JP6133785B2 - Curable resin composition for coating and automotive clear coating agent - Google Patents
Curable resin composition for coating and automotive clear coating agent Download PDFInfo
- Publication number
- JP6133785B2 JP6133785B2 JP2013547194A JP2013547194A JP6133785B2 JP 6133785 B2 JP6133785 B2 JP 6133785B2 JP 2013547194 A JP2013547194 A JP 2013547194A JP 2013547194 A JP2013547194 A JP 2013547194A JP 6133785 B2 JP6133785 B2 JP 6133785B2
- Authority
- JP
- Japan
- Prior art keywords
- group
- resin composition
- curable resin
- coating
- meth
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000011248 coating agent Substances 0.000 title claims description 86
- 238000000576 coating method Methods 0.000 title claims description 78
- 239000011342 resin composition Substances 0.000 title claims description 38
- 239000000178 monomer Substances 0.000 claims description 50
- -1 tertiary amine compound Chemical class 0.000 claims description 36
- 229920002554 vinyl polymer Polymers 0.000 claims description 34
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 33
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 32
- 125000004432 carbon atom Chemical group C* 0.000 claims description 28
- 229920006163 vinyl copolymer Polymers 0.000 claims description 26
- 150000001875 compounds Chemical class 0.000 claims description 20
- 125000000217 alkyl group Chemical group 0.000 claims description 15
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 15
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 12
- 239000003054 catalyst Substances 0.000 claims description 12
- 229910052782 aluminium Inorganic materials 0.000 claims description 9
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims description 9
- 229920001577 copolymer Polymers 0.000 claims description 9
- 239000012948 isocyanate Substances 0.000 claims description 9
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 claims description 9
- 239000011203 carbon fibre reinforced carbon Substances 0.000 claims description 8
- 150000002513 isocyanates Chemical class 0.000 claims description 7
- 125000003118 aryl group Chemical group 0.000 claims description 6
- 150000003606 tin compounds Chemical class 0.000 claims description 6
- 125000003647 acryloyl group Chemical group O=C([*])C([H])=C([H])[H] 0.000 claims description 5
- 125000002947 alkylene group Chemical group 0.000 claims description 5
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 5
- 230000009477 glass transition Effects 0.000 claims description 5
- 229920000642 polymer Polymers 0.000 claims description 5
- 239000013522 chelant Substances 0.000 claims description 4
- 229920006243 acrylic copolymer Polymers 0.000 claims description 3
- 125000001183 hydrocarbyl group Chemical group 0.000 claims 1
- 239000000126 substance Substances 0.000 description 37
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 33
- 238000001723 curing Methods 0.000 description 32
- 239000002904 solvent Substances 0.000 description 25
- 239000002253 acid Substances 0.000 description 17
- 238000000034 method Methods 0.000 description 17
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 17
- 239000003973 paint Substances 0.000 description 16
- 239000005056 polyisocyanate Substances 0.000 description 14
- 229920001228 polyisocyanate Polymers 0.000 description 14
- 239000000203 mixture Substances 0.000 description 12
- 238000011156 evaluation Methods 0.000 description 9
- 239000007788 liquid Substances 0.000 description 9
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 7
- AYOHIQLKSOJJQH-UHFFFAOYSA-N dibutyltin Chemical compound CCCC[Sn]CCCC AYOHIQLKSOJJQH-UHFFFAOYSA-N 0.000 description 7
- 229920003023 plastic Polymers 0.000 description 7
- 239000004033 plastic Substances 0.000 description 7
- 239000000047 product Substances 0.000 description 7
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 6
- 238000004132 cross linking Methods 0.000 description 6
- 229910052757 nitrogen Inorganic materials 0.000 description 6
- 230000000704 physical effect Effects 0.000 description 6
- 238000006116 polymerization reaction Methods 0.000 description 6
- 150000003839 salts Chemical class 0.000 description 6
- 229920000877 Melamine resin Polymers 0.000 description 5
- 239000003795 chemical substances by application Substances 0.000 description 5
- 239000012024 dehydrating agents Substances 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 238000002156 mixing Methods 0.000 description 5
- 229920005989 resin Polymers 0.000 description 5
- 239000011347 resin Substances 0.000 description 5
- 238000003860 storage Methods 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 4
- 229910000831 Steel Inorganic materials 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 4
- 238000009472 formulation Methods 0.000 description 4
- JDSHMPZPIAZGSV-UHFFFAOYSA-N melamine Chemical compound NC1=NC(N)=NC(N)=N1 JDSHMPZPIAZGSV-UHFFFAOYSA-N 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 239000000049 pigment Substances 0.000 description 4
- 229920000515 polycarbonate Polymers 0.000 description 4
- 239000004417 polycarbonate Substances 0.000 description 4
- 239000004814 polyurethane Substances 0.000 description 4
- 229920002635 polyurethane Polymers 0.000 description 4
- 238000005507 spraying Methods 0.000 description 4
- 239000010959 steel Substances 0.000 description 4
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 3
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 3
- 241001134446 Niveas Species 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 229910052783 alkali metal Inorganic materials 0.000 description 3
- 150000003863 ammonium salts Chemical class 0.000 description 3
- 239000004566 building material Substances 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 230000000052 comparative effect Effects 0.000 description 3
- 238000011109 contamination Methods 0.000 description 3
- 239000003431 cross linking reagent Substances 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- HGQSXVKHVMGQRG-UHFFFAOYSA-N dioctyltin Chemical compound CCCCCCCC[Sn]CCCCCCCC HGQSXVKHVMGQRG-UHFFFAOYSA-N 0.000 description 3
- 150000002009 diols Chemical class 0.000 description 3
- 150000002148 esters Chemical class 0.000 description 3
- XPFVYQJUAUNWIW-UHFFFAOYSA-N furfuryl alcohol Chemical compound OCC1=CC=CO1 XPFVYQJUAUNWIW-UHFFFAOYSA-N 0.000 description 3
- 150000002430 hydrocarbons Chemical group 0.000 description 3
- 239000003999 initiator Substances 0.000 description 3
- 230000009257 reactivity Effects 0.000 description 3
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 2
- JWAZRIHNYRIHIV-UHFFFAOYSA-N 2-naphthol Chemical compound C1=CC=CC2=CC(O)=CC=C21 JWAZRIHNYRIHIV-UHFFFAOYSA-N 0.000 description 2
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 2
- QOXOZONBQWIKDA-UHFFFAOYSA-N 3-hydroxypropyl Chemical group [CH2]CCO QOXOZONBQWIKDA-UHFFFAOYSA-N 0.000 description 2
- XDQWJFXZTAWJST-UHFFFAOYSA-N 3-triethoxysilylpropyl prop-2-enoate Chemical compound CCO[Si](OCC)(OCC)CCCOC(=O)C=C XDQWJFXZTAWJST-UHFFFAOYSA-N 0.000 description 2
- SXIFAEWFOJETOA-UHFFFAOYSA-N 4-hydroxy-butyl Chemical group [CH2]CCCO SXIFAEWFOJETOA-UHFFFAOYSA-N 0.000 description 2
- FUGYGGDSWSUORM-UHFFFAOYSA-N 4-hydroxystyrene Chemical compound OC1=CC=C(C=C)C=C1 FUGYGGDSWSUORM-UHFFFAOYSA-N 0.000 description 2
- 239000004923 Acrylic lacquer Substances 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical compound C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 2
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- 239000007983 Tris buffer Substances 0.000 description 2
- ORLQHILJRHBSAY-UHFFFAOYSA-N [1-(hydroxymethyl)cyclohexyl]methanol Chemical compound OCC1(CO)CCCCC1 ORLQHILJRHBSAY-UHFFFAOYSA-N 0.000 description 2
- RMKZLFMHXZAGTM-UHFFFAOYSA-N [dimethoxy(propyl)silyl]oxymethyl prop-2-enoate Chemical compound CCC[Si](OC)(OC)OCOC(=O)C=C RMKZLFMHXZAGTM-UHFFFAOYSA-N 0.000 description 2
- 229920000122 acrylonitrile butadiene styrene Polymers 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 230000000996 additive effect Effects 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 2
- 125000003277 amino group Chemical group 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- 239000002981 blocking agent Substances 0.000 description 2
- BTANRVKWQNVYAZ-UHFFFAOYSA-N butan-2-ol Chemical compound CCC(C)O BTANRVKWQNVYAZ-UHFFFAOYSA-N 0.000 description 2
- 150000001735 carboxylic acids Chemical class 0.000 description 2
- 239000012986 chain transfer agent Substances 0.000 description 2
- 239000002131 composite material Substances 0.000 description 2
- 238000013329 compounding Methods 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 229910001873 dinitrogen Inorganic materials 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 229920001971 elastomer Polymers 0.000 description 2
- 235000019441 ethanol Nutrition 0.000 description 2
- XYIBRDXRRQCHLP-UHFFFAOYSA-N ethyl acetoacetate Chemical compound CCOC(=O)CC(C)=O XYIBRDXRRQCHLP-UHFFFAOYSA-N 0.000 description 2
- 229940093858 ethyl acetoacetate Drugs 0.000 description 2
- 238000010528 free radical solution polymerization reaction Methods 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 150000002576 ketones Chemical class 0.000 description 2
- RLSSMJSEOOYNOY-UHFFFAOYSA-N m-cresol Chemical compound CC1=CC=CC(O)=C1 RLSSMJSEOOYNOY-UHFFFAOYSA-N 0.000 description 2
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 2
- QWVGKYWNOKOFNN-UHFFFAOYSA-N o-cresol Chemical compound CC1=CC=CC=C1O QWVGKYWNOKOFNN-UHFFFAOYSA-N 0.000 description 2
- IWDCLRJOBJJRNH-UHFFFAOYSA-N p-cresol Chemical compound CC1=CC=C(O)C=C1 IWDCLRJOBJJRNH-UHFFFAOYSA-N 0.000 description 2
- 238000010422 painting Methods 0.000 description 2
- 150000002978 peroxides Chemical class 0.000 description 2
- 229920005668 polycarbonate resin Polymers 0.000 description 2
- 239000004431 polycarbonate resin Substances 0.000 description 2
- 229920000728 polyester Chemical class 0.000 description 2
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 2
- KIDHWZJUCRJVML-UHFFFAOYSA-N putrescine Chemical compound NCCCCN KIDHWZJUCRJVML-UHFFFAOYSA-N 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 125000004434 sulfur atom Chemical group 0.000 description 2
- 238000004381 surface treatment Methods 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 229920002803 thermoplastic polyurethane Polymers 0.000 description 2
- 229920001187 thermosetting polymer Polymers 0.000 description 2
- MGSRCZKZVOBKFT-UHFFFAOYSA-N thymol Chemical compound CC(C)C1=CC=C(C)C=C1O MGSRCZKZVOBKFT-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- PYOKUURKVVELLB-UHFFFAOYSA-N trimethyl orthoformate Chemical compound COC(OC)OC PYOKUURKVVELLB-UHFFFAOYSA-N 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 239000008096 xylene Substances 0.000 description 2
- DTGKSKDOIYIVQL-WEDXCCLWSA-N (+)-borneol Chemical group C1C[C@@]2(C)[C@@H](O)C[C@@H]1C2(C)C DTGKSKDOIYIVQL-WEDXCCLWSA-N 0.000 description 1
- QGKMIGUHVLGJBR-UHFFFAOYSA-M (4z)-1-(3-methylbutyl)-4-[[1-(3-methylbutyl)quinolin-1-ium-4-yl]methylidene]quinoline;iodide Chemical compound [I-].C12=CC=CC=C2N(CCC(C)C)C=CC1=CC1=CC=[N+](CCC(C)C)C2=CC=CC=C12 QGKMIGUHVLGJBR-UHFFFAOYSA-M 0.000 description 1
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 description 1
- NDQXKKFRNOPRDW-UHFFFAOYSA-N 1,1,1-triethoxyethane Chemical compound CCOC(C)(OCC)OCC NDQXKKFRNOPRDW-UHFFFAOYSA-N 0.000 description 1
- HDPNBNXLBDFELL-UHFFFAOYSA-N 1,1,1-trimethoxyethane Chemical compound COC(C)(OC)OC HDPNBNXLBDFELL-UHFFFAOYSA-N 0.000 description 1
- JWTGRKUQJXIWCV-UHFFFAOYSA-N 1,2,3-trihydroxypropyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OC(O)C(O)CO JWTGRKUQJXIWCV-UHFFFAOYSA-N 0.000 description 1
- DNPRCPUUZUVBMY-UHFFFAOYSA-N 1,4-diethyl-2-methylpiperazine Chemical compound CCN1CCN(CC)C(C)C1 DNPRCPUUZUVBMY-UHFFFAOYSA-N 0.000 description 1
- WFSQCDSLKHKXTP-UHFFFAOYSA-N 1-butoxyethane-1,1-diol Chemical compound CCCCOC(C)(O)O WFSQCDSLKHKXTP-UHFFFAOYSA-N 0.000 description 1
- OZFIGURLAJSLIR-UHFFFAOYSA-N 1-ethenyl-2h-pyridine Chemical compound C=CN1CC=CC=C1 OZFIGURLAJSLIR-UHFFFAOYSA-N 0.000 description 1
- OSSNTDFYBPYIEC-UHFFFAOYSA-N 1-ethenylimidazole Chemical compound C=CN1C=CN=C1 OSSNTDFYBPYIEC-UHFFFAOYSA-N 0.000 description 1
- XLPJNCYCZORXHG-UHFFFAOYSA-N 1-morpholin-4-ylprop-2-en-1-one Chemical compound C=CC(=O)N1CCOCC1 XLPJNCYCZORXHG-UHFFFAOYSA-N 0.000 description 1
- YAJYJWXEWKRTPO-UHFFFAOYSA-N 2,3,3,4,4,5-hexamethylhexane-2-thiol Chemical compound CC(C)C(C)(C)C(C)(C)C(C)(C)S YAJYJWXEWKRTPO-UHFFFAOYSA-N 0.000 description 1
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 1
- JAHNSTQSQJOJLO-UHFFFAOYSA-N 2-(3-fluorophenyl)-1h-imidazole Chemical compound FC1=CC=CC(C=2NC=CN=2)=C1 JAHNSTQSQJOJLO-UHFFFAOYSA-N 0.000 description 1
- OEPOKWHJYJXUGD-UHFFFAOYSA-N 2-(3-phenylmethoxyphenyl)-1,3-thiazole-4-carbaldehyde Chemical compound O=CC1=CSC(C=2C=C(OCC=3C=CC=CC=3)C=CC=2)=N1 OEPOKWHJYJXUGD-UHFFFAOYSA-N 0.000 description 1
- BUVLLMREBRZNHQ-UHFFFAOYSA-N 2-(diethylamino)ethanol;hexanedioic acid Chemical compound CCN(CC)CCO.CCN(CC)CCO.OC(=O)CCCCC(O)=O BUVLLMREBRZNHQ-UHFFFAOYSA-N 0.000 description 1
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 1
- WVEOWVCCOMZTBL-UHFFFAOYSA-N 2-[di(propan-2-yloxy)-propylsilyl]oxypropan-2-yl prop-2-enoate Chemical compound CCC[Si](OC(C)C)(OC(C)C)OC(C)(C)OC(=O)C=C WVEOWVCCOMZTBL-UHFFFAOYSA-N 0.000 description 1
- MMGVVYCBXBYXRR-UHFFFAOYSA-L 2-acetyl-3-oxobutanoate;dibutyltin(2+) Chemical compound CCCC[Sn+2]CCCC.CC(=O)C(C(C)=O)C([O-])=O.CC(=O)C(C(C)=O)C([O-])=O MMGVVYCBXBYXRR-UHFFFAOYSA-L 0.000 description 1
- POAOYUHQDCAZBD-UHFFFAOYSA-N 2-butoxyethanol Chemical compound CCCCOCCO POAOYUHQDCAZBD-UHFFFAOYSA-N 0.000 description 1
- SBYMUDUGTIKLCR-UHFFFAOYSA-N 2-chloroethenylbenzene Chemical compound ClC=CC1=CC=CC=C1 SBYMUDUGTIKLCR-UHFFFAOYSA-N 0.000 description 1
- VUIWJRYTWUGOOF-UHFFFAOYSA-N 2-ethenoxyethanol Chemical compound OCCOC=C VUIWJRYTWUGOOF-UHFFFAOYSA-N 0.000 description 1
- ZNQVEEAIQZEUHB-UHFFFAOYSA-N 2-ethoxyethanol Chemical compound CCOCCO ZNQVEEAIQZEUHB-UHFFFAOYSA-N 0.000 description 1
- AGBXYHCHUYARJY-UHFFFAOYSA-N 2-phenylethenesulfonic acid Chemical compound OS(=O)(=O)C=CC1=CC=CC=C1 AGBXYHCHUYARJY-UHFFFAOYSA-N 0.000 description 1
- UDXXYUDJOHIIDZ-UHFFFAOYSA-N 2-phosphonooxyethyl prop-2-enoate Chemical class OP(O)(=O)OCCOC(=O)C=C UDXXYUDJOHIIDZ-UHFFFAOYSA-N 0.000 description 1
- GQTFHSAAODFMHB-UHFFFAOYSA-N 2-prop-2-enoyloxyethanesulfonic acid Chemical compound OS(=O)(=O)CCOC(=O)C=C GQTFHSAAODFMHB-UHFFFAOYSA-N 0.000 description 1
- FRIBMENBGGCKPD-UHFFFAOYSA-N 3-(2,3-dimethoxyphenyl)prop-2-enal Chemical compound COC1=CC=CC(C=CC=O)=C1OC FRIBMENBGGCKPD-UHFFFAOYSA-N 0.000 description 1
- UDWIZRDPCQAYRF-UHFFFAOYSA-N 3-[diethoxy(methyl)silyl]propyl prop-2-enoate Chemical compound CCO[Si](C)(OCC)CCCOC(=O)C=C UDWIZRDPCQAYRF-UHFFFAOYSA-N 0.000 description 1
- JMTRBTMJIYBHIV-UHFFFAOYSA-N 3-[dimethoxy(2-phenylethoxy)silyl]-n-ethenylpropan-1-amine Chemical compound C=CNCCC[Si](OC)(OC)OCCC1=CC=CC=C1 JMTRBTMJIYBHIV-UHFFFAOYSA-N 0.000 description 1
- MCDBEBOBROAQSH-UHFFFAOYSA-N 3-[dimethoxy(methyl)silyl]propyl prop-2-enoate Chemical compound CO[Si](C)(OC)CCCOC(=O)C=C MCDBEBOBROAQSH-UHFFFAOYSA-N 0.000 description 1
- DGBFOBNYTYHFPN-UHFFFAOYSA-N 3-[ethoxy(dimethyl)silyl]propyl prop-2-enoate Chemical compound CCO[Si](C)(C)CCCOC(=O)C=C DGBFOBNYTYHFPN-UHFFFAOYSA-N 0.000 description 1
- ZCRUJAKCJLCJCP-UHFFFAOYSA-N 3-[methoxy(dimethyl)silyl]propyl prop-2-enoate Chemical compound CO[Si](C)(C)CCCOC(=O)C=C ZCRUJAKCJLCJCP-UHFFFAOYSA-N 0.000 description 1
- PNZVYZIRTOVNKZ-UHFFFAOYSA-N 3-[tris(2-methoxyethoxy)silyl]propyl prop-2-enoate Chemical compound COCCO[Si](OCCOC)(OCCOC)CCCOC(=O)C=C PNZVYZIRTOVNKZ-UHFFFAOYSA-N 0.000 description 1
- OFNISBHGPNMTMS-UHFFFAOYSA-N 3-methylideneoxolane-2,5-dione Chemical compound C=C1CC(=O)OC1=O OFNISBHGPNMTMS-UHFFFAOYSA-N 0.000 description 1
- UUEWCQRISZBELL-UHFFFAOYSA-N 3-trimethoxysilylpropane-1-thiol Chemical compound CO[Si](OC)(OC)CCCS UUEWCQRISZBELL-UHFFFAOYSA-N 0.000 description 1
- UPMLOUAZCHDJJD-UHFFFAOYSA-N 4,4'-Diphenylmethane Diisocyanate Chemical compound C1=CC(N=C=O)=CC=C1CC1=CC=C(N=C=O)C=C1 UPMLOUAZCHDJJD-UHFFFAOYSA-N 0.000 description 1
- LIEWIHQESDHHTI-UHFFFAOYSA-N 4-butoxy-3-hydroxy-4-oxobutanoic acid Chemical compound CCCCOC(=O)C(O)CC(O)=O LIEWIHQESDHHTI-UHFFFAOYSA-N 0.000 description 1
- KYQMQSJMTYTXLD-UHFFFAOYSA-N 4-ethenylphenol;prop-2-enylbenzene Chemical compound C=CCC1=CC=CC=C1.OC1=CC=C(C=C)C=C1 KYQMQSJMTYTXLD-UHFFFAOYSA-N 0.000 description 1
- NDWUBGAGUCISDV-UHFFFAOYSA-N 4-hydroxybutyl prop-2-enoate Chemical compound OCCCCOC(=O)C=C NDWUBGAGUCISDV-UHFFFAOYSA-N 0.000 description 1
- BTJIUGUIPKRLHP-UHFFFAOYSA-N 4-nitrophenol Chemical compound OC1=CC=C([N+]([O-])=O)C=C1 BTJIUGUIPKRLHP-UHFFFAOYSA-N 0.000 description 1
- QHPQWRBYOIRBIT-UHFFFAOYSA-N 4-tert-butylphenol Chemical compound CC(C)(C)C1=CC=C(O)C=C1 QHPQWRBYOIRBIT-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- NLHHRLWOUZZQLW-UHFFFAOYSA-N Acrylonitrile Chemical compound C=CC#N NLHHRLWOUZZQLW-UHFFFAOYSA-N 0.000 description 1
- 102100040440 Adenylate kinase isoenzyme 5 Human genes 0.000 description 1
- 101710168711 Adenylate kinase isoenzyme 5 Proteins 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 229910001369 Brass Inorganic materials 0.000 description 1
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 1
- CTRSDHVBJVYXSL-UHFFFAOYSA-J C(CCCCCCCCCCC)(=[O+][O-])[O-].[Sn+4].C(CCCCCCCCCCC)(=[O+][O-])[O-].C(CCCCCCCCCCC)(=[O+][O-])[O-].C(CCCCCCCCCCC)(=[O+][O-])[O-] Chemical group C(CCCCCCCCCCC)(=[O+][O-])[O-].[Sn+4].C(CCCCCCCCCCC)(=[O+][O-])[O-].C(CCCCCCCCCCC)(=[O+][O-])[O-].C(CCCCCCCCCCC)(=[O+][O-])[O-] CTRSDHVBJVYXSL-UHFFFAOYSA-J 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- SJIXRGNQPBQWMK-UHFFFAOYSA-N DEAEMA Natural products CCN(CC)CCOC(=O)C(C)=C SJIXRGNQPBQWMK-UHFFFAOYSA-N 0.000 description 1
- 239000004641 Diallyl-phthalate Substances 0.000 description 1
- 239000004593 Epoxy Substances 0.000 description 1
- 239000005057 Hexamethylene diisocyanate Substances 0.000 description 1
- 239000005058 Isophorone diisocyanate Substances 0.000 description 1
- 229910001209 Low-carbon steel Inorganic materials 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 1
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 1
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 1
- SVYKKECYCPFKGB-UHFFFAOYSA-N N,N-dimethylcyclohexylamine Chemical compound CN(C)C1CCCCC1 SVYKKECYCPFKGB-UHFFFAOYSA-N 0.000 description 1
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- 239000005062 Polybutadiene Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 208000034189 Sclerosis Diseases 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- BOTDANWDWHJENH-UHFFFAOYSA-N Tetraethyl orthosilicate Chemical compound CCO[Si](OCC)(OCC)OCC BOTDANWDWHJENH-UHFFFAOYSA-N 0.000 description 1
- 239000005844 Thymol Substances 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 238000003848 UV Light-Curing Methods 0.000 description 1
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 1
- UNIWFCPQNWBLKG-UHFFFAOYSA-L [[9-methyl-2-(7-methyloctyl)-2-(sulfanylmethyl)decanoyl]oxy-dioctylstannyl] 9-methyl-2-(7-methyloctyl)-2-(sulfanylmethyl)decanoate Chemical compound C(CCCCCC(C)C)C(C(=O)[O-])(CS)CCCCCCC(C)C.C(CCCCCCC)[Sn+2]CCCCCCCC.C(CCCCCC(C)C)C(C(=O)[O-])(CS)CCCCCCC(C)C UNIWFCPQNWBLKG-UHFFFAOYSA-L 0.000 description 1
- NOZAQBYNLKNDRT-UHFFFAOYSA-N [diacetyloxy(ethenyl)silyl] acetate Chemical compound CC(=O)O[Si](OC(C)=O)(OC(C)=O)C=C NOZAQBYNLKNDRT-UHFFFAOYSA-N 0.000 description 1
- UKLDJPRMSDWDSL-UHFFFAOYSA-L [dibutyl(dodecanoyloxy)stannyl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCC UKLDJPRMSDWDSL-UHFFFAOYSA-L 0.000 description 1
- YVHDRFKHKGNLNW-UHFFFAOYSA-L [dibutyl(octadecanoyloxy)stannyl] octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCCCCCCCC YVHDRFKHKGNLNW-UHFFFAOYSA-L 0.000 description 1
- NBJODVYWAQLZOC-UHFFFAOYSA-L [dibutyl(octanoyloxy)stannyl] octanoate Chemical compound CCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCC NBJODVYWAQLZOC-UHFFFAOYSA-L 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- WFDIJRYMOXRFFG-UHFFFAOYSA-N acetic anhydride Substances CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 1
- WFDIJRYMOXRFFG-XPULMUKRSA-N acetyl acetate Chemical compound [14CH3]C(=O)OC([14CH3])=O WFDIJRYMOXRFFG-XPULMUKRSA-N 0.000 description 1
- 150000008065 acid anhydrides Chemical class 0.000 description 1
- 238000003916 acid precipitation Methods 0.000 description 1
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 1
- 239000004676 acrylonitrile butadiene styrene Substances 0.000 description 1
- 239000005456 alcohol based solvent Substances 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 150000007824 aliphatic compounds Chemical class 0.000 description 1
- 125000005370 alkoxysilyl group Chemical group 0.000 description 1
- 229920000180 alkyd Polymers 0.000 description 1
- XYLMUPLGERFSHI-UHFFFAOYSA-N alpha-Methylstyrene Chemical compound CC(=C)C1=CC=CC=C1 XYLMUPLGERFSHI-UHFFFAOYSA-N 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 239000002518 antifoaming agent Substances 0.000 description 1
- 150000001491 aromatic compounds Chemical class 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- AYJRCSIUFZENHW-DEQYMQKBSA-L barium(2+);oxomethanediolate Chemical compound [Ba+2].[O-][14C]([O-])=O AYJRCSIUFZENHW-DEQYMQKBSA-L 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- QUDWYFHPNIMBFC-UHFFFAOYSA-N bis(prop-2-enyl) benzene-1,2-dicarboxylate Chemical compound C=CCOC(=O)C1=CC=CC=C1C(=O)OCC=C QUDWYFHPNIMBFC-UHFFFAOYSA-N 0.000 description 1
- 239000010951 brass Substances 0.000 description 1
- 238000012662 bulk polymerization Methods 0.000 description 1
- QVYARBLCAHCSFJ-UHFFFAOYSA-N butane-1,1-diamine Chemical compound CCCC(N)N QVYARBLCAHCSFJ-UHFFFAOYSA-N 0.000 description 1
- SDUJCTKTKOLVFG-UHFFFAOYSA-N butoxymethanediol Chemical compound CCCCOC(O)O SDUJCTKTKOLVFG-UHFFFAOYSA-N 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- YACLQRRMGMJLJV-UHFFFAOYSA-N chloroprene Chemical compound ClC(=C)C=C YACLQRRMGMJLJV-UHFFFAOYSA-N 0.000 description 1
- HNEGQIOMVPPMNR-IHWYPQMZSA-N citraconic acid Chemical compound OC(=O)C(/C)=C\C(O)=O HNEGQIOMVPPMNR-IHWYPQMZSA-N 0.000 description 1
- 229940018557 citraconic acid Drugs 0.000 description 1
- 239000004567 concrete Substances 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- LDHQCZJRKDOVOX-NSCUHMNNSA-N crotonic acid Chemical compound C\C=C\C(O)=O LDHQCZJRKDOVOX-NSCUHMNNSA-N 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 150000003946 cyclohexylamines Chemical class 0.000 description 1
- XXKOQQBKBHUATC-UHFFFAOYSA-N cyclohexylmethylcyclohexane Chemical compound C1CCCCC1CC1CCCCC1 XXKOQQBKBHUATC-UHFFFAOYSA-N 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- JGFBRKRYDCGYKD-UHFFFAOYSA-N dibutyl(oxo)tin Chemical compound CCCC[Sn](=O)CCCC JGFBRKRYDCGYKD-UHFFFAOYSA-N 0.000 description 1
- 239000012975 dibutyltin dilaurate Substances 0.000 description 1
- UHGHZQYYPSGQDA-UHFFFAOYSA-L dibutyltin(2+);9-methyl-2-(7-methyloctyl)-2-(sulfanylmethyl)decanoate Chemical compound CCCC[Sn+2]CCCC.CC(C)CCCCCCC(CS)(C([O-])=O)CCCCCCC(C)C.CC(C)CCCCCCC(CS)(C([O-])=O)CCCCCCC(C)C UHGHZQYYPSGQDA-UHFFFAOYSA-L 0.000 description 1
- VKNUORWMCINMRB-UHFFFAOYSA-N diethyl malate Chemical compound CCOC(=O)CC(O)C(=O)OCC VKNUORWMCINMRB-UHFFFAOYSA-N 0.000 description 1
- OEEJEJXWNGIUHQ-UHFFFAOYSA-M dimethyl-(2-prop-2-enoyloxyethyl)-(3-trimethoxysilylpropyl)azanium;chloride Chemical compound [Cl-].CO[Si](OC)(OC)CCC[N+](C)(C)CCOC(=O)C=C OEEJEJXWNGIUHQ-UHFFFAOYSA-M 0.000 description 1
- LQRUPWUPINJLMU-UHFFFAOYSA-N dioctyl(oxo)tin Chemical compound CCCCCCCC[Sn](=O)CCCCCCCC LQRUPWUPINJLMU-UHFFFAOYSA-N 0.000 description 1
- 238000003618 dip coating Methods 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- WNAHIZMDSQCWRP-UHFFFAOYSA-N dodecane-1-thiol Chemical compound CCCCCCCCCCCCS WNAHIZMDSQCWRP-UHFFFAOYSA-N 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 239000000806 elastomer Substances 0.000 description 1
- 238000004070 electrodeposition Methods 0.000 description 1
- 238000010556 emulsion polymerization method Methods 0.000 description 1
- 125000003700 epoxy group Chemical group 0.000 description 1
- 238000005530 etching Methods 0.000 description 1
- UIWXSTHGICQLQT-UHFFFAOYSA-N ethenyl propanoate Chemical compound CCC(=O)OC=C UIWXSTHGICQLQT-UHFFFAOYSA-N 0.000 description 1
- FWDBOZPQNFPOLF-UHFFFAOYSA-N ethenyl(triethoxy)silane Chemical compound CCO[Si](OCC)(OCC)C=C FWDBOZPQNFPOLF-UHFFFAOYSA-N 0.000 description 1
- NKSJNEHGWDZZQF-UHFFFAOYSA-N ethenyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)C=C NKSJNEHGWDZZQF-UHFFFAOYSA-N 0.000 description 1
- NNBRCHPBPDRPIT-UHFFFAOYSA-N ethenyl(tripropoxy)silane Chemical compound CCCO[Si](OCCC)(OCCC)C=C NNBRCHPBPDRPIT-UHFFFAOYSA-N 0.000 description 1
- MABAWBWRUSBLKQ-UHFFFAOYSA-N ethenyl-tri(propan-2-yloxy)silane Chemical compound CC(C)O[Si](OC(C)C)(OC(C)C)C=C MABAWBWRUSBLKQ-UHFFFAOYSA-N 0.000 description 1
- KUGSJJNCCNSRMM-UHFFFAOYSA-N ethoxyboronic acid Chemical compound CCOB(O)O KUGSJJNCCNSRMM-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- WBJINCZRORDGAQ-UHFFFAOYSA-N formic acid ethyl ester Natural products CCOC=O WBJINCZRORDGAQ-UHFFFAOYSA-N 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 125000003055 glycidyl group Chemical group C(C1CO1)* 0.000 description 1
- 238000013007 heat curing Methods 0.000 description 1
- RRAMGCGOFNQTLD-UHFFFAOYSA-N hexamethylene diisocyanate Chemical compound O=C=NCCCCCCN=C=O RRAMGCGOFNQTLD-UHFFFAOYSA-N 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical compound OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 description 1
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 150000002596 lactones Chemical class 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000000873 masking effect Effects 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 125000005395 methacrylic acid group Chemical group 0.000 description 1
- UYVXZUTYZGILQG-UHFFFAOYSA-N methoxyboronic acid Chemical compound COB(O)O UYVXZUTYZGILQG-UHFFFAOYSA-N 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- XJRBAMWJDBPFIM-UHFFFAOYSA-N methyl vinyl ether Chemical compound COC=C XJRBAMWJDBPFIM-UHFFFAOYSA-N 0.000 description 1
- LVHBHZANLOWSRM-UHFFFAOYSA-N methylenebutanedioic acid Natural products OC(=O)CC(=C)C(O)=O LVHBHZANLOWSRM-UHFFFAOYSA-N 0.000 description 1
- BFXIKLCIZHOAAZ-UHFFFAOYSA-N methyltrimethoxysilane Chemical compound CO[Si](C)(OC)OC BFXIKLCIZHOAAZ-UHFFFAOYSA-N 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 150000002780 morpholines Chemical class 0.000 description 1
- 239000004570 mortar (masonry) Substances 0.000 description 1
- 238000010137 moulding (plastic) Methods 0.000 description 1
- GDGOJEMDZGZECC-UHFFFAOYSA-N n'-[(3-ethenylphenyl)methyl]-n-(3-trimethoxysilylpropyl)ethane-1,2-diamine Chemical compound CO[Si](OC)(OC)CCCNCCNCC1=CC=CC(C=C)=C1 GDGOJEMDZGZECC-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- UBVMBXTYMSRUDX-UHFFFAOYSA-N n-prop-2-enyl-3-trimethoxysilylpropan-1-amine Chemical compound CO[Si](OC)(OC)CCCNCC=C UBVMBXTYMSRUDX-UHFFFAOYSA-N 0.000 description 1
- 125000002560 nitrile group Chemical group 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- JFOJYGMDZRCSPA-UHFFFAOYSA-J octadecanoate;tin(4+) Chemical compound [Sn+4].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O JFOJYGMDZRCSPA-UHFFFAOYSA-J 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 150000001451 organic peroxides Chemical class 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 150000003014 phosphoric acid esters Chemical class 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229920000058 polyacrylate Polymers 0.000 description 1
- 229920002857 polybutadiene Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920000139 polyethylene terephthalate Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 230000000379 polymerizing effect Effects 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 239000000737 potassium alginate Substances 0.000 description 1
- USHAGKDGDHPEEY-UHFFFAOYSA-L potassium persulfate Chemical compound [K+].[K+].[O-]S(=O)(=O)OOS([O-])(=O)=O USHAGKDGDHPEEY-UHFFFAOYSA-L 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- NPSSWQJHYLDCNV-UHFFFAOYSA-N prop-2-enoic acid;hydrochloride Chemical compound Cl.OC(=O)C=C NPSSWQJHYLDCNV-UHFFFAOYSA-N 0.000 description 1
- 239000000770 propane-1,2-diol alginate Substances 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 238000010526 radical polymerization reaction Methods 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 239000005060 rubber Substances 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 150000004756 silanes Chemical class 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 229920002050 silicone resin Polymers 0.000 description 1
- 239000010454 slate Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000010558 suspension polymerization method Methods 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 229940066771 systemic antihistamines piperazine derivative Drugs 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- LFQCEHFDDXELDD-UHFFFAOYSA-N tetramethyl orthosilicate Chemical compound CO[Si](OC)(OC)OC LFQCEHFDDXELDD-UHFFFAOYSA-N 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 229960000790 thymol Drugs 0.000 description 1
- KSBAEPSJVUENNK-UHFFFAOYSA-L tin(ii) 2-ethylhexanoate Chemical compound [Sn+2].CCCCC(CC)C([O-])=O.CCCCC(CC)C([O-])=O KSBAEPSJVUENNK-UHFFFAOYSA-L 0.000 description 1
- 239000005028 tinplate Substances 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- DVKJHBMWWAPEIU-UHFFFAOYSA-N toluene 2,4-diisocyanate Chemical compound CC1=CC=C(N=C=O)C=C1N=C=O DVKJHBMWWAPEIU-UHFFFAOYSA-N 0.000 description 1
- RUELTTOHQODFPA-UHFFFAOYSA-N toluene 2,6-diisocyanate Chemical compound CC1=C(N=C=O)C=CC=C1N=C=O RUELTTOHQODFPA-UHFFFAOYSA-N 0.000 description 1
- LDHQCZJRKDOVOX-UHFFFAOYSA-N trans-crotonic acid Natural products CC=CC(O)=O LDHQCZJRKDOVOX-UHFFFAOYSA-N 0.000 description 1
- 125000005270 trialkylamine group Chemical group 0.000 description 1
- SGCFZHOZKKQIBU-UHFFFAOYSA-N tributoxy(ethenyl)silane Chemical compound CCCCO[Si](OCCCC)(OCCCC)C=C SGCFZHOZKKQIBU-UHFFFAOYSA-N 0.000 description 1
- LGQXXHMEBUOXRP-UHFFFAOYSA-N tributyl borate Chemical compound CCCCOB(OCCCC)OCCCC LGQXXHMEBUOXRP-UHFFFAOYSA-N 0.000 description 1
- GQIUQDDJKHLHTB-UHFFFAOYSA-N trichloro(ethenyl)silane Chemical compound Cl[Si](Cl)(Cl)C=C GQIUQDDJKHLHTB-UHFFFAOYSA-N 0.000 description 1
- UMFJXASDGBJDEB-UHFFFAOYSA-N triethoxy(prop-2-enyl)silane Chemical compound CCO[Si](CC=C)(OCC)OCC UMFJXASDGBJDEB-UHFFFAOYSA-N 0.000 description 1
- GKASDNZWUGIAMG-UHFFFAOYSA-N triethyl orthoformate Chemical compound CCOC(OCC)OCC GKASDNZWUGIAMG-UHFFFAOYSA-N 0.000 description 1
- SOUMBTQFYKZXPN-UHFFFAOYSA-N trimethoxy(4-phenylbut-3-enyl)silane Chemical compound CO[Si](OC)(OC)CCC=CC1=CC=CC=C1 SOUMBTQFYKZXPN-UHFFFAOYSA-N 0.000 description 1
- 239000006097 ultraviolet radiation absorber Substances 0.000 description 1
- 229920001567 vinyl ester resin Polymers 0.000 description 1
- 239000005050 vinyl trichlorosilane Substances 0.000 description 1
- NLVXSWCKKBEXTG-UHFFFAOYSA-N vinylsulfonic acid Chemical compound OS(=O)(=O)C=C NLVXSWCKKBEXTG-UHFFFAOYSA-N 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 230000002087 whitening effect Effects 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
- 238000004383 yellowing Methods 0.000 description 1
- LRXTYHSAJDENHV-UHFFFAOYSA-H zinc phosphate Chemical compound [Zn+2].[Zn+2].[Zn+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O LRXTYHSAJDENHV-UHFFFAOYSA-H 0.000 description 1
- 229910000165 zinc phosphate Inorganic materials 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D175/00—Coating compositions based on polyureas or polyurethanes; Coating compositions based on derivatives of such polymers
- C09D175/04—Polyurethanes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F230/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal
- C08F230/04—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal
- C08F230/08—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal containing silicon
- C08F230/085—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal containing silicon the monomer being a polymerisable silane, e.g. (meth)acryloyloxy trialkoxy silanes or vinyl trialkoxysilanes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/62—Polymers of compounds having carbon-to-carbon double bonds
- C08G18/6295—Polymers of silicium containing compounds having carbon-to-carbon double bonds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/72—Polyisocyanates or polyisothiocyanates
- C08G18/77—Polyisocyanates or polyisothiocyanates having heteroatoms in addition to the isocyanate or isothiocyanate nitrogen and oxygen or sulfur
- C08G18/78—Nitrogen
- C08G18/79—Nitrogen characterised by the polyisocyanates used, these having groups formed by oligomerisation of isocyanates or isothiocyanates
- C08G18/791—Nitrogen characterised by the polyisocyanates used, these having groups formed by oligomerisation of isocyanates or isothiocyanates containing isocyanurate groups
- C08G18/792—Nitrogen characterised by the polyisocyanates used, these having groups formed by oligomerisation of isocyanates or isothiocyanates containing isocyanurate groups formed by oligomerisation of aliphatic and/or cycloaliphatic isocyanates or isothiocyanates
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/04—Homopolymers or copolymers of esters
- C09D133/14—Homopolymers or copolymers of esters of esters containing halogen, nitrogen, sulfur or oxygen atoms in addition to the carboxy oxygen
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Paints Or Removers (AREA)
- Polyurethanes Or Polyureas (AREA)
Description
本発明は、温和な硬化条件で塗膜を形成することが可能で、優れた耐溶剤性、耐酸性と耐薬品性を付与し、耐擦傷性、さらには耐汚染性にも優れるコーティング用硬化性樹脂組成物及び該樹脂組成物を含む自動車クリアーコーティング剤に関する。 The present invention is capable of forming a coating film under mild curing conditions, imparts excellent solvent resistance, acid resistance and chemical resistance, and has excellent scratch resistance and stain resistance, as well as coating curing. The present invention relates to a conductive resin composition and an automobile clear coating agent containing the resin composition.
自動車、建材、プラスチック部品、フィルムなどに塗膜を形成するために使用される上塗り塗料に対して、形成される塗膜の耐久性への要求が高まっている。外観性のほかに具体的には耐候性、耐溶剤性、耐酸性、耐薬品性、耐擦傷性等に優れる塗膜性能が要求されている。この様な高い塗膜性能と外観性を発現するため、従来から2液硬化型の塗料が使用されてきた。 The demand for the durability of the formed coating film is increasing for the top coating used to form a coating film on automobiles, building materials, plastic parts, films and the like. In addition to the appearance, specifically, coating film performance excellent in weather resistance, solvent resistance, acid resistance, chemical resistance, scratch resistance, etc. is required. In order to express such high coating film performance and appearance, a two-component curable coating material has been conventionally used.
自動車、建材、プラスチック部品、フィルムなどの保護や、意匠性を付与する目的で使用される塗料としては、アクリルラッカー塗料や2液性硬化型ウレタン樹脂塗料、2液性硬化型アクリルシリコン樹脂塗料等がある。アクリルラッカー塗料は、樹脂の架橋反応を伴わない1液型塗料であり、硬化剤の配合等の手間がなく扱い易いが、得られる塗膜は耐薬品性などに劣る。 Examples of paints used to protect automobiles, building materials, plastic parts, films, etc., and to provide design properties include acrylic lacquer paints, two-part curable urethane resin paints, and two-part curable acrylic silicon resin paints. There is. Acrylic lacquer paint is a one-pack paint that does not involve a crosslinking reaction of the resin and is easy to handle without the need for compounding a curing agent, but the resulting coating film is inferior in chemical resistance.
また、従来、自動車、産業機械、スチール製家具などの塗装には、主としてアルキドメラミンやアクリルメラミンなどのメラミン樹脂を含有する塗料が用いられてきたが(特許文献1)、かかるメラミン樹脂には、硬化時に有害なホルマリンが発生したり、硬化塗膜が耐酸性に劣るため、酸性雨に侵され、塗膜にエッチングや白化、シミなどが発生する現象が見られる。 Conventionally, paints containing melamine resins such as alkyd melamine and acrylic melamine have been mainly used for painting automobiles, industrial machines, steel furniture, etc. (Patent Document 1). A harmful formalin is generated at the time of curing, and the cured coating film is inferior in acid resistance, so that it is affected by acid rain, and etching, whitening, spots and the like are generated on the coating film.
このような欠点を解消するために、メラミン重合体を使用せず、水酸基とイソシアネートの反応を利用したいわゆるウレタン塗料が提案されている(特許文献2、特許文献3)。それぞれ水酸基含有樹脂と多官能イソシアネート化合物あるいは、ブロックイソシアネートとの組み合わせによる硬化性樹脂組成物であるが、耐酸性は改善されているものの、得られる塗膜の耐擦傷性や耐汚染性は充分ではない。 In order to eliminate such drawbacks, so-called urethane paints using a reaction between a hydroxyl group and an isocyanate without using a melamine polymer have been proposed (Patent Documents 2 and 3). Although each is a curable resin composition comprising a combination of a hydroxyl group-containing resin and a polyfunctional isocyanate compound or a blocked isocyanate, although the acid resistance has been improved, the resulting coating film has insufficient scratch resistance and stain resistance. Absent.
熱硬化型の2液ウレタン塗料を塗布し、塗膜を形成する方法(特許文献4)は、耐擦傷性と耐薬品性を得るためには架橋密度を上げる必要があり、水酸基価を高く設計することとなる。しかしながら耐擦傷性、耐薬品性ともに充分なレベルではなく、さらにプラスチック基材に対する付着性が低下する問題がある。 The method of forming a coating film by applying a thermosetting two-component urethane paint (Patent Document 4) needs to increase the crosslinking density in order to obtain scratch resistance and chemical resistance, and has a high hydroxyl value. Will be. However, both scratch resistance and chemical resistance are not sufficient levels, and there is a problem that adhesion to a plastic substrate is further reduced.
そこで耐擦傷性を向上させる目的で、アクリル樹脂などの水酸基含有樹脂を基体樹脂とし、ポリイソシアネート化合物を架橋剤とするウレタン架橋系塗料において、ポリカーボネートジオールを使用することが検討されている(特許文献5)。しかしながら、ポリカーボネートジオールは、アクリルポリオールとの相溶性が不充分であり更に、耐擦傷性確保のためにポリカーボネートジオールを多量に配合すると、架橋密度が低下するため耐候性、耐溶剤性が不充分となる問題がある。 Therefore, for the purpose of improving the scratch resistance, the use of polycarbonate diol in a urethane cross-linking paint using a hydroxyl group-containing resin such as an acrylic resin as a base resin and a polyisocyanate compound as a cross-linking agent has been studied (Patent Literature). 5). However, the polycarbonate diol has insufficient compatibility with the acrylic polyol, and further, when a large amount of the polycarbonate diol is blended in order to ensure scratch resistance, the crosslinking density is lowered, so that the weather resistance and solvent resistance are insufficient. There is a problem.
一方で、加水分解性シリル基を有する重合体を用いた塗料を使用することも提案されている(特許文献6)。加水分解性シリル基とアルコール性水酸基を有するビニル系重合体を用いることで、焼付け乾燥により、安定なシロキサン結合ないしはシロキシ結合を形成して硬化するため、耐酸性や耐候性に優れた塗膜が形成できる。しかしながら、得られる塗膜の耐擦傷性は充分ではないなどの問題があった。 On the other hand, it has also been proposed to use a paint using a polymer having a hydrolyzable silyl group (Patent Document 6). By using a vinyl polymer having a hydrolyzable silyl group and an alcoholic hydroxyl group, a stable siloxane bond or siloxy bond is formed and cured by baking and drying, so that a coating film excellent in acid resistance and weather resistance can be obtained. Can be formed. However, there is a problem that the obtained coating film has insufficient scratch resistance.
また、2液性硬化型アクリルシリコン樹脂塗料では、優れた耐溶剤性や耐薬品性を得ることができるものの、2液性硬化型ウレタン樹脂塗料と比べ加工性、リコート性、塗膜外観に劣るといった問題がある。 In addition, although the two-component curable acrylic silicone resin paint can obtain excellent solvent resistance and chemical resistance, it is inferior in workability, recoatability, and coating film appearance as compared with the two-component curable urethane resin paint. There is a problem.
また、多官能性モノマーやオリゴマーを主な構成成分とし、光ラジカル発生剤を用いてUV硬化する方法も報告されている(特許文献7)。本方法では、硬化に熱乾燥を必要としないため、プラスチックなどの基材を傷めることがなく、また短時間で高硬度の膜が得られる利点がある。しかしながら柔軟性に乏しく、衝撃により塗膜が割れたり、剥がれたりするなどの問題点がある。 In addition, a method in which a polyfunctional monomer or oligomer is used as a main constituent and UV curing is performed using a photo radical generator has been reported (Patent Document 7). Since this method does not require heat drying for curing, there is an advantage that a substrate having a high hardness can be obtained in a short time without damaging a substrate such as plastic. However, there are problems such as poor flexibility and cracking or peeling of the coating film by impact.
このため、自動車、建材、プラスチック部品、フィルムなどへ塗布可能で、耐候性、耐溶剤性、耐酸性、耐薬品性、耐擦傷性、耐汚染性等に優れる塗膜性能を有し、安価なコーティング剤(例えば自動車クリアーコーティング剤)の開発が求められていた。 For this reason, it can be applied to automobiles, building materials, plastic parts, films, etc., and it has coating performance with excellent weather resistance, solvent resistance, acid resistance, chemical resistance, scratch resistance, stain resistance, etc., and is inexpensive. Development of a coating agent (for example, an automobile clear coating agent) has been demanded.
本発明の課題は、プラスチック成型体やフィルムなど過度に熱をかけることができない材料に対しても、温和な硬化条件で塗膜を形成することが可能であり、耐候性、耐溶剤性、耐薬品性、耐擦傷性等に優れるコーティング用硬化性樹脂組成物を提供することであり、さらには傷に対して傷修復性が得られるコーティング用硬化性樹脂組成物を提供することである。また、耐酸性、耐薬品性、耐擦傷性、さらには耐汚染性も有する自動車クリアーコーティング剤を提供することである。 The problem of the present invention is that it is possible to form a coating film under mild curing conditions even on materials that cannot be heated excessively, such as plastic moldings and films, and weather resistance, solvent resistance, It is to provide a curable resin composition for coating that is excellent in chemical properties, scratch resistance, etc., and further to provide a curable resin composition for coating that can provide scratch repairability against scratches. Another object of the present invention is to provide an automobile clear coating agent having acid resistance, chemical resistance, scratch resistance, and contamination resistance.
本発明者らは、上記課題を解決するため鋭意検討した結果、加水分解性ケイ素基と水酸基を有する特定のビニル系共重合体、ポリイソシアネート化合物、及び、特定の硬化触媒を含有するコーティング用硬化性樹脂組成物が、温和な硬化条件で硬化可能で、耐候性と耐溶剤性、耐酸性、耐薬品性、さらには、耐擦傷性が良好で、各種基材への優れた付着性を示し、耐汚染性にも優れる塗膜を形成することを見出した。 As a result of intensive studies to solve the above-mentioned problems, the present inventors have found that a specific vinyl copolymer having a hydrolyzable silicon group and a hydroxyl group, a polyisocyanate compound, and a curing for coating containing a specific curing catalyst. The curable resin composition can be cured under mild curing conditions, and has good weather resistance, solvent resistance, acid resistance, chemical resistance, scratch resistance, and excellent adhesion to various substrates. The present inventors have found that a coating film having excellent stain resistance can be formed.
すなわち本発明は、
(A)主鎖が(メタ)アクリル系共重合体鎖であり、主鎖末端および/または側鎖に一般式(I):
R2 a
|
−Si−(OR1)3−a (I)
(式中、R1は水素原子または炭素数1〜10のアルキル基、R2は水素原子または炭素数1〜10のアルキル基、炭素数6〜25のアリール基および炭素数7〜12のアラルキル基から選ばれた1価の炭化水素基を示す。aは0〜2の整数を示す。)
で表される加水分解性ケイ素基を10個以上有し、且つ主鎖末端および/または側鎖に水酸基を10個以上有するビニル系共重合体、
(B)イソシアナート基を2個以上含有する化合物、および、
(C)加水分解性ケイ素基およびイソシアナートの硬化触媒を含有することを特徴とするコーティング用多液型硬化性樹脂組成物に関する。That is, the present invention
(A) The main chain is a (meth) acrylic copolymer chain, and the general formula (I):
R 2 a
|
-Si- (OR 1 ) 3-a (I)
Wherein R 1 is a hydrogen atom or an alkyl group having 1 to 10 carbon atoms, R 2 is a hydrogen atom or an alkyl group having 1 to 10 carbon atoms, an aryl group having 6 to 25 carbon atoms, and an aralkyl having 7 to 12 carbon atoms. And represents a monovalent hydrocarbon group selected from the group a represents an integer of 0 to 2.)
A vinyl-based copolymer having 10 or more hydrolyzable silicon groups represented by the formula, and having 10 or more hydroxyl groups in the main chain terminal and / or side chain,
(B) a compound containing two or more isocyanate groups, and
(C) The present invention relates to a multi-component curable resin composition for coating, comprising a hydrolyzable silicon group and an isocyanate curing catalyst.
前記ビニル系共重合体(A)は、その水酸基価が50〜300mgKOH/gである重合体であることが好ましい。 The vinyl copolymer (A) is preferably a polymer having a hydroxyl value of 50 to 300 mgKOH / g.
前記ビニル系共重合体(A)は、ガラス転移温度が−20〜80℃であることが好ましい。 The vinyl copolymer (A) preferably has a glass transition temperature of -20 to 80 ° C.
前記ビニル系共重合体(A)が、炭素数が3以上のアルキレン基を介して水酸基が(メタ)アクリロイル基と結合した単量体および/または炭素−炭素二重結合を末端に有するポリラクトン系単量体を共重合することによって得られるものであることが好ましい。 The vinyl copolymer (A) is a monomer having a hydroxyl group bonded to a (meth) acryloyl group via an alkylene group having 3 or more carbon atoms and / or a polylactone system having a carbon-carbon double bond at the terminal. It is preferable to be obtained by copolymerizing monomers.
前記硬化触媒(C)が、錫化合物、アルミキレート化合物、および、活性水素基を有さない3級アミン化合物からなる群から選ばれる1つ以上であることが好ましい。 It is preferable that the curing catalyst (C) is at least one selected from the group consisting of a tin compound, an aluminum chelate compound, and a tertiary amine compound having no active hydrogen group.
前記硬化触媒(C)が、環状アミン化合物であることが好ましい。 The curing catalyst (C) is preferably a cyclic amine compound.
また、本発明は、本発明のコーティング用多液型硬化性樹脂組成物を含む自動車クリアーコーティング剤に関する。 Moreover, this invention relates to the motor vehicle clear coating agent containing the multi-component curable resin composition for coating of this invention.
さらに本発明は、本発明のコーティング用多液型硬化性樹脂組成物を塗装した塗装物に関する。 Furthermore, the present invention relates to a coated product coated with the multi-component curable resin composition for coating of the present invention.
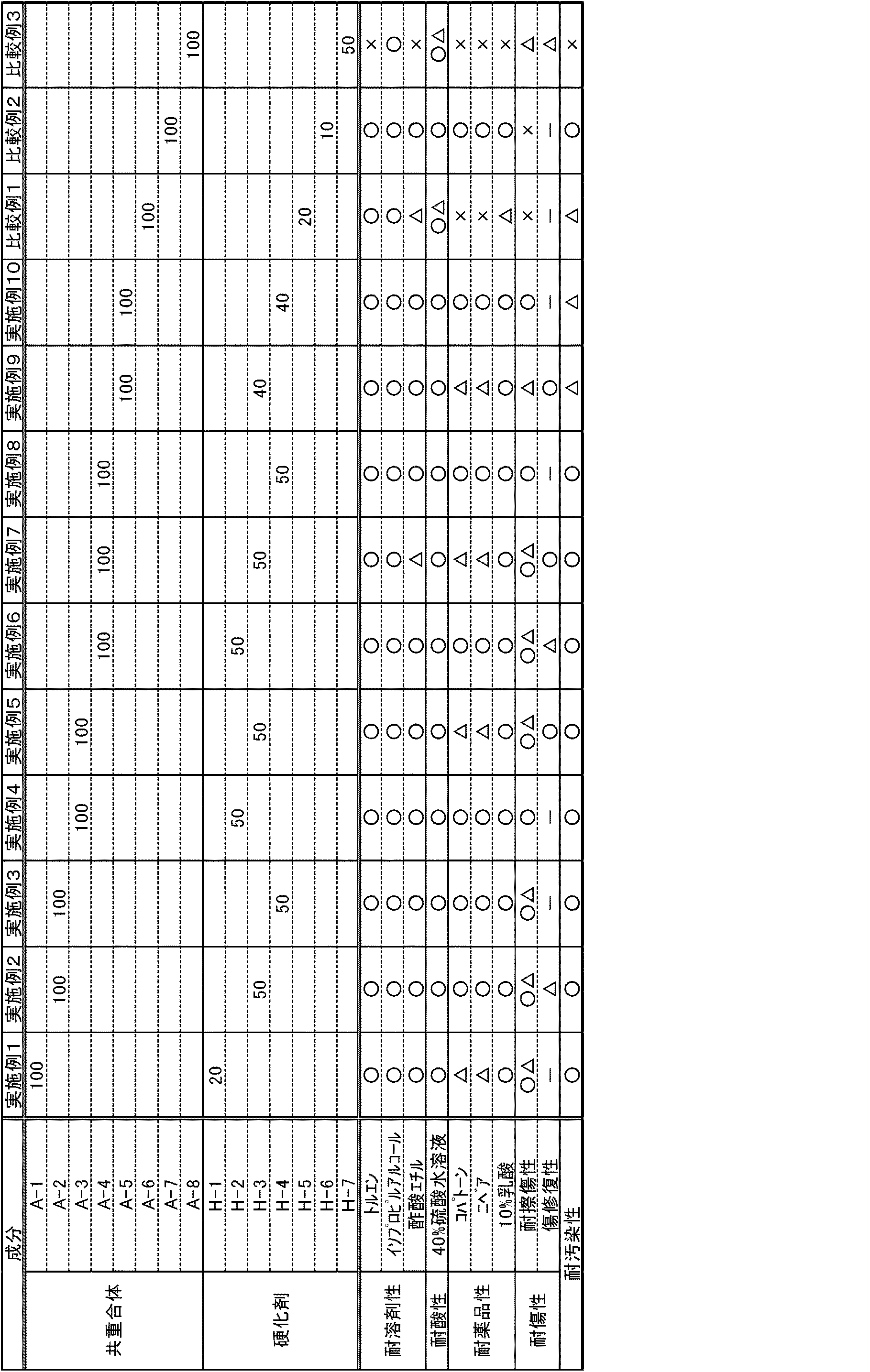
本発明のコーティング用多液型硬化性樹脂組成物は、温和な硬化条件で硬化可能で、耐候性、耐溶剤性、耐酸性、耐薬品性さらには、耐擦傷性が良好な塗膜を得ることができる。さらには、傷に対して傷修復性を有しており、耐汚染性にも優れた塗膜を形成し得る。 The multi-component curable resin composition for coating of the present invention can be cured under mild curing conditions to obtain a coating film having good weather resistance, solvent resistance, acid resistance, chemical resistance, and scratch resistance. be able to. Furthermore, it has a flaw-repairing property against scratches, and can form a coating film with excellent stain resistance.
以下に本発明をその実施の形態に基づき詳細に説明する。
本発明のコーティング用多液型硬化性樹脂組成物は、上記(A)〜(C)成分を含有する。
まず、各成分について説明する。Hereinafter, the present invention will be described in detail based on an embodiment thereof.
The multi-component curable resin composition for coating of the present invention contains the components (A) to (C).
First, each component will be described.
(A)ビニル系共重合体
(A)成分としては、主鎖が(メタ)アクリル系共重合体鎖であり、主鎖末端および/または側鎖に一般式(I):
R2 a
|
−Si−(OR1)3−a (I)
(式中、R1は水素原子または炭素数1〜10のアルキル基、R2は水素原子または炭素数1〜10のアルキル基、炭素数6〜25のアリール基および炭素数7〜12のアラルキル基から選ばれた1価の炭化水素基を示す。aは0〜2の整数を示す。)
で表される加水分解性ケイ素基を10個以上有し、且つ主鎖末端および/または側鎖に水酸基を10個以上有するビニル系共重合体であれば、特に限定することなく用いることができる。As the component (A) vinyl copolymer (A), the main chain is a (meth) acrylic copolymer chain, and the main chain terminal and / or side chain has the general formula (I):
R 2 a
|
-Si- (OR 1 ) 3-a (I)
Wherein R 1 is a hydrogen atom or an alkyl group having 1 to 10 carbon atoms, R 2 is a hydrogen atom or an alkyl group having 1 to 10 carbon atoms, an aryl group having 6 to 25 carbon atoms, and an aralkyl having 7 to 12 carbon atoms. And represents a monovalent hydrocarbon group selected from the group a represents an integer of 0 to 2.)
As long as it is a vinyl copolymer having 10 or more hydrolyzable silicon groups represented by the formula and 10 or more hydroxyl groups at the main chain terminal and / or side chain, it can be used without any particular limitation. .
加水分解性ケイ素基(以下、加水分解性シリル基とも記載する)とは、加水分解性基が結合したケイ素原子を含む基のことである。加水分解性シリル基は、(A)成分の主鎖末端に結合していてもよく、側鎖に結合していてもよく、主鎖末端および側鎖に結合していてもよい。 The hydrolyzable silicon group (hereinafter also referred to as a hydrolyzable silyl group) is a group containing a silicon atom to which a hydrolyzable group is bonded. The hydrolyzable silyl group may be bonded to the main chain terminal of the component (A), may be bonded to the side chain, or may be bonded to the main chain terminal and the side chain.
水酸基は、(A)成分の主鎖末端に結合していてもよく、側鎖に結合していてもよく、主鎖末端および側鎖に結合していてもよい。 The hydroxyl group may be bonded to the main chain end of the component (A), may be bonded to the side chain, or may be bonded to the main chain end and the side chain.
前記一般式(I)において、本発明の組成物の硬化性が良好になるという点から、aが0または1であることが好ましい。OR1またはR2が複数個ある場合は、それらは同一であっても異なっていてもよい。In the said general formula (I), it is preferable that a is 0 or 1 from the point that the sclerosis | hardenability of the composition of this invention becomes favorable. When there are a plurality of OR 1 or R 2 , they may be the same or different.
中でも、硬化速度が速く、得られる塗膜の耐溶剤性や耐薬品性、さらに耐酸性に優れるという観点から、R1は水素原子または炭素数1〜4のアルキル基であることが好ましく、水素原子または炭素数1〜2のアルキル基であることがより好ましい。Among these, R 1 is preferably a hydrogen atom or an alkyl group having 1 to 4 carbon atoms from the viewpoint that the curing speed is high, and the resulting coating film has excellent solvent resistance, chemical resistance, and acid resistance. It is more preferably an atom or an alkyl group having 1 to 2 carbon atoms.
また、硬化速度が速く、架橋度が高くなるとの観点から、R2は立体的に大きくないことがよく、水素原子、炭素数1〜6のアルキル基およびシクロアルキル基、ならびに、フェニル基が好ましく、炭素数1〜3のアルキル基であることがより好ましい。Further, from the viewpoint of a high curing rate and a high degree of crosslinking, R 2 is preferably not sterically large, and a hydrogen atom, an alkyl group having 1 to 6 carbon atoms and a cycloalkyl group, and a phenyl group are preferable. And more preferably an alkyl group having 1 to 3 carbon atoms.
ビニル系共重合体(A)は、ガラス転移温度が−20〜80℃であることが、耐薬品性と耐擦傷性のバランスに優れる点から好ましい。ガラス転移温度が−20℃未満である場合は、塗膜にタックが残るとともに耐溶剤や耐薬品性が悪化する傾向がある。一方、ガラス転移温度が80℃を超える場合には、耐溶剤性や耐薬品性は優れるものの、塗膜が脆くなり耐擦傷性が充分ではない。 The vinyl copolymer (A) preferably has a glass transition temperature of -20 to 80 ° C from the viewpoint of excellent balance between chemical resistance and scratch resistance. When the glass transition temperature is less than −20 ° C., tack remains in the coating film and the solvent resistance and chemical resistance tend to deteriorate. On the other hand, when the glass transition temperature exceeds 80 ° C., although the solvent resistance and chemical resistance are excellent, the coating film becomes brittle and the scratch resistance is not sufficient.
また、ビニル系共重合体(A)の水酸基価は50〜300mgKOH/gであることが好ましい。 The hydroxyl value of the vinyl copolymer (A) is preferably 50 to 300 mgKOH / g.
ビニル系共重合体(A)は、炭素数が3以上のアルキレン基を介して水酸基が(メタ)アクリロイル基と結合した単量体および/または炭素−炭素二重結合を末端に有するポリラクトン系単量体を共重合することによって得られるものであることが好ましい。 The vinyl copolymer (A) is a monomer having a hydroxyl group bonded to a (meth) acryloyl group via an alkylene group having 3 or more carbon atoms and / or a polylactone unit having a carbon-carbon double bond at the terminal. It is preferably obtained by copolymerizing a monomer.
ビニル系共重合体(A)の製造方法としては特に限定されないが、主鎖が(メタ)アクリル系共重合体鎖である共重合体が得られる方法の中で、例えば、(a)加水分解性シリル基含有ビニル系単量体、(b)水酸基含有ビニル系単量体、及び、(c)その他の重合性ビニル系単量体を重合させる方法などにより得られる。 Although it does not specifically limit as a manufacturing method of a vinyl type copolymer (A), For example, (a) Hydrolysis in the method in which the main chain is a (meth) acrylic-type copolymer chain is obtained. It is obtained by a method of polymerizing a polymerizable silyl group-containing vinyl monomer, (b) a hydroxyl group-containing vinyl monomer, and (c) another polymerizable vinyl monomer.
(a)加水分解性シリル基含有ビニル系単量体
加水分解性シリル基含有ビニル系単量体(a)としては、下記一般式(I)で表されるアルコキシシリル基を有するビニル系単量体が有用である。(A) Hydrolyzable silyl group-containing vinyl monomer As the hydrolyzable silyl group-containing vinyl monomer (a), a vinyl monomer having an alkoxysilyl group represented by the following general formula (I) The body is useful.
R2 a
|
−Si−(OR1)3−a (I)
(式中、R1は水素原子または炭素数1〜10のアルキル基、R2は水素原子または炭素数1〜10のアルキル基、炭素数6〜25のアリール基および炭素数7〜12のアラルキル基から選ばれた1価の炭化水素基を示す。aは0〜2の整数を示す。)R 2 a
|
-Si- (OR 1 ) 3-a (I)
Wherein R 1 is a hydrogen atom or an alkyl group having 1 to 10 carbon atoms, R 2 is a hydrogen atom or an alkyl group having 1 to 10 carbon atoms, an aryl group having 6 to 25 carbon atoms, and an aralkyl having 7 to 12 carbon atoms. And represents a monovalent hydrocarbon group selected from the group a represents an integer of 0 to 2.)
加水分解性シリル基含有ビニル系単量体(a)の具体例としては、ビニルトリメトキシシラン、ビニルトリエトキシシラン、ビニルトリ(n−プロポキシ)シラン、ビニルトリイソプロポキシシラン、ビニルトリブトキシシラン、ビニルトリス(β−メトキシエトキシ)シラン、アリルトリエトキシシラン、トリメトキシシリルプロピルアリルアミン、γ−(メタ)アクリロキシプロピルトリメトキシシラン、γ−(メタ)アクリロキシプロピルトリエトキシシラン、γ−(メタ)アクリロキシプロピルトリイソプロポキシシラン、γ−(メタ)アクリロキシプロピルトリス(β−メトキシエトキシ)シラン、γ−(メタ)アクリロキシプロピルメチルジメトキシシラン、γ−(メタ)アクリロキシプロピルメチルジエトキシシラン、γ−(メタ)アクリロキシプロピルジメチルメトキシシラン、γ−(メタ)アクリロキシプロピルジメチルエトキシシラン、N−ビニルベンジル−γ−アミノプロピルトリメトキシシラン、2−スチリルエチルトリメトキシシラン、3−(N−スチリルメチル−2−アミノエチルアミノ)プロピルトリメトキシシラン、(メタ)アクリロキシエチルジメチル(3−トリメトキシシリルプロピル)アンモニウムクロライド、ビニルトリアセトキシシラン、ビニルトリクロルシランなどを挙げることができ、これらの中では、特にアルコキシシリル基含有単量体であるγ−(メタ)アクリロキシプロピルトリメトキシシラン、γ−(メタ)アクリロキシプロピルトリエトキシシランが安定性の点で好ましい。 Specific examples of the hydrolyzable silyl group-containing vinyl monomer (a) include vinyltrimethoxysilane, vinyltriethoxysilane, vinyltri (n-propoxy) silane, vinyltriisopropoxysilane, vinyltributoxysilane, vinyltris. (Β-methoxyethoxy) silane, allyltriethoxysilane, trimethoxysilylpropylallylamine, γ- (meth) acryloxypropyltrimethoxysilane, γ- (meth) acryloxypropyltriethoxysilane, γ- (meth) acryloxy Propyltriisopropoxysilane, γ- (meth) acryloxypropyltris (β-methoxyethoxy) silane, γ- (meth) acryloxypropylmethyldimethoxysilane, γ- (meth) acryloxypropylmethyldiethoxysilane, γ- (Meta) Acryloxypropyldimethylmethoxysilane, γ- (meth) acryloxypropyldimethylethoxysilane, N-vinylbenzyl-γ-aminopropyltrimethoxysilane, 2-styrylethyltrimethoxysilane, 3- (N-styrylmethyl-2- Aminoethylamino) propyltrimethoxysilane, (meth) acryloxyethyldimethyl (3-trimethoxysilylpropyl) ammonium chloride, vinyltriacetoxysilane, vinyltrichlorosilane, etc. Γ- (meth) acryloxypropyltrimethoxysilane and γ- (meth) acryloxypropyltriethoxysilane, which are group-containing monomers, are preferable from the viewpoint of stability.
これらの加水分解性シリル基含有ビニル系単量体(a)は単独で用いても良く、2種以上併用しても良い。 These hydrolyzable silyl group-containing vinyl monomers (a) may be used alone or in combination of two or more.
なお、本明細書中では「(メタ)アクリル」とは、アクリルおよび/またはメタクリルを意味する。「(メタ)アクリロキシ」とは、メタアクリロキシおよび/またはアクリロキシを表すこととする。また、「(メタ)アクリレート」は、アクリレートおよび/またはメタクリレートを意味する。 In the present specification, “(meth) acryl” means acrylic and / or methacrylic. “(Meth) acryloxy” means methacryloxy and / or acryloxy. “(Meth) acrylate” means acrylate and / or methacrylate.
加水分解性シリル基含有ビニル系単量体(a)は、ビニル系共重合体(A)一分子につき加水分解性シリル基が10〜30個、特に好ましくは10〜20個含有されるように使用することが、充分な耐溶剤性と耐薬品性を与え、また擦傷性が良好になるという観点から好ましい。一分子中の加水分解性シリル基の個数は、単量体混合物中の加水分解性シリル基含有ビニル系単量体(a)の割合(モル数)と、ビニル系単量体(a)の数平均分子量から計算によって求めることができる。 The hydrolyzable silyl group-containing vinyl monomer (a) contains 10 to 30, particularly preferably 10 to 20, hydrolyzable silyl groups per molecule of the vinyl copolymer (A). It is preferable to use it from the viewpoint of providing sufficient solvent resistance and chemical resistance and improving scratch resistance. The number of hydrolyzable silyl groups in one molecule depends on the ratio (number of moles) of the hydrolyzable silyl group-containing vinyl monomer (a) in the monomer mixture and the vinyl monomer (a). It can be obtained by calculation from the number average molecular weight.
加水分解性シリル基含有ビニル系単量体(a)は、ビニル系共重合体(A)の全単量体100重量部中、5重量部以上80重量部以下配合するのが好ましく、10重量部以上60重量部以下配合するのが特に好ましい。 The hydrolyzable silyl group-containing vinyl monomer (a) is preferably blended in an amount of 5 to 80 parts by weight in 100 parts by weight of the total monomers of the vinyl copolymer (A). It is particularly preferable to blend in an amount of not less than 60 parts by weight.
(b)水酸基含有ビニル系単量体
水酸基含有ビニル系単量体(b)としては、特に限定されないが、例えば、2−ヒドロキシエチル(メタ)アクリレート、2−ヒドロキシプロピル(メタ)アクリレート、3−ヒドロキシプロピル(メタ)アクリレート、2−ヒドロキシブチル(メタ)アクリレート、4−ヒドロキシブチル(メタ)アクリレート、グリセロールモノ(メタ)アクリレート、シクロヘキサンジメタノールモノ(メタ)アクリレート、2−ヒドロキシエチルビニルエーテル、N−メチロール(メタ)アクリルアミド、4−ヒドロキシスチレンビニルトルエンなど水酸基含有ビニル系単量体;PlaccelFA−1、PlaccelFA−4、PlaccelFM−1、PlaccelFM−4(以上ダイセル化学(株)製)などの重合可能な炭素−炭素二重結合を末端に有する変性ラクトンまたはポリエステル;ブレンマーPPシリーズ、ブレンマーPEシリーズ、ブレンマーPEPシリーズ(以上日油(株)製)、MA−30、MA−50、MA−100、MA−150、RA−1120、RA−2614、RMA−564、RMA−568、RMA−1114、MPG130−MA(以上、日本乳化剤(株)製)などの重合可能な炭素−炭素二重結合を末端に有するポリオキシアルキレンなどが挙げられる。(B) Hydroxyl group-containing vinyl monomer The hydroxyl group-containing vinyl monomer (b) is not particularly limited, and examples thereof include 2-hydroxyethyl (meth) acrylate, 2-hydroxypropyl (meth) acrylate, 3- Hydroxypropyl (meth) acrylate, 2-hydroxybutyl (meth) acrylate, 4-hydroxybutyl (meth) acrylate, glycerol mono (meth) acrylate, cyclohexanedimethanol mono (meth) acrylate, 2-hydroxyethyl vinyl ether, N-methylol Hydroxyl group-containing vinyl monomers such as (meth) acrylamide and 4-hydroxystyrene vinyltoluene; PlaccelFA-1, PlaccelFA-4, PlaccelFM-1, PlaccelFM-4 (manufactured by Daicel Chemical Co., Ltd.) Modified lactone or polyester having a polymerizable carbon-carbon double bond at the end thereof; BLEMMER PP series, BLEMMER PE series, BLEMMER PEP series (manufactured by NOF CORPORATION), MA-30, MA-50, MA- 100, MA-150, RA-1120, RA-2614, RMA-564, RMA-568, RMA-1114, MPG130-MA (above, manufactured by Nippon Emulsifier Co., Ltd.) and other polymerizable carbon-carbon double bonds And polyoxyalkylene having a terminal.
中でも、イソシアナートとの反応性に優れ、耐候性や耐薬品性、耐擦傷性が良好な塗膜が得られるという点から、2−ヒドロキシエチル(メタ)アクリレート、2−ヒドロキシプロピル(メタ)アクリレート、2−ヒドロキシブチル(メタ)アクリレートが好ましい。 Among them, 2-hydroxyethyl (meth) acrylate and 2-hydroxypropyl (meth) acrylate are excellent in reactivity with isocyanates, and can provide a coating film having good weather resistance, chemical resistance, and scratch resistance. 2-hydroxybutyl (meth) acrylate is preferred.
さらに、3−ヒドロキシプロピル(メタ)アクリレートや4−ヒドロキシブチル(メタ)アクリレート、シクロヘキサンジメタノールモノ(メタ)アクリレートなどの、炭素数が3以上、より好ましくは炭素数が3以上10以下のアルキレン基を介して水酸基が(メタ)アクリロイル基と結合した単量体やPlaccelなどの炭素−炭素二重結合を末端に有するラクトン変性単量体(ポリラクトン系単量体)を使用すれば、傷修復性が得られる点から好ましい。 Further, an alkylene group having 3 or more carbon atoms, more preferably 3 to 10 carbon atoms, such as 3-hydroxypropyl (meth) acrylate, 4-hydroxybutyl (meth) acrylate, and cyclohexanedimethanol mono (meth) acrylate. If a lactone-modified monomer (polylactone monomer) having a terminal carbon-carbon double bond such as Placcel, such as a monomer in which a hydroxyl group is bonded to a (meth) acryloyl group via an acid, is repaired. Is preferable from the point that can be obtained.
これらの水酸基含有ビニル系単量体(b)成分は、単独で用いてもよいし、また2種以上を併用しても良い。 These hydroxyl group-containing vinyl monomers (b) may be used alone or in combination of two or more.
また使用量としては、ビニル系共重合体(A)一分子につき水酸基が10〜50個、特に好ましくは10〜30個含有されるように設計することが、良好な耐擦傷性を得られるという点から好ましい。一分子中の水酸基の個数は、単量体混合物中の水酸基含有ビニル系単量体(b)の割合(モル数)と、ビニル系単量体(A)の数平均分子量から計算によって求めることができる。 As for the amount used, it is said that it is possible to obtain good scratch resistance by designing the vinyl copolymer (A) so that it contains 10 to 50, particularly preferably 10 to 30, hydroxyl groups per molecule. It is preferable from the point. The number of hydroxyl groups in one molecule is determined by calculation from the ratio (number of moles) of the hydroxyl group-containing vinyl monomer (b) in the monomer mixture and the number average molecular weight of the vinyl monomer (A). Can do.
特に、ビニル系共重合体(A)の水酸基価が50〜300mgKOH/gになるよう設計することが、耐薬品性と耐擦傷性が良好になるという観点から好ましい。 In particular, it is preferable to design the vinyl copolymer (A) to have a hydroxyl value of 50 to 300 mgKOH / g from the viewpoint of improving chemical resistance and scratch resistance.
水酸基含有ビニル系単量体(b)は、ビニル系共重合体(A)の全単量体100重量部中、10重量部以上80重量部以下配合するのが好ましく、15重量部以上60重量部以下配合するのが特に好ましい。 The hydroxyl group-containing vinyl monomer (b) is preferably blended in an amount of 10 to 80 parts by weight, based on 100 parts by weight of all monomers of the vinyl copolymer (A), and 15 to 60 parts by weight. It is particularly preferable to blend in an amount of no more than parts.
(c)その他の重合性ビニル系単量体
その他の重合性ビニル系単量体(c)としては、炭素−炭素二重結合を有していれば特に限定がなく、例えば、メチル(メタ)アクリレート、エチル(メタ)アクリレート、n−ブチル(メタ)アクリレート、イソブチル(メタ)アクリレート、t−ブチル(メタ)アクリレート、2−エチルヘキシル(メタ)アクリレート等の炭素数1〜20のアルキル(メタ)アクリレート;シクロヘキシル(メタ)アクリレート、イソボルニル(メタ)アクリレート、ジシクロペンタニル(メタ)アクリレート等の炭素数4〜20のシクロアルキル(メタ)アクリレート;アリル(メタ)アクリレート;ジシクロペンテニル(メタ)アクリレート;ベンジル(メタ)アクリレート等の炭素数3〜20のアラルキル(メタ)アクリレート;グリシジル(メタ)アクリレート、オキシシクロヘキシニル(メタ)アクリレート等のエポキシ基含有ビニル系単量体;アクリル酸、メタクリル酸、マレイン酸、無水マレイン酸、イタコン酸、無水イタコン酸、クロトン酸、フマル酸、シトラコン酸などのα、β−エチレン性不飽和カルボン酸あるいは、それらの塩(アルカリ金属塩、アンモニウム塩、アミン塩など);無水マレイン酸などの酸無水物またはそれらと炭素数1〜20の直鎖または分岐のアルコールとのハーフエステルあるいは、それらの塩(アルカリ金属塩、アンモニウム塩、アミン塩など);スチレンスルホン酸、ビニルスルホン酸、2−(メタ)アクリロキシエチルスルホン酸、2−(メタ)アクリロキシエチルホスフェートなどの重合可能な炭素−炭素二重結合を有する酸、あるいは、それらの塩(アルカリ金属塩、アンモニウム塩、アミン塩など);N,N−ジメチルアミノエチル(メタ)アクリレート、N,N−ジメチルアミノプロピル(メタ)アクリレート、N,N−ジエチルアミノエチル(メタ)アクリレートなどのアミノ基含有ビニル単量体、あるいは、それらの塩(塩酸塩、酢酸塩など);トリメチルアミノエチル(メタ)アクリレート塩酸塩などの4級アミノ基を有するビニル単量体;(メタ)アクリルアミド、α−エチル(メタ)アクリルアミド、N−ブトキシメチル(メタ)アクリルアミド、N,N−ジメチル(メタ)アクリルアミド、N−メチル(メタ)アクリルアミド、アクリロイルモルホリン等の(メタ)アクリルアミド、あるいは、それらの塩(塩酸塩、酢酸塩など);N−ビニルピロリドン、N−ビニルピリジン、N−ビニルイミダゾールなどの含窒素ビニル単量体;α、β−エチレン性不飽和カルボン酸のヒドロキシアルキルエステルのリン酸エステル;ウレタン結合やシロキサン結合を含む(メタ)アクリレートなどのビニル化合物;東亜合成化学(株)製のマクロモノマーであるAS−6、AN−6、AA−6、AB−6、AK−5などの化合物;酢酸ビニル、プロピオン酸ビニル、バーサチック酸ビニル、ジアリルフタレートなどのビニルエステルやアリル化合物;(メタ)アクリロニトリルなどのニトリル基含有ビニル系単量体;スチレン、α−メチルスチレン、クロロスチレン、4−ヒドロキシスチレン、ビニルトルエンなどの芳香族炭化水素系ビニル単量体;ビニルメチルエーテル、塩化ビニル、塩化ビニリデン、クロロプレン、プロピレン、ブタジエンなどのその他のビニル系単量体などを挙げることができ、これらの群から選ばれる1種または2種以上の混合物を使用することができる。(C) Other polymerizable vinyl monomers Other polymerizable vinyl monomers (c) are not particularly limited as long as they have a carbon-carbon double bond. For example, methyl (meth) C1-C20 alkyl (meth) acrylates such as acrylate, ethyl (meth) acrylate, n-butyl (meth) acrylate, isobutyl (meth) acrylate, t-butyl (meth) acrylate, 2-ethylhexyl (meth) acrylate, etc. Cycloalkyl (meth) acrylate having 4 to 20 carbon atoms such as cyclohexyl (meth) acrylate, isobornyl (meth) acrylate, dicyclopentanyl (meth) acrylate, etc .; allyl (meth) acrylate; dicyclopentenyl (meth) acrylate; Aralkyl having 3 to 20 carbon atoms such as benzyl (meth) acrylate ( ) Acrylate; epoxy group-containing vinyl monomers such as glycidyl (meth) acrylate, oxycyclohexylinyl (meth) acrylate; acrylic acid, methacrylic acid, maleic acid, maleic anhydride, itaconic acid, itaconic anhydride, crotonic acid , Α-, β-ethylenically unsaturated carboxylic acids such as fumaric acid and citraconic acid or salts thereof (alkali metal salts, ammonium salts, amine salts, etc.); acid anhydrides such as maleic anhydride or those and carbon number 1 -20 half-esters with linear or branched alcohols or salts thereof (alkali metal salts, ammonium salts, amine salts, etc.); styrene sulfonic acid, vinyl sulfonic acid, 2- (meth) acryloxyethyl sulfonic acid, Polymerizable carbon such as 2- (meth) acryloxyethyl phosphate Acids having a carbon double bond, or salts thereof (alkali metal salts, ammonium salts, amine salts, etc.); N, N-dimethylaminoethyl (meth) acrylate, N, N-dimethylaminopropyl (meth) acrylate, An amino group-containing vinyl monomer such as N, N-diethylaminoethyl (meth) acrylate or a salt thereof (hydrochloride, acetate, etc.); a quaternary amino group such as trimethylaminoethyl (meth) acrylate hydrochloride; Vinyl monomers having; (meth) acrylamide, α-ethyl (meth) acrylamide, N-butoxymethyl (meth) acrylamide, N, N-dimethyl (meth) acrylamide, N-methyl (meth) acrylamide, acryloylmorpholine, etc. (Meth) acrylamide or their salts (hydrochloride, acetic acid) Salts); nitrogen-containing vinyl monomers such as N-vinylpyrrolidone, N-vinylpyridine, N-vinylimidazole; phosphate esters of hydroxyalkyl esters of α, β-ethylenically unsaturated carboxylic acids; urethane bonds and siloxanes Vinyl compounds such as (meth) acrylate containing a bond; compounds such as AS-6, AN-6, AA-6, AB-6, and AK-5, which are macromonomers manufactured by Toagosei Chemical Co., Ltd .; vinyl acetate, Vinyl esters and allyl compounds such as vinyl propionate, vinyl versatate and diallyl phthalate; nitrile group-containing vinyl monomers such as (meth) acrylonitrile; styrene, α-methylstyrene, chlorostyrene, 4-hydroxystyrene, vinyltoluene Aromatic hydrocarbon monomers such as vinyl methyl ether, Vinyl, vinylidene chloride, chloroprene, propylene, can be cited and other vinyl monomers such as butadiene, may be used alone or in combination of two or more thereof selected from these groups.
その他の重合性ビニル系単量体(c)は、ビニル系共重合体(A)の全単量体100重量部中、1重量部以上85重量部以下配合するのが好ましく、3重量部以上75重量部以下配合するのが特に好ましい。 The other polymerizable vinyl monomer (c) is preferably blended in an amount of 1 part by weight or more and 85 parts by weight or less, based on 100 parts by weight of the total monomers of the vinyl copolymer (A). It is particularly preferable to add 75 parts by weight or less.
重合方法は特に限定されず、例えば溶液重合法、乳化重合法、懸濁重合法、塊状重合法等が挙げられ、溶液重合法が合成の容易さなどの点で好ましい。 The polymerization method is not particularly limited, and examples thereof include a solution polymerization method, an emulsion polymerization method, a suspension polymerization method, a bulk polymerization method, and the like. The solution polymerization method is preferable in terms of ease of synthesis.
重合に用いる開始剤としては、公知のものを挙げることができ、例えば、クメンハイドロパーオキサイドなどの有機過酸化物系、アゾビス−2−メチルブチロニトリルなどのアゾ化合物系、過硫酸カリウムなどの無機過酸化物系、過酸化物と還元剤を組み合わせるレドックス系などの開始剤を挙げることができる。 Examples of the initiator used for the polymerization include known ones such as organic peroxides such as cumene hydroperoxide, azo compound types such as azobis-2-methylbutyronitrile, potassium persulfate, and the like. Examples include inorganic peroxide-based initiators, redox-based initiators combining peroxides and reducing agents.
得られるビニル系共重合体(A)の重合度は、数平均分子量で2000ないし50000とする事が好ましく、3000ないし25000がより好ましく、3000ないし10000が特に好ましい。数平均分子量が2000未満の場合は、未重合の単量体が残存し易く、好ましくない。一方、数平均分子量が50000を超える場合には、得られる硬化性樹脂組成物は、その塗装時に糸引き等の欠陥を生じることが多い。重合度は、ラジカル発生剤の種類及び使用量、重合温度、及び連鎖移動剤の使用によって調節することができる。連鎖移動剤としては、n−ドデシルメルカプタン、t−ドデシルメルカプタン、及びγ−メルカプトプロピルトリメトキシシラン等が好適に使用できる。 The degree of polymerization of the resulting vinyl copolymer (A) is preferably 2000 to 50000 in terms of number average molecular weight, more preferably 3000 to 25000, and particularly preferably 3000 to 10,000. When the number average molecular weight is less than 2,000, an unpolymerized monomer tends to remain, which is not preferable. On the other hand, when the number average molecular weight exceeds 50,000, the resulting curable resin composition often has defects such as stringing during coating. The degree of polymerization can be adjusted by the type and amount of the radical generator, the polymerization temperature, and the use of a chain transfer agent. As the chain transfer agent, n-dodecyl mercaptan, t-dodecyl mercaptan, γ-mercaptopropyltrimethoxysilane and the like can be preferably used.
(B)イソシアナート基を2個以上有する化合物
本発明においては、架橋剤としてイソシアナート基を2個以上有する化合物(B)(以下、ポリイソシアナート化合物(B)という)を使用する。イソシアナート基は7個未満であることが好ましい。イソシアナート基が2個未満の場合、架橋剤として働かないのは当然であるが、7個以上の場合には、架橋点が集中しすぎて付着性を阻害する場合がある。(B) Compound having two or more isocyanate groups In the present invention, a compound (B) having two or more isocyanate groups (hereinafter referred to as polyisocyanate compound (B)) is used as a crosslinking agent. The number of isocyanate groups is preferably less than 7. Naturally, when the number of isocyanate groups is less than 2, it does not work as a crosslinking agent. However, when the number of isocyanate groups is 7 or more, the crosslinking points may be concentrated so as to inhibit adhesion.
ポリイソシアナート化合物(B)としては、脂肪族系もしくは芳香族系のものが挙げられる。 Examples of the polyisocyanate compound (B) include aliphatic or aromatic compounds.
脂肪族系ポリイソシアナートの具体例として、ヘキサメチレンジイソシアナート、ジシクロヘキシルメタン4,4’−イソシアナート、2,2,4−トリメチル−1,6−ジイソシアナート、イソフォロンジイソシアナートがある。 Specific examples of the aliphatic polyisocyanate include hexamethylene diisocyanate, dicyclohexylmethane 4,4′-isocyanate, 2,2,4-trimethyl-1,6-diisocyanate, and isophorone diisocyanate. .
芳香族ポリイソシアナートとしては、2,4―トリレンジイソシアナート、2,6―トリレンジイソシアナート、ジフェニルメタン−4,4’−ジイソシアナート、キシレンジイソシアナート、ポリメチレン−ポリフェニレル−ポリイソシアナートなどがある。
黄変などがなく、耐候性が良好という観点から、脂肪族ポリイソシアナートがより好ましい。Aromatic polyisocyanates include 2,4-tolylene diisocyanate, 2,6-tolylene diisocyanate, diphenylmethane-4,4′-diisocyanate, xylene diisocyanate, polymethylene-polyphenylel-polyisocyanate, etc. There is.
From the viewpoint of no yellowing and good weather resistance, an aliphatic polyisocyanate is more preferable.
ポリイソシアナート化合物(B)の構造としては、単量体、ビュレット型、アダクト型、イソシアヌレート型がある。これらの化合物は常温硬化用に用いることができるが、さらにこれらのイソシアナート基をブロック剤でマスクしたものを用いることもできる。そのブロック剤としてはメチルアルコール、エチルアルコール、n−プロピルアルコール、イソプロピルアルコール、n−ブチルアルコール、sec−ブチルアルコール、メチルセルソルブ、エチルセルソルブ、ブチルセルソルブ、ベンジルアルコール、フルフリルアルコール、シクロヘキシルアルコール、フェノール、o−クレゾール、m−クレゾール、p―クレゾール、p−tert−ブチルフェノール、チモール、p−ニトロフェノール、β―ナフトールなどがある。また、これらの化合物は、2種以上混合して用いることもできる。 The structure of the polyisocyanate compound (B) includes a monomer, a burette type, an adduct type, and an isocyanurate type. These compounds can be used for normal temperature curing, and further, those obtained by masking these isocyanate groups with a blocking agent can also be used. As the blocking agent, methyl alcohol, ethyl alcohol, n-propyl alcohol, isopropyl alcohol, n-butyl alcohol, sec-butyl alcohol, methyl cellosolve, ethyl cellosolve, butyl cellosolve, benzyl alcohol, furfuryl alcohol, cyclohexyl alcohol Phenol, o-cresol, m-cresol, p-cresol, p-tert-butylphenol, thymol, p-nitrophenol, β-naphthol and the like. Moreover, these compounds can also be used in mixture of 2 or more types.
市販されているポリイソシアナート化合物(B)としては、コロネートHL(日本ポリウレタン工業(株)製)やデュラネートP−301、デュラネートE−402、デュラネートE−405(以上、旭化成ケミカルズ(株)製)などのアダクト型、デュラネート18H、デュラネート21S、デュラネート22A(以上、旭化成ケミカルズ(株)製)などのビュレット型、コロネートHX、コロネートHK、コロネート2760、コロネート2349(以上、日本ポリウレタン工業(株)製)、デュラネートMFA、デュラネートTPA100、デュラネートTHA100、デュラネートTSA100(以上、旭化成ケミカルズ(株)製)、コロネートAP−M、コロネートBI301、コロネート2507(以上、日本ポリウレタン工業(株)製)、デュラネート17B、デュラネートMF(以上、旭化成ケミカルズ(株)製)などのブロック型が挙げられる。 As commercially available polyisocyanate compounds (B), Coronate HL (manufactured by Nippon Polyurethane Industry Co., Ltd.), Duranate P-301, Duranate E-402, Duranate E-405 (above, manufactured by Asahi Kasei Chemicals Corporation) Adduct type such as Duranate 18H, Duranate 21S, Duranate 22A (above, manufactured by Asahi Kasei Chemicals Co., Ltd.) Duranate MFA, Duranate TPA100, Duranate THA100, Duranate TSA100 (above, manufactured by Asahi Kasei Chemicals Corporation), Coronate AP-M, Coronate BI301, Coronate 2507 (above, Nippon Polyurethane Industry Ltd.)), Duranate 17B, Duranate MF (manufactured by Asahi Kasei Chemicals), and the block type and the like.
ビニル系共重合体(A)とポリイソシアナート化合物(B)の使用割合は、ビニル系共重合体(A)中の水酸基に対してイソシアナート基が好ましくは0.3〜2当量、より好ましくは0.5〜1.5当量である。ポリイソシアナート化合物が少なすぎる場合には、得られる硬化物の耐薬品性が劣る傾向があり、また多すぎる場合には耐衝撃性等の加工性が劣ることになり、また、塗り重ね時にちぢみを生じたりするおそれがある。 The use ratio of the vinyl copolymer (A) and the polyisocyanate compound (B) is preferably 0.3 to 2 equivalents, more preferably an isocyanate group with respect to the hydroxyl group in the vinyl copolymer (A). Is 0.5 to 1.5 equivalents. If the polyisocyanate compound is too little, the resulting cured product tends to have poor chemical resistance, and if it is too much, the processability such as impact resistance is poor, and it is difficult to rub at the time of coating. May occur.
(C)硬化触媒
本発明においては、加水分解性シリル基およびイソシアナートの硬化触媒(C)を使用する。(C) Curing Catalyst In the present invention, a hydrolyzable silyl group and isocyanate curing catalyst (C) is used.
硬化触媒としては特に限定されず、錫化合物、アルミキレート化合物、及び、活性水素基を有さない3級アミン化合物が塗膜の硬化性や得られる塗膜の総合的な物性が優れる点から好ましい。 The curing catalyst is not particularly limited, and a tin compound, an aluminum chelate compound, and a tertiary amine compound having no active hydrogen group are preferable from the viewpoint of excellent curability of the coating film and overall physical properties of the resulting coating film. .
前記錫化合物の具体例としては、ジオクチル錫ビス(2−エチルヘキシルマレート)、ジオクチル錫オキサイドまたはジブチル錫オキサイドとシリケートとの縮合物、ジブチル錫ジオクトエート、ジブチル錫ジラウレート、ジブチル錫ジステアレート、ジブチル錫ジアセチルアセトナート、ジブチル錫ビス(エチルマレート)、ジブチル錫ビス(ブチルマレート)、ジブチル錫ビス(2−エチルヘキシルマレート)、ジブチル錫ビス(オレイルマレート)、スタナスオクトエート、ステアリン酸錫、ジ−n−ブチル錫ラウレートオキサイドがある。また、ジブチル錫ビスイソノニル−3―メルカプトプロピオネート、ジオクチル錫ビスイソノニル−3−メルカプトプロピオネート、オクチルブチル錫ビスイソノニル−3−メルカプトプロピオネート、ジブチル錫ビスイソオクチルチオグルコレート、ジオクチル錫ビスイソオクチルチオグルコレート、オクチルブチル錫ビスイソオクチルチオグルコレートなどの分子内にS原子を有する錫化合物が挙げられる。 Specific examples of the tin compound include dioctyltin bis (2-ethylhexylmalate), dioctyltin oxide or a condensate of dibutyltin oxide and silicate, dibutyltin dioctoate, dibutyltin dilaurate, dibutyltin distearate, dibutyltin diacetylacetate Nert, dibutyltin bis (ethylmalate), dibutyltin bis (butylmalate), dibutyltin bis (2-ethylhexylmalate), dibutyltin bis (oleylmalate), stannous octoate, tin stearate, di-n-butyl There is tin laurate oxide. Dibutyltin bisisononyl-3-mercaptopropionate, dioctyltin bisisononyl-3-mercaptopropionate, octylbutyltin bisisononyl-3-mercaptopropionate, dibutyltin bisisooctylthioglucorate, dioctyltin bisisooctyl Examples thereof include tin compounds having an S atom in the molecule, such as thioglucolate and octylbutyltin bisisooctylthioglucolate.
前記錫化合物のうちでは、分子内にS原子を有する化合物が、イソシアナートを配合した場合の貯蔵安定性および可使時間が良好であることから好ましく、特に、ジブチル錫ビスイソノニル−3−メルカプトプロピオネート、ジブチル錫ビスイソオクチルチオグルコレートがより好ましく、ジオクチル錫ビスイソオクチルチオグルコレートが硬化性と貯蔵安定性、可使時間のバランスの点から特に好ましい。 Among the tin compounds, a compound having an S atom in the molecule is preferable because of good storage stability and usable time when an isocyanate is blended, and in particular, dibutyltin bisisononyl-3-mercaptopropio Nate and dibutyltin bisisooctylthioglucolate are more preferable, and dioctyltin bisisooctylthioglucolate is particularly preferable from the viewpoint of balance between curability, storage stability, and pot life.
また、アルミキレート化合物としては、エチルアセトアセテートアルミニウムジイソプロピレート、アルミニウムトリス(アセチルアセテート)、アルミニウムトリス(エチルアセトアセテート)、アルミニウムモノアセチルアセトネートビス(エチルアセトアセテート)、アルキルアセチルアセテートアルミニウムジイソプロピレートなどが挙げられる。 Aluminum chelate compounds include ethyl acetoacetate aluminum diisopropylate, aluminum tris (acetylacetate), aluminum tris (ethylacetoacetate), aluminum monoacetylacetonate bis (ethylacetoacetate), alkylacetylacetate aluminum diisopropylate Etc.
活性水素基を有さない3級アミン化合物を使用すれば、非錫系で且つ優れた塗膜物性が得られるため好ましい。 The use of a tertiary amine compound having no active hydrogen group is preferable because it is non-tin and excellent film properties can be obtained.
活性水素基を有さない3級アミン化合物としては、例えば、トリエチルアミン、トリエチレンジアミン、トリメチルアミンのようなトリアルキルアミン、テトラメチレンジアミン、N,N,N’,N’−テトラメチル−1,3−ブタンジアミンのようなテトラアルキルジアミン、ビス(ジエチルエタノールアミン)アジペートのようなエステルアミン、N,N−ジメチルシクロヘキシルアミンのようなシクロヘキシルアミン誘導体、N−メチルモルホリンのようなモルホリン誘導体、N,N’−ジエチル−2−メチルピペラジンのようなピペラジン誘導体があげられるが、硬化活性の観点から環状アミン化合物が好ましく、特にトリエチレンジアミンが好ましい。 Examples of the tertiary amine compound having no active hydrogen group include trialkylamines such as triethylamine, triethylenediamine, and trimethylamine, tetramethylenediamine, N, N, N ′, N′-tetramethyl-1,3- Tetraalkyldiamines such as butanediamine, esteramines such as bis (diethylethanolamine) adipate, cyclohexylamine derivatives such as N, N-dimethylcyclohexylamine, morpholine derivatives such as N-methylmorpholine, N, N ′ -Piperazine derivatives such as diethyl-2-methylpiperazine can be mentioned. From the viewpoint of curing activity, a cyclic amine compound is preferable, and triethylenediamine is particularly preferable.
これら硬化触媒(C)は単独で用いてもよく、2種以上併用してもよい。 These curing catalysts (C) may be used alone or in combination of two or more.
硬化触媒(C)の使用量としては、硬化温度と時間により適宜調整すればよいが、(メタ)アクリル系重合体(A)100重量部に対して0.01重量部以上20重量部以下であることが好ましく、0.1重量部以上5重量部以下がより好ましい。0.01重量部未満の場合は、加水分解性シリル基の反応性が不充分となり塗膜性能のバランスが劣る傾向にあり、20重量部を超えると、可使時間が短くなり作業性が劣る傾向にある。 The amount of the curing catalyst (C) used may be appropriately adjusted depending on the curing temperature and time, but is 0.01 parts by weight or more and 20 parts by weight or less with respect to 100 parts by weight of the (meth) acrylic polymer (A). It is preferable that the amount is 0.1 to 5 parts by weight. When the amount is less than 0.01 part by weight, the reactivity of the hydrolyzable silyl group tends to be insufficient and the balance of the coating film performance tends to be inferior. When the amount exceeds 20 parts by weight, the pot life is shortened and the workability is inferior. There is a tendency.
(硬化性樹脂組成物)
本発明の硬化性樹脂組成物は、上記(A)〜(C)成分以外に、必要に応じて例えば下記の成分を含有してもよい。
なお、外観性と表面硬度が低下すること、さらには耐汚染性が低下するおそれがあることから、本系ではポリブタジエン系重合体は含有しないことが好ましい。(Curable resin composition)
The curable resin composition of the present invention may contain, for example, the following components as necessary in addition to the components (A) to (C).
In this system, it is preferable not to contain a polybutadiene-based polymer because the appearance and surface hardness may be decreased, and further, the contamination resistance may be decreased.
(溶媒)
ビニル系共重合体(A)を含有する多液型硬化性樹脂組成物に用いる溶媒としては特に限定はなく、公知の芳香族系、脂肪族炭化水素系、エーテル系、ケトン系、エステル系、アルコール系、水等の溶媒を用いることができる。トルエン、キシレン、酢酸ブチル等のエステル系、メチルイソブチルケトン等のケトン系、脂肪族炭化水素含有溶剤を用いるのがビニル系共重合体(A)の溶解性の点から好ましい。
また、硬化時の塗膜外観を考慮し、その他の溶剤を混合して揮発速度を調整することが望ましい。(solvent)
There is no particular limitation on the solvent used in the multi-component curable resin composition containing the vinyl copolymer (A), and known aromatic, aliphatic hydrocarbon, ether, ketone, ester, Alcohol-based solvents such as water can be used. From the viewpoint of the solubility of the vinyl copolymer (A), it is preferable to use an ester type such as toluene, xylene or butyl acetate, a ketone type such as methyl isobutyl ketone, or an aliphatic hydrocarbon-containing solvent.
In consideration of the appearance of the coating film during curing, it is desirable to adjust the volatilization rate by mixing other solvents.
(脱水剤)
加水分解性シリル基と水酸基を有するビニル系共重合体(A)は、湿分との反応性を有するため、さらに脱水剤を配合することによって、組成物の保存安定性を向上させることができる。(Dehydrating agent)
Since the vinyl copolymer (A) having a hydrolyzable silyl group and a hydroxyl group has reactivity with moisture, the storage stability of the composition can be improved by further adding a dehydrating agent. .
脱水剤としては、例えば、オルソ蟻酸メチル、オルソ蟻酸エチルもしくはオルソ蟻酸ブチル等のオルソ蟻酸アルキル;オルソ酢酸メチル、オルソ酢酸エチルもしくはオルソ酢酸ブチル等のオルソ酢酸アルキル;オルソほう酸メチル、オルソほう酸エチル、オルソほう酸ブチル等のオルソほう酸アルキル等のオルソカルボン酸エステルや、テトラメトキシシラン、テトラエトキシシラン、メチルトリメトキシシラン等の高活性シラン化合物などが挙げられる。 Examples of the dehydrating agent include alkyl orthoformate such as methyl orthoformate, ethyl orthoformate or butyl orthoformate; alkyl orthoacetate such as methyl orthoacetate, ethyl orthoacetate or butyl orthoacetate; methyl orthoborate, ethyl orthoborate, ortho Examples thereof include orthocarboxylic acid esters such as alkyl orthoborate such as butyl borate, and highly active silane compounds such as tetramethoxysilane, tetraethoxysilane, and methyltrimethoxysilane.
脱水剤の使用量としては、ビニル系共重合体(A)100重量部に対して0.1重量部以上10重量部以下であることが好ましい。 The amount of the dehydrating agent used is preferably 0.1 to 10 parts by weight with respect to 100 parts by weight of the vinyl copolymer (A).
(その他)
本発明のコーティング用多液型硬化性樹脂組成物には、必要に応じて添加剤を配合することができる。添加剤としては可塑剤、分散剤、脱水剤、湿潤剤、増粘剤、消泡剤、防腐剤、沈降防止剤、レベリング剤、紫外線吸収剤、光安定剤等があげられる。(Other)
An additive can be mix | blended with the multi-component curable resin composition for coating of this invention as needed. Examples of the additive include a plasticizer, a dispersant, a dehydrating agent, a wetting agent, a thickening agent, an antifoaming agent, a preservative, an anti-settling agent, a leveling agent, an ultraviolet absorber, and a light stabilizer.
さらに必要に応じて顔料を添加しても良く、顔料としては、二酸化チタン、炭酸カルシルム、炭酸バリウム、カオリン等の白色顔料、カーボンブラック、ベンガラ、シアニンブルー等の有色系顔料が挙げられる。 Further, a pigment may be added as necessary. Examples of the pigment include white pigments such as titanium dioxide, calcium carbonate, barium carbonate, and kaolin, and colored pigments such as carbon black, bengara, and cyanine blue.
本発明の硬化性樹脂組成物の調製方法としては、硬化性樹脂組成物が多液型となるのであれば特に限定されない。組成物の混合比、貯蔵安定性、混合方法、ポットライフ等を考慮して配合成分を多液に分けて配合しておき、使用時に混合する。すなわち、2液型の場合は、配合成分をA液とB液の2液に分割して配合しておき、2液を使用時に混合する。A液とB液への分割方法は、貯蔵安定性等に応じて種々の組合せが可能である。また、必要に応じて、3液型あるいはそれ以上に分割することも可能である。 The method for preparing the curable resin composition of the present invention is not particularly limited as long as the curable resin composition is multi-component. In consideration of the mixing ratio of the composition, storage stability, mixing method, pot life and the like, the compounding components are mixed in multiple liquids and mixed at the time of use. That is, in the case of the two-pack type, the blending component is divided into two liquids, liquid A and liquid B, and the liquids are mixed at the time of use. The method of dividing the liquid A and the liquid B can be variously combined depending on the storage stability and the like. Moreover, it is also possible to divide into three liquid type or more if necessary.
以上のようにして、本発明に係わるコーティング用多液型硬化性樹脂組成物が得られる。本発明のコーティング用多液型硬化性樹脂組成物を塗布する際には、ロールコーター法、ブレードコーター法、グラビアコーター法、ビートコーター法、カーテンフローコーター法、浸漬塗布法、及びスプレー塗布法のいずれも可能である。 As described above, the multi-component curable resin composition for coating according to the present invention is obtained. When applying the multi-component curable resin composition for coating of the present invention, a roll coater method, a blade coater method, a gravure coater method, a beat coater method, a curtain flow coater method, a dip coating method, and a spray coating method. Either is possible.
(硬化方法)
また、硬化性樹脂組成物は、常温〜300℃の任意の温度で硬化することができる。
硬化性樹脂組成物が加熱硬化型の場合は、加熱温度が高いほど加熱時間は短縮でき得る。その場合、65〜250℃の温度で5秒〜60分加熱硬化することが好ましい。
もちろん、過度に熱をかけることができない材料に対しても温和な硬化条件で塗膜を形成することが可能である。その場合は、常温程度で湿分により硬化することが好ましい。(Curing method)
Moreover, the curable resin composition can be cured at an arbitrary temperature ranging from room temperature to 300 ° C.
When the curable resin composition is a thermosetting type, the heating time can be shortened as the heating temperature increases. In that case, it is preferable to heat cure at a temperature of 65 to 250 ° C. for 5 seconds to 60 minutes.
Of course, it is possible to form a coating film under mild curing conditions even for materials that cannot be heated excessively. In that case, it is preferable to cure by moisture at about room temperature.
(塗膜)
本発明の多液型コーティング用硬化性樹脂組成物の塗膜の厚みとしては、0.5〜70μmが好ましく、5〜50μmがより好ましい。薄すぎる場合には、物理的な磨耗により塗膜が欠損し易くなり、耐久性の面で好ましくなく、厚すぎる場合には、塗材を加工する際に、塗膜に割れ及び剥がれが発生し易くなり、好ましくない。(Coating)
As thickness of the coating film of the curable resin composition for multi-component coatings of this invention, 0.5-70 micrometers is preferable and 5-50 micrometers is more preferable. If it is too thin, the coating film is likely to be damaged due to physical wear, which is not preferable in terms of durability, and if it is too thick, the coating film may be cracked and peeled off when the coating material is processed. It becomes easy and is not preferable.
本発明に係わるコーティング用多液型硬化性樹脂組成物の被塗物としては、金属、無機物、有機物、および複合材料が挙げられる。金属としては、ステンレス、アルミニウム、ブリキ、トタン、軟鋼板、銅、真鍮、各種メッキ鋼板およびチタン等が挙げられる。化成処理、アルマイト処理などの表面処理を施した基材でも好適に使用できる。無機物としてはガラス、モルタル、スレート、コンクリート及び瓦等が挙げられる。有機物としては、ポリプロピレン、ポリエチレン、アクリル、ポリカーボネート、ABS、ポリスチレン、PET、ナイロン、ポリエステル、ゴム、及びエラストマーのようなプラスチック成形品およびこれらをフィルム状に加工した製品などが挙げられる(尚、これらは、付着性の点から必要に応じて表面処理されたものであっても良い)。さらに本発明は、トップコートして充分な機能を有するため、エポキシ系塗料やウレタン系塗料、メラミン系塗料などの各種有機系の下塗り剤に対しても使用できる。複合材料としてはFRP、FRTP、積層板および金属と有機物を圧着したサンドイッチ材などが挙げられる。 Examples of the coated object of the multi-component curable resin composition for coating according to the present invention include metals, inorganic substances, organic substances, and composite materials. Examples of the metal include stainless steel, aluminum, tinplate, tin, mild steel plate, copper, brass, various plated steel plates, and titanium. A substrate subjected to a surface treatment such as a chemical conversion treatment or an alumite treatment can also be suitably used. Examples of the inorganic material include glass, mortar, slate, concrete and roof tile. Examples of organic substances include plastic molded products such as polypropylene, polyethylene, acrylic, polycarbonate, ABS, polystyrene, PET, nylon, polyester, rubber, and elastomer, and products obtained by processing these into films (note that these are From the viewpoint of adhesion, it may be subjected to surface treatment if necessary). Furthermore, since the present invention has a sufficient function when applied as a top coat, it can also be used for various organic undercoats such as epoxy paints, urethane paints and melamine paints. Examples of the composite material include FRP, FRTP, a laminate, and a sandwich material in which a metal and an organic material are pressure-bonded.
(用途)
硬化性樹脂組成物の具体的用途としては、携帯電話や(パーソナル)コンピューターの筐体や家電製品等のプラスチック成型体、フラットパネルディスプレイやタッチパネル用のフィルム、テーブルや机、家具などの木材製品さらに各種金属製品への塗装が挙げられる。また、耐候性、耐酸性、耐薬品性、耐擦傷性、耐汚染性に優れる塗膜が得られることから、自動車用トップコートとして使用される自動車クリアーコーティング剤も好ましい用途として挙げられる。(Use)
Specific applications of the curable resin composition include cell phones, (personal) computer casings, plastic molded products such as home appliances, flat panel displays, touch panel films, wood products such as tables, desks, and furniture. Examples include painting on various metal products. In addition, since a coating film excellent in weather resistance, acid resistance, chemical resistance, scratch resistance, and stain resistance can be obtained, an automotive clear coating agent used as a top coat for automobiles is also cited as a preferred application.
本発明に係る自動車クリアーコーティング剤は、本発明のコーティング用多液型硬化性樹脂組成物を含んでなるものである。 The automotive clear coating agent according to the present invention comprises the multi-component curable resin composition for coating according to the present invention.
また、本発明に係る塗装物は、本発明のコーティング用多液型硬化性樹脂組成物を塗装したものである。 Moreover, the coated object which concerns on this invention is what coated the multi-component curable resin composition for coating of this invention.
以下に、本発明を実施例に基づいて説明するが、本発明はこれによって限定されるものではない。 Hereinafter, the present invention will be described based on examples, but the present invention is not limited thereto.
(ビニル系共重合体)
攪拌機、温度計、還流冷却器、窒素ガス導入管および滴下ロートを備えた反応器に表1の(イ)成分を仕込み、窒素ガスを導入しつつ110℃に昇温した後、表1の(ア)成分の混合物を滴下ロートから5時間かけて等速滴下した。次に、(ウ)成分の混合溶液を1時間かけて等速滴下した。その後、引き続き、110℃で2時間攪拌した後に、室温まで冷却した。最後に表1の(エ)成分を加えて攪拌し、共重合体を合成した。(Vinyl copolymer)
In a reactor equipped with a stirrer, a thermometer, a reflux condenser, a nitrogen gas inlet tube and a dropping funnel, the components (a) in Table 1 were charged, and the temperature was raised to 110 ° C. while introducing nitrogen gas. The mixture of components a) was dropped from the dropping funnel at a constant rate over 5 hours. Next, the mixed solution of the component (c) was dropped at a constant rate over 1 hour. Then, after stirring at 110 degreeC for 2 hours continuously, it cooled to room temperature. Finally, component (D) in Table 1 was added and stirred to synthesize a copolymer.
得られた共重合体の固形分濃度、GPCで測定した数平均分子量などを表1に示した。尚、共重合体(A−1〜A−8)は、重合溶剤で固形分濃度が50%となるように一旦希釈して次の配合へと進めた。 Table 1 shows the solid content concentration of the obtained copolymer, the number average molecular weight measured by GPC, and the like. The copolymers (A-1 to A-8) were once diluted with a polymerization solvent so that the solid concentration was 50%, and proceeded to the next blending.
(硬化剤)
表2に示す配合に従い、硬化剤(H−1〜H−7)を調製した。(Curing agent)
Curing agents (H-1 to H-7) were prepared according to the formulation shown in Table 2.
FM−4:ダイセル化学工業(株)製ラクトン変性ヒドロキシエチルメタクリレート
コロネートHX:日本ポリウレタン工業(株)製ポリイソシアネート(NCO%=21.3%)
コロネート2349:日本ポリウレタン工業(株)製ポリイソシアネート(NCO%=12.2%)FM-4: Daicel Chemical Industries Co., Ltd. Lactone Modified Hydroxyethyl Methacrylate Coronate HX: Nippon Polyurethane Industry Co., Ltd. Polyisocyanate (NCO% = 21.3%)
Coronate 2349: Polyisocyanate manufactured by Nippon Polyurethane Industry Co., Ltd. (NCO% = 12.2%)
(物性評価1)
表3に示す配合に従い塗液を調製し、エアスプレー塗布法にて乾燥膜厚が20〜30μmになるようにポリカーボネート樹脂成形板に塗布し、80℃で30分乾燥させ、耐溶剤性、及び、耐薬品性の評価を行った。(Physical property evaluation 1)
A coating solution is prepared according to the formulation shown in Table 3, applied to a polycarbonate resin molded plate by an air spray coating method so that the dry film thickness is 20 to 30 μm, dried at 80 ° C. for 30 minutes, solvent resistance, and The chemical resistance was evaluated.
・耐溶剤性、耐薬品性
ポリカーボネート樹脂成型板に塗布した試験片を室温養生3日後、表3に示す溶剤、薬品をスポットし、耐溶剤性評価の場合は、常温で溶剤が揮発するまで静置し、耐薬品性評価の場合は、80℃で1時間静置した後、脱脂綿で拭取り、塗膜の状態を観察した。
○:変化なし
△:スポット跡が残る
×:塗膜が膨潤(溶解)している
コパトーン(登録商標)及びニベア(登録商標)としては、下記のものを使用した。
コパトーン:コパトーンSPF50(花王(株)製)
ニベア :ニベアSPF47((株)アイ・エー・シー製)・ Solvent resistance, chemical resistance Polycarbonate resin molded plate test piece 3 days after curing at room temperature, spot the solvents and chemicals shown in Table 3 and in the case of solvent resistance evaluation, keep static until the solvent volatilizes at room temperature. In the case of chemical resistance evaluation, the sample was allowed to stand at 80 ° C. for 1 hour, then wiped with absorbent cotton, and the state of the coating film was observed.
○: No change Δ: Spot mark remains ×: Copatone (registered trademark) and Nivea (registered trademark) in which the coating film is swollen (dissolved) were used as follows.
Copatone: Copatone SPF50 (manufactured by Kao Corporation)
Nivea: Nivea SPF47 (manufactured by IC Corporation)
(物性評価2)
カチオン電着塗装されたリン酸亜鉛処理鋼板(日本テストパネル(株)製バルボンド処理板)に、水性ベースコート白(AXUZ−DRY:イサム塗料(株)製)を塗装し、80℃で5分間プレヒートを行った。次に表3に示す配合に従い調製した塗液を、エアスプレー塗布法にて乾燥膜厚が30〜40μmになるように塗布し、140℃で30分焼き付けて塗膜を形成させ、評価を行った。(Physical property evaluation 2)
Aqueous base coat white (AXUZ-DRY: made by Isamu Paint Co., Ltd.) is applied to the cation electrodeposition-coated zinc phosphate treated steel plate (Nippon Test Panel Co., Ltd. Valbond treated plate) and preheated at 80 ° C. for 5 minutes. Went. Next, the coating liquid prepared according to the formulation shown in Table 3 was applied by an air spray coating method so that the dry film thickness was 30 to 40 μm, and baked at 140 ° C. for 30 minutes to form a coating film, which was evaluated. It was.
・耐酸性
室温養生1日後、40%硫酸水溶液をスポットして、70℃で30分間放置したのち、水で硫酸水溶液を洗い落して塗膜の状態を観察した。
○:変化なし
△:スポット跡が残る
×:塗膜が膨潤(溶解)している-After 1 day of acid-resistant room temperature curing, a 40% sulfuric acid aqueous solution was spotted and allowed to stand at 70 ° C. for 30 minutes, and then the sulfuric acid aqueous solution was washed off with water and the state of the coating film was observed.
○: No change Δ: Spot mark remains ×: The coating film is swollen (dissolved)
・耐擦傷性
室温養生3日後、消しゴム摩耗試験機((株)光本製作所製)を用い、スチールウール#0000に250g/cm2の荷重をかけて30回往復させ、塗膜に残った傷の本数を観察した。
○:傷なし
○△:数本の傷
△:数本から10本以内の浅い傷
×:傷が10本以上若しくは深い傷
・傷修復性
Knoop硬度計を用いて塗膜に傷を付け、30分後の傷の状態(修復性)を観察した。
○:傷が回復している
△:傷の面積が減少している
×:変化なし
−:未実施Scratch resistance After 3 days of room temperature curing, using an eraser abrasion tester (manufactured by Mitsumoto Seisakusho Co., Ltd.), the steel wound # 0000 was reciprocated 30 times with a load of 250 g / cm 2 , and the scratches remaining on the coating film Was observed.
○: No scratch ○ △: Several scratches Δ: Shallow scratches of several to 10 or less ×: Ten or more scratches or deep scratches / scratch repairability A scratch is applied to the coating film using a Knoop hardness tester, 30 The condition of the wound (restorability) after a minute was observed.
○: The wound has been recovered Δ: The area of the wound has decreased ×: No change −: Not implemented
・耐汚染性
作製した塗装板を、大阪府摂津市の屋外で30°の角度になるように設置して曝露に供した。2ヶ月間曝露したのち、塗膜表面の汚れ具合を目視にて観察、評価した。
○:汚れがあまり見られず、耐汚染性にすぐれている
△:薄めの汚れが確認できる
×:はっきりと目立った汚れが確認できる-The stain-resistant prepared coated plate was placed at an angle of 30 ° outdoors in Settsu City, Osaka, and exposed. After exposure for 2 months, the degree of contamination on the coating film surface was visually observed and evaluated.
○: Not very dirty and excellent in stain resistance △: Thin stain can be confirmed ×: Clearly noticeable stain can be confirmed
実施例1〜10では、硬化後すぐに高い耐溶剤性、耐酸性及び耐薬品性を示し、また耐擦傷性にも優れたバランスの良い塗膜が得られた。さらには、屋外に供した場合の汚れが付きにくく、耐汚染性に優れていた。また水酸基が炭素数4のアルキレン基を介してアクリロイル基と結合した単量体である4−ヒドロキシブチルアクリレートを使用した実施例5と実施例9、および炭素−炭素二重結合を末端に有するポリラクトン系単量体であるFM−4を使用した実施例7では、耐溶剤性、耐酸性、耐薬品性、耐擦傷性に加え、傷修復性も認められた。
なお、物性評価2において作製した塗装板についても、物性評価1と同様の耐薬品性評価を行ったところ、表3に記載した結果と同じ結果が得られた。
また、ABS樹脂成形板に表3に示す配合に従い塗液を調製し、エアスプレー塗布法にて乾燥膜厚が20〜30μmになるように塗布し、80℃で30分乾燥させ、物性評価2と同様の耐傷性評価を行ったところ、表3に記載した結果と同じ結果が得られた。In Examples 1 to 10, a well-balanced coating film having high solvent resistance, acid resistance and chemical resistance immediately after curing, and excellent scratch resistance was obtained. Furthermore, it was difficult to get dirty when used outdoors, and was excellent in stain resistance. Examples 5 and 9 using 4-hydroxybutyl acrylate, which is a monomer in which a hydroxyl group is bonded to an acryloyl group via an alkylene group having 4 carbon atoms, and a polylactone having a carbon-carbon double bond at the end In Example 7 using FM-4 which is a system monomer, in addition to solvent resistance, acid resistance, chemical resistance and scratch resistance, scratch repairability was also observed.
In addition, when the same chemical-resistance evaluation as the physical property evaluation 1 was performed also about the coating board produced in the physical property evaluation 2, the same result as the result described in Table 3 was obtained.
Also, a coating solution is prepared on an ABS resin molded plate according to the formulation shown in Table 3, and is applied by an air spray coating method so that the dry film thickness is 20 to 30 μm, dried at 80 ° C. for 30 minutes, and physical property evaluation 2 When the same scratch resistance evaluation was performed, the same results as described in Table 3 were obtained.
一方、一分子中の加水分解性基ケイ素基および水酸基が10個に満たない共重合体(A−6)を用いた比較例1では、耐薬品性と耐擦傷性が充分に得られなかった。また一分子中の加水分解性基ケイ素基は10個以上あるものの、水酸基を含まない比較例2では耐汚染性は優れるものの、耐擦傷性が、逆に一分子中の水酸基は10個以上あるものの、加水分解性ケイ素基を含まない比較例3は、耐薬品性及び耐汚染性が極めて劣る結果となった。 On the other hand, in Comparative Example 1 using a copolymer (A-6) having less than 10 hydrolyzable group silicon groups and hydroxyl groups in one molecule, sufficient chemical resistance and scratch resistance were not obtained. . Further, although there are 10 or more hydrolyzable silicon groups in one molecule, in Comparative Example 2 which does not contain a hydroxyl group, the stain resistance is excellent, but the scratch resistance is, on the contrary, there are 10 or more hydroxyl groups in one molecule. However, Comparative Example 3 containing no hydrolyzable silicon group resulted in extremely poor chemical resistance and stain resistance.
以上のように本発明のコーティング用硬化性樹脂組成物は、高い耐溶剤性、耐酸性と耐薬品性を示し、また耐擦傷性及び耐汚染性にも優れるものであり、目的とした課題をバランスよく解決できるコーティング用硬化性樹脂組成物であることが確認された。さらに、共重合体成分として特定の単量体を使用することで、傷修復性も期待できることが確認された。 As described above, the curable resin composition for coating of the present invention exhibits high solvent resistance, acid resistance and chemical resistance, and is excellent in scratch resistance and stain resistance. It was confirmed that it was a curable resin composition for coating that could be solved in a well-balanced manner. Furthermore, it was confirmed that the use of a specific monomer as the copolymer component can also be expected to provide wound repairability.
Claims (7)
R2 a
|
−Si−(OR1)3−a (I)
(式中、R1は水素原子または炭素数1〜10のアルキル基、R2は水素原子または炭素数1〜10のアルキル基、炭素数6〜25のアリール基および炭素数7〜12のアラルキル基から選ばれた1価の炭化水素基を示す。aは0〜2の整数を示す。)
で表される加水分解性ケイ素基を10個以上有し、且つ主鎖末端および/または側鎖に水酸基を10個以上有するビニル系共重合体、
(B)イソシアナート基を2個以上含有する化合物、および、
(C)加水分解性ケイ素基およびイソシアナートの硬化触媒を含有することを特徴とするコーティング用多液型硬化性樹脂組成物であって、
前記ビニル系共重合体(A)が、炭素数が3以上のアルキレン基を介して水酸基が(メタ)アクリロイル基と結合した単量体および/または炭素−炭素二重結合を末端に有するポリラクトン系単量体を共重合することによって得られるものであることを特徴とするコーティング用多液型硬化性樹脂組成物。 (A) The main chain is a (meth) acrylic copolymer chain, and the general formula (I):
R 2 a
|
-Si- (OR 1 ) 3-a (I)
Wherein R 1 is a hydrogen atom or an alkyl group having 1 to 10 carbon atoms, R 2 is a hydrogen atom or an alkyl group having 1 to 10 carbon atoms, an aryl group having 6 to 25 carbon atoms, and an aralkyl having 7 to 12 carbon atoms. And represents a monovalent hydrocarbon group selected from the group a represents an integer of 0 to 2.)
A vinyl-based copolymer having 10 or more hydrolyzable silicon groups represented by the formula, and having 10 or more hydroxyl groups in the main chain terminal and / or side chain,
(B) a compound containing two or more isocyanate groups, and
(C) A multi-component curable resin composition for coating, comprising a hydrolyzable silicon group and an isocyanate curing catalyst ,
The vinyl copolymer (A) is a monomer having a hydroxyl group bonded to a (meth) acryloyl group via an alkylene group having 3 or more carbon atoms and / or a polylactone system having a carbon-carbon double bond at the terminal. A multi-component curable resin composition for coating, which is obtained by copolymerizing monomers .
A coated product coated with the multi-component curable resin composition for coating according to any one of claims 1 to 5 .
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013547194A JP6133785B2 (en) | 2011-11-29 | 2012-11-28 | Curable resin composition for coating and automotive clear coating agent |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011260428 | 2011-11-29 | ||
| JP2011260428 | 2011-11-29 | ||
| JP2011267269 | 2011-12-06 | ||
| JP2011267269 | 2011-12-06 | ||
| JP2013547194A JP6133785B2 (en) | 2011-11-29 | 2012-11-28 | Curable resin composition for coating and automotive clear coating agent |
| PCT/JP2012/080780 WO2013081022A1 (en) | 2011-11-29 | 2012-11-28 | Curable resin composition for coating, and clear coating material for vehicle |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JPWO2013081022A1 JPWO2013081022A1 (en) | 2015-04-27 |
| JP6133785B2 true JP6133785B2 (en) | 2017-05-24 |
Family
ID=48535472
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013547194A Active JP6133785B2 (en) | 2011-11-29 | 2012-11-28 | Curable resin composition for coating and automotive clear coating agent |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JP6133785B2 (en) |
| WO (1) | WO2013081022A1 (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101615713B1 (en) * | 2014-07-25 | 2016-04-26 | 양철호 | Eco-Friendly Inorganic Coating Composition and Preparation Methods Thereof |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3954721B2 (en) * | 1997-04-11 | 2007-08-08 | 株式会社カネカ | Curable composition for top coat and coated product formed by applying the same |
| JPH115943A (en) * | 1997-04-24 | 1999-01-12 | Kansai Paint Co Ltd | Coating composition |
| JPH11100566A (en) * | 1997-09-26 | 1999-04-13 | Nippon Nsc Ltd | Acryl-urethane sealing material composition improved in initial tack |
| JPH11285670A (en) * | 1998-04-01 | 1999-10-19 | Kansai Paint Co Ltd | Formation of double-layered coating film |
| JP2007186690A (en) * | 2005-12-16 | 2007-07-26 | Sanyo Chem Ind Ltd | Resin composition for powder coating |
-
2012
- 2012-11-28 JP JP2013547194A patent/JP6133785B2/en active Active
- 2012-11-28 WO PCT/JP2012/080780 patent/WO2013081022A1/en not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| WO2013081022A1 (en) | 2013-06-06 |
| JPWO2013081022A1 (en) | 2015-04-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO1998022547A1 (en) | Non-staining coating composition | |
| WO2016005115A1 (en) | Two-component paint composition and multilayer coating formation method using this | |
| JP5609355B2 (en) | Urethane coating composition and resin member | |
| JP7777447B2 (en) | Primer composition | |
| JPH1036767A (en) | Two pack type aqueous coating composition | |
| JP4073180B2 (en) | Resin composition for coating and curable coating composition | |
| JP6133785B2 (en) | Curable resin composition for coating and automotive clear coating agent | |
| JP5916711B2 (en) | Multi-component curable coating composition, coating film forming method, and coated body | |
| JP4913941B2 (en) | Weak solvent-based coating for base preparation and paint finishing method using the same | |
| JP4334188B2 (en) | A curable resin composition for top coating and a coated product obtained by applying the same. | |
| JP3702381B2 (en) | Curable composition for top coat and coated product obtained by applying the same | |
| JP5261902B2 (en) | Water-based paint for floor and floor coating method | |
| JPH10219190A (en) | Multi-package water-based coating composition | |
| JP4627143B2 (en) | High weather resistance resin composition for paint | |
| JP2003268323A (en) | Adhesive sheet for outer wall of building | |
| JP3088073B2 (en) | Paint finishing method | |
| JP2002241680A (en) | Coating composition | |
| WO1998046691A1 (en) | Curable composition for topcoating and articles coated therewith | |
| JP2001081393A (en) | Curable composition for top-coat paint and coated product using the same | |
| JP2000160120A (en) | Topcoating composition | |
| JP2007186707A (en) | Resin composition for coating and curable coating composition | |
| JPH10101765A (en) | Curable resin composition and coating material containing the same | |
| KR20110116121A (en) | 2-component urethane paint composition | |
| JP2008169398A (en) | Curable coating composition for floor and method for forming coating film for floor by using the same | |
| JP2008144178A (en) | Curable resin composition for top coating and coated article coated with the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20151102 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20161122 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170118 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20170328 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20170420 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6133785 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |