JP4401818B2 - Briquette manufacturing method - Google Patents
Briquette manufacturing method Download PDFInfo
- Publication number
- JP4401818B2 JP4401818B2 JP2004059203A JP2004059203A JP4401818B2 JP 4401818 B2 JP4401818 B2 JP 4401818B2 JP 2004059203 A JP2004059203 A JP 2004059203A JP 2004059203 A JP2004059203 A JP 2004059203A JP 4401818 B2 JP4401818 B2 JP 4401818B2
- Authority
- JP
- Japan
- Prior art keywords
- dust
- mass
- briquette
- plastic
- lime
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 239000004484 Briquette Substances 0.000 title claims description 36
- 238000004519 manufacturing process Methods 0.000 title claims description 14
- 239000000428 dust Substances 0.000 claims description 50
- 239000004033 plastic Substances 0.000 claims description 29
- 229920003023 plastic Polymers 0.000 claims description 29
- 238000000034 method Methods 0.000 claims description 24
- 238000002844 melting Methods 0.000 claims description 22
- 230000008018 melting Effects 0.000 claims description 22
- 235000008733 Citrus aurantifolia Nutrition 0.000 claims description 19
- 235000011941 Tilia x europaea Nutrition 0.000 claims description 19
- 239000004571 lime Substances 0.000 claims description 19
- 239000003575 carbonaceous material Substances 0.000 claims description 17
- ODINCKMPIJJUCX-UHFFFAOYSA-N Calcium oxide Chemical group [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 claims description 16
- 239000000292 calcium oxide Substances 0.000 claims description 8
- 235000012255 calcium oxide Nutrition 0.000 claims description 8
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 claims description 5
- 239000000920 calcium hydroxide Substances 0.000 claims description 5
- 235000011116 calcium hydroxide Nutrition 0.000 claims description 5
- 229910001861 calcium hydroxide Inorganic materials 0.000 claims description 5
- 238000007670 refining Methods 0.000 claims description 5
- 239000010802 sludge Substances 0.000 claims description 5
- 238000005554 pickling Methods 0.000 claims description 4
- 238000005096 rolling process Methods 0.000 claims description 4
- 238000006722 reduction reaction Methods 0.000 description 56
- 239000000843 powder Substances 0.000 description 24
- 238000000465 moulding Methods 0.000 description 18
- 229910052751 metal Inorganic materials 0.000 description 14
- 239000002184 metal Substances 0.000 description 14
- 238000002156 mixing Methods 0.000 description 11
- 239000002699 waste material Substances 0.000 description 11
- 239000002994 raw material Substances 0.000 description 10
- 239000011230 binding agent Substances 0.000 description 8
- 238000010438 heat treatment Methods 0.000 description 8
- 230000001603 reducing effect Effects 0.000 description 8
- 239000003638 chemical reducing agent Substances 0.000 description 6
- 238000003723 Smelting Methods 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 239000008188 pellet Substances 0.000 description 5
- 239000000571 coke Substances 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 238000013329 compounding Methods 0.000 description 4
- 238000011084 recovery Methods 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 238000002485 combustion reaction Methods 0.000 description 3
- 239000002440 industrial waste Substances 0.000 description 3
- 229910044991 metal oxide Inorganic materials 0.000 description 3
- 150000004706 metal oxides Chemical class 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 239000012752 auxiliary agent Substances 0.000 description 2
- 239000003610 charcoal Substances 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 229910001385 heavy metal Inorganic materials 0.000 description 2
- 239000000155 melt Substances 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000003245 coal Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 238000003912 environmental pollution Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000010304 firing Methods 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 238000010309 melting process Methods 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229920001225 polyester resin Polymers 0.000 description 1
- 239000004645 polyester resin Substances 0.000 description 1
- 238000004064 recycling Methods 0.000 description 1
- 238000011946 reduction process Methods 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 229920005792 styrene-acrylic resin Polymers 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 239000004416 thermosoftening plastic Substances 0.000 description 1
- 239000002351 wastewater Substances 0.000 description 1
Images
Description
本発明は、溶解炉および精錬炉より発生する集塵ダストの成型および還元処理に関し、成型性に優れ、かつ高還元性を持つブリケットの製造方法に関する。 The present invention relates to molding and reduction treatment of dust collection dust generated from a melting furnace and a refining furnace, and relates to a method for producing briquettes having excellent moldability and high reducing ability.
溶解炉および精錬炉より発生する集塵ダストは有価な金属酸化物を含有するが、微粉体であり、かつ重金属を含有することから、集塵ダストの処分や処理に多額の費用を要している。つまり、集塵ダストの多くは産業廃棄物処理業者への委託廃棄されている。この場合、通常は埋立て処分が行われるが、処分費用の負担、微粉ダストのハンドリング時の周辺環境への飛散、埋立て後の排水への重金属の混入による環境汚染の恐れがある。 Dust collection dust generated from melting furnaces and smelting furnaces contains valuable metal oxides, but they are fine powders and contain heavy metals. Yes. In other words, most of the dust collection dust is discarded by an industrial waste disposal contractor. In this case, landfill disposal is usually performed, but there is a risk of environmental pollution due to the burden of disposal costs, scattering of the fine dust to the surrounding environment during handling, and mixing of heavy metals into the wastewater after landfill.
近年、これらの問題を解決するために、集塵ダストをペレット状あるいはブリケット状に成型し、溶解炉にて溶解し、ダスト中の有価金属を回収する試みがなされるようになってきている。例えば、微粉体を成型する方法として、特許文献1、特許文献2が開示されている。
In recent years, in order to solve these problems, attempts have been made to collect dust-collected dust into pellets or briquettes, dissolve it in a melting furnace, and recover valuable metals in the dust. For example, Patent Literature 1 and
これらの方法は粉体にプラスチックを加え、ブリケット状に成型する製造方法であり、特許文献1は熱可塑性廃棄プラスチックを加え、混錬して金属粉体の摩擦熱でプラスチックを溶融させて圧縮、成型を行う方法であり、特許文献2は金属系粉体とプラスチック原料を混合および加熱して、プラスチック原料の一部を溶融させてバインダーとし、ブリケット化する方法である。
These methods are manufacturing methods in which plastic is added to powder and molded into a briquette shape. Patent Document 1 adds thermoplastic waste plastic, kneads and melts and compresses the plastic with frictional heat of metal powder,
これらの方法は粉体の成型には優れた方法であるが、粉体中の有価な金属酸化物の還元、回収には問題がある。つまり、プラスチックの一部は還元剤となり得るが、燃焼・飛散しやすく、加熱直後にはなくなり、バインダーとして役割が失うために、固体状態で還元を行う場合には成型体の強度が低下して、粉体状に戻ってしまう欠点がある。また、溶解炉で使用する場合でも、炉内に装入する以前は低温状態で保持しなければならず、また溶解処理で多量の還元剤が必要であり、処理コストが高くなる欠点を持つ。 Although these methods are excellent methods for molding powder, there are problems in reducing and recovering valuable metal oxides in the powder. In other words, a part of the plastic can be a reducing agent, but it is easy to burn and scatter, disappears immediately after heating, and loses its role as a binder. , There is a drawback of returning to powder. Further, even when used in a melting furnace, it must be kept at a low temperature before being charged into the furnace, and a large amount of reducing agent is required for the melting process, which has the disadvantage of increasing the processing cost.
次に、金属酸化物を含んだ粉体から金属を回収する方法としては、特許文献3、特許文献4が開示されている。特許文献3による方法は粉体ダストを廃トナー粉と混和した後、圧縮成型してペレット状に造粒し、造粒ペレットをキューポラ等の金属溶解炉に原料として供給し、金属を回収すると共に燃料としても利用する方法である。
Next, Patent Literature 3 and
本方法では前述した問題が解決されていない。つまり、粉体ダストと廃トナー粉のみを混合、成型したペレットを金属溶解炉に原料として供給するために、溶解炉に供給するまでは低温状態で保持する必要があり、溶解炉に供給した直後に、燃焼が起こり、廃トナー分がなくなるために、十分な還元作用が得られず、金属分を回収するために、別途供給する還元剤が多量に必要となり、処理コストが高くなる。 This method does not solve the problems described above. In other words, in order to supply pellets formed by mixing and molding powder dust and waste toner powder as raw materials to the metal melting furnace, it is necessary to keep them at a low temperature until they are supplied to the melting furnace. In addition, since combustion occurs and the amount of waste toner disappears, a sufficient reducing action cannot be obtained, and a large amount of a reducing agent supplied separately is required to recover the metal, resulting in an increase in processing cost.
特許文献4による方法は、ダストに炭素を含む還元剤を添加して成型したペレットまたはブリケットを1000℃から1100℃で5分以上焼成する方法であるが、この方法では粉末状のダストを成型するためのバインダーが添加されていないために、成型状態が不安定である。また、1000℃から1100℃での焼成では脈石部分が溶融して結合状態は強くなるが、還元反応を進めるには不十分であり、十分な還元状態が得られない。
The method according to
ここに述べた特許文献以外にも、種々の粉体ダストの製造方法、または粉体ダストの還元方法が開示されているが、いずれも成型性および還元性の双方を満足するものではなかった。 In addition to the patent documents mentioned here, various methods for producing powder dust or methods for reducing powder dust have been disclosed, but none of them satisfy both moldability and reducibility.
本発明の目的は、産業廃棄物とされる溶解炉および精錬炉より発生する集塵ダストの成型および還元処理に対し、以上述べたような従来の成型性および還元性の問題に対し、成型性に優れ、かつ高還元性を持つブリケットの製造方法を得ることにある。 The object of the present invention is to solve the conventional moldability and reducibility problems as described above, with respect to the molding and reduction treatment of dust collection dust generated from melting furnaces and refining furnaces that are industrial waste. It is to obtain a method for producing briquettes that is excellent in high reducibility.
本発明は、集塵ダストに、プラスチックを1〜10mass%、石灰成分を5〜30mass%、および炭材を5〜20mass%混合、成型することを特徴とするブリケットの製造方法である。また、集塵ダストが、溶解炉集塵ダストまたは精錬炉集塵ダストであることを特徴とし、プラスチックがトナーであることを特徴とし、石灰成分が生石灰または消石灰であることを特徴とし、集塵ダストに圧延工程で発生するスケールまたは酸洗工程で発生する中和スラッジの1種または2種を混合させることを特徴とするブリケットの製造方法である。さらに、上記ブリケットの製造方法に続いて、1200℃〜1350℃で15分以上、60分以下に保持することを特徴とするブリケットの製造方法である。 The present invention is a briquette manufacturing method characterized by mixing and molding 1 to 10 mass% of plastic, 5 to 30 mass% of a lime component, and 5 to 20 mass% of a carbonaceous material in dust collection dust. The dust collecting dust is melting furnace dust collecting dust or smelting furnace dust collecting dust, the plastic is toner, the lime component is quick lime or slaked lime, A briquette manufacturing method characterized in that dust is mixed with one or two kinds of scales generated in a rolling process or neutralized sludge generated in a pickling process. Further, following the above briquette production method, the briquette production method is characterized by holding at 1200 ° C. to 1350 ° C. for 15 minutes or more and 60 minutes or less.
本発明法により製造されたブリケットは、成型性に優れ、高温状態でも安定した成型状態が確保できる。溶解炉で溶融処理する前工程で還元処理を行う場合、ブリケット自体に還元剤、還元補助剤および高温用のバインダーを有しているので、安定して高還元率が得られ、還元後でも成型状態の維持されたブリケットを得ることが可能である。また、直接溶解炉で処理する場合も、装入するまでの過程で特別に管理する必要がなく、溶解炉で新たに還元剤を添加する必要がない。以上より、本発明法で製造されたブリケットは処理が容易であり、また、安価に還元処理が行える。 The briquettes manufactured by the method of the present invention are excellent in moldability and can secure a stable molded state even at high temperatures. When the reduction process is performed in the previous step of melting in the melting furnace, the briquette itself has a reducing agent, a reducing auxiliary agent, and a binder for high temperature, so a high reduction rate can be obtained stably, and molding is performed even after reduction. It is possible to obtain a briquette in which the state is maintained. In addition, when processing directly in the melting furnace, it is not necessary to perform special management in the process until charging, and it is not necessary to newly add a reducing agent in the melting furnace. As described above, the briquette manufactured by the method of the present invention is easy to process and can be reduced at a low cost.
本発明者らは粉体状の集塵ダストの成型および還元処理に関し、成型性に優れ、かつ高還元性を持つブリケットの製造方法を種々検討した結果、ブリケットの製造において、下記点を考慮する必要があることを究明した。 The inventors of the present invention have studied various methods for producing briquettes having excellent moldability and high reducibility with respect to the molding and reduction treatment of powdered dust collection dust. As a result, the following points are considered in the production of briquettes. Investigated that there is a need.
(1)低温および高温状態の両方でバインダー効果のある物質として、石灰分を添加することが有効である。(2)ブリケット自体の還元性を付与するために炭材の添加が必要である。(3)従来は低温用バインダーとして用いられたプラスチックはバインダーとして使用するよりも、高温の処理で炭材の燃焼を抑え、炭材中のCによる還元を促進する還元補助剤として有効である。 (1) It is effective to add lime as a substance having a binder effect at both low and high temperatures. (2) It is necessary to add a carbonaceous material in order to impart the reducibility of the briquette itself. (3) Conventionally, a plastic used as a binder for low temperature is more effective as a reducing auxiliary agent that suppresses combustion of the carbonaceous material by high-temperature treatment and promotes reduction by C in the carbonaceous material than when used as a binder.
これらの結果を受けて、次に適正な配合割合の検討を行った。図1に溶解炉および精錬炉の集塵ダストにプラスチック、炭材に石灰分を加えて、混合、成型した場合の石灰配合率とブリケット成型後5mm以上塊の歩留割合の関係を示す。なお、図中の○印は石灰分として生石灰、□印は消石灰を用いた場合を示す。また、この場合、プラスチックにはコピー機、画像処理装置等から回収された廃トナー粉を用いて5mass%添加し、炭材は微粉コークスを15mass%添加し、ブリケットは幅40mm×長さ60mm×厚み20mmの卵型に成型し、成型後、5mmの篩いを用いて、5mm以上塊の歩留を評価した。 Based on these results, the appropriate blending ratio was then examined. FIG. 1 shows the relationship between the lime mixing ratio and the yield ratio of a block of 5 mm or more after briquette molding when plastic and carbon material are added to the dust collected in the melting furnace and the refining furnace and mixed and molded. In the figure, ◯ indicates the case where quick lime is used as the lime, and □ indicates the case where slaked lime is used. Further, in this case, 5 mass% is added to the plastic using waste toner powder collected from a copying machine, an image processing apparatus, etc., carbon material is added 15 mass% of fine coke, and the briquette is 40 mm wide × 60 mm long × It was molded into an egg shape having a thickness of 20 mm, and after molding, the yield of masses of 5 mm or more was evaluated using a 5 mm sieve.
図1より、石灰分としての生石灰、消石灰の差はなく、5mass%以上の添加で歩留70%以上を確保できる。また、5mass%以上の添加により、徐々に歩留は向上する傾向にあるが、30mass%以上の添加は歩留の向上代がなく、30mass%以上の添加では集塵ダストの配合量の低下に繋がるために、石灰分の添加は5〜30mass%とする必要がある。 From FIG. 1, there is no difference between quick lime and slaked lime as lime components, and a yield of 70% or more can be secured by adding 5 mass% or more. The addition of 5 mass% or more tends to gradually improve the yield, but the addition of 30 mass% or more has no margin to improve the yield, and the addition of 30 mass% or more reduces the amount of dust collection dust. In order to connect, it is necessary to add 5 to 30 mass% of lime.
ここで得られた石灰分を5〜30mass%添加した5mm以上の塊ブリケットを回転炉床型の加熱炉で還元処理を行った。1300℃で30分保持した後、還元、冷却後ブリケットの5mm以上塊の歩留を評価した結果、いずれも70%以上の歩留となった。これより、石灰分が成型時の低温および還元処理時の高温においてもバインダーとしての役割を持つことを確認した。 The 5 mm or more lump briquettes to which 5-30 mass% of the lime content obtained here was added were subjected to reduction treatment in a rotary hearth type heating furnace. After maintaining at 1300 ° C. for 30 minutes, after reducing and cooling, the yield of the briquette of 5 mm or more was evaluated, and as a result, the yield was 70% or more. From this, it was confirmed that lime content has a role as a binder even at a low temperature during molding and a high temperature during reduction treatment.
図2に溶解炉および精錬炉の集塵ダストにプラスチック、石灰分に炭材を加えて、混合、成型し、成型後の5mm以上ブリケットを回転炉床型の加熱炉にて1300℃で30分還元処理した場合の炭材の配合率とブリケット内Fe分の還元率の関係を示す。なお、プラスチックにはコピー機、画像処理装置等から回収された廃トナー粉を用いて5mass%添加し、石灰分は生石灰を20mass%添加し、炭材にはコークス粉を用いた。 Figure 2 shows the dust collected in the melting furnace and smelting furnace. Add plastic and carbon material to the lime, mix and mold, and then form a briquette of 5 mm or more at 1300 ° C for 30 minutes in a rotary hearth-type heating furnace. The relationship between the mixing ratio of the carbonaceous material and the reduction ratio of Fe content in the briquette in the case of reduction treatment is shown. In addition, 5 mass% was added to the plastic using waste toner powder collected from a copying machine, an image processing apparatus, etc., 20 mass% of quicklime was added to the lime content, and coke powder was used as the carbon material.
図2より、石炭分の配合率が5mass%以上であれば、Fe還元率70%以上となり、高還元率が得られる。5mass%以上の添加では20mass%までは徐々にFe還元率が上昇する傾向にあるが、20mass%以上添加ではFe還元率の上昇は殆どなく、集塵ダストの配合量の低下、ブリケット製造コストの増加を招くことから、炭材の配合率は5〜20mass%とする必要がある。 As shown in FIG. 2, when the coal content is 5 mass% or more, the Fe reduction rate is 70% or more, and a high reduction rate is obtained. When the addition is 5 mass% or more, the Fe reduction rate tends to increase gradually up to 20 mass%. However, when the addition is 20 mass% or more, there is almost no increase in the Fe reduction rate. Since it causes an increase, the blending ratio of the carbon material needs to be 5 to 20 mass%.
図3に溶解炉および精錬炉の集塵ダストに石灰分、炭材にプラスチックを加えて、混合、成型し、成型後の5mm以上塊ブリケットを回転炉床型の加熱炉にて1300℃で30分還元処理した場合のプラスチックの配合率とブリケット内Fe分の還元率の関係を示す。なお、石灰分は生石灰を20mass%添加し、炭材は粉コークスを10mass%添加し、プラスチックにはコピー機、画像処理装置等から回収された廃トナー粉を用いた。 In FIG. 3, lime content is added to the dust collected in the melting furnace and smelting furnace, plastic is added to the carbonaceous material, and the mixture is mixed and molded. The molded briquette of 5 mm or more is formed at 1300 ° C. in a rotary hearth-type heating furnace. The relation between the blending ratio of the plastic and the reduction ratio of Fe in the briquette in the case of partial reduction treatment is shown. In addition, 20 mass% of quick lime was added to the lime content, 10 mass% of powder coke was added to the carbon material, and waste toner powder collected from a copying machine, an image processing apparatus, or the like was used as the plastic.
図3より、プラスチックの配合率が1mass%以上であれば、Fe還元率70%以上が安定的に達成される。プラスチックの配合率が10mass%までは徐々にFe還元率が上昇すると共に、Fe還元率のばらつきも小さくなる傾向を持つが、10mass%以上の添加での向上代は殆どない。また、10mass%以上の添加ではプラスチックによる燃焼が強くなり、低温でも発火し易く、取り扱いに注意を要する必要が生じることから、プラスチックの配合率は1〜10mass%とする必要がある。 From FIG. 3, if the compounding ratio of the plastic is 1 mass% or more, the Fe reduction ratio of 70% or more is stably achieved. Although the Fe reduction rate gradually increases and the variation of the Fe reduction rate tends to be reduced until the plastic content rate is 10 mass%, there is almost no improvement margin with addition of 10 mass% or more. Addition of 10% by mass or more increases the combustion by the plastic, easily ignites even at low temperatures, and requires careful handling. Therefore, the blending ratio of the plastic needs to be 1 to 10% by mass.
ここで、還元反応でのプラスチックの効果は、粉コークスのような炭材よりも燃焼しやすいことから、還元初期にプラスチック分の燃焼が起こるために、ブリケット周囲および内部の酸素濃度を下げ、ブリケット内部への酸素の侵入を抑え、炭材による還元反応進行を促進する還元補助剤の役割を果たすと考えられる。このような作用を持つプラスチックとしては、融点が100℃前後であり、かつ燃焼性の強いスチレンアクリル樹脂、ポリエステル樹脂を含むものが有効であり、その中で、コピー機、画像処理装置等に使用されるトナー(又はこれらから回収された廃トナー粉)は、これらの樹脂を50mass%以上含み、廃トナー粉は産業廃棄物とされることから、資源リサイクル面からも有効である。 Here, the effect of plastic in the reduction reaction is easier to burn than charcoal such as powdered coke, so the plastic content burns in the early stage of reduction, so the oxygen concentration around and inside the briquette is lowered, and the briquette It is thought to play the role of a reducing aid that suppresses the ingress of oxygen into the interior and promotes the progress of the reduction reaction by the carbonaceous material. As plastics having such an action, those having a melting point of around 100 ° C. and containing highly flammable styrene acrylic resin and polyester resin are effective. Among them, they are used for copying machines, image processing apparatuses, etc. The toner (or waste toner powder collected from them) contains 50 mass% or more of these resins, and the waste toner powder is industrial waste, which is also effective in terms of resource recycling.
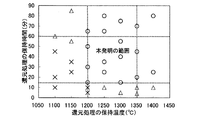
次に、ブリケットの還元処理条件について検討した。図4に溶解炉および精錬炉の集塵ダストにプラスチック、石灰分、炭材を加えて、混合、成型し、成型後の5mm以上ブリケットを小規模の大気加熱実験炉にて種々の温度、保持時間で還元処理した場合の温度と保持時間の関係におけるブリケット内Fe分の還元率の分布を示す。なお、図中の○印はFe還元率が70%以上、△印は50〜70%、×印は50%以下を示す。また、プラスチックは廃トナー粉を5mass%、石灰分は生石灰を20mass%添加し、炭材は粉コークスを10mass%添加した。 Next, briquette reduction treatment conditions were examined. Fig. 4 shows the dust collected in the melting furnace and smelting furnace, added with plastic, lime, and charcoal, mixed and molded, and then held the various briquettes of 5 mm or more at various temperatures in a small-scale atmospheric heating experimental furnace. The distribution of the reduction rate of Fe content in the briquette in the relationship between the temperature and the holding time when the reduction treatment is performed by time is shown. In the figure, ◯ indicates that the Fe reduction rate is 70% or more, Δ indicates 50 to 70%, and X indicates 50% or less. Further, 5 mass% of waste toner powder was added to plastic, 20 mass% of quicklime was added to the lime, and 10 mass% of powder coke was added to the carbon material.
図4より、1200℃以上で15分以上保持すれば、70%以上のFe還元率が得られる。実験では1350℃を超える温度ではブリケットの融体化が起こり、耐火物に付着して、分離できない現象が生じた。また、保持時間60分以上ではFe還元率の上昇は小さく、加熱処理コストの方が高くなることが確認された。以上より、還元処理条件としては、1200〜1350℃で15分以上、60分以下とするが望ましい。 As shown in FIG. 4, if the temperature is maintained at 1200 ° C. or higher for 15 minutes or longer, an Fe reduction rate of 70% or higher can be obtained. In the experiment, the briquette melted at a temperature exceeding 1350 ° C. and adhered to the refractory, resulting in a phenomenon that could not be separated. Further, it was confirmed that when the holding time was 60 minutes or more, the increase in Fe reduction rate was small and the heat treatment cost was higher. From the above, it is preferable that the reduction treatment condition is 1200 to 1350 ° C. and 15 minutes or more and 60 minutes or less.
本発明者らは、集塵ダストと同様に、有価な金属成分を含み、粉末状に存在する圧延工程で発生するスケール、酸洗工程で発生する中和スラッジの処理の可能性を調査した。その結果、集塵ダストにスケールまたは中和スラッジの1種または2種を混合しても、前記配合条件であれば、成型性および還元性とも劣化することはないことを確認した。 The present inventors investigated the possibility of treatment of the neutral sludge generated in the pickling process and the scale generated in the rolling process which contains valuable metal components and exists in a powder form, as in the dust collection dust. As a result, it was confirmed that even if one kind or two kinds of scale or neutralized sludge were mixed with the dust collection dust, the moldability and the reducibility were not deteriorated under the above-mentioned blending conditions.
以下、本発明例および比較例に基づいて、本発明を更に詳細に説明する。表1に本発明例と比較例の原料配合組成、ブリケット成型後の5mm以上塊の歩留、還元処理を行った時の加熱温度、保持時間、還元処理後のFe還元率、還元処理後の5mm以上塊の歩留、および金属回収コスト指数を示す。なお、原料としては、溶解炉および精錬炉の集塵ダスト、圧延工程で発生するスケール、酸洗工程で発生する中和スラッジ、プラスチックとしては廃トナー、石灰成分としては生石灰および消石灰、炭材としてはコークス粉を使用し、還元処理ではブリケット成型後5mm以上の塊のみを使用した。また、金属回収コスト指数は、原料コスト、還元処理コストおよび還元処理後のブリケットを溶解炉に供給して金属を回収するためのコストを基に、本発明例のNo.1を100として比例換算して求めた。 Hereinafter, the present invention will be described in more detail based on examples of the present invention and comparative examples. Table 1 shows the raw material composition of the present invention and comparative examples, the yield of lumps of 5 mm or more after briquette molding, the heating temperature when holding the reduction treatment, the holding time, the Fe reduction rate after the reduction treatment, the post-reduction treatment The yield of a mass of 5 mm or more and the metal recovery cost index are shown. In addition, as raw materials, dust collection dust of melting furnace and refining furnace, scale generated in rolling process, neutralized sludge generated in pickling process, waste toner as plastic, quick lime and slaked lime as lime component, as carbon material Used coke powder, and in the reduction treatment, only a lump of 5 mm or more was used after briquette molding. Further, the metal recovery cost index is based on the raw material cost, the reduction processing cost, and the cost for recovering the metal by supplying the briquette after the reduction processing to the melting furnace. It was determined by proportionally converting 1 as 100.
表1より、本発明例のNo.1〜10では原料配合および還元処理条件とも、本発明の条件範囲を満足しており、その結果、ブリケット成型後の5mm以上塊の歩留、還元処理後の5mm以上塊の歩留とも70%以上の高歩留を達成している。また、還元処理後のFe還元率も70%以上で高位安定していることから、金属回収コストも低位安定している。 From Table 1, No. of the present invention example. 1 to 10 satisfy the condition range of the present invention for both the raw material composition and the reduction treatment condition. As a result, the yield of the block of 5 mm or more after briquette molding and the yield of the block of 5 mm or more after the reduction treatment are 70%. The above high yield has been achieved. In addition, since the Fe reduction rate after the reduction treatment is highly stable at 70% or more, the metal recovery cost is also stable at a low level.
一方、比較例のNo.11〜16では原料配合或いは還元処理条件が本発明の条件外であり、ブリケット成型後の5mm以上塊の歩留、還元処理後の5mm以上塊の歩留、或いは還元処理後のFe還元率が低く、その結果、還元処理コストが高くなっている。また、比較例のNo.17は、還元処理温度が本発明の条件外にあり、還元処理時にブリケットが耐火物に付着し、回収不能となった。さらに、比較例のNo.18〜20では、ブリケット成型後の5mm以上塊の歩留、還元処理後の5mm以上塊の歩留、および還元処理後のFe還元率は本発明例と同等であるが、原料コスト或いは還元処理コストが高いために、金属回収コストが本発明例に比べ、高位となっている。 On the other hand, no. In 11 to 16, the raw material composition or the reduction treatment condition is outside the conditions of the present invention, and the yield of the mass of 5 mm or more after briquette molding, the yield of the mass of 5 mm or more after the reduction treatment, or the Fe reduction rate after the reduction treatment is As a result, the reduction treatment cost is high. Moreover, No. of the comparative example. In No. 17, the reduction treatment temperature was outside the conditions of the present invention, and the briquette adhered to the refractory during the reduction treatment, making it impossible to recover. Furthermore, No. of the comparative example. 18-20, the yield of the lump of 5 mm or more after briquette molding, the yield of the lump of 5 mm or more after the reduction treatment, and the Fe reduction rate after the reduction treatment are the same as those of the present invention example, but the raw material cost or the reduction treatment Since the cost is high, the metal recovery cost is higher than that of the example of the present invention.
Claims (5)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004059203A JP4401818B2 (en) | 2004-03-03 | 2004-03-03 | Briquette manufacturing method |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004059203A JP4401818B2 (en) | 2004-03-03 | 2004-03-03 | Briquette manufacturing method |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2005248235A JP2005248235A (en) | 2005-09-15 |
| JP4401818B2 true JP4401818B2 (en) | 2010-01-20 |
Family
ID=35028998
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2004059203A Expired - Lifetime JP4401818B2 (en) | 2004-03-03 | 2004-03-03 | Briquette manufacturing method |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP4401818B2 (en) |
-
2004
- 2004-03-03 JP JP2004059203A patent/JP4401818B2/en not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| JP2005248235A (en) | 2005-09-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1290232B1 (en) | Method of producing a metallized briquette | |
| JP4348152B2 (en) | Method for producing ferronickel and ferronickel refining raw material | |
| US5807420A (en) | Process for reduction of iron with solid fuel objects as amended by exam | |
| US20110024681A1 (en) | Titanium oxide-containing agglomerate for producing granular metallic iron | |
| JP2010111941A (en) | Method for producing ferrovanadium | |
| JP2010229525A (en) | Method for producing ferronickel and ferrovanadium | |
| US6342089B1 (en) | Direct reduced iron pellets | |
| US20070266824A1 (en) | Using a slag conditioner to beneficiate bag house dust from a steel making furnace | |
| US9011573B2 (en) | Process for recycling of steel industry iron bearing by-products, pellet obtained in that process and use thereof | |
| KR101234388B1 (en) | Process for production of direct-reduced iron | |
| JP3502064B2 (en) | Method for producing agglomerates of ironmaking raw materials | |
| JP2011246760A (en) | Method of manufacturing ferromolybdenum, and ferromolybdenum | |
| JP2010090431A (en) | Method for producing ferro-alloy containing nickel and vanadium | |
| JP2006152432A (en) | Method for producing molten iron | |
| JP3035285B1 (en) | Method for producing carburized material for steel making containing electric furnace dust, carburized material for steel making obtained thereby and method for recycling electric furnace dust | |
| JP4401818B2 (en) | Briquette manufacturing method | |
| JPH09316512A (en) | Method for melting steel using iron oxide briquette as auxiliary raw material | |
| KR20080112818A (en) | Method for recovering high value metals from waste materials of steel making process | |
| JP2017128786A (en) | Carbon material interior ore and manufacturing method therefor | |
| US20170130284A1 (en) | Products and processes for producing steel alloys using an electric arc furnace | |
| KR102469639B1 (en) | Method for removing impurities in dust | |
| JP2011179090A (en) | Method for producing granulated iron | |
| JP5397020B2 (en) | Reduced iron production method | |
| JP4192042B2 (en) | Shredder dust processing method in coke oven | |
| Pal et al. | Development of carbon composite iron ore slime briquettes for using in ironmaking |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20061215 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20090519 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20090526 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090629 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20091027 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20091028 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4401818 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20121106 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20131106 Year of fee payment: 4 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R371 | Transfer withdrawn |
Free format text: JAPANESE INTERMEDIATE CODE: R371 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |