (en) | 2014-08-15 | 2016-02-18 | Diomics Corporation | Films for biologic analyte collection and analysis and methods of production and use thereof |
| EP4066836A1 (en) | 2014-08-18 | 2022-10-05 | Ampio Pharmaceuticals, Inc. | Treatment of joint conditions |
| WO2016164861A1 (en) * | 2015-04-10 | 2016-10-13 | The Regents Of The University Of California | Microfluidic assisted perfusion devices |
| US10702688B2 (en) | 2015-05-11 | 2020-07-07 | Access For Life Inc. | Vascular access device |
| AU2016265286B2 (en) | 2015-05-15 | 2021-01-21 | Lifecell Corporation | Tissue matrices for plastic surgery |
| DK3310405T4 (en) | 2015-06-17 | 2023-05-22 | Hollister Inc | WATER-SOLUBLE FLUSHABLE CATHETER |
| AU2016280079B2 (en) | 2015-06-17 | 2021-04-15 | Hollister Incorporated | Selectively water disintegrable materials and catheters made of such materials |
| US11389512B2 (en) | 2015-06-22 | 2022-07-19 | Ampio Pharmaceuticals, Inc. | Use of low molecular weight fractions of human serum albumin in treating diseases |
| CA2996287A1 (en) | 2015-08-21 | 2017-03-02 | Lifecell Corporation | Breast treatment device |
| US11925578B2 (en) | 2015-09-02 | 2024-03-12 | Glaukos Corporation | Drug delivery implants with bi-directional delivery capacity |
| EP4226868A1 (en) | 2015-09-15 | 2023-08-16 | Savage Medical, Inc. | Devices and methods for anchoring a sheath in a tissue cavity |
| WO2017053885A1 (en) | 2015-09-25 | 2017-03-30 | Glaukos Corporation | Punctal implants with controlled drug delivery features and methods of using same |
| US11420174B2 (en) | 2015-12-18 | 2022-08-23 | Safeguard Biosystems Holdings Ltd. | Three-dimensional polymer networks with channels situated therein |
| EP3181700A1 (en) * | 2015-12-18 | 2017-06-21 | Safeguard Biosystems Holdings Ltd. | Three-dimensional polymer networks with channels situated therein |
| US12102973B2 (en) | 2015-12-18 | 2024-10-01 | Safeguard Biosystems Holdings Ltd. | Three-dimensional polymer networks with channels situated therein |
| MX393577B (en) | 2015-12-29 | 2025-03-24 | Galderma Sa | CARBOHYDRATE CROSS-CROSSLINKING AGENT. |
| CN105606556B (en) * | 2016-01-28 | 2018-09-21 | 广西壮族自治区中医药研究院 | The ultraviolet spectrophotometry of polyoses content in fokien angiopteris rhizome medicinal material |
| US10792477B2 (en) * | 2016-02-08 | 2020-10-06 | Orbusneich Medical Pte. Ltd. | Drug eluting balloon |
| AU2017252294B2 (en) | 2016-04-20 | 2021-12-02 | Glaukos Corporation | Bioresorbable ocular drug delivery device |
| WO2018013805A2 (en) | 2016-07-14 | 2018-01-18 | Hollister Incorporated | Hygienic medical devices having hydrophilic coating and methods of forming the same |
| MX2019002263A (en) | 2016-08-31 | 2019-09-19 | Lifecell Corp | Breast treatment device. |
| CN106474565A (en) * | 2016-12-09 | 2017-03-08 | 泰州学院 | A kind of tussah silk fibroin is combined the preparation method of maltodextrin active scaffold material |
| US10898446B2 (en) | 2016-12-20 | 2021-01-26 | Surmodics, Inc. | Delivery of hydrophobic active agents from hydrophilic polyether block amide copolymer surfaces |
| US20180221021A1 (en) * | 2017-02-08 | 2018-08-09 | Ethicon, Inc. | Braided suture with filament containing a medicant |
| JP2018153296A (en) * | 2017-03-16 | 2018-10-04 | Jsr株式会社 | Compositions containing unsaturated compounds |
| CN112638436A (en) | 2018-05-22 | 2021-04-09 | 界面生物公司 | Compositions and methods for drug delivery to vessel walls |
| US11628466B2 (en) | 2018-11-29 | 2023-04-18 | Surmodics, Inc. | Apparatus and methods for coating medical devices |
| FR3089753B1 (en) * | 2018-12-13 | 2022-05-13 | Franc Cecile | Bi-component gels for the controlled application of an oxidizing treatment on surfaces |
| WO2020210647A2 (en) * | 2019-04-12 | 2020-10-15 | The Trustees Of Columbia University In The City Of New York | Methods and compositions for protection of bioprosthetic heart valve tissue from glycation and associated protein incorporation |
| US11298220B2 (en) | 2019-05-03 | 2022-04-12 | Lifecell Corporation | Breast treatment device |
| US11819590B2 (en) | 2019-05-13 | 2023-11-21 | Surmodics, Inc. | Apparatus and methods for coating medical devices |
| US12226552B2 (en) | 2019-09-30 | 2025-02-18 | Surmodics, Inc. | Active agent depots formed in situ |
| US11235087B2 (en) * | 2019-10-28 | 2022-02-01 | Galderma Holding SA | Ready-to-use esthetic compositions |
| CN115335414B (en) | 2019-12-02 | 2024-03-19 | 高德美控股有限公司 | High molecular weight cosmetic compositions |
| US11739166B2 (en) | 2020-07-02 | 2023-08-29 | Davol Inc. | Reactive polysaccharide-based hemostatic agent |
| US12161777B2 (en) | 2020-07-02 | 2024-12-10 | Davol Inc. | Flowable hemostatic suspension |
| CN112618802B (en) * | 2020-12-08 | 2021-12-24 | 南方医科大学口腔医院 | Fluorine-containing antibacterial invisible appliance for improving enamel demineralization and preparation method thereof |
| EP4267210A1 (en) | 2020-12-28 | 2023-11-01 | Davol Inc. | Reactive dry powdered hemostatic materials comprising a protein and a multifunctionalized modified polyethylene glycol based crosslinking agent |
| CN114452442A (en) * | 2022-01-30 | 2022-05-10 | 上海松力生物技术有限公司 | Implant for in-situ induction of uterine wall tissue regeneration |
| WO2024030418A2 (en) * | 2022-08-02 | 2024-02-08 | The Secant Group, Llc | Biodegradable, elastomeric anastomotic tissue junction stabilizer |
| WO2025159745A1 (en) * | 2024-01-24 | 2025-07-31 | Bard Peripheral Vascular, Inc. | Embolic hydrogels for female sterilization |
Citations (55)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US627899A (en) * | 1897-07-03 | 1899-06-27 | George Martin Brill | Car-truck. |
| US2668156A (en) * | 1950-04-06 | 1954-02-02 | Nat Starch Products Inc | Unsaturated starch compounds and insoluble derivatives thereof |
| US4060506A (en) * | 1976-04-27 | 1977-11-29 | A. E. Staley Manufacturing Company | Starch acrylamides and the method for preparing the same |
| US4079025A (en) * | 1976-04-27 | 1978-03-14 | A. E. Staley Manufacturing Company | Copolymerized starch composition |
| US5116927A (en) * | 1989-06-27 | 1992-05-26 | Sequa Chemicals, Inc. | Starch polymer graft composition and method of preparation |
| US5160745A (en) * | 1986-05-16 | 1992-11-03 | The University Of Kentucky Research Foundation | Biodegradable microspheres as a carrier for macromolecules |
| US5272181A (en) * | 1992-11-18 | 1993-12-21 | Evergreen Solutions, Inc. | Biodegradable expanded foam material |
| US5488102A (en) * | 1992-11-30 | 1996-01-30 | Ciba Geigy Corporation | Polymerisable carbohydrate esters, polymers therefrom and their use |
| US5514379A (en) * | 1992-08-07 | 1996-05-07 | The General Hospital Corporation | Hydrogel compositions and methods of use |
| US5563056A (en) * | 1992-02-13 | 1996-10-08 | Bsi Corporation | Preparation of crosslinked matrices containing covalently immobilized chemical species and unbound releasable chemical species |
| US5589577A (en) * | 1993-04-07 | 1996-12-31 | Alko Group Ltd. | Applications and methods for the preparation of fatty acid esters of polysaccharides |
| US5626863A (en) * | 1992-02-28 | 1997-05-06 | Board Of Regents, The University Of Texas System | Photopolymerizable biodegradable hydrogels as tissue contacting materials and controlled-release carriers |
| US5668193A (en) * | 1993-01-19 | 1997-09-16 | Medicarb Ab | Solid substrate coated with an aminopolysaccharide |
| US5733994A (en) * | 1992-03-30 | 1998-03-31 | Deutsche Gelatine-Fabriken Stoess Ag | Biodegradable, water-resistant polymer material |
| US5736371A (en) * | 1991-06-04 | 1998-04-07 | A Et S Biovecteurs | Biodegradable particulate vector for transporting molecules having biological activity |
| US5770229A (en) * | 1994-05-13 | 1998-06-23 | Kuraray Co., Ltd. | Medical polymer gel |
| US5773021A (en) * | 1994-03-14 | 1998-06-30 | Vetoquinol S.A. | Bioadhesive ophthalmic insert |
| US5837747A (en) * | 1991-10-29 | 1998-11-17 | Vivorx, Inc. | Crosslinkable polysaccharides, polycations and lipids useful for encapsulation and drug release |
| US5866619A (en) * | 1990-05-04 | 1999-02-02 | Perio Products Ltd. | Colonic drug delivery system |
| US5879707A (en) * | 1996-10-30 | 1999-03-09 | Universite De Montreal | Substituted amylose as a matrix for sustained drug release |
| US5885615A (en) * | 1996-04-10 | 1999-03-23 | Labopharm Inc. | Pharmaceutical controlled release tablets containing a carrier made of cross-linked amylose and hydroxypropylmethylcellulose |
| US5993530A (en) * | 1995-09-13 | 1999-11-30 | Japan Corn Starch Co., Ltd. | Aqueous dispersion of biodegradable resin composition |
| US6001395A (en) * | 1995-07-13 | 1999-12-14 | Danbiosyst Uk Limited | Polymeric lamellar substrate particles for drug delivery |
