KR930003750B1 - Process for manufacturing magnesium hydroxide - Google Patents
Process for manufacturing magnesium hydroxide Download PDFInfo
- Publication number
- KR930003750B1 KR930003750B1 KR1019870015179A KR870015179A KR930003750B1 KR 930003750 B1 KR930003750 B1 KR 930003750B1 KR 1019870015179 A KR1019870015179 A KR 1019870015179A KR 870015179 A KR870015179 A KR 870015179A KR 930003750 B1 KR930003750 B1 KR 930003750B1
- Authority
- KR
- South Korea
- Prior art keywords
- magnesium hydroxide
- reaction
- synthesizing
- magnesia
- hours
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01F—COMPOUNDS OF THE METALS BERYLLIUM, MAGNESIUM, ALUMINIUM, CALCIUM, STRONTIUM, BARIUM, RADIUM, THORIUM, OR OF THE RARE-EARTH METALS
- C01F5/00—Compounds of magnesium
- C01F5/14—Magnesium hydroxide
- C01F5/16—Magnesium hydroxide by treating magnesia, e.g. calcined dolomite, with water or solutions of salts not containing magnesium
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01F—COMPOUNDS OF THE METALS BERYLLIUM, MAGNESIUM, ALUMINIUM, CALCIUM, STRONTIUM, BARIUM, RADIUM, THORIUM, OR OF THE RARE-EARTH METALS
- C01F5/00—Compounds of magnesium
- C01F5/14—Magnesium hydroxide
Abstract
내용 없음.No content.
Description
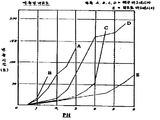
제1도는 샘플 A,B,C,D,E에 대한 반응성을 비교한 그래프.1 is a graph comparing the reactivity for samples A, B, C, D, E.
본원 발명은 천연으로 산출되는 마그네사이트(MgCO3)를 소성시켜서 경소(輕燒)마그네시아(MgO)로 한 것을 물과의 소화(消話)반응에 있어서, 고활성 히드로겔형 수산화마그네슘(이하 "히드로 마그네시아"라 함)을 합성하는 방법에 관한 것이다. 또한, 경소마그네시아와 물과의 반응으로 수산화마그네슘을 합성하는 것을 소화(消話)라고 한다. 상기 경소마그네시아는 대부분이 중국에서 생산되어 수입되고 있는 것이다. 원광(原鑛)은 마그네사이트(MgCO3)를 주성분으로 하고, 불순물로서는 헤레브란다이트(Ca2SiO4·H2O) 및 미량의 포르스테라이트(Mg2SiO4)를 함유하고 있다.The present invention is a high activity hydrogel-type magnesium hydroxide (hereinafter referred to as "hydromagnesia) in the digestion reaction with water to calcinate naturally produced magnesite (MgCO 3 ) to form a light magnesium (MgO). "," Is referred to. In addition, digestion is called synthesis of magnesium hydroxide by reaction of light magnesium with water. Most of the small magnesia is produced and imported from China. Ore contains magnesite (MgCO 3 ) as a main component and contains impurities such as herebrandite (Ca 2 SiO 4 · H 2 O) and trace amounts of forsterite (Mg 2 SiO 4 ).
이 원광을 800℃ 주변에서 소성함으로써 마그네사이트는 페리클레이스(periclase)형 MgO로 되고, 불순물의 헤레브란다이트는 랄사이트로 변하고, 포르스테라이트는 대략 이전의 상태로 남아 있다. 이 소성광석을 미분쇄(微粉碎)한 것을 경소마그네시아라고 부른다.By firing this ore at around 800 ° C, the magnesite turns into periclase type MgO, the herbbrandite of impurities turns to ralsite, and forsterite remains in its approximately previous state. The fine pulverization of this calcined ore is called light magnesium.
일본국에 있어서, 이 경소마그네시아는 고토(苦土)비료의 원료로써 수입되고 있다. 이 경소마그네시아는 MgO이므로 알칼리제로서 사용할 수 있고, 특히 배연(排煙) 탈황용의 알칼리제로서 사용할 수 있으면 현재 일본국에서 해수로부터 생산하고 있는 해수마그네시아의 생산코스트의 절반 이하로 공급할 수 있으며, 막대한 코스트절감 및 에너지절감이 될 것이다. 그러므로, 많은 사람이들이 상기 알칼리제로서의 사용을 시도하고 있었다.In Japan, this light magnesia is imported as a raw material of goto fertilizer. Since the light magnesium is MgO, it can be used as an alkaline agent, especially if it can be used as an alkaline agent for flue gas desulfurization, it can supply less than half of the production coast of seawater magnesia currently produced from seawater in Japan, It will be saving and energy saving. Therefore, many people have tried to use it as the alkali agent.
그러나, 상기 경소마그네시아는 알칼리제로서의 반응특성이 나쁜 것이다. 즉, ① 물에 녹지 않는다. ②물과의 반응성(소화)이 느리다. ③ 소화마그네시아는 해수마그네시아에 비해 반응성이 나쁘다, 등으로 인해 여러가지의 개질(改質)이 시도되었으나, 반응특성을 높일 수는 없었다. 이것은 경소마그네시아의 페리클레이스형 결정구조(Mg원자가 6개의 O원자에 둘러싸인 밀(密)패킹구조)에 의한 숙명적인 것이라고 해석되고 있었다.However, the light magnesia is poor in reaction characteristics as an alkaline agent. That is, ① insoluble in water. ② Reactivity (digestion) with water is slow. ③ Digested magnesia is less responsive than seawater magnesia, and various modifications have been attempted, but the reaction characteristics could not be improved. This has been interpreted to be fatal due to the pericle type crystal structure of small and small magnesia (a wheat packing structure in which Mg atoms are surrounded by six O atoms).
그러나, 발명자는 예의 연구의 결과, 페리클레이스형 구조의 해열(解裂)은 액의 OH-이온농도를 NaOH등을 가하여 높이는 것이며, 6개의 O원자중 4∼5개가 OH-로 변하고, 1∼2개의 O원자는 프리라디칼형의 활성원자로 되는 것을 발견하였다.However, as a result of intensive research, the inventors have found that the antipyretic structure of the pericle-type structure increases the OH - ion concentration of the liquid by adding NaOH, and 4 to 5 of the 6 O atoms are changed to OH − , Two O atoms were found to be active radicals of the free radical type.
이 프리라디칼원자를 가지므로, 반응성은 극단적으로 커지고, 해수마그네시아의 4∼5배의 반응속도 상수를 가지고 있다. 특히 pH가 높은 곳에서의 반응속도가 크다.Since it has this free radical atom, the reactivity becomes extremely large and has a reaction rate constant of 4 to 5 times that of seawater magnesia. In particular, the reaction rate is high at high pH.
히드로마그네시아의 합성에 있어서는, 히드로화의 반응속도를 높이기 위해 합성액 온도를 85℃이상, 바람직하게는 95℃이상으로 하고, 페리클레이스구조의 해열을 위해 가성소다 등의 강알칼리제를 가하고, 액의 pH를 상온에서 측정해서 11이상, 바람직하게는 12이상으로 한다. 또한, 반응시간은 20분 이상 5시간 정도, 바람직하게는 1시간∼3시간 정도로 하고, 충분히 교반하여 반응을 완결시킨다.In the synthesis of hydromagnesia, in order to increase the reaction rate of hydrolysis, the temperature of the synthesis liquid is set to 85 ° C or higher, preferably 95 ° C or higher, and a strong alkaline agent such as caustic soda is added to dissolve the pericle structure. Is measured at room temperature to 11 or more, preferably 12 or more. The reaction time is 20 minutes or more and about 5 hours, preferably 1 hour to 3 hours, and the mixture is sufficiently stirred to complete the reaction.
해열 반응을 개시하는 것은 예를들면 합성액 온도 95℃에 있어서, 합성개시후 10분 정도에서 시작되고, 60분내에 90%는 진행한다.The initiation of the antipyretic reaction starts at about 10 minutes after the start of the synthesis, for example, at a synthesis liquid temperature of 95 ° C., and 90% proceeds within 60 minutes.
다음에, 본원 발명의 실시예에 대하여 상세히 설명한다.Next, the Example of this invention is described in detail.
철제의 교반기가 부착된 15㎥반응조에 공업용수 10㎥를 채워 교반하면서 스팀으로 액온 50℃까지 가온한 후, 스팀을 정지한다. 이것에 플레이크형 가성소다 30㎏을 가하여 충분히 용해시킨다. 이어서, 경소(輕燒)마그네시아 4000㎏을 가하여 혼합 교반하면 20∼30분 내에 반응조 액온 100℃로 된다. 그 온도가 내려가지 않도록 스팀으로 온도조정을 하면서, 2∼3시간 반응시켜 반응을 종료한다. 합성액은 냉각후 히드로마그네시아로서 제품으로 한다.After filling 10 ㎥ of industrial water into a 15 ㎥ reaction tank equipped with an iron stirrer and stirring, the solution was heated with steam to 50 ° C., and then the steam was stopped. 30 kg of flake caustic soda is added to this, and it fully melt | dissolves. Subsequently, when 4000 kg of light magnesia is added and mixed and stirred, it becomes a reaction tank liquid temperature of 100 degreeC within 20-30 minutes. The reaction is completed by reacting for 2 to 3 hours while adjusting the temperature with steam so that the temperature does not decrease. The synthetic solution is cooled down to hydromagnesia as a product.
히드로마그네시아는 해수마그네시아와 다르고, 극단적으로 점성이 낮고 고농도 슬러리(45%)로서 운반 및 저장도 할 수 있다. 이 히드로마그네시아는 배탈용(排脫用) 알칼리제로서는 정상기한 바와 같이 코스트절감과 에너지절감에 대단한 공헌을 하는 것이다.Hydromagnesia differs from seawater magnesia and is extremely low viscosity and can be transported and stored as a high concentration slurry (45%). This hydromagnesia makes a significant contribution to cost reduction and energy reduction, as is the normal term as an alkaline agent for upset stomach.
다음에, 본원 발명 실시예의 히드로마그네시아와 각 회사제 해수마그네시아와의 각 물성 및 반응성의 비교측정치를 표 1 및 표 2로 나타낸다.Next, Table 1 and Table 2 show comparative measurements of the physical properties and reactivity of the hydromagnesia of Examples of the present invention with seawater magnesia from each company.
그리고, 반응성의 비교에 대해서는 다시 알기 쉽게 그래프로 나타냈다.In addition, the comparison of reactivity was shown by the graph again clearly.
샘플 A 해수마그네시아(A회사제)Sample A seawater magnesia (product made in A company)
" B " (A회사제)"B" (A company)
" C " (A회사제)"C" (A company)
" D " (S회사제)"D" (S company made)
" E 히드로마그네시아(본원 발명 실시예)"E hydromagnesia (inventive examples herein)
[표 1]TABLE 1
[표 2]TABLE 2
(반응성)(Reactivity)
* 측정조건 : 점 도-B형 점도계/NO. 2로터 사용* Measurement condition: Viscosity-B viscometer / NO. 2 rotor use
액온=약 20℃Liquid temperature = approximately 20 degrees Celsius
점도분포-세디그래프/용매=물Viscosity Distribution-Cedigraph / Solvent = Water
액온=34∼35℃Liquid temperature = 34-35 degrees Celsius
진비중=2.41Heavy weight = 2.41
반응성-2N 황산 200cc(0.2mol)와의 반응Reaction with 200 cc (0.2 mol) of reactive-2N sulfuric acid
반응시 액온=약 45∼50℃Liquid temperature at the time of reaction = about 45-50 degreeC
샘플의 농도=0.208molConcentration of sample = 0.208mol
Claims (5)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP62184584A JPS6428214A (en) | 1987-07-23 | 1987-07-23 | Synthesis of active magnesium hydroxide |
| JP184584 | 1987-07-23 | ||
| JP62-184584 | 1987-07-23 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR890001876A KR890001876A (en) | 1989-04-06 |
| KR930003750B1 true KR930003750B1 (en) | 1993-05-10 |
Family
ID=16155766
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1019870015179A KR930003750B1 (en) | 1987-07-23 | 1987-12-29 | Process for manufacturing magnesium hydroxide |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JPS6428214A (en) |
| KR (1) | KR930003750B1 (en) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01294520A (en) * | 1988-01-13 | 1989-11-28 | Sankurei:Kk | Method for slaking naturally occurring light burned magnesia |
| JPH0363741U (en) * | 1989-10-26 | 1991-06-21 | ||
| JPH03151543A (en) * | 1989-11-08 | 1991-06-27 | Mazda Motor Corp | Idle revolution controller of engine |
| KR0163963B1 (en) * | 1992-10-29 | 1998-11-16 | 시미즈 고우헤이 | Method for the production of active magnesium hydroxide |
| CN103115837B (en) * | 2013-01-05 | 2015-05-20 | 江苏苏博特新材料股份有限公司 | Method for testing content of magnesium oxide in magnesium oxide expanding agent |
| WO2015058236A1 (en) | 2013-10-24 | 2015-04-30 | Calix Ltd | Process and apparatus for manufacture of hydroxide slurry |
-
1987
- 1987-07-23 JP JP62184584A patent/JPS6428214A/en active Granted
- 1987-12-29 KR KR1019870015179A patent/KR930003750B1/en not_active IP Right Cessation
Also Published As
| Publication number | Publication date |
|---|---|
| JPH0360774B2 (en) | 1991-09-17 |
| KR890001876A (en) | 1989-04-06 |
| JPS6428214A (en) | 1989-01-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102583409B (en) | Method for producing mullite and calcium silicate by using high-alumina fly ash | |
| US3953584A (en) | Process for the preparation of basic aluminum chlorides | |
| KR930003750B1 (en) | Process for manufacturing magnesium hydroxide | |
| CN103274632A (en) | Reinforced cement grinding aid and preparation method thereof | |
| US2996369A (en) | Zirconium oxide and process of making the same | |
| AU607920B2 (en) | Preparation of hydrolysed zirconium salt precursors and high purity zirconia | |
| US4271130A (en) | Process for the preparation of zeolite A from kaolin | |
| CN109761256A (en) | It is a kind of to prepare high activity porous C a (OH) with shell2Method | |
| US4065396A (en) | Magnesium oxide process | |
| CN113716588B (en) | Low-cost preparation method of magnesium-aluminum hydrotalcite | |
| KR20190052632A (en) | Method for preparing Sodium Antimonate | |
| CN114609338A (en) | Method for detecting activity of red mud | |
| JPH06191832A (en) | Preparation of activated magnesium hydroxide | |
| US2393080A (en) | Charge preparation | |
| US1530139A (en) | Art of manufacturing zirconium compounds | |
| KR930001256B1 (en) | Process for manufacturing active magnesium hydrate | |
| CN117049581A (en) | Preparation method of light calcium carbonate for food | |
| CN109336146A (en) | A kind of method that flyash alkali soluble improves pre-desiliconizing rate | |
| CN109627742B (en) | Method for preparing polyurethane board from sintering flue gas desulfurization ash | |
| JPS6186421A (en) | Preparation of white electroconductive powder | |
| US2859100A (en) | Process of extracting alumina from raw calcium aluminates | |
| CN114408935B (en) | Preparation method of high-modulus lithium silicate aqueous solution | |
| CN100378004C (en) | Method for producing potassium stannate | |
| JPH0231007B2 (en) | ITAJOTANSAN KARUSHIUMUNOSEIZOHO | |
| US4447351A (en) | Preparation of alumina hydrosol from aluminum sulfate solution |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| E902 | Notification of reason for refusal | ||
| G160 | Decision to publish patent application | ||
| E701 | Decision to grant or registration of patent right | ||
| GRNT | Written decision to grant | ||
| FPAY | Annual fee payment |
Payment date: 20020329 Year of fee payment: 10 |
|
| LAPS | Lapse due to unpaid annual fee |