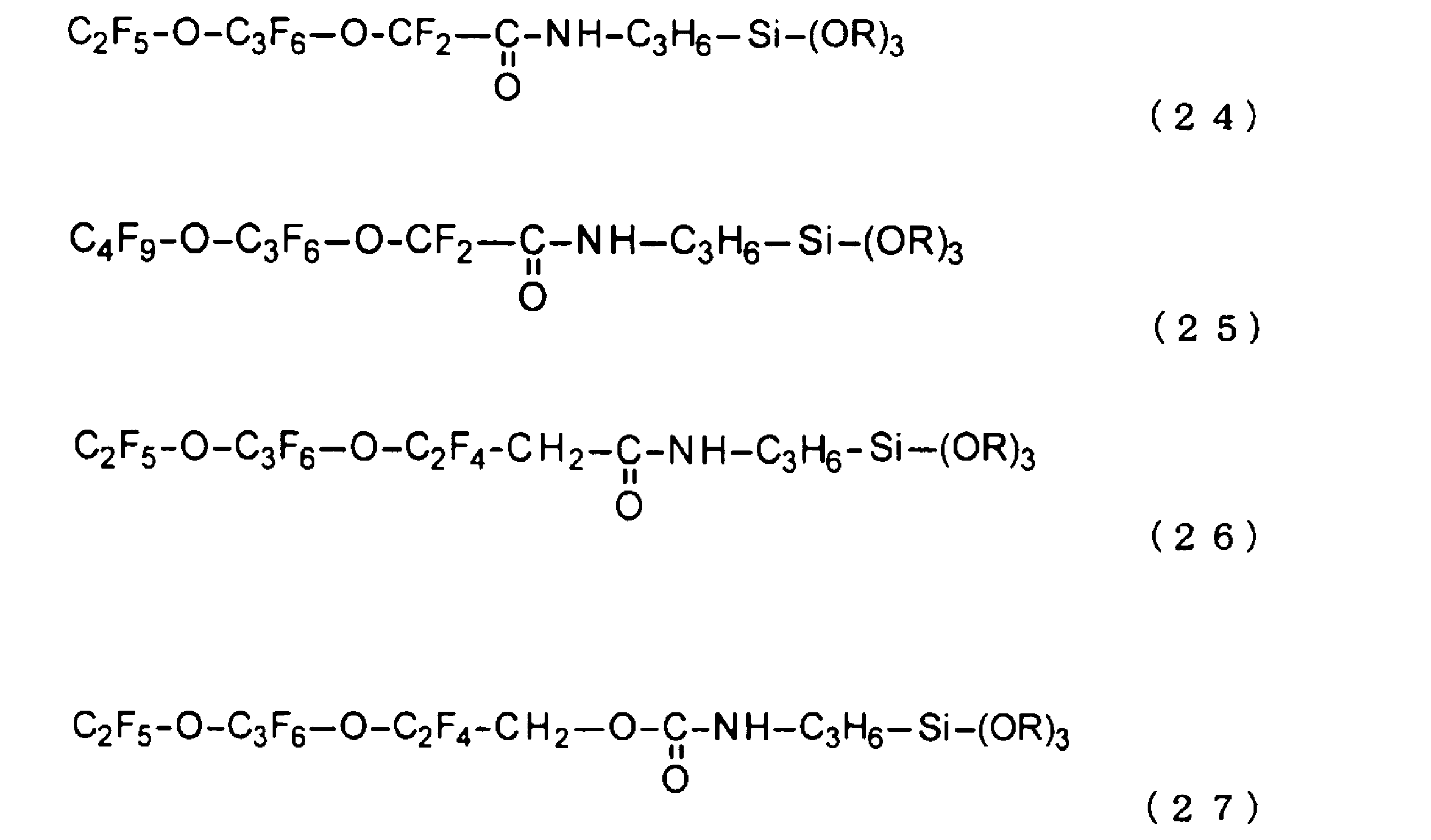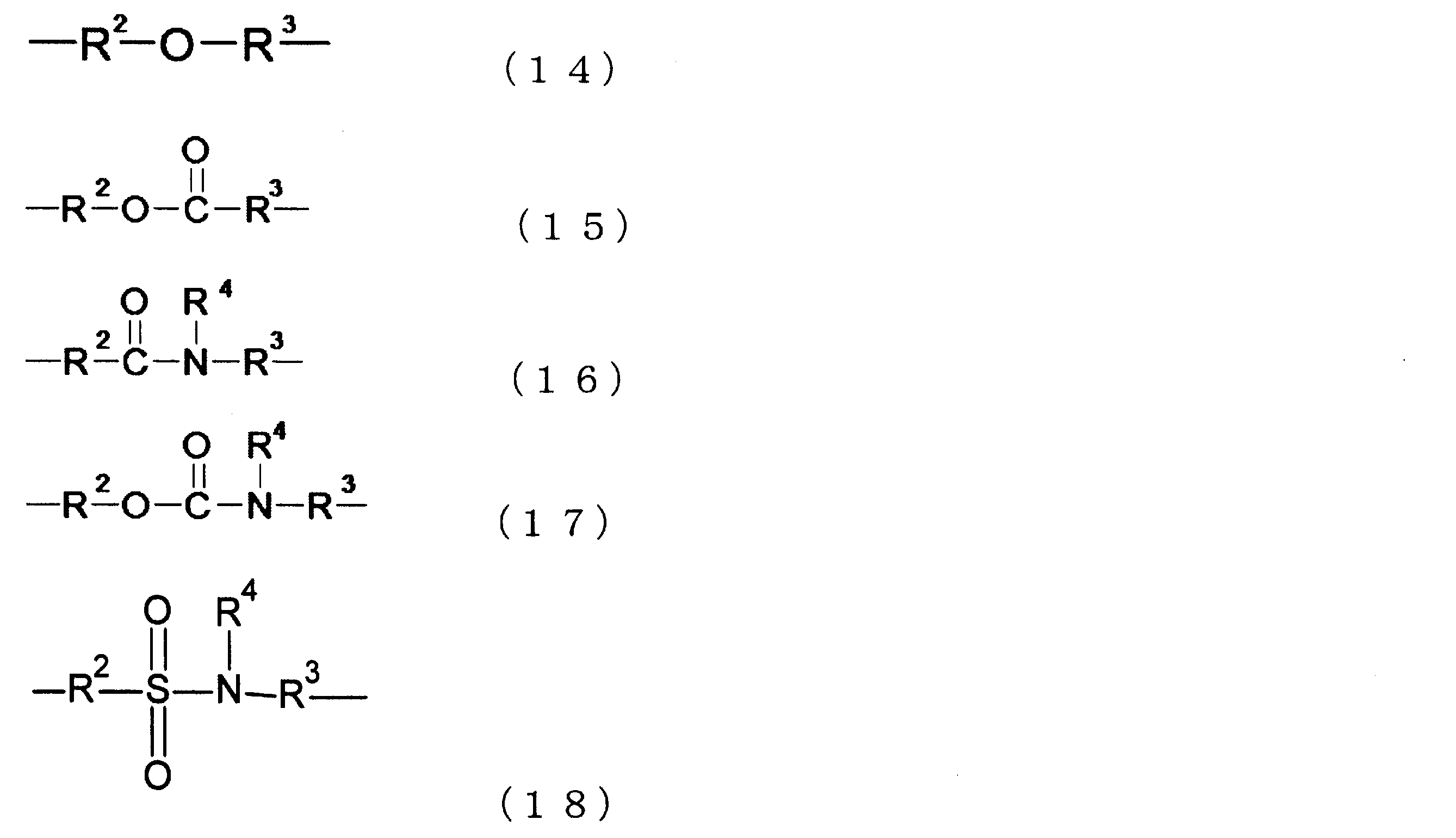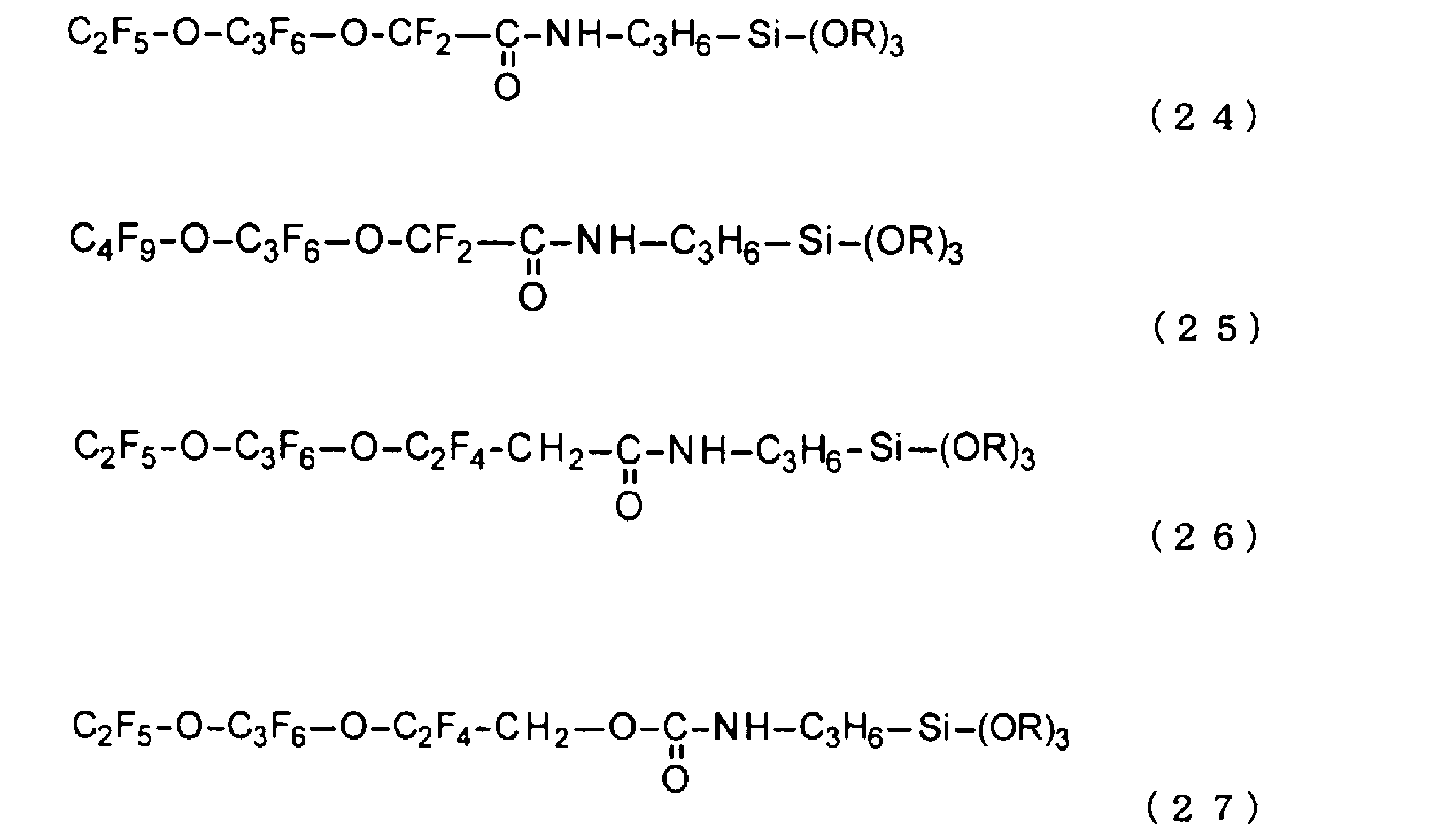JP7577472B2 - Water- and oil-repellent film-forming liquid composition and method for producing same - Google Patents
Water- and oil-repellent film-forming liquid composition and method for producing same Download PDFInfo
- Publication number
- JP7577472B2 JP7577472B2 JP2020120307A JP2020120307A JP7577472B2 JP 7577472 B2 JP7577472 B2 JP 7577472B2 JP 2020120307 A JP2020120307 A JP 2020120307A JP 2020120307 A JP2020120307 A JP 2020120307A JP 7577472 B2 JP7577472 B2 JP 7577472B2
- Authority
- JP
- Japan
- Prior art keywords
- fluorine
- water
- oil
- oxide particles
- metal oxide
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000007788 liquid Substances 0.000 title claims description 118
- 239000000203 mixture Substances 0.000 title claims description 86
- 239000005871 repellent Substances 0.000 title claims description 66
- 238000004519 manufacturing process Methods 0.000 title claims description 22
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 185
- 239000011737 fluorine Substances 0.000 claims description 149
- 229910052731 fluorine Inorganic materials 0.000 claims description 149
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 claims description 147
- 239000002245 particle Substances 0.000 claims description 131
- 239000000377 silicon dioxide Substances 0.000 claims description 91
- 229910044991 metal oxide Inorganic materials 0.000 claims description 84
- 150000004706 metal oxides Chemical class 0.000 claims description 84
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 66
- 239000006185 dispersion Substances 0.000 claims description 61
- 150000001875 compounds Chemical class 0.000 claims description 46
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 45
- 150000002222 fluorine compounds Chemical class 0.000 claims description 32
- 239000003921 oil Substances 0.000 claims description 25
- 229910052710 silicon Inorganic materials 0.000 claims description 23
- -1 silicon alkoxide Chemical class 0.000 claims description 23
- 239000002904 solvent Substances 0.000 claims description 20
- 125000002947 alkylene group Chemical group 0.000 claims description 19
- 239000003054 catalyst Substances 0.000 claims description 19
- 239000010703 silicon Substances 0.000 claims description 17
- 125000004432 carbon atom Chemical group C* 0.000 claims description 15
- 238000002156 mixing Methods 0.000 claims description 13
- UJMWVICAENGCRF-UHFFFAOYSA-N oxygen difluoride Chemical group FOF UJMWVICAENGCRF-UHFFFAOYSA-N 0.000 claims description 10
- 239000003960 organic solvent Substances 0.000 claims description 9
- 229910052782 aluminium Inorganic materials 0.000 claims description 7
- 229910052791 calcium Inorganic materials 0.000 claims description 6
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 6
- 229910052749 magnesium Inorganic materials 0.000 claims description 6
- 239000012046 mixed solvent Substances 0.000 claims description 6
- 229910052719 titanium Inorganic materials 0.000 claims description 6
- 229910052725 zinc Inorganic materials 0.000 claims description 6
- 229910052726 zirconium Inorganic materials 0.000 claims description 6
- 229910052751 metal Inorganic materials 0.000 claims description 5
- 239000002184 metal Substances 0.000 claims description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 5
- 150000002739 metals Chemical class 0.000 claims description 4
- 230000002940 repellent Effects 0.000 claims description 2
- 230000015572 biosynthetic process Effects 0.000 description 55
- 238000003786 synthesis reaction Methods 0.000 description 55
- 239000000499 gel Substances 0.000 description 51
- DCAYPVUWAIABOU-UHFFFAOYSA-N hexadecane Chemical compound CCCCCCCCCCCCCCCC DCAYPVUWAIABOU-UHFFFAOYSA-N 0.000 description 40
- 239000000758 substrate Substances 0.000 description 40
- 230000000052 comparative effect Effects 0.000 description 29
- 125000000524 functional group Chemical group 0.000 description 28
- 238000012360 testing method Methods 0.000 description 23
- 235000012239 silicon dioxide Nutrition 0.000 description 22
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 18
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 18
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 18
- 229910017604 nitric acid Inorganic materials 0.000 description 18
- 238000000034 method Methods 0.000 description 16
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 12
- LFQCEHFDDXELDD-UHFFFAOYSA-N tetramethyl orthosilicate Chemical compound CO[Si](OC)(OC)OC LFQCEHFDDXELDD-UHFFFAOYSA-N 0.000 description 11
- 239000011246 composite particle Substances 0.000 description 10
- 239000011248 coating agent Substances 0.000 description 9
- 238000000576 coating method Methods 0.000 description 9
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 8
- BOTDANWDWHJENH-UHFFFAOYSA-N Tetraethyl orthosilicate Chemical compound CCO[Si](OCC)(OCC)OCC BOTDANWDWHJENH-UHFFFAOYSA-N 0.000 description 8
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 8
- 229910052681 coesite Inorganic materials 0.000 description 8
- 229910052906 cristobalite Inorganic materials 0.000 description 8
- 125000003700 epoxy group Chemical group 0.000 description 8
- 238000006460 hydrolysis reaction Methods 0.000 description 8
- 229910052682 stishovite Inorganic materials 0.000 description 8
- 229910052905 tridymite Inorganic materials 0.000 description 8
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 7
- 229910000077 silane Inorganic materials 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- 235000010215 titanium dioxide Nutrition 0.000 description 7
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 239000011230 binding agent Substances 0.000 description 6
- 150000002484 inorganic compounds Chemical class 0.000 description 6
- 229910010272 inorganic material Inorganic materials 0.000 description 6
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 6
- 239000002114 nanocomposite Substances 0.000 description 6
- 238000006116 polymerization reaction Methods 0.000 description 6
- 239000004408 titanium dioxide Substances 0.000 description 6
- 150000001298 alcohols Chemical class 0.000 description 5
- 230000007062 hydrolysis Effects 0.000 description 5
- 150000007522 mineralic acids Chemical class 0.000 description 5
- 150000007524 organic acids Chemical class 0.000 description 5
- 239000002002 slurry Substances 0.000 description 5
- 239000000243 solution Substances 0.000 description 5
- 150000003609 titanium compounds Chemical class 0.000 description 5
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 4
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- MCMNRKCIXSYSNV-UHFFFAOYSA-N ZrO2 Inorganic materials O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 4
- 239000011575 calcium Substances 0.000 description 4
- 229910052799 carbon Inorganic materials 0.000 description 4
- 125000001153 fluoro group Chemical group F* 0.000 description 4
- 150000002430 hydrocarbons Chemical group 0.000 description 4
- 239000011777 magnesium Substances 0.000 description 4
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 4
- 239000011859 microparticle Substances 0.000 description 4
- 239000004745 nonwoven fabric Substances 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- 239000010936 titanium Substances 0.000 description 4
- 239000013638 trimer Substances 0.000 description 4
- 239000011701 zinc Substances 0.000 description 4
- 239000011787 zinc oxide Substances 0.000 description 4
- 238000005299 abrasion Methods 0.000 description 3
- 238000009835 boiling Methods 0.000 description 3
- 239000002131 composite material Substances 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 239000010419 fine particle Substances 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 230000009257 reactivity Effects 0.000 description 3
- VXUYXOFXAQZZMF-UHFFFAOYSA-N titanium(IV) isopropoxide Chemical compound CC(C)O[Ti](OC(C)C)(OC(C)C)OC(C)C VXUYXOFXAQZZMF-UHFFFAOYSA-N 0.000 description 3
- BPSIOYPQMFLKFR-UHFFFAOYSA-N trimethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](OC)(OC)CCCOCC1CO1 BPSIOYPQMFLKFR-UHFFFAOYSA-N 0.000 description 3
- AIFLGMNWQFPTAJ-UHFFFAOYSA-J 2-hydroxypropanoate;titanium(4+) Chemical compound [Ti+4].CC(O)C([O-])=O.CC(O)C([O-])=O.CC(O)C([O-])=O.CC(O)C([O-])=O AIFLGMNWQFPTAJ-UHFFFAOYSA-J 0.000 description 2
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- ODINCKMPIJJUCX-UHFFFAOYSA-N Calcium oxide Chemical compound [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 description 2
- 229920000298 Cellophane Polymers 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 239000012295 chemical reaction liquid Substances 0.000 description 2
- SWXVUIWOUIDPGS-UHFFFAOYSA-N diacetone alcohol Chemical compound CC(=O)CC(C)(C)O SWXVUIWOUIDPGS-UHFFFAOYSA-N 0.000 description 2
- 235000019253 formic acid Nutrition 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 239000012535 impurity Substances 0.000 description 2
- 235000005985 organic acids Nutrition 0.000 description 2
- 235000006408 oxalic acid Nutrition 0.000 description 2
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 description 2
- 125000005010 perfluoroalkyl group Chemical group 0.000 description 2
- 229920000139 polyethylene terephthalate Polymers 0.000 description 2
- 239000005020 polyethylene terephthalate Substances 0.000 description 2
- HKJYVRJHDIPMQB-UHFFFAOYSA-N propan-1-olate;titanium(4+) Chemical compound CCCO[Ti](OCCC)(OCCC)OCCC HKJYVRJHDIPMQB-UHFFFAOYSA-N 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- SZUVGFMDDVSKSI-WIFOCOSTSA-N (1s,2s,3s,5r)-1-(carboxymethyl)-3,5-bis[(4-phenoxyphenyl)methyl-propylcarbamoyl]cyclopentane-1,2-dicarboxylic acid Chemical compound O=C([C@@H]1[C@@H]([C@](CC(O)=O)([C@H](C(=O)N(CCC)CC=2C=CC(OC=3C=CC=CC=3)=CC=2)C1)C(O)=O)C(O)=O)N(CCC)CC(C=C1)=CC=C1OC1=CC=CC=C1 SZUVGFMDDVSKSI-WIFOCOSTSA-N 0.000 description 1
- HCGFUIQPSOCUHI-UHFFFAOYSA-N 2-propan-2-yloxyethanol Chemical compound CC(C)OCCO HCGFUIQPSOCUHI-UHFFFAOYSA-N 0.000 description 1
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 1
- KCBAMQOKOLXLOX-BSZYMOERSA-N CC1=C(SC=N1)C2=CC=C(C=C2)[C@H](C)NC(=O)[C@@H]3C[C@H](CN3C(=O)[C@H](C(C)(C)C)NC(=O)CCCCCCCCCCNCCCONC(=O)C4=C(C(=C(C=C4)F)F)NC5=C(C=C(C=C5)I)F)O Chemical compound CC1=C(SC=N1)C2=CC=C(C=C2)[C@H](C)NC(=O)[C@@H]3C[C@H](CN3C(=O)[C@H](C(C)(C)C)NC(=O)CCCCCCCCCCNCCCONC(=O)C4=C(C(=C(C=C4)F)F)NC5=C(C=C(C=C5)I)F)O KCBAMQOKOLXLOX-BSZYMOERSA-N 0.000 description 1
- 229920003043 Cellulose fiber Polymers 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 229910020068 MgAl Inorganic materials 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- 229910004298 SiO 2 Inorganic materials 0.000 description 1
- 239000006087 Silane Coupling Agent Substances 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 229910002808 Si–O–Si Inorganic materials 0.000 description 1
- 229910010413 TiO 2 Inorganic materials 0.000 description 1
- GEIAQOFPUVMAGM-UHFFFAOYSA-N ZrO Inorganic materials [Zr]=O GEIAQOFPUVMAGM-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 125000004423 acyloxy group Chemical group 0.000 description 1
- 239000002390 adhesive tape Substances 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 230000003373 anti-fouling effect Effects 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 125000002102 aryl alkyloxo group Chemical group 0.000 description 1
- 125000004104 aryloxy group Chemical group 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 125000001231 benzoyloxy group Chemical group C(C1=CC=CC=C1)(=O)O* 0.000 description 1
- 125000000051 benzyloxy group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])O* 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- FPCJKVGGYOAWIZ-UHFFFAOYSA-N butan-1-ol;titanium Chemical compound [Ti].CCCCO.CCCCO.CCCCO.CCCCO FPCJKVGGYOAWIZ-UHFFFAOYSA-N 0.000 description 1
- YHWCPXVTRSHPNY-UHFFFAOYSA-N butan-1-olate;titanium(4+) Chemical compound [Ti+4].CCCC[O-].CCCC[O-].CCCC[O-].CCCC[O-] YHWCPXVTRSHPNY-UHFFFAOYSA-N 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 229940126543 compound 14 Drugs 0.000 description 1
- 229940125833 compound 23 Drugs 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 238000007607 die coating method Methods 0.000 description 1
- OTARVPUIYXHRRB-UHFFFAOYSA-N diethoxy-methyl-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CCO[Si](C)(OCC)CCCOCC1CO1 OTARVPUIYXHRRB-UHFFFAOYSA-N 0.000 description 1
- WHGNXNCOTZPEEK-UHFFFAOYSA-N dimethoxy-methyl-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](C)(OC)CCCOCC1CO1 WHGNXNCOTZPEEK-UHFFFAOYSA-N 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 239000005357 flat glass Substances 0.000 description 1
- 150000002221 fluorine Chemical class 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 238000010191 image analysis Methods 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 239000010721 machine oil Substances 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 125000006551 perfluoro alkylene group Chemical group 0.000 description 1
- 239000010702 perfluoropolyether Substances 0.000 description 1
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920001084 poly(chloroprene) Polymers 0.000 description 1
- 229920003207 poly(ethylene-2,6-naphthalate) Polymers 0.000 description 1
- 229920001707 polybutylene terephthalate Polymers 0.000 description 1
- 229920006267 polyester film Polymers 0.000 description 1
- 239000011112 polyethylene naphthalate Substances 0.000 description 1
- 239000012462 polypropylene substrate Substances 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000007650 screen-printing Methods 0.000 description 1
- 150000004756 silanes Chemical class 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 238000004528 spin coating Methods 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 229940124530 sulfonamide Drugs 0.000 description 1
- 238000011282 treatment Methods 0.000 description 1
- JXUKBNICSRJFAP-UHFFFAOYSA-N triethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CCO[Si](OCC)(OCC)CCCOCC1CO1 JXUKBNICSRJFAP-UHFFFAOYSA-N 0.000 description 1
- DQZNLOXENNXVAD-UHFFFAOYSA-N trimethoxy-[2-(7-oxabicyclo[4.1.0]heptan-4-yl)ethyl]silane Chemical compound C1C(CC[Si](OC)(OC)OC)CCC2OC21 DQZNLOXENNXVAD-UHFFFAOYSA-N 0.000 description 1
- 238000009423 ventilation Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Landscapes
- Materials Applied To Surfaces To Minimize Adherence Of Mist Or Water (AREA)
Description
本発明は、撥水性と撥油性を有する撥水撥油性膜を形成するための液組成物及びその製造方法に関する。更に詳しくは、金属酸化物粒子を含む撥水撥油性膜形成用液組成物及びその製造方法に関するものである。 The present invention relates to a liquid composition for forming a water- and oil-repellent film having water and oil repellency, and a method for producing the same. More specifically, the present invention relates to a liquid composition for forming a water- and oil-repellent film containing metal oxide particles, and a method for producing the same.
これまで、アルコール類の有機溶媒中にSiO2、CaCO3、TiO2等の無機化合物を分散させて無機化合物の分散液を調整する工程(第1工程)と、無機化合物の分散液中に、ペルフルオロアルキル基を含むフッ素化合物および触媒を添加して、無機化合物とフッ素化合物とがナノコンポジット化された複合材料を合成する工程(第2工程)と、を含むフッ素含有ナノコンポジット粒子の製造方法が開示されている(例えば、特許文献1(段落[0047]、段落[0057]、段落[0067]~[0070]参照。))。 So far, a method for producing fluorine-containing nanocomposite particles has been disclosed, which includes a step (first step) of preparing a dispersion of an inorganic compound by dispersing an inorganic compound such as SiO2 , CaCO3 , TiO2 , etc. in an organic solvent such as an alcohol, and a step (second step) of adding a fluorine compound containing a perfluoroalkyl group and a catalyst to the dispersion of the inorganic compound to synthesize a composite material in which the inorganic compound and the fluorine compound are nanocomposite (for example, see Patent Document 1 (paragraphs [0047], [0057], and paragraphs [0067] to [0070]).
特許文献1には、第2工程において、無機化合物の分散液中にテトラメトキシシラン(TMOS)、テトラエトキシシラン(TEOS)等のケイ素アルコキシドを含むシランカップリング剤を添加してもよい旨が記載されている。また特許文献1には、フッ素含有ナノコンポジット粒子を水又はアルコール類の有機溶媒に分散させることによって、フッ素含有ナノコンポジット粒子の分散液にして、この分散液をコーティング剤として用いた場合には、各種材料表面に対して撥水撥油性や親水撥油性等の機能を付与することが可能となる旨が記載されている。
しかしながら、特許文献1記載の製造方法で作られたフッ素含有ナノコンポジット粒子の分散液(膜形成用液組成物)では、無機化合物(金属酸化物粒子)が親水撥油性であるため、形成した膜の撥油性能を保持しながら、膜の硬さ及び膜の強度を向上させることができなかった。
However, in the dispersion of fluorine-containing nanocomposite particles (film-forming liquid composition) made by the manufacturing method described in
このため、従来よりも高い撥水性及び撥油性を発揮できるフッ素含有複合粒子が提案されている(例えば、特許文献2(請求項1、段落[0008]、段落[0039]~段落[0054])参照。)。このフッ素含有複合粒子は、シリカ系微粒子の表面が1)フッ素含有化合物及び2)それがシリカ系微粒子のケイ素とシロキサン結合してなるフッ素含有基の少なくとも1種で被覆されている複合粒子であって、前記フッ素含有化合物が、パーフルオロポリエーテルからなる主鎖の少なくとも一方の末端側に[-Si(OR)3](但し、3つのRは、互いに同一又は異なって、水素又は炭素数1~10のアルキル基を示す。)で示される官能基Aを含む化合物であり、前記シリカ系微粒子の単位表面積当たりのフッ素含有量が0.8mg/m2~1.0g/m2であることを特徴とする。
For this reason, fluorine-containing composite particles that can exhibit higher water repellency and oil repellency than conventional ones have been proposed (for example, see Patent Document 2 (
上記フッ素含有複合粒子は、シリカ系微粒子又はその分散液とフッ素含有化合物の溶液とを混合し、特に両者を混合した後、その混合液を攪拌することにより製造される。シリカ系微粒子の分散液及びフッ素含有化合物の溶液は、それぞれ水、水溶性有機溶媒又はそれらの混合溶媒に分散して調製される。シリカ系微粒子の分散液とフッ素含有化合物の溶液を混合した後、混合液を攪拌することにより、官能基Aの一部又は全部がシリカ系微粒子表面のシリカ又はそのシリカ表面の官能基と十分に反応してシロキサン結合を確実に形成させることが可能となる。 The fluorine-containing composite particles are produced by mixing silica-based microparticles or a dispersion thereof with a solution of a fluorine-containing compound, and in particular by mixing the two and then stirring the mixture. The dispersion of silica-based microparticles and the solution of the fluorine-containing compound are each prepared by dispersing in water, a water-soluble organic solvent, or a mixture of these. By mixing the dispersion of silica-based microparticles with the solution of the fluorine-containing compound and then stirring the mixture, it becomes possible for part or all of the functional group A to sufficiently react with the silica on the surface of the silica-based microparticles or the functional groups on the silica surface to reliably form siloxane bonds.
攪拌が完了した後、スラリーの形態で特許文献2記載のフッ素含有複合粒子が得られ、用途に応じて、フッ素含有複合粒子をそのままスラリーの形態で使用するか、或いは必要に応じてスラリーに対して固液分離、洗浄等の処理を施した後、実質的に乾燥した粉末の形態で使用することもできる。フッ素含有複合粒子を更に溶媒に分散させて得られた分散液の形態で使用することもできる。その他にも、前記スラリーを固液分離して得られたケーキを別の溶媒に分散させることによって得られた分散液の形態で使用することも可能である。
After the stirring is completed, the fluorine-containing composite particles described in
特許文献2に開示されたフッ素含有複合粒子のスラリー、フッ素含有複合粒子を溶媒に分散させて得られた分散液等の膜形成用液組成物では、この液組成物を各種の物品に塗布して塗膜を物品表面に形成することにより、物品に撥水性及び/又は撥油性を付与することができるけれども、撥油性能がまだ十分でないこと、及びフッ素含有複合粒子同士の結着力が十分に大きくなく、形成した膜の強度、硬さが高くないこと等の課題がり、更なる改善が求められていた。
In the film-forming liquid compositions disclosed in
本発明の目的は、従来の液組成物で形成した膜と比較して、形成した膜の撥水性、撥油性、膜の強度及び膜の硬さがより高く、膜表面に指紋が目立ちにくい撥水撥油性膜形成用液組成物及びその製造方法を提供することにある。 The object of the present invention is to provide a liquid composition for forming a water- and oil-repellent film, which has higher water repellency, oil repellency, strength and hardness than films formed with conventional liquid compositions, and which makes fingerprints less noticeable on the film surface, and a method for producing the same.
本発明者は、金属酸化物粒子にフッ素系官能基成分を結合させること及びシリカゾルゲルにフッ素系官能基成分を含有させることにより、形成した膜により高い撥油性が付与される点、及び金属酸化物粒子同士をフッ素含有シリカゾルゲルで結着させて膜を形成すると、膜の硬さと膜の摩耗強度が向上し、かつ膜表面に凹凸が形成されて耐指紋性が改善される点に着目し、本発明に到達した。 The inventors of the present invention have arrived at the present invention by noting that by bonding fluorine-based functional group components to metal oxide particles and by incorporating fluorine-based functional group components into silica sol-gel, the formed film is endowed with high oil repellency, and that by forming a film by bonding metal oxide particles together with fluorine-containing silica sol-gel, the hardness and abrasion resistance of the film are improved, and unevenness is formed on the film surface, improving fingerprint resistance.
本発明の第1の観点は、下記の式(19)から式(27)のいずれか1つで示される、ペルフルオロエーテル構造を含む第1フッ素系化合物(A1)が結合した平均粒子径2nm~90nmのフッ素含有金属酸化物粒子(B)と、下記の式(19)から式(27)のいずれか1つで示される、ペルフルオロエーテル構造を含む第2フッ素系化合物(A2)を含有するフッ素含有シリカゾルゲル(C)と、溶媒(D)とを含み、前記溶媒(D)を除く全成分量を100質量%としたとき、前記第1フッ素化合物(A1)と前記第2フッ素化合物(A2)とを合計した含有割合が、1質量%~30質量%であり、前記フッ素含有シリカゾルゲル(C)と前記フッ素含有金属酸化物粒子(B)の質量比(C:B)が、10:90~90:10の範囲にあることを特徴とする撥水撥油性膜形成用液組成物である。 A first aspect of the present invention is a water- and oil-repellent film-forming liquid composition comprising: fluorine-containing metal oxide particles (B) having an average particle size of 2 nm to 90 nm and bonded to a first fluorine-based compound (A1) containing a perfluoroether structure , which is represented by any one of the following formulas (19) to (27); fluorine-containing silica sol-gel (C) containing a second fluorine-based compound (A2) containing a perfluoroether structure , which is represented by any one of the following formulas (19) to (27); and a solvent (D), wherein, when the amount of all components excluding the solvent (D) is taken as 100 mass%, the total content of the first fluorine compound (A1) and the second fluorine compound (A2) is 1 mass% to 30 mass%, and the mass ratio (C:B) of the fluorine-containing silica sol-gel (C) to the fluorine-containing metal oxide particles (B) is in the range of 10:90 to 90:10.
上記式(19)~(27)中、Rはメチル基又はエチル基である。In the above formulas (19) to (27), R is a methyl group or an ethyl group.
本発明の第2の観点は、第1の観点に基づく発明であって、前記第1フッ素化合物(A1)の含有割合が、質量比で前記第2フッ素化合物(A2)の含有割合以上である撥水撥油性膜形成用液組成物である。 A second aspect of the present invention is an invention based on the first aspect, which is a water- and oil-repellent film-forming liquid composition, wherein a content ratio of the first fluorine compound (A1) is equal to or greater than a content ratio of the second fluorine compound (A2) by mass ratio.
本発明の第3の観点は、第1の観点に基づく発明であって、前記フッ素含有金属酸化物粒子(B)に用いる金属酸化物粒子は、Si,Al、Mg、Ca、Ti、Zn及びZrからなる群より選ばれた1種又は2種以上の金属の酸化物粒子である。 A third aspect of the present invention is the invention based on the first aspect, wherein the metal oxide particles used for the fluorine-containing metal oxide particles (B) are oxide particles of one or more metals selected from the group consisting of Si, Al, Mg, Ca, Ti, Zn and Zr.
本発明の第4の観点は、第1の観点に基づく発明であって、前記フッ素含有シリカゾルゲル(C)は、前記フッ素含有シリカゾルゲル(C)を100質量%としたときに、炭素数2~7のアルキレン基成分を0.5質量%~20質量%含む撥水撥油性膜形成用液組成物である。 A fourth aspect of the present invention is the invention based on the first aspect, wherein the fluorine-containing silica sol-gel (C) is a water- and oil-repellent film-forming liquid composition containing 0.5% by mass to 20% by mass of an alkylene group component having 2 to 7 carbon atoms, when the fluorine-containing silica sol-gel (C) is taken as 100% by mass.
本発明の第5の観点は、第1の観点に基づく発明であって、前記溶媒(D)は、水と炭素数1~4のアルコールとの混合溶媒であるか、或いは水と炭素数1~4のアルコールと前記アルコール以外の有機溶媒との混合溶媒である撥水撥油性膜形成用液組成物である。 The fifth aspect of the present invention is an invention based on the first aspect, and is a liquid composition for forming a water- and oil-repellent film, in which the solvent (D) is a mixed solvent of water and an alcohol having 1 to 4 carbon atoms, or a mixed solvent of water, an alcohol having 1 to 4 carbon atoms, and an organic solvent other than the alcohol.
本発明の第6の観点は、図1に示すように、第1の観点の撥水撥油性膜形成用液組成物の製造方法であって、フッ素含有金属酸化物粒子(B)の分散液とフッ素含有シリカゾルゲル液(C)とを混合して撥水撥油性膜形成用液組成物を製造する撥水撥油性膜形成用液組成物の製造方法である。 A sixth aspect of the present invention is a method for producing a water- and oil-repellent film-forming liquid composition according to the first aspect , as shown in FIG. 1, which comprises mixing a dispersion of fluorine-containing metal oxide particles (B) and a fluorine-containing silica sol-gel liquid (C) to produce the water- and oil-repellent film-forming liquid composition .
本発明の第7の観点は、第5の観点に基づく発明であって、図1に示すように、前記フッ素含有金属酸化物粒子(B)の分散液が、金属酸化物粒子の分散液にフッ素系化合物(A1)を添加混合し、この混合液に水と触媒を添加混合して、調製される撥水撥油性膜形成用液組成物の製造方法である。 A seventh aspect of the present invention is an invention based on the fifth aspect, and relates to a method for producing a water- and oil-repellent film-forming liquid composition, in which the dispersion of fluorine-containing metal oxide particles (B) is prepared by adding and mixing a fluorine-based compound (A1) to a dispersion of metal oxide particles, and then adding and mixing water and a catalyst to the mixture, as shown in FIG.
本発明の第8の観点は、第6の観点に基づく発明であって、前記フッ素含有金属酸化物粒子(B)に用いる金属酸化物粒子がSi,Al、Mg、Ca、Ti、Zn及びZrからなる群より選ばれた1種又は2種以上の金属の酸化物粒子である撥水撥油性膜形成用液組成物の製造方法である。 An eighth aspect of the present invention is an invention based on the sixth aspect, and relates to a method for producing a water- and oil-repellent film-forming liquid composition, wherein the metal oxide particles used for the fluorine-containing metal oxide particles (B) are oxide particles of one or more metals selected from the group consisting of Si, Al, Mg, Ca, Ti, Zn and Zr.
本発明の第9の観点は、第5の観点に基づく発明であって、図1に示すように、前記フッ素含有シリカゾルゲル液(C)が、ケイ素アルコキシドとアルコールとフッ素系化合物(A2)と水を混合した混合液に触媒を添加混合して、調製される撥水撥油性膜形成用液組成物の製造方法である。 A ninth aspect of the present invention is an invention based on the fifth aspect, and relates to a method for producing a water- and oil-repellent film-forming liquid composition, in which the fluorine-containing silica sol-gel liquid (C) is prepared by adding a catalyst to a mixed liquid of a silicon alkoxide, an alcohol, a fluorine-based compound (A2) and water, as shown in FIG. 1 .
本発明の第1の観点の撥水撥油性膜形成用液組成物では、成膜したときに、第1フッ素化合物(A1)が結合したフッ素含有金属酸化物粒子(B)を含み、かつ第2フッ素化合物(A2)を含むフッ素含有シリカゾルゲル(C)で金属酸化物粒子同士が結着するため、形成した膜の撥油性が非常に高い。また、膜の硬さと膜の摩耗強度を向上させることができる。形成した膜の表面が平滑でないため、膜表面に指紋を付着させた後に、膜表面に指紋が目立ちにくい。 In the water- and oil-repellent film-forming liquid composition according to the first aspect of the present invention, when a film is formed, the film has very high oil repellency because the metal oxide particles are bound together by the fluorine-containing silica sol-gel (C) containing the first fluorine compound (A1) and the second fluorine compound (A2). In addition , the film has high hardness and abrasion resistance. The surface of the film formed is not smooth, so that fingerprints are not easily noticeable on the film surface after they are applied to the film surface.
本発明の第2の観点の撥水撥油性膜形成用液組成物では、第1フッ素化合物(A1)が第2フッ素化合物(A2)と同じ質量割合であるか、又は第2フッ素化合物(A2)より多く含有するため、バインダ成分であるシリカゾルゲル(C)にフッ素成分が少ないので、塗膜の外観や塗膜の密着性に優れる。 In the water/oil repellent film-forming liquid composition according to the second aspect of the present invention, the first fluorine compound (A1) is contained in the same mass proportion as the second fluorine compound (A2) or in a larger amount than the second fluorine compound (A2), and therefore the binder component, silica sol-gel (C ), contains a small amount of fluorine components, resulting in excellent appearance and adhesion of the coating film.
本発明の第3の観点の液組成物では、フッ素含有金属酸化物粒子(B)に用いる金属酸化物粒子が、Si,Al、Mg、Ca、Ti、Zn及びZrからなる群より選ばれた1種又は2種以上の金属酸化物粒子であるため、多種の金属酸化物粒子の中から、形成する膜の用途又は使用環境に適した金属酸化物粒子を含むことができる。 In the liquid composition according to the third aspect of the present invention, the metal oxide particles used for the fluorine-containing metal oxide particles (B) are one or more types of metal oxide particles selected from the group consisting of Si, Al, Mg, Ca, Ti, Zn and Zr, and therefore it is possible to include metal oxide particles suitable for the application or usage environment of the film to be formed from among the many types of metal oxide particles.
本発明の第4の観点の液組成物では、フッ素含有シリカゾルゲル中に炭素数2~7のアルキレン基成分を0.5質量%~20質量%含むため、撥水撥油性膜を形成する基材に良好に密着し、かつ撥水撥油性膜の厚さが均一になり、撥水撥油性膜により一層優れた撥油性能を付与することができる。 In the liquid composition according to the fourth aspect of the present invention, the fluorine-containing silica sol-gel contains 0.5% by mass to 20% by mass of an alkylene group component having 2 to 7 carbon atoms, and therefore the water- and oil-repellent film adheres well to the substrate on which it is formed, and the thickness of the water- and oil-repellent film becomes uniform, making it possible to impart even more excellent oil repellency to the water- and oil-repellent film.
本発明の第5の観点の液組成物では、溶媒(D)は、水と炭素数1~4のアルコールとの混合溶媒であるか、或いは水と炭素数1~4のアルコールとこのアルコール以外の有機溶媒との混合溶媒であるため、液組成物の乾燥速度が向上し、液組成物の粘度が低減する。このため、この液組成物は取り扱いが容易で、基材への成膜性に優れる。 In the liquid composition according to the fifth aspect of the present invention, the solvent (D) is a mixed solvent of water and an alcohol having 1 to 4 carbon atoms, or a mixed solvent of water, an alcohol having 1 to 4 carbon atoms, and an organic solvent other than this alcohol, so that the drying speed of the liquid composition is improved and the viscosity of the liquid composition is reduced. Therefore, this liquid composition is easy to handle and has excellent film-forming properties on a substrate.
本発明の第6の観点の液組成物の製造方法では、図1に示すように、フッ素含有金属酸化物粒子(B)の分散液とフッ素含有シリカゾルゲル液(C)とを混合して撥水撥油性膜形成用液組成物を製造する。これにより、粒子表面が撥水撥油性である金属酸化物粒子がフッ素系化合物を含むシリカゾルゲル中に存在し、液組成物を成膜したときに、膜により一層の撥水撥油性を付与する。 In the method for producing a liquid composition according to the sixth aspect of the present invention, a liquid composition for forming a water- and oil-repellent film is produced by mixing a dispersion of fluorine-containing metal oxide particles (B) with a fluorine-containing silica sol-gel liquid (C) as shown in Fig. 1. As a result, metal oxide particles having water- and oil-repellent particle surfaces are present in a silica sol-gel containing a fluorine-based compound, and when the liquid composition is formed into a film, an even greater layer of water- and oil-repellency is imparted to the film.
本発明の第7の観点の液組成物の製造方法では、金属酸化物粒子の分散液にフッ素系化合物を添加混合し、この混合液に水と触媒を添加混合するため、フッ素含有金属酸化物粒子(B)が均一に分散した分散液が得られる。 In the method for producing a liquid composition according to the seventh aspect of the present invention, a fluorine-based compound is added and mixed into a dispersion of metal oxide particles, and water and a catalyst are then added and mixed into this mixture, thereby obtaining a dispersion in which the fluorine-containing metal oxide particles (B) are uniformly dispersed.
本発明の第8の観点の液組成物の製造方法では、フッ素含有金属酸化物粒子(B)に用いる金属酸化物粒子が、Si,Al、Mg、Ca、Ti、Zn及びZrからなる群より選ばれた1種又は2種以上の金属酸化物粒子であるため、多種の金属酸化物粒子の中から、形成する膜の用途又は使用環境に適した金属酸化物粒子を含んだ液組成物を製造することができる。 In the liquid composition production method according to the eighth aspect of the present invention, the metal oxide particles used for the fluorine-containing metal oxide particles (B) are one or more types of metal oxide particles selected from the group consisting of Si, Al, Mg, Ca, Ti, Zn and Zr, so that it is possible to produce a liquid composition containing metal oxide particles suitable for the application or usage environment of the film to be formed, from among many types of metal oxide particles.
本発明の第9の観点の液組成物の製造方法では、ケイ素アルコキシドとアルコールとフッ素系化合物と水の混合液に触媒を添加混合して調製されたフッ素含有シリカゾルゲル液(C)は、フッ素含有金属酸化物粒子のバインダとして作用するとともに、液組成物を基材表面に成膜したときに、膜を基材表面に堅牢に結着させるとともにより膜に一層の撥水撥油性を付与する。 In the method for producing a liquid composition according to the ninth aspect of the present invention, the fluorine-containing silica sol-gel liquid (C) prepared by adding and mixing a catalyst to a mixed liquid of a silicon alkoxide, an alcohol, a fluorine-based compound and water acts as a binder for the fluorine-containing metal oxide particles, and when the liquid composition is formed into a film on the surface of a substrate, it firmly binds the film to the surface of the substrate and imparts further water and oil repellency to the film.
次に本発明を実施するための形態について図面を参照して説明する。 Next, the embodiment of the present invention will be described with reference to the drawings.
〔撥水撥油性膜形成用液組成物の製造方法〕
撥水撥油性膜形成用液組成物は次の方法により、概略製造される。
図1に示すように、金属酸化物粒子11と有機溶媒12を混合して金属酸化物粒子の分散液13を調製する。この分散液13に第1フッ素系官能基成分(A1)を含むフッ素系化合物14を混合し、更に水15と触媒16を混合してフッ素含有金属酸化物粒子の分散液17を調製する。一方、ケイ素アルコキシド21とアルコール22と第2フッ素系官能基成分(A2)を含むフッ素系化合物23と水25と、必要に応じてアルキレン基成分24を混合し、この混合液に触媒26を加えることにより、フッ素含有シリカゾルゲル液27を調製する。
このフッ素含有シリカゾルゲル液27に溶媒28を混合し、この混合液と上記フッ素含有金属酸化物粒子の分散液17とを混合することにより、撥水撥油性膜形成用液組成物30を製造する。以下、各工程毎に詳しく述べる。
[Method for producing water- and oil-repellent film-forming liquid composition]
The water- and oil-repellent film-forming liquid composition is generally produced by the following method.
As shown in Fig. 1,
A
〔金属酸化物粒子分散液の調製〕
先ず、有機溶媒中に、金属酸化物粒子を分散させて金属酸化物粒子の分散液を調製する。金属酸化物粒子は、2nm~90nm、好ましくは2nm~85nmの平均粒子径を有する。平均粒子径が2nm未満では、金属酸化物粒子の凝集が起こりやすくなり、媒体中に分散しにくくなる。90nmを超えると、液組成物を成膜したときに、金属酸化物粒子が撥水撥油性膜から脱落する。金属酸化物粒子としては、SiO2、Al2O3、MgO、CaO、TiO2、ZnO、ZrO2の粒子、これらの混合粒子、MgAl2O4等の複合酸化物粒子等が例示される。
有機溶媒としては、メタノール、エタノール、イソプロパノール(以下、IPAということもある。)、テトラヒドロフラン、ヘキサン、クロロホルム、トルエン、酢酸エチル、ジメチルスルホキシド(DMSO)、ジメチルホルムアミド(DMF)、アセトン、フッ素系溶剤などが例示される。これらの中でも、沸点が120℃未満の炭素数1~4の範囲にあるメタノール、エタノール、イソプロパノールなどのアルコールが好ましい。なお、本明細書において、金属酸化物粒子の平均粒子径とは、透過型電子顕微鏡(TEM)で観察した粒子形状のうち、200点の粒子サイズを画像解析により測定したものの平均値をいう。
[Preparation of Metal Oxide Particle Dispersion]
First, the metal oxide particles are dispersed in an organic solvent to prepare a dispersion liquid of the metal oxide particles. The metal oxide particles have an average particle size of 2 nm to 90 nm, preferably 2 nm to 85 nm. If the average particle size is less than 2 nm, the metal oxide particles are likely to aggregate and are difficult to disperse in the medium. If the average particle size exceeds 90 nm, the metal oxide particles will fall off from the water-repellent and oil-repellent film when the liquid composition is formed into a film. Examples of the metal oxide particles include particles of SiO 2 , Al 2 O 3 , MgO, CaO, TiO 2 , ZnO, and ZrO 2 , mixed particles of these, and composite oxide particles such as MgAl 2 O 4 .
Examples of organic solvents include methanol, ethanol, isopropanol (hereinafter sometimes referred to as IPA), tetrahydrofuran, hexane, chloroform, toluene, ethyl acetate, dimethyl sulfoxide (DMSO), dimethylformamide (DMF), acetone, and fluorine-based solvents. Among these, alcohols having a boiling point of less than 120° C. and a carbon number in the range of 1 to 4, such as methanol, ethanol, and isopropanol, are preferred. In this specification, the average particle size of the metal oxide particles refers to the average value of the particle sizes of 200 points among the particle shapes observed with a transmission electron microscope (TEM) measured by image analysis.
〔フッ素含有金属酸化物粒子分散液の調製〕
次に、調製された金属酸化物粒子の分散液中に、上述した式(1)又は式(2)で表される第1フッ素系官能基成分(A1)を含むフッ素系化合物を添加して、金属酸化物粒子と第1フッ素系官能基成分(A1)とがナノコンポジット化された複合材料を合成する。更に反応を促進するために、水及び触媒を添加する。これにより、フッ素含有金属酸化物粒子の分散液を調製する。
[Preparation of Fluorine-Containing Metal Oxide Particle Dispersion]
Next, a fluorine-based compound containing the first fluorine-based functional group component (A1) represented by the above formula (1) or formula (2) is added to the prepared dispersion of metal oxide particles to synthesize a composite material in which the metal oxide particles and the first fluorine-based functional group component (A1) are nanocomposite. In order to further promote the reaction, water and a catalyst are added. In this way, a dispersion of fluorine-containing metal oxide particles is prepared.
上記触媒としては、有機酸、無機酸又はチタン化合物が挙げられ、有機酸としてはギ酸、シュウ酸が例示され、無機酸としては塩酸、硝酸、リン酸が例示され、チタン化合物としてはテトラプロポキシチタン、テトラブトキシチタン、テトライソプロポキシチタン、乳酸チタン等が例示される。触媒は上記のものに限定されない。上記水としては、不純物の混入防止のため、イオン交換水や純水等を使用するのが望ましい。 The catalyst may be an organic acid, an inorganic acid, or a titanium compound. Examples of organic acids include formic acid and oxalic acid. Examples of inorganic acids include hydrochloric acid, nitric acid, and phosphoric acid. Examples of titanium compounds include titanium tetrapropoxide, titanium tetrabutoxide, titanium tetraisopropoxide, and titanium lactate. The catalyst is not limited to the above. As the water, it is preferable to use ion-exchanged water or pure water to prevent the inclusion of impurities.
第1フッ素系官能基成分(A1)を含むフッ素系化合物は、下記一般式(3)又は式(4)で示される。これらの式(3)又は式(4)中のペルフルオロエーテル基としては、より具体的には、下記式(5)~(13)で示されるペルフルオロエーテル構造を挙げることができる。 The fluorine-based compound containing the first fluorine-based functional group component (A1) is represented by the following general formula (3) or formula (4). More specifically, the perfluoroether group in formula (3) or formula (4) may be the perfluoroether structure represented by the following formulas (5) to (13).
また、上記式(3)及び式(4)中のXとしては、下記式(14)~(18)で示される構造を挙げることができる。なお、下記式(14)はエーテル結合、下記式(15)はエステル結合、下記式(16)はアミド結合、下記式(17)はウレタン結合、下記式(18)はスルホンアミド結合を含む例を示している。 In addition, examples of X in the above formulas (3) and (4) include structures shown in the following formulas (14) to (18). Note that the following formula (14) shows an example containing an ether bond, the following formula (15) an ester bond, the following formula (16) an amide bond, the following formula (17) a urethane bond, and the following formula (18) a sulfonamide bond.
ここで、上記式(14)~(18)中、R2及びR3は炭素数が0から10の炭化水素基、R4は水素原子又は炭素数1から6の炭化水素基である。R3の炭化水素基の例とは、メチレン基、エチレン基等のアルキレン基が挙げられ、R4の炭化水素基の例とは、メチル基、エチル基等のアルキル基の他、フェニル基等も挙げられる。 In the above formulas (14) to (18), R2 and R3 are hydrocarbon groups having 0 to 10 carbon atoms, and R4 is a hydrogen atom or a hydrocarbon group having 1 to 6 carbon atoms. Examples of the hydrocarbon group for R3 include alkylene groups such as a methylene group and an ethylene group, and examples of the hydrocarbon group for R4 include alkyl groups such as a methyl group and an ethyl group, as well as a phenyl group.
また、上記式(3)及び式(4)中、R1は、メチル基、エチル基等が挙げられる。 In the above formulas (3) and (4), R 1 may be a methyl group, an ethyl group, or the like.
また、上記式(3)及び式(4)中、Zは、加水分解されてSi-O-Si結合を形成可能な加水分解性基であれば特に限定されるものではない。このような加水分解性基としては、具体的には、例えば、メトキシ基、エトキシ基、プロポキシ基、ブトキシ基などのアルコキシ基、フェノキシ基、ナフトキシ基などのアリールオキシ基、ベンジルオキシ基、フェネチルオキシ基などのアラルキルオキシ基、アセトキシ基、プロピオニルオキシ基、ブチリルオキシ基、バレリルオキシ基、ピバロイルオキシ基、ベンゾイルオキシ基などのアシルオキシ基等が挙げられる。これらの中でも、メトキシ基、エトキシ基を適用することが好ましい。 In addition, in the above formulas (3) and (4), Z is not particularly limited as long as it is a hydrolyzable group that can be hydrolyzed to form a Si-O-Si bond. Specific examples of such hydrolyzable groups include alkoxy groups such as methoxy, ethoxy, propoxy, and butoxy groups, aryloxy groups such as phenoxy and naphthoxy groups, aralkyloxy groups such as benzyloxy and phenethyloxy groups, and acyloxy groups such as acetoxy, propionyloxy, butyryloxy, valeryloxy, pivaloyloxy, and benzoyloxy groups. Among these, it is preferable to apply a methoxy group or an ethoxy group.
ここで、上記式(3)又は式(4)で表されるペルフルオロエーテル構造を有する第1フッ素系官能基成分(A1)を含むフッ素系化合物の具体例としては、例えば、下記式(19)~(27)で表される構造が挙げられる。なお、下記式(19)~(27)中、Rはメチル基又はエチル基である。 Specific examples of fluorine-based compounds containing the first fluorine-based functional group component (A1) having a perfluoroether structure represented by the above formula (3) or formula (4) include structures represented by the following formulas (19) to (27). In the following formulas (19) to (27), R is a methyl group or an ethyl group.
〔フッ素含有シリカゾルゲル液の調製〕
先ず、ケイ素アルコキシドとしてのテトラメトキシシラン又はテトラエトキシシランと、沸点が120℃未満の炭素数1~4の範囲にあるアルコールと、上述した式(1)又は式(2)で表される第2フッ素系官能基成分(A2)を含むフッ素系化合物と、水とを混合して混合液を調製する。このときアルキレン基成分となるエポキシ基含有シランを一緒に混合してもよい。このケイ素アルコキシドとしては、具体的には、テトラメトキシシラン(TMOS)、そのオリゴマー又はテトラエトキシシラン(TEOS)、そのオリゴマーが挙げられる。例えば、耐久性の高い撥水撥油性膜を得る目的には、テトラメトキシシランを用いることが好ましく、一方、加水分解時に発生するメタノールを避ける場合は、テトラエトキシシランを用いることが好ましい。
[Preparation of fluorine-containing silica sol-gel liquid]
First, a mixed solution is prepared by mixing tetramethoxysilane or tetraethoxysilane as silicon alkoxide, alcohol having a boiling point of less than 120° C. and a carbon number in the range of 1 to 4, a fluorine-based compound containing the second fluorine-based functional group component (A2) represented by the above formula (1) or formula (2), and water. At this time, an epoxy group-containing silane as an alkylene group component may be mixed together. Specific examples of this silicon alkoxide include tetramethoxysilane (TMOS) and its oligomers, or tetraethoxysilane (TEOS) and its oligomers. For example, in order to obtain a highly durable water-repellent and oil-repellent film, it is preferable to use tetramethoxysilane, while in order to avoid methanol generated during hydrolysis, it is preferable to use tetraethoxysilane.
フッ素系化合物に含まれる第2フッ素系官能基成分(A2)は、上述した式(1)又は式(2)表され、第1フッ素系官能基成分(A1)を含む具体的なフッ素系化合物と第2フッ素系官能基成分(A2)を含む具体的なフッ素系化合物とは、同一であっても、異なってもよい。 The second fluorine-based functional group component (A2) contained in the fluorine-based compound is represented by the above-mentioned formula (1) or formula (2), and the specific fluorine-based compound containing the first fluorine-based functional group component (A1) and the specific fluorine-based compound containing the second fluorine-based functional group component (A2) may be the same or different.
上記アルキレン基成分となるエポキシ基含有シランとしては、具体的には、2-(3,4-エポキシシクロヘキシル)エチルトリメトキシシラン、3-グリシドキシプロピルメチルジメトキシシラン、3-グリシドキシプロピルメチルジエトキシシラン、3-グリシドキシプロピルトリメトキシシラン、3-グリシドキシプロピルトリエトキシシラン又は多官能エポキシシランが挙げられる。アルキレン基成分はケイ素アルコキシドとアルキレン基成分の合計質量に対して1質量%~40質量%、好ましくは2.5質量%~20質量%含まれる。アルキレン基成分が下限値の1質量%未満では、水酸基を含まない基材に膜を形成した場合に、基材への密着性が不十分になる。また上限値の40質量%を超えると、形成した膜の耐久性が低くなる。アルキレン基成分を上記1質量%~40質量%の範囲になるようにエポキシ基含有シランを含むと、エポキシ基も加水分解重合過程において開環して重合に寄与し、これにより乾燥過程にレベリング性が改善し膜厚さが均一になる。なお、基材がガラス等の親水基を含む場合には、アルキレン基成分の含有量は極少量であるか、若しくはゼロでもよい。一方、基材が親水基を含まない場合には、このアルキレン基成分をシリカゾルゲル(C)中、0.5質量%~20質量%含むことが好ましい。 Specific examples of the epoxy group-containing silane that is the alkylene group component include 2-(3,4-epoxycyclohexyl)ethyltrimethoxysilane, 3-glycidoxypropylmethyldimethoxysilane, 3-glycidoxypropylmethyldiethoxysilane, 3-glycidoxypropyltrimethoxysilane, 3-glycidoxypropyltriethoxysilane, and polyfunctional epoxysilane. The alkylene group component is contained in an amount of 1% to 40% by mass, preferably 2.5% to 20% by mass, based on the total mass of the silicon alkoxide and the alkylene group component. If the alkylene group component is less than the lower limit of 1% by mass, when a film is formed on a substrate that does not contain hydroxyl groups, the adhesion to the substrate becomes insufficient. If the amount exceeds the upper limit of 40% by mass, the durability of the formed film decreases. If the epoxy group-containing silane is included so that the alkylene group component falls within the above-mentioned range of 1% by mass to 40% by mass, the epoxy group also opens during the hydrolysis polymerization process and contributes to the polymerization, thereby improving the leveling property during the drying process and making the film thickness uniform. If the substrate contains a hydrophilic group such as glass, the content of the alkylene group component may be very small or may be zero. On the other hand, if the substrate does not contain a hydrophilic group, it is preferable that the alkylene group component is included in the silica sol-gel (C) at 0.5% by mass to 20% by mass.
沸点が120℃未満の炭素数1~4の範囲にあるアルコールは、上述したアルコールが挙げられる。特にメタノール又はエタノールが好ましい。これらのアルコールは、ケイ素アルコキドとの混合がしやすいためである。上記水としては、不純物の混入防止のため、イオン交換水や純水等を使用するのが望ましい。ケイ素アルコキシドに、或いはケイ素アルコキシドとエポキシ基含有シランに、炭素数1~4の範囲にあるアルコールと水を添加して、好ましくは10℃~30℃の温度で5分~20分間撹拌することにより混合液を調製する。 Examples of alcohols having a carbon number of 1 to 4 and a boiling point of less than 120°C include the alcohols mentioned above. Methanol or ethanol are particularly preferred because these alcohols are easily mixed with silicon alkoxides. As the water, it is preferable to use ion-exchanged water or pure water to prevent the inclusion of impurities. A mixture is prepared by adding an alcohol having a carbon number of 1 to 4 and water to silicon alkoxide, or to silicon alkoxide and epoxy group-containing silane, and stirring for 5 to 20 minutes at a temperature of 10°C to 30°C.
上記調製された混合液に触媒を添加混合する。この触媒としては、有機酸、無機酸又はチタン化合物が例示される。このとき液温を好ましくは30℃~80℃の温度に保持して、好ましくは1時間~24時間撹拌する。これにより、フッ素含有シリカゾルゲル液が調製される。なお、次の工程のために、フッ素含有シリカゾルゲル液にアルコールを添加混合してもよい。 A catalyst is added to the mixed liquid prepared above. Examples of the catalyst include an organic acid, an inorganic acid, or a titanium compound. At this time, the liquid temperature is preferably kept at 30°C to 80°C, and the mixture is stirred for preferably 1 hour to 24 hours. This prepares a fluorine-containing silica sol-gel liquid. Note that alcohol may be added to the fluorine-containing silica sol-gel liquid for the next step.
上記アルコールを添加混合した場合には、フッ素含有シリカゾルゲル液は、ケイ素アルコキシドを2質量%~50質量%、炭素数1~4の範囲にあるアルコールを20質量%~98質量%、水を0.1質量%~40質量%、触媒として0.01質量%~5質量%の割合で含有する。アルキレン基成分となるエポキシ基含有シランを混合した場合には、エポキシ基含有シランを最大30質量%まで含有する。 When the above alcohol is added and mixed, the fluorine-containing silica sol-gel liquid contains 2% by mass to 50% by mass of silicon alkoxide, 20% by mass to 98% by mass of alcohol having 1 to 4 carbon atoms, 0.1% by mass to 40% by mass of water, and 0.01% by mass to 5% by mass of catalyst. When an epoxy group-containing silane, which is an alkylene group component, is mixed, the epoxy group-containing silane can be contained up to a maximum of 30% by mass.
炭素数1~4の範囲にあるアルコールの割合を上記範囲に限定したのは、アルコールの割合が下限値未満では、ケイ素アルコキシドが、溶液中に溶解せず分離してしまうこと、ケイ素アルコキシドの加水分解反応中に反応液がゲル化しやすく、一方、上限値を超えると、加水分解に必要な水、触媒量が相対的に少なくなるために、加水分解の反応性が低下して、重合が進まず、膜の密着性が低下するためである。水の割合を上記範囲に限定したのは、下限値未満では加水分解速度が遅くなるために、重合が進まず、撥水撥油性膜の密着性が不十分になり、一方、上限値を超えると加水分解反応中に反応液がゲル化し、水が多過ぎるためケイ素アルコキシド化合物がアルコール水溶液に溶解せず、分離する不具合を生じるからである。 The proportion of alcohol having 1 to 4 carbon atoms is limited to the above range because if the proportion of alcohol is less than the lower limit, the silicon alkoxide does not dissolve in the solution and separates, and the reaction liquid is prone to gelling during the hydrolysis reaction of the silicon alkoxide, while if the proportion exceeds the upper limit, the amount of water and catalyst required for hydrolysis becomes relatively small, which reduces the reactivity of the hydrolysis, prevents polymerization, and reduces the adhesion of the film. The proportion of water is limited to the above range because if the proportion is less than the lower limit, the hydrolysis rate becomes slow, which prevents polymerization from proceeding and results in insufficient adhesion of the water- and oil-repellent film, while if the proportion exceeds the upper limit, the reaction liquid gels during the hydrolysis reaction, and the silicon alkoxide compound does not dissolve in the alcohol aqueous solution due to the excessive water, resulting in the problem of separation.
シリカゾルゲル中のSiO2濃度(SiO2分)は1質量%~40質量%であるものが好ましい。このSiO2濃度が下限値未満では、重合が不十分であり、膜の密着性の低下やクラックの発生が起こり易く、上限値を超えると、相対的に水の割合が高くなりケイ素アルコキシドが溶解せず、反応液がゲル化する不具合を生じる。 The SiO2 concentration ( SiO2 content) in the silica sol-gel is preferably 1% by mass to 40% by mass. If the SiO2 concentration is less than the lower limit, polymerization is insufficient, and the adhesion of the film is reduced and cracks are likely to occur, while if the SiO2 concentration exceeds the upper limit, the proportion of water becomes relatively high, silicon alkoxide does not dissolve, and the reaction solution gels.
有機酸、無機酸又はチタン化合物は加水分解反応を促進させるための触媒として機能する。有機酸としてはギ酸、シュウ酸が例示され、無機酸としては塩酸、硝酸、リン酸が例示され、チタン化合物としてはテトラプロポキシチタン、テトラブトキシチタン、テトライソプロポキシチタン、乳酸チタン等が例示される。触媒は上記のものに限定されない。上記触媒の割合を上記範囲に限定したのは、下限値未満では反応性に乏しく重合が不十分になるため、膜が形成されず、一方、上限値を超えても反応性に影響はないが、残留する酸により、膜の形成された基材が腐食等を生じ易い。 The organic acid, inorganic acid, or titanium compound functions as a catalyst to promote the hydrolysis reaction. Examples of organic acids include formic acid and oxalic acid, examples of inorganic acids include hydrochloric acid, nitric acid, and phosphoric acid, and examples of titanium compounds include tetrapropoxytitanium, tetrabutoxytitanium, tetraisopropoxytitanium, and titanium lactate. The catalyst is not limited to the above. The proportion of the catalyst is limited to the above range because if it is less than the lower limit, the reactivity is poor and polymerization is insufficient, so a film is not formed, while if it exceeds the upper limit, there is no effect on reactivity, but the substrate on which the film is formed is prone to corrosion due to the remaining acid.
〔撥水撥油性膜形成用液組成物〕
本実施の形態の撥水撥油性膜形成用液組成物は、上記製造方法で製造され、前述した第1フッ素系官能基成分(A1)が結合した金属酸化物粒子(B)と、前述した第2フッ素系官能基成分(A2)を含有するシリカゾルゲル(C)と、溶媒(D)(図1の符号28で示される。)とを含む。溶媒(D)は、水、メタノール、エタノール、イソプロパノール(以下、IPAということもある。)、テトラヒドロフラン、ヘキサン、クロロホルム、トルエン、酢酸エチル、ジメチルスルホキシド(DMSO)、ジメチルホルムアミド(DMF)、アセトン、フッ素系溶剤などが例示される。これらのフッ素系官能基成分(A1)及び(A2)は、上記の一般式(1)又は式(2)で示されるペルフルオロエーテル構造を有し、溶媒(D)を除く全成分量を100質量%としたとき、液組成物中、合計して、1質量%~30質量%含まれる。フッ素系官能基成分の合計した含有割合(A1+A2)が1質量%未満では形成した膜に撥油性を付与できず、30質量%を超えると膜の弾き等が発生し成膜性に劣る。好ましいフッ素系官能基成分の合計した含有割合(A1+A2)は2質量%~28質量%である。
[Water- and oil-repellent film-forming liquid composition]
The water- and oil-repellent film-forming liquid composition of the present embodiment is produced by the above-mentioned production method, and contains metal oxide particles (B) to which the first fluorine-based functional group component (A1) is bonded, silica sol-gel (C) containing the second fluorine-based functional group component (A2), and a solvent (D) (indicated by
また第1フッ素官能基成分(A1)は、第2フッ素官能基成分(A2)と同じ質量割合であるか、又は第2フッ素官能基成分(A2)より多く含有することが塗膜の外観や塗膜の密着性が優れるため、好ましい。 In addition, it is preferable that the first fluorine functional group component (A1) is contained in the same mass proportion as the second fluorine functional group component (A2) or in a larger amount than the second fluorine functional group component (A2), since this leads to excellent appearance and adhesion of the coating film.
上述したように、本実施の形態の撥水撥油性膜形成用液組成物に含まれるフッ素系化合物は、分子内に酸素原子に炭素数が6以下の短鎖長のペルフルオロアルキル基とペルフルオロアルキレン基が複数結合したペルフルオロエーテル基を有しており、分子内のフッ素含有率が高いため、形成した膜に優れた撥水撥油性を付与することができる。ペルフルオロエーテル構造の具体例としては、上述した式(19)~(27)で示される構造を挙げることができる。 As described above, the fluorine-based compound contained in the water- and oil-repellent film-forming liquid composition of this embodiment has a perfluoroether group in which multiple short-chain perfluoroalkyl groups and perfluoroalkylene groups having 6 or less carbon atoms are bonded to an oxygen atom in the molecule, and the high fluorine content in the molecule can impart excellent water- and oil-repellency to the formed film. Specific examples of perfluoroether structures include the structures shown by the above formulas (19) to (27).
更に本実施の形態の撥水撥油性膜形成用液組成物は、シリカゾルゲル(C)と金属酸化物粒子(B)の質量比(C:B)が、10:90~90:10の範囲にあることが必要である。即ち、シリカゾルゲル(C)と金属酸化物粒子(B)を合計した量を100質量%とするとき、シリカゾルゲル(C)が10質量%未満であって金属酸化物粒子(B)が90質量%を超える場合には、液組成物中のバインダ成分が少なくなり過ぎて、後述する膜のセロテープ(登録商標)試験で、形成した膜が基材から剥離し易くなる。反対に金属酸化物粒子(B)が10質量%未満であってシリカゾルゲル(C)が90質量%を超える場合には、金属酸化物粒子が少な過ぎて、後述する膜の強度試験で、膜が基材から剥離し易くなる。好ましい質量比(C:B)は、20:80~80:20である。 Furthermore, in the liquid composition for forming a water- and oil-repellent film of this embodiment, the mass ratio (C:B) of silica sol-gel (C) to metal oxide particles (B) must be in the range of 10:90 to 90:10. That is, when the total amount of silica sol-gel (C) and metal oxide particles (B) is taken as 100 mass%, if the amount of silica sol-gel (C) is less than 10 mass% and the amount of metal oxide particles (B) is more than 90 mass%, the binder component in the liquid composition becomes too small, and the formed film is easily peeled off from the substrate in the Cellophane Tape (registered trademark) test of the film described below. On the other hand, if the amount of metal oxide particles (B) is less than 10 mass% and the amount of silica sol-gel (C) is more than 90 mass%, the amount of metal oxide particles is too small, and the film is easily peeled off from the substrate in the film strength test described below. The preferred mass ratio (C:B) is 20:80 to 80:20.
本実施の形態の撥水撥油性膜形成用液組成物がフッ素含有金属酸化物粒子の分散液と、フッ素含有シリカゾルゲル液を含むため、基材表面に成膜したときに、従来の液組成物と比較して、より一層優れた撥油性能を付与するとともに、撥水撥油性膜の基材表面への密着性に優れ、剥離しにくい高い強度の撥水撥油性膜が得られる。 The liquid composition for forming a water- and oil-repellent film of this embodiment contains a dispersion of fluorine-containing metal oxide particles and a fluorine-containing silica sol-gel liquid, and therefore when formed on the surface of a substrate, it imparts even better oil repellency than conventional liquid compositions, and also provides a water- and oil-repellent film with excellent adhesion to the substrate surface and high strength that is difficult to peel off.
〔撥水撥油性膜の基材表面への形成方法〕
本実施形態の撥水撥油性膜を基材表面に形成するには、撥水撥油性膜形成用液組成物を基材上に塗布した後に、大気中で室温乾燥させて上記液組成物を硬化することにより形成される。この基材としては、特に限定されないが、ステンレス鋼(SUS)、アルミニウム、鉄等の金属板、窓ガラス、鏡等のガラス、タイル、ポリ塩化ビニル(PVC)等のプラスチック又はポリエチレンテレフタレート(PET)、ポリブチレンテレフタレート、ポリエチレンナフタレート等のポリエステルフィルム等が挙げられる。上記液組成物の塗布方法としては、スクリーン印刷法、バーコート法、ダイコート法、ドクターブレード、スピン法、刷毛塗り法等が挙げられる。
[Method of forming a water-repellent and oil-repellent film on a substrate surface]
To form the water-repellent and oil-repellent film on the surface of a substrate, the water-repellent and oil-repellent film-forming liquid composition is applied to the substrate, and then dried at room temperature in the air to harden the liquid composition.The substrate is not particularly limited, but includes metal plates such as stainless steel (SUS), aluminum, and iron, glass such as window glass and mirror, tiles, plastics such as polyvinyl chloride (PVC), and polyester films such as polyethylene terephthalate (PET), polybutylene terephthalate, and polyethylene naphthalate.The coating method of the liquid composition includes screen printing, bar coating, die coating, doctor blade, spin coating, brush coating, and the like.
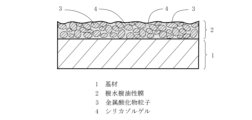
図2に示すように、基材1の表面に形成された撥水撥油性膜2は、粒子表面がフッ素系官能基成分に覆われた多数の金属酸化物粒子3がバインダとしてのフッ素含有シリカゾルゲル4で結着して構成される。撥水撥油性膜2はフッ素系官能基成分が結合した金属酸化物粒子3とフッ素含有シリカゾルゲル4を含むため、膜の撥油性能が保持される。また平均粒子径2nm~90nmの金属酸化物粒子3同士がシリカゾルゲル4により、膜2中で結合するため、膜2の硬さと膜2の摩耗強度を向上させることができる。また金属酸化物粒子3の存在により膜表面が凹凸になり、膜2表面に指紋を付着させた後に、膜表面に指紋が目立ちにくい利点もある。膜厚は、金属酸化物粒子の粒子径と膜成分中の金属酸化物粒子の含有割合を変えることにより制御することができる。
As shown in FIG. 2, the water- and oil-
次に本発明の実施例を比較例とともに詳しく説明する。先ず、金属酸化物粒子の分散液を調製するための合成例1~9及び比較合成例1~2を説明し、次いでフッ素含有シリカゾルゲル液を調製するための合成例10~13及び比較合成例3を説明し、次にこれらの合成例及び比較合成例を用いた撥水撥油性膜形成用液組成物の製造に関する実施例1~9及び比較例1~7を説明する。 Next, examples of the present invention will be described in detail together with comparative examples. First, Synthesis Examples 1 to 9 and Comparative Synthesis Examples 1 and 2 for preparing a dispersion liquid of metal oxide particles will be described, followed by Synthesis Examples 10 to 13 and Comparative Synthesis Example 3 for preparing a fluorine-containing silica sol-gel liquid, and then Examples 1 to 9 and Comparative Examples 1 to 7 for producing a water- and oil-repellent film-forming liquid composition using these Synthesis Examples and Comparative Synthesis Examples will be described.
〔金属酸化物粒子分散液を調製するための合成例1~9、比較合成例1~2〕
<合成例1>
平均粒子径が12nmの二酸化ケイ素のIPA分散液(IPA-ST、日産化学社製、SiO2濃度30%)が50.0g入ったビーカーに、上述した式(19)で表されるフッ素系化合物を1.50g添加し混合した。次に、水を0.10g添加し混合した。更に、硝酸を0.005g添加し、40℃で2時間混合し、フッ素系化合物が二酸化ケイ素粒子に結合した二酸化ケイ素(シリカ)粒子の分散液を得た。
[Synthesis Examples 1 to 9 for preparing metal oxide particle dispersion liquid, and Comparative Synthesis Examples 1 to 2]
<Synthesis Example 1>
To a beaker containing 50.0 g of an IPA dispersion of silicon dioxide having an average particle size of 12 nm (IPA-ST, manufactured by Nissan Chemical Industries, Ltd., SiO2
<合成例2>
平均粒子径が45nmの二酸化ケイ素のIPA分散液(IPA-ST-L、日産化学社製、SiO2濃度30%)が50.0g入ったビーカーに、上述した式(20)で表されるフッ素系化合物を1.50g添加し混合した。次に、水を0.10g添加し混合した。更に、硝酸を0.005g添加し、以下、合成例1と同様にして二酸化ケイ素(シリカ)粒子の分散液を得た。
<Synthesis Example 2>
1.50 g of the fluorine-based compound represented by the above formula (20) was added and mixed into a beaker containing 50.0 g of an IPA dispersion of silicon dioxide having an average particle size of 45 nm (IPA-ST-L, manufactured by Nissan Chemical Industries, Ltd., SiO2
<合成例3>
平均粒子径が80nmの二酸化ケイ素のIPA分散液(IPA-ST-ZL、日産化学社製、SiO2濃度30%)が50.0g入ったビーカーに、上述した式(21)で表されるフッ素系化合物を1.50g添加し混合した。次に、水を0.10g添加し混合した。更に、硝酸を0.005g添加し、以下、合成例1と同様にして二酸化ケイ素(シリカ)粒子の分散液を得た。
<Synthesis Example 3>
1.50 g of the fluorine-based compound represented by the above formula (21) was added and mixed into a beaker containing 50.0 g of an IPA dispersion of silicon dioxide having an average particle size of 80 nm (IPA-ST-ZL, manufactured by Nissan Chemical Industries, Ltd., SiO2
<合成例4>
合成例3と同じ二酸化ケイ素のIPA分散液が50.0g入ったビーカーに、上述した式(22)で表されるフッ素系化合物を0.75g添加し混合した。次に、水を0.05g添加し混合した。更に、硝酸を0.005g添加し、以下、合成例1と同様にして二酸化ケイ素(シリカ)粒子の分散液を得た。
<Synthesis Example 4>
0.75 g of the fluorine-based compound represented by the above formula (22) was added and mixed into a beaker containing 50.0 g of the same silicon dioxide IPA dispersion as in Synthesis Example 3. Next, 0.05 g of water was added and mixed. Furthermore, 0.005 g of nitric acid was added, and the same procedure as in Synthesis Example 1 was repeated to obtain a dispersion of silicon dioxide (silica) particles.
<合成例5>
合成例3と同じ二酸化ケイ素のIPA分散液が50.0g入ったビーカーに、上述した式(23)で表されるフッ素系化合物を2.25g添加し混合した。次に、水を0.15g添加し混合した。更に、硝酸を0.005g添加し、以下、合成例1と同様にして二酸化ケイ素(シリカ)粒子の分散液を得た。
<Synthesis Example 5>
2.25 g of the fluorine-based compound represented by the above formula (23) was added and mixed into a beaker containing 50.0 g of the same silicon dioxide IPA dispersion as in Synthesis Example 3. Next, 0.15 g of water was added and mixed. Furthermore, 0.005 g of nitric acid was added, and the same procedure as in Synthesis Example 1 was repeated to obtain a dispersion of silicon dioxide (silica) particles.
<合成例6>
平均粒子径が3nmの二酸化ジルコニウムのメタノール分散液(SZR-M、堺化学社製、ZrO2濃度30%)が50.0g入ったビーカーに、上述した式(27)で表されるフッ素系化合物を8.00g添加し混合した。次に、水を4.05g添加し混合した。更に、硝酸が0.035g添加し、以下、合成例1と同様にして二酸化ジルコニウム粒子の分散液を得た。
<Synthesis Example 6>
In a beaker containing 50.0 g of a methanol dispersion of zirconium dioxide having an average particle size of 3 nm (SZR-M, manufactured by Sakai Chemical Industry Co., Ltd., ZrO2
<合成例7>
平均粒子径が6nmの二酸化チタンのIPA分散液(TKD-701、テイカ社製、TiO2濃度18%)が50.0g入ったビーカーに、上述した式(27)で表されるフッ素系化合物を2.70g添加し混合した。次に、水を0.97g添加し混合した。更に、硝酸が0.010g添加し、以下、合成例1と同様にして二酸化チタン粒子の分散液を得た。
<Synthesis Example 7>
2.70 g of the fluorine-based compound represented by the above formula (27) was added and mixed into a beaker containing 50.0 g of an IPA dispersion of titanium dioxide having an average particle size of 6 nm (TKD-701, manufactured by Teika Corporation, TiO2 concentration 18%). Next, 0.97 g of water was added and mixed. Furthermore, 0.010 g of nitric acid was added, and the same procedure as in Synthesis Example 1 was repeated to obtain a dispersion of titanium dioxide particles.
<合成例8>
平均粒子径が60nmのアルミナと二酸化ケイ素のIPA分散液(バイラールAS-L10、多木化学社製、3Al2O3・2SiO2濃度10%)が50.0g入ったビーカーに、上述した式(27)で表されるフッ素系化合物を0.03g添加し混合した。次に、水0.02gを添加混合した。更に、硝酸0.005g添加し、以下、合成例1と同様にしてアルミナと二酸化ケイ素の粒子の分散液を得た。
<Synthesis Example 8>
0.03 g of the fluorine-based compound represented by the above formula (27) was added and mixed into a beaker containing 50.0 g of an IPA dispersion of alumina and silicon dioxide having an average particle size of 60 nm ( Baylal AS- L10 , manufactured by Taki Chemical Industry Co., Ltd., 3Al2O3.2SiO2 concentration 10%). Next, 0.02 g of water was added and mixed. Furthermore, 0.005 g of nitric acid was added, and the same procedure as in Synthesis Example 1 was repeated to obtain a dispersion of alumina and silicon dioxide particles.
<合成例9>
平均粒子径が25nmの酸化亜鉛のIPA分散液(MZ-500、テイカ社製、ZnO濃度30%)が50.0g入ったビーカーに、上述した式(27)で表されるフッ素系化合物を0.30g添加し混合した。次に、水を0.11g添加し混合した。更に、硝酸を0.005g添加し、以下、合成例1と同様にして酸化亜鉛粒子の分散液を得た。
<Synthesis Example 9>
0.30 g of the fluorine-based compound represented by the above formula (27) was added and mixed into a beaker containing 50.0 g of an IPA dispersion of zinc oxide having an average particle size of 25 nm (MZ-500, manufactured by Teika Corporation,
<比較合成例1>
平均粒子径が230nmの二酸化チタンのIPA分散液(R32、堺化学社製、TiO2濃度30%)が50.0g入ったビーカーに、上述した式(27)で表されるフッ素系化合物を0.75g添加し混合した。次に、水を0.27g添加し混合した。更に、硝酸を0.005g添加し、以下、合成例1と同様にして二酸化チタン粒子の分散液を得た。
Comparative Synthesis Example 1
0.75 g of the fluorine-based compound represented by the above formula (27) was added and mixed into a beaker containing 50.0 g of an IPA dispersion of titanium dioxide having an average particle size of 230 nm (R32, manufactured by Sakai Chemical Industry Co., Ltd.,
<比較合成例2>
合成例1と同じIPA分散液が50.0g入ったビーカーに、フッ素系化合物を全く添加せずに、水を0.10g添加し混合した。更に、硝酸を0.005g添加し、以下、合成例1と同様にして二酸化ケイ素粒子の分散液を得た。
Comparative Synthesis Example 2
To a beaker containing 50.0 g of the same IPA dispersion as in Synthesis Example 1, 0.10 g of water was added and mixed without adding any fluorine-based compound. Furthermore, 0.005 g of nitric acid was added, and the same procedure as in Synthesis Example 1 was repeated to obtain a dispersion of silicon dioxide particles.
以下の表1に、合成例1~9及び比較合成例1のフッ素含有金属酸化物粒子の分散液と比較合成例2のフッ素非含有の金属酸化物粒子の分散液の内容を示す。なお、表1において、フッ素系化合物として式(19)~式(22)及び式(27)で表わされるフッ素含有シランの式中のRはすべてエチル基である。 The following Table 1 shows the contents of the dispersions of fluorine-containing metal oxide particles in Synthesis Examples 1 to 9 and Comparative Synthesis Example 1, and the dispersion of fluorine-free metal oxide particles in Comparative Synthesis Example 2. In Table 1, the R in the formulas of the fluorine-containing silanes represented by formulas (19) to (22) and (27) as fluorine-based compounds is all an ethyl group.
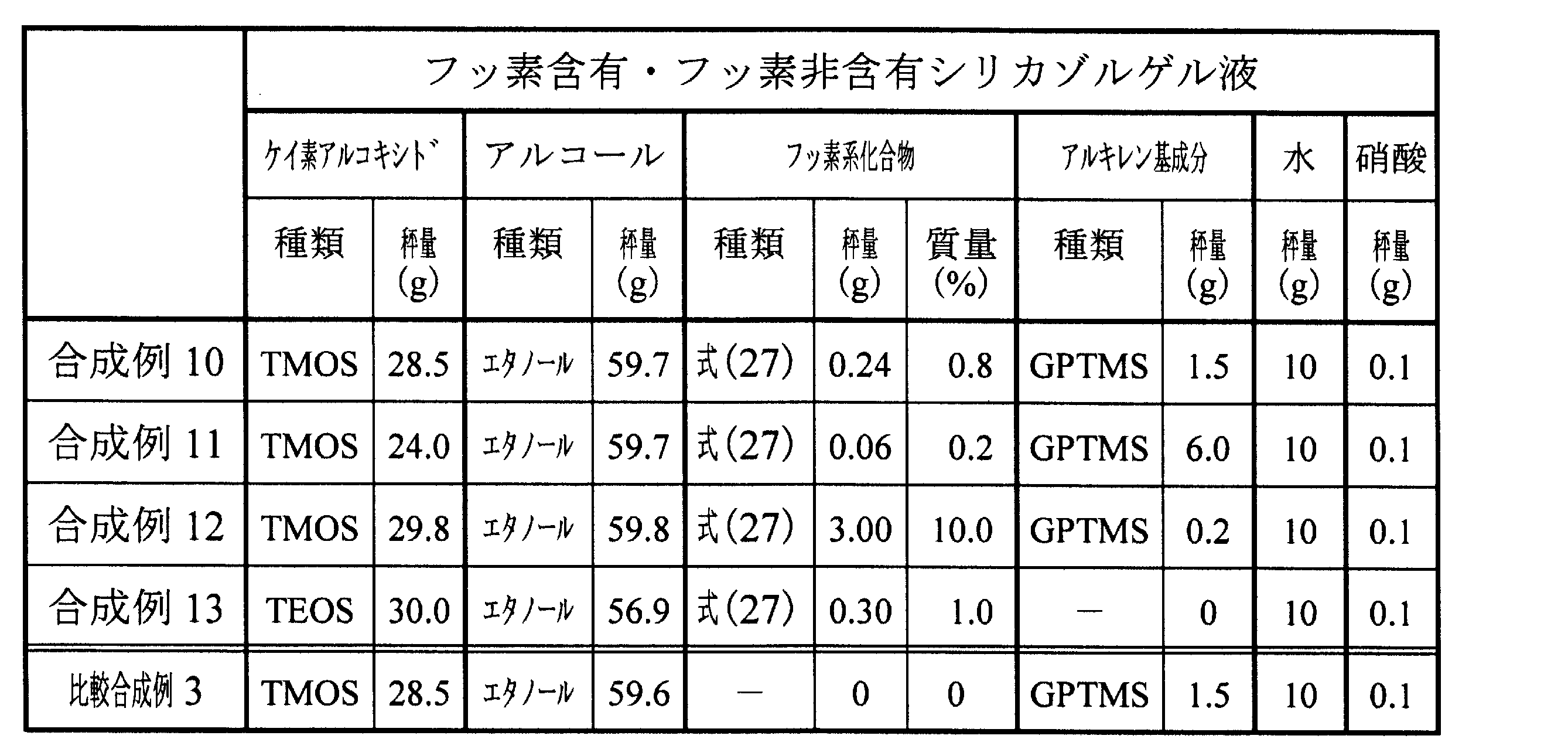
〔フッ素含有シリカゾルゲル液を調製するための合成例10~13、比較合成例3〕
<合成例10>
テトラメトキシシラン(TMOS)の3量体~5量体(三菱化学社製、商品名:MKCシリケートMS51)が28.5gと、エタノールが59.7g入ったビーカーに、上述した式(27)で表されるフッ素系化合物を0.24g(0.8質量%)添加し混合した。次に、アルキレン基成分としてエポキシ基含有シランである3-グリシドキシプロピルトリメトキシシラン(GPTMS:信越化学工業社製、商品名:KBM-403)を1.5gと、水を10g添加し混合した。更に、硝酸を0.1g添加し、30℃で3時間混合し、フッ素含有シリカゾルゲル液を得た。
[Synthesis Examples 10 to 13 for preparing fluorine-containing silica sol-gel liquid, and Comparative Synthesis Example 3]
<Synthesis Example 10>
0.24 g (0.8 mass%) of the fluorine-based compound represented by the above formula (27) was added to a beaker containing 28.5 g of trimer to pentamer of tetramethoxysilane (TMOS) (manufactured by Mitsubishi Chemical Corporation, product name: MKC Silicate MS51) and 59.7 g of ethanol, and mixed. Next, 1.5 g of 3-glycidoxypropyltrimethoxysilane (GPTMS: manufactured by Shin-Etsu Chemical Co., Ltd., product name: KBM-403), which is an epoxy group-containing silane as an alkylene group component, and 10 g of water were added and mixed. Furthermore, 0.1 g of nitric acid was added, and the mixture was mixed at 30° C. for 3 hours to obtain a fluorine-containing silica sol-gel liquid.
<合成例11>
合成例10と同一のTMOSの3量体~5量体が24.0gと、エタノールが59.7gが入ったビーカーに、上述した式(27)で表されるフッ素系化合物を0.06g(0.2質量%)添加し混合した。次に、アルキレン基成分として合成例10と同一のGPTMSを6.0gと、水を10g添加し混合した。更に、硝酸を0.1g添加し、30℃で3時間混合し、フッ素含有シリカゾルゲル液を得た。
<Synthesis Example 11>
To a beaker containing 24.0 g of the same TMOS trimer to pentamer as in Synthesis Example 10 and 59.7 g of ethanol, 0.06 g (0.2 mass%) of the fluorine-based compound represented by the above formula (27) was added and mixed. Next, 6.0 g of the same GPTMS as in Synthesis Example 10 as an alkylene group component and 10 g of water were added and mixed. Furthermore, 0.1 g of nitric acid was added and mixed at 30° C. for 3 hours to obtain a fluorine-containing silica sol-gel liquid.
<合成例12>
合成例10と同一のTMOSの3量体~5量体が29.8gと、エタノールが59.8gが入ったビーカーに、上述した式(27)で表されるフッ素系化合物を3.00g(10.0質量%)添加し混合した。次に、アルキレン基成分として合成例10と同一のGPTMSを0.2gと、水を10g添加し混合した。更に、硝酸を0.1g添加し、30℃で3時間混合し、フッ素含有シリカゾルゲル液を得た。
<Synthesis Example 12>
To a beaker containing 29.8 g of the same TMOS trimer to pentamer as in Synthesis Example 10 and 59.8 g of ethanol, 3.00 g (10.0 mass%) of the fluorine-based compound represented by the above formula (27) was added and mixed. Next, 0.2 g of the same GPTMS as in Synthesis Example 10 as an alkylene group component and 10 g of water were added and mixed. Furthermore, 0.1 g of nitric acid was added and mixed at 30° C. for 3 hours to obtain a fluorine-containing silica sol-gel liquid.
<合成例13>
テトラエトキシシラン(TEOS)が30.0gと、エタノールが56.9gが入ったビーカーに、上述した式(27)で表されるフッ素系化合物を0.30g(1.0質量%)添加し混合した。次に、アルキレン基成分を添加することなく、水を10g添加し混合した。更に、硝酸を0.1g添加し、30℃で3時間混合し、フッ素含有シリカゾルゲル液を得た。
<Synthesis Example 13>
0.30 g (1.0 mass%) of the fluorine-based compound represented by the above formula (27) was added to a beaker containing 30.0 g of tetraethoxysilane (TEOS) and 56.9 g of ethanol, and mixed. Next, 10 g of water was added without adding any alkylene group component, and mixed. Furthermore, 0.1 g of nitric acid was added, and mixed at 30° C. for 3 hours to obtain a fluorine-containing silica sol-gel liquid.
<比較合成例3>
合成例11と同一のTMOSの3量体~5量体が28.5gと、エタノールが59.6gが入ったビーカーに、フッ素系化合物を全く添加せずに、アルキレン基成分として合成例10と同一のGPTMSを1.5gと、水を10g添加し混合した。更に、硝酸を0.1g添加し、30℃で3時間混合し、フッ素を含有しないシリカゾルゲル液を得た。
Comparative Synthesis Example 3
To a beaker containing 28.5 g of the same TMOS trimer to pentamer as in Synthesis Example 11 and 59.6 g of ethanol, 1.5 g of the same GPTMS as in Synthesis Example 10 as an alkylene group component and 10 g of water were added and mixed without adding any fluorine-based compound. Furthermore, 0.1 g of nitric acid was added and mixed at 30° C. for 3 hours to obtain a fluorine-free silica sol-gel liquid.
以下の表2に、合成例10~13のフッ素含有シリカゾルゲル液と比較合成例3のフッ素非含有のシリカゾルゲル液の内容を示す。 Table 2 below shows the contents of the fluorine-containing silica sol-gel liquids of Synthesis Examples 10 to 13 and the fluorine-free silica sol-gel liquid of Comparative Synthesis Example 3.
〔撥水撥油性膜形成用液組成物の製造のための実施例1~9、比較例1~7〕
<実施例1>
合成例10で得られたフッ素含有シリカゾルゲル液1.9gに溶媒26.0gを添加し混合した。溶媒は、工業アルコール(AP-7、日本アルコール産業社製)22.0gに、ジアセトンアルコール1.0gと、イソプロピルグリコール3.0gとを混合して調製された。その後、合成例1の金属酸化物粒子の分散液1.5gを添加し混合し、撥水撥油性膜形成用液組成物を調製した。この内容を以下の表3に示す。
[Examples 1 to 9 and Comparative Examples 1 to 7 for the production of water- and oil-repellent film-forming liquid compositions]
Example 1
26.0 g of a solvent was added to 1.9 g of the fluorine-containing silica sol-gel liquid obtained in Synthesis Example 10 and mixed. The solvent was prepared by mixing 1.0 g of diacetone alcohol and 3.0 g of isopropyl glycol with 22.0 g of industrial alcohol (AP-7, manufactured by Japan Alcohol Industry Co., Ltd.). Then, 1.5 g of the dispersion of metal oxide particles of Synthesis Example 1 was added and mixed to prepare a water- and oil-repellent film-forming liquid composition. The details are shown in Table 3 below.
<実施例2~9及び比較例1~7>
実施例2~9及び比較例1~7について、表3に示すように、フッ素含有金属酸化物粒子の分散液の種類と秤量、フッ素含有シリカゾルゲル液の種類と秤量、及び実施例1と同一の溶媒の秤量をそれぞれ決定して、実施例2~9及び比較例1~7の各撥水撥油性膜形成用液組成物を調製した。
<Examples 2 to 9 and Comparative Examples 1 to 7>
For Examples 2 to 9 and Comparative Examples 1 to 7, the type and weight of the dispersion liquid of the fluorine-containing metal oxide particles, the type and weight of the fluorine-containing silica sol-gel liquid, and the weight of the same solvent as in Example 1 were determined as shown in Table 3, and the water- and oil-repellent film-forming liquid compositions of Examples 2 to 9 and Comparative Examples 1 to 7 were prepared.
<比較試験及び評価>
実施例1~9及び比較例1~7で得られた16種類の液組成物を、刷毛(末松刷子製ナイロン刷毛マイスター)を用いて、厚さ2mm、たて150mm、よこ75mmのSUS304基材上にそれぞれ乾燥後の厚さが1~3μmとなるように塗布し、16種類の塗膜を形成した。すべての塗膜を室温の大気雰囲気中にて3時間静置し、塗膜を乾燥させて上記SUS304基材上に16種類の膜を得た。これらの膜について、膜表面の水濡れ性(撥水性)、撥油性、n-ヘキサデカンの転落性及び膜の外観を評価し、膜の強度試験、膜のセロテープ(登録商標)剥離試験及び耐指紋性試験を行った。これらの結果を表4に示す。なお、膜の耐指紋性とは、膜に指紋付着後に指紋が膜表面に目立ちにくく、また付着した場合の指紋の拭き取りが容易であることに加えて、指紋が膜に付着しにくい特性である。
<Comparative testing and evaluation>
The 16 types of liquid compositions obtained in Examples 1 to 9 and Comparative Examples 1 to 7 were applied to a SUS304 substrate having a thickness of 2 mm, a length of 150 mm, and a width of 75 mm using a brush (Nylon Brush Meister manufactured by Suematsu Brush Co., Ltd.) so that the thickness after drying was 1 to 3 μm, and 16 types of coating films were formed. All coating films were left to stand in an atmospheric atmosphere at room temperature for 3 hours, and the coating films were dried to obtain 16 types of films on the SUS304 substrate. For these films, the water wettability (water repellency), oil repellency, n-hexadecane sliding property, and appearance of the film were evaluated, and a film strength test, a cellophane tape peeling test, and a fingerprint resistance test were performed. The results are shown in Table 4. The fingerprint resistance of the film is a property in which fingerprints are not easily noticeable on the film surface after they are attached to the film, and fingerprints are easily wiped off when they are attached, and fingerprints are not easily attached to the film.
(1) 膜表面の撥水性(接触角)
協和界面科学製ドロップマスターDM-700を用いて、シリンジに22℃±1℃のイオン交換水を準備し、シリンジの針の先端から2μLの液滴を飛び出した状態にする。次いで評価するSUS304基材上の膜をこの液滴に近づけて膜に液滴を付着させる。この付着した水の接触角を測定した。静止状態で水が膜表面に触れた1秒後の接触角をθ/2法により解析した値を水の接触角とし、膜表面の水濡れ性(撥水性)を評価した。84度以上であれば『良好』とした。
(1) Water repellency of the film surface (contact angle)
Using a Dropmaster DM-700 manufactured by Kyowa Interface Science, ion-exchanged water at 22°C ± 1°C was prepared in a syringe, and a 2μL droplet was ejected from the tip of the needle of the syringe. The film on the SUS304 substrate to be evaluated was then brought close to the droplet, and the droplet was attached to the film. The contact angle of the attached water was measured. The contact angle after 1 second when the water touched the film surface in a stationary state was analyzed by the θ/2 method, and the water wettability (water repellency) of the film surface was evaluated. If it was 84 degrees or more, it was rated as "good".
(2) 膜表面の撥油性(接触角)
協和界面科学製ドロップマスターDM-700を用いて、シリンジに22℃±1℃のn-ヘキサデカンを準備し、シリンジの針の先端から2μLの液滴を飛び出した状態にする。次いで評価するSUS304基材上の膜をこの液滴に近づけて膜に液滴を付着させる。この付着したn-ヘキサデカンの接触角を測定した。静止状態でn-ヘキサデカンが膜表面に触れた1秒後の接触角をθ/2法により解析した値をn-ヘキサデカンの接触角とし、膜表面の撥油性を評価した。42度以上であれば『良好』とした。
(2) Oil repellency of the film surface (contact angle)
Using a Dropmaster DM-700 manufactured by Kyowa Interface Science, n-hexadecane at 22°C±1°C was prepared in a syringe, and a 2μL droplet was ejected from the tip of the needle of the syringe. Next, the film on the SUS304 substrate to be evaluated was brought close to the droplet, and the droplet was attached to the film. The contact angle of the attached n-hexadecane was measured. The
(3) n-ヘキサデカンの転落性試験
協和界面科学製ドロップマスターDM-700を用いて、シリンジに25±1℃のn-ヘキサデカンを準備し、水平に置いたポリプロピレン基材上にシリンジからn-ヘキサデカンを9μLの液滴を滴下し、基材を2度/分の速度で傾斜させ、n-ヘキサデカンの液滴が移動開始するときの基材の傾けた角度を測定した。(2)の接触角が低くてもこの転落角度が小さい方が防汚性に優れていることを意味する。50度以下であれば『良好』とした。またn-ヘキサデカンの液滴が移動して基材から転落した後で、基材表面に液滴の痕跡が有る場合を『油痕有り』とした。
(3) n-Hexadecane sliding test Using a Kyowa Interface Science Dropmaster DM-700, n-hexadecane at 25±1°C was prepared in a syringe, and 9 μL of n-hexadecane was dropped from the syringe onto a horizontally placed polypropylene substrate. The substrate was tilted at a rate of 2 degrees/min, and the tilt angle of the substrate was measured when the n-hexadecane droplet began to move. Even if the contact angle in (2) is low, a smaller sliding angle means better antifouling properties. If it was 50 degrees or less, it was rated as "good." In addition, if there was a trace of the droplet on the surface of the substrate after the n-hexadecane droplet moved and fell off the substrate, it was rated as "oil stains present."
(4) 膜の外観
評価するSUS304基材上の膜を目視で観察して、膜が透明であるか否か、また膜が白濁しているか否かを調べた。膜が透明であるものを『良好』とし、膜が白濁しているものを『白濁不良』とした。
(4) Appearance of the film The film on the SUS304 substrate to be evaluated was visually observed to check whether the film was transparent or not, and whether the film was cloudy or not. A transparent film was rated as "good", and a cloudy film was rated as "bad cloudiness".
(5) 膜の強度試験
評価するSUS304基材上の膜に下記の接触子を所定の荷重をかけながら、次の条件で10往復移動した後で、基材上の膜が基材から剥離しないか否かを目視で調べた。膜が剥離しない場合を『合格』とし、剥離した場合を『不合格』とした。
(a) 測定器:静・動摩擦測定機TL201Tt(株式会社トリニティーラボ)
(b) 測定条件:
・移動距離:30mm
・垂直荷重:500g重
・移動速度:50mm/秒
・接触子:5mm×15mm角のネオプレーンゴム
(5) Film strength test The contactor described below was applied with a specified load to the film on the SUS304 substrate to be evaluated, and after 10 reciprocating movements under the following conditions, the film on the substrate was visually inspected for peeling from the substrate. If the film did not peel off, it was judged as "passed", and if it peeled off, it was judged as "failed".
(a) Measuring instrument: Static and dynamic friction measuring instrument TL201Tt (Trinity Lab Co., Ltd.)
(b) Measurement conditions:
・Traveling distance: 30mm
Vertical load: 500 g; Movement speed: 50 mm/sec.; Contact: 5 mm x 15 mm square neoprene rubber
(6) 膜のセロテープ(登録商標)剥離試験
評価するSUS304基材上の膜に碁盤目状に1mm幅のクロスカットを施し、その碁盤目状にクロスカットされた膜に粘着テープ(ニチバン社製、商品名「セロテープ(登録商標)」)を貼り、JISK5600-5-6(クロスカット法)の碁盤目テープ法に準拠してセロテープ(登録商標)剥離試験(以下、単に剥離試験という。)を行った。クロスカットを施したマス目100個を分母で表し、剥離試験後に基材上に残存するマス目の数を分子で表した。
(6) Cellotape (registered trademark) peeling test of
(6) 膜の耐指紋性試験
セルロース連続長繊維不織布(旭化成社製、製品名:ベンコットM-3II)を評価するSUS304基材上の膜の上に載せ、この不織布に人工指紋液(JIS K 2246に準じる)を100マイクロリットル滴下した。この不織布に黒ゴム24号(質量300g)を載せ、更に1kgの重りを載せて、30秒間静置することにより、膜に指紋液を付着させた。その後、不織布を剥がして、膜の耐指紋性を目視にて観察した。膜表面に指紋液が付着していなかったものを『良好』とし、付着しているものを『不良』とした。
(6) Fingerprint resistance test of film A continuous cellulose fiber nonwoven fabric (manufactured by Asahi Kasei Corporation, product name: BEMCOT M-3II) was placed on the film on the SUS304 substrate to be evaluated, and 100 microliters of artificial fingerprint liquid (based on JIS K 2246) was dropped onto the nonwoven fabric. Black rubber No. 24 (mass 300 g) was placed on the nonwoven fabric, and a weight of 1 kg was placed on top. The film was left to stand for 30 seconds to allow the fingerprint liquid to adhere to the film. The nonwoven fabric was then peeled off, and the fingerprint resistance of the film was visually observed. Films that had no fingerprint liquid on the surface were rated as "good", and films that had fingerprint liquid on the surface were rated as "poor".
表4から明らかなように、比較例1では、平均粒子径が230nmである金属酸化物(二酸化チタン)粒子を含む比較合成例1の二酸化チタン粒子の分散液から撥水撥油性膜形成用液組成物を調製し、この液組成物により膜を形成したため、膜中の金属酸化物粒子の平均粒子径が大き過ぎ、バインダ成分であるシリカゾルゲルで金属酸化物粒子が基材表面に結着しにくかった。この結果、水及びn-ヘキサデカンの接触角は悪く、膜が白濁していて、膜の外観が不良であった。また膜の強度試験では膜が基材から剥離し、剥離試験では基材上に残存するマス目の数は20個しかなく、大部分が剥離した。更に膜の耐指紋性は不良であった。 As is clear from Table 4, in Comparative Example 1, a liquid composition for forming a water- and oil-repellent film was prepared from a dispersion of titanium dioxide particles in Comparative Synthesis Example 1, which contained metal oxide (titanium dioxide) particles with an average particle diameter of 230 nm, and a film was formed using this liquid composition. As a result, the average particle diameter of the metal oxide particles in the film was too large, and the metal oxide particles were difficult to bind to the substrate surface with the silica sol-gel binder component. As a result, the contact angles of water and n-hexadecane were poor, the film was cloudy, and the appearance of the film was poor. In addition, in the strength test of the film, the film peeled off from the substrate, and in the peel test, only 20 squares remained on the substrate, with most peeling off. Furthermore, the fingerprint resistance of the film was poor.
比較例2では、フッ素系化合物を含まない比較合成例3のシリカゾルゲル液から撥水撥油性膜形成用液組成物を調製し、この液組成物により膜を形成したため、n-ヘキサデカンの転落角が悪かった。 In Comparative Example 2, a liquid composition for forming a water- and oil-repellent film was prepared from the silica sol-gel liquid of Comparative Synthesis Example 3, which does not contain a fluorine-based compound, and a film was formed using this liquid composition, resulting in a poor sliding angle of n-hexadecane.
比較例3では、シリカゾルゲル(C)と金属酸化物粒子(B)を合計した量を100質量%とするとき、シリカゾルゲル(C)が5質量%であったため、液組成物中のバインダ成分が少なくなり過ぎて、膜の剥離試験では基材上に残存するマス目の数は10個しかなく、大部分が剥離した。またn-ヘキサデカンの転落角が悪く、『油痕有り』であった。また膜が白濁していて、膜の外観が不良であった。更に膜の耐指紋性は不良であった。 In Comparative Example 3, when the total amount of silica sol-gel (C) and metal oxide particles (B) is taken as 100% by mass, the amount of silica sol-gel (C) was 5% by mass, so the binder component in the liquid composition was too little, and in the film peeling test, only 10 squares remained on the substrate, with most peeling off. In addition, the n-hexadecane had a poor sliding angle and was "oil stained." The film was also cloudy and had a poor appearance. Furthermore, the film had poor fingerprint resistance.
比較例4では、シリカゾルゲル(C)と金属酸化物粒子(B)を合計した量を100質量%とするとき、金属酸化物粒子(B)が5質量%であったため、液組成物中の金属酸化物粒子が少なくなり過ぎて、膜の強度試験では基材上から膜が剥離した。更に膜の耐指紋性は不良であった。 In Comparative Example 4, when the total amount of the silica sol-gel (C) and the metal oxide particles (B) was taken as 100 mass %, the metal oxide particles (B) were 5 mass %, so the amount of metal oxide particles in the liquid composition was too small, and the film peeled off from the substrate in the film strength test. Furthermore, the fingerprint resistance of the film was poor.
比較例5では、フッ素系官能基成分(A1)とフッ素系官能基成分(A2)の合計した含有割合(A1+A2)が0.8質量%であったため、形成した膜に撥油性を付与できず、水及びn-ヘキサデカンの接触角は悪く、n-ヘキサデカンの転落角試験では『油痕有り』であった。更に膜の耐指紋性は不良であった。 In Comparative Example 5, the combined content (A1+A2) of the fluorine-based functional group component (A1) and the fluorine-based functional group component (A2) was 0.8% by mass, so the formed film could not be imparted with oil repellency, the contact angles of water and n-hexadecane were poor, and the n-hexadecane sliding angle test showed "oil stains." Furthermore, the film had poor fingerprint resistance.
比較例6では、フッ素系官能基成分(A1)とフッ素系官能基成分(A2)の合計した含有割合(A1+A2)が33.4質量%であったため、形成した膜に撥油性を付与できず、n-ヘキサデカンの転落角試験では70度であった。また膜が白濁していて、膜の外観が不良であった。更に膜の耐指紋性は不良であった。 In Comparative Example 6, the combined content (A1+A2) of the fluorine-based functional group component (A1) and the fluorine-based functional group component (A2) was 33.4% by mass, so the formed film could not be imparted with oil repellency, and the n-hexadecane sliding angle test showed a value of 70 degrees. In addition, the film was cloudy and had a poor appearance. Furthermore, the film had poor fingerprint resistance.
比較例7では、フッ素系化合物を含まない比較合成例2の金属酸化物粒子の分散液から撥水撥油性膜形成用液組成物を調製し、この液組成物により膜を形成したため、水及びn-ヘキサデカンの接触角は悪く、n-ヘキサデカンの転落角試験では『油痕有り』であった。更に膜の耐指紋性は不良であった。 In Comparative Example 7, a liquid composition for forming a water- and oil-repellent film was prepared from a dispersion of metal oxide particles of Comparative Synthesis Example 2 that did not contain a fluorine-based compound, and a film was formed using this liquid composition. As a result, the contact angles of water and n-hexadecane were poor, and the n-hexadecane sliding angle test showed "oil stains." Furthermore, the fingerprint resistance of the film was poor.
それに対して、実施例1~9では、第1及び第2フッ素系官能基成分(A1、A2)が上述した式(19)~式(23)及び式(27)であり、金属酸化物粒子の平均粒子径が2nm~90nmの範囲にあり、溶媒(D)を除く液組成物中の第1及び第2フッ素系官能基成分(A1、A2)を合計した含有割合(A1+A2)が1質量%~30質量%であり、シリカゾルゲル(C)と金属酸化物粒子(B)の質量比(C:B)が10:90~90:10の範囲にある、第1の観点の発明の範囲を満たしていることから、水及びn-ヘキサデカンの接触角、n-ヘキサデカンの転落性及び膜の外観は、いずれも良好であり、膜の強度試験、剥離試験及び耐指紋性試験はいずれも合格していた。 In contrast, in Examples 1 to 9, the first and second fluorine-based functional group components (A1, A2) are represented by the above formulas (19) to (23) and (27), the average particle size of the metal oxide particles is in the range of 2 nm to 90 nm, the total content (A1+A2) of the first and second fluorine-based functional group components (A1, A2) in the liquid composition excluding the solvent (D) is 1% by mass to 30% by mass, and the mass ratio (C:B) of the silica sol-gel (C) to the metal oxide particles (B) is in the range of 10:90 to 90:10, which satisfies the scope of the invention of the first aspect. Therefore, the contact angles of water and n-hexadecane, the sliding properties of n-hexadecane, and the appearance of the film are all good, and the film strength test, peel test, and fingerprint resistance test all passed.
本発明の撥水撥油性膜形成用液組成物は、機械油を使用する工場、油が飛散する厨房、油蒸気が立ちこめるレンジフード、換気扇、冷蔵庫扉等において、油汚れを防止する分野に用いられる。 The water- and oil-repellent film-forming liquid composition of the present invention is used in fields where oil stains need to be prevented, such as in factories where machine oil is used, in kitchens where oil is scattered, and on range hoods, ventilation fans, refrigerator doors, and other areas where oil vapor is present.
1 基材
2 撥水撥油性膜
3 金属酸化物粒子
4 シリカゾルゲル
1
Claims (9)
下記の式(19)から式(27)のいずれか1つで示される、ペルフルオロエーテル構造を含む第2フッ素系化合物(A2)を含有するフッ素含有シリカゾルゲル(C)と、
溶媒(D)とを含み、
前記溶媒(D)を除く全成分量を100質量%としたとき、前記第1フッ素化合物(A1)と前記第2フッ素化合物(A2)とを合計した含有割合が、1質量%~30質量%であり、
前記フッ素含有シリカゾルゲル(C)と前記フッ素含有金属酸化物粒子(B)の質量比(C:B)が、10:90~90:10の範囲にあることを特徴とする撥水撥油性膜形成用液組成物。
A fluorine-containing silica sol-gel (C) containing a second fluorine-based compound (A2) having a perfluoroether structure represented by any one of the following formulas (19) to (27) :
A solvent (D),
the total content of the first fluorine compound (A1) and the second fluorine compound (A2) is 1% by mass to 30% by mass when the total amount of all components excluding the solvent (D) is 100% by mass,
A water- and oil-repellent film-forming liquid composition, characterized in that a mass ratio (C:B) of the fluorine-containing silica sol-gel (C) to the fluorine-containing metal oxide particles (B) is in the range of 10:90 to 90:10.
フッ素含有金属酸化物粒子(B)の分散液とフッ素含有シリカゾルゲル液(C)とを混合して調製される撥水撥油性膜形成用液組成物の製造方法。 A method for producing the water- and oil-repellent film-forming liquid composition according to claim 1, comprising the steps of:
A method for producing a water- and oil-repellent film-forming liquid composition, which is prepared by mixing a dispersion of fluorine-containing metal oxide particles (B) and a fluorine-containing silica sol-gel liquid (C) .
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020120307A JP7577472B2 (en) | 2020-07-14 | 2020-07-14 | Water- and oil-repellent film-forming liquid composition and method for producing same |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020120307A JP7577472B2 (en) | 2020-07-14 | 2020-07-14 | Water- and oil-repellent film-forming liquid composition and method for producing same |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2022017640A JP2022017640A (en) | 2022-01-26 |
| JP7577472B2 true JP7577472B2 (en) | 2024-11-05 |
Family
ID=80185935
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2020120307A Active JP7577472B2 (en) | 2020-07-14 | 2020-07-14 | Water- and oil-repellent film-forming liquid composition and method for producing same |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP7577472B2 (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023153135A1 (en) | 2022-02-08 | 2023-08-17 | 日本碍子株式会社 | Membrane reactor and method for operating membrane reactor |
| TW202438636A (en) * | 2022-11-18 | 2024-10-01 | 日商三菱綜合材料股份有限公司 | Hydrophobic film, hydrophobic electrostatic adsorption sheet, electrostatic adsorption support member, and method for producing hydrophobic electrostatic adsorption sheet |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009154480A (en) | 2007-12-27 | 2009-07-16 | Nof Corp | Water repellent film |
| JP2017171701A (en) | 2016-03-18 | 2017-09-28 | 三菱マテリアル株式会社 | Composition for film formation and method for producing the same, and film |
| WO2019026816A1 (en) | 2017-07-31 | 2019-02-07 | 東洋アルミニウム株式会社 | COMPOSITE PARTICLES CONTAINING FLUORINE |
| JP2020083836A (en) | 2018-11-28 | 2020-06-04 | 三菱マテリアル電子化成株式会社 | Perfluoroether-containing silane compound |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1192177A (en) * | 1997-09-18 | 1999-04-06 | Sony Corp | Water repellent glass and method for producing the same |
-
2020
- 2020-07-14 JP JP2020120307A patent/JP7577472B2/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009154480A (en) | 2007-12-27 | 2009-07-16 | Nof Corp | Water repellent film |
| JP2017171701A (en) | 2016-03-18 | 2017-09-28 | 三菱マテリアル株式会社 | Composition for film formation and method for producing the same, and film |
| WO2019026816A1 (en) | 2017-07-31 | 2019-02-07 | 東洋アルミニウム株式会社 | COMPOSITE PARTICLES CONTAINING FLUORINE |
| JP2020083836A (en) | 2018-11-28 | 2020-06-04 | 三菱マテリアル電子化成株式会社 | Perfluoroether-containing silane compound |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2022017640A (en) | 2022-01-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10889725B2 (en) | Sulfonate-functional coatings and methods | |
| JP5716679B2 (en) | Water-repellent substrate and method for producing the same | |
| JPWO2008072707A1 (en) | Articles having a water repellent surface | |
| WO2010007956A1 (en) | Water-repellent substrate and process for production thereof | |
| WO2018092543A1 (en) | Laminate, method for producing same and composition for anti-fog coating | |
| WO2008038782A1 (en) | Vapor deposition material, process for producing optical member or plastic lens for spectacle with use thereof, and plastic lens for spectacle | |
| WO2017159854A1 (en) | Coating-forming composition, method for producing same, and coating | |
| JP7577472B2 (en) | Water- and oil-repellent film-forming liquid composition and method for producing same | |
| JP4502112B2 (en) | Antifouling coating agent and coated article | |
| JP2009154480A (en) | Water repellent film | |
| JP7596074B2 (en) | Method for producing water- and oil-repellent film-forming liquid composition | |
| JP3576580B2 (en) | Water repellent glass | |
| JP6060698B2 (en) | Curable resin composition and coated article | |
| JP2021172768A (en) | Liquid composition for forming water- and oil-repellent film and method for producing the same | |
| JP7574037B2 (en) | Antifouling film-forming liquid composition | |
| JP2008119924A (en) | Articles having a water repellent surface | |
| JP2011006653A (en) | Substrate with cured film | |
| JP7705316B2 (en) | Water- and oil-repellent black film-forming liquid composition and method for producing same | |
| JP7574022B2 (en) | Antifouling film-forming liquid composition | |
| JP2023038692A (en) | Liquid composition for forming water- and oil-repellent black film and method for producing the same | |
| JP7177889B2 (en) | Liquid composition for film formation | |
| JP4725073B2 (en) | Coating material composition and painted product | |
| JP2009029882A (en) | Water / oil repellent resin composition, water / oil repellent coating and member | |
| Farhadyar et al. | Synthesis and characterization of ZnO-SiO2/epoxy nanocomposite coating by sol-gel process | |
| JP2023031897A (en) | Liquid composition for forming water and oil repellent black film and production method therefor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7423 Effective date: 20220427 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20230517 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20240214 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20240305 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20240430 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20240704 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20241008 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20241023 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7577472 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |