KR20200081943A - Method for producing Aralia continentalis herbal drink - Google Patents
Method for producing Aralia continentalis herbal drink Download PDFInfo
- Publication number
- KR20200081943A KR20200081943A KR1020180171995A KR20180171995A KR20200081943A KR 20200081943 A KR20200081943 A KR 20200081943A KR 1020180171995 A KR1020180171995 A KR 1020180171995A KR 20180171995 A KR20180171995 A KR 20180171995A KR 20200081943 A KR20200081943 A KR 20200081943A
- Authority
- KR
- South Korea
- Prior art keywords
- weight
- herbal
- mixture
- beverage
- prepared
- Prior art date
Links
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 20
- 241000838298 Aralia continentalis Species 0.000 title abstract 4
- 235000013361 beverage Nutrition 0.000 claims abstract description 46
- 239000000203 mixture Substances 0.000 claims abstract description 39
- HELXLJCILKEWJH-NCGAPWICSA-N rebaudioside A Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O HELXLJCILKEWJH-NCGAPWICSA-N 0.000 claims abstract description 16
- 244000223760 Cinnamomum zeylanicum Species 0.000 claims abstract description 15
- 235000001453 Glycyrrhiza echinata Nutrition 0.000 claims abstract description 15
- 235000006200 Glycyrrhiza glabra Nutrition 0.000 claims abstract description 15
- 235000017382 Glycyrrhiza lepidota Nutrition 0.000 claims abstract description 15
- 235000001287 Guettarda speciosa Nutrition 0.000 claims abstract description 15
- GXCLVBGFBYZDAG-UHFFFAOYSA-N N-[2-(1H-indol-3-yl)ethyl]-N-methylprop-2-en-1-amine Chemical compound CN(CCC1=CNC2=C1C=CC=C2)CC=C GXCLVBGFBYZDAG-UHFFFAOYSA-N 0.000 claims abstract description 15
- 235000006886 Zingiber officinale Nutrition 0.000 claims abstract description 15
- 235000006533 astragalus Nutrition 0.000 claims abstract description 15
- 235000017803 cinnamon Nutrition 0.000 claims abstract description 15
- 235000008397 ginger Nutrition 0.000 claims abstract description 15
- 229940010454 licorice Drugs 0.000 claims abstract description 15
- 241001061264 Astragalus Species 0.000 claims abstract description 14
- 210000004233 talus Anatomy 0.000 claims abstract description 14
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 13
- 235000006484 Paeonia officinalis Nutrition 0.000 claims abstract description 12
- 238000000034 method Methods 0.000 claims abstract description 9
- 235000008216 herbs Nutrition 0.000 claims abstract description 7
- 244000303040 Glycyrrhiza glabra Species 0.000 claims abstract description 6
- 244000170916 Paeonia officinalis Species 0.000 claims abstract description 6
- 244000273928 Zingiber officinale Species 0.000 claims abstract description 6
- 240000002045 Guettarda speciosa Species 0.000 claims abstract 4
- 244000228451 Stevia rebaudiana Species 0.000 claims abstract 4
- 231100000614 poison Toxicity 0.000 claims description 13
- 241000208340 Araliaceae Species 0.000 claims description 10
- 235000005035 Panax pseudoginseng ssp. pseudoginseng Nutrition 0.000 claims description 10
- 235000003140 Panax quinquefolius Nutrition 0.000 claims description 10
- 235000008434 ginseng Nutrition 0.000 claims description 10
- 241000202807 Glycyrrhiza Species 0.000 claims description 9
- 238000002360 preparation method Methods 0.000 claims description 8
- 231100000331 toxic Toxicity 0.000 claims description 7
- 230000002588 toxic effect Effects 0.000 claims description 7
- 239000002574 poison Substances 0.000 claims description 5
- 241000675108 Citrus tangerina Species 0.000 claims 3
- 241001672694 Citrus reticulata Species 0.000 abstract description 12
- 241000722818 Aralia Species 0.000 abstract 1
- 231100000572 poisoning Toxicity 0.000 description 15
- 230000000607 poisoning effect Effects 0.000 description 15
- 241000544066 Stevia Species 0.000 description 12
- 241000125175 Angelica Species 0.000 description 11
- 241000411851 herbal medicine Species 0.000 description 10
- 238000003860 storage Methods 0.000 description 10
- 241000234314 Zingiber Species 0.000 description 9
- 238000002156 mixing Methods 0.000 description 8
- 230000007096 poisonous effect Effects 0.000 description 8
- 239000000463 material Substances 0.000 description 7
- 241000736199 Paeonia Species 0.000 description 6
- 150000008442 polyphenolic compounds Chemical class 0.000 description 6
- 235000013824 polyphenols Nutrition 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- 239000000796 flavoring agent Substances 0.000 description 5
- 235000019634 flavors Nutrition 0.000 description 5
- 239000004615 ingredient Substances 0.000 description 5
- 230000001953 sensory effect Effects 0.000 description 5
- 241000894006 Bacteria Species 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 235000013305 food Nutrition 0.000 description 4
- 235000019629 palatability Nutrition 0.000 description 4
- 238000002835 absorbance Methods 0.000 description 3
- 230000003110 anti-inflammatory effect Effects 0.000 description 3
- 238000011109 contamination Methods 0.000 description 3
- MGJZITXUQXWAKY-UHFFFAOYSA-N diphenyl-(2,4,6-trinitrophenyl)iminoazanium Chemical compound [O-][N+](=O)C1=CC([N+](=O)[O-])=CC([N+]([O-])=O)=C1N=[N+](C=1C=CC=CC=1)C1=CC=CC=C1 MGJZITXUQXWAKY-UHFFFAOYSA-N 0.000 description 3
- 241000196324 Embryophyta Species 0.000 description 2
- 241000588724 Escherichia coli Species 0.000 description 2
- 230000002292 Radical scavenging effect Effects 0.000 description 2
- 239000007844 bleaching agent Substances 0.000 description 2
- HHEAADYXPMHMCT-UHFFFAOYSA-N dpph Chemical compound [O-][N+](=O)C1=CC([N+](=O)[O-])=CC([N+]([O-])=O)=C1[N]N(C=1C=CC=CC=1)C1=CC=CC=C1 HHEAADYXPMHMCT-UHFFFAOYSA-N 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- LNTHITQWFMADLM-UHFFFAOYSA-N gallic acid Chemical compound OC(=O)C1=CC(O)=C(O)C(O)=C1 LNTHITQWFMADLM-UHFFFAOYSA-N 0.000 description 2
- 210000004209 hair Anatomy 0.000 description 2
- 230000037231 joint health Effects 0.000 description 2
- 230000000813 microbial effect Effects 0.000 description 2
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 description 2
- MHVJRKBZMUDEEV-UHFFFAOYSA-N (-)-ent-pimara-8(14),15-dien-19-oic acid Natural products C1CCC(C(O)=O)(C)C2C1(C)C1CCC(C=C)(C)C=C1CC2 MHVJRKBZMUDEEV-UHFFFAOYSA-N 0.000 description 1
- MHVJRKBZMUDEEV-XIHRTOKZSA-N (1r,4as,4br,7r,10as)-7-ethenyl-1,4a,7-trimethyl-3,4,4b,5,6,9,10,10a-octahydro-2h-phenanthrene-1-carboxylic acid Chemical compound [C@@H]1([C@](CCC2)(C)C(O)=O)[C@]2(C)[C@@H]2CC[C@@](C=C)(C)C=C2CC1 MHVJRKBZMUDEEV-XIHRTOKZSA-N 0.000 description 1
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- 241000332371 Abutilon x hybridum Species 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- 241000361919 Metaphire sieboldi Species 0.000 description 1
- 208000000112 Myalgia Diseases 0.000 description 1
- 206010033799 Paralysis Diseases 0.000 description 1
- 206010033892 Paraplegia Diseases 0.000 description 1
- 235000014676 Phragmites communis Nutrition 0.000 description 1
- 244000000231 Sesamum indicum Species 0.000 description 1
- 235000003434 Sesamum indicum Nutrition 0.000 description 1
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 238000001467 acupuncture Methods 0.000 description 1
- 238000007792 addition Methods 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 235000021028 berry Nutrition 0.000 description 1
- 238000011088 calibration curve Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 230000036570 collagen biosynthesis Effects 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 235000019788 craving Nutrition 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 235000013399 edible fruits Nutrition 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 230000007760 free radical scavenging Effects 0.000 description 1
- 229940074391 gallic acid Drugs 0.000 description 1
- 235000004515 gallic acid Nutrition 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 230000005965 immune activity Effects 0.000 description 1
- 230000036737 immune function Effects 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 210000002540 macrophage Anatomy 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 239000008213 purified water Substances 0.000 description 1
- 229940079877 pyrogallol Drugs 0.000 description 1
- 235000021067 refined food Nutrition 0.000 description 1
- 230000001850 reproductive effect Effects 0.000 description 1
- 239000002689 soil Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 238000005728 strengthening Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/38—Other non-alcoholic beverages
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/52—Adding ingredients
- A23L2/60—Sweeteners
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2200/00—Function of food ingredients
- A23V2200/30—Foods, ingredients or supplements having a functional effect on health
- A23V2200/302—Foods, ingredients or supplements having a functional effect on health having a modulating effect on age
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/20—Natural extracts
- A23V2250/21—Plant extracts
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2300/00—Processes
- A23V2300/14—Extraction
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2300/00—Processes
- A23V2300/24—Heat, thermal treatment
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Nutrition Science (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Food Science & Technology (AREA)
- Polymers & Plastics (AREA)
- Botany (AREA)
- Mycology (AREA)
- Non-Alcoholic Beverages (AREA)
- Medicines Containing Plant Substances (AREA)
Abstract
Description
본 발명은 독활, 작약, 당귀, 백출, 귤피, 복령, 사삼, 계피, 맥아, 황기, 감초, 생강 및 스테비아를 혼합한 한약재 혼합물을 이용하여 제조하는 것을 특징으로 하는 독활 한방 음료의 제조방법 및 상기 방법으로 제조된 독활 한방 음료에 관한 것이다.The present invention is a method of manufacturing a medicinal herbal beverage characterized in that it is prepared using a mixture of herbal medicines containing poisonous, peony, Angelica, Baekchul, Tangerine, Bokryeong, Ginseng, cinnamon, malt, astragalus, licorice, ginger and stevia. It relates to a unique herbal beverage prepared by the method.
독활은 쌍떡잎식물로 산형화목 두릅나무과의 여러해살이풀로 땅두릅이라고도 한다. 유사한 발음 때문에 땃두릅이라 불리기도 하나 땃두릅나무와는 다른 종이다. 산에서 자라며, 높이는 1.5 m이고 꽃을 제외한 전체에 털이 약간 있다. 잎은 어긋나고 길이 50~100 cm, 너비 3~20 cm이며 어릴 때에는 연한 갈색 털이 있다. 작은잎은 달걀 모양 또는 타원형이고 가장자리에 톱니가 있다. 잎 표면은 녹색이고 뒷면은 흰빛이 돌며 잎자루 밑부분 양쪽에 작은 떡잎이 있다. 꽃은 7~8월에 크고 연한 녹색으로 피고 원추꽃차례가 자라며 총상(總狀)으로 갈라진 가지 끝에 산형꽃차례로 달린 양성화이다. 열매는 장과로서 9~10월에 검게 익는다. 바람에 움직이지 않는다는 뜻으로 독활이라고 부른다. 이른봄 어린 순은 식용하며, 가을에 잎이 죽은 다음 흙을 덮어서 어린 순이 길게 자랄 수 있도록 한다. 뿌리는 약용하는데, 근육통·하반신·마비·두통·중풍의 반신불수 등에 많이 쓰인다.Poison is a dicotyledonous plant, a perennial plant of the elmaceae family of mountain-type trees, and is also called earthworm. Because of its similar pronunciation, it is also called 땃durum, but it is a different species from 땃durmu. It grows in the mountains, is 1.5 m high and has some hairs except for flowers. Leaves are alternate, 50~100 cm long, 3~20 cm wide, and have light brown hair when young. The small leaves are egg-shaped or oval-shaped and have serrated edges. The surface of the leaf is green, the back is white, and there are small cotyledons on both sides of the bottom of the petiole. The flower blooms in large and light green in July-August, and the cone inflorescence grows, and it is a cultivation with a mountain-shaped inflorescence at the end of a branch with a gunshot. Fruits are berries and ripen black in September-October. It is called poisoning, meaning that it does not move in the wind. Early spring young shoots are edible, and after the leaves die in the fall, cover the soil so that the young shoots can grow long. The roots are medicinal, and they are often used for myalgia, lower body, paralysis, headache, and paraplegia.
독활의 국내 연구에서는 관절건강과 관련되는 면역기능 강화 분야에서 독활 물 추출물이 대식세포 면역 활성을 증진시키는 효능이 있다고 다수 보고되었으며, 독활은 항염증 유효성분 continentalic acid 화학구조 등 다수의 항염증 관련 보고가 있다. 또한, 독활의 국외연구로는 면역활성 증진이 독보적이고, 관절건강과 관련된 콜라겐 생합성을 증가시켰다는 보고가 있다. 우리나라 농가의 소득 대체 작목인 독활의 어린 새순을 식품에 적용하는 연구가 보고되고 있으며 성숙한 독활 잎은 가을이 되면 쓸모없이 그대로 버려지고 있어서 이를 채취하여 조사료원으로 이용한 연구가 보고되었다. 독활을 깻잎과 비교해보면 질기고 더 두꺼운 특성이 있으며, 독활을 이용하여 식품에 적용한 연구는 미미한 실정이다. 우리나라의 소득작물인 독활 잎은 2-5월에 비가림 및 노지재배 상품이 출하되는데 5월 중순 이후에는 온도상승으로 생식용으로 부적합한 상품이 다량 발생하여 저장식품인 김치를 제조하여 판매를 시도하고 있으나 저품질로 인해 판매의 어려움을 겪고 있어 독활 잎을 활용한 가공식품의 레시피 개발과 품질 개선이 필요한 실정이다.In a domestic study of poisoning, a number of anti-inflammatory extracts have been reported to enhance macrophage immune activity in the field of strengthening immune function related to joint health, and poisoning has been reported for a number of anti-inflammatory properties, such as the chemical structure of continentalic acid, an anti-inflammatory active ingredient. There is. In addition, there have been reports that boosting immunity is unique and increased collagen biosynthesis related to joint health. Studies have been reported on the application of young shoots of poisonous rot, which is an alternative crop for income of Korean farmers, to mature food, and mature leaves are discarded as they are in the fall. Compared to sesame leaves, it has a tougher and thicker characteristic, and studies applied to foods using it are insignificant. In Korea, the poisonous leaves, which are crops for income, are shipped from May to May in May, and after mid-May, there have been many unsuitable products for reproductive use due to temperature rise. However, due to low quality, it is difficult to sell, so it is necessary to develop recipes and improve quality of processed foods using toxic leaves.
한국등록특허 제0931880호에는 침향, 조릿대, 원지 및 산조인을 함유하는 한방 음료의 제조방법이 개시되어 있으며, 한국등록특허 제0931879호에는 황금, 갈대뿌리 및 왕대를 포함하는 한방 음료가 개시되어 있으나, 본 발명의 독활 한방 음료의 제조방법과는 상이하다.Korean Registered Patent No. 0931880 discloses a method of manufacturing a herbal beverage containing acupuncture, stalk, base paper, and sanjoin, and Korean Registered Patent No. 0931879 discloses a herbal beverage containing golden, reed roots, and Wangdae, It is different from the manufacturing method of the poisonous herbal beverage of the present invention.
본 발명은 상기와 같은 요구에 의해 안출된 것으로서, 본 발명은 독활을 이용하여 음료 제조 시 품질 및 기호도가 우수한 음료를 제조하기 위해, 부재료 선정, 재료 전처리, 배합비 등의 제조조건을 최적화하여, 이취 및 이미가 없고 풍미 및 기호도가 우수한 독활 한방 음료를 개발하여 본 발명에 이르렀다.The present invention was devised by the above-mentioned demands, and the present invention optimizes manufacturing conditions such as selection of subsidiary materials, pretreatment of materials, mixing ratios, etc. in order to produce beverages having excellent quality and preference when manufacturing beverages using poisoning. And has developed a unique herbal beverage with no taste and excellent taste and has reached the present invention.
상기 과제를 해결하기 위해, 본 발명은 (1) 독활, 작약, 당귀, 백출, 귤피, 복령, 사삼, 계피, 맥아, 황기, 감초, 생강 및 스테비아를 혼합한 한약재 혼합물을 준비하는 단계; 및 (2) 상기 (1)단계의 준비한 한약재 혼합물에 물을 첨가한 후 추출하는 단계를 포함하여 제조하는 것을 특징으로 하는 독활 한방 음료의 제조방법을 제공한다.In order to solve the above problems, the present invention (1) poison, peony, Angelica, Baekchul, mandarin orange, Bokryeong, ginseng, cinnamon, malt, astragalus, licorice, ginger and preparing a mixture of herbal mixtures of stevia; And (2) adding and extracting water after adding water to the prepared herbal mixture prepared in step (1).
또한, 본 발명은 상기 방법으로 제조된 독활 한방 음료를 제공한다.In addition, the present invention provides a unique herbal beverage prepared by the above method.
본 발명의 독활 및 한약재를 이용한 음료는 재료 특유의 이취는 제거하고, 온화한 맛으로 인해 기호성이 증진되며, 항산화 활성이 높아 소비자들이 더욱 선호하는 음료를 제공할 수 있다.The beverage using the poison and herbal medicines of the present invention removes the odor peculiar to the material, enhances palatability due to the mild taste, and has high antioxidant activity, thereby providing a beverage preferred by consumers.
도 1은 본 발명의 독활 한방 음료의 제조과정을 보여준다.
도 2는 독활 한방 음료의 저장 온도 및 기간에 따른 총 폴리페놀 함량 변화를 비교한 그래프이다.
도 3은 독활 한방 음료의 저장 온도 및 기간에 따른 DPPH 라디칼 소거능 변화를 비교한 그래프이다.
도 4는 독활 한방 음료의 저장 온도 및 기간에 따른 SOD 유사활성 변화를 비교한 그래프이다.Figure 1 shows the manufacturing process of the unique herbal beverage of the present invention.
Figure 2 is a graph comparing the change in the total polyphenol content according to the storage temperature and duration of the toxic herbal drink.
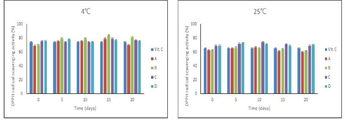
Figure 3 is a graph comparing the change in the DPPH radical scavenging ability according to the storage temperature and duration of the toxic herbal drink.
Figure 4 is a graph comparing the changes in the SOD-like activity according to the storage temperature and duration of the oriental herbal drink.
본 발명의 목적을 달성하기 위하여, 본 발명은 In order to achieve the object of the present invention, the present invention
(1) 독활, 작약, 당귀, 백출, 귤피, 복령, 사삼, 계피, 맥아, 황기, 감초, 생강 및 스테비아를 혼합한 한약재 혼합물을 준비하는 단계; 및(1) preparing a mixture of herbal medicines containing poison, peony, Angelica, Baekchul, Tangerine, Bokryeong, Ginseng, cinnamon, malt, astragalus, licorice, ginger and stevia; And
(2) 상기 (1)단계의 준비한 한약재 혼합물에 물을 첨가한 후 추출하는 단계를 포함하여 제조하는 것을 특징으로 하는 독활 한방 음료의 제조방법을 제공한다.(2) It provides a method of manufacturing a toxic herbal beverage, characterized in that it comprises a step of extracting after adding water to the mixture of medicinal herbs prepared in step (1).
본 발명의 독활 한방 음료의 제조방법에서, 상기 (1)단계의 한약재 혼합물은 바람직하게는 한약재 혼합물 총 중량 기준으로, 독활 13~17 중량%, 작약 13~17 중량%, 당귀 6~9 중량%, 백출 5~7 중량%, 귤피 6~9 중량%, 복령 4~5 중량%, 사삼 5~7 중량%, 계피 2~4 중량%, 맥아 13~17 중량%, 황기 6~9 중량%, 감초 4~5 중량%, 생강 5~6 중량% 및 스테비아 2~4 중량%를 혼합하여 준비할 수 있으며, 더욱 바람직하게는 한약재 혼합물 총 중량 기준으로, 독활 15 중량%, 작약 15 중량%, 당귀 7.5 중량%, 백출 6 중량%, 귤피 7.5 중량%, 복령 4.5 중량%, 사삼 6 중량%, 계피 3 중량%, 맥아 15 중량%, 황기 7.5 중량%, 감초 4.5 중량%, 생강 5.5 중량% 및 스테비아 3 중량%를 혼합하여 준비할 수 있다. 상기와 같은 한약재 종류 및 배합비로 준비한 한약재 혼합물을 이용하여 음료를 제조하는 것이 독활과 한약재료들의 맛과 향이 잘 어우러지고 적절한 단맛과 깔끔한 맛으로 소비자들의 입맛에 맞는 음료를 제공할 수 있었다.In the method of manufacturing a poisonous herbal beverage of the present invention, the herbal medicine mixture in the step (1) is preferably based on the total weight of the herbal medicine mixture, 13 to 17% by weight, 13 to 17% by weight, 6 to 9% by weight of Angelica , Baekchul 5~7 wt%, Tangerine peel 6~9 wt%, Bokryeong 4~5 wt%, Sasam 5~7 wt%, Cinnamon 2~4 wt%, Malt 13~17 wt%, Astragalus 6~9 wt%, Licorice can be prepared by mixing 4-5% by weight, 5-6% by weight of ginger, and 2-4% by weight of stevia, more preferably, based on the total weight of the herbal mixture, 15% by weight, 15% by weight, Angelica 7.5 wt%, Baekchul 6 wt%, Tangerine peel 7.5 wt%, Bokryeong 4.5 wt%, Ginseng 6 wt%, Cinnamon 3 wt%, Malt 15 wt%, Astragalus 7.5 wt%, Licorice 4.5 wt%, Ginger 5.5 wt% and Stevia It can be prepared by mixing 3% by weight. Producing beverages using the herbal medicine mixture prepared in the above-described types and combination ratios of the herbal medicines, the taste and aroma of the herbal ingredients are well harmonized, and it is possible to provide a beverage suitable for consumers' taste with proper sweetness and neat taste.
또한, 본 발명의 독활 한방 음료의 제조방법에서, 상기 (2)단계는 바람직하게는 한약재 혼합물에 혼합물 대비 물을 80~120배(v/w) 첨가한 후 90~110℃에서 1~3시간 동안 추출할 수 있으며, 더욱 바람직하게는 한약재 혼합물에 혼합물 대비 물을 100배(v/w) 첨가한 후 100℃에서 2시간 동안 추출할 수 있다. 상기와 같은 조건으로 추출하는 것이 각 재료들이 갖는 영양성분 및 풍미를 손상시키지 않으면서 재료들의 맛과 향이 잘 어우러지고 은은하게 느껴지는 한약재의 향과 맛으로 인해 기호도를 높일 수 있었다.In addition, in the method of manufacturing a toxic herbal beverage of the present invention, step (2) is preferably 80 to 120 times (v/w) of water compared to the mixture to the medicinal herb mixture, and then 1 to 3 hours at 90 to 110°C. It can be extracted during, and more preferably, 100 times (v/w) of water is added to the mixture of medicinal herbs and then extracted at 100° C. for 2 hours. The extractability under the above conditions could enhance the palatability due to the flavor and taste of the herbal medicine that harmonizes well with the taste and aroma of the ingredients without compromising the nutrients and flavors of each material.
본 발명의 독활 한방 음료의 제조방법은, 보다 구체적으로는The method of manufacturing a unique herbal beverage of the present invention, more specifically
(1) 한약재 혼합물 총 중량 기준으로, 독활 13~17 중량%, 작약 13~17 중량%, 당귀 6~9 중량%, 백출 5~7 중량%, 귤피 6~9 중량%, 복령 4~5 중량%, 사삼 5~7 중량%, 계피 2~4 중량%, 맥아 13~17 중량%, 황기 6~9 중량%, 감초 4~5 중량%, 생강 5~6 중량% 및 스테비아 2~4 중량%를 혼합한 한약재 혼합물을 준비하는 단계; 및(1) Based on the total weight of the herbal mixture, 13~17% by weight, 13~17% by weight, 6~9% by weight, Angelica 5~7% by weight, 6~9% by weight of tangerine, 4~5% by weight of Bokryeong %, Ginseng 5-7% by weight, Cinnamon 2-4% by weight, Malt 13-17% by weight, Astragalus 6-9% by weight, Licorice 4-5% by weight, Ginger 5-6% by weight, and Stevia 2-4% by weight Preparing a mixture of medicinal herbs mixed with; And
(2) 상기 (1)단계의 준비한 한약재 혼합물에 혼합물 대비 물을 80~120배(v/w) 첨가한 후 90~110℃에서 1~3시간 동안 추출하는 단계를 포함할 수 있으며,(2) 80 to 120 times (v/w) of water compared to the mixture is added to the prepared herbal mixture prepared in step (1), and may include extracting at 90 to 110° C. for 1 to 3 hours,
더욱 구체적으로는More specifically
(1) 한약재 혼합물 총 중량 기준으로, 독활 15 중량%, 작약 15 중량%, 당귀 7.5 중량%, 백출 6 중량%, 귤피 7.5 중량%, 복령 4.5 중량%, 사삼 6 중량%, 계피 3 중량%, 맥아 15 중량%, 황기 7.5 중량%, 감초 4.5 중량%, 생강 5.5 중량% 및 스테비아 3 중량%를 혼합한 한약재 혼합물을 준비하는 단계; 및(1) Based on the total weight of the herbal mixture, 15% by weight, 15% by weight peony, 7.5% by weight Angelica, 6% by weight Baekchul, 7.5% by weight tangerine, 4.5% by weight Bokryeong, 6% by weight ginseng, 3% by weight cinnamon, Preparing 15% by weight of malt, 7.5% by weight of astragalus, 4.5% by weight of licorice, 5.5% by weight of ginger, and 3% by weight of stevia; And
(2) 상기 (1)단계의 준비한 한약재 혼합물에 혼합물 대비 물을 100배(v/w) 첨가한 후 100℃에서 2시간 동안 추출하는 단계를 포함할 수 있다.(2) 100% (v/w) of water is added to the mixture of the herbal preparation prepared in the step (1), followed by extraction at 100° C. for 2 hours.
본 발명은 또한, 상기 방법으로 제조된 독활 한방 음료를 제공한다.The present invention also provides a unique herbal beverage prepared by the above method.
이하, 본 발명의 제조예 및 실시예를 들어 상세히 설명한다. 단, 하기 제조예 및 실시예는 본 발명을 예시하는 것일 뿐, 본 발명의 내용이 하기 제조예 및 실시예에 한정되는 것은 아니다.Hereinafter, the production examples and examples of the present invention will be described in detail. However, the following production examples and examples are only to illustrate the present invention, the content of the present invention is not limited to the following production examples and examples.
제조예Manufacturing example 1. One. 독활Poisoning 한방 음료 Herbal drink
(1) 한약재 혼합물 총 중량 기준으로, 독활 15 중량%, 작약 15 중량%, 당귀 7.5 중량%, 백출 6 중량%, 귤피 7.5 중량%, 복령 4.5 중량%, 사삼 6 중량%, 계피 3 중량%, 맥아 15 중량%, 황기 7.5 중량%, 감초 4.5 중량%, 생강 5.5 중량% 및 스테비아 3 중량%를 혼합한 한약재 혼합물을 준비하였다.(1) Based on the total weight of the herbal mixture, 15% by weight, 15% by weight peony, 7.5% by weight Angelica, 6% by weight Baekchul, 7.5% by weight tangerine, 4.5% by weight Bokryeong, 6% by weight ginseng, 3% by weight cinnamon, A herbal medicine mixture was prepared by mixing
(2) 상기 (1)단계의 준비한 한약재 혼합물에 혼합물 대비 물을 100배(v/w) 첨가한 후 100℃에서 2시간 동안 추출하였다.(2) 100 times (v/w) of water was added to the mixture of the medicinal herbs prepared in step (1), followed by extraction at 100° C. for 2 hours.
상기 제조예 1의 방법으로 독활 한방 음료를 제조하되, (1)단계의 한약재 혼합물 준비 시 재료 배합비를 표 1의 A, B 및 D의 배합비로 준비한 한약재 혼합물을 이용하여 독활 한방 음료를 각각 제조한 후, 하기 실시예 1 내지 6과 같이 품질을 비교하였다.In the preparation of the herbal herbal beverage in the method of Preparation Example 1, (1) when preparing the herbal mixture in step 1, the herbal compound mixture was prepared using the herbal mixture prepared in the mixing ratio of A, B and D in Table 1, respectively. Then, the quality was compared as in Examples 1 to 6 below.
실시예Example 1. One. 독활Poisoning 한방 음료의 관능검사 Sensory test of herbal beverages
한방 음료의 기호도 검사를 실시하였다. 평가 항목으로는 색, 향, 맛, 전반적인 기호도로 하였고, 각 항목에 대하여 9점 척도법(1점: 아주 많이 싫음, 5점: 보통, 9점: 아주 많이 좋음)을 사용하여 평가하도록 하였다. 한방 음료는 제조하여 4℃ 및 25℃에 보관한 후 검사 직전에 꺼내어 일회용 종이컵에 약 30 mL씩 담아 입안을 헹굴 수 있도록 정수된 물을 함께 제공하였다. 기호도 조사 결과는 향과 전체적인 기호도에서 C 음료가 가장 높은 점수로 평가되었다.The preference for herbal beverages was also tested. The evaluation items were color, aroma, taste, and overall preference. Each item was evaluated using a 9-point scale (1 point: very disliked, 5 points: normal, 9 points: very good). Medicinal beverages were prepared, stored at 4°C and 25°C, taken out immediately before the test, and placed in disposable paper cups of approximately 30 mL to provide purified water to rinse the mouth. The results of the palatability survey evaluated C beverages as the highest scores for aroma and overall palatability.
실시예Example 2. 2. 독활Poisoning 한방 음료의 pH 및 당도 PH and sugar content of herbal beverages
독활 한방 음료의 pH는 pH 미터(Orion star A211, Thermo scientific)를 이용하여 측정하였고, 당도 변화는 디지털 당도계(RA-250WE, ATAGO)로 측정하였다. pH 변화는 4℃ 및 25℃에서 저장기간이 경과함에 따라 큰 차이 없이 4.9~5.1 사이로 나타났다.The pH of the oriental herbal drink was measured using a pH meter (Orion star A211, Thermo scientific), and the change in sugar content was measured with a digital sugar meter (RA-250WE, ATAGO). The pH change was found to be between 4.9 and 5.1 with no significant difference as the storage period elapsed at 4°C and 25°C.
당도는 A 음료는 스테비아 첨가 B, C 및 D 음료보다 당도 1 Brix 정도 낮게 나타내었고, 저장온도 및 기간에 따라 큰 변화량이 없었다.The sugar content of the A beverage was lower than that of the B, C and D beverages with stevia by 1 Brix, and there was no significant change depending on the storage temperature and duration.
실시예Example 3. 3. 독활Poisoning 한방 음료의 총 폴리페놀 Total polyphenols in herbal beverages
총 폴리페놀은 Folin-Ciocaltus 발색법에 준해 분석하였다. 시료 1 mL에 증류수 7.5 mL를 가한 후 3분간 방치시킨 다음 Folin-Ciocalteu 시약 0.5 mL를 첨가하고 3.5% 무수탄산나트륨(sodium carbonate anhydrous) 1 mL를 넣고 1시간 동안 방치 후, 760 nm에서 흡광도를 측정하였다. 표준물질은 갈산을 사용하여 표준 검량 곡선을 작성 후 총 폴리페놀 함량을 정량하였다. 한방 음료의 저장기간이 경과함에 따라 총 폴리페놀 함량은 점차 증가하다가 일정한 수준을 나타내었으며 스테비아 첨가군들이 무첨가군에 비해 총 폴리페놀 함량이 높았지만 25℃에서는 무첨가군이 다소 높게 나타났다(도 2).Total polyphenols were analyzed according to the Folin-Ciocaltus color development method. 7.5 mL of distilled water was added to 1 mL of the sample, and then allowed to stand for 3 minutes, 0.5 mL of Folin-Ciocalteu reagent was added, 1 mL of 3.5% sodium carbonate anhydrous was added and left for 1 hour, and absorbance was measured at 760 nm. . As a standard, a standard calibration curve was prepared using gallic acid, and then the total polyphenol content was quantified. The total polyphenol content gradually increased with the storage period of the herbal beverage, and then showed a certain level. The stevia-added groups had higher total polyphenol content than the non-added group, but the non-added group was slightly higher at 25°C (Fig. 2). .
실시예Example 4. 4. 독활Poisoning 한방 음료의 Herbal DPPHDPPH 라디칼Radical 소거능Erasing ability
DPPH 자유 라디칼 소거활성은 Blois의 방법을 변형하여 측정하였다. 0.1 mM DPPH 용액 150 ㎕와 시료 50 ㎕를 혼합하여 30분간 암소에서 반응시킨 후 530 nm에서 흡광도를 측정하였다. 제조 당일 DPPH 라디칼 소거능은 스테비아(당성분) 첨가량이 많을수록 약간 높게 나타났으며 저장기간 및 온도에 따라 큰 변화가 없는 것으로 보아 영향을 미치지 않았다(도 3).DPPH free radical scavenging activity was measured by modifying the method of Blois. 150 μl of 0.1 mM DPPH solution and 50 μl of sample were mixed and reacted in the dark for 30 minutes, and absorbance was measured at 530 nm. The DPPH radical scavenging ability on the day of manufacture was slightly higher as the amount of stevia (sugar component) added increased, and was not affected as there was no significant change depending on the storage period and temperature (FIG. 3).
실시예Example 5. 5. 독활Poisoning 한방 음료의 SOD 유사 활성 SOD-like activity of herbal beverages
SOD 유사 활성은 시료 0.5 mL, Tris-HCl buffer(pH 8.5) 3 mL, 7.2 mM 피로갈롤 0.2 mL를 혼합하여 25℃에서 10분간 반응시킨 후, 1N HCl 1 mL로 반응을 정지시키고 420 nm에서 흡광도를 측정하였다. SOD 유사 활성 함량은 모든 음료가 저장기간에 따라 꾸준히 증가하였으며, 저장온도에는 큰 영향을 미치지 않는 것으로 나타났다(도 4).SOD-like activity is 0.5 mL of sample, 3 mL of Tris-HCl buffer (pH 8.5), 0.2 mL of 7.2 mM pyrogallol and reacted at 25° C. for 10 minutes, the reaction is stopped with 1 mL of 1N HCl and absorbance at 420 nm. Was measured. The SOD-like active content was steadily increased according to the storage period of all beverages, and it was found that the storage temperature had no significant effect (FIG. 4).
실시예Example 6. 6. 독활Poisoning 한방 음료의 미생물 오염도 Microbial contamination of herbal beverages
무균적으로 시료 1 mL를 채취하여 3M 페트리 필름에 분주한 후 일반세균 및 대장균군을 확인하기 위하여 37℃에서 24~48시간 배양한 후 미생물의 오염 여부를 확인하였다. 일반세균 및 대장균군은 저장기간 동안 음료에서 검출되지 않아 미생물학적으로 안전한 것으로 나타났다(표 5). 따라서 독활 첨가 한방 음료는 식품위생학적으로 안전한 것으로 판단되며 식품 공전상의 기준치(세균수: 1 mL당 100 이하, 대장균군: 음성)에 적합한 것으로 나타났다.After aseptically collecting 1 mL of the sample and dispensing it into a 3M Petri film, it was incubated at 37°C for 24 to 48 hours to check the general bacteria and coliform group, and then checked for contamination of microorganisms. The general bacteria and E. coli group were found to be microbiologically safe because they were not detected in the beverage during the storage period (Table 5). Therefore, it was found that herbal beverages with poisonous additions are considered to be food hygienically safe and suitable for food revolution standards (number of bacteria: 100 or less per 1 mL, E. coli group: negative).
(Log cfu/g)General bacteria
(Log cfu/g)
(Log cfu/g)Coliform
(Log cfu/g)
실시예Example 7. 재료 종류에 따른 7. Depending on the material type 독활Poisoning 한방 음료의 관능검사 Sensory test of herbal beverages
상기 제조예 1의 방법으로 독활 한방 음료를 제조하되, (1)단계의 한약재 혼합물 준비 시 재료 배합비를 표 6의 E 및 F의 재료 및 배합비로 준비한 한약재 혼합물을 이용하여 독활 한방 음료를 각각 제조한 후, 실시예 1과 동일한 방법으로 관능검사를 실시하였다.In the preparation of the herbal herbal drink in the method of Preparation Example 1, (1) when preparing the herbal mixture of the ingredients, each of the herbal herbal mixtures prepared by using the herbal mixture prepared with the ingredients and blending ratio of the ingredients E and F in Table 6, respectively. Subsequently, a sensory test was conducted in the same manner as in Example 1.
그 결과, C 음료와 같이, 독활, 작약, 당귀, 백출, 귤피, 복령, 사삼, 계피, 맥아, 황기, 감초 및 생강의 12가지 한약재만을 사용하여 음료를 제조하는 것이 향, 맛 및 전체적인 기호도가 가장 우수함을 확인할 수 있었다.As a result, like beverage C, the preparation of a drink using only 12 herbal medicines such as poisonous, peony, Angelica, Baekchul, Tangerine, Bokryeong, Sassam, cinnamon, malt, Hwanggi, licorice and ginger has a flavor, taste and overall preference It was confirmed that the most excellent.
Claims (4)
(2) 상기 (1)단계의 준비한 한약재 혼합물에 물을 첨가한 후 추출하는 단계를 포함하여 제조하는 것을 특징으로 하는 독활 한방 음료의 제조방법. (1) preparing a medicinal herb mixture of poison, peony, Angelica, Baekchul, Tangerine, Bokryeong, Ginseng, cinnamon, malt, astragalus, licorice, ginger and stevia; And
(2) The method of manufacturing a toxic herbal beverage comprising the step of extracting after adding water to the mixture of the herbal preparation prepared in the step (1).
(1) 한약재 혼합물 총 중량 기준으로, 독활 13~17 중량%, 작약 13~17 중량%, 당귀 6~9 중량%, 백출 5~7 중량%, 귤피 6~9 중량%, 복령 4~5 중량%, 사삼 5~7 중량%, 계피 2~4 중량%, 맥아 13~17 중량%, 황기 6~9 중량%, 감초 4~5 중량%, 생강 5~6 중량% 및 스테비아 2~4 중량%를 혼합한 한약재 혼합물을 준비하는 단계; 및
(2) 상기 (1)단계의 준비한 한약재 혼합물에 혼합물 대비 물을 80~120배(v/w) 첨가한 후 90~110℃에서 1~3시간 동안 추출하는 단계를 포함하여 제조하는 것을 특징으로 하는 독활 한방 음료의 제조방법. According to claim 2,
(1) Based on the total weight of the herbal mixture, 13~17% by weight, 13~17% by weight, 6~9% by weight, Angelica 5~7% by weight, 6~9% by weight of tangerine, 4~5% by weight of Bokryeong %, Ginseng 5-7% by weight, Cinnamon 2-4% by weight, Malt 13-17% by weight, Astragalus 6-9% by weight, Licorice 4-5% by weight, Ginger 5-6% by weight, and Stevia 2-4% by weight Preparing a mixture of medicinal herbs mixed with; And
(2) characterized in that it comprises the step of extracting for 1 to 3 hours at 90 to 110 ℃ after adding 80 to 120 times (v / w) of water compared to the mixture to the mixture of the herbal preparation prepared in the step (1) Method of manufacturing a unique oriental herbal beverage.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180171995A KR102196680B1 (en) | 2018-12-28 | 2018-12-28 | Method for producing Aralia continentalis herbal drink |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180171995A KR102196680B1 (en) | 2018-12-28 | 2018-12-28 | Method for producing Aralia continentalis herbal drink |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20200081943A true KR20200081943A (en) | 2020-07-08 |
| KR102196680B1 KR102196680B1 (en) | 2020-12-30 |
Family
ID=71600130
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020180171995A KR102196680B1 (en) | 2018-12-28 | 2018-12-28 | Method for producing Aralia continentalis herbal drink |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR102196680B1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20220028746A (en) * | 2020-08-31 | 2022-03-08 | 재단법인 임실치즈앤식품연구소 | Method for producing solid tea using powder of Aralia continentalis mixed extract having immune activity |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100972116B1 (en) * | 2009-10-29 | 2010-07-23 | (주)한국파비스 알엔디 | Method of producing fermentation of herbal medicine, the fermentation produced thereby, and food comprising the fermentation |
| KR20150040392A (en) * | 2013-10-04 | 2015-04-15 | 허윤정 | undiluted stevia liquid and the berages, and its manufacturing method |
-
2018
- 2018-12-28 KR KR1020180171995A patent/KR102196680B1/en active IP Right Grant
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100972116B1 (en) * | 2009-10-29 | 2010-07-23 | (주)한국파비스 알엔디 | Method of producing fermentation of herbal medicine, the fermentation produced thereby, and food comprising the fermentation |
| KR20150040392A (en) * | 2013-10-04 | 2015-04-15 | 허윤정 | undiluted stevia liquid and the berages, and its manufacturing method |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20220028746A (en) * | 2020-08-31 | 2022-03-08 | 재단법인 임실치즈앤식품연구소 | Method for producing solid tea using powder of Aralia continentalis mixed extract having immune activity |
Also Published As
| Publication number | Publication date |
|---|---|
| KR102196680B1 (en) | 2020-12-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Eyduran et al. | Organic acids, sugars, vitamin C, antioxidant capacity, and phenolic compounds in fruits of white (Morus alba L.) and black (Morus nigra L.) mulberry genotypes | |
| KR101127659B1 (en) | Preparation method of jujube wine | |
| KR101817282B1 (en) | Method for producing mixed barley sprout beverage with increased liver function and antioxidant activity | |
| Bisht et al. | Mahua an important Indian species: A review | |
| KR101260466B1 (en) | Preparation method for mother chrysanthemum vinegar | |
| KR100738658B1 (en) | Composition comprising propolis for producing meat seasoning sauce and method thereof | |
| Chawafambira | The effect of incorporating herbal (Lippia javanica) infusion on the phenolic, physicochemical, and sensorial properties of fruit wine | |
| Marahatta et al. | The phytochemical and nutritional analysis and biological activity of Annona squamosa Linn | |
| KR102196680B1 (en) | Method for producing Aralia continentalis herbal drink | |
| CN116869884A (en) | Composition containing lemon peel extract and preparation method and application thereof | |
| Mani et al. | Variability in physico-chemical properties of Indian olive (Elaeocarpus floribundus Blume.) grown under north Bengal conditions | |
| KR20180060215A (en) | A syrup comprising camellia flower extract and a method for preparing the same | |
| KR20180057356A (en) | A liquid tea comprising camellia flower extract and a method for preparaing the same | |
| KR101903633B1 (en) | Method for producing fermented Allium hookeri using blueberry solution fermented by lactic acid bacteria | |
| KR101851996B1 (en) | Method for producing non-heating beverage comprising barley sprout using UV sterilizer | |
| KR100749086B1 (en) | A Method for Preparing Extract of Xanthoxylum piperitum and Anti-cariogenic Products Containing Extract of Xanthoxylum piperitum | |
| KR101958439B1 (en) | Method for producing Hovenia dulcis beverage product comprising Hovenia dulcis mixed concentrate | |
| Lego et al. | Physico-chemical characteristics and sensory evaluation of Sikkim mandarin wine | |
| Pillwan et al. | To study extraction, phytochemical screening and formulation from Stevia rebaudiana bertoni | |
| KR101963839B1 (en) | A jam comprising camellia flower extract and a method for preparing the same | |
| KR100998246B1 (en) | Preparation Method of Kimchi Using Extract of Houttuynia cordata Thunb and Saururus chinensis Baill | |
| Dwivedi et al. | Mahua (Madhuca longifolia) flower and its application in food industry: A review | |
| Luvonga | Nutritional & Phytochemical Composition, Functional Properties of Roselle (Hibiscus sabdariffa) and Sensory Evaluation of Some Beverages made from Roselle Calyces | |
| KR20180047503A (en) | Lacquer seed tea beverage without urushiol and the manufacturing method thereof | |
| Cinkmanis et al. | Analysis of organic acids in herbal and fruit syrups by liquid chromatography. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| E902 | Notification of reason for refusal | ||
| E701 | Decision to grant or registration of patent right | ||
| GRNT | Written decision to grant |