JP7586832B2 - その場調整可能な眼内レンズ - Google Patents
その場調整可能な眼内レンズ Download PDFInfo
- Publication number
- JP7586832B2 JP7586832B2 JP2021559326A JP2021559326A JP7586832B2 JP 7586832 B2 JP7586832 B2 JP 7586832B2 JP 2021559326 A JP2021559326 A JP 2021559326A JP 2021559326 A JP2021559326 A JP 2021559326A JP 7586832 B2 JP7586832 B2 JP 7586832B2
- Authority
- JP
- Japan
- Prior art keywords
- iol
- lcn
- haptic
- haptics
- optic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/14—Eye parts, e.g. lenses or corneal implants; Artificial eyes
- A61F2/16—Intraocular lenses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting in contact-lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/0079—Methods or devices for eye surgery using non-laser electromagnetic radiation, e.g. non-coherent light or microwaves
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting in contact-lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/008—Methods or devices for eye surgery using laser
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/16—Macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/26—Mixtures of macromolecular compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L33/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- C08L33/04—Homopolymers or copolymers of esters
- C08L33/14—Homopolymers or copolymers of esters of esters containing halogen, nitrogen, sulfur, or oxygen atoms in addition to the carboxy oxygen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/14—Eye parts, e.g. lenses or corneal implants; Artificial eyes
- A61F2/16—Intraocular lenses
- A61F2/1613—Intraocular lenses having special lens configurations, e.g. multipart lenses; having particular optical properties, e.g. pseudo-accommodative lenses, lenses having aberration corrections, diffractive lenses, lenses for variably absorbing electromagnetic radiation, lenses having variable focus
- A61F2/1624—Intraocular lenses having special lens configurations, e.g. multipart lenses; having particular optical properties, e.g. pseudo-accommodative lenses, lenses having aberration corrections, diffractive lenses, lenses for variably absorbing electromagnetic radiation, lenses having variable focus having adjustable focus; power activated variable focus means, e.g. mechanically or electrically by the ciliary muscle or from the outside
- A61F2/1629—Intraocular lenses having special lens configurations, e.g. multipart lenses; having particular optical properties, e.g. pseudo-accommodative lenses, lenses having aberration corrections, diffractive lenses, lenses for variably absorbing electromagnetic radiation, lenses having variable focus having adjustable focus; power activated variable focus means, e.g. mechanically or electrically by the ciliary muscle or from the outside for changing longitudinal position, i.e. along the visual axis when implanted
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/14—Eye parts, e.g. lenses or corneal implants; Artificial eyes
- A61F2/16—Intraocular lenses
- A61F2/1613—Intraocular lenses having special lens configurations, e.g. multipart lenses; having particular optical properties, e.g. pseudo-accommodative lenses, lenses having aberration corrections, diffractive lenses, lenses for variably absorbing electromagnetic radiation, lenses having variable focus
- A61F2/1624—Intraocular lenses having special lens configurations, e.g. multipart lenses; having particular optical properties, e.g. pseudo-accommodative lenses, lenses having aberration corrections, diffractive lenses, lenses for variably absorbing electromagnetic radiation, lenses having variable focus having adjustable focus; power activated variable focus means, e.g. mechanically or electrically by the ciliary muscle or from the outside
- A61F2/1632—Intraocular lenses having special lens configurations, e.g. multipart lenses; having particular optical properties, e.g. pseudo-accommodative lenses, lenses having aberration corrections, diffractive lenses, lenses for variably absorbing electromagnetic radiation, lenses having variable focus having adjustable focus; power activated variable focus means, e.g. mechanically or electrically by the ciliary muscle or from the outside for changing radial position, i.e. perpendicularly to the visual axis when implanted
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/14—Eye parts, e.g. lenses or corneal implants; Artificial eyes
- A61F2/16—Intraocular lenses
- A61F2002/1681—Intraocular lenses having supporting structure for lens, e.g. haptics
- A61F2002/1683—Intraocular lenses having supporting structure for lens, e.g. haptics having filiform haptics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/14—Eye parts, e.g. lenses or corneal implants; Artificial eyes
- A61F2/16—Intraocular lenses
- A61F2002/1681—Intraocular lenses having supporting structure for lens, e.g. haptics
- A61F2002/1683—Intraocular lenses having supporting structure for lens, e.g. haptics having filiform haptics
- A61F2002/1686—Securing a filiform haptic to a lens body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/14—Eye parts, e.g. lenses or corneal implants; Artificial eyes
- A61F2/16—Intraocular lenses
- A61F2002/1681—Intraocular lenses having supporting structure for lens, e.g. haptics
- A61F2002/1689—Intraocular lenses having supporting structure for lens, e.g. haptics having plate-haptics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/14—Eye parts, e.g. lenses or corneal implants; Artificial eyes
- A61F2/16—Intraocular lenses
- A61F2002/1681—Intraocular lenses having supporting structure for lens, e.g. haptics
- A61F2002/169—Surrounding optic
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/14—Eye parts, e.g. lenses or corneal implants; Artificial eyes
- A61F2/16—Intraocular lenses
- A61F2002/1681—Intraocular lenses having supporting structure for lens, e.g. haptics
- A61F2002/16901—Supporting structure conforms to shape of capsular bag
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting in contact-lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/008—Methods or devices for eye surgery using laser
- A61F2009/00861—Methods or devices for eye surgery using laser adapted for treatment at a particular location
- A61F2009/0087—Lens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0004—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof adjustable
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/16—Materials or treatment for tissue regeneration for reconstruction of eye parts, e.g. intraocular lens, cornea
Landscapes
- Health & Medical Sciences (AREA)
- Ophthalmology & Optometry (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Transplantation (AREA)
- Chemical & Material Sciences (AREA)
- Vascular Medicine (AREA)
- Heart & Thoracic Surgery (AREA)
- Biomedical Technology (AREA)
- Engineering & Computer Science (AREA)
- Medicinal Chemistry (AREA)
- Cardiology (AREA)
- Dermatology (AREA)
- Epidemiology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Surgery (AREA)
- Optics & Photonics (AREA)
- Physics & Mathematics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electromagnetism (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Prostheses (AREA)
- Materials For Medical Uses (AREA)
Description
本願は、2019年4月9日に出願された、Xuwei Jiang、Ali Akinay、Jingbo Liu、及びJian Liuを発明者とする米国仮特許出願第62/831,520号の“IN-SITU ADJUSTABLE INTRAOCULAR LENS”の優先権の利益を主張するものであり、その全体を本明細書中に全文が完全に記載されているかのように本願に援用する。
i)少なくとも1つの支持部は、その少なくとも一部がPD-LCNから形成される支持部接合部を含んでいてよく、
ii)少なくとも1つの支持部は光学部に支持部接合部を介して取り付けられてよく、
iv)IOLは、光学部を保持するベースをさらに含み、少なくとも1つの支持部はベースに取り付けられてよく、
v)IOLは複数の支持部を含んでいてよく、各々の少なくとも一部はPD-LCNから形成されてよく、
vi)各支持部は、その少なくとも一部がPD-LCNから形成されてよい支持部接合部を含んでいてよく、
vii)PD-LCNは架橋されたジアクリレート液晶モノマとジアクリレートアゾベンゼン液晶モノマとを含んでいてよく、
viii)PD-LCNは25wt%以下のジアクリレートアゾベンゼン液晶モノマを含んでいてよく、
ix)PD-LCNの架橋密度は1.0モル/dm3~8.0モル/dm3であってよく、
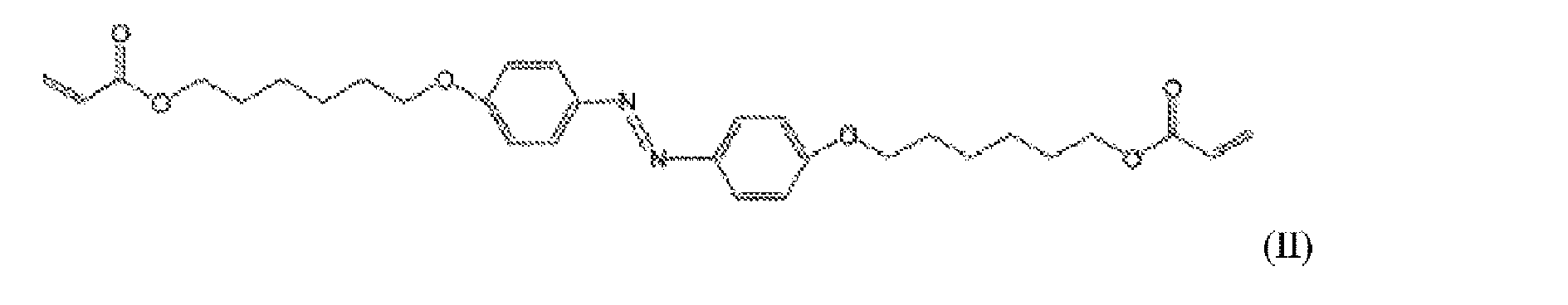
x)ジアクリレート液晶モノマは4-(3-アクリロイルオキシプロピルオキシ)-benzoesure 2-メチル-1,4-フェニルエステルを含んでいてよく、
xi)ジアクリレートアゾベンゼン液晶モノマは4,4’-ビス[6-アクリロルオキシ(acryloloxy))ヘキシルオキシ]アゾベンゼンを含んでいてよく、
xii)ジアクリレート液晶モノマは4-(3-アクリロイルオキシルオキシプロピルオキシ)-benzoesure 2-メチル-1,4-フェニルエステルを含んでいてよく、ジアクリレートアゾベンゼン液晶モノマは4,4’-ビス[6-アクリロルオキシ(acryloloxy))ヘキシルオキシ]アゾベンゼンを含んでいてよい。
i)光誘起形状記憶ポリマ網目構造は、ポリドメインアゾ液晶ポリマ網目構造(PD-LCN)を含んでいてよく、
ii)少なくとも1つの支持部は、その少なくとも一部が光誘起形状記憶ポリマ網目構造から形成されてよい支持部接合部を含んでいてよく、
iii)少なくとも1つの支持部は光学部に支持部接合部を介して取り付けられてよく、
iv)少なくとも1つの支持部は複数の支持部を含んでいてよく、各々の少なくとも一部は光誘起形状記憶ポリマ網目構造から形成されてよく、
v)PD-LCDは架橋されたジアクリレート液晶モノマとジアクリレートアゾベンゼン液晶モノマを含んでいてよく、
vi)PD-LCNは25wt%以下のジアクリレートアゾベンゼン液晶モノマを含んでいてよく、
vii)PD-LCNの架橋密度は1.0モル/dm3~8.0モル/dm3であってよく、
viii)ジアクリレート液晶モノマは4-(3-アクリロイルオキシプロピルオキシ)-benzoesure 2-メチル-1,4-フェニルエステルを含んでいてよく、
ix)ジアクリレートアゾベンゼン液晶モノマは4,4’-ビス[6-アクリロルオキシ(acryloloxy))ヘキシルオキシ]アゾベンゼンを含んでいてよく、
x)ジアクリレート液晶モノマは4-(3-アクリルオキシルオキシプロピルオキシ)-benzoesure 2-メチル-1,4-フェニルエステルを含んでいてよく、ジアクリレートアゾベンゼン液晶モノマは4,4’-ビス[6-アクリロルオキシ(acryloloxy))ヘキシルオキシ]アゾベンゼンを含んでいてよい。
i)偏光レーザ放射は、両端の値を含む440nm~514nmの範囲の波長を有していてよく、
ii)IOLの位置は軸方向に前後に調整されてよく、
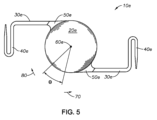
iii)IOLの位置は半径方向にある角度だけ調整されてよく、
iv)IOLは、実際の位置が水晶体嚢内の標的位置とは異なる位置であってもよく、IOlの位置を調整することは、IOLを標的位置まで移動させることを含んでいてよく、
v)照射は、両端の値を含む0.5秒~5分にわたり行われてよい。
i)光誘起形状記憶ポリマ網目構造はPD-LCNを含み、
ii)偏光レーザ放射は、両端の値を含む440nm~514nmの範囲の波長を有していてよく、
iii)IOLの位置は軸方向に前後に調整されてよく、
iv)IOLの位置は半径方向に角度θだけ調整されてよく、
v)照射は、両端の値を含む0.5秒~5分にわたり行われてよく、
vi)PD-LCNを含む支持部の照射される部分は支持部接合部を含んでいてよい。
i)支持部の放射が照射される部分は支持部接合部を含み、
ii)レーザはフェムト秒レーザ又はエキシマレーザを含んでいてよい。
また、本開示は以下の発明を含む。
第1の態様は、
光学部と、
少なくとも一部がポリドメインアゾ液晶ポリマ網目構造(PD-LCN)を含む少なくとも1つの支持部と、
を含む、眼内レンズ(IOL)である。
第2の態様は、
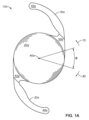
前記少なくとも1つの支持部は支持部接合部を含み、前記支持部接合部の少なくとも一部はPD-LCNから形成される、第1の態様におけるIOLである。
第3の態様は、
前記少なくとも1つの支持部は前記光学部に前記支持部接合部を介して取り付けられる、第1の態様又は第2の態様におけるIOLである。
第4の態様は、
前記光学部を保持するように構成されたベースをさらに含み、前記少なくとも1つの支持部は前記ベースに取り付けられる、第1の態様又は第2の態様におけるIOLである。
第5の態様は、
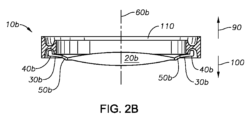
複数の支持部を含み、前記複数の支持部の各々の少なくとも一部はPD-LCNを含む、第1の態様~第4の態様の何れか一つにおけるIOLである。
第6の態様は、
前記PD-LCNは架橋されたジアクリレート液晶モノマとジアクリレートアゾベンゼン液晶モノマとを含む、第1の態様~第5の態様の何れか一つにおけるIOLである。
第7の態様は、
前記PD-LCNは25wt%以下のジアクリレートアゾベンゼン液晶モノマを含む、第6の態様におけるIOLである。
第8の態様は、
前記PD-LCNの架橋密度は1.0モル/dm 3 ~8.0モル/dm 3 である、第6の態様におけるIOLである。
第9の態様は、
前記ジアクリレートアゾベンゼン液晶モノマは4-(3-アクリロイルオキシプロピルオキシ)-benzoesure 2-メチル-1,4-フェニルエステルを含む、第6の態様におけるIOLである。
第10の態様は、
前記ジアクリレートアゾベンゼン液晶モノマは4,4’-ビス[6-アクリロルオキシ(acryloloxy))ヘキシルオキシ]アゾベンゼンを含む、第6の態様におけるIOLである。
第11の態様は、
眼内レンズ(IOL)を調整する方法において、前記IOLの支持部であって、ポリドメインアゾ液晶ポリマ網目構造(PD-LCN)を含む支持部の一部に偏光レーザ放射を照射して、前記PD-LCNをある曲げ角度まで曲げ、それによって前記IOLが入っている水晶体嚢に押し付け、前記水晶体嚢内の前記IOLの位置を調整するステップを含む方法である。
第12の態様は、
前記偏光レーザ放射の波長は、両端の数値を含む440nm~514nmの範囲である、第11の態様における方法である。
第13の態様は、
前記IOLの位置は軸方向に前後に調整される、第11の態様又は第12の態様における方法である。
第14の態様は、
前記IOLの位置は半径方向に角度θだけ調整される、第11の態様~第13の態様の何れか一つにおける方法である。
第15の態様は、
前記PD-LCNを含む前記支持部の前記照射される部分は支持部接合部を含む、第11の態様~第14の態様の何れか一つにおける方法である。
Claims (9)
- 光学部と、
少なくとも一部がポリドメインアゾ液晶ポリマ網目構造(PD-LCN)を含む複数の支持部と、
を含み、
前記支持部の前記PD-LCNの部分は、前記光学部の位置を可逆的に調整するように構成されており、
前記支持部は支持部接合部を含み、前記複数の支持部の内の2つの支持部の支持部接合部は、異なるwt%のジアクリレートアゾベンゼン液晶モノマ又は異なる架橋密度を有する、眼内レンズ(IOL)。 - 前記支持部接合部の少なくとも一部はPD-LCNから形成される、請求項1に記載のIOL。
- 前記支持部は前記光学部に前記支持部接合部を介して取り付けられる、請求項2に記載のIOL。
- 前記光学部を保持するように構成されたベースをさらに含み、前記支持部は前記ベースに取り付けられる、請求項1又は2に記載のIOL。
- 前記PD-LCNは架橋されたジアクリレート液晶モノマとジアクリレートアゾベンゼン液晶モノマとを含む、請求項1~4の何れか一項に記載のIOL。
- 前記PD-LCNは25wt%以下のジアクリレートアゾベンゼン液晶モノマを含む、請求項5に記載のIOL。
- 前記PD-LCNの架橋密度は1.0モル/dm3~8.0モル/dm3である、請求項5に記載のIOL。
- 前記ジアクリレートアゾベンゼン液晶モノマは4-(3-アクリロイルオキシプロピルオキシ)-benzoesure 2-メチル-1,4-フェニルエステルを含む、請求項5に記載のIOL。
- 前記ジアクリレートアゾベンゼン液晶モノマは4,4’-ビス[6-アクリロルオキシ(acryloloxy))ヘキシルオキシ]アゾベンゼンを含む、請求項5に記載のIOL。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2024194971A JP2025020330A (ja) | 2019-04-09 | 2024-11-07 | その場調整可能な眼内レンズ |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201962831520P | 2019-04-09 | 2019-04-09 | |
| US62/831,520 | 2019-04-09 | ||
| PCT/IB2020/053279 WO2020208501A1 (en) | 2019-04-09 | 2020-04-06 | In-situ adjustable intraocular lens |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2024194971A Division JP2025020330A (ja) | 2019-04-09 | 2024-11-07 | その場調整可能な眼内レンズ |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2022527831A JP2022527831A (ja) | 2022-06-06 |
| JP7586832B2 true JP7586832B2 (ja) | 2024-11-19 |
Family
ID=70289428
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2021559326A Active JP7586832B2 (ja) | 2019-04-09 | 2020-04-06 | その場調整可能な眼内レンズ |
| JP2024194971A Pending JP2025020330A (ja) | 2019-04-09 | 2024-11-07 | その場調整可能な眼内レンズ |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2024194971A Pending JP2025020330A (ja) | 2019-04-09 | 2024-11-07 | その場調整可能な眼内レンズ |
Country Status (7)
| Country | Link |
|---|---|
| US (3) | US11701220B2 (ja) |
| EP (1) | EP3927386A1 (ja) |
| JP (2) | JP7586832B2 (ja) |
| CN (2) | CN116327427A (ja) |
| AU (1) | AU2020270548B2 (ja) |
| CA (1) | CA3131226A1 (ja) |
| WO (1) | WO2020208501A1 (ja) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA3096886A1 (en) * | 2018-05-30 | 2019-12-05 | Alcon Inc. | System and method for nomogram-based refractive laser surgery |
| WO2020086312A1 (en) | 2018-10-23 | 2020-04-30 | The Board Of Trustees Of The Leland Stanford Junior University | Intraocular artificial lens capsule |
| US12419736B2 (en) | 2020-04-29 | 2025-09-23 | Long Bridge Medical, Inc. | Devices to support and position an intraocular lens within the eye and methods of use |
| US11759309B2 (en) | 2020-04-29 | 2023-09-19 | Long Bridge Medical, Inc. | Devices to support and position an intraocular lens within the eye and methods of use |
| CA3227200A1 (en) | 2021-07-28 | 2023-02-02 | SpyGlass Pharma, Inc. | Stabilized intraocular drug delivery systems and methods of use |
| DE102022122962A1 (de) * | 2022-09-09 | 2024-03-14 | Carl Zeiss Meditec Ag | Intraokularlinse mit einer zwei arme aufweisenden ersten haptik und einer zweiten haptik |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006517447A (ja) | 2003-02-06 | 2006-07-27 | パワービジョン, インコーポレイテッド | 適合性光学レンズシステムおよび使用方法 |
| JP2007262195A (ja) | 2006-03-28 | 2007-10-11 | Fujifilm Corp | 光駆動型高分子アクチュエータ、光駆動型高分子アクチュエータの製造方法、多官能性モノマー、および高分子 |
| JP2008019744A (ja) | 2006-07-11 | 2008-01-31 | Fujifilm Corp | 光駆動型アクチュエータ、受光素子、光ゲート素子、光反射素子、および光駆動型アクチュエータの使用方法 |
| US20100082017A1 (en) | 2008-09-26 | 2010-04-01 | Advanced Medical Optics, Inc. | Laser modification of intraocular lens |
| WO2010125596A1 (en) | 2009-04-30 | 2010-11-04 | Promacon Italia Oftalmologia S.R.L. | Intraocular lens with system of laser adjustable haptics |
| JP2013245296A (ja) | 2012-05-25 | 2013-12-09 | Daiken Iki Kk | 架橋型液晶高分子材料の変形方法、光駆動型成形体 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5728155A (en) * | 1996-01-22 | 1998-03-17 | Quantum Solutions, Inc. | Adjustable intraocular lens |
| WO2004081613A2 (en) * | 2003-03-06 | 2004-09-23 | Shadduck John H | Adaptive optic lens and method of making |
| US20070010881A1 (en) * | 2005-07-11 | 2007-01-11 | Alcon, Inc. | Intraocular lens system |
| DE102005045540A1 (de) | 2005-09-23 | 2007-03-29 | Hampp, Norbert, Prof. Dr. | Intraokularlinse |
| CN101827565B (zh) | 2007-10-15 | 2014-04-02 | 爱克透镜国际公司 | 可调节的调节型人工晶体及其定位装置 |
| US20140094908A1 (en) * | 2012-09-19 | 2014-04-03 | Roger Zaldivar | Intraocular lens and method |
| US10159566B2 (en) * | 2016-09-26 | 2018-12-25 | Novartis Ag | Heat tunable intraocular lens |
| TW201912675A (zh) * | 2017-08-18 | 2019-04-01 | 鴻海精密工業股份有限公司 | 偶氮苯凝膠、製備方法及自動變焦人工晶狀體 |
-
2020
- 2020-04-06 AU AU2020270548A patent/AU2020270548B2/en active Active
- 2020-04-06 JP JP2021559326A patent/JP7586832B2/ja active Active
- 2020-04-06 CA CA3131226A patent/CA3131226A1/en active Pending
- 2020-04-06 CN CN202310559961.5A patent/CN116327427A/zh active Pending
- 2020-04-06 CN CN202080025981.9A patent/CN113677378B/zh active Active
- 2020-04-06 EP EP20719231.1A patent/EP3927386A1/en active Pending
- 2020-04-06 US US16/840,660 patent/US11701220B2/en active Active
- 2020-04-06 WO PCT/IB2020/053279 patent/WO2020208501A1/en not_active Ceased
-
2023
- 2023-05-30 US US18/325,280 patent/US12193929B2/en active Active
-
2024
- 2024-11-07 JP JP2024194971A patent/JP2025020330A/ja active Pending
- 2024-12-02 US US18/965,096 patent/US20250090304A1/en active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006517447A (ja) | 2003-02-06 | 2006-07-27 | パワービジョン, インコーポレイテッド | 適合性光学レンズシステムおよび使用方法 |
| JP2007262195A (ja) | 2006-03-28 | 2007-10-11 | Fujifilm Corp | 光駆動型高分子アクチュエータ、光駆動型高分子アクチュエータの製造方法、多官能性モノマー、および高分子 |
| JP2008019744A (ja) | 2006-07-11 | 2008-01-31 | Fujifilm Corp | 光駆動型アクチュエータ、受光素子、光ゲート素子、光反射素子、および光駆動型アクチュエータの使用方法 |
| US20100082017A1 (en) | 2008-09-26 | 2010-04-01 | Advanced Medical Optics, Inc. | Laser modification of intraocular lens |
| WO2010125596A1 (en) | 2009-04-30 | 2010-11-04 | Promacon Italia Oftalmologia S.R.L. | Intraocular lens with system of laser adjustable haptics |
| JP2013245296A (ja) | 2012-05-25 | 2013-12-09 | Daiken Iki Kk | 架橋型液晶高分子材料の変形方法、光駆動型成形体 |
Non-Patent Citations (1)
| Title |
|---|
| 池田 富樹ら,光駆動高分子液晶フィルム-光で自由に曲がるプラスチック-,応用物理,2004年07月10日,73, 7,pp.947-951,https://doi.org/10.11470/oubutsu.73.7_947 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113677378A (zh) | 2021-11-19 |
| WO2020208501A1 (en) | 2020-10-15 |
| US20250090304A1 (en) | 2025-03-20 |
| US20200323626A1 (en) | 2020-10-15 |
| US11701220B2 (en) | 2023-07-18 |
| AU2020270548A1 (en) | 2021-09-09 |
| EP3927386A1 (en) | 2021-12-29 |
| US12193929B2 (en) | 2025-01-14 |
| US20230320841A1 (en) | 2023-10-12 |
| CN113677378B (zh) | 2023-05-16 |
| JP2022527831A (ja) | 2022-06-06 |
| JP2025020330A (ja) | 2025-02-12 |
| CA3131226A1 (en) | 2020-10-15 |
| CN116327427A (zh) | 2023-06-27 |
| AU2020270548B2 (en) | 2026-02-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7586832B2 (ja) | その場調整可能な眼内レンズ | |
| Charman | Developments in the correction of presbyopia II: surgical approaches | |
| US11311372B2 (en) | Intraocular lens implant for the correction of vision | |
| EP1296616B1 (en) | Method of correcting optical aberrations | |
| US10213102B2 (en) | Presbyopia correction through negative spherical aberration | |
| US5288293A (en) | In vivo modification of refractive power of an intraocular lens implant | |
| KR20170058951A (ko) | 안구내 인공수정체 콘택트 렌즈와 관련 시스템 및 방법 | |
| CA2829374C (en) | Capsular membrane implants to increase accommodative amplitude | |
| US5984961A (en) | Intracorneal prism and method of improving vision in a human eye having macula disease | |
| Schwiegerling | LASIK and beyond | |
| Ciralsky et al. | Presbyopia and Future Intraocular Lenses | |
| Wallace et al. | Toric intraocular lenses: advancements in astigmatism correction | |
| EP3536281B1 (en) | Intraocular lens | |
| Jacob et al. | Angle kappa may play important role in success of multifocal IOLs. | |
| Tomalla | Accommodating IOL achieves good 3-month European results. | |
| Dick et al. | the Correction of Astigmatism | |
| ARTAL | HOW IS EXTENDED DEPTH OF FOCUS ACHIEVED WITH A SMALL-APERTURE LENS? | |
| Megur | Intra-‐Operative Strategies to prevent post op surprises. Tips & Pearls |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20230327 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20240130 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20240426 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20240611 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20240904 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20241008 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20241107 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7586832 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |