US10577358B2 - Strigolactone formulations and uses thereof - Google Patents
Strigolactone formulations and uses thereof Download PDFInfo
- Publication number
- US10577358B2 US10577358B2 US15/947,508 US201815947508A US10577358B2 US 10577358 B2 US10577358 B2 US 10577358B2 US 201815947508 A US201815947508 A US 201815947508A US 10577358 B2 US10577358 B2 US 10577358B2
- Authority
- US
- United States
- Prior art keywords
- mixture
- strigolactones
- plant
- orobanchol
- formulations
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 1504
- 238000009472 formulation Methods 0.000 title abstract description 408
- XHSDUVBUZOUAOQ-WJQMYRPNSA-N (3e,3ar,8bs)-3-[[(2r)-4-methyl-5-oxo-2h-furan-2-yl]oxymethylidene]-4,8b-dihydro-3ah-indeno[1,2-b]furan-2-one Chemical compound O1C(=O)C(C)=C[C@@H]1O\C=C/1C(=O)O[C@@H]2C3=CC=CC=C3C[C@@H]2\1 XHSDUVBUZOUAOQ-WJQMYRPNSA-N 0.000 title abstract description 174
- 238000000034 method Methods 0.000 claims abstract description 171
- 238000004519 manufacturing process Methods 0.000 claims abstract description 19
- 150000001875 compounds Chemical class 0.000 claims description 201
- 125000000217 alkyl group Chemical group 0.000 claims description 82
- 125000001188 haloalkyl group Chemical group 0.000 claims description 54
- TZIHFWKZFHZASV-UHFFFAOYSA-N methyl formate Chemical compound COC=O TZIHFWKZFHZASV-UHFFFAOYSA-N 0.000 claims description 48
- 125000001475 halogen functional group Chemical group 0.000 claims description 36
- IMKJGXCIJJXALX-UHFFFAOYSA-N ent-Norambreinolide Natural products C1CC2C(C)(C)CCCC2(C)C2C1(C)OC(=O)C2 IMKJGXCIJJXALX-UHFFFAOYSA-N 0.000 claims description 33
- 229940096995 sclareolide Drugs 0.000 claims description 29
- IMKJGXCIJJXALX-SHUKQUCYSA-N Norambreinolide Chemical compound CC([C@@H]1CC2)(C)CCC[C@]1(C)[C@@H]1[C@]2(C)OC(=O)C1 IMKJGXCIJJXALX-SHUKQUCYSA-N 0.000 claims description 28
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 claims description 11
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 8
- ZCSHNCUQKCANBX-UHFFFAOYSA-N lithium diisopropylamide Chemical compound [Li+].CC(C)[N-]C(C)C ZCSHNCUQKCANBX-UHFFFAOYSA-N 0.000 claims 8
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 claims 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 claims 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims 2
- 229910000027 potassium carbonate Inorganic materials 0.000 claims 1
- 239000000463 material Substances 0.000 abstract description 325
- QXTUQXRFEBHUBA-DYLOANJQSA-N 5-Deoxystrigol Chemical compound O1C(=O)C(C)=C[C@@H]1O\C=C/1C(=O)O[C@@H]2C(C(C)(C)CCC3)=C3C[C@@H]2\1 QXTUQXRFEBHUBA-DYLOANJQSA-N 0.000 abstract description 144
- KEOQXENEHBJOJF-NTEUORMPSA-N 5-deoxystrigol Natural products CC1CCC(C)(C)C2=C1CC3C2OC(=O)/C/3=C/OC4OC(=O)C(=C4)C KEOQXENEHBJOJF-NTEUORMPSA-N 0.000 abstract description 142
- 239000000126 substance Substances 0.000 abstract description 123
- 230000003278 mimic effect Effects 0.000 abstract description 106
- 108090000623 proteins and genes Proteins 0.000 abstract description 50
- 102000040430 polynucleotide Human genes 0.000 abstract description 34
- 108091033319 polynucleotide Proteins 0.000 abstract description 34
- 239000002157 polynucleotide Substances 0.000 abstract description 34
- 230000015572 biosynthetic process Effects 0.000 abstract description 32
- 230000037361 pathway Effects 0.000 abstract description 12
- 239000002207 metabolite Substances 0.000 abstract description 8
- 238000001311 chemical methods and process Methods 0.000 abstract description 6
- 241000196324 Embryophyta Species 0.000 description 913
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 329
- CDBBMEYPRMUMTR-UHFFFAOYSA-N (+)-(3aR,4S,8bS,2'R)-3-[(E)-2',5'-dihydro-4'-methyl-5'-oxo-2'-furanyloxymethylene]-4-hydroxy-8,8-dimethyl-3,3a,4,5,6,7,8,8b-octahydroindeno[1,2-b]furan-2-one Natural products O1C(=O)C(C)=CC1OC=C1C(=O)OC2C(C(C)(C)CCC3)=C3C(O)C21 CDBBMEYPRMUMTR-UHFFFAOYSA-N 0.000 description 200
- CDBBMEYPRMUMTR-KIKJJFSISA-N (3E,3aR,4R,8bR)-4-hydroxy-8,8-dimethyl-3-[[(2R)-4-methyl-5-oxo-2H-furan-2-yl]oxymethylidene]-3a,4,5,6,7,8b-hexahydroindeno[1,2-b]furan-2-one Chemical compound O1C(=O)C(C)=C[C@@H]1O\C=C/1C(=O)O[C@H]2C(C(C)(C)CCC3)=C3[C@H](O)[C@H]2\1 CDBBMEYPRMUMTR-KIKJJFSISA-N 0.000 description 200
- CDBBMEYPRMUMTR-MHHPOBJPSA-N Orobanchol Natural products O(/C=C/1\C(=O)O[C@H]2[C@@H]\1[C@H](O)C1=C2C(C)(C)CCC1)[C@@H]1OC(=O)C(C)=C1 CDBBMEYPRMUMTR-MHHPOBJPSA-N 0.000 description 200
- 150000003839 salts Chemical class 0.000 description 175
- 239000012453 solvate Substances 0.000 description 173
- VOFXXOPWCBSPAA-UPXBXCCMSA-N (+)-Strigol Natural products O(/C=C/1\C(=O)O[C@H]2[C@@H]\1CC=1[C@H](O)CCC(C)(C)C2=1)[C@@H]1OC(=O)C(C)=C1 VOFXXOPWCBSPAA-UPXBXCCMSA-N 0.000 description 142
- VOFXXOPWCBSPAA-KCNJUGRMSA-N strigol Chemical compound O1C(=O)C(C)=C[C@@H]1O\C=C/1C(=O)O[C@@H]2C(C(C)(C)CC[C@@H]3O)=C3C[C@@H]2\1 VOFXXOPWCBSPAA-KCNJUGRMSA-N 0.000 description 142
- KHSREFIWULNDAB-YCUBLIQYSA-N Sorgolactone Chemical compound O([C@@H]1C2=C(C[C@@H]11)CCC[C@@H]2C)C(=O)\C1=C\O[C@@H]1OC(=O)C(C)=C1 KHSREFIWULNDAB-YCUBLIQYSA-N 0.000 description 140
- KHSREFIWULNDAB-MRZMDFQOSA-N Sorgolactone Natural products O(/C=C/1\C(=O)O[C@H]2[C@@H]\1CC1=C2[C@@H](C)CCC1)[C@@H]1OC(=O)C(C)=C1 KHSREFIWULNDAB-MRZMDFQOSA-N 0.000 description 140
- ARGBKKMNXCMJSK-UHFFFAOYSA-N Strigol Natural products CC1C=C(OC=C2/C3CC4=C(C3OC2=O)C(C)(C)CCC4O)OC1=O ARGBKKMNXCMJSK-UHFFFAOYSA-N 0.000 description 138
- FIKOOQXJBAJJSE-FYTPEDKISA-N Alectrol Natural products O(/C=C/1\C(=O)O[C@@H]2[C@]3(O)C(C)(C)CCCC3=C[C@H]\12)[C@@H]1OC(=O)C(C)=C1 FIKOOQXJBAJJSE-FYTPEDKISA-N 0.000 description 83
- DLRIUVHQJRZTMZ-UKTHLTGXSA-N orobanchyl acetate Natural products CC(=O)OC1C2C(OC(=O)/C/2=C/OC3OC(=O)C(=C3)C)C4=C1CCCC4(C)C DLRIUVHQJRZTMZ-UKTHLTGXSA-N 0.000 description 83
- 229930009674 sesquiterpene lactone Natural products 0.000 description 80
- 150000002107 sesquiterpene lactone derivatives Chemical class 0.000 description 80
- HATRDXDCPOXQJX-UHFFFAOYSA-N Thapsigargin Natural products CCCCCCCC(=O)OC1C(OC(O)C(=C/C)C)C(=C2C3OC(=O)C(C)(O)C3(O)C(CC(C)(OC(=O)C)C12)OC(=O)CCC)C HATRDXDCPOXQJX-UHFFFAOYSA-N 0.000 description 78
- KYSWLLSZOWOOGF-ZMJOFITKSA-N (3e,3ar,8r,8bs)-8-(hydroxymethyl)-8-methyl-3-[[(2r)-4-methyl-5-oxo-2h-furan-2-yl]oxymethylidene]-3a,4,5,6,7,8b-hexahydroindeno[1,2-b]furan-2-one Chemical compound O1C(=O)C(C)=C[C@@H]1O\C=C/1C(=O)O[C@@H]2C([C@](C)(CO)CCC3)=C3C[C@@H]2\1 KYSWLLSZOWOOGF-ZMJOFITKSA-N 0.000 description 67
- JMOKGABSEPTJPV-SDHIYRAFSA-N [(1S,2R,5E,6R,7R,8R)-12,12-dimethyl-5-[[(2R)-4-methyl-5-oxo-2H-furan-2-yl]oxymethylidene]-4-oxo-3,13-dioxatetracyclo[6.4.1.01,8.02,6]tridecan-7-yl] acetate Chemical compound O([C@@H]1[C@H]2[C@H]([C@]34O[C@]41C(CCC3)(C)C)OC(=O)C)C(=O)\C2=C\O[C@@H]1OC(=O)C(C)=C1 JMOKGABSEPTJPV-SDHIYRAFSA-N 0.000 description 67
- 229930002875 chlorophyll Natural products 0.000 description 67
- 235000019804 chlorophyll Nutrition 0.000 description 67
- ATNHDLDRLWWWCB-AENOIHSZSA-M chlorophyll a Chemical compound C1([C@@H](C(=O)OC)C(=O)C2=C3C)=C2N2C3=CC(C(CC)=C3C)=[N+]4C3=CC3=C(C=C)C(C)=C5N3[Mg-2]42[N+]2=C1[C@@H](CCC(=O)OC\C=C(/C)CCC[C@H](C)CCC[C@H](C)CCCC(C)C)[C@H](C)C2=C5 ATNHDLDRLWWWCB-AENOIHSZSA-M 0.000 description 67
- JMOKGABSEPTJPV-FMIVXFBMSA-N fabacyl acetate Natural products CC(=O)OC1C2C(OC(=O)/C/2=C/OC3OC(=O)C(=C3)C)C45OC14CCCC5(C)C JMOKGABSEPTJPV-FMIVXFBMSA-N 0.000 description 67
- KYSWLLSZOWOOGF-UHFFFAOYSA-N sorghumol Natural products O1C(=O)C(C)=CC1OC=C1C(=O)OC2C(C(C)(CO)CCC3)=C3CC21 KYSWLLSZOWOOGF-UHFFFAOYSA-N 0.000 description 67
- QXTUQXRFEBHUBA-MDVYPADHSA-N (3e,3ar,8bs)-8,8-dimethyl-3-[[(2s)-4-methyl-5-oxo-2h-furan-2-yl]oxymethylidene]-3a,4,5,6,7,8b-hexahydroindeno[1,2-b]furan-2-one Chemical compound O1C(=O)C(C)=C[C@H]1O\C=C/1C(=O)O[C@@H]2C(C(C)(C)CCC3)=C3C[C@@H]2\1 QXTUQXRFEBHUBA-MDVYPADHSA-N 0.000 description 63
- CHKDOMXJQUEQLN-AORPGMBKSA-N (3e,3as,4s,8bs)-4-hydroxy-8,8-dimethyl-3-[[(2r)-4-methyl-5-oxo-2h-furan-2-yl]oxymethylidene]-4,5,6,8b-tetrahydro-3ah-indeno[1,2-b]furan-2,7-dione Chemical compound O1C(=O)C(C)=C[C@@H]1O\C=C/1C(=O)O[C@@H]2C(C(C)(C)C(=O)CC3)=C3[C@@H](O)[C@@H]2\1 CHKDOMXJQUEQLN-AORPGMBKSA-N 0.000 description 63
- CDBBMEYPRMUMTR-XCPYXGTHSA-N (3e,3as,4s,8bs)-4-hydroxy-8,8-dimethyl-3-[[(2s)-4-methyl-5-oxo-2h-furan-2-yl]oxymethylidene]-3a,4,5,6,7,8b-hexahydroindeno[1,2-b]furan-2-one Chemical compound O1C(=O)C(C)=C[C@H]1O\C=C/1C(=O)O[C@@H]2C(C(C)(C)CCC3)=C3[C@@H](O)[C@@H]2\1 CDBBMEYPRMUMTR-XCPYXGTHSA-N 0.000 description 63
- CHKDOMXJQUEQLN-UHFFFAOYSA-N 7-oxoorobanchol Natural products O1C(=O)C(C)=CC1OC=C1C(=O)OC2C(C(C)(C)C(=O)CC3)=C3C(O)C21 CHKDOMXJQUEQLN-UHFFFAOYSA-N 0.000 description 63
- 230000001965 increasing effect Effects 0.000 description 53
- 210000004027 cell Anatomy 0.000 description 47
- 230000003111 delayed effect Effects 0.000 description 45
- 238000005804 alkylation reaction Methods 0.000 description 39
- 240000008042 Zea mays Species 0.000 description 38
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 37
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 31
- 238000007031 hydroxymethylation reaction Methods 0.000 description 30
- 230000002829 reductive effect Effects 0.000 description 29
- 238000006482 condensation reaction Methods 0.000 description 26
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 26
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 24
- 235000005822 corn Nutrition 0.000 description 24
- -1 phosphate ester Chemical class 0.000 description 24
- 239000000575 pesticide Substances 0.000 description 21
- 239000003337 fertilizer Substances 0.000 description 20
- 239000000047 product Substances 0.000 description 19
- JLIDBLDQVAYHNE-YKALOCIXSA-N (+)-Abscisic acid Chemical compound OC(=O)/C=C(/C)\C=C\[C@@]1(O)C(C)=CC(=O)CC1(C)C JLIDBLDQVAYHNE-YKALOCIXSA-N 0.000 description 18
- 125000003118 aryl group Chemical group 0.000 description 18
- 125000001072 heteroaryl group Chemical group 0.000 description 18
- 230000008569 process Effects 0.000 description 17
- 238000006243 chemical reaction Methods 0.000 description 16
- 230000029936 alkylation Effects 0.000 description 13
- 238000003786 synthesis reaction Methods 0.000 description 13
- 101100129496 Arabidopsis thaliana CYP711A1 gene Proteins 0.000 description 12
- 101150030337 CCD7 gene Proteins 0.000 description 12
- 101100083446 Danio rerio plekhh1 gene Proteins 0.000 description 12
- 101150081158 crtB gene Proteins 0.000 description 12
- 101150000046 crtE gene Proteins 0.000 description 12
- 101150011633 crtI gene Proteins 0.000 description 12
- 230000002035 prolonged effect Effects 0.000 description 12
- 239000002689 soil Substances 0.000 description 12
- 101100327165 Arabidopsis thaliana CCD8 gene Proteins 0.000 description 11
- 241000233866 Fungi Species 0.000 description 11
- 101100061456 Streptomyces griseus crtB gene Proteins 0.000 description 11
- 101100114901 Streptomyces griseus crtI gene Proteins 0.000 description 11
- 235000016383 Zea mays subsp huehuetenangensis Nutrition 0.000 description 11
- 239000012535 impurity Substances 0.000 description 11
- 235000009973 maize Nutrition 0.000 description 11
- 241000208000 Striga Species 0.000 description 10
- 230000000694 effects Effects 0.000 description 10
- 230000003032 phytopathogenic effect Effects 0.000 description 10
- FCRACOPGPMPSHN-UHFFFAOYSA-N desoxyabscisic acid Natural products OC(=O)C=C(C)C=CC1C(C)=CC(=O)CC1(C)C FCRACOPGPMPSHN-UHFFFAOYSA-N 0.000 description 9
- 238000007726 management method Methods 0.000 description 9
- LMBFAGIMSUYTBN-MPZNNTNKSA-N teixobactin Chemical compound C([C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(=O)N[C@H](CCC(N)=O)C(=O)N[C@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(=O)N[C@H]1C(N[C@@H](C)C(=O)N[C@@H](C[C@@H]2NC(=N)NC2)C(=O)N[C@H](C(=O)O[C@H]1C)[C@@H](C)CC)=O)NC)C1=CC=CC=C1 LMBFAGIMSUYTBN-MPZNNTNKSA-N 0.000 description 9
- 208000005156 Dehydration Diseases 0.000 description 8
- 241000238631 Hexapoda Species 0.000 description 8
- 230000024346 drought recovery Effects 0.000 description 8
- 239000008187 granular material Substances 0.000 description 8
- 238000003973 irrigation Methods 0.000 description 8
- 230000002262 irrigation Effects 0.000 description 8
- 229910052760 oxygen Inorganic materials 0.000 description 8
- 230000003071 parasitic effect Effects 0.000 description 8
- FKOULKPQMBAJDG-UHFFFAOYSA-N 2-bromo-4-methyl-2h-furan-5-one Chemical compound CC1=CC(Br)OC1=O FKOULKPQMBAJDG-UHFFFAOYSA-N 0.000 description 7
- QSYHSDNIQZDYGF-UHFFFAOYSA-N 4-bromo-2h-furan-5-one Chemical compound BrC1=CCOC1=O QSYHSDNIQZDYGF-UHFFFAOYSA-N 0.000 description 7
- 241000607479 Yersinia pestis Species 0.000 description 7
- 239000000969 carrier Substances 0.000 description 7
- 230000008859 change Effects 0.000 description 7
- 238000011161 development Methods 0.000 description 7
- 230000018109 developmental process Effects 0.000 description 7
- 239000000843 powder Substances 0.000 description 7
- 240000007594 Oryza sativa Species 0.000 description 6
- 239000002585 base Substances 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 6
- 239000007795 chemical reaction product Substances 0.000 description 6
- 239000002270 dispersing agent Substances 0.000 description 6
- 235000013305 food Nutrition 0.000 description 6
- 229910052757 nitrogen Inorganic materials 0.000 description 6
- 230000035882 stress Effects 0.000 description 6
- 235000002637 Nicotiana tabacum Nutrition 0.000 description 5
- 244000061176 Nicotiana tabacum Species 0.000 description 5
- 235000007164 Oryza sativa Nutrition 0.000 description 5
- 244000062793 Sorghum vulgare Species 0.000 description 5
- 150000001408 amides Chemical class 0.000 description 5
- 230000035784 germination Effects 0.000 description 5
- 230000000116 mitigating effect Effects 0.000 description 5
- 235000009566 rice Nutrition 0.000 description 5
- 239000007921 spray Substances 0.000 description 5
- 229910052717 sulfur Inorganic materials 0.000 description 5
- XUFAHPAHNJCLLZ-WYEAJDFLSA-N (3as,4s,5as,9as,9br)-4-hydroxy-3a,6,6,9a-tetramethyl-1,4,5,5a,7,8,9,9b-octahydrobenzo[e][1]benzofuran-2-one Chemical compound C([C@@]12C)CCC(C)(C)[C@@H]1C[C@H](O)[C@]1(C)[C@@H]2CC(=O)O1 XUFAHPAHNJCLLZ-WYEAJDFLSA-N 0.000 description 4
- 235000007688 Lycopersicon esculentum Nutrition 0.000 description 4
- ZSXGLVDWWRXATF-UHFFFAOYSA-N N,N-dimethylformamide dimethyl acetal Chemical compound COC(OC)N(C)C ZSXGLVDWWRXATF-UHFFFAOYSA-N 0.000 description 4
- 241000235648 Pichia Species 0.000 description 4
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 4
- 240000003768 Solanum lycopersicum Species 0.000 description 4
- 230000002152 alkylating effect Effects 0.000 description 4
- 238000013459 approach Methods 0.000 description 4
- 230000000853 biopesticidal effect Effects 0.000 description 4
- 235000013339 cereals Nutrition 0.000 description 4
- 210000003527 eukaryotic cell Anatomy 0.000 description 4
- 230000012010 growth Effects 0.000 description 4
- 230000007246 mechanism Effects 0.000 description 4
- 238000005580 one pot reaction Methods 0.000 description 4
- 239000000546 pharmaceutical excipient Substances 0.000 description 4
- 235000002020 sage Nutrition 0.000 description 4
- 230000011664 signaling Effects 0.000 description 4
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 239000004094 surface-active agent Substances 0.000 description 4
- 239000004562 water dispersible granule Substances 0.000 description 4
- 210000005253 yeast cell Anatomy 0.000 description 4
- FQTLCLSUCSAZDY-UHFFFAOYSA-N (+) E(S) nerolidol Natural products CC(C)=CCCC(C)=CCCC(C)(O)C=C FQTLCLSUCSAZDY-UHFFFAOYSA-N 0.000 description 3
- SQEBMLCQNJOCBG-HVHJFMEUSA-N (5s)-3-(hydroxymethyl)-5-methoxy-4-methyl-5-[(e)-2-phenylethenyl]furan-2-one Chemical compound C=1C=CC=CC=1/C=C/[C@]1(OC)OC(=O)C(CO)=C1C SQEBMLCQNJOCBG-HVHJFMEUSA-N 0.000 description 3
- GCVGDGFZLSSUIO-UHFFFAOYSA-N 4-chloro-2h-furan-5-one Chemical compound ClC1=CCOC1=O GCVGDGFZLSSUIO-UHFFFAOYSA-N 0.000 description 3
- 235000003261 Artemisia vulgaris Nutrition 0.000 description 3
- 240000006891 Artemisia vulgaris Species 0.000 description 3
- FBMORZZOJSDNRQ-UHFFFAOYSA-N Demethoxy,B,HCl-Adriamycin Natural products C1C2C(=C)CCCC2(C)CC2(O)C1=C(C)C(=O)O2 FBMORZZOJSDNRQ-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 244000068988 Glycine max Species 0.000 description 3
- 235000010469 Glycine max Nutrition 0.000 description 3
- 241000219823 Medicago Species 0.000 description 3
- 235000017587 Medicago sativa ssp. sativa Nutrition 0.000 description 3
- 241001508464 Orobanche Species 0.000 description 3
- 244000182022 Salvia sclarea Species 0.000 description 3
- 235000011684 Sorghum saccharatum Nutrition 0.000 description 3
- 240000001705 Striga asiatica Species 0.000 description 3
- 241000960395 Striga hermonthica Species 0.000 description 3
- 229910021627 Tin(IV) chloride Inorganic materials 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 235000021307 Triticum Nutrition 0.000 description 3
- 241000209140 Triticum Species 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- 230000008901 benefit Effects 0.000 description 3
- 230000033228 biological regulation Effects 0.000 description 3
- 230000001276 controlling effect Effects 0.000 description 3
- 239000003995 emulsifying agent Substances 0.000 description 3
- 229930002882 germacranolide Natural products 0.000 description 3
- 150000003073 germacranolide derivatives Chemical class 0.000 description 3
- XDDAORKBJWWYJS-UHFFFAOYSA-N glyphosate Chemical compound OC(=O)CNCP(O)(O)=O XDDAORKBJWWYJS-UHFFFAOYSA-N 0.000 description 3
- 238000003306 harvesting Methods 0.000 description 3
- 230000002363 herbicidal effect Effects 0.000 description 3
- 239000004009 herbicide Substances 0.000 description 3
- 239000005556 hormone Substances 0.000 description 3
- 229940088597 hormone Drugs 0.000 description 3
- 230000001939 inductive effect Effects 0.000 description 3
- JUTMAMXOAOYKHT-UHFFFAOYSA-N karrikinolide Natural products C1=COC=C2OC(=O)C(C)=C21 JUTMAMXOAOYKHT-UHFFFAOYSA-N 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 125000004433 nitrogen atom Chemical group N* 0.000 description 3
- 239000002245 particle Substances 0.000 description 3
- 230000008635 plant growth Effects 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 230000001850 reproductive effect Effects 0.000 description 3
- 230000004044 response Effects 0.000 description 3
- 238000007363 ring formation reaction Methods 0.000 description 3
- 229920002994 synthetic fiber Polymers 0.000 description 3
- HPGGPRDJHPYFRM-UHFFFAOYSA-J tin(iv) chloride Chemical compound Cl[Sn](Cl)(Cl)Cl HPGGPRDJHPYFRM-UHFFFAOYSA-J 0.000 description 3
- 239000013598 vector Substances 0.000 description 3
- QXTUQXRFEBHUBA-UHFFFAOYSA-N (+)-5-deoxystrigol Natural products O1C(=O)C(C)=CC1OC=C1C(=O)OC2C(C(C)(C)CCC3)=C3CC21 QXTUQXRFEBHUBA-UHFFFAOYSA-N 0.000 description 2
- PLQMEXSCSAIXGB-SAXRGWBVSA-N (+)-artemisinic acid Chemical compound C1=C(C)CC[C@H]2[C@H](C)CC[C@@H](C(=C)C(O)=O)[C@H]21 PLQMEXSCSAIXGB-SAXRGWBVSA-N 0.000 description 2
- FQTLCLSUCSAZDY-GOFCXVBSSA-N (3R,6E)-nerolidol Chemical compound CC(C)=CCC\C(C)=C\CC[C@@](C)(O)C=C FQTLCLSUCSAZDY-GOFCXVBSSA-N 0.000 description 2
- HSJKGGMUJITCBW-UHFFFAOYSA-N 3-hydroxybutanal Chemical compound CC(O)CC=O HSJKGGMUJITCBW-UHFFFAOYSA-N 0.000 description 2
- HTTKFVOETMSRQY-UHFFFAOYSA-N 4-iodo-2h-furan-5-one Chemical compound IC1=CCOC1=O HTTKFVOETMSRQY-UHFFFAOYSA-N 0.000 description 2
- OQYBLUDOOFOBPO-UHFFFAOYSA-N Asterolide Natural products C1C2C(=C)CCCC2(C)CC2C1=C(C)C(=O)O2 OQYBLUDOOFOBPO-UHFFFAOYSA-N 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- 102000004127 Cytokines Human genes 0.000 description 2
- 108090000695 Cytokines Proteins 0.000 description 2
- CCOGMNXJHQYPKW-UHFFFAOYSA-N Desacetyldihydro-beta-cycloisopyrethrosin Natural products CC1C2C(O)CC3(C)C(O)CCC(=C)C3C2OC1=O CCOGMNXJHQYPKW-UHFFFAOYSA-N 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- 239000005562 Glyphosate Substances 0.000 description 2
- 244000020551 Helianthus annuus Species 0.000 description 2
- 235000003222 Helianthus annuus Nutrition 0.000 description 2
- PATCUDSVUMFCMB-UHFFFAOYSA-N Lettucenin A Chemical compound C1=CC2=C(C=O)C(=O)OC2=C2C(=C)CCC2=C1C PATCUDSVUMFCMB-UHFFFAOYSA-N 0.000 description 2
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 2
- 241000235070 Saccharomyces Species 0.000 description 2
- 240000000111 Saccharum officinarum Species 0.000 description 2
- 235000007201 Saccharum officinarum Nutrition 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 241000201402 Striga gesnerioides Species 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- 240000004460 Tanacetum coccineum Species 0.000 description 2
- LIDPBIULZNRIJE-QHSBEEBCSA-N Telekin Chemical compound C1CCC(=C)[C@]2(O)C[C@@H]3C(=C)C(=O)O[C@@H]3C[C@]21C LIDPBIULZNRIJE-QHSBEEBCSA-N 0.000 description 2
- 241000482268 Zea mays subsp. mays Species 0.000 description 2
- AOHYMVJOAOUQHB-XDFQPYMHSA-N [(3e,3ar,5s,8bs)-8,8-dimethyl-3-[[(2r)-4-methyl-5-oxo-2h-furan-2-yl]oxymethylidene]-2-oxo-3a,4,5,6,7,8b-hexahydroindeno[1,2-b]furan-5-yl] acetate Chemical compound O([C@H]1[C@@H]2CC3=C1C(C)(C)CC[C@@H]3OC(=O)C)C(=O)\C2=C\O[C@@H]1OC(=O)C(C)=C1 AOHYMVJOAOUQHB-XDFQPYMHSA-N 0.000 description 2
- DLRIUVHQJRZTMZ-CQMYTRALSA-N [(3e,3as,4s,8bs)-8,8-dimethyl-3-[[(2r)-4-methyl-5-oxo-2h-furan-2-yl]oxymethylidene]-2-oxo-3a,4,5,6,7,8b-hexahydroindeno[1,2-b]furan-4-yl] acetate Chemical compound O([C@H]1[C@@H]2[C@@H](C3=C1C(CCC3)(C)C)OC(=O)C)C(=O)\C2=C\O[C@@H]1OC(=O)C(C)=C1 DLRIUVHQJRZTMZ-CQMYTRALSA-N 0.000 description 2
- 230000006978 adaptation Effects 0.000 description 2
- XXROGKLTLUQVRX-UHFFFAOYSA-N allyl alcohol Chemical compound OCC=C XXROGKLTLUQVRX-UHFFFAOYSA-N 0.000 description 2
- TUFPZQHDPZYIEX-UHFFFAOYSA-N alpha-Santonin Natural products C1CC2(C)C=CC(=O)C=C2C2C1C(C)C(=O)O2 TUFPZQHDPZYIEX-UHFFFAOYSA-N 0.000 description 2
- 238000003339 best practice Methods 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 244000038559 crop plants Species 0.000 description 2
- 238000012258 culturing Methods 0.000 description 2
- 239000008121 dextrose Substances 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 230000008641 drought stress Effects 0.000 description 2
- 239000004495 emulsifiable concentrate Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 229930000731 eudesmanolide Natural products 0.000 description 2
- 238000009313 farming Methods 0.000 description 2
- 230000004720 fertilization Effects 0.000 description 2
- 235000021393 food security Nutrition 0.000 description 2
- 230000000855 fungicidal effect Effects 0.000 description 2
- 239000000417 fungicide Substances 0.000 description 2
- 230000014509 gene expression Effects 0.000 description 2
- 229940097068 glyphosate Drugs 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- 238000010348 incorporation Methods 0.000 description 2
- 239000002917 insecticide Substances 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 239000003621 irrigation water Substances 0.000 description 2
- 150000002596 lactones Chemical class 0.000 description 2
- 239000004530 micro-emulsion Substances 0.000 description 2
- 230000000813 microbial effect Effects 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- 235000019713 millet Nutrition 0.000 description 2
- PSZYNBSKGUBXEH-UHFFFAOYSA-M naphthalene-1-sulfonate Chemical compound C1=CC=C2C(S(=O)(=O)[O-])=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-M 0.000 description 2
- 239000003895 organic fertilizer Substances 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- LLQCRTZROWMVOL-UHFFFAOYSA-N parthenin Natural products CC1CCC2C(=C)C(=O)OC2C2(C)C(=O)C=CC12O LLQCRTZROWMVOL-UHFFFAOYSA-N 0.000 description 2
- 230000000243 photosynthetic effect Effects 0.000 description 2
- 239000005648 plant growth regulator Substances 0.000 description 2
- 230000037039 plant physiology Effects 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 230000003938 response to stress Effects 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 239000013589 supplement Substances 0.000 description 2
- 239000004546 suspension concentrate Substances 0.000 description 2
- 150000003505 terpenes Chemical class 0.000 description 2
- 230000009105 vegetative growth Effects 0.000 description 2
- 239000008096 xylene Substances 0.000 description 2
- LLPYDSMSNNUQCD-UHFFFAOYSA-N (1alpha,3beta,4Z,6alpha,11betaH)-1,3-Dihydroxy-4,10(14)-germacradien-12,6-olide Natural products C1CC(=C)C(O)CC(O)C(C)=CC2OC(=O)C(C)C21 LLPYDSMSNNUQCD-UHFFFAOYSA-N 0.000 description 1
- WHOZNOZYMBRCBL-OUKQBFOZSA-N (2E)-2-Tetradecenal Chemical compound CCCCCCCCCCC\C=C\C=O WHOZNOZYMBRCBL-OUKQBFOZSA-N 0.000 description 1
- BOPADYWRUULRBD-MBICNOSFSA-N (3as,5r,5as,8s,8as,9ar)-8-hydroxy-5,8a-dimethyl-1-methylidene-4,5,5a,6,7,8,9,9a-octahydro-3ah-azuleno[6,5-b]furan-2-one Chemical compound C[C@@H]1C[C@@H]2OC(=O)C(=C)[C@H]2C[C@]2(C)[C@@H](O)CC[C@@H]12 BOPADYWRUULRBD-MBICNOSFSA-N 0.000 description 1
- LLQCRTZROWMVOL-DYDIJXKYSA-N (3as,6s,6ar,9as,9br)-6a-hydroxy-6,9a-dimethyl-3-methylidene-4,5,6,9b-tetrahydro-3ah-azuleno[8,7-b]furan-2,9-dione Chemical compound C[C@H]1CC[C@H]2C(=C)C(=O)O[C@H]2[C@]2(C)C(=O)C=C[C@]12O LLQCRTZROWMVOL-DYDIJXKYSA-N 0.000 description 1
- FQTLCLSUCSAZDY-SDNWHVSQSA-N (6E)-nerolidol Chemical compound CC(C)=CCC\C(C)=C\CCC(C)(O)C=C FQTLCLSUCSAZDY-SDNWHVSQSA-N 0.000 description 1
- MUROMQNYCWNWFJ-UHFFFAOYSA-N 3-Ketone-9beta-Hydroxy-4beta, 11alpha, 13, 15-tetrahydrozaluzanin C Natural products C1C(O)C(=C)C2CC(=O)C(C)C2C2OC(=O)C(C)C21 MUROMQNYCWNWFJ-UHFFFAOYSA-N 0.000 description 1
- XQVSREKNQZKAKU-UHFFFAOYSA-N 4'-demethuylpodophyllotoxin-7-Deoxy Natural products C1CC(C)=CCC(O)C(C)=CC2OC(=O)C(=C)C21 XQVSREKNQZKAKU-UHFFFAOYSA-N 0.000 description 1
- 244000013628 Achillea ptarmica Species 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 235000003129 Ambrosia artemisiifolia var elatior Nutrition 0.000 description 1
- 235000009051 Ambrosia paniculata var. peruviana Nutrition 0.000 description 1
- 241000219194 Arabidopsis Species 0.000 description 1
- 235000003130 Arctium lappa Nutrition 0.000 description 1
- 244000294263 Arctium minus Species 0.000 description 1
- 235000008078 Arctium minus Nutrition 0.000 description 1
- 235000003097 Artemisia absinthium Nutrition 0.000 description 1
- 235000015701 Artemisia arbuscula Nutrition 0.000 description 1
- 235000017731 Artemisia dracunculus ssp. dracunculus Nutrition 0.000 description 1
- 235000002657 Artemisia tridentata Nutrition 0.000 description 1
- 229930192334 Auxin Natural products 0.000 description 1
- 235000007319 Avena orientalis Nutrition 0.000 description 1
- 244000075850 Avena orientalis Species 0.000 description 1
- 241000219310 Beta vulgaris subsp. vulgaris Species 0.000 description 1
- 239000002028 Biomass Substances 0.000 description 1
- 240000002791 Brassica napus Species 0.000 description 1
- 235000004977 Brassica sinapistrum Nutrition 0.000 description 1
- 101150094887 CCD8 gene Proteins 0.000 description 1
- JFLRKDZMHNBDQS-UCQUSYKYSA-N CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C(=C[C@H]3[C@@H]2CC(=O)O1)C)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C.CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C=C[C@H]3C2CC(=O)O1)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C Chemical compound CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C(=C[C@H]3[C@@H]2CC(=O)O1)C)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C.CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C=C[C@H]3C2CC(=O)O1)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C JFLRKDZMHNBDQS-UCQUSYKYSA-N 0.000 description 1
- 240000004160 Capsicum annuum Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 235000003255 Carthamus tinctorius Nutrition 0.000 description 1
- 244000020518 Carthamus tinctorius Species 0.000 description 1
- 241000752190 Centaurea solstitialis Species 0.000 description 1
- 235000007866 Chamaemelum nobile Nutrition 0.000 description 1
- 240000003538 Chamaemelum nobile Species 0.000 description 1
- 244000281762 Chenopodium ambrosioides Species 0.000 description 1
- 240000006162 Chenopodium quinoa Species 0.000 description 1
- 229920001661 Chitosan Polymers 0.000 description 1
- 241000195628 Chlorophyta Species 0.000 description 1
- 235000007516 Chrysanthemum Nutrition 0.000 description 1
- 235000008495 Chrysanthemum leucanthemum Nutrition 0.000 description 1
- 235000000604 Chrysanthemum parthenium Nutrition 0.000 description 1
- 244000189548 Chrysanthemum x morifolium Species 0.000 description 1
- 235000008733 Citrus aurantifolia Nutrition 0.000 description 1
- 244000019459 Cynara cardunculus Species 0.000 description 1
- 235000019106 Cynara scolymus Nutrition 0.000 description 1
- 101150025945 D27 gene Proteins 0.000 description 1
- 240000008570 Digitaria exilis Species 0.000 description 1
- 239000001692 EU approved anti-caking agent Substances 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 235000009419 Fagopyrum esculentum Nutrition 0.000 description 1
- 240000008620 Fagopyrum esculentum Species 0.000 description 1
- 235000019715 Fonio Nutrition 0.000 description 1
- 235000008100 Ginkgo biloba Nutrition 0.000 description 1
- 244000194101 Ginkgo biloba Species 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- XJHMNKQRSRSCRT-UHFFFAOYSA-N Hanphyllin Natural products CC1=C/CC(O)C(=CCC2OC(=O)C(=C)C2C1)C XJHMNKQRSRSCRT-UHFFFAOYSA-N 0.000 description 1
- 235000006900 Helenium autumnale Nutrition 0.000 description 1
- 240000005979 Hordeum vulgare Species 0.000 description 1
- 235000007340 Hordeum vulgare Nutrition 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 240000007232 Illicium verum Species 0.000 description 1
- 235000008227 Illicium verum Nutrition 0.000 description 1
- 241000189522 Iva Species 0.000 description 1
- 241000235058 Komagataella pastoris Species 0.000 description 1
- 240000008415 Lactuca sativa Species 0.000 description 1
- 235000003228 Lactuca sativa Nutrition 0.000 description 1
- TZXUUTQEICXCOH-RYGBUWAXSA-N Lactuside A Chemical compound O([C@H]\1C\C=C(C=O)/CC[C@H]2[C@@H](C(O[C@@H]2/C=C/1C)=O)C)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O TZXUUTQEICXCOH-RYGBUWAXSA-N 0.000 description 1
- CLLVDTCVSNACGX-UHFFFAOYSA-N Lactuside A Natural products CC1C2CCC(=CCC(OC(=O)CN)C(=CC2OC1=O)C)C=O CLLVDTCVSNACGX-UHFFFAOYSA-N 0.000 description 1
- VMOFNEIWZSLTBX-UHFFFAOYSA-N Laurenobiolide Natural products COC(=O)C1C=C(/C)CCC=C(/C)CC2OC(=O)C(=C)C12 VMOFNEIWZSLTBX-UHFFFAOYSA-N 0.000 description 1
- 235000017858 Laurus nobilis Nutrition 0.000 description 1
- 244000147568 Laurus nobilis Species 0.000 description 1
- 229920001732 Lignosulfonate Polymers 0.000 description 1
- UPYKUZBSLRQECL-UKMVMLAPSA-N Lycopene Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C1C(=C)CCCC1(C)C)C=CC=C(/C)C=CC2C(=C)CCCC2(C)C UPYKUZBSLRQECL-UKMVMLAPSA-N 0.000 description 1
- JEVVKJMRZMXFBT-XWDZUXABSA-N Lycophyll Natural products OC/C(=C/CC/C(=C\C=C\C(=C/C=C/C(=C\C=C\C=C(/C=C/C=C(\C=C\C=C(/CC/C=C(/CO)\C)\C)/C)\C)/C)\C)/C)/C JEVVKJMRZMXFBT-XWDZUXABSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 240000003183 Manihot esculenta Species 0.000 description 1
- 235000016735 Manihot esculenta subsp esculenta Nutrition 0.000 description 1
- 235000007232 Matricaria chamomilla Nutrition 0.000 description 1
- TZXUUTQEICXCOH-UHFFFAOYSA-N Melampolide glucoside Natural products CC1=CC2OC(=O)C(C)C2CCC(C=O)=CCC1OC1OC(CO)C(O)C(O)C1O TZXUUTQEICXCOH-UHFFFAOYSA-N 0.000 description 1
- 241000372535 Mikania guaco Species 0.000 description 1
- QMGVPVSNSZLJIA-UHFFFAOYSA-N Nux Vomica Natural products C1C2C3C4N(C=5C6=CC=CC=5)C(=O)CC3OCC=C2CN2C1C46CC2 QMGVPVSNSZLJIA-UHFFFAOYSA-N 0.000 description 1
- 240000007817 Olea europaea Species 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 241000520272 Pantoea Species 0.000 description 1
- 241000588696 Pantoea ananatis Species 0.000 description 1
- LLQCRTZROWMVOL-JISBIHODSA-N Parthenin Chemical compound C[C@H]1CC[C@H]2C(=C)C(=O)O[C@H]2[C@]2(C)C(=O)C=C[C@@]12O LLQCRTZROWMVOL-JISBIHODSA-N 0.000 description 1
- 241001495454 Parthenium Species 0.000 description 1
- AVFIYMSJDDGDBQ-UHFFFAOYSA-N Parthenium Chemical compound C1C=C(CCC(C)=O)C(C)CC2OC(=O)C(=C)C21 AVFIYMSJDDGDBQ-UHFFFAOYSA-N 0.000 description 1
- BUQLXKSONWUQAC-UHFFFAOYSA-N Parthenolide Natural products CC1C2OC(=O)C(=C)C2CCC(=C/CCC1(C)O)C BUQLXKSONWUQAC-UHFFFAOYSA-N 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 235000002911 Salvia sclarea Nutrition 0.000 description 1
- 241000013709 Salvia yosgadensis Species 0.000 description 1
- LSMIOFMZNVEEBR-KIZPAXIPSA-N Scilliroside Natural products O=C(O[C@@H]1C=2[C@@](C)([C@@H]3[C@](O)([C@]4(O)[C@@](C)([C@H](C5=COC(=O)C=C5)CC4)CC3)C1)CC[C@H](O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1)C=2)C LSMIOFMZNVEEBR-KIZPAXIPSA-N 0.000 description 1
- 241000209056 Secale Species 0.000 description 1
- 235000007238 Secale cereale Nutrition 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 235000002595 Solanum tuberosum Nutrition 0.000 description 1
- 244000061456 Solanum tuberosum Species 0.000 description 1
- 244000300264 Spinacia oleracea Species 0.000 description 1
- 235000009337 Spinacia oleracea Nutrition 0.000 description 1
- 239000005930 Spinosad Substances 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 235000021536 Sugar beet Nutrition 0.000 description 1
- ULUAUXLGCMPNKK-UHFFFAOYSA-N Sulfobutanedioic acid Chemical compound OC(=O)CC(C(O)=O)S(O)(=O)=O ULUAUXLGCMPNKK-UHFFFAOYSA-N 0.000 description 1
- 229940100389 Sulfonylurea Drugs 0.000 description 1
- 239000005864 Sulphur Substances 0.000 description 1
- 235000005865 Symphytum officinale Nutrition 0.000 description 1
- 240000002299 Symphytum officinale Species 0.000 description 1
- CNIULSUYTFOEHN-LJDNLLIVSA-N Tenulin Natural products O=C1[C@]2(C)[C@](O)(C)O[C@H]3[C@@]4(C)C(=O)C=C[C@@H]4[C@H](C)C[C@H](O1)[C@@H]23 CNIULSUYTFOEHN-LJDNLLIVSA-N 0.000 description 1
- 229910003074 TiCl4 Inorganic materials 0.000 description 1
- 235000011941 Tilia x europaea Nutrition 0.000 description 1
- 235000019714 Triticale Nutrition 0.000 description 1
- 235000013018 Vernonia anthelmintica Nutrition 0.000 description 1
- 244000145469 Vernonia anthelmintica Species 0.000 description 1
- 235000010726 Vigna sinensis Nutrition 0.000 description 1
- 244000042314 Vigna unguiculata Species 0.000 description 1
- QYKIQEUNHZKYBP-UHFFFAOYSA-N Vinyl ether Chemical compound C=COC=C QYKIQEUNHZKYBP-UHFFFAOYSA-N 0.000 description 1
- 244000067505 Xanthium strumarium Species 0.000 description 1
- ROVGZAWFACYCSP-MQBLHHJJSA-N [2-methyl-4-oxo-3-[(2z)-penta-2,4-dienyl]cyclopent-2-en-1-yl] (1r,3r)-2,2-dimethyl-3-(2-methylprop-1-enyl)cyclopropane-1-carboxylate Chemical compound CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)OC1C(C)=C(C\C=C/C=C)C(=O)C1 ROVGZAWFACYCSP-MQBLHHJJSA-N 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 239000000642 acaricide Substances 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 230000008649 adaptation response Effects 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 238000005054 agglomeration Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 239000003619 algicide Substances 0.000 description 1
- 238000005904 alkaline hydrolysis reaction Methods 0.000 description 1
- 229930013930 alkaloid Natural products 0.000 description 1
- 150000004996 alkyl benzenes Chemical class 0.000 description 1
- OENHQHLEOONYIE-UKMVMLAPSA-N all-trans beta-carotene Natural products CC=1CCCC(C)(C)C=1/C=C/C(/C)=C/C=C/C(/C)=C/C=C/C=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C OENHQHLEOONYIE-UKMVMLAPSA-N 0.000 description 1
- XJHDMGJURBVLLE-BOCCBSBMSA-N alpha-santonin Chemical compound C([C@]1(C)CC2)=CC(=O)C(C)=C1[C@@H]1[C@@H]2[C@H](C)C(=O)O1 XJHDMGJURBVLLE-BOCCBSBMSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 235000003484 annual ragweed Nutrition 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 230000003466 anti-cipated effect Effects 0.000 description 1
- 230000002528 anti-freeze Effects 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 239000001138 artemisia absinthium Substances 0.000 description 1
- LZMOBPWDHUQTKL-RWMBFGLXSA-N artemisinic acid Natural products CC1=C[C@@H]2[C@@H](CCC[C@H]2C(=C)C(=O)O)CC1 LZMOBPWDHUQTKL-RWMBFGLXSA-N 0.000 description 1
- BLUAFEHZUWYNDE-NNWCWBAJSA-N artemisinin Chemical compound C([C@](OO1)(C)O2)C[C@H]3[C@H](C)CC[C@@H]4[C@@]31[C@@H]2OC(=O)[C@@H]4C BLUAFEHZUWYNDE-NNWCWBAJSA-N 0.000 description 1
- 229930101531 artemisinin Natural products 0.000 description 1
- 229960004191 artemisinin Drugs 0.000 description 1
- PLQMEXSCSAIXGB-UHFFFAOYSA-N artemisininic acid Natural products C1=C(C)CCC2C(C)CCC(C(=C)C(O)=O)C21 PLQMEXSCSAIXGB-UHFFFAOYSA-N 0.000 description 1
- 235000016520 artichoke thistle Nutrition 0.000 description 1
- 239000002363 auxin Substances 0.000 description 1
- 239000003899 bactericide agent Substances 0.000 description 1
- 239000000440 bentonite Substances 0.000 description 1
- 229910000278 bentonite Inorganic materials 0.000 description 1
- SVPXDRXYRYOSEX-UHFFFAOYSA-N bentoquatam Chemical compound O.O=[Si]=O.O=[Al]O[Al]=O SVPXDRXYRYOSEX-UHFFFAOYSA-N 0.000 description 1
- 229940077388 benzenesulfonate Drugs 0.000 description 1
- LUHMMHZLDLBAKX-UHFFFAOYSA-N beta-Santonin Natural products O=C1C=CC2(C)CC(O)C3C(C)C(=O)OC3C2=C1C LUHMMHZLDLBAKX-UHFFFAOYSA-N 0.000 description 1
- 235000013734 beta-carotene Nutrition 0.000 description 1
- 239000011648 beta-carotene Substances 0.000 description 1
- TUPZEYHYWIEDIH-WAIFQNFQSA-N beta-carotene Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C1=C(C)CCCC1(C)C)C=CC=C(/C)C=CC2=CCCCC2(C)C TUPZEYHYWIEDIH-WAIFQNFQSA-N 0.000 description 1
- XJHDMGJURBVLLE-OMSPQPPYSA-N beta-santonin Chemical compound C([C@]1(C)CC2)=CC(=O)C(C)=C1[C@@H]1[C@@H]2[C@@H](C)C(=O)O1 XJHDMGJURBVLLE-OMSPQPPYSA-N 0.000 description 1
- 229960002747 betacarotene Drugs 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000008236 biological pathway Effects 0.000 description 1
- 230000003592 biomimetic effect Effects 0.000 description 1
- VEMKTZHHVJILDY-UXHICEINSA-N bioresmethrin Chemical compound CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)OCC1=COC(CC=2C=CC=CC=2)=C1 VEMKTZHHVJILDY-UXHICEINSA-N 0.000 description 1
- 150000001647 brassinosteroids Chemical class 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 244000309464 bull Species 0.000 description 1
- 235000006263 bur ragweed Nutrition 0.000 description 1
- LGJQJUDAOYMIQX-UHFFFAOYSA-N cadinanolide Natural products CCOCC1=C2C(CC(C)(OC(=O)C)C3(O)CCC(C)(O)C(OC(=O)C)C23OC1=O)OC(=O)C LGJQJUDAOYMIQX-UHFFFAOYSA-N 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- VTYYLEPIZMXCLO-UHFFFAOYSA-L calcium carbonate Substances [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 235000019519 canola oil Nutrition 0.000 description 1
- 239000000828 canola oil Substances 0.000 description 1
- 239000000152 carbamate pesticide Substances 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 235000021466 carotenoid Nutrition 0.000 description 1
- 150000001747 carotenoids Chemical class 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000025084 cell cycle arrest Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000005660 chlorination reaction Methods 0.000 description 1
- 238000011097 chromatography purification Methods 0.000 description 1
- 235000019506 cigar Nutrition 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 235000003488 common ragweed Nutrition 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 230000000875 corresponding effect Effects 0.000 description 1
- 238000009347 cover cropping Methods 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- SKNVIAFTENCNGB-UHFFFAOYSA-N dehydroleucodine Natural products C1CC2C(=C)C(=O)OC2C2C(C)=CC(=O)C2=C1C SKNVIAFTENCNGB-UHFFFAOYSA-N 0.000 description 1
- 230000002939 deleterious effect Effects 0.000 description 1
- GUJOJGAPFQRJSV-UHFFFAOYSA-N dialuminum;dioxosilane;oxygen(2-);hydrate Chemical compound O.[O-2].[O-2].[O-2].[Al+3].[Al+3].O=[Si]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O GUJOJGAPFQRJSV-UHFFFAOYSA-N 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 235000013399 edible fruits Nutrition 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002085 enols Chemical class 0.000 description 1
- 230000006353 environmental stress Effects 0.000 description 1
- 230000006718 epigenetic regulation Effects 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- WOAQOADWJMRARB-XSFVSMFZSA-N ethyl (E)-2-(1-but-3-enyl-3-cyanoisoindol-2-yl)-3-[(4-methyl-5-oxo-2H-furan-2-yl)oxy]prop-2-enoate Chemical compound C=CCCC1=C2C=CC=CC2=C(C#N)N1/C(C(=O)OCC)=C/OC1OC(=O)C(C)=C1 WOAQOADWJMRARB-XSFVSMFZSA-N 0.000 description 1
- 235000019439 ethyl acetate Nutrition 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000001125 extrusion Methods 0.000 description 1
- 235000008384 feverfew Nutrition 0.000 description 1
- 244000037666 field crops Species 0.000 description 1
- 235000013312 flour Nutrition 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000013505 freshwater Substances 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 229930015714 guaianolide Natural products 0.000 description 1
- 150000004273 guaianolide derivatives Chemical class 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 229930016880 heliangolide Natural products 0.000 description 1
- 150000002550 heliangolide derivatives Chemical class 0.000 description 1
- 235000008216 herbs Nutrition 0.000 description 1
- 238000011905 homologation Methods 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- MRMGABUTBNWSLA-UHFFFAOYSA-N hymenine Natural products N1C(N)=NC(C2C=3C(Br)=C(Br)NC=3C(=O)NCC2)=C1 MRMGABUTBNWSLA-UHFFFAOYSA-N 0.000 description 1
- 208000006278 hypochromic anemia Diseases 0.000 description 1
- APCVEXBOZSBQLY-UHFFFAOYSA-N hypocretenolide Natural products CC1=C2C(=O)C=C(C)C23CC(CC1)C(=C)C(=O)O3 APCVEXBOZSBQLY-UHFFFAOYSA-N 0.000 description 1
- 150000004153 hypocretenolide derivatives Chemical class 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000015788 innate immune response Effects 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 235000021374 legumes Nutrition 0.000 description 1
- 239000004571 lime Substances 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 238000002370 liquid polymer infiltration Methods 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 235000012661 lycopene Nutrition 0.000 description 1
- 239000001751 lycopene Substances 0.000 description 1
- OAIJSZIZWZSQBC-GYZMGTAESA-N lycopene Chemical compound CC(C)=CCC\C(C)=C\C=C\C(\C)=C\C=C\C(\C)=C\C=C\C=C(/C)\C=C\C=C(/C)\C=C\C=C(/C)CCC=C(C)C OAIJSZIZWZSQBC-GYZMGTAESA-N 0.000 description 1
- 229960004999 lycopene Drugs 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical class [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 235000011160 magnesium carbonates Nutrition 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- GBMDVOWEEQVZKZ-UHFFFAOYSA-N methanol;hydrate Chemical compound O.OC GBMDVOWEEQVZKZ-UHFFFAOYSA-N 0.000 description 1
- 201000002266 mite infestation Diseases 0.000 description 1
- 239000003750 molluscacide Substances 0.000 description 1
- 230000002013 molluscicidal effect Effects 0.000 description 1
- 229910052901 montmorillonite Inorganic materials 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 239000005645 nematicide Substances 0.000 description 1
- 239000000618 nitrogen fertilizer Substances 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 235000014571 nuts Nutrition 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 125000001117 oleyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C([H])=C([H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000003992 organochlorine insecticide Substances 0.000 description 1
- 239000003987 organophosphate pesticide Substances 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- KTEXNACQROZXEV-PVLRGYAZSA-N parthenolide Chemical compound C1CC(/C)=C/CC[C@@]2(C)O[C@@H]2[C@H]2OC(=O)C(=C)[C@@H]21 KTEXNACQROZXEV-PVLRGYAZSA-N 0.000 description 1
- 229940069510 parthenolide Drugs 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 230000000361 pesticidal effect Effects 0.000 description 1
- 229940044654 phenolsulfonic acid Drugs 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 239000002686 phosphate fertilizer Substances 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000003375 plant hormone Substances 0.000 description 1
- 229920005646 polycarboxylate Polymers 0.000 description 1
- 235000013824 polyphenols Nutrition 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 210000001236 prokaryotic cell Anatomy 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 229930009585 pseudoguaianolide Natural products 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 239000002728 pyrethroid Substances 0.000 description 1
- 229940015367 pyrethrum Drugs 0.000 description 1
- 229910052903 pyrophyllite Inorganic materials 0.000 description 1
- 235000009736 ragweed Nutrition 0.000 description 1
- 230000008707 rearrangement Effects 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 238000012827 research and development Methods 0.000 description 1
- 230000025508 response to water Effects 0.000 description 1
- 230000025469 response to water deprivation Effects 0.000 description 1
- 238000013349 risk mitigation Methods 0.000 description 1
- 239000003128 rodenticide Substances 0.000 description 1
- 238000005096 rolling process Methods 0.000 description 1
- 239000001691 salvia sclarea Substances 0.000 description 1
- 238000013341 scale-up Methods 0.000 description 1
- LSMIOFMZNVEEBR-ICLSSMQGSA-N scilliroside Chemical compound C=1([C@@H]2[C@@]3(C)CC[C@H]4[C@@]([C@]3(CC2)O)(O)C[C@H](C2=C[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O3)O)CC[C@@]24C)OC(=O)C)C=CC(=O)OC=1 LSMIOFMZNVEEBR-ICLSSMQGSA-N 0.000 description 1
- 230000009291 secondary effect Effects 0.000 description 1
- 229930004725 sesquiterpene Natural products 0.000 description 1
- 150000004354 sesquiterpene derivatives Chemical class 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 235000004833 smallhead sneezeweed Nutrition 0.000 description 1
- 235000006891 sneezeweed Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000004550 soluble concentrate Substances 0.000 description 1
- 235000013599 spices Nutrition 0.000 description 1
- 229940014213 spinosad Drugs 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 239000000021 stimulant Substances 0.000 description 1
- 230000015378 stomatal closure Effects 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N succinic acid Chemical compound OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- YROXIXLRRCOBKF-UHFFFAOYSA-N sulfonylurea Chemical class OC(=N)N=S(=O)=O YROXIXLRRCOBKF-UHFFFAOYSA-N 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 229940104261 taurate Drugs 0.000 description 1
- XOAAWQZATWQOTB-UHFFFAOYSA-N taurine Chemical compound NCCS(O)(=O)=O XOAAWQZATWQOTB-UHFFFAOYSA-N 0.000 description 1
- CNIULSUYTFOEHN-AYUOKEMLSA-N tenulin Chemical compound C[C@@H]([C@H]1[C@@]2(C)C(C=C1)=O)C[C@@H]1OC(=O)[C@@]3(C)[C@](O)(C)O[C@@H]2[C@@H]13 CNIULSUYTFOEHN-AYUOKEMLSA-N 0.000 description 1
- 235000007586 terpenes Nutrition 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 239000004753 textile Substances 0.000 description 1
- XJDNKRIXUMDJCW-UHFFFAOYSA-J titanium tetrachloride Chemical compound Cl[Ti](Cl)(Cl)Cl XJDNKRIXUMDJCW-UHFFFAOYSA-J 0.000 description 1
- ZCIHMQAPACOQHT-ZGMPDRQDSA-N trans-isorenieratene Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/c1c(C)ccc(C)c1C)C=CC=C(/C)C=Cc2c(C)ccc(C)c2C ZCIHMQAPACOQHT-ZGMPDRQDSA-N 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical class [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 239000012873 virucide Substances 0.000 description 1
- 238000011179 visual inspection Methods 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 239000004563 wettable powder Substances 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 241000228158 x Triticosecale Species 0.000 description 1
- OENHQHLEOONYIE-JLTXGRSLSA-N β-Carotene Chemical compound CC=1CCCC(C)(C)C=1\C=C\C(\C)=C\C=C\C(\C)=C\C=C\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C OENHQHLEOONYIE-JLTXGRSLSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D407/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00
- C07D407/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00 containing two hetero rings
- C07D407/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/04—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom
- A01N43/06—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings
- A01N43/08—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings with oxygen as the ring hetero atom
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/04—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom
- A01N43/06—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings
- A01N43/12—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings condensed with a carbocyclic ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/34—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D307/56—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D307/60—Two oxygen atoms, e.g. succinic anhydride
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/92—Naphthofurans; Hydrogenated naphthofurans
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/93—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems condensed with a ring other than six-membered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/0004—Oxidoreductases (1.)
- C12N9/001—Oxidoreductases (1.) acting on the CH-CH group of donors (1.3)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/0004—Oxidoreductases (1.)
- C12N9/0069—Oxidoreductases (1.) acting on single donors with incorporation of molecular oxygen, i.e. oxygenases (1.13)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/10—Transferases (2.)
- C12N9/1085—Transferases (2.) transferring alkyl or aryl groups other than methyl groups (2.5)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/90—Isomerases (5.)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y103/00—Oxidoreductases acting on the CH-CH group of donors (1.3)
- C12Y103/99—Oxidoreductases acting on the CH-CH group of donors (1.3) with other acceptors (1.3.99)
- C12Y103/99031—Phytoene desaturase (lycopene-forming) (1.3.99.31)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y113/00—Oxidoreductases acting on single donors with incorporation of molecular oxygen (oxygenases) (1.13)
- C12Y113/11—Oxidoreductases acting on single donors with incorporation of molecular oxygen (oxygenases) (1.13) with incorporation of two atoms of oxygen (1.13.11)
- C12Y113/11068—9-Cis-beta-carotene 9',10'-cleaving dioxygenase (1.13.11.68)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y113/00—Oxidoreductases acting on single donors with incorporation of molecular oxygen (oxygenases) (1.13)
- C12Y113/11—Oxidoreductases acting on single donors with incorporation of molecular oxygen (oxygenases) (1.13) with incorporation of two atoms of oxygen (1.13.11)
- C12Y113/1107—All-trans-10'-apo-beta-carotenal 13,14-cleaving dioxygenase (1.13.11.70)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y205/00—Transferases transferring alkyl or aryl groups, other than methyl groups (2.5)
- C12Y205/01—Transferases transferring alkyl or aryl groups, other than methyl groups (2.5) transferring alkyl or aryl groups, other than methyl groups (2.5.1)
- C12Y205/01029—Geranylgeranyl diphosphate synthase (2.5.1.29)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y205/00—Transferases transferring alkyl or aryl groups, other than methyl groups (2.5)
- C12Y205/01—Transferases transferring alkyl or aryl groups, other than methyl groups (2.5) transferring alkyl or aryl groups, other than methyl groups (2.5.1)
- C12Y205/01099—All-trans-phytoene synthase (2.5.1.99)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y502/00—Cis-trans-isomerases (5.2)
- C12Y502/01—Cis-trans-Isomerases (5.2.1)
- C12Y502/01014—Beta-carotene isomerase (5.2.1.14)
Definitions
- Strigolactones are a recently discovered class of hormones known to regulate development and stress response. Strigolactone has not been evaluated as a product due to its high cost of production.
- the plant propagation materials may comprise strigolactone. Alternatively, or additionally, the plant propagation materials comprise chemical mimics of strigolactone.
- the plant propagation materials may be administered to plants, such as maize. Plants treated with strigolactones may show a significant resistance to the adverse effects of water-limited conditions. In addition, plants treated with strigolactones may show increased plant yield. Further disclosed herein are uses of plant propagation materials.
- R 1 , R 2 , R 4 , R 5 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , and R 17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ;
- R 3 and R 6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ; or R 3 and R 6 together form a direct bond to provide a double bond;
- each R 18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, or —C(O)R 19 ;
- each R 19 is independently alkyl, haloalkyl, aryl, or heteroaryl;
- m is 0, 1, or 2; and n is 1 or 2.
- m is 0 and n is 1. In some instances, m is 0 and n is 2. In some instances, m is 1 and n is 2. In some instances, m is 2 and n is 1. In some instances, m is 2 and n is 2. In some instances, m is 1 and n is 1.
- R 1 , R 2 , R 4 , R 5 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , and R 17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ;
- R 3 and R 6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ; or R 3 and R 6 together form a direct bond to provide a double bond;
- each R 18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, or —C(O)R 19 ; and each R 19 is independently alkyl, haloalkyl, aryl, or heteroaryl
- R 1 , R 2 , R 7 , R 8 , R 9 , R 16 , and R 17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ;
- R 3 and R 6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ; or R 3 and R 6 together form a direct bond to provide a double bond;
- each R 18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, or —C(O)R 19 ; and each R 19 is independently alkyl, haloalkyl, aryl, or heteroaryl.
- R 1 , R 2 , R 4 , R 5 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , and R 17 are each independently H, alkyl, or —OR 18 .
- R 3 and R 6 together form a direct bond to provide a double bond.
- R 3 and R 6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 .
- R 3 and R 6 are each independently H, alkyl, or —OR 18 .
- R 17 is alkyl.
- the chemical mimic of Strigolactone can be compound of Formula (VII), a salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer thereof:
- a, b, c are each independently 0, 1, or 2;
- each A is independently O, or S;
- each E is independently O, S, or —NR 18 ;
- each G is independently C or N;
- R 5 , R 6 , R 11 , R 12 , R 14 , R 15 and R 17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ;
- R 2 , R 3 , R 7 , R 8 , R 9 , and R 10 are each independently H, alkyl, haloalkyl, amino, halo, —OR 18 or a lone electron pair;
- R 1 and R 16 are each independently H, alkyl, haloalkyl, amino, halo, lone electron pair, or —OR 18 ; or R 1 and R 16 together form a direct bond to provide a double bond;
- R 4 and R 13 are each independently H, alkyl, haloalkyl, amino, halo, lone electron pair, or —OR 18 ; or R 1 and R 16 together form a direct bond to provide a double bond;
- each R 18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, —C(O)R 19 or
- each R 19 is independently H, alkyl, haloalkyl, aryl, or heteroaryl.
- Another chemical mimic of Strigolactone can be compound of Formula (VIII), a salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer thereof:
- a, b, c are each independently 0, 1, or 2;
- each A is independently O, or S;
- each E is independently O, S, or —NR 18 ;
- each G is independently C or N;
- R 5 , R 6 , R 11 , R 12 , R 14 , R 15 and R 17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ;
- R 2 , R 3 , R 7 , R 8 , R 9 , and R 10 are each independently H, alkyl, haloalkyl, amino, halo, —OR 18 or a lone electron pair;
- R 1 and R 16 are each independently H, alkyl, haloalkyl, amino, halo, lone electron pair, or —OR 18 ; or R 1 and R 16 together form a direct bond to provide a double bond;
- R 4 and R 13 are each independently H, alkyl, haloalkyl, amino, halo, lone electron pair, or —OR 18 ; or R 1 and R 16 together form a direct bond to provide a double bond;
- each R 18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, —C(O)R 19 or
- each R 19 is independently H, alkyl, haloalkyl, aryl, or heteroaryl.
- each A in the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone is independently O.
- each E in the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone is independently O.
- each G in the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone is independently C.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone wherein R 2 , R 3 , R 4 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , and R 16 are each independently H.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone wherein R 1 , R 5 , R 6 , R 13 , and R 17 are each independently alkyl.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone wherein R 1 , R 5 , R 6 , R 13 , and R 17 are each independently methyl.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer disclosed herein is not (+)-Strigol
- R 4 and R 13 together do not form a direct bond to provide a double bond. In another embodiment, R 4 and R 13 together forms a direct bond to provide a double bond. In another embodiment, b+c equals at least 2. In another embodiment, b is 1 or 2.
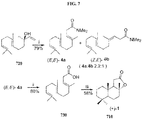
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer may be one of the following:
- the chemical mimic of strigolactone may be a compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer, having the structure of Formula (IX):
- the chemical mimic of strigolactone may be a compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer, having the structure of Formula (X):
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer disclosed herein may have a diastereomeric excess of at least about 15%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 85%, 90%, 95%, or 99%.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer disclosed herein may have a diastereomeric excess of about 15%-99%, 20%-99%, 30%-99%, 40-99%, 50-99%, 60-99%, 70-99%, 80-99%, 90-99%, 15%-90%, 20%-90%, 30%-90%, 40-90%, 50-90%, 60-90%, 70-90%, 80-90%, 15%-80%, 20%-80%, 30%-80%, 40-80%, 50-80%, 60-80%, 70-80%, 15%-70%, 20%-70%, 30%-70%, 40-70%, 50-70%, 60-70%, 15%-60%, 20%-60%, 30%-60%, 40-60%, 50-60%, 15%-50%, 20%-50%, 30%-50%, 40-50%, 15%-40%, 20%-40%, 30%-40%, 15%-30%, 20%-30%, or 15-20%.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer or formulation disclosed herein may comprise about: 2, 3, 4, 5, 6, 7, 8, 9, or 10 individual diastereoisomer of Formula I, II, III, IV, V, VI, VII, VIII, IX, or X.
- formulations comprising the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer disclosed herein.
- the formulation may further comprise an excipient.
- the excipient comprises water, a surfactant, an alcohol, or any combination thereof.
- the formulation comprises the surfactant, wherein the surfactant comprises sulfosuccinate, naphthalene sulfonate, sulfated ester, phosphate ester, sulfated alcohol, alkyl benzene sulfonate, polycarboxylate, naphthalene sulfonate condensate, phenol sulfonic acid condensate, lignosulfonate, methyl oleyl taurate, polyvinyl alcohol, or any combination thereof.
- the formulation may further comprise a fertilizer.
- the fertilizer comprises nitrogen fertilizer, phosphate fertilizer, potassium fertilizer, calcium fertilizer, magnesium fertilizer, sulfur fertilizer, compound fertilizer, organic fertilizer, or any combination thereof.
- the formulation may further comprise an insecticide, a fungicide, a herbicide, or any combination thereof.
- the herbicide comprises a glyphosate.
- the glyphosate comprises N-(phosphonomethyl)glycine.
- Disclosed herein can be a method comprising contacting a plant with the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation disclosed herein.
- the contacting the plant comprises administering the compound, salt, solvate, polymorph, stereoisomer, isomer or formulation as a spray.
- the contacting the plant further comprises adding the compound, salt, solvate, polymorph, stereoisomer, isomer or formulation to irrigation water of the plant.
- the plant disclosed herein can be cereals, such as millet, barley, maize, oats, triticale, rye, buckwheat, fonio, quinoa, sorghum, corn, wheat, and rice.
- the plant can be staple crops such as potato, cassava, and legumes.
- the plant can be vegetables, spices, fruits, nuts, herbs, and edible flowers.
- the plant can be sugar cane and sugar beet.
- the plant can be maize, soybean, rapeseed, safflower, sunflower, and olive.
- the plant is soybean, tomato, soybean, corn, rice, tomato, alfalfa, wheat, green algae or any combination thereof.
- a yield of the contacted plant is increased as compared to an uncontacted plant, a life of the contacted plant is extended as compared to an uncontacted plant, a wilting of the contacted plant is reduced or delayed as compared to an uncontacted plant, a turgidity of the contacted plant is prolonged or maintained as compared to an uncontacted plant, a loss of one or more petals of the contacted plant is reduced or delayed as compared to an uncontacted plant, a chlorophyll content of the contacted plant is maintained as compared to an uncontacted plant, a loss of the chlorophyll content of the contacted plant is reduced or delayed as compared to an uncontacted plant, a chlorophyll content of the contacted plant is increased as compared to an uncontacted plant, a salinity tolerance of the contacted plant is increased as compared to an uncontacted plant, a water consumption of the contacted plant is reduced as compared to an uncontacted plant, a drought tolerance of the contacted plant is increased as compared to an uncontacted plant
- a yield of the contacted plant is increased as compared to an uncontacted plant.
- a life of the contacted plant is extended as compared to an uncontacted plant.
- a wilting of the contacted plant is reduced or delayed as compared to an uncontacted plant.
- a turgidity of the contacted plant is prolonged or maintained as compared to an uncontacted plant.
- a loss of one or more petals of the contacted plant is reduced or delayed as compared to an uncontacted plant.
- a chlorophyll content of the contacted plant is maintained as compared to an uncontacted plant.
- a loss of the chlorophyll content of the contacted plant is reduced or delayed as compared to an uncontacted plant.
- a chlorophyll content of the contacted plant is increased as compared to an uncontacted plant.
- a salinity tolerance of the contacted plant is increased as compared to an uncontacted plant.
- a water consumption of the contacted plant is reduced as compared to an uncontacted plant.
- a drought tolerance of the contacted plant is increased as compared to an uncontacted plant.
- a pest resistance of the contacted plant is increased as compared to an uncontacted plant.
- a pesticides consumption of the contacted plant is reduced as compared to an uncontacted plant.
- the method comprises contacting a plant with the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer or formulation disclosed herein, in an amount effective to increase a yield of the contacted plant as compared to an uncontacted plant, extend a life of the contacted plant as compared to an uncontacted plant, reduce or delay a wilting of the contacted plant as compared to an uncontacted plant, prolong or maintain a turgidity of the contacted plant as compared to an uncontacted plant, reduce or delay a loss of one or more petals of the contacted plant as compared to an uncontacted plant, maintain a chlorophyll content of the contacted plant as compared to an uncontacted plant, reduce or delay a loss of the chlorophyll content of the contacted plant as compared to an uncontacted plant, increase a chlorophyll content of the contacted plant as compared to an uncontacted plant, increase a salinity tolerance of the contacted plant as compared to an uncontacted plant, reduce a
- the method may comprise contacting a plant with the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer or formulation disclosed herein.
- the method contacts the plant in an amount effective to increase a yield of the contacted plant as compared to an uncontacted plant.
- the method contacts the plant in an amount effective to extend a life of the contacted plant as compared to an uncontacted plant.
- the method contacts the plant in an amount effective to reduce or delay a wilting of the contacted plant as compared to an uncontacted plant.
- the method contacts the plant in an amount effective to prolong or maintain a turgidity of the contacted plant as compared to an uncontacted plant.
- the method contacts the plant in an amount effective to reduce or delay a loss of one or more petals of the contacted plant as compared to an uncontacted plant. In another embodiment, the method contacts the plant in an amount effective to maintain a chlorophyll content of the contacted plant as compared to an uncontacted plant. In another embodiment, the method contacts the plant in an amount effective to reduce or delay a loss of the chlorophyll content of the contacted plant as compared to an uncontacted plant. In another embodiment, the method contacts the plant in an amount effective to increase a chlorophyll content of the contacted plant as compared to an uncontacted plant. In another embodiment, the method contacts the plant in an amount effective to increase a salinity tolerance of the contacted plant as compared to an uncontacted plant.
- the method contacts the plant in an amount effective to reduce a water consumption of the contacted plant as compared to an uncontacted plant. In another embodiment, the method contacts the plant in an amount effective to increase a drought tolerance of the contacted plant as compared to an uncontacted plant. In another embodiment, the method contacts the plant in an amount effective to increase a pest resistance of the contacted plant as compared to an uncontacted plant. In another embodiment, the method contacts the plant in an amount effective to reduce a pesticides consumption of the contacted plant as compared to an uncontacted plant.
- the method may comprise increasing the yield of the contacted plant, wherein the yield of the contacted plant is increased by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the yield of the contacted plant may be increased from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the yield of the contacted plant is increased from about 5% to 50% as compared to an uncontacted plant.
- the yield of the contacted plant may be increased under adequately irrigated condition.
- the yield of the contacted plant may be increased under drought condition.
- the method may comprise extending the life of the contacted plant, wherein the life of the contacted plant is extended by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the life of the contacted plant may be extended from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the life of the contacted plant is extended from about 5% to 50% as compared to an uncontacted plant.
- a plant is determined to be dead if the metabolic activity of the plant has ceased. In some embodiments, a plant is determined to be dead if the vegetative growth of the plant has ceased.
- the method may comprise extending the life of the contacted plant, wherein the life of the contacted plant is extended by at least about 6 hours, 12 hours, 24 hours, 36 hours, or 48 hours as compared to an uncontacted plant.
- the life of the contacted plant may be extended by at least about 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 8 days, 9 days, or 10 days as compared to an uncontacted plant.
- the life of the contacted plant may be extended by at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, or 6 weeks as compared to an uncontacted plant.
- the life of the contacted plant may be extended by at least about 1 month, 2 months, 3 months, 4 months, 5 months, or 6 months as compared to an uncontacted plant.
- the life of the contacted plant may be extended from 6 hours to 48 hours as compared to an uncontacted plant.
- the life of the contacted plant may be extended from 1 day to 10 days as compared to an uncontacted plant.
- the life of the contacted plant may be extended from 1 week to 6 weeks as compared to an uncontacted plant.
- the life of the contacted plant may be extended from 1 month to 6 months as compared to an uncontacted plant. In one example, the life of the contacted plant is extended from at least about 6 hours to 1 month as compared to an uncontacted plant.
- the method may comprise reducing the wilting of the contacted plant, wherein the wilting of the contacted plant is reduced by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the wilting of the contacted plant may be reduced from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the wilting of the contacted plant is reduced from about 5% to 50% as compared to an uncontacted plant.
- the wilting can be determined by visual inspection. In some embodiments, the wilting can be determined by the change in leaf angle. The angle between the stem and leaf can change drastically during wilting. For example, it is determined to be wilting when the angle between the stem and leaf changes 10°, 20°, 30°, 40°, 50°, 60°, 70°, 80°, 90°, 100°, 110°, 120°, 130°, 140°, 150°, 160°, 170°, or 180°. In some embodiments, the wilting can be determined by the total leaf volume.
- the method may comprise delaying the wilting of the contacted plant, wherein the wilting of the contacted plant is delayed by at least about 6 hours, 12 hours, 24 hours, 36 hours, or 48 hours as compared to an uncontacted plant.
- the wilting of the contacted plant may be delayed by at least about 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 8 days, 9 days, or 10 days as compared to an uncontacted plant.
- the wilting of the contacted plant may be delayed by at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, or 6 weeks as compared to an uncontacted plant.
- the wilting of the contacted plant may be delayed by at least about 1 month, 2 months, 3 months, 4 months, 5 months, or 6 months as compared to an uncontacted plant.
- the wilting of the contacted plant may be delayed from 6 hours to 48 hours as compared to an uncontacted plant.
- the wilting of the contacted plant may be delayed from 1 day to 10 days as compared to an uncontacted plant.
- the wilting of the contacted plant may be delayed from 1 week to 6 weeks as compared to an uncontacted plant.
- the wilting of the contacted plant may be delayed from 1 month to 6 months as compared to an uncontacted plant.
- the wilting of the contacted plant is delayed from at least about 6 hours to 1 month as compared to an uncontacted plant.
- the method may comprise prolonging or maintaining the turgidity of the contacted plant, wherein the turgidity of the contacted plant is prolonged or maintained by at least about 6 hours, 12 hours, 24 hours, 36 hours, or 48 hours as compared to an uncontacted plant.
- the turgidity of the contacted plant may be prolonged or maintained by at least about 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 8 days, 9 days, or 10 days as compared to an uncontacted plant.
- the turgidity of the contacted plant may be prolonged or maintained by at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, or 6 weeks as compared to an uncontacted plant.
- the turgidity of the contacted plant may be prolonged or maintained by at least about 1 month, 2 months, 3 months, 4 months, 5 months, or 6 months as compared to an uncontacted plant.
- the turgidity of the contacted plant may be prolonged or maintained from 6 hours to 48 hours as compared to an uncontacted plant.
- the turgidity of the contacted plant may be prolonged or maintained from 1 day to 10 days as compared to an uncontacted plant.
- the turgidity of the contacted plant may be prolonged or maintained from 1 week to 6 weeks as compared to an uncontacted plant.
- the turgidity of the contacted plant may be prolonged or maintained from 1 month to 6 months as compared to an uncontacted plant.
- the turgidity of the contacted plant is prolonged or maintained from at least about 6 hours to 1 month as compared to an uncontacted plant.
- the method may comprise reducing the loss of one or more petals of the contacted plant, wherein the loss of one or more petals of the contacted plant is reduced by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant may be reduced from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant is reduced from about 5% to 50% as compared to an uncontacted plant.
- the method may comprise delaying the loss of one or more petals of the contacted plant, wherein the loss of one or more petals of the contacted plant is delayed by at least about 6 hours, 12 hours, 24 hours, 36 hours, or 48 hours as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant may be delayed by at least about 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 8 days, 9 days, or 10 days as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant may be delayed by at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, or 6 weeks as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant may be delayed by at least about 1 month, 2 months, 3 months, 4 months, 5 months, or 6 months as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant may be delayed from 6 hours to 48 hours as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant may be delayed from 1 day to 10 days as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant may be delayed from 1 week to 6 weeks as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant may be delayed from 1 month to 6 months as compared to an uncontacted plant.
- the loss of one or more petals of the contacted plant is delayed from at least about 6 hours to 1 month as compared to an uncontacted plant.
- the method may comprise delaying the chlorophyll content of the contacted plant, wherein the chlorophyll content of the contacted plant is maintained for at least about 6 hours, 12 hours, 24 hours, 36 hours, or 48 hours as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant may be maintained for at least about 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 8 days, 9 days, or 10 days as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant may be maintained for at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, or 6 weeks as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant may be maintained for at least about 1 month, 2 months, 3 months, 4 months, 5 months, or 6 months as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant may be maintained from 6 hours to 48 hours as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant may be maintained from 1 day to 10 days as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant may be maintained from 1 week to 6 weeks as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant may be maintained from 1 month to 6 months as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant is maintained from at least about 6 hours to 1 month as compared to an uncontacted plant.
- the method may comprise delaying the loss of the chlorophyll content of the contacted plant, wherein the loss of the chlorophyll content of the contacted plant is delayed by at least about 6 hours, 12 hours, 24 hours, 36 hours, or 48 hours as compared to an uncontacted plant.
- the loss of the chlorophyll content of the contacted plant may be delayed by at least about 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 8 days, 9 days, or 10 days as compared to an uncontacted plant.
- the loss of the chlorophyll content of the contacted plant may be delayed by at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, or 6 weeks as compared to an uncontacted plant.
- the loss of the chlorophyll content of the contacted plant may be delayed by at least about 1 month, 2 months, 3 months, 4 months, 5 months, or 6 months as compared to an uncontacted plant.
- the loss of the chlorophyll content of the contacted plant may be delayed from 6 hours to 48 hours as compared to an uncontacted plant.
- the loss of the chlorophyll content of the contacted plant may be delayed from 1 day to 10 days as compared to an uncontacted plant.
- the loss of the chlorophyll content of the contacted plant may be delayed from 1 week to 6 weeks as compared to an uncontacted plant.
- the loss of the chlorophyll content of the contacted plant may be delayed from 1 month to 6 months as compared to an uncontacted plant.
- the loss of the chlorophyll content of the contacted plant is delayed from at least about 6 hours to 1 month as compared to an uncontacted plant.
- the chlorophyll can be measured by using a chlorophyll meter, such as a SPAD 502 PLUS meter. In some embodiments, the chlorophyll meter measures absorbance at 502 nm through the leaf.
- the method may comprise increasing the chlorophyll content of the contacted plant, wherein the chlorophyll content of the contacted plant is increased by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant may be increased from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the chlorophyll content of the contacted plant is increased from about 5% to 50% as compared to an uncontacted plant.
- the method may comprise increasing the salinity tolerance of the contacted plant, wherein the yield of the contacted plant under salinity condition is increased by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the yield of the contacted plant under salinity condition may be increased from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the yield of the contacted plant under salinity condition is increased from about 5% to 50% as compared to an uncontacted plant.
- the yield of the contacted plant is measured by weight.
- the method may comprise increasing the salinity tolerance of the contacted plant, wherein a water consumption per weight unit of the contacted plant produced is reduced by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the water consumption per weight unit of the contacted plant produced is reduced from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the water consumption per weight unit of the contacted plant produced is reduced from about 5% to 50% as compared to an uncontacted plant.
- the water consumption is measured by weight.
- the method may comprise increasing the drought tolerance of the contacted plant, wherein the yield of the contacted plant under drought condition is increased by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the yield of the contacted plant under drought condition may be increased from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the yield of the contacted plant under drought condition is increased from about 5% to 50% as compared to an uncontacted plant.
- the yield of the contacted plant is measured by weight.
- the method may comprise increasing the pest resistance of the contacted plant, wherein the yield of the contacted plant without using any pesticides is increased by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the yield of the contacted plant without using any pesticides may be increased from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the yield of the contacted plant without using any pesticides is increased from about 5% to 50% as compared to an uncontacted plant.
- the yield of the contacted plant is measured by weight.
- the method may comprise reducing the pesticides consumption of the contacted plant, wherein a pesticides consumption per weight unit of the contacted plant produced is reduced by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted plant.
- the pesticides consumption per weight unit of the contacted plant produced is reduced from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted plant.
- the pesticides consumption per weight unit of the contacted plant produced is reduced from about 5% to 50% as compared to an uncontacted plant.
- the pesticides consumption is measured by weight.
- the contacted plant comprises a corn.
- a production of the corn may be increased as compared to an uncontacted plant.
- the method may comprise increasing the production of the corn, wherein an average kernel mass of the corn is increased by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted corn.
- the average kernel mass of the corn may be increased from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted corn.
- the average kernel mass of the corn is increased from about 5% to 50% as compared to an uncontacted corn.
- the method may comprise increasing the production of the corn, wherein an average ear volume of the corn is increased by at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, or 90% as compared to an uncontacted corn.
- the average ear volume of the corn may be increased from about 5% to 90%, for example, 5%-25%, 10%-30%, 20%-40%, 30%-50%, 40%-50%, 50%-60%, 60%-70%, or 70%-90% as compared to an uncontacted corn.
- the average ear volume of the corn is increased from about 5% to 50% as compared to an uncontacted corn.
- the method may inhibit the growth of a weed.
- the weed comprises a parasitic weed.
- the parasitic weed comprises a weed from the genus of Striga .
- the Striga genus may comprise species such as Striga asiatica, S. gesnerioides , and S. hermonthica .
- the growth of the weed is measured in biomass (grams) over time.
- a method of making a formulation comprising forming the formulation with the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer disclosed herein.
- the formulation further comprises an excipient.
- the excipient comprises water, a surfactant, an alcohol, or any combination thereof.
- the amount of the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation disclosed herein may comprise from at least about 1 mg to 1000 kg.
- the amount of the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation disclosed herein may comprise at least about 1 mg, 5 mg, 10 mg, 20 mg, 30 mg, 40 mg, 50 mg, 100 mg, 200 mg, 300 mg, 400 mg, 500 mg, 1 g, 5 g, 10 g, 50 g, 100 g, 500 g, 1 kg, 5 kg, 10 kg, 50 kg, 100 kg, or 1000 kg.
- the amount of the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation disclosed herein may comprise from at least about 1% to 99% of the total weight.
- the amount of the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation disclosed herein may comprise comprises about 1%, 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 99% of the total weight.
- a soil comprising the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation disclosed herein.
- a plant grown using the method disclosed herein, or an edible portion thereof.
- a food comprising an ingredient from the plant disclosed herein, or an edible portion thereof.
- a food comprising the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation disclosed herein.
- a seed comprising the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation disclosed herein.
- Disclosed herein may be an engineered cell comprising a plurality of polynucleotides, wherein (i) the plurality of polynucleotides encode one or more metabolites; and/or (ii) the plurality of polynucleotides comprise one or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- Disclosed herein is a method of producing the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation comprising alkylating
- the method comprises: i) hydroxymethylation of an optionally substituted decahydronaphtho[2,1-b]furan-2(3aH)-one; and ii) subsequent alkylation of
- the hydroxymethylation comprises a reaction between sclareolide and methyl formate in the presence of potassium tert-butoxide and the alkylation comprises a reaction between the hydroxymethylation product and 5-bromo-3-methylfuran-2(5H)-one.
- the optionally substituted decahydronaphtho[2,1-b]furan-2(3aH)-one comprises sclareolide.
- the R 17 is alkyl.
- the R 17 is methyl.
- the X is Cl.
- the hydroxymethylation and alkylation are a one pot procedure.
- the method may comprise (i) hydroxymethylation of an optionally substituted decahydronaphtho[2,1-b]furan-2(3aH)-one; and (ii) subsequent alkylation with
- R 17 is H, alkyl, halo, or haloalkyl and X is Cl, Br, or I.
- hydroxymethylation and alkylation is a one pot procedure.
- the optionally substituted decahydronaphtho[2,1-b]furan-2(3aH)-one is sclareolide.
- R 17 is alkyl. In some instances, X is Br.
- the hydroxymethylation is a reaction between sclareolide and methyl formate in the presence of potassium tert-butoxide and the alkylation is a reaction between the hydroxymethylation product and 5-bromo-3-methylfuran-2(5H)-one.
- strigolactone examples include, but are not limited to strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, and sorgolactone.
- orobanchol examples include, but are not limited to, 7-oxo-orobanchol, 2′epi-orobanchol, ent-2′-epi-orobanchol and ent-orobanchol.
- 5-deoxystrigol examples include, but are not limited to, 2′-epi-5-deoxystrigol, ent-2′-epi-5-deoxystrigol, and ent-5-deoxystrigol.
- the chemical mimics of strigolactone may be based on or derived from a plant.
- the plant may be rice.
- the plant may be tobacco.
- strigolactone analogs include, but are not limited to, 3′-methyl-GR24, thia-3′-methyl-debranone-like molecule, AR36, and CISA-1 (Boyer F D, Mol Plant, 2013 November).
- strigolactone analogs have been disclosed in Cohen (2013, Mol Plant, 2013, (6):1:141-52), Ruyter-Spira (2011 , Plant Physiol, 155(2):721-34), Tanaka M (2013 , Biosci Biotechnol Biochem, 77(4):832-5), Mwakaboko (2011 , Plant Cell Physiol, 52(4):699-715), and Besserer (2008 , Plant Physiol, 148(1); 402-13).
- the plant propagation material may comprise a compound having of Formula I, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula II, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula III, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having a structure of Formula (VI)
- plant propagation materials comprising one or more compounds having the structure of Formula IV, Formula V, or a salt, solvate, polymorph, stereoisomer, or isomer thereof. Further disclosed herein are plant propagation materials comprising two or more compounds having the structure of Formula IV and Formula V.
- strigolactones comprising mixtures of strigolactones, or salts, solvates, polymorphs, stereoisomers, or isomers thereof.
- the mixture of strigolactones may comprise two or more of strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, sorgolactone, 7-oxo-orobanchol, 2′epi-orobanchol, ent-2′-epi-orobanchol, ent-orobanchol, 2′-epi-5-deoxystrigol, ent-2′-epi-5-deoxystrigol, and ent-5-deoxystrigol.
- the mixture of strigolactones may comprise three or more of strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, sorgolactone, 7-oxo-orobanchol, 2′epi-orobanchol, ent-2′-epi-orobanchol, ent-orobanchol, 2′-epi-5-deoxystrigol, ent-2′-epi-5-deoxystrigol, and ent-5-deoxystrigol.
- the mixture of strigolactones may comprise four or more of strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, sorgolactone, 7-oxo-orobanchol, 2′epi-orobanchol, ent-2′-epi-orobanchol, ent-orobanchol, 2′-epi-5-deoxystrigol, ent-2′-epi-5-deoxystrigol, and ent-5-deoxystrigol.
- the mixture of strigolactones may comprise five or more of strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, sorgolactone, 7-oxo-orobanchol, 2′epi-orobanchol, ent-2′-epi-orobanchol, ent-orobanchol, 2′-epi-5-deoxystrigol, ent-2′-epi-5-deoxystrigol, and ent-5-deoxystrigol.
- the mixture of strigolactones may comprise two or more of strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise three or more of strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise four or more of strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise five or more of strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, and sorgolactone.
- the method may comprise chemical synthesis of the plant propapagation material.
- the plant propagation material may be a chemical mimic of strigolactone, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be a chemical mimic of 5-deoxystrigol, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be a chemical mimic of strigol, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be a chemical mimic of orobanchol, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be a chemical mimic of orobanchol acetate, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be a chemical mimic of strigyl acetate, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be a chemical mimic of sorgolactone, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material is a compound having the structure of Formula I or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material is a compound having the structure of Formula II or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material is a compound having the structure of Formula III or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material is a compound having the structure of Formula IV or a salt, solvate, polymorph, stereoisomer, or isomer thereof. In some instances, the plant propagation material is a compound having the structure of Formula V or a salt, solvate, polymorph, stereoisomer, or isomer thereof. In some instances, the plant propagation material is a compound having the structure of Formula VI or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the method of producing a plant propagation material may comprise conducting a hydroxymethylation and/or alkylation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, thereby producing a plant propagation material.
- the sesquiterpene lactone for use in the methods disclosed herein may be sclareolide.
- the sesquiterpene lactone may be extracted from a sage plant.
- the sage plant may be a clary sage plant.
- the condensation reaction may comprise condensing the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, with methyl formate to produce a hydroxymethylene lactone.
- the condensation reaction may further comprise potassium tert-butoxide.
- the method may comprise condensing the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof with an excess of methyl formate.
- the method may comprise condensing the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof with two-fold excess of methyl formate.
- the method may comprise condensing the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof with three-fold excess of methyl formate.
- the method may comprise condensing the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof with four-fold excess of methyl formate.
- the method may comprise condensing the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof with five-fold excess of methyl formate.
- the method may further comprises conducting an alkylation reaction.
- the alkylation reaction may comprise alkylating the condensation reaction product with a bromobutenolide.
- the alkylation reaction may comprise alkylating the hydroxymethylene lactone with a bromobutenolide.
- the alkylation reaction may produce a mixture of two diastereomers.
- the two diasteromers are
- the method of producing the plant propagation material does not require a catalyst. In some instances, the method of producing the plant propagation material does not require two or more reaction volumes. In some instances, the method of producing the plant propagation material not require a chromatographic purification.
- the efficiency of producing the plant propagation material is at least about 50%.
- the efficiency of producing the plant propagation material may be at least about 60%.
- the efficiency of producing the plant propagation material may be at least about 70%.
- the efficiency of producing the plant propagation material may be at least about 75%.
- the efficiency of producing the plant propagation material may be at least about 80%.
- the efficiency of producing the plant propagation material may be at least about 85%.
- the efficiency of producing the plant propagation material may be at least about 90%.
- the plant propagation material may be used to inhibit one or more weeds.
- the one or more weeds may be a parasitic weed.
- the parasitic weed may be Striga .
- the parasitic weed may be Orobanche.
- the biosynthetic process may comprise introducing one or more genes into a cell.
- the biosynthetic process may comprise transfecting one or more genes into a cell.
- the biosynthetic process may comprise transforming one or more cells with one or more genes.
- the one or more genes may encode a component of a strigolactone pathway.
- the one or more genes may encode a metabolite. In some instances, the one or more genes are not natural to the cell.
- a method of producing a plant propagation material comprising expressing a plurality of polynucleotides in a cell to produce a plant propagation material, wherein (i) the plurality of polynucleotides encode one or more metabolites; and/or (ii) the plurality of polynucleotides may comprise one or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- polynucleotides encoding the one or more genes for use in the production of a plant propagation material.
- vectors comprising the one or more polynucleotides encoding the one or more genes for use in the production of a plant propagation material.
- cells for use in producing the plant propagation material disclosed herein The cell may be an engineered cell.
- the engineered cell may comprise a plurality of polynucleotides, wherein (i) the plurality of polynucleotides encode one or more metabolites; and/or (ii) the plurality of polynucleotides comprise one or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- the plurality of polynucleotides may encode one or more metabolities and comprise one or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- the plurality of polynucleotides may encode one or more metabolites.
- the one or more metabolites may comprise lycopene. In some instances, the one or more metabolites are not natural to the cell.
- the plurality of polynucleotides may comprise one or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- the plurality of polynucleotides may comprise two or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- the plurality of polynucleotides may comprise three or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- the plurality of polynucleotides may comprise four or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- the plurality of polynucleotides may comprise five or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- the plurality of polynucleotides may comprise six or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- the plurality of polynucleotides comprise seven or more genes selected from a group comprising crtE, crtB, crtI, D27, CCD7, CCD8, and MAX1.
- the plurality of polynucleotides may comprise a crtE gene.
- the plurality of polynucleotides may comprise a crtB gene.
- the plurality of polynucleotides may comprise a crtI gene.
- the plurality of polynucleotides may comprise a D27 gene.
- the plurality of polynucleotides may comprise a CCD7 gene.
- the plurality of polynucleotides may comprise a CCD8 gene.
- the plurality of polynucleotides may comprise a MAX1 gene.
- the one or more genes may be based on or derived from a plant.
- the plant may be a tobacco plant.
- the plant may be a rice plant.
- the one or more genes may be based on or derived from a fungi.
- the one or more genes may be based on or derived from yeast.
- the yeast may be a Pantoea .
- the yeast may be P. ananatis.
- the cell may be a prokaryotic cell.
- the cell may be a eukaryotic cell.
- the eukaryotic cell may be a yeast cell.
- the yeast cell may be a Pichia cell.
- the Pichia cell may be a Pichia pastoris cell.
- the Pichia cell may be a Pichia anantais cell.
- the yeast cell may be a Saccharomyces cell.
- the Saccharomyces cell may be a Saccharaomyces cerevesiae.
- the one or more cells may be cultured.
- the cells may be cultured under conditions to express the one or more genes that were introduced into the cell.
- the cells may be cultured to express the plurality of polynucleotides.
- Purifying the plant propagation material may comprise extracting the plant propagation material from the cell.
- Purifying the plant propagation material may comprise an ethyl acetate purification.
- formulations comprising the compounds disclosed herein.
- formulations comprising the plant propagation materials disclosed herein.
- the formulation may be formulated as a powder, seed coating, or granule.
- the powder may be a wettable powder.
- the formulation may be formulated as a spray.
- the formulation may be formulated as an irrigation supplement.
- the formulation may be formulated as a seed coating.
- the method may comprise administering a formulation comprising a plant propagation material disclosed herein to a plant, thereby improving agriculture.
- the plant propagation material may comprise strigolactone, wherein the strigolactone is obtained by a biosynthetic process.
- the plant propagation material may comprise a chemical mimic of strigolactone, wherein the chemical mimic of strigolactone is obtained by a chemical process.
- the plant propagation material may comprise a compound having the structure of Formula I or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula II or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula III or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula IV or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula V or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula VI or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be applied directly to the plant.
- the plant propagation may be applied indirectly to the plant.
- the plant propagation material may be applied to the plant's habitat.
- the plant propagation material may be applied to the soil.
- the method may comprise causing a formulation comprising a plant propagation material disclosed herein to act on the phytopathogenic fungi.
- the plant propagation material may comprise strigolactone, wherein the strigolactone is obtained by a biosynthetic process.
- the plant propagation material may comprise a chemical mimic of strigolactone, wherein the chemical mimic of strigolactone is obtained by a chemical process.
- the plant propagation material may comprise a compound having the structure of Formula I or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula II or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula III or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula IV or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula V or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula VI or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be applied directly to the phytopathogenic fungi.
- the plant propagation may be applied indirectly to the phytopathogenic fungi.
- the plant propagation material may be applied to the phytopathogenic fungi's habitat.
- the plant propagation material may be applied to the soil.
- the plant propagation material may be applied to a plant within the vicinity of the phytopathogenic fungi.
- the plant within the vicinity of the phytopathogenic fungi may be a plant targeted by the phytopathogenic fungi.
- the plant propagation material may be applied directly to the plant.
- the plant propagation may be applied indirectly to the plant.
- the plant propagation material may be applied to the plant's habitat.
- the method may comprise causing a formulation comprising a plant propagation material disclosed herein to act on the unwanted plant.
- the unwanted plant may be a Striga plant or Orobanche plant.
- the plant propagation material may comprise strigolactone, wherein the strigolactone is obtained by a biosynthetic process.
- the plant propagation material may comprise a chemical mimic of strigolacatone, wherein the chemical mimic of strigolactone is obtained by a chemical process.
- the plant propagation material may comprise a compound having the structure of Formula I or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula II or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula III or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula IV or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula V or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula VI or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be applied directly to the unwanted plant.
- the plant propagation may be applied indirectly to the unwanted plant.
- the plant propagation material may be applied to the unwanted plant's habitat.
- the plant propagation may be applied to the soil.
- the plant propagation material may be applied to another plant within the vicinity of the unwanted plant.
- the plant within the vicinity of the unwanted plant may be a desirable plant.
- the plant propagation material may be applied directly to the desirable plant.
- the plant propagation may be applied indirectly to the desirable plant.
- the plant propagation material may be applied to the desirable plant's habitat.
- the method may comprise causing a formulation comprising a plant propagation material disclosed herein to act on the unwanted insect or mite.
- the plant propagation material may comprise strigolactone, wherein the strigolactone is obtained by a biosynthetic process.
- the plant propagation material may comprise a chemical mimic of strigolacatone, wherein the chemical mimic of strigolactone is obtained by a chemical process.
- the plant propagation material may comprise a compound having the structure of Formula I or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula II or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula III or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula IV or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula V or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula VI or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be applied to directly to the mite or insect.
- the plant propagation material may be applied indirectly to the mite or insect.
- the plant propagation material may be ingested by the mite or insect.
- the plant propagation material may be applied to a plant targeted by the mite or insect.
- the plant propagation material may be applied directly to the plant.
- the plant propagation may be applied indirectly to the plant.
- the plant propagation material may be applied to the plant's habitat.
- the plant propagation material may be applied to the soil.
- the method may comprise causing a formulation comprising a plant propagation material disclosed herein to act on the plant or its habitat.
- the plant propagation material may comprise strigolactone, wherein the strigolactone is obtained by a biosynthetic process.
- the plant propagation material may comprise a chemical mimic of strigolactone, wherein the chemical mimic of strigolactone is obtained by a chemical process.
- the plant propagation material may comprise a compound having the structure of Formula I or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula II or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula III or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula IV or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula V or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound having the structure of Formula VI or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may be applied directly to the plant.
- the plant propagation may be applied indirectly to the plant.
- the plant propagation material may be applied to the plant's habitat.
- the plant propagation material may be applied to the soil.
- the plant, desirable plant or plant targeted by the phytopathogenic fungi or mite/insect may be a crop plant.
- Crop plants include, but are not limited to, corn, rice, sorghum, millets, and sugar cane.
- the plant, desirable plant or plant targeted by the phytopathogenic fungi or mite/insect may be tobacco.
- the method may comprise contacting the plant with a plant propagation material disclosed herein.
- the plant propagation material may comprise a compound of Formula (I) or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula (II) or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula (III) or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a strigolactone or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a strigolactone mimic or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material for use in preserving or extending the life of a plant may be produced by any of the methods disclosed herein.
- the plant propagation material is produced by conducting a condensation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the plant propagation material may be produced by conducting a hydroxymethylation on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the plant propagation material may be produced by (a) conducting a hydroxymethylation on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof to produce a first product; and (b) conducting an alkylation reaction on the first product.
- the plant may be a cut plant.
- the plant may be an uncut plant.
- the plant may be a potted plant.
- the plant may be a flower.
- the plant may be a bush or shrub.
- the plant may be a tree.
- Preserving or extending the life of a plant may comprise contacting the plant with a plant propagation material disclosed herein. Contacting the plant with the plant propagation material may comprise administering the plant propagation material as a spray. Contacting the plant with the plant propagation material may comprise adding the plant growth material to the irrigation water of the plant. Contacting the plant with the plant propagation material may comprise applying the plant propagation material to the habitat of the plant. Contacting the plant with the plant propagation material may comprise adding the plant propagation material to a plant container (e.g., vase) and placing the plant in the plant container. Contacting the plant with the plant propagation material may comprise adding the plant propagation material to soil.
- a plant container e.g., vase
- the life of the plant may be extended by at least about 20% as compared to an untreated plant.
- the life of the plant may be extended by at least about 30% as compared to an untreated plant.
- the life of the plant may be extended by at least about 40% as compared to an untreated plant.
- the life of the plant may be extended by at least about 50% as compared to an untreated plant.
- the life of the plant may be extended by at least about 55% as compared to an untreated plant.
- the life of the plant may be extended by at least about 60% as compared to an untreated plant.
- the life of the plant may be extended by at least about 65% as compared to an untreated plant.
- the life of the plant may be extended by at least about 70% as compared to an untreated plant.
- the life of the plant may be extended by at least about 6, 12, 24, 30, 36, 42, 48, 54, 60, 66, or 72 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 24 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 36 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 48 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 72 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, or 7 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 1 day as compared to an untreated plant.
- the life of the plant may be extended by at least about 2 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 2.5 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 3 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, or 7 weeks as compared to an untreated plant.
- the life of the plant may be extended by at least about 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 weeks as compared to an untreated plant.
- the life of the plant may be extended by at least about 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, or 7 months as compared to an untreated plant.
- the life of the plant may be extended by at least about 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 months as compared to an untreated plant.
- Preserving or extending the life of the plant may comprise reducing wilting of the plant.
- Reducing wilting of the plant may comprise reducing flower or leaf rolling of the plant.
- the wilting of the plant may be reduced by at least about 20% as compared to an untreated plant.
- the wilting of the plant may be reduced by at least about 40% as compared to an untreated plant.
- the wilting of the plant may be reduced by at least about 60% as compared to an untreated plant.
- Reducing the wilting of the plant may comprise delaying the wilting of the plant as compared to an untreated plant.
- the wilting of the plant may be delayed by at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours as compared to an untreated plant.
- the wilting of the plant may be delayed by at least about 12 hours as compared to an untreated plant.
- the wilting of the plant may be delayed by at least about 24 hours as compared to an untreated plant.
- the wilting of the plant may be delayed by at least about 36 hours as compared to an untreated plant.
- the wilting of the plant may be delayed by at least about 48 hours as compared to an untreated plant.
- Preserving or extending the life of the plant may comprise prolonging or maintaining turgidity of the plant.
- the turgidity of the plant may be greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 20% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 30% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 40% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 50% greater than the turgidity of an untreated plant.
- Preserving or extending the life of the plant may comprise prolonging the turgid state of the plant.
- the turgid state of the plant may be increased by at least about 20% as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 30% as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 40% as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 50% as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 6 hours as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 12 hours as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 24 hours as compared to an untreated plant.
- Preserving or extending the life of the plant may comprise reducing or delaying the loss of one or more petals of the plant.
- the loss of the one or more petals of the plant may be reduced by least about 20% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be reduced by least about 30% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be reduced by least about 40% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 6 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 12 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 18 hours as compared to the loss of one or more petals of an untreated plant.
- Preserving or extending the life of the plant may comprise maintaining the chlorophyll content of the plant.
- the chlorophyll content of the plant may be maintained for at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours.
- the chlorophyll content of the plant may be maintained for at least about 6 hours.
- the chlorophyll content of the plant may be maintained for at least about 12 hours.
- the chlorophyll content of the plant may be maintained for at least about 24 hours.
- Preserving or extending the life of the plant may comprise reducing or delaying the loss of the chlorophyll content of the plant.
- the chlorophyll content of the plant may be greater than the chlorophyll content of an untreated plant.
- the chlorophyll content of the plant may be at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, or 50% greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 20% greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, 7, 8, 9, or 10-fold greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 2-fold greater than the content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 6 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 12 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, or 60% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 10% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 20% less than the loss of the chlorophyll content of an untreated plant.
- the stereocenter * is selected from the group consisting of: (S), (R), racemic, and a non-racemic mixture of (R) and (S).
- any stereocenter can be selected from the group consisting of: (S), (R), racemic, and a non-racemic mixture of (R) and (S).
- FIG. 1 (A) Synthetic SL shows potent bioactivity, with nanograms of material inducing germination of Striga seeds. (B) Bioactivity of synthetic SL. Seeds of Striga asiatica were exposed to SL (right) or a mock treatment (left). SL induces Striga germination as evident by radicle emergence from seeds. (C) Synthetic biological pathway for the production of SL in yeast, consisting of enzymes of plant and microbial origin.
- FIG. 2 Effect of SL on tolerance to water stress in Capsicum annum. Plants were either treated with SL or mock treated. Irrigation was stopped for 4 weeks. Untreated plant displayed symptoms of severe water stress and chlorosis, while the SL-treated plant appeared healthy and unstressed.
- FIG. 3 shows the effect of a plant propagation material on vase life extension of cut flowers.
- FIG. 4 Timeline of experiments to determine effect of SL application on yield. Plants are water-stressed starting in the early reproductive stage, prior to tasseling. SL is applied as either a ‘preventative’ dose during vegetative growth or as a ‘protective’ dose at the onset of water stress. Irrigation resumes during grain fill and grain yield is determined at maturity.
- FIG. 5 shows a schematic for the synthesis of AB01.
- FIG. 6 shows bioactivity of AB01 in Arabidopsis .
- A Max1+mock treated;
- B Max1+AB01.
- FIG. 7 shows an exemplary synthetic approach for (+)-sclareolide.
- FIG. 8 shows exemplary synthetic approaches for nitrogen-containing compounds.
- FIG. 9 shows the synthesis of formyl sclareolide.
- FIG. 10 shows the synthesis of chlorobutenolide.
- FIG. 11 shows the Synthesis of AB01.
- FIG. 12 shows improved germination triggering of parasitic weeds.
- FIG. 13 shows drought tolerance in alfalfa enabled by AB01 treatment.
- FIG. 14 shows AB01 enhances ear fertilization in corn field trial.
- FIG. 15 shows AB01 enhances kernel set in corn field trial.
- FIG. 16 shows AB01 enhances ear volume.
- FIG. 17 AB01 treatment enhances average kernel weight.
- FIG. 18 shows AB01 treatment enhances harvest yield.
- FIG. 19 shows AB01 treatment enables salinity tolerance in alfalfa.
- FIG. 20 shows AB01 treatment enables salinity tolerance in tomato.
- FIG. 21 shows AB01 treatment enhances drought tolerance in wheat.
- FIG. 22 shows reduction of Striga and enhancement of grain yield in AB01 treated fields (Siaya County, Kenya).
- FIG. 23 shows reduction of acute water stress in AB01 treated corn.
- formulations herein can be powdery.
- genes listed herein may be heterologous genes.
- powder formulations herein can contain water in an amount from about 0% to about 15% w/w, for example 0-10%, 0-5%, or 0-1% w/w; or about: 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 11%, 12%, 13%, 14%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90% or 99% w/w, based on the weight of the formulation.
- plant propagation material can refer to any compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, isomer, or formulation described herein.
- stereocenter in a structure disclosed or illustrated herein, the stereocenter can be R or S in each case.
- the wavy bond can be
- amino can refer be monosubstituted, disubstituted or trisubstituted.
- alkyl may comprise lower alkyl.
- the alkyl can be from C1 to C8, for example, C1, C2, C3, C4, C5, C6, or C8.
- the alkyl can be linear or branched.
- the alkyl may exclude cycloalkyl.
- treated can be referred to “contacted.”
- untreated can be referred to “uncontacted.”
- Drought is one of the most significant risks for farmers, rural economies, and the food supply chain; limited precipitation and irrigation acts as a major constraint on crop productivity. climate change threatens to further exacerbate crop losses due to drought by shifting rainfall patterns and weather conditions across e.g. America's most productive agricultural regions. There is a need for tools and strategies to enable e.g. American farmers to adapt to climate change by reducing the water footprint of major commercial crops. While there have been promising advances in field management (such as low-till and no-till systems) and in the development of drought tolerant crops, there is currently no crop protection product that enhances the robustness of field crops to periods of prolonged drought and water-limitation stress.
- a product that could be applied to crops as a foliar spray or irrigation supplement to boost the health and productivity of plants during drought stress would allow farmers to reduce the risk of crop loss and maintain productivity throughout variable weather. Due to the large magnitude of drought-related crop losses and the substantial threats that climate change poses for the agricultural system, there is a significant opportunity for development of crop protection products that enhance drought tolerance.
- Plant hormones also known as plant growth regulators, PGRs
- PGRs plant growth regulators
- Processes such as growth, development, and morphology are regulated by a stress-responsive hormone signaling network that includes the signaling molecules abscisic acid, cytokines, auxins, brassinosteroids, and strigolactones [11].
- the hormone-signaling network also orchestrates stress adaptive responses such as stomata opening, nutrient allocation, induction of innate immunity, and source-sink distribution [12].
- the understanding of the interactions between environmental stress, PGRs, and plant physiology are important targets for biotechnological improvement of crops. For example, tobacco plants genetically modified to overproduce cytokines maintained their photosynthetic capacity under water limitation by inhibiting degradation of the photosynthetic machinery [13].
- the PGR abscisic acid plays a major role in adaptation to drought conditions. Upon water limitation, abscisic acid biosynthesis is induced, with concentrations in cells reaching micromolar levels [14]. Abscisic acid triggers fast responses, such as stomata closure and cell cycle arrest, as well as slower responses such as transcriptional and epigenetic regulation of plant metabolism [15]. This coordinated response to water stress enables plants to adapt to periods of water limitation.
- Strigolactones are a recently discovered class of PGRs involved in the regulation of root and shoot morphology and interactions with rhizosphere-associated symbionts. SLs were first characterized as germination stimulants for seeds of the parasitic plants Striga and Orobanche [16]. SLs are derived from carotenoid biosynthesis, sharing a common precursor (beta-carotene) with abscisic acid [17]. The common pathway of abscisic acid and SL biosynthesis has lead to investigations into potential correlations in the levels of each PGR and their co-regulation of critical plant functions.
- SL levels were correlated with abscisic acid levels by an unknown mechanism [18]. Due to this correlation, we hypothesized that SLs may play a role in the regulation of stress response, either by influencing the levels and dynamics of abscisic acid signaling or by unknown mechanisms. SLs are among the few PGRs that have not been evaluated as crop protection products or as components of integrated crop management strategies.
- Crop failure, reduced crop harvest yields, and loss of pasture are the primary agricultural results of drought.
- the outcomes of drought affects individual farm revenues as well as the regional and national economies.
- the anticipated endpoint of the research and development project proposed here would deliver a product to protect crops from the effects of drought, increasing yields and reducing the risk of crop loss and failure.
- the development of this drought-protective product would have several benefits to society at large, including enhanced food security and food price stability.
- the secondary effects of drought and crop loss are increased food prices for consumers, which could be partially mitigated by deployment of a drought-protective product. It is also feasible that deployment of a drought-protective product will lower the water demands of agriculture, relieving existing and future stress on aquifers and fresh water supplies.
- the compounds may have the structure of Formula I-VI, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the compounds may comprise chemical mimics of strigolactone. Further disclosed are methods of producing the compounds and uses thereof.
- the compounds may have the structure of Formula I-VI, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the compounds may comprise chemical mimics of strigolactone. Further disclosed are methods of producing the compounds and uses thereof.
- strigolactone, or a salt, solvate, polymorph, stereoisomer, or isomer thereof may be obtained by chemical synthesis. Chemical synthesis methods are disclosed herein.
- the strigolactone, or a salt, solvate, polymorph, stereoisomer, or isomer thereof may be obtained by a biosynthetic process. Biosynthetic processes are disclosed herein.
- polynucleotides comprising one or more genes for use in a biosynthetic process.
- the one or more genes may encode one or more components of a strigolactone pathway.
- vectors comprising the polynucleotides.
- cells comprising the vectors comprising the polynucleotides.
- the methods may comprise a chemical synthesis.
- the method comprises a biosynthetic process.
- the plant propagation materials may be used to control the parasitic weeds of the Striga genus.
- the Striga genus may comprise species such as Striga asiatica, S. gesnerioides , and S. hermonthica .
- the plant propagation materials may be used to improve agriculture.
- the plant propagation material may be used to improve crop yield.
- the plant propagation material may be used to improve crop yield of staples such as maize, sorghum, rice, and cowpea.
- the plant propagation material may be use to preserve or extend the life of a plant.
- the plant propagation material may be used to prevent or reduce wilting of a plant.
- the plant propagation material may be used to delay the wilting of a plant.
- the plant propagation material may be used to maintain turgidity of a plant.
- the plant propagation material may be used to prolong the turgid state of a plant.
- the plant propagation material may be used to prevent or delay the loss of plant leaves or petals.
- the plant propagation material may be used to maintain the chlorophyll content of the plant.
- the plant propagation material may be used to reduce or delay the loss of the chlorophyll content of the plant.
- a chemical mimic of strigolactone may be a compound of Formula (I), a salt, solvate, polymorph, stereoisomer, or isomer thereof:
- R 1 , R 2 , R 4 , R 5 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , and R 17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ;
- R 3 and R 6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ; or R 3 and R 6 together form a direct bond to provide a double bond;
- each R 18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, or —C(O)R 19 ;
- each R 19 is independently alkyl, haloalkyl, aryl, or heteroaryl;
- n 0, 1, or 2;
- n 1 or 2.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein m is 0 and n is 1.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein m is 0 and n is 2.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein m is 1 and n is 2.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein m is 2 and n is 1.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein m is 2 and n is 2.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein m is 1 or 2.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein m is 1 or 2.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein R 3 and R 6 together form a direct bond to provide a double bond.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein R 3 and R 6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 .
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein R 3 and R 6 are each independently H or alkyl.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein R 1 , R 2 , R 4 , R 5 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , and R 17 are each independently H, alkyl, or —OR 18 ; R 3 and R 6 are each independently H or alkyl.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein R 1 , R 2 , R 4 , R 5 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , and R 16 are each independently H, alkyl, or —OR 18 ; R 3 and R 6 are each independently H or alkyl; and R 17 is alkyl.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein m is 1, n is 1, and the compound has the structure of Formula (II):
- the chemical mimic of strigolactone may be a compound of Formula (II) wherein R 1 , R 2 , R 4 , R 5 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , and R 17 are each independently H, alkyl, or —OR 18 .
- the chemical mimic of strigolactone may be a compound of Formula (II) wherein R 1 , R 2 , R 4 , R 5 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , and R 17 are each independently H, alkyl, or —OR 18 and R 18 is H or alkyl.
- the chemical mimic of strigolactone may be a compound of Formula (II) wherein R 3 and R 6 together form a direct bond to provide a double bond.
- the chemical mimic of strigolactone may be a compound of Formula (II) wherein R 3 and R 6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 .
- the chemical mimic of strigolactone may be a compound of Formula (II) wherein R 3 and R 6 are each independently H or alkyl.
- the chemical mimic of strigolactone may be a compound of Formula (II) wherein R 17 is alkyl.
- the chemical mimic of strigolactone may be a compound of Formula (I) wherein m is 1; n is 1; each of R 4 , R 5 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , and R 15 are hydrogen; and the compound has the structure of Formula (III):
- the chemical mimic of strigolactone may be a compound of Formula (III) wherein R 3 and R 6 together form a direct bond to provide a double bond.
- the chemical mimic of strigolactone may be a compound of Formula (III) wherein R 3 and R 6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 .
- the chemical mimic of strigolactone may be a compound of Formula (III) wherein R 3 and R 6 are each independently H or alkyl.
- the chemical mimic of strigolactone may be a compound of Formula (III) wherein R 17 is alkyl.
- the chemical mimic of Strigolactone can be compound of Formula (VII), a salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer thereof:
- a, b, c are each independently 0, 1, or 2;
- each A is independently O, or S;
- each E is independently O, S, or —NR 18 ;
- each G is independently C or N;
- R 5 , R 6 , R 11 , R 12 , R 14 , R 15 and R 17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ;
- R 2 , R 3 , R 7 , R 8 , R 9 , and R 10 are each independently H, alkyl, haloalkyl, amino, halo, —OR 18 or a lone electron pair;
- R 1 and R 16 are each independently H, alkyl, haloalkyl, amino, halo, lone electron pair, or —OR 18 ; or R 1 and R 16 together form a direct bond to provide a double bond;
- R 4 and R 13 are each independently H, alkyl, haloalkyl, amino, halo, lone electron pair, or —OR 18 ; or R 1 and R 16 together form a direct bond to provide a double bond;
- each R 18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, —C(O)R 19 or
- each R 19 is independently H, alkyl, haloalkyl, aryl, or heteroaryl.
- Another chemical mimic of Strigolactone can be a compound of Formula (VIII), a salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer thereof:
- a, b, c are each independently 0, 1, or 2;
- each A is independently O, or S;
- each E is independently O, S, or —NR 18 ;
- each G is independently C or N;
- R 5 , R 6 , R 11 , R 12 , R 14 , R 15 and R 17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ;
- R 2 , R 3 , R 7 , R 8 , R 9 , and R 10 are each independently H, alkyl, haloalkyl, amino, halo, —OR 18 or a lone electron pair;
- R 1 and R 16 are each independently H, alkyl, haloalkyl, amino, halo, lone electron pair, or —OR 18 ; or R 1 and R 16 together form a direct bond to provide a double bond;
- R 4 and R 13 are each independently H, alkyl, haloalkyl, amino, halo, lone electron pair, or —OR 18 ; or R 1 and R 16 together form a direct bond to provide a double bond;
- each R 18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, —C(O)R 19 or
- each R 19 is independently H, alkyl, haloalkyl, aryl, or heteroaryl.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 0, b is 0, and c is 0.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 0, b is 0, and c is 1.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 0, b is 0, and c is 2.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 0, b is 1, and c is 0.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 0, b is 1, and c is 1.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 0, b is 1, and c is 2.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 0, b is 2, and c is 0.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 0, b is 2, and c is 1.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 0, b is 2, and c is 2.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 1, b is 0, and c is 0.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 1, b is 0, and c is 1.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 1, b is 0, and c is 2.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 1, b is 1, and c is 0.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 1, b is 1, and c is 1.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 1, b is 1, and c is 2.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 1, b is 2, and c is 0.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 1, b is 2, and c is 1.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 1, b is 2, and c is 2.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 2, b is 0, and c is 0.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 2, b is 0, and c is 1.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 2, b is 0, and c is 2.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 2, b is 1, and c is 0.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 2, b is 1, and c is 1.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 2, b is 1, and c is 2.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 2, b is 2, and c is 0.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 2, b is 2, and c is 1.
- the chemical mimic of strigolactone may be a compound of Formula (VII) or (VIII) wherein a is 2, b is 2, and c is 2.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer disclosed herein is not (+)-Strigol
- each A in the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone is independently O.
- each E in the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone is independently O.
- each G in the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone is independently C.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone wherein R 2 , R 3 , R 4 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , and R 16 are each independently H.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone wherein R 1 , R 5 , R 6 , R 13 , and R 17 are each independently alkyl.
- the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer of the chemical mimic of strigolactone wherein R 1 , R 5 , R 6 , R 13 , and R 17 are each independently methyl.
- the chemical mimic of strigolactone may be a compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer, having the structure of Formula (IX):
- the chemical mimic of strigolactone may be a compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer, having the structure of Formula (X):
- the chemical mimic of strigolactone may be a compound having the structure of
- a chemical mimic of strigolactone may be a compound, a salt, solvate, polymorph, stereoisomer, or isomer thereof, obtained by a process comprising a condensation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the process may comprise a condensation reaction on a sesquiterpene lactone.
- the condensation reaction may comprise condensing the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, with methyl formate to produce a hydroxymethylene lactone.
- the condensation reaction may comprise condesing the sesquiterpene lactone with methyl formate to produce a hydroxymethylene lactone.
- the condensation reaction may further comprise a base.
- the base may be potassium tert-butoxide.
- the base may be sodium tert-butoxide.
- the process may further comprise conducting an alkylation reaction.
- the alkylation reaction may comprise reaction of the condensation reaction product with an electrophilic butenolide.
- the alkylation reaction may comprise reaction of the condensation reaction product with a halobutenolide.
- the halobutenolide may be chlorobutenolide.
- the halobutenolide may be bromobutenolide.
- the halobutenolide may be iodobutenolide.
- the sesquiterpene lactone may be sclareolide.
- the bromobutenolide may be 5-bromo-3-methylfuran-2(5H)-one.
- a chemical mimic of strigolactone may be a compound having the structure of
- a salt, solvate, polymorph, stereoisomer, or isomer thereof obtained by a process comprising a condensation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the chemical mimic of strigolactone may be obtained by a process comprising a condensation reaction on a sesquiterpene lactone.
- the sesquiterpene lactone may be sclareolide.
- a plant propagation material comprising strigolactone, wherein the plant propagation material is produced by a recombinant cell; and wherein the recombinant cell comprises one or more foreign genes.
- the one or more foreign genes are genes that do not naturally occur in the cell.
- the one or more foreign genes may comprise one or more genes that encode a strigolactone pathway.
- the plant propagation material may comprise a mixture of strigolactones.
- strigolactone examples include, but are not limited to strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, and sorgolactone.
- orobanchol examples include, but are not limited to, 7-oxo-orobanchol, 2′epi-orobanchol, ent-2′-epi-orobanchol and ent-orobanchol.
- Examples of 5-deoxystrigol include, but are not limited to, 2′-epi-5-deoxystrigol, ent-2′-epi-5-deoxystrigol, and ent-5-deoxystrigol.
- the strigolactone may comprise strigol.
- the strigolactone may comprise strigyl acetate.
- the strigolactone may comprise orobanchol.
- the strigolactone may comprise orobanchyl acetate.
- the strigolactone may comprise 5-deoxystrigol.
- the strigolactone may comprise sorgolactone.
- strigolactone examples include, but are not limited to strigol, strigyl acetate, orobanchol, orobanchyl acetate, 7-orobanchyl acetate, 7-hydroxy-orobanchyl acetate, sorgomol, fabacyl acetate, 5-deoxystrigol, and sorgolactone.
- orobanchol examples include, but are not limited to, 7-oxo-orobanchol, 2′epi-orobanchol, ent-2′-epi-orobanchol and ent-orobanchol.
- Examples of 5-deoxystrigol include, but are not limited to, 2′-epi-5-deoxystrigol, ent-2′-epi-5-deoxystrigol, and ent-5-deoxystrigol.
- the mixture of strigolactones may comprise one or more strigolactones selected from strigol, strigyl acetate, orobanchol, orobanchyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise two or more strigolactones selected from strigol, strigyl acetate, orobanchol, orobanchyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise three or more strigolactones selected from strigol, strigyl acetate, orobanchol, orobanchyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise four or more strigolactones selected from strigol, strigyl acetate, orobanchol, orobanchyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise five or more strigolactones selected from strigol, strigyl acetate, orobanchol, orobanchyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise six or more strigolactones selected from strigol, strigyl acetate, orobanchol, orobanchyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise at least about 1% of strigol.
- the mixture of strigolactones may comprise at least about 2% of strigol.
- the mixture of strigolactones may comprise at least about 5% of strigol.
- the mixture of strigolactones may comprise at least about 7% of strigol.
- the mixture of strigolactones may comprise at least about 10% of strigol.
- the mixture of strigolactones may comprise at least about 20% of strigol.
- the mixture of strigolactones may comprise at least about 30% of strigol.
- the mixture of strigolactones may comprise at least about 40% of strigol.
- the mixture of strigolactones may comprise at least about 50% of strigol.
- the mixture of strigolactones may comprise at least about 60% of strigol.
- the mixture of strigolactones may comprise at least about 70% of strigol.
- the mixture of strigolactones may comprise at least about 80% of strigol.
- the mixture of strigolactones may comprise at least about 85% of strigol.
- the mixture of strigolactones may comprise at least about 90% of strigol.
- the mixture of strigolactones may comprise at least about 95% of strigol.
- the mixture of strigolactones may comprise less than about 95% of strigol.
- the mixture of strigolactones may comprise less than about 90% of strigol.
- the mixture of strigolactones may comprise less than about 85% of strigol.
- the mixture of strigolactones may comprise less than about 80% of strigol.
- the mixture of strigolactones may comprise less than about 75% of strigol.
- the mixture of strigolactones may comprise less than about 70% of strigol.
- the mixture of strigolactones may comprise less than about 60% of strigol.
- the mixture of strigolactones may comprise less than about 55% of strigol.
- the mixture of strigolactones may comprise less than about 50% of strigol.
- the mixture of strigolactones may comprise less than about 40% of strigol.
- the mixture of strigolactones may comprise less than about 30% of strigol.
- the mixture of strigolactones may comprise less than about 25% of strigol.
- the mixture of strigolactones may comprise less than about 20% of strigol.
- the mixture of strigolactones may comprise less than about 15% of strigol.
- the mixture of strigolactones may comprise less than about 10% of strigol.
- the mixture of strigolactones may comprise less than about 5% of strigol.
- the mixture of strigolactones may comprise less than about 3% of strigol.
- the mixture of strigolactones may comprise between about 1% and 90% of strigol.
- the mixture of strigolactones may comprise between about 1% and 80% of strigol.
- the mixture of strigolactones may comprise between about 1% and 70% of strigol.
- the mixture of strigolactones may comprise between about 1% and 60% of strigol.
- the mixture of strigolactones may comprise between about 1% and 50% of strigol.
- the mixture of strigolactones may comprise between about 1% and 40% of strigol.
- the mixture of strigolactones may comprise between about 1% and 30% of strigol.
- the mixture of strigolactones may comprise between about 1% and 20% of strigol.
- the mixture of strigolactones may comprise between about 1% and 10% of strigol.
- the mixture of strigolactones may comprise between about 1% and 5% of strigol.
- the mixture of strigolactones may comprise between about 5% and 90% of strigol.
- the mixture of strigolactones may comprise between about 10% and 90% of strigol.
- the mixture of strigolactones may comprise between about 20% and 90% of strigol.
- the mixture of strigolactones may comprise between about 30% and 90% of strigol.
- the mixture of strigolactones may comprise between about 40% and 90% of strigol.
- the mixture of strigolactones may comprise between about 50% and 90% of strigol.
- the mixture of strigolactones may comprise between about 60% and 90% of strigol.
- the mixture of strigolactones may comprise between about 70% and 90% of strigol.
- the mixture of strigolactones may comprise between about 80% and 90% of strigol.
- the mixture of strigolactones may comprise between about 10% and 80% of strigol.
- the mixture of strigolactones may comprise between about 20% and 70% of strigol.
- the mixture of strigolactones may comprise between about 30% and 60% of strigol.
- the mixture of strigolactones may comprise between about 20% and 50% of strigol.
- the mixture of strigolactones may comprise between about 25% and 50% of strigol.
- the mixture of strigolactones may comprise at least about 1% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 2% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 5% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 7% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 10% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 20% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 30% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 40% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 50% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 60% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 70% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 80% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 85% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 90% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 95% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 95% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 90% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 85% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 80% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 75% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 70% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 60% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 55% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 50% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 40% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 30% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 25% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 20% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 15% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 10% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 5% of strigyl acetate.
- the mixture of strigolactones may comprise less than about 3% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 80% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 70% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 60% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 50% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 40% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 30% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 20% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 10% of strigyl acetate.
- the mixture of strigolactones may comprise between about 1% and 5% of strigyl acetate.
- the mixture of strigolactones may comprise between about 5% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 10% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 20% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 30% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 40% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 50% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 60% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 70% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 80% and 90% of strigyl acetate.
- the mixture of strigolactones may comprise between about 10% and 80% of strigyl acetate.
- the mixture of strigolactones may comprise between about 20% and 70% of strigyl acetate.
- the mixture of strigolactones may comprise between about 30% and 60% of strigyl acetate.
- the mixture of strigolactones may comprise between about 20% and 50% of strigyl acetate.
- the mixture of strigolactones may comprise between about 25% and 50% of strigyl acetate.
- the mixture of strigolactones may comprise at least about 1% orobanchol.
- the mixture of strigolactones may comprise at least about 2% orobanchol.
- the mixture of strigolactones may comprise at least about 5% orobanchol.
- the mixture of strigolactones may comprise at least about 7% orobanchol.
- the mixture of strigolactones may comprise at least about 10% orobanchol.
- the mixture of strigolactones may comprise at least about 20% orobanchol.
- the mixture of strigolactones may comprise at least about 30% orobanchol.
- the mixture of strigolactones may comprise at least about 40% orobanchol.
- the mixture of strigolactones may comprise at least about 50% orobanchol.
- the mixture of strigolactones may comprise at least about 60% orobanchol.
- the mixture of strigolactones may comprise at least about 70% orobanchol.
- the mixture of strigolactones may comprise at least about 80% orobanchol.
- the mixture of strigolactones may comprise at least about 85% orobanchol.
- the mixture of strigolactones may comprise at least about 90% orobanchol.
- the mixture of strigolactones may comprise at least about 95% orobanchol.
- the mixture of strigolactones may comprise less than about 95% orobanchol.
- the mixture of strigolactones may comprise less than about 90% orobanchol.
- the mixture of strigolactones may comprise less than about 85% orobanchol.
- the mixture of strigolactones may comprise less than about 80% orobanchol.
- the mixture of strigolactones may comprise less than about 75% orobanchol.
- the mixture of strigolactones may comprise less than about 70% orobanchol.
- the mixture of strigolactones may comprise less than about 60% orobanchol.
- the mixture of strigolactones may comprise less than about 55% orobanchol.
- the mixture of strigolactones may comprise less than about 50% orobanchol.
- the mixture of strigolactones may comprise less than about 40% orobanchol.
- the mixture of strigolactones may comprise less than about 30% orobanchol.
- the mixture of strigolactones may comprise less than about 25% orobanchol.
- the mixture of strigolactones may comprise less than about 20% orobanchol.
- the mixture of strigolactones may comprise less than about 15% orobanchol.
- the mixture of strigolactones may comprise less than about 10% orobanchol.
- the mixture of strigolactones may comprise less than about 5% orobanchol.
- the mixture of strigolactones may comprise less than about 3% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 80% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 70% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 60% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 50% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 40% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 30% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 20% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 10% orobanchol.
- the mixture of strigolactones may comprise between about 1% and 5% orobanchol.
- the mixture of strigolactones may comprise between about 5% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 10% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 20% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 30% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 40% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 50% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 60% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 70% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 80% and 90% orobanchol.
- the mixture of strigolactones may comprise between about 10% and 80% orobanchol.
- the mixture of strigolactones may comprise between about 20% and 70% orobanchol.
- the mixture of strigolactones may comprise between about 30% and 60% orobanchol.
- the mixture of strigolactones may comprise between about 20% and 50% orobanchol.
- the mixture of strigolactones may comprise between about 25% and 50% orobanchol.
- the mixture of strigolactones may comprise at least about 1% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 2% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 5% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 7% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 10% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 20% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 30% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 40% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 50% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 60% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 70% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 80% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 85% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 95% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 95% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 85% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 80% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 75% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 70% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 60% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 55% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 50% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 40% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 30% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 25% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 20% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 15% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 10% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 5% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise less than about 3% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 80% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 70% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 60% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 50% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 40% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 30% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 20% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 10% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 5% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 5% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 10% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 30% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 40% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 50% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 60% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 70% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 80% and 90% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 10% and 80% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 70% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 30% and 60% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 50% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise between about 25% and 50% of 2′ epi-orobanchol.
- the mixture of strigolactones may comprise at least about 1% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 2% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 5% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 7% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 10% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 20% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 30% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 40% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 50% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 60% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 70% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 80% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 85% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 95% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 95% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 85% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 80% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 75% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 70% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 60% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 55% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 50% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 40% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 30% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 25% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 20% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 15% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 10% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 5% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise less than about 3% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 80% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 70% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 60% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 50% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 40% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 30% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 20% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 10% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 5% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 5% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 10% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 30% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 40% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 50% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 60% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 70% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 80% and 90% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 10% and 80% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 70% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 30% and 60% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 50% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise between about 25% and 50% of ent-2′-epi-orobanchol.
- the mixture of strigolactones may comprise at least about 1% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 2% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 5% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 7% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 10% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 20% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 30% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 40% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 50% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 60% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 70% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 80% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 85% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 90% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 95% of ent-orobanchol.
- the mixture of strigolactones may comprise less than about 50% of ent-orobanchol.
- the mixture of strigolactones may comprise less than about 40% of ent-orobanchol.
- the mixture of strigolactones may comprise less than about 30% of ent-orobanchol.
- the mixture of strigolactones may comprise less than about 25% of ent-orobanchol.
- the mixture of strigolactones may comprise less than about 20% of ent-orobanchol.
- the mixture of strigolactones may comprise less than about 15% of ent-orobanchol.
- the mixture of strigolactones may comprise less than about 10% of ent-orobanchol.
- the mixture of strigolactones may comprise less than about 5% of ent-orobanchol.
- the mixture of strigolactones may comprise less than about 3% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 80% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 70% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 60% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 50% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 40% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 30% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 20% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 10% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 5% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 5% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 10% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 30% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 40% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 50% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 60% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 70% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 80% and 90% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 10% and 80% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 70% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 30% and 60% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 50% of ent-orobanchol.
- the mixture of strigolactones may comprise between about 25% and 50% of ent-orobanchol.
- the mixture of strigolactones may comprise at least about 1% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise at least about 2% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise at least about 5% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise at least about 7% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise at least about 10% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise at least about 20% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise at least about 30% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 95% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 85% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 80% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 75% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 70% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 60% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 55% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 50% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 40% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 30% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 25% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 20% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 15% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 10% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 5% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise less than about 3% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 80% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 70% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 60% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 50% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 40% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 30% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 20% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 10% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 5% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 5% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 10% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 20% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 30% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 40% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 50% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 60% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 70% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 80% and 90% of 7-oxo-orobanchol.
- the mixture of strigolactones may comprise between about 1% and 90% of orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 80% of orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 70% of orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 60% of orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 50% of orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 40% of orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 30% of orobanchyl acetate.
- the mixture of strigolactones may comprise between about 30% and 60% of orobanchyl acetate.
- the mixture of strigolactones may comprise between about 20% and 50% of orobanchyl acetate.
- the mixture of strigolactones may comprise between about 25% and 50% of orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 1% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 2% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 5% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 7% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 10% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 20% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 30% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 40% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 50% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 60% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 70% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 80% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 85% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 90% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 95% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 95% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 90% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 85% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 80% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 75% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 70% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 60% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 90% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 80% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 70% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 60% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 50% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 40% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 30% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 20% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 10% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 5% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 5% and 90% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 10% and 90% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 10% and 80% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 20% and 70% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 30% and 60% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 20% and 50% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 25% and 50% of 7-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 1% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 2% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 5% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 7% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 10% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 20% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 85% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 95% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 60% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 55% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 50% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 40% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 30% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise less than about 25% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 30% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 20% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 10% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 1% and 5% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 5% and 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 10% and 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 20% and 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 30% and 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 40% and 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 50% and 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 60% and 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 70% and 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 80% and 90% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 10% and 80% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 20% and 70% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 30% and 60% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 20% and 50% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise between about 25% and 50% of 7-hydroxy-orobanchyl acetate.
- the mixture of strigolactones may comprise at least about 1% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 2% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 5% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 7% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 10% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 20% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 30% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 40% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 50% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 60% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 70% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 80% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 85% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 95% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 95% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 85% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 80% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 75% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 70% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 60% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 55% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 50% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 40% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 30% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 25% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 20% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 15% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 10% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 5% of 5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 3% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 80% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 70% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 60% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 50% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 40% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 30% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 20% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 10% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 5% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 5% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 10% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 30% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 40% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 50% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 60% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 70% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 80% and 90% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 10% and 80% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 70% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 30% and 60% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 50% of 5-deoxystrigol.
- the mixture of strigolactones may comprise between about 25% and 50% of 5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 1% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 2% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 5% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 7% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 10% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 20% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 30% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 40% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 50% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 60% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 70% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 80% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 85% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 95% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 95% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 85% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 80% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 75% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 70% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 60% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 55% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 50% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 40% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 30% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 25% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 20% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 15% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 10% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 5% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 3% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 80% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 70% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 60% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 50% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 40% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 30% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 20% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 10% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 5% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 5% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 10% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 30% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 40% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 50% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 60% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 70% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 80% and 90% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 10% and 80% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 70% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 30% and 60% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 50% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 25% and 50% of 2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 1% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 2% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 5% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 7% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 10% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 20% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 30% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 40% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 50% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 60% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 70% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 80% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 85% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 95% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 95% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 85% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 80% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 75% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 70% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 60% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 55% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 50% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 40% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 30% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 25% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 20% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 15% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 10% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 5% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 3% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 80% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 70% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 60% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 50% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 40% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 30% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 20% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 10% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 5% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 5% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 10% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 30% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 40% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 50% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 60% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 70% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 80% and 90% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 10% and 80% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 70% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 30% and 60% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 50% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 25% and 50% of ent-2′-epi-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 1% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 2% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 5% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 7% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 10% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 20% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 30% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 40% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 50% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 60% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 70% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 80% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 85% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 95% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 95% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 85% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 80% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 75% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 70% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 60% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 55% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 50% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 40% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 30% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 25% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 20% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 15% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 10% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 5% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise less than about 3% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 80% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 70% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 60% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 50% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 40% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 30% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 20% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 10% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 1% and 5% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 5% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 10% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 30% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 40% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 50% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 60% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 70% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 80% and 90% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 10% and 80% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 70% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 30% and 60% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 20% and 50% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise between about 25% and 50% of ent-5-deoxystrigol.
- the mixture of strigolactones may comprise at least about 1% of sorgolactone.
- the mixture of strigolactones may comprise at least about 2% of sorgolactone.
- the mixture of strigolactones may comprise at least about 5% of sorgolactone.
- the mixture of strigolactones may comprise at least about 7% of sorgolactone.
- the mixture of strigolactones may comprise at least about 10% of sorgolactone.
- the mixture of strigolactones may comprise at least about 20% of sorgolactone.
- the mixture of strigolactones may comprise at least about 30% of sorgolactone.
- the mixture of strigolactones may comprise at least about 40% of sorgolactone.
- the mixture of strigolactones may comprise at least about 50% of sorgolactone.
- the mixture of strigolactones may comprise at least about 60% of sorgolactone.
- the mixture of strigolactones may comprise at least about 70% of sorgolactone.
- the mixture of strigolactones may comprise at least about 80% of sorgolactone.
- the mixture of strigolactones may comprise at least about 85% of sorgolactone.
- the mixture of strigolactones may comprise at least about 90% of sorgolactone.
- the mixture of strigolactones may comprise at least about 95% of sorgolactone.
- the mixture of strigolactones may comprise less than about 95% of sorgolactone.
- the mixture of strigolactones may comprise less than about 90% of sorgolactone.
- the mixture of strigolactones may comprise less than about 85% of sorgolactone.
- the mixture of strigolactones may comprise less than about 80% of sorgolactone.
- the mixture of strigolactones may comprise less than about 75% of sorgolactone.
- the mixture of strigolactones may comprise less than about 70% of sorgolactone.
- the mixture of strigolactones may comprise less than about 60% of sorgolactone.
- the mixture of strigolactones may comprise less than about 55% of sorgolactone.
- the mixture of strigolactones may comprise less than about 50% of sorgolactone.
- the mixture of strigolactones may comprise less than about 40% of sorgolactone.
- the mixture of strigolactones may comprise less than about 30% of sorgolactone.
- the mixture of strigolactones may comprise less than about 25% of sorgolactone.
- the mixture of strigolactones may comprise less than about 20% of sorgolactone.
- the mixture of strigolactones may comprise less than about 15% of sorgolactone.
- the mixture of strigolactones may comprise less than about 10% of sorgolactone.
- the mixture of strigolactones may comprise less than about 5% of sorgolactone.
- the mixture of strigolactones may comprise less than about 3% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 80% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 70% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 60% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 50% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 40% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 30% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 20% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 10% of sorgolactone.
- the mixture of strigolactones may comprise between about 1% and 5% of sorgolactone.
- the mixture of strigolactones may comprise between about 5% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 10% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 20% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 30% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 40% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 50% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 60% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 70% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 80% and 90% of sorgolactone.
- the mixture of strigolactones may comprise between about 10% and 80% of sorgolactone.
- the mixture of strigolactones may comprise between about 20% and 70% of sorgolactone.
- the mixture of strigolactones may comprise between about 30% and 60% of sorgolactone.
- the mixture of strigolactones may comprise between about 20% and 50% of sorgolactone.
- the mixture of strigolactones may comprise between about 25% and 50% of sorgolactone.
- the mixture of strigolactones may comprise at least about 1% of sorgomol.
- the mixture of strigolactones may comprise at least about 2% of sorgomol.
- the mixture of strigolactones may comprise at least about 5% of sorgomol.
- the mixture of strigolactones may comprise at least about 7% of sorgomol.
- the mixture of strigolactones may comprise at least about 10% of sorgomol.
- the mixture of strigolactones may comprise at least about 20% of sorgomol.
- the mixture of strigolactones may comprise at least about 30% of sorgomol.
- the mixture of strigolactones may comprise at least about 40% of sorgomol.
- the mixture of strigolactones may comprise at least about 50% of sorgomol.
- the mixture of strigolactones may comprise at least about 60% of sorgomol.
- the mixture of strigolactones may comprise at least about 70% of sorgomol.
- the mixture of strigolactones may comprise at least about 80% of sorgomol.
- the mixture of strigolactones may comprise at least about 85% of sorgomol.
- the mixture of strigolactones may comprise at least about 90% of sorgomol.
- the mixture of strigolactones may comprise at least about 95% of sorgomol.
- the mixture of strigolactones may comprise less than about 95% of sorgomol.
- the mixture of strigolactones may comprise less than about 90% of sorgomol.
- the mixture of strigolactones may comprise less than about 85% of sorgomol.
- the mixture of strigolactones may comprise less than about 80% of sorgomol.
- the mixture of strigolactones may comprise less than about 75% of sorgomol.
- the mixture of strigolactones may comprise less than about 70% of sorgomol.
- the mixture of strigolactones may comprise less than about 60% of sorgomol.
- the mixture of strigolactones may comprise less than about 55% of sorgomol.
- the mixture of strigolactones may comprise less than about 50% of sorgomol.
- the mixture of strigolactones may comprise less than about 40% of sorgomol.
- the mixture of strigolactones may comprise less than about 30% of sorgomol.
- the mixture of strigolactones may comprise less than about 25% of sorgomol.
- the mixture of strigolactones may comprise less than about 20% of sorgomol.
- the mixture of strigolactones may comprise less than about 15% of sorgomol.
- the mixture of strigolactones may comprise less than about 10% of sorgomol.
- the mixture of strigolactones may comprise less than about 5% of sorgomol.
- the mixture of strigolactones may comprise less than about 3% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 80% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 70% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 60% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 50% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 40% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 30% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 20% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 10% of sorgomol.
- the mixture of strigolactones may comprise between about 1% and 5% of sorgomol.
- the mixture of strigolactones may comprise between about 5% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 10% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 20% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 30% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 40% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 50% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 60% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 70% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 80% and 90% of sorgomol.
- the mixture of strigolactones may comprise between about 10% and 80% of sorgomol.
- the mixture of strigolactones may comprise between about 20% and 70% of sorgomol.
- the mixture of strigolactones may comprise between about 30% and 60% of sorgomol.
- the mixture of strigolactones may comprise between about 20% and 50% of sorgomol.
- the mixture of strigolactones may comprise between about 25% and 50% of sorgomol.
- the mixture of strigolactones may comprise at least about 1% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 2% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 5% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 7% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 10% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 20% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 30% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 40% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 50% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 60% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 70% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 80% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 85% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 90% of fabacyl acetate.
- the mixture of strigolactones may comprise at least about 95% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 95% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 90% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 85% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 80% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 75% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 70% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 60% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 55% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 50% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 40% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 30% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 25% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 20% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 15% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 10% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 5% of fabacyl acetate.
- the mixture of strigolactones may comprise less than about 3% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 80% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 70% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 60% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 50% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 40% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 30% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 20% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 10% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 1% and 5% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 5% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 10% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 20% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 30% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 40% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 50% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 60% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 70% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 80% and 90% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 10% and 80% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 20% and 70% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 30% and 60% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 20% and 50% of fabacyl acetate.
- the mixture of strigolactones may comprise between about 25% and 50% of fabacyl acetate.
- the plant propagation material may be at least about 60%, 70%, 75%, 80%, 85%, 90%, 95% or more pure.
- the plant propagation material may be at least about 60% pure.
- the plant propagation material may be at least about 75% pure.
- the plant propagation material may be at least about 80% pure.
- the plant propagation material may be at least about 87% pure.
- the plant propagation material may be at least about 92% pure.
- the plant propagation material may be at least about 95% pure.
- the plant propagation material may be at least about 97% pure.
- the plant propagation material may be a chemical mimic of strigolactone.
- the chemical mimic of strigolactone may be a compound of Formula I, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the chemical mimic of strigolactone may be a compound of Formula II, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the chemical mimic of strigolactone may be a compound of Formula III, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a mixture of chemical mimics of strigolactone.
- the mixture of chemical mimics of strigolactone may comprise the compounds of Formula IV and V.
- the plant propagation material may comprise strigolactone.
- the plant propagation material may comprise a mixture of strigolactones.
- the plant propagation material may comprise strigol.
- the plant propagation material may comprise strigyl acetate.
- the plant propagation material may comprise orobanchol.
- the plant propagation material may comprise orobanchyl acetate.
- the plant propagation material may comprise 5-deoxystrigol.
- the plant propagation material may comprise sorgolactone.
- the plant propagation material may be characterized by having less than 30% impurities.
- the plant propagation material may be characterized by having less than 25% impurities.
- the plant propagation material may be characterized by having less than 20% impurities.
- the plant propagation material may be characterized by having less than 15% impurities.
- the plant propagation material may be characterized by having less than 10% impurities.
- the plant propagation material may be characterized by having less than 7% impurities.
- the plant propagation material may be characterized by having less than 5% impurities.
- the plant propagation material may be characterized by having less than 3% impurities.
- the plant propagation material may be characterized by having less than 2% impurities.
- the plant propagation material may be characterized by having less than 1% impurities.
- the plant propagation material may be characterized by having less than 0.5% impurities.
- the plant propagation material may be a chemical mimic of strigolactone.
- the chemical mimic of strigolactone may be a compound of Formula I, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the chemical mimic of strigolactone may be a compound of Formula II, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the chemical mimic of strigolactone may be a compound of Formula III, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a mixture of chemical mimics of strigolactone.
- the mixture of chemical mimics of strigolactone may comprise the compounds of Formula IV and V.
- the plant propagation material may comprise strigolactone.
- the plant propagation material may comprise a mixture of strigolactones.
- the plant propagation material may comprise strigol.
- the plant propagation material may comprise strigyl acetate.
- the plant propagation material may comprise orobanchol.
- the plant propagation material may comprise orobanchyl acetate.
- the plant propagation material may comprise 5-deoxystrigol.
- the plant propagation material may comprise sorgolactone.
- the plant propagation material may comprise one or more chemical mimics of strigolactone as disclosed herein.
- the plant propagation material may comprise a compound of Formula I, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula II, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula III, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a mixture of chemical mimics of strigolactone.
- formulations comprising a plant propagation material, wherein the plant propagation material comprises a mixture of two diastereomers.
- the two diastereomers may be
- the two diastereomers may have a ratio of 1:1.
- the ratio of Formula IV to Formula V is 1:2.
- the ratio of Formula IV to Formula V may be 1:3.
- the ratio of Formula IV to Formula V may be 1:4.
- the ratio of Formula IV to Formula V may be 1:5.
- the ratio of Formula IV to Formula V may be at least about 1:6; 1:7; 1:8; 1:9; 1:10; 1:11; 1:12; 1:13; 1:14; 1:15: 1:16: 1:17; 1:18; 1:19; 1:20; 1:25; 1:30; 1:35; 1:40; 1:45; 1:50; 1:60; 1:70; 1:80; 1:90; or 1:100.
- the ratio of Formula IV to Formula V may be 1:10.
- the ratio of Formula IV to Formula V may be 1:20.
- the ratio of Formula IV to Formula V may be 1:40.
- the ratio of Formula V to Formula IV may be 1:2.
- the ratio of Formula V to Formula IV may be 1:3.
- the ratio of Formula V to Formula IV may be 1:4.
- the ratio of Formula V to Formula IV may be 1:5.
- the ratio of Formula V to Formula IV may be at least about 1:6; 1:7; 1:8; 1:9; 1:10; 1:11; 1:12; 1:13; 1:14; 1:15: 1:16: 1:17; 1:18; 1:19; 1:20; 1:25; 1:30; 1:35; 1:40; 1:45; 1:50; 1:60; 1:70; 1:80; 1:90; or 1:100.
- the ratio of Formula V to Formula IV may be 1:10.
- the ratio of Formula V to Formula IV may be 1:20.
- the ratio of Formula V to Formula IV may be 1:40.
- the mixture of the two diastereomers may comprise at least about 1% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 5% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 10% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 15% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 20% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 25% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 30% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 40% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 50% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 60% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 70% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 80% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 85% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 90% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 95% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 97% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 95% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 90% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 85% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 80% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 70% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 60% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 50% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 40% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 30% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 20% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 15% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 10% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 5% of Formula IV.
- the mixture of the two diastereomers may comprise less than about 1% of Formula IV.
- the mixture of the two diastereomers may comprise between about 1% to about 90% of Formula IV.
- the mixture of the two diastereomers may comprise between about 5% to about 90% of Formula IV.
- the mixture of the two diastereomers may comprise between about 10% to about 90% of Formula IV.
- the mixture of the two diastereomers may comprise between about 20% to about 90% of Formula IV.
- the mixture of the two diastereomers may comprise between about 30% to about 90% of Formula IV.
- the mixture of the two diastereomers may comprise between about 50% to about 90% of Formula IV.
- the mixture of the two diastereomers may comprise between about 70% to about 90% of Formula IV.
- the mixture of the two diastereomers may comprise between about 1% to about 80% of Formula IV.
- the mixture of the two diastereomers may comprise between about 1% to about 70% of Formula IV.
- the mixture of the two diastereomers may comprise between about 1% to about 60% of Formula IV.
- the mixture of the two diastereomers may comprise between about 1% to about 50% of Formula IV.
- the mixture of the two diastereomers may comprise between about 5% to about 80% of Formula IV.
- the mixture of the two diastereomers may comprise between about 10% to about 80% of Formula IV.
- the mixture of the two diastereomers may comprise between about 10% to about 70% of Formula IV.
- the mixture of the two diastereomers may comprise between about 20% to about 70% of Formula IV.
- the mixture of the two diastereomers may comprise between about 30% to about 90% of Formula IV.
- the mixture of the two diastereomers may comprise at least about 1% of Formula V.
- the mixture of the two diastereomers may comprise at least about 5% of Formula V.
- the mixture of the two diastereomers may comprise at least about 10% of Formula V.
- the mixture of the two diastereomers may comprise at least about 15% of Formula V.
- the mixture of the two diastereomers may comprise at least about 20% of Formula V.
- the mixture of the two diastereomers may comprise at least about 25% of Formula V.
- the mixture of the two diastereomers may comprise at least about 30% of Formula V.
- the mixture of the two diastereomers may comprise at least about 40% of Formula V.
- the mixture of the two diastereomers may comprise at least about 50% of Formula V.
- the mixture of the two diastereomers may comprise at least about 60% of Formula V.
- the mixture of the two diastereomers may comprise at least about 70% of Formula V.
- the mixture of the two diastereomers may comprise at least about 80% of Formula V.
- the mixture of the two diastereomers may comprise at least about 85% of Formula V.
- the mixture of the two diastereomers may comprise at least about 90% of Formula V.
- the mixture of the two diastereomers may comprise at least about 95% of Formula V.
- the mixture of the two diastereomers may comprise less than about 97% of Formula V.
- the mixture of the two diastereomers may comprise less than about 95% of Formula V.
- the mixture of the two diastereomers may comprise less than about 90% of Formula V.
- the mixture of the two diastereomers may comprise less than about 85% of Formula V.
- the mixture of the two diastereomers may comprise less than about 80% of Formula V.
- the mixture of the two diastereomers may comprise less than about 70% of Formula V.
- the mixture of the two diastereomers may comprise less than about 60% of Formula V.
- the mixture of the two diastereomers may comprise less than about 50% of Formula V.
- the mixture of the two diastereomers may comprise less than about 40% of Formula V.
- the mixture of the two diastereomers may comprise less than about 30% of Formula V.
- the mixture of the two diastereomers may comprise less than about 20% of Formula V.
- the mixture of the two diastereomers may comprise less than about 15% of Formula V.
- the mixture of the two diastereomers may comprise less than about 10% of Formula V.
- the mixture of the two diastereomers may comprise less than about 5% of Formula V.
- the mixture of the two diastereomers may comprise less than about 1% of Formula V.
- the mixture of the two diastereomers may comprise between about 1% to about 90% of Formula V.
- the mixture of the two diastereomers may comprise between about 5% to about 90% of Formula V.
- the mixture of the two diastereomers may comprise between about 10% to about 90% of Formula V.
- the mixture of the two diastereomers may comprise between about 20% to about 90% of Formula V.
- the mixture of the two diastereomers may comprise between about 30% to about 90% of Formula V.
- the mixture of the two diastereomers may comprise between about 50% to about 90% of Formula V.
- the mixture of the two diastereomers may comprise between about 70% to about 90% of Formula V.
- the mixture of the two diastereomers may comprise between about 1% to about 80% of Formula V.
- the mixture of the two diastereomers may comprise between about 1% to about 70% of Formula V.
- the mixture of the two diastereomers may comprise between about 1% to about 60% of Formula V.
- the mixture of the two diastereomers may comprise between about 1% to about 50% of Formula V.
- the mixture of the two diastereomers may comprise between about 5% to about 80% of Formula V.
- the mixture of the two diastereomers may comprise between about 10% to about 80% of Formula V.
- the mixture of the two diastereomers may comprise between about 10% to about 70% of Formula V.
- the mixture of the two diastereomers may comprise between about 20% to about 70% of Formula V.
- the mixture of the two diastereomers may comprise between about 30% to about 90% of Formula V.
- formulations comprising a plant propagation material, wherein the plant propagation material is obtained by a process comprising a condensation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the condensation reaction may comprise condensing the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, with methyl formate to produce a hydroxymethylene lactone.
- the condensation reaction may comprise condesing the sesquiterpene lactone with methyl formate to produce a hydroxymethylene lactone.
- the condensation reaction may further comprise a base.
- the base may be potassium tert-butoxide.
- the base may be sodium tert-butoxide.
- the process may further comprise conducting an alkylation reaction.
- the alkylation reaction may comprise reaction of the condensation reaction product with an electrophilic butenolide.
- the alkylation reaction may comprise reaction of the condensation reaction product with a halobutenolide.
- the halobutenolide may be chlorobutenolide.
- the halobutenolide may be bromobutenolide.
- the halobutenolide may be iodobutenolide.
- the sesquiterpene lactone may be sclareolide.
- the bromobutenolide may be 5-bromo-3-methylfuran-2(5H)-one.
- the plant propagation material may comprise a compound of Formula I, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula II, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula III, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- formulations comprising a plant propagation material, wherein the plant propagation material is obtained by a process comprising conducting a hydroxymethylation and/or alkylation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the sesquiterpene lactone may be sclareolide.
- formulations comprising a plant propagation material, wherein the plant propagation material is obtained by a process comprising conducting a hydroxymethylation and/or alkylation reaction on a sesquiterpene lactone.
- the sesquiterpene lactone may be sclareolide.
- the plant propagation material may comprise a compound of Formula I, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula II, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula III, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- formulations comprising a plant propagation material, wherein the plant propagation material is obtained by a process comprising conducting a hydroxymethylation and alkylation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the sesquiterpene lactone may be sclareolide.
- the hydroxymethylation and alkylation may be a one pot procedure.
- the hydroxymethylation may be a reaction between sclareolide and methyl formate in the presence of potassium tert-butoxide.
- the alkylation may be a reaction between the hydroxymethylation product and 5-bromo-3-methylfuran-2(5H)-one.
- the plant propagation material may comprise a compound of Formula I, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula II, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula III, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- formulations comprising a plant propagation material, wherein the plant propagation material is obtained by a process comprising conducting a hydroxymethylation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the sesquiterpene lactone may be sclareolide.
- the hydroxymethylation may be a reaction between sclareolide and methyl formate in the presence of potassium tert-butoxide.
- the plant propagation material may comprise a compound of Formula I, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula II, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula III, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula IV.
- the plant propagation material may comprise a compound of Formula V.
- formulations comprising a plant propagation material, wherein the plant propagation material is obtained by a process comprising conducting an alkylation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the sesquiterpene lactone may be sclareolide.
- the alkylation may be a reaction between the sesquiterpene lactone or product thereof and 5-bromo-3-methylfuran-2(5H)-one.
- the plant propagation material may comprise a compound of Formula I, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula II, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula III, or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula IV.
- the plant propagation material may comprise a compound of Formula V.
- formulations comprising a plant propagation material, wherein the plant propagation material is obtained by a biosynthetic process.
- the biosynthetic process may comprise introducing one or more genes into a cell.
- the biosynthetic process may further comprise culturing the cell.
- the biosynthetic process may further comprise inducing the cell to express one or more genes. Gene expression may result in production of the plant propagation material.
- the biosynthetic process may further comprise purifying the plant propagation material.
- the plant propagation material may comprise strigolactone.
- the plant propagation material may comprise a mixture of strigolactones.
- the plant propagation material may comprise strigol.
- the plant propagation material may comprise strigyl acetate.
- the plant propagation material may comprise orobanchol.
- the plant propagation material may comprise orobanchyl acetate.
- the plant propagation material may comprise 5-deoxystrigol.
- the plant propagation material may comprise sorgolactone.
- formulations comprising a plant propagation material, wherein the plant propagation material is obtained by a process comprising introducing one or more genes into a cell, wherein the one or more genes do not naturally occur in the cell.
- the one or more genes may be referred to as foreign genes.
- the one or more genes may encode a strigolactone pathway.
- the cell may be a eukaryotic cell.
- the one or more genes may be from a eukaryotic cell.
- the eukaryotice cell may be a yeast cell.
- the process may further comprise culturing the cell.
- the process may further comprise inducing the cell to express one or more genes. Gene expression may result in production of the plant propagation material.
- the process may further comprise purifying the plant propagation material.
- the plant propagation material may comprise strigolactone.
- the plant propagation material may comprise a mixture of strigolactones.
- the plant propagation material may comprise strigol.
- the plant propagation material may comprise strigyl acetate.
- the plant propagation material may comprise orobanchol.
- the plant propagation material may comprise orobanchyl acetate.
- the plant propagation material may comprise 5-deoxystrigol.
- the plant propagation material may comprise sorgolactone.
- the plant propagation material may comprise strigolactone.
- the plant propagation material may comprise a mixture of strigolactones.
- the plant propagation material may comprise strigol.
- the plant propagation material may comprise strigyl acetate.
- the plant propagation material may comprise orobanchol.
- the plant propagation material may comprise orobanchyl acetate.
- the plant propagation material may comprise 5-deoxystrigol.
- the plant propagation material may comprise sorgolactone.
- formulations comprising a plant propagation material comprising a mixture of strigolactones.
- the mixture of strigolactones may comprise two or more strigolactones selected from the group comprising strigol, strigyl acetate, orobanchol, orobanchyl acetate, 5-deoxystrigol, and sorgolactone.
- the mixture of strigolactones may comprise strigol.
- the mixture of strigolactones may comprise strigyl acetate.
- the mixture of strigolactones may comprise orobanchol.
- the mixture of strigolactones may comprise orobanchyl acetate.
- the mixture of strigolactones may comprise 5-deoxystrigol.
- the mixture of strigolactones may comprise sorgolactone.
- the formulations may comprise at least about 1% of strigol.
- the formulations may comprise at least about 2% of strigol.
- the formulations may comprise at least about 5% of strigol.
- the formulations may comprise at least about 7% of strigol.
- the formulations may comprise at least about 10% of strigol.
- the formulations may comprise at least about 20% of strigol.
- the formulations may comprise at least about 30% of strigol.
- the formulations may comprise at least about 40% of strigol.
- the formulations may comprise at least about 50% of strigol.
- the formulations may comprise at least about 60% of strigol.
- the formulations may comprise at least about 70% of strigol.
- the formulations may comprise at least about 80% of strigol.
- the formulations may comprise at least about 85% of strigol.
- the formulations may comprise at least about 90% of strigol.
- the formulations may comprise at least about 95% of strigol.
- the formulations may comprise less than about 95% of strigol.
- the formulations may comprise less than about 90% of strigol.
- the formulations may comprise less than about 85% of strigol.
- the formulations may comprise less than about 80% of strigol.
- the formulations may comprise less than about 75% of strigol.
- the formulations may comprise less than about 70% of strigol.
- the formulations may comprise less than about 60% of strigol.
- the formulations may comprise less than about 55% of strigol.
- the formulations may comprise less than about 50% of strigol.
- the formulations may comprise less than about 40% of strigol.
- the formulations may comprise less than about 30% of strigol.
- the formulations may comprise less than about 25% of strigol.
- the formulations may comprise less than about 20% of strigol.
- the formulations may comprise less than about 15% of strigol.
- the formulations may comprise less than about 10% of strigol.
- the formulations may comprise less than about
- the formulations may comprise between about 1% and 90% of strigol.
- the formulations may comprise between about 1% and 80% of strigol.
- the formulations may comprise between about 1% and 70% of strigol.
- the formulations may comprise between about 1% and 60% of strigol.
- the formulations may comprise between about 1% and 50% of strigol.
- the formulations may comprise between about 1% and 40% of strigol.
- the formulations may comprise between about 1% and 30% of strigol.
- the formulations may comprise between about 1% and 20% of strigol.
- the formulations may comprise between about 1% and 10% of strigol.
- the formulations may comprise between about 1% and 5% of strigol.
- the formulations may comprise between about 5% and 90% of strigol.
- the formulations may comprise between about 10% and 90% of strigol.
- the formulations may comprise between about 20% and 90% of strigol.
- the formulations may comprise between about 30% and 90% of strigol.
- the formulations may comprise between about 40% and 90% of strigol.
- the formulations may comprise between about 50% and 90% of strigol.
- the formulations may comprise between about 60% and 90% of strigol.
- the formulations may comprise between about 70% and 90% of strigol.
- the formulations may comprise between about 80% and 90% of strigol.
- the formulations may comprise between about 10% and 80% of strigol.
- the formulations may comprise between about 20% and 70% of strigol.
- the formulations may comprise between about 30% and 60% of strigol.
- the formulations may comprise between about 20% and 50% of strigol.
- the formulations may comprise between about 25% and 50% of strigol.
- the formulations may comprise at least about 1% of strigyl acetate.
- the formulations may comprise at least about 2% of strigyl acetate.
- the formulations may comprise at least about 5% of strigyl acetate.
- the formulations may comprise at least about 7% of strigyl acetate.
- the formulations may comprise at least about 10% of strigyl acetate.
- the formulations may comprise at least about 20% of strigyl acetate.
- the formulations may comprise at least about 30% of strigyl acetate.
- the formulations may comprise at least about 40% of strigyl acetate.
- the formulations may comprise at least about 50% of strigyl acetate.
- the formulations may comprise at least about 60% of strigyl acetate.
- the formulations may comprise at least about 70% of strigyl acetate.
- the formulations may comprise at least about 80% of strigyl acetate.
- the formulations may comprise at least about 85% of strigyl acetate.
- the formulations may comprise at least about 90% of strigyl acetate.
- the formulations may comprise at least about 95% of strigyl acetate.
- the formulations may comprise less than about 95% of strigyl acetate.
- the formulations may comprise less than about 90% of strigyl acetate.
- the formulations may comprise less than about 85% of strigyl acetate.
- the formulations may comprise less than about 80% of strigyl acetate.
- the formulations may comprise less than about 75% of strigyl acetate.
- the formulations may comprise less than about 70% of strigyl acetate.
- the formulations may comprise less than about 60% of strigyl acetate.
- the formulations may comprise less than about 55% of strigyl acetate.
- the formulations may comprise less than about 50% of strigyl acetate.
- the formulations may comprise less than about 40% of strigyl acetate.
- the formulations may comprise less than about 30% of strigyl acetate.
- the formulations may comprise less than about 25% of strigyl acetate.
- the formulations may comprise less than about 20% of strigyl acetate.
- the formulations may comprise less than about 15% of strigyl acetate.
- the formulations may comprise less than about 10% of strigyl acetate.
- the formulations may comprise less than about 5% of strigyl acetate.
- the formulations may comprise less than about 3% of strigyl acetate.
- the formulations may comprise between about 1% and 90% of strigyl acetate.
- the formulations may comprise between about 1% and 80% of strigyl acetate.
- the formulations may comprise between about 1% and 70% of strigyl acetate.
- the formulations may comprise between about 1% and 60% of strigyl acetate.
- the formulations may comprise between about 1% and 50% of strigyl acetate.
- the formulations may comprise between about 1% and 40% of strigyl acetate.
- the formulations may comprise between about 1% and 30% of strigyl acetate.
- the formulations may comprise between about 1% and 20% of strigyl acetate.
- the formulations may comprise between about 1% and 10% of strigyl acetate.
- the formulations may comprise between about 1% and 5% of strigyl acetate.
- the formulations may comprise between about 5% and 90% of strigyl acetate.
- the formulations may comprise between about 10% and 90% of strigyl acetate.
- the formulations may comprise between about 20% and 90% of strigyl acetate.
- the formulations may comprise between about 30% and 90% of strigyl acetate.
- the formulations may comprise between about 40% and 90% of strigyl acetate.
- the formulations may comprise between about 50% and 90% of strigyl acetate.
- the formulations may comprise between about 60% and 90% of strigyl acetate.
- the formulations may comprise between about 70% and 90% of strigyl acetate.
- the formulations may comprise between about 80% and 90% of strigyl acetate.
- the formulations may comprise between about 10% and 80% of strigyl acetate.
- the formulations may comprise between about 20% and 70% of strigyl acetate.
- the formulations may comprise between about 30% and 60% of strigyl acetate.
- the formulations may comprise between about 20% and 50% of strigyl acetate.
- the formulations may comprise between about 25% and 50% of strigyl acetate.
- the formulations may comprise at least about 1% of orobanchol.
- the formulations may comprise at least about 2% of orobanchol.
- the formulations may comprise at least about 5% of orobanchol.
- the formulations may comprise at least about 7% of orobanchol.
- the formulations may comprise at least about 10% of orobanchol.
- the formulations may comprise at least about 20% of orobanchol.
- the formulations may comprise at least about 30% of orobanchol.
- the formulations may comprise at least about 40% of orobanchol.
- the formulations may comprise at least about 50% of orobanchol.
- the formulations may comprise at least about 60% of orobanchol.
- the formulations may comprise at least about 70% of orobanchol.
- the formulations may comprise at least about 80% of orobanchol.
- the formulations may comprise at least about 85% of orobanchol.
- the formulations may comprise at least about 90% of orobanchol.
- the formulations may comprise at least about 95%
- the formulations may comprise less than about 95% of orobanchol.
- the formulations may comprise less than about 90% of orobanchol.
- the formulations may comprise less than about 85% of orobanchol.
- the formulations may comprise less than about 80% of orobanchol.
- the formulations may comprise less than about 75% of orobanchol.
- the formulations may comprise less than about 70% of orobanchol.
- the formulations may comprise less than about 60% of orobanchol.
- the formulations may comprise less than about 55% of orobanchol.
- the formulations may comprise less than about 50% of orobanchol.
- the formulations may comprise less than about 40% of orobanchol.
- the formulations may comprise less than about 30% of orobanchol.
- the formulations may comprise less than about 25% of orobanchol.
- the formulations may comprise less than about 20% of orobanchol.
- the formulations may comprise less than about 15% of orobanchol.
- the formulations may comprise less than about 10% of orobanchol.
- the formulations may comprise less than about 5% of orobanchol.
- the formulations may comprise less than about 3% of orobanchol.
- the formulations may comprise between about 1% and 90% of orobanchol.
- the formulations may comprise between about 1% and 80% of orobanchol.
- the formulations may comprise between about 1% and 70% of orobanchol.
- the formulations may comprise between about 1% and 60% of orobanchol.
- the formulations may comprise between about 1% and 50% of orobanchol.
- the formulations may comprise between about 1% and 40% of orobanchol.
- the formulations may comprise between about 1% and 30% of orobanchol.
- the formulations may comprise between about 1% and 20% of orobanchol.
- the formulations may comprise between about 1% and 10% of orobanchol.
- the formulations may comprise between about 1% and 5% of orobanchol.
- the formulations may comprise between about 5% and 90% of orobanchol.
- the formulations may comprise between about 10% and 90% of orobanchol.
- the formulations may comprise between about 20% and 90% of orobanchol.
- the formulations may comprise between about 30% and 90% of orobanchol.
- the formulations may comprise between about 40% and 90% of orobanchol.
- the formulations may comprise between about 50% and 90% of orobanchol.
- the formulations may comprise between about 60% and 90% of orobanchol.
- the formulations may comprise between about 70% and 90% of orobanchol.
- the formulations may comprise between about 80% and 90% of orobanchol.
- the formulations may comprise between about 10% and 80% of orobanchol.
- the formulations may comprise between about 20% and 70% of orobanchol.
- the formulations may comprise between about 30% and 60% of orobanchol.
- the formulations may comprise between about 20% and 50% of orobanchol.
- the formulations may comprise between about 25% and 50% of orobanchol.
- the formulations may comprise at least about 1% of orobanchol acetate.
- the formulations may comprise at least about 2% of orobanchol acetate.
- the formulations may comprise at least about 5% of orobanchol acetate.
- the formulations may comprise at least about 7% of orobanchol acetate.
- the formulations may comprise at least about 10% of orobanchol acetate.
- the formulations may comprise at least about 20% of orobanchol acetate.
- the formulations may comprise at least about 30% of orobanchol acetate.
- the formulations may comprise at least about 40% of orobanchol acetate.
- the formulations may comprise at least about 50% of orobanchol acetate.
- the formulations may comprise at least about 60% of orobanchol acetate.
- the formulations may comprise at least about 70% of orobanchol acetate.
- the formulations may comprise at least about 80% of orobanchol acetate.
- the formulations may comprise at least about 85% of orobanchol acetate.
- the formulations may comprise at least about 90% of orobanchol acetate.
- the formulations may comprise at least about 95% of orobanchol acetate.
- the formulations may comprise less than about 95% of orobanchol acetate.
- the formulations may comprise less than about 90% of orobanchol acetate.
- the formulations may comprise less than about 85% of orobanchol acetate.
- the formulations may comprise less than about 80% of orobanchol acetate.
- the formulations may comprise less than about 75% of orobanchol acetate.
- the formulations may comprise less than about 70% of orobanchol acetate.
- the formulations may comprise less than about 60% of orobanchol acetate.
- the formulations may comprise less than about 55% of orobanchol acetate.
- the formulations may comprise less than about 50% of orobanchol acetate.
- the formulations may comprise less than about 40% of orobanchol acetate.
- the formulations may comprise less than about 30% of orobanchol acetate.
- the formulations may comprise less than about 25% of orobanchol acetate.
- the formulations may comprise less than about 20% of orobanchol acetate.
- the formulations may comprise less than about 15% of orobanchol acetate.
- the formulations may comprise less than about 10% of orobanchol acetate.
- the formulations may comprise less than about 5% of orobanchol acetate.
- the formulations may comprise less than about 3% of orobanchol acetate.
- the formulations may comprise between about 1% and 90% of orobanchol acetate.
- the formulations may comprise between about 1% and 80% of orobanchol acetate.
- the formulations may comprise between about 1% and 70% of orobanchol acetate.
- the formulations may comprise between about 1% and 60% of orobanchol acetate.
- the formulations may comprise between about 1% and 50% of orobanchol acetate.
- the formulations may comprise between about 1% and 40% of orobanchol acetate.
- the formulations may comprise between about 1% and 30% of orobanchol acetate.
- the formulations may comprise between about 1% and 20% of orobanchol acetate.
- the formulations may comprise between about 1% and 10% of orobanchol acetate.
- the formulations may comprise between about 1% and 5% of orobanchol acetate.
- the formulations may comprise between about 5% and 90% of orobanchol acetate.
- the formulations may comprise between about 10% and 90% of orobanchol acetate.
- the formulations may comprise between about 20% and 90% of orobanchol acetate.
- the formulations may comprise between about 30% and 90% of orobanchol acetate.
- the formulations may comprise between about 40% and 90% of orobanchol acetate.
- the formulations may comprise between about 50% and 90% of orobanchol acetate.
- the formulations may comprise between about 60% and 90% of orobanchol acetate.
- the formulations may comprise between about 70% and 90% of orobanchol acetate.
- the formulations may comprise between about 80% and 90% of orobanchol acetate.
- the formulations may comprise between about 10% and 80% of orobanchol acetate.
- the formulations may comprise between about 20% and 70% of orobanchol acetate.
- the formulations may comprise between about 30% and 60% of orobanchol acetate.
- the formulations may comprise between about 20% and 50% of orobanchol acetate.
- the formulations may comprise between about 25% and 50% of orobanchol acetate.
- the formulations may comprise at least about 1% of 5-deoxystrigol.
- the formulations may comprise at least about 2% of 5-deoxystrigol.
- the formulations may comprise at least about 5% of 5-deoxystrigol.
- the formulations may comprise at least about 7% of 5-deoxystrigol.
- the formulations may comprise at least about 10% of 5-deoxystrigol.
- the formulations may comprise at least about 20% of 5-deoxystrigol.
- the formulations may comprise at least about 30% of 5-deoxystrigol.
- the formulations may comprise at least about 40% of 5-deoxystrigol.
- the formulations may comprise at least about 50% of 5-deoxystrigol.
- the formulations may comprise at least about 60% of 5-deoxystrigol.
- the formulations may comprise at least about 70% of 5-deoxystrigol.
- the formulations may comprise at least about 80% of 5-deoxystrigol.
- the formulations may comprise at least about 85% of 5-deoxystrigol.
- the formulations may comprise at least about 90% of 5-deoxystrigol.
- the formulations may comprise at least about 95% of 5-deoxystrigol.
- the formulations may comprise less than about 95% of 5-deoxystrigol.
- the formulations may comprise less than about 90% of 5-deoxystrigol.
- the formulations may comprise less than about 85% of 5-deoxystrigol.
- the formulations may comprise less than about 80% of 5-deoxystrigol.
- the formulations may comprise less than about 75% of 5-deoxystrigol.
- the formulations may comprise less than about 70% of 5-deoxystrigol.
- the formulations may comprise less than about 60% of 5-deoxystrigol.
- the formulations may comprise less than about 55% of 5-deoxystrigol.
- the formulations may comprise less than about 50% of 5-deoxystrigol.
- the formulations may comprise less than about 40% of 5-deoxystrigol.
- the formulations may comprise less than about 30% of 5-deoxystrigol.
- the formulations may comprise less than about 25% of 5-deoxystrigol.
- the formulations may comprise less than about 20% of 5-deoxystrigol.
- the formulations may comprise less than about 15% of 5-deoxystrigol.
- the formulations may comprise less than about 10% of 5-deoxystrigol.
- the formulations may comprise less than about 5% of 5-deoxystrigol.
- the formulations may comprise less than about 3% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 80% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 70% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 60% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 50% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 40% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 30% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 20% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 10% of 5-deoxystrigol.
- the formulations may comprise between about 1% and 5% of 5-deoxystrigol.
- the formulations may comprise between about 5% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 10% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 20% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 30% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 40% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 50% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 60% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 70% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 80% and 90% of 5-deoxystrigol.
- the formulations may comprise between about 10% and 80% of 5-deoxystrigol.
- the formulations may comprise between about 20% and 70% of 5-deoxystrigol.
- the formulations may comprise between about 30% and 60% of 5-deoxystrigol.
- the formulations may comprise between about 20% and 50% of 5-deoxystrigol.
- the formulations may comprise between about 25% and 50% of 5-deoxystrigol.
- the formulations may comprise at least about 1% of sorgolactone.
- the formulations may comprise at least about 2% of sorgolactone.
- the formulations may comprise at least about 5% of sorgolactone.
- the formulations may comprise at least about 7% of sorgolactone.
- the formulations may comprise at least about 10% of sorgolactone.
- the formulations may comprise at least about 20% of sorgolactone.
- the formulations may comprise at least about 30% of sorgolactone.
- the formulations may comprise at least about 40% of sorgolactone.
- the formulations may comprise at least about 50% of sorgolactone.
- the formulations may comprise at least about 60% of sorgolactone.
- the formulations may comprise at least about 70% of sorgolactone.
- the formulations may comprise at least about 80% of sorgolactone.
- the formulations may comprise at least about 85% of sorgolactone.
- the formulations may comprise at least about 90% of sorgolactone.
- the formulations may comprise at least about 95% of sorgolactone.
- the formulations may comprise less than about 95% of sorgolactone.
- the formulations may comprise less than about 90% of sorgolactone.
- the formulations may comprise less than about 85% of sorgolactone.
- the formulations may comprise less than about 80% of sorgolactone.
- the formulations may comprise less than about 75% of sorgolactone.
- the formulations may comprise less than about 70% of sorgolactone.
- the formulations may comprise less than about 60% of sorgolactone.
- the formulations may comprise less than about 55% of sorgolactone.
- the formulations may comprise less than about 50% of sorgolactone.
- the formulations may comprise less than about 40% of sorgolactone.
- the formulations may comprise less than about 30% of sorgolactone.
- the formulations may comprise less than about 25% of sorgolactone.
- the formulations may comprise less than about 20% of sorgolactone.
- the formulations may comprise less than about 15% of sorgolactone.
- the formulations may comprise less than about 10% of sorgolactone.
- the formulations may comprise less than about 5% of sorgolactone.
- the formulations may comprise less than about 3% of sorgolactone.
- the formulations may comprise between about 1% and 90% of sorgolactone.
- the formulations may comprise between about 1% and 80% of sorgolactone.
- the formulations may comprise between about 1% and 70% of sorgolactone.
- the formulations may comprise between about 1% and 60% of sorgolactone.
- the formulations may comprise between about 1% and 50% of sorgolactone.
- the formulations may comprise between about 1% and 40% of sorgolactone.
- the formulations may comprise between about 1% and 30% of sorgolactone.
- the formulations may comprise between about 1% and 20% of sorgolactone.
- the formulations may comprise between about 1% and 10% of sorgolactone.
- the formulations may comprise between about 1% and 5% of sorgolactone.
- the formulations may comprise between about 5% and 90% of sorgolactone.
- the formulations may comprise between about 10% and 90% of sorgolactone.
- the formulations may comprise between about 20% and 90% of sorgolactone.
- the formulations may comprise between about 30% and 90% of sorgolactone.
- the formulations may comprise between about 40% and 90% of sorgolactone.
- the formulations may comprise between about 50% and 90% of sorgolactone.
- the formulations may comprise between about 60% and 90% of sorgolactone.
- the formulations may comprise between about 70% and 90% of sorgolactone.
- the formulations may comprise between about 80% and 90% of sorgolactone.
- the formulations may comprise between about 10% and 80% of sorgolactone.
- the formulations may comprise between about 20% and 70% of sorgolactone.
- the formulations may comprise between about 30% and 60% of sorgolactone.
- the formulations may comprise between about 20% and 50% of sorgolactone.
- the formulations may
- the formulations disclosed herein may further comprise one or more pesticides.
- the pesticide may be a biopesticide.
- a biopesticide may be a form of a pesticide that is based on microorganisms or natural products.
- a biopesticide may include naturally occurring substances that control pests (biochemical pesticides), microorganisms that control pests (microbial pesticides), and pesticidal substances produced by plants containing added genetic material (plant-incorporated protectants) or PIPs.
- biopesticides include, but are not limited to, gluocosinolate, chitosan, spinosad, alkaloids, terpenoids, phenolics, pyrethroids, rotenoids, nicotinoids, strychnine, scilliroside, canola oil and baking soda.
- the pesticide may be an organophosphate pesticide, carbamate pesticide, organochlorine insecticide, pyrethroid pesticide, sulfonylurea pesticides, or a combination thereof.
- the pesticide may be a herbicide, algicide, avidicide, bactericide, fungicide, insecticide, miticide, molluscicide, nematicide, rodenticide, virucide, or a combination thereof.
- the formulations disclosed herein may be formulated as a dry sprayable formulation.
- dry sprayable formulations include, but are not limited to, wettable powders and water dispersible granules.
- Wettable powders may comprise plant propagation materials that have been microionized to powder form. Wettable powders may be applied as suspended particles after dispertion into water.
- Water dispersible granules may consist of granules that are applied after disintegration or dispersion in water.
- the water dispersible granules may comprise particles within the range of 0.2 to 4 mm. Water dispersible granules may be formed by agglomeration, spray drying, or extrusion techniques.
- the formulations may be formulated as a liquid sprayable formulation.
- liquid sprayable formulations include, but are not limited to, soluble concentrates, suspension concentrates, emulsifiable concentrates, microemulsions, oil dispersions, and microencapsulated particles.
- Suspension concentrates may comprise a stable suspension of the propagation material in a fluid usually intended for dilution with water before use.
- Emulsifiable concentrates may comprise a plant propagation material with an emulsifying agent in a water insoluble organic solvent which will form an emulsion when added to water.
- Microemulsions may comprise a plant propagation material with an emulsifying agent in a water insoluble organic solvent which will form a solution/emulsion when added to water.
- compositions may be formulated as a dry spreadable granule formulation.
- the dry spreadable granule formulation may comprise soil applied granule on inert or fertilizer carriers.
- the formulations may be formulated as a seed treatment or seed dressing.
- the formulations may be formulated for rapid release.
- the formulations may be formulated for slow release.
- the formulations may further comprise one or more stabilizers and/or other additives.
- stabilizers and/or additives include, but are not limited to, penetration agents, adhesives, anticaking agents, dyes, dispersants, wetting agents, emulsifying agents, defoamers, antimicrobials, antifreeze, pigments, colorants, buffers, and carriers.
- the formulations may further comprise surfanctans and/or adjuvants.
- the formulations may further comprise one or more carriers.
- carriers include, but are not limited to, solid carriers, sponges, textiles, and synthetic materials.
- the synthetic material may be a porous synthetic material.
- Additional carriers include organic carriers, such as waxes, linolin, paraffin, dextrose granules, sucrose granules and maltose-dextrose granules.
- the carrier is an anorganic carrier such as natural clays, kaolin, pyrophyllite, bentonite, alumina, montmorillonite, kieselguhr, chalk, diatomaceous earths, calcium phosphates, calcium and magnesium carbonates, sulphur, lime, flours or talc.
- the formulation may be adsorbed into the carrier.
- the carrier may be characterized by enabling release of the plant propagation material.
- the formulations may further comprise one or more dispersants.
- the dispersant may be an negatively charged anion dispersant.
- the dispersant may be a nonionic dispersant.
- the formulations may further comprise fertilizer.
- the fertilizer may be a chemical fertilizer.
- the fertilizer may be an organic fertilizer.
- the fertilizer may be an inorganic fertilizer.
- the fertilizer may be a granulated or powdered fertilizers.
- the fertilizer may be a liquid fertilizer.
- the fertilizer may be a slow-release fertilizer.
- the method may comprise a chemical synthesis.
- the method may comprise (i) hydroxymethylation of an optionally substituted decahydronaphtho[2,1-b]furan-2(3aH)-one; and (ii) subsequent alkylation with
- the plant propagation material may be a chemical mimic of strigolactone.
- the plant propagation material may be a compound of Formula I, II, III, IV, V, VI, VII, VIII, IX or X.
- the method of preparing a compound of Formula I, II, III, IV, V, VI, VII, VIII, IX or X may comprise (i) hydroxymethylation of an optionally substituted decahydronaphtho[2,1-b]furan-2(3aH)-one; and (ii) subsequent alkylation with
- R 17 is H, alkyl, halo, or haloalkyl and X is Cl, Br, or I.
- the hydroxymethylation and alkylation may be a one pot procedure.
- the optionally substituted decahydronaphtho[2,1-b]furan-2(3aH)-one may be sclareolide.
- R 17 may be H or alkyl.
- R 17 may be H.
- R 17 may be alkyl.
- the hydroxymethylation may be a reaction between sclareolide and methyl formate in the presence of potassium tert-butoxide.
- the alkylation may be a reaction between the hydroxymethylation product and 5-bromo-3-methylfuran-2(5H)-one.
- (+)-sclareolide 710 from (E)-(+)-nerolidol 720 involves the [2,3] sigmatropic rearrangement of an allylic alcohol to the homologous amide followed by hydrolysis of the amide to the acid and biomimetic enantioselective cyclization of acid promoted by (R)-2-benzyloxy-20-hydroxy-1,10-binaphthyl[(R)-benzyl-BINOL] and SnCl4 (chiral LBA).
- (+)-sclareolide and (+)-tetrahydroactinidiolid reaction conditions in FIG. 7 can be: (i) DMFDMA, xylene, reflux, 14 h; (ii) KOH, MeOH-water, reflux, 12 h; (iii) 2-benzyloxy-20-hydroxy-1,10-binaphthyl, SnCl4, toluene, ⁇ 78° C., 3 h, and at ⁇ 20° C., 3 d.
- DMFDMA DMFDMA
- xylene xylene
- reflux 14 h
- KOH MeOH-water
- reflux reflux
- 2-benzyloxy-20-hydroxy-1,10-binaphthyl, SnCl4, toluene ⁇ 78° C., 3 h, and at ⁇ 20° C., 3 d.
- (E)-(+)-Nerolidol 720 were heated with N,N-dimethylformamide dimethyl acetal (DMFDMA) to achieve one carbon homologation to the corresponding starting materials with incorporation of a terminal amide functionality.
- DMFDMA N,N-dimethylformamide dimethyl acetal
- Similar methods can also be used to synthesize compound I, II, III, IV, V, VI, VII, VIII, IX or X comprising a nitrogen atom.
- compound I, II, III, IV, V, VI, VII, VIII, IX or X comprising one or more nitrogen atoms can be synthesized by substituting a carbon atom with a nitrogen atom in the starting compound in the disclosed synthetic approach.
- a method of producing a plant propagation material may comprise conducting a condensation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, thereby producing a plant propagation material.
- a method of producing a plant propagation material may comprise conducting a condensation reaction on a sesquiterpene lactone, thereby producing a plant propagation material.
- the sesquiterpene lactone may be sclareolide.
- the plant propagation material may be a compound of Formula (I).
- the plant propagation material may be a compound of Formula (II).
- the plant propagation material may be a compound of Formula (III).
- a method of producing a plant propagation material comprises conducting a hydroxymethylation and/or alkylation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, thereby producing a plant propagation material.
- a method of producing a plant propagation material may comprise conducting a hydroxymethylation and/or alkylation reaction on a sesquiterpene lactone, thereby producing a plant propagation material.
- a method of producing a plant propagation material may comprise conducting a hydroxymethylation and alkylation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, thereby producing a plant propagation material.
- a method of producing a plant propagation material may comprise conducting a hydroxymethylation and alkylation reaction on a sesquiterpene lactone, thereby producing a plant propagation material.
- a method of producing a plant propagation material may comprise conducting a hydroxymethylation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, thereby producing a plant propagation material.
- a method of producing a plant propagation material may comprise conducting a hydroxymethylation reaction on a sesquiterpene lactone, thereby producing a plant propagation material.
- a method of producing a plant propagation material may comprise conducting an alkylation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, thereby producing a plant propagation material.
- a method of producing a plant propagation material may comprise conducting an alkylation reaction on a sesquiterpene lactone, thereby producing a plant propagation material.
- the sesquiterpene lactone may be sclareolide.
- the plant propagation material may be a compound of Formula (I).
- the plant propagation material may be a compound of Formula (II).
- the plant propagation material may be a compound of Formula (III).
- the plant propagation material may have the structure of Formula (I):
- R 1 , R 2 , R 4 , R 5 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , and R 17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ;
- R 3 and R 6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR 18 ; or R 3 and R 6 together form a direct bond to provide a double bond;
- each R 18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, or —C(O)R 19 ;
- each R 19 is independently alkyl, haloalkyl, aryl, or heteroaryl;
- n 0, 1, or 2;
- n 1 or 2.
- the methods disclosed herein may comprise one or more condensation reactions.
- the condensation reaction may comprise condensing the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof, with methyl formate to produce a hydroxymethylene lactone.
- the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof may be condensed with an excess of methyl formate.
- the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof may be condensed with two-fold excess of methyl formate.
- the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof may be condensed with three-fold excess of methyl formate.
- the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof may be condensed with four-fold excess of methyl formate.
- the sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof may be condensed with five-fold excess of methyl formate.
- the condensation reaction may further comprise potassium tert-butoxide.
- the methods disclosed herein may comprise one or more alkylation reactions.
- the method of producing a plant propagation material may further comprise conducting an alkylation reaction.
- the alkylation reaction may comprise alkylating the condensation reaction product with a bromobutenolide.
- the alkylation reaction may produce a mixture of two diastereomers. The two diastereomers may be
- the methods disclosed herein may comprise the use of a terpene.
- the methods disclosed herein may comprise the use of a lactone.
- the methods disclosed herein may comprise the use of a sesquiterpene.
- the methods disclosed herein may comprise the use of a sesquiterpene lactone.
- the sesquiterpene lactone may be a germacranolide. Examples of germacranolides include, but are not limited to, germacranolides from Mikania guaco as disclosed in Rüngeler et al. (2001 , Phytochemistry, 56(5):475-489).
- the sesquiterpene lactone may be a heliangolide.
- the sesquiterpene lactone may be a guaianolide.
- the sesquiterpene lactone may be a pseudoguaianolide.
- the sesquiterpene lactone may be a hypocretenolide.
- the sesquiterpene lactone may be an eudesmanolide.
- the sesquiterpene lactone may be an eudesmanolide ⁇ -santonin.
- the sesquiterpene lactone may be a ⁇ -santonin.
- the sesquiterpene lactone may be a parthenolide.
- the sesquiterpene lactone may be a lactuside A.
- the sesquiterpene lactone may be a helanalin.
- the sesquiterpene lactone may be a hymenin.
- the sesquiterpene lactone may be a lettucenin A.
- the sesquiterpene lactone may be a parthenin.
- the sesquiterpene lactone may be a tenulin.
- the sesquiterpene lactone may be a cadinanolide.
- the sesquiterpene lactone may be an artemisinin.
- the sesquiterpene lactone may be a seco-cadinanolide.
- the sesquiterpene lactone may be an artemisinic acid.
- the sesquiterpene lactone may be extracted or derived from a plant.
- the plant may be Salvia sclarea, Salvia yosgadensis , or cigar tobacco.
- the sesquiterpene lactone may be extracted or derived from one or more plants selected from laurus nobilis, chrysanthemum, pyrethrum, Star anise, Ragweed, Sneezeweed, Ironweed, Sagebrush, Wormwood, Mugwort, Boneset, Poverty weed, Marsh elder, Cocklebur, Burdock, Chamomile, Feverfew, Artichoke, Gailladrin, Parthenium, Sunflower, Lettuce, Spinach, Yellow star thistle, Ginkgo biloba, or a combination thereof.
- the sesquiterpene lactone may be extracted or derived from a sage plant.
- the sage plant may be a clary sage plant.
- the method may comprise a biological synthesis.
- the method may comprise introducing one or more genes that encode a strigolactone pathway into a cell.
- the method may comprise introducing two or more genes that encode a strigolactone pathway into a cell.
- the method may comprise introducing three or more genes that encode a strigolactone pathway into a cell.
- the method may comprise introducing four or more genes that encode a strigolactone pathway into a cell.
- the method may comprise introducing five or more genes that encode a strigolactone pathway into a cell.
- the method may comprise introducing six or more genes that encode a strigolactone pathway into a cell.
- the method may comprise introducing 7, 8, 9, 10, 11, 12, 13, 14, 15 or more genes that encode a strigolactone pathway into a cell.
- the method of preparing a plant propagation material may comprise introducing a plurality of genes into the cell. At least one of the plurality of genes may encode a strigolactone pathway. At least two of the plurality of genes may encode a strigolactone pathway. At least three of the plurality of genes may encode a strigolactone pathway. At least four of the plurality of genes may encode a strigolactone pathway. At least five of the plurality of genes may encode a strigolactone pathway. At least six of the plurality of genes may encode a strigolactone pathway. At least 7, 8, 9, 10, 11, 12, 13, 14, 15 or more of the plurality of genes may encode a strigolactone pathway.
- the method of preparing the plant propagation material may comprise engineering a cell to produce a metabolite, wherein the metabolite is not a natural metabolite of the cell.
- the metabolite may be lycopene.
- Engineering the cell to produce the metabolite may comprise introducing one or more genes into the cell.
- the one or more genes may comprise crtE, crtB, crtI or a combination thereof.
- Engineering the cell to produce the metabolite may comprise introducing one or more genes into the cell, wherein the one or more genes are selected from crtE, crtB, crtI or a combination thereof.
- Engineering the cell to produce the metabolite may comprise introducing a plurality of genes into the cell, wherein at least two of the plurality of genes are selected from crtE, crtB, and crtI.
- Engineering the cell to produce the metabolite may comprise introducing a plurality of genes into the cell, wherein at least three of the plurality of genes are selected from crtE, crtB, and crtI.
- the methods may further comprise introducing one or more additional genes into the engineered cell.
- the one or more genes may be introduced into the engineered cell by transformation.
- Introduction of the one or more genes into the engineered cell may comprise use of a gene cassette comprising the one or more genes.
- the one or more genes may comprise D27, CCD7, CCD8, P450 enzyme, or a combination thereof.
- the P450 enzyme may be MAX1. Transformation of the engineered cell with the one or more genes may result in production of the plant propagation material.
- the plant propagation material may be strigolactone.
- the plant propagation material may comprise a strigolactone analogue.
- the methods may further comprise introducing a plurality of additional genes into the engineered cell.
- the plurality of additional genes may be introduced into the engineered cell by transformation.
- Introduction of the plurality of genes into the engineered cell may comprise use of a gene cassette comprising at least one of the plurality of genes.
- Introduction of the plurality of genes into the engineered cell may comprise use of a gene cassette comprising at least two of the plurality of genes.
- Introduction of the plurality of genes into the engineered cell may comprise use of a gene cassette comprising at least three of the plurality of genes.
- Introduction of the plurality of genes into the engineered cell may comprise use of a gene cassette comprising at least four of the plurality of genes.
- At least one of the plurality of genes may be selected from CCD7, CCD8, or P450 enzyme.
- At least two of the plurality of genes may be selected from CCD7, CCD8, or P450 enzyme.
- At least three of the plurality of genes may be selected from CCD7, CCD8 and P450
- Introduction of the plurality of genes may comprise use of a gene cassette comprising at least one of the plurality of genes.
- Introduction of the plurality of genes may comprise use of a gene cassette comprising at least two of the plurality of genes.
- Introduction of the plurality of genes may comprise use of a gene cassette comprising at least three of the plurality of genes.
- Introduction of the plurality of genes may comprise use of a gene cassette comprising at least four of the plurality of genes.
- Introduction of the plurality of genes may comprise use of a gene cassette comprising at least five of the plurality of genes.
- Introduction of the plurality of genes may comprise use of a gene cassette comprising at least six of the plurality of genes.
- Introduction of the plurality of genes may comprise use of a gene cassette comprising at least seven of the plurality of genes.
- Introduction of the plurality of genes may comprise use of two or more gene cassettes. Introduction of the plurality of genes may comprise use of three or more gene cassettes. Introduction of the plurality of genes may comprise use of four or more gene cassettes. Introduction of the plurality of genes may comprise use of five or more gene cassettes. Introduction of the plurality of genes may comprise use of six or more gene cassettes. Introduction of the plurality of genes may comprise use of 7, 8, 9, 10, 11, 12, 13, 14, 15 or more gene cassettes.
- the plurality of genes may be introduced into the cell at the same time. Alternatively, the plurality of genes are introduced into the cell at two or more timepoints. The plurality of genes may be introduced into the cell at three or more timepoints. The plurality of genes may be introduced into the cell at four or more timepoints. The plurality of genes may be introduced into the cell at five or more timepoints. The plurality of genes may be introduced into the cell at six or more timepoints. The plurality of genes may be introduced into the cell at 7, 8, 9, 10, 11, 12, 13, 14, 15 or more timepoints.
- the one or more genes may comprise crtE, crtB, crtI, CCD7, CCD8, P450 enzyme or a combination thereof. At least two of the plurality of genes may comprise crtE, crtB, crtI, CCD7, CCD8, or P450 enzyme. At least three of the plurality of genes may comprise crtE, crtB, crtI, CCD7, CCD8, or P450 enzyme. At least four of the plurality of genes may comprise crtE, crtB, crtI, CCD7, CCD8, or P450 enzyme. At least five of the plurality of genes may comprise crtE, crtB, crtI, CCD7, CCD8, or P450 enzyme.
- At least six of the plurality of genes may comprise crtE, crtB, crtI, CCD7, CCD8, and P450 enzyme.
- the one or more genes may comprise crtE, crtB, crtI or a combination thereof. At least two of the plurality of genes may be selected from crtE, crtB, or crtI. At least three of the plurality of genes may be selected from crtE, crtB, and crtI.
- the one or more genes may comprise CCD7, CCD8, P450 enzyme or a combination thereof. At least one of the plurality of genes may be selected from CCD7, CCD8, or P450 enzyme. At least two of the plurality of genes may be selected from CCD7, CCD8, or P450 enzyme. At least three of the plurality of genes may be selected from CCD7, CCD8 and P450 enzyme.
- the P450 enzyme may be MAXI.
- the one or more genes may be introduced into the chromosome of the cell.
- the ono or more genes may be introduced episomally.
- the one or more genes may be introduced into the cell by transfection.
- Transfection may comprise physical treatment. Physical treatment may include, but is not limited to, electroporation, nanoparticles or magnetofection.
- Transfection may comprise chemical-based transfection. Chemical-based transfection may include, but is not limited to, cyclodextrin, polymers, liposomes, or nanoparticles. Chemical-based transfection may comprise calcium phosphate. Chemical-based transfection may comprise dendrimers. Chemical-based transfection may comprise cationic liposomes. Chemical-based transfection may comprise cationic polymers.
- Transfection may comprise non-chemical methods.
- Non-chemical methods include, but are not limited to, electroporation, sono-poration, optical transfection, protoplast fusion, impalefection, or hydrodynamic delivery.
- Transfection may comprise particle-based methods.
- Particle-based methods include, but are not limited to gene gun, mangentofection (e.g., magnet assisted transfection), or impalefection.
- Impalefection may comprise impaling cells by elongated nanostructures and arrays of such nanostructures. Examples of nanostructures include, but are not limited to, carbon nanofibers and silicon nanowires.
- Particle-based methods may also comprise particle bombardment.
- Particle bombardment may comprise delivery of the nucleic acid through membrane penetration at a high velocity.
- the nucleic acid may be connected to one or more microprojectiles.
- transfection may comprise nucleofection.
- the one or more gene may be introduced into the cell by transduction.
- Transduction may comprise viral transduction.
- Viral transduction may comprise the use of one or more viral vectors.
- the viral vector may be an adenoviral vector.
- the viral vector may be a retroviral vector.
- Transduction may comprise the use of a bacteriophage virus.
- Transformation may comprise electroporation. Transformation may comprise chemical-based transformation. Chemical-based transformation may comprise calcium phosphate. Transformation may comprise treating cells with one or more enzymes to degrade their cell walls. Transformation may comprise exposing cells to alkali cations. Alkali cations include, but are not limited to, cesium or lithium. Transformation may comprise exposing cells to lithium acetate, polyethylene glycole, or a combination thereof. Transformation may comprise enzymatic digestion. Transformation may comprise agitation or agitation with glass beads. Transformation may comprise bacterial-mediated transformation.
- Introducing the one or more genes may comprise transformation of the cell with a polynucleotide comprising the one or more genes. Introducing the one or more genes may comprise transformation of the cell with a vector comprising a polynucleotide comprising the one or more genes. The one or more genes may be introduced into the cell by a single polynucleotide or multiple polynucleotides. The one or more genes may be introduced into the cell by a single vector or multiple vectors.
- the one or more genes may be under the control of one or more promoters.
- the one or more genes may be under the control of two or more promoters.
- the two or more promoters may be the same.
- the two or more promoters may be different.
- the one or more promoters may be constitutive.
- the promoter may be regulated.
- the promoter may be a GAP promoter.
- Cells containing the one or more genes may be selected, isolated, and/or purified.
- the methods may further comprise drug selection.
- the methods may further comprise detection of a color change in cells containing the one or more genes.
- the methods may further comprise spectrophotometry.
- the methods may further comprise UV-Vis detection.
- the methods may further comprise chromatography.
- chromatography examples include, but are not limited to, column chromatography, planar chromatography, paper chromatography, thin layer chromatography, displacement chromatography, gas chromatography, liquid chromatograpy, affinity chromatography, supercritical fluid chromatography, ion exchange chromatography, size exclusion chromatography, expanded bed adsorption chromatographic separation, reversed-phase chromatography, two-dimensional chromatography, simulated moving-bed chromatography, pyrolysis gas chromatography, fast protein liquid chromatography, countercurrent chromatography, and chiral chromatography.
- Chromatography may comprise high performance liquid chromatography (HPLC). Selection may comprise the use of one or more known standards.
- the method may further comprise culturing the cells.
- the cells may be cultured for at least 6, 8, 10, 12, 16, 18, 20, 24, 26, 28, 30, 32, 36, 40, 44, 48, 56, 60, 64, 72 or more hours.
- the cells may be cultured for at least 6 hours.
- the cells may be cultured for at least 12 hours.
- the cells may be cultured for at least 24 hours.
- the cells may be cultured for at least 36 hours.
- the cells may be cultured for at least 48 hours.
- the cells may be cultured for at least 56 hours.
- the cells may be cultured for at least 64 hours.
- the cells may be cultured for at least 72 hours.
- the cells may be cultured prior to introduction of the one or more genes. Alternatively, or additionally, the cells may be cultured after introduction of the one or more genes.
- the cells may be cultured in a flask.
- the cells may be cultured in a petri dish.
- the cells may be cultured on a solid or semi-solid substrate.
- the cells may be cultured in a fermentor.
- the cells may be cultured in a cell culture media.
- the cell culture media may comprise one or more sugars and/or alcohols.
- the cell culture media may comprise one or more sugars.
- the sugars may be simple sugars.
- the sugar may be a monosaccharide.
- the monosaccharide may be linear.
- the monosaccharide may be cyclic. Examples of monosaccharids include pyranoses, furanoses, heptoses, deoxyribose, ribose, arabinose, gulose, allose, altrose, iodose, galactose, talose, mannose, lyxose, and xylose.
- the monosaccharide may be glucose.
- the sugar may be a disaccharide.
- disaccharides include, but are not limited to, sucrose, lactulose, lactose, maltose, trehalose, and cellobiose.
- the sugar may be lactose.
- the sugar may be a polysaccharide.
- polysaccharides include, but are not limited to, storage polysaccharides, starches, glycogen, structural polysaccharides, arabinoxylans, cellulose, chitin, pectin, acidic polysaccharides, and bacterial capsular polysaccharides.
- the cell culture media comprises one or more alcohols.
- alcohols include, but are not limited to, methanol, ethanol, propanol, isopropanol, and butanol.
- the alcohol may be methanol.
- the alcohol may be ethanol.
- the cells may be cultured with agitation.
- the cells may be cultured without agitation.
- the cells may be cultured at about 23° C.
- the cells may be cultured at about 25° C.
- the cells may be cultured at about 30° C.
- the cells may be cultured at about 37° C.
- the cells may be cultured at about 42° C.
- the method may further comprise purification of the plant propagation material from the cells or cell culture media.
- Purification may comprise solvent extraction.
- Solvent extraction may comprise ethyl acetate extraction.
- Purification may comprise a batchwise single stage extraction.
- Purification may comprise a multistage countercurrent continuous process.
- Purification may comprise the use of multistage countercurrent arrays.
- Purification may comprise an ion exchange mechanism.
- Purification may comprise aqueous two-phase extraction.
- Aqueous two-phase extraction may include, but is not limited to, polymer-polymer systems, polymer-salt systems, and ionic liquids.
- engineered cells comprising one or more polynucleotides and uses thereof.
- the engineered cells may be obtained by a process comprising introducing one or more polynucleotides into one or more cells.
- the engineered cells may be used to manufacture a plant propagation material.
- the one or more cells may be a prokaryotic cell.
- the one or more genes may be from a prokaryotic cell.
- the prokaryotic cell may be a bacterial cell.
- the bacterial cell may be a Gram negative cell.
- the Gram negative cell may be a Gram negative cocci.
- the Gram negative cell may be a Gram negative bacilli.
- the bacterial cell may be a Gram positive cell.
- the Gram positive cell may be a Gram positive cocci.
- the Gram positive cell may be a Gram positive bacilli.
- bacteria examples include, but are not limited to, chlamydiae, green nonsulfure bacteria, acinobacteria, planctomycetes, spirochaetes, fusobacteria, cyanobacteria, thermophilic bacteria, acidobacteria, and proteobacteria.
- the prokaryotic cell may be an archae. Examples of archae include, but are not limited to, crenarchaeota, nanoarchaeota, and euryarchaeota.
- the one or more cells may be an eukaryotic cell.
- the one or more genes may be from an eukaryotic cell.
- eukaryotes include, but are not limited to, fungi, animals, slime moulds, plants, algae, and protzoa.
- the eukaryotic cell may be a fungal cell. Examples of fungi include, but are not limited to, glomeromycota, chytridomycota, zygomycota, ascomycota, basidiomycota, and deuteromycetes.
- the eukaryotic cell may be a yeast cell.
- the yeast cell may be Saccharomyces, Cryptococcus, Candida .
- the yeast cell may be Pichia .
- the yeast cell may be P. pastoris .
- the yeast cell may be Pantoea .
- the yeast cell may be P. ananatis .
- the yeast cell may be Saccharomyces .
- the yeast cell may be
- the eukaryote may be an animal.
- the animal may be a mammal. Examples of mammals include, but are not limited to, humans, goats, monkeys, dogs, sheep, cows, cats, rodents, rabbits and lions.
- the animal may be an avian. Avians include, but are not limited to, eagles, hawks, chickens, and penguins.
- the animal may be a reptile. Examples of reptiles include, but are not limited to, lizards, alligators, crocodiles, turtles, snakes and tortoises.
- the animal may be a fish. Fish include, but are not limited to, trout, sharks, whales, dolphins and bass.
- the cells from which the genes are from and the cells in which the genes are introduced may be of the same species.
- the cells from which the genes are from and the cells in which the genes are introduced may be of different species.
- the cells from which the genes are from and the cells in which the genes are introduced may be of different cell types.
- the plant propagation materials and formulations disclosed herein may be used in agriculture.
- the plant propagation materials and formulations may be used to promote plant growth.
- the plant propagation materials and formulations disclosed herein may be used for enhancing shoot stability in plants.
- the plant propagation materials and formulations may be used for increasing transport capacity in plants.
- the plant propagation materials and formulations may be used for increasing drought tolerance of a plant.
- Improving agriculture may comprise promoting plant growth.
- Improving agriculture may comprise enhancing shoot stability in plants.
- Improving agriculture may comprise increasing transport capacity in plants.
- Improving agriculture may comprise increasing drought tolerance.
- Improving agriculture may comprise reducing an application of one or more pesticides.
- Improving agriculture may comprise terminating application of one or more pesticides.
- Improving agriculture may comprise reducing watering amounts applied to the plants.
- Improving agriculture may comprise reducing watering frequency to the plants.
- Improving agriculture may comprise controlling phytopathogenic fungi.
- Improving agriculture may comprise controlling unwanted plant growth.
- Improving agriculture may comprise controlling unwanted insect or mite infestation.
- Improving agriculture may comprise regulating growth of the plant.
- Improving agriculture may comprise promoting or stimulating activity in one or more fungi.
- the methods may comprise use of a formulation comprising a propagation plant propagation material disclosed herein to act on the respective pests, their habitat or the plants to be protected from the respective pest, to the soil and/or to unwanted plants and/or the crop plants and/or their habitat.
- the plant propagation materials may increase plant growth by at least about 5%.
- the plant propagation materials may increase plant growth by at least about 10%.
- the plant propagation materials may increase plant growth by at least about 15%.
- the plant propagation materials may increase plant growth by at least about 20%.
- the plant propagation materials may increase plant growth by at least about 25%.
- the plant propagation materials may increase plant growth by at least about 30%.
- the plant propagation materials may increase plant growth by at least about 50%.
- the plant propagation materials may increase plant growth by at least about 60%, 70%, 80%, 90%, 95%. 100% or more.
- the plant propagation materials may increase plant growth by at least about 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 40, 50-fold or more.
- the plant propagation materials may increase plant growth by at least about 1.5-fold or more.
- the plant propagation materials may increase plant growth by at least about 2-fold or more.
- the plant propagation materials may increase plant growth by at least about 3-fold or more.
- the plant propagation materials may increase plant growth by at least about 5-fold or more.
- the plant propagation materials may increase plant growth by at least about 10-fold or more.
- Plant growth may comprise secondary plant growth.
- the plant propagation materials may enhance shoot growth by at least about 5%.
- the plant propagation materials may enhance shoot growth by at least about 10%.
- the plant propagation materials may enhance shoot growth by at least about 15%.
- the plant propagation materials may enhance shoot growth by at least about 20%.
- the plant propagation materials may enhance shoot growth by at least about 25%.
- the plant propagation materials may enhance shoot growth by at least about 30%.
- the plant propagation materials may enhance shoot growth by at least about 50%.
- the plant propagation materials may enhance shoot growth by at least about 60%, 70%, 80%, 90%, 95%. 100% or more.
- the plant propagation materials may enhance shoot growth by at least about 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 40, 50-fold or more.
- the plant propagation materials may enhance shoot growth by at least about 1.5-fold or more.
- the plant propagation materials may enhance shoot growth by at least about 2-fold or more.
- the plant propagation materials may enhance shoot growth by at least about 3-fold or more.
- the plant propagation materials may enhance shoot growth by at least about 5-fold or more.
- the plant propagation materials may enhance shoot growth by at least about 10-fold or more.
- the plant propagation materials may increase transport capacity in plants by at least about 5%.
- the plant propagation materials may increase transport capacity in plants by at least about 10%.
- the plant propagation materials may increase transport capacity in plants by at least about 15%.
- the plant propagation materials may increase transport capacity in plants by at least about 20%.
- the plant propagation materials may increase transport capacity in plants by at least about 25%.
- the plant propagation materials may increase transport capacity in plants by at least about 30%.
- the plant propagation materials may increase transport capacity in plants by at least about 50%.
- the plant propagation materials may increase transport capacity in plants by at least about 60%, 70%, 80%, 90%, 95%. 100% or more.
- the plant propagation materials may increase transport capacity in plants by at least about 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 40, 50-fold or more.
- the plant propagation materials may increase transport capacity in plants by at least about 1.5-fold or more.
- the plant propagation materials may increase transport capacity in plants by at least about 2-fold or more.
- the plant propagation materials may increase transport capacity in plants by at least about 3-fold or more.
- the plant propagation materials may increase transport capacity in plants by at least about 5-fold or more.
- the plant propagation materials may increase transport capacity in plants by at least about 10-fold or more.
- the plant propagation materials may increase drought tolerance in plants by at least about 5%.
- the plant propagation materials may increase drought tolerance in plants by at least about 10%.
- the plant propagation materials may increase drought tolerance in plants by at least about 15%.
- the plant propagation materials may increase drought tolerance in plants by at least about 20%.
- the plant propagation materials may increase drought tolerance in plants by at least about 25%.
- the plant propagation materials may increase drought tolerance in plants by at least about 30%.
- the plant propagation materials may increase drought tolerance in plants by at least about 50%.
- the plant propagation materials may increase drought tolerance in plants by at least about 60%, 70%, 80%, 90%, 95%. 100% or more.
- the plant propagation materials may increase drought tolerance in plants by at least about 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 40, 50-fold or more.
- the plant propagation materials may increase drought tolerance in plants by at least about 1.5-fold or more.
- the plant propagation materials may increase drought tolerance in plants by at least about 2-fold or more.
- the plant propagation materials may increase drought tolerance in plants by at least about 3-fold or more.
- the plant propagation materials may increase drought tolerance in plants by at least about 5-fold or more.
- the plant propagation materials may increase drought tolerance in plants by at least about 10-fold or more.
- the plant propagation materials may reduce the application of one or more pesticides.
- Reducing the application of one or more pesticides may comprise reducing an amount of the one or more pesticides that are applied to the plant.
- the amount of the one or more pesticides applied to the plant may be reduced by at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%.
- the amount of the one or more pesticides applied to the plant may be reduced by at least about 10%.
- the amount of the one or more pesticides applied to the plant may be reduced by at least about 20%.
- the amount of the one or more pesticides applied to the plant may be reduced by at least about 30%.
- the amount of the one or more pesticides applied to the plant may be reduced by at least about 50%.
- reducing the application of the one or more pesticides may comprise reducing a frequency of which the one or more pesticides are applied to the plant.
- the frequency of which the one or more pesticides are applied to the plant may be reduced by at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%.
- the frequency of which the one or more pesticides are applied to the plant may be reduced by at least about 10%.
- the frequency of which the one or more pesticides are applied to the plant may be reduced by at least about 20%.
- the frequency of which the one or more pesticides are applied to the plant may be reduced by at least about 30%.
- the frequency of which the one or more pesticides are applied to the plant may be reduced by at least about 40%.
- the frequency of which the one or more pesticides are applied to the plant may be reduced by at least about 50%.
- Use of the plant propagation materials may allow a reduction in the amount of water applied to the plants.
- the amount of the water applied to the plant may be reduced by at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%.
- the amount of the water applied to the plant may be reduced by at least about 10%.
- the amount of the water applied to the plant may be reduced by at least about 20%.
- the amount of the water applied to the plant may be reduced by at least about 30%.
- the amount of the water applied to the plant may be reduced by at least about 50%.
- Use of the plant propagation materials may allow a reduction in the frequency of which the water is applied to the plant.
- the frequency of which the water is applied to the plant may be reduced by at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%.
- the frequency of which the water is applied to the plant may be reduced by at least about 10%.
- the frequency of which the water is applied to the plant may be reduced by at least about 20%.
- the frequency of which the water is applied to the plant may be reduced by at least about 30%.
- the frequency of which the water is applied to the plant may be reduced by at least about 40%.
- the frequency of which the water is applied to the plant may be reduced by at least about 50%.
- the plant propagation material disclosed herein may be used to control phytopathogenic fungi. Improving agriculture may comprise controlling unwanted plant growth. Controlling unwanted plant growth may comprise stimulating germination activity of the unwanted plant.
- the unwanted plant may be a parasitic plant.
- the unwanted plant may be a root parasitic plant. Examples of parasitic plants include, but are not limited to, witchweeds ( Striga spp.), broomrapes ( Orobanche spp, Phelipanche spp), Alectra , dodders, and mistletoes.
- the unwanted plant may belong to the family Orobanchaceae.
- the unwanted plant may be witchweed.
- the unwanted plant may be Orobanche spp.
- the plant propagation material may be applied directly to the unwanted plant.
- the plant propagation material may be applied indirectly to the unwanted plant.
- insects and mites include, but are not limited to spiders, gnats, mealybugs, whiteflies, predator mites, spider mites and aphids.
- the plant propagation material disclosed herein may be used to regulate growth of the plant.
- Regulating plant growth may comprise regulating plant breeding
- Regulating plant growth may comprise inhibiting shoot branching.
- Regulating plant growth may comprise regulating one or more plant products.
- Regulating plant growth may comprise inhibiting root development.
- the plant propagation material disclosed herein may be used to promote or stimulate activity in fungi.
- the plant propagation material may stimulate hyphal branching activity of one or more fungi.
- the plant propagation material may induce spore germination of one or more fungi.
- the one or more fungi may be arbuscular mycorrhizal (AM) fungi.
- the method may comprise contacting the plant with a plant propagation material disclosed herein.
- the plant propagation material may comprise a compound of Formula (I) or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula (II) or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a compound of Formula (III) or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a strigolactone or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material may comprise a strigolactone mimic or a salt, solvate, polymorph, stereoisomer, or isomer thereof.
- the plant propagation material for use in preserving or extending the life of a plant may be produced by any of the methods disclosed herein.
- the plant propagation material may be produced by chemical synthesis.
- the plant propagation material is produced by conducting a condensation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the plant propagation material may be produced by conducting a hydroxymethylation on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof.
- the plant propagation material may be produced by (a) conducting a hydroxymethylation on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof to produce a first product; and (b) conducting an alkylation reaction on the first product.
- the plant propagation material is produced by biological synthesis.
- Biological synthesis may comprise the use of one or more cells, genes, or vectors disclosed herein.
- the plant propagation material may be used to preserve or extend the life of a cut plant.
- the cut plant may be a flower.
- the cut plant may be a tree.
- the cut plant may be bush or shrub.
- the cut plant may be a vegetable.
- the plant propagation material may be used to preserve or extend the life of an uncut plant.
- the uncut plant may be a flower.
- the uncut plant may be a tree.
- the uncut plant may be bush or shrub.
- the uncut plant may be a vegetable.
- the plant propagation material may be used to preserve or extend the life of a potted plant.
- the potted plant may be a flower.
- the potted plant may be a tree.
- the potted plant may be bush or shrub.
- the potted plant may be a vegetable.
- the plant propagation material may be used to preserve or extend the life of a flower.
- flowers include, but are not limited to, lilies, daisies, roses, marigolds, Angel's trumpet, phlox, vinca, snapdragons, toadflax, orchids, ferns, black-eyed Susans, blood flowers, blue lobelias, morning glories, poppies, calendulas, geraniums, impatiens, lantanas, larkspurs, calla lilies, hyacinths, azaleas, pointsettias, and begonias.
- the plant propagation material may be used to preserve or extend the life of a bush or shrub.
- bushes and shrubs include, but are not limited to, forsynthia, fuchsia, hibiscus, currant, lilac, rose, hydrangea, willow, magnolia, thyme, snowberry, dogwood and holly.
- the plant propagation material may be used to preserve or extend the life of a tree.
- trees include, but are not limited to, cypress, poinsettia, palm, fir, pine, spruce, cedar, oak, mulberry, chestnut, hawthorn, poplar, and maple.
- the tree may be a fir tree.
- the fir tree may be a Douglas, Balsam or Fraser fir tree.
- the tree may be a pine tree.
- the pine tree may be a Scotch or White pine tree.
- the tree may be a spruce tree.
- the spruce tree may be a White, Norway or Blue spruce tree.
- the tree may be a cedar tree.
- the cedar tree may be a Deodara or Eastern red cedar.
- the tree may be a cypress tree.
- the cypress tree may be an Arizona or Leland cypress tree.
- the plant may be contacted with a plant propagation material disclosed herein, thereby extending or preserving the life of the plant.
- Contacting the plant with the plant propagation material may comprise administering the plant propagation material as a spray.
- Contacting the plant with the plant propagation material may comprise adding the plant growth material to the irrigation water of the plant.
- Contacting the plant with the plant propagation material may comprise applying the plant propagation material to the habitat of the plant.
- Contacting the plant with the plant propagation material may comprise adding the plant propagation material to a plant container (e.g., vase) and placing the plant in the plant container.
- Contacting the plant with the plant propagation material may comprise adding the plant propagation material to soil.
- the life of the plant may be extended by at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 97% as compared to an untreated plant.
- the life of the plant may be extended by at least about 20% as compared to an untreated plant.
- the life of the plant may be extended by at least about 30% as compared to an untreated plant.
- the life of the plant may be extended by at least about 40% as compared to an untreated plant.
- the life of the plant may be extended by at least about 50% as compared to an untreated plant.
- the life of the plant may be extended by at least about 55% as compared to an untreated plant.
- the life of the plant may be extended by at least about 60% as compared to an untreated plant.
- the life of the plant may be extended by at least about 65% as compared to an untreated plant.
- the life of the plant may be extended by at least about 70% as compared to an untreated plant.
- the life of the plant may be extended by at least about 75% as compared to an untreated plant.
- the life of the plant may be extended by at least about 80% as compared to an untreated plant.
- the life of the plant may be extended by at least about 6, 12, 24, 30, 36, 42, 48, 54, 60, 66, 72, 78, 84, 90, 96, 102, 108, 114, or 120 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 24 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 36 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 48 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 72 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 96 hours as compared to an untreated plant.
- the life of the plant may be extended by at least about 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, or 7 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 1 day as compared to an untreated plant.
- the life of the plant may be extended by at least about 2 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 2.5 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 3 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 3.5 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 4 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 4.5 days as compared to an untreated plant.
- the life of the plant may be extended by at least about 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, or 7 weeks as compared to an untreated plant.
- the life of the plant may be extended by at least about 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 weeks as compared to an untreated plant.
- the life of the plant may be extended by at least about 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, or 7 months as compared to an untreated plant.
- the life of the plant may be extended by at least about 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 months as compared to an untreated plant.
- Preserving or extending the life of the plant may comprise reducing wilting of the plant.
- Reducing wilting of the plant may comprise reducing flower or leaf rolling of the plant.
- the wilting of the plant may be reduced by at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 97% as compared to an untreated plant.
- the wilting of the plant may be reduced by at least about 10% as compared to an untreated plant.
- the wilting of the plant may be reduced by at least about 30% as compared to an untreated plant.
- the wilting of the plant may be reduced by at least about 50% as compared to an untreated plant.
- the wilting of the plant may be reduced by at least about 70% as compared to an untreated plant.
- the wilting of the plant may be reduced by at least about 80% as compared to an untreated plant.
- a sign of plant stress may include wilting of the plant.
- stressed plants may have rolled leaves or petals.
- the plant growth materials disclosed herein may promote the life of the plant by reducing the wilting of the plant.
- Reducing the wilting of the plant may comprise delaying the wilting of the plant as compared to an untreated plant.
- an untreated cut plant may show signs of wilting within 36 hours of being cut, however, a cut plant treated with a plant growth material may have delayed wilting.
- the wilting of the plant may be delayed by at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours as compared to an untreated plant.
- the wilting of the plant may be delayed by at least about 12 hours as compared to an untreated plant.
- the wilting of the plant may be delayed by at least about 24 hours as compared to an untreated plant.
- the wilting of the plant may be delayed by at least about 36 hours as compared to an untreated plant.
- the wilting of the plant may be delayed by at least about 48 hours as compared to an untreated plant.
- Turgidity may refer to pressure caused by the osmotic flow of water from an area of low solute concentration outside of the cell into the cell cell's vacuole. Turgidity may be used by plants to maintain rigidity. Often, healthy plants are turgid, whereas, unhealthy plants are less turgid. Preserving or extending the life of the plant may comprise prolonging or maintaining the turgidity of the plant. The turgidity of the plant may be greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 97% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 10% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 15% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 25% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 35% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 45% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 60% greater than the turgidity of an untreated plant.
- the turgidity of the plant may be at least about 75% greater than the turgidity of an untreated plant.
- a stressed plant may also show a reduction in the turgid state.
- the turgid state may refer to a period of time in which the plant maintains its rigidity.
- the rigidity of the plant may refer to the rigidity of the stem of the plant. For example, as cut plants die, the stem of the plant may be less rigid, thereby causing the cut plant to fall over or bend.
- a stressed plant may be unable to hold itself upright. Preserving or extending the life of the plant may comprise prolonging the turgid state of the plant.
- the turgid state of the plant may be increased by at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 97% as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 20% as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 30% as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 40% as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 50% as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 6 hours as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 12 hours as compared to an untreated plant.
- the turgid state of the plant may be increased by at least about 24 hours as compared to an untreated plant.
- a stressed plant may lose leaves or petals. Contacting a plant with a plant growth material may reduce or delay the loss of one or more petals or leaves of the plant. For example, an untreated plant may lose 50% of its leaves or petals, whereas a treated plant may lose 10-25% of its leaves or petals. The loss of the one or more petals of the plant may be reduced by least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 97% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be reduced by least about 10% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be reduced by least about 20% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be reduced by least about 35% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be reduced by least about 50% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be reduced by least about 60% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be reduced by least about 70% as compared to the loss of the one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95 or 100 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 6 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 12 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 18 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 36 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 48 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 60 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 72 hours as compared to the loss of one or more petals of an untreated plant.
- the loss of the one or more petals of the plant may be delayed by at least about 96 hours as compared to the loss of one or more petals of an untreated plant.
- a stressed plant may show signs of discoloration.
- the stressed plant may appear brownish.
- the stressed plant shows a reduction in the appearance of green leaves.
- the chlorohyll content of the stressed plant may also be reduced.
- Preserving or extending the life of the plant may comprise maintaining the chlorophyll content of the plant.
- a reduction in the chlorophyll content of an untreated plant may appear within 48 hours of being cut.
- a reduction in the chlorophyll content of a treated plant may appear after 60 hours of being cut.
- the chlorophyll content of the plant may be maintained for at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours.
- the chlorophyll content of the plant may be maintained for at least about 6 hours.
- the chlorophyll content of the plant may be maintained for at least about 12 hours.
- the chlorophyll content of the plant may be maintained for at least about 24 hours.
- Preserving or extending the life of the plant may comprise reducing or delaying the loss of the chlorophyll content of the plant.
- the chlorophyll content of the plant may be greater than the chlorophyll content of an untreated plant.
- the chlorophyll content of the plant may be at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, or 50% greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95% or 97% greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 20% greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 30% greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 40% greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 50% greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 60% greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, 7, 8, 9, or 10-fold greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95 or 100-fold greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 2-fold greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 3-fold greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 4-fold greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 5-fold greater than the content of an untreated plant.
- the chlorophyll content of the plant may be at least about 10-fold greater than the content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, or 100 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 6 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 12 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 24 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 36 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 48 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 60 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be delayed by at least about 72 hours as compared to the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, or 60% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 65%, 70%, 72%, 75%, 77%, 80%, 85%, 90%, 92%, 95%, or 97% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 5% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 10% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 20% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 30% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 40% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 50% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 60% less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, 7, 7.5, 8, 8.5, 9, 9.5, or 10-fold less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95 or 100-fold less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 2-fold less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 3-fold less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 5-fold less than the loss of the chlorophyll content of an untreated plant.
- the loss of the chlorophyll content of the plant may be at least about 10-fold less than the loss of the chlorophyll content of an untreated plant.
- the plant propagation material or a formulation thereof may be applied directly to the plant.
- the plant propagation material or a formulation thereof may be applied to one or more parts of the plant.
- the one or more parts of the plant may comprise a terminal bud, flower, lateral bud, leaf blade, leaf axil, node, internode, petiole, primary root, lateral root, root hair, root cap, or a combination thereof.
- the formulations may be applied to the leaf blade of the plant.
- the formulations may be applied to the root of the plant.
- the plant propagation material or a formulation thereof is applied indirectly to the plant.
- the formulation may be applied to an area around the plant.
- the area around the plant may comprise soil.
- the area around the plant may comprise an adjacent plant.
- the plant propagation material or a formulation thereof may be applied to a plant that is susceptible to a parasitic weed.
- plants include, but are not limited to, corn, rice, sorghum, millets, and sugar cane.
- the plant may be corn.
- the plant may be tobacco.
- the plant may be rice.
- the plant propagation material or a formulation thereof may be applied as a seed coating.
- the plant propagation material or a formulation thereof may be applied as a seed treatment.
- the plant propagation material or a formulation thereof may be applied as a seed dressing.
- the plant propagation material or a formulation thereof may be applied as a spray.
- the plant propagation material or a formulation thereof may be applies as a foliar spray.
- the plant propagation material or a formulation thereof may be applied as a powder.
- the plant propagation material or a formulation thereof may be applied 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more times a day.
- the plant propagation material or a formulation thereof may be applied once a day.
- the plant propagation material or a formulation thereof may be applied twice a day.
- the plant propagation material or a formulation thereof may be applied 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more times per week.
- the plant propagation material or a formulation thereof may be applied once a week.
- the plant propagation material or a formulation thereof may be applied twice a week.
- the plant propagation material or a formulation thereof may be applied three times a week.
- the plant propagation material or a formulation thereof may be applied four times a week.
- the formulations may be applied 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more times a month.
- the formulations may be applied once a month.
- the plant propagation material or a formulation thereof may be applied twice a month.
- the plant propagation material or a formulation thereof may be applied three times a month.
- the plant propagation material or a formulation thereof may be applied four times a month.
- the formulations may be applied ten times a month.
- the plant propagation material or a formulation thereof may be applied 15 times a month.
- the formulations may be applied 20 times a month.
- This example evaluates the ability of SL to mitigate the negative effects of water stress in maize.
- Several standard metrics are used to determine efficacy of SL treatment, including onset of water stress symptoms (leaf rolling, reduction in chlorophyll) and grain yield. Using these metrics to evaluate efficacy in enhancing drought tolerance, we are evaluating (1) the best methods for application of SL, (2) the dose concentration of SL, and (3) the dose schedule of SL, thereby determining the stage of plant growth where application has the largest effect. Taken together, this data enables us to estimate the magnitude improvement in crop health and harvest yield to expect in future phases of the project.
- the overall efficacy of SL treatment on harvest yield (per plant and bushels per acre) is key in determining the value proposition of technology adoption for growers.
- This example focuses on formulation, initial safety testing (both toxicology and environmental fate), and field efficacy of SL under different conditions. Data from Example 2 on the application method and dose with the highest efficacy guides product formulation efforts. Formulation of the SL active ingredient with inert carriers is tested for effective delivery to maize fields. For either a foliar spray or an irrigation supplement, it is likely that a wettable powder is the preferred formulation. Wettable powders contain relatively low amounts of the active ingredient along with inert carriers such as surfactants to allow even spraying. Formulations are tested for active ingredient release profiles using analytical chemistry (GC-MS), while formulation efficacy is measured in greenhouse maize and small-scale field experiments.
- GC-MS analytical chemistry
- Initial safety testing are focused on generating mammalian toxicology data and environmental fate data to support registration of SL as a new active ingredient with the Environmental Protection Agency. This work is performed using certified contract research organizations, and can also be used to request an Experimental Use Permit from the EPA for use of SL in field trials larger than 10 acres.
- the field trials with formulated SL are also performed, wherein the effects of SL treatment on harvest yield with different maize hybrid varieties, soil conditions, weather conditions, and drought severity are examined. The positive impacts of SL as a product for enhancing drought tolerance and improving harvest yield is determined.
- this example tests whether strigolactone has a positive effect on water stressed corn plants.
- This experiment examines whether SL application affects growth and/or morphology in healthy plants experiencing adequate irrigation.
- the rationale for this experiment is two-fold: first, due to the historical high costs of SL and mimic synthesis, it is unknown what effect the exogenous application of SL will have on vegetative growth. While there is no prior evidence to suggest deleterious effects, a crop protection product that negatively modulates vegetative growth would have a difficult path to adoption. Second, observation of the growth rates of morphology of SL-treated, unstressed plants provides a baseline for downstream experiments.
- This experiment tests and quantifies the drought protective effect of SL treatment in terms of the appearance of drought stress symptoms in plants in the vegetative stage of growth.
- Water stress in growing maize is manifest as leaf rolling (wilting), reductions in chlorophyll and photosynthetic activity, and plant height in severe cases.
- the experiments involve growing plants under adequate irrigation, applying a range of SL concentrations or a mock treatment, and stopping irrigation to impose water limitation.
- We perform these experiments to provide the range of SL concentrations that elicit a drought protective response, the duration of the drought protective response, and the magnitude of the drought protective response. This data may be used in determining the impact of SL application on grain yield in water stressed and unstressed corn and in validating SL as a drought protective product.
- This experiment seeks to determine directly whether SL application affects harvest grain yield, both for plants experiencing adequate irrigation and for plants subjected to water stress. These experiments are performed in a greenhouse for maximum control over conditions. We impose a severe water stress during the early reproductive phase of the maize growth cycle, starting at tasseling and continuing to early grain fill. From these results, we determine the best application method, dose, and application timing to enhance grain yields. This data also provides a foundation for scale up and field trials of SL as a drought protective product.
- SL SL to provide a protective effect to corn under drought stress.
- One key metric to determine economic and technical feasibility is the grain yield of drought stressed plants treated with SL. Maize are grown under greenhouse conditions for precise control of irrigation and conditions.
- water stress during the period from two weeks prior to silking to two weeks after silking can reduce harvest yield by 3 to 8 percent for each day of stress [2].
- Silking occurs 4 to 8 days after tasseling, with pollination 1 to 3 days after the emergence of silk.
- Subjecting plants to water limitation stress at this time results in significantly lower kernel number as well as kernel weight, due to desiccation of the silks and poor pollination efficiency.
- Foliar sprays are widely used to apply fertilizers, pesticides, and micronutrients to maize and other crops. Foliar sprays are particularly advantageous during drought conditions where soil moisture does not support efficient uptake of nutrients. Typical foliar spray application volumes use 3-10 gallons of liquid per acre for micronutrients, which we will follow here.
- Typical foliar spray application volumes use 3-10 gallons of liquid per acre for micronutrients, which we will follow here.
- This experiment incorporates the knowledge gained from previous experiments to test the ‘best practice’ application method with a higher number of experimental replicates in order to generate highly statistically significant data.
- the data from this experiment test the utility of SL as a drought-protective product and provide the foundation for field trials in the next phase of this project.
- the experiments evaluate application timing, application method, and two application concentrations on irrigated and water-stressed corn.
- kernel morphology and grain yield at maturity Severely water-stressed plants are known to have both unfertilized kernels and aborted kernels.
- AB01 The synthesis of AB01 starts from a readily available sesquiterpene lactone, sclareolide.
- Sclareolide is extracted from species of the Salvia plant and is currently used in industrial production of perfumes.
- the synthesis of AB01 is a straightforward hydroxymethylation and alkylation in a one-pot procedure.
- Sclareolide is condensed with a three-fold excess of methyl formate in the presence of potassium tert-butoxide and the solution of hydroxymethylene lactone is then alkylated with bromobutenolide to give a mixture of two diastereomers. Resolution of stereomers is not necessary for downstream application.
- reaction mixture was filtered to remove succinimide to provide a clear orange solution.
- Mixture was concentrated to provide an orange oil that was partitioned between toluene (150 mL) and de-ionized water (150 mL). Rapid stirring of the yellow emulsion for 30 minutes, then the toluene layer was separated, dried with Na2SO4, filtered and concentrated to give a deep orange oil.
- 3-methyl-2(5H)-furanone is commercially available (Aldrich, 90% technical grade) and by synthesis via bromination/elimination of 3-methyl-2,3-dihydro-2(3H)-furanone ( ⁇ -methyl- ⁇ -butyrolactone)1, from citraconic anhydride via a retro Diels-Alder sequence2 or by regiocontrolled ring opening and reduction3 and finally by ring-closing methathesis of allyl methyacrylate4.
- commercial material contains polar impurities that are not readily removed by column chromatography or distillation at the cost of considerable loss of material. While these polar impurities severely retard the rate of the radical bromination3, they are removed from the desired bromobutenolide as detailed above. Material without distillation performs well in the alkylation and it is stable over periods of months when stored cold protected from light.
- the flask was charged with sclareolide (0.4126 g, 2.0 mmol, Sigma-Aldrich) and dissolved in dry THF (20 mL).
- the clear, colourless solution was cooled under inert gas to ⁇ 0° C. using an ice-water bath.
- the pale yellow-white suspension was stirred under nitrogen at ⁇ 0° C. for 3 hours.
- Bromobutenolide (0.3540 g, 2.0 mmol, 1 equivalent) was added as a solution in dry THF (6 mL) via syringe.
- the reaction suspension was left to stir overnight, warming to room temperature.
- the suspension was quenched with distilled water (50 mL) and diluted with EtOAc (100 mL).
- This example determines the effect of the application of a plant propagation material comprising strigolactone or analogs or mimics thereof on the onset of water-stress symptoms in cut flowers during storage, shipping, and/or life in retail or consumer environment.
- concentrations to focus our investigations of the effects of the plant propagation material on cut flowers.
- Indicators of water-limitation stress in flowers are measured (approximately daily).
- the concentration of the strigolactone or analog or mimic thereof in the plant propagation material spanned a concentration range from 1 gram per liter to 1 picogram per liter.
- FIG. 3 shows the results from a preliminary experiment in which cut floweres were treated with a water-only control or a plant propagation material comprising a strigolactone mimic (e.g., AB01). Various concentrations of the strigolactone mimic were added to the irrigation water of the cut flowers.
- the vase life for cut flowers treated with water-only was approximately 4 days, whereas the vase life for the cut flowers treated with the plant propagation material was approximately 6-7 days.
- the purpose of this example is to also determine the best practice of cut flower treatment and to inform commercial considerations.
- Application through irrigation is commonly used for cut flower preservation, including granule or liquid-based formulations that can be added to irrigation water.
- a dose of the plant propagation material comprising strigolactone or analog or mimic thereof is dissolved in a small volume (0.1 mL) of acetone, diluted into the unit irrigation volume of water, and applied in the vase of cut flowers.
- a mock application of diluted acetone is performed.
- Plants receiving the spray application are physically separated from control plants using temporary plastic barriers.
- Each formulation is tested in combination with other cut flower preservative and shelf life extension products, such as nutrients, antimicrobials, surfactants, and plant growth regulators.
- a chemical mimic of the plant hormone strigolactone has been developed displaying biological activity similar to the natural product and potencies several orders of magnitude greater than those of previously described mimics.
- the synthesis of AB01 starts from a readily available sesquiterpene, lactone, sclareolide.
- Sclareolide is extracted from species of the Salvia plant and is currently used in industrial production of perfumes.
- Sclareolide is condensed with a two-fold excess of methyl formate in the presence of lithium diisopropylamide.
- the isolated formyl lactone is then alkylated with chlorobutenolide to give a mixture of two diastereomers.
- a concise synthesis of chlorobutenolide is provided here. Resolution of stereomers is not necessary for downstream application.
- an oven-dried 100 mL 2-necked round bottom flask (2 ⁇ 14j) with stirbar, capped with a rubber septa and nitrogen bubbler was cooled under nitrogen flow.
- the flask was charged with sclareolide (1.50 g, 6.0 mmol, Sigma-Aldrich) and dissolved in dry THF (42 mL).
- the clear, colourless solution was cooled under inert gas to ⁇ 0° C. using an ice water bath.
- LDA solution (3.60 mL, 7.20 mmol, 1.2 equivalents, 2.0M solution Sigma-Aldrich) was added dropwise via syringe to give a yellow-orange solution. Stirred at ⁇ 78° C.
- Step 1 TiCl4 aldol ( FIG. 10A )
- a 1000 mL 3-necked (19j, 34j, 19j) round bottom flask was equipped with an oversized stirbar, nitrogen bubbler (19j), reducing adapter (19j to 34j) topped with a pressure equalizing dropping funnel capped with 19j rubber septa (34j) and rubber septa (19j).
- the assembled glassware was flushed under nitrogen and flame-dried under nitrogen purge.
- CH 2 Cl 2 was charged to the flask (212 mL, anhydrous) and dropping funnel (106 mL).
- TiCl 4 (16.5 mL, 150 mmol) was added to the flask to give a clear, colourless solution.
- the titanium tetrachloride solution was cooled in an ice water bath and the dropping funnel charged with ethyl pyruvate (16.7 mL, 150 mmol) and vinyl acetate (13.8 mL, 150 mmol).
- the carbonyl solution in CH 2 Cl 2 was added dropwise to the titanium tetrachloride solution over two hours, generating a bright yellow-orange suspension.
- the suspension was further stirred for two hours at 0° C. (ice water bath).
- the clear orange-red solution was quenched with deionized water (140 mL) (caution: exothermic with vigorous gas production).
- CH 2 Cl 2 separated and the aqueous layer extracted with CH 2 Cl 2 (2 ⁇ 100 mL).
- Step 2 Hydrolysis and cyclization ( FIG. 10B )
- the condenser was returned to reflux set up and the deep golden reaction mixture heated at reflux for 45 minutes.
- the cooled reaction mixture was extracted with ethyl acetate (3 ⁇ 150 mL). Combined extracts were washed with brine (1 ⁇ 100 mL) and dried Na 2 SO 4 . Filtered to give a golden solution, concentrated to give an orange oil (15.19 g).
- the crude orange oil was subjected to bulb-to-bulb distillation, collecting material at 120-135° C./8 mbar.
- the pale yellow oil (9.47 g, 65%) slowly solidified on standing.
- Step 3 Chlorination ( FIG. 10C )
- a 25 mL 19j rbf was capped with a take-off head (2 necked, 2 ⁇ 19j), capped with a 19 j glass stopper and 19j reflux condenser.
- the flask was charged with CH 2 Cl 2 (5 mL), SOCl 2 (1 mL, 14 mmol, 1.4 equiv) and a drop of DMF, then heated to reflux.
- a golden solution of hydroxybutenolide (1.15 g, 10 mmol) in CH 2 Cl 2 (5 mL) was added dropwise to the refluxing vapours at such a rate to maintain reflux with immediate gas evolution.
- a 100 mL round bottom flask containing the formyl sclareolide (1.57 g, 5.64 mmol) was flushed under nitrogen and dissolved in DMF (15 mL, anhydrous, Sigma-Aldrich) at room temperature.
- the clear yellow solution was treated with potassium carbonate (858 mg, 6.2 mmol, 1.1 equivalents) under nitrogen flow to give a yellow-white suspension.
- To the suspension was added dropwise via syringe a clear golden solution of chlorobutenolide (5.52 mmol, 1.2 equivalents) in DMF (5 mL, anhydrous). Addition of the chlorobutenolide solution causes a colour change of the reaction mixture from yellow to orange to brown.
- the nitrogen-substituted sclareolides are synthesised using an approach similar to FIG. 7 .
- This complex of 2-benzyloxy-20-hydroxy-1,10-binapthyl-SnCl4 prepared in situ is cooled to ⁇ 78° C.
- Alfalfa was grown from seeds for 14 days. Seedlings were treated with 1 mg/mL AB01 or mock treated. Irrigation was stopped after treatment and seedlings were monitored for symptoms of water limitation stress. At approximately 7 days post-treatment, the untreated seedlings appeared dessicated (left), while treated seedlings appeared robust (right).
- the fraction of fertilized ears was measured by visual inspection of the ear silk at the R2-R3 stage. Unfertilized silks that appear yellow/green, while fertilized silks appear reddish brown. Completely unfertilized ears have silks that are completely yellow/green, fertilized ears appear brown, while ears with an incomplete degree of fertilized silks have a mixed population of silks. We found that AB01 treatment decreased the fraction of unfertilized ears while increasing the fraction of fertilized ears, compared to the mock treated control.
- Ears were visually inspected at harvest to monitor kernel set (the number of fully formed kernels in the mature ear). Control (mock treated, left) ears showed incomplete kernel set and kernel abortion, symptoms of severe drought stress. AB01 treated ears (right) showed more complete kernel set and few signs of kernel abortion.
- Ears volume was measured in the field at the R3 stage to calculate ear volume. The length and circumference of each ear was measure, and volume was calculated by treating the ear as a cylinder. Approximately 100 ears from each plot were measured. AB01 treated ears averaged significantly higher volumes in mid-season as compared to the mock treated control.
- Control plots averaged 120 bushels per acre, while AB01 treated plots averaged 143 bushels per acre, a 19% increase. Grain yield is calculated at 15% moisture, a standard measure of harvest output.
- Alfalfa was grown from seeds for 14 days. Seedlings were treated with 1 mg/mL AB01 or mock treated. After treatment, plants were irrigated with 100 mL water containing 35 g/L dissolved salt (NaCl). Irrigation was performed every 48 hours and plants monitored for symptoms of salinity stress. After 5 days, control plants (left) displayed near complete chlorosis, while treated plants (right) displayed significantly higher vigor.
- Tomato seedlings were treated with 10 mg/mL AB01 or mock treated. 24 hours after treatment, they were irrigated with 250 mL water containing 29.2 g/L salt (NaCl). At 6 hours after salt irrigation, the control seedling (left) displayed symptoms of salinity shock which are manifest as severe wilting. The treated seedling (right) appeared unstressed.
- Corn seedlings were scored as unstressed, moderately water stressed and severely water stressed as measured by visual inspection of leaf rolling, a response of corn to water stress.
- Dekalb 67-86 RR corn seeds were planted and then irrigated through 7 days after emergence in a greenhouse setting. Seedlings were then treated with 40 mL of water, 0.1 mM AB01 or 1.0 mM AB01 and irrigation ceased. On the 6th day without irrigation, seedlings were scored for water stress. Seedlings with open leaves were scored as unstressed; seedlings with partial leaf rolling were scored as moderately stressed; seedlings with completely rolled leaves were scored as severely stressed. We found that AB01 treatment decreased the fraction of severely stressed seedlings while increasing the fraction of unstressed seedlings in a dose-dependent manner.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Zoology (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Genetics & Genomics (AREA)
- General Health & Medical Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- General Engineering & Computer Science (AREA)
- Environmental Sciences (AREA)
- Dentistry (AREA)
- Plant Pathology (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Toxicology (AREA)
- Gastroenterology & Hepatology (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Breeding Of Plants And Reproduction By Means Of Culturing (AREA)
- Cultivation Of Plants (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Furan Compounds (AREA)
Abstract
Description
wherein:
R1, R2, R4, R5, R7, R8, R9, R10, R11, R12, R13, R14, R15, R16, and R17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR18;
R3 and R6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR18; or R3 and R6 together form a direct bond to provide a double bond;
each R18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, or —C(O)R19;
each R19 is independently alkyl, haloalkyl, aryl, or heteroaryl;
m is 0, 1, or 2; and
n is 1 or 2.
or a salt, solvate, polymorph, stereoisomer, or isomer thereof, wherein:
R1, R2, R4, R5, R7, R8, R9, R10, R11, R12, R13, R14, R15, R16, and R17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR18;
R3 and R6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR18; or R3 and R6 together form a direct bond to provide a double bond;
each R18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, or —C(O)R19; and
each R19 is independently alkyl, haloalkyl, aryl, or heteroaryl
or a salt, solvate, polymorph, stereoisomer, or isomer thereof,
wherein:
R1, R2, R7, R8, R9, R16, and R17 are each independently H, alkyl, haloalkyl, amino, halo, or —OR18; R3 and R6 are each independently H, alkyl, haloalkyl, amino, halo, or —OR18; or R3 and R6 together form a direct bond to provide a double bond;
each R18 is independently H, alkyl, haloalkyl, aryl, heteroaryl, or —C(O)R19; and
each R19 is independently alkyl, haloalkyl, aryl, or heteroaryl.
or a salt thereof, wherein R17 is H, alkyl, halo, or haloalkyl and X is Cl, Br, or I. In one embodiment, the method comprises: i) hydroxymethylation of an optionally substituted decahydronaphtho[2,1-b]furan-2(3aH)-one; and ii) subsequent alkylation of
or a salt thereof, wherein R17 is H, alkyl, halo, or haloalkyl and X is Cl, Br, or I. In another embodiment, the hydroxymethylation comprises a reaction between sclareolide and methyl formate in the presence of potassium tert-butoxide and the alkylation comprises a reaction between the hydroxymethylation product and 5-bromo-3-methylfuran-2(5H)-one. In another embodiment, the optionally substituted decahydronaphtho[2,1-b]furan-2(3aH)-one comprises sclareolide. In another embodiment, the R17 is alkyl. In another embodiment, the R17 is methyl. In another embodiment, the X is Cl. In another embodiment, the hydroxymethylation and alkylation are a one pot procedure.
the stereocenter * is selected from the group consisting of: (S), (R), racemic, and a non-racemic mixture of (R) and (S). In another embodiment, the compound, salt, solvate, polymorph, diastereoisomer, stereoisomer, or isomer, wherein in the moiety:
a salt, solvate, polymorph, stereoisomer, or isomer thereof, obtained by a process comprising a condensation reaction on a sesquiterpene lactone, salt, solvate, polymorph, stereoisomer, isomer or derivative thereof. The chemical mimic of strigolactone may be obtained by a process comprising a condensation reaction on a sesquiterpene lactone. The sesquiterpene lactone may be sclareolide.
The two diastereomers may have a ratio of 1:1. Alternatively, the ratio of Formula IV to Formula V is 1:2. The ratio of Formula IV to Formula V may be 1:3. The ratio of Formula IV to Formula V may be 1:4. The ratio of Formula IV to Formula V may be 1:5. The ratio of Formula IV to Formula V may be at least about 1:6; 1:7; 1:8; 1:9; 1:10; 1:11; 1:12; 1:13; 1:14; 1:15: 1:16: 1:17; 1:18; 1:19; 1:20; 1:25; 1:30; 1:35; 1:40; 1:45; 1:50; 1:60; 1:70; 1:80; 1:90; or 1:100. The ratio of Formula IV to Formula V may be 1:10. The ratio of Formula IV to Formula V may be 1:20. The ratio of Formula IV to Formula V may be 1:40. The ratio of Formula V to Formula IV may be 1:2. The ratio of Formula V to Formula IV may be 1:3. The ratio of Formula V to Formula IV may be 1:4. The ratio of Formula V to Formula IV may be 1:5. The ratio of Formula V to Formula IV may be at least about 1:6; 1:7; 1:8; 1:9; 1:10; 1:11; 1:12; 1:13; 1:14; 1:15: 1:16: 1:17; 1:18; 1:19; 1:20; 1:25; 1:30; 1:35; 1:40; 1:45; 1:50; 1:60; 1:70; 1:80; 1:90; or 1:100. The ratio of Formula V to Formula IV may be 1:10. The ratio of Formula V to Formula IV may be 1:20. The ratio of Formula V to Formula IV may be 1:40.
wherein R17 is H, alkyl, halo, or haloalkyl and X is Cl, Br, or I. The plant propagation material may be a chemical mimic of strigolactone. The plant propagation material may be a compound of Formula I, II, III, IV, V, VI, VII, VIII, IX or X.
- 1. Boyer, J. S. and M. E. Westgate, Grain yields with limited water. J Exp Bot, 2004. 55(407): p. 2385-94.
- 2. Nielsen, R., Corn growth and development, what goes on from planting to harvest? Extension University, 1997.
- 3. Schoper, J. B., et al., Plant factors controlling seed set in maize: the influence of silk, pollen, and ear-leaf water status and tassel heat treatment at pollination. Plant Physiol, 1987. 83(1): p. 121-5.
- 4. United States Department of Agriculture, National Agricultural Statistics Service Crop production report released September 2013.
- 5. United States Department of Agriculture, Risk Management Agency RMA Indemnities (As of Jul. 8, 2013). Accessed 26 Sep. 2013 from: http://www.rma.usda.gov/data/indemnity/2013/070813table.pdf.
- 6. O'Connor, C., Soil Matters: How the Federal Crop Insurance Program should be reformed to encourage low-risk farming methods with high-reward environmental outcomes. 2013.
- 7. Climate Stabilization Targets: Emissions, Concentrations, and Impacts over Decades to Millennia. 2011: The National Academies Press.
- 8. Bruce, W. B., G. O. Edmeades, and T. C. Barker, Molecular and physiological approaches to maize improvement for drought tolerance. J Exp Bot, 2002. 53(366): p. 13-25.
- 9. Harrigan, G. G., et al., The forage and grain of MON 87460, a drought-tolerant corn hybrid, are formulationally equivalent to that of conventional corn. J Agric Food Chem, 2009. 57(20): p. 9754-63.
- 10. Tollefson, J., Drought-tolerant maize gets US debut. Nature, 2011. 469(7329): p. 144.
- 11. Peleg, Z. and E. Blumwald, Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol, 2011. 14(3): p. 290-5.
- 12. Prasch, C. M. and U. Sonnewald, Simultaneous application of heat, drought, and virus to Arabidopsis plants reveals significant shifts in signaling networks. Plant Physiol, 2013. 162(4): p. 1849-66.
- 13. Rivero, R. M., et al., Enhanced cytokinin synthesis in tobacco plants expressing PSARK::IPT prevents the degradation of photosynthetic protein complexes during drought. Plant Cell Physiol, 2010. 51(11): p. 1929-41.
- 14. Harris, M. J., et al., Water-stress-induced changes in the abscisic acid content of guard cells and other cells of Vicia faba L. leaves as determined by enzyme-amplified immunoassay. Proc Natl Acad Sci USA, 1988. 85(8): p. 2584-8.
- 15. Kim, T. H., et al., Guard cell signal transduction network: advances in understanding abscisic acid, CO2, and Ca2+ signaling. Annu Rev Plant Biol, 2010. 61: p. 561-91.
- 16. Tsuchiya, Y. and P. McCourt, Strigolactones: a new hormone with a past. Curr Opin Plant Biol, 2009. 12(5): p. 556-61.
- 17. Alder, A., et al., The path from beta-carotene to carlactone, a strigolactone-like plant hormone. Science, 2012. 335(6074): p. 1348-51.
- 18. Lopez-Raez, J. A., et al., Does abscisic acid affect strigolactone biosynthesis? New Phytol, 2010. 187(2): p. 343-54.
- 19. Peppi, M. C., M. W. Fidelibus, and N. Dokoozlian, Abscisic Acid Application Timing and Concentration Affect Firmness, Pigmentation, and Color of Flame Seedless' Grapes. HortScience, 2006. 41(6): p. 1440-1445.
- 20. Lawrence, B. Production of clary sage oil and sclareol in North America. In Proceedings of the 4th international symposium on medicinal and aromatic plants. 1994.
- 21. Stephanopoulos, G., Synthetic biology and metabolic engineering. ACS Synth Biol, 2012. 1(11): p. 514-25.
- 22. Clark, M., et al., Agronomic, economic, and environmental comparison of pest management in conventional and alternative tomato and corn systems in northern California. Agriculture, Ecosystems & Environment, 1998. 68(1): p. 51-71.
- 23. Witt, S., et al., Metabolic and phenotypic responses of greenhouse-grown maize hybrids to experimentally controlled drought stress. Mol Plant, 2012. 5(2): p. 401-17.
- 24. Chugh, V., et al., Differential antioxidative response of tolerant and sensitive maize (Zea mays L.) genotypes to drought stress at reproductive stage. Indian J Biochem Biophys, 2013. 50(2): p. 150-8.
- 25. Eddy, R. and D. T Hahn, Optimizing Greenhouse Corn Production: Materials and Methods. Purdue Methods for Corn Growth, 2010.
- 26. Eddy, R. and D. T Hahn, Optimizing Greenhouse Corn Production: Summary. Purdue Methods for Corn Growth, 2012.
- 27. Rink, W., et al., Optimizing Greenhouse Corn Production: What Is the Best Irrigation Strategy? Purdue Methods for Corn Growth, 2010.
- 28. Eddy, R. and D. T Hahn, Optimizing Greenhouse Corn Production: What Is the Best Lighting and Plant Density? Purdue Methods for Corn Growth, 2010.
- 29. Leonberger, A., R. Eddy, and D. T Hahn, Optimizing Greenhouse Corn Production: What Is the Best Open Pollination Method? Purdue Methods for Corn Growth, 2010.
- 30. Gambrel, D., R. Eddy, and D. T Hahn, Optimizing Greenhouse Corn Production: What Is the Best Pot Size? Purdue Methods for Corn Growth, 2010.
- 31. Gambrel, D., et al., Optimizing Greenhouse Corn Production: What Is the Best Root Medium? Purdue Methods for Corn Growth, 2010.
- 32. Tarkalson, D. D., S. J. Van Donk, and J. L. Petersen, Effect of nitrogen application timing on corn production using subsurface drip irrigation. Soil Science, 2009. 174(3): p. 174-179.
- 33. Yoshida, S. and K. Shirasu, Plants that attack plants: molecular elucidation of plant parasitism. Curr Opin Plant Biol, 2012. 15(6): p. 708-13.
- 34. Wigchert, S. C., et al., Dose-response of seeds of the parasitic weeds Striga and Orobanche toward the synthetic germination stimulants GR 24 and
Nijmegen 1. J Agric Food Chem, 1999. 47(4): p. 1705-10. - 35. Pimentel, D., et al., Environmental and economic costs of pesticide use. BioScience, 1992. 42(10): p. 750-760.
- 36. Lake, L. K., et al., Regulation of Biochemical Plant Growth Regulators at the US Environmental Protection Agency. HortTechnology, 2002. 12(1): p. 55-58.
- 37. Okamoto, M., et al., Activation of dimeric ABA receptors elicits guard cell closure, ABA-regulated gene expression, and drought tolerance. Proc Natl Acad Sci USA, 2013. 110(29): p. 12132-7.
- 38. E. M. Magnus, B. Zwanenburg. J. Agric. Food. Chem. 1992, 40, 1066-1070.
- 39. B. Zwanenburg, A. S. Mwakaboko, A. Reizelman, G. Anilkumar, D. Sethumadhavan. Pest Manag. Sci. 2009, 65, 478-491.
- 40. B. Zwanenburg, T. Pospisil. Molecular Plant 2013, 6, 38-62.
Claims (14)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/947,508 US10577358B2 (en) | 2013-10-25 | 2018-04-06 | Strigolactone formulations and uses thereof |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361895893P | 2013-10-25 | 2013-10-25 | |
| US201361918552P | 2013-12-19 | 2013-12-19 | |
| PCT/US2014/062297 WO2015061764A1 (en) | 2013-10-25 | 2014-10-24 | Strigolactone formulations and uses thereof |
| US14/856,908 US9994557B2 (en) | 2013-10-25 | 2015-09-17 | Strigolactone formulations and uses thereof |
| US15/947,508 US10577358B2 (en) | 2013-10-25 | 2018-04-06 | Strigolactone formulations and uses thereof |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/856,908 Continuation US9994557B2 (en) | 2013-10-25 | 2015-09-17 | Strigolactone formulations and uses thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20190084970A1 US20190084970A1 (en) | 2019-03-21 |
| US10577358B2 true US10577358B2 (en) | 2020-03-03 |
Family
ID=52993675
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/856,908 Active US9994557B2 (en) | 2013-10-25 | 2015-09-17 | Strigolactone formulations and uses thereof |
| US15/947,508 Active US10577358B2 (en) | 2013-10-25 | 2018-04-06 | Strigolactone formulations and uses thereof |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/856,908 Active US9994557B2 (en) | 2013-10-25 | 2015-09-17 | Strigolactone formulations and uses thereof |
Country Status (16)
| Country | Link |
|---|---|
| US (2) | US9994557B2 (en) |
| EP (1) | EP3060561A4 (en) |
| JP (1) | JP6485839B2 (en) |
| KR (1) | KR102299236B1 (en) |
| CN (1) | CN105849104B (en) |
| AP (1) | AP2016009221A0 (en) |
| AU (1) | AU2014339828B2 (en) |
| CA (1) | CA2926731C (en) |
| CL (1) | CL2016000981A1 (en) |
| EA (1) | EA201690849A1 (en) |
| GB (1) | GB2535911B (en) |
| IL (1) | IL244883B (en) |
| MX (1) | MX381481B (en) |
| UA (1) | UA121855C2 (en) |
| WO (1) | WO2015061764A1 (en) |
| ZA (1) | ZA201602510B (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11678668B2 (en) | 2018-06-27 | 2023-06-20 | Pivot Bio, Inc. | Agricultural compositions comprising remodeled nitrogen fixing microbes |
| US12365868B2 (en) | 2023-06-20 | 2025-07-22 | Tenfold Technologies, LLC | Systems for production of products to promote nitrogen use efficiency in plants |
Families Citing this family (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9512431B2 (en) | 2011-06-16 | 2016-12-06 | The Regents Of The University Of California | Synthetic gene clusters |
| US10968446B2 (en) | 2012-11-01 | 2021-04-06 | Massachusetts Institute Of Technology | Directed evolution of synthetic gene cluster |
| CN105849104B (en) | 2013-10-25 | 2020-05-22 | 健康农业公司 | Strigolide preparation and use thereof |
| PL3286171T3 (en) | 2015-04-24 | 2022-01-10 | Sound Agriculture Company | Methods for hydraulic enhancement of crops |
| KR102197507B1 (en) | 2015-07-13 | 2020-12-31 | 피벗 바이오, 인크. | Methods and compositions for improving plant traits |
| GB201513971D0 (en) * | 2015-08-07 | 2015-09-23 | Syngenta Participations Ag | Plant growth regulator compounds |
| CN108291219B (en) | 2015-10-05 | 2023-02-17 | 麻省理工学院 | Nitrogen fixation using reconstructed nif clusters |
| GB201615544D0 (en) | 2016-09-13 | 2016-10-26 | Syngenta Participations Ag | Plant Growth regulator compounds |
| CN106636180B (en) * | 2016-09-18 | 2020-04-07 | 山东农业大学 | Plasmid vector and method for obtaining plant highly sensitive to salt stress |
| CA3049258A1 (en) | 2017-01-12 | 2018-07-19 | Pivot Bio, Inc. | Methods and compositions for improving plant traits |
| GB201702158D0 (en) | 2017-02-09 | 2017-03-29 | Syngenta Participations Ag | Germination promoters |
| JP7203747B2 (en) | 2017-02-24 | 2023-01-13 | ビーエーエスエフ ソシエタス・ヨーロピア | Process for the preparation of unsaturated carboxylic acids by carbonylation of allyl alcohols and their acylation products |
| US12151988B2 (en) | 2017-10-25 | 2024-11-26 | Pivot Bio, Inc. | Gene targets for nitrogen fixation targeting for improving plant traits |
| RU2020116764A (en) | 2017-10-25 | 2021-11-25 | Пивот Байо, Инк. | METHODS AND COMPOSITIONS FOR IMPROVING CONSTRUCTED NITROGEN-FIXING MICROORGANISMS |
| CN107653252B (en) * | 2017-11-16 | 2021-07-06 | 河南大学 | Application of cotton GbSLR1 gene in plant root and branch development |
| BR112020009330A2 (en) | 2017-11-17 | 2020-10-13 | Koch Biological Solutions, Llc | strigolactone derivatives |
| CN108912074B (en) * | 2018-06-15 | 2020-09-25 | 清华大学 | High-activity strigolactone derivative and preparation and application thereof |
| CN108623397A (en) * | 2018-07-19 | 2018-10-09 | 安徽袁粮水稻产业有限公司 | A kind of rice fertilizer inhibiting ineffective tillering |
| CN109321510B (en) * | 2018-09-29 | 2021-08-20 | 昆明理工大学 | Application of strigolactone in promoting the accumulation of lipids in the algae |
| EP3662752A1 (en) | 2018-12-03 | 2020-06-10 | Institutul National de Cercetare-Dezvoltare Pentru Chimie si Petrochimie-Icehim | Biostimulant for plants obtained from root exudates accumulated in recirculated hydroponic environments |
| CN109452311B (en) * | 2018-12-12 | 2021-04-06 | 四川国光农化股份有限公司 | Plant growth regulating composition, preparation and application thereof |
| CN109430271A (en) * | 2018-12-12 | 2019-03-08 | 四川国光农化股份有限公司 | A kind of plant growth regualting composition, preparation and its application |
| US12281299B2 (en) | 2019-03-19 | 2025-04-22 | Massachusetts Institute Of Technology | Control of nitrogen fixation in rhizobia that associate with cereals |
| AU2020445067A1 (en) | 2020-05-01 | 2022-12-01 | Pivot Bio, Inc. | Measurement of nitrogen fixation and incorporation |
| US20230295559A1 (en) | 2020-05-13 | 2023-09-21 | Pivot Bio, Inc. | De-repression of nitrogen fixation in gram-positive microorganisms |
| CN113383941B (en) * | 2021-06-25 | 2022-12-06 | 仲景食品股份有限公司 | Preparation method of high-hemp-degree pepper powder |
| EP4637358A1 (en) | 2022-12-23 | 2025-10-29 | Pivot Bio, Inc. | Combination of various nitrogen fixing bacteria with various biological products to achieve synergistic effects |
| CN116448929A (en) * | 2023-06-02 | 2023-07-18 | 湖南农业大学 | A kind of pretreatment method of strigolactone in plant sample |
| CN116982617A (en) * | 2023-07-14 | 2023-11-03 | 贵州大学 | Application of strigolactone in prolonging ornamental life of fresh cut China rose |
| CN118077719B (en) * | 2024-04-25 | 2024-07-09 | 赤峰市乾铭农业科技发展有限公司 | Application of a microbial preparation in Platycodon grandiflorum cultivation |
Citations (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1141952A (en) | 1995-06-09 | 1997-02-05 | 霍夫曼-拉罗奇有限公司 | Carotenoid Fermentation Production Method |
| WO1998031837A1 (en) | 1997-01-17 | 1998-07-23 | Maxygen, Inc. | Evolution of whole cells and organisms by recursive sequence recombination |
| US7038064B2 (en) | 2000-07-19 | 2006-05-02 | Diacel Chemical Industries, Ltd. | Process for producing hydroxylactones |
| US20080318773A1 (en) | 2004-02-10 | 2008-12-25 | Guillaume Becard | Modulators of the Development of Mychorrizal Fungi with Arbuscules, and Uses Thereof |
| US7825268B2 (en) | 2003-12-23 | 2010-11-02 | Henkel Ag & Co. Kgaa | Alkoxylactones, alkoxylactams and alkoxythiolactams for controlling processes based on microbial interaction |
| WO2010125065A2 (en) | 2009-04-28 | 2010-11-04 | Bayer Cropscience Ag | Compositions comprising a strigolactone compound and a chito-oligosaccharide compound for enhanced plant growth and yield |
| US20110207608A1 (en) | 2008-04-30 | 2011-08-25 | The Regents Of The University Of California | Transcriptional and post-transcription regulation of transcription factor for drought resistance |
| US20110230352A1 (en) | 2008-04-23 | 2011-09-22 | Institut National De La Recherche Agronomique | Treatment process for a superior plant in order to control its growth and architecture |
| WO2012057404A1 (en) | 2010-10-25 | 2012-05-03 | Lee Ji Hoon | Portable smokescreen insecticide sprayer |
| CN103068241A (en) | 2010-06-04 | 2013-04-24 | 先正达参股股份有限公司 | plant watering method |
| WO2013092430A1 (en) | 2011-12-19 | 2013-06-27 | Syngenta Participations Ag | Strigolactam derivatives as plant growth regulating compounds |
| CN103396390A (en) | 2013-07-22 | 2013-11-20 | 南京农业大学 | Total synthesis method of strigolactones GR24 |
| US8946280B2 (en) | 2010-12-14 | 2015-02-03 | Syngenta Participations Ag | Plant growth regulating compounds |
| WO2015061764A1 (en) | 2013-10-25 | 2015-04-30 | Asilomar Bio, Inc. | Strigolactone formulations and uses thereof |
| US20150274690A1 (en) | 2013-10-25 | 2015-10-01 | Asilomar Bio, Inc. | Strigolactone Compositions And Uses Thereof |
| WO2016172655A1 (en) | 2015-04-24 | 2016-10-27 | Asilomar Bio, Inc. | Methods for hydraulic enhancement of crops |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012157404A1 (en) | 2011-05-18 | 2012-11-22 | 国立大学法人 神戸大学 | Root parasitic plant germination modifier and method for control of root parasitic plant using same |
-
2014
- 2014-10-24 CN CN201480070453.XA patent/CN105849104B/en active Active
- 2014-10-24 UA UAA201605593A patent/UA121855C2/en unknown
- 2014-10-24 MX MX2016005373A patent/MX381481B/en unknown
- 2014-10-24 WO PCT/US2014/062297 patent/WO2015061764A1/en not_active Ceased
- 2014-10-24 GB GB1608553.2A patent/GB2535911B/en active Active
- 2014-10-24 EP EP14855263.1A patent/EP3060561A4/en active Pending
- 2014-10-24 EA EA201690849A patent/EA201690849A1/en unknown
- 2014-10-24 CA CA2926731A patent/CA2926731C/en active Active
- 2014-10-24 JP JP2016550683A patent/JP6485839B2/en active Active
- 2014-10-24 AP AP2016009221A patent/AP2016009221A0/en unknown
- 2014-10-24 AU AU2014339828A patent/AU2014339828B2/en active Active
- 2014-10-24 KR KR1020167013677A patent/KR102299236B1/en active Active
-
2015
- 2015-09-17 US US14/856,908 patent/US9994557B2/en active Active
-
2016
- 2016-04-04 IL IL244883A patent/IL244883B/en active IP Right Grant
- 2016-04-13 ZA ZA2016/02510A patent/ZA201602510B/en unknown
- 2016-04-25 CL CL2016000981A patent/CL2016000981A1/en unknown
-
2018
- 2018-04-06 US US15/947,508 patent/US10577358B2/en active Active
Patent Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1141952A (en) | 1995-06-09 | 1997-02-05 | 霍夫曼-拉罗奇有限公司 | Carotenoid Fermentation Production Method |
| WO1998031837A1 (en) | 1997-01-17 | 1998-07-23 | Maxygen, Inc. | Evolution of whole cells and organisms by recursive sequence recombination |
| US7038064B2 (en) | 2000-07-19 | 2006-05-02 | Diacel Chemical Industries, Ltd. | Process for producing hydroxylactones |
| US7825268B2 (en) | 2003-12-23 | 2010-11-02 | Henkel Ag & Co. Kgaa | Alkoxylactones, alkoxylactams and alkoxythiolactams for controlling processes based on microbial interaction |
| US20080318773A1 (en) | 2004-02-10 | 2008-12-25 | Guillaume Becard | Modulators of the Development of Mychorrizal Fungi with Arbuscules, and Uses Thereof |
| US8101171B2 (en) | 2004-02-10 | 2012-01-24 | Universite Paul Sabatier Toulouse Iii | Modulators of the development of mychorrizal fungi with arbuscules, and uses thereof |
| US20110230352A1 (en) | 2008-04-23 | 2011-09-22 | Institut National De La Recherche Agronomique | Treatment process for a superior plant in order to control its growth and architecture |
| US20110207608A1 (en) | 2008-04-30 | 2011-08-25 | The Regents Of The University Of California | Transcriptional and post-transcription regulation of transcription factor for drought resistance |
| US9131685B2 (en) | 2009-04-28 | 2015-09-15 | Bayer Cropscience Ag | Compositions comprising a strigolactone compound and a chito-oligosaccharide compound for enhanced plant growth and yield |
| WO2010125065A2 (en) | 2009-04-28 | 2010-11-04 | Bayer Cropscience Ag | Compositions comprising a strigolactone compound and a chito-oligosaccharide compound for enhanced plant growth and yield |
| US20120046169A1 (en) | 2009-04-28 | 2012-02-23 | Peter Dahman | Compositions comprising a strigolactone compound and a chito-oligosaccharide compound for enhanced plant growth and yield |
| CN103068241A (en) | 2010-06-04 | 2013-04-24 | 先正达参股股份有限公司 | plant watering method |
| WO2012057404A1 (en) | 2010-10-25 | 2012-05-03 | Lee Ji Hoon | Portable smokescreen insecticide sprayer |
| US8946280B2 (en) | 2010-12-14 | 2015-02-03 | Syngenta Participations Ag | Plant growth regulating compounds |
| WO2013092430A1 (en) | 2011-12-19 | 2013-06-27 | Syngenta Participations Ag | Strigolactam derivatives as plant growth regulating compounds |
| CN103396390A (en) | 2013-07-22 | 2013-11-20 | 南京农业大学 | Total synthesis method of strigolactones GR24 |
| WO2015061764A1 (en) | 2013-10-25 | 2015-04-30 | Asilomar Bio, Inc. | Strigolactone formulations and uses thereof |
| US20150274690A1 (en) | 2013-10-25 | 2015-10-01 | Asilomar Bio, Inc. | Strigolactone Compositions And Uses Thereof |
| US9994557B2 (en) | 2013-10-25 | 2018-06-12 | Asilomar Bio, Inc. | Strigolactone formulations and uses thereof |
| WO2016172655A1 (en) | 2015-04-24 | 2016-10-27 | Asilomar Bio, Inc. | Methods for hydraulic enhancement of crops |
Non-Patent Citations (81)
| Title |
|---|
| "Mwakaboko, A. S. Synthesis and Biological Evaluation of new Strigolactone Analogues as Germination Stimulants for the Seeds of the Parasitic Weeds Striga and Orobanche spp. Thesis. Catholic University Nijmegen, Netherlands. Mar. 25, 2003.". |
| Akiyama, et al. Strigolactones: chemical signals for fungal symbionts and parasitic weeds in plant roots. Ann Bot. Jun. 2006;97(6):925-31. Epub Mar. 30, 2006. |
| Alder, et al. The path from β-carotene to carlactone, a strigolactone-like plant hormone. Science. Mar. 16, 2012;335(6074):1348-51. doi: 10.1126/science.1218094. |
| Besserer, et al. GR24, a synthetic analog of strigolactones, stimulates the mitosis and growth of the arbuscular mycorrhizal fungus Gigaspora rosea by boosting its energy metabolism. Plant Physiol. Sep. 2008;148(1):402-13. doi: 10.1104/pp.108.121400. Epub Jul. 9, 2008. |
| Bouwmeester, et al. Secondary metabolite signalling in host-parasitic plant interactions. Curr Opin Plant Biol. Aug. 2003;6(4):358-64. |
| Boyer, et al. Grain yields with limited water. J Exp Bot. Nov. 2004;55(407):2385-94. Epub Jul. 30, 2004. |
| Boyer, et al. New Strigolactone Analogs as Plant Hormones with Low Activities in the Rhizosphere. Molecular Plant Advance Access, published Dec. 26, 2013. |
| Boyer, et al. Structure-activity relationship studies of strigolactone-related molecules for branching inhibition in garden pea: molecule design for shoot branching. Plant Physiol. Aug. 2012;159(4):1524-44. doi: 10.1104/pp.112.195826. Epub Jun. 21, 2012. |
| Bruce, et al. Molecular and physiological approaches to maize improvement for drought tolerance. J Exp Bot. Jan. 2002;53(366):13-25. |
| CAS Registry No. 1401956-60-2, STN Entry Date Oct. 24, 2012. |
| CAS Registry No. 920959-02-0, STN Entry Date Feb. 14, 2007. |
| Chadwick, et al. Sesquiterpenoids lactones: benefits to plants and people. Int J Mol Sci. Jun. 19, 2013;14(6):12780-805. doi: 10.3390/ijms140612780. |
| Chugh, et al. Differential antioxidative response of tolerant and sensitive maize (Zea mays L.) genotypes to drought stress at reproductive stage. Indian J Biochem Biophys. Apr. 2013;50(2):150-8. |
| Clark, et al. Agronomic, economic, and environmental comparison of pest management in conventional and alternative tomato and corn systems in northern California. Agriculture, Ecosystems & Environment. vol. 68, Issues 1-2, Mar. 1998, pp. 51-71. |
| Climate Stabilization Targets: Emissions, Concentrations, and Impacts over Decades to Millenina. 2011: The National Academies Press. |
| Cohen, et al. Structure-function relations of strigolactone analogs: activity as plant hormones and plant interactions. Mol Plant. Jan. 2013;6(1):141-52. doi: 10.1093/mp/sss134. Epub Dec. 8, 2012. |
| Co-pending U.S. Appl. No. 15/568,716, filed Oct. 23, 2017. |
| Eddy, et al. Optimizing Greenhouse Corn Prduction: Summary. Purdue Methods for Corn Growth, 2012. |
| Eddy, et al. Optimizing Greenhouse Corn Production: Materials and Methods. Purdue Methods for Corn Growth, 2010. |
| Eddy, et al. Optimizing Greenhouse corn Production: What is the Best Lighting and Plant Density? Purdue Methods for Corn Growth, 2010. |
| European search report and search opinion dated Jun. 8, 2017 for EP Application No. 14855263.1. |
| Gambrel, et al. Optimizing Greenhouse Corn Production: What Is the Best Pot Size? Purdue Methods for Corn Growth, 2010. |
| Gambrel, et al. Optimizing Greenhouse Corn Production: What Is the Best Root Medium? Purdue Methods for Corn Growth, 2010. |
| Goulet, et al. Climbing the branches of the strigolactones pathway one discovery at a time. Plant Physiol. Oct. 2010;154(2):493-6. doi: 10.1104/pp.110.161026. |
| Harrigan, et al. The forage and grain of MON 87460, a drought-tolerant corn hybrid, are compositionally equivalent to that of conventional corn. J Agric Food Chem. Oct. 28, 2009;57(20):9754-63. doi: 10.1021/jf9021515. |
| Harris, et al. Water-stress-induced changes in the abscisic acid content of guard cells and other cells of Vicia faba L. leaves as determined by enzyme-amplified immunoassay. Proc Natl Acad Sci USA. Apr. 1988;85(8):2584-8. |
| Heetika Malik et al.,Aromatic A-ring analogues of orobanchol, new germination stimulants for seeds of parasitic weeds, Org. Biomol.Chem, Sep. 2011, 2286-2293. * |
| International search report and written opinion dated Jan. 16, 2015 for PCT Application No. US2014/062297. |
| International Search Report and Written Opinion dated Sep. 14, 2016 for International Application No. PCT/US2016/029080. |
| Kim, et al. Guard cell signal transduction network: advances in understanding abscisic acid, CO2, and Ca2+ signaling. Annu Rev Plant Biol. 2010;61:561-91. doi: 10.1146/annurev-arplant-042809-112226. |
| Lake, et al. Regulation of Biochemical Plant Growth Regulators at the U.S. Environmental Protection Agency. HortTechnology Jan.-Mar. 2002, vol. 12, No. 1, pp. 55-58. |
| Lawrence, B. Production of clary sage oil and sclareol in North America. In Proceedings of the 4th international symposium on medicinal and aromatic plants. 1994. |
| Leonberger, et al. Optimizing Greenhouse Corn Production: What Is the Best Open Pollination Method? Purdue Methods for Corn Growth, 2010. |
| Lopez-Raez, et al. Does abscisic acid affect strigolactone biosynthesis? New Phytol. Jul. 2010;187(2):343-54. doi: 10.1111/j.1469-8137.2010.03291.x. Epub May 10, 2010. |
| Lopez-Raez, et al. Strigolactones: ecological significance and use as a target for parasitic plant control. Pest Manag Sci. May 2009;65(5):471-7. doi: 10.1002/ps.1692. |
| Macias, et al. New Chemical Clues for Broomrape-Sunflower Host-Parasite Interactions: Synthesis of Guaianestrigolactones. Journal of agricultural and food chemistry 57.13 (2009): 5853-5864. |
| Macias, et al. New Chemical Clues for Broomrape-Sunflower Host—Parasite Interactions: Synthesis of Guaianestrigolactones. Journal of agricultural and food chemistry 57.13 (2009): 5853-5864. |
| Magnus, et al. Tentative Molecular Mechanism for Germination Stimulation of Striga and Orobanche Seeds by Strigol and Its Synthetic Analogues. J. Agric. Food. Chem. 1992, 40 1066-1070. |
| Malik, et al. Aromatic A-ring analogues of orbanchol, new germinati on stimulants for seeds of parasitic weeds. Organic & Biomolecular Chemistry. Apr. 7, 2011. 9(7), pp. 2286-2293. |
| Mwakaboko, et al. Single step synthesis of strigolactone analogues from cyclic keto enols, germination stimulants for seeds of parasitic weeds. Bioorg Med Chem. Aug. 15, 2011;19(16):5006-11. |
| Mwakaboko, et al. Strigolactone analogs derived from ketones using a working model for germination stimulants as a blueprint. Plant Cell Physiol. Apr. 2011;52(4):699-715. doi: 10.1093/pcp/pcr031. Epub Mar. 18, 2011. |
| Nielsen, R. Corn growth and development, what goes on from planting to harvest? Extension University, 1997. |
| O'Connor, C. Soil Matters: How the Federal Crop Insurance Program should be reformed to encourage low-risk farming methods with high-reward environmental outcomes. 2013. |
| Office action dated Apr. 17, 2017 for U.S. Appl. No. 14/856,908. |
| Office action dated Dec. 14, 2017 for U.S. Appl. No. 14/856,908. |
| Office action dated Oct. 21, 2016 for U.S. Appl. No. 14/856,908. |
| Okamoto, et al. Activation of dimeric ABA receptors elicits guard cell closure, ABA-regulated gene expression, and drought tolerance. Proc Natl Acad Sci U S A. Jul. 16, 2013;110(29):12132-7. doi: 10.1073/pnas.1305919110. Epub Jul. 1, 2013. |
| Peleg, et al. Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol. Jun. 2011;14(3):290-5. doi: 10.1016/j.pbi.2011.02.001. Epub Mar. 4, 2011. |
| Peppi, et al. Abscisic Acid Application Timing and Concentration Affect Firmness, Pigmentation, and Color of ‘Flame Seedless’ Grapes. HortScience Oct. 2006, vol. 41, No. 6, 1440-1445. |
| Peppi, et al. Abscisic Acid Application Timing and Concentration Affect Firmness, Pigmentation, and Color of 'Flame Seedless' Grapes. HortScience Oct. 2006, vol. 41, No. 6, 1440-1445. |
| Pimentel, et al. Environmental and Economic Costs of Pesticide Use. BioScience, vol. 42, No. 10, 1992. |
| Prasch, et al. Simultaneous application of heat, drought, and virus to Arabidopsis plants reveals significant shifts in signaling networks. Plant Physiol. Aug. 2013;162(4):1849-66. doi: 10.1104/pp.113.221044. Epub Jun. 10, 2013. |
| Pubchem, Substance Record for SID 54096325 Create Date: Oct. 8, 2008. [retrieved on Aug. 16, 2016]. Retrieved from the Internet: https://pubchem.ncbi.nlm.nih.gov/substance/54096325. |
| Pubchem, Substance Record for SID 55052802 Create Date: Oct. 8, 2008. [retrieved on Jun. 1, 2016]. Retrieved from the Internet: https://pubchem.ncbi.nlm.nih.gov/substance/55052802. |
| Qin, et al. Sesquiterpene lactones from Inula hupehensis inhibit nitric oxide production in RAW264.7 macrophages. Planta Med. Jun. 2012;78(10):1002-9. doi: 10.1055/s-0031-1298621. Epub May 30, 2012. |
| Raupp, et al. New sesquiterpene lactones from sunflower root exudate as germination stimulants for Orobanche cumana. J Agric Food Chem. Nov. 6, 2013;61(44):10481-7. doi: 10.1021/jf402392e. Epub Oct. 24, 2013. |
| Ren, et al. Cytotoxic and NF-κB inhibitory sesquiterpene lactones from Piptocoma rufescens. Tetrahedron. Mar. 25, 2012;68(12):2671-2678. Epub Jan. 26, 2012. |
| Rink, et al. Optimizing Greenouse Corn Production: What is the Best Irrigation Strategy? Purdue Methods for Corn Growth, 2010. |
| Rivero, et al. Enhanced cytokinin synthesis in tobacco plants expressing PSARK::IPT prevents the degradation of photosynthetic protein complexes during drought. Plant Cell Physiol. Nov. 2010;51(11):1929-41. doi: 10.1093/pcp/pcq143. Epub Sep. 24, 2010. |
| Rungeler, et al. Germacranolides from Mikania guaco. Phytochemistry. Mar. 2001;56(5):475-89. |
| Ruyter-Spira, et al. Physiological effects of the synthetic strigolactone analog GR24 on root system architecture in Arabidopsis: another belowground role for strigolactones? Plant Physiol. Feb. 2011;155(2):721-34. doi: 10.1104/pp.110.166645. Epub Nov. 30, 2010. |
| Schoper, et al. Plant factors controlling seed set in maize : the influence of silk, pollen, and ear-leaf water status and tassel heat treatment at pollination. Plant Physiol. Jan. 1987;83(1):121-5. |
| Shin, et al. Sesquiterpenes and other constituents from Dendranthema zawadskii var. latilobum. Chem Pharm Bull (Tokyo). 2012;60(3):306-14. |
| Stephanopoulous, G. Synthetic biology and metabolic engineering. ACS Synth Biol. Nov. 16, 2012;1(11):514-25. doi: 10.1021/sb300094q. |
| Tanaka, et al. Synthesis of 7-oxo-5-deoxystrigol, a 7-oxygenated strigolactone analog. Biosci Biotechnol Biochem. 2013;77(4):832-5. Epub Apr. 7, 2013. |
| Tarklason, et al. Effect of Nitrogen Application Timing on Corn Production Using Subsurface Drip Irrigation. Soil Science, Mar. 2009, vol. 174, Issue 3, pp. 174-179. |
| Toh, et al. Thermoinhibition uncovers a role for strigolactones in Arabidopsis seed germination. Plant Cell Physiol. Jan. 2012;53(1):107-17. |
| Tollefson, J. Drought-tolerant maize gets US debut. Nature. Jan. 13, 2011;469(7329):144. doi: 10.1038/469144a. |
| Tsuchiya, et al. Strigolactones: a new hormone with a past. Curr Opin Plant Biol. Oct. 2009;12(5):556-61. doi: 10.1016/j.pbi.2009.07.018. Epub Aug. 31, 2009. |
| U.S. Appl. No. 14/856,908 Notice of Allowance dated Mar. 29, 2018. |
| United States Department of Agriculture, National Agricultural Statistics Service Crop production report released Sep. 2013. |
| United States Department of Agriculture, Risk Management Agency RMA Indemnities (AS of Jul. 8, 2013). Accessed Sep. 26, 2013 from: http://www.rma.usda.gov/data/indemnity/2013/070813table.pdf. |
| Upar, et al. Efficient enantioselective synthesis of (+)-sclareolide and (+)-tetrahydroactinidiolide: chiral LBA-induced biomimetic cyclization. Tetrahedron: Asymmetry vol. 20, Issue 14, Jul. 29, 2009, pp. 1637-1640. |
| Wigchert, et al. Dose-response of seeds of the parasitic weeds Striga and Orobanche toward the synthetic germination stimulants GR 24 and Nijmegen 1. J Agric Food Chem. Apr. 1999;47(4):1705-10. |
| Witt, et al. Metabolic and phenotypic responses of greenhouse-grown maize hybrids to experimentally controlled drought stress. Mol Plant. Mar. 2012;5(2):401-17. doi: 10.1093/mp/ssr102. Epub Dec. 15, 2011. |
| Xie, et al. Fabacyl acetate, a germination stimulant for root parasitic plants from Pisum sativum. Phytochemistry 70.2 (2009): 211-215. |
| Yoneyama, et al. Characterization of strigolactones exuded by Asteraceae plants. Plant growth regulation 65.3 (2011): 495-504. |
| Yoneyma, et al. Strigolactones as a new plant growth regulator. Presentation at the MARCO Symposium 2009, Tsukuba, Japan, on Oct. 6, 2009. |
| Yoshida, et al. Plants that attack plants: molecular elucidation of plant parasitism. Curr Opin Plant Biol. Dec. 2012;15(6):708-13. doi: 10.1016/j.pbi.2012.07.004. Epub Aug. 13, 2012. |
| Zwanenburg, et al. Structure and Activity of Strigolactones: New Plant Hormones with a Rich Future. Mol. Plant, vol. 6, Issue 1, pp. 38-62. |
| Zwanenburg, et al. Structure and function of natural and synthetic signalling molecules in parasitic weed germination. Pest Manag Sci 2009; 65: 478-491. |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11678668B2 (en) | 2018-06-27 | 2023-06-20 | Pivot Bio, Inc. | Agricultural compositions comprising remodeled nitrogen fixing microbes |
| US11678667B2 (en) | 2018-06-27 | 2023-06-20 | Pivot Bio, Inc. | Agricultural compositions comprising remodeled nitrogen fixing microbes |
| US11963530B2 (en) | 2018-06-27 | 2024-04-23 | Pivot Bio, Inc. | Agricultural compositions comprising remodeled nitrogen fixing microbes |
| US12268212B2 (en) | 2018-06-27 | 2025-04-08 | Pivot Bio, Inc. | Agricultural compositions comprising remodeled nitrogen fixing microbes |
| US12290074B2 (en) | 2018-06-27 | 2025-05-06 | Pivot Bio, Inc. | Agricultural compositions comprising remodeled nitrogen fixing microbes |
| US12471599B2 (en) | 2018-06-27 | 2025-11-18 | Pivot Bio, Inc. | Agricultural compositions comprising remodeled nitrogen fixing microbes |
| US12365868B2 (en) | 2023-06-20 | 2025-07-22 | Tenfold Technologies, LLC | Systems for production of products to promote nitrogen use efficiency in plants |
| US12365869B2 (en) | 2023-06-20 | 2025-07-22 | Tenfold Technologies, LLC | Systems for production of products to promote nitrogen use efficiency in plants |
Also Published As
| Publication number | Publication date |
|---|---|
| KR102299236B1 (en) | 2021-09-06 |
| US20160159780A1 (en) | 2016-06-09 |
| GB2535911B (en) | 2020-10-21 |
| EP3060561A1 (en) | 2016-08-31 |
| AU2014339828B2 (en) | 2019-02-14 |
| CN105849104A (en) | 2016-08-10 |
| CN105849104B (en) | 2020-05-22 |
| US9994557B2 (en) | 2018-06-12 |
| WO2015061764A1 (en) | 2015-04-30 |
| JP6485839B2 (en) | 2019-03-20 |
| EP3060561A4 (en) | 2017-07-05 |
| US20190084970A1 (en) | 2019-03-21 |
| MX381481B (en) | 2025-03-12 |
| MX2016005373A (en) | 2017-01-05 |
| EA201690849A1 (en) | 2016-11-30 |
| AP2016009221A0 (en) | 2016-05-31 |
| IL244883B (en) | 2020-04-30 |
| GB2535911A (en) | 2016-08-31 |
| CA2926731A1 (en) | 2015-04-30 |
| ZA201602510B (en) | 2018-07-25 |
| UA121855C2 (en) | 2020-08-10 |
| CL2016000981A1 (en) | 2017-01-13 |
| CA2926731C (en) | 2022-08-30 |
| IL244883A0 (en) | 2016-05-31 |
| JP2017503002A (en) | 2017-01-26 |
| KR20160079013A (en) | 2016-07-05 |
| GB201608553D0 (en) | 2016-06-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10577358B2 (en) | Strigolactone formulations and uses thereof | |
| AU2014339828A1 (en) | Strigolactone formulations and uses thereof | |
| US12239132B2 (en) | Compounds and methods for increasing soil nutrient availability | |
| TW201138632A (en) | Active compound combinations | |
| US20180310557A1 (en) | Methods for hydraulic enhancement of crops | |
| KR20140062099A (en) | Insecticidal lipid agents isolated from entomopathogenic fungi and uses thereof | |
| US20150274690A1 (en) | Strigolactone Compositions And Uses Thereof | |
| TW200911119A (en) | Fungicidal active compound combination | |
| JP2022097560A (en) | Plant irrigation reducing agents and methods for reducing plant irrigation | |
| UA127646C2 (en) | Use of alkoxypyrazoles as nitrification inhibitors | |
| CN101828557A (en) | Pyrimidine nucleoside antibiotic and hexaconazole compound bactericide | |
| KR20120068674A (en) | Streptomyces griseus big105 for controlling plant diseases and uses thereof | |
| CN112825861A (en) | Compound pesticide herbicide | |
| CN102217616B (en) | Fluopicolpkd-containing bactericidal composition | |
| CN106922703A (en) | A kind of bactericidal composition | |
| CN106922694A (en) | A kind of bactericidal composition | |
| TW201204260A (en) | Active compound combinations | |
| CN105475357A (en) | Bactericidal composition | |
| UA127764C2 (en) | SILYLETHYNYLHETARYLIC COMPOUNDS AS NITRIFICATION INHIBITORS | |
| KR102653110B1 (en) | Composition for controlling plant diseases comprising Boesenbergia pulcherrima extract | |
| CN108419798A (en) | A kind of bactericidal composition and its application containing alkene oxime amine | |
| CN106922704A (en) | A kind of bactericidal composition | |
| RU2331194C1 (en) | Remedy for controlling wheat diseases | |
| CN103988846B (en) | A kind of bactericidal composition | |
| CN109907053A (en) | A kind of composition pesticide and its application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FEPP | Fee payment procedure |
Free format text: ENTITY STATUS SET TO UNDISCOUNTED (ORIGINAL EVENT CODE: BIG.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NOTICE OF ALLOWANCE MAILED -- APPLICATION RECEIVED IN OFFICE OF PUBLICATIONS |
|
| AS | Assignment |
Owner name: ASILOMAR BIO, INC., CALIFORNIA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:DAVIDSON, ERIC A.;BAYER, TRAVIS S.;WINDRAM, OLIVER;AND OTHERS;SIGNING DATES FROM 20160406 TO 20160408;REEL/FRAME:050894/0423 Owner name: SOUND AGRICULTURE COMPANY, CALIFORNIA Free format text: CHANGE OF NAME;ASSIGNOR:ASILOMAR BIO, INC.;REEL/FRAME:050915/0656 Effective date: 20180918 |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: PUBLICATIONS -- ISSUE FEE PAYMENT VERIFIED |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1551); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 4 |