| US6007833A (en) * | 1998-03-19 | 1999-12-28 | Surmodics, Inc. | Crosslinkable macromers bearing initiator groups |
| US6140313A (en) * | 1996-11-29 | 2000-10-31 | Perbellini; Alberto | Butyric esters with antiproliferative activity and the pharmaceutical compositions containing them |
| US6197757B1 (en) * | 1998-07-09 | 2001-03-06 | Coletica | Particles, especially microparticles or nanoparticles, of crosslinked monosaccharides and oligosaccharides, processes for their preparation and cosmetic, pharmaceutical or food compositions in which they are present |
| US20010018072A1 (en) * | 1997-05-13 | 2001-08-30 | Imarx Therapeutics, Inc. | Solid matrix therapeutic compositions |
| US6303047B1 (en) * | 1999-03-22 | 2001-10-16 | Lsi Logic Corporation | Low dielectric constant multiple carbon-containing silicon oxide dielectric material for use in integrated circuit structures, and method of making same |
| US6346263B1 (en) * | 1996-09-27 | 2002-02-12 | Sarl Kappa Biotech | Biodegradable ionic matrix of variable internal polarity with grafted polymer |
| US6410044B1 (en) * | 1998-03-19 | 2002-06-25 | Surmodics, Inc. | Crosslinkable macromers |
| US6419957B1 (en) * | 1998-02-24 | 2002-07-16 | Labopharm, Inc. | Cross-linked high amylose starch having functional groups as a matrix for the slow release of pharmaceutical agents |
| US20020161163A1 (en) * | 1998-03-06 | 2002-10-31 | Ube Industries, Ltd. | Nylon 12, nylon 12 composition, method for producing nylon 12, and tubular molded product using nylon 12 |
| US6514734B1 (en) * | 1997-08-15 | 2003-02-04 | Surmodics, Inc. | Polybifunctional reagent having a polymeric backbone and latent reactive moieties and bioactive groups |
| US20030077242A1 (en) * | 1998-08-14 | 2003-04-24 | Incept Llc | Methods for forming regional tissue adherent barriers and drug delivery systems |
| US6559257B2 (en) * | 1998-06-12 | 2003-05-06 | Lord Corporation | Adhesive formulations |
| US6586493B1 (en) * | 2001-03-07 | 2003-07-01 | Arizona Board Of Regents Arizona State University | Polysaccharide-based hydrogels and pre-gel blends for the same |
| US20030125509A1 (en) * | 1999-04-12 | 2003-07-03 | Chee-Youb Won | Hydrogel-forming system with hydrophobic and hydrophilic components |
| US6596860B1 (en) * | 1998-07-23 | 2003-07-22 | Cooperatieve Verkoop-En Productievereniging Van Aardappelmeel En Derivaten Avebe B.A. | Adhesive composition |
| US20030218130A1 (en) * | 2002-05-02 | 2003-11-27 | Ciphergen Biosystems, Inc. | Biochips with surfaces coated with polysaccharide-based hydrogels |
| US6660827B2 (en) * | 1997-08-18 | 2003-12-09 | Scimed Life Systems, Inc. | Bioresorbable hydrogel compositions for implantable prostheses |
| US20040009218A1 (en) * | 2000-07-17 | 2004-01-15 | Shinichi Kitamura | Biodegradable articles obtained from enzymatically synthesized amylose |
| US6703048B1 (en) * | 1997-08-28 | 2004-03-09 | Celanese Ventures Gmbh | Spherical microparticles containing linear polysaccharides |
| US6706288B2 (en) * | 2000-10-06 | 2004-03-16 | Jagotec Ag | Microparticles |
| US20040062778A1 (en) * | 2002-09-26 | 2004-04-01 | Adi Shefer | Surface dissolution and/or bulk erosion controlled release compositions and devices |
| US6716445B2 (en) * | 1999-04-12 | 2004-04-06 | Cornell Research Foundation, Inc. | Hydrogel entrapping therapeutic agent and stent with coating comprising this |
| US20040091605A1 (en) * | 2002-05-24 | 2004-05-13 | Biotronik Mess- Und Therapiegeraete Gmbh & Co Ingenieurbuero Berlin | Method of coating a stent with a polysaccharide layer and associated stents |
| US6748954B2 (en) * | 2000-10-27 | 2004-06-15 | The Regents Of The University Of Michigan | Drug release from polymer matrices through mechanical stimulation |
| US20040133155A1 (en) * | 2000-08-30 | 2004-07-08 | Varner Sign Erickson | Devices for intraocular drug delivery |
| US20040202719A1 (en) * | 2002-12-17 | 2004-10-14 | Zion Todd C. | Stimuli-responsive systems for controlled drug delivery |
| US20050019371A1 (en) * | 2003-05-02 | 2005-01-27 | Anderson Aron B. | Controlled release bioactive agent delivery device |
| US20050025797A1 (en) * | 2003-04-08 | 2005-02-03 | Xingwu Wang | Medical device with low magnetic susceptibility |
| US20050255142A1 (en) * | 2004-05-12 | 2005-11-17 | Surmodics, Inc. | Coatings for medical articles including natural biodegradable polysaccharides |
| US20060036029A1 (en) * | 2004-08-12 | 2006-02-16 | Tomko Richard F | Carbohydrate based alkyds |
| US20070065481A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | Coatings including natural biodegradable polysaccharides and uses thereof |
| US20070065484A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | In situ occlusion using natural biodegradable polysaccharides |
Family Cites Families (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4320221A (en) * | 1980-12-12 | 1982-03-16 | The Dow Chemical Company | Addition polymerizable isocyanate-polyol anaerobic adhesives |
| US5318780A (en) * | 1991-10-30 | 1994-06-07 | Mediventures Inc. | Medical uses of in situ formed gels |
| US6277899B1 (en) * | 1992-08-03 | 2001-08-21 | Novamont S.P.A. | Biodegradable polymeric composition |
| US5414075A (en) | 1992-11-06 | 1995-05-09 | Bsi Corporation | Restrained multifunctional reagent for surface modification |
| US5466233A (en) | 1994-04-25 | 1995-11-14 | Escalon Ophthalmics, Inc. | Tack for intraocular drug delivery and method for inserting and removing same |
| TW362100B (en) | 1995-07-21 | 1999-06-21 | Novartis Ag | The preparation and use of photochemically cross-linked polysaccharide derivatives having no photopolymerisable functional groups |
| US5714360A (en) * | 1995-11-03 | 1998-02-03 | Bsi Corporation | Photoactivatable water soluble cross-linking agents containing an onium group |
| US5749848A (en) | 1995-11-13 | 1998-05-12 | Cardiovascular Imaging Systems, Inc. | Catheter system having imaging, balloon angioplasty, and stent deployment capabilities, and method of use for guided stent deployment |
| ZA978537B (en) * | 1996-09-23 | 1998-05-12 | Focal Inc | Polymerizable biodegradable polymers including carbonate or dioxanone linkages. |
| EP0842657A1 (en) | 1996-11-19 | 1998-05-20 | OctoPlus B.V. | Microspheres for controlled release and processes to prepare these microspheres |
| CA2274004A1 (en) * | 1996-12-03 | 1998-06-11 | Osteobiologics, Inc. | Biodegradable polymeric film |
| US5866165A (en) * | 1997-01-15 | 1999-02-02 | Orquest, Inc. | Collagen-polysaccharide matrix for bone and cartilage repair |
| US6668935B1 (en) | 1999-09-24 | 2003-12-30 | Schlumberger Technology Corporation | Valve for use in wells |
| US7595392B2 (en) * | 2000-12-29 | 2009-09-29 | University Of Iowa Research Foundation | Biodegradable oxidized cellulose esters |
| WO2002100453A1 (en) * | 2001-06-07 | 2002-12-19 | Surmodics, Inc. | Crosslinkable macromers |
| US7501157B2 (en) * | 2001-06-26 | 2009-03-10 | Accelr8 Technology Corporation | Hydroxyl functional surface coating |
| US7438925B2 (en) * | 2002-08-26 | 2008-10-21 | Biovention Holdings Ltd. | Drug eluting coatings for medical implants |
| US7862831B2 (en) | 2002-10-09 | 2011-01-04 | Synthasome, Inc. | Method and material for enhanced tissue-biomaterial integration |
| US20050143363A1 (en) | 2002-09-29 | 2005-06-30 | Innorx, Inc. | Method for subretinal administration of therapeutics including steroids; method for localizing pharmacodynamic action at the choroid of the retina; and related methods for treatment and/or prevention of retinal diseases |
-
2005
- 2005-11-11 AT AT05849204T patent/ATE540705T1/en active
- 2005-11-11 JP JP2008532203A patent/JP5139301B2/en not_active Expired - Fee Related
- 2005-11-11 EP EP05849204A patent/EP1926509B1/en not_active Not-in-force
- 2005-11-11 US US11/271,213 patent/US8512736B2/en not_active Expired - Fee Related
- 2005-11-11 US US11/271,238 patent/US20070065483A1/en not_active Abandoned
- 2005-11-11 WO PCT/US2005/040927 patent/WO2007040557A1/en active Application Filing
- 2005-11-11 CN CNA2005800520005A patent/CN101309709A/en active Pending
- 2005-11-11 US US11/271,237 patent/US8241656B2/en not_active Expired - Fee Related
- 2005-11-11 CA CA002621595A patent/CA2621595A1/en not_active Abandoned
-
2006
- 2006-09-21 US US11/524,926 patent/US20100226960A1/en not_active Abandoned
- 2006-09-21 CN CNA2006800412325A patent/CN101300036A/en active Pending
- 2006-09-21 CA CA002622401A patent/CA2622401A1/en not_active Abandoned
- 2006-09-21 AT AT06815123T patent/ATE554801T1/en active
- 2006-09-21 WO PCT/US2006/036861 patent/WO2007035886A1/en active Application Filing
- 2006-09-21 CN CNA2006800433482A patent/CN101316619A/en active Pending
- 2006-09-21 JP JP2008532392A patent/JP5226518B2/en not_active Expired - Fee Related
- 2006-09-21 EP EP06815123A patent/EP1937327B1/en not_active Not-in-force
Patent Citations (61)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US627899A (en) * | 1897-07-03 | 1899-06-27 | George Martin Brill | Car-truck. |
| US2668156A (en) * | 1950-04-06 | 1954-02-02 | Nat Starch Products Inc | Unsaturated starch compounds and insoluble derivatives thereof |
| US4060506A (en) * | 1976-04-27 | 1977-11-29 | A. E. Staley Manufacturing Company | Starch acrylamides and the method for preparing the same |
| US4079025A (en) * | 1976-04-27 | 1978-03-14 | A. E. Staley Manufacturing Company | Copolymerized starch composition |
| US5160745A (en) * | 1986-05-16 | 1992-11-03 | The University Of Kentucky Research Foundation | Biodegradable microspheres as a carrier for macromolecules |
| US5116927A (en) * | 1989-06-27 | 1992-05-26 | Sequa Chemicals, Inc. | Starch polymer graft composition and method of preparation |
| US5866619A (en) * | 1990-05-04 | 1999-02-02 | Perio Products Ltd. | Colonic drug delivery system |
| US5736371A (en) * | 1991-06-04 | 1998-04-07 | A Et S Biovecteurs | Biodegradable particulate vector for transporting molecules having biological activity |
| US5837747A (en) * | 1991-10-29 | 1998-11-17 | Vivorx, Inc. | Crosslinkable polysaccharides, polycations and lipids useful for encapsulation and drug release |
| US5563056A (en) * | 1992-02-13 | 1996-10-08 | Bsi Corporation | Preparation of crosslinked matrices containing covalently immobilized chemical species and unbound releasable chemical species |
| US5626863A (en) * | 1992-02-28 | 1997-05-06 | Board Of Regents, The University Of Texas System | Photopolymerizable biodegradable hydrogels as tissue contacting materials and controlled-release carriers |
| US5733994A (en) * | 1992-03-30 | 1998-03-31 | Deutsche Gelatine-Fabriken Stoess Ag | Biodegradable, water-resistant polymer material |
| US5514379A (en) * | 1992-08-07 | 1996-05-07 | The General Hospital Corporation | Hydrogel compositions and methods of use |
| US5272181A (en) * | 1992-11-18 | 1993-12-21 | Evergreen Solutions, Inc. | Biodegradable expanded foam material |
| US5488102A (en) * | 1992-11-30 | 1996-01-30 | Ciba Geigy Corporation | Polymerisable carbohydrate esters, polymers therefrom and their use |
| US5668193A (en) * | 1993-01-19 | 1997-09-16 | Medicarb Ab | Solid substrate coated with an aminopolysaccharide |
| US5589577A (en) * | 1993-04-07 | 1996-12-31 | Alko Group Ltd. | Applications and methods for the preparation of fatty acid esters of polysaccharides |
| US5773021A (en) * | 1994-03-14 | 1998-06-30 | Vetoquinol S.A. | Bioadhesive ophthalmic insert |
| US5770229A (en) * | 1994-05-13 | 1998-06-23 | Kuraray Co., Ltd. | Medical polymer gel |
| US6001395A (en) * | 1995-07-13 | 1999-12-14 | Danbiosyst Uk Limited | Polymeric lamellar substrate particles for drug delivery |
| US5993530A (en) * | 1995-09-13 | 1999-11-30 | Japan Corn Starch Co., Ltd. | Aqueous dispersion of biodegradable resin composition |
| US5885615A (en) * | 1996-04-10 | 1999-03-23 | Labopharm Inc. | Pharmaceutical controlled release tablets containing a carrier made of cross-linked amylose and hydroxypropylmethylcellulose |
| US6346263B1 (en) * | 1996-09-27 | 2002-02-12 | Sarl Kappa Biotech | Biodegradable ionic matrix of variable internal polarity with grafted polymer |
| US5879707A (en) * | 1996-10-30 | 1999-03-09 | Universite De Montreal | Substituted amylose as a matrix for sustained drug release |
| US6140313A (en) * | 1996-11-29 | 2000-10-31 | Perbellini; Alberto | Butyric esters with antiproliferative activity and the pharmaceutical compositions containing them |
| US20010018072A1 (en) * | 1997-05-13 | 2001-08-30 | Imarx Therapeutics, Inc. | Solid matrix therapeutic compositions |
| US6514734B1 (en) * | 1997-08-15 | 2003-02-04 | Surmodics, Inc. | Polybifunctional reagent having a polymeric backbone and latent reactive moieties and bioactive groups |
| US6660827B2 (en) * | 1997-08-18 | 2003-12-09 | Scimed Life Systems, Inc. | Bioresorbable hydrogel compositions for implantable prostheses |
| US6703048B1 (en) * | 1997-08-28 | 2004-03-09 | Celanese Ventures Gmbh | Spherical microparticles containing linear polysaccharides |
| US6419957B1 (en) * | 1998-02-24 | 2002-07-16 | Labopharm, Inc. | Cross-linked high amylose starch having functional groups as a matrix for the slow release of pharmaceutical agents |
| US20020161163A1 (en) * | 1998-03-06 | 2002-10-31 | Ube Industries, Ltd. | Nylon 12, nylon 12 composition, method for producing nylon 12, and tubular molded product using nylon 12 |
| US6156345A (en) * | 1998-03-19 | 2000-12-05 | Surmodics, Inc. | Crosslinkable macromers bearing initiator groups |
| US20030031697A1 (en) * | 1998-03-19 | 2003-02-13 | Chudzik Stephen J. | Crosslinkable macromers |
| US6410044B1 (en) * | 1998-03-19 | 2002-06-25 | Surmodics, Inc. | Crosslinkable macromers |
| US6007833A (en) * | 1998-03-19 | 1999-12-28 | Surmodics, Inc. | Crosslinkable macromers bearing initiator groups |
| US6559257B2 (en) * | 1998-06-12 | 2003-05-06 | Lord Corporation | Adhesive formulations |
| US6197757B1 (en) * | 1998-07-09 | 2001-03-06 | Coletica | Particles, especially microparticles or nanoparticles, of crosslinked monosaccharides and oligosaccharides, processes for their preparation and cosmetic, pharmaceutical or food compositions in which they are present |
| US6596860B1 (en) * | 1998-07-23 | 2003-07-22 | Cooperatieve Verkoop-En Productievereniging Van Aardappelmeel En Derivaten Avebe B.A. | Adhesive composition |
| US20030077242A1 (en) * | 1998-08-14 | 2003-04-24 | Incept Llc | Methods for forming regional tissue adherent barriers and drug delivery systems |
| US6303047B1 (en) * | 1999-03-22 | 2001-10-16 | Lsi Logic Corporation | Low dielectric constant multiple carbon-containing silicon oxide dielectric material for use in integrated circuit structures, and method of making same |
| US20030125509A1 (en) * | 1999-04-12 | 2003-07-03 | Chee-Youb Won | Hydrogel-forming system with hydrophobic and hydrophilic components |
| US6716445B2 (en) * | 1999-04-12 | 2004-04-06 | Cornell Research Foundation, Inc. | Hydrogel entrapping therapeutic agent and stent with coating comprising this |
| US20050129734A1 (en) * | 1999-04-12 | 2005-06-16 | Cornell Research Foundation, Inc. | Hydrogel entrapping therapeutic agent and stent with coating comprising this |
| US6709668B2 (en) * | 1999-04-12 | 2004-03-23 | Cornell Research Foundation, Inc. | Hydrogel-forming system with hydrophobic and hydrophilic components |
| US20040122165A1 (en) * | 1999-04-12 | 2004-06-24 | Chee-Youb Won | Hydrogel-forming system with hydrophobic and hydrophilic components |
| US20040009218A1 (en) * | 2000-07-17 | 2004-01-15 | Shinichi Kitamura | Biodegradable articles obtained from enzymatically synthesized amylose |
| US20040133155A1 (en) * | 2000-08-30 | 2004-07-08 | Varner Sign Erickson | Devices for intraocular drug delivery |
| US6706288B2 (en) * | 2000-10-06 | 2004-03-16 | Jagotec Ag | Microparticles |
| US6748954B2 (en) * | 2000-10-27 | 2004-06-15 | The Regents Of The University Of Michigan | Drug release from polymer matrices through mechanical stimulation |
| US6586493B1 (en) * | 2001-03-07 | 2003-07-01 | Arizona Board Of Regents Arizona State University | Polysaccharide-based hydrogels and pre-gel blends for the same |
| US20030218130A1 (en) * | 2002-05-02 | 2003-11-27 | Ciphergen Biosystems, Inc. | Biochips with surfaces coated with polysaccharide-based hydrogels |
| US20040091605A1 (en) * | 2002-05-24 | 2004-05-13 | Biotronik Mess- Und Therapiegeraete Gmbh & Co Ingenieurbuero Berlin | Method of coating a stent with a polysaccharide layer and associated stents |
| US20040062778A1 (en) * | 2002-09-26 | 2004-04-01 | Adi Shefer | Surface dissolution and/or bulk erosion controlled release compositions and devices |
| US20040202719A1 (en) * | 2002-12-17 | 2004-10-14 | Zion Todd C. | Stimuli-responsive systems for controlled drug delivery |
| US20050025797A1 (en) * | 2003-04-08 | 2005-02-03 | Xingwu Wang | Medical device with low magnetic susceptibility |
| US20050019371A1 (en) * | 2003-05-02 | 2005-01-27 | Anderson Aron B. | Controlled release bioactive agent delivery device |
| US20050255142A1 (en) * | 2004-05-12 | 2005-11-17 | Surmodics, Inc. | Coatings for medical articles including natural biodegradable polysaccharides |
| US20060036029A1 (en) * | 2004-08-12 | 2006-02-16 | Tomko Richard F | Carbohydrate based alkyds |
| US20070065481A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | Coatings including natural biodegradable polysaccharides and uses thereof |
| US20070065482A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | Articles including natural biodegradable polysaccharides and uses thereof |
| US20070065484A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | In situ occlusion using natural biodegradable polysaccharides |
Cited By (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110129513A1 (en) * | 2002-05-24 | 2011-06-02 | Angiotech International Ag | Compositions and methods for coating medical implants |
| US8313760B2 (en) | 2002-05-24 | 2012-11-20 | Angiotech International Ag | Compositions and methods for coating medical implants |
| US8425927B2 (en) | 2002-05-24 | 2013-04-23 | Angiotech International Ag | Compositions and methods for coating medical implants |
| US20090060973A1 (en) * | 2002-05-24 | 2009-03-05 | Angiotech International Ag | Compositions and methods for coating medical implants |
| US8372420B2 (en) | 2002-05-24 | 2013-02-12 | Angiotech International Ag | Compositions and methods for coating medical implants |
| US8241655B2 (en) | 2004-05-12 | 2012-08-14 | Surmodics, Inc. | Coatings for medical articles including natural biodegradable polysaccharides |
| US20050255142A1 (en) * | 2004-05-12 | 2005-11-17 | Surmodics, Inc. | Coatings for medical articles including natural biodegradable polysaccharides |
| US9910020B1 (en) | 2005-03-30 | 2018-03-06 | Copilot Ventures Fund Iii Llc | Methods and articles for identifying objects using encapsulated perfluorocarbon tracers |
| US20100226960A1 (en) * | 2005-09-21 | 2010-09-09 | Surmodics, Inc. | Natural biodegradable matrices and uses thereof |
| US8241656B2 (en) | 2005-09-21 | 2012-08-14 | Surmodics, Inc | Articles including natural biodegradable polysaccharides and uses thereof |
| US20070065482A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | Articles including natural biodegradable polysaccharides and uses thereof |
| US20070065484A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | In situ occlusion using natural biodegradable polysaccharides |
| US8512736B2 (en) | 2005-09-21 | 2013-08-20 | Surmodics, Inc. | Coatings including natural biodegradable polysaccharides and uses thereof |
| US20070065481A1 (en) * | 2005-09-21 | 2007-03-22 | Chudzik Stephen J | Coatings including natural biodegradable polysaccharides and uses thereof |
| US8816022B2 (en) | 2006-03-30 | 2014-08-26 | Becton, Dickinson And Company | Sealing members, articles using the same and methods of reducing sticktion |
| US20120058351A1 (en) * | 2006-03-30 | 2012-03-08 | Becton, Dickinson And Company | Coating System, Articles and Assembly Using the Same and Methods of Reducing Sticktion |
| US9234118B2 (en) | 2006-03-30 | 2016-01-12 | Becton, Dickinson And Company | Coating system, articles and assembly using the same and methods of reducing sticktion |
| US8603638B2 (en) | 2006-03-30 | 2013-12-10 | Becton, Dickinson And Company | Sealing members, articles using the same and methods of reducing sticktion |
| US20080114096A1 (en) * | 2006-11-09 | 2008-05-15 | Hydromer, Inc. | Lubricious biopolymeric network compositions and methods of making same |
| US8790701B2 (en) | 2008-04-28 | 2014-07-29 | Surmodics, Inc. | Poly-α(1→4)glucopyranose-based matrices with hydrazide crosslinking |
| US20090269407A1 (en) * | 2008-04-28 | 2009-10-29 | Surmodics, Inc. | Poly-alpha(1-4)glucopyranose-based matrices with hydrazide crosslinking |
| US8936811B2 (en) | 2008-05-07 | 2015-01-20 | Surmodics, Inc. | Device coated with glycogen particles comprising nucleic acid complexes |
| US8114429B2 (en) | 2008-09-15 | 2012-02-14 | Cv Ingenuity Corp. | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US10314948B2 (en) | 2008-09-15 | 2019-06-11 | The Spectranetics Coporation | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US8491925B2 (en) | 2008-09-15 | 2013-07-23 | Cv Ingenuity Corp. | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US8673332B2 (en) | 2008-09-15 | 2014-03-18 | Covidien Lp | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US8734825B2 (en) | 2008-09-15 | 2014-05-27 | Covidien Lp | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US10987452B2 (en) | 2008-09-15 | 2021-04-27 | The Spectranetics Corporation | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US8257722B2 (en) | 2008-09-15 | 2012-09-04 | Cv Ingenuity Corp. | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US10046093B2 (en) | 2008-09-15 | 2018-08-14 | The Spectranetics Corporation | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US8128951B2 (en) | 2008-09-15 | 2012-03-06 | Cv Ingenuity Corp. | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US9034362B2 (en) | 2008-09-15 | 2015-05-19 | The Spectranetics Corporation | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US9132211B2 (en) | 2008-09-15 | 2015-09-15 | The Spectranetics Corporation | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US8563023B2 (en) | 2008-09-15 | 2013-10-22 | Covidien Lp | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US9198968B2 (en) | 2008-09-15 | 2015-12-01 | The Spectranetics Corporation | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US10117970B2 (en) | 2008-09-15 | 2018-11-06 | The Spectranetics Corporation | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US9603973B2 (en) | 2008-09-15 | 2017-03-28 | The Spectranetics Corporation | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US20100068170A1 (en) * | 2008-09-15 | 2010-03-18 | Michal Eugene T | Local delivery of water-soluble or water-insoluble therapeutic agents to the surface of body lumens |
| US9181352B2 (en) | 2009-05-27 | 2015-11-10 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. | Method for producing polysaccharide esters or polysaccharide mixed esters |
| WO2010136191A1 (en) * | 2009-05-27 | 2010-12-02 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. | Process for preparing polysaccharide esters or polysaccharide mixed esters |
| US8901092B2 (en) | 2010-12-29 | 2014-12-02 | Surmodics, Inc. | Functionalized polysaccharides for active agent delivery |
| GB2493263A (en) * | 2011-07-19 | 2013-01-30 | Neoss Ltd | Implantable medical device with a surface coating |
| US9956385B2 (en) | 2012-06-28 | 2018-05-01 | The Spectranetics Corporation | Post-processing of a medical device to control morphology and mechanical properties |
| US10525171B2 (en) | 2014-01-24 | 2020-01-07 | The Spectranetics Corporation | Coatings for medical devices |
| US20200215238A1 (en) * | 2017-05-17 | 2020-07-09 | Phenox Gmbh | Coating for medical devices |
| US11779686B2 (en) * | 2017-05-17 | 2023-10-10 | Phenox Gmbh | Coating for medical devices |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5139301B2 (en) | 2013-02-06 |
| US8241656B2 (en) | 2012-08-14 |
| JP2009508626A (en) | 2009-03-05 |
| ATE554801T1 (en) | 2012-05-15 |
| CN101309709A (en) | 2008-11-19 |
| EP1937327A1 (en) | 2008-07-02 |
| US20070065481A1 (en) | 2007-03-22 |
| WO2007035886A1 (en) | 2007-03-29 |
| US20100226960A1 (en) | 2010-09-09 |
| EP1937327B1 (en) | 2012-04-25 |
| US8512736B2 (en) | 2013-08-20 |
| CA2621595A1 (en) | 2007-04-12 |
| CN101316619A (en) | 2008-12-03 |
| EP1926509B1 (en) | 2012-01-11 |
| CA2622401A1 (en) | 2007-03-29 |
| JP2009508658A (en) | 2009-03-05 |
| WO2007040557A1 (en) | 2007-04-12 |
| EP1926509A1 (en) | 2008-06-04 |
| JP5226518B2 (en) | 2013-07-03 |
| CN101300036A (en) | 2008-11-05 |
| ATE540705T1 (en) | 2012-01-15 |
| US20070065482A1 (en) | 2007-03-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8512736B2 (en) | Coatings including natural biodegradable polysaccharides and uses thereof | |
| US8241655B2 (en) | Coatings for medical articles including natural biodegradable polysaccharides | |
| CA2621657C (en) | In situ occluding compositions comprising natural biodegradable polysaccharides polymerized by a redox pair | |
| US8663674B2 (en) | Microparticle containing matrices for drug delivery | |
| US7550444B2 (en) | Composition and method for preparing biocompatible surfaces | |
| US20080154241A1 (en) | Latent stabilization of bioactive agents releasable from implantable medical articles | |
| US20070260054A1 (en) | Hydrophobic derivatives of natural biodegradable polysaccharides and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: SURMODICS, INC., MINNESOTA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:CHUDZIK, STEVEN J.;SWAN, DALE G.;BURKSTRAND, MICHAEL J.;REEL/FRAME:017814/0005;SIGNING DATES FROM 20060407 TO 20060410 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- AFTER EXAMINER'S ANSWER OR BOARD OF APPEALS DECISION |