JP6814044B2 - 薬剤溶出性バルーンを有する画像ガイドされる治療カテーテル - Google Patents
薬剤溶出性バルーンを有する画像ガイドされる治療カテーテル Download PDFInfo
- Publication number
- JP6814044B2 JP6814044B2 JP2016570826A JP2016570826A JP6814044B2 JP 6814044 B2 JP6814044 B2 JP 6814044B2 JP 2016570826 A JP2016570826 A JP 2016570826A JP 2016570826 A JP2016570826 A JP 2016570826A JP 6814044 B2 JP6814044 B2 JP 6814044B2
- Authority
- JP
- Japan
- Prior art keywords
- deb
- assembly
- catheter
- drug
- balloon
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 230000001225 therapeutic effect Effects 0.000 title description 4
- 238000003384 imaging method Methods 0.000 claims description 83
- 238000000576 coating method Methods 0.000 claims description 68
- 239000011248 coating agent Substances 0.000 claims description 62
- 229940079593 drug Drugs 0.000 claims description 47
- 239000003814 drug Substances 0.000 claims description 47
- 238000010828 elution Methods 0.000 claims description 37
- 238000011282 treatment Methods 0.000 claims description 25
- 239000003550 marker Substances 0.000 claims description 6
- 238000012014 optical coherence tomography Methods 0.000 claims description 4
- 230000003902 lesion Effects 0.000 description 59
- 210000004204 blood vessel Anatomy 0.000 description 41
- 238000000034 method Methods 0.000 description 19
- 239000012530 fluid Substances 0.000 description 15
- 238000002608 intravascular ultrasound Methods 0.000 description 11
- 238000003780 insertion Methods 0.000 description 8
- 230000037431 insertion Effects 0.000 description 8
- 239000000463 material Substances 0.000 description 8
- 239000004033 plastic Substances 0.000 description 7
- 229920003023 plastic Polymers 0.000 description 7
- 238000012545 processing Methods 0.000 description 7
- 210000001367 artery Anatomy 0.000 description 6
- 230000002792 vascular Effects 0.000 description 6
- 230000008901 benefit Effects 0.000 description 5
- 229920000642 polymer Polymers 0.000 description 5
- 239000004698 Polyethylene Substances 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- 230000009977 dual effect Effects 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 210000005036 nerve Anatomy 0.000 description 4
- 238000012384 transportation and delivery Methods 0.000 description 4
- 238000002604 ultrasonography Methods 0.000 description 4
- 230000017531 blood circulation Effects 0.000 description 3
- 239000004020 conductor Substances 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- 230000010339 dilation Effects 0.000 description 3
- 239000013307 optical fiber Substances 0.000 description 3
- -1 polyethylene Polymers 0.000 description 3
- 229920000573 polyethylene Polymers 0.000 description 3
- 229930012538 Paclitaxel Natural products 0.000 description 2
- 208000031481 Pathologic Constriction Diseases 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 230000006835 compression Effects 0.000 description 2
- 238000007906 compression Methods 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 238000002594 fluoroscopy Methods 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 238000007429 general method Methods 0.000 description 2
- 208000014674 injury Diseases 0.000 description 2
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 2
- 239000011976 maleic acid Substances 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 229960001592 paclitaxel Drugs 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 230000036262 stenosis Effects 0.000 description 2
- 208000037804 stenosis Diseases 0.000 description 2
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 230000008733 trauma Effects 0.000 description 2
- 208000031104 Arterial Occlusive disease Diseases 0.000 description 1
- 201000000057 Coronary Stenosis Diseases 0.000 description 1
- 206010011089 Coronary artery stenosis Diseases 0.000 description 1
- HKVAMNSJSFKALM-GKUWKFKPSA-N Everolimus Chemical compound C1C[C@@H](OCCO)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 HKVAMNSJSFKALM-GKUWKFKPSA-N 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 238000002583 angiography Methods 0.000 description 1
- 230000001028 anti-proliverative effect Effects 0.000 description 1
- 208000021328 arterial occlusion Diseases 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 238000002591 computed tomography Methods 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 230000002638 denervation Effects 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000002592 echocardiography Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 229960005167 everolimus Drugs 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 229920005570 flexible polymer Polymers 0.000 description 1
- 230000014509 gene expression Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000010687 lubricating oil Substances 0.000 description 1
- 238000012806 monitoring device Methods 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000001902 propagating effect Effects 0.000 description 1
- 238000005086 pumping Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000008660 renal denervation Effects 0.000 description 1
- 208000037803 restenosis Diseases 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 230000003595 spectral effect Effects 0.000 description 1
- 230000002966 stenotic effect Effects 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 230000026676 system process Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 230000006496 vascular abnormality Effects 0.000 description 1
- 239000011800 void material Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/0059—Measuring for diagnostic purposes; Identification of persons using light, e.g. diagnosis by transillumination, diascopy, fluorescence
- A61B5/0062—Arrangements for scanning
- A61B5/0066—Optical coherence imaging
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/0059—Measuring for diagnostic purposes; Identification of persons using light, e.g. diagnosis by transillumination, diascopy, fluorescence
- A61B5/0082—Measuring for diagnostic purposes; Identification of persons using light, e.g. diagnosis by transillumination, diascopy, fluorescence adapted for particular medical purposes
- A61B5/0084—Measuring for diagnostic purposes; Identification of persons using light, e.g. diagnosis by transillumination, diascopy, fluorescence adapted for particular medical purposes for introduction into the body, e.g. by catheters
- A61B5/0086—Measuring for diagnostic purposes; Identification of persons using light, e.g. diagnosis by transillumination, diascopy, fluorescence adapted for particular medical purposes for introduction into the body, e.g. by catheters using infrared radiation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B8/00—Diagnosis using ultrasonic, sonic or infrasonic waves
- A61B8/08—Detecting organic movements or changes, e.g. tumours, cysts, swellings
- A61B8/0891—Detecting organic movements or changes, e.g. tumours, cysts, swellings for diagnosis of blood vessels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B8/00—Diagnosis using ultrasonic, sonic or infrasonic waves
- A61B8/12—Diagnosis using ultrasonic, sonic or infrasonic waves in body cavities or body tracts, e.g. by using catheters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B8/00—Diagnosis using ultrasonic, sonic or infrasonic waves
- A61B8/13—Tomography
- A61B8/14—Echo-tomography
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M2025/1043—Balloon catheters with special features or adapted for special applications
- A61M2025/105—Balloon catheters with special features or adapted for special applications having a balloon suitable for drug delivery, e.g. by using holes for delivery, drug coating or membranes
Description
Claims (6)
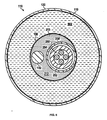
- 一体型治療及びイメージングカテーテルであって、
ガイドワイヤルーメンを規定する内側部材と、
前記内側部材上に位置付けられるバルーンアセンブリと、
前記バルーンアセンブリ上に位置付けられる薬剤溶出コーティングと、
前記内側部材上に位置付けられるイメージング装置と、
を有し、
前記イメージング装置及び前記バルーンアセンブリは、互いに対し移動可能である、
カテーテル。 - 前記イメージング装置が、血管内超音波トランスデューサ及び光学コヒーレンストモグラフィ装置の少なくとも一方を有する、請求項1に記載のカテーテル。
- 前記イメージング装置及び前記バルーンアセンブリが、互いから固定の距離隔てたところに位置付けられる、請求項1又は2に記載のカテーテル。
- 患者の身体又は前記バルーンアセンブリの外側に延在する前記カテーテルの少なくとも一部に位置付けられる距離マーカを更に有する、請求項1乃至3のいずれか1項に記載のカテーテル。
- 前記薬剤溶出コーティングがパクリテキセルを含む、請求項1乃至4のいずれか1項に記載のカテーテル。
- 前記バルーンアセンブリが非従順である、請求項1乃至5のいずれか1項に記載のカテーテル。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201462011276P | 2014-06-12 | 2014-06-12 | |
| US62/011,276 | 2014-06-12 | ||
| PCT/US2015/035609 WO2015192041A1 (en) | 2014-06-12 | 2015-06-12 | Image guided therapeutic catheter with drug eluting balloon |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2017524397A JP2017524397A (ja) | 2017-08-31 |
| JP2017524397A5 JP2017524397A5 (ja) | 2018-07-05 |
| JP6814044B2 true JP6814044B2 (ja) | 2021-01-13 |
Family
ID=54834424
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016570826A Active JP6814044B2 (ja) | 2014-06-12 | 2015-06-12 | 薬剤溶出性バルーンを有する画像ガイドされる治療カテーテル |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20150359433A1 (ja) |
| EP (1) | EP3154620B1 (ja) |
| JP (1) | JP6814044B2 (ja) |
| CN (1) | CN106456945B (ja) |
| WO (1) | WO2015192041A1 (ja) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3307172A4 (en) | 2015-06-15 | 2019-05-22 | Sunnybrook Research Institute | INTRAVASCULAR IMAGING CATHETERS AND METHODS OF USING SAME |
| US10813680B2 (en) * | 2017-03-27 | 2020-10-27 | Medtronic Cryocath Lp | Cryoballoon contact assessment using capacitive or resistive sensors |
| WO2023187510A1 (en) * | 2022-03-31 | 2023-10-05 | Medtronic, Inc. | Vessel modification using a therapeutic agent |
Family Cites Families (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5090959A (en) * | 1987-04-30 | 1992-02-25 | Advanced Cardiovascular Systems, Inc. | Imaging balloon dilatation catheter |
| US5372138A (en) * | 1988-03-21 | 1994-12-13 | Boston Scientific Corporation | Acousting imaging catheters and the like |
| EP0544820B1 (en) * | 1990-08-21 | 2000-05-24 | Boston Scientific Limited | Acoustic imaging catheter and the like |
| US5102402A (en) * | 1991-01-04 | 1992-04-07 | Medtronic, Inc. | Releasable coatings on balloon catheters |
| US5167233A (en) * | 1991-01-07 | 1992-12-01 | Endosonics Corporation | Dilating and imaging apparatus |
| US5183048A (en) * | 1991-06-24 | 1993-02-02 | Endosonics Corporation | Method and apparatus for removing artifacts from an ultrasonically generated image of a small cavity |
| AU1374095A (en) * | 1993-12-17 | 1995-07-03 | G. David Jang | Sliding receptacle catheter systems |
| GB2365127A (en) * | 2000-07-20 | 2002-02-13 | Jomed Imaging Ltd | Catheter |
| AU2003256856A1 (en) * | 2002-08-05 | 2004-02-23 | Miravant Medical Technologies, Inc. | Catheter for diagnosis and treatment of diseased vessels |
| DE102004001498B4 (de) * | 2004-01-09 | 2008-01-10 | Siemens Ag | Katheter zur Einführung in ein Gefäß |
| US8396548B2 (en) * | 2008-11-14 | 2013-03-12 | Vessix Vascular, Inc. | Selective drug delivery in a lumen |
| US20070060995A1 (en) * | 2005-08-19 | 2007-03-15 | Oliver Meissner | Workflow for cardiovascular intervention |
| US20070073364A1 (en) * | 2005-09-29 | 2007-03-29 | Siemens Aktiengesellschaft | Combined OCT catheter device and method for combined optical coherence tomography (OCT) diagnosis and photodynamic therapy (PDT) |
| US20090187108A1 (en) * | 2006-09-29 | 2009-07-23 | Cornova, Inc. | Systems and methods for analysis and treatment of a body lumen |
| US20080175887A1 (en) * | 2006-11-20 | 2008-07-24 | Lixiao Wang | Treatment of Asthma and Chronic Obstructive Pulmonary Disease With Anti-proliferate and Anti-inflammatory Drugs |
| JP5237567B2 (ja) * | 2007-02-21 | 2013-07-17 | 株式会社グツドマン | バルーンカテーテル |
| EP2865404B1 (en) * | 2007-06-01 | 2019-09-11 | Covidien LP | Extension tubes for balloon catheters |
| US8100855B2 (en) * | 2007-09-17 | 2012-01-24 | Abbott Cardiovascular Systems, Inc. | Methods and devices for eluting agents to a vessel |
| US8076529B2 (en) * | 2008-09-26 | 2011-12-13 | Abbott Cardiovascular Systems, Inc. | Expandable member formed of a fibrous matrix for intraluminal drug delivery |
| US20120330334A1 (en) * | 2010-12-31 | 2012-12-27 | Volcano Corporation | Therapeutic Cutting Devices, Systems, and Methods for Multiple Sclerosis, Deep Vein Thrombosis, and Pulmonary Embolism |
| US20120310210A1 (en) * | 2011-03-04 | 2012-12-06 | Campbell Carey V | Eluting medical devices |
| CN106421933A (zh) * | 2011-10-18 | 2017-02-22 | 米歇尔技术公司 | 药物递送医疗装置 |
| WO2014031736A1 (en) * | 2012-08-22 | 2014-02-27 | Volcano Corporation | Balloon catheter junction |
| US9314226B2 (en) * | 2012-08-24 | 2016-04-19 | Volcano Corporation | System and method for focusing ultrasound image data |
| EP2928540B1 (en) * | 2012-12-07 | 2020-10-07 | Volcano Corporation | High pressure therapeutic and imaging catheter |
| US20140276028A1 (en) * | 2013-03-15 | 2014-09-18 | Volcano Corporation | Integrated Therapeutic Imaging Catheter and Methods |
-
2015
- 2015-06-12 US US14/738,372 patent/US20150359433A1/en active Pending
- 2015-06-12 JP JP2016570826A patent/JP6814044B2/ja active Active
- 2015-06-12 WO PCT/US2015/035609 patent/WO2015192041A1/en active Application Filing
- 2015-06-12 EP EP15806028.5A patent/EP3154620B1/en active Active
- 2015-06-12 CN CN201580031200.6A patent/CN106456945B/zh active Active
Also Published As
| Publication number | Publication date |
|---|---|
| US20150359433A1 (en) | 2015-12-17 |
| CN106456945A (zh) | 2017-02-22 |
| EP3154620B1 (en) | 2020-09-09 |
| EP3154620A4 (en) | 2017-05-31 |
| WO2015192041A1 (en) | 2015-12-17 |
| CN106456945B (zh) | 2020-06-16 |
| JP2017524397A (ja) | 2017-08-31 |
| EP3154620A1 (en) | 2017-04-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20190175148A1 (en) | Distance, diameter and area determining device | |
| JP4621771B2 (ja) | 撮像システムおよび撮像ガイドワイヤ | |
| JP5107931B2 (ja) | 改良された超音波画像を生成するように構成されたエコー源性針カテーテル | |
| US10709312B2 (en) | Transitional region having cuts and a skive for an imaging catheter | |
| US20140276028A1 (en) | Integrated Therapeutic Imaging Catheter and Methods | |
| JP6814044B2 (ja) | 薬剤溶出性バルーンを有する画像ガイドされる治療カテーテル | |
| EP3229700B1 (en) | Distance, diameter and area measuring device | |
| US20220409858A1 (en) | Electromagnetic-radiation-cured radiopaque marker and associated devices, systems, and methods | |
| CN113301939B (zh) | 可变硬度管腔内设备 | |
| JP6382833B2 (ja) | 高圧の治療用および画像化用カテーテル | |
| US20180317879A1 (en) | Guarded imaging devices and methods | |
| US11707258B2 (en) | Device and method for intravascular imaging and sensing | |
| EP3672491B1 (en) | Adjustable flexibility/stiffness intraluminal device | |
| US11819360B2 (en) | Intraluminal rotational ultrasound for diagnostic imaging and therapy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20170214 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20180524 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20180524 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20190226 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20190319 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20190612 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190826 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20200107 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20200407 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20201124 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20201218 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6814044 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |