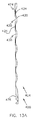JP5406840B2 - 眼内移植片 - Google Patents
眼内移植片 Download PDFInfo
- Publication number
- JP5406840B2 JP5406840B2 JP2010527081A JP2010527081A JP5406840B2 JP 5406840 B2 JP5406840 B2 JP 5406840B2 JP 2010527081 A JP2010527081 A JP 2010527081A JP 2010527081 A JP2010527081 A JP 2010527081A JP 5406840 B2 JP5406840 B2 JP 5406840B2
- Authority
- JP
- Japan
- Prior art keywords
- spine
- schlemm
- intraocular implant
- canal
- implant
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000007943 implant Substances 0.000 claims description 273
- 239000012530 fluid Substances 0.000 claims description 29
- 238000004891 communication Methods 0.000 claims description 28
- 239000003814 drug Substances 0.000 claims description 18
- 239000000463 material Substances 0.000 claims description 17
- 229940124597 therapeutic agent Drugs 0.000 claims description 17
- 208000010412 Glaucoma Diseases 0.000 claims description 16
- 150000003180 prostaglandins Chemical class 0.000 claims description 6
- GGXICVAJURFBLW-CEYXHVGTSA-N latanoprost Chemical compound CC(C)OC(=O)CCC\C=C/C[C@H]1[C@@H](O)C[C@@H](O)[C@@H]1CC[C@@H](O)CCC1=CC=CC=C1 GGXICVAJURFBLW-CEYXHVGTSA-N 0.000 claims description 5
- 229960001160 latanoprost Drugs 0.000 claims description 5
- 239000012781 shape memory material Substances 0.000 claims description 2
- 210000001742 aqueous humor Anatomy 0.000 description 69
- 210000001508 eye Anatomy 0.000 description 60
- 210000002159 anterior chamber Anatomy 0.000 description 42
- 210000001585 trabecular meshwork Anatomy 0.000 description 25
- 210000001519 tissue Anatomy 0.000 description 22
- 230000002093 peripheral effect Effects 0.000 description 18
- 238000000034 method Methods 0.000 description 16
- 210000004087 cornea Anatomy 0.000 description 12
- 210000000695 crystalline len Anatomy 0.000 description 12
- 239000007788 liquid Substances 0.000 description 11
- 238000005452 bending Methods 0.000 description 10
- 230000004410 intraocular pressure Effects 0.000 description 10
- 239000011162 core material Substances 0.000 description 9
- 238000003698 laser cutting Methods 0.000 description 7
- 210000003462 vein Anatomy 0.000 description 7
- 238000004804 winding Methods 0.000 description 7
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 6
- 210000004240 ciliary body Anatomy 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 238000011282 treatment Methods 0.000 description 5
- 210000002919 epithelial cell Anatomy 0.000 description 4
- 235000015097 nutrients Nutrition 0.000 description 4
- 230000008320 venous blood flow Effects 0.000 description 4
- 239000002699 waste material Substances 0.000 description 4
- 206010030348 Open-Angle Glaucoma Diseases 0.000 description 3
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 230000009975 flexible effect Effects 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 230000003068 static effect Effects 0.000 description 3
- 238000001356 surgical procedure Methods 0.000 description 3
- 229920001187 thermosetting polymer Polymers 0.000 description 3
- 229910052719 titanium Inorganic materials 0.000 description 3
- 239000010936 titanium Substances 0.000 description 3
- 201000004569 Blindness Diseases 0.000 description 2
- 229910045601 alloy Inorganic materials 0.000 description 2
- 239000000956 alloy Substances 0.000 description 2
- 230000017531 blood circulation Effects 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- 230000001886 ciliary effect Effects 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 238000002513 implantation Methods 0.000 description 2
- 239000007769 metal material Substances 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- 229910001000 nickel titanium Inorganic materials 0.000 description 2
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 description 2
- 210000001328 optic nerve Anatomy 0.000 description 2
- 210000000056 organ Anatomy 0.000 description 2
- 238000005192 partition Methods 0.000 description 2
- 230000003014 reinforcing effect Effects 0.000 description 2
- GDOPTJXRTPNYNR-UHFFFAOYSA-N CC1CCCC1 Chemical compound CC1CCCC1 GDOPTJXRTPNYNR-UHFFFAOYSA-N 0.000 description 1
- 229910000990 Ni alloy Inorganic materials 0.000 description 1
- 206010036346 Posterior capsule opacification Diseases 0.000 description 1
- 229910001069 Ti alloy Inorganic materials 0.000 description 1
- 229910010380 TiNi Inorganic materials 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000000560 biocompatible material Substances 0.000 description 1
- 238000007664 blowing Methods 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000002872 contrast media Substances 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000007599 discharging Methods 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000004438 eyesight Effects 0.000 description 1
- 210000000887 face Anatomy 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 238000007726 management method Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 230000001991 pathophysiological effect Effects 0.000 description 1
- 230000005043 peripheral vision Effects 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 201000006366 primary open angle glaucoma Diseases 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 125000003259 prostaglandin group Chemical class 0.000 description 1
- 230000001953 sensory effect Effects 0.000 description 1
- 239000004071 soot Substances 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 229910052715 tantalum Inorganic materials 0.000 description 1
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 230000004797 therapeutic response Effects 0.000 description 1
- 210000004127 vitreous body Anatomy 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting-in contact lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/00781—Apparatus for modifying intraocular pressure, e.g. for glaucoma treatment
Description
本願は、2007年9月24日出願の米国特許出願連続番号11/860,318号および2008年3月4日出願の米国特許出願連続番号61/033,746号の優先権を主張するものであり、両出願の開示内容はここに引例に挙げることにより本件の一部を構成しているものとする。
本件明細書中で言及される刊行物および特許出願についてここに引例に挙げることにより本件の一部に編入するにあたり、その程度はいずれも、ここで援用されている個々の刊行物または特許出願を引例に挙げて本件の一部を構成していると特に個別的に示してある限りにおいてという意味である。
Claims (16)
- 少なくとも一部が眼のシュレム管の或る部位に載置されるのに好適な構成の眼内移植片であって、前記移植片は、
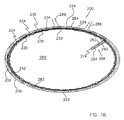
湾曲した容積部の態様で延びており、前記容積部の長軸線が円弧の形状である本体部と、
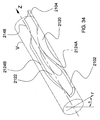
前記本体部に形成された複数の開放領域と、複数の背骨状部と、複数の支柱領域とを備えており、前記開放領域は前記湾曲した容積部を規定している表面の50%を超える領域に亘って広がっており、前記開放領域は、前記本体部に形成された開放流路と、該開放流路の反対側に形成された複数の開口を有し、前記支柱領域は前記開口を包囲し、前記背骨状部は前記開口の間にあり、
前記本体部は直径が0.005インチから0.04インチの間であることを特徴とする、眼内移植片。 - 前記開放流路は、前記湾曲した容積部の最大半径部位に沿って形成されていることを特徴とする、請求項2に記載の移植片。
- 前記移植片は形状記憶素材から前記湾曲した容積部に概ね等しい形状に形成されていることを特徴とする、請求項1に記載の移植片。
- 前記長軸線に直交している横断面で素材が空乏ではない率は50%未満であり、この条件は前記移植片の90%を超える領域で満たされていることを特徴とする、請求項1に記載の移植片。
- 前記移植片は、中心角度60度から180度の円弧に亘って延びていることを特徴とする、請求項1に記載の移植片。
- 前記本体部の一方端に位置して本体部と流体連絡状態にあるとともに、前記円弧を基点に内向きに延びている入口部を更に備えている、請求項1に記載の移植片。
- 前記入口部は、入口部と本体部とを接続する点から引かれている正接線から90度の角度で延びていることを特徴とする、請求項6に記載の移植片。
- 前記入口部の長さは前記本体部の直径よりも大きいことを特徴とする、請求項6に記載の移植片。
- 前記入口部はコイルとして形成されていることを特徴とする、請求項6に記載の移植片。
- 前記本体部の一方端に位置している入口部を更に備えており、前記入口部は前記本体部と流体連絡状態である少なくとも1個の長軸線方向区分が設けられていることを特徴とする、請求項1に記載の移植片。
- 前記移植片はその一方端に鈍先端部が設けられていることを特徴とする、請求項1に記載の移植片。
- 前記鈍先端部の中を通る管腔が形成されていることを特徴とする、請求項11に記載の移植片。
- 治療剤が前記本体部上に被膜堆積されていることを特徴とする、請求項1に記載の移植片。
- 前記治療剤の具体例として緑内障治療薬が挙げられる、請求項13に記載の移植片。
- 前記緑内障治療薬の具体例としてプロスタグラジンアナログが挙げられることを特徴とする、請求項14に記載の移植片。
- 前記プロスタグラジンアナログの具体例としてラタノプロストが挙げられることを特徴とする、請求項15に記載の移植片。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/860,318 | 2007-09-24 | ||
| US11/860,318 US7740604B2 (en) | 2007-09-24 | 2007-09-24 | Ocular implants for placement in schlemm's canal |
| US3374608P | 2008-03-04 | 2008-03-04 | |
| US61/033,746 | 2008-03-04 | ||
| PCT/US2008/077380 WO2009042596A2 (en) | 2007-09-24 | 2008-09-23 | Ocular implants and methods |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013228621A Division JP5788459B2 (ja) | 2007-09-24 | 2013-11-01 | 眼内移植片 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2010540096A JP2010540096A (ja) | 2010-12-24 |
| JP2010540096A5 JP2010540096A5 (ja) | 2011-11-10 |
| JP5406840B2 true JP5406840B2 (ja) | 2014-02-05 |
Family
ID=40380344
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2010527081A Active JP5406840B2 (ja) | 2007-09-24 | 2008-09-23 | 眼内移植片 |
| JP2013228621A Active JP5788459B2 (ja) | 2007-09-24 | 2013-11-01 | 眼内移植片 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013228621A Active JP5788459B2 (ja) | 2007-09-24 | 2013-11-01 | 眼内移植片 |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP2194941B1 (ja) |
| JP (2) | JP5406840B2 (ja) |
| CN (2) | CN101868201B (ja) |
| AU (1) | AU2008304581B2 (ja) |
| CA (1) | CA2700503C (ja) |
| WO (1) | WO2009042596A2 (ja) |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7909789B2 (en) | 2006-06-26 | 2011-03-22 | Sight Sciences, Inc. | Intraocular implants and methods and kits therefor |
| AU2010271218B2 (en) * | 2009-07-09 | 2017-02-02 | Alcon Inc. | Ocular implants and methods for delivering ocular implants into the eye |
| US8951221B2 (en) * | 2009-08-20 | 2015-02-10 | Grieshaber Ophthalmic Research Foundation | Method and device for the treatment of glaucoma |
| JP5856569B2 (ja) | 2010-02-05 | 2016-02-10 | サイト サイエンシーズ, インコーポレイテッド | 眼内圧を低減するためのデバイスと、それを含むキット |
| EP2677981B1 (de) | 2011-02-23 | 2020-04-01 | Grieshaber Ophthalmic Research Foundation | Implantat zur behandlung von glaukom |
| AU2012308317B2 (en) * | 2011-09-14 | 2017-01-05 | Forsight Vision5, Inc. | Ocular insert apparatus and methods |
| EP4302734A3 (en) | 2012-03-20 | 2024-03-20 | Sight Sciences, Inc. | Ocular delivery systems and methods |
| US9358156B2 (en) | 2012-04-18 | 2016-06-07 | Invantis, Inc. | Ocular implants for delivery into an anterior chamber of the eye |
| US9462973B2 (en) * | 2012-07-27 | 2016-10-11 | Biocrine Ab | Methods for monitoring physiological status of a body organ |
| EP3094291A4 (en) * | 2014-01-14 | 2017-09-20 | University of Utah Research Foundation | Schlemm's canal stent-sieve |
| JP6099589B2 (ja) * | 2014-03-25 | 2017-03-22 | 株式会社三共 | 遊技機 |
| US10299958B2 (en) | 2015-03-31 | 2019-05-28 | Sight Sciences, Inc. | Ocular delivery systems and methods |
| EP4265231A3 (en) * | 2015-08-14 | 2023-12-20 | Alcon Inc. | Ocular implant with pressure sensor |
| US11504270B1 (en) | 2019-09-27 | 2022-11-22 | Sight Sciences, Inc. | Ocular delivery systems and methods |
| WO2022149249A1 (ja) * | 2021-01-07 | 2022-07-14 | 株式会社ドックスネット | インプラント、及びインプラントシステム |
| CN114652488A (zh) * | 2022-03-22 | 2022-06-24 | 海思盖德(苏州)生物医学科技有限公司 | 一种眼内植入物及植入物外表复合式药物涂覆工艺 |
| CN115006103B (zh) * | 2022-06-10 | 2023-07-18 | 健诺维(成都)生物科技有限公司 | 眼部植入管 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4733665C2 (en) * | 1985-11-07 | 2002-01-29 | Expandable Grafts Partnership | Expandable intraluminal graft and method and apparatus for implanting an expandable intraluminal graft |
| US4968296A (en) | 1989-12-20 | 1990-11-06 | Robert Ritch | Transscleral drainage implant device for the treatment of glaucoma |
| US5180362A (en) | 1990-04-03 | 1993-01-19 | Worst J G F | Gonio seton |
| US5360399A (en) * | 1992-01-10 | 1994-11-01 | Robert Stegmann | Method and apparatus for maintaining the normal intraocular pressure |
| US5911732A (en) * | 1997-03-10 | 1999-06-15 | Johnson & Johnson Interventional Systems, Co. | Articulated expandable intraluminal stent |
| EP0898947A3 (de) * | 1997-08-15 | 1999-09-08 | GRIESHABER & CO. AG SCHAFFHAUSEN | Verfahren und Vorrichtung zum Verbessern des Kammerwasserabflusses in einem Auge |
| DE19840047B4 (de) * | 1998-09-02 | 2004-07-08 | Neuhann, Thomas, Prof.Dr.med. | Vorrichtung zur gezielten Verbesserung und/oder dauerhaften Gewährleistung des Durchlässigkeitsvermögens für Augenkammerwasser durch das Trabekelwerk in den Schlemmschen Kanal |
| EP2260804B2 (en) | 1999-04-26 | 2022-03-02 | Glaukos Corporation | Trabeculotomy device for treating glaucoma |
| US20050119601A9 (en) * | 1999-04-26 | 2005-06-02 | Lynch Mary G. | Shunt device and method for treating glaucoma |
| MXPA04001330A (es) * | 2001-08-16 | 2004-05-05 | Gmp Vision Solutions Inc | DISPOSITIVO DE DERIVACION MEJORADO Y MeTODO PARA EL TRATAMIENTO DEL GLAUCOMA. |
| US7799336B2 (en) * | 2004-04-30 | 2010-09-21 | Allergan, Inc. | Hypotensive lipid-containing biodegradable intraocular implants and related methods |
| KR20070092279A (ko) * | 2004-12-16 | 2007-09-12 | 아이싸이언스 인터벤셔날 코포레이션 | 녹내장 치료를 위한 안과 이식물 |
| WO2007035356A2 (en) * | 2005-09-16 | 2007-03-29 | Bg Implant, Inc. | Glaucoma treatment devices and methods |
| AU2006304553A1 (en) * | 2005-10-14 | 2007-04-26 | Alcon, Inc. | Method for treating primary and secondary forms of glaucoma |
-
2008
- 2008-09-23 JP JP2010527081A patent/JP5406840B2/ja active Active
- 2008-09-23 WO PCT/US2008/077380 patent/WO2009042596A2/en active Application Filing
- 2008-09-23 CN CN200880116684.4A patent/CN101868201B/zh active Active
- 2008-09-23 EP EP08833245.7A patent/EP2194941B1/en active Active
- 2008-09-23 CA CA2700503A patent/CA2700503C/en active Active
- 2008-09-23 CN CN201410095603.4A patent/CN103932840B/zh active Active
- 2008-09-23 AU AU2008304581A patent/AU2008304581B2/en active Active
-
2013
- 2013-11-01 JP JP2013228621A patent/JP5788459B2/ja active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN101868201A (zh) | 2010-10-20 |
| CN103932840B (zh) | 2017-04-12 |
| EP2194941A2 (en) | 2010-06-16 |
| AU2008304581A1 (en) | 2009-04-02 |
| WO2009042596A2 (en) | 2009-04-02 |
| CN103932840A (zh) | 2014-07-23 |
| CN101868201B (zh) | 2014-04-16 |
| CA2700503A1 (en) | 2009-04-02 |
| JP2014061406A (ja) | 2014-04-10 |
| CA2700503C (en) | 2016-05-24 |
| AU2008304581B2 (en) | 2013-11-21 |
| JP5788459B2 (ja) | 2015-09-30 |
| JP2010540096A (ja) | 2010-12-24 |
| WO2009042596A3 (en) | 2009-05-28 |
| EP2194941B1 (en) | 2013-07-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5406840B2 (ja) | 眼内移植片 | |
| US8282592B2 (en) | Glaucoma treatment method | |
| US9610196B2 (en) | Ocular implants with asymmetric flexibility | |
| US11504275B2 (en) | Methods and apparatus for treating glaucoma | |
| US11744734B2 (en) | Method of implanting an ocular implant | |
| US9402767B2 (en) | Ocular implant architectures | |
| AU2016203606B2 (en) | Ocular implants and methods | |
| AU2014200768B2 (en) | Ocular implants and methods |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110916 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110916 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20130214 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130221 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130521 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130528 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130821 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20131002 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20131101 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 Ref document number: 5406840 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: R3D03 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: R3D02 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313113 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| S633 | Written request for registration of reclamation of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313633 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| S633 | Written request for registration of reclamation of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313633 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |