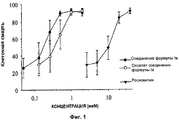
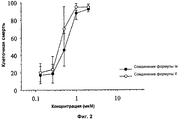
RU2500400C2 - Применение производных пурина для изготовления лекарственного препарата - Google Patents
Применение производных пурина для изготовления лекарственного препарата Download PDFInfo
- Publication number
- RU2500400C2 RU2500400C2 RU2010108505/15A RU2010108505A RU2500400C2 RU 2500400 C2 RU2500400 C2 RU 2500400C2 RU 2010108505/15 A RU2010108505/15 A RU 2010108505/15A RU 2010108505 A RU2010108505 A RU 2010108505A RU 2500400 C2 RU2500400 C2 RU 2500400C2
- Authority
- RU
- Russia
- Prior art keywords
- compound
- formula
- compounds
- esters
- hydrates
- Prior art date
Links
- 239000003814 drug Substances 0.000 title claims abstract description 13
- 229940079593 drug Drugs 0.000 title abstract description 7
- 238000002360 preparation method Methods 0.000 title abstract description 4
- 229940083251 peripheral vasodilators purine derivative Drugs 0.000 title abstract description 3
- 125000000561 purinyl group Chemical class N1=C(N=C2N=CNC2=C1)* 0.000 title abstract 2
- 150000001875 compounds Chemical class 0.000 claims abstract description 103
- 208000032852 chronic lymphocytic leukemia Diseases 0.000 claims abstract description 31
- 208000030761 polycystic kidney disease Diseases 0.000 claims abstract description 12
- 208000010839 B-cell chronic lymphocytic leukemia Diseases 0.000 claims description 32
- 208000031422 Lymphocytic Chronic B-Cell Leukemia Diseases 0.000 claims description 22
- 150000002148 esters Chemical class 0.000 claims description 20
- 150000003839 salts Chemical class 0.000 claims description 15
- 150000004677 hydrates Chemical class 0.000 claims description 11
- 238000004519 manufacturing process Methods 0.000 claims description 10
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 claims description 5
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical group OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 claims description 4
- 229940095064 tartrate Drugs 0.000 claims description 3
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 claims 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims 1
- 230000000694 effects Effects 0.000 abstract description 20
- 239000000126 substance Substances 0.000 abstract description 2
- 208000028018 Lymphocytic leukaemia Diseases 0.000 abstract 1
- 208000003747 lymphoid leukemia Diseases 0.000 abstract 1
- 210000004027 cell Anatomy 0.000 description 17
- 201000010099 disease Diseases 0.000 description 14
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 14
- 230000006907 apoptotic process Effects 0.000 description 12
- 230000007170 pathology Effects 0.000 description 12
- 208000004736 B-Cell Leukemia Diseases 0.000 description 11
- KDCGOANMDULRCW-UHFFFAOYSA-N 7H-purine Chemical compound N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 description 10
- 229960000390 fludarabine Drugs 0.000 description 10
- GIUYCYHIANZCFB-FJFJXFQQSA-N fludarabine phosphate Chemical compound C1=NC=2C(N)=NC(F)=NC=2N1[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@@H]1O GIUYCYHIANZCFB-FJFJXFQQSA-N 0.000 description 10
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 9
- 230000030833 cell death Effects 0.000 description 9
- 102100032857 Cyclin-dependent kinase 1 Human genes 0.000 description 7
- 101710106279 Cyclin-dependent kinase 1 Proteins 0.000 description 7
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 7
- 210000003719 b-lymphocyte Anatomy 0.000 description 7
- 230000032823 cell division Effects 0.000 description 7
- 230000002401 inhibitory effect Effects 0.000 description 7
- 210000004698 lymphocyte Anatomy 0.000 description 7
- 239000000203 mixture Substances 0.000 description 7
- 102000002427 Cyclin B Human genes 0.000 description 6
- 108010068150 Cyclin B Proteins 0.000 description 6
- 229910052757 nitrogen Inorganic materials 0.000 description 6
- 150000003891 oxalate salts Chemical class 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 108091000080 Phosphotransferase Proteins 0.000 description 5
- 102000020233 phosphotransferase Human genes 0.000 description 5
- 150000003212 purines Chemical class 0.000 description 5
- 230000004044 response Effects 0.000 description 5
- 230000000638 stimulation Effects 0.000 description 5
- AOPLJKWUEJZIPY-UHFFFAOYSA-N 2-chloro-9-propan-2-yl-n-[(4-pyridin-2-ylphenyl)methyl]purin-6-amine Chemical compound N1=C(Cl)N=C2N(C(C)C)C=NC2=C1NCC(C=C1)=CC=C1C1=CC=CC=N1 AOPLJKWUEJZIPY-UHFFFAOYSA-N 0.000 description 4
- 108010024986 Cyclin-Dependent Kinase 2 Proteins 0.000 description 4
- 108010025454 Cyclin-Dependent Kinase 5 Proteins 0.000 description 4
- 102100036239 Cyclin-dependent kinase 2 Human genes 0.000 description 4
- 102100026805 Cyclin-dependent-like kinase 5 Human genes 0.000 description 4
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 4
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 4
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 4
- 239000000427 antigen Substances 0.000 description 4
- 108091007433 antigens Proteins 0.000 description 4
- 102000036639 antigens Human genes 0.000 description 4
- 239000000872 buffer Substances 0.000 description 4
- 229960004397 cyclophosphamide Drugs 0.000 description 4
- 238000000034 method Methods 0.000 description 4
- 229960004641 rituximab Drugs 0.000 description 4
- -1 1,3-dihydroxyprop-2-ylamino Chemical group 0.000 description 3
- KJJPLEZQSCZCKE-UHFFFAOYSA-N 2-aminopropane-1,3-diol Chemical compound OCC(N)CO KJJPLEZQSCZCKE-UHFFFAOYSA-N 0.000 description 3
- NAMYKGVDVNBCFQ-UHFFFAOYSA-N 2-bromopropane Chemical compound CC(C)Br NAMYKGVDVNBCFQ-UHFFFAOYSA-N 0.000 description 3
- 241000282472 Canis lupus familiaris Species 0.000 description 3
- 102100024457 Cyclin-dependent kinase 9 Human genes 0.000 description 3
- 101000980930 Homo sapiens Cyclin-dependent kinase 9 Proteins 0.000 description 3
- 229960000548 alemtuzumab Drugs 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 230000018109 developmental process Effects 0.000 description 3
- VHJLVAABSRFDPM-QWWZWVQMSA-N dithiothreitol Chemical compound SC[C@@H](O)[C@H](O)CS VHJLVAABSRFDPM-QWWZWVQMSA-N 0.000 description 3
- DEFVIWRASFVYLL-UHFFFAOYSA-N ethylene glycol bis(2-aminoethyl)tetraacetic acid Chemical compound OC(=O)CN(CC(O)=O)CCOCCOCCN(CC(O)=O)CC(O)=O DEFVIWRASFVYLL-UHFFFAOYSA-N 0.000 description 3
- 210000003734 kidney Anatomy 0.000 description 3
- 208000017169 kidney disease Diseases 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- BXHXBVNRKRXSHM-UHFFFAOYSA-N (4-pyridin-2-ylphenyl)methanamine Chemical compound C1=CC(CN)=CC=C1C1=CC=CC=N1 BXHXBVNRKRXSHM-UHFFFAOYSA-N 0.000 description 2
- VZARATVQVHMJRO-UHFFFAOYSA-N 2-chloro-n-[(4-pyridin-2-ylphenyl)methyl]-7h-purin-6-amine Chemical compound C=12NC=NC2=NC(Cl)=NC=1NCC(C=C1)=CC=C1C1=CC=CC=N1 VZARATVQVHMJRO-UHFFFAOYSA-N 0.000 description 2
- 206010065553 Bone marrow failure Diseases 0.000 description 2
- 102000002554 Cyclin A Human genes 0.000 description 2
- 108010068192 Cyclin A Proteins 0.000 description 2
- 102000003909 Cyclin E Human genes 0.000 description 2
- 108090000257 Cyclin E Proteins 0.000 description 2
- 102000002435 Cyclin T Human genes 0.000 description 2
- 108010068106 Cyclin T Proteins 0.000 description 2
- 102100028554 Dual specificity tyrosine-phosphorylation-regulated kinase 1A Human genes 0.000 description 2
- 108010014905 Glycogen Synthase Kinase 3 Proteins 0.000 description 2
- 102000002254 Glycogen Synthase Kinase 3 Human genes 0.000 description 2
- 241000238631 Hexapoda Species 0.000 description 2
- 101000838016 Homo sapiens Dual specificity tyrosine-phosphorylation-regulated kinase 1A Proteins 0.000 description 2
- 101000878605 Homo sapiens Low affinity immunoglobulin epsilon Fc receptor Proteins 0.000 description 2
- 102000003855 L-lactate dehydrogenase Human genes 0.000 description 2
- 108700023483 L-lactate dehydrogenases Proteins 0.000 description 2
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 2
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 2
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 2
- 102100038007 Low affinity immunoglobulin epsilon Fc receptor Human genes 0.000 description 2
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical class OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 102000001253 Protein Kinase Human genes 0.000 description 2
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 2
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 2
- 238000009825 accumulation Methods 0.000 description 2
- 125000000218 acetic acid group Chemical group C(C)(=O)* 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 229940045686 antimetabolites antineoplastic purine analogs Drugs 0.000 description 2
- 239000012736 aqueous medium Substances 0.000 description 2
- 210000001185 bone marrow Anatomy 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 150000001733 carboxylic acid esters Chemical class 0.000 description 2
- 230000022131 cell cycle Effects 0.000 description 2
- 230000004663 cell proliferation Effects 0.000 description 2
- 230000003833 cell viability Effects 0.000 description 2
- JCKYGMPEJWAADB-UHFFFAOYSA-N chlorambucil Chemical compound OC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 JCKYGMPEJWAADB-UHFFFAOYSA-N 0.000 description 2
- 210000000349 chromosome Anatomy 0.000 description 2
- 208000020832 chronic kidney disease Diseases 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 230000006735 deficit Effects 0.000 description 2
- 235000019439 ethyl acetate Nutrition 0.000 description 2
- 108010049611 glycogen synthase kinase 3 alpha Proteins 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 230000002489 hematologic effect Effects 0.000 description 2
- 230000001506 immunosuppresive effect Effects 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 229940043355 kinase inhibitor Drugs 0.000 description 2
- 210000001165 lymph node Anatomy 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 125000004433 nitrogen atom Chemical group N* 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 239000003757 phosphotransferase inhibitor Substances 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 229940002612 prodrug Drugs 0.000 description 2
- 239000000651 prodrug Substances 0.000 description 2
- 238000004393 prognosis Methods 0.000 description 2
- 230000035755 proliferation Effects 0.000 description 2
- XJMOSONTPMZWPB-UHFFFAOYSA-M propidium iodide Chemical compound [I-].[I-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CCC[N+](C)(CC)CC)=C1C1=CC=CC=C1 XJMOSONTPMZWPB-UHFFFAOYSA-M 0.000 description 2
- 108060006633 protein kinase Proteins 0.000 description 2
- 125000004076 pyridyl group Chemical group 0.000 description 2
- UQDJGEHQDNVPGU-UHFFFAOYSA-N serine phosphoethanolamine Chemical compound [NH3+]CCOP([O-])(=O)OCC([NH3+])C([O-])=O UQDJGEHQDNVPGU-UHFFFAOYSA-N 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 235000002906 tartaric acid Nutrition 0.000 description 2
- 239000011975 tartaric acid Substances 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 239000004474 valine Substances 0.000 description 2
- MUVQIIBPDFTEKM-QWWZWVQMSA-N (2r,3r)-2-aminobutane-1,3-diol Chemical compound C[C@@H](O)[C@H](N)CO MUVQIIBPDFTEKM-QWWZWVQMSA-N 0.000 description 1
- ZGCHLAJIRWDGFE-UHFFFAOYSA-N 1-aminopropane-1,1-diol Chemical compound CCC(N)(O)O ZGCHLAJIRWDGFE-UHFFFAOYSA-N 0.000 description 1
- YQGHJCYLMLPCCB-UHFFFAOYSA-N 2,4-diaminopyrimidin-5-ol Chemical compound NC1=NC=C(O)C(N)=N1 YQGHJCYLMLPCCB-UHFFFAOYSA-N 0.000 description 1
- RMFWVOLULURGJI-UHFFFAOYSA-N 2,6-dichloro-7h-purine Chemical compound ClC1=NC(Cl)=C2NC=NC2=N1 RMFWVOLULURGJI-UHFFFAOYSA-N 0.000 description 1
- XDLJIIILKATPAQ-UHFFFAOYSA-N 2,8-dichloro-7h-purine Chemical compound ClC1=NC=C2NC(Cl)=NC2=N1 XDLJIIILKATPAQ-UHFFFAOYSA-N 0.000 description 1
- GKRXRQZDDJHENX-UHFFFAOYSA-N 2-[[9-propan-2-yl-6-[(4-pyridin-2-ylphenyl)methylamino]purin-2-yl]amino]propane-1,3-diol Chemical compound N1=C(NC(CO)CO)N=C2N(C(C)C)C=NC2=C1NCC(C=C1)=CC=C1C1=CC=CC=N1 GKRXRQZDDJHENX-UHFFFAOYSA-N 0.000 description 1
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- ASNHGEVAWNWCRQ-UHFFFAOYSA-N 4-(hydroxymethyl)oxolane-2,3,4-triol Chemical compound OCC1(O)COC(O)C1O ASNHGEVAWNWCRQ-UHFFFAOYSA-N 0.000 description 1
- XZKIHKMTEMTJQX-UHFFFAOYSA-N 4-Nitrophenyl Phosphate Chemical compound OP(O)(=O)OC1=CC=C([N+]([O-])=O)C=C1 XZKIHKMTEMTJQX-UHFFFAOYSA-N 0.000 description 1
- APRZHQXAAWPYHS-UHFFFAOYSA-N 4-[5-[3-(carboxymethoxy)phenyl]-3-(4,5-dimethyl-1,3-thiazol-2-yl)tetrazol-3-ium-2-yl]benzenesulfonate Chemical compound S1C(C)=C(C)N=C1[N+]1=NC(C=2C=C(OCC(O)=O)C=CC=2)=NN1C1=CC=C(S([O-])(=O)=O)C=C1 APRZHQXAAWPYHS-UHFFFAOYSA-N 0.000 description 1
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 1
- 241000258957 Asteroidea Species 0.000 description 1
- 102100022005 B-lymphocyte antigen CD20 Human genes 0.000 description 1
- 208000018240 Bone Marrow Failure disease Diseases 0.000 description 1
- HOCBJBNQIQQQGT-UHFFFAOYSA-N CCC(CO)Nc1nc(NCc(cc2)ccc2-c2ccccn2)c2nc[n](C(C)C)c2n1 Chemical compound CCC(CO)Nc1nc(NCc(cc2)ccc2-c2ccccn2)c2nc[n](C(C)C)c2n1 HOCBJBNQIQQQGT-UHFFFAOYSA-N 0.000 description 1
- 101150047910 CSNK1D gene Proteins 0.000 description 1
- 102000011727 Caspases Human genes 0.000 description 1
- 108010076667 Caspases Proteins 0.000 description 1
- 102000002495 Cyclin H Human genes 0.000 description 1
- 108010068237 Cyclin H Proteins 0.000 description 1
- 102100026810 Cyclin-dependent kinase 7 Human genes 0.000 description 1
- 241000701022 Cytomegalovirus Species 0.000 description 1
- 206010011831 Cytomegalovirus infection Diseases 0.000 description 1
- 206010061818 Disease progression Diseases 0.000 description 1
- 102100040862 Dual specificity protein kinase CLK1 Human genes 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 102000001267 GSK3 Human genes 0.000 description 1
- 108060006662 GSK3 Proteins 0.000 description 1
- 108010051975 Glycogen Synthase Kinase 3 beta Proteins 0.000 description 1
- 102100022975 Glycogen synthase kinase-3 alpha Human genes 0.000 description 1
- 102100038104 Glycogen synthase kinase-3 beta Human genes 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- 108010033040 Histones Proteins 0.000 description 1
- 102000006947 Histones Human genes 0.000 description 1
- 101000897405 Homo sapiens B-lymphocyte antigen CD20 Proteins 0.000 description 1
- 101000911952 Homo sapiens Cyclin-dependent kinase 7 Proteins 0.000 description 1
- 101000749294 Homo sapiens Dual specificity protein kinase CLK1 Proteins 0.000 description 1
- 108700029228 Immunoglobulin Heavy Chain Genes Proteins 0.000 description 1
- 208000019693 Lung disease Diseases 0.000 description 1
- 239000007993 MOPS buffer Substances 0.000 description 1
- 101150024075 Mapk1 gene Proteins 0.000 description 1
- 208000001388 Opportunistic Infections Diseases 0.000 description 1
- 208000033759 Prolymphocytic T-Cell Leukemia Diseases 0.000 description 1
- 108010090931 Proto-Oncogene Proteins c-bcl-2 Proteins 0.000 description 1
- 102000013535 Proto-Oncogene Proteins c-bcl-2 Human genes 0.000 description 1
- 206010037660 Pyrexia Diseases 0.000 description 1
- 206010040047 Sepsis Diseases 0.000 description 1
- 206010042970 T-cell chronic lymphocytic leukaemia Diseases 0.000 description 1
- 208000026651 T-cell prolymphocytic leukemia Diseases 0.000 description 1
- 102000006601 Thymidine Kinase Human genes 0.000 description 1
- 108020004440 Thymidine kinase Proteins 0.000 description 1
- VWQVUPCCIRVNHF-OUBTZVSYSA-N Yttrium-90 Chemical compound [90Y] VWQVUPCCIRVNHF-OUBTZVSYSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- AWUCVROLDVIAJX-UHFFFAOYSA-N alpha-glycerophosphate Natural products OCC(O)COP(O)(O)=O AWUCVROLDVIAJX-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 108700000711 bcl-X Proteins 0.000 description 1
- 102000055104 bcl-X Human genes 0.000 description 1
- 239000012267 brine Substances 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 230000000711 cancerogenic effect Effects 0.000 description 1
- 231100000357 carcinogen Toxicity 0.000 description 1
- 239000003183 carcinogenic agent Substances 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 230000006037 cell lysis Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 229940000425 combination drug Drugs 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 238000010835 comparative analysis Methods 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 229940125890 compound Ia Drugs 0.000 description 1
- 238000012217 deletion Methods 0.000 description 1
- 230000037430 deletion Effects 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 230000005750 disease progression Effects 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000009093 first-line therapy Methods 0.000 description 1
- 238000000684 flow cytometry Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-L fumarate(2-) Chemical class [O-]C(=O)\C=C\C([O-])=O VZCYOOQTPOCHFL-OWOJBTEDSA-L 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 208000035474 group of disease Diseases 0.000 description 1
- 231100000226 haematotoxicity Toxicity 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 150000003840 hydrochlorides Chemical class 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 238000003674 kinase activity assay Methods 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 229940126601 medicinal product Drugs 0.000 description 1
- NFVJNJQRWPQVOA-UHFFFAOYSA-N n-[2-chloro-5-(trifluoromethyl)phenyl]-2-[3-(4-ethyl-5-ethylsulfanyl-1,2,4-triazol-3-yl)piperidin-1-yl]acetamide Chemical compound CCN1C(SCC)=NN=C1C1CN(CC(=O)NC=2C(=CC=C(C=2)C(F)(F)F)Cl)CCC1 NFVJNJQRWPQVOA-UHFFFAOYSA-N 0.000 description 1
- 210000000287 oocyte Anatomy 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 230000007918 pathogenicity Effects 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- CMPQUABWPXYYSH-UHFFFAOYSA-N phenyl phosphate Chemical compound OP(O)(=O)OC1=CC=CC=C1 CMPQUABWPXYYSH-UHFFFAOYSA-N 0.000 description 1
- 229940080469 phosphocellulose Drugs 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 235000015320 potassium carbonate Nutrition 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 238000011363 radioimmunotherapy Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- BTIHMVBBUGXLCJ-OAHLLOKOSA-N seliciclib Chemical compound C=12N=CN(C(C)C)C2=NC(N[C@@H](CO)CC)=NC=1NCC1=CC=CC=C1 BTIHMVBBUGXLCJ-OAHLLOKOSA-N 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- AWUCVROLDVIAJX-GSVOUGTGSA-N sn-glycerol 3-phosphate Chemical compound OC[C@@H](O)COP(O)(O)=O AWUCVROLDVIAJX-GSVOUGTGSA-N 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- CMZUMMUJMWNLFH-UHFFFAOYSA-N sodium metavanadate Chemical compound [Na+].[O-][V](=O)=O CMZUMMUJMWNLFH-UHFFFAOYSA-N 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 238000001665 trituration Methods 0.000 description 1
- 229910000166 zirconium phosphate Inorganic materials 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
- A61K31/52—Purines, e.g. adenine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/02—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6
- C07D473/16—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 two nitrogen atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Epidemiology (AREA)
- Oncology (AREA)
- Virology (AREA)
- Communicable Diseases (AREA)
- Hematology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Urology & Nephrology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0706390A FR2920776B1 (fr) | 2007-09-12 | 2007-09-12 | Utilisation de derives de purine pour la fabrication d'un medicament |
| FR0706390 | 2007-09-12 | ||
| PCT/FR2008/001278 WO2009068761A2 (fr) | 2007-09-12 | 2008-09-12 | Utilisation de derives de purine pour la fabrication d'un medicament |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| RU2010108505A RU2010108505A (ru) | 2011-10-20 |
| RU2500400C2 true RU2500400C2 (ru) | 2013-12-10 |
Family
ID=39382006
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2010108505/15A RU2500400C2 (ru) | 2007-09-12 | 2008-09-12 | Применение производных пурина для изготовления лекарственного препарата |
Country Status (13)
| Country | Link |
|---|---|
| US (1) | US8431583B2 (enExample) |
| EP (1) | EP2187889B1 (enExample) |
| JP (1) | JP5485893B2 (enExample) |
| KR (1) | KR20100075908A (enExample) |
| CN (1) | CN101918000B (enExample) |
| BR (1) | BRPI0816810A2 (enExample) |
| CA (1) | CA2699590A1 (enExample) |
| DK (1) | DK2187889T3 (enExample) |
| ES (1) | ES2444419T3 (enExample) |
| FR (1) | FR2920776B1 (enExample) |
| MX (1) | MX2010002680A (enExample) |
| RU (1) | RU2500400C2 (enExample) |
| WO (1) | WO2009068761A2 (enExample) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2920776B1 (fr) | 2007-09-12 | 2012-09-28 | Centre Nat Rech Scient | Utilisation de derives de purine pour la fabrication d'un medicament |
| WO2010103473A1 (en) * | 2009-03-10 | 2010-09-16 | Chu De Brest | Method of treatment of polycystic diseases and chronic lymphocytic leukemia |
| WO2013130461A1 (en) * | 2012-02-29 | 2013-09-06 | The Scripps Research Institute | Wee1 degradation inhibitors |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998005335A1 (en) * | 1996-08-02 | 1998-02-12 | Cv Therapeutics, Inc. | PURINE INHIBITORS OF CYCLIN DEPENDENT KINASE 2 AND IλB-$g(a) |
| WO2003002565A1 (en) * | 2001-06-27 | 2003-01-09 | Cyclacel Limited | 2,6,9-substituted purine derivatives and their use n the treatment of proliferative disorders |
| WO2003022805A2 (en) * | 2001-09-11 | 2003-03-20 | Albany Molecular Research, Inc. | Heterocycle substituted purines as antiproliferative agents |
| WO2005016528A2 (en) * | 2003-08-15 | 2005-02-24 | Irm Llc | 6-substituted anilino purines as rtk inhibitors |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6627633B2 (en) * | 1999-03-17 | 2003-09-30 | Albany Molecular Research, Inc. | 6-substituted biaryl purine derivatives as potent cyclin/CDK inhibitors and antiproliferative agents |
| US6969720B2 (en) * | 1999-03-17 | 2005-11-29 | Amr Technology, Inc. | Biaryl substituted purine derivatives as potent antiproliferative agents |
| US6667311B2 (en) * | 2001-09-11 | 2003-12-23 | Albany Molecular Research, Inc. | Nitrogen substituted biaryl purine derivatives as potent antiproliferative agents |
| GB0315259D0 (en) | 2003-06-30 | 2003-08-06 | Cyclacel Ltd | Use |
| FR2920776B1 (fr) | 2007-09-12 | 2012-09-28 | Centre Nat Rech Scient | Utilisation de derives de purine pour la fabrication d'un medicament |
-
2007
- 2007-09-12 FR FR0706390A patent/FR2920776B1/fr active Active
-
2008
- 2008-09-12 KR KR1020107007886A patent/KR20100075908A/ko not_active Ceased
- 2008-09-12 WO PCT/FR2008/001278 patent/WO2009068761A2/fr not_active Ceased
- 2008-09-12 CN CN2008801067270A patent/CN101918000B/zh not_active Expired - Fee Related
- 2008-09-12 MX MX2010002680A patent/MX2010002680A/es active IP Right Grant
- 2008-09-12 BR BRPI0816810A patent/BRPI0816810A2/pt not_active IP Right Cessation
- 2008-09-12 JP JP2010524542A patent/JP5485893B2/ja not_active Expired - Fee Related
- 2008-09-12 CA CA2699590A patent/CA2699590A1/fr not_active Abandoned
- 2008-09-12 ES ES08855583.4T patent/ES2444419T3/es active Active
- 2008-09-12 RU RU2010108505/15A patent/RU2500400C2/ru not_active IP Right Cessation
- 2008-09-12 US US12/675,945 patent/US8431583B2/en not_active Expired - Fee Related
- 2008-09-12 DK DK08855583.4T patent/DK2187889T3/da active
- 2008-09-12 EP EP08855583.4A patent/EP2187889B1/fr not_active Not-in-force
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998005335A1 (en) * | 1996-08-02 | 1998-02-12 | Cv Therapeutics, Inc. | PURINE INHIBITORS OF CYCLIN DEPENDENT KINASE 2 AND IλB-$g(a) |
| WO2003002565A1 (en) * | 2001-06-27 | 2003-01-09 | Cyclacel Limited | 2,6,9-substituted purine derivatives and their use n the treatment of proliferative disorders |
| WO2003022805A2 (en) * | 2001-09-11 | 2003-03-20 | Albany Molecular Research, Inc. | Heterocycle substituted purines as antiproliferative agents |
| WO2005016528A2 (en) * | 2003-08-15 | 2005-02-24 | Irm Llc | 6-substituted anilino purines as rtk inhibitors |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2444419T3 (es) | 2014-02-25 |
| KR20100075908A (ko) | 2010-07-05 |
| DK2187889T3 (da) | 2014-02-03 |
| BRPI0816810A2 (pt) | 2017-05-16 |
| CA2699590A1 (fr) | 2009-06-04 |
| WO2009068761A3 (fr) | 2010-01-07 |
| RU2010108505A (ru) | 2011-10-20 |
| FR2920776A1 (fr) | 2009-03-13 |
| CN101918000B (zh) | 2012-11-21 |
| EP2187889A2 (fr) | 2010-05-26 |
| CN101918000A (zh) | 2010-12-15 |
| US20100311768A1 (en) | 2010-12-09 |
| EP2187889B1 (fr) | 2013-11-06 |
| JP2010539146A (ja) | 2010-12-16 |
| JP5485893B2 (ja) | 2014-05-07 |
| MX2010002680A (es) | 2010-08-26 |
| FR2920776B1 (fr) | 2012-09-28 |
| WO2009068761A2 (fr) | 2009-06-04 |
| US8431583B2 (en) | 2013-04-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11851445B2 (en) | Compounds and uses thereof | |
| JP2000501408A (ja) | 特に抗増殖性を有する新規プリン誘導体およびそれらの生物学的用途 | |
| WO2021022163A2 (en) | Compounds and uses thereof | |
| US20180207170A1 (en) | Methods and compositions useful for treating diseases involving bcl-2 family proteins with isoquinoline and quinoline derivatives | |
| EP2640718B1 (de) | Substituiertes natrium-1h-pyrazol-5-olat | |
| RU2500400C2 (ru) | Применение производных пурина для изготовления лекарственного препарата | |
| US20220280492A1 (en) | Therapeutic agent for cancer having resistance to anti-ccr4 antibody | |
| US11427574B2 (en) | Compounds for treating Rac-GTPase mediated disorder | |
| EP3921308B1 (en) | Sodium-hydrogen exchanger 3 inhibitor compounds | |
| US12215106B2 (en) | Pharmaceutical compounds and therapeutic methods | |
| WO2014059305A1 (en) | Compounds for treating rac-gtpase mediated disorder | |
| Hirschberg | Thiaxanthenones: Miracil D and hycanthone | |
| RU2526902C1 (ru) | Средство, обладающее кардиопротекторным действием, и галогениды 1,3-дизамещенных 2-аминобензимидазолия | |
| WO2016206097A1 (zh) | 新的5型磷酸二酯酶抑制剂及其应用 | |
| WO2024118830A1 (en) | Atropisomers of mat2a inhibitors | |
| WO2007074871A1 (ja) | 新規cxcr4拮抗剤及びその用途 | |
| CN106279164A (zh) | 新的5型磷酸二酯酶抑制剂及其应用 | |
| HK40066190A (en) | Therapeutic agent for cancer having resistance to anti-ccr4 antibody | |
| HK40064283B (en) | Sodium-hydrogen exchanger 3 inhibitor compounds | |
| JPWO2017175859A1 (ja) | 乳酸アシドーシスの予防又は治療のための医薬 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| MM4A | The patent is invalid due to non-payment of fees |
Effective date: 20180913 |