JP6483655B2 - 細胞注入針 - Google Patents
細胞注入針 Download PDFInfo
- Publication number
- JP6483655B2 JP6483655B2 JP2016500811A JP2016500811A JP6483655B2 JP 6483655 B2 JP6483655 B2 JP 6483655B2 JP 2016500811 A JP2016500811 A JP 2016500811A JP 2016500811 A JP2016500811 A JP 2016500811A JP 6483655 B2 JP6483655 B2 JP 6483655B2
- Authority
- JP
- Japan
- Prior art keywords
- tissue
- needle
- priming
- cavity
- cells
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000002347 injection Methods 0.000 title 1
- 239000007924 injection Substances 0.000 title 1
- 230000037452 priming Effects 0.000 claims description 92
- 239000012530 fluid Substances 0.000 claims description 23
- 230000000149 penetrating effect Effects 0.000 claims description 17
- 238000003780 insertion Methods 0.000 claims description 16
- 230000037431 insertion Effects 0.000 claims description 16
- 230000004044 response Effects 0.000 claims description 14
- 230000007704 transition Effects 0.000 claims description 12
- 230000035876 healing Effects 0.000 claims description 9
- 238000004891 communication Methods 0.000 claims description 6
- 238000000227 grinding Methods 0.000 claims description 4
- 230000009719 regenerative response Effects 0.000 claims description 4
- 238000002560 therapeutic procedure Methods 0.000 claims description 4
- 230000035515 penetration Effects 0.000 claims description 3
- 230000002093 peripheral effect Effects 0.000 claims 2
- 238000007789 sealing Methods 0.000 claims 1
- 210000004027 cell Anatomy 0.000 description 129
- 210000001519 tissue Anatomy 0.000 description 125
- 239000000463 material Substances 0.000 description 14
- 238000000034 method Methods 0.000 description 12
- 239000003550 marker Substances 0.000 description 7
- 230000008439 repair process Effects 0.000 description 7
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 230000001413 cellular effect Effects 0.000 description 4
- 238000000926 separation method Methods 0.000 description 4
- 230000008878 coupling Effects 0.000 description 3
- 238000010168 coupling process Methods 0.000 description 3
- 238000005859 coupling reaction Methods 0.000 description 3
- 238000003384 imaging method Methods 0.000 description 3
- 230000010354 integration Effects 0.000 description 3
- 238000011068 loading method Methods 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 230000008929 regeneration Effects 0.000 description 3
- 238000011069 regeneration method Methods 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 230000001225 therapeutic effect Effects 0.000 description 3
- 238000002604 ultrasonography Methods 0.000 description 3
- 230000005653 Brownian motion process Effects 0.000 description 2
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 2
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 238000005537 brownian motion Methods 0.000 description 2
- 239000012876 carrier material Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 238000005520 cutting process Methods 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 210000002744 extracellular matrix Anatomy 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 210000000056 organ Anatomy 0.000 description 2
- 210000000813 small intestine Anatomy 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 210000004876 tela submucosa Anatomy 0.000 description 2
- 210000005166 vasculature Anatomy 0.000 description 2
- 241001631457 Cannula Species 0.000 description 1
- 229920001661 Chitosan Polymers 0.000 description 1
- 206010020843 Hyperthermia Diseases 0.000 description 1
- 206010061216 Infarction Diseases 0.000 description 1
- 239000004696 Poly ether ether ketone Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 229920000954 Polyglycolide Polymers 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- 206010066218 Stress Urinary Incontinence Diseases 0.000 description 1
- WYTGDNHDOZPMIW-RCBQFDQVSA-N alstonine Natural products C1=CC2=C3C=CC=CC3=NC2=C2N1C[C@H]1[C@H](C)OC=C(C(=O)OC)[C@H]1C2 WYTGDNHDOZPMIW-RCBQFDQVSA-N 0.000 description 1
- 230000003466 anti-cipated effect Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000000560 biocompatible material Substances 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 238000002716 delivery method Methods 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 210000003238 esophagus Anatomy 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 238000011010 flushing procedure Methods 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 230000036031 hyperthermia Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000007574 infarction Effects 0.000 description 1
- 229940102223 injectable solution Drugs 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 238000000608 laser ablation Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 238000009428 plumbing Methods 0.000 description 1
- 229920000747 poly(lactic acid) Polymers 0.000 description 1
- 229920001610 polycaprolactone Polymers 0.000 description 1
- 239000004632 polycaprolactone Substances 0.000 description 1
- 229920002530 polyetherether ketone Polymers 0.000 description 1
- -1 polyethylene Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 230000001172 regenerating effect Effects 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- 210000005070 sphincter Anatomy 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 239000002470 thermal conductor Substances 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 210000003708 urethra Anatomy 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M5/00—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests
- A61M5/178—Syringes
- A61M5/31—Details
- A61M5/32—Needles; Details of needles pertaining to their connection with syringe or hub; Accessories for bringing the needle into, or holding the needle on, the body; Devices for protection of needles
- A61M5/34—Constructions for connecting the needle, e.g. to syringe nozzle or needle hub
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M5/00—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests
- A61M5/14—Infusion devices, e.g. infusing by gravity; Blood infusion; Accessories therefor
- A61M5/158—Needles for infusions; Accessories therefor, e.g. for inserting infusion needles, or for holding them on the body
- A61M5/1582—Double lumen needles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B18/18—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by applying electromagnetic radiation, e.g. microwaves
- A61B18/20—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by applying electromagnetic radiation, e.g. microwaves using laser
- A61B18/22—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by applying electromagnetic radiation, e.g. microwaves using laser the beam being directed along or through a flexible conduit, e.g. an optical fibre; Couplings or hand-pieces therefor
- A61B18/24—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by applying electromagnetic radiation, e.g. microwaves using laser the beam being directed along or through a flexible conduit, e.g. an optical fibre; Couplings or hand-pieces therefor with a catheter
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0067—Catheters; Hollow probes characterised by the distal end, e.g. tips
- A61M25/0068—Static characteristics of the catheter tip, e.g. shape, atraumatic tip, curved tip or tip structure
- A61M25/0069—Tip not integral with tube
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0067—Catheters; Hollow probes characterised by the distal end, e.g. tips
- A61M25/0068—Static characteristics of the catheter tip, e.g. shape, atraumatic tip, curved tip or tip structure
- A61M25/007—Side holes, e.g. their profiles or arrangements; Provisions to keep side holes unblocked
Description
細胞送達に先立って、使用者(例えば医師又は他の医学専門家)は細胞のための送達部位を選択する。例えば、患者が心臓発作を患っているのであれば施術者は梗塞帯域又はその付近の場所を選定することになろう。腹圧性尿失禁を患う患者については、施術者は横紋筋性括約筋(rhabdosphincter)又はその付近に標的場所を選定することになろう。
102 シリンジ
104 中空針
106 接続部分
108 第1操作部材
110 第2操作部材
112 プライミング部分
200 細胞送達システム
202 シリンジ
204 中空針
206 接続部材
208 第1操作部材
210 第2操作部材
212 プライミング部分
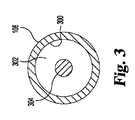
300 壁
302 中央ルーメン
304 先端制御部材
402 制御ルーメン
404 流体送達ルーメン
500 患者の身体
600 針
602 シース部分
604 中心キャリア部分
606 内面
608 空洞部
610 キャリアシャフト
612 ノーズ部分
614 連結部分
616 連結面
618 組織穿通部分
620 組織分離部分
622 プライミング部分
624 リップ又は縁部
626 長手方向軸線
800 針
802 シース部分
804 中心キャリア部分
806 内面
808 空洞部
810 キャリアシャフト
812 端領域
814 遠位領域
816 接点
818 組織穿通遠位先端
820 組織分離部分
822 プライミング部分
824 追従縁
826 長手方向軸線
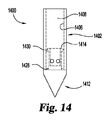
1400 針
1402 本体部分
1406 内面
1408 空洞部
1412 ノーズ部分
1414 面
1422 孔又はポート
1424 開口
1426 空洞部
1428 ストッパ部材
1430 捕捉部材
1600 針
1602 シース部分
1606 内面
1608 空洞部
1612 ノーズ部分
1622 ポート又は孔
1800 針
1802 シース部分
1806 内面
1808 第1空洞部
1810 中空シャフト部分
1811 第2空洞部
1814 放出領域
1822 シース部分のポート又は孔
1824 シャフト部分の孔
1826 長手方向軸線
2000 針
2002 シース部分
2004 中心キャリア部分
2006 内面
2007 外面
2008 第1空洞部
2009 第2空洞部
2010 シャフト部分
2011 シャフト空洞部
2022 プライミング部分
2024 孔
2032 シール
Claims (11)
- 細胞送達システムであって、
長手方向軸線に沿って延びる第1部分と、前記長手方向軸線に沿って延びる第2部分と、を備え、組織穿通に適合されている遠位先端で終端している細長い針本体、を備えており、
前記第1部分は前記長手方向軸線に沿って伸びる空洞部と開口した遠位端を画定するシース部分を有し、前記第2部分は前記シース部分内を前記長手方向軸線に沿って延びるシャフト部分と該シャフト部分の遠位に配置された遠位部分とを有し、前記遠位部分は前記シャフト部分の直径よりも大きくされて前記遠位先端を形成するようにされており、
前記針本体は、組織内への刺入のための第1形態から、前記第2部分を前記第1部分に対して遠位方向に変位させることにより、前記針本体の一部分が療法の所望される組織場所に隣接したときに細胞を送達するための第2形態へ移行でき、
前記針本体が前記第1形態にあるときには、前記第2部分の前記遠位部分が、前記シース部分の前記開口した遠位端を封止して前記シース部分の内周面と前記シャフト部分の外周面との間の前記空洞部に複数の細胞を保持し、前記遠位部分から前記シース部分の外面にかけて連続する滑らかな移行部が形成されるようになっており、
前記針本体が前記第2形態にあるときには、前記遠位部分が前記開口した遠位端から遠位に延びて、前記シース部分の前記開口した遠位端が開放されて前記空洞部内に保持されている細胞が療法の所望される組織場所に向けて送達されるようにされた、システム。 - 前記空洞部と流体連通しているルーメンを更に備えている、請求項1に記載のシステム。
- 前記第1部分と前記第2部分の一方又は両方が、前記第1形態から前記第2形態へ移行する際に前記長手方向軸線に沿って動く、請求項1又は請求項2に記載のシステム。
- 組織を穿通する前記遠位先端に隣接するプライミング部分を更に備えており、
前記プライミング部分は、当該プライミング部分に隣接する組織に、前記空洞部から前記組織への細胞到着に先立って治癒応答又は再生応答を生成するように配設されている、請求項1から請求項3の何れか一項に記載のシステム。 - 前記プライミング部分は研削面を備えている、請求項4に記載のシステム。
- 前記プライミング部分は少なくとも1つの流体射出ポートを備えている、請求項4又は請求項5の何れか一項に記載のシステム。
- 前記空洞部の内部に配置されている複数の細胞を更に備えている、請求項1から請求項6の何れか一項に記載のシステム。
- 細胞送達システムであって、
直線状のシャフト部分を有する中心キャリア部分、前記シャフト部分の遠位端に配置されたプライミング部分を有する遠位部分、及び開口した遠位端と細胞を送達するための空洞部を画定する内周面とを有するシース部分を備えていて、前記シャフト部分が前記シース部分の前記空洞部内を延びており、前記遠位部分の組織穿通部分で終端している細長い本体、を備えており、
前記本体が、前記遠位部分が前記開口した遠位端を封止して前記遠位部分から前記シース部分の外面にかけて連続する滑らかな移行部を形成する第1形態にあるときに、前記組織穿通部分は組織の中へ押し進められることで組織を穿通するように配設されており、
前記プライミング部分は、前記組織穿通部分に隣接して配置されていて、当該プライミング部分が療法の所望される組織場所に隣接したときに当該プライミング部分の隣接の組織に治癒応答又は再生応答を生成するように配設されており、
前記本体が、前記シャフト部分が遠位方向に押されて前記遠位部分が前記シャフト部分の前記開口した遠位端から遠位方向に延在して前記開口した遠位端を開放する第2形態にあるときに、前記空洞部が、複数の細胞を前記本体に隣接する組織へ送達するように配設されている、システム。 - 前記空洞部の内部に配置されている複数の細胞を更に備えている請求項8に記載のシステム。
- 前記プライミング部分は研削面を備えている、請求項8又は請求項9に記載のシステム。
- 前記プライミング部分は少なくとも1つの流体射出ポートを備えている、請求項8から請求項10の何れか一項に記載のシステム。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361789518P | 2013-03-15 | 2013-03-15 | |
| US61/789,518 | 2013-03-15 | ||
| PCT/US2014/021699 WO2014149977A1 (en) | 2013-03-15 | 2014-03-07 | Cell injection needle |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018220197A Division JP2019048157A (ja) | 2013-03-15 | 2018-11-26 | 細胞注入針 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2016515004A JP2016515004A (ja) | 2016-05-26 |
| JP2016515004A5 JP2016515004A5 (ja) | 2017-02-09 |
| JP6483655B2 true JP6483655B2 (ja) | 2019-03-13 |
Family
ID=51530681
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016500811A Active JP6483655B2 (ja) | 2013-03-15 | 2014-03-07 | 細胞注入針 |
| JP2018220197A Pending JP2019048157A (ja) | 2013-03-15 | 2018-11-26 | 細胞注入針 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018220197A Pending JP2019048157A (ja) | 2013-03-15 | 2018-11-26 | 細胞注入針 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US10328213B2 (ja) |
| EP (1) | EP2968752B1 (ja) |
| JP (2) | JP6483655B2 (ja) |
| WO (1) | WO2014149977A1 (ja) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160287795A1 (en) * | 2015-04-02 | 2016-10-06 | XEND Medical, LLC | Method of using a hypodermic needle system |
| CN110382020A (zh) * | 2016-12-28 | 2019-10-25 | 桑比欧公司 | 细胞递送系统及操作细胞递送系统的方法 |
| JP2024040532A (ja) * | 2021-02-08 | 2024-03-26 | テルモ株式会社 | 医療器具 |
Family Cites Families (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4698056A (en) * | 1986-03-21 | 1987-10-06 | Medi-Tech, Inc. | Enteric feeding device |
| JP2702086B2 (ja) | 1995-02-13 | 1998-01-21 | 株式会社湘南合成樹脂製作所 | 管ライニング材の製造方法 |
| US6120520A (en) | 1997-05-27 | 2000-09-19 | Angiotrax, Inc. | Apparatus and methods for stimulating revascularization and/or tissue growth |
| US6332877B1 (en) | 1998-05-12 | 2001-12-25 | Novartis Ag | Ostomy tube placement tip |
| US6432119B1 (en) | 1999-03-17 | 2002-08-13 | Angiotrax, Inc. | Apparatus and methods for performing percutaneous myocardial revascularization and stimulating angiogenesis using autologous materials |
| US6689103B1 (en) * | 1999-05-07 | 2004-02-10 | Scimed Life System, Inc. | Injection array apparatus and method |
| US6344027B1 (en) | 1999-12-08 | 2002-02-05 | Scimed Life Systems, Inc. | Needle-less injection apparatus and method |
| US7399304B2 (en) * | 2000-03-03 | 2008-07-15 | C.R. Bard, Inc. | Endoscopic tissue apposition device with multiple suction ports |
| US6549803B1 (en) | 2000-05-08 | 2003-04-15 | Image-Guided Neurologics Inc. | Method and apparatus for targeting material delivery to tissue |
| US6464662B1 (en) | 2000-07-26 | 2002-10-15 | Image-Guided Neurologics, Inc. | Drug delivery and catheter systems, apparatus and processes |
| US6572579B1 (en) | 2000-09-13 | 2003-06-03 | Image-Guided Neurologics, Inc. | Drug delivery and catheter systems, apparatus and processes |
| US6425854B1 (en) | 2000-10-02 | 2002-07-30 | Genyx Medical, Inc. | Method for delivering non-biodegradable bulking composition to a urological site |
| US6478775B1 (en) | 2000-10-02 | 2002-11-12 | Genyx Medical Inc. | Device for delivering non-biodegradable bulking composition to a urological site |
| US6602241B2 (en) * | 2001-01-17 | 2003-08-05 | Transvascular, Inc. | Methods and apparatus for acute or chronic delivery of substances or apparatus to extravascular treatment sites |
| WO2002062231A2 (en) * | 2001-02-05 | 2002-08-15 | Tyco Healthcare Group Lp | Biopsy apparatus and method |
| US6544220B2 (en) | 2001-02-14 | 2003-04-08 | Scimed Life Systems, Inc. | Fluid jet PMR |
| US6969373B2 (en) * | 2001-04-13 | 2005-11-29 | Tricardia, Llc | Syringe system |
| US6749833B2 (en) | 2001-05-25 | 2004-06-15 | Bresagen Limited | Design and planning system for improving the survival rate of injected structures |
| US7674250B2 (en) | 2002-08-05 | 2010-03-09 | Boston Scientific Scimed, Inc. | Methods of delivering therapeutic agents |
| CA2506033A1 (en) * | 2002-11-25 | 2004-06-10 | Boston Scientific Limited | Injection device, screw-type syringe for the use therewith, and method for treating mammalian body |
| US7481798B2 (en) * | 2003-03-20 | 2009-01-27 | Boston Scientific Scimed, Inc. | Devices and methods for delivering therapeutic or diagnostic agents |
| US8512290B2 (en) * | 2003-03-20 | 2013-08-20 | Boston Scientific Scimed, Inc. | Devices and methods for delivering therapeutic or diagnostic agents |
| US20050070874A1 (en) | 2003-09-30 | 2005-03-31 | Hikaru Matsuda | System and method for injecting liquid drug containing biological material |
| US7273469B1 (en) * | 2003-12-31 | 2007-09-25 | Advanced Cardiovascular Systems, Inc. | Modified needle catheter for directional orientation delivery |
| US7632262B2 (en) | 2004-07-19 | 2009-12-15 | Nexeon Medical Systems, Inc. | Systems and methods for atraumatic implantation of bio-active agents |
| WO2006023203A1 (en) * | 2004-08-19 | 2006-03-02 | Vein Rx, Inc. | An occludable intravascular catheter for drug delivery and method of using the same |
| JP2008519659A (ja) | 2004-11-12 | 2008-06-12 | リージェンツ オブ ザ ユニバーシティ オブ ミネソタ | 静脈閉塞装置および使用法 |
| US7651482B2 (en) * | 2005-02-04 | 2010-01-26 | Boston Scientific Scimed, Inc. | Non-coring needles and methods of manufacturing same |
| US20100280493A1 (en) | 2005-06-23 | 2010-11-04 | Asha Nayak | Methods and Systems for Treating Injured Cardiac Tissue |
| US8182444B2 (en) | 2005-11-04 | 2012-05-22 | Medrad, Inc. | Delivery of agents such as cells to tissue |
| US20090312696A1 (en) * | 2005-12-28 | 2009-12-17 | Copa Vincent G | Devices, Systems, and Related Methods for Delivery of Fluid to Tissue |
| US20080039776A1 (en) | 2006-08-08 | 2008-02-14 | Ragae Ghabrial | Devices for endoscopic therapeutic cell delivery |
| US20080086111A1 (en) | 2006-10-09 | 2008-04-10 | Medrad, Inc. | Fluid delivery systems and volume metering in cell delivery |
| WO2009055640A1 (en) * | 2007-10-25 | 2009-04-30 | Epitome Pharmaceuticals Limited | Tissue splitting biopsy needle |
| US8784800B2 (en) | 2009-03-09 | 2014-07-22 | Medtronic, Inc. | Method of delivering cell therapy to a target site |
| US20110014181A1 (en) | 2009-07-20 | 2011-01-20 | Medtronic Vascular, Inc. | Microneedle Delivery Device and Methods of Using Same |
| US8926552B2 (en) * | 2009-08-12 | 2015-01-06 | Medtronic, Inc. | Particle delivery |
-
2014
- 2014-03-07 WO PCT/US2014/021699 patent/WO2014149977A1/en active Application Filing
- 2014-03-07 JP JP2016500811A patent/JP6483655B2/ja active Active
- 2014-03-07 EP EP14767516.9A patent/EP2968752B1/en active Active
- 2014-03-14 US US14/211,702 patent/US10328213B2/en active Active
-
2018
- 2018-11-26 JP JP2018220197A patent/JP2019048157A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| US10328213B2 (en) | 2019-06-25 |
| EP2968752A1 (en) | 2016-01-20 |
| JP2016515004A (ja) | 2016-05-26 |
| JP2019048157A (ja) | 2019-03-28 |
| US20140276368A1 (en) | 2014-09-18 |
| EP2968752B1 (en) | 2023-09-06 |
| EP2968752A4 (en) | 2017-03-22 |
| WO2014149977A1 (en) | 2014-09-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4282898B2 (ja) | 心筋の血液潅流を増大させるための装置 | |
| US8012096B2 (en) | Surgical device and method for performing combination revascularization and therapeutic substance delivery to tissue | |
| JP3712668B2 (ja) | 生体の目標組織に作用物質を送達するためのシステム | |
| US7776025B2 (en) | Method for providing medicament to tissue | |
| JP4846166B2 (ja) | インプラント供給カテーテルシステム | |
| US6152141A (en) | Method for delivery of therapeutic agents to the heart | |
| JP5072850B2 (ja) | 埋め込み型注入装置 | |
| JP2019048157A (ja) | 細胞注入針 | |
| WO2020232849A1 (zh) | 一种可协同给药的电脉冲消融装置 | |
| EP1188417A2 (en) | Method and apparatus for delivery of therapeutic agents to the heart | |
| EP1383551B1 (en) | Multilumen catheter and implants for the delivery of therapeutic agents to tissues | |
| AU782484B2 (en) | Methods and apparatus for delivering medicament to tissue | |
| KR102576604B1 (ko) | 의료용 정밀 약물주입 및 전기천공 융합 전극 | |
| EP3254723A1 (en) | Catheter for delivering a therapeutic agent into a substrate | |
| KR20240008935A (ko) | 표적 치료제를 전달하는 루멘 병렬 스텐트 | |
| JP2012005537A (ja) | カテーテル |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170105 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20170105 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20171020 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20171107 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20180207 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20180409 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20180724 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20181126 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20181217 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20190115 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20190214 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6483655 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |