JP2015513349A - 組織修復のための基礎材システム - Google Patents
組織修復のための基礎材システム Download PDFInfo
- Publication number
- JP2015513349A JP2015513349A JP2014556808A JP2014556808A JP2015513349A JP 2015513349 A JP2015513349 A JP 2015513349A JP 2014556808 A JP2014556808 A JP 2014556808A JP 2014556808 A JP2014556808 A JP 2014556808A JP 2015513349 A JP2015513349 A JP 2015513349A
- Authority
- JP
- Japan
- Prior art keywords
- base material
- fibers
- tissue
- support structure
- electrospun
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
-
- D—TEXTILES; PAPER
- D04—BRAIDING; LACE-MAKING; KNITTING; TRIMMINGS; NON-WOVEN FABRICS
- D04H—MAKING TEXTILE FABRICS, e.g. FROM FIBRES OR FILAMENTARY MATERIAL; FABRICS MADE BY SUCH PROCESSES OR APPARATUS, e.g. FELTS, NON-WOVEN FABRICS; COTTON-WOOL; WADDING ; NON-WOVEN FABRICS FROM STAPLE FIBRES, FILAMENTS OR YARNS, BONDED WITH AT LEAST ONE WEB-LIKE MATERIAL DURING THEIR CONSOLIDATION
- D04H1/00—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres
- D04H1/40—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres from fleeces or layers composed of fibres without existing or potential cohesive properties
- D04H1/42—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres from fleeces or layers composed of fibres without existing or potential cohesive properties characterised by the use of certain kinds of fibres insofar as this use has no preponderant influence on the consolidation of the fleece
- D04H1/4391—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres from fleeces or layers composed of fibres without existing or potential cohesive properties characterised by the use of certain kinds of fibres insofar as this use has no preponderant influence on the consolidation of the fleece characterised by the shape of the fibres
- D04H1/43918—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres from fleeces or layers composed of fibres without existing or potential cohesive properties characterised by the use of certain kinds of fibres insofar as this use has no preponderant influence on the consolidation of the fleece characterised by the shape of the fibres nonlinear fibres, e.g. crimped or coiled fibres
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/04—Hollow or tubular parts of organs, e.g. bladders, tracheae, bronchi or bile ducts
- A61F2/06—Blood vessels
- A61F2/07—Stent-grafts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/18—Macromolecular materials obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/22—Polypeptides or derivatives thereof, e.g. degradation products
- A61L27/227—Other specific proteins or polypeptides not covered by A61L27/222, A61L27/225 or A61L27/24
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/28—Materials for coating prostheses
- A61L27/34—Macromolecular materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/507—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials for artificial blood vessels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/54—Biologically active materials, e.g. therapeutic substances
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/56—Porous materials, e.g. foams or sponges
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/58—Materials at least partially resorbable by the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/08—Materials for coatings
- A61L31/10—Macromolecular materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/146—Porous materials, e.g. foams or sponges
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/148—Materials at least partially resorbable by the body
-
- D—TEXTILES; PAPER
- D01—NATURAL OR MAN-MADE THREADS OR FIBRES; SPINNING
- D01D—MECHANICAL METHODS OR APPARATUS IN THE MANUFACTURE OF ARTIFICIAL FILAMENTS, THREADS, FIBRES, BRISTLES OR RIBBONS
- D01D5/00—Formation of filaments, threads, or the like
- D01D5/0007—Electro-spinning
- D01D5/0015—Electro-spinning characterised by the initial state of the material
- D01D5/003—Electro-spinning characterised by the initial state of the material the material being a polymer solution or dispersion
-
- D—TEXTILES; PAPER
- D04—BRAIDING; LACE-MAKING; KNITTING; TRIMMINGS; NON-WOVEN FABRICS
- D04H—MAKING TEXTILE FABRICS, e.g. FROM FIBRES OR FILAMENTARY MATERIAL; FABRICS MADE BY SUCH PROCESSES OR APPARATUS, e.g. FELTS, NON-WOVEN FABRICS; COTTON-WOOL; WADDING ; NON-WOVEN FABRICS FROM STAPLE FIBRES, FILAMENTS OR YARNS, BONDED WITH AT LEAST ONE WEB-LIKE MATERIAL DURING THEIR CONSOLIDATION
- D04H1/00—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres
- D04H1/70—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres characterised by the method of forming fleeces or layers, e.g. reorientation of fibres
- D04H1/72—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres characterised by the method of forming fleeces or layers, e.g. reorientation of fibres the fibres being randomly arranged
- D04H1/728—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres characterised by the method of forming fleeces or layers, e.g. reorientation of fibres the fibres being randomly arranged by electro-spinning
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2210/00—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2210/0004—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof bioabsorbable
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2210/00—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2210/0076—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof multilayered, e.g. laminated structures
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0063—Three-dimensional shapes
- A61F2230/0069—Three-dimensional shapes cylindrical
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/20—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing organic materials
- A61L2300/21—Acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/40—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a specific therapeutic activity or mode of action
- A61L2300/412—Tissue-regenerating or healing or proliferative agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/22—Materials or treatment for tissue regeneration for reconstruction of hollow organs, e.g. bladder, esophagus, urether, uterus
-
- D—TEXTILES; PAPER
- D01—NATURAL OR MAN-MADE THREADS OR FIBRES; SPINNING
- D01D—MECHANICAL METHODS OR APPARATUS IN THE MANUFACTURE OF ARTIFICIAL FILAMENTS, THREADS, FIBRES, BRISTLES OR RIBBONS
- D01D5/00—Formation of filaments, threads, or the like
- D01D5/0007—Electro-spinning
- D01D5/0061—Electro-spinning characterised by the electro-spinning apparatus
- D01D5/0076—Electro-spinning characterised by the electro-spinning apparatus characterised by the collecting device, e.g. drum, wheel, endless belt, plate or grid
Abstract
Description
基礎材の基礎材処理は、基礎材を反応性ガスにより形成されるプラズマにさらすことを含んでもよい。反応性ガスは、酸素、窒素、アルゴン、アンモニアまたはその組み合わせを含んでもよい。
PTFE包覆ステント(心房I−cast,9mm)を、7Fの導入器シースを通して挿入した。バルーンを10秒間15ATMまで膨張し、その後収縮させ取り除いた。それから第2のバルーン(パワーフレックスP3 CTAカテーテル12mm×4mm)を置き、10秒間12ATMまで近位および遠位に膨張させた。6FのMP−1カテーテルを再度置き、血管造影を使用してAAAが適切に閉塞したかどうかを判断した。それから、大腿動脈が絹縫合素材を用いて連結されたとき、ガイドワイヤおよびカテーテル、続いてシースを取り除いた。アクセス部位を3−0Vicrylで閉じ、動物を回復させた。
Claims (27)
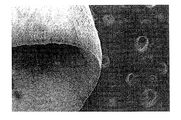
- 拡張可能な基礎材を備え、前記基礎材は不織繊維で構成され、前記基礎材は実質的に曲線状の繊維を有する第1の面、および実質的に直線状の繊維を有する第2の面を有し、
前記基礎材が哺乳類内に位置決められるとき、前記基礎材の前記第1の面は細胞の付着に適切な面を提供し、一方で前記基礎材の前記第2の面は細胞の侵入および組織化を促進する、
組織修復装置。 - 前記装置が動脈瘤の修復のために構成される、請求項1に記載の装置。
- 前記装置が、内腔組織構造、組織内の空洞または半空洞のスペースの修復のために構成される、請求項1に記載の装置。
- 前記繊維が電界紡糸される、請求項1に記載の装置。
- 前記繊維が、押出、延伸、および/または引抜技術を用いて製造される、請求項1に記載の装置。
- 前記繊維が、生分解性および/または生体吸収性素材で構成される、請求項1に記載の装置。
- 基礎材が、1つまたは複数のポリ(α−ヒドロキシエステル)で構成される、請求項1に記載の装置。
- 基礎材がポリカプロラクトンで構成される、請求項1に記載の装置。
- 前記基礎材が、生分解性および/または生体吸収性の天然ポリマーで構成される、請求項1に記載の装置。
- 前記基礎材が、エラスチン、コラーゲン、DNA、RNA、グルコサミノグルカンまたはその混合物で構成される、請求項1に記載の装置。
- 前記第1の面が凹面であり、前記第2の面が凸面である、請求項1に記載の装置。
- 前記基礎材が支持構造により支持される、請求項1に記載の装置。
- 前記支持構造が拡張可能である、請求項8に記載の装置。
- 前記支持構造が拡張可能なステントである、請求項8に記載の装置。
- 前記支持構造が柔軟な構造である、請求項8に記載の装置。
- 前記支持構造が再配置可能な構造である、請求項8に記載の装置。
- 前記支持構造が生体吸収性および/または生分解性である、請求項8に記載の装置。
- 前記基礎材が、生分解性素材および/または生体吸収性素材化合物から電界紡糸される不織マイクロファイバで構成される、請求項1に記載の装置。
- 前記基礎材が、生分解性素材および/または生体吸収性素材化合物から電界紡糸される不織ナノファイバで構成される、請求項1に記載の装置。
- 前記基礎材が実質的にチューブ状である、請求項1に記載の装置。
- 前記基礎材が、医療装置の少なくとも一部により支持される、請求項1に記載の装置。
- 前記基礎材が、支持構造に縫合または機械的に固定される、請求項1に記載の装置。
- 前記基礎材が支持構造に化学的に接着される、請求項1に記載の装置。
- 前記基礎材が、支持構造上に直接または間接的に電界紡糸される、請求項1に記載の装置。
- 前記支持構造が、電界紡糸された前記基礎材内に組み込まれる、請求項1に記載の装置。
- 装置を内腔組織構造内に挿入し、前記装置は拡張可能な基礎材を備え、前記基礎材は生分解性素材および/または生体吸収性素材から電界紡糸された不織繊維で構成され、前記基礎材は、実質的に曲線状の繊維を有する第1の面、および実質的に直線状の繊維を有する第2の面を備えるステップと、
前記基礎材が前記内腔組織構造の少なくとも一部と接触するように、前記基礎材を拡張し、前記基礎材の第1の面が細胞の付着に適切な面を提供し、一方で前記第2の表面が細胞の侵入および組織化を促進するように前記基礎材が位置決められるステップと、
前記内腔組織構造内に前記装置を固定するステップと、を含む、
組織修復方法。 - 拡張可能な基礎材を備え、前記基礎材は、実質的に曲線状の繊維を有する面、および実質的に直線状の繊維を有する対向する面で構成される、
医学的状態を治療する装置。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201261598125P | 2012-02-13 | 2012-02-13 | |
| US61/598,125 | 2012-02-13 | ||
| PCT/US2013/025829 WO2013122981A1 (en) | 2012-02-13 | 2013-02-13 | Scaffold system for tissue repair |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2015513349A true JP2015513349A (ja) | 2015-05-11 |
Family
ID=48982851
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2014556808A Pending JP2015513349A (ja) | 2012-02-13 | 2013-02-13 | 組織修復のための基礎材システム |
Country Status (8)
| Country | Link |
|---|---|
| US (4) | US9259334B2 (ja) |
| EP (1) | EP2814528A4 (ja) |
| JP (1) | JP2015513349A (ja) |
| KR (1) | KR20140138709A (ja) |
| AU (2) | AU2013221777B2 (ja) |
| CA (1) | CA2867167A1 (ja) |
| IN (1) | IN2014DN07298A (ja) |
| WO (1) | WO2013122981A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016523604A (ja) * | 2013-05-31 | 2016-08-12 | フランシスコ セリーノ | 血管組織の修復および操作のための方法および構成物 |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2867167A1 (en) * | 2012-02-13 | 2013-08-22 | Board Of Regents, The University Of Texas System | Scaffold system for tissue repair |
| US10149923B2 (en) * | 2013-01-15 | 2018-12-11 | Tepha, Inc. | Implants for soft and hard tissue regeneration |
| US10201640B2 (en) * | 2013-03-13 | 2019-02-12 | Tepha, Inc. | Ultrafine electrospun fibers of poly-4-hydroxybutyrate and copolymers thereof |
| US9302029B2 (en) | 2013-10-31 | 2016-04-05 | Tepha, Inc. | Pultrusion of poly-4-hydroxybutyrate and copolymers thereof |
| US11000285B2 (en) * | 2013-12-17 | 2021-05-11 | 3Dt Holdings, Llc | Luminal grafts and methods of making and using the same |
| US10626521B2 (en) | 2014-12-11 | 2020-04-21 | Tepha, Inc. | Methods of manufacturing mesh sutures from poly-4-hydroxybutyrate and copolymers thereof |
| EP3230500A1 (en) | 2014-12-11 | 2017-10-18 | Tepha, Inc. | Methods of orienting multifilament yarn and monofilaments of poly-4-hydroxybutyrate and copolymers thereof |
| WO2019112184A1 (ko) | 2017-12-05 | 2019-06-13 | 한국생산기술연구원 | 3차원 섬유형 스캐폴드 |
| EP4100074A4 (en) * | 2020-02-06 | 2023-10-18 | Board of Trustees of the University of Arkansas | EXPANDABLE BONE AND TISSUE REGENERATION SYSTEM AND ITS APPLICATIONS |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008532643A (ja) * | 2005-03-09 | 2008-08-21 | ザ ユニバーシティー オブ テネシー リサーチ ファウンデーション | 障壁ステントおよびその使用 |
| US20090018643A1 (en) * | 2007-06-11 | 2009-01-15 | Nanovasc, Inc. | Stents |
| WO2011113001A2 (en) * | 2010-03-11 | 2011-09-15 | Board Of Regents Of The University Of Texas System | Scaffold system to repair cardiovascular conditions |
Family Cites Families (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4329743A (en) | 1979-04-27 | 1982-05-18 | College Of Medicine And Dentistry Of New Jersey | Bio-absorbable composite tissue scaffold |
| US4883618A (en) * | 1983-02-02 | 1989-11-28 | Minnesota Mining And Manufacturing Company | Absorbable nerve repair device and method |
| US7611533B2 (en) | 1995-06-07 | 2009-11-03 | Cook Incorporated | Coated implantable medical device |
| US20020090725A1 (en) * | 2000-11-17 | 2002-07-11 | Simpson David G. | Electroprocessed collagen |
| US7615373B2 (en) * | 1999-02-25 | 2009-11-10 | Virginia Commonwealth University Intellectual Property Foundation | Electroprocessed collagen and tissue engineering |
| US20050271701A1 (en) | 2000-03-15 | 2005-12-08 | Orbus Medical Technologies, Inc. | Progenitor endothelial cell capturing with a drug eluting implantable medical device |
| WO2002007749A2 (en) * | 2000-07-21 | 2002-01-31 | Board Of Regents, The University Of Texas System | Device providing regulated growth factor delivery for the regeneration of peripheral nerves |
| US20030100945A1 (en) * | 2001-11-23 | 2003-05-29 | Mindguard Ltd. | Implantable intraluminal device and method of using same in treating aneurysms |
| US6790455B2 (en) * | 2001-09-14 | 2004-09-14 | The Research Foundation At State University Of New York | Cell delivery system comprising a fibrous matrix and cells |
| JP4605985B2 (ja) * | 2002-12-27 | 2011-01-05 | ニプロ株式会社 | 神経再生誘導管 |
| US20050038498A1 (en) * | 2003-04-17 | 2005-02-17 | Nanosys, Inc. | Medical device applications of nanostructured surfaces |
| WO2005003300A2 (en) | 2003-06-04 | 2005-01-13 | University Of South Carolina | Tissue scaffold having aligned fibrils, apparatus and method for producing same, and methods of using same |
| CA2536042A1 (en) * | 2003-11-10 | 2005-06-02 | Angiotech International Ag | Medical implants and anti-scarring agents |
| CA2583469C (en) * | 2004-10-06 | 2013-03-19 | Research Foundation Of State University Of New York | High flux and low fouling filtration media |
| US20060085063A1 (en) * | 2004-10-15 | 2006-04-20 | Shastri V P | Nano- and micro-scale engineering of polymeric scaffolds for vascular tissue engineering |
| US8871237B2 (en) * | 2005-04-04 | 2014-10-28 | Technion Research & Development Foundation Limited | Medical scaffold, methods of fabrication and using thereof |
| US8282873B2 (en) * | 2006-01-03 | 2012-10-09 | Victor Barinov | Controlled electrospinning of fibers |
| US20080220042A1 (en) * | 2006-01-27 | 2008-09-11 | The Regents Of The University Of California | Biomolecule-linked biomimetic scaffolds |
| EP2599858A3 (en) * | 2006-01-27 | 2013-09-18 | The Regents of The University of California | Biomimetic scaffolds |
| WO2008024640A2 (en) * | 2006-08-10 | 2008-02-28 | University Of Pittsburgh-Of The Commonwealth System Of Higher Education | Biodegradable elastomeric scaffolds containing microintegrated cells |
| EP2066363A2 (en) * | 2006-09-15 | 2009-06-10 | Boston Scientific Limited | Endoprosthesis containing magnetic induction particles |
| US20080112998A1 (en) * | 2006-11-14 | 2008-05-15 | Hongjun Wang | Innovative bottom-up cell assembly approach to three-dimensional tissue formation using nano-or micro-fibers |
| US9011754B2 (en) * | 2006-12-05 | 2015-04-21 | Nanyang Technological University | Manufacturing three-dimensional scaffolds using electrospinning at low temperatures |
| WO2008118228A2 (en) * | 2006-12-05 | 2008-10-02 | Stonybrook Water Purification | Articles comprising a fibrous support |
| US8753391B2 (en) * | 2007-02-12 | 2014-06-17 | The Trustees Of Columbia University In The City Of New York | Fully synthetic implantable multi-phased scaffold |
| US9163338B2 (en) * | 2007-04-11 | 2015-10-20 | Drexel University | Fibrous mats containing chitosan nanofibers |
| US10155881B2 (en) | 2007-05-30 | 2018-12-18 | Abbott Cardiovascular Systems Inc. | Substituted polycaprolactone for coating |
| US8790684B2 (en) * | 2007-10-31 | 2014-07-29 | Cordis Corporation | Vascular closure device |
| WO2009099570A2 (en) * | 2008-02-01 | 2009-08-13 | Wake Forest University Health Sciences | Aligned scaffolding system for skeletal muscle regeneration |
| US9029149B2 (en) * | 2008-07-31 | 2015-05-12 | Carnegie Mellon University | Methods, apparatus, and systems for fabrication of polymeric nano- and micro-fibers in aligned configurations |
| EP2962704A1 (en) * | 2008-10-07 | 2016-01-06 | Nanonerve, Inc. | Multilayer fibrous polymer scaffolds, methods of production and methods of use |
| AU2009320446B2 (en) * | 2008-11-25 | 2015-08-20 | The Henry M. Jackson Foundation For The Advancement Of Military Medicine, Inc. | Regenerative tissue grafts and methods of making same |
| MX2011009282A (es) * | 2009-03-10 | 2011-10-28 | Medprin Regenerative Medical Technologies Co Ltd | Duramadre artificial y su metodo de fabricacion. |
| US9476026B2 (en) * | 2009-03-12 | 2016-10-25 | New Jersey Institute Of Technology | Method of tissue repair using a piezoelectric scaffold |
| WO2010127119A2 (en) * | 2009-04-29 | 2010-11-04 | The Regents Of The University Of Michigan | Multiphasic microfibers for spatially guided cell growth |
| US9107739B2 (en) * | 2009-11-25 | 2015-08-18 | Drexel University | Small diameter vascular graft produced by a hybrid method |
| US10292808B2 (en) | 2010-06-07 | 2019-05-21 | Q3 Medical Devices Limited | Device and method for management of aneurism, perforation and other vascular abnormalities |
| WO2011156586A2 (en) * | 2010-06-09 | 2011-12-15 | Trustees Of Tufts College | Multilayered silk scaffolds for meniscus tissue engineering |
| WO2011163232A2 (en) * | 2010-06-21 | 2011-12-29 | University Of South Florida | Polypeptide electrospun nanofibrils of defined composition |
| WO2012014205A1 (en) * | 2010-07-27 | 2012-02-02 | Technion Research & Development Foundation Ltd. | Isolated mesenchymal progenitor cells and extracellular matrix produced thereby |
| US20130180917A1 (en) * | 2010-08-23 | 2013-07-18 | The Research Foundation Of State University Of New York | High flux microfiltration membranes with virus and metal ion adsorption capability for liquid purification |
| WO2012048188A1 (en) * | 2010-10-07 | 2012-04-12 | Drixel University | Electrospun mineralized chitosan nanofibers crosslinked with genipin for bone tissue enginering |
| IN2014KN01509A (ja) * | 2012-01-27 | 2015-10-23 | Zeus Ind Products Inc | |
| CA2867167A1 (en) * | 2012-02-13 | 2013-08-22 | Board Of Regents, The University Of Texas System | Scaffold system for tissue repair |
-
2013
- 2013-02-13 CA CA2867167A patent/CA2867167A1/en not_active Abandoned
- 2013-02-13 EP EP13748603.1A patent/EP2814528A4/en not_active Withdrawn
- 2013-02-13 JP JP2014556808A patent/JP2015513349A/ja active Pending
- 2013-02-13 US US13/766,530 patent/US9259334B2/en not_active Expired - Fee Related
- 2013-02-13 KR KR1020147025331A patent/KR20140138709A/ko not_active Application Discontinuation
- 2013-02-13 WO PCT/US2013/025829 patent/WO2013122981A1/en active Application Filing
- 2013-02-13 AU AU2013221777A patent/AU2013221777B2/en not_active Ceased
- 2013-02-13 IN IN7298DEN2014 patent/IN2014DN07298A/en unknown
-
2016
- 2016-02-15 US US15/043,968 patent/US9849007B2/en not_active Expired - Fee Related
-
2017
- 2017-01-12 AU AU2017200200A patent/AU2017200200B2/en not_active Ceased
- 2017-12-26 US US15/854,314 patent/US10376394B2/en not_active Expired - Fee Related
-
2019
- 2019-06-18 US US16/444,518 patent/US20190321202A1/en not_active Abandoned
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008532643A (ja) * | 2005-03-09 | 2008-08-21 | ザ ユニバーシティー オブ テネシー リサーチ ファウンデーション | 障壁ステントおよびその使用 |
| US20090018643A1 (en) * | 2007-06-11 | 2009-01-15 | Nanovasc, Inc. | Stents |
| WO2011113001A2 (en) * | 2010-03-11 | 2011-09-15 | Board Of Regents Of The University Of Texas System | Scaffold system to repair cardiovascular conditions |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016523604A (ja) * | 2013-05-31 | 2016-08-12 | フランシスコ セリーノ | 血管組織の修復および操作のための方法および構成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| IN2014DN07298A (ja) | 2015-04-24 |
| AU2013221777A1 (en) | 2014-09-18 |
| AU2013221777B2 (en) | 2016-10-13 |
| WO2013122981A1 (en) | 2013-08-22 |
| KR20140138709A (ko) | 2014-12-04 |
| US9259334B2 (en) | 2016-02-16 |
| CA2867167A1 (en) | 2013-08-22 |
| US10376394B2 (en) | 2019-08-13 |
| EP2814528A1 (en) | 2014-12-24 |
| US20180200084A1 (en) | 2018-07-19 |
| AU2017200200B2 (en) | 2018-07-12 |
| US20130218253A1 (en) | 2013-08-22 |
| EP2814528A4 (en) | 2015-09-30 |
| US9849007B2 (en) | 2017-12-26 |
| AU2017200200A1 (en) | 2017-02-02 |
| US20160287415A1 (en) | 2016-10-06 |
| US20190321202A1 (en) | 2019-10-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10376394B2 (en) | Scaffold system for tissue repair | |
| US11850141B2 (en) | Bioerodible wraps and uses therefor | |
| AU2011226695B2 (en) | Scaffold system to repair cardiovascular conditions | |
| EP2111185B1 (en) | Bioerodible wraps and uses therefor | |
| Li et al. | Extrahepatic bile duct regeneration in pigs using collagen scaffolds loaded with human collagen-binding bFGF | |
| Yang et al. | VEGF combined with DAPT promotes tissue regeneration and remodeling in vascular grafts |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20160215 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160222 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20161222 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20170110 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20170407 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20170801 |