US11053265B2 - Optically active substituted 2,3-bisphosphinoquinoxalines and processes for producing the same - Google Patents
Optically active substituted 2,3-bisphosphinoquinoxalines and processes for producing the same Download PDFInfo
- Publication number
- US11053265B2 US11053265B2 US16/641,845 US201816641845A US11053265B2 US 11053265 B2 US11053265 B2 US 11053265B2 US 201816641845 A US201816641845 A US 201816641845A US 11053265 B2 US11053265 B2 US 11053265B2
- Authority
- US
- United States
- Prior art keywords
- alkyl
- group
- cycloalkyl
- general formula
- carbon atoms
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 0 CC.[1*]P([1*])C1=NC2=C(C=CC=C2)N=C1P([2*])C Chemical compound CC.[1*]P([1*])C1=NC2=C(C=CC=C2)N=C1P([2*])C 0.000 description 36
- WGIYRZRBKZOUSQ-UHFFFAOYSA-N CC.CC1=NC2=C(C=CC=C2)N=C1C Chemical compound CC.CC1=NC2=C(C=CC=C2)N=C1C WGIYRZRBKZOUSQ-UHFFFAOYSA-N 0.000 description 5
- VEZLVPXBIWEVFU-UHFFFAOYSA-N CB(C)B(C)C Chemical compound CB(C)B(C)C VEZLVPXBIWEVFU-UHFFFAOYSA-N 0.000 description 2
- WMHPGOJXAKRBNO-UHFFFAOYSA-N C=CC.CCCC.[H]C(C)(C)C Chemical compound C=CC.CCCC.[H]C(C)(C)C WMHPGOJXAKRBNO-UHFFFAOYSA-N 0.000 description 1
- ZWMSWLWWQGLWDT-UHFFFAOYSA-N C=CCCC1=CC=CC=C1.CCCCCC1=CC=CC=C1.[H]C(C)(C)CCC1=CC=CC=C1 Chemical compound C=CCCC1=CC=CC=C1.CCCCCC1=CC=CC=C1.[H]C(C)(C)CCC1=CC=CC=C1 ZWMSWLWWQGLWDT-UHFFFAOYSA-N 0.000 description 1
- OVTBFRIUQNPGJK-DEOSSOPVSA-N C[P@@](C1=NC2=C(C=CC=C2)N=C1P(C(C)(C)C)C(C)(C)C)C(C)(C)C Chemical compound C[P@@](C1=NC2=C(C=CC=C2)N=C1P(C(C)(C)C)C(C)(C)C)C(C)(C)C OVTBFRIUQNPGJK-DEOSSOPVSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/645—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having two nitrogen atoms as the only ring hetero atoms
- C07F9/6509—Six-membered rings
- C07F9/650952—Six-membered rings having the nitrogen atoms in the positions 1 and 4
- C07F9/650994—Six-membered rings having the nitrogen atoms in the positions 1 and 4 condensed with carbocyclic rings or carbocyclic ring systems
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/24—Phosphines, i.e. phosphorus bonded to only carbon atoms, or to both carbon and hydrogen atoms, including e.g. sp2-hybridised phosphorus compounds such as phosphabenzene, phosphole or anionic phospholide ligands
- B01J31/2404—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring
- B01J31/2442—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring comprising condensed ring systems
- B01J31/2447—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring comprising condensed ring systems and phosphine-P atoms as substituents on a ring of the condensed system or on a further attached ring
- B01J31/2452—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring comprising condensed ring systems and phosphine-P atoms as substituents on a ring of the condensed system or on a further attached ring with more than one complexing phosphine-P atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F5/00—Compounds containing elements of Groups 3 or 13 of the Periodic Table
- C07F5/02—Boron compounds
- C07F5/025—Boronic and borinic acid compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F5/00—Compounds containing elements of Groups 3 or 13 of the Periodic Table
- C07F5/02—Boron compounds
- C07F5/027—Organoboranes and organoborohydrides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2231/00—Catalytic reactions performed with catalysts classified in B01J31/00
- B01J2231/30—Addition reactions at carbon centres, i.e. to either C-C or C-X multiple bonds
- B01J2231/32—Addition reactions to C=C or C-C triple bonds
- B01J2231/323—Hydrometalation, e.g. bor-, alumin-, silyl-, zirconation or analoguous reactions like carbometalation, hydrocarbation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/10—Complexes comprising metals of Group I (IA or IB) as the central metal
- B01J2531/16—Copper
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B2200/00—Indexing scheme relating to specific properties of organic compounds
- C07B2200/07—Optical isomers
Definitions
- the present invention relates to an optically active 2,3-bisphosphinopyrazine derivative, a method for producing the same, a transition metal complex, an asymmetric catalyst, and a method for producing an organoboron compound.
- An optically active phosphine ligand having an asymmetric center on a phosphorus atom plays an important role in a catalytic asymmetric synthesis reaction using a transition metal complex.
- a 1,2-bis(dialkylphosphino)benzene derivative is proposed in Patent Literature 1.
- Patent Literature 2 a 2,3-bis(dialkylphosphino)pyrazine derivative is proposed.
- This pyrazine derivative is characterized in that the electron-withdrawing properties are extremely high due to the pyrazine skeleton, and thereby the electron density of the phosphorus atoms at the phosphine sites is low.
- a 2,3-bis(dialkylphosphino)pyrazine derivative represented by the following chemical formula (A) is proposed.
- a catalyst for a catalytic asymmetric synthesis reaction using a transition metal complex one having better catalytic activity is required.
- a hydroboration reaction generally, the reaction proceeds with anti-Markovnikov type regioselectivity.
- a hydroboration reaction showing Markovnikov type selectivity is limited to styrene type substrates, and there are few examples of reports on such hydroboration reactions of terminal alkenes substituted by alkyl groups.
- the 2,3-bis(dialkylphosphino)pyrazine derivative represented by the chemical formula (A) is used as a ligand, the substrate application range, selectivity, and yield are insufficient.
- the present invention provides an optically active 2,3-bisphosphinopyrazine derivative represented by the following general formula (1):
- R 1 represents a group selected from a branched alkyl group having 3 or more carbon atoms, an adamantyl group, an optionally substituted cycloalkyl group, and an optionally substituted aryl group;
- R 2 represents a group selected from a branched alkyl group having 3 or more carbon atoms, an adamantyl group, and an optionally substituted cycloalkyl group, provided that when R 1 is a tert-butyl group, R 1 and R 2 are not the same;
- R 3 represents a monovalent substituent
- n represents an integer of 0 to 4
- * represents an asymmetric center on a phosphorus atom.
- R 1 is as defined in the general formula (1), to act on a 2,3-dihalogenopyrazine derivative represented by the following general formula (2):
- R 3 and n are as defined in the general formula (1); and X represents a halogen atom, to perform an aromatic nucleophilic substitution reaction (1), then performing a deboranation reaction (1) to obtain a phosphinopyrazine derivative represented by the following general formula (4):
- R 2 and * are as defined in the general formula (1), to act on the phosphinopyrazine derivative to perform a nucleophilic substitution reaction (2), and then performing a deboranation reaction (2).
- the present invention provides a method for producing an optically active 2,3-bisphosphinopyrazine derivative represented by the following general formula (1′):
- R 1 , R 3 , n, and * are as defined in the general formula (1);
- R 2′ represents a group selected from a branched alkyl group having 3 or more carbon atoms, an adamantyl group, and an optionally substituted cycloalkyl group, comprising allowing a deprotonation product of a phosphine-borane represented by the following general formula (3):
- R 2′ is as defined in the general formula (1′; and * is as defined in the general formula (1), and a deboranating agent with a base to perform a reaction.
- the present invention provides an asymmetric catalyst comprising the transition metal complex.
- the present invention provides a method for producing an organoboron compound, comprising subjecting an alkene compound represented by the following general formula (6):
- R represents a hydrogen atom, an alkyl group, a substituted alkyl group, a cycloalkyl group, a substituted cycloalkyl group, an aralkyl group, a substituted aralkyl group, an aryl group, a substituted aryl group, an aliphatic heterocyclic group, a substituted aliphatic heterocyclic group, an aromatic heterocyclic group, a substituted aromatic heterocyclic group, an alkoxy group, a substituted alkoxy group, an aralkyloxy group, a substituted aralkyloxy group, an aryloxy group, a substituted aryloxy group, an alkyloxycarbonyl group, an aralkyloxycarbonyl group, a substituted silyl group or a substituted silyloxy group, an amino group, a substituted amino group, an alkylaminocarboxy group, a substituted alkylaminocarboxy group, an aryla
- Z represents the same or a different atom having a lone electron pair, and a dotted line connecting neighboring Z to each other indicates that another atom is bonded to Z; and neighboring Z optionally forms a ring with each other via another atom, in the presence of the asymmetric catalyst, the organoboron compound being represented by the following general formula (8):
- R and t are as defined in the general formula (6); Z is as defined in the general formula (7); and * indicates an asymmetric carbon atom.
- R 1 represents a group selected from a branched alkyl group having 3 or more carbon atoms, an adamantyl group, an optionally substituted cycloalkyl group, and an optionally substituted aryl group.
- Examples of the an optionally substituted cycloalkyl group represented by R 1 can include the groups described later as examples of the cycloalkyl group and the substituted cycloalkyl group represented by R in the formula of a general formula (6).
- Examples of the optionally substituted aryl group and the aryl group represented by R 1 can include the groups described later as examples of the optionally substituted aryl group and the aryl group represented by R in the formula of the general formula (6).
- R 2 represents a group selected from a branched alkyl group having 3 or more carbon atoms, an adamantyl group, and an optionally substituted cycloalkyl group.
- the branched alkyl group represented by R 2 is preferably one having 3 to 8 carbon atoms and particularly preferably one having 4 to 8 carbon atoms.
- the branched alkyl group represented by R 2 is preferably a tertiary alkyl group.
- R 1 and R 2 are not the same.
- the branched alkyl group having 3 or more carbon atoms represented by R 2 is one other than a tert-butyl group and particularly preferably a branched tertiary alkyl group having 5 to 8 carbon atoms.
- examples of a particularly preferred branched alkyl group having 3 or more carbon atoms represented by R 2 include an amyl group and a 1,1,3,3-tetramethylbutyl group.
- R 1 is a branched alkyl group having 3 or more carbon atoms other than a tert-butyl group, an adamantyl group, an optionally substituted cycloalkyl group, or an optionally substituted aryl group
- examples of a particularly preferred branched alkyl group having 3 or more carbon atoms represented by R 2 include a tert-butyl group, an amyl group, and a 1,1,3,3-tetramethylbutyl group.
- Examples of the optionally substituted cycloalkyl group represented by R 2 can include the groups described later as examples of the cycloalkyl group and the substituted cycloalkyl group represented by R in the formula of the general formula (6).
- R 1 is preferably a group selected from a tert-butyl group, an adamantyl group, a 1,1,3,3-tetramethylbutyl group, and an optionally substituted aryl group, and particularly preferred combinations with R 2 will be shown below.
- R 1 is a tert-butyl group in the general formula (1)
- R 2 is preferably a 1,1,3,3-tetramethylbutyl group or an adamantyl group from the viewpoint of being able to effectively shield the space of a reaction field.
- R 1 is an adamantyl group, a 1,1,3,3-tetramethylbutyl group, or an optionally substituted aryl group, in the general formula (1), similarly, the space of a reaction field can be effectively shielded.
- R 2 is preferably a tert-butyl group, a 1,1,3,3-tetramethylbutyl group, or an adamantyl group.
- R 1 is a group other than a tert-butyl group, especially when R 1 is an adamantyl group or a 1,1,3,3-tetramethylbutyl group
- R 2 is preferably a tert-butyl group, a 1,1,3,3-tetramethylbutyl group, or an adamantyl group.
- R 1 is an adamantyl group
- R 2 is particularly preferably a tert-butyl group, a 1,1,3,3-tetramethylbutyl group, or an adamantyl group.
- R 3 represents a monovalent substituent.
- R 3 is not particularly limited as long as it is a monovalent substituent.
- examples of R 3 include an alkyl group linear or branched and having 1 to 5 carbon atoms, a nitro group, an amino group, a hydroxyl group, an alkoxy group, a fluoro group, a chloro group, a bromo group, and an iodo group.
- n in the general formula (1) represents an integer of 0 to 4, and * represents an asymmetric center on a phosphorus atom.
- the electron density of the P atoms at the phosphine sites is lowered due to the electron-withdrawing properties due to the pyrazine skeleton.
- the phosphine sites are inert to oxidation by air, and the storage stability is high.
- the electron density decreasing properties at the phosphine sites do not impair catalytic activity.
- Examples of specific compounds of the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) include 2-[(1S)-(1-adamantyl)(methyl)phosphanyl]-3-(di-tert-butylphosphanyl)quinoxaline, (S)-2-(di-tert-butylphosphanyl)-3-[methyl(2,4,4-trimethylpentan-2-yl)phosphanyl]quinoxaline, 2-[(diadamantan-1-yl)phosphanyl]-3-[(R)-tert-butyl(methyl)phosphanyl]quinoxaline, (S)-2-(adamantan-1-yl(methyl)phosphanyl)-3-[(diadamantan-1-yl)phosphanyl]quinoxaline, and 2-[(diadamantan-1-yl)phosphanyl]-3-[(S)-methyl(
- a first preferred method for producing the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) comprises deprotonating the phosphine-borane represented by the general formula (3), allowing the deprotonation product produced thereby to act on the 2,3-dihalogenopyrazine derivative represented by the general formula (2) to perform a nucleophilic substitution reaction (1), then performing a deboranation reaction (1) to obtain the phosphinopyrazine derivative represented by the general formula (4), then allowing a deprotonation product of the optically active phosphine-borane represented by the general formula (5) to act on the phosphinopyrazine derivative represented by the general formula (4) to perform a nucleophilic substitution reaction (2), and then performing a deboranation reaction (2).
- the method for producing the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) according to the present invention has the following four steps:
- R 1 , R 3 , X, and n are as defined in the general formula (1).
- the 2,3-dihalogenopyrazine derivative according to the A liquid is represented by the following general formula (2):
- R 3 and n are as defined in the general formula (1); and X represents a halogen atom.
- X in the formula of the general formula (2) is a halogen atom, and examples of the halogen atom include a chlorine atom, a bromine atom, and an iodine atom. Among these, as X, a chlorine atom is preferred.
- R 3 and n in the formula of the general formula (2) correspond to R 3 and n in the formula of the optically active 2,3-bisphosphinopyrazine derivative of the general formula (1), and R 3 represents a monovalent substituent, and n represents an integer of 0 to 4.
- the B liquid is a solution comprising a phosphineborane compound obtained by deprotonating a phosphine-borane.
- the phosphine-borane according to the B liquid is represented by the following general formula (3):
- R 1 in the formula of the general formula (3) corresponds to R 1 in the formula of the optically active 2,3-bisphosphinopyrazine derivative of the general formula (1).
- R 1 in the formula (3) represents a group selected from a branched alkyl group having 3 or more carbon atoms, an adamantyl group, an optionally substituted cycloalkyl group, and an optionally substituted aryl group, and specific examples thereof and preferred groups are as described above for R 1 in the formula.
- the phosphine-borane represented by the general formula (3) can be produced by a known method.
- Examples of the method for producing the phosphine-borane represented by the general formula (3) include Japanese Patent Laid-Open No. 2001-253889, Japanese Patent Laid-Open No. 2003-300988, Japanese Patent Laid-Open No. 2007-70310, Japanese Patent Laid-Open No. 2010-138136, and J. Org. Chem, 2000, vol. 65, P4185-4188.
- the phosphine-borane represented by the general formula (3) is dissolved in a solvent, and then a base is added.
- a base is added.
- a solvent inert to the phosphine-borane represented by the general formula (3) and the phosphine compound produced from the phosphine-borane by deprotonation can be used without particular limitation.
- the solvent include tetrahydrofuran, N,N-dimethylformamide, diethyl ether, tert-butyl methyl ether, cyclopentyl methyl ether, dioxane, hexane, and toluene. These solvents can be used as a single or mixed solvent.
- the amount of the base added be in the range of 1.0 to 1.5, particularly 1.0 to 1.2, in the molar ratio of the base to the phosphine-borane represented by the general formula (3).
- the temperature at which the base is added be ⁇ 20 to 20° C., particularly ⁇ 20 to 0° C.
- reaction in the first step it is possible to add the A liquid to the B liquid or add the B liquid to the A liquid to perform the nucleophilic substitution reaction (1) to obtain the phosphinopyrazine-borane derivative represented by the general formula (9).
- the A liquid or the B liquid be added so that the molar ratio of the phosphine compound obtained by deprotonation from the phosphine-borane represented by the general formula (3) to the 2,3-dihalogenopyrazine derivative represented by the general formula (2) in the A liquid is in the range of 1.0 to 2.0, particularly 1.0 to 1.5.
- the rate at which the A liquid or the B liquid is added is preferably a constant rate from the viewpoint of obtaining a product having stable quality.
- the temperature at which the A liquid or the B liquid is added be ⁇ 20 to 50° C., particularly ⁇ 20 to 5° C.
- an aging reaction can be continuously performed as needed, in order to complete the nucleophilic substitution reaction (1). It is preferred from the viewpoint of the reaction rate and the purity of the obtained target that the reaction temperature when this aging reaction is performed be ⁇ 20 to 80° C., particularly 0 to 50° C.
- the second step is the step of subjecting the phosphinopyrazine-borane derivative represented by the general formula (9) obtained in the first step to the deboranation reaction (1) with a deboranating agent in a solvent to obtain a phosphinopyrazine derivative represented by the following general formula (4):
- R 1 , R 3 , and n are as defined in the general formula (1); and X is as defined in the general formula (2).
- boranating agent examples include N,N,N′,N′-tetramethylethylenediamine (TMEDA), triethylenediamine (DABCO), triethylamine, HBF 4 , and trifluoromethanesulfonic acid, and the boranating agent is preferably TMEDA.
- the amount of the deboranating agent added is usually 2 to 20 equivalents, preferably 3 to 10 equivalents, based on the phosphinopyrazine-borane derivative represented by the general formula (9).
- a solvent that can dissolve the phosphinopyrazine-borane derivative represented by the general formula (9) and is inert to the phosphinopyrazine-borane derivative and the phosphinopyrazine derivative represented by the general formula (4) produced can be used without particular limitation.
- the solvent include ethyl acetate, tetrahydrofuran, N,N-dimethylformamide, diethyl ether, tert-butyl methyl ether, cyclopentyl methyl ether, dioxane, hexane, and toluene, and these solvents can be used as a single or mixed solvent.
- the reaction temperature of the deboranation reaction (1) be preferably ⁇ 20 to 80° C., more preferably ⁇ 20 to 50° C.
- the reaction time of the deboranation reaction (1) is preferably 30 min or more, more preferably 1 to 5 h.
- the third step is the step of allowing a deprotonation product of the optically active phosphine-borane represented by the general formula (5) to act on the phosphinopyrazine derivative represented by the general formula (4) obtained in the second step to perform the nucleophilic substitution reaction (2) to obtain an optically active phosphinopyrazine-borane derivative represented by the following general formula (10):
- R 1 , R 2 , R 3 , n, and * are as defined in the general formula (1).
- a liquid comprising the phosphinopyrazine derivative represented by the general formula (4) (hereinafter referred to as a “C liquid”) is prepared, and apart from the C liquid, a liquid in which the optically active phosphine-borane represented by the general formula (5) is deprotonated (hereinafter referred to as a “D liquid”) is prepared.
- the C liquid may be a solution or a slurry.
- a solvent that can dissolve the phosphinopyrazine derivative represented by the general formula (4) and is inert to the phosphinopyrazine derivative represented by the general formula (4) is preferably used.
- the solvent include tetrahydrofuran, 1,2-dimethoxyethane, N,N-dimethylformamide, diethyl ether, tert-butyl methyl ether, cyclopentyl methyl ether, dioxane, hexane, and toluene. These solvents can be used as a single or mixed solvent. In addition, it is not always necessary to completely dissolve the phosphinopyrazine derivative represented by the general formula (4), and the reaction can be initiated even from a slurry state.
- the D liquid is a solution comprising an optically active phosphineborane compound obtained by deprotonating the optically active phosphine-borane represented by the general formula (5).
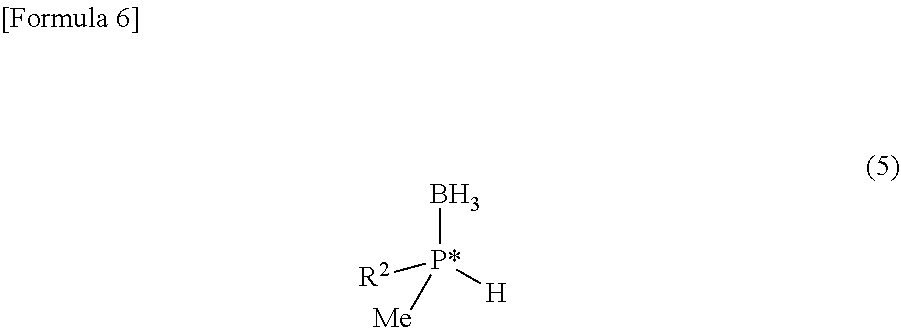
- optically active phosphine-borane is represented by the following general formula (5):
- R 2 in the formula of the general formula (5) corresponds to R 2 in the formula of the optically active 2,3-bisphosphinopyrazine derivative of the general formula (1).
- R 2 in the formula (5) represents a group selected from a branched alkyl group having 3 or more carbon atoms, an adamantyl group, and an optionally substituted cycloalkyl group, on the condition that when R 1 in the formula (4) is a tert-butyl group, R 2 in the formula (5) is a group other than a tert-butyl group.
- Examples of R 2 in the formula (5) and preferred R 2 in the formula (5) include those described above for R 2 in the formula (1).
- branched alkyl group having 3 or more carbon atoms represented by R 2 in the formula (5) branched tertiary alkyl groups having 4 to 8 carbon atoms are preferred.
- R 1 in the formula (4) is a group other than a tert-butyl group
- examples of the branched alkyl group having 3 or more carbon atoms represented by R 2 in the formula (5) include a tert-butyl group, an amyl group, and a 1,1,3,3-tetramethylbutyl group.
- examples of the branched alkyl group having 3 or more carbon atoms represented by R 2 in the formula (5) include an amyl group and a 1,1,3,3-tetramethylbutyl group.
- the optically active phosphine-borane represented by the general formula (5) can be produced by a known method.
- Examples of the method for producing the phosphine-borane represented by the general formula (5) include Japanese Patent Laid-Open No. 2001-253889, Japanese Patent Laid-Open No. 2003-300988, Japanese Patent Laid-Open No. 2007-70310, Japanese Patent Laid-Open No. 2010-138136, and J. Org. Chem, 2000, vol. 65, P4185-4188.
- the optically active phosphine-borane represented by the general formula (5) is dissolved in a solvent, and then a base is added.
- a base is added.
- the amount of the base added be in the range of 1.0 to 2.0, particularly 1.0 to 1.5, in the molar ratio of the base to the optically active phosphine-borane represented by the general formula (5).
- the temperature at which the base is added be ⁇ 20 to 20° C., particularly ⁇ 20 to 0° C., from the viewpoint of being able to deprotonate the optically active phosphine-borane represented by the general formula (5) while maintaining its optical purity.
- the C liquid or the D liquid be added so that the molar ratio of the optically active phosphine compound obtained by deprotonation from the optically active phosphine-borane represented by the general formula (5) to the phosphinopyrazine derivative represented by the general formula (4) in the C liquid is in the range of 1.0 to 2.0, particularly 1.0 to 1.5.
- the C liquid or the D liquid is preferably added at a constant rate from the viewpoint of obtaining a product having stable quality.
- R 1 , R 2 , R 3 , n, and * are as defined above.
- boranating agent examples include N,N,N′,N′-tetramethylethylenediamine (TMEDA), triethylenediamine (DABCO), triethylamine, HBF 4 , and trifluoromethanesulfonic acid, and the boranating agent is preferably TMEDA.
- the amount of the deboranating agent added is usually 2 to 20 equivalents, preferably 3 to 10 equivalents, based on the optically active phosphinopyrazine-borane derivative represented by the general formula (10).
- a solvent that can dissolve the phosphinopyrazine-borane derivative represented by the general formula (10) and is inert to the phosphinopyrazine-borane derivative and the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) produced can be used without particular limitation.
- solvent examples include ethyl acetate, tetrahydrofuran, 1,2-dimethoxyethane, N,N-dimethylformamide, diethyl ether, tert-butyl methyl ether, cyclopentyl methyl ether, dioxane, hexane, and toluene, and these solvents can be used as a single or mixed solvent.
- the Ath step is the step of adding a base to a liquid comprising the phosphinopyrazine derivative represented by the general formula (4) obtained in the second step, the optically active phosphine-borane represented by the general formula (5), and a deboranating agent (hereinafter referred to as a “Y liquid”) to obtain the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) at once.
- the second production method allows both R 1 and R 2 to be t-butyl.
- the general formula (1) when both R 1 and R 2 are allowed to be t-butyl will be described as a general formula (1′), and the general formula (5) will be described as a general formula (5′).
- the general formula (1) and the general formula (1′) are the same except that the former does not include the case where both R 1 and R 2 are t-butyl, and the latter includes the case.
- the content of the phosphinopyrazine derivative represented by the general formula (4) in the Y liquid is 1 to 50% by mass, preferably 5 to 20% by mass, based on the total amount of the Y liquid.
- optically active phosphine-borane represented by the general formula (5′) one the same as the optically active phosphine-borane represented by the general formula (5) used in the third step except that the case where both R 1 and R 2 are t-butyl as described above is allowed as described above can be used. It is preferred from the viewpoint of reactivity and economy that the optically active phosphine-borane represented by the general formula (5′) be added so that the amount of the optically active phosphine-borane represented by the general formula (5′) added is in the range of 1.0 to 2.0, particularly 1.0 to 1.5, in the molar ratio to the phosphinopyrazine derivative represented by the general formula (4).
- the amount of the deboranating agent added is usually preferably 2 to 20 equivalents, more preferably 3 to 10 equivalents, based on the phosphinopyrazine derivative represented by the general formula (4).
- the Y liquid is a liquid in which the phosphinopyrazine derivative represented by the general formula (4), the optically active phosphine-borane represented by the general formula (5′), and a deboranating agent are dissolved or dispersed in a solvent.
- the solvent used in the Y liquid is not particularly limited as long as it is a solvent that can dissolve or disperse the phosphinopyrazine derivative represented by the general formula (4) and is inert to the phosphinopyrazine derivative represented by the general formula (4).
- the solvent that can be used for the preparation of the Y liquid include tetrahydrofuran, 1,2-dimethoxyethane, N,N-dimethylformamide, diethyl ether, tert-butyl methyl ether, cyclopentyl methyl ether, dioxane, hexane, and toluene. These solvents can be used as a single or mixed solvent.
- Examples of the base according to the Ath step include n-butyllithium, lithium diisopropylamide, methylmagnesium bromide, potassium-tert-butoxide, sodium-tert-butoxide, Hunig's base, potassium hydroxide, and sodium hydroxide.
- potassium-tert-butoxide is preferred in that a product having an excellent reaction yield and excellent quality is obtained.
- the Y liquid and the base are mixed to perform a reaction to obtain an optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1′).
- adding the base to the Y liquid is preferred.
- a solution in which the base is dissolved in a solvent hereinafter referred to as a “Z liquid”
- Z liquid a solution in which the base is dissolved in a solvent
- adding the Z liquid to the Y liquid is preferred in that the reaction is easily controlled, and a product having stable quality is easily obtained.
- the content of the base in the Z liquid is not particularly limited, but it is preferred in terms of high reactivity and productivity that the content is 1 to 50% by mass, preferably 5 to 30% by mass, based on the total amount of the Z liquid.
- the solvent used in the Z liquid is not particularly limited as long as it is a solvent that can dissolve the base and is inert.
- the solvent according to the Z liquid include tetrahydrofuran, 1,2-dimethoxyethane, N,N-dimethylformamide, diethyl ether, tert-butyl methyl ether, cyclopentyl methyl ether, dioxane, hexane, and toluene. These solvents are used as a single or mixed solvent.
- the amount of the base added to the Y liquid is 1.0 to 1.5 mol, preferably 1.0 to 1.2 mol, based on 1 mol of the optically active phosphine-borane represented by the general formula (5′) in the Y liquid.
- the rate at which the base is added to the Y liquid is not particularly limited as long as the reaction heat can be controlled in a range in which no side reactions occur. But, the rate at which the base is added to the Y liquid is preferably a constant rate in that a product having stable quality is obtained.
- the solid base is desirably added in portions while the state of the reaction heat is observed.
- the temperature of the Y liquid (the temperature of the reaction liquid) when the base is added to the Y liquid is preferably ⁇ 25 to 50° C. in that it is industrially advantageous, and particularly preferably ⁇ 25 to 20° C. in that a product having high optical purity is obtained in high yield.
- the aging temperature when aging is performed is preferably ⁇ 25 to 80° C. in that it is industrially advantageous, and particularly preferably ⁇ 25 to 30° C. in that a product having high optical purity is obtained in high yield.
- the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) according to the present invention, as a ligand, can form a complex with a transition metal.
- This transition metal complex is useful as an asymmetric synthesis catalyst.
- Examples of asymmetric synthesis include the hydroboration reactions of olefins, Enantioselective Substitution of Allylic Carbonates with Diboron, the synthesis of optically active piperidine and tetrahydroquinoline derivative compounds by Dearomatization/Borylation of Pyridines, enantioselective borylation reactions to allyl acetal derivatives and allyl ketal derivatives, the asymmetric hydrogenation reactions of dehydroamino acids, and asymmetric coupling reactions, asymmetric hydrosilylation reactions, and asymmetric Michael reactions involving C—C bonds and C—N bonds.
- transition metal examples include rhodium, ruthenium, iridium, palladium, nickel, iron, and copper, and the transition metal is preferably a rhodium metal, a palladium metal, or a copper metal.
- the method for producing the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) is as described above, but in the production method having the above-described first step to fourth step, instead of previously allowing the phosphine-borane represented by the formula (3) deprotonated, to act on the 2,3-dihalogenopyrazine derivative represented by the general formula (2), the optically active phosphine-borane represented by the formula (5) deprotonated may be previously allowed to act to perform an aromatic nucleophilic substitution reaction and a deboranation reaction.
- the phosphine-borane represented by the formula (3) deprotonated is allowed to act on the obtained phosphinopyrazine derivative to perform an aromatic nucleophilic substitution reaction and a deboranation reaction to obtain the optically active 2,3-bisphosphinopyrazine derivative represented by the formula (1).
- the phosphine-borane represented by the formula (3) deprotonated be previously allowed to act, as described above, because the optically active 2,3-bisphosphinopyrazine derivative represented by the formula (1) can be obtained with the optical purity increased.
- the phosphine-borane represented by the formula (3) deprotonated be previously allowed to act, and then the optically active phosphine-borane represented by the formula (5′) be allowed to act, because the optically active 2,3-bisphosphinopyrazine derivative represented by the formula (1′) can be obtained with the optical purity increased.
- a rhodium complex can be produced by reacting the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) with bis(cyclooctane-1,5-diene)rhodium hexafluoroantimonate, bis(cyclooctane-1,5-diene)rhodium tetrafluoroborate, or the like.
- a palladium complex can be produced, for example, by reacting the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) with n-allylpalladium chloride, for example, according to a method described in “Y. Uozumi and T. Hayashi, J. Am. Chem. Soc., 1991, 113, 9887.”
- a complex can be easily produced by mixing the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) and Cu(OtBu) in a solvent.
- a complex can also be produced by mixing a copper(I) salt and KOtBu in a solvent in the coexistence of a 2,3-bisphosphinopyrazine derivative.
- the copper(I) salt include CuF, CuCl, CuBr, CuI, CuPF 6 , CuBPh 4 , CuBF 4 , CuOAc, and CuBF 4 (MeCN) 4 .
- a monovalent Cu ion and the derivative represented by the general formula (1) usually form a complex in a molar ratio of 1:1.
- the copper metal complex obtained by forming the complex with a copper metal using the optically active 2,3-bisphosphinopyrazine derivative represented by the general formula (1) as a ligand is useful particularly as an asymmetric catalyst for various borylation reactions such as a hydroboration reaction.
- the alkene compound represented by the general formula (6) is subjected to a coupling reaction, using the copper metal complex of the present invention as an asymmetric catalyst (hereinafter sometimes simply referred to as an “asymmetric catalyst”), with the diboron compound represented by the general formula (7) in the presence of the asymmetric catalyst to produce the organoboron compound represented by the general formula (8).
- an asymmetric catalyst hereinafter sometimes simply referred to as an “asymmetric catalyst”
- the alkene compound that is a starting raw material in the method for producing an organoboron compound according to the present invention is represented by the following general formula (6):
- R represents a hydrogen atom, an alkyl group, a substituted alkyl group, a cycloalkyl group, a substituted cycloalkyl group, an aralkyl group, a substituted aralkyl group, an aryl group, a substituted aryl group, an aliphatic heterocyclic group, a substituted aliphatic heterocyclic group, an aromatic heterocyclic group, a substituted aromatic heterocyclic group, an alkoxy group, a substituted alkoxy group, an aralkyloxy group, a substituted aralkyloxy group, an aryloxy group, a substituted aryloxy group, an alkyloxycarbonyl group, an aralkyloxycarbonyl group, a substituted silyl group or a substituted silyloxy group, an amino group, a substituted amino group, an alkylaminocarboxy group, a substituted alkylaminocarboxy group, an aryla
- Examples of the aralkyl group represented by R in the formula of the general formula (6) include aralkyl groups having 7 to 12 carbon atoms. Specific examples include a benzyl group, a 2-phenylethyl group, a 1-phenylpropyl group, a 2-phenylpropyl group, a 3-phenylpropyl group, a 1-phenylbutyl group, a 2-phenylbutyl group, a 3-phenylbutyl group, a 4-phenylbutyl group, a 1-phenylpentyl group, a 2-phenylpentyl group, a 3-phenylpentyl group, a 4-phenylpentyl group, a 5-phenylpentyl group, a 1-phenylhexyl group, a 2-phenylhexyl group, a 3-phenylhexyl group, a 4-phenylhexyl group, a 5-phenylhexyl
- Examples of the aryl group represented by R in the formula of the general formula (6) include aryl groups having 6 to 18 carbon atoms. Specific examples include a phenyl group, a methylphenyl group, an ethylphenyl group, a dimethylphenyl group, a naphthyl group, an anthracenyl group, and a phenanthrenyl group.
- aliphatic heterocyclic group represented by R in the formula of the general formula (6) for example, five-membered or six-membered aliphatic heterocyclic groups are preferred, and examples of the aliphatic heterocyclic group include aliphatic heterocyclic groups comprising one to three heteroatoms, for example, nitrogen atoms, oxygen atoms, or sulfur atoms, as different atoms. Specific examples include a pyrrolidyl-2-one group, a piperidino group, a piperazinyl group, a morpholino group, a tetrahydrofuryl group, and a tetrahydropyranyl group.
- aromatic heterocyclic group represented by R in the formula of the general formula (6) for example, five-membered or six-membered monocyclic aromatic heterocyclic groups and polycyclic aromatic heterocyclic groups are preferred, and examples of the aromatic heterocyclic group include aromatic heterocyclic groups comprising one to three heteroatoms, for example, nitrogen atoms, oxygen atoms, or sulfur atoms, as different atoms. Specific examples include a pyridyl group, an imidazolyl group, a thiazolyl group, a furfuryl group, a pyranyl group, a furyl group, a benzofuryl group, and a thienyl group.
- Examples of the aryloxy group represented by R in the formula of the general formula (6) include aryloxy groups having 6 to 14 carbon atoms. Specific examples include a phenyloxy group, a naphthyloxy group, and an anthryloxy group.
- Examples of the substituted alkyl group represented by R in the formula of the general formula (6) include alkyl groups in which at least one hydrogen atom of the above alkyl groups is replaced by a substituent such as an alkyl group, a cycloalkyl group, an alkoxy group, a halogen atom, an amino group, or an amino group having a protecting group.
- Examples of the substituted aliphatic heterocyclic group represented by R in the formula of the general formula (6) include aliphatic heterocyclic groups in which at least one hydrogen atom of the above aliphatic heterocyclic groups is replaced by a substituent such as an alkyl group, a cycloalkyl group, an alkyl halide group, an alkoxy group, or a halogen atom.
- Examples of the substituted alkoxy group represented by R in the formula of the general formula (6) include alkoxy groups in which at least one hydrogen atom of the above alkoxy groups is replaced by a substituent such as an alkyl group, a cycloalkyl group, an alkoxy group, a halogen atom, an amino group, or an amino group having a protecting group.
- Examples of the substituted aralkyloxy group represented by R in the formula of the general formula (6) include aralkyloxy groups in which at least one hydrogen atom of the above aralkyloxy groups is replaced by a substituent such as an alkyl group, a cycloalkyl group, an alkyl halide group, an alkoxy group, a halogen atom, an amino group, or an alkyl group-substituted amino group.
- Examples of the substituted aryloxy group represented by R in the formula of the general formula (6) include aryloxy groups in which at least one hydrogen atom of the above aryloxy groups is replaced by a substituent such as an alkyl group, a cycloalkyl group, an alkyl halide group, an alkoxy group, a halogen atom, an amino group, or an alkyl group-substituted amino group, or aryloxy groups in which two adjacent hydrogen atoms of the above aryloxy groups are replaced by an alkylenedioxy group or the like.
- a substituent such as an alkyl group, a cycloalkyl group, an alkyl halide group, an alkoxy group, a halogen atom, an amino group, or an alkyl group-substituted amino group, or aryloxy groups in which two adjacent hydrogen atoms of the above aryloxy groups are replaced by an alkylenedioxy group or the like.
- Examples of the substituted silyl group represented by R in the formula of the general formula (6) include silyl groups in which at least one hydrogen atom of a silyl group is replaced by a substituent such as an alkyl group, a cycloalkyl group, an alkyl halide group, an alkoxy group, a halogen atom, an amino group, an alkyl group-substituted amino group, or an aryl group.
- a substituent such as an alkyl group, a cycloalkyl group, an alkyl halide group, an alkoxy group, a halogen atom, an amino group, an alkyl group-substituted amino group, or an aryl group.
- Examples of the substituted silyloxy group represented by R in the formula of the general formula (6) include silyloxy groups in which at least one hydrogen atom of a silyloxy group is replaced by a substituent such as an alkyl group, a cycloalkyl group, an alkyl halide group, an alkoxy group, a halogen atom, an amino group, an alkyl group-substituted amino group, or an aryl group.
- a substituent such as an alkyl group, a cycloalkyl group, an alkyl halide group, an alkoxy group, a halogen atom, an amino group, an alkyl group-substituted amino group, or an aryl group.
- Examples of the alkyl group, the cycloalkyl group, and the alkoxy group as the substituents include the examples of the alkyl group, the cycloalkyl group, and the alkoxy group represented by R described above, respectively.
- Examples of the halogen atom include a fluorine atom, a chlorine atom, a bromine atom, and an iodine atom.
- alkylenedioxy group examples include alkylenedioxy groups having 1 to 3 carbon atoms, and specific examples include a methylenedioxy group, an ethylenedioxy group, a propylenedioxy group, and a trimethylenedioxy group.
- alkyl halide group examples include alkyl halide groups having 1 to 6 carbon atoms in which the above alkyl groups are halogenated (for example, fluorinated, chlorinated, brominated, or iodinated). Specific examples include a chloromethyl group, a bromomethyl group, a trifluoromethyl group, a 2-chloroethyl group, a 3-chloropropyl group, a 3-bromopropyl group, and a 3,3,3-trifluoropropyl group.
- halogenated for example, fluorinated, chlorinated, brominated, or iodinated.
- Specific examples include a chloromethyl group, a bromomethyl group, a trifluoromethyl group, a 2-chloroethyl group, a 3-chloropropyl group, a 3-bromopropyl group, and a 3,3,3-trifluoropropyl group.
- alkyl group-substituted amino group examples include amino groups in which one or two hydrogen atoms of an amino group are replaced by the above alkyl group(s) and/or the above cycloalkyl group(s).
- Specific examples of the alkyl group-substituted amino group include monosubstituted amino groups such as a methylamino group, an ethylamino group, a propylamino group, a butylamino group, a pentylamino group, and a hexylamino group, and disubstituted amino groups such as a dimethylamino group, a diethylamino group, a dipropylamino group, a dibutylamino group, a dipentylamino group, and a dihexylamino group.
- any one used as an amino-protecting group can be used, and examples thereof include those described in “PROTECTIVE GROUPS IN ORGANIC SYNTHESIS Second Edition (JOHN WILEY & SONS, INC.)” as amino-protecting groups.
- Specific examples of the amino-protecting group include an alkyl group, a cycloalkyl group, an aralkyl group, an acyl group, and an alkyloxycarbonyl group.
- Examples of the alkyl group, the cycloalkyl group, and the aralkyl group here include the examples of the alkyl group, the cycloalkyl group, and the alkoxy group represented by R described above, respectively.
- the acyl group may be linear or branched or cyclic.
- Examples of the acyl group include acyl groups derived from carboxylic acids and, for example, having 2 to 7 carbon atoms. Specific examples include an acetyl group, a propionyl group, a butyryl group, a pentanoyl group, a hexanoyl group, and a benzoyl group.
- Examples of the alkyloxycarbonyl group include a tert-butyloxycarbonyl group and a benzyloxycarbonyl group.
- amino group having a protecting group examples include amino groups protected by the above protecting groups.
- Specific examples of the amino group having a protecting group include an acetylamino group, a benzoylamino group, a tert-butyloxycarbonylamino group, a benzyloxycarbonylamino group, and a cyclic amino group.
- cyclic amino group examples include cyclic amines in which an alkylene chain such as a butylene group or a pentylene group, a —CH 2 CH 2 OCH 2 CH 2 —, —CH 2 CH 2 NHCH 2 CH 2 —, or —CH 2 CH 2 OCO— group, or the like is bonded to a nitrogen atom, and specific examples thereof include a morpholino group, a piperidino group, and a 1,3-oxazolin-2-on-1-yl group.
- Examples of the substituted amino group represented by R in the formula of the general formula (6) include those illustrated above as the amino group and the amino group having a protecting group.
- Examples of the alkylaminocarboxy group, the substituted alkylaminocarboxy group, and the arylaminocarboxy group represented by R in the formula of the general formula (6) include those represented by —CON(R m ) 2 (R m is a substituted or unsubstituted alkyl group having 1 to 8 carbon atoms, a substituted or unsubstituted aryl group having 6 to 12 carbon atoms, or a hydrogen atom, and at least one of R m is a substituted or unsubstituted alkyl group having 1 to 8 carbon atoms or a substituted or unsubstituted aryl group having 6 to 12 carbon atoms).
- Examples of the substituents in the substituted alkyl group and the substituted aryl group here include the same as those mentioned above as the examples of the substituents in the substituted alkyl group and the substituted aryl group represented by R.
- Examples of the alkyloxycarbonyloxy group represented by R in the formula of the general formula (6) can include methoxycarbonyloxy, ethoxycarbonyloxy, n-propoxycarbonyloxy, isopropoxycarbonyloxy, n-butoxycarbonyloxy, isobutoxycarbonyloxy, tert-butoxycarbonyloxy, sec-butoxycarbonyloxy, n-pentyloxycarbonyloxy, neopentyloxycarbonyloxy, n-hexyloxycarbonyloxy, isohexyloxycarbonyloxy, a 3-methylpentyloxycarbonyloxy group, cyclopropyloxycarbonyloxy, cyclobutyloxycarbonyloxy, cyclopentyloxycarbonyloxy, cyclohexyloxycarbonyloxy, cycloheptyloxycarbonyloxy, and cyclooctyloxycarbonyloxy.
- Examples of the aryloxycarbonyloxy group represented by R in the formula of the general formula (6) can include phenoxycarbonyloxy, 1-naphthyloxycarbonyloxy, and 2-naphthyloxycarbonyloxy.
- Examples of the halogen atom represented by R in the formula of the general formula (6) include a fluorine atom, a chlorine atom, a bromine atom, and an iodine atom.
- the diboron compound that is a starting raw material in the method for producing an organoboron compound according to the present invention is represented by the following general formula (7):
- Z represents the same or a different atom having a lone electron pair, and a dotted line connecting neighboring Z to each other indicates that another atom is bonded to Z; and neighboring Z optionally forms a ring with each other via another atom.
- Z in the diboron compound in the formula of the general formula (7) its type is not particularly limited as long as it is an atom having a lone electron pair, such as an oxygen atom or a nitrogen atom.
- Z has a lone electron pair, and thus the lone electron pair acts with the vacant orbital of adjacent boron, and the reactivity of the diboron compound becomes suitable.
- four Z may be the same or different. Two neighboring Z may form a ring via another atom or may not form a ring.
- Examples of an atomic group forming a ring in the diboron compound represented by the general formula (7) include those shown in the following (7a) to (7c). Examples of an atomic group not forming a ring include those shown in the following (7d) to (7f).
- Ra and Rb represent the same or different optionally substituted alkyl groups or aryl groups; and * represents a bond.
- the amount of the asymmetric catalyst used differs depending on the type of the alkene compound used, the reaction container used, the form of the reaction, the economy, and the like, and it is preferred that the amount be 0.1 to 20 mol %, particularly 0.5 to 10 mol %, based on the alkene compound.
- t-butoxypotassium is preferably used as one component of the catalyst.
- a copper metal complex CuOtBu is produced with this additive, and the catalytic reaction proceeds in good yield.
- t-Butoxylithium or t-butoxysodium may be used, but t-butoxypotassium is preferred.
- the amount of t-butoxypotassium used is preferably 1.0 to 5.0, more preferably 1.0 to 2.5, in the molar ratio to copper.
- the reaction can be performed in a solvent as needed.
- the solvent is preferably one that dissolves the alkene compound that is a starting raw material and the organoboron compound that is a product, and does not react with the reaction reagents.
- the solvent include aromatic hydrocarbons such as benzene, toluene, and xylene; aliphatic hydrocarbons such as pentane, hexane, heptane, and octane; halogenated hydrocarbons such as methylene chloride, chloroform, carbon tetrachloride, and dichloroethane; ethers such as diethyl ether, diisopropyl ether, tert-butyl methyl ether, dimethoxyethane, tetrahydrofuran (THF), tetrahydropyran (THP), dioxane, and dioxolane; amides such as N,N-dimethylformamide (DMF) and N,N-dimethylacetamide (DMA); acetonitrile, N-methylpyrrolidone (NMP), dimethyl sulfoxide (DMSO), and dimethylimidazolidinone (DMI).
- the reaction temperature is usually ⁇ 78 to 20° C., and preferably ⁇ 50 to 0° C. considering economy and chiral purity.
- the reaction time differs depending on the type and amount of the catalyst used, the types and concentrations of the starting raw materials used, the reaction conditions such as reaction temperature, and the like and is usually 1 h or more, preferably 5 to 24 h.
- optically active organoboron compound obtained by this production method is used as a chiral intermediate raw material for medicines and agrochemicals, and physiologically active substances and is useful, for example, as a synthetic intermediate for antibiotics.
- R 1 represents an adamantyl group
- R 2 represents a tert-butyl group
- * represents an asymmetric center on a phosphorus atom
- a phosphine-borane (3a) (1.68 mmol, 531.4 mg) was dissolved in dehydrated THF (3.6 ml).
- a hexane solution of n-BuLi (1.64 mol/L, 1.68 mmol, 1.02 ml) was dropped into this solution at ⁇ 5° C. under a nitrogen atmosphere, and this was a B liquid.
- the B liquid was dropped into the A liquid at 0° C. After the completion of the dropping, the mixture was stirred at room temperature for 30 min. Water was added to the reaction liquid for quenching, followed by extraction with hexane three times. Then, the organic layer was subjected to dehydration treatment with sodium sulfate and then filtered, and with the filtrate, the solvent was removed under reduced pressure by an evaporator to obtain an oily residue (phosphinopyrazine-borane derivative (9a)).
- the C liquid was dropped into the D liquid at ⁇ 5° C., and the mixture was stirred at room temperature for 5 h. Water was added to the reaction liquid for quenching, followed by extraction with ethyl acetate two times. Then, the organic layer was washed with brine, subjected to dehydration treatment with sodium sulfate, and then filtered, and with the filtrate, the solvent was removed under reduced pressure by an evaporator to obtain an optically active phosphinopyrazine-borane derivative (10a).
- TMEDA/ethyl acetate (volume ratio 1:2) was added to the optically active phosphinopyrazine-borane derivative (10a) to adjust the concentration at 0.07 mol/L, and the mixture was stirred at room temperature for 2 h.
- further ethyl acetate was added to the reaction liquid for extraction with the ethyl acetate.
- the organic layer was washed well with water, a 6 mol/L hydrochloric acid aqueous solution, and brine. Then, the organic layer was subjected to dehydration treatment with sodium sulfate and then filtered, and with the filtrate, the solvent was removed under reduced pressure by an evaporator to obtain a residue.
- R 1 and R 2 represent an adamantyl group.
- R 1 represents an adamantyl group
- R 2 represents a 1,1,3,3-tetramethylbutyl group
- R 1 represents a tert-butyl group
- R 2 represents an adamantyl group
- a phosphine-borane (3d) was used instead of the phosphine-borane (3a) in the first step to obtain a phosphinopyrazine derivative (4d) in the second step.
- R 1 represents a tert-butyl group
- R 2 represents a 1,1,3,3-tetramethylbutyl group
- the phosphine-borane (3d) was used instead of the phosphine-borane (3a) in the first step to obtain the phosphinopyrazine derivative (4d).
- R 1 and R 2 represent a tert-butyl group.
- the phosphine-borane (3d) was used instead of the phosphine-borane (3a) in the first step to obtain the phosphinopyrazine derivative (4d).
- Target hydroboration products were obtained as in Example 6 except that the amounts of CuBF 4 (MeCN) 4 and (S)-Quniox-tODAd were each 5 mol % based on the alkene compound, and the compounds shown in Table 2 were used as the alkene compound.
- Table 2 Ph means a phenyl group
- Me means a methyl group
- Bn means a benzyl group.
- 3b in the “3b:4b” ratio represents the amount of the S form.
- “Optical purity of 3b” is the optical purity of the S form.
- a phosphinopyrazine derivative (4e) was obtained as in Example 1 except that a phosphine-borane (3e) was used instead of the phosphine-borane (3b).
- the phosphinopyrazine derivative (4e) (1.05 g, 3 mmol) and the (S)-t-butylmethylphosphineborane (5a) (390 mg, 3.3 mmol) were placed in a 30 mL three-necked flask equipped with a three-way cock, a pressure-equalizing dropping funnel, and a septum, and the interior of the system was purged with argon.
- Dehydrated THF (7.5 mL) and N,N,N′,N′-tetramethylethylenediamine (1.3 mL, 9.0 mmol) were added by a syringe through the septum (Y liquid).
- the flask was immersed in a low temperature bath at ⁇ 20° C., and while the Y liquid was stirred by a magnetic stirrer, a 1.0 M THF solution of potassium tert-butoxide (Z liquid) (3.6 mL, 3.6 mmol) was dropped into the Y liquid over 20 min. After the dropping, the mixture was maintained at the same temperature for 30 min, and then the temperature was increased to room temperature over about 1 h, and further stirring was continued for 4 h. The flask was immersed in an ice water bath, ethyl acetate (10 mL), water (7 mL), and 2 M hydrochloric acid (6.5 mL) were sequentially added, and the mixture was stirred well.
- Z liquid potassium tert-butoxide
- the mixture was transferred to a separatory funnel, and the upper layer was separated.
- the lower layer was subjected to extraction with ethyl acetate, and the organic layers were combined, washed with a saturated saline solution, and then dried with anhydrous sodium sulfate.
- the solvent was distilled off by an evaporator followed by vacuum drying to obtain an amorphous solid (1.22 g, 98%).
- Methanol (7 mL) was added to this product, and the mixture was stirred well by a spatula for crystallization.
- the obtained solid was filtered using a glass filter, and washed with methanol. The solid after the washing was vacuum-dried to obtain a yellow powder (990 mg, 78%).
- the present invention it is possible to provide a novel optically active 2,3-bisphosphinopyrazine derivative having an asymmetric center on a phosphorus atom useful for catalytic asymmetric synthesis reactions.
- the 2,3-bisphosphinopyrazine derivative of the present invention is useful particularly as the ligand of a catalyst for the asymmetric hydroboration reaction of an alkene compound.
- the optically active 2,3-bisphosphinopyrazine derivative of the present invention can be easily produced.
- a transition metal complex particularly a transition metal complex with a copper metal, comprising the optically active 2,3-bisphosphinopyrazine derivative of the present invention as a ligand, and an asymmetric catalyst having the same have high regioselectivity and high enantioselectivity and reaction activity for an asymmetric Markovnikov type hydroboration reaction using a terminal alkene.
- the desired organoboron compound can be produced by an industrially advantageous method due to high regioselectivity and high enantioselectivity.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Materials Engineering (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Inorganic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Catalysts (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
Abstract
Description
- Patent Literature 1: Japanese Patent Laid-Open No. 2000-319288
- Patent Literature 2: US2007021610 A1
- Non Patent Literature 1: Journal of Organic Chemistry Vol. 77, 4184-4188 (2012)
wherein R1 represents a group selected from a branched alkyl group having 3 or more carbon atoms, an adamantyl group, an optionally substituted cycloalkyl group, and an optionally substituted aryl group;
wherein R1 is as defined in the general formula (1), to act on a 2,3-dihalogenopyrazine derivative represented by the following general formula (2):
wherein R3 and n are as defined in the general formula (1); and X represents a halogen atom, to perform an aromatic nucleophilic substitution reaction (1), then performing a deboranation reaction (1) to obtain a phosphinopyrazine derivative represented by the following general formula (4):
wherein R1, R3, and n are as defined in the general formula (1), then allowing a deprotonation product of an optically active phosphine-borane represented by the following general formula (5):
wherein R2 and * are as defined in the general formula (1), to act on the phosphinopyrazine derivative to perform a nucleophilic substitution reaction (2), and then performing a deboranation reaction (2).
wherein R1 is as defined in the general formula (1), to act on a 2,3-dihalogenopyrazine derivative represented by the following general formula (2):
wherein R3 and n are as defined in the general formula (1); and X represents a halogen atom, to perform an aromatic nucleophilic substitution reaction (1), then performing a deboranation reaction (1) to obtain a phosphinopyrazine derivative represented by the following general formula (4):
wherein R1, R3, and n are as defined in the general formula (1), and then mixing a liquid comprising the phosphinopyrazine derivative, an optically active phosphine-borane represented by the following general formula (5′):
wherein R2′ is as defined in the general formula (1′; and * is as defined in the general formula (1), and a deboranating agent with a base to perform a reaction.
wherein R represents a hydrogen atom, an alkyl group, a substituted alkyl group, a cycloalkyl group, a substituted cycloalkyl group, an aralkyl group, a substituted aralkyl group, an aryl group, a substituted aryl group, an aliphatic heterocyclic group, a substituted aliphatic heterocyclic group, an aromatic heterocyclic group, a substituted aromatic heterocyclic group, an alkoxy group, a substituted alkoxy group, an aralkyloxy group, a substituted aralkyloxy group, an aryloxy group, a substituted aryloxy group, an alkyloxycarbonyl group, an aralkyloxycarbonyl group, a substituted silyl group or a substituted silyloxy group, an amino group, a substituted amino group, an alkylaminocarboxy group, a substituted alkylaminocarboxy group, an arylaminocarboxy group, an alkyloxycarbonyloxy group, an aryloxycarbonyloxy group, or a halogen atom; and t represents an integer of 0 to 10, to a coupling reaction with a diboron compound represented by the following general formula (7):
wherein Z represents the same or a different atom having a lone electron pair, and a dotted line connecting neighboring Z to each other indicates that another atom is bonded to Z; and neighboring Z optionally forms a ring with each other via another atom, in the presence of the asymmetric catalyst, the organoboron compound being represented by the following general formula (8):
wherein R and t are as defined in the general formula (6); Z is as defined in the general formula (7); and * indicates an asymmetric carbon atom.
wherein R1, R3, and n are as defined in the general formula (1); and X is as defined in the general formula (2).
wherein R represents a hydrogen atom, an alkyl group, a substituted alkyl group, a cycloalkyl group, a substituted cycloalkyl group, an aralkyl group, a substituted aralkyl group, an aryl group, a substituted aryl group, an aliphatic heterocyclic group, a substituted aliphatic heterocyclic group, an aromatic heterocyclic group, a substituted aromatic heterocyclic group, an alkoxy group, a substituted alkoxy group, an aralkyloxy group, a substituted aralkyloxy group, an aryloxy group, a substituted aryloxy group, an alkyloxycarbonyl group, an aralkyloxycarbonyl group, a substituted silyl group or a substituted silyloxy group, an amino group, a substituted amino group, an alkylaminocarboxy group, a substituted alkylaminocarboxy group, an arylaminocarboxy group, an alkyloxycarbonyloxy group, an aryloxycarbonyloxy group, or a halogen atom; and t represents an integer of 0 to 10.
wherein Z represents the same or a different atom having a lone electron pair, and a dotted line connecting neighboring Z to each other indicates that another atom is bonded to Z; and neighboring Z optionally forms a ring with each other via another atom.
wherein Ra and Rb represent the same or different optionally substituted alkyl groups or aryl groups; and * represents a bond.
wherein R1 represents an adamantyl group, R2 represents a tert-butyl group, and * represents an asymmetric center on a phosphorus atom.
<First Step>
| TABLE 1 | |||||
| Yield | Optical purity | ||||
| Ligand | (%) | 3a/4a1) | of 3a (ee %) | ||
| Example 6 | (S) -Quinox-tODAd | 94 | 92:8 | 98 |
| Example 7 | (R) -Quinox-CFDAd | 40 | 84:16 | −91 |
| Example 8 | (S) -Quinox-TAd | 85 | 86:14 | 97 |
| Example 9 | (S) -Quinox-AdDCF | 69 | 82:18 | 91 |
| Example 10 | (S) -Quinox-tODCF | 37 | 80:20 | 93 |
| Comparative | (R) -Quinox-TCF | 39 | 78:22 | −87 |
| Example 1 | ||||
| Note) | ||||
| 1)3a/4a is an area ratio measured by gas chromatography | ||||
| TABLE 2 | |||||
| Type of R′ in | |||||
| alkene compound | Yield | Optical purity | |||
| general formula (1b) | (%) | 3b/4b2) | of 3b (ee %) | ||
| Example 11 | CH3—(CH2)7— | 64 | 90:10 | 96 |
| Example 12 | Ph—(CH2)— | 88 | 85:15 | 99 |
| Example 13 | Cl—(CH2)4— | 89 | 86:14 | 98 |
| Example 14 | Ph2MeSiO—(CH2)3— | 52 | 89:11 | 97 |
| Example 15 | BnO—(CH2)4— | 60 | 88:12 | 95 |
| Note) | ||||
| 2)3b/4b is an area ratio measured by gas chromatography | ||||
wherein R1 represents a phenyl group, and R2 represents a tert-butyl group.
<First Step and Second Step>
Claims (5)
Applications Claiming Priority (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JPJP2017-195879 | 2017-10-06 | ||
| JP2017195879 | 2017-10-06 | ||
| JP2017-195879 | 2017-10-06 | ||
| JP2018-119159 | 2018-06-22 | ||
| JPJP2018-119159 | 2018-06-22 | ||
| JP2018119159 | 2018-06-22 | ||
| PCT/JP2018/036440 WO2019069828A1 (en) | 2017-10-06 | 2018-09-28 | Optically active 2,3-bisphosphinopyrazine derivative, method for producing same, transition metal complex, and method for producing organic boron compound |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20200377536A1 US20200377536A1 (en) | 2020-12-03 |
| US11053265B2 true US11053265B2 (en) | 2021-07-06 |
Family
ID=65994811
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/641,845 Active US11053265B2 (en) | 2017-10-06 | 2018-09-28 | Optically active substituted 2,3-bisphosphinoquinoxalines and processes for producing the same |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US11053265B2 (en) |
| JP (1) | JP6547087B1 (en) |
| CN (1) | CN111032668B (en) |
| WO (1) | WO2019069828A1 (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110563752B (en) * | 2019-08-06 | 2022-04-01 | 河北科技大学 | Method for synthesizing secondary or tertiary alkyl borate by iron-catalyzed Ma's selective hydroboration of olefin |
| CN110483560B (en) * | 2019-08-06 | 2022-03-15 | 河北科技大学 | Iron catalysis system for synthesizing alkyl borate through aliphatic olefin hydroboration reaction and application method thereof |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000319288A (en) | 1999-05-10 | 2000-11-21 | Nippon Chem Ind Co Ltd | Optically active 1,2-bis (dialkylphosphino) benzene derivative, method for producing the same, and rhodium metal complex having the compound as a ligand |
| US20070021610A1 (en) | 2005-07-25 | 2007-01-25 | Tsuneo Imamoto | 2, 3-Bis(dialkylphosphino)pyrazine derivative, process of producing the same, and metal complex having the same as ligand |
| JP2011219413A (en) | 2010-04-09 | 2011-11-04 | Nippon Chem Ind Co Ltd | Method for producing pyrazine derivative |
-
2018
- 2018-09-28 CN CN201880051140.8A patent/CN111032668B/en active Active
- 2018-09-28 JP JP2019507354A patent/JP6547087B1/en active Active
- 2018-09-28 WO PCT/JP2018/036440 patent/WO2019069828A1/en not_active Ceased
- 2018-09-28 US US16/641,845 patent/US11053265B2/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000319288A (en) | 1999-05-10 | 2000-11-21 | Nippon Chem Ind Co Ltd | Optically active 1,2-bis (dialkylphosphino) benzene derivative, method for producing the same, and rhodium metal complex having the compound as a ligand |
| US20070021610A1 (en) | 2005-07-25 | 2007-01-25 | Tsuneo Imamoto | 2, 3-Bis(dialkylphosphino)pyrazine derivative, process of producing the same, and metal complex having the same as ligand |
| JP2011219413A (en) | 2010-04-09 | 2011-11-04 | Nippon Chem Ind Co Ltd | Method for producing pyrazine derivative |
Non-Patent Citations (10)
| Title |
|---|
| Cai et al., "Copper-Catalyed Enantioseletive Markovnikov Protoboration of x-Olefins Enabled by a Buttressed N-Hetereocylic Carbene Ligand", Angewandte Chemie International Edition, Dec. 12, 2017, pp. 1376-1380, 57. Cited in ISR. (5 pages). |
| Hoge et al., "Highly Selective Asymmetric Hydrogenation Using a Three Hindered Quadrant Bisphosphine Rhodium Catalyst", Journal of the American Chemical society, (2004), pp. 5966-5967, 126. Cited in ISR. (2 pages). |
| Imamoto et al., "Air-Stable P-Chiral Bidentate Phosphine Ligand with (1-Adamanty)methylphosphino Group", Chemistry Letters, (2007), pp. 500-501, vol. 36, No. 4. Cited in ISR. (4 pages). |
| Imamoto et al., "An Air-Stable P-Chiral Phosphine Ligand for Highly Enantioselective Transition-Metal-Catalyzed Reactions", Journal of the American Chemical Society, (2005), pp. 11934-11935, 127. Cited in ISR. (2 pages). |
| Imamoto et al., "Searching for Practically Useful P-Chirogenic Phosphine Ligands", The Chemical Record, (2016), pp. 2659-2673, 16. Cited in ISR. (25 pages). |
| International Search Report dated Nov. 13, 2018, issued in counterpart International Application No. PCT/JP2018/036440. (3 pages). |
| Iwamoto et al., "Computational design of high-performance ligand for enantioselective Markovnikov hydroboration of aliphatic terminal alkenes", Nature Communications, Jun. 12, 2018, pp. 2041-1723, 9 (1), 1-10. Cited in ISR. (10pages). |
| Iwamoto et al., "Computational design of high-performance ligand for enantioselective Markovnikov hydroboration of aliphatic terminal alkenes", Nature Communications, Jun. 12, 2018, pp. 2041-1723, 9 (1), 1-10. Cited in ISR. with English Translation. (14 pages). |
| Liu et al., "A new and convenient approach for the synthesis of P-stereogenic intermediates bearing a tert-butyl (methyl)phosphino group", Research on Chemical Intermediates, Apr. 7, 2017, pp. 4959-4966, 43 (8). Cited in ISR. (8 pages). |
| Zhang et a., "Three-Hindered Quadrant Phosphine Ligands with an Aromatic Ring Backbone for the Rhodium-Catalyzed Asymmetric Hydrogenation of Functionalized Alkenes", The Journal of Organic Chemistry, (2012), pp. 11934-11935, 77. Cited in Specification & ISR. (5 pages). |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2019069828A1 (en) | 2019-04-11 |
| US20200377536A1 (en) | 2020-12-03 |
| JPWO2019069828A1 (en) | 2019-11-14 |
| JP6547087B1 (en) | 2019-07-17 |
| CN111032668A (en) | 2020-04-17 |
| CN111032668B (en) | 2022-10-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5718178B2 (en) | Ruthenium-diamine complex and method for producing optically active compound | |
| US11053265B2 (en) | Optically active substituted 2,3-bisphosphinoquinoxalines and processes for producing the same | |
| JP5271503B2 (en) | Method for producing organoboron compound | |
| JP5465584B2 (en) | Method for producing pyrazine derivative | |
| JP6048762B2 (en) | Process for producing optically active β-hydroxy-α-aminocarboxylic acid ester | |
| CN106748917A (en) | A kind of chiral sulfenamide part and its preparation method and application | |
| JP5963140B2 (en) | Asymmetric dehydration condensation agent | |
| JP6476497B2 (en) | Process for producing optically active compound, and novel metal-diamine complex | |
| JP5350767B2 (en) | Novel phosphine borane compound and method for producing the same, and method for producing hydrogen-phosphine borane compound | |
| US11084835B2 (en) | 2,3-bisphosphinopyrazine derivative, method for producing same, transition metal complex, asymmetric catalyst, and method for producing organic boron compound | |
| JP4649645B2 (en) | Process for producing optically active alcohol compounds | |
| US11453687B2 (en) | Production method of biarylphosphine | |
| US9382280B2 (en) | Diamine compound and metal complexes, and method for producing optically active compounds | |
| US20090030200A1 (en) | Process of preparing optically active allyl compound | |
| JPWO2019012918A1 (en) | Method for producing optically active 2,3-bisphosphinopyrazine derivative and method for producing optically active phosphine transition metal complex | |
| US11021500B2 (en) | Process for preparing optically active 2,3-bisphosphino-substituted quinoxalines | |
| WO2016056669A1 (en) | Solid-supported ruthenium-diamine complex, and method for manufacturing optically active compound | |
| WO2026023360A1 (en) | Method for producing optically active hydrogen-phosphine borane compound and method for producing optically active 2,3-bisphosphinopyrazine derivative | |
| CN105237435B (en) | A kind of preparation method of α cyano group amine | |
| JP2009046469A (en) | Process for producing optically active allyl compounds | |
| JP2019135225A (en) | Iodination of aromatic ring by hydrochloric acid solution of monochloroiodine | |
| JP2013139417A (en) | Bis-imidazolidine pincer complex and bis-imidazolidine pincer catalyst, and methods for preparing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: NIPPON CHEMICAL INDUSTRIAL CO., LTD., JAPAN Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:ITO, HAJIME;IWAMOTO, HIROAKI;IMAMOTO, TSUNEO;AND OTHERS;REEL/FRAME:051921/0452 Effective date: 20200109 Owner name: NATIONAL UNIVERSITY CORPORATION HOKKAIDO UNIVERSITY, JAPAN Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:ITO, HAJIME;IWAMOTO, HIROAKI;IMAMOTO, TSUNEO;AND OTHERS;REEL/FRAME:051921/0452 Effective date: 20200109 |
|
| FEPP | Fee payment procedure |
Free format text: ENTITY STATUS SET TO UNDISCOUNTED (ORIGINAL EVENT CODE: BIG.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NOTICE OF ALLOWANCE MAILED -- APPLICATION RECEIVED IN OFFICE OF PUBLICATIONS |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: PUBLICATIONS -- ISSUE FEE PAYMENT RECEIVED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: PUBLICATIONS -- ISSUE FEE PAYMENT VERIFIED |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1551); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 4 |