JP4197159B2 - 混成の生物学的合成の生体吸収性支持骨格材料 - Google Patents
混成の生物学的合成の生体吸収性支持骨格材料 Download PDFInfo
- Publication number
- JP4197159B2 JP4197159B2 JP2003513456A JP2003513456A JP4197159B2 JP 4197159 B2 JP4197159 B2 JP 4197159B2 JP 2003513456 A JP2003513456 A JP 2003513456A JP 2003513456 A JP2003513456 A JP 2003513456A JP 4197159 B2 JP4197159 B2 JP 4197159B2
- Authority
- JP
- Japan
- Prior art keywords
- sis
- layer
- ecm
- tissue
- bioprosthesis
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/36—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix
- A61L27/38—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix containing added animal cells
- A61L27/3839—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix containing added animal cells characterised by the site of application in the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/064—Surgical staples, i.e. penetrating the tissue
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/064—Surgical staples, i.e. penetrating the tissue
- A61B17/0642—Surgical staples, i.e. penetrating the tissue for bones, e.g. for osteosynthesis or connecting tendon to bone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/0063—Implantable repair or support meshes, e.g. hernia meshes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/08—Muscles; Tendons; Ligaments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30721—Accessories
- A61F2/30749—Fixation appliances for connecting prostheses to the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/18—Macromolecular materials obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/36—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix
- A61L27/3604—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix characterised by the human or animal origin of the biological material, e.g. hair, fascia, fish scales, silk, shellac, pericardium, pleura, renal tissue, amniotic membrane, parenchymal tissue, fetal tissue, muscle tissue, fat tissue, enamel
- A61L27/3629—Intestinal tissue, e.g. small intestinal submucosa
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/36—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix
- A61L27/3604—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix characterised by the human or animal origin of the biological material, e.g. hair, fascia, fish scales, silk, shellac, pericardium, pleura, renal tissue, amniotic membrane, parenchymal tissue, fetal tissue, muscle tissue, fat tissue, enamel
- A61L27/3633—Extracellular matrix [ECM]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/36—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix
- A61L27/3683—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix subjected to a specific treatment prior to implantation, e.g. decellularising, demineralising, grinding, cellular disruption/non-collagenous protein removal, anti-calcification, crosslinking, supercritical fluid extraction, enzyme treatment
- A61L27/3691—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix subjected to a specific treatment prior to implantation, e.g. decellularising, demineralising, grinding, cellular disruption/non-collagenous protein removal, anti-calcification, crosslinking, supercritical fluid extraction, enzyme treatment characterised by physical conditions of the treatment, e.g. applying a compressive force to the composition, pressure cycles, ultrasonic/sonication or microwave treatment, lyophilisation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/56—Porous materials, e.g. foams or sponges
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/005—Ingredients of undetermined constitution or reaction products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/064—Surgical staples, i.e. penetrating the tissue
- A61B2017/0646—Surgical staples, i.e. penetrating the tissue for insertion into cartillege, e.g. meniscus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/08—Muscles; Tendons; Ligaments
- A61F2/0811—Fixation devices for tendons or ligaments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30756—Cartilage endoprostheses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/3094—Designing or manufacturing processes
- A61F2/30965—Reinforcing the prosthesis by embedding particles or fibres during moulding or dipping
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/38—Joints for elbows or knees
- A61F2/3872—Meniscus for implantation between the natural bone surfaces
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/44—Joints for the spine, e.g. vertebrae, spinal discs
- A61F2/442—Intervertebral or spinal discs, e.g. resilient
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/28—Bones
- A61F2002/2817—Bone stimulation by chemical reactions or by osteogenic or biological products for enhancing ossification, e.g. by bone morphogenetic or morphogenic proteins [BMP] or by transforming growth factors [TGF]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30003—Material related properties of the prosthesis or of a coating on the prosthesis
- A61F2002/30004—Material related properties of the prosthesis or of a coating on the prosthesis the prosthesis being made from materials having different values of a given property at different locations within the same prosthesis
- A61F2002/30032—Material related properties of the prosthesis or of a coating on the prosthesis the prosthesis being made from materials having different values of a given property at different locations within the same prosthesis differing in absorbability or resorbability, i.e. in absorption or resorption time
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30003—Material related properties of the prosthesis or of a coating on the prosthesis
- A61F2002/3006—Properties of materials and coating materials
- A61F2002/30062—(bio)absorbable, biodegradable, bioerodable, (bio)resorbable, resorptive
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30108—Shapes
- A61F2002/3011—Cross-sections or two-dimensional shapes
- A61F2002/30138—Convex polygonal shapes
- A61F2002/30153—Convex polygonal shapes rectangular
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30108—Shapes
- A61F2002/30199—Three-dimensional shapes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30108—Shapes
- A61F2002/30199—Three-dimensional shapes
- A61F2002/30224—Three-dimensional shapes cylindrical
- A61F2002/30225—Flat cylinders, i.e. discs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30108—Shapes
- A61F2002/30199—Three-dimensional shapes
- A61F2002/30261—Three-dimensional shapes parallelepipedal
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30108—Shapes
- A61F2002/30199—Three-dimensional shapes
- A61F2002/30291—Three-dimensional shapes spirally-coiled, i.e. having a 2D spiral cross-section
- A61F2002/30294—Conical or frustoconical body made by spirally rolling up a sheet or a strip around itself
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30667—Features concerning an interaction with the environment or a particular use of the prosthesis
- A61F2002/30677—Means for introducing or releasing pharmaceutical products, e.g. antibiotics, into the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30721—Accessories
- A61F2/30749—Fixation appliances for connecting prostheses to the body
- A61F2002/30751—Fixation appliances for connecting prostheses to the body for attaching cartilage scaffolds to underlying bone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30756—Cartilage endoprostheses
- A61F2002/30766—Scaffolds for cartilage ingrowth and regeneration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30767—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth
- A61F2/30771—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth applied in original prostheses, e.g. holes or grooves
- A61F2002/30772—Apertures or holes, e.g. of circular cross section
- A61F2002/30784—Plurality of holes
- A61F2002/30785—Plurality of holes parallel
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30767—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth
- A61F2/30907—Nets or sleeves applied to surface of prostheses or in cement
- A61F2002/30909—Nets
- A61F2002/30914—Details of the mesh structure, e.g. disposition of the woven warp and weft wires
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/3094—Designing or manufacturing processes
- A61F2/30942—Designing or manufacturing processes for designing or making customized prostheses, e.g. using templates, CT or NMR scans, finite-element analysis or CAD-CAM techniques
- A61F2002/30957—Designing or manufacturing processes for designing or making customized prostheses, e.g. using templates, CT or NMR scans, finite-element analysis or CAD-CAM techniques using a positive or a negative model, e.g. moulds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/3094—Designing or manufacturing processes
- A61F2002/30971—Laminates, i.e. layered products
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2210/00—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2210/0004—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof bioabsorbable
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0002—Two-dimensional shapes, e.g. cross-sections
- A61F2230/0017—Angular shapes
- A61F2230/0019—Angular shapes rectangular
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0063—Three-dimensional shapes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0063—Three-dimensional shapes
- A61F2230/0069—Three-dimensional shapes cylindrical
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0063—Three-dimensional shapes
- A61F2230/0082—Three-dimensional shapes parallelepipedal
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0063—Three-dimensional shapes
- A61F2230/0091—Three-dimensional shapes helically-coiled or spirally-coiled, i.e. having a 2-D spiral cross-section
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2240/00—Manufacturing or designing of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2240/001—Designing or manufacturing processes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2240/00—Manufacturing or designing of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2240/001—Designing or manufacturing processes
- A61F2240/002—Designing or making customized prostheses
- A61F2240/004—Using a positive or negative model, e.g. moulds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0014—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof having different values of a given property or geometrical feature, e.g. mechanical property or material property, at different locations within the same prosthesis
- A61F2250/003—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof having different values of a given property or geometrical feature, e.g. mechanical property or material property, at different locations within the same prosthesis differing in adsorbability or resorbability, i.e. in adsorption or resorption time
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0014—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof having different values of a given property or geometrical feature, e.g. mechanical property or material property, at different locations within the same prosthesis
- A61F2250/003—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof having different values of a given property or geometrical feature, e.g. mechanical property or material property, at different locations within the same prosthesis differing in adsorbability or resorbability, i.e. in adsorption or resorption time
- A61F2250/0031—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof having different values of a given property or geometrical feature, e.g. mechanical property or material property, at different locations within the same prosthesis differing in adsorbability or resorbability, i.e. in adsorption or resorption time made from both resorbable and non-resorbable prosthetic parts, e.g. adjacent parts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0058—Additional features; Implant or prostheses properties not otherwise provided for
- A61F2250/0067—Means for introducing or releasing pharmaceutical products into the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2310/00—Prostheses classified in A61F2/28 or A61F2/30 - A61F2/44 being constructed from or coated with a particular material
- A61F2310/00005—The prosthesis being constructed from a particular material
- A61F2310/00365—Proteins; Polypeptides; Degradation products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/06—Materials or treatment for tissue regeneration for cartilage reconstruction, e.g. meniscus
Description
同時係属の「メニスカス・リジェネレーション・デバイス・アンド・メソッド(Meniscus Regeneration Device and Method)」を発明の名称とする米国特許出願第10/195,794号(代理人明細書番号第265280−71141号,DEP−745)、「デバイシーズ・フロム・ナチュラリー・オカーリング・バイオロジカリー・デライブド・マテリアルズ(Devices from Naturally Occurring Biologically Derived Materials)」を発明の名称とする米国特許出願第10/195,719号(代理人明細書番号第265280−71142号,DEP−748)、「カーテイレイジ・リペア・アパレイタス・アンド・メソッド(Cartilage Repair Apparatus and Method)」を発明の名称とする米国特許出願第10/195,347号(代理人明細書番号第265280−71143号,DEP−749)、「ユニタリー・サージカル・デバイス・アンド・メソッド(Unitary Surgical Device and Method)」を発明の名称とする米国特許出願第10/195,344号(代理人明細書番号DEP−750)、「ハイブリッド・バイオロジック/シンセチック・ポーラス・エクストラセルラー・マトリクス・スキャフォルド(Hybrid Biologic/Synthetic Porous Extracellular Matrix Scaffolds)」を発明の名称とする米国特許出願第10/195,341号(代理人明細書番号第265280−71144号,DEP−751)、「カーテイレイジ・リペア・アンド・リジェネレーション・デバイス・アンド・メソッド(Cartilage Repair and Regeneration Device and Method)」を発明の名称とする米国特許出願第10/195,606号(代理人明細書番号第265280−71145号,DEP−752)、「ポーラス・エクストラセルラー・マトリクス・スキャフォルド・アンド・メソッド(Porous Extracellular Matrix Scaffold and Method)」を発明の名称とする米国特許出願第10/195,354号(代理人明細書番号第265280−71146号,DEP−747)、「カーテイレイジ・リペア・アンド・リジェネレーション・スキャフォルド・アンド・メソッド(Cartilage Repair and Regeneration Scaffolds and Method)」を発明の名称とする米国特許出願第10/195,334号(代理人明細書番号第265280−71180号,DEP−763)、および「ポーラス・デリバリー・スキャフォルド・アンド・メソッド(Porous Delivery Scaffold and Method)」を発明の名称とする米国特許出願第10/195,633号(代理人明細書番号第265280−71207号,DEP−762)に対してクロス・リファレンスが行なわれており、これらのそれぞれは本特許出願と同一の譲受人に譲渡されており、これらのそれぞれは本特許出願と同時に出願されていて、これらのそれぞれは本明細書に参考文献として含まれる。
本発明は生体プロテーゼに関連しており、特に、結合組織の修復および置換のための生体プロテーゼの使用方法に関連している。特に、本発明は合成部分および異種動物組織により作成されている複合生体プロテーゼ装置の使用方法に関連している。
現在において、小腸粘膜下組織(SIS)の種々の特徴および特性を詳細に記載している多数の特許および公告が存在している。例えば、本明細書に参考文献として含まれている米国特許第5,352,463号、同第4,902,508号、同第4,956,179号、同第5,281,422号、同第5,372,821号、同第5,445,833号、同第5,516,533号、同第5,573,784号、同第5,641,518号、同第5,645,860号、同第5,668,288号、同第5,695,998号、同第5,711,969号、同第5,730,933号、同第5,733,868号、同第5,753,267号、同第5,755,791号、同第5,762,966号、同第5,788,625号、同第5,866,414号、同第5,885,619号、同第5,922,028号、同第6,056,777号、およびPCT国際公開第WO 97/37613号を参照されたい。SISは、種々の形態で、クック・バイオテック・インコーポレイテッド社(Cook Biotech Incorporated)(ブルーミントン、インディアナ州)から市場において入手可能である。さらに、カーランド(Kurland)に発行されている米国特許第4,400,833号および国際公開番号第WO 00/16822号を有するPCT特許公開は種々の生体プロテーゼに関連している情報を提供しており、これらもまた本明細書に参考文献として含まれる。
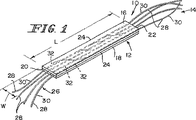
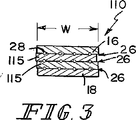
図1において示されている複合型生体プロテーゼ装置10が軟質組織の結合、補強、および/または再構成の目的のために提供されている。この生体プロテーゼ装置10は小腸粘膜下組織(SIS)部分12および合成部分14を備えている。SIS部分12は体内に吸収されて宿主組織により置換されるように備えられている。このSIS部分12は組織の内部成長および再造形のための支持骨格として作用する。また、上記生体プロテーゼ装置10の合成部分14は付加的な初期の機械的強度をこの生体プロテーゼ装置10に与えている。この装置10はSIS部分12および合成部分14を備えているので、この生体プロテーゼ装置10はその生体崩壊速度および生体再造形速度においてある差を備えている。例えば、合成部分14はSIS部分12よりも遅い速度で崩壊するように構成できる。さらに、合成部分14は生体プロテーゼ装置10を手術中に周囲の軟質組織(図示せず)に連結するための固定手段として作用できる。あるいは、SIS部分を縫合して上記生体プロテーゼ装置を周囲の組織に連結することも可能である。
清浄な消毒したブタのSIS材料の複数のシートを米国特許第4,902,508号および同第4,956,178号において記載されているように得た。3.5インチ(89mm)の幅および6インチ(152mm)の長さの10個の部材片を切断した。これらの部材片を室温で5分間にわたりRO水中に入れることにより水和した。
この実施例は生体崩壊性のSIS積層化シート、生体崩壊性のメッシュ材料の形態の合成補強部材、および合成の崩壊性の発泡体を含む三次元的な複合型組織移植片の調製方法を説明している。
この実施例は本発明の生体崩壊性の複合型支持骨格材料を製造するために上記実施例2において概説されている方法を用いており、この場合に、上記発泡体部品は65:35のPGA/PCLコポリマーである。
この実施例は本発明の生体崩壊性の複合型支持骨格材料を製造するために上記実施例2において概説されている方法を用いており、この場合に、上記合成の編み状メッシュ材料は100%PDOにより構成されている。
この実施例は本発明の生体崩壊性の複合型支持骨格材料を製造するために上記実施例2において概説されている方法を用いており、この場合に、三次元的なメッシュ材料の代わりに、上記の合成部品は100%PDO、100%90/10PGA/PLA、またはこれらの2種類の材料の組み合わせ物のいずれかにより構成されている不織状の繊維質構造である。
この実施例は本発明の生体崩壊性の複合型支持骨格材料を製造するために上記実施例2において概説されている方法を用いており、この場合に、上記SIS部品は上記合成メッシュ材料の上に配置する前にポリマー溶液(ジオキサン中における5%重量の60/40PLA/PCL)中において一晩にわたり浸漬されている。比較的に優れた取扱性を有する複合体により立証されているように上記の付加的な浸漬工程を上記方法に加えた場合に各構成部品の間において改善された積層状態が見られた。
この実施例は本発明の生体崩壊性の複合型支持骨格材料を製造するために上記実施例2において概説されている方法を用いており、この場合に、上記SIS部品は積層化したシート材料ではなく単一層のシート材料である。
この実施例は本発明の生体崩壊性の複合型支持骨格材料を製造するために上記実施例2において概説されている方法を用いており、この場合に、上記SISの積層化したシート材料が1mm乃至1cmの範囲の複数の孔を伴って孔あけ処理されている。これらの孔は上記SISシート材料の中を通るポリマー溶液の促進された浸透を可能にする。
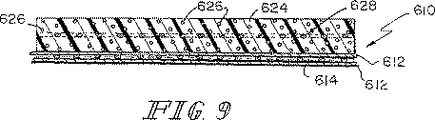
この実施例は本発明の生体崩壊性の複合型支持骨格材料を製造するために上記実施例2において概説されている方法を用いており、この場合に、上記SISの補強部品は60/40PLA/PCL発泡体の2個の層の間に挟み込まれている「織り状のメッシュ材料(woven mesh)」の積層化された部材片である。図13はこのような織り状のメッシュ材料を示している図である。このSIS層が図13における織り状のメッシュ材料である場合の図11がこの実施例の構成を示している。
なお、本発明の好ましい実施態様は以下の通りである。
(1) 生体プロテーゼ装置において、
第1の表面を有しているECM材料の層、および
第1の表面を有している三次元的な合成部分を備えており、
前記ECM層の第1の表面が前記三次元的な合成部分の第1の表面に連結している生体プロテーゼ装置。
(2) 前記ECM層がSIS層を含む実施態様1に記載の生体プロテーゼ装置。
(3) 前記SIS層が一体に積層化されている幾つかのSIS部材片を含む実施態様2に記載の生体プロテーゼ装置。
(4) 前記三次元的な部分が繊維質材料を含む実施態様1に記載の生体プロテーゼ装置。
(5) 前記繊維質材料がメッシュ材料、織物、およびフェルト材料から成る群から選択される実施態様4に記載の生体プロテーゼ装置。
(6) 前記繊維質材料がPLA、PGA、PCL、PDO、TMC、PVA、これらのコポリマー、およびこれらの混合物から成る群から選択される生体吸収性の材料である実施態様4に記載の生体プロテーゼ装置。
(7) 前記三次元的な部分が合成発泡体を含む実施態様1に記載の生体プロテーゼ装置。
(8) 前記ECM層が孔あけ処理されており、前記合成発泡体が前記ECM層の中の各孔の中に侵入している実施態様7に記載の生体プロテーゼ装置。
(9) さらに、生体活性物質、生物学的に誘導されている物質、細胞、生物学的な潤滑剤、生体相容性の無機材料、および生体相容性の合成ポリマーから成る群から選択される付加的な外因性の物質を含有している実施態様1に記載の生体プロテーゼ装置。
(10) 前記ECM材料が胃、膀胱、消化管、呼吸器、および生殖器の粘膜下組織および肝臓基底膜から選択される材料の層を含む実施態様1に記載の生体プロテーゼ装置。
(11) 1種類以上の三次元的な合成の本体部分に連結して組織の結合、補強、または再構成のための三次元的な複合材料を形成するための生体再造形可能な膠原性の組織材料の1個以上の層を備えているプロテーゼ装置。
(12) 生体プロテーゼ装置を作成するための方法において、
第1の表面を有するECM材料の層を供給する工程、
ポリマー溶液を前記ECM材料の第1の表面に接触させて配置することにより組立体を作成する工程を含み、前記ポリマーが凍結乾燥時に発泡体を形成するように選択され、さらに
前記組立体を凍結乾燥する工程を含む方法。
(13) 前記ECM材料が一体に積層化されている複数のSIS層を含む実施態様12に記載の方法。
(14) 前記ポリマー溶液がPCL、PLA、PGA、PDO、TMC、PVA、これらのコポリマー、およびこれらの混合物から成る群から選択される実施態様12に記載の方法。
12 小腸粘膜下組織(SIS)部分
14 合成部分
16 上部組織層
18 下部組織層
20 第1の端部
22 第2の端部
24 側面部
26 繊維の列
28 繊維
30 外側端部部分
32 中間部分
L 長さ
W 幅
Claims (15)
- 生体プロテーゼ装置において、
小腸、胃、膀胱、消化管、呼吸器、および生殖器の粘膜下組織ならびに肝臓基底膜から選択されたECM材料の層であって、第1の表面を有しており、組織の内部成長および再造形のための支持骨格として作用するECM材料の層と、
第1の表面を有しており、三つの相異なる軸に沿って繊維が配された三次元的な繊維質材料を含む、三次元的に機械的強度を賦与するための合成部分と、
を備えており、
前記ECM層の第1の表面が前記合成部分の第1の表面に連結されている、生体プロテーゼ装置。 - 請求項1に記載の生体プロテーゼ装置において、
前記ECM層がSIS層を含む、生体プロテーゼ装置。 - 請求項2に記載の生体プロテーゼ装置において、
前記SIS層が一体に積層化されている複数のSIS部材片を含む、生体プロテーゼ装置。 - 請求項3に記載の生体プロテーゼ装置において、
前記積層化したSIS部材片が孔あけ処理されている、生体プロテーゼ装置。 - 請求項2〜4のいずれかに記載の生体プロテーゼ装置において、
前記SIS層がSIS部材片の織り状のメッシュ材料を含む、生体プロテーゼ装置。 - 請求項1〜5のいずれかに記載の生体プロテーゼ装置において、
前記繊維質材料がPLA、PGA、PCL、PDO、TMC、PVA、これらのコポリマー、およびこれらの混合物から成る群から選択される生体吸収性の材料である、生体プロテーゼ装置。 - 請求項1〜6のいずれかに記載の生体プロテーゼ装置において、
前記合成部分が合成発泡体を含み、当該合成発泡体が、20〜400μmのサイズの隙間を有する、生体プロテーゼ装置。 - 請求項7に記載の生体プロテーゼ装置において、
前記隙間が、前記合成発泡体の体積の70〜95%を占める、生体プロテーゼ装置。 - 請求項7または8に記載の生体プロテーゼ装置において、
前記ECM層が孔あけ処理されており、前記合成発泡体が前記ECM層の中の孔の中に侵入している、生体プロテーゼ装置。 - 請求項1〜9のいずれかに記載の生体プロテーゼ装置において、
第2の表面を有しており、合成発泡体を含む第2の三次元的な合成部分をさらに備え、
前記ECM層が、前記第1の表面の反対側に第2の表面を有し、当該第2の合成部分の第2の表面が当該ECM層の第2の表面に連結されている、生体プロテーゼ装置。 - 請求項1〜10のいずれかに記載の生体プロテーゼ装置において、
生体活性物質、生物学的に誘導されている物質、細胞、生物学的な潤滑剤、生体相容性の無機材料、および生体相容性の合成ポリマーから成る群から選択される付加的な外因性の物質をさらに含有している、生体プロテーゼ装置。 - 生体プロテーゼ装置において、
第1の表面を有しており、小腸、胃、膀胱、消化管、呼吸器、および生殖器の粘膜下組織ならびに肝臓基底膜から選択された、組織の内部成長および再造形のための支持骨格として作用するECM材料の非微粒子状のシートと、
第1の表面を有しており、合成発泡体を含む、三次元的に機械的強度を賦与するための三次元的な合成部分と
を備えており、
当該ECMシートの第1の表面が、当該三次元的な合成部分の第1の表面に連結されている、生体プロテーゼ装置。 - 生体プロテーゼ装置を作成するための方法において、
第1の表面を有するECM材料の層を供給する工程と、
ポリマー溶液および前記ECM層を、金型内に、当該ポリマー溶液を前記ECM材料の第1の表面に接触させて配置することにより組立体を作成する工程であって、前記ポリマーが凍結乾燥時に発泡体を形成するように選択されている、工程と、
前記組立体を凍結乾燥する工程と
を含む方法。 - 請求項13に記載の方法において、
前記ECM材料が一体に積層化されている複数のSIS層を含む、方法。 - 請求項13または14に記載の方法において、
前記ポリマー溶液がPCL、PLA、PGA、PDO、TMC、PVA、これらのコポリマー、およびこれらの混合物から成る群から選択される、方法。
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US30578601P | 2001-07-16 | 2001-07-16 | |
| US09/918,116 US6638312B2 (en) | 2000-08-04 | 2001-07-30 | Reinforced small intestinal submucosa (SIS) |
| US10/172,347 US8366787B2 (en) | 2000-08-04 | 2002-06-14 | Hybrid biologic-synthetic bioabsorbable scaffolds |
| PCT/US2002/022364 WO2003007847A1 (en) | 2001-07-16 | 2002-07-15 | Hybrid biologic-synthetic bioabsorable scaffolds |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2004522555A JP2004522555A (ja) | 2004-07-29 |
| JP2004522555A5 JP2004522555A5 (ja) | 2008-09-04 |
| JP4197159B2 true JP4197159B2 (ja) | 2008-12-17 |
Family
ID=27390127
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003513456A Expired - Fee Related JP4197159B2 (ja) | 2001-07-16 | 2002-07-15 | 混成の生物学的合成の生体吸収性支持骨格材料 |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP1416878A4 (ja) |
| JP (1) | JP4197159B2 (ja) |
| AU (1) | AU2002320517B2 (ja) |
| WO (1) | WO2003007847A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011072797A (ja) * | 2009-10-01 | 2011-04-14 | Tyco Healthcare Group Lp | メッシュインプラント |
Families Citing this family (65)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2280812A1 (en) | 1997-02-13 | 1998-08-20 | Rodney Brenneman | Percutaneous and hiatal devices and methods for use in minimally invasive pelvic surgery |
| CA2365376C (en) | 2000-12-21 | 2006-03-28 | Ethicon, Inc. | Use of reinforced foam implants with enhanced integrity for soft tissue repair and regeneration |
| US9149261B2 (en) | 2001-03-09 | 2015-10-06 | Boston Scientific Scimed, Inc. | Systems, methods and devices relating to delivery of medical implants |
| AU2002254157B2 (en) | 2001-03-09 | 2006-07-27 | Boston Scientific Limited | Medical slings |
| US6666817B2 (en) | 2001-10-05 | 2003-12-23 | Scimed Life Systems, Inc. | Expandable surgical implants and methods of using them |
| WO2003105727A1 (en) | 2002-06-12 | 2003-12-24 | Scimed Life Systems, Inc. | Medical slings |
| US7824701B2 (en) | 2002-10-18 | 2010-11-02 | Ethicon, Inc. | Biocompatible scaffold for ligament or tendon repair |
| US8197837B2 (en) | 2003-03-07 | 2012-06-12 | Depuy Mitek, Inc. | Method of preparation of bioabsorbable porous reinforced tissue implants and implants thereof |
| GB0307011D0 (en) | 2003-03-27 | 2003-04-30 | Regentec Ltd | Porous matrix |
| US8226715B2 (en) | 2003-06-30 | 2012-07-24 | Depuy Mitek, Inc. | Scaffold for connective tissue repair |
| US10583220B2 (en) | 2003-08-11 | 2020-03-10 | DePuy Synthes Products, Inc. | Method and apparatus for resurfacing an articular surface |
| WO2005016184A1 (en) * | 2003-08-14 | 2005-02-24 | Scimed Life System, Inc. | Surgical slings |
| US8545386B2 (en) | 2003-08-14 | 2013-10-01 | Boston Scientific Scimed, Inc. | Surgical slings |
| AU2004279388B2 (en) | 2003-10-03 | 2010-10-14 | Boston Scientific Limited | Systems for a delivering a medical implant to an anatomical location in a patient |
| US7901461B2 (en) | 2003-12-05 | 2011-03-08 | Ethicon, Inc. | Viable tissue repair implants and methods of use |
| US11395865B2 (en) | 2004-02-09 | 2022-07-26 | DePuy Synthes Products, Inc. | Scaffolds with viable tissue |
| US8221780B2 (en) | 2004-04-20 | 2012-07-17 | Depuy Mitek, Inc. | Nonwoven tissue scaffold |
| US8657881B2 (en) * | 2004-04-20 | 2014-02-25 | Depuy Mitek, Llc | Meniscal repair scaffold |
| US8137686B2 (en) | 2004-04-20 | 2012-03-20 | Depuy Mitek, Inc. | Nonwoven tissue scaffold |
| US8628465B2 (en) | 2004-06-14 | 2014-01-14 | Boston Scientific Scimed, Inc. | Systems, methods and devices relating to implantable supportive slings |
| US8329202B2 (en) | 2004-11-12 | 2012-12-11 | Depuy Products, Inc. | System and method for attaching soft tissue to an implant |
| US7556598B2 (en) | 2005-04-04 | 2009-07-07 | Boston Scientific Scimed, Inc. | Dissolvable protective treatment for an implantable supportive sling |
| ES2536748T3 (es) | 2005-04-06 | 2015-05-28 | Boston Scientific Limited | Sistemas y dispositivos para tratar trastornos del suelo pélvico |
| EP1909687A1 (en) | 2005-07-13 | 2008-04-16 | Boston Scientific Scimed, Inc. | Snap fit sling anchor system and related methods |
| US7981023B2 (en) | 2005-07-25 | 2011-07-19 | Boston Scientific Scimed, Inc. | Elastic sling system and related methods |
| EP2974692B1 (en) | 2005-07-25 | 2019-03-13 | Boston Scientific Limited | Pelvic floor repair system |
| US8033982B2 (en) | 2005-08-03 | 2011-10-11 | Boston Scientific Scimed, Inc. | Systems, devices and methods relating to a shape resilient sling-like support for treating urinary incontinence |
| US7935046B2 (en) | 2005-08-11 | 2011-05-03 | Boston Scientific Scimed, Inc. | Systems, methods and devices relating to a removable sleeve for an implantable sling |
| US7951066B2 (en) | 2005-08-17 | 2011-05-31 | Boston Scientific Scimed, Inc. | Medical device positioning assemblies and methods |
| US7699769B2 (en) | 2005-09-01 | 2010-04-20 | Boston Scientific Scimed, Inc. | Adjustable surgical sling |
| EP3505137A1 (en) * | 2006-05-09 | 2019-07-03 | LifeCell Corporation | Reinforced biological tissue |
| US20100204775A1 (en) * | 2006-05-22 | 2010-08-12 | Tarun John Edwin | Tissue Synthetic- Biomaterial Hybrid Medical Devices |
| US8524265B2 (en) * | 2006-08-17 | 2013-09-03 | Warsaw Orthopedic, Inc. | Medical implant sheets useful for tissue regeneration |
| US20090157193A1 (en) * | 2007-12-18 | 2009-06-18 | Warsaw Orthopedic, Inc. | Tendon and Ligament Repair Sheet and Methods of Use |
| EP2271354B1 (en) | 2008-03-27 | 2017-07-05 | Cleveland Clinic Foundation | Reinforced tissue graft |
| US20130116799A1 (en) | 2008-03-27 | 2013-05-09 | The Cleveland Clinic Foundation | Reinforced tissue graft |
| CN101332134B (zh) * | 2008-07-15 | 2012-01-11 | 许和平 | I型胶原人工肌腱、韧带的制备及应用 |
| WO2010014021A1 (en) * | 2008-07-30 | 2010-02-04 | Mesynthes Limited | Tissue scaffolds derived from forestomach extracellular matrix |
| FR2937243B1 (fr) * | 2008-10-22 | 2011-11-18 | Sofradim Production | Implant de renfort |
| US8758799B2 (en) | 2010-03-24 | 2014-06-24 | Covidien Lp | Therapeutic implant |
| US8758798B2 (en) | 2010-03-24 | 2014-06-24 | Covidien Lp | Therapeutic implant |
| US8758800B2 (en) | 2010-03-24 | 2014-06-24 | Covidien Lp | Therapeutic implant |
| CN103249375A (zh) * | 2010-06-09 | 2013-08-14 | 瑞士商麦斯特医疗股份有限公司 | 抗沾粘外科入路、加固性与封闭假体 |
| FR2962646B1 (fr) | 2010-07-16 | 2012-06-22 | Sofradim Production | Prothese avec element radio-opaque |
| US9861590B2 (en) | 2010-10-19 | 2018-01-09 | Covidien Lp | Self-supporting films for delivery of therapeutic agents |
| US9017711B2 (en) | 2011-04-28 | 2015-04-28 | Warsaw Orthopedic, Inc. | Soft tissue wrap |
| US9113991B2 (en) | 2011-05-12 | 2015-08-25 | Boston Scientific Scimed, Inc. | Anchors for bodily implants and methods for anchoring bodily implants into a patient's body |
| US9636201B2 (en) | 2011-05-12 | 2017-05-02 | Boston Scientific Scimed, Inc. | Delivery members for delivering an implant into a body of a patient |
| FR2977790B1 (fr) | 2011-07-13 | 2013-07-19 | Sofradim Production | Prothese pour hernie ombilicale |
| US8579924B2 (en) | 2011-07-26 | 2013-11-12 | Covidien Lp | Implantable devices including a mesh and a pivotable film |
| GB201113303D0 (en) | 2011-08-02 | 2011-09-14 | Xiros Ltd | Connective tissue repair pad |
| US9782957B2 (en) | 2011-08-24 | 2017-10-10 | Covidien Lp | Medical device films |
| US8932621B2 (en) | 2011-10-25 | 2015-01-13 | Covidien Lp | Implantable film/mesh composite |
| US9005308B2 (en) | 2011-10-25 | 2015-04-14 | Covidien Lp | Implantable film/mesh composite for passage of tissue therebetween |
| US9179994B2 (en) | 2011-10-25 | 2015-11-10 | Covidien Lp | Implantable film/mesh composite |
| US10206769B2 (en) | 2012-03-30 | 2019-02-19 | Covidien Lp | Implantable devices including a film providing folding characteristics |
| FR2992662B1 (fr) | 2012-06-28 | 2014-08-08 | Sofradim Production | Tricot avec picots |
| FR2992547B1 (fr) | 2012-06-29 | 2015-04-24 | Sofradim Production | Prothese pour hernie |
| GB201301784D0 (en) | 2013-02-01 | 2013-03-20 | Xiros Ltd | Connective tissue repair technology |
| US10548705B2 (en) * | 2014-12-22 | 2020-02-04 | Aroa Biosurgery Limited | Laminated tissue graft product |
| US10888407B2 (en) * | 2015-12-11 | 2021-01-12 | Poly-Med, Inc. | Method for forming a multi-layer construct |
| US11484401B2 (en) | 2016-02-01 | 2022-11-01 | Medos International Sarl | Tissue augmentation scaffolds for use in soft tissue fixation repair |
| US11357495B2 (en) | 2016-02-01 | 2022-06-14 | Medos International Sarl | Tissue augmentation scaffolds for use with soft tissue fixation repair systems and methods |
| CN105664257B (zh) * | 2016-03-01 | 2019-02-19 | 上海卓阮医疗科技有限公司 | 一种修复区稳固的复合软组织修复材料 |
| CN109331229A (zh) * | 2018-11-28 | 2019-02-15 | 上海白衣缘生物工程有限公司 | 一种医用生物线、医用生物修补网片及其制备方法 |
Family Cites Families (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1405886A (en) | 1971-11-01 | 1975-09-10 | Astra Laekemedel Ab | Synergistic compositions comprising penicillin and cephalosporin antibiotics |
| US4352463A (en) | 1979-01-18 | 1982-10-05 | Leisure Lawn, Inc. | Motorized combination wet and dry lawn treatment spreader |
| US4902508A (en) | 1988-07-11 | 1990-02-20 | Purdue Research Foundation | Tissue graft composition |
| US5281422A (en) | 1991-09-24 | 1994-01-25 | Purdue Research Foundation | Graft for promoting autogenous tissue growth |
| US5800537A (en) * | 1992-08-07 | 1998-09-01 | Tissue Engineering, Inc. | Method and construct for producing graft tissue from an extracellular matrix |
| US5275826A (en) | 1992-11-13 | 1994-01-04 | Purdue Research Foundation | Fluidized intestinal submucosa and its use as an injectable tissue graft |
| DE4302709C1 (de) * | 1993-02-01 | 1994-07-28 | Kirsch Axel | Abdeckeinrichtung mit Abdeckmembran |
| EP0713364A4 (en) * | 1993-08-13 | 1996-12-27 | Shalaby W Shalaby | MICROPOROUS POLYMERIC FOAMS AND MICROTEXTURED SURFACES |
| DE69426414T2 (de) * | 1993-09-24 | 2001-05-03 | Takiron Co | Implantatmaterial |
| GB2282328B (en) * | 1993-09-29 | 1997-10-08 | Johnson & Johnson Medical | Absorbable structures for ligament and tendon repair |
| US5759205A (en) * | 1994-01-21 | 1998-06-02 | Brown University Research Foundation | Negatively charged polymeric electret implant |
| CA2186374A1 (en) * | 1994-04-29 | 1995-11-09 | William Carl Bruchman | Improved blood contact surfaces employing natural subendothelial matrix and method for making and using the same |
| DE69527141T2 (de) * | 1994-04-29 | 2002-11-07 | Scimed Life Systems Inc | Stent mit kollagen |
| US5891558A (en) * | 1994-11-22 | 1999-04-06 | Tissue Engineering, Inc. | Biopolymer foams for use in tissue repair and reconstruction |
| US5709934A (en) * | 1994-11-22 | 1998-01-20 | Tissue Engineering, Inc. | Bipolymer foams having extracellular matrix particulates |
| GB9721585D0 (en) * | 1997-10-10 | 1997-12-10 | Geistlich Soehne Ag | Chemical product |
| US5855613A (en) * | 1995-10-13 | 1999-01-05 | Islet Sheet Medical, Inc. | Retrievable bioartificial implants having dimensions allowing rapid diffusion of oxygen and rapid biological response to physiological change |
| US5965125A (en) * | 1995-10-25 | 1999-10-12 | Transkaryotic Therapies, Inc. | Hybrid matrix implants and explants |
| US5755791A (en) * | 1996-04-05 | 1998-05-26 | Purdue Research Foundation | Perforated submucosal tissue graft constructs |
| US5788625A (en) * | 1996-04-05 | 1998-08-04 | Depuy Orthopaedics, Inc. | Method of making reconstructive SIS structure for cartilaginous elements in situ |
| FR2766717B1 (fr) * | 1997-08-01 | 2000-06-09 | Cogent Sarl | Prothese composite pour la prevention des adherences post-chirurgicales et son procede d'obtention |
| US6409764B1 (en) * | 1998-12-03 | 2002-06-25 | Charles F. White | Methods and articles for regenerating bone or peridontal tissue |
-
2002
- 2002-07-15 WO PCT/US2002/022364 patent/WO2003007847A1/en active Application Filing
- 2002-07-15 AU AU2002320517A patent/AU2002320517B2/en not_active Ceased
- 2002-07-15 EP EP02750038A patent/EP1416878A4/en not_active Withdrawn
- 2002-07-15 JP JP2003513456A patent/JP4197159B2/ja not_active Expired - Fee Related
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011072797A (ja) * | 2009-10-01 | 2011-04-14 | Tyco Healthcare Group Lp | メッシュインプラント |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1416878A4 (en) | 2007-04-18 |
| EP1416878A1 (en) | 2004-05-12 |
| JP2004522555A (ja) | 2004-07-29 |
| WO2003007847A1 (en) | 2003-01-30 |
| AU2002320517B2 (en) | 2008-03-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4197159B2 (ja) | 混成の生物学的合成の生体吸収性支持骨格材料 | |
| JP2004522555A5 (ja) | ||
| US7569233B2 (en) | Hybrid biologic-synthetic bioabsorbable scaffolds | |
| US8366787B2 (en) | Hybrid biologic-synthetic bioabsorbable scaffolds | |
| US20050249772A1 (en) | Hybrid biologic-synthetic bioabsorbable scaffolds | |
| US20220331491A1 (en) | Scaffolds with viable tissue | |
| US7914808B2 (en) | Hybrid biologic/synthetic porous extracellular matrix scaffolds | |
| US7163563B2 (en) | Unitary surgical device and method | |
| US7871440B2 (en) | Unitary surgical device and method | |
| US8025896B2 (en) | Porous extracellular matrix scaffold and method | |
| JP4194774B2 (ja) | 生体プロテーゼ装置 | |
| EP1593400A1 (en) | Hybrid biologic-synthetic bioabsorbable scaffolds | |
| US7201917B2 (en) | Porous delivery scaffold and method | |
| JP4201134B2 (ja) | 軟骨修復装置および方法 | |
| AU2002320517A1 (en) | Hybrid biologic-synthetic bioabsorable scaffolds | |
| JP4294474B2 (ja) | 半月板再生装置 | |
| US20040166169A1 (en) | Porous extracellular matrix scaffold and method | |
| AU2002316694A1 (en) | Hybrid biologic/synthetic porous extracellular matrix scaffolds | |
| EP1618903A1 (en) | Hybrid biologic-synthetic bioabsorbable scaffolds |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20041126 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20071127 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20080115 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20080415 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20080422 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20080714 |
|
| A524 | Written submission of copy of amendment under section 19 (pct) |
Free format text: JAPANESE INTERMEDIATE CODE: A524 Effective date: 20080714 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20080909 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20080910 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20080924 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20111010 Year of fee payment: 3 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| LAPS | Cancellation because of no payment of annual fees |