JP5886202B2 - ガイドワイヤ型ペーシングリード - Google Patents
ガイドワイヤ型ペーシングリード Download PDFInfo
- Publication number
- JP5886202B2 JP5886202B2 JP2012530991A JP2012530991A JP5886202B2 JP 5886202 B2 JP5886202 B2 JP 5886202B2 JP 2012530991 A JP2012530991 A JP 2012530991A JP 2012530991 A JP2012530991 A JP 2012530991A JP 5886202 B2 JP5886202 B2 JP 5886202B2
- Authority
- JP
- Japan
- Prior art keywords
- lead
- contact means
- pacing
- thread
- lead body
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000000576 coating method Methods 0.000 claims description 18
- 239000011248 coating agent Substances 0.000 claims description 12
- 238000009413 insulation Methods 0.000 claims description 11
- 239000012774 insulation material Substances 0.000 claims 1
- 239000002184 metal Substances 0.000 description 13
- 210000001519 tissue Anatomy 0.000 description 13
- 238000001514 detection method Methods 0.000 description 12
- 210000002216 heart Anatomy 0.000 description 8
- 210000001174 endocardium Anatomy 0.000 description 7
- 238000000034 method Methods 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 230000000638 stimulation Effects 0.000 description 7
- 239000000463 material Substances 0.000 description 6
- 238000001356 surgical procedure Methods 0.000 description 6
- 239000004642 Polyimide Substances 0.000 description 5
- 238000002513 implantation Methods 0.000 description 5
- 229920001721 polyimide Polymers 0.000 description 5
- 210000005003 heart tissue Anatomy 0.000 description 4
- 210000003462 vein Anatomy 0.000 description 4
- 230000005540 biological transmission Effects 0.000 description 3
- 210000005242 cardiac chamber Anatomy 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 210000005241 right ventricle Anatomy 0.000 description 3
- 238000004873 anchoring Methods 0.000 description 2
- 125000003118 aryl group Chemical group 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 210000002837 heart atrium Anatomy 0.000 description 2
- 210000003709 heart valve Anatomy 0.000 description 2
- 239000011810 insulating material Substances 0.000 description 2
- 239000012212 insulator Substances 0.000 description 2
- 210000005245 right atrium Anatomy 0.000 description 2
- 230000000451 tissue damage Effects 0.000 description 2
- 231100000827 tissue damage Toxicity 0.000 description 2
- 230000002792 vascular Effects 0.000 description 2
- 238000004804 winding Methods 0.000 description 2
- 239000010963 304 stainless steel Substances 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- 229910000589 SAE 304 stainless steel Inorganic materials 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 210000000709 aorta Anatomy 0.000 description 1
- 238000003491 array Methods 0.000 description 1
- 206010003119 arrhythmia Diseases 0.000 description 1
- 230000001746 atrial effect Effects 0.000 description 1
- 238000013158 balloon valvuloplasty Methods 0.000 description 1
- 238000010009 beating Methods 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 239000010839 body fluid Substances 0.000 description 1
- 230000000747 cardiac effect Effects 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 230000004087 circulation Effects 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 210000004351 coronary vessel Anatomy 0.000 description 1
- 238000012937 correction Methods 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 230000001862 defibrillatory effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 238000010292 electrical insulation Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 238000007667 floating Methods 0.000 description 1
- IPENDKRRWFURRE-UHFFFAOYSA-N fluoroimide Chemical compound C1=CC(F)=CC=C1N1C(=O)C(Cl)=C(Cl)C1=O IPENDKRRWFURRE-UHFFFAOYSA-N 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 238000010329 laser etching Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000028161 membrane depolarization Effects 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 210000005259 peripheral blood Anatomy 0.000 description 1
- 239000011886 peripheral blood Substances 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 230000002787 reinforcement Effects 0.000 description 1
- 210000005247 right atrial appendage Anatomy 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000005482 strain hardening Methods 0.000 description 1
- 210000001321 subclavian vein Anatomy 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 210000005166 vasculature Anatomy 0.000 description 1
- 230000008320 venous blood flow Effects 0.000 description 1
- 230000002861 ventricular Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/05—Electrodes for implantation or insertion into the body, e.g. heart electrode
- A61N1/056—Transvascular endocardial electrode systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/09—Guide wires
- A61M2025/09058—Basic structures of guide wires
- A61M2025/09066—Basic structures of guide wires having a coil without a core possibly combined with a sheath
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/09—Guide wires
- A61M2025/09058—Basic structures of guide wires
- A61M2025/09083—Basic structures of guide wires having a coil around a core
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/09—Guide wires
- A61M2025/09175—Guide wires having specific characteristics at the distal tip
- A61M2025/09183—Guide wires having specific characteristics at the distal tip having tools at the distal tip
Description
Saulsonら、米国特許第4,530,368号明細書、「一時的バイポーラペーシングリード」
Tarjanら、米国特許第4,475,560号明細書、「一時的ペーシングリード組立体」
Parsonnet、米国特許第4,541,440号明細書、「バイポーラ心外膜一時的ペーシングリード」
Barringtonら、米国特許第4,602,645号明細書、「房室ペーシングカテーテル」
Williams、米国特許第4,338,947号明細書、「ポジティブ固定心臓ワイヤ」
Authら、米国特許出願公開第2007/0088355号明細書、「経中隔左心房アクセス及び中隔閉鎖」
Pedersenら、米国特許出願公開第2005/00075662号明細書、「弁形成術カテーテル」
Schwartzら、米国特許出願公開第2002/0098307号明細書、「医療用バルーン及びカテーテルに使用可能な材料」
Schwartzら、米国特許出願公開第2009/0018608号明細書、「高血圧制御用心臓刺激装置及び方法」
Pedersenら、米国特許出願公開第2005/0090846号明細書、「弁形成術装置及び方法」
Claims (6)
- ガイドワイヤ型ペーシングリードであって、
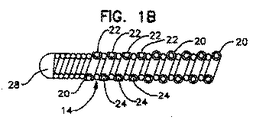
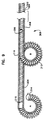
遠位端及び近位端を有する、複糸線の、細長い、螺旋巻リード本体を含み、
螺旋巻リード本体は、電気的に絶縁され管腔を画成する少なくとも第1の糸線及び第2の糸線を含み、第1の糸線は、第1の接触手段及び第1のコネクタ手段に電気的に結合され、第2の糸線は、第2の接触手段及び第2のコネクタ手段に結合され、第1の接触手段と第2の接触手段は、第1の接触手段及び第2の接触手段の両方が組織に適用されたとき、これらの間に電気的短絡経路が存在しないように、十分に間隔を隔てられ、
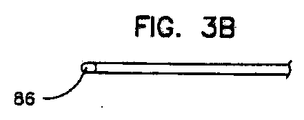
更に、リード本体は、リード本体の遠位端に非外傷性先端を含み、第1の糸線及び第2の糸線は、異なる直径を有し、リード本体の長さの大部分にわたって巻き合わされ、上記糸線の一方又は他方は、リードの遠位端のすぐ近くで終わっている、
ガイドワイヤ型ペーシングリード。 - 上記第1の糸線及び第2の糸線は、糸線の電気絶縁被覆によって電気的に絶縁されている、請求項1に記載のリード。
- 上記第1の接触手段又は第2の接触手段の一方は、上記リード本体によって画成された外側へ片寄った円形構造体である、請求項1に記載のリード。
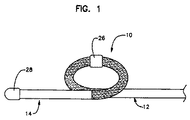
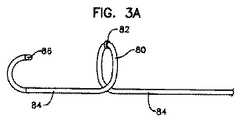
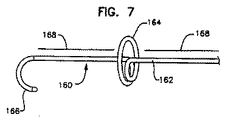
- 上記リード本体はループを画成し、上記糸線の一方は、上記ループによって支持されたリング形接触手段に電気的に結合される、請求項1に記載のリード。
- 少なくとも上記第1の糸線及び第2の糸線のうちの一方は、接触手段を画成するために除去された絶縁材の少なくとも一部分を有する、請求項1に記載のリード。
- 上記螺旋巻糸線によって画成された上記管腔内に配置され、且つ非外傷性先端に電気的に結合されている、電気的絶縁層を表面に備える導電性の芯線を更に含む、請求項1に記載のペーシングリード。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US24511709P | 2009-09-23 | 2009-09-23 | |
| US61/245,117 | 2009-09-23 | ||
| PCT/US2010/049789 WO2011037978A2 (en) | 2009-09-23 | 2010-09-22 | Guidewire-style pacing lead |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2013505781A JP2013505781A (ja) | 2013-02-21 |
| JP2013505781A5 JP2013505781A5 (ja) | 2013-11-14 |
| JP5886202B2 true JP5886202B2 (ja) | 2016-03-16 |
Family
ID=43757300
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2012530991A Active JP5886202B2 (ja) | 2009-09-23 | 2010-09-22 | ガイドワイヤ型ペーシングリード |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US9387323B2 (ja) |
| EP (1) | EP2480282B1 (ja) |
| JP (1) | JP5886202B2 (ja) |
| CN (1) | CN102711902B (ja) |
| CA (1) | CA2775081A1 (ja) |
| ES (1) | ES2550034T3 (ja) |
| WO (1) | WO2011037978A2 (ja) |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5886202B2 (ja) | 2009-09-23 | 2016-03-16 | レイク リージョン マニュファクチュアリング インコーポレイテッド | ガイドワイヤ型ペーシングリード |
| US20120109190A1 (en) * | 2010-05-06 | 2012-05-03 | Cvdevices, Llc (A California Limited Liability Company) | Devices, systems, and methods for closing an aperture in a bodily tissue |
| US10086191B2 (en) | 2012-04-19 | 2018-10-02 | Medtronic, Inc. | Medical leads having a distal body and an openly coiled filar |
| US9545207B2 (en) | 2012-07-25 | 2017-01-17 | Cardiac Pacemakers, Inc. | Temporary implantable medical electrical stimulation lead |
| AU2013318149B2 (en) * | 2012-09-19 | 2016-09-22 | Cardiac Pacemakers, Inc. | Fixation mechanisms for temporary implantable medical electrical stimulation leads |
| US9351783B2 (en) | 2013-05-01 | 2016-05-31 | Medtronic Cryocath Lp | Diagnostic guidewire for cryoablation sensing and pressure monitoring |
| EP3212068B1 (en) | 2014-10-31 | 2020-11-25 | Lake Region Medical, Inc. | Fiber bragg grating multi-point pressure sensing guidewire with birefringent component |
| CN104399174A (zh) * | 2014-11-28 | 2015-03-11 | 中山市普利斯微创介入医械有限公司 | 绝缘导丝 |
| US20170189674A1 (en) * | 2016-01-04 | 2017-07-06 | Medtronic, Inc. | Medical electrical lead |
| US10881851B2 (en) * | 2016-03-18 | 2021-01-05 | Cardiac Interventions And Aviation Llc | Pacing guidewire |
| US10773076B2 (en) | 2016-07-05 | 2020-09-15 | Wesley Robert Pedersen | Temporary pacing lead |
| WO2018017731A1 (en) | 2016-07-19 | 2018-01-25 | Cygnus Investment Corporation | Guidewire having conductive elements |
| EP3856326A1 (en) * | 2018-09-27 | 2021-08-04 | Verily Life Sciences LLC | Implantable medical devices with microfabricated leads |
| JP2022530872A (ja) | 2019-04-22 | 2022-07-04 | ボストン サイエンティフィック サイムド,インコーポレイテッド | 癌治療のための電気刺激機器 |
| WO2020219517A2 (en) | 2019-04-23 | 2020-10-29 | Boston Scientific Scimed, Inc. | Electrical stimulation for cancer treatment with internal and external electrodes |
| EP3958956B1 (en) | 2019-04-23 | 2024-02-28 | Boston Scientific Scimed Inc. | Electrodes for electrical stimulation to treat cancer |
| EP3958960A1 (en) | 2019-04-23 | 2022-03-02 | Boston Scientific Scimed Inc. | Electrical stimulation with thermal treatment or thermal monitoring |
| EP4110455A1 (en) | 2020-02-24 | 2023-01-04 | Boston Scientific Scimed, Inc. | Systems and methods for treatment of pancreatic cancer |
| WO2022094459A1 (en) * | 2020-11-02 | 2022-05-05 | Edwards Lifesciences Corporation | Assemblies, apparatuses, and methods for electrically stimulating the body |
Family Cites Families (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4317459A (en) * | 1979-06-28 | 1982-03-02 | Medtronic, Inc. | Fixation loop for transvenous leads |
| US4338947A (en) | 1980-11-03 | 1982-07-13 | Medtronic, Inc. | Positive fixation heart wire |
| US4475560A (en) | 1982-04-29 | 1984-10-09 | Cordis Corporation | Temporary pacing lead assembly |
| US4437474A (en) * | 1982-07-16 | 1984-03-20 | Cordis Corporation | Method for making multiconductor coil and the coil made thereby |
| US4602645A (en) | 1982-12-16 | 1986-07-29 | C. R. Bard, Inc. | Atrio-ventricular pacing catheter |
| US4530368A (en) | 1984-05-24 | 1985-07-23 | Cordis Corporation | Temporary bipolar pacing lead |
| US4541440A (en) | 1984-11-14 | 1985-09-17 | Cordis Corporation | Bipolar epicardial temporary pacing lead |
| US5154705A (en) * | 1987-09-30 | 1992-10-13 | Lake Region Manufacturing Co., Inc. | Hollow lumen cable apparatus |
| US4972848A (en) * | 1989-08-23 | 1990-11-27 | Medtronic, Inc. | Medical electrical lead with polymeric monolithic controlled release device and method of manufacture |
| JPH066170B2 (ja) * | 1991-08-28 | 1994-01-26 | 中島 博 | ペースメーカのペーシング・リード |
| JPH0778282B2 (ja) | 1992-07-24 | 1995-08-23 | 株式会社クシタニ | つなぎ服 |
| US5354327A (en) | 1993-04-07 | 1994-10-11 | Medtronic, Inc. | Conductor coil with specific ratio of torque to bending stiffness |
| US5824030A (en) * | 1995-12-21 | 1998-10-20 | Pacesetter, Inc. | Lead with inter-electrode spacing adjustment |
| US5851226A (en) | 1996-10-22 | 1998-12-22 | Medtronic, Inc. | Temporary transvenous endocardial lead |
| WO1998040122A1 (en) | 1997-03-14 | 1998-09-17 | University Of Alabama At Birmingham Research Foundation | Method and apparatus for treating cardiac arrhythmia |
| US6161029A (en) | 1999-03-08 | 2000-12-12 | Medtronic, Inc. | Apparatus and method for fixing electrodes in a blood vessel |
| US6400992B1 (en) * | 1999-03-18 | 2002-06-04 | Medtronic, Inc. | Co-extruded, multi-lumen medical lead |
| US6408213B1 (en) * | 1999-09-29 | 2002-06-18 | Cardiac Pacemakers, Inc. | Low profile, ventricular, transvenous, epicardial defibrillation lead |
| US6584362B1 (en) | 2000-08-30 | 2003-06-24 | Cardiac Pacemakers, Inc. | Leads for pacing and/or sensing the heart from within the coronary veins |
| US20020098307A1 (en) | 2000-10-09 | 2002-07-25 | Schwartz Robert S. | Material useable for medical balloons and catheters |
| US7020529B2 (en) * | 2001-05-02 | 2006-03-28 | Gore Enterprise Holdings, Inc. | Defibrillation electrode cover |
| US6978185B2 (en) * | 2001-11-09 | 2005-12-20 | Oscor Inc. | Multifilar conductor for cardiac leads |
| US7904178B2 (en) * | 2002-04-11 | 2011-03-08 | Medtronic, Inc. | Medical electrical lead body designs incorporating energy dissipating shunt |
| US6968237B2 (en) * | 2002-05-22 | 2005-11-22 | Pacesetter, Inc. | Implantable coronary sinus lead and lead system |
| US7313445B2 (en) * | 2002-09-26 | 2007-12-25 | Medtronic, Inc. | Medical lead with flexible distal guidewire extension |
| US7031777B2 (en) | 2002-09-27 | 2006-04-18 | Medtronic, Inc. | Cardiac vein lead with flexible anode and method for forming same |
| US8010207B2 (en) * | 2002-10-31 | 2011-08-30 | Medtronic, Inc. | Implantable medical lead designs |
| US7184839B2 (en) * | 2002-12-16 | 2007-02-27 | Medtronic, Inc. | Catheter-delivered cardiac lead |
| US8021359B2 (en) | 2003-02-13 | 2011-09-20 | Coaptus Medical Corporation | Transseptal closure of a patent foramen ovale and other cardiac defects |
| US7065411B2 (en) * | 2003-04-23 | 2006-06-20 | Medtronic, Inc. | Electrical medical leads employing conductive aerogel |
| US7744620B2 (en) | 2003-07-18 | 2010-06-29 | Intervalve, Inc. | Valvuloplasty catheter |
| US8086315B2 (en) | 2004-02-12 | 2011-12-27 | Asap Medical, Inc. | Cardiac stimulation apparatus and method for the control of hypertension |
| US7225035B2 (en) * | 2004-06-24 | 2007-05-29 | Medtronic, Inc. | Multipolar medical electrical lead |
| US7067765B2 (en) | 2004-08-23 | 2006-06-27 | Medtronic, Inc. | Methods for forming electrically active surfaces for medical electrical leads |
| US7627382B2 (en) | 2005-05-25 | 2009-12-01 | Lake Region Manufacturing, Inc. | Medical devices with aromatic polyimide coating |
| US7917213B2 (en) * | 2005-11-04 | 2011-03-29 | Kenergy, Inc. | MRI compatible implanted electronic medical lead |
| US7860580B2 (en) * | 2006-04-24 | 2010-12-28 | Medtronic, Inc. | Active fixation medical electrical lead |
| US8150536B2 (en) * | 2006-06-12 | 2012-04-03 | Oscor Inc. | Low profile active fixation cardiac lead having torque transmitting means |
| US8155756B2 (en) * | 2007-02-16 | 2012-04-10 | Pacesetter, Inc. | Motion-based optimization for placement of cardiac stimulation electrodes |
| ES2615402T3 (es) * | 2007-03-19 | 2017-06-06 | Boston Scientific Neuromodulation Corporation | Cables compatibles con IRM y RF |
| US20080242976A1 (en) | 2007-03-30 | 2008-10-02 | Proteus Biomedical, Inc. | Electric field tomography |
| US20090088827A1 (en) * | 2007-10-02 | 2009-04-02 | Cardiac Pacemakers, Inc | Lead assembly providing sensing or stimulation of spaced-apart myocardial contact areas |
| US8574849B2 (en) | 2007-10-03 | 2013-11-05 | The University Of Vermont And State Agriculture College | Methods of detection of factor XIa and tissue factor |
| US8285395B2 (en) * | 2008-11-29 | 2012-10-09 | Medtronic, Inc. | Conductive couplings, and components thereof, for medical electrical leads |
| JP5886202B2 (ja) | 2009-09-23 | 2016-03-16 | レイク リージョン マニュファクチュアリング インコーポレイテッド | ガイドワイヤ型ペーシングリード |
| WO2011049684A1 (en) * | 2009-10-19 | 2011-04-28 | Cardiac Pacemakers, Inc. | Mri compatible tachycardia lead |
-
2010
- 2010-09-22 JP JP2012530991A patent/JP5886202B2/ja active Active
- 2010-09-22 CA CA2775081A patent/CA2775081A1/en not_active Abandoned
- 2010-09-22 EP EP10819375.6A patent/EP2480282B1/en active Active
- 2010-09-22 US US12/887,871 patent/US9387323B2/en active Active
- 2010-09-22 WO PCT/US2010/049789 patent/WO2011037978A2/en active Application Filing
- 2010-09-22 CN CN201080045698.9A patent/CN102711902B/zh active Active
- 2010-09-22 ES ES10819375.6T patent/ES2550034T3/es active Active
Also Published As
| Publication number | Publication date |
|---|---|
| EP2480282A4 (en) | 2013-03-13 |
| EP2480282B1 (en) | 2015-08-26 |
| EP2480282A2 (en) | 2012-08-01 |
| JP2013505781A (ja) | 2013-02-21 |
| WO2011037978A8 (en) | 2012-07-19 |
| CN102711902A (zh) | 2012-10-03 |
| US9387323B2 (en) | 2016-07-12 |
| WO2011037978A2 (en) | 2011-03-31 |
| ES2550034T3 (es) | 2015-11-04 |
| CA2775081A1 (en) | 2011-03-31 |
| WO2011037978A3 (en) | 2011-09-09 |
| CN102711902B (zh) | 2015-09-16 |
| US20110071608A1 (en) | 2011-03-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5886202B2 (ja) | ガイドワイヤ型ペーシングリード | |
| US11478636B2 (en) | Tube-cut helical fixation anchor for electrotherapy device | |
| US6871085B2 (en) | Cardiac vein lead and guide catheter | |
| US6574512B1 (en) | Lead system with main lead and transverse lead | |
| US11383080B2 (en) | Implantable medical devices and methods for making and delivering implantable medical devices | |
| US6544270B1 (en) | Multi-lumen cardiac catheter and system | |
| US8509916B2 (en) | Bilumen guide catheters for accessing cardiac sites | |
| US5231996A (en) | Removable endocardial lead | |
| US7801622B2 (en) | Medical electrical lead and delivery system | |
| US20090259283A1 (en) | Sheathed lead for pacing or defibrillation | |
| US9265937B2 (en) | Implantable indifferent reference electrode pole | |
| JP2008539031A (ja) | 心臓箇所にアクセスするためのガイドカテーテル | |
| WO2006055556A2 (en) | Method and apparatus for reliably placing and adjusting a left ventricular pacemaker lead | |
| US20240058603A1 (en) | Temporary pacing lead | |
| US20100228330A1 (en) | Lead configured for hisian, para-hisian, rv septum and rv outflow tract pacing | |
| US8750996B2 (en) | Medical implantable lead | |
| US20220118261A1 (en) | Curled shaft temporary pacing lead |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130924 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20130924 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140618 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20140620 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20140918 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20140926 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20141020 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20141027 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20141218 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20150525 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20150825 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20151125 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20160112 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20160210 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5886202 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |