JP6654898B2 - 標的組織指向型薬物送達のための装置および方法 - Google Patents
標的組織指向型薬物送達のための装置および方法 Download PDFInfo
- Publication number
- JP6654898B2 JP6654898B2 JP2015531989A JP2015531989A JP6654898B2 JP 6654898 B2 JP6654898 B2 JP 6654898B2 JP 2015531989 A JP2015531989 A JP 2015531989A JP 2015531989 A JP2015531989 A JP 2015531989A JP 6654898 B2 JP6654898 B2 JP 6654898B2
- Authority
- JP
- Japan
- Prior art keywords
- heart
- patient
- minimally invasive
- drug
- invasive device
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000012377 drug delivery Methods 0.000 title claims description 42
- 238000000034 method Methods 0.000 title description 53
- 210000002216 heart Anatomy 0.000 claims description 172
- 239000003814 drug Substances 0.000 claims description 145
- 229940079593 drug Drugs 0.000 claims description 144
- 239000012528 membrane Substances 0.000 claims description 47
- 239000000463 material Substances 0.000 claims description 34
- 239000003416 antiarrhythmic agent Substances 0.000 claims description 28
- 206010003658 Atrial Fibrillation Diseases 0.000 claims description 23
- 230000003444 anaesthetic effect Effects 0.000 claims description 13
- REQCZEXYDRLIBE-UHFFFAOYSA-N procainamide Chemical group CCN(CC)CCNC(=O)C1=CC=C(N)C=C1 REQCZEXYDRLIBE-UHFFFAOYSA-N 0.000 claims description 12
- 229960000244 procainamide Drugs 0.000 claims description 12
- NNJVILVZKWQKPM-UHFFFAOYSA-N Lidocaine Chemical compound CCN(CC)CC(=O)NC1=C(C)C=CC=C1C NNJVILVZKWQKPM-UHFFFAOYSA-N 0.000 claims description 11
- 229960004194 lidocaine Drugs 0.000 claims description 11
- 229960004919 procaine Drugs 0.000 claims description 11
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 claims description 11
- 239000000853 adhesive Substances 0.000 claims description 10
- 230000001070 adhesive effect Effects 0.000 claims description 10
- 229960005260 amiodarone Drugs 0.000 claims description 9
- IYIKLHRQXLHMJQ-UHFFFAOYSA-N amiodarone Chemical compound CCCCC=1OC2=CC=CC=C2C=1C(=O)C1=CC(I)=C(OCCN(CC)CC)C(I)=C1 IYIKLHRQXLHMJQ-UHFFFAOYSA-N 0.000 claims 3
- 239000002998 adhesive polymer Substances 0.000 claims 1
- 230000001746 atrial effect Effects 0.000 description 49
- 210000004379 membrane Anatomy 0.000 description 42
- 239000004020 conductor Substances 0.000 description 34
- 230000033764 rhythmic process Effects 0.000 description 34
- 230000000694 effects Effects 0.000 description 32
- 108091006146 Channels Proteins 0.000 description 30
- 238000011282 treatment Methods 0.000 description 29
- 238000013194 cardioversion Methods 0.000 description 27
- 210000002837 heart atrium Anatomy 0.000 description 22
- 239000000126 substance Substances 0.000 description 20
- 210000000779 thoracic wall Anatomy 0.000 description 19
- 230000001684 chronic effect Effects 0.000 description 18
- 230000001154 acute effect Effects 0.000 description 17
- 230000000747 cardiac effect Effects 0.000 description 17
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 16
- 208000035475 disorder Diseases 0.000 description 16
- 206010003119 arrhythmia Diseases 0.000 description 14
- 229910052751 metal Inorganic materials 0.000 description 13
- 239000002184 metal Substances 0.000 description 13
- 210000005245 right atrium Anatomy 0.000 description 13
- 238000010438 heat treatment Methods 0.000 description 12
- 238000001990 intravenous administration Methods 0.000 description 12
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 11
- 238000002513 implantation Methods 0.000 description 11
- 230000002829 reductive effect Effects 0.000 description 11
- 229910052709 silver Inorganic materials 0.000 description 11
- 239000004332 silver Substances 0.000 description 11
- 239000003795 chemical substances by application Substances 0.000 description 10
- 238000001816 cooling Methods 0.000 description 10
- 230000006870 function Effects 0.000 description 10
- 238000002360 preparation method Methods 0.000 description 10
- 230000007012 clinical effect Effects 0.000 description 9
- 230000009885 systemic effect Effects 0.000 description 9
- 231100000057 systemic toxicity Toxicity 0.000 description 9
- 229940127291 Calcium channel antagonist Drugs 0.000 description 8
- 238000004519 manufacturing process Methods 0.000 description 8
- 150000002739 metals Chemical class 0.000 description 8
- 210000001519 tissue Anatomy 0.000 description 8
- 230000008901 benefit Effects 0.000 description 7
- 239000002876 beta blocker Substances 0.000 description 7
- 229940097320 beta blocking agent Drugs 0.000 description 7
- 239000000480 calcium channel blocker Substances 0.000 description 7
- 210000005242 cardiac chamber Anatomy 0.000 description 7
- 238000001647 drug administration Methods 0.000 description 7
- 238000001746 injection moulding Methods 0.000 description 7
- 210000005246 left atrium Anatomy 0.000 description 7
- 238000012546 transfer Methods 0.000 description 7
- ITPDYQOUSLNIHG-UHFFFAOYSA-N Amiodarone hydrochloride Chemical compound [Cl-].CCCCC=1OC2=CC=CC=C2C=1C(=O)C1=CC(I)=C(OCC[NH+](CC)CC)C(I)=C1 ITPDYQOUSLNIHG-UHFFFAOYSA-N 0.000 description 6
- 230000002159 abnormal effect Effects 0.000 description 6
- 230000005856 abnormality Effects 0.000 description 6
- 239000000956 alloy Substances 0.000 description 6
- 229910045601 alloy Inorganic materials 0.000 description 6
- 230000006793 arrhythmia Effects 0.000 description 6
- 210000001992 atrioventricular node Anatomy 0.000 description 6
- 230000001186 cumulative effect Effects 0.000 description 6
- 230000009545 invasion Effects 0.000 description 6
- 229920002379 silicone rubber Polymers 0.000 description 6
- 239000004945 silicone rubber Substances 0.000 description 6
- 238000001356 surgical procedure Methods 0.000 description 6
- 229940035674 anesthetics Drugs 0.000 description 5
- 239000003193 general anesthetic agent Substances 0.000 description 5
- 239000012212 insulator Substances 0.000 description 5
- 230000007246 mechanism Effects 0.000 description 5
- 230000004048 modification Effects 0.000 description 5
- 238000012986 modification Methods 0.000 description 5
- 238000001050 pharmacotherapy Methods 0.000 description 5
- 230000035939 shock Effects 0.000 description 5
- 230000000638 stimulation Effects 0.000 description 5
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 4
- 230000006378 damage Effects 0.000 description 4
- 230000036541 health Effects 0.000 description 4
- 210000005003 heart tissue Anatomy 0.000 description 4
- 230000002980 postoperative effect Effects 0.000 description 4
- 238000002560 therapeutic procedure Methods 0.000 description 4
- 241001465754 Metazoa Species 0.000 description 3
- 208000005228 Pericardial Effusion Diseases 0.000 description 3
- 206010039897 Sedation Diseases 0.000 description 3
- 230000005540 biological transmission Effects 0.000 description 3
- 239000002327 cardiovascular agent Substances 0.000 description 3
- 229940125692 cardiovascular agent Drugs 0.000 description 3
- 229910052802 copper Inorganic materials 0.000 description 3
- 239000010949 copper Substances 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 230000004064 dysfunction Effects 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 150000004677 hydrates Chemical class 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 239000002207 metabolite Substances 0.000 description 3
- 210000004912 pericardial fluid Anatomy 0.000 description 3
- 229940002612 prodrug Drugs 0.000 description 3
- 239000000651 prodrug Substances 0.000 description 3
- 230000036280 sedation Effects 0.000 description 3
- 229940125723 sedative agent Drugs 0.000 description 3
- 239000000932 sedative agent Substances 0.000 description 3
- 238000005476 soldering Methods 0.000 description 3
- 230000002861 ventricular Effects 0.000 description 3
- SGTNSNPWRIOYBX-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-{[2-(3,4-dimethoxyphenyl)ethyl](methyl)amino}-2-(propan-2-yl)pentanenitrile Chemical compound C1=C(OC)C(OC)=CC=C1CCN(C)CCCC(C#N)(C(C)C)C1=CC=C(OC)C(OC)=C1 SGTNSNPWRIOYBX-UHFFFAOYSA-N 0.000 description 2
- 206010003130 Arrhythmia supraventricular Diseases 0.000 description 2
- 206010003662 Atrial flutter Diseases 0.000 description 2
- 208000017667 Chronic Disease Diseases 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- 208000001871 Tachycardia Diseases 0.000 description 2
- 208000027418 Wounds and injury Diseases 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 102000012740 beta Adrenergic Receptors Human genes 0.000 description 2
- 108010079452 beta Adrenergic Receptors Proteins 0.000 description 2
- 238000007675 cardiac surgery Methods 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 238000002716 delivery method Methods 0.000 description 2
- 238000003745 diagnosis Methods 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 230000037024 effective refractory period Effects 0.000 description 2
- 230000008030 elimination Effects 0.000 description 2
- 238000003379 elimination reaction Methods 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 238000002695 general anesthesia Methods 0.000 description 2
- 239000007943 implant Substances 0.000 description 2
- 238000001802 infusion Methods 0.000 description 2
- 208000014674 injury Diseases 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- 238000002690 local anesthesia Methods 0.000 description 2
- 230000033001 locomotion Effects 0.000 description 2
- 238000012806 monitoring device Methods 0.000 description 2
- 238000012544 monitoring process Methods 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 230000036407 pain Effects 0.000 description 2
- 230000036961 partial effect Effects 0.000 description 2
- 210000003516 pericardium Anatomy 0.000 description 2
- 229920001296 polysiloxane Polymers 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 239000003450 potassium channel blocker Substances 0.000 description 2
- 210000003492 pulmonary vein Anatomy 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 150000003839 salts Chemical class 0.000 description 2
- 231100000241 scar Toxicity 0.000 description 2
- 238000009958 sewing Methods 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 239000010703 silicon Substances 0.000 description 2
- 239000012453 solvate Substances 0.000 description 2
- 230000002889 sympathetic effect Effects 0.000 description 2
- 229920002994 synthetic fiber Polymers 0.000 description 2
- 238000007910 systemic administration Methods 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 230000004797 therapeutic response Effects 0.000 description 2
- 210000000115 thoracic cavity Anatomy 0.000 description 2
- 210000003462 vein Anatomy 0.000 description 2
- 229960001722 verapamil Drugs 0.000 description 2
- 206010001497 Agitation Diseases 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 108090000312 Calcium Channels Proteins 0.000 description 1
- 102000003922 Calcium Channels Human genes 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 208000030453 Drug-Related Side Effects and Adverse reaction Diseases 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 102000004310 Ion Channels Human genes 0.000 description 1
- 108090000862 Ion Channels Proteins 0.000 description 1
- 206010033372 Pain and discomfort Diseases 0.000 description 1
- 208000031481 Pathologic Constriction Diseases 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 208000003734 Supraventricular Tachycardia Diseases 0.000 description 1
- 239000004809 Teflon Substances 0.000 description 1
- 229920006362 Teflon® Polymers 0.000 description 1
- 108090000190 Thrombin Proteins 0.000 description 1
- 206010070863 Toxicity to various agents Diseases 0.000 description 1
- 238000002679 ablation Methods 0.000 description 1
- 230000036982 action potential Effects 0.000 description 1
- 239000000332 adrenergic beta-1 receptor antagonist Substances 0.000 description 1
- 229910002065 alloy metal Inorganic materials 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 210000003484 anatomy Anatomy 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 150000001557 benzodiazepines Chemical class 0.000 description 1
- 230000002051 biphasic effect Effects 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 238000013153 catheter ablation Methods 0.000 description 1
- 210000000038 chest Anatomy 0.000 description 1
- 230000015271 coagulation Effects 0.000 description 1
- 238000005345 coagulation Methods 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000011443 conventional therapy Methods 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 125000004925 dihydropyridyl group Chemical group N1(CC=CC=C1)* 0.000 description 1
- HSUGRBWQSSZJOP-RTWAWAEBSA-N diltiazem Chemical compound C1=CC(OC)=CC=C1[C@H]1[C@@H](OC(C)=O)C(=O)N(CCN(C)C)C2=CC=CC=C2S1 HSUGRBWQSSZJOP-RTWAWAEBSA-N 0.000 description 1
- 229960004166 diltiazem Drugs 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 238000002651 drug therapy Methods 0.000 description 1
- 210000005081 epithelial layer Anatomy 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 230000005021 gait Effects 0.000 description 1
- 230000035876 healing Effects 0.000 description 1
- 210000002064 heart cell Anatomy 0.000 description 1
- 208000019622 heart disease Diseases 0.000 description 1
- 230000000004 hemodynamic effect Effects 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 238000011866 long-term treatment Methods 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 230000007721 medicinal effect Effects 0.000 description 1
- 238000002324 minimally invasive surgery Methods 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 230000002107 myocardial effect Effects 0.000 description 1
- 210000004165 myocardium Anatomy 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 230000001151 other effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 230000036470 plasma concentration Effects 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 229940125422 potassium channel blocker Drugs 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 230000010349 pulsation Effects 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 230000036279 refractory period Effects 0.000 description 1
- 230000002336 repolarization Effects 0.000 description 1
- 238000002633 shock therapy Methods 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 230000037380 skin damage Effects 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000003195 sodium channel blocking agent Substances 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 229960002370 sotalol Drugs 0.000 description 1
- ZBMZVLHSJCTVON-UHFFFAOYSA-N sotalol Chemical compound CC(C)NCC(O)C1=CC=C(NS(C)(=O)=O)C=C1 ZBMZVLHSJCTVON-UHFFFAOYSA-N 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 230000036262 stenosis Effects 0.000 description 1
- 208000037804 stenosis Diseases 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000006794 tachycardia Effects 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 239000004416 thermosoftening plastic Substances 0.000 description 1
- 229960004072 thrombin Drugs 0.000 description 1
- 239000012780 transparent material Substances 0.000 description 1
- 238000009966 trimming Methods 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 238000009423 ventilation Methods 0.000 description 1
- 208000003663 ventricular fibrillation Diseases 0.000 description 1
- 206010047302 ventricular tachycardia Diseases 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M31/00—Devices for introducing or retaining media, e.g. remedies, in cavities of the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0021—Catheters; Hollow probes characterised by the form of the tubing
- A61M25/0023—Catheters; Hollow probes characterised by the form of the tubing by the form of the lumen, e.g. cross-section, variable diameter
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/05—Electrodes for implantation or insertion into the body, e.g. heart electrode
- A61N1/0587—Epicardial electrode systems; Endocardial electrodes piercing the pericardium
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/05—Electrodes for implantation or insertion into the body, e.g. heart electrode
- A61N1/0587—Epicardial electrode systems; Endocardial electrodes piercing the pericardium
- A61N1/0597—Surface area electrodes, e.g. cardiac harness
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/38—Applying electric currents by contact electrodes alternating or intermittent currents for producing shock effects
- A61N1/39—Heart defibrillators
- A61N1/3956—Implantable devices for applying electric shocks to the heart, e.g. for cardioversion
- A61N1/3962—Implantable devices for applying electric shocks to the heart, e.g. for cardioversion in combination with another heart therapy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M19/00—Local anaesthesia; Hypothermia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M2025/0004—Catheters; Hollow probes having two or more concentrically arranged tubes for forming a concentric catheter system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/0404—Electrodes for external use
- A61N1/0408—Use-related aspects
- A61N1/046—Specially adapted for shock therapy, e.g. defibrillation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/0404—Electrodes for external use
- A61N1/0472—Structure-related aspects
- A61N1/0476—Array electrodes (including any electrode arrangement with more than one electrode for at least one of the polarities)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/05—Electrodes for implantation or insertion into the body, e.g. heart electrode
- A61N1/0587—Epicardial electrode systems; Endocardial electrodes piercing the pericardium
- A61N1/0595—Temporary leads
Description
・クラスI――ナトリウムチャネル遮断薬
・クラスII――βブロッカー
・クラスIII――カリウムチャネル遮断薬
・クラスIV――カルシウムチャンネル遮断薬
例として、下の表に本発明に基づいて臨床的に使用可能な典型的なクラスI薬剤を挙げる。下の表には例として、それぞれの薬剤の典型的な治療上の用途のいくつかも挙げる。
例として、下の表に本発明に基づいて臨床的に使用可能な典型的なβブロッカーを挙げる。ここに含まれるのは(1)非選択的遮断薬(β1受容体およびβ2受容体の両方を遮断)または(2)比較的選択的なβ1ブロッカー(「心選択性」βブロッカー)。βブロッカーの中にはβ遮断に加えて独特の薬効薬理を示すものがあることが知られている。βブロッカーの2つのクラスそれぞれを代表する薬剤を下の表にあくまでも例として示す。
例として、下の表に本発明に基づいて臨床的に使用可能な典型的なクラスIII薬剤(カリウムチャネル遮断薬)を挙げる。下の表には例として、それぞれの薬剤の典型的な治療上の用途のいくつかも挙げる。
(クラスIV抗不整脈薬である)カルシウムチャネル遮断薬(CCB)の薬効薬理や特性はよく知られており、それは同薬剤の心臓内の歩調取り異常部位の発信ペースを下げる能力と、特に房室結節(AV)における伝導速度を下げかつ再分極を引き延ばす能力とに関係がある。後者の房室結節(AV)における作用により、上室性頻拍を引き起こすリエントリー機構を遮断する。本発明に基づいて使用可能な典型的なCCBには例えば次のようなものがある。ベラパミル(フェニルアルキルアミンのクラス)は心筋に対して選択性を示し、患者の不整脈を治療するのに用いられる。ジルチアゼム(ベンゾジアゼピンのクラス)は、CCBの他の一例であって、脈管カルシウムチャネルに対する選択性の点で、ベラパミルとジヒドロピリジン系薬物との中間にあたるものである。これらは本発明に従って使用可能な数多いCCBの例のうちのごく一部に過ぎないと理解されたい。
本発明の好ましい実施形態によれば、1つ以上のアルゴリズムを用いて、例えば本発明に係るインターフェース装置の動作を制御/プログラムすることができる。例えば、1つ以上のアルゴリズムを用いて、例えば本発明に係るインターフェース装置を介して患者の心臓の目標領域へ1種以上の薬剤の送達するための同装置の動作を制御/プログラムすることができる。本明細書に記載のとおり、心臓に送達される薬物(薬剤)は任意数の種類の薬物であって、例えばこれには1種以上の麻酔薬、鎮静剤および/または他のクラスの薬剤あるいはこれらの任意の組み合わせが含まれる。このような薬物を本発明に係るインターフェース装置に供給する手段として、例えば1つ以上のアルゴリズムを用いて、有効薬物を他の製剤、例えば生理食塩水、ヘパリン、抗生剤、その他、と共にパルス様に投入して、パルス幅変調を行うことができる。典型的な一態様によれば、急性症例に対する除細動中に、単位時間ごとの薬剤の量を他の薬物の量に対して増やしていくことができる。いくつかの実施形態によれば、低侵襲で植え込み型の装置内を正圧に維持することで、凝固や目詰まりを防ぎ、また治療中の重大な局面での処置の中断を防ぐことができる。
本例では、術後の心臓組織に対して直接行われるアミオダロン、ソタロールおよびプロカインアミドの標的指向型局所投与において、従来の方法で用いられる全身薬量と比べてはるかに少ない薬量で本発明が効果を上げることが実証された。本例について、下の表1に各薬物の、術後の経口および静脈経由投与(従来の治療における投与方法)による薬量と、それと対照して本発明に係る装置を用いた標的組織指向型送達による薬量を示す。表1から理解されるように、本発明を利用して薬物の標的指向型送達を実施することにより、治療に必要な薬量が遥かに少なくなる。これはまた本発明を利用すれば全身毒性が著しく低減されることを意味する
抗不整脈薬プロカインアミドの心膜内(IPC)送達と静脈経由(IV)送達とを比較する実験を行った。実験の1つにおいて、2グループのブタ(以下動物)に対してプロカインアミドを静脈経由送達(n=6)または心膜送達(n=7)によって投与した。投与量は静脈経由が順次2、8および16各mg/kg(累積2、10および26各mg/kg)、心膜が順次0.5、1および2各mg/kg(累積0.5、1.5、および3.5各mg/kg)であった。いずれの投与量においても、10mLで再懸濁し、1mL/分のレートで投与した。2つの薬物送達方法間の薬物動態の違いは統計学的に著しいものであった。心膜送達では、ピーク薬物濃度が250〜900μg/mLの範囲内であったのに対し、血漿中で測定し得たレベルは低かった(<1μg/mL)。静脈経由送達では、ピーク薬物濃度は<1〜40μg/mLで、心嚢液中濃度は血漿中濃度に等しかった。静脈経由送達による心嚢液中濃度が、心膜内送達による心嚢液中濃度の1/100であることに注目すべきである。心房の電気生理学的数値は2つの送達方法間で同様であった。これはより低い直接投与量で同様の治療反応が得られたことを示している。心膜送達での累積量3.5mg/kgは右心房のERPをベースラインより22%増加させたのに対して、静脈経由送達における累積量10および26各mg/kgは心房ERPをそれぞれ24%および18%引き延ばした。加えて、累積量3.5mg/kg(心膜)および26mg/kg(静脈経由)は動物のそれぞれ71%および83%において心房細動の誘発を防いだ。心室の電気生理学的特性に関しては、心膜送達は心室の電気生理学的特性、心拍数、あるいは全身血圧にさしたる影響を及ぼさなかった。しかしながら、26mg/kgの投与量で静脈注射で投与したプロカインアミドは心房/心室伝導時間を引き延ばし、両心室間伝導(RV興奮およびLV興奮)を低減した。これは、静脈経由送達が心臓の全体に影響を与えるのに対して、IPCが特に心房を標的とすることを示している。また、別の5頭の動物に2mg/kgの投与量で1回限り心膜送達での投与を行ったところ、電気生理学的特性および薬物動態に対する影響の点では累積投与と同様の結果が得られた。以上の典型的な実験からIPCであればより少ない(約10分の1)投与量で、IV注射によるより多い投与量と同等の効果を心房の電気生理学的特性に及ぼすことができ、しかも心室の電気生理学的特性に影響しない。
Claims (9)
- 患者の治療に用いる低侵襲装置であって、
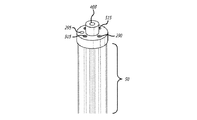
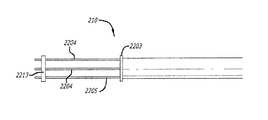
(a)前記患者の心臓の表面に薬物を送達可能に構成される少なくとも1つの薬物送達チャネルを含む少なくとも1つの内側カテーテルと、
(b)前記少なくとも1つの内側カテーテルを実質的に取り囲む外側カテーテルと、
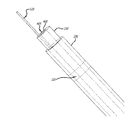
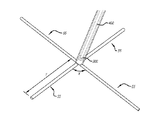
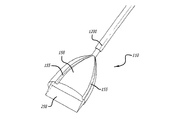
(c)前記内側カテーテル内にあり且つ前記薬物送達チャネルを取り囲み尚且つ前記低侵襲装置の実質的に遠位部に設けられるように延びる複数の導電性エレメントと、
(d)前記低侵襲装置の実質的に遠位部で前記複数の導電性エレメントを互いに接続し、心臓の拍動に合わせて変形できる、少なくとも1枚の膜と、
を備え、
前記複数の導電性エレメントは、該低侵襲装置の前記実質的に遠位部にて前記膜と相互に接続されるとともに、中心部から漏斗状に広がって花状に配置され、
前記少なくとも1つの薬物送達チャネルが前記内側カテーテル内に設けられ、
前記複数の導電性エレメントが前記患者の心臓表面に除細動エネルギーを供給可能に構成され、
前記低侵襲装置は、慢性的な心房細動を治療することに効果を有する
ことを特徴とする低侵襲装置。 - 請求項1に記載の低侵襲装置であって、少なくとも1種の接着性素材が前記膜の裏側に設けられていることを特徴とする低侵襲装置。
- 請求項2に記載の低侵襲装置であって、前記少なくとも1種の接着性素材は少なくとも1種の接着性ポリマーからなることを特徴とする低侵襲装置。
- 請求項1に記載の低侵襲装置であって、少なくとも1種の抗不整脈薬を備えることを特徴とする低侵襲装置。
- 請求項4に記載の低侵襲装置であって、前記少なくとも1種の抗不整脈薬はプロカインアミド、アミオダロン、プロカインアミドと他の少なくとも1種の抗不整脈薬との組み合わせ、アミオダロンと他の少なくとも1種の抗不整脈薬との組み合わせ、およびプロカインアミドとアミオダロンと他の少なくとも1種の抗不整脈薬との組み合わせからなる群から選ばれることを特徴とする低侵襲装置。
- 請求項1に記載の低侵襲装置であって、少なくとも1種の麻酔薬を備えることを特徴とすることを特徴とする低侵襲装置。
- 請求項6に記載の低侵襲装置であって、前記麻酔薬はプロカイン、リドカイン、プロカインと他の少なくとも1種の麻酔薬との組み合わせ、リドカインと他の少なくとも1種の麻酔薬との組み合わせ、およびプロカインとリドカインと他の少なくとも1種の麻酔薬との組み合わせからなる群から選ばれることを特徴とする低侵襲装置。
- 患者の治療に用いる低侵襲装置であって、
(a)少なくとも1つの内側カテーテルと、
(b)前記内側カテーテル内に設けられ、前記患者の心臓の表面に薬物を送達可能に構成される少なくとも1つの薬物送達チャネルと、
(c)前記少なくとも1つの内側カテーテルを実質的に取り囲む外側カテーテルと、
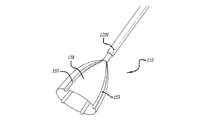
(d)前記低侵襲装置の実質的に遠位部に設けられ尚且つ前記内側カテーテル内に位置して前記薬物送達チャネルを取り囲む小葉状アセンブリと、
を備え、
前記小葉状アセンブリは、少なくとも1つの導電性エレメントを有して、少なくとも1つの膜により相互に接続され、
前記少なくとも1つの膜は、心臓の拍動に合わせて変形可能であり、
前記低侵襲装置の前記実質的に遠位部には少なくとも1つの薬物放出部が設けられ、該薬物放出部を介して少なくとも1種の薬物が前記患者の前記心臓表面に送達される
ことを特徴とする低侵襲装置。 - 請求項8に記載の低侵襲装置であって、前記小葉状アセンブリは、中心部から漏斗状に広がって花状に配置される複数の小葉状片を有し、
前記複数の小葉状片は、前記患者の心臓表面に除細動エネルギーを供給する複数の前記導電性エレメントと結合されることを特徴とする低侵襲装置。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201261743759P | 2012-09-11 | 2012-09-11 | |
| US61/743,759 | 2012-09-11 | ||
| US201361749713P | 2013-01-07 | 2013-01-07 | |
| US61/749,713 | 2013-01-07 | ||
| PCT/US2013/058814 WO2014043039A1 (en) | 2012-09-11 | 2013-09-09 | Device and methods for targeted tissue drug delivery |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2015529531A JP2015529531A (ja) | 2015-10-08 |
| JP6654898B2 true JP6654898B2 (ja) | 2020-02-26 |
Family
ID=50278622
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2015531989A Active JP6654898B2 (ja) | 2012-09-11 | 2013-09-09 | 標的組織指向型薬物送達のための装置および方法 |
Country Status (9)
| Country | Link |
|---|---|
| US (4) | US10118014B2 (ja) |
| EP (1) | EP2895236B1 (ja) |
| JP (1) | JP6654898B2 (ja) |
| KR (1) | KR102223218B1 (ja) |
| CN (2) | CN107837453A (ja) |
| BR (1) | BR112015005450B1 (ja) |
| ES (1) | ES2697683T3 (ja) |
| HK (1) | HK1211249A1 (ja) |
| WO (1) | WO2014043039A1 (ja) |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107837453A (zh) * | 2012-09-11 | 2018-03-27 | 本土心血管公司 | 用于向靶组织给药的装置和方法 |
| US11690645B2 (en) | 2017-05-03 | 2023-07-04 | Medtronic Vascular, Inc. | Tissue-removing catheter |
| EP3618737B1 (en) | 2017-05-03 | 2022-03-16 | Medtronic Vascular Inc. | Tissue-removing catheter |
| CA3066361A1 (en) | 2017-06-07 | 2018-12-13 | Shifamed Holdings, Llc | Intravascular fluid movement devices, systems, and methods of use |
| CN107617140B (zh) * | 2017-08-15 | 2022-06-24 | 杭州德柯医疗科技有限公司 | 心室壁注射辅助器械 |
| CN107638615B (zh) * | 2017-08-15 | 2022-06-24 | 杭州德柯医疗科技有限公司 | 心室壁注射辅助器械 |
| EP3710076B1 (en) | 2017-11-13 | 2023-12-27 | Shifamed Holdings, LLC | Intravascular fluid movement devices, systems, and methods of use |
| EP3746149A4 (en) | 2018-02-01 | 2021-10-27 | Shifamed Holdings, LLC | INTRAVASCULAR BLOOD PUMPS AND METHODS OF USE AND METHODS OF MANUFACTURING |
| EP3880096A1 (en) | 2018-11-16 | 2021-09-22 | Medtronic Vascular Inc. | Tissue-removing catheter |
| US11819236B2 (en) | 2019-05-17 | 2023-11-21 | Medtronic Vascular, Inc. | Tissue-removing catheter |
| JP2022540616A (ja) | 2019-07-12 | 2022-09-16 | シファメド・ホールディングス・エルエルシー | 血管内血液ポンプならびに製造および使用の方法 |
| US11654275B2 (en) | 2019-07-22 | 2023-05-23 | Shifamed Holdings, Llc | Intravascular blood pumps with struts and methods of use and manufacture |
| WO2021062265A1 (en) | 2019-09-25 | 2021-04-01 | Shifamed Holdings, Llc | Intravascular blood pump systems and methods of use and control thereof |
| US20220379111A1 (en) * | 2019-10-29 | 2022-12-01 | Nippon Telegraph And Telephone Corporation | Biological electrode and cardiac pacemaker |
Family Cites Families (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5282845A (en) * | 1990-10-01 | 1994-02-01 | Ventritex, Inc. | Multiple electrode deployable lead |
| US6086582A (en) * | 1997-03-13 | 2000-07-11 | Altman; Peter A. | Cardiac drug delivery system |
| US7481759B2 (en) * | 2001-08-03 | 2009-01-27 | Cardiac Pacemakers, Inc. | Systems and methods for treatment of coronary artery disease |
| US6745079B2 (en) * | 2001-11-07 | 2004-06-01 | Medtronic, Inc. | Electrical tissue stimulation apparatus and method |
| US7270669B1 (en) | 2002-03-14 | 2007-09-18 | Medtronic, Inc. | Epicardial lead placement for bi-ventricular pacing using thoracoscopic approach |
| US6965798B2 (en) * | 2002-06-17 | 2005-11-15 | Kim Young D | Implantable device for treating atrial fibrillation and method of using same |
| US7130700B2 (en) * | 2002-11-19 | 2006-10-31 | Medtronic, Inc. | Multilumen body for an implantable medical device |
| FR2862521B1 (fr) * | 2003-11-24 | 2006-09-22 | Juan Carlos Chachques | Catheter de diagnostic et d'injection, notamment pour application cardiologique a coeur battant |
| US20050245972A1 (en) * | 2004-04-29 | 2005-11-03 | Medtronic, Inc. | Apparatus and methods for treating arrhythmia and lowering defibrillation threshold |
| US7567841B2 (en) * | 2004-08-20 | 2009-07-28 | Cardiac Pacemakers, Inc. | Method and apparatus for delivering combined electrical and drug therapies |
| EP1809374A4 (en) * | 2004-10-12 | 2008-09-10 | Closed Loop Therapies Ltd | IMPLANTABLE METHODS AND DEVICES FOR TREATING SUPRAVENTRICULAR ARRHYTHMIAS |
| US7899555B2 (en) * | 2006-04-11 | 2011-03-01 | Pacesetter, Inc. | Intrapericardial lead |
| AU2008269932A1 (en) | 2007-06-29 | 2009-01-08 | Cardiac Pacemakers, Inc. | Methods, devices and systems for cardiac rhythm management using an electrode arrangement |
| EP2195071A1 (en) | 2007-09-06 | 2010-06-16 | Boston Scientific Scimed, Inc. | Methods and devices for local therapeutic agent delivery to heart valves |
| WO2009099550A1 (en) * | 2008-02-07 | 2009-08-13 | Cardiac Pacemakers, Inc. | Wireless tissue electrostimulation |
| US20090287166A1 (en) * | 2008-05-19 | 2009-11-19 | Cna Tek Inc. | Integrated device for ischemic treatment |
| CN102159257B (zh) * | 2008-07-17 | 2015-11-25 | 米歇尔技术公司 | 药物递送医疗设备 |
| JP6200895B2 (ja) * | 2011-11-21 | 2017-09-20 | インキューブ ラブズ, エルエルシー | 心筋薬剤送達装置および方法 |
| CN107837453A (zh) * | 2012-09-11 | 2018-03-27 | 本土心血管公司 | 用于向靶组织给药的装置和方法 |
-
2013
- 2013-09-09 CN CN201711086410.2A patent/CN107837453A/zh active Pending
- 2013-09-09 WO PCT/US2013/058814 patent/WO2014043039A1/en active Application Filing
- 2013-09-09 CN CN201380058893.9A patent/CN105142719B/zh active Active
- 2013-09-09 BR BR112015005450-1A patent/BR112015005450B1/pt active IP Right Grant
- 2013-09-09 JP JP2015531989A patent/JP6654898B2/ja active Active
- 2013-09-09 EP EP13836415.3A patent/EP2895236B1/en active Active
- 2013-09-09 ES ES13836415T patent/ES2697683T3/es active Active
- 2013-09-09 US US14/427,607 patent/US10118014B2/en active Active
- 2013-09-09 KR KR1020157009296A patent/KR102223218B1/ko active IP Right Grant
-
2015
- 2015-12-04 HK HK15111973.1A patent/HK1211249A1/xx unknown
-
2018
- 2018-09-18 US US16/133,977 patent/US10456553B2/en active Active
-
2019
- 2019-09-13 US US16/569,789 patent/US11224718B2/en active Active
-
2021
- 2021-12-20 US US17/556,176 patent/US20230372667A9/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| US20200078553A1 (en) | 2020-03-12 |
| CN105142719B (zh) | 2017-12-08 |
| BR112015005450A2 (pt) | 2019-12-17 |
| KR102223218B1 (ko) | 2021-03-04 |
| CN105142719A (zh) | 2015-12-09 |
| US10118014B2 (en) | 2018-11-06 |
| CN107837453A (zh) | 2018-03-27 |
| EP2895236A1 (en) | 2015-07-22 |
| BR112015005450B1 (pt) | 2021-03-02 |
| JP2015529531A (ja) | 2015-10-08 |
| US20150224281A1 (en) | 2015-08-13 |
| ES2697683T3 (es) | 2019-01-25 |
| HK1211249A1 (en) | 2016-05-20 |
| WO2014043039A1 (en) | 2014-03-20 |
| US20230372667A9 (en) | 2023-11-23 |
| US10456553B2 (en) | 2019-10-29 |
| EP2895236B1 (en) | 2018-08-15 |
| US20190083742A1 (en) | 2019-03-21 |
| US20220168543A1 (en) | 2022-06-02 |
| US11224718B2 (en) | 2022-01-18 |
| EP2895236A4 (en) | 2016-07-13 |
| KR20150080491A (ko) | 2015-07-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6654898B2 (ja) | 標的組織指向型薬物送達のための装置および方法 | |
| US6296630B1 (en) | Device and method to slow or stop the heart temporarily | |
| US20010044619A1 (en) | Cardiac drug delivery system and method for use | |
| US20030074039A1 (en) | Devices and methods for vagus nerve stimulation | |
| US20060058597A1 (en) | Intraluminal electrode assembly | |
| CN113226443A (zh) | 平面外可偏转导管 | |
| JP2006501894A (ja) | 能動的流体送り込みカテーテル | |
| US20070253997A1 (en) | Drug delivery to alleviate chronic pelvic pain | |
| US7186247B2 (en) | Apparatus and system for delivery of drug therapies | |
| US6965798B2 (en) | Implantable device for treating atrial fibrillation and method of using same | |
| EP3790620B1 (en) | Bonding strip for fixing an electrode coil to a lead body | |
| US20150073288A1 (en) | Method and apparatus to control conduction through the heart to treat cardiac conditions | |
| Rosenheck et al. | The effect of overdrive pacing rate and duration on ventricular escape rhythms in patients with chronic complete atrioventricular block | |
| JP2002515766A (ja) | 埋め込み型薬理学的除細動器システム | |
| WO1997004834A2 (en) | Implantable pharmacological defibrillator system |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20160701 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160905 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20161028 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20170525 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20170606 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20170817 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20171106 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20171206 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20180123 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20180523 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20180531 |
|
| A912 | Re-examination (zenchi) completed and case transferred to appeal board |
Free format text: JAPANESE INTERMEDIATE CODE: A912 Effective date: 20180713 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20191120 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20200131 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6654898 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |