KR20140053419A - 약제학적 조성물 - Google Patents
약제학적 조성물 Download PDFInfo
- Publication number
- KR20140053419A KR20140053419A KR1020147010618A KR20147010618A KR20140053419A KR 20140053419 A KR20140053419 A KR 20140053419A KR 1020147010618 A KR1020147010618 A KR 1020147010618A KR 20147010618 A KR20147010618 A KR 20147010618A KR 20140053419 A KR20140053419 A KR 20140053419A
- Authority
- KR
- South Korea
- Prior art keywords
- pharmaceutical composition
- tablet
- minutes
- composition
- calcitonin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 239000008194 pharmaceutical composition Substances 0.000 title claims abstract description 91
- 238000004090 dissolution Methods 0.000 claims abstract description 57
- 239000000203 mixture Substances 0.000 claims description 112
- XRTHAPZDZPADIL-UHFFFAOYSA-N 8-[(5-chloro-2-hydroxybenzoyl)amino]octanoic acid Chemical compound OC(=O)CCCCCCCNC(=O)C1=CC(Cl)=CC=C1O XRTHAPZDZPADIL-UHFFFAOYSA-N 0.000 claims description 53
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 40
- 229940124447 delivery agent Drugs 0.000 claims description 37
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical group C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 claims description 32
- 108090000445 Parathyroid hormone Proteins 0.000 claims description 27
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical class [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 claims description 25
- 229960000913 crospovidone Drugs 0.000 claims description 21
- 229920000523 polyvinylpolypyrrolidone Polymers 0.000 claims description 21
- 235000013809 polyvinylpolypyrrolidone Nutrition 0.000 claims description 21
- 239000007884 disintegrant Substances 0.000 claims description 19
- 150000001875 compounds Chemical class 0.000 claims description 17
- 239000012453 solvate Substances 0.000 claims description 16
- 239000003085 diluting agent Substances 0.000 claims description 15
- 238000002156 mixing Methods 0.000 claims description 14
- 239000012634 fragment Substances 0.000 claims description 8
- 229920000036 polyvinylpyrrolidone Polymers 0.000 claims description 8
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 claims description 8
- 229940069328 povidone Drugs 0.000 claims description 8
- 125000002947 alkylene group Chemical group 0.000 claims description 7
- 239000002702 enteric coating Substances 0.000 claims description 4
- 238000009505 enteric coating Methods 0.000 claims description 4
- 229910052739 hydrogen Inorganic materials 0.000 claims description 4
- 239000001257 hydrogen Substances 0.000 claims description 4
- 125000004450 alkenylene group Chemical group 0.000 claims description 3
- 125000000217 alkyl group Chemical group 0.000 claims description 3
- 125000000732 arylene group Chemical group 0.000 claims description 3
- 229910052736 halogen Inorganic materials 0.000 claims description 3
- 150000002367 halogens Chemical class 0.000 claims description 3
- 108010073509 parathyroid hormone (1-31) Proteins 0.000 claims description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 2
- 125000000729 N-terminal amino-acid group Chemical group 0.000 claims description 2
- 229940122344 Peptidase inhibitor Drugs 0.000 claims description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 2
- 150000002431 hydrogen Chemical class 0.000 claims description 2
- 229910052760 oxygen Inorganic materials 0.000 claims description 2
- 239000001301 oxygen Substances 0.000 claims description 2
- 108700000288 parathyroid hormone (1-28) Proteins 0.000 claims description 2
- 125000003118 aryl group Chemical group 0.000 claims 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 claims 1
- 125000001424 substituent group Chemical group 0.000 claims 1
- 239000004480 active ingredient Substances 0.000 abstract description 45
- 229920001308 poly(aminoacid) Polymers 0.000 abstract description 35
- 239000003814 drug Substances 0.000 abstract description 28
- 229940079593 drug Drugs 0.000 abstract description 26
- 108090000765 processed proteins & peptides Proteins 0.000 abstract description 23
- 102000004169 proteins and genes Human genes 0.000 abstract description 21
- 108090000623 proteins and genes Proteins 0.000 abstract description 21
- 239000008203 oral pharmaceutical composition Substances 0.000 abstract description 13
- 230000001225 therapeutic effect Effects 0.000 abstract description 11
- 229940088597 hormone Drugs 0.000 abstract description 6
- 239000005556 hormone Substances 0.000 abstract description 6
- 239000008177 pharmaceutical agent Substances 0.000 abstract 1
- BBBFJLBPOGFECG-VJVYQDLKSA-N calcitonin Chemical compound N([C@H](C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N1[C@@H](CCC1)C(N)=O)C(C)C)C(=O)[C@@H]1CSSC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1 BBBFJLBPOGFECG-VJVYQDLKSA-N 0.000 description 79
- 239000003826 tablet Substances 0.000 description 52
- 238000000034 method Methods 0.000 description 43
- 108010068072 salmon calcitonin Proteins 0.000 description 42
- 102000055006 Calcitonin Human genes 0.000 description 39
- 108060001064 Calcitonin Proteins 0.000 description 39
- 229960004015 calcitonin Drugs 0.000 description 38
- 229960003773 calcitonin (salmon synthetic) Drugs 0.000 description 38
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 24
- 150000003839 salts Chemical class 0.000 description 21
- 102100036893 Parathyroid hormone Human genes 0.000 description 17
- 239000013543 active substance Substances 0.000 description 17
- 239000003795 chemical substances by application Substances 0.000 description 17
- 238000010521 absorption reaction Methods 0.000 description 15
- 239000000314 lubricant Substances 0.000 description 14
- 239000007787 solid Substances 0.000 description 14
- -1 For example Proteins 0.000 description 13
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 12
- 239000004615 ingredient Substances 0.000 description 12
- 235000019359 magnesium stearate Nutrition 0.000 description 12
- 230000036470 plasma concentration Effects 0.000 description 12
- 201000010099 disease Diseases 0.000 description 11
- 230000000694 effects Effects 0.000 description 10
- 238000009472 formulation Methods 0.000 description 10
- 108010000521 Human Growth Hormone Proteins 0.000 description 9
- 102000002265 Human Growth Hormone Human genes 0.000 description 9
- 239000000854 Human Growth Hormone Substances 0.000 description 9
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 9
- 239000002245 particle Substances 0.000 description 9
- 102000004196 processed proteins & peptides Human genes 0.000 description 9
- 229920003084 Avicel® PH-102 Polymers 0.000 description 8
- 230000002917 arthritic effect Effects 0.000 description 8
- 239000002775 capsule Substances 0.000 description 8
- 239000011734 sodium Substances 0.000 description 8
- 238000011282 treatment Methods 0.000 description 8
- 241000282693 Cercopithecidae Species 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- 108010051696 Growth Hormone Proteins 0.000 description 6
- 102000018997 Growth Hormone Human genes 0.000 description 6
- 208000001132 Osteoporosis Diseases 0.000 description 6
- 239000000199 parathyroid hormone Substances 0.000 description 6
- 229910052708 sodium Inorganic materials 0.000 description 6
- 241000972773 Aulopiformes Species 0.000 description 5
- 229940078581 Bone resorption inhibitor Drugs 0.000 description 5
- 102000003982 Parathyroid hormone Human genes 0.000 description 5
- 102000015731 Peptide Hormones Human genes 0.000 description 5
- 108010038988 Peptide Hormones Proteins 0.000 description 5
- 150000001413 amino acids Chemical group 0.000 description 5
- 210000000988 bone and bone Anatomy 0.000 description 5
- 239000002552 dosage form Substances 0.000 description 5
- 210000001035 gastrointestinal tract Anatomy 0.000 description 5
- 239000000122 growth hormone Substances 0.000 description 5
- 238000004519 manufacturing process Methods 0.000 description 5
- 201000008482 osteoarthritis Diseases 0.000 description 5
- 239000000813 peptide hormone Substances 0.000 description 5
- 238000002360 preparation method Methods 0.000 description 5
- 235000019515 salmon Nutrition 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- 208000006386 Bone Resorption Diseases 0.000 description 4
- 208000010191 Osteitis Deformans Diseases 0.000 description 4
- 208000027868 Paget disease Diseases 0.000 description 4
- 241000288906 Primates Species 0.000 description 4
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 230000024279 bone resorption Effects 0.000 description 4
- 238000007906 compression Methods 0.000 description 4
- 230000006835 compression Effects 0.000 description 4
- 238000001035 drying Methods 0.000 description 4
- 208000027202 mammary Paget disease Diseases 0.000 description 4
- 239000000137 peptide hydrolase inhibitor Substances 0.000 description 4
- 210000002784 stomach Anatomy 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 229910002016 Aerosil® 200 Inorganic materials 0.000 description 3
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 3
- 101000741445 Homo sapiens Calcitonin Proteins 0.000 description 3
- 241000282560 Macaca mulatta Species 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 3
- 230000002159 abnormal effect Effects 0.000 description 3
- 239000011575 calcium Substances 0.000 description 3
- 229910052791 calcium Inorganic materials 0.000 description 3
- 210000000845 cartilage Anatomy 0.000 description 3
- 230000015556 catabolic process Effects 0.000 description 3
- 239000007891 compressed tablet Substances 0.000 description 3
- 238000006731 degradation reaction Methods 0.000 description 3
- XGSDAQOSOKPJML-UHFFFAOYSA-L disodium;8-[(5-chloro-2-oxidobenzoyl)amino]octanoate Chemical compound [Na+].[Na+].[O-]C(=O)CCCCCCCNC(=O)C1=CC(Cl)=CC=C1[O-] XGSDAQOSOKPJML-UHFFFAOYSA-L 0.000 description 3
- 239000006185 dispersion Substances 0.000 description 3
- 239000002706 dry binder Substances 0.000 description 3
- 229940045644 human calcitonin Drugs 0.000 description 3
- 150000004677 hydrates Chemical class 0.000 description 3
- 239000003112 inhibitor Substances 0.000 description 3
- 230000000968 intestinal effect Effects 0.000 description 3
- 239000011777 magnesium Substances 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 239000002609 medium Substances 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 239000002002 slurry Substances 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- 230000004936 stimulating effect Effects 0.000 description 3
- OGBMKVWORPGQRR-UMXFMPSGSA-N teriparatide Chemical compound C([C@H](NC(=O)[C@H](CCSC)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@@H](N)CO)C(C)C)[C@@H](C)CC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1N=CNC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1N=CNC=1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(O)=O)C1=CNC=N1 OGBMKVWORPGQRR-UMXFMPSGSA-N 0.000 description 3
- 208000020084 Bone disease Diseases 0.000 description 2
- 229940122156 Cathepsin K inhibitor Drugs 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 239000000095 Growth Hormone-Releasing Hormone Substances 0.000 description 2
- 102000038461 Growth Hormone-Releasing Hormone Human genes 0.000 description 2
- 206010062767 Hypophysitis Diseases 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 241000316144 Macrodon ancylodon Species 0.000 description 2
- 208000003076 Osteolysis Diseases 0.000 description 2
- 102000057297 Pepsin A Human genes 0.000 description 2
- 108090000284 Pepsin A Proteins 0.000 description 2
- 101710142969 Somatoliberin Proteins 0.000 description 2
- AUYYCJSJGJYCDS-LBPRGKRZSA-N Thyrolar Chemical class IC1=CC(C[C@H](N)C(O)=O)=CC(I)=C1OC1=CC=C(O)C(I)=C1 AUYYCJSJGJYCDS-LBPRGKRZSA-N 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- 208000019664 bone resorption disease Diseases 0.000 description 2
- 108010006025 bovine growth hormone Proteins 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 229940126523 co-drug Drugs 0.000 description 2
- 229940111134 coxibs Drugs 0.000 description 2
- 239000003255 cyclooxygenase 2 inhibitor Substances 0.000 description 2
- 229940088598 enzyme Drugs 0.000 description 2
- 229940011871 estrogen Drugs 0.000 description 2
- 239000000262 estrogen Substances 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 230000002496 gastric effect Effects 0.000 description 2
- 230000036571 hydration Effects 0.000 description 2
- 238000006703 hydration reaction Methods 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- 210000000936 intestine Anatomy 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- 229960000994 lumiracoxib Drugs 0.000 description 2
- KHPKQFYUPIUARC-UHFFFAOYSA-N lumiracoxib Chemical compound OC(=O)CC1=CC(C)=CC=C1NC1=C(F)C=CC=C1Cl KHPKQFYUPIUARC-UHFFFAOYSA-N 0.000 description 2
- 208000029791 lytic metastatic bone lesion Diseases 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 230000004060 metabolic process Effects 0.000 description 2
- 150000004682 monohydrates Chemical class 0.000 description 2
- 239000006186 oral dosage form Substances 0.000 description 2
- 230000011164 ossification Effects 0.000 description 2
- 229960001319 parathyroid hormone Drugs 0.000 description 2
- 108010073230 parathyroid hormone (1-38) Proteins 0.000 description 2
- 229940111202 pepsin Drugs 0.000 description 2
- 230000001817 pituitary effect Effects 0.000 description 2
- 210000003635 pituitary gland Anatomy 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 230000017854 proteolysis Effects 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 238000007670 refining Methods 0.000 description 2
- RZJQGNCSTQAWON-UHFFFAOYSA-N rofecoxib Chemical compound C1=CC(S(=O)(=O)C)=CC=C1C1=C(C=2C=CC=CC=2)C(=O)OC1 RZJQGNCSTQAWON-UHFFFAOYSA-N 0.000 description 2
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 2
- 159000000000 sodium salts Chemical class 0.000 description 2
- XUHVCHNJCBBXMP-UHFFFAOYSA-M sodium;10-[(2-hydroxybenzoyl)amino]decanoate Chemical compound [Na+].OC1=CC=CC=C1C(=O)NCCCCCCCCCC([O-])=O XUHVCHNJCBBXMP-UHFFFAOYSA-M 0.000 description 2
- UOENJXXSKABLJL-UHFFFAOYSA-M sodium;8-[(2-hydroxybenzoyl)amino]octanoate Chemical compound [Na+].OC1=CC=CC=C1C(=O)NCCCCCCCC([O-])=O UOENJXXSKABLJL-UHFFFAOYSA-M 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 238000013268 sustained release Methods 0.000 description 2
- 239000012730 sustained-release form Substances 0.000 description 2
- 229920001059 synthetic polymer Polymers 0.000 description 2
- 239000005495 thyroid hormone Substances 0.000 description 2
- 229940036555 thyroid hormone Drugs 0.000 description 2
- 241000251468 Actinopterygii Species 0.000 description 1
- OGSPWJRAVKPPFI-UHFFFAOYSA-N Alendronic Acid Chemical compound NCCCC(O)(P(O)(O)=O)P(O)(O)=O OGSPWJRAVKPPFI-UHFFFAOYSA-N 0.000 description 1
- 102100032187 Androgen receptor Human genes 0.000 description 1
- 229930192334 Auxin Natural products 0.000 description 1
- 229940122361 Bisphosphonate Drugs 0.000 description 1
- KSIYPKPZIBBUFR-LJNLPFSOSA-N CSCC[C@H](NC(=O)CNC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@@H]1CSSC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1)C(C)C)C(=O)NCC(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1CCC[C@H]1C(N)=O Chemical compound CSCC[C@H](NC(=O)CNC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@@H]1CSSC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1)C(C)C)C(=O)NCC(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1CCC[C@H]1C(N)=O KSIYPKPZIBBUFR-LJNLPFSOSA-N 0.000 description 1
- 102000004171 Cathepsin K Human genes 0.000 description 1
- 108090000625 Cathepsin K Proteins 0.000 description 1
- 229940123003 Cathepsin inhibitor Drugs 0.000 description 1
- 102000005600 Cathepsins Human genes 0.000 description 1
- 108010084457 Cathepsins Proteins 0.000 description 1
- 102000029816 Collagenase Human genes 0.000 description 1
- 108060005980 Collagenase Proteins 0.000 description 1
- 229920002683 Glycosaminoglycan Polymers 0.000 description 1
- 208000037147 Hypercalcaemia Diseases 0.000 description 1
- 206010020584 Hypercalcaemia of malignancy Diseases 0.000 description 1
- MPBVHIBUJCELCL-UHFFFAOYSA-N Ibandronate Chemical compound CCCCCN(C)CCC(O)(P(O)(O)=O)P(O)(O)=O MPBVHIBUJCELCL-UHFFFAOYSA-N 0.000 description 1
- 108090000723 Insulin-Like Growth Factor I Proteins 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 102100037611 Lysophospholipase Human genes 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 108700020797 Parathyroid Hormone-Related Proteins 0.000 description 1
- 102000043299 Parathyroid hormone-related Human genes 0.000 description 1
- 102000035195 Peptidases Human genes 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 108010058864 Phospholipases A2 Proteins 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 102100038277 Prostaglandin G/H synthase 1 Human genes 0.000 description 1
- 108050003243 Prostaglandin G/H synthase 1 Proteins 0.000 description 1
- 229940124158 Protease/peptidase inhibitor Drugs 0.000 description 1
- IIDJRNMFWXDHID-UHFFFAOYSA-N Risedronic acid Chemical compound OP(=O)(O)C(P(O)(O)=O)(O)CC1=CC=CN=C1 IIDJRNMFWXDHID-UHFFFAOYSA-N 0.000 description 1
- 241000277263 Salmo Species 0.000 description 1
- 102000013275 Somatomedins Human genes 0.000 description 1
- 101000910302 Sus scrofa Calcitonin Proteins 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- 229930003316 Vitamin D Natural products 0.000 description 1
- QYSXJUFSXHHAJI-XFEUOLMDSA-N Vitamin D3 Natural products C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C/C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-XFEUOLMDSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 229940062527 alendronate Drugs 0.000 description 1
- 108010080146 androgen receptors Proteins 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000002363 auxin Substances 0.000 description 1
- 229960000817 bazedoxifene Drugs 0.000 description 1
- UCJGJABZCDBEDK-UHFFFAOYSA-N bazedoxifene Chemical compound C=1C=C(OCCN2CCCCCC2)C=CC=1CN1C2=CC=C(O)C=C2C(C)=C1C1=CC=C(O)C=C1 UCJGJABZCDBEDK-UHFFFAOYSA-N 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 150000004663 bisphosphonates Chemical class 0.000 description 1
- 230000036765 blood level Effects 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 239000010839 body fluid Substances 0.000 description 1
- 230000004097 bone metabolism Effects 0.000 description 1
- 230000003913 calcium metabolism Effects 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 210000003321 cartilage cell Anatomy 0.000 description 1
- 238000011260 co-administration Methods 0.000 description 1
- 229960002424 collagenase Drugs 0.000 description 1
- 238000000748 compression moulding Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 239000003260 cyclooxygenase 1 inhibitor Substances 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- GHVNFZFCNZKVNT-UHFFFAOYSA-M decanoate Chemical compound CCCCCCCCCC([O-])=O GHVNFZFCNZKVNT-UHFFFAOYSA-M 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 230000005786 degenerative changes Effects 0.000 description 1
- DCOPUUMXTXDBNB-UHFFFAOYSA-N diclofenac Chemical compound OC(=O)CC1=CC=CC=C1NC1=C(Cl)C=CC=C1Cl DCOPUUMXTXDBNB-UHFFFAOYSA-N 0.000 description 1
- 229960001259 diclofenac Drugs 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 229940042399 direct acting antivirals protease inhibitors Drugs 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 239000012738 dissolution medium Substances 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 239000008298 dragée Substances 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
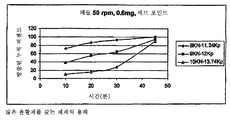
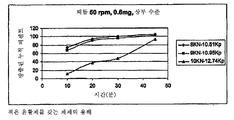
- 238000009506 drug dissolution testing Methods 0.000 description 1
- 229940126534 drug product Drugs 0.000 description 1
- 230000002183 duodenal effect Effects 0.000 description 1
- 210000001198 duodenum Anatomy 0.000 description 1
- VSHJAJRPRRNBEK-LMVCGNDWSA-N eel calcitonin Chemical compound C([C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCCN)NC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CS)[C@@H](C)O)C(C)C)CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)NCC(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N1[C@@H](CCC1)C(N)=O)C1=CN=CN1 VSHJAJRPRRNBEK-LMVCGNDWSA-N 0.000 description 1
- 210000002308 embryonic cell Anatomy 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 230000007515 enzymatic degradation Effects 0.000 description 1
- MVPICKVDHDWCJQ-UHFFFAOYSA-N ethyl 3-pyrrolidin-1-ylpropanoate Chemical compound CCOC(=O)CCN1CCCC1 MVPICKVDHDWCJQ-UHFFFAOYSA-N 0.000 description 1
- 229960004945 etoricoxib Drugs 0.000 description 1
- MNJVRJDLRVPLFE-UHFFFAOYSA-N etoricoxib Chemical compound C1=NC(C)=CC=C1C1=NC=C(Cl)C=C1C1=CC=C(S(C)(=O)=O)C=C1 MNJVRJDLRVPLFE-UHFFFAOYSA-N 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 235000019688 fish Nutrition 0.000 description 1
- 238000005187 foaming Methods 0.000 description 1
- 238000003304 gavage Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 229920001519 homopolymer Polymers 0.000 description 1
- 208000008750 humoral hypercalcemia of malignancy Diseases 0.000 description 1
- 230000000887 hydrating effect Effects 0.000 description 1
- 230000000148 hypercalcaemia Effects 0.000 description 1
- 208000030915 hypercalcemia disease Diseases 0.000 description 1
- 229940015872 ibandronate Drugs 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- SEOVTRFCIGRIMH-UHFFFAOYSA-N indole-3-acetic acid Chemical compound C1=CC=C2C(CC(=O)O)=CNC2=C1 SEOVTRFCIGRIMH-UHFFFAOYSA-N 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000004898 kneading Methods 0.000 description 1
- 238000009533 lab test Methods 0.000 description 1
- GXESHMAMLJKROZ-IAPPQJPRSA-N lasofoxifene Chemical compound C1([C@@H]2[C@@H](C3=CC=C(C=C3CC2)O)C=2C=CC(OCCN3CCCC3)=CC=2)=CC=CC=C1 GXESHMAMLJKROZ-IAPPQJPRSA-N 0.000 description 1
- 229960002367 lasofoxifene Drugs 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 229940126601 medicinal product Drugs 0.000 description 1
- 239000003475 metalloproteinase inhibitor Substances 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 230000021332 multicellular organism growth Effects 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- WWZKQHOCKIZLMA-UHFFFAOYSA-M octanoate Chemical compound CCCCCCCC([O-])=O WWZKQHOCKIZLMA-UHFFFAOYSA-M 0.000 description 1
- 230000002188 osteogenic effect Effects 0.000 description 1
- 238000010647 peptide synthesis reaction Methods 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 230000003285 pharmacodynamic effect Effects 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 229920000191 poly(N-vinyl pyrrolidone) Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 208000001685 postmenopausal osteoporosis Diseases 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 239000011164 primary particle Substances 0.000 description 1
- 235000019833 protease Nutrition 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 238000003127 radioimmunoassay Methods 0.000 description 1
- 229960004622 raloxifene Drugs 0.000 description 1
- GZUITABIAKMVPG-UHFFFAOYSA-N raloxifene Chemical compound C1=CC(O)=CC=C1C1=C(C(=O)C=2C=CC(OCCN3CCCCC3)=CC=2)C2=CC=C(O)C=C2S1 GZUITABIAKMVPG-UHFFFAOYSA-N 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 229940089617 risedronate Drugs 0.000 description 1
- 229960000371 rofecoxib Drugs 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 229940045902 sodium stearyl fumarate Drugs 0.000 description 1
- 239000008279 sol Substances 0.000 description 1
- 239000007909 solid dosage form Substances 0.000 description 1
- 239000008247 solid mixture Substances 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 239000003270 steroid hormone Substances 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 229920002994 synthetic fiber Polymers 0.000 description 1
- 230000001839 systemic circulation Effects 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 239000007916 tablet composition Substances 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 210000001685 thyroid gland Anatomy 0.000 description 1
- WZDGZWOAQTVYBX-XOINTXKNSA-N tibolone Chemical compound C([C@@H]12)C[C@]3(C)[C@@](C#C)(O)CC[C@H]3[C@@H]1[C@H](C)CC1=C2CCC(=O)C1 WZDGZWOAQTVYBX-XOINTXKNSA-N 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 238000005292 vacuum distillation Methods 0.000 description 1
- 229940087652 vioxx Drugs 0.000 description 1
- 235000019166 vitamin D Nutrition 0.000 description 1
- 239000011710 vitamin D Substances 0.000 description 1
- 150000003710 vitamin D derivatives Chemical class 0.000 description 1
- 229940046008 vitamin d Drugs 0.000 description 1
- XRASPMIURGNCCH-UHFFFAOYSA-N zoledronic acid Chemical compound OP(=O)(O)C(P(O)(O)=O)(O)CN1C=CN=C1 XRASPMIURGNCCH-UHFFFAOYSA-N 0.000 description 1
- 229960004276 zoledronic acid Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/195—Carboxylic acids, e.g. valproic acid having an amino group
- A61K31/197—Carboxylic acids, e.g. valproic acid having an amino group the amino and the carboxyl groups being attached to the same acyclic carbon chain, e.g. gamma-aminobutyric acid [GABA], beta-alanine, epsilon-aminocaproic acid or pantothenic acid
- A61K31/198—Alpha-amino acids, e.g. alanine or edetic acid [EDTA]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/23—Calcitonins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/29—Parathyroid hormone, i.e. parathormone; Parathyroid hormone-related peptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2095—Tabletting processes; Dosage units made by direct compression of powders or specially processed granules, by eliminating solvents, by melt-extrusion, by injection molding, by 3D printing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/06—Drugs for disorders of the endocrine system of the anterior pituitary hormones, e.g. TSH, ACTH, FSH, LH, PRL, GH
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Endocrinology (AREA)
- Physical Education & Sports Medicine (AREA)
- Immunology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Rheumatology (AREA)
- Biophysics (AREA)
- Molecular Biology (AREA)
- Diabetes (AREA)
- Medicinal Preparation (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US73763105P | 2005-11-17 | 2005-11-17 | |
| US60/737,631 | 2005-11-17 | ||
| PCT/US2006/044642 WO2007061829A2 (en) | 2005-11-17 | 2006-11-16 | Pharmaceutical composition |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020087011726A Division KR101432313B1 (ko) | 2005-11-17 | 2006-11-16 | 약제학적 조성물 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20140053419A true KR20140053419A (ko) | 2014-05-07 |
Family
ID=37875983
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020147010618A Ceased KR20140053419A (ko) | 2005-11-17 | 2006-11-16 | 약제학적 조성물 |
| KR1020087011726A Expired - Fee Related KR101432313B1 (ko) | 2005-11-17 | 2006-11-16 | 약제학적 조성물 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020087011726A Expired - Fee Related KR101432313B1 (ko) | 2005-11-17 | 2006-11-16 | 약제학적 조성물 |
Country Status (10)
| Country | Link |
|---|---|
| US (3) | US20080255048A1 (OSRAM) |
| EP (1) | EP1951207A2 (OSRAM) |
| JP (3) | JP2009515993A (OSRAM) |
| KR (2) | KR20140053419A (OSRAM) |
| CN (1) | CN101309674A (OSRAM) |
| AU (1) | AU2006318815B2 (OSRAM) |
| BR (1) | BRPI0618723A2 (OSRAM) |
| CA (1) | CA2626933C (OSRAM) |
| RU (1) | RU2469709C2 (OSRAM) |
| WO (1) | WO2007061829A2 (OSRAM) |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8497258B2 (en) | 2005-11-12 | 2013-07-30 | The Regents Of The University Of California | Viscous budesonide for the treatment of inflammatory diseases of the gastrointestinal tract |
| JP2009515993A (ja) * | 2005-11-17 | 2009-04-16 | ノバルティス アクチエンゲゼルシャフト | 医薬組成物 |
| EP2440210A4 (en) | 2009-06-12 | 2014-01-29 | Meritage Pharma Inc | METHOD FOR THE TREATMENT OF STOMACH DARM DISEASES |
| RS60321B1 (sr) | 2010-12-16 | 2020-07-31 | Novo Nordisk As | Čvrste kompozicije koje sadrže glp-1 agonist i so n-(8-(2- hidroksibenzoil)amino)kaprilne kiseline |
| DK2696687T3 (en) | 2011-04-12 | 2017-02-06 | Novo Nordisk As | Double-acylated GLP-1 derivatives |
| RS58636B1 (sr) | 2012-03-22 | 2019-05-31 | Novo Nordisk As | Kompozicije koje sadrže sredstvo za isporuku i njihova priprema |
| HUE042757T2 (hu) | 2012-03-22 | 2019-07-29 | Novo Nordisk As | Szállító szert tartalmazó készítmények és elõállításuk |
| PL2827885T3 (pl) | 2012-03-22 | 2019-01-31 | Novo Nordisk A/S | Kompozycje peptydów GLP-1 i ich otrzymywanie |
| WO2013189988A1 (en) | 2012-06-20 | 2013-12-27 | Novo Nordisk A/S | Tablet formulation comprising a peptide and a delivery agent |
| KR20210086717A (ko) | 2013-05-02 | 2021-07-08 | 노보 노르디스크 에이/에스 | Glp-1 화합물의 경구 투여 |
| CN104095828A (zh) * | 2014-07-29 | 2014-10-15 | 中国药科大学 | 一种降钙素口服肠溶组合物及其制备方法 |
| WO2016128974A1 (en) | 2015-02-09 | 2016-08-18 | Entera Bio Ltd. | Formulations for oral administration of active agents |
| WO2018033927A1 (en) * | 2016-08-17 | 2018-02-22 | Entera Bio Ltd. | Formulations for oral administration of active agents |
| CN111683676B (zh) | 2018-02-02 | 2024-06-18 | 诺和诺德股份有限公司 | 包含glp-1激动剂、n-(8-(2-羟基苯甲酰基)氨基)辛酸的盐和润滑剂的固体组合物 |
| CN111116684B (zh) * | 2019-12-31 | 2020-09-25 | 厦门甘宝利生物医药有限公司 | 一种具有甲状腺激素受体激动剂特性的肝靶向化合物及其药物组合物 |
Family Cites Families (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US647A (en) | 1838-03-21 | Improved fishing-seine for deep water | ||
| US608618A (en) | 1898-08-09 | Thomas millen | ||
| US5866A (en) | 1848-10-17 | Dentist s deill | ||
| US536A (en) | 1837-12-26 | Beady binder for binding newspapers | ||
| US5773A (en) | 1848-09-19 | Sizing and drying cotton-batting | ||
| AU508480B2 (en) * | 1977-04-13 | 1980-03-20 | Asahi Kasei Kogyo Kabushiki Kaisha | Microcrystalline cellulose excipient and pharmaceutical composition containing thesame |
| DE3524572A1 (de) * | 1985-07-10 | 1987-01-15 | Thomae Gmbh Dr K | Feste arzneimittelformen zur peroralen anwendung enthaltend 9-deoxo-11-deoxy-9,11-(imino(2-(2-methoxyethoxy)ethyliden)-oxy)-(9s)-erythromycin und verfahren zu ihrer herstellung |
| US5096714A (en) * | 1988-06-28 | 1992-03-17 | Hauser-Kuhrts, Inc. | Prolonged release drug tablet formulations |
| JP2609022B2 (ja) * | 1990-11-29 | 1997-05-14 | 北陸製薬株式会社 | ポリカルボフィルカルシウム含有製剤 |
| US5359030A (en) | 1993-05-10 | 1994-10-25 | Protein Delivery, Inc. | Conjugation-stabilized polypeptide compositions, therapeutic delivery and diagnostic formulations comprising same, and method of making and using the same |
| US5681811A (en) | 1993-05-10 | 1997-10-28 | Protein Delivery, Inc. | Conjugation-stabilized therapeutic agent compositions, delivery and diagnostic formulations comprising same, and method of making and using the same |
| US6191105B1 (en) | 1993-05-10 | 2001-02-20 | Protein Delivery, Inc. | Hydrophilic and lipophilic balanced microemulsion formulations of free-form and/or conjugation-stabilized therapeutic agents such as insulin |
| EA000361B1 (ru) * | 1995-02-08 | 1999-06-24 | Яманучи Юроп Б.В. | Способ приготовления гранулята, содержащего активный при пероральном введении бета-лактамный антибиотик |
| TW442301B (en) * | 1995-06-07 | 2001-06-23 | Sanofi Synthelabo | Pharmaceutical compositions containing irbesartan |
| US5912014A (en) | 1996-03-15 | 1999-06-15 | Unigene Laboratories, Inc. | Oral salmon calcitonin pharmaceutical products |
| US6361794B1 (en) * | 1996-06-12 | 2002-03-26 | Basf Corporation | Method of making ibuprofen and narcotic analgesic composition |
| EP1175390B1 (en) | 1999-04-05 | 2005-02-02 | Emisphere Technologies, Inc. | Disodium salts, monohydrates, and ethanol solvates |
| US6309633B1 (en) | 1999-06-19 | 2001-10-30 | Nobex Corporation | Amphiphilic drug-oligomer conjugates with hydroyzable lipophile components and methods for making and using the same |
| US6380405B1 (en) | 1999-09-13 | 2002-04-30 | Nobex Corporation | Taxane prodrugs |
| US6436990B1 (en) | 1999-10-27 | 2002-08-20 | Nobex Corporation | 6-methoxy-2-naphthylacetic acid prodrugs |
| WO2002018330A1 (en) | 2000-08-29 | 2002-03-07 | Nobex Corporation | 5-asa derivatives having anti-inflammatory and antibiotic activity and methods of treating diseases therewith |
| US7049283B2 (en) | 2000-12-06 | 2006-05-23 | Novartis Ag | Pharmaceutical compositions for the oral delivery of pharmacologically active agents |
| US6479692B1 (en) | 2001-05-02 | 2002-11-12 | Nobex Corporation | Methods of synthesizing acylanilides including bicalutamide and derivatives thereof |
| ES2282436T3 (es) * | 2001-06-01 | 2007-10-16 | Novartis Ag | Administracion oral de hormona paratiroide y calcitonina. |
| US20050054557A1 (en) * | 2002-05-09 | 2005-03-10 | Goldberg Michael M. | Compositions for delivering parathyroid hormone and calcitonin |
| CA2498410A1 (en) * | 2002-09-13 | 2004-03-25 | Cydex, Inc. | Capsules containing aqueous fill compositions stabilized with derivatized cyclodextrin |
| EP1643978A1 (en) * | 2003-07-04 | 2006-04-12 | Nycomed Danmark A/S | Parathyroid hormone (pth) containing pharmaceutical compositions for oral use |
| BRPI0412265A (pt) | 2003-07-23 | 2006-09-05 | Novartis Ag | uso de calcitonina em osteoartrite |
| GB0422644D0 (en) * | 2004-10-12 | 2004-11-10 | Novartis Ag | Organic compounds |
| JP2009515993A (ja) * | 2005-11-17 | 2009-04-16 | ノバルティス アクチエンゲゼルシャフト | 医薬組成物 |
-
2006
- 2006-11-16 JP JP2008541365A patent/JP2009515993A/ja not_active Withdrawn
- 2006-11-16 AU AU2006318815A patent/AU2006318815B2/en not_active Ceased
- 2006-11-16 BR BRPI0618723-4A patent/BRPI0618723A2/pt not_active IP Right Cessation
- 2006-11-16 CA CA2626933A patent/CA2626933C/en not_active Expired - Fee Related
- 2006-11-16 CN CNA2006800429006A patent/CN101309674A/zh active Pending
- 2006-11-16 RU RU2008123380/15A patent/RU2469709C2/ru not_active IP Right Cessation
- 2006-11-16 WO PCT/US2006/044642 patent/WO2007061829A2/en not_active Ceased
- 2006-11-16 KR KR1020147010618A patent/KR20140053419A/ko not_active Ceased
- 2006-11-16 KR KR1020087011726A patent/KR101432313B1/ko not_active Expired - Fee Related
- 2006-11-16 US US12/093,383 patent/US20080255048A1/en not_active Abandoned
- 2006-11-16 EP EP06837887A patent/EP1951207A2/en not_active Withdrawn
-
2011
- 2011-04-04 US US13/079,400 patent/US20110218148A1/en not_active Abandoned
-
2012
- 2012-06-14 JP JP2012135063A patent/JP6049317B2/ja not_active Expired - Fee Related
-
2015
- 2015-04-08 JP JP2015079415A patent/JP6147290B2/ja not_active Expired - Fee Related
- 2015-05-21 US US14/718,595 patent/US9622975B2/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| BRPI0618723A2 (pt) | 2011-09-06 |
| US9622975B2 (en) | 2017-04-18 |
| CA2626933C (en) | 2015-12-29 |
| JP2012211159A (ja) | 2012-11-01 |
| WO2007061829A2 (en) | 2007-05-31 |
| US20110218148A1 (en) | 2011-09-08 |
| CA2626933A1 (en) | 2007-05-31 |
| KR20080066958A (ko) | 2008-07-17 |
| JP6147290B2 (ja) | 2017-06-14 |
| JP6049317B2 (ja) | 2016-12-21 |
| KR101432313B1 (ko) | 2014-08-20 |
| AU2006318815B2 (en) | 2010-08-19 |
| EP1951207A2 (en) | 2008-08-06 |
| WO2007061829A3 (en) | 2007-08-30 |
| US20080255048A1 (en) | 2008-10-16 |
| CN101309674A (zh) | 2008-11-19 |
| JP2015145410A (ja) | 2015-08-13 |
| RU2469709C2 (ru) | 2012-12-20 |
| RU2008123380A (ru) | 2009-12-27 |
| AU2006318815A1 (en) | 2007-05-31 |
| JP2009515993A (ja) | 2009-04-16 |
| US20150250729A1 (en) | 2015-09-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9622975B2 (en) | Pharmaceutical composition | |
| AU2008202879B2 (en) | Use of calcitonin in osteoarthritis | |
| EP1945243B1 (en) | Use of calcitonin for the treatment of ra | |
| MX2008006319A (en) | Pharmaceutical composition | |
| HK1152229B (en) | Use of calcitonin in osteoarthritis | |
| HK1089935B (en) | Use of calcitonin in osteoarthritis |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A107 | Divisional application of patent | ||
| PA0104 | Divisional application for international application |
Comment text: Divisional Application for International Patent Patent event code: PA01041R01D Patent event date: 20140421 Application number text: 1020087011726 Filing date: 20080516 |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20140521 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20140812 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20150107 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20140812 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |