KR20200082006A - Extended release formulation containing tofacitinib or pharmaceutically acceptable salts thereof as an active ingredient and the preparation method for the same - Google Patents
Extended release formulation containing tofacitinib or pharmaceutically acceptable salts thereof as an active ingredient and the preparation method for the same Download PDFInfo
- Publication number
- KR20200082006A KR20200082006A KR1020180172103A KR20180172103A KR20200082006A KR 20200082006 A KR20200082006 A KR 20200082006A KR 1020180172103 A KR1020180172103 A KR 1020180172103A KR 20180172103 A KR20180172103 A KR 20180172103A KR 20200082006 A KR20200082006 A KR 20200082006A
- Authority
- KR
- South Korea
- Prior art keywords
- sustained
- tofacitinib
- sustained release
- salt
- release preparation
- Prior art date
Links
- 239000004012 Tofacitinib Substances 0.000 title claims abstract description 49
- 229960001350 tofacitinib Drugs 0.000 title claims abstract description 46
- UJLAWZDWDVHWOW-YPMHNXCESA-N tofacitinib Chemical compound C[C@@H]1CCN(C(=O)CC#N)C[C@@H]1N(C)C1=NC=NC2=C1C=CN2 UJLAWZDWDVHWOW-YPMHNXCESA-N 0.000 title claims abstract description 46
- 239000000203 mixture Substances 0.000 title claims abstract description 35
- 238000009472 formulation Methods 0.000 title claims abstract description 27
- 150000003839 salts Chemical class 0.000 title claims abstract description 20
- 238000002360 preparation method Methods 0.000 title claims abstract description 13
- 239000004480 active ingredient Substances 0.000 title description 10
- 238000013265 extended release Methods 0.000 title description 2
- 239000003405 delayed action preparation Substances 0.000 claims abstract description 54
- 238000013268 sustained release Methods 0.000 claims abstract description 39
- 239000012730 sustained-release form Substances 0.000 claims abstract description 39
- 239000011230 binding agent Substances 0.000 claims abstract description 33
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 21
- 239000011159 matrix material Substances 0.000 claims abstract description 12
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 claims description 26
- 239000001863 hydroxypropyl cellulose Substances 0.000 claims description 26
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 claims description 26
- 229920000036 polyvinylpyrrolidone Polymers 0.000 claims description 24
- 239000001267 polyvinylpyrrolidone Substances 0.000 claims description 24
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 claims description 24
- 229940079593 drug Drugs 0.000 claims description 18
- 239000003814 drug Substances 0.000 claims description 18
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 claims description 15
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 claims description 15
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 claims description 15
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 claims description 14
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 claims description 14
- 238000004090 dissolution Methods 0.000 claims description 13
- WSVLPVUVIUVCRA-KPKNDVKVSA-N Alpha-lactose monohydrate Chemical compound O.O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O WSVLPVUVIUVCRA-KPKNDVKVSA-N 0.000 claims description 11
- 229960001021 lactose monohydrate Drugs 0.000 claims description 8
- 238000000034 method Methods 0.000 claims description 8
- 238000002156 mixing Methods 0.000 claims description 8
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical class OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 claims description 7
- 239000002202 Polyethylene glycol Substances 0.000 claims description 7
- 239000003085 diluting agent Substances 0.000 claims description 7
- 235000019359 magnesium stearate Nutrition 0.000 claims description 7
- 229920001223 polyethylene glycol Polymers 0.000 claims description 7
- 229920000168 Microcrystalline cellulose Polymers 0.000 claims description 6
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 6
- 229920000609 methyl cellulose Polymers 0.000 claims description 6
- 239000001923 methylcellulose Substances 0.000 claims description 6
- 235000010981 methylcellulose Nutrition 0.000 claims description 6
- 229940016286 microcrystalline cellulose Drugs 0.000 claims description 6
- 235000019813 microcrystalline cellulose Nutrition 0.000 claims description 6
- 239000008108 microcrystalline cellulose Substances 0.000 claims description 6
- 239000001856 Ethyl cellulose Substances 0.000 claims description 5
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 claims description 5
- 150000001860 citric acid derivatives Chemical class 0.000 claims description 5
- 235000019325 ethyl cellulose Nutrition 0.000 claims description 5
- 229920001249 ethyl cellulose Polymers 0.000 claims description 5
- 239000000314 lubricant Substances 0.000 claims description 5
- 229920002261 Corn starch Polymers 0.000 claims description 4
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 claims description 4
- 229930006000 Sucrose Natural products 0.000 claims description 4
- 239000008120 corn starch Substances 0.000 claims description 4
- -1 cyclam salt salts Chemical class 0.000 claims description 4
- 239000005720 sucrose Substances 0.000 claims description 4
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims description 4
- OMDQUFIYNPYJFM-XKDAHURESA-N (2r,3r,4s,5r,6s)-2-(hydroxymethyl)-6-[[(2r,3s,4r,5s,6r)-4,5,6-trihydroxy-3-[(2s,3s,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxan-2-yl]methoxy]oxane-3,4,5-triol Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1OC[C@@H]1[C@@H](O[C@H]2[C@H]([C@@H](O)[C@H](O)[C@@H](CO)O2)O)[C@H](O)[C@H](O)[C@H](O)O1 OMDQUFIYNPYJFM-XKDAHURESA-N 0.000 claims description 3
- 229920000858 Cyclodextrin Polymers 0.000 claims description 3
- 229920000926 Galactomannan Polymers 0.000 claims description 3
- 108010010803 Gelatin Proteins 0.000 claims description 3
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 claims description 3
- 239000004354 Hydroxyethyl cellulose Substances 0.000 claims description 3
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 claims description 3
- 239000002253 acid Substances 0.000 claims description 3
- 239000001768 carboxy methyl cellulose Substances 0.000 claims description 3
- 239000003086 colorant Substances 0.000 claims description 3
- 229940097362 cyclodextrins Drugs 0.000 claims description 3
- MOTZDAYCYVMXPC-UHFFFAOYSA-N dodecyl hydrogen sulfate Chemical class CCCCCCCCCCCCOS(O)(=O)=O MOTZDAYCYVMXPC-UHFFFAOYSA-N 0.000 claims description 3
- 235000003599 food sweetener Nutrition 0.000 claims description 3
- 239000008273 gelatin Substances 0.000 claims description 3
- 229920000159 gelatin Polymers 0.000 claims description 3
- 235000019322 gelatine Nutrition 0.000 claims description 3
- 235000011852 gelatine desserts Nutrition 0.000 claims description 3
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 claims description 3
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 claims description 3
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 claims description 3
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical class CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 claims description 3
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical class CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 claims description 3
- PXQPEWDEAKTCGB-UHFFFAOYSA-N orotic acid Chemical class OC(=O)C1=CC(=O)NC(=O)N1 PXQPEWDEAKTCGB-UHFFFAOYSA-N 0.000 claims description 3
- 239000001814 pectin Substances 0.000 claims description 3
- 229920001277 pectin Polymers 0.000 claims description 3
- 235000010987 pectin Nutrition 0.000 claims description 3
- 239000011734 sodium Substances 0.000 claims description 3
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 claims description 3
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 claims description 3
- 239000003765 sweetening agent Substances 0.000 claims description 3
- 150000002688 maleic acid derivatives Chemical class 0.000 claims description 2
- 150000002823 nitrates Chemical class 0.000 claims description 2
- 239000003381 stabilizer Substances 0.000 claims description 2
- 150000003467 sulfuric acid derivatives Chemical class 0.000 claims description 2
- 239000004094 surface-active agent Substances 0.000 claims description 2
- 150000001558 benzoic acid derivatives Chemical class 0.000 claims 1
- 150000002942 palmitic acid derivatives Chemical class 0.000 claims 1
- 125000002467 phosphate group Chemical class [H]OP(=O)(O[H])O[*] 0.000 claims 1
- 150000003890 succinate salts Chemical class 0.000 claims 1
- 150000008054 sulfonate salts Chemical class 0.000 claims 1
- 239000012729 immediate-release (IR) formulation Substances 0.000 abstract description 15
- 238000001727 in vivo Methods 0.000 abstract description 10
- 238000004519 manufacturing process Methods 0.000 abstract description 10
- 230000000694 effects Effects 0.000 abstract description 6
- 230000000052 comparative effect Effects 0.000 description 7
- 206010009900 Colitis ulcerative Diseases 0.000 description 6
- 201000001263 Psoriatic Arthritis Diseases 0.000 description 6
- 208000036824 Psoriatic arthropathy Diseases 0.000 description 6
- 201000006704 Ulcerative Colitis Diseases 0.000 description 6
- 239000000654 additive Substances 0.000 description 6
- 229940009098 aspartate Drugs 0.000 description 6
- 238000010828 elution Methods 0.000 description 6
- 239000000499 gel Substances 0.000 description 6
- 206010039073 rheumatoid arthritis Diseases 0.000 description 6
- 239000008187 granular material Substances 0.000 description 5
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 4
- 229920003114 HPC-L Polymers 0.000 description 4
- 229920003093 Methocel™ K100 LV Polymers 0.000 description 4
- 229920003081 Povidone K 30 Polymers 0.000 description 4
- 235000012054 meals Nutrition 0.000 description 4
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 3
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- 235000010980 cellulose Nutrition 0.000 description 3
- 229920002678 cellulose Polymers 0.000 description 3
- 239000001913 cellulose Substances 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 238000004128 high performance liquid chromatography Methods 0.000 description 3
- 230000003204 osmotic effect Effects 0.000 description 3
- 230000002265 prevention Effects 0.000 description 3
- SYIKUFDOYJFGBQ-YLAFAASESA-N tofacitinib citrate Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O.C[C@@H]1CCN(C(=O)CC#N)C[C@@H]1N(C)C1=NC=NC2=C1C=CN2 SYIKUFDOYJFGBQ-YLAFAASESA-N 0.000 description 3
- 229940039916 xeljanz Drugs 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 229940072107 ascorbate Drugs 0.000 description 2
- 235000010323 ascorbic acid Nutrition 0.000 description 2
- 239000011668 ascorbic acid Substances 0.000 description 2
- 229940050390 benzoate Drugs 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 239000007888 film coating Substances 0.000 description 2
- 238000009501 film coating Methods 0.000 description 2
- IPCSVZSSVZVIGE-UHFFFAOYSA-M hexadecanoate Chemical compound CCCCCCCCCCCCCCCC([O-])=O IPCSVZSSVZVIGE-UHFFFAOYSA-M 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 229960001375 lactose Drugs 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 229940049964 oleate Drugs 0.000 description 2
- 230000035699 permeability Effects 0.000 description 2
- 239000008057 potassium phosphate buffer Substances 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 229940114926 stearate Drugs 0.000 description 2
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- GHCZTIFQWKKGSB-UHFFFAOYSA-N 2-hydroxypropane-1,2,3-tricarboxylic acid;phosphoric acid Chemical compound OP(O)(O)=O.OC(=O)CC(O)(C(O)=O)CC(O)=O GHCZTIFQWKKGSB-UHFFFAOYSA-N 0.000 description 1
- 208000024827 Alzheimer disease Diseases 0.000 description 1
- 208000011231 Crohn disease Diseases 0.000 description 1
- UDIPTWFVPPPURJ-UHFFFAOYSA-M Cyclamate Chemical compound [Na+].[O-]S(=O)(=O)NC1CCCCC1 UDIPTWFVPPPURJ-UHFFFAOYSA-M 0.000 description 1
- 206010012438 Dermatitis atopic Diseases 0.000 description 1
- 208000002249 Diabetes Complications Diseases 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 206010062016 Immunosuppression Diseases 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 229920000881 Modified starch Polymers 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- MHQJUHSHQGQVTM-HNENSFHCSA-N Octadecyl fumarate Chemical compound CCCCCCCCCCCCCCCCCCOC(=O)\C=C/C(O)=O MHQJUHSHQGQVTM-HNENSFHCSA-N 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 102000001253 Protein Kinase Human genes 0.000 description 1
- 201000004681 Psoriasis Diseases 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 239000004376 Sucralose Substances 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 206010067584 Type 1 diabetes mellitus Diseases 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- GZCGUPFRVQAUEE-SLPGGIOYSA-N aldehydo-D-glucose Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O GZCGUPFRVQAUEE-SLPGGIOYSA-N 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 201000008937 atopic dermatitis Diseases 0.000 description 1
- 208000010928 autoimmune thyroid disease Diseases 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 229940109275 cyclamate Drugs 0.000 description 1
- HCAJEUSONLESMK-UHFFFAOYSA-N cyclohexylsulfamic acid Chemical class OS(=O)(=O)NC1CCCCC1 HCAJEUSONLESMK-UHFFFAOYSA-N 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000012738 dissolution medium Substances 0.000 description 1
- 229940043264 dodecyl sulfate Drugs 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 229960003943 hypromellose Drugs 0.000 description 1
- 239000012728 immediate-release (IR) tablet Substances 0.000 description 1
- 230000001506 immunosuppresive effect Effects 0.000 description 1
- 229960003444 immunosuppressant agent Drugs 0.000 description 1
- 230000001861 immunosuppressant effect Effects 0.000 description 1
- 239000003018 immunosuppressive agent Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 229910000358 iron sulfate Inorganic materials 0.000 description 1
- BAUYGSIQEAFULO-UHFFFAOYSA-L iron(2+) sulfate (anhydrous) Chemical compound [Fe+2].[O-]S([O-])(=O)=O BAUYGSIQEAFULO-UHFFFAOYSA-L 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 206010025135 lupus erythematosus Diseases 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 201000006417 multiple sclerosis Diseases 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 230000000291 postprandial effect Effects 0.000 description 1
- 230000003449 preventive effect Effects 0.000 description 1
- 108060006633 protein kinase Proteins 0.000 description 1
- 238000004513 sizing Methods 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 229940032147 starch Drugs 0.000 description 1
- 229940071138 stearyl fumarate Drugs 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- BAQAVOSOZGMPRM-QBMZZYIRSA-N sucralose Chemical compound O[C@@H]1[C@@H](O)[C@@H](Cl)[C@@H](CO)O[C@@H]1O[C@@]1(CCl)[C@@H](O)[C@H](O)[C@@H](CCl)O1 BAQAVOSOZGMPRM-QBMZZYIRSA-N 0.000 description 1
- 235000019408 sucralose Nutrition 0.000 description 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 1
- 150000003871 sulfonates Chemical class 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 239000007916 tablet composition Substances 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 238000002054 transplantation Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2072—Pills, tablets, discs, rods characterised by shape, structure or size; Tablets with holes, special break lines or identification marks; Partially coated tablets; Disintegrating flat shaped forms
- A61K9/2077—Tablets comprising drug-containing microparticles in a substantial amount of supporting matrix; Multiparticulate tablets
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
Abstract
Description
본 발명은 토파시티닙 또는 그의 약제학적으로 허용되는 염을 함유한 서방성 제제 및 그의 제조방법에 관한 것이다. 구체적으로는, 토파시티닙 또는 그의 약제학적으로 허용되는 염 및 서방화제로 친수성 기제를 포함하는 서방성 제제에 관한 것이다. 이러한 서방성 제제는 생체내에서 약물이 안정적이면서 지속적으로 방출되어 1일 1회 복용만으로도 원하는 치료 효과를 얻을 수 있다. 보다 특별하게는, 현재 시판중인 서방성 제품보다 적은 함량 즉, 10mg으로도 생체내에서 속방성 제제와 동등한 효과를 나타낼 수 있으며, 그 제조 과정 또한 시판 중인 서방성 제품에 비해 간단하여 생산성 및 경제성 측면에서도 장점이 있다는 것을 특징으로 한다. The present invention relates to a sustained release preparation containing tofacitinib or a pharmaceutically acceptable salt thereof and a method for preparing the same. Specifically, it relates to a sustained release preparation comprising a hydrophilic base as tofacitinib or a pharmaceutically acceptable salt and sustained release agent thereof. Such sustained-release preparations can stably and continuously release the drug in vivo, thereby obtaining a desired therapeutic effect by taking only once a day. More specifically, a content lower than that of a commercially available sustained release product, i.e., 10 mg, may have an effect equivalent to that of an immediate release preparation in vivo, and the manufacturing process is also simpler than that of a commercially available sustained release product. It is also characterized by having an advantage.
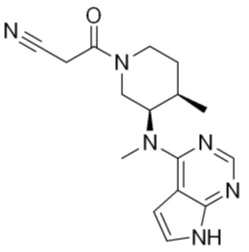
본 발명의 유효성분인 토파시티닙(Tofacitinib)은 화합물명이 3-{(3R,4R)-4-메틸-3-[메틸-(7H-피롤로[2,3-d]피리미딘-4-일)-아미노]-피페리딘-1-일}-3-옥소-프로피오니트릴으로 하기 화학식 1로 표시되는 구조를 가진다. Tofacitinib, an active ingredient of the present invention, has a compound name of 3-{(3R,4R)-4-methyl-3-[methyl-(7H-pyrrolo[2,3-d]pyrimidine-4- 1)-Amino]-piperidin-1-yl}-3-oxo-propionitrile, and has a structure represented by Formula 1 below.
상기 화학식 1로 표시되는 토파시티닙은 국제공개특허 WO2001/042246호에 야누스 키나아제(Janus Kinas)3 (JAK3) 효소와 같은 단백질 키나아제의 저해 활성을 갖는 것으로 개시되어 있고, 장기 이식, 이종 장기 이식, 루푸스, 다발성 경화증, 류마티스 관절염, 건선, 건선성 관절염, I형 당뇨병 및 당뇨합병증, 암, 천식, 아토피성 피부염, 자가면역 갑상선 질환, 궤양성 대장염, 크론병, 알츠하이머병, 백혈병 및 면역억제가 필요한 다른 질병에 대한 면역억제제로서 유용한 것으로 공지되어 있다. Tofacitinib represented by Chemical Formula 1 is disclosed in International Publication No. WO2001/042246 as having inhibitory activity of protein kinase, such as Janus Kinas3 (JAK3) enzyme, organ transplantation, xenograft, Lupus, multiple sclerosis, rheumatoid arthritis, psoriasis, psoriatic arthritis, type I diabetes and diabetes complications, cancer, asthma, atopic dermatitis, autoimmune thyroid disease, ulcerative colitis, Crohn's disease, Alzheimer's disease, leukemia and immunosuppression It is known to be useful as an immunosuppressant for other diseases.
또한, 상기 화학식 1 및 그 약제학적으로 허용가능한 염의 제조방법은 국제공개특허WO2002/096909에 개시되어 있으며, 현재 국내에서 시판되고 있는 젤잔즈®(Xeljanz)정의 주성분인 토파시티닙 시트레이트염은 국제공개특허 WO2003/048162에 구체적으로 개시되어 있다. 젤잔즈®(Xeljanz)정은 류마티스 관절염, 건선성 관절염 및 궤양성 대장염 치료제로, 토파시티닙 시트레이트염을 5mg 함유하는 속방성 제제(1일 2회 복용)이다. In addition, the formula I and their pharmaceutically acceptable salt thereof is disclosed in International Patent Publication WO2002 / 096909, gel janjeu ® (Xeljanz) defining a main component topa City nip citrate salt are currently commercially available in Korea, International It is specifically disclosed in published patent WO2003/048162. Gel is janjeu ® (Xeljanz) Chung rheumatoid arthritis, psoriatic arthritis or ulcerative colitis to treatment, topa City nip sheet in immediate release formulation containing the citrate salt 5mg (1 twice-daily dose).
통상 서방성 제제(extended release form)는 속방성(immediate release form) 제제에 비해 체내에서 일정한 시간 동안 약물을 지속적으로 방출하기 때문에, 약물의 유효혈중농도를 장기간 유지시켜 속방성 제제를 자주 투여하여 발생하는 혈중농도의 진폭을 감소시키고 그에 따라 부작용도 줄일 수 있으며, 투여빈도를 줄임으로써 환자의 복약순응도(compliance)를 향상시킬 수 있다. 또한, 토파시티닙과 같이 낮은 투여량이 요구되는 약물을 정제로 제조하는 경우 유효성분에 비해 첨가제의 양이 많기 때문에 개별 정제마다 유효성분의 함량 균일성을 확보하는데 어려움이 있어서 보다 많은 함량이 함유되어 함량 균일성 확보가 더 유리하다는 측면에서도 서방성 제제가 요구될 수 있다. Usually, extended release form releases the drug for a certain period of time in the body compared to immediate release form, so it occurs by maintaining the effective blood concentration of the drug for a long time and frequently administering the immediate release agent. By reducing the amplitude of the blood concentration and reducing side effects accordingly, it is possible to improve patient compliance by reducing the frequency of administration. In addition, when a drug requiring a low dosage such as tofacitinib is prepared as a tablet, since the amount of the additive is greater than that of the active ingredient, it is difficult to ensure uniformity of the content of the active ingredient for each individual tablet, and thus, a higher content is contained. Sustained-release preparations may also be required in terms of securing more uniformity of content.
토파시티닙을 유효성분으로 하는 서방성 제제에 관해서는 국제공개특허 WO2012/100949에 토파시티닙이 방출 조절 제제로 제제화될 수 있다고 언급되어 있으며, 국제공개특허 WO2014/147526에 매트릭스 시스템(정제 또는 다중입자), 삼투 시스템, 저장소 시스템 등 통상 서방성 제제를 제조하는 기술을 이용하여 토파시티닙을 유효성분으로 하는 서방성 제제를 제조할 수 있다고 언급하면서 삼투 시스템을 이용한 압출성 코어 시스템을 대표적인 예로 기재하고 있다. 그러나, 토파시티닙의 경우 방출 지속시간이 연장됨에 따라 생체이용률이 감소되어 국제공개특허 WO2012/100949와 같이 1일 투여용량인 10mg으로 서방성 제제를 제조하면 속방성 제제와 약동학적 동등성을 확보할 수 없고, 이 문제를 해결하기 위해 초과 용량(11mg)으로 제조된 서방성 제제를 개시하고 있는 국제공개특허 WO2014/147526에서도 삼투성 압출성 코어 시스템을 이용한 제제의 경우에만 90% 신뢰구간(CI) 기준 범위 내에서 속방성 제제와 동등한 생체이용률을 보일 뿐 친수성 매트릭스 시스템의 경우에는 초과 용량을 함유하여도 동등한 생체이용률을 확보하지 못하였다. 실제 미국에서 젤잔즈®XR(Xeljanz XR)이라는 상품명으로 판매되고 있는 제품이 속방성 제제 1일 투여용량인 10mg을 초과하는 용량인 11mg을 함유하는 OROS(Osmotic Controlled-release Oral Delivery) 제형이다. 하지만, OROS 제형은 그 제조과정이 복잡하여 생산성 및 경제성이 떨어지는 단점이 있다. Regarding sustained-release preparations using tofacitinib as an active ingredient, it is mentioned in WO2012/100949 that tofacitinib can be formulated as a release-controlling agent, and matrix systems (tablets or multiples) are disclosed in WO2014/147526. Particles), osmotic systems, storage systems, etc., mentioning that it is possible to manufacture sustained-release preparations using tofacitinib as an active ingredient using techniques for preparing sustained-release preparations, and describe extruded core systems using osmotic systems as representative examples. Doing. However, in the case of tofacitinib, the bioavailability decreases as the release duration is extended, and thus, when a sustained release preparation is prepared at a daily dosage of 10 mg, as in International Publication Patent WO2012/100949, pharmacokinetic equivalence with the immediate release preparation is secured In order to solve this problem, the international publication patent WO2014/147526, which discloses a sustained-release preparation prepared in an excess dose (11mg), solves this problem. Within the reference range, the bioavailability equivalent to the immediate release formulation was shown, but in the case of the hydrophilic matrix system, the bioavailability equivalent to the excess dose was not secured. A (Osmotic Controlled-release Oral Delivery) OROS formulation of the product that is sold under the trade name of gel janjeu ® XR (XR Xeljanz) in the United States contains a physical dose of 11mg beyond the immediate release formulation daily dosage dose of 10mg. However, the OROS formulation has a disadvantage in that its manufacturing process is complicated and productivity and economic efficiency are poor.
이에, 본 발명자는 상기와 같은 문제를 해결하기 위하여 연구한 결과, 서방화제에 추가로 친수성 결합제를 사용하는 경우 속방성 제제 1일 투여용량만으로도 약물이 안정적이면서도 지속적인 방출을 나타내어 속방성 제제와 동등한 생체이용률을 확보할 수 있는 서방성 제제를 제조할 수 있다는 것을 확인하여, 본 발명을 완성하였다. Thus, the present inventors have studied in order to solve the above problems, and when using a hydrophilic binder in addition to the sustained release agent, the drug is stable and sustained release even with a daily dose of the immediate release preparation, so that the bioequivalent to the immediate release preparation It was confirmed that a sustained release formulation capable of securing a utilization rate could be prepared, and the present invention was completed.
본 발명은 류마티스 관절염, 건선성 관절염 및 궤양성 대장염 예방 또는 치료용 서방성 제제를 제공하는 것을 해결과제로 하며, 유효성분으로 토파시티닙을 포함하고, 서방화제에 추가로 친수성 결합제를 사용하여 유효성분이 친수성 매트릭스 내에 분산된 형태로 안정적이면서 지속적으로 방출되는 서방성 제제를 제공하는 것을 구체적인 해결과제로 한다. The present invention is to solve the problem of providing a sustained release preparation for the prevention or treatment of rheumatoid arthritis, psoriatic arthritis and ulcerative colitis, contains tofacitinib as an active ingredient, and is effective by using a hydrophilic binder in addition to a sustained release agent. A specific solution is to provide a sustained release formulation that is stable and continuously released in a form in which the powder is dispersed in a hydrophilic matrix.
나아가, 본 발명은 2종 이상의 친수성 기제를 사용하여 현재 시판중인 서방성 제품보다 토파시티닙 또는 그의 약제학적으로 허용되는 염의 함량이 낮음에도 불구하고 생체내에서 속방성 제제와 동등한 효과를 나타내고, 그 제조 과정이 간단하여 생산성 및 경제성이 우수한 친수성 매트릭스 제형의 서방성 제제를 제공하는 것을 본 발명의 특별한 해결과제로 한다. Furthermore, the present invention uses two or more hydrophilic bases and exhibits an equivalent effect to a rapid release preparation in vivo despite the lower content of tofacitinib or a pharmaceutically acceptable salt thereof than a commercially available sustained release product. It is a special problem of the present invention to provide a sustained release preparation of a hydrophilic matrix formulation that is excellent in productivity and economic efficiency due to a simple manufacturing process.
상기 과제를 해결하기 위하여, 본 발명에서는 하기와 같은 수단을 개시한다. In order to solve the above problems, the present invention discloses the following means.
일 양태에서, 본 발명은 토파시티닙 또는 그의 약제학적으로 허용되는 염, 서방화제 및 친수성 결합제를 포함하는 서방성 제제를 개시한다. In one aspect, the present invention discloses a sustained release preparation comprising tofacitinib or a pharmaceutically acceptable salt, sustained release agent and hydrophilic binder.
상기 서방화 제제에 있어서, 상기 토파시티닙 또는 그의 약제학적으로 허용되는 염은 시트레이트염, 아스파르트산염, 숙신산염, 오로트산염, 팔미트산염, 스테아르산염, 벤조산염, 아스코르빈산염, 올레산염, 술폰산염, 황산염, 도데실황산염, 사이클람산염, 에디실산염, 질산염, 말레산염 또는 인산염 중에서 선택될 수 있다. In the sustained release formulation, the tofacitinib or a pharmaceutically acceptable salt thereof is a citrate salt, aspartate, succinate, orotate, palmitate, stearate, benzoate, ascorbate, and oleate. Acid salts, sulfonates, sulfates, dodecyl sulfates, cyclamates, edisylates, nitrates, maleates or phosphates.
상기 서방성 제제에 있어서, 상기 서방화제는 약제학적으로 허용이 가능한 서방화 기제라면 모두 사용할 수 있으며, 바람직하게는 폴리비닐피롤리돈(PVP), 히드록시프로필메틸셀룰로오스(HPMC), 히드록시프로필메틸셀룰로오스프탈레이트, 메틸셀룰로오스(MC), 카르복시메틸셀룰로오스나트륨(CMC-Na), 히드록시에틸셀룰로오스, 히드록시프로필셀룰로오스(HPC), 펙틴, 시클로덱스트린류, 갈락토만난, 폴리에틸렌글리콜(PEG), 에틸셀룰로오스(EC) 및 젤라틴으로 구성된 그룹으로부터 선택된 1종 이상일 수 있다. 보다 바람직하게는 HPMC가 사용될 수 있다. In the sustained release preparation, the sustained release agent can be used as long as it is a pharmaceutically acceptable sustained release base, preferably polyvinylpyrrolidone (PVP), hydroxypropylmethylcellulose (HPMC), hydroxypropyl Methylcellulose phthalate, methylcellulose (MC), sodium carboxymethylcellulose (CMC-Na), hydroxyethylcellulose, hydroxypropylcellulose (HPC), pectin, cyclodextrins, galactomannan, polyethylene glycol (PEG), ethyl It may be one or more selected from the group consisting of cellulose (EC) and gelatin. More preferably, HPMC can be used.
상기 친수성 결합제는 약제학적으로 허용이 가능한 친수성 결합제라면 모두 사용할 수 있으며, 바람직하게는 미결정셀룰로오스, 슈크로오스, 산화티탄, 히드록시프로필셀룰로오스(HPC), 폴리비닐피롤리돈(PVP), 옥수수 전분 및 유당수화물로 구성된 그룹으로부터 선택된 1종 또는 2종 이상일 수 있다. 바람직하게는 HPC, PVP 또는 이들의 조합일 수 있다. The hydrophilic binder may be used as long as it is a pharmaceutically acceptable hydrophilic binder, preferably microcrystalline cellulose, sucrose, titanium oxide, hydroxypropyl cellulose (HPC), polyvinylpyrrolidone (PVP), corn starch. And it may be one or two or more selected from the group consisting of lactose hydrate. Preferably it may be HPC, PVP or a combination thereof.
상기 친수성 결합제의 2종 조합시, 선택된 2종의 혼합 비율이 1:4 내지 4: 1일 수 있다. When combining two types of the hydrophilic binder, the mixing ratio of the two selected types may be 1:4 to 4:1.
상기 서방성 제제에 있어서, 토파시티닙이 10mg 포함될 수 있다. In the sustained release preparation, tofacitinib may be included 10mg.
상기 서방성 제제에 있어서, 상기 친수성 결합제는 제제 총 중량 기준으로 5.0~20.0중량%로 포함될 수 있다. In the sustained release formulation, the hydrophilic binder may be included in an amount of 5.0 to 20.0 wt% based on the total weight of the formulation.
상기 서방성 제제에 있어서, 상기 토파시티닙은 친수성 매트릭스 내에 분산된 형태일 수 있다.In the sustained release formulation, the tofacitinib may be in a form dispersed in a hydrophilic matrix.
다른 양태에서, 본 발명은 토파시티닙 또는 그의 약제학적으로 허용되는 염; 서방화제로 히드록시프로필메틸셀룰로오스(HPMC); 친수성 결합제로 히드록시프로필셀룰로오스(HPC) 및 폴리비닐피롤리돈(PVP); 희석제로 유당 일수화물; 및 활택제로 스테아르산 마그네슘을 포함하는 서방성 제제를 개시한다. In another aspect, the invention provides tofacitinib or a pharmaceutically acceptable salt thereof; Hydroxypropyl methylcellulose (HPMC) as a sustained release agent; Hydroxypropyl cellulose (HPC) and polyvinylpyrrolidone (PVP) as hydrophilic binders; Lactose monohydrate as a diluent; And a sustained release formulation comprising magnesium stearate as a lubricant.
또 다른 양태에서, 본 발명은 주성분 토파시티닙 또는 그 약제학적으로 허용되는 염 및 기타 첨가제를 혼합하여 혼합물을 제조하는 단계; 상기 혼합물을 압축, 정립하여 과립화하는 단계; 상기 과립물을 타정하는 단계; 및 상기 나정을 코팅하는 단계를 포함하는 서방성 제제의 제조방법을 개시한다. In another aspect, the present invention comprises the steps of preparing a mixture by mixing the main component tofacitinib or a pharmaceutically acceptable salt and other additives thereof; Compressing and sizing the mixture to granulate; Tableting the granules; And it discloses a method for producing a sustained release preparation comprising the step of coating the tablet.
본 발명에 따른 서방성 제제는 류마티스 관절염, 건선성 관절염 및 궤양성 대장염 예방 또는 치료 효과를 나타낸다. 보다 특별하게는, 서방화제에 추가로 친수성 결합제를 사용함으로써 유효성분이 안정적이면서도 지속적인 방출을 나타내어 속방성 제제 1일 투여용량과 동일한 함량만으로도 동등한 생체이용률을 나타낼 수 있다. 특히, 시판 중인 OROS 제품에 비해 제조 과정이 비교적 간단하여 생산성 및 경제성이 우수한 서방성 제제를 제공할 수 있다. The sustained-release preparations according to the present invention show the prevention or treatment effect of rheumatoid arthritis, psoriatic arthritis and ulcerative colitis. More specifically, by using a hydrophilic binder in addition to a sustained release agent, the active ingredient exhibits a stable and sustained release, and thus, an equivalent bioavailability can be exhibited only with the same amount as the daily dosage of the immediate release preparation. In particular, compared to a commercially available OROS product, the manufacturing process is relatively simple, thereby providing a sustained release preparation excellent in productivity and economic efficiency.
도 1은 본 발명의 서방성 제제, 1종의 친수성 결합제만을 포함한 서방성 제제 및 시판 서방성 제품의 용출률을 비교한 그래프이다.
도 2는 본 발명의 서방성 제제 및 시판 속방성 제품의 PK 프로파일을 비교한 그래프이다.
도 3은 본 발명의 서방성 제제의 식전 vs 식후 PK 프로파일을 비교한 그래프이다. 1 is a graph comparing the dissolution rate of the sustained-release preparation of the present invention, a sustained-release preparation containing only one hydrophilic binder and a commercially available sustained-release product.
Figure 2 is a graph comparing the PK profile of the sustained-release preparations and commercially-released products of the present invention.
Figure 3 is a graph comparing the PK profile before and after meal of the sustained-release preparation of the present invention.
본 발명은, 기존 알려진 토파시티닙 서방성 제제의 경우 속방성 제제 1일 투여용량을 초과하는 함량을 함유하며, 제조과정이 복잡한 OROS 제형인데 반해, 서방화제에 추가로 친수성 결합제를 사용하여 친수성 매트릭스의 점도를 조절하면 속방성 제제 1일 투여용량과 동일한 함량만으로도 동등한 효과를 나타낼 수 있는 서방성 제제를 간단한 제조과정을 통해 제조할 수 있음에 기초하여 이루어진 것이다.In the present invention, in the case of the known tofacitinib sustained-release preparation, the content of the immediate-release preparation exceeds the daily dosage, and the manufacturing process is a complicated OROS formulation, whereas the hydrophilic matrix is added using a hydrophilic binder in addition to the sustained release agent. By adjusting the viscosity of the immediate-release preparation is made on the basis that the sustained-release preparation that can exhibit the same effect with only the same amount as the daily dosage is produced through a simple manufacturing process.
특히, 1종의 친수성 결합제만을 사용할 경우 용출이 너무 늦어지거나 초기 용출이 너무 빠르게 진행되는데 반해 2종 이상의 친수성 결합제 조합, 예를 들어 히드록시프로필셀룰로오스(HPC) 및 폴리비닐피롤리돈(PVP)를 조합하는 경우 친수성 매트릭스로부터 활성성분인 토파시티닙이 안정적이면서도 지속적으로 용출이 가능하다는 점에 기초한 것이다. In particular, when only one type of hydrophilic binder is used, the elution is too late or the initial elution proceeds too quickly, whereas a combination of two or more hydrophilic binders such as hydroxypropylcellulose (HPC) and polyvinylpyrrolidone (PVP) When combined, it is based on the fact that the active ingredient tofacitinib from the hydrophilic matrix is stable and can continuously elute.
본 발명에 따라, 류마티스 관절염, 건선성 관절염 및 궤양성 대장염 예방 또는 치료용 서방성 제제는 토파시티닙 또는 그의 약제학적으로 허용되는 염, 서방화제 및 친수성 결합제를 포함한다. According to the present invention, sustained-release preparations for the prevention or treatment of rheumatoid arthritis, psoriatic arthritis and ulcerative colitis include tofacitinib or a pharmaceutically acceptable salt thereof, a sustained release agent and a hydrophilic binder.
본 발명에 따르면, 미국약전(USP)에 따른 제2법 패들법(50rpm)에 따라 900 ml 인산칼륨 완충액으로 pH 6.8 및 37℃ 조건에서 적절한 시간에 얻어진 용출액을 여과한 후 고성능 액체 크로마토그래피(HPLC)에 의해 측정하였을 때, 본 발명의 서방성 제제는 2시간에서의 제제로부터의 약물 용출률이 45% 이하, 3시간에서의 제제로부터의 약물 용출률이 55% 이상 80% 이하이다. According to the present invention, high-performance liquid chromatography (HPLC) after filtering the eluate obtained at a suitable time at pH 6.8 and 37° C. with 900 ml potassium phosphate buffer according to the second method paddle method (50 rpm) according to the United States Pharmacopeia (USP) When measured by ), the sustained release formulation of the present invention has a drug dissolution rate from the formulation at 2 hours of 45% or less, and a drug dissolution rate from the formulation at 3 hours of 55% or more and 80% or less.
본 발명의 사용가능한 토파시티닙은 토파시티닙 유리 염기 또는 약제학적으로 허용가능한 그의 산부가염(이하, 명세서에서는, 특별히 다르게 표시되지 않는 한, 토파시티닙 또는 그의 약제학적으로 허용되는 염을 '토파시티닙'로 통칭한다)이 포함될 수 있으며, 예를 들어, 시트레이트염, 아스파르트산염, 숙신산염, 오로트산염, 팔미트산염, 스테아르산염, 벤조산염, 아스코르빈산염, 올레산염, 술폰산염, 황산염, 도데실황산염, 사이클람산염, 에디실산염, 질산염, 말레산염, 인산염을 들 수 있으며, 이 중에서 시트레이트염이 가장 바람직하다. The tofacitinib usable in the present invention is tofacitinib free base or a pharmaceutically acceptable acid addition salt (hereinafter, unless indicated otherwise, in the specification, tofacitinib or a pharmaceutically acceptable salt thereof is referred to as'topa Cytinib'), for example, citrate salt, aspartate, Succinate, orotate, palmitate, stearate, benzoate, ascorbate, oleate, sulfonate, sulfate, dodecyl sulfate, cyclamate, edisylate, nitrate, maleate, phosphate Citrate salt is the most preferred.
본 발명의 서방성 제제에 사용되는 서방화제는 약제학적으로 허용이 가능한 서방화기제라면 모두 사용할 수 있으며, 바람직하게는 폴리비닐피롤리돈(PVP), 셀룰로오스폴리머 예를 들어, 히드록시프로필메틸셀룰로오스(HPMC), 히드록시프로필메틸셀룰로오스프탈레이트, 메틸셀룰로오스(MC), 카르복시메틸셀룰로오스나트륨(CMC-Na), 히드록시에틸셀룰로오스 또는 히드록시프로필셀룰로오스(HPC), 펙틴, 시클로덱스트린류, 갈락토만난, 폴리에틸렌글리콜(PEG), 에틸셀룰로오스(EC) 및 젤라틴으로 구성된 그룹으로부터 선택된 1종 이상일 수 있다. 보다 바람직하게는 HPMC가 사용될 수 있다. The sustained-release agent used in the sustained-release preparation of the present invention can be used as long as it is a pharmaceutically acceptable sustained-release agent, preferably polyvinylpyrrolidone (PVP), a cellulose polymer, for example, hydroxypropylmethylcellulose (HPMC), hydroxypropylmethylcellulose phthalate, methylcellulose (MC), sodium carboxymethylcellulose (CMC-Na), hydroxyethylcellulose or hydroxypropylcellulose (HPC), pectin, cyclodextrins, galactomannan, It may be one or more selected from the group consisting of polyethylene glycol (PEG), ethyl cellulose (EC) and gelatin. More preferably, HPMC can be used.
상기 친수성 결합제는 약제학적으로 허용이 가능한 친수성 결합제라면 모두 사용할 수 있으며, 바람직하게는 미결정셀룰로오스, 슈크로오스, 산화티탄, 히드록시프로필셀룰로오스(HPC), 폴리비닐피롤리돈(PVP), 옥수수 전분 및 유당수화물로 구성된 그룹으로부터 선택된 1종 또는 2종 이상일 수 있다. 바람직하게는 HPC, PVP 등이고, 가장 바람직하게는 HPC와 PVP의 조합일 수 있다. 특히, 저점도의 HPC를 사용하면 더욱 바람직한 서방 효과를 얻을 수 있고, 바람직하게는 2% 수용액을 20℃로 Brookfield viscometer 이용하여 측정한 점도가 6.0-10.0 mPa*s이다. The hydrophilic binder may be used as long as it is a pharmaceutically acceptable hydrophilic binder, preferably microcrystalline cellulose, sucrose, titanium oxide, hydroxypropyl cellulose (HPC), polyvinylpyrrolidone (PVP), corn starch. And it may be one or two or more selected from the group consisting of lactose hydrate. Preferably it is HPC, PVP, and the like, and most preferably, it may be a combination of HPC and PVP. Particularly, when a low viscosity HPC is used, a more preferable sustained effect can be obtained, and the viscosity measured by using a 2% aqueous solution at 20°C using a Brookfield viscometer is 6.0-10.0 mPa*s.
본 발명의 서방성 제제는 친수성 매트릭스 내에 토파시티닙이 분산된 형태로, 생체 내의 소화관에서 수용성 매질과 친수성 매트릭스가 반응하여 정제의 바깥 외층부터 겔층의 막을 형성하며, 이 겔층의 막을 통해서 토파시티닙이 일정 시간 동안 서서히 방출된다. 본 발명의 서방성 제제는 서방화제에 추가로 친수성 결합제를 사용하여 안정적이면서 일정한 속도로 유효성분인 토파시티닙이 방출하는 것이 가능하다. The sustained release preparation of the present invention is a form in which tofacitinib is dispersed in a hydrophilic matrix, and a hydrophilic matrix reacts with a water-soluble medium in the digestive tract in vivo to form a gel layer film from the outer outer layer of the tablet, and through this gel layer film, tofacitinib It is released slowly over a period of time. The sustained-release preparation of the present invention is capable of releasing topacitinib as an active ingredient at a stable and constant rate by using a hydrophilic binder in addition to a sustained release agent.
상기 친수성 결합제는 제제 총 중량 기준으로 5.0~20.0중량%로 포함될 수 있으며, 바람직하게는 10.0~17.5중량%로 포함될 수 있다. 친수성 결합제가 5.0중량% 미만인 경우 지속적인 용출이 가능하도록 친수성 매트릭스의 점도를 조절하기에 부족하고, 20.0중량%를 초과하는 경우 비경제적이고, 친수성 결합제의 과도한 양으로 인해 과립의 제조 공정상 문제를 일으킬 소지가 있으며, 단위 제형의 전체 중량이 커져 복용에 불편을 초래하기 때문이다. 또한, 상기 친수성 결합제를 2종 이상 혼합하는 경우 그 혼합 비율은 1∼4 : 4∼1가 바람직하다. 특히, 그 혼합 비율이 2:3이 가장 바람직하다. The hydrophilic binder may be included in 5.0 to 20.0% by weight based on the total weight of the formulation, preferably 10.0 to 17.5% by weight. When the hydrophilic binder is less than 5.0% by weight, it is insufficient to adjust the viscosity of the hydrophilic matrix to enable continuous elution, and when it exceeds 20.0% by weight, it is uneconomical, and excessive amounts of the hydrophilic binder may cause problems in the manufacturing process of granules. This is because the total weight of the unit dosage form increases, causing inconvenience to taking. Moreover, when mixing 2 or more types of the said hydrophilic binder, the mixing ratio is preferably 1-4:4-1. In particular, the mixing ratio is most preferably 2:3.
본 발명의 서방성 제제는 추가적으로 약제학적으로 허용되는 첨가제, 예를 들어 희석제, 착색제, 감미제, 계면활성제, 활택제, 안정화제 등을 포함할 수 있다. The sustained release preparations of the present invention may additionally include pharmaceutically acceptable additives such as diluents, colorants, sweeteners, surfactants, lubricants, stabilizers, and the like.
상기 희석제는 유당류, 미결정 셀룰로오스류, 전분류 등을 포함할 수 있으며, [0036] 구체적으로 유당류에는 유당 일수화물, 유당 무수물, 분무건조 유당 일수화물 등이 있고, 미결정 셀룰오로스류에는 미결정 셀룰로오스, 실리케이트화 미결정 셀룰로오스 등이 있으며, 전분류에는 옥수수전분, 전호화 전분 등이 있으나 이에 한정되지는 않는다. The diluent may include lactose, microcrystalline cellulose, starch, etc., specifically, lactose includes lactose monohydrate, lactose anhydride, spray dried lactose monohydrate, etc., and microcrystalline cellulose Cellulose, silicate microcrystalline cellulose, and the like, corn starch, pregelatinized starch, and the like, but are not limited thereto.
상기 착색제로는 산화티탄, 황산산화철 또는 FD&C에서 추천된 색소 중에서 선택된 1종 이상이 포함될 수 있다. The colorant may include one or more selected from titanium oxide, iron sulfate, or pigments recommended by FD&C.
상기 감미제로는 수크랄로스, 수크로스, 덱스트로스, 과당, 포도당, 액체포도당 또는 말토스 중에서 선택된 1종 이상이 포함될 수 있다. The sweetener may include one or more selected from sucralose, sucrose, dextrose, fructose, glucose, liquid glucose or maltose.
상기 활택제로는 탈크, 스테아릴푸마레이트, 스테아르산마그네슘 또는 수소화 피마자유 등이 사용될 수 있다. As the lubricant, talc, stearyl fumarate, magnesium stearate, or hydrogenated castor oil may be used.
본 발명의 서방성 제제는 약제학적으로 사용 가능한 필름 코팅을 포함할 수 있다. 상기 필름 코팅은 히프로멜로오스(hyperomellose), 이산화티타늄(titanium dioxide), PEG, 산화철(iron oxide), 탈크(talc) 등의 일반적으로 경구용 제제의 코팅에 사용되는 첨가제들 중에서 1종 이상을 선택하여 사용할 수 있다.The sustained release formulation of the present invention may include a pharmaceutically usable film coating. The film coating is one or more of the additives commonly used in the coating of oral preparations such as hypromellose, titanium dioxide, PEG, iron oxide, and talc. You can choose and use.
본 발명의 서방성 제제는 다양한 제형으로 제조될 수 있다. 예를 들어 나정, 코팅정, 다층정 또는 유핵정 등의 정제, 분말제, 과립제 또는 캡슐제 등으로 제형화될 수 있으며, 바람직하게는 정제일 수 있다. The sustained release preparations of the present invention can be prepared in various dosage forms. For example, it may be formulated into tablets, powders, granules or capsules, such as tablets, coated tablets, multilayer tablets or nucleated tablets, and preferably tablets.
본 발명의 서방성 제제는 약 5 내지 15mg의 토파시티닙, 바람직하게는 10mg의 토파시티닙을 포함할 수 있으며, 투여 경로가 적절하게 조절될 수 있으나 바람직하게는 경구 투여될 수 있다. The sustained release preparations of the present invention may include about 5 to 15 mg of tofacitinib, preferably 10 mg of tofacitinib, and the route of administration may be appropriately controlled, but may preferably be administered orally.
본 발명은 또한 서방성 제제의 제조 방법을 제공한다. 한 구체예로서, 본 발명의 서방성 제제의 제조 방법은 The present invention also provides a method for preparing a sustained release preparation. In one embodiment, the method of preparing the sustained release formulation of the present invention
(1) 주성분 토파시티닙 또는 그 약제학적으로 허용되는 염 및 기타 첨가제를 혼합하여 혼합물을 제조하는 단계;(1) mixing the main component tofacitinib or a pharmaceutically acceptable salt and other additives to prepare a mixture;
(2) 상기 혼합물을 압축, 정립하여 과립화하는 단계;(2) compacting and granulating the mixture;
(3) 상기 과립물을 타정하는 단계; 및(3) tableting the granules; And
(4) 상기 나정을 코팅하는 단계를 포함할 수 있다. (4) may include the step of coating the tablet.
이하, 본 발명을 실시예에 의해 상세히 설명한다. 단, 하기 실시예는 본 발명을 예시하는 것일 뿐, 본 발명의 내용이 하기 실시예에 한정되는 것은 아니다. Hereinafter, the present invention will be described in detail by examples. However, the following examples are only to illustrate the present invention, the content of the present invention is not limited to the following examples.
실시예Example 1 내지 4 1 to 4
하기 표 1의 조성에 따라 주성분인 토파시티닙 아스파르트산염, 희석제인 유당 일수화물, 서방화제인 HPMC(Methocel K100LV CR®) 및 2종의 친수성 결합제(HPC-L 및 PVP K30)를 혼합한 후 활택제인 스테아르산마그네슘을 추가 투입하여 혼합하였다. 이 혼합물은 건식과립기에서 압축 및 정립하여 건식과립물로 제조한 후 로터리 타정기에서 1정 분량으로 압축하여 타정하였다. According to the composition of Table 1, the main components of tofacitinib aspartate, diluent lactose monohydrate, sustained release agent HPMC (Methocel K100LV CR ® ) and two hydrophilic binders (HPC-L and PVP K30) were mixed and then lubricated Jane magnesium stearate was further added and mixed. This mixture was compressed and granulated in a dry granulator to prepare a dry granulated product, and then compressed and compressed into 1 tablet portions in a rotary tableting machine.
아스파르트산염Tofacitinib
Aspartate
비교예Comparative example 1 및 2 1 and 2
하기 표 2의 조성에 따라 주성분인 토파시티닙 아스파르트산염, 희석제인 유당 일수화물, 서방화제인 HPMC(Methocel K100LV CR®) 및 1종의 친수성 결합제(HPC-L 또는 PVP K30)를 혼합한 후 활택제인 스테아르산마그네슘을 추가 투입하여 혼합하였다. 이 혼합물은 건식과립기에서 압축 및 정립하여 건식과립물로 제조한 후 로터리 타정기에서 1정 분량으로 압축하여 타정하였다.According to the composition of Table 2, the main component of tofacitinib aspartate, diluent lactose monohydrate, sustained release agent HPMC (Methocel K100LV CR ® ) and one hydrophilic binder (HPC-L or PVP K30) are mixed and then lubricated Jane magnesium stearate was further added and mixed. This mixture was compressed and granulated in a dry granulator to prepare a dry granulated product, and then compressed and compressed into 1 tablet portions in a rotary tableting machine.
아스파르트산염Tofacitinib
Aspartate
실험예Experimental Example 1. 용출 프로파일 실험 1. Elution Profile Experiment
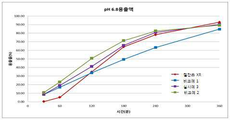
실시예 1 내지 4 및 비교예 1과 2에서 제조된 서방성 제제와 현재 시판 중인 젤잔즈 XR정(대조약, 토파시티닙으로 11mg)에 대하여 용출시험을 미국약전(USP) 용출시험 2 회전 패들 장치에서 수행하였다. 구체적으로, 패들의 속도는 50rpm으로 회전시키고, 용해 매체로서 900mL의 pH 6.8 인산칼륨 완충액을 사용하며 완충액의 온도는 37℃로 유지하면서, 시험 중 적절한 시간에 얻어진 용출액을 필터 진행 후 고성능 액체 크로마토그래피(HPLC)에 의해 방출된 토파시티닙을 분석하여 용출 특성을 조사하고 그 결과를 표 3 및 도 1에 나타내었다. The sustained-release preparations prepared in Examples 1 to 4 and Comparative Examples 1 and 2 and the currently available gelzand XR tablets (control, 11 mg as tofacitinib) were tested for dissolution in the United States Pharmacopoeia (USP). It was performed on the device. Specifically, the speed of the paddle is rotated at 50 rpm, 900 mL of pH 6.8 potassium phosphate buffer is used as the dissolution medium, and the temperature of the buffer is maintained at 37° C., while the eluate obtained at the appropriate time during the test is filtered and then subjected to high performance liquid chromatography Analysis of the tofacitinib released by (HPLC) investigated the dissolution characteristics and the results are shown in Table 3 and FIG. 1.
토파시티닙은 BCS Class 3의 약물로 약물의 용해도는 높고, 생체막 투과도가 낮기 때문에 생체내에서 약물의 흡수는 약물 자체의 투과 속도에 의해 결정된다. 본 서방성 제제와 같이 생체막 투과도에 영향을 줄 수 있는 첨가제를 추가로 포함하지 않는 경우 생체내 투과는 토파시티닙의 물리화학적 특성에 따르므로 생체외 용출 결과에 따라 생체내 방출 정도를 예측할 수 있다. 현재 시판되고 있는 젤잔즈 XR정의 Tmax는 3∼4h으로 이와 동등한 수준으로 생체내 방출되기 위해서는 생체외 용출 실험에서 2∼4h 시점의 용출이 중요하며, 적절하게는 2h에 20~42%, 2.5h에 33-56%, 3h에 46~65%, 3.5h에 55~70%, 4h에 72~76%의 용출을 보이는 것이다. Tofacitinib is a drug of BCS Class 3, and since the drug has high solubility and low biomembrane permeability, absorption of the drug in vivo is determined by the rate of permeation of the drug itself. When the additives that may affect the biomembrane permeability, such as the sustained-release preparation, are not included, in vivo permeation is dependent on the physicochemical properties of tofacitinib, so the degree of in vivo release can be predicted according to the results of in vitro dissolution. . The Tmax of the currently available Gelzanz XR tablets is 3 to 4 h, and in order to be released in vivo at an equivalent level, it is important to dissolve at a time of 2 to 4 h in an in vitro dissolution experiment, suitably 20 to 42% at 2 h, 2.5 h Elution of 33-56%, 46-65% at 3h, 55-70% at 3.5h, and 72-76% at 4h.
표 3과 도 1의 용출 결과를 보면 본 발명의 서방성 제제인 실시예 1 내지 4의 경우 2∼4h 시점에서 대조약인 젤잔즈 XR정과 유사한 용출률(± 10% 이내)을 나타내었다. 그러나, 친수성 결합제를 HPC 1종만 사용한 비교예 1의 경우에는 약물 방출 속도는 일정하게 유지되지만 대조약에 비해 전체적으로 볼 때 용출이 지연되었으며, PVP 1종만 사용한 비교예 2의 경우에는 초반에 빠른 속도로 약물이 방출되고 시간이 지날수록 방출속도가 늦어지는 경향을 보이며 전체적으로 볼 때는 대조약에 비해 용출이 빨리 진행되는 것을 확인하였다. 이를 통해 본 발명의 서방성 제제는 친수성 결합제인 HPC와 PVP를 적절한 비율로 혼합하여 사용함으로써 원하는 용출 패턴을 확보할 수 있었음을 확인하였다. Looking at the dissolution results in Table 3 and Figure 1, in the case of Examples 1 to 4, which is a sustained release formulation of the present invention, the dissolution rate (within ± 10%) similar to that of the gelz XR tablet as a control drug was exhibited at 2 to 4 h. However, in the case of Comparative Example 1 using only the hydrophilic binding agent HPC, the drug release rate was kept constant, but dissolution was delayed when viewed as a whole compared to the control drug. As the drug was released, the release rate tended to be slower with time, and as a whole, it was confirmed that the elution proceeded faster than the control drug. Through this, it was confirmed that the sustained-release preparation of the present invention was able to secure a desired dissolution pattern by mixing HPC and PVP, which are hydrophilic binders, in an appropriate ratio.
실험예Experimental Example 2. PK 프로파일 시험 ( 2.PK profile test ( 젤잔즈Gel glasses IR정IR tablet 5mg BID vs 본 발명의 서방성 제제 5mg BID vs slow release formulation of the present invention QDQD ))
건강한 남성에서 랜덤화된, 개방-라벨, 단일 용량, 2기간, 2처리, 4교차 연구를 진행하였다. 대상체는 투여간 7일의 약효세척 기간을 갖는 토파시티닙의 지속 방출 제제인 본 발명의 실시예 X 및 즉시방출 정제 제제인 젤잔즈 IR정을 수용하였다. 지속 방출 제제는 10mg 단일 용량으로서 제공되었고, 즉시 방출 제제는 단일 용량으로 2개의 5mg정제로서 제공되었으며, 그 PK 프로파일을 비교하고 그 결과를 도 2에 나타내었다. 도 2의 그래프에서 보는 바와 같이, 실제 생체내에서 토파시티닙을 10mg을 함유하는 본 발명의 서방성 제제를 1일 1회 복용한 경우 젤잔즈 IR정 5mg을 1일 2회 복용한 경우와 동등한 Cmax와 AUC를 나타내는 것을 확인하였다. Randomized, open-label, single dose, two-period, two-treatment, four-cross studies were conducted in healthy men. Subjects received Example X of the present invention, a sustained release formulation of tofacitinib having a 7-day washout period between doses, and Gelzand IR tablets, an immediate release tablet formulation. Sustained release formulations were provided as a single 10 mg dose, and immediate release formulations were provided as two 5 mg tablets in a single dose, the PK profiles were compared and the results are shown in FIG. 2. As shown in the graph of FIG. 2, when the sustained-release preparation of the present invention containing 10 mg of tofacitinib is taken once a day in vivo, it is equivalent to the case of taking 5 mg of Gelzanz IR tablet twice a day. It was confirmed that Cmax and AUC were shown.
실험예Experimental Example 3. 식전/식후 PK 프로파일 시험 (본 발명의 서방성 제제 3. PK profile test before/after meal (sustained release preparation of the present invention QDQD 식전 vs 식후) Before and after meals)
본 발명의 같은 서방성 제제의 식이 영향 평가를 진행하였으며, 그 결과를 표 4 및 도 3에 나타내었다. Evaluation of the dietary effect of the same sustained-release preparation of the present invention was performed, and the results are shown in Table 4 and FIG. 3.
표 4와 도 3의 그래프에서 보는 바와 같이, 본 발명의 서방성 제제의 식전 vs 식후 AUC값은 동등한 수준을 나타내었으며, 이는 본 발명의 서방성 제제의 경우 식이에 영향을 받지 않아 서방성 제제가 갖추어야 하는 기준을 확보하였을 의미한다. As shown in the graphs of Table 4 and FIG. 3, the pre-prediction vs post-prandial AUC value of the sustained-release preparation of the present invention exhibited an equivalent level, and the sustained-release preparation was not affected by the diet in the case of the sustained-release preparation of the present invention. It means that you have secured the standards you must have.
본 발명에 따른 서방성 제제는 류마티스 관절염, 건선성 관절염 및 궤양성 대장염 예방 또는 치료 효과를 나타내므로, 제약산업 및 의료현장에서 의약품으로서 이용 가능하다. The sustained-release preparation according to the present invention exhibits a preventive or therapeutic effect on rheumatoid arthritis, psoriatic arthritis and ulcerative colitis, and thus can be used as a pharmaceutical in the pharmaceutical industry and the medical field.
Claims (12)
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180172103A KR20200082006A (en) | 2018-12-28 | 2018-12-28 | Extended release formulation containing tofacitinib or pharmaceutically acceptable salts thereof as an active ingredient and the preparation method for the same |
| PCT/KR2019/017717 WO2020138791A2 (en) | 2018-12-28 | 2019-12-13 | Sustained-release preparation comprising tofacitinib or pharmaceutically acceptable salt thereof and manufacturing method therefor |
| TW108147892A TW202038917A (en) | 2018-12-28 | 2019-12-26 | Extended release formulation containing tofacitinib or pharmaceutically acceptable salts thereof and preparation method for the same |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180172103A KR20200082006A (en) | 2018-12-28 | 2018-12-28 | Extended release formulation containing tofacitinib or pharmaceutically acceptable salts thereof as an active ingredient and the preparation method for the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20200082006A true KR20200082006A (en) | 2020-07-08 |
Family
ID=71126346
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020180172103A KR20200082006A (en) | 2018-12-28 | 2018-12-28 | Extended release formulation containing tofacitinib or pharmaceutically acceptable salts thereof as an active ingredient and the preparation method for the same |
Country Status (3)
| Country | Link |
|---|---|
| KR (1) | KR20200082006A (en) |
| TW (1) | TW202038917A (en) |
| WO (1) | WO2020138791A2 (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114588158B (en) * | 2022-03-14 | 2023-09-05 | 武汉工程大学 | Application of indole piperidine pyrimidine derivative in preparation of novel coronavirus inhibitor |
| WO2024042218A1 (en) * | 2022-08-26 | 2024-02-29 | Synthon B.V. | Prolonged release tofacitinib compositions without functional coating |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012100949A1 (en) | 2011-01-27 | 2012-08-02 | Ratiopharm Gmbh | Oral dosage forms for modified release comprising tasocitinib |
| WO2014147526A1 (en) | 2013-03-16 | 2014-09-25 | Pfizer Inc. | Tofacitinib oral sustained release dosage forms |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0613310D0 (en) * | 2006-07-05 | 2006-08-16 | Merck Sharp & Dohme | The use of pvp to control the release profile of an active ingredient from a hydrophilic polymer matrix tablet |
| WO2014174073A1 (en) * | 2013-04-26 | 2014-10-30 | Sandoz Ag | Sustained release formulations of tofacitinib |
| US10166183B2 (en) * | 2014-02-07 | 2019-01-01 | Auspex Pharmaceuticals, Inc. | Pharmaceutical formulations |
| US20170049774A1 (en) * | 2015-08-19 | 2017-02-23 | Sun Pharmaceutical Industries Limited | Sustained release oral pharmaceutical compositions of tofacitinib |
-
2018
- 2018-12-28 KR KR1020180172103A patent/KR20200082006A/en not_active Application Discontinuation
-
2019
- 2019-12-13 WO PCT/KR2019/017717 patent/WO2020138791A2/en active Application Filing
- 2019-12-26 TW TW108147892A patent/TW202038917A/en unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012100949A1 (en) | 2011-01-27 | 2012-08-02 | Ratiopharm Gmbh | Oral dosage forms for modified release comprising tasocitinib |
| WO2014147526A1 (en) | 2013-03-16 | 2014-09-25 | Pfizer Inc. | Tofacitinib oral sustained release dosage forms |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202038917A (en) | 2020-11-01 |
| WO2020138791A3 (en) | 2020-08-20 |
| WO2020138791A2 (en) | 2020-07-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1340495B1 (en) | Extended release tablet of clarithromycin | |
| AU2012210777B2 (en) | Oral dosage forms for modified release comprising tasocitinib | |
| KR100780553B1 (en) | Pharmaceutical compositions and formulations of Metformin extended release tablets and its preparing method | |
| JP6122098B2 (en) | Pharmaceutical composition comprising olmesartan medoxomil and rosuvastatin or a salt thereof | |
| WO2011111818A1 (en) | Sustained release type pharmaceutical composition containing mosapride or salt thereof | |
| WO2009034541A9 (en) | Controlled release pharmaceutical dosage forms of trimetazidine | |
| WO2020138791A2 (en) | Sustained-release preparation comprising tofacitinib or pharmaceutically acceptable salt thereof and manufacturing method therefor | |
| JP2017523149A (en) | Edoxaban pharmaceutical composition | |
| WO2009027786A2 (en) | Matrix dosage forms of varenicline | |
| EP3697392B1 (en) | Tablets comprising tamsulosin and solifenacin | |
| KR102389339B1 (en) | Controlled release high-dose tamsulosin hydrochloride tablet and its preparing method | |
| US20120195966A1 (en) | Oral dosage form for modified release comprising a jak3 inhibitor | |
| CN110623934B (en) | Trimetazidine hydrochloride sustained release tablet and preparation method thereof | |
| KR101265491B1 (en) | Dissolution rate controlled multilayered tablet for oral administration containing sarpogrelate hydrochloride and manufacturing method thereof | |
| WO2020109319A1 (en) | Pharmaceutical composition comprising ramipril and indapamide | |
| KR20210096162A (en) | pharmaceutical composition | |
| KR101910707B1 (en) | Metformin Extended-release Tablets Having Enhanced Patient Compliance and its Preparing Method | |
| KR20210012082A (en) | A pharmaceutical composition comprising mirabegron and tamsulosin | |
| KR20180101307A (en) | An oral sustained-release tablet comprising tianeptine or pharmaceutically acceptable salts thereof | |
| CN109864978B (en) | Sustained-release preparation of 5-methyltetrahydrofolic acid and preparation method thereof | |
| AU2016210691A1 (en) | Controlled release formulations of paracetamol | |
| KR20120092993A (en) | An oral sustained-release tablet comprising tianeptine or pharmaceutically acceptable salts thereof | |
| EP3335703A1 (en) | Pharmaceutical composition comprising omarigliptin | |
| WO2022117594A1 (en) | Orally-administered preparation containing solifenacin and tamsulosin | |
| TW201729814A (en) | Solid preparation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| E902 | Notification of reason for refusal | ||
| E601 | Decision to refuse application |