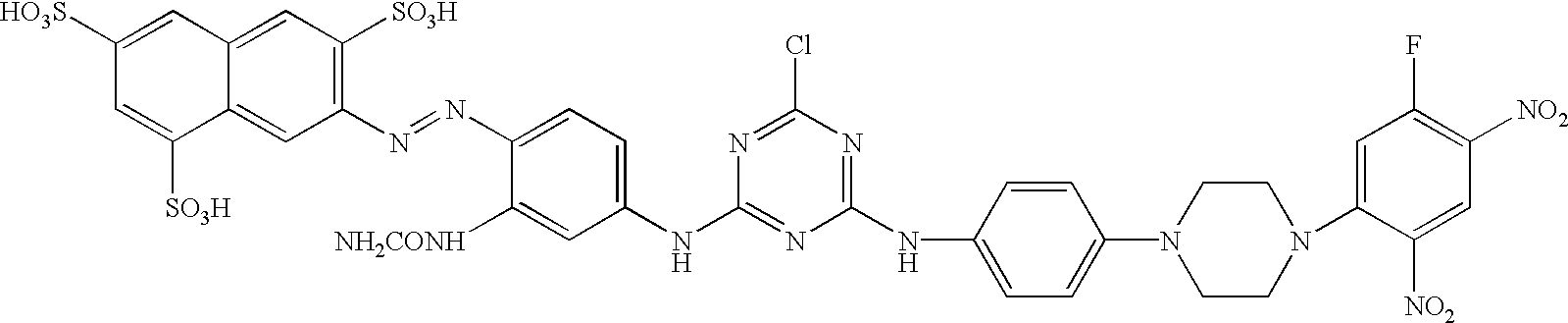
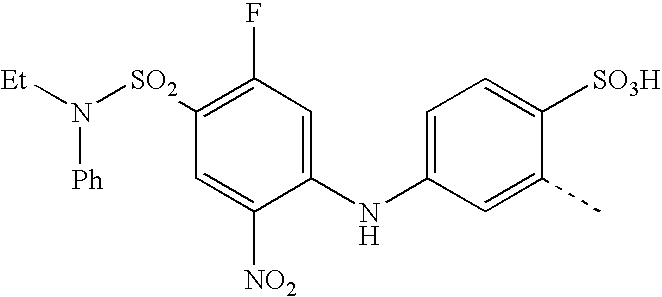
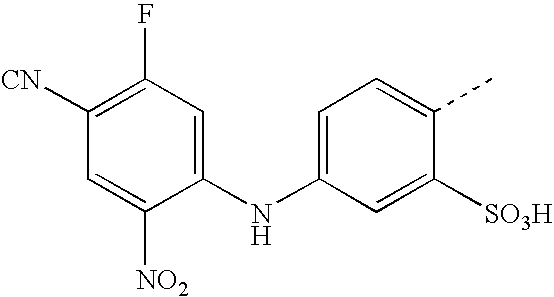
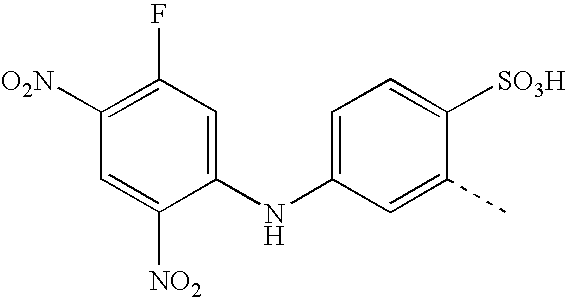
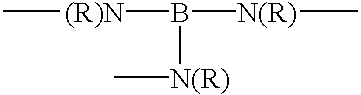US20030191293A1 - Reactive dyes containing a halobenzene nucleus - Google Patents
Reactive dyes containing a halobenzene nucleus Download PDFInfo
- Publication number
- US20030191293A1 US20030191293A1 US10/117,279 US11727902A US2003191293A1 US 20030191293 A1 US20030191293 A1 US 20030191293A1 US 11727902 A US11727902 A US 11727902A US 2003191293 A1 US2003191293 A1 US 2003191293A1
- Authority
- US
- United States
- Prior art keywords
- group
- formula
- independently
- dye according
- given
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 150000005171 halobenzenes Chemical class 0.000 title claims abstract description 21
- 239000000985 reactive dye Substances 0.000 title abstract description 6
- 239000000975 dye Substances 0.000 claims description 208
- 125000005647 linker group Chemical group 0.000 claims description 55
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 50
- 229910052739 hydrogen Inorganic materials 0.000 claims description 38
- 239000001257 hydrogen Substances 0.000 claims description 38
- 238000000034 method Methods 0.000 claims description 32
- 125000005843 halogen group Chemical group 0.000 claims description 30
- 150000003839 salts Chemical class 0.000 claims description 27
- 150000001875 compounds Chemical class 0.000 claims description 26
- 125000003107 substituted aryl group Chemical group 0.000 claims description 26
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 25
- 125000000664 diazo group Chemical group [N-]=[N+]=[*] 0.000 claims description 17
- 239000004215 Carbon black (E152) Substances 0.000 claims description 14
- 229930195733 hydrocarbon Natural products 0.000 claims description 14
- 229910052757 nitrogen Inorganic materials 0.000 claims description 14
- BDHFUVZGWQCTTF-UHFFFAOYSA-N sulfonic acid Chemical compound OS(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-N 0.000 claims description 14
- 229910052727 yttrium Inorganic materials 0.000 claims description 13
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 12
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 claims description 11
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 11
- 239000000460 chlorine Substances 0.000 claims description 11
- 229910004727 OSO3H Inorganic materials 0.000 claims description 10
- 125000003118 aryl group Chemical group 0.000 claims description 10
- 125000006575 electron-withdrawing group Chemical group 0.000 claims description 10
- NYGZLYXAPMMJTE-UHFFFAOYSA-M metanil yellow Chemical group [Na+].[O-]S(=O)(=O)C1=CC=CC(N=NC=2C=CC(NC=3C=CC=CC=3)=CC=2)=C1 NYGZLYXAPMMJTE-UHFFFAOYSA-M 0.000 claims description 10
- UHOVQNZJYSORNB-UHFFFAOYSA-N monobenzene Natural products C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 claims description 10
- QPILZZVXGUNELN-UHFFFAOYSA-M sodium;4-amino-5-hydroxynaphthalene-2,7-disulfonate;hydron Chemical compound [Na+].OS(=O)(=O)C1=CC(O)=C2C(N)=CC(S([O-])(=O)=O)=CC2=C1 QPILZZVXGUNELN-UHFFFAOYSA-M 0.000 claims description 10
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 9
- 125000000751 azo group Chemical group [*]N=N[*] 0.000 claims description 9
- 238000007641 inkjet printing Methods 0.000 claims description 8
- 150000002790 naphthalenes Chemical class 0.000 claims description 8
- 239000000758 substrate Substances 0.000 claims description 8
- 125000003277 amino group Chemical group 0.000 claims description 7
- VMGAPWLDMVPYIA-HIDZBRGKSA-N n'-amino-n-iminomethanimidamide Chemical compound N\N=C\N=N VMGAPWLDMVPYIA-HIDZBRGKSA-N 0.000 claims description 7
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 7
- PYKYMHQGRFAEBM-UHFFFAOYSA-N anthraquinone Natural products CCC(=O)c1c(O)c2C(=O)C3C(C=CC=C3O)C(=O)c2cc1CC(=O)OC PYKYMHQGRFAEBM-UHFFFAOYSA-N 0.000 claims description 6
- 150000004056 anthraquinones Chemical class 0.000 claims description 6
- 125000004432 carbon atom Chemical group C* 0.000 claims description 6
- 229910052736 halogen Chemical group 0.000 claims description 6
- 150000002367 halogens Chemical group 0.000 claims description 6
- 238000004519 manufacturing process Methods 0.000 claims description 6
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 6
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 6
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 5
- 238000006243 chemical reaction Methods 0.000 claims description 5
- 229910052801 chlorine Inorganic materials 0.000 claims description 5
- 125000005842 heteroatom Chemical group 0.000 claims description 5
- BOLDJAUMGUJJKM-LSDHHAIUSA-N renifolin D Natural products CC(=C)[C@@H]1Cc2c(O)c(O)ccc2[C@H]1CC(=O)c3ccc(O)cc3O BOLDJAUMGUJJKM-LSDHHAIUSA-N 0.000 claims description 5
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims description 4
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 claims description 4
- 125000004390 alkyl sulfonyl group Chemical group 0.000 claims description 4
- 239000011737 fluorine Substances 0.000 claims description 4
- 229910052731 fluorine Inorganic materials 0.000 claims description 4
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 claims description 4
- 238000007639 printing Methods 0.000 claims description 4
- 150000001408 amides Chemical class 0.000 claims description 3
- 150000001412 amines Chemical class 0.000 claims description 3
- 238000012505 colouration Methods 0.000 claims description 3
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 3
- 238000010016 exhaust dyeing Methods 0.000 claims description 3
- FDDDEECHVMSUSB-UHFFFAOYSA-N sulfanilamide Chemical compound NC1=CC=C(S(N)(=O)=O)C=C1 FDDDEECHVMSUSB-UHFFFAOYSA-N 0.000 claims description 3
- 229940124530 sulfonamide Drugs 0.000 claims description 3
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical group C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 claims description 2
- HMNPDEGBVWDHAR-UHFFFAOYSA-N 8-aminonaphthalen-1-ol Chemical group C1=CC(O)=C2C(N)=CC=CC2=C1 HMNPDEGBVWDHAR-UHFFFAOYSA-N 0.000 claims description 2
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 claims description 2
- 125000003282 alkyl amino group Chemical group 0.000 claims description 2
- 239000000987 azo dye Substances 0.000 claims description 2
- RBTKNAXYKSUFRK-UHFFFAOYSA-N heliogen blue Chemical compound [Cu].[N-]1C2=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=NC([N-]1)=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=N2 RBTKNAXYKSUFRK-UHFFFAOYSA-N 0.000 claims description 2
- 125000000623 heterocyclic group Chemical group 0.000 claims description 2
- 229910052751 metal Inorganic materials 0.000 claims description 2
- 239000002184 metal Substances 0.000 claims description 2
- 125000004193 piperazinyl group Chemical group 0.000 claims description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-O pyridinium Chemical class C1=CC=[NH+]C=C1 JUJWROOIHBZHMG-UHFFFAOYSA-O 0.000 claims description 2
- 125000005309 thioalkoxy group Chemical group 0.000 claims description 2
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 2
- 125000001183 hydrocarbyl group Chemical group 0.000 claims 7
- 150000002431 hydrogen Chemical group 0.000 claims 6
- -1 sulpho Chemical class 0.000 description 47
- 239000000243 solution Substances 0.000 description 45
- 0 *C.*C(C)N1CCN(C)CC1 Chemical compound *C.*C(C)N1CCN(C)CC1 0.000 description 36
- 229920000742 Cotton Polymers 0.000 description 36
- 229910006069 SO3H Inorganic materials 0.000 description 35
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 29
- 125000000020 sulfo group Chemical group O=S(=O)([*])O[H] 0.000 description 26
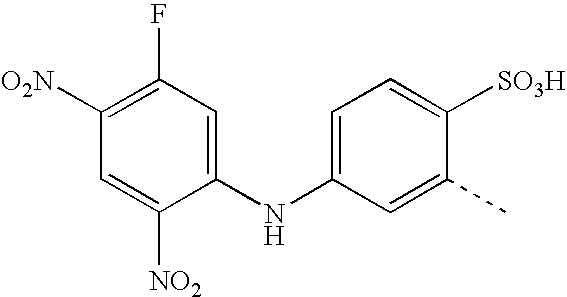
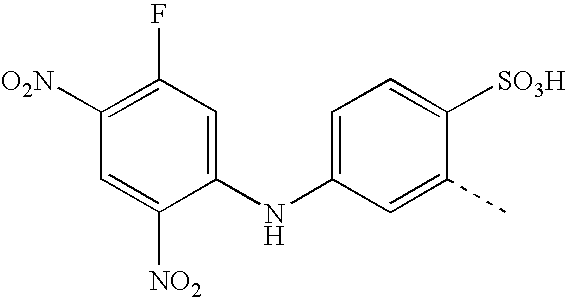
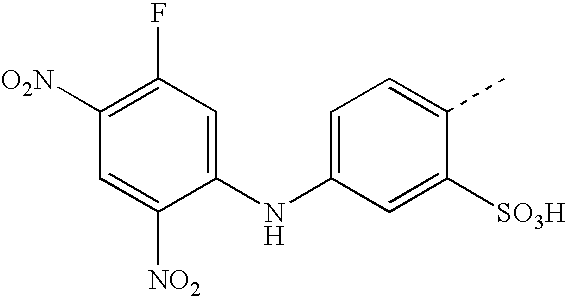
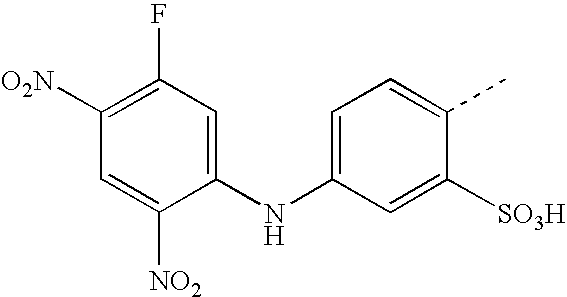
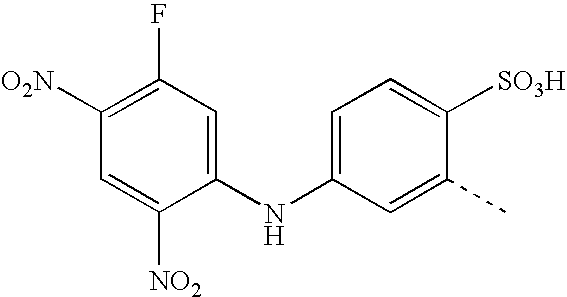
- BKDGBKHAJZPRLA-UHFFFAOYSA-N CC1=CC(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])=CC=C1S(=O)(=O)O BKDGBKHAJZPRLA-UHFFFAOYSA-N 0.000 description 23
- CMIMRMOSIVTUAA-UHFFFAOYSA-N CC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] Chemical compound CC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] CMIMRMOSIVTUAA-UHFFFAOYSA-N 0.000 description 20
- FFRBMBIXVSCUFS-UHFFFAOYSA-N 2,4-dinitro-1-naphthol Chemical compound C1=CC=C2C(O)=C([N+]([O-])=O)C=C([N+]([O-])=O)C2=C1 FFRBMBIXVSCUFS-UHFFFAOYSA-N 0.000 description 19
- 239000000976 ink Substances 0.000 description 16
- MPTVBFPSRSTYAU-UHFFFAOYSA-N C1=CC=CC=C1.CC.CC.C[Y] Chemical compound C1=CC=CC=C1.CC.CC.C[Y] MPTVBFPSRSTYAU-UHFFFAOYSA-N 0.000 description 15
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 14
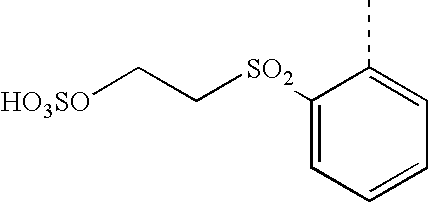
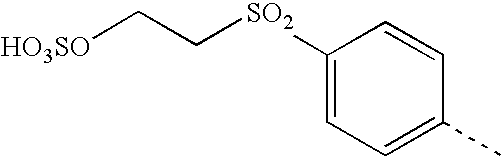
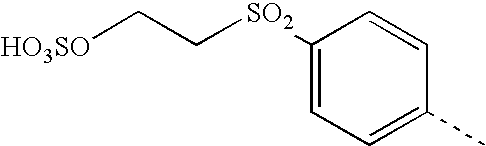
- FIDYJRHFNNOTKT-UHFFFAOYSA-N CC1=CC=C(S(=O)(=O)CCOSOOO)C=C1 Chemical compound CC1=CC=C(S(=O)(=O)CCOSOOO)C=C1 FIDYJRHFNNOTKT-UHFFFAOYSA-N 0.000 description 14
- 239000000203 mixture Substances 0.000 description 14
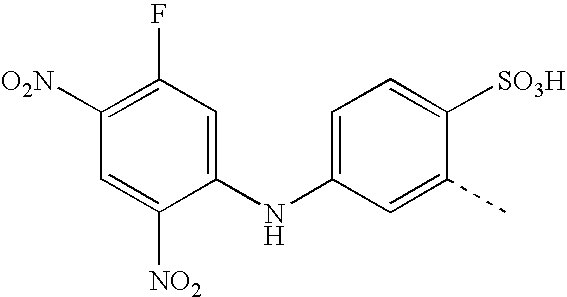
- MPXNIMFYTUIMRP-UHFFFAOYSA-N CC1=CC(NC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])=CC=C1S(=O)(=O)O MPXNIMFYTUIMRP-UHFFFAOYSA-N 0.000 description 13
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 13
- 239000000463 material Substances 0.000 description 13
- SKQBNRFJWOCVMH-UHFFFAOYSA-N CC1=CC=C(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])C=C1S(=O)(=O)O Chemical compound CC1=CC=C(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])C=C1S(=O)(=O)O SKQBNRFJWOCVMH-UHFFFAOYSA-N 0.000 description 12
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 12
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 12
- 239000004753 textile Substances 0.000 description 12
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 10
- GXKQTVXQUIWMGN-UHFFFAOYSA-N CC1=CC(S(=O)(=O)CCOSOOO)=CC=C1 Chemical compound CC1=CC(S(=O)(=O)CCOSOOO)=CC=C1 GXKQTVXQUIWMGN-UHFFFAOYSA-N 0.000 description 9
- UNSRNCGHWMJSAA-UHFFFAOYSA-N CC1=CC=C(NC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])C=C1S(=O)(=O)O Chemical compound CC1=CC=C(NC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])C=C1S(=O)(=O)O UNSRNCGHWMJSAA-UHFFFAOYSA-N 0.000 description 9
- 238000004043 dyeing Methods 0.000 description 9
- FSHNWWKZVLPEQS-UHFFFAOYSA-N CC1=CC(F)=C(C#N)C=C1[N+](=O)[O-] Chemical compound CC1=CC(F)=C(C#N)C=C1[N+](=O)[O-] FSHNWWKZVLPEQS-UHFFFAOYSA-N 0.000 description 8
- GIHNKADIUDISBR-UHFFFAOYSA-N CC1=CC(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])=C(C)C=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])=C(C)C=C1S(=O)(=O)O GIHNKADIUDISBR-UHFFFAOYSA-N 0.000 description 8
- IBNDQRXXEISNBE-UHFFFAOYSA-N CC1=CC=C(S(=O)(=O)CCOSOOO)C=C1S(=O)(=O)O Chemical compound CC1=CC=C(S(=O)(=O)CCOSOOO)C=C1S(=O)(=O)O IBNDQRXXEISNBE-UHFFFAOYSA-N 0.000 description 8
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 8
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 8
- 125000001424 substituent group Chemical group 0.000 description 8
- BOWBAYAOKGAFGS-UHFFFAOYSA-N CC1=CC=CC=C1S(=O)(=O)CCOSOOO Chemical compound CC1=CC=CC=C1S(=O)(=O)CCOSOOO BOWBAYAOKGAFGS-UHFFFAOYSA-N 0.000 description 7
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 7
- 239000004744 fabric Substances 0.000 description 7
- 150000002430 hydrocarbons Chemical group 0.000 description 7
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 7
- 239000000725 suspension Substances 0.000 description 7
- LDWJUYKHAMEDKV-UHFFFAOYSA-N C1=CC=CC=C1.CC.C[Y].C[Y] Chemical compound C1=CC=CC=C1.CC.C[Y].C[Y] LDWJUYKHAMEDKV-UHFFFAOYSA-N 0.000 description 6
- ZBCMUMCFIHLOJE-UHFFFAOYSA-N C1=NC=NC=N1.CC.CC.CC Chemical compound C1=NC=NC=N1.CC.CC.CC ZBCMUMCFIHLOJE-UHFFFAOYSA-N 0.000 description 6
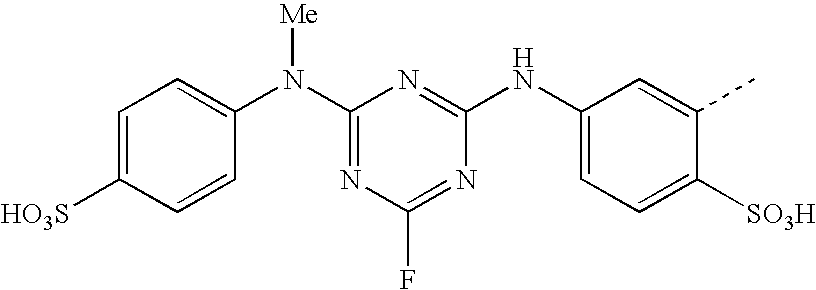
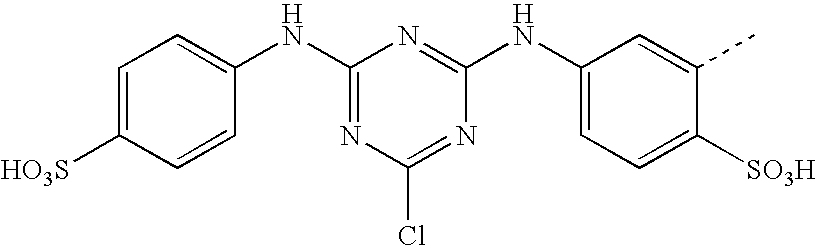
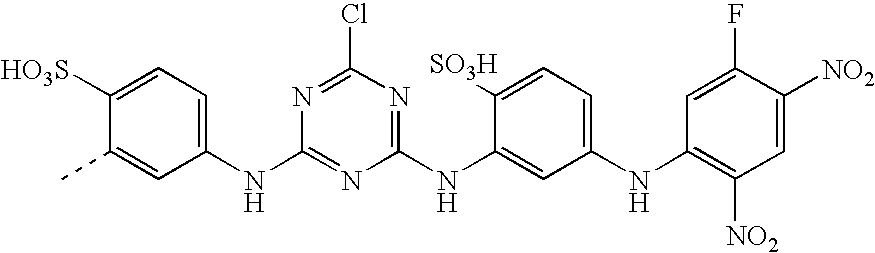
- CWVMSYMQQKGWLH-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=C(S(=O)(=O)O)C=CC(S(=O)(=O)O)=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=NC(NC3=C(S(=O)(=O)O)C=CC(S(=O)(=O)O)=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O CWVMSYMQQKGWLH-UHFFFAOYSA-N 0.000 description 6
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 239000002253 acid Substances 0.000 description 6
- 230000008878 coupling Effects 0.000 description 6
- 238000010168 coupling process Methods 0.000 description 6
- 238000005859 coupling reaction Methods 0.000 description 6
- 238000001914 filtration Methods 0.000 description 6
- 239000003906 humectant Substances 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 229910000029 sodium carbonate Inorganic materials 0.000 description 6
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 6
- VILFTWLXLYIEMV-UHFFFAOYSA-N 1,5-difluoro-2,4-dinitrobenzene Chemical compound [O-][N+](=O)C1=CC([N+]([O-])=O)=C(F)C=C1F VILFTWLXLYIEMV-UHFFFAOYSA-N 0.000 description 5
- IIBNEDZSCBOTMT-UHFFFAOYSA-N C1=CN=CN=C1.CC.CC.CC Chemical compound C1=CN=CN=C1.CC.CC.CC IIBNEDZSCBOTMT-UHFFFAOYSA-N 0.000 description 5
- KSNGEKYSDKGEOO-UHFFFAOYSA-N CC1=C(S(=O)(=O)O)C2=CC=CC(S(=O)(=O)O)=C2C=C1 Chemical compound CC1=C(S(=O)(=O)O)C2=CC=CC(S(=O)(=O)O)=C2C=C1 KSNGEKYSDKGEOO-UHFFFAOYSA-N 0.000 description 5
- RJAGFXJVYIDRNG-UHFFFAOYSA-N CC1=CC(C)=C(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])C=C1S(=O)(=O)O Chemical compound CC1=CC(C)=C(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])C=C1S(=O)(=O)O RJAGFXJVYIDRNG-UHFFFAOYSA-N 0.000 description 5
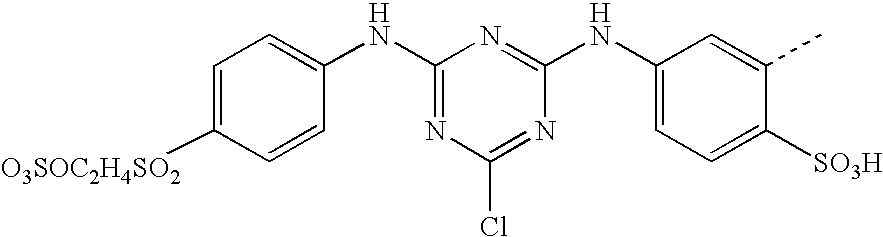
- SWFVSSUEELNOJR-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC=C(S(=O)(=O)CCOSOOO)C=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=NC(NC3=CC=C(S(=O)(=O)CCOSOOO)C=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O SWFVSSUEELNOJR-UHFFFAOYSA-N 0.000 description 5
- 239000012535 impurity Substances 0.000 description 5
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 description 5
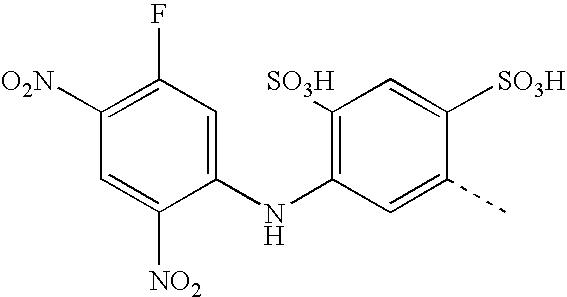
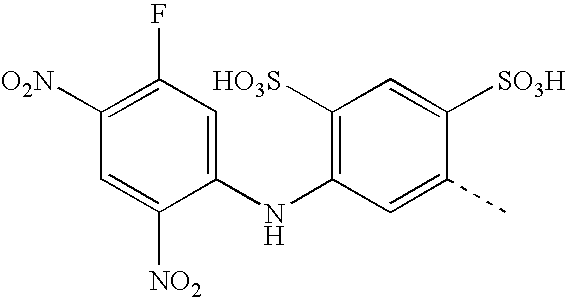
- MCQQYROYWRNAQV-UHFFFAOYSA-N CC1=CC(NC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])=C(S(=O)(=O)O)C=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])=C(S(=O)(=O)O)C=C1S(=O)(=O)O MCQQYROYWRNAQV-UHFFFAOYSA-N 0.000 description 4
- BZIXXXFXKVKVNH-UHFFFAOYSA-N CC1=CC(S(=O)(=O)CCOSOOO)=C(SOOO)C=C1S(=O)(=O)O Chemical compound CC1=CC(S(=O)(=O)CCOSOOO)=C(SOOO)C=C1S(=O)(=O)O BZIXXXFXKVKVNH-UHFFFAOYSA-N 0.000 description 4
- FSVBQHNAXLISLJ-UHFFFAOYSA-N CC1=CC=C(CCCOSOOO)C=C1S(=O)(=O)O Chemical compound CC1=CC=C(CCCOSOOO)C=C1S(=O)(=O)O FSVBQHNAXLISLJ-UHFFFAOYSA-N 0.000 description 4
- CLEDTPYCWWUOEX-UHFFFAOYSA-N CCCNC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] Chemical compound CCCNC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] CLEDTPYCWWUOEX-UHFFFAOYSA-N 0.000 description 4
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 4
- IOVCWXUNBOPUCH-UHFFFAOYSA-N Nitrous acid Chemical compound ON=O IOVCWXUNBOPUCH-UHFFFAOYSA-N 0.000 description 4
- 125000001309 chloro group Chemical group Cl* 0.000 description 4
- 150000004985 diamines Chemical class 0.000 description 4
- 125000001624 naphthyl group Chemical group 0.000 description 4
- 239000000123 paper Substances 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 159000000000 sodium salts Chemical class 0.000 description 4
- BFXZVZRCZMIZTH-UHFFFAOYSA-N CC1=CC(NC2=NC(Cl)=NC(NC3=CC=C(S(=O)(=O)O)C(C)=C3)=N2)=CC=C1 Chemical compound CC1=CC(NC2=NC(Cl)=NC(NC3=CC=C(S(=O)(=O)O)C(C)=C3)=N2)=CC=C1 BFXZVZRCZMIZTH-UHFFFAOYSA-N 0.000 description 3
- IRLYGRLEBKCYPY-UHFFFAOYSA-N CC1=CC(S(=O)(=O)O)=C(C)C=C1 Chemical compound CC1=CC(S(=O)(=O)O)=C(C)C=C1 IRLYGRLEBKCYPY-UHFFFAOYSA-N 0.000 description 3
- ZKNSSSGTQJLXMT-UHFFFAOYSA-N CC1=CC=C(NC2=NC(Cl)=NC(NC3=CC=C(S(=O)(=O)O)C(C)=C3)=N2)C=C1 Chemical compound CC1=CC=C(NC2=NC(Cl)=NC(NC3=CC=C(S(=O)(=O)O)C(C)=C3)=N2)C=C1 ZKNSSSGTQJLXMT-UHFFFAOYSA-N 0.000 description 3
- BFUGAHPTVMZLAC-UHFFFAOYSA-N CC1=NC(Cl)=NC(Cl)=N1 Chemical compound CC1=NC(Cl)=NC(Cl)=N1 BFUGAHPTVMZLAC-UHFFFAOYSA-N 0.000 description 3
- TZHRITUJLCGSMV-UHFFFAOYSA-N CC1=NC(Cl)=NC(NC2=CC(S(=O)(=O)O)=CC=C2)=N1 Chemical compound CC1=NC(Cl)=NC(NC2=CC(S(=O)(=O)O)=CC=C2)=N1 TZHRITUJLCGSMV-UHFFFAOYSA-N 0.000 description 3
- AVQZBECLWBSIMX-UHFFFAOYSA-N CCN(C1=CC=CC=C1)S(=O)(=O)C1=C(F)C=C(NC2=CC=C(S(=O)(=O)O)C(C)=C2)C([N+](=O)[O-])=C1 Chemical compound CCN(C1=CC=CC=C1)S(=O)(=O)C1=C(F)C=C(NC2=CC=C(S(=O)(=O)O)C(C)=C2)C([N+](=O)[O-])=C1 AVQZBECLWBSIMX-UHFFFAOYSA-N 0.000 description 3
- IXLATQSGFPMCGE-UHFFFAOYSA-N CNC1=C(S(=O)(=O)O)C=C(NC2=NC(NC3=C(S(=O)(=O)O)C=CC(NC4=CC(F)=C([N+](=O)[O-])C=C4[N+](=O)[O-])=C3)=NC(Cl)=N2)C=C1 Chemical compound CNC1=C(S(=O)(=O)O)C=C(NC2=NC(NC3=C(S(=O)(=O)O)C=CC(NC4=CC(F)=C([N+](=O)[O-])C=C4[N+](=O)[O-])=C3)=NC(Cl)=N2)C=C1 IXLATQSGFPMCGE-UHFFFAOYSA-N 0.000 description 3
- FESVWAFSXUZEHC-UHFFFAOYSA-N CNc(cc(c([N+]([O-])=O)c1)F)c1[N+]([O-])=O Chemical compound CNc(cc(c([N+]([O-])=O)c1)F)c1[N+]([O-])=O FESVWAFSXUZEHC-UHFFFAOYSA-N 0.000 description 3
- 239000004115 Sodium Silicate Substances 0.000 description 3
- 206010042618 Surgical procedure repeated Diseases 0.000 description 3
- AHWXCYJGJOLNFA-UHFFFAOYSA-N [1,4]benzoxazino[2,3-b]phenoxazine Chemical compound O1C2=CC=CC=C2N=C2C1=CC1=NC3=CC=CC=C3OC1=C2 AHWXCYJGJOLNFA-UHFFFAOYSA-N 0.000 description 3
- GPYBEPUOAJFJFU-UHFFFAOYSA-N [C-]#[N+]C1=C(F)C=C(NC2=CC=C(S(=O)(=O)O)C(C)=C2)C([N+](=O)[O-])=C1 Chemical compound [C-]#[N+]C1=C(F)C=C(NC2=CC=C(S(=O)(=O)O)C(C)=C2)C([N+](=O)[O-])=C1 GPYBEPUOAJFJFU-UHFFFAOYSA-N 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 229910052744 lithium Inorganic materials 0.000 description 3
- 229960004063 propylene glycol Drugs 0.000 description 3
- 235000013772 propylene glycol Nutrition 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 235000010288 sodium nitrite Nutrition 0.000 description 3
- NTHWMYGWWRZVTN-UHFFFAOYSA-N sodium silicate Chemical compound [Na+].[Na+].[O-][Si]([O-])=O NTHWMYGWWRZVTN-UHFFFAOYSA-N 0.000 description 3
- 229910052911 sodium silicate Inorganic materials 0.000 description 3
- IIACRCGMVDHOTQ-UHFFFAOYSA-N sulfamic acid Chemical compound NS(O)(=O)=O IIACRCGMVDHOTQ-UHFFFAOYSA-N 0.000 description 3
- 238000011282 treatment Methods 0.000 description 3
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- QKMRGQVJHDZIHK-UHFFFAOYSA-N C.C1=NC=NC=N1.C1=NC=NC=N1.CC.CC.CC.CC.C[Y].C[Y] Chemical compound C.C1=NC=NC=N1.C1=NC=NC=N1.CC.CC.CC.CC.C[Y].C[Y] QKMRGQVJHDZIHK-UHFFFAOYSA-N 0.000 description 2
- JQCAFFFZPXGCQK-UHFFFAOYSA-N C=CS(=O)(=O)C1=C(SOOO)C=C(S(=O)(=O)O)C(C)=C1 Chemical compound C=CS(=O)(=O)C1=C(SOOO)C=C(S(=O)(=O)O)C(C)=C1 JQCAFFFZPXGCQK-UHFFFAOYSA-N 0.000 description 2
- IXPHUOQMDNFWCC-UHFFFAOYSA-N CC(=O)NCCNC1=NC(Cl)=NC(NC2=CC=C(S(=O)(=O)O)C(C)=C2)=N1 Chemical compound CC(=O)NCCNC1=NC(Cl)=NC(NC2=CC=C(S(=O)(=O)O)C(C)=C2)=N1 IXPHUOQMDNFWCC-UHFFFAOYSA-N 0.000 description 2
- LGLNSAXKLYESSP-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC(CCCOSOOO)=CC=C3)=NC(Cl)=N2)=C(S(=O)(=O)O)C=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=NC(NC3=CC(CCCOSOOO)=CC=C3)=NC(Cl)=N2)=C(S(=O)(=O)O)C=C1S(=O)(=O)O LGLNSAXKLYESSP-UHFFFAOYSA-N 0.000 description 2
- VQGBIYOUNFUVHR-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC=C(SOOO)C=C3)=NC(Cl)=N2)=C(S(=O)(=O)O)C=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=NC(NC3=CC=C(SOOO)C=C3)=NC(Cl)=N2)=C(S(=O)(=O)O)C=C1S(=O)(=O)O VQGBIYOUNFUVHR-UHFFFAOYSA-N 0.000 description 2
- QJZAWTCNRNIMQX-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC=C(SOOO)C=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=NC(NC3=CC=C(SOOO)C=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O QJZAWTCNRNIMQX-UHFFFAOYSA-N 0.000 description 2
- AXKFSCWBZOICCL-ANAXZMQJSA-N CC1=CC(S(=O)(=O)O)=C(N=NC2=C(S(O)(O)O)C=C3C=C(S(=O)(=O)O)C(/N=N\C4=CC(S(=O)(=O)O)=C(NC5=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C5)C=C4S(=O)(=O)O)=C(C)C3=C2C)C=C1 Chemical compound CC1=CC(S(=O)(=O)O)=C(N=NC2=C(S(O)(O)O)C=C3C=C(S(=O)(=O)O)C(/N=N\C4=CC(S(=O)(=O)O)=C(NC5=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C5)C=C4S(=O)(=O)O)=C(C)C3=C2C)C=C1 AXKFSCWBZOICCL-ANAXZMQJSA-N 0.000 description 2
- MLZMFXOTPJARMB-UHFFFAOYSA-N CC1=CC=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1NC1=NC(Cl)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 Chemical compound CC1=CC=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1NC1=NC(Cl)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 MLZMFXOTPJARMB-UHFFFAOYSA-N 0.000 description 2
- HKZPAOPAWMAWMN-BYYHNAKLSA-N CC1=CC=C2C(C)=CC=C(/N=N/C3=CC=C(NC4=CC(F)=C(NOO)C=C4[N+](=O)[O-])C=C3S(=O)(=O)O)C2=C1 Chemical compound CC1=CC=C2C(C)=CC=C(/N=N/C3=CC=C(NC4=CC(F)=C(NOO)C=C4[N+](=O)[O-])C=C3S(=O)(=O)O)C2=C1 HKZPAOPAWMAWMN-BYYHNAKLSA-N 0.000 description 2
- DNEMDBRILBVUAP-UHFFFAOYSA-N CC1=NC(Cl)=NC(NC2=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=CC=C2S(=O)(=O)O)=N1 Chemical compound CC1=NC(Cl)=NC(NC2=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=CC=C2S(=O)(=O)O)=N1 DNEMDBRILBVUAP-UHFFFAOYSA-N 0.000 description 2
- FHPSEQAHSCPZRG-UHFFFAOYSA-N CC1=NC(Cl)=NC(NC2=CC(S(=O)(=O)CCOS(=O)(=O)O)=CC=C2)=N1 Chemical compound CC1=NC(Cl)=NC(NC2=CC(S(=O)(=O)CCOS(=O)(=O)O)=CC=C2)=N1 FHPSEQAHSCPZRG-UHFFFAOYSA-N 0.000 description 2
- WKSWRVHHGGGNCW-UHFFFAOYSA-N CC1=NC(Cl)=NC(NC2=CC(SO(O)CCOS(=O)(=O)O)=CC=C2)=N1 Chemical compound CC1=NC(Cl)=NC(NC2=CC(SO(O)CCOS(=O)(=O)O)=CC=C2)=N1 WKSWRVHHGGGNCW-UHFFFAOYSA-N 0.000 description 2
- CKDDTPPDCGUFOX-UHFFFAOYSA-N CC1=NC(F)=NC(NC2=CC(S(=O)(=O)O)=CC=C2)=N1 Chemical compound CC1=NC(F)=NC(NC2=CC(S(=O)(=O)O)=CC=C2)=N1 CKDDTPPDCGUFOX-UHFFFAOYSA-N 0.000 description 2
- CHBAWFGIXDBEBT-UHFFFAOYSA-N CCCC(C)CCC Chemical compound CCCC(C)CCC CHBAWFGIXDBEBT-UHFFFAOYSA-N 0.000 description 2
- HXSJUHLLFBPGJQ-UHFFFAOYSA-N CCCCNC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] Chemical compound CCCCNC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] HXSJUHLLFBPGJQ-UHFFFAOYSA-N 0.000 description 2
- UMGNJVBIZCNRNF-UHFFFAOYSA-N CCCNC1=NC(NC2=CC(S(=O)(=O)O)=C(NC3=CC(Cl)=C([N+](=O)[O-])C=C3[N+](=O)[O-])C=C2S(=O)(=O)O)=NC(Cl)=N1 Chemical compound CCCNC1=NC(NC2=CC(S(=O)(=O)O)=C(NC3=CC(Cl)=C([N+](=O)[O-])C=C3[N+](=O)[O-])C=C2S(=O)(=O)O)=NC(Cl)=N1 UMGNJVBIZCNRNF-UHFFFAOYSA-N 0.000 description 2
- ZBBLANZKYWTTHK-UHFFFAOYSA-N CCCNC1=NC(NC2=CC(S(=O)(=O)O)=C(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])C=C2S(=O)(=O)O)=NC(F)=N1 Chemical compound CCCNC1=NC(NC2=CC(S(=O)(=O)O)=C(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])C=C2S(=O)(=O)O)=NC(F)=N1 ZBBLANZKYWTTHK-UHFFFAOYSA-N 0.000 description 2
- CVBQDJLIPNQXCZ-UHFFFAOYSA-N CCCNC1=NC(NC2=CC=C(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])C=C2S(=O)(=O)O)=NC(Cl)=N1 Chemical compound CCCNC1=NC(NC2=CC=C(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])C=C2S(=O)(=O)O)=NC(Cl)=N1 CVBQDJLIPNQXCZ-UHFFFAOYSA-N 0.000 description 2
- JWPLLRNJPFDGEL-UHFFFAOYSA-N CCCNC1=NC(NCCNC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])=NC(Cl)=N1 Chemical compound CCCNC1=NC(NCCNC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])=NC(Cl)=N1 JWPLLRNJPFDGEL-UHFFFAOYSA-N 0.000 description 2
- GKQAMOGKNSSIDJ-UHFFFAOYSA-N CNBCC(C)C Chemical compound CNBCC(C)C GKQAMOGKNSSIDJ-UHFFFAOYSA-N 0.000 description 2
- LZEBHQQBLHMJOG-UHFFFAOYSA-N CNCCN1CCN(C)CC1 Chemical compound CNCCN1CCN(C)CC1 LZEBHQQBLHMJOG-UHFFFAOYSA-N 0.000 description 2
- UMXJXWRHGNZYNI-UHFFFAOYSA-N CSO(O)C1=C(F)C=C(NC2=CC=C(S(=O)(=O)O)C(C)=C2)C([N+](=O)[O-])=C1 Chemical compound CSO(O)C1=C(F)C=C(NC2=CC=C(S(=O)(=O)O)C(C)=C2)C([N+](=O)[O-])=C1 UMXJXWRHGNZYNI-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 2
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 2
- 239000004952 Polyamide Substances 0.000 description 2
- LQBOFIKUDKPJHF-UHFFFAOYSA-N [C-]#[N+]C1=C(F)C=C(NC2=CC=C(C)C(S(=O)(=O)O)=C2)C([N+](=O)[O-])=C1 Chemical compound [C-]#[N+]C1=C(F)C=C(NC2=CC=C(C)C(S(=O)(=O)O)=C2)C([N+](=O)[O-])=C1 LQBOFIKUDKPJHF-UHFFFAOYSA-N 0.000 description 2
- 238000013019 agitation Methods 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 125000000043 benzamido group Chemical group [H]N([*])C(=O)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 description 2
- 239000006227 byproduct Substances 0.000 description 2
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 2
- 150000003857 carboxamides Chemical class 0.000 description 2
- 239000011928 denatured alcohol Substances 0.000 description 2
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 125000001072 heteroaryl group Chemical group 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 2
- 238000005342 ion exchange Methods 0.000 description 2
- TWBYWOBDOCUKOW-UHFFFAOYSA-N isonicotinic acid Chemical compound OC(=O)C1=CC=NC=C1 TWBYWOBDOCUKOW-UHFFFAOYSA-N 0.000 description 2
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 239000012466 permeate Substances 0.000 description 2
- 150000004986 phenylenediamines Chemical class 0.000 description 2
- 229920002647 polyamide Polymers 0.000 description 2
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 2
- YPFDHNVEDLHUCE-UHFFFAOYSA-N propane-1,3-diol Chemical compound OCCCO YPFDHNVEDLHUCE-UHFFFAOYSA-N 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 239000008149 soap solution Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 210000002268 wool Anatomy 0.000 description 2
- ZNXSFVXZQBETRJ-UHFFFAOYSA-N (3-aminophenyl)urea Chemical compound NC(=O)NC1=CC=CC(N)=C1 ZNXSFVXZQBETRJ-UHFFFAOYSA-N 0.000 description 1
- ZPXDNSYFDIHPOJ-UHFFFAOYSA-N 1,5-dichloro-2,4-dinitrobenzene Chemical compound [O-][N+](=O)C1=CC([N+]([O-])=O)=C(Cl)C=C1Cl ZPXDNSYFDIHPOJ-UHFFFAOYSA-N 0.000 description 1
- LOTKRQAVGJMPNV-UHFFFAOYSA-N 1-fluoro-2,4-dinitrobenzene Chemical group [O-][N+](=O)C1=CC=C(F)C([N+]([O-])=O)=C1 LOTKRQAVGJMPNV-UHFFFAOYSA-N 0.000 description 1
- RJXOVESYJFXCGI-UHFFFAOYSA-N 2,4-difluoro-1-nitrobenzene Chemical compound [O-][N+](=O)C1=CC=C(F)C=C1F RJXOVESYJFXCGI-UHFFFAOYSA-N 0.000 description 1
- MREZSSGRNNMUKZ-UHFFFAOYSA-N 2,4-difluoro-5-nitrobenzonitrile Chemical compound [O-][N+](=O)C1=CC(C#N)=C(F)C=C1F MREZSSGRNNMUKZ-UHFFFAOYSA-N 0.000 description 1
- KTVOLKQQNNIGGM-UHFFFAOYSA-N 2-[[4-[4-[(2-hydroxy-5-nonylphenyl)diazenyl]-3-methylphenyl]-2-methylphenyl]diazenyl]-4-nonylphenol Chemical compound CCCCCCCCCC1=CC(N=NC2=C(C)C=C(C=C2)C2=CC(C)=C(C=C2)N=NC2=CC(CCCCCCCCC)=CC=C2O)=C(O)C=C1 KTVOLKQQNNIGGM-UHFFFAOYSA-N 0.000 description 1
- ZAJAQTYSTDTMCU-UHFFFAOYSA-N 3-aminobenzenesulfonic acid Chemical compound NC1=CC=CC(S(O)(=O)=O)=C1 ZAJAQTYSTDTMCU-UHFFFAOYSA-N 0.000 description 1
- FOINSAWEWXUXPQ-UHFFFAOYSA-N 4-acetamido-2-aminobenzenesulfonic acid Chemical compound CC(=O)NC1=CC=C(S(O)(=O)=O)C(N)=C1 FOINSAWEWXUXPQ-UHFFFAOYSA-N 0.000 description 1
- VAVOYRCCWLRTMS-UHFFFAOYSA-N 4-piperazin-1-ylaniline Chemical compound C1=CC(N)=CC=C1N1CCNCC1 VAVOYRCCWLRTMS-UHFFFAOYSA-N 0.000 description 1
- GFPQSWFFPRQEHH-UHFFFAOYSA-N 7-aminonaphthalene-1,3,6-trisulfonic acid Chemical compound C1=C(S(O)(=O)=O)C=C2C=C(S(O)(=O)=O)C(N)=CC2=C1S(O)(=O)=O GFPQSWFFPRQEHH-UHFFFAOYSA-N 0.000 description 1
- RZVHIXYEVGDQDX-UHFFFAOYSA-N 9,10-anthraquinone Chemical group C1=CC=C2C(=O)C3=CC=CC=C3C(=O)C2=C1 RZVHIXYEVGDQDX-UHFFFAOYSA-N 0.000 description 1
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical class OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 241001674044 Blattodea Species 0.000 description 1
- 239000004358 Butane-1, 3-diol Substances 0.000 description 1
- NYHTYWRSMPZXNR-ZWGKIWOBSA-N C.C.C.C[2H]C(C)[Ar]C Chemical compound C.C.C.C[2H]C(C)[Ar]C NYHTYWRSMPZXNR-ZWGKIWOBSA-N 0.000 description 1
- RSRRNKYQOUNDGQ-UHFFFAOYSA-N C.C.CC.CC.CC(C)N=NC1=C(S(O)(O)O)C=C2C=CC=CC2=C1O Chemical compound C.C.CC.CC.CC(C)N=NC1=C(S(O)(O)O)C=C2C=CC=CC2=C1O RSRRNKYQOUNDGQ-UHFFFAOYSA-N 0.000 description 1
- ASZQZSGKEBTWRY-UHFFFAOYSA-N C.C.CC.CC.CC(C)N=NC1=CC=C2C=CC=CC2=C1O Chemical compound C.C.CC.CC.CC(C)N=NC1=CC=C2C=CC=CC2=C1O ASZQZSGKEBTWRY-UHFFFAOYSA-N 0.000 description 1
- LZYUAZVFRHIASD-SNHBFGBXSA-N C.C.C[2H]C(C)[Ar]C Chemical compound C.C.C[2H]C(C)[Ar]C LZYUAZVFRHIASD-SNHBFGBXSA-N 0.000 description 1
- FMLSDGARSGZLFP-RRHCXGJISA-N C.C/N=C/B(N)NC.CC Chemical compound C.C/N=C/B(N)NC.CC FMLSDGARSGZLFP-RRHCXGJISA-N 0.000 description 1
- BCUCQTKCLSIHHB-NSKUCRDLSA-N C.CC1=C(/N=N/C2=C(S(=O)(=O)O)C=CC=C2)C(=O)N(C)C(O)=C1[Y] Chemical compound C.CC1=C(/N=N/C2=C(S(=O)(=O)O)C=CC=C2)C(=O)N(C)C(O)=C1[Y] BCUCQTKCLSIHHB-NSKUCRDLSA-N 0.000 description 1
- CXFGXJWDSSIMMQ-UHFFFAOYSA-N C.CC1=CC(NC(N)=O)=C(N=NC2=CC3=C(S(=O)(=O)O)C=C(S(O)(O)O)C=C3C=C2S(=O)(=O)O)C=C1 Chemical compound C.CC1=CC(NC(N)=O)=C(N=NC2=CC3=C(S(=O)(=O)O)C=C(S(O)(O)O)C=C3C=C2S(=O)(=O)O)C=C1 CXFGXJWDSSIMMQ-UHFFFAOYSA-N 0.000 description 1
- XTUOOKVDAKXOHN-UHFFFAOYSA-N C.N#CC1=CC([N+](=O)[O-])=C(NC2=CC(N=NC3=C(O)C4=C(N)C(N=NC5=CC(NC6=CC(F)=C(C#N)C=C6[N+](=O)[O-])=CC=C5S(=O)(=O)O)=C(S(O)(O)O)C=C4C=C3S(=O)(=O)O)=C(S(=O)(=O)O)C=C2)C=C1F Chemical compound C.N#CC1=CC([N+](=O)[O-])=C(NC2=CC(N=NC3=C(O)C4=C(N)C(N=NC5=CC(NC6=CC(F)=C(C#N)C=C6[N+](=O)[O-])=CC=C5S(=O)(=O)O)=C(S(O)(O)O)C=C4C=C3S(=O)(=O)O)=C(S(=O)(=O)O)C=C2)C=C1F XTUOOKVDAKXOHN-UHFFFAOYSA-N 0.000 description 1
- RYPCIZRIUMIPJS-UHFFFAOYSA-N C.NC1=C(N=NC2=C(S(=O)(=O)O)C=CC(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=C2)C(S(=O)(=O)O)=CC2=CC(S(O)(O)O)=C(N=NC3=CC=C(SOOCCS(=O)(O)(O)O)C=C3)C(O)=C21 Chemical compound C.NC1=C(N=NC2=C(S(=O)(=O)O)C=CC(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=C2)C(S(=O)(=O)O)=CC2=CC(S(O)(O)O)=C(N=NC3=CC=C(SOOCCS(=O)(O)(O)O)C=C3)C(O)=C21 RYPCIZRIUMIPJS-UHFFFAOYSA-N 0.000 description 1
- PAIWIPJGQBVXBO-UHFFFAOYSA-N C.NC1=C2C(=CC(S(O)(O)O)=C1N=NC1=CC(NC3=CC(Cl)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=CC=C1S(=O)(=O)O)C=C(S(=O)(=O)O)C(N=NC1=C(S(=O)(=O)O)C=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(Cl)=C3)=C1)=C2O Chemical compound C.NC1=C2C(=CC(S(O)(O)O)=C1N=NC1=CC(NC3=CC(Cl)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=CC=C1S(=O)(=O)O)C=C(S(=O)(=O)O)C(N=NC1=C(S(=O)(=O)O)C=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(Cl)=C3)=C1)=C2O PAIWIPJGQBVXBO-UHFFFAOYSA-N 0.000 description 1
- MZXFMMGIYIVGIM-UHFFFAOYSA-N C.NC1=C2C(=CC(S(O)(O)O)=C1N=NC1=CC(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=CC=C1S(=O)(=O)O)C=C(S(=O)(=O)O)C(N=NC1=C(S(=O)(=O)O)C=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=C1)=C2O Chemical compound C.NC1=C2C(=CC(S(O)(O)O)=C1N=NC1=CC(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=CC=C1S(=O)(=O)O)C=C(S(=O)(=O)O)C(N=NC1=C(S(=O)(=O)O)C=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=C1)=C2O MZXFMMGIYIVGIM-UHFFFAOYSA-N 0.000 description 1
- BEJKICHTVFJRFD-UHFFFAOYSA-N C.NC1=C2C(=CC(S(O)(O)O)=C1N=NC1=CC(NC3=CC(F)=CC=C3[N+](=O)[O-])=CC=C1S(=O)(=O)O)C=C(S(=O)(=O)O)C(N=NC1=C(S(=O)(=O)O)C=CC(NC3=C([N+](=O)[O-])C=CC(F)=C3)=C1)=C2O Chemical compound C.NC1=C2C(=CC(S(O)(O)O)=C1N=NC1=CC(NC3=CC(F)=CC=C3[N+](=O)[O-])=CC=C1S(=O)(=O)O)C=C(S(=O)(=O)O)C(N=NC1=C(S(=O)(=O)O)C=CC(NC3=C([N+](=O)[O-])C=CC(F)=C3)=C1)=C2O BEJKICHTVFJRFD-UHFFFAOYSA-N 0.000 description 1
- UDNQMMQNXQVWSC-UHFFFAOYSA-N C.NC1=C2C(=CC(S(O)(O)O)=C1N=NC1=CC=C(SOOCCS(=O)(O)(O)O)C=C1)C=C(S(=O)(=O)O)C(N=NC1=C(S(=O)(=O)O)C=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=C1)=C2O Chemical compound C.NC1=C2C(=CC(S(O)(O)O)=C1N=NC1=CC=C(SOOCCS(=O)(O)(O)O)C=C1)C=C(S(=O)(=O)O)C(N=NC1=C(S(=O)(=O)O)C=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=C1)=C2O UDNQMMQNXQVWSC-UHFFFAOYSA-N 0.000 description 1
- GLIYSWVETSGAFO-UHFFFAOYSA-N C1=CN=CN=C1.CC.CC Chemical compound C1=CN=CN=C1.CC.CC GLIYSWVETSGAFO-UHFFFAOYSA-N 0.000 description 1
- WOKFZFYPVVMXMC-NVQSTNCTSA-N C=CC(=O)NC1=CC=C(/N=N/C2=C(O)N(C3=CC(S(=O)(=O)O)=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C3C)N=C2C(=O)O)C(S(=O)(=O)O)=C1 Chemical compound C=CC(=O)NC1=CC=C(/N=N/C2=C(O)N(C3=CC(S(=O)(=O)O)=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C3C)N=C2C(=O)O)C(S(=O)(=O)O)=C1 WOKFZFYPVVMXMC-NVQSTNCTSA-N 0.000 description 1
- QTTZZUJXQPUOTF-UHFFFAOYSA-N C=CCS(=O)(=O)C1=CC=C(C)C=C1 Chemical compound C=CCS(=O)(=O)C1=CC=C(C)C=C1 QTTZZUJXQPUOTF-UHFFFAOYSA-N 0.000 description 1
- VZQFRPMWVXCURA-UHFFFAOYSA-N C=CS(=O)(=O)C1=CC=C(C)C=C1 Chemical compound C=CS(=O)(=O)C1=CC=C(C)C=C1 VZQFRPMWVXCURA-UHFFFAOYSA-N 0.000 description 1
- FPIKNCRVEWBGTD-VHEBQXMUSA-N CC(=O)C(/N=N/C1=CC=CC=C1)C(=O)NC1=CC=CC=C1 Chemical compound CC(=O)C(/N=N/C1=CC=CC=C1)C(=O)NC1=CC=CC=C1 FPIKNCRVEWBGTD-VHEBQXMUSA-N 0.000 description 1
- GCHHLSJQPNNOMT-UHFFFAOYSA-K CC.CC.CC.CC.CC.O=C1O[Cu]23OC4=C(C=CC=C4)N2N=C(C2=CC=CC=C2)N=N3C2=CC=CC=C12 Chemical compound CC.CC.CC.CC.CC.O=C1O[Cu]23OC4=C(C=CC=C4)N2N=C(C2=CC=CC=C2)N=N3C2=CC=CC=C12 GCHHLSJQPNNOMT-UHFFFAOYSA-K 0.000 description 1
- RIFKRKAJSINCNQ-OBNSMGDVSA-N CC.CC.CC.CC.CC1=C(/N=N\C2=CC=CC=C2)C=CC2=CC=C(N=NC3=CC=CC=C3)C(C)=C21.CNC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] Chemical compound CC.CC.CC.CC.CC1=C(/N=N\C2=CC=CC=C2)C=CC2=CC=C(N=NC3=CC=CC=C3)C(C)=C21.CNC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] RIFKRKAJSINCNQ-OBNSMGDVSA-N 0.000 description 1
- RIFKRKAJSINCNQ-UHFFFAOYSA-N CC.CC.CC.CC.CC1=C(N=NC2=CC=CC=C2)C=CC2=CC=C(N=NC3=CC=CC=C3)C(C)=C21.CNC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] Chemical compound CC.CC.CC.CC.CC1=C(N=NC2=CC=CC=C2)C=CC2=CC=C(N=NC3=CC=CC=C3)C(C)=C21.CNC1=CC(F)=C([N+](=O)[O-])C=C1[N+](=O)[O-] RIFKRKAJSINCNQ-UHFFFAOYSA-N 0.000 description 1
- QYMQYNZZLFFQJU-WPDLWGESSA-N CC1=C(/N=N/C2=C(S(=O)(=O)O)C=CC=C2)C(=O)N(C)C(O)=C1[Y] Chemical compound CC1=C(/N=N/C2=C(S(=O)(=O)O)C=CC=C2)C(=O)N(C)C(O)=C1[Y] QYMQYNZZLFFQJU-WPDLWGESSA-N 0.000 description 1
- BSXBEOLOTRYICT-UHFFFAOYSA-N CC1=C(C)C(SOOO)=C(C)C(NC2=NC(Cl)=NC(NC3=CC=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(Cl)=C4)C=C3S(=O)(=O)O)=N2)=C1C Chemical compound CC1=C(C)C(SOOO)=C(C)C(NC2=NC(Cl)=NC(NC3=CC=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(Cl)=C4)C=C3S(=O)(=O)O)=N2)=C1C BSXBEOLOTRYICT-UHFFFAOYSA-N 0.000 description 1
- LIHDFUKYZLUQQA-UHFFFAOYSA-N CC1=C(C)C(SOOO)=C(C)C(NC2=NC(Cl)=NC(NC3=CC=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)C=C3S(=O)(=O)O)=N2)=C1C Chemical compound CC1=C(C)C(SOOO)=C(C)C(NC2=NC(Cl)=NC(NC3=CC=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)C=C3S(=O)(=O)O)=N2)=C1C LIHDFUKYZLUQQA-UHFFFAOYSA-N 0.000 description 1
- PDEJXIOMKPKEAK-UHFFFAOYSA-N CC1=C(C)C=C(SOOO)C=C1 Chemical compound CC1=C(C)C=C(SOOO)C=C1 PDEJXIOMKPKEAK-UHFFFAOYSA-N 0.000 description 1
- WODGMMJHSAKKNF-UHFFFAOYSA-N CC1=C(S(=O)(=O)O)C2=CC=CC=C2C=C1 Chemical compound CC1=C(S(=O)(=O)O)C2=CC=CC=C2C=C1 WODGMMJHSAKKNF-UHFFFAOYSA-N 0.000 description 1
- LBLYYCQCTBFVLH-UHFFFAOYSA-N CC1=C(S(=O)(=O)O)C=CC=C1 Chemical compound CC1=C(S(=O)(=O)O)C=CC=C1 LBLYYCQCTBFVLH-UHFFFAOYSA-N 0.000 description 1
- HOQNPPNARFOZRQ-UHFFFAOYSA-N CC1=CC(C)=C(SOOO)C=C1 Chemical compound CC1=CC(C)=C(SOOO)C=C1 HOQNPPNARFOZRQ-UHFFFAOYSA-N 0.000 description 1
- UVIDNIYQDXRHKL-UHFFFAOYSA-N CC1=CC(NC(N)=O)=CC=C1.CN=NC1=CC2=C(S(=O)(=O)O)C=C(S(O)(O)O)C=C2C=C1S(=O)(=O)O Chemical compound CC1=CC(NC(N)=O)=CC=C1.CN=NC1=CC2=C(S(=O)(=O)O)C=C(S(O)(O)O)C=C2C=C1S(=O)(=O)O UVIDNIYQDXRHKL-UHFFFAOYSA-N 0.000 description 1
- HZEJIEXSIOGYDK-UHFFFAOYSA-N CC1=CC(NC2=C([N+](=O)[O-])C([N+](=O)[O-])=CC(F)=C2)=CC=C1NC1=NC(Cl)=NC(NC2=CC=C(C)C(SOOO)=C2)=N1 Chemical compound CC1=CC(NC2=C([N+](=O)[O-])C([N+](=O)[O-])=CC(F)=C2)=CC=C1NC1=NC(Cl)=NC(NC2=CC=C(C)C(SOOO)=C2)=N1 HZEJIEXSIOGYDK-UHFFFAOYSA-N 0.000 description 1
- SCLZFEAFLITPSS-UHFFFAOYSA-N CC1=CC(NC2=C([N+](=O)[O-])C([N+](=O)[O-])=CC(F)=C2)=CC=C1NC1=NC(Cl)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 Chemical compound CC1=CC(NC2=C([N+](=O)[O-])C([N+](=O)[O-])=CC(F)=C2)=CC=C1NC1=NC(Cl)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 SCLZFEAFLITPSS-UHFFFAOYSA-N 0.000 description 1
- QMQBJVJFRWSNTQ-UHFFFAOYSA-N CC1=CC(NC2=CC(Cl)=C(NOO)C=C2[N+](=O)[O-])=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=CC(Cl)=C(NOO)C=C2[N+](=O)[O-])=CC=C1S(=O)(=O)O QMQBJVJFRWSNTQ-UHFFFAOYSA-N 0.000 description 1
- JQCXACWKWRXJBJ-UHFFFAOYSA-N CC1=CC(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])=C(SOOO)C=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=CC(F)=C(NOO)C=C2[N+](=O)[O-])=C(SOOO)C=C1S(=O)(=O)O JQCXACWKWRXJBJ-UHFFFAOYSA-N 0.000 description 1
- PZGIUJSBVQXMEZ-UHFFFAOYSA-N CC1=CC(NC2=NC(N(C)C3=CC=C(SOOO)C=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=NC(N(C)C3=CC=C(SOOO)C=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O PZGIUJSBVQXMEZ-UHFFFAOYSA-N 0.000 description 1
- OPXAVJHLTDTHNX-UHFFFAOYSA-N CC1=CC(NC2=NC(N(C)C3=CC=C(SOOO)C=C3)=NC(F)=N2)=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=NC(N(C)C3=CC=C(SOOO)C=C3)=NC(F)=N2)=CC=C1S(=O)(=O)O OPXAVJHLTDTHNX-UHFFFAOYSA-N 0.000 description 1
- SGTGRJVREYMPGS-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=C(S(=O)(=O)O)C=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)C=C3)=NC(N3=CC(C(=O)O)=CC=C3)=N2)=C(S(=O)(=O)O)C=C1SOOO Chemical compound CC1=CC(NC2=NC(NC3=C(S(=O)(=O)O)C=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)C=C3)=NC(N3=CC(C(=O)O)=CC=C3)=N2)=C(S(=O)(=O)O)C=C1SOOO SGTGRJVREYMPGS-UHFFFAOYSA-N 0.000 description 1
- LMZTYHCIXOIPEV-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(Cl)=C4)=CC=C3OS(=O)O)=NC(Cl)=N2)=CC=C1SOOO Chemical compound CC1=CC(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(Cl)=C4)=CC=C3OS(=O)O)=NC(Cl)=N2)=CC=C1SOOO LMZTYHCIXOIPEV-UHFFFAOYSA-N 0.000 description 1
- BFDUNKYMWYUEBV-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C(S(=O)(=O)O)C=C3)=NC(NC3=CC=C(SO(O)CCOS(=O)(=O)O)C=C3)=N2)=CC=C1SOOO Chemical compound CC1=CC(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C(S(=O)(=O)O)C=C3)=NC(NC3=CC=C(SO(O)CCOS(=O)(=O)O)C=C3)=N2)=CC=C1SOOO BFDUNKYMWYUEBV-UHFFFAOYSA-N 0.000 description 1
- XNPFAYNKVNXWDV-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=CC=C3)=NC(Cl)=N2)=CC=C1SOOO Chemical compound CC1=CC(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=CC=C3)=NC(Cl)=N2)=CC=C1SOOO XNPFAYNKVNXWDV-UHFFFAOYSA-N 0.000 description 1
- NIISIDHHFQZICY-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=CC=C3OS(=O)O)=NC(NC3=CC=CC([SH](CCOS(=O)(=O)O)OO)=C3)=N2)=CC=C1SOOO Chemical compound CC1=CC(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=CC=C3OS(=O)O)=NC(NC3=CC=CC([SH](CCOS(=O)(=O)O)OO)=C3)=N2)=CC=C1SOOO NIISIDHHFQZICY-UHFFFAOYSA-N 0.000 description 1
- ANQGAUIVMCURRH-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)C=C3)=NC(Cl)=N2)=CC=C1SOOO Chemical compound CC1=CC(NC2=NC(NC3=CC=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)C=C3)=NC(Cl)=N2)=CC=C1SOOO ANQGAUIVMCURRH-UHFFFAOYSA-N 0.000 description 1
- WQSJPRHMYPAHJN-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC=CC(S(=O)(=O)O)=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=NC(NC3=CC=CC(S(=O)(=O)O)=C3)=NC(Cl)=N2)=CC=C1S(=O)(=O)O WQSJPRHMYPAHJN-UHFFFAOYSA-N 0.000 description 1
- BNLGTQNTKITRCX-UHFFFAOYSA-N CC1=CC(NC2=NC(NC3=CC=CC=C3NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=NC(F)=N2)=CC=C1SOOO Chemical compound CC1=CC(NC2=NC(NC3=CC=CC=C3NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=NC(F)=N2)=CC=C1SOOO BNLGTQNTKITRCX-UHFFFAOYSA-N 0.000 description 1
- NZPVAOKVAHHWFY-UHFFFAOYSA-N CC1=CC(NC2=NC(NCCNO[Ce])=NC(Cl)=N2)=CC=C1S(=O)(=O)O Chemical compound CC1=CC(NC2=NC(NCCNO[Ce])=NC(Cl)=N2)=CC=C1S(=O)(=O)O NZPVAOKVAHHWFY-UHFFFAOYSA-N 0.000 description 1
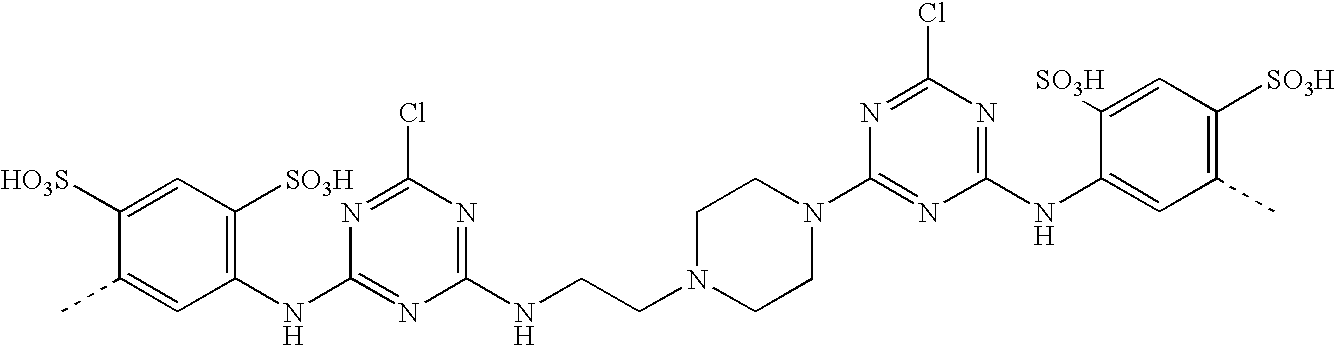
- SXNGAPXBTPCBJA-UHFFFAOYSA-N CC1=CC(S(=O)(=O)O)=C(C)C=C1NC1=NC(Cl)=NC(N2CCN(CCNC3=NC(Cl)=NC(NC4=C(S(=O)(=O)O)C=C(SOOO)C(C)=C4)=N3)CC2)=N1 Chemical compound CC1=CC(S(=O)(=O)O)=C(C)C=C1NC1=NC(Cl)=NC(N2CCN(CCNC3=NC(Cl)=NC(NC4=C(S(=O)(=O)O)C=C(SOOO)C(C)=C4)=N3)CC2)=N1 SXNGAPXBTPCBJA-UHFFFAOYSA-N 0.000 description 1
- KNGZDCRBFPTPNZ-UHFFFAOYSA-N CC1=CC(S(=O)(=O)O)=C(C)C=C1NC1=NC(Cl)=NC(NC2=C(S(=O)(=O)O)C=C(NC3=NC(Cl)=NC(NC4=C(S(=O)(=O)O)C=C(SOOO)C(C)=C4)=N3)C=C2)=N1 Chemical compound CC1=CC(S(=O)(=O)O)=C(C)C=C1NC1=NC(Cl)=NC(NC2=C(S(=O)(=O)O)C=C(NC3=NC(Cl)=NC(NC4=C(S(=O)(=O)O)C=C(SOOO)C(C)=C4)=N3)C=C2)=N1 KNGZDCRBFPTPNZ-UHFFFAOYSA-N 0.000 description 1
- LSKXRQNEHXMDQQ-UHFFFAOYSA-N CC1=CC(S(=O)(=O)O)=C(C)C=C1NC1=NC(Cl)=NC=N1.CNCCNC1=NC(Cl)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 Chemical compound CC1=CC(S(=O)(=O)O)=C(C)C=C1NC1=NC(Cl)=NC=N1.CNCCNC1=NC(Cl)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 LSKXRQNEHXMDQQ-UHFFFAOYSA-N 0.000 description 1
- YGHFQDDLMGZONS-UHFFFAOYSA-N CC1=CC(S(=O)(=O)O)=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1NC1=NC(Cl)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 Chemical compound CC1=CC(S(=O)(=O)O)=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1NC1=NC(Cl)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 YGHFQDDLMGZONS-UHFFFAOYSA-N 0.000 description 1
- KSSIJEXQTWFYBS-UHFFFAOYSA-N CC1=CC(S(=O)(=O)O)=C(NC2=NC(C)=NC(Cl)=N2)C=C1 Chemical compound CC1=CC(S(=O)(=O)O)=C(NC2=NC(C)=NC(Cl)=N2)C=C1 KSSIJEXQTWFYBS-UHFFFAOYSA-N 0.000 description 1
- GWVJIAHICLIACH-UHFFFAOYSA-N CC1=CC(S(=O)(=O)O)=C(NC2=NC(C)=NC(F)=N2)C=C1S(=O)(=O)O Chemical compound CC1=CC(S(=O)(=O)O)=C(NC2=NC(C)=NC(F)=N2)C=C1S(=O)(=O)O GWVJIAHICLIACH-UHFFFAOYSA-N 0.000 description 1
- ZBPBWTXDCUOZBT-UHFFFAOYSA-N CC1=CC(SOOO)=C(C)C=C1 Chemical compound CC1=CC(SOOO)=C(C)C=C1 ZBPBWTXDCUOZBT-UHFFFAOYSA-N 0.000 description 1
- NUHQMDNPTWPDSB-UHFFFAOYSA-N CC1=CC(SOOO)=CC=C1 Chemical compound CC1=CC(SOOO)=CC=C1 NUHQMDNPTWPDSB-UHFFFAOYSA-N 0.000 description 1
- QCWBVCNHQOXOLG-UHFFFAOYSA-N CC1=CC(SOOO)=CC=C1S(=O)(=O)O Chemical compound CC1=CC(SOOO)=CC=C1S(=O)(=O)O QCWBVCNHQOXOLG-UHFFFAOYSA-N 0.000 description 1
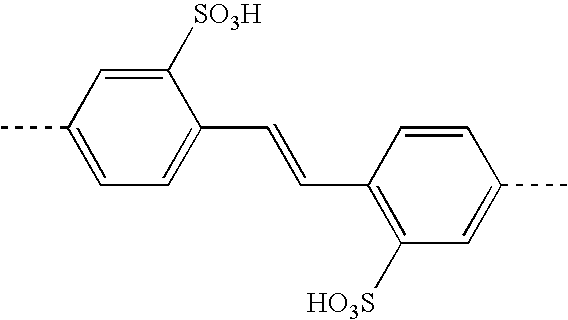
- BFRBWJUMOZLXLT-BQYQJAHWSA-N CC1=CC=C(/C=C/C2=C(SOOO)C=C(C)C=C2)C(S(=O)(=O)O)=C1 Chemical compound CC1=CC=C(/C=C/C2=C(SOOO)C=C(C)C=C2)C(S(=O)(=O)O)=C1 BFRBWJUMOZLXLT-BQYQJAHWSA-N 0.000 description 1
- CAFIIZXOHDZHLD-UHFFFAOYSA-N CC1=CC=C(C2=C(SOOO)C=C(C)C=C2)C(S(=O)(=O)O)=C1 Chemical compound CC1=CC=C(C2=C(SOOO)C=C(C)C=C2)C(S(=O)(=O)O)=C1 CAFIIZXOHDZHLD-UHFFFAOYSA-N 0.000 description 1
- ICSCYSJFLVXYOX-UHFFFAOYSA-N CC1=CC=C(CCCOSOOO)C=C1 Chemical compound CC1=CC=C(CCCOSOOO)C=C1 ICSCYSJFLVXYOX-UHFFFAOYSA-N 0.000 description 1
- UXOFNNFABVDHRZ-UHFFFAOYSA-N CC1=CC=C(CNC2=NC(NCCNC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=NC(Cl)=N2)C=C1 Chemical compound CC1=CC=C(CNC2=NC(NCCNC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=NC(Cl)=N2)C=C1 UXOFNNFABVDHRZ-UHFFFAOYSA-N 0.000 description 1
- XBYAFZQTQXDDSC-UHFFFAOYSA-N CC1=CC=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1NC1=NC(F)=NC(NC2=C(S(=O)(=O)O)C=C(SOOO)C(C)=C2)=N1 Chemical compound CC1=CC=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1NC1=NC(F)=NC(NC2=C(S(=O)(=O)O)C=C(SOOO)C(C)=C2)=N1 XBYAFZQTQXDDSC-UHFFFAOYSA-N 0.000 description 1
- LRCDZTXDRCQWMJ-UHFFFAOYSA-N CC1=CC=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1NC1=NC(F)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 Chemical compound CC1=CC=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1NC1=NC(F)=NC(NC2=CC=C(SOOO)C(C)=C2)=N1 LRCDZTXDRCQWMJ-UHFFFAOYSA-N 0.000 description 1
- CPYHHBQJAKAYES-UHFFFAOYSA-O CC1=CC=C(NC2=NC(C)=NC([N+]3=CC(C(=O)O)=CC=C3)=N2)C=C1S(=O)(=O)O Chemical compound CC1=CC=C(NC2=NC(C)=NC([N+]3=CC(C(=O)O)=CC=C3)=N2)C=C1S(=O)(=O)O CPYHHBQJAKAYES-UHFFFAOYSA-O 0.000 description 1
- VSPGRDFHXGHNCS-UHFFFAOYSA-O CC1=CC=C(NC2=NC(C)=NC([N+]3=CC=C(C(=O)O)C=C3)=N2)C=C1 Chemical compound CC1=CC=C(NC2=NC(C)=NC([N+]3=CC=C(C(=O)O)C=C3)=N2)C=C1 VSPGRDFHXGHNCS-UHFFFAOYSA-O 0.000 description 1
- MDVGCKCRZAGYJU-UHFFFAOYSA-N CC1=CC=C(NC2=NC(Cl)=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=CC=C3S(=O)(=O)O)=N2)C(S(=O)(=O)O)=C1 Chemical compound CC1=CC=C(NC2=NC(Cl)=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=CC=C3S(=O)(=O)O)=N2)C(S(=O)(=O)O)=C1 MDVGCKCRZAGYJU-UHFFFAOYSA-N 0.000 description 1
- KISAUXKQODEGOH-UHFFFAOYSA-N CC1=CC=C(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C(S(=O)(=O)O)C=C3)=NC(F)=N2)C=C1SOOO Chemical compound CC1=CC=C(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C(S(=O)(=O)O)C=C3)=NC(F)=N2)C=C1SOOO KISAUXKQODEGOH-UHFFFAOYSA-N 0.000 description 1
- VTYGJRRABRVKRS-UHFFFAOYSA-N CC1=CC=C(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C(S(=O)(=O)O)C=C3C)=NC(Cl)=N2)C=C1 Chemical compound CC1=CC=C(NC2=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C(S(=O)(=O)O)C=C3C)=NC(Cl)=N2)C=C1 VTYGJRRABRVKRS-UHFFFAOYSA-N 0.000 description 1
- WSOARTNEKOMTSU-UHFFFAOYSA-N CC1=CC=C(NC2=NC(NCCCNC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=NC(F)=N2)C=C1C Chemical compound CC1=CC=C(NC2=NC(NCCCNC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=NC(F)=N2)C=C1C WSOARTNEKOMTSU-UHFFFAOYSA-N 0.000 description 1
- JPWXCUWXCWDEMO-UHFFFAOYSA-N CC1=CC=C(NC2=NC(NCCN3CCN(C4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)CC3)=NC(Cl)=N2)C=C1SOOO Chemical compound CC1=CC=C(NC2=NC(NCCN3CCN(C4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)CC3)=NC(Cl)=N2)C=C1SOOO JPWXCUWXCWDEMO-UHFFFAOYSA-N 0.000 description 1
- ZASXSXHDMAHYDI-UHFFFAOYSA-N CC1=CC=C(NC2=NC(NCCNC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=NC(Cl)=N2)C=C1SOOO Chemical compound CC1=CC=C(NC2=NC(NCCNC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)=NC(Cl)=N2)C=C1SOOO ZASXSXHDMAHYDI-UHFFFAOYSA-N 0.000 description 1
- INCBFVBICVXKHY-UHFFFAOYSA-N CC1=CC=C(S(=O)(=O)O)C(NC2=NC(Cl)=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C(S(=O)(=O)O)C=C3S(=O)(=O)O)=N2)=C1 Chemical compound CC1=CC=C(S(=O)(=O)O)C(NC2=NC(Cl)=NC(NC3=CC(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)=C(S(=O)(=O)O)C=C3S(=O)(=O)O)=N2)=C1 INCBFVBICVXKHY-UHFFFAOYSA-N 0.000 description 1
- SREMWZSURQFEFS-UHFFFAOYSA-N CC1=CC=C(SO(O)C2=CC=C(C)C=C2)C=C1 Chemical compound CC1=CC=C(SO(O)C2=CC=C(C)C=C2)C=C1 SREMWZSURQFEFS-UHFFFAOYSA-N 0.000 description 1
- MOXLNBCPCZQVME-UHFFFAOYSA-N CC1=CC=C(SOOO)C=C1 Chemical compound CC1=CC=C(SOOO)C=C1 MOXLNBCPCZQVME-UHFFFAOYSA-N 0.000 description 1
- YXFVVABEGXRONW-UHFFFAOYSA-N CC1=CC=CC=C1 Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 1
- SPXXCMKFDVVWRL-UHFFFAOYSA-N CC1=NC(Cl)=NC(NC2=C(S(=O)(=O)O)C=C(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)C(S(=O)(=O)O)=C2)=N1 Chemical compound CC1=NC(Cl)=NC(NC2=C(S(=O)(=O)O)C=C(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)C(S(=O)(=O)O)=C2)=N1 SPXXCMKFDVVWRL-UHFFFAOYSA-N 0.000 description 1
- UYIYMGIPCNMUEQ-UHFFFAOYSA-N CC1=NC(Cl)=NC(NC2=CC(NC3=C([N+](=O)[O-])C=C(C#N)C(F)=C3)=C(S(=O)(=O)O)C=C2S(=O)(=O)O)=N1 Chemical compound CC1=NC(Cl)=NC(NC2=CC(NC3=C([N+](=O)[O-])C=C(C#N)C(F)=C3)=C(S(=O)(=O)O)C=C2S(=O)(=O)O)=N1 UYIYMGIPCNMUEQ-UHFFFAOYSA-N 0.000 description 1
- FTFGRPMHUNJHRF-UHFFFAOYSA-N CC1=NC(Cl)=NC(NC2=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(Cl)=C3)=CC=C2S(=O)(=O)O)=N1 Chemical compound CC1=NC(Cl)=NC(NC2=CC(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(Cl)=C3)=CC=C2S(=O)(=O)O)=N1 FTFGRPMHUNJHRF-UHFFFAOYSA-N 0.000 description 1
- CWUNWTDMQOCVQP-UHFFFAOYSA-N CC1=NC(Cl)=NC(NCCNC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)=N1 Chemical compound CC1=NC(Cl)=NC(NCCNC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)=N1 CWUNWTDMQOCVQP-UHFFFAOYSA-N 0.000 description 1
- IXSKRFFOBQWKAE-UHFFFAOYSA-N CC1=NC(F)=NC(NC2=CC=C(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)C(S(=O)(=O)O)=C2)=N1 Chemical compound CC1=NC(F)=NC(NC2=CC=C(NC3=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C3)C(S(=O)(=O)O)=C2)=N1 IXSKRFFOBQWKAE-UHFFFAOYSA-N 0.000 description 1
- VKQTZSLTIYVUTL-ULDVOPSXSA-N CC1=NC(NC2=C(C)C(N3N=C(C(=O)O)C(/N=N/C4=CC=C(NC5=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C5)C=C4S(=O)(=O)O)=C3O)=CC(S(=O)(=O)O)=C2)=NC(Cl)=N1 Chemical compound CC1=NC(NC2=C(C)C(N3N=C(C(=O)O)C(/N=N/C4=CC=C(NC5=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C5)C=C4S(=O)(=O)O)=C3O)=CC(S(=O)(=O)O)=C2)=NC(Cl)=N1 VKQTZSLTIYVUTL-ULDVOPSXSA-N 0.000 description 1
- MSBMKLYGKTVRNU-XAHDOWKMSA-N CC1=NC(NC2=CC(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=CC=C2/N=N/C2=C(S(=O)(=O)O)C=C3C=C(SOOO)C=C(S(=O)(=O)O)C3=C2)=NC(Cl)=N1 Chemical compound CC1=NC(NC2=CC(NC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=CC=C2/N=N/C2=C(S(=O)(=O)O)C=C3C=C(SOOO)C=C(S(=O)(=O)O)C3=C2)=NC(Cl)=N1 MSBMKLYGKTVRNU-XAHDOWKMSA-N 0.000 description 1
- XNEZMDMUSLJQSC-UHFFFAOYSA-N CC1=NC(NC2=CC=CC=C2)=NC(NC2=CC=C(S(=O)(=O)CCOS(=O)(=O)O)C=C2)=N1 Chemical compound CC1=NC(NC2=CC=CC=C2)=NC(NC2=CC=C(S(=O)(=O)CCOS(=O)(=O)O)C=C2)=N1 XNEZMDMUSLJQSC-UHFFFAOYSA-N 0.000 description 1
- TVJQOFSFKTXYMS-UHFFFAOYSA-N CC1=NC(NC2=CC=CC=C2)=NC(NC2=CC=C(SO(O)COS(=O)(=O)O)C=C2)=N1 Chemical compound CC1=NC(NC2=CC=CC=C2)=NC(NC2=CC=C(SO(O)COS(=O)(=O)O)C=C2)=N1 TVJQOFSFKTXYMS-UHFFFAOYSA-N 0.000 description 1
- XELKQMHTUTYRNW-UHFFFAOYSA-N CC1=NC(NCCOCCO)=NC(NC2=CC=C(S(=O)(=O)CCOS(=O)(=O)O)C=C2)=N1 Chemical compound CC1=NC(NCCOCCO)=NC(NC2=CC=C(S(=O)(=O)CCOS(=O)(=O)O)C=C2)=N1 XELKQMHTUTYRNW-UHFFFAOYSA-N 0.000 description 1
- SUNWEAQHEBYYEL-UHFFFAOYSA-N CCNC1=CC=C2N=C3C(C)=C4OC5=C(C=CC(NCC)=C5C)N=C4C(C)=C3OC2=C1C Chemical compound CCNC1=CC=C2N=C3C(C)=C4OC5=C(C=CC(NCC)=C5C)N=C4C(C)=C3OC2=C1C SUNWEAQHEBYYEL-UHFFFAOYSA-N 0.000 description 1
- DVCXZWPOFWIXAV-UHFFFAOYSA-N CNC1=C(S(=O)(=O)O)C=C(NC2=NC(NC3=CC=C(S(=O)(=O)O)C(NC4=CC(F)=C([N+](=O)[O-])C=C4[N+](=O)[O-])=C3)=NC(F)=N2)C(S(=O)(=O)O)=C1 Chemical compound CNC1=C(S(=O)(=O)O)C=C(NC2=NC(NC3=CC=C(S(=O)(=O)O)C(NC4=CC(F)=C([N+](=O)[O-])C=C4[N+](=O)[O-])=C3)=NC(F)=N2)C(S(=O)(=O)O)=C1 DVCXZWPOFWIXAV-UHFFFAOYSA-N 0.000 description 1
- IJOCFVWMLHISQO-UHFFFAOYSA-N CNC1=C(S(=O)(=O)O)C=CC(N(C)C)=C1 Chemical compound CNC1=C(S(=O)(=O)O)C=CC(N(C)C)=C1 IJOCFVWMLHISQO-UHFFFAOYSA-N 0.000 description 1
- DQJBLZQDIIPPDV-UHFFFAOYSA-N CNC1=CC(S(=O)(=O)O)=C(NC)C=C1S(=O)(=O)O Chemical compound CNC1=CC(S(=O)(=O)O)=C(NC)C=C1S(=O)(=O)O DQJBLZQDIIPPDV-UHFFFAOYSA-N 0.000 description 1
- CPWLPMXXAPPGBN-ONEGZZNKSA-N CNC1=CC=C(/C=C/C2=C(SOOO)C=C(NC)C=C2)C(S(=O)(=O)O)=C1 Chemical compound CNC1=CC=C(/C=C/C2=C(SOOO)C=C(NC)C=C2)C(S(=O)(=O)O)=C1 CPWLPMXXAPPGBN-ONEGZZNKSA-N 0.000 description 1
- OVXXORDMCYZOQN-GHVJWSGMSA-N CNC1=CC=C(/N=N/C2=C(S(=O)(=O)O)C=C3C=C(SOOO)C=C(S(=O)(=O)O)C3=C2)C(NC(N)=O)=C1 Chemical compound CNC1=CC=C(/N=N/C2=C(S(=O)(=O)O)C=C3C=C(SOOO)C=C(S(=O)(=O)O)C3=C2)C(NC(N)=O)=C1 OVXXORDMCYZOQN-GHVJWSGMSA-N 0.000 description 1
- JNDNNYQYVIHCRJ-QURGRASLSA-N CNC1=CC=C(/N=N/C2=C(S(=O)(=O)O)C=C3C=C(SOOO)C=C(S(=O)(=O)O)C3=C2)C(NC)=C1 Chemical compound CNC1=CC=C(/N=N/C2=C(S(=O)(=O)O)C=C3C=C(SOOO)C=C(S(=O)(=O)O)C3=C2)C(NC)=C1 JNDNNYQYVIHCRJ-QURGRASLSA-N 0.000 description 1
- AWXUBHMURFEJMY-UHFFFAOYSA-N CNC1=CC=C(N(C)C)C=C1 Chemical compound CNC1=CC=C(N(C)C)C=C1 AWXUBHMURFEJMY-UHFFFAOYSA-N 0.000 description 1
- PVRZMTHMPKVOBP-UHFFFAOYSA-N CNC1=CC=C(NC)C=C1 Chemical compound CNC1=CC=C(NC)C=C1 PVRZMTHMPKVOBP-UHFFFAOYSA-N 0.000 description 1
- UQBNGMRDYGPUOO-UHFFFAOYSA-N CNC1=CC=CC(NC)=C1 Chemical compound CNC1=CC=CC(NC)=C1 UQBNGMRDYGPUOO-UHFFFAOYSA-N 0.000 description 1
- RHGPWAAAFFMVIH-UHFFFAOYSA-N CNC1=NC(Cl)=NC(NC2=CC(S(=O)(=O)O)=CC=C2)=N1 Chemical compound CNC1=NC(Cl)=NC(NC2=CC(S(=O)(=O)O)=CC=C2)=N1 RHGPWAAAFFMVIH-UHFFFAOYSA-N 0.000 description 1
- PNFRMZHLDYVYTK-UHFFFAOYSA-N CNC1=NC(Cl)=NC(NC2=CC=C(S(=O)(=O)O)C=C2)=N1 Chemical compound CNC1=NC(Cl)=NC(NC2=CC=C(S(=O)(=O)O)C=C2)=N1 PNFRMZHLDYVYTK-UHFFFAOYSA-N 0.000 description 1
- NSKGIXLYTHJZTI-UHFFFAOYSA-N CNC1=NC(F)=NC(NC2=CC(S(=O)(=O)CCOS(=O)(=O)O)=CC=C2)=N1 Chemical compound CNC1=NC(F)=NC(NC2=CC(S(=O)(=O)CCOS(=O)(=O)O)=CC=C2)=N1 NSKGIXLYTHJZTI-UHFFFAOYSA-N 0.000 description 1
- KPASVHQXQQFJTO-UHFFFAOYSA-N CNCC1=C(NC)C=CC=C1 Chemical compound CNCC1=C(NC)C=CC=C1 KPASVHQXQQFJTO-UHFFFAOYSA-N 0.000 description 1
- LNDGSYRDGSSTLI-UHFFFAOYSA-N CNCC1=CC=C(NC)C=C1 Chemical compound CNCC1=CC=C(NC)C=C1 LNDGSYRDGSSTLI-UHFFFAOYSA-N 0.000 description 1
- OGFXPDAIXCWRDX-UHFFFAOYSA-M C[V]NC1=CC(S(=O)(=O)O)=C(C)C2=C1C(=O)C1=CC=CC=C1C2=O Chemical compound C[V]NC1=CC(S(=O)(=O)O)=C(C)C2=C1C(=O)C1=CC=CC=C1C2=O OGFXPDAIXCWRDX-UHFFFAOYSA-M 0.000 description 1
- LTYBYSFEGHYUDB-UHFFFAOYSA-N Cc(c(S(O)(=O)=O)c1)ccc1S(O)(=O)=O Chemical compound Cc(c(S(O)(=O)=O)c1)ccc1S(O)(=O)=O LTYBYSFEGHYUDB-UHFFFAOYSA-N 0.000 description 1
- ZIIOAIUTJGCVQQ-UHFFFAOYSA-N Cc(ccc(Nc1nc(Cl)nc(NCCN(CC2)CCN2c(cc(c([N+]([O-])=O)c2)F)c2[N+]([O-])=O)n1)c1)c1S(O)(=O)=O Chemical compound Cc(ccc(Nc1nc(Cl)nc(NCCN(CC2)CCN2c(cc(c([N+]([O-])=O)c2)F)c2[N+]([O-])=O)n1)c1)c1S(O)(=O)=O ZIIOAIUTJGCVQQ-UHFFFAOYSA-N 0.000 description 1
- JAPCBKZBZUMSOQ-UHFFFAOYSA-N Cc1c(C)c(Nc2nc(Cl)nc(Nc(ccc(Nc(c([N+]([O-])=O)c3)cc(Cl)c3[N+]([O-])=O)c3)c3S(O)(=O)=O)n2)c(C)c(S(O)(=O)=O)c1C Chemical compound Cc1c(C)c(Nc2nc(Cl)nc(Nc(ccc(Nc(c([N+]([O-])=O)c3)cc(Cl)c3[N+]([O-])=O)c3)c3S(O)(=O)=O)n2)c(C)c(S(O)(=O)=O)c1C JAPCBKZBZUMSOQ-UHFFFAOYSA-N 0.000 description 1
- DIOILCAIBASXMB-UHFFFAOYSA-N Cc1cc(Nc(c([N+]([O-])=O)c2)cc(F)c2C#N)ccc1S(O)(=O)=O Chemical compound Cc1cc(Nc(c([N+]([O-])=O)c2)cc(F)c2C#N)ccc1S(O)(=O)=O DIOILCAIBASXMB-UHFFFAOYSA-N 0.000 description 1
- GHTBENICNSFIMT-UHFFFAOYSA-N Cc1cc(Nc2nc(Cl)nc(Nc(cc3)ccc3S(O)(=O)=O)n2)ccc1S(O)(=O)=O Chemical compound Cc1cc(Nc2nc(Cl)nc(Nc(cc3)ccc3S(O)(=O)=O)n2)ccc1S(O)(=O)=O GHTBENICNSFIMT-UHFFFAOYSA-N 0.000 description 1
- WAORBOMKYPLLGG-UHFFFAOYSA-N Cc1cc(Nc2nc(F)nc(N(C)c(cc3)ccc3S(O)(=O)=O)n2)ccc1S(O)(=O)=O Chemical compound Cc1cc(Nc2nc(F)nc(N(C)c(cc3)ccc3S(O)(=O)=O)n2)ccc1S(O)(=O)=O WAORBOMKYPLLGG-UHFFFAOYSA-N 0.000 description 1
- FTBFHGAQUAJCSU-UHFFFAOYSA-N Cc1ccc(-c(ccc(C)c2)c2S(O)(=O)=O)c(S(O)(=O)=O)c1 Chemical compound Cc1ccc(-c(ccc(C)c2)c2S(O)(=O)=O)c(S(O)(=O)=O)c1 FTBFHGAQUAJCSU-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 241000195493 Cryptophyta Species 0.000 description 1
- 241000233866 Fungi Species 0.000 description 1
- 229920000433 Lyocell Polymers 0.000 description 1
- DDTSCELTJPSQRX-CPHIHMHPSA-N NC(=O)NC1=CC(NC2=NC(NC3=CC=C(N4CCN(C5=CC(F)=C([N+](=O)[O-])C=C5[N+](=O)[O-])CC4)C=C3)=NC(Cl)=N2)=CC=C1/N=N/C1=C(S(=O)(=O)O)C=C2C=C(S(O)(O)O)C=C(S(=O)(=O)O)C2=C1 Chemical compound NC(=O)NC1=CC(NC2=NC(NC3=CC=C(N4CCN(C5=CC(F)=C([N+](=O)[O-])C=C5[N+](=O)[O-])CC4)C=C3)=NC(Cl)=N2)=CC=C1/N=N/C1=C(S(=O)(=O)O)C=C2C=C(S(O)(O)O)C=C(S(=O)(=O)O)C2=C1 DDTSCELTJPSQRX-CPHIHMHPSA-N 0.000 description 1
- OHHATHRXAKJVNQ-HBSCQBRPSA-N NC(=O)NC1=CC(NC2=NC(NCCNCCNC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=NC(Cl)=N2)=CC=C1/N=N/C1=C(S(=O)(=O)O)C=C2C=C(SOOO)C=C(S(=O)(=O)O)C2=C1 Chemical compound NC(=O)NC1=CC(NC2=NC(NCCNCCNC3=CC(F)=C([N+](=O)[O-])C=C3[N+](=O)[O-])=NC(Cl)=N2)=CC=C1/N=N/C1=C(S(=O)(=O)O)C=C2C=C(SOOO)C=C(S(=O)(=O)O)C2=C1 OHHATHRXAKJVNQ-HBSCQBRPSA-N 0.000 description 1
- IXZJXKKKUODOLO-BYYHNAKLSA-N O=CNC1=C(/N=N/C2=C(S(=O)(=O)O)C=C3C=C(SOOO)C=C(S(=O)(=O)O)C3=C2)C=CC(NC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])=C1 Chemical compound O=CNC1=C(/N=N/C2=C(S(=O)(=O)O)C=C3C=C(SOOO)C=C(S(=O)(=O)O)C3=C2)C=CC(NC2=CC(F)=C([N+](=O)[O-])C=C2[N+](=O)[O-])=C1 IXZJXKKKUODOLO-BYYHNAKLSA-N 0.000 description 1
- BHVVEFPUYKSWNC-UHFFFAOYSA-N O=S(=O)=O.O=S(=O)=O.[H]C1=CC=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1N=NC1=C(O)C2=C(N)C(N=NC3=CC=C(SOOCCS(=O)(O)(O)O)C=C3)=C([H])C=C2C=C1S(=O)(=O)O Chemical compound O=S(=O)=O.O=S(=O)=O.[H]C1=CC=C(NC2=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C2)C=C1N=NC1=C(O)C2=C(N)C(N=NC3=CC=C(SOOCCS(=O)(O)(O)O)C=C3)=C([H])C=C2C=C1S(=O)(=O)O BHVVEFPUYKSWNC-UHFFFAOYSA-N 0.000 description 1
- COJLIMLKVCPTRO-UHFFFAOYSA-N O=S(=O)=O.[H]C1=C(NC)C=C(NC)C=C1 Chemical compound O=S(=O)=O.[H]C1=C(NC)C=C(NC)C=C1 COJLIMLKVCPTRO-UHFFFAOYSA-N 0.000 description 1
- QJWWTNLEWUFCFG-IRWWKPKRSA-N O=S(=O)=O.[H]C1=CC(NC)=C(C)C(N2N=C(C(=O)O)C(/N=N/C3=CC=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)C=C3S(=O)(=O)O)=C2O)=C1 Chemical compound O=S(=O)=O.[H]C1=CC(NC)=C(C)C(N2N=C(C(=O)O)C(/N=N/C3=CC=C(NC4=C([N+](=O)[O-])C=C([N+](=O)[O-])C(F)=C4)C=C3S(=O)(=O)O)=C2O)=C1 QJWWTNLEWUFCFG-IRWWKPKRSA-N 0.000 description 1
- DUFKCOQISQKSAV-UHFFFAOYSA-N Polypropylene glycol (m w 1,200-3,000) Chemical compound CC(O)COC(C)CO DUFKCOQISQKSAV-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 239000004146 Propane-1,2-diol Substances 0.000 description 1
- 229920000297 Rayon Polymers 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- PJANXHGTPQOBST-VAWYXSNFSA-N Stilbene Natural products C=1C=CC=CC=1/C=C/C1=CC=CC=C1 PJANXHGTPQOBST-VAWYXSNFSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 125000004442 acylamino group Chemical group 0.000 description 1
- 229910000288 alkali metal carbonate Inorganic materials 0.000 description 1
- 150000008041 alkali metal carbonates Chemical class 0.000 description 1
- 229910000318 alkali metal phosphate Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 239000001000 anthraquinone dye Substances 0.000 description 1
- 125000000732 arylene group Chemical group 0.000 description 1
- 238000006149 azo coupling reaction Methods 0.000 description 1
- WDEQGLDWZMIMJM-UHFFFAOYSA-N benzyl 4-hydroxy-2-(hydroxymethyl)pyrrolidine-1-carboxylate Chemical compound OCC1CC(O)CN1C(=O)OCC1=CC=CC=C1 WDEQGLDWZMIMJM-UHFFFAOYSA-N 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 235000019437 butane-1,3-diol Nutrition 0.000 description 1
- 150000001733 carboxylic acid esters Chemical class 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 239000006184 cosolvent Substances 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- AFOSIXZFDONLBT-UHFFFAOYSA-N divinyl sulfone Chemical group C=CS(=O)(=O)C=C AFOSIXZFDONLBT-UHFFFAOYSA-N 0.000 description 1
- 229940093476 ethylene glycol Drugs 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 210000004209 hair Anatomy 0.000 description 1
- 150000005694 halopyrimidines Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 150000007857 hydrazones Chemical class 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 150000002500 ions Chemical group 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000010985 leather Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 239000010446 mirabilite Substances 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000001007 phthalocyanine dye Substances 0.000 description 1
- 229920002239 polyacrylonitrile Polymers 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 125000006239 protecting group Chemical group 0.000 description 1
- 125000005494 pyridonyl group Chemical group 0.000 description 1
- 239000004627 regenerated cellulose Substances 0.000 description 1
- 238000001223 reverse osmosis Methods 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 235000017550 sodium carbonate Nutrition 0.000 description 1
- 235000011121 sodium hydroxide Nutrition 0.000 description 1
- 235000019795 sodium metasilicate Nutrition 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- PJANXHGTPQOBST-UHFFFAOYSA-N stilbene Chemical compound C=1C=CC=CC=1C=CC1=CC=CC=C1 PJANXHGTPQOBST-UHFFFAOYSA-N 0.000 description 1
- 235000021286 stilbenes Nutrition 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 229910021653 sulphate ion Inorganic materials 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 239000007966 viscous suspension Substances 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000001043 yellow dye Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B62/00—Reactive dyes, i.e. dyes which form covalent bonds with the substrates or which polymerise with themselves
- C09B62/44—Reactive dyes, i.e. dyes which form covalent bonds with the substrates or which polymerise with themselves with the reactive group not directly attached to a heterocyclic ring
- C09B62/78—Reactive dyes, i.e. dyes which form covalent bonds with the substrates or which polymerise with themselves with the reactive group not directly attached to a heterocyclic ring with other reactive groups
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B62/00—Reactive dyes, i.e. dyes which form covalent bonds with the substrates or which polymerise with themselves
- C09B62/44—Reactive dyes, i.e. dyes which form covalent bonds with the substrates or which polymerise with themselves with the reactive group not directly attached to a heterocyclic ring
- C09B62/4401—Reactive dyes, i.e. dyes which form covalent bonds with the substrates or which polymerise with themselves with the reactive group not directly attached to a heterocyclic ring with two or more reactive groups at least one of them being directly attached to a heterocyclic system and at least one of them being directly attached to a non-heterocyclic system
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B62/00—Reactive dyes, i.e. dyes which form covalent bonds with the substrates or which polymerise with themselves
- C09B62/44—Reactive dyes, i.e. dyes which form covalent bonds with the substrates or which polymerise with themselves with the reactive group not directly attached to a heterocyclic ring
- C09B62/443—Reactive dyes, i.e. dyes which form covalent bonds with the substrates or which polymerise with themselves with the reactive group not directly attached to a heterocyclic ring the reactive group being alternatively specified
Definitions
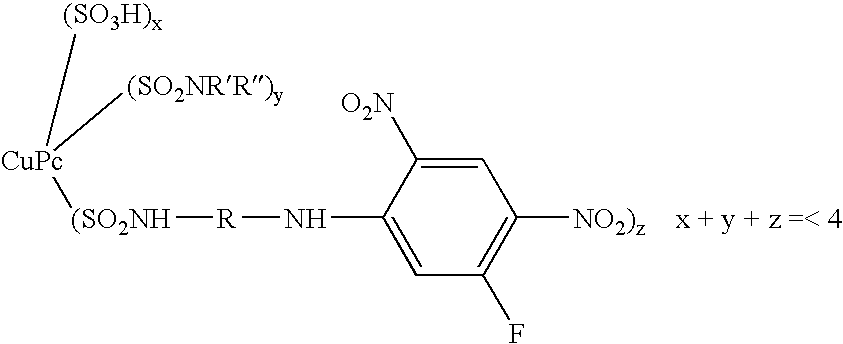
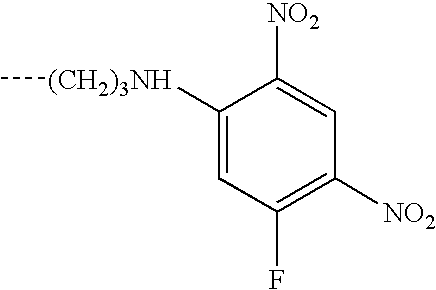
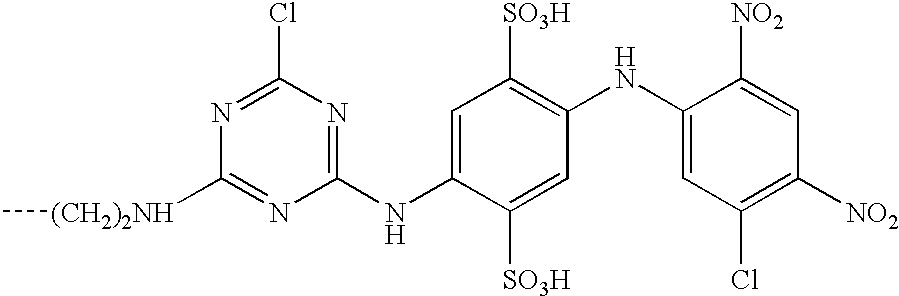
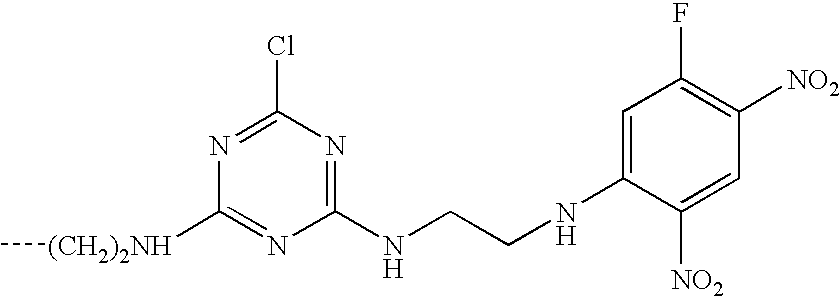
- This invention relates to reactive dyes containing a halobenzene nucleus and, in particular, reactive dyes of this type containing two or more reactive components.
- Dyes which contain a halobenzene nucleus linked via an azo group to another aromatic nucleus such that the halobenzene nucleus forms part of the chromophoric chain (see for example GB-A-882001).
- Dyes of this type which contain two such halobenzene nuclei are disclosed in CA64,14316d (1966), which is an English language abstract of an article by Matsui et al, Yuki Gosei Kagaku Kyokai Shi(1966), 24-(2), 132-136.
- Dyes are also known in which the halobenzene nucleus is attached to a chromophoric group by a sulphonamide or amide linkage; see, for example, GB-A-978162 and CA59,12949g (1963), which is an English language abstract of an article by Matsui et al in Yuki Gosei Kagaku Kyokai Shi (1962), 20,1100-1112. Again dyes of this type may contain two such halobenzene nuclei; see GB-A-978162.
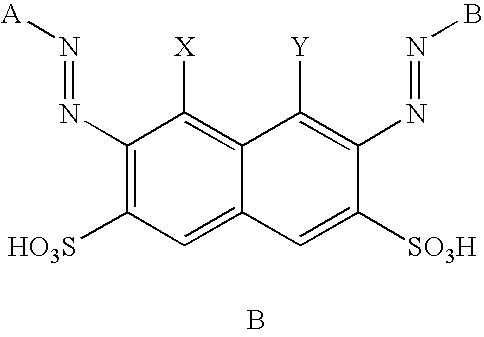
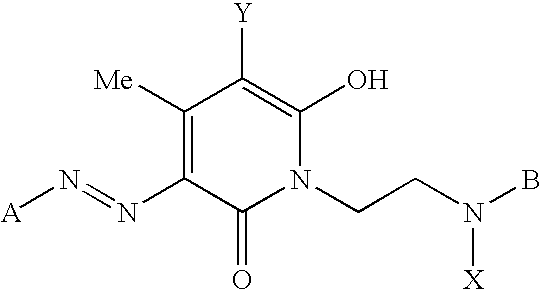
- n 1 or 2
- X or each X independently, is an electron withdrawing group
- Y is a halogen atom
- m is 1 or 2; p is 0 or 1; when m is 1, p is 1; and when m is 2, p is 0;
- Y 1 or each Y 1 independently, is a halogen atom or an optionally substituted pyridinium group
- T is C 1-4 alkoxy, C 1-4 thioalkoxy or N(R 1 ) (R 2 ), in which each of R 1 and R 2 independently is hydrogen, optionally substituted C 1-4 alkyl or optionally substituted aryl;
- Y 2 is a halogen atom or an optionally substituted pyridinium group
- U or each U independently, is C 1-4 alkyl or C 1-4 alkylsulphonyl
- X 1 is an eliminatable group
- z is zero or 1
- R 10 is hydrogen, C 1-4 alkyl or halogen
- W is —OC( ⁇ O)— or —N(R 11 )C( ⁇ O)— in which R 11 is hydrogen or C 1-4 alkyl;
- At least a first linking group L 1 linking the said first, halobenzene, reactive group Z 1 to one of components (i) the or a chromophore D and (ii) the second reactive group Z 2 , which said first linking group L 1 presents an amino nitrogen to the reactive group Z 1 and to the component (i) or (ii) or, when component (i) contains a heterocyclic nitrogen atom, is linked directly to the nitrogen atom and which said first linking group L 1 optionally includes a hydrocarbon bridging group, which hydrocarbon bridging group B has at least two carbon atoms, is optionally substituted, optionally includes at least one hetero atom and is optionally a chromophore; and
- optionally at least one aromatic group Ar which, when Z 2 is selected from the said groups (IV)-(VI), may carry the said reactive group Z 2 .
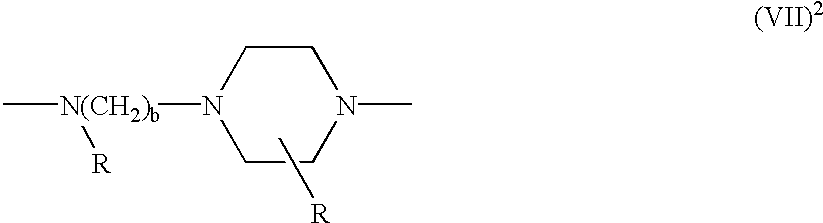
- the linking group L 1 has the formula (VII) 1
- R is hydrogen or optionally substituted C 1-4 alkyl, such that the same amino group presents itself to each of the reactive group Z 1 and the component (i) or (ii), as defined above; or
- each R independently, is as defined above, such that respective amino nitrogens, one of the piperazine group and the other of the alkylamino group, present themselves respectively, to the reactive group Z 1 and to the component (i) or (ii), as defined above; or
- B is a hydrocarbon bridging group as defined above, each R, independently , is as defined above and B is optionally linked additionally to at least one additional group —N(R).
- the hydrocarbon bridging group B may be a straight or branched, optionally substituted, C 2-6 alkylene group optionally interrupted by at least one hetero atom, for example, O,S or N.
- Optional substituents are OH alkoxy, carboxy, carboxylic ester or carboxamide.
- the bonding group B may be an optionally substituted arylene especially phenylene group.
- Optional substituents are SO 3 H and salts thereof, C 1-4 alkyl, C 1-4 alkoxy and chloro.
- the bridging group B is is especially preferably an optionally substituted aryl group.
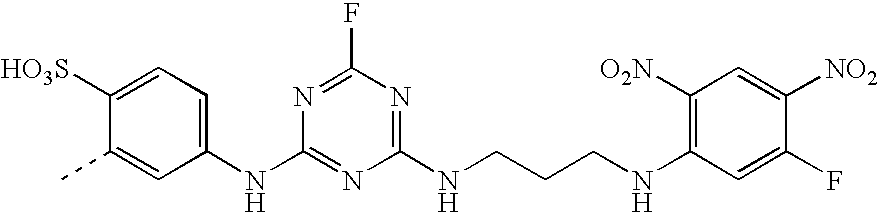
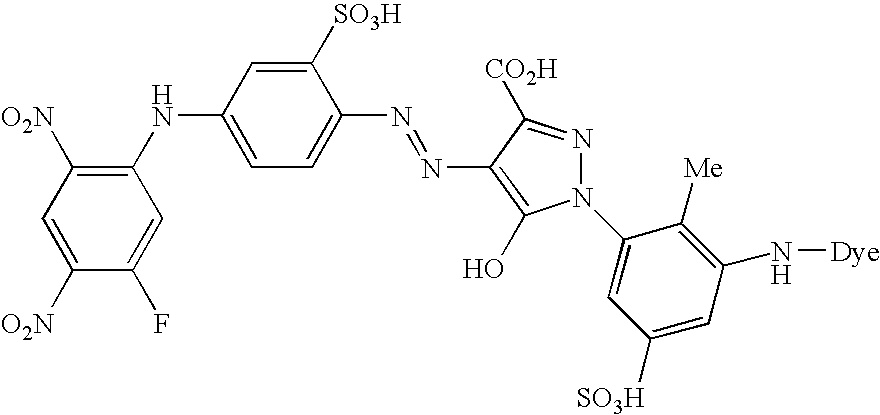
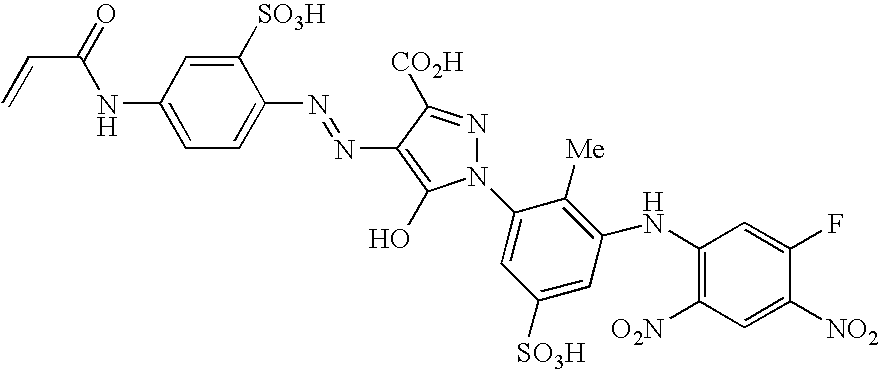
- An especially preferred dye embodying the invention has the formula (VIII)
- D is a chromophore
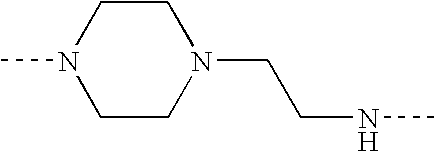
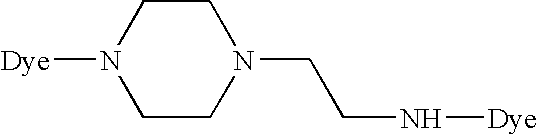
- each of L 1 and L 2 is an amine or piperazine linkage of the formula
- R or each R independently, is hydrogen or C 1-4 alkyl
- B is a hydrocarbon bridging group which has at least two carbon atoms, is optionally substituted, optionally includes at least one hetero atom and is optionally a chromophore;
- a is zero or 1
- b is from 2 to 6 inclusive;
- Z 1 is a group
- n 1 or 2;
- X or each X independently, is an electron withdrawing group
- Y is a halogen atom
- m is 1 or 2; p is 0 or 1; when m is 1, p is 1; and when m is 2, p is 0;
- Y 1 is a halogen atom or an optionally substituted pyridinium group
- T is C 1-4 alkoxy, thioalkoxy or N(R 1 ) (R 2 ) in which R 1 is hydrogen, optionally substituted C 1-4 alkyl or optionally substituted aryl and
- R 2 is hydrogen or optionally substituted C 1-4 alkyl
- Y 2 is a halogen atom
- U or each U independently, is C 1-4 alkyl or C 1-4 alkylsulphonyl
- R 10 is hydrogen, C 1-4 alkyl or halogen
- W is —OC( ⁇ O)— or —N(R 11 )C( ⁇ O)— in which R 11 is hydrogen or C 1-4 alkyl.
- X or each X independently may be selected from nitro, cyano, alkylsulphonyl, dialkylaminosulphonyl and sulphonic acid groups and salts thereof.
- X or each X independently is selected from nitro and cyano.
- the halogen atom in the halobenzene nucleus of formula (I) is preferably fluorine or chlorine.
- the group R in the linking group L 1 of formulae (1)-(3) may be any of hydrogen, methyl, ethyl, n- or i-propyl or n-, s- or t-butyl, but is preferably hydrogen.
- each of Z 1 and Z 2 is preferably a group of the above mentioned formula (I). Moreover, it is even more preferred that each of Z 1 and Z 2 is the same group as the other.
- Z 1 may be a halobenzene nucleus of the formula (I)
- A may be zero and Z 2 a group of the formula
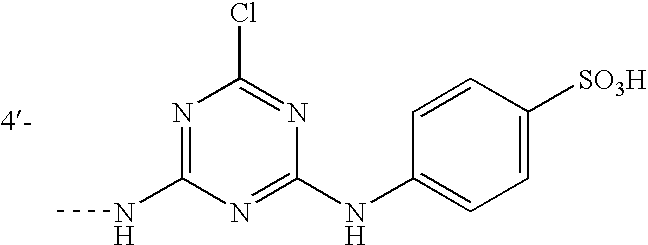
- X 1 is an eliminatable group such as OSO 3 H, OPO 3 H 2 and salts of any of these, and Cl, a preferred group (IV) being
- Z 1 is a halobenzene nucleus of the formula (I) above and Z 2 is a halotriazine nucleus of the formula (II)
- Y 1 1 or each Y 1 independently, is a halogen atom or an optionally substituted pyridinium group
- T is C 1-4 alkoxy, C 1-4 thioalkoxy or N(R 1 ) (R 2 ) in which R 1 is hydrogen, optionally substituted C 1-4 alkyl or optionally substituted aryl; and
- R 2 is hydrogen or optionally substituted C 1-4 alkyl.
- Y 1 is preferably fluorine, chlorine or optionally substituted pyridinium which may be derived from, for example, nicotinic or isonicotinic acid or their carboxamides.
- R 1 and R 2 are preferably hydrogen, but at least one of R 1 and R 2 may be a C 1-4 alkyl group and indeed R 1 may additionally be an optionally substituted aryl, preferably phenyl, group.
- a preferred substituent is hydroxyl, amino, halo, carboxy or sulpho.
- Z 1 is a halobenzene nucleus of the formula (I) and Z 2 is a halopyrimidine nucleus of the formula
- U or each U independently, is C 1-4 alkyl or C 1-4 alkylsulphonyl.
- Y 2 is preferably fluorine or chlorine.
- a preferred range of dyes embodying the invention has the formula (XXX)
- Z 3 is a third reactive group selected from the groups of the formulae (I)-(III), given and defined above;
- each of J 1 and J 2 is an optionally substituted aryl group or a chromophore
- L 3 is a linking group linking Z 3 and D;
- L 4 is a linking group linking Z 3 and J 1 ;
- L 1 is a linking group linking Z 2 and J 2 ;
- each of q, r, s and t independently, is zero or 1;
- each of Z 1 , Z 2 , L 1 , L 2 and a is as defined above;
- Z 2 is selected from the groups of the formulae (I)-(III), given and defined above.
- each of L 3 and L 4 1 independently, is preferably selected from one of the groups of the formulae (VII) 1 , (VII) 2 and (VII) 3 , given and defined above;
- each of L 3 and L 4 is preferably a group of the formulae (VII) 1 , given and defined above.
- B is a triazine group substituted by a non-reactive group.
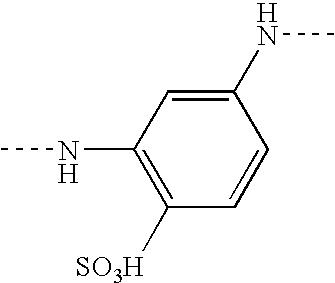
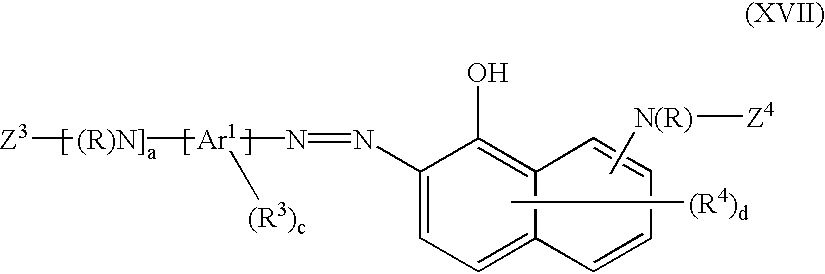
- the chromophore D is preferably an azo chromophore derived from 1-hydroxy-8-aminonaphthalene substituted by at least one sulphonic acid group.
- D 1 is a first chromophore
- D 2 is a second chromophore
- Z 3 when present, is a third reactive group selected from the groups of the formulae (I)-(III), given and defined in claim 1;
- Z 4 when present, is a fourth reactive group selected from the groups of the formulae (I)-(III), given and defined in claim 1;
- L 3 is a linking group linking Z 3 to D 1 ;
- L 4 is a linking group linking Z 3 to Z 4 ;
- L 1 is a linking group linking D 2 to one of Z 3 and Z 4 ;
- each of k and l independently, is zero or 1;
- each of Z 1 , Z 2 , L 1 , L 2 and a is as defined in claim 1.
- a is 1
- Z 2 is a group of the formula (I), given and defined above
- k is zero
- D 1 is a tetrakisazo chromophore containing two residues of H-acid linked together by a group forming part of the chromophore.
- a is 1, each of Z 1 and Z 2 is a group of the formula (I), given and defined above, each of k and l is 1;
- each of D 1 and D 1 is a disazo chromophore containing a respective residue of H-acid
- each of Z 3 and Z 4 is a group of the formula (II), given and defined above, and
- L 4 is a linking group of the formula (VII) 2 or (VII) 3 , given and defined above.
- Z 3 is a third reactive group selected from the groups (I)-(III), given and defined above.
- L 3 is a third linking group selected from the groups (VII) 1 , (VII) 2 and (VII), 3 given and defined above;
- Z 2 is a second reactive group selected from the groups (IV)-(VI), given and defined above;
- each of Z 1 and L 1 is as defined above.
- L 1 is preferably a linking group of the formula (VII) 2 or (VII) 3 , given and defined above and Z 3 is preferably a group of the formula (II), given and defined above.
- Z 2 is selected from groups of the formulae I-(III), given and defined above.
- each of D, Z 1 , L 1 and L 1 is as defined above.
- Z 2 is preferably a group of the formula (II), given and defined above and L 2 is preferably a linking group selected from the groups (VII) 1 , (VII) 2 and (VII) 3 , given and defined above, more preferably a group of the formula (VII) 2 or (VII) 3 , given and defined above; and
- L 1 is a group of the formula (VII) 3 , given and defined above, in which B is an optionally substituted aryl group, or is alternatively a chromophoric bridging group.
- a preferred chromophore D is a disazo dye containing a residue derived from H-acid and having azo groups at the 2- and 7- positions.
- Another preferred chromophore D is a group of the formula
- each of D 1 and D 2 is a chromophore
- L 3 is a linking group selected from groups of the formula (I)-(III), given and defined above;
- each of Z 1 , Z 2 1 L 1 and L 2 is as defined above;
- each of D 1 and D 2 1 independently, is a chromophore
- L 3 is a linking group selected from the groups of the formulae (I)-(III), given and defined above;
- each of Z 1 , Z 2 , L 1 and L 2 is as defined above;
- one of Z 3 and Z 4 is a reactive group Z 2 ;
- each of Z 3 and Z 4 is a reactive group selected from the formulae (IV)-(VI), given and defined above;
- each of t and u independently, is zero or 1 and at least one of t and u is 1;
- D is a chromophore
- Ar is an optionally substituted aryl group
- L 1 is a group of the formula
- each R independently, is as defined in claim;
- Z 1 is as defined above.
- D is preferably a disazo dye containing a residue derived from H-acid.
- the or a chromophore D may contain a heterocyclic nitrogen atom, in which case the linking group may have the formula
- each of B,R and b is as defined above and the bond ⁇ circle over (1) ⁇ is linked to the heterocyclic nitrogen atom of the chromophore.
- Such dyes may have the formula
- D N is a chromophore containing a heterocyclic group including a nitrogen atom
- L 1 is a group of the formula (VII) 4 or (VII) 5 , given and defined above, directly attached via the bond ⁇ circle over (1) ⁇ , to the nitrogen atom of the said chromophore D N ;
- Ar is an optionally substituted aryl group
- each of Z 1 , Z 2 , L 2 , L 5 , a and t is as defined above.
- a typical chromophore D containing a heterocyclic nitrogen atom has the formula
- the or each chromophoric group independently preferably comprises an azo (which may be a monoazo, polyazo or metal complex azo), anthraquinone, hydrazone, phthalocyanine, triphenodioxazine or formazan group.
- azo which may be a monoazo, polyazo or metal complex azo
- anthraquinone hydrazone
- phthalocyanine triphenodioxazine or formazan group.
- Examples of chromophoric groups which may be present are those given as types (a)-(g) of the group “D 1 ” in formula (I) of U.S. Pat. No. 5,484,899, which is incorporated herein by reference.
- Preferred azo groups are monoazo and disazo groups.
- Preferred monoazo groups have the formula
- Ar 1 is an aryl or heteroaryl group and Ar 2 is an aryl group.
- each aryl group independently is a mono- or di-cyclic aryl group.
- Preferred aryl groups are optionally substituted phenyl and optionally substituted naphthyl.
- Preferred heteroaryl groups are pyridonyl and pyrazolonyl.
- a first preferred monoazo group is of the Formula (IX) (or salt thereof):
- Ar 1 is an aryl group, preferably a benzene or naphthalene nucleus
- R 3 is C 1-4 alkyl, nitro, halo or sulphonic acid or a salt thereof;
- c is zero or 1 to 4;
- R 4 is a sulphonic acid or a salt thereof.
- d is 1 or 2;
- Ar 1 is preferably optionally substituted phenyl or naphthyl, especially a phenyl or naphthyl group having at least one sulpho substituent.
- substituents which may be present on Ar include a halogen atom, especially chlorine; an alkyl radical, especially C 1-4 alkyl, more especially methyl; an acylamido radical, especially acetylamino, benzamido or sulphonated benzamido; amino; hydroxy; and an alkoxy radical, especially C 1-4 alkoxy, more especially methoxy.
- phenyl groups having at least one sulpho substituent there may be mentioned 2-, 3- or 4-sulphophenyl; 2-sulpho-4-nitrophenyl; 2-sulpho-5-nitrophenyl; 4-sulpho-2-methylphenyl; 5-sulpho-2-methylphenyl; 2-sulpho-4-methylphenyl; 5-sulpho-2-methoxyphenyl; 2-sulpho-4-methoxyphenyl; 4-sulpho-2-chlorophenyl; 5-acetamido-2-sulphophenyl; 5-sulpho-2-carboxyphenyl; 2,4-disulphophenyl; 2,5-disulphophenyl; and 3,5-disulphophenyl.
- naphthyl groups having at least one sulpho substituent there may be mentioned 1-sulphonaphth-2-yl; 1,5-disulphonaphth-2-yl; 1,5,7-trisulphonaphth-2-yl; 3,6,8-trisulphonaphth-2-yl; 5,7-disulphonaphth-2-yl; 6-sulphonaphth-2-yl; 4-, 5-, 6-, or 7-sulphonaphth-1-yl; 4,8-disulphonaphth-1-yl; 3,8-disulphonaphth-1-yl; 2,5,7-trisulphonaphth-1-yl; and 3,5,7-trisulphonaphth-1-yl.
- Preferred optional substituents which may be present on the naphthalene nucleus of Formula (IX) are those mentioned above for Ar 1 .
- Groups of the Formula (IX) are preferably linked to a group L 1 or L 2 at the 6-, 7- or 8-position, especially the 6- or 8-position.
- L 1 or L 2 is to be linked at the 8-position, it is preferred that R 5 is a sulpho group at the 5- or 6- position.
- each R independently and a is as defined above;
- one of Z 3 and Z 4 is a group Z 1 and the other is a group Z 2 ;
- the group Z 4 is selected from the groups of the formulae (I)-(III), given and defined above.
- Ar 1 is a benzene or naphthalene nucleus
- R 3 is C 1-4 alkyl, nitro, halo or sulphonic acid or salt thereof;
- c is zero or 1-4;
- R 4 is a sulphonic acid or a salt thereof
- d is 1 or 2.
- a preferred disazo group is of the Formula (XI) (or salt thereof):
- M and E are each independently optionally substituted phenylene or naphthalene
- Ar 1 is as defined above.
- E is optionally substituted naphthalene and M is optionally substituted phenylene.
- the optional substituents which may be present on M or E are preferably independently selected from halo, especially chloro; alkoxy, especially C 1-4 alkoxy; alkyl, especially methyl; sulpho; carboxy; hydroxy; amino; acylamino, especially acetamido, benzamido and sulphonated benzamido, and pyrimidinylamino or triazinylamino cellulose-reactive groups.
- Examples of groups represented by M and E there may be mentioned phenylene, 2-methyl-1,4-phenylene, sulphophenylene, ureidophenylene, 7-sulpho-1,4-naphthalene, 6-sulpho-1,4-naphthalene; 8-sulpho-1,4-naphthalene and 6-hydroxy-4-sulpho-1,5-naphthalene.
- one of Z 5 and Z 6 is a group Z 1 and the other is a group Z 2 ;
- each of f and g independently is zero or 1;
- each of c and e independently, is zero or 1-4;
- d is 1 or 2;
- each R independently is as defined above;
- each of Ar 1 and Ar 2 independently is an optionally substituted aryl group
- each of R 3 and R 4 is as defined above.
- each of Z 5 and Z 6 may be the same group
- one of Z 5 and Z 6 may be a group of the formula (I), given and defined above, and the other of Z 5 and Z 6 may be selected from groups of the formulae (II) and (III), given and defined above.
- one of Z 5 and Z 6 is a group of the formula (I), given and defined above, and the other of Z 5 and Z 6 is a group of the formula (II).
- Another especially preferred range of disazo dyes has the formula
- B is a hydrocarbon bridging group as defined above, and preferably an optionally substituted aryl group;
- one of G 1 and G 2 is OH and the other is NH 2 ;
- each of X, Y, Y 1 , Ar 1 Ar 2 , R 3 , R 4 , R 5 , c, d and e is as defined above.
- one of Z 5 and Z 6 may be a group
- X, Y and n are as defined in claim 1 and the other of Z 5 and Z 6 is the group —SO 2 CH 2 CH 2 OSO 3 H or —SO 2 CH ⁇ CH 2 .
- G 3 is C 2 H 4 OSO 3 H or a salt thereof or —CH ⁇ CH 2 ;
- G 1 and G 2 are as defined above;
- R 4 and d are as defined above;
- each of h and i is zero, 1 or 2.
- a preferred anthraquinone group is of the Formula (XII) (or a salt thereof).
- anthraquinone nucleus optionally contains a sulphonic acid group in the 5-, 6-, 7-, or 8-position and V is a divalent organic linking group, preferably of the benzene series.
- V is a bridging group B, preferably phenylene, diphenylene, or 4,4′-divalent stilbene or azobenzene radicals which are optionally sulphonated. It is preferred that V contains one sulphonic acid group for each benzene ring present therein.
- a preferred anthraquinone dye of has the formula
- D A is an anthraquinone chromophore
- L 3 is a linking of the formula (VII) 1 , given and defined above;
- Ar is an optionally substituted aryl group
- each of Z 1 , Z 2 , L 1 and L 2 is as defined above.
- each of L 1 and L 2 is a linking group of the formula (VII) 1 , (VII) 2 or (VII) 3 , given and defined above; and still more preferably, each of L 1 and L 2 1 independently is a group of the formula (VII) 3 , given and defined above, in which B is an optionally substituted aryl group.
- a preferred phthalocyanine group is of the Formula (XIII) (or a salt thereof).
- Pc is a metallo-phthalocyanine nucleus, preferably copper or nickel phthalocyanine; L is as hereinbefore defined; each W independently is a hydroxy or a substituted or unsubstituted amino group, V 1 is a divalent organic linking group, preferably a C 1-4 -alkylene or phenylene linking group; and a and b are each independently 1, 2 or 3 provided that a+b is not greater than 4.
- such a metal phthalocyanine dye has the formula
- Cu Pc is a copper phthalocyanine chromophore
- each of R 21 and R 22 is hydrogen or optionally substituted C 1-4 alkyl
- B is a hydrocarbon bridging group
- Z 1 is as defined above.
- chromophore D is a triphenodioxazine
- a preferred triphenodioxazine group is of the Formula (XIV) (or a salt thereof).
- each Y 3 independently is a covalent bond, C 2-4 -alkylene, phenylene or sulphophenylene;
- U 1 is H or SO 3 H
- T 1 and T 2 are halo, especially chloro, C 1-4 -alkyl, or C 1-4 alkoxy.
- Each Y 3 is preferably —CH 2 H 4 — or —C 3 H 6 —, U 1 is preferably SO 3 H and T 1 and T 2 are preferably Cl, methyl or ethyl.
- triphendioxazine dye has the formula
- D T is a triphendioxazine chromophore
- each of L 2 , L 3 and L 4 is a linking group of the formula (VII) 1 1 (VII) 2 or (VII) 3 given and defined above;
- each of Z 2 , Z 3 and Z 4 is a reactive group selected from groups of the formulae (I)-(III), given and defined above;
- each of q and r is zero or 1;
- each of Z 1 and L 1 is as defined above.
- Z 2 is preferably a group of the formula (I), given and defined above and each of Z 3 and Z 4 is a group of the formula (II), given and defined above; and each of L 1 , L 2 , L 3 and L 4 is preferably a group of the formula (VII) 3 , given and defined above.
- chromophore D is a formazan
- a preferred group is of the Formula (XV) (or a salt thereof).
- X 1 is H, SO 3 H or Cl
- each r independently has a value of 0, 1 or 2.
- the formazan group has at least one, and preferably at least two, sulpho groups.
- each r has a value of 1.
- Such a formazan dye has the formula
- D F is a formazan chromophore
- one of Z A and Z B is a group Z 2 , given and defined above;
- each of L 3 and L 4 is a linking group of the formula (VII) 1 , (VII) 2 or (VII) 3 , given and defined above;
- each of Z 1 and L 1 is as defined above;
- a is zero or 1;
- Z A is selected from groups of the formulae (IV)-(VI), given and defined above;
- Z 8 is selected from groups of the formulae (I)-(III),given and defined above.
- the invention provides a process for preparing a dye of the formula (VIII) 1
- D is a chromophore
- each R independently, is hydrogen or C 1-4 -alkyl
- each of Z 1 and Z 2 is a group
- n 1 or 2
- X or each X independently, is an electron withdrawing group
- Y is a halogen atom
- each of D and R is as defined above, with at least two moles, per mole of the chromophoric compound of the formula (XX), of a dihalobenzene component comprising at least one dihalobenzene compound of the formula (XXI)
- the chromophoric compound of the formula (XX) can be prepared by protecting one amino group of a phenylene diamine sulphonic acid and then diazotising this and coupling a first portion of the diazotised phenylene diamine sulphonic acid to the naphthalene nucleus under acid conditions so as to couple on to the benzene ring of the naphthalene nucleus containing an amino group and then taking a second portion of the same diazotised and protected phenylene diamine sulphonic acid compound referred to above (or a different such compound) and coupling this under neutral or alkaline conditions to the benzene ring of the naphthalene nucleus containing a hydroxyl group to obtain a diamine dyestuff having respective protected amino groups.
- the protective group can then be removed by hydrolysis.
- the invention provides a process for preparing a dye of the formula (VIII) 1
- D is a chromophore
- each of L 1 and L 2 independently, is N(R);
- each R independently, is hydrogen or C 1-4 alkyl
- Z 1 is a group
- n 1 or 2
- X or each X independently, is an electron withdrawing group
- Y is a halogen atom
- Z 2 is selected from the groups (II) and (III) defined above;
- each of D and R is as defined above, with one mole, per mole of the chromophoric compound of the formula (XX), of each of
- m is 1 or 2; p is 0 or 1; when m is 1, p is 1; and when m is 2, p is 0;
- Y 1 is a halogen atom or an optionally substituted pyridinium group
- T is C 1-4 alkoxy, C 1-4 thioalkoxy or N(R 1 ) (R 2 ), in which each of R 1 and R 2 independently is hydrogen, optionally substituted C 1-4 alkyl or optionally substituted aryl;
- Y 2 is a halogen atom or an optionally substituted pyridinium group
- U or each U independently, is C 1-4 alkyl or C 1-4 alkylsulphonyl
- Y is as defined above;
- the invention provides a process for preparing a dye of the formula (VIII) 3
- D is a chromophore
- L 1 is N(R), in which R is hydrogen or C 1-4 alkyl;
- Z 1 is a group
- n 1 or 2; X, or each X independently, is an electron withdrawing group; and Y is a hydrogen atom; and Z 2 is a group of the formula (IV)
- X 1 is an eliminatable group
- z is zero or 1
- R 10 is hydrogen, C 1-4 alkyl or halogen
- W is —OC( ⁇ O)— or —N(R 11 )C( ⁇ O)—
- R 11 is hydrogen or C 1-4 alkyl
- dye formulae have been shown in the form of their free acid in this specification, the invention also includes dyes and processes using dyes in the salt form, particularly their salts with alkali metals such as the potassium, sodium, lithium or mixed sodium/lithium salt and their salts with tetraalkylammonium ions.
- the dyes may be used for dyeing, printing or ink-jet printing, for example, of textile materials and paper.
- the process for colouration is preferably performed at a pH of 7.1 to 13, more preferably 10 to 12. pH levels above 7 can be achieved by performing the process for colouration in the presence of an acid-binding agent.
- the substrate may be any of a textile material, leather, paper, hair or film, but is preferably a natural or artificial textile material containing amino or hydroxyl groups, for example textile material such as wool, silk, polyamides and modified polyacrylonitrile fibres, and more preferably a cellulosic material, especially cotton, viscose and regenerated cellulose, for example, that commercially available as Tencel.
- the dyes can be applied to the textile materials at a pH above 7 by, for example, exhaust dyeing, padding or printing, including ink-jet printing. Textile materials are coloured bright shades and possess good fastness to light and wet treatments such as washing.
- the new dyes are particularly valuable for colouring cellulosic textile materials.
- the dyes are preferably applied to the cellulosic textile material at a pH above 7 in conjunction with a treatment with an acid binding agent.
- Preferred acid-binding agents include alkali metal carbonates, bicarbonates, hydroxides, metasilicates and mixtures thereof, for example sodium bicarbonate, sodium carbonate, sodium metasilicate, sodium hydroxide and the corresponding potassium salts.
- the dyes benefit from the excellent build-up and high fixation.
- exhaust dyeing can be carried out at a relatively low temperature of about 50-70° C., especially about 60° C.
- the new dyes can be applied to textile material containing amine groups, such as wool and polyamide textile materials, from a neutral to mildly alkaline dyebath.
- the dyeing process can be carried out at a constant or substantially constant pH, that is to say that the pH of the dyebath remains constant or substantially constant during the dyeing process, or if desired the pH of the dyebath can be altered at any stage of the dyeing process.
- the dyes may be in a liquid form, or solid form, for example in granular or powdered form.
- Dyes embodying the invention are especially useful for application to substrates by ink-jet technologies.
- Substrates which are particularly useful are cellulosic textiles and paper.
- the dye used in the ink is preferably purified by removal of substantially all the inorganic salts and by-products which are generally present in a commercial dye at the end of its synthesis. Such purification assists in the preparation of a low viscosity aqueous solution suitable for use in an ink jet printer.
- the dye should preferably have a water-solubility of at least 5%, and more preferably from 5% to 25%, by weight. Solubility of the dye can be enhanced by converting the sodium salt, in which form it is normally synthesised, either partially or wholly, into the lithium or ammonium salt. Purification and ion exchange can conveniently be effected by use of membrane separation processes which permit the separation of unwanted inorganic materials and by-products directly from an aqueous solution or dispersion of the dye followed by partial or complete exchange of the counter-ion.
- the ink preferably contains up to 20% by weight of dye and more preferably from 2% to 10%, especially from 3% to 8%.
- the ink may also contain a humectant, which may also function as a water miscible solvent, which preferably comprises a glycol or dihydroxyglycolether, or mixture thereof, in which one or both hydroxy groups are secondary hydroxy groups, such as propane-1,2-diol,butane-1,3-diol and 3-(3-hydroxy-prop-2-oxy)propan-2-ol.
- a humectant which may also function as a water miscible solvent, which preferably comprises a glycol or dihydroxyglycolether, or mixture thereof, in which one or both hydroxy groups are secondary hydroxy groups, such as propane-1,2-diol,butane-1,3-diol and 3-(3-hydroxy-prop-2-oxy)propan-2-ol.
- the humectant has a primary hydroxy group this is prefereably attached to a carbon atom adjacent to a carbon atom carrying a secondary or tertiary hydroxy group.
- the humectant may comprise up to a total of 10% by weight of a polyol, especially a glycol or dihydroxyglycolether, having two or more primary hydroxy groups, such as ethyleneglycol, propane-1,3-diol,butane-1,4-diol, 2-(2-hydroxyethoxy)ethanol and 2-(2-[2-hydroxyethoxy)ethanol and/or an alcohol with a primary hydroxy group, such as ethanol,n-propanol and n-butanol.
- the ink preferably contains not more than 5% by weight, and is more preferably free from, such compounds.
- the term “alcohol” means a compound having only one hydroxy group attached to an aliphatic carbon atom.
- the ink preferably contains from 5% to 25%, by weight, more especially from 10% to 20%, of humectant.
- the ink may be buffered to a pH from 5 to 8, especially to a pH from 6 to 7, with a buffer such as the sodium salt of metanilic acid or an alkali metal phosphate, or di- or triethanolamine.
- a buffer such as the sodium salt of metanilic acid or an alkali metal phosphate, or di- or triethanolamine.
- the ink preferably also contains one or more preservatives to inhibit the growth of fungi, bacteria and/or algae because these can block the jet of the ink jet printing equipment.
- the solution preferably contains a conducting material such as an ionised salt to enhance the accumulation of charge on the drop.
- Suitable salts for this purpose are alkali metal salts of mineral acids.
- the remainder of the ink is preferably water, especially de-ionised water to avoid the introduction of impurities into the ink.
- H-Acid (0.475M, 183 g) was dissolved in water (800 ml) at pH 7.5. This solution was added dropwise to the above diazo suspension with good agitation over 1 hr at 0-2° C. The mixture was then stirred at 0-2° C. for a further 2 hrs at pH 2-3, and allowed to warm to room temperature over 18 hrs.
- This Example describes how a dye may be synthesised where the fibre-reactive halobenzene group is attached via a linking diamine to a second fibre-reactive group, and thereby to a chromophore.
- N-dichlorotriazinyl derivative of the azo dye resulting from azo-coupling 7-aminonaphthalene-1,3,6-trisulphonic acid with m-ureidoaniline was prepared by conventional means. A solution of this dye (35 mmol in 450 ml) was added at room temperature with stirring to N-(4-aminophenyl)piperazine (7.5 g, 42 mmol) dissolved in 50/50 acetone/water (400 ml) maintained at pH 6-6.5 by addition of sodium carbonate solution. After completion of the reaction the solution was concentrated, and the product was precipitated by addition of methylated spirit.
- dyes of a similar nature may be prepared as further exemplified by dyes 69-77 listed in Table 3.
- Disazo naphthalene dyes containing two reactive groups attached to the chromophore as described in Example 68 are exemplified by Examples 78 to 97 listed in Table 4.
- Each of the dyes (0.2 parts) of respective Examples 1, 3 and 5 was dissolved in respective amounts of water (50 parts) at 25° C. and pH 9.
- Cotton fabric (5 parts) and Glaubers salt (2.5 parts) were added.
- the dyes were fixed to the cotton by raising the temperature to between 50° C.-60° C., basifying to pH 11.0 and maintaining this for 1 hr.
- the cotton was removed and washed in soapy water to give a dark greenish-navy cotton having high general fastness properties.
- Each of the dyes (0.2 parts) of respective Examples 2 and 4 was dissolved in water (50 parts) at 25° C. and pH 9.
- Cotton fabric (5 parts) and salt (4 parts) were added.
- the dyes were fixed to cotton by raising the temperature to 80-100° C. and maintaining this for 1 hr.
- the cotton was removed and washed in soapy water to give a deep greenish-navy cotton having good fastness properties.
- Methodology for applying dyes embodying the present invention to cotton may be further exemplified by means of the following pad-batch dyeing protocol.
- Example 195.-Dye from Example 5 (0.5 parts) was dissolved in water (30 parts) at 25° C. and the following agents were added: Primasol NF (1 part of 20% solution) and sodium silicate Q70 (9.5 parts of 50% solution), sodium hydroxide (5.1 parts of 10% solution). The solution was made up to 50 parts by addition of water, and then padded onto woven cotton fabric (70% pick-up). The cloth was wrapped in cling film and batched at room temperature for 24 hours. The cling film was removed and the dyed cloth was rinsed successively with cold water and hot water, then washed with a soap solution, rinsed with water, and dried to give cotton coloured a dull greenish blue shade.
- Example 193 If the amount of dye used in Example 193 is doubled and the procedure repeated, a greenish-navy shade is obtained.
- Example 193 If the amount of dye used in Example 193 is trebled and the procedure repeated, a dark navy shade is obtained.
- Dyes embodying the present Invention may be applied to textile substrates, especially cotton, by conventional printing technology, as the following exemplifies.
- a dye from Example 5 (30 parts) was dissolved in a solution containing Manutex F 700 (500 parts of a 10% solution) and Vitexol D (20 parts). The solution was made up to 1000 parts by the addition of water and printed by means of a Zimmer screen printer. The printed cloth was dried and padded through a solution containing sodium silicate (48 degBe, 700 parts) made up to 1000 parts by the addition of water (80% pickup) . Immediately after padding, the printed cotton was steamed in a Roaches Flash-ager steam chamber at 120 deg C. for 45 seconds. The printed cloth was rinsed in cold water, washed with a soap solution at the boil, rinsed again in cold water and dried to give a dull greenish navy print on the cotton.
- Dyes embodying the present invention may be used in the preparation of inks specially formulated for application by ink-jet technology.
- the dye from Example 5 (6 parts) was dissolved in a solution of propylene glycol (15 parts) and water (79 parts) .
- this solution was applied to cotton which had previously been pretreated (for example, with a pretreatment agent described in EP-A-0534660) by means of commercial ink-jet printing equipment, deep navy shades were obtained.
- Reactive dyes including those of Examples 1-189, are usually isolated as their sodium salts, and are contaminated with inorganic impurities resulting from the method of preparation. Dyes free of impurities suitable for ink jet printing, and/or with increased solubility, may be prepared by conventional ion exchange techniques, where for example sodium is replaced by lithium and inorganic impurities are simultaneously removed.
- Dye from Example 1 (10 parts) was dissolved in water (100 parts) and treated on a reverse osmosis rig until the permeate conductivity was 10 micro reciprocal ohms. A solution of lithium chloride was added to the dye solution and treatment on the r.o. rig was continued until the permeate conductivity had decreased to 1 micro reciprocal ohm. The sample was concentrated to a volume of about 80 parts, after which the solution was buffered and other formulating agents were added. The dye solution was then diluted to 90 parts by addition of water, at which stage it was suitable for storage.
- Dye solution prepared in this way could be diluted and applied to cotton by the methods described in any of the Examples 190, 195-198, or formulated into an ink by addition of suitable humectants and/or cosolvents, and applied by ink-jet methodology to cotton, for example by the method described in Example 200. In all these cases, dull blue, greenish navy or black shades were imparted to the substrate, depending on the amount of dye applied.
- Dye from Example 1 was ion exchanged to the lithium form as in Example 201, and formulated into an ink with the composition: dye (5 parts), propylene glycol (12 parts), diethanolamine (sufficient to buffer the final pH to 7-8.5), and water (to bring the total to 100 parts).
- the ink was added to the ink reservoir of an ink jet printer (e.g. HP Desk Jet 500) and printed onto paper (Logic 300), to give a black print of generally good fastness properties.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Plural Heterocyclic Compounds (AREA)
- Coloring (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Inks, Pencil-Leads, Or Crayons (AREA)
- Pyridine Compounds (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Ink Jet (AREA)
- Ink Jet Recording Methods And Recording Media Thereof (AREA)
- Cosmetics (AREA)
Abstract
The invention relates to reactive dyes containing a halobenzene nucleus and, in particular, reactive dyes containing a halobenzene nucleus and two or more reactive components.
Description
- This invention relates to reactive dyes containing a halobenzene nucleus and, in particular, reactive dyes of this type containing two or more reactive components.
- Dyes are known which contain a halobenzene nucleus linked via an azo group to another aromatic nucleus such that the halobenzene nucleus forms part of the chromophoric chain (see for example GB-A-882001). Dyes of this type which contain two such halobenzene nuclei are disclosed in CA64,14316d (1966), which is an English language abstract of an article by Matsui et al, Yuki Gosei Kagaku Kyokai Shi(1966), 24-(2), 132-136.
- Dyes are also known in which the halobenzene nucleus is attached to a chromophoric group by a sulphonamide or amide linkage; see, for example, GB-A-978162 and CA59,12949g (1963), which is an English language abstract of an article by Matsui et al in Yuki Gosei Kagaku Kyokai Shi (1962), 20,1100-1112. Again dyes of this type may contain two such halobenzene nuclei; see GB-A-978162.
- In other known dyes the halobenzene nucleus is linked to a chromophoric group by an amine linkage; see U.S. Pat. No. 3,301,847 and CA61,16193f (1964) which is an English language abstract of an article by Matsui et al in Kogyo Kagaku Zasshi, (1964), 67(1), 94-97. However, such dyes contain only one reactive group.
- We have now found surprisingly that dyes having at least one halobenzene nucleus linked to a chromophoric group via an amino linkage and additionally containing a second reactive group have particularly high build up, especially in warm dyeing applications.
- According to the invention there is provided a dye containing
- at least one chromophore D;
-
- in which:
- n is 1 or 2
- X, or each X independently, is an electron withdrawing group; and
- Y is a halogen atom;
- at least a second reactive group Z 2 selected from
- (1) a group of the formula (I), given and defined above, but selected independently thereof;
-
- wherein
- m is 1 or 2; p is 0 or 1; when m is 1, p is 1; and when m is 2, p is 0;
- Y 1 or each Y1 independently, is a halogen atom or an optionally substituted pyridinium group; and
- T is C 1-4alkoxy, C1-4thioalkoxy or N(R1) (R2), in which each of R1 and R2 independently is hydrogen, optionally substituted C1-4alkyl or optionally substituted aryl;
-
- wherein:
- x is 1, 2 or 3; y is zero, 1 or 2; and
- x+y≦3;
- Y 2, or each Y2 independently, is a halogen atom or an optionally substituted pyridinium group; and
- U or each U independently, is C 1-4alkyl or C1-4alkylsulphonyl;
- (4) a group of the formula (IV)
- —SO2CH2CH2X1 (IV)
- wherein
- X 1 is an eliminatable group;
- (5) a group of the formula (V)
- —SO2(CH2)zCH═CH2 (V)
- wherein
- z is zero or 1; and
- (6) a group of formula (VI)
- —W—C (R10)═CH2 (VI)
- wherein:
- R 10 is hydrogen, C1-4alkyl or halogen; and
- W is —OC(═O)— or —N(R 11)C(═O)— in which R11 is hydrogen or C1-4 alkyl;
- at least a first linking group L 1, linking the said first, halobenzene, reactive group Z1 to one of components (i) the or a chromophore D and (ii) the second reactive group Z2, which said first linking group L1 presents an amino nitrogen to the reactive group Z1 and to the component (i) or (ii) or, when component (i) contains a heterocyclic nitrogen atom, is linked directly to the nitrogen atom and which said first linking group L1 optionally includes a hydrocarbon bridging group, which hydrocarbon bridging group B has at least two carbon atoms, is optionally substituted, optionally includes at least one hetero atom and is optionally a chromophore; and
- when Z 2 is selected from the said groups (I)-(III), at least a second linking group L2 linking the second reactive group Z2 to one of (i) the or a chromophore D and (ii) the said first reactive group Z1, which said linking group L2 is selected from
- (1) a linking group L 1, but selected independently thereof; or
- (2) an amide linkage; and
- ( 3) a sulphonamide linkage; and
- optionally at least one aromatic group Ar which, when Z 2 is selected from the said groups (IV)-(VI), may carry the said reactive group Z2.
- Preferably, the linking group L 1 has the formula (VII)1
- N(R) (VII)1
- wherein R is hydrogen or optionally substituted C 1-4alkyl, such that the same amino group presents itself to each of the reactive group Z1 and the component (i) or (ii), as defined above; or
-
- wherein each R, independently, is as defined above, such that respective amino nitrogens, one of the piperazine group and the other of the alkylamino group, present themselves respectively, to the reactive group Z 1 and to the component (i) or (ii), as defined above; or
- has the formula (VII) 3
- —N(R)BN(R)— (VII)3
- wherein B is a hydrocarbon bridging group as defined above, each R, independently , is as defined above and B is optionally linked additionally to at least one additional group —N(R).
- The hydrocarbon bridging group B may be a straight or branched, optionally substituted, C 2-6alkylene group optionally interrupted by at least one hetero atom, for example, O,S or N. Optional substituents are OH alkoxy, carboxy, carboxylic ester or carboxamide. Alternatively the bonding group B may be an optionally substituted arylene especially phenylene group. Optional substituents are SO3H and salts thereof, C1-4alkyl, C1-4alkoxy and chloro. The bridging group B is is especially preferably an optionally substituted aryl group.
- An especially preferred dye embodying the invention has the formula (VIII)
- Z1-L1-D-(L2)aZ2 (VIII)
- wherein:
- D is a chromophore;
- each of L 1 and L2 is an amine or piperazine linkage of the formula
- —N(R)— (VII)1;
-
- —N(R)BN(R)— (VII)3
- wherein:
- R, or each R independently, is hydrogen or C 1-4 alkyl;
- B is a hydrocarbon bridging group which has at least two carbon atoms, is optionally substituted, optionally includes at least one hetero atom and is optionally a chromophore;
- a is zero or 1; and
- b is from 2 to 6 inclusive;
-
- in which:
- n is 1 or 2;
- X, or each X independently, is an electron withdrawing group; and
- Y is a halogen atom; and
- when a is 1, Z 2 is:
- a group of the formula (1), given and defined above but selected independently thereof; or
-
- wherein:
- m is 1 or 2; p is 0 or 1; when m is 1, p is 1; and when m is 2, p is 0;
- Y 1, or each Y1 independently, is a halogen atom or an optionally substituted pyridinium group; and
- T is C 1-4 alkoxy, thioalkoxy or N(R1) (R2) in which R1 is hydrogen, optionally substituted C1-4 alkyl or optionally substituted aryl and
- R 2 is hydrogen or optionally substituted C1-4 alkyl; or
-
- wherein:
- x is 1, 2 or 3; y is zero, 1 or 2; and x+y≦3;
- Y 2, or each Y2 independently, is a halogen atom; and
- U, or each U independently, is C 1-4 alkyl or C1-4 alkylsulphonyl; and
- when a is zero, Z 2 is:
- —SO2CH2CH2X1 (IV)
- in which X 1 is an eliminatable group; or
- —SO2(CH2)zCH═CH2 (V)
- wherein z is zero or 1; or
- a group of formula (VI)
- —W—C(R10)═CH2 (VI)
- wherein:
- R 10 is hydrogen, C1-4alkyl or halogen; and
- W is —OC(═O)— or —N(R 11)C(═O)— in which R11 is hydrogen or C1-4 alkyl.
- In the halobenzene nucleus of the formula (I), X or each X independently, may be selected from nitro, cyano, alkylsulphonyl, dialkylaminosulphonyl and sulphonic acid groups and salts thereof. Preferably, X or each X independently, is selected from nitro and cyano.
- The halogen atom in the halobenzene nucleus of formula (I) is preferably fluorine or chlorine.
- The group R in the linking group L 1 of formulae (1)-(3) may be any of hydrogen, methyl, ethyl, n- or i-propyl or n-, s- or t-butyl, but is preferably hydrogen.
- In the dye of formula (VII), each of Z 1 and Z2, independently, is preferably a group of the above mentioned formula (I). Moreover, it is even more preferred that each of Z1 and Z2 is the same group as the other.
- However, alternatively, Z 1 may be a halobenzene nucleus of the formula (I), A may be zero and Z2 a group of the formula
- —SO2CH2CH2X1 (IV)
- in which X 1 is an eliminatable group such as OSO3H, OPO3H2 and salts of any of these, and Cl, a preferred group (IV) being
- —SO2CH2CH2OSO3H (or a salt thereof)
- or
- —SO2(CH2)zCH═CH2 (V)
- wherein z is zero or 1.
-
- wherein m is 1 or 2, p is 0 or 1,
- when m is 1, p is 1 and
- when m is 2, p is 0;
- Y 1 1 or each Y1 independently, is a halogen atom or an optionally substituted pyridinium group; and
- T is C 1-4 alkoxy, C1-4thioalkoxy or N(R1) (R2) in which R1 is hydrogen, optionally substituted C1-4 alkyl or optionally substituted aryl; and
- R 2 is hydrogen or optionally substituted C1-4alkyl.
- In the above formula (II), Y 1 is preferably fluorine, chlorine or optionally substituted pyridinium which may be derived from, for example, nicotinic or isonicotinic acid or their carboxamides.
- Each of R 1 and R2 is preferably hydrogen, but at least one of R1 and R2 may be a C1-4 alkyl group and indeed R1 may additionally be an optionally substituted aryl, preferably phenyl, group.
- When such a C 1-4 alkyl or aryl group is substituted, a preferred substituent is hydroxyl, amino, halo, carboxy or sulpho.
-
- wherein
- x is 1, 2 or 3; y is 0, 1 or 2; and x+y≦3;
- Y 2 1 or each Y2 independently, is halogen atom; and
- U, or each U independently, is C 1-4alkyl or C1-4alkylsulphonyl.
- In the above formula (III) Y 2 is preferably fluorine or chlorine.
- A preferred range of dyes embodying the invention has the formula (XXX)
- Z1-L1-D[L3]q-[Z3-L4 r[J1]s[L2]aZ2[L5-J2]t (XXX)
- wherein:
- Z 3 is a third reactive group selected from the groups of the formulae (I)-(III), given and defined above;
- each of J 1 and J2, independently, is an optionally substituted aryl group or a chromophore;
- L 3 is a linking group linking Z3 and D;
- L 4 is a linking group linking Z3 and J1;
- L 1 is a linking group linking Z2 and J2;
- each of q, r, s and t independently, is zero or 1;
- each of Z 1, Z2, L1, L2 and a is as defined above; and
- when at least one of a and t is 1, Z 2 is selected from the groups of the formulae (I)-(III), given and defined above.
- In one such range of dyes, q is 1, r is 1, s is 1, each of a and t is zero and Z 2 is selected from the groups of the formulae (IV)-(VI), given and defined above.
- In another such range, q is 1, r is 1, s is zero, a is zero and t is 1.
- In a dye of the formula (XXX), each of L 3 and L4 1 independently, is preferably selected from one of the groups of the formulae (VII)1, (VII)2 and (VII)3, given and defined above;
- each of L 3 and L4 is preferably a group of the formulae (VII)1, given and defined above.
- In one preferred range of dyes of the formula (XXX), q is 1, r is zero, s is 1, a is zero and t is zero and in such a range L 3 is preferably a group of the formula (VII)3
- —N(R)BN(R)— (VII)3
- wherein B is a triazine group substituted by a non-reactive group.
- In a dye of the formula (XXX), the chromophore D is preferably an azo chromophore derived from 1-hydroxy-8-aminonaphthalene substituted by at least one sulphonic acid group.
- Yet another range of dyes embodying the invention has the formula
- Z1-L1-D1[L3-Z3(L4-Z4)lL5-D2]k[L2]aZ2
- wherein:
- D 1 is a first chromophore;
- D 2 is a second chromophore;
- Z 3, when present, is a third reactive group selected from the groups of the formulae (I)-(III), given and defined in claim 1;
- Z 4, when present, is a fourth reactive group selected from the groups of the formulae (I)-(III), given and defined in claim 1;
- L 3 is a linking group linking Z3 to D1;
- L 4 is a linking group linking Z3 to Z4;
- L 1 is a linking group linking D2 to one of Z3 and Z4;
- each of k and l, independently, is zero or 1; and
- each of Z 1, Z2, L1, L2 and a is as defined in claim 1.
- In one range of such dye of formula (XXXI), a is 1, Z 2 is a group of the formula (I), given and defined above, k is zero and D1 is a tetrakisazo chromophore containing two residues of H-acid linked together by a group forming part of the chromophore.
- In another such range, a is 1, each of Z 1 and Z2 is a group of the formula (I), given and defined above, each of k and l is 1;
- each of D 1 and D1 is a disazo chromophore containing a respective residue of H-acid,
- each of Z 3 and Z4 is a group of the formula (II), given and defined above, and
- L 4 is a linking group of the formula (VII)2 or (VII)3, given and defined above.
- In still further such ranges
- (i) a is 1, Z 2 is a group of the formula (I), given and defined above, k is 1 and l is 1; or
- (ii) a is 1, Z 2 is a group of the formula (I), given and defined above, k is 1 and l is zero.
- Yet another preferred range of dyes embodying the invention has the formula (XXXII)
- Z1-L1-Z3-L3-D-Z2 (XXXII)
- wherein:
- Z 3 is a third reactive group selected from the groups (I)-(III), given and defined above; and
- L 3 is a third linking group selected from the groups (VII)1, (VII)2 and (VII),3 given and defined above;
- Z 2 is a second reactive group selected from the groups (IV)-(VI), given and defined above; and
- each of Z 1 and L1 is as defined above.
- In such dyes of the formula (XXXII), L 1 is preferably a linking group of the formula (VII)2 or (VII)3, given and defined above and Z3 is preferably a group of the formula (II), given and defined above.
- Another range of dyes embodying the invention has the formula (XXXIII)
- D-L2-Z2-L1-Z1 (XXXIII)
- wherein:
- Z 2 is selected from groups of the formulae I-(III), given and defined above; and
- each of D, Z 1, L1 and L1 is as defined above.
- In such dyes of the formula (XXXIII), Z 2 is preferably a group of the formula (II), given and defined above and L2 is preferably a linking group selected from the groups (VII)1, (VII)2 and (VII)3, given and defined above, more preferably a group of the formula (VII)2 or (VII)3, given and defined above; and
- L 1 is a group of the formula (VII)3, given and defined above, in which B is an optionally substituted aryl group, or is alternatively a chromophoric bridging group.
- In the range of dyes of formulae (XXXII), a preferred chromophore D is a disazo dye containing a residue derived from H-acid and having azo groups at the 2- and 7- positions.
-
- Other ranges of dyes embodying the invention are of the respective formulae
- D2-L2-Z2-L3-D1-L1-Z1 (XXXIV)
- wherein:
- each of D 1 and D2, independently, is a chromophore;
- L 3 is a linking group selected from groups of the formula (I)-(III), given and defined above; and
-
- wherein:
- each of D 1 and D2 1 independently, is a chromophore;
- L 3 is a linking group selected from the groups of the formulae (I)-(III), given and defined above; and
-
- wherein:
- one of Z 3 and Z4 is a reactive group Z2;
- each of Z 3 and Z4, independently, is a reactive group selected from the formulae (IV)-(VI), given and defined above;
- each of t and u, independently, is zero or 1 and at least one of t and u is 1;
- D is a chromophore;
- Ar is an optionally substituted aryl group;
-
- wherein each R, independently, is as defined in claim; and
- Z 1 is as defined above.
- In such dyes of formulae (XXXVI), D is preferably a disazo dye containing a residue derived from H-acid.
-
- wherein each of B,R and b is as defined above and the bond {circle over (1)} is linked to the heterocyclic nitrogen atom of the chromophore.
- Such dyes, may have the formula
- Z1-L1-DNL2a Z2 L5—Ar]t
- wherein:
- D N is a chromophore containing a heterocyclic group including a nitrogen atom;
- L 1 is a group of the formula (VII)4 or (VII)5, given and defined above, directly attached via the bond {circle over (1)}, to the nitrogen atom of the said chromophore DN;
- Ar is an optionally substituted aryl group;
- and each of Z 1, Z2, L2, L5, a and t is as defined above.
-
- In the above formulae, wherever L 1-L5, J1, J2 or B is or has an optionally substituted phenyl group, optional substituents are SO3H or a salt thereof, C1-4alkyl, especially methyl and chloro, especially SO3H.
- Again, in all of the above formulae, where the groups Z 2 is any of the groups of the formulae (I)-(III), it is most preferably of the formula (II).
- Likewise in all such formulae, where Z 2 i s any of the groups (IV)(VI), it is most preferably of the formula (IV) or (V), wherein Z is zero.
- In a dye according to the invention, in general, the or each chromophoric group independently preferably comprises an azo (which may be a monoazo, polyazo or metal complex azo), anthraquinone, hydrazone, phthalocyanine, triphenodioxazine or formazan group. Examples of chromophoric groups which may be present are those given as types (a)-(g) of the group “D 1” in formula (I) of U.S. Pat. No. 5,484,899, which is incorporated herein by reference.
- Preferred azo groups are monoazo and disazo groups. Preferred monoazo groups have the formula
- —Ar1—N═N—Ar2—
- wherein Ar 1 is an aryl or heteroaryl group and Ar2 is an aryl group.
- It is preferred that each aryl group independently is a mono- or di-cyclic aryl group. Preferred aryl groups are optionally substituted phenyl and optionally substituted naphthyl. Preferred heteroaryl groups are pyridonyl and pyrazolonyl.
-
- wherein:
- Ar 1 is an aryl group, preferably a benzene or naphthalene nucleus;
- R 3, or each R3 independently, is C1-4 alkyl, nitro, halo or sulphonic acid or a salt thereof;
- c is zero or 1 to 4;
- R 4, or each R4 independently, is a sulphonic acid or a salt thereof; and
- d is 1 or 2;
-
- wherein each of Ar 1R3 and a are as defined above, R4 is sulpho and c is zero or 1.
- Ar 1 is preferably optionally substituted phenyl or naphthyl, especially a phenyl or naphthyl group having at least one sulpho substituent. Further optional substituents which may be present on Ar include a halogen atom, especially chlorine; an alkyl radical, especially C1-4 alkyl, more especially methyl; an acylamido radical, especially acetylamino, benzamido or sulphonated benzamido; amino; hydroxy; and an alkoxy radical, especially C1-4 alkoxy, more especially methoxy.
- As examples of phenyl groups having at least one sulpho substituent there may be mentioned 2-, 3- or 4-sulphophenyl; 2-sulpho-4-nitrophenyl; 2-sulpho-5-nitrophenyl; 4-sulpho-2-methylphenyl; 5-sulpho-2-methylphenyl; 2-sulpho-4-methylphenyl; 5-sulpho-2-methoxyphenyl; 2-sulpho-4-methoxyphenyl; 4-sulpho-2-chlorophenyl; 5-acetamido-2-sulphophenyl; 5-sulpho-2-carboxyphenyl; 2,4-disulphophenyl; 2,5-disulphophenyl; and 3,5-disulphophenyl.
- As examples of naphthyl groups having at least one sulpho substituent there may be mentioned 1-sulphonaphth-2-yl; 1,5-disulphonaphth-2-yl; 1,5,7-trisulphonaphth-2-yl; 3,6,8-trisulphonaphth-2-yl; 5,7-disulphonaphth-2-yl; 6-sulphonaphth-2-yl; 4-, 5-, 6-, or 7-sulphonaphth-1-yl; 4,8-disulphonaphth-1-yl; 3,8-disulphonaphth-1-yl; 2,5,7-trisulphonaphth-1-yl; and 3,5,7-trisulphonaphth-1-yl.
- Preferred optional substituents which may be present on the naphthalene nucleus of Formula (IX) are those mentioned above for Ar 1.
- Groups of the Formula (IX) are preferably linked to a group L 1 or L2 at the 6-, 7- or 8-position, especially the 6- or 8-position. When L1 or L2 is to be linked at the 8-position, it is preferred that R5 is a sulpho group at the 5- or 6- position.
-
- wherein:
- each R independently and a is as defined above;
- one of Z 3 and Z4 is a group Z1 and the other is a group Z2;
- the group Z 4 is selected from the groups of the formulae (I)-(III), given and defined above.
- Ar 1 is a benzene or naphthalene nucleus;
- R 3, or each R3 independently, is C1-4 alkyl, nitro, halo or sulphonic acid or salt thereof;
- c is zero or 1-4;
- R 4, or each R4 independently, is a sulphonic acid or a salt thereof; and
- d is 1 or 2.
- A preferred disazo group is of the Formula (XI) (or salt thereof):
- Ar1—N═N—M—N═N—E (XI)
- wherein:
- M and E are each independently optionally substituted phenylene or naphthalene; and
- Ar 1 is as defined above.
- It is preferred that E is optionally substituted naphthalene and M is optionally substituted phenylene. The optional substituents which may be present on M or E are preferably independently selected from halo, especially chloro; alkoxy, especially C 1-4 alkoxy; alkyl, especially methyl; sulpho; carboxy; hydroxy; amino; acylamino, especially acetamido, benzamido and sulphonated benzamido, and pyrimidinylamino or triazinylamino cellulose-reactive groups.
- As Examples of groups represented by M and E, there may be mentioned phenylene, 2-methyl-1,4-phenylene, sulphophenylene, ureidophenylene, 7-sulpho-1,4-naphthalene, 6-sulpho-1,4-naphthalene; 8-sulpho-1,4-naphthalene and 6-hydroxy-4-sulpho-1,5-naphthalene.
-
- wherein:
- one of Z 5 and Z6 is a group Z1 and the other is a group Z2;
- each of f and g independently is zero or 1;
- when Z 5 or Z6 is any of the groups of the formulae (I)-(III), given and defined above, f or g respectively is 1 and when Z5 or Z6 is any of the groups of the formulae (IV)-(VI), given and defined above, f or g respectively is zero;
- each of c and e, independently, is zero or 1-4;
- d is 1 or 2;
- each R independently is as defined above;
- each of Ar 1 and Ar2 independently is an optionally substituted aryl group; and
- each of R 3 and R4 is as defined above.
-
- wherein X, Y and n are as defined above.
- Alternatively, one of Z 5 and Z6 may be a group of the formula (I), given and defined above, and the other of Z5 and Z6 may be selected from groups of the formulae (II) and (III), given and defined above. In such a dye it is preferred that one of Z5 and Z6 is a group of the formula (I), given and defined above, and the other of Z5 and Z6 is a group of the formula (II).
-
- wherein:
- B is a hydrocarbon bridging group as defined above, and preferably an optionally substituted aryl group;
- one of G 1 and G2 is OH and the other is NH2;
- each of X, Y, Y 1, Ar1Ar2, R3, R4, R5, c, d and e is as defined above.
-
- wherein: X, Y and n are as defined in claim 1 and the other of Z5 and Z6 is the group —SO2CH2CH2OSO3H or —SO2CH═CH2.
-
- wherein
- G 3 is C2H4OSO3H or a salt thereof or —CH═CH2;
- G 1 and G2 are as defined above;
- R 4 and d are as defined above; and
- each of h and i, independently, is zero, 1 or 2.
-
- where each of G 1 G2 and G3 is as defined above.
-
- wherein the anthraquinone nucleus optionally contains a sulphonic acid group in the 5-, 6-, 7-, or 8-position and V is a divalent organic linking group, preferably of the benzene series.
- V is a bridging group B, preferably phenylene, diphenylene, or 4,4′-divalent stilbene or azobenzene radicals which are optionally sulphonated. It is preferred that V contains one sulphonic acid group for each benzene ring present therein.
- A preferred anthraquinone dye of has the formula
- DA-L3-Ar- L2-Z2- L1-Z1
- wherein:
- D A is an anthraquinone chromophore;
- L 3 is a linking of the formula (VII)1, given and defined above;
- Ar is an optionally substituted aryl group;
- and
- each of Z 1, Z2, L1 and L2 is as defined above.
- More preferably each of L 1 and L2, independently, is a linking group of the formula (VII)1, (VII)2 or (VII)3, given and defined above; and still more preferably, each of L1 and L2 1 independently is a group of the formula (VII)3, given and defined above, in which B is an optionally substituted aryl group.
-
- wherein Pc is a metallo-phthalocyanine nucleus, preferably copper or nickel phthalocyanine; L is as hereinbefore defined; each W independently is a hydroxy or a substituted or unsubstituted amino group, V 1 is a divalent organic linking group, preferably a C1-4-alkylene or phenylene linking group; and a and b are each independently 1, 2 or 3 provided that a+b is not greater than 4.
-
- wherein:
- Cu Pc is a copper phthalocyanine chromophore;
- X+Y+Z≦4;
- each of R 21 and R22, independently, is hydrogen or optionally substituted C1-4alkyl;
- B is a hydrocarbon bridging group; and
- Z 1 is as defined above.
-
- wherein:
- each Y 3 independently is a covalent bond, C2-4-alkylene, phenylene or sulphophenylene;
- U 1 is H or SO3H; and
- T 1 and T2 are halo, especially chloro, C1-4-alkyl, or C1-4 alkoxy.
- Each Y 3 is preferably —CH2H4— or —C3H6—, U1 is preferably SO3H and T1 and T2 are preferably Cl, methyl or ethyl.
- Preferably such a triphendioxazine dye has the formula
- Z1-L1-[Z3-L3]q-DT-[Z4]r-L2-Z2
- wherein:
- D T is a triphendioxazine chromophore;
- each of L 2, L3 and L4, independently, is a linking group of the formula (VII)1 1 (VII)2 or (VII)3 given and defined above;
- each of Z 2, Z3 and Z4 is a reactive group selected from groups of the formulae (I)-(III), given and defined above;
- each of q and r is zero or 1; and
- each of Z 1 and L1 is as defined above.
- In such a dye, Z 2 is preferably a group of the formula (I), given and defined above and each of Z3 and Z4 is a group of the formula (II), given and defined above; and each of L1, L2, L3 and L4 is preferably a group of the formula (VII)3, given and defined above.
-
- wherein:
- X 1 is H, SO3H or Cl; and
- each r independently has a value of 0, 1 or 2.
- provided that the formazan group has at least one, and preferably at least two, sulpho groups.
- It is preferred that each r has a value of 1.
- Preferably such a formazan dye has the formula
- [ZA]a-DF-L-ZB-L1-Z1
- wherein:
- D F is a formazan chromophore;
- one of Z A and ZB is a group Z2, given and defined above;
- each of L 3 and L4 is a linking group of the formula (VII)1, (VII)2 or (VII)3, given and defined above;
- each of Z 1 and L1 is as defined above;
- a is zero or 1;
- Z A is selected from groups of the formulae (IV)-(VI), given and defined above; and
- Z 8 is selected from groups of the formulae (I)-(III),given and defined above.
- According to one process aspect, the invention provides a process for preparing a dye of the formula (VIII) 1
- Z1 -L1-D-L2-Z2 (VIII)1
- wherein:
- D is a chromophore;
- each of L 1 and L2 independently, is N(R);
- each R, independently, is hydrogen or C 1-4-alkyl;
-
- in which:
- n is 1 or 2
- X, or each X independently, is an electron withdrawing group; and
- Y is a halogen atom,
- which process comprises reacting a chromophoric compound of the formula (XX)
- H(R)N-D-N(R)H (XX)
-
- wherein each of X, Y and n is as defined above, to obtain the dye of the formula (VIII) 1.
- When the dye is of the formula (XVIII) given above, the chromophoric compound of the formula (XX) can be prepared by protecting one amino group of a phenylene diamine sulphonic acid and then diazotising this and coupling a first portion of the diazotised phenylene diamine sulphonic acid to the naphthalene nucleus under acid conditions so as to couple on to the benzene ring of the naphthalene nucleus containing an amino group and then taking a second portion of the same diazotised and protected phenylene diamine sulphonic acid compound referred to above (or a different such compound) and coupling this under neutral or alkaline conditions to the benzene ring of the naphthalene nucleus containing a hydroxyl group to obtain a diamine dyestuff having respective protected amino groups. The protective group can then be removed by hydrolysis.
- According to another process aspect, the invention provides a process for preparing a dye of the formula (VIII) 1
- Z1-L1-D-L2-Z2 (VIII)2
- wherein:
- D is a chromophore;
- each of L 1 and L2 independently, is N(R);
- each R, independently, is hydrogen or C 1-4alkyl;
-
- in which:
- n is 1 or 2
- X, or each X independently, is an electron withdrawing group; and
- Y is a halogen atom; and
- Z 2 is selected from the groups (II) and (III) defined above;
- which process comprises reacting a chromophoric compound of the formula (XX)
- H(R)N-D-N(R)H (XX)
- wherein each of D and R is as defined above, with one mole, per mole of the chromophoric compound of the formula (XX), of each of
-
- wherein each of Z, X, Y and n is as defined above; and
-
- wherein:
- m is 1 or 2; p is 0 or 1; when m is 1, p is 1; and when m is 2, p is 0;
- Y 1, or each Y1 independently, is a halogen atom or an optionally substituted pyridinium group; and
- T is C 1-4alkoxy, C1-4thioalkoxy or N(R1) (R2), in which each of R1and R2 independently is hydrogen, optionally substituted C1-4alkyl or optionally substituted aryl;
- x is 1, 2 or 3; y is zero, 1 or 2; and x+y≦3;
- Y 2, or each Y2 independently, is a halogen atom or an optionally substituted pyridinium group;
- U or each U independently, is C 1-4alkyl or C1-4alkylsulphonyl; and
- Y is as defined above;
- the said reactions of the compound of the formula (XX) with each of the respective compounds of the formulae (XII) and (XIII) being carried out simultaneously or one before the other, in either order, to obtain a dye of the formula (VIII) 2.
- According to yet another process aspect, the invention provides a process for preparing a dye of the formula (VIII) 3
- Z1-L1-D-Z2 (VIII)3
- wherein:
- D is a chromophore
- L 1 is N(R), in which R is hydrogen or C1-4alkyl;
-
- in which n is 1 or 2; X, or each X independently, is an electron withdrawing group; and Y is a hydrogen atom; and Z 2 is a group of the formula (IV)
- —SO2 CH2 CH2 X1 (IV)
- wherein
- X 1 is an eliminatable group;
- a group of the formula (V)
- —SO2(CH2)2CH═CH2 (V)
- wherein
- z is zero or 1; and
- a group of formula (VI)
- —W—C(R10)═CH2 (VI)
- wherein:
- R 10 is hydrogen, C1-4alkyl or halogen; and
- W is —OC(═O)— or —N(R 11)C(═O)—
- in which R 11 is hydrogen or C1-4alkyl;
- which process comprises reacting a chromophoric compound of the formula (XXIV)
- H(R)N-D-Z2 (XXIV)
-
- wherein each of X, Y and n is as defined above, to obtain the dye of the formula (VIII) 3.
- Although dye formulae have been shown in the form of their free acid in this specification, the invention also includes dyes and processes using dyes in the salt form, particularly their salts with alkali metals such as the potassium, sodium, lithium or mixed sodium/lithium salt and their salts with tetraalkylammonium ions.
- The dyes may be used for dyeing, printing or ink-jet printing, for example, of textile materials and paper.
- The process for colouration is preferably performed at a pH of 7.1 to 13, more preferably 10 to 12. pH levels above 7 can be achieved by performing the process for colouration in the presence of an acid-binding agent.
- The substrate may be any of a textile material, leather, paper, hair or film, but is preferably a natural or artificial textile material containing amino or hydroxyl groups, for example textile material such as wool, silk, polyamides and modified polyacrylonitrile fibres, and more preferably a cellulosic material, especially cotton, viscose and regenerated cellulose, for example, that commercially available as Tencel. For this purpose the dyes can be applied to the textile materials at a pH above 7 by, for example, exhaust dyeing, padding or printing, including ink-jet printing. Textile materials are coloured bright shades and possess good fastness to light and wet treatments such as washing.
- The new dyes are particularly valuable for colouring cellulosic textile materials. For this purpose, the dyes are preferably applied to the cellulosic textile material at a pH above 7 in conjunction with a treatment with an acid binding agent.
- Preferred acid-binding agents include alkali metal carbonates, bicarbonates, hydroxides, metasilicates and mixtures thereof, for example sodium bicarbonate, sodium carbonate, sodium metasilicate, sodium hydroxide and the corresponding potassium salts. The dyes benefit from the excellent build-up and high fixation.
- At least for cellulosic materials, exhaust dyeing can be carried out at a relatively low temperature of about 50-70° C., especially about 60° C.
- The new dyes can be applied to textile material containing amine groups, such as wool and polyamide textile materials, from a neutral to mildly alkaline dyebath. The dyeing process can be carried out at a constant or substantially constant pH, that is to say that the pH of the dyebath remains constant or substantially constant during the dyeing process, or if desired the pH of the dyebath can be altered at any stage of the dyeing process.
- The dyes may be in a liquid form, or solid form, for example in granular or powdered form.
- We find surprisingly that such dyes give a much higher build up, as compared with known dyestuffs, particularly in warm dyeing applications at about 60° C.
- In addition, a wide selection of dye backbones is possible, giving the potential to provide high fastness dyes.
- Dyes embodying the invention are especially useful for application to substrates by ink-jet technologies. Substrates which are particularly useful are cellulosic textiles and paper.
- The dye used in the ink is preferably purified by removal of substantially all the inorganic salts and by-products which are generally present in a commercial dye at the end of its synthesis. Such purification assists in the preparation of a low viscosity aqueous solution suitable for use in an ink jet printer.
- To assist in the achievement of heavy depths of shades the dye should preferably have a water-solubility of at least 5%, and more preferably from 5% to 25%, by weight. Solubility of the dye can be enhanced by converting the sodium salt, in which form it is normally synthesised, either partially or wholly, into the lithium or ammonium salt. Purification and ion exchange can conveniently be effected by use of membrane separation processes which permit the separation of unwanted inorganic materials and by-products directly from an aqueous solution or dispersion of the dye followed by partial or complete exchange of the counter-ion. The ink preferably contains up to 20% by weight of dye and more preferably from 2% to 10%, especially from 3% to 8%.
- The ink may also contain a humectant, which may also function as a water miscible solvent, which preferably comprises a glycol or dihydroxyglycolether, or mixture thereof, in which one or both hydroxy groups are secondary hydroxy groups, such as propane-1,2-diol,butane-1,3-diol and 3-(3-hydroxy-prop-2-oxy)propan-2-ol.
- Where the humectant has a primary hydroxy group this is prefereably attached to a carbon atom adjacent to a carbon atom carrying a secondary or tertiary hydroxy group. The humectant may comprise up to a total of 10% by weight of a polyol, especially a glycol or dihydroxyglycolether, having two or more primary hydroxy groups, such as ethyleneglycol, propane-1,3-diol,butane-1,4-diol, 2-(2-hydroxyethoxy)ethanol and 2-(2-[2-hydroxyethoxy)ethanol and/or an alcohol with a primary hydroxy group, such as ethanol,n-propanol and n-butanol. However, it preferably contains not more than 5% by weight, and is more preferably free from, such compounds. In the context of the humectant, the term “alcohol” means a compound having only one hydroxy group attached to an aliphatic carbon atom. The ink preferably contains from 5% to 25%, by weight, more especially from 10% to 20%, of humectant.
- If desired, the ink may be buffered to a pH from 5 to 8, especially to a pH from 6 to 7, with a buffer such as the sodium salt of metanilic acid or an alkali metal phosphate, or di- or triethanolamine.
- The ink preferably also contains one or more preservatives to inhibit the growth of fungi, bacteria and/or algae because these can block the jet of the ink jet printing equipment. Where the ink jet printing technique involves the charging and electrically controlled deflection of drops the solution preferably contains a conducting material such as an ionised salt to enhance the accumulation of charge on the drop. Suitable salts for this purpose are alkali metal salts of mineral acids. The remainder of the ink is preferably water, especially de-ionised water to avoid the introduction of impurities into the ink.
- Especially preferred embodiments of the invention will now be described in more detail with reference to the following Examples in which all parts and percentages are by weight unless otherwise stated. Although preparation and dyeing with any single dye is exemplified, particular advantage could be seen when dyeing with mixtures of dyes.
- 2-amino-4-(N-acetyl)aminobenzene-1-sulphonic acid (0.5M, 182 g) was dissolved in water (600 ml) at pH 7 and 2M sodium nitrite solution added (0.526M, 263 ml). This solution was added dropwise to a mixture of hydrochloric acid 35% (1.13M, 100 ml, SG=1.18) and ice (1 Kg), maintaining a temperature of 0-5° C. The mixture was stirred for 0.5 hrs with excess nitrous acid at 0-5° C. A solution of sulphamic acid (10%) was added to remove excess nitrous acid to obtain a suspension of a diazotized diamine for coupling.
- H-Acid (0.475M, 183 g) was dissolved in water (800 ml) at pH 7.5. This solution was added dropwise to the above diazo suspension with good agitation over 1 hr at 0-2° C. The mixture was then stirred at 0-2° C. for a further 2 hrs at pH 2-3, and allowed to warm to room temperature over 18 hrs.
- The viscous suspension was adjusted to pH 6 using sodium hydroxide solution (46/48%) and a solution of a monoazo dye was thereby obtained.
- The above monoazo dye solution was cooled to 0-5° C. and a batch of the diazotized diamine prepared as above was added. The mixture was stirred at 0-5° C. and pH 6-7 for 2 hrs and subsequently over 18 hrs at room temperature while maintaining the pH at 6-7 using 2M sodium carbonate solution, thereby obtaining a disazo dye solution.
- Hydrolysis was then carried out by adding sodium hydroxide solution (46/48%, 800 g) to the above disazo dye solution (vol=5 L) and heating at 70-75° C. for 1.5 hrs.
- The reaction mixture was cooled to 20° C. and neutralised by controlled addition of 35% hydrochloric acid. After screening to remove a small amount of impurity sodium chloride was added (15% w/v) and stirring continued to allow precipitation of the resultant diaminodisazo product. This was collected by filtration and dried at 40° C. (285 g; 0.242M; strength=68.3%).
- The above diaminodisazo dye (0.01M, 11.78 g) was dissolved in water/acetone (100 ml, 1:1) at 50° C. A solution of 1,5-difluoro-2,4-dinitrobenzene (0.023M, 4.7 g taken as 100% strength) in acetone (20 ml) was added over 0.25 hr and the mixture heated at 55° C. for 3 hrs. The pH was maintained at 7 throughout the reaction by the addition of 2N sodium carbonate solution.
- The reaction mixture was cooled to 20° C. and acetone (150 ml) added to precipitate the dye. The product was collected by filtration and washed with acetone (50 ml) and dried at 40° C. to give a greenish-navy dye(11.12 g; 0.0075M; strength 74.8%) (λ Max=607 nm; εMax=57,036).
- The method of Example 1 was followed except that the diaminodisazo was reacted with 1,5-dichloro-2,4-dinitrobenzene in place of 1,5-difluoro-2,4-dinitrobenzene to give a greenish-navy dye(λ Max=607 nm; εMax=48,212).
- The method of Example 1 was followed except that the diaminodisazo was reacted with 1,5-difluoro-2-cyan-4-nitrobenzene in place of 1,5-difluoro-2,4-dinitrobenzene to give a greenish-navy dye(λ Max=607 nm; εMax=56,416).
- The method of Example 1 was followed except that the diaminodisazo was reacted with 1,5-difluoro-2-nitrobenzene in place of 1,5-difluoro-2,4-dinitrobenzene to give a greenish-navy dye(λ Max=608 nm; εMax=54,660).
- 4-Amino benzene sulphatoethylsulphone (0.1M, 30 g) was stirred in ice/water (400 ml) and hydrochloric acid 35% (0.58M, 52 ml, SG=1.18) and the temperature maintained at below 5° C. 2M Sodium nitrite solution (0.104M, 52 ml) was added dropwise at below 5° C. and the mixture stirred for a further 2 hours. A solution of sulphamic acid was added (10%) to remove excess nitrous acid and provide a diazo suspension for coupling.
- H-Acid (0.103M, 42.6 g) was dissolved in water (300 ml) at pH 6 and the solution cooled to 5° C. This solution was added dropwise to the above diazo suspension with good agitation, while maintaining the temperature below 5° C. The mixture was then stirred for 18 hours, allowing the temperature to rise to 20° C. Sodium chloride (10% w/v) was added and the mixture stirred for 1 hour. The precipitated monoazo dye was collected by filtration and reslurried in ethanol (600 ml) for 1 hour at 20° C. The product was collected by filtration and dried at 40° C. (70 g; 0.0825M; strength=72%).
- 3-Amino-5′-fluoro-2′,4′-dinitrodiphenylamine-4-sulphonic acid (0.0068M, 3.2 g) was dissolved in water (100 ml) at 50-60° C. and the solution cooled to 20° C. 2M Sodium nitrite solution (0.008M, 4 ml) was added and the mixture cooled to 0-2° C. and added dropwise to ice (50 g) and hydrochloric acid 35% (0.09M, 8 ml), while maintaining the temperature at 0-2° C. The resulting yellow suspension was stirred at 0-2° C. for 0.5 hour and a solution of sulphamic acid (10%) added to remove excess nitrous acid and provide a diazo suspension for coupling.
- The diazo suspension was then added dropwise to a solution of the above monoazo dye (0.006M, 5.1 g) in water (150 ml) at pH 5 and 0-2° C. The pH of the coupling mixture was maintained at pH 5 by the addition of 2M sodium carbonate solution and stirred at this pH for 18 hours, allowing the temperature to rise to 20° C. Sodium chloride was added (20% w/v) and the product collected by filtration. The solid was reslurried in ethanol (100 ml) for 1 hour at 20° C., collected by filtration and dried at 40° C. to obtain a greenish-navy dye (4.4 g; 0.003M; strength=67%) (λ Max=603 nm; εMax=54,279) of the structure given below:
- Other disazo naphthalene structures embodying the invention are shown as Examples 6 to 61 in Table 1. They can be prepared by methods analogous to those outlined in Examples 1 to 5.
- Still further disazo naphthalene dyes embodying the invention, in which the chromophore has been doubled up via a linking group, are shown in Examples 62 to 67 in Table 2.
-
- The N-dichlorotriazinyl derivative of the azo dye resulting from azo-coupling 7-aminonaphthalene-1,3,6-trisulphonic acid with m-ureidoaniline was prepared by conventional means. A solution of this dye (35 mmol in 450 ml) was added at room temperature with stirring to N-(4-aminophenyl)piperazine (7.5 g, 42 mmol) dissolved in 50/50 acetone/water (400 ml) maintained at pH 6-6.5 by addition of sodium carbonate solution. After completion of the reaction the solution was concentrated, and the product was precipitated by addition of methylated spirit. To a solution of this intermediate (8.4 mmol) in 50/50 acetone/water (200 ml) was added a solution of 1,5-difluoro-2,4-dinitrobenzene (8.5 mmol) in acetone (20 ml), maintaining the pH at 8.5 by addition of sodium carbonate solution. On completion of the reaction, the pH was adjusted to 6.5 and the solution was concentrated to ca 100 ml. Methylated spirit was added, and the product dye was filtered, washed with meths and dried. λ Max(water)=379 nm, εMax=33000 1 mol−1 cm−1, half band width >150 nm. This material dyed cotton a bright golden yellow shade with very good fastness properties.
- By following the principles described in Example 68, dyes of a similar nature may be prepared as further exemplified by dyes 69-77 listed in Table 3.
- Disazo naphthalene dyes containing two reactive groups attached to the chromophore as described in Example 68 are exemplified by Examples 78 to 97 listed in Table 4.
- Further yellow dyes may be prepared by the methods described in Examples 1-6 and 68, and are listed in Tables 5 to 9.
- Monoazo naphthalene dyes embodying the invention are listed in Tables 10 to 12.
- A variety of dyes embodying the invention, containing blue and green chromophores, are listed in Tables 13 to 16.
- Each of the dyes (0.2 parts) of respective Examples 1, 3 and 5 was dissolved in respective amounts of water (50 parts) at 25° C. and pH 9. Cotton fabric (5 parts) and Glaubers salt (2.5 parts) were added. The dyes were fixed to the cotton by raising the temperature to between 50° C.-60° C., basifying to pH 11.0 and maintaining this for 1 hr. The cotton was removed and washed in soapy water to give a dark greenish-navy cotton having high general fastness properties.
- Each of the dyes (0.2 parts) of respective Examples 2 and 4 was dissolved in water (50 parts) at 25° C. and pH 9. Cotton fabric (5 parts) and salt (4 parts) were added. The dyes were fixed to cotton by raising the temperature to 80-100° C. and maintaining this for 1 hr. The cotton was removed and washed in soapy water to give a deep greenish-navy cotton having good fastness properties.
- Methodology for applying dyes embodying the present invention to cotton may be further exemplified by means of the following pad-batch dyeing protocol.
- Example 195.-Dye from Example 5 (0.5 parts) was dissolved in water (30 parts) at 25° C. and the following agents were added: Primasol NF (1 part of 20% solution) and sodium silicate Q70 (9.5 parts of 50% solution), sodium hydroxide (5.1 parts of 10% solution). The solution was made up to 50 parts by addition of water, and then padded onto woven cotton fabric (70% pick-up). The cloth was wrapped in cling film and batched at room temperature for 24 hours. The cling film was removed and the dyed cloth was rinsed successively with cold water and hot water, then washed with a soap solution, rinsed with water, and dried to give cotton coloured a dull greenish blue shade.
- If the amount of dye used in Example 193 is doubled and the procedure repeated, a greenish-navy shade is obtained.
- If the amount of dye used in Example 193 is trebled and the procedure repeated, a dark navy shade is obtained.
- If six times the amount of dye in Example 193 is used and the procedure repeated, an almost black shade is obtained.
- Other dyes from the above Examples, particularly those containing the 2,4-dinitrofluorobenzene unit and/or a vinyl sulphone group or its sulphate half-ester precursor, can be applied to cotton by the same method.
- Dyes embodying the present Invention may be applied to textile substrates, especially cotton, by conventional printing technology, as the following exemplifies.
- A dye from Example 5 (30 parts) was dissolved in a solution containing Manutex F 700 (500 parts of a 10% solution) and Vitexol D (20 parts). The solution was made up to 1000 parts by the addition of water and printed by means of a Zimmer screen printer. The printed cloth was dried and padded through a solution containing sodium silicate (48 degBe, 700 parts) made up to 1000 parts by the addition of water (80% pickup) . Immediately after padding, the printed cotton was steamed in a Roaches Flash-ager steam chamber at 120 deg C. for 45 seconds. The printed cloth was rinsed in cold water, washed with a soap solution at the boil, rinsed again in cold water and dried to give a dull greenish navy print on the cotton.
- Dyes embodying the present invention may be used in the preparation of inks specially formulated for application by ink-jet technology. As an Example, the dye from Example 5 (6 parts) was dissolved in a solution of propylene glycol (15 parts) and water (79 parts) . When this solution was applied to cotton which had previously been pretreated (for example, with a pretreatment agent described in EP-A-0534660) by means of commercial ink-jet printing equipment, deep navy shades were obtained.
- Other dyes from the above examples as well as others described by the Invention may be used to prepare inks suitable for ink-jet printing.
- Reactive dyes, including those of Examples 1-189, are usually isolated as their sodium salts, and are contaminated with inorganic impurities resulting from the method of preparation. Dyes free of impurities suitable for ink jet printing, and/or with increased solubility, may be prepared by conventional ion exchange techniques, where for example sodium is replaced by lithium and inorganic impurities are simultaneously removed.
- Dye from Example 1 (10 parts) was dissolved in water (100 parts) and treated on a reverse osmosis rig until the permeate conductivity was 10 micro reciprocal ohms. A solution of lithium chloride was added to the dye solution and treatment on the r.o. rig was continued until the permeate conductivity had decreased to 1 micro reciprocal ohm. The sample was concentrated to a volume of about 80 parts, after which the solution was buffered and other formulating agents were added. The dye solution was then diluted to 90 parts by addition of water, at which stage it was suitable for storage. Dye solution prepared in this way could be diluted and applied to cotton by the methods described in any of the Examples 190, 195-198, or formulated into an ink by addition of suitable humectants and/or cosolvents, and applied by ink-jet methodology to cotton, for example by the method described in Example 200. In all these cases, dull blue, greenish navy or black shades were imparted to the substrate, depending on the amount of dye applied.
- Dye from Example 1 was ion exchanged to the lithium form as in Example 201, and formulated into an ink with the composition: dye (5 parts), propylene glycol (12 parts), diethanolamine (sufficient to buffer the final pH to 7-8.5), and water (to bring the total to 100 parts). The ink was added to the ink reservoir of an ink jet printer (e.g. HP Desk Jet 500) and printed onto paper (Logic 300), to give a black print of generally good fastness properties.
TABLE 1 λmax nm (w1/2 nm) Example A B Colour on cotton 6 606 (121) greenish-navy 7 607 (105) greenish-navy 8 623 (121) dark bluish-green 9 618 (121) greenish-navy 10 603 (105) greenish-navy 11 618 (130) greenish-navy 12 614 (110) dark bluish-green 13 594 (112) greenish-navy 14 595 (108) greenish-navy 15 604 (110) dark bluish-green 16 645 (137) very dull dark green 17 604 (119) greenish-navy 18 603 (127) greenish-navy 19 600 (134) greenish-navy 20 596 120) greenish-navy 21 609 (111) greenish-navy 22 604 (120) greenish-navy 23 625 (104) greenish-navy 24 620 (112) dark bluish-green 25 632 (114) dark bluish-green 26 609 (141) dark bluish-green 27 602 (125) greenish-navy 28 605 (143) dark bluish-green 29 596 (129) greenish-navy 30 607 (109) greenish-navy 31 595 (107) greenish-navy 32 622 (123) greenish-navy 33 619 (114) dark bluish-green 34 606 (114) dark bluish-green 35 618 (113) dark bluish-green 36 616 (110) dark bluish-green 37 616 (106) greenish-navy 38 616 (111) dark bluish-green 39 608 (111) greenish-navy 40 608 (116) greenish-navy 41 608 (116) greenish-navy 42 616 (111) dark bluish-green 43 615 (120) dark bluish-green 44 615 (118) dark bluish-green 45 greenish-navy 46 dark bluish-green 47 greenish-navy 48 dark bluish-green 49 greenish-navy 50 dark bluish-green 51 greenish-navy 52 dark bluish-green 53 dark bluish-green 54 greenish-navy 55 dark bluish-green 56 dark bluish-green 57 greenish-navy 58 greenish-navy 59 dark bluish-green 60 dark bluish-green 61 greenish-navy -
-
-
TABLE 4 Exam- Colour on ple A B X Y cotton 78 NH2 OH Greenish navy 79 OH NH2 Greenish navy 80 NH2 OH Greenish navy 81 OH NH2 Greenish navy 82 NH2 OH Greenish navy 83 OH NH2 Greenish navy 84 NH2 OH Greenish navy 85 OH NH2 Greenish navy 86 NH2 OH Greenish navy 87 OH NH2 Greenish navy 88 NH2 OH Greenish navy 89 OH NH2 Navy 90 NH2 OH Navy 91 OH NH2 Greenish navy 92 NH2 OH Greenish navy 93 OH NH2 Greenish navy 94 OH NH2 Greenish navy 95 NH2 OH Greenish navy 96 OH NH2 Greenish navy 97 OH NH2 Greenish navy -
TABLE 5 Example Structure λmax nm w1/2 nm Colour on cotton 98 Dye-SC2H4OH 407 148 Golden yellow 99 Dye-N(CH3)Ph 403 138 Golden yellow 100 Dye-NHC2H4SO3H 407 129 Golden yellow 101 Dye-NHC2H4OC2H4OH 412 134 Golden yellow 102 Dye-NHC6H3-m-SO3H 409 143 Golden yellow 103 Dye-NHC2H4NH-Dye Golden yellow 104 412 130 Golden yellow -
TABLE 6 Example Structure λmax nm w1/2 nm Colour on cotton 105 Dye-SC2H4OH 437 151 Mid yellow 106 430 135 Mid yellow 107 Dye-NHC2H4OC2H4OH 436 141 Mid yellow 108 435 132 Mid yellow 109 439 143 Mid yellow 110 438 149 Mid yellow 111 437 131 Mid yellow 112 Dye-NHC2H4NH-Dye Mid yellow 113 Dye-NHC3H6NH-Dye Mid yellow -
-
-
-
-
-
-
-
-
-
Claims (83)
1. A dye containing
at least one chromophore D;
at least a first, halobenzene, reactive group Z1, of the formula (I)
in which:
n is 1 or 2
X, or each X independently, is an electron withdrawing group; and
Y is a halogen atom;
at least a second reactive group Z2 selected from
(1) a group of the formula (I), given and defined above, but selected independently thereof;
(2) a group of the formula (II)
wherein
m is 1 or 2; p is 0 or 1; when m is 1, p is 1; and when m is 2, p is 0;
Y1, or each Y1 independently, is a halogen atom or an optionally substituted pyridinium group; and
T is C1-4alkoxy, C1-4thioalkoxy or N(R1) (R2), in which each of R1 and R2 independently is hydrogen, optionally substituted C1-4alkyl or optionally substituted aryl;
(3) a group of the formula (III)
wherein:
x is 1, 2 or 3; y is zero, 1 or 2; and
x+y≦3;
Y2, or each Y2 independently, is a halogen atom or an optionally substituted pyridinium group; and
U or each U independently, is C1-4alkyl or C1-4alkylsulphonyl;
(4) a group of the formula (IV)
—SO2CH2CH2X1 (IV)
wherein
X1 is an eliminatable group;
(5) a group of the formula (V)
—SO2(CH2)zCH═CH2 (V)
wherein
z is zero or 1; and
(6) a group of formula (VI)
—W—C(R10)═CH2 (VI)
wherein:
R10 is hydrogen, C1-4alkyl or halogen; and
W is —OC(═O)— or —N(R11)C(═O)— in which R11 is hydrogen or C1-4 alkyl;
at least a first linking group L1, linking the said first, halobenzene, reactive group Z1 to one of components (i) the or a chromophore D and (ii) the second reactive group Z2, which said first linking group L1 presents an amino nitrogen to the reactive group Z1 and to the component (i) or (ii) or, when component (i) contains a heterocyclic nitrogen atom, is linked directly to the nitrogen atom and which said first linking group L1 optionally includes a hydrocarbon bridging group, which hydrocarbon bridging group B has at least two carbon atoms, is optionally substituted, optionally includes at least one hetero atom and is optionally a chromophore; and
when Z2 is selected from the said groups (I)-(III), at least a second linking group L2 linking the second reactive group Z2 to one of (i) the or a chromophore D and (ii) the said first reactive group Z1, which said linking group L2 is selected from
(1) a linking group L1, but selected independently thereof; or
(2) an amide linkage; and
(3) a sulphonamide linkage; and optionally at least one aromatic group Ar which, when Z2 is selected from the said groups (IV)-(VI), may carry the said reactive group Z2.
2. A dye according to claim 1 , wherein the linking group L1 has the formula (VII)1
N(R) (VII)1
wherein R is hydrogen or optionally substituted C1-4alkyl, such that the same amino group presents itself to each of the reactive group Z1 and the component (i) or (ii), as defined in claim 1 .
3. A dye according to claim 1 , wherein the linking group L1 is a piperazinoalkylamino group of the formula (VII)2
4. A dye according to claim 1 , wherein the linking group L1 has the formula (VII)3
—N(R)BN(R)— (VII)3
wherein B is a hydrocarbon bridging group as defined in claim 1 , each R, independently , is as defined in claim 1 and B is optionally linked additionally to at least one additional group —N(R).
5. A dye according to claim 4 , wherein the hydrocarbon bridging group B is an optionally substituted aryl group.
6. A dye according to any preceding claim, of the formula (VIII)
Z1-L1-D-(L2)a-Z2 (VIII)
wherein:
D is a chromophore;
each of L1 and L2 is an amine or piperazine linkage of the formula
—N(R)— (VII)1;
wherein:
R, or each R independently, is hydrogen or C1-4 alkyl;
B is a hydrocarbon bridging group which has at least two carbon atoms, is optionally substituted, optionally includes at least one hetero atom and is optionally a chromophore;
a is zero or 1; and
b is from 2 to 6 inclusive;
Z1 is a group
in which:
n is 1 or 2;
X, or each X independently, is an electron withdrawing group; and
Y is a halogen atom; and
when a is 1, Z2 is:
a group of the formula (I), given and defined above but selected independently thereof; or
a group of the formula (II)
wherein:
m is 1 or 2; p is 0 or 1; when m is 1, p is 1; and when m is 2, p is 0;
Y1, or each Y1 independently, is a halogen atom or an optionally substituted pyridinium group; and
T is C1-4 alkoxy, thioalkoxy or N(R1) (R2) in which R1 is hydrogen, optionally substituted C1-4 alkyl or optionally substituted aryl and
R2 is hydrogen or optionally substituted C1-4 alkyl; or
a group of the formula (III)
wherein: x is 1, 2 or 3; y is zero, 1 or 2; and x+y≦3;
Y2, or each Y2 independently, is a halogen atom; and
U, or each U independently, is C1-4 alkyl or C1-4 alkylsulphonyl; and
when a is zero, Z2 is:
—SO2CH2CH2X1 (IV)
in which
X1 is an eliminatable group; or
—SO2(CH2)zCH═CH2 (V)
wherein
z is zero or 1; or
a group of formula (VI)
—W—C(R10)═CH2 (VI)
wherein:
R10 is hydrogen, C1-4alkyl or halogen; and
W is —OC(═O)— or —N(R11)C(═O)— in which R is hydrogen or C1-4 alkyl.
7. A dye according to any preceding claim , wherein, in formula (I), X, or each X independently, is selected from nitro, cyano, alkylsulphonyl, dialkylaminosulphonyl and sulphonic acid or a salt thereof.
8. A dye according to any preceding claim, wherein a group of the formula (II) is present, in which Y1, or each Y independently, is fluorine, chlorine or optionally substituted pyridinium.
9. A dye according to any one of claims 2 to 8 , wherein R, or each R independently, is hydrogen.
11. A dye according to claim 10 , wherein each of Z1 and Z2 is the same as the other.
14. A dye according to any one of claims 1 to 9 , wherein a is zero and Z2 is
—SO2CH2CH2X1 (IV)
where X1 is as defined in claim 1; or
—SO2(CH2)zCH═CH2 (V)
wherein z is as defined in claim 1 .
15. A dye according to any preceding claim, wherein D is an azo chromophore.
16. A dye according to claim 15 , wherein D is a monoazo chromophore.
17. A dye according to claim 16 , which has the formula (XVII)
wherein:
each R independently and a is as defined in claim 1;
one of Z3 and Z4 is a group Z1 and the other is is a group Z2;
the group Z4 is selected from the groups of the formulae (I)-(III), given and defined in claim 1;
Ar is a benzene or naphthalene nucleus;
R3, or each R3 independently, is C1-4 alkyl, nitro, halo or sulphonic acid or salt thereof;
c is zero or 1-4;
R4, or each R4 independently, is a sulphonic acid or a salt thereof; and
d is 1 or 2.
18. A dye according to claim 15 , wherein D is a disazo chromophore.
19. A dye according to claim 18 , which has the formula (XVIII)
wherein:
one of Z5 and Z6 is a group Z1 and the other is a group Z2;
each of f and g independently is zero or 1;
when Z5 or Z6 is any of the groups of the formulae (I)-(III), given and defined in claim 1 , f or g respectively is 1 and when Z5 or Z6 is any of the groups of the formulae (IV)-(VI), given and defined in claim 1 , f or g respectively is zero;
each of c and e, independently, is zero or 1-4;
d is 1 or 2;
each R independently is as defined in claim 1;
each of Ar1 and Ar2 is an optionally substituted aryl group; and
each of R3 and R4 is as defined in claim 17 .
21. A dye according to claim 19 , wherein one of Z5 and Z6 is a group of the formula (I), given and defined in claim 1 , and the other of Z5 and 6 Z is selected from groups of the formulae (II) and (III), given and defined in claim 1 .
22. A dye according to claim 18 , which dye has the formula
wherein:
B is a hydrocarbon bridging group as defined in claim 1;
one of G1 and G2 is ODH and the other is NH2;
each of X, Y, Y1 and n is as defined in claim 1;
each of Ar1, Ar2, R3, R4, c and d is as defined in claim 19; and
each of R5 and e is as defined in claim 19 .
23. A dye according to claim 22 , wherein B is an optionally substituted aryl group.
27. A dye according to claim 1 , of the formula
Z1-L1-D [L3]q-[Z3-L4 r[J1]s[L2]aZ2[L5-J2]t
wherein:
Z3 is a third reactive group selected from the groups of the formulae (I)-(III), given and defined in claim 1;
each of J1 and J2, independently, is an optionally substituted aryl group or a chromophore;
L3 is a linking group linking Z3 and D;
L4 is a linking group linking Z3 and J1;
L5 is a linking group linking Z2 and J2;
each of q, r, s and t independently, is zero or 1;
each of Z1, Z2, L1, L2 and a is as defined in claim 1; and
when at least one of a and t is 1, Z2 is selected from the groups of the formulae (I)-(III), given and defined in claim 1 .
28. A dye according to claim 27 , wherein q is 1, r is 1, s is 1, each of a and t is zero and Z2 is selected from the groups of the formulae (IV)-(VI), given and defined in claim 1 .
29. A dye according to claim 27 , wherein q is 1, r is 1, s is zero, a is zero and t is 1.
30. A dye according to any one of claims 27 to 29 , wherein each of L3 and L4, independently, is selected from one of the groups of the formulae (VII)1, (VII)2 and (VII)3, given and defined in claim 6 .
31. A dye according to claim 30 , wherein each of L3 and L4 is a group of the formulae (VII)1, given and defined in claim 6 .
32. A dye according to claim 27 , wherein q is 1, r is zero, s is 1, a is zero and t is zero.
33. A dye according to claim 32 , wherein L3 is a group of the formula (VII)3
—N(R)BN(R)— (VII)3
wherein B is a triazine group substituted by a non-reactive group.
34. A dye according to any one of claims 27 to 33 , wherein the chromophore D is an azo chromophore derived from 1-hydroxy-8-aminonaphthalene substituted by at least one sulphonic acid group.
35. A dye according to claim 1 of the formula
Z1-L1-D1[L3-Z3(L4-Z4)1L5-D2]k[L2]aZ2
wherein:
D1 is a first chromophore;
D2 is a second chromophore;
Z3, when present, is a third reactive group selected from the groups of the formulae (I)-(III), given and defined in claim 1;
Z4, when present, is a fourth reactive group selected from the groups of the formulae (I)-(III), given and defined in claim 1;
L3 is a linking group linking Z3 to D1;
L4 is a linking group linking Z3 to Z4;
L5 is a linking group linking D2 to one of Z3 and Z4;
each of k and l, independently, is zero or 1; and
each of Z1, Z2, L1, L2 and a is as defined in claim 1 .
36. A dye according to claim 35 wherein a is 1, Z2 is a group of the formula (I), given and defined in claim 1 , k is zero and D1 is a tetrakisazo chromophore containing two residues of H-acid linked together by a group forming part of the chromophore.
37. A dye according to claim 35 , wherein a is 1, each of Z1 and Z2 is a group of the formula (I), given and defined in claim 1 , each of k and l is 1;
each of D1 and D2 is a disazo chromophore containing a respective residue of H-acid,
each of Z3 and Z4 is a group of the formula (II), given and defined in claim 1 , and
L4 is a linking group of the formula (VII)2 or (VII)3, given and defined in claim 6 .
38. A dye according to claim 35 , wherein a is 1, Z2 is a group of the formula (I), given and defined in claim 1 , k is 1 and l is 1.
39. A dye according to claim 35 , wherein a is 1, Z2 is a group of the formula (I), given and defined in claim 1 , k is 1 and l is zero.
40. A dye according to claim 1 , of the formula
Z1-L1-Z3-L3-D-Z2
wherein:
Z3 is a third reactive group selected from the groups (I)-(III), given and defined in claim 1; and
L3 is a third linking group selected from the groups (VII)1, (VII)2 and (VII)3, given and defined in claim 6;
Z2 is a second reactive group selected from the groups (IV)-(VI), given and defined in claim 1; and
each of Z1 and L1 is as defined in claim 1 .
41. A dye according to claim 40 , wherein L1 is a linking group of the formula (VII)2 or (VII)3, given and defined in claim 6 .
42. A dye according to claim 40 or claim 41 , wherein Z3 is a group of the formula (II), given and defined in claim 1 .
43. A dye according to claim 1 , of the formula
D-L2-Z2-L1-Z1
wherein:
Z2 is selected from groups of the formulae I-(III), given and defined in claim 1; and
each of D, Z1, L1 and L2 is as defined in claim 1 .
44. A dye according to claim 43 , wherein Z4 is a group of the formula (II), given and defined in claim 1 .
45. A dye according to claim 43 or claim 44 , wherein L2 is a linking group selected from the groups (VII)1, (VII) 2 and (VII)3 1, given and defined in claim 6 .
46. A dye according to claim 43 , wherein L2 is a linking group of the formula (VII)2 or (VII)3, given and defined in claim 6 .
47. A dye according to any one of claims 43 to 46 , wherein L1 is a group of the formula (VII3) , given and defined in claim 6 , in which B is an optionally substituted aryl group.
48. A dye according to any one of claims 43 to 46 , wherein L1 is a group of the formula (VII3) , given and defined in claim 6 , in which B is a chromophoric bridging group.
49. A dye according to any one of claims 40 to 48 , wherein the chromophore D is a disazo dye containing a residue derived from H-acid and having azo groups at the 2- and 7-positions.
51. A dye according to claim 1 , of the formula
D2-L2-Z2-L3-D1-L1-Z1
wherein:
each of D1 and D2, independently, is a chromophore;
L3 is a linking group selected from groups of the formula (I)-(III), given and defined in claim 1; and
each of Z1, Z2, L1 and L2 is as defined in claim 1 .
53. A dye according to claim 1 , of the formula.
wherein:
one of Z3 and Z4 is a reactive group Z2;
each of Z3 and Z4, independently, is a reactive group selected from the formulae (IV)-(VI), given and defined in claim 1;
each of t and u, independently, is zero or 1 and at least one of t and u is 1;
D is a chromophore;
Ar is an optionally substituted aryl group;
L1 is a group of the formula
wherein each R, independently, is as defined in claim; and
Z1 is as defined in claim 1 .
54. A dye according to claim 53 , wherein D is a disazo dye containing a residue derived from H-acid.
56. A dye according to claim 55 , of the formula
Z1-L1-DNL2aZ2L5—Ar]t
wherein:
DN is a chromophore containing a heterocyclic group including a nitrogen atom;
L1 is a group of the formula (VII)4 or (VII)5, given and defined in claim 55 , directly attached via the bond {circle over (1)}, to the nitrogen atom of the said chromophore DN;
Ar is an optionally substituted aryl group;
and each of Z1, Z2, L2, L5, a and t is as defined in claim 27 .
58. A dye according to any one of claims 27 to 56 , which is an azo dye having at least two azo groups therein.
59. A dye according to claim 15 , wherein D is a trisazo or tetrakisazo chromophore.
60. A dye according to claim 1 , wherein the chromophore D is an anthraquinone.
61. A dye according to claim 60 , which is of the formula
DA-L3-Ar-L2-Z2-L1-Z1
wherein:
DA is an anthraquinone chromophore;
L3 is a linking of the formula (VII)1, given and defined in claim 6;
Ar is an optionally substituted aryl group; and
each of Z1, Z2, L1 and L2 is as defined in claim 1 .
62. A dye according to claim 61 , wherein each of L1 and L2, independently, is a linking group of the formula (VII)1, (VII)2 or (VII)3, given and defined in claim 6 .
63. A dye according to claim 62 , wherein each of L1 and L2, independently is a group of the formula (VII)3, given and defined in claim 6 , in which B is an optionally substituted aryl group.
64. A dye according to claim 1 , wherein the chromophore D is a metal phthalocyanine.
66. A dye according to claim 1 , wherein the chromophore D is a triphendioxazine chromophore.
67. A dye according to claim 66 , which is of the formula
Z1-L1-[Z3-L3]q-DT-[Z4]r-L2-Z2
wherein:
DT is a triphendioxazine chromophore;
each of L2, L3 and L4, independently, is a linking group of the formula (VII)1, (VII)2 or (VII)3, given and defined in claim 6;
each of Z2, Z3 and Z4 is a reactive group selected from groups of the formulae (I)-(III), given and defined in claim 1;
each of q and r is zero or 1; and
each of Z1 and L1 is as defined in claim 1 .
68. A dye according to claim 67 , wherein Z2 is a group of the formula (I), given and defined in claim 1 and each of Z3 and Z4 is a group of the formula (II), given and defined in claim 1 .
69. A dye according to claim 67 or claim 68 , wherein each of L1, L2, L3 and L4 is a group of the formula (VII)3, given and defined in claim 6 .
70. A dye according to claim 1 , wherein the chromophore D is a formazan chromophore.
71. A dye according to claim 70 , of the formula
[ZA]a-DF-L-ZB-L1-Z1
wherein:
DF is a formazan chromophore;
one of ZA and ZB is a group Z2, given and defined in claim 1;
L is a linking group of the formula (VII)1, (VII)2 or (VII)3, given and defined in claim 6;
each of Z1 and L1 is as defined in claim 1 ,
a is zero or 1;
ZA is selected from groups of the formulae (IV)-(VI), given and defined in claim 1; and
ZB is selected from groups of the formulae (I)-(III),given and defined in claim 1 .
78. A process for preparing a dye of the formula (VIII)1
Z1-L1-D-L2-Z2 (VIII)1
wherein:
D is a chromophore;
each of L1 and L2 independently, is N(R);
each R, independently, is hydrogen or C1-4alkyl;
each of Z1 and Z2 is a group
in which:
n is 1 or 2
X, or each X independently, is an electron withdrawing group; and
Y is a halogen atom,
which process comprises reacting a chromophoric compound of the formula (XX)
H(R)N-D-N(R)H (XX)
wherein each of D and R is as defined above, with at least two moles, per mole of the chromophoric compound of the formula (XX) , of a dihalobenzene component comprising at least one dihalobenzene compound of the formula (XXI)
wherein each of X, Y and n is as defined above, to obtain the dye of the formula (VIII)1.
79. A process for preparing a dye of the formula (VIII)1
Z1-L1-D-L2-Z2 (VIII)2
wherein:
D is a chromophore;
each of L1 and L2 independently, is N(R);
each R, independently, is hydrogen or C1-4alkyl;
Z1 is a group
in which:
n is 1 or 2
X, or each X independently, is an electron withdrawing group; and
Y is a halogen atom; and
Z2 is selected from the groups (II) and (III) given and defined in claim 1;
which process comprises reacting a chromophoric compound of the formula (XX)
H(R)N—D—N(R)H (XX)
wherein each of D and R is as defined above, with one mole, per mole of the chromophoric compound of the formula (XX), of each of
(a) a dihalobenzene compound of the formula (XXI)
wherein each of Z, X, Y and n is as defined above; and
(b) a compound selected from
wherein:
m is 1 or 2; p is 0 or 1; when m is 1, p is 1; and when m is 2, p is 0;
Y1, or each Y1 independently, is a halogen atom or an optionally substituted pyridinium group; and
T is C1-4alkoxy, C1-4thioalkoxy or N(R1) (R2), in which each of R1 and R2 independently is hydrogen, optionally substituted C1-4alkyl or optionally substituted aryl;
x is 1, 2 or 3; y is zero, 1 or 2; and x+y≦3;
Y2, or each Y2 independently, is a halogen atom or an optionally substituted pyridinium group;
U or each U independently, is C1-4alkyl or C1-4alkylsulphonyl; and
Y is as defined above;
the said reactions of the compound of the formula (XX) with each of the respective compounds of the formulae (XII) and (XIII) being carried out simultaneously or one before the other, in either order, to obtain a dye of the formula (VIII)2.
80. A process for preparing a dye of the formula (VIII)3
Z1-L1-D-Z2 (VIII)3
wherein:
D is a chromophore
L1 is N(R), in which R is hydrogen or C1-4alkyl;
Z1 is a group
in which n is 1 or 2; X, or each X independently, is an electron withdrawing group; and Y is a hydrogen atom; and Z2 is a group of the formula (IV)
—SO2CH2CH2X1 (IV)
wherein X1 is an eliminatable group;
a group of the formula (V)
—SO2(CH2)zCH═CH2 (V)
wherein z is zero or 1; and
a group of formula (VI)
—W—C(R10)═CH2 (VI)
wherein:
R10 is hydrogen, C1-4alkyl or halogen; and
W is —OC(═O)— or —N(R11)C(═O)—
in which R11 is hydrogen or C1-4alkyl;
which process comprises reacting a chromophoric compound of the formula (XXIV)
H(R)N-D-Z2 (XXIV)
wherein D and Z2 are as defined above, with a dihalobenzene compound of the formula (XXI)
wherein each of X, Y and n is as defined above, to obtain the dye of the formula (VIII)3.
81. A process for the colouration of a substrate, which process comprises applying to the substrate a dye according to any one of claims 1 to 77 .
82. A process according to claim 81 , wherein the dye is applied to the substrate by exhaust dyeing, padding or printing.
83. A process according to claim 82 , wherein the dye is applied by ink jet printing.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/117,279 US20030191293A1 (en) | 1997-11-12 | 2002-04-08 | Reactive dyes containing a halobenzene nucleus |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GBGB9723924.8A GB9723924D0 (en) | 1997-11-12 | 1997-11-12 | Reactive dyes containing a halobenzene nucleus |
| GB9723924.8 | 1997-11-12 | ||
| US09/554,325 US6399751B1 (en) | 1997-11-12 | 1998-11-12 | Reactive dyes containing a halobenzene nucleus |
| US10/117,279 US20030191293A1 (en) | 1997-11-12 | 2002-04-08 | Reactive dyes containing a halobenzene nucleus |
Related Parent Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/554,325 Division US6399751B1 (en) | 1997-11-12 | 1998-11-12 | Reactive dyes containing a halobenzene nucleus |
| PCT/GB1998/003406 Division WO1999027019A2 (en) | 1997-11-12 | 1998-11-12 | Reactive dyes containing a halobenzene nucleus |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20030191293A1 true US20030191293A1 (en) | 2003-10-09 |
Family
ID=10821975
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/554,325 Expired - Fee Related US6399751B1 (en) | 1997-11-12 | 1998-11-12 | Reactive dyes containing a halobenzene nucleus |
| US10/117,279 Abandoned US20030191293A1 (en) | 1997-11-12 | 2002-04-08 | Reactive dyes containing a halobenzene nucleus |
| US10/158,879 Abandoned US20030158395A1 (en) | 1997-11-12 | 2002-06-03 | Reactive dyes containing a halobenzene nucleus |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/554,325 Expired - Fee Related US6399751B1 (en) | 1997-11-12 | 1998-11-12 | Reactive dyes containing a halobenzene nucleus |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/158,879 Abandoned US20030158395A1 (en) | 1997-11-12 | 2002-06-03 | Reactive dyes containing a halobenzene nucleus |
Country Status (12)
| Country | Link |
|---|---|
| US (3) | US6399751B1 (en) |
| EP (2) | EP1333062A1 (en) |
| JP (1) | JP2001524570A (en) |
| KR (1) | KR20010032016A (en) |
| CN (2) | CN1491994A (en) |
| DE (1) | DE69823859T2 (en) |
| ES (1) | ES2221216T3 (en) |
| GB (1) | GB9723924D0 (en) |
| ID (1) | ID26617A (en) |
| PT (1) | PT1029002E (en) |
| TW (1) | TW508365B (en) |
| WO (1) | WO1999027019A2 (en) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101121827B (en) * | 2006-08-09 | 2010-07-14 | 明德国际仓储贸易(上海)有限公司 | Chemically-reactive dyes with thioalkyl-s-triazine reactive group |
| EP3296363B1 (en) * | 2010-11-30 | 2023-06-21 | Kemira Oyj | Azo dyes |
| CN102719113B (en) * | 2011-03-29 | 2014-09-03 | 上海汇友精密化学品有限公司 | Ink-jet ink with light resistance and preparation method thereof |
| CN106833017B (en) * | 2016-11-28 | 2018-09-11 | 浙江瑞华化工有限公司 | A kind of blue active dye composition and its preparation method and application |
| CN112111174B (en) * | 2020-09-27 | 2022-05-31 | 苏州澳缘盛新材料科技有限公司 | Preparation method of reactive dye blue |
| CN112175413B (en) * | 2020-09-27 | 2022-05-06 | 苏州澳缘盛新材料科技有限公司 | Reactive dye blue and application thereof |
| CN116144195B (en) * | 2022-12-12 | 2024-04-02 | 江苏德美科化工有限公司 | High-performance alkali-resistant yellow reactive dye and preparation method thereof |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB887870A (en) * | 1958-02-26 | 1962-01-24 | Cassella Farbwerke Mainkur Ag | New monoazo dyestuffs |
| GB882001A (en) * | 1959-09-16 | 1961-11-08 | Ici Ltd | New colouration process for cellulose textile materials |
| GB978162A (en) * | 1962-02-07 | 1964-12-16 | Ici Ltd | New dyestuffs containing pyridinium groups |
| FR1321960A (en) * | 1962-05-14 | 1963-03-22 | Acna | Reactive dyes |
| US3301847A (en) * | 1962-05-14 | 1967-01-31 | Acna | Reactive disazo dyestuffs |
-
1997
- 1997-11-12 GB GBGB9723924.8A patent/GB9723924D0/en not_active Ceased
-
1998
- 1998-11-12 ID IDW20000884A patent/ID26617A/en unknown
- 1998-11-12 EP EP03007521A patent/EP1333062A1/en not_active Withdrawn
- 1998-11-12 JP JP2000522167A patent/JP2001524570A/en not_active Withdrawn
- 1998-11-12 CN CNA021248893A patent/CN1491994A/en active Pending
- 1998-11-12 CN CN98811133A patent/CN1121456C/en not_active Expired - Fee Related
- 1998-11-12 US US09/554,325 patent/US6399751B1/en not_active Expired - Fee Related
- 1998-11-12 KR KR1020007005134A patent/KR20010032016A/en not_active Ceased
- 1998-11-12 DE DE69823859T patent/DE69823859T2/en not_active Expired - Fee Related
- 1998-11-12 ES ES98952935T patent/ES2221216T3/en not_active Expired - Lifetime
- 1998-11-12 EP EP98952935A patent/EP1029002B1/en not_active Expired - Lifetime
- 1998-11-12 PT PT98952935T patent/PT1029002E/en unknown
- 1998-11-12 WO PCT/GB1998/003406 patent/WO1999027019A2/en not_active Ceased
- 1998-12-29 TW TW087121801A patent/TW508365B/en active
-
2002
- 2002-04-08 US US10/117,279 patent/US20030191293A1/en not_active Abandoned
- 2002-06-03 US US10/158,879 patent/US20030158395A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| JP2001524570A (en) | 2001-12-04 |
| EP1029002B1 (en) | 2004-05-12 |
| ES2221216T3 (en) | 2004-12-16 |
| CN1121456C (en) | 2003-09-17 |
| DE69823859D1 (en) | 2004-06-17 |
| KR20010032016A (en) | 2001-04-16 |
| US20030158395A1 (en) | 2003-08-21 |
| EP1029002A2 (en) | 2000-08-23 |
| TW508365B (en) | 2002-11-01 |
| ID26617A (en) | 2001-01-25 |
| EP1333062A1 (en) | 2003-08-06 |
| WO1999027019A2 (en) | 1999-06-03 |
| GB9723924D0 (en) | 1998-01-07 |
| US6399751B1 (en) | 2002-06-04 |
| WO1999027019A3 (en) | 1999-07-15 |
| CN1491994A (en) | 2004-04-28 |
| WO1999027019A8 (en) | 1999-08-19 |
| CN1278848A (en) | 2001-01-03 |
| PT1029002E (en) | 2004-09-30 |
| DE69823859T2 (en) | 2005-04-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6458936B2 (en) | Fiber-reactive disazo compounds | |
| US6359121B1 (en) | Reactive dyes containing a linkage | |
| US6399751B1 (en) | Reactive dyes containing a halobenzene nucleus | |
| US5059681A (en) | Bis-azotriazinyl reactive dyes having an n-alkyl-phenylenediamine link | |
| JP2747500B2 (en) | Fiber reactive phthalocyanine dye | |
| CN1322063C (en) | Fiber-reactive mono-Azo dyes | |
| EP1230304B1 (en) | Azo dyestuffs | |
| JPH0931352A (en) | Dye mixture and its production | |
| CA2446784C (en) | Fibre reactive scarlet azo dyes | |
| EP1380621B1 (en) | Fibre reactive azo dyes | |
| KR20040045493A (en) | Organic compounds | |
| US6180765B1 (en) | Reactive diamine-linked azo dyes | |
| EP1506981B1 (en) | Fibre reactive azo dyes | |
| EP1311622A1 (en) | Fibre-reactive monoazo dyestuffs | |
| JPS62192466A (en) | Water-soluble disazo compound, manufacture and use as dyestuff | |
| US20050020820A1 (en) | Fiber-reactive azo dyestuffs | |
| KR0131760B1 (en) | Phthalocyanine compounds and methods for dyeing or printing organic substrates using the same | |
| WO2004026966A1 (en) | Dye mixtures of fibre-reactive azo dyes and use thereof for dyeing material containing hydroxy- and/or carboxamido groups | |
| WO1997027250A1 (en) | Dyes containing a 2,4,5-trifluoro-3,6-dicyanophenyl reactive group and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |